Anti-nkg2a Antibodies And Uses Thereof
Spee; Petrus Johannes Louis ; et al.
U.S. patent application number 16/397271 was filed with the patent office on 2019-08-15 for anti-nkg2a antibodies and uses thereof. The applicant listed for this patent is NOVO NORDISK A/S. Invention is credited to Soeren Berg Padkaer, Petrus Johannes Louis Spee.
| Application Number | 20190248896 16/397271 |
| Document ID | / |
| Family ID | 38626279 |
| Filed Date | 2019-08-15 |











View All Diagrams
| United States Patent Application | 20190248896 |
| Kind Code | A1 |
| Spee; Petrus Johannes Louis ; et al. | August 15, 2019 |
ANTI-NKG2A ANTIBODIES AND USES THEREOF
Abstract
Described herein are anti-NKG2A antibodies suitable for human therapy, including humanized versions of murine anti-NKG2A antibody Z270, as well as related methods and materials for producing and using such antibodies. Exemplary complementarity-determining regions (CDRs) sequences and sites for optional amino acid back-substitutions in framework region (FR) and/or CDRs of such antibodies are also described.
| Inventors: | Spee; Petrus Johannes Louis; (Alleroed, DK) ; Padkaer; Soeren Berg; (Vaerloese, DK) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 38626279 | ||||||||||
| Appl. No.: | 16/397271 | ||||||||||
| Filed: | April 29, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15624882 | Jun 16, 2017 | |||
| 16397271 | ||||
| 14548327 | Nov 20, 2014 | 9683041 | ||
| 15624882 | ||||
| 13477888 | May 22, 2012 | 8901283 | ||
| 14548327 | ||||
| 12305683 | May 26, 2009 | 8206709 | ||
| PCT/EP2007/056485 | Jun 28, 2007 | |||
| 13477888 | ||||
| 60818550 | Jul 5, 2006 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C07K 2317/56 20130101; A61P 31/12 20180101; C07K 2317/24 20130101; A61K 2039/505 20130101; C07K 16/2803 20130101; C07K 16/2851 20130101; A61P 35/00 20180101; C07K 16/468 20130101; C07K 2317/31 20130101; C07K 2317/76 20130101; A61P 29/00 20180101; A61P 37/00 20180101; C07K 2317/565 20130101; A61P 37/08 20180101; C07K 2317/92 20130101; A61P 37/06 20180101; C07K 2317/73 20130101 |
| International Class: | C07K 16/28 20060101 C07K016/28; C07K 16/46 20060101 C07K016/46 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Jun 30, 2006 | EP | 06116429.9 |
Claims
1-18. (canceled)
19. An isolated antibody that specifically binds NKG2A, comprising antigen-binding residues from the complementarity-determining regions (CDRs) of murine antibody Z270 and human acceptor framework sequences, wherein said antibody comprises a VL domain that is at least 90% identical to SEQ ID NO: 4 and a VH domain that is at least 90% identical to SEQ ID NO: 5, and wherein said antibody is more effective than an antibody comprising murine antibody Z270 variable light (VL) (SEQ ID NO: 1) and VH (SEQ ID NO: 2) sequences in potentiating the cytotoxic activity of a CD94/NKG2A-expressing cytotoxic lymphocyte.
20. The antibody of claim 19, comprising a CDR-H1 corresponding to residues 31-35 of SEQ ID NO:5 and a CDR-H3 corresponding to residues 95-102 of SEQ ID NO:5, wherein the CDR-H2 comprises residues 50-59 of SEQ ID NO:5, wherein the residue numbering is referenced with respect to Kabat numbering.
21. The antibody of claim 19, wherein (a) the amino acid at position 5 of the VH domain is V or Q; (b) the amino acid at position 69 of the VH domain is M or L; (c) the amino acid at position 71 of the VH domain is T or V; (d) the amino acid at position 73 of the VH domain is T or K; or (e) the amino acid at position 75 of the VH domain is T or S, wherein the residue numbering is referenced with respect to Kabat numbering.
22. The antibody of claim 19, wherein the VH domain human acceptor framework sequences are free of any back-mutations.
23. The antibody of claim 20, wherein the VH domain comprises the sequence of SEQ ID NO:5.
24. The antibody of claim 23, comprising a CDR-L1 corresponding to residues 24-34 of SEQ ID NO:4, a CDR-L2 corresponding to residues 50-56 of SEQ ID NO:4, and a CDR-L3 corresponding to residues 89-97 of SEQ ID NO:4, wherein the residue numbering is referenced with respect to Kabat numbering.
25. The antibody of claim 24, wherein the VL domain comprises SEQ ID NO:4.
26. The antibody of claim 19, which is an IgG4 antibody.
27. The antibody of claim 19, which is an antibody fragment or a multispecific antibody.
28. The antibody of claim 19, wherein the antibody VH domain comprises residues D52, D54, F99, T(100C), and W(100F) from the VH CDRs of murine antibody Z270, wherein the residue numbering is referenced with respect to Kabat numbering.
29. The antibody of claim 28, wherein the antibody VH domain comprises residues N35, Y53, E56, D98, V(100A), and L(100D) from the VH CDRs of murine antibody Z270, wherein the residue numbering is referenced with respect to Kabat numbering.
30. The antibody of claim 29, comprising a CDR-H1 corresponding to residues 31-35 of SEQ ID NO: 5 and a CDR-H3 corresponding to residues 95-102 of SEQ ID NO: 5, wherein the CDR-H2 comprises residues 50-59 of SEQ ID NO: 5, wherein the residue numbering is referenced with respect to Kabat numbering.
31. The antibody of claim 19, comprising a CDR-H1 corresponding to residues 31-35 of SEQ ID NO: 5 and a CDR-H3 corresponding to residues 95-102 of SEQ ID NO: 5, wherein the CDR-H2 comprises residues 50-59 of SEQ ID NO: 5, wherein the residue numbering is referenced with respect to Kabat numbering.
32. A method for producing an anti-NKG2A antibody, comprising culturing a host cell comprising a nucleic acid encoding the anti-NKG2A antibody of claim 19 so that the nucleic acid is expressed and the antibody produced.
33. A pharmaceutical composition comprising the antibody of claim 19 and a pharmaceutically acceptable carrier.
34. A method of treating a human patient suffering from a disorder selected from a cancer, a viral disease, an inflammatory disorder, and an autoimmune disorder, comprising administering the pharmaceutical composition of claim 33.
35. A diagnostic method for NKG2A protein comprising contacting the antibody of claim 19 with a sample selected from cells, tissue or serum and detecting the binding of said antibody to NKG2A in said sample.
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application is a continuation of U.S. application Ser. No. 15/624,882, filed Jun. 16, 2017, which is a continuation of U.S. application Ser. No. 14/548,327, filed Nov. 20, 2014, now U.S. Pat. No. 9,683,041, which is a divisional of U.S. application Ser. No. 13/477,888, filed May 22, 2012, now U.S. Pat. No. 8,901,283, which is a continuation of U.S. application Ser. No. 12/305,683, filed May 26, 2009, now U.S. Pat. No. 8,206,709, which is a 35 U.S.C. .sctn. 371 national stage application of International Patent Application PCT/EP2007/056485 (published as WO 2008/009545), filed Jun. 28, 2007, which claimed priority of European Patent Application 06116429.9, filed Jun. 30, 2006; this application further claims priority under 35 U.S.C. .sctn. 119 of U.S. Provisional Application 60/818,550, filed Jul. 5, 2006; the contents of all above-named applications are incorporated herein by reference.
FIELD OF THE INVENTION
[0002] The present invention relates to anti-NKG2A antibodies, in particular humanized versions of murine anti-NKG2A antibody Z270, as well as methods of producing and using such antibodies.
BACKGROUND OF THE INVENTION
[0003] CD94/NKG2A is a cytotoxicity inhibitory receptor found on subsets of NK, NKT and T cells, which restricts their killing of cells expressing the CD94/NKG2A-ligand HLA-E (see, e.g., WO99/28748). Antibodies that inhibit CD94/NKG2A may increase the cytolytic activity of tumor-specific lymphocytes against tumor cells. Therefore, therapeutic antibodies that inhibit CD94/NKG2A in cancer patients without killing CD94/NKG2A-expressing cells may be able to control tumor-growth. In addition, certain lymphomas such as, e.g., NK-lymphomas, are characterized by CD94/NKG2A expression. In such patients, therapeutic antibodies that target and kill CD94/NKG2A-expressing cells may be able to eradicate tumor cells. Anti-NKG2A antibodies have also been suggested for use in treating autoimmune or inflammatory diseases (see, e.g., US20030095965A1, WO02006070286).
[0004] Various antibodies against CD94/NKG2A have been described in the art. For example, Sivori et al. (Eur J Immunol 1996; 26:2487-92) refers to the murine anti-NKG2A antibody Z270; Carretero et al. (Eur J Immunol 1997; 27:563-7) describes murine anti-NKG2A antibody Z199 (now commercially available via Beckman Coulter, Inc., Product No. IM2750, USA); Vance et al. (J Exp Med 1999; 190: 1801-12) refers to murine anti-NKG2-antibody 20D5 (now commercially available via BD Biosciences Pharmingen, Catalog No. 550518, USA); and U.S. patent application publication 20030095965 describes murine antibody 3S9, which purportedly binds to NKG2A, NKG2C and NKG2E.
[0005] Currently available anti-CD94/NKG2A antibodies are of non-human origin, which makes them unsuitable for most therapeutic applications in humans due to their immunogenicity. While simple humanization approaches such as, e.g., CDR-grafting, are available, individualized humanization approaches are typically needed to obtain an optimal humanized variant, minimizing immunogenicity while sufficiently retaining or improving functional properties. Accordingly, there is a need for anti-CD94/NKG2A antibodies that are suitable for treatment of human patients.
SUMMARY OF THE INVENTION
[0006] The present invention provides anti-NKG2A antibodies, as well as compositions comprising such antibodies, and methods of producing and using such antibodies. In one embodiment, the antibody is a humanized version of murine anti-NKG2A antibody Z270, herein denoted a "humZ270". In another embodiment, the antibody is a humanized version of an anti-NKG2A antibody having substantially identical variable heavy-chain (VH) and/or variable light-chain (VL) domains to those of Z270.
[0007] Exemplary complementarity-determining region (CDR) residues and/or sites for amino acid substitutions in framework region (FR) and/or of such antibodies to produce antibodies having improved properties such as, e.g., lower immunogenicity, improved antigen-binding or other functional properties, and/or improved physicochemical properties such as, e.g., better stability, are provided. In one aspect, the invention provides humanized antibodies in which at least a portion of a Kabat CDR is identical to the corresponding portion in the human acceptor sequence. In one embodiment, the human acceptor framework sequence does not comprise any amino acid substitutions or back-mutations. In another embodiment, the human framework sequence comprises at least one amino acid substitution. Kabat positions for such exemplary amino acid substitutions include 5, 66, 67, 69, 71, 73, and 75 in the framework region of the VH domain, 46 and 48 in the framework region of the VL domain, and 60, 63, 64, and 65 in CDR-H2.
[0008] In other aspects, the invention provides for pharmaceutical compositions comprising such antibodies and a carrier, and for immunoconjugates comprising such antibodies conjugated to a cytotoxic or detectable agent.
[0009] In other aspects, the invention provides for nucleic acids and vectors encoding such antibodies, and host cells containing such nucleic acids and/or vectors. Also provided for are recombinant methods of producing anti-NKG2A antibodies by culturing such host cells so that the nucleic acids are produced.
[0010] In other aspects, the invention provides for articles of manufacture comprising a container comprising such anti-NKG2A antibodies and instructions directing a user to treat a disorder such as cancer or a viral disease in a patient. Optionally, the article may comprise another container containing another agent, wherein the instructions direct the user to treat the disorder with the antibody in combination with the agent.
[0011] The invention also provides for methods of using such anti-NKG2A antibodies in the treatment of disorders such as cancer, a viral disease, an inflammatory disorder or an autoimmune disorder in a patient, optionally in conjunction with another anti-cancer, anti-viral disease agent, or anti-inflammatory agent.
DESCRIPTION OF THE DRAWINGS
[0012] FIG. 1 shows an alignment of Z270VL and Z270VH with selected germline V- and J-segments and exemplary humZ270VL1 (SEQ ID NO:4) and humZ270VH1 (SEQ ID NO:5) sequences, with amino acid residue numbering according to the Kabat scheme (Kabat et al, 1991, Sequences of Proteins of Immunological Interest, 5th Ed. Public Health Service, National Institutes of Health, Bethesda, Md.) Mask residues are shaded in the Kabat scheme; CDR residues are shown in bold in the Kabat scheme; mouse/germline differences are shaded in the VKI_02/JK4 (SEQ ID NO:9) and VH1_18/JH6 (SEQ ID NO:10) sequences; and potential back-mutation residues are shaded in the humZ270VHNL sequences. The identified potential back-mutations in the VL were L46F and 148V. The identified potential back-mutations in the heavy chain were V5Q, M69L, T71V, T73K, and T75S. Also shown are the resulting humZ270VL consensus ("humZ270VL1 cons;" SEQ ID NO:6) and humZ270VH1 ("humZ270VH1 cons;" SEQ ID NO:7) consensus sequences. In humZ270VL1 cons, the amino acid at position 46 is L or F, and the amino acid at position 48 is I or V. In humZ270VH1 cons, the amino acid at position 5 is V or Q; the amino acid at position 69 is M or L; the amino acid at position 71 is T or V; the amino acid at position 73 is T or K; and/or the amino acid at position 75 is T or S. In an alternative humZ270VH1, humZ270VH1cons2 (SEQ ID NO:8), the amino acid at position 5 is V or Q; the amino acid at position 60 is S or A; the amino acid at position 63 is L or F; the amino acid at position 64 is Q or K; the amino acid at position 65 is G or D; the amino acid at position 66 is R or K; the amino acid at position 67 is V or A; the amino acid at position 69 is M or L; the amino acid at position 71 is T or V; the amino acid at position 73 is T or K; and/or the amino acid at position 75 is T or S.
[0013] FIG. 2 shows the CDRs of an exemplary humZ270 antibody, according to the Kabat definitions. The differences compared to murine Z270 CDRs are shown in bold. CDR_L1 corresponds to residues 24-34 of SEQ ID NO: 4. CDR_L2 corresponds to residues 50-56 of SEQ ID NO: 4. CDR_L3 corresponds to residues 89-97 of SEQ ID NO: 4. CDR_H1 corresponds to residues 31-35 of SEQ ID NO: 5. CDR_H2 corresponds to residues 50-66 of SEQ ID NO: 5. CDR_H3 corresponds to residues 99-114 of SEQ ID NO: 5. The residue numbering is referenced with respect to each SEQ ID NO recited.
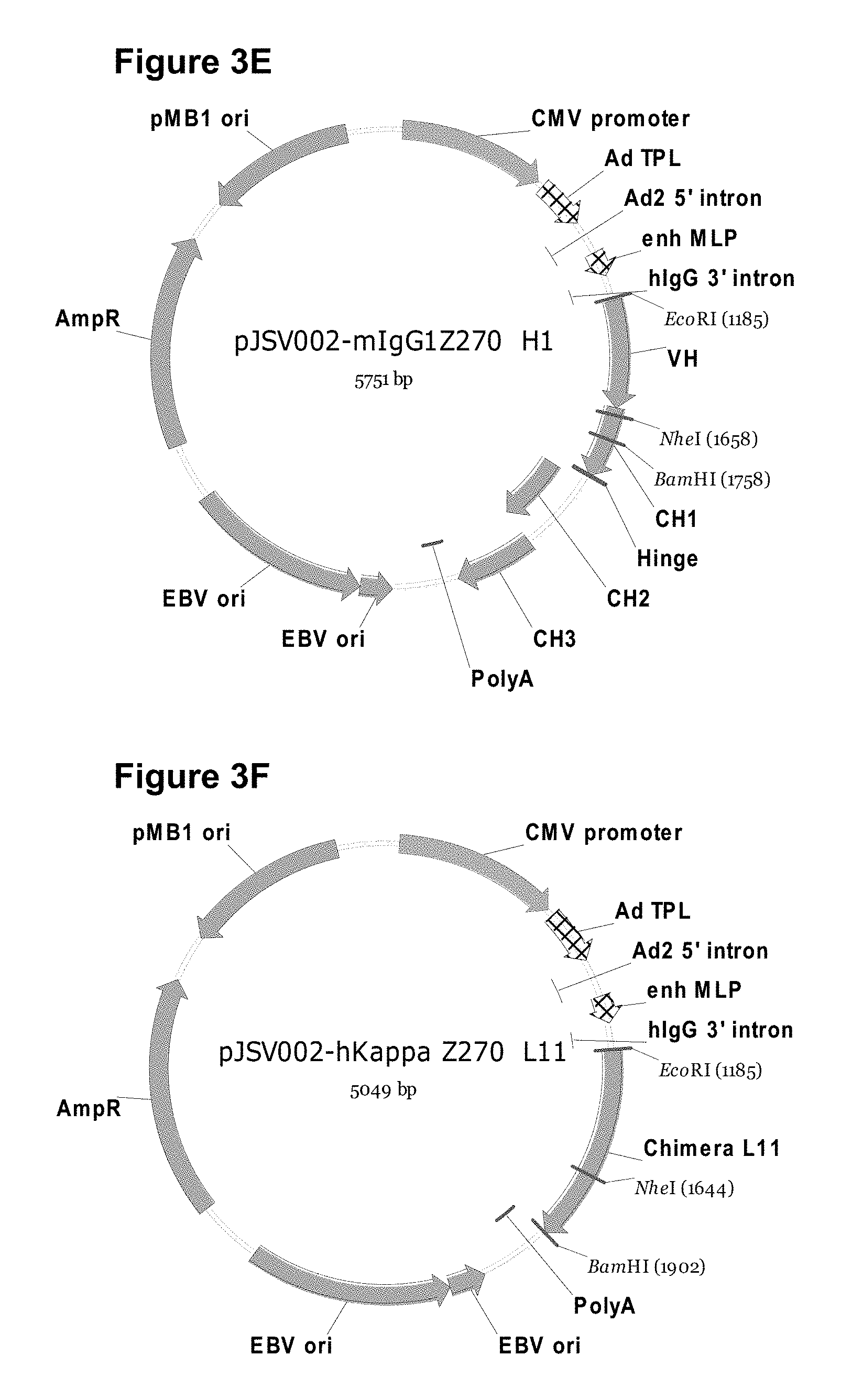
[0014] FIGS. 3A-3H show plasmids referred to in the Examples. (FIG. 3A) pMD19-T vector map for TA cloning. (FIG. 3B) pJSV002 cloning vector map for transient expression. (FIG. 3C) Light chain is inserted into pJSV002 with murine kappa constant region. (FIG. 3D) pJSV002-mlgG1 variant containing murine IgG1 constant region. (FIG. 3E) pJSV002-mlgG1 Z270 H1 containing Z270 heavy chain variable region and IgG1 heavy chain constant region. (FIG. 3F) pJSV002-hKappa Z270 L11 containing light chain variable region and human kappa chain constant region. (FIG. 3G) pJSV002-lgG4-S241P. (FIG. 3H) pJSV002-lgG4-S241P Z270H1.
[0015] FIGS. 4A-4J show different sequences derived from or based on Z270, FIG. 4A to FIG. 4J, SEQ ID NOS:14-23, respectively. See Examples 2-5 for details.
[0016] FIG. 5 shows an exemplary design of a vector for expression of humanized Z270 heavy-chains.
[0017] FIG. 6 shows an exemplary design of a vector for expression of humanized Z270 light-chains.
[0018] FIG. 7 shows an exemplary outline of a procedure for transient expression of humanized Z270 antibodies in HEK293 cells.
[0019] FIG. 8 shows an exemplary Biacore assay to determine humZ270 binding affinity for antigen.
[0020] FIGS. 9A-9B show affinity determinations of chimeric Z270 (FIG. 9A) and humZ270VL1NH1 (FIG. 9B).
[0021] FIG. 10 shows affinity determination of humZ270VL1NH1, having different back-mutations in the light chain or heavy chain. Chimeric Z270 was used as a comparison.
[0022] FIG. 11 shows the strategy for standard CDR-grafting of murine Z270 Kabat CDRs H1-H3 into a VH1_18/JH6 heavy chain acceptor framework, without (humZ270H3) or with (humZ270VH4) back-mutations.
[0023] FIG. 12 shows an alignment between humZ270VH constructs in different human acceptor sequences, all with a partly human portion of CDR-H2.
[0024] FIG. 13 shows the results of Biacore affinity evaluation of humZ270 variantsVL1NH1 and VH3-VH8, all normalized to the KD of hZ270VL1NH1.
[0025] FIG. 14 shows the selected residues for alanine-scan mutagenesis to identify critical paratope residues in the Z270 VL (SEQ ID NO: 1) and VH (SEQ ID NO: 2) segments.
[0026] FIG. 15 shows the results of Biacore affinity evaluation of Z270VH alanine mutants, normalized to the binding of chimeric Z270.
[0027] FIG. 16 shows the results of Biacore affinity evaluation of Z270VL alanine mutants, normalized to the binding of chimeric Z270.
[0028] FIG. 17 shows the binding of various recombinant Z270 variants to Ba/F3 cells stably over-expressing either CD94/NKG2A or CD94/NKG2C, using flow-cytometry.
[0029] FIG. 18 shows the results of an assay to evaluate the ability of recombinant Z270, chimeric Z270, humZ270VL1NH1, and Z199 to induce killing of .sup.51Cr-labeled LCL 721.221-Cw3 cells by CD94/NKG2A+ NKL cells, showing that humZ270VL1NH1 was more efficient in inducing killing.
[0030] FIG. 19 shows that humZ270VL1NH1 efficiently binds Ba/F3-CD94/NKG2A cells in a concentration dependent fashion (diamonds). However, when cells were pre-incubated with HLA-E tetramers, humZ270VL1NH1 was prevented from binding to Ba/F3-CD94/NKG2A cells (squares).
[0031] FIG. 20 shows that two different preparations of humZ270VL1NH1 and a humZ270 variant with a V5Q mutation in VH bind to CD94/NKG2A--but not CD94/NKG2C-expressing cells, though the V5Q-variant slightly less efficiently.
DEFINITIONS
[0032] The term "antibody" herein is used in the broadest sense and specifically includes full-length monoclonal antibodies, polyclonal antibodies, multispecific antibodies (e.g., bispecific antibodies), and antibody fragments, so long as they exhibit the desired biological activity. Various techniques relevant to the production of antibodies are provided in, e.g., Harlow, et al., ANTIBODIES: A LABORATORY MANUAL, Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y. (1988).
[0033] An "antibody fragment" comprises a portion of a full-length antibody, preferably antigen-binding or variable regions thereof. Examples of antibody fragments include Fab, Fab', F(ab).sub.2, F(ab').sub.2, F(ab).sub.3, Fv (typically the VL and VH domains of a single arm of an antibody), single-chain Fv (scFv), dsFv, Fd fragments (typically the VH and CH1 domain), and dAb (typically a VH domain) fragments; VH, VL, VhH, and V-NAR domains; minibodies, diabodies, triabodies, tetrabodies, and kappa bodies (see, e.g., III et al., Protein Eng 1997; 10: 949-57); camel IgG; IgNAR; and multispecific antibody fragments formed from antibody fragments, and one or more isolated CDRs or a functional paratope, where isolated CDRs or antigen-binding residues or polypeptides can be associated or linked together so as to form a functional antibody fragment. Various types of antibody fragments have been described or reviewed in, e.g., Holliger and Hudson, Nat Biotechnol 2005; 23, 1126-1136; WO2005040219, and published U.S. Patent Applications 20050238646 and 20020161201.
[0034] The term "antibody derivative", as used herein, comprises a full-length antibody or a fragment of an antibody, preferably comprising at least antigen-binding or variable regions thereof, wherein one or more of the amino acids are chemically modified, e.g., by alkylation, PEGylation, acylation, ester formation or amide formation or the like, e.g., for linking the antibody to a second molecule. This includes, but is not limited to, PEGylated antibodies, cysteine-PEGylated antibodies, and variants thereof.
[0035] An "immunoconjugate" comprises an antibody derivative associated with or linked to a second agent, such as a cytotoxic agent, a detectable agent, etc.
[0036] A "humanized" antibody is a human/non-human chimeric antibody that contains a minimal sequence derived from non-human immunoglobulin. For the most part, humanized antibodies are human immunoglobulins (recipient antibody) in which residues from a hypervariable region of the recipient are replaced by residues from a hypervariable region of a non-human species (donor antibody) such as mouse, rat, rabbit, or non-human primate having the desired specificity, affinity, and capacity. In some instances, framework region (FR) residues of the human immunoglobulin are replaced by corresponding non-human residues. Furthermore, humanized antibodies may comprise residues that are not found in the recipient antibody or in the donor antibody. These modifications are made to further refine antibody performance. In general, a humanized antibody will comprise substantially all of at least one, and typically two, variable domains, in which all or substantially all of the hypervariable loops correspond to those of a non-human immunoglobulin and all or substantially all of the FR residues are those of a human immunoglobulin sequence. The humanized antibody can optionally also comprise at least a portion of an immunoglobulin constant region (Fc), typically that of a human immunoglobulin. For further details, see, e.g., Jones et al., Nature 321:522-525 (1986); Riechmann et al., Nature 332:323-329 (1988); and Presta, Curr. Op. Struct Biol. 2:593-596 (1992), WO 92/02190, US Patent Application 20060073137, and U.S. Pat. Nos. 6,750,325, 6,632,927, 6,639,055, 6,548,640, 6,407,213, 6,180,370, 6,054,297, 5,929,212, 5,895,205, 5,886,152, 5,877,293, 5,869,619, 5,821,337, 5,821,123, 5,770,196, 5,777,085, 5,766,886, 5,714,350, 5,693,762, 5,693,761, 5,530,101, 5,585,089, and 5,225,539.
[0037] The term "hypervariable region" when used herein refers to the amino acid residues of an antibody that are responsible for antigen-binding. The hypervariable region generally comprises amino acid residues from a "complementarity-determining region" or "CDR" (residues 24-34 (L1), 50-56 (L2) and 89-97 (L3) in the light-chain variable domain and 31-35 (H1), 50-65 (H2) and 95-102 (H3) in the heavy-chain variable domain; Kabat et al. 1991, supra) and/or those residues from a "hypervariable loop" (residues 26-32 (L1), 50-52 (L2) and 91-96 (L3) in the light-chain variable domain and 26-32 (H1), 53-55 (H2) and 96-101 (H3) in the heavy-chain variable domain; Chothia and Lesk, J. Mol. Biol 1987; 196:901-917). Typically, the numbering of amino acid residues in this region is performed by the method described in Kabat et al., supra. Phrases such as "Kabat position", "using Kabat numbering", "variable domain residue numbering as in Kabat" and "according to Kabat" herein refer to this numbering system for heavy chain variable domains or light chain variable domains. Using the Kabat numbering system, the actual linear amino acid sequence of a peptide may contain fewer or additional amino acids corresponding to a shortening of, or insertion into, a FR or CDR of the variable domain. For example, a heavy chain variable domain may include a single amino acid insert (residue 52a according to Kabat) after residue 52 of CDR H2 and inserted residues (e.g. residues 82a, 82b, and 82c, etc. according to Kabat) after heavy chain FR residue 82. The Kabat numbering of residues may be determined for a given antibody by alignment at regions of homology of the sequence of the antibody with a "standard" Kabat numbered sequence. Unless otherwise indicated or contrary to context, the position of all amino acid residues in a VL or VH sequence described herein are according to Kabat.
[0038] "Framework region" or "FR" residues are those VH or VL residues other than the CDRs as herein defined.
[0039] A "variant" of a polypeptide refers to a polypeptide having an amino acid sequence that is substantially identical to a reference polypeptide, typically a native or "parent" polypeptide. The polypeptide variant may possess one or more amino acid substitutions, deletions, and/or insertions at certain positions within the native amino acid sequence.
[0040] "Conservative" amino acid substitutions are those in which an amino acid residue is replaced with an amino acid residue having a side chain with similar physicochemical properties. Families of amino acid residues having similar side chains are known in the art, and include amino acids with basic side chains (e.g., lysine, arginine, histidine), acidic side chains (e.g., aspartic acid, glutamic acid), uncharged polar side chains (e.g., glycine, asparagine, glutamine, serine, threonine, tyrosine, cysteine, tryptophan), nonpolar side chains (e.g., alanine, valine, leucine, isoleucine, proline, phenylalanine, methionine), beta-branched side chains (e.g., threonine, valine, isoleucine) and aromatic side chains (e.g., tyrosine, phenylalanine, tryptophan, histidine).
[0041] The term "substantially identical" in the context of two amino acid sequences means that the sequences, when optimally aligned, such as by the programs GAP or BESTFIT using default gap weights, share at least about 50, at least about 60, at least about 70, at least about 80, at least about 90, at least about 95, at least about 98, or at least about 99 percent sequence identity. In one embodiment, residue positions that are not identical differ by conservative amino acid substitutions. Sequence identity is typically measured using sequence analysis software. Protein analysis software matches similar sequences using measures of similarity assigned to various substitutions, deletions and other modifications, including conservative amino acid substitutions. For instance, the publicly available GCG software contains programs such as "Gap" and "BestFit" which can be used with default parameters to determine sequence homology or sequence identity between closely related polypeptides, such as homologous polypeptides from different species of organisms or between a wild-type protein and a mutein thereof. See, e.g., GCG Version 6.1. Polypeptide sequences can also be compared using FASTA or ClustalW, applying default or recommended parameters. A program in GCG Version 6.1., FASTA (e.g., FASTA2 and FASTA3) provides alignments and percent sequence identity of the regions of the best overlap between the query and search sequences (Pearson, Methods Enzymol. 1990; 183:63-98; Pearson, Methods Mol. Biol. 2000; 132:185-219). Another preferred algorithm when comparing a sequence to a database containing a large number of sequences from various organisms, or when deducing the computer program BLAST, especially blastp, using default parameters. See, e.g., Altschul et al., J. Mol. Biol. 1990; 215:403-410; Altschul et al., Nucleic Acids Res. 1997; 25:3389-402 (1997); each herein incorporated by reference. "Corresponding" amino acid positions in two substantially identical amino acid sequences are those aligned by any of the protein analysis software mentioned herein, typically using default parameters.
[0042] An antibody having a "biological characteristic" of a reference antibody, (e.g., Z270), is one that possesses one or more of the biological characteristics of that antibody that distinguish it from other antibodies that bind to the same antigen (e.g. NKG2A). For example, an antibody with a biological characteristic of Z270 may block activation of NKG2A, and/or cross-compete with Z270 in binding the extracellular domain of NKG2A.
[0043] NKG2A (OMIM 161555, the entire disclosure of which is herein incorporated by reference) is a member of the NKG2 group of transcripts (Houchins, et al. (1991) J. Exp. Med. 173:1017-1020). NKG2A is encoded by 7 exons spanning 25 kb, showing some differential splicing. NKG2A is an inhibitory receptor found on the surface of subsets of NK cells, .alpha./.beta. T cells, .gamma./.delta. cells, and NKT cells. Like inhibitory KIR receptors, it possesses an ITIM in its cytoplasmic domain. As used herein, "NKG2A" refers to any variant, derivative, or isoform of the NKG2A gene or encoded protein. Also encompassed are any nucleic acid or protein sequences sharing one or more biological properties or functions with wild type, full length NKG2A, and sharing at least 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99%, or higher nucleotide or amino acid identity. Human NKG2A comprises 233 amino acids in 3 domains, with a cytoplasmic domain comprising residues 1-70, a transmembrane region comprising residues 71-93, and an extracellular region comprising residues 94-233, of the following sequence:
TABLE-US-00001 (SEQ ID NO: 11) MDNQGVIYSDLNLPPNPKRQQRKPKGNKSSILATEQEITYAELNLQKASQ DFQGNDKTYHCKDLPSAPEKLIVGILGIICLILMASVVTIVVIPSTLIQR HNNSSLNTRTQKARHCGHCPEEWITYSNSCYYIGKERRTWEESLLACTSK NSSLLSIDNEEEMKFLSIISPSSWIGVFRNSSHHPWVTMNGLAFKHEIKD SDNAELNCAVLQVNRLKSAQCGSSIIYHCKHKL
[0044] NKG2C (OMIM 602891, the entire disclosure of which is herein incorporated by reference) and NKG2E (OMIM 602892, the entire disclosure of which is herein incorporated by reference) are two other members of the NKG2 group of transcripts (Gilenke, et al. (1998) Immunogenetics 48:163-173). NKG2C and NKG2E are activating receptors found on the surface of NK cells.
[0045] HLA-E (OMIM 143010, the entire disclosure of which is herein incorporated by reference) is a nonclassical MHC molecule that is expressed on the cell surface and regulated by the binding of peptides derived from the signal sequence of other MHC class I molecules. HLA-E binds natural killer (NK) cells and some T cells, binding specifically to CD94/NKG2A, CD94/NKG2B, and CD94/NKG2C (see, e.g., Braud et al. (1998) Nature 391:795-799, the entire disclosure of which is herein incorporated by reference). Surface expression of HLA-E is sufficient to protect target cells from lysis by CD94/NKG2A+NK, T, or NKT cell clones. As used herein, "HLA-E" refers to any variant, derivative, or isoform of the HLA-E gene or encoded protein. Also encompassed are any nucleic acid or protein sequences sharing one or more biological properties or functions with wild type, full length HLA-E, and sharing at least 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99%, or higher nucleotide or amino acid identity.
[0046] A nucleic acid is "operably linked" when it is placed into a functional relationship with another nucleic acid sequence. For example, DNA for a presequence or secretory leader is operably linked to DNA for a polypeptide if it is expressed as a preprotein that participates in the secretion of the polypeptide; a promoter or enhancer is operably linked to a coding sequence if it affects the transcription of the sequence; or a ribosome-binding site is operably linked to a coding sequence if it is positioned so as to facilitate translation. Generally, "operably linked" means that the DNA sequences being linked are contiguous, and, in the case of a secretory leader, contiguous and in reading phase. However, enhancers do not have to be contiguous. Linking is accomplished by ligation at convenient restriction sites. If such sites do not exist, the synthetic oligonucleotide adaptors or linkers are used in accordance with conventional practice.
[0047] An "isolated" molecule is a molecule that is the predominant species in the composition wherein it is found with respect to the class of molecules to which it belongs (i.e., it makes up at least about 50% of the type of molecule in the composition and typically will make up at least about 70%, at least about 80%, at least about 85%, at least about 90%, at least about 95%, or more of the species of molecule, e.g., peptide, in the composition). Commonly, a composition of an antibody molecule will exhibit 98%, 98%, or 99% homogeneity for antibody molecules in the context of all present peptide species in the composition or at least with respect to substantially active peptide species in the context of proposed use.
[0048] In the context of the present invention, "treatment" or "treating" refers to preventing, alleviating, managing, curing or reducing one or more symptoms or clinically relevant manifestations of a disease or disorder, unless contradicted by context. For example, "treatment" of a patient in whom no symptoms or clinically relevant manifestations of a disease or disorder have been identified is preventive or prophylactic therapy, whereas "treatment" of a patient in whom symptoms or clinically relevant manifestations of a disease or disorder have been identified generally does not constitute preventive or prophylactic therapy.
DETAILED DESCRIPTION OF THE INVENTION
[0049] The present invention concerns antibodies binding to NKG2A. In one aspect, the antibody is a humanized version of antibody Z270, which is a murine monoclonal antibody that specifically binds NKG2A, but not to human NKG2C or NKG2E. Z270 can block the function of human CD94/NKG2A, and specifically induce killing of cells by CD94/NKG2A-restricted lymphocytes in a concentration-dependent fashion.
[0050] The invention provides, e.g., humZ270 variants in which at least a portion of a VH CDR such as the CDR-H2 is identical to the corresponding portion of the human VH acceptor sequence, thus reducing the immunogenicity of the humanized antibody. Surprisingly, such humanized variants can be more effective in potentiating the cytotoxicity of a CD94/NKG2A-expressing cytotoxic lymphocyte than the murine or a chimeric form of Z270. In other aspects, the invention provides antibodies having CDRs comprising certain antigen-binding residues corresponding to those in murine antibody Z270, and human framework sequences. These and other aspects are described in more detail in the following sections and in the Examples.
Humanized Anti-NKG2A Antibodies
[0051] Methods for humanizing non-human antibodies have been described in the art. Generally, in a humanization process, nucleotides encoding the interaction-regions of a murine antibody can be cloned into a cDNA-vector encoding human IgG, which can be done such that a chimeric antibody is generated consisting of a human IgG backbone harbouring the murine CDRs. Such chimeric antibodies may exhibit a lower affinity, lower stability, or other undesired features in comparison with the original murine antibody, and may also be immunogenic. Therefore, individual amino acids in the chimeric Ab may need to be optimized to obtain a functional mAb of high quality for therapeutic applications in humans.
[0052] Typically, a humanized antibody has one or more amino acid residues introduced into it from a source that is non-human. These non-human amino acid residues are often referred to as "import" residues, which are typically taken from an "import" variable domain. Humanization can be essentially performed following the method of Winter and co-workers (Jones et al, Nature, 321: 522-525 (1986); Riechmann et al., Nature, 332: 323-327 (1988); Verhoeyen et al., Science, 239: 1534-1536 (1988)), by substituting hypervariable region sequences for the corresponding sequences of a human "acceptor" antibody. Accordingly, such "humanized" antibodies are chimeric antibodies (U.S. Pat. No. 4,816,567) wherein substantially less than an intact human variable domain has been substituted by the corresponding sequence from a non-human species. In practice, humanized antibodies are typically human antibodies in which some hypervariable region residues and possibly some FR residues are substituted by residues from analogous sites in rodent antibodies.
[0053] Another method for making humanized antibodies is described in U.S. patent application publication 2003/0017534, wherein humanized antibodies and antibody preparations are produced from transgenic non-human animals. The non-human animals are genetically engineered to contain one or more humanized immunoglobulin loci that are capable of undergoing gene rearrangement and gene conversion in the transgenic non-human animals to produce diversified humanized immunoglobulins.
[0054] The choice of human variable domains, both light and heavy, to be used in making the humanized antibodies is very important to reduce antigenicity. According to the so-called "best-fit" method, the sequence of the variable domain of a rodent antibody is screened against a library of known human variable-domain sequences or a library of human germline sequences. The human sequence that is closest to that of the rodent can then be accepted as the human framework region for the humanized antibody (Sims et al., J. Immunol. 1993; 151:2296 et seq.; Chothia et al, Chothia and Lesk, J. Mol. Biol 1987; 196:901-917). Another method uses a particular framework region derived from the consensus sequence of all human antibodies of a particular subgroup of light or heavy chains. The same framework may be used for several different humanized antibodies (Carter et al., PNAS USA, 1992; 89:4285 et seq.; Presta et al., J Immunol 1993; 151:2623 et seq.). Other methods designed to reduce the immunogenicity of the antibody molecule in a human patient include veneered antibodies (see, e.g., U.S. Pat. No. 6,797,492 and U.S. patent application publications 20020034765 and 20040253645) and antibodies that have been modified by T-cell epitope analysis and removal (see, e.g., U.S. patent application publications 20030153043 and U.S. Pat. No. 5,712,120).
[0055] It is further important that antibodies be humanized with retention of high affinity for the antigen and other favorable biological properties. To achieve this goal, according to a preferred method, humanized antibodies are prepared by a process of analysis of the parental sequences and various conceptual humanized products using three-dimensional models of the parental and humanized sequences. Three-dimensional immunoglobulin models are commonly available and are familiar to those skilled in the art. Computer programs are available that illustrate and display probable three-dimensional conformational structures of selected candidate immunoglobulin sequences. Inspection of these displays permits analysis of the likely role of the residues in the functioning of the candidate immunoglobulin sequence, i.e., the analysis of residues that influence the ability of the candidate immunoglobulin to bind its antigen. In this way, FR residues can be selected and combined from the recipient and import sequences so that the desired antibody characteristic, such as increased affinity for the target antigen(s), is achieved. In general, the hypervariable region residues are directly and most substantially involved in influencing antigen binding.
[0056] Example 1 below describes the design of exemplary humanized anti-NKG2A antibodies that bind NKG2A, and the analysis is, at least in part, illustrated in FIG. 1. As shown in FIG. 1, in one humZ270 antibody of the invention, the C-terminal portion of CDR-H2 (corresponding to Kabat residues 61-65) are identical to the corresponding portion of the human acceptor sequence. Further, as described in the figure legend, a humZ270VL sequence (SEQ ID NO:4) may optionally comprise mutations in one or both of the indicated FR residues L46 and 148, and a humZ270VH sequence (SEQ ID NO:5) may optionally comprise mutations in one or more of the indicated FR residues V5, M69, T71, T73 and T75, with amino acid numbering according to Kabat. Table 1 describes exemplary humZ270VL and humZ270VH variants comprising exemplary human-to-murine back-mutations in the humZ270VH and humZ270VL FR sequences, as well as exemplary combinations of FR mutations. In Table 1 and elsewhere herein, the amino acid positions are designated according to Kabat, in which amino acids V5, M69, T71, T73, and T75 in the humZ270VH domain correspond to amino acids V5, M70, T72, T74, and T76 in SEQ ID NO:5, SEQ ID NO:7, SEQ ID NO:8, or other exemplary humZ270VH sequences described herein.
TABLE-US-00002 TABLE 1 humZ270VL and humZ270VH variants comprising exemplary FR back-mutations in native or consensus humZ270VL (SEQ ID NO: 4 or 6) and/or humZ270VH (SEQ ID NO: 5, 7, or 8) sequences. humZ270VL Variants humZ270VH Variants None None L46F V5Q I48V M69L L46F and I48V T71V T73K T75S V5Q and M69L V5Q and T71V V5Q and T73K V5Q and T75S M69L and T71V M69L and T73K M69L and T75S T71V and T73K T71V and T75S T73K and T75S V5Q, T73K and T75S V5Q, T71V and T75S V5Q, T71V and T73K V5Q, M69L and T75S V5Q, M69L and T73K V5Q, M69L and T71V T71V, T73K and T75S M69L, T73K and T75S M69L, T71V and T75S, M69L, T71V and T73K, V5Q, M69L, T71V and T73K, V5Q, M69L, T71V and T75S, V5Q, M69L, T73K and T75S, V5Q, T71V, T73K and T75S, M69L, T71V, T73K and T75S, V5Q, M69L, T71V, T73K and T75S
[0057] Accordingly, the present invention provides for humanized versions of an anti-NKG2A antibody produced by the Z270 hybridoma, as well as for humanized versions of non-human antibodies sharing biological characteristics and/or substantial sequence identity with Z270. In another embodiment, the monoclonal antibody or a fragment or derivative thereof is capable of binding to a non-human primate NKG2A.
[0058] The humanized antibody herein comprises non-human hypervariable region or CDR residues incorporated into human VH and VL domains.
[0059] In one aspect, the invention provides a humanized antibody comprising antigen-binding residues from the CDRs of murine antibody Z270 in a human acceptor framework, wherein at least the 6 C-terminal amino acid residues of the CDR-H2 are the same as those in the human acceptor sequence. Such humanized antibodies can be more effective than the original murine Z270 antibody or a chimeric version thereof in, e.g., potentiating the cytotoxic activity of a CD94/NKG2A-expressing cytotoxic lymphocyte, such as, e.g., an NK-cell, an NKT-cell, an .alpha./.beta. T-cell, and/or a .gamma./.delta. T-cell, or of a population of CD94/NKG2A-expressing cytotoxic lymphocytes.
[0060] Typical antibodies of the invention comprise antigen-binding residues corresponding to, or portions of the Z270 CDR-H2 and CDR-H3 that comprise, residues D52, D54, R94, F99, T(100C), and W(100F) of SEQ ID NO:2 or SEQ ID NO:5, which have been shown in the Examples to be critical for antigen-binding. Optionally, the antibodies also comprise VH residues N35, Y53, E56, D98, V(100A), and L(100D).
[0061] In another aspect, a humanized antibody comprises a CDR-H1 sequence corresponding to residues 31-35 of SEQ ID NO:5 and a CDR-H3 sequence corresponding to residues 95-102 of SEQ ID NO:5, wherein the CDR-H2 sequence comprises residues 50-59 of SEQ ID NO:5. In such humanized antibodies, the VH region can have, for example 50% or more sequence identity to SEQ ID NO:5, such as at least 60%, at least 70%, or at least 75% sequence identity. Exemplary humanized VH domains having such sequences are described in the examples and below.
[0062] In one embodiment, in such humanized antibodies, the amino acid at position 5 of the VH region can be V or Q; the amino acid at position 69 of the VH region can be M or L; the amino acid at position 71 of the VH region can be T or V; the amino acid at position 73 of the VH region can be T or K; and/or the amino acid at position 75 of the VH region can be T or S. In separate embodiments, the VH region comprises V5 or Q5, M69 or L69, T71 or V71, T73, or K71, and/or T75 or S75 residues. In another embodiment, the amino acid at position 69 is L. In another additional or alternative embodiment, the amino acid at position 71 is V.
[0063] The humanized antibody may comprise a VH human acceptor framework from a human acceptor sequence selected from, e.g., VH1_18, VH5_a, VH5_51, VH1_f, and VH1_46, and the J-segment is JH6, such as, e.g., a VH1_18, VH5_a, VH5_51, or VH1_f, or other human germline VH framework sequences known in the art. In one embodiment, the VH segment is VH1_18. In a particular embodiment, the VH region comprises the sequence of SEQ ID NO:8. In another particular embodiment, the VH region comprises the sequence of SEQ ID NO:7. For example, the VH region may comprise the sequence of SEQ ID NO:5.
[0064] The VL region human acceptor sequence may be, e.g., VKI_02/JK4. In a particular embodiment, the VL region comprises SEQ ID NO:4.
[0065] In one aspect, a humanized antibody of the invention comprises a CDR-H2 comprising a murine portion consisting of residues 50-59 of SEQ ID NO:2 linked to a CDR-H3 consisting of residues 95-102 of SEQ ID NO:2 via suitable FR sequences.
[0066] As described herein, in one embodiment, the humanized antibody comprises no FR substitution in the VH domain. In another embodiment, the humanized antibody comprises a VH domain FR substitution at one or more positions selected from 5, 69, 71, 73 and 75, utilizing the variable domain numbering system according to Kabat. In another embodiment, the humanized antibody comprises VH domain FR substitutions at two or more of positions 5, 69, 71, 73 and 75; and in other embodiments, at three, four, or all of such positions. In separate and independent embodiments, the humanized antibody comprises one VH domain FR substitution at a position selected from 5, 69, 71, 73 and 75. In other separate and independent embodiments, the humanized antibody comprises VH domain FR substitutions at positions 5 and 69, or 5 and 71, or 5 and 73, or 5 and 75, or 69 and 71, 69 and 73, or 69 and 75, or 71 and 73, or 71 and 75, or 73 and 75, or 5, 69 and 71, or 5, 69 and 73, or 5, 69 and 75, or 69, 71 and 73, or 69, 71 and 75, or 71, 73, and 75, or 5, 69, 71, and 73, or 5, 69, 71, and 75, or 5, 71, 73, and 75, or 69, 71, 73, and 75, or 5, 69, 71, 73, and 75. Fewer rather than more framework substitutions can minimize immunogenicity, but binding efficacy may be an important consideration for some applications. Thus, preferred substitutions are back-mutations, i.e., mutations which replace an amino acid at a certain position in the human FR with the amino acid at the corresponding position in a non-human donor FR. Thus, in separate and independent embodiments, the VH domain amino acid substitution at position 5 is V5Q, the amino acid substitution at position 69 is M69L, the amino acid substitution at position 71 is T71V, the amino acid substitution at position 73 is T73K, and the amino acid substitution at position 75 is T75S. In a particular embodiment, the humanized antibody comprises a V at position 71 and/or an L at position 69.
[0067] The humanized antibody herein also comprises non-human hypervariable region residues incorporated into a human VL domain. In one embodiment, the humanized antibody comprises no FR substitution in the VL domain. In another embodiment, the humanized antibody comprises a VL domain FR substitution at one of positions 46 and 48, utilizing the variable domain numbering system according to Kabat. In another embodiment, the humanized antibody comprises VL domain FR substitutions both of positions 46 and 48. Preferred substitutions are back-mutations, i.e., mutations which replace an amino acid at a certain position in the human FR with the amino acid at the corresponding position in a non-human donor FR. Thus, in separate and independent embodiments, the VL domain amino acid substitution at position 46 is L46F, and the VL domain amino acid substitution at position 48 is 148V.
[0068] An exemplary humanized antibody comprises a VH domain comprising a CDR-H1 sequence corresponding to residues 31-35 of SEQ ID NOS:5 or 7, a CDR-H2 sequence corresponding to residues 50-66 of SEQ ID NOS:5 or 7, and a CDR-H3 sequence corresponding to residues 95-102 of SEQ ID NOS:5 or 7. The humanized antibody may further comprise an amino acid at Kabat position 5 which is V or Q, an amino acid at Kabat position 69 which is M or L, an amino acid at Kabat position 71 which is T or V, an amino acid at Kabat position 73 is T or K, and/or an amino acid at Kabat position 75 which is T or S, in the VH domain. In one embodiment, the VH domain comprises a framework region substitution in at least one Kabat position selected from the group consisting of 5, 69, 71, 73, and 75, e.g., corresponding to any of the VH FR substitutions, or combinations thereof, listed in Table 1. In one embodiment, the VH domain comprises the sequence of SEQ ID NO:7. In another embodiment, the VH domain comprises the sequence of SEQ ID NO:5.
[0069] An exemplary humanized antibody may also or alternatively comprise a VL domain comprising a CDR-L1 sequence corresponding to residues 24-34 of SEQ ID NO:4, a CDR-L2 sequence corresponding to residues 50-56 of SEQ ID NO:4, and an CDR-L3 sequence corresponding to residues 89-97 of SEQ ID NO:4. The humanized antibody may further comprise an amino acid at Kabat position 46 which is L or F and/or an amino acid at Kabat position 48 which is I or V. In one embodiment, the VL domain comprises a framework region substitution in at least one Kabat position selected from 46 and 48, e.g., corresponding to any of the VL FR substitutions, or combinations thereof, listed in Table 1. In one embodiment, the VL domain comprises the amino acid sequence of SEQ ID NO:4. In another embodiment, the VL domain comprises the sequence of SEQ ID NO:6.
[0070] In another aspect, the invention provides a humanized antibody that binds human NKG2A, the antibody comprising a VH domain that comprises the amino acid sequence of SEQ ID NO:5, optionally with one or more FR substitutions at Kabat positions 5, 69, 71, 73, and/or 75. The optional FR substitutions can be selected from, e.g., V5Q, M69L, T71V, T73K, and/or T75S, as well as any combination thereof. Such a humanized antibody may also or alternatively comprise a VL domain that comprises the amino acid sequence of SEQ ID NO:4, optionally with one or more FR substitutions at Kabat positions 46 and/or 48. The optional FR substitutions can be selected from, e.g., L46F and/or 148V.
[0071] Optionally, in a particular aspect, the VH domain comprises amino acid modifications of one or more CDR residues, e.g. where the modifications essentially maintain or improve affinity of the antibody. For example, the antibody variant may have one, two, three, or from one to about seven amino acid substitutions in the above VH CDR sequences.
[0072] In a particular aspect, amino acids in the humanized antibody VH CDRs which are different from the amino acids at the corresponding positions in the non-human donor VH CDRs can be substituted to improve the binding properties and/or stability of the humanized antibody. For example, one or more of these amino acids can be substituted for the amino acid at the corresponding position(s) in the non-human donor VH CDR. In one embodiment, the variant antibody comprises CDRH2 substitutions at one or more positions selected from 60, 63, 64, and 65, according to Kabat (corresponding to positions 61, 64, 65, and 66 in SEQ ID NO:5 or 7). In separate and independent embodiments, the antibody variant comprises a CDRH2 amino acid substitution at one position selected from 60, 63, 64, and 65. In other separate and independent embodiments, the antibody variant comprises CDRH2 amino acid substitution at positions 60 and 63, or 60 and 64, or 60 and 65, or 63 and 64, or 63 and 65, or 64 and 65, or 60, 63, and 64, or 60, 63, and 65, or 60, 64, and 65, or 63, 64, and 65, or 60, 63, 64, and 65. Preferred substitutions are back-mutations, i.e., mutations which replace an amino acid at a certain position in the humanized CDR with the amino acid at the corresponding position in a non-human donor CDR. Thus, in separate and independent embodiments, the CDRH2 amino acid substitution at position 60 is A60S, the amino acid substitution at position 63 is L63F, the amino acid substitution at position 64 is Q64K, and the amino acid substitution at position 65 is G65D. In aspects where the antibody variant comprises one or more of the CDRH2 amino acid substitutions A60S, L63F, Q64K, and G65D, the antibody variant comprises the VH FR amino acid substitutions at positions 66 and 67 according to Kabat, optionally in conjunction with one or more of the VH FR substitutions previously described. Preferably, the amino acid substitutions are R66K and V67A. For example, in one embodiment, the antibody variant comprises a VH domain comprising a CDR-H1 sequence corresponding to residues 31-35 of SEQ ID NO:5, a CDR-H2 sequence corresponding to residues 50-66 of SEQ ID NO:5, and a CDR-H3 sequence corresponding to residues 95-102 of SEQ ID NO:5, with A60S, L63F, Q64K, G65D, R66K and V67A "back-mutations," optionally in conjunction with additional amino acid modifications.
[0073] In another particular aspect, the invention provides for a humanized antibody that binds human NKG2A, the antibody comprising a VH domain that comprises non-human CDR residues incorporated into a human VH domain, the VH domain comprising a CDR-H1 sequence corresponding to residues 31-35 of SEQ ID NO:8, a CDR-H2 sequence corresponding to residues 50-66 of SEQ ID NO:8, and a CDR-H3 sequence corresponding to residues 95-102 of SEQ ID NO:8, wherein the amino acids at Kabat positions 63, 64, 65, 66, and 67 are F, K, D, K, A, respectively. In one embodiment, the VH domain comprises the amino acid sequence of SEQ ID NO:8. In another embodiment, the residue at Kabat position 60 is A. In another embodiment, the residue at Kabat position 60 is S, and the humanized antibody comprises the CDR-H1, -H2, and H3 sequences of Z270, with FR back-mutations in the two amino acids adjacent to the C-terminal end of CDR-H2. The humanized antibodies described in this section may further comprise FR back-mutations in other residues, e.g., at Kabat positions 5, 69, 71, 73, and/or 75, as described herein.
[0074] In another aspect, the invention provides for a humanized antibody wherein the Kabat CDRs in the VH region all derive from the murine Z270VH (SEQ ID NO:2), further comprising S60A, F63L, K64Q, and/or D65G mutations. In one embodiment, the humanized antibody comprises all of the mutations S60A, F63L, K64Q, and/or D65G.
[0075] The humanized antibody can also comprise a VL domain comprising a CDR-L1 sequence corresponding to residues 24-34 of SEQ ID NO:6, a CDR-L2 sequence corresponding to residues 50-56 of SEQ ID NO:6, and an CDR-L3 sequence corresponding to residues 89-97 of SEQ ID NO:6, e.g., in addition to the VH domain CDRs described above. In one embodiment, the VL domain of the humanized antibody comprises the amino acid sequence of SEQ ID NO:6. In another embodiment, the VL domain of the humanized antibody comprises the amino acid sequence of SEQ ID NO:4. Optionally, such a humanized antibody comprises amino acid modifications of one or more VL CDR residues, e.g., where the modifications essentially maintain or improve affinity of the antibody. For example, the antibody variant may have one, two, three, or from one to about seven amino acid substitutions in the above VL CDR sequences.
[0076] The present application also contemplates affinity-matured antibodies that bind anti-NKG2A, containing additional CDR or FR mutations that improve the affinity of the humanized antibody for the antigen. Methods for preparing such affinity-matured antibodies are known in the art. The parent antibody may be a humanized antibody, e.g., one comprising the VL/VH sequences of SEQ ID NOS:4 and 5 (optionally with one or more of the CDRH2 and/or FR substitutions described herein), or one comprising the consensus VL/VH sequences of SEQ ID NOS:6 and 7, respectively. The affinity-matured antibody preferably binds to NKG2A with an affinity comparable or superior to that of murine Z270, e.g. from at least about one-, about two- or about four-fold to about 100-fold or about 1000-fold improved affinity, e.g. as assessed using a binding assay as described below.
[0077] In another aspect, the invention provides humanized antibodies that comprise a VH domain having at least about 50%, at least about 70%, at least about 80% sequence identity (e.g., at least about 85%, 90%, 95%, 97%, or more identity) to the VH domain of Z270, humZ270, humZ270cons, and/or humZ270cons2 (SEQ ID NOS:2, 5, 7, and 8, respectively). In another particular aspect, the invention provides a humanized antibody that binds NKG2A, comprising a VH domain that comprises non-human CDR residues incorporated into a human VH domain, wherein the VH domain is at least about 50% identical to humZ270VH1 (SEQ ID NO:5). In one embodiment, the VH domain is at least 90% identical to SEQ ID NO:5. For example, the humanized antibody may comprise a CDR-H1 sequence corresponding to residues 31-35 of SEQ ID NO:5, a CDR-H2 sequence corresponding to residues 50-66 of SEQ ID NO:5, and a CDR-H3 sequence corresponding to residues 95-102 of SEQ ID NO:5. The humanized antibody may also or alternatively comprise a V or Q at Kabat position 5, an M or L at Kabat position 69, a T or V at Kabat position 71, a T or K at Kabat position 73, and a T or S at Kabat position 75, in the VH domain.
[0078] In another aspect, the invention provides an antibody molecule that also or alternatively comprises a VL domain having at least about 50%, at least about 70%, or at least about 80% sequence identity (e.g., at least about 85%, 90%, 95%, 97%, or more identity) to the VL domain of Z270, humZ270, and/or humZ270 cons (SEQ ID NOS:1, 4, and 6, respectively). In another particular aspect, the invention provides a humanized antibody that binds NKG2A, comprising a VL region that comprises non-human CDR residues incorporated into a human VL domain, wherein the VL domain is at least 50% identical to SEQ ID NO:4. In one embodiment, the VL region is at least 90% identical to SEQ ID NO:4. For example, the humanized antibody may comprise a CDR-L1 sequence corresponding to residues 24-34 of SEQ ID NO:4, a CDR-L2 sequence corresponding to residues 50-56 of SEQ ID NO:4, and an CDR-L3 sequence corresponding to residues 89-97 of SEQ ID NO:4. The humanized antibody may also or alternatively comprise a L or F at Kabat position 46 and/or an I or V at Kabat position 48, in the VL domain.
[0079] In another aspect, the invention provides an isolated humanized antibody that specifically binds NKG2A and that comprises either (a) CDR-L1, CDR-L2, and/or CDR-L3 sequences of Z270 VL (SEQ ID NO:1), humZ270 VL (SEQ ID NO:4), or humZ270 VL cons (SEQ ID NO:6) or (b) CDR-H1, CDR-H2, and/or CDR-H3 sequences of Z270 VH (SEQ ID NO:2), humZ270 (SEQ ID NO:5), humZ270 cons (SEQ ID NO:7), or humZ270 cons2 (SEQ ID NO:8). In another aspect, the invention provides an antibody molecule that comprises at least a complete set of VH CDRs from Z270, humZ270, or humZ270 cons, wherein the 6 C-terminal amino acids are identical to those of the human acceptor sequence. In a particular aspect, the invention provides an antibody molecule that comprises CDR-H1, CDR-H2 (or an N-terminal portion thereof), and CDR-H3 of Z270, humZ270, or humZ270 cons and at least some of CDR-L1, CDR-L2, and CDR-L3 of Z270, humZ270, or humZ270 cons. In a more particular aspect, the invention provides an antibody molecule wherein the antibody molecule comprises CDR-L1, CDR-L2, and CDR-L3, CDR-H1, CDR-H2, and CDR-H3 of Z270, humZ270, or humZ270 cons, wherein CDR-L1 is linked to CDR-L2, CDR-L2 is linked to CDR-L3, CDR-H1 is linked to CDR-H2, and CDR-H2 is linked to CDR-H3, via suitable FR sequences.
[0080] In a particular aspect, the invention provides antibody molecules that comprise essentially all of the VH and/or VL domains of humZ270, humZ270 cons, or humZ270 cons2.
[0081] A humanized anti-NKG2A antibody according to the invention may comprise any full-length or partial heavy-chain (HC) comprising a humZ270VH described herein and/or any full-length or partial HC comprising a humZ270VH may be combined with any humZ270VL described herein in, and the resulting antibody or fragment tested for antigen binding, functional effects on CD94/NKG2A-expressing cells, and/or immunogenicity.
[0082] Various forms of the humanized antibody are contemplated. For example, the humanized antibody may be an antibody fragment, such as a Fab or other type of fragment described herein. Alternatively, the humanized antibody may be a full-length or intact antibody, such as a full-length or intact IgG1 or IgG4 antibody. In one embodiment, the humanized antibody is a full-length IgG4 antibody or a fragment thereof.
[0083] In one aspect, the present invention provides a humanized antibody characterized by: a) specifically binding to NKG2A; b) not specifically binding to an Fc receptor; and c) when bound to NKG2A on a human NK cell, causing said NK cell to lyse a target human cell bearing HLA-E on the target cell surface, when said target cell comes into contact with said NK cell. In one embodiment, the humanized antibody comprises a mouse or human IgG1 constant region that has been modified to prevent binding to an Fc receptor, or a human IgG4 constant region. Such antibodies, as well as antibody fragments that do not bind an Fc receptor, are particularly useful in applications where it is desired to activate NK cells (e.g. cancer, infectious disease), without leading to the depletion of the NK cell themselves, as might be mediated by antibody dependent cell cytotoxicity, and can be referred to as "non-depleting" antibodies.
[0084] In another aspect, the humanized antibody comprises a mouse or human IgG1 constant region that binds an Fc receptor, or a human IgG1, 2, 3 or 4 constant region has been modified to bind an Fc receptor or increase binding to an Fc receptor, or a human IgG.sub.4 constant region. In another embodiment, the monoclonal antibody or a fragment thereof is linked to a moiety that is toxic to a cell to which the antibody is bound. Such antibodies are particularly useful in applications where it is desired to deplete an NK cell, useful in certain applications such as NK-LDGL (NK-type lymphoproliferative disease of granular lymphocytes; alternatively called NK-LGL), and can be referred to as "depleting" antibodies.
[0085] For recombinant production of humanized antibodies, humanized VH and VL regions, or variant versions thereof, can be cloned into expression vectors encoding full-length or truncated constant regions from a human antibody according to standard recombinant methods (see, e.g., Sambrook et al., Molecular Cloning: A Laboratory Manual, 2.sup.nd Ed., Cold Spring Harbor Laboratory, Cold Spring Harbor, N.Y., 1989). The result is a transfected cell line that expresses and secretes the humanized antibody molecule of interest, comprising the selected VH and VL regions and constant regions. cDNA sequences encoding the constant regions of human antibodies are known. Exemplary cDNA sequences available via, e.g., GenBank, each of which incorporated by reference in its entirety, are as follows:
[0086] Human IgG1 constant heavy chain region: GenBank accession No.: J00228;
[0087] Human IgG2 constant heavy chain region: GenBank accession No.: J00230;
[0088] Human IgG3 constant heavy chain region: GenBank accession No.: X04646;
[0089] Human IgG4 constant heavy chain region: GenBank accession No.: K01316; and
[0090] Human kappa light chain constant region: GenBank accession No.: J00241.
[0091] If desired, the class of a humanized antibody may also be "switched" by known methods. For example, an antibody that was originally produced as an IgM molecule may be class switched to an IgG antibody. Class switching techniques also may be used to convert one IgG subclass to another, e.g., from IgG1 to IgG2. Thus, the effector function of the antibodies of the invention may be changed by isotype switching to, e.g., an IgG1, IgG2, IgG3, IgG4, IgD, IgA, IgE, or IgM antibody for various therapeutic uses.
[0092] The constant region may further be modified according to known methods. For example, in an IgG4 constant region, residue S241 may be mutated to a proline (P) residue to allow complete disulphide bridge formation at the hinge (see, e.g., Angal et al., Mol Immunol. 1993; 30:105-8).
Antibody Fragments
[0093] The humanized antibodies of the invention may be prepared as antibody fragments, or antibody fragments may be prepared from humanized full-length antibodies.
[0094] Various techniques have been developed for the production of antibody fragments of humanized antibodies. Traditionally, these fragments were derived via proteolytic digestion of full-length antibodies (see, e.g., Morimoto et al., Journal of Biochemical and Biophysical Methods, 24:107-117 (1992); and Brennan et al., Science, 229:81 (1985)). However, these fragments can now be produced directly by recombinant host cells. Alternatively, Fab'-SH fragments can be directly recovered from E. coli and chemically coupled to form F(ab')2 fragments (Carter et al., Bio/Technology, 10:163-167 (1992)). According to another approach, F(ab')2 fragments can be isolated directly from recombinant host cell culture. Other techniques for the production of antibody fragments will be apparent to the skilled practitioner. In other embodiments, the antibody of choice is a single-chain Fv fragment (scFv). See WO 1993/16185; U.S. Pat. Nos. 5,571,894; and 5,587,458. The antibody fragment may also be a "linear antibody", e.g., as described in U.S. Pat. No. 5,641,870, for example. Such linear antibody fragments may be monospecific or bispecific.
[0095] Bispecific antibodies are antibodies that have binding specificities for at least two different epitopes. Methods for making bispecific antibodies are known in the art, and traditional production of full-length bispecific antibodies is usually based on the coexpression of two immunoglobulin heavy-chain-light-chain pairs, where the two chains have different specificities (Millstein et al., Nature, 305: 537-539 (1983)). In the bispecific antibodies according to the present invention, at least one binding epitope is on the NKG2A protein. The anti-NKG2A-binding "arm" may be combined with an "arm" that binds to a triggering molecule on a leukocyte such as a T-cell receptor molecule (e.g. CD2 or CD3), or Fc receptors for IgG (Fcgamma-R), such as Fo-gamma-RI (CD64), Fo-gamma-RII (CD32) and Fo-gamma-RIII (CD16), so as to focus cellular defense mechanisms to the NKG2A-expressing cell. Bispecific antibodies may also be used to localize cytotoxic agents to cells that express NKG2A. These antibodies possess a NKG2A-binding arm and an arm that binds the cytotoxic agent (e.g. saporin, anti-interferon-alpha, vinca alkaloid, ricin A chain, methotrexate, or radioactive isotope hapten). Bispecific antibodies can be prepared as full-length antibodies or antibody fragments (e.g. F(ab')2 bispecific antibodies). Antibodies with more than two valencies are contemplated. For example, trispecific antibodies can be prepared. Tutt et al., J. Immunol, 147: 60 (1991).
[0096] Typical antibodies, antibody fragments and multispecific antibodies of the invention comprise portions of the CDR-H2 and CDR-H3 that comprise residues D52, D54, R94, F99, T(100C), and W(100F) of SEQ ID NO:2 or SEQ ID NO:5, which have been shown to be critical for antigen-binding (see Examples). Optionally, the humanized antibody fragments and multispecific antibodies also comprise Z270 heavy chain residues N35, Y53, E56, D98, V(100A), and L(100D). In one aspect, a humanized antibody fragment or multispecific antibody of the invention comprises residues 50-59 of the CDR-H2 and residues 95-102 of the CDR-H3 of SEQ ID NO:2 or SEQ ID NO:5. In one embodiment, the humanized antibody fragment or multispecific antibody does not comprise one, two, or all of Z270 CDR-L1, CDR-L2, and CDR-L3. In an additional or alternative embodiment, the antibody fragment or multispecific antibody does not comprise the Z270 CDR-H1.
Antibody Derivatives
[0097] Antibody derivatives within the scope of this invention include humanized antibodies conjugated or covalently bound to a second agent.
[0098] For example, in one aspect, the invention provides immunoconjugates comprising a humanized antibody conjugated or covalently bonded to a cytotoxic agent. The term "cytotoxic agent" as used herein is a molecule that is capable of killing a cell bearing a NKG2A receptor on its cell surface. Any type of moiety with a cytotoxic or cytoinhibitory effect can be conjugated to the present antibodies to form a cytotoxic conjugate of the present invention and to inhibit or kill specific NK receptor expressing cells, including therapeutic radioisotopes, toxic proteins, toxic small molecules, such as drugs, toxins, immunomodulators, hormones, hormone antagonists, enzymes, oligonucleotides, enzyme inhibitors, therapeutic radionuclides, angiogenesis inhibitors, chemotherapeutic drugs, vinca alkaloids, anthracyclines, epidophyllotoxins, taxanes, antimetabolites, alkylating agents, antibiotics, COX-2 inhibitors, SN-38, antimitotics, antiangiogenic and apoptotoic agents, particularly doxorubicin, methotrexate, taxol, CPT-11, camptothecans, nitrogen mustards, gemcitabine, alkyl sulfonates, nitrosoureas, triazenes, folic acid analogs, pyrimidine analogs, purine analogs, platinum coordination complexes, Pseudomonas exotoxin, ricin, abrin, 5-fluorouridine, ribonuclease (RNase), DNase I, Staphylococcal enterotoxin-A, pokeweed antiviral protein, gelonin, diphtherin toxin, Pseudomonas exotoxin, and Pseudomonas endotoxin and others (see, e.g., Remington's Pharmaceutical Sciences, 19th Ed. (Mack Publishing Co. 1995); Goodman and Gilman's The Pharmacological Basis of Therapeutics (McGraw Hill, 2001); Pastan et al. (1986) Cell 47:641; Goldenberg (1994) Cancer Journal for Clinicians 44:43; U.S. Pat. No. 6,077,499; the entire disclosures of which are herein incorporated by reference). It will be appreciated that a toxin can be of animal, plant, fungal, or microbial origin, or can be created de novo by chemical synthesis.
[0099] In another embodiment, the antibody is derivatized with a radioactive isotope, such as a therapeutic radionuclide or a radionuclide suitable for detection purposes. Any of a number of suitable radioactive isotopes can be used, including, but not limited to, 1-131, Indium-111, Lutetium-171, Bismuth-212, Bismuth-213, Astatine-211, Copper-62, Copper-64, Copper-67, Yttrium-90, Iodine-125, Iodine-131, Phosphorus-32, Phosphorus-33, Scandium-47, Silver-Ill, Gallium-67, Praseodymium-142, Samarium-153, Terbium-161, Dysprosium-166, Holmium-166, Rhenium-186, Rhenium-188, Rhenium-189, Lead-212, Radium-223, Actinium-225, Iron-59, Selenium-75, Arsenic-77, Strontium-89, Molybdenum-99, Rhodium-105, Palladium-109, Praseodymium-143, Promethium-149, Erbium-169, Iridium-194, Gold-198, Gold-199, and Lead-211. In general, the radionuclide preferably has a decay energy in the range of 20 to 6,000 keV, preferably in the ranges 60 to 200 keV for an Auger emitter, 100-2,500 keV for a beta emitter, and 4,000-6,000 keV for an alpha emitter. Also preferred are radionuclides that substantially decay with generation of alpha-particles.
[0100] In other embodiments, the second agent is a detectable moiety, which can be any molecule that can be quantitatively or qualitatively observed or measured. Examples of detectable markers useful in the conjugated antibodies of this invention are radioisotopes, fluorescent dyes, or a member of a complementary binding pair, such as a member of any one of: and antigen/antibody (other than an antibody to NKG2A), lectin/carbohydrate; avidin/biotin; receptor/ligand; or molecularly imprinted polymer/print molecule systems.
[0101] The second agent may also or alternatively be a polymer, intended to increase the circulating half-life of the humanized antibody, for example. Exemplary polymers and methods to attach such polymers to peptides are illustrated in, e.g., U.S. Pat. Nos. 4,766,106; 4,179,337; 4,495,285; and 4,609,546. Additional illustrative polymers include polyoxyethylated polyols and polyethylene glycol (PEG) moieties (e.g., a full-length antibody or antibody fragment can be conjugated to one or more PEG molecules with a molecular weight of between about 1,000 and about 40,000, such as between about 2000 and about 20,000, e.g., about 3,000-12,000).
[0102] The cytotoxic agents or other compounds can be linked to the antibody directly or indirectly, using any of a large number of available methods. For example, an agent can be attached at the hinge region of the reduced antibody component via disulfide bond formation, using cross-linkers such as N-succinyl 3-(2-pyridyldithio)proprionate (SPDP), or via a carbohydrate moiety in the Fc region of the antibody (see, e.g., Yu et al. (1994) Int. J. Cancer 56: 244; Wong, Chemistry of Protein Conjugation and Cross-linking (CRC Press 1991); Upeslacis et al., "Modification of Antibodies by Chemical Methods," in Monoclonal antibodies: principles and applications, Birch et al. (eds.), pages 187-230 (Wiley-Liss, Inc. 1995); Price, "Production and Characterization of Synthetic Peptide-Derived Antibodies," in Monoclonal antibodies: Production, engineering and clinical application, Ritter et al. (eds.), pages 60-84 (Cambridge University Press 1995), Cattel et al. (1989) Chemistry today 7:51-58, Delprino et al. (1993) J. Pharm. Sci 82:699-704; Arpicco et al. (1997) Bioconjugate Chemistry 8:3; Reisfeld et al. (1989) Antibody, Immunicon. Radiopharm. 2:217; the entire disclosures of each of which are herein incorporated by reference).
[0103] Alternatively, a fusion protein comprising the anti-NKG2A antibody and a second (cytotoxic or other) polypeptide agent may be made, e.g. by recombinant techniques or peptide synthesis.
Binding Assays
[0104] The present invention provides for antibodies that bind human NKG2A, in particular humanized versions of an anti-NKG2A antibody produced by the Z270 hybridoma.
[0105] Any of a wide variety of assays can be used to assess binding of an antibody to human NKG2A. Protocols based upon ELISAs, radioimmunoassays, Western blotting, BIACORE, and other competition assays, inter alia, are suitable for use and are well known in the art.
[0106] For example, simple binding assays can be used, in which a test antibody is incubated in the presence of a target protein or epitope (e.g., NKG2A or a portion thereof), unbound antibodies are washed off, and the presence of bound antibodies is assessed using, e.g., radiolabels, physical methods such as mass spectrometry, or direct or indirect fluorescent labels detected using, e.g., cytofluorometric analysis (e.g. FACScan). Such methods are well known to those of skill in the art. Any amount of binding above the amount seen with a control, non-specific antibody indicates that the antibody binds specifically to the target.
[0107] In such assays, the ability of the test antibody to bind to the target cell or human NKG2A can be compared with the ability of a (negative) control protein, e.g. an antibody raised against a structurally unrelated antigen, or a non-Ig peptide or protein, to bind to the same target. Antibodies or fragments that bind to the target cells or NKG2A using any suitable assay with 25%, 50%, 100%, 200%, 1000%, or higher increased affinity relative to the control protein, are said to "specifically bind to" or "specifically interact with" the target, and are preferred for use in the therapeutic methods described below. The ability of a test antibody to affect the binding of a (positive) control antibody against NKG2A, e.g. Z270, or derivatives thereof, may also be assessed.
[0108] The humanized anti-NKG2A antibodies may or may not bind human NKG2C, may or may not bind human NKG2E, or may or may not bind any of human NKG2C and E. In a particular embodiment, the monoclonal antibody or fragment does not bind to other human NKG2 receptors, specifically the activating receptors NKG2C or NKG2E. The NKG2C- and NKG2E-binding properties of the antibodies of the invention can be evaluated in similar assays as those described above, simply exchanging NKG2A for the molecule of interest.
[0109] In one aspect, the invention provides for humanized versions of non-human antibodies sharing biological characteristics and/or substantial sequence identity with Z270. One exemplary biological characteristic is the binding to the Z270 epitope, i.e., the region in the extracellular domain of NKG2A to which the Z270 antibody binds. To screen for antibodies that bind to the Z270 epitope, a routine cross-blocking assay, such as that described in Antibodies, A Laboratory Manual, Cold Spring Harbor Laboratory, Ed Harlow and David Lane (1988), can be performed.
[0110] In an exemplary cross-blocking or competition assay, Z270 (control) antibody and a test antibody are admixed (or pre-adsorbed) and applied to a sample containing NKG2A. In certain embodiments, one would pre-mix the control antibodies with varying amounts of the test antibody (e.g., 1:10 or 1:100) for a period of time prior to applying to the NKG2A-containing sample. In other embodiments, the control and varying amounts of test antibody can simply be admixed during exposure to the antigen/target sample. As long as one can distinguish bound from free antibodies (e.g., by using separation or washing techniques to eliminate unbound antibodies) and the control antibody from test antibody (e.g., by using species- or isotype-specific secondary antibodies, by specifically labeling the control antibody with a detectable label, or by using physical methods such as mass spectrometry to distinguish between different compounds) one will be able to determine if the test antibody reduces the binding of the control antibody to the antigen, indicating that the test antibody recognizes substantially the same epitope as the control. In this assay, the binding of the (labeled) control antibody in the presence of a completely irrelevant antibody is the control high value. The control low value is be obtained by incubating the labeled (positive) control antibody (Z270) with unlabeled control antibody, where competition would occur and reduce binding of the labeled antibody.
[0111] In a test assay, a significant reduction in labeled antibody reactivity in the presence of a test antibody is indicative of a test antibody that recognizes the same epitope, i.e., one that "cross-reacts" with the labeled control antibody. Any test antibody or compound that reduces the binding of the labeled control to the antigen/target by at least 50% or more preferably 70%, at any ratio of control:test antibody or compound between about 1:10 and about 1:100 is considered to be an antibody or compound that binds to substantially the same epitope or determinant as the control. Preferably, such test antibody or compound will reduce the binding of the control to the antigen/target by at least 90%. Nevertheless, any compound or antibody that reduces the binding of a control antibody or compound to any measurable extent can be used in the present invention.
[0112] Similar cross-blocking assays can also be used to evaluate whether a test (humanized) antibody affects the binding of the natural ligand for human NKG2A, HLA-E, to NKG2A, by exchanging Z270 for a suitable form of HLA-E. For example, to determine whether a humanized anti-NKG2A antibody preparation reduces or blocks CD94/NKG2A interactions with HLA-E, the following test can be performed: A cell line expressing CD94/NKG2A, such as Ba/F3-CD94/NKG2A, NKL or NK92, is incubated for 30 min on ice, with increasing concentrations of a test anti-NKG2A antibody. The cells are then incubated with PE-labeled HLA-E tetramers for 30 minutes on ice, washed again, and HLA-E tetramer binding analyzed on a flow cytometer (FACScalibur, Beckton Dickinson), by standard methods. In the absence of test antibodies, the HLA-E tetramer binds to the cells. In the presence of an antibody preparation that blocks CD94/NKG2A-binding to HLA-E, there is a reduced binding of HLA-E tetramers to the cells, and such mAbs are designated "blocking antibodies".
[0113] In some aspects of the invention, e.g., where it is not desired to kill NKG2A-expressing cells, the humanized antibodies of this invention preferably do not demonstrate substantial specific binding to Fc receptors. Such antibodies may comprise constant regions of various heavy chains that are known not to bind Fc receptors. One such example is an IgG4 constant region. Alternatively, antibody fragments that do not comprise constant regions, such as Fab or F(ab')2 fragments, can be used to avoid Fc receptor binding. Fc receptor binding can be assessed according to methods known in the art, including for example testing binding of an antibody to Fc receptor protein in a BIACORE assay. Also, any other antibody type can be used in which the Fc portion is modified to minimize or eliminate binding to Fc receptors (see, e.g., WO03101485, the disclosure of which is herein incorporated by reference). Assays such as, e.g., cell based assays, to assess Fc receptor binding are well known in the art, and are described in, e.g., WO03101485.
Cytotoxicity Assays
[0114] If an anti-NKG2A antibody reduces or blocks CD94/NKG2A interactions with HLA-E, it may increase the cytotoxicity of CD94/NKG2A-restricted lymphocytes. This can be evaluated by a typical cytotoxicity assay, examples of which are described below.
[0115] The ability of an antibody to reduce CD94/NKG2A-mediated signaling can be tested in a standard 4-hour in vitro cytotoxicity assay using, e.g., NKL cells that express CD94/NKG2A, and target cells that express HLA-E. Such NKL cells do not efficiently kill targets that express HLA-E because CD94/NKG2A recognizes HLA-E, leading to initiation and propagation of inhibitory signaling that prevents lymphocyte-mediated cytolysis. Such an in vitro cytotoxicity assay can be carried out by standard methods that are well known in the art, as described for example in Coligan et al., eds., Current Protocols in Immunology, Greene Publishing Assoc. and Wiley Interscience, N.Y., (1992, 1993). The target cells are labeled with .sup.51Cr prior to addition of NKL cells, and then the killing is estimated as proportional to the release of .sup.51Cr from the cells to the medium, as a result of killing. The addition of an antibody that prevents CD94/NKG2A from binding to HLA-E results in prevention of the initiation and propagation of inhibitory signaling via CD94/NKG2A. Therefore, addition of such agents results in increases in lymphocyte-mediated killing of the target cells. This step thereby identifies agents that prevent CD94/NKG2A-induced negative signaling by, e.g., blocking ligand binding. In a particular .sup.51Cr-release cytotoxicity assay, CD94/NKG2A-expressing NKL effector-cells can kill HLA-E-negative LCL 721.221 target cells, but less well HLA-E-expressing LCL 721.221-Cw3 control cells. In contrast, YTS effector-cells that lack CD94/NKG2A kill both cell-lines efficiently. Thus, NKL effector cells kill less efficiently HLA-E.sup.+ LCL 721.221-Cw3 cells due to HLA-E-induced inhibitory signaling via CD94/NKG2A. When NKL cells are pre-incubated with blocking anti-CD94/NKG2A antibodies according to the present invention in such a .sup.51Cr-release cytotoxicity assay, HLA-E-expressing LCL 721.221-Cw3 cells are more efficiently killed, in an antibody-concentration-dependent fashion.
[0116] The inhibitory or potentiating activity of an antibody of this invention can also be assessed in any of a number of other ways, e.g., by its effect on intracellular free calcium as described, e.g., in Sivori et al., J. Exp. Med. 1997; 186:1129-1136, the disclosure of which is herein incorporated by reference. NK, T, or NKT cell activity can also be assessed using a cell based cytotoxicity assays, e.g., measuring chromium release or other parameter to assess the ability of the antibody to stimulate NK cells to kill target cells such as P815, K562 cells, or appropriate tumor cells as disclosed in Sivori et al., J. Exp. Med. 1997; 186:1129-1136; Vitale et al., J. Exp. Med. 1998; 187:2065-2072; Pessino et al. J. Exp. Med. 1998; 188:953-960; Neri et al. Clin. Diag. Lab. Immun. 2001; 8:1131-1135; Pende et al. J. Exp. Med. 1999; 190:1505-1516, the entire disclosures of each of which are herein incorporated by reference.
[0117] In one embodiment, an antibody preparation causes at least a 10% augmentation in the cytotoxicity of a CD94/NKG2A-restricted lymphocyte, preferably at least a 40% or 50% augmentation in NK cytotoxicity, or more preferably at least a 70% augmentation in NK cytotoxicity.
[0118] The activity of a cytotoxic lymphocyte can also be addressed using a cytokine-release assay, wherein NK cells are incubated with the antibody to stimulate the cytokine production of the NK cells (for example IFN-.gamma. and TNF-.alpha. production). In an exemplary protocol, IFN-.gamma. production from PBMC is assessed by cell surface and intracytoplasmic staining and analysis by flow cytometry after 4 days in culture. Briefly, Brefeldin A (Sigma Aldrich) is added at a final concentration of 5 .mu.g/ml for the last 4 hours of culture. The cells are then incubated with anti-CD3 and anti-CD56 mAb prior to permeabilization (IntraPrep.TM.; Beckman Coulter) and staining with PE-anti-IFN-.gamma. or PE-IgG1 (Pharmingen). GM-CSF and IFN-.gamma. production from polyclonal activated NK cells are measured in supematants using ELISA (GM-CSF: DuoSet Elisa, R&D Systems, Minneapolis, Minn., IFN-: OptEIA set, Pharmingen).
[0119] In a particular aspect, the invention provides antibodies that are more capable of, or more effective in, increasing the cytotoxicity of CD94/NKG2A-restricted lymphocytes, potentiating cytotoxic activity of a CD94/NKG2A-restricted lymphocyte, or reducing or inhibiting CD94/NKG2A-mediated signaling, than the original, non-humanized antibody and/or a chimeric version thereof. Such antibodies can be, for example, at least 2%, at least 5%, at least 10%, at least 15%, or at least 20% more capable or effective an original, non-humanized antibody or chimeric version thereof.
Recombinant Methods
[0120] The invention also provides isolated nucleic acids encoding the anti-NKG2A antibodies described herein, as well as vectors and host cells comprising such nucleic acids. Also provided for are methods of producing such anti-NKG2A antibodies using recombinant techniques such as, e.g., culturing suitable host cells comprising such nucleic acids or vectors so that the nucleic acid is expressed and the humanized antibody produced. Before culturing, the host cell may, for example, be co-transfected with a vector comprising nucleic acids encoding a variable heavy domain and with a vector comprising nucleic acid encoding a variable light domain. Additionally, the antibody may be recovered and/or purified from the host cell culture using known techniques. Useful vectors, host cells, and techniques are further described below and in the Examples. Additionally, strategies for expressing humanized anti-NKG2A antibodies are outlined in FIGS. 4-6.
[0121] Generally, for recombinant production of the antibody, a nucleic acid encoding it is isolated and inserted into a replicable vector for further cloning (amplification of the DNA) or for expression, typically operably linked to one or more expression control elements. DNA encoding the monoclonal antibody is readily isolated and sequenced using conventional procedures (e.g., by using oligonucleotide probes that are capable of binding specifically to genes encoding the heavy and light chains of the antibody). Many vectors are known and available. The vector components generally include, but are not limited to, one or more of the following: a signal sequence, an origin of replication, one or more marker genes, an enhancer element, a promoter, and a transcription-termination sequence.
Signal Sequence Component
[0122] The anti-NKG2A antibody of this invention may be produced recombinantly not only directly, but also as a fusion polypeptide with a heterologous polypeptide, which is preferably a signal sequence or other polypeptide having a specific cleavage site at the N-terminus of the mature protein or polypeptide. The heterologous signal sequence selected preferably is one that is recognized and processed (i.e., cleaved by a signal peptidase) by the host cell. See, e.g., Example 2 for exemplary signal sequences for expression of humanized heavy and/or light chains of anti-NKG2A antibodies such as humanized Z270 antibodies.
[0123] For prokaryotic host cells that do not recognize and process the native anti-NKG2A antibody signal sequence, the signal sequence is substituted by a prokaryotic signal sequence selected, for example, from the group of the alkaline phosphatase, penicillinase, Ipp, or heat-stable enterotoxin II leaders. For yeast secretion the native signal sequence may be substituted by, e.g., the yeast invertase leader, alpha-factor leader (including Saccharomyces and Kluyveromyces alpha-factor leaders), acid-phosphatase leader, the C. albicans glucoamylase leader, or the signal described in WO 1990/13646. In mammalian cell expression, mammalian signal sequences as well as viral secretory leaders, for example, the herpes simplex gD signal, are available.
[0124] The DNA for such precursor region is ligated in reading frame to DNA encoding the anti-NKG2A antibody.
Origin of Replication Component
[0125] Both expression and cloning vectors contain a nucleic acid sequence that enables the vector to replicate in one or more selected host cells. Generally, in cloning vectors this sequence is one that enables the vector to replicate independently of the host chromosomal DNA, and includes origins of replication or autonomously replicating sequences. Such sequences are well known for a variety of bacteria, yeast, and viruses. The origin of replication from the plasmid pBR322 is suitable for most Gram-negative bacteria, the 2p plasmid origin is suitable for yeast, and various viral origins (SV40, polyoma, adenovirus, VSV, EBV, or BPV) are useful for cloning vectors in mammalian cells. Generally, the origin of replication component is not needed for mammalian expression vectors (the SV40 origin may typically be used only because it contains the early promoter).
Selection Gene Component
[0126] Expression and cloning vectors may contain a selection gene, also termed a selectable marker. Typical selection genes encode proteins that (a) confer resistance to antibiotics or other toxins, e.g., ampicillin, neomycin, methotrexate, or tetracycline, (b) complement auxotrophic deficiencies, or (c) supply critical nutrients not available from complex media, e.g., the gene encoding D-alanine racemase for Bacilli.
[0127] One example of a selection scheme utilizes a drug to arrest growth of a host cell. Those cells that are successfully transformed with a heterologous gene produce a protein conferring drug resistance and thus survive the selection regimen. Examples of such dominant selection use the drugs neomycin, mycophenolic acid and hygromycin.
[0128] Another example of suitable selectable markers for mammalian cells are those that enable the identification of cells competent to take up the anti-NKG2A antibody-encoding nucleic acid, such as DHFR, thymidine kinase, metallothionein-I and -II, preferably primate metallothionein genes, aderosine deaminase, omithine decarboxylase, etc.
[0129] For example, cells transformed with the DHFR selection gene are first identified by culturing all of the transformants in a culture medium that contains methotrexate (Mtx), a competitive antagonist of DHFR. An appropriate host cell when wild-type DHFR is employed is the Chinese hamster ovary (CHO) cell line deficient in DHFR activity.
[0130] Alternatively, host cells (particularly wild-type hosts that contain endogenous DHFR) transformed or co-transformed with DNA sequences encoding anti-NKG2A antibody, wild-type DHFR protein, and another selectable marker such as aminoglycoside 3'-phosphotransferase (APH) can be selected by cell growth in medium containing a selection agent for the selectable marker such as an aminoglycosidic antibiotic, e.g., kanamycin, neomycin, or G418. See U.S. Pat. No. 4,965,199.
[0131] A suitable selection gene for use in yeast is the trp1 gene present in the yeast plasmid YRp7 (Stinchcomb et al., Nature, 282:39 (1979)). The trp1 gene provides a selection marker for a mutant strain of yeast lacking the ability to grow in tryptophan, for example, ATCC No. 44076 or PEP4-1. Jones, Genetics, 85:12 (1977). The presence of the trp1 lesion in the yeast host cell genome then provides an effective environment for detecting transformation by growth in the absence of tryptophan. Similarly, Leu2-deficient yeast strains (ATCC 20,622 or 38,626) are complemented by known plasmids bearing the Leu2 gene.
[0132] In addition, vectors derived from the 1.6-.mu.m circular plasmid pKD1 can be used for transformation of Kluyveromyces yeasts. Alternatively, an expression system for large-scale production of recombinant calf chymosin was reported for K. lactis (Van den Berg, Bio/Technology, 8:135 (1990). Stable multi-copy expression vectors for secretion of mature recombinant human serum albumin by industrial strains of Kluyveromyces have also been disclosed. Fleer et al., Bio/Technolog, 9: 968-975 (1991).
Promoter Component
[0133] Expression and cloning vectors usually contain a promoter that is recognized by the host organism and is operably linked to the anti-NKG2A antibody-encoding nucleic acid. Promoters suitable for use with prokaryotic hosts include the phoA promoter, .beta.-lactamase and lactose promoter systems, alkaline phosphatase, a tryptophan (trp) promoter system, and hybrid promoters such as the tac promoter. However, other known bacterial promoters are suitable. Promoters for use in bacterial systems also will contain a Shine-Dalgamo (S.D.) sequence operably linked to the DNA encoding the anti-NKG2A antibody.
[0134] Various promoter sequences are known for eukaryotes. Virtually all eukaryotic genes have an AT-rich region located approximately 25 to 30 bases upstream from the site where transcription is initiated. Another sequence found 70 to 80 bases upstream from the start of transcription of many genes is a CNCAAT region where N may be any nucleotide. At the 3' end of most eukaryotic genes is an AATAAA sequence that may be the signal for addition of the poly-A tail to the 3' end of the coding sequence. All of these sequences are suitably inserted into eukaryotic expression vectors. Examples of suitable promoting sequences for use with yeast hosts include the promoters for 3-phosphoglycerate kinase or other glycolytic enzymes, such as enolase, glyceraldehyde-3-phosphate dehydrogenase, hexokinase, pyruvate decarboxylase, phosphofructokinase, glucose-6-phosphate isomerase, 3-phosphoglycerate mutase, pyruvate kinase, triosephosphate isomerase, phosphoglucose isomerase, and glucokinase.
[0135] Other yeast promoters, which are inducible promoters having the additional advantage of transcription controlled by growth conditions, are the promoter regions for alcohol dehydrogenase 2, isocytochrome C, acid phosphatase, degradative enzymes associated with nitrogen metabolism, metallothionein, glyceraldehyde-3-phosphate dehydrogenase, and enzymes responsible for maltose and galactose utilization. Suitable vectors and promoters for use in yeast expression are further described in EP73657. Yeast enhancers also are advantageously used with yeast promoters.
[0136] Anti-NKG2A antibody transcription from vectors in mammalian host cells is controlled, for example, by promoters obtained from the genomes of viruses such as polyoma virus, fowipox virus, adenovirus (such as Adenovirus 2), bovine papilloma virus, avian sarcoma virus, cytomegalovirus (CMV), a retrovirus, hepatitis-B virus and most preferably Simian Virus 40 (SV40), heterologous mammalian promoters, e.g., the actin promoter or an immunoglobulin promoter, and heat-shock promoters, provided such promoters are compatible with the host cell systems.
[0137] The early and late promoters of the SV40 virus are conveniently obtained as an SV40 restriction fragment that also contains the SV40 viral origin of replication. The immediate early promoter of the human cytomegalovirus is conveniently obtained as a HindIII E restriction fragment. A system for expressing DNA in mammalian hosts using the bovine papilloma virus as a vector is disclosed in U.S. Pat. No. 4,419,446. A modification of this system is described in U.S. Pat. No. 4,601,978. See also Reyes et al., Nature, 297:598-601 (1982) on expression of human .beta.-interferon cDNA in mouse cells under the control of a thymidine kinase promoter from herpes simplex virus. Alternatively, the rous sarcoma virus long-terminal repeat can be used as the promoter.
Enhancer Element Component
[0138] Transcription of a DNA encoding the anti-NKG2A antibody of this invention by higher eukaryotes is often increased by inserting an enhancer sequence into the vector. Many enhancer sequences are now known from mammalian genes (globin, elastase, albumin, .alpha.-fetoprotein, and insulin). Typically, however, one will use an enhancer from a eukaryotic cell virus. Examples include the SV40 enhancer on the late side of the replication origin (bp 100-270), the cytomegalovirus early-promoter enhancer, the polyoma enhancer on the late side of the replication origin, and adenovirus enhancers. See also Yaniv, Nature, 297:17-18 (1982) on enhancing elements for activation of eukaryotic promoters. The enhancer may be spliced into the vector at a position 5' or 3' to the anti-NKG2A antibody-encoding sequence, but is preferably located at a site 5' from the promoter.
Transcription Termination Component
[0139] Expression vectors used in eukaryotic host cells (for example, yeast, fungi, insect, plant, animal, human, or nucleated cells from other multicellular organisms) will also contain sequences necessary for the termination of transcription and for stabilizing the mRNA. Such sequences are commonly available from the 5' end, occasionally 3' end, of untranslated regions of eukaryotic or viral DNAs or cDNAs. These regions contain nucleotide segments transcribed as polyadenylated fragments in the untranslated portion of the mRNA encoding anti-NKG2A antibody. One useful transcription termination component is the bovine growth hormone polyadenylation region. See WO 1994/11026 and the expression vector disclosed therein.
Selection and Transformation of Host Cells
[0140] Suitable host cells for cloning or expressing the DNA in the vectors herein are the prokaryote, yeast, or higher eukaryote cells described above. Suitable prokaryotes for this purpose include eubacteria, such as Gram-negative or Gram-positive organisms, for example, Enterobacteriaceae such as Escherichia, e.g., E. coli, Enterobacter, Erwinia, Klebsiella, Proteus, Salmonella, e.g., Salmonella typhimurium, Serratia, e.g., Serratia marcescans, and Shigella, as well as Bacilli such as B. subtilis and B. licheniformis (e.g. B. licheniformis 41 P disclosed in DD 266,710 published 12 Apr. 1989), Pseudomonas such as P. aeruginosa, and Streptomyces. One preferred E. coli cloning host is E. coli 294 (ATCC 31,446), although other strains such as E. coli B, E. coli X1776 (ATCC 31,537), and E. coli W3110 (ATCC 27,325) are suitable. These examples are illustrative rather than limiting.
[0141] In addition to prokaryotes, eukaryotic microbes such as filamentous fungi or yeast are suitable cloning or expression hosts for anti-NKG2A antibody-encoding vectors. Saccharomyces cerevisiae, or common baker's yeast, is the most commonly used among lower eukaryotic host microorganisms. However, a number of other genera, species, and strains are commonly available and useful herein, such as Schizosaccharomyces pombe; Kluyveromyces hosts such as, e.g., K. lactis, K. fragilis (ATCC 12,424), K. bulgaricus (ATCC 16,045), K. wickeramii (ATCC 24,178), K. waltii (ATCC 56,500), K. drosophilarum (ATCC 36,906), K. thermotolerans, and K. marxianus; yarrowia (EP 402,226); Pichia pastoris (EP 183,070); Candida; Trichoderma reesia (EP 244,234); Neurospora crassa; Schwanniomyces such as Schwanniomyces occidentalis; and filamentous fungi such as, e.g., Neurospora, Penicillium, Tolypocladium, and Aspergillus hosts such as A. nidulans and A. niger.
[0142] Suitable host cells for the expression of glycosylated anti-NKG2A antibody are derived from multicellular organisms. Examples of invertebrate cells include plant and insect cells. Numerous baculoviral strains and variants and corresponding permissive insect host cells from hosts such as Spodoptera frugiperda (caterpillar), Aedes aegypti (mosquito), Aedes albopictus (mosquito), Drosophila melanogaster (fruitfly), and Bombyx mori have been identified. A variety of viral strains for transfection are publicly available, e.g. the L-1 variant of Autographa californica NPV and the Bm-5 strain of Bombyx mori NPV, and such viruses may be used as the virus herein according to the present invention, particularly for transfection of Spodoptera frugiperda cells.
[0143] Plant cell cultures of cotton, corn, potato, soybean, petunia, tomato, and tobacco can also be utilized as hosts.
[0144] However, interest has been greatest in vertebrate cells, and propagation of vertebrate cells in culture (tissue culture) has become a routine procedure. Examples of useful mammalian host cell lines are monkey kidney CV1 line transformed by SV40 (COS-7, ATCC CRL 1651); human embryonic kidney (HEK) line (293 or 293 cells subcloned for growth in suspension culture, Graham et al., J. Gen Virol., 36:59 (1977)); baby hamster kidney cells (BHK, ATCC CCL 10); Chinese hamster ovary cells/-DHFR (CHO, Urlaub et al., Proc. Natl. Acad. Sci. USA, 77:4216 (1980), including DG44 (Urlaub et al., Som. Cell and Mol. Gen., 12: 555-566 (1986)) and DP12 cell lines); mouse sertoli cells (TM4, Mather, Biol. Reprod., 23:243-251 (1980)); monkey kidney cells (CV1 ATCC CCL 70); African green monkey kidney cells (VERO-76, ATCC CRL-1587); human cervical carcinoma cells (HELA, ATCC CCL 2); canine kidney cells (MDCK, ATCC CCL 34); buffalo rat liver cells (BRL 3A, ATCC CRL 1442); human lung cells (W138, ATCC CCL 75); human liver cells (Hep G2, HB 8065); mouse mammary tumor (MMT 060562, ATCC CCL51); TRI cells (Mather et al., Annals N.Y. Acad. Sci., 383:44-68 (1982)); MRC 5 cells; FS4 cells; and a human hepatoma line (Hep G2).
[0145] Host cells are transformed with the above-described expression or cloning vectors for anti-NKG2A antibody production and cultured in conventional nutrient media modified as appropriate for inducing promoters, selecting transformants, or amplifying the genes encoding the desired sequences.
Culturing the Host Cells
[0146] The host cells used to produce the anti-NKG2A antibody of this invention may be cultured in a variety of media. Commercially available media such as Ham's F10 (Sigma), Minimal Essential Medium ((MEM), Sigma), RPMI-1640 (Sigma), FreeStyle.TM. (Cibco) and Dulbecco's Modified Eagle's Medium ((DMEM), Sigma) are suitable for culturing the host cells. In addition, any of the media described, for example, in Ham et al., Meth. Enz. 58:44 (1979); Barnes et al., Anal. Biochem., 102:255 (1980); U.S. Pat. Nos. 4,767,704; 4,657,866; 4,927,762; 4,560,655; or 5,122,469; WO 1990/03430; WO 1987/00195; or U.S. Pat. Re. 30,985 may be used as culture media for the host cells. Any of these media may be supplemented as necessary with hormones and/or other growth factors (such as insulin, transferrin, or epidermal growth factor), salts (such as sodium chloride, calcium, magnesium, and phosphate), buffers (such as HEPES), nucleotides (such as adenosine and thymidine), antibiotics (such as GENTAMYCIN.TM. drug), trace elements (defined as inorganic compounds usually present at final concentrations in the micromolar range), and glucose or an equivalent energy source. Any other necessary supplements may also be included at appropriate concentrations that would be known to those skilled in the art. The culture conditions, such as temperature, pH, and the like, are those previously used with the host cell selected for expression, and will be apparent to the ordinarily skilled artisan.
Purification of Anti-NKG2A Antibody
[0147] When using recombinant techniques, the antibody can be produced intracellularty or in the periplasmic space, or directly secreted into the medium. If the antibody is produced intracellularly, as a first step, the particulate debris, either host cells or lysed fragments, is removed, for example, by centrifugation or ultrafiltration. Carter et al., Bio/Technology, 10: 163-167 (1992) describes a procedure for isolating antibodies that are secreted to the periplasmic space of E. coli. Briefly, cell paste is thawed in the presence of sodium acetate (pH 3.5), EDTA, and phenylmethylsulfonylfluoride (PMSF) over about 30 min. Cell debris can be removed by centrifugation. Where the antibody is secreted into the medium, superatants from such expression systems are generally first concentrated using a commercially available protein concentration filter, for example, an AMICON.TM. or MILLIPORE PELLICON.TM. ultrafiltration unit. A protease inhibitor such as phenylmethylsulphonyl fluoride (PMSF) may be included in any of the foregoing steps to inhibit proteolysis, and antibiotics may be included to prevent the growth of adventitious contaminants.
[0148] The antibody composition prepared from the cells can be purified using, for example, hydroxylapatite chromatography, gel electrophoresis, dialysis, and affinity chromatography, with affinity chromatography being the preferred purification technique. The suitability of protein A as an affinity ligand depends on the species and isotype of any immunoglobulin Fc domain that is present in the antibody. Protein A can be used to purify antibodies that are based on human gamma1, gamma, or gamma4 heavy chains (iUndmark et al., J. Immunol. Meth., 62:1-13 (1983)). Protein G is recommended for all mouse isotypes and for human y3 (Guss et al., EMBO J., 5:15671575 (1986)). The matrix to which the affinity ligand is attached is most often agarose, but other matrices are available. Mechanically stable matrices such as controlled-pore glass or poly(styrenedivinyl)benzene allow for faster flow rates and shorter processing times than can be achieved with agarose. Where the antibody comprises a CH3 domain, the BAKERBOND ABX.TM. resin (J. T. Baker, Phillipsburg, N.J.) is useful for purification. Other techniques for protein purification such as fractionation on an ion-exchange column, ethanol precipitation, reverse-phase HPLC, chromatography on silica, chromatography on heparin SEPHAROSE.TM., chromatography on an anion- or cation-exchange resin (such as a polyaspartic acid column), chromatofocusing, SDS-PAGE, and ammonium-sulfate precipitation are also available depending on the antibody to be recovered.
[0149] An exemplary Protein A-based purification method for Z270 or humanized versions thereof is described in Example 6.
Pharmaceutical Formulations
[0150] In one embodiment, the present invention provides pharmaceutical composition comprising antibodies as described herein together with one or more carriers.
[0151] Accordingly, one object of the invention is to provide a pharmaceutical formulation comprising such an antibody which is present in a concentration from 1 mg/ml to 500 mg/ml, and wherein said formulation has a pH from 2.0 to 10.0. The formulation may further comprise a buffer system, preservative(s), tonicity agent(s), chelating agent(s), stabilizers and surfactants. In one embodiment, the pharmaceutical formulation is an aqueous formulation, i.e., formulation comprising water. Such formulation is typically a solution or a suspension. In a further embodiment, the pharmaceutical formulation is an aqueous solution. The term "aqueous formulation" is defined as a formulation comprising at least 50% w/w water. Likewise, the term "aqueous solution" is defined as a solution comprising at least 50% w/w water, and the term "aqueous suspension" is defined as a suspension comprising at least 50% w/w water.
[0152] In another embodiment, the pharmaceutical formulation is a freeze-dried formulation, whereto the physician or the patient adds solvents and/or diluents prior to use.
[0153] In another embodiment, the pharmaceutical formulation is a dried formulation (e.g. freeze-dried or spray-dried) ready for use without any prior dissolution.
[0154] In a further aspect, the pharmaceutical formulation comprises an aqueous solution of such an antibody, and a buffer, wherein the antibody is present in a concentration from 1 mg/ml or above, and wherein said formulation has a pH from about 2.0 to about 10.0.
[0155] In another embodiment, the pH of the formulation is in the range selected from the list consisting of from about 2.0 to about 10.0, about 3.0 to about 9.0, about 4.0 to about 8.5, about 5.0 to about 8.0, and about 5.5 to about 7.5.
[0156] In a further embodiment, the buffer is selected from the group consisting of sodium acetate, sodium carbonate, citrate, glycylglycine, histidine, glycine, lysine, arginine, sodium dihydrogen phosphate, disodium hydrogen phosphate, sodium phosphate, and tris(hydroxymethyl)-aminomethan, bicine, tricine, malic acid, succinate, maleic acid, fumaric acid, tartaric acid, aspartic acid or mixtures thereof. Each one of these specific buffers constitutes an alternative embodiment of the invention.
[0157] In a further embodiment, the formulation further comprises a pharmaceutically acceptable preservative. The preservative may be selected from, e.g., the group consisting of phenol, o-cresol, m-cresol, p-cresol, methyl p-hydroxybenzoate, propyl p-hydroxybenzoate, 2-phenoxyethanol, butyl p-hydroxybenzoate, 2-phenylethanol, benzyl alcohol, chlorobutanol, and thiomerosal, bronopol, benzoic acid, imidurea, chlorohexidine, sodium dehydroacetate, chlorocresol, ethyl p-hydroxybenzoate, benzethonium chloride, chlorphenesine (3p-chlorphenoxypropane-1,2-diol) or mixtures thereof. The preservative may, e.g., be present in a concentration from 0.1 mg/ml to 20 mg/ml, from 0.1 mg/ml to 5 mg/ml, from 5 mg/ml to 10 mg/ml, or from 10 mg/ml to 20 mg/ml. Each one of these specific preservatives constitutes an alternative embodiment of the invention. The use of a preservative in pharmaceutical compositions is well-known to the skilled person. For convenience reference is made to Remington: The Science and Practice of Pharmacy, 19.sup.th edition, 1995.
[0158] In a further embodiment, the formulation further comprises an isotonic agent. The isotonic agent may be, e.g., selected from the group consisting of a salt (e.g. sodium chloride), a sugar or sugar alcohol, an amino acid (e.g. L-glycine, L-histidine, arginine, lysine, isoleucine, aspartic acid, tryptophan, threonine), an alditol (e.g. glycerol (glycerine), 1,2-propanediol (propyleneglycol), 1,3-propanediol, 1,3-butanediol) polyethyleneglycol (e.g. PEG400), or mixtures thereof. Any sugar such as mono-, di-, or polysaccharides, or water-soluble glucans, including for example fructose, glucose, mannose, sorbose, xylose, maltose, lactose, sucrose, trehalose, dextran, pullulan, dextrin, cyclodextrin, soluble starch, hydroxyethyl starch and carboxymethylcellulose-Na may be used. In one embodiment, the sugar additive is sucrose. Sugar alcohol is defined as a C4-C8 hydrocarbon having at least one --OH group and includes, for example, mannitol, sorbitol, inositol, galactitol, dulcitol, xylitol, and arabitol. In one embodiment, the sugar alcohol additive is mannitol. The sugars or sugar alcohols mentioned above may be used individually or in combination. There is no fixed limit to the amount used, as long as the sugar or sugar alcohol is soluble in the liquid preparation and does not adversely effect the stabilizing effects achieved using the methods of the invention. The sugar or sugar alcohol concentration can, e.g., be between about 1 mg/ml and about 150 mg/ml. The isotonic agent can be present in a concentration from, e.g., 1 mg/ml to 50 mg/ml, from 1 mg/ml to 7 mg/ml, from 8 mg/ml to 24 mg/ml, or from 25 mg/ml to 50 mg/ml. Each one of these specific isotonic agents constitutes an alternative embodiment of the invention. The use of an isotonic agent in pharmaceutical compositions is well-known to the skilled person. For convenience reference is made to Remington: The Science and Practice of Pharmacy, 19.sup.th edition, 1995.
[0159] In a further embodiment, the formulation also comprises a chelating agent. The chelating agent can, for example, be selected from salts of ethylenediaminetetraacetic acid (EDTA), citric acid, and aspartic acid, and mixtures thereof. The chelating agent may, for example, be present in a concentration from 0.1 mg/ml to 5 mg/ml, from 0.1 mg/ml to 2 mg/ml, or from 2 mg/ml to 5 mg/ml. Each one of these specific chelating agents constitutes an alternative embodiment of the invention. The use of a chelating agent in pharmaceutical compositions is well-known to the skilled person. For convenience reference is made to Remington: The Science and Practice of Pharmacy, 19.sup.th edition, 1995.
[0160] In a further embodiment of the invention the formulation further comprises a stabilizer. The use of a stabilizer in pharmaceutical compositions is well-known to the skilled person. For convenience reference is made to Remington: The Science and Practice of Pharmacy, 19.sup.th edition, 1995. More particularly, compositions of the invention can be stabilized liquid pharmaceutical compositions whose therapeutically active components include a polypeptide that possibly exhibits aggregate formation during storage in liquid pharmaceutical formulations. By "aggregate formation" is intended a physical interaction between the polypeptide molecules that results in formation of oligomers, which may remain soluble, or large visible aggregates that precipitate from the solution. By "during storage" is intended a liquid pharmaceutical composition or formulation once prepared, is not immediately administered to a subject. Rather, following preparation, it is packaged for storage, either in a liquid form, in a frozen state, or in a dried form for later reconstitution into a liquid form or other form suitable for administration to a subject. By "dried form" is intended the liquid pharmaceutical composition or formulation is dried either by freeze drying (i.e., lyophilization; see, for example, Williams and Polli (1984) J. Parenteral Sci. Technol. 38:48-59), spray drying (see Masters (1991) in Spray-Drying Handbook (5th ed; Longman Scientific and Technical, Essez, U.K.), pp. 491-676; Broadhead et al. (1992) Drug Devel. Ind. Pharm. 18:1169-1206; and Mumenthaler et al. (1994) Pharm. Res. 11:12-20), or air drying (Carpenter and Crowe (1988) Cryobiology 25:459-470; and Roser (1991) Biopharm. 4:47-53). Aggregate formation by a polypeptide during storage of a liquid pharmaceutical composition can adversely affect biological activity of that polypeptide, resulting in loss of therapeutic efficacy of the pharmaceutical composition. Furthermore, aggregate formation may cause other problems such as blockage of tubing, membranes, or pumps when the polypeptide-containing pharmaceutical composition is administered using an infusion system.
[0161] The pharmaceutical compositions of the invention may further comprise an amount of an amino acid base sufficient to decrease aggregate formation by the polypeptide during storage of the composition. By "amino acid base" is intended an amino acid or a combination of amino acids, where any given amino acid is present either in its free base form or in its salt form. Where a combination of amino acids is used, all of the amino acids may be present in their free base forms, all may be present in their salt forms, or some may be present in their free base forms while others are present in their salt forms. In one embodiment, amino acids to use in preparing the compositions of the invention are those carrying a charged side chain, such as arginine, lysine, aspartic acid, and glutamic acid. Any stereoisomer (i.e., L, D, or a mixture thereof) of a particular amino acid (e.g. methionine, histidine, imidazole, arginine, lysine, isoleucine, aspartic acid, tryptophan, threonine and mixtures thereof) or combinations of these stereoisomers, may be present in the pharmaceutical compositions of the invention so long as the particular amino acid is present either in its free base form or its salt form. In one embodiment the L-stereoisomer is used. Compositions of the invention may also be formulated with analogues of these amino acids. By "amino acid analogue" is intended a derivative of the naturally occurring amino acid that brings about the desired effect of decreasing aggregate formation by the polypeptide during storage of the liquid pharmaceutical compositions of the invention. Suitable arginine analogues include, for example, aminoguanidine, omithine and N-monoethyl L-arginine, suitable methionine analogues include ethionine and buthionine and suitable cysteine analogues include S-methyl-L cysteine. As with the other amino acids, the amino acid analogues are incorporated into the compositions in either their free base form or their salt form. In a further embodiment of the invention the amino acids or amino acid analogues are used in a concentration, which is sufficient to prevent or delay aggregation of the protein.
[0162] In a further embodiment of the invention methionine (or other sulphuric amino acids or amino acid analogous) may be added to inhibit oxidation of methionine residues to methionine sulfoxide when the polypeptide acting as the therapeutic agent is a polypeptide comprising at least one methionine residue susceptible to such oxidation. By "inhibit" is intended minimal accumulation of methionine oxidized species over time. Inhibiting methionine oxidation results in greater retention of the polypeptide in its proper molecular form. Any stereoisomer of methionine (L or D) or combinations thereof can be used. The amount to be added should be an amount sufficient to inhibit oxidation of the methionine residues such that the amount of methionine sulfoxide is acceptable to regulatory agencies. Typically, this means that the composition contains no more than about 10% to about 30% methionine sulfoxide. Generally, this can be achieved by adding methionine such that the ratio of methionine added to methionine residues ranges from about 1:1 to about 1000:1, such as 10:1 to about 100:1.
[0163] In a further embodiment, the formulation further comprises a stabilizer selected from the group of high molecular weight polymers or low molecular compounds. In a further embodiment of the invention the stabilizer is selected from polyethylene glycol (e.g. PEG 3350), polyvinyl alcohol (PVA), polyvinylpyrrolidone, carboxy-/hydroxycellulose or derivates thereof (e.g. HPC, HPC-SL, HPC-L and HPMC), cyclodextrins, sulphur-containing substances as monothioglycerol, thioglycolic acid and 2-methylthioethanol, and different salts (e.g. sodium chloride). Each one of these specific stabilizers constitutes an alternative embodiment of the invention.
[0164] The pharmaceutical compositions may also comprise additional stabilizing agents, which further enhance stability of a therapeutically active polypeptide therein. Stabilizing agents of particular interest to the present invention include, but are not limited to, methionine and EDTA, which protect the polypeptide against methionine oxidation, and a nonionic surfactant, which protects the polypeptide against aggregation associated with freeze-thawing or mechanical shearing.
[0165] In a further embodiment, the formulation further comprises a surfactant. The surfactant may, for example, be selected from a detergent, ethoxylated castor oil, polyglycolyzed glycerides, acetylated monoglycerides, sorbitan fatty acid esters, polyoxypropylene-polyoxyethylene block polymers (eg. poloxamers such as Pluronic F68, poloxamer 188 and 407, Triton X-100), polyoxyethylene sorbitan fatty acid esters, polyoxyethylene and polyethylene derivatives such as alkylated and alkoxylated derivatives (tweens, e.g. Tween-20, Tween-40, Tween-80 and Brij-35), monoglycerides or ethoxylated derivatives thereof, diglycerides or polyoxyethylene derivatives thereof, alcohols, glycerol, lectins and phospholipids (eg. phosphatidyl serine, phosphatidyl choline, phosphatidyl ethanolamine, phosphatidyl inositol, diphosphatidyl glycerol and sphingomyelin), derivates of phospholipids (eg. dipalmitoyl phosphatidic acid) and lysophospholipids (eg. palmitoyl lysophosphatidyl-L-serine and 1-acyl-sn-glycero-3-phosphate esters of ethanolamine, choline, serine or threonine) and alkyl, alkoxyl (alkyl ester), alkoxy (alkyl ether)-derivatives of lysophosphatidyl and phosphatidylcholines, e.g. lauroyl and myristoyl derivatives of lysophosphatidylcholine, dipalmitoylphosphatidylcholine, and modifications of the polar head group, that is cholines, ethanolamines, phosphatidic acid, serines, threonines, glycerol, inositol, and the positively charged DODAC, DOTMA, DCP, BISHOP, lysophosphatidylserine and lysophosphatidylthreonine, and glycerophospholipids (eg. cephalins), glyceroglycolipids (eg. galactopyransoide), sphingoglycolipids (eg. ceramides, gangliosides), dodecylphosphocholine, hen egg lysolecithin, fusidic acid derivatives--(e.g. sodium tauro-dihydrofusidate etc.), long-chain fatty acids and salts thereof C6-C12 (eg. oleic acid and caprylic acid), acylcamitines and derivatives, N.sup..alpha.-acylated derivatives of lysine, arginine or histidine, or side-chain acylated derivatives of lysine or arginine, Na-acylated derivatives of dipeptides comprising any combination of lysine, arginine or histidine and a neutral or acidic amino acid, Na-acylated derivative of a tripeptide comprising any combination of a neutral amino acid and two charged amino acids, DSS (docusate sodium, CAS registry no [577-11-7]), docusate calcium, CAS registry no [128-49-4]), docusate potassium, CAS registry no [7491-09-0]), SDS (sodium dodecyl sulphate or sodium lauryl sulphate), sodium caprylate, cholic acid or derivatives thereof, bile acids and salts thereof and glycine or taurine conjugates, ursodeoxycholic acid, sodium cholate, sodium deoxycholate, sodium taurocholate, sodium glycocholate, N-Hexadecyl-N, N-dimethyl-3-ammonio-1-propanesulfonate, anionic (alkyl-aryl-sulphonates) monovalent surfactants, zwitterionic surfactants (e.g. N-alkyl-N,N-dimethylammonio-1-propanesulfonates, 3-cholamido-1-propyldimethylammonio-1-propanesulfonate, cationic surfactants (quaternary ammonium bases) (e.g. cetyl-trimethylammonium bromide, cetylpyridinium chloride), non-ionic surfactants (eg. Dodecyl .beta.-D-glucopyranoside), poloxamines (eg. Tetronic's), which are tetrafunctional block copolymers derived from sequential addition of propylene oxide and ethylene oxide to ethylenediamine, or the surfactant may be selected from the group of imidazoline derivatives, or mixtures thereof. Each one of these specific surfactants constitutes an alternative embodiment of the invention.
[0166] The use of a surfactant in pharmaceutical compositions is well-known to the skilled person. For convenience reference is made to Remington: The Science and Practice of Pharmacy, 19.sup.th edition, 1995.
[0167] In a further embodiment, the formulation further comprises protease inhibitors such as EDTA (ethylenediamine tetraacetic acid) and benzamidineHCl, but other commercially available protease inhibitors may also be used. The use of a protease inhibitor is particular useful in pharmaceutical compositions comprising zymogens of proteases in order to inhibit autocatalysis.
[0168] It is possible that other ingredients may be present in the peptide pharmaceutical formulation of the present invention. Such additional ingredients may include wetting agents, emulsifiers, antioxidants, bulking agents, tonicity modifiers, chelating agents, metal ions, oleaginous vehicles, proteins (e.g., human serum albumin, gelatine or proteins) and a zwitterion (e.g., an amino acid such as betaine, taurine, arginine, glycine, lysine and histidine). Such additional ingredients, of course, should not adversely affect the overall stability of the pharmaceutical formulation of the present invention.
[0169] Pharmaceutical compositions containing an antibody according to the present invention may be administered to a patient in need of such treatment at several sites, for example, at topical sites, for example, skin and mucosal sites, at sites which bypass absorption, for example, administration in an artery, in a vein, in the heart, and at sites which involve absorption, for example, administration in the skin, under the skin, in a muscle or in the abdomen.
[0170] Administration of pharmaceutical compositions according to the invention may be through several routes of administration, for example, subcutaneous, intramuscular, intraperitoneal, intravenous, lingual, sublingual, buccal, in the mouth, oral, in the stomach and intestine, nasal, pulmonary, for example, through the bronchioles and alveoli or a combination thereof, epidermal, dermal, transdermal, vaginal, rectal, ocular, for examples through the conjunctiva, uretal, and parenteral to patients in need of such a treatment.
[0171] Compositions of the current invention may be administered in several dosage forms, for example, as solutions, suspensions, emulsions, microemulsions, multiple emulsion, foams, salves, pastes, plasters, ointments, tablets, coated tablets, rinses, capsules, for example, hard gelatine capsules and soft gelatine capsules, suppositories, rectal capsules, drops, gels, sprays, powder, aerosols, inhalants, eye drops, ophthalmic ointments, ophthalmic rinses, vaginal pessaries, vaginal rings, vaginal ointments, injection solution, in situ transforming solutions, for example in situ gelling, in situ setting, in situ precipitating, in situ crystallization, infusion solution, and implants.
[0172] Compositions of the invention may further be compounded in, or attached to, for example through covalent, hydrophobic and electrostatic interactions, a drug carrier, drug delivery system and advanced drug delivery system in order to further enhance stability of the antibody, increase bioavailability, increase solubility, decrease adverse effects, achieve chronotherapy well known to those skilled in the art, and increase patient compliance or any combination thereof. Examples of carriers, drug delivery systems and advanced drug delivery systems include, but are not limited to, polymers, for example cellulose and derivatives, polysaccharides, for example dextran and derivatives, starch and derivatives, poly(vinyl alcohol), acrylate and methacrylate polymers, polylactic and polyglycolic acid and block co-polymers thereof, polyethylene glycols, carrier proteins, for example albumin, gels, for example, thermogelling systems, for example block co-polymeric systems well known to those skilled in the art, micelles, liposomes, microspheres, nanoparticulates, liquid crystals and dispersions thereof, L2 phase and dispersions there of, well known to those skilled in the art of phase behaviour in lipid-water systems, polymeric micelles, multiple emulsions, self-emulsifying, self-microemulsifying, cyclodextrins and derivatives thereof, and dendrimers.
[0173] Compositions of the current invention are useful in the formulation of solids, semisolids, powder and solutions for pulmonary administration of an antibody, using, for example a metered dose inhaler, dry powder inhaler and a nebulizer, all being devices well known to those skilled in the art.
[0174] Compositions of the current invention are also useful in the formulation of controlled, sustained, protracting, retarded, and slow release drug delivery systems. More specifically, but not limited to, compositions are useful in formulation of parenteral controlled release and sustained release systems (both systems leading to a many-fold reduction in number of administrations), well known to those skilled in the art. Even more preferably, are controlled release and sustained release systems administered subcutaneous. Without limiting the scope of the invention, examples of useful controlled release system and compositions are hydrogels, oleaginous gels, liquid crystals, polymeric micelles, microspheres, nanoparticles,
[0175] Methods to produce controlled release systems useful for compositions of the current invention include, but are not limited to, crystallization, condensation, co-crystallization, precipitation, co-precipitation, emulsification, dispersion, high pressure homogenisation, encapsulation, spray drying, microencapsulating, coacervation, phase separation, solvent evaporation to produce microspheres, extrusion and supercritical fluid processes. General reference is made to Handbook of Pharmaceutical Controlled Release (Wise, D. L., ed. Marcel Dekker, New York, 2000) and Drug and the Pharmaceutical Sciences vol. 99: Protein Formulation and Delivery (MacNally, E. J., ed. Marcel Dekker, New York, 2000).
[0176] Parenteral administration may be performed by subcutaneous, intramuscular, intraperitoneal or intravenous injection by means of a syringe, optionally a pen-like syringe. Alternatively, parenteral administration can be performed by means of an infusion pump. A further option is a composition which may be a solution or suspension for the administration of the antibody compound in the form of a nasal or pulmonal spray. As a still further option, the pharmaceutical compositions containing an antibody of the invention can also be adapted to transdermal administration, e.g. by needle-free injection or from a patch, optionally an iontophoretic patch, or transmucosal, e.g. buccal, administration.
[0177] The antibody can be administered via the pulmonary route in a vehicle, as a solution, suspension or dry powder using any of known types of devices suitable for pulmonary drug delivery. Examples of these comprise of, but are not limited to, the three general types of aerosol-generating for pulmonary drug delivery, and may include jet or ultrasonic nebulizers, metered-dose inhalers, or dry powder inhalers (Cf. Yu J, Chien Y W. Pulmonary drug delivery: Physiologic and mechanistic aspects. Crit Rev Ther Drug Carr Sys 14(4) (1997) 395-453).
[0178] Based on standardized testing methodology, the aerodynamic diameter (da) of a particle is defined as the geometric equivalent diameter of a reference standard spherical particle of unit density (1 g/cm.sup.3). In the simplest case, for spherical particles, da is related to a reference diameter (d) as a function of the square root of the density ratio as described by:
d a = .rho. .rho. a d ##EQU00001##
[0179] Modifications to this relationship occur for non-spherical particles (cf. Edwards D A, Ben-Jebria A, Langer R. Recent advances in pulmonary drug delivery using large, porous inhaled particles. J Appl Physiol 84(2) (1998) 379-385). The terms "MMAD" and "MMEAD" are well-described and known to the art (cf. Edwards D A, Ben-Jebria A, Langer R and represent a measure of the median value of an aerodynamic particle size distribution. Recent advances in pulmonary drug delivery using large, porous inhaled particles. J Appl Physiol 84(2) (1998) 379-385). Mass median aerodynamic diameter (MMAD) and mass median effective aerodynamic diameter (MMEAD) are used inter-changeably, are statistical parameters, and empirically describe the size of aerosol particles in relation to their potential to deposit in the lungs, independent of actual shape, size, or density (cf. Edwards D A, Ben-Jebria A, Langer R. Recent advances in pulmonary drug delivery using large, porous inhaled particles. J Appl Physiol 84(2) (1998) 379-385). MMAD is normally calculated from the measurement made with impactors, an instrument that measures the particle inertial behaviour in air.
[0180] In a further embodiment, the formulation could be aerosolized by any known aerosolisation technology, such as nebulisation, to achieve a MMAD of aerosol particles less than 10 .mu.m, more preferably between 1-5 .mu.m, and most preferably between 1-3 .mu.m. The preferred particle size is based on the most effective size for delivery of drug to the deep lung, where protein is optimally absorbed (cf. Edwards D A, Ben-Jebria A, Langer A, Recent advances in pulmonary drug delivery using large, porous inhaled particles. J Appl Physiol 84(2) (1998) 379-385).
[0181] Deep lung deposition of the pulmonal formulations comprising the antibody may optional be further optimized by using modifications of the inhalation techniques, for example, but not limited to: slow inhalation flow (eg. 30 L/min), breath holding and timing of actuation.
[0182] The term "stabilized formulation" refers to a formulation with increased physical stability, increased chemical stability or increased physical and chemical stability.
[0183] The term "physical stability" of the protein formulation as used herein refers to the tendency of the protein to form biologically inactive and/or insoluble aggregates of the protein as a result of exposure of the protein to thermo-mechanical stresses and/or interaction with interfaces and surfaces that are destabilizing, such as hydrophobic surfaces and interfaces. Physical stability of the aqueous protein formulations is evaluated by means of visual inspection and/or turbidity measurements after exposing the formulation filled in suitable containers (e.g. cartridges or vials) to mechanical/physical stress (e.g. agitation) at different temperatures for various time periods. Visual inspection of the formulations is performed in a sharp focused light with a dark background. The turbidity of the formulation is characterized by a visual score ranking the degree of turbidity for instance on a scale from 0 to 3 (a formulation showing no turbidity corresponds to a visual score 0, and a formulation showing visual turbidity in daylight corresponds to visual score 3). A formulation is classified physically unstable with respect to protein aggregation, when it shows visual turbidity in daylight. Alternatively, the turbidity of the formulation can be evaluated by simple turbidity measurements well-known to the skilled person. Physical stability of the aqueous protein formulations can also be evaluated by using a spectroscopic agent or probe of the conformational status of the protein. The probe is preferably a small molecule that preferentially binds to a non-native conformer of the protein. One example of a small molecular spectroscopic probe of protein structure is Thioflavin T. Thioflavin T is a fluorescent dye that has been widely used for the detection of amyloid fibrils. In the presence of fibrils, and perhaps other protein configurations as well, Thioflavin T gives rise to a new excitation maximum at about 450 nm and enhanced emission at about 482 nm when bound to a fibril protein form. Unbound Thioflavin T is essentially non-fluorescent at the wavelengths.
[0184] Other small molecules can be used as probes of the changes in protein structure from native to non-native states. For instance the "hydrophobic patch" probes that bind preferentially to exposed hydrophobic patches of a protein. The hydrophobic patches are generally buried within the tertiary structure of a protein in its native state, but become exposed as a protein begins to unfold or denature. Examples of these small molecular, spectroscopic probes are aromatic, hydrophobic dyes, such as antrhacene, acridine, phenanthroline or the like. Other spectroscopic probes are metal-amino acid complexes, such as cobalt metal complexes of hydrophobic amino acids, such as phenylalanine, leucine, isoleucine, methionine, and valine, or the like.
[0185] The term "chemical stability" of the protein formulation as used herein refers to chemical covalent changes in the protein structure leading to formation of chemical degradation products with potential less biological potency and/or potential increased immunogenic properties compared to the native protein structure. Various chemical degradation products can be formed depending on the type and nature of the native protein and the environment to which the protein is exposed. Elimination of chemical degradation can most probably not be completely avoided and increasing amounts of chemical degradation products is often seen during storage and use of the protein formulation as well-known by the person skilled in the art. Most proteins are prone to deamidation, a process in which the side chain amide group in glutaminyl or asparaginyl residues is hydrolysed to form a free carboxylic acid. Other degradation pathways involve formation of high molecular weight transformation products where two or more protein molecules are covalently bound to each other through transamidation and/or disulfide interactions leading to formation of covalently bound dimer, oligomer and polymer degradation products (Stability of Protein Pharmaceuticals, Ahem. T. J. & Manning M. C., Plenum Press, New York 1992). Oxidation (of for instance methionine residues) can be mentioned as another variant of chemical degradation. The chemical stability of the protein formulation can be evaluated by measuring the amount of the chemical degradation products at various time-points after exposure to different environmental conditions (the formation of degradation products can often be accelerated by for instance increasing temperature). The amount of each individual degradation product is often determined by separation of the degradation products depending on molecule size and/or charge using various chromatography techniques (e.g. SEC-HPLC and/or RP-HPLC).
[0186] Hence, as outlined above, a "stabilized formulation" refers to a formulation with increased physical stability, increased chemical stability or increased physical and chemical stability. In general, a formulation must be stable during use and storage (in compliance with recommended use and storage conditions) until the expiration date is reached.
[0187] In one embodiment of the invention the pharmaceutical formulation comprising the antibody is stable for more than 6 weeks of usage and for more than 3 years of storage.
[0188] In another embodiment of the invention the pharmaceutical formulation comprising the antibody is stable for more than 4 weeks of usage and for more than 3 years of storage.
[0189] In a further embodiment of the invention the pharmaceutical formulation comprising the antibody is stable for more than 4 weeks of usage and for more than two years of storage.
[0190] In an even further embodiment of the invention the pharmaceutical formulation comprising the antibody is stable for more than 2 weeks of usage and for more than two years of storage.
[0191] Suitable antibody formulations can also be determined by examining experiences with other already developed therapeutic monoclonal antibodies. Several monoclonal antibodies have been shown to be efficient in clinical situations, such as Rituxan (Rituximab), Herceptin (Trastuzumab) Xolair (Omalizumab), Bexxar (Tositumomab), Campath (Alemtuzumab), Zevalin, Oncolym and similar formulations may be used with the antibodies of this invention. For example, a monoclonal antibody can be supplied at a concentration of 10 mg/mL in either 100 mg (10 mL) or 500 mg (50 mL) single-use vials, formulated for IV administration in 9.0 mg/mL sodium chloride, 7.35 mg/mL sodium citrate dihydrate, 0.7 mg/mL polysorbate 80, and Sterile Water for Injection. The pH is adjusted to 6.5. In another embodiment, the antibody is supplied in a formulation comprising about 20 mM Na-Citrate, about 150 mM NaCl, at pH of about 6.0.
Therapeutic Applications
[0192] Methods of treating a patient using an anti-NKG2A antibody as described herein are also provided for. In one embodiment, the invention provides for the use of an antibody as described herein in the preparation of a pharmaceutical composition for administration to a human patient. Typically, the patient suffers from, or is at risk for, cancer, a viral disease, an inflammatory disorder, or an autoimmune disorder. Alternatively, the antibody of the invention is used to improve bone marrow transplantation in a patient.
[0193] For example, in one aspect, the invention provides a method of potentiating the activity of CD94/NKG2A-restricted lymphocytes in a patient in need thereof, comprising the step of administering a human or humanized anti-NKG2A antibody to said patient, which antibody reduces or prevents HLA-E-mediated activation of the CD94/NKG2A receptor. In one embodiment, the method directed at increasing the activity of such lymphocytes in patients having a disease in which increased NK, T, and/or NKT cell activity is beneficial, which involves, affects or is caused by cells susceptible to lysis by NK, T, or NKT cells, or which is caused or characterized by insufficient NK, T, or NKT cell activity, such as a cancer, an infectious disease or an immune disorder.
[0194] More specifically, the methods and compositions of the present invention are utilized for the treatment of a variety of cancers and other proliferative diseases including, but not limited to: carcinoma, including that of the bladder, breast, colon, kidney, liver, lung, ovary, prostate, pancreas, stomach, cervix, thyroid and skin, including squamous cell carcinoma; hematopoietic tumors of lymphoid lineage, including leukemia, acute lymphocytic leukemia, chronic lymphocytic leukemia, acute lymphoblastic leukemia, B-cell lymphoma, T-cell lymphoma, Hodgkin's lymphoma, non-Hodgkin's lymphoma, hairy cell lymphoma and Burkitt's lymphoma, and multiple myeloma; hematopoietic tumors of myeloid lineage, including acute and chronic myelogenous leukemias, promyelocytic leukemia, and myelodysplastic syndrome; tumors of mesenchymal origin, including fibrosarcoma and rhabdomyosarcoma; other tumors, including melanoma, seminoma, terato-carcinoma, neuroblastoma and glioma; tumors of the central and peripheral nervous system, including astrocytoma, neuroblastoma, glioma, and schwannomas; tumors of mesenchymal origin, including fibrosarcoma, rhabdomyosarcoma, and osteosarcoma; and other tumors, including melanoma, xeroderma pigmentosum, keratoacanthoma, seminoma, thyroid follicular cancer and teratocarcinoma.
[0195] Particular disorders that can be treated according to the invention include hematopoietic tumors of lymphoid lineage, for example T-cell and B-cell tumors, including but not limited to T-cell disorders such as T-prolymphocytic leukemia (T-PLL), including of the small cell and cerebriform cell type; large granular lymphocyte leukemia (LGL) preferably of the T-cell type; Sezary syndrome (SS); adult T-cell leukemia lymphoma (ATLL); T-NHL hepatosplenic lymphoma; peripheral/post-thymic T cell lymphoma (pleomorphic and immunoblastic subtypes); angio immunoblastic T-cell lymphoma; angiocentric (nasal) T-cell lymphoma; anaplastic (Ki 1+) large cell lymphoma; intestinal T-cell lymphoma; T-lymphoblastic; lym-phoma/leukaemia (T-Lbly/T-ALL), multiple myeloma.
[0196] Other proliferative disorders can also be treated according to the invention, including for example hyperplasias, fibrosis (especially pulmonary, but also other types of fibrosis, such as renal fibrosis), angiogenesis, psoriasis, atherosclerosis and smooth muscle proliferation in the blood vessels, such as stenosis or restenosis following angioplasty.
[0197] In a particular aspect, antibodies of the invention are used to treat NK-type lymphoproliferative disease of granular lymphocytes; alternatively called NK-LGL), referring to a class of proliferative disorders that is caused by the clonal expansion of NK cells or NK-like cells, i.e., large granular lymphocytes showing a characteristic combination of surface antigen expression (e.g., CD3-, CD56+, CD16+, etc.; see, e.g., Loughran (1993) Blood 82:1). The cell proliferation underlying these disorders can have variable effects, ranging from the mild symptoms seen in some patients to the aggressive, often-fatal form of the disease called NK-LDGL leukemia. Symptoms of this class of disorders can include fever, mild neutropenia, thrombocytopenia, anemia, lymphocytosis, splenomegaly, hepatomegaly, lymphadenopathy, marrow infiltration, and others (see, e.g., Zambello et al. (2003) Blood 102:1797; Loughran (1993) Blood 82:1; Epling-Bumette et al. (2004) Blood-2003-02-400).
[0198] The CD94/NKG2A antibody based treatment can also be used to treat or prevent infectious diseases, including preferably any infections caused by infection by viruses, bacteria, protozoa, molds or fungi. Such viral infectious organisms include, but are not limited to, hepatitis type A, hepatitis type B, hepatitis type C, influenza, varicella, adenovirus, herpes simplex type I (HSV-1), herpes simplex type 2 (HSV-2), rinderpest, rhinovirus, echovirus, rotavirus, respiratory syncytial virus, papilloma virus, cytomegalovirus, echinovirus, arbovirus, huntavirus, coxsackie virus, mumps virus, measles virus, rubella virus, polio virus and human immunodeficiency virus type I or type 2 (HIV-1, HIV-2). Bacteria constitute another preferred class of infectious organisms including but are not limited to the following: Staphylococcus; Streptococcus, including S. pyogenes; Enterococcl; Bacillus, including Bacillus anthracis, and Lactobacillus; Listeria; Corynebacterium diphtheriae; Gardnerella including G. vaginalis; Nocardia; Streptomyces; Thermoactinomyces vulgaris; Treponema; Camplyobacter, Pseudomonas including P. aeruginosa; Legionella; Neisseria including N. gonorrhoeae and N. meningitides; Flavobacterium including F. meningosepticum and F. odoratum; Brucella; Bordetella including B. pertussis and B. bronchiseptica; Escherichia including E. coli, Klebsiella; Enterobacter, Serratia including S. marcescens and S. liquefaciens; Edwardsiella; Proteus including P. mirabilis and P. vulgaris; Streptobacillus; Rickettsiaceae including R. fickettsfi, Chlamydia including C. psittaci and C. trachomatis; Mycobacterium including M. tuberculosis, M. intracellulare, M. folluitum, M. laprae, M. avium, M. bovis, M. africanum, M. kansasii, M. intracellulare, and M. lepraemurium; and Nocardia. Protozoa may include but are not limited to, leishmania, kokzidioa, and trypanosoma. Parasites include but are not limited to, chlamydia and rickettsia. A complete list of infectious diseases can be found on the website of the National Center for Infectious Disease (NCID) at the Center for Disease Control (CDC) (World-Wide Web (www) address cdc.gov/ncidod/diseases/), which list is incorporated herein by reference. All of these diseases are candidates for treatment using the inhibitory anti-CD94/NKG2A antibodies of the invention.
[0199] In an alternative aspect, the anti-NKG2A antibodies are used to target and kill NKG2A-expressing cells in, e.g., a patient suffering from a cancer characterized by CD94/NKG2A expression on cancerous cells, for example an NK-lymphoma. In one embodiment, the humanized antibody is administered in the form of an immunoconjugate comprising the humanized antibody and a cytotoxic agent.
[0200] In alternative aspect, the anti-NKG2A antibodies are used to treat or prevent an autoimmune or inflammatory disorder. Exemplary autoimmune disorders treatable using the present methods include, inter alia, hemolytic anemia, pemicious anemia, polyarteritis nodosa, systemic lupus erythematosus, Wegener's granulomatosis, autoimmune hepatitis, Behcet's disease, Crohn's disease, primary bilary cirrhosis, scleroderma, ulcerative colitis, Sjogren's syndrome, Type 1 diabetes mellitus, uveitis, Graves' disease, Alzheimer's disease, thyroiditis, myocarditis, rheumatic fever, scleroderma, ankylosing spondylitis, rheumatoid arthritis, glomerulonephritis, sarcoidosis, dermatomyositis, myasthenia gravis, polymyositis, Guillain-Barre syndrome, multiple sclerosis, alopecia areata, pemphigus/pemphigoid, Bullous pemphigoid, Hashimoto's thyroiditis, psoriasis, and vitiligo.
[0201] Examples of inflammatory disorders that can be treated by these methods include, but not limited to, adrenalitis, alveolitis, angiocholecystitis, appendicitis, balanitis, blepharitis, bronchitis, bursitis, carditis, cellulitis, cervicitis, cholecystitis, chorditis, cochlitis, colitis, conjunctivitis, cystitis, dermatitis, diverticulitis, encephalitis, endocarditis, esophagitis, eustachitis, fibrositis, folliculitis, gastritis, gastroenteritis, gingivitis, glossitis, hepatosplenitis, keratitis, labyrinthitis, laryngitis, lymphangitis, mastitis, media otitis, meningitis, metritis, mucitis, myocarditis, myosititis, myringitis, nephritis, neuritis, orchitis, osteochondritis, otitis, pericarditis, peritendonitis, peritonitis, pharyngitis, phlebitis, poliomyelitis, prostatitis, pulpitis, retinitis, rhinitis, salpingitis, scleritis, selerochoroiditis, scrotitis, sinusitis, spondylitis, steatitis, stornatitis, synovitis, syringitis, tendonitis, tonsillitis, urethritis, and vaginitis.
[0202] It has also been shown that alloreactive NK cell killing of dendritic cells improved engraftment of hematopoietic cells in a bone marrow transplant (L. Rugged et al., Science, 2002, 295:2097-2100). Thus, in another embodiment, the invention provides a method of improving the engraftment of hematopoietic cells in a patient comprising the step administering to said patient a composition of this invention comprising an activating antibody. Improvement in grafting is manifest by any one of reduced incidience or severity of graft versus host disease, prolonged survival of the graft, or a reduction in or elimination of the symptoms of the disease being treated by the graft (e.g., a hematopoietic cancer). This method is preferably used in the treatment of leukemia.
Combinations
[0203] A number of therapeutic agents are available for the treatment of cancers. The antibody compositions and methods of the present invention may thus also be combined with any other methods generally employed in the treatment of the particular disease, particularly a tumor, cancer disease, or other disease or disorder that the patient exhibits. So long as a particular therapeutic approach is not known to be detrimental to the patient's condition in itself, and does not significantly counteract the anti-CD94/NKG2A antibody-based treatment, its combination with the present invention is contemplated.
[0204] In connection with solid tumor treatment, the present invention may be used in combination with classical approaches, such as surgery, radiotherapy, chemotherapy, and the like. The invention therefore provides combined therapies in which anti-CD94/NKG2A antibodies according to the invention are used simultaneously with, before, or after surgery or radiation treatment; or are administered to patients with, before, or after administration of another anti-cancer agent. One would ensure that the surgery, radiotherapy, or anti-cancer agent in combination with the active agent in the composition of this invention exert an advantageously combined effect on the cancer.
[0205] Exemplary anti-cancer agents include chemotherapeutic agents, hormonal agents, anti-angiogenic agents, anti-metastatic agents, anti-cancer antibodies (e.g., Rituximab), antibodies against inhibitory KIR-molecules, growth-factor inhibitors, apoptosis-promoting compounds, cytokines and other immunomodulatory agents, tumor-targeting agents conjugated to toxins or radionuclides, compounds that interfere with DNA replication, mitosis and chromosomal segregation, and agents that disrupt the synthesis and fidelity of polynucleotide precursors.
[0206] For autoimmune or inflammatory disorders, any other compound known to be effective for one or more types of autoimmune or inflammatory disorders, or any symptom or feature of autoimmune or inflammatory disorders, including inter alia, immunosuppressants, e.g., azathioprine (e.g., Imuran), chiorambucil (e.g., Leukeran), cyclophosphamide (e.g., Cytoxan), cyclosporine (e.g., Sandimmune, Neoral), methotrexate (e.g., Rheumatrex), corticosteroids, prednisone (e.g., Deltasone, Meticorten), Etanercept (e.g., Enbrel), infliximab (e.g., Remicade), inhibitors of TNF, FK-506, rapamycin, mycophenolate mofetil, leflunomide, anti-lymphocyte globulin, deoxyspergualin or OKT.
[0207] Preferred examples of immunomodulatory compounds include cytokines. Other examples include compounds that have an effect, preferably an effect of activation or potentiation NK cell activity, or of inducing or supporting the proliferation of NK cells. Other compounds for administration before, simultaneously with, or after compositions comprising the agents of the invention are adjunct compounds (e.g., anti-emetics and analgesic agents) and anti-viral agents.
[0208] As will be understood by those of ordinary skill in the art, the appropriate doses of anti-cancer agents will approximate those already employed in clinical therapies wherein the anti-cancer agents are administered alone or in combination with other agents. Variation in dosage will likely occur depending on the condition being treated. The physician administering treatment will be able to determine the appropriate dose for the individual subject.
Articles of Manufacture
[0209] In another embodiment of the invention, an article of manufacture containing materials useful for the treatment of the disorders described above is provided. For example, the article of manufacture can comprise a container containing an antibody as described herein together with instructions directing a user to treat a disorder such as a cancer or a viral disease in a mammal with the antibody in an effective amount. In a preferred embodiment, the mammal is a human. The article of manufacture typically comprises a container and a label or package insert on or associated with the container. Suitable containers include, for example, bottles, vials, syringes, etc. The containers may be formed from a variety of materials such as glass or plastic. The container holds a composition that is effective for treating the condition and may have a sterile access port (for example, the container may be an intravenous solution bag or a vial having a stopper pierceable by a hypodermic injection needle). At least one active agent in the composition is the humanized anti-NKG2A antibody herein, or an antibody derivative (e.g., an immunoconjugate) comprising such a humanized antibody. The label or package insert indicates that the composition is used for treating the condition of choice, such as cancer or a viral disease.
[0210] Moreover, the article of manufacture may comprise (a) a first container with a composition contained therein, wherein the composition comprises the antibody described herein, and (b) a second container with a composition contained therein, wherein the composition comprises a therapeutic agent other than the first antibody. The article of manufacture in this embodiment of the invention may further comprise a package insert indicating that the first and second compositions can be used in combination to treat a cancer or viral disease. Such therapeutic agent may be any of the adjunct therapies described in the preceding section (e.g., a chemotherapeutic agent, an anti-angiogenic agent, an anti-hormonal compound, a cardioprotectant, and/or a regulator of immune function in a mammal, including a cytokine). Alternatively, or additionally, the article of manufacture may further comprise a second (or third) container comprising a pharmaceutically acceptable buffer, such as bacteriostatic water for injection (BWFI), phosphate-buffered saline, Ringer's solution and dextrose solution. It may further include other materials desirable from a commercial and user standpoint, including other buffers, diluents, filters, needles, and syringes.
Administration
[0211] As described above, several monoclonal antibodies have been shown to be efficient in clinical situations (such as, e.g., Rituxan (Rituximab) and others), and similar administration regimens (i.e., doses and/or administration protocols) may be used with the antibodies of this invention. Schedules and dosages for administration can be determined in accordance with known methods for these products, for example using the manufacturers' instructions. For example, an antibody preparation can be supplied at a concentration of 10 mg/mL in either 100 mg (10 mL) or 500 mg (50 mL) single-use vials. An exemplary suitable dosage range for an antibody of the invention may between about 10 mg/m.sup.2 and 500 mg/m.sup.2. Quantities and schedule of injection of anti-NKG2A antibodies that, e.g., saturate cells for 24 hours, 48 hours, 72 hours or a week or a month can be determined considering the affinity of the antibody and its pharmacokinetic parameters. However, it will be appreciated that these schedules are exemplary and that optimal schedule and regimen and the tolerability of the antibodies must be determined in clinical trials.
Non-Therapeutic Applications
[0212] The antibodies (e.g. the humanized anti-NKG2A antibodies) of the invention also have non-therapeutic applications.
[0213] For example, the antibodies may be used as affinity-purification agents. In this process, the antibodies are immobilized on a solid phase such as a SEPHADEX.TM. resin or filter paper, using methods well known in the art. The immobilized antibody is contacted with a sample containing the NKG2A protein (or fragment thereof) to be purified, and thereafter the support is washed with a suitable solvent that will remove substantially all the material in the sample except the NKG2A protein, which is bound to the immobilized antibody. Finally, the support is washed with another suitable solvent, such as glycine buffer, pH 5.0, that will release the NKG2A protein from the antibody.
[0214] Anti-NKG2A antibodies may also be useful in diagnostic assays for NKG2A protein, e.g. detecting its expression in specific cells, tissues, or serum.
[0215] For diagnostic applications, the antibody typically will be labeled with a detectable moiety. Numerous labels are available that can be generally grouped into the following categories:
[0216] (a) Radioisotopes, such as .sup.35S, .sup.14C, .sup.125I, .sup.3H, and .sup.131I. The antibody can be labeled with the radioisotope using the techniques described in Current Protocols in Immunology, Volumes 1 and 2, Coligen et al., Ed. Wiley-Interscience, New York, N.Y., Pubs. (1991), for example, and radioactivity can be measured using scintillation counting.
[0217] (b) Fluorescent labels such as rare-earth chelates (europium chelates) or fluorescein and its derivatives, rhodamine and its derivatives, dansyl, Lissamine, phycoerythrin and Texas Red are available. The fluorescent labels can be conjugated to the antibody using the techniques disclosed in Current Protocols in Immunology, supra, for example. Fluorescence can be quantified using a fluorimeter.
[0218] (c) Various enzyme-substrate labels are available and U.S. Pat. No. 4,275,149 provides a review of some of these. The enzyme generally catalyzes a chemical alteration of the chromogenic substrate that can be measured using various techniques. For example, the enzyme may catalyze a color change in a substrate, which can be measured spectrophotometrically. Alternatively, the enzyme may alter the fluorescence or chemiluminescence of the substrate. Techniques for quantifying a change in fluorescence are described above. The chemiluminescent substrate becomes electronically excited by a chemical reaction and may then emit light that can be measured (using a chemiluminometer, for example) or donates energy to a fluorescent acceptor. Examples of enzymatic labels include luciferases (e.g., firefly luciferase and bacterial luciferase; U.S. Pat. No. 4,737,456), luciferin, 2,3-dihydrophthalazinediones, malate dehydrogenase, urease, peroxidase such as horseradish peroxidase (HRPO), alkaline phosphatase, .beta.-galactosidase, glucoamylase, lysozyme, saccharide oxidases (e.g., glucose oxidase, galactose oxidase, and glucose-6-phosphate dehydrogenase), heterocyclic oxidases (such as uricase and xanthine oxidase), lactoperoxidase, microperoxidase, and the like. Techniques for conjugating enzymes to antibodies are described in O'Sullivan et al, "Methods for the Preparation of Enzyme-Antibody Conjugates for use in Enzyme Immunoassay," in Methods in Enzym. (Ed., J. Langone & H. Van Vunakis), Academic Press, New York, 73:147-166 (1981).
[0219] Examples of enzyme-substrate combinations include, for example:
[0220] (i) Horseradish peroxidase (HRPO) with hydrogen peroxidase as a substrate, wherein the hydrogen peroxidase oxidizes a dye precursor (e.g., orthophenylene diamine (OPD) or 3,3',5,5'-tetramethyl benzidine hydrochloride (TMB));
[0221] (ii) alkaline phosphatase (AP) with para-nitrophenyl phosphate as chromogenic substrate; and
[0222] (iii) beta-D-galactosidase (beta-D-Gal) with a chromogenic substrate (e.g., p-nitrophenyl-beta-D-galactosidase) or fluorogenic substrate 4-methylumbelliferyl-p-beta-galactosidase.
[0223] Numerous other enzyme-substrate combinations are available to those skilled in the art. For a general review of these, see U.S. Pat. Nos. 4,275,149 and 4,318,980.
[0224] Sometimes, the label is indirectly conjugated with the antibody. The skilled artisan will be aware of various techniques for achieving this. For example, the antibody can be conjugated with biotin, and any of the three broad categories of labels mentioned above can be conjugated with avidin, or vice versa. Biotin binds selectively to avidin, and thus, the label can be conjugated with the antibody in this indirect manner. Alternatively, to achieve indirect conjugation of the label with the antibody, the antibody is conjugated with a small hapten (e.g., digoxin) and one of the different types of labels mentioned above is conjugated with an anti-hapten antibody (e.g., anti-digoxin antibody). Thus, indirect conjugation of the label with the antibody can be achieved.
[0225] In another embodiment of the invention, the anti-NKG2A antibody need not be labeled, and the presence thereof can be detected using a labeled secondary antibody that binds to the NKG2A antibody.
[0226] The antibodies of the present invention may be employed in any known assay method, such as competitive-binding assays, direct and indirect sandwich assays, and immunoprecipitation assays. Zola, Monoclonal Antibodies: A Manual of Techniques, pp. 147-158 (CRC Press, Inc. 1987).
[0227] For immunohistochemistry, the tumor sample may be fresh or frozen or may be embedded in paraffin and fixed with a preservative such as formalin, for example.
[0228] The antibodies may also be used for in vivo diagnostic assays. Generally, the antibody is labeled with a radionuclide or a non-radioactive indicator detectable by, e.g., nuclear magnetic resonance, or other means known in the art. Preferably, the label is a radiolabel, such as, e.g., .sup.125I, .sup.131I, .sup.67Cu, .sup.99mTc, or .sup.111In. The labeled antibody is administered to a host, preferably via the bloodstream, and the presence and location of the labeled antibody in the host is assayed. This imaging technique is suitably used in the detection, staging and treatment of neoplasms. The radioisotope is conjugated to the protein by any means, including metal-chelating compounds or lactoperoxidase, or iodogen techniques for iodination.
[0229] As a matter of convenience, the antibodies of the present invention can be provided in a kit, i.e., a packaged combination of reagents in predetermined amounts with instructions for performing the diagnostic assay. Where the antibody is labeled with an enzyme, the kit will include substrates and cofactors required by the enzyme (e.g., a substrate precursor that provides the detectable chromophore or fluorophore). In addition, other additives may be included such as stabilizers, buffers (e.g., a block buffer or lysis buffer) and the like. The relative amounts of the various reagents may be varied widely to provide for concentrations in solution of the reagents that substantially optimize the sensitivity of the assay. Particularly, the reagents may be provided as dry powders, usually lyophilized, including excipients that on dissolution will provide a reagent solution having the appropriate concentration.
Deposits
[0230] The Z270 hybridoma was deposited on Dec. 22, 2005 at the Collection Nationale de Culture de Microorganismes, Institute Pasteur, 25, Rue du Docteur Roux, F-75725 Paris, France, under accession number 1-3549.
EXAMPLES
[0231] Further details of the invention are illustrated by the following non-limiting Examples.
Example I--Selection of Parent humZ270VL and humZ270VH Sequences
[0232] This Example describes the selection of parent humZ270VL and humZ270VH sequences as well as optional back mutations for variant h270VL and humZ270VH sequences.
[0233] As described in Example 2, the Z270 hybridoma was cloned, and the Z270 VH and VL chain sequences of the corresponding antibody from were determined to be:
[0234] Z270VL:
DIQMTQSPASLSASVGETVTITCRASENIYSYLAWYQQKQGKSPQFLVYNAKTLAEGVPSRF SGSGSGTQFSLKINSLQPEDFGSYYCQHHYGTPRTFGGGTKLEIK (SEQ ID NO:1), with an optional arginine (R) residue at Kabat position 108.
[0235] Z270VH:
QVQLQQPGAELVRPGASVKLSCKASGYTFTSYWMNWVKQRPEQGLQWIGRIDPYDSETH YSQKFKDKAILTVDKSSSTAYMRLSSLTSEDSAVYYCARGGYDFDVGTLYWFFDVWGAGTT VTVS (SEQ ID NO:2), with an optional C-terminal serine (S) residue.
[0236] A second light chain, Z270VL-NB, was also identified. However, this was a common myeloma light chain.
[0237] Z270VL-NB:
NIVMTQSPKSMSMSVGERVTLTCKASENVVTYVSWYQQKPEQSPKLLIYGASNRY TGVPDRFTGSGSATDFTLTISSVQAEDLADYHCGQGYSYPYTFGGGTKLEIKRA (SEQ ID NO:3), with an optional arginine (R) residue at Kabat position 108, and an optional alanine (A) residue at Kabat position 109.
[0238] From an analysis of the murine Z270 sequences, the CDRs according to Kabats definitions were determined as:
TABLE-US-00003 CDR-L1: RASENIYSYLA (residues 24-34 of SEQ ID NO: 1) CDR-L2: NAKTLAE (residues 50-56 of SEQ ID NO: 1) CDR-L3: QHHYGTPRT (residues 89-97 of SEQ ID NO: 1) CDR-H1: SYWMN (residues 31-35 of SEQ ID NO: 2) CDR-H2: RIDPYDSETHYSQKFKD (residues 50-66 of SEQ ID NO: 2) CDR-H3: GGYDFDVGTLYWFFDV (residues 95-102 of SEQ ID NO: 2).
[0239] A 3D protein structure model was built using MOE (molecular Operating Environment; available at www.chemcomp.com) with structural templates from the Protein Database Bank (PDB): 1OPG and 1XF4. The PDB is described in Berman et al. (Nuc Acids Res 2000; 28:235-242), and is available at www.rcsb.org/pdb. Based on a statistical analysis of 201 antibody-antigen complexes in the PDB database the most probable residues in the paratope were determined to be:
[0240] Z270VL: residues 24-34, 49-56, 89-97 of SEQ ID NO:1
[0241] Z270VH: residues 23-35, 49-58, 93-102 of SEQ ID NO:2.
[0242] Using MOE, residues interacting (hydrophobic, hydrogen binding, or charge) with the paratope were identified and the combined set of residues (paratope+interacting residues) were taken as the mask of Z270.
[0243] Searching the germline V databases (V-base; available at vbase.mrc-cpe.cam.ac.uk/) with the Z270VL and Z270VH returned the following potential framework templates (E-value given in parenthesis):
[0244] Heavy chain: VH1_46 (2e-036), VH1_f (2e-035), VH1_02 (3e-035), VH1_18 (4e-035), VH1_03 (6e-035); and Light chain: VKI_O2 (1e-039), VKI_O12 (1e-039), VKI_L12 (9e-038), VKI_L8 (9e-038), VKI_A20 (9e-038).
[0245] Searching the germline databases with the mask returned the following potential framework templates (E-value given in parenthesis):
[0246] Heavy chain: VH5_a (4e-013), VH5_51 (3e-011), VH1_f (1e-010), VH1_18 (2e-010), VH1_46 (4e-010): and
[0247] Light chain: VKI_L9 (2e-012), VKI_O2 (3e-012), VKI_O12 (3e-012), VKI_L24 (2e-011), VKI_A20 (2e-011)
[0248] After manual inspections of the alignments and the hits, VH1_18 and VKI_O2 were selected as the human scaffolds. JH6 and JK4 were chosen as germline J-segments.
[0249] Humanization was now designed with the following rules: [0250] Residues outside the mask are taken as human. [0251] Residues inside the mask and inside the Kabat CDR are taken as murine. [0252] Residues inside the mask and outside the Kabat CDR with mouse/germline consensus are taken as the consensus sequence. [0253] Residues inside the mask and outside the Kabat CDR with mouse/germline difference are subject to potential back mutations.
[0254] The analysis is illustrated in FIG. 1 for Z270VL and Z270VH, where mask residues are those shaded in the Kabat scheme; CDR residues are shown in bold in the Kabat scheme; and mouse/germline differences are shaded in the VKI_02/JK4 and VH1_18/JH6 sequences. Z270VL and humZ270VL1, and humZ270VL1 cons may optionally comprise an arginine (R) residue at Kabat position 108.
[0255] The resulting sequences, humZ270VL1 and humZ270VH1, are given with the potential back mutation residues as human.
[0256] The CDRs of a humanized Z270 antibody according to the Kabat definitions are shown in FIG. 2. Of the humZ270 CDRs, only the CDR-H2 sequence was different than that of the corresponding murine CDR, differing in 4 positions. However, in effect, this meant that Kabat CDR-H2 residues 60-65 were identical to the human acceptor sequence, providing for a more human molecule and a lesser risk for immunogenicity. In FIG. 2, the differences are indicated in bold text.
Example 2--Cloning of Z270 IgG1 VH and VL Regions
[0257] This Example describes the cloning and sequencing of murine Z270 VH and VL regions.
[0258] Z270 Hybridoma Cell Culture for Total RNA Extraction.
[0259] Z270 hybridoma was cultured in RPMI 1640 (Hyclone Cat# SH30011.04) plus 10% FCS (Biochrom Cat#S0115). 5.times.10.sup.8 to 1.times.10.sup.7 cells were harvested for total RNA extraction.
[0260] Z270 Hybridoma Cell Culture of Antibody Production.
[0261] One week batch culture strategy was adopted for the production of the Z270 mAb. The culture medium was RPMI 1640 with 10% FCS. The supernatant was harvested every 7 days. The starting cell density in the culture chamber of CL-1000 flask (INTEGRA Biosciences, Item No. 90005, Lot No. 08541150) was be 2.times.10.sup.8 cells/ml. During the culture, the cell density and viability was checked regularly. After 3 days culture in CL-1000 flask, the density was above 1.times.10.sup.7 and the viability was about 85%. In the next 4 days the cell density varied between 1.5-2.5.times.10.sup.7 cells/ml and the viability decreased gradually to 60.about.70%. At the 7.sup.th day the supernatant was collected and reducing SDS-PAGE was used to estimate the antibody concentration with a quantification control (A-TNP 20050118 2 mg/ml).
[0262] Z270 Total RNA Extraction.
[0263] This was done using TRIZOL reagent Invitrogen, Cat. No. 15596-026, according to the manufacturer's instructions.
[0264] 5'-RACE (Rapid Amplification of cDNA Ends).
[0265] A protocol was adapted from the manufacturer's instructions for use of SMART.TM. RACE cDNA Amplification Kit product by Clontech, Catalog no. 634914, with the following design of Gene-Specific Primers (GSPs):
TABLE-US-00004 GSP1 for amplification of IgG1 heavy chain variable region (SEQ ID NO: 12) RacePrimerheavy: 5'-GCCAGTGGATAGACAGATGG-3' GSP2: for amplification of IgG1 Kappa chain variable region (SEQ ID NO: 13) RacePrimerkappa: 5'-GATGGATACAGTTGGTGCAGC-3'.
[0266] After first-strand cDNA synthesis, RACE, and analysis of the resulting samples on a 1.5% agarose gel, the RACE DNA band was cut out and purified with Gel purification kit (QIAGEN QIAquick Cat#28706) then cloned into pMD-19 using DNA Ligation Kit (Cat#D6022 from TAKARA) Ver. 2.0 by TA cloning (FIG. 3A). The positive clones were sent out for DNA sequencing.
[0267] Several clones of light chain and heavy chain were sequenced with identical sequence as shown in FIG. 4A-D (with bold text indicating signal peptide sequences):
[0268] Z270 VL cDNA: (SEQ ID NO:14)
[0269] Z270 VL protein (SEQ ID NO:15)
[0270] Z270 VH cDNA (SEQ ID NO:16)
[0271] Z270 VH protein (SEQ ID NO:17).
Example 3--Cloning of Murine IgG1 Light Chain and Heavy Chain into pJSV002
[0272] In order to test antibody affinity and to use it for positive control, Z270 antibody light chain and heavy chain is inserted into pJSV002 as murine antibody IgG1. pJSV002 is a transient expression vector that can be used in combination with HEK293 6E cells for Z270 transient expression (FIG. 3B).
[0273] Z270 VL and IgG1 constant region are cloned into EcoRI and Nhe I sites of pJSV002. The resulted plasmid is as shown in FIG. 3C. The inserted sequence with EcoRI and Nhe I (uppercase letters) at both ends is shown in FIG. 4E (SEQ ID NO:18).
[0274] H1 variable region and IgG1 constant region is cloned into EcoRI and Nhe I sites of pJSV002-mlgGI-variant (FIG. 3D). The pJSV002-mlgGI-variant contains murine IgG1 heavy chain constant region (FIG. 3E). The inserted sequence is between EcoRI and Nhe I (gctagc) sites, with restriction sites and murine IgG1 constant region indicated in lowercase letters, is shown in FIG. 4F (SEQ ID NO:19).
Example 4--Plasmid Constructs for Chimeric Z270 Antibody
[0275] In order to test antibody affinity and to use it as positive control, murine Z270 antibody light chain and heavy chain are inserted into pJSV002 as human chimeric antibody IgG4 S241P. The antibody contains murine variable region and human IgG4 constant region with an S241P mutation in the heavy chain.
[0276] For expression of chimeric Z270 antibody (chimZ270), Z270VL sequence and human light chain (kappa) constant region is inserted into EcoR I and BamH I sites of pJSV002 (FIG. 3F). The sequence shown in FIG. 4G is used, where uppercase letters indicate restriction sites (SEQ ID NO:20).
[0277] Z270 VH and human heavy chain constant region is inserted into EcoR I and Nhe I sites of pJSV002-IgG4-S241P (FIGS. 3G and 3H). The sequence shown in FIG. 4H is used, with the actual inserted sequence being between Eco RI and Nhe I (gctagc) sites, where upper-case letters indicate restriction sites (SEQ ID NO:21).
Example 5--Expression of Humanized Z270
[0278] According to the Z270 humanization strategy described in Example 1, Z270 light chain CDRs are grafted into the VKI_02/JK4 template to prepare humZ270VL1. The sequence shown in FIG. 4I results from synthesizing the sequence between EcoRI and KasI sites and inserting the it into pJSV002-hKappaC (with human Kappa constant region in pJSV002), with CDR-encoding sequences in uppercase letters and restriction sites in bold text (SEQ ID NO:22).
[0279] For humanization of the heavy chain, Z270 heavy chain CDRs (optionally with optimized CDR-H2 are grafted into the VH1_18/JK6 template to prepare humZ270VH1. The sequence between EcoRI and NheI sites is synthesized and cloned into pJSV002-hlgG4 S241P (human IgG4 S241P constant region in pJSV002, FIG. 3G), with CDR-encoding sequences in uppercase letters and restriction sites in bold text (FIG. 4J, SEQ ID NO:23).
[0280] Mutated constructs are likewise cloned into pJSV002 with the corresponding IgG4 S241P constant region and human Kappa chain constant region, with the following combinations of framework back-mutations in each of humZ270VL and humZ270VH:
[0281] humZ270VL: MWt (i.e., no back-mutation), L46F, 148V, L46F_148V
[0282] humZ270VH: MWt (i.e., no back-mutation), V5Q, M69L, T71V, T73K, T75S, V5Q_M69L, V5Q_T71V, V5Q_T73K, V5Q_T75S, M69L_T71V, M69L_T73K, M69L_T75S, T71V_T73K, T71V_T75S, T73K_T75S, V5Q_M69L_T71V_T73K_T75S, M69L_T71V_T73K_T75S, V5Q_T71V_T73K_T75S, V5Q_M69L_T73K_T75S, V5Q_M69L_T71V_T75S, V5Q_M69L_T71V_T73K, T71V_T73K_T75S, M69L_T73K_T75S, M69L_T71V_T75S, M69L_T71V_T73K, V5Q_T73K_T75S, V5Q_T71V_T75S, V5Q_T71V_T73K, V5Q_M69L_T75S, V5Q_M69L_T73K, V5Q_M69L_T71V. An exemplary vector design for the heavy chain (HC) is illustrated in FIG. 5, and an exemplary vector design for the light chain (LC) is provided in FIG. 6.
[0283] The plasmids are transfected into HEK293 6E for transient expression, using 293fectin. Materials: Cells: HEK 293 6E (293-6E) cells are grown in exponential growth phase (0.8 to 1.2.times.106 cells/ml). Culture Medium: FreeStyle.TM. (Cat. No. 12338-018 from Gibco); 25 .mu.g/ml Geneticin 418 (Cat. No. 10131-019 from Gibco); 0.1% pluronic F-68 (Cat. No. 24040-032 from Gibco). Transfection Medium: Opti-MEM (Cat. No. 51985-026 from Gibco); 293fectin (Cat. No. 12347019 from Invitrogen). Plasmid DNA: Purified plasmid DNA of interest (see above).
[0284] Cell Count and Inoculation.
[0285] Two days before transfection, the necessary volume to get 7.5.times.10.sup.6 cells is transferred into a 125 ml flask, and fresh Freestyle medium is added to complete to 30 ml (final cell density should be 0.25.times.10.sup.6 cells/ml). Two days later (the day of transfection), cell density should be between 1 and 1.2.times.10.sup.6 cells/ml. Alternatively, one day before transfection, the necessary volume to get 1.5.times.10.sup.7 cells is transferred into a 125 ml flask, and fresh Freestyle medium is added to complete to 30 ml (final cell density should be of 0.50.times.10.sup.6 cells/ml). 24h later (the day of transfection), cell density should be between 0.9 and 1.2.times.10.sup.6 cells/ml.
[0286] 293Fectin-DNA Complexes Preparation.
[0287] To prepare DNA solution, 30 .mu.g DNA is diluted in a total volume of 1 ml Opti-MEM. To prepare 293fectin solution, 40 .mu.l is diluted in 960 .mu.l Opti-MEM. After 5 min. incubation at room temperature, the 293fectin solution and the DNA solution are mixed, and then incubated 25 minutes at room temperature. The cells are then transfected with 2 ml 293fectin-DNA mixture, and incubate at 37.degree. C. in a humidified incubator (orbital shaker) containing 5% CO2 for 4 to 6 days. FIG. 7 outlines a general procedure for transient expression in HEK293 cells, such as e.g., HEK693 6E cells.
Example 6--Purification of IgG1 from Z270 Hybridoma Culture
[0288] IgG1 Z270 antibody is purified from Z270 hybridoma cell culture fluid by adding the sample onto a HiTrap Protein A HP (1 ml) column equilibrated in 3M NaCl 50 mM Tris pH8.5, at a flow rate: 1.0 ml/min, and eluting antibody using 25 mM citric acid, 4.5 mM sodium citric acid pH3.0.
Example 7--Antibody Quantification and Affinity Determination
[0289] Plasmids containing light chain and equivalent heavy chain expression constructs are mixed in pairs, and the plasmids are used to transfect HEK6E cells. The culture medium supernatant is then collected.
[0290] To quantify mouse IgG1 (or IgG4) antibody quantification, an ELISA plate is coated with Goat poly anti mouse IgG1 (or IgG4) Fc specific capture antibody. Antibody expression supernatant is applied, followed by HRP-Goat poly anti mouse kappa or Fab secondary antibody. HRP substrate is applied, and conversion detected at OD450.
[0291] To analyse antigen-binding of humanized Z270 antibodies, the following Biacore assay, illustrated in FIG. 8, is used. Antigen-capture antibody is immobilized on a Biacore chip. The antigen and culture supernatant are applied. The on- and off-rates are analyzed to calculate the affinity.
Example 8--Biacore Analysis of Chimeric, Humanized, and Back-Mutation Variants of Z270
Materials and Methods
[0292] Chimeric Z270 and humZ270 in VKI_O2/JK4 light chain and VH1_18/JH6 heavy chain acceptor frameworks were produced according to the methods described in Examples 4 and 5. The antigen-binding properties of chimeric Z270, humZ270 and back-mutation variants were analyzed on a Biacore T100 (Biacore AB, Uppsala, Sweden). The antigen was in the form of a single-chain NKG2A-CD94-mFc construct was covalently immobilized on the sensor CM5 chip (Biacore AB, Uppsala, Sweden) via amine groups using 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride (EDC) and N-hydroxysuccinimide (NHS). The immobilization level was targeted at 300 RU. Z270 antibody variants were diluted to a concentration series (0.157, 0.313, 0.625, 1.25, 2.5 nM) in the running buffer HBS-EP (10 mM HEPES pH 7.4, 150 mM NaCl, 3 mM EDTA, 0.005% (v/v) Tween-20). All the samples were then injected over the immobilized antigen for 2 min at the flow rate of 40 ul/min. Subsequently, the running buffer was injected for 3 min at 40 ul/min for antibody dissociation analysis. After each run, the regeneration buffer (10 mM NaOH, 500 mM NaCl) was injected (30 seconds, 10 ul/min) to completely strip the remaining antibodies off the antigen. Data were evaluated with Biacore T100 evaluation software.
Results
[0293] The affinity of humZ270 was determined as 67 pM. This KD value was higher than that of chimeric Z270 (50 pM) (FIG. 9 and Table 2). Introduction of back mutations to humZ270 in VKI_02/JK4 light chain and VH1_18/JH6 heavy chain acceptor frameworks did not substantially improve its affinity (FIG. 10).
TABLE-US-00005 TABLE 2 Chimeric Z270 humZ270 ka (1/Ms) kd (1/s) KD (M) Chi.sup.2 (RU.sup.2) ka (1/Ms) Kd (1/s) KD (M) Chi.sup.2 (RU.sup.2) 7.970E+6 3.972E-4 4.983E-11 0.0968 7.492E+6 4.982E-4 6.650E-11 0.044
Example 9--Generation of humZ270 with Full-Length CDR-H2
[0294] In an alternative strategy, shown in FIG. 11, it was investigated whether full-length Kabat CDRs (including a full-length CDR-H2) would result in an improved affinity of humZ270 in VKI_O2/JK4 light chain and VH1_18/JH6 heavy chain acceptor frameworks. humZ270VH3 (SEQ ID NO:24) follows directly from the difference in CDR definitions, resulting basically in a Kabat CDR-grafted humanized Z270 antibody. humZ270VH4 (SEQ ID NO:25) follows from the observation that K38, Q46, W47, 148, G49, Y59, Q61, K62, K66, and A67 are in proximity to S60, F63, K64, D65 side-chains, resulting in a CDR-grafted humanized Z270 antibody with R38K, E46Q, M481, R66K, and V67A back-mutations.
Example 10--Generation of humZ270 in Other Human Acceptor Frameworks
[0295] A number of different humanized constructs with different human heavy chain acceptor framework sequences were made to explore the framework choice.
[0296] FIG. 12 shows an alignment between the different humanized Z270VH constructs prepared. humZ270VH5 (SEQ ID NO:26) is based on VH5_a, humZ270VH6 (SEQ ID NO:27) is based on VH5_51, humZ270VH7 (SEQ ID NO:28) is based on VH1_f, and humZ270VH8 (SEQ ID NO:29) is based on VH1_46, all with a JH6 J-segment. The 6 C-terminal amino acid residues of the Kabat CDR-H2 of all humanized constructs were identical to the human acceptor framework.
[0297] Using the alignment program Vector NTI, the following sequence identities between humZ270VH1 and humZ270VH5, -6, -7, and -8 were obtained: 78.2% (VH1 vs. VH5), 79.0% (VH1 vs. VH6), 88.7% (VH1 vs. VH7), and 96.0% (VH1 vs. VH8).
Example 11--Biacore Analysis of Different humZ270 Variants
[0298] The affinities of huZ270 variants, all comprising a humZ270VL1 sequence but with different strategies employed for VH sequence humanization, were analyzed.
Materials and Methods
[0299] A Biacore T100 (Biacore AB, Uppsala, Sweden) was used. CD94/NKG2A antigen was used in the form of a single-chain NKG2A-CD94-mFc construct, covalently immobilized on the sensor CM5 chip (Biacore AB, Uppsala, Sweden) via amine groups using 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride (EDC) and N-hydroxysuccinimide (NHS). The immobilization level was targeted at 300 RU. humZ270 antibody variants were diluted to a concentration series (0.157, 0.313, 0.625, 1.25, 2.5 nM) in the running buffer HBS-EP. All the samples were then injected over the immobilized antigen for 2 min at the flow rate of 40 ul/min. Subsequently, the running buffer was injected for 3 min at 40 ul/min for antibody dissociation analysis. After each run, the regeneration buffer (10 mM NaOH, 500 mM NaCl) was injected (40 seconds, 10 ul/min) to completely strip the remaining antibodies off the antigen. Data were evaluated with Biacore T100 evaluation software. The KD value of each variant was divided by that of huZ270 with a humZ270VH1 heavy chain to obtain the relative KD fold change.
Results and Conclusions
[0300] The results are shown in FIG. 13, where the KD value of each variant was normalized to that of huZ270 with a humZ270VH1 heavy chain to obtain the relative change in KD. As shown in FIG. 13, there were no significant differences between the CDR-grafted variants (VH3, VH4) and the humZ270 variant comprising fewer murine residues in the CDR-H2 segment, (humZ270VL1NH1). Nor were there substantial differences between the "humanized-CDR-H2" variants in different human acceptor frameworks. Since a more human humZ270 antibody has the benefit of a lower risk for an immunogenic response in human patients, the humanized-CDR-H2 variants such as VH1, VH5, VH6, and VH7 can be chosen for therapeutic applications without the compromise of a significantly lower affinity as compared to a standard CDR-grafted humZ270 antibody, and with a lower likelihood of a host immune response.
Example 12--Identification of Critical Residues in Z270VL and VH
[0301] In order to identify the paratope of Z270, alanine scan mutagenesis was conducted on the CDRs of the murine antibody. The following amino acids were selected for alanine mutagenesis (FIG. 14):
[0302] Z270VL: R24A, S26A, E27A, N28A, Y30A, S31A, N50A, K52A, T53A, E56A, Y92A, T94A
[0303] Z270VH: K23A, S25A, T28A, T30A, S31A, N35A, D52A, Y53A, D54A, S55A, E56A, R94A, D98A, F99A, D100A, V(100A)A, T(100C)A, L(100D)A, W(100F)A, D101A.
[0304] Materials and Methods
[0305] The antigen-binding properties of the alanine mutants were analyzed on a Biacore T100 (Biacore AB, Uppsala, Sweden). Antigen in the form of sc-NKG2A-CD94-mFc was covalently immobilized on the sensor CM5 chip (Biacore AB, Uppsala, Sweden) via amine groups using 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride (EDC) and N-hydroxysuccinimide (NHS). The immobilization level was targeted at 300 RU. Purified Z270 alanine mutants were diluted to 0.75 nM or 1.5 nM in the running buffer HBS-EP. All the samples were then injected over the immobilized antigen for 3 min at the flow rate of 10 ul/min. Subsequently, the running buffer was injected for 1 min at 10 ul/min for antibody binding stability analysis. After each run, the regeneration buffer (10 mM NaOH, 500 mM NaCl) was injected (35 seconds, 10 ul/min) to completely strip the remaining antibodies off the antigen. Data were evaluated with Biacore T100 evaluation software. The relative binding of each mutant was calculated through dividing its binding level (RU) obtained from Biacore by that of chimZ270.
Results and Conclusions
[0306] As shown in FIG. 15, Z270VH alanine mutants D52A, D54A, F99A, T(100C)A, and W(100F)A completely lost their antigen-binding properties. The heavy-chain mutant R94 remained around 20% antigen-binding ability. The relative binding levels of heavy-chain mutants N35A, Y53A, E56A, D98A, V(100A)A, and L(100D)A were between 40-70% (FIG. 1). Accordingly, the amino acids D52, D54, R94, F99, T(100C), and W(100F) in the Z270 heavy chain CDR-H2 and CDR-H3 are the critical residues to recognize the antigen. Meanwhile, the amino acids N35, Y53, E56, D98, V(100A), and L(100D) in the heavy chain moderately affect the antigen-binding. Interestingly, all Z270 light-chain alanine mutants retain comparable antigen-binding properties to that of chimeric Z270 (FIG. 16). Therefore, no amino acid in the Z270 light chain significantly contributes to antigen recognition.
Example 13--HumZ270 Specifically Binds Cells Expressing CD94/NKG2A
[0307] The strength and specificity of humZ270 binding to CD94/NKG2A was tested in flow-cytometry, by analyzing the binding of in HEK293 cells produced wild-type Z270 (recZ270), chimeric Z270 with human IgG4 (chimZ270) or humanized Z270 (humZ270VL1NH1) to Ba/F3 cells stably over-expressing either CD94/NKG2A or CD94/NKG2C. For this purpose, Ba/F3-CD94/NKG2A and -C cells were incubated with various concentrations of Z270 variants in tissue-culture medium containing 2% FCS, for at least 30 minutes on ice. Subsequently cells were washed, and the cells incubated in similar medium with APC conjugated secondary Ab's, again for at least 30 minutes on ice. After two times washing with ice-cold PBS, the binding of mAb's to cells was visualized using a BD Biosciences FACSarray.
[0308] As shown in FIG. 17, all Z270 variants bind in a dose-dependent fashion to Ba/F3-CD94/NKG2A cells, but not to Ba/F3-CD94/NKG2C cells. Thus all variants specifically bind NKG2A, with humZ270 binding with similar efficacy to NKG2A as chimZ270, whereas recZ270 binds slightly more efficiently.
Example 14--humZ270 Induces Killing of HLA-E+ Target Cells by CD94/NKG2A-Expressing NKL Cells
[0309] The ability of recZ270, chimZ270, humZ270VL1NH1 and Z199 to induce killing of .sup.51Cr-labeled LCL 721.221-Cw3 cells by CD94/NKG2A+ NKL cells was investigated. In this assay, .sup.51Cr-labeled LCL 721.221-Cw3 target-cells (HLA-E+) were incubated with NKL cells in a humidified incubator containing 5% CO.sub.2, for 4 hours at 37.degree. C. (E:T ratio=6:1), in the presence or absence of various concentrations of anti-NKG2A mAb's. The killing of target-cells was analyzed by measuring the amount of .sup.51Cr in the tissue-culture medium, which was released by target cells upon killing.
[0310] In FIG. 18, it shown that increasing concentrations of anti-NKG2A antibody induced the killing of LCL 721.221-Cw3 cells by NKL cells. Z199, chimZ270 and recZ270 were all equally efficient, whereas humZ270 induced higher killing of LCL 721.221-Cw3 cells by NKL cells. Thus, humZ270 can efficiently block the inhibitory function of CD94/NKG2A on CD94/NKG2A-expressing cytotoxic lymphocytes, such as subsets of NK-cells, NKT-cells, .alpha./.beta. T-cells and .gamma./.delta. T-cells, and was more efficient than the other recombinant variants tested.
Example 15--humZ270 is a Competitive CD94/NKG2A Antagonist
[0311] To test whether humZ270VL1NH1 prevents ligand (i.e. HLA-E) binding to CD94/NKG2A, we analyzed whether humZ270 could prevent the binding of HLA-E tetramers to CD94/NKG2A over-expressing Ba/F3 cells (Ba/F3-CD94/NKG2A). For this, Ba/F3-CD94/NKG2A were incubated with 1) various concentrations of humZ270 or 2) first incubated with a saturing concentration of HLA-E tetramers (4.7 .mu.g/ml) and then incubated with various concentrations of humZ270. All incubations were performed in tissue-culture medium containing 2% FCS, on ice. Subsequently, cells were incubated with APC-conjugated secondary antibodies specific for mouse Ab's, and analyzed by flowcytometry using a BD Biosciences FACSarray.
[0312] As shown in FIG. 19, humZ270 efficiently binds Ba/F3-CD94/NKG2A cells in a concentration dependent fashion (diamonds). However, when cells were pre-incubated with HLA-E tetramers, humZ270 was prevented from binding to Ba/F3-CD94/NKG2A cells. Thus HumZ270 and HLA-E bind overlapping epitopes on CD94/NKG2A. Therefore, the CD94/NKG2A-inhibitory effect of humZ270 in NK-cytotoxicity assays is likely a consequence of preventing the ability of HLA-E inducing negative signals to cytotoxic lymphocytes via CD94/NKG2A. As such, humZ270 can be considered a competitive CD94/NKG2A antagonist.
Example 16--humZ270 Specifically Binds to CD94/NKG2A
[0313] The specificity and efficacy of humZ270VL1NH1, and various humZ270VL1NH1 variants with back-mutations in the variable light (VL) or variable heavy (VH) regions were tested for binding to CD94/NKG2A in flow-cytometry. For this, Ba/F3-CD94/NKG2A cells were incubated with humZ270-L46F (VL), humZ270-148V (VL), humZ270-L46F/148V (VL), humZ270-V5Q (VH), humZ270-M69L (VH), humZ270-T71V (VH), humZ270-T73K (VH), humZ270-T75S (VH), humZ270-V5Q/M69LJT71V/T73K/T75S (VH), humZ270-M69L/T71V/T73KIT75S (VH) or two different batches of humZ270VL1NH1 without back-mutations ("DK" and "CHN"). For this purpose, Ba/F3-CD94/NKG2A or -C cells were incubated with various concentrations of the humZ270 variants in tissue-culture medium containing 2% FCS, for at least 30 minutes on ice. The cells were then washed, and incubated in similar medium with APC conjugated secondary antibodies specific for human antibodies, again for at least 30 minutes on ice. After two times washing with ice-cold PBS, the binding of secondary antibodies to cells was visualized using a BD Biosciences FACSarray.
[0314] All variants bound specifically to CD94/NKG2A, and not to CD94/NKG2C. All variants bound with similar efficacy to CD94/NKG2A, with the exception of variants containing the V5Q (VL) mutation, which bound slightly less efficiently (see FIG. 20).
Exemplary Embodiments
[0315] The following paragraphs describe exemplary embodiments of the invention.
[0316] 1. An antibody that specifically binds NKG2A, comprising antigen-binding residues from the complementarity-determining regions (CDRs) of murine antibody Z270 and human acceptor framework sequences, wherein at least the 6 C-terminal amino acid residues of the CDR-H2 are the same as those in the variable heavy (VH) human acceptor sequence.
[0317] 2. The antibody of embodiment 1, which is more effective than an antibody comprising murine antibody Z270 variable light (VL) and VH sequences in potentiating the cytotoxic activity of a CD94/NKG2A-expressing cytotoxic lymphocyte.
[0318] 3. The antibody of embodiment 1, which is more effective than an antibody comprising murine antibody Z270 variable light (VL) and VH sequences in neutralizing the inhibitory activity of a CD94/NKG2A receptor expressed on the surface of a cytotoxic lymphocyte.
[0319] 4. The antibody of embodiment 1, which is more effective than an antibody comprising murine antibody Z270 variable light (VL) and VH sequences in reducing CD94/NKG2A-mediated inhibition of the cytotoxic activity of a CD94/NKG2A-expressing cytotoxic lymphocyte.
[0320] 5. The antibody of embodiment 1, which is more effective than an antibody comprising murine antibody Z270 variable light (VL) and VH sequences in inducing the killing of a Cw3-expressing target cell by a CD94/NKG2A-expressing cytotoxic lymphocyte.
[0321] 6. The antibody of any of embodiments 2-5, wherein the CD94/NKG2A-expressing cytotoxic lymphocyte is an NK cell, an NKT cell, an .alpha./.beta. T-cell, or a .gamma./.delta. T-cell.
[0322] 7. The antibody of embodiment 6, wherein the CD94/NKG2A-expressing cytotoxic lymphocyte is an NK cell.
[0323] 8. The antibody of any of embodiments 1-7, wherein the antibody VH domain comprises residues D52, D54, F99, T(100C), and W(100F) from the VH CDRs of murine antibody Z270.
[0324] 9. The antibody of embodiment 8, wherein the antibody VH domain further comprises residues N35, Y53, E56, D98, V(100A), and L(100D) from the VH CDRs of murine antibody Z270.
[0325] 10. The antibody of any of embodiments 1-9, comprising a CDR-H1 corresponding to residues 31-35 of SEQ ID NO:5 and a CDR-H3 corresponding to residues 95-102 of SEQ ID NO:5, wherein the CDR-H2 comprises residues 50-59 of SEQ ID NO:5.
[0326] 11. The antibody of any of embodiments 1-10, wherein the VH domain human acceptor framework has 70% or more sequence identity to SEQ ID NO:5.
[0327] 12. The antibody of any of embodiments 1-11, wherein the VH segment of the VH human acceptor framework is VH1_18, VH5_a, VH5_51, VH1_f, or VH1_46, and the J-segment is JH6.
[0328] 13. The antibody of embodiment 12, wherein the VH segment is VH1_18, VH5_a, VH5_51, or VH1_f.
[0329] 14. The antibody of embodiment 13, wherein the VH segment is VH1_18.
[0330] 15. The antibody of any of embodiments 1-14, wherein the VH domain human acceptor sequence is free of any back-mutations.
[0331] 16. The antibody of any of embodiments 1-14, wherein
[0332] (a) the amino acid at position 5 of the VH domain is V or Q;
[0333] (b) the amino acid at position 69 of the VH domain is M or L;
[0334] (c) the amino acid at position 71 of the VH domain is T or V;
[0335] (d) the amino acid at position 73 of the VH domain is T or K; or
[0336] (e) the amino acid at position 75 of the VH domain is T or S.
[0337] 17. The antibody of embodiment 16, wherein the amino acid at position 69 is L.
[0338] 18. The antibody of embodiment 16, wherein the amino acid at position 71 is V.
[0339] 19. The antibody of any of embodiments 1-15, wherein the VH domain comprises the sequence of SEQ ID NO:5.
[0340] 20. The antibody of any of embodiments 1-19, comprising a CDR-L1 corresponding to residues 24-34 of SEQ ID NO:4, a CDR-L2 corresponding to residues 50-56 of SEQ ID NO:4, and a CDR-L3 corresponding to residues 89-97 of SEQ ID NO:4.
[0341] 21. The antibody of embodiment 20, wherein the VL domain human acceptor sequence is free of any back-mutations.
[0342] 22. The antibody of any of embodiments 20-21, wherein the VL domain human acceptor framework is from VKI_02/JK4.
[0343] 23. The antibody of any of embodiments 20-22, wherein the VL domain human acceptor framework comprises SEQ ID NO:4.
[0344] 24. A humanized antibody or antibody fragment that specifically binds NKG2A, comprising
[0345] (a) a CDR-L1 corresponding to residues 24-34 of SEQ ID NO:4;
[0346] (b) a CDR-L2 corresponding to residues 50-56 of SEQ ID NO:4;
[0347] (c) a CDR-L3 corresponding to residues 89-97 of SEQ ID NO:4;
[0348] (d) a CDR-H1 corresponding to residues 31-35 of SEQ ID NO:5;
[0349] (e) a CDR-H3 corresponding to residues 95-102 of SEQ ID NO:5; and
[0350] (f) a CDR-H2 comprising residues 50-59 of SEQ ID NO:5; and
[0351] (g) human acceptor framework sequences;
[0352] wherein residues 60-65 in the CDR-H2 are from the VH human acceptor sequence, and
[0353] wherein the humanized antibody is more effective than an antibody comprising a variable light (VL) sequence corresponding to SEQ ID NO:1 and a VH sequences corresponding to SEQ ID NO:2 in potentiating the cytotoxic activity of a CD94/NKG2A-expressing NK cell.
[0354] 25. A humanized antibody that binds human NKG2A, the antibody comprising a VH domain that comprises non-human CDR residues and a human VH acceptor framework, the VH domain comprising a CDR-H1 corresponding to residues 31-35 of SEQ ID NO:5, a CDR-H2 corresponding to residues 50-65 of SEQ ID NO:5, and a CDR-H3 corresponding to residues 95-102 of SEQ ID NO:5.
[0355] 26. The humanized antibody of embodiment 25, wherein the human VH acceptor framework does not comprise any back-mutation.
[0356] 27. The humanized antibody of embodiment 25, wherein the amino acid at Kabat position 5 of the VH domain is V or Q.
[0357] 28. The humanized antibody of embodiment 25, wherein the amino acid at Kabat position of the VH domain 69 is M or L.
[0358] 29. The humanized antibody of embodiment 25, wherein the amino acid at Kabat position 71 of the VH domain is T or V.
[0359] 30. The humanized antibody of embodiment 25, wherein the amino acid at Kabat position 73 of the VH domain is T or K.
[0360] 31. The humanized antibody of embodiment 25, wherein the amino acid at Kabat position 75 of the VH domain is T or S.
[0361] 32. The humanized antibody of embodiment 25, wherein the VH domain comprises a framework region substitution in at least one Kabat position selected from the group consisting of 5, 69, 71, 73, and 75.
[0362] 33. The humanized antibody of embodiment 32, wherein the VH domain comprises the amino acid sequence of SEQ ID NO:5, with framework substitutions according to any one of the following options:
[0363] (a) none
[0364] (b) V5Q
[0365] (c) M69L
[0366] (d) T71V
[0367] (e) T73K
[0368] (f) T75S
[0369] (g) V5Q and M69L
[0370] (h) V5Q and T71V
[0371] (i) V5Q and T73K
[0372] (j) V5Q and T75S
[0373] (k) M69L and T71V
[0374] (l) M69L and T73K
[0375] (m) M69L and T75S
[0376] (n) T71V and T73K
[0377] (o) T71V and T75S
[0378] (p) T73K and T75S
[0379] (q) V5Q, T73K and T75S
[0380] (r) V5Q, T71V and T75S
[0381] (s) V5Q, T71V and T73K
[0382] (t) V5Q, M69L and T75S
[0383] (u) V5Q, M69L and T73K
[0384] (v) V5Q, M69L and T71V
[0385] (w) T71V, T73K and T75S
[0386] (x) M69L, T73K and T75S
[0387] (y) M69L, T71V and T75S,
[0388] (z) M69L, T71V and T73K,
[0389] (aa) V5Q, M69L, T71V and T73K,
[0390] (bb) V5Q, M69L, T71V and T75S,
[0391] (cc) V5Q, M69L, T73K and T75S,
[0392] (dd) V5Q, T71V, T73K and T75S,
[0393] (ee) M69L, T71V, T73K and T75S, and
[0394] (ff) V5Q, M69L, T71V, T73K and T75S.
[0395] 34. The humanized antibody of any of embodiments 25-32, comprising a VL domain that comprises non-human CDR residues incorporated into a human VL acceptor framework, the VL domain comprising a CDR-L1 corresponding to residues 24-34 of SEQ ID NO:4, a CDR-L2 corresponding to residues 50-56 of SEQ ID NO:4, and an CDR-L3 corresponding to residues 89-97 of SEQ ID NO:4.
[0396] 35. The humanized antibody of embodiment 34, wherein the human VL acceptor framework does not comprise any back-mutation.
[0397] 36. The humanized antibody of embodiment 34, wherein the amino acid at Kabat position 46 of the VL domain is L or F.
[0398] 37. The humanized antibody of embodiment 34, wherein the amino acid at Kabat position 48 of the VL domain is I or V.
[0399] 38. The humanized antibody of embodiment 34, wherein the VL domain comprises a framework region substitution in at least one Kabat position selected from 46 and 48.
[0400] 39. The humanized antibody of embodiment 38, wherein the VL domain comprises the amino acid sequence of SEQ ID NO:4, with framework substitutions according to any one of the following options:
[0401] (a) None
[0402] (b) L46F
[0403] (c) 148V
[0404] (d) L46 and 148V.
[0405] 40. A humanized antibody that binds human NKG2A, the antibody comprising a VH domain that comprises non-human CDR residues incorporated into a human VH domain, the VH domain comprising a framework region substitution in at least one Kabat position in SEQ ID NO:7 selected from the group consisting of 5, 69, 71, 73, and 75.
[0406] 41. The humanized antibody of embodiment 40, comprising a V5Q substitution.
[0407] 42. The humanized antibody of embodiment 40, comprising a M69L substitution.
[0408] 43. The humanized antibody of embodiment 40, comprising a T71V substitution.
[0409] 44. The humanized antibody of embodiment 40, comprising a T73K substitution.
[0410] 45. The humanized antibody of embodiment 40, comprising a T75S substitution.
[0411] 46. The humanized antibody of embodiment 40, comprising a VL domain that comprises non-human CDR residues incorporated into a human VL domain, the VL domain comprising a framework region substitution in at least one Kabat position in SEQ ID NO:6 selected from 46 and 48.
[0412] 47. The humanized antibody of embodiment 40, comprising a L46F substitution.
[0413] 48. The humanized antibody of embodiment 40, comprising a 148V substitution.
[0414] 49. The humanized antibody of any of embodiments 40-48, comprising a CDR-H1 corresponding to residues 31-35 of SEQ ID NO:5, a CDR-H2 corresponding to residues 50-66 of SEQ ID NO:5, and a CDR-H3 corresponding to residues 95-102 of SEQ ID NO:5.
[0415] 50. The humanized antibody of any of embodiments 40-49, comprising a VH domain comprising the sequence of SEQ ID NO:7.
[0416] 51. The humanized antibody of any of embodiments 40-50, comprising a CDR-L1 corresponding to residues 24-34 of SEQ ID NO:6, a CDR-L2 corresponding to residues 50-56 of SEQ ID NO:6, and an CDR-L3 corresponding to residues 89-97 of SEQ ID NO:6.
[0417] 52. The humanized antibody of any of embodiments 40-52, comprising a VL domain comprising the sequence of SEQ ID NO:6.
[0418] 53. A humanized antibody that binds human NKG2A, the antibody comprising a VH domain that comprises the amino acid sequence of SEQ ID NO:5, optionally with one or more FR substitutions at Kabat positions 5, 69, 71, 73, and/or 75.
[0419] 54. The humanized antibody of embodiment 53, wherein the optional FR substitutions are V5Q, M69L, T71V, T73K, and/or T75S.
[0420] 55. The humanized antibody of embodiment 52, further comprising a VL domain that comprises the amino acid sequence of SEQ ID NO:4, optionally with one or more FR substitutions at Kabat positions 46 and/or 48.
[0421] 56. The humanized antibody of embodiment 55, wherein the optional FR substitutions are L46F and/or 148V.
[0422] 57. A humanized antibody that binds NKG2A, comprising a VH domain that comprises non-human CDR residues incorporated into a human VH domain, wherein the VH domain is at least 50% identical to SEQ ID NO:5.
[0423] 58. The humanized antibody of embodiment 46, wherein the VH domain is at least 90% identical to SEQ ID NO:5.
[0424] 59. The humanized antibody of any of embodiments 57 and 58, comprising a CDR-H1 corresponding to residues 31-35 of SEQ ID NO:5, a CDR-H2 corresponding to residues 50-66 of SEQ ID NO:5, and a CDR-H3 corresponding to residues 95-102 of SEQ ID NO:5.
[0425] 60. The humanized antibody of any of embodiments 57-59, comprising a V or Q at Kabat position 5, an M or L at Kabat position 69, a T or V at Kabat position 71, a T or K at Kabat position 73, and a T or S at Kabat position 75, in the VH domain.
[0426] 61. The humanized antibody of any of embodiments 57-60, comprising a VL domain that comprises non-human CDR residues incorporated into a human VL domain, wherein the VL domain is at least 50% identical to SEQ ID NO:4.
[0427] 62. The humanized antibody of any of embodiments 57-61, comprising a VL domain at least 90% identical to SEQ ID NO:4.
[0428] 63. The humanized antibody of any of embodiments 57-62, comprising a CDR-L1 sequence corresponding to residues 24-34 of SEQ ID NO:4, a CDR-L2 sequence corresponding to residues 50-56 of SEQ ID NO:4, and an CDR-L3 sequence corresponding to residues 89-97 of SEQ ID NO:4.
[0429] 64. The humanized antibody of any of embodiments 57-63, comprising a L or F at Kabat position 46 and/or an I or V at Kabat position 48.
[0430] 65. A humanized antibody that binds human NKG2A, the antibody comprising a VH domain that comprises non-human CDR residues incorporated into a human VH domain, the VH domain comprising a CDR-H1 corresponding to residues 31-35 of SEQ ID NO:8, a CDR-H2 corresponding to residues 50-66 of SEQ ID NO:8, and a CDR-H3 corresponding to residues 95-102 of SEQ ID NO:8, wherein the amino acids at Kabat positions 63, 64, 65, 66, and 67 are F, K, D, K, A, respectively.
[0431] 66. The humanized antibody of embodiment 65, wherein the amino acid at Kabat position 60 is A.
[0432] 67. The humanized antibody of any of embodiments 65 and 66, wherein the VH domain comprises the amino acid sequence of SEQ ID NO:8.
[0433] 68. The humanized antibody of any of embodiments 65-67, further comprising a VL domain comprising a CDR-L1 corresponding to residues 24-34 of SEQ ID NO:6, a CDR-L2 corresponding to residues 50-56 of SEQ ID NO:6, and an CDR-L3 corresponding to residues 89-97 of SEQ ID NO:6.
[0434] 69. The humanized antibody of any of embodiments 65-68, wherein the VL domain comprises the amino acid sequence of SEQ ID NO:6.
[0435] 70. A humanized version of an anti-NKG2A antibody produced by the Z270 hybridoma.
[0436] 71. The antibody of any of embodiments 1-70, which is a multispecific antibody or an antibody fragment.
[0437] 72. The antibody of embodiment 71, which is an antibody fragment selected from a Fab, a Fab', a F(ab).sub.2, a F(ab').sub.2, a F(ab).sub.3, an Fv, a single-chain Fv, a dsFv, an Fd fragment, a dAb fragment, a minibody, a diabody, a triabody, a tetrabody, a kappa body; a camel IgG; an IgNAR; and a multispecific antibody fragment.
[0438] 73. The antibody of embodiment 72, which is a bispecific antibody.
[0439] 74. The antibody of any of embodiments 1-70, which is a full-length IgG4 antibody or fragment thereof.
[0440] 75. The antibody of embodiment 74, wherein the heavy-chain constant domain comprises an S241P mutation.
[0441] 76. An isolated nucleic acid encoding the antibody of any of the preceding embodiments.
[0442] 77. A vector comprising the nucleic acid of embodiment 76.
[0443] 78. A host cell comprising the vector of embodiment 76.
[0444] 79. A method of producing an antibody comprising culturing the host cell of embodiment 78 so that the nucleic acid is expressed and the antibody produced.
[0445] 80. The method of embodiment 79, further comprising recovering the antibody from the host cell culture.
[0446] 81. The method of embodiment 79 wherein, before culturing, the host cell is co-transfected with a vector comprising nucleic acid encoding a variable heavy domain and with a vector comprising nucleic acid encoding a variable light domain.
[0447] 82. An immunoconjugate comprising an antibody according to any of embodiments 1-75 and a second agent.
[0448] 83. The immunoconjugate of embodiment 82, wherein the second agent is a cytotoxic agent.
[0449] 84. The immunoconjugate of embodiment 82, wherein the second agent is a PEG-molecule.
[0450] 85. A pharmaceutical composition comprising the antibody of any of embodiments 1-75 or the immunoconjugate of any of embodiments 82-84, and a carrier.
[0451] 86. The pharmaceutical composition of embodiment 74, comprising a buffer selected from citrate, phosphate, and a combination thereof, having a pH from about 6.0 to about 7.5.
[0452] 87. An article of manufacture comprising a container containing the antibody of any of embodiments 1-75 and instructions directing a user to treat a disorder selected from a cancer, a viral disease, an inflammatory disorder, and an autoimmune disorder, in a mammal with the antibody in an effective amount.
[0453] 88. The article of embodiment 87, wherein the mammal is a human.
[0454] 89. The antibody of any of embodiments 1-75 for use in neutralizing the inhibitory activity of a CD94/NKG2A receptor expressed on the surface of a cytotoxic lymphocyte in a human patient.
[0455] 90. The antibody of any of embodiments 1-75 for use in reducing CD94/NKG2A-mediated inhibition of the cytotoxicity activity of a CD94/NKG2A-expressing cytotoxic lymphocyte in a human patient.
[0456] 91. The antibody of any of embodiments 1-75 for use in potentiating the cytotoxic activity of a CD94/NKG2A-expressing cytotoxic lymphocyte in a human patient.
[0457] 92. The antibody of any of embodiments 1-75 for use in inducing the killing of a Cw3-expressing target cell by a CD94/NKG2A-expressing cytotoxic lymphocyte in a human patient.
[0458] 93. The antibody of any embodiments 89-92, wherein the CD94/NKG2A-expressing cytotoxic lymphocyte is an NK cell, an NKT cell, an .alpha./.beta. T-cell, or a .gamma./.delta. T-cell.
[0459] 94. A method of treating a human patient suffering from a disorder selected from a cancer, a viral disease, an inflammatory disorder, and an autoimmune disorder, comprising administering the pharmaceutical composition of any of embodiments 85-86.
[0460] 95. The use of the antibody of any of embodiments 1-75 in the preparation of a medicament for administration to a human patient suffering from a disorder selected from a cancer, a viral disease, an inflammatory disorder, and an autoimmune disorder.
[0461] 96. The antibody of any of embodiments 1-75 for use in the treatment of a human patient suffering from a disorder selected from a cancer, a viral disease, an inflammatory disorder, and an autoimmune disorder.
[0462] 97. The method, use, or antibody of any of embodiments 89-96, wherein the patient suffers from squamous cell carcinoma, leukemia, acute lymphocytic leukemia, acute lymphoblastic leukemia, B-cell lymphoma, T-cell lymphoma, Hodgkin's lymphoma, non-Hodgkin's lymphoma, hairy cell lymphoma, Burkitt's lymphoma, multiple myeloma, acute or chronic myelogenous leukemias, promyelocytic leukemia, fibrosarcoma, rhabdomyosarcoma; melanoma, seminoma, teratocarcinoma, neuroblastoma, astrocytoma, neuroblastoma, glioma, schwannomas; fibrosarcoma, rhabdomyosarcoma, osteosarcoma, melanoma, xeroderma pigmentosum, keratoacanthoma, seminoma, thyroid follicular cancer, teratocarcinoma, other carcinoma of the bladder, breast, colon, kidney, liver, lung, ovary, prostate, pancreas, stomach, cervix, thyroid or skin, other hematopoietic tumors of lymphoid lineage, other hematopoietic tumors of myeloid lineage, other tumors of mesenchymal origin, other tumors of the central or peripheral nervous system, or other tumors of mesenchymal origin.
[0463] 98. The use according to embodiment 97, wherein the patient suffers from multiple myeloma, Non-Hodgkin's lymphoma, or acute myelogeous lymphoma.
[0464] All references, including publications, patent applications and patents, cited herein are hereby incorporated by reference to the same extent as if each reference was individually and specifically indicated to be incorporated by reference and was set forth in its entirety herein.
[0465] All headings and sub-headings are used herein for convenience only and should not be construed as limiting the invention in any way,
[0466] Any combination of the above-described elements in all possible variations thereof is encompassed by the invention unless otherwise indicated herein or otherwise clearly contradicted by context.
[0467] Unless otherwise expressly indicated or clearly contradicted by context, the term "or" herein is used in the inclusive sense of "and/or", and, accordingly, as implicitly providing support for an embodiment or aspect in which the term is to be interpreted in the exclusive sense of "either this or that".
[0468] The terms "a" and "an" and "the" and similar referents as used in the context of describing the invention are to be construed to cover both the singular and the plural, unless otherwise indicated herein or clearly contradicted by context.
[0469] Recitation of ranges of values herein are merely intended to serve as a shorthand method of referring individually to each separate value falling within the range, unless otherwise indicated herein, and each separate value is incorporated into the specification as if it were individually recited herein. Unless otherwise stated, all exact values provided herein are representative of corresponding approximate values (e.g., all exact exemplary values provided with respect to a particular factor or measurement can be considered to also provide a corresponding approximate measurement, modified by "about," where appropriate).
[0470] All methods described herein can be performed in any suitable order unless otherwise indicated herein or otherwise clearly contradicted by context.
[0471] The use of any and all examples, or exemplary language (e.g., "such as") provided herein, is intended merely to better illuminate the invention and does not pose a limitation on the scope of the invention unless otherwise indicated. No language in the specification should be construed as indicating any element is essential to the practice of the invention unless as much is explicitly stated.
[0472] The citation and incorporation of patent documents herein is done for convenience only and does not reflect any view of the validity, patentability and/or enforceability of such patent documents,
[0473] The description herein of any aspect or embodiment of the invention using terms such as "comprising", "having", "including" or "containing" with reference to an element or elements is intended to provide support for a similar aspect or embodiment of the invention that "consists of", "consists essentially of", or "substantially comprises" that particular element or elements, unless otherwise stated or clearly contradicted by context (e.g., a composition described herein as comprising a particular element should be understood as also describing a composition consisting of that element, unless otherwise stated or clearly contradicted by context).
[0474] This invention includes all modifications and equivalents of the subject matter recited in the aspects or claims presented herein to the maximum extent permitted by applicable law.
Sequence CWU 1
1
291107PRTMus musculus 1Asp Ile Gln Met Thr Gln Ser Pro Ala Ser Leu
Ser Ala Ser Val Gly1 5 10 15Glu Thr Val Thr Ile Thr Cys Arg Ala Ser
Glu Asn Ile Tyr Ser Tyr 20 25 30Leu Ala Trp Tyr Gln Gln Lys Gln Gly
Lys Ser Pro Gln Phe Leu Val 35 40 45Tyr Asn Ala Lys Thr Leu Ala Glu
Gly Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr Gln Phe
Ser Leu Lys Ile Asn Ser Leu Gln Pro65 70 75 80Glu Asp Phe Gly Ser
Tyr Tyr Cys Gln His His Tyr Gly Thr Pro Arg 85 90 95Thr Phe Gly Gly
Gly Thr Lys Leu Glu Ile Lys 100 1052124PRTMus musculus 2Gln Val Gln
Leu Gln Gln Pro Gly Ala Glu Leu Val Arg Pro Gly Ala1 5 10 15Ser Val
Lys Leu Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Ser Tyr 20 25 30Trp
Met Asn Trp Val Lys Gln Arg Pro Glu Gln Gly Leu Gln Trp Ile 35 40
45Gly Arg Ile Asp Pro Tyr Asp Ser Glu Thr His Tyr Ser Gln Lys Phe
50 55 60Lys Asp Lys Ala Ile Leu Thr Val Asp Lys Ser Ser Ser Thr Ala
Tyr65 70 75 80Met Arg Leu Ser Ser Leu Thr Ser Glu Asp Ser Ala Val
Tyr Tyr Cys 85 90 95Ala Arg Gly Gly Tyr Asp Phe Asp Val Gly Thr Leu
Tyr Trp Phe Phe 100 105 110Asp Val Trp Gly Ala Gly Thr Thr Val Thr
Val Ser 115 1203107PRTMus musculus 3Asn Ile Val Met Thr Gln Ser Pro
Lys Ser Met Ser Met Ser Val Gly1 5 10 15Glu Arg Val Thr Leu Thr Cys
Lys Ala Ser Glu Asn Val Val Thr Tyr 20 25 30Val Ser Trp Tyr Gln Gln
Lys Pro Glu Gln Ser Pro Lys Leu Leu Ile 35 40 45Tyr Gly Ala Ser Asn
Arg Tyr Thr Gly Val Pro Asp Arg Phe Thr Gly 50 55 60Ser Gly Ser Ala
Thr Asp Phe Thr Leu Thr Ile Ser Ser Val Gln Ala65 70 75 80Glu Asp
Leu Ala Asp Tyr His Cys Gly Gln Gly Tyr Ser Tyr Pro Tyr 85 90 95Thr
Phe Gly Gly Gly Thr Lys Leu Glu Ile Lys 100
1054107PRTArtificialHumanized sequence 4Asp Ile Gln Met Thr Gln Ser
Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile Thr
Cys Arg Ala Ser Glu Asn Ile Tyr Ser Tyr 20 25 30Leu Ala Trp Tyr Gln
Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu Ile 35 40 45Tyr Asn Ala Lys
Thr Leu Ala Glu Gly Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser
Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65 70 75 80Glu
Asp Phe Ala Thr Tyr Tyr Cys Gln His His Tyr Gly Thr Pro Arg 85 90
95Thr Phe Gly Gly Gly Thr Lys Val Glu Ile Lys 100
1055124PRTArtificialHumanized sequence 5Gln Val Gln Leu Val Gln Ser
Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys Val Ser Cys
Lys Ala Ser Gly Tyr Thr Phe Thr Ser Tyr 20 25 30Trp Met Asn Trp Val
Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Met 35 40 45Gly Arg Ile Asp
Pro Tyr Asp Ser Glu Thr His Tyr Ala Gln Lys Leu 50 55 60Gln Gly Arg
Val Thr Met Thr Thr Asp Thr Ser Thr Ser Thr Ala Tyr65 70 75 80Met
Glu Leu Arg Ser Leu Arg Ser Asp Asp Thr Ala Val Tyr Tyr Cys 85 90
95Ala Arg Gly Gly Tyr Asp Phe Asp Val Gly Thr Leu Tyr Trp Phe Phe
100 105 110Asp Val Trp Gly Gln Gly Thr Thr Val Thr Val Ser 115
1206107PRTArtificialHumanized sequenceUNSURE(46)..(46)Xaa is L or
FUNSURE(48)..(48)Xaa is I or V 6Asp Ile Gln Met Thr Gln Ser Pro Ser
Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys Arg
Ala Ser Glu Asn Ile Tyr Ser Tyr 20 25 30Leu Ala Trp Tyr Gln Gln Lys
Pro Gly Lys Ala Pro Lys Xaa Leu Xaa 35 40 45Tyr Asn Ala Lys Thr Leu
Ala Glu Gly Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr
Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe
Ala Thr Tyr Tyr Cys Gln His His Tyr Gly Thr Pro Arg 85 90 95Thr Phe
Gly Gly Gly Thr Lys Val Glu Ile Lys 100
1057124PRTArtificialHumanized sequenceUNSURE(5)..(5)Xaa is V or
QUNSURE(70)..(70)Kabat position 69; Xaa is M or
LUNSURE(72)..(72)Kabat position 71; Xaa is T or
VUNSURE(74)..(74)Kabat position 73; Xaa is T or
KUNSURE(76)..(76)Kabat position 75; Xaa is T or S 7Gln Val Gln Leu
Xaa Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys
Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Ser Tyr 20 25 30Trp Met
Asn Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Met 35 40 45Gly
Arg Ile Asp Pro Tyr Asp Ser Glu Thr His Tyr Ala Gln Lys Leu 50 55
60Gln Gly Arg Val Thr Xaa Thr Xaa Asp Xaa Ser Xaa Ser Thr Ala Tyr65
70 75 80Met Glu Leu Arg Ser Leu Arg Ser Asp Asp Thr Ala Val Tyr Tyr
Cys 85 90 95Ala Arg Gly Gly Tyr Asp Phe Asp Val Gly Thr Leu Tyr Trp
Phe Phe 100 105 110Asp Val Trp Gly Gln Gly Thr Thr Val Thr Val Ser
115 1208124PRTArtificialHumanized sequenceUNSURE(5)..(5)Xaa is V or
QUNSURE(61)..(61)Kabat position 60; Xaa is S or
AUNSURE(64)..(64)Kabat position 63; Xaa is L or
FUNSURE(65)..(65)Kabat position 64; Xaa is Q or
KUNSURE(66)..(66)Kabat position 65; Xaa is G or
DUNSURE(67)..(67)Kabat position 66; Xaa is R or
KUNSURE(68)..(68)Kabat position 67; Xaa is V or
AUNSURE(70)..(70)Kabat position 69; Xaa is M or
LUNSURE(72)..(72)Kabat position 71; Xaa is T or
VUNSURE(74)..(74)Kabat position 73; Xaa is T or
KUNSURE(76)..(76)Kabat position 75; Xaa is T or S 8Gln Val Gln Leu
Xaa Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys
Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Ser Tyr 20 25 30Trp Met
Asn Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Met 35 40 45Gly
Arg Ile Asp Pro Tyr Asp Ser Glu Thr His Tyr Xaa Gln Lys Xaa 50 55
60Xaa Xaa Xaa Xaa Thr Xaa Thr Xaa Asp Xaa Ser Xaa Ser Thr Ala Tyr65
70 75 80Met Glu Leu Arg Ser Leu Arg Ser Asp Asp Thr Ala Val Tyr Tyr
Cys 85 90 95Ala Arg Gly Gly Tyr Asp Phe Asp Val Gly Thr Leu Tyr Trp
Phe Phe 100 105 110Asp Val Trp Gly Gln Gly Thr Thr Val Thr Val Ser
115 1209107PRTHomo sapiens 9Asp Ile Gln Met Thr Gln Ser Pro Ser Ser
Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile Thr Cys Arg Ala
Ser Gln Ser Ile Ser Ser Tyr 20 25 30Leu Asn Trp Tyr Gln Gln Lys Pro
Gly Lys Ala Pro Lys Leu Leu Ile 35 40 45Tyr Ala Ala Ser Ser Leu Gln
Ser Gly Val Pro Ser Arg Phe Ser Gly 50 55 60Ser Gly Ser Gly Thr Asp
Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro65 70 75 80Glu Asp Phe Ala
Thr Tyr Tyr Cys Gln Gln Ser Tyr Ser Thr Pro Leu 85 90 95Thr Phe Gly
Gly Gly Thr Lys Val Glu Ile Lys 100 10510117PRTHomo sapiens 10Gln
Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10
15Ser Val Lys Val Ser Cys Lys Ala Ser Gly Tyr Thr Phe Thr Ser Tyr
20 25 30Gly Ile Ser Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp
Met 35 40 45Gly Trp Ile Ser Ala Tyr Asn Gly Asn Thr Asn Tyr Ala Gln
Lys Leu 50 55 60Gln Gly Arg Val Thr Met Thr Thr Asp Thr Ser Thr Ser
Thr Ala Tyr65 70 75 80Met Glu Leu Arg Ser Leu Arg Ser Asp Asp Thr
Ala Val Tyr Tyr Cys 85 90 95Ala Arg Tyr Tyr Tyr Tyr Tyr Gly Met Asp
Val Trp Gly Gln Gly Thr 100 105 110Thr Val Thr Val Ser
11511233PRTHomo sapiens 11Met Asp Asn Gln Gly Val Ile Tyr Ser Asp
Leu Asn Leu Pro Pro Asn1 5 10 15Pro Lys Arg Gln Gln Arg Lys Pro Lys
Gly Asn Lys Ser Ser Ile Leu 20 25 30Ala Thr Glu Gln Glu Ile Thr Tyr
Ala Glu Leu Asn Leu Gln Lys Ala 35 40 45Ser Gln Asp Phe Gln Gly Asn
Asp Lys Thr Tyr His Cys Lys Asp Leu 50 55 60Pro Ser Ala Pro Glu Lys
Leu Ile Val Gly Ile Leu Gly Ile Ile Cys65 70 75 80Leu Ile Leu Met
Ala Ser Val Val Thr Ile Val Val Ile Pro Ser Thr 85 90 95Leu Ile Gln
Arg His Asn Asn Ser Ser Leu Asn Thr Arg Thr Gln Lys 100 105 110Ala
Arg His Cys Gly His Cys Pro Glu Glu Trp Ile Thr Tyr Ser Asn 115 120
125Ser Cys Tyr Tyr Ile Gly Lys Glu Arg Arg Thr Trp Glu Glu Ser Leu
130 135 140Leu Ala Cys Thr Ser Lys Asn Ser Ser Leu Leu Ser Ile Asp
Asn Glu145 150 155 160Glu Glu Met Lys Phe Leu Ser Ile Ile Ser Pro
Ser Ser Trp Ile Gly 165 170 175Val Phe Arg Asn Ser Ser His His Pro
Trp Val Thr Met Asn Gly Leu 180 185 190Ala Phe Lys His Glu Ile Lys
Asp Ser Asp Asn Ala Glu Leu Asn Cys 195 200 205Ala Val Leu Gln Val
Asn Arg Leu Lys Ser Ala Gln Cys Gly Ser Ser 210 215 220Ile Ile Tyr
His Cys Lys His Lys Leu225 2301220DNAArtificialPrimer 12gccagtggat
agacagatgg 201321DNAArtificialPrimer 13gatggataca gttggtgcag c
2114381DNAMus musculus 14atgagtgtgc ccactcaggt cctggggttg
ctgctgctgt ggcttacagg tgccagatgt 60gacatccaga tgactcagtc tccagcctcc
ctatctgcat ctgtgggaga aactgtcacc 120atcacatgtc gagcaagtga
gaatatttac agttatttag catggtatca gcagaaacag 180ggaaaatctc
ctcagttctt ggtctataat gcaaaaacct tagcagaagg tgtgccatca
240aggttcagtg gcagtggatc aggcacacag ttttctctga agatcaacag
cctgcagcct 300gaagattttg ggagttatta ctgtcaacat cactatggta
ctcctcggac gttcggtgga 360ggcaccaagc tggaaatcaa a 38115127PRTMus
musculus 15Met Ser Val Pro Thr Gln Val Leu Gly Leu Leu Leu Leu Trp
Leu Thr1 5 10 15Gly Ala Arg Cys Asp Ile Gln Met Thr Gln Ser Pro Ala
Ser Leu Ser 20 25 30Ala Ser Val Gly Glu Thr Val Thr Ile Thr Cys Arg
Ala Ser Glu Asn 35 40 45Ile Tyr Ser Tyr Leu Ala Trp Tyr Gln Gln Lys
Gln Gly Lys Ser Pro 50 55 60Gln Phe Leu Val Tyr Asn Ala Lys Thr Leu
Ala Glu Gly Val Pro Ser65 70 75 80Arg Phe Ser Gly Ser Gly Ser Gly
Thr Gln Phe Ser Leu Lys Ile Asn 85 90 95Ser Leu Gln Pro Glu Asp Phe
Gly Ser Tyr Tyr Cys Gln His His Tyr 100 105 110Gly Thr Pro Arg Thr
Phe Gly Gly Gly Thr Lys Leu Glu Ile Lys 115 120 12516432DNAMus
musculus 16atgggatgga gctatatcat cctcttcttg ttagcaacag ctacatgtgt
ccactcccag 60gtccaactgc agcagcctgg ggctgagctg gtgaggcctg gggcttcagt
gaagctgtcc 120tgcaaggctt ctggctacac gttcaccagc tactggatga
actgggttaa gcagaggcct 180gagcaaggcc ttcagtggat tggaaggatt
gatccttacg atagtgaaac tcactacagt 240caaaagttca aggacaaggc
catattgact gtagacaaat cctccagcac agcctacatg 300cgactcagca
gcctgacatc tgaggactct gcggtctatt actgtgcaag agggggctat
360gatttcgacg taggaactct ctactggttc ttcgatgtct ggggcgcagg
gaccacggtc 420accgtctcct ca 43217144PRTMus musculus 17Met Gly Trp
Ser Tyr Ile Ile Leu Phe Leu Leu Ala Thr Ala Thr Cys1 5 10 15Val His
Ser Gln Val Gln Leu Gln Gln Pro Gly Ala Glu Leu Val Arg 20 25 30Pro
Gly Ala Ser Val Lys Leu Ser Cys Lys Ala Ser Gly Tyr Thr Phe 35 40
45Thr Ser Tyr Trp Met Asn Trp Val Lys Gln Arg Pro Glu Gln Gly Leu
50 55 60Gln Trp Ile Gly Arg Ile Asp Pro Tyr Asp Ser Glu Thr His Tyr
Ser65 70 75 80Gln Lys Phe Lys Asp Lys Ala Ile Leu Thr Val Asp Lys
Ser Ser Ser 85 90 95Thr Ala Tyr Met Arg Leu Ser Ser Leu Thr Ser Glu
Asp Ser Ala Val 100 105 110Tyr Tyr Cys Ala Arg Gly Gly Tyr Asp Phe
Asp Val Gly Thr Leu Tyr 115 120 125Trp Phe Phe Asp Val Trp Gly Ala
Gly Thr Thr Val Thr Val Ser Ser 130 135
14018723DNAArtificialChimeric sequence 18gaattcgcca ccatgagtgt
gcccactcag gtcctggggt tgctgctgct gtggcttaca 60ggtgccagat gtgacatcca
gatgactcag tctccagcct ccctatctgc atctgtggga 120gaaactgtca
ccatcacatg tcgagcaagt gagaatattt acagttattt agcatggtat
180cagcagaaac agggaaaatc tcctcagttc ttggtctata atgcaaaaac
cttagcagaa 240ggtgtgccat caaggttcag tggcagtgga tcaggcacac
agttttctct gaagatcaac 300agcctgcagc ctgaagattt tgggagttat
tactgtcaac atcactatgg tactcctcgg 360acgttcggtg gaggcaccaa
gctggaaatc aaacgggctg atgctgcacc aactgtatcc 420atcttcccac
catccagtga gcagttaaca tctggaggtg cctcagtcgt gtgcttcttg
480aacaacttct accccaaaga catcaatgtc aagtggaaga ttgatggcag
tgaacgacaa 540aatggcgtcc tgaacagttg gactgatcag gacagcaaag
acagcaccta cagcatgagc 600agcaccctca cgttgaccaa ggacgagtat
gaacgacata acagctatac ctgtgaggcc 660actcacaaga catcaacttc
acccattgtc aagagcttca acaggaatga gtgttaggct 720agc
723191419DNAArtificialChimeric sequence 19gaattcgcca ccatgggatg
gagctatatc atcctcttct tgttagcaac agctacatgt 60gtccactccc aggtccaact
gcagcagcct ggggctgagc tggtgaggcc tggggcttca 120gtgaagctgt
cctgcaaggc ttctggctac acgttcacca gctactggat gaactgggtt
180aagcagaggc ctgagcaagg ccttcagtgg attggaagga ttgatcctta
cgatagtgaa 240actcactaca gtcaaaagtt caaggacaag gccatattga
ctgtagacaa atcctccagc 300acagcctaca tgcgactcag cagcctgaca
tctgaggact ctgcggtcta ttactgtgca 360agagggggct atgatttcga
cgtaggaact ctctactggt tcttcgatgt ctggggcgca 420gggaccacgg
tcaccgtctc ctcagccaaa acgacacccc catctgtcta tccgctagcc
480cctggatctg ctgcccaaac taactccatg gtgaccctgg gatgcctggt
caagggctat 540ttccctgagc cagtgacagt gacctggaac tctggatccc
tgtccagcgg tgtgcacacc 600ttcccagctg tcctgcagtc tgacctctac
actctgagca gctcagtgac tgtcccctcc 660agcacctggc ccagcgagac
cgtcacctgc aacgttgccc acccggccag cagcaccaag 720gtggacaaga
aaattgtgcc cagggattgt ggttgtaagc cttgcatatg tacagtccca
780gaagtatcat ctgtcttcat cttcccccca aagcccaagg atgtgctcac
cattactctg 840actcctaagg tcacgtgtgt tgtggtagac atcagcaagg
atgatcccga ggtccagttc 900agctggtttg tagatgatgt ggaggtgcac
acagctcaga cgcaaccccg ggaggagcag 960ttcaacagca ctttccgctc
agtcagtgaa cttcccatca tgcaccagga ctggctcaat 1020ggcaaggagt
tcaaatgcag ggtcaacagt gcagctttcc ctgcccccat cgagaaaacc
1080atctccaaaa ccaaaggcag accgaaggct ccacaggtgt acaccattcc
acctcccaag 1140gagcagatgg ccaaggataa agtcagtctg acctgcatga
taacagactt cttccctgaa 1200gacattactg tggagtggca gtggaatggg
cagccagcgg agaactacaa gaacactcag 1260cccatcatgg acacagatgg
ctcttacttc gtctacagca agctcaatgt gcagaagagc 1320aactgggagg
caggaaatac tttcacctgc tctgtgttac atgagggcct gcacaaccac
1380catactgaga agagcctctc ccactctcct ggtaaatga
141920723DNAArtificialChimeric sequence 20gaattcgcca ccatgagtgt
gcccactcag gtcctggggt tgctgctgct gtggcttaca 60ggtgccagat gtgacatcca
gatgactcag tctccagcct ccctatctgc atctgtggga 120gaaactgtca
ccatcacatg tcgagcaagt gagaatattt acagttattt agcatggtat
180cagcagaaac agggaaaatc tcctcagttc ttggtctata atgcaaaaac
cttagcagaa 240ggtgtgccat caaggttcag tggcagtgga tcaggcacac
agttttctct gaagatcaac 300agcctgcagc ctgaagattt tgggagttat
tactgtcaac atcactatgg tactcctcgg 360acgttcggtg gaggcaccaa
gctggaaatc aaacggactg tggcggcgcc atctgtcttc 420atcttcccgc
catctgatga gcagttgaaa tctggtaccg ctagcgttgt gtgcctgctg
480aataacttct atcccagaga ggccaaagta cagtggaagg tggataacgc
cctccaatcg 540ggtaactccc aggagagtgt cacagagcag gacagcaagg
acagcaccta cagcctcagc 600agcaccctga cgctgagcaa agcagactac
gagaaacaca aagtctacgc ctgcgaagtc 660acccatcagg gcctgagctc
gcccgtcaca aagagcttca acaggggaga gtgttaggga 720tcc
723211434DNAArtificialChimeric sequence 21gaattcgcca ccatgggatg
gagctatatc atcctcttct tgttagcaac agctacatgt 60gtccactccc aggtccaact
gcagcagcct ggggctgagc tggtgaggcc tggggcttca 120gtgaagctgt
cctgcaaggc ttctggctac acgttcacca gctactggat gaactgggtt
180aagcagaggc ctgagcaagg ccttcagtgg attggaagga ttgatcctta
cgatagtgaa 240actcactaca gtcaaaagtt caaggacaag gccatattga
ctgtagacaa atcctccagc 300acagcctaca tgcgactcag cagcctgaca
tctgaggact ctgcggtcta ttactgtgca 360agagggggct atgatttcga
cgtaggaact ctctactggt tcttcgatgt ctggggcgca 420gggaccacgg
tcaccgtctc ctcagctagc accaagggcc catccgtctt ccccctggcg
480ccctgctcca ggagcacctc cgagagcaca gccgccctgg gctgcctggt
caaggactac 540ttccccgaac cggtgacggt gtcgtggaac tcaggcgccc
tgaccagcgg cgtgcacacc 600ttcccggctg tcctacagtc ctcaggactc
tactccctca gcagcgtggt gaccgtgccc 660tccagcagct tgggcacgaa
gacctacacc tgcaacgtag atcacaagcc cagcaacacc 720aaggtggaca
agagagttga gtccaaatat ggtcccccat gcccaccatg cccagcacct
780gagttcctgg ggggaccatc agtcttcctg ttccccccaa aacccaagga
cactctcatg 840atctcccgga cccctgaggt cacgtgcgtg gtggtggacg
tgagccagga agaccccgag 900gtccagttca actggtacgt ggatggcgtg
gaggtgcata atgccaagac aaagccgcgg 960gaggagcagt tcaacagcac
gtaccgtgtg gtcagcgtcc tcaccgtcct gcaccaggac 1020tggctgaacg
gcaaggagta caagtgcaag gtctccaaca aaggcctccc gtcctccatc
1080gagaaaacca tctccaaagc caaagggcag ccccgagagc cacaggtgta
caccctgccc 1140ccatcccagg aggagatgac caagaaccag gtcagcctga
cctgcctggt caaaggcttc 1200taccccagcg acatcgccgt ggagtgggag
agcaatgggc agccggagaa caactacaag 1260accacgcctc ccgtgctgga
ctccgacggc tccttcttcc tctacagcag gctaaccgtg 1320gacaagagca
ggtggcagga ggggaatgtc ttctcatgct ccgtgatgca tgaggctctg
1380cacaaccact acacacagaa gagcctctcc ctgtctctgg gtaaatgagg atcc
143422723DNAArtificialHumanized sequence 22gaattcgcca ccatggacat
gagggtcccc gctcagctcc tggggctcct gctactctgg 60ctccgaggtg ccagatgtga
catccagatg acccagtctc catcctccct gtctgcatct 120gtaggagaca
gagtcaccat cacttgccga gcaagtgaga atatttacag ttatttagca
180tggtatcagc agaaaccagg gaaagcccct aagctcctga tctataatgc
aaaaacctta 240gcagaagggg tcccatcaag gttcagtggc agtggatctg
ggacagattt cactctcacc 300atcagcagtc tgcaacctga agattttgca
acttactact gtcaacatca ctatggtact 360cctcggacgt tcggcggagg
gaccaaggtg gagatcaaac ggactgtggc ggcgccatct 420gtcttcatct
tcccgccatc tgatgagcag ttgaaatctg gtaccgctag cgttgtgtgc
480ctgctgaata acttctatcc cagagaggcc aaagtacagt ggaaggtgga
taacgccctc 540caatcgggta actcccagga gagtgtcaca gagcaggaca
gcaaggacag cacctacagc 600ctcagcagca ccctgacgct gagcaaagca
gactacgaga aacacaaagt ctacgcctgc 660gaagtcaccc atcagggcct
gagctcgccc gtcacaaaga gcttcaacag gggagagtgt 720tag
723231428DNAArtificialHumanized sequence 23gaattcgcca ccatggactg
gacctggagc atccttttct tggtggcagc agcaacaggt 60gcccactccc aggttcagct
ggtgcagtct ggagctgagg tgaagaagcc tggggcctca 120gtgaaggtct
cctgcaaggc ttctggttac acctttacca gctactggat gaactgggtg
180cgacaggccc ctggacaagg gcttgagtgg atgggaagga ttgatcctta
cgatagtgaa 240actcactatg cacagaagct ccagggcaga gtcaccatga
ccacagacac atccacgagc 300acagcctaca tggagctgag gagcctgaga
tctgacgaca cggccgtgta ttactgtgcg 360agagggggct atgatttcga
cgtaggaact ctctactggt tcttcgatgt ctggggccaa 420gggacaacgg
tcaccgtctc ttcagctagc accaagggcc catccgtctt ccccctggcg
480ccctgctcca ggagcacctc cgagagcaca gccgccctgg gctgcctggt
caaggactac 540ttccccgaac cggtgacggt gtcgtggaac tcaggcgccc
tgaccagcgg cgtgcacacc 600ttcccggctg tcctacagtc ctcaggactc
tactccctca gcagcgtggt gaccgtgccc 660tccagcagct tgggcacgaa
gacctacacc tgcaacgtag atcacaagcc cagcaacacc 720aaggtggaca
agagagttga gtccaaatat ggtcccccat gcccaccatg cccagcacct
780gagttcctgg ggggaccatc agtcttcctg ttccccccaa aacccaagga
cactctcatg 840atctcccgga cccctgaggt cacgtgcgtg gtggtggacg
tgagccagga agaccccgag 900gtccagttca actggtacgt ggatggcgtg
gaggtgcata atgccaagac aaagccgcgg 960gaggagcagt tcaacagcac
gtaccgtgtg gtcagcgtcc tcaccgtcct gcaccaggac 1020tggctgaacg
gcaaggagta caagtgcaag gtctccaaca aaggcctccc gtcctccatc
1080gagaaaacca tctccaaagc caaagggcag ccccgagagc cacaggtgta
caccctgccc 1140ccatcccagg aggagatgac caagaaccag gtcagcctga
cctgcctggt caaaggcttc 1200taccccagcg acatcgccgt ggagtgggag
agcaatgggc agccggagaa caactacaag 1260accacgcctc ccgtgctgga
ctccgacggc tccttcttcc tctacagcag gctaaccgtg 1320gacaagagca
ggtggcagga ggggaatgtc ttctcatgct ccgtgatgca tgaggctctg
1380cacaaccact acacacagaa gagcctctcc ctgtctctgg gtaaatga
142824124PRTArtificialHumanized sequence 24Gln Val Gln Leu Val Gln
Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys Val Ser
Cys Lys Ala Ser Gly Tyr Thr Phe Thr Ser Tyr 20 25 30Trp Met Asn Trp
Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Met 35 40 45Gly Arg Ile
Asp Pro Tyr Asp Ser Glu Thr His Tyr Ser Gln Lys Phe 50 55 60Lys Asp
Arg Val Thr Met Thr Thr Asp Thr Ser Thr Ser Thr Ala Tyr65 70 75
80Met Glu Leu Arg Ser Leu Arg Ser Asp Asp Thr Ala Val Tyr Tyr Cys
85 90 95Ala Arg Gly Gly Tyr Asp Phe Asp Val Gly Thr Leu Tyr Trp Phe
Phe 100 105 110Asp Val Trp Gly Gln Gly Thr Thr Val Thr Val Ser 115
12025124PRTArtificialHumanized sequence 25Gln Val Gln Leu Val Gln
Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys Val Ser
Cys Lys Ala Ser Gly Tyr Thr Phe Thr Ser Tyr 20 25 30Trp Met Asn Trp
Val Lys Gln Ala Pro Gly Gln Gly Leu Gln Trp Ile 35 40 45Gly Arg Ile
Asp Pro Tyr Asp Ser Glu Thr His Tyr Ser Gln Lys Phe 50 55 60Lys Asp
Lys Ala Thr Met Thr Thr Asp Thr Ser Thr Ser Thr Ala Tyr65 70 75
80Met Glu Leu Arg Ser Leu Arg Ser Asp Asp Thr Ala Val Tyr Tyr Cys
85 90 95Ala Arg Gly Gly Tyr Asp Phe Asp Val Gly Thr Leu Tyr Trp Phe
Phe 100 105 110Asp Val Trp Gly Gln Gly Thr Thr Val Thr Val Ser 115
12026124PRTArtificialHumanized sequence 26Glu Val Gln Leu Val Gln
Ser Gly Ala Glu Val Lys Lys Pro Gly Glu1 5 10 15Ser Leu Arg Ile Ser
Cys Lys Gly Ser Gly Tyr Ser Phe Thr Ser Tyr 20 25 30Trp Met Asn Trp
Val Arg Gln Met Pro Gly Lys Gly Leu Glu Trp Met 35 40 45Gly Arg Ile
Asp Pro Tyr Asp Ser Glu Thr His Tyr Ser Pro Ser Phe 50 55 60Gln Gly
His Val Thr Ile Ser Ala Asp Lys Ser Ile Ser Thr Ala Tyr65 70 75
80Leu Gln Trp Ser Ser Leu Lys Ala Ser Asp Thr Ala Met Tyr Tyr Cys
85 90 95Ala Arg Gly Gly Tyr Asp Phe Asp Val Gly Thr Leu Tyr Trp Phe
Phe 100 105 110Asp Val Trp Gly Gln Gly Thr Thr Val Thr Val Ser 115
12027124PRTArtificialHumanized sequence 27Glu Val Gln Leu Val Gln
Ser Gly Ala Glu Val Lys Lys Pro Gly Glu1 5 10 15Ser Leu Lys Ile Ser
Cys Lys Gly Ser Gly Tyr Ser Phe Thr Ser Tyr 20 25 30Trp Met Asn Trp
Val Arg Gln Met Pro Gly Lys Gly Leu Glu Trp Met 35 40 45Gly Arg Ile
Asp Pro Tyr Asp Ser Glu Thr His Tyr Ser Pro Ser Phe 50 55 60Gln Gly
Gln Val Thr Ile Ser Ala Asp Lys Ser Ile Ser Thr Ala Tyr65 70 75
80Leu Gln Trp Ser Ser Leu Lys Ala Ser Asp Thr Ala Met Tyr Tyr Cys
85 90 95Ala Arg Gly Gly Tyr Asp Phe Asp Val Gly Thr Leu Tyr Trp Phe
Phe 100 105 110Asp Val Trp Gly Gln Gly Thr Thr Val Thr Val Ser 115
12028124PRTArtificialHumanized sequence 28Glu Val Gln Leu Val Gln
Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10 15Thr Val Lys Ile Ser
Cys Lys Val Ser Gly Tyr Thr Phe Thr Ser Tyr 20 25 30Trp Met Asn Trp
Val Gln Gln Ala Pro Gly Lys Gly Leu Glu Trp Met 35 40 45Gly Arg Ile
Asp Pro Tyr Asp Ser Glu Thr His Tyr Ala Glu Lys Phe 50 55 60Gln Gly
Arg Val Thr Ile Thr Ala Asp Thr Ser Thr Asp Thr Ala Tyr65 70 75
80Met Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr Cys
85 90 95Ala Thr Gly Gly Tyr Asp Phe Asp Val Gly Thr Leu Tyr Trp Phe
Phe 100 105 110Asp Val Trp Gly Gln Gly Thr Thr Val Thr Val Ser 115
12029124PRTArtificialHumanized sequence 29Gln Val Gln Leu Val Gln
Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val Lys Val Ser
Cys Lys Ala Ser Gly Tyr Thr Phe Thr Ser Tyr 20 25 30Trp Met Asn Trp
Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Met 35 40 45Gly Arg Ile
Asp Pro Tyr Asp Ser Glu Thr His Tyr Ala Gln Lys Phe 50 55 60Gln Gly
Arg Val Thr Met Thr Arg Asp Thr Ser Thr Ser Thr Val Tyr65 70 75
80Met Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr Cys
85 90 95Ala Arg Gly Gly Tyr Asp Phe Asp Val Gly Thr Leu Tyr Trp Phe
Phe 100 105 110Asp Val Trp Gly Gln Gly Thr Thr Val Thr Val Ser 115
120
References
D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

D00013

D00014

D00015

D00016

D00017

D00018

D00019

D00020

D00021

D00022

D00023

D00024

D00025


S00001
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.