Systems And Methods For Simulation Of Hemodialysis Access And Optimization
SANKARAN; Sethuraman ; et al.
U.S. patent application number 16/397218 was filed with the patent office on 2019-08-15 for systems and methods for simulation of hemodialysis access and optimization. This patent application is currently assigned to HeartFlow, Inc.. The applicant listed for this patent is HeartFlow, Inc.. Invention is credited to Leo Grady, Sethuraman SANKARAN, Charles A. Taylor, Christopher K. Zarins.
| Application Number | 20190247124 16/397218 |
| Document ID | / |
| Family ID | 55852965 |
| Filed Date | 2019-08-15 |





| United States Patent Application | 20190247124 |
| Kind Code | A1 |
| SANKARAN; Sethuraman ; et al. | August 15, 2019 |
SYSTEMS AND METHODS FOR SIMULATION OF HEMODIALYSIS ACCESS AND OPTIMIZATION
Abstract
Systems and methods are disclosed for simulating or optimizing hemodialysis access. One method includes receiving a patient-specific anatomic model of a patient's vasculature; computing a pre-treatment hemodynamic characteristic of a pre-treatment geometry of a portion of the anatomic model; simulating a post-treatment geometry of a vascular access in the portion of the anatomic model; computing a post-treatment hemodynamic characteristic of the post-treatment geometry of the portion of the anatomic model having the vascular access; and generating a representation of the pre-treatment hemodynamic characteristic or the post-treatment hemodynamic characteristic.
| Inventors: | SANKARAN; Sethuraman; (Palo Alto, CA) ; Grady; Leo; (Millbrae, CA) ; Taylor; Charles A.; (Atherton, CA) ; Zarins; Christopher K.; (Menlo Park, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | HeartFlow, Inc. |
||||||||||
| Family ID: | 55852965 | ||||||||||
| Appl. No.: | 16/397218 | ||||||||||
| Filed: | April 29, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15054622 | Feb 26, 2016 | 10314654 | ||
| 16397218 | ||||
| 14595503 | Jan 13, 2015 | 9336354 | ||
| 15054622 | ||||
| 62074698 | Nov 4, 2014 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | G16H 50/50 20180101; A61B 2034/105 20160201; A61B 2034/104 20160201; G16H 50/30 20180101; A61M 1/3655 20130101; A61M 1/3661 20140204; A61M 1/3653 20130101; A61M 1/3659 20140204; A61M 1/00 20130101; A61B 34/10 20160201 |
| International Class: | A61B 34/10 20060101 A61B034/10; G16H 50/50 20060101 G16H050/50; A61M 1/36 20060101 A61M001/36; A61M 1/00 20060101 A61M001/00 |
Claims
1-20. (canceled)
21. A computer-implemented method of simulating or optimizing hemodialysis access, the method comprising: receiving a patient-specific anatomic model of a patient's vasculature; modifying the geometry of the received patient-specific anatomic model such that the geometry of the received patient-specific anatomic model defines a post-treatment geometry of a vascular access; determining a computational model of a hemodynamic characteristic of the modified anatomic model; computing a post-treatment hemodynamic characteristic using the determined computational model; and outputting or generating a representation of the post-treatment hemodynamic characteristic.
22. The computer-implemented method of claim 21, wherein the hemodynamic characteristic includes blood pressure, blood velocity, or cardiac output.
23. The computer-implemented method of claim 21, further comprising: receiving or determining a geometry of a planned treatment defining the vascular access; and modifying the received patient-specific anatomic model such that the post-treatment geometry is based on the geometry of the planned treatment.
24. The computer-implemented method of claim 23, wherein the planned treatment includes a graft.
25. The computer-implemented method of claim 21, further comprising: receiving or measuring one or more candidate locations of the received patient-specific anatomic model, wherein the portion of the received patient-specific anatomic model is selected from one of the one or more candidate locations.
26. The computer-implemented method of claim 21, further comprising: defining a cost function for optimizing the vascular access; and solving the cost function using the post-treatment hemodynamic characteristic.
27. The computer-implemented method of claim 21, further comprising: receiving one or more infeasible surgical geometries for the hemodialysis access, and computing the post-treatment hemodynamic characteristic using the one or more infeasible geometries as constraints for the simulating of the post-treatment geometry.
28. The computer-implemented method of claim 21, further comprising: selecting a treatment or treatment location of the vascular access based on the post-treatment hemodynamic characteristic.
29. A system for simulating or optimizing hemodialysis access, the system comprising: a data storage device storing instructions for simulating or optimizing hemodialysis access; and a processor configured to execute the instructions to perform a method including: receiving a patient-specific anatomic model of a patient's vasculature; modifying the geometry of the received patient-specific anatomic model such that the geometry of the received patient-specific anatomic model defines a post-treatment geometry of a vascular access; determining a computational model of a hemodynamic characteristic of the modified anatomic model; computing a post-treatment hemodynamic characteristic using the determined computational model; and outputting or generating a representation of the post-treatment hemodynamic characteristic.
30. The system of claim 29, wherein the hemodynamic characteristic includes blood pressure, blood velocity, or cardiac output.
31. The system of claim 29, wherein the system is further configured for: receiving or determining a geometry of a planned treatment defining the vascular access; and modifying the received patient-specific anatomic model such that the post-treatment geometry is based on the geometry of the planned treatment.
32. The system of claim 31, wherein the planned treatment includes a graft.
33. The system of claim 29, wherein the system is further configured for: receiving or measuring one or more candidate locations of the received patient-specific anatomic model, wherein the portion of the received patient-specific anatomic model is selected from one of the one or more candidate locations.
34. The system of claim 29, wherein the system is further configured for: defining a cost function for optimizing the vascular access; and solving the cost function using the post-treatment hemodynamic characteristic.
35. The system of claim 29, wherein the system is further configured for: receiving one or more infeasible surgical geometries for the hemodialysis access, and computing the post-treatment hemodynamic characteristic using the one or more infeasible geometries as constraints for the simulating of the post-treatment geometry.
36. The system of claim 29, wherein the system is further configured for: selecting a treatment or treatment location of the vascular access based on the post-treatment hemodynamic characteristic.
37. A non-transitory computer readable medium for use on a computer system containing computer-executable programming instructions for performing a method of simulating or optimizing hemodialysis access, the method comprising: receiving a patient-specific anatomic model of a patient's vasculature; modifying the geometry of the received patient-specific anatomic model such that the geometry of the received patient-specific anatomic model defines a post-treatment geometry of a vascular access; determining a computational model of a hemodynamic characteristic of the modified anatomic model; computing a post-treatment hemodynamic characteristic using the determined computational model; and outputting or generating a representation of the post-treatment hemodynamic characteristic.
38. The non-transitory computer readable medium of claim 37, wherein the hemodynamic characteristic includes blood pressure, blood velocity, or cardiac output.
39. The non-transitory computer readable medium of claim 37, the method further comprising: receiving or determining a geometry of a planned treatment defining the vascular access; and modifying the received patient-specific anatomic model such that the post-treatment geometry is based on the geometry of the planned treatment.
40. The non-transitory computer readable medium of claim 39, wherein the planned treatment includes a graft.
Description
RELATED APPLICATION(S)
[0001] This application claims priority to U.S. Provisional Application No. 62/074,698 filed Nov. 4, 2014, the entire disclosure of which is hereby incorporated herein by reference in its entirety.
FIELD OF THE DISCLOSURE
[0002] Various embodiments of the present disclosure relate generally to disease assessment, treatment planning, and related methods. More specifically, particular embodiments of the present disclosure relate to systems and methods for simulating and optimizing hemodialysis access.
BACKGROUND
[0003] Hemodialysis is a process in which an external machine is used to filter blood to remove excess salt and harmful wastes. For example, blood may be sent to a hemodialysis machine and back to the patient's circulation. The blood may enter and exit a body via a vascular access. For instance, a vascular access for the blood may be created by introducing cannulas into a patient's vein. Blood may then be sent to a hemodialysis machine and back to the patient's circulation via the cannulas. To enable ease of cannulation and ensure availability of many candidate access sites, a larger vein may be used. This may be possible by shunting arterial blood flow through veins. Two kinds of vascular access procedures include--arteriovenous fistula (AVF) (in which an artery and a vein may be directly connected) and arteriovenous graft (AVG) (in which a synthetic graft may be attached between an artery and a vein). Demand for cardiac output may change from before and after treatment (e.g., pre- and post-shunting of blood flow). For example, the arteriovenous connection may increase blood pressure and blood flow in the veins. The veins may slowly adapt to this shunting by enlarging in diameter and increasing in thickness. This adaptation process might take anywhere from one month to a year. Once adapted, many different candidate sites may be available for repeated cannulation and hemodialysis.
[0004] However, the change in cardiac workload for the heart post-treatment may create a health risk. For instance, shunting the blood flow from an artery to a vein may reduce overall system resistance, thus changing hemodynamics in a way that increases cardiac output. This increased demand for cardiac output may result in a larger workload for the heart, and may be linked to an increased risk of congestive heart failure. Further, treatment may change vessel geometry, thus altering regions in vessels that may be prone to thrombosis. An optimal AVG may minimize regions of disturbed hemodynamics and, consequently, minimize regions prone to thrombosis.
[0005] Thus, a desire exists to ensure that there is enough blood flow to allow successful dialysis and maintain sufficient perfusion to extremities, and at the same time, ensure that change to hemodynamics is minimal so that cardiac demand may not increase to a point that endangers a patient. Furthermore, a desire exists to improve treatment planning by optimizing vascular access graft locations and/or vascular access graft types. The present disclosure is directed to improving treatment planning by predicting changes in hemodynamics that may result from vascular access procedures.
[0006] The foregoing general description and the following detailed description are exemplary and explanatory only and are not restrictive of the disclosure.
SUMMARY
[0007] According to certain aspects of the present disclosure, systems and methods are disclosed for simulating and optimizing hemodialysis access.
[0008] One method includes: receiving a patient-specific anatomic model of a patient's vasculature; computing a pre-treatment hemodynamic characteristic of a pre-treatment geometry of a portion of the anatomic model; simulating a post-treatment geometry of a vascular access in the portion of the anatomic model; computing a post-treatment hemodynamic characteristic of the post-treatment geometry of the portion of the anatomic model having the vascular access; and generating a representation of the pre-treatment hemodynamic characteristic or the post-treatment hemodynamic characteristic.
[0009] In accordance with another embodiment, a system for simulating or optimizing hemodialysis access: a data storage device storing instructions for simulating or optimizing hemodialysis access; and a processor configured for: receiving a patient-specific anatomic model of a patient's vasculature; computing a pre-treatment hemodynamic characteristic of a pre-treatment geometry of a portion of the anatomic model; simulating a post-treatment geometry of a vascular access in the portion of the anatomic model; computing a post-treatment hemodynamic characteristic of the post-treatment geometry of the portion of the anatomic model having the vascular access; and generating a representation of the pre-treatment hemodynamic characteristic or the post-treatment hemodynamic characteristic.
[0010] In accordance with another embodiment, a non-transitory computer readable medium for use on a computer system containing computer-executable programming instructions for performing a method of simulating or optimizing hemodialysis access, the method comprising: receiving a patient-specific anatomic model of a patient's vasculature; computing a pre-treatment hemodynamic characteristic of a pre-treatment geometry of a portion of the anatomic model; simulating a post-treatment geometry of a vascular access in the portion of the anatomic model; computing a post-treatment hemodynamic characteristic of the post-treatment geometry of the portion of the anatomic model having the vascular access; and generating a representation of the pre-treatment hemodynamic characteristic or the post-treatment hemodynamic characteristic.
[0011] Additional objects and advantages of the disclosed embodiments will be set forth in part in the description that follows, and in part will be apparent from the description, or may be learned by practice of the disclosed embodiments. The objects and advantages of the disclosed embodiments will be realized and attained by means of the elements and combinations particularly pointed out in the appended claims.
[0012] It is to be understood that both the foregoing general description and the following detailed description are exemplary and explanatory only and are not restrictive of the disclosed embodiments, as claimed.
BRIEF DESCRIPTION OF THE DRAWINGS
[0013] The accompanying drawings, which are incorporated in and constitute a part of this specification, illustrate various exemplary embodiments and together with the description, serve to explain the principles of the disclosed embodiments.
[0014] FIG. 1 is a block diagram of an exemplary system and network for simulating or optimizing hemodialysis access, according to an exemplary embodiment of the present disclosure.
[0015] FIG. 2A is a block diagram of an exemplary method of simulating hemodialysis access, according to an exemplary embodiment of the present disclosure.
[0016] FIG. 2B is a block diagram of an exemplary method of optimizing hemodialysis access, according to an exemplary embodiment of the present disclosure.
[0017] FIG. 3A is a block diagram of an exemplary method of performing predictive modeling and simulation of an AVG, a specific embodiment of simulating hemodialysis access according to an exemplary embodiment of the present disclosure.
[0018] FIG. 3B is a block diagram of an exemplary method of optimizing an AVG, according to an exemplary embodiment of the present disclosure.
DESCRIPTION OF THE EMBODIMENTS
[0019] Reference will now be made in detail to the exemplary embodiments of the disclosure, examples of which are illustrated in the accompanying drawings. Wherever possible, the same reference numbers will be used throughout the drawings to refer to the same or like parts.
[0020] Shunting of blood flow from an artery to a vein via AVGs or AVFs may reduce overall systemic resistance, thus causing increased cardiac output. This increased demand for cardiac output may result in a larger workload for the heart, which may be linked to an increased risk of congestive heart failure. Thus, a goal of vascular access may include increasing blood flow to the veins, while maintaining sufficient perfusion to extremities and ensuring that cardiac demand may not increase to a dangerous level. Further, an optimal AVG may minimize regions of disturbed hemodynamics so that regions prone to thrombosis may be minimized as well.
[0021] The present disclosure includes systems and methods for simulating and optimizing hemodialysis access in order to better provide vascular access that improves blood flow but does not endanger the patient. For example, the simulations and optimizations include evaluating or comparing various location(s) for the vascular access, as well as graft or fistula geometries. Each "treatment" may include vascular access at one location, multiple locations, a single graft or fistula, multiple grafts or fistulas, or a combination thereof. One embodiment may include evaluating various treatments, e.g., by simulating and comparing pre- or post-treatment blood flow for different treatments.
[0022] One embodiment may further include performing optimization for the hemodialysis access. For example, the optimization may include defining a cost function for determining an optimal hemodialysis characteristic (e.g., blood flow). The cost function may incorporate pre- and post-treatment calculations from the simulations of blood flow for different treatments. In one case, selecting or finding an optimal hemodialysis access treatment may include performing optimization over all possible vascular grafts (e.g., including graft types and/or graft locations in the patient vasculature) to minimize the cost function. In another case, the optimization may be performed for a subset of available vascular access grafts to minimize a cost function.
[0023] For example, the increase in cardiac demand due to an AVG or an AVF may be modeled by performing a pre-dialysis blood-flow simulation with the original geometry, e.g., by solving 3D Navier-Stokes equations, reduced order models, or using machine learning methods. Resistance boundary condition(s) relating blood pressure to flow rate may be provided at the model boundaries. At the aortic inlet surface, cardiac flow and pressure may be coupled to the systemic circulation using a ventricular elastance function. The ventricular elastance function may reflect the relationship between cardiac flow, pressure, and systemic circulation since pressure may be based on ventricular volume which, in turn, may be related to a flow-rate using an elastance function.
[0024] Hemodynamics post-treatment may be computed by solving the same equations using a modified geometry reflecting the procedure based on geometric variables that determine the AVG or the AVF. The resulting aortic pressure and flow rate, as well as the area enclosed in the left ventricle PV loop, may be used to estimate the changes in cardiac workload, post-treatment. The post-treatment cardiac demand and other hemodynamic quantities of interest may be reported to a user (e.g., a physician).
[0025] Hemodynamic quantities of interest may include blood flow, e.g., volume of blood or velocity of blood through a patient's vasculature or portion of a patient's vasculature. Other hemodynamic characteristics that may be quantities of interest may include shear stress and/or particle residence time through an AVG. Shear stress and particle residence time through a particular AVG may be observed to provide sufficient blood flow to allow successful dialysis, while reducing the risk of thrombosis in the AVG over time when it is not being used for dialysis. Yet another example of hemodynamic characteristics may include distal perfusion pressure and flow in arteries past a fistula. For example, evaluating distal perfusion and flow in vessel locations past an AVF may help to identify whether a graft may "steal" blood from the extremity causing ischemia/symptoms or gangrene in fingers.
[0026] Such methods may help in treatment planning and predicting changes in hemodynamics that result from vascular access procedures.
[0027] Referring now to the figures, FIG. 1 depicts a block diagram of an exemplary system 100 and network for simulating or optimizing hemodialysis access, according to an exemplary embodiment. Specifically, FIG. 1 depicts a plurality of physicians 102 and third party providers 104, any of whom may be connected to an electronic network 101, such as the Internet, through one or more computers, servers, and/or handheld mobile devices. Physicians 102 and/or third party providers 104 may create or otherwise obtain images of one or more patients' anatomy. The physicians 102 and/or third party providers 104 may also obtain any combination of patient-specific information, such as age, medical history, blood pressure, blood viscosity, patient activity or exercise level, etc. Physicians 102 and/or third party providers 104 may transmit the anatomical images and/or patient-specific information to server systems 106 over the electronic network 101. Server systems 106 may include storage devices for storing images and data received from physicians 102 and/or third party providers 104. Server systems 106 may also include processing devices for processing images and data stored in the storage devices. For the present disclosure, "patient" may refer to any individual of interest.
[0028] FIGS. 2A and 2B depict flowcharts of a general embodiment for simulating hemodialysis access and optimizing hemodialysis access, respectively. FIG. 3A depicts a flowchart of a specific embodiment for simulating hemodialysis access. FIG. 3B depicts a flowchart of a specific embodiment for optimizing hemodialysis access.
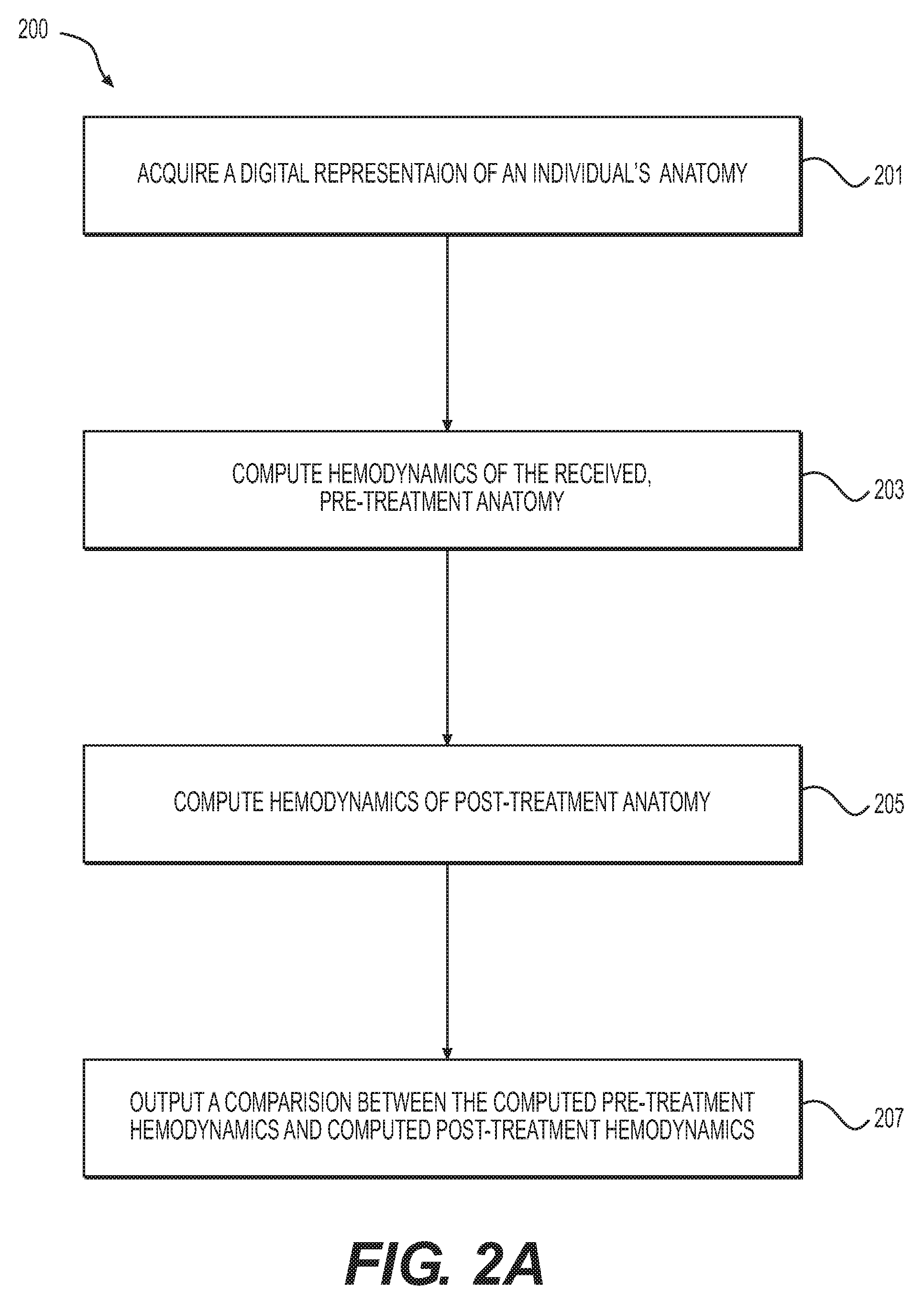
[0029] FIG. 2A is a block diagram of an exemplary method 200 of simulating hemodialysis access, according to an exemplary embodiment. The method of FIG. 2A may be performed by server systems 106, based on information, images, and data received from physicians 102 and/or third party providers 104 over electronic network 101.
[0030] In one embodiment, step 201 may include acquiring a digital representation of a system of interest (e.g., of an individual's anatomy) and reconstructing a computational model (e.g., representing the functioning of the system of interest). For example, the digital representation may include an artery and a vein that may be anastomosed or shunted, as well as all vessels branching off the arteriovenous system encompassing arteries and veins of a system of interest. In one embodiment, the vessels may be traced back to the aorta. A three dimensional model of the arteries may be reconstructed, e.g., by computing centerlines through the model and segmenting the arteries and veins across centerlines.
[0031] In one embodiment, step 201 may further include acquiring candidate parameters for anastomoses (e.g., location of an anastomosis). Step 201 may also include acquiring infeasible surgeries or any extraneous constraints that may make certain surgical configurations infeasible.
[0032] In one embodiment, step 203 may include computing hemodynamics of a pre-treatment AV system geometry. Blood flow characteristics in the reconstructed geometry may be computed by solving the Navier-Stokes equations. For example, the computation may include constructing a finite element mesh over the reconstructed geometry (e.g., from step 201) and specifying appropriate boundary conditions that relate blood pressure to flow rate at outlet surfaces. The boundary conditions may be calculated based on local tissue perfusion demand. Other boundary conditions, including capacitance of micro-vessels, may also be provided. At the aortic inlet, a time-varying elastance function of the left ventricle may be given as input. Otherwise, blood pressure or velocity may be provided as input at inlet surfaces.
[0033] Alternatively or in addition, step 203 may include using a reduced order model to solve for the hemodynamics and related quantities of interest. The reduced order model may involve combining the Poiseulle's law with non-linear stenosis pressure loss models, as well as Bernoulli's equation to account for kinetic energy of blood. Alternately or in addition, step 203 may involve solving the one dimensional wave equation.
[0034] Alternatively or in addition, step 203 may include using a machine learning approach to solve for the hemodynamics (e.g., as described in U.S. Nonprovisional application Ser. No. 13/895,893 filed May 16, 2013, the entire disclosure of which is hereby incorporated by reference in its entirety). Features of the computational model that affect hemodynamics may be calculated. In some cases, a regressor computed using a database of 3D simulations may be used to map the inputs to hemodynamic quantities of interest (e.g., as described in the application).
[0035] In one embodiment, step 205 may include simulating and computing hemodynamics for a post-treatment anatomy geometry. Post-treatment patient geometry may be created by altering the segmentation at the candidate anastomoses sites to mimic the planned treatment geometry. Any of the three broad classes of methods described in step 203 may be used to calculate hemodynamic quantities of interest post-treatment.
[0036] In one embodiment, step 207 may include outputting the results of the simulation. For example, the difference in hemodynamics pre-treatment versus post-treatment may be displayed or reported to a user (e.g., a physician). In one embodiment, step 207 may further include creating a rendering for the comparison, including graphical, pictorial, and/or interactive features.
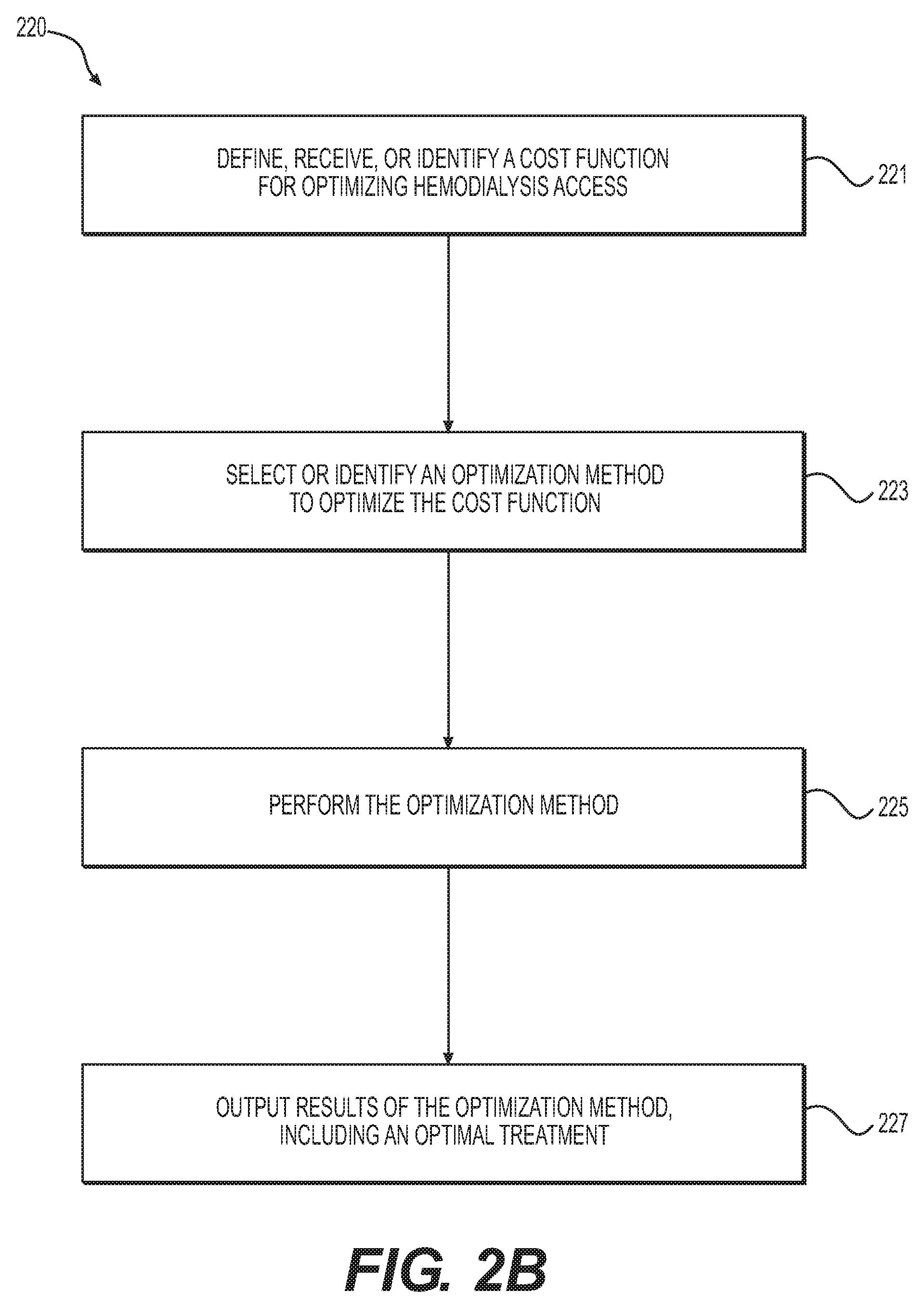
[0037] FIG. 2B is a block diagram of an exemplary method 220 of optimizing hemodialysis access, according to an exemplary embodiment. The method of FIG. 2B may be performed by server systems 106, based on information, images, and data received from physicians 102 and/or third party providers 104 over electronic network 101.
[0038] Simulations may offer a viable alternative to calculating optimal treatment geometry, including one or more AVGs and/or AVFs at one or more locations. Optimal parameters may be computed by minimizing a cost function. In one embodiment, step 221 may include defining, receiving, or identifying a cost function for optimizing hemodialysis access. For example, a general cost function may be defined as:
C=.alpha..sub.1C.sub.output(pre,post).alpha..sub.2C.sub.flow+.alpha..sub- .3C.sub.hemo+.alpha..sub.4C.sub.clot+.alpha..sub.5C.sub.other
C.sub.output(pre, post) may be a function of the cardiac output of the heart, C.sub.flow may be a function of blood flow through the veins and flow through arteries distal to shunt location, C.sub.hemo may be a function of risk of clot formation near shunt location, and C.sub.other may be a function of any other problem-specific cost function that may depend on the hemodynamics. More specifically, the cost function may be defined as:
C=.alpha..sub.1(E.sub.post-E.sub.pre)-.alpha..sub.2(Q.sub.dialyzer).alph- a..sub.3(H.sub.post-H.sub.pre)+.alpha..sub.4(T.sub.post-T.sub.pre)+.SIGMA.- .sub.i=1.sup.N.beta..sub.if.sub.i(P,v)
[0039] .alpha..sub.1, .alpha..sub.2, .alpha..sub.3, and .alpha..sub.4 may include weights for different cost functions. .alpha..sub.2, .alpha..sub.3, and .alpha..sub.4 may have different units and a user may choose to pick values based on patient-history (e.g., for a patient who has had previous coronary artery bypass graft surgery, .alpha..sub.2, .alpha..sub.3 and .beta..sub.i may be taken to be zero so that only the first term may be used in the optimization). E in the first term may include the difference between cardiac outputs (E.sub.post-E.sub.pre), which may be defined as:
E=.intg.PdV
[0040] P and V may be defined as pressure and volume of left ventricle. The second term, Q.sub.dialyzer may include the flow through the dialyzer intended to be maximized. H in the third term (H.sub.post-H.sub.pre), may include terms which quantify disturbed hemodynamics. For instance, H may be an area of low wall shear stresses defined as:
H=.intg.I(.tau.<.tau..sub.c)dA
[0041] .tau. may be the local WSS and .tau..sub.c may be the critical cutoff, below which surfaces may be deemed disturbed.
[0042] Furthermore, thrombosis of a graft may contribute to AVG failure. In one embodiment, the cost function may include a term (e.g., "T") that indicates a propensity for thrombosis, e.g., by modeling blood rheology or clotting. Various coagulation models T based on hemodynamics and blood viscosity may be used to account for one or more characteristics of blood that may promote or inhibit intravascular coagulation, for instance: blood composition, factors that influence activation or inhibition of platelets, factors that influence activation or inhibition of a clotting cascade (e.g., at any point in a multilayered cascade), anti-coagulant presence or usage versus no anti-coagulation presence or usage, viscosity of blood (since dialysis patients may be anemic), etc. Factors that influence activation or inhibition of platelets may include, for example, initiation of platelet aggregation inhibition. The coagulation models may further account for shear rate and/or chemical pathways, for instance, in modeling the blood flow of a patient using anticoagulants.
[0043] In one embodiment, T may be calculated by advecting particles using the blood velocity field calculated from the hemodynamics. For example, particles may be defined based on a platelet activation function, which, in turn, may be defined based on a velocity gradient (e.g., with a deformation tensor). For instance, an active platelet may be identified when an activation threshold is crossed. Calculating T may then involve tracing the evolution or activity of the active platelet over time. Another exemplary platelet activation model may include a power law model based on shear stresses, in which advecting particles may involve releasing simulated blood cells at the inlet of a problem geometry and propagating the cells based on the blood velocity field to trace the path of the simulated blood cells over a period of time. Such a power law model may be used to calculate the residence time of the blood cells, which quantifies the propensity of the blood cells to form clots. For instance, the higher the residence time, the higher the propensity for thrombus formation. A patient's blood viscosity may affect the velocity field and, consequently, the residence time.
[0044] Any other term encoding hemodynamic quantities of interest that may be added to the cost function, which may be represented by the term .SIGMA..sub.i=1.sup.N.beta..sub.if.sub.i(P,v). Non-linear combinations of the terms may also be used.
[0045] To find an optimal treatment, one embodiment of step 223 may include selecting or identifying an optimization method to find the parameters that minimize the cost function C. Any optimization method, including (i) derivative-free optimization techniques (e.g., pattern search, Nelder-Mead algorithm, etc.), (ii) gradient-based quasi-Newtonian algorithm (e.g., BFGS), or (iii) global optimization methods (e.g., evolutionary search algorithm) may be used to find the optima. Constraints may be incorporated by mapping parameters in the constraint region to a very high value (e.g., 10{circumflex over ( )}10). Step 225 may include performing the optimization method, and step 227 may include outputting the results of the optimization method and/or updating or changing the cost function to solve for a different optima.
[0046] FIG. 3A is a block diagram of an exemplary method 300 of performing predictive modeling and simulation of an AVG between the brachial artery and antecubital vein, which is one exemplary embodiment of simulating hemodialysis access. FIG. 3B is a block diagram of an exemplary method 320 of optimizing an AVG, according to an exemplary embodiment. The methods of FIGS. 3A and 3B may be performed by server systems 106, based on information, images, and data received from physicians 102 and/or third party providers 104 over electronic network 101. Exemplary methods 300 and 320 may apply to an AVG between any other artery and vein as well. Furthermore, exemplary methods 300 and 320 may be extended to an AVF where graft-specific parameters, e.g., radius, may be removed from the optimization parameter set.
[0047] In one embodiment, step 301 may include acquiring a digital representation of a system (e.g., a patient's vasculature or anatomy of interest) and reconstructing a computational model to represent a hemodynamic characteristic of the system. For example, the system and computational model may include a portion of a patient's aorta, brachial artery, antecubital vein, and (if available) other arteries, e.g., cephalic, subclavian, and radial artery. A three dimensional model of the brachial artery and antecubital vein, part of the aorta, and other visible arteries and veins may be reconstructed using a combination of automated algorithms and manual editing.
[0048] Step 301 may further include receiving or measuring available graft radii. Candidate sites or parameters for anastomoses in the artery and veins may also be measured or received as input, and infeasible surgical geometries may be used as constraints.
[0049] In one embodiment, step 303 may include computing hemodynamics in the model pre-AVG implantation. Blood flow simulations in the reconstructed geometry (e.g., from step 301) may be performed by constructing a finite element mesh of the computational model. Blood pressure and velocities may be calculated at the vertices of the finite element mesh, e.g., by solving the Navier-Stokes equations. At the inlet to the model, the aortic inlet flow and blood pressure may be coupled to a reduced-order heart model driven by a ventricular elastance function. At the outlets, resistance boundary conditions which relate blood pressure to flow-rate may be prescribed. These values may be obtained for different arteries by determining the ratio of population-averaged nominal pressure to nominal flow-rate. For instance, pressure and flow-rate for the radial artery may be .about.150 mm Hg and .about.600 ml/min, respectively. Hemodynamic quantities of interest may be calculated from blood pressure and velocity found using the Navier-Stokes equations.
[0050] In one embodiment, step 305 may include modeling a virtual AVG. For example, step 305 may include constructing a virtual model of the AVG by inserting a graft at candidate sites (e.g., sites provided in step 301). This may be performed by creating a cylindrical vessel of the radius (e.g., from step 301) and creating an anastomosis using the cylindrical vessel and a series of Boolean operations on the computational geometry constructed from step 301. Alternatively, if an implicit representation of the patient-specific geometry is available, an implicit model of a cylindrical graft may be used to model the geometric changes associated with inserting a graft. In one embodiment, this procedure may be performed twice, once at the brachial artery and once at the antecubital vein. A finite element mesh of the new geometry may be constructed.
[0051] In one embodiment, step 307 may include calculating hemodynamic quantities of interest post-implantation of an AVG. In one embodiment, step 309 may include reporting a comparison of the pre-AVG hemodynamic quantities of interest to modeled post-AVG hemodynamic quantities of interest. For example, step 309 may include reporting the comparison for one or more of the candidate AVG parameters that were provided as input in step 301.
[0052] Regarding FIG. 3B, method 320 shown in FIG. 3B may include optimizing the radius and location of an AVG. In one embodiment, step 321 of method 320 may include defining a cost function for optimizing AVG implantation. The cost function may include parameters that contribute to AVG optimization. For example, one parameter may include graft radius. Graft radius may affect shunting resistance of the AVG. A higher graft radius may result in larger blood flow being shunted through the AVG. Length, as well as curvature of vascular graft, may also be treated as a parameter. The curvature of the graft may be important since the graft may go down the forearm, take a 180.degree. turn, and return to the antecubital space. Further, the angle of the AVG may impact the local hemodynamics and may result in adverse hemodynamics, e.g., flow stasis, recirculation, etc. Location of anastomosis may be another parameter which impacts the outcome of treatment. The graft may take various different pathways, e.g., depending on the location of the arterial and venous anastomosis. A desired output of the model may include a choice of an optimal site for anastomosis. An exemplary cost function may be defined as
C = ( E post - E pre ) E pre - ( Q dialyzer ) Q max - ( Q distal ) Q max + ( H post - H pre ) H pre + ( T - T pre ) T pre ##EQU00001##
[0053] The first, second, and last terms of the above equation may be the same as defined in the general embodiment (and normalized by their nominal value). An AVG-specific term (e.g., Q.sub.distal) may be added to ensure that most of the flow is not shunted and that enough blood flow goes to the extremities. This may help prevent a condition where a patient loses sensation or has compromised circulation in extremities soon after the surgery. Specifically, Q.sub.distal may be the blood flow distal to the arterial anastomosis site.
[0054] In one embodiment, step 323 may include receiving, creating, or identifying a termination criterion. In one embodiment, step 325 may include using a derivative-free Nelder-Mead optimization method to compute parameters that may minimize the cost function. A random initial parameter set may be chosen. Subsequently, at each step in the Nelder-Mead algorithm, a new parameter may be identified by constructing simplexes using existing points and choosing an operation between reflection, reduction, contraction or reduction. In one embodiment, step 327 may include determining whether the computed parameters from step 325 meet a termination criterion and performing step 325 successively until the termination criterion is reached. In one embodiment, step 329 may include outputting the parameters computed where the termination criterion was met, as optimal therapy parameters, e.g., to an electronic storage medium and/or to a user interface.
[0055] Hemodialysis may present its own set of risks, including cardiovascular risks stemming from dialysis access. Various access treatments may change vessel geometry, altering hemodynamics and causing thrombosis or dangerous levels of cardiac demand. A desire exists to ensure that there is enough blood flow to allow successful dialysis while also ensuring that change to hemodynamics does not endanger a patient. The present disclosure thus provides systems and methods to improve treatment planning by predicting changes in hemodynamics that may result from vascular access procedures. The systems and methods include simulating access treatments and comparing results from the simulations to provide optimal treatments for a patient.
[0056] Other embodiments of the invention will be apparent to those skilled in the art from consideration of the specification and practice of the invention disclosed herein. It is intended that the specification and examples be considered as exemplary only, with a true scope and spirit of the invention being indicated by the following claims.
* * * * *
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.