Metal Electrodeposition Cathode Plate And Production Method Therefor
Watanabe; Hiroto ; et al.
U.S. patent application number 16/317141 was filed with the patent office on 2019-08-01 for metal electrodeposition cathode plate and production method therefor. This patent application is currently assigned to SUMITOMO METAL MINING CO., LTD.. The applicant listed for this patent is SUMITOMO METAL MINING CO., LTD.. Invention is credited to Hiroshi Kobayashi, Itsumi Matsuoka, Yusuke Senba, Hiroto Watanabe.
| Application Number | 20190233960 16/317141 |
| Document ID | / |
| Family ID | 60992989 |
| Filed Date | 2019-08-01 |




| United States Patent Application | 20190233960 |
| Kind Code | A1 |
| Watanabe; Hiroto ; et al. | August 1, 2019 |
METAL ELECTRODEPOSITION CATHODE PLATE AND PRODUCTION METHOD THEREFOR
Abstract
Provided are a metal electrodeposition cathode plate, the non-conductive film of which is not susceptible to failure and which can be used repeatedly, and a production method therefor. This cathode plate comprises a metal plate on which multiple disc-shaped protrusions are disposed, and a non-conductive film formed on the non-protrusion flat areas of the metal plate. The minimum film thickness Y of the non-conductive film at positions between the centers of adjacent protrusions is the same or greater than the height X of the protrusions. It is preferred that the height X of the protrusions is 50 .mu.m to 1000 .mu.m.
| Inventors: | Watanabe; Hiroto; (Niihama-shi, JP) ; Matsuoka; Itsumi; (Niihama-shi, JP) ; Senba; Yusuke; (Niihama-shi, JP) ; Kobayashi; Hiroshi; (Niihama-shi, JP) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | SUMITOMO METAL MINING CO.,
LTD. Tokyo JP |
||||||||||
| Family ID: | 60992989 | ||||||||||
| Appl. No.: | 16/317141 | ||||||||||
| Filed: | July 10, 2017 | ||||||||||
| PCT Filed: | July 10, 2017 | ||||||||||
| PCT NO: | PCT/JP2017/025093 | ||||||||||
| 371 Date: | January 11, 2019 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C25C 7/02 20130101; C25D 17/12 20130101; C25C 1/08 20130101; C25C 7/08 20130101 |
| International Class: | C25C 1/08 20060101 C25C001/08; C25C 7/02 20060101 C25C007/02; C25C 7/08 20060101 C25C007/08; C25D 17/12 20060101 C25D017/12 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Jul 21, 2016 | JP | 2016-143531 |
Claims
1. A metal electrodeposition cathode plate comprising: a metal plate having a plurality of disc-shaped protrusions disposed on at least one surface of the metal plate; and a non-conductive film formed on a surface of the metal plate except the protrusions, wherein a minimum film thickness of the non-conductive film at a position between centers of the adjacent protrusions is the same as or greater than a height of the protrusion.
2. The metal electrodeposition cathode plate according to claim 1, wherein the height of the protrusion is 50 .mu.m or more and 1000 .mu.m or less.
3. The metal electrodeposition cathode plate according to claim 1, wherein a difference between the minimum film thickness of the non-conductive film at the position between centers of the adjacent protrusions and the height of the protrusion is 200 .mu.m or less.
4. The metal electrodeposition cathode plate according to claim 1, wherein the metal plate is formed of titanium or stainless steel.
5. The metal electrodeposition cathode plate according to claim 1, wherein the metal electrodeposition cathode plate is used in production of electric nickel for plating.
6. A method for producing a metal electrodeposition cathode plate, comprising: a first step of forming a plurality of disc-shaped protrusions on at least one surface of a metal plate; and a second step of forming a non-conductive film on a surface of the metal plate except the protrusions, wherein a minimum film thickness of the non-conductive film at a position between centers of the adjacent protrusions is set to be the same as or greater than a height of the protrusion in the second step.
7. The metal electrodeposition cathode plate according to claim 2, wherein a difference between the minimum film thickness of the non-conductive film at the position between centers of the adjacent protrusions and the height of the protrusion is 200 .mu.m or less.
8. The metal electrodeposition cathode plate according to claim 2, wherein the metal plate is formed of titanium or stainless steel.
9. The metal electrodeposition cathode plate according to claim 3, wherein the metal plate is formed of titanium or stainless steel.
10. The metal electrodeposition cathode plate according to claim 7, wherein the metal plate is formed of titanium or stainless steel.
11. The metal electrodeposition cathode plate according to claim 2, wherein the metal electrodeposition cathode plate is used in production of electric nickel for plating.
12. The metal electrodeposition cathode plate according to claim 3, wherein the metal electrodeposition cathode plate is used in production of electric nickel for plating.
13. The metal electrodeposition cathode plate according to claim 4, wherein the metal electrodeposition cathode plate is used in production of electric nickel for plating.
14. The metal electrodeposition cathode plate according to claim 7, wherein the metal electrodeposition cathode plate is used in production of electric nickel for plating.
15. The metal electrodeposition cathode plate according to claim 8, wherein the metal electrodeposition cathode plate is used in production of electric nickel for plating.
16. The metal electrodeposition cathode plate according to claim 9, wherein the metal electrodeposition cathode plate is used in production of electric nickel for plating.
17. The metal electrodeposition cathode plate according to claim 10, wherein the metal electrodeposition cathode plate is used in production of electric nickel for plating.
Description
TECHNICAL FIELD
[0001] The present invention relates to a metal electrodeposition cathode plate and a production method therefor.
BACKGROUND ART
[0002] Conventionally, electric nickel serving as an anode raw material for nickel plating has been used by being placed in a titanium basket to be an anode holding tool and hung in a nickel plating tank. At this time, as the electric nickel of an anode raw material, those obtained by cutting plate-shaped electric nickel electrodeposited on a cathode plate into small pieces have been used.
[0003] However, the corner of the small pieces of electric nickel is sharp, and it has been thus difficult to handle the electric nickel when charging the electric nickel into a titanium basket. In addition, the small pieces of electric nickel cause so-called scaffold bridging as the corner thereof is caught by the mesh of the titanium basket after the electric nickel was charged in the titanium basket, the filling state of electric nickel in the titanium basket changes, and this causes plating unevenness in some cases.
[0004] Hence, it has been proposed to use blobby (button-shaped) electric nickel with rounded corner. The blobby electric nickel can be produced, for example, by precipitating nickel on a conductive portion by using a cathode plate on which a plurality of circular conductive portions is disposed at regular intervals by electrolysis and then peeling off the electrodeposited nickel from the conductive portion. According to such a method, it is possible to efficiently produce a plurality of pieces of blobby electric nickel from one cathode plate.
[0005] FIG. 5 is a view illustrating an example of a conventional cathode plate to be used in production of blobby electric nickel. A cathode plate 11 is masked with a non-conductive film 13 on a flat plate-shaped metal plate 12 except the place to be a conductive portion 12a, and the conductive portion 12a is a concave portion and the non-conductive film 13 is a convex portion on this cathode plate 11. Nickel having a proper size is electrodeposited on the conductive portion 12a and blobby electric nickel is thus produced by using such a cathode plate 11.
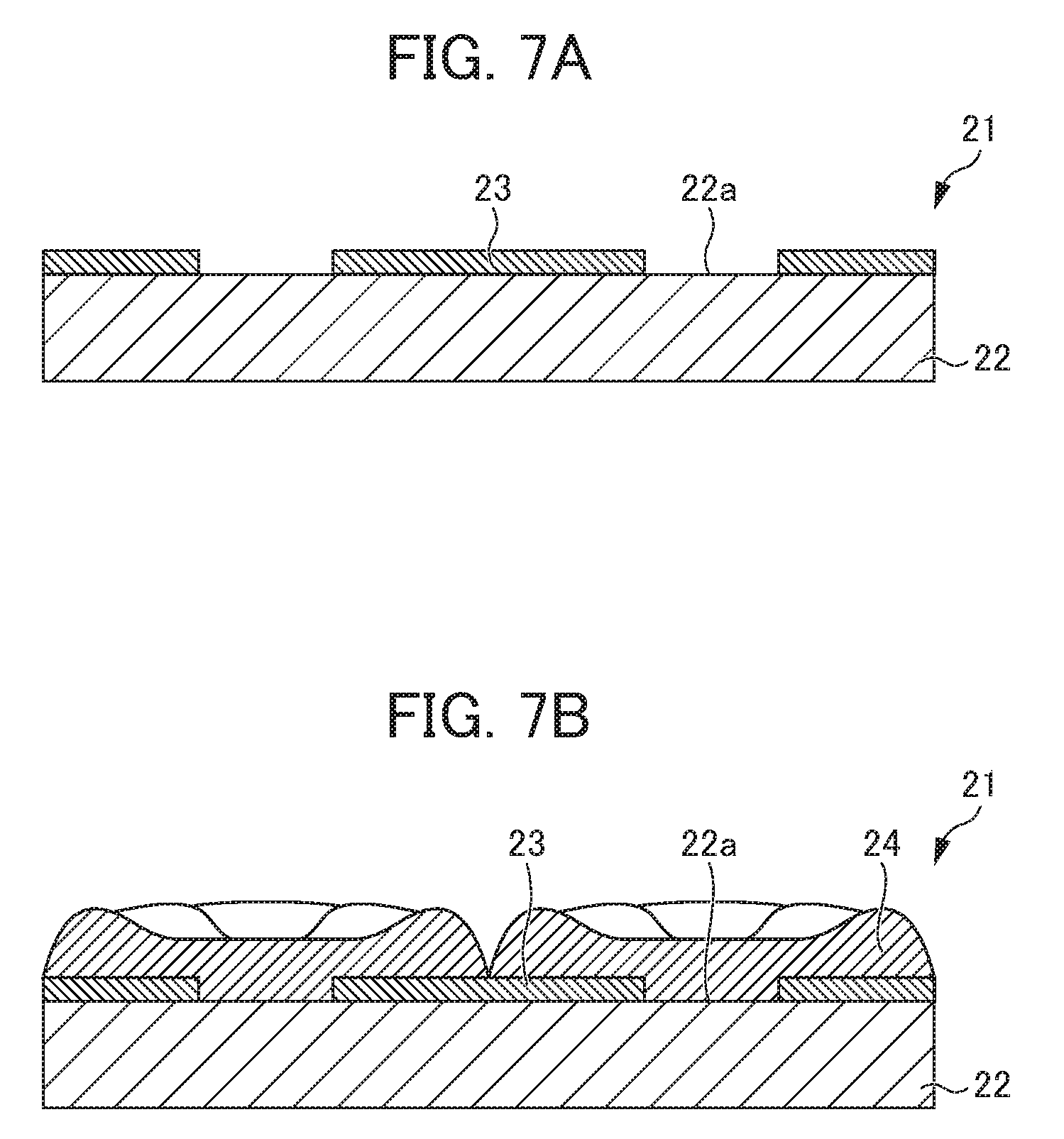
[0006] As a method for forming the non-conductive film 13 on the metal plate 12 as the cathode plate 11, for example, there is a method for forming a non-conductive film 13 having a desired pattern by coating a thermosetting non-conductive resin such as an epoxy resin on the flat plate-shaped metal plate 12 by a screen printing method and heating the thermosetting non-conductive resin as illustrated in FIG. 6A (see Patent Documents 1 and 2). Incidentally, FIG. 6B illustrates a state in which nickel (electric nickel) 14 is electrodeposited and precipitated on the conductive portion 12a by using the cathode plate 11 on which the non-conductive film 13 is formed. In the cathode plate 11, the nickel 14 begins to be electrodeposited and precipitated from the conductive portion 12a, grows not only in the thickness (longitudinal) direction but also in the planar (lateral) direction, and is in the state of being piled on the upper portion of the non-conductive film 13 as well.
[0007] In addition, for example, there has also been proposed a method for forming a non-conductive film 23 having a desired pattern by coating a photosensitive non-conductive resin on a metal plate 22 and removing the non-conductive resin at the place corresponding to a conductive portion 22a by exposure and development as illustrated in FIG. 7A. Incidentally, FIG. 7B illustrates a state in which nickel (electric nickel) 24 is electrodeposited and precipitated on the conductive portion 22a by using the cathode plate 21 on which the non-conductive film 23 is formed. In the cathode plate 21 as well, the nickel 24 begins to be electrodeposited and precipitated from the conductive portion 22a and grows not only in the thickness direction but also in the planar direction.
[0008] Furthermore, there has also been proposed a method for producing a cathode plate constituting a non-conductive portion by solidifying the periphery of a metal structure incorporated so that a plurality of studs to be a conductive portion is disposed at regular intervals with an insulating resin by an injection molding method (see Patent Document 3).
Patent Document 1: Japanese Examined Patent Application Publication No. S51-036693 Patent Document 2: Japanese Unexamined Patent Application, Publication No. S52-152832 Patent Document 3: Japanese Examined Patent Application Publication No. S56-029960
DISCLOSURE OF THE INVENTION
Problems to be Solved by the Invention
[0009] Meanwhile, in a case in which blobby electric nickel is produced using a cathode plate as described above, it is required that the non-conductive film (non-conductive portion) to be formed on the cathode plate has a long service life and can be easily maintained even in the case of being lost (deteriorated).
[0010] The film thickness of the non-conductive film 13 gradually decreases toward the conductive portion 12a and is thus significantly thin at the boundary with the conductive portion 12a in a case in which the non-conductive film 13 is formed by coating a non-conductive resin on the metal plate 12 by screen printing as illustrated in FIG. 6A. Such a change in the film thickness of the non-conductive film 13 depends on the amount of the non-conductive resin coated, the viscosity and temperature characteristics of viscosity of the non-conductive resin, the curing temperature of the non-conductive resin, the surface roughness and surface free energy of the metal surface, and the like. Hence, the film thickness of the non-conductive film 13 is significantly thin at the boundary with the conductive portion 12a.
[0011] As described above, the nickel 14 begins to be electrodeposited and precipitated from the conductive portion 12a, grows not only in the longitudinal direction but also in the lateral direction, and thus is in the state of gradually being piled on the non-conductive film 13 as well when blobby electric nickel is produced by using the cathode plate 11 as illustrated in FIG. 5 and FIG. 6. Hence, the part of the thin non-conductive film 13 to be formed in the vicinity of the boundary with the conductive portion 12a is likely to be lost by the stress at the time of electrodeposition of the nickel 14 and the impact at the time of peeling off of the electric nickel as well as the adhesive property of the part with the metal plate 12 is likely to diminish by penetration of the electrolytic solution. In addition, the non-conductive film 13 in the vicinity of the non-conductive film 13 lost rises from the surface of the metal plate 12 when loss of the non-conductive film 13 once occurs, thus the electrolytic solution is more likely to enter the gap, and as a result, the electrolytic solution gets into the gap of the non-conductive film 13 risen from the surface of the metal plate 12 and the nickel 14 is electrodeposited when it is attempted to continuously electrodeposit nickel. Thereafter, the non-conductive film 13 in which the nickel 14 is bitten is further lost when it is attempted to peel off the nickel 14 electrodeposited by being gotten into the gap.
[0012] In this manner, in the conventional cathode plate 11, when loss of the non-conductive film 13 occurs and the lost part expands in a chain reaction, the nickel 14 grown from the adjacent conductive portions 12a is likely to be connected to each other, electric nickel having a desired shape cannot be obtained, and a defective product is produced. Accordingly, it is required to peel off the entire non-conductive films 13 before loss of the non-conductive film 13 occurs, to form the non-conductive film 3 again, and thus to maintain the cathode plate 11. However, in reality, it is required to perform maintenance of the cathode plate 11 at the stage at which the electrodeposition treatment of nickel is conducted about from several times to at most less than 10 times, and not only the productivity decreases but the maintenance cost also increases.
[0013] On the other hand, it is possible to form the non-conductive film 23 having a uniform film thickness in the cathode plate 21 in which the non-conductive film 23 is formed using a photosensitive non-conductive resin by exposure and development as illustrated in FIG. 7A. However, the nickel 24 is caught by the step of the non-conductive film 23 constituting the convex portion when the nickel 24 is peeled off after the electrodeposition, a large impact is likely to be applied to the non-conductive film 23, and thus loss of the non-conductive film 23 occurs in this case as well.
[0014] Incidentally, in the method for forming a non-conductive portion by injection molding as in Patent Document 3, the production cost of the cathode plate itself increases and it is difficult to maintain the cathode plate in a case in which the non-conductive portion is deteriorated although the service life of the non-conductive portion to be formed increases.
[0015] In view of such conventional circumstances, an object of the present invention is to provide a metal electrodeposition cathode plate in which a non-conductive film on a metal plate is hardly lost and which can be repeatedly used and a production method therefor.
Means for Solving the Problems
[0016] The inventors of the present invention have carried out intensive investigations in order to solve the problems described above. As a result, it has been found out that the non-conductive film is hardly lost as protrusions are provided on a metal plate to form a conductive portion and a non-conductive film is provided on the metal surface except the protrusions, whereby the present invention has been completed.
[0017] (1) A first aspect of the present invention is a metal electrodeposition cathode plate, which includes a metal plate having a plurality of disc-shaped protrusions disposed on at least one surface of the metal plate and a non-conductive film formed on a surface of the metal plate except the protrusions, in which a minimum film thickness of the non-conductive film at a position between centers of the adjacent protrusions is the same as or greater than a height of the protrusion.
[0018] (2) A second aspect of the present invention is the metal electrodeposition cathode plate according to the first aspect, in which the height of the protrusion is 50 .mu.m or more and 1000 .mu.m or less.
[0019] (3) A third aspect of the present invention is the metal electrodeposition cathode plate according to the first or second aspect, in which a difference between the minimum film thickness of the non-conductive film at the position between centers of the adjacent protrusions and the height of the protrusion is 200 .mu.m or less.
[0020] (4) A fourth aspect of the present invention is the metal electrodeposition cathode plate according to any one of the first to third aspects, in which the metal plate is formed of titanium or stainless steel.
[0021] (5) A fifth aspect of the present invention is the metal electrodeposition cathode plate according to any one of the first to fourth aspects, in which the metal electrodeposition cathode plate is used in production of electric nickel for plating.
[0022] (6) A sixth aspect of the present invention is a method for producing a metal electrodeposition cathode plate, which includes a first step of forming a plurality of disc-shaped protrusions on at least one surface of a metal plate and a second step of forming a non-conductive film on a surface of the metal plate except the protrusions, in which a minimum film thickness of the non-conductive film at a position between centers of the adjacent protrusions is set to be the same as or greater than a height of the protrusion in the second step.
Effects of the Invention
[0023] According to the present invention, it is possible to provide a metal electrodeposition cathode plate in which a non-conductive film is hardly lost and which can be repeatedly used and a production method therefor.
BRIEF DESCRIPTION OF THE DRAWINGS
[0024] FIG. 1 is a plan view illustrating a configuration of a cathode plate.
[0025] FIG. 2 is an enlarged cross-sectional view of a main part illustrating a configuration of a cathode plate, FIG. 2A is an enlarged cross-sectional view of a main part for describing the state of a cathode plate before nickel electrodeposition, and FIG. 2B is an enlarged cross-sectional view of a main part for describing the state of a cathode plate after nickel electrodeposition.
[0026] FIG. 3 is an enlarged cross-sectional view of a main part illustrating a configuration of a cathode plate in a case in which the film thickness of the non-conductive film is thin, FIG. 3A is an enlarged cross-sectional view of a main part for describing the state of a cathode plate before nickel electrodeposition, and FIG. 3B is an enlarged cross-sectional view of a main part for describing the state of a cathode plate after nickel electrodeposition.
[0027] FIG. 4 is an enlarged cross-sectional view of a main part for describing a method for producing a cathode plate, FIG. 4A is an enlarged cross-sectional view of a main part for describing a first step, and FIG. 4B is an enlarged cross-sectional view of a main part for describing a second step.
[0028] FIG. 5 is a plan view illustrating a configuration of a conventional cathode plate.
[0029] FIG. 6 is an enlarged cross-sectional view of a main part illustrating a configuration of a conventional cathode plate, FIG. 6A is an enlarged cross-sectional view of a main part for describing the state of a cathode plate before nickel electrodeposition, and FIG. 6B is an enlarged cross-sectional view of a main part for describing the state of a cathode plate after nickel electrodeposition.
[0030] FIG. 7 is an enlarged cross-sectional view of a main part illustrating a configuration of a conventional cathode plate, FIG. 7A is an enlarged cross-sectional view of a main part for describing the state of a cathode plate before nickel electrodeposition, and FIG. 7B is an enlarged cross-sectional view of a main part for describing the state of a cathode plate after nickel electrodeposition.
PREFERRED MODE FOR CARRYING OUT THE INVENTION
[0031] Hereinafter, an embodiment (hereinafter referred to as the "present embodiment") in which the metal electrodeposition cathode plate of the present invention is applied to a metal electrodeposition cathode plate to be used in the production of electric nickel will be described in detail. It should be noted that the present invention is not limited to the following embodiments and can be appropriately changed without changing the gist of the present invention.
<1. Metal Electrodeposition Cathode Plate>
(1) Configuration of Cathode Plate
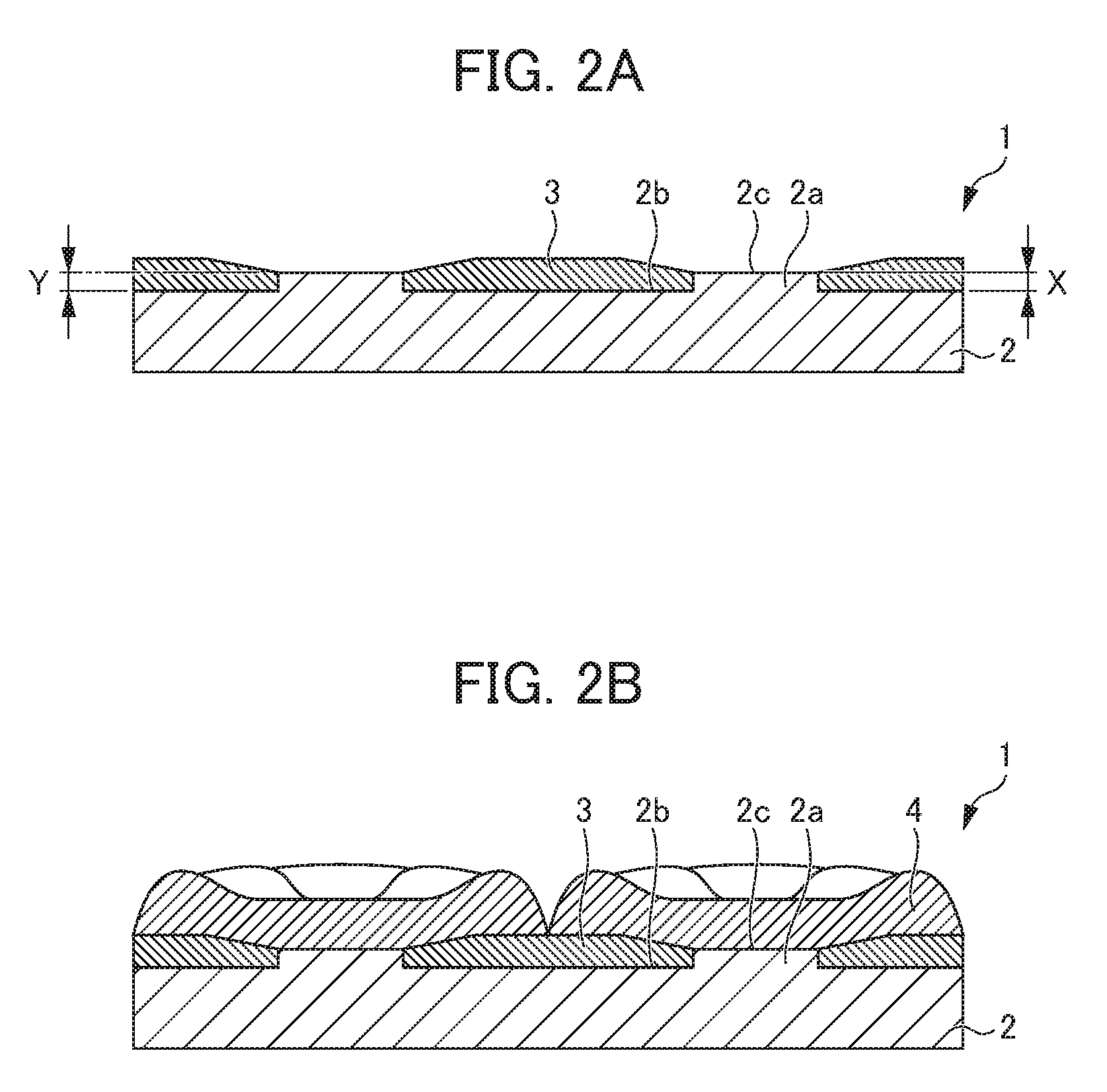
[0032] A cathode plate 1 according to the present embodiment includes a metal plate 2 on which a plurality of disc-shaped protrusions 2a is disposed and a non-conductive film 3 formed on the surface of the metal plate 2 except the protrusions 2a as illustrated in FIG. 1. The cathode plate 1 is used, for example, by being hung in an electrolytic cell containing an electrolytic solution containing nickel and an anode by a hanging member 5 and nickel having a desired shape is electrodeposited and precipitated on the surface of the cathode plate as to be described later.
[Metal Plate]
[0033] The metal plate 2 is a plate of a metal having a flat plate shape and has a plurality of disc-shaped protrusions 2a as illustrated in FIG. 1 and FIG. 2A. Here, the surface of the metal plate 2 except the protrusion 2a is referred to as a "flat area 2b" with respect to the protrusion 2a. In addition, the "height X of the protrusion" is the protruding height from the surface of the flat area 2b of the metal plate 2.
[0034] Incidentally, an example in which the protrusion 2a is provided on one surface of the metal plate 2 is illustrated in FIG. 2, but the protrusion 2a may be provided on both surfaces of the metal plate 2.
[0035] The size of the metal plate 2 is not particularly limited, and it may be set according to the desired size and number of electric nickel to be produced as appropriate. For example, the size can be set to a rectangular size of which one side is 100 mm or more and 2000 mm or less. In addition, the thickness of the metal plate 2 is preferably, for example, about 1.5 mm or more and about 5 mm or less in a case in which the protrusion 2a is provided on one surface, and it is preferably, for example, about 3 mm or more and about 10 mm or less in a case in which the protrusion 2a is provided on both surfaces. There is a tendency that warpage is likely to occur by the protrusion 2a and the flat area 2b when the thickness of the metal plate 2 is too thin. On the other hand, the weight of the metal plate 2 increases and it is difficult to handle the metal plate 2 when the thickness of the metal plate 2 is too thick.
[0036] The material for the metal plate 2 is not particularly limited as long as it is a metal which is less susceptible to corrosion by the electrolytic solution to be used and forms only loose bonding with an electrodeposit such as nickel, but preferred examples thereof may include titanium and stainless steel.
[0037] On the metal plate 2, a concave step is formed by the adjacent protrusions 2a in order to form the non-conductive film 3 having a predetermined thickness as well as the surface of a plurality of disc-shaped protrusions 2a is exposed from the non-conductive film 3 to be described later and functions as a conductive portion. Hereinafter, the surface of the protrusions 2a to be exposed from the non-conductive film 3 is referred to as a "conductive portion 2c" in some cases. Nickel 4 is electrodeposited and precipitated on the conductive portion 2c by an electrolytic treatment.
[0038] The size of the disc-shaped protrusion 2a may be set according to the desired size of electric nickel as appropriate, but the diameter thereof can be set to, for example, 5 mm or more and 30 mm or less. In addition, the height X of the protrusion 2a is preferably 50 .mu.m or more and 1000 .mu.m or less and more preferably 100 .mu.m or more and 500 .mu.m or less. When the height X of the protrusion 2a is too low, the film thickness of the non-conductive film 3 to be formed on the flat area 2b of the metal plate 2 is insufficient and the non-conductive film is likely to be lost by the stress at the time of electrodeposition of the nickel 4 and the impact at the time of peeling off of the electric nickel. On the other hand, when the height X of the protrusion 2a is too high, for example, the number of coating increases and the productivity decreases when forming a non-conductive film by screen printing. In addition, when the height X is too high, distortion of the metal plate 2 is likely to occur at the time of processing of the protrusion 2a, the metal plate 2 is likely to warp, and it is thus difficult to form the non-conductive film 3. Incidentally, it is also possible to increase the thickness of the metal plate 2 in order to diminish the influence of distortion of the metal plate 2, but the weight of the metal plate 2 increases and it is difficult to handle the metal plate.
[0039] In addition, fine concave and convex may be provided on the surface of the metal plate 2, namely, on the surface of the disc-shaped protrusion 2a of the metal plate 2 by sand blasting or etching. This makes it possible to peel off the nickel 4 electrodeposited on the protrusion 2a with a proper impact without falling off the nickel 4 during the electrolytic treatment. In this case, it is preferable that the film thickness of the non-conductive film 3 to be described later is two or more times the maximum surface roughness Rz of the metal plate 2. There is concern that pinholes and insulation failure portions are generated on the non-conductive film 3 when the film thickness of the non-conductive film 3 is thinner than two times the maximum surface roughness Rz of the metal plate 2.
[Non-Conductive Film]
[0040] The non-conductive film 3 is formed on the flat area 2b, which is the surface of the metal plate 2 except the protrusion 2a, as illustrated in FIG. 2, and the surface of a plurality of protrusions 2a disposed on the metal plate 2, namely, the conductive portion 2c is put into a state of being exposed by this. Moreover, the nickel 4 is formed by being individually divided into a small blobby shape as the nickel 4 is electrodeposited and precipitated on such a conductive portion 2c of the metal plate 2.
[0041] Here, in the cathode plate 1, the non-conductive film 3 is formed on the flat area 2b having a concave step formed by the adjacent protrusions 2a and thus the non-conductive film 3 having a predetermined thickness is formed. In the cathode plate 1 according to the present embodiment, the minimum film thickness Y of the non-conductive film 3 is the same as or greater than the height X of the protrusion 2a and it is preferably the same as the height X.
[0042] Incidentally, the "minimum film thickness Y of the non-conductive film" is defined as the minimum film thickness of the non-conductive film 3 at a position between the centers of the adjacent protrusions 2a. The non-conductive film 3 is formed as the central portion between adjacent protrusions 2a is piled by the surface tension as illustrated in FIG. 2A. In this case, the minimum film thickness Y of the non-conductive film 3 is the film thickness of the end portion in contact with the side face of the protrusion 2a. In addition, the non-conductive film 3 may be formed on the surface of the protrusion 2a in a case in which the film thickness is thick. As the minimum film thickness Y of the non-conductive film 3 at this time, not the film thickness of the non-conductive film 3 formed on the surface of the protrusion 2a but the minimum value among the film thicknesses of the non-conductive films 3 formed at the position on the flat areas 2b is taken. Incidentally, in the cathode plate 1, the film thickness varies depending on the position of the protrusion 2a to be selected but the minimum value among the film thicknesses is taken as the minimum film thickness Y.
[0043] The non-conductive film 3 is formed on the flat area 2b which is formed by the adjacent protrusions 2a and has a concave step. Hence, the film thickness of the end portion of the non-conductive film 3 is hardly thinned and the non-conductive film 3 is hardly lost even by the stress at the time of electrodeposition of the nickel 4 and the impact at the time of peeling off of the nickel 4 after electrodeposition as the conventional non-conductive film 13 illustrated in FIG. 6. In addition, the non-conductive film 3 does not protrude in a convex shape and the end portion thereof is protected by the concave step as the conventional non-conductive film 23 illustrated in FIG. 7. Consequently, the impact to be applied to the end portion of the non-conductive film 3 by the nickel 4 is minor and the non-conductive film 3 is hardly lost even when the nickel 4 is peeled off from the cathode plate 1. In this manner, in the cathode plate 1, the non-conductive film 3 is hardly lost and it is thus possible to repeatedly use the non-conductive film 3 in electrodeposition without replacing the non-conductive film 3, to decrease the maintenance cost, and to achieve improvement in the productivity.
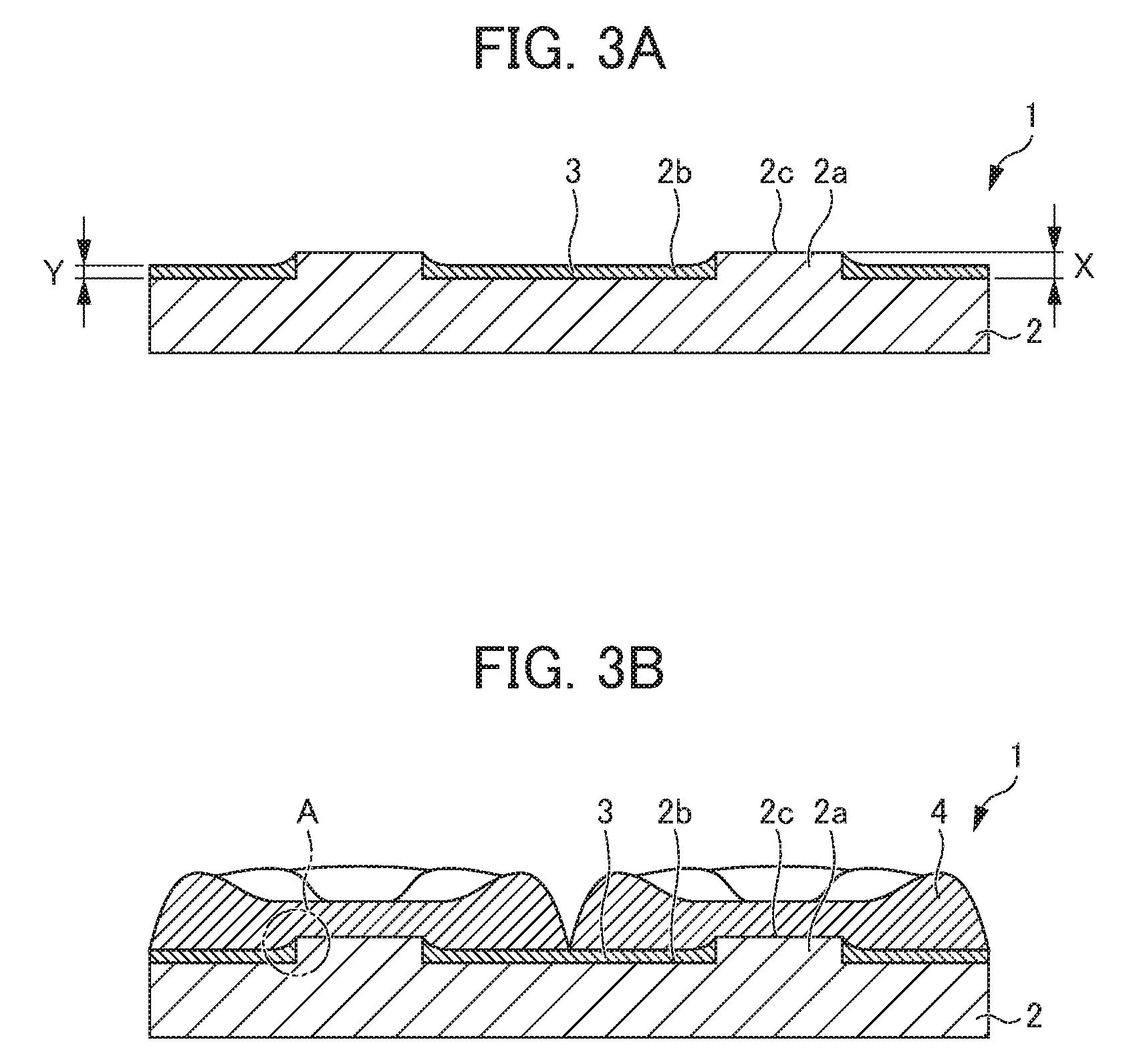
[0044] Furthermore, the minimum film thickness Y of the non-conductive film 3 is the same as or greater than the height X of the protrusion 2a, and the nickel 4 can be thus peeled off without being caught by the peripheral portion of the protrusion 2a when the nickel 4 is peeled off from the cathode plate 1. On the other hand, in a case in which the minimum film thickness Y of the non-conductive film 3 is less than the height X of the protrusion 2a as illustrated in FIG. 3, it is difficult to peel off the electrodeposited nickel 4 since the electrodeposited nickel 4 is caught by the peripheral portion of the protrusion 2a, for example, at the place denoted by "A" in the drawing when the electrodeposited nickel 4 is peeled off from the cathode plate 1.
[0045] The upper limit of the minimum film thickness Y of the non-conductive film 3 is not particularly limited, but the difference (Y-X) between the minimum film thickness Y and the height X of the protrusion 2a is preferably 200 .mu.m or less, more preferably 100 .mu.m or less, still more preferably 50 .mu.m or less, and particularly preferably 5 .mu.m or less. Here, as described above, the minimum film thickness Y of the non-conductive film 3 is not particularly limited as long as it is the same as or greater than the height X of the protrusion 2a, but it is not required to set the minimum film thickness Y thicker than necessary. For example, it is difficult to coat the non-conductive film 3 so as to have a film thickness thicker than the height X of the protrusion 2a by more than 200 .mu.m by screen printing. It is required to conduct coating while finely adjusting the size of the pattern of the screen plate plural times when it is attempted to form the non-conductive film 3 having a film thickness thicker than the height X of the protrusion 2a by more than 200 .mu.m by screen printing, and thus the adjustment is difficult and the productivity decreases.
[0046] Incidentally, in a case in which the non-conductive film 3 is formed on the flat area 2b on the metal plate 2 by the screen printing method, the material for the non-conductive film 3 is coated on the surface of the protrusion 2a as well, thus the surface area of the conductive portion 2c decreases and the initial current density increases in some cases, but there is no problem as long as troubles are not caused in the characteristics of the electrodeposited nickel 4. In addition, the non-conductive film 3 attached on the surface of the protrusion 2a is likely to be lost since the film thickness thereof is extremely thin, but the non-conductive film 3 to be formed on the flat area 2b has no problem since the film thickness thereof is thick and the loss thereof is suppressed.
[0047] The non-conductive film 3 is not particularly limited as long as it is formed from a material which is non-conductive and is less susceptible to corrosion by the electrolytic solution to be used. For example, it is preferable that the non-conductive film 3 is composed of a thermosetting resin or a photocuring (ultraviolet curing and the like) resin from the viewpoint of being easy to form the film. Specific examples thereof may include an insulating resin such as an epoxy-based resin, a phenol-based resin, a polyamide-based resin, or a polyimide-based resin.
(2) Production of Electric Nickel Using Cathode Plate
[0048] In the cathode plate 1 having the configuration described above, the surface of the protrusion 2a to be exposed from the non-conductive film 3 is the conductive portion 2c and the nickel 4 is electrodeposited and precipitated thereon as illustrated in FIG. 2B. In the cathode plate 1, the nickel 4 grows not only in the thickness direction but also in the planar direction and is thus in the state of being piled on the upper part of the non-conductive film 3. For this reason, it is preferable to terminate the electrodeposition before the nickel 4 grown from the conductive portion 2c of the surface of the adjacent protrusion 2a comes into contact with each other.
[0049] Thereafter, a plurality of pieces of blobby electric nickel can be obtained from one cathode plate 1 by peeling off the nickel 4 from the cathode plate 1 after the electrodeposition of nickel is terminated. As described above, in the cathode plate 1 according to the present embodiment, the non-conductive film 3 is hardly lost and it is thus possible to repeatedly use the non-conductive film 3 without replacing the non-conductive film 3, to decrease the maintenance cost, and to achieve improvement in the productivity.
[0050] Incidentally, in the cathode plate 1 according to the present embodiment, the nickel 4 is electrodeposited but silver, gold, zinc, tin, chromium, cobalt, or any alloy thereof may be electrodeposited without being limited to nickel.
<2. Method for Producing Metal Electrodeposition Cathode Plate>
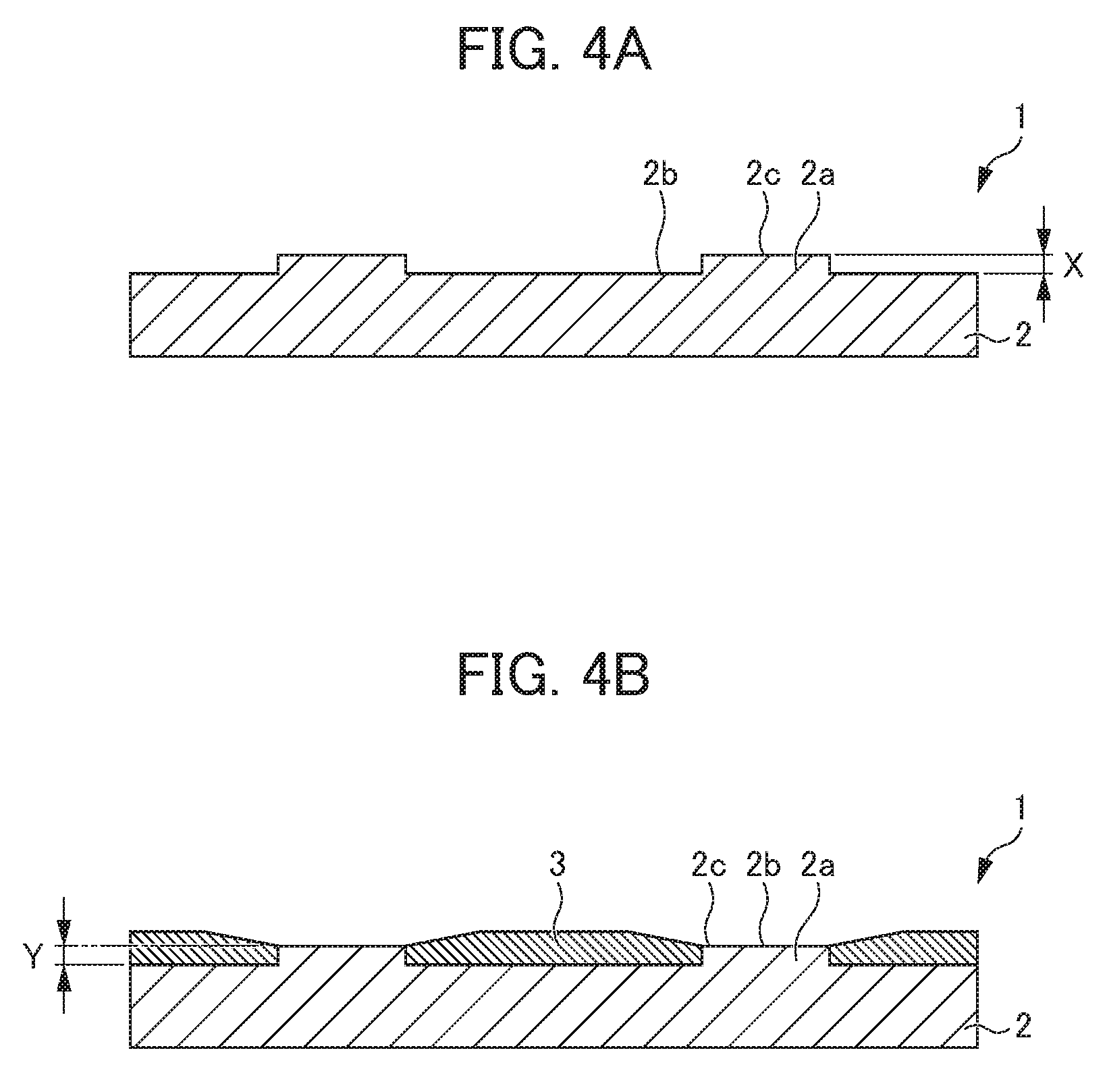
[0051] The method for producing a cathode plate 1 according to the present embodiment includes a first step (FIG. 4A) of forming a plurality of disc-shaped protrusions 2a on at least one surface of a metal plate 2 and a second step (FIG. 4B) of forming a non-conductive film 3 on the surface of the metal plate 2 except the protrusions 2a as illustrated in FIG. 4.
[First Step]
[0052] In the first step, a plurality of disc-shaped protrusions 2a is formed on the surface of the metal plate 2. For example, the parts of the flat plate-shaped metal plate 2 except the protrusions 2a are scraped, the protrusions 2a having a height X are left, and flat areas 2b are thus formed. The processing method is not particularly limited, and the formation of flat areas 2b can be conducted by, for example, wet etching processing, end mill processing, and laser processing.
[0053] For example, in the case of processing a flat plate-shaped stainless steel plate by wet etching, a photosensitive etching resist is coated on the surface of a stainless steel plate and is then exposed by passing through a film or glass on which a desired pattern is drawn and the etching resist of the part to be etched is removed by a development treatment. Thereafter, the stainless steel plate developed is dipped in an etching solution (for example, a ferric chloride solution), a part of the stainless steel plate from which the etching resist has been removed is removed, and finally, the etching resist is peeled off, whereby a plurality of disc-shaped protrusions 2a matching with a desired pattern can be formed.
[0054] Incidentally, the protrusions 2a may be formed only on one surface of the metal plate 2 or on both surfaces of the metal plate 2.
[Second Step]
[0055] In the second step, the non-conductive film 3 is formed on the flat areas 2b to be the surface of the metal plate 2 except the protrusions 2a. The method for forming the non-conductive film 3 is not particularly limited, and the formation of the non-conductive film 3 can be conducted by screen printing. In a case in which the material for the non-conductive film 3 is a thermosetting resin or a photocurable resin, heat curing or photocuring may be conducted if necessary.
[0056] At this time, the non-conductive film 3 is formed so that the minimum film thickness Y of the non-conductive film 3 at the position between the centers of adjacent protrusions 2a is the same as or greater than the height X of the protrusion 2a. In a case in which a desired film thickness cannot be obtained by one time of screen printing, the above-described screen printing and heat curing or photocuring may be repeated until the desired film thickness is obtained.
[0057] According to the method for producing a cathode plate according to the present embodiment, it is possible to obtain the cathode plate 1 in which the non-conductive film on the metal plate is hardly lost and which can be repeatedly used.
EXAMPLES
[0058] Hereinafter, the present invention will be described more specifically with reference to Examples, but the present invention is not limited by these Examples at all. It should be noted that members having the same functions as the members illustrated in FIG. 1 to FIG. 6 are denoted by the same reference numerals for the sake of convenience.
<Fabrication of Cathode Plate>
Example 1
[0059] A cathode plate 1 as illustrated in FIG. 1 and FIG. 2 was fabricated. Specifically, first, a metal plate 2 which was made of stainless steel and had a size of 200 mm.times.100 mm.times.4 mm was subjected to wet etching to form disc-shaped protrusions 2a (18 pieces). At this time, the size of the protrusion 2a was set to a diameter of 14 mm and a height X of 300 .mu.m, and the minimum center-distance between adjacent protrusions 2a was set to 21 mm.
[0060] Next, a thermosetting epoxy resin was coated on flat areas 2b of the metal plate 2 by a screen printing method and cured by heating at 150.degree. C. for 60 minutes to form a non-conductive film 3. In the cathode plate 1 fabricated in this manner, the difference between the minimum film thickness Y of the non-conductive film 3 and the height X of the protrusion at a position between the centers of adjacent protrusions 2a was measured at arbitrary 10 places by using a laser displacement meter, and the results were in a range of from 40 to 70 .mu.m and the minimum film thickness Y of the non-conductive film 3 was thus 340 .mu.m.
Example 2
[0061] A cathode plate 1 was fabricated in the same manner as in Example 1 except that the height X of the protrusion 2a of the metal plate 2 was set to 500 .mu.m and the non-conductive film 3 was formed on the flat area 2b so as to have a predetermined thickness. In the cathode plate 1 fabricated in this manner, the difference between the minimum film thickness Y of the non-conductive film 3 and the height X of the protrusion 2a was measured at arbitrary 10 places by using a laser displacement meter, and the results were in a range of from 10 to 50 .mu.m and the minimum film thickness Y of the non-conductive film 3 was thus 510 .mu.m.
Example 3
[0062] A cathode plate 1 was fabricated in the same manner as in Example 1 except that the height X of the protrusion 2a of the metal plate 2 was set to 60 .mu.m and the non-conductive film 3 was formed on the flat area 2b so as to have a predetermined thickness. In the cathode plate 1 fabricated in this manner, the difference between the minimum film thickness Y of the non-conductive film 3 and the height X of the protrusion was measured at arbitrary 10 places by using a laser displacement meter, and the results were in a range of from 60 to 90 .mu.m and the minimum film thickness Y of the non-conductive film 3 was thus 120 .mu.m.
Example 4
[0063] A cathode plate 1 was fabricated in the same manner as in Example 1 except that the height X of the protrusion 2a of the metal plate 2 was set to 100 .mu.m and the non-conductive film 3 was formed on the flat area 2b so as to have a predetermined thickness. In the cathode plate 1 fabricated in this manner, the difference between the minimum film thickness Y of the non-conductive film 3 and the height X of the protrusion was measured at arbitrary 10 places by using a laser displacement meter, and the results were in a range of from 100 to 150 .mu.m and the minimum film thickness Y of the non-conductive film 3 was thus 200 .mu.m.
Example 5
[0064] A cathode plate 1 was fabricated in the same manner as in Example 1 except that the height X of the protrusion 2a of the metal plate 2 was set to 40 .mu.m and the non-conductive film 3 was formed on the flat area 2b so as to have a predetermined thickness. In the cathode plate 1 fabricated in this manner, the difference between the minimum film thickness Y of the non-conductive film 3 and the height X of the protrusion 2a was measured at arbitrary 10 places by using a laser displacement meter, and the results were in a range of from 10 to 40 .mu.m and the minimum film thickness Y of the non-conductive film 3 was thus 50 .mu.m.
Comparative Example 1
[0065] In Comparative Example 1, a conventional cathode plate 11 as illustrated in FIG. 5 and FIG. 6 was fabricated.
[0066] Specifically, a thermosetting epoxy resin was coated on a flat plate-shaped metal plate 12 which was made of stainless steel and had a size of 200 mm.times.100 mm.times.4 mm except conductive portions 12a (18 pieces) having a diameter of 14 mm by a screen printing method and cured by heating at 150.degree. C. for 60 minutes to form a non-conductive film 13, whereby the cathode plate 11 was fabricated. In the cathode plate 11 fabricated in this manner, the maximum film thickness of the non-conductive film 13 was measured at arbitrary 10 places by using a laser displacement meter, and the results were in a range of from 90 to 110 .mu.m.
Comparative Example 2
[0067] A cathode plate was fabricated in the same manner as in Example 1 except that the height X of the protrusion of the metal plate was set to 500 .mu.m and the non-conductive film was formed on the flat area so as to have a predetermined thickness. In the cathode plate fabricated in this manner, the difference between the minimum film thickness of the non-conductive film and the height of the protrusion was measured at arbitrary 10 places by using a laser displacement meter, and the results were in a range of from -200 to -150 .mu.m and the minimum film thickness Y of the non-conductive film 3 was thus 300 .mu.m. Incidentally, the minimum film thickness Y of the non-conductive film 3 is thinner than 500 .mu.m of the height of the protrusion.
Comparative Example 3
[0068] A metal plate which was made of stainless steel and had a size of 200 mm.times.100 mm.times.4 mm was subjected to wet etching to form protrusions (18 pieces) having a height of 2000 .mu.m. However, warpage of the metal plate was severe and it was difficult to form a non-conductive film by screen printing.
<Production of Electric Nickel>
[0069] Electric nickel was produced by an electrolytic treatment using the cathode plates fabricated in the respective Examples and Comparative Examples. Specifically, the cathode plate and an anode plate which was composed of electric nickel and had a size of 200 mm.times.100 mm.times.10 mm were dipped in an electrolytic tank containing a nickel chloride electrolytic solution so as to face each other. Thereafter, nickel was electrodeposited on the surface of the cathode plate under the conditions of an initial current density of 710 A/m.sup.2 and an electrolysis time of 3 days. After the electrolysis, the electric nickel precipitated on the cathode plate was peeled off to obtain blobby electric nickel for plating.
<Evaluation>
[0070] The number of times, by which the cathode plate used in the electrolysis treatment was able to be repeatedly utilized as it was, was evaluated. Nickel electrodeposited at the adjacent protrusions and conductive portions are connected to each other and electric nickel having a desired shape cannot be obtained in some cases when the loss of the non-conductive film expands. Hence, the use was stopped and the number of repetitions up to this time point was evaluated in a case in which the non-conductive film was lost from the boundary with the protrusion in the direction of the flat area by 1 mm or more. In addition, the use was stopped and the number of repetitions up to this time point was evaluated in a case in which the non-conductive film was lost and the diameter of the conductive portion increased by 1 mm or more as well.
[0071] The evaluation results are presented in the following Table 1 together with the configuration of the cathode plate.
TABLE-US-00001 TABLE 1 Height Minimum Maximum X of film film protrusion thickness thickness Y - X Number of (.mu.m) Y (.mu.m) (.mu.m) (.mu.m) repeated use Example 1 300 340 -- 40 20 or more Example 2 500 510 -- 10 20 or more Example 3 60 120 -- 60 16 Example 4 100 200 -- 100 20 or more Example 5 40 50 -- 10 9 Comparative -- -- 90~110 -- 7 Example 1 Comparative 500 300 -- -200 (Difficult Example 2 to peel off)
[0072] As presented in Table 1, in Examples 1 to 5 using the cathode plates 1 in which the non-conductive film 3 was formed on the flat area 2b of the metal plate 2 and the minimum film thickness Y of the non-conductive film 3 was the same as or greater than the height X of the protrusion 2a, loss of the non-conductive film 3 was suppressed and it was possible to sufficiently repeatedly use the cathode plates 1. Particularly, in Examples 1 to 4 in which the height X of the protrusion 2a was 50 .mu.m or more, the number of repeated use was more than 10 times.
[0073] On the other hand, in Comparative Example 1 in which the non-conductive film 13 was formed in a convex shape on the flat plate-shaped metal plate 12, the non-conductive film was lost and it was not possible to sufficiently repeatedly use the cathode plate. In addition, in Comparative Example 2 in which the minimum film thickness Y of the non-conductive film was less than the height X of the protrusion, nickel was caught by the peripheral portion of the protrusion at the time of peeling off of nickel and it was difficult to peel off nickel.
EXPLANATION OF REFERENCE NUMERALS
[0074] 1 CATHODE PLATE [0075] 2 METAL PLATE [0076] 2a PROTRUSION [0077] 2b FLAT AREA [0078] 2c CONDUCTIVE PORTION [0079] 3 NON-CONDUCTIVE FILM [0080] 4 NICKEL
* * * * *
D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.