Steel for Machine Structural Use
Hashimura; Masayuki ; et al.
U.S. patent application number 16/313931 was filed with the patent office on 2019-08-01 for steel for machine structural use. The applicant listed for this patent is Nippon Steel & Sumitomo Metal Corporation. Invention is credited to Makoto Egashira, Masayuki Hashimura, Takanori Iwahashi, Shouji Toudou.
| Application Number | 20190233927 16/313931 |
| Document ID | / |
| Family ID | 60912701 |
| Filed Date | 2019-08-01 |

| United States Patent Application | 20190233927 |
| Kind Code | A1 |
| Hashimura; Masayuki ; et al. | August 1, 2019 |
Steel for Machine Structural Use
Abstract
A steel for machine structural use is provided which is excellent in machinability, rusting characteristics, and hot ductility. The steel for machine structural use according to the present embodiment has a chemical composition which consists of, in mass %, C: 0.30 to 0.80%, Si: 0.01 to 0.80%, Mn: 0.20 to 2.00%, P: 0.030% or less, S: 0.010 to 0.100%, Pb: 0.010 to 0.100%, Al: 0.010 to 0.050%, N: 0.015% or less and O: 0.0005 to 0.0030%, with the balance being Fe and impurities, the chemical composition satisfying Formula (1). The total number of specific inclusions included in the steel which are any of MnS inclusions, Pb inclusions and composite inclusions containing MnS and Pb and which have an equivalent circular diameter of 5 .mu.m or more is 40 per mm.sup.2 or more. Mn/S.gtoreq.8.0 (1) Where, a content (mass %) of a corresponding element is substituted for each symbol of an element in Formula (1).
| Inventors: | Hashimura; Masayuki; (Chiyoda-ku, Tokyo, JP) ; Egashira; Makoto; (Chiyoda-ku, Tokyo, JP) ; Iwahashi; Takanori; (Chiyoda-ku, Tokyo, JP) ; Toudou; Shouji; (Chiyoda-ku, Tokyo, JP) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 60912701 | ||||||||||
| Appl. No.: | 16/313931 | ||||||||||
| Filed: | July 4, 2017 | ||||||||||
| PCT Filed: | July 4, 2017 | ||||||||||
| PCT NO: | PCT/JP2017/024442 | ||||||||||
| 371 Date: | December 28, 2018 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | B22D 11/001 20130101; C22C 38/12 20130101; C21D 8/065 20130101; C22C 38/60 20130101; C22C 38/16 20130101; C22C 38/08 20130101; C22C 38/001 20130101; B22D 11/124 20130101; C22C 38/06 20130101; C22C 38/14 20130101; C21D 2211/004 20130101; C22C 38/38 20130101; C22C 38/02 20130101; C22C 38/04 20130101; C22C 38/002 20130101 |
| International Class: | C22C 38/60 20060101 C22C038/60; C22C 38/38 20060101 C22C038/38; C22C 38/02 20060101 C22C038/02; C22C 38/04 20060101 C22C038/04; C22C 38/08 20060101 C22C038/08; C22C 38/12 20060101 C22C038/12; C22C 38/16 20060101 C22C038/16; C22C 38/14 20060101 C22C038/14; C22C 38/06 20060101 C22C038/06; C22C 38/00 20060101 C22C038/00 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Jul 4, 2016 | JP | 2016-132902 |
Claims
1. A steel for machine structural use having a chemical composition consisting of, in mass %: C: 0.30 to 0.80%, Si: 0.01 to 0.80%, Mn: 0.20 to 2.00%, P: 0.030% or less, S: 0.010 to 0.100%, Pb: 0.010 to 0.100%, Al: 0.010 to 0.050%, N: 0.015% or less, O: 0.0005 to 0.0030%, Cr: 0 to 0.70%, Ni: 0 to 3.50%, B: 0 to 0.0050%, V: 0 to 0.70%, Mo: 0 to 0.70%, W: 0 to 0.70%, Nb: 0 to less than 0.050%, Cu: 0 to 0.50%, Ti: 0 to 0.100%, and Ca: 0 to 0.0030%, with the balance being Fe and impurities, the chemical composition satisfying Formula (1), wherein, in the steel, a total number of specific inclusions which are any of MnS inclusions, Pb inclusions and composite inclusions containing MnS and Pb, and which have an equivalent circular diameter of 5 .mu.m or more is 40 per mm.sup.2 or more; Mn/S.gtoreq.8.0 (1) where, a content (mass %) of a corresponding element is substituted for each symbol of an element in Formula (1).
2. The steel for machine structural use according to claim 1, wherein the chemical composition contains one or more types of element selected from a group consisting of: Cr: 0.10 to 0.70%, Ni: 0.02 to 3.50%, B: 0.0005 to 0.0050%, V: 0.05 to 0.70%, Mo: 0.05 to 0.70%, W: 0.05 to 0.70%, Nb: 0.001 to less than 0.050%, Cu: 0.05 to 0.50%, and Ti: 0.003 to 0.100%.
3. The steel for machine structural use according to claim 1, wherein the chemical composition contains: Ca: 0.0001 to 0.0030%.
4. The steel for machine structural use according to claim 1, wherein: a ratio of a number of the composite inclusions with respect to the specific inclusions is 40% or more.
5. The steel for machine structural use according to claim 2, wherein the chemical composition contains: Ca: 0.0001 to 0.0030%.
6. The steel for machine structural use according to claim 2, wherein: a ratio of a number of the composite inclusions with respect to the specific inclusions is 40% or more.
7. The steel for machine structural use according to claim 3, wherein: a ratio of a number of the composite inclusions with respect to the specific inclusions is 40% or more.
8. The steel for machine structural use according to claim 5, wherein: a ratio of a number of the composite inclusions with respect to the specific inclusions is 40% or more.
Description
TECHNICAL FIELD
[0001] The present invention relates to a steel, and more particularly relates to a steel for machine structural use.
BACKGROUND ART
[0002] Machine components that are to be used for structural use and power transmission use such as general machine components and automobile components are produced using a steel for machine structural use. An example of a method for producing such kind of machine components is as follows. A steel for machine structural use is subjected to hot working (hot forging or the like) to produce an intermediate product. The intermediate product is subjected to machining (cutting or grinding) to produce a machine component. There are also cases in which, as necessary, the machine component is subjected to a heat treatment (normalizing or the like), a case hardening heat treatment (induction hardening or the like), or quenching and tempering. A steel for machine structural use for producing such kind of machine components is required to be excellent not only in hot workability, but also in machinability.
[0003] A steel for machine structural use that is excellent in machinability is also called a "free-cutting steel", and is defined in JIS G 4804 (2008) (Non Patent Literature 1). The machinability of a free-cutting steel is enhanced by containing Pb.
[0004] A steel for machine structural use that contains Pb is disclosed, for example, in Japanese Patent Application Publication No. 2000-282172 (Patent Literature 1). The steel material for machine structural use disclosed in Patent Literature 1 has a chemical composition which contains, in mass %, C: 0.05 to 0.55%, Si: 0.50 to 2.5%, Mn: 0.01 to 2.00%, S: 0.005 to 0.080%, Cr: 0 to 2.0%, P: 0.035% or less, V: 0 to 0.50%, N: 0.0150% or less, Al: 0.04% or less, Ni: 0 to 2.0%, Mo: 0 to 1.5%, B: 0 to 0.01%, Bi: 0 to 0.10%, Ca: 0 to 0.05%, Pb: 0 to 0.12%, Ti: 0 to less than 0.04%, Zr: 0 to less than 0.04% and Ti (%)+Zr (%): 0 to less than 0.04%, Te: 0 to 0.05%, Nd: 0 to 0.05%, Nb: 0 to 0.1%, Cu: 0 to 1.5%, and Se: 0 to 0.5%, the chemical composition satisfying a condition that a value of fn1 represented by a formula hereunder is 100 or less, a value of fn2 represented by a formula hereunder is 0 or more, and a value of fn3 represented by a formula hereunder is 3.0 or more, with the balance being Fe and impurities. In addition, a proportion that a ferritic phase occupies in the micro-structure is, with respect to the area ratio, from 10 to 80%, and Hv hardness is in a range from 160 to 350. Here, fn1=100C+11Si+18Mn+32Cr+45Mo+6V, fn2=-23C+Si(5-2Si)-4Mn+104S-3Cr-9V+10, and fn3=3.2C+0.8Mn+5.2S+0.5Cr-120N+2.6Pb+4.1Bi-0.001.alpha..sup.2+0.13.alpha.- . A symbol of an element in the respective formulas represents the content in mass % of the corresponding element, and .alpha. represents the area ratio (%) of the ferritic phase in the micro-structure. It is described in Patent Literature 1 that the steel material for machine structural use is excellent in machinability and toughness.
CITATION LIST
Patent Literature
[0005] Patent Literature 1: Japanese Patent Application Publication No. 2000-282172
Non Patent Literature
[0005] [0006] Non Patent Literature 1: Japanese Industrial Standards Committee, Standard No.: JIS G 4804 (2008), Standard Name: Free-cutting Steels
SUMMARY OF INVENTION
Technical Problem
[0007] In some cases machining such as cutting is performed using an automated equipment system. In an automated equipment system, in the case of producing a large amount of machine components by cutting intermediate products, such as producing several hundred or more machine components per day, excellent chip treatability is required. It is preferable that chips that are to be discharged accompanying cutting are split into small pieces and discharged. If the chips remain connected in a long length, the chips are liable to become entwined around the intermediate product, and defects are liable to arise on the surface of the machine component after cutting. If a chip is entwined around a machine component, it is also necessary to temporarily stop the production line to remove the chip that is entwined around the machine component. In this case, it is difficult to perform unattended production, and it is necessary to assign personnel to monitor the production process. Thus, chip treatability affects both the quality of the machine components and the production cost. In addition, in an automated equipment system, productivity decreases as tool wear increases. Therefore, a steel for machine structural use is required to have high machinability, such as being capable of suppressing tool wear and being excellent in chip treatability.
[0008] In addition, when cutting is performed using automated equipment system, in some cases rust occurs in the machine component. In an automated equipment system, a water-soluble cutting oil is utilized from the viewpoint of performing unattended operations. Consequently, in some cases rust occurs in the machine components. Rust is not only a cause of the occurrence of shape errors, but is also a cause of quality defects when performing a plating treatment on the machine component. In addition, after cutting, the machine components are sometimes stored in a bucket or the like for a long time period until undergoing the next process after the cutting process. For example, in a case where machine components are cut in a certain country and the next process is performed in a separate factory in a different country, a period of several days to several months may pass after cutting until the machine components are subjected to the next process. Therefore, a steel for machine structural use is required to be not only excellent in machinability, but also to have characteristics that suppress the occurrence of rust (hereunder, referred to as "rusting characteristics").
[0009] An objective of the present invention is to provide a steel for machine structural use that is excellent in machinability, rusting characteristics and hot workability.
Solution to Problem
[0010] A steel for machine structural use according to the present invention has a chemical composition which consists of, in mass %, C: 0.30 to 0.80%, Si: 0.01 to 0.80%, Mn: 0.20 to 2.00%, P: 0.030% or less, S: 0.010 to 0.100%, Pb: 0.010 to 0.100%, Al: 0.010 to 0.050%, N: 0.015% or less, O: 0.0005 to 0.0030%, Cr: 0 to 0.70%, Ni: 0 to 3.50%, B: 0 to 0.0050%, V: 0 to 0.70%, Mo: 0 to 0.70%, W: 0 to 0.70%, Nb: 0 to less than 0.050%, Cu: 0 to 0.50%, Ti: 0 to 0.100% and Ca: 0 to 0.0030%, with the balance being Fe and impurities, the chemical composition satisfying Formula (1). In the steel, a total number of specific inclusions which are any of MnS inclusions, Pb inclusions, and composite inclusions containing MnS and Pb and which have an equivalent circular diameter of 5 .mu.m or more is 40 per mm.sup.2 or more.
Mn/S.gtoreq.8.0 (1)
[0011] Where, a content (mass %) of a corresponding element is substituted for each symbol of an element in Formula (1).
Advantageous Effects of Invention
[0012] A steel for machine structural use according to the present invention is excellent in machinability, rusting characteristics and hot workability.
BRIEF DESCRIPTION OF DRAWINGS
[0013] FIG. 1A is a schematic diagram illustrating an S distribution in an observation surface, that was obtained by EPMA analysis.
[0014] FIG. 1B is a schematic diagram illustrating a Pb distribution in the same observation surface as in FIG. 1A, that was obtained by EPMA analysis.
[0015] FIG. 1C is a schematic diagram of an image obtained by combining FIG. 1A and FIG. 1B.
[0016] FIG. 2 is a schematic diagram for describing a criterion for determining whether or not to regard adjacent inclusions as a single inclusion.
[0017] FIG. 3A is a photographic image of an S distribution obtained by performing EPMA analysis on the steel for machine structural use of the present embodiment.
[0018] FIG. 3B is a photographic image of a Pb distribution obtained by performing EPMA with respect to the same visual fields as in FIG. 3A.
[0019] FIG. 3C is a composite image of the images shown in FIG. 3A and FIG. 3B.
[0020] FIG. 4 is a transverse sectional view of a starting material that was cast.
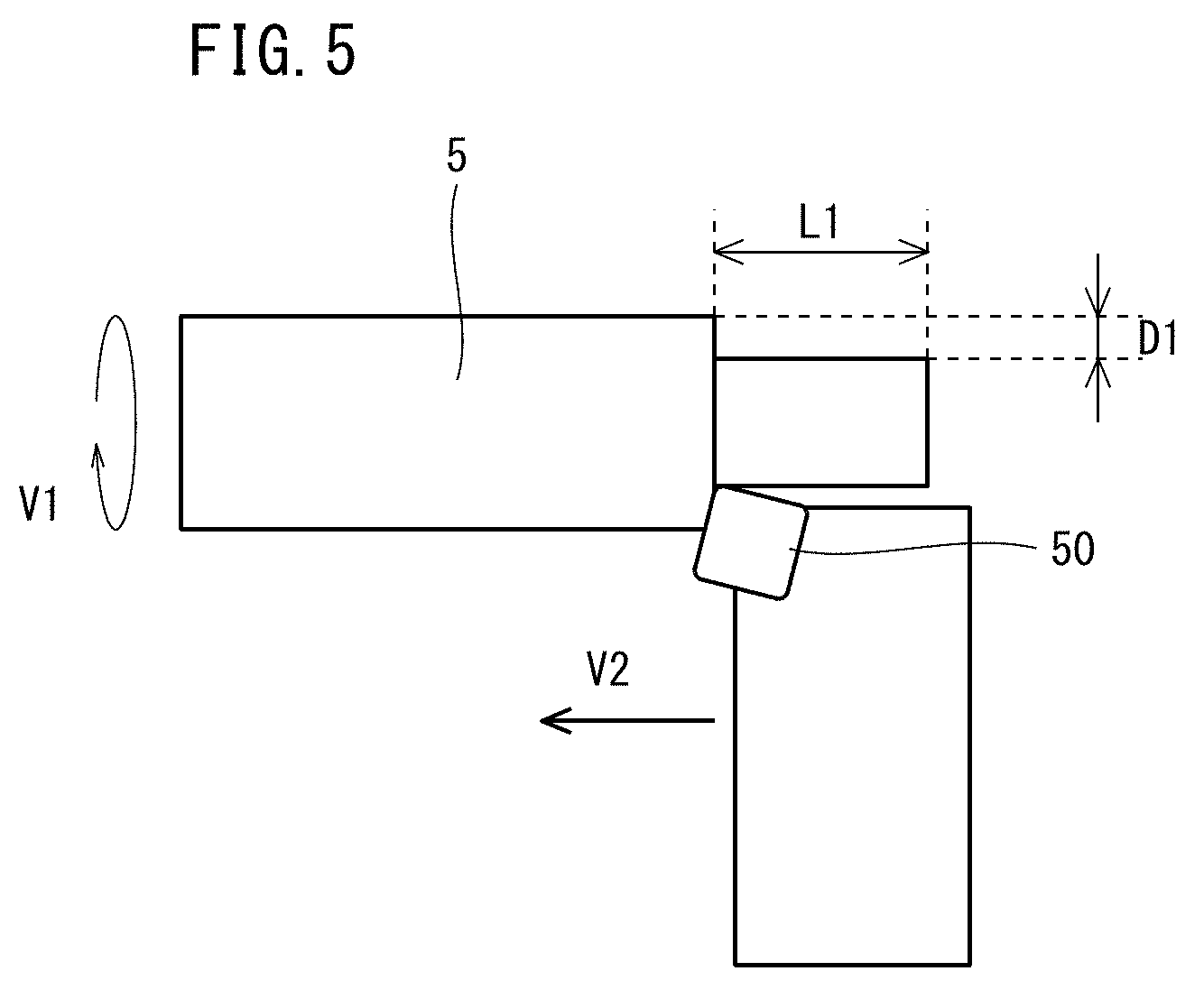
[0021] FIG. 5 is a schematic diagram of a cutting test machine for describing a cutting test.
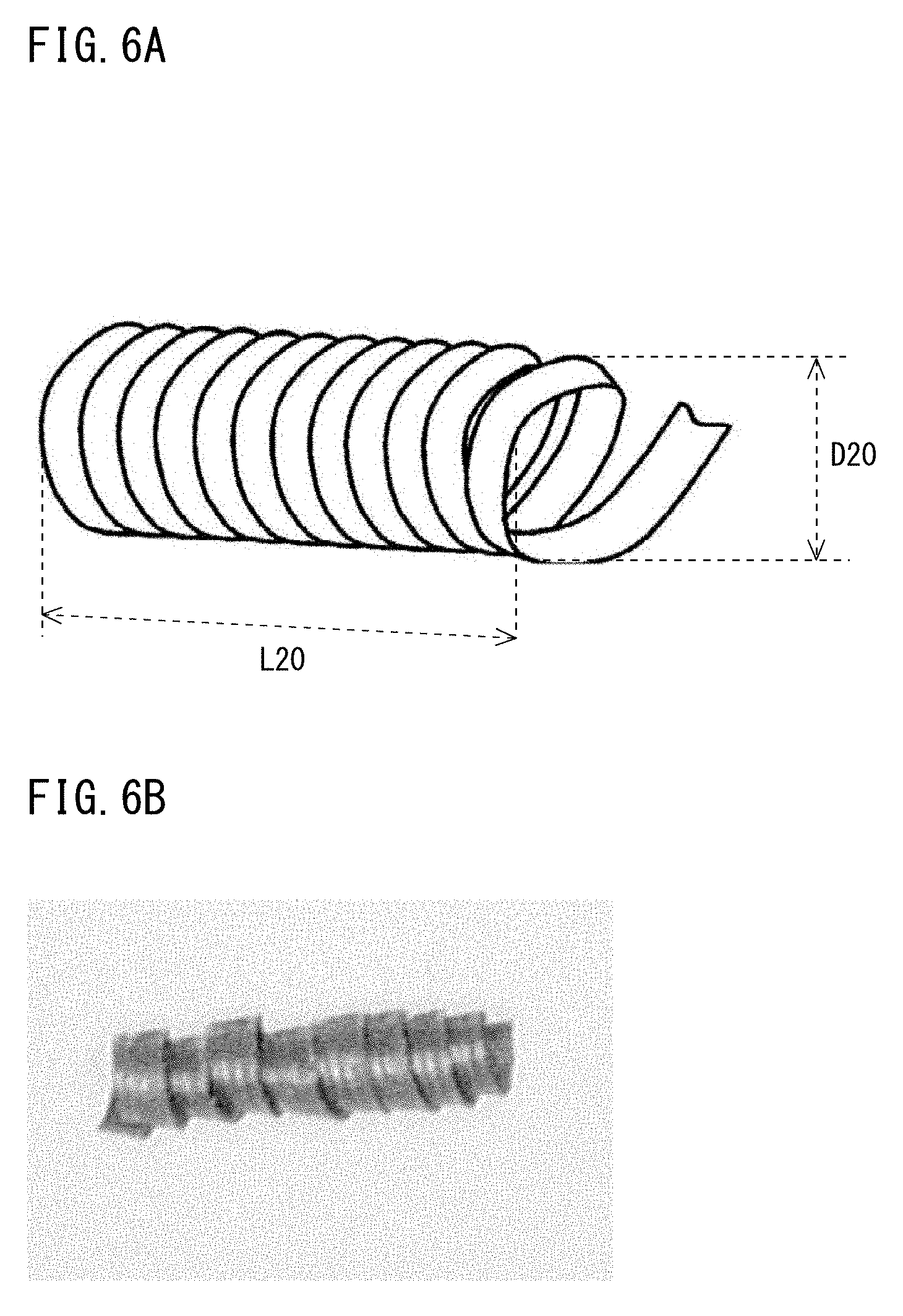
[0022] FIG. 6A is a perspective view of a chip.
[0023] FIG. 6B is a planar photographic view of a chip.
DESCRIPTION OF EMBODIMENTS
[0024] The present inventors conducted investigations and studies regarding the machinability, rusting characteristics and hot workability of steels for machine structural use. As a result, the present inventors found that if a steel for machine structural use has a chemical composition consisting of, in mass %, C: 0.30 to 0.80%, Si: 0.01 to 0.80%, Mn: 0.20 to 2.00%, P: 0.030% or less, S: 0.010 to 0.100%, Pb: 0.010 to 0.100%, Al: 0.010 to 0.050%, N: 0.015% or less, O: 0.0005 to 0.0030%, Cr: 0 to 0.70%, Ni: 0 to 3.50%, B: 0 to 0.0050%, V: 0 to 0.70%, Mo: 0 to 0.70%, W: 0 to 0.70%, Nb: 0 to less than 0.050%, Cu: 0 to 0.50%, Ti: 0 to 0.100%, and Ca: 0 to 0.0030%, with the balance being Fe and impurities, there is a possibility that excellent machinability and excellent hot workability are obtained.
[0025] Mn in the steel combines with S to form MnS. The MnS is divided into MnS inclusions and MnS precipitates according to the formation process. MnS inclusions crystallize in the molten steel before solidification. On the other hand, MnS precipitates precipitate in the steel after solidification. The MnS inclusions form in the molten steel. Therefore, the size of the MnS inclusions tends to be large in comparison to the MnS precipitates that form after solidification.
[0026] On the other hand, most of the Pb in the steel does not dissolve in the steel, and is present as Pb inclusions (Pb particles). The MnS inclusions and Pb inclusions each enhance the machinability of the steel.
[0027] In addition, in a case where Mn and Pb are present in the steel, in addition to the aforementioned MnS inclusions and Pb inclusions, the Mn and Pb also form composite inclusions containing MnS and Pb (hereinafter, also referred to simply as "composite inclusions"). The term "composite inclusions" means inclusions that contain MnS and Pb, with the balance being impurities. More specifically, there are cases where composite inclusions are composed by MnS and Pb that are adjacent to each other, and there are also cases where Pb dissolves into MnS to form a composite inclusion. In the present description, "MnS inclusions", "Pb inclusions" and "composite inclusions" are identified by a method described in the section "Method of measuring number TN of specific inclusions and composite ratio RA" that is described later. In the present description, the term "MnS inclusions" refers to inclusions that contain Mn and S and do not contain Pb. The term "Pb inclusions" refers to inclusions which are composed of Pb and impurities and which do not contain Mn. The term "composite inclusions" refers to inclusions that contain Mn, S and Pb.
[0028] MnS inclusions are known as inclusions that enhance machinability. On the other hand, the fusing point of Pb inclusions is lower than the fusing point of MnS inclusions. Therefore, Pb inclusions exert a lubricating action during cutting, and as a result the machinability of the steel is enhanced.
[0029] In addition, it is considered that composite inclusions enhance the machinability of steel more than individual MnS inclusions and Pb inclusions. In a case where a fissure has arisen at the periphery of a composite inclusion, liquefied Pb enters into the open crack. By this means, propagation of the crack is promoted and machinability is enhanced. Accordingly, if composite inclusions are also formed, and not just MnS inclusions and Pb inclusions, the machinability is enhanced further.
[0030] The mechanism by which composite inclusions are formed is considered to be as follows. It is easier for Pb to move in liquid phase than in solid phase. Therefore, almost no composite inclusions can be formed from MnS precipitates which form after solidification of the steel, and the composite inclusions are instead formed by adherence of Pb to MnS inclusions that are formed in the molten steel before solidification. Accordingly, in order to form a large number of composite inclusions, it is desirable to form a large number of MnS inclusions in the molten steel rather than forming MnS precipitates after solidification.
[0031] As described above, in order to enhance the machinability of steel, it suffices to form a large number of MnS inclusions, Pb inclusions and composite inclusions. As described above, MnS inclusions are formed in molten steel by crystallization. In addition, as described above, the greater the number of MnS inclusions that are present, the greater the number of composite inclusions that will be formed. Therefore, it is considered that the machinability of the steel is enhanced by causing a large number of MnS inclusions to crystallize in the molten steel.
[0032] On the other hand, a steel for machine structural use that contains MnS inclusions, MnS precipitates, Pb inclusions and composite inclusions is susceptible to rusting. However, hitherto the mechanism of rusting with respect to a steel for machine structural use had not been studied in detail. Therefore the present inventors conducted investigations and studies regarding the rusting mechanism. As a result, the present inventors obtained the following finding.
[0033] MnS inclusions, MnS precipitates, Pb inclusions and composite inclusions become starting points for rust. In this case, the susceptibility to rusting depends more on the total number of MnS inclusions, MnS precipitates, Pb inclusions and composite inclusions than the size of the MnS inclusions, MnS precipitates, Pb inclusions and composite inclusions. Specifically, the susceptibility to rusting of the steel increases as the total number of MnS inclusions, MnS precipitates, Pb inclusions and composite inclusions increases. Based on the above finding, the present inventors concluded that in order to suppress rusting while obtaining excellent machinability, it is effective to decrease the total number of MnS inclusions, MnS precipitates, Pb inclusions and composite inclusions. Therefore, the present inventors studied methods for decreasing the total number of MnS inclusions, MnS precipitates, Pb inclusions and composite inclusions.
[0034] As described above, MnS inclusions that are formed by crystallization in molten steel are liable to grow (coarsen) in the molten steel. Therefore, the size of MnS inclusions is larger than the size of MnS precipitates that are formed by precipitation in the steel after solidification. That is, the MnS precipitates precipitate more finely than the MnS inclusions. Therefore, in a steel having a certain Mn content and S content, if a case in which MnS inclusions are caused to crystallize and a case in which MnS precipitates are caused to precipitate are supposed, the number of MnS precipitates that are formed by precipitation will be noticeably greater than the number of MnS inclusions that are formed by crystallization. Accordingly, to improve the rusting characteristics of a steel, it suffices to suppress precipitation of MnS precipitates by crystallizing MnS inclusions in the molten steel and causing the MnS inclusions to grow (coarsen).
[0035] In order to cause MnS inclusions to crystallize and grow in molten steel and suppress precipitation of MnS precipitates and, as a result, decrease the total number of MnS inclusions, MnS precipitates, Pb inclusions and composite inclusions, it suffices to significantly increase the Mn content in comparison to the S content. If the Mn content is sufficiently higher than the S content, coarse MnS inclusions are likely to form in the molten steel. In this case, because S is consumed by the crystallization of the coarse MnS inclusions, the amount of dissolved S in the steel after solidification is lowered. Consequently, precipitation of MnS precipitates can be suppressed, and the total number of MnS inclusions, MnS precipitates, Pb inclusions and composite inclusions can be decreased. As a result, excellent rusting characteristics are obtained.
[0036] Specifically, the Mn content and S content satisfy the following Formula (1).
Mn/S.gtoreq.8.0 (1)
[0037] Where, the content (mass %) of a corresponding element is substituted for each symbol of an element in Formula (1).
[0038] Here, it is defined that F1=Mn/S. If F1 is less than 8.0, it is difficult for MnS inclusions to adequately crystallize in the molten steel. Therefore the amount of dissolved S in the steel after solidification cannot be adequately decreased, and a large number of fine MnS precipitates are formed after solidification. In this case, because the total number of MnS inclusions, MnS precipitates, Pb inclusions and composite inclusions cannot be decreased, the rusting characteristics of the steel decline. On the other hand, if F1 is 8.0 or more, the Mn content is adequately high in comparison with the S content. In this case, by using an appropriate production method, MnS inclusions in the molten steel adequately crystallize and grow. As a result, the amount of dissolved S in the steel after solidification is adequately decreased, and precipitation of MnS precipitates in the steel after solidification can be suppressed. Therefore, the total number of MnS inclusions, MnS precipitates, Pb inclusions and composite inclusions can be adequately reduced, and the rusting characteristics of the steel are enhanced.
[0039] Here, inclusions which are any of MnS inclusions, Pb inclusions and composite inclusions and which have an equivalent circular diameter of 5 .mu.m or more are defined as "specific inclusions". In the present description, the term "equivalent circular diameter" means the diameter of a circle in a case where the area of an inclusion or a precipitate that is observed during micro-structure observation is converted into a circle having the same area. In this case, in the present embodiment, in addition, in a steel for machine structural use having the aforementioned chemical composition and satisfying Formula (1), the total number of specific inclusions is 40 per mm.sup.2 or more.
[0040] If the total number of specific inclusions in the steel is 40 per mm.sup.2 or more, coarse MnS inclusions adequately crystallize and formation of MnS precipitates can be suppressed. As a result, the total number of MnS inclusions, MnS precipitates, Pb inclusions and composite inclusions which become starting points for rusting can be adequately reduced. Therefore, excellent machinability and excellent rusting characteristics can both be realized in a compatible manner. On the other hand, if the total number of specific inclusions in the steel is less than 40 per mm.sup.2, MnS inclusions do not adequately crystallize, and a large number of MnS precipitates form. As a result, the formation of MnS precipitates can be suppressed. Consequently, the total number of MnS inclusions, MnS precipitates, Pb inclusions and composite inclusions which become starting points for rusting cannot be adequately reduced. Therefore, although excellent machinability is obtained, adequate rusting characteristics are not obtained.
[0041] A steel for machine structural use according to the present embodiment that was completed based on the above findings has a chemical composition which consists of, in mass %, C: 0.30 to 0.80%, Si: 0.01 to 0.80%, Mn: 0.20 to 2.00%, P: 0.030% or less, S: 0.010 to 0.100%, Pb: 0.010 to 0.100%, Al: 0.010 to 0.050%, N: 0.015% or less, O: 0.0005 to 0.0030%, Cr: 0 to 0.70%, Ni: 0 to 3.50%, B: 0 to 0.0050%, V: 0 to 0.70%, Mo: 0 to 0.70%, W: 0 to 0.70%, Nb: 0 to less than 0.050%, Cu: 0 to 0.50%, Ti: 0 to 0.100% and Ca: 0 to 0.0030%, with the balance being Fe and impurities, the chemical composition satisfying Formula (1). In the steel, the total number of specific inclusions which are any of MnS inclusions, Pb inclusions, and composite inclusions containing MnS and Pb, and which have an equivalent circular diameter of 5 .mu.m or more is 40 per mm.sup.2 or more.
Mn/S.gtoreq.8.0 (1)
[0042] Where, the content (mass %) of a corresponding element is substituted for the respective elements in Formula (1).
[0043] The chemical composition of the steel for machine structural use that is described above may contain one or more types of element selected from a group consisting of Cr: 0.10 to 0.70%, Ni: 0.02 to 3.50%, B: 0.0005 to 0.0050%, V: 0.05 to 0.70%, Mo: 0.05 to 0.70%, W: 0.05 to 0.70%, Nb: 0.001 to less than 0.050%, Cu: 0.05 to 0.50% and Ti: 0.003 to 0.100%.
[0044] The chemical composition of the steel for machine structural use that is described above may contain Ca: 0.0001 to 0.0030%.
[0045] In the steel for machine structural use that is described above, a ratio of the number of the composite inclusions to the specific inclusions may be 40% or more.
[0046] Hereunder, the steel for machine structural use according to the present embodiment is described in detail. The symbol "%" in the chemical composition means "mass percent" unless specifically stated otherwise.
[0047] [Chemical Composition]
[0048] The chemical composition of the steel for machine structural use of the present embodiment contains the following elements.
[0049] C: 0.30 to 0.80%
[0050] Carbon (C) enhances the strength of the steel. When producing a component using a steel for machine structural use, as necessary, a heat treatment (normalizing or the like), a case hardening heat treatment (induction hardening or the like), or quenching and tempering are performed after forging the steel for machine structural use. In such a case, C increases the strength of the steel. If the C content is less than 0.30%, sufficient strength will not be obtained. On the other hand, if the C content is more than 0.80%, a large amount of retained austenite will form after tempering. In such a case, not only will an increase in the strength be saturated, but hard cementite will form and the machinability of the steel will decrease. Accordingly, the C content is in a range of 0.30 to 0.80%.
[0051] A preferable lower limit of the C content in the case of using a component as it is in a normalized state is 0.34%, and more preferably is 0.40%. In the case of performing quenching such as induction hardening, a preferable upper limit of the C content is 0.70%. In such a case, a strength that is commensurate with the C content is obtained. Further, in the steel for machine structural use of the present embodiment, a temperature region in which an austenite single-phase structure is formed during quenching is extremely narrow. Accordingly, in the case of large-scale production, a preferable upper limit of the C content is 0.60%.
[0052] Si: 0.01 to 0.80%
[0053] Silicon (Si) deoxidizes the steel. By adding Si after adding Mn at the time of deoxidation, the Si modifies oxides. Specifically, Si added to molten steel modifies oxides that are mainly composed of Mn into oxides that are mainly composed of Si. By adding Al after adding Si, composite oxides containing Si and Al form in the steel. The composite oxides serve as nuclei for crystallization of MnS inclusions. Therefore, the composite oxides enhance the rusting characteristics of the steel. Si also enhances temper softening resistance and raises the strength. The aforementioned effects are not obtained if the Si content is less than 0.01%.
[0054] On the other hand, Si is a ferrite forming element. If the Si content is more than 0.80%, the outer layer of the steel may be decarburized. Furthermore, if the Si content is more than 0.80%, the ferrite fraction may increase and the strength decrease in some cases. Accordingly, the Si content is from 0.01 to 0.80%. A preferable lower limit of the Si content for increasing the temper softening resistance is 0.10%, and more preferably is 0.20%. A preferable upper limit of the Si content for keeping the ferrite fraction low is 0.70%, and more preferably is 0.50%.
[0055] Mn: 0.20 to 2.00%
[0056] Manganese (Mn) forms MnS inclusions and composite inclusions containing MnS and Pb, and enhances the machinability of the steel.
[0057] Mn also deoxidizes the steel. The deoxidizing power of Mn is weak compared to Si or Al. Therefore, a large amount of Mn may be contained. In a case where another strong deoxidizing element is not present in the molten steel, oxides that are mainly composed of Mn form in the molten steel. Thereafter, if another strong deoxidizing element (Si or Al) is added to the molten steel, the Mn contained in the oxides is discharged into the molten steel, and the oxides are modified. Hereunder, the modified oxides are referred to as "composite oxides". The Mn that is discharged into the molten steel from the oxides combines with S to form MnS inclusions. Note that, composite oxides formed by modification of oxides easily become nuclei for crystallization of MnS inclusions. Therefore, if composite oxides are formed, crystallization of MnS inclusions is promoted. The MnS inclusions formed by crystallization easily form composite inclusions also.
[0058] If the Mn content is less than 0.20%, it is difficult for MnS inclusions to adequately crystallize. Therefore, a large number of MnS precipitates will form in the steel after solidification. In this case, the total number of MnS inclusions, MnS precipitates, Pb inclusions and composite inclusions will increase. Consequently, the rusting characteristics of the steel will decrease. In contrast, if the Mn content is more than 2.00%, the hardenability of the steel will be too high and, as a result, the hardness of the steel will be too high. In this case, the machinability of the steel will decrease. Accordingly, the Mn content is from 0.20 to 2.00%. A preferable lower limit of the Mn content is 0.50%. A preferable upper limit of the Mn content is 1.50%, and more preferably is 1.20%.
[0059] P: 0.030% or less
[0060] Phosphorus (P) is unavoidably contained. P embrittles the steel and enhances the machinability. On the other hand, if the P content is more than 0.030%, hot ductility decreases. In such a case, rolling defects and the like occur, and the productivity decreases. Accordingly, the P content is 0.030% or less. A preferable lower limit of the P content for enhancing the machinability is 0.005%. In this case, the machinability, particularly the chip treatability, is enhanced. A preferable upper limit of the P content is 0.015%.
[0061] S: 0.010 to 0.100%
[0062] Sulfur (S) forms MnS in the steel and enhances the machinability. In particular, MnS suppresses tool wear. If the S content is less than 0.010%, MnS will not crystallize adequately and it will be difficult for composite inclusions containing MnS and Pb to form. As a result, the rusting characteristics will decrease. On the other hand, if the S content is more than 0.100%, S will segregate at grain boundaries and the steel will become brittle, and the hot workability of the steel will decrease. Accordingly, the S content is from 0.010 to 0.100%. When prioritizing mechanical properties over machinability, a preferable lower limit of the S content is 0.015%, and a preferable upper limit is 0.030%. When prioritizing machinability, a preferable lower limit of the S content is 0.030%, and a preferable upper limit is 0.050%.
[0063] Pb: 0.010 to 0.100%
[0064] Lead (Pb) forms Pb inclusions (Pb particles) by itself, and enhances the machinability of the steel. Pb also combines with MnS inclusions to form composite inclusions and enhance the machinability of the steel, and in particular enhance the chip treatability. The aforementioned effects are not obtained if the Pb content is less than 0.010%. On the other hand, if the Pb content is more than 0.100%, although the machinability will be enhanced, the steel will become brittle. As a result, the hot workability of the steel will decrease. In addition, if the Pb content is more than 0.100%, because the Pb inclusions will excessively increase, the rusting characteristics of the steel will decrease. Accordingly, the Pb content is from 0.010 to 0.100%. A preferable lower limit of the Pb content for promoting the formation of composite inclusions and enhancing the machinability is 0.020%, and more preferably is 0.025%. A preferable upper limit of the Pb content for enhancing the rusting characteristics is 0.050%.
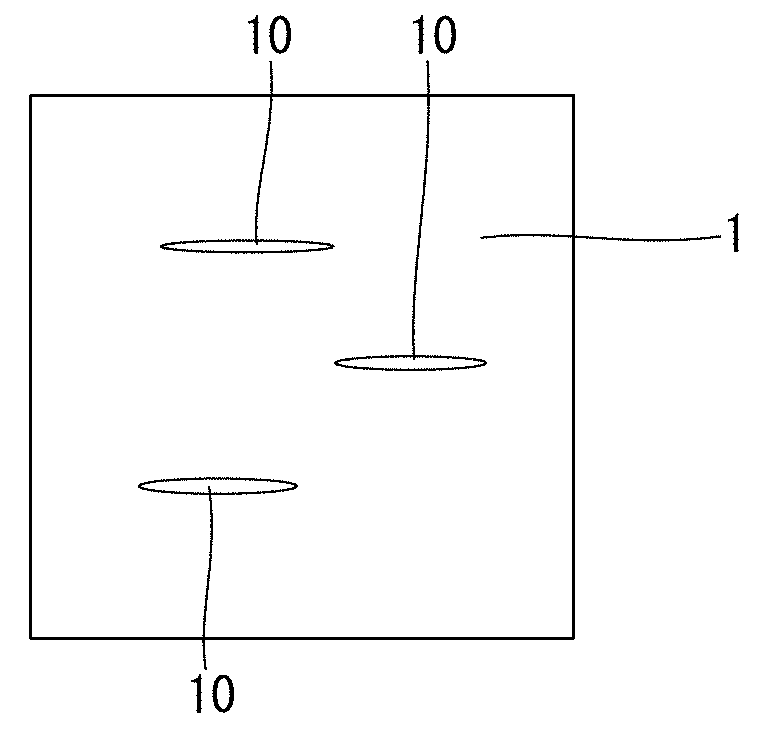
[0065] Al: 0.010 to 0.050%
[0066] Aluminum (Al) deoxidizes the steel. In the steel for machine structural use according to the present invention, in order to suppress the formation of holes and surface defects during solidification, deoxidation is performed by aluminum killing. As described later, if deoxidation is performed by adding Al into the molten steel after Mn and Si, oxides in the steel are modified and composite oxides containing Si and Al are formed. The composite oxides easily become nuclei for crystallization of MnS inclusions. Therefore, it is easy for MnS inclusions to disperse and crystallize, and to grow and coarsen, and it is also easy for composite inclusions containing MnS and Pb to form. In this case, the machinability of the steel improves. In addition, in a case where MnS inclusions disperse and crystallize, precipitation of fine MnS precipitates is suppressed. In this case, the total number of MnS inclusions, MnS precipitates, Pb inclusions and composite inclusions decreases. Consequently, the rusting characteristics of the steel improve. Al also combines with N to form AlN and thereby suppress coarsening of austenite grains in various kinds of heat treatment. The aforementioned effects are not obtained if the Al content is less than 0.010%.
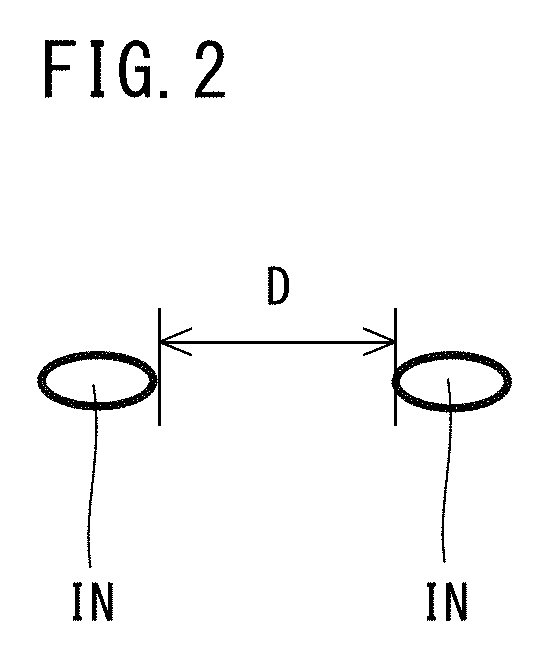
[0067] On the other hand, if the Al content is more than 0.050%, coarse composite oxides are liable to form. The composite oxides are liable to become coarse. If coarse composite oxides are formed in the steel, surface defects are liable to occur on the steel. If coarse composite oxides are formed in the steel, the fatigue strength of the steel will also decrease. In addition, if the Al content is more than 0.050%, deoxidation will proceed excessively, and the amount of oxygen in the molten steel will decrease. In this case, it will be difficult to form MnS inclusions, and the machinability (particularly, suppression of tool wear) of the steel will decrease. In such a case, in addition, it will be difficult for composite inclusions in which Pb is combined with MnS inclusions to form, and a large number of independent Pb inclusions will remain in the steel. As a result, the total number of MnS inclusions, MnS precipitates, Pb inclusions and composite inclusions will increase, and the rusting characteristics will decrease. Accordingly, the Al content is from 0.010 to 0.050%. A preferable lower limit of the Al content for obtaining a further effect of suppressing the coarsening of grains by formation of AlN is 0.015%, and more preferably is 0.020%. A preferable upper limit of the Al content is 0.035%. In the present description, the term "Al content" means the content of acid-soluble Al (sol. Al).
[0068] N: 0.015% or less
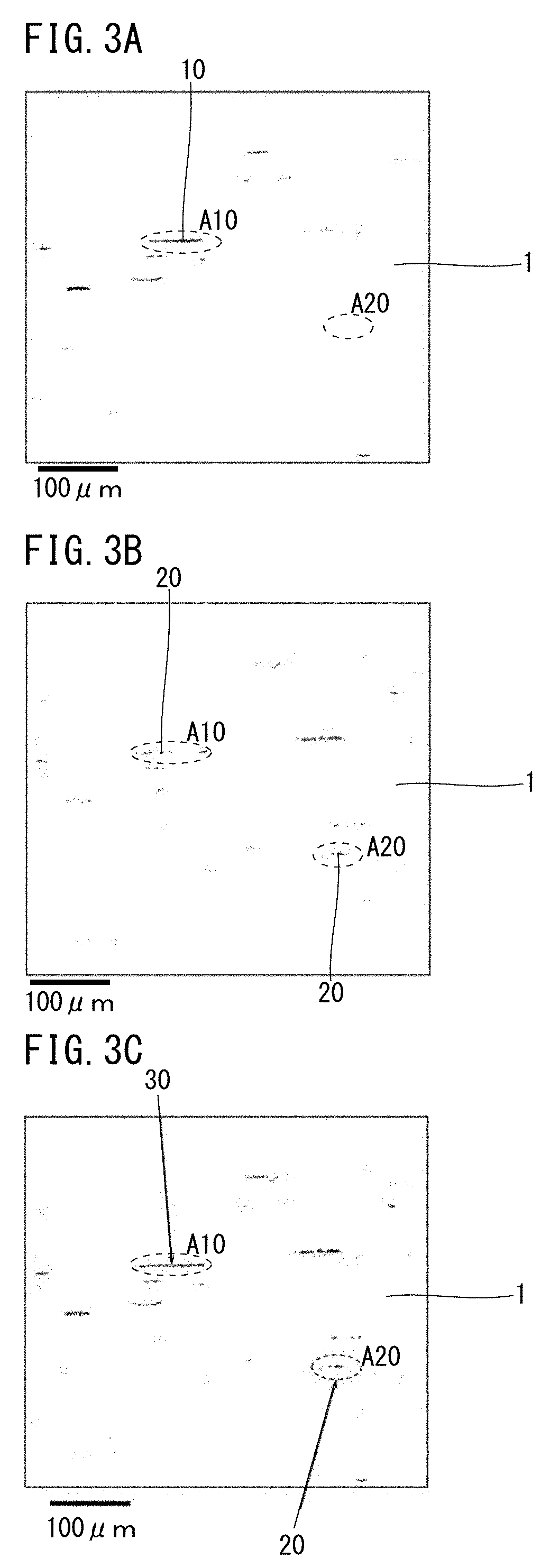
[0069] Nitrogen (N) is unavoidably contained. N combines with Al to form AlN to thereby suppress coarsening of austenite grains during heat treatment and enhance the strength of the steel. On the other hand, if the N content is more than 0.015%, the cutting resistance of the steel increases and the machinability decreases. If the N content is more than 0.015%, the hot workability also decreases. Accordingly, the N content is 0.015% or less. A preferable lower limit of the N content is 0.002%, and more preferably is 0.004%. A preferable upper limit of the N content is 0.012%, and more preferably is 0.008%. In the present description, the term "N content" means the total content of N (t-N).
[0070] O: 0.0005 to 0.0030%
[0071] Oxygen (O) is contained not only in oxides, but also in MnS inclusions. O forms composite oxides that serve as nuclei for crystallization of MnS inclusions. If the O content is less than 0.0005%, the formed amount of composite oxides will be insufficient, and it will be difficult for MnS inclusions to crystallize in the molten steel. In such a case, the machinability of the steel will decrease. Furthermore, in such a case, a large number of fine MnS precipitates will form after solidification. As a result, the total number of MnS inclusions, MnS precipitates, Pb inclusions and composite inclusions will increase, and rusting characteristics will decrease. On the other hand, if the O content is more than 0.0030%, coarse alumina-based oxides will form and promote cutting wear of the tools, and hence the machinability of the steel will decrease. Furthermore, if the O content is more than 0.0030%, in some cases coarse oxides that become the starting points for fractures are formed. In this case, the rolling contact fatigue properties of the machine component decrease. Accordingly, the O content is from 0.0005 to 0.0030%. A preferable lower limit of the O content for further improving the machinability of the steel as well as the rusting characteristics of the steel is 0.0007%, and more preferably is 0.0010%. A preferable upper limit of the O content is 0.0025%, and more preferably is 0.0020%. In the present description, the term "O content" means the total content of oxygen (t-O).
[0072] The balance of the chemical composition of the steel for machine structural use according to the present embodiment is Fe and impurities. Here, the term "impurities" refers to elements which, during industrial production of the steel for machine structural use, are mixed in from ore or scrap that is used as a raw material, or from the production environment or the like, and which are allowed within a range that does not adversely affect the steel for machine structural use of the present embodiment.
[0073] [Optional Elements]
[0074] The chemical composition of the steel for machine structural use of the present embodiment may further contain one or more types of element selected from the group consisting of Cr, Ni, B, V, Mo, W, Nb, Cu and Ti.
[0075] Cr: 0 to 0.70%
[0076] Chromium (Cr) is an optional element and need not be contained. If contained, Cr dissolves in the steel and increases the hardenability and temper softening resistance of the steel and enhances the steel strength. In a case where nitriding is performed as a case hardening treatment, Cr also deepens the hardened layer depth. The aforementioned effects are obtained to a certain extent if even a small amount of Cr is contained. On the other hand, if the Cr content is more than 0.70%, if quenching and tempering are performed, cementite in the steel will coarsen. In addition, if the Cr content is more than 0.70%, if induction hardening is performed, cementite in the steel will not dissolve. Furthermore, if the Cr content is more than 0.70%, austenite will stabilize at even a low temperature. In such a case, the steel will become brittle. Accordingly, the Cr content is in a range from 0 to 0.70%. A preferable lower limit of the Cr content for increasing hardenability is 0.10%, and more preferably is 0.30%. A preferable upper limit of the Cr content is 0.60%.
[0077] Ni: 0 to 3.50%
[0078] Nickel (Ni) is an optional element and need not be contained. If contained, Ni dissolves in the steel and increases the hardenability of the steel, and enhances the steel strength. Ni also improves the ductility of the matrix. In addition, Ni increases the toughness of the steel. Furthermore, Ni increases the corrosion resistance of the steel. The aforementioned effects are obtained to a certain extent if even a small amount of Ni is contained. On the other hand, if the Ni content is more than 3.50%, a large amount of retained austenite will remain. In such a case, a part of the retained austenite will transform into martensite by strain induced transformation, and the ductility of the steel will decrease. Accordingly, the Ni content is from 0 to 3.50%.
[0079] A preferable lower limit of the Ni content for stably obtaining the aforementioned effects is 0.02%, and more preferably is 0.05%. A preferable upper limit of the Ni content for further suppressing the formation of retained austenite is 2.50%, and more preferably is 2.00%. When prioritizing toughness, a preferable lower limit of the Ni content is 0.20%. Note that, Ni detoxifies Cu and enhances the toughness. If the steel contains Cu, a preferable lower limit of the Ni content is equal to or more than the Cu content.
[0080] B: 0 to 0.0050%
[0081] Boron (B) is an optional element and need not be contained. If contained, B increases the hardenability of the steel and increases the steel strength. B also suppresses segregation at the grain boundaries of P and S that decrease toughness, and thus enhances the fracture characteristics. The aforementioned effects are obtained to a certain extent if even a small amount of B is contained. On the other hand, if the B content is more than 0.0050%, a large amount of BN will be formed and the steel will become brittle. Accordingly, the B content is from 0 to 0.0050%. A preferable lower limit of the B content in a case where Ti or Nb that are nitride-forming elements is contained is 0.0005%. A preferable upper limit of the B content is 0.0020%.
[0082] V: 0 to 0.70%
[0083] Vanadium (V) is an optional element and need not be contained. If contained, V precipitates as carbides, nitrides, or carbo-nitrides during tempering and during nitriding, and enhances the strength of the steel. The V precipitates (nitrides, carbides and carbo-nitrides) also suppress coarsening of austenite grains and increase the toughness of the steel. In addition, V dissolves in the steel and thereby increases the temper softening resistance of the steel. The aforementioned effects are obtained to a certain extent if even a small amount of V is contained.
[0084] On the other hand, if the V content is more than 0.70%, V precipitates will form even at a temperature equal to or higher than the A.sub.3 point. It is difficult for V precipitates that are formed at a temperature equal to or higher than the A.sub.3 point to dissolve in the steel, and such V precipitates remain in the steel as insoluble precipitates. When insoluble precipitates remain, the amount of dissolved V decreases. Consequently, the temper softening resistance of the steel decreases. In addition, when insoluble precipitates remain, it is difficult for fine V precipitates to precipitate by means of a heat treatment conducted thereafter. In such a case, the strength of the steel decreases. Accordingly, the V content is from 0 to 0.70%. A preferable lower limit of the V content for stably obtaining the aforementioned effects is 0.05%, and more preferably is 0.10%. A preferable upper limit of the V content is 0.50%, and more preferably is 0.30%.
[0085] Mo: 0 to 0.70%
[0086] Molybdenum (Mo) is an optional element and need not be contained. If contained, Mo precipitates as Mo carbides during a heat treatment at a low temperature that is not more than the A.sub.1, such as a heat treatment for tempering or nitriding. Therefore, the strength and temper softening resistance of the steel increase. Mo also dissolves in the steel and increases the hardenability of the steel. The aforementioned effects are obtained to a certain extent if even a small amount of Mo is contained. On the other hand, if the Mo content is more than 0.70%, the hardenability of the steel will be too high. In such a case, a supercooled structure is liable to form during rolling or a softening heat treatment before wire drawing or the like. Accordingly, the Mo content is from 0 to 0.70%.
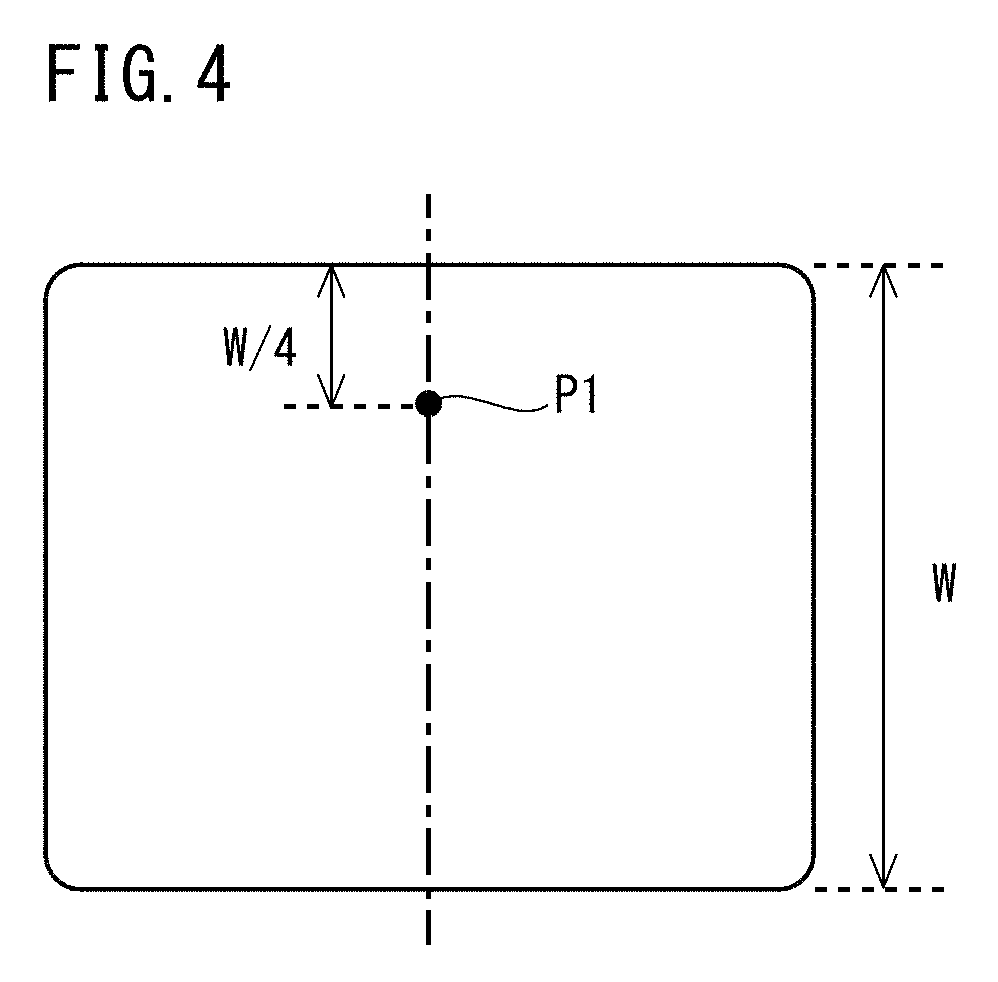
[0087] A preferable lower limit of the Mo content for stably obtaining the aforementioned effects is 0.05%, more preferably is 0.10%, and further preferably is 0.15%. A preferable upper limit of the Mo content for stably obtaining ferrite-pearlite structure is 0.40%, and more preferably is 0.30%.
[0088] W: 0 to 0.70%
[0089] Tungsten (W) is an optional element and need not be contained. If contained, W precipitates as W carbides in the steel and enhances the strength and temper softening resistance of the steel. W carbides form at a low temperature that is not more than the A.sub.3 point. Therefore, unlike V, Nb, Ti and the like, it is difficult for W to form insoluble precipitates. Consequently, W carbides increase the strength and temper softening resistance of the steel by precipitation strengthening. W also dissolves in the steel and thereby increases the hardenability of the steel and increases the steel strength. The aforementioned effects are obtained to a certain extent if even a small amount of W is contained.
[0090] On the other hand, if the W content is more than 0.70%, a supercooled structure is liable to form, and the hot workability of the steel will thus decrease. Accordingly, the W content is from 0 to 0.70%. A preferable lower limit of the W content for stably increasing the temper softening resistance of the steel is 0.05%, and more preferably is 0.10%. A preferable upper limit of the W content for stably obtaining ferrite-pearlite structure is 0.40%, and more preferably is 0.30%.
[0091] It is difficult for W and Mo to form nitrides. Therefore, these elements can enhance the temper softening resistance of the steel without being influenced by the N content. A preferable total content of W and Mo for obtaining a high temper softening resistance is from 0.10 to 0.30%.
[0092] Nb: 0 to less than 0.050%
[0093] Niobium (Nb) is an optional element and need not be contained. If contained, Nb forms nitrides, carbides, or carbo-nitrides and suppresses coarsening of austenite grains during quenching or during normalizing. Nb also increases the strength of the steel by precipitation strengthening. The aforementioned effects are obtained to a certain extent even if a small amount of Nb is contained. On the other hand, if the Nb content is more than 0.050%, insoluble precipitates form and the toughness of the steel decreases. In addition, if the Nb content is more than 0.050%, a supercooled structure is liable to form and consequently the hot workability of the steel will decrease. Accordingly, the Nb content is from 0 to less than 0.050%. A preferable lower limit of the Nb content for stably obtaining the aforementioned effects is 0.001%, and more preferably is 0.005%. A preferable upper limit of the Nb content is 0.030%, and more preferably is 0.015%.
[0094] Cu: 0 to 0.50%
[0095] Copper (Cu) is an optional element and need not be contained. If contained, Cu prevents decarburization. Cu also increases corrosion resistance, similarly to Ni. The aforementioned effects are obtained to a certain extent if even a small amount of Cu is contained. On the other hand, if the Cu content is more than 0.50%, the steel will become brittle and rolling defects are liable to arise. Accordingly, the Cu content is from 0 to 0.50%. A preferable lower limit of the Cu content for stably obtaining the aforementioned effects is 0.05%, and more preferably is 0.10%. In a case where 0.30% or more of Cu is contained, the hot ductility can be maintained if the Ni content is higher than the Cu content.
[0096] Ti: 0 to 0.100%
[0097] Titanium (Ti) is an optional element and need not be contained. If contained, Ti forms nitrides, carbides or carbo-nitrides, and suppresses coarsening of austenite grains during quenching and during normalizing. Ti also increases the strength of the steel by precipitation strengthening. Ti also deoxidizes the steel. In addition, in a case where B is contained, Ti combines with dissolved N and maintains the amount of dissolved B. In this case, the hardenability increases. The aforementioned effects are obtained to a certain extent if even a small amount of Ti is contained.
[0098] On the other hand, because Ti forms the aforementioned nitrides and sulfides, Ti influences MnS inclusions and composite inclusions. Specifically, if the Ti content is more than 0.100%, the crystallized amount of MnS inclusions decreases, and formation of composite inclusions also decreases. In this case, the rusting characteristics of the steel decrease. In addition, if the Ti content is too high, the Ti forms nitrides and sulfides, and the fatigue strength decreases. Accordingly, the Ti content is from 0 to 0.100%. A preferable lower limit of the Ti content for effectively obtaining the aforementioned effects is 0.003%. In particular, in a case where B is contained, a preferable lower limit of the Ti content for reducing dissolved N is 0.005%. A preferable upper limit of the Ti content for increasing corrosion resistance is 0.090%, and more preferably is 0.085%.
[0099] The steel for machine structural use of the present embodiment may further contain Ca.
[0100] Ca: 0 to 0.0030%
[0101] Calcium (Ca) is an optional element and need not be contained. If contained, Ca forms CaS or (Mn, Ca)S and spheroidizes MnS inclusions, and reduces the amount of tool wear. As a result, the machinability of the steel increases. The aforementioned effects are obtained to a certain extent if even a small amount of Ca is contained. On the other hand, if the Ca content is more than 0.0030%, oxide-based inclusions coarsen and the fatigue strength of the steel decreases. Accordingly, the Ca content is from 0 to 0.0030%. A preferable lower limit of the Ca content for further enhancing the machinability is 0.0001%. When prioritizing fatigue strength over machinability, a preferable upper limit of the Ca content is 0.0015%, and more preferably is 0.0003%.
[0102] [Regarding Formula (1)]
[0103] The chemical composition of the steel for machine structural use of the present embodiment also satisfies Formula (1).
Mn/S.gtoreq.8.0 (1)
[0104] Where, a content (mass %) of a corresponding element is substituted for each element in Formula (1).
[0105] Here, it is defined that F1=Mn/S. F1 means the Mn content relative to the S content. If F1 is less than 8.0, it will be difficult for MnS inclusions to adequately crystallize. Consequently, the amount of dissolved S in the steel after solidification will not adequately decrease, and a large number of fine MnS precipitates will form after solidification. In such a case, the rusting characteristics of the steel will decrease because the total number of MnS inclusions, MnS precipitates, Pb inclusions and composite inclusions cannot decrease. If the amount of dissolved S in the steel after solidification cannot be adequately decreased, dissolved S will remain at crystal grain boundaries after solidification. As a result, in some cases the hot workability of the steel will decrease.
[0106] On the other hand, if F1 is 8.0 or more, the Mn content will be adequately high in comparison to the S content. In this case, MnS inclusions in the molten steel will adequately crystallize and grow. As a result, the amount of dissolved S in the steel after solidification will be adequately decreased, and precipitation of MnS precipitates in the steel after solidification can be suppressed. Therefore, the total number of MnS inclusions, MnS precipitates, Pb inclusions and composite inclusions in the steel can be adequately reduced, and the rusting characteristics of the steel improve. A preferable lower limit of F1 for improving the rusting characteristics of the steel is 10.0, and more preferably is 20.0.
[0107] [Regarding the Steel Micro-Structure]
[0108] The micro-structure of the steel for machine structural use according to the present invention is mainly composed of ferrite and pearlite. Specifically, a total area fraction of ferrite and pearlite in the micro-structure of the steel for machine structural use having the aforementioned chemical composition is 99% or more.
[0109] The total area fraction of ferrite and pearlite in the micro-structure can be measured by the following method. A sample is taken from the steel for machine structural use. For example, in a case where the steel for machine structural use is a steel bar or a wire rod, in a transverse section (a face perpendicular to the axial direction), a sample is taken from a middle part of a radius R (hereunder, referred to as "R/2 part") that links the external surface and the central axis. Of the entire area of a transverse section (surface) of the sample of the R/2 part, a surface that is perpendicular to the central axis of the steel for machine structural use is adopted as an observation surface. After polishing the observation surface, the observation surface is subjected to etching using 3% nitric acid-alcohol (nital etching reagent). The etched observation surface is observed with an optical microscope having a magnification of .times.200, and photographic images of an arbitrary five visual fields are generated.
[0110] In each visual field, the contrast differs for each of the respective phases of ferrite, pearlite, bainite and the like. Accordingly, the respective phases are identified based on the contrast. The total area (.mu.m.sup.2) of ferrite and pearlite among the identified phases is determined for each visual field. The total area in the respective visual fields is totaled for all of the visual fields (five visual fields), and the ratio relative to the gross area of all the visual fields is determined. The determined ratio is defined as the total area fraction (%) of ferrite and pearlite.
[0111] [Number TN of Specific Inclusions]
[0112] According to the steel for machine structural use of the present invention, a total number TN of inclusions (that is, specific inclusions) which are any of MnS inclusions, Pb inclusions and composite inclusions containing MnS and Pb and which have an equivalent circular diameter of 5 .mu.m or more in the steel is 40 per mm.sup.2 or more.
[0113] If the number TN of specific inclusions is 40 per mm.sup.2 or more, coarse MnS inclusions having an equivalent circular diameter of 5 .mu.m or more will adequately crystallize, and as a result the total number of MnS inclusions, MnS precipitates, Pb inclusions and composite inclusions can be adequately reduced. Therefore, excellent machinability and excellent rusting characteristics can both be realized in a compatible manner. On the other hand, if the number TN of specific inclusions in the steel is less than 40 per mm.sup.2, coarse MnS inclusions having an equivalent circular diameter of 5 .mu.m or more do not adequately crystallize, and as a result the total number of MnS inclusions, MnS precipitates, Pb inclusions and composite inclusions cannot be adequately reduced. Therefore, adequate rusting characteristics are not obtained. A preferable lower limit of the number TN of specific inclusions is 80 per mm.sup.2, and more preferably is 150 per mm.sup.2. A preferable upper limit of the number TN of specific inclusions is 300 per mm.sup.2. Note that, although an upper limit of the equivalent circular diameter of the specific inclusions is not particularly limited, for example, the upper limit is 200 .mu.m.
[0114] [Ratio of Number of Composite Inclusions Among Specific Inclusions (Composite Ratio) RA]
[0115] Preferably, a ratio (hereunder, also referred to as "composite ratio") RA of the total number (number per mm.sup.2) of composite inclusions having an equivalent circular diameter of 5 .mu.m or more with respect to the number (number per mm.sup.2) of specific inclusions is 40% or more.
[0116] As described above, the susceptibility of the steel to rusting increases as the total number of MnS inclusions, MnS precipitates, Pb inclusions and composite inclusions increases. In this case, the larger the number of composite inclusions that the MnS inclusions and Pb inclusions form, the more that the total number of MnS inclusions, MnS precipitates, Pb inclusions and composite inclusions can be reduced. In particular, the total number of Pb inclusions in the steel can be reduced. Pb inclusions, in particular, are liable to decrease the rusting characteristics. If the composite ratio is 40% or more, the total number of MnS inclusions, MnS precipitates, Pb inclusions and composite inclusions can be reduced, and the number of Pb inclusions that are independently present can also be reduced. As a result, the rusting characteristics of the steel can be further enhanced. Accordingly, the composite ratio RA is preferably 40% or more. In this case, the rusting characteristics of the steel can be further enhanced. A more preferable lower limit of the composite ratio RA is 60%, and further preferably is 75%.
[0117] [Method of Measuring Number TN of Specific Inclusions and Composite Ratio RA]
[0118] The number TN of specific inclusions and the composite ratio RA can be measured by the following methods. A sample is taken from the steel for machine structural use by the method described above. Using a scanning electron microscope (SEM), 20 visual fields at a transverse section (surface) of the sample of the R/2 part are randomly observed at a magnification of .times.1000. Specific inclusions (any of MnS inclusions, Pb inclusions and composite inclusions for which an equivalent circular diameter is 5 .mu.m or more) are identified in the respective visual fields (referred to as "observation surfaces"). It is possible to distinguish specific inclusions and other inclusions based on contrast. In addition, among the specific inclusions, MnS inclusions, Pb inclusions and composite inclusions are respectively identified by the following method.
[0119] For each observation surface, an image of the S distribution and Pb distribution in the observation surface is obtained by means of an X-ray spectroscopy wavelength dispersion device (EPMA). FIG. 1A is a schematic diagram illustrating the S distribution in an observation surface, which was obtained by EPMA analysis. FIG. 1B is a schematic diagram illustrating the Pb distribution in the same observation surface as in FIG. 1A, which was obtained by EPMA analysis.
[0120] Reference numeral 10 in FIG. 1A denotes a region in which S is present. Because S is almost entirely present as MnS, MnS can be regarded as being present at the locations indicated by each reference numeral 10 in FIG. 1A. Reference numeral 20 in FIG. 1B denotes a region in which Pb is present.
[0121] As illustrated in FIG. 1B, in some cases, as shown by reference numeral 20A, Pb is divided by rolling or the like and is arranged in the rolling direction. The same applies with respect to S. As illustrated in FIG. 2, in an image obtained by EPMA analysis, in a case where adjacent inclusions IN each have an equivalent circular diameter of 5 .mu.m or more, if a distance D between the adjacent inclusions IN is not more than 10 .mu.m, these inclusions IN are regarded as a single inclusion. Note that, as described above, the term "equivalent circular diameter" means the diameter of a circle in a case where the area of the respective inclusions or respective precipitates is converted into a circle that has the same area. Even when an inclusion group is defined as a single inclusion, the equivalent circular diameter is the diameter of a circle having the same total area as the inclusion group.
[0122] FIG. 1C is an image obtained by combining FIG. 1B with FIG. 1A. Referring to FIG. 1C, in the case where the Pb inclusions 20 overlap with the MnS inclusions 10, the relevant inclusions are recognized as being composite inclusions 30. On the other hand, referring to FIG. 1C, in the case where the MnS inclusion 10 and the Pb inclusion 20 do not overlap (region A.sub.1 and region A2 in FIG. 1C), the relevant inclusions are identified as an MnS inclusion and a Pb inclusion.
[0123] FIG. 3A is a photographic image of an S distribution obtained by performing EPMA analysis on the steel for machine structural use of the present embodiment, and FIG. 3B is a photographic image of the Pb distribution. FIG. 3C is a photographic image obtained by superposing the images in FIG. 3A and FIG. 3B. Referring to FIG. 3A to FIG. 3C. The MnS inclusions 10 are observed in an area A10 in FIG. 3A, and the Pb inclusions 20 are observed in the area A10 in FIG. 3B. Accordingly, it can be recognized that the composite inclusions 30 are present in the area A10 in FIG. 3C. Further, the MnS inclusions 10 are not observed in an area A20 in FIG. 3A, and the Pb inclusions 20 are observed in the area A20 in FIG. 3B. Therefore, it can be recognized that the inclusions present in the area A20 in FIG. 3C are the Pb inclusions 20.
[0124] By the above method, MnS inclusions, Pb inclusions and composite inclusions are identified using a scanning microscope and EPMA. The area of each inclusion that is identified is determined, and the diameter of a circle with the same area is determined as the equivalent circular diameter (.mu.m) for each of the inclusions.
[0125] Among the respective inclusions, specific inclusions for which the equivalent circular diameter is 5 .mu.m or more are identified. The total number (number in 20 visual fields) of the specific inclusions that are identified is determined, and is converted to a number TN per mm.sup.2 (inclusions/mm.sup.2). The number TN of specific inclusions is determined by the above method. In addition, among the identified specific inclusions, a number MN of composite inclusions (inclusions/mm.sup.2) for which the equivalent circular diameter is 5 .mu.m or more is determined, and the composite ratio RA (%) is determined based on the following Formula (2).
RA=MN/TN.times.100 (2)
[0126] [Production Method]
[0127] An example of a method for producing the steel for machine structural use according to the present invention will now be described. According to the present embodiment, a method for producing a steel bar or a wire rod as an example of the steel for machine structural use will be described. However, a steel for machine structural use according to the present invention is not limited to a steel bar or a wire rod.
[0128] One example of the production method includes a steel making process of refining and casting molten steel to produce a starting material (a cast piece or an ingot), and a hot working process of subjecting the starting material to hot working to produce a steel for machine structural use. Hereunder, each of these processes is described.
[0129] [Steel Making Process]
[0130] The steel making process includes a refining process and a casting process.
[0131] [Refining Process]
[0132] In the refining process, firstly, hot metal that was produced by a well-known method is subjected to refining (primary refining) using a converter. Molten steel that was tapped from the converter is subjected to secondary refining. In the secondary refining, an alloy whose components have been adjusted is added to the molten steel to thereby produce a molten steel having the aforementioned chemical composition.
[0133] Specifically, Mn is added to the molten steel that was tapped from the converter. As a result, oxides that are mainly composed of Mn form in the molten steel. After addition of the Mn is completed, Si which has a stronger deoxidizing power than Mn is added. As a result, the oxides that are mainly composed of Mn are modified to oxides that are mainly composed of Si. After addition of the Si is completed, Al which has an even stronger deoxidizing power than Si is added. As a result, the oxides that are mainly composed of Si are modified to composite oxides containing Si and Al (hereinafter, also referred to simply as "composite oxides").
[0134] The composite oxides that were formed by the above described refining process serve as nuclei for crystallization of MnS inclusions. Therefore, by forming the composite oxides, MnS inclusions adequately crystallize and grow coarse. That is, if composite oxides form, it is easy for specific inclusions that are inclusions having an equivalent circular diameter of 5 .mu.m or more to form, and the number TN of specific inclusions becomes 40 per mm.sup.2 or more. As a result, the amount of dissolved S in the steel after solidification is adequately reduced, and precipitation of MnS precipitates in the steel after solidification can be suppressed. Therefore, the total number of MnS inclusions, MnS precipitates, Pb inclusions and composite inclusions can be adequately reduced, and the rusting characteristics of the steel are enhanced.
[0135] A well-known removing slag is performed after performing the deoxidation. After the removing slag, secondary refining is performed. For example, composite refining is performed as the secondary refining. For example, first a primary treatment that uses an LF (ladle furnace) or VAD (vacuum arc degassing) is performed. In addition an RH (Ruhrstahl-Hausen) vacuum degassing treatment may be performed. In the secondary refining, Mn, Si, and other elements are added as necessary to adjust the components of the molten steel. After adjusting the components of the molten steel, a casting process is performed.
[0136] [Casting Process]
[0137] A starting material (a cast piece or an ingot) is produced using the molten steel produced by the above described refining process. Specifically, a cast piece is produced by a continuous casting process using the molten steel. Alternatively, an ingot may be produced by an ingot-making process using the molten steel. Hereinafter, a cast piece and an ingot are referred to generically as "starting material". A cross-sectional area of the starting material in this case is, for example, 200 to 350 mm.times.200 to 600 mm.
[0138] A solidification cooling rate RC during casting is 100.degree. C./min or less. If the solidification cooling rate RC is 100.degree. C./min or less, MnS inclusions adequately crystallize and grow in the molten steel. Therefore, it is easy for specific inclusions to form, and the number TN thereof becomes 40 per mm.sup.2 or more. As a result, the amount of dissolved S in the steel after solidification is adequately reduced, and precipitation of MnS precipitates in the steel after solidification can be suppressed. Therefore, the total number of MnS inclusions, MnS precipitates, Pb inclusions and composite inclusions can be adequately reduced, and the rusting characteristics of the steel are enhanced.
[0139] On the other hand, if the solidification cooling rate RC is more than 100.degree. C./min, MnS inclusions do not adequately crystallize, and MnS inclusions also do not adequately grow. Therefore, it will be difficult for specific inclusions to be formed, and the number TN of specific inclusions will be less than 40 per mm.sup.2. In this case, the amount of dissolved S in the steel after solidification cannot be adequately reduced, and a large number of fine MnS precipitates will form after solidification. As a result, since the total number of MnS inclusions, MnS precipitates, Pb inclusions and composite inclusions cannot be reduced, the rusting characteristics of the steel will decline. Accordingly, the solidification cooling rate RC is 100.degree. C./min or less.
[0140] A preferable solidification cooling rate RC is from 8 to less than 50.degree. C./min. In this case, it is even easier for MnS inclusions to crystallize and grow. Furthermore, if the solidification cooling rate RC is from 8 to less than 50.degree. C./min, because the time period until solidifying is long, a sufficient time period for Pb to move through the molten steel and adhere to MnS inclusions can be secured. Therefore, it is easy for composite inclusions containing MnS and Pb to form, and the composite ratio RA becomes 40% or more. A more preferable upper limit of the solidification cooling rate RC is 30.degree. C./min. A more preferable lower limit of the solidification cooling rate RC is 10.degree. C./min, and further preferably is 15.degree. C./min.
[0141] The solidification cooling rate RC can be determined based on the starting material that was cast. FIG. 4 is a transverse sectional view of a starting material that was cast. In the starting material having a thickness W (mm), at a point P1 located at a position at a depth of W/4 towards the center of the starting material from the surface, the cooling rate from the liquidus temperature to the solidus temperature is defined as the solidification cooling rate RC (.degree. C./min) in the casting process. The solidification cooling rate RC can be determined by the following method. After solidification, the starting material is cut in the transverse direction. In the transverse section of the starting material, a secondary dendrite arm spacing .lamda.2 (.mu.m) in the thickness direction of the solidification structure at the point P1 is measured. Using the measurement value .lamda.2, the solidification cooling rate RC (.degree. C./min) is determined based on the following Formula (3).
RC=(.lamda.2/770).sup.-(1/0.41) (3)
[0142] The secondary dendrite arm spacing .lamda.2 depends on the solidification cooling rate RC. Accordingly, the solidification cooling rate RC can be determined by measuring the secondary dendrite arm spacing .lamda.2.
[0143] [Hot Working Process]
[0144] In the hot working process, hot working is usually performed one or a plurality of times. The starting material is heated before each hot working operation is performed. Thereafter, the starting material is subjected to the hot working. The hot working is, for example, hot forging or hot rolling. In the case of performing hot working a plurality of times, the initial hot working is, for example, blooming or hot forging, and the next hot working is finish rolling using a continuous mill. In the hot rolling mill, a horizontal stand having a pair of horizontal rolls, and a vertical stand having a pair of vertical rolls are alternately arranged in a row. The starting material after hot working is cooled by a well-known cooling method such as air cooling.
[0145] The steel for machine structural use according to the present embodiment is produced by the above described processes. The steel for machine structural use is, for example, a steel bar or a wire rod.
[0146] The steel for machine structural use produced by the above described method is excellent in machinability and rusting characteristics. Production of the steel for machine structural use into a machine component is performed, for example, by the following method.
[0147] The steel for machine structural use is subjected to hot forging to produce an intermediate product having a rough shape. As necessary, the intermediate product is subjected to a normalizing treatment. The intermediate product is also subjected to machining. The machining is, for example, cutting. The intermediate product that underwent machining may be subjected to a thermal refining treatment (quenching and tempering). In a case where a thermal refining treatment is performed, the machining such as cutting may be performed on the intermediate product after the thermal refining treatment. A machine component is produced by the above process. A machine component may also be produced by performing cold forging instead of hot forging.
EXAMPLES
[0148] Molten steels having the chemical compositions shown in Table 1 were produced.
TABLE-US-00001 TABLE 1 Test Num- Chemical Composition (unit is mass %; balance is Fe and impurities) ber C Si Mn P S Pb Al t-N t-O Cr Ni B V Mo W Nb Cu Ti Ca 1 0.49 0.80 0.94 0.014 0.046 0.048 0.026 0.004 0.0015 -- -- -- -- -- -- -- -- -- -- 2 0.38 0.38 0.20 0.014 0.013 0.034 0.033 0.004 0.0007 -- -- -- -- -- -- -- -- -- -- 3 0.69 0.34 1.33 0.007 0.045 0.045 0.050 0.005 0.0012 -- -- -- -- -- -- -- -- -- -- 4 0.48 0.25 1.17 0.005 0.033 0.025 0.022 0.005 0.0030 -- -- -- -- -- -- -- -- -- -- 5 0.63 0.39 0.65 0.015 0.010 0.041 0.029 0.008 0.0015 -- -- -- -- -- -- -- -- -- -- 6 0.54 0.50 1.66 0.010 0.022 0.051 0.029 0.006 0.0005 -- -- -- -- -- -- -- -- -- -- 7 0.80 0.42 1.36 0.012 0.021 0.035 0.029 0.005 0.0018 -- -- -- -- -- -- -- -- -- -- 8 0.30 0.39 1.01 0.010 0.021 0.025 0.020 0.007 0.0019 -- -- -- -- -- -- -- -- -- -- 9 0.53 0.01 1.48 0.009 0.025 0.035 0.032 0.007 0.0019 -- -- -- -- -- -- -- -- -- -- 10 0.56 0.24 1.90 0.014 0.033 0.045 0.023 0.007 0.0017 -- -- -- -- -- -- -- -- -- -- 11 0.55 0.41 1.55 0.030 0.024 0.043 0.029 0.006 0.0009 -- -- -- -- -- -- -- -- -- -- 12 0.56 0.46 0.92 0.009 0.100 0.040 0.026 0.007 0.0008 -- -- -- -- -- -- -- -- -- -- 13 0.47 0.29 1.59 0.017 0.017 0.100 0.023 0.006 0.0011 -- -- -- -- -- -- -- -- -- -- 14 0.66 0.32 1.59 0.014 0.026 0.010 0.023 0.005 0.0011 -- -- -- -- -- -- -- -- -- -- 15 0.51 0.43 0.90 0.005 0.021 0.035 0.010 0.005 0.0016 -- -- -- -- -- -- -- -- -- -- 16 0.43 0.49 1.22 0.007 0.047 0.049 0.026 0.015 0.0009 -- -- -- -- -- -- -- -- -- -- 17 0.57 0.34 1.68 0.014 0.040 0.030 0.025 0.005 0.0015 0.70 -- -- -- -- -- -- -- -- -- 18 0.53 0.44 0.85 0.012 0.031 0.045 0.031 0.007 0.0018 -- 3.50 -- -- -- -- -- -- -- -- 19 0.69 0.29 1.08 0.014 0.028 0.047 0.023 0.005 0.0013 -- -- 0.0050 -- -- -- -- -- -- -- 20 0.42 0.34 1.01 0.015 0.035 0.029 0.034 0.006 0.0013 -- -- -- 0.70 -- -- -- -- -- -- 21 0.46 0.32 0.66 0.015 0.020 0.025 0.029 0.008 0.0011 -- -- -- -- 0.70 -- -- -- -- -- 22 0.41 0.50 1.44 0.014 0.027 0.039 0.035 0.005 0.0013 -- -- -- -- -- 0.70 -- -- -- -- 23 0.44 0.42 0.49 0.011 0.052 0.031 0.036 0.008 0.0013 -- -- -- -- -- -- 0.049 -- -- -- 24 0.50 0.28 1.32 0.010 0.016 0.040 0.026 0.006 0.0011 -- -- -- -- -- -- -- 0.50 -- -- 25 0.51 0.44 1.42 0.012 0.026 0.030 0.029 0.006 0.0010 -- -- -- -- -- -- -- -- 0.100 -- 26 0.60 0.27 0.56 0.015 0.047 0.048 0.030 0.004 0.0012 -- -- -- -- -- -- -- -- -- 0.0030 27 0.80 0.43 1.35 0.011 0.023 0.032 0.033 0.006 0.0017 -- -- -- -- -- -- -- -- -- -- 28 0.54 0.01 1.47 0.008 0.027 0.036 0.033 0.007 0.0018 -- -- -- -- -- -- -- -- -- -- 29 0.57 0.39 1.59 0.030 0.026 0.044 0.031 0.004 0.0008 -- -- -- -- -- -- -- -- -- -- 30 0.48 0.30 1.60 0.015 0.018 0.100 0.024 0.007 0.0013 -- -- -- -- -- -- -- -- -- -- 31 0.50 0.42 0.91 0.006 0.019 0.033 0.010 0.004 0.0017 -- -- -- -- -- -- -- -- -- -- 32 0.55 0.49 1.68 0.009 0.024 0.049 0.031 0.005 0.0005 -- -- -- -- -- -- -- -- -- -- 33 0.54 0.45 0.82 0.013 0.030 0.042 0.033 0.008 0.0019 -- 3.50 -- -- -- -- -- -- -- -- 34 0.43 0.33 1.04 0.014 0.036 0.030 0.034 0.005 0.0012 -- -- -- 0.70 -- -- -- -- -- -- 35 0.43 0.44 0.51 0.009 0.049 0.034 0.035 0.007 0.0014 -- -- -- -- -- -- 0.049 -- -- -- 36 0.49 0.22 0.57 0.029 0.076 0.044 0.045 0.006 0.0017 -- -- -- -- -- -- -- -- -- -- 37 0.51 0.22 0.34 0.021 0.056 0.055 0.031 0.009 0.0012 -- -- -- -- -- -- -- -- -- -- 38 0.55 0.27 0.27 0.011 0.038 0.041 0.026 0.006 0.0013 -- -- -- -- -- -- -- -- -- -- 39 0.58 0.55 2.14 0.012 0.044 0.043 0.024 0.006 0.0013 -- -- -- -- -- -- -- -- -- -- 40 0.57 0.28 0.16 0.006 0.012 0.047 0.026 0.004 0.0015 -- -- -- -- -- -- -- -- -- -- 41 0.42 0.25 1.18 0.008 0.113 0.048 0.021 0.008 0.0020 -- -- -- -- -- -- -- -- -- -- 42 0.44 0.22 0.58 0.013 0.008 0.030 0.029 0.006 0.0010 -- -- -- -- -- -- -- -- -- -- 43 0.46 0.23 0.86 0.007 0.014 0.113 0.025 0.005 0.0015 -- -- -- -- -- -- -- -- -- -- 44 0.49 0.37 1.03 0.014 0.012 0.008 0.020 0.005 0.0007 -- -- -- -- -- -- -- -- -- -- 45 0.49 0.25 0.89 0.008 0.022 0.034 0.004 0.008 0.0014 -- -- -- -- -- -- -- -- -- -- 46 0.63 0.31 0.58 0.008 0.043 0.041 0.030 0.019 0.0014 -- -- -- -- -- -- -- -- -- -- 47 0.45 0.37 1.41 0.007 0.021 0.029 0.027 0.005 0.0034 -- -- -- -- -- -- -- -- -- -- 48 0.45 0.26 0.28 0.007 0.014 0.021 0.034 0.007 0.0003 -- -- -- -- -- -- -- -- -- -- 49 0.45 0.31 1.61 0.015 0.016 0.035 0.033 0.007 0.0016 -- -- -- -- -- -- -- -- -- -- 50 0.48 0.25 1.42 0.007 0.013 0.039 0.035 0.005 0.0018 -- -- -- -- -- -- -- -- -- --
[0149] The molten steel of each test number was produced by the following method. Hot metals produced by a well-known method were subjected to primary refining under the same conditions using a converter to thereby produce the molten steels of the respective test numbers.
[0150] For the molten steels of test numbers other than Test Numbers 49 and 50, after tapping the molten steel from the converter, Mn, Si and Al were added in that order to perform deoxidation. For the molten steel of Test Number 49, after tapping the molten steel from the converter, Si, Al and Mn were added in that order to perform deoxidation. For the molten steel of Test Number 50, after tapping the molten steel from the converter, Mn, Al and Si were added in that order to perform deoxidation.
[0151] After deoxidation, slag is removed. After removing slag, a primary treatment was performed using VAD, and thereafter an RH vacuum degassing treatment was performed. After the RH vacuum degassing treatment, final adjustment of alloying elements was performed. Molten steels having the chemical compositions shown in Table 1 were produced by the above described process.
[0152] Each of the molten steels was cast to produce an ingot for test use that had a rectangular parallelepiped shape. The cross sectional shape of the ingot was a rectangular shape with dimensions of 190 mm.times.190 mm. The solidification cooling rates RC (.degree. C./min) for the respective test numbers were as listed in Table 2. The solidification cooling rate RC was determined by measuring a secondary dendrite arm spacing of the ingot and applying the determined value to the aforementioned Formula (3).
TABLE-US-00002 TABLE 2 Machinability Amount of Chip Rusting Hot Test Deoxidation RC F1 = Micro- TN RA Hardness Tool Wear Treat- Charac- Ductility Overall Number Order (.degree. C./min) Mn/S structure (inclusions/mm.sup.2) (%) HV (.mu.m) ability teristics (%) Evaluation 1 Mn.fwdarw.Si.fwdarw.Al 8.3 20.4 F + P 254.4 89.0 198 115 .largecircle. .circle-w/dot. 75 .largecircle. 2 Mn.fwdarw.Si.fwdarw.Al 21.1 15.4 F + P 66.3 89.6 168 192 .largecircle. .circle-w/dot. 76 .largecircle. 3 Mn.fwdarw.Si.fwdarw.Al 40.5 29.6 F + P 138.1 79.2 275 167 .largecircle. .circle-w/dot. 79 .largecircle. 4 Mn.fwdarw.Si.fwdarw.Al 30.8 35.5 F + P 58.0 71.3 200 181 .largecircle. .circle-w/dot. 81 .largecircle. 5 Mn.fwdarw.Si.fwdarw.Al 33.2 65.0 F + P 91.7 79.8 255 181 .largecircle. .circle-w/dot. 79 .largecircle. 6 Mn.fwdarw.Si.fwdarw.Al 11.8 75.5 F + P 106.4 77.8 219 168 .largecircle. .circle-w/dot. 83 .largecircle. 7 Mn.fwdarw.Si.fwdarw.Al 54.2 64.8 F + P 59.8 38.3 272 188 .largecircle. .largecircle. 74 .largecircle. 8 Mn.fwdarw.Si.fwdarw.Al 68.2 48.1 F + P 55.9 32.6 188 181 .largecircle. .largecircle. 71 .largecircle. 9 Mn.fwdarw.Si.fwdarw.Al 65.3 59.2 F + P 61.2 36.5 211 163 .largecircle. .largecircle. 82 .largecircle. 10 Mn.fwdarw.Si.fwdarw.Al 72.7 45.5 F + P 57.4 35.8 270 179 .largecircle. .largecircle. 84 .largecircle. 11 Mn.fwdarw.Si.fwdarw.Al 88.2 64.6 F + P 49.1 35.6 215 162 .largecircle. .largecircle. 75 .largecircle. 12 Mn.fwdarw.Si.fwdarw.Al 59.9 9.2 F + P 110.2 38.0 228 110 .largecircle. .largecircle. 72 .largecircle. 13 Mn.fwdarw.Si.fwdarw.Al 72.5 93.5 F + P 48.3 37.2 244 106 .largecircle. .largecircle. 78 .largecircle. 14 Mn.fwdarw.Si.fwdarw.Al 65.1 61.2 F + P 50.5 35.8 269 197 .largecircle. .largecircle. 81 .largecircle. 15 Mn.fwdarw.Si.fwdarw.Al 64.3 42.9 F + P 59.8 31.4 191 179 .largecircle. .largecircle. 76 .largecircle. 16 Mn.fwdarw.Si.fwdarw.Al 88.0 26.0 F + P 68.7 21.6 177 187 .largecircle. .largecircle. 73 .largecircle. 17 Mn.fwdarw.Si.fwdarw.Al 39.4 42.0 F + P 125.4 74.8 265 187 .largecircle. .circle-w/dot. 79 .largecircle. 18 Mn.fwdarw.Si.fwdarw.Al 77.5 27.4 F + P 52.3 36.1 243 161 .largecircle. .largecircle. 79 .largecircle. 19 Mn.fwdarw.Si.fwdarw.Al 42.4 38.6 F + P 121.4 65.0 285 187 .largecircle. .circle-w/dot. 75 .largecircle. 20 Mn.fwdarw.Si.fwdarw.Al 59.6 28.9 F + P 48.7 33.1 268 156 .largecircle. .largecircle. 79 .largecircle. 21 Mn.fwdarw.Si.fwdarw.Al 61.9 33.0 F + P 47.3 31.2 231 188 .largecircle. .largecircle. 82 .largecircle. 22 Mn.fwdarw.Si.fwdarw.Al 9.3 53.3 F + P 88.2 60.4 266 198 .largecircle. .circle-w/dot. 81 .largecircle. 23 Mn.fwdarw.Si.fwdarw.Al 69.7 9.4 F + P 66.9 36.2 218 129 .largecircle. .largecircle. 79 .largecircle. 24 Mn.fwdarw.Si.fwdarw.Al 72.1 82.5 F + P 41.6 32.0 232 182 .largecircle. .largecircle. 82 .largecircle. 25 Mn.fwdarw.Si.fwdarw.Al 44.1 54.6 F + P 101.6 59.2 231 162 .largecircle. .circle-w/dot. 79 .largecircle. 26 Mn.fwdarw.Si.fwdarw.Al 54.6 11.9 F + P 51.1 39.4 244 149 .largecircle. .largecircle. 76 .largecircle. 27 Mn.fwdarw.Si.fwdarw.Al 148.5 58.7 F + P 33.2 28.4 285 190 .largecircle. X 75 X 28 Mn.fwdarw.Si.fwdarw.Al 195.6 54.4 F + P 37.2 32.8 219 174 .largecircle. X 85 X 29 Mn.fwdarw.Si.fwdarw.Al 102.5 61.2 F + P 31.5 30.4 229 165 .largecircle. X 79 X 30 Mn.fwdarw.Si.fwdarw.Al 111.3 88.9 F + P 28.9 38.1 246 107 .largecircle. X 80 X 31 Mn.fwdarw.Si.fwdarw.Al 188.7 47.9 F + P 32.8 28.4 203 187 .largecircle. X 73 X 32 Mn.fwdarw.Si.fwdarw.Al 175.0 70.0 F + P 39.4 31.4 222 162 .largecircle. X 84 X 33 Mn.fwdarw.Si.fwdarw.Al 173.2 27.3 F + P 29.2 29.9 244 167 .largecircle. X 81 X 34 Mn.fwdarw.Si.fwdarw.Al 109.9 28.9 F + P 37.0 39.6 255 176 .largecircle. X 73 X 35 Mn.fwdarw.Si.fwdarw.Al 102.1 10.4 F + P 25.9 38.4 221 133 .largecircle. X 81 X 36 Mn.fwdarw.Si.fwdarw.Al 31.2 7.5 F + P 31.5 58.1 201 62 .largecircle. X 49 X 37 Mn.fwdarw.Si.fwdarw.Al 71.5 6.1 F + P 29.6 33.8 198 122 .largecircle. X 53 X 38 Mn.fwdarw.Si.fwdarw.Al 200.0 7.1 F + P 27.5 34.6 225 148 .largecircle. X 57 X 39 Mn.fwdarw.Si.fwdarw.Al 79.6 35.0 F + P 67.7 79.1 304 288 .largecircle. .largecircle. 82 X 40 Mn.fwdarw.Si.fwdarw.Al 187.4 13.3 F + P 30.9 34.2 237 144 .largecircle. X 58 X 41 Mn.fwdarw.Si.fwdarw.Al 63.1 10.4 F + P 113.0 79.8 171 102 .largecircle. .largecircle. 51 X 42 Mn.fwdarw.Si.fwdarw.Al 54.4 72.5 F + P 37.8 32.8 181 144 .largecircle. X 83 X 43 Mn.fwdarw.Si.fwdarw.Al 21.7 59.7 F + P 118.3 35.8 188 111 .largecircle. X 61 X 44 Mn.fwdarw.Si.fwdarw.Al 198.5 85.8 F + P 7.1 92.8 205 270 X .largecircle. 79 X 45 Mn.fwdarw.Si.fwdarw.Al 56.2 115.6 F + P 35.6 35.8 226 134 .largecircle. X 75 X 46 Mn.fwdarw.Si.fwdarw.Al 94.4 13.5 F + P 43.0 84.7 260 256 .largecircle. .largecircle. 63 X 47 Mn.fwdarw.Si.fwdarw.Al 187.8 67.1 F + P 38.7 81.1 193 210 .largecircle. .largecircle. 80 X 48 Mn.fwdarw.Si.fwdarw.Al 177.4 67.1 F + P 15.7 61.1 181 215 X .largecircle. 81 X 49 Si.fwdarw.Al.fwdarw.Mn 42.8 100.6 F + P 29.8 37.2 244 106 .largecircle. X 78 X 50 Mn.fwdarw.Al.fwdarw.Si 44.6 109.2 F + P 31.1 33.7 204 158 .largecircle. X 77 X
[0153] The produced ingots for test use were subjected to hot working twice to produce a steel bar. In the hot working, blooming was performed, and thereafter finish rolling (steel bar rolling) was performed. The produced test ingot was subjected to hot forging to produce a steel bar having a diameter of 50 mm. Alternatively, the test ingot was subjected to blooming, and then subjected to finish rolling to produce a steel bar having a diameter of 50 mm. A normalizing treatment in a range of 800 to 950.degree. C. was performed on the produced steel bar. The cooling method adopted in the normalizing treatment was to allow cooling of the steel bar. A steel bar (steel for machine structural use) having a diameter of 50 mm was produced by the above-described production process.
[0154] [Evaluation Tests]
[0155] [Micro-Structure Observation]
[0156] A test specimen for micro-structure observation use was taken from the R/2 part of the steel bar of each test number. Of the entire surface of the test specimen, a cross-section parallel to the longitudinal direction (that is, the rolling direction or elongation direction) of the steel bar was defined as the observation surface. The total area fraction (%) of ferrite and pearlite were determined based on the method described above. The total area fraction was 99% or more in the micro-structure of the steel bar of each test number. A micro-structure in which the total area fraction was 99% or more is shown as "F+P" in Table 2.
[0157] [Number TN of Specific Inclusions and Composite Ratio RA]
[0158] A test specimen for observing the micro-structure was taken from the R/2 part of the steel bar of each test number. Of the entire surface of the test specimen, a cross-section that was parallel to the longitudinal direction (that is, the rolling direction or elongation direction) of the steel bar was defined as the observation surface. For the observation surface of the test specimen for observing the micro-structure of each test number, the specific inclusions number TN (inclusions/mm.sup.2) and the composite ratio RA (%) were determined based on the above described method. The results are shown in Table 2.
[0159] [Vickers Hardness Test]
[0160] A Vickers hardness test was performed in conformity with JIS Z 2244 (1981) at an arbitrary five points of the R/2 part of the steel bar of each test number. The test force was set to 100 N. The average of the obtained five values was defined as the Vickers hardness (HV) of the steel bar of the relevant test number. If the Vickers hardness was HV 160 or more, it was determined that the steel bar had sufficient strength. On the other hand, if the Vickers hardness was less than HV 160, it was determined that the steel bar had insufficient strength. The results are shown in Table 2. The results show that the Vickers hardness was HV 160 or more for each test number, indicating sufficient strength.
[0161] [Machinability]
[0162] The machinability was evaluated by evaluating the amount of tool wear (.mu.m) and the chip treatability. Specifically, a steel bar having a diameter of 50 mm was cut to a predetermined length and adopted as a cutting test specimen. The cutting test specimen was subjected to outer circumferential lathe turning as illustrated in FIG. 5. The conditions of the outer circumferential lathe turning are shown in Table 3.
TABLE-US-00003 TABLE 3 Cutting Speed 200 m/min Depth-of-cut Amount 2 mm Feed Rate 0.2 mm/rev Wet Type Water-soluble Cutting Oil Tool Chip P20 Cemented Carbide Tool SNMG 120408 Circumference Groove With Breaker Holder DSBN-R2525
[0163] Specifically, a P20 cemented carbide tool was used as a tool 50. The nose radius of the tool 50 was 0.4, and the rake angle thereof was 5.degree.. Outer circumferential lathe turning was performed under the following conditions: cutting speed V1: 200 m/min; feed speed V2: 0.2 mm/rev; depth-of-cut amount D1: 2 mm; and longitudinal direction cutting length L1: 200 mm. After cutting the outer circumference, cutting lathe turning was repeated again so as to obtain a small diameter of D1: 2 mm, and with respect to test specimen 5, a lathe turning test was performed under the aforementioned conditions for four minutes.
[0164] [Service Life Evaluation]
[0165] The amount of tool wear (mm) of the minor flank was measured with respect to the tool 50 after lathe turning of the 1,000.sup.th test specimen was completed. The measurement results are shown in the "amount of tool wear" column in Table 2. The service life was determined as excellent if the amount of tool wear was 200 .mu.m or less. On the other hand, if the amount of tool wear was more than 200 .mu.m, the service life was determined as being not excellent.
[0166] [Chip Treatability Evaluation]
[0167] In the lathe turning for the 1000.sup.th test specimen, a chip as illustrated in FIG. 6A and FIG. 6B was obtained. A length L20 and a diameter D20 of the chip were measured. Based on the measurement result, the chip treatability was evaluated as follows. If the chip was a coil shape of not more than 30 mm in diameter, or if the chip length was less than 50 mm even if the chip was not a coil shape, the chip treatability was determined as being excellent (".largecircle." in Table 2). On the other hand, if the chip was not a coil shape of not more than 30 mm in diameter, and the chip length was also 50 mm or more, the chip treatability was determined as being poor ("x" in Table 2).
[0168] [Rusting characteristics (corrosion resistance) evaluation test] A rust test specimen was prepared by cutting the steel bar having a diameter of 50 mm to a predetermined length. The rust test specimen was subjected to lathe turning under similar conditions as in the aforementioned cutting test. Thereafter, the test specimen was stored for one hour in an atmosphere with a humidity of 70% and a temperature of 20.degree. C. while spraying tap water onto the cut surface. After storage, the cut surface of the test specimen was observed and the number of rust points was measured. The measurement results are shown in the "rusting characteristics" column in Table 2. If the number of rust points was less than 10 (indicated by ".circle-w/dot." in Table 2), and if the number of rust points was 10 or more and less than 20 (indicated by ".largecircle." in Table 2), the rusting characteristics were determined as being excellent. On the other hand, if the number of rust points was 20 or more ("x" in Table 2), the rusting characteristics were determined as being not excellent.
[0169] [Hot Ductility Evaluation Test]
[0170] A hot tension test was performed by electrical heating, and the hot ductility was evaluated. Specifically, from the cast piece of each test number, a round bar specimen that had a diameter of 10 mm and a length of 100 mm and in which both ends had been subjected to screw machining was prepared. The round bar specimen was heated to 1100.degree. C. by electrical heating, and held at that temperature for three minutes. Thereafter, the round bar specimen was cooled to 900.degree. C. by being allowed to cool. The tension test was executed in a state in which the temperature of the round bar specimen was 900.degree. C., and the reduction of area (%) at the time of breaking off was determined. The tension test was performed on three round bar specimens for each test number, and the average of the three values was defined as the reduction of area (%) of the relevant test number. The reduction of area is shown in the "hot ductility" column in Table 2. If the reduction of area was 70% or more, the hot ductility was evaluated as excellent. On the other hand, if the reduction of area was less than 70%, the hot ductility was evaluated as not excellent.
[0171] [Test Results]
[0172] In Test Numbers 1 to 26, the chemical composition was appropriate, F1 was 8.0 or more, the deoxidation order was appropriate, and the solidification cooling rate RC was 100.degree. C./min or less. Therefore, the number TN of specific inclusions was 40 per mm.sup.2 or more. As a result, the amount of tool wear was 200 .mu.m or less, and excellent chip treatability was obtained. That is, excellent machinability was obtained. In addition, in the rusting characteristics evaluation test, the number of rust points was less than 20 for each of these test numbers, and thus excellent rusting characteristics were obtained. In addition, in the hot ductility evaluation test, the reduction of area was 70% or more, showing that excellent hot ductility was obtained.
[0173] In Test Numbers 1 to 6, 17, 19, 22 and 25, the solidification cooling rate RC was within the range of 8 to 50.degree. C./min. Therefore, not only was the number TN of specific inclusions 40 per mm.sup.2 or more, but furthermore the composite ratio RA was 40% or more. As a result, for each of these test numbers, the number of rust points was less than 10, and thus rusting characteristics that were even more excellent in comparison to Test Numbers 7 to 16, 18, 20, 21, 23, 24 and 26 were obtained.
[0174] On the other hand, in Test Numbers 27 to 35, although the chemical composition was appropriate and F1 was 8.0 or more and the deoxidation order was appropriate, the solidification cooling rate RC was more than 100.degree. C./min. Consequently, the number TN of specific inclusions was less than 40 per mm.sup.2. As a result, excellent rusting characteristics were not obtained.
[0175] In Test Numbers 36 and 37, although the chemical composition was appropriate and the deoxidation order was appropriate and the solidification cooling rate RC was not more than 100.degree. C./min, F1 was less than 8.0. Consequently, the number TN of specific inclusions was less than 40 per mm.sup.2. As a result, excellent rusting characteristics were not obtained. In addition, the reduction of area was less than 70%, and thus excellent hot ductility was not obtained.
[0176] In Test Number 38, although the chemical composition was appropriate and the deoxidation order was appropriate, the solidification cooling rate RC was more than 100.degree. C./min and F1 was less than 8.0. Consequently, the number TN of specific inclusions was less than 40 per mm.sup.2. As a result, excellent rusting characteristics were not obtained. In addition, the reduction of area was less than 70%, and thus excellent hot ductility was not obtained.
[0177] In Test Number 39, the Mn content was too high. As a result, the amount of tool wear was more than 200 .mu.m, and thus excellent machinability was not obtained.
[0178] In Test Number 40, the Mn content was too low. In addition, the solidification cooling rate RC was more than 100.degree. C./min. Therefore, the number TN of specific inclusions was less than 40 per mm.sup.2. As a result, excellent rusting characteristics were not obtained. In addition, the point value was less than 70%, and thus excellent hot ductility was not obtained.
[0179] In Test Number 41, the S content was too high. As a result, the point value was less than 70%, and thus excellent hot ductility was not obtained.
[0180] In Test Number 42, the S content was too low. Consequently, the number TN of specific inclusions was less than 40 per mm.sup.2. As a result, excellent rusting characteristics were not obtained.
[0181] In Test Number 43, the Pb content was too high. As a result, excellent rusting characteristics were not obtained.
[0182] In Test Number 44, the Pb content was too low. In addition, the solidification cooling rate RC was more than 100.degree. C./min. Therefore, the number TN of specific inclusions was less than 40 per mm.sup.2. As a result, the amount of tool wear was more than 200 .mu.m and, furthermore, excellent chip treatability was also not obtained. That is, excellent machinability was not obtained.
[0183] In Test Number 45, the Al content was too low. Therefore, the number TN of specific inclusions was less than 40 per mm.sup.2. As a result, excellent rusting characteristics were not obtained.
[0184] In Test Number 46, the N content was too high. As a result, the amount of tool wear was more than 200 .mu.m, and thus excellent machinability was not obtained. In addition, the point value was less than 70%, and thus excellent hot ductility was not obtained.
[0185] In Test Number 47, the O content was too high. In addition, the solidification cooling rate RC was more than 100.degree. C./min. Therefore, the number TN of specific inclusions was less than 40 per mm.sup.2. As a result, the amount of tool wear was more than 200 .mu.m, and thus excellent machinability was not obtained.
[0186] In Test Number 48, the O content was too low. In addition, the solidification cooling rate RC was more than 100.degree. C./min. Therefore, the number TN of specific inclusions was less than 40 per mm.sup.2. As a result, the amount of tool wear was more than 200 .mu.m and, furthermore, excellent chip treatability was also not obtained. That is, excellent machinability was not obtained.
[0187] In Test Number 49, although the chemical composition was appropriate and F1 was 8.0 or more and the solidification cooling rate RC was not more than 100.degree. C./min, the deoxidation order was inappropriate. Consequently, the number TN of specific inclusions was less than 40 per mm.sup.2. As a result, excellent rusting characteristics were not obtained.
[0188] In Test Number 50, although the chemical composition was appropriate and F1 was 8.0 or more and the solidification cooling rate RC was not more than 100.degree. C./min, the deoxidation order was inappropriate. Consequently, the number TN of specific inclusions was less than 40 per mm.sup.2. As a result, excellent rusting characteristics were not obtained.
[0189] An embodiment of the present invention has been described above. However, the foregoing embodiment is merely an example for implementing the present invention. Accordingly, the present invention is not limited to the above embodiment, and the above embodiment can be appropriately modified within a range which does not deviate from the gist of the present invention.
REFERENCE SIGNS LIST
[0190] 10 MnS Inclusions [0191] 20 Pb Inclusions [0192] 30 Composite Inclusions
* * * * *
D00000

D00001

D00002

D00003

D00004

D00005

D00006

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.