Heterocyclic Compound, And Organic Light-emitting Device Using Same
OH; Han-Kook ; et al.
U.S. patent application number 16/315271 was filed with the patent office on 2019-08-01 for heterocyclic compound, and organic light-emitting device using same. This patent application is currently assigned to HEESUNG MATERIAL LTD.. The applicant listed for this patent is HEESUNG MATERIAL LTD.. Invention is credited to Dae-Hyuk CHOI, Jin-Seok CHOI, Won-Jang JEONG, Yong-Geun JUNG, Joo-Dong LEE, Jun-Tae MO, Han-Kook OH.
| Application Number | 20190233398 16/315271 |
| Document ID | / |
| Family ID | 60913070 |
| Filed Date | 2019-08-01 |












View All Diagrams
| United States Patent Application | 20190233398 |
| Kind Code | A1 |
| OH; Han-Kook ; et al. | August 1, 2019 |
HETEROCYCLIC COMPOUND, AND ORGANIC LIGHT-EMITTING DEVICE USING SAME
Abstract
The present application provides a hetero-cyclic compound capable of significantly enhancing lifespan, efficiency, electrochemical stability and thermal stability of an organic light emitting device, and an organic light emitting device containing the hetero-cyclic compound in an organic compound layer.
| Inventors: | OH; Han-Kook; (Osan-si, KR) ; MO; Jun-Tae; (Osan-si, KR) ; JUNG; Yong-Geun; (Seoul, KR) ; JEONG; Won-Jang; (Hwaseong-si, KR) ; CHOI; Jin-Seok; (Suwon-si, KR) ; CHOI; Dae-Hyuk; (Yongin-si, KR) ; LEE; Joo-Dong; (Seongnam-si, KR) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | HEESUNG MATERIAL LTD. Yongin-City, Gyeonggi-do KR |
||||||||||
| Family ID: | 60913070 | ||||||||||
| Appl. No.: | 16/315271 | ||||||||||
| Filed: | July 6, 2017 | ||||||||||
| PCT Filed: | July 6, 2017 | ||||||||||
| PCT NO: | PCT/KR2017/007256 | ||||||||||
| 371 Date: | January 4, 2019 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C09K 11/06 20130101; C07D 401/14 20130101; H01L 51/5012 20130101; C07D 221/06 20130101; H01L 51/0067 20130101; H01L 51/5044 20130101; C07D 409/14 20130101; H01L 51/0072 20130101; C07D 471/04 20130101; H01L 51/0073 20130101; C07D 405/14 20130101; H01L 51/0074 20130101; C07D 401/10 20130101; H01L 51/5056 20130101; H01L 51/5092 20130101; C07D 401/06 20130101; H01L 51/00 20130101; H01L 51/50 20130101; H01L 51/5072 20130101; H01L 51/5016 20130101; H01L 51/5096 20130101; H01L 51/5088 20130101; H01L 51/0054 20130101 |
| International Class: | C07D 401/14 20060101 C07D401/14; C07D 221/06 20060101 C07D221/06; C07D 405/14 20060101 C07D405/14; C07D 409/14 20060101 C07D409/14; C07D 401/10 20060101 C07D401/10; C07D 471/04 20060101 C07D471/04; H01L 51/00 20060101 H01L051/00; H01L 51/50 20060101 H01L051/50 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Jul 6, 2016 | KR | 10-2016-0085746 |
Claims
1. A hetero-cyclic compound represented by the following Chemical Formula 1: ##STR00130## wherein, in Chemical Formula 1, L is a direct bond; a substituted or unsubstituted C.sub.6 to C.sub.60 arylene group; or a C.sub.2 to C.sub.60 heteroarylene group; Z is selected from the group consisting of hydrogen; deuterium; a halogen group; --CN; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group; --SiRR'R''; --P(.dbd.O)RR'; and an amine group unsubstituted or substituted with a C.sub.1 to C.sub.20 alkyl group, a C.sub.6 to C.sub.60 aryl group or a C.sub.2 to C.sub.60 heteroaryl group; m is an integer of 0 to 4; n is an integer of 1 to 4; R1 to R6, and R8 are the same as or different from each other, and each independently selected from the group consisting of hydrogen; deuterium; a halogen group; --CN; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 alkenyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 alkynyl group; a substituted or unsubstituted C.sub.1 to C.sub.60 alkoxy group; a substituted or unsubstituted C.sub.3 to C.sub.60 cycloalkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heterocycloalkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group; --SiRR'R''; --P(.dbd.O)RR'; and an amine group unsubstituted or substituted with a C.sub.1 to C.sub.20 alkyl group, a C.sub.6 to C.sub.60 aryl group or a C.sub.2 to C.sub.60 heteroaryl group, or two or more groups adjacent to each other bond to each other to form a substituted or unsubstituted aliphatic or aromatic hydrocarbon ring; R7 is selected from the group consisting of hydrogen; deuterium; a halogen group; --CN; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 alkenyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 alkynyl group; a substituted or unsubstituted C.sub.1 to C.sub.60 alkoxy group; a substituted or unsubstituted C.sub.3 to C.sub.60 cycloalkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heterocycloalkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group; --SiRR'R''; --P(.dbd.O)RR'; and an amine group unsubstituted or substituted with a C.sub.1 to C.sub.2 alkyl group, a C.sub.6 to C.sub.60 aryl group or a C.sub.2 to C.sub.60 heteroaryl group; and R, R' and R'' are the same as or different from each other, and each independently hydrogen; deuterium; --CN; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.3 to C.sub.60 cycloalkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; or a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group.
2. The hetero-cyclic compound of claim 1, wherein at least one of R1 and R6 of Chemical Formula 1 is a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group, and the rest are hydrogen or deuterium.
3. The hetero-cyclic compound of claim 1, wherein Z of Chemical Formula 1 is selected from the group consisting of hydrogen; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; and a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group.
4. The hetero-cyclic compound of claim 1, wherein Chemical Formula 1 is represented by any one of the following Chemical Formulae 2 to 6: ##STR00131## wherein, in Chemical Formulae 2 to 6, L, R1 to R8, and m have the same definitions as in Chemical Formula 1; at least one of X1 to X3 is N, and the rest are each independently N or CR23; R9 to R23 are the same as or different from each other, and each independently selected from the group consisting of hydrogen; deuterium; a halogen group; --CN; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 alkenyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 alkynyl group; a substituted or unsubstituted C.sub.1 to C.sub.60 alkoxy group; a substituted or unsubstituted C.sub.3 to C.sub.60 cycloalkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heterocycloalkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group; --SiRR'R''; --P(.dbd.O)RR'; and an amine group unsubstituted or substituted with a C.sub.1 to C.sub.20 alkyl group, a C.sub.6 to C.sub.60 aryl group or a C.sub.2 to C.sub.60 heteroaryl group, or two or more groups adjacent to each other bond to each other to form a substituted or unsubstituted aliphatic or aromatic hydrocarbon ring; and p, q, r and s are each independently an integer of 0 to 3.
5. The hetero-cyclic compound of claim 4, wherein at least one of R1 and R6 and at least one of R11 and R16 of Chemical Formula 2 are a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group, and the rest are hydrogen or deuterium.
6. The hetero-cyclic compound of claim 1, wherein Chemical Formula 1 is represented by any one of the following compounds: ##STR00132## ##STR00133## ##STR00134## ##STR00135## ##STR00136## ##STR00137## ##STR00138## ##STR00139## ##STR00140## ##STR00141## ##STR00142## ##STR00143## ##STR00144## ##STR00145## ##STR00146## ##STR00147## ##STR00148## ##STR00149## ##STR00150## ##STR00151## ##STR00152## ##STR00153## ##STR00154## ##STR00155## ##STR00156## ##STR00157## ##STR00158## ##STR00159## ##STR00160## ##STR00161## ##STR00162## ##STR00163## ##STR00164## ##STR00165## ##STR00166## ##STR00167## ##STR00168## ##STR00169##
7. An organic light emitting device comprising: an anode; a cathode; and one or more organic material layers provided between the anode and the cathode, wherein one or more layers of the organic material layers comprise the hetero-cyclic compound of claim 1.
8. The organic light emitting device of claim 7, wherein the organic material layer comprises at least one of a hole blocking layer, an electron injection layer and an electron transfer layer, and at least one of the hole blocking layer, the electron injection layer and the electron transfer layer comprises the hetero-cyclic compound.
9. The organic light emitting device of claim 7, wherein the organic material layer comprises a light emitting layer, and the light emitting layer comprises the hetero-cyclic compound.
10. The organic light emitting device of claim 7, wherein the organic material layer comprises one or more of a hole injection layer, a hole transfer layer, and a layer carrying out hole injection and hole transfer at the same time, and one of the above-mentioned layers comprises the hetero-cyclic compound.
11. The organic light emitting device of claim 7, wherein the organic material layer comprises a charge generation layer, and the charge generation layer comprises the hetero-cyclic compound.
12. The organic light emitting device of claim 7, comprising: an anode; a first stack provided on the anode and comprising a first light emitting layer; a charge generation layer provided on the first stack; a second stack provided on the charge generation layer and comprising a second light emitting layer; and a cathode provided on the second stack.
Description
TECHNICAL FIELD
[0001] The present application claims priority to and the benefits of Korean Patent Application No. 10-2016-0085746, filed with the Korean Intellectual Property Office on Jul. 6, 2016, the entire contents of which are incorporated herein by reference.
[0002] The present application relates to a hetero-cyclic compound and an organic light emitting device using the same.
BACKGROUND ART
[0003] An electroluminescent device is one type of self-emissive display devices, and has an advantage of having a wide viewing angle, and a high response speed as well as having an excellent contrast.
[0004] An organic light emitting device has a structure disposing an organic thin film between two electrodes. When a voltage is applied to an organic light emitting device having such a structure, electrons and holes injected from the two electrodes bind and pair in the organic thin film, and light emits as these annihilate. The organic thin film may be formed in a single layer or a multilayer as necessary.
[0005] A material of the organic thin film may have a light emitting function as necessary. For example, as a material of the organic thin film, compounds capable of forming a light emitting layer themselves may be used alone, or compounds capable of performing a role of a host or a dopant of a host-dopant-based light emitting layer may also be used. In addition thereto, compounds capable of performing roles of hole injection, hole transfer, electron blocking, hole blocking, electron transfer, electron injection and the like may also be used as a material of the organic thin film.
[0006] Development of an organic thin film material has been continuously required for enhancing performance, lifespan or efficiency of an organic light emitting device.
PRIOR ART DOCUMENTS
Patent Documents
[0007] U.S. Pat. No. 4,356,429
DISCLOSURE
Technical Problem
[0008] Researches for an organic light emitting device comprising a compound capable of satisfying conditions required for materials usable in an organic light emitting device, for example, a proper energy level, electrochemical stability, thermal stability and the like, and having a chemical structure that may perform various roles required in an organic light emitting device depending on substituents have been required.
Technical Solution
[0009] One embodiment of the present application provides a hetero-cyclic compound represented by the following Chemical Formula 1:
##STR00001##
[0010] in Chemical Formula 1,
[0011] L is a direct bond; a substituted or unsubstituted C.sub.6 to C.sub.60 arylene group; or a C.sub.2 to C.sub.60 heteroarylene group,
[0012] Z is selected from the group consisting of hydrogen;
[0013] deuterium; a halogen group; --CN; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group; --SiRR'R''; --P(.dbd.O)RR'; and an amine group unsubstituted or substituted with a C.sub.1 to C.sub.20 alkyl group, a C.sub.6 to C.sub.60 aryl group or a C.sub.2 to C.sub.60 heteroaryl group,
[0014] m is an integer of 0 to 4,
[0015] n is an integer of 1 to 4,
[0016] R1 to R6, and R8 are the same as or different from each other, and each independently selected from the group consisting of hydrogen; deuterium; a halogen group; --CN; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 alkenyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 alkynyl group; a substituted or unsubstituted C.sub.1 to C.sub.60 alkoxy group; a substituted or unsubstituted C.sub.3 to C.sub.60 cycloalkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heterocycloalkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group; --SiRR'R''; --P(.dbd.O)RR'; and an amine group unsubstituted or substituted with a C.sub.1 to C.sub.20 alkyl group, a C.sub.6 to C.sub.60 aryl group or a C.sub.2 to C.sub.60 heteroaryl group, or two or more groups adjacent to each other bond to each other to form a substituted or unsubstituted aliphatic or aromatic hydrocarbon ring,
[0017] R7 is selected from the group consisting of hydrogen; deuterium; a halogen group; --CN; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 alkenyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 alkynyl group; a substituted or unsubstituted C.sub.1 to C.sub.60 alkoxy group; a substituted or unsubstituted C.sub.3 to C.sub.60 cycloalkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heterocycloalkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group; --SiRR'R''; --P(.dbd.O)RR'; and an amine group unsubstituted or substituted with a C.sub.1 to C.sub.20 alkyl group, a C.sub.6 to C.sub.60 aryl group or a C.sub.2 to C.sub.60 heteroaryl group, and
[0018] R, R' and R'' are the same as or different from each other, and each independently hydrogen; deuterium; --CN; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.3 to C.sub.60 cycloalkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; or a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group.
[0019] Another embodiment of the present application provides an organic light emitting device comprising an anode, a cathode, and one or more organic material layers provided between the anode and the cathode, wherein one or more layers of the organic material layers comprise the hetero-cyclic compound represented by Chemical Formula 1.
Advantageous Effects
[0020] A hetero-cyclic compound according to one embodiment of the present application can be used as an organic material layer material of an organic light emitting device. The hetero-cyclic compound can be used as a material of a hole injection layer, a hole transfer layer, a light emitting layer, an electron transfer layer, an electron injection layer, a charge generation layer or the like in an organic light emitting device. Particularly, the hetero-cyclic compound represented by Chemical Formula 1 can be used as a material of an electron transfer layer or a charge generation layer in an organic light emitting device. In addition, using the hetero-cyclic compound represented by Chemical Formula 1 in an organic light emitting device lowers a driving voltage of the device, enhances light efficiency, and can enhance a lifespan property of the device with thermal stability of the compound.
DESCRIPTION OF DRAWINGS
[0021] FIG. 1 to FIG. 4 are diagrams each schematically illustrating a lamination structure of an organic light emitting device according to one embodiment of the present application.
REFERENCE NUMERAL
[0022] 100: Substrate [0023] 200: Anode [0024] 300: Organic Material Layer [0025] 301: Hole Injection Layer [0026] 302: Hole Transfer Layer [0027] 303: Light Emitting Layer [0028] 304: Hole Blocking Layer [0029] 305: Electron Transfer Layer [0030] 306: Electron Injection Layer [0031] 400: Cathode
MODE FOR DISCLOSURE
[0032] Hereinafter, the present application will be described in detail.
[0033] A hetero-cyclic compound according to one embodiment of the present application is represented by Chemical Formula 1. More specifically, the hetero-cyclic compound represented by Chemical Formula 1 is capable of being used as an organic material layer material of an organic light emitting device with such a core structure and structural characteristics of substituents.
[0034] In one embodiment of the present application, when m of Chemical Formula 1 is 2 or greater, two or more Ls may be the same as or different from each other. In addition, when n of Chemical Formula 1 is 2 or greater, two or more Zs may be the same as or different from each other.
[0035] In one embodiment of the present application, m of Chemical Formula 1 may be an integer of 1 to 4.
[0036] In one embodiment of the present application, at least one of R1 and R6 of Chemical Formula 1 is a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group, and the rest may be hydrogen or deuterium.
[0037] In another embodiment, at least one of R1 and R6 of Chemical Formula 1 is a C.sub.6 to C.sub.60 aryl group, and the rest may be hydrogen or deuterium.
[0038] In another embodiment, at least one of R1 and R6 of Chemical Formula 1 is a phenyl group, and the rest may be hydrogen.
[0039] In one embodiment of the present application, Z of Chemical Formula 1 may be selected from the group consisting of hydrogen; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; and a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group.
[0040] In another embodiment, Z of Chemical Formula 1 may be hydrogen; a C.sub.6 to C.sub.60 aryl group unsubstituted or substituted with one or more substituents selected from the group consisting of a C.sub.6 to C.sub.60 aryl group and a C.sub.2 to C.sub.60 heteroaryl group; or a C.sub.2 to C.sub.60 heteroaryl group unsubstituted or substituted with one or more substituents selected from the group consisting of a C.sub.1 to C.sub.60 alkyl group, a C.sub.6 to C.sub.60 aryl group and a C.sub.2 to C.sub.60 heteroaryl group.
[0041] In another embodiment, Z of Chemical Formula 1 may be hydrogen; a phenyl group; a naphthyl group unsubstituted or substituted with a phenyl group; an anthracene group; a triphenylene group; a pyrene group; a phenanthrene group; or a perylene group.
[0042] In another embodiment, Z of Chemical Formula 1 may be hydrogen; a benzoquinoline group unsubstituted or substituted with a phenyl group; a triazine group unsubstituted or substituted with one or more substituents selected from the group consisting of a phenyl group and a biphenyl group; a pyrimidine group unsubstituted or substituted with one or more substituents selected from the group consisting of a phenyl group and a biphenyl group; a pyridine group unsubstituted or substituted with one or more substituents selected from the group consisting of a phenyl group, a pyrene group, a triphenylene group, a quinoline group and a naphthyl group; a quinoline group unsubstituted or substituted with one or more substituents selected from the group consisting of a pyridine group, a phenyl group and pyrene group; a phenanthroline group unsubstituted or substituted with one or more substituents selected from the group consisting of a naphthyl group, a methyl group, a quinoline group, a phenyl group, a pyridine group and a biphenyl group; a benzimidazole group unsubstituted or substituted with a phenyl group; a dibenzothiophene group; a dibenzofuran group; or a carbazole group.
[0043] In one embodiment of the present application, Z of Chemical Formula 1 may be substituted again with one or more substituents selected from the group consisting of a C.sub.6 to C.sub.60 aryl group and a C.sub.6 to C.sub.60 heteroaryl group.
[0044] In another embodiment, Z of Chemical Formula 1 may be substituted again with one or more substituents selected from the group consisting of a pyridine group; a quinoline group; a pyrene group; and a pyrimidine group.
[0045] In one embodiment of the present application, L may be a direct bond; a substituted or unsubstituted C.sub.6 to C.sub.40 arylene group; or a substituted or unsubstituted C.sub.2 to C.sub.40 heteroarylene group.
[0046] In another embodiment, L may be a direct bond; a C.sub.6 to C.sub.40 arylene group unsubstituted or substituted with one or more substituents selected from the group consisting of a C.sub.1 to C.sub.40 alkyl group, a C.sub.6 to C.sub.40 aryl group and a C.sub.2 to C.sub.40 heteroaryl group; or a C.sub.2 to C.sub.40 heteroarylene group unsubstituted or substituted with one or more substituents selected from the group consisting of a C.sub.1 to C.sub.40 alkyl group, a C.sub.6 to C.sub.40 aryl group and a C.sub.2 to C.sub.40 heteroaryl group.
[0047] In another embodiment, L may be a direct bond; a phenylene group unsubstituted or substituted with one or more substituents selected from the group consisting of a pyridine group, a pyrimidine group, a quinoline group, a phenanthroline group, an anthracene group, a benzoquinoline group, a naphthyl group, a phenyl group, a triazine group, a dibenzofuran group, a dibenzothiophene group and a carbazole group; a divalent anthracene group unsubstituted or substituted with one or more substituents selected from the group consisting of a phenyl group, a naphthyl group, a pyridine group and a quinoline group; a biphenylene group; a naphthylene group; a phenalene group; a divalent phenanthrene group; or a divalent pyrene group.
[0048] In another embodiment, L may be a direct bond; a divalent pyridine group unsubstituted or substituted with one or more substituents selected from the group consisting of a phenyl group, a pyridine group, a pyrimidine group, a quinoline group, a phenanthroline group, an anthracene group, a benzoquinoline group, a naphthyl group and a triazine group; a divalent phenanthroline group unsubstituted or substituted with one or more substituents selected from the group consisting of a phenyl group and a methyl group; a divalent benzoquinoline group; a divalent pyrimidine group or a divalent quinoline group.
[0049] In one embodiment of the present application, L may be substituted again with one or more substituents selected from the group consisting of a C.sub.1 to C.sub.60 alkyl group; a C.sub.6 to C.sub.60 aryl group; and a C.sub.6 to C.sub.60 heteroaryl group.
[0050] In another embodiment, L may be substituted again with one or more substituents selected from the group consisting of a methyl group; a phenyl group; a biphenyl group; a naphthyl group; a pyridine group; a pyrimidine group; a quinoline group; and a benzoquinoline group.
[0051] According to one embodiment of the present application, Chemical Formula 1 may be represented by any one of the following Chemical Formulae 2 to 6.
##STR00002##
[0052] In Chemical Formulae 2 to 6,
[0053] L, R1 to R8, and m have the same definitions as in Chemical Formula 1,
[0054] at least one of X1 to X3 is N, and the rest are each independently N or CR23,
[0055] R9 to R23 are the same as or different from each other, and each independently selected from the group consisting of hydrogen; deuterium; a halogen group; --CN; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 alkenyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 alkynyl group; a substituted or unsubstituted C.sub.1 to C.sub.60 alkoxy group; a substituted or unsubstituted C.sub.3 to C.sub.60 cycloalkyl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heterocycloalkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group; --SiRR'R''; --P(.dbd.O)RR'; and an amine group unsubstituted or substituted with a C.sub.1 to C.sub.20 alkyl group, a C.sub.6 to C.sub.60 aryl group or a C.sub.2 to C.sub.60 heteroaryl group, or two or more groups adjacent to each other bond to each other to form a substituted or unsubstituted aliphatic or aromatic hydrocarbon ring, and
[0056] p, q, r and s are each independently an integer of 0 to 3.
[0057] In one embodiment of the present application, R23 is hydrogen.
[0058] In one embodiment of the present application, at least one of R1 and R6 and at least one of R11 and R16 of Chemical Formulae 2 to 6 are a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group, and the rest may be hydrogen or deuterium.
[0059] In another embodiment, at least one of R1 and R6 and at least one of R11 and R16 of Chemical Formulae 2 to 6 are a C.sub.6 to C.sub.60 aryl group, and the rest may be hydrogen or deuterium.
[0060] In another embodiment, at least one of R1 and R6 and at least one of R11 and R16 of Chemical Formulae 2 to 6 are a phenyl group, and the rest may be hydrogen.
[0061] In one embodiment of the present application, R2 to R5, R7 to R10, and R12 to R15 of Chemical Formulae 1 and 2 may be each independently hydrogen or deuterium.
[0062] In one embodiment of the present application, R, R' and R'' of Chemical Formulae 1 and 2 are the same as or different from each other, and may be each independently hydrogen; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; or a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group.
[0063] In one embodiment of the present application, R17 and R18 are the same as or different from each other, and may be each independently hydrogen; or a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group.
[0064] In another embodiment, R17 and R18 are the same as or different from each other, and may be each independently hydrogen; or a C.sub.6 to C.sub.60 aryl group.
[0065] In another embodiment, R17 and R18 are the same as or different from each other, and may be each independently hydrogen; a phenyl group; a naphthyl group; or a biphenyl group.
[0066] In one embodiment of the present application, R19 to R21 are the same as or different from each other, and may be each independently hydrogen; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; or a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl 10 group.
[0067] In another embodiment, R19 to R21 are the same as or different from each other, and may be each independently hydrogen; a C.sub.1 to C.sub.60 alkyl group; a C.sub.6 to C.sub.60 aryl group; or a C.sub.2 to C.sub.60 heteroaryl group.
[0068] In another embodiment, R19 to R21 are the same as or different from each other, and may be each independently hydrogen; a methyl group; a phenyl group; a pyridine group; a naphthyl group; a quinoline group; or a benzoquinoline group.
[0069] In one embodiment of the present application, R22 may be hydrogen; a C.sub.1 to C.sub.60 alkyl group; a C.sub.6 to C.sub.60 aryl group; or a C.sub.2 to C.sub.60 heteroaryl group.
[0070] In another embodiment, R22 may be hydrogen; a phenyl group unsubstituted or substituted with one or more substituents selected from the group consisting of a quinoline group, a pyridine group and a benzoquinoline group; a pyrene group; or a pyridine group.
[0071] In the present specification, the term "substituted or unsubstituted" means being substituted with one or more substituents selected from the group consisting of deuterium; a halogen group; --CN; a C.sub.1 to C.sub.60 alkyl group; a C.sub.2 to C.sub.60 alkenyl group; a C.sub.2 to C.sub.60 alkynyl group; a C.sub.3 to C.sub.60 cycloalkyl group; a C.sub.2 to C.sub.60 heterocycloalkyl group; a C.sub.6 to C.sub.60 aryl group; a C.sub.2 to C.sub.60 heteroaryl group; --SiRR'R''; --P(.dbd.O)RR'; a C.sub.1 to C.sub.20 alkylamine group; a C.sub.6 to C.sub.60 arylamine group; and a C.sub.2 to C.sub.60 heteroarylamine group, or being unsubstituted, or being substituted with a substituent bonding two or more of the above-mentioned substituents, or being substituted, or being substituted with a substituent linking two or more substituents selected from among the above-mentioned substituents, or being unsubstituted. For example, "a substituent linking two or more substituents" may comprise a biphenyl group. In other words, a biphenyl group may be an aryl group, or may be interpreted as a substituent linking two phenyl groups. The additional substituents may be further substituted. R, R' and R'' are the same as or different from each other, and each independently hydrogen; deuterium; --CN; a substituted or unsubstituted C.sub.1 to C.sub.60 alkyl group; a substituted or unsubstituted C.sub.3 to C.sub.60 cycloalkyl group; a substituted or unsubstituted C.sub.6 to C.sub.60 aryl group; or a substituted or unsubstituted C.sub.2 to C.sub.60 heteroaryl group.
[0072] According to one embodiment of the present application, the "substituted or unsubstituted" means being substituted with one or more substituents selected from the group consisting of deuterium, a halogen group, --CN, SiRR'R'', P(.dbd.O)RR', a C.sub.1 to C.sub.20 linear or branched alkyl group, a C.sub.6 to C.sub.60 aryl group, and a C.sub.2 to C.sub.60 heteroaryl group, or being unsubstituted, and
[0073] R, R' and R'' are the same as or different from each other, and each independently hydrogen; deuterium; --CN; a C.sub.1 to C.sub.60 alkyl group unsubstituted or substituted with deuterium, a halogen group, --CN, a C.sub.1 to C.sub.20 alkyl group, a C.sub.6 to C.sub.60 aryl group and a C.sub.2 to C.sub.60 heteroaryl group; a C.sub.3 to C.sub.60 cycloalkyl group unsubstituted or substituted with deuterium, halogen, --CN, a C.sub.1 to C.sub.20 alkyl group, a C.sub.6 to C.sub.60 aryl group and a C.sub.2 to C.sub.60 heteroaryl group; a C.sub.6 to C.sub.60 aryl group unsubstituted or substituted with deuterium, halogen, --CN, a C.sub.1 to C.sub.20 alkyl group, a C.sub.6 to C.sub.60 aryl group and a C.sub.2 to C.sub.60 heteroaryl group; or a C.sub.2 to C.sub.60 heteroaryl group unsubstituted or substituted with deuterium, halogen, --CN, a C.sub.1 to C.sub.20 alkyl group, a C.sub.6 to C.sub.60 aryl group and a C.sub.2 to C.sub.60 heteroaryl group.
[0074] The term "substituted" means a hydrogen atom bonding to a carbon atom of a compound is changed to another substituent, and the position of substitution is not limited as long as it is a position at which the hydrogen atom is substituted, that is, a position at which a substituent can substitute, and when two or more substituents substitute, the two or more substituents may be the same as or different from each other.
[0075] In the present specification, the halogen may be fluorine, chlorine, bromine or iodine.
[0076] In the present specification, the alkyl group comprises linear or branched having 1 to 60 carbon atoms, and may be further substituted with other substituents. The number of carbon atoms of the alkyl group may be from 1 to 60, specifically from 1 to 40 and more specifically from 1 to 20. Specific examples thereof may comprise a methyl group, an ethyl group, a propyl group, an n-propyl group, an isopropyl group, a butyl group, an n-butyl group, an isobutyl group, a tert-butyl group, a sec-butyl group, a 1-methyl-butyl group, a 1-ethylbutyl group, a pentyl group, an n-pentyl group, an isopentyl group, a neopentyl group, a tert-pentyl group, a hexyl group, an n-hexyl group, a 1-methylpentyl group, a 2-methylpentyl group, a 4-methyl-2-pentyl group, a 3,3-dimethylbutyl group, a 2-ethylbutyl group, a heptyl group, an n-heptyl group, a 1-methylhexyl group, a cyclopentylmethyl group, a cyclohexylmethyl group, an octyl group, an n-octyl group, a tert-octyl group, a 1-methylheptyl group, a 2-ethylhexyl group, a 2-propylpentyl group, an n-nonyl group, a 2,2-dimethylheptyl group, a 1-ethyl-propyl group, a 1,1-dimethyl-propyl group, an isohexyl group, a 2-methylpentyl group, a 4-methylhexyl group, a 5-methylhexyl group and the like, but are not limited thereto.
[0077] In the present specification, the alkenyl group comprises linear or branched having 2 to 60 carbon atoms, and may be further substituted with other substituents. The number of carbon atoms of the alkenyl group may be from 2 to 60, specifically from 2 to 40 and more specifically from 2 to 20. Specific examples thereof may comprise a vinyl group, a 1-propenyl group, an isopropenyl group, a 1-butenyl group, a 2-butenyl group, a 3-butenyl group, a 1-pentenyl group, a 2-pentenyl group, a 3-pentenyl group, a 3-methyl-1-butenyl group, a 1,3-butadienyl group, an allyl group, a 1-phenylvinyl-1-yl group, a 2-phenylvinyl-1-yl group, a 2,2-diphenylvinyl-1-yl group, a 2-phenyl-2-(naphthyl-1-yl)vinyl-1-yl group, a 2,2-bis(diphenyl-1-yl)vinyl-1-yl group, a stilbenyl group, a styrenyl group and the like, but are not limited thereto.
[0078] In the present specification, the alkynyl group comprises linear or branched having 2 to 60 carbon atoms, and may be further substituted with other substituents. The number of carbon atoms of the alkynyl group may be from 2 to 60, specifically from 2 to 40 and more specifically from 2 to 20.
[0079] In the present specification, the cycloalkyl group comprises monocyclic or multicyclic having 3 to 60 carbon atoms, and may be further substituted with other substituents. Herein, the multicyclic means a group in which the cycloalkyl group is directly linked to or fused with other cyclic groups. Herein, the other cyclic groups may be a cycloalkyl group, however, may also be different types of cyclic groups such as a heterocycloalkyl group, an aryl group and a heteroaryl group. The number of carbon groups of the cycloalkyl group may be from 3 to 60, specifically from 3 to 40 and more specifically from 5 to 20. Specific examples thereof may comprise a cyclopropyl group, a cyclobutyl group, a cyclopentyl group, a 3-methylcyclopentyl group, a 2,3-dimethylcyclopentyl group, a cyclohexyl group, a 3-methylcyclohexyl group, a 4-methylcyclohexyl group, a 2,3-dimethylcyclohexyl group, a 3,4,5-trimethylcyclohexyl group, a 4-tert-butylcyclohexyl group, a cycloheptyl group, a cyclooctyl group and the like, but are not limited thereto.
[0080] In the present specification, the heterocycloalkyl group comprises O, S, Se, N or Si as a heteroatom, comprises monocyclic or multicyclic having 2 to 60 carbon atoms, and may be further substituted with other substituents. Herein, the multicyclic means a group in which the heterocycloalkyl group is directly linked to or fused with other cyclic groups. Herein, the other cyclic groups may be a heterocycloalkyl group, however, may also be different types of cyclic groups such as a cycloalkyl group, an aryl group and a heteroaryl group. The number of carbon atoms of the heterocycloalkyl group may be from 2 to 60, specifically from 2 to 40 and more specifically from 3 to 20.
[0081] In the present specification, the aryl group comprises monocyclic or multicyclic having 6 to 60 carbon atoms, and may be further substituted with other substituents. Herein, the multicyclic means a group in which the aryl group is directly linked to or fused with other cyclic groups. Herein, the other cyclic groups may be an aryl group, however, may also be different types of cyclic groups such as a cycloalkyl group, a heterocycloalkyl group and a heteroaryl group. The aryl group comprises a spiro group. The number of carbon atoms of the aryl group may be from 6 to 60, specifically from 6 to 40 and more specifically from 6 to 25. Specific examples of the aryl group may comprise a phenyl group, a biphenyl group, a triphenyl group, a naphthyl group, an anthryl group, a chrysenyl group, a phenanthrenyl group, a perylenyl group, a fluoranthenyl group, a triphenylenyl group, a phenalenyl group, a pyrenyl group, a tetracenyl group, a pentacenyl group, a fluorenyl group, an indenyl group, an acenaphthylenyl group, a benzofluorenyl group, a spirobifluorenyl group, a 2,3-dihydro-1H-indenyl group, a fused ring thereof, and the like, but are not limited thereto.
[0082] In the present specification, the spiro group is a group comprising a spiro structure, and may have 15 to 60 carbon atoms. For example, the spiro group may comprise a structure in which a 2,3-dihydro-1H-indene group or a cyclohexane group spiro bonds to a fluorenyl group. Specifically, the following spiro group may comprise any one of the groups having the following structural formulae.
##STR00003##
[0083] In the present specification, the heteroaryl group comprises O, S, Se, N or Si as a heteroatom, comprises monocyclic or multicyclic having 2 to 60 carbon atoms, and may be further substituted with other substituents. Herein, the multicyclic means a group in which the heteroaryl group is directly linked to or fused with other cyclic groups. Herein, the other cyclic groups may be a heteroaryl group, however, may also be different types of cyclic groups such as a cycloalkyl group, a heterocycloalkyl group and an aryl group. The number of carbon atoms of the heteroaryl group may be from 2 to 60, specifically from 2 to 40 and more specifically from 3 to 25. Specific examples of the heteroaryl group may comprise a pyridyl group, a pyrrolyl group, a pyrimidyl group, a pyridazinyl group, a furanyl group, a thiophene group, an imidazolyl group, a pyrazolyl group, an oxazolyl group, an isoxazolyl group, a thiazolyl group, an isothiazolyl group, a triazolyl group, a furazanyl group, an oxadiazolyl group, a thiadiazolyl group, a dithiazolyl group, a tetrazolyl group, a pyranyl group, a thiopyranyl group, a diazinyl group, an oxazinyl group, a thiazinyl group, a dioxynyl group, a triazinyl group, a tetrazinyl group, a quinolyl group, an isoquinolyl group, a quinazolinyl group, an isoquinazolinyl group, a qninozolinyl group, a naphthyridyl group, an acridinyl group, a phenanthridinyl group, an imidazopyridinyl group, a diazanaphthalenyl group, a triazaindene group, an indolyl group, an indolizinyl group, a benzothiazolyl group, a benzoxazolyl group, a benzimidazolyl group, a benzothiophene group, a benzofuran group, a dibenzothiophene group, a dibenzofuran group, a carbazolyl group, a benzocarbazolyl group, a dibenzocarbazolyl group, a phenazinyl group, a dibenzosilole group, spirobi(dibenzosilole), a dihydrophenazinyl group, a phenoxazinyl group, a phenanthridyl group, an imidazopyridinyl group, a thienyl group, an indolo[2,3-a]carbazolyl group, an indolo[2,3-b]carbazolyl group, an indolinyl group, a 10,11-dihydro-dibenzo[b,f]azepine group, a 9,10-dihydroacridinyl group, a phenanthrazinyl group, a phenothiathiazinyl group, a phthalazinyl group, a naphthylidinyl group, a phenanthrolinyl group, a benzo[c][1,2,5]thiadiazolyl group, a 5,10-dihydrobenzo[b,e][1,4]azasilinyl, a pyrazolo[1,5-c]quinazolinyl group, a pyrido[1,2-b]indazolyl group, a pyrido[1,2-a]imidazo[1,2-e]indolinyl group, a 5,11-dihydroindeno[1,2-b]carbazolyl group and the like, but are not limited thereto.
[0084] In the present specification, the amine group may be selected from the group consisting of a monoalkylamine group; a monoarylamine group; a monoheteroarylamine group; --NH.sub.2; a dialkylamine group; a diarylamine group; a diheteroarylamine group; an alkylarylamine group; an alkylheteroarylamine group; and an arylheteroarylamine group, and although not particularly limited thereto, the number of carbon atoms is preferably from 1 to 30. Specific examples of the amine group may comprise a methylamine group, a dimethylamine group, an ethylamine group, a diethylamine group, a phenylamine group, a naphthylamine group, a biphenylamine group, a dibiphenylamine group, an anthracenylamine group, a 9-methyl-anthracenylamine group, a diphenylamine group, a phenylnaphthylamine group, a ditolylamine group, a phenyltolylamine group, a triphenylamine group, a biphenylnaphthylamine group, a phenylbiphenylamine group, a biphenylfluorenylamine group, a phenyltriphenylenylamine group, a biphenyltriphenylenylamine group and the like, but are not limited thereto.
[0085] In the present specification, the arylene group means the aryl group having two bonding sites, that is, a divalent group. Descriptions on the aryl group provided above may be applied thereto except for each being a divalent. In addition, the heteroarylene group means the heteroaryl group having two bonding sites, that is, a divalent group. Descriptions on the heteroaryl group provided above may be applied thereto except for each being a divalent.
[0086] According to one embodiment of the present application, Chemical Formula 1 may be represented by any one of the following compounds, but is not limited thereto.
##STR00004## ##STR00005## ##STR00006## ##STR00007## ##STR00008## ##STR00009## ##STR00010## ##STR00011## ##STR00012## ##STR00013## ##STR00014## ##STR00015## ##STR00016## ##STR00017## ##STR00018## ##STR00019## ##STR00020## ##STR00021## ##STR00022## ##STR00023## ##STR00024## ##STR00025## ##STR00026## ##STR00027## ##STR00028## ##STR00029## ##STR00030## ##STR00031## ##STR00032## ##STR00033## ##STR00034## ##STR00035## ##STR00036## ##STR00037## ##STR00038## ##STR00039## ##STR00040## ##STR00041## ##STR00042## ##STR00043## ##STR00044## ##STR00045##
[0087] In addition, by introducing various substituents to the structure of Chemical Formula 1, compounds having unique properties of the introduced substituents may be synthesized. For example, by introducing substituents normally used as hole injection layer materials, hole transfer layer materials, light emitting layer materials, electron transfer layer materials and charge generation layer materials used for manufacturing an organic light emitting device to the core structure, materials satisfying conditions required for each organic material layer may be synthesized.
[0088] In addition, by introducing various substituents to the structure of Chemical Formula 1, the energy band gap may be finely controlled, and meanwhile, properties at interfaces between organic materials are enhanced, and material applications may become diverse.
[0089] Meanwhile, the hetero-cyclic compound has excellent thermal stability with a high glass transition temperature (Tg). Such an increase in the thermal stability becomes an important factor in providing driving stability to a device.
[0090] The hetero-cyclic compound according to one embodiment of the present application may be prepared through a multistep chemical reaction. Some intermediate compounds are prepared first, and the compound of Chemical Formula 1 may be prepared from the intermediate compounds. More specifically, the hetero-cyclic compound according to one embodiment of the present application may be prepared based on preparation examples to be described below.
[0091] Another embodiment of the present application provides an organic light emitting device comprising the hetero-cyclic compound represented by Chemical Formula 1.
[0092] The organic light emitting device according to one embodiment of the present application may be manufactured using common organic light emitting device manufacturing methods and materials except that one or more organic material layers are formed using the hetero-cyclic compound described above.
[0093] The hetero-cyclic compound may be formed into an organic material layer through a solution coating method as well as a vacuum deposition method when manufacturing the organic light emitting device. Herein, the solution coating method means spin coating, dip coating, inkjet printing, screen printing, a spray method, roll coating and the like, but is not limited thereto.
[0094] Specifically, the organic light emitting device according to one embodiment of the present application comprises an anode, a cathode, and one or more organic material layers provided between the anode and the cathode, wherein one or more layers of the organic material layers comprise the hetero-cyclic compound represented by Chemical Formula 1.
[0095] FIGS. 1 to 3 illustrate a lamination order of electrodes and organic material layers of an organic light emitting device according to one embodiment of the present application. However, the scope of the present application is not limited to these diagrams, and structures of organic light emitting devices known in the art may also be used in the present application.
[0096] FIG. 1 illustrates an organic light emitting device in which an anode (200), an organic material layer (300) and a cathode (400) are consecutively laminated on a substrate (100). However, the structure is not limited to such a structure, and as illustrated in FIG. 2, an organic light emitting device in which a cathode, an organic material layer and an anode are consecutively laminated on a substrate may also be obtained.
[0097] FIG. 3 illustrates a case of the organic material layer being a multilayer. The organic light emitting device according to FIG. 3 comprises a hole injection layer (301), a hole transfer layer (302), a light emitting layer (303), a hole blocking layer (304), an electron transfer layer (305) and an electron injection layer (306). However, the scope of the present application is not limited to such a lamination structure, and as necessary, other layers except the light emitting layer may not be included, and other necessary functional layers may be further included.
[0098] In addition, the organic light emitting device according to one embodiment of the present application comprises an anode, a cathode, and two or more stacks provided between the anode and the cathode, wherein the two or more stacks each independently comprise a light emitting layer, a charge generation layer is included between the two or more stacks, and the charge generation layer comprises the hetero-cyclic compound represented by Chemical Formula 1.
[0099] In addition, the organic light emitting device according to one embodiment of the present application comprises an anode, a first stack provided on the anode and comprising a first light emitting layer, a charge generation layer provided on the first stack, a second stack provided on the charge generation layer and comprising a second light emitting layer, and a cathode provided on the second stack. Herein, the charge generation layer may comprise the hetero-cyclic compound represented by Chemical Formula 1. In addition, the first stack and the second stack may each independently further comprise one or more types of the hole injection layer, the hole transfer layer, the hole blocking layer, the electron transfer layer, the electron injection layer described above and the like.
[0100] The charge generation layer may be an N-type charge generation layer, and the charge generation layer may further comprise a dopant known in the art in addition to the hetero-cyclic compound represented by Chemical Formula 1.
[0101] As the organic light emitting device according to one embodiment of the present application, an organic light emitting device having a 2-stack tandem structure is schematically illustrated in FIG. 4.
[0102] Herein, the first electron blocking layer, the first bole blocking layer, the second hole blocking layer and the like described in FIG. 4 may not be included in some cases.
[0103] The organic light emitting device according to the present specification may be manufactured using materials and methods known in the art except that one or more layers of the organic material layers comprise the hetero-cyclic compound represented by Chemical Formula 1.
[0104] The hetero-cyclic compound represented by Chemical Formula 1 may form one or more layers of the organic material layers of the organic light emitting device alone. However, as necessary, the hetero-cyclic compound represented by Chemical Formula 1 may be mixed with other materials to form the organic material layers.
[0105] The hetero-cyclic compound represented by Chemical Formula 1 may be used as a material of the charge generation layer in the organic light emitting device.
[0106] The hetero-cyclic compound represented by Chemical Formula 1 may be used as a material of the electron transfer layer, the hole blocking layer, the light emitting layer or the like in the organic light emitting device. As one example, the hetero-cyclic compound represented by Chemical Formula 1 may be used as a material of the electron transfer layer, the hole transfer layer or the light emitting layer in the organic light emitting device.
[0107] In addition, the hetero-cyclic compound represented by Chemical Formula 1 may be used as a material of the light emitting layer in the organic light emitting device. As one example, the hetero-cyclic compound represented by Chemical Formula 1 may be used as a phosphorescent host material of the light emitting layer in the organic light emitting device.
[0108] In the organic light emitting device according to one embodiment of the present application, materials other than the hetero-cyclic compound of Chemical Formula 1 are illustrated below, however, these are for illustrative purposes only and not for limiting the scope of the present application, and may be replaced by materials known in the art.
[0109] As the anode material, materials having relatively large work function may be used, and transparent conductive oxides, metals, conductive polymers or the like may be used. Specific examples of the anode material comprise metals such as vanadium, chromium, copper, zinc and gold, or alloys thereof; metal oxides such as zinc oxide, indium oxide, indium tin oxide (ITO) and indium zinc oxide (IZO); combinations of metals and oxides such as ZnO:Al or SnO.sub.2:Sb; conductive polymers such as poly(3-methylcompound), poly[3,4-(ethylene-1,2-dioxy)compound](PEDOT), polypyrrole and polyaniline, but are not limited thereto.
[0110] As the cathode material, materials having relatively small work function may be used, and metals, metal oxides, conductive polymers or the like may be used. Specific examples of the cathode material comprise metals such as magnesium, calcium, sodium, potassium, titanium, indium, yttrium, lithium, gadolinium, aluminum, silver, tin and lead, or alloys thereof; multilayer structure materials such as LiF/Al or LiO.sub.2/Al, and the like, but are not limited thereto.
[0111] As the hole injection material, known hole injection materials may be used, and for example, phthalocyanine compounds such as copper phthalocyanine disclosed in U.S. Pat. No. 4,356,429, or starburst-type amine derivatives such as tris(4-carbazoyl-9-ylphenyl)amine (TCTA), 4,4',4''-tri[phenyl(m-tolyl)amino]triphenylamine (m-MTDATA) or 1,3,5-tris[4-(3-methylphenylphenylamino)phenyl]benzene (m-MTDAPB) described in the literature [Advanced Material, 6, p. 677 (1994)], polyaniline/dodecylbenzene sulfonic acid, poly(3,4-ethylenedioxythiophene)/poly(4-styrenesulfonate), polyaniline/camphor sulfonic acid or polyaniline/poly(4-styrene-sulfonate) that are conductive polymers having solubility, and the like, may be used.
[0112] As the hole transfer material, pyrazoline derivatives, arylamine-based derivatives, stilbene derivatives, triphenyldiamine derivatives and the like may be used, and low molecular or high molecular materials may also be used.
[0113] As the electron transfer material, metal complexes of oxadiazole derivatives, anthraquinodimethane and derivatives thereof, benzoquinone and derivatives thereof, naphthoquinone and derivatives thereof, anthraquinone and derivatives thereof, tetracyanoanthraquinodimethane and derivatives thereof, fluorenone derivatives, diphenyldicyanoethylene and derivatives thereof, diphenoquinone derivatives, 8-hydroxyquinoline and derivatives thereof, and the like, may be used, and high molecular materials may also be used as well as low molecular materials.
[0114] As examples of the electron injection material, LiF is typically used in the art, however, the present application is not limited thereto.
[0115] As the light emitting material, red, green or blue light emitting materials may be used, and as necessary, two or more light emitting materials may be mixed and used. In addition, fluorescent materials may also be used as the light emitting material, however, phosphorescent materials may also be used. As the light emitting material, materials emitting light by bonding electrons and holes injected from an anode and a cathode, respectively, may be used alone, however, materials having a host material and a dopant material involved in light emission together may also be used.
[0116] The organic light emitting device according to one embodiment of the present application may be a top-emission type, a bottom-emission type or a dual-emission type depending on the materials used.
[0117] The hetero-cyclic compound according to one embodiment of the present application may also be used in an organic electronic device comprising an organic solar cell, an organic photo conductor, an organic transistor and the like under a similar principle used in the organic light emitting device.
[0118] Hereinafter, the present specification will be described in more detail with reference to examples, however, these are for illustrative purposes only, and the scope of the present application is not limited thereto.
EXAMPLE
<Preparation Example 1> Preparation of Compound 3
##STR00046##
[0120] 1) Preparation of Compound 3-2
[0121] A 3-amino-2-naphthaldehyde compound (16.8 g, 85 mmol) and 1,1'-(5-bromo-1,3-phenylene)diethanone (12.5 g, 40 mmol) were introduced to EtOH (300 mL), 5 mL of a KOH solution saturated in EtOH was added dropwise thereto, and then the result was refluxed for 2 hours. After the reaction was completed, the result was cooled to room temperature, and extracted with distilled water and Mc. After the organic layer was dried with anhydrous Na.sub.2O.sub.4, the solvent was removed using a rotary evaporator, and the result was purified using column chromatography with ethyl acetate and hexane as a developing solvent to obtain target Compound 3-2 (27 g, 95%).
[0122] 2) Preparation of Compound 3-1
[0123] After placing Compound 3-2 (13.5 g, 40 mmol), 4,4,4',4',5,5,5',5'-octamethyl-2,2'-bi(1,3,2-dioxaborolane) (20 g, 80 mmol), Pd(dppf).sub.2Cl.sub.2 (1.4 g, 2 mmol) and KOAc (11.7 g, 120 mmol) in a reactor, 0.3 M dioxane was added thereto, and the result was refluxed for 12 hours. After the reaction was completed, the result was cooled to room temperature, and extracted with distilled water and EA. After the organic layer was dried with anhydrous Na.sub.2O.sub.4, the solvent was removed using a rotary evaporator, and the result was purified using column chromatography with dichloromethane and hexane as a developing solvent to obtain target Compound 3-1 (12.2 g, 80%).
[0124] 3) Preparation of Compound 3
[0125] Compound 3-1 (7.6 g, 20 mmol) and iodobenzene (5.7 g, 20 mmol) were dissolved in toluene (80 mL), then Pd(PPh.sub.3).sub.4 (1.1 g, 1 mmol) and K.sub.2CO.sub.3 (8.3 g, 60 mmol) were added thereto, and the result was stirred for 10 minutes. After that, H.sub.2O (16 mL) and EtOH (16 mL) were added thereto, and the result was refluxed for 12 hours. After the reaction was completed, the result was cooled to room temperature, and extracted with distilled water and Mc. After the organic layer was dried with anhydrous Na.sub.2O.sub.4, the solvent was removed using a rotary evaporator, and the result was purified using column chromatography with ethyl acetate and hexane as a developing solvent to obtain target Compound 3 (8.3 g, 95%).
[0126] Target Compound A was synthesized in the same manner as the preparation of Compound 3 except that, in Preparation Example 1, Intermediate A of the following Table 1 was used instead of iodobenzene.
TABLE-US-00001 TABLE 1 Com- pound Number Intermediate A Target Compound A Yield 57 ##STR00047## ##STR00048## 85% 62 ##STR00049## ##STR00050## 82% 63 ##STR00051## ##STR00052## 88% 138 ##STR00053## ##STR00054## 78% 142 ##STR00055## ##STR00056## 72% 147 ##STR00057## ##STR00058## 81% 148 ##STR00059## ##STR00060## 83%
<Preparation Example 2> Preparation of Compound 41
##STR00061##
[0128] 1) Preparation of Compound 41-3
[0129] A 4-phenyl-1H-benzo[f]isoindole-1,3(2H)-dione compound (20 g, 73 mmol), KOBr (9.9 g, 73 mmol) and KOH (10.2 g, 183 mMol) were introduced to H.sub.2O (300 mL), and the result was refluxed for 2 hours. After the reaction was completed, the result was cooled to room temperature, and extracted with distilled water and Mc. After the organic layer was dried with anhydrous Na.sub.2O.sub.4, the solvent was removed using a rotary evaporator, and the result was purified using column chromatography with ethyl acetate and hexane as a developing solvent to obtain target Compound 41-3 (15 g, 78%).
[0130] 2) Preparation of Compound 41-2
[0131] LiAlH.sub.4 (3.5 g, 91 mmol) was introduced to THF (50 mL), and the temperature was maintained at 0.degree. C. Compound 41-3 (15 g, 57 mmol) was dissolved in THF (50 mL) and slowly added dropwise thereto. After that, the temperature was slowly raised to room temperature, and the result was stirred for 12 hours. After the reaction was completed, the result was extracted with distilled water and Mc. After the organic layer was dried with anhydrous Na.sub.2O.sub.4, the solvent was removed using a rotary evaporator, and the result was purified using column chromatography with ethyl acetate and hexane as a developing solvent to obtain target Compound 41-2 (13 g, 92%).
[0132] 3) Preparation of Compound 41-1
[0133] After dissolving Compound 41-2 (13 g, 52 mmol) in MC (250 mL), MnO.sub.2 (18 g, 208 mmol) was slowly added thereto, and the result was stirred for 1 hour at room temperature. After the reaction was completed, the result was Celite filtered, and the organic layer was rotary evaporated to remove the solvent. The result was purified using column chromatography with ethyl acetate and hexane as a developing solvent to obtain target Compound 41-1 (10 g, 77%).
[0134] 4) Preparation of Compound 41
[0135] Compound 41-1 (10 g, 40 mmol) and 1,1'-(1,3-phenylene)diethanone (3.5 g, 20 mmol) were introduced to EtOH (50 mL), 5 mL of a KOH solution saturated in EtOH was added dropwise thereto, and then the result was refluxed for 2 hours. After the reaction was completed, the result was cooled to room temperature, and extracted with distilled water and Mc. After the organic layer was dried with anhydrous Na.sub.2O.sub.4, the solvent was removed using a rotary evaporator, and the result was purified using column chromatography with ethyl acetate and hexane as a developing solvent to obtain target Compound 41 (7 g, 60%).
[0136] Target Compound B was synthesized in the same manner as the preparation of Compound 41 except that, in Preparation Example 2, Intermediate B of the following Table 2 was used instead of 1,1'-(1,3-phenylene)diethanone.
TABLE-US-00002 TABLE 2 Compound Number Intermediate B Target Compound B Yield 42 ##STR00062## ##STR00063## 55% 43 ##STR00064## ##STR00065## 65% 44 ##STR00066## ##STR00067## 68% 45 ##STR00068## ##STR00069## 50% 46 ##STR00070## ##STR00071## 54% 47 ##STR00072## ##STR00073## 61%
<Preparation Example 3> Preparation of Compound 88
##STR00074##
[0138] 1) Preparation of Compound 88-2
[0139] A 3-amino-2-naphthaldehyde compound (16.8 g, 85 mmol) and 1,1'-(4-bromopyridine-2,6-diyl) diethanone (12.5 g, 40 mmol) were introduced to EtOH (300 mL), 5 mL of a KOH solution saturated in EtOH was added dropwise thereto, and then the result was refluxed for 2 hours. After the reaction was completed, the result was cooled to room temperature, and extracted with distilled water and Mc. After the organic layer was dried with anhydrous Na.sub.2O.sub.4, the solvent was removed using a rotary evaporator, and the result was purified using column chromatography with ethyl acetate and hexane as a developing solvent to obtain target Compound 88-2 (27 g, 95%).
[0140] 2) Preparation of Compound 88-1
[0141] After placing Compound 88-2 (13.5 g, 40 mmol), 4,4,4',4',5,5,5',5'-octamethyl-2,2'-bi(1,3,2-dioxaborolane) (20 g, 80 mmol), Pd(dppf).sub.2Cl.sub.2 (1.4 g, 2 mmol) and KOAc (11.7 g, 120 mmol) in a reactor, 0.3 M dioxane was added thereto, and the result was refluxed for 12 hours. After the reaction was completed, the result was cooled to room temperature, and extracted with distilled water and EA. After the organic layer was dried with anhydrous Na.sub.2O.sub.4, the solvent was removed using a rotary evaporator, and the result was purified using column chromatography with dichloromethane and hexane as a developing solvent to obtain target Compound 88-1 (14 g, 90%).
[0142] 3) Preparation of Compound 88
[0143] Compound 88-1 (7.6 g, 20 mmol) and 2-bromopyridine (4.7 g, 20 mmol) were dissolved in toluene (80 mL), then Pd(PPh.sub.3).sub.4 (1.1 g, 1 mmol) and K.sub.2CO.sub.3 (8.3 g, 60 mmol) were added thereto, and the result was stirred for 10 minutes. After that, H.sub.2O (16 mL) and EtOH (16 mL) were added thereto, and the result was refluxed for 12 hours. After the reaction was completed, the result was cooled to room temperature, and extracted with distilled water and Mc. After the organic layer was dried with anhydrous Na.sub.2O.sub.4, the solvent was removed using a rotary evaporator, and the result was purified using column chromatography with ethyl acetate and hexane as a developing solvent to obtain target Compound 88 (7.3 g, 85%).
[0144] Target Compound C was synthesized in the same manner as the preparation of Compound 88 except that, in Preparation Example 3, Intermediate C of the following Table 3 was used instead of 2-bromopyridine.
TABLE-US-00003 TABLE 3 Compound Number Intermediate C Target Compound C Yield 89 ##STR00075## ##STR00076## 78% 90 ##STR00077## ##STR00078## 81% 91 ##STR00079## ##STR00080## 91% 92 ##STR00081## ##STR00082## 78% 93 ##STR00083## ##STR00084## 72% 94 ##STR00085## ##STR00086## 81% 95 ##STR00087## ##STR00088## 83%
<Preparation Example 4> Preparation of Compound 164
##STR00089##
[0146] 1) Preparation of Compound 164-2
[0147] A 3-amino-2-naphthaldehyde compound (16.8 g, 85 mmol) and 1-(3-bromophenyl)ethan-1-one (16.9 g, 85 mmol) were introduced to EtOH (300 mL), 5 mL of a KOH solution saturated in EtOH was added dropwise thereto, and then the result was refluxed for 2 hours. After the reaction was completed, the result was cooled to room temperature, and extracted with distilled water and Mc. After the organic layer was dried with anhydrous Na.sub.2O.sub.4, the solvent was removed using a rotary evaporator, and the result was purified using column chromatography with ethyl acetate and hexane as a developing solvent to obtain target Compound 164-2 (27 g, 95%).
2) Preparation of Compound 164-1
[0148] After placing Compound 164-2 (13.5 g, 40 mmol), 4,4,4',4',5,5,5',5'-octamethyl-2,2'-bi(1,3,2-dioxaborolane) (20 g, 80 mmol), Pd(dppf).sub.2Cl.sub.2 (1.4 g, 2 mmol) and KOAc (11.7 g, 120 mmol) in a reactor, 0.3 M dioxane was added thereto, and the result was refluxed for 12 hours. After the reaction was completed, the result was cooled to room temperature, and extracted with distilled water and EA. After the organic layer was dried with anhydrous Na.sub.2O.sub.4, the solvent was removed using a rotary evaporator, and the result was purified using column chromatography with dichloromethane and hexane as a developing solvent to obtain target Compound 164-1 (12.2 g, 80%).
[0149] 3) Preparation of Compound 164
[0150] Compound 164-1 (7.6 g, 20 mmol) and 2-chloro-4,6-diphenyl-1,3,5-triazine (5.7 g, 20 mmol) were dissolved in toluene (80 mL), then Pd(PPh.sub.3).sub.4 (1.1 g, 1 mmol) and K.sub.2CO.sub.3 (8.3 g, 60 mmol) were added thereto, and the result was stirred for 10 minutes. After that, H.sub.2O (16 mL) and EtOH (16 mL) were added thereto, and the result was refluxed for 12 hours. After the reaction was completed, the result was cooled to room temperature, and extracted with distilled water and Mc. After the organic layer was dried with anhydrous Na.sub.2O.sub.4, the solvent was removed using a rotary evaporator, and the result was purified using column chromatography with ethyl acetate and hexane as a developing solvent to obtain target Compound 164 (7 g, 75%).
[0151] Target Compound D was synthesized in the same manner as the preparation of Compound 164 except that, in Preparation Example 4, Intermediate D of the following Table 4 was used instead of 2-chloro-4,6-diphenyl-1,3,5-triazine.
TABLE-US-00004 TABLE 4 Com- pound Number Intermediate D Target Compound D Yield 169 ##STR00090## ##STR00091## 75% 192 ##STR00092## ##STR00093## 90% 193 ##STR00094## ##STR00095## 83% 210 ##STR00096## ##STR00097## 81% 228 ##STR00098## ##STR00099## 83% 230 ##STR00100## ##STR00101## 85% 247 ##STR00102## ##STR00103## 89%
<Preparation Example 5> Preparation of Compound 226
##STR00104##
[0153] 1) Preparation of Compound 226-2
[0154] A 1-(3,5-dibromophenyl)ethan-1-one compound (11.1 g, 40 mmol) and 2-amino-5-bromobenzaldehyde (6.8 g, 40 mmol) were introduced to EtOH (300 mL), 2 mL of a KOH solution saturated in EtOH was added dropwise thereto, and then the result was refluxed for 4 hours. After the reaction was completed, the result was cooled to room temperature, and extracted with distilled water and Mc. After the organic layer was dried with anhydrous Na.sub.2O.sub.4, the solvent was removed using a rotary evaporator, and the result was purified using column chromatography with ethyl acetate and hexane as a developing solvent to obtain target Compound 226-2 (11.5 g, 70%).
[0155] 2) Preparation of Compound 226-1
[0156] After placing Compound 226-2 (13.5 g, 40 mmol), 4,4,4',4',5,5,5',5'-octamethyl-2,2'-bi(1,3,2-dioxaborolane) (25 g, 100 mmol), Pd(dppf).sub.2Cl.sub.2 (1.4 g, 2 mmol) and KOAc (11.7 g, 120 mmol) in a reactor, 0.3 M dioxane was added thereto, and the result was refluxed for 12 hours. After the reaction was completed, the result was cooled to room temperature, and extracted with distilled water and EA. After the organic layer was dried with anhydrous Na.sub.2O.sub.4, the solvent was removed using a rotary evaporator, and the result was purified using column chromatography with dichloromethane and hexane as a developing solvent to obtain target Compound 226-1 (12.2 g, 80%).
[0157] 3) Preparation of Compound 226
[0158] Compound 226-1 (4.1 g, 10 mmol) and 5-bromo-1,10-phenanthroline (3.3 g, 10 mmol) were dissolved in toluene (30 mL), then Pd(PPh.sub.3).sub.4 (0.6 g, 0.5 mmol) and K.sub.2CO.sub.3 (2.8 g, 30 mmol) were added thereto, and the result was stirred for 10 minutes. After that, H.sub.2O (6 mL) and EtOH (6 mL) were added thereto, and the result was refluxed for 12 hours. After the reaction was completed, the result was cooled to room temperature, and extracted with distilled water and Mc. After the organic layer was dried with anhydrous Na.sub.2O.sub.4, the solvent was removed using a rotary evaporator, and the result was purified using column chromatography with ethyl acetate and hexane as a developing solvent to obtain target Compound 226 (5.0 g, 76%).
[0159] 4) Preparation of Compound 203
[0160] Compound 226-2 (8 g, 20 mmol) and 9H-carbazole (3.4 g, 20 mmol) were dissolved in toluene (310 mL), then Pd.sub.2dba.sub.3 (0.2 g, 0.2 mmol), 2 M (t-Bu).sub.3P (0.2 mL, 0.4 mmol) and NaOt-Bu (5.4 g, 60 mmol) were added thereto, and the result was refluxed for 12 hours. After the reaction was completed, the result was cooled to room temperature, and extracted with distilled water and Mc. After the organic layer was dried with anhydrous Na.sub.2SO.sub.4, the solvent was removed using a rotary evaporator, and the result was purified using column chromatography with ethyl acetate and hexane as a developing solvent to obtain target Compound 203 (10.4 g, 89%).
[0161] Target Compound E was synthesized in the same manner as the preparation of Compound 226 except that, in Preparation Example 5, Intermediate E of the following Table 5 was used instead of 5-bromo-1,10-phenanthroline.
TABLE-US-00005 TABLE 5 Com- pound Number Intermediate E Target Compound E Yield 203 ##STR00105## ##STR00106## 89% 260 ##STR00107## ##STR00108## 78% 261 ##STR00109## ##STR00110## 86%
<Preparation Example 6> Preparation of Compound 195
##STR00111##
[0163] 1) Preparation of Compound 195-2
[0164] A 3-amino-2-naphthaldehyde compound (16.8 g, 85 mmol) and acetyl bromide (10.5 g, 85 mmol) were introduced to EtOH (300 mL), 5 mL of a KOH solution saturated in EtOH was added dropwise thereto, and then the result was refluxed for 2 hours. After the reaction was completed, the result was cooled to room temperature, and extracted with distilled water and Mc. After the organic layer was dried with anhydrous Na.sub.2O.sub.4, the solvent was removed using a rotary evaporator, and the result was purified using column chromatography with ethyl acetate and hexane as a developing solvent to obtain target Compound 195-2 (19.7 g, 90%).
[0165] 2) Preparation of Compound 195-1
[0166] Compound 195-2 (19.7 g, 74 mmol) was introduced to THF (200 mL), and the temperature was maintained at -78.degree. C. At -78.degree. C., 2.5 M n-BuLi (36 mL, 89 mmol) was slowly added dropwise thereto. After 30 minutes, n-Bu.sub.3SnCl (22 mL, 81.4 mmol) was added thereto, and the temperature was maintained for 2 hours. After that, the temperature was slowly raised to room temperature, and the result was quenched with an aqueous NH.sub.4Cl solution. The result was extracted with distilled water and Mc, and after the organic layer was dried with anhydrous Na.sub.2O.sub.4, the solvent was removed using a rotary evaporator to obtain target Compound 195-1 (33 g, 95%).
[0167] 3) Preparation of Compound 195
[0168] Compound 195-1 (10 g, 20.5 mmol) and 6-bromo-1,9-dihydropyrene (5.8 g, 20.5 mmol) were dissolved in toluene (80 mL), then Pd(PPh.sub.3).sub.4 (1.2 g, 1 mmol) was were added thereto, and the result was refluxed for 24 hours. After the reaction was completed, the result was cooled to room temperature, and extracted with distilled water and Mc. After the organic layer was dried with anhydrous Na.sub.2O.sub.4, the solvent was removed using a rotary evaporator, and the result was purified using column chromatography with ethyl acetate and hexane as a developing solvent to obtain target Compound 195 (5.5 g, 70%).
[0169] Target Compound F was synthesized in the same manner as the preparation of Compound 195 except that, in Preparation Example 6, Intermediate F of the following Table 6 was used instead of 6-bromo-1,9-dihydropyrene.
TABLE-US-00006 TABLE 6 Compound Number Intermediate F Target Compound F Yield 207 ##STR00112## ##STR00113## 66% 213 ##STR00114## ##STR00115## 72% 217 ##STR00116## ##STR00117## 59%
[0170] Compounds were prepared in the same manner as in the preparation examples, and the synthesis identification results are shown in Table 7 and Table 8. Table 7 shows measurement values of 1H NMR (CDCl.sub.3, 200 MHz), and Table 8 shows measurement values of field desorption mass spectrometry (FD-MS).
TABLE-US-00007 TABLE 7 Example .sup.1H NMR (CDCl.sub.3, 400 MHz) 3 .delta. = 8.68 (s, 1H), 8.23 (s, 2H), 8.16-8.10 (m, 6H), 8.05 (d, 2H), 7.68-7.67 (m, 6H), 7.52-7.41 (m, 5H), 7.35 (d, 2H) 41 .delta. = 8.72 (s, 1H), 8.54 (d, 2H), 8.32 (d, 2H), 8.16 (td, 2H), 8.10 (dd, 2H), 7.67-7.63 (m, 7H), 7.52-7.51 (m, 8H), 7.41-7.35 (m, 4H) 42 .delta. = 8.54 (d, 2H), 8.26-8.21 (m, 4H), 8.16-8.10 (m, 4H), 7.67-7.52 (m, 16H), 7.51-7.41 (m, 6H) 43 .delta. = 8.55-8.54 (m, 6H), 8.16 (td, 2H), 8.10 (dd, 2H), 7.67-7.64 (m, 6H), 7.55-7.41 (m, 14H) 44 .delta. = 8.64 (t, 2H), 8.54 (d, 2H), 8.31 (s, 2H), 8.16-8.10 (m, 6H), 8.00 (d, 2H), 7.67-7.64 (m, 6H), 7.52-7.35 (m, 12H) 45 .delta. = 8.54 (t, 2H), 8.16-8.10 (m, 4H), 7.91 (q, 4H), 7.67-7.64 (m, 6H), 7.52-7.39 (m, 16H) 46 .delta. = 9.04 (d, 2H), 8.54 (t, 2H), 8.30 (d, 4H), 8.16 (dd, 2H), 7.82 (t, 1H), 7.67-7.64 (m, 6H), 7.52-7.41 (m, 10H) 47 .delta. = 9.04 (d, 4H), 8.54 (d, 2H), 8.30 (d, 4H), 8.16 (dd, 2H), 7.82 (t, 2H), 7.67-7.64 (m, 6H), 7.52-7.41 (m, 10H) 57 .delta. = 8.86 (d, 2H), 8.68 (s, 1H), 8.23 (s, 2H), 8.16-8.10 (m, 6H), 8.05 (d, 2H), 7.68-7.67 (m, 6H), 7.35-7.31 (m, 3H) 62 .delta. = 8.83 (d, 1H), 8.74 (s, 3H), 8.38 (d, 1H), 8.16-8.05 (m, 8H), 7.81 (d, 1H), 7.68-7.67 (m, 6H), 7.58 (t, 1H), 7.35 (d, 3H) 63 .delta. = 8.74 (s, 3H), 8.30 (d, 2H), 8.16-8.05 (m, 11H), 7.81 (d, 1H), 7.68-7.67 (m, 6H), 7.35 (d, 4H) 88 .delta. = 9.66 (s, 2H), 8.59 (d, 1H), 8.30 (d, 4H), 8.16 (td, 4H), 8.05 (s, 2H), 7.85 (t, 1H), 7.68-7.67 (m, 6H), 7.36-7.32 (dd, 2H) 89 .delta. = 9.24 (s, 1H), 9.15 (s, 2H), 8.70 (d, 1H), 8.30 (d, 4H), 8.16 (td, 4H), 8.05 (s, 2H), 7.85 (t, 1H), 7.68-7.67 (m, 6H) 90 .delta. = 9.15 (s, 2H), 8.75 (d, 2H), 8.30 (d, 4H), 8.16 (td, 4H), 8.05-7.99 (m, 4H), 7.68-7.67 (m, 6H) 91 .delta. = 9.15 (s, 2H), 9.08 (d, 2H), 8.30 (d, 4H), 8.16 (td, 4H), 8.05 (s, 2H), 7.68-7.67 (m, 7H) 92 .delta. = 9.15 (s, 2H), 8.83 (d, 1H), 8.38-8.29 (m, 5H), 8.21-8.16 (m, 5H), 8.05-8.02 (m, 3H), 7.68-7.58 (m, 8H) 93 .delta. = 9.15 (s, 2H), 8.57 (s, 1H), 8.31-8.16 (m, 9H), 8.06-7.98 (m, 4H), 7.68-7.60 (m, 7H) 94 .delta. = 9.15 (s, 2H), 8.83 (d, 1H), 8.38-8.27 (m, 6H), 8.16-8.03 (m, 8H), 7.68-7.58 (m, 7H) 95 .delta. = 9.66 (s, 2H), 8.83 (d, 1H), 8.44 (d, 1H), 8.38 (d, 1H), 8.30 (m, 4H), 8.16 (q, 4H), 8.06-8.05 (m, 3H), 7.81 (d, 1H), 7.68-7.67 (m, 6H), 7.58 (t, 1H), 7.41 (d, 1H) 138 .delta. = 8.68 (s, 1H), 8.23 (s, 3H), 8.16-8.05 (m, 8H), 7.79 (d, 4H), 7.68-7.67 (m, 6H), 7.51 (t, 4H), 7.41-7.35 (m, 4H) 142 .delta. = 8.68 (s, 1H), 8.28 (d, 2H), 8.23 (s, 3H), 8.16-8.05 (m, 8H), 7.79 (d, 2H), 7.68-7.67 (m, 6H), 7.51 (t, 4H), 7.41-7.35 (m, 4H) 147 .delta. = 8.68 (s, 1H), 8.28 (d, 4H), 8.23 (s, 2H), 8.16-8.05 (m, 8H), 7.68-7.67 (m, 6H), 7.51 (t, 4H), 7.41-7.35 (m, 4H) 148 .delta. = 8.68 (s, 1H), 8.28 (d, 2H), 8.23 (s, 2H), 8.16-8.05 (m, 8H), 7.85 (d, 2H), 7.68-7.67 (m, 6H), 7.51 (t, 4H), 7.52-7.35 (m, 10H), 7.25 (d, 2H) 164 .delta. = 8.30-8.28 (m, 5H), 8.26-8.10 (m, 5H), 8.05 (s, 1H), 7.68-7.67 (m, 3H), 7.60 (t, 1H), 7.51-7.41 (m, 7H) 169 .delta. = 8.30-8.28 (m, 5H), 8.26-8.10 (m, 5H), 8.05 (s, 1H), 7.85 (d, 2H), 7.68-7.67 (m, 3H), 7.60 (t, 1H), 7.51-7.41 (m, 6H), 7.41-7.35 (m, 3H), 7.25 (d, 2H) 192 .delta. = 8.26 (td, 1H), 8.21-8.05 (m, 7H), 7.80 (q, 2H), 7.71-7.54 (m, 9H), 7.35 (d, 1H) 193 .delta. = 9.15 (s, 1H), 8.93 (d, 2H), 8.26-8.04 (m, 10H), 7.68-7.54 (m, 5H), 7.35 (d, 1H) 195 .delta. = 8.55 (d, 1H), 8.21-8.05 (m, 7H), 7.88-7.82 (m, 2H), 7.71-7.67 (m, 7H), 7.35 (d, 1H) 203 .delta. = 9.15 (s, 2H), 8.52 (d, 8H), 8.17-8.04 (m, 10H), 7.85 (t, 8H), 7.72-7.67 (m, 4H), 7.35 (d, 1H) 207 .delta. = 8.83 (d, 2H), 8.38 (d, 2H), 8.16-8.05 (m, 5H), 7.68-7.67 (m, 3H), 7.58 (t, 2H), 7.35 (d, 1H) 210 .delta. = 8.30-8.05 (m, 12H), 7.68-7.47 (m, 12H), 7.35 (d, 3H) 213 .delta. = 8.26-8.05 (m, 11H), 7.81 (d, 1H), 8.38 (d, 2H), 7.68-7.51 (m, 9H), 7.41-7.35 (m, 2H) 217 .delta. = 8.85 (s, 1H), 8.38-8.29 (m, 5H), 8.16-8.00 (m, 8H), 7.81 (d, 1H), 7.68-7.67 (m, 3H), 7.59 (dd, 2H), 7.35 (d, 1H) 226 .delta. = 8.83 (d, 4H), 8.38 (d, 4H), 8.17-8.05 (m, 6H), 7.72-7.58 (m, 10H), 7.35 (d, 1H) 228 .delta. = 8.83 (d, 1H), 8.72 (s, 1H), 8.83 (d, 1H), 8.32 (d, 2H), 8.16-8.05 (m, 6H), 7.61 (d, 1H), 7.68-7.58 (m, 5H), 7.35 (d, 2H) 230 .delta. = 8.72 (s, 1H), 8.32 (d, 2H), 8.30 (d, 2H), 8.16-8.05 (m, 7H), 7.81 (d, 1H), 7.68-7.63 (m, 4H), 7.54-7.47 (m, 3H), 7.35 (d, 3H) 247 .delta. = 9.30 (d, 1H), 8.53 (d, 1H), 8.31-8.05 (m, 10H), 7.90 (s, 1H), 7.70-7.54 (m, 6H), 7.35d (, 1H), 7.14 (t, 1H) 260 .delta. = 9.30 (d, 2H), 8.53 (d, 2H), 8.31 (d, 2H), 8.29-8.05 (m, 10H), 8.04 (d, 2H), 7.90 (s, 1H), 7.70-7.67 (m, 6H), 7.35 (d, 1H), 7.14 (t, 2H) 261 .delta. = 8.83 (d, 2H), 8.74 (s, 3H), 8.38 (d, 2H), 8.16-8.05 (m, 8H), 7.81 (d, 2H), 7.68-7.67 (m, 3H), 7.58 (t, 2H), 7.35 (d, 3H)
TABLE-US-00008 TABLE 8 Compound FD-Mass Compound FD-Mass 1 m/z = 432.51 2 m/z = 508.61 (C32H20N2 = 432.16) (C38H24N2 = 508.19) 3 m/z = 508.61 4 m/z = 482.57 (C38H24N2 = 508.19) (C36H22N2 = 482.18) 5 m/z = 482.57 6 m/z = 482.57 (C36H22N2 = 482.18) (C36H22N2 = 482.18) 7 m/z = 520.62 8 m/z = 532.63 (C39H24N2 = 520.19) (C40H24N2 = 532.19) 9 m/z = 532.63 10 m/z = 584.71 (C40H24N2 = 532.19) (C44H28N2 = 584.23) 11 m/z = 585.69 12 m/z = 556.65 (C43H27N3 = 585.22) (C42H24N2 = 556.19) 13 m/z = 556.65 14 m/z = 556.65 (C42H24N2 = 556.19) (C42H24N2 = 556.19) 15 m/z = 508.61 16 m/z = 532.63 (C38H24N2 = 508.19) (C40H24N2 = 532.19) 17 m/z = 533.62 18 m/z = 433.50 (C39H23N3 = 533.19) (C31H19N3 = 433.16) 19 m/z = 434.49 20 m/z = 483.56 (C30H18N4 = 434.15) (C35H21N3 = 483.17) 21 m/z = 483.56 22 m/z = 483.56 (C35H21N3 = 483.17) (C35H21N3 = 483.17) 23 m/z = 510.59 24 m/z = 558.67 (C36H22N4 = 510.18) (C42H26N2 = 558.21) 25 m/z = 559.66 26 m/z = 560.65 (C41H25N3 = 559.20) (C40H24N4 = 560.20) 27 m/z = 559.66 28 m/z = 608.73 (C41H25N3 = 559.20) (C46H28N2 = 608.23) 29 m/z = 609.72 30 m/z = 610.70 (C45H27N3 = 609.22) (C44H26N4 = 610.22) 31 m/z = 610.70 32 m/z = 608.73 (C44H26N4 = 610.22) (C46H28N2 = 608.23) 33 m/z = 609.72 34 m/z = 658.79 (C45H27N3 = 609.22) (C50H30N2 = 658.24) 35 m/z = 659.77 36 m/z = 534.61 (C49H29N3 = 659.24) (C38H22N4 = 534.18) 37 m/z = 558.67 38 m/z = 559.66 (C42H26N2 = 558.21) (C41H25N3 = 559.20) 39 m/z = 608.73 40 m/z = 708.85 (C46H28N2 = 608.23) (C54H32N2 = 708.26) 41 m/z = 584.71 42 m/z = 660.80 (C44H28N2 = 584.23) (C50H32N2 = 660.26) 43 m/z = 634.77 44 m/z = 684.82 (C48H30N2 = 634.24) (C52H32N2 = 684.26) 45 m/z = 684.82 46 m/z = 585.69 (C52H32N2 = 684.26) (C43H27N3 = 585.22) 47 m/z = 662.78 48 m/z = 711.85 (C48H30N4 = 662.25) (C53H33N3 = 711.27) 49 m/z = 584.71 50 m/z = 660.80 (C44H28N2 = 584.23) (C50H32N2 = 660.26) 51 m/z = 634.77 52 m/z = 684.82 (C48H30N2 = 634.24) (C52H32N2 = 684.26) 53 m/z = 784.94 54 m/z = 509.60 (C60H36N2 = 784.29) (C37H23N3 = 509.19) 55 m/z = 509.60 56 m/z = 509.60 (C37H23N3 = 509.19) (C37H23N3 = 509.19) 57 m/z = 510.59 58 m/z = 509.60 (C36H22N4 = 510.18) (C37H23N3 = 509.19) 59 m/z = 559.66 60 m/z = 559.66 (C41H25N3 = 559.20) (C41H25N3 = 559.20) 61 m/z = 559.66 62 m/z = 610.70 (C41H25N3 = 559.20) (C44H26N4 = 610.22) 63 m/z = 686.80 64 m/z = 687.79 (C50H30N4 = 686.25) (C49H29N5 = 687.24) 65 m/z = 624.73 66 m/z = 700.83 (C45H28N4 = 624.23) (C51H32N4 = 700.26) 67 m/z = 762.90 68 m/z = 533.62 (C56H34N4 = 762.28) (C39H23N3 = 533.19) 69 m/z = 608.73 70 m/z = 684.82 (C46H28N2 = 608.23) (C52H32N2 = 684.26) 71 m/z = 609.72 72 m/z = 735.87 (C45H27N3 = 609.22) (C55H33N3 = 735.27) 73 m/z = 785.93 74 m/z = 685.81 (C59H35N3 = 785.28) (C51H31N3 = 685.25) 75 m/z = 735.87 76 m/z = 685.81 (C55H33N3 = 735.27) (C51H31N3 = 685.25) 77 m/z = 786.92 78 m/z = 863.01 (C58H34N4 = 786.28) (C64H38N4 = 862.31) 79 m/z = 685.81 80 m/z = 735.87 (C51H31N3 = 685.25) (C55H33N3 = 735.27) 81 m/z = 635.75 82 m/z = 685.81 (C47H29N3 = 635.24) (C51H31N3 = 685.25) 83 m/z = 635.75 84 m/z = 686.80 (C47H29N3 = 635.24) (C50H30N4 = 686.25) 85 m/z = 838.99 86 m/z = 762.90 (C62H38N4 = 838.31) (C56H34N4 = 762.28) 87 m/z = 762.90 88 m/z = 510.59 (C56H34N4 = 762.28) (C36H22N4 = 510.18) 89 m/z = 510.59 90 m/z = 510.59 (C36H22N4 = 510.18) (C36H22N4 = 510.18) 91 m/z = 511.57 92 m/z = 560.65 (C35H21N5 = 511.18) (C40H24N4 = 560.20) 93 m/z = 560.65 94 m/z = 560.65 (C40H24N4 = 560.20) (C40H24N4 = 560.20) 95 m/z = 611.69 96 m/z = 687.79 (C43H25N5 = 611.21) (C49H29N5 = 687.24) 97 m/z = 688.78 98 m/z = 625.72 (C48H28N6 = 688.24) (C44H27N5 = 625.23) 99 m/z = 701.81 100 m/z = 763.88 (C50H31N5 = 701.26) (C55H33N5 = 763.27) 101 m/z = 609.72 102 m/z = 686.80 (C45H27N3 = 609.22) (C50H30N4 = 686.25) 103 m/z = 685.81 104 m/z = 610.70 (C51H31N3 = 685.25) (C44H26N4 = 610.22) 105 m/z = 736.86 106 m/z = 786.92 (C54H32N4 = 736.26) (C58H34N4 = 786.28) 107 m/z = 686.80 108 m/z = 736.86 (C50H30N4 = 686.25) (C54H32N4 = 736.26) 109 m/z = 737.85 110 m/z = 686.80 (C53H31N5 = 737.26) (C50H30N4 = 686.25) 111 m/z = 687.79 112 m/z = 787.91 (C49H29N5 = 687.24) (C57H33N5 = 787.27) 113 m/z = 864.00 114 m/z = 864.99 (C63H37N5 = 863.30) (C62H36N6 = 864.30) 115 m/z = 686.80 116 m/z = 736.86 (C50H30N4 = 686.25) (C54H32N4 = 736.26) 117 m/z = 636.74 118 m/z = 686.80 (C46H28N4 = 636.23) (C50H30N4 = 686.25) 119 m/z = 636.74 120 m/z = 637.73 (C46H28N4 = 636.23) (C45H27N5 = 637.23) 121 m/z = 687.79 122 m/z = 839.98 (C49H29N5 = 687.24) (C61H37N5 = 839.30) 123 m/z = 839.98 124 m/z = 763.88 (C61H37N5 = 839.30) (C55H33N5 = 763.27) 125 m/z = 764.87 126 m/z = 763.88 (C54H32N6 = 764.27) (C55H33N5 = 763.27) 127 m/z = 764.87 128 m/z = 635.75 (C54H32N6 = 764.27) (C47H29N3 = 635.24) 129 m/z = 685.81 130 m/z = 712.84 (C51H31N3 = 685.25) (C52H32N4 = 712.26) 131 m/z = 585.69 132 m/z = 662.78 (C43H27N3 = 585.22) (C48H30N4 = 662.25) 133 m/z = 635.75 134 m/z = 658.79 (C47H29N3 = 635.24) (C50H30N4 = 686.25) 135 m/z = 686.80 136 m/z = 686.80 (C50H30N4 = 686.25) (C50H30N4 = 686.25) 137 m/z = 786.92 138 m/z = 662.78 (C58H34N4 = 786.28) (C48H30N4 = 662.25) 139 m/z = 738.87 140 m/z = 814.97 (C54H34N4 = 738.28) (C60H38N4 = 814.31) 141 m/z = 762.90 142 m/z = 662.78 (C56H34N4 = 762.28) (C48H30N4 = 662.25) 143 m/z = 738.87 144 m/z = 738.87 (C54H34N4 = 738.28) (C54H34N4 = 738.28) 145 m/z = 814.97 146 m/z = 762.90 (C60H38N4 = 814.31) (C56H34N4 = 762.28) 147 m/z = 663.77 148 m/z = 739.86 (C47H29N5 = 663.24) (C53H33N5 = 739.27) 149 m/z = 815.96 150 m/z = 763.88 (C59H37N5 = 815.30) (C55H33N5 = 763.27) 151 m/z = 663.77 152 m/z = 739.86 (C47H29N5 = 663.24) (C53H33N5 = 739.27) 153 m/z = 763.88 154 m/z = 663.77 (C55H33N5 = 763.27) (C47H29N5 = 663.24) 155 m/z = 739.86 156 m/z = 739.86 (C53H33N5 = 739.27) (C53H33N5 = 739.27) 157 m/z = 815.96 158 m/z = 763.88 (C59H37N5 = 815.30) (C55H33N5 = 763.27) 159 m/z = 664.75 160 m/z = 740.85 (C46H28N6 = 664.24) (C52H32N6 = 740.27) 161 m/z = 816.95 162 m/z = 764.87 (C58H36N6 = 816.30) (C54H32N6 = 764.27) 163 m/z = 486.57 164 m/z = 486.57 (C34H22N4 = 486.18) (C34H22N4 = 486.18) 165 m/z = 485.58 166 m/z = 485.58 (C41H25N3 = 485.19) (C35H23N3 = 485.19) 167 m/z = 485.58 168 m/z = 485.58 (C35H23N3 = 485.19) (C35H23N3 = 485.19) 169 m/z = 562.66 170 m/z = 638.76 (C40H26N4 = 562.22) (C46H30N4 = 638.25) 171 m/z = 561.67 172 m/z = 561.67 (C41H27N3 = 561.22) (C41H27N3 = 561.22) 173 m/z = 816.95 174 m/z = 764.87 (C58H36N6 = 816.30) (C54H32N6 = 764.27) 175 m/z = 486.57 176 m/z = 486.57 (C34H22N4 = 486.18) (C34H22N4 = 486.18) 177 m/z = 485.58 178 m/z = 485.58 (C35H23N3 = 485.19) (C35H23N3 = 485.19) 179 m/z = 485.58 180 m/z = 485.58 (C35H23N3 = 485.19) (C35H23N3 = 485.19) 181 m/z = 562.66 182 m/z = 562.66 (C40H26N4 = 562.22) (C40H26N4 = 562.22) 183 m/z = 638.76 184 m/z = 638.76 (C46H30N4 = 638.25) (C46H30N4 = 638.25) 185 m/z = 561.67 186 m/z = 561.67 (C41H27N3 = 561.22) (C41H27N3 = 561.22) 187 m/z = 561.67 188 m/z = 561.67 (C41H27N3 = 561.22) (C41H27N3 = 561.22) 189 m/z = 561.67 190 m/z = 561.67 (C41H27N3 = 561.22) (C41H27N3 = 561.22) 191 m/z = 431.53 192 m/z = 455.55 (C33H21N = 431.17) (C35H21N = 455.17) 193 m/z = 481.59 194 m/z = 505.61 (C37H23N = 481.18) (C39H23N = 505.18) 195 m/z = 379.45 196 m/z = 457.56 (C29H17N = 379.14) (C35H23N = 457.18) 197 m/z = 507.62 198 m/z = 557.68 (C39H25N = 507.20) (C43H27N = 557.21) 199 m/z = 557.68 200 m/z = 557.68 (C43H27N = 557.21) (C43H27N = 557.21) 201 m/z = 607.74 202 m/z = 655.78 (C47H29N = 607.23) (C51H29N = 655.23) 203 m/z = 665.22 204 m/z = 659.81 (C49H35N3 = 665.28) (C51H33N = 659.26) 205 m/z = 659.81 206 m/z = 589.20 (C51H33N = 659.26) (C43H31N3 = 589.25) 207 m/z = 357.41 208 m/z = 433.50 (C25H15N3 = 357.13) (C31H19N3 = 433.16) 209 m/z = 509.60 210 m/z = 585.69 (C37H23N3 = 509.19) (C43H27N3 = 585.22) 211 m/z = 511.57 212 m/z = 407.47 (C35H21N5 = 511.18) (C29H17N3 = 407.14) 213 m/z = 509.60 214 m/z = 610.70 (C37H23N3 = 509.19) (C44H26N4 = 610.22) 215 m/z = 559.66 216 m/z = 701.81 (C41H25N3 = 559.20) (C50H31N5 = 701.26) 217 m/z = 483.56 218 m/z = 510.59 (C35H21N3 = 483.17) (C36H22N4 = 510.18) 219 m/z = 482.57 220 m/z = 482.57 (C36H22N2 = 482.18) (C36H22N2 = 482.18) 221 m/z = 609.72 222 m/z = 511.57 (C45H27N3 = 609.22) (C35H21N5 = 511.18) 223 m/z = 587.67 224 m/z = 461.56 (C41H25N5 = 587.21) (C33H23N3 = 461.19) 225 m/z = 685.81 226 m/z = 611.69 (C51H31N3 = 685.25) (C43H25N5 = 611.21) 227 m/z = 667.80 228 m/z = 433.50 (C47H33N5 = 667.27) (C31H19N3 = 433.16) 229 m/z = 447.53 230 m/z = 509.60 (C32H21N3 = 447.17) (C37H23N3 = 509.19) 231 m/z = 434.49 232 m/z = 448.52 (C30H18N4 = 434.15) (C31H20N4 = 448.17) 233 m/z = 510.59 234 m/z = 511.57 (C36H22N4 = 510.18) (C35H21N5 = 511.18) 235 m/z = 637.73 236 m/z = 687.79 (C45H27N5 = 637.23) (C49H29N5 = 687.24) 237 m/z = 587.67 238 m/z = 483.56 (C41H25N5 = 587.21) (C35H21N3 = 483.17) 239 m/z = 559.66 240 m/z = 560.65 (C41H25N3 = 559.20) (C40H24N4 = 560.20) 241 m/z = 484.55 242 m/z = 560.65 (C34H20N4 = 484.17) (C40H24N4 = 560.20) 243 m/z = 561.63 244 m/z = 560.65 (C39H23N5 = 561.20) (C40H24N4 = 558.21) 245 m/z = 636.74 246 m/z = 637.73 (C46H28N4 = 636.23) (C45H27N5 = 637.23)
247 m/z = 459.54 248 m/z = 612.72 (C33H21N3 = 459.17) (C44H28N4 = 612.23) 249 m/z = 612.72 250 m/z = 712.84 (C44H28N4 = 612.23) (C52H32N4 = 712.26) 251 m/z = 582.69 252 m/z = 585.69 (C44H26N2 = 582.21) (C43H27N3 = 585.22) 253 m/z = 585.69 254 m/z = 460.53 (C43H27N3 = 585.22) (C32H20N4 = 460.17) 255 m/z = 613.71 256 m/z = 613.71 (C43H27N5 = 613.23) (C43H27N5 = 613.23) 257 m/z = 583.68 258 m/z = 586.68 (C43H25N3 = 583.20) (C42H26N4 = 586.22) 259 m/z = 509.60 260 m/z = 663.77 (C37H23N3 = 509.19) (C47H29N5 = 663.24) 261 m/z = 612.68 262 m/z = 656.77 (C42H24N6 = 612.21) (C50H28N2 = 656.23) 263 m/z = 708.85 264 m/z = 662.78 (C54H32N2 = 708.26) (C48H30N4 = 662.25) 265 m/z = 664.75 266 m/z = 510.59 (C46H28N6 = 664.24) (C36H22N4 = 510.18) 267 m/z = 455.55 268 m/z = 431.53 (C35H21N = 455.17) (C33H21N = 431.17) 269 m/z = 431.53 270 m/z = 431.53 (C33H21N = 431.17) (C33H21N = 431.17) 271 m/z = 507.62 272 m/z = 509.60 (C39H25N = 507.20) (C37H23N3 = 509.19) 273 m/z = 661.79 274 m/z = 587.67 (C49H31N3 = 661.25) (C41H25N5 = 587.21) 275 m/z = 559.66 276 m/z = 558.67 (C41H25N3 = 559.20) (C42H26N2 = 558.21) 277 m/z = 685.81 278 m/z = 537.65 (C51H31N3 = 685.25) (C39H27N3 = 537.22) 279 m/z = 687.79 280 m/z = 509.60 (C49H29N5 = 687.24) (C37H23N3 = 509.19) 281 m/z = 510.59 282 m/z = 559.66 (C36H22N4 = 510.18) (C41H25N3 = 559.20) 283 m/z = 599.72 284 m/z = 561.67 (C44H29N3 = 599.24) (C41H27N3 = 561.22) 285 m/z = 561.67 286 m/z = 562.66 (C41H27N3 = 561.22) (C40H26N4 = 562.22) 287 m/z = 561.67 288 m/z = 561.67 (C41H27N3 = 561.22) (C41H27N3 = 561.22) 289 m/z = 562.66 290 m/z = 523.63 (C40H26N4 = 562.22) (C38H25N3 = 523.20) 291 m/z = 675.82 292 m/z = 583.18 (C50H33N3 = 675.27) (C45H29N = 583.23) 293 m/z = 583.18 294 m/z = 587.13 (C45H29N = 583.23) (C43H25NO2 = 587.19) 295 m/z = 619.08 296 m/z = 652.18 (C43H25NS2 = 619.14) (C46H28N4O = 652.23) 297 m/z = 668.15 298 m/z = 651.17 (C46H28N4S = 668.20) (C46H29N5 = 651.24) 299 m/z = 652.18 300 m/z = 728.20 (C46H28N4O = 652.23) (C52H32N4O = 728.26) 301 m/z = 744.17 302 m/z = 727.21 (C52H32N4S = 744.23) (C52H33N5 = 727.27) 303 m/z = 728.21 304 m/z = 727.21 (C52H32N4O = 728.26) (C53H33N3O = 727.26) 305 m/z = 743.19 306 m/z = 726.23 (C53H33N3S = 743.24) (C53H34N4 = 726.28) 307 m/z = 727.21 308 m/z = 651.18 (C53H33N3O = 727.26) (C47H29N3O = 651.23) 309 m/z = 667.15 310 m/z = 650.20 (C47H29N3S = 667.21) (C47H30N4 = 650.25) 311 m/z = 651.17 (C47H29N3O = 651.23)
EXPERIMENTAL EXAMPLE
Experimental Example 1
[0171] 1) Manufacture of Organic Light Emitting Device
[0172] A glass substrate on which ITO was coated as a thin film to a thickness of 1500 .ANG. was cleaned with distilled water and ultrasonic waves. After the cleaning with distilled water was finished, the substrate was ultrasonic cleaned with solvents such as acetone, methanol and isopropyl alcohol, then dried, and UVO treatment was carried out for 5 minutes in a UV cleaner using UV. After that, the substrate was transferred to a plasma cleaner (PT), and plasma treatment was carried out under vacuum for removing ITO work function and remaining film, and the substrate was transferred to a thermal deposition apparatus for organic deposition.
[0173] On the ITO transparent electrode (anode), organic materials were formed in a two-stack white organic light emitting diode (WOLED) structure. As for the first stack, a hole transfer layer was formed first by thermal vacuum depositing TAPC to a thickness of 300 .ANG.. After forming the hole transfer layer, a light emitting layer was thermal vacuum deposited thereon as follows. The light emitting layer was deposited to 300 .ANG. by doping FIrpic in 8% as a blue phosphorescent dopant to TCzl, a host. An electron transfer layer was formed to 400 .ANG. using TmPyPB, and then a charge generation layer was formed to 100 .ANG. by doping Cs.sub.2CO.sub.3 in 20% to a compound described in the following Table 9.
[0174] As for the second stack, a hole injection layer was formed first by thermal vacuum depositing MoO.sub.3 to a thickness of 50 .ANG.. A hole transfer layer, a common layer, was formed by doping MoO.sub.3 to TAPC in 20% and forming to 100 .ANG., and then depositing TAPC to 300 .ANG.. After depositing a light emitting layer to 300 .ANG. thereon by doping Ir(ppy).sub.3, a green phosphorescent dopant, in 8% to TCzl, a host, an electron transfer layer was formed to 600 .ANG. using TmPyPB. Lastly, an electron injection layer was formed on the electron transfer layer by depositing lithium fluoride (LiF) to a thickness of 10 .ANG., and then a cathode was formed on the electron injection layer by depositing an aluminum (Al) cathode to a thickness of 1,200 .ANG. to manufacture an organic electroluminescent device.
[0175] Meanwhile, all the organic compounds required to manufacture the OLED device were vacuum sublimation purified under 10.sup.-6 torr to 10.sup.-8 torr by each material to be used in the OLED manufacture.
##STR00118## ##STR00119## ##STR00120##
[0176] 2) Driving Voltage and Light Emission Efficiency of Organic Electroluminescent Device
[0177] For the organic electroluminescent devices manufactured as above, electroluminescent light emission (EL) characteristics were measured using M7000 manufactured by McScience Inc., and with the measurement results, T95 when standard luminance was 3,500 cd/m.sup.2 was measured using a lifetime test system (M6000) manufactured by McScience Inc. Results of measuring a driving voltage, light emission efficiency, external quantum efficiency and a color coordinate (CIE) of the white organic electroluminescent devices manufactured according to the present disclosure are as shown in Table 9.
TABLE-US-00009 TABLE 9 Light Driving Emission Voltage Efficiency CIE Lifespan Compound (V) (cd/A) (x, y) (T95) Example 1 3 7.89 59.32 (0.221, 28 0.434) Example 2 5 7.96 59.45 (0.220, 27 0.430) Example 3 7 7.82 58.98 (0.219, 28 0.422) Example 4 41 7.88 61.78 (0.221, 29 0.427) Example 5 42 7.93 58.66 (0.225, 25 0.424) Example 6 43 7.88 59.74 (0.219, 24 0.425) Example 7 44 7.79 59.42 (0.222, 27 0.430) Example 8 45 7.90 60.17 (0.221, 26 0.415) Example 9 46 7.91 59.87 (0.228, 28 0.430) Example 10 47 7.95 58.26 (0.218, 28 0.411) Example 11 57 7.99 60.11 (0.223, 28 0.422) Example 12 62 7.06 69.82 (0.221, 42 0.440) Example 13 63 7.08 69.45 (0.220, 40 0.430) Example 14 64 7.12 68.55 (0.216, 35 0.426) Example 15 65 7.06 66.75 (0.225, 36 0.430) Example 16 66 7.01 68.21 (0.220, 34 0.415) Example 17 67 7.11 67.99 (0.217, 35 0.413) Example 18 89 7.99 58.73 (0.211, 26 0.421) Example 19 90 7.97 59.02 (0.218, 27 0.425) Example 20 91 7.99 59.64 (0.211, 31 0.423) Example 21 92 7.88 58.98 (0.227, 23 0.422) Example 22 93 7.89 58.77 (0.230, 29 0.431) Example 23 94 7.96 59.25 (0.230, 22 0.424) Example 24 95 7.12 68.55 (0.215, 44 0.422) Example 25 96 7.23 67.58 (0.221, 39 0.422) Example 26 97 7.20 69.45 (0.223, 37 0.426) Example 27 100 7.11 67.44 (0.215, 38 0.422) Example 28 138 7.90 60.91 (0.220, 29 0.428) Example 29 142 7.77 64.77 (0.216, 30 0.430) Example 30 147 7.82 59.99 (0.216, 31 0.422) Example 31 148 7.80 61.74 (0.220, 31 0.432) Example 32 164 7.90 63.52 (0.210, 32 0.430) Example 33 169 7.96 58.77 (0.221, 26 0.422) Example 34 192 7.98 63.35 (0.223, 29 0.424) Example 35 193 7.74 64.20 (0.224, 29 0.424) Example 36 195 7.85 60.57 (0.226, 27 0.433) Example 37 203 7.82 60.65 (0.210, 31 0.423) Example 38 207 7.08 68.21 (0.214, 45 0.422) Example 39 208 7.02 67.44 (0.212, 40 0.417) Example 40 209 7.09 68.01 (0.211, 42 0.422) Example 41 210 7.01 69.67 (0.223, 40 0.429) Example 42 213 7.21 68.96 (0.215, 39 0.426) Example 43 217 7.05 67.93 (0.221, 50 0.428) Example 44 226 7.11 69.44 (0.223, 41 0.428) Example 45 228 7.02 68.08 (0.222, 42 0.430) Example 46 230 7.04 69.11 (0.231, 40 0.434) Example 47 233 7.25 68.11 (0.221, 38 0.430) Example 48 234 7.13 69.24 (0.214, 39 0.421) Example 49 238 7.22 67.88 (0.222, 42 0.413 Example 50 242 7.01 68.43 (0.212, 41 0.431) Example 51 247 7.29 65.55 (0.220, 53 0.432) Example 52 260 7.31 66.32 (0.221, 51 0.433) Example 53 261 7.10 69.22 (0.229, 40 0.432) Example 54 272 7.22 68.46 (0.228, 41 0.421) Example 55 279 7.14 69.21 (0.219, 39 0.430) Example 56 280 7.20 67.88 (0.226, 44 0.416) Example 57 296 7.87 57.56 (0.220, 26 0.420) Example 58 302 7.93 59.67 (0.226, 28 0.423) Example 59 305 7.85 58.69 (0.217, 26 0.419) Example 60 310 7.79 59.11 (0.230, 27 0.425) Comparative TmPyPB 8.50 57.63 (0.213, 25 Example 0.431) 1-1 Comparative N-ADN2 8.05 58.45 (0.211, 26 Example 0.427) 1-2 Comparative N-ADN5 8.11 57.12 (0.220, 25 Example 0.421) 1-3 Comparative N-AN9 8.38 58.11 (0.218, 26 Example 0.439) 1-4 Comparative N-AN11 8.34 57.55 (0.234, 24 Example 0.423) 1-5
[0178] As shown from the results of Table 9, the organic electroluminescent devices using the charge generation layer material of the 2-stack white organic electroluminescent device of the present disclosure had a low driving voltage and improved light emission efficiency compared to Comparative Examples 1-1, 1-2, 1-3, 1-4 and 1-5. Particularly, it was identified that Compounds 62, 63, 95, 207, 210, 217, 226, 228, 230, 247, 260 and 261 were significantly excellent in all of driving, efficiency and lifespan.
[0179] The presumed reason for such results is that the compound of the present disclosure used as an N-type charge generation layer formed with an invented skeleton having proper length, strength and flat property and a proper hetero-compound capable of binding with metals is doped with an alkali metal or an alkali-earth metal to form a gap state within the N-type charge generation layer, and electrons produced from a P-type charge generation layer are readily injected to the electron transfer layer through the gap state produced within the N-type charge generation layer. Accordingly, the P-type charge generation layer favorably carried out electron injection and electron transfer to the N-type charge generation layer, and as a result, it is considered that a driving voltage of the organic light emitting device decreased, and efficiency and lifespan were improved.
Experimental Example 2
[0180] 1) Manufacture of Organic Light Emitting Device
[0181] A glass substrate on which ITO was coated as a thin film to a thickness of 1500 .ANG. was cleaned with distilled water and ultrasonic waves. After the cleaning with distilled water was finished, the substrate was ultrasonic cleaned with solvents such as acetone, methanol and isopropyl alcohol, then dried, and UVO treatment was carried out for 5 minutes in a UV cleaner using UV. After that, the substrate was transferred to a plasma cleaner (PT), and plasma treatment was carried out under vacuum for removing ITO work function and remaining film, and the substrate was transferred to a thermal deposition apparatus for organic deposition. On the ITO transparent electrode (anode), organic materials were formed in a single-stack structure. As a hole injection layer, HAT-CN was deposited to a thickness of 50 .ANG., and subsequently, a hole transfer layer was formed by doping DNTPD within 10% to NPD, depositing the result to a thickness of 1500 .ANG., and continuously depositing TCTA to a thickness of 200 .ANG.. Subsequently, a light emitting layer comprising a t-Bu-perylene dopant in an ADN host was formed to a thickness of 250 .ANG.. Next, Alq.sub.3, an electron transfer layer, was formed to a thickness of 250 .ANG., and an N-type charge transfer layer was formed to a thickness of 100 .ANG. by doping Li, an alkali metal, to a compound described in the following Table 10, and Al, a cathode, was formed to a thickness of approximately 1,000 .ANG. to manufacture an organic electroluminescent device.
##STR00121## ##STR00122## ##STR00123## ##STR00124##
[0182] 2) Driving Voltage and Light Emission Efficiency of Organic Electroluminescent Device
[0183] For the organic electroluminescent devices manufactured as above, electroluminescent light emission (EL) characteristics were measured using M7000 manufactured by McScience Inc., and with the measurement results, T.sub.95 when standard luminance was 750 cd/m.sup.2 was measured using a lifetime test system (M6000) manufactured by McScience Inc. Results of measuring a driving voltage, light emission efficiency, external quantum efficiency and a color coordinate (CIE) of the white organic electroluminescent devices manufactured according to the present disclosure are as shown in Table 10.
TABLE-US-00010 TABLE 10 Light Driving Emission Voltage Efficiency CIE Lifespan Compound (V) (cd/A) (x, y) (T95) Example 3 5.68 6.42 (0.134 30 61 0.102) Example 5 5.75 6.38 (0.134 29 62 0.102) Example 7 5.62 6.32 (0.134 32 63 0.101) Example 41 5.58 6.33 (0.134, 29 64 0.104) Example 42 5.79 6.21 (0.134, 28 65 0.099) Example 43 5.83 6.15 (0.132, 29 66 0.100) Example 44 5.65 6.07 (0.128, 25 67 0.095) Example 45 5.77 6.21 (0.134, 30 68 0.099) Example 46 5.85 6.43 (0.134, 28 69 0.100 Example 47 5.86 6.22 (0.131, 30 70 0.098) Example 57 5.87 6.31 (0.130, 29 71 0.0910 Example 62 4.48 6.89 (0.134 41 72 0.100) Example 63 4.51 6.90 (0.134 40 73 0.100) Example 64 4.61 6.75 (0.132, 42 74 0.095) Example 65 4.72 6.71 (0.131, 39 75 0.100) Example 66 4.55 6.66 (0.129, 40 76 0.098) Example 67 4.60 6.70 (0.130, 40 77 0.100) Example 89 5.79 6.11 (0.133, 29 78 0.102 Example 90 5.91 6.03 (0.130, 27 79 0.099) Example 91 5.91 6.17 (0.134, 31 80 0.101) Example 92 5.87 6.21 (0.128, 30 81 0.103) Example 93 5.67 6.35 (0.134, 29 82 0.103) Example 94 5.78 6.22 (0.130, 31 83 0.099) Example 95 4.51 6.89 (0.134, 49 84 0.100) Example 96 4.62 6.91 (0.132, 42 85 0.102) Example 97 4.68 6.75 (0.129, 40 86 0.098) Example 100 4.82 6.64 (0.128, 43 87 0.102) Example 138 5.65 6.46 (0.134 31 88 0.101) Example 142 5.70 6.57 (0.134 29 89 0.100) Example 147 5.50 6.39 (0.134 30 90 0.100) Example 148 5.56 6.47 (0.134 30 91 0.100) Example 164 5.62 6.43 (0.134 32 92 0.101) Example 169 5.78 6.27 (0.130, 28 93 0.102) Example 192 5.46 6.34 (0.134 32 94 0.102) Example 193 5.76 6.51 (0.134 34 95 0.101) Example 195 5.76 6.44 (0.134, 28 96 0.102) Example 203 5.62 6.58 (0.134 35 97 0.102) Example 207 4.53 6.79 (0.134, 48 98 0.098) Example 208 4.73 6.88 (0.131, 42 99 0.100) Example 209 4.69 6.76 (0.130, 40 100 0.102) Example 210 4.60 6.90 (0.134 40 101 0.099) Example 213 4.77 6.80 (0.128, 39 102 0.097) Example 217 4.65 6.93 (0.134, 49 103 0.100) Example 226 4.44 6.89 (0.134 40 104 0.099) Example 228 4.57 6.95 (0.134 41 105 0.100) Example 230 4.50 6.98 (0.134 41 106 0.101) Example 233 4.69 6.78 (0.130, 39 107 0.099) Example 234 4.70 6.80 (0.129, 38 108 0.100) Example 238 4.65 6.62 (0.128, 40 109 0.101) Example 242 4.89 6.79 (0.130, 40 110 0.100) Example 247 4.83 6.67 (0.134 48 111 0.103) Example 260 4.90 6.58 (0.134 47 112 0.103) Example 261 4.51 6.99 (0.134 42 113 0.100) Example 272 4.91 6.78 (0.131, 38 114 0.102) Example 279 4.78 6.80 (0.128, 42 115 0.099) Example 280 4.69 6.82 (0.130, 40 116 0.098) Example 296 5.88 6.21 (0.133, 25 117 0.103) Example 302 5.97 6.01 (0.127, 29 118 0.101) Example 305 5.79 6.15 (0.129, 27 119 0.100) Example 310 5.80 6.23 (0.130, 30 120 0.098) Comparative Bphen 5.82 6.23 (0.134 27 Example 0.110) 2-1 Comparative N-ADN2 5.79 6.31 (0.134, 31 Example 0.100) 2-2 Comparative N-ADN5 5.68 6.24 (0.134, 29 Example 0.100) 2-3 Comparative N-AN9 5.83 6.15 (0.134, 27 Example 0.104) 2-4 Comparative N-AN11 5.86 6.26 (0.134, 26 Example 0.103) 2-5
[0184] As shown from the results of Table 10, the organic electroluminescent devices using the charge generation layer material of the blue organic electroluminescent device of the present disclosure had a low driving voltage and improved light emission efficiency compared to Comparative Examples 2-1, 2-2, 2-3, 2-4 and 2-5. Particularly, it was identified that Compounds 62, 63, 95, 207, 210, 217, 226, 228, 230, 247, 260 and 261 were significantly excellent in all of driving, efficiency and lifespan.
[0185] The presumed reason for such results is that the compound of the present disclosure used as an N-type charge generation layer formed with an invented skeleton having proper length, strength and flat property and a proper hetero-compound capable of binding with metals is doped with an alkali metal or an alkali-earth metal to form a gap state within the N-type charge generation layer, and electrons produced from a P-type charge generation layer are readily injected to the electron transfer layer through the gap state produced within the N-type charge generation layer. Accordingly, the P-type charge generation layer favorably carried out electron injection and electron transfer to the N-type charge generation layer, and as a result, it is considered that a driving voltage of the organic light emitting device decreased, and efficiency and lifespan were improved.
Experimental Example 3
[0186] 1) Manufacture of Organic Light Emitting Device
[0187] A transparent electrode ITO thin film obtained from glass for an OLED (manufactured by Samsung Corning Advanced Glass) was ultrasonic cleaned consecutively using trichloroethylene, acetone, ethanol and distilled water for 5 minutes each, placed in isopropanol and stored, and then used.
[0188] Next, the ITO substrate was installed in a substrate folder of vacuum deposition equipment, and the following 4,4',4''-tris(N,N-(2-naphthyl)-phenylamino)triphenyl amine (2-TNATA) was introduced to a cell in the vacuum deposition equipment.
##STR00125##
[0189] Subsequently, the chamber was exhausted until the degree of vacuum inside the chamber reached 10.sup.-6 torr, and then a current was applied to the cell to evaporate the 2-TNATA to deposit a hole injection layer having a thickness of 600 .ANG. on the ITO substrate.
[0190] The following N,N'-bis(.alpha.-naphthyl)-N,N'-diphenyl-4,4'-diamine (NPB) was introduced to a different cell in the vacuum deposition equipment, a current was applied to the cell to evaporate to deposit a hole transfer layer having a thickness of 300 .ANG. on the hole injection layer.
##STR00126##
[0191] After forming the hole injection layer and the hole transfer layer as above, a blue light emitting material having a structure as follows was deposited thereon as a light emitting layer. Specifically, H1, a blue light emitting host material, was vacuum deposited to a thickness of 200 .ANG. on one cell in the vacuum deposition equipment, and D1, a blue light emitting dopant material, was vacuum deposited thereon in 5% with respect to the host material.
##STR00127##
[0192] Subsequently, compounds of the following structural formulae E1, N-ADN2, N-ADN5, N-AN9 and N-AN11 were deposited to a thickness of 300 .ANG. as an electron transfer layer.
##STR00128## ##STR00129##
[0193] As an electron injection layer, lithium fluoride (LiF) was deposited to a thickness of 10 .ANG., and an Al cathode was formed to a thickness of 1000 .ANG. to manufacture an OLED device.
[0194] Meanwhile, all the organic compounds required to manufacture the OLED device were vacuum sublimation purified under 10.sup.-6 torr to 10.sup.-8 torr by each material to be used in the OLED manufacture.
[0195] 2) Driving Voltage and Light Emission Efficiency of Organic Electroluminescent Device
[0196] For the organic electroluminescent devices manufactured as above, electroluminescent light emission (EL) characteristics were measured using M7000 manufactured by McScience Inc., and with the measurement results, Tss when standard luminance was 700 cd/m.sup.2 was measured using a lifetime test system (M6000) manufactured by McScience Inc. Results of measuring a driving voltage, light emission efficiency, external quantum efficiency and a color coordinate (CIE) of the white organic electroluminescent devices manufactured according to the present disclosure are as shown in Table 11.
TABLE-US-00011 TABLE 11 Light Driving Emission Voltage Efficiency CIE Lifespan Compound (V) (cd/A) (x, y) (T95) Example 3 4.42 6.93 (0.134 40 121 0.099) Example 5 4.48 6.86 (0.134 41 122 0.099) Example 7 4.47 6.87 (0.134 40 123 0.100) Example 41 4.51 7.05 (0.134, 42 124 0.099) Example 42 4.67 6.64 (0.129, 38 125 0.100) Example 43 4.82 6.55 (0.130, 36 126 0.099) Example 44 4.77 6.68 (0.132, 36 127 0.098) Example 45 4.44 6.97 (0.134, 40 128 0.101) Example 46 4.61 6.89 (0.134, 40 129 0.103) Example 47 4.88 6.72 (0.128, 36 130 0.099) Example 57 4.70 6.75 (0.127, 37 131 0.100) Example 62 5.35 6.30 (0.134 33 132 0.102) Example 63 5.28 6.28 (0.134 32 133 0.102) Example 64 5.61 6.19 (0.130, 28 134 0.099) Example 65 5.60 6.07 (0.129, 29 135 0.100) Example 66 5.55 6.10 (0.130, 30 136 0.101) Example 67 5.71 6.15 (0.129, 28 137 0.098) Example 89 4.57 6.74 (0.128, 36 138 0.099) Example 90 4.60 6.69 (0.129, 36 139 0.102) Example 91 4.49 6.96 (0.134, 40 140 0.100) Example 92 4.62 6.72 (0.133, 37 141 0.100) Example 93 4.55 6.85 (0.134, 39 142 0.101) Example 94 4.80 6.69 (0.130, 38 143 0.100) Example 95 5.42 6.13 (0.134, 29 144 0.101) Example 96 5.67 6.02 (0.132, 25 145 0.103) Example 97 5.58 6.15 (0.130, 25 146 0.099) Example 100 5.70 6.07 (0.129, 28 147 0.100) Example 138 4.54 6.92 (0.134 41 148 0.101) Example 142 4.30 6.81 (0.134 41 149 0.101) Example 147 4.50 6.98 (0.134 40 150 0.100) Example 148 4.44 7.09 (0.134 39 151 0.100) Example 164 4.61 7.01 (0.134 38 152 0.099) Example 169 4.60 6.87 (0.131, 37 153 0.100) Example 192 4.64 7.08 (0.134 40 154 0.099) Example 193 4.53 6.91 (0.134 41 155 0.101) Example 195 4.97 6.59 (0.134, 34 156 0.100) Example 203 4.54 6.95 (0.134 42 157 0.103) Example 207 5.33 6.17 (0.134, 29 158 0.102) Example 208 5.63 6.11 (0.131, 28 159 0.098) Example 209 5.58 6.23 (0.129, 30 160 0.101) Example 210 5.61 6.20 (0.134 31 161 0.102) Example 213 5.66 6.04 (0.131, 30 162 0.102) Example 217 5.65 6.08 (0.134, 28 163 0.101) Example 226 5.35 6.32 (0.134 30 164 0.102) Example 228 5.43 6.25 (0.134 29 165 0.101) Example 230 5.40 6.49 (0.134 31 166 0.101) Example 233 5.59 6.14 (0.131, 28 167 0.101) Example 234 5.60 6.22 (0.129, 29 168 0.100) Example 238 5.48 6.18 (0.129, 28 169 0.099) Example 242 5.59 6.07 (0.128, 30 170 0.102 Example 247 4.74 5.89 (0.134 37 171 0.100) Example 260 4.70 6.01 (0.134 36 172 0.101) Example 261 5.40 6.31 (0.134 31 173 0.102) Example 272 5.71 6.19 (0.131, 27 174 0.100) Example 279 5.63 6.06 (0.128, 27 175 0.102) Example 280 5.49 6.14 (0.130, 29 176 0.101) Example 296 4.73 6.78 (0.130, 35 177 0.100) Example 302 4.67 6.65 (0.129, 36 178 0.098) Example 305 4.69 6.92 (0.130, 35 179 0.101) Example 310 4.52 6.75 (0.129, 37 180 0.097) Comparative E1 5.79 6.10 (0.134 27 Example 0.100) 3-1 Comparative N-ADN2 5.54 6.24 (0.134, 33 Example 0.100) 3-2 Comparative N-ADN5 5.68 6.21 (0.134, 31 Example 0.104) 3-3 Comparative N-AN9 5.77 6.18 (0.134, 33 Example 0.103) 3-4 Comparative N-AN11 5.72 6.14 (0.134, 35 Example 0.100) 3-5
[0197] As shown from the results of Table 11, the organic electroluminescent devices using the electron transfer layer material of the blue organic electroluminescent device of the present disclosure had a low driving voltage and significantly improved light emission efficiency and lifespan compared to Comparative Examples 3-1, 3-2, 3-3, 3-4 and 3-5. Particularly, it was identified that Compounds 3, 5, 7, 41, 45, 46, 91, 93, 138, 142, 147, 148, 164, 192, 193 and 203 were significantly excellent in all of driving, efficiency and lifespan.
[0198] The presumed reason for such results is that, when the invented compound having proper length, strength and flat property is used as an electron transfer layer, a compound in an excited state is produced by receiving electrons under a specific condition, and particularly, when the excited state is formed in the heteroskeleton site of the compound, excited energy moves to a stable state before the excited heteroskeleton site goes through a different reaction, and the relatively stabilized compound is capable of efficiently transferring electrons without compound decomposition or destruction. As a reference, it is considered that those having a stable state when excited are aryl or acene series compounds or multicyclic hetero-compounds. Accordingly, it is considered that the compound of the present disclosure enhances electron-transport properties or improved stability resulting in excellency in all of driving, efficiency and lifespan.
* * * * *






































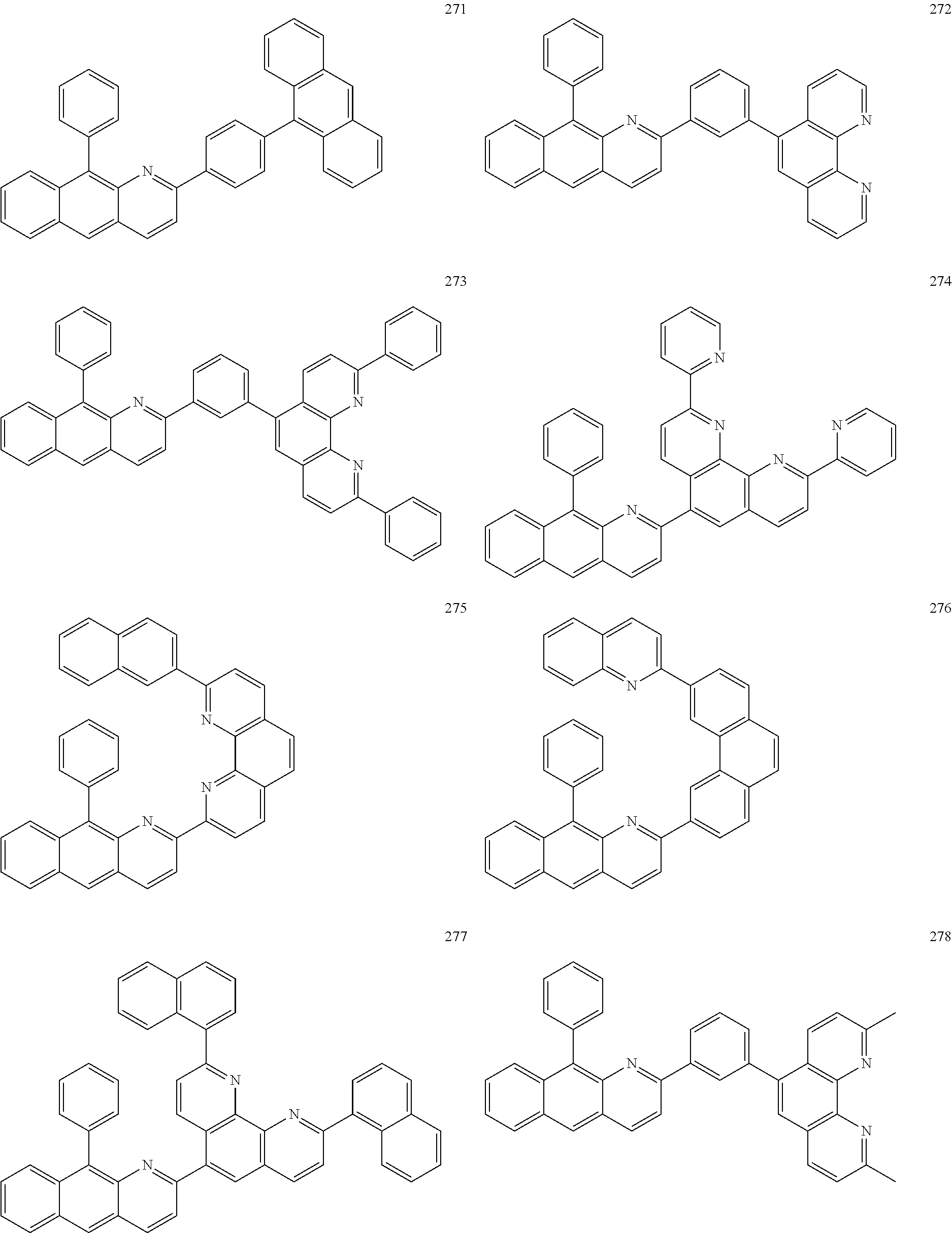











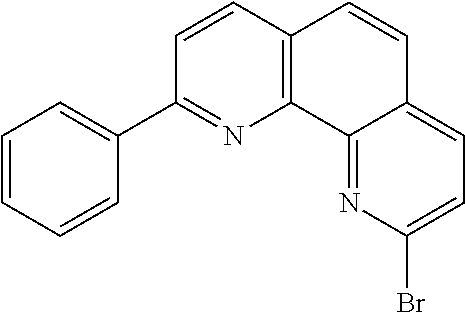


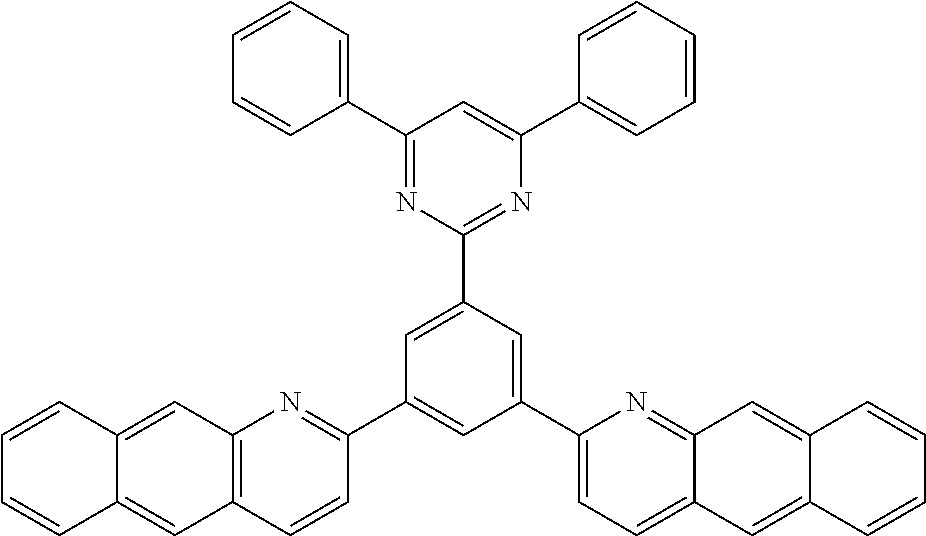





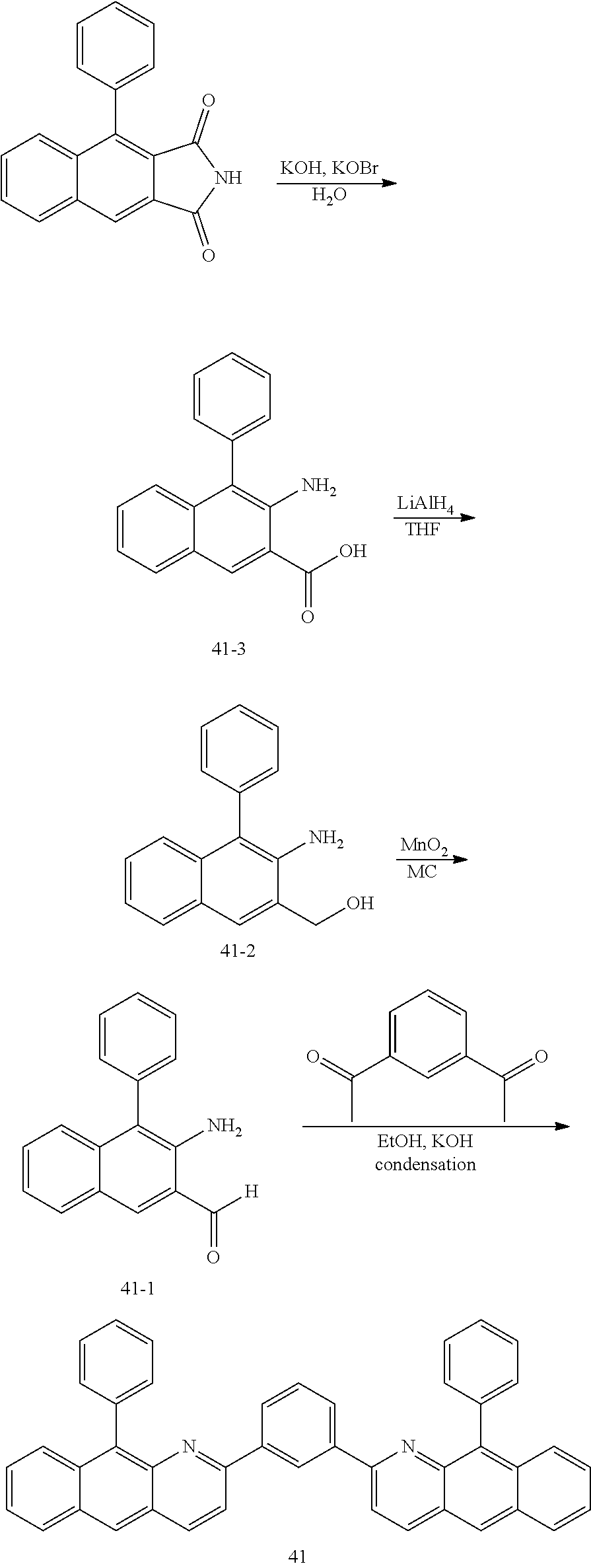








































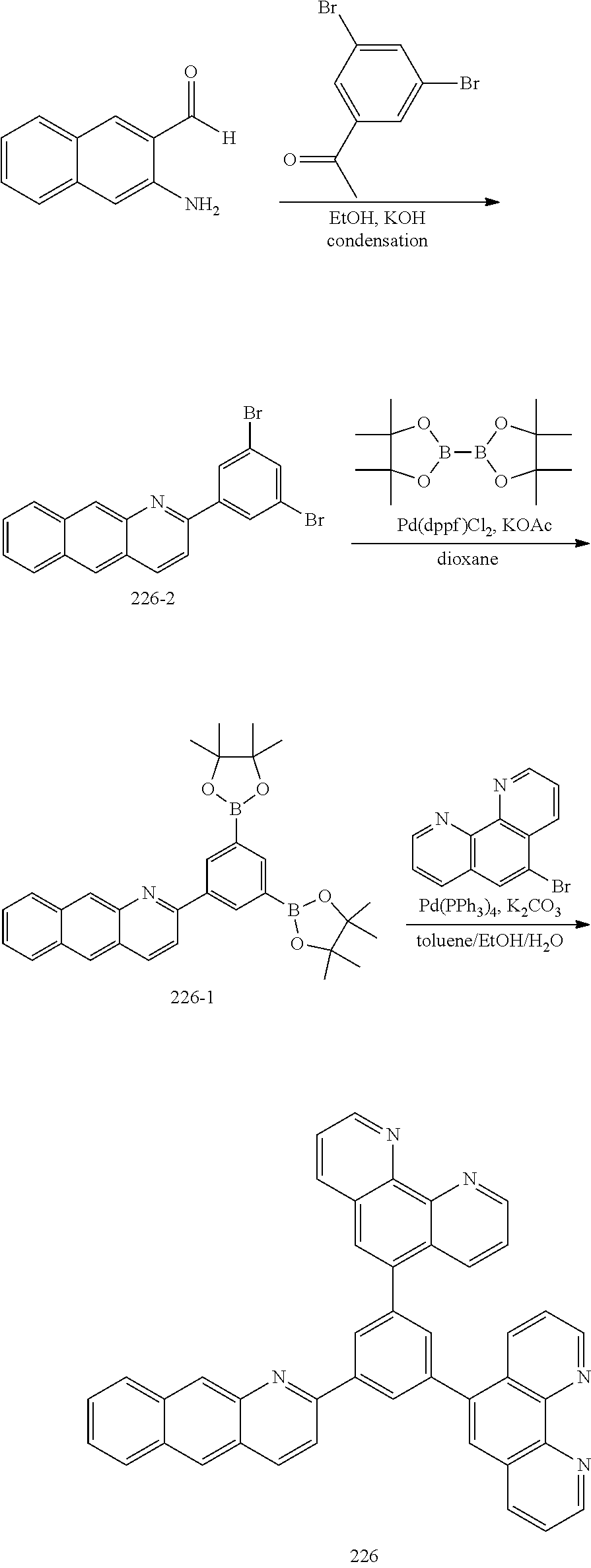









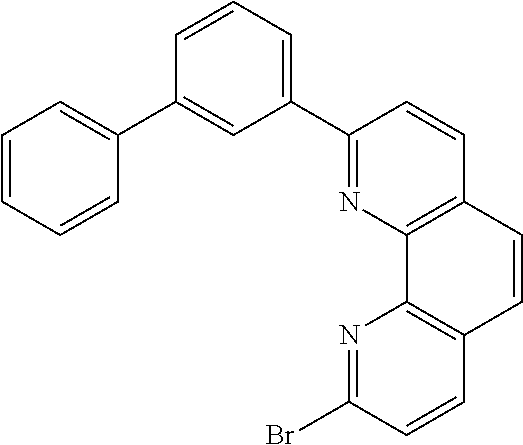









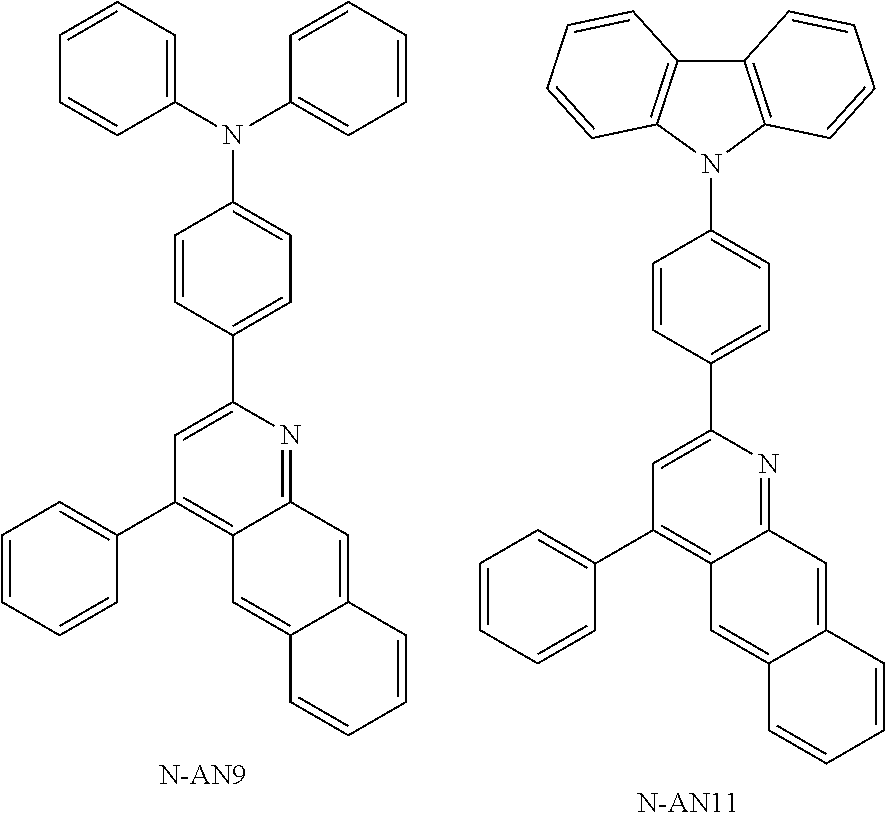
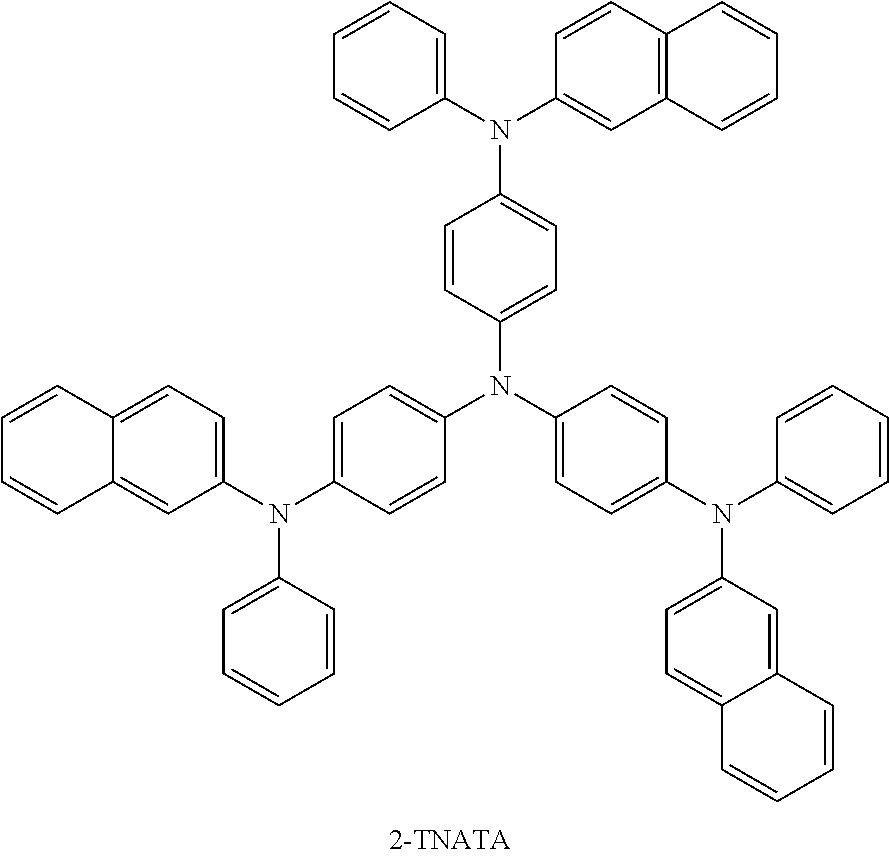




































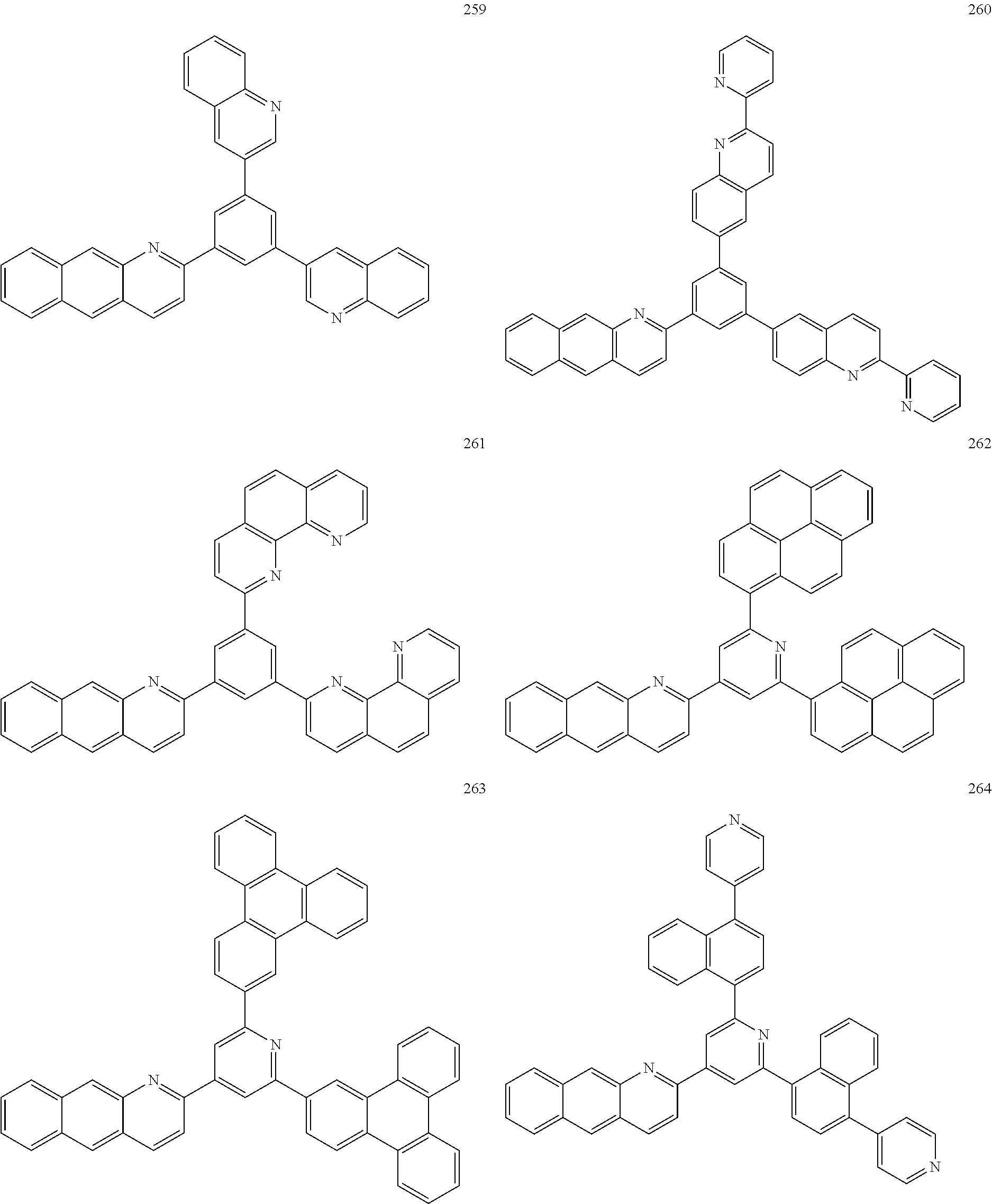






D00000

D00001

D00002

D00003

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.