Upconversion Of Light For Use In Optogenetic Methods
Deisseroth; Karl ; et al.
U.S. patent application number 16/267144 was filed with the patent office on 2019-07-18 for upconversion of light for use in optogenetic methods. The applicant listed for this patent is The Board of Trustees of the Leland Stanford Junior University. Invention is credited to Polina Anikeeva, Karl Deisseroth.
| Application Number | 20190217118 16/267144 |
| Document ID | / |
| Family ID | 46024837 |
| Filed Date | 2019-07-18 |

| United States Patent Application | 20190217118 |
| Kind Code | A1 |
| Deisseroth; Karl ; et al. | July 18, 2019 |
UPCONVERSION OF LIGHT FOR USE IN OPTOGENETIC METHODS
Abstract
Provided herein are compositions comprising lanthanide-doped nanoparticles which upconvert electromagnetic radiation from infrared or near infrared wavelengths into the visible light spectrum. Also provided herein are methods activating light-responsive opsin proteins expressed on plasma membranes of neurons and selectively altering the membrane polarization state of the neurons using the light delivered by the lanthanide-doped nanoparticles.
| Inventors: | Deisseroth; Karl; (Stanford, CA) ; Anikeeva; Polina; (Somerville, MA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 46024837 | ||||||||||
| Appl. No.: | 16/267144 | ||||||||||
| Filed: | February 4, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15214403 | Jul 19, 2016 | 10252076 | ||
| 16267144 | ||||
| 13882703 | Jul 16, 2013 | 9522288 | ||
| PCT/US11/59287 | Nov 4, 2011 | |||
| 15214403 | ||||
| 61410729 | Nov 5, 2010 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61N 2005/0663 20130101; C12N 2750/14143 20130101; A61K 9/1611 20130101; A61K 41/00 20130101; A61N 5/062 20130101; A61N 5/0613 20130101; A61K 48/0083 20130101; C12N 7/00 20130101; A61N 2005/0659 20130101; A61K 41/0038 20130101; B82Y 5/00 20130101; A61K 41/008 20130101; A61K 38/177 20130101; A61N 2005/0662 20130101; A61N 5/0622 20130101; A61D 7/00 20130101; A61K 9/0019 20130101 |
| International Class: | A61N 5/06 20060101 A61N005/06; A61K 48/00 20060101 A61K048/00; A61K 41/00 20060101 A61K041/00; B82Y 5/00 20060101 B82Y005/00; A61K 9/16 20060101 A61K009/16; A61K 38/17 20060101 A61K038/17; C12N 7/00 20060101 C12N007/00; A61D 7/00 20060101 A61D007/00; A61K 9/00 20060101 A61K009/00 |
Claims
1.-13. (canceled)
14. A method to hyperpolarize the plasma membrane of a neural cell in an individual comprising: (a) placing a plurality of lanthanide-doped nanoparticles in proximity to the neural cell, wherein the nanoparticles comprise NaYF.sub.4:Yb/X/Gd, wherein X is erbium (Er), thulium (Tm), or Er/Tm; and (b) exposing the plurality of nanoparticles to electromagnetic radiation in the infrared (IR) or near infrared (NIR) spectrum, wherein the electromagnetic radiation in the IR or NIR spectrum is upconverted into yellow, amber or red light in the visible spectrum by the nanoparticles, and wherein a light-responsive opsin comprising a light-responsive chloride pump is expressed on the plasma membrane and activation of the opsin by the light in the visible spectrum induces the hyperpolarization of the plasma membrane.
15. A method to hyperpolarize the plasma membrane of a neural cell in an individual comprising: (a) administering a polynucleotide encoding a light-responsive opsin to an individual, wherein the light-responsive opsin comprises a light-responsive chloride pump and is expressed on the plasma membrane of a neural cell in the individual and the opsin is capable of inducing membrane hyperpolarization of the neural cell when illuminated with light; (b) administering a plurality of lanthanide-doped nanoparticles in proximity to the neural cell, wherein the nanoparticles comprise NaYF.sub.4:Yb/X/Gd, wherein X is erbium (Er), thulium (Tm), or Er/Tm; and (c) exposing the plurality of nanoparticles to electromagnetic radiation in the infrared (IR) or near infrared (NIR) spectrum, wherein the electromagnetic radiation in the IR or NIR spectrum is upconverted into yellow, amber or red light in the visible spectrum and the activation of the opsin by the light in the visible spectrum induces the hyperpolarization of the plasma membrane.
16. The method of claim 14, wherein the light-responsive opsin comprises an amino acid sequence having at least 85% amino acid sequence identity to SEQ ID NO:1.
17. (canceled)
18. The method of claim 14, wherein the electromagnetic energy in the IR or NIR spectrum is upconverted into light having a wavelength of about 580 nm to about 630 nm.
19. The method of claim 14, wherein the electromagnetic energy in the IR or NIR spectrum is upconverted into light having a wavelength of about 630 nm to about 740 nm.
20. The method of claim 14, wherein the electromagnetic energy in the IR or NIR spectrum is upconverted into light having a wavelength corresponding to yellow or amber light.
21. The method of claim 14, wherein the electromagnetic energy in the IR or NIR spectrum is upconverted into light having a wavelength corresponding to red light.
22. The method of claim 14, wherein the individual is a non-human animal.
23. The method of claim 14, wherein the individual is a human.
24. The method of claim 14, wherein the neural cell is a neural cell in the central nervous system.
25. The method of claim 14, wherein the neural cell is a neural cell in the peripheral nervous system.
26.-32. (canceled)
33. The method of claim 14, wherein X is Er.
34. The method of claim 14, wherein X is Tm.
35. The method of claim 14, wherein X is Er/Tm.
36. The method of claim 14, wherein the neural cell is a neural cell in the nucleus accumbens of the individual.
37. The method of claim 14, wherein the light-responsive opsin comprises an amino acid sequence having at least 95% amino acid sequence identity to SEQ ID NO:2 or SEQ ID NO:3.
38. The method of claim 14, wherein the light-responsive opsin comprises a NpHR protein.
39. A method to hyperpolarize the plasma membrane of a neural cell in an individual comprising: (a) placing a plurality of lanthanide-doped nanoparticles in proximity to the neural cell, wherein the nanoparticles comprise NaYF.sub.4:Yb/X/Gd, wherein X is erbium (Er), thulium (Tm), or Er/Tm; and (b) exposing the plurality of nanoparticles to electromagnetic radiation in the infrared (IR) or near infrared (NIR) spectrum, wherein the electromagnetic radiation in the IR or NIR spectrum is upconverted into green or blue light in the visible spectrum by the nanoparticles, and wherein a light-responsive opsin comprising a light-responsive proton pump is expressed on the plasma membrane and activation of the opsin by the light in the visible spectrum induces the hyperpolarization of the plasma membrane.
40. A method to hyperpolarize the plasma membrane of a neural cell in an individual comprising: (a) administering a polynucleotide encoding a light-responsive opsin to an individual, wherein the light-responsive opsin comprises a light-responsive proton pump and is expressed on the plasma membrane of a neural cell in the individual and the opsin is capable of inducing membrane hyperpolarization of the neural cell when illuminated with light; (b) administering a plurality of lanthanide-doped nanoparticles in proximity to the neural cell, wherein the nanoparticles comprise NaYF.sub.4:Yb/X/Gd, wherein X is erbium (Er), thulium (Tm), or Er/Tm; and (c) exposing the plurality of nanoparticles to electromagnetic radiation in the infrared (IR) or near infrared (NIR) spectrum, wherein the electromagnetic radiation in the IR or NIR spectrum is upconverted into green or blue light in the visible spectrum and the activation of the opsin by the light in the visible spectrum induces the hyperpolarization of the plasma membrane.
41. The method of claim 39, wherein the light-responsive opsin comprises an amino acid sequence having at least 85% amino acid sequence identity to SEQ ID NO:4.
42. The method of claim 39, wherein the electromagnetic energy in the IR or NIR spectrum is upconverted into light having a wavelength of about 450 nm to about 495 nm.
43. The method of claim 39, wherein the electromagnetic energy in the IR or NIR spectrum is upconverted into light having a wavelength corresponding to green light.
44. The method of claim 39, wherein the electromagnetic energy in the IR or NIR spectrum is upconverted into light having a wavelength corresponding to blue light.
45. The method of claim 39, wherein the individual is a non-human animal.
46. The method of claim 39, wherein the individual is a human.
47. The method of claim 39, wherein the neural cell is a neural cell in the central nervous system.
48. The method of claim 39, wherein the neural cell is a neural cell in the peripheral nervous system.
49. The method of claim 39, wherein X is Er.
50. The method of claim 39, wherein X is Tm.
51. The method of claim 39, wherein X is Er/Tm.
52. The method of claim 39, wherein the light-responsive opsin comprises an amino acid sequence having at least 95% amino acid sequence identity to SEQ ID NO:4.
53. The method of claim 14, wherein the light-responsive opsin comprises a GtR3 protein.
Description
CROSS REFERENCE TO RELATED APPLICATION
[0001] This application claims priority to U.S. Provisional Application No. 61/410,729 filed Nov. 5, 2010, the disclosure of which is incorporated herein by reference in its entirety.
FIELD OF THE INVENTION
[0002] This application pertains to compositions comprising lanthanide-doped nanoparticles which upconvert electromagnetic radiation from infrared or near infrared wavelengths into the visible light spectrum and methods of using lanthanide-doped nanoparticles to deliver light to activate light-responsive opsin proteins expressed in neurons and selectively alter the membrane polarization state of the neurons.
BACKGROUND
[0003] Optogenetics is the combination of genetic and optical methods used to control specific events in targeted cells of living tissue, even within freely moving mammals and other animals, with the temporal precision (millisecond-timescale) needed to keep pace with functioning intact biological systems. The hallmark of optogenetics is the introduction of fast light-responsive opsin channel or pump proteins to the plasma membranes of target neuronal cells that allow temporally precise manipulation of neuronal membrane potential while maintaining cell-type resolution through the use of specific targeting mechanisms. Among the microbial opsins which can be used to investigate the function of neural systems are the halorhodopsins (NpHRs), used to promote membrane hyperpolarization when illuminated, and the channel rhodopsins, used to depolarize membranes upon exposure to light. In just a few short years, the field of optogenetics has furthered the fundamental scientific understanding of how specific cell types contribute to the function of biological tissues, such as neural circuits, in vivo. Moreover, on the clinical side, optogenetics-driven research has led to insights into the neurological mechanisms underlying complex mammalian behaviors such as anxiety, memory, fear, and addiction.
[0004] In spite of these advances, use of optogenetic methods in animals suffers from the significant drawback of requiring the animal to either be tethered to a light source or to have a light source surgically implanted into the animal. Moreover, when optogenetic methods are used to alter the function of neurons in the brain, a light source must be placed in proximity to those neurons. This requires drilling a hole in the animal's skull and also presents practical difficulties when the brain region of interest is located deep within the brain itself. Since light poorly passes through neural tissue, this necessitates inserting a fiber optic light source into the brain, which can result in unintended damage to surrounding brain tissue.
[0005] What is needed, therefore, is a method to non-invasively deliver light to neurons located within the brain and the peripheral nervous system of animals expressing light-responsive opsin proteins on the plasma membranes of neural cells.
[0006] Throughout this specification, references are made to publications (e.g., scientific articles), patent applications, patents, etc., all of which are herein incorporated by reference in their entirety.
SUMMARY OF THE INVENTION
[0007] Provided herein are compositions and methods for non-invasively delivering light to neurons expressing light-responsive opsin proteins on neural plasma membranes via the use of nanoparticles capable of upshifting electromagnetic radiation from wavelengths associated with the infrared (IR) or near infrared (NIR) spectrum into wavelengths associated with visible light.
[0008] Accordingly, provided herein is a method to depolarize the plasma membrane of a neural cell in an individual comprising: (a) placing a plurality of lanthanide-doped nanoparticles in proximity to the neural cell; and (b) exposing the plurality of nanoparticles to electromagnetic radiation in the infrared (IR) or near infrared (NIR) spectrum, wherein the electromagnetic radiation in the IR or NIR spectrum is upconverted into light in the visible spectrum by the nanoparticles, and wherein a light-responsive opsin is expressed on the plasma membrane of the neural cells and activation of the opsin by the light in the visible spectrum induces the depolarization of the plasma membrane.
[0009] In other aspects, provided herein is a method to depolarize the plasma membrane of a neural cell in an individual comprising: (a) administering a polynucleotide encoding a light-responsive opsin to an individual, wherein the light-responsive protein is expressed on the plasma membrane of a neural cell in the individual, and the opsin is capable of inducing membrane depolarization of the neural cell when illuminated with light; (b) administering a plurality of lanthanide-doped nanoparticles in proximity to the neural cell; and (c) exposing the plurality of nanoparticles to electromagnetic radiation in the infrared (IR) or near infrared (NIR) spectrum, wherein the electromagnetic radiation in the IR or NIR spectrum is upconverted into light in the visible spectrum and the activation of the opsin by the light in the visible spectrum induces the depolarization of the plasma membrane.
[0010] In some aspects, provided herein is a method to hyperpolarize the plasma membrane of a neural cell in an individual comprising: (a) placing a plurality of lanthanide-doped nanoparticles in proximity to the neural cell; and (b) exposing the plurality of nanoparticles to electromagnetic radiation in the infrared (IR) or near infrared (NIR) spectrum, wherein the electromagnetic radiation in the IR or NIR spectrum is upconverted into light in the visible spectrum by the nanoparticles, and wherein a light-responsive opsin is expressed on the plasma membrane and activation of the opsin by the light in the visible spectrum induces the hyperpolarization of the plasma membrane.
[0011] In yet other aspects, provided herein is a method to hyperpolarize the plasma membrane of a neural cell in an individual comprising: (a) administering a polynucleotide encoding a light-responsive opsin to an individual, wherein the light-responsive protein is expressed on the plasma membrane of a neural cell in the individual, and the opsin is capable of inducing membrane hyperpolarization of the neural cell when illuminated with light; (b) administering a plurality of lanthanide-doped nanoparticles in proximity to the neural cell; and (c) exposing the plurality of nanoparticles to electromagnetic radiation in the infrared (IR) or near infrared (NIR) spectrum, wherein the electromagnetic radiation in the IR or NIR spectrum is upconverted into light in the visible spectrum and the activation of the opsin by the light in the visible spectrum induces the hyperpolarization of the plasma membrane.
[0012] The present disclosure is directed to apparatuses and methods involving upconversion for deep delivery of light in vivo. Aspects of the present disclosure relate generally to delivery of light to tissue in vivo using upconversion of near infrared light to the visible light spectrum and methods relating to the applications discussed herein.
[0013] Certain aspects of the present disclosure are directed to a light source that is implanted within living tissue. Nanoparticles from the nanoparticle solution anchor to a target cell population that includes cells expressing light responsive channels/opsins. The nanoparticles are configured to respond to receipt of light of a first wavelength by emitting light of a second, different wavelength. For example, the nanoparticles can upconvert received light and thereby emit light of a higher frequency.
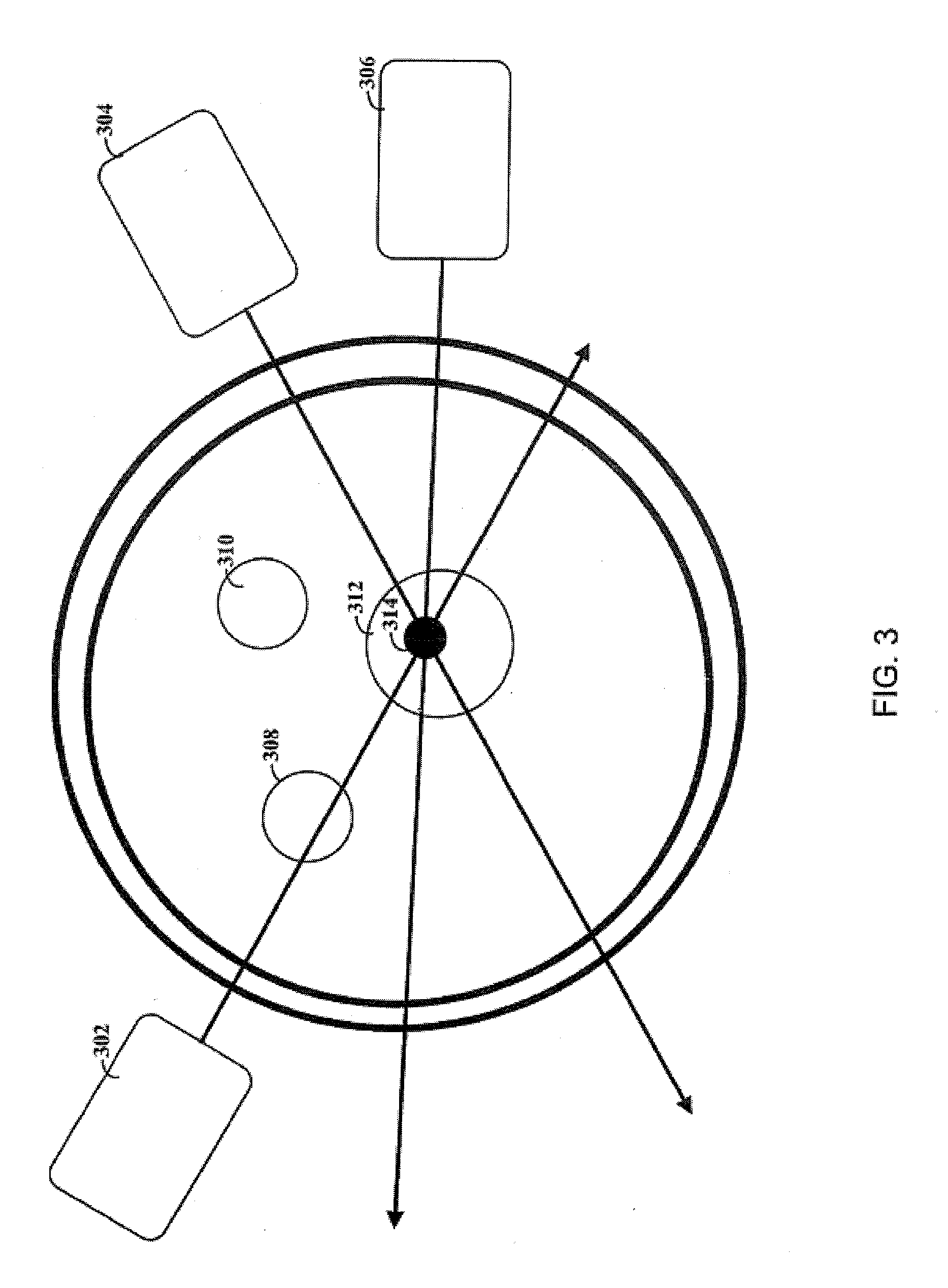
[0014] Embodiments of the present disclosure are directed towards injection of a site of interest with a virus, caring an opsin gene and a nanoparticle solution. The virus causes a target cell population at the site of interest to express the opsin gene. Various different light sources are possible. The use of different wavelengths can be particularly useful for facilitating the use of different (external) light sources, e.g., as certain wavelengths exhibit corresponding decreases in absorption by tissue of the brain or otherwise.
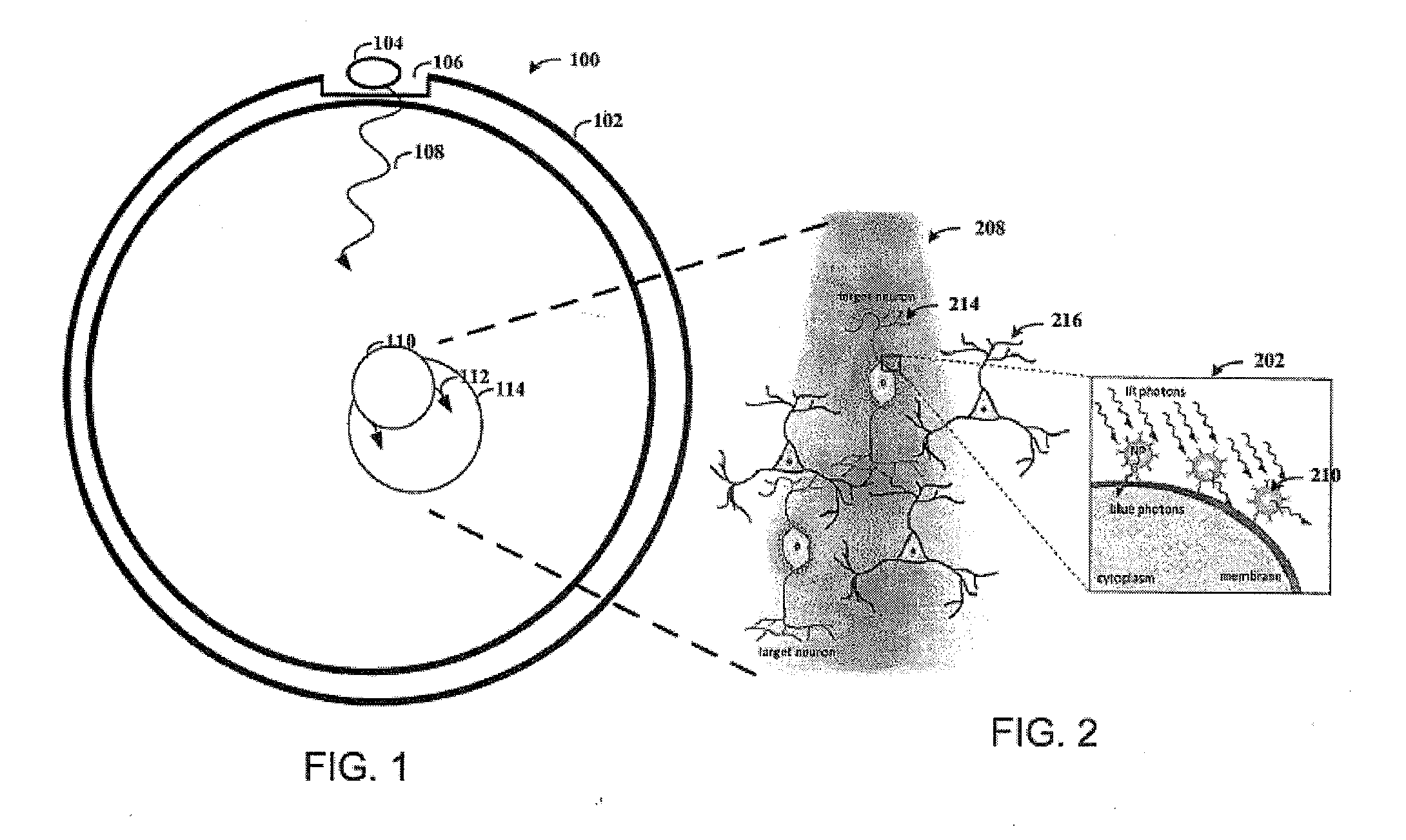
[0015] Consistent with a particular embodiment of the present disclosure, a light-emitting diode ("LED") is placed on a portion of a skull that has been thinned. The LED is placed under the skin near the thinned portion of the skull, and the location and/or orientation of the LED is chosen, at least in part, based on the location of the target cell population. For example, the LED can be placed to reduce the distance between the LED and the target cell population and oriented accordingly.
[0016] In certain more specific aspects of the present disclosure, light from the LED travels through surrounding tissue to the nanoparticles. When (near) infrared light hits the nanoparticles, the nanoparticles absorb the infrared (IR) photons and emit visible photons. The visible photons are then absorbed by the opsins expressed within the target cell population causing a response therein (e.g., triggering neural excitation or inhibition).
[0017] The LED can be powered by a battery similar to those used for pacemakers. The LED can emit light in the infrared spectrum, and particularly between 700 nm-1000 nm, which can travel through the skull and intervening tissue. The light emitted from the nanoparticles has a spectra centered between 450-550 nm. The wavelength of the light emitted is dependent on characteristics of the nanoparticle.
[0018] The above overview is not intended to describe each illustrated embodiment or every implementation of the present disclosure.
BRIEF DESCRIPTION OF THE DRAWINGS
[0019] Various example embodiments may be more completely understood in consideration of the following description and the accompanying drawings, in which:
[0020] FIG. 1 shows a cross section of a skull, consistent with an embodiment of the present disclosure.
[0021] FIG. 2 shows light delivery to target neurons, consistent with an embodiment of the present disclosure.
[0022] FIG. 3 depicts a system that uses multiple light sources, consistent with an embodiment of the present disclosure.
[0023] While the present disclosure is amenable to various modifications and alternative forms, specifics thereof have been shown by way of example in the drawings and will be described in detail. It should be understood, however, that the intention is not to limit the present disclosure to the particular embodiments described. On the contrary, the intention is to cover all modifications, equivalents, and alternatives falling within the scope of the present disclosure including aspects defined in the claims.
DETAILED DESCRIPTION
[0024] This invention provides, inter alia, compositions and methods for delivering light to neural cells expressing one or more light-responsive opsin proteins on the plasma membranes of those neural cells. The inventors have discovered that nanoparticles doped with a lanthanide metal (for example, Gadolinium) that converts infrared (IR) or near infrared (NIR) electromagnetic radiation into wavelengths corresponding to the visible light spectrum can be used to activate light-responsive opsin proteins on the plasma membrane of a neural cell and selectively alter the membrane polarization state of the cell. Unlike visible light, IR or NIR electromagnetic energy readily penetrates biological tissues. For example, NIR can penetrate biological tissues for distances of up to 4 centimeters (Heyward & Dale Wagner, "Applied Body Composition Assessment", 2nd edition (2004), p. 100). Certain equations useful for calculating light penetration in tissue as a function of wavelength are disclosed in U.S. Pat. No. 7,043,287, the contents of which are incorporated herein by reference. Similarly, U.S. Patent Application Publication No. 2007/0027411 discloses that near infrared Low Level Laser Treatment light penetrates the body to a depth of between 3-5 cm. Therefore, use of IR or NIR sources of electromagnetic radiation in optogenetic methods can alleviate the need to place a light source in direct proximity to neural cells. In particular, for optogenetic techniques in the brain, use of lanthanide-doped nanoparticles in combination with IR or NIR electromagnetic energy can permit activation of the opsin protein without the need to puncture the skull or insert a fiber optic light source into the brain. Similarly, in the peripheral nervous system, opsin-expressing nerves can be activated via IR or NM sources placed under the skin or worn against the skin.
[0025] General Techniques
[0026] The practice of the present invention will employ, unless otherwise indicated, conventional techniques of molecular biology, microbiology, cell biology, biochemistry, nucleic acid chemistry, immunology, and physiology, which are well known to those skilled in the art. Such techniques are explained fully in the literature, such as, Molecular Cloning: A Laboratory Manual, second edition (Sambrook et al., 1989) and Molecular Cloning: A Laboratory Manual, third edition (Sambrook and Russel, 2001), (jointly referred to herein as "Sambrook"); Current Protocols in Molecular Biology (F. M. Ausubel et al., eds., 1987, including supplements through 2001); PCR: The Polymerase Chain Reaction, (Mullis et al., eds., 1994); Harlow and Lane (1988), Antibodies, A Laboratory Manual, Cold Spring Harbor Publications, New York; Harlow and Lane (1999), Using Antibodies: A Laboratory Manual, Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y. (jointly referred to herein as "Harlow and Lane"), Beaucage et al. eds., Current Protocols in Nucleic Acid Chemistry, John Wiley & Sons, Inc., New York, 2000), Handbook of Experimental Immunology, 4th edition (D. M. Weir & C. C. Blackwell, eds., Blackwell Science Inc., 1987), and Gene Transfer Vectors for Mammalian Cells (J. M. Miller & M. P. Calos, eds., 1987). Other useful references include Harrison's Principles of Internal Medicine (McGraw Hill; J. Isseleacher et al., eds.) and Lanthanide Luminescence: Photophysical, Analytical and Biological Aspects (Springer-Verlag, Berlin, Heidelberg; Hanninen & Harma, eds., 2011).
Definitions
[0027] As used herein, "infrared" or "near infrared" or "infrared light" or "near infrared light" refers to electromagnetic radiation in the spectrum immediately above that of visible light, measured from the nominal edge of visible red light at 0.74 .mu.m, and extending to 300 .mu.m. These wavelengths correspond to a frequency range of approximately 1 to 400 THz. In particular, "near infrared" or "near infrared light" also refers to electromagnetic radiation measuring 0.75-1.4 .mu.m in wavelength, defined by the water absorption.
[0028] "Visible light" is defined as electromagnetic radiation with wavelengths between 380 nm and 750 nm. In general, "electromagnetic radiation," including light, is generated by the acceleration and deceleration or changes in movement (vibration) of electrically charged particles, such as parts of molecules (or adjacent atoms) with high thermal energy, or electrons in atoms (or molecules).
[0029] The term "nanoparticles" as used herein, can also refer to nanocrystals, nanorods, nanoclusters, clusters, particles, dots, quantum dots, small particles, and nanostructured materials. The term "nanoparticle" encompasses all materials with small size (generally, though not necessarily) less than 100 nm associated with quantum size effects.
[0030] An "individual" is a mammal including a human. Mammals include, but are not limited to, farm animals, sport animals, pets, primates, mice and rats. Individuals also include companion animals including, but not limited to, dogs and cats. In some aspects, an individual is a non-human animal, such as a mammal. In another aspect, an individual is a human.
[0031] As used herein, the singular form "a", "an", and "the" includes plural references unless indicated otherwise.
[0032] It is intended that every maximum numerical limitation given throughout this specification includes every lower numerical limitation, as if such lower numerical limitations were expressly written herein. Every minimum numerical limitation given throughout this specification will include every higher numerical limitation, as if such higher numerical limitations were expressly written herein. Every numerical range given throughout this specification will include every narrower numerical range that falls within such broader numerical range, as if such narrower numerical ranges were all expressly written herein.
[0033] Lanthanide-Doped Nanoparticles
[0034] In materials science, doping is commonly used to incorporate specific species of ions or atoms into a host lattice core structure to produce hybrid materials with new and useful properties. When synthesizing nanoparticles, doping can influence not only the size and shape of the particles, but also other properties, such as the ability to convert near infrared (NIR) excitation into a visible emission of light.
[0035] The lanthanide metals, or lanthanoids (also known as the "Rare Earth" metals), are elements of atomic number 57 (Lanthanum) through 71 (Lutetium), and often include Yttrium (atomic number 39) and Scandium (atomic number 21) because of their chemical similarities. Lanthanide ions exhibit unique luminescent properties, including the ability to convert near infrared long-wavelength excitation radiation into shorter visible wavelengths through a process known as photon upconversion. Lanthanides usually exist as trivalent cations, in which case their electronic configuration is (Xe) 4f, with n varying from 1 (Ce.sup.3+) to 14 (Lu.sup.3+). The transitions within the f-manifold are responsible for many of the photo-physical properties of the lanthanide ions, such as long-lived luminescence and sharp absorption and emission lines. The f-electrons are shielded from external perturbations by filled 5s and 5p orbitals, thus giving rise to line-like spectra. Additionally, the f-f electronic transitions of lanthanides are LaPorte forbidden, leading to long excited state lifetimes, in the micro- to millisecond range.
[0036] In some embodiments, any known method can be used to synthesize lanthanide-doped nanoparticles. Such methods are well known in the art (See, e.g., Xu & Li, 2007, Clin Chem., 53(8):1503-10; Wang et al., 2010, Nature, 463(7284):1061-5; U.S. Patent Application Publication Nos.: 2003/0030067 and 2010/0261263; and U.S. Pat. No. 7,550,201, the disclosures of each of which are incorporated herein by reference in their entireties). For example, in some embodiments, lanthanide-doped nanorods can be synthesized with a NaYF.sub.4 dielectric core, wherein a DI water solution (1.5 ml) of 0.3 g NaOH is mixed with 5 ml of ethanol and 5 ml of oleic acid under stirring. To the resulting mixture is selectively added 2 ml of RECl.sub.3 (0.2 M, RE=Y, Yb, Er, Gd, Sm, Nd or La) and 1 ml of NH.sub.4F (2 M). The solution is then transferred into an autoclave and heated at 200.degree. C. for 2 h. Nanorods are then obtained by centrifugation, washed with water and ethanol several times, and finally re-dispersed in cyclohexane. In another non-limiting example, nanoparticles can be synthesized using 2 ml of RECl.sub.3 (0.2 M, RE=Y, Yb, Er, Gd, or Tm) in methanol added to a flask containing 3 ml oleic acid and 7 ml of 1-octadecene. This solution is then heated to 160.degree. C. for 30 min and cooled down to room temperature. Thereafter, a 5 ml methanol solution of NH.sub.4F (1.6 mmol) and NaOH (1 mmol) is added and the solution is stirred for 30 min. After methanol evaporation, the solution is next heated to 300.degree. C. under argon for 1.5 h and cooled down to room temperature. The resulting nanoparticles are precipitated by the addition of ethanol, collected by centrifugation, washed with methanol and ethanol several times, and finally re-dispersed in cyclohexane.
[0037] In one embodiment, the materials for the lanthanide-doped nanoparticle core can include a wide variety of dielectric materials. In various embodiments, the dielectric core can include lanthanide-doped oxide materials. Lanthanides include lanthanum (La), cerium (Ce), praseodymium (Pr), neodymium (Nd), promethium (Pm), samarium (Sm), europium (Eu), gadolinium (Gd), terbium (Tb), dysprosium (Dy), holmium (Ho), erbium (Er), thulium (Tm), ytterbium (Yb), and lutetium (Lu). Other suitable dielectric core materials include non-lanthanide elements such as yttrium (Y) and scandium (Sc). Hence, suitable dielectric core materials include, but are not limited to, Y.sub.2O.sub.3, Y.sub.2O.sub.2S, NaYF.sub.4, NaYbF4, Na doped YbF.sub.3, YAG, YAP, Nd.sub.2O.sub.3, LaF.sub.3, LaCl.sub.3, La.sub.2O.sub.3, TiO.sub.2, LuPO.sub.4, YVO.sub.4, YbF.sub.3, YF.sub.3, or SiO.sub.2. In one embodiment, the dielectric nanoparticle core is NaYF.sub.4. These dielectric cores can be doped with one or more Er, Eu, Yb, Tm, Nd, Tb, Ce, Y, U, Pr, La, Gd and other rare-earth species or a combination thereof. In one embodiment, the dielectric core material is doped with Gd. In another embodiment, the lanthanide-doped nanoparticle comprises NaYF.sub.4:Yb/X/Gd, wherein X is Er, Tm, or Er/Tm. In some embodiments, the lanthanide-doped nanoparticles comprise a NaYF.sub.4:Yb/Er (18/2 mol %) dielectric core doped with any of about 0 mol %, about 5 mol %, about 10 mol %, about 15 mol %, about 20 mol %, about 25 mol %, about 30 mol %, about 35 mol %, about 40 mol %, about 45 mol %, about 50 mol %, about 55 mol %, about or 60 mol % Gd.sup.3+ ions, inclusive, including any mol % in between these values. In other embodiments, the lanthanide-doped nanoparticles comprise a NaYF.sub.4:Yb/Er (18/2 mol %) dielectric core doped with any of about 0 mol %, about 5 mol %, about 10 mol %, about 15 mol %, about 20 mol %, about 25 mol %, or about 30 mol % Yb.sup.3+ ions, inclusive, including any mol % in between these values. In yet other embodiments, the lanthanide-doped nanoparticles comprise a NaYF.sub.4:Yb/Er (18/2 mol %) dielectric core doped with any of about 0 mol %, about 5 mol %, about 10 mol %, about 15 mol %, about 20 mol %, about 25 mol %, or about 30 mol % Er.sup.3+ ions, inclusive, including any mol % in between these values. In other embodiments, the lanthanide-doped nanoparticles comprise a NaYF.sub.4:Yb/Er (18/2 mol %) dielectric core doped with any of about 0 mol %, about 5 mol %, about 10 mol %, about 15 mol %, about 20 mol %, about 25 mol %, or about 30 mol % Tm.sup.3+ ions, inclusive, including any mol % in between these values. In another embodiment, the lanthanide-doped nanoparticle is selected from the group consisting of NaYF.sub.4:Yb/Er/Gd (18/2/5 mol %), NaYF.sub.4:Yb/Tm/Er/Gd (20/0.2/0.1/5 mol %), NaYF.sub.4:Yb/Tm/Er/Gd (20/0.2/0.05/5 mol %), and NaYF.sub.4:Yb/Tm/Gd (20/0.2/5 mol %).
[0038] In some aspects, the lanthanide-doped nanoparticles disclosed herein are conjugated to one or more delivery molecules to target them to one or more molecules expressed on the surface of a neural cell of interest (such as a neural cell expressing one or more light-responsive opsin proteins on its plasma membrane). These can include, without limitation, antibodies or fragments thereof, small molecules, as well as lectins or any other carbohydrate motif. The delivery molecules ensure that the lanthanide-doped nanoparticles remain in close proximity to the opsin proteins to permit activation upon upconversion of IR or NIR electromagnetic radiation. Antibody conjugation to nanoparticles is well-known in the art (See, e.g., U.S. Patent Application Publication No.: 2010/0209352 and 2008/0267876, the contents of each of which are incorporated by reference herein in their entireties).
[0039] In another aspect, lanthanide-doped nanoparticles can be embedded or trapped within a biocompatible material which is surgically placed proximal to (such as adjacent to or around) the neural cell of interest (such as a neural cell expressing one or more light-responsive opsin proteins on its plasma membrane). In some embodiments, the biocompatible material is transparent, so that visible light produced by the upconversion of IR or NIR electromagnetic radiation by the lanthanide-doped nanoparticles can reach the light-responsive opsin proteins expressed on the surface of the neural cell of interest. The biocompatible materials used to embed or trap the lanthanide-doped nanoparticles can include, but are not limited to, Ioplex materials and other hydrogels such as those based on 2-hydroxyethyl methacrylate or acrylamide, and poly ether polyurethane ureas (PEUU) including Biomer (Ethicon Corp.), Avcothane (Avco-Everrett Laboratories), polyethylene, polypropylene, polytetrafluoroethylene (Gore-Tex.TM.), poly(vinylchloride), polydimethylsiloxane, an ethylene-acrylic acid copolymer, knitted or woven Dacron, polyester-polyurethane, polyurethane, polycarbonatepolyurethane (Corethane.TM.), polyamide (Nylon) and polystyrene. In one embodiment, the biocompatible material can be polydimethylsiloxane (PDMS). Additional compounds that may be used for embedding and/or trapping the lanthanide-doped nanoparticles disclosed herein are described in Kirk-Othmer, Encyclopedia of Chemical Technology, 3rd Edition 1982 (Vol. 19, pp. 275-313, and Vol. 18, pp. 219-2220), van der Giessen et al., 1996, Circulation, 94:1690-1997 (1996), U.S. Patent Application Publication No.: 2011/0054305, and U.S. Pat. No. 6,491,965, the contents of each which are incorporated herein by reference in their entireties.
[0040] Light-Responsive Opsin Proteins
[0041] Provided herein are optogenetic-based compositions for selectively hyperpolarizing or depolarizing neurons of the central or peripheral nervous system. Optogenetics refers to the combination of genetic and optical methods used to control specific events in targeted cells of living tissue, even within freely moving mammals and other animals, with the temporal precision (millisecond-timescale) needed to keep pace with functioning intact biological systems. Optogenetics requires the introduction of fast light-responsive channel or pump proteins to the plasma membranes of target neuronal cells that allow temporally precise manipulation of neuronal membrane potential while maintaining cell-type resolution through the use of specific targeting mechanisms.
[0042] Light-responsive opsins that may be used in the present invention include opsins that induce hyperpolarization in neurons by light and opsins that induce depolarization in neurons by light. Examples of opsins are shown in Tables 1 and 2 below.
TABLE-US-00001 Opsin Biological Wavelength Type Origin Sensitivity Defined action NpHR Natronomonas 589 nm max Inhibition pharaonis (hyperpolarization) BR Halobacterium 570 nm max Inhibition helobium (hyperpolarization) AR Acetabulaira 518 nm max Inhibition acetabulum (hyperpolarization) GtR3 Guillardia 472 nm max Inhibition theta (hyperpolarization) Mac Leptosphaeria 470-500 nm max Inhibition maculans (hyperpolarization) NpHr3.0 Natronomonas 680 nm utility Inhibition pharaonis 589 nm max (hyperpolarization) NpHR3.1 Natronomonas 680 nm utility Inhibition pharaonis 589 nm max (hyperpolarization)
TABLE-US-00002 Wavelength Opsin Type Biological Origin Sensitivity Defined action VChR1 Volvox carteri 589 nm utility Excitation 535 nm max (depolarization) DChR Dunaliella salina 500 nm max Excitation (depolarization) ChR2 Chlamydomonas 470 nm max Excitation reinhardtii 380-405 nm utility (depolarization) ChETA Chlamydomonas 470 nm max Excitation reinhardtii 380-405 nm utility (depolarization) SFO Chlamydomonas 470 nm max Excitation reinhardtii 530 nm max (depolarization) Inactivation SSFO Chlamydomonas 445 nm max Step-like activation reinhardtii 590 nm; 390-400 nm (depolarization) Inactivation C1V1 Volvox carteri and 542 nm max Excitation Chlamydomonas (depolarization) reinhardtii C1V1 E122 Volvox carteri and 546 nm max Excitation Chlamydomonas (depolarization) reinhardtii C1V1 E162 Volvox carteri and 542 nm max Excitation Chlamydomonas (depolarization) reinhardtii C1V1 E122/E162 Volvox carteri and 546 nm max Excitation Chlamydomonas (depolarization) reinhardtii
[0043] As used herein, a light-responsive opsin (such as NpHR, BR, AR, GtR3, Mac, ChR2, VChR1, DChR, and ChETA) includes naturally occurring protein and functional variants, fragments, fusion proteins comprising the fragments, or the full length protein. For example, the signal peptide may be deleted. A variant may have an amino acid sequence at least about any of 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% identical to the naturally occurring protein sequence. A functional variant may have the same or similar hyperpolarization function or depolarization function as the naturally occurring protein.
[0044] Enhanced Intracellular Transport Amino Acid Motifs
[0045] The present disclosure provides for the modification of light-responsive opsin proteins expressed in a cell by the addition of one or more amino acid sequence motifs which enhance transport to the plasma membranes of mammalian cells. Light-responsive opsin proteins having components derived from evolutionarily simpler organisms may not be expressed or tolerated by mammalian cells or may exhibit impaired subcellular localization when expressed at high levels in mammalian cells. Consequently, in some embodiments, the light-responsive opsin proteins expressed in a cell can be fused to one or more amino acid sequence motifs selected from the group consisting of a signal peptide, an endoplasmic reticulum (ER) export signal, a membrane trafficking signal, and/or an N-terminal golgi export signal. The one or more amino acid sequence motifs which enhance light-responsive opsin protein transport to the plasma membranes of mammalian cells can be fused to the N-terminus, the C-terminus, or to both the N- and C-terminal ends of the light-responsive opsin protein. Optionally, the light-responsive opsin protein and the one or more amino acid sequence motifs may be separated by a linker. In some embodiments, the light-responsive opsin protein can be modified by the addition of a trafficking signal (ts) which enhances transport of the protein to the cell plasma membrane. In some embodiments, the trafficking signal can be derived from the amino acid sequence of the human inward rectifier potassium channel Kir2.1. In other embodiments, the trafficking signal can comprise the amino acid sequence KSRITSEGEYIPLDQIDINV.
[0046] Additional protein motifs which can enhance light-responsive opsin protein transport to the plasma membrane of a cell are described in U.S. Patent Application Publication No. 2009/0093403, which is incorporated herein by reference in its entirety. In some embodiments, the signal peptide sequence in the protein can be deleted or substituted with a signal peptide sequence from a different protein.
[0047] Light-Responsive Chloride Pumps
[0048] In some aspects, the light-responsive opsin proteins described herein are light-responsive chloride pumps. In some aspects of the methods provided herein, one or more members of the Halorhodopsin family of light-responsive chloride pumps are expressed on the plasma membranes of neurons of the central and peripheral nervous systems.
[0049] In some aspects, said one or more light-responsive chloride pump proteins expressed on the plasma membranes of nerve cells of the central or peripheral nervous systems can be derived from Natronomonas pharaonic. In some embodiments, the light-responsive chloride pump proteins can be responsive to amber light as well as red light and can mediate a hyperpolarizing current in the interneuron when the light-responsive chloride pump proteins are illuminated with amber or red light. The wavelength of light which can activate the light-responsive chloride pumps can be between about 580 and about 630 nm. In some embodiments, the light can be at a wavelength of about 590 nm or the light can have a wavelength greater than about 630 nm (e.g. less than about 740 nm). In another embodiment, the light has a wavelength of around 630 nm. In some embodiments, the light-responsive chloride pump protein can hyperpolarize a neural membrane for at least about 90 minutes when exposed to a continuous pulse of light. In some embodiments, the light-responsive chloride pump protein can comprise an amino acid sequence at least about 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% identical to the sequence shown in SEQ ID NO: 1. Additionally, the light-responsive chloride pump protein can comprise substitutions, deletions, and/or insertions introduced into a native amino acid sequence to increase or decrease sensitivity to light, increase or decrease sensitivity to particular wavelengths of light, and/or increase or decrease the ability of the light-responsive protein to regulate the polarization state of the plasma membrane of the cell. In some embodiments, the light-responsive chloride pump protein contains one or more conservative amino acid substitutions. In some embodiments, the light-responsive protein contains one or more non-conservative amino acid substitutions. The light-responsive protein comprising substitutions, deletions, and/or insertions introduced into the native amino acid sequence suitably retains the ability to hyperpolarize the plasma membrane of a neuronal cell in response to light.
[0050] Additionally, in other aspects, the light-responsive chloride pump protein can comprise a core amino acid sequence at least about 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% identical to the sequence shown in SEQ ID NO: 1 and an endoplasmic reticulum (ER) export signal. This ER export signal can be fused to the C-terminus of the core amino acid sequence or can be fused to the N-terminus of the core amino acid sequence. In some embodiments, the ER export signal is linked to the core amino acid sequence by a linker. The linker can comprise any of about 5, 10, 20, 30, 40, 50, 75, 100, 125, 150, 175, 200, 225, 250, 275, 300, 400, or 500 amino acids in length. The linker may further comprise a fluorescent protein, for example, but not limited to, a yellow fluorescent protein, a red fluorescent protein, a green fluorescent protein, or a cyan fluorescent protein. In some embodiments, the ER export signal can comprise the amino acid sequence FXYENE, where X can be any amino acid. In another embodiment, the ER export signal can comprise the amino acid sequence VXXSL, where X can be any amino acid. In some embodiments, the ER export signal can comprise the amino acid sequence FCYENEV.
[0051] In other aspects, the light-responsive chloride pump proteins provided herein can comprise a light-responsive protein expressed on the cell membrane, wherein the protein comprises a core amino acid sequence at least about 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% identical to the sequence shown in SEQ ID NO: 1 and a trafficking signal (e.g., which can enhance transport of the light-responsive chloride pump protein to the plasma membrane). The trafficking signal may be fused to the C-terminus of the core amino acid sequence or may be fused to the N-terminus of the core amino acid sequence. In some embodiments, the trafficking signal can be linked to the core amino acid sequence by a linker which can comprise any of about 5, 10, 20, 30, 40, 50, 75, 100, 125, 150, 175, 200, 225, 250, 275, 300, 400, or 500 amino acids in length. The linker may further comprise a fluorescent protein, for example, but not limited to, a yellow fluorescent protein, a red fluorescent protein, a green fluorescent protein, or a cyan fluorescent protein. In some embodiments, the trafficking signal can be derived from the amino acid sequence of the human inward rectifier potassium channel Kir2.1. In other embodiments, the trafficking signal can comprise the amino acid sequence KSRITSEGEYIPLDQIDINV.
[0052] In some aspects, the light-responsive chloride pump protein can comprise a core amino acid sequence at least about 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% identical to the sequence shown in SEQ ID NO: 1 and at least one (such as one, two, three, or more) amino acid sequence motifs which enhance transport to the plasma membranes of mammalian cells selected from the group consisting of an ER export signal, a signal peptide, and a membrane trafficking signal. In some embodiments, the light-responsive chloride pump protein comprises an N-terminal signal peptide, a C-terminal ER Export signal, and a C-terminal trafficking signal. In some embodiments, the C-terminal ER Export signal and the C-terminal trafficking signal can be linked by a linker. The linker can comprise any of about 5, 10, 20, 30, 40, 50, 75, 100, 125, 150, 175, 200, 225, 250, 275, 300, 400, or 500 amino acids in length. The linker can also further comprise a fluorescent protein, for example, but not limited to, a yellow fluorescent protein, a red fluorescent protein, a green fluorescent protein, or a cyan fluorescent protein. In some embodiments the ER Export signal can be more C-terminally located than the trafficking signal. In other embodiments the trafficking signal is more C-terminally located than the ER Export signal. In some embodiments, the signal peptide comprises the amino acid sequence MTETLPPVTESAVALQAE. In another embodiment, the light-responsive chloride pump protein comprises an amino acid sequence at least 95% identical to SEQ ID NO:2.
[0053] Moreover, in other aspects, the light-responsive chloride pump proteins can comprise a core amino acid sequence at least about 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% identical to the sequence shown in SEQ ID NO: 1, wherein the N-terminal signal peptide of SEQ ID NO:1 is deleted or substituted. In some embodiments, other signal peptides (such as signal peptides from other opsins) can be used. The light-responsive protein can further comprise an ER transport signal and/or a membrane trafficking signal described herein. In some embodiments, the light-responsive chloride pump protein comprises an amino acid sequence at least 95% identical to SEQ ID NO:3.
[0054] In some embodiments, the light-responsive opsin protein is a NpHR opsin protein comprising an amino acid sequence at least 95%, at least 96%, at least 97%, at least 98%, at least 99% or 100% identical to the sequence shown in SEQ ID NO:1. In some embodiments, the NpHR opsin protein further comprises an endoplasmic reticulum (ER) export signal and/or a membrane trafficking signal. For example, the NpHR opsin protein comprises an amino acid sequence at least 95% identical to the sequence shown in SEQ ID NO:1 and an endoplasmic reticulum (ER) export signal. In some embodiments, the amino acid sequence at least 95% identical to the sequence shown in SEQ ID NO:1 is linked to the ER export signal through a linker. In some embodiments, the ER export signal comprises the amino acid sequence FXYENE, where X can be any amino acid. In another embodiment, the ER export signal comprises the amino acid sequence VXXSL, where X can be any amino acid. In some embodiments, the ER export signal comprises the amino acid sequence FCYENEV. In some embodiments, the NpHR opsin protein comprises an amino acid sequence at least 95% identical to the sequence shown in SEQ ID NO:1, an ER export signal, and a membrane trafficking signal. In other embodiments, the NpHR opsin protein comprises, from the N-terminus to the C-terminus, the amino acid sequence at least 95% identical to the sequence shown in SEQ ID NO:1, the ER export signal, and the membrane trafficking signal. In other embodiments, the NpHR opsin protein comprises, from the N-terminus to the C-terminus, the amino acid sequence at least 95% identical to the sequence shown in SEQ ID NO:1, the membrane trafficking signal, and the ER export signal. In some embodiments, the membrane trafficking signal is derived from the amino acid sequence of the human inward rectifier potassium channel Kir2.1. In some embodiments, the membrane trafficking signal comprises the amino acid sequence KSRITSEGEYIPLDQIDINV. In some embodiments, the membrane trafficking signal is linked to the amino acid sequence at least 95% identical to the sequence shown in SEQ ID NO:1 by a linker. In some embodiments, the membrane trafficking signal is linked to the ER export signal through a linker. The linker may comprise any of 5, 10, 20, 30, 40, 50, 75, 100, 125, 150, 175, 200, 225, 250, 275, 300, 400, or 500 amino acids in length. The linker may further comprise a fluorescent protein, for example, but not limited to, a yellow fluorescent protein, a red fluorescent protein, a green fluorescent protein, or a cyan fluorescent protein. In some embodiments, the light-responsive opsin protein further comprises an N-terminal signal peptide. In some embodiments, the light-responsive opsin protein comprises the amino acid sequence of SEQ ID NO:2. In some embodiments, the light-responsive opsin protein comprises the amino acid sequence of SEQ ID NO:3.
[0055] Also provided herein are polynucleotides encoding any of the light-responsive chloride ion pump proteins described herein, such as a light-responsive protein comprising a core amino acid sequence at least about 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% identical to the sequence shown in SEQ ID NO:1, an ER export signal, and a membrane trafficking signal. In another embodiment, the polynucleotides comprise a sequence which encodes an amino acid at least 95% identical to SEQ ID NO:2 and/or SEQ ID NO:3. The polynucleotides may be in an expression vector (such as, but not limited to, a viral vector described herein). The polynucleotides may be used for expression of the light-responsive chloride ion pump proteins in neurons of the central or peripheral nervous systems.
[0056] Further disclosure related to light-responsive chloride pump proteins can be found in U.S. Patent Application Publication Nos: 2009/0093403 and 2010/0145418 as well as in International Patent Application No: PCT/US2011/028893, the disclosures of each of which are hereby incorporated by reference in their entireties.
[0057] Light-Responsive Proton Pumps
[0058] In some aspects, the light-responsive opsin proteins described herein are light-responsive proton pumps. In some aspects of the compositions and methods provided herein, one or more light-responsive proton pumps are expressed on the plasma membranes of neurons of the central or peripheral nervous systems.
[0059] In some embodiments, the light-responsive proton pump protein can be responsive to blue light and can be derived from Guillardia theta, wherein the proton pump protein can be capable of mediating a hyperpolarizing current in the cell when the cell is illuminated with blue light. The light can have a wavelength between about 450 and about 495 nm or can have a wavelength of about 490 nm. In another embodiment, the light-responsive proton pump protein can comprise an amino acid sequence at least about 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% identical to the sequence shown in SEQ ID NO:4. The light-responsive proton pump protein can additionally comprise substitutions, deletions, and/or insertions introduced into a native amino acid sequence to increase or decrease sensitivity to light, increase or decrease sensitivity to particular wavelengths of light, and/or increase or decrease the ability of the light-responsive proton pump protein to regulate the polarization state of the plasma membrane of the cell. Additionally, the light-responsive proton pump protein can contain one or more conservative amino acid substitutions and/or one or more non-conservative amino acid substitutions. The light-responsive proton pump protein comprising substitutions, deletions, and/or insertions introduced into the native amino acid sequence suitably retains the ability to hyperpolarize the plasma membrane of a neuronal cell in response to light.
[0060] In other aspects of the methods disclosed herein, the light-responsive proton pump protein can comprise a core amino acid sequence at least about 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% identical to the sequence shown in SEQ ID NO:4 and at least one (such as one, two, three, or more) amino acid sequence motifs which enhance transport to the plasma membranes of mammalian cells selected from the group consisting of a signal peptide, an ER export signal, and a membrane trafficking signal. In some embodiments, the light-responsive proton pump protein comprises an N-terminal signal peptide and a C-terminal ER export signal. In some embodiments, the light-responsive proton pump protein comprises an N-terminal signal peptide and a C-terminal trafficking signal. In some embodiments, the light-responsive proton pump protein comprises an N-terminal signal peptide, a C-terminal ER Export signal, and a C-terminal trafficking signal. In some embodiments, the light-responsive proton pump protein comprises a C-terminal ER Export signal and a C-terminal trafficking signal. In some embodiments, the C-terminal ER Export signal and the C-terminal trafficking signal are linked by a linker. The linker can comprise any of about 5, 10, 20, 30, 40, 50, 75, 100, 125, 150, 175, 200, 225, 250, 275, 300, 400, or 500 amino acids in length. The linker may further comprise a fluorescent protein, for example, but not limited to, a yellow fluorescent protein, a red fluorescent protein, a green fluorescent protein, or a cyan fluorescent protein. In some embodiments the ER Export signal is more C-terminally located than the trafficking signal. In some embodiments the trafficking signal is more C-terminally located than the ER Export signal.
[0061] Also provided herein are isolated polynucleotides encoding any of the light-responsive proton pump proteins described herein, such as a light-responsive proton pump protein comprising a core amino acid sequence at least about 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% identical to the sequence shown in SEQ ID NO:4. Also provided herein are expression vectors (such as a viral vector described herein) comprising a polynucleotide encoding the proteins described herein, such as a light-responsive proton pump protein comprising a core amino acid sequence at least about 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% identical to the sequence shown in SEQ ID NO:4. The polynucleotides may be used for expression of the light-responsive proton pumps in neural cells of the central or peripheral nervous systems.
[0062] Further disclosure related to light-responsive proton pump proteins can be found in International Patent Application No. PCT/US2011/028893, the disclosure of which is hereby incorporated by reference in its entirety.
[0063] Light-Activated Cation Channel Proteins
[0064] In some aspects, the light-responsive opsin proteins described herein are light-activated cation channel proteins. In some aspects of the methods provided herein, one or more light-activated cation channels can be expressed on the plasma membranes of the neural cells of the central or peripheral nervous systems.
[0065] In some aspects, the light-activated cation channel protein can be derived from Chlamydomonas reinhardtii, wherein the cation channel protein can be capable of mediating a depolarizing current in the cell when the cell is illuminated with light. In another embodiment, the light-activated cation channel protein can comprise an amino acid sequence at least about 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% identical to the sequence shown in SEQ ID NO:5. The light used to activate the light-activated cation channel protein derived from Chlamydomonas reinhardtii can have a wavelength between about 460 and about 495 nm or can have a wavelength of about 480 nm. Additionally, the light can have an intensity of at least about 100 Hz. In some embodiments, activation of the light-activated cation channel derived from Chlamydomonas reinhardtii with light having an intensity of 100 Hz can cause depolarization-induced synaptic depletion of the neurons expressing the light-activated cation channel. The light-activated cation channel protein can additionally comprise substitutions, deletions, and/or insertions introduced into a native amino acid sequence to increase or decrease sensitivity to light, increase or decrease sensitivity to particular wavelengths of light, and/or increase or decrease the ability of the light-activated cation channel protein to regulate the polarization state of the plasma membrane of the cell.
[0066] Additionally, the light-activated cation channel protein can contain one or more conservative amino acid substitutions and/or one or more non-conservative amino acid substitutions. The light-activated proton pump protein comprising substitutions, deletions, and/or insertions introduced into the native amino acid sequence suitably retains the ability to depolarize the plasma membrane of a neuronal cell in response to light.
[0067] Further disclosure related to light-activated cation channel proteins can be found in U.S. Patent Application Publication No. 2007/0054319 and International Patent Application Publication Nos. WO 2009/131837 and WO 2007/024391, the disclosures of each of which are hereby incorporated by reference in their entireties.
[0068] Step Function Opsins and Stabilized Step Function Opsins
[0069] In other embodiments, the light-activated cation channel protein can be a step function opsin (SFO) protein or a stabilized step function opsin (SSFO) protein that can have specific amino acid substitutions at key positions throughout the retinal binding pocket of the protein. In some embodiments, the SFO protein can have a mutation at amino acid residue C128 of SEQ ID NO:5. In other embodiments, the SFO protein has a C128A mutation in SEQ ID NO:5. In other embodiments, the SFO protein has a C128S mutation in SEQ ID NO:5. In another embodiment, the SFO protein has a C128T mutation in SEQ ID NO:5. In some embodiments, the SFO protein can comprise an amino acid sequence at least about 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% identical to the sequence shown in SEQ ID NO:6.
[0070] In some embodiments, the SSFO protein can have a mutation at amino acid residue D156 of SEQ ID NO:5. In other embodiments, the SSFO protein can have a mutation at both amino acid residues C128 and D156 of SEQ ID NO:5. In one embodiment, the SSFO protein has an C128S and a D156A mutation in SEQ ID NO:5. In another embodiment, the SSFO protein can comprise an amino acid sequence at least about 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% identical to the sequence shown in SEQ ID NO:7.
[0071] In some embodiments the SFO or SSFO proteins provided herein can be capable of mediating a depolarizing current in the cell when the cell is illuminated with blue light. In other embodiments, the light can have a wavelength of about 445 nm. Additionally, the light can have an intensity of about 100 Hz. In some embodiments, activation of the SFO or SSFO protein with light having an intensity of 100 Hz can cause depolarization-induced synaptic depletion of the neurons expressing the SFO or SSFO protein. In some embodiments, each of the disclosed step function opsin and stabilized step function opsin proteins can have specific properties and characteristics for use in depolarizing the membrane of a neuronal cell in response to light.
[0072] Further disclosure related to SFO or SSFO proteins can be found in International Patent Application Publication No. WO 2010/056970 and U.S. Provisional Patent Application Nos. 61/410,704 and 61/511,905, the disclosures of each of which are hereby incorporated by reference in their entireties.
[0073] C1V1 Chimeric Cation Channels
[0074] In other embodiments, the light-activated cation channel protein can be a C1V1 chimeric protein derived from the VChR1 protein of Volvox carteri and the ChR1 protein from Chlamydomonas reinhardti, wherein the protein comprises the amino acid sequence of VChR1 having at least the first and second transmembrane helices replaced by the first and second transmembrane helices of ChR1; is responsive to light; and is capable of mediating a depolarizing current in the cell when the cell is illuminated with light. In some embodiments, the C1V1 protein can further comprise a replacement within the intracellular loop domain located between the second and third transmembrane helices of the chimeric light responsive protein, wherein at least a portion of the intracellular loop domain is replaced by the corresponding portion from ChR1. In another embodiment, the portion of the intracellular loop domain of the C1V1 chimeric protein can be replaced with the corresponding portion from ChR1 extending to amino acid residue A145 of the ChR1. In other embodiments, the C1V1 chimeric protein can further comprise a replacement within the third transmembrane helix of the chimeric light responsive protein, wherein at least a portion of the third transmembrane helix is replaced by the corresponding sequence of ChR1. In yet another embodiment, the portion of the intracellular loop domain of the C1V1 chimeric protein can be replaced with the corresponding portion from ChR1 extending to amino acid residue W163 of the ChR1. In other embodiments, the C1V1 chimeric protein can comprise an amino acid sequence at least about 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% identical to the sequence shown in SEQ ID NO:8.
[0075] In some embodiments, the C1V1 protein can mediate a depolarizing current in the cell when the cell is illuminated with green light. In other embodiments, the light can have a wavelength of between about 540 nm to about 560 nm. In some embodiments, the light can have a wavelength of about 542 nm. In some embodiments, the C1V1 chimeric protein is not capable of mediating a depolarizing current in the cell when the cell is illuminated with violet light. In some embodiments, the chimeric protein is not capable of mediating a depolarizing current in the cell when the cell is illuminated with light having a wavelength of about 405 nm. Additionally, the light can have an intensity of about 100 Hz. In some embodiments, activation of the C1V1 chimeric protein with light having an intensity of 100 Hz can cause depolarization-induced synaptic depletion of the neurons expressing the C1V1 chimeric protein. In some embodiments, the disclosed C1V1 chimeric protein can have specific properties and characteristics for use in depolarizing the membrane of a neuronal cell in response to light.
[0076] C1V1 Chimeric Mutant Variants
[0077] In some aspects, the invention can include polypeptides comprising substituted or mutated amino acid sequences, wherein the mutant polypeptide retains the characteristic light-responsive nature of the precursor C1V1 chimeric polypeptide but may also possess altered properties in some specific aspects. For example, the mutant light-activated C1V1 chimeric proteins described herein can exhibit an increased level of expression both within an animal cell or on the animal cell plasma membrane; an altered responsiveness when exposed to different wavelengths of light, particularly red light; and/or a combination of traits whereby the chimeric C1V1 polypeptide possess the properties of low desensitization, fast deactivation, low violet-light activation for minimal cross-activation with other light-activated cation channels, and/or strong expression in animal cells.
[0078] Accordingly, provided herein are C1V1 chimeric light-activated proteins that can have specific amino acid substitutions at key positions throughout the retinal binding pocket of the VChR1 portion of the chimeric polypeptide. In some embodiments, the C1V1 protein can have a mutation at amino acid residue E122 of SEQ ID NO:7. In some embodiments, the C1V1 protein can have a mutation at amino acid residue E162 of SEQ ID NO:7. In other embodiments, the C1V1 protein can have a mutation at both amino acid residues E162 and E122 of SEQ ID NO:7. In other embodiments, the C1V1 protein can comprise an amino acid sequence at least about 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% identical to the sequence shown in SEQ ID NO:9, SEQ ID NO:10, or SEQ ID NO:11. In some embodiments, each of the disclosed mutant C1V1 chimeric proteins can have specific properties and characteristics for use in depolarizing the membrane of an animal cell in response to light.
[0079] In some aspects, the C1V1-E122 mutant chimeric protein is capable of mediating a depolarizing current in the cell when the cell is illuminated with light. In some embodiments the light can be green light. In other embodiments, the light can have a wavelength of between about 540 nm to about 560 nm. In some embodiments, the light can have a wavelength of about 546 nm. In other embodiments, the C1V1-E122 mutant chimeric protein can mediate a depolarizing current in the cell when the cell is illuminated with red light. In some embodiments, the red light can have a wavelength of about 630 nm. In some embodiments, the C1V1-E122 mutant chimeric protein does not mediate a depolarizing current in the cell when the cell is illuminated with violet light. In some embodiments, the chimeric protein does not mediate a depolarizing current in the cell when the cell is illuminated with light having a wavelength of about 405 nm. Additionally, the light can have an intensity of about 100 Hz. In some embodiments, activation of the C1V1-E122 mutant chimeric protein with light having an intensity of 100 Hz can cause depolarization-induced synaptic depletion of the neurons expressing the C1V1-E122 mutant chimeric protein. In some embodiments, the disclosed C1V1-E122 mutant chimeric protein can have specific properties and characteristics for use in depolarizing the membrane of a neuronal cell in response to light.
[0080] In other aspects, the C1V1-E162 mutant chimeric protein is capable of mediating a depolarizing current in the cell when the cell is illuminated with light. In some embodiments the light can be green light. In other embodiments, the light can have a wavelength of between about 540 nm to about 535 nm. In some embodiments, the light can have a wavelength of about 542 nm. In other embodiments, the light can have a wavelength of about 530 nm. In some embodiments, the C1V1-E162 mutant chimeric protein does not mediate a depolarizing current in the cell when the cell is illuminated with violet light. In some embodiments, the chimeric protein does not mediate a depolarizing current in the cell when the cell is illuminated with light having a wavelength of about 405 nm. Additionally, the light can have an intensity of about 100 Hz. In some embodiments, activation of the C1V1-E162 mutant chimeric protein with light having an intensity of 100 Hz can cause depolarization-induced synaptic depletion of the neurons expressing the C1V1-E162 mutant chimeric protein. In some embodiments, the disclosed C1V1-E162 mutant chimeric protein can have specific properties and characteristics for use in depolarizing the membrane of a neuronal cell in response to light.
[0081] In yet other aspects, the C1V1-E122/E162 mutant chimeric protein is capable of mediating a depolarizing current in the cell when the cell is illuminated with light. In some embodiments the light can be green light. In other embodiments, the light can have a wavelength of between about 540 nm to about 560 nm. In some embodiments, the light can have a wavelength of about 546 nm. In some embodiments, the C1V1-E122/E162 mutant chimeric protein does not mediate a depolarizing current in the cell when the cell is illuminated with violet light. In some embodiments, the chimeric protein does not mediate a depolarizing current in the cell when the cell is illuminated with light having a wavelength of about 405 nm. In some embodiments, the C1V1-E122/E162 mutant chimeric protein can exhibit less activation when exposed to violet light relative to C1V1 chimeric proteins lacking mutations at E122/E162 or relative to other light-activated cation channel proteins. Additionally, the light can have an intensity of about 100 Hz. In some embodiments, activation of the C1V1-E122/E162 mutant chimeric protein with light having an intensity of 100 Hz can cause depolarization-induced synaptic depletion of the neurons expressing the C1V1-E122/E162 mutant chimeric protein. In some embodiments, the disclosed C1V1-E122/E162 mutant chimeric protein can have specific properties and characteristics for use in depolarizing the membrane of a neuronal cell in response to light.
[0082] Further disclosure related to C1V1 chimeric cation channels as well as mutant variants of the same can be found in U.S. Provisional Patent Application Nos. 61/410,736, 61/410,744, and 61/511,912, the disclosures of each of which are hereby incorporated by reference in their entireties.
[0083] Polynucleotides
[0084] The disclosure also provides polynucleotides comprising a nucleotide sequence encoding a light-responsive opsin protein described herein. In some embodiments, the polynucleotide comprises an expression cassette. In some embodiments, the polynucleotide is a vector comprising the above-described nucleic acid(s). In some embodiments, the nucleic acid encoding a light-activated protein of the disclosure is operably linked to a promoter. Promoters are well known in the art. Any promoter that functions in the host cell can be used for expression of the light-responsive opsin proteins and/or any variant thereof of the present disclosure. In one embodiment, the promoter used to drive expression of the light-responsive opsin proteins is a promoter that is specific to motor neurons. In another embodiment, the promoter used to drive expression of the light-responsive opsin proteins is a promoter that is specific to central nervous system neurons. In other embodiments, the promoter is capable of driving expression of the light-responsive opsin proteins in neurons of both the sympathetic and/or the parasympathetic nervous systems. Initiation control regions or promoters, which are useful to drive expression of the light-responsive opsin proteins or variant thereof in a specific animal cell are numerous and familiar to those skilled in the art. Virtually any promoter capable of driving these nucleic acids can be used. Examples of motor neuron-specific genes can be found, for example, in Kudo, et al., Human Mol. Genetics, 2010, 19(16): 3233-3253, the contents of which are hereby incorporated by reference in their entirety. In some embodiments, the promoter used to drive expression of the light-activated protein can be the Thy1 promoter, which is capable of driving robust expression of transgenes in neurons of both the central and peripheral nervous systems (See, e.g., Llewellyn, et al., 2010, Nat. Med., 16(10):1161-1166). In other embodiments, the promoter used to drive expression of the light-responsive opsin protein can be the EF1.alpha. promoter, a cytomegalovirus (CMV) promoter, the CAG promoter, the sinapsin promoter, or any other ubiquitous promoter capable of driving expression of the light-responsive opsin proteins in the peripheral and/or central nervous system neurons of mammals.
[0085] Also provided herein are vectors comprising a nucleotide sequence encoding a light-responsive opsin protein or any variant thereof described herein. The vectors that can be administered according to the present invention also include vectors comprising a nucleotide sequence which encodes an RNA (e.g., an mRNA) that when transcribed from the polynucleotides of the vector will result in the accumulation of light-responsive opsin proteins on the plasma membranes of target animal cells. Vectors which may be used, include, without limitation, lentiviral, HSV, adenoviral, and andeno-associated viral (AAV) vectors. Lentiviruses include, but are not limited to HW-1, HIV-2, SW, FW and EIAV. Lentiviruses may be pseudotyped with the envelope proteins of other viruses, including, but not limited to VSV, rabies, Mo-MLV, baculovirus and Ebola. Such vectors may be prepared using standard methods in the art.
[0086] In some embodiments, the vector is a recombinant AAV vector. AAV vectors are DNA viruses of relatively small size that can integrate, in a stable and site-specific manner, into the genome of the cells that they infect. They are able to infect a wide spectrum of cells without inducing any effects on cellular growth, morphology or differentiation, and they do not appear to be involved in human pathologies. The AAV genome has been cloned, sequenced and characterized. It encompasses approximately 4700 bases and contains an inverted terminal repeat (ITR) region of approximately 145 bases at each end, which serves as an origin of replication for the virus. The remainder of the genome is divided into two essential regions that carry the encapsidation functions: the left-hand part of the genome, that contains the rep gene involved in viral replication and expression of the viral genes; and the right-hand part of the genome, that contains the cap gene encoding the capsid proteins of the virus.
[0087] AAV vectors may be prepared using standard methods in the art. Adeno-associated viruses of any serotype are suitable (See, e.g., Blacklow, pp. 165-174 of "Parvoviruses and Human Disease" J. R. Pattison, ed. (1988); Rose, Comprehensive Virology 3:1, 1974; P. Tattersall "The Evolution of Parvovirus Taxonomy" in Parvoviruses (J R Kerr, S F Cotmore. M E Bloom, R M Linden, C R Parrish, Eds.) p 5-14, Hudder Arnold, London, U K (2006); and D E Bowles, J E Rabinowitz, R T Samulski "The Genus Dependovirus" (J R Kerr, S F Cotmore. M E Bloom, R M Linden, C R Parrish, Eds.) p 15-23, Hudder Arnold, London, UK (2006), the disclosures of each of which are hereby incorporated by reference herein in their entireties). Methods for purifying for vectors may be found in, for example, U.S. Pat. Nos. 6,566,118, 6,989,264, and 6,995,006 and International Patent Application Publication No.: WO/1999/011764 titled "Methods for Generating High Titer Helper-free Preparation of Recombinant AAV Vectors", the disclosures of which are herein incorporated by reference in their entirety. Preparation of hybrid vectors is described in, for example, PCT Application No. PCT/US2005/027091, the disclosure of which is herein incorporated by reference in its entirety. The use of vectors derived from the AAVs for transferring genes in vitro and in vivo has been described (See e.g., International Patent Application Publication Nos.: WO 91/18088 and WO 93/09239; U.S. Pat. Nos. 4,797,368, 6,596,535, and 5,139,941; and European Patent No.: 0488528, all of which are hereby incorporated by reference herein in their entireties). These publications describe various AAV-derived constructs in which the rep and/or cap genes are deleted and replaced by a gene of interest, and the use of these constructs for transferring the gene of interest in vitro (into cultured cells) or in vivo (directly into an organism). The replication defective recombinant AAVs according to the invention can be prepared by co-transfecting a plasmid containing the nucleic acid sequence of interest flanked by two AAV inverted terminal repeat (ITR) regions, and a plasmid carrying the AAV encapsidation genes (rep and cap genes), into a cell line that is infected with a human helper virus (for example, an adenovirus). The AAV recombinants that are produced are then purified by standard techniques.
[0088] In some embodiments, the vector(s) for use in the methods of the invention are encapsidated into a virus particle (e.g. AAV virus particle including, but not limited to, AAV1, AAV2, AAV3, AAV4, AAV5, AAV6, AAV7, AAV8, AAV9, AAV10, AAV11, AAV12, AAV13, AAV14, AAV15, and AAV16). Accordingly, the invention includes a recombinant virus particle (recombinant because it contains a recombinant polynucleotide) comprising any of the vectors described herein. Methods of producing such particles are known in the art and are described in U.S. Pat. No. 6,596,535, the disclosure of which is hereby incorporated by reference in its entirety.
[0089] Delivery of Light-Responsive Opsin Proteins and Lanthanide-Doped Nanoparticles
[0090] In some aspects, polynucleotides encoding the light-responsive opsin proteins disclosed herein (for example, an AAV1 vector) can be delivered directly to neurons of the central or peripheral nervous system with a needle, catheter, or related device, using neurosurgical techniques known in the art, such as by stereotactic injection (See, e.g., Stein et al., J. Virol., 1999, 73:34243429; Davidson et al., Proc. Nat. Acad. Sci. U.S.A., 2000, 97:3428-3432; Davidson et al., Nat. Genet., 1993, 3:219-223; and Alisky & Davidson, Hum. Gene Ther., 2000, 11:2315-2329, the contents of each of which are hereby incorporated by reference herein in their entireties) or fluoroscopy. In some embodiments, the polynucleotide encoding the light-responsive opsin proteins disclosed herein (for example, an AAV1 vector) can be delivered to neurons of the peripheral nervous system by injection into any one of the spinal nerves (such as the cervical spinal nerves, the thoracic spinal nerves, the lumbar spinal nerves, the sacral spinal nerves, and/or the coccygeal spinal nerves).
[0091] Other methods to deliver the light-responsive opsin proteins to the nerves of interest can also be used, such as, but not limited to, transfection with ionic lipids or polymers, electroporation, optical transfection, impalefection, or via gene gun.
[0092] In another aspect, the polynucleotide encoding the light-responsive opsin proteins disclosed herein (for example, an AAV2 vector) can be delivered directly to muscles innervated by the neurons of the peripheral nervous system. Because of the limitations inherent in injecting viral vectors directly into the specific cell bodies which innvervate particular muscles, researchers have attempted to deliver transgenes to peripheral neurons by injecting viral vectors directly into muscle. These experiments have shown that some viral serotypes such as adenovirus, AAV2, and Rabies glycoprotein-pseudotyped lentivirus can be taken up by muscle cells and retrogradely transported to motor neurons across the neuromuscular synapse (See, e.g., Azzouz et al., 2009, Antioxid Redox Signal., 11(7):1523-34; Kaspar et al., 2003, Science, 301(5634):839-842; Manabe et al., 2002, Apoptosis, 7(4):329-334, the disclosures of each of which are herein incorporated by reference in their entireties).
[0093] Accordingly, in some embodiments, the vectors expressing the light-responsive opsin proteins disclosed herein (for example, an AAV2 vector) can be delivered to the neurons responsible for the innervation of muscles by direct injection into the muscle of interest.
[0094] The lanthanide-doped nanoparticles disclosed herein can be delivered to neurons expressing one or more light-responsive opsin proteins by any route, such as intravascularly, intracranially, intracerebrally, intramuscularly, intradermally, intravenously, intraocularly, orally, nasally, topically, or by open surgical procedure, depending upon the anatomical site or sites to which the nanoparticles are to be delivered. The nanoparticles can additionally be delivered by the same route used for delivery of the polynucleotide vectors expressing the light-responsive opsin proteins, such as any of those described above. The nanoparticles can also be administered in an open manner, as in the heart during open heart surgery, or in the brain during stereotactic surgery, or by intravascular interventional methods using catheters going to the blood supply of specific organs, or by other interventional methods.
[0095] Pharmaceutical compositions used for the delivery and/or storage of polynucleotides encoding the light-responsive opsin proteins disclosed herein and/or the lanthanide-doped nanoparticles disclosed herein can be formulated according to known methods for preparing pharmaceutically useful compositions. Formulations are described in a number of sources which are well known and readily available to those skilled in the art. For example, Remington's Pharmaceutical Sciences (Martin E W, 1995, Easton Pa., Mack Publishing Company, 19.sup.th ed.) describes formulations which can be used in connection with the subject invention. Formulations suitable for parenteral administration include, for example, aqueous sterile injection solutions, which may contain antioxidants, buffers, bacteriostats, and solutes which render the formulation isotonic with the blood of the intended recipient; and aqueous and non-aqueous sterile suspensions which may include suspending agents and thickening agents. The formulations may be presented in unit-dose or multi-dose containers, for example, sealed ampoules and vials, and may be stored in a freeze dried (lyophilized) condition requiring only the condition of the sterile liquid carrier, for example, water for injections, prior to use.
[0096] The lanthanide-doped nanoparticles may also be administered intravenously or intraperitoneally by infusion or injection. Solutions of the nanoparticles and/or cells can be prepared in water, optionally mixed with a nontoxic surfactant. Dispersions can also be prepared in glycerol, liquid polyethylene glycols, triacetin, and mixtures thereof and in oils. Under ordinary conditions of storage and use, these preparations contain a preservative to prevent the growth of microorganisms.
[0097] The pharmaceutical dosage forms suitable for injection or infusion of the lanthanide-doped nanoparticles described herein can include sterile aqueous solutions or dispersions or sterile powders comprising the active ingredient which are adapted for the extemporaneous preparation of sterile injectable or infusible solutions or dispersions. The liquid carrier or vehicle can be a solvent or liquid dispersion medium comprising, for example, water, ethanol, a polyol (for example, glycerol, propylene glycol, liquid polyethylene glycols, and the like), vegetable oils, nontoxic glyceryl esters, and suitable mixtures thereof. The prevention of the action of microorganisms can be brought about by various antibacterial and antifungal agents, for example, parabens, chlorobutanol, phenol, sorbic acid, thimerosal, and the like.
[0098] Sources of Infrared or Near Infrared Electromagnetic Radiation
[0099] Any device that is capable of producing a source of electromagnetic radiation having a wavelength in the infrared (IR) or near infrared (NIR) spectrum may be used to activate one or more light-responsive proteins expressed on the surface of a neuron in combination with the lanthanide-doped nanoparticles described herein. The IR or NIR source can be configured to provide optical stimulus to a specific target region of the brain. The IR or NIR source can additionally provide continuous IR or NIR electromagnetic radiation and/or pulsed IR or NIR electromagnetic radiation, and may be programmable to provide IR or NIR electromagnetic radiation in pre-determined pulse sequences.
[0100] In other aspects, the implantable IR or NIR source does not require physical tethering to an external power source. In some embodiments, the power source can be an internal battery for powering the IR or NIR source. In another embodiment, the implantable IR or NIR source can comprise an external antenna for receiving wirelessly transmitted electromagnetic energy from an external power source for powering the IR or NIR source. The wirelessly transmitted electromagnetic energy can be a radio wave, a microwave, or any other electromagnetic energy source that can be transmitted from an external source to power the IR or NIR-generating source. In one embodiment, the IR or NIR source is controlled by an integrated circuit produced using semiconductor or other processes known in the art.
[0101] In some aspects, the implantable IR or NIR electromagnetic radiation source can be externally activated by an external controller. The external controller can comprise a power generator which can be mounted to a transmitting coil. In some embodiments of the external controller, a battery can be connected to the power generator, for providing power thereto. A switch can be connected to the power generator, allowing an individual to manually activate or deactivate the power generator. In some embodiments, upon activation of the switch, the power generator can provide power to the IR or NIR electromagnetic radiation source through electromagnetic coupling between the transmitting coil on the external controller and the external antenna of the implantable IR or NIR source. When radio-frequency magnetic inductance coupling is used, the operational frequency of the radio wave can be between about 1 and 20 MHz, inclusive, including any values in between these numbers (for example, about 1 MHz, about 2 MHz, about 3 MHz, about 4 MHz, about 5 MHz, about 6 MHz, about 7 MHz, about 8 MHz, about 9 MHz, about 10 MHz, about 11 MHz, about 12 MHz, about 13 MHz, about 14 MHz, about 15 MHz, about 16 MHz, about 17 MHz, about 18 MHz, about 19 MHz, or about 20 MHz). However, other coupling techniques may be used, such as an optical receiver or a biomedical telemetry system (See, e.g., Kiourti, "Biomedical Telemetry: Communication between Implanted Devices and the External World, Opticon 1826, (8): Spring, 2010).
[0102] In some aspects, the intensity of the IR or NIR electromagnetic radiation reaching the neural cells (such as neural cells expressing one or more light-responsive opsin proteins) produced by the IR or NW electromagnetic radiation source has an intensity of any of about 0.05 mW/mm.sup.2, 0.1 mW/mm.sup.2, 0.2 mW/mm.sup.2, 0.3 mW/mm.sup.2, 0.4 mW/mm.sup.2, 0.5 mW/mm.sup.2, about 0.6 mW/mm.sup.2, about 0.7 mW/mm.sup.2, about 0.8 mW/mm.sup.2, about 0.9 mW/mm.sup.2, about 1.0 mW/mm.sup.2, about 1.1 mW/mm.sup.2, about 1.2 mW/mm.sup.2, about 1.3 mW/mm.sup.2, about 1.4 mW/mm.sup.2, about 1.5 mW/mm.sup.2, about 1.6 mW/mm.sup.2, about 1.7 mW/mm.sup.2, about 1.8 mW/mm.sup.2, about 1.9 mW/mm.sup.2, about 2.0 mW/mm.sup.2, about 2.1 mW/mm.sup.2, about 2.2 mW/mm.sup.2, about 2.3 mW/mm.sup.2, about 2.4 mW/mm.sup.2, about 2.5 mW/mm.sup.2, about 3 mW/mm.sup.2, about 3.5 mW/mm.sup.2, about 4 mW/mm.sup.2, about 4.5 mW/mm.sup.2, about 5 mW/mm.sup.2, about 5.5 mW/mm.sup.2, about 6 mW/mm.sup.2, about 7 mW/mm.sup.2, about 8 mW/mm.sup.2, about 9 mW/mm.sup.2, or about 10 mW/mm.sup.2, inclusive, including values in between these numbers.
[0103] In other aspects, the IR or NIR electromagnetic radiation produced by the IR or NW electromagnetic radiation source can have a wavelength encompassing the entire infrared spectrum, such as from about 740 nm to about 300,000 nm. In other embodiments, the IR or NIR electromagnetic radiation produced by the IR or NIR electromagnetic radiation source can have a wavelength corresponding to the NIR spectrum, such as about 740 nm to about 1400 nm. In other embodiments, NIR electromagnetic radiation produced has a wavelength between 700 nm and 1000 nm.
[0104] In some aspects, an IR or NIR electromagnetic radiation source is used to hyperpolarize or depolarize the plasma membranes of neural cells (such as neural cells expressing one or more light-responsive opsin proteins) in the brain or central nervous system of an individual when used in combination with the lanthanide-doped nanoparticles disclosed herein. In some embodiments, the skull of the individual is surgically thinned in an area adjacent to the brain region of interest without puncturing the bone. The IR or NW electromagnetic radiation source can then be placed directly over the thinned-skull region. In other embodiments, the IR or NIR electromagnetic radiation generator is implanted under the skin of the individual directly adjacent to the thinned skull region.
[0105] In some aspects, an IR or NIR electromagnetic radiation source is used to hyperpolarize or depolarize the plasma membranes of neural cells (such as neural cells expressing one or more light-responsive opsin proteins) in the peripheral nervous system of an individual when used in combination with the lanthanide-doped nanoparticles disclosed herein. In some embodiments, the IR or NIR electromagnetic radiation source is surgically implanted under the skin of the individual directly adjacent to the peripheral neural cell of interest. In other embodiments, the IR or NIR electromagnetic radiation source is placed against the skin directly adjacent to the peripheral neural cell of interest. In one embodiment, the IR or NIR electromagnetic radiation source is held against the skin in a bracelet or cuff configuration.
[0106] Examples of the IR or NIR electromagnetic radiation sources, particularly those small enough to be implanted under the skin, can be found in U.S. Patent Application Publication Nos.: 2009/0143842, 2011/0152969, 2011/0144749, and 2011/0054305, the disclosures of each of which are incorporated by reference herein in their entireties.
[0107] In still other aspects, the lanthanide-doped nanoparticles disclosed herein can be exposed to higher wavelength light in the visible spectrum (such as red light) to upconvert the higher wavelength visible light into lower wavelength visible light (such as blue or green light). As described above, light passes through biological tissue poorly. However, when visible light does penetrate into tissues, it typically does so in higher wavelengths which correspond to red light (for example, between about 620 nm to 740 nm). Accordingly, the lanthanide-doped nanoparticles disclosed herein can additionally be used in combination with optical sources of visible light to upshift wavelengths corresponding to red light into wavelengths corresponding to green or blue light (for example, between about 440 nm and 570 nm). Examples of light stimulation devices, including light sources, can be found in International Patent Application Nos.: PCT/US08/50628 and PCT/US09/49936 and in Llewellyn et al., 2010, Nat. Med., 16(10):161-165, the disclosures of each of which are hereby incorporated herein in their entireties.
Methods of the Invention
[0108] Depolarization of Neural Cells
[0109] Provided herein are methods to depolarize the plasma membrane of a neural cell in an individual comprising placing a plurality of lanthanide-doped nanoparticles in proximity to the neural cell; and exposing the plurality of nanoparticles to electromagnetic radiation in the infrared (IR) or near infrared (NM) spectrum, wherein the electromagnetic radiation in the IR or NIR spectrum is upconverted into light in the visible spectrum by the nanoparticles, and wherein a light-responsive opsin is expressed on the plasma membrane of the neural cells and activation of the opsin by the light in the visible spectrum induces the depolarization of the plasma membrane.
[0110] Also provided herein is a method to depolarize the plasma membrane of a neural cell in an individual comprising administering a polynucleotide encoding a light-responsive opsin to a neural cell in the brain of an individual, wherein the light-responsive protein is expressed on the plasma membrane of the neural cell and the opsin is capable of inducing membrane depolarization of the neural cell when illuminated with light administering a plurality of lanthanide-doped nanoparticles in proximity to the neural cell; and exposing the plurality of nanoparticles to electromagnetic radiation in the infrared (IR) or near (IR) spectrum, wherein the electromagnetic radiation in the IR or near IR spectrum is upconverted into light in the visible spectrum and the activation of the opsin by the light in the visible spectrum induces the depolarization of the plasma membrane.
[0111] In some embodiments, the light-responsive opsin protein is ChR2, VChR1, or C1V1. In other embodiments, the light-responsive opsin protein is selected from the group consisting of SFO, SSFO, C1V1-E122, C1V1-E162, and C1V1-E122/E162.
[0112] The lanthanide metal can be ions or atoms from any of the lanthanide series of elements, such as Lanthanum, Cerium, Praseodymium, Neodymium, Promethium, Samarium, Europium, Gadolinium, Terbium, Dysprosium, Holmium, Erbium, Thulium, Ytterbium, or Lutetium. In other embodiments, the nanoparticles comprise NaYF4:Yb/X/Gd, wherein X is Er, Tm, or Er/Tm.
[0113] The electromagnetic radiation in the IR or near IR spectrum can be upconverted into light having a wavelength of about 450 nm to about 550 nm. The light can have wavelengths corresponding to red, yellow, amber, orange, green, or blue light. In some embodiments, the individual is a human or a non-human animal. In other embodiments, the neural cell is in the peripheral nervous system. In another embodiment, the neural cell is in the central nervous system.
[0114] Hyperpolarization of Neural Cells
[0115] Provided herein are methods to hyperpolarize the plasma membrane of a neural cell in an individual comprising placing a plurality of lanthanide-doped nanoparticles in proximity to the neural cell; and exposing the plurality of nanoparticles to electromagnetic radiation in the infrared (IR) or near infrared (NIR) spectrum, wherein the electromagnetic radiation in the IR or NIR spectrum is upconverted into light in the visible spectrum by the nanoparticles, and wherein a light-responsive opsin is expressed on the plasma membrane of the neural cells and activation of the opsin by the light in the visible spectrum induces the hyperpolarization of the plasma membrane.
[0116] Also provided herein is a method to hyperpolarize the plasma membrane of a neural cell in an individual comprising administering a polynucleotide encoding a light-responsive opsin to a neural cell in the brain of an individual, wherein the light-responsive protein is expressed on the plasma membrane of the neural cell and the opsin is capable of inducing membrane depolarization of the neural cell when illuminated with light administering a plurality of lanthanide-doped nanoparticles in proximity to the neural cell; and exposing the plurality of nanoparticles to electromagnetic radiation in the infrared (IR) or near (IR) spectrum, wherein the electromagnetic radiation in the IR or near IR spectrum is upconverted into light in the visible spectrum and the activation of the opsin by the light in the visible spectrum induces the hyperpolarization of the plasma membrane.
[0117] In some embodiments, the light-responsive opsin protein is an NpHR or a GtR3.
[0118] The lanthanide metal can be ions or atoms from any of the lanthanide series of elements, such as Lanthanum, Cerium, Praseodymium, Neodymium, Promethium, Samarium, Europium, Gadolinium, Terbium, Dysprosium, Holmium, Erbium, Thulium, Ytterbium, or Lutetium. In other embodiments, the nanoparticles comprise NaYF4:Yb/X/Gd, wherein X is Er, Tm, or Er/Tm.
[0119] The electromagnetic radiation in the IR or near IR spectrum can be upconverted into light having a wavelength of about 450 nm to about 550 nm. The light can have wavelengths corresponding to red, yellow, amber, orange, green, or blue light. In some embodiments, the individual is a human or a non-human animal. In other embodiments, the neural cell is in the peripheral nervous system. In another embodiment, the neural cell is in the central nervous system.
[0120] Kits
[0121] Also provided herein are kits comprising polynucleotides encoding a light-responsive opsin protein (such as any of the light-responsive opsin proteins described herein) and lanthanide-doped nanoparticles for use in any of the methods disclosed herein to alter the membrane polarization state of one or more neurons of the central and/or peripheral nervous system. In some embodiments, the kits further comprise an infrared or near infrared electromagnetic radiation source. In other embodiments, the kits further comprise instructions for using the polynucleotides and lanthanide-doped nanoparticles described herein. In still other embodiments, the lanthanide-doped nanoparticles described herein are embedded and/or trapped in a biocompatible material (such as any of the biocompatible materials described above).
EXEMPLARY EMBODIMENTS
[0122] Aspects of the present disclosure may be more completely understood in consideration of the detailed description of various embodiments of the present disclosure that follows in connection with the accompanying drawings. This description and the various embodiments are presented as follows:
[0123] The embodiments and specific applications discussed herein may be implemented in connection with one or more of the above-described aspects, embodiments and implementations, as well as with those shown in the figures and described below. Reference may also be made to Wang et al., 2010, Nature, 463(7284):1061-5, which is fully incorporated herein by reference. For further details on light responsive molecules and/or opsins, including methodology, devices and substances, reference may also be made to the following background publications: U.S. Patent Publication No. 2010/0190229, entitled "System for Optical Stimulation of Target Cells" to Zhang et al.; U.S. Patent Publication No. 2007/0261127, entitled "System for Optical Stimulation of Target Cells" to Boyden et al. These applications form part of the provisional patent document and are fully-incorporated herein by reference. Consistent with these publications, numerous opsins can be used in mammalian cells in vivo and in vitro to provide optical stimulation and control of target cells. For example, when ChR2 is introduced into an electrically-excitable cell, such as a neuron, light activation of the ChR2 channel rhodopsin can result in excitation and/or firing of the cell. In instances when NpHR is introduced into an electrically-excitable cell, such as a neuron, light activation of the NpHR opsin can result in inhibition of firing of the cell. These and other aspects of the disclosures of the above-referenced patent applications may be useful in implementing various aspects of the present disclosure.
[0124] In various embodiments of the present disclosure, minimally invasive delivery of light, for example as can be useful for manipulation of neural circuits with optogenetics, using near infrared up-conversion nanocrystals, is achieved. This is used to avoid the implantation of light sources within living tissues, including, for example, a subject's brain. Mammalian tissue has a transparency window in near infrared part of the spectrum (700-1000 nm). Accordingly, aspects of the present disclosure relate to the use of nanoparticles for the purpose of using (near) infrared light to deliver energy into the depth of a brain by converting the infrared light into visible wavelengths at a site of interest.
[0125] In certain embodiments, delivering visible wavelengths at a site of interest within the brain is achieved through a process of optical upconversion in Lanthanide-doped nanocrystals. During upconversion 3-4 photons are absorbed by the material which then emits one photon with the energy .about.1.5-2 times the energy of absorbed photons. For example NaYF4:Yb/X/Gd nanocrystals can absorb 980 nm light and emit light with spectra centered between 450-550 nm depending on the nature and relative content of dopants (X.dbd.Er, Tm, Er/Tm). For more information regarding modifying the light emitted from the nanoparticles, see Wang et al., Nature, 2010, 463(7284):1061-5, the disclosure of which is incorporated by reference herein in its entirety.
[0126] In certain embodiments a single step surgery is performed to modify a target cell population and provide nanoparticles to convert near infrared light to visible light that stimulates the modified target cell population. During the surgery, the surgeon injects both an adeno-associated virus carrying an opsin gene and a nanoparticle solution to a site of interest.
[0127] The virus is optimized to only infect the target cell population. Similarly, the nanoparticles are functionalized with antibodies so that the nanoparticles anchor to the target cell population as well. In certain more specific embodiments the target cell population is a particular neuron type. After surgery is completed, a LED that emits near infrared light is placed on a thinned portion of the patient's skull, underneath the skin. A battery can also be implanted underneath the skin to power the LED. In certain embodiments the battery has characteristics similar to those of a pacemaker battery. A microcontroller can be used to control the battery to deliver energy to the LED at specified intervals, resulting in LED light pulses at specified intervals.
[0128] Certain aspects of the present disclosure are directed to the use of optogenetics in vivo. Optogenetics, applied in vivo, relies on light delivery to specific neuron populations that can be located deep within the brain. Mammalian tissue is highly absorptive and scatters light in the visible spectrum. However, near infrared light is able to penetrate to deep levels of the brain without excessive absorption or scattering.
[0129] Certain aspects of the present disclosure are directed to imbedding nanoparticles in the brain near target neurons. The nanoparticles can be lanthanide doped-nanoparticle. Nanoparticles doped with Lanthanides or with other dopants can be optimized with respect to a particular opsin's activation spectra. As discussed in more detail in Wang et al., Nature, 2010, 463(7284):1061-5, the disclosure of which is incorporated by reference herein in its entirety, the spectra of the light emitted from lanthanide-doped nanocrystals can be manipulated based on which dopants are used, and how much. Similarly, the light emitted from nanoparticles doped with other molecules can be manipulated based on the concentration of dopants.
[0130] The ability to provide different output spectra depending on the doping of nanoparticles allows for a non-invasive approach to acute neural manipulation. A light source, such as a LED can be mounted onto a thinned skull under the skin. Depending on the composition of nanoparticles, and the opsin delivered to the target neurons, aspects of the present disclosure can be used for neural excitation or silencing. Similarly, multiple neural populations may be controlled simultaneously through the use of various dopants and opsins in combination.
[0131] Turning to FIG. 1, a patient's head 100 is shown. A target (neural) cell population 114 includes light responsive molecules. These light responsive molecules can include, but are not necessarily limited to, opsins derived from Channel rhodopsins (e.g. ChR1 or ChR2) or Halorhodopins (NpHR). The specific molecule can be tailored/selected based upon the desired effect on the target cell population and/or the wavelength at which the molecules respond to light.
[0132] Nanocrystals 110 are introduced near or at the target cell populate. Various embodiments of the present disclosure are directed toward methods and devices for positioning and maintaining positioning of the nanocrystals near the target cell population. Certain embodiments are directed toward anchoring the nanocrystals to cells of (or near) the target cell population using antibodies.
[0133] According to other example embodiments, a structure can be introduced that includes the nanocrystals. For instance, a mesh structure can be coated with the nanocrystals. The synthetic mesh can be constructed so as to allow the dendrites and axons to pass through the mess without allowing the entire neuron (e.g., the cell body) to pass. One example of such a mesh has pores that are on the order of 3-7 microns in diameter and is made from polyethylene terephthalate. This mesh structure can be constructed with light-responsive cells/neurons contained therein and/or be placed near the target cell population, which includes the light-responsive cells. Consistent with another embodiment, one or more transparent capsules, each containing a solution of nanocrystals, can be positioned near the target cell populations.
[0134] Embodiments of the present disclosure are also directed toward various optical sources of stimulation. These sources can include, but are not limited to, external laser sources and light-emitting didoes (LEDs). Particular aspects of the present disclosure are directed toward the relatively low absorption and/or scattering/diffusion caused by intervening material when the light is at certain wavelengths (e.g., (near) infrared). Accordingly, the light source can be externally located because of the ability to penetrate the tissue with little loss of optical intensity or power. Moreover, reduced diffusion can be particularly useful for providing a relatively-high spatial-precision for the delivery of the light. Thus, embodiments of the present disclosure are directed toward multiple target cell populations with respective nanocrystals that can be individually controlled using spatially-precise optical stimulus. For instance, the nanocrystals can be implanted in several locations within the brain. The light source can then be aimed at a respective and particular location. Multiple light sources can also be used for simultaneous stimulation of a plurality of locations.
[0135] Consistent with a particular embodiment of the present disclosure, the skull 102 has a thinned portion 106. An LED 104 is located above the thinned portion of the skull and emits near infrared light 108. When the IR hits nanocrystal 110, it is absorbed. The nanocrystal emits visible light 112 in response to absorbing the IR light 108. The visible light 112 is absorbed by modified cell 114.
[0136] The system shown in FIG. 1 allows for delivery of light to a target cell deep within a patient's brain tissue. The light responsive molecule can be specifically targeted to a neural cell type of interest. Similarly, the nanocrystals 112 are anchored to the neural cell with antibodies chosen based on the type of neural cell 114 being targeted.
[0137] Turning to FIG. 2, a group of neurons is illuminated with infrared light 208 between 700-1000 nm. Target neurons 214 express an opsin gene, allowing the neurons to be activated or inhibited, depending on which opsin, and what wavelength of light is absorbed by the neurons 214. The target neurons 214 can be interspersed between other neurons 216. As shown in inset 202, target neurons 214 are coated with upconverting nanoparticles 210 that are anchored to the neural membrane via antibodies. The nanoparticles 210 absorb IR photons and emit visible photons that are then absorbed by opsins triggering neural activation.
[0138] The system of FIG. 2 can be used with a variety of target neurons 214. The opsin gene 215 expressed in the target neurons 214 is modified based on the target neuron. Similarly, the antibodies used to anchor the nanoparticles 210 to the target neuron membranes are modified to attach to a specific membrane type. As shown in inset 202, the nanoparticles 210 are closely linked to the target neurons so that visible light photons emitted by the nanoparticles 210 are absorbed by the target neurons 214.
[0139] FIG. 3 depicts a system that uses multiple light sources, consistent with an embodiment of the present disclosure. A patient has nanoparticles located at target locations 308-312. The system includes light sources 302-306, which can be configured to generate light at a frequency that is upconverted by the nanoparticles located at target locations 308-312. Although three light sources are depicted, there can be any number of light sources. These light sources can be external to the patient (e.g., a targeting system that directs several light sources using mechanical positioning), using embedded lights sources (e.g., LEDs implanted on the skull) or combinations thereof. The target locations 308-312 include cells that have optically-responsive membrane molecules. These optically-responsive membrane molecules react to light at the upconverted frequency.
[0140] Nanoparticles located at the intersection 314 of the light from the different light sources 302-306 receive increased intensity of optical stimulus relative to other locations, including those locations within the path of light from a single light source. In this manner, the light intensity of each of the light sources can be set below a threshold level. When multiple light sources are directed at the same location, the threshold intensity level can be exceeded at the location. This allows for spatial control in three-dimensions and also allows for reduced inadvertent effects on non-targeted tissue. Consistent with one embodiment, the threshold level can be set according to an amount of light necessary to cause the desired effect (e.g., excitation or inhibition) on the target cells. Consistent with other embodiments, the threshold level can be set to avoid adverse effects on non-targeted tissue (e.g., heating).
[0141] The use of multiple light sources can also bring about a step-wise increase in light intensity. For instance, a disease model could be tested by monitoring the effects of additional stimulation caused by the increase in light intensity. The use of independent light sources allows for relatively simple control over temporal and spatial increases or decreases. Consistent with other embodiments of the present disclosure, the spatial precision of the light sources can be varied between the different light sources. For example, a first light source can provide light that illuminates the entire target cell location. This allows for all cells within the population to be illuminated. A second light source can provide light having a focal point that illuminates less than all of the entire target cell location. The combination of the first and second (or more) light sources can be used to provide different levels of stimulation within the same cell population.
[0142] Embodiments of the present disclosure relate to the use of one or more light sources operating in a scanning mode. The light source(s) are aimed at specific locations within a target cell population. The effects of the stimulation can be monitored as the light source is used to scan or otherwise move within the target cell population. This can be particularly useful in connection with the three-dimensional control provided by the use of multiple light sources.
[0143] Various embodiments of the present disclosure are directed toward the use of nanocrystals that emit light at different wavelengths. This can be particularly useful when using multiple opsins having different light-absorption spectrums. The nanocrystals can be targeted toward different opsins and/or placed in the corresponding locations. While the present disclosure is amenable to various modifications and alternative forms, specifics thereof have been shown by way of example in the drawings and will be described in further detail. It should be understood that the intention is not to limit the disclosure to the particular embodiments and/or applications described. On the contrary, the intention is to cover all modifications, equivalents, and alternatives falling within the spirit and scope of the present disclosure.
EXAMPLES
Example 1: Use of Lanthanide-Doped Nanoparticles in the Use of Optogenetics to Hyperpolarize the Cholinergic Interneurons of the Nucleus Accumbens
[0144] The nucleus accumbens (NAc) is a collection of neurons that forms the main part of the ventral striatum. The NAc is thought to play an important role in the complex mammalian behaviors associated with reward, pleasure, laughter, addiction, aggression, fear, and the placebo effect. Cholinergic interneurons within the NAc constitute less than 1% of the local neural population, yet they project throughout the NAc and provide its only known cholinergic input. In this Example, an optogenetic approach using a light-responsive chloride pump protein in combination with lanthanide-doped nanoparticles is used to block action potential firing in these cells, with both high temporal resolution and high cell-type specificity. To express microbial opsin genes specifically in cholinergic interneurons, a transgenic mouse line expressing Cre recombinase is employed under the choline acetyltransferase (ChAT) promoter. A Cre-inducible adeno-associated virus (AAV) vector carrying a yellow-light gated third-generation chloride pump halorhodopsin (eNpHR3.0) gene fused in-frame with coding sequence for enhanced yellow fluorescent protein (eYFP) is stereotactically injected.
[0145] Specifically, mice are anesthetized and then placed in a stereotactic head apparatus. Surgeries are performed on 4-6 week old mice and ophthalmic ointment is applied throughout to prevent the eyes from drying. A midline scalp incision is made followed by a craniotomy, and then AAV vector is injected with a 10 .mu.l syringe and a 34 gauge metal needle. The injection volume and flow rate (1 .mu.l at 0.15 .mu.l/min) are controlled by an injection pump. Each NAc receives two injections (injection 1: AP 1.15 mm, ML 0.8 mm, DV -4.8 mm; injection 2: AP 1.15 mm, ML 0.8 mm, DV -4.2 mm). The virus injection and fiber position are chosen so that virtually the entire shell is stimulated.
[0146] Next, before withdrawing the needle, NaYF.sub.4:Yb/Er/Gd, nanoparticles are injected into the Nac. Concentrations of 3.4, 8.5, or 17 nmoles of NaYF4:Yb/Er/Gd, nanoparticles are used. After injection of both the AAV vector and the lanthanide-doped nanoparticles is complete, the needle is left in place for 5 additional minutes and then very slowly withdrawn.
[0147] Following a recovery period, the mice are again anesthetized, the skulls of the mice are thinned and an NIR source of electromagnetic radiation is placed adjacent to the thinned skull-region. Simultaneous NW stimulation and extracellular electrical recording are performed based on methods described previously using optical stimulation (Gradinaru et al., J. Neurosci., 27, 14231-14238 (2007)). The electrode consists of a tungsten electrode (1 M.OMEGA.; 0.005 in; parylene insulation) with the tip of the electrode projecting beyond the fiber by 300-500 .mu.m. The electrode is lowered through the NAc in approximately 100 .mu.m increments, and NIR-upconverted optical responses are recorded at each increment. Signals are amplified and band-pass filtered (300 Hz low cut-off, 10 kHz high cut-off) before digitizing and recording to disk. At each site, 5 stimulation repetitions are presented and saved.
[0148] The examples, which are intended to be purely exemplary of the invention and should therefore not be considered to limit the invention in any way, also describe and detail aspects and embodiments of the invention discussed above. The foregoing examples and detailed description are offered by way of illustration and not by way of limitation. All publications, patent applications, and patents cited in this specification are herein incorporated by reference as if each individual publication, patent application, or patent were specifically and individually indicated to be incorporated by reference. In particular, all publications cited herein are expressly incorporated herein by reference for the purpose of describing and disclosing compositions and methodologies which might be used in connection with the invention. Although the foregoing invention has been described in some detail by way of illustration and example for purposes of clarity of understanding, it will be readily apparent to those of ordinary skill in the art in light of the teachings of this invention that certain changes and modifications may be made thereto without departing from the spirit or scope of the appended claims.
Sequence CWU 1
1
151273PRTNatronomonas pharaonis 1Val Thr Gln Arg Glu Leu Phe Glu
Phe Val Leu Asn Asp Pro Leu Leu1 5 10 15Ala Ser Ser Leu Tyr Ile Asn
Ile Ala Leu Ala Gly Leu Ser Ile Leu 20 25 30Leu Phe Val Phe Met Thr
Arg Gly Leu Asp Asp Pro Arg Ala Lys Leu 35 40 45Ile Ala Val Ser Thr
Ile Leu Val Pro Val Val Ser Ile Ala Ser Tyr 50 55 60Thr Gly Leu Ala
Ser Gly Leu Thr Ile Ser Val Leu Glu Met Pro Ala65 70 75 80Gly His
Phe Ala Glu Gly Ser Ser Val Met Leu Gly Gly Glu Glu Val 85 90 95Asp
Gly Val Val Thr Met Trp Gly Arg Tyr Leu Thr Trp Ala Leu Ser 100 105
110Thr Pro Met Ile Leu Leu Ala Leu Gly Leu Leu Ala Gly Ser Asn Ala
115 120 125Thr Lys Leu Phe Thr Ala Ile Thr Phe Asp Ile Ala Met Cys
Val Thr 130 135 140Gly Leu Ala Ala Ala Leu Thr Thr Ser Ser His Leu
Met Arg Trp Phe145 150 155 160Trp Tyr Ala Ile Ser Cys Ala Cys Phe
Leu Val Val Leu Tyr Ile Leu 165 170 175Leu Val Glu Trp Ala Gln Asp
Ala Lys Ala Ala Gly Thr Ala Asp Met 180 185 190Phe Asn Thr Leu Lys
Leu Leu Thr Val Val Met Trp Leu Gly Tyr Pro 195 200 205Ile Val Trp
Ala Leu Gly Val Glu Gly Ile Ala Val Leu Pro Val Gly 210 215 220Val
Thr Ser Trp Gly Tyr Ser Phe Leu Asp Ile Val Ala Lys Tyr Ile225 230
235 240Phe Ala Phe Leu Leu Leu Asn Tyr Leu Thr Ser Asn Glu Ser Val
Val 245 250 255Ser Gly Ser Ile Leu Asp Val Pro Ser Ala Ser Gly Thr
Pro Ala Asp 260 265 270Asp2559PRTArtificial SequenceSynthetic
polypeptide 2Met Thr Glu Thr Leu Pro Pro Val Thr Glu Ser Ala Val
Ala Leu Gln1 5 10 15Ala Glu Val Thr Gln Arg Glu Leu Phe Glu Phe Val
Leu Asn Asp Pro 20 25 30Leu Leu Ala Ser Ser Leu Tyr Ile Asn Ile Ala
Leu Ala Gly Leu Ser 35 40 45Ile Leu Leu Phe Val Phe Met Thr Arg Gly
Leu Asp Asp Pro Arg Ala 50 55 60Lys Leu Ile Ala Val Ser Thr Ile Leu
Val Pro Val Val Ser Ile Ala65 70 75 80Ser Tyr Thr Gly Leu Ala Ser
Gly Leu Thr Ile Ser Val Leu Glu Met 85 90 95Pro Ala Gly His Phe Ala
Glu Gly Ser Ser Val Met Leu Gly Gly Glu 100 105 110Glu Val Asp Gly
Val Val Thr Met Trp Gly Arg Tyr Leu Thr Trp Ala 115 120 125Leu Ser
Thr Pro Met Ile Leu Leu Ala Leu Gly Leu Leu Ala Gly Ser 130 135
140Asn Ala Thr Lys Leu Phe Thr Ala Ile Thr Phe Asp Ile Ala Met
Cys145 150 155 160Val Thr Gly Leu Ala Ala Ala Leu Thr Thr Ser Ser
His Leu Met Arg 165 170 175Trp Phe Trp Tyr Ala Ile Ser Cys Ala Cys
Phe Leu Val Val Leu Tyr 180 185 190Ile Leu Leu Val Glu Trp Ala Gln
Asp Ala Lys Ala Ala Gly Thr Ala 195 200 205Asp Met Phe Asn Thr Leu
Lys Leu Leu Thr Val Val Met Trp Leu Gly 210 215 220Tyr Pro Ile Val
Trp Ala Leu Gly Val Glu Gly Ile Ala Val Leu Pro225 230 235 240Val
Gly Val Thr Ser Trp Gly Tyr Ser Phe Leu Asp Ile Val Ala Lys 245 250
255Tyr Ile Phe Ala Phe Leu Leu Leu Asn Tyr Leu Thr Ser Asn Glu Ser
260 265 270Val Val Ser Gly Ser Ile Leu Asp Val Pro Ser Ala Ser Gly
Thr Pro 275 280 285Ala Asp Asp Ala Ala Ala Lys Ser Arg Ile Thr Ser
Glu Gly Glu Tyr 290 295 300Ile Pro Leu Asp Gln Ile Asp Ile Asn Val
Val Ser Lys Gly Glu Glu305 310 315 320Leu Phe Thr Gly Val Val Pro
Ile Leu Val Glu Leu Asp Gly Asp Val 325 330 335Asn Gly His Lys Phe
Ser Val Ser Gly Glu Gly Glu Gly Asp Ala Thr 340 345 350Tyr Gly Lys
Leu Thr Leu Lys Phe Ile Cys Thr Thr Gly Lys Leu Pro 355 360 365Val
Pro Trp Pro Thr Leu Val Thr Thr Phe Gly Tyr Gly Leu Gln Cys 370 375
380Phe Ala Arg Tyr Pro Asp His Met Lys Gln His Asp Phe Phe Lys
Ser385 390 395 400Ala Met Pro Glu Gly Tyr Val Gln Glu Arg Thr Ile
Phe Phe Lys Asp 405 410 415Asp Gly Asn Tyr Lys Thr Arg Ala Glu Val
Lys Phe Glu Gly Asp Thr 420 425 430Leu Val Asn Arg Ile Glu Leu Lys
Gly Ile Asp Phe Lys Glu Asp Gly 435 440 445Asn Ile Leu Gly His Lys
Leu Glu Tyr Asn Tyr Asn Ser His Asn Val 450 455 460Tyr Ile Met Ala
Asp Lys Gln Lys Asn Gly Ile Lys Val Asn Phe Lys465 470 475 480Ile
Arg His Asn Ile Glu Asp Gly Ser Val Gln Leu Ala Asp His Tyr 485 490
495Gln Gln Asn Thr Pro Ile Gly Asp Gly Pro Val Leu Leu Pro Asp Asn
500 505 510His Tyr Leu Ser Tyr Gln Ser Ala Leu Ser Lys Asp Pro Asn
Glu Lys 515 520 525Arg Asp His Met Val Leu Leu Glu Phe Val Thr Ala
Ala Gly Ile Thr 530 535 540Leu Gly Met Asp Glu Leu Tyr Lys Phe Cys
Tyr Glu Asn Glu Val545 550 5553542PRTArtificial SequenceSynthetic
polypeptide 3Met Val Thr Gln Arg Glu Leu Phe Glu Phe Val Leu Asn
Asp Pro Leu1 5 10 15Leu Ala Ser Ser Leu Tyr Ile Asn Ile Ala Leu Ala
Gly Leu Ser Ile 20 25 30Leu Leu Phe Val Phe Met Thr Arg Gly Leu Asp
Asp Pro Arg Ala Lys 35 40 45Leu Ile Ala Val Ser Thr Ile Leu Val Pro
Val Val Ser Ile Ala Ser 50 55 60Tyr Thr Gly Leu Ala Ser Gly Leu Thr
Ile Ser Val Leu Glu Met Pro65 70 75 80Ala Gly His Phe Ala Glu Gly
Ser Ser Val Met Leu Gly Gly Glu Glu 85 90 95Val Asp Gly Val Val Thr
Met Trp Gly Arg Tyr Leu Thr Trp Ala Leu 100 105 110Ser Thr Pro Met
Ile Leu Leu Ala Leu Gly Leu Leu Ala Gly Ser Asn 115 120 125Ala Thr
Lys Leu Phe Thr Ala Ile Thr Phe Asp Ile Ala Met Cys Val 130 135
140Thr Gly Leu Ala Ala Ala Leu Thr Thr Ser Ser His Leu Met Arg
Trp145 150 155 160Phe Trp Tyr Ala Ile Ser Cys Ala Cys Phe Leu Val
Val Leu Tyr Ile 165 170 175Leu Leu Val Glu Trp Ala Gln Asp Ala Lys
Ala Ala Gly Thr Ala Asp 180 185 190Met Phe Asn Thr Leu Lys Leu Leu
Thr Val Val Met Trp Leu Gly Tyr 195 200 205Pro Ile Val Trp Ala Leu
Gly Val Glu Gly Ile Ala Val Leu Pro Val 210 215 220Gly Val Thr Ser
Trp Gly Tyr Ser Phe Leu Asp Ile Val Ala Lys Tyr225 230 235 240Ile
Phe Ala Phe Leu Leu Leu Asn Tyr Leu Thr Ser Asn Glu Ser Val 245 250
255Val Ser Gly Ser Ile Leu Asp Val Pro Ser Ala Ser Gly Thr Pro Ala
260 265 270Asp Asp Ala Ala Ala Lys Ser Arg Ile Thr Ser Glu Gly Glu
Tyr Ile 275 280 285Pro Leu Asp Gln Ile Asp Ile Asn Val Val Ser Lys
Gly Glu Glu Leu 290 295 300Phe Thr Gly Val Val Pro Ile Leu Val Glu
Leu Asp Gly Asp Val Asn305 310 315 320Gly His Lys Phe Ser Val Ser
Gly Glu Gly Glu Gly Asp Ala Thr Tyr 325 330 335Gly Lys Leu Thr Leu
Lys Phe Ile Cys Thr Thr Gly Lys Leu Pro Val 340 345 350Pro Trp Pro
Thr Leu Val Thr Thr Phe Gly Tyr Gly Leu Gln Cys Phe 355 360 365Ala
Arg Tyr Pro Asp His Met Lys Gln His Asp Phe Phe Lys Ser Ala 370 375
380Met Pro Glu Gly Tyr Val Gln Glu Arg Thr Ile Phe Phe Lys Asp
Asp385 390 395 400Gly Asn Tyr Lys Thr Arg Ala Glu Val Lys Phe Glu
Gly Asp Thr Leu 405 410 415Val Asn Arg Ile Glu Leu Lys Gly Ile Asp
Phe Lys Glu Asp Gly Asn 420 425 430Ile Leu Gly His Lys Leu Glu Tyr
Asn Tyr Asn Ser His Asn Val Tyr 435 440 445Ile Met Ala Asp Lys Gln
Lys Asn Gly Ile Lys Val Asn Phe Lys Ile 450 455 460Arg His Asn Ile
Glu Asp Gly Ser Val Gln Leu Ala Asp His Tyr Gln465 470 475 480Gln
Asn Thr Pro Ile Gly Asp Gly Pro Val Leu Leu Pro Asp Asn His 485 490
495Tyr Leu Ser Tyr Gln Ser Ala Leu Ser Lys Asp Pro Asn Glu Lys Arg
500 505 510Asp His Met Val Leu Leu Glu Phe Val Thr Ala Ala Gly Ile
Thr Leu 515 520 525Gly Met Asp Glu Leu Tyr Lys Phe Cys Tyr Glu Asn
Glu Val 530 535 5404223PRTGuillardia theta 4Ala Ser Ser Phe Gly Lys
Ala Leu Leu Glu Phe Val Phe Ile Val Phe1 5 10 15Ala Cys Ile Thr Leu
Leu Leu Gly Ile Asn Ala Ala Lys Ser Lys Ala 20 25 30Ala Ser Arg Val
Leu Phe Pro Ala Thr Phe Val Thr Gly Ile Ala Ser 35 40 45Ile Ala Tyr
Phe Ser Met Ala Ser Gly Gly Gly Trp Val Ile Ala Pro 50 55 60Asp Cys
Arg Gln Leu Phe Val Ala Arg Tyr Leu Asp Trp Leu Ile Thr65 70 75
80Thr Pro Leu Leu Leu Ile Asp Leu Gly Leu Val Ala Gly Val Ser Arg
85 90 95Trp Asp Ile Met Ala Leu Cys Leu Ser Asp Val Leu Met Ile Ala
Thr 100 105 110Gly Ala Phe Gly Ser Leu Thr Val Gly Asn Val Lys Trp
Val Trp Trp 115 120 125Phe Phe Gly Met Cys Trp Phe Leu His Ile Ile
Phe Ala Leu Gly Lys 130 135 140Ser Trp Ala Glu Ala Ala Lys Ala Lys
Gly Gly Asp Ser Ala Ser Val145 150 155 160Tyr Ser Lys Ile Ala Gly
Ile Thr Val Ile Thr Trp Phe Cys Tyr Pro 165 170 175Val Val Trp Val
Phe Ala Glu Gly Phe Gly Asn Phe Ser Val Thr Phe 180 185 190Glu Val
Leu Ile Tyr Gly Val Leu Asp Val Ile Ser Lys Ala Val Phe 195 200
205Gly Leu Ile Leu Met Ser Gly Ala Ala Thr Gly Tyr Glu Ser Ile 210
215 2205310PRTChlamydomonas reinhardtii 5Met Asp Tyr Gly Gly Ala
Leu Ser Ala Val Gly Arg Glu Leu Leu Phe1 5 10 15Val Thr Asn Pro Val
Val Val Asn Gly Ser Val Leu Val Pro Glu Asp 20 25 30Gln Cys Tyr Cys
Ala Gly Trp Ile Glu Ser Arg Gly Thr Asn Gly Ala 35 40 45Gln Thr Ala
Ser Asn Val Leu Gln Trp Leu Ala Ala Gly Phe Ser Ile 50 55 60Leu Leu
Leu Met Phe Tyr Ala Tyr Gln Thr Trp Lys Ser Thr Cys Gly65 70 75
80Trp Glu Glu Ile Tyr Val Cys Ala Ile Glu Met Val Lys Val Ile Leu
85 90 95Glu Phe Phe Phe Glu Phe Lys Asn Pro Ser Met Leu Tyr Leu Ala
Thr 100 105 110Gly His Arg Val Gln Trp Leu Arg Tyr Ala Glu Trp Leu
Leu Thr Cys 115 120 125Pro Val Ile Leu Ile His Leu Ser Asn Leu Thr
Gly Leu Ser Asn Asp 130 135 140Tyr Ser Arg Arg Thr Met Gly Leu Leu
Val Ser Asp Ile Gly Thr Ile145 150 155 160Val Trp Gly Ala Thr Ser
Ala Met Ala Thr Gly Tyr Val Lys Val Ile 165 170 175Phe Phe Cys Leu
Gly Leu Cys Tyr Gly Ala Asn Thr Phe Phe His Ala 180 185 190Ala Lys
Ala Tyr Ile Glu Gly Tyr His Thr Val Pro Lys Gly Arg Cys 195 200
205Arg Gln Val Val Thr Gly Met Ala Trp Leu Phe Phe Val Ser Trp Gly
210 215 220Met Phe Pro Ile Leu Phe Ile Leu Gly Pro Glu Gly Phe Gly
Val Leu225 230 235 240Ser Val Tyr Gly Ser Thr Val Gly His Thr Ile
Ile Asp Leu Met Ser 245 250 255Lys Asn Cys Trp Gly Leu Leu Gly His
Tyr Leu Arg Val Leu Ile His 260 265 270Glu His Ile Leu Ile His Gly
Asp Ile Arg Lys Thr Thr Lys Leu Asn 275 280 285Ile Gly Gly Thr Glu
Ile Glu Val Glu Thr Leu Val Glu Asp Glu Ala 290 295 300Glu Ala Gly
Ala Val Pro305 3106310PRTArtificial SequenceSynthetic polypeptide
6Met Asp Tyr Gly Gly Ala Leu Ser Ala Val Gly Arg Glu Leu Leu Phe1 5
10 15Val Thr Asn Pro Val Val Val Asn Gly Ser Val Leu Val Pro Glu
Asp 20 25 30Gln Cys Tyr Cys Ala Gly Trp Ile Glu Ser Arg Gly Thr Asn
Gly Ala 35 40 45Gln Thr Ala Ser Asn Val Leu Gln Trp Leu Ala Ala Gly
Phe Ser Ile 50 55 60Leu Leu Leu Met Phe Tyr Ala Tyr Gln Thr Trp Lys
Ser Thr Cys Gly65 70 75 80Trp Glu Glu Ile Tyr Val Cys Ala Ile Glu
Met Val Lys Val Ile Leu 85 90 95Glu Phe Phe Phe Glu Phe Lys Asn Pro
Ser Met Leu Tyr Leu Ala Thr 100 105 110Gly His Arg Val Gln Trp Leu
Arg Tyr Ala Glu Trp Leu Leu Thr Ser 115 120 125Pro Val Ile Leu Ile
His Leu Ser Asn Leu Thr Gly Leu Ser Asn Asp 130 135 140Tyr Ser Arg
Arg Thr Met Gly Leu Leu Val Ser Asp Ile Gly Thr Ile145 150 155
160Val Trp Gly Ala Thr Ser Ala Met Ala Thr Gly Tyr Val Lys Val Ile
165 170 175Phe Phe Cys Leu Gly Leu Cys Tyr Gly Ala Asn Thr Phe Phe
His Ala 180 185 190Ala Lys Ala Tyr Ile Glu Gly Tyr His Thr Val Pro
Lys Gly Arg Cys 195 200 205Arg Gln Val Val Thr Gly Met Ala Trp Leu
Phe Phe Val Ser Trp Gly 210 215 220Met Phe Pro Ile Leu Phe Ile Leu
Gly Pro Glu Gly Phe Gly Val Leu225 230 235 240Ser Val Tyr Gly Ser
Thr Val Gly His Thr Ile Ile Asp Leu Met Ser 245 250 255Lys Asn Cys
Trp Gly Leu Leu Gly His Tyr Leu Arg Val Leu Ile His 260 265 270Glu
His Ile Leu Ile His Gly Asp Ile Arg Lys Thr Thr Lys Leu Asn 275 280
285Ile Gly Gly Thr Glu Ile Glu Val Glu Thr Leu Val Glu Asp Glu Ala
290 295 300Glu Ala Gly Ala Val Pro305 3107310PRTArtificial
SequenceSynthetic polypeptide 7Met Asp Tyr Gly Gly Ala Leu Ser Ala
Val Gly Arg Glu Leu Leu Phe1 5 10 15Val Thr Asn Pro Val Val Val Asn
Gly Ser Val Leu Val Pro Glu Asp 20 25 30Gln Cys Tyr Cys Ala Gly Trp
Ile Glu Ser Arg Gly Thr Asn Gly Ala 35 40 45Gln Thr Ala Ser Asn Val
Leu Gln Trp Leu Ala Ala Gly Phe Ser Ile 50 55 60Leu Leu Leu Met Phe
Tyr Ala Tyr Gln Thr Trp Lys Ser Thr Cys Gly65 70 75 80Trp Glu Glu
Ile Tyr Val Cys Ala Ile Glu Met Val Lys Val Ile Leu 85 90 95Glu Phe
Phe Phe Glu Phe Lys Asn Pro Ser Met Leu Tyr Leu Ala Thr 100 105
110Gly His Arg Val Gln Trp Leu Arg Tyr Ala Glu Trp Leu Leu Thr Ser
115 120 125Pro Val Ile Leu Ile His Leu Ser Asn Leu Thr Gly Leu Ser
Asn Asp 130 135 140Tyr Ser Arg Arg Thr Met Gly Leu Leu Val Ser Ala
Ile Gly Thr Ile145 150 155 160Val Trp Gly Ala Thr Ser Ala Met Ala
Thr Gly Tyr Val Lys Val Ile 165 170 175Phe Phe Cys Leu Gly Leu Cys
Tyr Gly Ala Asn Thr Phe Phe His Ala 180 185 190Ala Lys Ala Tyr Ile
Glu Gly Tyr His Thr Val Pro Lys Gly Arg Cys 195 200 205Arg Gln Val
Val Thr Gly Met Ala Trp Leu Phe Phe Val Ser Trp Gly 210 215 220Met
Phe Pro Ile Leu Phe Ile Leu Gly Pro Glu Gly
Phe Gly Val Leu225 230 235 240Ser Val Tyr Gly Ser Thr Val Gly His
Thr Ile Ile Asp Leu Met Ser 245 250 255Lys Asn Cys Trp Gly Leu Leu
Gly His Tyr Leu Arg Val Leu Ile His 260 265 270Glu His Ile Leu Ile
His Gly Asp Ile Arg Lys Thr Thr Lys Leu Asn 275 280 285Ile Gly Gly
Thr Glu Ile Glu Val Glu Thr Leu Val Glu Asp Glu Ala 290 295 300Glu
Ala Gly Ala Val Pro305 3108344PRTArtificial SequenceSynthetic
polypeptide 8Met Ser Arg Arg Pro Trp Leu Leu Ala Leu Ala Leu Ala
Val Ala Leu1 5 10 15Ala Ala Gly Ser Ala Gly Ala Ser Thr Gly Ser Asp
Ala Thr Val Pro 20 25 30Val Ala Thr Gln Asp Gly Pro Asp Tyr Val Phe
His Arg Ala His Glu 35 40 45Arg Met Leu Phe Gln Thr Ser Tyr Thr Leu
Glu Asn Asn Gly Ser Val 50 55 60Ile Cys Ile Pro Asn Asn Gly Gln Cys
Phe Cys Leu Ala Trp Leu Lys65 70 75 80Ser Asn Gly Thr Asn Ala Glu
Lys Leu Ala Ala Asn Ile Leu Gln Trp 85 90 95Ile Thr Phe Ala Leu Ser
Ala Leu Cys Leu Met Phe Tyr Gly Tyr Gln 100 105 110Thr Trp Lys Ser
Thr Cys Gly Trp Glu Glu Ile Tyr Val Ala Thr Ile 115 120 125Glu Met
Ile Lys Phe Ile Ile Glu Tyr Phe His Glu Phe Asp Glu Pro 130 135
140Ala Val Ile Tyr Ser Ser Asn Gly Asn Lys Thr Val Trp Leu Arg
Tyr145 150 155 160Ala Glu Trp Leu Leu Thr Cys Pro Val Leu Leu Ile
His Leu Ser Asn 165 170 175Leu Thr Gly Leu Lys Asp Asp Tyr Ser Lys
Arg Thr Met Gly Leu Leu 180 185 190Val Ser Asp Val Gly Cys Ile Val
Trp Gly Ala Thr Ser Ala Met Cys 195 200 205Thr Gly Trp Thr Lys Ile
Leu Phe Phe Leu Ile Ser Leu Ser Tyr Gly 210 215 220Met Tyr Thr Tyr
Phe His Ala Ala Lys Val Tyr Ile Glu Ala Phe His225 230 235 240Thr
Val Pro Lys Gly Ile Cys Arg Glu Leu Val Arg Val Met Ala Trp 245 250
255Thr Phe Phe Val Ala Trp Gly Met Phe Pro Val Leu Phe Leu Leu Gly
260 265 270Thr Glu Gly Phe Gly His Ile Ser Pro Tyr Gly Ser Ala Ile
Gly His 275 280 285Ser Ile Leu Asp Leu Ile Ala Lys Asn Met Trp Gly
Val Leu Gly Asn 290 295 300Tyr Leu Arg Val Lys Ile His Glu His Ile
Leu Leu Tyr Gly Asp Ile305 310 315 320Arg Lys Lys Gln Lys Ile Thr
Ile Ala Gly Gln Glu Met Glu Val Glu 325 330 335Thr Leu Val Ala Glu
Glu Glu Asp 3409344PRTArtificial SequenceSynthetic polypeptide 9Met
Ser Arg Arg Pro Trp Leu Leu Ala Leu Ala Leu Ala Val Ala Leu1 5 10
15Ala Ala Gly Ser Ala Gly Ala Ser Thr Gly Ser Asp Ala Thr Val Pro
20 25 30Val Ala Thr Gln Asp Gly Pro Asp Tyr Val Phe His Arg Ala His
Glu 35 40 45Arg Met Leu Phe Gln Thr Ser Tyr Thr Leu Glu Asn Asn Gly
Ser Val 50 55 60Ile Cys Ile Pro Asn Asn Gly Gln Cys Phe Cys Leu Ala
Trp Leu Lys65 70 75 80Ser Asn Gly Thr Asn Ala Glu Lys Leu Ala Ala
Asn Ile Leu Gln Trp 85 90 95Ile Thr Phe Ala Leu Ser Ala Leu Cys Leu
Met Phe Tyr Gly Tyr Gln 100 105 110Thr Trp Lys Ser Thr Cys Gly Trp
Glu Thr Ile Tyr Val Ala Thr Ile 115 120 125Glu Met Ile Lys Phe Ile
Ile Glu Tyr Phe His Glu Phe Asp Glu Pro 130 135 140Ala Val Ile Tyr
Ser Ser Asn Gly Asn Lys Thr Val Trp Leu Arg Tyr145 150 155 160Ala
Glu Trp Leu Leu Thr Cys Pro Val Leu Leu Ile His Leu Ser Asn 165 170
175Leu Thr Gly Leu Lys Asp Asp Tyr Ser Lys Arg Thr Met Gly Leu Leu
180 185 190Val Ser Asp Val Gly Cys Ile Val Trp Gly Ala Thr Ser Ala
Met Cys 195 200 205Thr Gly Trp Thr Lys Ile Leu Phe Phe Leu Ile Ser
Leu Ser Tyr Gly 210 215 220Met Tyr Thr Tyr Phe His Ala Ala Lys Val
Tyr Ile Glu Ala Phe His225 230 235 240Thr Val Pro Lys Gly Ile Cys
Arg Glu Leu Val Arg Val Met Ala Trp 245 250 255Thr Phe Phe Val Ala
Trp Gly Met Phe Pro Val Leu Phe Leu Leu Gly 260 265 270Thr Glu Gly
Phe Gly His Ile Ser Pro Tyr Gly Ser Ala Ile Gly His 275 280 285Ser
Ile Leu Asp Leu Ile Ala Lys Asn Met Trp Gly Val Leu Gly Asn 290 295
300Tyr Leu Arg Val Lys Ile His Glu His Ile Leu Leu Tyr Gly Asp
Ile305 310 315 320Arg Lys Lys Gln Lys Ile Thr Ile Ala Gly Gln Glu
Met Glu Val Glu 325 330 335Thr Leu Val Ala Glu Glu Glu Asp
34010344PRTArtificial SequenceSynthetic polypeptide 10Met Ser Arg
Arg Pro Trp Leu Leu Ala Leu Ala Leu Ala Val Ala Leu1 5 10 15Ala Ala
Gly Ser Ala Gly Ala Ser Thr Gly Ser Asp Ala Thr Val Pro 20 25 30Val
Ala Thr Gln Asp Gly Pro Asp Tyr Val Phe His Arg Ala His Glu 35 40
45Arg Met Leu Phe Gln Thr Ser Tyr Thr Leu Glu Asn Asn Gly Ser Val
50 55 60Ile Cys Ile Pro Asn Asn Gly Gln Cys Phe Cys Leu Ala Trp Leu
Lys65 70 75 80Ser Asn Gly Thr Asn Ala Glu Lys Leu Ala Ala Asn Ile
Leu Gln Trp 85 90 95Ile Thr Phe Ala Leu Ser Ala Leu Cys Leu Met Phe
Tyr Gly Tyr Gln 100 105 110Thr Trp Lys Ser Thr Cys Gly Trp Glu Glu
Ile Tyr Val Ala Thr Ile 115 120 125Glu Met Ile Lys Phe Ile Ile Glu
Tyr Phe His Glu Phe Asp Glu Pro 130 135 140Ala Val Ile Tyr Ser Ser
Asn Gly Asn Lys Thr Val Trp Leu Arg Tyr145 150 155 160Ala Thr Trp
Leu Leu Thr Cys Pro Val Leu Leu Ile His Leu Ser Asn 165 170 175Leu
Thr Gly Leu Lys Asp Asp Tyr Ser Lys Arg Thr Met Gly Leu Leu 180 185
190Val Ser Asp Val Gly Cys Ile Val Trp Gly Ala Thr Ser Ala Met Cys
195 200 205Thr Gly Trp Thr Lys Ile Leu Phe Phe Leu Ile Ser Leu Ser
Tyr Gly 210 215 220Met Tyr Thr Tyr Phe His Ala Ala Lys Val Tyr Ile
Glu Ala Phe His225 230 235 240Thr Val Pro Lys Gly Ile Cys Arg Glu
Leu Val Arg Val Met Ala Trp 245 250 255Thr Phe Phe Val Ala Trp Gly
Met Phe Pro Val Leu Phe Leu Leu Gly 260 265 270Thr Glu Gly Phe Gly
His Ile Ser Pro Tyr Gly Ser Ala Ile Gly His 275 280 285Ser Ile Leu
Asp Leu Ile Ala Lys Asn Met Trp Gly Val Leu Gly Asn 290 295 300Tyr
Leu Arg Val Lys Ile His Glu His Ile Leu Leu Tyr Gly Asp Ile305 310
315 320Arg Lys Lys Gln Lys Ile Thr Ile Ala Gly Gln Glu Met Glu Val
Glu 325 330 335Thr Leu Val Ala Glu Glu Glu Asp
34011344PRTArtificial SequenceSynthetic polypeptide 11Met Ser Arg
Arg Pro Trp Leu Leu Ala Leu Ala Leu Ala Val Ala Leu1 5 10 15Ala Ala
Gly Ser Ala Gly Ala Ser Thr Gly Ser Asp Ala Thr Val Pro 20 25 30Val
Ala Thr Gln Asp Gly Pro Asp Tyr Val Phe His Arg Ala His Glu 35 40
45Arg Met Leu Phe Gln Thr Ser Tyr Thr Leu Glu Asn Asn Gly Ser Val
50 55 60Ile Cys Ile Pro Asn Asn Gly Gln Cys Phe Cys Leu Ala Trp Leu
Lys65 70 75 80Ser Asn Gly Thr Asn Ala Glu Lys Leu Ala Ala Asn Ile
Leu Gln Trp 85 90 95Ile Thr Phe Ala Leu Ser Ala Leu Cys Leu Met Phe
Tyr Gly Tyr Gln 100 105 110Thr Trp Lys Ser Thr Cys Gly Trp Glu Thr
Ile Tyr Val Ala Thr Ile 115 120 125Glu Met Ile Lys Phe Ile Ile Glu
Tyr Phe His Glu Phe Asp Glu Pro 130 135 140Ala Val Ile Tyr Ser Ser
Asn Gly Asn Lys Thr Val Trp Leu Arg Tyr145 150 155 160Ala Thr Trp
Leu Leu Thr Cys Pro Val Leu Leu Ile His Leu Ser Asn 165 170 175Leu
Thr Gly Leu Lys Asp Asp Tyr Ser Lys Arg Thr Met Gly Leu Leu 180 185
190Val Ser Asp Val Gly Cys Ile Val Trp Gly Ala Thr Ser Ala Met Cys
195 200 205Thr Gly Trp Thr Lys Ile Leu Phe Phe Leu Ile Ser Leu Ser
Tyr Gly 210 215 220Met Tyr Thr Tyr Phe His Ala Ala Lys Val Tyr Ile
Glu Ala Phe His225 230 235 240Thr Val Pro Lys Gly Ile Cys Arg Glu
Leu Val Arg Val Met Ala Trp 245 250 255Thr Phe Phe Val Ala Trp Gly
Met Phe Pro Val Leu Phe Leu Leu Gly 260 265 270Thr Glu Gly Phe Gly
His Ile Ser Pro Tyr Gly Ser Ala Ile Gly His 275 280 285Ser Ile Leu
Asp Leu Ile Ala Lys Asn Met Trp Gly Val Leu Gly Asn 290 295 300Tyr
Leu Arg Val Lys Ile His Glu His Ile Leu Leu Tyr Gly Asp Ile305 310
315 320Arg Lys Lys Gln Lys Ile Thr Ile Ala Gly Gln Glu Met Glu Val
Glu 325 330 335Thr Leu Val Ala Glu Glu Glu Asp 3401220PRTArtificial
SequenceSynthetic peptide 12Lys Ser Arg Ile Thr Ser Glu Gly Glu Tyr
Ile Pro Leu Asp Gln Ile1 5 10 15Asp Ile Asn Val 20136PRTArtificial
SequenceSynthetic peptideVARIANT(2)...(2)x = any amino acid 13Phe
Xaa Tyr Glu Asn Glu1 5147PRTArtificial SequenceSynthetic peptide
14Phe Cys Tyr Glu Asn Glu Val1 51518PRTArtificial SequenceSynthetic
peptide 15Met Thr Glu Thr Leu Pro Pro Val Thr Glu Ser Ala Val Ala
Leu Gln1 5 10 15Ala Glu
D00000

D00001

D00002

S00001
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.