Device For Vaporized Substance Dosage Metering Based On An Input Dosage
FREEMAN; Daniel ; et al.
U.S. patent application number 16/249389 was filed with the patent office on 2019-07-18 for device for vaporized substance dosage metering based on an input dosage. This patent application is currently assigned to INDOSE INC. The applicant listed for this patent is INDOSE INC. Invention is credited to Ari FREEMAN, Daniel FREEMAN.
| Application Number | 20190217027 16/249389 |
| Document ID | / |
| Family ID | 67213452 |
| Filed Date | 2019-07-18 |
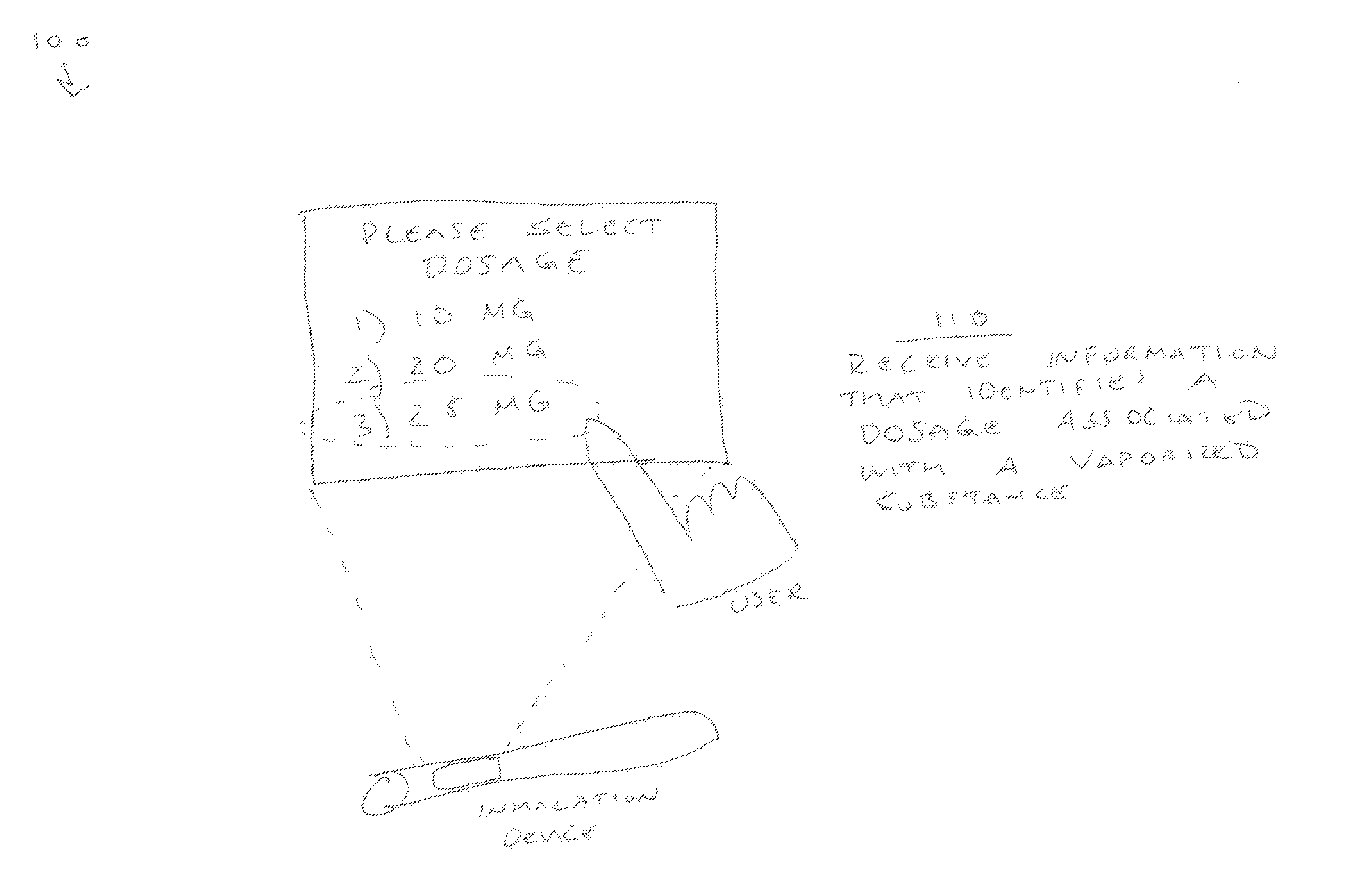
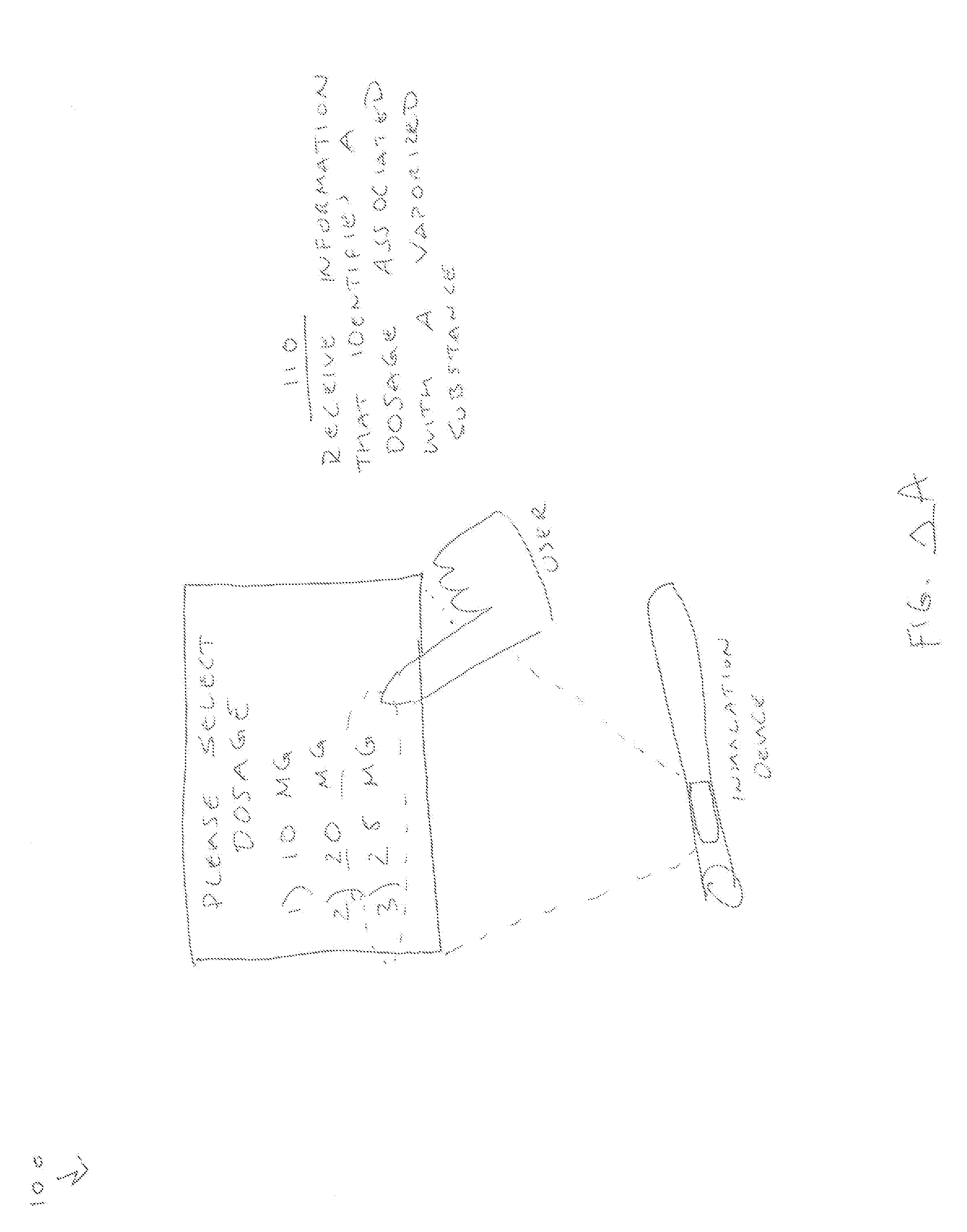




| United States Patent Application | 20190217027 |
| Kind Code | A1 |
| FREEMAN; Daniel ; et al. | July 18, 2019 |
DEVICE FOR VAPORIZED SUBSTANCE DOSAGE METERING BASED ON AN INPUT DOSAGE
Abstract
A device may receive information that identifies a dosage associated with a vaporized substance. The device may determine that the dosage associated with the vaporized substance has been generated. The device may provide information that identifies that the dosage associated with the vaporized substance has been generated.
| Inventors: | FREEMAN; Daniel; (Agoura, CA) ; FREEMAN; Ari; (Lafayette, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | INDOSE INC Woodland Hills CA |
||||||||||
| Family ID: | 67213452 | ||||||||||
| Appl. No.: | 16/249389 | ||||||||||
| Filed: | January 16, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 62621795 | Jan 25, 2018 | |||
| 62585565 | Jan 17, 2018 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A24F 7/00 20130101; A61M 2205/52 20130101; A61M 2205/3306 20130101; A61M 2205/502 20130101; A24F 47/008 20130101; A61M 15/0065 20130101; A24F 40/50 20200101; A61M 2205/3584 20130101; G01F 13/006 20130101; A61M 15/06 20130101; A61M 11/042 20140204; A61M 15/0085 20130101; G01F 1/05 20130101; A61M 2205/3334 20130101; A61M 2205/3592 20130101; A61M 2205/582 20130101; H05B 1/0297 20130101; A61M 2205/3379 20130101; A61M 2205/581 20130101; A61M 15/0001 20140204; A61M 2205/587 20130101; A61M 15/0066 20140204; A61M 15/008 20140204; A61M 2205/583 20130101 |
| International Class: | A61M 15/06 20060101 A61M015/06; A61M 15/00 20060101 A61M015/00; A24F 47/00 20060101 A24F047/00 |
Claims
1. A device, comprising: a memory configured to store one or more instructions; and one or more processors configured to execute the one or more instructions to: receive information that identifies a dosage associated with a vaporized substance; determine that the dosage associated with the vaporized substance has been generated; and provide information that identifies that the dosage associated with the vaporized substance has been generated.
2. The device of claim 1, further comprising: a vapor sensing component that is configured to detect a flow of the vaporized substance; and a flow pathway that is substantially visible to the vapor sensing component.
3. The device of claim 1, further comprising: a reservoir configured to hold a substance, and generate a vibration of a predetermined frequency to generate the vaporized substance using the substance.
4. The device of claim 1, wherein the vaporized substance is generated using at least one of a plant, a herb, or a naturally occurring ingredient.
5. The device of claim 1, wherein the one or more processors are configured to perform metering in association with dabbing.
6. The device of claim 1, wherein the one or more processors are configured to meter a substance before the device generates the vaporized substance; and wherein the one or more processors are configured to determine that the dosage associated with the vaporized substance has been generated based on metering the substance before the device generates the vaporized substance.
7. The device of claim 1, wherein the one or more processors are further configured to measure a density of a vapor in a flow pathway; and wherein the one or more processors are configured to determine that the dosage associated with the vaporized substance has been generated based on measuring the density of the vapor in the flow pathway.
8. A method, comprising: receiving, by a device, information that identifies a dosage associated with a vaporized substance; determining, by the device, that the dosage associated with the vaporized substance has been generated; and providing, by the device, information that identifies that the dosage associated with the vaporized substance has been generated.
9. The method of claim 8, further comprising: providing a user interface that permits the dosage associated with the vaporized substance to be input; and wherein receiving the information that identifies the dosage associated with the vaporized substance comprises receiving the information that identifies the dosage associated with the vaporized substance based on providing the user interface.
10. The method of claim 8, wherein the device is a user device that is configured to connect to an inhalation device, and wherein the inhalation device is configured to generate the dosage associated with the vaporized substance by vaporizing a substance.
11. The method of claim 8, wherein the information that identifies the dosage includes an input value of the dosage.
12. The method of claim 8, wherein the information that identifies the dosage includes a selection of a predetermined dosage.
13. The method of claim 8, further comprising: providing, to an inhalation device, the information that identifies the input dosage to permit the inhalation device to generate the input dosage; receiving, from the inhalation device, information that identifies that the input dosage has been generated, based on providing the information that identifies the input dosage; and wherein determining that the dosage associated with the vaporized substance has been generated comprises determining that dosage associated with the vaporized substance has been generated based on receiving the information that identifies that the input dosage has been generated.
14. The method of claim 8, further comprising: receiving, based on a user input via a user interface of the device, the information that identifies the dosage associated with the vaporized substance, and wherein the device is in communication with an inhalation device that is configured to generate the dosage associated with the vaporized substance.
15. A non-transitory computer-readable medium storing instructions, the instructions comprising: one or more instructions that, when executed by one or more processors of a device, cause the one or more processors to: receive information that identifies a dosage associated with a vaporized substance; determine that the dosage associated with the vaporized substance has been generated; and provide information that identifies that the dosage associated with the vaporized substance has been generated.
16. The non-transitory computer-readable medium of claim 15, wherein the information that identifies the dosage includes an input value of the dosage.
17. The non-transitory computer-readable medium of claim 15, wherein the information that identifies the dosage includes a selection of a predetermined dosage.
18. The non-transitory computer-readable medium of claim 15, wherein the device is an inhalation device configured to generate the vaporized substance.
19. The non-transitory computer-readable medium of claim 15, wherein the information that identifies the dosage is received from a user device.
20. The non-transitory computer-readable medium of claim 15, wherein the device is a vaporizer including a user interface configured to receive the information that identifies the input dosage.
Description
CROSS-REFERENCE TO RELATED APPLICATION
[0001] This application claims priority under 35 U.S.C. .sctn. 119 to U.S. Application Nos. 62/585,565 filed on Jan. 17, 2018, and 62/621,795, filed on Jan. 25, 2018, in the United States Patent & Trademark Office, the disclosures of which are incorporated herein by reference in their entirety.
BACKGROUND
[0002] A metered-dose inhaler (MDI) permits the delivery of a known dosage of an underlying substance for consumption by a user. For example, the underlying substance to be delivered by an MDI includes a known quantity, and, typically, a known potency. In contrast, usage of an inhalation device, such as a vaporizer, often results in the consumption of an unknown, or at least imprecise, dosage of an underlying substance. For example, the underlying substance to be delivered by the inhalation device is typically of both unknown quantity and potency. In this way, a user of an inhalation device may consume an unintended quantity of an underlying substance.
SUMMARY
[0003] According to some possible implementations, a device may receive information that identifies a dosage associated with a vaporized substance; determine that the dosage associated with the vaporized substance has been generated; and provide information that identifies that the dosage associated with the vaporized substance has been generated.
[0004] According to some possible implementations, a method includes receiving information that identifies a dosage associated with a vaporized substance; determining that the dosage associated with the vaporized substance has been generated; and providing information that identifies that the dosage associated with the vaporized substance has been generated
[0005] According to some possible implementations, a non-transitory computer-readable medium stores instructions, the instructions comprising: one or more instructions that, when executed by one or more processors of a device, cause the one or more processors to: receive information that identifies a dosage associated with a vaporized substance; determine that the dosage associated with the vaporized substance has been generated; and provide information that identifies that the dosage associated with the vaporized substance has been generated.
BRIEF DESCRIPTION OF THE DRAWINGS
[0006] FIGS. 1A and 1B are diagrams of an overview of an example implementation described herein;
[0007] FIG. 2 is a diagram of an example environment in which systems and/or methods, described herein, may be implemented;
[0008] FIG. 3 is a diagram of example components of one or more devices of FIG. 2; and
[0009] FIG. 4 is a flow chart of an example process for vaporized substance dosage metering based on an input dosage.
DETAILED DESCRIPTION
[0010] As described above, an inhalation device may provide an unintended dosage of an underlying substance for consumption by a user. Some implementations herein permit vaporized substance dosage metering based on an input dosage. In this way, some implementations herein permit the delivery, via an inhalation device, of an intended dosage of an underlying substance despite the underlying substance being of unknown quantity and/or potency.
[0011] FIGS. 1A and 1B are diagrams of an overview 100 of an embodiment described herein. As shown in FIG. 1A, and by reference number 110, an inhalation device may receive information that identifies a dosage associated with a vaporized substance. For example, as shown, a user may interact with a user interface to input a desired input dosage of 25 milligrams (mg). That is, assume that the user desires to consume 25 mg of a tetrahydrocannabinol oil that is to be vaporized by the inhalation device. In this case, the input dosage of 25 mg is the intended (desired) dosage for consumption by the user.
[0012] As shown in FIG. 1B, and by reference number 120, the inhalation device may determine that the dosage associated with the vaporized substance has been generated. For example, the inhalation device may vaporize the tetrahydrocannabinol oil, and determine that a 25 mg dosage has been generated. The inhalation device may determine that the input dosage has been generated using myriad sensor-based and/or time-based techniques, such as an optical technique, a flow measuring technique, and/or the like.
[0013] As further shown in FIG. 1B, and by reference number 130, the inhalation device may provide information that identifies that the dosage associated with the vaporized substance has been generated. For example, as shown, the inhalation device may provide, for display, information that identifies that the input dosage of 25 mg has been generated by the inhalation device (and thereby likely consumed by the user). In turn, the user may identify that the desired input dosage has been produced by the device, and cease consumption. In this way, some implementations herein permit the delivery of a known dosage of an underlying substance despite the underlying substance being of potentially both unknown quantity and potency.
[0014] FIG. 2 is a diagram of an example environment 200 in which systems and/or methods, described herein, may be implemented. As shown in FIG. 2, environment 200 may include an inhalation device 210, a user device 220, and a network 230. Devices of environment 200 may interconnect via wired connections, wireless connections, or a combination of wired and wireless connections.
[0015] Inhalation device 210 includes a device capable of generating a vaporized substance, determining that a dosage associated with the vaporized substance has been generated, and/or providing information that identifies that the vaporized substance has been generated. For example, inhalation device 210 may include a vaporizer, an electronic cigarette, a vaporizing pen, a vaporizing machine, and/or the like. Inhalation device 210 may vaporize a substance for inhalation by a user. As examples, the substance may include tetrahydrocannabinol oil, cannabis, tobacco, propylene glycol, glycerin, and/or the like.
[0016] Inhalation device 210 includes a component configured to vaporize an underlying substance to generate a vaporized substance. For example, inhalation device 210 includes a heating component configured to vaporize a substance. Additionally, inhalation device 210 includes a component that permits inhalation device 210 to determine a dosage associated with a vaporized substance. For example, inhalation device 210 includes a sensor (e.g., an optical sensor, a flowmeter, a pressure sensor, a transducer, a microphone, a temperature sensor, and/or the like), a timer, and/or the like.
[0017] User device 220 includes a device capable of receiving, generating, storing, processing, and/or providing information associated with inhalation device 210. For example, user device 220 may include a computing device (e.g., a desktop computer, a laptop computer, a tablet computer, a handheld computer, a smart speaker, a server, etc.), a mobile phone (e.g., a smart phone, a radiotelephone, etc.), a wearable device (e.g., a pair of smart glasses or a smart watch), or a similar device. In some implementations, user device 220 may receive information from and/or transmit information to inhalation device 210.
[0018] User device 220 may connect to inhalation device 210. User device 220 may execute an application that permits a user to input information associated with a dosage. The user may interact with a user interface of user device 220 to input information that identifies a dosage. For example, the user may input information that identifies a particular dosage, may select a dosage, may set a dosage, and/or the like.
[0019] User device 220 may provide information that identifies the input dosage to inhalation device 210. The inhalation device 210 may receive the information that identifies the input dosage, and utilize the information when performing metering as described elsewhere herein.
[0020] Network 230 includes one or more wired and/or wireless networks. For example, network 230 may include a cellular network (e.g., a fifth generation (5G) network, a long-term evolution (LTE) network, a third generation (3G) network, a code division multiple access (CDMA) network, etc.), a public land mobile network (PLMN), a local area network (LAN), a wide area network (WAN), a metropolitan area network (MAN), a telephone network (e.g., the Public Switched Telephone Network (PSTN)), a private network, an ad hoc network, an intranet, the Internet, a fiber optic-based network, or the like, and/or a combination of these or other types of networks.
[0021] The number and arrangement of devices and networks shown in FIG. 2 are provided as an example. In practice, there may be additional devices and/or networks, fewer devices and/or networks, different devices and/or networks, or differently arranged devices and/or networks than those shown in FIG. 2. Furthermore, two or more devices shown in FIG. 2 may be implemented within a single device, or a single device shown in FIG. 2 may be implemented as multiple, distributed devices. Additionally, or alternatively, a set of devices (e.g., one or more devices) of environment 200 may perform one or more functions described as being performed by another set of devices of environment 200.
[0022] FIG. 3 is a diagram of example components of a device 300. Device 300 may correspond to inhalation device 210 and/or user device 220. As shown in FIG. 3, device 300 may include a bus 310, a processor 320, a memory 330, a storage component 340, an input component 350, an output component 360, and a communication interface 370.
[0023] Bus 310 includes a component that permits communication among the components of device 300. Processor 320 is implemented in hardware, firmware, or a combination of hardware and software. Processor 320 is a central processing unit (CPU), a graphics processing unit (GPU), an accelerated processing unit (APU), a microprocessor, a microcontroller, a digital signal processor (DSP), a field-programmable gate array (FPGA), an application-specific integrated circuit (ASIC), or another type of processing component. In some implementations, processor 320 includes one or more processors capable of being programmed to perform a function. Memory 330 includes a random access memory (RAM), a read only memory (ROM), and/or another type of dynamic or static storage device (e.g., a flash memory, a magnetic memory, and/or an optical memory) that stores information and/or instructions for use by processor 320.
[0024] Storage component 340 stores information and/or software related to the operation and use of device 300. For example, storage component 340 may include a hard disk (e.g., a magnetic disk, an optical disk, a magneto-optic disk, and/or a solid state disk), a compact disc (CD), a digital versatile disc (DVD), a floppy disk, a cartridge, a magnetic tape, and/or another type of non-transitory computer-readable medium, along with a corresponding drive.
[0025] Input component 350 includes a component that permits device 300 to receive information, such as via user input (e.g., a touch screen display, a keyboard, a keypad, a mouse, a button, a switch, and/or a microphone). Additionally, or alternatively, input component 350 may include a sensor for sensing information (e.g., a global positioning system (GPS) component, an accelerometer, a gyroscope, an actuator, a light sensor, a flowmeter, and/or the like). Output component 360 includes a component that provides output information from device 300 (e.g., a display, a speaker, light-emitting diodes (LEDs), and/or the like).
[0026] Communication interface 370 includes a transceiver-like component (e.g., a transceiver and/or a separate receiver and transmitter) that enables device 300 to communicate with other devices, such as via a wired connection, a wireless connection, or a combination of wired and wireless connections. Communication interface 370 may permit device 300 to receive information from another device and/or provide information to another device. For example, communication interface 370 may include an Ethernet interface, an optical interface, a coaxial interface, an infrared interface, a radio frequency (RF) interface, a universal serial bus (USB) interface, a Wi-Fi interface, a cellular network interface, or the like.
[0027] Device 300 may perform one or more processes described herein. Device 300 may perform these processes in response to processor 320 executing software instructions stored by a non-transitory computer-readable medium, such as memory 330 and/or storage component 340. A computer-readable medium is defined herein as a non-transitory memory device. A memory device includes memory space within a single physical storage device or memory space spread across multiple physical storage devices.
[0028] Software instructions may be read into memory 330 and/or storage component 340 from another computer-readable medium or from another device via communication interface 370. When executed, software instructions stored in memory 330 and/or storage component 340 may cause processor 320 to perform one or more processes described herein. Additionally, or alternatively, hardwired circuitry may be used in place of or in combination with software instructions to perform one or more processes described herein. Thus, implementations described herein are not limited to any specific combination of hardware circuitry and software.
[0029] The number and arrangement of components shown in FIG. 3 are provided as an example. In practice, device 300 may include additional components, fewer components, different components, or differently arranged components than those shown in FIG. 3. Additionally, or alternatively, a set of components (e.g., one or more components) of device 300 may perform one or more functions described as being performed by another set of components of device 300.
[0030] FIG. 4 is a flow chart of an example process 400 for vaporized substance dosage metering based on an input dosage. In some implementations, one or more process blocks of FIG. 4 may be performed by inhalation device 210. In some implementations, one or more process blocks of FIG. 4 may be performed by another device or a group of devices separate from or including inhalation device 210, such as user device 220.
[0031] As shown in FIG. 4, process 400 may include receiving information that identifies a dosage associated with a vaporized substance (block 410).
[0032] In some implementations, inhalation device 210 may receive the input dosage based on a user interaction with inhalation device 210. For example, a user may interact with an input component of inhalation device 210 to set an input dosage.
[0033] As an example, inhalation device 210 may provide a UI that permits the user to input the input dosage. In this case, the user might input a value for the input dosage, such as "20," "25," "40," etc.
[0034] As another example, inhalation device 210 may include an input component that permits a user to select from predetermined input dosages. For example, the user may select "low," "medium," "heavy," "20 mg," "30 mg," "full," "half," and/or the like.
[0035] As another example, inhalation device 210 may provide, via an output component, an initial input dosage, and the user may interact with an input component to adjust the initial input dosage to a final input dosage. For example, the user may select "more," "less," "+5," "-5," "+," "-," and/or the like. Continuing the example, inhalation device 210 may update a value of the input dosage based on the user's adjustments in real time to permit the user to identify the updated input dosage.
[0036] In some implementations, inhalation device 210 may receive the input dosage from user device 220. For example, user device 220 may receive the input dosage, and provide the input dosage to inhalation device 210. The user may interact with user device 220 to set an input dosage in a similar manner and/or using a similar technique as described above. That is, user device 220, according to various implementations, includes myriad interfaces that permit the input of an input dosage.
[0037] In some implementations, user device 220 may receive the input dosage. For example, a user may interact with user device 220 to set an input dosage in a similar manner as described above. In this case, the user device 220 might not provide the input dosage to inhalation device 210. For example, user device 220 might perform the operations described in process 400. In such cases, the need of user device 220 to provide the input dosage to inhalation device 210 is obviated.
[0038] User device 220 may execute an application that permits a user to input the information associated with the dosage. Based on executing the application, user device 220 may provide, for display via a user interface, information that permits a user to input information associated with the dosage.
[0039] The user may interact with a user interface (or another input component) to input the information associated with the dosage. For example, the user may input a discrete amount (e.g., "20 MG," "27.4 MG," etc.), may select from a predetermined set of dosages, may increase or decrease a dosage by manipulating an input component, and/or the like. It should be understood that the input dosage may be input to user device 220 in a multitude of ways using any variety of techniques.
[0040] In this way, a user may interact with user device 220 to input the dosage instead of setting a dosage using inhalation device 210.
[0041] In this way, a user may select an input dosage that corresponds to a desired dosage that is to be consumed by the user.
[0042] As further shown in FIG. 4, process 400 may include determining that the dosage associated with the vaporized substance has been generated (block 420).
[0043] In some implementations, inhalation device 210 may determine that the input dosage has been generated. For example, inhalation device 210 may generate the vaporized substance, and determine that an amount of the vaporized substance is equal to the input dosage.
[0044] In some implementations, inhalation device 210 may determine, using a sensor of inhalation device 210, that the input dosage has been generated. For example, inhalation device 210 may use an optical sensor, a flow sensor, a pressure sensor, and/or the like, to determine that the input dosage has been generated.
[0045] Alternatively, inhalation device 210 may determine, using a timer, that the input dosage has been generated. For example, inhalation device 210 may store a data structure that maps time values and dosages, and determine, using the data structure, that an amount of time corresponding to the input dosage has elapsed. In this case, inhalation device 210 may initiate the timer based on initiating a heating component, based on detecting flow of vapor, based on detecting that vapor is generated, based on detecting that the user's lips are contacting inhalation device 210, and/or the like.
[0046] In some implementations, user device 220 may determine that the input dosage has been generated. For example, user device 220 may store a data structure similar as described above, and may determine, using the data structure, that an amount of time corresponding to the input dosage has elapsed. In this case, inhalation device 210 may transmit an initiation signal that permits user device 220 to initiate timing, and user device 220 may initiate timing based on the initiation signal. Inhalation device 210 may transmit the initiation signal based on any of the foregoing factors mentioned above.
[0047] In some implementations, inhalation device 210 is configured such that a flow pathway of the vaporized substance is substantially in view of a sensor configured to meter the vaporized substance. For example, inhalation device 210 may include a flow pathway having at least one cross section that is entirely within view (i.e., capable of being detected) of the sensor.
[0048] In this way, substantially all of the vaporized substance is required to pass through the foregoing cross-sectional area, thereby rendering the vaporized substance detectable by the sensor. In some cases, a frequency of measurement is configured to satisfy the flow rate of the vaporized substance to permit substantially all of the vaporized substance to be detected, and, ultimately, metered.
[0049] The cross-sectional area can be elongated to create a three-dimensional area in which the vapor/air must pass. This space is substantially visible to a sensor of inhalation device 210. In this case, the frequency of measurements by the sensor might be less than the frequency mentioned elsewhere herein because the three-dimensional space incorporates a larger volume of vapor visible to the sensor.
[0050] In some implementations, vapor/air mixtures tend to be non-homogenous and poorly mixed. The density of the vapor may vary greatly in small distances. The density of the vapor may also change quickly depending on temperature, pressure, motion and turbulence. One can anticipate that measuring substantially all the air/vapor will yield better results that measuring only a portion of the air/vapor.
[0051] When measuring vapor it is important to measure the vapor density often enough to properly characterize the vapor quantity. Due to the nature of vapor, it will probably be poorly mixed and non-homogeneous. In a flowing environment, one may find snapshots of high density flowed by low density. Ideally, the frequency of the snapshots would match the flow speed in such a way that all the vapor cross sections are captured. Such a setup may require that the snapshot frequency vary according to the flow rate. As an example, assume that the vapor will travel a length L in a certain amount of time dependent on flow rate. Let say that this time is 0.25 seconds. In such a case, it would be advantageous to take a snapshot at least once per 0.25 seconds to ensure all vapor is seen by the sensor. It may also be found that substantially good characterization of the flow is possible with less frequent snapshots. Such a determination can be made after proper consideration to liquid characteristics, physical pathway constraints and dynamics, temperature, desired level of accuracy and various other factors.
[0052] In yet another embodiment is an inhalation device 210, with metering capabilities as described in this disclosure, where the vapor is produced by vibrations (rather than heat). Such a device would have a reservoir for holding the drug in liquid form (could also be in solid form), and creating a vibration of certain frequency in order to transform the liquid into a vapor. A piezoelectric may be used to create the vibrations. The liquid may be held/suspended in a membrane that has small holes. The membrane may be metal and have porous qualities. The vapor produced may then be inhaled by the user. Adding heat to the vaporization method, may help the performance of this device. Further, it may help create vapor particle sizes that are better suited for inhalation and absorption by the lungs. Particle size has an effect on how far into the lungs the particles may travel, thus affecting where the particles settle and may get absorbed.
[0053] In yet another embodiment is an inhalation device 210, with metering capabilities as described in this disclosure, for use with plants and herbs (or other naturally occurring materials). This device would include a heating element, a location or chamber to hold the plant material, a vapor sensor for measuring the vapor, it may have a pressure or airflow sensor for determining air flow speed. It may have a puff switch for detecting a puff.
[0054] In another embodiment is an inhalation device 210 including metering ability for "dabbing." This embodiment includes an inhalation device 210 with metering, as described in this disclosure, for use with highly concentrated extracts. These extract may be solid or waxy. They may be substantially solid and non-fluid. This device would include a heating element, a location or chamber to hold substantially solid material, a vapor sensor for measuring the vapor, it may have a pressure or airflow sensor for determining air flow speed. It may have a puff switch for detecting a puff.
[0055] In embodiments described above, vapor is metered after it is produced. In another embodiment the material/drug is metered before it is vaporized (or as it is being vaporized). This embodiment requires metering the drug in to the vaporizing unit such that the amount that is being vapor is controlled by the metering process. For example, the drug may be made into a solid and fed into the heating element by a lead screw. The feed rate would be controlled and metered. This embodiment can also work with a liquid and fed at a certain rate into a heating element for evaporation. Another embodiment includes a piezoelectric/vibration unit to be used a mechanical way to feed the drug into a heating element.
[0056] Another embodiment provides an alternative way to meter vapor in an inhalation device 210. More specifically, this embodiment discusses a way by which to measure that density of vapor in a pathway. There would be two probes positioned in the vapor pathway. The probes would be located at a set distance from one another. The probes may be made of a conductive material. There would be a certain high amount of electrical resistance between the tips of the probes in the default `no vapor` state. This resistance would be measured and used as a baseline. As vapor flows past the probes, it will fill the space between the probes with particles of vapor. The vapor particles are more electronically conductive that air. Hence, the vapor will change the resistance reading between the probes.
[0057] The drug can be situated in such a way that it can only move into the heating element. This can be achieved by a ratchet design or one way valves that only allows motion in one direction. The vibration caused by the piezoelectric/vibrator can be setup in such a way that it will bias the drug into the heating element when activated. Another way is to feed the drug into the heating element by discrete individual touches. By touching the drug to the heating element for a certain amount of time and pressure. It may be necessary to repeat this action with high frequency to get the desired amount.
[0058] Another embodiment provides an alternative way to meter vapor in an inhalation device. More specifically, this embodiment discusses a way by which to measure that density of vapor in a pathway. There would be two probe positioned in the vapor pathway. The probes would be located at a set distance from one another. The probes may be made of a conductive material. There would be a certain high amount of electrical resistance between the tips of the probes in the default `no vapor` state. This resistance would be measured and used as a baseline. As vapor flows past the probes, it will fill the space between the probes with particles of vapor. The vapor particles are more electronically conductive that air. Hence, the vapor will change the resistance reading between the probes.
[0059] The resistance readings may be amplified by changing the shape and orientation of the probes. Heat or electrical charge may also improve the results. Another embodiment may include connecting the probes by a thin wire that is positioned to accumulate tiny particles of the passing vapor during flow. The resistance of the wire will change accordingly.
[0060] In another embodiment, an inhalation device 210 according to this disclosure may use a sensor positioned on the mouthpiece such that when the user's lips touch the mouthpiece, the sensor can detect this action. Preferably, a set of sensors will be positioned such that the one sensor touches the top lip and the other the bottom lip. Capacitive and resistive touch sensors may be used for this. The above described embodiments may also be fitted with a push button that can be used by the user to initiate and/or activate the device. When the user stops pushing the button, the device can stop.
[0061] In yet another embodiment, an inhalation device 210 according to this disclosure may be configured in such a way that the user may define when the device will turn off. The user can define this by setting an amount of drug (dose) that they want to consume. The unit will remain operational until the dose is fully consumed. The device will measure the amount inhaled in real-time and will stop supplying vapor once the dose is met. This allows the user to get the dose they want without actively monitoring the metering interface.
[0062] As further shown in FIG. 4, process 400 may include providing information that identifies that the dosage associated with the vaporized substance has been generated (block 430).
[0063] In some implementations, inhalation device 210 may provide, via an output component, information that identifies that the input dosage has been generated. For example, inhalation device 210 may provide a visual indication, an audio indication, a tactile indication, and/or the like. In some implementations, user device 220 may provide, via an output component, the information that identifies that the input dosage has been generated.
[0064] It should be understood that some or all of the operations of process 400 may be performed by inhalation device 210 and user device 220, and that myriad permutations of operations of process 400 may be performed by inhalation device 210 and user device 220.
[0065] In some implementations, inhalation device 210 may adjust a state of inhalation device 210 based on determining that the input dosage has been generated. For example, inhalation device 210 may stop the operation of a heating element of inhalation device 210, may turn off entirely, and/or the like.
[0066] In some implementations, inhalation device 210 may determine that an amount of the generated vaporized substance is less than the input dosage. For example, assume that an amount of available substance is insufficient to generate the input dosage. In this case, inhalation device (or user device 220) may provide information that identifies that the input dosage has not been generated.
[0067] Although FIG. 4 shows example blocks of process 400, in some implementations, process 400 may include additional blocks, fewer blocks, different blocks, or differently arranged blocks than those depicted in FIG. 4. Additionally, or alternatively, two or more of the blocks of process 400 may be performed in parallel.
[0068] The foregoing disclosure provides illustration and description, but is not intended to be exhaustive or to limit the implementations to the precise form disclosed. Modifications and variations are possible in light of the above disclosure or may be acquired from practice of the implementations.
[0069] As used herein, the term component is intended to be broadly construed as hardware, firmware, or a combination of hardware and software.
[0070] It will be apparent that systems and/or methods, described herein, may be implemented in different forms of hardware, firmware, or a combination of hardware and software. The actual specialized control hardware or software code used to implement these systems and/or methods is not limiting of the implementations. Thus, the operation and behavior of the systems and/or methods were described herein without reference to specific software code--it being understood that software and hardware may be designed to implement the systems and/or methods based on the description herein.
[0071] Even though particular combinations of features are recited in the claims and/or disclosed in the specification, these combinations are not intended to limit the disclosure of possible implementations. In fact, many of these features may be combined in ways not specifically recited in the claims and/or disclosed in the specification. Although each dependent claim listed below may directly depend on only one claim, the disclosure of possible implementations includes each dependent claim in combination with every other claim in the claim set.
[0072] No element, act, or instruction used herein should be construed as critical or essential unless explicitly described as such. Also, as used herein, the articles "a" and "an" are intended to include one or more items, and may be used interchangeably with "one or more."
[0073] Furthermore, as used herein, the term "set" is intended to include one or more items (e.g., related items, unrelated items, a combination of related and unrelated items, etc.), and may be used interchangeably with "one or more." Where only one item is intended, the term "one" or similar language is used. Also, as used herein, the terms "has," "have," "having," or the like are intended to be open-ended terms. Further, the phrase "based on" is intended to mean "based, at least in part, on" unless explicitly stated otherwise.
* * * * *
D00000
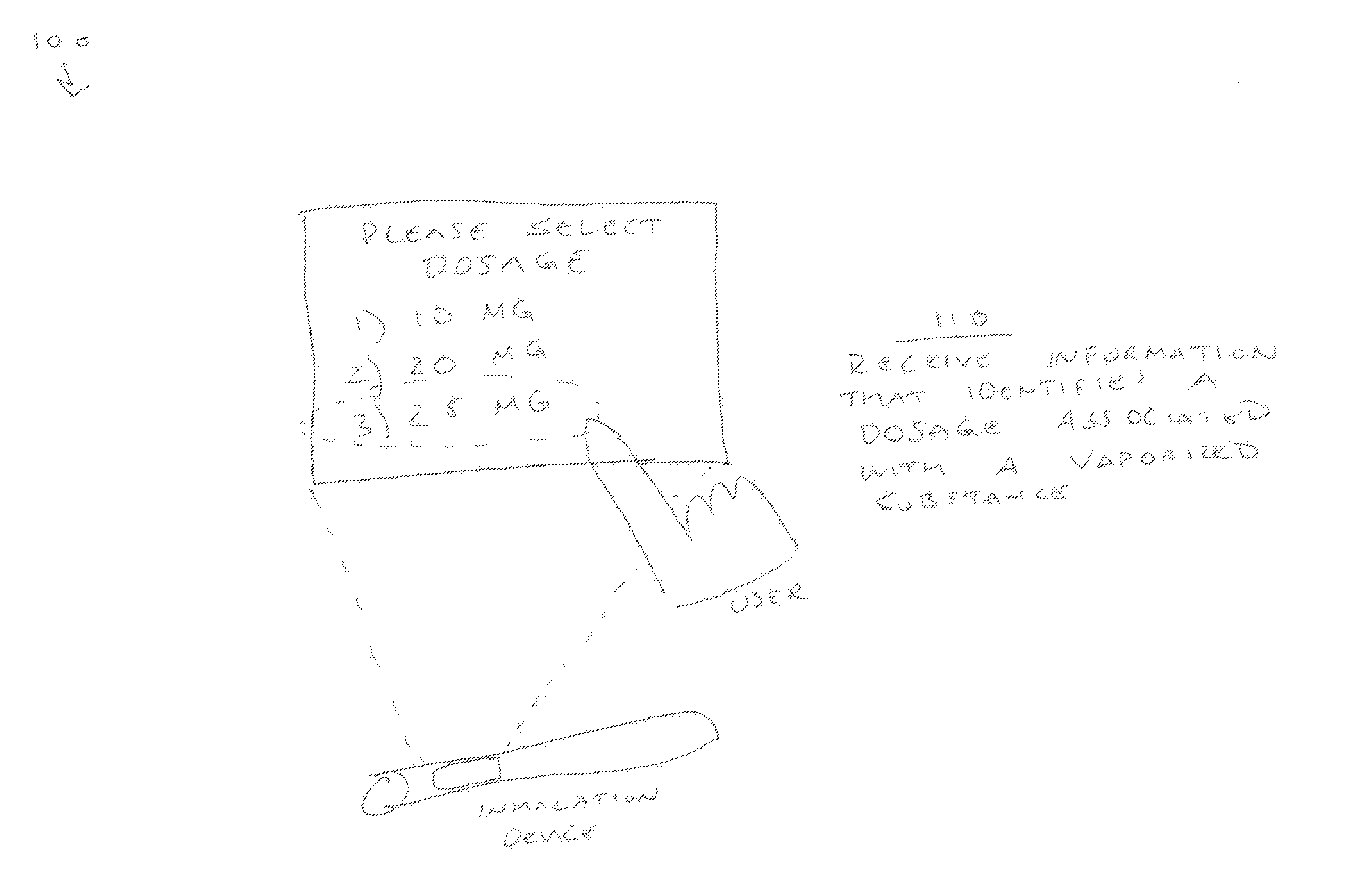
D00001
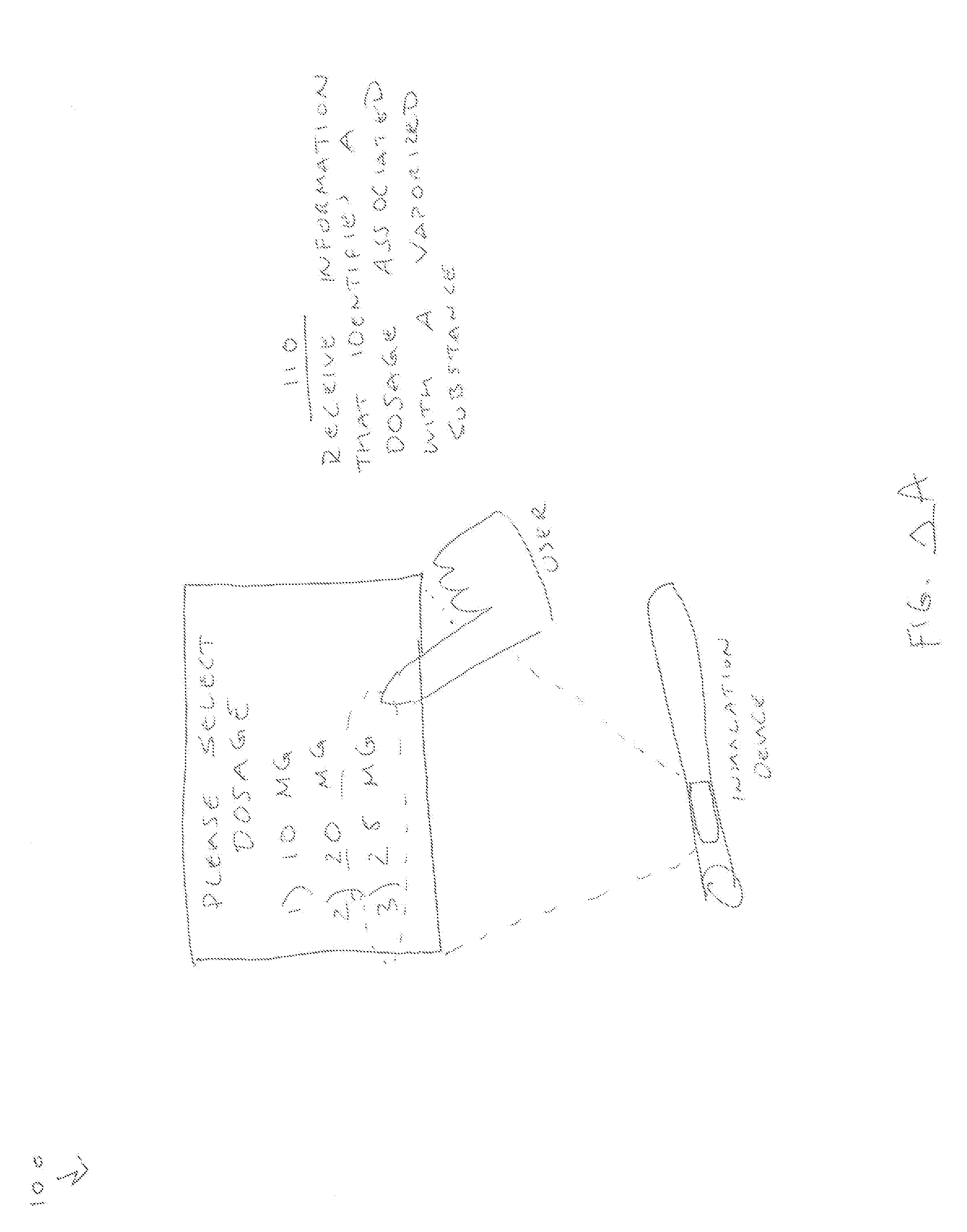
D00002

D00003

D00004

D00005

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.