Devices And Methods For Assisting Valve Function, Replacing Venous Valves, And Predicting Valve Treatment Success
Kassab; Ghassan S.
U.S. patent application number 16/365373 was filed with the patent office on 2019-07-18 for devices and methods for assisting valve function, replacing venous valves, and predicting valve treatment success. This patent application is currently assigned to 3DT Holdings, LLC. The applicant listed for this patent is 3DT Holdings, LLC. Invention is credited to Ghassan S. Kassab.
| Application Number | 20190216590 16/365373 |
| Document ID | / |
| Family ID | 47881375 |
| Filed Date | 2019-07-18 |








| United States Patent Application | 20190216590 |
| Kind Code | A1 |
| Kassab; Ghassan S. | July 18, 2019 |
DEVICES AND METHODS FOR ASSISTING VALVE FUNCTION, REPLACING VENOUS VALVES, AND PREDICTING VALVE TREATMENT SUCCESS
Abstract
Devices and methods for assisting valve function, replacing venous valves, and predicting valve treatment successes. In at least one embodiment of an endograft valve device of the present disclosure, the device comprises an endograft body configured for expansion from a collapsed configuration to an expanded configuration within a luminal organ and comprising a first portion having a proximal end defining a proximal end aperture and a distal end defining a distal end aperture, the first portion tapering toward the distal end so that the distal end aperture has a relatively smaller cross-sectional area than the proximal end aperture when the valve device is expanded, and a valve portion positioned at or near the distal end of the first portion, the valve portion configured to receive fluid flowing through the distal end aperture of the first portion.
| Inventors: | Kassab; Ghassan S.; (La Jolla, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | 3DT Holdings, LLC San Diego CA |
||||||||||
| Family ID: | 47881375 | ||||||||||
| Appl. No.: | 16/365373 | ||||||||||
| Filed: | March 26, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 13539607 | Jul 2, 2012 | 10238483 | ||
| 16365373 | ||||
| 61547378 | Oct 14, 2011 | |||
| 61535689 | Sep 16, 2011 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61F 2/06 20130101; A61B 17/12013 20130101; A61F 2240/002 20130101; A61F 2002/068 20130101; A61F 2/2475 20130101; A61B 2017/00557 20130101; A61F 2/2433 20130101 |
| International Class: | A61F 2/06 20060101 A61F002/06; A61B 17/12 20060101 A61B017/12; A61F 2/24 20060101 A61F002/24 |
Claims
1. An endograft valve device comprising: a first portion having a proximal end, a distal end, an outer portion wall surface and an inner portion wall surface, wherein a tapered wall thickness is defined between the inner and outer portion wall surfaces, and wherein the inner portion wall surface further defines a lumen therethrough; and wherein the first portion has a tapered wall thickness that has a greater wall thickness at the distal end than at the proximal end.
2. The endograft valve device of claim 1, further comprising: a second portion comprising a second portion outer portion wall surface and a second portion inner portion wall surface, wherein a tapered wall thickness is defined between the second portion inner wall surface and second portion outer portion wall surfaces; the second portion inner wall surface further defining the lumen therethrough, the lumen extending from a second portion distal end to a second portion proximal end; wherein the second portion has a tapered wall thickness that has a greater wall thickness at the second portion distal end than at the second portion proximal end; and wherein the distal end of the first portion is adjacent to the second portion proximal end.
3. The endograft valve device of claim 1, wherein the first portion comprises a distal end aperture and a proximal end aperture and the distal end aperture has a relatively smaller cross-sectional area than the proximal end aperture.
4. The endograft valve device of claim 2, wherein a valve is coupled to a portion of the endograft body selected from the group consisting of the first portion at or near the distal end of the first portion and the second portion at or near the second portion proximal end.
5. The endograft valve device of claim 2, wherein the second portion comprises a second portion distal end aperture and a second portion proximal end aperture, where the second portion distal end aperture has a relatively smaller cross-sectional area than the second portion proximal end aperture.
6. The endograft valve device of claim 2, wherein the first portion comprises a distal end aperture and a proximal end aperture and the distal end aperture has a relatively smaller cross-sectional area than the proximal end aperture.
7. The endograft valve device of claim 6, further comprising: the second portion having a second portion proximal end aperture in fluid communication with the distal end aperture of the first portion, and the second portion having a second portion distal end aperture wherein the second portion distal end aperture has a relatively smaller cross-sectional area than the second portion proximal end aperture.
8. An endograft valve device, comprising: a first portion having a proximal end and a distal end; a second portion comprising a second portion outer portion wall surface and a second portion inner portion wall surface, wherein a tapered wall thickness is defined between the second portion inner wall surface and second portion outer portion wall surfaces; the second portion inner wall surface further defining the lumen therethrough, the lumen extending from a second portion distal end to a second portion proximal end; wherein the second portion has a tapered wall thickness that has a greater wall thickness at the second portion distal end than at the second portion proximal end; and wherein the distal end of the first portion is adjacent to the second portion proximal end.
9. The endograft valve device of claim 8, wherein the first portion comprises a distal end aperture and a proximal end aperture; and the distal end aperture has a relatively smaller cross-sectional area than the proximal end aperture.
10. The endograft valve device of claim 9, wherein the second portion comprises a second portion distal end aperture and a second portion proximal end aperture; and the second portion distal end aperture has a relatively smaller cross-sectional area than the second portion proximal end aperture.
11. The endograft valve device of claim 8, wherein the second portion comprises a second portion distal end aperture and a second portion proximal end aperture; and the second portion distal end aperture has a relatively smaller cross-sectional area than the second portion proximal end aperture.
12. The endograft valve device of claim 8, wherein a valve is coupled to a portion of the endograft body selected from the group consisting of the first portion at or near the distal end of the first portion and the second portion at or near the second portion proximal end.
13. A system comprising an endograft valve device comprising: an endograft body comprising: a first portion having a proximal end, a distal end, a first outer portion wall surface and a first inner portion wall surface, wherein a tapered wall thickness is defined between the first inner portion wall surface and first outer portion wall surface, and wherein the first inner portion wall surface further defines a lumen therethrough; wherein the first portion has a tapered wall thickness that has a greater wall thickness at the distal end than at the proximal end; a second portion comprising a second portion inner portion wall surface, further defining the lumen therethrough, the lumen extending from a second portion distal end to a second portion proximal end; wherein the distal end of the first portion is adjacent to the second portion proximal end; and a valve coupled to a portion of the endograft body selected from the group consisting of the first portion at or near the distal end of the first portion and the second portion at or near the second portion proximal end.
14. The system comprising an endograft valve device of claim 13 wherein the first portion comprises a distal end aperture and a proximal end aperture and the distal end aperture has a relatively smaller cross-sectional area than the proximal end aperture.
15. The system comprising an endograft valve device of claim 13 wherein the second portion comprises a second portion distal end aperture and a second portion proximal end aperture; and the second portion distal end aperture has a relatively smaller cross-sectional area than the second portion proximal end aperture.
16. The system comprising an endograft valve device of claim 14 wherein the second portion comprises a second portion distal end aperture and a second portion proximal end aperture; and the second portion distal end aperture has a relatively smaller cross-sectional area than the second portion proximal end aperture.
17. The system comprising an endograft valve device of claim 13 wherein the second portion proximal end is in fluid communication with the distal end of the first portion.
18. The system comprising an endograft valve device of claim 13 wherein the endograft body is configured for expansion from a collapsed configuration to an expanded configuration within a luminal organ.
19. The system of claim 18, wherein the collapsed configuration is sized so that the endograft body fits within the luminal organ, and wherein the expanded configuration is sized so that the endograft body is securely positioned within the luminal organ upon expansion.
20. The system comprising an endograft valve device of claim 13 further comprising: a cuff configured to fit around a blood vessel and further configured to periodically compress the blood vessel; and a processor operably coupled to the cuff, the processor configured to control a compression rate and a relaxation rate; and wherein when the cuff is positioned around the blood vessel at a first location, operation of the processor causes the cuff to compress the blood vessel and relax compression of the blood vessel, whereby relaxation at the relaxation rate causes blood to move through the blood vessel at the first location.
Description
PRIORITY
[0001] The present application is related to, claims the priority benefit of, and is a U.S. continuation patent application of, U.S. patent application Ser. No. 13/539,607, filed Jul. 2, 2012 and issued as U.S. Pat. No. 10,238,483 on Mar. 26, 2019, which is related to, and claims the priority benefit of, U.S. Provisional Patent Application Ser. No. 61/547,378, filed Oct. 14, 2011, and U.S. Provisional Patent Application Ser. No. 61/535,689, filed Sep. 16, 2011. The contents of each of these applications are incorporated by reference in their entirety into this disclosure.
BACKGROUND
[0002] It has been known for several years that the three contributing factors to venous thrombosis are stasis (slow blood flow), changes in blood composition, and changes in vessel wall. These three contributing factors are known as Virchow's triad.
[0003] Venous insufficiency is a complex disease that involves thrombosis (blood clot abnormalities and/or endothelial dysfunction), venous hypertension, reverse flow (reflux), and venous remodeling, amongst others. The two major contributors to venous insufficiency are venous occlusion and valve incompetence (reflux).
[0004] Blood stasis promotes a thrombogenic response through various biochemical activators. Attempts have been made to create percutaneous venous valve stent systems to replace a prolapsed or otherwise dysfunctional venous valve, but no attempt has previously been proven successful. In order for such a system to work, one of the contributing factors to venous blood thrombosis, namely stasis, must be overcome in order for the valve to remain patent and to avoid thrombosis/clotting
[0005] A valve device or system, operable to replace a prolapsed or otherwise dysfunctional venous valve that effectively operates and overcomes the stasis contributing factor, would be well-received in the marketplace. In addition, a device useful to assist blood flow through a blood vessel, methods of using the same, and methods for predicting a potential success for an individual patient in connection with a potential valve treatment procedure, would also be well-received in the marketplace.
BRIEF SUMMARY
[0006] In at least one exemplary embodiment of an endograft valve device of the present disclosure, the endograft valve device comprises an endograft body configured for expansion from a collapsed configuration to an expanded configuration within a luminal organ, the endograft body comprising a first portion having a proximal end defining a proximal end aperture and a distal end defining a distal end aperture, the first portion tapering toward the distal end so that the distal end aperture has a relatively smaller cross-sectional area than the proximal end aperture when the valve device is expanded, and a valve portion positioned at or near the distal end of the first portion, the valve portion configured to receive fluid flowing through the distal end aperture of the first portion. In another embodiment, the endograft valve device further comprises a second portion having a second portion proximal end defining a second portion proximal end aperture and a second portion distal end defining a second portion distal end aperture, the second portion tapering toward the second portion distal end so that the second portion distal end aperture has a relatively smaller cross-sectional area than the second portion proximal end aperture when the valve device is expanded, wherein the distal end of the first portion is adjacent to the second portion proximal end, and wherein the valve portion is positioned at or near the second portion proximal end.
[0007] In at least one exemplary embodiment of an endograft valve device of the present disclosure, the endograft valve device comprises an endograft body configured for expansion within a luminal organ, the endograft body comprising a first portion having a proximal end defining a proximal end aperture and a distal end defining a distal end aperture, the first portion tapering toward the distal end so that the distal end aperture has a relatively smaller cross-sectional area than the proximal end aperture when the valve device is expanded, a second portion having a proximal end defining a proximal end aperture and a distal end defining a distal end aperture, the second portion tapering toward the distal end so that the distal end aperture has a relatively smaller cross-sectional area than the proximal end aperture when the valve device is expanded, wherein the distal end of the first portion is adjacent to the proximal end of the second portion, and a valve portion positioned at or near the proximal end of the second portion, the valve portion configured to receive fluid flowing through the distal end aperture of the first portion. In another embodiment, the endograft body is configured to expand from a collapsed configuration to an expanded configuration. In yet another embodiment, the endograft body has a first configuration, the first configuration sized so that the endograft body may fit within the luminal organ. In an additional embodiment, the endograft has a second configuration, the second configuration sized so that the endograft body may be securely positioned within the luminal organ upon expansion.
[0008] In at least one exemplary embodiment of an endograft valve device of the present disclosure, the valve portion is coupled to the first portion. In an additional embodiment, the valve portion is coupled to the second portion. In yet an additional embodiment, the endograft body is sized and shaped to fit around a guidewire. In another embodiment, the endograft body is sized and shaped to fit around a catheter. In yet another embodiment, the endograft body is capable of expanding due to inflation of a balloon coupled to the catheter.
[0009] In at least one exemplary embodiment of an endograft valve device of the present disclosure, when the endograft body is expanded within the luminal organ, an outer portion of the endograft body contacts the luminal organ, an inner portion of the endograft body is configured to permit fluid to flow therethrough. In another embodiment, the outer portion defines an outer portion wall, and the inner portion defines an inner portion wall. In yet another embodiment, the outer portion defines an outer portion relative surface, and the inner portion defines an inner portion relative surface. In at least one exemplary embodiment of an endograft valve device of the present disclosure, when the endograft body is expanded within the luminal organ, fluid flowing through the first portion increases in velocity as the fluid approaches the valve portion. In an additional embodiment, when the endograft body is expanded within the luminal organ, fluid flowing through the first portion increases in velocity as the fluid approaches the valve portion and increases shear stresses at the valve portion. In yet an additional embodiment, the valve portion comprises leaflets.
In at least one exemplary embodiment of an endograft valve device of the present disclosure, the endograft valve device further comprises an anti-clotting agent positioned upon at least a portion of the endograft valve device. In another embodiment, the anti-clotting agent is positioned upon at least an inner portion of the endograft valve device. In yet another embodiment, the anti-clotting agent is positioned upon at least an outer portion of the endograft valve device. In an additional embodiment, the anti-clotting agent is selected from the group consisting of heparin, thrombomodulin, and/or endothelial cell protein C. In yet an additional embodiment, the endograft body is comprises of a biologically-compatible material selected from the group consisting of polytetrafluoroethylene, Gore-Tex.RTM., and/or a nickel titanium alloy, such as nitinol. In an additional embodiment, the endograft valve device further comprises a guidewire and a catheter, wherein the catheter is configured to fit around the guidewire, and wherein the endograft valve device is configured to fit around the catheter. In yet an additional embodiment, the endograft valve device comprises a venous endograft valve device configured to fit within a vein.
[0010] In at least one exemplary embodiment of a valve system of the present disclosure, the valve system comprises an exemplary endograft valve device of the present disclosure, a guidewire, and a catheter, wherein the catheter is configured to fit around the guidewire, and wherein the endograft valve device is configured to fit around the catheter. In another embodiment, the catheter comprises a balloon catheter.
[0011] In at least one exemplary embodiment of a method of using an exemplary endograft valve device of the present disclosure, the method comprises the steps of introducing a guidewire into a luminal organ of a patient, advancing an endograft valve device of the present disclosure along the guidewire to a desired location within the luminal organ, and expanding the endograft valve device within the luminal organ. In another embodiment, the method further comprises the step of withdrawing the guidewire from the luminal organ. In yet another embodiment, the luminal organ is a vein, and the endograft valve device is configured as a venous endograft valve device.
[0012] In at least one exemplary embodiment of a method of determining whether a patient is suitable for a valve procedure of the present disclosure, the method comprises the steps of obtaining data indicative to a patient's venous geometry at a first location, obtaining data indicative of a flow velocity of blood within a vein of the patient at or near the first location, preparing a digital model of the vein of the patient using the data indicative to the patient's venous geometry and/or the data indicative of the flow velocity to obtain patient venous data, and determining whether the patient is suitable for a valve procedure based at least in part on the patient venous data. In another embodiment, the steps of obtaining data are performed using duplex ultrasonography. In yet another embodiment, the steps of obtaining data are performed using an impedance device selected from the group consisting of an impedance wire and an impedance catheter. In an additional embodiment, the steps of obtaining data are performed using an impedance device comprising a device body and at least four electrodes positioned thereon, the at least four electrodes comprising two electrodes configured to excite an electric field and two electrodes configured to obtain a conductance measurement within the electric field.
[0013] In at least one exemplary embodiment of a method of determining whether a patient is suitable for a valve procedure of the present disclosure, the step of preparing the digital model of the vein of the patient is performed using the data indicative to the patient's venous geometry and the data indicative of the flow velocity to obtain patient venous data. In an additional embodiment, the step of preparing the digital model of the vein of the patient is performed using the data indicative to the patient's venous geometry to prepare a first digital model and the data indicative of the flow velocity to prepare a second digital model, wherein the patient venous data is indicative of the first digital model and the second digital model. In yet an additional embodiment, the method further comprises the step of incorporating data indicative of at least one valve into the digital model.
[0014] In at least one exemplary embodiment of a method of determining whether a patient is suitable for a valve procedure of the present disclosure, the step of determining whether the patient is suitable for a valve procedure based at least in part on the patient venous data and the data indicative of at least one valve. In another embodiment, the step of determining whether the patient is suitable for a valve procedure is based upon at least patient venous data indicative of flow and shear stress, and an ultimate determination is based upon a comparison of the patient venous data indicative of flow and shear stress to at least one threshold.
[0015] In at least one exemplary embodiment of a method of preparing a digital model of a vein of the present disclosure, the method comprising the steps of obtaining data indicative to a patient's venous geometry at a first location within the patient, obtaining data indicative of a flow velocity of blood within a vein of the patient at or near the first location, preparing at least one digital model of the vein of the patient using the data indicative to the patient's venous geometry and/or the data indicative of the flow velocity to obtain patient venous data, and using the patient venous data for at least one test purpose, the at least one test purpose selected from the group consisting of i) determining whether the patient is suitable for a valve procedure based at least in part on the patient venous data and ii) testing one or more virtual valve device configurations in at least one simulation using the digital model. In another embodiment, the steps of obtaining data are performed using a device selected from the group consisting of a duplex ultrasound device, an impedance wire, and impedance catheter, an impedance device comprising a device body and at least four electrodes positioned thereon, the at least four electrodes comprising two electrodes configured to excite an electric field and two electrodes configured to obtain a conductance measurement within the electric field. In an additional embodiment, the step of using the patient venous data is performed by using the patient venous data for the at least one test purpose of determining whether the patient is suitable for the valve procedure based at least in part on the patient venous data. In yet an additional embodiment, the method further comprises the step of incorporating data indicative of at least one valve into the digital model. In another embodiment, the determination of whether the patient is suitable for the valve procedure based at least in part on information selected from the group consisting of i) the patient venous data and the data indicative of at least one valve, and ii) patient venous data indicative of flow and shear stress, and wherein an ultimate determination is based upon a comparison of the patient venous data indicative of flow and shear stress to at least one threshold.
[0016] In at least one exemplary embodiment of a method of preparing a digital model of a vein of the present disclosure, the step of using the patient venous data is performed by using the patient venous data for the at least one test purpose of testing one or more virtual valve device configurations in at least one simulation using the digital model. In an additional embodiment, the method further comprises the step of adjusting the one or more virtual valve device configurations and retesting the adjusted one or more virtual valve device configurations. In yet an additional embodiment, the method further comprises the step of preparing a physical valve device based upon at least one of the adjusted one or more virtual valve device configurations.
[0017] In at least one exemplary embodiment of an external assist device of the present disclosure, the device comprises a cuff configured to fit around a blood vessel and further configured to periodically compress the blood vessel, and a processor operably coupled to the cuff, the processor configured to control a compression rate and a relaxation rate, wherein when the device is positioned around the blood vessel at a first location, operation of the processor causes the cuff to compress the blood vessel and relax compression of the blood vessel, whereby relaxation at the relaxation rate causes blood to move through the blood vessel at the first location. In another embodiment, the compression rate is slower than the relaxation rate. In yet another embodiment, the device further comprises a power source operably coupled to the cuff, the power source configured to provide power to the cuff and/or the processor to facilitate compression and relaxation of the cuff. In an additional embodiment, the device further comprises a connector coupled to the power source and to the cuff, the connector configured to allow power from the power source to be transmitted therethrough to the cuff. In yet an additional embodiment, the connector comprises a wire.
[0018] In at least one exemplary embodiment of an external assist device of the present disclosure, the processor is configured so that the compression rate and the relaxation rate can be changed to a different compression rate and a different relaxation rate. In an additional embodiment, when the device is positioned distal to a blood vessel valve, operation of the device causes blood to flow through the vessel valve toward the device. In yet an additional embodiment, the blood vessel valve is selected from the group consisting of a native valve and a prosthetic valve. In another embodiment, when the device is positioned around the blood vessel at a first location, the blood flows through the blood vessel at the first location a first rate without operation of the device, and the blood flows through the blood vessel at the first location at a second rate during operation of the device, wherein the second rate is faster than the first rate. In yet another embodiment, when the device is positioned around the blood vessel at a first location, the blood flows through the blood vessel at the first location a first rate range without operation of the device, and the blood flows through the blood vessel at the first location at a second rate range during operation of the device, wherein the second rate range has a faster top rate than the first rate range.
[0019] In at least one exemplary embodiment of a method of facilitating blood flow through a blood vessel of the present disclosure, the method comprises the steps of positioning an exemplary external assist device of the present disclosure around a blood vessel, and operating the external assist device to alternately compress the blood vessel and relax compression of the blood vessel, wherein relaxation of compression causes blood to flow through the blood vessel. In another embodiment, relaxation of compression causes blood to flow through the blood vessel at a faster rate than a native blood flow rate. In yet another embodiment, the step of operating the external assist device comprises operating the external assist device to alternative compress the blood vessel at a first rate and to relax compression of the blood vessel at a second rate, wherein the second rate is faster than the first rate.
BRIEF DESCRIPTION OF THE DRAWINGS
[0020] The disclosed embodiments and other features, advantages, and disclosures contained herein, and the matter of attaining them, will become apparent and the present disclosure will be better understood by reference to the following description of various exemplary embodiments of the present disclosure taken in conjunction with the accompanying drawings, wherein:
[0021] FIG. 1 shows a portion of a guidewire used to position an exemplary endograft valve device into a luminal organ, according to at least one exemplary embodiment of the present disclosure;
[0022] FIG. 2 shows an endograft valve device in a first configuration positioned over a guidewire, according to at least one exemplary embodiment of the present disclosure;
[0023] FIG. 3 shows an endograft valve device in a second configuration positioned over a guidewire, according to at least one exemplary embodiment of the present disclosure;
[0024] FIG. 4 shows an endograft valve device in a second configuration, according to at least one exemplary embodiment of the present disclosure;
[0025] FIG. 5 shows fluid velocities (represented by position and length of arrows) when a valve portion of an endograft valve device is open, according to at least one exemplary embodiment of the present disclosure;
[0026] FIG. 6 shows fluid velocities (represented by position and length of arrows) when a valve portion of an endograft valve device is partially closed, according to at least one exemplary embodiment of the present disclosure;
[0027] FIG. 7 shows a block diagram of components of a valve system, according to at least one exemplary embodiment of the present disclosure;
[0028] FIG. 8 shows a diagram of method steps, according to at least one exemplary embodiment of the present disclosure;
[0029] FIG. 9 shows steps of a method of determining whether a patient is suitable for a valve procedure, according to at least one exemplary embodiment of the present disclosure;
[0030] FIG. 10A shows an external assist device positioned around a blood vessel, according to at least one exemplary embodiment of the present disclosure;
[0031] FIG. 10B shows an external assist device positioned around and constricting a blood vessel, according to at least one exemplary embodiment of the present disclosure;
[0032] FIG. 11 shows an external assist device positioned around a blood vessel and an endograft valve device positioned within a vessel, according to at least one exemplary embodiment of the present disclosure; and
[0033] FIG. 12 shows steps of a method of facilitating blood flow through a blood vessel, according to at least one exemplary embodiment of the present disclosure.
[0034] An overview of the features, functions and/or configurations of the components depicted in the various figures will now be presented. It should be appreciated that not all of the features of the components of the figures are necessarily described. Some of these non-discussed features, such as various couplers, etc., as well as discussed features are inherent from the figures themselves. Other non-discussed features may be inherent in component geometry and/or configuration.
DETAILED DESCRIPTION
[0035] For the purposes of promoting an understanding of the principles of the present disclosure, reference will now be made to the embodiments illustrated in the drawings, and specific language will be used to describe the same. It will nevertheless be understood that no limitation of the scope of this disclosure is thereby intended.
[0036] With respect to patient treatment for valve and/or venous insufficiency, the disclosure of the present application includes a strategy based on the following three pillars, namely (1) that valve insufficiency must be treated, (2) that no particular therapy can apply to the entire patient population given the various stages of venous insufficiency and the complex etiology thereof, and (3) that no valve can function long-term in the absence of flow (and more specifically shear stress). The present disclosure addresses each pillar.
[0037] The first pillar (namely that valve insufficiency must be treated) is addressed by way of one or more exemplary endograft valve devices.
Shear Enhancing Valve
[0038] An exemplary endograft valve device 100 of the present disclosure is described as follows. As shown in FIG. 3, for example, an exemplary valve device 100 of the present disclosure comprises an endograft body 102 having a first portion 104, a second portion 106, and a valve portion 108. Endograft body 102, in various embodiments, is configured to fit within a luminal organ and further configured for expansion within the luminal organ. The first portion 104 of the endograft body 102, as shown in FIG. 3 in an expanded configuration, has a proximal end 110 defining a proximal end aperture 112 and a distal end 114 defining a distal end aperture 116, the first portion 104 tapering toward distal end 114 so that distal end aperture 116 has a relatively smaller cross-sectional area than proximal end aperture 112. Second portion 106 of the endograft body 102, as shown in FIG. 3 in an expanded configuration, also has a proximal end 118 defining a proximal end aperture 120 and a distal end 122 defining a distal end aperture 124, the second portion 106 tapering toward distal end 122 so that distal end aperture 124 has a relatively smaller cross-sectional area than proximal end aperture 120. As shown in FIG. 3, and in various embodiments of the present disclosure, distal end 114 of first portion 104 is positioned adjacent to (and/or coupled to) proximal end 118 of second portion 106. Valve portion 108, as referenced above and shown in FIGS. 3 and 4 for example, is positioned at or near proximal end 118 of second portion 106 (or coupled to first portion 104 or second portion 106), wherein valve portion 108 is configured to receive fluid flowing through distal end aperture 116 of first portion 104.
[0039] An exemplary endograft valve device 100 of the present disclosure may be positioned within a luminal organ of a patient as follows. In at least one embodiment, and as shown in FIG. 1, a guidewire 200 having an optional compliant end 202 may be inserted into a lumen 250 of a luminal organ 252 of a patient. In at least one embodiment, luminal organ 252 comprises a patient's vein, and an exemplary endograft valve device 100 of the present disclosure is configured to fit within the patient's vein and configured to operate in accordance with the present disclosure.
[0040] Upon insertion and placement of a portion of a guidewire 200 within a luminal organ 252, an exemplary endograft valve device 100 of the present disclosure may be advanced over guidewire 200 to a desired location within luminal organ 252 as shown in FIG. 2. As shown in FIG. 2, endograft valve device 100 is in a first, or collapsed, configuration, and as shown in FIG. 3, endograft valve device 100 is in a second, or expanded, configuration. Endograft valve device 100 may be positioned around and/or coupled to an optional catheter 260, as shown in FIG. 2, to facilitate positioning of endograft valve device 100 within luminal organ 252. Expansion of endograft valve device 100 may be due to movement of guidewire 200, movement of optional catheter 260, inflation of a balloon 702 (as shown in FIG. 7, for example) positioned upon catheter 260, or other mechanisms/procedures known to facilitate expansion of an endograft and/or stent.
[0041] A side view of an exemplary endograft valve device 100 is shown in FIG. 2, while a cross-sectional side view of an exemplary endograft valve device 100 is shown in FIG. 3. Expansion of endograft valve device 100 (from FIG. 2 to FIG. 3) causes at least a portion of endograft valve device 100 to physically contact luminal organ 252 as shown in FIG. 3. As shown in FIG. 3, an outer portion 300 of endograft valve device 100 contacts luminal organ 252, while an inner portion 302 of endograft valve device 100 is configured so that fluid may flow therethrough and through valve portion 108. Outer portion 300 may generally define an outer portion wall 310, and inner portion 302 may generally define an inner portion wall 312, as shown in FIG. 3. In at least one embodiment, outer portion wall 310 and/or inner portion wall 312 may be relative walls, as endograft body 102 may itself comprise a mesh that does not create a formal wall. In such an embodiment, for example, outer portion 300 may define an outer portion relative surface 320, and inner portion 302 may define an inner portion relative surface 322, as shown in FIG. 4. As shown in each of FIGS. 3 and 4, the interior portion of an exemplary endograft valve device 100 is generally referred to as a lumen 350, whereby lumen 350 tapers toward distal end 114 of first portion 104 and again tapers toward distal end 122 of second portion 106.
[0042] FIG. 4 shows an exemplary endograft valve device 100 positioned within a lumen 250 of a luminal organ 252 with guidewire 200 and optional catheter 260 withdrawn. FIG. 4 shows "CSA1" and "CSA2," which are indicative of a first cross-sectional area and a second cross-sectional area, respectively. As shown in FIG. 4, CSA1 is relatively larger than CSA2, with CSA1 being indicative of a proximal end aperture 112 of first portion 104, and with CSA2 being indicative of a distal end aperture 116 of first portion 104. As shown therein, first portion 104 tapers inward toward valve portion 108.
[0043] Valve portion 108, as generally referenced above, is configured in at least one embodiment as a venous valve system. In at exemplary embodiment, valve portion 108 is configured as an effective bicuspid valve system for a vein, noting that the arterial system has substantially higher shear stresses than the venous system. In view of the same, exemplary valve portions 108 of the present disclosure are configured to increase shear stress at the leaflets 500 of valve portion 108 as shown in FIG. 5 so that they effectively operate as venous valves.
[0044] As shown in FIGS. 3-6, exemplary endograft valve devices 100 are provided for potential use within a patient's body instead of a traditional stent that may carry a valve. As generally referenced above and shown in FIGS. 3-6, endograft valve devices 100 are configured to taper endoluminally, either linerally as shown in the figures or in some other fashion, so that blood flowing therethrough will increase in velocity and hence increase the shear stress at valve portion 108.
[0045] Conservation of mass requires that volumetric flow rate (Q) to remain constant throughout the graft (i.e., Q=constant=V*CSA, where V and CSA represent the velocity of blood and luminal cross-sectional area of the endograft). Since the CSA is made, by novel design of the various endograft valve devices 100 of the present disclosure, to decrease towards valve portion 108 (i.e., CSA2<CSA1 as shown in FIG. 4), the velocity will be much larger at valve portion 108. The wall shear stress (WSS, referring to the stresses of inner portion 302 of endograft valve device 100) is related to the CSA as WSS.about.1/CSA3/2 (Poiseuille's law) and hence any decrease in CSA will amplify the WSS. For example, a 10% decrease in diameter (CSA.about.D2) is expected to increase the WSS by 33%.
[0046] The increase in WSS and the corresponding reduction in thrombogenecity at the valve is weighted against the potential increase in the pressure drop (.DELTA.P, as shown in FIG. 5) to overcome the resistance to flow. A 50% decrease in CSA, for example is non-flow limiting (i.e., only a minimal pressure drop). Hence, even at this level of CSA design, the WSS can be nearly tripled (.about.2.8.times.). A reverse tapering on the distal portion of the valve, in at least one embodiment and as shown in FIG. 6, is considered to ensure flow recirculation and hence the establishment of a negative pressure drop to close valve portion 108.
[0047] The present disclosure considers various hemodynamic variables to ensure a correct design that prevents stasis. The third point of the triad, namely changes in the vessel wall as referenced herein, can be considered as various endograft valve devices 100 can be coated similarly to a luminal organ (such as a biological vein, for example) at least acutely until the endograft valve device 100 endothelializes. Such considerations may require that the inner portion 302 (such as inner portion wall 312 or inner portion relative surface 322, for example) of the endograft valve device 100 be at least partially covered or coated one or more anti-clotting agents, such as, for example, heparin, thrombomodulin, endothelial cell protein C, and/or another anti-clotting agent. FIG. 5 shows agent 510 positioned upon endograft valve device 100. In addition to providing an anticoagulant endoluminal surface (such as inner portion wall 312 or inner portion relative surface 322, for example), the same can also be provided to the endograft outer portion 300 (such as outer portion wall 310 or outer portion relative surface 320, for example) to dissolve any existing clots in the luminal organ upon deployment of the endograft valve device 100.
[0048] Various embodiments of endograft valve devices 100 of the present disclosure may be made of one or more standard biologically-compatible materials, such as polytetrafluoroethylene (PTFE), Gore-Tex.RTM., etc., containing, for example, a nickel titanium alloy such as nitinol and/or another memory metal in the endograft body 102 so that the desired memory shape of the endograft valve device 100, in an open or a closed configuration, is accomplished while maintaining the desired hemodynamic effects noted above.
[0049] At least one exemplary embodiment of a valve system 700 of the present disclosure is shown in the block diagram of FIG. 7. As shown in FIG. 7, an exemplary valve system 700 comprises a number of components of an exemplary endograft valve device 100 of the present disclosure, such as endograft body 102, valve portion 108, etc. In addition, an exemplary valve system 700 may comprise one or more components useful to deliver and/or position an exemplary valve device 100 of the present disclosure, including a guidewire 200, a catheter 260, and an optional balloon 702 coupled to or positioned adjacent to catheter 260.
[0050] FIG. 8 shows a diagram of steps of an exemplary method of using an endograft valve device 100 and/or valve system 700 of the present disclosure. As shown in FIG. 8, an exemplary method 800 comprises the steps of introducing a guidewire into a luminal organ (such as a vein) of a patient (an exemplary guidewire insertion step 802) and advancing an endograft valve device 100 along guidewire 200 to a desired location within a patient (an exemplary advancement step 804). Endograft valve device 100 is then expanded as referenced herein (an exemplary expansion step 806), and the guidewire 200 and/or any other device used to deliver endograft valve device 100 (such as a catheter 260, for example), is withdrawn from the area of the endograft valve device 100 (an exemplary removal step 808).
[0051] The second pillar, namely that no particular therapy can apply to the entire patient population given the various stages of venous insufficiency and the complex etiology thereof, is addressed as follows.
Patient-Specific Virtual Venous Valve Simulation
[0052] An analogy can be drawn with mitral valve and heart failure (HF) whose etiology may be of ischemic, electrical, or valvular origin. A number of therapies for HF exist such as revascularization (such as coronary artery bypass graft (CABG) surgery and percutaneous coronary intervention (PCI)), valve replacement, cardiac resynchronization therapy (CRT), use of a left ventricular assist device (LVAD), and the like. Each of these therapies has guidelines for patient selection (inclusion/exclusion criterion). A similar paradigm for patient selection must be established for venous insufficiency in order for a therapy to be effective.
[0053] The disclosure of the present application includes a patient-specific, physics-based approach to determine whether or not the patient is suitable for a potential vein valve procedure. Such an approach, in at least one embodiment, may be useful to develop a validated, patient-specific, physics-based computational model to predict the clinical function of a prosthetic valve replacement device. Existing clinical imaging modality, such as duplex ultrasonography (US) may be used in connection therewith to provide both the venous geometry and flow velocity of a patient.
[0054] An exemplary patient selection method of the present disclosure uses one or more computer models of various venous valves, whereby said valves can be virtually implanted into a model of the specific patient's geometry. Various laws of physics, such as the conservation of mass and momentum, can be used in conjunction with the patient-specific boundary condition (flow velocity) to simulate the entire shear field on the valve leaflets as well as the mechanical stresses and strains in the leaflets and/or other functional surfaces of the devices. Various sample prototype devices may also be tested in said simulations, providing data indicative of the temporal and spatial distributions of the stresses. These simulations would then provide the physical predictions of the expected levels of the mechanical environment of the prototype valves and their propensities for success or failure based, in part, on the various intramural stresses and strains in the valve device materials.
[0055] By way of example, patients that have low shear stresses (i.e., stasis) on the valve leaflets can be excluded since these patients are likely to have a poor outcome. The relation between the cutoff for the various mechanical forces, deformations, and biological responses can be determined through in vivo experiments that include realistic models of venous hypertension and insufficiency (e.g., venous hypertension using an arteriovenous (AV) fistula and occlusion models to perturb flow and shear stress and evaluate the biological response of the valve implant). Hence, various guidelines and criterion for acceptable mechanical regimes in animal studies and from the published literature can be established and used to guide the initial patient experience.
[0056] Exemplary computational platforms for both fluid and solid mechanics of a valve can be obtained using duplex US, an impedance device (as referenced below), or another mechanism useful to obtain geometric and flow data of a valve within a vessel. Such a computer simulation of a flow field and wall stress can be generated for an idealized valve with idealized geometries and flows, and said platforms can then be repeated for specific patients, so that flows and stresses representative of a specific patient can be provided.
[0057] As can be identified by such a platform, idealized valves prominently show regions of stagnant flow in the base region of the valve under the provided flow conditions, as well as showing flow alterations at the hinge region of leaflets. Flow rates and direction can then be indicated using a series of arrows to show direction of flow (via arrow point) within a vessel and rate of flow (as given by the overall length of the arrows). A longer arrow denotes a faster flow rate. Wall stresses can be shown using, for example, various colors or pixel concentrations to show areas with higher stresses (generally at or near the valve leaflets) and areas of lower stresses (generally away from the valve leaflets).
[0058] To minimize ad hoc assumptions in the simulations, accurate data would be established based upon the anatomy (geometry) and material properties of both the potentially-used prosthetic valve and the vein itself. Furthermore, and in addition to duplex US, additional technologies (such as impedance wires and catheters, for example) can be leveraged to determine both the size of the vein to match the prosthesis (to prevent migration) as well as compliance of the vessel (for accurate simulation of vein wall motion and coupling to blood flow).
[0059] As such, the various virtual simulations used to test various device/valve designs would allow researchers to design and redesign such devices until the devices/valves have the desired functionality. Specific devices/valves, including those specific to venous diseases, can be optimized so that their use in vivo would be effective for any number of luminal organ needs.
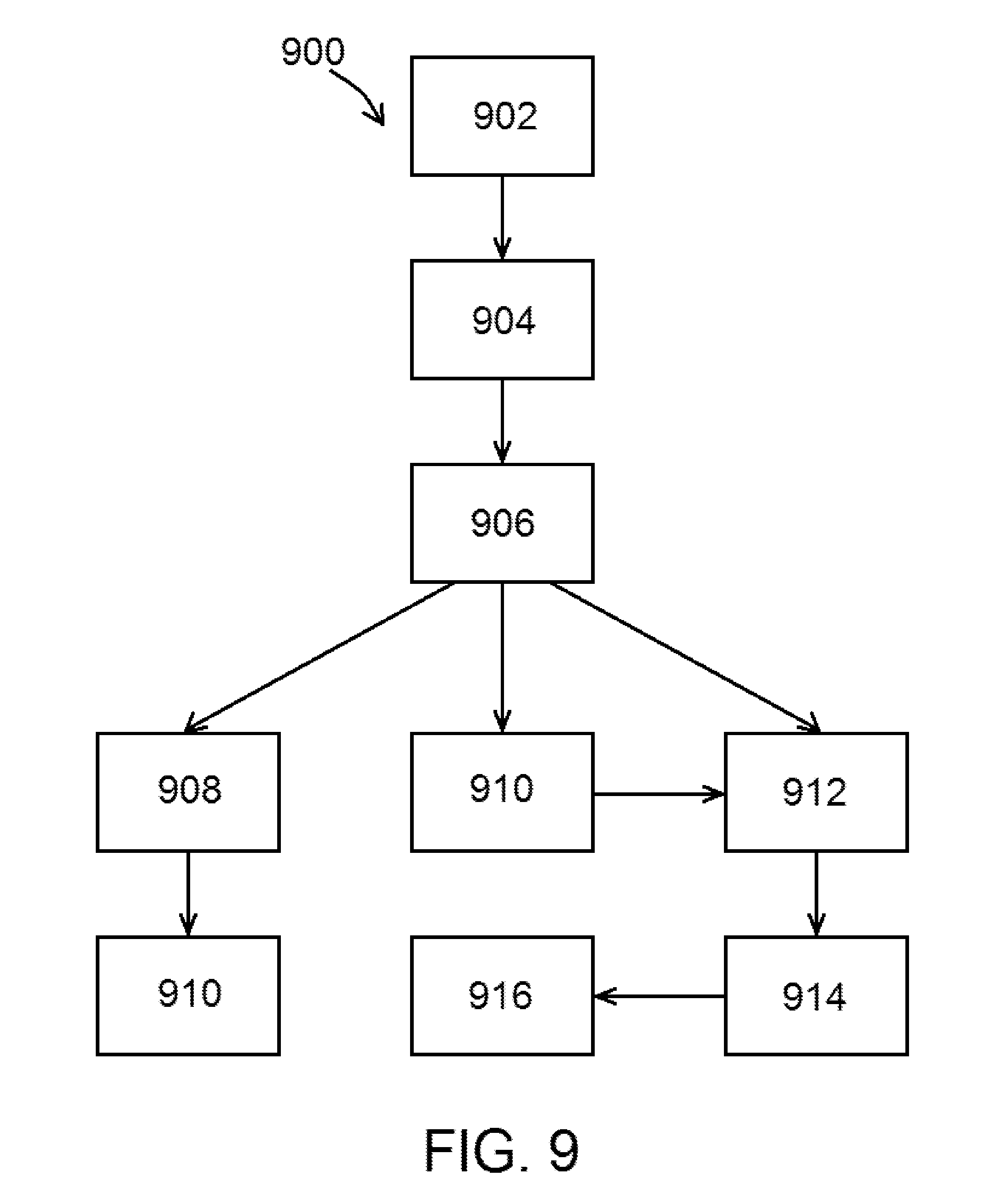
[0060] Accordingly, the present disclosure includes disclosure of a method of determining whether a patient is suitable for a valve procedure. In at least one embodiment of a method 900, as indicated by the method steps shown in FIG. 9, method 900 comprises the steps of obtaining data indicative to a patient's venous geometry at a first location (an exemplary first data obtaining step 902) and obtaining data indicative of a flow velocity of blood within a vein of the patient at or near the first location (an exemplary second data obtaining step 904), wherein steps 902 and 904 can be performed in either order. Method 900, in various embodiments, further comprises the steps of preparing a digital model of the vein of the patient using the data indicative to the patient's venous geometry and/or the data indicative of the flow velocity to obtain patient venous data (an exemplary digital model preparation step 906), and determining whether the patient is suitable for a valve procedure based at least in part of the patient venous data (an exemplary determination step 908).
[0061] In at least one embodiment, steps 902 and/or 904 is/are performed using duplex ultrasonography. In various embodiments, steps 902 and/or 904 is/are performed using an impedance device, such as an impedance wire and an impedance catheter. An exemplary impedance device may comprise a device body and at least four electrodes positioned thereon, wherein the at least four electrodes comprising two electrodes configured to excite an electric field and two electrodes configured to obtain a conductance measurement within the electric field.
[0062] In various embodiments of methods 900 of the present disclosure, digital model preparation step 906 is performed using the data indicative to the patient's venous geometry and the data indicative of the flow velocity to obtain patient venous data. In other embodiments, digital model preparation step 906 is performed using the data indicative to the patient's venous geometry to prepare a first digital model and the data indicative of the flow velocity to prepare a second digital model, wherein the patient venous data is indicative of the first digital model and the second digital model.
[0063] In at least one embodiment of a method 900 of the present disclosure, and as shown in FIG. 9, method 900 further comprises the step of incorporating data indicative of at least one valve into the digital model (an exemplary valve data incorporation step 910). Step 910 may be performed by incorporating native valve data or prosthetic valve data therein. Upon performance of step 910, in at least one embodiment, determination step 908 can be performed based at least in part on information including the patient venous data and the data indicative of at least one valve and/or patient venous data indicative of flow and shear stress. Further and in at least one embodiment, an ultimate determination can be made based upon a comparison of the patient venous data indicative of flow and shear stress to at least one threshold.
[0064] An exemplary method 900, as shown in FIG. 9, may comprise the step of testing one or more virtual valve device configurations in at least one simulation using the digital model prepared in digital model preparation step 906 (an exemplary valve testing step 912). In at least one embodiment, the device configurations can be incorporated into the digital model such as by valve data incorporation step 910 referenced above. Valve testing step 912, as shown in FIG. 9, may be performed after digital model preparation step 906 and optionally after valve data incorporation step 910, and involves performing at least one simulation using the digital model. Determination step 908 and valve testing step 912 may be generally referred to herein as a test purpose.
[0065] Valve testing step 912, in various embodiments, may be performed to determine whether or not a virtual valve model is suitable for the patient that the digital model is based upon. For example, if a virtual valve model is tested in valve testing step 912 and it does not perform optimally, the virtual valve model can be modified and retested. In view of the same, an exemplary method 900 of the present disclosure may further comprise the steps of adjusting one or more virtual valve device configurations and retesting the adjusted one or more virtual valve device configurations (an exemplary adjustment step 914). Should steps 912 or 914 yield satisfactory results (indicative of a suitable virtual valve for the patient), a physical valve device based upon at least one of the adjusted virtual valve device configurations may be prepared (an exemplary valve preparation step 916), whereby the prepared valve (such as an endograft valve device 100 and/or valve system 700 of the present disclosure) may be positioned within the patient consistent with an exemplary method 800 of the present disclosure, for example.
[0066] Steps 912, 914, and/or 916 may be performed, as referenced above, to test, optimize, and produce a valve (such as a vein valve device) best suited for the modeled patient. The virtual testing, as referenced herein, allows for dozens, if not hundreds or thousands or more, of virtual devices to be tested, saving significant time and money over traditional physical valve manufacture and testing. In addition, such a method, as referenced above, is patient-specific, allowing for an optimized valve device, specifically tailored for the patient in need of the valve procedure (such as a valve replacement or valve insertion where no valve is present), resulting in optimal patient treatment. Such valve optimization (by way of performing steps 912 and/or 914) may be done to change one or more parameters, such as valve length, width, wall thickness, leaflet size, leaflet configuration, leaflet number(s), materials, curvatures, and/or a combination of the foregoing, for example, to optimize a valve for that particular patient. Such an optimized valve (ultimately produced in step 916, for example), may have one or more of the desired hemodynamic, mechanical, and/or functional properties sufficient for that particular patient's needs. For example, such an optimized valve may have two or more leaflets instead of one leaflet, and may be optimized so that a minimum amount of energy (or a reduced amount of energy) is needed to open and/or close the valve. Other valve configurations may be preferred based upon a different patient digital model.
[0067] In various embodiments, determination step 908 is based at least in part on the patient venous data and the data indicative of at least one valve. In other embodiments, determination step 908 is based upon at least patient venous data indicative of flow and shear stress, and wherein an ultimate determination is based upon a comparison of the patient venous data indicative of flow and shear stress to at least one threshold.
[0068] Such a tailored approach defines the range of in vivo performance of the valve in a patient-specific mechanical environment. Although there are factors beyond mechanics that can be considered, a mechanical approach, and the data emanating therefrom, allows a practitioner to potentially identify non-mechanical factors. For example, if the valve fails in a percentage of the patients that satisfy mechanically-based inclusion criteria, then additional, non-mechanical biomarkers may be identified (such as blood chemistry, risk factors, co-morbidities, etc.). This data will then form the basis of a systematic and rigorous approach to embracing this complex patient population.
[0069] The third pillar, namely that no valve can function long-term in the absence of flow (and more specifically shear stress, is addressed as follows.
Venous Return Assist Device
[0070] Transcatheter aortic valve implantation (TAVI) has been successful, in large part, because the aortic valve prosthesis is coupled with the heart, namely that the heart pump ensures sufficient flow through the prosthetic. However, this is limited to aortic valves, and does not apply to venous valves, as the heart pump cannot ensure sufficient flow through a venous valve.
[0071] No valve can function without flow. The disclosure of the present application includes methods to generate venous blood flow (propulsion) through various assist devices (compression) and suction (release) within the vein, such as within the abdominal vena cavae. As discussed in further detail herein, said devices and methods can be used with our without valve implants, as if a native valve is functional, said devices and methods can be used to facilitate blood flow therethrough.
[0072] Under physiologic conditions, the peripheral pump (skeletal muscle), as well as the respiratory and abdominal phasic pressures, work in conjunction with the compliant veins to assist venous return in the presence of valves. Unfortunately, in patients of interest, venous hypertension-induced remodeling, thrombosis or fibrosis reduces the compliance of the veins (thicker and stiffer) and compromises the normal venous assist mechanisms. In such patients, an active assist mechanism for venous return is needed.
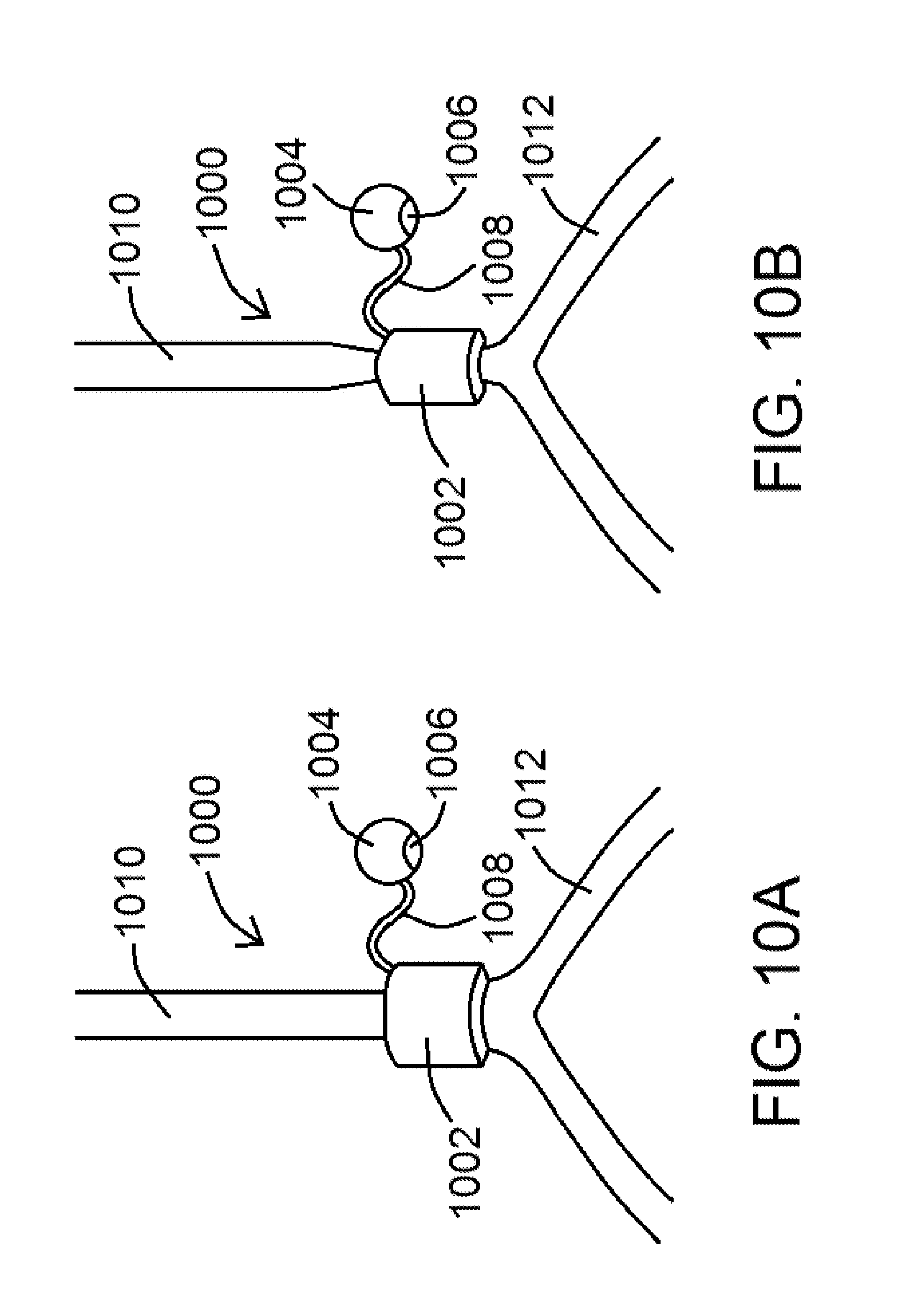
[0073] The present disclosure includes disclosure of an exemplary external assist device operable to impose a force directly on the external surface of the vein to overcome the increased stiffness of the vein. As shown in FIGS. 10A and 10B, an exemplary assist device 1000 of the present disclosure comprises a cuff 1002 configured to fit around a blood vessel and further configured to periodically compress the blood vessel. Devices 1000, in various embodiments, are operably by way of a power source 1004 operably coupled to cuff 1002, so that power from power source 1004 can control the compression and relaxation of cuff 1002. Power source 1004 may comprise, for example, an implanted battery, which may be rechargeable, and/or a power source 1004 positioned external to the patient's body. A processor 1006, operably coupled to power source 1004, is configured to control the rates of compression and relaxation of cuff 1002. In at least one embodiment, processor 1006 is configured so that the compression rate and the relaxation rate can be changed to a different compression rate and a different relaxation rate. As shown in FIGS. 10A and 10B, an optional connector 1008, such as a wire, may be used to connect power source 1004 to cuff 1002, wherein connector 1008 configured to allow power from power source 1004 to be transmitted therethrough to cuff 1002.
[0074] FIG. 10A shows an exemplary assist device 1000 of the present disclosure positioned around an abdominal vena cava (an exemplary blood vessel 1010). Blood flowing from an iliac vein 1012, for example, would flow through the vena cava and be assisted using an exemplary assist device 1000. Device 1000 is shown in FIG. 10A as positioned around, but not compressing, the vena cava, while an exemplary device 1000 of the present disclosure is shown in FIG. 10B positioned around and compressing the vena cava. Devices 1000 of the present disclosure are not limited to being configured around a vena cava, as said devices 1000 may be configured to fit around any number of blood vessels within a mammalian body.
[0075] In at least one embodiment of a device 1000 of the present disclosure, when device 1000 is positioned around a blood vessel at a first location, operation of processor 1006 causes cuff 1002 to alternately compress the blood vessel and relax compression of the blood vessel. Processor 1006 controls a compression rate and a relaxation rate (which may be the same or different), whereby relaxation at the relaxation rate causes blood to move through the blood vessel at the first location. In at least one embodiment, the compression rate is slower than the relaxation rate, as a relatively faster relaxation rate allows the blood vessel to open quicker and effectively pull blood through the blood vessel at the first location. A power source 1004 operably coupled to cuff 1002 would be configured to provide power to cuff 1002 and/or processor 1006 to facilitate compression and relaxation of cuff 1002.
[0076] In various embodiments of devices 1000, processor 1006 is configured so that the compression rate and the relaxation rate can be changed to a different compression rate and a different relaxation rate. In at least one embodiment, when device 1000 is positioned distal to a blood vessel valve (such as an endograft valve device 100 as referenced herein), operation of device 1000 causes blood to flow through the vessel valve toward device 1000. The blood vessel valve may be a native valve or a prosthetic valve, as devices 1000 of the present disclosure are configured to facilitate blood flow through both types of valves.
[0077] In at least one embodiment, when device 1000 is positioned around the blood vessel at a first location, the blood flows through the blood vessel at the first location a first rate without operation of device 100, and the blood flows through the blood vessel at the first location at a second rate during operation of device 1000, wherein the second rate is faster than the first rate. Furthermore, and in various embodiments, when device 1000 is positioned around the blood vessel at a first location, the blood flows through the blood vessel at the first location a first rate range without operation of device 100, and the blood flows through the blood vessel at the first location at a second rate range during operation of device 1000, wherein the second rate range has a faster top rate than the first rate range. The two ranges include the slowest relative flow rate, the fastest relative flow rate, and potentially various flow rates in between.
[0078] FIG. 11 shows a larger view of an exemplary device 1000 of the present disclosure positioned around a blood vessel 1010, and further shows an exemplary endograft valve device 100 positioned within a lumen 1100 of a blood vessel 1012 proximal to where device 1000 is positioned. As shown in FIG. 11, device 1000 is positioned about a vena cava, while device 100 is positioned within an iliac vein 1012 proximal to device 1000. Operation of device 1000, when positioned as shown in FIG. 11 relative to device 100, causes blood to be pulled through vessel 1010 at the location of device 1000, and therefore causes blood to be pulled through device 100 proximal to device 1000.
[0079] Use of various devices 1000 of the present disclosure provides an assist mechanism where compression of the vein propels the blood flow (in the presence of functional valve, either native or prosthetic) towards the heart. A quick release of the compression of device 1000 can create the effect of suction to "pull" the blood from the periphery again in the direction of the heart. Other devices, such as the devices disclosed within US2010/0179376 of Kassab and Navia, may also be configured to fit around a vessel (such as a vein, as referenced herein) and further configured to compress and release the blood vessel so to, for example, facilitate blood flow through a vein when the device is positioned around the vein. Said devices are hereby incorporated into the present disclosure by reference.
[0080] The present disclosure also includes disclosure of a method of facilitating blood flow through a blood vessel. In at least one embodiment of a method 1200 of the present disclosure, as shown by the method steps in FIG. 12, the method comprises the steps of positioning an exemplary external assist device 1000 of the present disclosure around a blood vessel (an exemplary positioning step 1202), and operating external assist device 1000 to alternatively compress the blood vessel and relax compression of the blood vessel (an exemplary operating step 1204), wherein relaxation of compression causes blood to flow through the blood vessel. Relaxation of compression of device 1000, as referenced herein, causes blood to flow through the blood vessel at a faster rate than a native blood flow rate. In at least one embodiment of method 1200 of the present disclosure, operating step 1204 comprises operating external assist device 1000 to alternative compress the blood vessel at a first rate and to relax compression of the blood vessel at a second rate, wherein the second rate is faster than the first rate.
[0081] Various devices 1000 of the present disclosure may be delivered minimally invasively through a laparoscopic approach to induce a pumping action to propel the flow forward in opposition to gravity.
[0082] While various embodiments of devices and methods for assisting valve function, replacing venous valves, and predicting valve treatment successes been described in considerable detail herein, the embodiments are merely offered as non-limiting examples of the disclosure described herein. It will therefore be understood that various changes and modifications may be made, and equivalents may be substituted for elements thereof, without departing from the scope of the present disclosure. The present disclosure is not intended to be exhaustive or limiting with respect to the content thereof.
[0083] Further, in describing representative embodiments, the present disclosure may have presented a method and/or a process as a particular sequence of steps. However, to the extent that the method or process does not rely on the particular order of steps set forth therein, the method or process should not be limited to the particular sequence of steps described, as other sequences of steps may be possible. Therefore, the particular order of the steps disclosed herein should not be construed as limitations of the present disclosure. In addition, disclosure directed to a method and/or process should not be limited to the performance of their steps in the order written. Such sequences may be varied and still remain within the scope of the present disclosure.
* * * * *
D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.