Methods Of Applying Printable Lithium Compositions For Forming Battery Electrodes
Fitch; Kenneth Brian ; et al.
U.S. patent application number 16/359725 was filed with the patent office on 2019-07-11 for methods of applying printable lithium compositions for forming battery electrodes. The applicant listed for this patent is FMC Lithium USA Corp.. Invention is credited to Kenneth Brian Fitch, William Arthur Greeter, JR., Jian Xia, Marina Yakovleva.
| Application Number | 20190214631 16/359725 |
| Document ID | / |
| Family ID | 67139926 |
| Filed Date | 2019-07-11 |
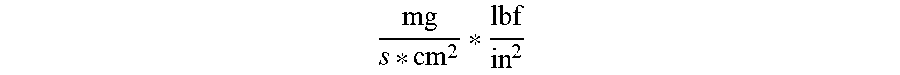

| United States Patent Application | 20190214631 |
| Kind Code | A1 |
| Fitch; Kenneth Brian ; et al. | July 11, 2019 |
METHODS OF APPLYING PRINTABLE LITHIUM COMPOSITIONS FOR FORMING BATTERY ELECTRODES
Abstract
A method for depositing lithium on a substrate to form an electrode is provided. The method includes applying a printable lithium composition comprised of lithium metal powder, a polymer binder compatible with the lithium metal powder, a rheology modifier compatible with the lithium metal powder and a solvent compatible with the lithium metal powder and with the polymer binder, to a substrate.
| Inventors: | Fitch; Kenneth Brian; (Cherryville, NC) ; Yakovleva; Marina; (Gastonia, NC) ; Greeter, JR.; William Arthur; (Dallas, NC) ; Xia; Jian; (Belmont, NC) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 67139926 | ||||||||||
| Appl. No.: | 16/359725 | ||||||||||
| Filed: | March 20, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 62464521 | Feb 28, 2017 | |||
| 62691819 | Jun 29, 2018 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | H01M 10/052 20130101; H01M 10/0585 20130101; H01M 2004/027 20130101; H01M 10/0525 20130101; H01M 4/139 20130101; H01M 4/0404 20130101; H01M 4/0409 20130101; H01M 4/0414 20130101; H01M 4/485 20130101; H01M 4/364 20130101; H01M 4/622 20130101; H01M 4/134 20130101; H01M 4/0411 20130101; H01M 10/0565 20130101; H01M 4/602 20130101; H01M 4/382 20130101; H01M 4/624 20130101; H01M 4/1395 20130101 |
| International Class: | H01M 4/1395 20060101 H01M004/1395; H01M 4/38 20060101 H01M004/38; H01M 4/62 20060101 H01M004/62; H01M 4/134 20060101 H01M004/134; H01M 4/04 20060101 H01M004/04; H01M 10/0585 20060101 H01M010/0585; H01M 10/052 20060101 H01M010/052 |
Claims
1. A method for depositing lithium on a substrate to form an electrode comprising applying a printable lithium composition comprised of lithium metal powder, a polymer binder compatible with the lithium metal powder, a rheology modifier compatible with the lithium metal powder and a solvent compatible with the lithium metal powder and with the polymer binder, to a substrate.
2. The method of claim 1, wherein applying the printable lithium composition to the substrate comprises printing the printable lithium composition onto the substrate with a slot die print head.
3. The method of claim 2 further including loading the printable lithium composition to the print head.
4. The method of claim 2 further including loading the printable lithium composition into a cartridge, loading the cartridge into a dispenser attached to a slot die print head, and mounting the slot die head onto a printer for printing the printable lithium composition onto the substrate.
5. The method of claim 1, wherein applying the printable lithium composition to the substrate comprises extruding the printable lithium composition onto the substrate.
6. The method of claim 5, wherein extruding the printable lithium composition onto the substrate comprises inserting the printable lithium composition into an extruder and extruding the printable lithium composition out of a nozzle.
7. The method of claim 5, wherein extruding the printable lithium composition onto the substrate comprises extruding the printable lithium composition onto the substrate as a pattern.
8. The method of claim 1, wherein applying the printable lithium composition comprises applying coating to a roller and passing the substrate through the roller.
9. The method of claim 1, wherein applying the printable lithium composition to the substrate comprises applying the printable lithium composition onto an anode.
10. The method of claim 9 further including the step of forming a battery comprised of the printed anode.
11. The method of claim 9, wherein applying the printable lithium composition onto an anode comprises applying the printable lithium composition onto a current collector at a thickness between about 10 microns to about 200 microns.
12. The method of claim 1, wherein applying the printable lithium composition to the substrate comprises applying the printable lithium composition as a monolithic coating forming an anode for a solid-state battery.
13. The method of claim 1, wherein applying the printable lithium composition to the substrate comprises applying the printable lithium composition to a solid electrolyte for a solid-state battery.
14. The method of claim 1, wherein the printable lithium composition includes a solvent compatible with the lithium powder and with the polymer binder.
15. A method for depositing lithium on a substrate to form an electrode comprising printing a printable lithium composition comprised of lithium metal powder, a polymer binder compatible with the lithium metal powder, a rheology modifier compatible with the lithium powder and a solvent compatible with the lithium powder and with the polymer binder, to a substrate with a slot die print head.
16. The method of claim 15 further including loading the printable lithium composition into a cartridge, loading the cartridge into a dispenser attached to a slot die print head, and mounting the slot die head onto a printer for printing the printable lithium composition onto the substrate.
17. The method of claim 15, wherein applying the printable lithium composition to the substrate comprises applying the printable lithium composition onto an anode, cathode, solid electrolyte, current collector, or carrier material.
18. The method of claim 17 further including the step of forming a battery comprised of the anode, cathode, solid electrolyte, current collector, or carrier material.
19. A method for depositing lithium on a substrate to form an electrode comprising extruding a printable lithium composition comprised of lithium metal powder, a polymer binder compatible with the lithium metal powder, a rheology modifier compatible with the lithium powder and the polymer binder, and a solvent compatible with the lithium powder and with the polymer binder, onto a substrate.
20. The method of claim 19, wherein extruding the printable lithium composition onto the substrate comprises inserting the printable lithium composition into an extruder and extruding the printable lithium composition out of a nozzle.
21. The method of claim 19, wherein extruding the printable lithium composition onto the substrate comprises extruding the printable lithium composition onto the substrate as a pattern.
22. The method of claim 19, wherein applying the printable lithium composition to the substrate comprises applying the printable lithium composition onto an anode, cathode, solid electrolyte, current collector, or carrier material.
23. The method of claim 22 further including the step of forming a battery comprised of the anode, cathode, solid electrolyte, current collector, or carrier material.
24. A method for depositing lithium on a substrate to form an electrode comprising roller coating a substrate with a printable lithium composition comprised of lithium metal powder, a polymer binder compatible with the lithium metal powder, a rheology modifier compatible with the lithium powder and the polymer binder, and a solvent compatible with the lithium powder and with the polymer binder.
25. The method of claim 24, wherein roller coating the substrate comprises applying coating to a roller and passing the substrate over the roller.
26. The method of claim 24, wherein applying the printable lithium composition to the substrate comprises applying the printable lithium composition onto an anode, cathode, solid electrolyte, current collector, or carrier material.
27. The method of claim 26 further including the step of forming a battery comprised of the anode, cathode, solid electrolyte, current collector, or carrier material.
Description
RELATED APPLICATION
[0001] The following application claims priority to U.S. Provisional No. 62/646,521 filed Mar. 22, 2018, and U.S. Provisional No. 62/691,819 filed Jun. 29, 2018, the disclosures of which are incorporated by reference in their entireties.
FIELD OF THE INVENTION
[0002] The present invention relates to a method for applying a printable lithium composition suitable for formation of electrodes suitable for use in a wide variety of energy storage devices, including batteries and capacitors.
BACKGROUND OF THE INVENTION
[0003] Lithium and lithium-ion secondary or rechargeable batteries have found use in certain applications such as in cellular phones, camcorders, and laptop computers, and even more recently, in larger power application such as in electric vehicles and hybrid electric vehicles. It is preferred in these applications that the secondary batteries have the highest specific capacity possible but still provide safe operating conditions and good cyclability so that the high specific capacity is maintained in subsequent recharging and discharging cycles.
[0004] Although there are various constructions for secondary batteries, each construction includes a positive electrode (or cathode), a negative electrode (or anode), a separator that separates the cathode and anode, an electrolyte in electrochemical communication with the cathode and anode. For secondary lithium batteries, lithium ions are transferred from the anode to the cathode through the electrolyte when the secondary battery is being discharged, i.e., used for its specific application. During the discharge process, electrons are collected from the anode and pass to the cathode through an external circuit. When the secondary battery is being charged, or recharged, the lithium ions are transferred from the cathode to the anode through the electrolyte.
[0005] Historically, secondary lithium batteries were produced using non-lithiated compounds having high specific capacities such as TiS.sub.2, MoS.sub.2, MnO2, and V.sub.2O.sub.5, as the cathode active materials. These cathode active materials were coupled with a lithium metal anode. When the secondary battery was discharged, lithium ions were transferred from the lithium metal anode to the cathode through the electrolyte. Unfortunately, upon cycling, the lithium metal developed dendrites that ultimately caused unsafe conditions in the battery. As a result, the production of these types of secondary batteries was stopped in the early 1990s in favor of lithium-ion batteries.
[0006] Lithium-ion batteries typically use lithium metal oxides such as LiCoO.sub.2 and LiNiO.sub.2 as cathode active materials coupled with an active anode material such as a carbon-based material. It is recognized that there are other anode types based on silicon oxide, silicon particles and the like. In batteries utilizing carbon-based anode systems, the lithium dendrite formation on the anode is substantially avoided, thereby making the battery safer. However, the lithium, the amount of which determines the battery capacity, is totally supplied from the cathode. This limits the choice of cathode active materials because the active materials must contain removable lithium. Also, delithiated products corresponding to Li.sub.xCoO.sub.2, Li.sub.xNiO.sub.2 formed during charging and overcharging are not stable. In particular, these delithiated products tend to react with the electrolyte and generate heat, which raises safety concerns.
[0007] New lithium-ion cells or batteries are initially in a discharged state. During the first charge of lithium-ion cell, lithium moves from the cathode material to the anode active material. The lithium moving from the cathode to the anode reacts with an electrolyte material at the surface of the graphite anode, causing the formation of a passivation film on the anode. The passivation film formed on the graphite anode is a solid electrolyte interface (SEI). Upon subsequent discharge, the lithium consumed by the formation of the SEI is not returned to the cathode. This results in a lithium-ion cell having a smaller capacity compared to the initial charge capacity because some of the lithium has been consumed by the formation of the SEI. The partial consumption of the available lithium on the first cycle reduces the capacity of the lithium-ion cell. This phenomenon is called irreversible capacity and is known to consume about 10% to more than 20% of the capacity of a lithium ion cell. Thus, after the initial charge of a lithium-ion cell, the lithium-ion cell loses about 10% to more than 20% of its capacity.
[0008] One solution has been to use stabilized lithium metal powder to pre-lithiate the anode. For example, lithium powder can be stabilized by passivating the metal powder surface with carbon dioxide such as described in U.S. Pat. Nos. 5,567,474, 5,776,369, and 5,976,403, the disclosures of which are incorporated herein in their entireties by reference. The CO.sub.2-passivated lithium metal powder can be used only in air with low moisture levels for a limited period of time before the lithium metal content decays because of the reaction of the lithium metal and air. Another solution is to apply a coating such as fluorine, wax, phosphorus or a polymer to the lithium metal powder such as described in U.S. Pat. Nos. 7,588,623, 8,021,496, 8,377,236 and U.S. Patent Publication No. 2017/0149052, for example.
[0009] There, however, remains a need for processes and compositions for applying lithium metal powder to various substrates to provide electrodes for capacitors, lithium-ion cells and other lithium metal batteries.
SUMMARY OF THE INVENTION
[0010] To this end, the present invention provides methods for depositing lithium on a substrate to form an electrode by applying a printable lithium composition to the substrate. The printable lithium composition may be applied by various means, including printing, extruding, spraying, and coating.
[0011] The printable lithium composition of the present invention comprises a lithium metal powder, a polymer binder, wherein the polymer binder is compatible with the lithium powder, and a rheology modifier compatible with the lithium powder and the polymer binder. A solvent may be included in the printable lithium composition, wherein the solvent is compatible with the lithium powder and compatible with (e.g., able to form suspension or to dissolve in) the polymer binder. The solvent may be included as a component during the initial preparation of the printable lithium composition, or added later after the printable lithium composition is prepared.
BRIEF DESCRIPTION OF THE DRAWINGS
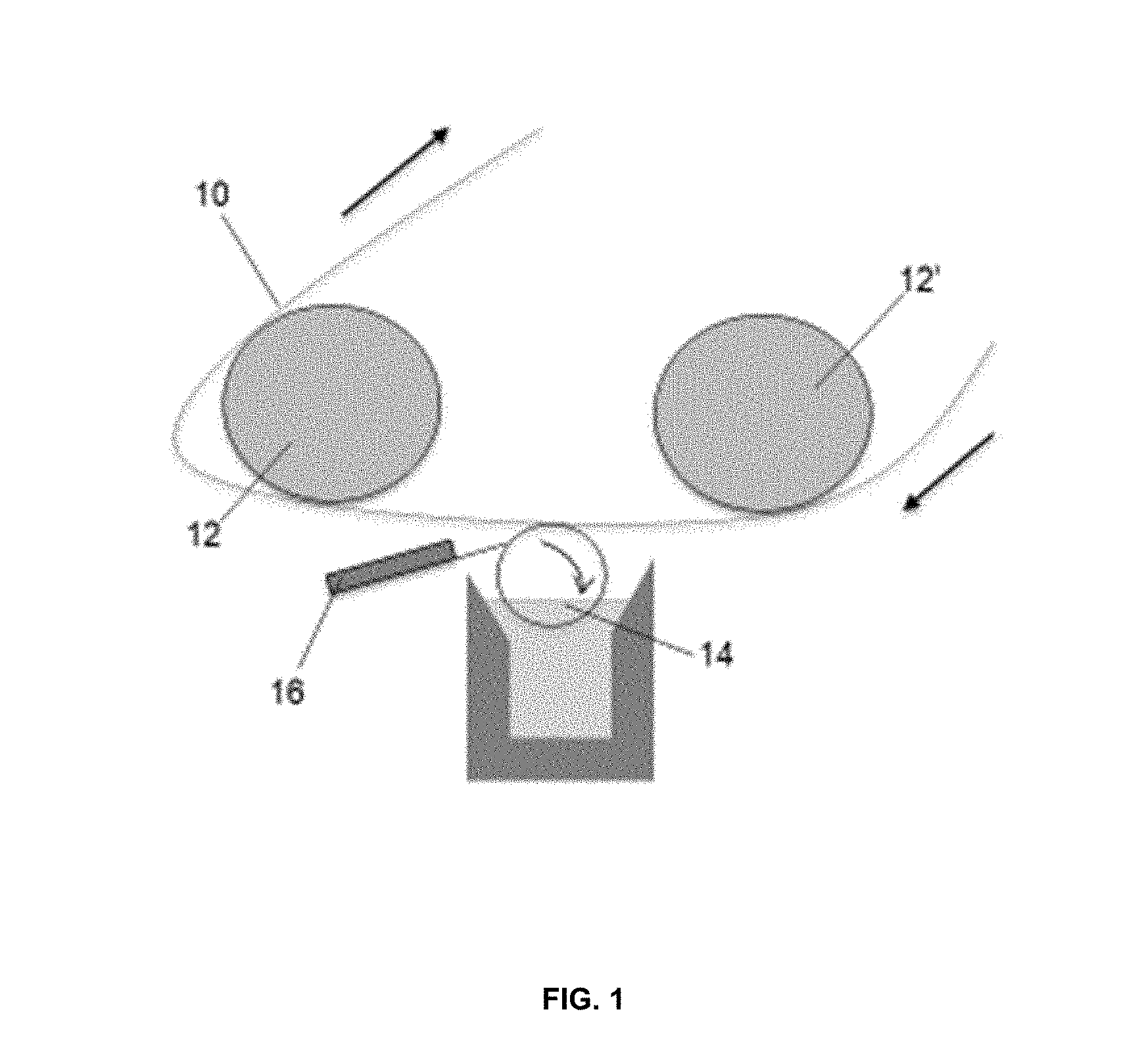
[0012] FIG. 1 is a schematic of one embodiment of a printable lithium composition coated onto a substrate;
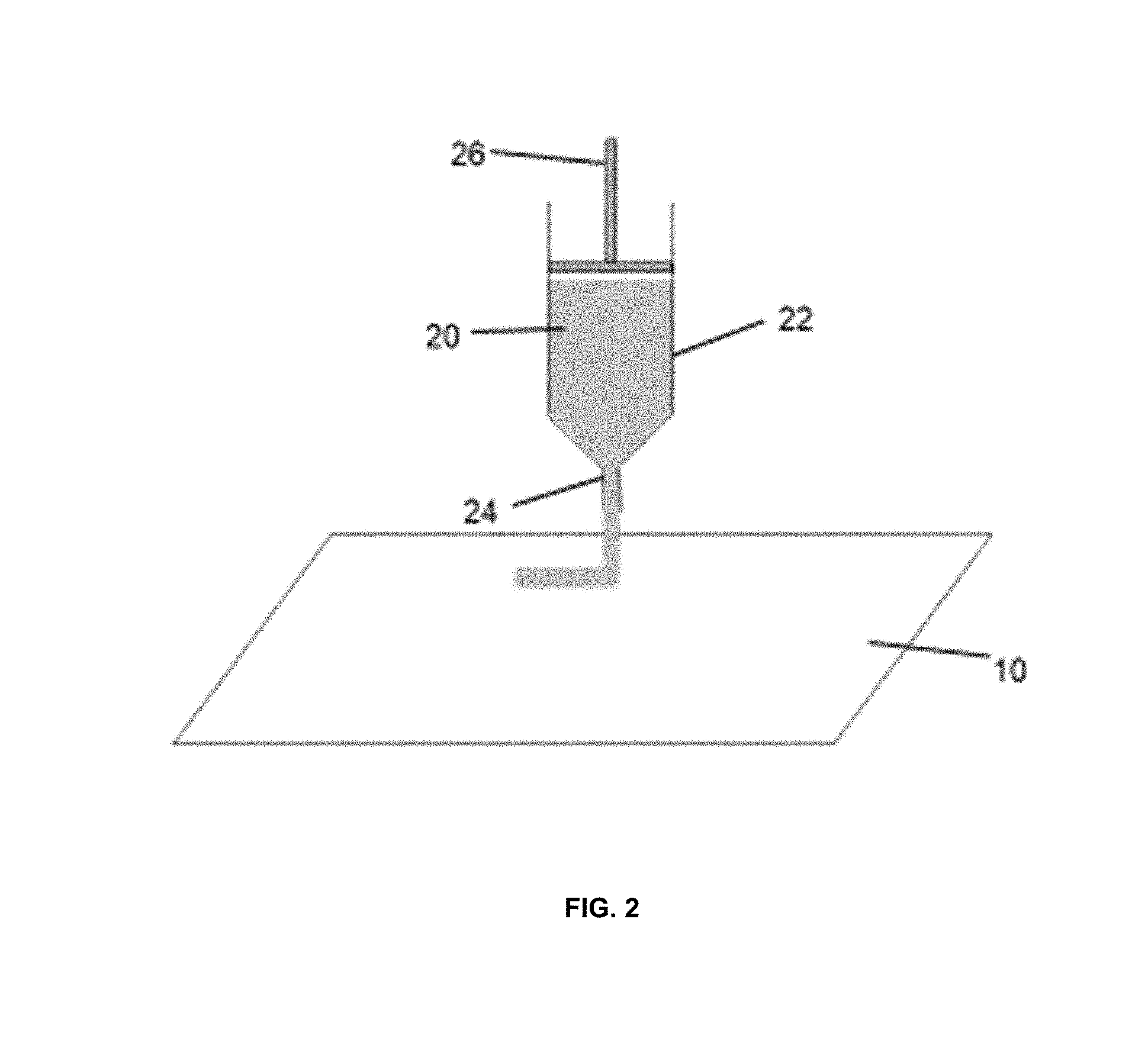
[0013] FIG. 2 is a schematic of one embodiment of a printable lithium composition extruded onto a substrate;
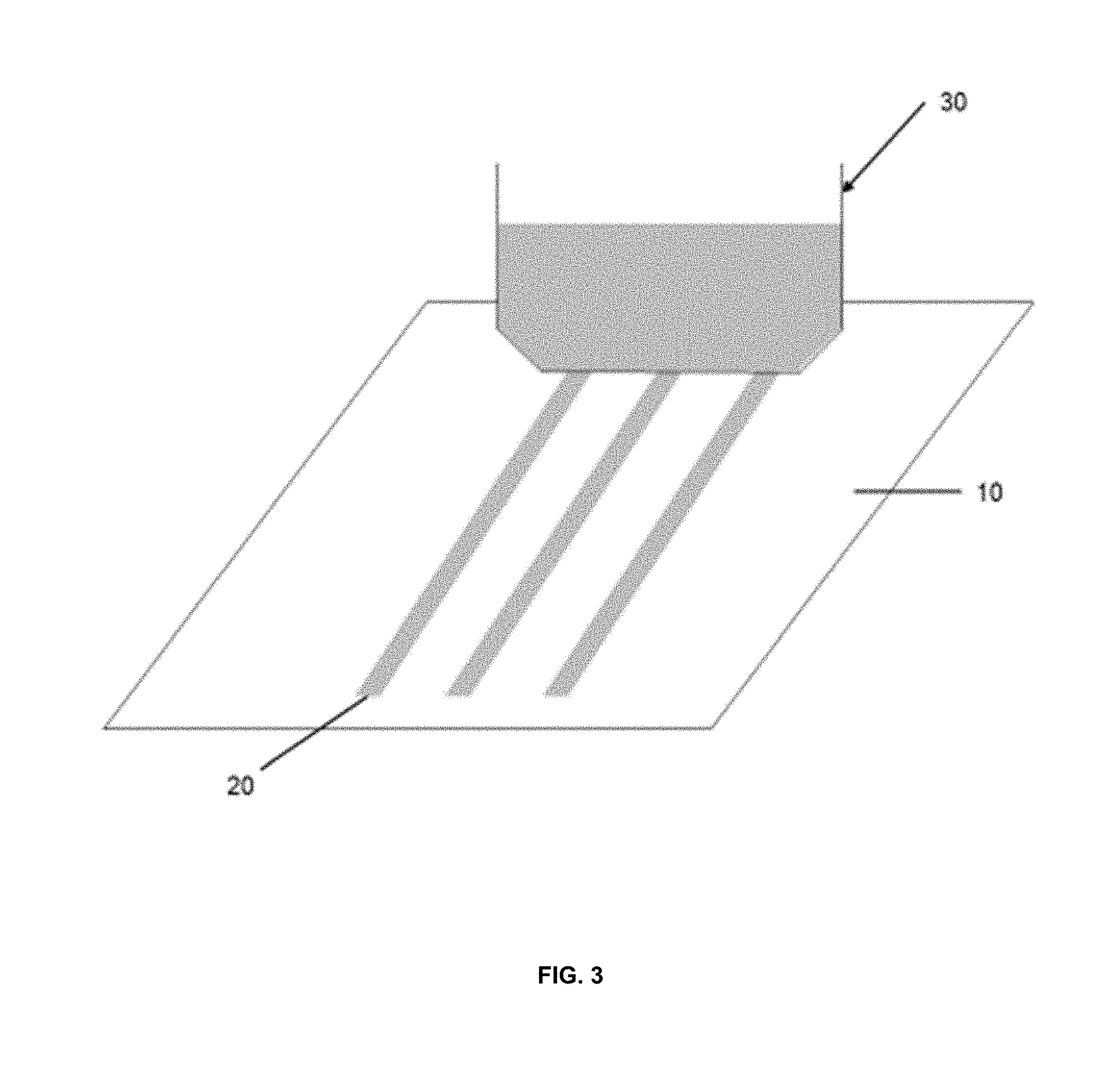
[0014] FIG. 3 is a schematic of one embodiment of a printable lithium composition printed onto a substrate using a slot die;
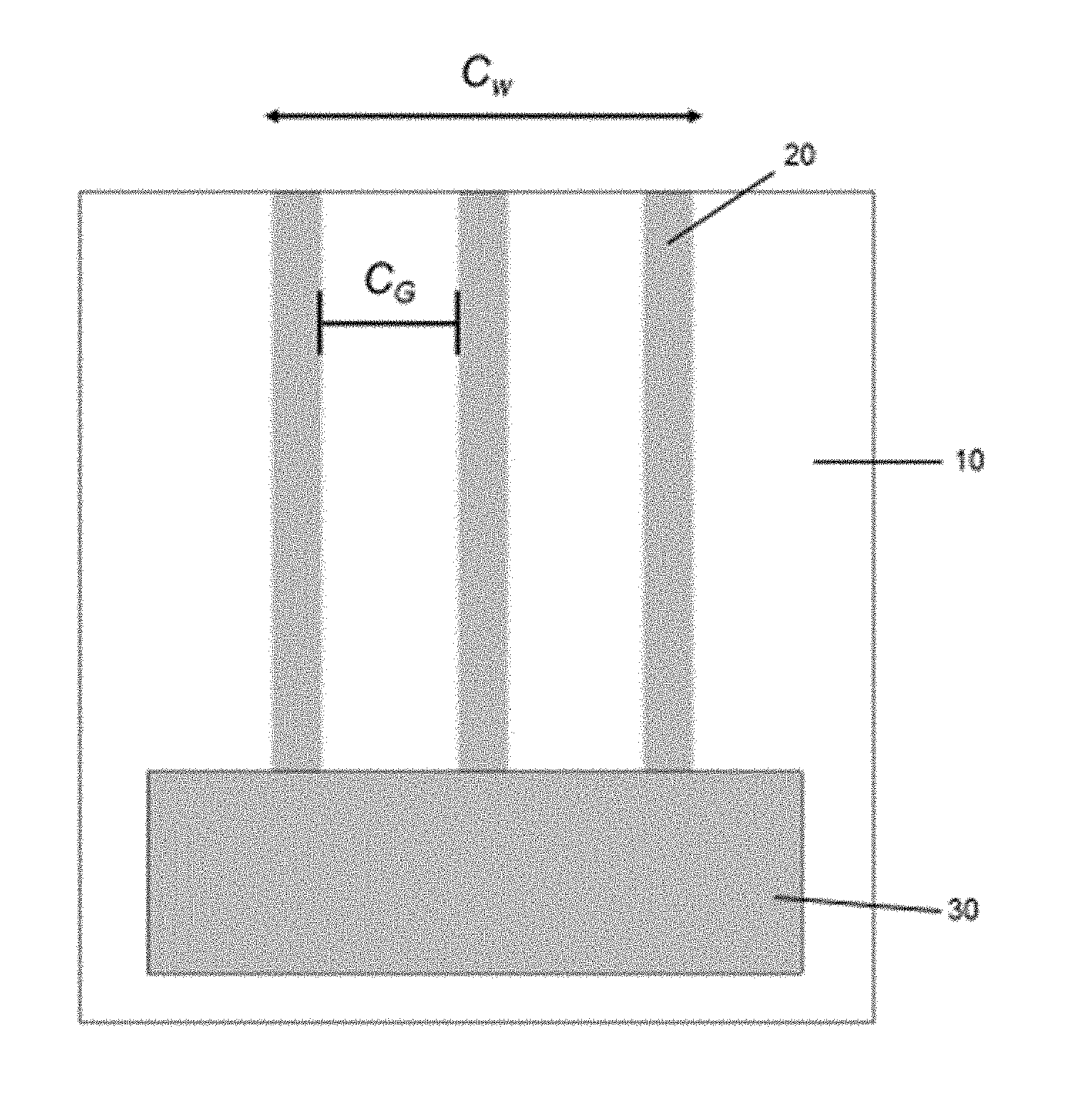
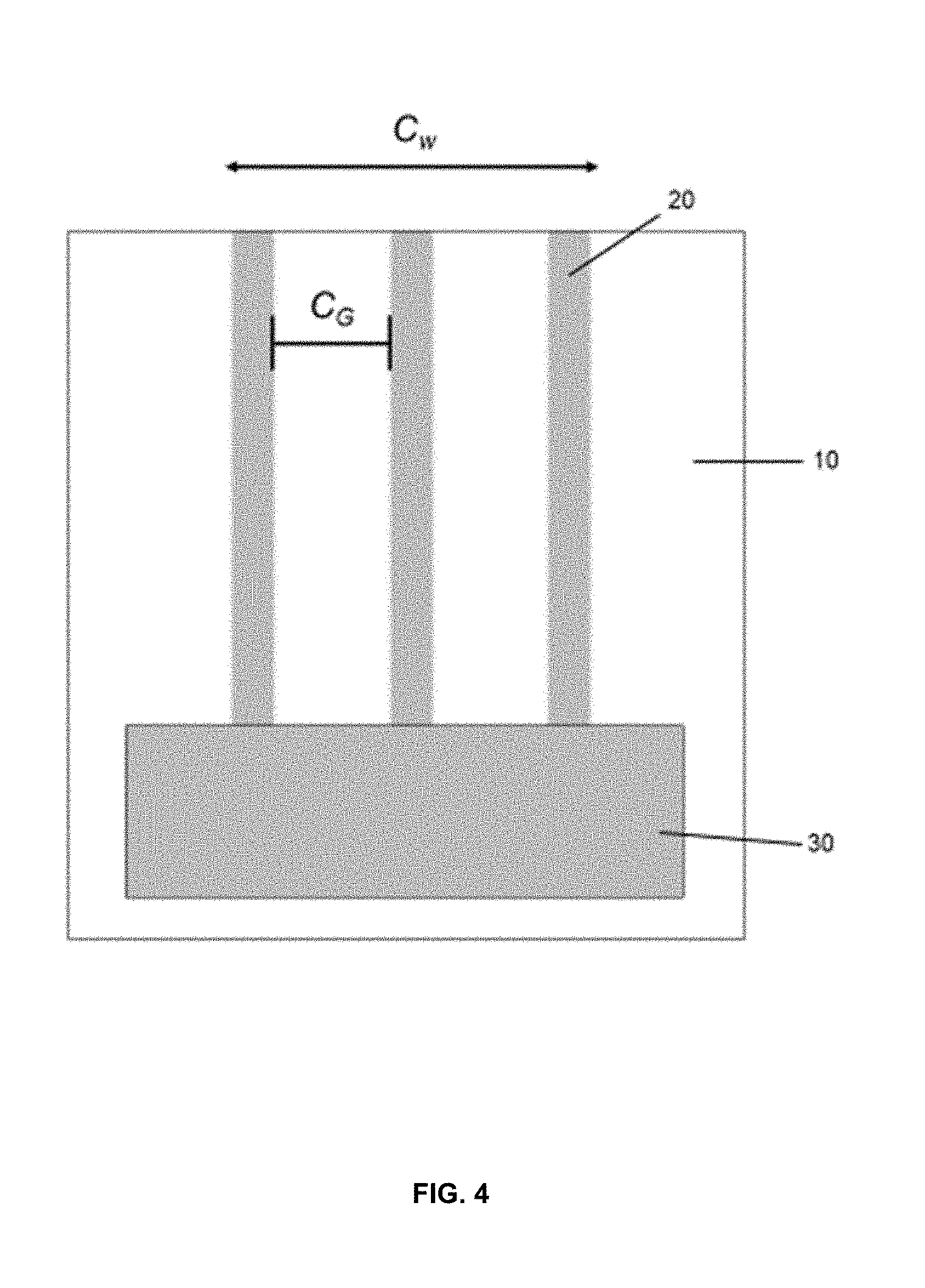
[0015] FIG. 4 is a plan view of a printable lithium composition printed onto a substrate;
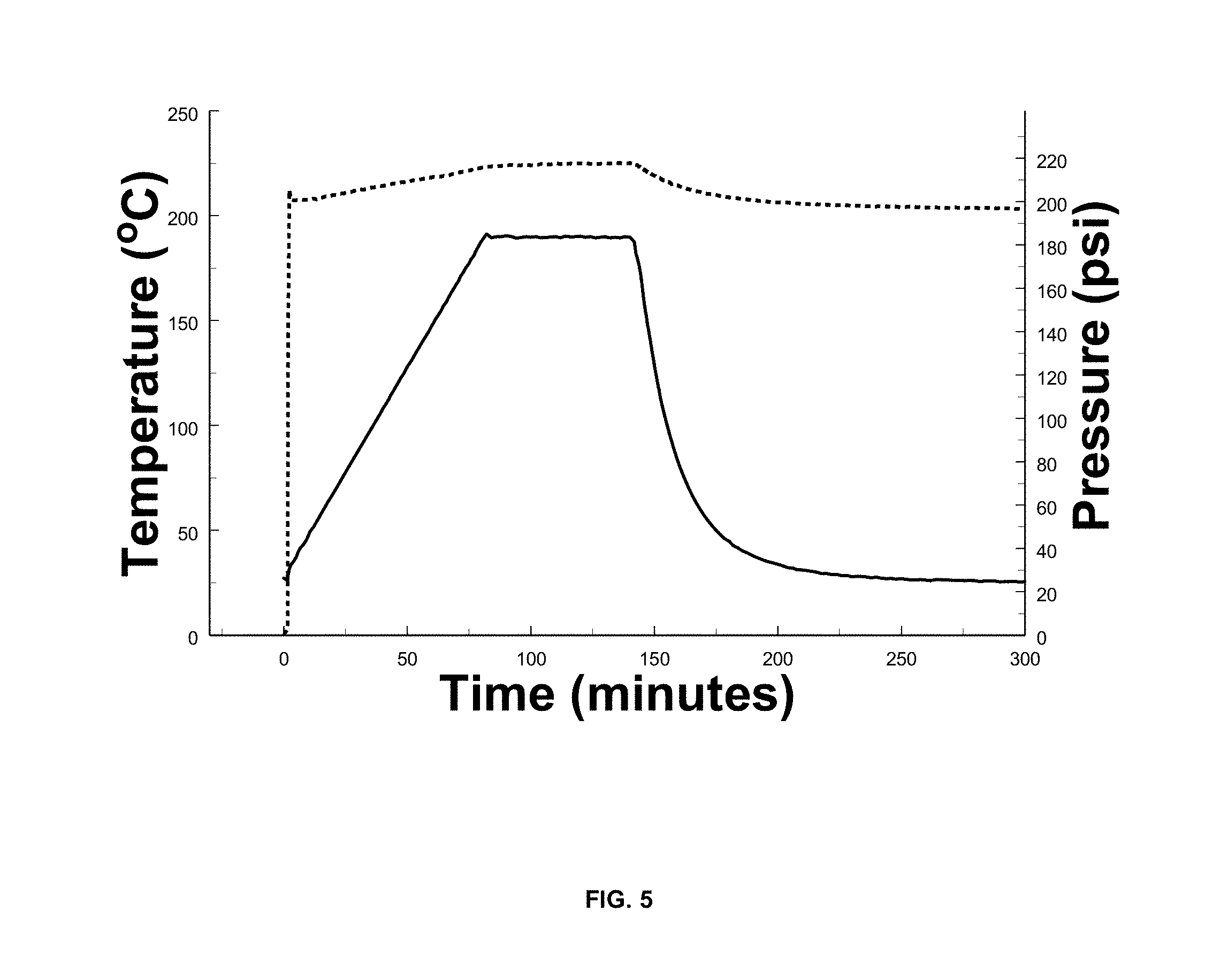
[0016] FIG. 5 is a temperature and pressure profile for the reactivity testing of SLMP/styrene butadiene/toluene printable lithium composition; and
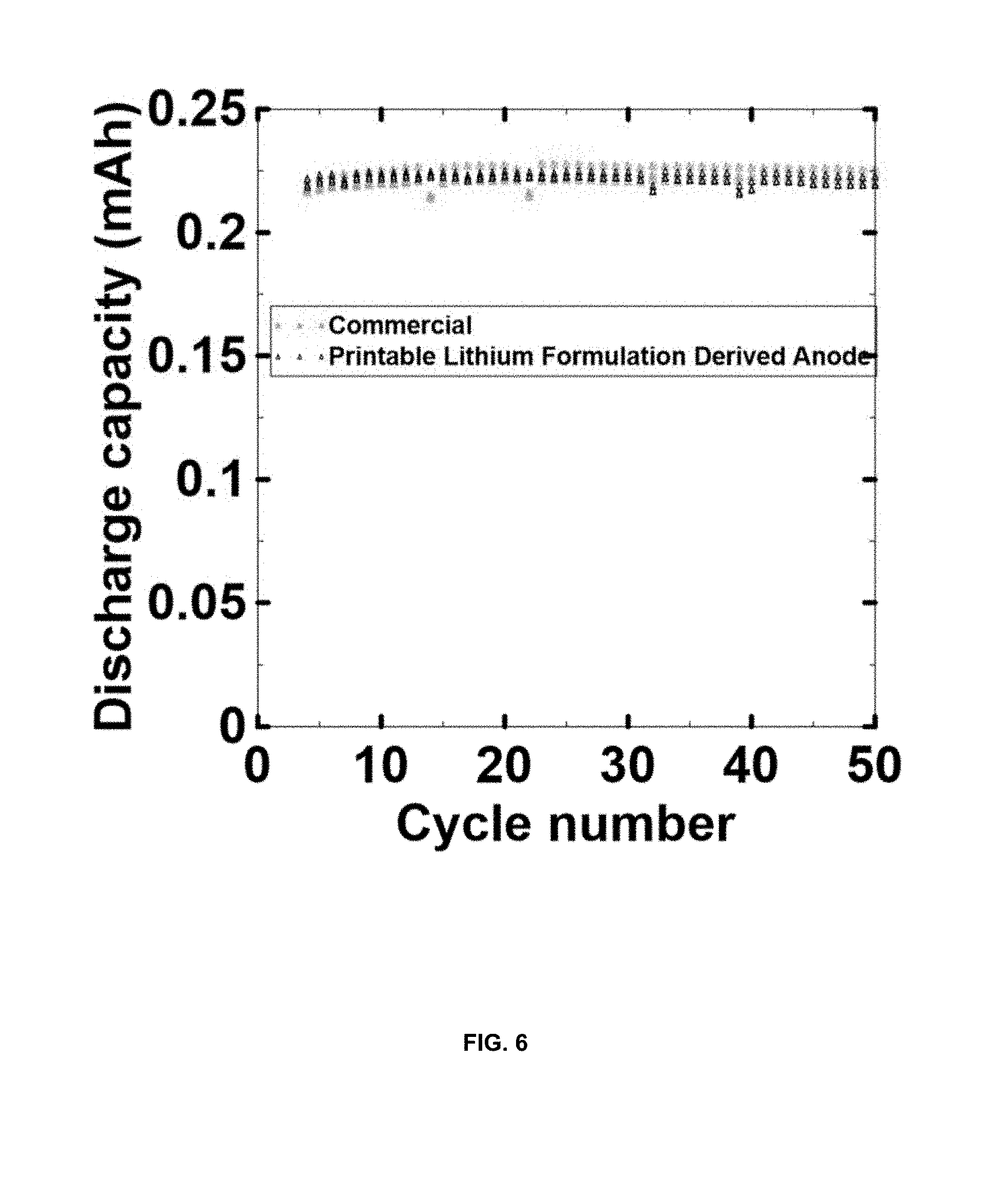
[0017] FIG. 6 is a plot showing the cycle performance for a pouch cell with printable lithium derived thin lithium film as the anode vs. commercial thin lithium foil.
DETAILED DESCRIPTION OF THE INVENTION
[0018] The foregoing and other aspects of the present invention will now be described in more detail with respect to the description and methodologies provided herein. It should be appreciated that the invention can be embodied in different forms and should not be construed as limited to the embodiments set forth herein. Rather, these embodiments are provided so that this disclosure will be thorough and complete, and will fully convey the scope of the invention to those skilled in the art.
[0019] The terminology used in the description of the invention herein is for the purpose of describing particular embodiments only and is not intended to be limiting of the invention. As used in the description of the embodiments of the invention and the appended claims, the singular forms "a", "an" and "the" are intended to include the plural forms as well, unless the context clearly indicates otherwise. Also, as used herein, "and/or" refers to and encompasses any and all possible combinations of one or more of the associated listed items.
[0020] The term "about," as used herein when referring to a measurable value such as an amount of a compound, dose, time, temperature, and the like, is meant to encompass variations of 20%, 10%, 5%, 1%, 0.5%, or even 0.1% of the specified amount. Unless otherwise defined, all terms, including technical and scientific terms used in the description, have the same meaning as commonly understood by one of ordinary skill in the art to which this invention belongs.
[0021] As used herein, the terms "comprise," "comprises," "comprising," "include," "includes" and "including" specify the presence of stated features, integers, steps, operations, elements, and/or components, but do not preclude the presence or addition of one or more other features, integers, steps, operations, elements, components, and/or groups thereof.
[0022] As used herein, the term "consists essentially of" (and grammatical variants thereof), as applied to the compositions and methods of the present invention, means that the compositions/methods may contain additional components so long as the additional components do not materially alter the composition/method. The term "materially alter," as applied to a composition/method, refers to an increase or decrease in the effectiveness of the composition/method of at least about 20% or more.
[0023] All patents, patent applications and publications referred to herein are incorporated by reference in their entirety. In case of a conflict in terminology, the present specification is controlling.
[0024] In accordance with the present invention, a method for applying a printable lithium composition is provided. In one embodiment, the printable lithium composition is electrochemically active and may be used to form an anode by applying or depositing the printable lithium composition on an anode conductive or carrier material (e.g., copper or polymer or ceramic films). The printable lithium composition comprises a lithium metal powder, one or more polymer binders, one or more rheology modifiers and may further include a solvent or co-solvent.
[0025] In another embodiment, the printable lithium composition may be applied or deposited to prelithiate an anode or cathode. The prelithiated anode or cathode may be incorporated into an energy storage device such as a capacitor or battery. The battery may be comprised of liquid electrolytes. In another embodiment, the battery may be comprised of solid electrolytes to form a solid-state battery. In another embodiment, the printable lithium composition may be used applied or deposited to form a monolithic lithium metal anode for use in conventional and solid-state batteries.
[0026] In yet another embodiment, the printable lithium composition may be applied or deposited so as to form a solid electrolyte for a solid-state battery, and includes combining the printable lithium composition with a polymer or ceramic material to form a solid electrolyte.
[0027] In one embodiment, the active anode material and the printable lithium composition are provided together and extruded onto the current collector (e.g., copper, nickel, etc.). For instance, the active anode material and printable lithium composition may be mixed and co-extruded together. Examples of active anode materials include graphite, graphite-SiO, graphite-SnO, SiO, hard carbon and other lithium ion battery and lithium ion capacitor anode materials. In another embodiment, the active anode material and the printable lithium composition are co-extruded to form a layer of the printable lithium composition on the current collector. The deposition of the printable lithium composition including the above extrusion technique may include depositing as wide variety patterns (e.g., dots, stripes), thicknesses, widths, etc. For example, the printable lithium composition and active anode material may be deposited as a series of stripes, such as described in US Publication No. 2014/0186519 incorporated herein by reference in its entirety. The stripes would form a 3D structure that would account for expansion of the active anode material during lithiation. For example, silicon may expand by 300 to 400 percent during lithiation. Such swelling potentially adversely affects the anode and its performance. By depositing the printable lithium as a thin stripe in the Y-plane as an alternating pattern between the silicon anode stripes, the silicon anode material can expand in the X-plane alleviating electrochemical grinding and loss of particle electrical contact. Thus, the printing method can provide a buffer for expansion. In another example, where the printable lithium formulation is used to form the anode, it could be co-extruded in a layered fashion along with the cathode and separator, resulting in a solid-state battery.
[0028] In one embodiment, the printable lithium composition may be applied to a substrate or a preformed anode by coating the substrate with a roller. One example is a gravure coating device, such as one described in U.S. Pat. No. 4,948,635 herein incorporated by reference in its entirety. In the example shown in FIG. 1, a pair of spaced rollers 12, 12' support the substrate 10 as it advances toward a gravure roller 14. A nozzle is utilized to apply the coating material to the gravure roller 14 while a doctor blade 16 is utilized to remove excess coating from the gravure roller 14. The gravure roller 14 contacts the substrate 10 as it travels through the gravure roller to apply the ink composition. The gravure roller can be designed to print various patterns on the surface of the substrate; for example, lines or dots.
[0029] In another embodiment, the printable lithium composition may be applied to a substrate by extruding the printable lithium composition onto the substrate from an extruder. One example of an extruder is described in U.S. Pat. No. 5,318,600 herein incorporated by reference in its entirety. In such an embodiment, high pressure forces the printable lithium composition through an extrusion nozzle to coat the exposed surface area of the substrate. As seen in FIG. 2, the ink composition 20 is contained within a container 22. A high pressure forces the ink composition 20 through an extrusion nozzle 24 to coat the exposed surface area of the substrate 10. One example of such an extruding apparatus may be mounted upon a suitable table and includes the hydraulic cylinder having a high pressure oil feed inlet and an oil outlet. The hydraulic cylinder typically contains a piston that drives the piston head into engagement with the upper end of the extrusion plunger 26.
[0030] In another embodiment, the printable lithium composition may be applied to a substrate by printing the printable lithium composition onto the substrate. Slot die print heads may be used to print monolithic, stripe or other patterns of the printable lithium composition onto the substrate. One example of a compatible printer utilizing a slot die print head is described in U.S. Pat. No. 5,494,518 herein incorporated by reference in its entirety. As seen in FIGS. 3 and 4, a slot coating die typically comprises a slot coating die head 30 through which the ink composition 20 is extruded onto the substrate 10, as the substrate 10 is moved past the slot coating die head 30. FIG. 4 illustrates one example of a slot coating wherein the ink composition 20 having a coating width C.sub.w and gaps between each strip designated as C.sub.G. Both C.sub.w and C.sub.G may vary depending on the configuration of the slot coating die head 30.
[0031] In another embodiment, a conventional carbon anode may be prelithiated by depositing the printable lithium composition on the carbon anode. This will obviate the problem associated with carbon anodes in which upon initial charging of the cell when lithium is intercalated into the carbon some irreversibility occurs due to some lithium and cell electrolyte being consumed resulting in an initial capacity loss.
[0032] In one embodiment, the printable lithium composition may be used to pre-lithiate an anode as described in U.S. Pat. No. 9,837,659 herein incorporated by reference in its entirety. For example, the method includes disposing a layer of printable lithium composition adjacent to a surface of a pre-fabricated/pre-formed anode. The pre-fabricated electrode comprises an electroactive material. In certain variations, the printable lithium composition may be applied to the carrier/substrate via a deposition process. A carrier substrate on which the layer of printable lithium composition may be disposed may be selected from the group consisting of: polymer films (e.g., polystyrene, polyethylene, polyethyleneoxide, polyester, polypropylene, polypolytetrafluoroethylene), ceramic films, copper foil, nickel foil, or metal foams by way of non-limiting example. Heat may then be applied to the printable lithium composition layer on the substrate or the pre-fabricated anode. The printable lithium composition layer on the substrate or the pre-fabricated anode may be further compressed together, under applied pressure. The heating, and optional applied pressure, facilitates transfer of lithium onto the surface of the substrate or anode. In case of transfer to the pre-fabricated anode, pressure and heat can result in mechanical lithiation, especially where the pre-fabricated anode comprises graphite. In this manner, lithium transfers to the electrode and due to favorable thermodynamics is incorporated into the active material.
[0033] In one embodiment, the printable lithium composition may be incorporated within the anode as described in US Publication No. 2018/0269471 herein incorporated by reference in its entirety. For example, the anode can comprise an active anode composition and the printable lithium composition, and any electrically conductive powder if present. In additional or alternative embodiments, the printable lithium composition is placed along the surface of the electrode. For example, the anode can comprise an active layer with an active anode composition and a printable lithium composition source layer on the surface of active layer. In an alternative configuration, the printable lithium composition source layer is between the active layer and a current collector. Also, in some embodiments, the anode can comprise printable lithium composition source layers on both surfaces of the active layer.
[0034] In one embodiment, the printable lithium composition may be incorporated into a three-dimensional electrode structure as described in US Publication No. 2018/0013126 herein incorporated by reference in its entirety. For example, the printable lithium composition may be incorporated into a three-dimensional porous anode, porous current collector or porous polymer or ceramic film, wherein the printable lithium composition may be deposited therein.
[0035] In some embodiments, an electrode prelithiated with the printable lithium composition can be assembled into a cell with the electrode to be preloaded with lithium. A separator can be placed between the respective electrodes. Current can be allowed to flow between the electrodes. For example, an anode prelithiated with the printable lithium composition of the present invention may be formed into a second battery such as described in U.S. Pat. No. 6,706,447 herein incorporated by reference in its entirety.
[0036] In one embodiment, the printable lithium composition is deposited or applied to an active anode material on a current collector namely to form a prelithiated anode. Suitable active anode materials include graphite and other carbon-based materials, alloys such as tin/cobalt, tin/cobalt/carbon, silicon-carbon, variety of silicone/tin based composite compounds, germanium-based composites, titanium based composites, elemental silicon, and germanium. The anode materials may be a foil, mesh or foam. Application may be via spraying, extruding, coating, printing, painting, dipping, and spraying.
[0037] Anodes prelithiated using the printable lithium composition may be incorporated into various types of batteries. For example, the prelithiated anodes may be incorporated into batteries as disclosed in U.S. Pat. Nos. 7,851,083, 8,088,509, 8,133,612, 8,276,695, and 9,941,505, which are incorporated herein by reference in their entireties. Printing the printable lithium composition on an anode material may be an alternative to smearing lithium as disclosed in U.S. Pat. No. 7,906,233 incorporated herein by reference in its entirety.
[0038] The cathode is formed of an active material, which is typically combined with a carbonaceous material and a binder polymer. The active material used in the cathode is preferably a material that can be lithiated. Preferably, non-lithiated materials such as MnO.sub.2, V.sub.2O.sub.5, MoS.sub.2, metal fluorides or mixtures thereof, Sulphur and sulfur composites can be used as the active material. However, lithiated materials such as LiMn.sub.2O.sub.4 and LiMO.sub.2 wherein M is Ni, Co or Mn that can be further lithiated can also be used. The non-lithiated active materials are preferred because they generally have higher specific capacities, lower cost and broader choice of cathode materials in this construction that can provide increased energy and power over conventional secondary batteries that include lithiated active materials.
[0039] In one embodiment, the printable lithium composition may be used to prelithiate a capacitor, such as an anode in a lithium-ion capacitor as described in US Publication No. 2017/0301485 herein incorporated by reference. For example, the anode can be constructed using hard carbon, soft carbon or graphite. The anode may then be attached to a current collector before or during having a printable lithium composition layer coated on the top surface of the anode. The printable lithium composition may also be used to prelithiate an energy storage device such as a lithium-ion capacitor as described in U.S. Pat. No. 9,711,297 herein incorporated by reference in its entirety.
[0040] In one embodiment, the printable lithium composition may be used to prelithiate a hybrid battery/capacitor as described in US Publication No. 2018/0241079 herein incorporated by reference in its entirety. The term "hybrid electrode" refers to an electrode that includes both battery electrode materials and capacitor electrode materials. In one embodiment, the hybrid cathode may comprise a blend of higher energy materials, such as battery cathode materials, and high power materials, such as capacitor cathode materials. For example, lithium-ion battery cathode materials may be combined with ultracapacitor or supercapacitor cathode materials. To complete the hybrid lithium-ion cell assembly, the hybrid cathode may be disposed against an anode electrode with a polyolefin separator in between the electrodes and is placed in a confined packaging, such as an energy storage device container, e.g. housing. The electrode stack is filled and contacted with a suitable electrolyte, such as a solvent containing a lithium-ion electrolyte salt and optionally including an electrolyte additive. The energy storage device package can be sealed.
[0041] The anode used in combination with the hybridized cathode can comprise elemental metal, such as elemental lithium. A method for prelithiation is direct addition of the printable lithium composition to the electrode formulation. This printable lithium composition uniformly integrated into the electrode formulation can then be used to form an electrode film, in a dry process, which can then be laminated onto a current collector, such as a metal foil, to form the electrode, such as an anode. The printable lithium composition can be also applied to the current collector prior to the lamination with the dry electrode. Embodiments herein can allow for a homogenous, and in some embodiments, dry, and/or particulate material, to be used as a raw material in the anode and hybridized cathode. Some embodiments herein can avoid the need for two separate layers on each electrode (such as a "battery material" layer and a "capacitor material" layer), which can avoid the need to introduce manufacturing complexity and added production cost. In further embodiments, the pre-doped electrode is a hybrid cathode. It will be understood that the elemental metal and related concepts described herein with respect to an energy storage device with lithium may be implemented with other energy storage devices, and other metals.
[0042] As disclosed in US application Ser. No. ______ (Attorney Matter ID. 073396.1116, filed concurrently with this application) and hereby incorporated by reference in its entirety, the printable lithium composition comprises a lithium metal powder, one or more polymer binders, one or more rheology modifiers and may further include a solvent or co-solvent. The polymer binder may be compatible with the lithium metal powder. The rheology modifier may be compatible with the lithium metal powder and the polymer binder. The solvent may be compatible with the lithium metal powder and with the polymer binder.
[0043] The lithium metal powder may be in the form of a finely divided powder. The lithium metal powder typically has a mean particle size of less than about 80 microns, often less than about 40 microns and sometimes less than about 20 microns. The lithium metal powder may be a low pyrophoricity stabilized lithium metal power (SLMP.RTM.) available from FMC. The lithium metal powder may also include a substantially continuous layer or coating of fluorine, wax, phosphorus or a polymer or the combination thereof (as disclosed in U.S. Pat. Nos. 5,567,474, 5,776,369, and 5,976,403). Lithium metal powder has a significantly reduced reaction with moisture and air.
[0044] The lithium metal powder may also be alloyed with a metal. For example, the lithium metal powder may be alloyed with a Group I-VIII element. Suitable elements from Group IB may include, for example, copper, silver, or gold. Suitable elements from Group IIB may include, for example, zinc, cadmium, or mercury. Suitable elements from Group IIA of the Periodic Table may include beryllium, magnesium, calcium, strontium, barium, and radium. Elements from Group IIIA that may be used in the present invention may include, for example, boron, aluminum, gallium, indium, or thallium. Elements from Group IVA that may be used in the present invention may include, for example, carbon, silicon, germanium, tin, or lead. Elements from Group VA that may be used in the present invention may include, for example, nitrogen, phosphorus, or bismuth. Suitable elements from Group VIIIB may include, for example, nickel, palladium, or platinum.
[0045] The polymer binder is selected so as to be compatible with the lithium metal powder. "Compatible with" or "compatibility" is intended to convey that the polymer binder does not violently react with the lithium metal powder resulting in a safety hazard. The lithium metal powder and the polymer binder may react to form a lithium-polymer complex, however, such complex should be stable at various temperatures. It is recognized that the amount (concentration) of lithium and polymer binder contribute to the stability and reactivity. The polymer binder may have a molecular weight of about 1,000 to about 8,000,000, and often has a molecular weight of 2,000,000 to 5,000,000. Suitable polymer binders may include one or more of poly(ethylene oxide), polystyrene, polyisobutylene, natural rubbers, butadiene rubbers, styrene-butadiene rubber, polyisoprene rubbers, butyl rubbers, hydrogenated nitrile butadiene rubbers, epichlorohydrin rubbers, acrylate rubbers, silicon rubbers, nitrile rubbers, polyacrylic acid, polyvinylidene chloride, polyvinyl acetate, ethylene propylene diene termonomer, ethylene vinyl acetate copolymer, ethylene-propylene copolymers, ethylene-propylene terpolymers, polybutenes. The binder may also be a wax.
[0046] The rheology modifier is selected to be compatible with the lithium metal powder and the polymer binder. The rheology modifier provides rheology properties such as viscosity and flow under shear conditions. The rheology modifier may also provide conductivity, improved capacity and/or improved stability/safety depending on the selection of the rheology modifier. To this end, the rheology modifier may be the combination of two or more compounds so as to provide different properties or to provide additive properties. Exemplary rheology modifiers may include one or more of carbon black, carbon nanotubes, graphene, silicon nanotubes, graphite, hard carbon and mixtures, fumed silica, titanium dioxide, zirconium dioxide and other Group IIA, IIIA, IVB, VB and VIA elements/compounds and mixtures or blends thereof. Other additives intended to increase lithium ion conductivity can be used; for example, electrochemical device electrolyte salts such as lithium perchlorate (LiClO.sub.4), lithium hexafluorophosphate (LiPF.sub.6), lithium nitrate (LiNO.sub.3), lithium bis(oxalate) borate (LiBOB), and lithium trifluoromethanesulfonimide (LiTFSI).
[0047] Solvents compatible with lithium may include acyclic hydrocarbons, cyclic hydrocarbons, aromatic hydrocarbons, symmetrical ethers, unsymmetrical ethers, cyclic ethers, alkanes, sulfones, mineral oil, and mixtures, blends or cosolvents thereof. Examples of suitable acyclic and cyclic hydrocarbons include n-hexane, n-heptane, cyclohexane, and the like. Examples of suitable aromatic hydrocarbons include toluene, ethylbenzene, xylene, isopropylbenzene (cumene), and the like. Examples of suitable symmetrical, unsymmetrical and cyclic ethers include di-n-butyl ether, methyl t-butyl ether, tetrahydrofuran, glymes and the like. Commercially available isoparaffinic synthetic hydrocarbon solvents with tailored boiling point ranges such as Shell Sol.RTM. (Shell Chemicals) or Isopar.RTM. (Exxon) are also suitable.
[0048] The polymer binder and solvents are selected to be compatible with each other and with the lithium metal powder. In general, the binder or solvent should be non-reactive with the lithium metal powder or in amounts so that any reaction is kept to a minimum and violent reactions are avoided. The binder and solvent should be compatible with each other at the temperatures at which the printable lithium composition is made and will be used. Preferably the solvent (or co-solvent) will have sufficient volatility to readily evaporate from the printable lithium composition (e.g., in slurry form) to provide drying of the printable lithium composition (slurry) after application.
[0049] The components of the printable lithium composition may be mixed together as a slurry or paste to have a high concentration of solid. Thus the slurry/paste may be in the form of a concentrate with not all of the solvent necessarily added prior to the time of depositing or applying. In one embodiment, the lithium metal powder should be uniformly suspended in the solvent so that when applied or deposited a substantially uniform distribution of lithium metal powder is deposited or applied. Dry lithium powder may be dispersed such as by agitating or stirring vigorously to apply high sheer forces.
[0050] In another embodiment, a mixture of the polymer binder, rheology modifier, coating reagents, and other potential additives for the lithium metal powder may be formed and introduced to contact the lithium droplets during the dispersion at a temperature above the lithium melting point, or at a lower temperature after the lithium dispersion has cooled such as described in U.S. Pat. No. 7,588,623 the disclosure of which is incorporated by reference in its entirety. The thusly modified lithium metal may be introduced in a crystalline form or in a solution form in a solvent of choice. It is understood that combinations of different process parameters could be used to achieve specific coating and lithium powder characteristics for particular applications.
[0051] Conventional pre-lithiation surface treatments require compositions having very low binder content and very high lithium; for example, see U.S. Pat. No. 9,649,688 the disclosure of which is incorporated by reference in its entirety. However, embodiments of the printable lithium composition in accordance with the present invention can accommodate higher binder ratios, including up to 20 percent on dry basis. Various properties of the printable lithium composition, such as viscosity and flow, may be modified by increasing the binder and modifier content up to 50% dry basis without loss of electrochemical activity of lithium. Increasing the binder content facilitates the loading of the printable lithium composition and the flow during printing. For example, in one embodiment the printable lithium composition comprises about 70% lithium metal powder and about 30% polymer binder and rheology modifiers. In another embodiment, the printable lithium composition may comprise about 85% lithium metal powder and about 15% polymer binder and rheology modifiers.
[0052] An important aspect of printable lithium compositions is the rheological stability of the suspension. Because lithium metal has a low density of 0.534 g/cc, it is difficult to prevent lithium powder from separating from solvent suspensions. By selection of lithium metal powder loading, polymer binder and conventional modifier types and amounts, viscosity and rheology may be tailored to create the stable suspension of the invention. A preferred embodiment shows no separation at greater than 90 days. This can be achieved by designing compositions with very high zero shear viscosity in the range of 1.times.10.sup.4 cps to 1.times.10.sup.7 cps. It is however very important to the application process that the compositions, when exposed to shear, exhibit viscosity characteristics in the ranges claimed.
[0053] The resulting printable lithium composition preferably may have a viscosity at 10 s.sup.-1 about 20 to about 20,000 cps, and often a viscosity of about 100 to about 10,000 cps. At such viscosity, the printable lithium composition is a flowable suspension or gel. The printable lithium composition preferably has an extended shelf life at room temperature and is stable against metallic lithium loss at temperatures up to 60.degree. C., often up to 120.degree. C., and sometimes up to 180.degree. C. The printable lithium composition may separate somewhat over time but can be placed back into suspension by mild agitation and/or application of heat.
[0054] In one embodiment, the printable lithium composition comprises on a solution basis about 5 to 50 percent lithium metal powder, about 0.1 to 20 percent polymer binder, about 0.1 to 30 percent rheology modifier and about 50 to 95 percent solvent. In one embodiment, the printable lithium composition comprises on a solution basis about 15 to 25 percent lithium metal powder, about 0.3 to 0.6 percent polymer binder having a molecular weight of 4,700,000, about 0.5 to 0.9 percent rheology modifier, and about 75 to 85 percent solvent. Typically, the printable lithium composition is applied or deposited to a thickness of about 10 microns to 200 microns prior to pressing. After pressing, the thickness can be reduced to between about 1 to 50 microns. Examples of pressing techniques are described, for example, in U.S. Pat. Nos. 3,721,113 and 6,232,014 which are incorporated herein by reference in their entireties.
[0055] In another embodiment, the printable lithium composition may be applied or deposited to prelithiate an anode or cathode of a solid-state battery. For example, the printable lithium composition may be used to form a monolithic lithium metal anode for use in a solid-state battery, including solid-state batteries as described in U.S. Pat. Nos. 8,252,438 and 9,893,379 and incorporated herein by reference in their entireties.
[0056] In another embodiment, the printable lithium composition may be used to form or in conjunction with a solid electrolyte for use in a solid-state battery. For instance, the printable lithium composition may be deposited on a variety of solid-state electrolytes as described in U.S. Pat. No. 7,914,930 herein incorporated by reference in its entirety. One example of a solid-state secondary battery may include a positive electrode capable of electrochemically absorbing and desorbing lithium; a negative electrode capable of electrochemically absorbing and desorbing lithium, the negative electrode including an active material layer that comprises an active material, the active material layer being carried on a current collector; and a non-aqueous electrolyte. A method includes the steps of: reacting lithium with the active material of the negative electrode by bringing the printable lithium composition into contact with a surface of the active material layer of the negative electrode; and thereafter combining the negative electrode with the positive electrode to form an electrode assembly.
[0057] The following examples are merely illustrative of the invention and are not limiting thereon.
EXAMPLES
Example 1
[0058] 10 g of solution styrene butadiene rubber (S-SBR Europrene Sol R 72613) is dissolved in 90 g toluene (99% anhydrous, Sigma Aldrich) by stirring at 21.degree. C. for 12 hours. 6 g of the 10 wt % SBR (polymer binder) in toluene (solvent) is combined with 0.1 g carbon black (Timcal Super P) (rheology modifier) and 16 g of toluene and dispersed in a Thinky ARE 250 planetary mixer for 6 minutes at 2000 rpm. 9.3 g of stabilized lithium metal powder (SLMP.RTM., FMC Lithium Corp.) having polymer coating of 20 to 200 .mu.m and d50 of 20 .mu.m is added to this suspension and dispersed for 3 minutes at 1000 rpm in a Thinky mixer. The printable lithium is then filtered through 180 .mu.m opening stainless steel mesh. The printable lithium suspension is then doctor blade coated on to a copper current collector at a wet thickness of 2 mil (.about.50 .mu.m). FIG. 6 is a plot showing the cycle performance for a pouch cell LMP with printable lithium derived thin lithium film as the anode vs. commercial thin lithium foil.
Example 2
[0059] 10 g of 135,000 molecular weight ethylene propylene diene terpolymer (EPDM) (Dow Nordel IP 4725P) is dissolved in 90 g p-xylene (99% anhydrous, Sigma Aldrich) by stirring at 21.degree. C. for 12 hours. 6 g of the 10 wt % EPDM (polymer binder) in p-xylene (solvent) is combined with 0.1 g TiO2 (Evonik Industries) (rheology modifier) and 16 g of toluene and dispersed in a Thinky ARE 250 planetary mixer for 6 minutes at 2000 rpm. 9.3 g of stabilized lithium metal powder (SLMP.RTM., FMC Lithium Corp.) having polymer coating of 20 to 200 .mu.m and d50 of 20 .mu.m is added to this suspension and dispersed for 3 minutes at 1000 rpm in a Thinky mixer. The printable lithium is then filtered through 180 .mu.m opening stainless steel mesh. The printable lithium composition is then doctor blade coated on to a copper current collector at a wet thickness of 2 mil (.about.50 .mu.m).
Shelf Life Stability
[0060] Printable lithium components must be selected to ensure chemical stability for long shelf life at room temperature and stability at elevated temperature for shorter durations such as during transport or during the drying process. The printable lithium composition stability was tested using calorimetry. 1.5 g SLMP was added to a 10 ml volume Hastelloy ARC bomb sample container. 2.4 g of 4% SBR binder solution was added to the container. The container was fitted with a 24-ohm resistance heater and a thermocouple to monitor and control sample temperature. The bomb sample set-up was loaded into a 350 ml containment vessel along with insulation. An Advance Reactive Screening Systems Tool calorimeter by Fauske Industries was used to assess the compatibility of the printable lithium solutions during a constant rate temperature ramp to 190.degree. C. The temperature ramp rate was 2.degree. C./min and the sample temperature was held at 190.degree. C. for 60 minutes. The test was conducted under 200 psi Argon pressure to prevent boiling of the solvent. FIG. 5 shows the temperature and pressure profiles for the reactivity testing of a SLMP/styrene butadiene/toluene printable lithium composition.
Printing Performance
[0061] The quality of the printable lithium composition with regard to printability is measured by several factors, for example, consistency of flow which directly impact one's ability to control lithium loading on a substrate or an electrode surface. An effective means of measuring flow is Flow Conductance which is an expression of the loading per square centimeter in relation to the factors which control the loading--the pressure during extrusion and the speed of the printer head. It can most simply be thought of as the inverse of flow resistance.
[0062] The expression is used to allow comparisons between prints of varying pressures and speeds, and changes in Flow Conductance can alert one to non-linear relationships of flow with pressure. These are important for scaling the loading for a printable lithium up or down depending on the need of the anode or cathode. An ideal printable lithium composition would behave in a linear fashion to changes in extrusion pressure.
[0063] To test printability, a printable lithium composition is filtered through 180 .mu.m opening stainless steel mesh and loaded into a Nordson EFD 10 ml syringe. The syringe is loaded into a Nordson EFD HP4x syringe dispenser and attached to a slot die print head. The slot die print head is equipped with a 100 .mu.m-300 .mu.m thick shim with channel openings designed to deliver the desired printable lithium composition loading. The slot die head is mounted on a Loctite 300 Series robot. The print head speed is set to 200 mm/s and the printing pressure is between 20 and 200 psi argon, depending on shim and channel design. The print length is 14 cm. In an example printing trial experiment, the printable lithium composition was printed 30 times from a single syringe at dispenser settings ranging from 80 psi to 200 psi. For this print trial experiment, the flow conductance average was 0.14
mg s * cm 2 * lbf in 2 ##EQU00001##
with standard deviation of 0.02. Although this printable composition does not behave in a perfectly linear fashion, the composition flow response to changes in dispenser pressure is predictable to allow one skilled in the art to fine tune lithium loading to the desired level. Thus, at fixed dispenser pressure conditions the loading of lithium can be controlled very consistently. For example, for a print of 0.275
mAh cm 2 ##EQU00002##
lithium metal, the CV is about 5%.
Electrochemical Testing
[0064] The pre-lithiation effect of printable lithium composition can be evaluated by printing the required amount of printable lithium onto the surface of prefabricated electrodes. The pre-lithiation lithium amount is determined by testing the anode material in half-cell format and calculating the lithium required to compensate for the first cycle losses due to formation of SEI, or other side reactions. To calculate the necessary amount of printable lithium, the capacity as lithium metal of the composition must be known and is approximately 3600 mAh/g dry lithium basis for the compositions used as examples.
[0065] The pre-lithiation effect is tested using Graphite-SiO/NCA pouch cells. The Graphite-SiO anode sheet has the following formulation: artificial graphite (90.06%)+SiO (4.74%)+carbon black (1.4%)+SBR/CMC (3.8%). The capacity loading of the electrode is 3.59 mAh/cm.sup.2 with 87% first cycle CE (columbic efficiency). The printable lithium is applied onto a Graphite-SiO anode at 0.15 mg/cm.sup.2 lithium metal. The electrode is dried at 80.degree. C. for 100 min followed by lamination at a roller gap approximately 75% of the thickness of the electrode. A 7 cm.times.7 cm electrode is punched from the printable lithium treated anode sheet. The positive electrode has the following formulation: NCA (96%)+carbon black (2%)+PVdF (2%). The positive electrode is 6.8 cm.times.6.8 cm with capacity loading of 3.37 mAh/cm.sup.2. The NCA cathode has 90% first cycle CE. The anode to cathode capacity ratio is 1.06 and the baseline for full cell first cycle CE is 77%. Single layer pouch cells are assembled and 1M LiPF.sub.6/EC+DEC (1:1) is used as the electrolyte. The cells are pre-conditioned for 12 hours at 21.degree. C. and then the formation cycle is conducted at 40.degree. C. The formation protocol is 0.1 C charge to 4.2V, constant voltage to 0.01 C and 0.1 C discharge to 2.8V. In the described test 89% first cycle CE was demonstrated.
[0066] Although the present approach has been illustrated and described herein with reference to preferred embodiments and specific examples thereof, it will be readily apparent to those of ordinary skill in the art that other embodiments and examples may perform similar functions and/or achieve like results. All such equivalent embodiments and examples are within the spirit and scope of the present approach.
* * * * *
D00000

D00001

D00002

D00003

D00004

D00005

D00006

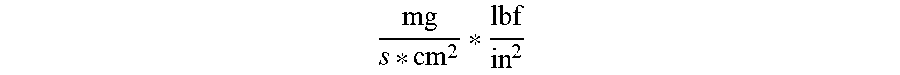

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.