Biochar As A Microbial Carrier
Belcher; Richard Wilson ; et al.
U.S. patent application number 16/238995 was filed with the patent office on 2019-07-11 for biochar as a microbial carrier. The applicant listed for this patent is Cool Planet Energy Systems, Inc.. Invention is credited to Richard Wilson Belcher, Brian Buege, Michael C. Cheiky, Mark L. Jarand, Han Suk Kim, Ronald A. Sills.
| Application Number | 20190210935 16/238995 |
| Document ID | / |
| Family ID | 59065069 |
| Filed Date | 2019-07-11 |











View All Diagrams
| United States Patent Application | 20190210935 |
| Kind Code | A1 |
| Belcher; Richard Wilson ; et al. | July 11, 2019 |
BIOCHAR AS A MICROBIAL CARRIER
Abstract
The invention relates to a microbial delivery system where biochar acts as a carrier for microbes.
| Inventors: | Belcher; Richard Wilson; (Oxnard, CA) ; Kim; Han Suk; (Thousand Oaks, CA) ; Buege; Brian; (Centennial, CO) ; Cheiky; Michael C.; (US) ; Sills; Ronald A.; (Houston, TX) ; Jarand; Mark L.; (Rotorua, NZ) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 59065069 | ||||||||||
| Appl. No.: | 16/238995 | ||||||||||
| Filed: | January 3, 2019 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15393214 | Dec 28, 2016 | 10173937 | ||
| 16238995 | ||||
| 15350920 | Nov 14, 2016 | 10093588 | ||
| 15393214 | ||||
| 14385986 | Dec 23, 2014 | 9493380 | ||
| PCT/US12/39862 | May 29, 2012 | |||
| 15350920 | ||||
| 13154213 | Jun 6, 2011 | 8317891 | ||
| 14385986 | ||||
| 15156256 | May 16, 2016 | 9809502 | ||
| 15393214 | ||||
| 14873053 | Oct 1, 2015 | 10252951 | ||
| 15393214 | ||||
| 14036480 | Sep 25, 2013 | 9359268 | ||
| 15393214 | ||||
| 13189709 | Jul 25, 2011 | 8568493 | ||
| 14036480 | ||||
| 62271486 | Dec 28, 2015 | |||
| 62290285 | Feb 2, 2016 | |||
| 62344865 | Jun 2, 2016 | |||
| 62432253 | Dec 9, 2016 | |||
| 62162219 | May 15, 2015 | |||
| 62058445 | Oct 1, 2014 | |||
| 62058472 | Oct 1, 2014 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C12N 1/14 20130101; C10B 53/02 20130101; C05G 3/00 20130101; C10B 57/02 20130101; C05F 11/08 20130101; C12N 11/12 20130101; Y02E 50/14 20130101; C01B 32/05 20170801; Y10S 71/903 20130101; C12N 1/20 20130101; Y02E 50/10 20130101; C05D 9/00 20130101; C05G 5/40 20200201; C09K 17/40 20130101; C05F 11/00 20130101; C09K 17/04 20130101; C05G 5/00 20200201; C12N 1/16 20130101; Y02P 20/145 20151101; C05F 11/02 20130101; C09K 17/02 20130101; C05F 11/02 20130101; C05F 11/08 20130101 |
| International Class: | C05F 11/08 20060101 C05F011/08; C12N 1/20 20060101 C12N001/20; C01B 32/05 20060101 C01B032/05; C12N 1/14 20060101 C12N001/14; C12N 11/12 20060101 C12N011/12; C12N 1/16 20060101 C12N001/16; C09K 17/04 20060101 C09K017/04; C05D 9/00 20060101 C05D009/00; C05F 11/00 20060101 C05F011/00; C10B 57/02 20060101 C10B057/02; C10B 53/02 20060101 C10B053/02; C09K 17/02 20060101 C09K017/02; C05G 3/00 20060101 C05G003/00; C05F 11/02 20060101 C05F011/02 |
Claims
1. A microbial delivery system comprising: biochar having pores; and microbes retained on the surface or in the pores of the biochar.
2. The delivery system of claim 1, where at least some of the microbes are inoculated into the pores of the biochar.
3. The delivery system of claim 2, where the microbes are inoculated into the pores of the biochar using mechanical, chemical, or biological assistance to move the microbes either into the pores of the biochar or onto the surfaces of the biochar.
4. The delivery system of claim 3 where the microbes are inoculated into the pores of the biochar using the application of positive or negative pressure.
5. The delivery system of claim 3 where the microbes are inoculated into the pores of the biochar using a surfactant.
6. The delivery system of claim 1 where the microbes are retained by the biochar through mixing the biochar and microbes together.
7. The delivery system of claim 6 where the microbes are retained on the biochar by suspending the microbes in liquid and depositing the microbes on the biochar.
8. The delivery system of claim 1 where the microbes retained by the biochar are selected from the group consisting of: Bacillus, Pseudomonas, Rhizobium, Burkholderia, Achromobacter, Agrobacterium, Microccocus, Aereobacter, Flavobacterium, Erwinia, Klebsiella, and Enterobacter, including Bacillus mucilaginosus, Bacillus edaphicus, Bacillus circulans, Paenibacillus spp., Acidothiobacillus ferrooxidans, Pseudomonas cepacia, Burkholderia cepacia, Klebsiella varticola, Pantoea aggioinerans; fungi such as Glomus mosseae, Glomus intraradices, Aspergillus terreus and Aspergillus niger.
9. The delivery system of claim 1 where the biochar is treated biochar.
10. The delivery system of claim 9 where the biochar has been treated to alter one or more of the following properties of the biochar: pH; hydrophobicity; hydrophilicity; ability of the biochar to hold moisture; ability of the biochar to retain and exchange certain types of nutrients; ion exchange capacity; physical protection from environmental hazards or protozoa; presence or absence of nutrients, micronutrients, or sources of metabolic carbon; or ability of the biochar to host other symbiotic microbes or plant systems.
11. The delivery system of claim 1, where at least some of the microbes are retained on the biochar through integrated growth with the biochar.
12. A method for delivering microbes into an environment, the method comprising combining biochar and microbes in the environment, where the biochar has been treated to have suitable properties for the microbes in the environment in which the biochar and microbes are combined.
13. Biochar having pores where pores are filled with a media containing microbes, where the media is infused into the pores of the biochar.
14. The biochar of claim 13 where the microbes includes microorganisms selected from the group consisting of: Bacillus, Pseudomonas, Rhizobium, Burkholderia, Achromobacter, Agrobacterium, Microccocus, Aereobacter, Flavobacterium, Erwinia, Klebsiella, and Enterobacter, including Bacillus mucilaginosus, Bacillus edaphicus, Bacillus circulans, Paenibacillus spp., Acidothiobacillus ferrooxidans, Pseudomonas cepacia, Burkholderia cepacia, Klebsiella variicola, Pantoea agglomerans; fungi such as Glomus mosseae, Glomus intraradices, Aspergillus terreus and Aspergillus niger.
15. The biochar of claim 13 where the media is infused into the pores of the biochar using positive or negative pressure.
16. The biochar of claim 13 where the media is infused into the pores of the biochar using a surfactant.
17. The biochar of claim 16 where the surfactant treatment comprises adding 1% surfactant to the media.
18. The biochar of claim 13 where the pores of the biochar are treated prior to infusion with the media.
19. The biochar of claim 13 where the moisture content of the pores of the biochar are adjusted prior to infusion with the media.
20. Biochar for use in agricultural, remediation, public health, or animal application, the biochar comprising a porous carbonaceous particle that has been treated and/or mixed with media containing microbes, whereby the porous carbonaceous particle after treatment and/or mixing has retained the media in at least some of the pores of the porous carbonaceous particle.
21. The biochar of claim 20 where the microbes are microorganisms selected from the group consisting of: Bacillus, Pseudomonas, Rhizobium, Burkholderia, Achromobacter, Agrobacterium, Microccocus, Aereobacter, Flavobacterium, Erwinia, Klebsiella, and Enterobacter, including Bacillus mucilaginosus, Bacillus edaphicus, Bacillus circulans, Paenibacillus spp., Acidothiobacillus ferrooxidans, Pseudomonas cepacia, Burkholderia cepacia, Klebsiella variicola, Pantoea agglomerans; fungi such as Glomus mosseae, Glomus intraradices, Aspergillus terreus and Aspergillus niger.
22. A method for creating enhanced biochar, the method comprising the steps of infusing media containing microbes into the pores of biochar.
23. The method of claim 22 where the media is infused into the pores of the biochar using positive or negative pressure.
24. The method of claim 22 where the media is infused into the pores of the biochar using a surfactant.
25. The method of claim 22 where the media includes microorganisms selected from the group consisting of: Bacillus, Pseudomonas, Rhizobium, Burkholderia, Achromobacter, Agrobacterium, Microccocus, Aereobacter, Flavobacterium, Erwinia, Klebsiella, and Enterobacter, including Bacillus mucilaginosus, Bacillus edaphicus, Bacillus circulans, Paenibacillus spp., Acidothiobacillus ferrooxidans, Pseudomonas cepacia, Burkholderia cepacia, Klebsiella variicola, Pantoea agglomerans; fungi such as Glomus mosseae, Glomus intraradices, Aspergillus terreus and Aspergillus niger.
26. A methods for integrating a microbial community with a biochar particle, the method selected from the group consisting of: while under vacuum, pulling the microbial solution through a treated biochar bed that is resting on a membrane filter; spraying a microbial solution on top of a treated biochar bed; lyophilizing a microbial solution and then blending the freeze dried solution with the treated biochar; again infusing, the treated biochar with a microbial solution; adding treated biochar to a growth medium, inoculating with the microbe, and incubating to allow the microbe to grow in said biochar containing medium; infusing, as defined previously, the biochar with a food source and then introducing the substrate infused biochar to a microbe and incubating to allow the microbes to grow; blending commercially available strains in dry form with treated biochar; adding the treated biochar to a microbial solution and then centrifuging at a high speed, potentially with a density gradient in order to promote the biochar to spin down with the microbes; densely packing a column with treated biochar and then gravity flowing a microbial solution through the column and possibly repeating this multiple times; or adding the microbe to a solution based binder that is well known to enter the treated biochar pores and then adding said solution to the treated biochar.
27. The method of claim 26 where biochar is sterilized before being infused with the microbial community.
28. The method of claim 26 where the microbial community includes a microorganism selected from the group consisting of: Bacillus, Pseudomonas, Rhizobium, Burkholderia, Achromobacter, Agrobacterium, Microccocus, Aereobacter, Flavobacterium, Erwinia, Klebsiella, and Enterobacter, including Bacillus mucilaginosus, Bacillus edaphicus, Bacillus circulans, Paenibacillus spp., Acidothiobacillus ferrooxidans, Pseudomonas cepacia, Burkholderia cepacia, Klebsiella variicola, Pantoea agglomerans; fungi such as Glomus mosseae, Glomus intraradices, Aspergillus terreus and Aspergillus niger.
29. The biochar of claim 20 where the biochar has been treated to alter one or more of the following properties of the biochar: pH; hydrophobicity; hydrophilicity; ability of the biochar to hold moisture; ability of the biochar to retain and exchange certain types of nutrients; ion exchange capacity; physical protection from environmental hazards or protozoa; presence or absence of nutrients, micronutrients, or sources of metabolic carbon; or ability of the biochar to host other symbiotic microbes or plant systems.
Description
RELATED APPLICATIONS
[0001] This application is a continuation of and claims priority to U.S. patent application Ser. No. 15/393,214 filed Dec. 28, 2016 titled BIOCHAR AS A MICROBIAL CARRIER, which claims priority to U.S. Provisional Patent Application Ser. No. 62/271,486 filed on Dec. 28, 2015 titled ADDITIVE INFUSED BIOCHARS, U.S. Provisional Patent Application Ser. No. 62/290,285 filed on Feb. 2, 2016 titled ADDITIVE INFUSED BIOCHARS, U.S. Provisional Patent Application Ser. No. 62/344,865 filed on Jun. 2, 2016 titled MINERAL SOLUBILIZING MICROORGANISMS INFUSED BIOCHARS and U.S. Provisional Patent Application Ser. No. 62/432,253 filed on Dec. 9, 2016 titled ADDITIVE INFUSED BIOCHARS, which U.S. patent application Ser. No. 15/393,214 filed Dec. 28, 2016 titled BIOCHAR AS A MICROBIAL CARRIER is a continuation-in-part of U.S. patent application Ser. No. 15/350,920 filed on Nov. 14, 2016 titled METHOD FOR ENHANCING SOIL GROWTH USING BIO-CHAR (now U.S. Pat. No. 10,093,588 that issued on Oct. 9, 2018), which is a continuation of U.S. patent application Ser. No. 14/385,986 filed on May 29, 2012, titled METHOD FOR ENHANCING SOIL GROWTH USING BIO-CHAR (now U.S. Pat. No. 9,493,380 that issued on Nov. 15, 2016), which is a 371 national stage filing of PCT US12/39862 filed May 29, 2012 titled METHOD FOR ENHANCING SOIL GROWTH USING BIO-CHAR, which is a continuation of U.S. patent application Ser. No. 13/154,213 filed on Jun. 6, 2011, titled METHOD FOR ENHANCING SOIL GROWTH USING BIO-CHAR (now U.S. Pat. No. 8,317,891 that issued on Nov. 27, 2012), which U.S. patent application Ser. No. 15/393,214 filed Dec. 28, 2016 titled BIOCHAR AS A MICROBIAL CARRIER is also a continuation-in-part of U.S. patent application Ser. No. 15/156,256, filed on May 16, 2016, titled ENHANCED BIOCHAR (now U.S. Pat. No. 9,809,502 that issued Nov. 7, 2017), which claims priority to U.S. Provisional Patent Application No. 62/162,219, filed on May 15, 2015, titled ENHANCED BIOCHAR, which U.S. patent application Ser. No. 15/393,214 filed Dec. 28, 2016 titled BIOCHAR AS A MICROBIAL CARRIER is also a continuation-in-part of U.S. patent application Ser. No. 14/873,053 filed on Oct. 1, 2015, titled BIOCHARS AND BIOCHAR TREATMENT PROCESSES, which claims priority to U.S. Provisional Patent Application No. 62/058,445, filed on Oct. 1, 2014, titled METHODS, MATERIALS AND APPLICATIONS FOR CONTROLLED POROSITY AND RELEASE STRUCTURES AND APPLICATIONS and U.S. Provisional Patent Application No. 62/058,472, filed on Oct. 1, 2014, titled HIGH ADDITIVE RETENTION BIOCHARS, METHODS AND APPLICATIONS, and which U.S. patent application Ser. No. 15/393,214 filed Dec. 28, 2016 titled BIOCHAR AS A MICROBIAL CARRIER is also a continuation-in-part of U.S. patent application Ser. No. 14/036,480, filed on Sep. 25, 2013, titled METHOD FOR PRODUCING NEGATIVE CARBON FUEL (now U.S. Pat. No. 9,359,268 that issued Jun. 7, 2016), which is a continuation of U.S. patent application Ser. No. 13/189,709, filed on Jul. 25, 2011 (now U.S. Pat. No. 8,568,493), all of the above of which are incorporated in their entirety by reference in this application.
FIELD OF INVENTION
[0002] The invention relates to porous carbonaceous structures having increased capabilities to retain additives for use in applications, including, but not limited to, agricultural applications. These additives include but are not limited to beneficial nutrients, substances, microbes, or enzymes. The additives can be incorporated with the biochar in various ways, including but not limited to infusing the biochar with the additives to provide for the gradual delivery of the additive to the surrounding environment, such as soil.
BACKGROUND
[0003] During the last decades, soil degradation has increased due to deforestation, agricultural activities, industrial activities, vegetation overexploitation and excessive grazing. To avoid and potentially reverse soil degradation, many different types of soil enhancers have been developed and are already being used today. Among the most common soil enhancers are fertilizers. Fertilizers improve the supply of nutrients in the soil, directly affecting plant growth. However, despite the wide spread use of fertilizers, conventional fertilizers are inefficient, particularly in soils with low cation exchange capacities and humid climate conditions. The demand for frequent application, susceptibility to being washed out/leaching, and the need for nutrients to be constantly replenished are a few of the many problems associated with conventional fertilizers. Therefore, controlled released fertilizers (CRF) have become the preferred type of fertilizers to improve nutrient yield while minimizing losses. CRF's have the ability to supply nutrients gradually to soil and plants over a longer period of time. By coinciding with the nutrient requirements of the plant, CRF's ensure improved effectiveness through minimizing the losses between application and absorption, thus avoiding losses by leaching, runoff, and nutrient volatilization.
[0004] A second known type of soil enhancer consists of microbes, such as beneficial fungi or plant growth promoting bacteria ("PGPB"). PGPB are rhizosphere-associated organisms that colonize the rhizosphere and rhizoplane and improve plant growth when artificially inoculated into soil. PGPB can both promote plant growth and fight pathogenic fungi. Current methods for deploying microbes into the environment often lead to the microbes being compromised and even dying before they can be fully incorporated into the environment. Thus a system or method to deploy them which will better maintain their viability and effectiveness is needed for the industry to fully realize the benefits of microbial use.
[0005] A third known type of soil enhancer is biochar. Biochar has been known for many years as a soil enhancer. It contains highly porous, high carbon content material similar to the type of very dark, fertile anthropogenic soil found in the Amazon Basin known as Terra Preta, which has very high carbon content and historically has been made from a mixture of charcoal, bone, and manure. Biochar is created by the pyrolysis of biomass, which generally involves heating and/or burning of organic matter, in a reduced oxygen environment, at a predetermined rate. Such heating and/or burning is stopped when the matter reaches a charcoal like stage. The highly porous material of biochar is suited to host beneficial microbes, retain nutrients, hold water, and act as a delivery system for a range of beneficial compounds and additives suited to specific applications.
[0006] Raw biochar, while known for its soil enhancing characteristics, does not always benefit soil and, depending upon the biomass from which the biochar is produced and the method of production, can potentially be harmful to the soil, making it unsuitable for various types of crops or other productive uses. In particular, biochar can be detrimental, or even toxic, to 1) soil microbes involved in nutrient transport to the plant; 2) plants and 3) humans. Biochars derived from different biomass or produced with differing parameters, such as higher or lower pyrolysis temperature or variations in residence time, will have different physical and chemical properties and can behave quite differently when used in agriculture. For example, biochar having pH levels too high, containing too much ash, inorganics, or containing toxins or heavy metal content too high can be harmful and/or have minimal benefit to the soil and the plant life it supports. Biochar can also contain unacceptable levels of residual organic compounds such as acids, esters, ethers, ketones, alcohols, sugars, phenyls, alkanes, alkenes, phenols, polychlorinated biphenyls or poly or mono aromatic hydrocarbons which are either toxic or not beneficial to plant or animal life.
[0007] Due to the unpredictable performance of biochar and its potential to be detrimental to plant life and growth, it has mostly been a scientific curiosity, not found wide spread use, not found large scale commercial application, and has been relegated to small niche applications. It is, however, known, as noted above, that biochar, having certain characteristics can host beneficial microbes, retain nutrients, hold water, and act as a delivery system for a range of beneficial compounds suited to specific applications. Thus, it has been a continued desire to capture the beneficial soil enhancing characteristics of biochar in a more consistent, predictable way. Biochar research has continued in an attempt to harness biochar having predictable, controllable, and beneficial results as a soil amendment for large scale applications.
[0008] Additionally, attempts have been made to narrowly combine the benefits of fertilizer with biochar by mixing it with, coating it with or submersing it in the fertilizer. The results of these attempts, however, have failed to adequately allow soil nutrient exposure and plant nutrient uptake to occur over a longer period of time throughout a growing season from the same application.
[0009] There are currently around 7 billion people in the world and this is expected to increase to approximately 8 billion around the year 2020. In light of both the expected worldwide population increase and the increasing environmental damage caused by ever greater levels of industrialization, it will become more and more of a challenge to feed all of the world's people, a problem that will only increase with time. Thus, a need exists, in order to feed this growing population, for a method of combining the benefits of fertilizer, beneficial fungi, PGPB or other additives with biochar in a manner that reduces the cost and impact of the frequent application of nutrients to the soil and increases agricultural productivity in a sustainable and environmentally friendly manner.
SUMMARY
[0010] The present invention relates to biochar having increased capabilities to retain and then deploy additives more effectively. The biochar may be infused with beneficial additives to allow for a more gradual, prolonged release of the compounds to the soil. This time release effect in agricultural applications can dramatically reduce the need for high frequency application in the period immediately following planting and can also increase plant growth and sustain plant life. The present invention can be used in connection with any type of beneficial additive--including, but not limited to, plant nutrients, beneficial fungi, PGPB, hormones, enzymes, bio pesticides, herbicides, fungicides, nematicides, bacteriacides, fumigants among others additives, as will be described more fully below. In addition, the biochar can make a superior microbial carrier for various applications, including but not limited to agriculture, as the properties of the biochar can improve the viability of the microbes and their effectiveness after deployment with the biochar.
[0011] The method includes producing an additive infused biochar that may contain biochar, plant nutrients, beneficial fungi, PGPB and/or other additives. The method includes impregnating at least some of the pores of the biochar with liquid additives or additives in liquid solution through an infusion process. The resulting infused biochar provides for gradual and/or steady delivery of the additives to the soil and plants. The utilization of additive infused biochar allows the delivery of more nutrients or additives per unit of biochar and also provides for a more gradual release of the additives to the surrounding soil. In turn, this enables different soils to provide an environment well suited to the long term success of the desired plant. The use of additive infused biochar results in visibly fuller plants, increases plant yield with improved vitality and longevity that can be maintained with less frequent additive application and reduced additive effectiveness from leaching or runoff.
[0012] The present invention teaches treating the biochar in a manner that forces, accelerates or assists the infusion of additives into the pores of the biochar. Treatment in this manner allows for the impregnation or inoculation of the pores of the biochar with additives, which can be beneficial for the intended use of the biochar.
[0013] In one example of an implementation of the present invention, the method for treating the biochar includes placing porous carbonaceous materials in a tank or chamber; adding an additive solution to the tank; and changing the pressure in the tank by, for example, placing the contents of the tank under a partial vacuum. In this example, the additive solution may be added to the tank either before the pressure change is applied or while the pressure change is being applied. In addition to subjecting the contents to a partial vacuum, the pores of the biochar may be impregnated with the additive solution using a surfactant solution (e.g., a liquid solution containing 0.1-20% surfactant) or ultrasonic treatment, as will be further described below. Through the above treatment methods, at least 10% or more of the pore volume of the pores of the biochar material may be filled with the additive solution within a time period where it would not otherwise be possible to achieve the same results by simply contact or immersion of the biochar with the additive solution alone.
[0014] Other devices, apparatus, systems, methods, features and advantages of the invention are or will become apparent to one with skill in the art upon examination of the following figures and detailed description. It is intended that all such additional systems, methods, features and advantages be included within this description, be within the scope of the invention, and be protected by the accompanying claims.
BRIEF DESCRIPTION OF THE FIGURES
[0015] The invention may be better understood by referring to the following figures. The components in the figures are not necessarily to scale, emphasis instead being placed upon illustrating the principles of the invention.
[0016] FIG. 1 illustrates a cross-section of one example of a raw biochar particle.
[0017] FIG. 2a is a SEM (10 KV.times.3.00K 10.0 .mu.m) of pore morphology of treated biochar made from pine.
[0018] FIG. 2b is a SEM (10 KV.times.3.00K 10.0 .mu.m) of pore morphology of treated biochar made from birch.
[0019] FIG. 2c is a SEM (10 KV.times.3.00K 10.0 .mu.m) of pore morphology of treated biochar made from coconut shells.
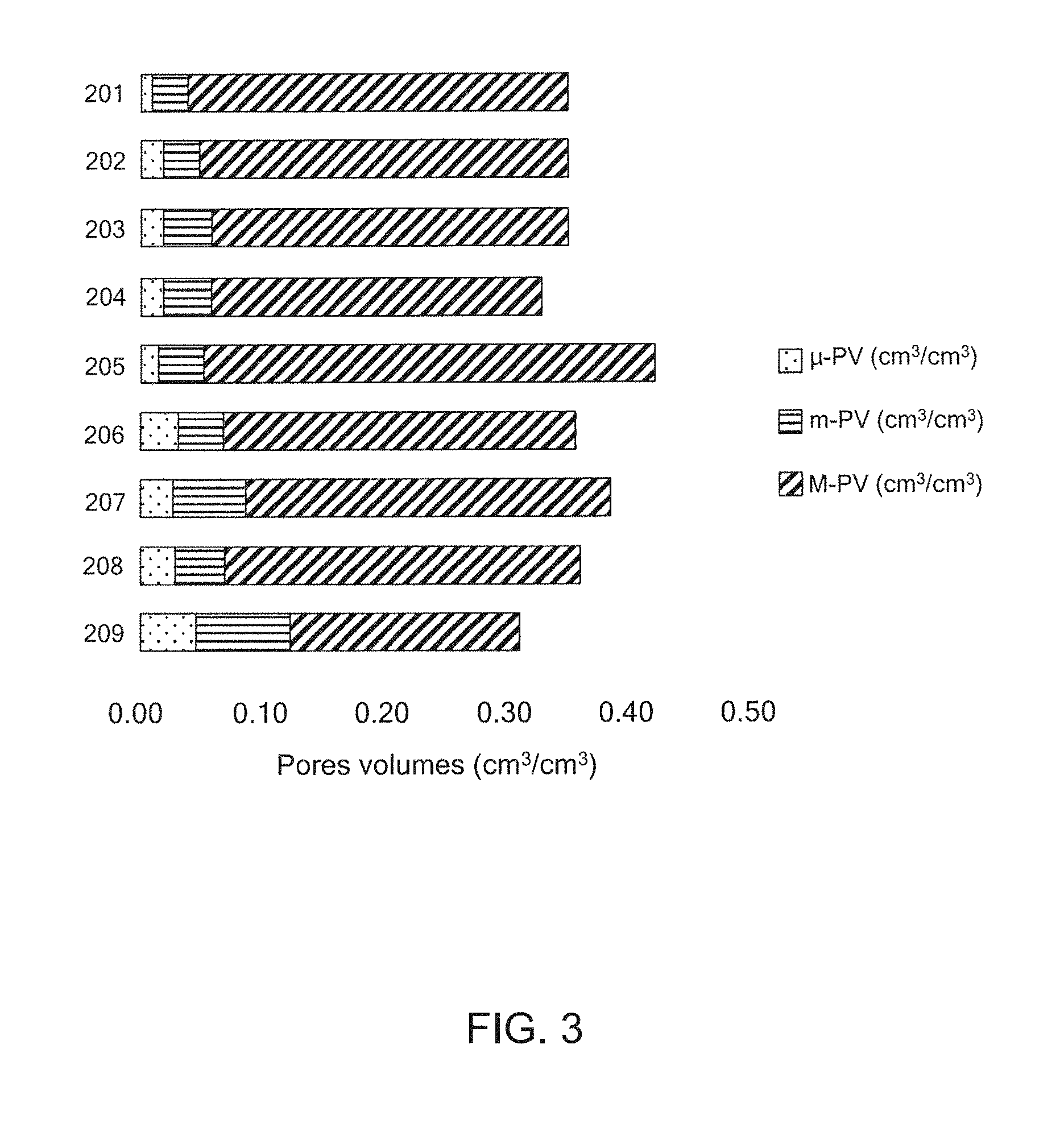
[0020] FIG. 3 is a chart showing porosity distribution of various biochars.
[0021] FIG. 4 is a flow chart process diagram of one implementation of a process for treating the raw biochar in accordance with the invention.
[0022] FIG. 4a illustrates a schematic of one example of an implementation of a biochar treat processes that that includes washing, pH adjustment and moisture adjustment.
[0023] FIG. 4b illustrates yet another example of an implementation of a biochar treatment processing that includes inoculation.
[0024] FIG. 5 is a schematic flow diagram of one example of a treatment system for use in accordance with the present invention.
[0025] FIG. 6 is a chart showing the water holding capacities of treated biochar as compared to raw biochar and sandy clay loam soil and as compared to raw biochar and sunshine potting soil.
[0026] FIG. 7 illustrates the different water retention capacities of raw biochar versus treated biochar measured gravimetrically.
[0027] FIG. 8 is a chart showing the retained water in vacuum impregnated biochar over other biochars after a seven week period.
[0028] FIG. 9 is a chart showing the weight loss of treated biochars versus raw biochar samples when heated at varying temperatures using a TGA testing method.
[0029] FIG. 10 illustrates the plant available water in raw biochar, versus treated biochar and treated dried biochar.
[0030] FIG. 11 is a graph showing the pH of various starting biochars that were made from different starting materials and pyrolysis process temperatures.
[0031] FIG. 12 is a chart showing various pH ranges and germination for treated biochars.
[0032] FIG. 13 is a Thermogravimetric Analysis (TGA) plot showing the measurement of water content, heavy organics and light organics in a sample.
[0033] FIG. 14 is a chart showing the impact of treatment on pores sizes of biochar derived from coconut.
[0034] FIG. 15 is a chart showing the impact of treatment on pores sizes of biochar derived from pine.
[0035] FIG. 16 is a chart showing the measured hydrophobicity index raw biochar, vacuum treated biochar and surfactant treated biochar.
[0036] FIG. 17 is a flow diagram showing one example of a method for infusing biochar.
[0037] FIG. 18 illustrates the improved liquid content of biochar using vacuum impregnation as against soaking the biochar in liquid.
[0038] FIG. 19a is a chart comparing total retained water of treated biochar after soaking and after vacuum impregnation.
[0039] FIG. 19b is a chart comparing water on the surface, interstitially and in the pores of biochar after soaking and after vacuum impregnation.
[0040] FIG. 20 illustrates how the amount of water or other liquid in the pores of vacuum processed biochars can be increased varied based upon the applied pressure.
[0041] FIG. 21 illustrates the effects of NPK impregnation of biochar on lettuce yield.
[0042] FIG. 22 is a chart showing nitrate release curves of treated biochars infused with nitrate fertilizer.
[0043] FIGS. 23 and 24 are images that show how different sized bacteria will fit in different biochar pore size structures.
[0044] FIG. 25 illustrates release rate data verse total pore volume data for both coconut shell and pine based treated biochars inoculated with a releasable bacteria.
[0045] FIG. 26 is a chart comparing examples of biochars.
[0046] FIGS. 27a, 27b, 27c are charts comparing different examples of biochars.
[0047] FIG. 28 is a chart comparing shoot biomass when the biochar added to a soilless mix containing soybean seeds is treated with microbial product containing bradyrhizobium japonicum, and when it is untreated.
[0048] FIG. 29 shows the comparison of root biomass in a treated verses an untreated environment.
[0049] FIG. 30 is a chart comparing the nitrogen levels when the biochar is inoculated with the rhizobial inoculant verses when it is not inoculated.
[0050] FIG. 31 illustrates the three day release rates of water infused biochar compared to other types of biochar.
[0051] FIG. 32a is a SEM (10 KV.times.3.00K 10.0 .mu.m) of pore morphology of raw biochar.
[0052] FIG. 32b is a SEM (10 KV.times.3.00K 10.0 .mu.m) of pore morphology of raw biochar of FIG. 32a after it has been infused with microbial species.
[0053] FIG. 32c is a SEM (10 KV.times.3.00K 10.0 .mu.m) of a pore morphology of another example of raw biochar of FIG. 17a after it has been infused with microbial species.
[0054] FIG. 33 contains charts illustrating improved results obtained through the use of biochars.
[0055] FIG. 34 is an example of carbon dioxide production captured as a continuous gas bubble in BGB (left two tubes) and LTB (right two tubes) growth medium.
[0056] FIGS. 35 and 36 illustrate improved growth rates of colonies of Streptomyces lydicus using biochars.
DESCRIPTION OF THE INVENTION
[0057] As illustrated in the attached figures, the present invention relates to biochar having increased capabilities to retain and then deploy additives more effectively. In addition, it includes additive infused biochars and methods for infusing biochars with additives that allow for the release of the additives into the environment gradually. When used in agricultural applications, the gradual release of the additive into the soil, can result in an increase of plant growth, vigor, or survivability, while minimizing the loss of beneficial compounds in the root zone. In particular, the biochar can make a superior microbial carrier for various applications, including but not limited to agriculture, as the properties of the biochar can improve the viability of the microbes and their effectiveness after deployment with the biochar. This present invention of the biochar as a microbial carrier can either be or not be in conjunction with the invention of the additive infusion, in which the additive infused is either the microbe itself or something else. As described below, through treatment, the properties of the raw biochar can be modified to significantly increase the biochar's ability to retain water, nutrients and additives useful for an end application while also, in many cases, creating an environment beneficial to microorganisms. Generally, for agricultural applications, such enhanced abilities could include holding water, and nutrients, e.g. fertilizer, or removing compounds, such as volatile organic compounds (VOCs), that may react with or negatively impact either the additive itself or microbial or plant life in general.
[0058] For example, through treatment, in addition to nutrients, other material additives, e.g., beneficial fungi, PGPB, herbicides and pesticides, can be utilized and benefit from the increased holding and retention capacities of the treated biochar. For certain biochar, the processing can also ensure that properties of the biochar, including its pH, used in the present application is suitable for creating conditions beneficial for plant or microbial growth, which has been a known challenge for raw biochars.
[0059] Generally, treated biochar of the present invention can be used throughout the world, in numerous soil types, agricultural applications, horticultural, large and small scale farming, organic farming, and in a variety of soil management applications and systems, and combinations and variations of these. Examples of these applications include for example, use in acidic and highly weathered tropical field soils, use in temperate soils of higher fertility, use in large commercial applications, use for the production of large scale crops such as, soybean, corn, sugarcane and rice, in forestry applications, for golf courses (e.g., greens, fairways), for general purpose turf grasses, wine grapes, table grapes, raisin grapes, fruit and nut trees, ground fruits (e.g., strawberries, blueberries, blackberries), row crops (e.g., tomatoes, celery, lettuce, leafy greens), root crops (e.g., tubers, potatoes, beets, carrots), mushrooms, and combinations and variations of these and other agricultural applications. As discussed in more detail below, biochar treated in this way may also be used in other applications, such as animal feed, composting, water treatment, and heavy metal remediation, to name a few.
[0060] For purposes of this application, the term "biochar" shall be given its broadest possible meaning and shall include any solid carbonaceous materials obtained from the pyrolysis, torrefaction, gasification or any other thermal and/or chemical conversion of a biomass. For purposes of this application, the solid carbonaceous material may include, but not be limited to, BMF char disclosed and taught by U.S. Pat. No. 8,317,891, which is incorporated into this application by reference. Pyrolysis is generally defined as a thermochemical decomposition of organic material at elevated temperatures in the absence of, or with reduced levels of oxygen. When the biochar is referred to as "treated" or undergoes "treatment," it shall mean raw, pyrolyzed biochar that has undergone additional physical, biological, and/or chemical processing.
[0061] As used herein, unless specified otherwise, the terms "carbonaceous", "carbon based", "carbon containing", and similar such terms are to be given their broadest possible meaning, and would include materials containing carbon in various states, crystallinities, forms and compounds.
[0062] As used herein, unless stated otherwise, room temperature is 25.degree. C. And, standard temperature and pressure is 25.degree. C. and 1 atmosphere. Unless stated otherwise, generally, the term "about" is meant to encompass a variance or range of .+-.10%, the experimental or instrument error associated with obtaining the stated value, and preferably the larger of these.
[0063] A. Biochars
[0064] Typically, biochars include porous carbonaceous materials, such as charcoal, that are used as soil amendments or other suitable applications. Biochar most commonly is created by pyrolysis of a biomass. In addition to the benefits to plant growth, yield and quality, etc.; biochar provides the benefit of reducing carbon dioxide (CO.sub.2) in the atmosphere by serving as a method of carbon sequestration. Thus, biochar has the potential to help mitigate climate change, via carbon sequestration. However, to accomplish this important, yet ancillary benefit, to any meaningful extent, the use of biochar in agricultural applications must become widely accepted, e.g., ubiquitous. Unfortunately, because of the prior failings in the biochar arts, this has not occurred. It is believed that with the solutions of the present invention may this level of use of biochar be achieved; and more importantly, yet heretofore unobtainable, realize the benefit of significant carbon dioxide sequestration.
[0065] In general, one advantage of putting biochar in soil includes long term carbon sequestration. It is theorized that as worldwide carbon dioxide emissions continue to mount, benefits may be obtained by, controlling, mitigating and reducing the amount of carbon dioxide in the atmosphere and the oceans. It is further theorized that increased carbon dioxide emissions are associated with the increasing industrial development of developing nations, and are also associated with the increase in the world's population. In addition to requiring more energy, the increasing world population will require more food. Thus, rising carbon dioxide emissions can be viewed as linked to the increasing use of natural resources by an ever increasing global population. As some suggest, this larger population brings with it further demands on food production requirements. Biochar uniquely addresses both of these issues by providing an effective carbon sink, e.g., carbon sequestration agent, as well as, an agent for improving and increasing agricultural output. In particular, biochar is unique in its ability to increase agricultural production, without increasing carbon dioxide emission, and preferably reducing the amount of carbon dioxide in the atmosphere. However, as discussed above, this unique ability of biochar has not been realized, or seen, because of the inherent problems and failings of prior biochars including, for example, high pH, phytotoxicity due to high metals content and/or residual organics, and dramatic product inconsistencies.
[0066] Biochar can be made from basically any source of carbon, for example, from hydrocarbons (e.g., petroleum based materials, coal, lignite, peat) and from a biomass (e.g., woods, hardwoods, softwoods, waste paper, coconut shell, manure, chaff, food waste, etc.). Combinations and variations of these starting materials, and various and different members of each group of starting materials can be, and are, used. Thus, the large number of vastly different starting materials leads to biochars having different properties.
[0067] Many different pyrolysis or carbonization processes can be, and are used, to create biochars. In general, these processes involve heating the starting material under positive pressure, reduced pressure, vacuum, inert atmosphere, or flowing inert atmosphere, through one or more heating cycles where the temperature of the material is generally brought above about 400.degree. C., and can range from about 300.degree. C. to about 900.degree. C. The percentage of residual carbon formed and several other initial properties are strong functions of the temperature and time history of the heating cycles. In general, the faster the heating rate and the higher the final temperature the lower the char yield. Conversely, in general, the slower the heating rate or the lower the final temperature the greater the char yield. The higher final temperatures also lead to modifying the char properties by changing the inorganic mineral matter compositions, which in turn, modify the char properties. Ramp, or heating rates, hold times, cooling profiles, pressures, flow rates, and type of atmosphere can all be controlled, and typically are different from one biochar supplier to the next. These differences potentially lead to a biochar having different properties, further framing the substantial nature of one of the problems that the present inventions address and solve. Generally, in carbonization most of the non-carbon elements, hydrogen and oxygen are first removed in gaseous form by the pyrolytic decomposition of the starting materials, e.g., the biomass. The free carbon atoms group or arrange into crystallographic formations known as elementary graphite crystallites. Typically, at this point the mutual arrangement of the crystallite is irregular, so that free interstices exist between them. Thus, pyrolysis involves thermal decomposition of carbonaceous material, e.g., the biomass, eliminating non-carbon species, and producing a fixed carbon structure.
[0068] As noted above, raw or untreated biochar is generally produced by subjecting biomass to either a uniform or varying pyrolysis temperature (e.g., 300.degree. C. to 550.degree. C. to 750.degree. C. or more) for a prescribed period of time in a reduced oxygen environment. This process may either occur quickly, with high reactor temperature and short residence times, slowly with lower reactor temperatures and longer residence times, or anywhere in between. To achieve better results, the biomass from which the char is obtained may be first stripped of debris, such as bark, leaves and small branches, although this is not necessary. The biomass may further include feedstock to help adjust the pH and particle size distribution in the resulting raw biochar. In some applications, it is desirous to have biomass that is fresh, less than six months old, and with an ash content of less than 3%. Further, by using biochar derived from different biomass, e.g., pine, oak, hickory, birch and coconut shells from different regions, and understanding the starting properties of the raw biochar, the treatment methods can be tailored to ultimately yield a treated biochar with predetermined, predictable physical and chemical properties.
[0069] In general, biochar particles can have a very wide variety of particle sizes and distributions, usually reflecting the sizes occurring in the input biomass. Additionally, biochar can be ground or crushed after pyrolysis to further modify the particle sizes. Typically, for agricultural uses, biochars with consistent, predictable particle sizes are more desirable. By way of example, the biochar particles can have particle sizes as shown or measured in Table 1 below. When referring to a batch having 1/4 inch particles, the batch would have particles that will pass through a 3 mesh sieve, but will not pass through (i.e., are caught by or sit atop) a 4 mesh sieve.
TABLE-US-00001 TABLE 1 U.S. Mesh Microns Millimeters (i.e., mesh) Inches (.mu.m) (mm) 3 0.2650 6730 6.370 4 0.1870 4760 4.760 5 0.1570 4000 4.000 6 0.1320 3360 3.360 7 0.1110 2830 2.830 8 0.0937 2380 2.380 10 0.0787 2000 2.000 12 0.0661 1680 1.680 14 0.0555 1410 1.410 16 0.0469 1190 1.190 18 0.0394 1000 1.000 20 0.0331 841 0.841 25 0.0280 707 0.707 30 0.0232 595 0.595 35 0.0197 500 0.500 40 0.0165 400 0.400 45 0.0138 354 0.354 50 0.0117 297 0.297 60 0.0098 250 0.250 70 0.0083 210 0.210 80 0.0070 177 0.177 100 0.0059 149 0.149 120 0.0049 125 0.125 140 0.0041 105 0.105 170 0.0035 88 0.088 200 0.0029 74 0.074 230 0.0024 63 0.063 270 0.0021 53 0.053 325 0.0017 44 0.044 400 0.0015 37 0.037
[0070] For most applications, it is desirable to use biochar particles having particle sizes from about 3/4 mesh to about 60/70 mesh, about 4/5 mesh to about 20/25 mesh, or about 4/5 mesh to about 30/35 mesh. It being understood that the desired mesh size, and mesh size distribution can vary depending upon a particular application for which the biochar is intended.
[0071] FIG. 1 illustrates a cross-section of one example of a raw biochar particle. As illustrated in FIG. 1, a biochar particle 100 is a porous structure that has an outer surface 100a and a pore structure 101 formed within the biochar particle 100. As used herein, unless specified otherwise, the terms "porosity", "porous", "porous structure", and "porous morphology" and similar such terms are to be given their broadest possible meaning, and would include materials having open pores, closed pores, and combinations of open and closed pores, and would also include macropores, mesopores, and micropores and combinations, variations and continuums of these morphologies. Unless specified otherwise, the term "pore volume" is the total volume occupied by the pores in a particle or collection of particles; the term "inter-particle void volume" is the volume that exists between a collection of particle; the term "solid volume or volume of solid means" is the volume occupied by the solid material and does not include any free volume that may be associated with the pore or inter-particle void volumes; and the term "bulk volume" is the apparent volume of the material including the particle volume, the inter-particle void volume, and the internal pore volume.
[0072] The pore structure 101 forms an opening 121 in the outer surface 100a of the biochar particle 100. The pore structure 101 has a macropore 102, which has a macropore surface 102a, and which surface 102a has an area, i.e., the macropore surface area. (In this diagram only a single micropore is shown. If multiple micropores are present than the sum of their surface areas would equal the total macropore surface area for the biochar particle.) From the macropore 102, several mesopores 105, 106, 107, 108 and 109 are present, each having its respective surfaces 105a, 106a, 107a, 108a and 109a. Thus, each mesopore has its respective surface area; and the sum of all mesopore surface areas would be the total mesopore surface area for the particle. From the mesopores, e.g., 107, there are several micropores 110, 111, 112, 113, 114, 115, 116, 117, 118, 119 and 120, each having its respective surfaces 110a, 111a, 112a, 113a, 114a, 115a, 116a, 117a, 118a, 119a and 120a. Thus, each micropore has its respective surface area and the sum of all micropore surface areas would be the total micropore surface area for the particle. The sum of the macropore surface area, the mesopore surface area and the micropore surface area would be the total pore surface area for the particle.
[0073] Macropores are typically defined as pores having a diameter greater than 300 nm, mesopores are typically defined as diameter from about 1-300 nm, and micropores are typically defined as diameter of less than about 1 nm, and combinations, variations and continuums of these morphologies. The macropores each have a macropore volume, and the sum of these volumes would be the total macropore volume. The mesopores each have a mesopore volume, and the sum of these volumes would be the total mesopore volume. The micropores each have a micropore volume, and the sum of these volumes would be the total micropore volume. The sum of the macropore volume, the mesopore volume and the micropore volume would be the total pore volume for the particle.
[0074] Additionally, the total pore surface area, volume, mesopore volume, etc., for a batch of biochar would be the actual, estimated, and preferably calculated sum of all of the individual properties for each biochar particle in the batch.
[0075] It should be understood that the pore morphology in a biochar particle may have several of the pore structures shown, it may have mesopores opening to the particle surface, it may have micropores opening to particle surface, it may have micropores opening to macropore surfaces, or other combinations or variations of interrelationship and structure between the pores. It should further be understood that the pore morphology may be a continuum, where moving inwardly along the pore from the surface of the particle, the pore transitions, e.g., its diameter becomes smaller, from a macropore, to a mesopore, to a micropore, e.g., macropore 102 to mesopore 109 to micropore 114.
[0076] In general, the biochars have porosities that can range from 0.2 cm.sup.3/cm.sup.3 to about 0.8 cm.sup.3/cm.sup.3 and more preferably about 0.2 cm.sup.3/cm.sup.3 to about 0.5 cm.sup.3/cm.sup.3. (Unless stated otherwise, porosity is provided as the ratio of the total pore volumes (the sum of the micro+meso+macro pore volumes) to the solid volume of the biochar. Porosity of the biochar particles can be determined, or measured, by measuring the micro-, meso-, and macro pore volumes, the bulk volume, and the inter particle volumes to determine the solid volume by difference. The porosity is then calculated from the total pore volume and the solid volume.
[0077] As noted above, the use of different biomass potentially leads to biochars having different properties, including, but not limited to different pore structures. By way of example, FIGS. 2A, 2B and 2C illustrate Scanning Electron Microscope ("SEM") images of various types of treated biochars showing the different nature of their pore morphology. FIG. 2A is biochar derived from pine. FIG. 2B is biochar derived from birch. FIG. 2C is biochar derived from coconut shells.
[0078] The surface area and pore volume for each type of pore, e.g., macro-, meso- and micro can be determined by direct measurement using CO.sub.2 adsorption for micro-, N.sub.2 adsorption for meso- and macro pores and standard analytical surface area analyzers and methods, for example, particle analyzers such as Micrometrics instruments for meso- and micro pores and impregnation capacity for macro pore volume. Mercury porosimetry, which measures the macroporosity by applying pressure to a sample immersed in mercury at a pressure calibrated for the minimum pore diameter to be measured, may also be used to measure pore volume.
[0079] The total micropore volume can be from about 2% to about 25% of the total pore volume. The total mesopore volume can be from about 4% to about 35% of the total pore volume. The total macropore volume can be from about 40% to about 95% of the total pore volume. By way of example, FIG. 3 shows a bar chart setting out examples of the pore volumes for sample biochars made from peach pits 201, juniper wood 202, a first hard wood 203, a second hard wood 204, fir and pine waste wood 205, a first pine 206, a second pine 207, birch 208 and coconut shells 209.
[0080] As explained further below, treatment can increase usable pore volumes and, among other things, remove obstructions in the pores, which leads to increased retention properties and promotes further performance characteristics of the biochar. Knowing the properties of the starting raw biochar, one can treat the biochar to produce controlled, predictable and optimal resulting physical and chemical properties.
[0081] B. Treatment
[0082] The rationale for treating the biochar after pyrolysis is that given the large pore volume and large surface are of the biochars, it is most efficient to make significant changes in the physical and chemical properties of the biochar by treating both the internal and external surfaces and internal pore volume of the char. Testing has demonstrated that if the biochar is treated, at least partially, in a manner that causes the forced infusion and/or diffusion of liquids into and/or out of the biochar pores (through mechanical, physical, or chemical means), certain properties of the biochar can be altered or improved over and above simply contacting these liquids with the biochar. By knowing the properties of the raw biochar and the optimal desired properties of the treated biochar, the raw biochar can then be treated in a manner that results in the treated biochar having controlled optimized properties.
[0083] For purposes of this application, treating and/or washing the biochar in accordance with the present invention involves more than a simple wash or soak, which generally only impacts the exterior surfaces and a small percentage of the interior surface area. "Washing" or "treating" in accordance with the present invention, and as used below, involves treatment of the biochar in a manner that causes the forced, accelerated or assisted infusion and/or diffusion of liquids and/or additivities into and/or out of the biochar pores (through mechanical, physical, or chemical means) such that certain properties of the biochar can be altered or improved over and above simply contacting these liquids with the biochar or so that treatment becomes more efficient or rapid from a time standpoint over simple contact or immersion.
[0084] In particular, effective treatment processes can mitigate deleterious pore surface properties, remove undesirable substances from pore surfaces or volume, and impact anywhere from between 10% to 99% or more of pore surface area of a biochar particle. By modifying the usable pore surfaces through treatment and removing deleterious substances from the pore volume, the treated biochars can exhibit a greater capacity to retain water and/or other nutrients as well as being more suitable habitats for some forms of microbial life. Through the use of treated biochars, agricultural applications can realize increased moisture control, increased nutrient retention, reduced water usage, reduced water requirements, reduced runoff or leaching, increased nutrient efficiency, reduced nutrient usage, increased yields, increased yields with lower water requirements and/or nutrient requirements, increases in beneficial microbial life, improved performance and/or shelf life for inoculated bacteria, and any combination and variation of these and other benefits.
[0085] Treatment further allows the biochar to be modified to possess certain known properties that enhance the benefits received from the use of biochar. While the selection of feedstock, raw biochar and/or pyrolysis conditions under which the biochar was manufactured can make treatment processes less cumbersome, more efficient and further controlled, treatment processes can be utilized that provide for the biochar to have desired and generally sustainable resulting properties regardless of the biochar source or pyrolysis conditions. As explained further below, treatment can (i) repurpose problematic biochars, (ii) handle changing biochar material sources, e.g., seasonal and regional changes in the source of biomass, (iii) provide for custom features and functions of biochar for particular soils, regions or agricultural purposes; (iv) increase the retention properties of biochar, (v) provide for large volumes of biochar having desired and predictable properties, (vi) provide for biochar having custom properties, (vii) handle differences in biochar caused by variations in pyrolysis conditions or manufacturing of the "raw" biochar; and (viii) address the majority, if not all, of the problems that have, prior to the present invention, stifled the large scale adoption and use of biochars.
[0086] Treatment can wash both the interior and exterior pore surfaces, remove harmful chemicals, introduce beneficial substances, and alter certain properties of the biochar and the pore surfaces and volumes. This is in stark contrast to simple washing which generally only impacts the exterior surfaces and a small percentage of the interior surface area. Treatment can further be used to coat substantially all of the biochar pore surfaces with a surface modifying agent or impregnate the pore volume with additives or treatment to provide a predetermined feature to the biochar, e.g., surface charge and charge density, surface species and distribution, targeted nutrient addition, magnetic modifications, root growth facilitator, and water absorptivity and water retention properties. Just as importantly, treatment can also be used to remove undesirable substances from the biochar, such as dioxins or other toxins either through physical removal or through chemical reactions causing neutralization.
[0087] FIG. 4 is a schematic flow diagram of one example treatment process 400 for use in accordance with the present invention. As illustrated, the treatment process 400 starts with raw biochar 402 that may be subjected to one or more reactors or treatment processes prior to bagging 420 the treated biochar for resale. For example, 404 represents reactor 1, which may be used to treat the biochar. The treatment may be a simple water wash or may be an acid wash used for the purpose of altering the pH of the raw biochar particles 402. The treatment may also contain a surfactant or detergent to aid the penetration of the treatment solution into the pores of the biochar. The treatment may optionally be heated, cooled, or may be used at ambient temperature or any combination of the three. For some applications, depending upon the properties of the raw biochar, a water and/or acid/alkaline wash 404 (the latter for pH adjustment) may be the only necessary treatment prior to bagging the biochar 420. If, however, the moisture content of the biochar needs to be adjusted, the treated biochar may then be put into a second reactor 406 for purposes of reducing the moisture content in the washed biochar. From there, the treated and moisture adjusted biochar may be bagged 420.
[0088] Again, depending upon the starting characteristics of the raw biochar and the intended application for the resale product, further processing may still be needed or desired. In this case, the treated moisture adjusted biochar may then be passed to a third reactor 408 for inoculation, which may include the impregnation of biochar with beneficial additives, such as nutrients, bacteria, microbes, fertilizers or other additives. Thereafter, the inoculated biochar may be bagged 420, or may be yet further processed, for example, in a fourth reactor 410 to have further moisture removed from or added to the biochar. Further moisture adjustment may be accomplished by placing the inoculated biochar in a fourth moisture adjustment reactor 410 or circulating the biochar back to a previous moisture adjustment reactor (e.g. reactor 406). Those skilled in the art will recognize that the ordering in which the raw biochar is processed and certain processes may be left out, depending on the properties of the starting raw biochar and the desired application for the biochar. For example, the treatment and inoculation processes may be performed without the moisture adjustment step, inoculation processes may also be performed with or without any treatment, pH adjustment or any moisture adjustment. All the processes may be completed alone or in the conjunction with one or more of the others.
[0089] For example, FIG. 4a illustrates a schematic of one example of an implementation of biochar processing that includes washing the pores and both pH and moisture adjustment. FIG. 4b illustrates yet another example of an implementation of biochar processing that includes inoculation.
[0090] As illustrated in FIG. 4a, raw biochar 402 is placed into a reactor or tank 404. A washing or treatment liquid 403 is then added to a tank and a partial vacuum, using a vacuum pump, 405 is pulled on the tank. The treating or washing liquid 403 may be used to clean or wash the pores of the biochar 402 or adjust the chemical or physical properties of the surface area or pore volume, such as pH level, usable pore volume, or VOC content, among other things. The vacuum can be applied after the treatment liquid 403 is added or while the treatment liquid 403 is added. Thereafter, the washed/adjusted biochar 410 may be moisture adjusted by vacuum exfiltration 406 to pull the extra liquid from the washed/moisture adjusted biochar 410 or may be placed in a centrifuge 407, heated or subjected to pressure gradient changes (e.g., blowing air) for moisture adjustment. The moisture adjusted biochar 412 may then be bagged or subject to further treatment. Any excess liquids 415 collected from the moisture adjustment step may be disposed of or recycled, as desired. Optionally, biochar fines may be collected from the excess liquids 415 for further processing, for example, to create a slurry, cakes, or biochar extrudates.
[0091] Optionally, rather than using a vacuum pump 405, a positive pressure pump may be used to apply positive pressure to the tank 404. In some situations, applying positive pressure to the tank may also function to force or accelerate the washing or treating liquid 403 into the pores of the biochar 402. Any change in pressure in the tank 404 or across the surface of the biochar could facilitate the exchange of gas and/or moisture into and out of the pores of the biochar with the washing or treating liquid 403 in the tank. Accordingly, changing the pressure in the tank and across the surface of the biochar, whether positive or negative, is within the scope of this invention.
[0092] As illustrated FIG. 4b, the washed/adjusted biochar 410 or the washed/adjusted and moisture adjusted biochar 412 may be further treated by inoculating or impregnating the pores of the biochar with an additive 425. The biochar 410, 412 placed back in a reactor 401, an additive solution 425 is placed in the reactor 401 and a vacuum, using a vacuum pump, 405 is applied on the tank. Again, the vacuum can be applied after the additive solution 425 is added to the tank or while the additive solution 425 is being added to the tank. Thereafter, the washed, adjusted and inoculated biochar 428 can be bagged. Alternatively, if further moisture adjustment is required, the biochar can be further moisture adjusted by vacuum filtration 406 to pull the extra liquid from the washed/moisture adjusted biochar 410 or may be placed in a centrifuge 407 for moisture adjustment. The resulting biochar 430 can then be bagged. Any excess liquids 415 collected from the moisture adjustment step may be disposed of or recycled, as desired. Optionally, biochar particulates or "fines" which easily are suspended in liquid may be collected from the excess liquids 415 for further processing, for example, to create a slurry, biochar extrudates, or merely a biochar product of a consistently smaller particle size. As described above, both processes of the FIGS. 4a and 4b can be performed with a surfactant solution in place of, or in conjunction with, the vacuum 405.
[0093] While known processes exist for the above described processes, research associated with the present invention has shown improvement and the ability to better control the properties and characteristics of the biochar if the processes are performed through the infusion and diffusion of liquids into and out of the biochar pores. One such treatment process that can be used is vacuum impregnation and vacuum and/or centrifuge extraction. Another such treatment process that can be used is the addition of a surfactant to infused liquid, which infused liquid may be optionally heated, cooled, or used at ambient temperature or any combination of the three.
[0094] Since research associated with the present invention has identified what physical and chemical properties have the highest impact on plant growth and/or soil health, the treatment process can be geared to treat different forms of raw biochar to achieve treated biochar properties known to enhance these characteristics. For example, if the pH of the biochar needs to be adjusted to enhance the raw biochar performance properties, the treatment may be the infusion of an acid solution into the pores of the biochar using vacuum, surfactant, or other treatment means. This treatment of pore infusion through, for example, the rapid, forced infusion of liquid into and out the pores of the biochar, has further been proven to sustain the adjusted pH levels of the treated biochar for much longer periods than biochar that is simply immersed in an acid solution for the same period of time. By way of another example, if the moisture content needs to be adjusted, then excess liquid and other selected substances (e.g. chlorides, dioxins, and other chemicals, to include those previously deposited by treatment to catalyze or otherwise react with substances on the interior or exterior surfaces of the biochar) can be extracted from the pores using vacuum and/or centrifuge extraction or by using various heating techniques. The above describes a few examples of treatment that result in treated biochar having desired performance properties identified to enhance soil health and plant life.
[0095] FIG. 5 illustrates one example of a system 500 that utilizes vacuum impregnation to treat raw biochar. Generally, raw biochar particles, and preferably a batch of biochar particles, are placed in a reactor, which is connected to a vacuum pump, and a source of treating liquid (i.e. water or acidic/basis solution). When the valve to the reactor is closed, the pressure in the reactor is reduced to values ranging from 750 Torr to 400 Torr to 10 Torr or less. The biochar is maintained under vacuum ("vacuum hold time") for anywhere from seconds to 1 minute to 10 minutes, to 100 minutes, or possibly longer. By way of example, for about a 500 pound batch of untreated biochar, a vacuum hold time of from about 1 to about 5 minutes can be used if the reactor is of sufficient size and sufficient infiltrate is available to adjust the necessary properties. While under the vacuum the treating liquid may then be introduced into the vacuum chamber containing the biochar. Alternatively, the treating liquid may be introduced into the vacuum chamber before the biochar is placed under a vacuum. Optionally, treatment may also include subjecting the biochar to elevated temperatures from ambient to about 250.degree. C. or reduced temperatures to about -25.degree. C. or below, with the limiting factor being the temperature and time at which the infiltrate can remain flowable as a liquid or semi-liquid.
[0096] The infiltrate or treating liquid is drawn into the biochar pore, and preferably drawn into the macropores and mesopores. Depending upon the specific doses applied and pore structure of the biochar, the infiltrate can coat anywhere from 10% to 50% to 100% of the total macropore and mesopore surface area and can fill or coat anywhere from a portion to nearly all (10%-100%) of the total macropore and mesopore volume.
[0097] As described above, the treating liquid can be left in the biochar, with the batch being a treated biochar batch ready for packaging, shipment and use in an agricultural or other application. The treating liquid may also be removed through drying, subsequent vacuum processing, centrifugal force (e.g., cyclone drying machines or centrifuges), with the batch being a treated biochar batch ready for packaging, shipment and use in an agricultural application. A second, third or more infiltration, removal, infiltration and removal, and combinations and variations of these may also be performed on the biochar with optional drying steps between infiltrations to remove residual liquid from and reintroduce gasses to the pore structure if needed. In any of these stages the liquid may contain organic or inorganic surfactants to assist with the penetration of the treating liquid.
[0098] As illustrated in FIG. 5, a system 500 for providing a biochar, preferably having predetermined and uniform properties. The system 500 has a vacuum infiltration tank 501. The vacuum infiltration tank 501 has an inlet line 503 that has a valve 504 that seals the inlet line 503. In operation, the starting biochar is added to vacuum infiltration tank 501 as shown by arrow 540. Once the tank is filled with the starting biochar, a vacuum is pulled on the tank, by a vacuum pump connected to vacuum line 506, which also has valve 507. The starting biochar is held in the vacuum for a vacuum hold time. Infiltrate, as shown by arrow 548 is added to the tank 501 by line 508 having valve 509. The infiltrate is mixed with the biochar in the tank 501 by agitator 502. The mixing process is done under vacuum for a period of time sufficient to have the infiltrate fill the desired amount of pore volume, e.g., up to 100% of the macropores and mesopores.
[0099] Alternatively, the infiltrate may be added to the vacuum infiltration tank 501 before vacuum is pulled on the tank. In this manner, infiltrate is added in the tank in an amount that can be impregnated into the biochar. As the vacuum is pulled, the biochar is circulated in the tank to cause the infiltrate to fill the pore volume. To one skilled in the art, it should be clear that the agitation of the biochar during this process can be performed through various means, such as a rotating tank, rotating agitator, pressure variation in the tank itself, or other means. Additionally, the biochar may be dried using conventional means before even the first treatment. This optional pre-drying can remove liquid from the pores and in some situations may increase the efficiency of impregnation due to pressure changes in the tank.
[0100] Pressure is then restored in the tank 501 and the infiltrated biochar is removed, as shown by arrow 541, from the tank 501 to bin 512, by way of a sealing gate 511 and removal line 510. The infiltrated biochar is collected in bin 512, where it can be further processed in several different ways. The infiltrated biochar can be shipped for use as a treated biochar as shown by arrow 543. The infiltrated biochar can be returned to the tank 501 (or a second infiltration tank). If returned to the tank 501 the biochar can be processed with a second infiltration step, a vacuum drying step, a washing step, or combinations and variations of these. The infiltrated biochar can be moved by conveyor 514, as shown by arrow 542, to a drying apparatus 516, e.g., a centrifugal dryer or heater, where water, infiltrate or other liquid is removed by way of line 517, and the dried biochar leaves the dryer through discharge line 518 as shown by arrow 545, and is collected in bin 519. The biochar is removed from the bin by discharge 520. The biochar may be shipped as a treated biochar for use in an agriculture application, as shown by arrow 547. The biochar may also be further processed, as shown by 546. Thus, the biochar could be returned to tank 501 (or a second vacuum infiltration tank) for a further infiltration step. The drying step may be repeated either by returning the dry biochar to the drying apparatus 516, or by running the biochar through a series of drying apparatus, until the predetermined dryness of the biochar is obtained, e.g., between 50% to less than 1% moisture.
[0101] The system 500 is illustrative of the system, equipment and processes that can be used for, and to carry out the present inventions. Various other implementations and types of equipment can be used. The vacuum infiltration tank can be a sealable off-axis rotating vessel, chamber or tank. It can have an internal agitator that also when reversed can move material out, empty it, (e.g., a vessel along the lines of a large cement truck, or ready mix truck, that can mix and move material out of the tank, without requiring the tank's orientation to be changed). Washing equipment may be added or utilized at various points in the process, or may be carried out in the vacuum tank, or drier, (e.g., wash fluid added to biochar as it is placed into the drier for removal). Other steps, such as bagging, weighing, the mixing of the biochar with other materials, e.g., fertilized, peat, soil, etc. can be carried out. In all areas of the system referring to vacuum infiltration, optionally positive pressure can be applied, if needed, to enhance the penetration of the infiltrate or to assist with re-infusion of gaseous vapors into the treated char. Additionally, where feasible, especially in positive pressure environments, the infiltrate may have soluble gasses added which then can assist with removal of liquid from the pores, or gaseous treatment of the pores upon equalization of pressure.
[0102] As noted above, the biochar may also be treated using a surfactant. The same or similar equipment used in the vacuum infiltration process can be used in the surfactant treatment process. Although it is not necessary to apply a vacuum in the surfactant treatment process, the vacuum infiltration tank or any other rotating vessel, chamber or tank can be used. In the surfactant treatment process, a surfactant, such as yucca extract, is added to the infiltrate, e.g., acid wash or water. The quantity of the surfactant added to the infiltrate may vary depending upon the surfactant used. For example, organic yucca extract can be added at a rate of between 0.1-20%, but more preferably 1-5% by volume of the infiltrate. The infiltrate with surfactant is then mixed with the biochar in a tumbler for several minutes, e.g., 3-5 minutes, without applied vacuum. Optionally, a vacuum or positive pressure may be applied with the surfactant to improve efficiency, but is not necessary. Additionally, infiltrate to which the surfactant or detergent is added may be heated or may be ambient temperature or less. Similarly, the mixture of the surfactant or detergent, as well as the char being treated may be heated, or may be ambient temperature, or less. After tumbling, excess free liquid can be removed in the same manner as described above in connection with the vacuum infiltration process. Drying, also as described above in connection with the vacuum infiltration process, is an optional additional step. Besides yucca extract, a number of other surfactants may be used for surfactant treatment, which include, but are not limited to, the following: nonionic types, such as, ethoxylated alcohols, phenols-lauryl alcohol ethoxylates, Fatty acid esters-sorbitan, tween 20, amines, amides-imidazoles; anionic types, such as sulfonates-arylalkyl sulfonates and sulfate-sodium dodecyl sulfate; cationic types, such as alkyl-amines or ammoniums-quaternary ammoniums; and amphoteric types, such as betaines-cocamidopropyl betaine.
[0103] Optionally, the biochar may also be treated by applying ultrasonics. In this treatment process, the biochar may be contacted with a treating liquid that is agitated by ultrasonic waves. By agitating the treating liquid, contaminants may be dislodged or removed from the biochar due to bulk motion of the fluid in and around the biocarbon, pressure changes, including cavitation in and around contaminants on the surface, as well as pressure changes in or near pore openings (cavitation bubbles) and internal pore cavitation.
[0104] In this manner, agitation will cause contaminants of many forms to be released from the internal and external structure of the biochar. The agitation also encourages the exchange of water, gas, and other liquids with the internal biochar structure. Contaminants are transported from the internal structure to the bulk liquid (treating fluid) resulting in biochar with improved physical and chemical properties. The effectiveness of ultrasonic cleaning is tunable as bubble size and number is a function of frequency and power delivered by the transducer to the treating fluid
[0105] In one example, applying ultrasonic treatment, raw wood based biochar between 10 microns to 10 mm with moisture content from 0% to 90% may be mixed with a dilute mixture of acetic acid and water (together the treating liquid) in a processing vessel that also translates the slurry (the biochar/treating liquid mixture). During translation, the slurry passes near an ultrasonic transducer to enhance the interaction between the fluid and biochar. The biochar may experience one or multiple washes of dilute acetic acid, water, or other treating fluids. The biochar may also make multiple passes by ultrasonic transducers to enhance physical and chemical properties of the biochar. For example, once a large volume of slurry is made, it can continuously pass an ultrasonic device and be degassed and wetted to its maximum, at a rapid processing rate. The slurry can also undergo a separation process in which the fluid and solid biochar are separated at 60% effectiveness or greater.
[0106] Through ultrasonic treatment, the pH of the biochar, or other physical and chemical properties may be adjusted and the mesopore and macropore surfaces of the biochar may be cleaned and enhanced. Further, ultrasonic treatment can be used in combination with bulk mixing with water, solvents, additives (fertilizers, etc.), and other liquid based chemicals to enhance the properties of the biochar. After treatment, the biochar may be subject to moisture adjustment, further treatment and/or inoculation using any of the methods set forth above.
[0107] C. Benefits of Treatment
[0108] As illustrated above, the treatment process, whether using vacuum, surfactant or ultrasonic treatment, or a combination thereof, may include two steps, which in certain applications, may be combined: (i) washing and (ii) inoculation of the pores with an additive. When the desired additive is the same and that being inoculated into the pores, e.g., water, the step of washing the pores and inoculating the pores with an additive may be combined.
[0109] While not exclusive, washing is generally done for one of three purposes: (i) to modify the surface of the pore structure of the biochar (i.e., to allow for increased retention of liquids); (ii) to modify the pH of the biochar; and/or (iii) to remove undesired and potentially harmful compounds or gases.
[0110] 1. Increases Water Holding Capacity/Water Retention Capacity
[0111] As demonstrated below, the treatment processes of the invention modify the surfaces of the pore structure to provide enhanced functionality and to control the properties of the biochar to achieve consistent and predicable performance. Using the above treatment processes, anywhere from at least 10% of the total pore surface area up to 90% or more of the total pore surface area may be modified. In some implementations, it may be possible to achieve modification of up to 99% or more of the total pore surface area of the biochar particle. Using the processes set forth above, such modification may be substantially and uniformly achieved for an entire batch of treated biochar.
[0112] For example, it is believed that by treating the biochar as set forth above, the hydrophilicity of the surface of the pores of the biochar is modified, allowing for a greater water retention capacity. Further, by treating the biochars as set forth above, gases and other substances are also removed from the pores of the biochar particles, also contributing to the biochar particles' increased water holding capacity. Thus, the ability of the biochar to retain liquids, whether water or additives in solution, is increased, which also increases the ability to load the biochar particles with large volumes of inoculant, infiltrates and/or additives.
[0113] A batch of biochar has a bulk density, which is defined as weight in grams (g) per cm.sup.3 of loosely poured material that has or retains some free space between the particles. The biochar particles in this batch will also have a solid density, which is the weight in grams (g) per cm.sup.3 of just particles, i.e., with the free space between the particles removed. The solid density includes the air space or free space that is contained within the pores, but not the free space between particles. The actual density of the particles is the density of the material in grams (g) per cm.sup.3 of material, which makes up the biochar particles, i.e., the solid material with pore volume removed.
[0114] In general, as bulk density increases the pore volume is expected to decrease. When the pore volume is macro or mesoporous, the ability of the material to hold infiltrate, e.g., inoculant is directly proportional to the pore volume, thus it is also expected to decrease as bulk density increases. With the infiltration processes, the treated biochars can have impregnation capacities that are larger than could be obtained without infiltration, e.g., the treated biochars can readily have 30%, 40%, 50%, or most preferably, 60%-100% of their total pore volume filled with an infiltrate, e.g., an inoculant. The impregnation capacity is the amount of a liquid that a biochar particle, or batch of particles, can absorb. The ability to make the pores surface hydrophilic, and to infuse liquid deep into the pore structure through the application of positive or negative pressure and/or a surfactant, alone or in combination, provides the ability to obtain these high impregnation capabilities. The treated biochars can have impregnation capacities, i.e., the amount of infiltrate that a particle can hold on a volume held/total volume of a particle basis, that is greater than 0.2 cm.sup.3/cm.sup.3 to 0.8 cm.sup.3/cm.sup.3. Resulting bulk densities of treated biochar can range from 0.1-0.6 g/cm.sup.3 and sometimes preferably between 0.3-0.6 g/cm.sup.3 and can have solid densities ranging from 0.2-1.2 g/cm.sup.3.
[0115] Accordingly, by using the treatment above, the water retention capacity of biochar can be greatly increased over the water retention capacities of various soil types and even raw biochar, thereby holding water and/or nutrients in the plant's root zone longer and ultimately reducing the amount of applied water (through irrigation, rainfall, or other means) needed by up to 50% or more. FIG. 6 is a chart showing the water retention capacities of soils versus raw and treated biochar. The soils sampled are loam and sandy clay soil and a common commercial horticultural mix. The charts show the retained water as a function of time.
[0116] In chart A, the bottom line represents the retained water in the sandy claim loam soil over time. The middle line represents the retained water in the sandy clay soil with 20% by volume percent of unprocessed raw biochar. The top line represents the retained water in the sandy clay loam soil with 20% by volume percent of treated biochar (adjusted and inoculated biochar). Chart B represents the same using a soilless potting mix rather than sandy clay loam soil.
[0117] As illustrated in FIG. 7, testing showed a treated biochar had an increased water retention capacity of approximately 1.5 times that of the raw biochar from the same feedstock. Similar results have been seen with biochars derived from various feedstocks. With certain biochar types, the water retention capacity of treated biochar could be as great as three times that of raw biochar.
[0118] "Water holding capacity," which may also be referred to as "Water Retention Capacity," is the amount of water that can be held both internally within the porous structure and in the interparticle void spaces in a given batch of particles. While a summary of the method of measure is provided above, a more specific method of measuring water holding capacity/water retention capacity is measured by the following procedure: (i) drying a sample of material under temperatures of 105.degree. C. for a period of 24 hours or using another scientifically acceptable technique to reduce the moisture content of the material to less than 2%, less than 1%; and preferably less than 0.5% (ii) placing a measured amount of dry material in a container; (iii) filling the container having the measured amount of material with water such that the material is completely immersed in the water; (iv) letting the water remain in the container having the measured amount of material for at least ten minutes or treating the material in accordance with the invention by infusing with water when the material is a treated biochar; (v) draining the water from the container until the water ceases to drain; (vi) weighing the material in the container (i.e., wet weight); (vii) again drying the material by heating it under temperatures of 105.degree. C. for a period of 24 hours or using another scientifically acceptable technique to reduce the moisture content of the material to less than 2% and preferably less than 1%; and (viii) weighing the dry material again (i.e., dry weight) and, for purposes of a volumetric measure, determining the volume of the material.
[0119] Measured gravimetrically, the water holding/water retention capacity is determined by measuring the difference in weight of the material from step (vi) to step (viii) over the weight of the material from step (viii) (i.e., wet weight-dry weight/dry weight). FIG. 8 illustrates the different water retention capacities of raw biochar versus treated biochar measured gravimetrically. As illustrated, water retention capacity of raw biochar can be between 100-200%, whereas treated biochar can have water retention capacities measured gravimetrically between 200-400%.
[0120] Water holding capacity can also be measured volumetrically and represented as a percent of the volume of water retained in the biochar after gravitationally draining the excess water/volume of biochar The volume of water retained in the biochar after draining the water can be determined from the difference between the water added to the container and water drained off the container or from the difference in the weight of the wet biochar from the weight of the dry biochar converted to a volumetric measurement. This percentage water holding capacity for treated biochar may be 50-55 percent and above by volume.
[0121] Given biochar's increased water retention capacity, the application of the treated biochar and even the raw biochar can greatly assist with the reduction of water and/or nutrient application. It has been discovered that these same benefits can be imparted to agricultural growth.
[0122] Treated biochar of the present invention has also demonstrated the ability to retain more water than raw biochar after exposure to the environment for defined periods of time. For purposes of this application "remaining water content" can be defined as the total amount of water that remains held by the biochar after exposure to the environment for certain amount of time. Exposure to environment is exposure at ambient temperature and pressures. Under this definition, remaining water content can be measured by (i) creating a sample of biochar that has reached its maximum water holding capacity; (ii) determining the total water content by thermogravimetric analysis (H.sub.2O (TGA)), as described above on a sample removed from the output of step (i) above, (iii) exposing the biochar in the remaining sample to the environment for a period of 2 weeks (15 days, 360 hrs.); (iv) determining the remaining water content by thermogravimetric analysis (H.sub.2O (TGA)); and (v) normalizing the remaining (retained) water in mL to 1 kg or 1 L biochar. The percentage of water remaining after exposure for this two-week period can be calculated by the remaining water content of the biochar after the predetermined period over the water content of the biochar at the commencement of the two-week period. Using this test, treated biochar has shown to retain water at rates over 4.times. that of raw biochar. Testing has further demonstrated that the following amount of water can remain in treated biochar after two weeks of exposure to the environment: 100-650 mL/kg; 45-150 mL/L; 12-30 gal/ton; 3-10 gal/yd.sup.3 after 360 hours (15 days) of exposure to the environment. In this manner, and as illustrated in FIG. 8, biochar treated with a process consistent with those described in this disclosure can increase the amount of retained water in biochar about 3 times compared to other methods even after seven weeks. In general, the more porous and the higher the surface area of a given material, the higher the water retention capacity. Further, it is theorized that by modifying the hydrophilicity/hydrophobicity of the pore surfaces, greater water holding capacity and controlled release may be obtained. Thus, viewed as a weight percent, e.g., the weight of retained water to weight of biochar, examples of the present biochars can retain more than 5% of their weight, more than 10% of their weight, and more than 15% of their weight, and even more than 50% of their weight compared to an average soil which may retain 2% or less, or between 100-600 ml/kg by weight of biochar.
[0123] Tests have also shown that treated biochars that show weight loss of >1% in the interval between 43-60.degree. C. when analyzed by the Thermal Gravimetric Analysis (TGA) (as described below) demonstrate greater water holding and content capacities over raw biochars. Weight loss of >5%-15% in the interval between 38-68.degree. C. when analyzed by the Thermal Gravimetric Analysis (TGA) using sequences of time and temperature disclosed in the following paragraphs or others may also be realized. Weight percentage ranges may vary from between >1%-15% in temperature ranges between 38-68.degree. C., or subsets thereof, to distinguish between treated biochar and raw biochar.
[0124] FIG. 9 is a chart 900 showing the weight loss of treated biochars 902 versus raw biochar samples 904 when heated at varying temperatures using the TGA testing described below. As illustrated, the treated biochars 902 continue to exhibit weight loss when heated between 40-60.degree. C. when analyzed by the Thermal Gravimetric Analysis (TGA) (described below), whereas the weight loss in raw biochar 804 between the same temperature ranges levels off. Thus, testing demonstrates the presence of additional moisture content in treated biochars 902 versus raw biochars 904.
[0125] In particular, the treated biochars 902 exhibit substantial water loss when heated in inert gas such as nitrogen following treatment. More particularly, when heated for 25 minutes at each of the following temperatures 20, 30, 40, 50 and 60.degree. C. the treated samples lose about 5-% to 15% in the interval 43-60.degree. C. and upward of 20-30% in the interval between 38-68.degree. C. The samples to determine the water content of the raw biochar were obtained by mixing a measured amount of biochar and water, stirring the biochar and water for 2 minutes, draining off the water, measuring moisture content and then subjecting the sample to TGA. The samples for the treated biochar were obtained by using the same measured amount of biochar as used in the raw biochar sample and using treatment process consistent with those described in this disclosure. The moisture content is then measured and the sample is subjected to TGA described above.
[0126] The sequences of time and temperature conditions for evaluating the effect of biochars heating in inert atmosphere is defined in this application as the "Bontchev-Cheyne Test" ("BCT"). The BCT is run using samples obtained, as described above, and applying Thermal Gravimetric Analysis (TGA) carried out using a Hitachi STA 7200 analyzer under nitrogen flow at the rate of 110 mL/min. The biochar samples are heated for 25 minutes at each of the following temperatures: 20, 30, 40, 50 and 60.degree. C. The sample weights are measured at the end of each dwell step, at the beginning and at the end of the experiment. The analyzer also continually measures and records weight over time. Biochars treated with a process consistent with those described in this disclosure to enhance water holding or retention capacities typically exhibit weight loss of >5% in the interval between 38-68.degree. C., >1% in the interval between 43-60.degree. C. Biochars with greater water holding or retention capacities can exhibit >5% weight loss in the interval between 43-60.degree. C. measured using the above described BCT.
[0127] Lastly, as illustrated in FIG. 10, plant available water is greatly increased in treated biochar over that of raw biochar. FIG. 10 illustrates the plant available water in raw biochar, versus treated biochar and treated dried biochar and illustrates that treated biochar can have a plant available water percent of greater than 35% by volume.
[0128] "Plant Available Water" is the amount of unbound water in a material available for plants to uptake. This is calculated by subtracting the volumetric water content at the permanent wilting point from the volumetric water content at field capacity, which is the point when no water is available for the plants. Field capacity is generally expressed as the bulk water content retained in at -33 J/kg (or -0.33 bar) of hydraulic head or suction pressure. Permanent wilting point is generally expressed as the bulk water content retained at -1500 J/kg (or -15 bar) of hydraulic head or suction pressure. Methods for measuring plant available water are well-known in the industry and use pressure plate extractors, which are commercially available or can be built using well-known principles of operation.
[0129] 2. Adjusts pH
[0130] With regard to treatment for pH adjustment, the above described vacuum infiltration processes and/or surfactant treatment processes have the ability to take raw biochars having detrimental or deleterious pHs and transform those biochars into a treated biochar having pH that is in an optimal range for most plant growth, and soil health. Turning to FIG. 11, a graph 1100 is provided that shows the pH of various starting raw biochars that were made from different starting materials and pyrolysis process temperatures, including coconut shells 1104, pistachio shells 1101, corn at 500.degree. C. 1105, corn at 900.degree. C. 1102, bamboo 1103, mesquite 1106, wood and coffee 1108, wood (Australia) 1109, various soft woods 1110, 1111, 1112, 1113, 1114, 1115, 1116, 1117, red fir at 900.degree. C. 1107, various grasses at 500.degree. C. 1118, 1119, 1120, grass 1121, and grass at 900.degree. C. 1123. The treatment processes described in this disclosure, can be used to alter the pH from the various undesirable pH levels and bring the pH into the preferred, optimal range 1124 for most plant growth, soil health and combinations of these. FIG. 12 is a chart 1200 showing percentage of germination for lettuce plants for particular pHs, and a desired germination range 1201. A control 1204 is compared with an optimal pH range 1202, and a distribution 1203 of growth rates across pHs is shown.
[0131] If treated for pH adjustment, the treated biochar takes a few days after treatment for the pH to normalize. Once normalized, tests have proven that pH altered biochar remains at a stable pH, typically the treatment is used to lower the stable pH to below that of the raw biochar, for up to 12 months or more after treatment. Although in certain situations, the pH could be altered to be higher than the raw biochar when needed.
[0132] For example, the treatment process of the present invention can remove and/or neutralize inorganic compounds, such as the calcium hydroxide ((CaOH).sub.2), potassium oxide (K.sub.2OK.sub.2OK.sub.20), magnesium oxide (MgO), magnesium hydroxide (Mg(OH).sub.2), and many others that are formed during pyrolysis, and are fixed to the biochar pore surfaces. These inorganics, in particular calcium hydroxide, adversely affect the biochar's pH, making the pH in some instances as high as 8.5, 9.5, 10.5 and 11.2. These high pH ranges are deleterious, detrimental to crops, and may kill or adversely affect the plants, sometimes rendering an entire field a loss.
[0133] The calcium hydroxide, and other inorganics, cannot readily and quickly be removed by simple washing of the biochar, even in an acid bath. It cannot be removed by drying the biochar, such as by heating or centrifugal force. It is theorized that these techniques and methodologies cannot reach or otherwise affect the various pore surfaces, e.g., macro-, meso- and micro- in any viable or efficacious manner; and thus cannot remove or otherwise neutralize the calcium hydroxide.
[0134] Upon modification of the pore surface area by removal and/or neutralization of deleterious substances, such as calcium hydroxide, the pH of the biochar can be reduced to the range of about pH 5 to about pH 8, and more preferably from about pH 6.4 to about 7.2, and still more preferably around 6.5 to 6.8, recognizing that other ranges and pHs are contemplated and may prove useful, under specific environmental or agricultural situations or for other applications. Thus, the present treated biochars, particles, batches and both, have most, essentially all, and more preferably all, of their pore surfaces modified by the removal, neutralization and both, of the calcium hydroxide that is present in the starting biochar material. These treated biochars have pHs in the range of about 5 to about 8, about 6.5 to about 7.5, about 6.4 to about 7, and about 6.8. Prior to and before testing, biochar is passed through a 2 mm sieve before pH is measured. All measurements are taken according to Rajkovich et. al, Corn growth and nitrogen nutrition after additions of biochars with varying properties to a temperate soil, Biol. Fertil. Soils (2011), from which the International Biochar Initiative (IBI) method is based.
[0135] There are a wide variety of tests, apparatus and equipment for making pH measurements. For example, and preferably when addressing the pH of biochar, batches, particles and pore surfaces of those particles, two appropriates for measuring pH are the Test Method for the US Composting Council ("TMCC") 4.11-A and the pH Test Method promulgated by the International Biochar Initiative. The test method for the TMCC comprises mixing biochar with distilled water in 1:5 [mass:volume] ratio, e.g., 50 grams of biochar is added to 250 ml of pH 7.0.+-.0.02 water and is stirred for 10 minutes; the pH is then the measured pH of the slurry. The pH Test Method promulgated by the International Biochar Initiative comprises 5 grams of biochar is added to 100 ml of water pH=7.0.+-.0.02 and the mixture is tumbled for 90 minutes; the pH of the slurry is measured at the end of the 90 minutes of tumbling.
[0136] 3. Removing/Neutralizing Deleterious Materials
[0137] Further, the treatment processes are capable of modifying the pore surfaces to remove or neutralize deleterious materials that are otherwise difficult, if not for all practical purpose, impossible to mitigate. For example, heavy metals, transition metals, sodium and phytotoxic organics, polycyclic aromatic hydrocarbons, volatile organic compounds (VOCs), and perhaps other phytotoxins. Thus, by treating the biochar in accordance with the treatment processes set forth and described above, the resulting treated biochar has essentially all, and more preferably all, of their pore surfaces modified by the removal, neutralization and both, of one or more deleterious, harmful, or potentially harmful material that is present in the starting biochar material.
[0138] For example, treatment can reduce the total percentage of residual organic compounds (ROC), including both the percentage of heavy ROCs and percentage of VOCs. Through treatment, the total ROC can be reduced to 0-25% wt. %, percentage heavy ROC content can be reduced to 0-20% wt. % and VOC content can be reduced to less than 5% wt. %. For purposes of this application, "Residual organic compounds" (ROCs) are defined as compounds that burn off during thermogravimetric analysis, as defined above, between 150 degrees C. and 950 degrees C. Residual organic compounds include, but are not limited to, phenols, polyaromatic hydrocarbons, monoaromatic hydrocarbons, acids, alcohols, esters, ethers, ketones, sugars, alkanes and alkenes. Of the ROCs, those that burn off using thermogravimetric analysis between 150 degrees C. and 550 degrees are considered light organic compounds (volatiles or VOCs), and those that burn off between 550 degrees C. and 950 degrees C. are heavy residual organic compounds. It should be noted that there may be some inorganic compounds which also are burned off during TGA analysis in these temperature ranges, but these are generally a very low percentage of the total emission and can be disregarded in the vast majority of cases as slight variations. In any of these measurements, a gas chromatograph/mass spectrometer may be used if needed for higher degrees of precision.
[0139] The percent water, total organic compounds, total light organic compounds (volatiles or VOC) and total heavy organic compounds, as referenced in this application as contained in a biochar particle or particles in a sample may all be measured by thermogravimetric analysis. Thermogravimetric analysis is performed by a Hitachi STA 7200 analyzer or similar piece of equipment under nitrogen flow at the rate of 110 mL/min. The biochar samples are heated for predetermined periods of time, e.g., 20 minutes, at a variety of temperatures between 100 and 950.degree. C. The sample weights are measured at the end of each dwell step and at the beginning and at the end of the experiment. Thermogravimetric analysis of a given sample indicating percentage water in a sample is determined by % mass loss measured between standard temperature and 150 degrees C. Thermogravimetric analysis of a given sample indicating percentage of residual organic compounds is measured by percentage mass loss sustained between 150 degrees C. and 950 degrees C. Thermogravimetric analysis of a given sample indicating percentage of light organic compounds (volatiles) is measured by percentage mass loss sustained between 150 degrees C. and 550 degrees C. Thermogravimetric analysis of a given sample indicating percentage of heavy organic compounds is measured by percentage mass loss sustained between 550 degrees C. and 950 degrees C. FIG. 13 is an example of a Thermogravimetric Analysis (TGA) plot outlining the above explanation and the measure of water, light organics and heavy organics.
[0140] As noted above, treatment can remove or neutralize heavy metals, transition metals, sodium and phytotoxic organics, polycyclic aromatic hydrocarbons, volatile organic compounds (VOCs), other phytotoxins, and even dioxins. Thus, by treating the biochar in accordance with the treatment processes set forth and described above, the resulting treated biochar has essentially all, and more preferably all, of their pore surfaces modified by the removal, neutralization or both, of one or more deleterious, harmful, or potentially harmful material that is present in the starting biochar material.
[0141] Dioxins may also be removed through the treatment processes of the present invention. Dioxins are released from combustion processes and thus are often found in raw biochar. Dioxins include polychlorinated dibenzo-p-dioxins (PCDDs) (i.e., 75 congeners (10 are specifically toxic)); polychlorinated dibenzofurans (PCDFs) (i.e., 135 congeners (7 are specifically toxic)) and polychlorinated biphenyls (PCBs) (Considered dioxin-like compounds (DLCs)).
[0142] Since some dioxins may be carcinogenic even at low levels of exposure over extended periods of time, the FDA views dioxins as a contaminant and has no tolerances or administrative levels in place for dioxins in animal feed. Dioxins in animal feed can cause health problems in the animals themselves. Additionally, the dioxins may accumulate in the fat of food-producing animals and thus consumption of animal derived foods (e.g. meat, eggs, milk) could be a major route of human exposure to dioxins. Thus, if biochar is used in animal applications, where the animals ingest the biochar, the ability to remove dioxins from the raw biochar prior to use is of particular significance.
[0143] Results have proven the removal of dioxins from raw biochar by applying the treatment process of the present invention. To demonstrate the removal of dioxins, samples of both raw biochar and biochar, treated within the parameters set forth above, were sent out for testing. The results revealed that the dioxins in the raw biochar were removed through treatment as the dioxins detected in the raw biochar sample were not detected in the treated biochar sample. Below is a chart comparing the test results of measured dioxins in the raw verses the treated biochar.
TABLE-US-00002 Amount Detected Amount Detected in Raw Biochar in Treated Biochar Dioxins Sample Sample Tetradioxins 26.4 ng/Kg-dry Not detectable Pentadioxins 5.86 ng/Kg-dry Not detectable Hexadioxins 8.41 ng/Kg-dry Not detectable
[0144] A number of different dioxins exist, several of which are known to be toxic or undesirable for human consumption. Despite the test results above, it is possible that any number of dioxins could be present in raw biochar depending on the biomass or where the biomass is grown. It is shown, however, in the above testing, that the treatment process of the present invention can be used to eliminate dioxins present in raw biochar.
[0145] Seventeen tetra-octo dioxins and furan congeners are the basis for regulatory compliance. Other dioxins are much less toxic. Dioxins are generally regulated on toxic equivalents (TEQ) and are represented by the sum of values weighted by Toxic Equivalency
TEQ=.SIGMA.[C.sub.i].times.TEF.sub.i
Factor (TEF)
[0146] 2,3,7,8-TCDD has a TEF of 1 (most toxic). TEQ is measured as ng/kg WHO-PCDD/F-TEQ//kg NDs are also evaluated. Two testing methods are generally used to determine TEQ values: EPA Method 8290 (for research and understanding at low levels (ppt-ppq); and EPA Method 1613B (for regulatory compliance). Both are based on high resolution gas chromatography (HRGC)/high resolution mass spectrometry (HRMS).
[0147] The required EU Feed Value is equal to or less than 0.75 ng/kg WHO-PCDD/F-TEQ//kg. Treated biochar, in accordance with the present invention, has shown to have TEQ dioxins less than 0.5 ng/kg WHO-PCDD/F-TEQ//kg, well below the requirement for EU Feed limits of 0.75 ng/kg WHO-PCDD/F-TEQ//kg. As further set forth above, treatment can reduce the amount of detectable dioxins from raw biochar such that the dioxins are not detectible in treated biochar. Two methods are used: EPA Method 8290 (for research and understanding at low levels (ppt-ppq); and EPA Method 1613B (for regulatory compliance). Both are based on high resolution gas chromatography (HRGC)/high resolution mass spectrometry (HRMS).
[0148] 4. Pore Volume
[0149] Generally, a treated biochar sample has greater than 50% by volume of its porosity in macropores (pores greater than 300 nanometers). Further, results indicate that greater than 75% of pores in treated biochar are below 50,000 nanometers. Also, results indicate that greater than 50% by volume of treated biochar porosity are pores in the range of 500 nanometers and 100,000 nanometers. Bacterial sizes are typically 500 nanometers to several thousand nanometers. Bacteria and other microbes have been observed to fit and colonize in the pores of treated biochar, thus supporting the pore size test results.
[0150] Macropore volume is determined by mercury porosimetry, which measures the meso and/or macro porosity by applying pressure to a sample immersed in mercury at a pressure calibrated for the minimum pore diameter to be measured (for macroporosity this is 300 nanometers). This method can be used to measure pores in the range of 3 nm to 360,000 nm. Total volume of pores per volumetric unit of substance is measured using gas expansion method.
[0151] Depending upon the biomass from which the biochar is derived, mercury porosimetry testing has shown that washing under differential pressure, using the processes described above, can increase the number of both the smallest and larger pores in certain biochar (e.g., pine) and can increase the number of usable smaller pores. Treatment of biochar using either vacuum or surfactant does alter the percentage of total usable pores between 500 to 100,000 nanometers and further has varying impact on pores less than 50,000 nanometers and less than 10,000 nanometers.
[0152] FIG. 14 is a chart 1400 showing the impact of treatment on pores sizes of biochar derived from coconut. The majority of the coconut based biochar pores are less than 10 microns. Many are less than 1 micron. Vacuum processing of the biochar results in small reduction of 10 to 50 micron pores, with increase of smaller pores on vacuum processing. The mercury porosimetry results of the raw biochar are represented by 1402 (first column in the group of three). The vacuum treated biochar is represented by 1404 (second column in the group of three) and the surfactant treated biochar is 1406 (third column in the group of three).
[0153] FIG. 15 is a chart 1500 showing the impact of treatment on pores sizes of biochar derived from pine. The majority of the pine based biochar pores are 1 to 50 microns, which is a good range for micro-biologicals. Vacuum processing results in significant reduction of the 10 to 50 micron pores, with an increase of smallest and largest pores. The mercury porosimetry results of the raw biochar are represented by 1502 (first column in the group of three). The vacuum treated biochar is represented by 1504 (second column in the group of three) and the surfactant treated biochar is 3006 (third column in the group of three).
[0154] 5. Electrical Conductivity
[0155] The electrical conductivity (EC) of a solid material-water mixture indicates the amount of salts present in the solid material. Salts are essential for plant growth. The EC measurement detects the amount of cations or anions in solution; the greater the amount of ions, the greater the EC. The ions generally associated with salinity are Ca.sup.2+, Mg.sup.2+, K.sup.+, Na.sup.+, H.sup.+, NO.sub.3.sup.-, SO.sub.4.sup.2-, Cl--, HCO.sub.3.sup.-, OH.sup.-. Electrical conductivity testing of biochar was done following the method outlined in the USDA's Soil Quality Test Kit Guide and using a conventional EC meter. The biochar sample is mixed with DI water in a 1:1 biochar to water ratio on a volume basis. After thorough mixing, the EC (dS/m) is measured while the biochar particles are still suspended in solution. Treatment, as outlined in this disclosure can be used to adjust the ions in the char. Testing of treated biochar shows its EC is generally greater than 0.2 dS/m and sometimes greater than 0.5 dS/m.
[0156] 6. Cation Exchange Capacity
[0157] One method for cation exchange capacity ("CEC") determination is the use of ammonium acetate buffered at pH 7.0 (see Schollenberger, C. J. and Dreibelbis, E R. 1930, Analytical methods in base-exchange investigations on soils, Soil Science, 30, 161-173). The material is saturated with 1M ammonium acetate, (NH.sub.4OAc), followed by the release of the NH.sub.4.sup.+ ions and its measurement in meq/100 g (milliequivalents of charge per 100 g of dry soil) or cmolc/kg (centimoles of charge per kilogram of dry soil). Instead of ammonium acetate another method uses barium chloride according to Mehlich, 1938, Use of triethanolamine acetate-barium hydroxide buffer for the determination of some base exchange properties and lime requirement of soil, Soil Sci. Soc. Am. Proc. 29:374-378. 0.1 M BaCl.sub.2 is used to saturate the exchange sites followed by replacement with either MgSO.sub.4 or MgCl.sub.2.
[0158] Indirect methods for CEC calculation involves the estimation of extracted Ca.sub.2.sup.+, Mg.sub.2.sup.+, K.sup.+, and Na.sup.+ in a standard soil test using Mehlich 3 and accounting for the exchangeable acidity (sum of H.sup.+, Al.sub.3.sup.+, Mn.sub.2.sup.+, and Fe.sub.2.sup.+) if the pH is below 6.0 (see Mehlich, A. 1984, Mehlich-3 soil test extractant: a modification of Mehlich-2 extractant, Commun. Soil Sci. Plant Anal. 15(12): 1409-1416). When treated using the above methods, including but not limited by washing under a vacuum, treated biochars generally have a CEC greater than 5 millieq/l and some even have a CEC greater than 25 (millieq/l). To some extent, treatment can be used to adjust the CEC of a char.
[0159] 7. Anion Exchange Capacity
[0160] Similar to CEC measurements, anion exchange capacity ("AEC") may be calculated directly or indirectly-saturated paste extraction of exchangeable anions, Cl.sup.-, NO.sub.3.sup.-, SO.sub.4.sup.2-, and PO.sub.4.sup.3- to calculate anion sum or the use of potassium bromide to saturate anions sites at different pHs and repeated washings with calcium chloride and final measurement of bromide (see Rhoades, J. D. 1982, Soluble salts, p. 167-179. In: A. L. Page et al. (ed.) Methods of soil analysis: Part 2: Chemical and microbiological properties; and Michael Lawrinenkoa and David A. Laird, 2015, Anion exchange capacity of biochar, Green Chem., 2015, 17, 4628-4636). When treated using the above methods, including but not limited by washing under a vacuum, treated biochars generally have an AEC greater than 5 millieq/l and some even have an AEC greater than 20 (millieq/l). To some extent, treatment can be used to adjust the CEC of a char.
[0161] 8. Hydrophilicity/Hydrophobicity
[0162] The ability to control the hydrophilicity of the pores provides the ability to load the biochar particles with larger volumes of inoculant. The more hydrophilic the more the biochars can accept inoculant or infiltrate. Tests show that biochar treated in accordance with the above processes, using either vacuum or surfactant treatment processes increase the hydrophilicity of raw biochar. Two tests may be used to test the hydrophobicity/hydrophilicity of biochar: (i) the Molarity of Ethanol Drop ("MED") Test; and (ii) the Infiltrometer Test.
[0163] The MED test was originally developed by Doerr in 1998 and later modified by other researchers for various materials. The MED test is a timed penetration test that is noted to work well with biochar soil mixtures. For 100% biochar, penetration time of different mixtures of ethanol/water are noted to work better. Ethanol/Water mixtures verses surface tension dynes were correlated to determine whether treated biochar has increased hydrophilicity over raw biochar. Seven mixtures of ethanol and deionized water were used with a sorption time of 3 seconds on the biochar.
[0164] Seven solutions of deionized ("DI") water with the following respective percentages of ethanol: 3, 5, 11, 13, 18, 24 and 36, were made for testing. The test starts with a mixture having no DI. If the solution is soaked into the biochar in 3 seconds for the respective solution, it receives the corresponding Hydrophobicity Index value below.
TABLE-US-00003 Ethanol % Hydrophobicity Index 0: DI Water 0 Very Hydrophillic 3% 1 5% 2 11% 3 13% 4 18% 5 24% 6 36% 7 Strongly Hydrophillic
[0165] To start the test the biochar ("material/substrate") is placed in convenient open container prepared for testing. Typically, materials to be tested are dried 110.degree. C. overnight and cooled to room temperature. The test starts with a deionized water solution having no ethanol. Multiple drips of the solution are then laid onto the substrate surface from low height. If drops soak in less than 3 seconds, test records substrate as "0". If drops take longer than 3 seconds or don't soak in, go to test solution 1. Then, using test solution 1, multiple drops from dropper are laid onto the surface from low height. If drops soak into the substrate in less than 3 seconds, test records material as "1". If drops take longer than 3 seconds, or don't soak in, go to test solution 2. Then, using test solution 2, multiple drops from dropper laid onto the surface from low height. If drops soak into the substrate in less than 3 seconds, test records material as "2". If drops take longer than 3 seconds, or don't soak in, go to test solution 3. Then, using test solution 3, multiple drops from dropper laid onto the surface from low height. If drops soak into the substrate in less than 3 seconds, test records material as "3". If drops take longer than 3 seconds, or don't soak in, go to solution 4.
[0166] The process above is repeated, testing progressively higher numbered MED solutions until the tester finds the solution that soaks into the substrate in 3 seconds or less. The substrate is recorded as having that hydrophobicity index number that correlates to the solution number assigned to it (as set forth in the chart above).
[0167] Example test results using the MED test method is illustrated below.
TABLE-US-00004 MATERIAL HYDROPHOBICITY INDEX Raw Biochars 3 to 5 Treated Biochars 1 to 3
[0168] Another way to measure and confirm that treatment decreases hydrophobicity and increases hydrophilicity is by using a mini disk infiltrometer. For this test procedure, the bubble chamber of the infiltrometer is filled three quarters full with tap water for both water and ethanol sorptivity tests. Deionized or distilled water is not used. Once the upper chamber is full, the infiltrometer is inverted and the water reservoir on the reserve is filled with 80 mL. The infiltrometer is carefully set on the position of the end of the mariotte tube with respect to the porous disk to ensure a zero suction offset while the tube bubbles. If this dimension is changed accidentally, the end of the mariotte tube should be reset to 6 mm from the end of the plastic water reservoir tube. The bottom elastomer is then replaced, making sure the porous disk is firmly in place. If the infiltrometer is held vertically using a stand and clamp, no water should leak out.
[0169] The suction rate of 1 cm is set for all samples. If the surface of the sample is not smooth, a thin layer of fine biochar can be applied to the area directly underneath the infiltrometer stainless steel disk. This ensures good contact between the samples and the infiltrometer. Readings are then taken at 1 min intervals for both water and ethanol sorptivity test. To be accurate, 20 mL water or 95% ethanol needs to be infiltrated into the samples. Record time and water/ethanol volumes at the times are recorded.
[0170] The data is then processed to determine the results. The data is processed by the input of the volume levels and time to the corresponding volume column. The following equation is used to calculate the hydrophobicity index of R
I=at+b {square root over (t)} [0171] a: Infiltration Rate, cm/s [0172] b: Sorptivity, cm/s.sup.1/2
[0172] R = 1.95 * b ethanol b water ##EQU00001##
[0173] FIG. 16 illustrates one example of the results of a hydrophobicity test performed on raw biochar, vacuum treated biochar and surfactant treated biochar. As illustrated, both the vacuum treated and surfactant treated biochar are more hydrophilic than the raw biochar based upon the lower Index rating. In accordance with the test data in FIG. 16, the hydrophobicity of raw biochar was reduced 23% by vacuum processing and 46% by surfactant addition.
[0174] As an example, raw biochar and treated biochar were tested with ethanol and water, five times for each. The results below show that the hydrophobicity index of the treated biochar is lower than the raw biochar. Thus, tests demonstrate that treating the biochar, using the methods set forth above, make the biochar less hydrophobic and more hydrophilic.
TABLE-US-00005 MATERIAL HYDROPHOBICITY INDEX Dried Raw Biochar 12.9 Dried Vacuum Treated Biochar 10.4 Dried Surfactant Treated Biochar 7.0 As Is Raw Biochar 5.8 As Is Vacuum Treated Biochar 2.9
[0175] Further, through the treatment processes of the present invention, the biochar can also be infused with soil enhancing agents. By infusing liquid into the pore structure through the application of positive or negative pressure and/or a surfactant, alone or in combination, provides the ability to impregnate the macropores of the biochar with soil enhancing solutions and solids. The soil enhancing agent may include, but not be limited to, any of the following: water, water solutions of salts, inorganic and organic liquids of different polarities, liquid organic compounds or combinations of organic compounds and solvents, mineral and organic oils, slurries and suspensions, supercritical liquids, fertilizers, plant growth promoting rhizobacteria, free-living and nodule-forming nitrogen fixing bacteria, organic decomposers, nitrifying bacteria, phosphate solubilizing bacteria, biocontrol agents, bioremediation agents, saprotrophic fungi, ectomycorrhizae and endomycorrhizae, among others.
[0176] 9. Impregnation and/or Inoculation with Infiltrates or Additives
[0177] In addition to mitigating or removing deleterious pore surface properties, by treating the pores of the biochar through a forced, assisted, accelerate or rapid infiltration process, such as those described above, the pore surface properties of the biochar can be enhanced. Such treatment processes may also permit subsequent processing, may modify the pore surface to provide predetermined properties to the biochar, and/or provide combinations and variations of these effects. For example, it may be desirable or otherwise advantageous to coat substantially all, or all of the biochar macropore and mesopore surfaces with a surface modifying agent or treatment to provide a predetermined feature to the biochar, e.g., surface charge and charge density, surface species and distribution, targeted nutrient addition, magnetic modifications, root growth facilitator, and water absorptivity and water retention properties.
[0178] By infusing liquids into the pores of biochar, it has been discovered that additives infused within the pores of the biochar provide a time release effect or steady flow of some beneficial substances to the environment, e.g. root zones of the plants, and also can improve and provide a more beneficial environment for microbes which may reside or take up residence within the pores of the biochar. In particular, additive infused biochars placed in the soil prior to or after planting can dramatically reduce the need for high frequency application of additives, minimize losses caused by leaching and runoff and/or reduce or eliminate the need for controlled release fertilizers. They can also be exceptionally beneficial in animal feed applications by providing an effective delivery mechanism for beneficial nutrients, pharmaceuticals, enzymes, microbes, or other substances.
[0179] For purposes of this application, "infusion" of a liquid or liquid solution into the pores of the biochar means the introduction of the liquid or liquid solution into the pores of the biochar by a means other than solely contacting the liquid or solution with the biochar, e.g., submersion. The infusion process, as described in this application in connection with the present invention, includes a mechanical, chemical or physical process that facilitates or assist with the penetration of liquid or solution into the pores of the biochar, which process may include, but not be limited to, positive and negative pressure changes, such as vacuum infusion, surfactant infusion, or infusion by movement of the liquid and/or biochar (e.g., centrifugal force and/or ultrasonic waves) or other method that facilitates, assists, forces or accelerates the liquid or solution into the pores of the biochar.
[0180] Prior to infusing the biochar, the biochar, as described in detail above, may be washed and/or moisture adjusted. FIG. 17 is a flow diagram 1700 of one example of a method for infusing biochar with an additive. Optionally, the biochar may first be washed or treated at step 1702, the wash may adjust the pH of the biochar, as described in more detail above, or may be used to remove elemental ash and other harmful organics that may be unsuitable for the desired infused additive. Optionally, the moisture content of the biochar may then be adjusted by drying the biochar at step 1704, also as described in further detail above, prior to infusion of the additive or inoculant at step 1706.
[0181] In summary, the infusion process may be performed with or without any washing, prior pH adjustment or moisture content adjustment. Optionally, the infusion process may be performed with the wash and/or the moisture adjustment step. All the processes may be completed alone or in the conjunction with one or more of the others.
[0182] Through the above process of infusing the additive into the pores of the biochar, the pores of the biochar may be filled by 25%, up to 100%, with an additive solution, as compared to 1-20% when the biochar is only submerged in the solution or washed with the solution for a period of less than twelve hours. Higher percentages may be achieved by washing and/or drying the pores of the biochar prior to infusion.
[0183] Data have been gathered from research conducted comparing the results of soaking or immersion of biochar in liquid versus vacuum impregnation of liquid into biochar. These data support the conclusion that vacuum impregnation provides greater benefits than simple soaking and results in a higher percentage volume of moisture on the surface, interstitially and in the pores of the biochar.
[0184] In one experiment, equal quantities of pine biochar were mixed with equal quantities of water, the first in a beaker, the second in a vacuum flask. The mixture in the beaker was continuously stirred for up to 24 hours, then samples of the suspended solid were taken, drained and analyzed for moisture content. The mixture in the vacuum flask was connected to a vacuum pump and negative pressure of 15'' was applied. Samples of the treated solid were taken, drained and analyzed for moisture content. FIG. 18 is a chart illustrating the results of the experiment. The lower graph 1802 of the chart, which shows the results of soaking over time, shows a Wt. % of water of approximately 52%. The upper graph 1804 of the chart, which shows the results of vacuum impregnation over time, shows a Wt. % of water of approximately 72%.
[0185] FIGS. 19a and 19b show two charts that further illustrate that the total water and/or any other liquid content in processed biochar can be significantly increased using vacuum impregnation instead of soaking. FIG. 19a compares the mL of total water or other liquid by retained by 1 mL of treated biochar. The graph 1902 shows that approximately 0.17 mL of water or other liquid are retained through soaking, while the graph 1904 shows that approximately 0.42 mL of water or other liquid are retained as a result of vacuum impregnation. FIG. 19b shows that the retained water of a biochar subjected to soaking consists entirely of surface and interstitial water 1906, while the retained water of a biochar subjected to vacuum impregnation consists not only of surface and interstitial water 1908a, but also water impregnated in the pores of the biochar 1908b.
[0186] In addition, as illustrated by FIG. 20, the amount of moisture content impregnated into the pores of vacuum processed biochars by varying the applied (negative) pressure during the treatment process. The graphs of four different biochars all show how the liquid content of the pours of each of them increase to 100% as the vacuum is increased.
[0187] The pores may be substantially filled or completely filled with additives to provide enhanced performance features to the biochar, such as increased plant growth, nutrient delivery, water retention, nutrient retention, disadvantageous species control, e.g., weeds, disease causing bacteria, insects, volunteer crops, etc. By infusing liquid deep into the pore structure through the application of positive or negative pressure, surfactant and/or ultrasonic waves, alone or in combination, provides the ability to impregnate the mesopores and macropores of the biochar with additives, that include, but are not limited to, soil enhancing solutions and solids. It should be noted that using these infusion techniques allows for impregnating the pores with additives that are more fragile. For example, since heating is not a requirement for these infusion techniques, microbes, chemicals, or compounds can be infused without risk of destroying the microbes or changing chemicals or compounds due to high temperatures. Also the process can be done at low temperatures to infuse chemicals that have low boiling points to keep them a liquid.
[0188] The additive may be a soil enhancing agent that includes, but is not be limited to, any of the following: water, water solutions of salts, inorganic and organic liquids of different polarities, liquid organic compounds or combinations of organic compounds and solvents, mineral and organic oils, slurries and suspensions, supercritical liquids, fertilizers, PGPB (including plant growth promoting rhizobacteria, free-living and nodule-forming nitrogen fixing bacteria, organic decomposers, nitrifying bacteria, and phosphate solubilizing bacteria), enzymes, biocontrol agents, bioremediation agents, saprotrophic fungi, ectomycorrhizae and endomycorrhizae, among others.
[0189] Fertilizers that may be infused into the biochar include, but are not limited to, the following sources of nitrogen, phosphorous, and potassium: urea, ammonium nitrate, calcium nitrate, sulfur, ammonium sulfate, monoammonium phosphate, ammonium polyphosphate, potassium sulfate, or potassium chloride.
[0190] Similar beneficial results are expected from other additives, such as: bio pesticides; herbicides; insecticides; nematicides; plant hormones; plant pheromones; organic or inorganic fungicides; algicides; antifouling agents; antimicrobials; attractants; biocides, disinfectants and sanitizers; miticides; microbial pesticides; molluscicides; bacteriacides; fumigants; ovicides; repellents; rodenticides, defoliants, desiccants; insect growth regulators; plant growth regulators; beneficial microbes; and, microbial nutrients or secondary signal activators, that may also be added to the biochar in a similar manner as a fertilizer. Additionally, beneficial macro- and micro-nutrients such as, calcium, magnesium, sulfur, boron, zinc, iron, manganese, molybdenum, copper and chloride may also be infused into the biochar in the form of a water solution or other solvent solution.
[0191] Examples of compounds, in addition to fertilizer, that may be infused into the pores of the biochar include, but are not limited to: phytohormones, such as, abscisic acid (ABA), auxins, cytokinins, gibberellins, brassinosteroies, salicylic acid, jasmonates, planet peptide hormones, polyamines, karrikins, strigolactones; 2,1,3-Benzothiadiazole (BTH), an inducer of systemic acquired resistance that confers broad spectrum disease resistance (including soil borne pathogens); signaling agents similar to BTH in mechanism or structure that protects against a broad range or specific plant pathogens; EPSPS inhibitors; synthetic auxins; photosystem I inhibitors photosystem II inhibitors; and HPPD inhibitors.
[0192] In one example, a 1000 ppm NO.sub.3.sup.- N fertilizer solution is infused into the pores of the biochar. As discussed above, the method to infuse biochar with the fertilizer solution may be accomplished generally by placing the biochar in a vacuum infiltration tank or other sealable mixing vessel, chamber or tank. When using vacuum infiltration, a vacuum may be applied to the biochar and then the solution may be introduced into the tank. Alternatively, the solution and biochar may both be introduced into the tank and, once introduced, a vacuum is applied. Based upon the determined total pore volume of the biochar or the incipient wetness, the amount of solution to introduce into the tank necessary to fill the pore of the biochar can be determined. When infused in this manner, significantly more nutrients can be held in a given quantity of biochar versus direct contact of the biochar with the nutrients alone.
[0193] When using a surfactant, the biochar and additive solution may be added to a tank along with 0.01-20% of surfactant, but more preferably 1-5% of surfactant by volume of fertilizer solution. The surfactant or detergent aids in the penetration of the wash solution into the pores of the biochar. The same or similar equipment used in the vacuum infiltration process can be used in the surfactant treatment process. Although it is not necessary to apply a vacuum in the surfactant treatment process, the vacuum infiltration tank or any other mixing vessel, chamber or tank can be used. Again, while it is not necessary to apply a vacuum, a vacuum may be applied or the pressure in the vessel may be changed. Further, the surfactant can be added with or without heat or cooling either of the infiltrate, the biochar, the vessel itself, or any combination of the three.
[0194] The utility of infusing the biochar with an additive is that the pores in biochar create a protective "medium" for carrying said additive to the environment. As an example when the additive is a fertilizer the nutrient infused biochar provides a more constant supply of available nutrients to the soil and plants and continues to act beneficially, potentially sorbing more nutrients or nutrients in solution even after introduction to the soil. By infusing the nutrients in the pores of the biochar, immediate oversaturation of the soil with the nutrients is prevented and a time released effect is provided. This effect is illustrated in connection with FIGS. 18 and 19 below. As demonstrated in connection with FIGS. 18 & 19 below, biochars having pores infused with additives, using the infusion methods described above, have been shown to increase nutrient retention, increase crop yields and provide a steadier flow of fertilizer to the root zones of the plants.
[0195] FIG. 21 is a chart showing improved mass yield in lettuce with fertilizer infused biochar using vacuum impregnation. FIG. 21 compares the mass yield results of lettuce grown in different environments. One set of data measurements represents lettuce grown in soil over a certain set time period with certain, predetermined amounts of fertilizer infused into the biochar. A second set of data represents lettuce grown in soil over a certain set period of time with the same amount of unimpregnated biochar added at the beginning of the trial and certain predetermined amounts of NPK solution added to the soil over time. Growth comparisons were made between the same amount of fertilizer solution infused into the biochar as added directly to the soil, using the same watering schedule. As illustrated, the test results demonstrated a 15% yield increase in growth when infusing approximately 750 mg/pot of NPK into the biochar than when applying it directly to the soil. Similarly, the same mass yield of lettuce is achieved at 400 mg NPK/pot with infused biochar than at 750 mg/pot when adding the fertilizer solution directly to the soil.
[0196] FIG. 22 is a chart illustrating the concentration of nitrate (N) found in distilled water after washing differentially treated biochar. In the illustrated example, two biochar samples (500 ml each) mixed with 1000 ppm NO.sub.3.sup.- N fertilizer solution were washed with distilled water. The resulting wash was then tested for the presence of nitrate (N), measured in ppm. In one sample, the biochar was submerged in and mixed with the nutrient solution. In the other example, the biochar was mixed or washed with a nutrient solution augmented with 1% surfactant by volume (i.e., 1 ml of surfactant per 100 ml of fertilizer solution) in a tumbler. In both examples, the biochar was not dried completely before infusion with the NO.sub.3.sup.- N fertilizer solution, but used as received with a moisture content of approximately 10-15%. In both examples, the biochar was mixed with solution and/or surfactant (in the case of a second sample) with a bench scale tumbler, rotating the drum for four (4) minutes without vacuum. The results demonstrate that the biochar treated with the 1% surfactant increases the efficiency of infiltrating nitrate fertilizer into biochar and then demonstrates the release of the nutrient over time. To yield the above data, the test was repeated six times for each treatment sample, with 10 washes for each sample per repeat test.
[0197] The above are only a few examples of how additive infused biochar may be produced for different uses. Those skilled in the art will recognize that there may be other mechanisms for infusing fertilizer or other soil additives into the pores of the biochar without departing from the scope of the invention. Those skilled in the art will further recognize that the present invention can be used on any type of soil application, including, but not limited to, the following: crops, turf grasses, potted plants, flowering plants, annuals, perennials, evergreens and seedlings, as will be further described below.
[0198] For example, in another implementation, additive infused biochar may be produced for use for consumption by animals and/or humans. Biochar may be infused in the same manner as described above with nutrients (such as carbohydrates, minerals, proteins, lipids), vitamins, drugs and/or other supplements (such as enzymes or hormones, to name a few), or a combination of any of the foregoing, for consumption by either humans and/or animals. Coloring, flavor agents and/or coating may also be infused into the pores of the biochar or applied to the surface. The foregoing may be included to enhance the performance of the substance in the digestive tract or to ease or facilitate the ingestion of the biochar.
[0199] D. Biochar as a Habitat for Microorganisms
[0200] Biotechnology, specifically the use of biological organisms, usually microorganisms, to address chemical, industrial, medical, or agricultural problems is a growing field with new applications being discovered daily. To date, much research has focused on identifying, developing, producing and deploying microbes for various uses. However, despite much work on the microbes themselves, relatively little work has been performed on how to carry, deliver, and encourage the successful establishment of these microbes in their targeted environment. Most current technology for microbial carriers in agriculture is based on technologies or products that are highly variable and, in many cases, lead to highly unpredictable performance of microbes in the field. For example, many commercial microbes in agricultural settings are delivered on peat, clay, or other carriers derived from natural sources, accompanied by limited engineering or process control.
[0201] Biochar have a proclivity to interact positively with many microbes relevant to plant health, animal health, and human public health applications. In fact, there has been a level of initial research focused on inoculating biochar with microbes and/or using biochar in conjunction with microbes or materials with microbes, e.g. compost. See co-owned U.S. Pat. No. 8,317,891 Method for Enhancing Soil Growth Using Bio-char and Fischer et al., and Synergisms between compost and biochar for sustainable soil amelioration 2012 http://www.intechopen.com/source/pdfs/27163/InTech-Synergisms_between_com- post_and_biochar_for_sustainable_soil_amelioration.pdf.
[0202] However, biochars, especially in raw form, often suffer from many characteristics which make their interaction with microbial organisms extremely unpredictable. Key among these undesirable characteristics is a high degree of variability. Because of this and other factors, biochar has been, to date, unused in large scale commercial biotechnology applications. There are several methods by which this variability can be ameliorated. At a high level, the methods to overcome these challenges fall into two categories: (i) making the biochar a more favorable habitat for the microbes--either by modifying its properties, adding materials beneficial to microbes, or removing materials deleterious to microbes, or (ii) inoculating, applying, or immobilizing the microbes on the biochar in ways that mitigate the underlying variability in the material. Both of these high-level methods can be used independently or in conjunction and have been shown to have a significant impact on the suitability of biochar in many biotechnology applications.
[0203] Before delving into the varying treatment methods that will turn the biochar into a microbial carrier or co-deploying with microbes, it is important to be able to view biochar as a habitat for microbes. Biochar, especially treated biochar, has many physical properties that make it interesting as a microbial habitat. The most obvious of these is its porosity (most biochars have a surface area of over 100 m2/g and total porosity of 0.10 cm3/cm3 or above). Furthermore, many biochars have significant water holding and nutrient retention characteristics which may be beneficial to microbes. Previous disclosure has outlined how these characteristics can be further improved with treatment, e.g., U.S. patent application Ser. No. 15/156,256, filed on May 16, 2016, and titled Enhanced Biochar.
[0204] However, recent data indicates that the Earth may be home to more than one trillion independent species of microbes (See Kenneth J. Locey and Jay T. Lennon, Scaling laws predict global microbial diversity, Proceedings of the National Academy of Science, vol. 113 no. 21 (see full text at http://www.pnas.org/content/113/21/5970.full). Clearly, each of these microbial species does not require an identical habitat. In fact, many have evolved in different conditions and thrive in different environments. Biochar, due to its organic origins, porosity, and amenability to treatment seems to be an extremely desirable base product to be used in the construction of microbial carriers or co-deployment of microbes. If the properties of the biochar can be made to match the properties expected by particular microbes, or groups of microbes, empirical data has shown that a much greater impact can be delivered in Many applications--whether the targeted biochar is used as a carrier, substrate, co-deployed product, or merely is introduced into the same environment at a separate time. It stands to reason, as many real-world environments are composed of very complex microbial ecosystems, that giving certain microbes in these ecosystems a more favorable habitat, can ultimately help those microbes to become more successfully established, and potentially shift the entire ecosystem based on their improved ability to compete for resources. Clearly this is a very desirable characteristic when the successful deployment and establishment of a targeted microbe into a new environment is a desired outcome.
[0205] There are many properties of a habitat which may be important to certain microbes, but some of the most important are: pH, hydrophobicity or hydrophilicity, ability to hold moisture, ability to retain and exchange certain types of nutrients, ion exchange capacity (cationic and anionic), physical protection from predatory or competitive microbes or protozoa (usable and inhabitable porosity), presence or absence of nutrients, micronutrients, or sources of metabolic carbon, ability to host other symbiotic microbes or plant systems (such as plant root tissue), or others which may be important to various types or species of microbes. Ability to either enhance or suppress the availability of certain enzymes can also be an extremely important factor in building a viable habitat. This invention focuses on methods and systems that can be used to consistently produce biochar which has these targeted characteristics, methods that can be used to effectively create a particular formulation of biochar targeted to match a particular microbe or group of microbes, and techniques for deploying the desired microbes along with this targeted biochar, through inoculation, co-deployment, integrated growth/fermentation, or other methods.
[0206] By using treatment properties disclosed previously, proper feedstock selection, and control of the pyrolysis process, the following are some, but not all, of the properties that can be consistently targeted and controlled at production scales to improve the biochar for use with microbes or as a microbial carrier. Examples of those properties include (1) pH, (2) hydrophobicity, (3) sodium levels, (4) usable pore size distribution and usable pore volume, (5) particle size and distribution, (6) exterior and interior surface geometry, (7) nutrient exchange, (8) useable carbon or energy source, (9) toxic materials or compounds, (10) surface structure/crystals/tortuosity, (11) compatibility with biofilm formations, (12) surface charge, (13) enzyme activity and (14) sterilization.
[0207] 1. pH
[0208] It is well known that various microbes prefer varying levels of acidity or alkalinity. For example, acidophiles have evolved to inhabit extremely acidic environments. Likewise, aikaliphiles prefer more basic (alkali) environments. It has been clearly shown that the methods outlined for treating biochars can product targeted pH values that can be sustained over long periods of time.
[0209] 2. Hydrophobicity
[0210] There are several common sources of hydrophobicity in porous carbonaceous materials. One of them is the occurrence of hydrophobic organic compounds on the surface of the char--typically residual from the pyrolysis process. Targeted removal of these compounds is a method to improve the hydrophobicity of porous carbonaceous substances. These compounds can be removed in a non-selective way by increasing the pyrolysis temperature of the biomass to a level at which the compounds will disassociate with the material and become gaseous. This method, while useful, is very broad, and can also remove other desirable compounds as well as changing the surface chemistry of the residual carbon, increasing ash percentages, or reducing carbon yield by reacting and removing more carbon than is necessary. These compounds can also be selectively removed by the application of a targeted solvent using the mechanisms previously disclosed to infiltrate liquids into the pore volume of the material. This method is also effective, and has shown to be much more predictable in the removal of certain compounds. Since the vast majority of microbes rely heavily on water for both transport and life, the easy association of water with a material has a large bearing on its ability to sustain microbial life.
[0211] 3. Sodium Levels
[0212] Differing types of microbes have varying proclivities for the presence of sodium. Some microbes Halobacterium spp., Salinibacter ruber, Wallemia ichthyophaga prefer high levels of salinity, while others prefer moderate or limited levels of sodium. Sodium can be removed from biochar by either simple washing, or more preferably and effectively, treatment methods which infuse a solvent (most commonly water, although others may be used) into the pores of the material. Sodium can be added, by using the same methods except instead of using a solvent, the liquid being washed with or infused is a solution high in sodium content. Additionally, since sodium usually manifests itself as a cation in solution, temporary or permanent adjustment of the cationic exchange capacity (CEC) of the material through treatment which impacts the ability of the material to exchange cations. Lowering the CEC of the material will in many cases reduce its ability to exchange sodium cations, while raising the CEC will typically enhance the ability of the material to exchange sodium cations, with exceptions occurring if other cations are present in quantities that cause them to preferentially exchange instead of the sodium cations present. Finally, differing biomass feedstock contains differing levels of sodium--selecting an appropriate feedstock prior to pyrolysis will result in a raw or untreated biochar with reasonably controlled levels of sodium. For example, pine wood, when pyrolyzed, results in a raw char with lower levels of sodium, while coconut shells result in char with higher levels of sodium after pyrolysis.
TABLE-US-00006 Untreated Untreated Coconut Untreated Pine Pine ASH Composition Shell Biochar Biochar #1 Biochar #2 Ultimate Analysis - Moisture free results Ash 6.7% 9.2% 3.6% Ash Composition Sodium Oxide, as 5.7% 1.2% 0.8% Na2O
Regardless, it should be clear that there are various methods available to produce final product with a targeted sodium level, making it suitable for various microbes depending on their preference for an environment with a certain sodium level.
[0213] 4. Usable Pore Size Distribution and Usable Pore Volume
[0214] One very important quality of a microbial habitat is the availability of shelter from environmental or biological hazards. A few examples of environmental hazards are high temperature, UV radiation, or low moisture, while an example of a biological hazard is the existing of predatory multicellular microbes such as protozoa, including both flagellates and ciliates. In order for a particle or material to provide shelter for microbes, at least two conditions must be present: (i) The material must consist of pores or openings of a size which can be inhabited by the microbe in question (ii) but prevent the hazard from entering (e.g. pore size smaller than the size of predators, such as protozoa, or deep enough to be shaded from UV rays) and, (iii) the pores mentioned previously must be usable--namely, they should not be occupied by solid matter (clogged) and/or they should not contain substances that are toxic or undesirable for the microbe in question. In some cases, the pore size distribution of a biochar can be adjusted by the selection of the biomass feedstock to be pyrolyzed and the conditions of the pyrolysis process itself. For example, pine wood has a relatively narrow pore size distribution, with most pores falling in the range from 10-70 .mu.m. Coconut shells, on the other hand, have a much wider size distribution, with many pores below 1 .mu.m, and also a high percentage of porosity above 100 .mu.m. It is theorized that materials with pores of a single size or where most pores are of similar size can potentially be good carriers or habitats for certain, targeted microbes, while materials consisting of broader ranges of pore sizes may be better habitats for communities, consortia or groups of microbes, where each microbe may prefer a slightly different pore size. Furthermore, the pore size of a material may also be controlled during the pyrolysis process by increasing temperature or performing "activation" or other steps common in activated carbon production to react or remove carbon, leaving larger pores, or exposing availability of pores that were once inaccessible from the exterior surface of the material. Adjusting the particle size of the material may also change the pore size distribution in at least two ways: (i) exposing pores that were not available or accessible previously, or (ii) destroying larger pores by fracturing, splitting, or dividing them. In many cases, raw biochar may contain a proper pore size distribution, but for one reason or another, the pores are not usable by the microbes in question. In other cases, the pore size distribution provided by the natural feedstock may be undesirable. Both properties may also be impacted through treatment of the raw biochar itself. Larger pores can be created using strong acids or other caustic substances either by simple washing or through forced or rapid infusion into the pores. Conversely, a material with fewer usable pores may be created by intentionally "clogging" or filling the larger pores with either solids, gums, or liquids designed to stay resident in the pores themselves. This treatment may be done in a controlled way to only partially fill the pores. For example, one could infuse a limited amount of heated liquid, such as a resin, that will become solid at normal atmospheric temperatures. If the volume of liquid used is less than the available pore volume of the material being infused, some of the porosity of the material will be left untreated and available for use. Most importantly, and most commonly, usable pore volume may be increased through the act of simply removing contaminants (physical or chemical) from the pores. Rapid infusion and extraction of liquids may be used to accomplish this. As discussed previously, appropriate solvents may be infused or extracted to remove chemical contaminants. Additionally, gasses or liquids may be driven into or out of the pores to force the removal of many solid obstructions, such as smaller particles of ash or simply smaller particles of raw biochar which may have become lodged in the pore in question. Regardless of the mechanism used, it has been shown that the available, uncontaminated, usable pore volume and pore size has a major role in determining the efficacy biochars in microbial roles.
[0215] FIGS. 23 and 24 are images that show how different sized bacteria will fit in different biochar pore size structures. FIG. 2.3 is rod-shaped gram-positive bacteria, Bacillus thuringiensis israelensis, in a treated pine biochar, with pore openings of .about.10-20 .mu.m and bacteria of .about.2-5 .mu.m. FIG. 24 is rod-shaped gram-negative bacteria, Serratia liquefaciens, in a treated coconut shell biochar, with pore openings of .about.2-10 .mu.m and bacteria of .about.1-2 .mu.m.
[0216] In addition, total pore volume in the size of 5-50 .mu.m has been shown to correlate with microbial release rate after inoculation on treated biochar. FIG. 25 illustrates release rate data verse total pore volume data for both coconut shell and pine based treated biochars inoculated with a releasable bacteria. As illustrated in FIG. 25, the data was plotted in a graph, and clearly, shows that as pore volume increases so does the release rate.
[0217] 5. Exterior and Interior Surface Geometry
[0218] Two important properties of microbial carriers are: (i) their ability to release microbes from their surfaces and (ii) their ability to immobilize or stabilize microbes on their surfaces. Depending on the final application or use of the carrier, one or both of these properties may be desired. For example, for carriers designed to quickly release a microbe into a targeted domain such as a lake, river, or other waterway, the release characteristics of the material are paramount. For other applications, such as applications of certain symbiotic microbes in agriculture, rapid release may be undesirable, rather it may be important to sustain the microbes within the porosity of the material until plant tissue, such as root biomass, is nearby to provide nutrition for the microbes in question. The surface and pore geometry of the material used as a carrier can be critical to determine this behavior. For example, material with generally smooth, uniform surfaces will typically release many microbes much more effectively, while material with more rugged, varied, tortuous pore surfaces and geometry will typically retain and immobilize microbes more effectively. The biomass used in the production of the final material is one of the most important factors in surface geometry. However, even this quality can be altered through treatment. Specifically, smooth surfaces may be etched by implementing the treatment and infusion processes previously disclosed with strong acids, rendering them rougher. Conversely, rough surfaces may be treated with either organic or inorganic compounds to coat and remove contour. Mechanical means may also be used to affect changes in particle geometry. Many forms of charred material have relatively low crush strength and are relatively brittle. The method used to grind, or size particles can have a large impact on the geometry of the final particles. For example, particles milled using a ball mill or other type of grinding technology will typically have a smoother exterior geometry after the milling is complete and may lose a good amount of their porosity through the simple mechanical crushing of pores. However, particles sized using ultrasonic vibrations or even simple physical vibrations to shatter, rather than crush larger particles into smaller ones, will typically retain their geometry, or sometimes result in smaller particles with more rugged geometries than the particles at the beginning. It should be apparent to one skilled in the art that there are various mechanical mechanisms available to effect these changes, but the resulting particles can be tailored to meet a particular microbial release or immobilization outcome.
[0219] 6. Particle Size and Distribution
[0220] It is well known that the particle size and particle size distribution of a material has a key impact on its formulation as a microbial carrier. In many cases, these factors are very different for porous carbonaceous materials than they are for other common microbial carriers. In standard carriers, typically the reduction of particle size is a method used to increase surface area, and thus the area available to support, immobilize, and carry microbes. However, in porous materials, specifically materials with a large volume of usable interior porosity, sometimes a reduction in particle size does not cause a large increase in the usable surface area specifically because the interior surfaces of the material were already exposed, and reducing the size of the particle does not change that fact. This leads to a somewhat counterintuitive behavior in some cases in which the reduction of the particle size of a porous material actually degrades its performance as a microbial carrier, due to the phenomena that surfaces that were once sheltered inside the material are exposed as exterior surfaces when the material is split or crushed, making the material less desirable as a habitat for microbes that require shelter from the surrounding environment. Additionally, at times the actual distribution of particle sizes can be a key factor in performance. As a simple example, imagine an aggregated material which consists of only two particle sizes: 1 mm and 1 .mu.m. Furthermore, imagine that 50% of the mass of the aggregate resided in the 1 mm particles with the remainder in the 1 .mu.m particles. Lastly, imagine that the 1 mm particles were porous carbonaceous particles with an average pore size of approximately 50 .mu.m. It should be clear that if this aggregate was placed in a container and agitated, that a good portion of the 1 .mu.m particles would end up inhabiting the pore volume of the 1 mm particles, impacting their usability. In fact, this is the behavior that we see in practice. Therefore, for certain microbial applications, it is desirable to remove extremely small particles, often referred to as fines, from the aggregate. This has the additional benefit of reducing dust during application, which is particularly important in aerial applications, and reducing the level of surface runoff for applications in water, which also is important in certain microbial applications. The small particles may be removed through several methods such as sieving, blowing or aerodynamic removal, separation with either stationary or moving liquids (hydrostatic or hydrodynamic separation) of various viscosities, temperatures, flow rates, etc. However, at times, having a mixture of smaller and larger particles can be desirable. The most common cases are when communities of microbes are to be deployed, or the aggregate is to remain generally intact for a period of time (fermentation applications, long term storage applications, or preparation for other formulation uses such as palletization), in which case, the interparticle void space is also an important factor and can be optimized for a particular microbe or set of microbes by providing a range of particle sizes and geometries.
[0221] 7. Nutrient Exchange
[0222] The ability of a material to hold or exchange nutrients is an incredibly important characteristic, not only for microbial, but also for general agricultural applications. There are two primary mechanisms that porous carbonaceous materials can exchange nutrients: (i) sorption or retention of the nutrients on the interior and exterior surfaces of the material, and (ii) retention of the nutrients either in suspension or solution in liquid or gasses residing in the pore volume of the material. Both mechanisms are very useful, but also very different in function. Surface sorption or retention is driven by two main properties, among others: (i) ion exchange capacity of the material and. (ii) reactivity or electrical charge of compounds present on or coating the surfaces of the material. Retention of nutrients in solution or suspension are impacted by other, different characteristics of the material, such as hydrophilicity, oil sorption capacity, usable pore volume and pore size distribution, and interior pore geometry and tortuosity. The surface retention of nutrients can be targeted by selecting the feedstock biomass (some materials render a char after pyrolysis with vastly differing ionic exchange capacities (CEC and AEC) than others). It can also be impacted by adjusting pyrolysis conditions. Higher pyrolysis temperatures tend to reduce CEC and nutrient adsorption capability. See Gai, Xiapu et al. "Effects of Feedstock and Pyrolysis Temperature on Biochar Adsorption of Ammonium and Nitrate." Ed. Jonathan A. Coles. PLoS ONE 9.12 (2014): e113888. PMC. Web. 19 Nov. 2016. In addition, the surface retention of nutrients can be impacted by treating the surfaces of the material with substances targeted towards adjusting the ionic exchange characteristics. For example, using the previously disclosed treatment methods to infuse H.sub.2O.sub.2 into the pores of the carbonaceous material and then evaporating the liquid can increase the cationic exchange properties of the material.
[0223] Furthermore, another way to exchange nutrients more efficiently is to use the pore volume rather than, or in addition to, the pore surfaces--namely keeping the nutrients in solution or liquid or gaseous form and placing them in the volume of the pores rather than attempting to sorb them on the surfaces of the material. This can be an incredibly useful technique not only for plant life and soil health, but also for microbes. The food sources can vary from simple to complex such as glucose, molasses, yeast extract, kelp meal, or bacteria media (e.g. MacConkey, Tryptic Soy, Luria-Bertani). When using the pore volume to exchange nutrients in this way, it should be clear that a wide variety of nutrients may be used, and targeted combinations of pore volume, size, and nutrition can be produced to assist in the delivery, establishment, or successful colonization of targeted microorganisms or groups of microorganisms. It should be clear by this point that merely immersing the biochar or porous carbonaceous material in a liquid nutrient broth may be partially effective in filling the pore volume or coating the pore surfaces with these nutrients and should be considered within the scope of this invention, however using the treatment techniques outlined in this and related disclosures is much more effective at both coating the surfaces and infusing nutrition into the pore volume of the material itself. Since many microbes rely on liquid for mobility, placing liquid into the pore volume of the material is in many cases a prerequisite for successfully infusing, carrying, or delivering microbes.
[0224] 8. Usable Carbon or Energy Sources
[0225] Related to the ability to improve nutrient exchange is the ability to treat the pore volume, pore surfaces, exterior surfaces, or any combination of these with not only custom broths or growth media, but also other forms of carbon known to be beneficial to microbes and plant life. Some examples of this are carbohydrates (simple and complex), humic substances, plant macro and micronutrients such as nitrogen (in many forms, such as ammonium and nitrates), phosphorous, potassium, iron, magnesium, calcium, and sulfur and trace elements such as manganese, cobalt, zinc, copper, molybdenum. These nutrients may either be infused in liquid or gaseous form, or even as a suspended solid in liquid. The liquid may be left in the pores, or may be removed. If removed through evaporation, nutrients in solution or suspended solids may be left behind, while if removed by mechanical or physical means, a portion of the liquid may be left behind as well as some solids. It should be noted that the various forms of removal have differing advantages and disadvantages and that many energy sources may be added either at the same time or in sequence, with one, or many, removal steps in between treatment or infusion steps.
[0226] 9. Toxic Materials or Compounds
[0227] The selective addition or removal of materials or substances known to be toxic to a certain microbe or lifeform is a key step in preparation of biochar for use as a microbial habitat or carrier. It has been shown, that through treatment, potentially toxic compounds can be removed with much greater effectiveness than through simple pyrolysis alone. Some examples of the potentially deleterious compounds that may be removed are: volatile organic compounds (VOCs), monoaromatics, polycyclic aromatic hydrocarbons (PAHs), heavy metals, and chlorinated compounds (e.g. dioxins and furans). A proven approach to remove these substances is to wash the exterior surfaces with and/or rapidly infuse a solvent into the pore volume of the material targeted to remove these substances. Following the infusion with either mechanical extraction, drying, or other methods to remove the solvent laden with the substances in question, from the pores and interparticle, spaces is a desirable, but not strictly necessary step to further reduce the levels of toxicity. For example, the following data shows removal of dioxins using the treatment process of the present invention.
TABLE-US-00007 Raw coconut Treated coconut Raw pine Treated pine shell biochar shell biochar biochar biochar TEQ ng/kg 0.7 0.4 9.6 0.4 (method 8290A)
[0228] Another approach for some toxic compounds (benzene as one example) is, rather than removing the compounds in question, to react them in place with other compounds to neutralize the toxicant. This approach can be used either with washing, or forced assisted infusion, and in these cases a removal step is less necessary although it still can be used to prepare the material for another, subsequent phase of treatment.
[0229] Much attention is given to the removal of toxic compounds, but it should be also be noted that at times, it can be extremely beneficial to actually add or treat the material with toxic compounds. A primary example of this is sterilization, or preparation for selective infusion. Even after pyrolysis, residual biological life has been found to potentially establish itself in biochars given the right conditions. Treating, washing, or infusing the material with antiseptics such as methanol, ethanol, or other antibacterial or antiviral substances can be a key step in removing contamination and preparing the material for use in microbial applications. A variation on this approach is to infuse, treat, or wash the material with a selectively toxic compound, such as a targeted antibiotic or pharmaceutical targeted towards interrupting the lifecycle of a specific set of microorganisms or organisms, thereby giving other microbes, either through infusion or merely contact in situ the opportunity to establish. Some examples of this treatment would be the use of antifungals such as cycloheximide to suppress fungal growth and provide an environment more well suited toward the establishment of bacteria. As has been stated previously, the methods may be used alone, or in combination with one another. Specifically, a toxic compound such as ethanol, may be infused, removed, and then steps may be taken to remove other toxic compounds, followed by steps to add carbon sources or growth media.
[0230] 10. Surface Structures/Crystals/Tortuosity
[0231] The physical surface and pore structure of the material is critically important to its suitability as a microbial habitat. There are many factors that contribute to the surface structure of the material. The most notable of these factors is the biomass used to produce the carbonaceous material the cellular structure of the biomass dictates the basic shape of many of the pores. For example, pyrolyzed coconut shells typically have less surface area, and a more diverse distribution of pore sizes than pyrolyzed pine wood, which, when pyrolyzed at the same temperature, has greater surface area, but a more uniform (less diverse) pore size distribution. Tortuosity, or the amount of curvature in a given path through a selected pore volume is also an extremely important characteristic of engineered porous carbonaceous materials.
[0232] FIG. 26 shows the total fungi/bacteria ratio for two biochars derived from different biochar starting materials, e.g., feedstocks. Each biochar was loaded with different levels of moisture, and the total fungi/bacteria ratio was monitored during the first week. Biochar A 2301 showed a constant total fungi/bacteria ratio of 0.08 across moisture levels range 5000 ng from 15% to 40%, while Biochar B 2302 showed a constant total fungi/bacteria ratio of 0.50 for moisture levels ranging from 30% to 40%. It is theorized that, a fungi/bacteria ratio between 0.05 and 0.60 is an effective prescription for a stable biochar composition. This composition allows a commercially viable product, which has sufficient shelf life that it can be delivered to storage houses waiting for the proper planting window.
[0233] It is theorized that the difference in the observed total fungi/total bacteria ratios of may also be explainable by the structures of the biochars. Biochar's having an open pore structure, e.g., more interconnected pores, promotes more bacteria formation; while closed pores, e.g., relatively non-connected nature of the pores, tends to promote fungi formation. Biochars with differing microbial communities may be beneficial for specific applications in commercial agriculture. Thus, custom or tailored loading of the microbial population may be a desired implementation of the present invention.
[0234] For example, as shown in FIGS. 27a, 27b and 27c, Biochar A 2701 shows that it has a greater population of, i.e., is inhabited by, more gram negative, gram positive and actinomycetes than Biochar B 2702. Thus, for example, Biochar A would be more applicable for use with certain agricultural crops in which Plant Growth Promoting Bacteria (PGPB) species in the actinomycetes, gram (-) pseudomonas, and bacillus groups are used for nutrient utilization and uptake.
[0235] It should be noted that both pyrolysis and post-treatment can be used to further modify the shape of these pores and structures. Pyrolyzing at higher temperatures, injecting select gasses or liquids during pyrolysis, or both typically will increase the pore volume and surface area of the material in question. Steam is the most readily available gas to cause this effect, but hydrogen sulfide, carbon dioxide, carbon monoxide, as well as other reactive gasses can be used. Prior art has clearly shown that the surface area of a biochar changes based on feedstock and pyrolysis temperature. Post treatment focused on a forced infusion of a strong acid, or other reactive substance into the pore space of the carbonaceous material can also be used to modify the pore size and pore volume of material by removing or breaking down the carbon matrix which forms the structure of the biochar, or other porous carbonaceous material. Acid etching or infusion can also be used to make smoother surfaces rougher. Rough surfaces can be very useful in the attachment and immobilization of microbes. Smooth surfaces can be useful for the easy release of carried microbes. Coating the surface area with materials such as starches is a technique to make rough surfaces smoother. Ultrasound, with or without a transmission media (gel, liquid, oil, or other) can also be used to rupture interpose divisions and create more pore space. Flash gasification either at atmospheric pressure, or under negative or positive pressure, of liquid infused into the pores by the methods previously disclosed can also be used to crack, disrupt, or fracture solid material separating adjacent pores.
[0236] While much attention is given to modifying the pore structure by removing carbonaceous material, it should be noted that the pore structure can also be modified by the coating, forced infusion, and/or addition of materials which will bond to the carbon and consume pore volume, smooth surfaces, add tortuosity, change the exterior surfaces, or all of these. In the most simple form, it should be clear that materials may be added to coat surfaces or fill pore volume either through forced infusion, simple contact, or other means. However, if the material is infused or even simply contacted with a super saturated solution of a substance that will crystallize, such as sucrose, sodium chloride, or other common or uncommon substances known to form crystals. It should be noted that the crystals or substances used to create them do not need to be water soluble, and in fact in many cases it is desirable if they are not. The crystals may also be composed of nutrients or substances which may be beneficial to microbial or plant life. Examples of this are sucrose and monoammonium phosphate, both known for their ability to easily crystallize and be beneficial for microbial and plant life respectively. By adding material or even growing crystals on the carbon, a hybrid material is formed which can have many properties that are exceptionally useful for the delivery and establishment of microbial systems. Crystallization is also way to add tortuosity to a carbonaceous material and typically is much more effective in this aspect than coating with solids alone.
[0237] 11. Compatibility with Biofilm Formation
[0238] Biofilms can be an important factor in the survival of a microbe in extreme or challenging conditions. Bacterial communities can shift their morphology to increase nutritional access and decrease predation. One such modification is that the bacteria may attach to surfaces, such as those found in biochar, in a densely compacted community. In this compacted form, they may form an extracellular polymeric substance (EPS) matrix called a biofilm. These communities can contain hundreds of different species which find shelter under the protective EPS coating from predatory protozoa, pathogens, contaminants, and other environmental stressors. In some cases, usually related to public health or healthcare, biofilms are undesirable as they typically allow pathogenic microbes to survive exposure to antiseptics, antibiotics, predatory microbes such as protozoa, or other agents which may eliminate them or negatively impact their prospects for survival. But in agricultural settings, encouraging target biofilm establishment could lead to improved microbe survival and thus improved agricultural or crop benefits.
[0239] As outlined in the article titled The Effect of Environmental Conditions on Biofilm Formation of Burkholderia psudomallei Clinical Isolates, it can be seen that certain bacteria require certain environmental factors, among them surface pH, for the creation of biofilms. See Ramli, et al., The Effect of Environmental Conditions on Biofilm Formation of Burkholderia psudomallei Clinical Isolates (Sep. 6, 2012) (http://dx.doi.org/10.1371/journal.pone.0044104). It is believed that other surface characteristics (rugged vs. smooth surfaces, surface charge, and more), along with moisture levels and relative humidity also play a large role in biofilm formation.
[0240] But for certain microbes requiring deployment into environments known to present survival challenges, optimizing a delivery material to encourage the formation of these protective biofilms can provide the targeted microbes with a significant advantage. Also, many vegetable and short cycle row crops such as tomatoes, lettuce, and celery form mutualistic relationships with bacteria that lead to the formation of biofilms on root hairs that function not only in nutrient uptake but also in plant pathogen resistance.
[0241] As outlined in previous disclosure, treatment of raw biochar can be used to adjust the surface pH to a level suitable for biofilm formation. Similarly, adjusting the humidity by selectively leaving a measured or controlled amount of water resident in the pore volume of the material can also provide benefit. Lastly, the techniques outlines for modifying the physical surface properties of the material either by smoothing or roughening, can be key factors also.
[0242] It should be clear that these factors can also be reversed to create an environment that is unsuitable for biofilm formation in applications where the formation of biofilms on the carrier is not desirable--e.g. delivery or applications where quick release of microbes from the carrier is important.
[0243] 12. Surface Charge
[0244] The surface charge of a porous carbonaceous material can be crucially important in the association and establishment of targeted microbes with or on the material. For example, most bacteria have a net negative surface charge and in certain conditions a specific bacterium may favor attachment to positively charged surfaces. In some biological applications, this attachment may be preferred, in others, attachment may not be preferred. However, modifying the surface charge of the material is clearly a way to impact the suitability for attachment of certain microbes. There are many ways in which the surface charge of a carbonaceous material may be changed or modified. One way to accomplish this is by treating the surface area of the material with a solution containing a metal, such as Mn, Zn, Fe, or Ca. This can be performed either by doping the material with these metals prior to or during pyrolysis, or more preferably, by using a forced infusion or treatment technique after pyrolysis to deposit these substances on the interior and/or exterior surfaces of the carbonaceous material. By controlling the amount and or types of substances infused, the surface charge of the material can be modified by encouraging loading of O.sub.2.sup.- or other anions, or conversely, N.sup.+, NH.sub.2.sup.+, or other cations. This modification of surface charge can have a profound impact on the ability of certain microorganism to be immobilized on the interior and exterior surfaces of the material.
[0245] Another application of surface charge can be found by temporarily charging the carbonaceous material during inoculation with microbes. Carbon is used as a cathode or anode in many industrial applications. Because of its unique electrical properties, carbon, or more specifically porous carbonaceous materials, may be given a temporary surface charge by the application of a difference in electrical potential. One application of this mechanism is to create a temporarily positively charged surface to encourage microbial attachment. Then, while the charge is maintained, allowing the microbes to attach themselves to and colonize the carrier. Once the colonization is complete, the charge can be released and the carrier, laden with microbes can either be deployed as is, or can undergo further treatment to stabilize the microbes such as lyophilization, or freeze drying.
[0246] 13. Enzyme Activity
[0247] For some types of microbes, enzyme activity, or the presence of certain enzymes is every bit as important as the availability of energy or nutrition. Enzymes can be critical in the ability of microbes to metabolize nutrition, which in turn can be a key element of reproduction, survival, and effective deployment. There are six main types of enzymes: hydrolases, isomerases, ligases, lyases, oxidoreductases, and transferases. These enzymes can be important in microbial applications. Through treatment or even simple contact, enzymes, like nutrients and energy sources, can be deposited on the surfaces or within the pore volume of porous carbonaceous materials, either as solids, or in solution/suspension, ensuring the enzymes are not degraded through the process. However, forced infusion of enzymes through the treatment processes previously outlined allows for much greater storage capacity and much greater levels of contact with the interior surfaces of the biochar, and as such, is preferable to simple contact. In some cases, the carbonaceous material can be used to deliver enzymes alone into an environment where both a habitat and enzymes are needed to promote or encourage the growth of certain indigenous microbes.
[0248] Another important aspect of enzyme activity is that some bacteria make extra-cellular enzymes which could be bound by the biochar or either reduce or even stop biochemical reactions. Thus, in certain situations when application is appropriate the carbonaceous material can be used to inhibit or make certain enzymes ineffective. For example, if the biochar is being used as a carrier for food or certain chemicals that are vulnerable to breakdown by enzymatic degradation and these specific enzymes would be bound by the biochar, then using the carbonaceous material as the carrier would provide for greater shelf-life and viability of the product versus traditional carriers.
[0249] 14. Sterilization
[0250] In many cases, it is desirable to remove potential unwanted microbes from the surfaces and pore volume of the material through sterilization. At outlined above, infusion with antiseptics or antibiotics are a way to accomplish this. Boiling, or more preferably, forced infusion of steam is also a technique that can be used to remove resident microbial life. Heating to a temperature above 100 degrees C., and preferably between 100 and 150 degrees C. is also effective for removing some microbial life. Heating may be required for ideally 30 minutes or more, depending on volume, method, and extent (temperature, radiation). Autoclaving can also be used 30 minutes, 121 degrees C., 20 psig. For applications requiring a high level of sterility, gamma irradiation can be used, with dosages adjusted for the level of sterility needed in ranges of 5 to 10 kGy or even 50 to 100 kGy or even higher dosage levels. For all sterilization methods, the extent of treatment required will depend on the volume of material and the required level of sterilization. In general, sterilization, using heat, should be done for at least 30 minutes, but should be adjusted as needed.
[0251] At this point, it should be clear that all of these properties can be controlled and modified to create a treated, controlled biochar that is suitable for use as a microbial carrier, delivery system, habitat, fermentation substrate, or environmental (soil, water or other) enhancement. By controlling these properties and producing a material matched to the application and the microbe(s) in question, effectiveness can be dramatically improved over both traditional biological carriers, and many forms of raw, untreated, uncontrolled biochar. Furthermore, varying materials, with varying properties, may be aggregated to provide delivery systems or habitats targeted towards consortia, communities, or groups of microbes.
[0252] E. Inoculating, Applying, or Immobilizing the Microbes on the Biochar
[0253] Typically, the prior art teaches either placing biochar on soils alone or combining the biochar with compost and using this mixture as a soil amendment. The nature of the microbial population in this compost mixture is poorly disclosed by the prior art. Thus using more targeted methods to get the desired microbes into the suitable habitat created by the raw biochar, or more preferably treated or controlled biochar is desired. The following are some but not all, methods and systems that can be used to inoculate, deploy, or otherwise associate microbial life with a treated or untreated biochar:
[0254] 1. Co-Deployment
[0255] This method focuses on deploying the microbes at the same time as the biochar. This can be done either by deploying the biochar into the environment first, followed by microbes or by reversing the order, or even deploying the two components simultaneously. An example of this would be the deployment of a commercial brady rhizobium inoculant simultaneously with the introduction of a treated biochar into the soil media. The system here is the combination use of a biochar and microbes in the environment, and more preferably a char treated to have suitable properties for a target microbe or group of microbes which it is used with in a targeted application for a specified purpose, for example a symbiotic crop of said microbe(s).
[0256] In one experiment, various biochar feedstocks with various post-treatments were added to a soilless mix containing soybean seeds that had been treated with a commercial microbial product containing bradyrhizobium japonicum, and compared to both a control with microbe inoculant and one without. Some of the treated biochars co-deployed with the inoculant increased seed germination rates, one by 29%. Others increased nodulation measured at 10 weeks, one more than doubled the number of nodules. The use of the microbial inoculant increased shoot biomass in all treatments. FIG. 28 is a chart comparing shoot biomass when the biochar added to a soilless mix containing soybean seeds is treated with microbial product containing bradyrhizobium japonicum, and when it is untreated. As illustrated in FIG. 28, shoot biomass increased with the biochar was treated.
[0257] FIG. 29 shows the comparison of root biomass in a microbial inoculated environment versus one without inoculation. As illustrated in FIG. 29, when inoculated, root biomass decreased with the inoculant alone yet increased with the use of all the treated biochars with or without inoculant.
[0258] In addition leaf tissue analysis was done which showed some of the treated biochars co-deployed with the rhizobial inoculant showed a significant increase in nitrogen uptake. FIG. 30 is a chart comparing the nitrogen levels when the biochar is inoculated with the rhizobial inoculant verses when it is not inoculated. Statistical significance in the chart in FIG. 30 is marked with a star. In all cases, nitrogen levels increase with inoculation.
[0259] As outlined in these results, the addition of a treated biochar suitable for co-deployment with this particular microbe increased nodulation, increased nitrogen fixation/availability, and resulted in substantially increased root mass. It should be noted that to demonstrate the differing performance of varying formulations, two formulations were tested, each showing different interactions with the microbe in question, along with significant variations in performance. This is just one example to demonstrate the invention of how the specific combination of biochar feedstock, biochar treatment, co-deployed microbe, and application (this case plant species) can lead to improved microbial effectiveness and thus improved results (this case plant vigor), versus no treatment, applying the microbe alone, or applying the biochar alone. Another example of co-deployment benefit could be using a biochar that has strong absorption properties in combination with fertilizer (or infused with fertilizer) and microbes in an agricultural setting. The biochar properties that help retain and then slowly release nutrients and ions will also help the targeted microbe population to establish and grow without being impacted by the high levels of fertilizer salts or nutrients which can often impede and sometimes kill the microbes being deployed.
[0260] 2. Basic Inoculation
[0261] A more advanced method of inoculation centers on mixing the microbe or microbes in question with the treated or untreated biochar before deployment. In some cases, the biochar in question can be treated, produced, or controlled to assist with this deployment, making this case slightly different than merely inoculating a microbe on untreated biochar. In one form, microbes suspended in liquid (either water, growth media, or other liquids) are deposited on the biochar and mixed together until both materials are well integrated and then the material is deployed as a granular solid. It has been shown that materials that have been treated to be more hydrophilic typically accept this inoculation more readily than hydrophobic materials--demonstrating yet another way in which the treatment of biochar can enhance performance. In another form of basic inoculation, the biochar is delivered in suspension in the liquid also carrying the microbes. This biochar/liquid/microbe slurry is then deployed as a liquid. In this form, sizing the biochar particles in such a way that their surface properties and porosity is maintained is a key element of effectiveness. Additionally, ensuring that the pores are treated to allow easy association of both liquid and microbes with the surfaces of the biochar is important. An example of a basic inoculation method of biochar for a bacteria in lab scale is as follows: [0262] 1) Isolate Pseudomonas protegens on a plate with 1.5% w/v Tryptic Soy Broth solidified with 1.5% w/v agar and incubate at 30.degree. C. for 12 h [0263] 2) Take an isolated colony of Pseudomonas protegens and grow up in a 1.5% w/v TSB solution (90 ml) along with 10 g sterile biochar (sterilized at 110 C in small batches for 15-20 min) and combine both in a sterile 250 ml Erlenmeyer flask [0264] 3) Shake contents of flask at 150 rpm at 30.degree. C. for 12 h, or greater [0265] 4) Transfer contents of flask into a sterilized ultracentrifuge tube (250 ml) and spin at 10,000.times.g for 10 min [0266] 5) Carefully remove supernatant liquid fraction by filtering through a Whatman No 4 filter with a vacuum filtration system to separate out the bulk liquid from biochar. After basic inoculation, the material and the microbes may be deployed immediately, stored for future use, or stabilized using technology such as lyophilization.
[0267] 3. Assisted Inoculation
[0268] Another form of inoculation, which appears to have greater efficacy with some microbial systems, is assisted inoculation. Assisted inoculation involves providing mechanical, chemical, or biological assistance to move the targeted microbe either into the pore volume of the carrier or onto interior surfaces of the material that normally may not be accessible. Realizing that many microbes require liquid, and preferably water, for mobility, the most straightforward method of assisted inoculation requires infiltrating the pore volume of the material with water prior to contact with the targeted microbes. This water infusion can be done using the treatment methods described previously in this disclosure. It has been shown that, with certain microbes, making this change alone will have a positive impact on the ability of microbes to associate with and infiltrate the material. In one experiment, it was shown that water infusion improved release rate on both a treated pine biochar with granular particles and with a coconut biochar powder. FIG. 31 illustrates the three-day release rates of water infused biochar compared to other types of biochar. As illustrated, results vary depending upon the biomass.
[0269] Changes can also be made in the media to reduce surface tension and increase flowability through the addition of a surfactant to the water, either into the liquid used to carry the microbes, or into the pores of the material itself, through simple contact, or preferably forced infusion.
[0270] Additionally, the microbes themselves may be assisted into the pores using the treatment techniques previously outlined. Care needs to be taken to match the microbe to the technique used, but many microbes are capable of surviving vacuum infiltration if performed at relatively gentle, lower pressure differentials (+/-10% of standard temperature and pressure). Some microbes, and many spores however are capable of surviving vacuum infiltration even at relatively large pressure differentials (+/-50, 75, or even 90 or 95% or more variation from standard temperature and pressure). When this technique is used, a liquid mixture is constructed containing both liquid to be infused and the microbe or microbes in question. The liquid is then used as the "infiltrant" outlined in previous disclosure related to placing liquid into the pore volume of the material. The final material, infiltrated with microbes, may then be heated to incubate the microbes, cooled to slow development of the microbes or stabilize the microbes, or have other techniques applied such as lyophilization. The material may then be delivered in solid granular form, powdered, further sized downward by grinding or milling, upward by agglomerating, aggregating, or bonding, or suspended in a liquid carrier. A clear advantage to this assisted infusion approach is that the material can be processed or handled after inoculation with more microbial stability because the targeted microbes are inhabiting the interior pore volume of the material and are less prone to degradation due to contact with exterior surfaces, or other direct physical or environmental contact. This method may be applied repeatedly, with one or more microbes, and one to many moisture removal steps. It may also be combined with the other inoculation methods disclosed here either in whole or in part.
[0271] FIGS. 32a, 32b and 32c show scanning electron microscopy (SEM) images of raw biochar compared to ones that have been processed by being infused under vacuum with bio-extract containing different microbial species.
[0272] FIG. 32a is a SEM (10 KV.times.3.00K 10.0 .mu.m) of pore morphology of raw biochar. FIG. 32b is a SEM (10 KV.times.3.00K 10.0 .mu.m) of pore morphology of raw biochar of FIG. 32a after it has been infused with microbial species. FIG. 32c is a SEM (10 KV.times.3.00K 10.0 .mu.m) of a pore morphology of another example of raw biochar of FIG. 32a after it has been infused with microbial species. The images confirm the ability to incorporate different microbes into the pores of biochar by treatment. In turn, these beneficial microbes can interact with and enhance the performance of the environment they are deployed into, for example the plants' root systems when the inoculated biochar is mixed with the soil in the root zone.
[0273] Compared to a biochar that has immersed in a compost tea, which may have a relatively short, e.g., a few days for the life of the microbes, the impregnated populations of examples of the present treated biochars, are stable over substantially longer periods of time, e.g., at least an 8 week period and in some cases 1 year or more as measured by PLFA (Phospholipid-derived fatty acids) analysis. PLFA analysis extracts the fatty acid side chains of phospholipid bilayers and measures the quantity of these biomarkers using GC-MS. An estimate of the microbial community population can thus be determined through PLFA analysis. The microbial activity may also be inferred through PLFA analysis by monitoring the transformation of specific fatty acids. Thus, the impregnation of the biochar with a microbial population provides for extended life of the microbes by at least 5.times., 10.times., or more over simple contact or immersion. In fact, some microbes may be better suited to surfactant infiltration versus vacuum infiltration and vice versa and this may impact the shelf life, penetration, viability, or other characteristics of the microbes.
[0274] As used herein, unless stated otherwise, the stable shelf life of an example of a biochar product having a microbial population is the period of time over which the product can be stored in a warehouse, e.g., dry environment, temperature between 40.degree. F.-90.degree. F., with a less than 50% decrease in microbial population.
[0275] 4. Integrated Growth/Deployable Substrate
[0276] With many microbes, especially fungi, it can be helpful to develop or "grow" the microbes on the material itself. With porous materials, rather than mechanically or chemically assisting the infiltration of the microbes, it can be beneficial to allow the microbes themselves to inhabit the pore volume of the material prior to deployment. In fact, with materials constructed to effectively immobilize microbes, this can be the most efficient technique to stabilize, store, and ultimately deploy the microbes in question.
[0277] An example of this method involves preparing the biochar material for the microbes, sometimes through thorough cleansing, other times through addition of either enzymes or energy sources needed by the microbe in question, preferably using the treatment techniques described previously in this disclosure. Once the material is prepared, the microbes are placed onto the material, or infused into the material and then incubated for a period of time. In the case of many microbial systems, the microbes themselves will inhabit the material and form close affiliations with available surfaces and pore volume. At this point, the material can be deployed with the microbes actively attached and affiliated. With many microbes, especially fungi, this is a preferred method of deployment and shows many advantages over co-deployment, or basic inoculation because of the tight integration of biological life with the material itself.
[0278] An example of an integrated growth inoculation method of biochar for a fungus in lab scale is as follows: [0279] 1) Make petri dishes containing corn meal agar (17 g/L), glucose (10 g/L), and yeast extract (1 g/L) [0280] 2) Inoculate plates with Sordaria fimicola and incubate between 22-30 C for at least 1 day to produce hyphae [0281] 3) Sterilize an inoculating loop and slice "plugs" of the hyphae and agar generating cubes that are agar and hyphal mass [0282] 4) Inoculate a sterile plate with a "plug" in the center of the plate, around perimeter have sterile biochar [0283] 5) Incubate plate for at least a day and remove biochar (that are now covered with grown over hyphae)]
[0284] It should be noted that because of this effect, biochars, and specifically treated biochars can also be extremely effective substrates for solid state fermentation--particularly when growth media or energy sources are added to the pore volume of the material. So, once incubation is ongoing, the material can either be removed, with the integrated microbes, and deployed, or it can be stabilized for long term storage, or it can be left in situ and used as a fermentation or growth substrate to develop or grow more microbes--especially those that require a solid to propagate and develop.
[0285] 5. Media and/or Enzyme Infiltration
[0286] As mentioned previously, growth media, energy sources, enzymes, or other beneficial/necessary components for microbial growth may be infused into the pore volume or coated onto the surfaces of the material in question. This method can be combined with any of the other inoculation techniques disclosed here. It has been shown that with certain microbes and certain types of material, inoculation with growth media or enzymes can significant impact the effectiveness of the biochar material as a carrier.
[0287] 6. Habitat Pre-Establishment (Enhanced Rhizosphere)
[0288] There are certain microbes which, because of symbiotic associations with host organisms, such as plants, prefer to develop in the vicinity of the organism, such as the active root or other plant tissue. An effective method for deploying these organisms can be to develop and deploy the plant/microbe/habitat (biochar) system together as a unit.
[0289] An example of this is germinating seed or transplanting a seedling or developing juvenile plant in the presence of treated or untreated biochar, and the targeted microbes. Biochar that has been treated to encourage hydrophilicity and neutral pH typically allows for easier affiliation of plant root tissue with the material. As this affiliation occurs, a habitat for symbiotic organisms is developed within the material itself due to the proximity of active plant tissue to microbes reliant on the tissue for energy. As this symbiosis continues, the number, activity, and colony size of the targeted microbes will continue to grow. At this point, the plant and biochar can be deployed together into the target environment, acting as a pre-established habitat and carrying the microbes along with them.
[0290] Another option is to develop and then remove the biochar from the "incubation" system either by stripping the biochar material from the symbiotic organism, such as the root mass, or by sieving or sifting the media used to grow the plant. At this point, the microbes can either be deployed directly or stabilized for storage.
[0291] Thus, through more controlled inoculation of the biochar particles, one can achieve a predetermined and controllable amount of a microbial community, e.g., population, into the soil. This integration of a microbial community with a biochar particle, and biochar batches provides the ability to have controlled addition, use and release of the microbes in the community. In agricultural applications, this integration c a n further enhance, promote and facilitate the growth of roots, e.g., micro-roots, in the biochar pores, e.g., pore morphology, pore volume.
[0292] Other methods than those listed above exist for integrating a microbial community with an untreated or previously infused biochar particle. Different manners and methods would be preferred depending on needs to minimize contamination, encourage biochar pore colonization/infiltration, minimize labor and cost and producing a uniform, or mostly uniform, product.
[0293] Other methods for integrating a microbial community with a biochar particle may include, but are not be limited to the following: while under vacuum, pulling the microbial solution through a treated biochar bed that is resting on a membrane filter; spraying a microbial solution on top of a treated biochar bed; lyophilizing a microbial solution and then blending said freeze dried solution with the treated biochar; again infusing, as defined previously, the treated biochar with a microbial solution; adding treated biochar to a growth medium, inoculating with the microbe, and incubating to allow the microbe to grow in said biochar containing medium; infusing, as defined previously, the biochar with a food source and then introducing the substrate infused biochar to a microbe and incubating to allow the microbes to grow; blending commercially available strains in dry form with treated biochar; adding the treated biochar to a microbial solution and then centrifuging at a high speed, potentially with a density gradient in order to promote the biochar to spin down with the microbes; densely packing a column with treated biochar and then gravity flowing a microbial solution through the column and possibly repeating this multiple times; or adding the microbe to a solution based binder that is well known to enter the treated biochar pores and then adding said solution to the treated biochar. In order to insure the proper microbial community the treated biochar may need to be sterilized prior to these methods for integrating a microbial community. All or parts of the above manners and methods may be combined to create greater efficacy. In addition, those skilled in the art will recognize that there may be additional manners or methods of infusing biochars with microbials, including those created by the combination of one or more of the manners and methods listed above, without departing from the scope of the present invention.
[0294] F. Using Microbial Inoculated Biochars
[0295] Thus, treated biochar can have a microbial community in its pores (macro-, meso-, and combinations and variations of these), on its pore surfaces, embedded in it, located on its surface, and combinations and variations of these. The microbial community can have several different types, e.g., species, of biologics, such as different types of bacteria or fungi, or it may have only a single type. For example, a preferred functional biochar, can have a preferred range for bacterial population of from about 50-5000000 micrograms/g biochar; and for fungi, from about 5 to 500000 micrograms/g biochar. A primary purpose in agricultural settings, among many purposes, in selecting the microbial population is looking toward a population that will initiate a healthy soil, e.g., one that is beneficial for, enhances or otherwise advance the desired growth of plants under particular environmental conditions. Two types of microbial infused biochars will be discussed further for agricultural settings: bacteria and fungi. However, the microbes may also be used in other applications, including but not limited to animal health, either directly or through interactions with other microbes in the animals' digestive tract and public health applications, such as microbial larvicides (e.g. Bacillus thuringiensis var. israelensis (Bti)) and Bacillus sphaericus used to fight Malaria).
[0296] G. Bacteria Inoculated Biochars
[0297] PGPB include, for example, plant growth promoting rhizobacteria, free-living and nodule-forming nitrogen fixing bacteria, organic decomposers, nitrifying bacteria, phosphate solubilizing bacteria, biocontrol agents, bioremediation agents, archea, actinomycetes, thermophilic bacteria, purple sulfur bacteria, cyanobacteria, and combinations and variations of these.
[0298] PGPB may promote plant growth either by direct stimulation such as iron chelation, phosphate solubilization, nitrogen fixation and phytohormone production or by indirect stimulation, such as suppression of plant pathogens and induction of resistance in host plants against pathogens. In addition, some beneficial bacteria produce enzymes (including chitinases, cellulases, -1,3 glucanases, proteases, and lipases) that can lyse a portion of the cell walls of many pathogenic fungi. PGPB that synthesize one or more of these enzymes have been found to have biocontrol activity against a range of pathogenic fungi including Botrytis cinerea, Sclerotium rolfsii, Fusarium oxysporum, Phytophthora spp., Rhizoctonia solani, Pythium ultimum.
[0299] Currently the most economic conventional solid carrier used to deliver microbes is peat. A solid carrier allows for a relatively long shelf life and a more direct application to a plant's root system compared to a microbial liquid solution, which would be sprayed directly.
[0300] Research has shown a substantial increase in PGPB growth and distribution resulting from being infused in biochar. For example, data resulting from research conducted to compare the effects upon CO2 production (an indicator of bacterial growth) using peat and biochars show the beneficial effects of using various biochars in promoting PGPB growth. As illustrated in the left-hand chart in FIG. 33, peat results in CO2 production of between approximately 10% and 30% (depending upon the grown medium), whereas biochars result in CO2 production of approximately 48% and 80%. Replicated experimental results using different biochars confirm CO2 production of approximately 30% to 70% (depending on the grown medium), as compared to approximately 10% to 20% for the peat control.
[0301] The method developed for determining this CO2 production as an indicator of bacterial growth consists of the following. The substrate, here biochar or peat, is sterilized by heating at 110 C for 15 hours. A bacterial stock solution is then created, here Tryptic Soy Broth was solidified with agar at 1.5% w/v in petri plates to isolate the gram negative non-pathogenic organism Escherichia coli ATCC 51813 (15 h growth at 37.degree. C.). Then an isolated colony is captured with an inoculating loop and suspend in 10 ml sterile buffer (phosphate buffer saline or equivalent) to create the bacterial stock solution. Lactose containing assays are then used, here, test tubes that contain 13 ml of either Lauryl Tryptose Broth (LTB) or Brilliant Green Broth (BGB) that also contain a Durham tube. A negative control is generated by adding 10 .mu.L of sterile buffer to triplicate sets of LTB and BGB tubes. A positive control is generated by adding 10 .mu.L of bacterial stock solution to triplicate sets of LTB and BGB tubes. A negative substrate is generated by adding 1.25 ml (.about.1% v/v) of sterile substrate to triplicate sets of LTB and BGB tubes. A positive substrate is generated by adding 1.25 ml (.about.1% v/v) of sterile substrate and 10 .mu.L of bacterial stock solution to triplicate sets of LTB and BGB tubes. The tubes of the four treatments are then incubated statically in a test tube rack at 37.degree. C. for at least 15 h. The tubes are then carefully observed and any gas bubbles captured by the Durham tube within respective LTB or BGB tubes are closely measured with a ruler. Small bubbles <0.2 mm should not be considered. A continuous bubble as shown in individual tubes in FIG. 34 are what are observed and quantified. FIG. 34 is an example of carbon dioxide production captured as a continuous gas bubble in BGB (left two tubes) and LTB (right two tubes) growth medium. The percent carbon dioxide production is then calculated by dividing the recorded bubble length by the total Durham tube length and multiplying by 100.
[0302] Further tests were conducted using the Streptomyces lidicus WYEC 108 bacterium found in one of the commercially available products sold under the Actinovate brand. Actinovate products are biofungicides that protect against many common foliar and soil-borne diseases found in outdoor crops, greenhouses and nurseries. The formulations are water-soluble.
[0303] FIG. 35 illustrates the effects upon the growth of Streptomyces lidicus using conventional peat versus biochars. In the test illustrated by the photograph on the left of FIG. 35, an Actinovate powder was blended with peat, placed in an inoculated media and incubated at 25.degree. C. The photograph shows the distribution and density of white colonies after 3 days. In the test illustrated by the photograph on the right of FIG. 35, an Actinovate powder was blended with the treated biochar, placed in an inoculated media and incubated at 25.degree. C. The photograph also shows the distribution and density of white colonies after 3 days, the distribution and density of which are significantly greater than those achieved with peat.
[0304] FIG. 36 further illustrates the improved growth of the Actinovate bacterium using biochar versus peat. The left photograph shows only limited and restricted growth away from the peat carrier. The right photograph shows abundant growth of the bacterium spread much farther out from the biochar carrier.
[0305] Another application of using biochar inoculated with bacteria would be in the biofuel industry, where methanotroph inoculated biochar could be used to create methanol. Methanotrophic bacteria are proteobacteria with diverse respiration capabilities, enzyme types, and carbon assimilation pathways. However, Methylosinus trichosporium OB3b is one of the few methanotrophs that can be manipulated by environmental conditioning to exclusively produce methanol from methane. M. trichosporium OB3b is one of the most well studied aerobic C.sub.1 degraders and can be grown in either batch or continuous systems. As mentioned earlier, the large pore volume and surface area of biochar is suitable for bacterial colonization and should subsequently increase substrate access to enzyme activation sites. To improve the conversion rate, copper, nitrate, and phosphate should be included in the system. The use of biochar as a support material for the aerobic bioconversion of methane to methanol provides a pore distribution suitable for both adsorptions of methane and impregnation with bacteria. By providing biological and adsorptive functionality the biochar can intensify the bacteria in the biochar and increases the conversion rate.
[0306] In general, bacteria communicate via the distribution of signaling molecules which trigger a variety of behaviors like swarming (rapid surface colonization), nodulation (nitrogen fixation), and virulence. Biochars can bind signaling molecules and in particular it is believed can bind a major signaling molecule to their surface. This binding ability can be dependent upon many factors including on the pyrolysis temperature. This dependency on pyrolysis temperature and other factors can be overcome, mitigated, by the use of examples of the present vacuum infiltration techniques. For example, a signaling molecule that is involved in quorum sensing-multicellular-like cross-talk found in prokaryotes can be bound to the surface of biochars. Concentration of biochars required to bind the signaling molecule decreased as the surface area of biochars increased. These signaling molecules may be added to the surface of a biochar and may be used to manipulate the behavior of the bacteria. An example of such a use would be to bind the molecules which inhibit cell-to-cell communication and could be useful in hindering plant pathogens; using techniques in the present invention signaling molecules may be added to the surface of a biochar to engineer specific responses from various naturally occurring bacteria.
[0307] H. Fungi Inoculated Biochars
[0308] Beneficial fungi include, for example, saprotrophic fungi, biocontrol fungi, ectomycorrhizae, endomycorrhizae, ericoid mycorrhizae, and combinations and variations of these. It is further theorized that, in general, biochars with greater fungal development may be better suited for perennial crops such as grapes, almonds, blueberries, and strawberries in which symbiotic relationships with arbuscular mycorrhizal fungi (AMF) are favored over PGPBs. The presence of high concentrations of AMF spores in biochars can therefore rapidly promote fungal colonization of plant root hairs leading to extensive mycelial development. Increased plant root associations with mycelial filaments would consequently increase nutrient and water uptake.
[0309] Mycorrhizal fungi, including but not limited to Endomycorrhizae and Ectomycorrhizae, are known to be an important component of soil life. The mutualistic association between the fungi and the plant can be particularly helpful in improving plant survivability in nutrient-poor soils, plant resistance to diseases, e.g. microbial soil-borne pathogens, and plant resistance to contaminated soils, e.g. soils with high metal concentrations. Since mycorrhizal root systems significantly increase the absorbing area of plant roots, introducing mycorrhizal fungi may also reduce water and fertilizer requirements for plants.
[0310] Typically mycorrhizae are introduced into soil as a liquid formulation or as a solid in powder or granular form and contain dormant mycorrhizal spores and/or colonized root fragments. Often the most economic and efficient method is to treat the seeds themselves, but dealing with traditional liquid and powder inoculums to coat the seed can be difficult. In accordance with the present invention, inoculated biochar may be used to coat the seeds by, for example, using a starch binder on the seeds and then subjecting the seeds to inoculated biochar fines or powder. Another method is by placing the mycorrhizae inoculum in the soil near the seeding or established plant but is more costly and has delayed response as the plants initial roots form without a mycorrhizal system. This is because the dormant mycorrhizae are only activated when they come close enough to living roots which exude a signaling chemical. In addition if the phosphorus levels are high in the soil, e.g. greater than 70 ppm, the dormant mycorrhizae will not be activated until the phosphorus levels are reduced. Thus applying inoculant with or near fertilizers with readily available phosphorus levels can impede the desired mycorrhizal fungi growth. A third option is to dip plant roots into an inoculant solution prior to replanting, but this is costly as it is both labor and time intensive and only applicable to transplanting.
[0311] If the colonization of mycorrhizae can be quickened and the density of the mycorrhizae's hyphal network can be increased then the beneficial results of mycorrhizal root systems, e.g. increased growth, increased survivability, reduced water, and reduced fertilizer needs, can be realized sooner. Prior art shows that compost, compost teas, humates, and fish fertilizers can improve microbial activities and in more recent studies have shown physically combining arbuscular mycorrhizal fungi (AMF) inoculant with raw biochar has resulted in additional plant yield compared to each alone. See Hammer, et. al. Biochar Increases Arbuscular Mycorrhizal Plant Growth Enhancement and Ameliorates Salinity Stress, Applied Soil Ecology Vol 96, November 2015 (pg. 114-121).
[0312] An ideal carrier for the mycorrhizae would have moisture, air, a neutral pH, a surface for fungi to attach, and a space for the roots and fungi to meet. Thus a previously infused biochar created by the method disclosed above would be a better carrier than raw biochar alone. The infused biochar could be physically mixed with a solid mycorrhizal fungi inoculant or sprayed with a liquid mycorrhizal inoculant prior to or during application at seeding or to established plants. Additionally, the infused biochar and mycorrhizal fungi inoculant could be combined to form starter cubes, similar to Organo-Cubes, rockwool, oasis cubes, and peat pots.
[0313] The infused biochar could be further improved upon by initially infusing or further infusing the biochar with micronutrients for mycorrhizal fungi, for example but not limited to humic acid, molasses, or sugar. The growth nutrient infused biochar would further expedite the colonization of the mycorrhizal fungi when physically combined with the inoculant and applied to seeds or plants.
[0314] Another improvement to the infused biochar would be to initially infuse or further infuse the biochar with the signaling molecules of mycorrhizal fungi. The signaling molecule infused biochar would further expedite the colonization of the mycorrhizal fungi when physically combined with the inoculant and applied to seeds or plants, as it would bring the mycorrhizae out of dormancy quicker and thus establish the mycorrhizal root system quicker.
[0315] Another method for establishing and improving mycorrhizal fungi colonies would be by growing mycorrhizae into the infused biochar and then applying the mycorrhizal fungi inoculated biochar to seeds or plants. Similar to a sand culture (Ojala and Jarrell 1980 http://jhbiotech.com/docs/Mycorrhizae-Article.pdf), a bed of infused biochar is treated with a recycled inoculated nutrient solution by passing it through the bed multiple times.
[0316] I. Batch Treatment/Bulk Production
[0317] As demonstrated above, the treatment processes described above are particularly well suited for large scale production of biochar. The processes and biochars of the present invention provides a unique capability to select starting materials and pyrolysis techniques solely on the basis of obtaining a particular structure, e.g., pore size, density, pore volume, amount of open pores, interconnectivity, tortuosity, etc. Thus, these starting materials and processes can be selected without regard to adverse, harmful, phytotoxic side effects that may come from the materials and processes. This is possible, because the infiltration steps have the capability of mitigating, removing or otherwise address those adverse side effects. In this manner, a truly custom biochar can be made, with any adverse side effects of the material selection and pyrolysis process being mitigated in later processing steps.
[0318] Further, the processes are capable of treating a large, potentially variable, batch of biochar to provide the same, generally uniform, predetermined customized characteristics for which treatment was designed to achieve, e.g., pH adjustment. Treatment can result in treated biochar batches in which 50% to 70% to 80% to 99% of the biochar particles in the batch have same modified or customized characteristic, e.g., deleterious pore surface materials mitigated, pore surface modified to provide beneficial surface, pore volume containing beneficial additives.
[0319] Accordingly, the ability to produce large quantities of biochar having a high level of consistency, predictability and uniformity, provides numerous advantages in both large and small agricultural applications, among other things. For example, the ability to provide large quantities of biochar having predetermined and generally uniform properties will find applications in large scale agriculture applications. Thus, treated biochar batches from about 100 lbs up to 50,000+ lbs and between may have treated biochar particles with predetermined, uniform properties.
[0320] As the treated biochar batches are made up of individual biochar particles, when referring to uniformity of such batches it is understood that these batches are made up of tens and hundreds of thousands of particles. Uniformity is thus based upon a sampling and testing method that statistically establishes a level of certainty that the particles in the batch have the desired uniformity.
[0321] Thus, when referring to a treated batch of biochar as being "completely uniform" or having "complete uniformity" it means that at least about 99% of all particles in the batch have at least one or more property or feature that is the same. Same being within appropriately set tolerances for said property. When a treated batch of biochar is referred to as "substantially uniform" or having "substantial uniformity" it means that at least about 95% of all particles in the batch have at least one or more property or feature that is the same. When a treated batch of biochar is referred to as "essentially uniform" or having "essential uniformity" it means that at least about 80% of all particles in the batch have at least one or more property or feature that is the same. The batches can have less than 25%, 20% to 80%, and 80% or more particles in the batch that have at least one or more property or feature that is the same. Further, the batches can have less than 25%, 20% to 80%, and 80% or more particles in the batch that have at one, two, three, four, or all properties or features that are the same.
[0322] J. Applications
[0323] Generally, treated biochar of the present inventions can be used throughout the world, in numerous soil types, agricultural applications, horticultural, large and small scale farming, organic farming, and in a variety of soil management applications and systems, and combinations and variations of these. In fact, this particular solution provides the capability to custom-manufacture biochar for a particular climate, environment, geographical area, soil type, plant type, microbe type, or application by more precisely controlling key characteristics.
[0324] Examples of these applications include for example, use in acidic and highly weathered tropical field soils, use in temperate soils of higher fertility, use in large commercial applications, use for the production of large scale crops such as, soybean, corn, sugarcane and rice, in forestry applications, for golf courses (e.g., greens, fairways), for general purpose turf grasses, wine grapes, table grapes, raisin grapes, fruit and nut trees, ground fruits (e.g., strawberries, blueberries, blackberries), row crops (e.g., tomatoes, celery, lettuce, leafy greens), root crops (e.g., tubers, potatoes, beets, carrots), mushrooms, and combinations and variations of these.
[0325] Treated biochars and agriculture practices and methods, provide for improved soil structure, increased water retention capability, increased water holding ability of the soil over time, reduced runoff or leaching, increased holding ability for nutrients, increase holding of nutrients over time, and combinations and variations of these, and other features that relate to the increased holding and retention features and soil aggregation of the present biochars and processes. It further being understood that in addition to nutrients, other material additives, (e.g., herbicide, pesticide), can be utilized and benefit from the increased holding and retention capacities of the present biochars, systems, and methods.
[0326] Treated biochar may also be used in other applications, for example, such mixing with manure in holding ponds to potentially reduce gaseous nitrogen losses, soil remedial (for example absorption and capture of pesticide, contaminates, heavy metals, or other undesirable, disadvantageous soil components), ground water remediation, other bioremediations, storm water runoff remediation, mine remediation, mercury remediation and as a cattle or poultry feed additive.
[0327] Further, the present invention could be used to clean and/or infiltrate the pores of biochar with a variety of substances, for a number of purposes, including but not limited to, infiltrating the pores of biochar with nutrients, vitamins, microbes, drugs and/or other supplements, or a combination of any of the foregoing, for consumption by either humans and/or animals. The treated biochar may also be applied to animal pens, bedding, and/or other areas where animal waste is present to reduce odor and emission of unpleasant or undesirable vapors. Furthermore it may be applied to compost piles to reduce odor, emissions, and temperature to enable the use of the food waste and animal feed in composting. Biochar can also be applied to areas where fertilizer or pesticide runoff is occurring to slow or inhibit leaching and runoff. Additionally, it can be used in the public health sector to help control disease, for example carrying or deploying larvicides to reduce numbers of certain disease carrying or transmitting insects. The biochar may also be treated with additives which make it easier to dispense or apply, such as non-toxic oils, anti-clumping/binding additives, surface drying agents, or other materials.
[0328] In general, in the agricultural application of biochar to soil, the biochar should be located near the soil's surface in the root zone, or in or adjacent to the rhizosphere, where the bulk of nutrient cycling and uptake by plants takes place. Although benefits may be obtained from the application of biochar in layers above, below, in and combinations and variation of these, the root zone, for example during landscaping for carbon sequestration, or if using biochar for moisture management. Layering of biochar at various depths above, below, in and combinations and variation of these, the root zone, the surface, and combinations and variations of these, may also be employed. The biochar layers may have different predetermined properties for each layer, based upon, for example, the depth of the layer, soil type, geography, crop, climate and other factors.
[0329] The above are only a few examples of how additive infused or microbial carrying biochar may be produced for different uses. Those skilled in the art will recognize that there may be other mechanisms for infusing additives into the pores of the biochar without departing from the scope of the invention. Those skilled in the art will further recognize that the present invention can be used on any type of soil application, including, but not limited to, the following: crops, turf grasses, potted plants, flowering plants, annuals, perennials, evergreens and seedlings. By way of example, treated biochar may be incorporated into or around the root zone of a plant. As most trees, rows, and specialty crops extract large percentage of their water from the first twenty-four inches below the soil surface, the above applications will generally be effective incorporating the biochar around the root zone from the top surface of the soil and up to a depth of 24'' below the top surface of the soil, depending on the plant type and species, or alternatively, within a 24'' radius surrounding the roots regardless of root depth or proximity from the top surface of the soil. When the plant roots are closer to the surface, the incorporation of the biochar within the top 2-6'' inches of the soil surface may also be effective. Greater depths are more beneficial for plants having larger root zones, such as trees.
[0330] In certain examples of biochar applications, the treated biochar can be applied in amounts (e.g., rates of addition as measured by weight of treated biochar per area of field) of from about 0.001 ton of treated biochar per acre to about 150 tons of treated biochar per acre, from about 2.5 tons of treated biochar per acre to about 100 tons of treated biochar per acre, and from about 5 tons of treated biochar per acre to about 70 tons of treated biochar per acre, although larger and smaller amounts may be used. Additional rates of from about 1/2 tons of treated biochar to about 10 tons of treated biochar may be used. For example, application rates of 1 ton of treated biochar was added per acre to a soil for a lettuce crop where the soil had a pH of about 7. In another example, about 3 tons per acre of treated biochar was added to soil for a strawberry crop. In these examples, the plants showed enhanced growth rates and yields.
[0331] Generally, for conventional field cropping systems, biochar can be preferably added using existing farm equipment and incorporated into existing farming operations. For example, treated biochar can be applied and incorporated together with lime, since lime is often applied as a fine solid, which must be well incorporated into soil. However, it is also contemplated that the examples of the present inventions may give rise to new equipment and utilizations based upon the features, performance and capabilities of the present inventions. Generally, treated biochar may be applied to fields, by way of example through the use of manure or compost spreaders, lime spreaders, plowing method (e.g., from hand hoes, animal draft plows, disc harrows, chisels, rotary hoes, etc.), large scale tillage equipment, including rotary tillers, mulch finishers, draw offset discs, and disc harrows (such as for example JOHN DEERE DH51, DH52F, PC10, RT22, and RC22). Treated biochar may also be applied by modified large scale nutrient applicators (such as, for example, JOHN DEERE 2410C, 2510H, 25105 Strip-Till Medium Residue Applicator), large scale draw dry spreaders (such as JOHN DEERE DN345), large scale no-till planters, large scale dry fertilizer sub-surface applicators, and liquid slurry surface or subsurface applicators. Similar, and various other types of large farming, and earth moving and manipulation equipment may be used to apply the treated biochar to the field, such as for example, drop spreaders or drills.
[0332] For example, treated biochar may be applied using banding techniques, which is an operation involving applying the biochar in a narrow band, using equipment that cuts the soil open, without disturbing the entire soil surface. Using this technique the biochar can be placed inside the soil while minimizing soil disturbance, making it possible to apply biochar after crop establishment, among other applications.
[0333] In other examples, treated biochar may be mixed with other soil amendments, or other materials, such as for example manure, sand, topsoil, compost, turf grass substrate, peat, peat moss, or lime before soil application, which are already scheduled or part of the existing operations, and in this manner by combining these steps (e.g., biochar application with existing application step) can improve efficiency by reducing the number of field operations required. In other examples, treated biochar can also be mixed with liquid, (e.g., liquid manures) and applied as a slurry. Finer biochar particles may be preferably used with this type of slurry application using existing application equipment, and dust problems associated with these finer particles may be mitigated, managed or eliminated.
[0334] In further examples, treated biochar can be top dressed on perennial pastures or other perennial vegetation, such as spaces between fruit trees in orchards. Treated biochar may also be applied with individual plants while transplanting or mixed with topsoil and other amendments while preparing raised beds. In forestry or similar operations where replanting of seedlings takes place, treated biochar can be applied by broadcasting (e.g., surface application) or incorporation over the entire planting area, it can be added in the planting holes, and combinations and variations of these. Before or after tree establishment, biochar could also be applied by traditional and subsurface banding or top-dressed over perennial vegetation in orchards, but care should be taken to minimize root damage and soil compaction.
[0335] In other examples of applications, treated biochar can be applied in trenches radiating out from the base of established trees ("radial trenching") or in holes dug at some distance from the base of the tree ("vertical mulching"); biochar could also potentially be applied to soil using "air excavation tools". These tools use pressurized air to deliver material, e.g., compost, under the soil surface and reduce compaction. Alternatively, the soil around tree roots can be excavated and treated biochar applied before covering with soil.
[0336] While, in some examples, particle size distribution of treated biochar materials may vary widely depending on the feedstock and the pyrolysis technique used to produce the biochar, uniformity if required or preferred, can be achieved by various milling and grinding techniques that may be employed during processing or during the distribution and application to soil. When smaller particles are utilized, and in particular for surface applications, care should be taken to apply the treated biochar in ways that minimize loss due to wind or water erosion.
[0337] As set forth above, the treated biochar of the present invention may be used in various agriculture activities, and the fields of edaphology and pedology, as well as other activities and in other fields. Additionally, the treated biochar may be used, for example, with: farming systems and technologies, operations or activities that may be developed in the future; and with such existing systems, operations or activities which may be modified, in-part, based on the teachings of this specification. Further, the various treated biochar and treatment processes set forth in this specification may be used with each other in different and various combinations. Thus, for example, the processes and resulting biochar compositions provided in the various examples provided in this specification may be used with each other; and the scope of protection afforded the present inventions should not be limited to any particular example, process, configuration, application or arrangement that is set forth in a particular example or figure.
[0338] Although this specification focuses on agriculture, soil modification and plant growth, it should be understood that the materials, compositions, structures, apparatus, methods, and systems, taught and disclosed herein, may have applications and uses for many other activities in addition to agriculture for example, as filters, additives, and in remediation activities, among other things.
[0339] It being understood that one or more of these may be preferred for one application, and another of these may be preferred for a different application. Thus, these are only a general list of preferred features and are not required, necessary and may not be preferred in all applications and uses.
[0340] It is noted that there is no requirement to provide or address the theory underlying the novel and groundbreaking functionality, performance or other beneficial features and properties that are the subject of, or associated with, embodiments of the present inventions. Nevertheless, to the extent that various theories are provided in this specification to further advance the art in this important area. These theories put forth in this specification, and unless expressly stated otherwise, in no way limit, restrict or narrow the scope of protection to be afforded the claimed inventions. These theories many not be required or practiced to utilize the present inventions. It is further understood that the present inventions may lead to new, and heretofore unknown theories to explain the functionality, performance or other beneficial features and properties that are the subject of, or associated with, embodiments of the methods, articles, materials, and devices of the present inventions; and such later developed theories shall not limit the scope of protection afforded the present inventions.
[0341] Those skilled in the art will recognize that there are other methods that may be used to treat biochar in a manner that forces the infusion of liquids into the pores of the biochar without departing from the scope of the invention. The foregoing description of implementations has been presented for purposes of illustration and description. It is not exhaustive and does not limit the claimed inventions to the precise form disclosed. Modifications and variations are possible in light of the above description or may be acquired from practicing the invention. The claims and their equivalents define the scope of the invention.
* * * * *
References
D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

D00013

D00014

D00015

D00016

D00017

D00018

D00019

D00020

D00021

D00022

D00023
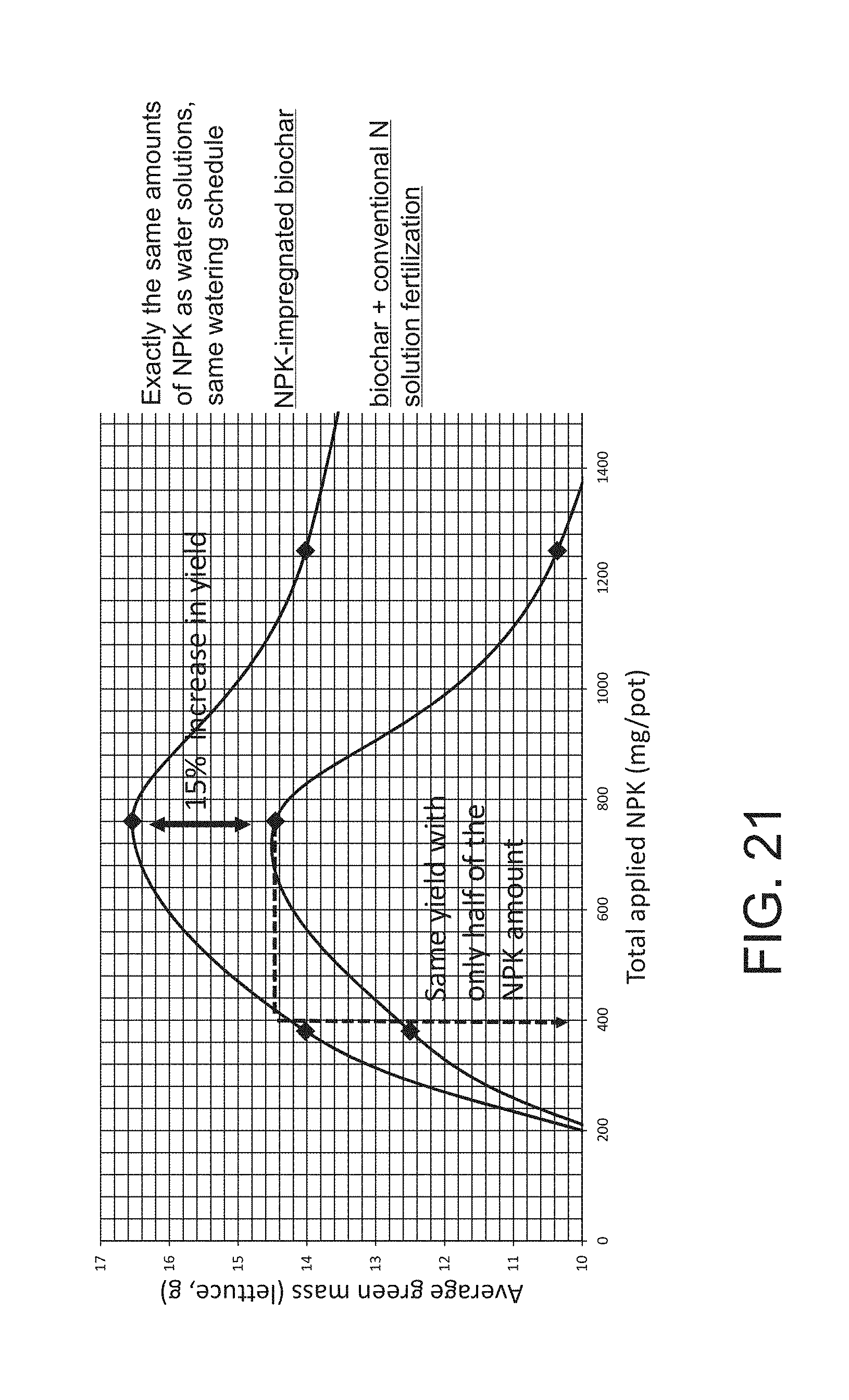
D00024

D00025

D00026

D00027

D00028

D00029

D00030

D00031

D00032

D00033

D00034

D00035

D00036

D00037


XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.