Anti-death Receptor Antibodies And Methods Of Use Thereof
BEURSKENS; Frank ; et al.
U.S. patent application number 15/780285 was filed with the patent office on 2019-07-04 for anti-death receptor antibodies and methods of use thereof. The applicant listed for this patent is GENMAB B.V.. Invention is credited to Frank BEURSKENS, Rob DE JONG, Marije OVERDIJK, Paul PARREN, David SATIJN, Janine SCHUURMAN, Kristin STRUMANE.
| Application Number | 20190202926 15/780285 |
| Document ID | / |
| Family ID | 58796495 |
| Filed Date | 2019-07-04 |


View All Diagrams
| United States Patent Application | 20190202926 |
| Kind Code | A1 |
| BEURSKENS; Frank ; et al. | July 4, 2019 |
ANTI-DEATH RECEPTOR ANTIBODIES AND METHODS OF USE THEREOF
Abstract
The present invention relates to monospecific or bispecific antibody molecules that specifically bind antigens of Death Receptors, which are members of the tumor necrosis factor (TNF) receptor Superfamily (TNFR-SF) with an intracellular death domain. The invention relates in particular to antibody molecules of the IgG1 isotype having a mutation in the Fc region that enhances clustering of IgG molecules after target binding. The invention further relates to a combination of antibody molecules binding different epitopes on one or more specific Death Receptors. The invention also relates to pharmaceutical compositions containing these molecules and the treatment of cancer using these compositions.
| Inventors: | BEURSKENS; Frank; (Utrecht, NL) ; OVERDIJK; Marije; (Utrecht, NL) ; DE JONG; Rob; (Utrecht, NL) ; SATIJN; David; (Utrecht, NL) ; STRUMANE; Kristin; (Werkhoven, NL) ; SCHUURMAN; Janine; (Diemen, NL) ; PARREN; Paul; (Odijk, NL) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 58796495 | ||||||||||
| Appl. No.: | 15/780285 | ||||||||||
| Filed: | December 1, 2016 | ||||||||||
| PCT Filed: | December 1, 2016 | ||||||||||
| PCT NO: | PCT/EP2016/079517 | ||||||||||
| 371 Date: | May 31, 2018 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C07K 16/46 20130101; A61K 2039/507 20130101; A61K 38/00 20130101; A61K 2039/505 20130101; C07K 2317/31 20130101; C07K 2317/526 20130101; C07K 2317/73 20130101; C07K 16/2878 20130101; A61P 35/00 20180101; C07K 2317/75 20130101; C12N 15/62 20130101; C07K 2317/24 20130101; C07K 16/30 20130101; C07K 2317/52 20130101 |
| International Class: | C07K 16/28 20060101 C07K016/28; A61P 35/00 20060101 A61P035/00; C07K 16/30 20060101 C07K016/30 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Dec 1, 2015 | DK | PA 2015 00771 |
| Dec 7, 2015 | DK | PA 2015 00787 |
| Dec 7, 2015 | DK | PA 2015 00788 |
| Nov 10, 2016 | DK | PA 2016 00701 |
| Nov 10, 2016 | DK | PA 2016 00702 |
Claims
1. An antibody comprising an Fc region of a human immunoglobulin IgG and an antigen binding region binding to a death receptor comprising an intracellular death domain, wherein the Fc region comprises a mutation at an amino acid position corresponding to position E430, E345, S440, and/or Y436 in human IgG1, wherein the positions are numbered according to EU Index.
2-4. (canceled)
5. The antibody according to claim 1, wherein the Fc region comprises a mutation selected from the group consisting of: E430G, E345K, E430S, E430F, E430T, E345Q, E345R, E345Y, S440W, S440Y, and Y436I.
6-8. (canceled)
9. The antibody according to claim 1, wherein the Fc region comprises a further mutation in an amino acid position corresponding to K439.
10. The antibody according to claim 1, wherein the Fc region comprises a mutation at an amino acid position corresponding to E430 and/or E345 in a human IgG1, and wherein said Fc region comprises a further mutation at an amino acid position corresponding to S440, with the proviso that the mutation is not S440Y or S440W.
11. The antibody according to claim 9, wherein the further mutation is selected from the group consisting of: K439E, K439D.
12. The antibody according to claim 10, wherein the further mutation is selected from the group consisting of: S440K, S440R and S440H.
13. The antibody according to claim 1, wherein the antibody further comprises a mutation selected from K439E or S440K.
14. The antibody according to claim 1, wherein the death receptor comprising an intracellular death domain is selected from the group consisting of: FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR, and NGFR.
15-16. (canceled)
17. The antibody according to claim 1, wherein the antibody is an IgG1, IgG2, IgG3, IgG4, IgE, IgD or IgM isotype.
18-19. (canceled)
20. The antibody according to claim 1, wherein the antibody is a monoclonal antibody.
21. The antibody according to claim 1, wherein the antibody is human, humanized or chimeric.
22. The antibody according to claim 1, wherein the antibody is agonistic.
23. The antibody according to claim 1, wherein the antibody induces programmed cell death in a target cell.
24-25. (canceled)
26. A multispecific antibody comprising one or more antigen binding regions according to claim 1.
27. The multispecific antibody according to claim 26, wherein said multispecific antibody is a bispecific antibody.
28. The bispecific antibody according to claim 27, wherein said first antigen binding region and said second antigen binding region binds different epitopes on one or more members of a death receptor comprising an intracellular death domain selected from the group consisting of: FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR and NGFR.
29. (canceled)
30. A composition comprising at least one antibody according to claim 1 and a carrier.
31. (canceled)
32. A composition comprising a first antibody and a second antibody, wherein both the first and second antibodies are according to claim 1.
33. The composition according to claim 30, which comprises i) a first antibody, wherein the Fc region comprises a first mutation at an amino acid position corresponding to E430 or E345 in human IgG1, and a further mutation at an amino acid position corresponding to K439 in human IgG1, EU numbering, and ii) a second antibody, wherein the Fc region comprises a first mutation at an amino acid position corresponding to E430 or E345 in human IgG1, EU numbering, and a further mutation at an amino acid position corresponding to S440 in human IgG1.
34. The composition according to claim 33, which comprises a first antibody wherein the further mutation is selected from the group of: K439E and K439D, and a second antibody wherein the further mutation is selected from the group of S440K, S440R or S440H.
35. (canceled)
36. The composition according to claim 32, wherein said first antibody and said second antibody bind different epitopes on one or more members of a death receptor comprising an intracellular death domain selected form the group consisting of: such as FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR and NGFR.
37-45. (canceled)
46. A method of treating an infectious disease, autoimmune disease, or cardiovascular anomalies comprising administering to a subject in need thereof an effective amount of the composition of claim 30.
47. A method of treating a solid tumor and/or hematological tumor comprising administering to a subject in need thereof an effective amount of the composition of claim 30.
48. The method of claim 47, wherein the solid tumor is selected from the group consisting of colorectal cancer, bladder cancer, osteosarcoma, chondrosarcoma, breast cancer, cancers of the central nervous system, cervical cancer, endometrium cancer, gastric cancer, head and neck cancer, kidney cancer, liver cancer, lung cancer, ovarian cancer, pancreatic cancer, sarcoma, and skin cancer; and the hematological tumor is selected from the group consisting of leukemia, lymphoma, and multiple myeloma.
49. (canceled)
50. A method of inhibiting growth of FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR expressing tumors comprising administering to a subject in need thereof an effective amount of the composition of claim 30.
51. A method of inducing apoptosis in FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR expressing tumors comprising administering to a subject in need thereof an effective amount of the composition of claim 30.
52. A method of treating an individual having a cancer comprising administering to said individual an effective amount of the antibody of claim 1.
53. The method according to claim 52 further comprising administering an additional therapeutic agent.
54. The method according to claim 53, wherein the additional therapeutic agent is one or more anti-cancer agent(s) selected from the group consisting of of chemotherapeutics, kinase inhibitors, apoptosis-modulating agents, RAS inhibitors, proteasome inhibitors, histone deacetylase inhibitors, antibodies or antibody mimetics, antibody-drug conjugates.
55. A kit comprising the antibody of claim 1, and instructions for use.
56-57. (canceled)
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application is a 35 U.S.C. 371 national stage filing of International Application No. PCT/EP2016/079517, filed Dec. 1, 2016, which claims priority to Danish Patent Application Nos. PA 2015 00771, filed Dec. 1, 2015, PA 2015 00787, filed Dec. 7, 2015, PA 2015 00788, filed Dec. 7, 2015, PA 2016 00701, filed Nov. 10, 2016, and PA 2016 00702, filed Nov. 10, 2016. The contents of the aforementioned applications are hereby incorporated by reference.
SEQUENCE LISTING
[0002] The instant application contains a Sequence Listing which has been submitted electronically in ASCII format and is hereby incorporated by reference in its entirety. Said ASCII copy, created on Jan. 29, 2019, is named GMI_169BUS_Sequence_Listing.txt and is 72,344 bytes in size.
FIELD OF THE INVENTION
[0003] The present invention relates to monospecific or bispecific antibodies that specifically bind antigens of Death Receptors, which are members of the tumor necrosis factor (TNF) receptor Superfamily (TNFR-SF) with an intracellular death domain. The invention relates in particular to antibody molecules of the IgG1 isotype having a mutation in the Fc region that enhances clustering of IgG molecules after target binding. The invention further relates to a combination of antibody molecules binding different epitopes on one or more specific Death Receptors. The invention also relates to pharmaceutical compositions containing these molecules and the treatment of cancer using these compositions.
BACKGROUND OF THE INVENTION
[0004] The Death Receptors (DR) are a subset of the TNFR-SF, which are plasma membrane receptors characterized by a cytoplasmic sequence of .sup..about.80 amino acids known as the death domain (DD) (Nagata et al., Cell. 1997 Feb. 7; 88(3):355-65; Ashkenzai et al., Science. 1998 Aug. 28; 281(5381):1305-8; Locksley et al., Cell. 2001 Feb. 23; 104(4):487-501; Wajant Cell Death Differ. 2015 November; 22(11):1727-41). The intracellular death-domain of the tumor necrosis factor (TNF) receptor superfamily (TNFR-SF) are known to activate two main signalling cascades: a kinase cascade leading to NF-kappaB and JNK activation and a caspase cascade leading to cell death (Ashkenazi et al., Science. 1998 Aug. 28; 281(5381):1305-8). Ligand-mediated activation of death receptors has been shown to trigger apoptosis in a variety of transformed cell lines. Accordingly, there has been considerable efforts to develop death receptor-targeting therapeutics for various diseases, including agonistic antibodies. However, these efforts only resulted in limited clinical efficacy.
[0005] Consequently, there is a need for providing improved antibodies binding to death receptors of the tumor necrosis factor (TNF) receptor Superfamily (TNFR-SF) with an intracellular death domain, such as improved anti-Death Receptor antibodies for the treatment of cancer, of infectious disease, autoimmune disease, cardiovascular anomalies and other diseases
SUMMARY OF THE INVENTION
[0006] Surprisingly the inventors of the present invention have found that the introduction of a specific point mutation in the Fc region of antibodies that specifically bind antigens of Death Receptors, which are members of the TNFR-SF comprising an intracellular death domain significantly enhances the potency of the antibody in vitro and in vivo by Fc.gamma.R-independent clustering after binding of the antibody to the target on the cell surface. Even more surprisingly the inventors have also found that a combination of two anti-Death Receptor antibodies with a mutations in the Fc region facilitate antibody clustering conditional on cell surface antigen binding, resulting in the formation of hetrohexamers and enhanced potency compared to a combination of the two anti-Death Receptor antibodies without the mutation.
[0007] The object of the present invention is to provide improved anti-Death Receptor antibodies, such as anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR, and anti-NGFR antibodies, e.g. for use in the treatment of cancer. Such an improved anti-Death Receptor antibody comprises a mutation in the Fc domain. A further object of the present invention is to provide an improved composition for the treatment of cancer comprising one or more anti-Death Receptor antibodies binding to different epitopes on Death Receptors, such as anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR, and anti-NGFR antibodies. Such an improved composition as described herein comprises at least one anti-Death Receptor antibody or the composition comprises two anti-Death Receptor antibodies binding to different regions on one or more Death Receptors, such as different epitopes on one or more of the following Death Receptors selected from the group consisting of: FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR, and NGFR.
[0008] The present invention provides an antibody comprising an Fc region of a human immunoglobulin IgG and an antigen binding region binding to a Death Receptor, such as an anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR, and anti-NGFR antibody, wherein the Fc region comprises a mutation at an amino acid corresponding to position E430, E345 or S440 in human IgG1 according to EU numbering (Edelman et al., Proc Natl Acad Sci USA. 1969 May; 63(1):78-85; Kabat et al., Sequences of Proteins of Immunological Interest, Fifth Edition. 1991 NIH Publication No. 91-3242). Unless contradicted by the context immunoglobulin IgG has the same meaning as IgG.
[0009] In one aspect the invention provides an antibody comprising an Fc region of a human immunoglobulin IgG and an antigen binding region binding to a Death Receptor comprising an intracellular death domain, wherein the Fc region comprises a mutation at an amino acid corresponding to position E430, E345 or S440 in human IgG1, EU numbering.
[0010] That is, the inventors of the present invention in a first aspect of the invention found that anti-Death Receptor antibodies, such as anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR, and anti-NGFR antibodies of the invention increase apoptosis of cells expressing FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR, or NGFR, such as tumor cells compared to anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR, and anti-NGFR antibodies without a mutation at an amino acid position corresponding to E430, E345 or 5440 of human IgG1, EU numbering. That is, an anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR, and anti-NGFR antibody of the present invention is suitable for the treatment of FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR, or NGFR positive or expressing tumors. Thus the antibodies according to the invention are suitable for treatment of tumors which are positive for or expressing one or more antigens consisting of the following group: FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR and NGFR.
[0011] In one embodiment of the present invention the anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR, and anti-NGFR antibody comprises an Fc region of a human immunoglobulin IgG and an antigen binding region binding to a Death Receptor, wherein the Fc region comprises a mutation corresponding to position E430G or E345K in human IgG1 according to EU numbering. Thus in one embodiment of the invention the anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR, and anti-NGFR antibody comprises an Fc region of a human immunoglobulin IgG and an antigen binding region binding to a Death Receptor, wherein the Fc region comprises a mutation at an E430G or E345K mutation.
[0012] In one embodiment of the present invention the anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR, and anti-NGFR antibody comprises an Fc region of a human immunoglobulin IgG, wherein the Fc region comprises an E430G mutation.
[0013] In one embodiment of the present invention the anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR, and anti-NGFR antibody comprises an Fc region of a human immunoglobulin IgG, wherein the Fc region comprises an E345K mutation.
[0014] In one aspect the invention provides a composition comprising one or more anti-Death Receptor antibodies selected from the group consisting of: anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR, and anti-NGFR. In one embodiment the composition comprises one or more antibodies binding to different epitopes on FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR and NGFR. In one embodiment the composition comprises at least a first and a second antibody selected from the group consisting of: anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR and anti-NGFR, wherein the first antibody does not block antigen binding of the second antibody.
[0015] In another aspect the invention provides a bispecific antibody comprising one or more antigen binding regions binding to FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR and NGFR. In one embodiment a bispecific antibody of the present invention comprises a first and a second heavy chain, wherein the first heavy chain comprises a F405L mutation and a second heavy chain comprises a K409R mutation, or vice versa. Thus in one embodiment a bispecific antibody according to the present invention the bispecific antibody comprises a first and a second heavy chain, wherein the first and the second heavy chain comprises a mutation at an amino acid positon corresponding to E430, E345 or S440 in human IgG1, EU numbering and wherein the first heavy chain comprises a F405L mutation and the second heavy chain comprises a K409R mutation. Thus in one embodiment a bispecific antibody according to the present invention the bispecific antibody comprises a first and a second heavy chain, wherein the first and the second heavy chain comprises a mutation at an amino acid position corresponding to E430, E345 or S440 in human IgG1, EU numbering and wherein the first heavy chain comprises a K409R mutation and the second heavy chain comprises a F405L mutation.
[0016] In yet another aspect the invention provides a method of treating a disease comprising administering to an individual in need thereof an effective amount of an antibody or composition as described herein. In one embodiment of the invention the disease is cancer.
[0017] In another aspect of the invention the anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR, or anti-NGFR antibody, bispecific antibody or composition according to the present invention is for use as a medicament. In one embodiment the anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR, or anti-NGFR antibody, bispecific antibody or composition is for use in treatment of a disease. In one embodiment the disease is a cancer or a tumor.
[0018] In yet another aspect the invention provides a method of treating an individual having a cancer comprising administering to said individual an effective amount of said antibody or composition as described herein.
[0019] In another aspect the invention provides a kit of parts comprising an antibody or composition according to any one of the preceding claims, wherein said antibody or composition is in one or more containers such as a vial.
[0020] In another aspect the invention provides the use of an antibody or a composition as described herein for the manufacture of a medicament for treatment of a disease. In one embodiment the invention provides the use of an antibody or a composition as described herein for the manufacture of a medicament for treatment of cancer.
[0021] The antibodies and compositions described herein are directed against or specific for human FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR. The antibodies and compositions described cross-react with rhesus and cynomolgus monkey FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR. In particular, in one embodiment the antibodies and compositions bind specifically to the extracellular domain of FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR. In one particular embodiment the antibodies and compositions bind to the same Death Receptor from the group consisting of: FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR and NGFR e.g. at non-overlapping epitopes. That is a first antibody described herein does not block binding of a second antibody described herein. In one particular embodiment a composition described herein comprises a first and a second antibody binding to FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR and the first antibody does not block binding of the second antibody to FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR.
[0022] The antibodies and compositions of the present invention can generally be used to modulate the activity of a Death Receptor such as FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR. In one embodiment the antibody or composition may trigger, activate and/or increase or enhance the signalling that is mediated by a Death Receptor such as FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR. In one embodiment the antibody or composition may have an agonistic effect on a Death Receptor such as FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR and in particular trigger or increase the biological mechanisms, responses and effects associated with FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR, their signalling and/or the pathway in which FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR is involved. That is antibodies or compositions of the present invention may induce apoptosis or cell death in cells or tissues expressing FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR, such as cancer cells or tumor cells.
[0023] In one embodiment the antibodies or compositions described herein induce, trigger, increase or enhance apoptosis, cell death or growth arrest in cells or tissues expressing FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR, such as cancer cells or tumor cells. In one embodiment the antibodies or compositions described herein are capable of binding to FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR on a cell surface, and in particular of binding to FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR in such a way that the signalling mediated by FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR is induced, triggered, increased or enhanced. In one embodiment the antibodies or compositions described herein may be such that they are capable of binding to a FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR in such a way that apoptosis or cell death is induced in cancer cells or tumor cells expressing FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR.
[0024] In one embodiment the antibodies or compositions of the present invention induce, trigger, increase or enhance apoptosis or cell death in cancer cells or tumor cells expressing FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR. The increased or enhanced apoptosis or cell death may be measured by an increase or enhanced level of phosphatidylserine exposure on cells exposed to or treated with one or more anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR, and anti-NGFR antibodies of the invention. Alternatively, the increase or enhanced apoptosis or cell death may be measured by measuring activation of caspase 3 or caspase 7 in cells that have been exposed to or treated with one or more anti-DR5 antibodies of the invention. Alternatively, the increase or enhanced apoptosis or cell death may be measured by a loss of viability in cell cultures that have been exposed to or treated with one or more anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR, and anti-NGFR antibodies of the invention, compared to untreated cell cultures. Induction of caspase-mediated apoptosis may be assessed by demonstrating inhibition of the loss of viability after exposure to DR5 antibody by a caspase-inhibitor, for example ZVAD.
BRIEF DESCRIPTION OF THE DRAWINGS
[0025] FIG. 1 shows an amino acid alignment of the four different human IgG1 Fc allotypes identified thus far. The Fc sequence of the IgG1m(f) (SEQ ID NO 1), IgG1m(z) (SEQ ID NO 2), IgG1m(a) (SEQ ID NO 3), IgG1m(x) (SEQ ID NO 4).
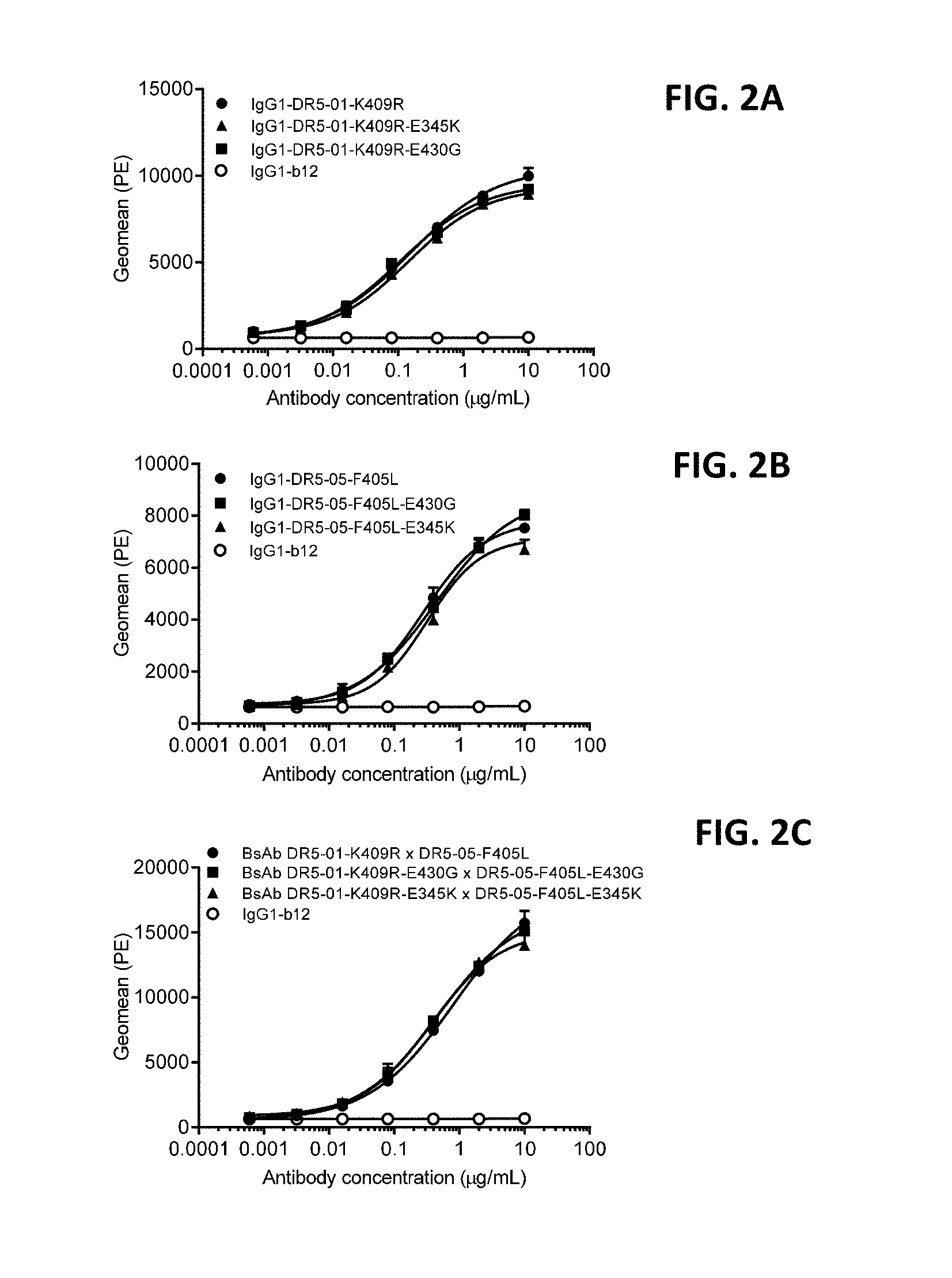
[0026] FIGS. 2A-2C show binding of DR5 antibodies with and without hexamerization-enhancing mutation E430G or E345K to DR5-positive COLO 205 cells. Variants of the human-mouse chimeric antibodies (FIG. 2A) IgG1-DR5-01, (FIG. 2B) IgG1-DR5-05 and (FIG. 2C) and bispecific antibody IgG1-DR5-01-K409R.times.IgG1-DR5-05-F405L (BsAb DR5-01-K409R.times.DR5-05-F405L) were tested in FACS analysis for binding to COLO 205 cells. Binding is expressed as Geometric mean of fluorescence intensity. Anti-gp120 antibody IgG1-b12 was used as negative control. Error bars indicate the standard deviation.
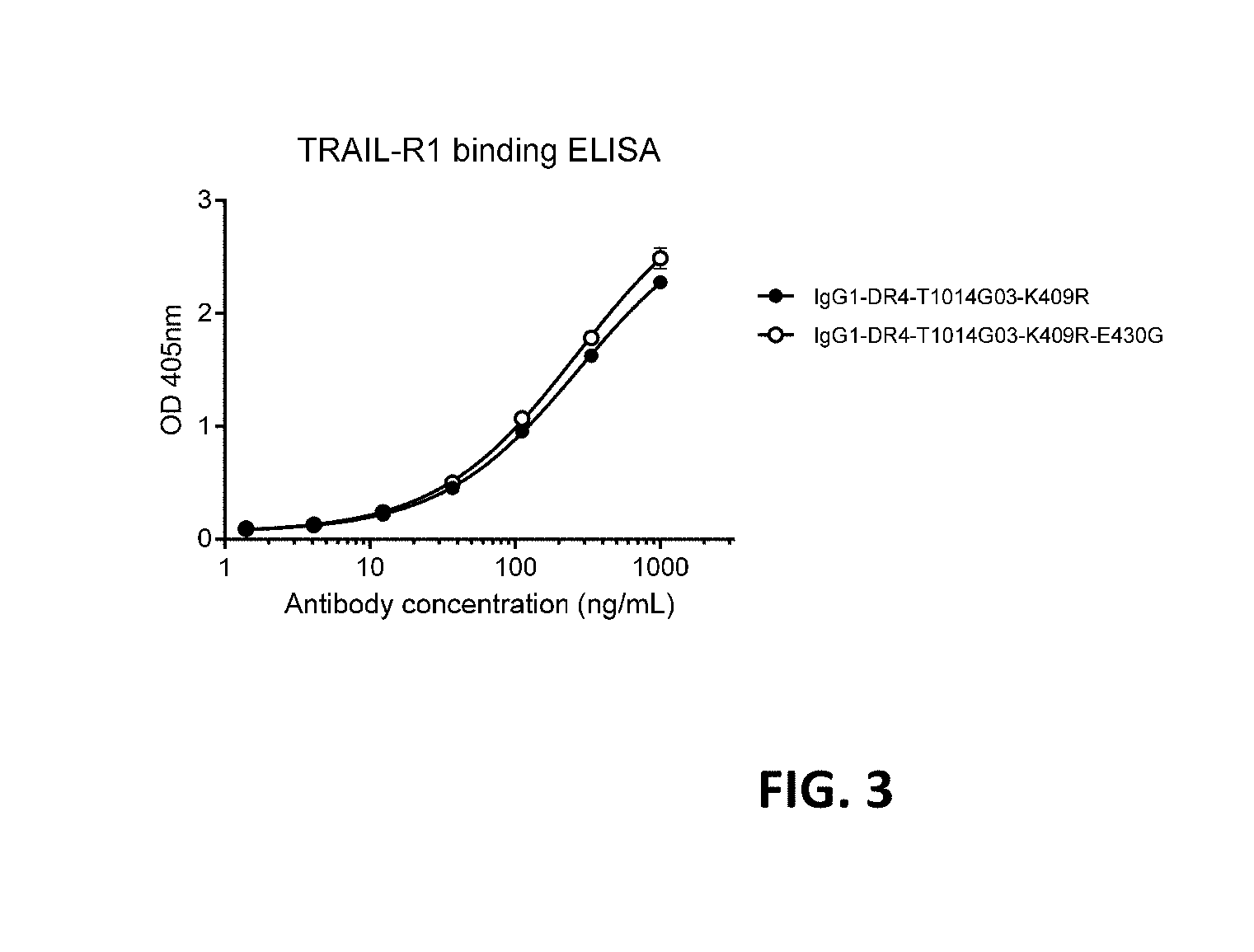
[0027] FIG. 3 shows a binding ELISA of DR4 antibodies to coated sTRAIL-R1. Graphs represent binding of the antibody IgG1-DR4-T1014G03-K409R with and without the E430G hexamerization-enhancing mutation to coated sTRAIL-R1.
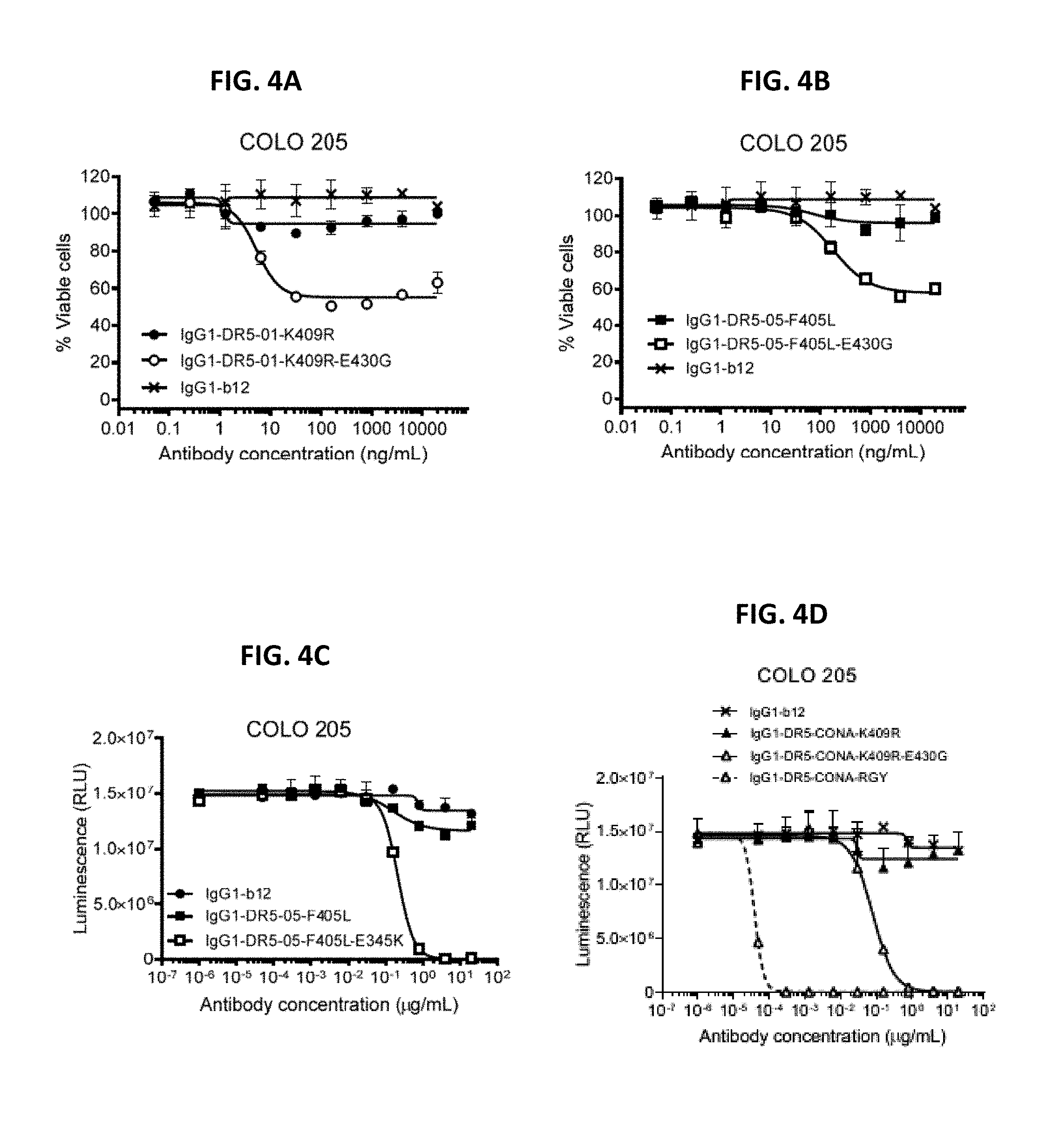
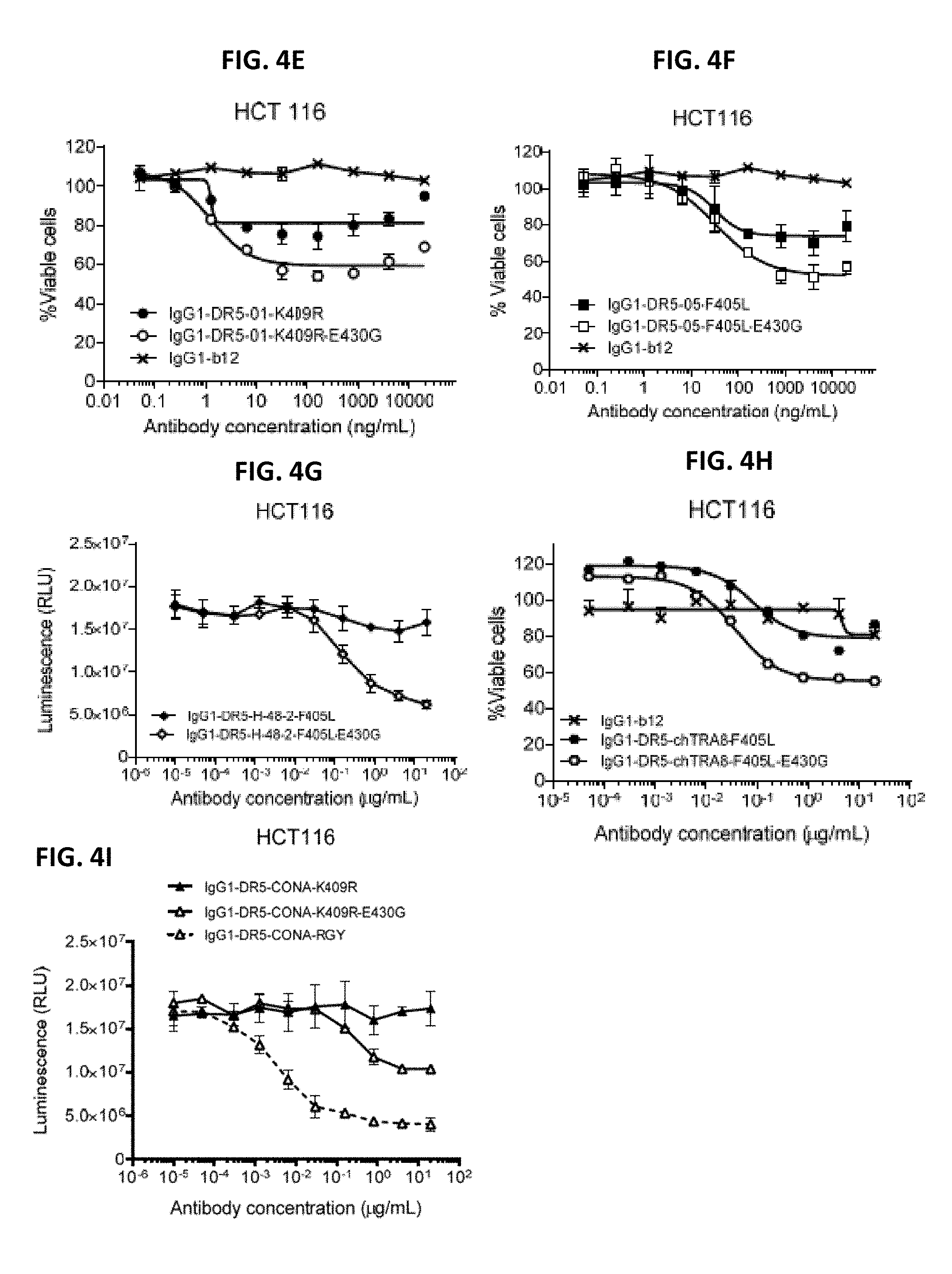
[0028] FIGS. 4A-4I show a viability assays with variants of different DR5 antibodies. Introduction of the E345K (FIG. 4C), E430G (FIGS. 4A, 4B, and 4D-4I) or E345R/E430G/S440Y (RGY) (FIG. 4D, FIG. 4I) hexamerization-enhancing mutations resulted in enhanced killing for the different DR5 antibodies on COLO 205 (FIGS. 4A-4D) and HCT116 (FIGS. 4E-4I) colon cancer cells. Error bars indicate standard deviation. Data are presented as Luminescence (RLU=relative luminescence units) or as % Viable cells calculated from the luminescence relative to samples incubated without antibody (no kill) and samples incubated with Staurosporine (maximal kill).
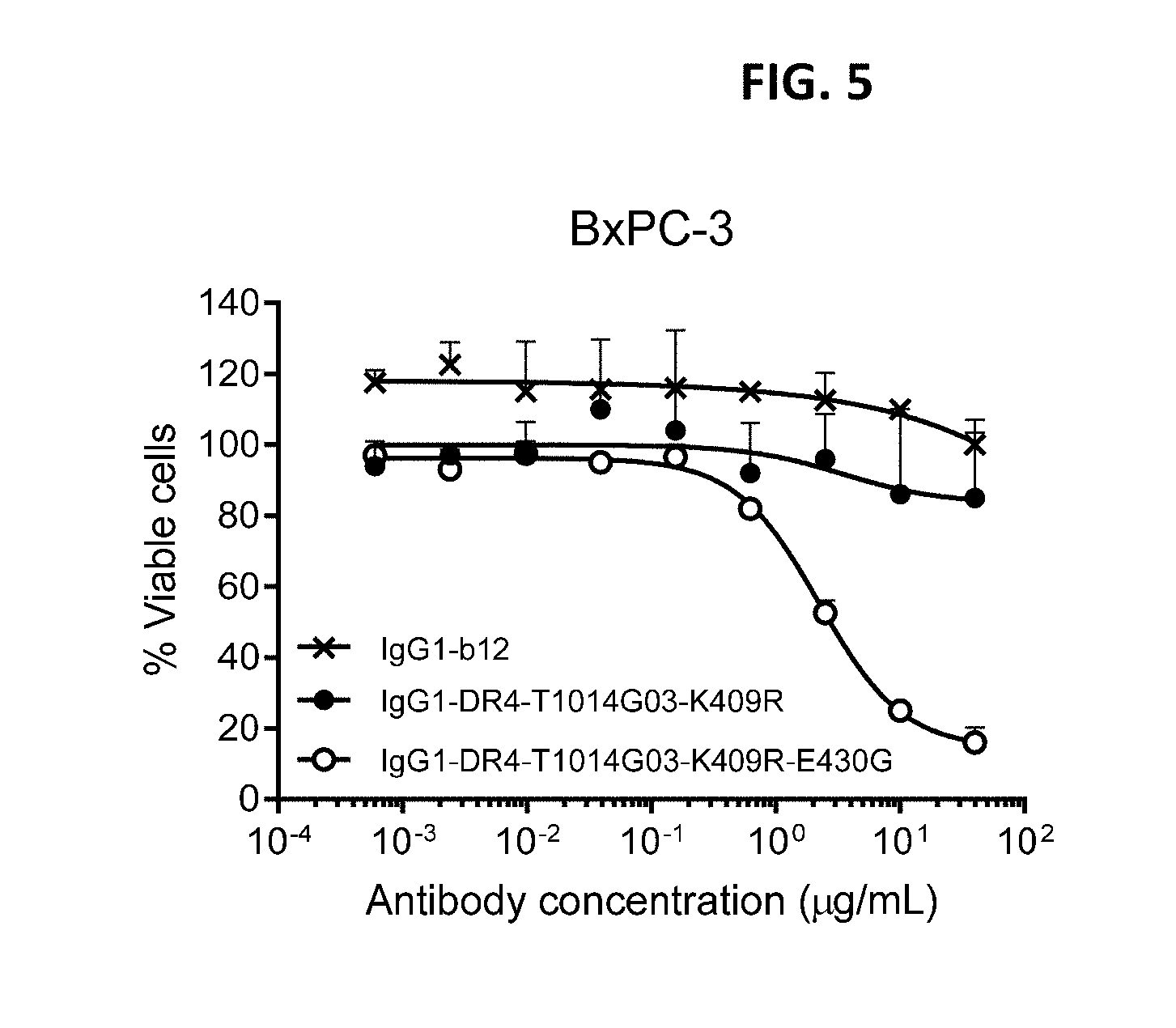
[0029] FIG. 5 shows a viability assay with variants of DR4 antibody IgG1-DR4-T1014G03. Introduction of the E430G hexamerization-enhancing mutation resulted in enhanced killing of BxPC-3 human pancreatic cancer cells. Error bars indicate standard deviation.
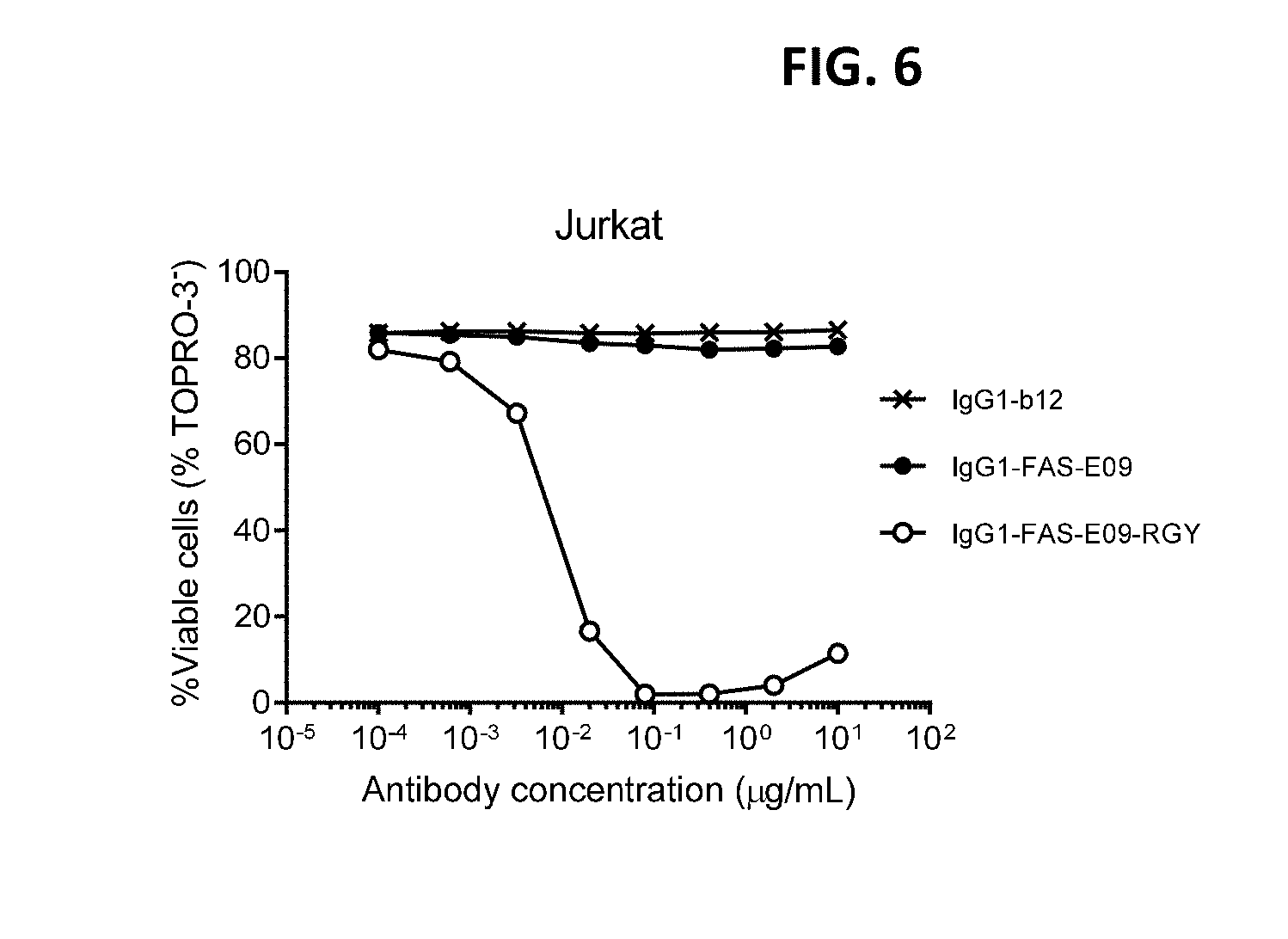
[0030] FIG. 6 shows a viability assay with variants of FAS antibody IgG1-FAS-E09. Introduction of the hexamerization-enhancing triple mutation E345R/E430G/S440Y (RGY) resulted in dose-dependent killing of Jurkat human T lymphocytes.
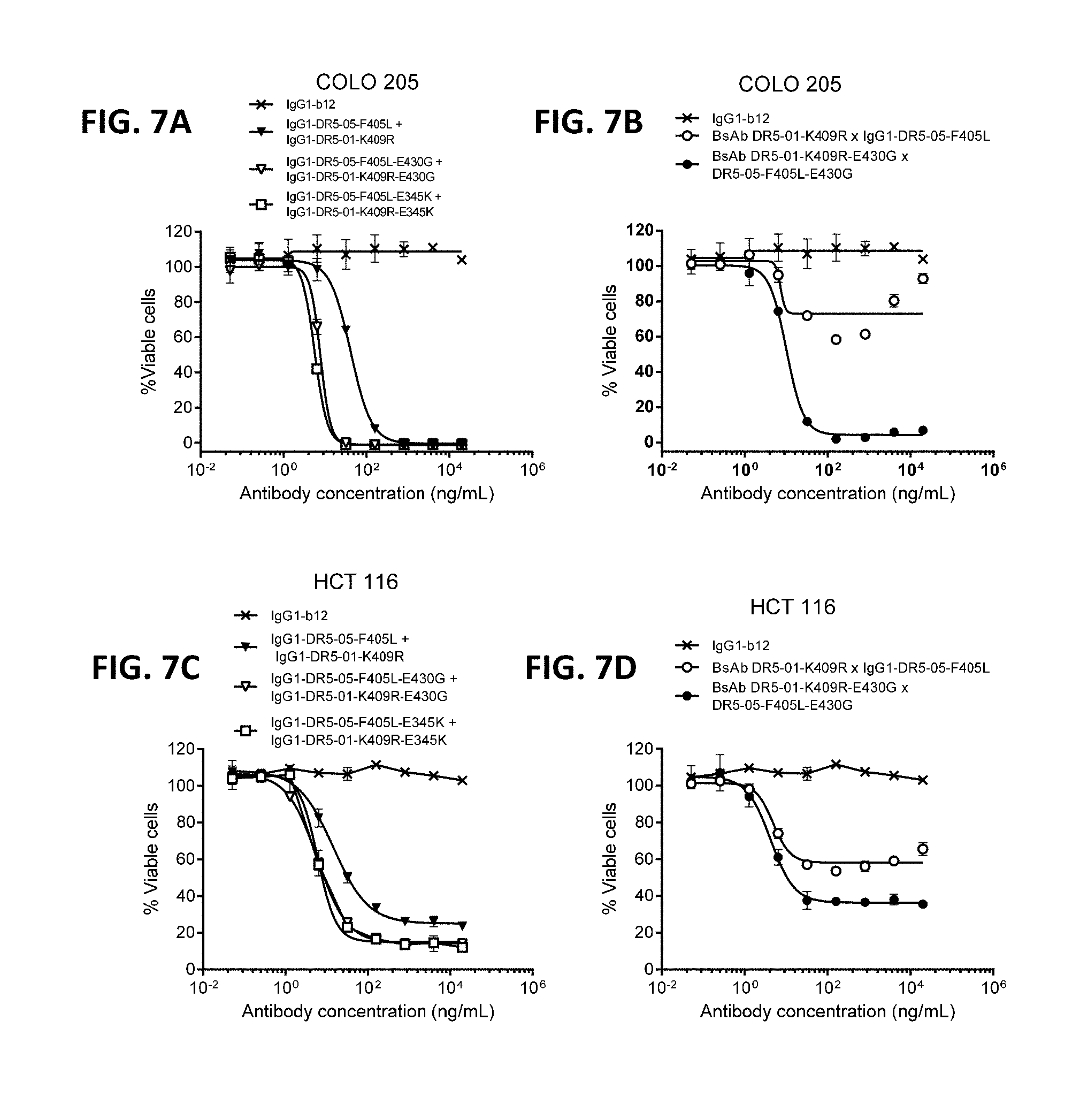
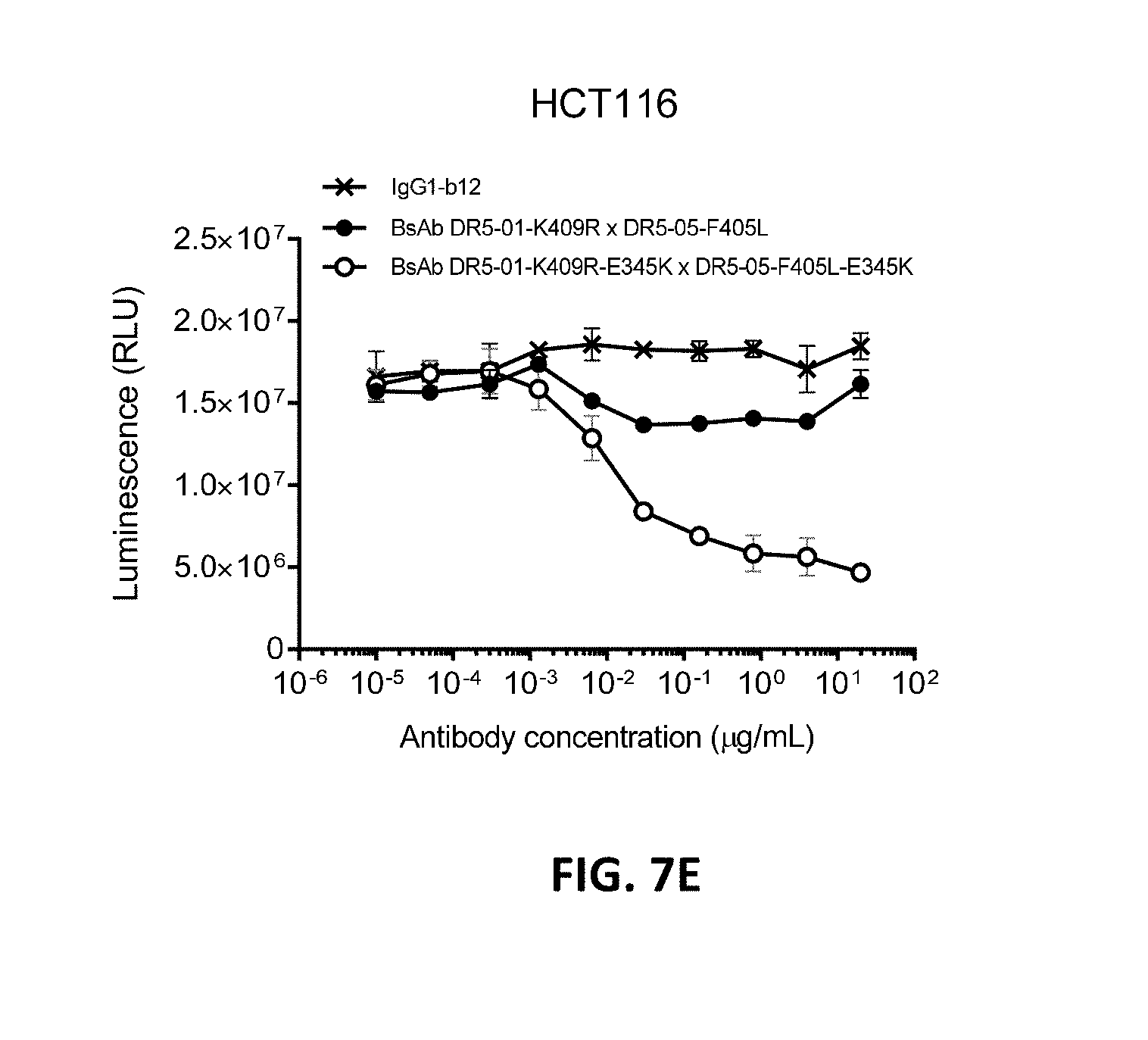
[0031] FIGS. 7A-7E show that introduction of a hexamerization-enhancing mutation resulted in enhanced induction of killing by the antibody combination IgG1-DR5-01-K409R+IgG1-DR5-05-F405 (FIGS. 7A and 7C) and the BsAb DR5-01-K409R.times.DR5-05-F405 (FIGS. 7B and 7D) on both COLO 205 (FIGS. 7A and 7B) and HCT116 (FIGS. 7C-7E) colon cancer cells. Error bars indicate standard deviation.
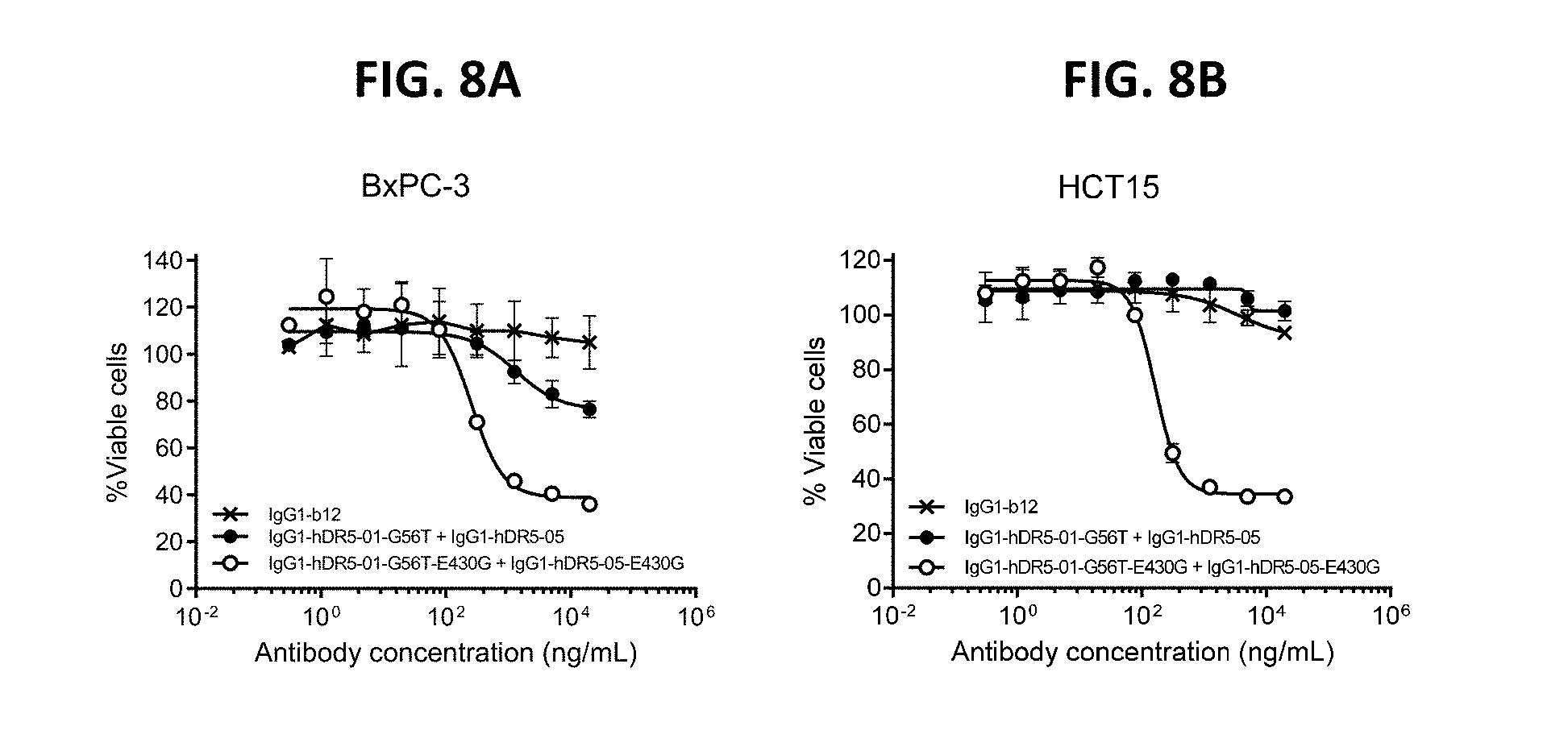
[0032] FIGS. 8A and 8B show the potency of the combination of the antibody combination IgG1-hDR5-01-G56T-E430G+IgG1-hDR5-05-E430G compared to the combination without E430G mutation as measured in a viability assay on BxPC-3 pancreatic (FIG. 8A) and HCT15 colon cancer cells (FIG. 8B). Graphs represent mean values of duplicate samples+/-standard deviation.
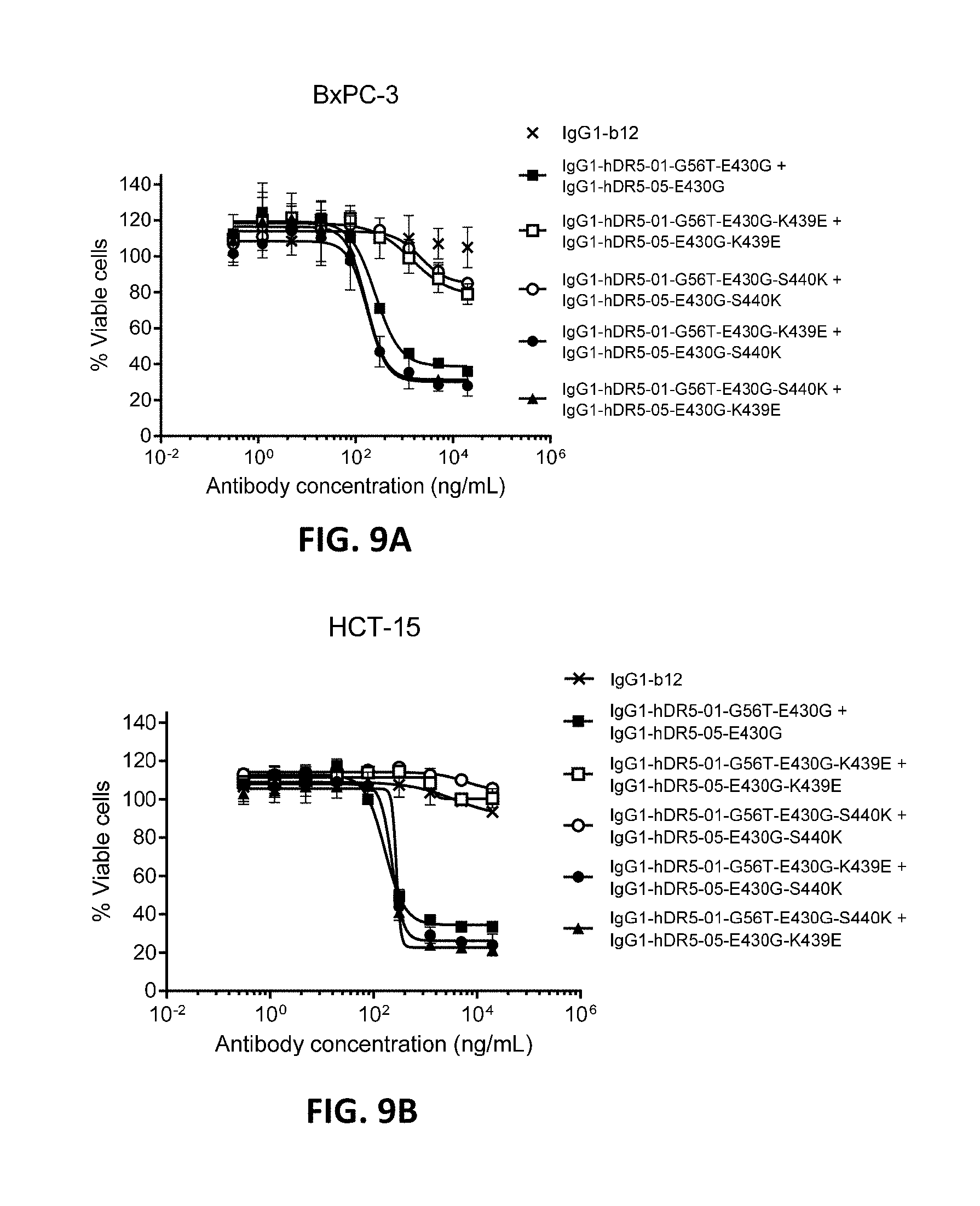
[0033] FIGS. 9A and 9B show a viability assay with repulsing and complementary variants of IgG1-hDR5-01-G56T-E430G and IgG1-hDR5-05-E430G. Introduction of the same repulsing mutation (K439E or S440K) in both antibodies resulted in diminished induction of killing of BxPC-3 pancreatic (FIG. 9A) and HCT-15 colon cancer cells (FIG. 9B). By combining the two mutations (K439E and S440K) in both antibodies, repulsion was neutralized and killing restored. Error bars indicate standard deviation.
[0034] FIG. 10 shows the involvement of Fc interactions in the capacity of the antibody combination IgG1-hDR5-01-G56T-E430G+IgG1-hDR5-05-E430G with hexamerization-enhancing mutation to induce receptor clustering on the cell surface and induction of apoptosis. Induction of apoptosis is inhibited by the Fc-binding peptide DCAWHLGELVWCT as shown in a 3-days viability assay on BxPC-3 human cancer cells.
[0035] FIG. 11 shows that the combination of IgG1-DR5-01-K409R-E430G+IgG1-DR5-05-F405L-E430G reduced the viability of different human cancer cell lines, as determined in a 3-days viability assay. Graphs show the mean+/-standard deviation from duplicate samples. *p<0.05, **p<0.01, ***p<0.001, ****p<0.001 (One-way ANOVA with Tukey's multiple comparisons test). 01 is IgG1-DR5-01-K409R, 05 is IgG1-DR5-05-F405L, 01-E430G is IgG1-DR5-01-K409R-E430G, 05-E430G is IgG1-DR5-05-F405L-E430G.
[0036] FIG. 12 shows that introduction of a hexamerization-enhancing mutation results in enhanced induction of killing of HCT 116 colon cancer cells by the antibody combination IgG1-DR5-05-F405L-E345K+IgG1-CONA-K409R-E430G and BsAb CONA-K409R-E430G.times.DR5-05-F405L-E345K as determined in a 3-days viability assay. Error bars indicate standard deviation. RLU: Relative Luminescence Units.
[0037] FIGS. 13A-13C show the efficacy of the antibody combination IgG1-DR5-01-K409R-E430G+IgG1-DR5-05-F405L-E430G and BsAb DR5-01-K409R-E430G.times.DR5-05-F405L-E430G in the presence or absence of secondary Fc crosslinking by anti-human IgG F(ab')2 and in comparison to DR5 antibodies IgG1-DR5-CONA and IgG1-DR5-chTRA8-F405L in a 3-days viability assay on adherent COLO 205 (FIG. 13A) colorectal and PANC-1 (FIG. 13B) and BxPC-3 (FIG. 13C) pancreatic cancer cells. The non-target binding antibody IgG1-b12 was included as a negative control. Graphs show the mean+/-standard deviation from duplicate samples.
[0038] FIGS. 14A-14C show caspase-dependent programmed cell death by the combination of humanized IgG1-hDR5-01-E430G+IgG1-hDR5-05-E430G (01-E430G+05-E430G) antibodies as measured in a viability assay on PANC-1 (FIGS. 14A and 14B) and BxPC-3 (FIG. 14C) pancreatic cancer cells. ZVAD, Z-VAD-FMK.
[0039] FIGS. 15A-15E show that cell death induction upon binding of DR5 antibody combinations on COLO 205 colon cancer cells. COLO 205 cells were incubated with antibody sample for 5 hours (FIGS. 15A-15C) and 24 hours (FIGS. 15D and 15E). Different stages of cell death induction were analyzed by Annexin V/PI double staining and Active caspase-3 staining. Error bars indicate the standard deviation of 2 duplicate samples. 01 is IgG1-DR5-01-K409R, 05 is IgG1-DR5-05-F405L, 01-E430G is IgG1-DR5-01-K409R-E430G, 05-E430G is IgG1-DR5-05-F405L-E430G, 01x05 is BsAb DR5-01-K409R.times.DR5-05-F405L, 01-E430G.times.05-E430G is BsAb DR5-01-K409R-E430G.times.DR5-05-F405L-E430G.
[0040] FIGS. 16A and 16B show the kinetics of Caspase-3/7 activation upon binding of antibody combination IgG1-DR5-01-K409R-E430G+IgG1-DR5-05-F405L-E430G (FIG. 16A) and BsAb DR5-01-K409R-E430G.times.DR5-05-F405L-E430G (FIG. 16B) on COLO 205 colon cancer cells. COLO 205 cells were incubated with antibody for 1, 2, 5 and 24 hours. Caspase-3/7 activation was analyzed in a homogenous luminescence assay. AU, arbitrary units. Error bars indicate the standard deviation of duplicate samples.
[0041] FIG. 17 shows the potency of the antibody combination IgG1-hDR5-01-K409R-E430G+IgG1-hDR5-05-F405L-E430G and of the antibody combination IgG1-hDR5-01-E430G+IgG1-hDR5-05-E430G as measured in a viability assay on BxPC-3 pancreatic cancer cells. Graphs represent mean values of duplicate samples+/-standard deviation.
[0042] FIG. 18 shows the efficacy of different ratios of IgG1-DR5-01-K409R-E430G and IgG1-DR5-05-F405L-E430G (DR5-01-E430G:DR5-05-E430G) on adherent BxPC-3 human cancer cells as determined in a 3-days viability assay.
[0043] FIGS. 19A and 19B show the efficacy of different ratios of IgG1-hDR5-01-G56T-E430G and IgG1-hDR5-05-E430G (DR5-01-E430G:DR5-05-E430G) on adherent BxPC-3 (FIG. 19A) and HCT-15 (FIG. 19B) human cancer cells as determined in a 3-days viability assay.
[0044] FIGS. 20A and 20B the evaluation of the in vivo efficacy of the chimeric IgG1-DR5-05-F405L with and without the hexamerization-enhancing mutation E430G in a subcutaneous xenograft model with COLO 205 human colon cancer cells. Tumor development (mean & SEM) in mice treated with the indicated antibodies (5 mg/kg) is shown in time (FIG. 20A). In (FIG. 20B) the percentage of mice with tumor sizes smaller than 750 mm3 is shown in a Kaplan-Meier plot.
[0045] FIG. 21 shows a viability assay on Jurkat human T lymphocytes with variants of FAS antibody IgG1-FAS-E09. In the presence of the Fc-Fc repulsing mutation K439E or S440K, killing by IgG1-FAS-E09 variants with hexamerization-enhancing mutations E345R/E430G/S440Y (RGY) or E345R/E430G/Y436I (RGI) was inhibited. RGEY: E345R/E430G/K439E/S440Y; RGIK: E345R/E430G/Y436I/S440K.
[0046] FIG. 22 shows a viability assays with DR5 antibodies IgG1-DR5-CONA and IgG1-DR5-CONA-E430G on attached COLO 205 human colon cancer cells. Introduction of the hexamerization-enhancing mutation E430G resulted in induction of killing. Data are presented as % viable cells calculated from the luminescence relative to samples incubated without antibody (no kill) and samples incubated with Staurosporine (maximal kill). Error bars indicate standard deviation.
[0047] FIGS. 23A and 23B show a viability assay with DR5 antibodies on COLO 205 human colon cancer cells. Introduction of the hexamerization-enhancing mutation S440Y resulted in induction of killing by the single antibodies IgG1-hDR5-01-G56T and IgG1-hDR5-05 (FIG. 23A) and increased efficacy of the antibody combination IgG1-hDR5-01-G56T+IgG1-hDR5-05 (FIG. 23B). Data are presented as % viable cells calculated from the luminescence relative to samples incubated without antibody (no kill) and samples incubated with Staurosporine (maximal kill). Error bars indicate standard deviation.
[0048] FIG. 24A shows crossblock ELISA between IgG1-DR5-CONA-K409R and IgG1-DR5-chTRA8-F405L. (FIG. 24B) Introduction of the E430G hexamerization-enhancing mutation resulted in enhanced induction of killing of BxPC-3 human pancreatic cancer cells by the combination of the non-crossblocking antibodies IgG1-DR5-CONA-E430G+IgG1-DR5-chTRA8-E430G as determined in a 3-days viability assay. Error bars indicate standard deviation.
[0049] FIGS. 25A-25C show the evaluation of the in vivo efficacy of the anti-DR5 antibody concentration IgG1-hDR5-01-G56T+IgG1-hDR5-05 with and without the hexamerization-enhancing mutation E430G in a subcutaneous xenograft model with HCT15 human colon cancer cells. Tumor development (mean & SEM) in mice treated with the 0.5 mg/kg antibodies is shown in time (FIG. 25A) and at day 21 after start treatment (FIG. 25B). **P<0.0011 (Mann Whitney test). In (FIG. 25C) the percentage of mice with tumor sizes smaller than 750 mm3 is shown in a Kaplan-Meier plot.
[0050] FIGS. 26A and 26B show evaluation of the in vivo efficacy of the antibodies IgG1-hDR5-01-G56T-E430G and IgG1-hDR5-05-E430G, both as single agents and as a combination in comparison to the parental antibodies without the E430G mutation in a subcutaneous xenograft model with COLO 205 human colon cancer cells. (FIG. 26A) Tumor size (mean & SEM) in mice treated with the indicated antibodies (0.5 mg/kg) as shown in time. (FIG. 26B) Kaplan-Meier plot of tumor progression, with a cutoff set at a tumor volume >500 mm.sup.3.
DETAILED DESCRIPTION OF THE INVENTION
[0051] In describing the embodiments of the invention specific terminology will be resorted to for the sake of clarity. However, the invention is not intended to be limited to the specific terms so selected, and it is understood that each specific term includes all technical equivalents which operate in a similar manner to accomplish a similar purpose.
[0052] As described herein, surprisingly it has been found that antibodies binding to a Death Receptor such as FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR and comprising a mutation at an amino acid in the Fc region corresponding to position E430, E345 or S440 in human IgG1 according to EU numbering, were found to be superior at inducing apoptosis in cancer cells expressing FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR compared to anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibodies without said mutation in one of the above mentioned positions. Furthermore, compositions comprising two or more anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR, and anti-NGFR antibodies of the invention, which bind different epitopes on FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR, were found superior to compositions comprising the same anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibodies without said mutation. That is compositions with two or more antibodies of the present invention were superior at inducing apoptosis and/or inhibiting cell growth of cancer cells expressing FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR compared to compositions comprising the two same anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibodies without said mutation in the Fc region. It is to be understood that in the context of this invention the same antibody is antibodies having the identical antigen binding region. Thus the same antibody has an identical amino acid sequence as an antibody of the present invention, but does not have said mutation in the Fc region. By introducing specific mutations in the Fc region, oligomerization such as hexamerization upon target binding on the cell surface can be enhanced, while the antibody molecules remain monomeric in solution WO2013/004842, WO2014/108198.
Definitions
[0053] The term "immunoglobulin" as used herein, refers to a class of structurally related glycoproteins consisting of two pairs of polypeptide chains, one pair of light (L) low molecular weight chains and one pair of heavy (H) chains, all four potentially inter-connected by disulfide bonds. The structure of immunoglobulins has been well characterized. See for instance Fundamental Immunology Ch. 7 (Paul, W., ed., 2nd ed. Raven Press, N.Y. (1989)). Briefly, each heavy chain typically is comprised of a heavy chain variable region (abbreviated herein as VH) and a heavy chain constant region. The heavy chain constant region of IgG antibodies typically is comprised of three domains, CH1, CH2, and CH3. The heavy chains are inter-connected via disulfide bonds in the so-called "hinge region". Each light chain typically is comprised of a light chain variable region (abbreviated herein as VL) and a light chain constant region. The light chain constant region typically is comprised of one domain, CL. The VH and VL regions may be further subdivided into regions of hypervariability (or hypervariable regions which may be hypervariable in sequence and/or form of structurally defined loops), also termed complementarity determining regions (CDRs), interspersed with regions that are more conserved, termed framework regions (FRs). Each VH and VL is typically composed of three CDRs and four FRs, arranged from amino-terminus to carboxy-terminus in the following order: FR1, CDR1, FR2, CDR2, FR3, CDR3, FR4 (see also Chothia and Lesk J. Mol. Biol. 196, 901 917 (1987)). Unless otherwise stated or contradicted by context, reference to amino acid positions in the present invention is according to the EU-numbering (Edelman et al., Proc Natl Acad Sci USA. 1969 May; 63(1):78-85; Kabat et al., Sequences of Proteins of Immunological Interest, Fifth Edition. 1991 NIH Publication No. 91-3242).
[0054] The term "hinge region" as used herein is intended to refer to the hinge region of an immunoglobulin heavy chain. Thus, for example the hinge region of a human IgG1 antibody corresponds to amino acids 216-230 according to the EU numbering.
[0055] The term "CH2 region" or "CH2 domain" as used herein is intended to refer the CH2 region of an immunoglobulin heavy chain. Thus, for example the CH2 region of a human IgG1 antibody corresponds to amino acids 231-340 according to the EU numbering. However, the CH2 region may also be any of the other isotypes or allotypes as described herein.
[0056] The term "CH3 region" or "CH3 domain" as used herein is intended to refer to the CH3 region of an immunoglobulin heavy chain. Thus, for example the CH3 region of a human IgG1 antibody corresponds to amino acids 341-447 according to the EU numbering. However, the CH3 region may also be any of the other isotypes or allotypes as described herein.
[0057] The term "fragment crystallizable region", "Fc region", "Fc fragment" or "Fc domain", which may be used interchangeably herein, refers to an antibody region comprising, arranged from amino-terminus to carboxy-terminus, at least a hinge region, a CH2 domain and a CH3 domain. An Fc region of an IgG1 antibody can, for example, be generated by digestion of an IgG1 antibody with papain. The Fc region of an antibody may mediate the binding of the immunoglobulin to host tissues or factors, including various cells of the immune system (such as effector cells) and components of the complement system such as C1q, the first component in the classical pathway of complement activation.
[0058] The term "Fab fragment" in the context of the present invention, refers to a fragment of an immunoglobulin molecule, which comprises the variable regions of the heavy chain and light chain as well as the constant region of the light chain and the CH1 region of the heavy chain of an immunoglobulin. The "CH1 region" refers e.g. to the region of a human IgG1 antibody corresponding to amino acids 118-215 according to the EU numbering. Thus, the Fab fragment comprises the binding region of an immunoglobulin.
[0059] The term "antibody" (Ab), as used herein refers to an immunoglobulin molecule, a fragment of an immunoglobulin molecule, or a derivative of either thereof. The antibody of the present invention comprises an Fc-region of an immunoglobulin and an antigen-binding region. The Fc region generally contains two CH2-CH3 regions and a connecting region, e.g. a hinge region. The variable regions of the heavy and light chains of the immunoglobulin molecule contain a binding domain that interacts with an antigen. The term "antibody" as used herein, also refers to, unless otherwise specified or contradicted by the context, polyclonal antibodies, oligoclonal antibodies, monoclonal antibodies (such as human monoclonal antibodies), antibody mixtures, recombinant polyclonal antibodies, chimeric antibodies, humanized antibodies and human antibodies. An antibody as generated can potentially possess any class or isotype.
[0060] The term "human antibody", as used herein, refers to antibodies having variable and constant regions derived from human germline immunoglobulin sequences. The human antibodies of the invention may include amino acid residues not encoded by human germline immunoglobulin sequences (e.g., mutations, insertions or deletions introduced by random or site-specific mutagenesis in vitro or by somatic mutation in vivo). However, the term "human antibody", as used herein, is not intended to include antibodies in which CDR sequences derived from the germline of another species, such as a mouse, have been grafted onto human framework sequences.
[0061] The term "chimeric antibody", as used herein, refers to an antibody in which both chain types are chimeric as a result of antibody engineering. A chimeric chain is a chain that contains a foreign variable domain (originating from a non-human species, or synthetic or engineered from any species including human) linked to a constant region of human origin.
[0062] The term "humanized antibody", as used herein, refers to an antibody in which both chain types are humanized as a result of antibody engineering. A humanized chain is typically a chain in which the complementarity determining regions (CDR) of the variable domains are foreign (originating from a species other than human, or synthetic) whereas the remainder of the chain is of human origin. Humanization assessment is based on the resulting amino acid sequence, and not on the methodology per se, which allows protocols other than grafting to be used.
[0063] The term "isotype", as used herein, refers to the immunoglobulin class (for instance IgG1, IgG2, IgG3, IgG4, IgD, IgA1, IgA2, IgE, or IgM) that is encoded by heavy chain constant region genes. To produce a canonical antibody, each heavy chain isotype is to be combined with either a kappa (.kappa.) or lambda (.lamda.) light chain.
[0064] The term "allotype", as used herein, refers to the amino acid variation within one isotype class in the same species. The predominant allotype of an antibody isotype varies between ethnicity individuals. The known allotype variations within the IgG1 isotype of the heavy chain result from 4 amino acid substitutions in the antibody frame as illustrated in FIG. 1. In one embodiment the antibody of the invention is of the IgG1m(f) allotype as defined in SEQ ID NO 1. In one embodiment of the invention the antibody is of the IgG1m(z) allotype as defined in SEQ ID NO 2, the IgG1m(a) allotype as defined in SEQ ID NO 3, the IgG1m(x) allotype as defined in SEQ ID NO 4, or any allotype combination, such as IgG1m(z,a), IgG1m(z,a,x), IgG1m(f,a) (de lange Exp Clin Immunogenet. 1989; 6(1):7-17).
[0065] The terms "monoclonal antibody", "monoclonal Ab", "monoclonal antibody composition", "mAb", or the like, as used herein refer to a preparation of Ab molecules of single molecular composition. A monoclonal antibody composition displays a single binding specificity and affinity for a particular epitope. Accordingly, the term "human monoclonal antibody" refers to Abs displaying a single binding specificity which have variable and constant regions derived from human germline immunoglobulin sequences. The human mAbs may be generated by a hybridoma which includes a B cell obtained from a transgenic or transchromosomal non-human animal, such as a transgenic mouse, having a genome comprising a human heavy chain transgene repertoire and a human light chain transgene repertoire, rearranged to produce a functional human antibody and fused to an immortalized cell. Alternatively, the human mAbs may be generated recombinantly.
[0066] The term "antibody mimetics" as used herein, refers to compounds that, like antibodies, can specifically bind antigens, but that are not structurally related to antibodies. They are usually artificial peptides, proteins, nucleic acids or small molecules.
[0067] The term "bispecific antibody" refers to antibody having specificities for at least two different, typically non-overlapping, epitopes. Such epitopes may be on the same or different targets Examples of different classes of bispecific antibodies comprising an Fc region include but are not limited to: asymmetric bispecific molecules e.g. IgG-like molecules with complementary CH3 domains and symmetric bispecific molecules e.g. recombinant IgG-like dual targeting molecules wherein each antigen-binding region of the molecule binds at least two different epitopes.
[0068] Examples of bispecific molecules include but are not limited to Triomab.RTM. (Trion Pharma/Fresenius Biotech, WO/2002/020039), Knobs-into-Holes (Genentech, WO9850431), CrossMAbs (Roche, WO 2009/080251, WO 2009/080252, WO 2009/080253), electrostatically-matched Fc-heterodimeric molecules (Amgen, EP1870459 and WO2009089004; Chugai, US201000155133; Oncomed, WO2010129304), LUZ-Y (Genentech), DIG-body, PIG-body and TIG-body (Pharmabcine), Strand Exchange Engineered Domain body (SEEDbody) (EMD Serono, WO2007110205), Bispecific IgG1 and IgG2 (Pfizer/Rinat, WO11143545), Azymetric scaffold (Zymeworks/Merck, WO2012058768), mAb-Fv (Xencor, WO2011028952), XmAb (Xencor), Bivalent bispecific antibodies (Roche, WO2009/080254), Bispecific IgG (Eli Lilly), DuoBody' molecules (Genmab A/S, WO 2011/131746), DuetMab (Medimmune, US2014/0348839), Biclonics (Merus, WO 2013/157953), NovImmune (.kappa..lamda.Bodies, WO 2012/023053), Fc.DELTA.Adp (Regeneron, WO 2010/151792), (DT)-Ig (GSK/Domantis), Two-in-one Antibody or Dual Action Fabs (Genentech, Adimab), mAb2 (F-Star, WO2008003116), Zybodies.TM. (Zyngenia), CovX-body (CovX/Pfizer), FynomAbs (Covagen/Janssen Cilag), DutaMab (Dutalys/Roche), iMab (Medimmune), Dual Variable Domain (DVD)-Ig.TM. (Abbott, U.S. Pat. No. 7,612,18), dual domain double head antibodies (Unilever; Sanofi Aventis, WO20100226923), Ts2Ab (Medimmune/AZ), BsAb (Zymogenetics), HERCULES (Biogen Idec, U.S. Ser. No. 00/795,1918), scFv-fusions (Genentech/Roche, Novartis, Immunomedics, Changzhou Adam Biotech Inc, CN 102250246), TvAb (Roche, WO2012025525, WO2012025530), ScFv/Fc Fusions, SCORPION (Emergent BioSolutions/Trubion, Zymogenetics/BMS), Interceptor (Emergent), Dual Affinity Retargeting Technology (Fc-DART.TM.) (MacroGenics, WO2008/157379, WO2010/080538), BEAT (Glenmark), Di-Diabody (Imclone/Eli Lilly) and chemically crosslinked mAbs (Karmanos Cancer Center), and covalently fused mAbs (AIMM therapeutics).
[0069] The term "full-length antibody" when used herein, refers to an antibody (e.g., a parent or variant antibody) which contains all heavy and light chain constant and variable domains corresponding to those that are normally found in a wild-type antibody of that class or isotype.
[0070] The term "oligomer" as used herein, refers to a molecule that consists of more than one but a limited number of monomer units (e.g. antibodies) in contrast to a polymer that, at least in principle, consists of an unlimited number of monomers. Exemplary oligomers are dimers, trimers, tetramers, pentamers and hexamers. Greek prefixes are often used to designate the number of monomer units in the oligomer, for example a tetramer being composed of four units and a hexamer of six units. Likewise, the term "oligomerization", as used herein, is intended to refer to a process that converts molecules to a finite degree of polymerization. Herein, it is observed, that antibodies and/or other dimeric proteins comprising target-binding regions according to the invention can form oligomers, such as hexamers, via non-covalent association of Fc-regions after target binding, e.g., at a cell surface.
[0071] The term "antigen-binding region", "antigen binding region", "binding region" or antigen binding domain, as used herein, refers to a region of an antibody which is capable of binding to the antigen. This binding region is typically defined by the VH and VL domains of the antibody which may be further subdivided into regions of hypervariability (or hypervariable regions which may be hypervariable in sequence and/or form of structurally defined loops), also termed complementarity determining regions (CDRs), interspersed with regions that are more conserved, termed framework regions (FRs). The antigen can be any molecule, such as a polypeptide, e.g. present on a cell, bacterium, or virion or in solution. The terms "antigen" and "target" may, unless contradicted by the context, be used interchangeably in the context of the present invention.
[0072] The term "target", as used herein, refers to a molecule to which the antigen binding region of the antibody binds. The target includes any antigen towards which the raised antibody is directed. The term "antigen" and "target" may in relation to an antibody be used interchangeably and constitute the same meaning and purpose with respect to any aspect or embodiment of the present invention.
[0073] The term "epitope" means a protein determinant capable of specific binding to an antibody. Epitopes usually consist of surface groupings of building blocks such as amino acids, sugar side chains or a combination thereof and usually have specific three-dimensional structural characteristics, as well as specific charge characteristics. Conformational and non-conformational epitopes are distinguished in that the binding to the former but not the latter is lost in the presence of denaturing solvents. The epitope may comprise amino acid residues directly involved in the binding and other amino acid residues, which are not directly involved in the binding, such as amino acid residues which are effectively blocked by the specifically antigen binding peptide (in other words, the amino acid residue is within the footprint of the specifically antigen binding peptide).
[0074] The term "binding" as used herein refers to the binding of an antibody to a predetermined antigen or target, typically with a binding affinity corresponding to a K.sub.D of about 10.sup.-6 M or less, e.g. 10.sup.-7 M or less, such as about 10.sup.-8 M or less, such as about 10.sup.-9 M or less, about 10.sup.-10 M or less, or about 10.sup.-11 M or even less when determined by for instance surface plasmon resonance (SPR) technology in a BIAcore 3000 instrument using the antigen as the ligand and the antibody as the analyte or visa versa, and binds to the predetermined antigen with an affinity corresponding to a K.sub.D that is at least ten-fold lower, such as at least 100 fold lower, for instance at least 1,000 fold lower, such as at least 10,000 fold lower, for instance at least 100,000 fold lower than its affinity for binding to a non-specific antigen (e.g., BSA, casein) other than the predetermined antigen or a closely-related antigen. The amount with which the affinity is lower is dependent on the K.sub.D of the antibody, so that when the K.sub.D of the antibody is very low (that is, the antibody is highly specific), then the degree with which the affinity for the antigen is lower than the affinity for a non-specific antigen may be at least 10,000 fold. The term "K.sub.D" (M), as used herein, refers to the dissociation equilibrium constant of a particular antibody-antigen interaction, and is obtained by dividing k.sub.d by k.sub.a.
[0075] The term "k.sub.d" (sec.sup.-1), as used herein, refers to the dissociation rate constant of a particular antibody-antigen interaction. Said value is also referred to as the k.sub.off value or off-rate.
[0076] The term "k.sub.a" (M.sup.-1.times.sec.sup.-1), as used herein, refers to the association rate constant of a particular antibody-antigen interaction. Said value is also referred to as the k.sub.on value or on-rate.
[0077] The term "K.sub.A" (M.sup.-1), as used herein, refers to the association equilibrium constant of a particular antibody-antigen interaction and is obtained by dividing k.sub.a by k.sub.d.
[0078] As used herein, the term "affinity" is the strength of binding of one molecule, e.g. an antibody, to another, e.g. a target or antigen, at a single site, such as the monovalent binding of an individual antigen binding site of an antibody to an antigen.
[0079] As used herein, the term "avidity" refers to the combined strength of multiple binding sites between two structures, such as between multiple antigen binding sites of antibodies simultaneously interacting with a target. When more than one binding interactions are present, the two structures will only dissociate when all binding sites dissociate, and thus, the dissociation rate will be slower than for the individual binding sites, and thereby providing a greater effective total binding strength (avidity) compared to the strength of binding of the individual binding sites (affinity).
[0080] The term "hexamerization enhancing mutation", as used herein, refers to a mutation of an amino acid position corresponding to E430, E345 or S440 in human IgG1 according to EU numbering. The hexamerization enhancing mutation strengthens Fc-Fc interactions between neighbouring IgG antibodies that are bound to a cell surface target, resulting in enhanced hexamer formation of the target-bound antibodies, while the antibody molecules remain monomeric in solution as described in WO2013/004842; WO2014/108198.
[0081] The term "repulsing mutation" or "self-repulsing mutation" or "hexamerization-inhibiting mutation", as used herein, refers to a mutation of an amino acid position of human IgG1 that can result in charge repulsion between amino acids at the Fc-Fc interface, resulting in weakening of the Fc-Fc interaction between two adjacent Fc region containing polypeptides, and thus inhibiting hexamerization. Examples of such a repulsing mutation in human IgG1 are K439E and S440K. The repulsion in the Fc-Fc interaction between two adjacent Fc region containing polypeptides at the position of a repulsing mutation can be neutralized by introduction of a second mutation (complementary mutation) in the amino acid position that interacts with the position harboring the first mutation. This second mutation can be present either in the same antibody or in a second antibody. The combination of the first and second mutation results in neutralization of the repulsion and restoration of the Fc-Fc interactions and thus hexamerization. Examples of such first and second mutations are K439E (repulsing mutation) and S440K (neutralizing the repulsion by K439E), and vice versa S440K (repulsing mutation) and K439E (neutralizing the repulsion by S440K).
[0082] The term "complementary mutation", as used herein, refers to a mutation of an amino acid position in a Fc region-containing polypeptide that relates to a first mutation in an adjacent Fc region containing polypeptide that preferably interacts with the Fc region-containing polypeptide containing the complementary mutation due to the combination of the two mutations in the two adjacent Fc region-containing polypeptides. The complementary mutation and the related first mutation can be present either in the same antibody (intramolecular) or in a second antibody (intermolecular). An example of intramolecular complementary mutations is the combination K409R and F405L that mediates preferential heterodimerization in a bispecific antibody according to WO 2011/131746. The combination of the K439E and S440K mutations that results in neutralization of repulsion and restoration of Fc-Fc interactions between two adjacent Fc region containing polypeptides and thus hexamerization is an example of complementary mutations that can be applied both inter- and intramolecularly.
[0083] The term "apoptosis", as used herein refers to the process of programmed cell death (PCD) that may occur in a cell. Biochemical events lead to characteristic cell changes (morphology) and death. These changes include blebbing, cell shrinkage, phosphatidylserine exposure, loss of mitochondrial function, nuclear fragmentation, chromatin condensation, caspase activation, and chromosomal DNA fragmentation.
[0084] The term "programmed cell-death" or "PCD", as used herein refers to the death of a cell in any form mediated by an intracellular signaling, e.g. apoptosis, autophagy or necroptosis.
[0085] The term "Annexin V", as used herein, refers to a protein of the annexin group that binds phosphatidylserine (PS) on the cell surface.
[0086] The term "caspase activation", as used herein, refers to cleavage of inactive pro-forms of effector caspases by initiator caspases, leading to their conversion into effector caspases, which in turn cleave protein substrates within the cell to trigger apoptosis.
[0087] The term "caspase-dependent programmed cell death", as used herein refers to any form of programmed cell death mediated by caspases. In a particular embodiment, caspase-dependent programmed cell death by one or more agonistic anti-DR5 antibodies can be determined by comparing the viability of a cell culture in the presence and absence of pan-caspase inhibitor Z-Val-Ala-DL-Asp-fluoromethylketone (Z-VAD-FMK). Pan-caspase inhibitor Z-VAD-FMK (5 .mu.M end concentration) may be added to adhered cells in 96-well flat bottom plates and incubated for one hour at 37.degree. C. Next, antibody concentration dilution series (e.g. starting from e.g. 20,000 ng/mL to 0.05 ng/mL final concentration in 5-fold dilutions) may be added and incubated for 3 days at 37.degree. C. Cell viability can be quantified using special kits for this purpose, such as the CellTiter-Glo luminescent cell viability assay of Promega (Cat nr G7571).
[0088] The term "cell viability", as used herein refers to the presence of metabolically active cells. In a particular embodiment, cell viability after incubation with one or more agonistic anti-Death Receptor antibodies can be determined by quantifying the ATP present in the cells. Antibody concentration dilution series (e.g. starting from e.g. 20,000 ng/mL to 0.05 ng/mL final concentration in 5-fold dilutions) may be added to cells in 96-well flat bottom plates, medium may be used as negative control and 5 .mu.M staurosporine may be used as positive control for the induction of cell death. After 3 days incubation cell viability may be quantified using special kits for this purpose, such as the CellTiter-Glo luminescent cell viability assay of Promega (Cat nr G7571). The percentage viable cells can be calculated using the following formula: % viable cells=[(luminescence antibody sample-luminescence staurosporine sample)/(luminescence no antibody sample-luminescence staurosporine sample)]*100.
[0089] The term "Death Receptor", as used herein, refers to a member of the tumor necrosis factor receptor superfamily (TNFR-SF) comprising an intracellular death domain (DD).
[0090] An intracellular death domain, as used herein, refers to the death domain in the intracellular part of the eight members of the TNFRSF comprising a death domain. The death domains (DDs) are well-known protein interaction modules that belong to the death domain superfamily (Park Apoptosis. 2011 March; 16(3):209-20).
[0091] The term DR1, as used herein, refers to death receptor 1, also known as "TNFR1", CD120a, p55 and tumor necrosis factor receptor superfamily member 1A (TNFRSF1A), which is a single-pass type I membrane protein with four extracellular cysteine-rich domains (CRD's), a transmembrane domain (TM) and a cytoplasmic domain containing a death domain (DD) (Schall et al., Cell. 1990 Apr. 20; 61(2):361-70). Natural ligands for TNFR1 are tumor necrosis factor alpha (TNF-alpha) and lymphotoxin-alpha (LT-alpha). In humans, the DR1 protein is encoded by a nucleic acid sequence encoding the amino acid sequence UniprotKB/Swissprot P19438.
[0092] The term "DR2", as used herein, refers to death receptor 2, also known as "FAS", CD95, APO-1 and tumor necrosis factor receptor superfamily member 6 (TNFRSF6), which is a single-pass type I membrane protein with three extracellular cysteine-rich domains (CRD's), a transmembrane domain (TM) and a cytoplasmic domain containing a death domain (DD) (Lichter et al., Genomics. 1992 September; 14(1):179-80; Inazawa et al., Genomics. 1992 November; 14(3):821-2). The natural ligand for FAS is FASL (CD95L). In humans, the DR2 protein is encoded by a nucleic acid sequence encoding the amino acid sequence UniprotKB/Swissprot P25445.
[0093] The term "DR3", as used herein, refers to death receptor 3, also known as APO3, Apoptosis-inducing receptor (AIR), TRAMP, Lymphocyte-associated receptor of death (LARD), APO-3 and tumor necrosis factor receptor superfamily member 25 (TNFRSF25), which is a single-pass type I membrane protein with four extracellular cysteine-rich domains (CRDs), a transmembrane domain (TM) and a cytoplasmic domain containing a death domain (DD) (Bodmer et al., Immunity. 1997 January; 6(1):79-88). The natural ligand for DR3 is TWEAK. In humans, the DR3 protein is encoded by a nucleic acid sequence encoding the amino acid sequence UniprotKB/Swissprot Q93038.
[0094] The term "DR4", as used herein, refers to death receptor 4, also known as CD261, TNF-related apoptosis-inducing ligand receptor 1 (TRAILR1), APO-2 and tumor necrosis factor receptor superfamily member 10A (TNFRSF10A), which is a single-pass type I membrane protein with three extracellular cysteine-rich domains (CRD's), a transmembrane domain (TM) and a cytoplasmic domain containing a death domain (DD) (Pan et al., Science. 1997 Apr. 4; 276(5309):111-3). The natural ligand for DR4 is TRAIL. In humans, the DR4 protein is encoded by a nucleic acid sequence encoding the amino acid sequence UniprotKB/Swissprot 000220.
[0095] The term "DR5", as used herein, refers to death receptor 5, also known as CD262 and TNF-related apoptosis-inducing ligand receptor 2 (TRAILR2) and tumor necrosis factor receptor superfamily member 1013 TNFRSF10B, which is a single-pass type I membrane protein with three extracellular cysteine-rich domains (CRD's), a transmembrane domain (TM) and a cytoplasmic domain containing a death domain (DD) (Walczak et al., EMBO J. 1997 Sep. 1; 16(17):5386-97). The natural ligand for DR5 is TRAIL. In humans, the DR5 protein is encoded by a nucleic acid sequence encoding the amino acid sequence UniprotKB/Swissprot 014763).
[0096] The term "DR6", as used herein, refers to death receptor 6, also known as CD358 and tumor necrosis factor receptor superfamily member 21 (TNFRSF21), which is a single-pass type I membrane protein with four extracellular cysteine-rich domains (CRD's), a transmembrane domain (TM) and a cytoplasmic domain containing a death domain (DD) (Pan et al., FEBS Lett. 1998 Jul. 24; 431(3):351-6). DR6 is activated by overexpression. A natural ligand for DR6 is alpha-amyloid precursor protein (APP). In humans, the DR6 protein is encoded by a nucleic acid sequence encoding the amino acid sequence UniprotKB/Swissprot 075509.
[0097] The term "EDAR", as used herein, refers to Ectodysplasin-A receptor, also known as Ectodermal dysplasia receptor, EDA-A1 receptor, Downless homolog, anhidrotic ectodysplasin receptor 1 and Tumor necrosis factor receptor superfamily member EDAR, which is a single-pass type I membrane protein with three extracellular cysteine-rich domains (CRD's), a transmembrane domain (TM) and a cytoplasmic domain containing a death domain (DD) (Kumar et al., J Biol Chem. 2001 Jan. 26; 276(4):2668-77). The natural ligand for EDAR is ectodysplasin A. In humans, the EDAR protein is encoded by a nucleic acid sequence encoding the amino acid sequence UniprotKB/Swissprot Q9UNE0.
[0098] The term "NGFR", as used herein, refers to nerve growth factor receptor, also known as low-affinity nerve growth factor receptor (LNGFR), p75NTR, CD271 and tumor necrosis factor receptor superfamily member 16 (TNFRSF16), which is a single-pass type I membrane protein with four extracellular cysteine-rich domains (CRD's), a serine/threonine-rich region, a transmembrane domain (TM) and a cytoplasmic domain containing a death domain (DD) (Johnson et al., Cell. 1986 Nov. 21; 47(4):545-54). The natural ligand for NGFR is nerve growth factor (NGF) that binds the serine/threonine-rich domain in NGFR. In humans, the NGFR protein is encoded by a nucleic acid sequence encoding the amino acid sequence UniprotKB/Swissprot P08138.
[0099] The term "antibody binding death receptor", "anti-death receptor antibody" death receptor-binding antibody", "death receptor-specific antibody", "death receptor antibody" which may be used interchangeably herein, refers to any antibody binding an epitope on the extracellular part of a death receptor such as FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR."
[0100] The term "antibody binding FAS", "anti-FAS antibody" FAS-binding antibody", "FAS-specific antibody", "FAS antibody" which may be used interchangeably herein, refers to any antibody binding an epitope on the extracellular FAS
[0101] The term "antibody binding DR4", "anti-DR4 antibody" DR4-binding antibody", "DR4-specific antibody", "DR4antibody" which may be used interchangeably herein, refers to any antibody binding an epitope on the extracellular part of DR4.
[0102] The term "antibody binding DR5", "anti-DR5 antibody" DR5-binding antibody", "DR5-specific antibody", "DR5 antibody" which may be used interchangeably herein, refers to any antibody binding an epitope on the extracellular part of DR5."
[0103] The term "antibody binding TNFR1", "anti-TNFR1 antibody" TNFR1-binding antibody", "TNFR1-specific antibody", "TNFR1 antibody" which may be used interchangeably herein, refers to any antibody binding an epitope on the extracellular part of TNFR1."
[0104] The term "antibody binding DR6", "anti-DR6 antibody" DR6-binding antibody", "DR6-specific antibody", "DR6 antibody" which may be used interchangeably herein, refers to any antibody binding an epitope on the extracellular part of DR6."
[0105] The term "antibody binding DR3", "anti-DR3 antibody" DR3-binding antibody", "DR3-specific antibody", "DR3 antibody" which may be used interchangeably herein, refers to any antibody binding an epitope on the extracellular part of DR3."
[0106] The term "antibody binding EDAR", "anti-EDAR antibody" EDAR-binding antibody", "EDAR-specific antibody", "EDAR antibody" which may be used interchangeably herein, refers to any antibody binding an epitope on the extracellular part of EDAR."
[0107] The term "antibody binding NGFR", "anti-NGFR antibody" NGFR-binding antibody", "NGFR-specific antibody", "NGFR antibody" which may be used interchangeably herein, refers to any antibody binding an epitope on the extracellular part of NGFR."
[0108] The term "agonist" as used herein, refers to a molecule such as an anti-Death Receptor antibody that triggers a response in a cell when bound to a Death Receptor, wherein the response may beactivation of the Death Receptor. That the anti-Death Receptor antibody is agonistic is to be understood as that the antibody stimulates, activates or clusters of the Death Receptor as a result of the anti-Death Receptor binding to said Death Receptor. That is an agonistic anti-Death Receptor antibody of the present invention bound to a Death Receptor results in Death Receptor stimulation, clustering or activation of downstream intracellular signaling pathways as the natural ligand bound to the Death Receptor.
[0109] A "variant" or "antibody variant" of the present invention is an antibody molecule which comprises one or more mutations as compared to a "parent" antibody. Exemplary parent antibody formats include, without limitation, a wild-type antibody, a full-length antibody or Fc-containing antibody fragment, a bispecific antibody, a human antibody, humanized antibody, chimeric antibody or any combination thereof.
[0110] Exemplary mutations include amino acid deletions, insertions, and substitutions of amino acids in the parent amino acid sequence. Amino acid substitutions may exchange a native amino acid for another naturally-occurring amino acid, or for a non-naturally-occurring amino acid derivative. The amino acid substitution may be conservative or non-conservative. In the context of the present invention, conservative substitutions may be defined by substitutions within the classes of amino acids reflected in one or more of the following three tables:
Amino Acid Residue Classes for Conservative Substitutions
TABLE-US-00001 [0111] Acidic Residues Asp (D) and Glu (E) Basic Residues Lys (K), Arg (R), and His (H) Hydrophilic Uncharged Residues Ser (S), Thr (T), Asn (N), and Gln (Q) Aliphatic Uncharged Residues Gly (G), Ala (A), Val (V), Leu (L), and Ile (I) Non-polar Uncharged Residues Cys (C), Met (M), and Pro (P) Aromatic Residues Phe (F), Tyr (Y), and Trp (W)
Alternative Conservative Amino Acid Residue Substitution Classes
TABLE-US-00002 [0112] 1 A S T 2 D E 3 N Q 4 R K 5 I L M 6 F Y W
Alternative Physical and Functional Classifications of Amino Acid Residues
TABLE-US-00003 [0113] Alcohol group-containing residues S and T Aliphatic residues I, L, V, and M Cycloalkenyl-associated residues F, H, W, and Y Hydrophobic residues A, C, F, G, H, I, L, M, R, T, V, W, and Y Negatively charged residues D and E Polar residues C, D, E, H, K, N, Q, R, S, and T Positively charged residues H, K, and R Small residues A, C, D, G, N, P, S, T, and V Very small residues A, G, and S Residues involved in turn formation A, C, D, E, G, H, K, N, Q, R, S, P, and T Flexible residues Q, T, K, S, G, D, E, and R
[0114] In the context of the present invention, a substitution in a variant is indicated as: [0115] Original amino acid-position-substituted amino acid;
[0116] The three letter code, or one letter code, are used, including the codes Xaa and X to indicate amino acid residue. Accordingly, the notation "E345R" or "Glu345Arg" means, that the variant comprises a substitution of Glutamic acid with Arginine in the variant amino acid position corresponding to the amino acid in position 345 in the parent antibody.
[0117] Where a position as such is not present in an antibody, but the variant comprises an insertion of an amino acid, for example: Position-substituted amino acid; the notation, e.g., "448E" is used. Such notation is particular relevant in connection with modification(s) in a series of homologous polypeptides or antibodies. Similarly when the identity of the substitution amino acid residues(s) is immaterial: Original amino acid-position; or "E345". For a modification where the original amino acid(s) and/or substituted amino acid(s) may comprise more than one, but not all amino acid(s), the substitution of Glutamic acid for Arginine, Lysine or Tryptophan in position 345: "Glu345Arg,Lys,Trp" or "E345R,K,W" or "E345R/K/W" or "E345 to R, K or W" may be used interchangeably in the context of the invention. Furthermore, the term "a substitution" embraces a substitution into any one of the other nineteen natural amino acids, or into other amino acids, such as non-natural amino acids. For example, a substitution of amino acid E in position 345 includes each of the following substitutions: 345A, 345C, 345D, 345G, 345H, 345F, 345I, 345K, 345L, 345M, 345N, 345Q, 345R, 345S, 345T, 345V, 345W, and 345Y. This is, by the way, equivalent to the designation 345X, wherein the X designates any amino acid. These substitutions can also be designated E345A, E345C, etc, or E345A,C,ect, or E345A/C/ect. The same applies to analogy to each and every position mentioned herein, to specifically include herein any one of such substitutions.
[0118] For the purposes of the present invention, the sequence identity between two amino acid sequences is determined using the Needleman-Wunsch algorithm (Needleman and Wunsch, 1970, J. Mol. Biol. 48: 443-453) as implemented in the Needle program of the EMBOSS package (EMBOSS: The European Molecular Biology Open Software Suite, Rice et al., 2000, Trends Genet. 16: 276-277), preferably version 5.0.0 or later. The parameters used are gap open penalty of 10, gap extension penalty of 0.5, and the EBLOSUM62 (EMBOSS version of BLOSUM62) substitution matrix. The output of Needle labeled "longest identity" (obtained using the -nobrief option) is used as the percent identity and is calculated as follows:
(Identical Residues.times.100)/(Length of Alignment-Total Number of Gaps in Alignment).
[0119] For the purposes of the present invention, the sequence identity between two deoxyribonucleotide sequences is determined using the Needleman-Wunsch algorithm (Needleman and Wunsch, 1970, supra) as implemented in the Needle program of the EMBOSS package (EMBOSS: The European Molecular Biology Open Software Suite, Rice et al., 2000, supra), preferably version 5.0.0 or later. The parameters used are gap open penalty of 10, gap extension penalty of 0.5, and the EDNAFULL (EMBOSS version of NCBI NUC4.4) substitution matrix. The output of Needle labeled "longest identity" (obtained using the -nobrief option) is used as the percent identity and is calculated as follows:
(Identical Deoxyribonucleotides.times.100)/(Length of Alignment-Total Number of Gaps in Alignment).
[0120] The sequence of CDR variants may differ from the sequence of the CDR of the parent antibody sequences through mostly conservative physical or functional amino acids substitutions at most 5 mutations or substitutions selected from conservative, physical or functional amino acids in total across the six CDR sequences of the antibody binding region, such as at most 4 mutations or substitutions selected from conservative, physical or functional amino acids, such as at most 3 mutations or substitutions selected from conservative, physical or functional amino acids, such as at most 2 mutations selected from conservative, physical or functional amino acids or substitutions, such as at most 1 mutation or substitution selected from a conservative, physical or functional amino acid, in total across the six CDR sequences of the antibody binding region. The conservative, physical or functional amino acids are selected from the 20 natural amino acids found i.e, Arg (R), His (H), Lys (K), Asp (D), Glu (E), Ser (S), Thr (T), Asn (N), Gln (Q), Cys (C), Gly (G), Pro (P), Ala (A), Ile (I), Leu (L), Met (M), Phe (F), Trp (W), Tyr (Y) and Val (V).
[0121] The sequence of CDR variants may differ from the sequence of the CDR of the parent antibody sequences through mostly conservative, physical or functional amino acids substitutions; for instance at least about 75%, about 80% or more, about 85% or more, about 90% or more, (e.g., about 75-95%, such as about 92%, 93% or 94%) of the substitutions in the variant are mutations or substitutions selected from conservative, physical or functional amino acids residue replacements.
[0122] The conservative, physical or functional amino acids are selected from the 20 natural amino acids found i.e, Arg (R), His (H), Lys (K), Asp (D), Glu (E), Ser (S), Thr (T), Asn (N), Gln (Q), Cys (C), Gly (G), Pro (P), Ala (A), Ile (I), Leu (L), Met (M), Phe (F), Trp (W), Tyr (Y) and Val (V).
[0123] An amino acid or segment in one sequence that "corresponds to" an amino acid or segment in another sequence is one that aligns with the other amino acid or segment using a standard sequence alignment program such as ALIGN, ClustalW or similar, typically at default settings. Hence a standard sequence alignment program can be used to identify which amino acid in an e.g. immunoglobulin sequence corresponds to a specific amino acid in e.g. human IgG1. Further a standard sequence alignment program can be used to identify sequence identity e.g. a sequence identity to SEQ ID NO:1 of at least 80%, or 85%, 90%, or at least 95%. For example, the sequence alignments shown in FIG. 1 can be used to identify any amino acid in the Fc region of one IgG1 allotype that corresponds to a particular amino acid in another allotype of an IgG1 Fc sequence.
[0124] The term "vector," as used herein, refers to a nucleic acid molecule capable of inducing transcription of a nucleic acid segment ligated into the vector. One type of vector is a "plasmid", which is in the form of a circular double stranded DNA loop. Another type of vector is a viral vector, wherein the nucleic acid segment may be ligated into the viral genome. Certain vectors are capable of autonomous replication in a host cell into which they are introduced (for instance bacterial vectors having a bacterial origin of replication and episomal mammalian vectors). Other vectors (such as non-episomal mammalian vectors) may be integrated into the genome of a host cell upon introduction into the host cell, and thereby are replicated along with the host genome. Moreover, certain vectors are capable of directing the expression of genes to which they are operatively linked. Such vectors are referred to herein as "recombinant expression vectors" (or simply, "expression vectors"). In general, expression vectors of utility in recombinant DNA techniques are often in the form of plasmids. In the present specification, "plasmid" and "vector" may be used interchangeably as the plasmid is the most commonly used form of vector. However, the present invention is intended to include such other forms of expression vectors, such as viral vectors (such as replication defective retroviruses, adenoviruses and adeno-associated viruses), which serve equivalent functions.
[0125] The term "recombinant host cell" (or simply "host cell"), as used herein, is intended to refer to a cell into which an expression vector has been introduced. It should be understood that such terms are intended to refer not only to the particular subject cell, but also to the progeny of such a cell. Because certain modifications may occur in succeeding generations due to either mutation or environmental influences, such progeny may not, in fact, be identical to the parent cell, but are still included within the scope of the term "host cell" as used herein. Recombinant host cells include, for example, transfectomas, such as CHO-S cells, HEK-293F cells, Expi293F cells, PER.C6, NS0 cells, and lymphocytic cells, and prokaryotic cells such as E. coli and other eukaryotic hosts such as plant cells and fungi, as well as prokaryotic cells such as E. coli.
Specific Embodiments of the Invention
[0126] The present invention is based, at least in part, on the discovery that the ability of antibodies targeting members of the TNFR-SF comprising an intracellular death domain, such as an anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibody to induce cell death in a target cell expressing FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR, or NGFR can be greatly enhanced by introducing a mutation at an amino acid in the Fc region corresponding to amino acid position E430, E345 or S440 in human IgG1 according to EU numbering. The invention is further based on the surprising finding that a combination of two antibodies binding to a first and a second epitope on FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR, or NGFR and each comprising a mutation in the Fc region show superior induction of cell death in a target cell compared to a combination of the two antibodies without the mutation in the Fc region.
[0127] In one aspect the present invention relates to an antibody comprising an Fc region of a human immunoglobulin IgG and an antigen binding region binding to FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR, or NGFR, wherein the Fc region comprises a mutation at an amino acid position corresponding to E430, E345 or S440 in human IgG1 according to EU numbering.
[0128] The positions corresponding to E430, E345 and S440 in human IgG1 according to EU numbering are located in the CH3 domain of the Fc region.
[0129] By introducing specific mutations in the Fc domain corresponding to at least one of the following positions E430, E345 and S440 in human IgG1 oligomerization such as hexamerization upon target binding on the cell surface is enhanced, while the antibody molecules remain monomeric in solution (WO2013/004842; WO2014/108198). The hexamerization enhancing mutation strengthens Fc-Fc interactions between neighbouring IgG antibodies that are bound to a cell surface target, resulting in enhanced hexamer formation of the target-bound antibodies.
[0130] In one embodiment of the present invention the Fc region of the anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR, and anti-NGFR antibody comprises a mutation corresponding to E430G, E430S, E430F, E430T, E345K, E345Q, E345R, E345Y, S440Y or S440W in human IgG1, EU numbering. Hereby are embodiments provided that allow for enhanced hexamerization of antibodies upon target binding on a cell surface.
[0131] In one embodiment of the present invention the anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR, and anti-NGFR antibody comprises a mutation at an amino acid position corresponding to E430 in human IgG1 according to EU numbering, wherein the mutation is selected from the group consisting of: E430G, E430S, E430F and E430T.
[0132] In one embodiment of the present invention the anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR, and anti-NGFR antibody comprises an E430G mutation in the Fc region.
[0133] In a preferred embodiment of the present invention the Fc region comprises a mutation corresponding to E430G or E345K in human IgG1 EU numbering.
[0134] In one embodiment of the present invention the anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR, and anti-NGFR antibody comprises a mutation at an amino acid position corresponding to E345 in human IgG1 according to EU numbering, wherein the mutation is selected from the group consisting of: E345K, E345Q, E345R and E345Y. In one embodiment of the present invention the anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR, and anti-NGFR antibody comprises an E345K mutation in the Fc region.
[0135] In one embodiment of the present invention the Fc region comprises a mutation at an amino acid position corresponding to position S440 in human IgG1, EU numbering where the mutation is S440Y or S440W.
[0136] In one embodiment of the present invention the anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR, and anti-NGFR antibody comprises a mutation at an amino acid position corresponding to S440 in human IgG1 according to EU numbering, wherein the mutation is selected from the group consisting of: S440Y and S440W.
[0137] In one embodiment of the present invention the Fc region comprises a mutation corresponding to S440Y. In one embodiment of the present invention the Fc region comprises a mutation corresponding to E430G. In one embodiment of the present invention the Fc region comprises a mutation corresponding to E345K.
[0138] In one embodiment of the present invention the Fc region comprises at least a first and a second mutation at an amino acid position corresponding to E430 and E345 in human IgG1, EU numbering.
[0139] In one embodiment of the present invention the Fc region further comprises a third mutation at an amino acid position selected form the group consisting of: Y436 and S440. Hereby embodiments are provided comprising a first, second and third mutations which allows for enhanced Fc-Fc interactions in solution.
[0140] In one embodiment of the invention the antibody comprises a first, second and third mutation at an amino acid position corresponding to E430, E345 and S440 in human IgG1, EU numbering.
[0141] In one embodiment of the invention the antibody comprises an Fc region wherein the first, second and third mutation at amino acid positions corresponding to E430, E345 and S440 in human IgG1, EU numbering, wherein the mutations are E430G, E345R, S440Y.
[0142] In one embodiment of the invention the antibody comprises a first, second and third mutation at an amino acid position corresponding to E430, E345 and Y436 in human IgG1, EU numbering.
[0143] In one embodiment of the invention the antibody comprises an Fc region wherein the first, second and third mutation at amino acid positions corresponding to E430, E345 and Y436 in human IgG1, EU numbering, wherein the mutations are E430G, E345R, Y436I.
[0144] In one embodiment of the present invention the Fc region comprises a mutation at an amino acid position corresponding to E430 and/or E345 and wherein the said Fc region comprises a further mutation at an amino acid position corresponding to S440, with the proviso that the mutation is not S440Y or S440W.
[0145] In one embodiment of the present invention the antibody comprises a further mutation at an amino acid position corresponding to one of the following positions S440 or K439 in human IgG1, EU numbering. In one embodiment of the invention the Fc region comprises a further mutation in a position corresponding to S440 or K439, with the proviso that the further mutation is not in position S440 if the hexamerization enhancing mutation is in S440. In one embodiment of the present invention the further mutation at an amino acid position corresponding to one of the following positions S440 or K439 may be a hexamerization-inhibiting mutation.
[0146] In one embodiment the Fc region comprises a further mutation at an amino acid position corresponding to K439 in human IgG1, EU numbering, wherein the further mutation is selected from the group consisting of: K439E and K439D. In on embodiment the further mutation is K439E.
[0147] In on embodiment the Fc region comprises a further mutation at an amino acid position corresponding to S440 in human IgG1, EU numbering, wherein the further mutation is selected from the group consisting of: S440K, S440R and S440H. In on embodiment the further mutation is S440K.
[0148] In one embodiment of the present invention the Fc region comprises a further hexamerization-inhibiting mutation such as K439E or S440K in human IgG1, EU numbering. The hexamerization-inhibiting mutation such as K439E or S440K prevents Fc-Fc interaction with antibodies comprising the same hexamerization inhibiting mutation, but by combining antibodies with a K439E mutation and antibodies with a S440K mutation the inhibiting effect is neutralized and Fc-Fc interactions is restored.
[0149] Antibodies comprising a mutation in a position corresponding to E430, E345 or S440 according to the present invention and a further mutation at an amino acid position corresponding to K439 such as a K439E mutation do not form oligomers with antibodies comprising a further mutation at an amino acid position corresponding to K439 such as a K439E mutation. However, antibodies comprising hexamerization enhancing mutation in E430, E345 or S440 and a further mutation in K439 such a K439E do form oligomers with antibodies comprising a hexamerization enhancing mutation in E430 or E345 and a further mutation in S440 such as S440K. Antibodies comprising a mutation in a position corresponding to E430 or E345 according to the present invention and a further mutation at an amino acid position corresponding to S440 such as a S440K mutation do not form oligomers with antibodies comprising a further mutation at an amino acid position corresponding to S440 such as a S440K mutation. However, antibodies comprising hexamerization enhancing mutation in E430 or E345 and a further mutation in S440 such a S440K do form oligomers with antibodies comprising a hexamerization enhancing mutation in E430 or E345 and a further mutation in K439 such as K439. In one embodiment of the present invention the Fc region comprises a hexamerization enhancing mutation such as E430G and a hexamerization inhibiting mutation such as K439E. In one embodiment of the present invention the Fc region comprises a hexamerization enhancing mutation such as E345K and a hexamerization inhibiting mutation such as K439E. In another embodiment of the present invention the Fc region comprises a hexamerization enhancing mutation such as E430G and a hexamerization inhibiting mutation such as S440K. In one embodiment of the present invention the Fc region comprises a hexamerization enhancing mutation such as E345K and a hexamerization inhibiting mutation such as S440K. In one embodiment of the present invention the Fc region comprises a hexamerization enhancing mutation such as S440Y and a hexamerization inhibiting mutation such as K439E Hereby embodiments are provided that allow for exclusive hexamerization between combinations of antibodies comprising a K439E mutation and antibodies comprising a S440K mutation.
[0150] In on embodiment the Fc region comprises a further mutation, wherein the further mutation is selected from the group consisting of: K439E and K439D. In on embodiment the further mutation is K439E.
[0151] In one embodiment the Fc region comprises a further mutation, wherein the further mutation is selected from the group consisting of: S440K, S440R and S440H. In on embodiment the further mutation is S440K.
[0152] The human FAS molecule is comprised of 335 amino acids in including the signaling peptide at the first 1-25 positions, followed by the extracellular domain at positions 26-173, a transmembrane domain at positions 174-190 and a cytoplasmic domain at positions 191-335. The extracellular domain is comprised of a 148 amino acid sequence.
[0153] In one embodiment the member of the death receptor comprising an intracellular death domain is FAS.
[0154] In one embodiment of the invention the anti-FAS antibody comprises an antigen binding region binding to an epitope within the extracellular domain of FAS.
[0155] In one embodiment of the present invention the anti-FAS antibody comprise an antigen binding region comprising a variable heavy chain (VH) region and a variable light chain (VL) region comprising the amino acid sequence of: (VH) SEQ ID NO 15: and (VL) SEQ ID NO:16.
[0156] In one embodiment of the invention the anti-FAS antibody comprises an Fc region comprising a mutation corresponding to E430G or E345K in human IgG1 EU numbering.
[0157] In one embodiment of the invention the anti-FAS antibody comprise a first, second and third mutation at an amino acid position corresponding to E430, E345 and S440 in human IgG1, EU numbering.
[0158] In one embodiment of the invention the anti-FAS antibody comprises an Fc region wherein the first, second and third mutation at amino acid positions corresponding to E430, E345 and S440 in human IgG1, EU numbering, wherein the mutations are E430G, E345R, S440Y.
[0159] In one embodiment of the invention the anti-FAS antibody comprises an Fc region wherein the first, second and third mutation at amino acid positions corresponding to E430, E345 and S440 in human IgG1, EU numbering, wherein the mutations are first, second a third E430G, E345R, S440Y and a further S440K mutation.
[0160] In one embodiment of the invention the anti-FAS antibody comprises a first, second and third mutation at an amino acid position corresponding to E430, E345 and Y436 in human IgG1, EU numbering.
[0161] In one embodiment of the invention the anti-FAS antibody comprises an Fc region wherein the first, second and third mutation at amino acid positions corresponding to E430, E345 and Y436 in human IgG1, EU numbering, wherein the mutations are E430G, E345R, Y436I.
[0162] In one embodiment of the invention the anti-FAS antibody comprises an Fc region wherein the first, second and third mutation at amino acid positions corresponding to E430, E345 and Y436 in human IgG1, EU numbering, wherein the first, second a third mutations are E430G, E345R, Y436I and a further S440K mutation.
[0163] The human TNFR1 molecule is comprise of 455 amino acids in including the signaling peptide at the first 1-21 positions, followed by the extracellular domain at positions 22-211, a transmembrane domain at positions 212-234 and a cytoplasmic domain at positions 235-455. The extracellular domain is comprised of a 190 amino acid sequence.
[0164] In one embodiment of the invention the anti-TNFR1 antibody comprises an antigen binding region binding to an epitope within the extracellular domain of TNFR1.
[0165] In one embodiment of the invention the anti-TNFR1 antibody comprises a Fc region comprising a mutation corresponding to E430G or E345K in human IgG1 EU numbering.
[0166] The human EDAR molecule is comprise of 448 amino acids in including the signaling peptide at the first 1-26 positions, followed by the extracellular domain at positions 27-187, a transmembrane domain at positions 188-208 and a cytoplasmic domain at positions 209-448. The extracellular domain is comprised of a 161 amino acid sequence.
[0167] In one embodiment of the invention the anti-EDAR antibody comprises an antigen binding region binding to an epitope within the extracellular domain of EDAR.
[0168] In one embodiment of the invention the anti-EDAR antibody comprises a Fc region comprising a mutation corresponding to E430G or E345K in human IgG1 EU numbering.
[0169] The human NGFR molecule is comprise of 427 amino acids in including the signaling peptide at the first 1-28 positions, followed by the extracellular domain at positions 29-250, a transmembrane domain at positions 251-272 and a cytoplasmic domain at positions 273-427. The extracellular domain is comprised of a 222 amino acid sequence.
[0170] In one embodiment of the invention the anti-NGFR antibody comprises an antigen binding region binding to an epitope within the extracellular domain of NGFR.
[0171] In one embodiment of the invention the anti-NGFR antibody comprises a Fc region comprising a mutation corresponding to E430G or E345K in human IgG1 EU numbering.
[0172] The human DR3 molecule is comprise of 417 amino acids including the signaling peptide at the first 1-24 positions, followed by the extracellular domain at positions 25-199, a transmembrane domain at positions 200-220 and a cytoplasmic domain at positions 221-417. The extracellular domain is comprised of a 175 amino acid sequence.
[0173] In one embodiment of the invention the anti-DR3 antibody comprises an antigen binding region binding to an epitope within the extracellular domain of DR3.
[0174] In one embodiment of the invention the anti-DR3 antibody comprises a Fc region comprising a mutation corresponding to E430G or E345K in human IgG1 EU numbering.
[0175] The human DR4 molecule is comprise of 468 amino acids including the signaling peptide at the first 1-23 positions, followed by the extracellular domain at positions 24-239, a transmembrane domain at positions 240-262 and a cytoplasmic domain at positions 263-468. The extracellular domain is comprised of a 216 amino acid sequence.
[0176] In one embodiment of the invention the anti-DR4 antibody comprises an antigen binding region binding to an epitope within the extracellular domain of DR4.
[0177] In one embodiment of the invention the member of the death receptor comprising an intracellular death domain is DR4.
[0178] In one embodiment of the present invention the anti-DR4 antibody comprise an antigen binding region comprising a variable heavy chain (VH) region and a variable light chain (VL) region comprising the amino acid sequence of: (VH) SEQ ID NO 13: and (VL) SEQ ID NO:14.
[0179] In one embodiment of the invention the anti-DR4 antibody comprises a mutation at an amino acid position corresponding to E430 in human IgG1, EU numbering, wherein the mutation is selected from the group consisting of: E430G, E430S, E40F and E430T. In one embodiment of the invention the anti-DR4 antibody comprises an E430G mutation.
[0180] In one embodiment of the invention the anti-DR4 antibody comprises a mutation at an amino acid position corresponding to E345 in human IgG1, EU numbering, wherein the mutation is selected from the group consisting of: E345K E345Q, E345R and E345Y. In one embodiment of the invention the anti-DR4 antibody comprises an E345K mutation.
[0181] In one embodiment of the invention the anti-DR4 antibody comprises an Fc region comprising a mutation corresponding to E430G or E345K in human IgG1 EU numbering.
[0182] The human DR5 molecule is comprised of 440 amino acids including a signaling peptide at the first 1-55 positions, followed by the extracellular domain at positions 56-210, a transmembrane domain at positions 211-231 and a cytoplasmic domain at positions 232-440. The extracellular domain is comprised of a 155 amino acid sequence. The isoform short of DR5 is missing 185-213 from the extracellular domain.
[0183] In one embodiment of the invention the anti-DR5 antibody comprises an antigen binding region binding to an epitope within the extracellular domain of DR5.
[0184] In one embodiment of the present invention the anti-DR5 antibody comprise an antigen binding region comprising a variable heavy chain (VH) region and a variable light chain (VL) region comprising the amino acid sequence from the group consisting of:
a) (VH) SEQ ID NO 19: and (VL) SEQ ID NO:23,
b) (VH) SEQ ID NO 26: and (VL) SEQ ID NO:23,
c) (VH) SEQ ID NO 31: and (VL) SEQ ID NO:35 and
d) (VH) SEQ ID NO 40: and (VL) SEQ ID NO:43.
[0185] In one embodiment of the invention the anti-DR5 antibody comprises a mutation at an amino acid position corresponding to E430 in human IgG1, EU numbering, wherein the mutation is selected from the group consisting of: E430G, E430S, E40F and E430T. In one embodiment of the invention the anti-DR5 antibody comprises an E430G mutation.
[0186] In one embodiment of the invention the anti-DR5 antibody comprises a mutation at an amino acid position corresponding to E345 in human IgG1, EU numbering, wherein the mutation is selected from the group consisting of: E345K E345Q, E345R and E345Y. In one embodiment of the invention the anti-DR5 antibody comprises an E345K mutation.
[0187] In one embodiment of the invention the anti-DR5 antibody comprises a Fc region comprising a mutation corresponding to E430G or E345K in human IgG1 EU numbering.
[0188] In one embodiment of the invention the antibody comprises an antigen binding region binding to the same binding site as TRAIL or a binding site overlapping with the binding site of TRAIL. The TRAIL binding motif is located in CRD2 and CRD3 based on a Crystal structure of TRAIL in complex with the DR5 ectodomain (Hymowitz et al., Mol Cell. 1999 October; 4(4):563-71) That is, in one embodiment of the invention the antibody comprises an antigen binding region binding to the same binding region on DR5 as TRAIL. In one embodiment of the invention the antibody comprises an antigen binding region that competes with TRAIL binding to DR5. In one embodiment of the invention the antibody blocks TRAIL induced mediated killing such as TRAIL induced apoptosis.
[0189] In another embodiment of the invention the antibody comprises an antigen binding region binding to an epitope on DR5 that is different from the binding site of TRAIL. In one embodiment of the invention the antibody comprises an antigen binding region binding to a different binding region on DR5 than TRAIL. In one embodiment of the invention the antibody does not block TRAIL induced mediated killing such as TRAIL induced apoptosis.
[0190] The human DR6 molecule is comprise of 655 amino acids in including the signaling peptide at the first 1-41 positions, followed by the extracellular domain at positions 42-349, a transmembrane domain at positions 350-370 and a cytoplasmic domain at positions 371-655. The extracellular domain is comprised of a 308 amino acid sequence.
[0191] In one embodiment of the invention the anti-DR6 antibody comprises an antigen binding region binding to an epitope within the extracellular domain of DR6.
[0192] In one embodiment of the invention the anti-DR6 antibody comprises a Fc region comprising a mutation corresponding to E430G or E345K in human IgG1 EU numbering.
[0193] In one embodiment of the present invention the antibody is a monoclonal antibody. In one embodiment of the present invention the antibody is of the IgG1, IgG2, IgG3, IgG4, IgA, IgE, IgD or IgM isotype.
[0194] In a preferred embodiment of the invention the antibody is an IgG1 antibody.
[0195] In one embodiment of the present invention the antibody is an IgG1m(f), IgG1m(z), IgG1m(a) or an IgG1m(x) allotype, or any allotype combination, such as IgG1m(z,a), IgG1m(z,a,x), IgG1m(f,a).
[0196] In one embodiment the antibody is a human antibody, a chimeric antibody or a humanized antibody.
[0197] In one embodiment of the present invention the anti-Death Receptor antibody selected from the group consisting of: anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR and anti-NGFR is agonistic. That the antibody is agonistic is to be understood as that the antibody clusters, stimulates or activates the Death Receptor to which it bind at least as good as the effect found by interaction between the Death Receptor and the natural ligand binding to the Death Receptor, or by overexpression of the Death Receptor. An agonistic anti-FAS antibody of the present invention bound to FAS activates the same intracellular pathways as FAS-Ligand bound to FAS. An agonistic anti-FAS antibody of the present invention is able to induce apoptosis in a cell expressing FAS.
[0198] An agonistic anti-DR4 antibody of the present invention bound to DR4 activates the same intracellular pathways as TRAIL bound to DR4.
[0199] An agonistic anti-DR5 antibody of the present invention bound to DR5 activates the same intracellular pathways as TRAIL bound to DR5.
[0200] An agonistic anti-TNFR1 antibody of the present invention bound to TNFR1 activates the same intracellular pathways as LT.alpha. or TNF bound to TNFR1.
[0201] An agonistic anti-DR6 antibody of the present invention bound to DR6 activates the same intracellular pathways as DR6 overexpression or APP bound to DR6.
[0202] An agonistic anti-DR3 antibody of the present invention bound to DR3 activates the same intracellular pathways as TWEAK bound to DR3.
[0203] An agonistic anti-EDAR antibody of the present invention bound to EDAR activates the same intracellular pathways as ectodysplasin A bound to EDAR.
[0204] An agonistic anti-NGFR antibody of the present invention bound to NGFR activates the same intracellular pathways as NGF bound to NGFR.
[0205] In one embodiment of the present invention the anti-Death Receptor antibody, such as anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibody has enhanced agonistic activity. That the anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibody has enhanced agonistic activity is to be understood as the antibody is able to cluster the FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR, or NGFR receptor or activate the same intracellular pathways as the natural ligand bound to FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR, or NGFR, but at an enhanced level. That is an anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibody of the invention, i.e having a mutation in the Fc region according to the invention, with enhanced agonistic activity is able to induce increased level of apoptosis or programmed cell death in a cell or tissue expressing the FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR, or NGFR receptor compared to the natural ligand or the same antibody without said mutation binding to the receptor.
[0206] Thus it is to be understood in the context of the present invention that the enhanced agonistic activity of an antibody according to the invention i.e. comprising an amino acid mutation at a position corresponding to E430, E345 or S440 in human IgG1, EU numbering, may be evaluated by comparing the antibody according to the invention with the same antibody without said mutation. In the context of the present invention the same antibody is to be understood as an antibody having the identical amino acid sequence as the antibody according to the invention, but without said mutation.
[0207] In one embodiment of the present invention the anti-Death Receptor receptor antibody, such as anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibody induces programmed cell death in a target cell. In one embodiment of the present invention the anti-DR5 antibody induces caspase-dependent cell death. Caspase-dependent cell death may be induced by activation of caspase-3 and/or caspase-7. In one embodiment of the present invention the antibody induces apoptosis.
[0208] In one embodiment of the present invention the anti-Death Receptor antibody, such as anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibody induces phosphatidylserine (PS) exposure, which can be measured by Annexin-V binding. Therefore, Annexin-V binding correlates to programmed cell death and can be used to measure the anti-Death Receptor antibody, such as anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibody's ability to induce cellular events leading to programmed cell death.
[0209] In a preferred embodiment of the present invention the anti-Death Receptor antibody, such as anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibody induces apoptosis in a target cell expressing the Death Receptor such as FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR, or NGFR, such as a tumor cell.
[0210] In one embodiment of the invention the anti-Death Receptor antibody, such as anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibody reduces cell viability.
[0211] In one embodiment the present invention the anti-Death Receptor antibody, such as anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibody induces clustering of the Death Receptor such as FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR, or NGFR. That the antibody can induce clustering and even enhance clustering leads to activation of the same intracellular signaling pathways as the natural ligand bound to one of the following group of targets FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR, or NGFR.
[0212] In one embodiment the antibodies or compositions of the present invention induce, trigger, increase or enhance apoptosis or cell death in cancer cells or cancer tissues expressing one or more Death Receptors such as FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR and/or NGFR. The increased or enhanced apoptosis or cell death can be measured by an increase or enhanced level of phosphatidylserine exposure on cells exposed to or treated with one or more anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR and/or anti-NGFR antibodies of the invention. Thus it is to be understood in the context of the present invention that induced, triggered, increased or enhanced apoptosis or cell death of an antibody according to the invention i.e. comprising an amino acid mutation at a position corresponding to E430, E345 or S440 in human IgG1, EU numbering, may be evaluated by comparing the antibody according to the invention with the same antibody without said mutation. In the context of the present invention the same antibody is to be understood as an antibody having the identical amino acid sequence as the antibody according to the invention, but without said mutation.
[0213] Alternatively, the increase or enhanced apoptosis or cell death can be measured by measuring activation of caspase 3 or caspase 7 in cells that have been exposed to or treated with one or more anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR and/or anti-NGFR antibodies of the invention. Alternatively, the increase or enhanced apoptosis or cell death can be measured by a loss of viability in cell cultures that have been exposed to or treated with one or more anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR and/or anti-NGFR antibodies of the invention, compared to untreated cell cultures, in which the loss of viability can be inhibited by a caspase-inhibitor, for example ZVAD.
[0214] In one embodiment of the present invention the anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibody induces hexamerization of antibodies on target cells expressing FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR.
Bispecific Antibodies
[0215] In another aspect, the present invention relates to a bispecific antibody comprising at least one antigen binding region binding a death receptor e.g. FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR as described herein.
[0216] In another aspect, the present invention comprises a bispecific antibody comprising one or more antigen binding regions binding a death receptor e.g. FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR as described herein.
[0217] In one embodiment of the invention the bispecific antibody comprises a first antigen binding region and a second antigen binding region binding a death receptor as defined herein.
[0218] In one embodiment of the invention the bispecific antibody comprises a first and a second antigen binding region, wherein said first antigen binding region and said second antigen binding region bind different epitopes on the same death receptor.
[0219] In one embodiment of the present invention the bispecific antibody comprises a first and a second Fc region, wherein the first and/or second Fc region comprises a mutation of an amino acid position corresponding to E430, E345 or S440 in human IgG1, EU numbering according to the invention. In one embodiment of the present invention the bispecific anti-DR5 antibody comprises a first and a second Fc region, wherein the first and second Fc region comprises a mutation of an amino acid position corresponding to E430, E345 or S440 in human IgG1, EU numbering. In one embodiment of the present invention the bispecific antibody comprises a first and a second Fc region, wherein the first Fc region comprises a mutation of an amino acid position corresponding to E430, E345 or S440 in human IgG1, EU numbering. In one embodiment of the present invention the bispecific antibody comprises a first and a second Fc region, wherein the second Fc region comprises a mutation of an amino acid position corresponding to E430, E345 or S440 in human IgG1, EU numbering.
[0220] In one embodiment of the invention the bispecific antibody comprises a first and a second antigen binding region, wherein said first antigen binding region binding to a death receptor selected from the following group FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR and NGFR does not block binding of said second antigen binding region binding a death receptor selected from the following group FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR and NGFR, wherein the first and the second antigen binding region does not bind to the same death receptor.
[0221] In a particular embodiment the antibody may be bispecific antibody such as the heterodimeric protein described in WO 11/131746, which is hereby incorporated herein by reference.
[0222] In one embodiment, the antibody is a bispecific antibody which comprises a first heavy chain comprising a first Fc region of an immunoglobulin and a first antigen-binding region, and a second heavy chain comprising a second Fc region of an immunoglobulin and a second antigen-binding region, wherein the first and second antigen-binding regions bind different epitopes on the same antigen or on different antigens.
[0223] In a further embodiment said first heavy chain comprising a first Fc region comprises a further amino acid substitution at a position selected from those corresponding to K409, T366, L368, K370, D399, F405, and Y407 in the Fc region of a human IgG1 heavy chain; and wherein said second heavy chain comprising a second Fc region comprises a further amino acid substitution at a position selected from those corresponding to F405, T366, L368, K370, D399, Y407, and K409 in the Fc region of a human IgG1 heavy chain, and wherein said further amino acid substitution in the first heavy chain comprising a first Fc region is different from the said further amino acid substitution in the second heavy chain comprising a second Fc region.
[0224] In a further embodiment said first heavy chain comprising a first Fc region comprises an amino acid substitution at a position corresponding to K409 in the Fc-region of a human IgG1 heavy chain; and said second heavy chain comprising a second Fc region comprises an amino acid substitution at a position corresponding to F405 in the Fc-region of a human IgG1 heavy chain.
[0225] In one embodiment of the invention the bispecific antibody comprises introducing a first and second Fc region comprising a mutation in at least one amino acid residue selected from those corresponding to E345, E430, S440, Q386, P247, I253, S254, Q311, D/E356, T359, E382, Y436, and K447 in the Fc-region of a human IgG1 heavy chain, with the proviso that the mutation in S440 is S440Y or S440W.
[0226] In a further embodiment the mutation in the first and second Fc region in at least one amino acid residue selected from those corresponding to E345, E430, S440, Q386, P247, I253, S254, Q311, D/E356, T359, E382, Y436, and K447 in the Fc-region of a human IgG1 heavy chain, with the proviso that the mutation in S440 is S440Y or S440W, may be in the same amino acid residue position or a different position. In a further embodiment it may be the same or a different mutation in the same amino acid residue position in the first and second Fc region.
[0227] In another embodiment the bispecific antibody comprises a first or second CH2-CH3 region comprising a mutation in at least one amino acid residue selected from those corresponding to E345, E430, S440, Q386, P247, I253, S254, Q311, D/E356, T359, E382, Y436, and K447 in the Fc-region of a human IgG1 heavy chain, with the proviso that the mutation in S440 is S440Y or S440W.
[0228] In one embodiment of the invention the bispecific antibody comprises a first and a second heavy chain, wherein said first heavy chain comprises a mutation corresponding to F405L in human IgG1 according to EU numbering and said second heavy chain comprises a mutation corresponding to K409R in human IgG1 according EU numbering.
[0229] In one embodiment of the invention the bispecific antibody is comprised in a pharmaceutical composition.
Anti-Death Receptor Antibody Compositions
[0230] The anti-death receptor antibodies i.e. anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibodies such as monoclonal antibodies or bispecific antibodies according to any aspect or embodiment of the present invention may be comprised in a composition, such as a pharmaceutical composition, diagnostic composition or any other composition.
[0231] In one aspect the invention relates to a composition comprising at least one anti-death receptor antibody according to any one of the embodiments described herein.
[0232] In one aspect the invention relates to a composition comprising one or more anti-death receptor antibodies according to any one the embodiments described herein. The composition may comprise one, two or more anti-death domain receptor antibodies according to the invention as described herein that are not identical, such as a combination of two different monoclonal anti-death domain receptor antibodies.
[0233] In one embodiment of the present invention the composition comprises a first anti-death receptor antibody and a second anti-death receptor antibody as described herein. That is in one embodiment of the present invention the composition comprises a first antibody as described herein and a second antibody as described herein, wherein the first and the second antibody are not identical. That is in one embodiment of the present invention the composition comprises a first antibody selected from the group consisting of: anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR and anti-NGFR antibody as described herein and a second antibody selected from the group consisting of: anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR and anti-NGFR antibody, wherein the first and the second antibody do not bind to the same antigen or epitope. Hereby antibody compositions are described wherein the first and second antibody is not identical.
[0234] In one embodiment of the present invention the composition comprises a first anti-death receptor antibody and a second anti-death receptor antibody as described herein i.e. having a mutation in an amino acid corresponding to position E430, E345 or S440 in human IgG1, EU numbering.
[0235] In one embodiment of the present invention the mutation in an amino acid corresponding to position E430 in human IgG1, EU numbering is selected from the group consisting of: E430G, E430S, E430F and E430T.
[0236] In one embodiment of the present invention the mutation in an amino acid corresponding to position E345 in human IgG1, EU numbering is selected from the group consisting of: E345K, E345Q, E345R and E345Y.
[0237] In one embodiment of the present invention the mutation in an amino acid corresponding to position S440 in human IgG1, EU numbering is selected from the group consisting of: S440W and S440Y
[0238] In one embodiment of the present invention the composition comprises a first anti-FAS antibody comprising a mutation at an amino acid position corresponding to E430, E345 or S440 in human IgG1, EU numbering and a second antibody comprising a mutation at an amino acid position corresponding to E430, E345 or S440 in human IgG1, EU numbering, wherein the second antibody is selected from the group consisting of:
a) anti-DR4 antibody; b) anti-DR5 antibody; c) anti-TNFR1 antibody; d) anti-DR6 antibody; c) anti-DR3 antibody; d) anti-EDAR antibody and e) anti-NGFR antibody.
[0239] In one embodiment of the present invention the first and second antibody comprises a mutation at an amino acid position, wherein said amino acid position is the same. In one embodiment of the present invention the first and second antibody comprises a mutation at an amino acid position, wherein said amino acid position is not the same. Thus in one embodiment of the present invention the first and antibody comprises a mutation at an amino acid position, wherein said amino acid position in said first and second antibody is different.
[0240] In one embodiment of the present invention the composition comprises a first anti-FAS antibody comprising a mutation at an amino acid position corresponding to E430 in human IgG1, EU numbering, and a second antibody comprising a mutation at an amino acid position corresponding to E430, in human IgG1, EU numbering, wherein the second antibody is selected from the group consisting of:
a) anti-DR4 antibody; b) anti-DR5 antibody; c) anti-TNFR1 antibody; d) anti-DR6 antibody; c) anti-DR3 antibody; d) anti-EDAR antibody and e) anti-NGFR antibody.
[0241] In one embodiment of the present invention the composition comprises a first anti-FAS antibody comprising a E430G mutation and a second antibody comprising a E430G mutation, wherein the second antibody is selected from the group consisting of:
a) anti-DR4 antibody; b) anti-DR5 antibody; c) anti-TNFR1 antibody; d) anti-DR6 antibody; c) anti-DR3 antibody; d) anti-EDAR antibody and e] anti-NGFR antibody.
[0242] In one embodiment of the present invention the composition comprises a first anti-FAS antibody comprising a mutation at an amino acid position corresponding to E345 in human IgG1, EU numbering, and a second antibody comprising a mutation at an amino acid position corresponding to E345, in human IgG1, EU numbering, wherein the second antibody is selected from the group consisting of:
a) anti-DR4 antibody; b) anti-DR5 antibody; c) anti-TNFR1 antibody; d) anti-DR6 antibody; c) anti-DR3 antibody; d) anti-EDAR antibody and e) anti-NGFR antibody.
[0243] In one embodiment of the present invention the composition comprises a first anti-FAS antibody comprising a E345K mutation in the Fc region, and a second antibody comprising a E345K mutation in the Fc region, wherein the second, wherein the second antibody is selected from the group consisting of:
a) anti-DR4 antibody; b) anti-DR5 antibody; c) anti-TNFR1 antibody; d) anti-DR6 antibody; c) anti-DR3 antibody; d) anti-EDAR antibody and e) anti-NGFR antibody.
[0244] In one embodiment of the present invention the composition comprises a first anti-DR4 antibody comprising a mutation at an amino acid position corresponding to E430, E345 or S440 in human IgG1, EU numbering and a second antibody comprising a mutation at an amino acid position corresponding to E430, E345 or S440 in human IgG1, EU numbering, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR5 antibody; c) anti-TNFR1 antibody; d) anti-DR6 antibody; c) anti-DR3 antibody; d) anti-EDAR antibody and e) anti-NGFR antibody.
[0245] In one embodiment of the present invention the composition comprises a first anti-DR4 antibody comprising a mutation at an amino acid position corresponding to E430 in human IgG1, EU numbering, and a second antibody comprising a mutation at an amino acid position corresponding to E430, in human IgG1, EU numbering, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR5 antibody; c) anti-TNFR1 antibody; d) anti-DR6 antibody; c) anti-DR3 antibody; d) anti-EDAR antibody and e) anti-NGFR antibody.
[0246] In one embodiment of the present invention the composition comprises a first anti-DR4 antibody comprising a mutation at an amino acid position corresponding to E345 in human IgG1, EU numbering, and a second antibody comprising a mutation at an amino acid position corresponding to E345, in human IgG1, EU numbering, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR5 antibody; c) anti-TNFR1 antibody; d) anti-DR6 antibody; c) anti-DR3 antibody; d) anti-EDAR antibody and e) anti-NGFR antibody.
[0247] In one embodiment of the present invention the composition comprises a first anti-DR4 antibody comprising an E345K mutation in the Fc region and a second antibody comprising an E345K mutation in the Fc region, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR5 antibody; c) anti-TNFR1 antibody; d) anti-DR6 antibody; c) anti-DR3 antibody; d) anti-EDAR antibody and e) anti-NGFR antibody.
[0248] In one embodiment of the present invention the composition comprises a first anti-DR5 antibody comprising a mutation at an amino acid position corresponding to E430, E345 or S440 in human IgG1, EU numbering and a second antibody comprising a mutation at an amino acid position corresponding to E430, E345 or S440 in human IgG1, EU numbering, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR4 antibody; c) anti-TNFR1 antibody; d) anti-DR6 antibody; c) anti-DR3 antibody; d) anti-EDAR antibody and e) anti-NGFR antibody.
[0249] In one embodiment of the present invention the composition comprises a first anti-DR5 antibody comprising a mutation at an amino acid position corresponding to E430 in human IgG1, EU numbering, and a second antibody comprising a mutation at an amino acid position corresponding to E430, in human IgG1, EU numbering, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR4 antibody; c) anti-TNFR1 antibody; d) anti-DR6 antibody; c) anti-DR3 antibody; d) anti-EDAR antibody and e) anti-NGFR antibody.
[0250] In one embodiment of the present invention the composition comprises a first anti-DR5 antibody comprising a mutation at an amino acid position corresponding to E345 in human IgG1, EU numbering, and a second antibody comprising a mutation at an amino acid position corresponding to E345, in human IgG1, EU numbering, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR4 antibody; c) anti-TNFR1 antibody; d) anti-DR6 antibody; c) anti-DR3 antibody; d) anti-EDAR antibody and e) anti-NGFR antibody.
[0251] In one embodiment of the present invention the composition comprises a first anti-DR5 antibody comprising an E430G mutation in the Fc region and a second antibody comprising an E430G mutation in the Fc region, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR4 antibody; c) anti-TNFR1 antibody; d) anti-DR6 antibody; c) anti-DR3 antibody; d) anti-EDAR antibody and e) anti-NGFR antibody.
[0252] In one embodiment of the present invention the composition comprises a first anti-DR5 antibody comprising an E345K mutation in the Fc region and a second antibody comprising an E345K mutation in the Fc region, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR4 antibody; c) anti-TNFR1 antibody; d) anti-DR6 antibody; c) anti-DR3 antibody; d) anti-EDAR antibody and e] anti-NGFR antibody.
[0253] In one embodiment of the present invention the composition comprises a first anti-TNFR1 antibody comprising a mutation at an amino acid position corresponding to E430, E345 or S440 in human IgG1, EU numbering and a second antibody comprising a mutation at an amino acid position corresponding to E430, E345 or S440 in human IgG1, EU numbering, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR4 antibody; c) anti-DR5 antibody; d) anti-DR6 antibody; c) anti-DR3 antibody; d) anti-EDAR antibody and e) anti-NGFR antibody.
[0254] In one embodiment of the present invention the composition comprises a first anti-TNFR1 antibody comprising an E430G mutation in the Fc region and a second antibody comprising an E430G mutation in the Fc region, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR4 antibody; c) anti-DR5 antibody; d) anti-DR6 antibody; c) anti-DR3 antibody; d) anti-EDAR antibody and e] anti-NGFR antibody.
[0255] In one embodiment of the present invention the composition comprises a first anti-TNFR1 antibody comprising an E345K mutation in the Fc region and a second antibody comprising a E345K mutation in the Fc region, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR4 antibody; c) anti-DR5 antibody; d) anti-DR6 antibody; c) anti-DR3 antibody; d) anti-EDAR antibody and e] anti-NGFR antibody.
[0256] In one embodiment of the present invention the composition comprises a first anti-DR6 antibody comprising a mutation at an amino acid position corresponding to E430, E345 or S440 in human IgG1, EU numbering and a second antibody comprising a mutation at an amino acid position corresponding to E430, E345 or S440 in human IgG1, EU numbering, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR4 antibody; c) anti-TNFR1 antibody; d) anti-DR5 antibody; c) anti-DR3 antibody; d) anti-EDAR antibody and e) anti-NGFR antibody.
[0257] In one embodiment of the present invention the composition comprises a first anti-DR6 antibody comprising an E430G mutation in the Fc region and a second antibody comprising an E430G mutation in the Fc region, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR4 antibody; c) anti-TNFR1 antibody; d) anti-DR5 antibody; c) anti-DR3 antibody; d) anti-EDAR antibody and e] anti-NGFR antibody.
[0258] In one embodiment of the present invention the composition comprises a first anti-DR6 antibody comprising an E345K mutation in the Fc region and a second antibody comprising an E345K mutation in the Fc region, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR4 antibody; c) anti-TNFR1 antibody; d) anti-DR5 antibody; c) anti-DR3 antibody; d) anti-EDAR antibody and e] anti-NGFR antibody.
[0259] In one embodiment of the present invention the composition comprises a first anti-DR3 antibody comprising a mutation at an amino acid position corresponding to E430, E345 or S440 in human IgG1, EU numbering and a second antibody comprising a mutation at an amino acid position corresponding to E430, E345 or S440 in human IgG1, EU numbering, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR4 antibody; c) anti-TNFR1 antibody; d) anti-DR5 antibody; c) anti-DR6 antibody; d) anti-EDAR antibody and e) anti-NGFR antibody.
[0260] In one embodiment of the present invention the composition comprises a first anti-DR3 antibody comprising an E430G mutation in the Fc region and a second antibody comprising an E430G mutation in the Fc region, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR4 antibody; c) anti-TNFR1 antibody; d) anti-DR5 antibody; c) anti-DR6 antibody; d) anti-EDAR antibody and e] anti-NGFR antibody.
[0261] In one embodiment of the present invention the composition comprises a first anti-DR3 antibody comprising an E345K mutation in the Fc region and a second antibody comprising an E345K mutation in the Fc region, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR4 antibody; c) anti-TNFR1 antibody; d) anti-DR5 antibody; c) anti-DR6 antibody; d) anti-EDAR antibody and e] anti-NGFR antibody.
[0262] In one embodiment of the present invention the composition comprises a first anti-EDAR antibody comprising a mutation at an amino acid position corresponding to E430, E345 or S440 in human IgG1, EU numbering and a second antibody comprising a mutation at an amino acid position corresponding to E430, E345 or S440 in human IgG1, EU numbering, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR4 antibody; c) anti-TNFR1 antibody; d) anti-DR5 antibody; c) anti-DR6 antibody; d) anti-DR3 antibody and e] anti-NGFR antibody.
[0263] In one embodiment of the present invention the composition comprises a first anti-EDAR antibody comprising an E430G mutation in the Fc region and a second antibody comprising an E430G mutation in the Fc region, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR4 antibody; c) anti-TNFR1 antibody; d) anti-DR5 antibody; c) anti-DR6 antibody; d) anti-DR3 antibody and e] anti-NGFR antibody.
[0264] In one embodiment of the present invention the composition comprises a first anti-EDAR antibody comprising an E345K mutation in the Fc region and a second antibody comprising an E345K mutation in the Fc region, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR4 antibody; c) anti-TNFR1 antibody; d) anti-DR5 antibody; c) anti-DR6 antibody; d) anti-DR3 antibody and e) anti-NGFR antibody.
[0265] In one embodiment of the present invention the composition comprises a first anti-NGFR antibody comprising a mutation at an amino acid position corresponding to E430, E345 or S440 in human IgG1, EU numbering and a second antibody comprising a mutation at an amino acid position corresponding to E430, E345 or S440 in human IgG1, EU numbering, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR4 antibody; c) anti-TNFR1 antibody; d) anti-DR5 antibody; c) anti-DR6 antibody; d) anti-DR3 antibody and e] anti-EDAR antibody.
[0266] In one embodiment of the present invention the composition comprises a first anti-NGFR antibody comprising an E430G mutation in the Fc region and a second antibody comprising an E430G mutation in the Fc region, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR4 antibody; c) anti-TNFR1 antibody; d) anti-DR5 antibody; c) anti-DR6 antibody; d) anti-DR3 antibody and e] anti-EDAR antibody.
[0267] In one embodiment of the present invention the composition comprises a first anti-NGFR antibody comprising an E345K mutation in the Fc region and a second antibody comprising an E345K mutation in the Fc region, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR4 antibody; c) anti-TNFR1 antibody; d) anti-DR5 antibody; c) anti-DR6 antibody; d) anti-DR3 antibody and e] anti-EDAR antibody.
[0268] The hexamerization-inhibiting mutation such as K439E or S440K prevents Fc-Fc interaction with antibodies comprising the same hexamerization-inhibiting mutation, but by combining antibodies with a K439E mutation and antibodies with a S440K mutation the inhibiting effect is neutralized and Fc-Fc interactions is restored.
[0269] In one embodiment of the present invention the composition comprises a first antibody selected from the group consisting of; anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR and anti-NGFR antibody and a second selected form the group consisting of; anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR and anti-NGFR antibody wherein the first and the second antibody comprises a further hexamerization-inhibiting mutation corresponding to K439E or S440K in human IgG1 EU numbering.
[0270] In one embodiment of the present invention the composition comprises a first and a second antibody selected from the group consisting of; anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR and anti-NGFR antibody, wherein the first antibody comprises a hexamerization enhancing mutation such as E430G and an hexamerization inhibiting mutation such as K439E, and wherein the second antibody comprises a hexamerization enhancing mutation such as E430G and an hexamerization inhibiting mutation such S440K. Hereby embodiments are provided that allow compositions wherein hexamerization exclusively occur between combinations of antibodies comprising a K439E mutation and antibodies comprising a S440K mutation thereby allowing for hexamerization between antibodies with different binding specificities.
[0271] In one embodiment of the present invention the composition comprises a first and a second antibody selected from the group consisting of; anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR and anti-NGFR antibody, wherein the first antibody comprises a hexamerization enhancing mutation such as E345K and an hexamerization inhibiting mutation such K439E, and wherein the second antibody comprises a hexamerization enhancing mutation such as E345K and an hexamerization inhibiting mutation such S440K. Hereby are embodiments provided that allow compositions wherein hexamerization exclusively occur between combinations of antibodies comprising a K439E mutation and antibodies comprising a S440K mutation thereby allowing for hexamerization between antibodies with different binding specificities.
[0272] In one embodiment of the present invention the composition comprises a first anti-FAS antibody comprising an E430G and a K439E mutation in the Fc region and a second antibody comprising a E430G and a S440K mutation in the Fc region, wherein the second antibody is selected from the group consisting of:
a) anti-DR4 antibody; b) anti-DR5 antibody; c) anti-TNFR1 antibody; d) anti-DR6 antibody; c) anti-DR3 antibody; d) anti-EDAR antibody and e] anti-NGFR antibody.
[0273] In one embodiment of the present invention the composition comprises a first anti-FAS antibody comprising an E345K and a K439E mutation in the Fc region and a second antibody comprising an E345K and a S440K mutation in the Fc region, wherein the second antibody is selected from the group consisting of:
a) anti-DR4 antibody; b) anti-DR5 antibody; c) anti-TNFR1 antibody; d) anti-DR6 antibody; c) anti-DR3 antibody; d) anti-EDAR antibody and e) anti-NGFR antibody.
[0274] In one embodiment of the present invention the composition comprises a first anti-DR4 antibody comprising an E430G and a K439E mutation in the Fc region and a second antibody comprising an E430G and a S440K mutation in the Fc region, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR5 antibody; c) anti-TNFR1 antibody; d) anti-DR6 antibody; c) anti-DR3 antibody; d) anti-EDAR antibody and e) anti-NGFR antibody.
[0275] In one embodiment of the present invention the composition comprises a first anti-DR4 antibody comprising an E345K and a K439E mutation in the Fc region and a second antibody comprising an E345K and a S440K mutation in the Fc region, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR5 antibody; c) anti-TNFR1 antibody; d) anti-DR6 antibody; c) anti-DR3 antibody; d) anti-EDAR antibody and e] anti-NGFR antibody.
[0276] In one embodiment of the present invention the composition comprises a first anti-DR5 antibody comprising an E430G and a K439E mutation in the Fc region and a second antibody comprising an E430G and a S440K mutation in the Fc region, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR4 antibody; c) anti-TNFR1 antibody; d) anti-DR6 antibody; c) anti-DR3 antibody; d) anti-EDAR antibody and e) anti-NGFR antibody.
[0277] In one embodiment of the present invention the composition comprises a first anti-DR5 antibody comprising an E345K and a K439E mutation in the Fc region and a second antibody comprising an E345K and a S440K mutation in the Fc region, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR4 antibody; c) anti-TNFR1 antibody; d) anti-DR6 antibody; c) anti-DR3 antibody; d) anti-EDAR antibody and e] anti-NGFR antibody.
[0278] In one embodiment of the present invention the composition comprises a first anti-TNFR1 antibody comprising an E430G and a K439E mutation in the Fc region and a second antibody comprising an E430G and a S440K mutation in the Fc region, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR4 antibody; c) anti-DR5 antibody; d) anti-DR6 antibody; c) anti-DR3 antibody; d) anti-EDAR antibody and e] anti-NGFR antibody.
[0279] In one embodiment of the present invention the composition comprises a first anti-TNFR1 antibody comprising an E345K and a K439E mutation in the Fc region and a second antibody comprising an E345K and a S440K mutation in the Fc region, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR4 antibody; c) anti-DR5 antibody; d) anti-DR6 antibody; c) anti-DR3 antibody; d) anti-EDAR antibody and e] anti-NGFR antibody.
[0280] In one embodiment of the present invention the composition comprises a first anti-DR6 antibody comprising an E430G and a K439E mutation in the Fc region and a second antibody comprising an E430G and a S440K mutation in the Fc region, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR4 antibody; c) anti-TNFR1 antibody; d) anti-DR5 antibody; c) anti-DR3 antibody; d) anti-EDAR antibody and e) anti-NGFR antibody.
[0281] In one embodiment of the present invention the composition comprises a first anti-DR6 antibody comprising an E345K and a K439E mutation in the Fc region and a second antibody comprising an E345K and a S440K mutation in the Fc region, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR4 antibody; c) anti-TNFR1 antibody; d) anti-DR5 antibody; c) anti-DR3 antibody; d) anti-EDAR antibody and e) anti-NGFR antibody.
[0282] In one embodiment of the present invention the composition comprises a first anti-DR3 antibody comprising an E430G and a K439E mutation in the Fc region and a second antibody comprising an E430G and a S440K mutation in the Fc region, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR4 antibody; c) anti-TNFR1 antibody; d) anti-DR5 antibody; c) anti-DR6 antibody; d) anti-EDAR antibody and e] anti-NGFR antibody.
[0283] In one embodiment of the present invention the composition comprises a first anti-DR3 antibody comprising an E345K and a K439E mutation in the Fc region and a second antibody comprising an E345K and a S440K mutation in the Fc region, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR4 antibody; c) anti-TNFR1 antibody; d) anti-DR5 antibody; c) anti-DR6 antibody; d) anti-EDAR antibody and e] anti-NGFR antibody.
[0284] In one embodiment of the present invention the composition comprises a first anti-EDAR antibody comprising an E430G and a K439E mutation in the Fc region and a second antibody comprising an E430G and a S440K mutation in the Fc region, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR4 antibody; c) anti-TNFR1 antibody; d) anti-DR5 antibody; c) anti-DR6 antibody; d) anti-DR3 antibody and e) anti-NGFR antibody.
[0285] In one embodiment of the present invention the composition comprises a first anti-EDAR antibody comprising an E345K and a K439E mutation in the Fc region and a second antibody comprising an E345K and a S440K mutation in the Fc region, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR4 antibody; c) anti-TNFR1 antibody; d) anti-DR5 antibody; c) anti-DR6 antibody; d) anti-DR3 antibody and e) anti-NGFR antibody.
[0286] In one embodiment of the present invention the composition comprises a first anti-NGFR antibody comprising an E430G and a K439E mutation in the Fc region and a second antibody comprising an E430G and a S440K mutation in the Fc region, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR4 antibody; c) anti-TNFR1 antibody; d) anti-DR5 antibody; c) anti-DR6 antibody; d) anti-DR3 antibody and e) anti-EDAR antibody.
[0287] In one embodiment of the present invention the composition comprises a first anti-NGFR antibody comprising an E345K and a K439E mutation in the Fc region and a second antibody comprising an E345K and a S440K mutation in the Fc region, wherein the second antibody is selected from the group consisting of:
a) anti-FAS antibody; b) anti-DR4 antibody; c) anti-TNFR1 antibody; d) anti-DR5 antibody; c) anti-DR6 antibody; d) anti-DR3 antibody and e) anti-EDAR antibody.
[0288] Hereby are embodiments provided that allow compositions wherein hexamerization exclusively occur between combinations of antibodies comprising a K439E mutation and antibodies comprising a S440K mutation thereby allowing for hexamerization between antibodies with different binding specificities.
[0289] In one embodiment of the present invention the composition comprises a first anti-death receptor antibody and a second anti-death receptor antibody binding different epitopes on the same death receptor.
[0290] In one embodiment of the present invention the composition comprises said first anti-death receptor antibody binding to a death receptor, which does not block binding of said second anti-death receptor antibody, when the first and the second anti-death receptor antibody bind to the same target. That is in one embodiment of the invention the composition comprises a first anti-death receptor antibody and a second anti-death receptor antibody, wherein the first and the second antibody does not compete for binding to the death domain receptor.
[0291] In one embodiment of the invention the composition comprises a first and a second anti-death receptor antibody selected from the following group anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR and anti-NGFR, wherein said first antibody and said second antibody are present in the composition at a 1:49 to 49:1 molar ratio, such as 1:1 molar ratio, a 1:2 molar ratio, a 1:3 molar ratio, a 1:4 molar ratio, a 1:5 molar ratio, a 1:6 molar ratio, a 1:7 molar ratio, a 1:8 molar ratio, a 1:9 molar ratio, a 1:5 molar ratio, a 1:5 molar ratio, a 1:5 molar ratio, a 1:10 molar ratio, a 1:15 molar ratio, a 1:20 molar ratio, a 1:25 molar ratio, a 1:30 molar ratio, a 1:35 molar ratio, a 1:40 molar ratio, a 1:45 molar ratio a 1:50 molar ratio, a 50:1 molar ratio, a 45:1 molar ratio, a 40:1 molar ratio, a 35:1 molar ratio, a 30:1 molar ratio a 25:1 molar ratio, a 20:1 molar ratio, a 15:1 molar ratio, a 10:1 molar ratio, a 9:1 molar ratio, a 8:1 molar ratio, a 7:1 molar ratio, a 6:1 molar ratio, a 5:1 molar ratio, a 4:1 molar ratio, a 3:1 molar ratio, a 2:1 molar ratio.
[0292] In one embodiment of the invention the composition comprises a first and a second antibody, wherein said first antibody and said second antibody are present in the composition at a 1:9 to 9:1 molar ratio.
[0293] In one embodiment of the invention the composition comprises a first and a second anti-death receptor antibody, wherein said first antibody and said second antibody are present in the composition at approximately a 1:1 molar ratio.
[0294] In one embodiment of the invention the composition comprises a first and a second anti-death receptor antibody, wherein said first antibody and said second antibody are present in the composition at a 1:1 molar ratio.
[0295] In a preferred embodiment of the invention the composition comprises a first and a second anti-death receptor antibody, wherein said first antibody and second antibody and/or any additional antibodies are present in the composition at an equimolar ratio.
[0296] In one embodiment of the invention the composition is a pharmaceutical composition.
[0297] In one embodiment of the invention the bispecific antibody is comprised in a pharmaceutical composition.
[0298] Pharmaceutical compositions of the present invention may comprise antibodies such as monoclonal antibodies or bispecific antibodies according to any aspect or embodiment of the present invention.
[0299] The pharmaceutical compositions may be formulated with pharmaceutically acceptable carriers or diluents as well as any other known adjuvants and excipients in accordance with conventional techniques such as those disclosed in (Rowe et al., Handbook of Pharmaceutical Excipients, 2012 June, ISBN 9780857110275)
[0300] The pharmaceutically acceptable carriers or diluents as well as any other known adjuvants and excipients should be suitable for the antibody or bispecific antibody of the present invention and the chosen mode of administration. Suitability for carriers and other components of pharmaceutical compositions is determined based on the lack of significant negative impact on the desired biological properties of the chosen compound or pharmaceutical composition of the present invention (e.g., less than a substantial impact (10% or less relative inhibition, 5% or less relative inhibition, etc.) upon antigen binding).
[0301] A pharmaceutical composition of the present invention may also include diluents, fillers, salts, buffers, detergents (e. g., a nonionic detergent, such as Tween-20 or Tween-80), stabilizers (e.g., sugars or protein-free amino acids), preservatives, tissue fixatives, solubilizers, and/or other materials suitable for inclusion in a pharmaceutical composition. The actual dosage levels of the active ingredients in the pharmaceutical compositions of the present invention may be varied so as to obtain an amount of the active ingredient which is effective to achieve the desired therapeutic response for a particular patient, composition, and mode of administration, without being toxic to the patient. The selected dosage level will depend upon a variety of pharmacokinetic factors including the activity of the particular compositions of the present invention employed, or the amide thereof, the route of administration, the time of administration, the rate of excretion of the particular compound being employed, the duration of the treatment, other drugs, compounds and/or materials used in combination with the particular compositions employed, the age, sex, weight, condition, general health and prior medical history of the patient being treated, and like factors well known in the medical arts.
[0302] The pharmaceutical composition may be administered by any suitable route and mode. Suitable routes of administering a compound of the present invention in vivo and in vitro are well known in the art and may be selected by those of ordinary skill in the art.
[0303] In one embodiment, the pharmaceutical composition of the present invention is administered parenterally.
[0304] The terms "parenteral administration" and "administered parenterally" as used herein refers to modes of administration other than enteral and topical administration, usually by injection, and include epidermal, intravenous, intramuscular, intra-arterial, intrathecal, intracapsular, intra-orbital, intracardiac, intradermal, intraperitoneal, intratendinous, transtracheal, subcutaneous, subcuticular, intra-articular, subcapsular, subarachnoid, intraspinal, intracranial, intrathoracic, epidural and intrasternal injection and infusion.
[0305] In one embodiment, the pharmaceutical composition of the present invention is administered by intravenous or subcutaneous injection or infusion.
[0306] In one embodiment of the present invention the pharmaceutical composition comprises one or more antibodies according to the invention such as monoclonal antibodies or bispecific antibodies together with a pharmaceutical carrier.
[0307] Pharmaceutically acceptable carriers include any and all suitable solvents, dispersion media, coatings, antibacterial and antifungal agents, isotonicity agents, antioxidants and absorption-delaying agents, and the like that are physiologically compatible with a compound of the present invention.
[0308] Examples of suitable aqueous and non-aqueous carriers which may be employed in the pharmaceutical compositions of the present invention include water, saline, phosphate-buffered saline, ethanol, dextrose, polyols (such as glycerol, propylene glycol, polyethylene glycol, and the like), and suitable mixtures thereof, vegetable oils, such as olive oil, corn oil, peanut oil, cottonseed oil, and sesame oil, carboxymethyl cellulose colloidal solutions, tragacanth gum and injectable organic esters, such as ethyl oleate, and/or various buffers. Other carriers are well known in the pharmaceutical arts.
[0309] Pharmaceutically acceptable carriers include sterile aqueous solutions or dispersions and sterile powders for the extemporaneous preparation of sterile injectable solutions or dispersion. The use of such media and agents for pharmaceutically active substances is known in the art. Except insofar as any conventional media or agent is incompatible with the active compound, use thereof in the pharmaceutical compositions of the present invention is contemplated.
[0310] Proper fluidity may be maintained, for example, by the use of coating materials, such as lecithin, by the maintenance of the required particle size in the case of dispersions, and by the use of surfactants.
[0311] Pharmaceutical compositions of the present invention may also comprise pharmaceutically acceptable antioxidants for instance (1) water-soluble antioxidants, such as ascorbic acid, cysteine hydrochloride, sodium bisulfate, sodium metabisulfite, sodium sulfite and the like; (2) oil-soluble antioxidants, such as ascorbyl palmitate, butylated hydroxyanisole (BHA), butylated hydroxytoluene (BHT), lecithin, propyl gallate, alpha-tocopherol, and the like; and (3) metal-chelating agents, such as citric acid, ethylenediamine tetraacetic acid (EDTA), sorbitol, tartaric acid, phosphoric acid, and the like.
[0312] Pharmaceutical compositions of the present invention may also comprise isotonicity agents, such as sugars, polyalcohols, such as mannitol, sorbitol, glycerol or sodium chloride in the compositions.
[0313] The pharmaceutical compositions of the present invention may also contain one or more adjuvants appropriate for the chosen route of administration such as preservatives, wetting agents, emulsifying agents, dispersing agents, preservatives or buffers, which may enhance the shelf life or effectiveness of the pharmaceutical composition. The compounds of the present invention may be prepared with carriers that will protect the compound against rapid release, such as a controlled release formulation, including implants, transdermal patches, and micro-encapsulated delivery systems. Such carriers may include gelatin, glyceryl monostearate, glyceryl distearate, biodegradable, biocompatible polymers such as ethylene vinyl acetate, polyanhydrides, polyglycolic acid, collagen, poly-ortho-esters, and polylactic acid alone or with a wax, or other materials well known in the art. Methods for the preparation of such formulations are generally known to those skilled in the art. In one embodiment, the compounds of the present invention may be formulated to ensure proper distribution in vivo. Pharmaceutically acceptable carriers for parenteral administration include sterile aqueous solutions or dispersions and sterile powders for the extemporaneous preparation of sterile injectable solutions or dispersion. The use of such media and agents for pharmaceutically active substances is known in the art. Except insofar as any conventional media or agent is incompatible with the active compound, use thereof in the pharmaceutical compositions of the present invention is contemplated. Other active or therapeutic compounds may also be incorporated into the compositions.
[0314] Pharmaceutical compositions for injection or infusion must typically be sterile and stable under the conditions of manufacture and storage. The composition may be formulated as a solution, micro-emulsion, liposome, or other ordered structure suitable to high drug concentration. The carrier may be an aqueous or a non-aqueous solvent or dispersion medium containing for instance water, ethanol, polyols (such as glycerol, propylene glycol, polyethylene glycol, and the like), and suitable mixtures thereof, vegetable oils, such as olive oil, and injectable organic esters, such as ethyl oleate. The proper fluidity may be maintained, for example, by the use of a coating such as lecithin, by the maintenance of the required particle size in the case of dispersion and by the use of surfactants. In many cases, it will be preferable to include isotonic agents, for example, sugars, polyalcohols such as glycerol, mannitol, sorbitol, or sodium chloride in the composition. Prolonged absorption of the injectable compositions may be brought about by including in the composition an agent that delays absorption, for example, monostearate salts and gelatin. Sterile injectable solutions may be prepared by incorporating the active compound in the required amount in an appropriate solvent with one or a combination of ingredients e.g. as enumerated above, as required, followed by sterilization microfiltration. Generally, dispersions are prepared by incorporating the active compound into a sterile vehicle that contains a basic dispersion medium and the required other ingredients e.g. from those enumerated above. In the case of sterile powders for the preparation of sterile injectable solutions, examples of methods of preparation are vacuum-drying and freeze-drying (lyophilization) that yield a powder of the active ingredient plus any additional desired ingredient from a previously sterile-filtered solution thereof.
[0315] Sterile injectable solutions may be prepared by incorporating the active compound in the required amount in an appropriate solvent with one or a combination of ingredients enumerated above, as required, followed by sterilization microfiltration. Generally, dispersions are prepared by incorporating the active compound into a sterile vehicle that contains a basic dispersion medium and the required other ingredients from those enumerated above. In the case of sterile powders for the preparation of sterile injectable solutions, examples of methods of preparation are vacuum-drying and freeze-drying (lyophilization) that yield a powder of the active ingredient plus any additional desired ingredient from a previously sterile-filtered solution thereof.
[0316] The pharmaceutical composition of the present invention may contain one or more monoclonal antibodies or one or more bispecific antibodies of the present invention, a combination of an antibody or a bispecific antibody according to the invention with another therapeutic compound, or a combination of compounds of the present invention.
Therapeutic Applications
[0317] The antibodies such as monoclonal antibodies, bispecific antibodies or compositions according to any aspect or embodiment of the present invention may be used as a medicament, i.e. for therapeutic applications.
[0318] In one embodiment of the present invention the composition comprises one or more antibodies according to the invention such as monoclonal antibodies or bispecific antibodies for use as a medicament.
[0319] In another aspect, the present invention provides methods for treating or preventing a disorder involving cells expressing a death receptor such as FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR in a subject, which method comprises administration of a therapeutically effective amount of an anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR, and anti-NGFR antibody, bispecific antibody or a composition comprising one or more antibodies of the present invention to a subject in need thereof. The method typically involves administering to a subject in need thereof an anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR, and anti-NGFR antibody, a bispecific antibody or composition according to the present invention in an amount effective to treat or prevent the disorder.
[0320] The anti-death receptor antibodies of the present invention can be used in the treatment or prevention of disorders involving cells expressing the death receptor. For example, the antibodies may be administered to human subjects, e.g., in vivo, to treat or prevent disorders involving FAS-expressing cells, DR4-expressing cells, DR5-expressing cells, TNFR1-expressing cells, DR6-expressing cells, DR3-expressing cells, EDAR-expressing cells or NGFR-expressing cells. As used herein, the term "subject" is typically a human to whom the anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibody or bispecific antibody is administered. Subjects may for instance include human patients having disorders that may be corrected or ameliorated by modulating FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR function or by killing of the FAS-expressing cells, DR4-expressing cells, DR5-expressing cells, TNFR1-expressing cells, DR6-expressing cells, DR3-expressing cells, EDAR-expressing cells or NGFR-expressing cells, directly or indirectly.
[0321] In one aspect, the present invention relates to an anti-death receptor antibody, bispecific antibody or composition as defined in any aspect or embodiment herein, for use in treatment or to ameliorate symptoms of a disease or disorder involving cells expressing one or more death receptors i.e. FAS-expressing cells, DR4-expressing cells, DR5-expressing cells, TNFR1-expressing cells, DR6-expressing cells, DR3-expressing cells, EDAR-expressing cells or NGFR-expressing cells. In some diseases or disorders the cells express more than one death receptor. That is in some diseases or disorders the cells expresses any combination of the following group of death receptors FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR and NGFR.
[0322] In one embodiment of the present invention the composition comprising an anti-death receptor i.e. an anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibody or bispecific antibody according to any aspect or embodiment as disclosed herein, for use in treatment of infectious disease, autoimmune disease or cardiovascular anomalies.
[0323] In one aspect, the present invention relates to an anti-death receptor i.e. an anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibody, bispecific antibody or composition as defined in any aspect or embodiment herein, for use in treatment or to ameliorate symptoms of cancer and/or tumors.
[0324] In one embodiment of the present invention the composition comprising an anti-death receptor i.e. an anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibody or bispecific antibody according to any aspect or embodiment of the invention is for use in treatment of cancer and/or tumors.
[0325] The term "cancer" refers to or describes the physiological condition in mammals such as humans that is typically characterized by unregulated growth. Most cancers belong to one of two larger groups of cancers i.e., solid tumors and hematological tumors.
[0326] In a particular aspect, an anti-death receptor i.e. an anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibody, bispecific antibody or composition is administered prophylactically in order to reduce the risk of developing cancer, delay the onset of an event in cancer progression or reduce the risk of recurrence when a cancer is in remission and/or a primary tumor has been surgically removed. In the latter case, the anti-death receptor i.e. an anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibody, bispecific antibody or composition could, for example, be administered in association with (i.e., before, during, or after) the surgery. Prophylactic administration may also be useful in patients where it is difficult to locate a tumor that is believed to be present due to other biological factors.
[0327] In one embodiment the composition comprising one or more anti-death receptor i.e. an anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibodies or bispecific antibodies of the present invention is for use in treatment of solid tumors and/or hematological tumors
[0328] In one embodiment the composition comprising one or more anti-death receptor i.e. an anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibodies or bispecific antibodies of the present invention is for use in treatment of solid tumors such as, colorectal cancer, including colorectal carcinoma and colorectal adenocarcinoma, bladder cancer, osteosarcoma, chondrosarcoma, breast cancer, including triple-negative breast cancer, cancers of the central nervous system, including glioblastoma, astrocytoma, neuroblastoma, neural fibrosarcoma, neuroendocrine tumors, cervical cancer, endometrium cancer, gastric cancer, including gastric adenocarcinoma, head and neck cancer, kidney cancer, liver cancer, including hepatocellular carcinoma, lung cancer, including NSCLC and SCLC, ovarian cancer, pancreatic cancer, including pancreatic ductal carcinoma and pancreatic adenocarcinoma, sarcoma or skin cancer, including malignant melanoma and non-melanoma skin cancers.
[0329] In one embodiment of the invention the composition comprising one or more anti-death receptor i.e. an anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibodies or bispecific antibodies is for use in treatment of hematological tumors such as, leukemia, including chronic lymphocytic leukemia and myeloid leukemia, including acute myeloid leukemia and chronic myeloid leukemia, lymphoma, including Non-Hodgkin lymphoma or multiple myeloma, including Hodgkin Lymphoma, and including myelodysplastic syndromes.
[0330] In a particular embodiment of the present invention the composition comprising one or more anti-death receptor i.e. an anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibodies or bispecific antibodies is for use in treatment of a cancer selected from the following group of cancers; bladder cancer, bone cancer, colorectal cancer, sarcoma, endometrium cancer, fibroblast cancer, gastric cancer, head and neck cancer, kidney cancer, leukemia, liver cancer, lung cancer, lymphoma, muscle cancer, neural tissue cancer, ovary cancer, pancreas cancer and skin cancer.
[0331] In one embodiment of the invention the composition comprising one or more anti-death receptor i.e. an anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibodies or bispecific antibodies is for use in inhibiting growth of FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR positive or FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR expressing tumors or cancers.
[0332] In the present invention FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR positive tumors or cancers are to be understood as tumor cells and/or cancer cells expressing DR5 on the cell surface. Such FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR expression may be detected by immunohistochemistry, flow cytometry or other suitable diagnostic method.
[0333] In one embodiment of the invention the composition comprising one or more anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibodies or bispecific antibodies is for use in inhibiting growth of FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR tumors or cancers. Tumors and cancer tissues that show heterogenous expression of FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR are also considered as FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR positive tumors and cancers.
[0334] Tumors and/or cancers may express FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR on some tumor and/or cancer cells and/or tissues showing FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR expression, some tumor and/or cancers may show over-expression or aberrant expression of FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR, whereas other tumors and/or cancers show heterogeneous expression of FAS, DR4, DR5, TNFR1, DR6, DR3, EDAR or NGFR. Such tumors and/or cancers may all be suitable targets for treatment with anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibodies, bispecific antibodies and compositions comprising such antibodies according to the present invention.
[0335] In one embodiment of the invention the composition comprising one or more anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibodies or bispecific antibodies is for use in induction of apoptosis in FAS-expressing tumors, DR4-expressing tumors, DR5-expressing tumors, TNFR1-expressing tumors, DR6-expressing tumors, DR3-expressing tumors, EDAR-expressing tumors, or NGFR-expressing tumors. In one embodiment the tumor is expressing a one or more death receptors, that is a combination of two death receptors, a combination of three death receptors, a combination of four death receptors, a combination of five death receptors, a combination of six death receptors, a combination of seven death receptors, a combination of eight death receptors.
[0336] Another aspect of the present invention comprises a method of treating an individual having a cancer comprising administering to said individual an effective amount of an anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibody, bispecific antibody or composition according to the invention.
[0337] In one embodiment of the invention the method of treating an individual having a cancer comprising administering to said individual an effective amount of an anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibody, bispecific antibody or composition according to the invention, further comprises administering an additional therapeutic agent to the said individual.
[0338] In one embodiment of the invention the additional therapeutic agent is a single agent or a combination of agents comprising an agent or regimen selected from the group chemotherapeutics (including but not limited to paclitaxel, temozolomide, cisplatin, carboplatin, oxaliplatin, irinotecan, doxorubicin, gemcitabine, 5-fluorouracil, pemetrexed), kinase inhibitors (including but not limited to sorafenib, sunitinib or everolimus), apoptosis-modulating agents (including but not limited to recombinant human TRAIL or birinapant), RAS inhibitors, proteasome inhibitors (including but not limited to bortezomib), histon deacetylase inhibitors (including but not limited to vorinostat), nutraceuticals, cytokines (including but not limited to IFN-.gamma.), antibodies or antibody mimetics (including but not limited to anti-TF, anti-AXL, anti-EGFR, anti-IGF-1R, anti-VEGF, anti-CD20, anti-CD38, anti-HER2, anti-PD-1, anti-PD-L1, anti-CTLA4, anti-CD40, anti-CD137, anti-GITR, anti-VISTA (or other immunomodulatory targets) antibodies and antibody mimetics), and antibody-drug conjugates such as brentuximab vedotin, trastuzumab emtansine, HuMax-TF-ADC or HuMax-AXL-ADC.
[0339] In a further aspect, the invention comprises a kit of parts comprising an anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibody, bispecific antibody or composition according to the, wherein said antibody, bispecific antibody or composition is in one or more containers such as one or more vials.
[0340] In one embodiment of the invention the kit of parts comprising an anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibody, bispecific antibody or composition according to the invention is for simultaneous, separate or sequential use in therapy.
[0341] In a further embodiment the present invention is for use of an anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibody, bispecific antibody or a composition according to the invention for the manufacture of a medicament for treatment of cancer.
[0342] When describing the embodiments of the present invention, the combinations and permutations of all possible embodiments have not been explicitly described. Nevertheless, the mere fact that certain measures are recited in mutually different dependent claims or described in different embodiments does not indicate that a combination of these measures cannot be used to advantage. The present invention envisages all possible combinations and permutations of the described embodiments.
[0343] In another aspect of the present invention, the invention comprises a nucleic acid construct encoding an antibody according to amino acid sequences set forth in table 1. That is in one embodiment, the present invention comprises, a nucleic acid construct encoding an antibody corresponding to the amino acid sequences set forth in table 1. In one embodiment of the present invention, the nucleic acid construct encodes an antibody according to any embodiments disclosed herein.
[0344] In a further aspect, the present invention relates to a nucleic acid encoding an antibody according to the present invention, wherein the Fc region comprises a mutation of an amino acids position corresponding to E430, E345 or S440 in a human IgG1, EU numbering. It is further contemplated that the nucleic acid encoding an antibody according to the invention comprises the amino acid substitutions in the specific amino acid positions herein described. Thus, in one embodiment, the nucleic acid encodes an antibody having the sequence according to SEQ ID NO: 1 to 50.
[0345] In another aspect, the invention relates to nucleic acids encoding a sequence of a human, humanized or chimeric anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibody for use in the invention, to expression vectors encoding the sequences of such an antibody, to host cells comprising such expression vectors, to hybridomas which produce such antibodies, and to methods of producing such an antibody by culturing such host cells or hybridomas under appropriate conditions whereby the antibody is produced and, optionally, retrieved.
[0346] In one embodiment, the invention provides an expression vector comprising a nucleotide sequence encoding one or more of the amino acid sequence according to SEQ ID Nos: 1 to 51.
[0347] In another embodiment, the expression vector comprises a nucleotide sequence encoding any one or more of the VH CDR3 amino acid sequences selected from SEQ ID NOs: 18, 22, 19, 30, 39 and 46. In another embodiment, the expression vector comprises a nucleotide sequence encoding a VH amino acid sequence selected from SEQ ID NOs: 13, 15, 19, 26, 31 and 40. In another embodiment, the expression vector comprises a nucleotide sequence encoding a VL amino acid sequence selected from SEQ ID NOs: 14, 16, 23, 35, 43 and 15. In another embodiment, the expression vector comprises a nucleotide sequence encoding the constant region of a human antibody light chain, of a human antibody heavy chain, or both. In another embodiment, the expression vector comprising a nucleotide sequence encoding the constant region of a human antibody heavy chain of selected from the group consisting of: SEQ ID NOs: 20, 27, 32 and 47.
[0348] In a particular embodiment, the expression vector comprises a nucleotide sequence encoding a variant of one or more of the above amino acid sequences, said variant having at most 25 amino acid modifications, such as at most 20, such as at most 15, 14, 13, 12, or 11 amino acid modifications, such as 10, 9, 8, 7, 6, 5, 4, 3, 2, or 1 amino acid modifications, such as deletions or insertions, preferably substitutions, such as conservative substitutions, or at least 80% identity to any of said sequences, such as at least 85% identity or 90% identity or 95% identity, such as 96% identity or 97% identity or 98% identity or 99% identity to any of the afore-mentioned amino acid sequences.
[0349] An expression vector in the context of the present invention may be any suitable vector, including chromosomal, non-chromosomal, and synthetic nucleic acid vectors (a nucleic acid sequence comprising a suitable set of expression control elements). Examples of such vectors include derivatives of SV40, bacterial plasmids, phage DNA, baculovirus, yeast plasmids, vectors derived from combinations of plasmids and phage DNA, and viral nucleic acid (RNA or DNA) vectors. In one embodiment, a humanized CD3 antibody-encoding nucleic acid is comprised in a naked DNA or RNA vector, including, for example, a linear expression element (as described in for instance Sykes and Johnston, Nat Biotech 17, 355-59 (1997)), a compacted nucleic acid vector (as described in for instance U.S. Pat. No. 6,077,835 and/or WO 00/70087), a plasmid vector such as pBR322, pUC 19/18, or pUC 118/119, a "midge" minimally-sized nucleic acid vector (as described in for instance Schakowski et al., Mol Ther 3, 793-800 (2001)), or as a precipitated nucleic acid vector construct, such as a CaPO.sub.4.sup.--precipitated construct (as described in for instance WO 00/46147, Benvenisty and Reshef, PNAS USA 83, 9551-55 (1986), Wigler et al., Cell 14, 725 (1978), and Coraro and Pearson, Somatic Cell Genetics 7, 603 (1981)). Such nucleic acid vectors and the usage thereof are well known in the art (see for instance U.S. Pat. Nos. 5,589,466 and 5,973,972). In one embodiment, the vector is suitable for expression of the humanized anti-DR5 antibody, in a bacterial cell. Examples of such vectors include expression vectors such as BlueScript (Stratagene), pIN vectors (Van Heeke & Schuster, J Biol Chem 264, 5503-5509 (1989)), pET vectors (Novagen, Madison Wis.) and the like.
[0350] An expression vector may also or alternatively be a vector suitable for expression in a yeast system. Any vector suitable for expression in a yeast system may be employed. Suitable vectors include, for example, vectors comprising constitutive or inducible promoters such as alpha factor, alcohol oxidase and PGH (reviewed in: F. Ausubel et al., ed. Current Protocols in Molecular Biology, Greene Publishing and Wiley InterScience New York (1987), and Grant et al., Methods in Enzymol 153, 516-544 (1987)).
[0351] A nucleic acid and/or vector may also comprise a nucleic acid sequence encoding a secretion/localization sequence, which can target a polypeptide, such as a nascent polypeptide chain, to the periplasmic space or into cell culture media. Such sequences are known in the art, and include secretion leader or signal peptides, organelle-targeting sequences (e.g., nuclear localization sequences, ER retention signals, mitochondrial transit sequences, chloroplast transit sequences), membrane localization/anchor sequences (e.g., stop transfer sequences, GPI anchor sequences), and the like.
[0352] In an expression vector of the invention, anti-DR5 antibody-encoding nucleic acids and the first and the second polypeptides nucleic acids may comprise or be associated with any suitable promoter, enhancer, and other expression-facilitating elements. Examples of such elements include strong expression promoters (e.g., human CMV IE promoter/enhancer as well as RSV, SV40, 5L3-3, MMTV, and HIV LTR promoters), effective poly (A) termination sequences, an origin of replication for plasmid product in E. coli, an antibiotic resistance gene as selectable marker, and/or a convenient cloning site (e.g., a polylinker). Nucleic acids may also comprise an inducible promoter as opposed to a constitutive promoter such as CMV IE (the skilled artisan will recognize that such terms are actually descriptors of a degree of gene expression under certain conditions).
[0353] In one embodiment, the anti-DR5 antibody-encoding expression is positioned in and/or delivered to the host cell or host animal via a viral vector.
[0354] Such expression vectors may be used for recombinant production of anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibodies.
[0355] In one aspect, the anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibodies of any aspect or embodiment described herein are provided by use of recombinant eukaryotic or prokaryotic host cell which produces the antibody. Accordingly, the invention provides a recombinant eukaryotic or prokaryotic host cell, such as a transfectoma, which produces an anti-DR5 antibody as defined herein. Examples of host cells include yeast, bacterial and mammalian cells, such as CHO or HEK-293 cells. For example, in one embodiment, the host cell comprises a nucleic acid stably integrated into the cellular genome that comprises a sequence coding for expression of a anti-DR5 antibody described herein. In one embodiment, the host cell comprises a nucleic acid stably integrated into the cellular genome that comprise a sequence coding for expression of a first or a second polypeptide described herein. In another embodiment, the host cell comprises a non-integrated nucleic acid, such as a plasmid, cosmid, phagemid, or linear expression element, which comprises a sequence coding for expression of an anti-FAS, anti-DR4, anti-DR5, anti-TNFR1, anti-DR6, anti-DR3, anti-EDAR or anti-NGFR antibody, a first or a second polypeptide described herein.
[0356] The term "recombinant host cell" (or simply "host cell"), as used herein, is intended to refer to a cell into which an expression vector has been introduced. It should be understood that such terms are intended to refer not only to the particular subject cell, but also to the progeny of such a cell. Because certain modifications may occur in succeeding generations due to either mutation or environmental influences, such progeny may not, in fact, be identical to the parent cell, but are still included within the scope of the term "host cell" as used herein. Recombinant host cells include, for example, transfectomas, such as CHO cells, HEK-293 cells, PER.C6, NS0 cells, and lymphocytic cells, and prokaryotic cells such as E. coli and other eukaryotic hosts such as plant cells and fungi.
[0357] The term "transfectoma", as used herein, includes recombinant eukaryotic host cells expressing the antibody or a target antigen, such as CHO cells, PER.C6, NS0 cells, HEK-293 cells, plant cells, or fungi, including yeast cells.
[0358] In a further aspect, the invention relates to a method for producing an antibody of the invention, said method comprising the steps of
a) culturing a hybridoma or a host cell of the invention as described herein above, and b) retrieving and/or purifying the antibody of the invention from the culture media.
[0359] In a further aspect, the nucleotide sequence encoding a sequence of an antibody further encodes a second moiety, such as a therapeutic polypeptide. Exemplary therapeutic antibodies are described elsewhere herein. In one embodiment, the invention relates to a method for producing an antibody fusion protein, said method comprising the steps of
a) culturing a host cell comprising an expression vector comprising such a nucleotide sequence, and b) retrieving and/or purifying the antibody fusion protein from the culture media.
[0360] In one aspect of the present invention, the invention comprises an expression vector comprising on or more nucleic acid constructs encoding an antibody according to any embodiment disclosed herein.
[0361] In a further aspect of the invention, the invention comprises a host cell comprising an expression vector.
TABLE-US-00004 Sequence Table 1 SEQ ID NO: Name Sequence Clone SEQ ID NO: 1 Fc IgG1m (f) ASTKGPSVFPLAPSSKST SGGTAALGCLVKDYFPE PVTVSWNSGALTSGVH TFPAVLQSSGLYSLSSVV TVPSSSLGTQTYICNVN HKPSNTKVDKRVEPKSC DKTHTCPPCPAPELLGG PSVFLFPPKPKDTLMISR TPEVTCVVVDVSHEDPE VKFNWYVDGVEVHNA KTKPREEQYNSTYRVVS VLTVLHQDWLNGKEYK CKVSNKALPAPIEKTISK AKGQPREPQVYTLPPSR EEMTKNQVSLTCLVKGF YPSDIAVEWESNGQPE NNYKTTPPVLDSDGSFF LYSKLTVDKSRWQQGN VFSCSVMHEALHNHYT QKSLSLSPGK SEQ ID NO: 2 Fc IgG1m(z) ASTKGPSVFPLAPSS KSTSGGTAALGCLV KDYFPEPVTVSWNS GALTSGVHTFPAVLQ SSGLYSLSSVVTVPS SSLGTQTYICNVNHK PSNTKVDKKVEPKS CDKTHTCPPCPAPEL LGGPSVFLFPPKPKD TLMISRTPEVTCVVV DVSHEDPEVKFNWY VDGVEVHNAKTKPR EEQYNSTYRVVSVLT VLHQDWLNGKEYKC KVSNKALPAPIEKTIS KAKGQPREPQVYTLP PSREEMTKNQVSLT CLVKGFYPSDIAVEW ESNGQPENNYKTTPP VLDSDGSFFLYSKLT VDKSRWQQGNVFS CSVMHEALHNHYTQ KSLSLSPGK SEQ ID NO: 3 Fc IgG1m(a) ASTKGPSVFPLAPSS KSTSGGTAALGCLV KDYFPEPVTVSWNS GALTSGVHTFPAVLQ SSGLYSLSSVVTVPS SSLGTQTYICNVNHK PSNTKVDKPVEPKSC DKTHTCPPCPAPELL GGPSVFLFPPKPKDT LMISRTPEVTCVVVD VSHEDPEVKFNWYV DGVEVHNAKTKPRE EQYNSTYRVVSVLTV LHQDWLNGKEYKCK VSNKALPAPIEKTISK AKGQPREPQVYTLPP SRDELTKNQVSLTCL VKGFYPSDIAVEWES NGQPENNYKTTPPVL DSDGSFFLYSKLTVD KSRWQQGNVFSCS VMHEALHNHYTQKS LSLSPGK SEQ ID NO: 4 Fc IgG1m(x) ASTKGPSVFPLAPSS KSTSGGTAALGCLV KDYFPEPVTVSWNS GALTSGVHTFPAVLQ SSGLYSLSSVVTVPS SSLGTQTYICNVNHK PSNTKVDKPVEPKSC DKTHTCPPCPAPELL GGPSVFLFPPKPKDT LMISRTPEVTCVVVD VSHEDPEVKFNWYV DGVEVHNAKTKPRE EQYNSTYRVVSVLTV LHQDWLNGKEYKCK VSNKALPAPIEKTISK AKGQPREPQVYTLPP SREEMTKNQVSLTCL VKGFYPSDIAVEWES NGQPENNYKTTPPVL DSDGSFFLYSKLTVD KSRWQQGNVFSCS VMHEGLHNHYTQKS LSLSPGK SEQ ID NO: 5 Fc IgG1m(f)-E430G ASTKGPSVFPLAPSS KSTSGGTAALGCLV KDYFPEPVTVSWNS GALTSGVHTFPAVLQ SSGLYSLSSVVTVPS SSLGTQTYICNVNHK PSNTKVDKRVEPKS CDKTHTCPPCPAPEL LGGPSVFLFPPKPKD TLMISRTPEVTCVVV DVSHEDPEVKFNWY VDGVEVHNAKTKPR EEQYNSTYRVVSVLT VLHQDWLNGKEYKC KVSNKALPAPIEKTIS KAKGQPREPQVYTLP PSREEMTKNQVSLT CLVKGFYPSDIAVEW ESNGQPENNYKTTPP VLDSDGSFFLYSKLT VDKSRWQQGNVFS CSVMHGALHNHYTQ KSLSLSPGK SEQ ID NO: 6 Fc IgG1m(f)-E345K ASTKGPSVFPLAPSS KSTSGGTAALGCLV KDYFPEPVTVSWNS GALTSGVHTFPAVLQ SSGLYSLSSVVTVPS SSLGTQTYICNVNHK PSNTKVDKRVEPKS CDKTHTCPPCPAPEL LGGPSVFLFPPKPKD TLMISRTPEVTCVVV DVSHEDPEVKFNWY VDGVEVHNAKTKPR EEQYNSTYRVVSVLT VLHQDWLNGKEYKC KVSNKALPAPIEKTIS KAKGQPRKPQVYTL PPSREEMTKNQVSLT CLVKGFYPSDIAVEW ESNGQPENNYKTTPP VLDSDGSFFLYSKLT VDKSRWQQGNVFS CSVMHEALHNHYTQ KSLSLSPGK SEQ ID NO: 7 Fc IgG1m(f)-S440Y ASTKGPSVFPLAPSS KSTSGGTAALGCLV KDYFPEPVTVSWNS GALTSGVHTFPAVLQ SSGLYSLSSVVTVPS SSLGTQTYICNVNHK PSNTKVDKRVEPKS CDKTHTCPPCPAPEL LGGPSVFLFPPKPKD TLMISRTPEVTCVVV DVSHEDPEVKFNWY VDGVEVHNAKTKPR EEQYNSTYRVVSVLT VLHQDWLNGKEYKC KVSNKALPAPIEKTIS KAKGQPREPQVYTLP PSREEMTKNQVSLT CLVKGFYPSDIAVEW ESNGQPENNYKTTPP VLDSDGSFFLYSKLT VDKSRWQQGNVFS CSVMHEALHNHYTQ KYLSLSPGK SEQ ID NO: 8 Fc IgG1m(f)-F405L ASTKGPSVFPLAPSS KSTSGGTAALGCLV KDYFPEPVTVSWNS GALTSGVHTFPAVLQ SSGLYSLSSVVTVPS SSLGTQTYICNVNHK PSNTKVDKRVEPKS CDKTHTCPPCPAPEL LGGPSVFLFPPKPKD TLMISRTPEVTCVVV DVSHEDPEVKFNWY VDGVEVHNAKTKPR EEQYNSTYRVVSVLT VLHQDWLNGKEYKC KVSNKALPAPIEKTIS KAKGQPREPQVYTLP PSREEMTKNQVSLT CLVKGFYPSDIAVEW ESNGQPENNYKTTPP VLDSDGSFLLYSKLT VDKSRWQQGNVFS CSVMHEALHNHYTQ KSLSLSPGK SEQ ID NO: 9 Fc IgG1m(f)-K409R ASTKGPSVFPLAPSS KSTSGGTAALGCLV KDYFPEPVTVSWNS GALTSGVHTFPAVLQ SSGLYSLSSVVTVPS SSLGTQTYICNVNHK PSNTKVDKRVEPKS CDKTHTCPPCPAPEL LGGPSVFLFPPKPKD TLMISRTPEVTCVVV DVSHEDPEVKFNWY VDGVEVHNAKTKPR EEQYNSTYRVVSVLT VLHQDWLNGKEYKC KVSNKALPAPIEKTIS KAKGQPREPQVYTLP PSREEMTKNQVSLT CLVKGFYPSDIAVEW ESNGQPENNYKTTPP VLDSDGSFFLYSRLT VDKSRWQQGNVFS CSVMHEALHNHYTQ KSLSLSPGK SEQ ID NO: 10 Fc IgG1m(f)-K439E ASTKGPSVFPLAPSS KSTSGGTAALGCLV KDYFPEPVTVSWNS GALTSGVHTFPAVLQ SSGLYSLSSVVTVPS SSLGTQTYICNVNHK PSNTKVDKRVEPKS CDKTHTCPPCPAPEL LGGPSVFLFPPKPKD TLMISRTPEVTCVVV DVSHEDPEVKFNWY VDGVEVHNAKTKPR EEQYNSTYRVVSVLT VLHQDWLNGKEYKC KVSNKALPAPIEKTIS KAKGQPREPQVYTLP PSREEMTKNQVSLT CLVKGFYPSDIAVEW ESNGQPENNYKTTPP VLDSDGSFFLYSKLT VDKSRWQQGNVFS CSVMHEALHNHYTQ ESLSLSPGK SEQ ID NO: 11 Fc IgG1m(f)-5440K ASTKGPSVFPLAPSS KSTSGGTAALGCLV KDYFPEPVTVSWNS GALTSGVHTFPAVLQ SSGLYSLSSVVTVPS SSLGTQTYICNVNHK PSNTKVDKRVEPKS CDKTHTCPPCPAPEL LGGPSVFLFPPKPKD
TLMISRTPEVTCVVV DVSHEDPEVKFNWY VDGVEVHNAKTKPR EEQYNSTYRVVSVLT VLHQDWLNGKEYKC KVSNKALPAPIEKTIS KAKGQPREPQVYTLP PSREEMTKNQVSLT CLVKGFYPSDIAVEW ESNGQPENNYKTTPP VLDSDGSFFLYSKLT VDKSRWQQGNVFS CSVMHEALHNHYTQ KKLSLSPGK SEQ ID NO: 12 Fc IgG1m(f)-Y436I ASTKGPSVFPLAPSS KSTSGGTAALGCLV KDYFPEPVTVSWNS GALTSGVHTFPAVLQ SSGLYSLSSVVTVPS SSLGTQTYICNVNHK PSNTKVDKRVEPKS CDKTHTCPPCPAPEL LGGPSVFLFPPKPKD TLMISRTPEVTCVVV DVSHEDPEVKFNWY VDGVEVHNAKTKPR EEQYNSTYRVVSVLT VLHQDWLNGKEYKC KVSNKALPAPIEKTIS KAKGQPREPQVYTLP PSREEMTKNQVSLT CLVKGFYPSDIAVEW ESNGQPENNYKTTPP VLDSDGSFFLYSKLT VDKSRWQQGNVFS CSVMHEALHNHITQ KSLSLSPGK SEQ ID NO: 13 VH DR4-T1014G03 EVQLVQSGAEVKMP GASVKLSCRVSGDT FTAYFIHWVRQAPG QGLEWMGWFNPISG TAGSAEKFRGRVAM TRDTSISTAYMELNR LTFDDTAVYYCARQH RGNTFDPWGQGTLV TVSS SEQ ID NO: 14 VL DR4-T1014G03 QSALTQPASVSGSP GQSITISCTGTSSDI GAYKYVSWYQQHPG KAPKLVIYEVSNRPS GVSSRFSGSKSGQT ASLTISGLQADDEAD YYCNSYQGYNTWVF GGGTKVTVLG SEQ ID NO: 15 VH FAS-E09 QLQLQESGPGLVKP SETLSLTCTVSGASI SANSYYGVWVRQSP GKGLEWVGSIAYRG NSNSGSTYYNPSLKS RATVSVDTSKNQVS LRLTSVTAADTALYY CARRQLLDDGTGYQ WAAFDVWGQGTMV TVSS SEQ ID NO: 16 VL FAS-E09 QSVLTQPPSVSEAPR QTVTISCSGNSFNIG RYPVNWYQQLPGKA PKLLIYYNNLRFSGV SDRFSGSKSGTSAS LAIRDLLSEDEADYY CSTWDDTLKGWVF GGGTKVTVL SEQ ID NO: 17 VH hDR5-01 CDR1 GFNIKDTF hDR5-01 SEQ ID NO: 2 VH hDR5-01 CDR2 IDPANGNT SEQ ID NO: 18 VH hDR5-01 CDR3 VRGLYTYYFDY SEQ ID NO: 19 VH hDR5-01 EVQLQQSGAEVVKPGA SVKLSCKASGFNIKDTFI HWVKQAPGQGLEWIG RIDPANGNTKYDPKFQ GKATITTDTSSNTAYME LSSLRSEDTAVYYCVRGL YTYYFDYWGQGTLVTV SS SEQ ID NO: 20 HC-hDR5-01 EVQLQQSGAEVVKPGA SVKLSCKASGFNIKDTFI HWVKQAPGQGLEWIG RIDPANGNTKYDPKFQ GKATITTDTSSNTAYME LSSLRSEDTAVYYCVRGL YTYYFDYWGQGTLVTV SSASTKGPSVFPLAPSSK STSGGTAALGCLVKDYF PEPVTVSWNSGALTSG VHTFPAVLQSSGLYSLSS VVTVPSSSLGTQTYICN VNHKPSNTKVDKRVEP KSCDKTHTCPPCPAPEL LGGPSVFLFPPKPKDTL MISRTPEVTCVVVDVSH EDPEVKFNWYVDGVEV HNAKTKPREEQYNSTYR VVSVLTVLHQDWLNGK EYKCKVSNKALPAPIEKT ISKAKGQPREPQVYTLP PSREEMTKNQVSLTCLV KGFYPSDIAVEWESNG QPENNYKTTPPVLDSD GSFFLYSKLTVDKSRWQ QGNVFSCSVMHEALHN HYTQKSLSLSPGK SEQ ID NO: 21 VL hDR5-01 CDR1 QSISNN VL hDR5-01 CDR2 FAS SEQ ID NO: 22 VL hDR5-01 CDR3 QQGNSWPYT SEQ ID NO: 23 VL hDR5-01 EIVMTQSPATLSVSPGE RATLSCRASQSISNNLH WYQQKPGQAPRLLIKF ASQSITGIPARFSGSGSG TEFTLTISSLQSEDFAVY YCQQGNSWPYTFGQG TKLEIK SEQ ID NO: 24 LC-hDR5-01 EIVMTQSPATLSVSPGE RATLSCRASQSISNNLH WYQQKPGQAPRLLIKF ASQSITGIPARFSGSGSG TEFTLTISSLQSEDFAVY YCQQGNSWPYTFGQG TKLEIKRTVAAPSVFIFPP SDEQLKSGTASVVCLLN NFYPREAKVQWKVDN ALQSGNSQESVTEQDS KDSTYSLSSTLTLSKADY EKHKVYACEVTHQGLSS PVTKSFNRGEC SEQ ID NO: 17 VH hDR5-01-G56T GFNIKDTF hDR5-01-G56T CDR1 SEQ ID NO: 25 VH hDR5-01-G56T IDPANTNT CDR2 SEQ ID NO: 19 VH hDR5-01-G56T VRGLYTYYFDY CDR3 SEQ ID NO: 26 VH hDR5-01-G56T EVQLQQSGAEVVKPGA SVKLSCKASGFNIKDTFI HWVKQAPGQGLEWIG RIDPANTNTKYDPKFQG KATITTDTSSNTAYMEL SSLRSEDTAVYYCVRGL YTYYFDYWGQGTLVTV SS SEQ ID NO: 27 HC-hDR5-01-G56T EVQLQQSGAEVVKPGA SVKLSCKASGFNIKDTFI HWVKQAPGQGLEWIG RIDPANTNTKYDPKFQG KATITTDTSSNTAYMEL SSLRSEDTAVYYCVRGL YTYYFDYWGQGTLVTV SSASTKGPSVFPLAPSSK STSGGTAALGCLVKDYF PEPVTVSWNSGALTSG VHTFPAVLQSSGLYSLSS VVTVPSSSLGTQTYICN VNHKPSNTKVDKRVEP KSCDKTHTCPPCPAPEL LGGPSVFLFPPKPKDTL MISRTPEVTCVVVDVSH EDPEVKFNWYVDGVEV HNAKTKPREEQYNSTYR VVSVLTVLHQDWLNGK EYKCKVSNKALPAPIEKT ISKAKGQPREPQVYTLP PSREEMTKNQVSLTCLV KGFYPSDIAVEWESNG QPENNYKTTPPVLDSD GSFFLYSKLTVDKSRWQ QGNVFSCSVMHEALHN HYTQKSLSLSPGK SEQ ID NO: 21 VL hDR5-01-G56T QSISNN CDR1 VL hDR5-01-G56T FAS CDR2 SEQ ID NO: 22 VL hDR5-01-G56T QQGNSWPYT CDR3 SEQ ID NO: 23 VL hDR5-01-G56T EIVMTQSPATLSVSPGE RATLSCRASQSISNNLH WYQQKPGQAPRLLIKF ASQSITGIPARFSGSGSG TEFTLTISSLQSEDFAVY YCQQGNSWPYTFGQG TKLEIK SEQ ID NO: 28 VH hDR5-05 CDR1 GFNIKDTH hDR5-05 SEQ ID NO: 29 VH hDR5-05 CDR2 IDPANGNT SEQ ID NO: 30 VH hDR5-05 CDR3 ARWGTNVYFAY SEQ ID NO: 31 VH hDR5-05 QVQLVQSGAEVKKPGA SVKVSCKASGFNIKDTH MHWVRQAPGQRLEWI GRIDPANGNTEYDQKF QGRVTITVDTSASTAYM ELSSLRSEDTAVYYCAR WGTNVYFAYWGQGTL VTVSS SEQ ID NO: 32 HC-hDR5-05 QVQLVQSGAEVKKPGA SVKVSCKASGFNIKDTH MHWVRQAPGQRLEWI GRIDPANGNTEYDQKF QGRVTITVDTSASTAYM ELSSLRSEDTAVYYCAR WGTNVYFAYWGQGTL VTVSSASTKGPSVFPLA PSSKSTSGGTAALGCLV KDYFPEPVTVSWNSGA LTSGVHTFPAVLQSSGL YSLSSVVTVPSSSLGTQT YICNVNHKPSNTKVDKR VEPKSCDKTHTCPPCPA PELLGGPSVFLFPPKPKD TLMISRTPEVTCVVVDV SHEDPEVKFNWYVDGV EVHNAKTKPREEQYNST YRVVSVLTVLHQDWLN GKEYKCKVSNKALPAPI EKTISKAKGQPREPQVY TLPPSREEMTKNQVSLT CLVKGFYPSDIAVEWES NGQPENNYKTTPPVLD
SDGSFFLYSKLTVDKSR WQQGNVFSCSVMHEA LHNHYTQKSLSLSPGK SEQ ID NO: 33 VL hDR5-05 CDR1 SSVSY VL hDR5-05 CDR2 RTS SEQ ID NO: 34 VL hDR5-05 CDR3 QQYHSYPPT SEQ ID NO: 35 VL hDR5-05 DIQLTQSPSSLSASVGD RVTITCSASSSVSYMYW YQQKPGKAPKPWIYRT SNLASGVPSRFSGSGSG TDFTLTISSLQPEDFATY YCQQYHSYPPTFGGGT KVEIK SEQ ID NO: 36 LC-hDR5-05 DIQLTQSPSSLSASVGD RVTITCSASSSVSYMYW YQQKPGKAPKPWIYRT SNLASGVPSRFSGSGSG TDFTLTISSLQPEDFATY YCQQYHSYPPTFGGGT KVEIKRTVAAPSVFIFPP SDEQLKSGTASVVCLLN NFYPREAKVQWKVDN ALQSGNSQESVTEQDS KDSTYSLSSTLTLSKADY EKHKVYACEVTHQGLSS PVTKSFNRGEC SEQ ID NO: 37 VH CONA-CDR1 GGSISSGDYF Conatumumab IgG1-DR5-CONA SEQ ID NO: 38 VH CONA-CDR2 IHNSGTT SEQ ID NO: 39 VH CONA-CDR3 ARDRGGDYYYGMDV SEQ ID NO: 40 VH CONA QVQLQESGPGLVKPSQ TLSLTCTVSGGSISSGDY FWSWIRQLPGKGLECIG HIHNSGTTYYNPSLKSR VTISVDTSKKQFSLRLSS VTAADTAVYYCARDRG GDYYYGMDVWGQGTT VTVSS SEQ ID NO: 41 VL CONA-CDR1 QGISRSY VL CONA-CDR2 GAS SEQ ID NO: 42 VL CONA-CDR3 QQFGSSPWT SEQ ID NO: 43 VL CONA EIVLTQSPGTLSLSPGER ATLSCRASQGISRSYLA WYQQKPGQAPSLLIYG ASSRATGIPDRFSGSGS GTDFTLTISRLEPEDFAV YYCQQFGSSPWTFGQG TKVEIK SEQ ID NO: 44 VH DR5-chTRA8 CDR1 GFTFSSYV SEQ ID NO: 45 VH DR5-chTRA8 CDR2 ISSGGSYT SEQ ID NO: 46 VH DR5-chTRA8 CDR3 ARRGDSMITTDY SEQ ID NO: 47 HC-DR5-chTRA8 EVMLVESGGGLVKP GGSLKLSCAASGFT FSSYVMSWVRQTPE KRLEWVATISSGGS YTYYPDSVKGRFTIS RDNAKNTLYLQMSS LRSEDTAMYYCARR GDSMITTDYWGQG TTLTVSSASTKGPSV FPLAPSSKSTSGGTA ALGCLVKDYFPEPVT VSWNSGALTSGVHT FPAVLQSSGLYSLSS VVTVPSSSLGTQTYI CNVNHKPSNTKVDK RVEPKSCDKTHTCPP CPAPELLGGPSVFLF PPKPKDTLMISRTPE VTCVVVDVSHEDPE VKFNWYVDGVEVHN AKTKPREEQYNSTYR VVSVLTVLHQDWLN GKEYKCKVSNKALPA PIEKTISKAKGQPRE PQVYTLPPSREEMTK NQVSLTCLVKGFYPS DIAVEWESNGQPEN NYKTTPPVLDSDGSF FLYSKLTVDKSRWQ QGNVFSCSVMHEAL HNHYTQKSLSLSPG K SEQ ID NO: 48 VL DR5-chTRA8 CDR1 QDVGTA SEQ ID NO: VL DR5-chTRA8 CDR2 WAS SEQ ID NO: 49 VL DR5-chTRA8 CDR3 QQYSSYRT SEQ ID NO: 50 LC-DR5-chTRA8 DIVMTQSHKFMSTS VGDRVSITCKASQD VGTAVAWYQQKPG QSPKLLIYWASTRH TGVPDRFTGSGSGT DFTLTISNVQSEDLA DYFCQQYSSYRTFG GGTKLEIKRTVAAPS VFIFPPSDEQLKSGT ASVVCLLNNFYPREA KVQWKVDNALQSG NSQESVTEQDSKDS TYSLSSTLTLSKADY EKHKVYACEVTHQG LSSPVTKSFNRGEC
EXAMPLES
Example 1: Antibodies and Antigens
[0362] Expression constructs for antibodies For antibody expression variable heavy (VH) chain and Variable light (VL) chain sequences were cloned in pcDNA3.3 expression vectors containing IgG1 heavy chain (HC) and light chain (LC) constant regions. Desired mutations were introduced either by gene synthesis or site directed mutagenesis. Antibodies mentioned in this application have VH and VL sequences derived from previously described chimeric human/mouse DR5 antibodies DR5-01 and DR5-05 (based on EP2684896A1), humanized DR5 antibodies hDR5-01 and hDR5-05 (based on WO2014/009358), IgG1-CONA (based on U.S. Pat. No. 7,521,048 B2 and WO2010/138725), IgG1-chTRA8 (based on EP1506285B1 and U.S. Pat. No. 7,244,429B2), IgG1-DR5-H48-2 (based on US 2004 0214235 A1), IgG1-DR4-T1014G03 (based on U.S. Pat. No. 7,361,341), and IgG1-FAS-E09 (based on Chodorge et al., Cell Death Differ. 2012 July; 19(7): 1187-1195). In some of the examples the human IgG1 antibody b12, a gp120-specific antibody was used as a negative control (Barbas et al., J Mol Biol. 1993 Apr. 5; 230(3):812-23).
Transient Expression
[0363] Antibodies were expressed as IgG1,K. Plasmid DNA mixtures encoding both heavy and light chains of antibodies were transiently transfected in Expi293F cells (Life technologies, USA) using 293fectin (Life technologies) essentially as described by Vink et al. (Vink et al., Methods, 65 (1), 5-10 2014).
Purification and Analysis of Proteins
[0364] Antibodies were purified by immobilized protein G chromatography. His-tagged recombinant protein was purified by immobilized metal affinity chromatography. Protein batches were analyzed by a number of bioanalytical assays including SDS-PAGE, size exclusion chromatography and measurement of endotoxin levels.
Generation of Bispecific Antibodies
[0365] Bispecific IgG1 antibodies were generated by Fab-arm-exchange under controlled reducing conditions. The basis for this method is the use of complimentary CH3 domains, which promote the formation of heterodimers under specific assay conditions as described in WO2011/131746 (Labrijn et al., Proc Natl Acad Sci USA. 2013 Mar. 26; 110(13):5145-50). To create antibody pairs with complementary CH3 domains, the F405L mutation (EU numbering) was introduced in IgG1-DR5-05, IgG1-DR5-05-E430G and IgG1-DR5-05-E345K; and the K409R mutation was introduced in IgG1-DR5-01, IgG1-DR5-01-E430G, IgG1-DR5-01-E345K and IgG1-CONA-E430G. To generate bispecific antibodies, two parental complementary antibodies, each antibody at a final concentration of 0.5 mg/mL, were incubated with 75 mM 2-mercaptoethylamine-HCl (2-MEA) in a total volume of 100 .mu.L TE at 31.degree. C. for 5 hours. The reduction reaction was stopped by removing the reducing agent 2-MEA using spin columns (Microcon centrifugal filters, 30 k, Millipore) according to the manufacturer's protocol. In this way the bispecific antibodies IgG1-DR5-01-K409R.times.IgG1-DR5-05-F405L (BsAb DR5-01-K409R.times.DR5-05-F405L), IgG1-DR5-01-K409R-E430G.times.IgG1-DR5-05-F405L-E430G (BsAb DR5-01-K409R-E430G.times.DR5-05-F405L-E430G), IgG1-DR5-01-K409R-E345K.times.IgG1-DR5-05-F405L-E345K (BsAb DR5-01-K409R-E345K.times.DR5-05-F405L-E345K) and IgG1-DR5-CONA-K409R-E430G.times.IgG1-DR5-05-F405L-E345K (BsAb DR5-CONA-K409R-E430G.times.DR5-05-F405L-E345K) were generated.
Example 2: Introduction of a Hexamerization-Enhancing Mutation does not Affect Binding of IgG1-DR5-01-K409R, IgG1-DR5-05-F405L and Bispecific Antibody IgG1-DR5-01-K409R.times.DR5-05-F405L to DR5-Positive Human Colon Cancer Cells
[0366] Binding of purified antibody variants of IgG1-DR5-01-K409R, IgG1-DR5-05-F405L and bispecific antibody IgG1-DR5-01-K409R.times.IgG1-DR5-05-F405L (BsAb DR5-01-K409R.times.DR5-05-F405L) with and without a hexamerization-enhancing mutation (E430G or E345K) to human colon cancer cells COLO 205 was analyzed by FACS analysis. Cells were harvested by pooling the culture supernatant containing non-adherent cells and trypsinized adherent COLO 205 cells. Cells were centrifuged for 5 minutes at 1,200 rpm and resuspended in 10 mL culture medium [RPMI 1640 with 25 mM Hepes and L-Glutamine (Lonza Cat nr BE12-115F)+10% Donor Bovine Serum with Iron (Life Technologies Cat nr 10371-029)+50 Units Penicillin/50 Units Streptomycin (Lonza Cat nr DE17-603E)]. Cells were counted, centrifuged again and resuspended in FACS buffer at a concentration of 0.3.times.10.sup.6 cells/mL. The next steps were performed at 4.degree. C. 100 .mu.L cell suspension samples (30,000 cells per well) were seeded in polystyrene 96-well round-bottom plates and pelleted by centrifugation at 300.times.g for 3 minutes at 4.degree. C. Cells were resuspended in 50 .mu.L samples of a serial dilution antibody preparation series (range 0 to 10 .mu.g/mL final concentrations in 5-fold dilutions) and incubated for 30 minutes at 4.degree. C. Plates were centrifuged at 300.times.g for 3 minutes at 4.degree. C. and cells were washed twice with 150 .mu.L FACS buffer. Cells were incubated with 50 .mu.L secondary antibody R-PE-conjugated goat-anti-human IgG F(ab')2 (Jackson ImmunoResearch; Cat nr 109-116-098; 1/100) for 30 minutes at 4.degree. C. protected from light. Cells were washed twice with 150 .mu.L FACS buffer, resuspended in 100 .mu.L FACS buffer, and antibody binding was analyzed on a FACS Canto II (BD Biosciences) by recording 5,000 events. Binding curves were analyzed using non-linear regression analysis (sigmoidal dose-response with variable slope) using GraphPad Prism software.
[0367] FIG. 2A shows that the antibodies IgG1-DR5-01-K409R-E430G and IgG1-DR5-01-K409R-E345K showed similar dose-dependent binding to human colon cancer cells COLO 205 as IgG1-DR5-01-K409R. FIG. 2B shows that the antibodies IgG1-DR5-05-F405L-E430G and IgG1-DR5-05-F405L-E345K showed similar dose-dependent binding to COLO 205 cells as IgG1-DR5-05-F405L. FIG. 2C shows that BsAb DR5-01-K409R-E430G.times.DR5-05-F405L-E430G and BsAb DR5-01-K409R-E345K.times.DR5-05-F405L-E345K showed similar dose-dependent binding to COLO 205 cells as BsAb DR5-01-K409R.times.DR5-05-F405L. These data indicate that introduction of the hexamerization-enhancing mutations E430G or E345K did not affect binding of antibodies IgG1-DR5-01-K409R, IgG1-DR5-05-F405L and BsAb DR5-01-K409R.times.DR5-05-F405L on DR5-positive COLO 205 cells.
Example 3: Introduction of a Hexamerization-Enhancing Mutation does not Affect Binding of DR4 Antibody to Soluble Human DR4
[0368] Binding of purified antibody variants of IgG1-DR4-T1014G03 with and without hexamerization-enhancing mutation E430G to coated human soluble DR4 was analyzed in a sandwich enzyme-linked immunosorbent assay (ELISA). 96-well flat bottom ELISA plates (Greiner bio-one; Cat nr 655092) were coated overnight at 4.degree. C. with 2 .mu.g/mL sTRAIL-R1 (Peprotech cat nr 310-18) in 100 .mu.L PBS. The wells were washed three times with PBST [PBS with 0.05% Tween-20 (Sigma-Aldrich; Cat nr 63158)]. The wells were blocked by adding 200 .mu.L PBSA [PBS with 1% Bovine Serum Albumin (BSA; Roche Cat #10735086001)] and incubated for 1 hour at room temperature while shaking. The wells were washed three times with PBST. Next, antibody samples of IgG1-DR4-T1014G03-K409R or IgG1-DR4-T1014G03-K409R-E430G (range 0 to 2,000 ng/mL final concentrations in 3-fold dilutions) were added in a total volume of 100 .mu.L PBSTA (PBST with 0.2% BSA) and incubated for 1.5 hour at room temperature while shaking. After washing three times with PBST, wells were incubated on an ELISA shaker with 100 .mu.L Horseradish Peroxidase (HRP)-conjugated goat anti-human IgG Fc.gamma. antibody (Jackson ImmunoResearch; Cat nr. 109-035-098; 1:10.000) in PBSTA for 1.5 hour at room temperature. After washing three times with PBST, the reaction was visualized through an incubation with 100 .mu.L 2,2'-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid [ABTS (Roche; Cat nr 11112597001)] at RT protected from light. Fluorescence at 405 nm was measured on an ELISA reader (BioTek ELx808 Absorbance Microplate Reader).
[0369] FIG. 3 shows that the antibodies IgG1-DR4-T1014G3-K409R and IgG1-DR4-T1014G3-K409R-E430G showed similar dose-dependent binding to coated soluble receptor, indicating that introduction of the hexamerization-enhancing mutation E430G did not affect binding of the antibody to its target.
Example 4: Introduction of a Hexamerization-Enhancing Mutation Improves the Efficacy of Cell Death Induction by DR5 Antibodies
[0370] Viability assays were performed to study the effect of introducing hexamerization-enhancing mutation E345K or E430G in different DR5 antibodies to induce killing of human colon cancer cells COLO 205 or HCT116. COLO 205 cells were harvested by pooling the culture supernatant containing non-adherent cells and trypsinized adherent cells. HCT 116 cells were harvested by trypsinization. Cells were passed through a cell strainer, pelleted by centrifugation for 5 minutes at 1,200 rpm and resuspended in culture medium at a concentration of 0.5.times.10.sup.5 cells/mL [COLO 205: RPMI 1640 with 25 mM Hepes and L-Glutamine (Lonza Cat nr BE12-115F)+10% Donor Bovine Serum with Iron (DBSI; Life Technologies Cat nr 10371-029)+50 Units Penicillin/50 Units Streptomycin (Pen/Strep; Lonza Cat nr DE17-603E); HCT 116: McCoy's5A Medium with L-Glutamine and Hepes (Lonza, Cat nr BE12-168F)+10% DBSI+Pen/Strep]. 100 .mu.L of the single cell suspension (5,000 cells per well) was seeded in polystyrene 96-well flat-bottom plates (Greiner Bio-One, Cat nr 655182). 50 .mu.L of serial dilution antibody preparation series (range 0.05 to 20,000 ng/mL final concentrations in 5-fold dilutions) were added and incubated for 3 days at 37.degree. C. The viability of the cultured cells was determined in a CellTiter-Glo luminescent cell viability assay (Promega, Cat nr G7571) that quantifies the ATP present, which is an indicator of metabolically active cells. From the kit, 20 .mu.L luciferin solution reagent was added per well and mixed by shaking the plate for 2 minutes at 500 rpm. Next, plates were incubated for 1.5 hours at 37.degree. C. 100 .mu.L supernatant was transferred to a white OptiPlate-96 (Perkin Elmer, Cat nr 6005299) and luminescence was measured on an EnVision Multilabel Reader (PerkinElmer). Data were analyzed and plotted using non-linear regression (sigmoidal dose-response with variable slope) using GraphPad Prism software. When samples with 5 .mu.M staurosporine (Sigma Aldrich, Cat nr S6942) were included as positive control, the percentage viable cells was calculated using the following formula: % viable cells=[(luminescence antibody sample-luminescence staurosporine sample)/(luminescence no antibody sample-luminescence staurosporine sample)]*100. For the experiments where no staurosporine control sample was included, data are presented as Luminescence.
[0371] E345K variants of IgG1-DR5-01-K409R and IgG1-DR5-05-F405L were tested on COLO 205. E430G-variants of IgG1-DR5-01-K409R, IgG1-DR5-05-F405L and IgG1-CONA-K409R were tested on both COLO 205 and HCT116 cells. IgG1-CONA was also tested as RGY-variant, a triple mutant E345K/E430G/S440Y that occurs as hexamer in solution (Diebolder et al., Science. 2014 Mar. 14; 343(6176):1260-3). IgG1-H48-2-F405L and IgG1-DR5-chTRA8-F405L were tested as E430G-variant on HCT116 cells. FIG. 4 shows that introduction of the hexamerization-enhancing mutations enhanced the potency of the different DR5 antibodies in COLO 205 and HCT 116 colon cancer cells.
Example 5: Introduction of a Hexamerization-Enhancing Mutation Improves the Efficacy of a DR4 Antibody to Induce Cell Death
[0372] A viability assay was performed to study the effect of introducing hexamerization-enhancing mutation E430G in DR4 antibody IgG1-DR4-T1014G03-K409R to induce killing of BxPC-3 human pancreatic cancer cells. Cells were harvested by trypsinization and passed through a cell strainer. Cells were pelleted by centrifugation for 5 minutes at 1,200 rpm and resuspended in culture medium at a concentration of 0.5.times.10.sup.5 cells/mL [RPMI 1640 with 25 mM Hepes and L-Glutamine (Lonza Cat nr BE12-115F)+10% DBSI+Pen/Strep]. 100 .mu.L of the single cell suspensions (5,000 cells per well) were seeded in polystyrene 96-well flat-bottom plates (Greiner Bio-One, Cat nr 655182) and incubated overnight at 37.degree. C. 50 .mu.L of serial dilution antibody preparation series (range 0.0006 to 40 .mu.g/mL final concentrations in 4-fold dilutions) were added and incubated for 3 days at 37.degree. C. As a negative and positive control, cells were incubated without antibody or with 5 .mu.M staurosporine, respectively. The viability of the cultured cells was determined in a CellTiter-Glo luminescent cell viability assay as described in Example 4. FIG. 5 shows that introduction of the hexamerization-enhancing mutation E430G enabled the DR4 antibody IgG1-DR4-T1014G03-K409R-E430G to induce dose-dependent killing of BxPC-3 pancreatic cancer cells, whereas the antibody without the E430G mutation was unable to induce killing at the tested antibody concentrations.
Example 6: Introduction of Hexamerization-Enhancing Mutations Improves the Efficacy of Cell Death Induction by a FAS Antibody
[0373] A viability assays was performed to study the effect of introducing hexamerization-enhancing mutations E345K/E430G/S440Y (RGY) in the FAS antibody IgG1-FAS-E09 to induce killing of Jurkat human T lymphocytes (ATTC TIB-152.TM.). Jurkat cells were harvested and resuspended in culture medium at a concentration of 0.3.times.10.sup.6 cells/mL (RPMI 1640 with 25 mM Hepes and L-Glutamine+10% Cosmic Calf Serum (CCS, Perbio Cat nr SH30087.03)+Pen/Strep). 100 .mu.L of the single cell suspension (30,000 cells per well) was seeded in polystyrene 96-well flat-bottom plates (Greiner Bio-One, Cat nr 655182). 50 .mu.L of serial dilution antibody preparation series (range 0.005 to 10,000 ng/mL final concentrations in 5-fold dilutions) were added and incubated for 3 days at 37.degree. C. The viability of the cultured cells was determined by TOPRO-3 iodine. TOPRO-3 binds to DNA but cannot pass intact plasma and nuclear membranes and will therefore only stain dying cells that have decreased membrane integrity. Cells were resuspended and transferred to a U-bottom 96-Wells plate (Greiner, Cat nr 650101). Cells were pelleted by centrifugation for 3 minutes at 300.times.g and washed with 150 .mu.L FACS buffer. Cells were pelleted by centrifugation for 3 minutes at 300.times.g and resuspended in 100 .mu.L FACS buffer supplemented with TOPRO-3 iodine (1:1,000; final concentration 1 .mu.M; Life Technologies, Cat nr T3605). TOPRO-3 staining was analyzed on a FACS Canto II (BD Biosciences) by recording 20,000 events. Data were analyzed and plotted using non-linear regression (sigmoidal dose-response with variable slope) using GraphPad Prism software. FIG. 6 shows the percentage viable cells, as calculated from the percentage TOPRO-3-negative cells. Introduction of the hexamerization-enhancing mutations RGY enabled the FAS antibody IgG1-FAS-E09 to induce dose-dependent killing of Jurkat human T lymphocytes, whereas the antibody without the E345R/E430G/S440Y triple mutation was unable to induce killing at the tested antibody concentrations.
Example 7: Introduction of Hexamerization-Enhancing Mutations Improves the Efficacy of Cell Death Induction by the Antibody Combination IgG1-DR5-01-K409R+IgG1-DR5-05-F405L and by the BsAb DR5-01-K409R.times.DR5-05-F405L
[0374] The effect of the hexamerization-enhancing mutation E345K or E430G on the capacity of the antibody combination IgG1-DR5-01-K409R+IgG1-DR5-05-F405L to kill human colon cancer cells COLO 205 and HCT116 was studied in a viability assay as described in Example 4. Also the effect of introducing the E345K or E430G mutation in the BsAb DR5-01-K409R.times.DR5-05-F405L was tested on COLO 205 or HCT116. FIG. 7 shows that the antibody combinations IgG1-DR5-01-K409R-E430G+IgG1-DR5-05-F405L-E430G and IgG1-DR5-01-K409R-E345K+IgG1-DR5-05-F405L-E345K showed enhanced potency compared to the antibody combination IgG1-DR5-01-K409R+IgG1-DR5-05-F405L without the E345K or E430G hexamerization-enhancing mutation on both COLO 205 (FIG. 7 A) and HCT116 cells (FIG. 7 C). The BsAb DR5-01-K409R-E430G.times.DR5-05-F405L-E430G also showed enhanced potency compared to the BsAb DR5-01-K409R.times.DR5-05-F405L without the E430G hexamerization-enhancing mutation on both COLO 205 (FIG. 7 B) and HCT116 cells (FIG. 7 D). The BsAb DR5-01-K409R-E345K.times.DR5-05-F405L-E345K showed enhanced potency compared to the BsAb DR5-01-K409R.times.DR5-05-F405L without the E430G hexamerization-enhancing mutation on HCT116 cells (FIG. 7 E)
Example 8: Introduction of a Hexamerization-Enhancing Mutation Improves the Efficacy of Cell Death Induction by the Combination of IgG1-hDR5-01-G56T+IgG1-hDR5-05 Antibodies
[0375] The effect of the hexamerization-enhancing mutation E430G on the capacity of the antibody combination IgG1-hDR5-01-G56T+IgG1-hDR5-05 to kill HCT15 colon and BxPC-3 pancreatic cancer cells was studied in a viability assay. Cells were harvested by trypsinization and passed through a cell strainer. Cells were pelleted by centrifugation for 5 minutes at 1,200 rpm and resuspended in culture medium at a concentration of 0.5.times.10.sup.5 cells/mL (RPMI 1640 with 25 mM Hepes and L-Glutamine (Lonza Cat nr BE12-115F)+10% DBSI+Pen/Strep). 100 .mu.L of the single cell suspensions (5,000 cells per well) were seeded in polystyrene 96-well flat-bottom plates (Greiner Bio-One, Cat nr 655182) and incubated overnight at 37.degree. C. 50 .mu.L antibody samples of serial dilution antibody preparation series (range 0.3 to 20,000 ng/mL final concentrations in 4-fold dilutions) were added and incubated for 3 days at 37.degree. C. As negative and positive control, cells were incubated without antibody and with 5 .mu.M staurosporine, respectively. The viability of the cell cultures was determined in a CellTiter-Glo luminescent cell viability assay as described in Example 4. FIG. 8 shows that the antibody combination IgG1-hDR5-01-G56T-E430G+IgG1-hDR5-05-E430G showed dose-dependent killing on both BxPC-3 (FIG. 8A) and HCT15 cells (FIG. 8B), whereas the antibody combination without the E430G hexamerization-enhancing mutation induced little to no killing at the tested antibody concentrations.
Example 9: Cell Death Induction by the Antibody Combination IgG1-hDR5-01-G56T-E430G+IgG1-hDR5-05-E430G Requires Fc-Fc Interactions to Form Hexamers
[0376] To analyze the requirement of antibody hexamer formation by IgG1-hDR5-01-G56T-E430G and IgG1-hDR5-05-E430G to induce cell death, we made use of the self-repulsing mutations K439E and S440K (Diebolder et al., Science. 2014 Mar. 14; 343(6176):1260-3). The Fc repulsion between antibodies that is introduced by the presence of either K439E or S440K in one IgG1 antibody or a combination of antibodies results in inhibition of hexamerization, even in the presence of a hexamerization enhancing mutation such as E345K or E430G (WO2013/0044842). The repulsion by the K439E and S440K mutations is neutralized by combining both mutations in a mixture of two antibodies each harboring one or the other mutation, resulting in restoration of the Fc-Fc interactions and hexamerization.
[0377] For both IgG1-hDR5-01-G56T-E430G and IgG1-hDR5-05-E430G, variants with either the K439E or S440K mutation were generated and tested in all different combinations. A viability assay was performed with serial dilution antibody preparation series ranging from 0.3 to 20,000 ng/mL total concentrations in 4-fold dilutions on BxPC-3 pancreatic and HCT-15 colon cancer cells as described in Example 4.
[0378] FIG. 9 shows that the antibody combinations with IgG1-hDR5-01-G56T-E430G and IgG1-hDR5-05-E430G variants harboring both the same repulsion mutation (K439E or S440K) showed strongly diminished killing efficacy in BxPC-3 (FIG. 9A) and HCT-15 cells (FIG. 9B). Killing efficacy was restored when repulsion was neutralized by combining two antibodies each having one of the complementary mutations K439E or S440K. These data indicate that hexamerization by Fc-Fc interactions is required for the induction of cell death by IgG1-hDR5-01-G56T-E430G and IgG1-hDR5-05-E430G.
Example 10: Antibody Fc-Fc Interactions are Involved in DR5 Clustering and Induction of Apoptosis by the Antibody Combination IgG1-hDR5-01-G56T-E430G+IgG1-hDR5-05-E430G with Hexamerization Enhancing Mutations
[0379] To test the involvement of Fc-Fc-mediated antibody hexamerization in the induction of cell death by the antibody combination IgG1-hDR5-01-G56T-E430G+IgG1-hDR5-05-E430G, we made use of the 13-residue peptide DCAWHLGELVWCT (DeLano et al., Science 2000 Feb. 18; 287(5456):1279-83) that binds the Fc in a region containing the core amino acids in the hydrophobic patch that are involved in Fc-Fc interactions (Diebolder et al., Science. 2014 Mar. 14; 343(6176):1260-3). A viability assay on BxPC-3 cells was performed as described in Example 4 for the antibody combination IgG1-hDR5-01-G56T-E430G+IgG1-hDR5-05-E430G in presence or absence of the DCAWHLGELVWCT peptide. Briefly, after overnight incubation of the cells at 37.degree. C., culture medium was removed and replaced by 100 .mu.L culture medium containing serial diluted peptide concentrations (range 0 to 100 .mu.g/mL final concentrations) of the Fc-binding DCAWHLGELVWCT peptide, a non-specific control peptide GWTVFQKRLDGSV, or no peptide. Next, 50 .mu.L antibody samples (833 ng/mL final concentration) were added and incubated for 3 days at 37.degree. C. The capacity of the antibody combination IgG1-hDR5-01-G56T-E430G+IgG1-hDR5-05-E430G to induce killing of BxPC-3 cells was strongly inhibited by 100 .mu.g/mL Fc-binding DCAWHLGELVWCT peptide (FIG. 10). These data indicate the involvement of Fc interactions in the capacity of the antibody combination IgG1-hDR5-01-G56T-E430G+IgG1-hDR5-05-E430G with hexamerization-enhancing mutations to induce DR5 clustering on the cell surface of cancer cells and induction of apoptosis.
Example 11: Capacity of the Antibody Combination IgG1-DR5-01-K409R-E430G+IgG1-DR5-05-F4051-E430G to Induce Target Cell Killing in Different Cancer Cell Lines
[0380] A viability assay was performed to study the capacity of the antibody combination IgG1-DR5-01-K409R+IgG1-DR5-05-F405L with and without the hexamerization-enhancing mutation E430G to induce killing of COLO 205, HCT-15, HCT 116, HT-29 and SW480 colon cancer, BxPC-3, HPAF-II and PANC-1 pancreatic cancer, SNU-5 gastric cancer, A549 and SK-MES-1 lung cancer, and A375 skin cancer cells. The assay was performed as described in Example 4, with the exception that here a fixed antibody concentration of 10 .mu.g/mL was used. Medium compositions of cell lines not previously described are as follows: SW480: RPMI 1640 with 25 mM Hepes and L-Glutamine+10% DBSI+Pen/Strep; HT-29: McCoy's5A Medium with L-Glutamine and Hepes+10% DBSI+Pen/Strep; HPAF-II and SK-MES-1: Eagle's Minimum Essential Medium (EMEM, ATCC Cat nr 30-2003)+10% DBSI+Pen/Strep; PANC-1 and A375: DMEM 4.5 g/L Glucose without L-Gln with HEPES (Lonza Cat nr LO BE12-709F)+10% DBSI+1 mM L-Glutamine (Lonza Cat nr BE17-605E)+Pen/Strep; SNU-5: IMDM (Lonza Cat nr BE12-722F)+10% DBSI+Pen/Strep; A549: F-12K Medium (ATCC Cat nr 30-2004)+10% DBSI+1 mM L-Glutamine+Pen/Strep).
[0381] For all tested cell lines, the percentage viable cells was significant lower after incubation with 10 .mu.g/mL of the antibody combination IgG1-DR5-01-K409R-E430G+IgG1-DR5-05-F405L-E430G than after incubation with the non-target binding negative control antibody IgG1-b12 (FIG. 11). In all but two of the tested cell lines, the efficacy of the antibody combination IgG1-DR5-01-K409R-E430G+IgG1-DR5-05-F405L-E430G was significant better than for the combination IgG1-DR5-01-K409R+IgG1-DR5-05-F405L without hexamerization-enhancing mutation. These data indicate that the combination of DR5 antibodies with hexamerization-enhancing mutations IgG1-DR5-01-K409R-E430G+IgG1-DR5-05-F405L-E430G was very effective in killing of cancer target cells of different origin, including colon, pancreatic, gastric, lung and skin cancer, without the requirement of a secondary cross-linking agent.
Example 12: Introduction of Hexamerization-Enhancing Mutations Improves the Efficacy of Cell Death Induction by the Antibody Combination IgG1-CONA-K409R+IgG1-DR5-05-F405L and BsAb CONA-K409R.times.DR5-05-F405L
[0382] The effect of the hexamerization-enhancing mutations on the capacity of the antibody combination IgG1-DR5-01-K409R+IgG1-CONA and BsAb CONA-K409R.times.DR5-05-F405L to kill HCT116 colon cancer cells was studied in a viability assay as described in Example 4. FIG. 12 shows that the antibody combination IgG1-CONA-K409R-E430G+IgG1-DR5-05-F405L-E345K and BsAb CONA-K409R-E430G.times.DR5-05-F405L-E345K with hexamerization-enhancing mutations showed enhanced efficacy in killing of HCT116 cells compared to the combination and bispecific antibody without the hexamerization-enhancing mutations E430G or E345K.
Example 13: The Potency of Antibody Combination IgG1-DR5-01-K409R-E430G+IgG1-DR5-05-F4051-E430G and BsAb DR5-01-K409R-E430G.times.DR5-05-F4051-E430G with Hexamerization-Enhancing Mutations is Independent of Fc.gamma.R Binding by a Secondary Crosslinker
[0383] A viability assay was performed to compare the capacity of the antibody combinations with hexamerization mutation in the absence and presence of secondary antibody crosslinker to induce killing of COLO 205 colorectal and BxPC-3 and PANC-1 pancreatic cancer cells. For comparison, the DR5 antibodies IgG1-CONA and IgG1-chTRA8-F405L that are known to require a secondary antibody crosslinker to induce killing, were tested in the same settings. The viability assay was performed as described in Example 4 in the absence or presence of goat-anti-human IgG F(ab')2 (1/150; Jackson ImmunoResearch; Cat nr 109-006-098). DR5 antibodies IgG1-CONA and IgG1-chTRA8-F405L did not induce target cell killing in the absence of an Fc crosslinker (FIG. 13). Fc crosslinking induced killing by IgG1-DR5-CONA and IgG1-DR5-chTRA8-F405L in COLO 205 and BxPC-3 cells The antibody combination IgG1-DR5-01-K409R-E430G+IgG1-DR5-05-F405L-E430G and BsAb DR5-01-K409R-E430G.times.DR5-05-F405L-E430G induced significant killing compared to the negative control, both in presence or absence of a secondary Fc crosslinker. These data indicate that killing of COLO 205, BxPC-3 and PANC-1 cancer cells by the antibody combination IgG1-DR5-01-K409R-E430G+IgG1-DR5-05-F405L-E430G and BsAb DR5-01-K409R-E430G.times.DR5-05-F405L-E430G is independent of Fc.gamma.R-mediated binding by a secondary Fc crosslinker and that this crosslinker-independent killing is more efficient than for Fc.gamma.R-crosslinked IgG1-DR5-CONA and IgG1-DR5-chTRA8-F405L.
Example 14: The Antibody Combination IgG1-hDR5-01-E430G+IgG1-hDR5-05-E430G with the E430G Hexamerization-Enhancing Mutation Induce Caspase-Dependent Cytotoxicity
[0384] A viability assay was performed to compare the cytotoxicity of the combination of humanized antibodies IgG1-hDR5-01-E430G+IgG1-hDR5-05-E430G in the presence and absence of a caspase inhibitor. PANC-1 and BxPC3 pancreatic cancer cells were harvested by trypsinization and passed through a cell strainer. Cells were pelleted by centrifugation for 5 minutes at 1,200 rpm and resuspended in culture medium at a concentration of 0.5.times.10.sup.5 cells/mL. 100 .mu.L of the single cell suspensions (5,000 cells per well) were seeded in polystyrene 96-well flat-bottom plates (Greiner Bio-One, Cat nr 655182) and incubated overnight at 37.degree. C. 25 .mu.L pan-caspase inhibitor Z-Val-Ala-DL-Asp-fluoromethylketone (Z-VAD-FMK, 5 .mu.M end concentration in 150 .mu.L, Bachem, Cat nr 4026865.0005) was added to the cell cultures and incubated for one hour at 37.degree. C. before adding 25 .mu.L antibody sample of a serial dilution antibody preparation series (range 1 to 20 .mu.g/mL final concentrations in 4-fold dilutions) and further incubation for 3 days at 37.degree. C. As a positive control, cells were incubated with 5 .mu.M staurosporine (Sigma Aldrich, Cat nr S6942). Recombinant human TRAIL/APO-2L (eBioscience, Cat nr BMS356) was used at 6 .mu.g/mL final concentration. The viability of the cell cultures was determined in a CellTiter-Glo luminescent cell viability assay as described in Example 4. The antibody combination with hexamerization-enhancing mutations IgG1-hDR5-01-E430G+IgG1-hDR5-05-E430G was unable to reduce the viability of PANC-1 and BxPC3 pancreatic cancer cells in presence of the pan-caspase inhibitor Z-VAD-FMK, indicating that the combination of IgG1-hDR5-01-E430G+IgG1-hDR5-05-E430G induced caspase-dependent programmed cell death (FIG. 14). This was also shown for the natural DR5 ligand TRAIL.
Example 15: Cell Death Induction Upon Binding of the Antibody Combination IgG1-DR5-01-K409R-E430G+IgG1-DR5-05-F4051-E430G and BsAb DR5-01-K409R-E430G.times.DR5-05-F4051-E430G on COLO 205 Colon Cancer Cells, as Assessed by Annexin V/Propidium Iodide and Active Caspase-3 Staining
[0385] The kinetics of cell death induction was analyzed by Annexin V/Propidium Iodide (PI) double staining and active caspase-3 staining. Annexin-V binds phosphatidylserine that is exposed on the cell surface after initiation of programmed cell death, which is a reversible process. PI is a dye that intercalates into double-stranded DNA and RNA when it enters cells. Because PI cannot pass intact plasma and nuclear membranes, it will not stain living cells but only enter and stain dying cells that have decreased membrane integrity. Due to these characteristics, the Annexin V/PI double staining can be applied to discriminate between initiation (Annexin V-positive/PI-negative) and irreversible (Annexin V-positive/PI-positive) programmed cell death. Caspase-3 is activated by both the extrinsic death receptor-induced and intrinsic mitochondrial cell death pathways. Therefore, active caspase-3 is also a marker for initiation of the death cascade. The induction of cell death upon binding of the combination of IgG1-DR5-01-K409R-E430G+IgG1-DR5-05-F405L-E430G and BsAb DR5-01-K409R-E430G.times.DR5-05-F405L-E430G was analyzed in the DR5-positive COLO 205 colon cancer cells. Cells were harvested by pooling the culture supernatant containing non-adherent cells and trypsinized adherent cells. Cells were passed through a cell strainer, pelleted by centrifugation for 5 minutes at 1,200 rpm and resuspended in culture medium at a concentration of 0.2.times.10.sup.6 cells/mL. 500 .mu.L of the single cell suspensions (100,000 cells per well) were seeded in 24-wells flat-bottom culture plates (Greiner Bio-One, Cat nr 662160) and incubated for 16 hours at 37.degree. C. 500 .mu.L antibody sample was added (1 .mu.g antibody final concentration) and incubated for 5 hours or 24 hours at 37.degree. C. As a positive control, cells were incubated with 5 .mu.M staurosporine (Sigma Aldrich, Cat nr S6942). Cells were washed once with 250 .mu.L 1.times.PBS. Adherent cells were harvested by incubating with 100 .mu.L 0.05% trypsin for 10 minutes at 37.degree. C. 200 .mu.L medium was added to the trypsinized cells and cells were transferred to a 96-wells round-bottom FACS plate (Greiner Bio-One, Cat nr 650101) and pooled with the non-adherent cells. Cells were pelleted by centrifugation for 5 minutes at 1,200 rpm, resuspended in 200 .mu.L ice cold PBS and divided into two samples of 100 .mu.L in 96-Wells round-bottom FACS plates for the Annexin V/PI and active caspase-3 staining, respectively.
[0386] Annexin V/PI double staining was performed using the FITC Annexin V Apoptosis Detection Kit I (BD Pharmingen, Cat nr 556547). Cells were washed once with ice cold PBS and incubated in 50 .mu.L Annexin V/PI Staining Solution (Annexin V-FITC 1:00 and PI 1:25) for 15 minutes at 4.degree. C. Cells were washed with 100 .mu.L Binding Buffer, resuspended in 20 .mu.L Binding Buffer and fluorescence was measured on an iQue Screener (IntelliCyt) within 1 hour. Data were analyzed and plotted using GraphPad Prism software.
[0387] Active caspase-3 staining was performed using the PE Active Caspase-3 Apoptosis Kit (BD Pharmingen, Cat nr 550914). Cells were washed once with ice cold PBS, resuspended in 100 .mu.L Cytofix/Cytoperm Fixation and Permeabilization Solution and incubated for 20 minutes on ice. Cells were pelleted at room temperature, washed twice with 100 .mu.L 1.times. Perm/Wash Buffer and resuspended in 100 .mu.L PE Rabbit Anti-Active Caspase-3 (1:10) for an incubation of 30 minutes at room temperature. Cells were pelleted at room temperature, washed once with 100 .mu.L 1.times. Perm/Wash Buffer and resuspended in 20 .mu.L 1.times. Perm/Wash Buffer. Fluorescence was measured on an iQue Screener. Data were analyzed and plotted using GraphPad Prism software.
[0388] FIG. 15 shows that, after 5 hours of incubation, the combination of the chimeric antibodies IgG1-DR5-01-K409R-E430G+IgG1-DR5-05-F405L-E430G efficiently induced the early stages of cell death as indicated by an increase in the percentage of Annexin V-positive/PI-negative (FIG. 15A) and Active Caspase-3-positive cells (FIG. 15B), compared to the negative control antibody IgG1-b12. The percentage of Annexin V-positive/PI-negative and Active Caspase-3 positive cells was higher in cells treated with the combination of IgG1-DR5-01-K409R-E430G+IgG1-DR5-05-F405L-E430G compared to the combination of the DR5 antibodies without the E430G mutation (IgG1-DR5-01-K409R+IgG1-DR5-05-F405L) or any of the single antibodies. At the 5 hour time point, the percentage of AnnexinV/PI double-positive cells was comparable to background levels in all samples (FIG. 15C).
[0389] BsAb DR5-01-K409R-E430G.times.DR5-05-F405L-E430G efficiently induced the early stages of cell death as indicated by an increase in the percentage of Annexin V-positive/PI-negative (FIG. 15A) and Active Caspase-3-positive cells (FIG. 15B) after 5 hours incubation, compared to the negative control antibody IgG1-b12. The percentage of Annexin V-positive/PI-negative and Active Caspase-3 positive cells was higher in cells that had been treated with BsAb DR5-01-K409R-E430G.times.DR5-05-F405L-E430G compared to the bispecific antibody without the E430G mutation (BsAb DR5-01-K409R.times.DR5-05-F405L) or any of the monospecific antibodies. At the 5 hour time point, the percentage of AnnexinV/PI double positive cells was comparable to background levels in all samples (FIG. 15C).
[0390] After 24 hours incubation, the percentage of Annexin V/PI double-positive cells (FIG. 15D) was enhanced in samples treated with IgG1-DR5-01-K409R-E430G+IgG1-DR5-05-F405L-E430G, indicating that the cells had entered the irreversible stages of cell death. Also at this stage, the effect of the combination of IgG1-DR5-01-K409R-E430G+IgG1-DR5-05-F405L-E430G was stronger (larger increase in the percentage of Annexin V/PI double-positive cells (FIG. 15E)) than in samples treated with a combination of DR5 antibodies without the E430G mutation (IgG1-DR5-01-K409R+IgG1-DR5-05-F405L) or any of the single antibodies. At the same time point, the percentage of Active Caspase 3 positive cells was highest in cells treated with IgG1-DR5-01-K409R-E430G+IgG1-DR5-05-F405L-E430G.
[0391] After 24 hours incubation, the percentage of Annexin V/PI double-positive cells (FIG. 15D) was enhanced in samples treated with BsAb DR5-01-K409R-E430G.times.DR5-05-F405L-E430G, indicating that the cells had entered the irreversible stages of cell death. Also at this stage, the effect of BsAb DR5-01-K409R-E430G.times.DR5-05-F405L-E430G was stronger (larger increase in the percentage of Annexin V/PI double-positive cells (FIG. 15E) than in samples treated with the bispecific antibody without the E430G mutation (BsAb DR5-01-K409R.times.DR5-05-F405L) or any of the monospecific antibodies. At the same time point, the percentage of Active Caspase 3 positive cells was highest in cells treated with BsAb DR5-01-K409R-E430G.times.DR5-05-F405L-E430G.
[0392] These data indicate that the antibody combination IgG1-DR5-01-K409R-E430G+IgG1-DR5-05-F405L-E430G and BsAB DR5-01-K409R-E430G.times.DR5-05-F405L-E430G induced both the early and late stages of cell death in COLO 205 colon cancer cells, and did so more effectively than the antibody combination and BsAb without the E430G hexamerization enhancing mutation.
Example 16: Caspase-3 and -7 Activation Upon Binding of the Antibody Combination IgG1-DR5-01-K409R-E430G+IgG1-DR5-05-F4051-E430G or BsAb DR5-01-K409R-E430G.times.DR5-05-F4051-E430G with Hexamerization-Enhancing Mutation on COLO 205 Colon Cancer Cells
[0393] In Example 15 it was described that incubation with the antibody combination IgG1-DR5-01-K409R-E430G+IgG1-DR5-05-F405L-E430G induced caspase-3 activation in COLO 205 colon cancer cells. The percentage of active caspase-3-positive cells was higher after 5 hours than after 24 hours of incubation with the antibody combination. In this example, Caspase-3/7 activation was measured in time using the Caspase-Glo 3/7 assay (Promega, Cat nr G8091), in which a substrate with the Caspase-3/7 recognition motif DEVD releases aminoluciferin, a substrate of luciferase, upon cleavage. Cells were harvested by pooling the culture supernatant containing non-adherent cells and trypsinized adherent COLO 205. Cells were passed through a cell strainer, pelleted by centrifugation for 5 minutes at 1,200 rpm and resuspended in culture medium at a concentration of 0.8.times.10.sup.5 cells/mL. 25 .mu.L of the single cell suspensions (2,000 cells per well) were seeded in 384-wells culture plates (Perkin Elmer, Cat nr 6007680) and incubated for 16 hours at 37.degree. C. 25 .mu.L antibody sample was added (1 .mu.g antibody final concentration) and incubated for 1, 2, 5 and 24 hours at 37.degree. C. Plates were removed from the incubator to let the temperature decrease till room temperature. Cells were pelleted by centrifugation for three minutes at 300.times.g. 25 .mu.L supernatant was removed and replaced by 25 .mu.L Caspase-Glo 3/7 Substrate. After mixing by shaking for one minute at 500 rpm, the plates were incubated for one hour at room temperature. Luminescence was measured on an EnVision Multilabel Reader (PerkinElmer). In the time course of 1, 2 to 5 hours, the antibody combination IgG1-DR5-01-K409R-E430G+IgG1-DR5-05-F405L-E430G showed faster and stronger induction of caspase-3/7 activation than the combination of IgG1-DR5-01-K409R+IgG1-DR5-05-F405L without the hexamerization-enhancing mutation (FIG. 16A). Similarly BsAb DR5-01-K409R-E430G.times.DR5-05-F405L-E430G showed faster and stronger induction of caspase-3/7 activation than BsAb DR5-01-K409R.times.DR5-05-F405L without the hexamerization-enhancing mutation (FIG. 16B). After 24 hours, caspase-3/7 activation was almost reduced to baseline levels for all tested DR5 antibodies.
Example 17: Introduction of the K409R or F405L Mutation has No Effect on the Potency of Antibodies with a Hexamerization-Enhancing Mutation
[0394] In many of the experiments described in this application, the anti-death receptor antibodies contain in the IgG Fc domain the K409R or F405L (EU numbering) mutation. These mutations enable the generation of bispecific death receptor antibodies by Fab-arm-exchange reaction between a K409R-containing IgG1 and a F405L-containing IgG1 under controlled reducing conditions as described in WO2011/131746. Without Fab-arm exchange, human IgG1 antibodies bearing the K409R or F405L mutation are thought to show the same functional characteristics as wild type human IgG1 (Labrijn et al., Proc Natl Acad Sci USA. 2013 Mar. 26; 110(13):5145-50). Here we show that the presence of the K409R or F405L mutations has no effect on the capacity of the combination of the parental IgG1-DR5-01-K409R-E430G and IgG1-DR5-05-F405L-E430G antibodies to induce cell death in tumor cells in vitro. A viability assay was performed as described in Example 5 to compare the capacity of the antibody combination IgG1-DR5-01-K409R-E430G+IgG1-DR5-05-F405L-E430G with the capacity of the antibody combination IgG1-hDR5-01-E430G+IgG1-hDR5-05-E430G to induce killing of BxPC-3 pancreatic cancer cells.
[0395] The BxPC-3 pancreatic cancer cell line showed similar viability curves after incubation with the antibody combination IgG1-hDR5-01-K409R-E430G+IgG1-hDR5-05-F405L-E430G as with the antibody combination IgG1-hDR5-01-E430G+IgG1-hDR5-05-E430G (FIG. 17). These data indicate that the K409R and F405L mutations had no effect on the potency of the combination of the antibodies with E430G hexamerization enhancing mutation.
Example 18: Cancer Cell Kill Capacity of Different Antibody Ratios in the Combination IgG1-DR5-01-K409R-E430G+IgG1-DR5-05-F4051-E430G
[0396] A viability assay was performed as described in Example 5 to study the capacity of the antibody combination IgG1-DR5-01-K409R-E430G+IgG1-DR5-05-F405L-E430G to induce killing of BxPC-3 pancreatic cancer cells, when combined at different ratios of IgG1-DR5-01-K409R-E430G and IgG1-DR5-05-F405L-E430G. The antibodies were combined at different ratios of IgG1-DR5-01-K409R-E430G and IgG1-DR5-05-F405L-E430G, indicated as Ratio DR5-01-E430G:DR5-05-E430G of 100:0, 90:10, 80:20, 70:30, 60:40, 50:50, 40:60, 30:70, 20:80, 10:90 and 0:100. At 20 .mu.g/mL and 4 .mu.g/mL total antibody concentrations, killing was equally effective at all tested antibody ratios containing both antibodies IgG1-DR5-01-K409R-E430G and IgG1-DR5-05-F405L-E430G. At 0.8 .mu.g/mL and 0.16 .mu.g/mL total antibody concentrations, all tested antibody ratios containing both antibodies IgG1-DR5-01-K409R-E430G and IgG1-DR5-05-F405L-E430G induced killing (FIG. 18).
Example 19: Cancer Cell Kill Capacity of Different Antibody Ratios in the Combination IgG1-hDR5-01-G56T-E430G+IgG1-hDR5-05-E430G
[0397] A viability assay was performed as described in Example 5 to study the capacity of the antibody combination IgG1-hDR5-01-G56T-E430G+IgG1-hDR5-05-E430G to induce killing of BxPC-3 pancreatic and HCT-15 colon cancer cells, when combined at different antibody ratios (indicated in FIG. 19 as Ratio DR5-01-E430G:DR5-05-E430G of 100:0, 98:2, 96:4, 94:6, 92:8, 90:10, 50:50, 10:90, 8:92, 6:94, 4:96, 2:98 and 0:100) at final antibody concentrations of 10 .mu.g/mL for BxPC-3 and 20 .mu.g/mL for HCT-15. Killing was equally effective at all tested antibody ratios containing both antibodies IgG1-hDR5-01-G56T-E430G and IgG1-hDR5-05-E430G (FIG. 19).
Example 20: Effect of a Hexamerization-Enhancing Mutation on the In Vivo Efficacy of an Anti-DR5 Antibody in a Subcutaneous COLO 205 Colon Cancer Xenograft Model
[0398] The in vivo anti-tumor efficacy of IgG1-DR5-05-F405L-E430G was compared to that of IgG1-DR5-05-F405L without a hexamerization-enhancing mutation in a subcutaneous model with COLO 205 human colon cancer cells. At day 0, cells were harvested by pooling the culture supernatant containing non-adherent cells and trypsinized adherent cells. 3.times.10.sup.6 cells were injected in a volume of 200 .mu.L PBS into the flank of 6-11 weeks old female SCID mice (C.B-17/IcrHan.RTM.Hsd-Prkdc.sup.scid; Harlan). All experiments and animal handlings were approved by the local authorities, and were conducted according to all applicable international, national and local laws and guidelines. Tumor development was monitored at least twice per week by caliper (PLEXX) measurement as 0.52.times.(length).times.(width).sup.2. Tumors were measured until an endpoint tumor volume of 1,500 mm.sup.3, until tumors showed ulcerations, until serious clinical signs were observed, or until tumor growth blocked movements of the mouse. At day 6, the average tumor volume was .about.200 mm3 and the mice were sorted into groups with equal tumor size variance (Table below). Mice were treated by intraperitoneal (i.p.) injection of 100 .mu.g antibody in 200 .mu.L PBS on day 6 and 13 (5 mg/kg per dose). To check for correct antibody administration, blood samples were obtained for IgG serum determination three days after the first dose. One individual mouse had no detectable human IgG plasma level and was excluded from statistical analysis (Table below). For the other mice, human antibody plasma concentrations were according to the expectations when assuming a 2-compartment model with Vcen=50 mL/kg, Vs=100 mL/kg and an elimination half-life of 11.6 days (data not shown). Tumors were measured until 16 weeks after tumor inoculation.
Treatment Groups and Dosing
TABLE-US-00005 [0399] Dosing day # # after tumor mice analyzed Antibody Total antibody dose inoculation 7 7 IgG1-DR5-05- 100 .mu.g (5 mg/kg) 6, 13 F405L (100 .mu.g) 8 8 IgG1-DR5-05- 100 .mu.g (5 mg/kg) 6, 13 F405L-E430G (100 .mu.g) 8 7 IgG1-b12 100 .mu.g (5 mg/kg) 6, 13 (100 .mu.g)
[0400] FIG. 20A shows mean tumor volumes per treatment group in time. Complete tumor abrogation was observed for the anti-DR5 antibody with hexamerization-enhancing mutation (IgG1-DR5-05-F405L-E430G). In contrast, IgG1-DR5-05-F405L without hexamerization-enhancing mutation strongly inhibited tumor growth compared to IgG1-b12, but did not result in complete tumor abrogation.
[0401] FIG. 20B shows a Kaplan-Meier plot of tumor progression, with a cutoff set at a tumor volume >750 mm3. Compared to mice treated with negative control antibody IgG1-b12, tumor outgrowth was significantly delayed in the groups treated with anti-DR5 antibodies (Mantel-Cox analysis at tumor size cut-off 750 mm3: p<0.001). At the end of the study (day 112), the group of mice treated with IgG1-DR5-05-F405L-E430G showed significant less mice with tumor outgrowth than the IgG1-DR5-05-F405L without hexamerization-enhancing mutation group (p<0.001).
[0402] These data show that introduction of the E430G hexamerization-enhancing mutation in IgG1-DR5-05-F405L resulted in enhanced tumor inhibition in the subcutaneous COLO 205 colon cancer tumor model compared to IgG1-DR5-05-F405L without the hexamerization-enhancing mutation.
Example 21: Cell Death Induction by IgG1-FAS-E09 Variants with Hexamerization-Enhancing Mutations
[0403] Introduction of the hexamerization-enhancing mutations E345R/E430G/S440Y enabled the FAS antibody IgG1-FAS-E09 to induce dose-dependent killing of Jurkat human T lymphocytes as described in Example 6. To analyze the requirement of antibody Fc-Fc interactions by hexamerized IgG1-FAS-E09 variants to induce cell death, we made use of the self-repulsing mutations K439E and S440K in combination with the hexamerization-enhancing mutations E345R/E430G/S440Y (RGY) and E345R/E430G/Y4361(RGI), respectively (WO2014006217).
[0404] A viability assay on Jurkat human T lymphocytes was performed, essentially as described in Example 6. Briefly, 19,200 cells in 100 .mu.L per well were seeded in 96-well plates. 50 .mu.L of serial dilution antibody preparation series (range 0.0006 to 10 .mu.g/mL final concentrations in 6-fold dilutions) were added and incubated for 4 days at 37.degree. C. The viability of the cultured cells was determined by TOPRO-3 iodine as described in Example 6. TOPRO-3 staining was analyzed by flow cytometry on a BD LSRFORTESSA cell analyzer (BD Biosciences). Data were analyzed and plotted using non-linear regression (sigmoidal dose-response with variable slope) using GraphPad Prism software. FIG. 21 shows the percentage viable cells, as calculated from the percentage TOPRO-3-negative cells. Introduction of the hexamerization-enhancing mutations RGY enabled the FAS antibody IgG1-FAS-E09 to induce dose-dependent killing of Jurkat human T lymphocytes. Killing by IgG1-FAS-E09-RGY was inhibited by presence of the Fc-Fc repulsion mutation K439E in IgG1-FAS-E09-RGEY. Also IgG1-FAS-E09-RGIK, containing the repulsion mutation S440K did not induce killing of Jurkat cells. Killing efficacy was restored when Fc-Fc repulsion was neutralized by combining the two antibodies IgG1-FAS-E09-RGEY and IgG1-FAS-E09-RGIK, each having one of the complementary mutations K439E or S440K. These data illustrate that hexamerization by Fc-Fc interactions is required for the induction of cell death by IgG1-FAS-E09 variants with the hexamerization mutation RGY or RGI.
Example 22: Anti-DR5 Antibody IgG1-DR5-CONA with a Hexamerization-Enhancing Mutation E430G is Able to Kill Human Colon Cancer Cells
[0405] The present study illustrate the ability of the anti-DR5 antibody IgG1-DR5-CONA with the hexamerization-enhancing mutation E430G to kill attached human colon cancer cells COLO 205. COLO 205 cells were harvested as described in Example 4. 100 .mu.L of the single cell suspensions (5,000 cells per well) were seeded in 96-well flat-bottom plates and incubated overnight at 37.degree. C. 50 .mu.L samples of antibody concentration series (range 0.04 to 10 .mu.g/mL final concentrations in 4-fold dilutions) were added and incubated for 3 days at 37.degree. C. As a positive control, cells were incubated with 5 .mu.M staurosporine. The viability of the cell cultures was determined in a CellTiter-Glo luminescent cell viability assay as described in Example 4. Luminescence was measured on an EnVision Multilabel Reader (PerkinElmer). Data were analyzed and plotted using non-linear regression (sigmoidal dose-response with variable slope) using GraphPad Prism software. The percentage viable cells was calculated using the following formula: % viable cells=[(luminescence antibody sample-luminescence staurosporine sample)/(luminescence no antibody sample-luminescence staurosporine sample)]*100.
[0406] FIG. 22 shows that introduction of the hexamerization-enhancing mutation E430G resulted in dose-dependent killing by IgG1-DR5-CONA-E430G, whereas the parental wild type antibody IgG1-DR5-CONA was not able to kill attached COLO 205 colon cancer cells.
Example 23: Introduction of Hexamerization-Enhancing Mutation S440Y Improves the Efficacy of Anti-DR5 Antibodies to Induce Cell Death on Human Colon Cancer Cells
[0407] The effect of the hexamerization-enhancing mutation S440Y on the capacity of the single antibodies and the combination of IgG1-hDR5-01-G56T and IgG1-hDR5-05 to kill COLO 205 human colon cancer cells was studied in a viability assay. Cells were harvested and a viability assay was performed as described in Example 4. Briefly, 100 .mu.L single cell suspensions (5,000 cells per well) were seeded in 96-well plates. 50 .mu.L of serial dilution antibody preparation series (range 0.0003 to 20 .mu.g/mL final concentrations in 4-fold dilutions) were added and incubated for 3 days at 37.degree. C. The viability of the cultured cells was determined in a CellTiter-Glo luminescent cell viability assay as described in Example 4. Luminescence data were analyzed as described in Example 22.
[0408] FIG. 23A shows that introduction of the hexamerization-enhancing mutation S440Y resulted in dose-dependent killing by the single antibodies IgG1-hDR5-01-G56T-S440Y and IgG1-hDR5-05-S440Y, whereas the parental wild type antibodies IgG1-hDR5-01-G56T and IgG1-hDR5-05 were not able to kill COLO 205 colon cancer cells. Also the efficacy of the combination of IgG1-hDR5-01-G56T+IgG1-hDR5-05 was improved by introduction of the S440Y mutation in both antibodies, represented by the decreased EC50 (FIG. 23B).
Example 24: Introduction of the Hexamerization-Enhancing Mutation E430G Improves the Efficacy of Cell Death Induction by the Combination of Anti-DR5 Antibodies IgG1-DR5-CONA+IgG1-DR5-chTRA8
[0409] The competition between IgG1-DR5-CONA-K409R and IgG1-DR5-chTRA8-F405L for binding to the extracellular domain of DR5 was measured by sandwich binding assays in a sandwich enzyme-linked immunosorbent assay (ELISA). 96-well flat bottom ELISA plates (Greiner bio-one; Cat nr 655092) were coated overnight at 4.degree. C. with 2 .mu.g/mL DR5 antibody (IgG1-DR5-CONA-K409R or IgG1-DR5-chTRA8-F405L) in 100 .mu.L PBS. The wells were blocked by adding 200 .mu.L PBSA [PBS/1% Bovine Serum Albumin (BSA; Roche Cat #10735086001)] and incubated for 1 hour at room temperature. The wells were washed three times with PBST [PBS/0.05% Tween-20 (Sigma-Aldrich; Cat nr 63158)]. Next, DR5ECD-FcHistag (0.2 .mu.g/mL final concentration) and competing antibody (1 .mu.g/mL final concentration) were added in a total volume of 100 .mu.L PBSTA (PBST/0.2% BSA) and incubated for 1 hour at room temperature while shaking. After washing three times with PBST, wells were incubated on an ELISA shaker with 100 .mu.L biotinylated anti-His tag antibody (R&D Systems; Cat nr BAM050; 1:2.000) in PBSTA for one hour at room temperature. After washing three times with PBST, wells were incubated with streptavidin-labelled Poly-HRP (Sanquin; Cat nr M2032; 1:10.000) in PBSTA for 20 minutes at room temperature on an ELISA shaker. After washing three times with PBST, the reaction was visualized through an incubation with 100 .mu.L 2,2'-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid [ABTS (Roche; Cat nr 11112597001)] for 30 minutes at RT protected from light. The substrate reaction was stopped by adding an equal volume of 2% oxalic acid. Fluorescence at 405 nm was measured on an ELISA reader (BioTek ELx808 Absorbance Microplate Reader).
[0410] FIG. 24A shows binding competition expressed as percentage inhibition of DR5ECD-FcHisCtag binding to coated antibody in presence of competing antibody, relative to binding of DR5ECD-FcHisCtag in absence of competing antibody (% inhibition=100-[(binding in presence of competing antibody/binding in absence of competing antibody)]*100). Binding of DR5ECD-FcHistag to coated IgG1-DR5-CONA-K409R was not inhibited in the presence of soluble IgG1-DR5-chTRA8-F405L. Vice versa, binding of DR5ECD-FcHistag to coated IgG1-DR5-chTRA8-F405L was also not inhibited in the presence of soluble IgG1-DR5-CONA-K409R. These data illustrate that IgG1-DR5-CONA-K409R and IgG1-DR5-chTRA8-E430G did not compete with each other for binding of DR5ECD-FcHisCtag.
[0411] Next, the effect of the hexamerization-enhancing mutation E430G on the capacity of the combination of the non-crossblocking anti-DR5 antibodies IgG1-DR5-CONA+IgG1-DR5-chTRA-8 to kill attached BxPC-3 human pancreatic cancer cells was studied in a viability assay as described in Example 5. FIG. 24 shows that the antibody combination IgG1-DR5-CONA-E430G+IgG1-DR5-chTRA8-E430G with hexamerization-enhancing mutations showed increased dose-dependent killing of BxPC-3 cells compared to the combination of the parental antibodies without the E430G hexamerization-enhancing mutation.
Example 25: Effect of a Hexamerization-Enhancing Mutation on the In Vivo Efficacy of the Combination of Anti-DR5 Antibodies IgG1-hDR5-01-G56T+IgG1-hDR5-05 in a Subcutaneous HCT15 Colon Cancer Xenograft Model
[0412] The in vivo anti-tumor efficacy of the anti-DR5 antibody combination IgG1-hDR5-01-G56T-E430G+IgG1-hDR5-05-E430G was compared to that of IgG1-hDR5-01-G56T+IgG1-hDR5-05 without the E430G hexamerization-enhancing mutation in the subcutaneous HCT15 human colon cancer xenograft model at CrownBiosciences, Taicang, China. The cells were maintained in vitro as a monolayer culture in RPMI-1640 medium supplemented with 10% fetal bovine serum at 37.degree. C. in an atmosphere of 5% CO2 in air. Adherent cells in an exponential growth phase were harvested by trypsin-EDTA treatment. 5.times.10.sup.6 cells were injected in a volume of 100 .mu.L PBS into the flank of 7-9 weeks old female BALB/c nude mice. The care and use of animals during the study were conducted in accordance with the regulations of the Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC). Tumor volumes were measured twice weekly in two dimensions using a caliper, and the volume was expressed in mm.sup.3 using the formula: V=0.5 a.times.b.sup.2 where a and b are the long and short diameters of the tumor, respectively. Mice were assigned into groups using randomized block design and treatments were started when the mean tumor size reached 161 mm.sup.3 (8 mice per group). Mice were treated three times according to a Q7D regimen by i.v. injection of 10 .mu.g antibody (0.5 mg/kg, i.e. 0.25 mg/kg of each antibody in the combination). Mice in the control group were treated in parallel with 0.5 mg/kg IgG1-b12.
[0413] FIG. 25A shows mean tumor volumes per treatment group. The antibody combination IgG1-hDR5-01-G56T-E430G+IgG1-hDR5-05-E430G showed better tumor growth inhibition than IgG1-hDR5-01-G56T+IgG1-hDR5-05. FIG. 25B shows tumor volume per treatment group at day 21. The combination IgG1-hDR5-01-G56T-E430G+IgG1-hDR5-05-E430G inhibited tumor growth progression significantly better than an equivalent dose IgG1-hDR5-01-G56T+IgG1-hDR5-05 (Mann Whitney test (P<0.0011)). FIG. 25C shows a Kaplan-Meier plot of tumor progression, with a cutoff set at a tumor volume >750 mm.sup.3. The combination IgG1-hDR5-01-G56T-E430G+IgG1-hDR5-05-E430G inhibited tumor growth progression better than an equivalent dose IgG1-hDR5-01-G56T+IgG1-hDR5-05.
[0414] These data illustrate that introduction of the E430G hexamerization-enhancing mutation in the anti-DR5 antibody combination IgG1-DR5-01-K409R-E430G+IgG1-DR5-05-F405L-E430G resulted in enhanced tumor growth inhibition in an in vivo xenograft model with HCT15 human colon cancer cells.
Example 26: In Vivo Efficacy of the Anti-DR5 Antibodies IgG1-hDR5-01-G56T-E430G and IgG1-hDR5-05-E430G in a Subcutaneous COLO 205 Colon Cancer Xenograft Model
[0415] The in vivo anti-tumor efficacy of antibodies IgG1-hDR5-01-G56T-E430G and IgG1-hDR5-05-E430G was evaluated for the single antibodies and the combination of both antibodies and compared to the parental antibodies without the E430G mutation in the subcutaneous COLO 205 human colon cancer xenograft model. Tumor cell inoculation, mice handling, tumor outgrowth measurements and endpoint determination were performed, essentially as described in Example 20. 3.times.10.sup.6 cells were injected in a volume of 100 1.11 PBS into the flank of 5-8 weeks old female SCID mice (C.B-17/IcrHan*Hsd-Prkdc.sup.scid; Harlan). At day 9, the average tumor volume was measured and the mice were sorted into groups with equal tumor size variance. Mice were treated by intravenous (i.v.) injection of 10 .mu.g (0.5 mg/kg) antibody in 200 .mu.L PBS on day 9. Mice in the control group were treated with 10 .mu.g (0.5 mg/kg) IgG1-b12.
TABLE-US-00006 TABLE 2 Treatment groups and dosing Dosing day after # # Total antibody tumor mice analyzed Antibody dose inoculation 8 8 IgG1-hDR5-01-G56T- 0.5 mg/kg 9 E430G 8 8 IgG1-hDR5-05-E430G 0.5 mg/kg 9 8 8 IgG1-hDR5-01-G56T- 0.5 mg/kg 9 E430G IgG1-hDR5-05-E430G 8 8 IgG1-hDR5-01-G56T 0.5 mg/kg 9 8 8 IgG1-hDR5-05 0.5 mg/kg 9 8 8 IgG1-hDR5-01-G56T 0.5 mg/kg 9 IgG1-hDR5-05 8 8 IgG1-b12 0.5 mg/kg 9
[0416] FIG. 26A shows mean tumor volumes per treatment group in time. Introduction of the E430G mutation in the single antibodies IgG1-hDR5-01-G56T-E430G and IgG1-hDR5-05-E430G resulted in enhanced inhibition of tumor growth compared to the parental antibodies without the E430G mutation. Treatment with the antibody combinations induced complete tumor regression, both for IgG1-hDR5-01-G56T-E430G+IgG1-hDR5-05-E430G and for the combination of parental antibodies without the E430G mutation. At day 19 the average tumor size in all groups treated with DR5-antibodies was significantly smaller than in animals treated with the negative control antibody IgG1-b12 (Mann Whitney test (P<0.001))(data not shown). FIG. 26B shows a Kaplan-Meier plot of tumor progression, with a cutoff set at a tumor volume >500 mm.sup.3. Compared to mice treated with negative control antibody IgG1-b12, tumor outgrowth was significantly delayed in all groups treated with anti-DR5 antibodies (Mantel-Cox analysis at tumor size cut-off 500 mm.sup.3: p<0.0001). Mice treated with the single antibodies IgG1-hDR5-01-G56T and IgG1-hDR5-05 without the hexamerization-enhancing mutation E430G showed tumor outgrowth significantly earlier compared to the mice treated with the other tested anti-DR5 antibodies ((Mantel-Cox analysis at tumor size cut-off 500 mm.sup.3: p<0.0001).
Sequence CWU 1
1
501330PRTHomo Sapiens 1Ala Ser Thr Lys Gly Pro Ser Val Phe Pro Leu
Ala Pro Ser Ser Lys1 5 10 15Ser Thr Ser Gly Gly Thr Ala Ala Leu Gly
Cys Leu Val Lys Asp Tyr 20 25 30Phe Pro Glu Pro Val Thr Val Ser Trp
Asn Ser Gly Ala Leu Thr Ser 35 40 45Gly Val His Thr Phe Pro Ala Val
Leu Gln Ser Ser Gly Leu Tyr Ser 50 55 60Leu Ser Ser Val Val Thr Val
Pro Ser Ser Ser Leu Gly Thr Gln Thr65 70 75 80Tyr Ile Cys Asn Val
Asn His Lys Pro Ser Asn Thr Lys Val Asp Lys 85 90 95Arg Val Glu Pro
Lys Ser Cys Asp Lys Thr His Thr Cys Pro Pro Cys 100 105 110Pro Ala
Pro Glu Leu Leu Gly Gly Pro Ser Val Phe Leu Phe Pro Pro 115 120
125Lys Pro Lys Asp Thr Leu Met Ile Ser Arg Thr Pro Glu Val Thr Cys
130 135 140Val Val Val Asp Val Ser His Glu Asp Pro Glu Val Lys Phe
Asn Trp145 150 155 160Tyr Val Asp Gly Val Glu Val His Asn Ala Lys
Thr Lys Pro Arg Glu 165 170 175Glu Gln Tyr Asn Ser Thr Tyr Arg Val
Val Ser Val Leu Thr Val Leu 180 185 190His Gln Asp Trp Leu Asn Gly
Lys Glu Tyr Lys Cys Lys Val Ser Asn 195 200 205Lys Ala Leu Pro Ala
Pro Ile Glu Lys Thr Ile Ser Lys Ala Lys Gly 210 215 220Gln Pro Arg
Glu Pro Gln Val Tyr Thr Leu Pro Pro Ser Arg Glu Glu225 230 235
240Met Thr Lys Asn Gln Val Ser Leu Thr Cys Leu Val Lys Gly Phe Tyr
245 250 255Pro Ser Asp Ile Ala Val Glu Trp Glu Ser Asn Gly Gln Pro
Glu Asn 260 265 270Asn Tyr Lys Thr Thr Pro Pro Val Leu Asp Ser Asp
Gly Ser Phe Phe 275 280 285Leu Tyr Ser Lys Leu Thr Val Asp Lys Ser
Arg Trp Gln Gln Gly Asn 290 295 300Val Phe Ser Cys Ser Val Met His
Glu Ala Leu His Asn His Tyr Thr305 310 315 320Gln Lys Ser Leu Ser
Leu Ser Pro Gly Lys 325 3302330PRTHomo Sapiens 2Ala Ser Thr Lys Gly
Pro Ser Val Phe Pro Leu Ala Pro Ser Ser Lys1 5 10 15Ser Thr Ser Gly
Gly Thr Ala Ala Leu Gly Cys Leu Val Lys Asp Tyr 20 25 30Phe Pro Glu
Pro Val Thr Val Ser Trp Asn Ser Gly Ala Leu Thr Ser 35 40 45Gly Val
His Thr Phe Pro Ala Val Leu Gln Ser Ser Gly Leu Tyr Ser 50 55 60Leu
Ser Ser Val Val Thr Val Pro Ser Ser Ser Leu Gly Thr Gln Thr65 70 75
80Tyr Ile Cys Asn Val Asn His Lys Pro Ser Asn Thr Lys Val Asp Lys
85 90 95Lys Val Glu Pro Lys Ser Cys Asp Lys Thr His Thr Cys Pro Pro
Cys 100 105 110Pro Ala Pro Glu Leu Leu Gly Gly Pro Ser Val Phe Leu
Phe Pro Pro 115 120 125Lys Pro Lys Asp Thr Leu Met Ile Ser Arg Thr
Pro Glu Val Thr Cys 130 135 140Val Val Val Asp Val Ser His Glu Asp
Pro Glu Val Lys Phe Asn Trp145 150 155 160Tyr Val Asp Gly Val Glu
Val His Asn Ala Lys Thr Lys Pro Arg Glu 165 170 175Glu Gln Tyr Asn
Ser Thr Tyr Arg Val Val Ser Val Leu Thr Val Leu 180 185 190His Gln
Asp Trp Leu Asn Gly Lys Glu Tyr Lys Cys Lys Val Ser Asn 195 200
205Lys Ala Leu Pro Ala Pro Ile Glu Lys Thr Ile Ser Lys Ala Lys Gly
210 215 220Gln Pro Arg Glu Pro Gln Val Tyr Thr Leu Pro Pro Ser Arg
Glu Glu225 230 235 240Met Thr Lys Asn Gln Val Ser Leu Thr Cys Leu
Val Lys Gly Phe Tyr 245 250 255Pro Ser Asp Ile Ala Val Glu Trp Glu
Ser Asn Gly Gln Pro Glu Asn 260 265 270Asn Tyr Lys Thr Thr Pro Pro
Val Leu Asp Ser Asp Gly Ser Phe Phe 275 280 285Leu Tyr Ser Lys Leu
Thr Val Asp Lys Ser Arg Trp Gln Gln Gly Asn 290 295 300Val Phe Ser
Cys Ser Val Met His Glu Ala Leu His Asn His Tyr Thr305 310 315
320Gln Lys Ser Leu Ser Leu Ser Pro Gly Lys 325 3303330PRTHomo
Sapiens 3Ala Ser Thr Lys Gly Pro Ser Val Phe Pro Leu Ala Pro Ser
Ser Lys1 5 10 15Ser Thr Ser Gly Gly Thr Ala Ala Leu Gly Cys Leu Val
Lys Asp Tyr 20 25 30Phe Pro Glu Pro Val Thr Val Ser Trp Asn Ser Gly
Ala Leu Thr Ser 35 40 45Gly Val His Thr Phe Pro Ala Val Leu Gln Ser
Ser Gly Leu Tyr Ser 50 55 60Leu Ser Ser Val Val Thr Val Pro Ser Ser
Ser Leu Gly Thr Gln Thr65 70 75 80Tyr Ile Cys Asn Val Asn His Lys
Pro Ser Asn Thr Lys Val Asp Lys 85 90 95Pro Val Glu Pro Lys Ser Cys
Asp Lys Thr His Thr Cys Pro Pro Cys 100 105 110Pro Ala Pro Glu Leu
Leu Gly Gly Pro Ser Val Phe Leu Phe Pro Pro 115 120 125Lys Pro Lys
Asp Thr Leu Met Ile Ser Arg Thr Pro Glu Val Thr Cys 130 135 140Val
Val Val Asp Val Ser His Glu Asp Pro Glu Val Lys Phe Asn Trp145 150
155 160Tyr Val Asp Gly Val Glu Val His Asn Ala Lys Thr Lys Pro Arg
Glu 165 170 175Glu Gln Tyr Asn Ser Thr Tyr Arg Val Val Ser Val Leu
Thr Val Leu 180 185 190His Gln Asp Trp Leu Asn Gly Lys Glu Tyr Lys
Cys Lys Val Ser Asn 195 200 205Lys Ala Leu Pro Ala Pro Ile Glu Lys
Thr Ile Ser Lys Ala Lys Gly 210 215 220Gln Pro Arg Glu Pro Gln Val
Tyr Thr Leu Pro Pro Ser Arg Asp Glu225 230 235 240Leu Thr Lys Asn
Gln Val Ser Leu Thr Cys Leu Val Lys Gly Phe Tyr 245 250 255Pro Ser
Asp Ile Ala Val Glu Trp Glu Ser Asn Gly Gln Pro Glu Asn 260 265
270Asn Tyr Lys Thr Thr Pro Pro Val Leu Asp Ser Asp Gly Ser Phe Phe
275 280 285Leu Tyr Ser Lys Leu Thr Val Asp Lys Ser Arg Trp Gln Gln
Gly Asn 290 295 300Val Phe Ser Cys Ser Val Met His Glu Ala Leu His
Asn His Tyr Thr305 310 315 320Gln Lys Ser Leu Ser Leu Ser Pro Gly
Lys 325 3304330PRTHomo Sapiens 4Ala Ser Thr Lys Gly Pro Ser Val Phe
Pro Leu Ala Pro Ser Ser Lys1 5 10 15Ser Thr Ser Gly Gly Thr Ala Ala
Leu Gly Cys Leu Val Lys Asp Tyr 20 25 30Phe Pro Glu Pro Val Thr Val
Ser Trp Asn Ser Gly Ala Leu Thr Ser 35 40 45Gly Val His Thr Phe Pro
Ala Val Leu Gln Ser Ser Gly Leu Tyr Ser 50 55 60Leu Ser Ser Val Val
Thr Val Pro Ser Ser Ser Leu Gly Thr Gln Thr65 70 75 80Tyr Ile Cys
Asn Val Asn His Lys Pro Ser Asn Thr Lys Val Asp Lys 85 90 95Pro Val
Glu Pro Lys Ser Cys Asp Lys Thr His Thr Cys Pro Pro Cys 100 105
110Pro Ala Pro Glu Leu Leu Gly Gly Pro Ser Val Phe Leu Phe Pro Pro
115 120 125Lys Pro Lys Asp Thr Leu Met Ile Ser Arg Thr Pro Glu Val
Thr Cys 130 135 140Val Val Val Asp Val Ser His Glu Asp Pro Glu Val
Lys Phe Asn Trp145 150 155 160Tyr Val Asp Gly Val Glu Val His Asn
Ala Lys Thr Lys Pro Arg Glu 165 170 175Glu Gln Tyr Asn Ser Thr Tyr
Arg Val Val Ser Val Leu Thr Val Leu 180 185 190His Gln Asp Trp Leu
Asn Gly Lys Glu Tyr Lys Cys Lys Val Ser Asn 195 200 205Lys Ala Leu
Pro Ala Pro Ile Glu Lys Thr Ile Ser Lys Ala Lys Gly 210 215 220Gln
Pro Arg Glu Pro Gln Val Tyr Thr Leu Pro Pro Ser Arg Glu Glu225 230
235 240Met Thr Lys Asn Gln Val Ser Leu Thr Cys Leu Val Lys Gly Phe
Tyr 245 250 255Pro Ser Asp Ile Ala Val Glu Trp Glu Ser Asn Gly Gln
Pro Glu Asn 260 265 270Asn Tyr Lys Thr Thr Pro Pro Val Leu Asp Ser
Asp Gly Ser Phe Phe 275 280 285Leu Tyr Ser Lys Leu Thr Val Asp Lys
Ser Arg Trp Gln Gln Gly Asn 290 295 300Val Phe Ser Cys Ser Val Met
His Glu Gly Leu His Asn His Tyr Thr305 310 315 320Gln Lys Ser Leu
Ser Leu Ser Pro Gly Lys 325 3305330PRTArtificial SequenceSynthetic
5Ala Ser Thr Lys Gly Pro Ser Val Phe Pro Leu Ala Pro Ser Ser Lys1 5
10 15Ser Thr Ser Gly Gly Thr Ala Ala Leu Gly Cys Leu Val Lys Asp
Tyr 20 25 30Phe Pro Glu Pro Val Thr Val Ser Trp Asn Ser Gly Ala Leu
Thr Ser 35 40 45Gly Val His Thr Phe Pro Ala Val Leu Gln Ser Ser Gly
Leu Tyr Ser 50 55 60Leu Ser Ser Val Val Thr Val Pro Ser Ser Ser Leu
Gly Thr Gln Thr65 70 75 80Tyr Ile Cys Asn Val Asn His Lys Pro Ser
Asn Thr Lys Val Asp Lys 85 90 95Arg Val Glu Pro Lys Ser Cys Asp Lys
Thr His Thr Cys Pro Pro Cys 100 105 110Pro Ala Pro Glu Leu Leu Gly
Gly Pro Ser Val Phe Leu Phe Pro Pro 115 120 125Lys Pro Lys Asp Thr
Leu Met Ile Ser Arg Thr Pro Glu Val Thr Cys 130 135 140Val Val Val
Asp Val Ser His Glu Asp Pro Glu Val Lys Phe Asn Trp145 150 155
160Tyr Val Asp Gly Val Glu Val His Asn Ala Lys Thr Lys Pro Arg Glu
165 170 175Glu Gln Tyr Asn Ser Thr Tyr Arg Val Val Ser Val Leu Thr
Val Leu 180 185 190His Gln Asp Trp Leu Asn Gly Lys Glu Tyr Lys Cys
Lys Val Ser Asn 195 200 205Lys Ala Leu Pro Ala Pro Ile Glu Lys Thr
Ile Ser Lys Ala Lys Gly 210 215 220Gln Pro Arg Glu Pro Gln Val Tyr
Thr Leu Pro Pro Ser Arg Glu Glu225 230 235 240Met Thr Lys Asn Gln
Val Ser Leu Thr Cys Leu Val Lys Gly Phe Tyr 245 250 255Pro Ser Asp
Ile Ala Val Glu Trp Glu Ser Asn Gly Gln Pro Glu Asn 260 265 270Asn
Tyr Lys Thr Thr Pro Pro Val Leu Asp Ser Asp Gly Ser Phe Phe 275 280
285Leu Tyr Ser Lys Leu Thr Val Asp Lys Ser Arg Trp Gln Gln Gly Asn
290 295 300Val Phe Ser Cys Ser Val Met His Gly Ala Leu His Asn His
Tyr Thr305 310 315 320Gln Lys Ser Leu Ser Leu Ser Pro Gly Lys 325
3306330PRTArtificial SequenceSynthetic 6Ala Ser Thr Lys Gly Pro Ser
Val Phe Pro Leu Ala Pro Ser Ser Lys1 5 10 15Ser Thr Ser Gly Gly Thr
Ala Ala Leu Gly Cys Leu Val Lys Asp Tyr 20 25 30Phe Pro Glu Pro Val
Thr Val Ser Trp Asn Ser Gly Ala Leu Thr Ser 35 40 45Gly Val His Thr
Phe Pro Ala Val Leu Gln Ser Ser Gly Leu Tyr Ser 50 55 60Leu Ser Ser
Val Val Thr Val Pro Ser Ser Ser Leu Gly Thr Gln Thr65 70 75 80Tyr
Ile Cys Asn Val Asn His Lys Pro Ser Asn Thr Lys Val Asp Lys 85 90
95Arg Val Glu Pro Lys Ser Cys Asp Lys Thr His Thr Cys Pro Pro Cys
100 105 110Pro Ala Pro Glu Leu Leu Gly Gly Pro Ser Val Phe Leu Phe
Pro Pro 115 120 125Lys Pro Lys Asp Thr Leu Met Ile Ser Arg Thr Pro
Glu Val Thr Cys 130 135 140Val Val Val Asp Val Ser His Glu Asp Pro
Glu Val Lys Phe Asn Trp145 150 155 160Tyr Val Asp Gly Val Glu Val
His Asn Ala Lys Thr Lys Pro Arg Glu 165 170 175Glu Gln Tyr Asn Ser
Thr Tyr Arg Val Val Ser Val Leu Thr Val Leu 180 185 190His Gln Asp
Trp Leu Asn Gly Lys Glu Tyr Lys Cys Lys Val Ser Asn 195 200 205Lys
Ala Leu Pro Ala Pro Ile Glu Lys Thr Ile Ser Lys Ala Lys Gly 210 215
220Gln Pro Arg Lys Pro Gln Val Tyr Thr Leu Pro Pro Ser Arg Glu
Glu225 230 235 240Met Thr Lys Asn Gln Val Ser Leu Thr Cys Leu Val
Lys Gly Phe Tyr 245 250 255Pro Ser Asp Ile Ala Val Glu Trp Glu Ser
Asn Gly Gln Pro Glu Asn 260 265 270Asn Tyr Lys Thr Thr Pro Pro Val
Leu Asp Ser Asp Gly Ser Phe Phe 275 280 285Leu Tyr Ser Lys Leu Thr
Val Asp Lys Ser Arg Trp Gln Gln Gly Asn 290 295 300Val Phe Ser Cys
Ser Val Met His Glu Ala Leu His Asn His Tyr Thr305 310 315 320Gln
Lys Ser Leu Ser Leu Ser Pro Gly Lys 325 3307330PRTArtificial
SequenceSynthetic 7Ala Ser Thr Lys Gly Pro Ser Val Phe Pro Leu Ala
Pro Ser Ser Lys1 5 10 15Ser Thr Ser Gly Gly Thr Ala Ala Leu Gly Cys
Leu Val Lys Asp Tyr 20 25 30Phe Pro Glu Pro Val Thr Val Ser Trp Asn
Ser Gly Ala Leu Thr Ser 35 40 45Gly Val His Thr Phe Pro Ala Val Leu
Gln Ser Ser Gly Leu Tyr Ser 50 55 60Leu Ser Ser Val Val Thr Val Pro
Ser Ser Ser Leu Gly Thr Gln Thr65 70 75 80Tyr Ile Cys Asn Val Asn
His Lys Pro Ser Asn Thr Lys Val Asp Lys 85 90 95Arg Val Glu Pro Lys
Ser Cys Asp Lys Thr His Thr Cys Pro Pro Cys 100 105 110Pro Ala Pro
Glu Leu Leu Gly Gly Pro Ser Val Phe Leu Phe Pro Pro 115 120 125Lys
Pro Lys Asp Thr Leu Met Ile Ser Arg Thr Pro Glu Val Thr Cys 130 135
140Val Val Val Asp Val Ser His Glu Asp Pro Glu Val Lys Phe Asn
Trp145 150 155 160Tyr Val Asp Gly Val Glu Val His Asn Ala Lys Thr
Lys Pro Arg Glu 165 170 175Glu Gln Tyr Asn Ser Thr Tyr Arg Val Val
Ser Val Leu Thr Val Leu 180 185 190His Gln Asp Trp Leu Asn Gly Lys
Glu Tyr Lys Cys Lys Val Ser Asn 195 200 205Lys Ala Leu Pro Ala Pro
Ile Glu Lys Thr Ile Ser Lys Ala Lys Gly 210 215 220Gln Pro Arg Glu
Pro Gln Val Tyr Thr Leu Pro Pro Ser Arg Glu Glu225 230 235 240Met
Thr Lys Asn Gln Val Ser Leu Thr Cys Leu Val Lys Gly Phe Tyr 245 250
255Pro Ser Asp Ile Ala Val Glu Trp Glu Ser Asn Gly Gln Pro Glu Asn
260 265 270Asn Tyr Lys Thr Thr Pro Pro Val Leu Asp Ser Asp Gly Ser
Phe Phe 275 280 285Leu Tyr Ser Lys Leu Thr Val Asp Lys Ser Arg Trp
Gln Gln Gly Asn 290 295 300Val Phe Ser Cys Ser Val Met His Glu Ala
Leu His Asn His Tyr Thr305 310 315 320Gln Lys Tyr Leu Ser Leu Ser
Pro Gly Lys 325 3308330PRTArtificial SequenceSynthetic 8Ala Ser Thr
Lys Gly Pro Ser Val Phe Pro Leu Ala Pro Ser Ser Lys1 5 10 15Ser Thr
Ser Gly Gly Thr Ala Ala Leu Gly Cys Leu Val Lys Asp Tyr 20 25 30Phe
Pro Glu Pro Val Thr Val Ser Trp Asn Ser Gly Ala Leu Thr Ser 35 40
45Gly Val His Thr Phe Pro Ala Val Leu Gln Ser Ser Gly Leu Tyr Ser
50 55 60Leu Ser Ser Val Val Thr Val Pro Ser Ser Ser Leu Gly Thr Gln
Thr65 70 75 80Tyr Ile Cys Asn Val Asn His Lys Pro Ser Asn Thr Lys
Val Asp Lys 85 90 95Arg Val Glu Pro Lys Ser Cys Asp Lys Thr His Thr
Cys Pro Pro Cys 100 105 110Pro Ala Pro Glu Leu Leu Gly Gly Pro Ser
Val Phe Leu Phe Pro Pro 115 120 125Lys Pro Lys Asp Thr Leu Met Ile
Ser Arg
Thr Pro Glu Val Thr Cys 130 135 140Val Val Val Asp Val Ser His Glu
Asp Pro Glu Val Lys Phe Asn Trp145 150 155 160Tyr Val Asp Gly Val
Glu Val His Asn Ala Lys Thr Lys Pro Arg Glu 165 170 175Glu Gln Tyr
Asn Ser Thr Tyr Arg Val Val Ser Val Leu Thr Val Leu 180 185 190His
Gln Asp Trp Leu Asn Gly Lys Glu Tyr Lys Cys Lys Val Ser Asn 195 200
205Lys Ala Leu Pro Ala Pro Ile Glu Lys Thr Ile Ser Lys Ala Lys Gly
210 215 220Gln Pro Arg Glu Pro Gln Val Tyr Thr Leu Pro Pro Ser Arg
Glu Glu225 230 235 240Met Thr Lys Asn Gln Val Ser Leu Thr Cys Leu
Val Lys Gly Phe Tyr 245 250 255Pro Ser Asp Ile Ala Val Glu Trp Glu
Ser Asn Gly Gln Pro Glu Asn 260 265 270Asn Tyr Lys Thr Thr Pro Pro
Val Leu Asp Ser Asp Gly Ser Phe Leu 275 280 285Leu Tyr Ser Lys Leu
Thr Val Asp Lys Ser Arg Trp Gln Gln Gly Asn 290 295 300Val Phe Ser
Cys Ser Val Met His Glu Ala Leu His Asn His Tyr Thr305 310 315
320Gln Lys Ser Leu Ser Leu Ser Pro Gly Lys 325 3309330PRTArtificial
SequenceSynthetic 9Ala Ser Thr Lys Gly Pro Ser Val Phe Pro Leu Ala
Pro Ser Ser Lys1 5 10 15Ser Thr Ser Gly Gly Thr Ala Ala Leu Gly Cys
Leu Val Lys Asp Tyr 20 25 30Phe Pro Glu Pro Val Thr Val Ser Trp Asn
Ser Gly Ala Leu Thr Ser 35 40 45Gly Val His Thr Phe Pro Ala Val Leu
Gln Ser Ser Gly Leu Tyr Ser 50 55 60Leu Ser Ser Val Val Thr Val Pro
Ser Ser Ser Leu Gly Thr Gln Thr65 70 75 80Tyr Ile Cys Asn Val Asn
His Lys Pro Ser Asn Thr Lys Val Asp Lys 85 90 95Arg Val Glu Pro Lys
Ser Cys Asp Lys Thr His Thr Cys Pro Pro Cys 100 105 110Pro Ala Pro
Glu Leu Leu Gly Gly Pro Ser Val Phe Leu Phe Pro Pro 115 120 125Lys
Pro Lys Asp Thr Leu Met Ile Ser Arg Thr Pro Glu Val Thr Cys 130 135
140Val Val Val Asp Val Ser His Glu Asp Pro Glu Val Lys Phe Asn
Trp145 150 155 160Tyr Val Asp Gly Val Glu Val His Asn Ala Lys Thr
Lys Pro Arg Glu 165 170 175Glu Gln Tyr Asn Ser Thr Tyr Arg Val Val
Ser Val Leu Thr Val Leu 180 185 190His Gln Asp Trp Leu Asn Gly Lys
Glu Tyr Lys Cys Lys Val Ser Asn 195 200 205Lys Ala Leu Pro Ala Pro
Ile Glu Lys Thr Ile Ser Lys Ala Lys Gly 210 215 220Gln Pro Arg Glu
Pro Gln Val Tyr Thr Leu Pro Pro Ser Arg Glu Glu225 230 235 240Met
Thr Lys Asn Gln Val Ser Leu Thr Cys Leu Val Lys Gly Phe Tyr 245 250
255Pro Ser Asp Ile Ala Val Glu Trp Glu Ser Asn Gly Gln Pro Glu Asn
260 265 270Asn Tyr Lys Thr Thr Pro Pro Val Leu Asp Ser Asp Gly Ser
Phe Phe 275 280 285Leu Tyr Ser Arg Leu Thr Val Asp Lys Ser Arg Trp
Gln Gln Gly Asn 290 295 300Val Phe Ser Cys Ser Val Met His Glu Ala
Leu His Asn His Tyr Thr305 310 315 320Gln Lys Ser Leu Ser Leu Ser
Pro Gly Lys 325 33010330PRTArtificial SequenceSynthetic 10Ala Ser
Thr Lys Gly Pro Ser Val Phe Pro Leu Ala Pro Ser Ser Lys1 5 10 15Ser
Thr Ser Gly Gly Thr Ala Ala Leu Gly Cys Leu Val Lys Asp Tyr 20 25
30Phe Pro Glu Pro Val Thr Val Ser Trp Asn Ser Gly Ala Leu Thr Ser
35 40 45Gly Val His Thr Phe Pro Ala Val Leu Gln Ser Ser Gly Leu Tyr
Ser 50 55 60Leu Ser Ser Val Val Thr Val Pro Ser Ser Ser Leu Gly Thr
Gln Thr65 70 75 80Tyr Ile Cys Asn Val Asn His Lys Pro Ser Asn Thr
Lys Val Asp Lys 85 90 95Arg Val Glu Pro Lys Ser Cys Asp Lys Thr His
Thr Cys Pro Pro Cys 100 105 110Pro Ala Pro Glu Leu Leu Gly Gly Pro
Ser Val Phe Leu Phe Pro Pro 115 120 125Lys Pro Lys Asp Thr Leu Met
Ile Ser Arg Thr Pro Glu Val Thr Cys 130 135 140Val Val Val Asp Val
Ser His Glu Asp Pro Glu Val Lys Phe Asn Trp145 150 155 160Tyr Val
Asp Gly Val Glu Val His Asn Ala Lys Thr Lys Pro Arg Glu 165 170
175Glu Gln Tyr Asn Ser Thr Tyr Arg Val Val Ser Val Leu Thr Val Leu
180 185 190His Gln Asp Trp Leu Asn Gly Lys Glu Tyr Lys Cys Lys Val
Ser Asn 195 200 205Lys Ala Leu Pro Ala Pro Ile Glu Lys Thr Ile Ser
Lys Ala Lys Gly 210 215 220Gln Pro Arg Glu Pro Gln Val Tyr Thr Leu
Pro Pro Ser Arg Glu Glu225 230 235 240Met Thr Lys Asn Gln Val Ser
Leu Thr Cys Leu Val Lys Gly Phe Tyr 245 250 255Pro Ser Asp Ile Ala
Val Glu Trp Glu Ser Asn Gly Gln Pro Glu Asn 260 265 270Asn Tyr Lys
Thr Thr Pro Pro Val Leu Asp Ser Asp Gly Ser Phe Phe 275 280 285Leu
Tyr Ser Lys Leu Thr Val Asp Lys Ser Arg Trp Gln Gln Gly Asn 290 295
300Val Phe Ser Cys Ser Val Met His Glu Ala Leu His Asn His Tyr
Thr305 310 315 320Gln Glu Ser Leu Ser Leu Ser Pro Gly Lys 325
33011330PRTArtificial SequenceSynthetic 11Ala Ser Thr Lys Gly Pro
Ser Val Phe Pro Leu Ala Pro Ser Ser Lys1 5 10 15Ser Thr Ser Gly Gly
Thr Ala Ala Leu Gly Cys Leu Val Lys Asp Tyr 20 25 30Phe Pro Glu Pro
Val Thr Val Ser Trp Asn Ser Gly Ala Leu Thr Ser 35 40 45Gly Val His
Thr Phe Pro Ala Val Leu Gln Ser Ser Gly Leu Tyr Ser 50 55 60Leu Ser
Ser Val Val Thr Val Pro Ser Ser Ser Leu Gly Thr Gln Thr65 70 75
80Tyr Ile Cys Asn Val Asn His Lys Pro Ser Asn Thr Lys Val Asp Lys
85 90 95Arg Val Glu Pro Lys Ser Cys Asp Lys Thr His Thr Cys Pro Pro
Cys 100 105 110Pro Ala Pro Glu Leu Leu Gly Gly Pro Ser Val Phe Leu
Phe Pro Pro 115 120 125Lys Pro Lys Asp Thr Leu Met Ile Ser Arg Thr
Pro Glu Val Thr Cys 130 135 140Val Val Val Asp Val Ser His Glu Asp
Pro Glu Val Lys Phe Asn Trp145 150 155 160Tyr Val Asp Gly Val Glu
Val His Asn Ala Lys Thr Lys Pro Arg Glu 165 170 175Glu Gln Tyr Asn
Ser Thr Tyr Arg Val Val Ser Val Leu Thr Val Leu 180 185 190His Gln
Asp Trp Leu Asn Gly Lys Glu Tyr Lys Cys Lys Val Ser Asn 195 200
205Lys Ala Leu Pro Ala Pro Ile Glu Lys Thr Ile Ser Lys Ala Lys Gly
210 215 220Gln Pro Arg Glu Pro Gln Val Tyr Thr Leu Pro Pro Ser Arg
Glu Glu225 230 235 240Met Thr Lys Asn Gln Val Ser Leu Thr Cys Leu
Val Lys Gly Phe Tyr 245 250 255Pro Ser Asp Ile Ala Val Glu Trp Glu
Ser Asn Gly Gln Pro Glu Asn 260 265 270Asn Tyr Lys Thr Thr Pro Pro
Val Leu Asp Ser Asp Gly Ser Phe Phe 275 280 285Leu Tyr Ser Lys Leu
Thr Val Asp Lys Ser Arg Trp Gln Gln Gly Asn 290 295 300Val Phe Ser
Cys Ser Val Met His Glu Ala Leu His Asn His Tyr Thr305 310 315
320Gln Lys Lys Leu Ser Leu Ser Pro Gly Lys 325
33012330PRTArtificial SequenceSynthetic 12Ala Ser Thr Lys Gly Pro
Ser Val Phe Pro Leu Ala Pro Ser Ser Lys1 5 10 15Ser Thr Ser Gly Gly
Thr Ala Ala Leu Gly Cys Leu Val Lys Asp Tyr 20 25 30Phe Pro Glu Pro
Val Thr Val Ser Trp Asn Ser Gly Ala Leu Thr Ser 35 40 45Gly Val His
Thr Phe Pro Ala Val Leu Gln Ser Ser Gly Leu Tyr Ser 50 55 60Leu Ser
Ser Val Val Thr Val Pro Ser Ser Ser Leu Gly Thr Gln Thr65 70 75
80Tyr Ile Cys Asn Val Asn His Lys Pro Ser Asn Thr Lys Val Asp Lys
85 90 95Arg Val Glu Pro Lys Ser Cys Asp Lys Thr His Thr Cys Pro Pro
Cys 100 105 110Pro Ala Pro Glu Leu Leu Gly Gly Pro Ser Val Phe Leu
Phe Pro Pro 115 120 125Lys Pro Lys Asp Thr Leu Met Ile Ser Arg Thr
Pro Glu Val Thr Cys 130 135 140Val Val Val Asp Val Ser His Glu Asp
Pro Glu Val Lys Phe Asn Trp145 150 155 160Tyr Val Asp Gly Val Glu
Val His Asn Ala Lys Thr Lys Pro Arg Glu 165 170 175Glu Gln Tyr Asn
Ser Thr Tyr Arg Val Val Ser Val Leu Thr Val Leu 180 185 190His Gln
Asp Trp Leu Asn Gly Lys Glu Tyr Lys Cys Lys Val Ser Asn 195 200
205Lys Ala Leu Pro Ala Pro Ile Glu Lys Thr Ile Ser Lys Ala Lys Gly
210 215 220Gln Pro Arg Glu Pro Gln Val Tyr Thr Leu Pro Pro Ser Arg
Glu Glu225 230 235 240Met Thr Lys Asn Gln Val Ser Leu Thr Cys Leu
Val Lys Gly Phe Tyr 245 250 255Pro Ser Asp Ile Ala Val Glu Trp Glu
Ser Asn Gly Gln Pro Glu Asn 260 265 270Asn Tyr Lys Thr Thr Pro Pro
Val Leu Asp Ser Asp Gly Ser Phe Phe 275 280 285Leu Tyr Ser Lys Leu
Thr Val Asp Lys Ser Arg Trp Gln Gln Gly Asn 290 295 300Val Phe Ser
Cys Ser Val Met His Glu Ala Leu His Asn His Ile Thr305 310 315
320Gln Lys Ser Leu Ser Leu Ser Pro Gly Lys 325
33013118PRTArtificial SequenceSynthetic 13Glu Val Gln Leu Val Gln
Ser Gly Ala Glu Val Lys Met Pro Gly Ala1 5 10 15Ser Val Lys Leu Ser
Cys Arg Val Ser Gly Asp Thr Phe Thr Ala Tyr 20 25 30Phe Ile His Trp
Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Met 35 40 45Gly Trp Phe
Asn Pro Ile Ser Gly Thr Ala Gly Ser Ala Glu Lys Phe 50 55 60Arg Gly
Arg Val Ala Met Thr Arg Asp Thr Ser Ile Ser Thr Ala Tyr65 70 75
80Met Glu Leu Asn Arg Leu Thr Phe Asp Asp Thr Ala Val Tyr Tyr Cys
85 90 95Ala Arg Gln His Arg Gly Asn Thr Phe Asp Pro Trp Gly Gln Gly
Thr 100 105 110Leu Val Thr Val Ser Ser 11514111PRTArtificial
SequenceSynthetic 14Gln Ser Ala Leu Thr Gln Pro Ala Ser Val Ser Gly
Ser Pro Gly Gln1 5 10 15Ser Ile Thr Ile Ser Cys Thr Gly Thr Ser Ser
Asp Ile Gly Ala Tyr 20 25 30Lys Tyr Val Ser Trp Tyr Gln Gln His Pro
Gly Lys Ala Pro Lys Leu 35 40 45Val Ile Tyr Glu Val Ser Asn Arg Pro
Ser Gly Val Ser Ser Arg Phe 50 55 60Ser Gly Ser Lys Ser Gly Gln Thr
Ala Ser Leu Thr Ile Ser Gly Leu65 70 75 80Gln Ala Asp Asp Glu Ala
Asp Tyr Tyr Cys Asn Ser Tyr Gln Gly Tyr 85 90 95Asn Thr Trp Val Phe
Gly Gly Gly Thr Lys Val Thr Val Leu Gly 100 105
11015132PRTArtificial SequenceSynthetic 15Gln Leu Gln Leu Gln Glu
Ser Gly Pro Gly Leu Val Lys Pro Ser Glu1 5 10 15Thr Leu Ser Leu Thr
Cys Thr Val Ser Gly Ala Ser Ile Ser Ala Asn 20 25 30Ser Tyr Tyr Gly
Val Trp Val Arg Gln Ser Pro Gly Lys Gly Leu Glu 35 40 45Trp Val Gly
Ser Ile Ala Tyr Arg Gly Asn Ser Asn Ser Gly Ser Thr 50 55 60Tyr Tyr
Asn Pro Ser Leu Lys Ser Arg Ala Thr Val Ser Val Asp Thr65 70 75
80Ser Lys Asn Gln Val Ser Leu Arg Leu Thr Ser Val Thr Ala Ala Asp
85 90 95Thr Ala Leu Tyr Tyr Cys Ala Arg Arg Gln Leu Leu Asp Asp Gly
Thr 100 105 110Gly Tyr Gln Trp Ala Ala Phe Asp Val Trp Gly Gln Gly
Thr Met Val 115 120 125Thr Val Ser Ser 13016110PRTArtificial
SequenceSynthetic 16Gln Ser Val Leu Thr Gln Pro Pro Ser Val Ser Glu
Ala Pro Arg Gln1 5 10 15Thr Val Thr Ile Ser Cys Ser Gly Asn Ser Phe
Asn Ile Gly Arg Tyr 20 25 30Pro Val Asn Trp Tyr Gln Gln Leu Pro Gly
Lys Ala Pro Lys Leu Leu 35 40 45Ile Tyr Tyr Asn Asn Leu Arg Phe Ser
Gly Val Ser Asp Arg Phe Ser 50 55 60Gly Ser Lys Ser Gly Thr Ser Ala
Ser Leu Ala Ile Arg Asp Leu Leu65 70 75 80Ser Glu Asp Glu Ala Asp
Tyr Tyr Cys Ser Thr Trp Asp Asp Thr Leu 85 90 95Lys Gly Trp Val Phe
Gly Gly Gly Thr Lys Val Thr Val Leu 100 105 110178PRTArtificial
SequenceSynthetic 17Gly Phe Asn Ile Lys Asp Thr Phe1
5188PRTArtificial SequenceSynthetic 18Ile Asp Pro Ala Asn Gly Asn
Thr1 51911PRTArtificial SequenceSynthetic 19Val Arg Gly Leu Tyr Thr
Tyr Tyr Phe Asp Tyr1 5 1020118PRTArtificial SequenceSynthetic 20Glu
Val Gln Leu Gln Gln Ser Gly Ala Glu Val Val Lys Pro Gly Ala1 5 10
15Ser Val Lys Leu Ser Cys Lys Ala Ser Gly Phe Asn Ile Lys Asp Thr
20 25 30Phe Ile His Trp Val Lys Gln Ala Pro Gly Gln Gly Leu Glu Trp
Ile 35 40 45Gly Arg Ile Asp Pro Ala Asn Gly Asn Thr Lys Tyr Asp Pro
Lys Phe 50 55 60Gln Gly Lys Ala Thr Ile Thr Thr Asp Thr Ser Ser Asn
Thr Ala Tyr65 70 75 80Met Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr
Ala Val Tyr Tyr Cys 85 90 95Val Arg Gly Leu Tyr Thr Tyr Tyr Phe Asp
Tyr Trp Gly Gln Gly Thr 100 105 110Leu Val Thr Val Ser Ser
11521448PRTArtificial SequenceSynthetic 21Glu Val Gln Leu Gln Gln
Ser Gly Ala Glu Val Val Lys Pro Gly Ala1 5 10 15Ser Val Lys Leu Ser
Cys Lys Ala Ser Gly Phe Asn Ile Lys Asp Thr 20 25 30Phe Ile His Trp
Val Lys Gln Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40 45Gly Arg Ile
Asp Pro Ala Asn Gly Asn Thr Lys Tyr Asp Pro Lys Phe 50 55 60Gln Gly
Lys Ala Thr Ile Thr Thr Asp Thr Ser Ser Asn Thr Ala Tyr65 70 75
80Met Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val Tyr Tyr Cys
85 90 95Val Arg Gly Leu Tyr Thr Tyr Tyr Phe Asp Tyr Trp Gly Gln Gly
Thr 100 105 110Leu Val Thr Val Ser Ser Ala Ser Thr Lys Gly Pro Ser
Val Phe Pro 115 120 125Leu Ala Pro Ser Ser Lys Ser Thr Ser Gly Gly
Thr Ala Ala Leu Gly 130 135 140Cys Leu Val Lys Asp Tyr Phe Pro Glu
Pro Val Thr Val Ser Trp Asn145 150 155 160Ser Gly Ala Leu Thr Ser
Gly Val His Thr Phe Pro Ala Val Leu Gln 165 170 175Ser Ser Gly Leu
Tyr Ser Leu Ser Ser Val Val Thr Val Pro Ser Ser 180 185 190Ser Leu
Gly Thr Gln Thr Tyr Ile Cys Asn Val Asn His Lys Pro Ser 195 200
205Asn Thr Lys Val Asp Lys Arg Val Glu Pro Lys Ser Cys Asp Lys Thr
210 215 220His Thr Cys Pro Pro Cys Pro Ala Pro Glu Leu Leu Gly Gly
Pro Ser225 230 235 240Val Phe Leu Phe Pro Pro Lys Pro Lys Asp Thr
Leu Met Ile Ser Arg 245 250 255Thr Pro Glu Val Thr Cys Val Val Val
Asp Val Ser His Glu Asp Pro 260 265 270Glu Val Lys Phe Asn Trp Tyr
Val Asp Gly Val
Glu Val His Asn Ala 275 280 285Lys Thr Lys Pro Arg Glu Glu Gln Tyr
Asn Ser Thr Tyr Arg Val Val 290 295 300Ser Val Leu Thr Val Leu His
Gln Asp Trp Leu Asn Gly Lys Glu Tyr305 310 315 320Lys Cys Lys Val
Ser Asn Lys Ala Leu Pro Ala Pro Ile Glu Lys Thr 325 330 335Ile Ser
Lys Ala Lys Gly Gln Pro Arg Glu Pro Gln Val Tyr Thr Leu 340 345
350Pro Pro Ser Arg Glu Glu Met Thr Lys Asn Gln Val Ser Leu Thr Cys
355 360 365Leu Val Lys Gly Phe Tyr Pro Ser Asp Ile Ala Val Glu Trp
Glu Ser 370 375 380Asn Gly Gln Pro Glu Asn Asn Tyr Lys Thr Thr Pro
Pro Val Leu Asp385 390 395 400Ser Asp Gly Ser Phe Phe Leu Tyr Ser
Lys Leu Thr Val Asp Lys Ser 405 410 415Arg Trp Gln Gln Gly Asn Val
Phe Ser Cys Ser Val Met His Glu Ala 420 425 430Leu His Asn His Tyr
Thr Gln Lys Ser Leu Ser Leu Ser Pro Gly Lys 435 440
445226PRTArtificial SequenceSynthetic 22Gln Ser Ile Ser Asn Asn1
5239PRTArtificial SequenceSynthetic 23Gln Gln Gly Asn Ser Trp Pro
Tyr Thr1 524107PRTArtificial SequenceSynthetic 24Glu Ile Val Met
Thr Gln Ser Pro Ala Thr Leu Ser Val Ser Pro Gly1 5 10 15Glu Arg Ala
Thr Leu Ser Cys Arg Ala Ser Gln Ser Ile Ser Asn Asn 20 25 30Leu His
Trp Tyr Gln Gln Lys Pro Gly Gln Ala Pro Arg Leu Leu Ile 35 40 45Lys
Phe Ala Ser Gln Ser Ile Thr Gly Ile Pro Ala Arg Phe Ser Gly 50 55
60Ser Gly Ser Gly Thr Glu Phe Thr Leu Thr Ile Ser Ser Leu Gln Ser65
70 75 80Glu Asp Phe Ala Val Tyr Tyr Cys Gln Gln Gly Asn Ser Trp Pro
Tyr 85 90 95Thr Phe Gly Gln Gly Thr Lys Leu Glu Ile Lys 100
10525214PRTArtificial SequenceSynthetic 25Glu Ile Val Met Thr Gln
Ser Pro Ala Thr Leu Ser Val Ser Pro Gly1 5 10 15Glu Arg Ala Thr Leu
Ser Cys Arg Ala Ser Gln Ser Ile Ser Asn Asn 20 25 30Leu His Trp Tyr
Gln Gln Lys Pro Gly Gln Ala Pro Arg Leu Leu Ile 35 40 45Lys Phe Ala
Ser Gln Ser Ile Thr Gly Ile Pro Ala Arg Phe Ser Gly 50 55 60Ser Gly
Ser Gly Thr Glu Phe Thr Leu Thr Ile Ser Ser Leu Gln Ser65 70 75
80Glu Asp Phe Ala Val Tyr Tyr Cys Gln Gln Gly Asn Ser Trp Pro Tyr
85 90 95Thr Phe Gly Gln Gly Thr Lys Leu Glu Ile Lys Arg Thr Val Ala
Ala 100 105 110Pro Ser Val Phe Ile Phe Pro Pro Ser Asp Glu Gln Leu
Lys Ser Gly 115 120 125Thr Ala Ser Val Val Cys Leu Leu Asn Asn Phe
Tyr Pro Arg Glu Ala 130 135 140Lys Val Gln Trp Lys Val Asp Asn Ala
Leu Gln Ser Gly Asn Ser Gln145 150 155 160Glu Ser Val Thr Glu Gln
Asp Ser Lys Asp Ser Thr Tyr Ser Leu Ser 165 170 175Ser Thr Leu Thr
Leu Ser Lys Ala Asp Tyr Glu Lys His Lys Val Tyr 180 185 190Ala Cys
Glu Val Thr His Gln Gly Leu Ser Ser Pro Val Thr Lys Ser 195 200
205Phe Asn Arg Gly Glu Cys 210268PRTArtificial SequenceSynthetic
26Ile Asp Pro Ala Asn Thr Asn Thr1 527118PRTArtificial
SequenceSynthetic 27Glu Val Gln Leu Gln Gln Ser Gly Ala Glu Val Val
Lys Pro Gly Ala1 5 10 15Ser Val Lys Leu Ser Cys Lys Ala Ser Gly Phe
Asn Ile Lys Asp Thr 20 25 30Phe Ile His Trp Val Lys Gln Ala Pro Gly
Gln Gly Leu Glu Trp Ile 35 40 45Gly Arg Ile Asp Pro Ala Asn Thr Asn
Thr Lys Tyr Asp Pro Lys Phe 50 55 60Gln Gly Lys Ala Thr Ile Thr Thr
Asp Thr Ser Ser Asn Thr Ala Tyr65 70 75 80Met Glu Leu Ser Ser Leu
Arg Ser Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Val Arg Gly Leu Tyr
Thr Tyr Tyr Phe Asp Tyr Trp Gly Gln Gly Thr 100 105 110Leu Val Thr
Val Ser Ser 11528448PRTArtificial SequenceSynthetic 28Glu Val Gln
Leu Gln Gln Ser Gly Ala Glu Val Val Lys Pro Gly Ala1 5 10 15Ser Val
Lys Leu Ser Cys Lys Ala Ser Gly Phe Asn Ile Lys Asp Thr 20 25 30Phe
Ile His Trp Val Lys Gln Ala Pro Gly Gln Gly Leu Glu Trp Ile 35 40
45Gly Arg Ile Asp Pro Ala Asn Thr Asn Thr Lys Tyr Asp Pro Lys Phe
50 55 60Gln Gly Lys Ala Thr Ile Thr Thr Asp Thr Ser Ser Asn Thr Ala
Tyr65 70 75 80Met Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val
Tyr Tyr Cys 85 90 95Val Arg Gly Leu Tyr Thr Tyr Tyr Phe Asp Tyr Trp
Gly Gln Gly Thr 100 105 110Leu Val Thr Val Ser Ser Ala Ser Thr Lys
Gly Pro Ser Val Phe Pro 115 120 125Leu Ala Pro Ser Ser Lys Ser Thr
Ser Gly Gly Thr Ala Ala Leu Gly 130 135 140Cys Leu Val Lys Asp Tyr
Phe Pro Glu Pro Val Thr Val Ser Trp Asn145 150 155 160Ser Gly Ala
Leu Thr Ser Gly Val His Thr Phe Pro Ala Val Leu Gln 165 170 175Ser
Ser Gly Leu Tyr Ser Leu Ser Ser Val Val Thr Val Pro Ser Ser 180 185
190Ser Leu Gly Thr Gln Thr Tyr Ile Cys Asn Val Asn His Lys Pro Ser
195 200 205Asn Thr Lys Val Asp Lys Arg Val Glu Pro Lys Ser Cys Asp
Lys Thr 210 215 220His Thr Cys Pro Pro Cys Pro Ala Pro Glu Leu Leu
Gly Gly Pro Ser225 230 235 240Val Phe Leu Phe Pro Pro Lys Pro Lys
Asp Thr Leu Met Ile Ser Arg 245 250 255Thr Pro Glu Val Thr Cys Val
Val Val Asp Val Ser His Glu Asp Pro 260 265 270Glu Val Lys Phe Asn
Trp Tyr Val Asp Gly Val Glu Val His Asn Ala 275 280 285Lys Thr Lys
Pro Arg Glu Glu Gln Tyr Asn Ser Thr Tyr Arg Val Val 290 295 300Ser
Val Leu Thr Val Leu His Gln Asp Trp Leu Asn Gly Lys Glu Tyr305 310
315 320Lys Cys Lys Val Ser Asn Lys Ala Leu Pro Ala Pro Ile Glu Lys
Thr 325 330 335Ile Ser Lys Ala Lys Gly Gln Pro Arg Glu Pro Gln Val
Tyr Thr Leu 340 345 350Pro Pro Ser Arg Glu Glu Met Thr Lys Asn Gln
Val Ser Leu Thr Cys 355 360 365Leu Val Lys Gly Phe Tyr Pro Ser Asp
Ile Ala Val Glu Trp Glu Ser 370 375 380Asn Gly Gln Pro Glu Asn Asn
Tyr Lys Thr Thr Pro Pro Val Leu Asp385 390 395 400Ser Asp Gly Ser
Phe Phe Leu Tyr Ser Lys Leu Thr Val Asp Lys Ser 405 410 415Arg Trp
Gln Gln Gly Asn Val Phe Ser Cys Ser Val Met His Glu Ala 420 425
430Leu His Asn His Tyr Thr Gln Lys Ser Leu Ser Leu Ser Pro Gly Lys
435 440 445298PRTArtificial SequenceSynthetic 29Gly Phe Asn Ile Lys
Asp Thr His1 53011PRTArtificial SequenceSynthetic 30Ala Arg Trp Gly
Thr Asn Val Tyr Phe Ala Tyr1 5 1031118PRTArtificial
SequenceSynthetic 31Gln Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys
Lys Pro Gly Ala1 5 10 15Ser Val Lys Val Ser Cys Lys Ala Ser Gly Phe
Asn Ile Lys Asp Thr 20 25 30His Met His Trp Val Arg Gln Ala Pro Gly
Gln Arg Leu Glu Trp Ile 35 40 45Gly Arg Ile Asp Pro Ala Asn Gly Asn
Thr Glu Tyr Asp Gln Lys Phe 50 55 60Gln Gly Arg Val Thr Ile Thr Val
Asp Thr Ser Ala Ser Thr Ala Tyr65 70 75 80Met Glu Leu Ser Ser Leu
Arg Ser Glu Asp Thr Ala Val Tyr Tyr Cys 85 90 95Ala Arg Trp Gly Thr
Asn Val Tyr Phe Ala Tyr Trp Gly Gln Gly Thr 100 105 110Leu Val Thr
Val Ser Ser 11532448PRTArtificial SequenceSynthetic 32Gln Val Gln
Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala1 5 10 15Ser Val
Lys Val Ser Cys Lys Ala Ser Gly Phe Asn Ile Lys Asp Thr 20 25 30His
Met His Trp Val Arg Gln Ala Pro Gly Gln Arg Leu Glu Trp Ile 35 40
45Gly Arg Ile Asp Pro Ala Asn Gly Asn Thr Glu Tyr Asp Gln Lys Phe
50 55 60Gln Gly Arg Val Thr Ile Thr Val Asp Thr Ser Ala Ser Thr Ala
Tyr65 70 75 80Met Glu Leu Ser Ser Leu Arg Ser Glu Asp Thr Ala Val
Tyr Tyr Cys 85 90 95Ala Arg Trp Gly Thr Asn Val Tyr Phe Ala Tyr Trp
Gly Gln Gly Thr 100 105 110Leu Val Thr Val Ser Ser Ala Ser Thr Lys
Gly Pro Ser Val Phe Pro 115 120 125Leu Ala Pro Ser Ser Lys Ser Thr
Ser Gly Gly Thr Ala Ala Leu Gly 130 135 140Cys Leu Val Lys Asp Tyr
Phe Pro Glu Pro Val Thr Val Ser Trp Asn145 150 155 160Ser Gly Ala
Leu Thr Ser Gly Val His Thr Phe Pro Ala Val Leu Gln 165 170 175Ser
Ser Gly Leu Tyr Ser Leu Ser Ser Val Val Thr Val Pro Ser Ser 180 185
190Ser Leu Gly Thr Gln Thr Tyr Ile Cys Asn Val Asn His Lys Pro Ser
195 200 205Asn Thr Lys Val Asp Lys Arg Val Glu Pro Lys Ser Cys Asp
Lys Thr 210 215 220His Thr Cys Pro Pro Cys Pro Ala Pro Glu Leu Leu
Gly Gly Pro Ser225 230 235 240Val Phe Leu Phe Pro Pro Lys Pro Lys
Asp Thr Leu Met Ile Ser Arg 245 250 255Thr Pro Glu Val Thr Cys Val
Val Val Asp Val Ser His Glu Asp Pro 260 265 270Glu Val Lys Phe Asn
Trp Tyr Val Asp Gly Val Glu Val His Asn Ala 275 280 285Lys Thr Lys
Pro Arg Glu Glu Gln Tyr Asn Ser Thr Tyr Arg Val Val 290 295 300Ser
Val Leu Thr Val Leu His Gln Asp Trp Leu Asn Gly Lys Glu Tyr305 310
315 320Lys Cys Lys Val Ser Asn Lys Ala Leu Pro Ala Pro Ile Glu Lys
Thr 325 330 335Ile Ser Lys Ala Lys Gly Gln Pro Arg Glu Pro Gln Val
Tyr Thr Leu 340 345 350Pro Pro Ser Arg Glu Glu Met Thr Lys Asn Gln
Val Ser Leu Thr Cys 355 360 365Leu Val Lys Gly Phe Tyr Pro Ser Asp
Ile Ala Val Glu Trp Glu Ser 370 375 380Asn Gly Gln Pro Glu Asn Asn
Tyr Lys Thr Thr Pro Pro Val Leu Asp385 390 395 400Ser Asp Gly Ser
Phe Phe Leu Tyr Ser Lys Leu Thr Val Asp Lys Ser 405 410 415Arg Trp
Gln Gln Gly Asn Val Phe Ser Cys Ser Val Met His Glu Ala 420 425
430Leu His Asn His Tyr Thr Gln Lys Ser Leu Ser Leu Ser Pro Gly Lys
435 440 445335PRTArtificial SequenceSynthetic 33Ser Ser Val Ser
Tyr1 5349PRTArtificial SequenceSynthetic 34Gln Gln Tyr His Ser Tyr
Pro Pro Thr1 535106PRTArtificial SequenceSynthetic 35Asp Ile Gln
Leu Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg
Val Thr Ile Thr Cys Ser Ala Ser Ser Ser Val Ser Tyr Met 20 25 30Tyr
Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Pro Trp Ile Tyr 35 40
45Arg Thr Ser Asn Leu Ala Ser Gly Val Pro Ser Arg Phe Ser Gly Ser
50 55 60Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro
Glu65 70 75 80Asp Phe Ala Thr Tyr Tyr Cys Gln Gln Tyr His Ser Tyr
Pro Pro Thr 85 90 95Phe Gly Gly Gly Thr Lys Val Glu Ile Lys 100
10536213PRTArtificial SequenceSynthetic 36Asp Ile Gln Leu Thr Gln
Ser Pro Ser Ser Leu Ser Ala Ser Val Gly1 5 10 15Asp Arg Val Thr Ile
Thr Cys Ser Ala Ser Ser Ser Val Ser Tyr Met 20 25 30Tyr Trp Tyr Gln
Gln Lys Pro Gly Lys Ala Pro Lys Pro Trp Ile Tyr 35 40 45Arg Thr Ser
Asn Leu Ala Ser Gly Val Pro Ser Arg Phe Ser Gly Ser 50 55 60Gly Ser
Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro Glu65 70 75
80Asp Phe Ala Thr Tyr Tyr Cys Gln Gln Tyr His Ser Tyr Pro Pro Thr
85 90 95Phe Gly Gly Gly Thr Lys Val Glu Ile Lys Arg Thr Val Ala Ala
Pro 100 105 110Ser Val Phe Ile Phe Pro Pro Ser Asp Glu Gln Leu Lys
Ser Gly Thr 115 120 125Ala Ser Val Val Cys Leu Leu Asn Asn Phe Tyr
Pro Arg Glu Ala Lys 130 135 140Val Gln Trp Lys Val Asp Asn Ala Leu
Gln Ser Gly Asn Ser Gln Glu145 150 155 160Ser Val Thr Glu Gln Asp
Ser Lys Asp Ser Thr Tyr Ser Leu Ser Ser 165 170 175Thr Leu Thr Leu
Ser Lys Ala Asp Tyr Glu Lys His Lys Val Tyr Ala 180 185 190Cys Glu
Val Thr His Gln Gly Leu Ser Ser Pro Val Thr Lys Ser Phe 195 200
205Asn Arg Gly Glu Cys 2103710PRTArtificial SequenceSynthetic 37Gly
Gly Ser Ile Ser Ser Gly Asp Tyr Phe1 5 10387PRTArtificial
SequenceSynthetic 38Ile His Asn Ser Gly Thr Thr1 53914PRTArtificial
SequenceSynthetic 39Ala Arg Asp Arg Gly Gly Asp Tyr Tyr Tyr Gly Met
Asp Val1 5 1040122PRTArtificial SequenceSynthetic 40Gln Val Gln Leu
Gln Glu Ser Gly Pro Gly Leu Val Lys Pro Ser Gln1 5 10 15Thr Leu Ser
Leu Thr Cys Thr Val Ser Gly Gly Ser Ile Ser Ser Gly 20 25 30Asp Tyr
Phe Trp Ser Trp Ile Arg Gln Leu Pro Gly Lys Gly Leu Glu 35 40 45Cys
Ile Gly His Ile His Asn Ser Gly Thr Thr Tyr Tyr Asn Pro Ser 50 55
60Leu Lys Ser Arg Val Thr Ile Ser Val Asp Thr Ser Lys Lys Gln Phe65
70 75 80Ser Leu Arg Leu Ser Ser Val Thr Ala Ala Asp Thr Ala Val Tyr
Tyr 85 90 95Cys Ala Arg Asp Arg Gly Gly Asp Tyr Tyr Tyr Gly Met Asp
Val Trp 100 105 110Gly Gln Gly Thr Thr Val Thr Val Ser Ser 115
120417PRTArtificial SequenceSynthetic 41Gln Gly Ile Ser Arg Ser
Tyr1 5429PRTArtificial SequenceSynthetic 42Gln Gln Phe Gly Ser Ser
Pro Trp Thr1 543108PRTArtificial SequenceSynthetic 43Glu Ile Val
Leu Thr Gln Ser Pro Gly Thr Leu Ser Leu Ser Pro Gly1 5 10 15Glu Arg
Ala Thr Leu Ser Cys Arg Ala Ser Gln Gly Ile Ser Arg Ser 20 25 30Tyr
Leu Ala Trp Tyr Gln Gln Lys Pro Gly Gln Ala Pro Ser Leu Leu 35 40
45Ile Tyr Gly Ala Ser Ser Arg Ala Thr Gly Ile Pro Asp Arg Phe Ser
50 55 60Gly Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Arg Leu
Glu65 70 75 80Pro Glu Asp Phe Ala Val Tyr Tyr Cys Gln Gln Phe Gly
Ser Ser Pro 85 90 95Trp Thr Phe Gly Gln Gly Thr Lys Val Glu Ile Lys
100 105448PRTArtificial SequenceSynthetic 44Gly Phe Thr Phe Ser Ser
Tyr Val1 5458PRTArtificial SequenceSynthetic 45Ile Ser Ser Gly Gly
Ser Tyr Thr1 54612PRTArtificial SequenceSynthetic 46Ala Arg Arg Gly
Asp Ser Met Ile Thr Thr Asp Tyr1 5 1047449PRTArtificial
SequenceSynthetic 47Glu Val Met Leu Val Glu Ser Gly Gly Gly Leu Val
Lys Pro Gly Gly1 5 10 15Ser Leu Lys Leu Ser Cys Ala Ala Ser Gly Phe
Thr Phe Ser Ser Tyr 20 25 30Val Met Ser Trp Val Arg Gln Thr Pro Glu
Lys Arg Leu Glu Trp Val 35 40 45Ala Thr Ile Ser Ser Gly Gly Ser Tyr
Thr Tyr Tyr Pro Asp Ser Val 50 55 60Lys Gly
Arg Phe Thr Ile Ser Arg Asp Asn Ala Lys Asn Thr Leu Tyr65 70 75
80Leu Gln Met Ser Ser Leu Arg Ser Glu Asp Thr Ala Met Tyr Tyr Cys
85 90 95Ala Arg Arg Gly Asp Ser Met Ile Thr Thr Asp Tyr Trp Gly Gln
Gly 100 105 110Thr Thr Leu Thr Val Ser Ser Ala Ser Thr Lys Gly Pro
Ser Val Phe 115 120 125Pro Leu Ala Pro Ser Ser Lys Ser Thr Ser Gly
Gly Thr Ala Ala Leu 130 135 140Gly Cys Leu Val Lys Asp Tyr Phe Pro
Glu Pro Val Thr Val Ser Trp145 150 155 160Asn Ser Gly Ala Leu Thr
Ser Gly Val His Thr Phe Pro Ala Val Leu 165 170 175Gln Ser Ser Gly
Leu Tyr Ser Leu Ser Ser Val Val Thr Val Pro Ser 180 185 190Ser Ser
Leu Gly Thr Gln Thr Tyr Ile Cys Asn Val Asn His Lys Pro 195 200
205Ser Asn Thr Lys Val Asp Lys Arg Val Glu Pro Lys Ser Cys Asp Lys
210 215 220Thr His Thr Cys Pro Pro Cys Pro Ala Pro Glu Leu Leu Gly
Gly Pro225 230 235 240Ser Val Phe Leu Phe Pro Pro Lys Pro Lys Asp
Thr Leu Met Ile Ser 245 250 255Arg Thr Pro Glu Val Thr Cys Val Val
Val Asp Val Ser His Glu Asp 260 265 270Pro Glu Val Lys Phe Asn Trp
Tyr Val Asp Gly Val Glu Val His Asn 275 280 285Ala Lys Thr Lys Pro
Arg Glu Glu Gln Tyr Asn Ser Thr Tyr Arg Val 290 295 300Val Ser Val
Leu Thr Val Leu His Gln Asp Trp Leu Asn Gly Lys Glu305 310 315
320Tyr Lys Cys Lys Val Ser Asn Lys Ala Leu Pro Ala Pro Ile Glu Lys
325 330 335Thr Ile Ser Lys Ala Lys Gly Gln Pro Arg Glu Pro Gln Val
Tyr Thr 340 345 350Leu Pro Pro Ser Arg Glu Glu Met Thr Lys Asn Gln
Val Ser Leu Thr 355 360 365Cys Leu Val Lys Gly Phe Tyr Pro Ser Asp
Ile Ala Val Glu Trp Glu 370 375 380Ser Asn Gly Gln Pro Glu Asn Asn
Tyr Lys Thr Thr Pro Pro Val Leu385 390 395 400Asp Ser Asp Gly Ser
Phe Phe Leu Tyr Ser Lys Leu Thr Val Asp Lys 405 410 415Ser Arg Trp
Gln Gln Gly Asn Val Phe Ser Cys Ser Val Met His Glu 420 425 430Ala
Leu His Asn His Tyr Thr Gln Lys Ser Leu Ser Leu Ser Pro Gly 435 440
445Lys486PRTArtificial SequenceSynthetic 48Gln Asp Val Gly Thr Ala1
5498PRTArtificial SequenceSynthetic 49Gln Gln Tyr Ser Ser Tyr Arg
Thr1 550213PRTArtificial SequenceSynthetic 50Asp Ile Val Met Thr
Gln Ser His Lys Phe Met Ser Thr Ser Val Gly1 5 10 15Asp Arg Val Ser
Ile Thr Cys Lys Ala Ser Gln Asp Val Gly Thr Ala 20 25 30Val Ala Trp
Tyr Gln Gln Lys Pro Gly Gln Ser Pro Lys Leu Leu Ile 35 40 45Tyr Trp
Ala Ser Thr Arg His Thr Gly Val Pro Asp Arg Phe Thr Gly 50 55 60Ser
Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Asn Val Gln Ser65 70 75
80Glu Asp Leu Ala Asp Tyr Phe Cys Gln Gln Tyr Ser Ser Tyr Arg Thr
85 90 95Phe Gly Gly Gly Thr Lys Leu Glu Ile Lys Arg Thr Val Ala Ala
Pro 100 105 110Ser Val Phe Ile Phe Pro Pro Ser Asp Glu Gln Leu Lys
Ser Gly Thr 115 120 125Ala Ser Val Val Cys Leu Leu Asn Asn Phe Tyr
Pro Arg Glu Ala Lys 130 135 140Val Gln Trp Lys Val Asp Asn Ala Leu
Gln Ser Gly Asn Ser Gln Glu145 150 155 160Ser Val Thr Glu Gln Asp
Ser Lys Asp Ser Thr Tyr Ser Leu Ser Ser 165 170 175Thr Leu Thr Leu
Ser Lys Ala Asp Tyr Glu Lys His Lys Val Tyr Ala 180 185 190Cys Glu
Val Thr His Gln Gly Leu Ser Ser Pro Val Thr Lys Ser Phe 195 200
205Asn Arg Gly Glu Cys 210
D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012
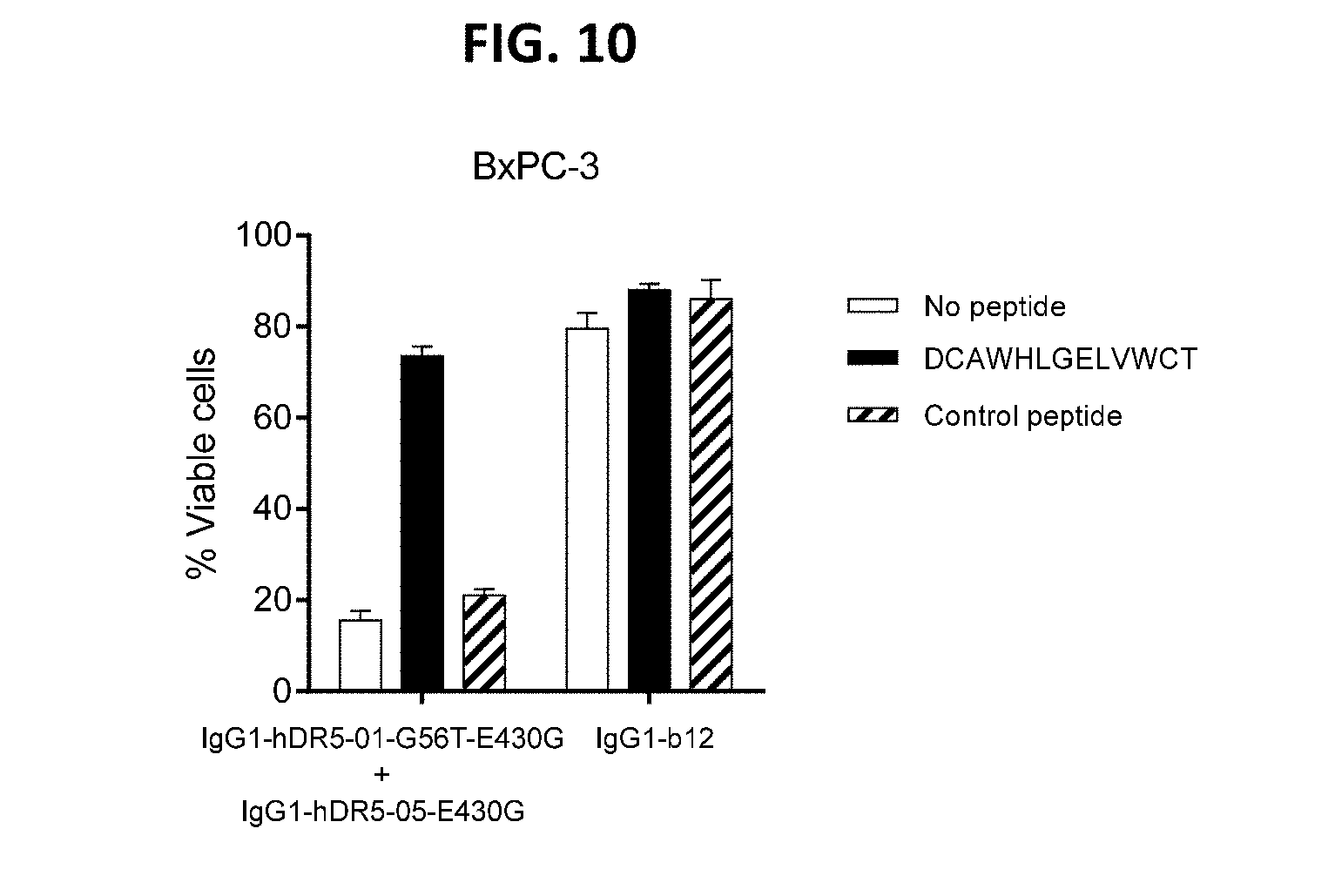
D00013
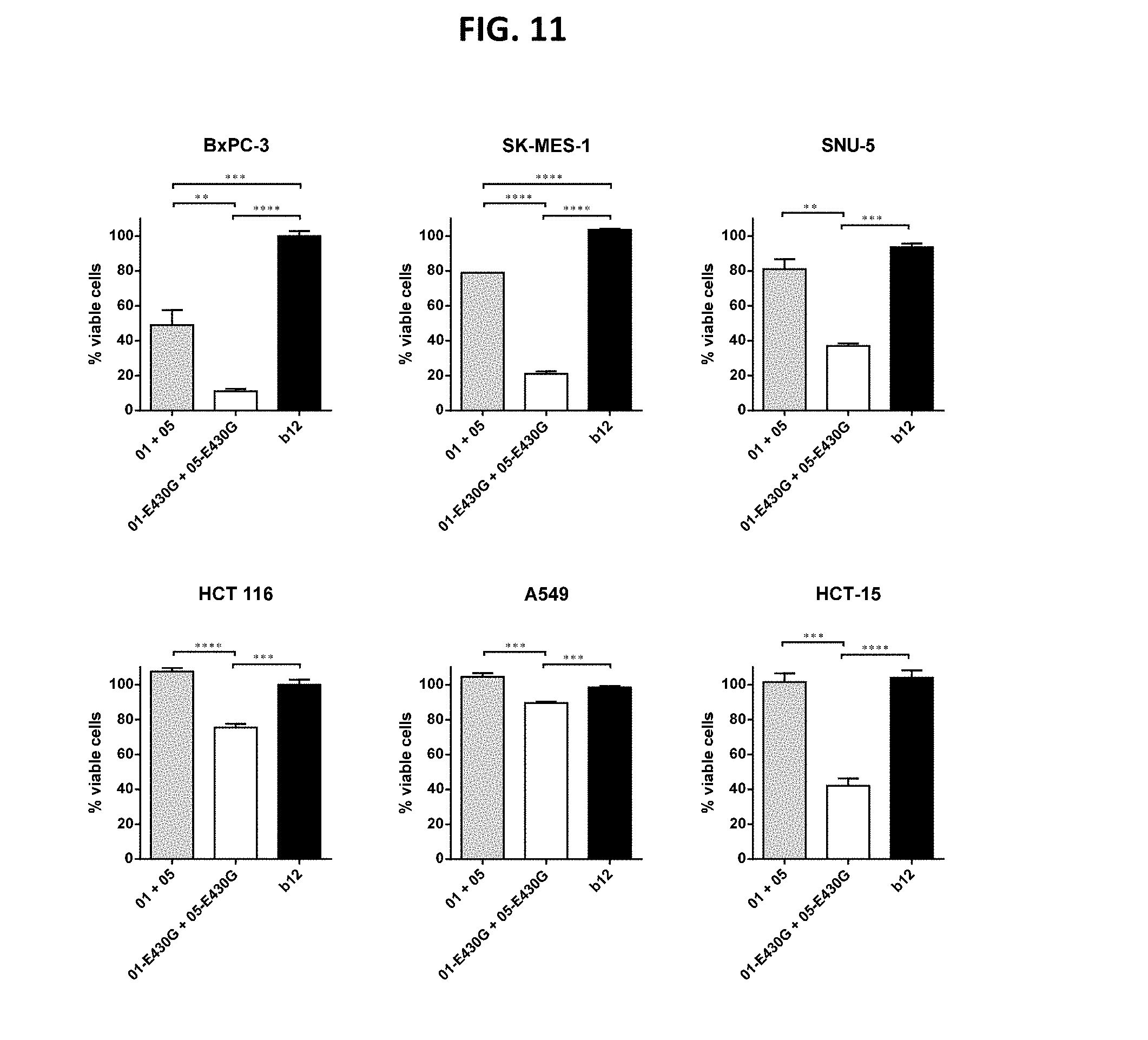
D00014
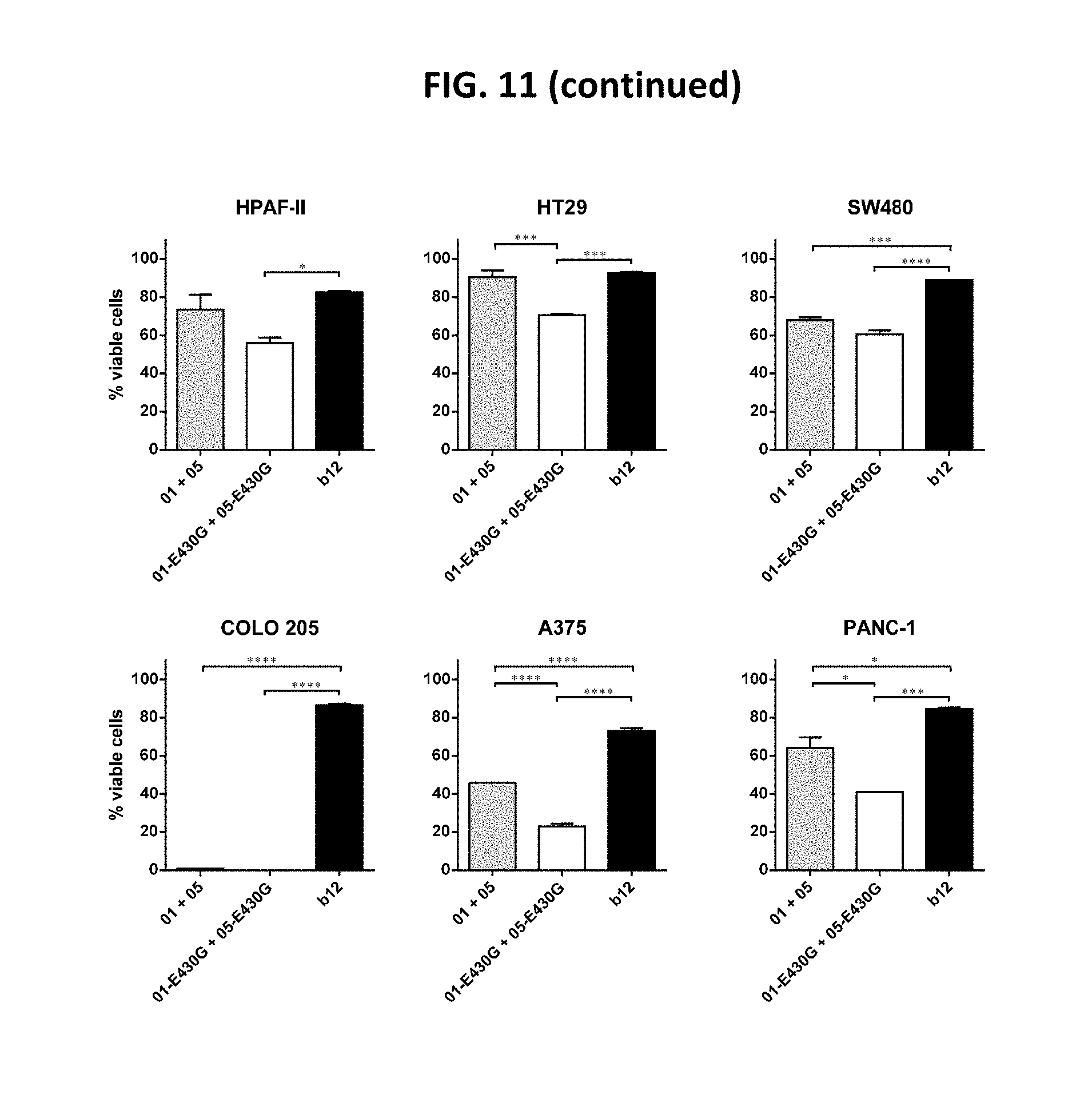
D00015
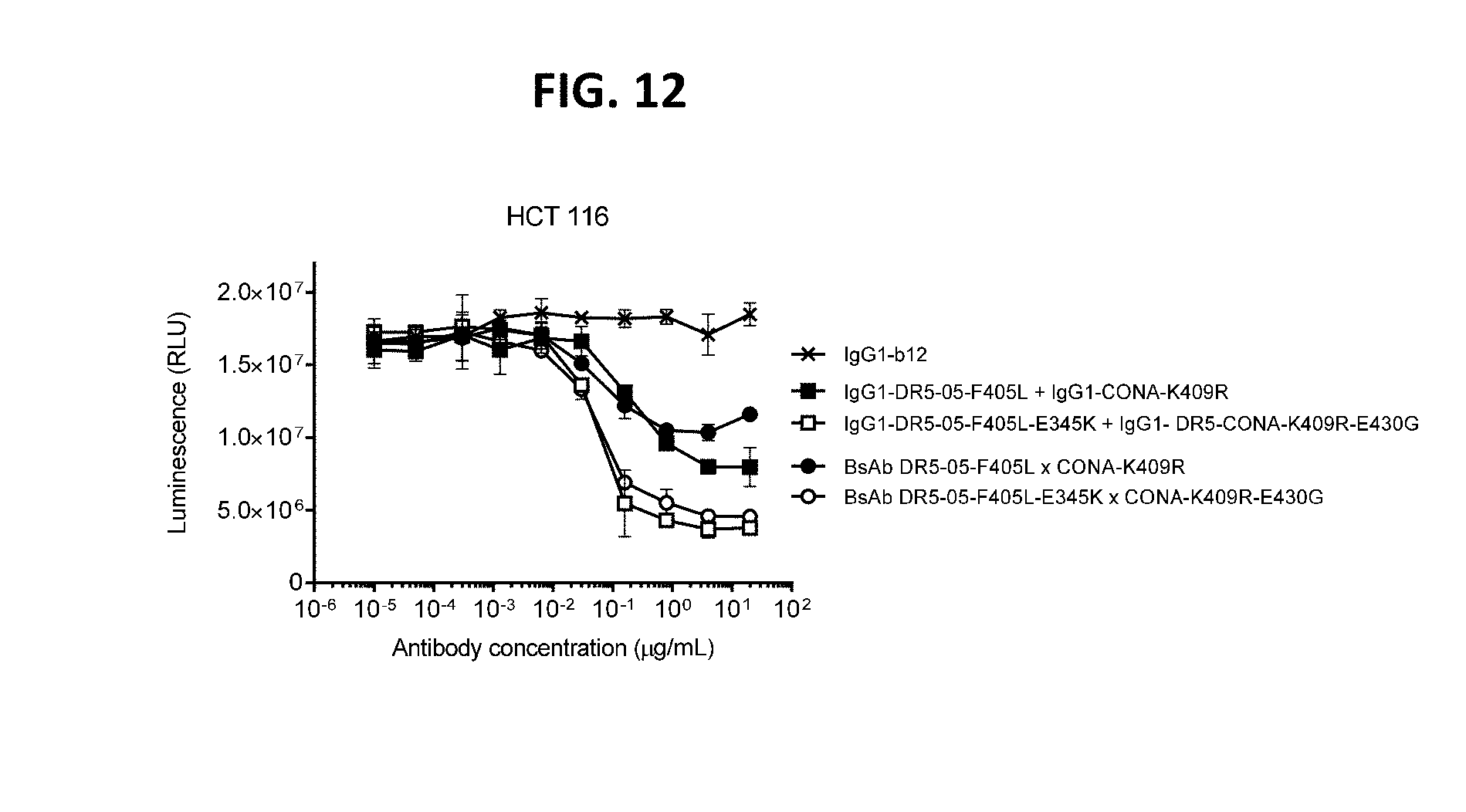
D00016

D00017
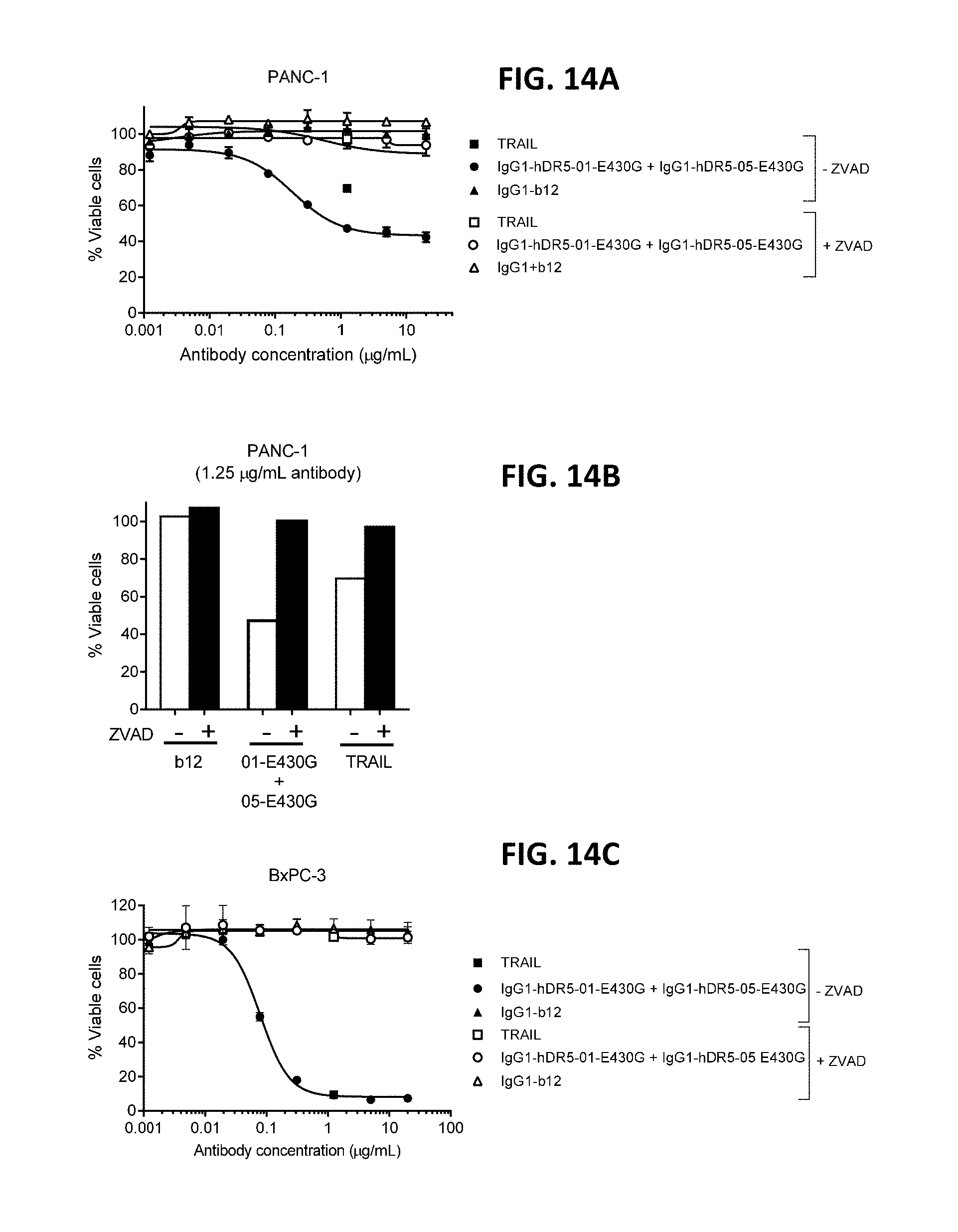
D00018

D00019
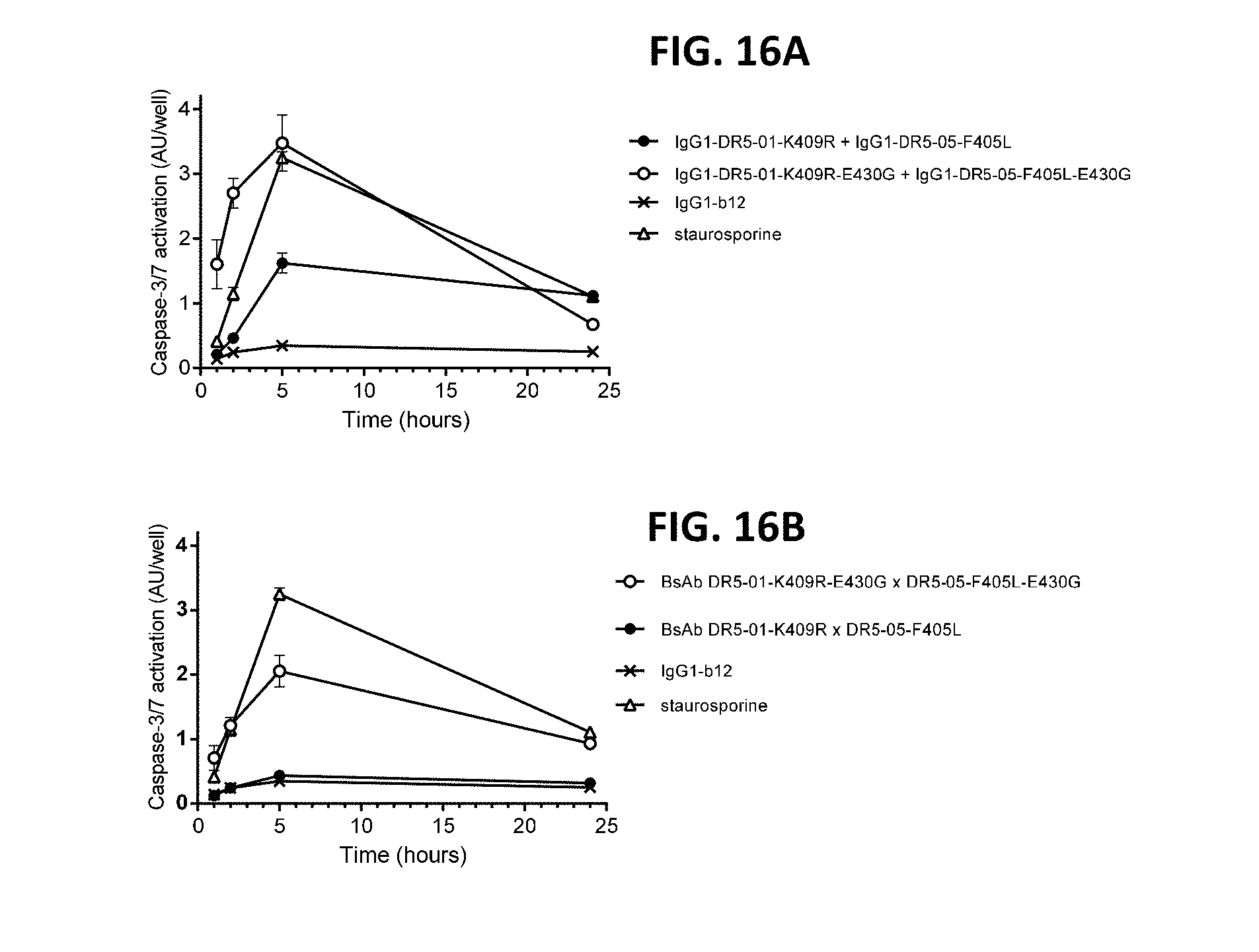
D00020
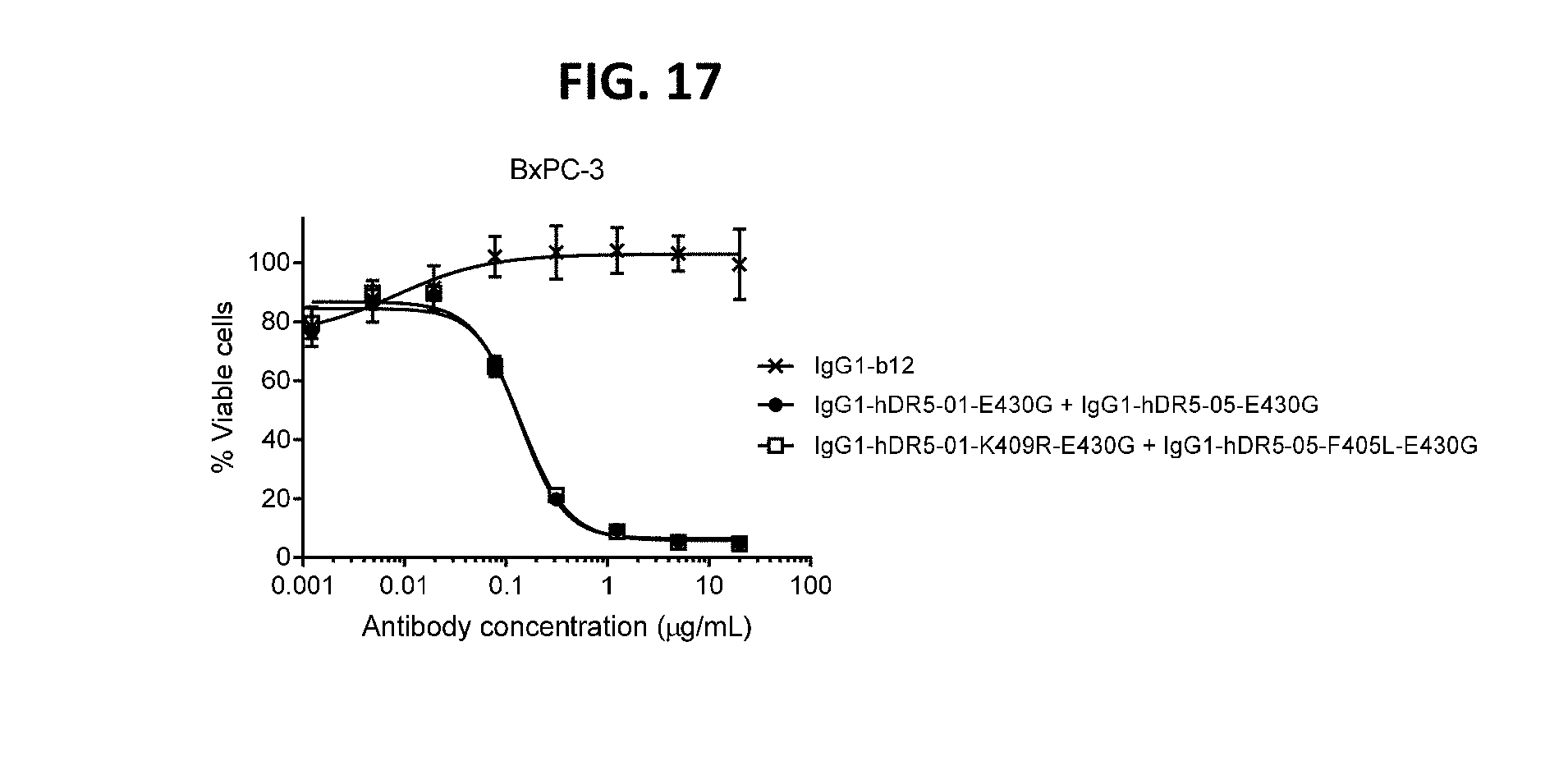
D00021
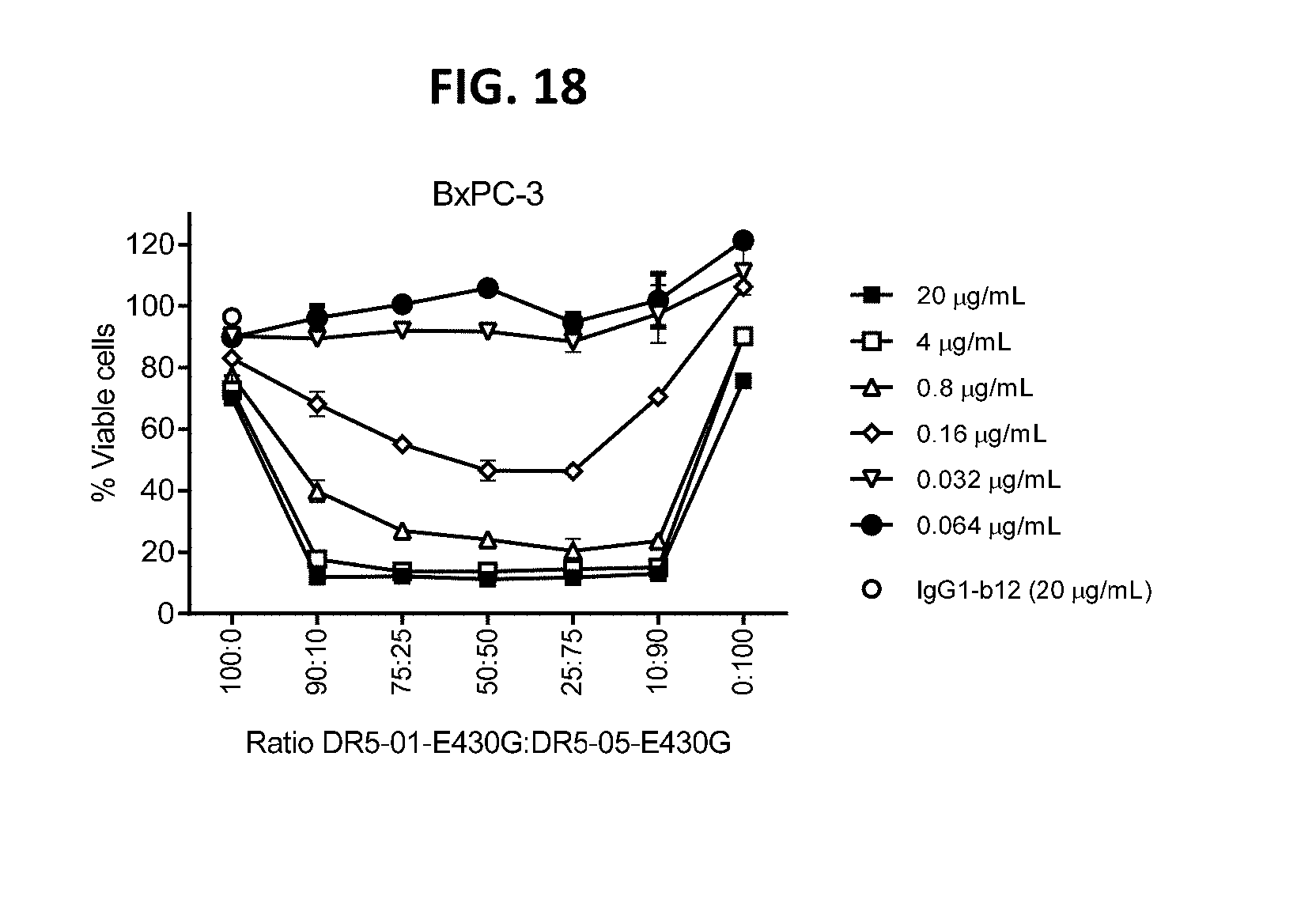
D00022
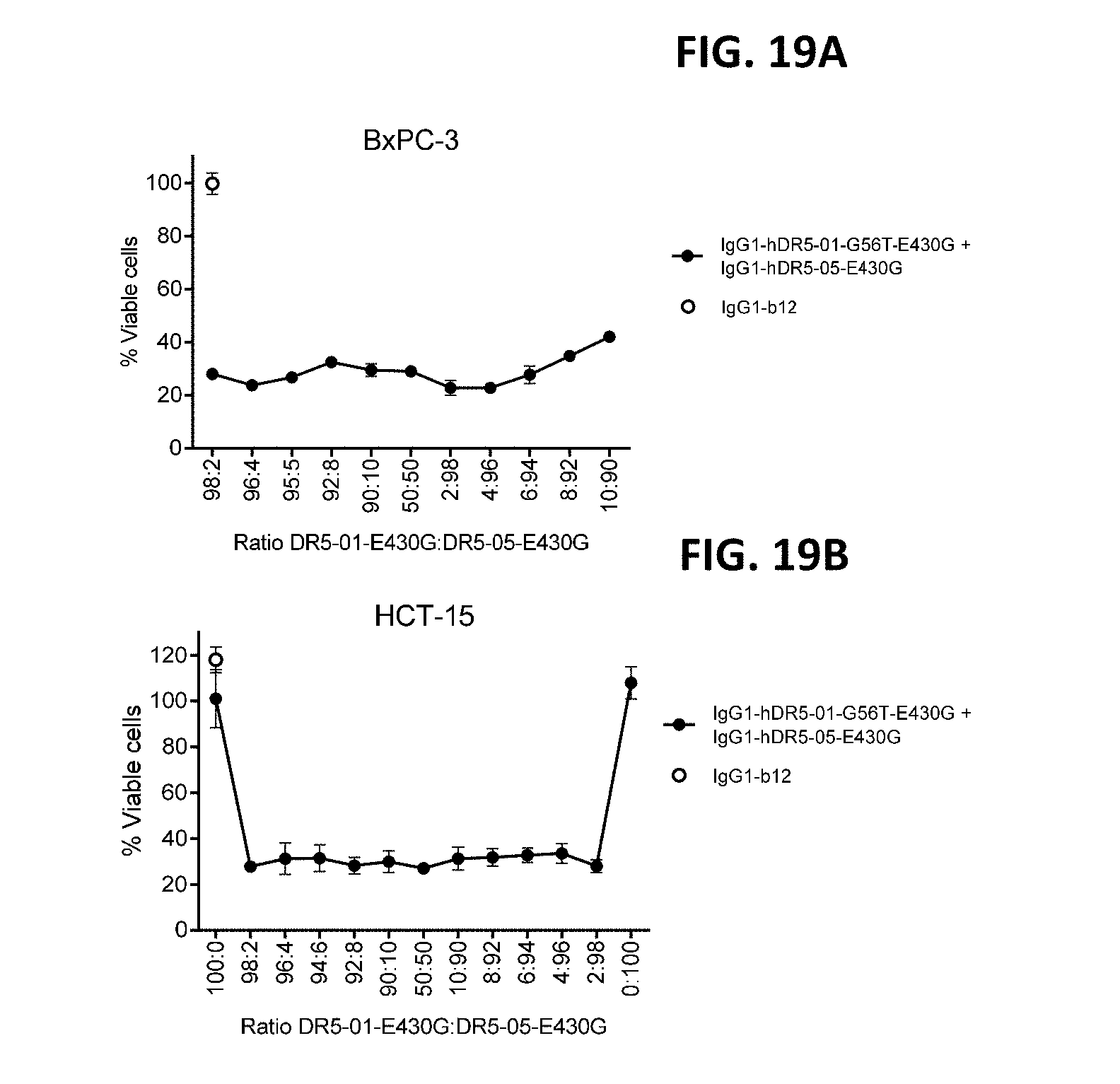
D00023
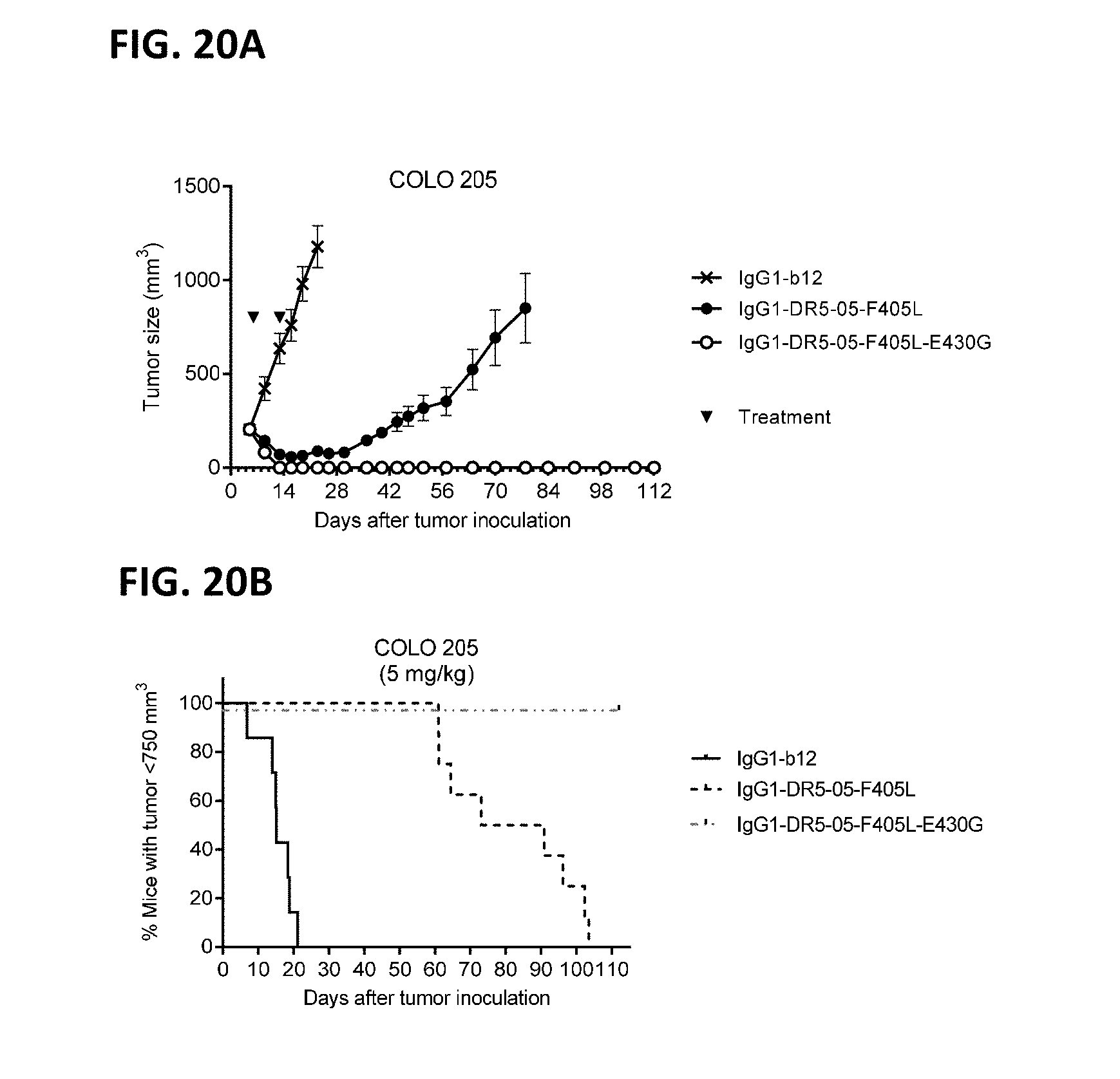
D00024
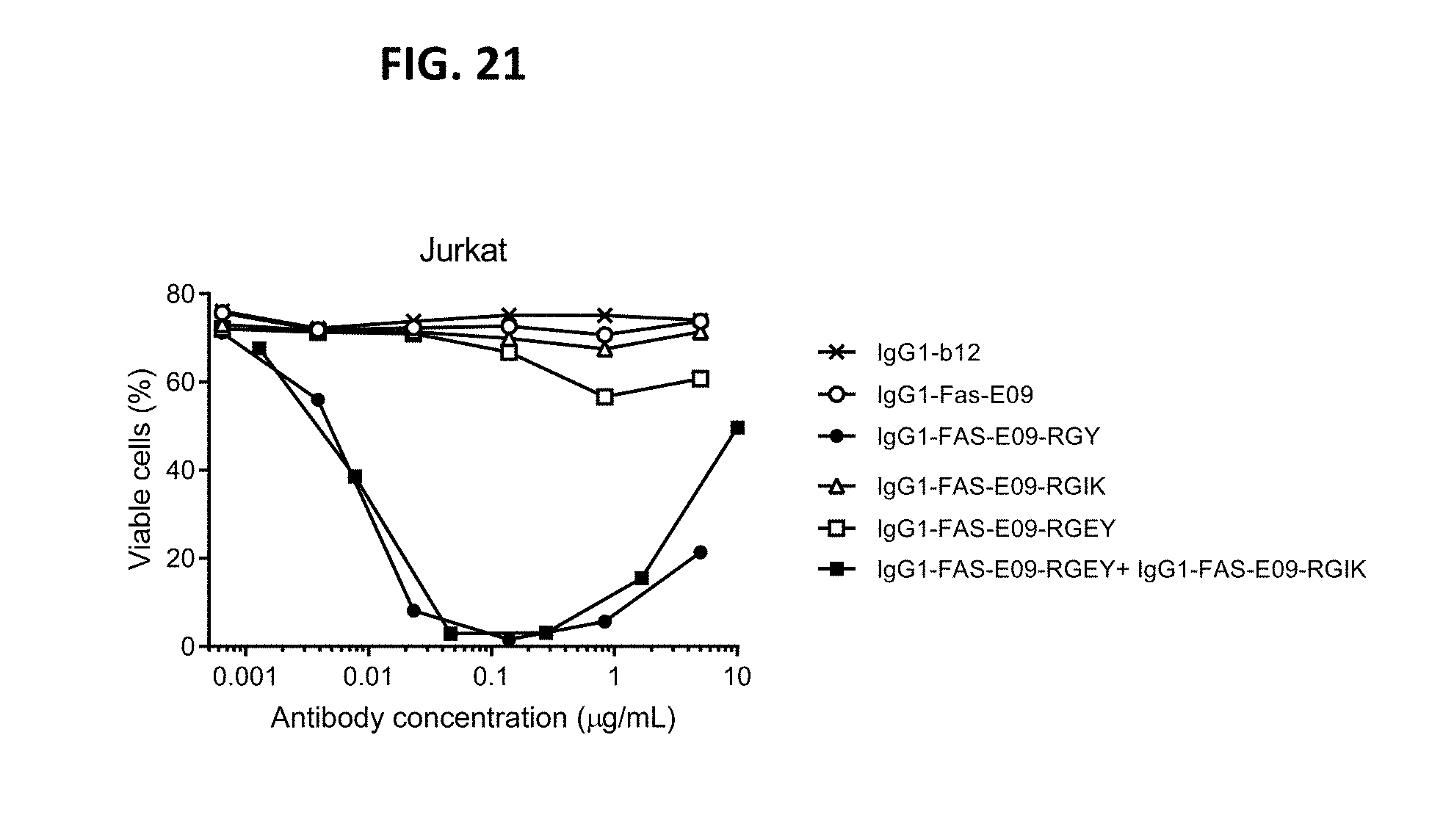
D00025
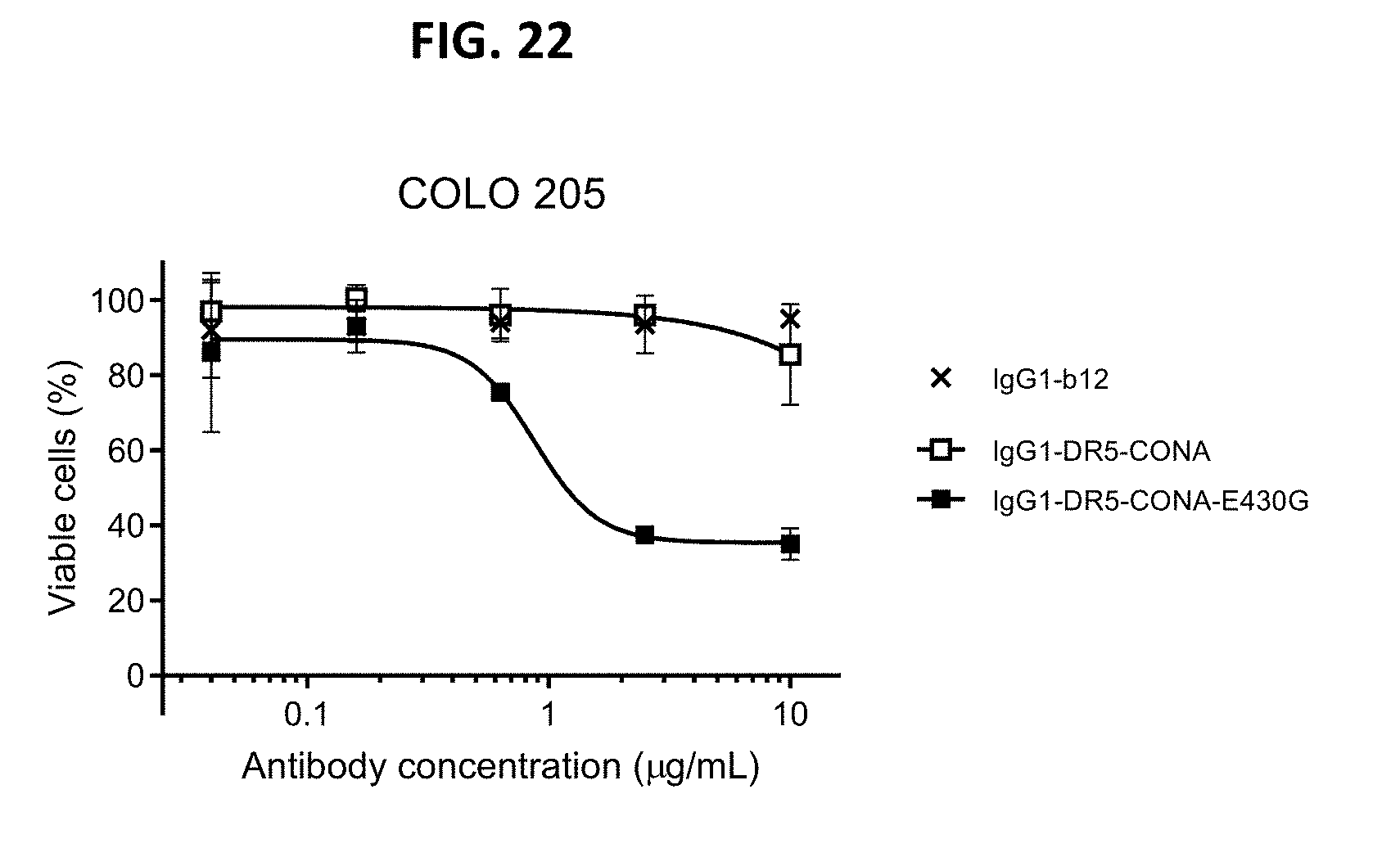
D00026
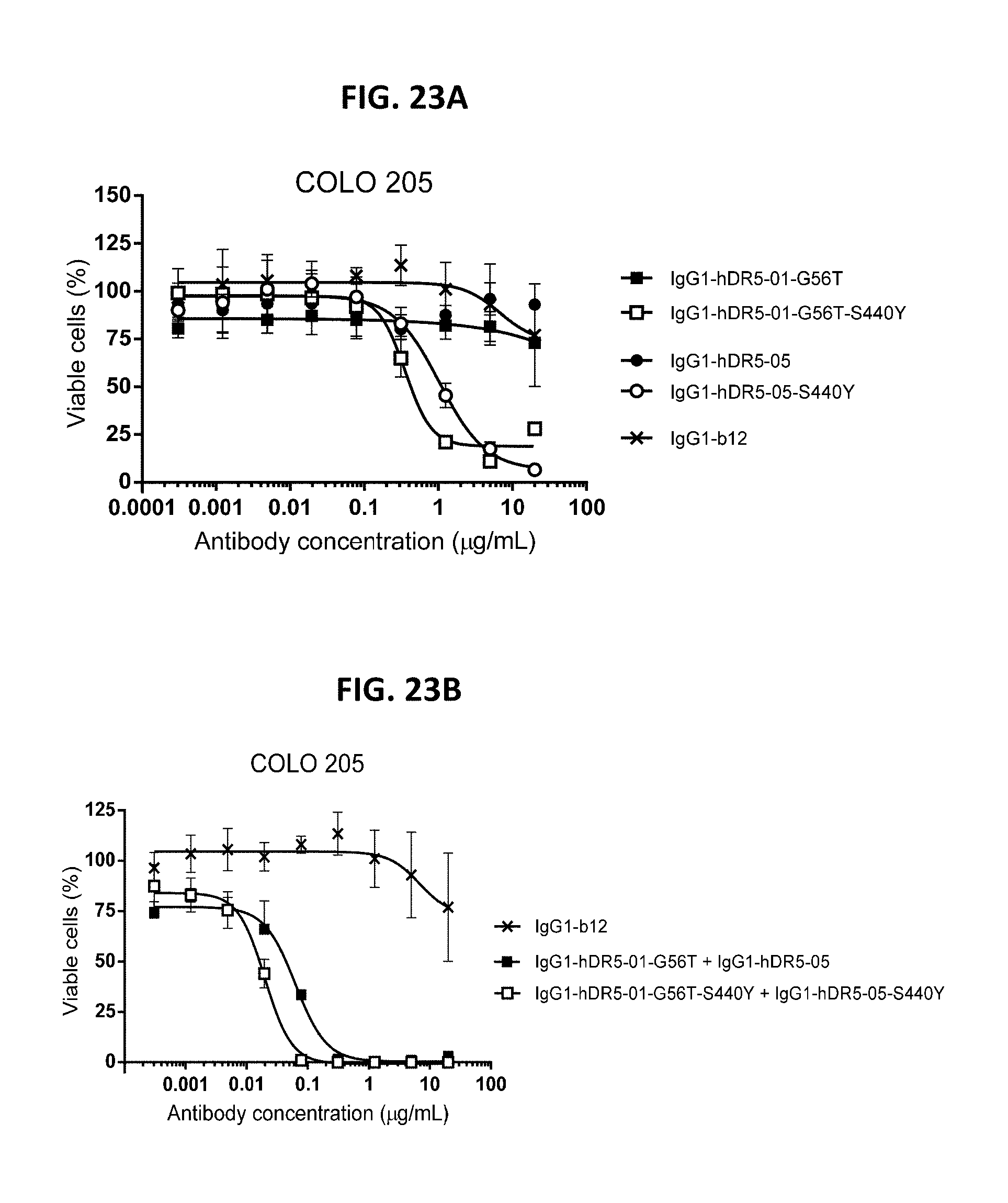
D00027

D00028
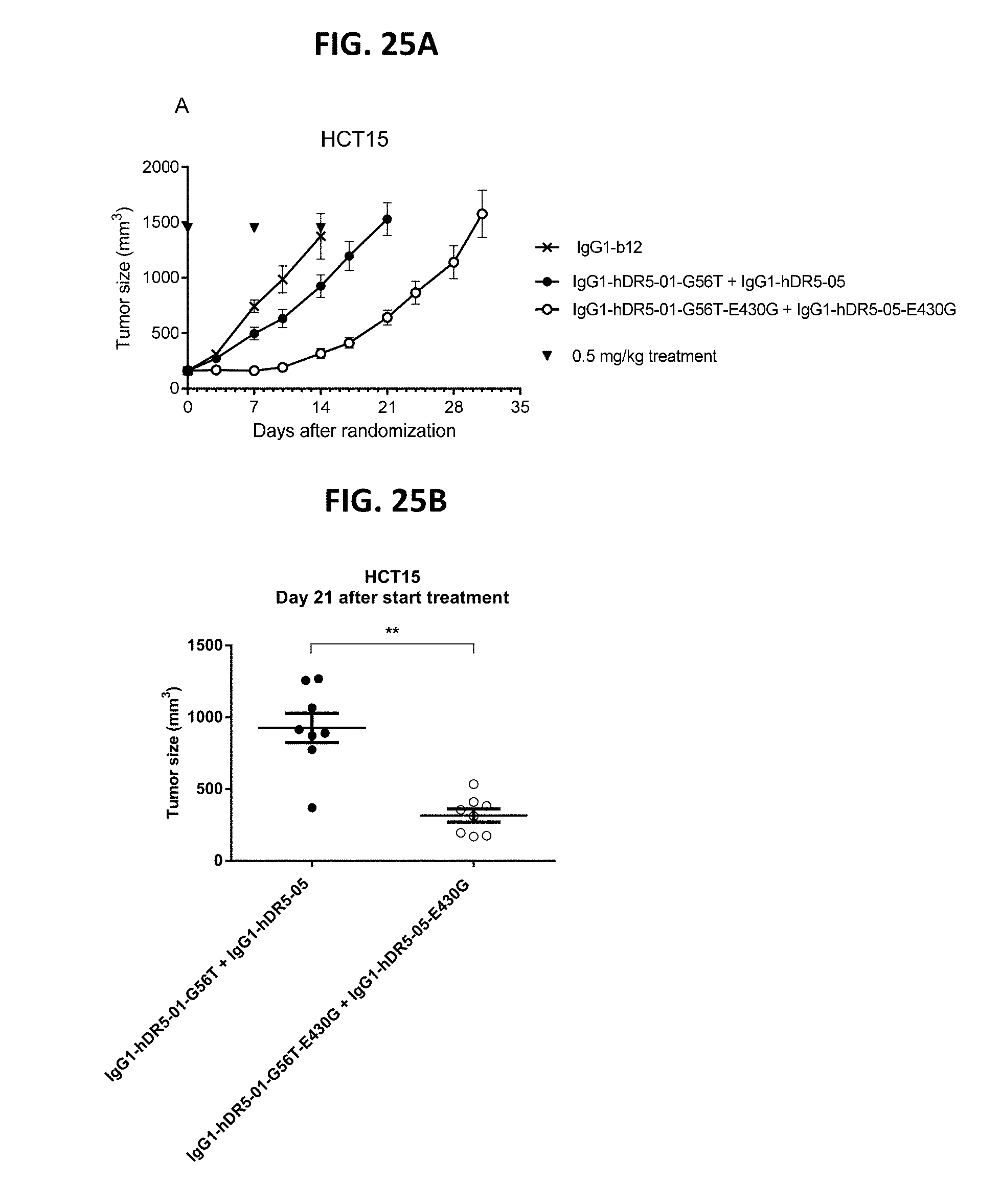
D00029
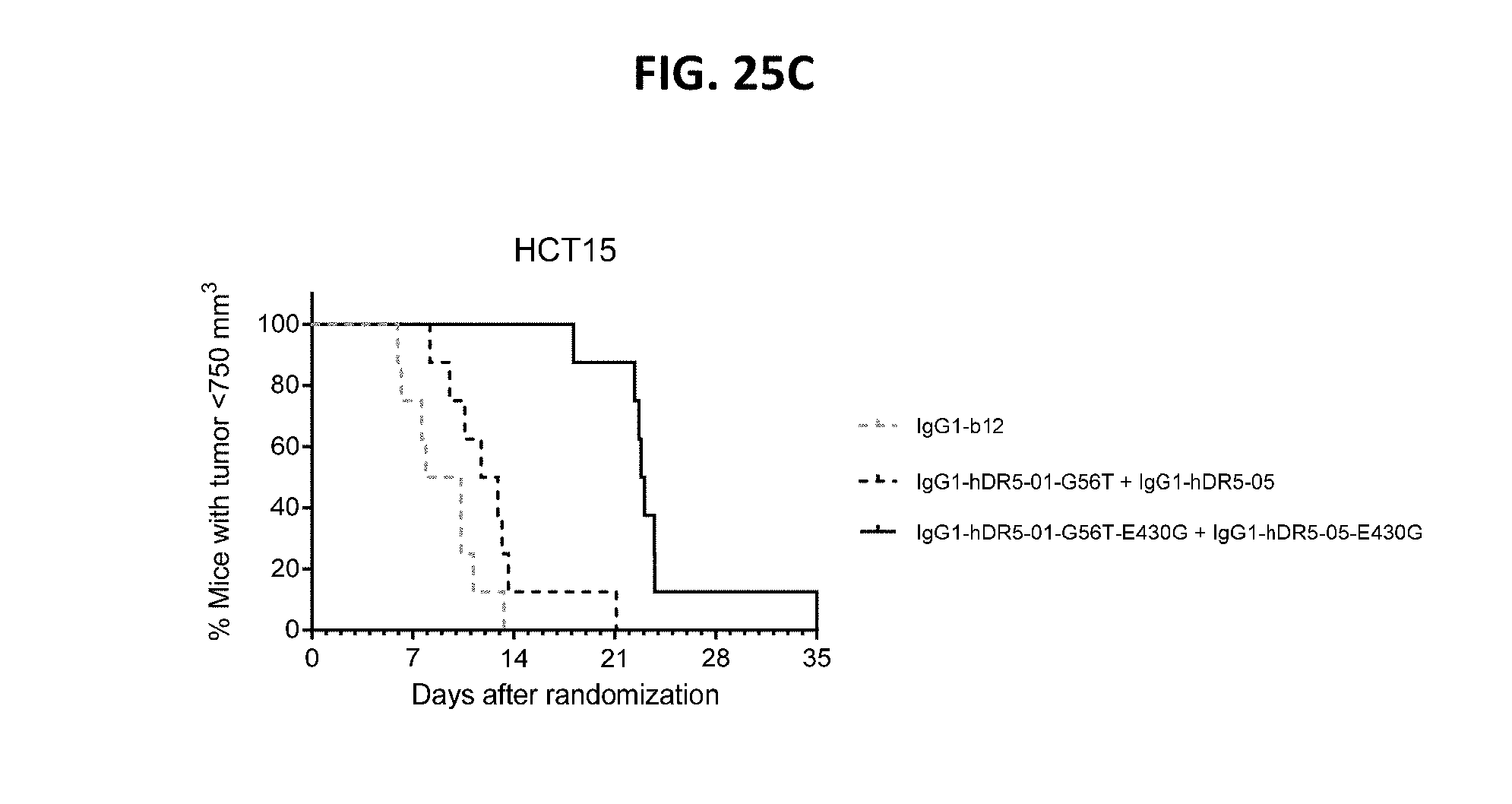
D00030
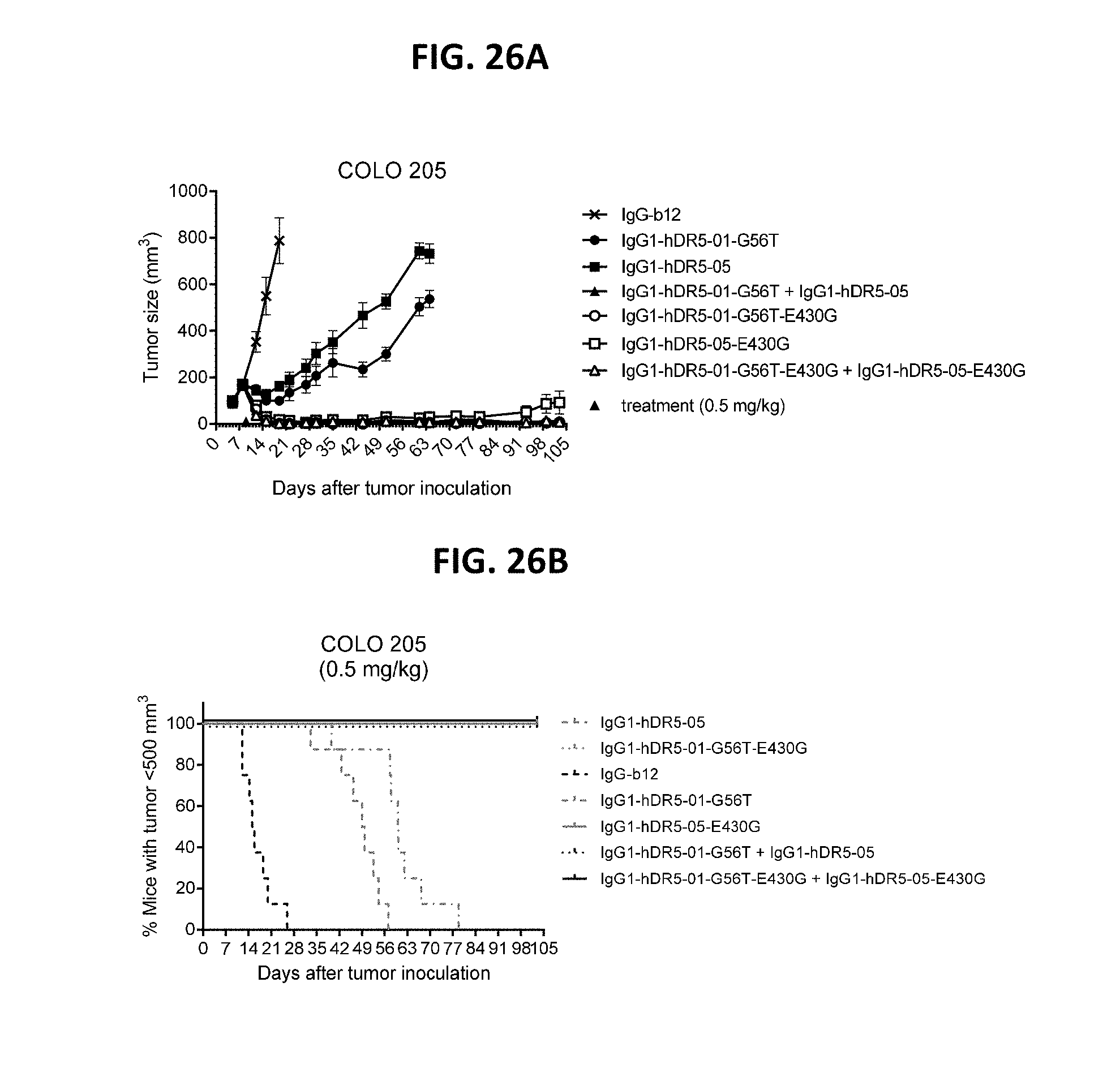
S00001
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.