2-spiro-5- And 6-hydroxamic Acid Indanes As Hdac Inhibitors
Zheng; Xiaozhang ; et al.
U.S. patent application number 16/309980 was filed with the patent office on 2019-07-04 for 2-spiro-5- and 6-hydroxamic acid indanes as hdac inhibitors. The applicant listed for this patent is FORMA Therapeutics, Inc.. Invention is credited to Pui Yee Ng, Mary-Margaret Zablocki, Xiaozhang Zheng.
| Application Number | 20190201384 16/309980 |
| Document ID | / |
| Family ID | 59276843 |
| Filed Date | 2019-07-04 |

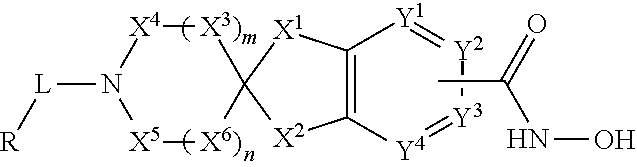










View All Diagrams
| United States Patent Application | 20190201384 |
| Kind Code | A1 |
| Zheng; Xiaozhang ; et al. | July 4, 2019 |
2-SPIRO-5- AND 6-HYDROXAMIC ACID INDANES AS HDAC INHIBITORS
Abstract
The present invention is directed to inhibitors of histone deacetylases (HDACs) such as HDAC6, and their use in the treatment of diseases such as cell proliferative diseases (e.g., cancer), neurological (e.g., neurodegenerative disease or neurodevelopmental disease), inflammatory or autoimmune disease, infection, metabolic disease, hematologic disease, or cardiovascular disease.
| Inventors: | Zheng; Xiaozhang; (Lexington, MA) ; Ng; Pui Yee; (Waltham, MA) ; Zablocki; Mary-Margaret; (Revere, MA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 59276843 | ||||||||||
| Appl. No.: | 16/309980 | ||||||||||
| Filed: | June 16, 2017 | ||||||||||
| PCT Filed: | June 16, 2017 | ||||||||||
| PCT NO: | PCT/US2017/037970 | ||||||||||
| 371 Date: | December 14, 2018 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 62351399 | Jun 17, 2016 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C07D 209/54 20130101; A61P 25/00 20180101; C07D 221/20 20130101; A61P 35/00 20180101; A61P 29/00 20180101; A61P 25/14 20180101; A61P 25/28 20180101; A61K 31/407 20130101; C07D 401/04 20130101; C07D 401/06 20130101; A61K 31/438 20130101 |
| International Class: | A61K 31/438 20060101 A61K031/438 |
Claims
1. A compound of the Formula I: ##STR00044## or a pharmaceutically acceptable salt thereof, wherein: X.sup.1, X.sup.2, X.sup.3, X.sup.4, X.sup.5, and X.sup.6 are each independently, at each occurrence, --CR.sup.1R.sup.2-- or --C(O)--; Y.sup.1, Y.sup.2, Y.sup.3 and Y.sup.4 are each independently, at each occurrence, N or CR.sup.1, wherein --C(O)NHOH is attached at Y.sup.2 or Y.sup.3, and Y.sup.2 or Y.sup.3 is a carbon atom when attached to --C(O)NHOH; L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--; R is --H, --C.sub.1-C.sub.6alkyl, --C.sub.2-C.sub.6alkenyl, --C.sub.4-C.sub.8cycloalkenyl, --C.sub.2-C.sub.6alkynyl, --C.sub.3-C.sub.8cycloalkyl, --C.sub.5-C.sub.12spirocycloalkyl, heterocyclyl, spiroheterocyclyl, aryl, or heteroaryl containing 1-5 heteroatoms selected from the group consisting of N, S, P, or O, wherein each alkyl, alkenyl, cycloalkenyl, alkynyl, cycloalkyl, spirocycloalkyl, heterocyclyl, spiroheterocyclyl, aryl, or heteroaryl is optionally substituted with one or more --OH, halogen, oxo, --NO.sub.2, --CN, --R.sup.1, --R.sup.2, --SR.sup.3, --OR.sup.3, --NHR.sup.3, --NR.sup.3R.sup.4, --S(O).sub.2NR.sup.3R.sup.4, --S(O).sub.2R.sup.1, --C(O)R.sup.1, --CO.sub.2R, --NR.sup.3S(O).sub.2R, --S(O)R.sup.1, --S(O)NR.sup.3R.sup.4, --NR.sup.3S(O)R.sup.1, heterocyclyl, aryl, or heteroaryl; R.sup.1 and R.sup.2 are independently, at each occurrence, --H, --R.sup.3, --R.sup.4, --C.sub.1-C.sub.6alkyl, --C.sub.2-C.sub.6alkenyl, --C.sub.4-C.sub.8cycloalkenyl, --C.sub.2-C.sub.6alkynyl, --C.sub.3-C.sub.8cycloalkyl, heterocyclyl, aryl, heteroaryl containing 1-5 heteroatoms selected from the group consisting of N, S, P and O, --OH, halogen, --NO.sub.2, --CN, --NHC.sub.1-C.sub.6alkyl, --N(C.sub.1-C.sub.6alkyl).sub.2, --S(O).sub.2N(C.sub.1-C.sub.6alkyl).sub.2, --N(C.sub.1-C.sub.6alkyl)S(O).sub.2R, --S(O).sub.2(C.sub.1-C.sub.6alkyl), --(C.sub.1-C.sub.6alkyl)S(O).sub.2R.sup.5, --C(O)C.sub.1-C.sub.6alkyl, --CO.sub.2C.sub.1-C.sub.6alkyl, --N(C.sub.1-C.sub.6alkyl)S(O).sub.2C.sub.1-C.sub.6alkyl, or --(CHR.sup.5).sub.pNR.sup.3R.sup.4, wherein each alkyl, alkenyl, cycloalkenyl, alkynyl, cycloalkyl, heterocyclyl, aryl, or heteroaryl is optionally substituted with one or more --OH, halogen, --NO.sub.2, oxo, --CN, --R.sup.5, --OR.sup.3, --NHR.sup.3, --NR.sup.3R.sup.4, --S(O).sub.2N(R.sup.3).sub.2, --S(O).sub.2R.sup.5, --C(O)R.sup.5, --CO.sub.2R.sup.5, --NR.sup.3S(O).sub.2R.sup.5, --S(O)R.sup.5, --S(O)NR.sup.3R.sup.4, --NR.sup.3S(O)R.sup.5, heterocyclyl, aryl, or heteroaryl; or R.sup.1 and R.sup.2, when on the same atom, can combine with the carbon atom to which they are attached to form a cycloalkyl, heterocycle, spirocycloalkyl, spiroheterocycle, or spirocycloalkenyl; or R.sup.1 and R.sup.2, when on adjacent or non-adjacent atoms, can combine to form a heterocycle, cycloalkyl, aryl, heteroaryl containing 1-5 heteroatoms selected from the group consisting of N, S, P and O, or cycloalkenyl; R.sup.3 and R.sup.4 are independently, at each occurrence, --H, --C.sub.1-C.sub.6alkyl, --C.sub.2-C.sub.6alkenyl, --C.sub.4-C.sub.8cycloalkenyl, --C.sub.2-C.sub.6alkynyl, --C.sub.3-C.sub.8cycloalkyl, heterocyclyl, aryl, heteroaryl containing 1-5 heteroatoms selected from N, S, P, and O, --S(O).sub.2N(C.sub.1-C.sub.6alkyl).sub.2, --S(O).sub.2(C.sub.1-C.sub.6alkyl), --(C.sub.1-C.sub.6alkyl)S(O).sub.2R.sup.5, --C(O)C.sub.1-C.sub.6alkyl, --CO.sub.2C.sub.1-C.sub.6alkyl, or --(CHR.sup.5).sub.pN(C.sub.1-C.sub.6alkyl).sub.2, wherein each alkyl, alkenyl, cycloalkenyl, alkynyl, cycloalkyl, heterocyclyl, aryl, and heteroaryl is optionally substituted with one or more substituents selected from --OH, halogen, --NO.sub.2, oxo, --CN, --R.sup.5, --O(C.sub.1-C.sub.6)alkyl, --NH(C.sub.1-C.sub.6)alkyl, --N(C.sub.1-C.sub.6alkyl).sub.2, --S(O).sub.2N(C.sub.1-C.sub.6alkyl).sub.2, --S(O).sub.2NHC.sub.1-C.sub.6alkyl, --C(O)C.sub.1-C.sub.6alkyl, --CO.sub.2C.sub.1-C.sub.6alkyl, --N(C.sub.1-C.sub.6alkyl)S(O).sub.2C.sub.1-C.sub.6alkyl, --S(O)R.sup.5, --S(O)N(C.sub.1-C.sub.6alkyl).sub.2, --N(C.sub.1-C.sub.6alkyl)S(O)R.sup.5, heterocyclyl, aryl, or heteroaryl; R.sup.5 is independently, at each occurrence, --H, --C.sub.1-C.sub.6alkyl, --C.sub.2-C.sub.6alkenyl, --C.sub.4-C.sub.8cycloalkenyl, --C.sub.2-C.sub.6alkynyl, --C.sub.3-C.sub.8cycloalkyl, heterocyclyl, aryl, heteroaryl containing 1-5 heteroatoms selected from N, S, P and O, --OH, halogen, --NO.sub.2, --CN, --NHC.sub.1-C.sub.6alkyl, --N(C.sub.1-C.sub.6alkyl).sub.2, --S(O).sub.2NH(C.sub.1-C.sub.6alkyl), --S(O).sub.2N(C.sub.1-C.sub.6alkyl).sub.2, --S(O).sub.2C.sub.1-C.sub.6alkyl, --C(O)C.sub.1-C.sub.6alkyl, --CO.sub.2C.sub.1-C.sub.6alkyl, --N(C.sub.1-C.sub.6alkyl)SO.sub.2C.sub.1-C.sub.6alkyl, --S(O)(C.sub.1-C.sub.6alkyl), --S(O)N(C.sub.1-C.sub.6alkyl).sub.2, --N(C.sub.1-C.sub.6alkyl)S(O)(C.sub.1-C.sub.6alkyl), or --(CH.sub.2).sub.pN(C.sub.1-C.sub.6alkyl).sub.2; p is 0, 1, 2, 3, 4, 5, or 6; n is 0, 1, 2, 3, or 4; m is 0, 1, or 2; and wherein the sum m+n.ltoreq.4.
2-31. (canceled)
32. The compound of claim 1, wherein n is 0.
33. The compound of claim 32, wherein m is 1.
34. The compound of claim 32, wherein m is 2.
35. The compound of claim 1, wherein n is 1.
36. The compound of claim 35, wherein m is 1.
37. The compound of claim 1, wherein X.sup.1 is --CH.sub.2-- and X.sup.2 is --CH.sub.2--.
38. The compound of claim 1, wherein X.sup.5 is --C(O)--.
39. The compound of claim 37, wherein Y.sup.1 and Y.sup.4 are each CR.sup.1, wherein --C(O)NHOH is attached at Y.sup.2 or Y.sup.3, and Y.sup.2 or Y.sup.3 is a carbon atom when attached to --C(O)NHOH.
40. The compound of claim 39, wherein L is a bond, --(CR.sup.1R.sup.2).sub.p--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
41. The compound of claim 40, wherein p is 0 or 1.
42. The compound of claim 41, wherein R is --H or an optionally substituted group selected from --C.sub.1-C.sub.6alkyl, aryl, and heteroaryl.
43. The compound of claim 42, wherein R is an optionally substituted group selected from aryl or heteroaryl.
44. The compound of claim 1, wherein the compound is of the Formula IA-1: ##STR00045##
45. The compound of claim 1, wherein the compound is of the Formula IA-2: ##STR00046##
46. The compound of claim 1, wherein the compound is of the Formula IA-3: ##STR00047##
47. The compound of claim 1, wherein the compound is of the Formula IA-4, Formula IA-5, Formula IA-6, Formula IA-7, or Formula IA-8: ##STR00048##
48. The compound of claim 1, wherein the compound is of the Formula IB-1, Formula IB-2, Formula IB-3, Formula IB-4, Formula IB-5, Formula IB-6, Formula IB-7, or Formula IB-8: ##STR00049##
49. A compound of claim 1 selected from: (R)--N-hydroxy-1'-methyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]- -5-carboxamide (I-1); (S)--N-hydroxy-1'-methyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]- -5-carboxamide (I-2); (R)--N-hydroxy-1'-isopropyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidi- ne]-5-carboxamide (I-3); (S)--N-hydroxy-1'-isopropyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidi- ne]-5-carboxamide (I-4); (R)-1'-ethyl-N-hydroxy-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5- -carboxamide (I-5); (S)-1'-ethyl-N-hydroxy-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5- -carboxamide (I-6); (R)--N-hydroxy-1'-isobutyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidin- e]-5-carboxamide (I-7); (S)--N-hydroxy-1'-isobutyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidin- e]-5-carboxamide (I-8); (R)--N-hydroxy-1'-(2-methylbenzyl)-2'-oxo-1,3-dihydrospiro[indene-2,3'-py- rrolidine]-5-carboxamide (I-9); (S)--N-hydroxy-1'-(2-methylbenzyl)-2'-oxo-1,3-dihydrospiro[indene-2,3'-py- rrolidine]-5-carboxamide (I-10); (R)-1'-benzyl-N-hydroxy-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-- 5-carboxamide (I-11); (S)-1'-benzyl-N-hydroxy-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-- 5-carboxamide (I-12); (R)--N-hydroxy-2'-oxo-1'-phenyl-1,3-dihydrospiro[indene-2,3'-pyrrolidine]- -5-carboxamide (I-13); (S)--N-hydroxy-2'-oxo-1'-phenyl-1,3-dihydrospiro[indene-2,3'-pyrrolidine]- -5-carboxamide (I-14); (R)-1'-(4-fluorophenyl)-N-hydroxy-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyr- rolidine]-5-carboxamide (I-15); (S)-1'-(4-fluorophenyl)-N-hydroxy-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyr- rolidine]-5-carboxamide (I-16); (R)--N-hydroxy-2'-oxo-1'-(pyridin-3-yl)-1,3-dihydrospiro[indene-2,3'-pyrr- olidine]-5-carboxamide (I-17); (S)--N-hydroxy-2'-oxo-1'-(pyridin-3-yl)-1,3-dihydrospiro[indene-2,3'-pyrr- olidine]-5-carboxamide (I-18); (R)--N-hydroxy-2'-oxo-1'-(pyridin-2-yl)-1,3-dihydrospiro[indene-2,3'-pyrr- olidine]-5-carboxamide (I-19); (S)--N-hydroxy-2'-oxo-1'-(pyridin-2-yl)-1,3-dihydrospiro[indene-2,3'-pyrr- olidine]-5-carboxamide (I-20); (R)--N-hydroxy-2'-oxo-1'-(pyridin-4-yl)-1,3-dihydrospiro[indene-2,3'-pyrr- olidine]-5-carboxamide (I-21); (S)--N-hydroxy-2'-oxo-1'-(pyridin-4-yl)-1,3-dihydrospiro[indene-2,3'-pyrr- olidine]-5-carboxamide (I-22); (R)--N-hydroxy-2'-oxo-1'-(pyridin-3-ylmethyl)-1,3-dihydrospiro[indene-2,3- '-pyrrolidine]-5-carboxamide (I-23); (R)--N-hydroxy-1'-methyl-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carb- oxamide (I-24); (S)--N-hydroxy-1'-methyl-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carb- oxamide (I-25); (R)-1'-benzyl-N-hydroxy-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carbo- xamide (I-26); (S)-1'-benzyl-N-hydroxy-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carbo- xamide (I-27); (R)-1'-acetyl-N-hydroxy-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carbo- xamide (I-28); (S)-1'-acetyl-N-hydroxy-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carbo- xamide (I-29); (R)-1'-benzoyl-N-hydroxy-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carb- oxamide (I-30); (S)-1'-benzoyl-N-hydroxy-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carb- oxamide (I-31); (R)--N-hydroxy-1'-methyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-- 5-carboxamide (I-32); (S)--N-hydroxy-1'-methyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-- 5-carboxamide (I-33); (R)-1'-benzyl-N-hydroxy-2'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-5- -carboxamide (I-34); (S)-1'-benzyl-N-hydroxy-2'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-5- -carboxamide (I-35); (R)--N-hydroxy-2'-oxo-1'-phenyl-1,3-dihydrospiro[indene-2,3'-piperidine]-- 5-carboxamide (I-36); (S)--N-hydroxy-2'-oxo-1'-phenyl-1,3-dihydrospiro[indene-2,3'-piperidine]-- 5-carboxamide (I-37); N-hydroxy-1'-methyl-5'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-ca- rboxamide (I-38); N-hydroxy-1'-methyl-6'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-5-car- boxamide (I-39); N-hydroxy-1'-methyl-2'-oxo-1,3-dihydrospiro[indene-2,4'-piperidine]-5-car- boxamide (I-40); N-hydroxy-1'-methyl-1,3-dihydrospiro[indene-2,3'-piperidine]-5-carboxamid- e (I-41); and N-hydroxy-1'-methyl-1,3-dihydrospiro[indene-2,4'-piperidine]-5-carboxamid- e (I-42).
50. A pharmaceutical composition comprising a compound of claim 1 and a pharmaceutically acceptable carrier.
Description
RELATED APPLICATIONS
[0001] This application claims the benefit of and priority to U.S. provisional application No. 62/351,399, filed Jun. 17, 2016, the entire contents of which are incorporated herein by reference in its entirety.
FIELD OF THE INVENTION
[0002] The present invention relates to inhibitors of zinc-dependent histone deacetylases (HDACs) useful in the treatment of diseases or disorders associated with HDACs including cell proliferation diseases (e.g., cancer), neurological and inflammatory diseases. Specifically, this invention is concerned with compounds and compositions inhibiting HDACs, methods of treating diseases associated with HDACs, and methods of synthesizing these compounds.
BACKGROUND OF THE INVENTION
[0003] Many members of the HDAC family require zinc (Zn) to function properly. For instance, the isozyme histone deacetylase 6 (HDAC6) is a zinc-dependent histone deacetylase that possesses histone deacetylase activity. Other family members include HDACs 1-5 and 7-11. (De Ruijter et al., Biochem. J. 2003. 370; 737-749).
[0004] HDAC6 is known to deacetylate and associate with .alpha.-tubulin, cortactin, heat shock protein 90, -catenin, glucose-regulated protein 78 kDa, myosin heavy chain 9, heat shock cognate protein 70, and dnaJ homolog subfamily A member 1 (reviewed in Li et al., EBS J. 2013, 280: 775-93; Zhang et al., Protein Cell. 2015, 6(1): 42-54). Diseases in which HDAC6 inhibition could have a potential benefit include cancer (reviewed in Aldana-Masangkay et al., J. Biomed. Biotechnol. 2011, 875824), specifically: multiple myeloma (Hideshima et al., Proc. Natl. Acad. Sci. USA 2005, 102(24):8567-8572); lung cancer (Kamemura et al., Biochem. Biophys. Res. Commun. 2008, 374(1); 84-89); ovarian cancer (Bazzaro et al., Clin. Cancer Res. 2008, 14(22):7340-7347); breast cancer (Lee et al., Cancer Res. 2008, 68(18):7561-7569; Park et al., Oncol. Rep. 2011, 25: 1677-81; Rey et al., Eur. J. Cell Biol. 2011, 90: 128-35); prostate cancer (Seidel et al., Biochem. Pharmacol. 2015 (15)00714-5); pancreatic cancer (Nawrocki et al., Cancer Res. 2006, 66(7):3773-3781); renal cancer (Cha et al., Clin. Cancer Res. 2009, 15(3): 840-850); hepatocellular cancer (Ding et al., FEBS Lett. 2013, 587:880-6; Kanno et al., Oncol. Rep. 2012, 28: 867-73); lymphomas (Ding et al., Cancer Cell Int. 2014, 14:139; Amengual et al., Clin Cancer Res. 2015, 21(20):4663-75); and leukemias such as acute myeloid leukemia (AML) (Fiskus et al., Blood 2008, 112(7); 2896-2905) and acute lymphoblastic leukemia (ALL) (Rodriguez-Gonzalez et al., Blood 2008, 1 12(11): Abstract 1923)).
[0005] Inhibition of HDAC6 may also have a role in cardiovascular disease, including pressure overload, chronic ischemia, and infarction-reperfusion injury (Tannous et al., Circulation 2008, 1 17(24):3070-3078); bacterial infection, including those caused by uropathogenic Escherichia coli (Dhakal and Mulve, J. Biol. Chem. 2008, 284(1):446-454); neurological diseases caused by accumulation of intracellular protein aggregates such as Alzheimer's, Parkinson's and Huntington's disease (reviewed in Simoes-Pires et al., Mol. Neurodegener. 2013, 8: 7) or central nervous system trauma caused by tissue injury, oxidative-stress induced neuronal or axomal degeneration (Rivieccio et al., Proc. Natl. Acad. Sci. USA 2009, 106(46):19599-195604); and inflammation and autoimmune diseases through enhanced T cell-mediated immune tolerance at least in part through effects on regulatory T cells, including rheumatoid arthritis, psoriasis, spondylitis arthritis, psoriatic arthritis, multiple sclerosis, lupus, colitis and graft versus host disease (reviewed in Wang et al., Nat. Rev. Drug Disc. 2009 8(12):969-981; Vishwakarma et al., Int. Immunopharmacol. 2013, 16:72-8; Kalin et al., J. Med. Chem. 2012, 55:639-51); and fibrotic disease, including kidney fibrosis (Choi et al., Vascul. Pharmacol. 2015 72:130-140).
[0006] Four HDAC inhibitors are currently approved for the treatment of some cancers. These are suberanilohydroxamic acid (Vorinostat; Zolinza.RTM.) for the treatment of cutaneous T cell lymphoma and multiple myeloma; Romidepsin (FK228; FR901228; Istodax.RTM.) for the treatment of peripheral T cell lymphoma; Panobinostat (LBH-589; Farydak.RTM.) for the treatment of multiple myeloma; and belinostat (PXD101; Beleodaq.RTM.) for the treatment of peripheral T cell lymphoma. However, these drugs are of limited effectiveness and can give rise to unwanted side effects. Thus there is a need for HDAC inhibitors with an improved safety-efficacy profile.
SUMMARY OF THE INVENTION
[0007] One aspect of the invention relates to compounds of Formula I:
##STR00001##
[0008] and pharmaceutically acceptable salts, prodrugs, solvates, hydrates, tautomers, or isomers thereof, wherein: [0009] X.sup.1, X.sup.2, X.sup.3, X.sup.4, X.sup.5, and X.sup.6 are each independently, at each occurrence, --CR.sup.1R.sup.2--, --NR.sup.3--, --O--, --C(O)--, --S(O).sub.2--, --S(O)--, or --S--; [0010] Y.sup.1, Y.sup.2, Y.sup.3 and Y.sup.4 are each independently, at each occurrence, N or CR.sup.1, wherein the hydroxamic acid is attached at Y.sup.2 or Y.sup.3 and Y.sup.2 or Y.sup.3 is a carbon atom when attached to the hydroxamic acid; [0011] L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--; [0012] R is independently --H, --C.sub.1-C.sub.6alkyl, --C.sub.2-C.sub.6alkenyl, --C.sub.4-C.sub.5cycloalkenyl, --C.sub.2-C.sub.6alkynyl, --C.sub.3-C.sub.8cycloalkyl, --C.sub.5-C.sub.12spirocycloalkyl, heterocyclyl, spiroheterocyclyl, aryl, or heteroaryl containing 1-5 heteroatoms selected from the group consisting of N, S, P, or O, wherein each alkyl, alkenyl, cycloalkenyl, alkynyl, cycloalkyl, spirocycloalkyl, heterocyclyl, spiroheterocyclyl, aryl, or heteroaryl is optionally substituted with one or more --OH, halogen, oxo, --NO.sub.2, --CN, --R.sup.1, --R.sup.2, --SR.sup.3, --OR.sup.3, --NHR.sup.3, --NR.sup.3R.sup.4, --S(O).sub.2NR.sup.3R.sup.4, --S(O).sub.2R.sup.1, --C(O)R.sup.1, --CO.sub.2R.sup.1, --NR.sup.3S(O).sub.2R.sup.1, --S(O)R.sup.1, --S(O)NR.sup.3R.sup.4, --NR.sup.3S(O)R.sup.1, heterocyclyl, aryl, or heteroaryl; [0013] R.sup.1 and R.sup.2 are independently, at each occurrence, --H, --R.sup.3, --R.sup.4, --C.sub.1-C.sub.6alkyl, --C.sub.2-C.sub.6alkenyl, --C.sub.4-C.sub.5cycloalkenyl, --C.sub.2-C.sub.6alkynyl. --C.sub.3-C.sub.8cycloalkyl, heterocyclyl, aryl, heteroaryl containing 1-5 heteroatoms selected from the group consisting of N, S, P and O, --OH, halogen, --NO.sub.2, --CN, --NHC.sub.1-C.sub.6alkyl, --N(C.sub.1-C.sub.6alkyl).sub.2, --S(O).sub.2N(C.sub.1-C.sub.6alkyl).sub.2, --N(C.sub.1-C.sub.6alkyl)S(O).sub.2R.sup.5, --S(O).sub.2(C.sub.1-C.sub.6alkyl), --(C.sub.1-C.sub.6alkyl)S(O).sub.2R.sup.5, --C(O)C.sub.1-C.sub.6alkyl, --CO.sub.2C.sub.1-C.sub.6alkyl, --N(C.sub.1-C.sub.6alkyl)S(O).sub.2C.sub.1-C.sub.6alkyl, or --(CHR.sup.5).sub.pNR.sup.3R.sup.4, wherein each alkyl, alkenyl, cycloalkenyl, alkynyl, cycloalkyl, heterocyclyl, aryl, or heteroaryl is optionally substituted with one or more --OH, halogen, --NO.sub.2, oxo, --CN, --R.sup.5, --OR.sup.3, --NHR.sup.3, --NR.sup.3R.sup.4, --S(O).sub.2N(R.sup.3).sub.2--, --S(O).sub.2R.sup.5, --C(O)R.sup.5, --CO.sub.2R.sup.5, --NR.sup.3S(O).sub.2R.sup.5, --S(O)R.sup.5, --S(O)NR.sup.3R.sup.4, --NR.sup.3S(O)R.sup.5, heterocyclyl, aryl, or heteroaryl; [0014] or R.sup.1 and R.sup.2 can combine with the carbon atom to which they are both attached to form a cycloalkyl, heterocycle, spirocycloalkyl, spiroheterocycle, or spirocycloalkenyl; [0015] or R.sup.1 and R.sup.2, when on adjacent or non-adjacent atoms, can combine to form a heterocycle, cycloalkyl, aryl, heteroaryl containing 1-5 heteroatoms selected from the group consisting of N, S, P and O, or cycloalkenyl; [0016] R.sup.3 and R.sup.4 are independently, at each occurrence, --H, --C.sub.1-C.sub.6alkyl, --C.sub.2-C.sub.6alkenyl, --C.sub.4-C.sub.8cycloalkenyl, --C.sub.2-C.sub.6alkynyl, --C.sub.3-C.sub.8cycloalkyl, heterocyclyl, aryl, heteroaryl containing 1-5 heteroatoms selected from N, S, P, and O, --S(O).sub.2N(C.sub.1-C.sub.6alkyl).sub.2, --S(O).sub.2(C.sub.1-C.sub.6alkyl), --(C.sub.1-C.sub.6alkyl)S(O).sub.2R.sup.5, --C(O)C.sub.1-C.sub.6alkyl, --CO.sub.2C.sub.1-C.sub.6alkyl, or --(CHR.sup.5).sub.pN(C.sub.1-C.sub.6alkyl).sub.2, wherein each alkyl, alkenyl, cycloalkenyl, alkynyl, cycloalkyl, heterocyclyl, aryl, and heteroaryl is optionally substituted with one or more substituents selected from --OH, halogen, --NO.sub.2, oxo, --CN, --R.sup.5, --O(C.sub.1-C.sub.6)alkyl, --NH(C.sub.1-C.sub.6)alkyl, --N(C.sub.1-C.sub.6alkyl).sub.2, --S(O).sub.2N(C.sub.1-C.sub.6alkyl).sub.2, --S(O).sub.2NHC.sub.1-C.sub.6alkyl, --C(O)C.sub.1-C.sub.6alkyl, --CO.sub.2C.sub.1-C.sub.6alkyl, --N(C.sub.1-C.sub.6alkyl)S(O).sub.2C.sub.1-C.sub.6alkyl, --S(O)R.sup.5, --S(O)N(C.sub.1-C.sub.6alkyl).sub.2, --N(C.sub.1-C.sub.6alkyl)S(O)R.sup.5, heterocyclyl, aryl, or heteroaryl; [0017] R.sup.5 is independently, at each occurrence, --H, --C.sub.1-C.sub.6alkyl, --C.sub.2-C.sub.6alkenyl, --C.sub.4-C.sub.8cycloalkenyl, --C.sub.2-C.sub.6alkynyl, --C.sub.3-C.sub.8cycloalkyl, heterocyclyl, aryl, heteroaryl containing 1-5 heteroatoms selected from N, S, P and O, --OH, halogen, --NO.sub.2, --CN, --NHC.sub.1-C.sub.6alkyl, --N(C.sub.1-C.sub.6alkyl).sub.2, --S(O).sub.2NH(C.sub.1-C.sub.6alkyl), --S(O).sub.2N(C.sub.1-C.sub.6alkyl).sub.2, --S(O).sub.2C.sub.1-C.sub.6alkyl, --C(O)C.sub.1-C.sub.6alkyl, --CO.sub.2C.sub.1-C.sub.6alkyl, --N(C.sub.1-C.sub.6alkyl)SO.sub.2C.sub.1-C.sub.6alkyl, --S(O)(C.sub.1-C.sub.6alkyl), --S(O)N(C.sub.1-C.sub.6alkyl).sub.2, --N(C.sub.1-C.sub.6alkyl)S(O)(C.sub.1-C.sub.6alkyl) or --(CH.sub.2).sub.pN(C.sub.1-C.sub.6alkyl).sub.2; [0018] p is 0, 1, 2, 3, 4, 5, or 6; [0019] n is 0, 1, 2, 3, or 4; [0020] m is 0, 1, or 2; and [0021] wherein the sum m+n.ltoreq.4.
[0022] Another aspect of the invention relates to a method of treating a disease or disorder associated with HDAC6 modulation in a subject in need thereof, comprising administering to the subject an effective amount of a compound of Formula I.
[0023] Another aspect of the invention is directed to a method of inhibiting a histone deacetylase (e.g., a zinc-dependent histone deacetylase such as HDAC6). The method involves administering to a patient in need thereof an effective amount of a compound of Formula I. In some embodiments, the method of treating a disease or disorder associated with HDAC6 modulation in a subject includes inhibiting a histone deacetylase (e.g., a zinc-dependent histone deacetylase such as HDAC6) in the subject.
[0024] Another aspect of the invention is directed to pharmaceutical compositions comprising a compound of Formula I and a pharmaceutically acceptable carrier. The pharmaceutically acceptable carrier can further include an excipient, diluent, or surfactant. The pharmaceutical composition can be effective for treating a disease or disorder associated with HDAC6 modulation in a subject in need thereof. The pharmaceutical compositions can comprise the compounds of the present invention for use in treating diseases described herein. The compositions can contain at least one compound of the invention and a pharmaceutically acceptable carrier.
[0025] Another aspect of the present disclosure relates to a compound of Formula I, or a pharmaceutically acceptable salt, hydrate, solvate, prodrug, stereoisomer, or tautomer thereof, for use in treating or preventing a disease associated with HDAC6 modulation. The invention also provides the use of the compounds described herein in the manufacture of a medicament for the treatment of a disease associated with HDACs.
[0026] The present invention also provides methods for the treatment of human diseases or disorders including, without limitation, oncological, neurological, inflammatory, autoimmune, infectious, metabolic, hematologic, or cardiovascular diseases or disorders.
[0027] The present invention also provides compounds that are useful in inhibiting of zinc-dependent HDAC enzymes, for instance HDAC6. These compounds can also be useful in the treatment of diseases including cancer.
[0028] The present invention further provides compounds that can inhibit HDAC6. In some embodiments, the efficacy-safety profile of the compounds of the current invention can be improved relative to other known HDAC (e.g. HDAC6) inhibitors. Additionally, the present technology also has the advantage of being able to be used for a number of different types of diseases, including cancer and non-cancer indications. Additional features and advantages of the present technology will be apparent to one of skill in the art upon reading the Detailed Description of the Invention, below.
DETAILED DESCRIPTION OF THE INVENTION
[0029] HDAC6 is a zinc-dependent histone deacetylase that has two catalytic domains. HDAC6 can interact with and deacetylate non-histone proteins, including HSP90 and .alpha.-tubulin. Acetylation of HSP90 is associated with loss of function of HSP90. HDAC6 is also implicated in the degradation of misfolded proteins as part of the aggresome. Accordingly, inhibition of HDAC6 can have downstream effects that can play a role in the development of certain diseases such as cancer. The present invention provides inhibitors of HDAC6 and methods for using the same to treat disease.
[0030] In a first aspect of the invention, compounds of Formula I are described:
##STR00002##
and pharmaceutically acceptable salts, prodrugs, solvates, hydrates, tautomers, and isomers thereof, wherein Y.sup.1, Y.sup.2, Y.sup.3, Y.sup.4, X.sup.1, X.sup.2, X.sup.3, X.sup.4, X.sup.5, X.sup.6, L, R, m and n are described as above.
[0031] The details of the invention are set forth in the accompanying description below. Although methods and materials similar or equivalent to those described herein can be used in the practice or testing of the present invention, illustrative methods and materials are now described. Other features, objects, and advantages of the invention will be apparent from the description and from the claims. In the specification and the appended claims, the singular forms also include the plural unless the context clearly dictates otherwise. Unless defined otherwise, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which this invention belongs. All patents and publications cited in this specification are incorporated herein by reference in their entireties.
Definitions
[0032] The articles "a" and "an" are used in this disclosure to refer to one or more than one (i.e., to at least one) of the grammatical object of the article. By way of example, "an element" means one element or more than one element.
[0033] The term "and/or" is used in this disclosure to mean either "and" or "or" unless indicated otherwise.
[0034] The term "optionally substituted" is understood to mean that a given chemical moiety (e.g. an alkyl group) can (but is not required to) be bonded other substituents (e.g. heteroatoms). For instance, an alkyl group that is optionally substituted can be a fully saturated alkyl chain (i.e. a pure hydrocarbon). Alternatively, the same optionally substituted alkyl group can have substituents different from hydrogen. For instance, it can, at any point along the chain be bonded to a halogen atom, a hydroxyl group, or any other substituent described herein. Thus the term "optionally substituted" means that a given chemical moiety has the potential to contain other functional groups, but does not necessarily have any further functional groups.
[0035] The term "aryl" refers to cyclic, aromatic hydrocarbon groups that have 1 to 2 aromatic rings, including monocyclic or bicyclic groups such as phenyl, biphenyl or naphthyl. Where containing two aromatic rings (bicyclic, etc.), the aromatic rings of the aryl group may be joined at a single point (e.g., biphenyl), or fused (e.g., naphthyl). The aryl group may be optionally substituted by one or more substituents, e.g., 1 to 5 substituents, at any point of attachment. Exemplary substituents include, but are not limited to, --H, -halogen, --O--C.sub.1-C.sub.6alkyl, --C.sub.1-C.sub.6alkyl, --OC.sub.2-C.sub.6alkenyl, --OC.sub.2-C.sub.6alkynyl, --C.sub.2-C.sub.6alkenyl, --C.sub.2-C.sub.6alkynyl, --OH, --OP(O)(OH).sub.2, --OC(O)C.sub.1-C.sub.6alkyl, --C(O)C.sub.1-C.sub.6alkyl, --OC(O)OC.sub.1-C.sub.6alkyl, --NH.sub.2, --NH(C.sub.1-C.sub.6alkyl), --N(C.sub.1-C.sub.6alkyl).sub.2, --S(O).sub.2--C.sub.1-C.sub.6alkyl, --S(O)NHC.sub.1-C.sub.6alkyl, and --S(O)N(C.sub.1-C.sub.6alkyl).sub.2. The substituents can themselves be optionally substituted. Furthermore when containing two fused rings the aryl groups herein defined may have an unsaturated or partially saturated ring fused with a fully saturated ring. Exemplary ring systems of these aryl groups include indanyl, indenyl, tetrahydronaphthalenyl, and tetrahydrobenzoannulenyl.
[0036] Unless otherwise specifically defined, "heteroaryl" means a monovalent monocyclic aromatic radical or a polycyclic aromatic radical of 5 to 24 ring atoms, containing one or more ring heteroatoms selected from N, S, P, and O, the remaining ring atoms being C. Heteroaryl as herein defined also means a bicyclic heteroaromatic group wherein the heteroatom is selected from N, S, P, and O. The aromatic radical is optionally substituted independently with one or more substituents described herein. Examples include, but are not limited to, furyl, thienyl, pyrrolyl, pyridyl, pyrazolyl, pyrimidinyl, imidazolyl, isoxazolyl, oxazolyl, oxadiazolyl, pyrazinyl, indolyl, thiophen-2-yl, quinolyl, benzopyranyl, isothiazolyl, thiazolyl, thiadiazole, indazole, benzimidazolyl, thieno[3,2-b]thiophene, triazolyl, triazinyl, imidazo[1,2-b]pyrazolyl, furo[2,3-c]pyridinyl, imidazo[1,2-a]pyridinyl, indazolyl, pyrrolo[2,3-c]pyridinyl, pyrrolo[3,2-c]pyridinyl, pyrazolo[3,4-c]pyridinyl, thieno[3,2-c]pyridinyl, thieno[2,3-c]pyridinyl, thieno[2,3-b]pyridinyl, benzothiazolyl, indolyl, indolinyl, indolinonyl, dihydrobenzothiophenyl, dihydrobenzofuranyl, benzofuran, chromanyl, thiochromanyl, tetrahydroquinolinyl, dihydrobenzothiazine, dihydrobenzoxanyl, quinolinyl, isoquinolinyl, 1,6-naphthyridinyl, benzo[de]isoquinolinyl, pyrido[4,3-b][1,6]naphthyridinyl, thieno[2,3-b]pyrazinyl, quinazolinyl, tetrazolo[1,5-a]pyridinyl, [1,2,4]triazolo[4,3-a]pyridinyl, isoindolyl, pyrrolo[2,3-b]pyridinyl, pyrrolo[3,4-b]pyridinyl, pyrrolo[3,2-b]pyridinyl, imidazo[5,4-b]pyridinyl, pyrrolo[1,2-a]pyrimidinyl, tetrahydro pyrrolo[1,2-a]pyrimidinyl, 3,4-dihydro-2H-1.lamda..sup.2-pyrrolo[2,1-b]pyrimidine, dibenzo[b,d] thiophene, pyridin-2-one, furo[3,2-c]pyridinyl, furo[2,3-c]pyridinyl, 1H-pyrido[3,4-b][1,4] thiazinyl, benzooxazolyl, benzoisoxazolyl, furo[2,3-b]pyridinyl, benzothiophenyl, 1,5-naphthyridinyl, furo[3,2-b]pyridine, [1,2,4]triazolo[1,5-a]pyridinyl, benzo [1,2,3]triazolyl, imidazo[1,2-a]pyrimidinyl, [1,2,4]triazolo[4,3-b]pyridazinyl, benzo[c][1,2,5]thiadiazolyl, benzo[c][1,2,5]oxadiazole, 1,3-dihydro-2H-benzo[d]imidazol-2-one, 3,4-dihydro-2H-pyrazolo [1,5-b][1,2]oxazinyl, 4,5,6,7-tetrahydropyrazolo[1,5-a]pyridinyl, thiazolo[5,4-d]thiazolyl, imidazo[2,1-b][1,3,4]thiadiazolyl, thieno[2,3-b]pyrrolyl, 3H-indolyl, and derivatives thereof. Furthermore when containing two fused rings the heteroaryl groups herein defined may have an unsaturated or partially saturated ring fused with a fully saturated ring. Exemplary ring systems of these heteroaryl groups include indolinyl, indolinonyl, dihydrobenzothiophenyl, dihydrobenzofuran, chromanyl, thiochromanyl, tetrahydroquinolinyl, dihydrobenzothiazine, 3,4-dihydro-1H-isoquinolinyl, 2,3-dihydrobenzofuran, indolinyl, indolyl, and dihydrobenzoxanyl.
[0037] "Alkyl" refers to a straight or branched chain saturated hydrocarbon. C.sub.1-C.sub.6alkyl groups contain 1 to 6 carbon atoms. Examples of a C.sub.1-C.sub.6alkyl group include, but are not limited to, methyl, ethyl, propyl, butyl, pentyl, isopropyl, isobutyl, sec-butyl and tert-butyl, isopentyl and neopentyl.
[0038] The term "alkenyl" means an aliphatic hydrocarbon group containing a carbon-carbon double bond and which may be straight or branched having about 2 to about 6 carbon atoms in the chain. Certain alkenyl groups have 2 to about 4 carbon atoms in the chain. Branched means that one or more lower alkyl groups such as methyl, ethyl, or propyl are attached to a linear alkenyl chain. Exemplary alkenyl groups include ethenyl, propenyl, n-butenyl, and i-butenyl. A C.sub.2-C.sub.6 alkenyl group is an alkenyl group containing between 2 and 6 carbon atoms.
[0039] The term "alkynyl" means an aliphatic hydrocarbon group containing a carbon-carbon triple bond and which may be straight or branched having about 2 to about 6 carbon atoms in the chain. Certain alkynyl groups have 2 to about 4 carbon atoms in the chain. Branched means that one or more lower alkyl groups such as methyl, ethyl, or propyl are attached to a linear alkynyl chain. Exemplary alkynyl groups include ethynyl, propynyl, n-butynyl, 2-butynyl, 3-methylbutynyl, and n-pentynyl. A C.sub.2-C.sub.6 alkynyl group is an alkynyl group containing between 2 and 6 carbon atoms.
[0040] The term "cycloalkyl" means monocyclic or polycyclic saturated carbon rings containing 3-18 carbon atoms. Examples of cycloalkyl groups include, without limitations, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptanyl, cyclooctanyl, norboranyl, norborenyl, bicyclo[2.2.2]octanyl, or bicyclo[2.2.2]octenyl. A C.sub.3-C.sub.8 cycloalkyl is a cycloalkyl group containing between 3 and 8 carbon atoms. A cycloalkyl group can be fused (e.g., decalin) or bridged (e.g., norbornane).
[0041] The term "cycloalkenyl" means monocyclic, non-aromatic unsaturated carbon rings containing 4-18 carbon atoms. Examples of cycloalkenyl groups include, without limitation, cyclopentenyl, cyclohexenyl, cycloheptenyl, cyclooctenyl, and norborenyl. A C.sub.4-C.sub.8 cycloalkenyl is a cycloalkenyl group containing between 4 and 8 carbon atoms.
[0042] The terms "heterocyclyl" or "heterocycloalkyl" or "heterocycle" refer to monocyclic or polycyclic 3 to 24-membered rings containing carbon and heteroatoms taken from oxygen, phosphorous nitrogen, or sulfur and wherein there is not delocalized n electrons (aromaticity) shared among the ring carbon or heteroatoms. Heterocyclyl rings include, but are not limited to, oxetanyl, azetadinyl, tetrahydrofuranyl, pyrrolidinyl, oxazolinyl, oxazolidinyl, thiazolinyl, thiazolidinyl, pyranyl, thiopyranyl, tetrahydropyranyl, dioxalinyl, piperidinyl, morpholinyl, thiomorpholinyl, thiomorpholinyl S-oxide, thiomorpholinyl S-dioxide, piperazinyl, azepinyl, oxepinyl, diazepinyl, tropanyl, and homotropanyl. A heteroycyclyl or heterocycloalkyl ring can also be fused or bridged, e.g., can be a bicyclic ring.
[0043] As used herein, the term "halo" or "halogen" means a fluoro, chloro, bromo, or iodo group.
[0044] The term "carbonyl" refers to a functional group composing a carbon atom double-bonded to an oxygen atom. It can be abbreviated herein as C(O), or as C.dbd.O.
[0045] The term "oxo" refers to an oxygen atom that is double-bonded to another atom. An "oxo" group can be connected to a carbon atom (e.g., to form a carbonyl, as defined above) or can be connected to a heteroatom such as sulfur (e.g., to form a sulfoxide or a sulfone) or phosphorous (e.g., to form a phosphorous ylide).
[0046] "Spirocycle" means carbogenic bicyclic ring systems with both rings connected through a single atom. A "spirocycloalkyl" or "spirocyclic" system is a spirocycle in which both rings are fully carbogenic. The ring can be different in size and nature, or identical in size and nature. Examples include spiropentane, spirohexane, spiroheptane, spirooctane, spirononane, or spirodecane. One or both of the rings in a spirocycle can be fused to another carbocyclic, heterocyclic, aromatic, or heteroaromatic ring. One or more of the carbon atoms in the spirocycle can be substituted with a heteroatom (e.g., O, N, S, or P). A C.sub.5-C.sub.12 spirocycle is a spirocycle containing between 5 and 12 carbon atoms. One or more of the carbon atoms can be substituted with a heteroatom.
[0047] The term "spirocyclic heterocycle" "spiroheterocyclyl" or "spiroheterocycle" is understood to mean a spirocycle wherein at least one of the rings is a heterocycle (e.g., at least one of the rings is furanyl, morpholinyl, or piperadinyl). A spirocyclic heterocycle can contain between 5 and 12 atoms, at least one of which is a heteroatom selected from N, O, S and P. Spiroheterocycloalkyl groups can be for instance, without limitation, azapiroheptanes; azaspirooctanes; azaspirononanes; azaspirodecanes; oxaspiroheptanes; oxaspirooctanes; octaspirononanes; or oxaspirodecanes.
[0048] The disclosure also includes pharmaceutical compositions comprising an effective amount of a disclosed compound and a pharmaceutically acceptable carrier. Representative "pharmaceutically acceptable salts" include, e.g., water-soluble and water-insoluble salts, such as the acetate, amsonate (4,4-diaminostilbene-2,2-disulfonate), benzenesulfonate, benzonate, bicarbonate, bisulfate, bitartrate, borate, bromide, butyrate, calcium, calcium edetate, camsylate, carbonate, chloride, citrate, clavulariate, dihydrochloride, edetate, edisylate, estolate, esylate, fiunarate, gluceptate, gluconate, glutamate, glycollylarsanilate, hexafluorophosphate, hexylresorcinate, hydrabamine, hydrobromide, hydrochloride, hydroxynaphthoate, iodide, sethionate, lactate, lactobionate, laurate, magnesium, malate, maleate, mandelate, mesylate, methylbromide, methylnitrate, methylsulfate, mucate, napsylate, nitrate, N-methylglucamine ammonium salt, 3-hydroxy-2-naphthoate, oleate, oxalate, palmitate, pamoate (1,1-methene-bis-2-hydroxy-3-naphthoate, einbonate), pantothenate, phosphate/diphosphate, picrate, polygalacturonate, propionate, p-toluenesulfonate, salicylate, stearate, subacetate, succinate, sulfate, sulfosalicylate, suramate, tannate, tartrate, teoclate, tosylate, triethiodide, and valerate salts.
[0049] The term "tautomers" refers to a set of compounds that have the same number and type of atoms, but differ in bond connectivity and are in equilibrium with one another. A "tautomer" is a single member of this set of compounds. Typically a single tautomer is drawn but it is understood that this single structure is meant to represent all possible tautomers that might exist. Examples include enol-ketone tautomerism. When a ketone is drawn it is understood that both the enol and ketone forms are part of the invention.
[0050] The term "prodrug," as used in this disclosure, means a compound which is convertible in vivo by metabolic means (e.g., by hydrolysis) to a disclosed compound. Furthermore, as used herein a prodrug is a drug which is inactive in the body, but is transformed in the body typically either during absorption or after absorption from the gastrointestinal tract into the active compound. The conversion of the prodrug into the active compound in the body may be done chemically or biologically (i.e., using an enzyme).
[0051] The term "solvate" refers to a complex of variable stoichiometry formed by a solute and solvent. Such solvents for the purpose of the invention may not interfere with the biological activity of the solute. Examples of suitable solvents include, but are not limited to, water, MeOH, EtOH, and AcOH. Solvates wherein water is the solvent molecule are typically referred to as hydrates. Hydrates include compositions containing stoichiometric amounts of water, as well as compositions containing variable amounts of water.
[0052] The term "isomer" refers to compounds that have the same composition and molecular weight but differ in physical and/or chemical properties. The structural difference may be in constitution (geometric isomers) or in the ability to rotate the plane of polarized light (stereoisomers). With regard to stereoisomers, the compounds of Formula I may have one or more asymmetric carbon atom and may occur as racemates, racemic mixtures and as individual enantiomers or diastereomers.
[0053] The term "stereoisomers" refers to the set of compounds which have the same number and type of atoms and share the same bond connectivity between those atoms, but differ in three dimensional structure. The term "stereoisomer" refers to any member of this set of compounds. For instance, a stereoisomer may be an enantiomer or a diastereomer.
[0054] The term "enantiomers" refers to a pair of stereoisomers which are non-superimposable mirror images of one another. The term "enantiomer" refers to a single member of this pair of stereoisomers. The term "racemic" refers to a 1:1 mixture of a pair of enantiomers.
[0055] The term "diastereomers" refers to the set of stereoisomers which cannot be made superimposable by rotation around single bonds. For example, cis- and trans-double bonds, endo- and exo-substitution on bicyclic ring systems, and compounds containing multiple stereogenic centers with different relative configurations are considered to be diastereomers. The term "diastereomer" refers to any member of this set of compounds. In some examples presented, the synthetic route may produce a single diastereomer or a mixture of diastereomers. In some cases these diastereomers were separated and in other cases a wavy bond is used to indicate the structural element where configuration is variable.
[0056] An "effective amount" when used in connection with a compound is an amount effective for treating or preventing a disease in a subject as described herein.
[0057] The term "carrier", as used in this disclosure, encompasses carriers, excipients, and diluents and means a material, composition or vehicle, such as a liquid or solid filler, diluent, excipient, solvent or encapsulating material, involved in carrying or transporting a pharmaceutical agent from one organ, or portion of the body, to another organ, or portion of the body of a subject.
[0058] The term "treating" with regard to a subject, refers to improving at least one symptom of the subject's disorder. Treating includes curing, improving, or at least partially ameliorating the disorder.
[0059] The term "disorder" is used in this disclosure to mean, and is used interchangeably with, the terms disease, condition, or illness, unless otherwise indicated.
[0060] The term "administer", "administering", or "administration" as used in this disclosure refers to either directly administering a disclosed compound or pharmaceutically acceptable salt of the disclosed compound or a composition to a subject, or administering a prodrug derivative or analog of the compound or pharmaceutically acceptable salt of the compound or composition to the subject, which can form an equivalent amount of active compound within the subject's body.
[0061] A "patient" or "subject" is a mammal, e.g., a human, mouse, rat, guinea pig, dog, cat, horse, cow, pig, or non-human primate, such as a monkey, chimpanzee, baboon or rhesus.
[0062] In one or more embodiments of the compounds of Formula I, n is 0 and m is 1.
[0063] In one or more embodiments of the compounds of Formula I, n is 1 and m is 1.
[0064] In one or more embodiments of the compounds of Formula I, X.sup.5 is C(O).
[0065] In one or more embodiments of the compounds of Formula I, L is a bond. In one or more embodiments, L is --(CR.sup.1R.sup.2).sub.p--. In one or more embodiments, L is --C(O)NR.sup.3--. In one or more embodiments, L is --S(O).sub.2--. In one or more embodiments, L is --S(O).sub.2NR.sup.3--. In one or more embodiments, L is --S(O)--. In one or more embodiments, L is --S(O)NR.sup.3--. In one or more embodiments, L is --C(O)(CR.sup.1R.sup.2).sub.pO--. In one or more embodiments, L is --C(O)(CR.sup.1R.sup.2).sub.p--.
[0066] In one or more embodiments of the compounds of Formula I, the compound is of the Formula IA:
##STR00003##
[0067] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2-- and X.sup.2 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, and X.sup.3 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0068] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0069] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0070] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --NH--. In one or more embodiments, X.sup.1 is --NH-- and X.sup.2 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, and X.sup.3 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--. X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0071] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond. --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0072] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--. X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0073] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --O--. In one or more embodiments, X.sup.1 is --O-- and X.sup.2 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, and X.sup.3 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0074] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0075] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond. --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0076] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --C(O)--. In one or more embodiments, X.sup.1 is --C(O)-- and X.sup.2 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, and X.sup.3 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0077] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0078] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0079] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --S(O).sub.2--. In one or more embodiments, X.sup.1 is --S(O).sub.2-- and X.sup.2 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, and X.sup.3 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0080] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0081] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0082] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --S(O)--. In one or more embodiments, X.sup.1 is --S(O)-- and X.sup.2 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, and X.sup.3 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0083] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0084] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)CR.sup.1R.sup.2).sub.p--.
[0085] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --S--. In one or more embodiments, X.sup.1 is --S-- and X.sup.2 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, and X.sup.3 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0086] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0087] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2)P--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0088] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2-- and X.sup.2 is --NH--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, and X.sup.3 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.4 is --CH.sub.2--, X is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3-, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0089] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0090] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0091] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2-- and X.sup.2 is --O--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, and X.sup.3 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0092] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0093] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0094] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2-- and X.sub.2 is --C(O)--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, and X.sup.3 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.3 is --CH.sub.1--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.1 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0095] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.1 is --CH.sub.2--, X.sup.1 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0096] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.3 is --CH.sub.2--. X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0097] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2-- and X.sup.2 is --S(O).sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, and X.sup.3 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0098] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.1 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0099] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0100] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2-- and X.sup.2 is --S(O)--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O--, and X.sup.3 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0101] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0102] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0103] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2-- and X.sup.2 is --S--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, and X.sup.3 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.4 is --CH.sub.2--, X.sup.2 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0104] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0105] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond. --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0106] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, and X.sup.3 is --NH--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --NH-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --NH--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --NH--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0107] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --NH--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --NH--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --NH--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --NH--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0108] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --NH--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.1 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.3 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --NH--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0109] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, and X.sup.3 is --O--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --O-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --O--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --O--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0110] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --O--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --O--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --O--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --O--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0111] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --O--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --O--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0112] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, and X.sup.3 is --S--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0113] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--. X.sup.3 is --S--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0114] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.1 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0115] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, and X.sup.3 is --C(O)--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0116] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0117] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0118] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, and X.sup.3 is --S(O).sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O).sub.2-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O).sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O).sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In any of the above-embodiments, L is a bond. --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0119] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O).sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O).sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O).sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O).sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0120] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O).sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O).sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O).sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond. --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0121] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, and X.sup.3 is --S(O)--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O)-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O)--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0122] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O)--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0123] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0124] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --NH--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --NH-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --NH--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --NH--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --NH--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --NH--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --NH--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --NH--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --NH--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0125] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --NH--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --NH--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --NH--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.1 is --C(O)--, X.sup.4 is --NH--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0126] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --NH--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --NH--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --NH--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0127] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --O--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --O-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --O--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --O--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --O--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --O--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --O--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --O--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --O--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0128] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --O--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --O--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --O--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --O--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0129] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --O--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --O--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --O--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0130] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --S--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0131] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --S--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0132] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --S--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0133] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --C(O)--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0134] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0135] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0136] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --S(O).sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S(O).sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S(O).sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S(O).sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S(O).sub.2--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S(O).sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S(O).sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S(O).sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S(O).sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0137] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S(O).sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S(O).sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S(O).sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --S(O).sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S(O).sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3-, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0138] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S(O).sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --S(O).sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S(O).sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0139] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --S(O)--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S(O)-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S(O)--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S(O)--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S(O)--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S(O)--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0140] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S(O)--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --S(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0141] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S(O)--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --S(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0142] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --NH--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --NH-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --NH--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --NH--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --NH--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --NH--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --NH--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --NH--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.pO--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0143] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --NH--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --NH--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --NH--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --NH--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --NH--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0144] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --NH--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --NH--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond. --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0145] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --O--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --O-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --O--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --O--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --O--, X.sup.1 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --O--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --O--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --O--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0146] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --O--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --O--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --O--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --O--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --O--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0147] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --O--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --O--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0148] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --S--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --S--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --S--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3-, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0149] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --S--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0150] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0151] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --C(O)--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0152] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.5 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0153] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0154] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --S(O).sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O).sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O).sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O).sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O).sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --S(O).sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O).sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O).sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --S(O).sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4; is --CH.sub.2--, X.sup.6 is --S(O).sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)CR.sup.1R.sup.2).sub.pO--, or --C(O)CR.sup.1R.sup.2).sub.p--.
[0155] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O).sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O).sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O).sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O).sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --S(O).sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0156] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.5 is --S(O).sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O).sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O).sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0157] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --S(O)--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O)-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O)--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O)--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --S(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --S(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0158] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O)--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O)--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --S(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0159] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0160] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --NH--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --NH-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --NH--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --NH--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --NH--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --NH--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --NH--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --NH--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3-, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(OX(R.sup.1R.sup.2).sub.p--.
[0161] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --NH-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --NH--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --NH--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --NH--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0162] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --NH--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --NH--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --NH--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0163] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --O--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --O-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --O--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --O--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --O--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --O--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --O--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --O--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0164] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.5 is --O-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --O--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --O--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --O--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0165] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --O--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --O--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --O--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O)--, --S(O).sub.zNR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0166] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --C(O)--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --C(O)-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --C(O)--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --C(O)--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --C(O)--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --C(O)--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --C(O)--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --C(O)--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O)--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0167] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --C(O)-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --C(O)--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --C.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --C(O)--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --C(O)--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0168] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --C(O)--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --C(O)--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --C(O)--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3-, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0169] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --S(O).sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O).sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O).sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O).sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --S(O).sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O).sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O).sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --S(O).sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0170] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O).sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O).sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O).sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O).sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0171] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --S(O).sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O).sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O).sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0172] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --S(O)--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O)-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O)--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O)--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --S(O)--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O)--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O)--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --S(O)--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0173] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O)-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O)--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O)--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O)--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0174] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --S(O)--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O)--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O)--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0175] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --S--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --S--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --S--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0176] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0177] In one or more embodiments of the compounds of Formula IA, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --S--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0178] In one or more embodiments of the compounds of Formula IA, the compound is of the formula IA-1, IA-2, IA-3, IA-4, IA-5, IA-6, IA-7, or IA-8:
##STR00004##
[0179] In one or more embodiments, of the compounds of Formula I, the compound is of the Formula IB:
##STR00005##
[0180] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2-- and X.sup.2 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, and X.sup.3 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0181] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0182] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0183] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --NH--. In one or more embodiments, X.sup.1 is --NH-- and X.sup.2 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, and X.sup.3 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--. X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0184] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond. --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0185] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --NH--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0186] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --O--. In one or more embodiments, X.sup.1 is --O-- and X.sup.2 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, and X.sup.3 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0187] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0188] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --O--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond. --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0189] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --C(O)--. In one or more embodiments, X.sup.1 is --C(O)-- and X.sup.2 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, and X.sup.3 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0190] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0191] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --C(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0192] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --S(O).sub.2--. In one or more embodiments, X.sup.1 is --S(O).sub.2-- and X.sup.2 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, and X.sup.3 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.1 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0193] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0194] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --S(O).sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0195] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --S(O)--. In one or more embodiments, X.sup.1 is --S(O)-- and X.sup.2 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, and X.sup.3 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0196] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0197] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --S(O)--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)CR.sup.1R.sup.2).sub.p--.
[0198] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --S--. In one or more embodiments, X.sup.1 is --S-- and X.sup.2 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, and X.sup.3 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0199] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sup.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0200] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --S--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2)P--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0201] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2-- and X.sup.2 is --NH--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, and X.sup.3 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0202] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0203] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --NH--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0204] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2-- and X.sup.2 is --O--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, and X.sup.3 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0205] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0206] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --O--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0207] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2-- and X.sub.2 is --C(O)--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, and X.sup.3 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0208] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0209] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sub.2 is --C(O)--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3-, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0210] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2-- and X.sup.2 is --S(O).sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, and X.sup.3 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.3 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0211] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0212] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O).sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0213] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2-- and X.sup.2 is --S(O)--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, and X.sup.3 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.1 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0214] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond. --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0215] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S(O)--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0216] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2-- and X.sup.2 is --S--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, and X.sup.3 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0217] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0218] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --S--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond. --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0219] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, and X.sup.3 is --NH--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --NH-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --NH--, X.sup.4 is --CH.sub.2- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --NH--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0220] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --NH--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --NH--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --NH--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --NH--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0221] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --NH--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --NH--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0222] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, and X.sup.3 is --O--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --O-- and X.sup.4 is --CH.sub.2--. In one or more embodiments. X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --O--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --O--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0223] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --O--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --O--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --O--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --O--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0224] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --O--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --O--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0225] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, and X.sup.3 is --S--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0226] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0227] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0228] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, and X.sup.3 is --C(O)--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0229] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0230] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0231] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, and X.sup.3 is --S(O).sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O).sub.2- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O).sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O).sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0232] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O).sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O).sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O).sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O).sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0233] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O).sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O).sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O).sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond. --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0234] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, and X.sup.3 is --S(O)--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O)-- and X.sup.4 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O)--, X.sup.4 is --CH.sub.2- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0235] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O)--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0236] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --S(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0237] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --NH--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --NH-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --NH--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --NH--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --NH--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --NH--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --NH--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --NH--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --NH--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0238] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --NH--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --NH--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --NH--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --NH--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0239] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --NH--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --NH--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --NH--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0240] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --O--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --O-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --O--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --O--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --O--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --O--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --O--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --O--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --O--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0241] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --O--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --O--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --O--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --O--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0242] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --O--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --O--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --O--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0243] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --S--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0244] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --S--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0245] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --S--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0246] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --C(O)--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.1 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0247] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0248] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0249] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --S(O).sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.1 is --CH.sub.2--, X.sup.4 is --S(O).sub.2-- and X.sup.1 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S(O).sub.2--, X.sup.1 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S(O).sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S(O).sub.2--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S(O).sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S(O).sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S(O).sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S(O).sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0250] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S(O).sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S(O).sub.2--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S(O).sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --S(O).sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S(O).sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0251] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S(O).sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --S(O).sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S(O).sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.4--, --C(O)(CR.sup.1R.sup.2).sup.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0252] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2-- and X.sup.4 is --S(O)--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S(O)-- and X.sup.5 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S(O)--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S(O)--, X.sup.5 is --CH.sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S(O)--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --S(O)--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0253] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S(O)--, X.sup.5 is --CH.sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --S(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0254] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S(O)--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --S(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --S(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0255] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --NH--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --NH-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --NH--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --NH--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --NH--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --NH--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --NH--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --NH--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0256] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --NH--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --NH--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --NH--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --NH--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --NH--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0257] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --NH--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --NH--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond. --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0258] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --O--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --O-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --O--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --O--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --O--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --O--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --O--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --O--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0259] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --O--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --O--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --O--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --O--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --O--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sup.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0260] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --O--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --O--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0261] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --S--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --S--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --S--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0262] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --S--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0263] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0264] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --C(O)--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0265] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.5 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0266] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0267] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2-- and X.sup.5 is --S(O).sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O).sub.2-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O).sub.2--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O).sub.2--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O).sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --S(O).sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O).sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O).sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.3 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --S(O).sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O).sub.2--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3-, --S(O)--, --S(O)NR.sup.3--, --C(O)CR.sup.1R.sup.2).sub.pO--, or --C(O)CR.sup.1R.sup.2).sub.p--.
[0268] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O).sub.2--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O).sub.2--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O).sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O).sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --S(O).sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0269] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O).sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O).sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O).sub.2--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond. --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0270] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.6 is --CH.sub.2-- and X.sup.5 is --S(O)--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O)-- and X.sup.6 is --CH.sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O)--, X.sup.6 is --CH.sub.2-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O)--, m is 0 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --S(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --S(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O)--, X.sup.6 is --CH.sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0271] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O)--, X.sup.6 is --CH.sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O)--, m is 1 and n is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --S(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0272] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --S(O)--, X.sup.6 is --CH.sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0273] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --NH--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --NH-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --NH--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --NH--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --NH--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --NH--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --NH--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --NH--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(OX(R.sup.1R.sup.2).sub.p--.
[0274] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --NH-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --NH--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --NH--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --NH--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0275] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --NH--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --NH--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --NH--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0276] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --O--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --O-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --O--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --O--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --O--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --O--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --O--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --O--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0277] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --O-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --O--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --O--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.1 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --O--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0278] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --O--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --O--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --O--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0279] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --C(O)--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --C(O)-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --C(O)--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --C(O)--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --C(O)--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --C(O)--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --C(O)--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --C(O)--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O)--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0280] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--. X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --C(O)-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --C(O)--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --C(O)--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --C(O)--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0281] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --C(O)--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --C(O)--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --C(O)--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--. --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0282] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --S(O).sub.2--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O).sub.2--, and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--. X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O).sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O).sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --S(O).sub.2--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O).sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O).sub.2--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --S(O).sub.2--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0283] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O).sub.2-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O).sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O).sub.2--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O).sub.2--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0284] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --S(O).sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O).sub.2--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O).sub.2--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0285] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --S(O)--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O)-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O)--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O)--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --S(O)--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O)--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O)--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --S(O)--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0286] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O)-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O)--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O)--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O)--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0287] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --S(O)--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O)--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S(O)--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0288] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2-- and X.sup.6 is --S--. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S-- and m is 0. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --(CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --S--, m is 0 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S--, m is 0 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --S--, m is 0 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0289] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S-- and m is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S--, m is 1 and n is 1. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --C(O)--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S--, m is 1 and n is 1. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0290] In one or more embodiments of the compounds of Formula IB, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --C(O)--, X.sup.6 is --S--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --C(O)--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S--, m is 1 and n is 2. In one or more embodiments, X.sup.1 is --CH.sub.2--, X.sup.2 is --CH.sub.2--, X.sup.3 is --CH.sub.2--, X.sup.4 is --CH.sub.2--, X.sup.5 is --CH.sub.2--, X.sup.6 is --S--, m is 1 and n is 2. In any of the above-embodiments, L is a bond, --(CR.sup.1R.sup.2).sub.p--, --C(O)NR.sup.3--, --S(O).sub.2--, --S(O).sub.2NR.sup.3--, --S(O)--, --S(O)NR.sup.3--, --C(O)(CR.sup.1R.sup.2).sub.pO--, or --C(O)(CR.sup.1R.sup.2).sub.p--.
[0291] In one or more embodiments of the compounds of Formula IB, the compound is of the formula IB-1, IB-2, IB-3, IB-4, IB-5, IB-6, IB-7, or IB-8:
##STR00006##
[0292] In one or more embodiments, a compound of Formula I can be: [0293] (R)--N-hydroxy-1'-methyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]- -5-carboxamide (I-1); [0294] (S)--N-hydroxy-1'-methyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]- -5-carboxamide (I-2); [0295] (R)--N-hydroxy-1'-isopropyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidi- ne]-5-carboxamide (I-3); [0296] (S)--N-hydroxy-1'-isopropyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidi- ne]-5-carboxamide (I-4); [0297] (R)-1'-ethyl-N-hydroxy-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5- -carboxamide (I-5); [0298] (S)-1'-ethyl-N-hydroxy-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5- -carboxamide (I-6); [0299] (R)--N-hydroxy-1'-isobutyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidin- e]-5-carboxamide (I-7). [0300] (S)--N-hydroxy-1'-isobutyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidin- e]-5-carboxamide (I-8); [0301] (R)--N-hydroxy-1'-(2-methylbenzyl)-2'-oxo-1,3-dihydrospiro[indene-2,3'-py- rrolidine]-5-carboxamide (I-9); [0302] (S)--N-hydroxy-1'-(2-methylbenzyl)-2'-oxo-1,3-dihydrospiro[indene-2,3'-py- rrolidine]-5-carboxamide (I-10); [0303] (R)-1'-benzyl-N-hydroxy-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-- 5-carboxamide (I-11); [0304] (S)-1'-benzyl-N-hydroxy-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-- 5-carboxamide (I-12); [0305] (R)--N-hydroxy-2'-oxo-1'-phenyl-1,3-dihydrospiro[indene-2,3'-pyrrolidine]- -5-carboxamide (I-13); [0306] (S)--N-hydroxy-2'-oxo-1'-phenyl-1,3-dihydrospiro[indene-2,3'-pyrrolidine]- -5-carboxamide (I-14); [0307] (R)-1'-(4-fluorophenyl)-N-hydroxy-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyr- rolidine]-5-carboxamide (I-15); [0308] (S)-1'-(4-fluorophenyl)-N-hydroxy-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyr- rolidine]-5-carboxamide (I-16); [0309] (R)--N-hydroxy-2'-oxo-1'-(pyridin-3-yl)-1,3-dihydrospiro[indene-2,3'-pyrr- olidine]-5-carboxamide (I-17); [0310] (S)--N-hydroxy-2'-oxo-1'-(pyridin-3-yl)-1,3-dihydrospiro[indene-2,3'-pyrr- olidine]-5-carboxamide [0311] (R)--N-hydroxy-2'-oxo-1'-(pyridin-2-yl)-1,3-dihydrospiro[indene-2,3'-pyrr- olidine]-5-carboxamide (I-19); [0312] (S)--N-hydroxy-2'-oxo-1'-(pyridin-2-yl)-1,3-dihydrospiro[indene-2,3'-pyrr- olidine]-5-carboxamide (I-20); [0313] (R)--N-hydroxy-2'-oxo-1'-(pyridin-4-yl)-1,3-dihydrospiro[indene-2,3'-pyrr- olidine]-5-carboxamide (I-21), [0314] (S)--N-hydroxy-2'-oxo-1'-(pyridin-4-yl)-1,3-dihydrospiro[indene-2,3'-pyrr- olidine]-5-carboxamide (I-22); [0315] (R)--N-hydroxy-2'-oxo-1'-(pyridin-3-ylmethyl)-1,3-dihydrospiro[indene-2,3- '-pyrrolidine]-5-carboxamide (I-23); [0316] (R)--N-hydroxy-1'-methyl-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carb- oxamide (I-24); [0317] (S)--N-hydroxy-1'-methyl-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carb- oxamide (I-25); [0318] (R)-1'-benzyl-N-hydroxy-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carbo- xamide (I-26); [0319] (S)-1'-benzyl-N-hydroxy-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carbo- xamide (I-27); [0320] (R)-1'-acetyl-N-hydroxy-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carbo- xamide (I-28); [0321] (S)-1'-acetyl-N-hydroxy-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carbo- xamide (I-29); [0322] (R)-1'-benzoyl-N-hydroxy-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carb- oxamide (I-30); [0323] (S)-1'-benzoyl-N-hydroxy-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carb- oxamide (I-31); [0324] (R)--N-hydroxy-1'-methyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-- 5-carboxamide (I-32); [0325] (S)--N-hydroxy-1'-methyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-- 5-carboxamide (I-33); [0326] (R)-1'-benzyl-N-hydroxy-2'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-5- -carboxamide (I-34); [0327] (S)-1'-benzyl-N-hydroxy-2'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-5- -carboxamide (I-35); [0328] (R)--N-hydroxy-2'-oxo-1'-phenyl-1,3-dihydrospiro[indene-2,3'-piperidine]-- 5-carboxamide (I-36); or [0329] (S)--N-hydroxy-2'-oxo-1'-phenyl-1,3-dihydrospiro[indene-2,3'-piperidine]-- 5-carboxamide (I-37).
[0330] In one or more embodiments, a compounds of Formula I can be: [0331] N-hydroxy-1'-methyl-5'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-ca- rboxamide (I-38); [0332] N-hydroxy-1'-methyl-6'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-5-car- boxamide (I-39); [0333] N-hydroxy-1'-methyl-2'-oxo-1,3-dihydrospiro[indene-2,4'-piperidine]-5-car- boxamide (I-40); [0334] N-hydroxy-1'-methyl-1,3-dihydrospiro[indene-2,3'-piperidine]-5-carboxamid- e (I-41); [0335] N-hydroxy-1'-methyl-1,3-dihydrospiro[indene-2,4'-piperidine]-5-carboxamid- e (I-42); [0336] N-hydroxy-1'-methyl-3H-spiro[benzofuran-2,4'-piperidine]-6-carboxamide (I-43); or [0337] N-hydroxy-1'-methyl-3H-spiro[benzofuran-2,4'-piperidine]-6-carboxamide (I-44).
[0338] In some embodiments of the invention, the compounds of Formula I are enantiomers. In some embodiments the compounds are the (S)-enantiomer. In other embodiments the compounds are the (R)-enantiomer. In yet other embodiments, the compounds of Formula I may be (+) or (-) enantiomers. As used herein, a chemical structure that is labelled as "R/S" indicates that the structure represents one enantiomer, the stereochemistry of which is not defined.
[0339] It should be understood that all isomeric forms are included within the present invention, including mixtures thereof. If the compound contains a double bond, the substituent may be in the E or Z configuration. If the compound contains a disubstituted cycloalkyl, the cycloalkyl substituent may have a cis- or trans-configuration. All tautomeric forms are also intended to be included.
Methods of Synthesizing the Disclosed Compounds
[0340] The compounds of the present invention may be made by a variety of methods, including standard chemistry. Suitable synthetic routes are depicted in the schemes given below.
[0341] The compounds of Formula I may be prepared by methods known in the art of organic synthesis as set forth in part by the following synthetic schemes and examples. In the schemes described below, it is well understood that protecting groups for sensitive or reactive groups are employed where necessary in accordance with general principles or chemistry. Protecting groups are manipulated according to standard methods of organic synthesis (T. W. Greene and P. G. M. Wuts, "Protective Groups in Organic Synthesis", Third edition, Wiley, New York 1999). These groups are removed at a convenient stage of the compound synthesis using methods that are readily apparent to those skilled in the art. The selection processes, as well as the reaction conditions and order of their execution, shall be consistent with the preparation of compounds of Formula I.
[0342] Those skilled in the art will recognize if a stereocenter exists in the compounds of Formula I. Accordingly, the present invention includes both possible stereoisomers (unless specified in the synthesis) and includes not only racemic compounds but the individual enantiomers and/or diastereomers as well. When a compound is desired as a single enantiomer or diastereomer, it may be obtained by stereospecific synthesis or by resolution of the final product or any convenient intermediate. Resolution of the final product, an intermediate, or a starting material may be affected by any suitable method known in the art. See, for example, "Stereochemistry of Organic Compounds" by E. L. Eliel, S. H. Wilen, and L. N. Mander (Wiley-Interscience, 1994).
Preparation of Compounds
[0343] The compounds described herein may be made from commercially available starting materials or synthesized using known organic, inorganic, and/or enzymatic processes.
[0344] The compounds of the present invention can be prepared in a number of ways well known to those skilled in the art of organic synthesis. By way of example, compounds of the Formula I (e.g., Formula IA-9 and IA-10) can be synthesized using the methods described below, together with synthetic methods known in the art of synthetic organic chemistry, or variations thereon as appreciated by those skilled in the art. These methods include but are not limited to those methods described below.
##STR00007## ##STR00008##
wherein L and R are defined as in Formula (I).
[0345] The general way of preparing target molecules of Formula (IA-9) by using intermediates 1a, 1b, 1c, 1d, 1e, 1f, 1g, and 1h is outlined in Scheme 1. Carbonylation of 5-bromo-2,3-dihydro-1H-inden-1-one (1a) in the presence of a metal catalyst, e.g., [1,1'-Bis(diphenylphosphino)ferrocene]dichloropalladium(II) (Pd(dppf)Cl.sub.2), carbon monoxide, and a base, e.g., triethylamine (Et.sub.3N), provides ester 1b. Deprotonation of 1b using a strong base, e.g., sodium hydride (NaH), in the presence of dimethyl carbonate affords Intermediate 1c, which can be alkylated by treatment with a halo-nitrile in the presence of a base to provide Intermediates 1d. Reduction, for example, with hydrogen gas in the presence of platinum (IV) oxide (PtO.sub.2), acetic acid, and methanol, can provide amino-alcohol Intermediates 1e. Spiro-lactams 1f can be obtained by treatment of 1e with ammonia (NH.sub.3) in methanol. Dehydroxylation of 1f by conversion to the silyl ether can be accomplished by treatment with triethylsilane and TFA to provide Intermediates 1g. Addition of the R-L moiety can be achieved via standard methods of alkylation or arylation. For example, alkylation of 1g with an alkyl halide in the presence of a base, e.g., sodium hydride, can provide compounds of Intermediates 1h. Alternatively, arylation of 1g with an aryl boronic acid in the presence of a metal catalyst, e.g., copper (II) acetate (Cu(OAc).sub.2), and a base, e.g., Et.sub.3N, can also provide compounds of Intermediates 1h. Treatment of 1h with hydroxylamine and a base, e.g., sodium hydroxide, provides compounds of Formula (IA-9).
##STR00009##
wherein L and R are defined as in Formula (I).
[0346] The general way of preparing target molecules of Formula (IA-10) by using intermediates 1g, 1i, 1j, and 1k is outlined in Scheme 2. Spiro-amines 1j can be obtained via reduction of thiolactam 1i by treatment of 1g with 2,4-bis(4-methoxyphenyl)-2,4-dithioxo-1,3,2,4-dithiadiphosphetane (Lawesson reagent), followed by sodium borohydride (NaBH.sub.4) in the presence of Nickel (II) chloride hexahydrate (NiCl.sub.2.6H.sub.2O). Addition of the R-L moiety can be achieved via standard methods of alkylation, arylation, acylation, urea formation, or sulfonation. Treatment of 1k with hydroxylamine and a base, e.g., sodium hydroxide, provides compounds of Formula (IA-10).
Methods of Using the Disclosed Compounds
[0347] Another aspect of the invention relates to a method of treating a disease associated with HDAC6 modulation in a subject in need thereof. The method involves administering to a patient in need of treatment for diseases or disorders associated with HDAC6 modulation an effective amount of a compound of Formula I. In an embodiment, the disease can be, but is not limited to, cancer, neurodegenerative disease, neurodevelopmental disease, inflammatory or autoimmune disease, infection, metabolic disease, hematologic disease, transplant rejection, or cardiovascular disease.
[0348] Another aspect of the invention is directed to a method of inhibiting HDAC6. The method involves administering to a patient in need thereof an effective amount of Formula I.
[0349] The present invention relates to compositions capable of modulating the activity of (e.g., inhibiting) HDACs, and in particular HDAC6. The present invention also relates to the therapeutic use of such compounds.
[0350] One therapeutic use of the compounds of the present invention is to treat proliferative diseases or disorders such as cancer. Cancer can be understood as abnormal or unregulated cell growth within a patient and can include but is not limited to lung cancer, ovarian cancer, breast cancer, prostate cancer, pancreatic cancer, hepatocellular cancer, renal cancer and leukemias such as acute myeloid leukemia and acute lymphoblastic leukemia. Additional cancer types include T-cell lymphoma (e.g., cutaneous T-cell lymphoma, peripheral T-cell lymphoma), Hodgkin lymphoma, melanoma and multiple myeloma. In other embodiments, treating proliferative diseases or disorders can include any cancer where there is evidence of an increase in Treg/effector T cell ratio or in an absolute Treg number, either in the periphery or in the tumor microenvironment or tertiary lymphoid structures, or increased expression of T cell tolerance-related genes. Such proliferative diseases or disorders can include but are not limited to: any Kras mutant carrying tumor (Zdanov et al., Mutant KRAS conversion of conventional T cells into regulatory T cells. Cancer Immunol. Res. Feb. 15, 2016, DOI: 10.1158/2326-6066.CIR-15-0241); renal cell carcinoma; lung carcinoma; cervical cancer, prostate cancer; ovarian cancer; head and neck cancer, lymphoma; colorectal cancer, non-small cell lung carcinoma; breast cancers (Gobert, M. et al., (2009) Cancer Res. 69, 2000-2009); and bladder cancer.
[0351] One therapeutic use of the compounds of the present disclosure is to treat neurological diseases or disorders or neurodegeneration. Neurological disorders are understood as disorders of the nervous system (e.g., the brain and spinal cord). Neurological disorders or neurodegenerative diseases can include but are not limited to epilepsy, attention deficit disorder (ADD), Alzheimer's disease, Parkinson's Disease, Huntington's Disease, amyotrophic lateral sclerosis, spinal muscular atrophy, essential tremor, central nervous system trauma caused by tissue injury, oxidative stress-induced neuronal or axomal degeneration, and multiple sclerosis.
[0352] Another therapeutic use of the compounds of the present disclosure is to treat neurodevelopmental disorders. Neurodevelopmental disorders can include, but are not limited to, Rett syndrome, intellectual disability, intellectual and developmental disability, autism spectrum disorder, fetal alcohol syndrome, developmental coordination disorder, stereotypic movement disorder, Tourette syndrome, cerebral palsy, fragile X syndrome, attention deficit hyperactivity disorder, and Mendelsohnn's syndrome.
[0353] Another therapeutic use of the compounds of the present invention is also to treat inflammatory diseases or disorders. Inflammation can be understood as a host's response to an initial injury or infection. Symptoms of inflammation can include but are not limited to redness, swelling, pain, heat and loss of function. Inflammation may be caused by the upregulation of pro-inflammatory cytokines such as IL-1.beta., and increased expression of the FOXP3 transcription factor. In some embodiments, the inflammatory diseases include fibrosis or fibrotic diseases. Types of fibrotic diseases include but are not limited to lung fibrosis or pulmonary fibrosis, Liver fibrosis; Heart fibrosis; Mediastinal fibrosis; Retroperitoneal cavity fibrosis; Bone marrow fibrosis; Skin fibrosis, and Scleroderma or systemic sclerosis.
[0354] Another therapeutic use of the compounds of the present invention is also to treat autoimmune diseases or disorders. Autoimmune disorders are understood as disorders wherein a host's own immune system responds to tissues and substances occurring naturally in the host's body. Autoimmune diseases can include but are not limited to rheumatoid arthritis, Crohn's disease, type-1 diabetes, systemic juvenile idiopathic arthritis; inflammatory bowel disease; allograft transplantation; eczema, psoriasis, idiopathic thrombocytopenic purpra, autoimmune thrombocytopenia, acquired immune thrombocytopenia, autoimmune neutropenia, autoimmune hemolyitic anemia, parvovirus B19-associated red cell aplasia, acquired antifactor VIII autoimmunity, acquired von Willebrand disease, monoclonal gammopathy, aplastic anemia, pure red cell aplasia, Diamond-Blackfan anemia, hemolytic disease of the newborn, immune mediated-refractoriness to platelet transfusion, hemolytic uremic syndrome, Evan's syndrome, Guillain-Barre syndrome, chronic demyelinating polyradiculoneuropathy, paraproteinemic IgM demyelinating polyneuropathy, Lamber-Eaton myasthenic syndrome, myasthenia gravis, multifocal motor neuropathy, stiff man syndrome, paraneoplastic encephalomyelitis, sensory neuropathy with anti-Hu antibodies, myelitis, autoimmune diabetic neuropathy, acute idiopathic neuropathy, toxic epidermal necrolysis, gangrene, granuloma, pemphigus vulgaris, bullous pemphigoid, vitiligo, scleroderma, atomic dermatitis, systemic and diffuse sclerosis, primary biliary cirrhosis, Celiac disease, dermatitis herpetiformis, cryptogenic cirrhosis, reactive arthritis, Hashimoto's thryroditis, Wegner's granulomoatosis, micropolyarterits, Churg-Strauss syndrome Type I and Type II autoimmune polyglandular syndromes, linear IgA disease, epidermolysis bullosa acquisita, erythema nodosa, pemphigoid gestationis, cicatricial pemphigoid, mixed essential cryoglobulinemia, chronic bullous disease of childhood, Goodpasture's syndrome, sclerosis cholangitis, ankylosing spondylitis, Bechet's syndrome temporal arteritis, Takayasu's arteritis, autoimmune urticaria, and Kawasaki's disease.
[0355] In some embodiments, the cancer to be treated is melanoma, cutaneous T-cell lymphoma, peripheral T-cell lymphoma, Hodgkin lymphoma, multiple myeloma, leukemia, lung, ovarian, breast, prostate, pancreatic, hepatocellular or renal cancer. In some embodiments, the cancer to be treated is cutaneous T-cell lymphoma, peripheral T-cell lymphoma, multiple myeloma, lymphoma, leukemia, lung, ovarian, breast, prostate, pancreatic, hepatocellular or renal cancer. In other embodiments, the neurodegenerative disease to be treated is Alzheimer's Disease, Huntington's Disease, Parkinson's Disease, Amyotrophic Lateral Sclerosis, or spinal muscular atrophy. In other embodiments, the neurodevelopmental disorder to be treated is Rett syndrome. In yet other embodiments, the inflammatory or autoimmune disease to be treated is rheumatoid arthritis, spondylitis arthritis, psoriatic arthritis, psoriasis, multiple sclerosis, systemic lupus erythematosus, inflammatory bowel diseases, graft versus host disease, transplant rejection or fibrotic disease.
[0356] Another therapeutic use of the compounds of the present invention is also to treat and/or prevent allergy and unwanted immune responses associated with allergy. A non-limiting list of allergies and related conditions includes, pollen allergy (e.g. Japanese Cedar Pollen), mold allergy, food allergies (including, but not limited to peanut, tree nut, milk, soy, gluten, and egg allergies), animal allergies (e.g. allergies to dogs, cats, rabbits), dust mite allergy, atopic dermatitis, allergic rhinitis, allergic otitis, allergic asthma, dry eye, ocular allergy, allergic urticaria, contact dermatitis, anaphalaxis, eosinophilic esophagitis.
[0357] Another therapeutic use of the compounds of the present invention is also to treat infectious diseases or disorders. Infections or infectious diseases are caused by the invasion of a foreign pathogen. The infection may be caused by, for instance, a bacteria, a fungus, or virus. Bacterial infections include, but are not limited to streptococcus infections, mycobacterial infections, bacillus infections, Salmonella infections, Vibrio infections, spirochete infections, and Neisseria infections. Viral infections include, but are not limited to herpes virus infections, hepatitis virus infections, west nile virus infections, flavivrus infections, influenza virus infections, rhinovirus infections, papillomavirus infections, paromyxovirus infections, parainfluenza virus infections, and retrovirus infections. In particular embodiments, the compounds of the present invention are useful for treating infections which result in an inflammatory cytokine burst. Nonlimiting examples of such infections include Ebola and the viral hemorghagic fever-causing viruses, and Malaria.
[0358] Yet another therapeutic use of the compounds of the present invention is also to treat metabolic diseases or disorders. Metabolic diseases can be characterized as abnormalities in the way that a subject stores energy. Metabolic disorders can include but are not limited to metabolic syndrome, diabetes, obesity, high blood pressure, non-alcoholic fatty liver disease and heart failure.
[0359] Yet another therapeutic use of the compounds of the present invention is also to treat hematologic disorders. Hematologic diseases primarily affect the blood. Hematologic disorders can include but are not limited to anemia, multiple myeloma, lymphoma, and leukemia.
[0360] Yet another therapeutic use of the compounds of the present invention is also to prevent and/or treat transplant rejection. Tissues that are transplanted include (but are not limited to) whole organs such as kidney, liver, heart, lung, organ components such as skin grafts and the cornea of the eye; and cell suspensions such as bone marrow cells and cultures of cells selected and expanded from bone marrow or circulating blood, and whole blood transfusions.
[0361] Yet another therapeutic use of the compounds of the present invention is also to treat cardiovascular diseases or disorders. Cardiovascular diseases affect the heart and blood vessels of a patient. Exemplary conditions include but are not limited to cardiovascular stress, pressure overload, chronic ischemia, infarction-reperfusion injury, hypertension, Brain infarct after cerebral artery occlusion, atherosclerosis, peripheral artery disease, cardiac hypertrophy, cardiac arrhythmias, stroke, and heart failure.
[0362] Another therapeutic use of the compounds of the present invention is for purging the reservoir of latently infected memory CD4+ T cells in HIV+ patients (Matalon, et al., Mol Med. 2011; 17(5-6); 466-472), which is hereby incorporated by reference in its entirety.
[0363] The disclosed compound can be administered in effective amounts to treat or prevent a disorder and/or prevent the development thereof in subjects.
[0364] Another aspect of the present disclosure relates to a compound of Formula I, or a pharmaceutically acceptable salt, hydrate, solvate, prodrug, stereoisomer, or tautomer thereof, for use in treating or preventing a disease associated with HDAC6 modulation. In some embodiments, the disease is cancer, neurodegenerative disease, neurodevelopmental disorder, inflammatory or autoimmune disease, infection, metabolic disease, hematologic disease, transplant rejection or cardiovascular disease. In some embodiments, the compound inhibits a histone deacetylase. In another embodiment, the compound inhibits a zinc-dependent histone deacetylase. In another embodiment, the compound inhibits the HDAC6 isozyme zinc-dependent histone deacetylase.
[0365] In another aspect, the present disclosure relates to the use of a compound of Formula (I), or a pharmaceutically acceptable salt, hydrate, solvate, prodrug, stereoisomer, or tautomer thereof, in the manufacture of a medicament for treating or preventing a disease associated with HDAC6 modulation. In some embodiments, the disease is cancer, neurodegenerative disease, neurodevelopmental disorder, inflammatory or autoimmune disease, infection, metabolic disease, hematologic disease, transplant rejection, or cardiovascular disease. In some embodiments, the compound inhibits a histone deacetylase. In another embodiment, the compound inhibits a zinc-dependent histone deacetylase. In another embodiment, the compound inhibits the HDAC6 isozyme zinc-dependent histone deacetylase.
[0366] In some embodiments, the cancer is melanoma, cutaneous T-cell lymphoma, peripheral T-cell lymphoma, Hodgkin lymphoma, multiple myeloma, leukemia, lung, ovarian, breast, prostate, pancreatic, hepatocellular or renal cancer. In some embodiments, the cancer is cutaneous T-cell lymphoma, peripheral T-cell lymphoma, multiple myeloma, lymphoma, leukemia, lung, ovarian, breast, prostate, pancreatic, hepatocellular or renal cancer. In other embodiments, the neurodegenerative disease is Alzheimer's Disease, Huntington's Disease, Parkinson's Disease, Amyotrophic Lateral Sclerosis, or spinal muscular atrophy. In other embodiments, the neurodevelopmental disorder is Rett syndrome. In yet other embodiments, the inflammatory or autoimmune disease is rheumatoid arthritis, spondylitis arthritis, psoriatic arthritis, psoriasis, multiple sclerosis, systemic lupus erythematosus, inflammatory bowel diseases, graft versus host disease, transplant rejection or fibrotic disease.
[0367] Administration of the disclosed compounds can be accomplished via any mode of administration for therapeutic agents. These modes include systemic or local administration such as oral, nasal, parenteral, transdermal, subcutaneous, vaginal, buccal, rectal or topical administration modes.
[0368] Depending on the intended mode of administration, the disclosed compositions can be in solid, semi-solid or liquid dosage form, such as, for example, injectables, tablets, suppositories, pills, time-release capsules, elixirs, tinctures, emulsions, syrups, powders, liquids, suspensions, or the like, sometimes in unit dosages and consistent with conventional pharmaceutical practices. Likewise, they can also be administered in intravenous (both bolus and infusion), intraperitoneal, subcutaneous or intramuscular form, and all using forms well known to those skilled in the pharmaceutical arts.
[0369] Illustrative pharmaceutical compositions are tablets and gelatin capsules comprising a compound of the disclosure and a pharmaceutically acceptable carrier, such as a) a diluent, e.g., purified water, triglyceride oils, such as hydrogenated or partially hydrogenated vegetable oil, or mixtures thereof, corn oil, olive oil, sunflower oil, safflower oil, fish oils, such as EPA or DHA, or their esters or triglycerides or mixtures thereof, omega-3 fatty acids or derivatives thereof, lactose, dextrose, sucrose, mannitol, sorbitol, cellulose, sodium, saccharin, glucose and/or glycine; b) a lubricant, e.g., silica, talcum, stearic acid, its magnesium or calcium salt, sodium oleate, sodium stearate, magnesium stearate, sodium benzoate, sodium acetate, sodium chloride and/or polyethylene glycol; for tablets also, c) a binder, e.g., magnesium aluminum silicate, starch paste, gelatin, tragacanth, methylcellulose, sodium carboxymethylcellulose, magnesium carbonate, natural sugars such as glucose or beta-lactose, corn sweeteners, natural and synthetic gums such as acacia, tragacanth or sodium alginate, waxes and/or polyvinylpyrrolidone, if desired; d) a disintegrant, e.g., starches, agar, methyl cellulose, bentonite, xanthan gum, algiic acid or its sodium salt, or effervescent mixtures; e) absorbent, colorant, flavorant and sweetener, f) an emulsifier or dispersing agent, such as Tween 80, Labrasol, HPMC, DOSS, caproyl 909, labrafac, labrafil, peceol, transcutol, capmul MCM, capmul PG-12, captex 355, gelucire, vitamin E TGPS or other acceptable emulsifier; and/or g) an agent that enhances absorption of the compound such as cyclodextrin, hydroxypropyl-cyclodextrin, PEG400, PEG200.
[0370] Liquid, particularly injectable, compositions can, for example, be prepared by dissolution, dispersion, etc. For example, the disclosed compound is dissolved in or mixed with a pharmaceutically acceptable solvent such as, for example, water, saline, aqueous dextrose, glycerol, ethanol, and the like, to thereby form an injectable isotonic solution or suspension. Proteins such as albumin, chylomicron particles, or serum proteins can be used to solubilize the disclosed compounds.
[0371] The disclosed compounds can be also formulated as a suppository that can be prepared from fatty emulsions or suspensions; using polyalkylene glycols such as propylene glycol, as the carrier.
[0372] The disclosed compounds can also be administered in the form of liposome delivery systems, such as small unilamellar vesicles, large unilamellar vesicles and multilamellar vesicles. Liposomes can be formed from a variety of phospholipids, containing cholesterol, stearylamine or phosphatidylcholines. In some embodiments, a film of lipid components is hydrated with an aqueous solution of drug to a form lipid layer encapsulating the drug, as described in U.S. Pat. No. 5,262,564.
[0373] Disclosed compounds can also be delivered by the use of monoclonal antibodies as individual carriers to which the disclosed compounds are coupled. The disclosed compounds can also be coupled with soluble polymers as targetable drug carriers. Such polymers can include polyvinylpyrrolidone, pyran copolymer, polyhydroxypropylmethacrylamide-phenol, polyhydroxyethylaspanamidephenol, or polyethyleneoxidepolylysine substituted with palmitoyl residues. Furthermore, the disclosed compounds can be coupled to a class of biodegradable polymers useful in achieving controlled release of a drug, for example, polylactic acid, polyepsilon caprolactone, polyhydroxy butyric acid, polyorthoesters, polyacetals, polydihydropyrans, polycyanoacrylates and cross-linked or amphipathic block copolymers of hydrogels. In one embodiment, disclosed compounds are not covalently bound to a polymer, e.g., a polycarboxylic acid polymer, or a polyacrylate.
[0374] Parental injectable administration is generally used for subcutaneous, intramuscular or intravenous injections and infusions. Injectables can be prepared in conventional forms, either as liquid solutions or suspensions or solid forms suitable for dissolving in liquid prior to injection.
[0375] Another aspect of the invention relates to a pharmaceutical composition comprising a compound of Formula I and a pharmaceutically acceptable carrier. The pharmaceutically acceptable carrier can further include an excipient, diluent, or surfactant.
[0376] Compositions can be prepared according to conventional mixing, granulating or coating methods, respectively, and the present pharmaceutical compositions can contain from about 0.1% to about 99%, from about 5% to about 90%, or from about 1% to about 20% of the disclosed compound by weight or volume.
[0377] The dosage regimen utilizing the disclosed compound is selected in accordance with a variety of factors including type, species, age, weight, sex and medical condition of the patient; the severity of the condition to be treated; the route of administration; the renal or hepatic function of the patient; and the particular disclosed compound employed. A physician or veterinarian of ordinary skill in the art can readily determine and prescribe the effective amount of the drug required to prevent, counter or arrest the progress of the condition.
[0378] Effective dosage amounts of the disclosed compounds, when used for the indicated effects, range from about 0.5 mg to about 5000 mg of the disclosed compound as needed to treat the condition. Compositions for in vivo or in vitro use can contain about 0.5, 5, 20, 50, 75, 100, 150, 250, 500, 750, 1000, 1250, 2500, 3500, or 5000 mg of the disclosed compound, or, in a range of from one amount to another amount in the list of doses. In one embodiment, the compositions are in the form of a tablet that can be scored.
[0379] Without wishing to be bound by any particular theory, the compounds of the present invention can inhibit HDACs such as HDAC6 by interacting with the zinc (Zn.sup.2+) ion in the protein's active site via the hydroxamic acid group bound to the aromatic ring of the compound. The binding can prevent the zinc ion from interacting with its natural substrates, thus inhibiting the enzyme.
EXAMPLES
[0380] The disclosure is further illustrated by the following examples and synthesis examples, which are not to be construed as limiting this disclosure in scope or spirit to the specific procedures herein described. It is to be understood that the examples are provided to illustrate certain embodiments and that no limitation to the scope of the disclosure is intended thereby. It is to be further understood that resort may be had to various other embodiments, modifications, and equivalents thereof which may suggest themselves to those skilled in the art without departing from the spirit of the present disclosure and/or scope of the appended claims.
[0381] The present invention includes a number of unique features and advantages compared with other inhibitors of HDAC enzymes, in particular HDAC6. For instance, the present invention features a unique class of small molecule therapeutic agents of Formula I.
[0382] Definitions used in the following examples and elsewhere herein are:
TABLE-US-00001 AcOH Acetic Acid CH.sub.2Cl.sub.2 Methylene chloride, Dichloromethane CH.sub.3CN Acetonitrile CH.sub.3I Iodomethane CO (g) Carbon monoxide (gas) CO.sub.2 Carbon dioxide Cs.sub.2CO.sub.3 Cesium carbonate Cu(OAc).sub.2 Copper (II) acetate DMA Dimethylacetamide DMF N,N-Dimethylformamide DMSO Dimethylsulfoxide Et.sub.3N Triethylamine EtOAc Ethyl acetate EtOH Ethanol h hours H.sub.2 (g) Hydrogen (gas) H.sub.2O Water HCl Hydrochloric acid MeOH Methanol min minutes Na.sub.2SO.sub.4 Sodium sulfate NaBH.sub.3CN Sodium cyanoborohydride NaBH.sub.4 Sodium borohydride NaH Sodium hydride NaHCO.sub.3 Sodium bicarbonate NaOH Sodium hydroxide NH.sub.2OH Hydroxylamine NH.sub.3 Ammonia NH.sub.4HCO.sub.3 Ammonium bicarbonate NiCl.sub.2.cndot.6H.sub.2O Nickel (II) chloride hexahydrate NMM 4-Methylmorpholine O.sub.2 (g) Oxygen (gas) Pd(dppf)Cl.sub.2 [1,1'- Bis(diphenylphosphino)ferrocene]dichloropalladium(II) PtO.sub.2 Platinum (IV) oxide RuPhos 2-Dicyclohexylphosphino-2',6'-diisopropoxybiphenyl RuPhos Pd G2 2nd Generation RuPhos Precatalyst, Chloro(2- dicyclohexylphosphino-2',6'-diisopropoxy-1,1'- biphenyl)[2-(2'-amino-1,1'-biphenyl)]palladium(II)
Example 1--Preparation of (R)--N-hydroxy-1'-methyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]- -5-carboxamide and (S)--N-hydroxy-1'-methyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]- -5-carboxamide (I-1 and I-2)
##STR00010##
[0383] Step-1: Methyl 1-oxo-2,3-dihydro-1H-indene-5-carboxylate
[0384] Into a 1-L round-bottom flask was placed a solution of 5-bromo-2,3-dihydro-1H-inden-1-one (30 g, 142 mmol, 1 equiv) in MeOH (450 mL), Pd(dppf)Cl.sub.2 (15.6 g, 21.32 mmol, 0.15 equiv) and Et.sub.3N (57.5 g, 568 mmol, 4 equiv). CO (g) was introduced to the flask. The reaction mixture was stirred for 24 h at 100.degree. C. in an oil bath, then cooled to room temperature. The solids were filtered and the filtrate was concentrated under vacuum. The residue was purified by normal phase chromatography on silica gel using EtOAc/petroleum ether (1:10). The collected fractions were concentrated under vacuum to afford 18 g (67% yield) of the title compound as a yellow solid. MS: (ES, m/z): 191 [M+H].sup.+.
Step-2: Dimethyl 1-oxo-2,3-dihydro-1H-indene-2,5-dicarboxylate
[0385] Into a 250-mL round-bottom flask was placed a solution of methyl 1-oxo-2,3-dihydro-1H-indene-5-carboxylate (10 g, 52.58 mmol, 1 equiv) in dimethyl carbonate (100 mL). NaH (60% in mineral oil, 4.2 g, 175 mmol, 2 equiv) was added. The resulting solution was stirred for 2 h at 80.degree. C. in an oil bath. The reaction mixture was cooled to room temperature and poured into ice water (50 mL). The solids were filtered and the filtrate was extracted with EtOAc (3.times.100 mL). The combined organic layers were dried over anhydrous Na.sub.2SO.sub.4, filtered and concentrated under vacuum. The residue purified by normal phase chromatography on silica gel using EtOAc/petroleum ether (1:10). The collected fractions were concentrated under vacuum to afford 5.8 g (44% yield) of the title compound as a yellow solid. MS: (ES, m/z): 249 [M+H].sup.+.
Step-3: Dimethyl 2-(cyanomethyl)-1-oxo-2,3-dihydro-1H-indene-2,5-dicarboxylate
[0386] Into a 1-L round-bottom flask was placed a solution of dimethyl 1-oxo-2,3-dihydro-1H-indene-2,5-dicarboxylate (11.8 g, 47.54 mmol, 1 equiv) in THF (500 mL), Et.sub.3N (24.0 g, 237.68 mmol, 5 equiv) and 2-bromoacetonitrile (17.1 g, 142.61 mmol, 3 equiv). The reaction was stirred for 16 h at 25.degree. C. The solids were filtered out and the filtrate was concentrated under vacuum. The residue was purified by normal phase chromatography on silica gel using EtOAc/petroleum ether (1:5). The collected fractions were concentrated under vacuum to afford 3.1 g (23% yield) of the title compound as a yellow solid. MS: (ES, m/z): 288 [M+H].sup.+.
Step-4: Dimethyl 2-(2-aminoethyl)-1-hydroxy-2,3-dihydro-1H-indene-2,5-dicarboxylate
[0387] Into a 500-mL round-bottom flask was placed dimethyl 2-(cyanomethyl)-1-oxo-2,3-dihydro-1H-indene-2,5-dicarboxylate (3 g, 10.44 mmol, 1 equiv), MeOH (100 mL), PtO.sub.2 (3 g) and AcOH (30 mL). H.sub.2 (g) was introduced into the flask. The resulting solution was stirred for 4 h at 25.degree. C. The reaction mixture was cooled to room temperature and the solids were filtered out. The filtrate was concentrated under vacuum to afford 2 g (crude) of the title compound as a yellow oil. MS: (ES, m/z): 294 [M+H].sup.+.
Step-5: Methyl 1-hydroxy-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxylate
[0388] Into a 250-mL round-bottom flask was placed dimethyl 2-(2-aminoethyl)-1-hydroxy-2,3-dihydro-H-indene-2,5-dicarboxylate (2 g, 6.82 mmol, 1 equiv) and NH.sub.3 (7 M in MeOH, 30 mL). The reaction was stirred for 1 h at 25.degree. C. The resulting mixture was concentrated under vacuum. The crude product was re-crystallized from CH.sub.2Cl.sub.2 to afford 500 mg (28% yield) of the title compound as a white solid. MS: (ES, m/z): 262 [M+H].sup.+.
Step-6: Methyl 2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxylate
[0389] Into a 100-mL round-bottom flask was placed a solution of methyl 1-hydroxy-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxylate (900 mg, 3.44 mmol, 1 equiv) in TFA (10 mL) and triethylsilane (10 mL). The reaction was stirred for 12 h at 80.degree. C. in an oil bath. The reaction mixture was cooled to room temperature and concentrated under vacuum. The residue was purified by normal phase chromatography on silica gel using an EtOAc/petroleum ether gradient. The collected fractions were concentrated under vacuum to afford 400 mg of the title compound as a white solid. MS: (ES, m/z): 246 [M+H].sup.+.
Step-7: Chiral separation of methyl (R)-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxylate and methyl (S)-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxylate
[0390] Racemic methyl 2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxylate was purified by Chiral-Prep-HPLC with the following conditions: Column: Phenomenex Lux.RTM. Cellulose-4 AXIA.TM. Packed, 250.times.21.2 mm, 5 .mu.m; Mobile Phase A: hexanes, Mobile Phase B: EtOH; Gradient: hold 30% B for 23 min, Detector: UV 254 nm and 220 nm. The first eluting isomer (Rt 2.46 min) was collected and concentrated under vacuum to afford 210 mg (25% yield) of a white solid which was assigned as the R isomer of methyl 2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxylate. MS: (ES, m/z): 246 [M+H].sup.+. The second eluting isomer (Rt 3.21 min) was collected and concentrated under vacuum to afford 200 mg (24% yield) of a white solid which was assigned as the S isomer of methyl 2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxylate. MS: (ES, m/z): 246 [M+H].sup.+.
Step-8: Methyl (R)-1'-methyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxyl- ate
[0391] Into an 8-mL vial was placed a solution of the first eluted isomer from Step 7, which was assigned as methyl (R)-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxylate as described above, (50 mg, 0.20 mmol, 1 equiv) in DMF (3 mL). The solution was cooled to 0.degree. C. NaH (60% in mineral oil, 16.3 mg, 0.41 mmol, 2 equiv), was added in portions at 0.degree. C. The resulting solution was stirred for 30 min at 0.degree. C. CH.sub.3I (57.8 mg, 0.41 mmol, 2 equiv) was added and the resulting solution was stirred for 2 h at 25.degree. C. The reaction mixture was poured into 10 mL of water and extracted with EtOAc (2.times.20 mL). The combined organic layers were dried over Na.sub.2SO.sub.4, filtered and concentrated under vacuum to afford 30 mg (51% yield) of the title compound as a yellow solid. MS: (ES, m/z): 260 [M+H].sup.+.
Step-9: (R)--N-Hydroxy-1'-methyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrro- lidine]-5-carboxamide
[0392] Into an 8-mL vial was placed methyl (R)-1'-methyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxyl- ate (30 mg, 0.12 mmol, 1 equiv), THF/MeOH (4:1, 2.5 mL), NH.sub.2OH (50% in water, 917 mg, 27.79 mmol, 120 equiv) and aq, 1N NaOH (0.232 mL, 0.24 mmol). The resulting solution was stirred for 2 h at 25.degree. C. The crude product was purified by Prep-HPLC with the following conditions: Column: XBridge Prep C18 OBD, 19.times.150 mm, 5 .mu.m; Mobile Phase A: Water/0.1% Formic acid, Mobile Phase B: CH.sub.3CN; Gradient: 5% B up to 20% B in 7 min; Detector: UV 254 nm, 220 nm. The collected fractions were lyophilized to afford 11.6 mg (39%, yield) of the title compound as a light pink solid. .sup.1H NMR (300 MHz, DMSO-d6) .delta. (ppm): 11.13 (s, 1H), 9.04 (s, 1H), 7.58-7.53 (t, J=7.4 Hz, 2H), 7.27-7.24 (d, J=7.8 Hz, 1H), 3.35-3.31 (t, J=6.6 Hz, 2H), 3.17-3.11 (d, J=16.2 Hz, 2H), 2.88-2.83 (d, J=16.2 Hz, 2H), 2.79 (s, 3H), 1.98-1.94 (t, J=6.6 Hz, 2H). MS: (ES, m/z): 261 [M+H]*.
Step-10: Methyl (S)-1'-methyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxyl- ate
[0393] The procedure from Step 8 was followed using the second eluted isomer from Step 7, which was assigned as methyl (S)-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxylate as described above, (50 mg, 0.20 mmol, 1 equiv) to afford 53 mg (94% yield) of the title compound as a light yellow oil. MS: (ES, m/z): 260 [M+H].sup.+.
Step-11: (S)--N-Hydroxy-1'-methyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrr- olidine]-5-carboxamide
[0394] The procedure from Step 9 was followed using methyl (S)-1'-methyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxyl- ate (53 mg, 0.20 mmol, 1 equiv) to afford 20.9 mg (39% yield) of the title compound as a white solid. .sup.1H NMR (300 MHz, DMSO-d6) .delta. (ppm): 11.11 (m, 1H), 8.98 (s, 1H), 7.59-7.54 (m, 2H), 7.28-7.25 (m, 1H), 3.36-3.32 (m, 2H), 3.18-3.16 (d, J=3.3 Hz, 1H), 3.12-3.11 (d, J=3.0 Hz, 1H), 2.89 (s, 1H), 2.84 (s, 1H), 2.80-2.75 (m, 3H), 1.99-1.95 (m, 2H). MS: (ES, m/z): 261 [M+H].sup.+.
TABLE-US-00002 TABLE 1 The following compounds were prepared according to the method of Example 1, with stereochemistry assigned as described, with the following modifications: (1) In Steps 8 and 10, the halide can be an iodide, a chloride, or a bromide; (2) In Steps 9 and 11, the Prep-HPLC column can be XBridge Prep C18 OBD, 19 .times. 150 mm, 5 .mu.m using formic acid or NH.sub.4HCO.sub.3 as the additive to the water Mobile Phase A; or the column XBridge Shield RP18 OBD, 19 .times. 150 mm, 5 .mu.m using formic acid as the additive to the water Mobile Phase A. Found (ES, m/z) Ex. Structure .sup.1H-NMR .delta. (ppm) [M + H].sup.+ I-3 ##STR00011## (400 MHz, DMSO-d6): 10.97 (s, 1H), 8.96 (s, 1H), 7.57 (s, 1H), 7.54 (d, J = 8.0 Hz, 1H), 7.23 (d, J = 16.8 Hz, 1H), 4.18-4.11 (m, 1H), 3.33-3.26 (m, 2H), 3.14-3.10 (d, J = 16.8 Hz, 2H), 2.87- 2.83 (d, J = 16.0 Hz, 2H), 1.94-19.1 (m, 2 H), 1.11-1.09 (m, 6H) 289 I-4 ##STR00012## (400 MHz, DMSO-d6): 11.09 (br, 1H), 8.96 (br, 1H), 7.58 (s, 1H), 7.54 (d, J = 7.6 Hz, 1H), 7.25 (d, J = 8.0 Hz, 1H), 4.19-4.11 (m, 1H), 3.30-3.26 (m, 2H), 3.13 (d, J = 16.0 Hz, 2H), 2.86 (d, J = 16.4 Hz, 2H), 1.95-1.92 (m, 2H), 1.15- 1.09 (m, 6H) 289 I-5 ##STR00013## (300 MHz, DMSO-d6): 8.55-8.41 (s, 2H), 7.58-7.53 (m, 2H), 7.26-7.24 (d, J = 8.0 Hz, 1H), 3.41-3.23 (m, 4H), 3.15- 3.10 (m 2H), 2.88-2.84 (d, J = 16.0 Hz, 2H), 1.96-1.93 (m, 2H), 1.07-1.03 (m, 3H) 275 I-6 ##STR00014## (400 MHz, DMSO-d6): 8.96 (br, 1H), 7.58 (s, 1H), 7.55 (d, J = 8.0 Hz, 1H), 7.25 (d, J = 8.0 Hz, 1H), 3.35-3.30 (m, 2H), 3.28-3.16 (m, 2H), 3.15-3.11 (m, 2H), 2.88-2.84 (m, 2H), 2.08-1.94 (m, 2H), 1.15-1.02 (m, 3H) 275 I-7 ##STR00015## (400 MHz, DMSO-d6): 11.06 (s, 1H), 8.98 (s, 1H), 7.58 (s, 1H), 7.54 (d, J = 7.6 Hz, 1H), 7.26 (d, J = 8.0 Hz, 1H), 3.34-3.31 (m, 2H), 3.16-3.12 (m, 2H), 3.05-3.04 (m, 2H), 2.90-2.86 (m, 2H), 1.91-1.87 (m, 3H), 0.85-0.83 (m, 6H) 303 I-8 ##STR00016## (400 MHz, DMSO-d6): 11.10 (br s, 1H), 8.98 (br s, 1H), 7.58 (s, 1H), 7.55 (d, J = 8.0 Hz, 1H), 7.26 (d, J = 7.6 Hz, 1H), 3.35 (s, 1H), 3.32 (s, 1H), 3.15 (d, J = 16.0 Hz, 2H), 3.06-3.04 (m, 2H), 2.89 (d, J = 12.0 Hz, 2H), 1.98-1.88 (m, 3H), 0.85-0.81 (m, 6H) 303 I-9 ##STR00017## (400 MHz, DMSO-d6): 11.02 (s, 1H), 9.02 (s, 1H), 7.59 (s, 1H), 7.55 (d, J = 8.0 Hz, 1H), 7.27 (d, J = 8.0 Hz, 1H), 7.21-7.12 (m, 4H), 4.44 (s, 2H), 3.22- 3.16 (m, 4H), 2.93-2.89 (m, 2H), 2.25 (s, 3H), 1.97-1.95 (m, 2H) 351 I-10 ##STR00018## (400 MHz, DMSO-d6): 11.12 (br, 1H), 8.97 (br, 1H) 7.60 (s, 1H), 7.55 (d, J = 8.0 Hz, 1H), 7.28 (d, J = 8.0 Hz, 1H), 7.21-7.15 (m, 3H), 7.13 (d, J = 3.6 Hz, 1H), 4.44 (s, 2H), 3.22-3.16 (m, 4H), 2.92 (d, J = 16.0 Hz, 1H), 2.32 (s, 3H), 1.99-1.96 (m, 2H) 351 I-12 ##STR00019## (300 MHz, DMSO-d6): 11.12 (s, 1H), 8.98 (s, 1H), 7.61-7.58 (m, 1H), 7.57- 7.55 (m, 1H), 7.41-7.37 (m, 2H), 7.57 7.31 (m, 1H), 7.30-7.23 (m, 3H), 4.45 (s, 2H), 3.27-3.23 (m, 2H), 3.19 (s, 1H), 2.95 (s, 1H), 2.90 (s, 1H), 2.08- 1.97 (m, 2H) 337
Example 2--Preparation of (R)-1'-benzyl-N-hydroxy-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-- 5-carboxamide (I-11)
##STR00020##
[0395] Step-1: (R)-1'-Benzyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxyl- ic acid
[0396] Into an 8-mL vial was placed a solution of the first eluted isomer from Example 1, Step 7, which was assigned as methyl (R)-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxylate as described above, (50 mg, 0.20 mmol, 1 equiv) in DMF (3 mL). The reaction was cooled to 0.degree. C. and NaH (60% in mineral oil, 16.3 mg, 0.41 mmol) was added portionwise at 0.degree. C. The resulting solution was stirred for 30 min at 0.degree. C. (Bromomethyl)benzene (69.7 mg, 0.41 mmol) was added at 0.degree. C. The reaction was stirred for 2 h at 25.degree. C. The reaction was then poured into 10 mL water. The solution was adjusted to pH 2 with 2N HCl and extracted with EtOAc (2.times.10 mL). The organic layers were combined, dried over Na.sub.2SO.sub.4, filtered and concentrated under vacuum to afford 40 mg (49% yield) of the title compound as a yellow oil. MS: (ES, m/z): 322 [M+H].sup.+.
Step-2: (R)-1'-Benzyl-N-hydroxy-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrol- idine]-5-carboxamide
[0397] Into an 8-mL vial was placed (R)-1'-benzyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxyl- ic acid (40 mg, 0.12 mmol, 1 equiv) in DMA (3 mL). This was followed by the addition of isopropyl chloroformate (78.7 mg, 0.64 mmol, 5 equiv) and NMM (63.1 mg, 0.62 mmol, 5 equiv) dropwise with stirring at 0.degree. C. The resulting solution was stirred for 30 min at 0.degree. C. To the mixture was added a solution of NH.sub.2OH.HCl (43.7 mg, 0.62 mmol, 5 equiv) in DMA (0.5 mL). The reaction was stirred for 16 h at 25.degree. C. The solids were filtered out and the crude product was purified by Prep-HPLC with the following conditions: Column: XBridge Prep C18 OBD, 19.times.150 mm, 5 .mu.m; Mobile Phase A: Water/0.1% Formic acid. Mobile Phase B: CH.sub.3CN; Gradient; 25% B up to 55% B in 7 min; Detector: UV 254 nm, 220 nm. The collected fractions were lyophilized to afford 16.9 mg (40% yield) of the title compound as an off-white solid. .sup.1H NMR (400 MHz, DMSO-d6) .delta. (ppm): 11.10 (s, 1H), 8.98 (s, 1H), 7.60-7.54 (m, 2H), 7.40-7.23 (m, 6H), 4.44 (s, 2H), 3.25-3.18 (m, 4H), 2.93-2.89 (d, J=12 Hz, 2H), 1.99-1.92 (m, 2H). MS: (ES, m/z): 337 [M+H].sup.+.
Example 3--Preparation of (R)--N-hydroxy-2'-oxo-1'-phenyl-1,3-dihydrospiro[indene-2,3'-pyrrolidine]- -5-carboxamide (I-13)
##STR00021##
[0398] Step-1: Methyl (R)-2'-oxo-1'-phenyl-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxyl- ate
[0399] Into a 100-mL round-bottom flask was placed a solution of the first eluted isomer from Example 1, Step 7, which was assigned as methyl (R)-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxylate as described above, (50 mg, 0.20 mmol, 1 equiv) in CH.sub.2Cl.sub.2 (10 mL). Cu(OAc).sub.2 (69.8 mg, 0.380 mmol, 2 equiv), Et.sub.3N (58.2 mg, 0.58 mmol, 3 equiv), phenylboronic acid (117 mg, 0.96 mmol, 5 equiv) and 4 .ANG. molecular sieves (100 mg) were added. O.sub.2 (g) was introduced to the reaction mixture. The resulting solution was stirred for 60 h at 25.degree. C. The solids were filtered out and the filtrate was diluted with 10 mL of water and extracted with CH.sub.2Cl.sub.2 (3.times.10 mL). The combined organic layers were dried over Na.sub.2SO.sub.4 and concentrated under vacuum to afford 20 mg (29% yield) of the title compound as an orange oil. MS: (ES, m/z): 322 [M+H].sup.+.
Step-2: (R)--N-Hydroxy-2'-oxo-1'-phenyl-1,3-dihydrospiro[indene-2,3'-pyrro- lidine]-5-carboxamide
[0400] Into an 8-mL vial was placed a solution of methyl (R)-2'-oxo-1'-phenyl-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxyl- ate (20 mg, 0.06 mmol, 1 equiv) in THF/MeOH (4:1, 2.5 mL). Then NH.sub.2OH (50% in water, 492 mg, 7.45 mmol, 120 equiv) was added followed by aq. 1N NaOH (0.124 mL, 2 equiv). The resulting solution was stirred for 2 h at 25.degree. C. The crude product was purified by Prep-HPLC with the following conditions: Column: XBridge Prep C18 OBD, 19.times.150 mm, 5 .mu.m; Mobile Phase A: Water/0.1% Formic acid, Mobile Phase B: CH.sub.3CN; Gradient: 5% B up to 57% B in 7 min; Detector: UV 254 nm, 220 nm. The collected fractions were lyophilized to afford 6.7 mg (33% yield) of the title compound as a white solid. .sup.1H NMR (300 MHz, DMSO-d6) .delta. (ppm): 10.88 (s, 1H), 9.00 (s, 1H), 7.73-7.71 (d, J=7.8 Hz, 2H), 7.61-7.56 (t, J=8.4 Hz, 6H), 7.42-7.37 (t, J=8.0 Hz, 2H), 7.30-7.27 (d, J=7.8 Hz, 1H), 7.18-7.13 (t, 0.1=7.4 Hz, 1H), 3.89-3.84 (t, J=6.8 Hz, 2H), 3.24 (s, 2H), 3.05-2.99 (d, 0.1=16.2 Hz, 2H), 2.15-2.07 (m, 2H). MS: (ES, m/z): 323 [M+H].sup.+.
Example 4--Preparation of (S)--N-hydroxy-2'-oxo-1'-phenyl-1,3-dihydrospiro[indene-2,3'-pyrrolidine]- -5-carboxamide (I-14)
##STR00022##
[0401] Step-1: Methyl (S)-2'-oxo-1'-phenyl-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxyl- ate
[0402] Into a 25-mL round-bottom flask was placed a solution of the second eluted isomer from Example 1, Step 7, which was assigned as methyl (S)-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxylate as described above, (50 mg, 0.20 mmol, 1 equiv) in THF (8.0 mL). Cu(OAc).sub.2 (37 mg, 0.20 mmol, 1 equiv), Et.sub.3N (62 mg, 0.61 mmol, 3 equiv), pyridine (24 mg, 0.31 mmol, 1.5 equiv), phenylboronic acid (49.7 mg, 0.41 mmol, 2 equiv) and 4 .ANG. molecular sieves were added. O.sub.2 (g) was introduced in. The resulting solution was stirred for 18 h at 60.degree. C. in an oil bath. The solids were filtered out and the filtrate was concentrated under vacuum. The residue was dissolved in 60 mL of EtOAc and was washed with 3.times.20 mL of H.sub.2O. The organic layer was separated and concentrated under vacuum. The residue purified by normal phase chromatography on silica gel using EtOAc/petroleum ether (1:2). The collected fractions were concentrated under vacuum to afford 60 mg (92%) of the title compound as a light yellow oil. MS: (ES, m/z): 322 [M+H].sup.+.
Step-2: (S)--N-Hydroxy-2'-oxo-1'-phenyl-1,3-dihydrospiro[indene-2,3'-pyrro- lidine]-5-carboxamide
[0403] Into an 8-mL vial was placed a solution of methyl (S)-2'-oxo-1'-phenyl-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxyl- ate (60 mg, 0.19 mmol, 1 equiv) in THF/MeOH (4:1, 2.5 mL). Then NH.sub.2OH (50% in water, 274 mg, 5.37 mmol, 30 equiv) was added followed by aq 1N NaOH (0.37 mL, 2 equiv). The resulting solution was stirred for 1 h at 25.degree. C. The crude product was purified by Prep-HPLC with the following conditions: Column: XBridge Prep C18 OBD, 19.times.150 mm, 5 .mu.m; Mobile Phase A: Water/0.1% Formic acid, Mobile Phase B: CH.sub.3CN; Gradient: 5% B up to 57% B in 7 min; Detector: UV 254 nm, 220 nm. The collected fractions were lyophilized to afford 24.3 mg (40% yield) of the title compound as an off-white solid. .sup.1H NMR (300 MHz, DMSO-d6) .delta. (ppm): 11.14 (s, 1H), 9.00 (s, 1H), 7.75-7.72 (m, 2H), 7.63-7.57 (m, 2H), 7.44-7.38 (m, 2H), 7.32-7.29 (m, 1H), 7.19-7.14 (m, 1H), 3.90-3.86 (m, 2H), 3.34-3.25 (m, 2H), 3.06-3.01 (d, 0.1=16.2 Hz, 2H), 2.16-2.08 (m, 2H). MS: (ES, m/z): 323 [M+H].sup.+.
TABLE-US-00003 TABLE 2 The following compounds were prepared according to the method of Example 4, with stereochemistry assigned as described, with the following modifications: In Prep-HPLC purification of Step 2, formic acid or NH.sub.4HCO.sub.3 can be used as the additive to the water Mobile Phase A. Found (ES, m/z) Ex. Structure .sup.1H-NMR .delta. (ppm) [M + H].sup.+ I-15 ##STR00023## (400 MHz, DMSO-d6): 10.71 (s, 1H), 8.95 (s, 1H), 7.74-7.71 (m, 2H), 7.60- 7.54 (m, 2H), 7.28-7.20 (m, 3H), 3.85- 3.82 (m, 2H), 3.30-63.22 (m, 2H), 3.02- 2.98 (m, 2H), 2.12-2.09 (m, 2H) 341 I-16 ##STR00024## (400 MHz, DMSO-d6): 11.10 (br s, 1H), 8.98 (br s, 1H), 7.77-7.73 (m, 2H), 7.62 (s, 1H), 7.57 (d, J = 7.6 Hz 1H), 7.29 (d, J = 8.0 Hz, 1H), 7.27-7.22 (m, 2H), 3.88-3.84 (m, 2H), 3.29-3.23 (m, 2H), 3.04-3.00 (m, 2H), 2.14-2.11 (m, 2H) 341 I-18 ##STR00025## (400 MHz, DMSO-d6): 8.95-8.94 (m, 2H), 8.37-8.36 (m, 1H), 8.17-8.15 (m, 1H), 7.62 (s, 1H), 7.58 (d, J = 7.6 Hz, 1H), 7.46-7.42 (m, 1H), 7.30 (d, J = 8.0 Hz, 1H), 3.93-3.90 (m, 2H), 3.32-3.29 (m, 1H), 3.26-3.25 (m, 1H), 3.07-3.02 (m, 2H), 2.19-2.07 (m, 2H) 324
Example 5--Preparation of (R)--N-hydroxy-2'-oxo-1'-(pyridin-3-yl)-1,3-dihydrospiro[indene-2,3'-pyrr- olidine]-5-carboxamide (I-17)
##STR00026##
[0404] Step-1: Methyl (R)-2'-oxo-1'-(pyridin-3-yl)-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-- carboxylate
[0405] Into a 10-mL sealed tube was placed a solution of the first eluted isomer from Example 1, Step 7, which was assigned as methyl (R)-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxylate as described above, (60 mg, 0.24 mmol, 1 equiv) in toluene (5 mL), 3-bromopyridine (77 mg, 0.49 mmol, 2 equiv), RuPhos (23 mg, 0.05 mmol), Cs.sub.2CO.sub.3 (240 mg, 0.74 mmol, 3 equiv) and RuPhos Pd G2 (19 mg, 0.02 mmol, 0.1 equiv). The reaction was stirred for 16 h at 110.degree. C. The reaction mixture was then cooled to room temperature and poured into 15 mL of water. The resulting solution was extracted with 3.times.30 mL of EtOAc. The combined organic layers were washed with 100 mL of brine, dried over Na.sub.2SO.sub.4, filtered and concentrated under vacuum. The residue was purified by normal phase chromatography on silica gel using EtOAc/petroleum ether (1:1). The collected fractions were concentrated under vacuum to afford 51 mg (60% yield) of the title compound as a yellow oil. MS: (ES, m/z): 323 [M+H].sup.+.
Step-2: (R)--N-Hydroxy-2'-oxo-1'-(pyridin-3-yl)-1,3-dihydrospiro[indene-2,- 3'-pyrrolidine]-5-carboxamide
[0406] Into a 25-mL round-bottom flask was placed methyl (R)-2'-oxo-1'-(pyridin-3-yl)-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-- carboxylate (51 mg, 0.16 mmol, 1 equiv), THF/MeOH (4:1, 4 mL), NH.sub.2OH (50% in water, 1426 mg, 43.17 mmol, 120 equiv) and aq. 1N NaOH (0.32 mL, 0.32 mmol, 2 equiv). The resulting solution was stirred for 2 h at room temperature. The solids were filtered out and the crude product was purified by Prep-HPLC with the following conditions: Column: XBridge Prep C18 OBD, 19.times.150 mm, 5 .mu.m; Mobile Phase A: Water/0.1% Formic acid, Mobile Phase B: CH.sub.3CN, Gradient: 5% B up to 14% B in 8 min, Detector: UV 254 nm, 220 nm. The collected fractions were lyophilized to afford 20.1 mg (39% yield) of the title compound as an off-white solid. .sup.1H NMR (300 MHz, DMSO-d6) .delta. (ppm): 8.94 (s, 1H), 8.37 (d, J=3.9 Hz, 1H), 8.17 (d, J=20.4 Hz, 1H), 7.61-7.56 (m, 2H), 7.50-7.46 (m, 1H), 7.34 (d, J=7.8 Hz, 1H), 3.93 (d, J=6.6 Hz, 2H), 3.29 (d, J=16.5 Hz, 2H), 3.07 (d, J=16.2 Hz, 2H), 2.20-2.16 (m, 2H). MS: (ES, m/z): 324 [M+H].sup.+.
TABLE-US-00004 TABLE 3 The following compounds were prepared according to the method of Example 5, with stereochemistry assigned as described, with the following modifications: In Prep-HPLC purification of Step 2, formic acid or NH.sub.4HCO.sub.3 can be used as the additive to the water Mobile Phase A. Found (ES, m/z) Ex. Structure .sup.1H-NMR .delta. (ppm) [M + H].sup.+ I-19 ##STR00027## (300 MHz, DMSO-d6): 11.11 (s, 1H), 8.98 (s, 1H), 8.43-8.41 (m, 1H), 8.33 (d, J = 8.4 Hz, 1H), 7.86-7.81 (m, 1H), 7.62-7.57 (m, 2H), 7.30 (d, J = 7.8 Hz, 1H), 7.19-7.15 (m, 1H), 4.05-4.00 (m, 2H), 3.30 (d, J = 17.7 Hz, 2H), 3.08 (d, J = 16.5 Hz, 2H), 2.14-2.08 (m, 2H) 324 I-20 ##STR00028## (400 MHz, DMSO-d6): 8.43-8.41 (m, 1H), 8.34-8.32 (m, 1H), 7.86-7.81 (m, 1H), 7.62 (s, 1H), 7.58 (d, J = 8.0 Hz, 1H), 7.29 (d, J = 8.0 Hz, 1H), 7.19- 7.15 (m, 1H), 4.04-4.01 (m, 2H), 3.32-3.27 (m, 2H), 3.07-3.03 (m, 2H), 2.14-2.07 (m, 2H) 324 I-21 ##STR00029## (300 MHz, DMSO-d6): 11.11 (s, 1H), 9.00 (d, J = 11.4 Hz, 1H), 8.52 (d, J = 6.3 Hz, 2H), 7.75-7.73 (m, 2H), 7.62- 7.57 (m, 2H), 7.30 (d, J = 7.8 Hz, 1H), 3.91-3.86 (m, 2H), 3.32-3.30 (m, 1H), 3.25 (s, 1H), 3.08 (s, 1H), 3.02 (s, 1H), 2.18-2.07 (m, 2H) 324 I-22 ##STR00030## (400 MHz, DMSO-d6): 11.12 (br s, 1H), 8.96 (br s, 1H), 8.53-8.51 (m, 2H), 7.75-7.73 (m, 2H), 7.62 (s, 1H), 7.58 (d, J = 8.0 Hz, 1H),7.29 (d, J = 8.0 Hz, 1H), 3.90-3.87 (m, 2H), 3.29-3.25 (m, 2H), 3.07-3.03 (m, 2H), 2.18-2.07 (m, 2H) 324
Example 6--Preparation of (R)--N-hydroxy-2'-oxo-1'-(pyridin-3-ylmethyl)-1,3-dihydrospiro[indene-2,3- '-pyrrolidine]-5-carboxamide (I-23)
##STR00031##
[0407] Step-1: Methyl (R)-2'-oxo-1'-(pyridin-3-ylmethyl)-1,3-dihydrospiro[indene-2,3'-pyrrolidi- ne]-5-carboxylate
[0408] Into a 25-mL round-bottom flask was placed a solution of the first eluted isomer from Example 1, Step 7, which was assigned as methyl (R)-2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxylate as described above, (50 mg, 0.20 mmol, 1 equiv) in DMF (3 mL). NaH (60% in mineral oil, 16 mg, 0.40 mmol, 2 equiv) was then added at 0.degree. C. The resulting solution was stirred for 30 min at 0.degree. C. A solution of 3-(bromomethyl) pyridine hydrobromide (77 mg, 0.30 mmol, 1.5 equiv) and Et.sub.3N (61 mg, 0.60 mmol, 3 equiv) in DMF (1 mL) were added dropwise with stirring at 0.degree. C. The resulting solution was allowed to stir for 1 h at room temperature. The reaction mixture was then poured into 15 mL of ice water and extracted with 3.times.15 mL of EtOAc. The combined organic layers were dried over Na.sub.2SO.sub.4, filtered and concentrated under vacuum to afford 55 mg of the title compound as a yellow oil. MS: (ES, m/z): 337 [M+H].sup.+.
Step-2: (R)--N-Hydroxy-2'-oxo-1'-(pyridin-3-ylmethyl)-1,3-dihydrospiro[ind- ene-2,3'-pyrrolidine]-5-carboxamide
[0409] Into a 25-mL round-bottom flask was placed methyl (R)-2'-oxo-1'-(pyridin-3-ylmethyl)-1,3-dihydrospiro[indene-2,3'-pyrrolidi- ne]-5-carboxylate (55 mg, 0.16 mmol, 1 equiv), THF/MeOH (4:1, 3.0 mL), NH.sub.2OH (50% in water, 648 mg, 9.81 mmol, 60 equiv) and aq. 1N NaOH (0.32 mL, 0.32 mmol, 2 equiv). The resulting solution was stirred for 2 h at 25.degree. C. The solids were filtered and the crude product was purified by Prep-HPLC with the following conditions: Column: XBridge Prep C18 OBD, 19.times.150 mm, 5 .mu.m; Mobile Phase A: Water/0.1% Formic acid, Mobile Phase B: CH.sub.3CN; Gradient: 3% B up to 23% B in 7 min; Detector: UV 254 nm, 220 nm. The collected fractions were lyophilized to afford 19.6 mg (36% yield) of the title compound as a light pink solid. .sup.1H NMR (400 MHz, DMSO-d6) .delta. (ppm): 11.05 (s, 1H), 8.95 (s, 1H), 8.51-8.47 (m, 2H), 7.64-7.52 (m, 3H), 7.40-7.39 (m, 1H), 7.37-7.24 (m, 1H), 4.46 (s, 2H), 3.27-3.24 (m, 2H), 3.19-3.15 (d, 0.1=16.4 Hz, 2H), 2.92-2.88 (d, J=16.4 Hz, 2H), 1.99-1.95 (m, 2H) MS: (ES, m/z); 338 [M+H].sup.+.
Example 7--Preparation of (R)--N-hydroxy-1'-methyl-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carb- oxamide and (S)--N-hydroxy-1'-methyl-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carb- oxamide (I-24 and I-25)
##STR00032##
[0410] Step-1: Methyl 2'-thioxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxylate
[0411] Into a 100-mL round-bottom flask was placed methyl 2'-oxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxylate (2.4 g, 9.78 mmol, 1 equiv), CH.sub.2Cl.sub.2 (30 mL) and 2,4-bis(4-methoxyphenyl)-2,4-dithioxo-, 3,2,4-dithiadiphosphetane (Lawesson reagent) (5.9 g, 14.59 mmol, 1.5 equiv). The resulting solution was stirred for 16 h at room temperature. The reaction was then quenched by the addition of aq NaHCO.sub.3 (30 mL). The resulting solution was extracted with 3.times.30 mL of CH.sub.2Cl.sub.2. The organic layers were combined, dried over Na.sub.2SO.sub.4, filtered and concentrated under vacuum. The residue was purified by normal phase column chromatography on silica gel using EtOAc/petroleum ether (1:1). The collected fractions were concentrated under vacuum to afford 2 g (78% yield) of the title compound as a yellow solid. MS: (ES, m/z): 262 [M+H].sup.+.
Step-2: Methyl 1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxylate
[0412] Into a 100-mL 3-necked round-bottom flask purged and maintained with an inert atmosphere of nitrogen, was placed methyl 2'-thioxo-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxylate (2 g, 7.65 mmol, 1 equiv), THF (40 mL), MeOH (10 mL) and NiCl.sub.2.6H.sub.2O (10.9 g, 45.86 mmol, 6 equiv). This was followed by the addition of NaBH.sub.4 (1.74 g, 45.99 mmol, 6 equiv) in several batches at 0.degree. C. The resulting solution was stirred for 18 h at room temperature. The reaction was concentrated under vacuum. The residue was dissolved in 10 mL of THF and filtered through a Celite pad. The filtrate was concentrated under reduced pressure and the residue was purified by normal phase column chromatography on silica gel using CH.sub.2Cl.sub.2/MeOH (10:1). The collected fractions were concentrated under vacuum to afford 1.4 g (79% yield) of the title compound as a green solid. MS: (ES, m/z): 232 [M+H].sup.+.
Step-3: 1'-(tert-Butyl) 5-methyl (R)-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-1',5-dicarboxylate and 1'-(tert-Butyl) 5-methyl (S)-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-1',5-dicarboxylate
[0413] Into a 50-mL round-bottom flask was placed methyl 1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxylate (1.4 g, 6.05 mmol, 1 equiv), CH.sub.2Cl.sub.2 (15 mL), Et.sub.3N (2.4 g, 23.72 mmol, 4 equiv) and di-tert-butyl dicarbonate (3.3 g, 15.12 mmol, 2.5 equiv). The resulting solution was stirred for 18 h at room temperature. The reaction was then quenched by the addition of 20 mL of water. The resulting solution was extracted with 3.times.20 mL of CH.sub.2C.sub.12. The combined organic layers were dried over Na.sub.2SO.sub.4, filtered, concentrated under vacuum, and the residue was purified by normal phase column chromatography on silica gel using EtOAc/petroleum ether (1:10) to afford the racemic mixture of the title compounds. The racemate was further separated by Prep-SFC with the following conditions: Column: EnantioCel-C1, 21.2.times.250 mm; Mobile Phase A: CO.sub.2, Mobile Phase B: EtOH; Gradient 20% B; Flow rate: 40 mL/min; Detector: UV 220 nm. The first eluting isomer (Rt 4.91 min) was collected and concentrated under vacuum to give 400 mg (20% yield) of a white solid which was assigned as the R isomer of 1'-(tert-butyl) 5-methyl 1,3-dihydrospiro[indene-2,3'-pyrrolidine]-1',5-dicarboxylate. MS: (ES, m/z): 332 [M+H].sup.+. The second eluting isomer (Rt 5.76 min) was collected and concentrated under vacuum to give 400 mg (20% yield) of a white solid which was assigned as the S isomer of 1'-(tert-butyl) 5-methyl 1,3-dihydrospiro[indene-2,3'-pyrrolidine]-1',5-dicarboxylate. MS: (ES, m/z): 332 [M+H].sup.+.
Step-4: Methyl (R)-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxylate
[0414] Into a 25-mL round-bottom flask was placed the first eluted isomer from Step 3, which was assigned as 1'-(tert-butyl) 5-methyl (R)-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-1',5-dicarboxylate as described above, (400 mg, 1.21 mmol, 1 equiv), CH.sub.2Cl.sub.2 (8 mL) and TFA (2 mL). The reaction was stirred for 2 h at room temperature. The reaction was concentrated under vacuum. The residue was dissolved in water (10 mL) and the pH was adjusted to 8 with aq. NaHCO.sub.3. The resulting solution was extracted with 3.times.15 mL of CH.sub.2Cl.sub.2. The combined organic layers were dried over Na.sub.2SO.sub.4, filtered and concentrated under vacuum to afford 270 mg (97% yield) of the title compound as a yellow oil. MS: (ES, m/z): 232 [M+H].
Step-5: Methyl (R)-1'-methyl-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxylate
[0415] Into a 10-mL round-bottom flask was placed methyl (R)-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxylate (50 mg, 0.22 mmol, 1 equiv), MeOH (3 mL) and paraformaldehyde (7.8 mg, 0.26 mmol, 1.2 equiv). The resulting solution was stirred for 1 h at room temperature. This was followed by the addition of NaBH.sub.3CN (42 mg, 0.67 mmol, 3 equiv) in several batches at 0.degree. C. The reaction was allowed to stir for 18 h at room temperature. The reaction was poured into 15 mL of water. The resulting solution was extracted with 3.times.15 mL of EtOAc. The combined organic layers were dried over Na.sub.2SO.sub.4, filtered, and concentrated under vacuum. The residue was purified by normal phase column chromatography on silica gel using EtOAc/petroleum ether (1:2). The collected fractions were concentrated under vacuum to afford 50 mg (94% yield) of the title compound as a yellow oil. MS: (ES, m/z): 246 [M+H].sup.+.
Step-6: (R)--N-Hydroxy-1'-methyl-1,3-dihydrospiro[indene-2,3'-pyrrolidine]- -5-carboxamide
[0416] Into a 10-mL round-bottom flask was placed methyl (R)-1'-methyl-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxylate (50 mg, 0.20 mmol, 1 equiv), THF/MeOH (4:1, 5.0 mL), NH.sub.2OH (50% in water, 1.58 g, 24 mmol, 120 equiv) and aq. 1N NaOH (16.3 mg, 0.41 mmol, 2 equiv). The resulting solution was stirred for 2 h at room temperature. The solids were filtered and the crude product was purified by Prep-HPLC with the following conditions: Column: XBridge Shield RP18 OBD, 19.times.150 mm, 5 .mu.m, Mobile Phase A: Water/NH.sub.4HCO.sub.3, Mobile Phase B: CH.sub.3CN; Gradient: 0% B up to 18% B in 7 min; Detector: UV 220 nm, 254 nm. The collected fractions were lyophilized to afford 7.9 mg (16% yield) of the title compound as a white solid. .sup.1H NMR (400 MHz, DMSO-d6) .delta. (ppm): 7.55 (s, 1H), 7.50 (d, J=8 Hz, 1H), 7.21 (d, J=7.6 Hz, 1H), 2.97-2.84 (m, 2H), 2.55-2.51 (m, 2H), 2.40 (s, 2H), 2.23 (s, 3H), 1.81-1.77 (m, 2H). MS: (ES, m/z): 247 [M+H].sup.+.
Step-7: Methyl (S)-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxylate
[0417] The procedure from Step 4 was followed using the second eluted isomer from Step 3, which was assigned as 1'-(tert-butyl) 5-methyl (S)-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-1',5-dicarboxylate as described above, (470 mg, 1.42 mmol, 1 equiv) to afford 230 mg (70% yield) of the title compound as a yellow oil. MS: (ES, m/z): 232 [M+H].sup.+.
Step-8: Methyl (S)-1'-methyl-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxylate
[0418] The procedure from Step 5 was followed using methyl (S)-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxylate (50 mg, 0.22 mmol, 1 equiv) to afford 27 mg (51% yield) of the title compound as a yellow oil. MS: (ES, m/z): 246 [M+H].sup.+.
Step-9: (S)--N-Hydroxy-1'-methyl-1,3-dihydrospiro[indene-2,3'-pyrrolidine]- -5-carboxamide
[0419] The procedure from Step 6 was followed using methyl (S)-1'-methyl-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxylate (27 mg, 0.11 mmol, 1 equiv) to afford 9.2 mg (34% yield) of the title compound as a white solid. .sup.1H NMR (400 MHz, DMSO-d6) .delta. (ppm); 7.55 (s, 1H), 7.51 (d, J=7.6 Hz, 1H), 7.23 (d, J=7.6 Hz, 1H), 2.95 (d, J=16.0 Hz, 2H), 2.86 (d, J=16.0 Hz, 2H), 2.56-2.51 (m, 2H), 2.42-2.39 (m, 2H), 2.23-2.21 (m, 3H), 1.80-1.77 (m, 2H). MS: (ES, m/z): 247 [M+H].sup.+.
TABLE-US-00005 TABLE 4 The following compounds were prepared according to the method of Example 7, with stereochemistry assigned as described, with the following modifications: In Prep-HPLC purification of Steps 6 and 9, formic acid or NH.sub.4HCO.sub.3 can be used as the additive to the water Mobile Phase A. Found (ES, m/z) Ex. Structure .sup.1H-NMR .delta. (ppm) [M + H].sup.+ I-26 ##STR00033## (400 MHz, DMSO-d6): 11.08 (s, 1H), 8.19 (s, 1H), 7.55-7.49 (m, 2H), 7.32 (d, J = 4.2 Hz, 4H), 7.29-7.22 (m, 2H), 3.63 (s, 2H), 2.99-2.86 (m, 4H), 2.69- 2.65 (m, 2H), 2.51 (s, 2H), 1.86-1.81 (m, 2H) 323 I-27 ##STR00034## (400 MHz, DMSO-d6): 11.07 (s, 1H), 8.18 (s, 1H), 7.54-7.48 (m, 2H), 7.32- 7.28 (m, 4H), 7.25-7.15 (m, 2H), 3.64 3.60 (m, 2H), 2.95 (d, J = 16.0 Hz, 2H), 2.88 (d, J = 16.0 Hz, 2H), 2.69-2.63 (m, 2H), 2.49-2.46 (m, 2H), 1.84-1.80 (m, 2H) 323
Example 8--Preparation of (S)-1'-acetyl-N-hydroxy-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carbo- xamide (I-29)
##STR00035##
[0420] Step-1: Methyl (S)-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxylate
[0421] Into a 50-mL round-bottom flask was placed the second eluted isomer from Example 7 Step 3, which was assigned as 1'-(tert-butyl) 5-methyl (S)-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-1',5-dicarboxylate as described above, (470 mg, 1.42 mmol, 1 equiv), TFA (5 mL) and CH.sub.2Cl.sub.2 (15 mL). The resulting solution was stirred for 2 h at room temperature. The resulting mixture was washed with 3.times.30 mL of aq NaHCO.sub.3, dried over Na.sub.2SO.sub.4, filtered and concentrated under vacuum to afford 230 mg (70% yield) of the title compound as a yellow oil. MS: (ES, m/z): 232 [M+H].sup.+.
Step-2: Methyl (S)-1'-acetyl-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxylate
[0422] Into a 10-mL vial was placed methyl (S)-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxylate (50 mg, 0.22 mmol, 1 equiv) and CH.sub.2C.sub.2 (5 mL). This was followed by the addition of Et.sub.3N (109 mg, 1.08 mmol, 5 equiv) dropwise at 0.degree. C. Acetyl chloride (51 mg, 0.65 mmol, 3 equiv) was added dropwise at 0.degree. C. The resulting solution was stirred for 2 h at room temperature. The reaction mixture was poured into 20 mL of ice water and extracted with 3.times.30 mL of EtOAc. The combined organic layers were dried over Na.sub.2SO.sub.4, filtered and concentrated under vacuum to afford 52 mg of the title compound as a yellow oil. MS: (ES, m/z): 274 [M+H].sup.+.
Step-3: (S)-1'-Acetyl-N-hydroxy-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-- 5-carboxamide
[0423] Into a 25-mL round-bottom flask was placed methyl (S)-1'-acetyl-1,3-dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxylate (62 mg, 0.23 mmol, 1 equiv), THF/MeOH (4:1, 2.0 mL), NH.sub.2OH (50.degree. % in water, 896 mg, 13.56 mmol, 60 equiv) and aq. 1N NaOH (0.45 mL, 0.45 mmol, 2 equiv). The resulting solution was stirred for 2 h at room temperature. The solids were filtered and the crude product was purified by Prep-HPLC with the following conditions; Column: XBridge Prep C18 OBD, 19.times.150 mm, 5 .mu.m; Mobile Phase A: Water/0.1% Formic acid, Mobile Phase B: CH.sub.3CN; Gradient: 5% B up to 30% B in 7 min; Detector: UV 254 nm, 220 nm. The collected fractions were lyophilized to afford 13.6 mg (22% yield) of the title compound as a yellow solid. .sup.1H NMR (300 MHz, DMSO-d6) .delta. (ppm): 11.07 (s, 1H), 9.09 (s, 1H), 7.59-7.54 (m, 2H), 7.27-7.24 (m, 1H), 3.56-3.52 (m, 1H), 3.41-3.26 (m, 3H), 2.98-2.86 (m, 4H), 1.94-1.79 (m, 5H). MS: (ES, m/z): 275 [M+H].sup.+.
TABLE-US-00006 TABLE 5 The following compounds were prepared according to the method of Example 8, with stereochemistry assigned as described, with the following modifications: In Step 3 the Prep-HPLC column can be XBridge Prep C18 OBD, 19 .times. 150 mm, 5 .mu.m using formic acid or NH.sub.4HCO.sub.3 as the additive to OBD, the water Mobile Phase A; or the column XBridge Shield RP18 19 .times. 150 mm, 5 .mu.m using formic acid as the additive to the water Mobile Phase A. Found (ES, m/z) Ex. Structure .sup.1H-NMR .delta. (ppm) [M + H].sup.+ I-28 ##STR00036## (400 MHz, DMSO-d6): 11.11 (br s, 1H), 8.99) (s, 1H), 7.60-7.54 (m, 2H), 7.29-7.24 (m, 1H), 3.59-3.54 (m, 1H), 3.40 (d, J = 6.9 Hz, 2H), 3.28 (s, 1H), 2.92 (d, J = 7.5 Hz, 4H), 1.96-1.82 (m, 5H) 275 I-30 ##STR00037## (400 MHz, DMSO-d6): 11.09 (br s, 1H), 8.97 (s, 1H), 7.56-7.40 (m, 7H), 7.30-7.20 (m, 1H), 3.62-3.49 (m, 3H), 3.36 (s, 1H), 3.01-2.91 (m, 2H), 2.87 (d, J = 7.6 Hz, 2H), 1.97-1.86 (m, 2H) 337 I-31 ##STR00038## (300 MHz, DMSO-d6): 11.09 (s, 1H), 8.97 (s, 1H), 7.60-7.39 (m, 7H), 7.29- 7.19 (m, 1H), 3.74-3.71 (m, 1H), 3.63-3.59 (m, 2H), 3.48-3.32 (m, 1H), 3.15-2.80 (m, 4H), 1.97-1.84 (m, 2H) 337
Example 9--Preparation of (R)--N-hydroxy-1'-methyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-- 5-carboxamide and (S)--N-hydroxy-1'-methyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-- 5-carboxamide (I-32 and I-33)
##STR00039##
[0424] Step-1: Dimethyl 2-(2-cyanoethyl)-1-oxo-2,3-dihydro-1H-indene-2,5-dicarboxylate
[0425] Into a 250-mL round-bottom flask was placed dimethyl 1-oxo-2,3-dihydro-1H-indene-2,5-dicarboxylate (4.4 g, 17.73 mmol, 1 equiv), DMF (50 mL), Et.sub.3N (5.3 g, 53.18 mmol, 3 equiv) and prop-2-enenitrile (2.3 g, 44.31 mmol, 2.5 equiv). The resulting solution was stirred for 16 h at 25.degree. C. The reaction was diluted with 100 mL of water and extracted with 3.times.100 mL of EtOAc. The organic layers were combined, washed with brine (2.times.50 mL), dried over Na.sub.2SO.sub.4, filtered and concentrated under vacuum. The residue was purified by normal phase chromatography on silica gel using EtOAc/petroleum ether (1:3). The collected fractions were concentrated under vacuum to afford 2.0 g (37% yield) of title compound as a yellow solid. MS: (ES, m/z): 302 [M+H].sup.+.
Step-2: Dimethyl 2-(3-aminopropyl)-1-hydroxy-2,3-dihydro-1H-indene-2,5-dicarboxylate
[0426] Into a 250-mL round-bottom flask was placed dimethyl 2-(2-cyanoethyl)-1-oxo-2,3-dihydro-1H-indene-2,5-dicarboxylate (1 g, 3.32 mmol, 1 equiv), MeOH (80 mL), PtO.sub.2 (1 g, 4.40 mmol, 1.33 equiv) and AcOH (20 mL). H.sub.2 was introduced to the flask and the reaction was stirred for 1 h at 25.degree. C. The solids were filtered out and the filtrate was concentrated under vacuum to afford 900 mg of title compound as a brown oil. MS: (ES, m/z): 308 [M+H].sup.+.
Step-3: Methyl 1-hydroxy-2'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-5-carboxylate
[0427] Into a 250-mL round-bottom flask was placed dimethyl 2-(3-aminopropyl)-1-hydroxy-2,3-dihydro-1H-indene-2,5-dicarboxylate (1 g, 3.25 mmol, 1 equiv). A solution of 7M NH.sub.3 in MeOH (30 mL) was added dropwise with stirring at 0.degree. C. The resulting solution was stirred for 1 h at 25.degree. C. The reaction was concentrated under vacuum. The residue purified by normal phase chromatography on silica gel using CH.sub.2Cl.sub.2/MeOH (20:1). The collected fractions were concentrated under vacuum to afford 1 g of the title compound as a brown oil. MS: (ES, m/z): 276 [M+H].sup.+.
Step-4: Methyl (R)-2'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-5-carboxylate and Methyl (S)-2'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-5-carboxylate
[0428] Into a 100-mL round-bottom flask was placed methyl 1-hydroxy-2'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-5-carboxylate (1 g, 3.63 mmol, 1 equiv), TFA (25 mL) and triethylsilane (25 mL). The resulting solution was stirred overnight at 80.degree. C. in an oil bath. After cooling to room temperature, the reaction was concentrated under vacuum. The residue purified by normal phase chromatography on silica gel using EtOAc/petroleum ether (99:1). The product was purified by Chiral-Prep-HPLC with the following conditions: Column: Phenomenex Lux.RTM. Cellulose-4 AXIA.TM. Packed, 250.times.21.2 mm, 5 .mu.m; Mobile Phase A: hexanes, Mobile Phase B: EtOH; Gradient: 30% B in 23 min; Detector: UV 254 nm, 220 nm. The first eluting isomer (Rt 2.8 min) was collected and concentrated under vacuum to give 310 mg (33% yield) of an off-white solid which was assigned as the R isomer of methyl 2'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-5-carboxylate. MS: (ES, m/z): 260 [M+H].sup.+. The second eluting isomer (Rt 3.8 min) was collected and concentrated under vacuum to give 280 mg (30% yield) of an off-white solid which was assigned as the R isomer of methyl 2'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-5-carboxylate. MS: (ES, m/z): 260 [M+H].sup.+.
Step-5: Methyl (R)-1'-methyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-5-carboxyla- te
[0429] Into a 10-mL vial purged and maintained with an inert atmosphere of nitrogen, was placed a solution of the first eluted isomer from Step 4, which was assigned as methyl (R)-2'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-5-carboxylate as described above, (50 mg, 0.19 mmol, 1 equiv) in DMF (2 mL). The solution was cooled to 0.degree. C. and NaH (60%, in mineral oil, 31 mg, 0.78 mmol, 4 equiv) was added. The mixture was allowed warm to room temperature and stirred for 30 min. CH.sub.3I (109 mg, 0.77 mmol, 4 equiv) was added and the reaction was stirred for 2 h at 25.degree. C. The reaction was then quenched by the addition of 20 mL of water and extracted with 3.times.20 mL of EtOAc. The organic layers were combined, washed with 2.times.50 mL of brine, dried over Na.sub.2SO.sub.4, filtered and concentrated under vacuum to afford 70 mg (crude) of title compound as a brown oil. MS: (ES, m/z): 274 [M+H].sup.+.
Step-6: (R)--N-Hydroxy-1'-methyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-piper- idine]-5-carboxamide
[0430] Into a 25-mL round-bottom flask was placed methyl (R)-1'-methyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-5-carboxyla- te (70 mg, 0.26 mmol, 1 equiv), THF/MeOH (4:1, 2.0 mL), NH.sub.2OH (50% in water, 2023 mg, 120 equiv) and aq. 1N NaOH (0.51 mg, 2 equiv). The resulting solution was stirred for 2 h at 25.degree. C. The solids were filtered out and the crude product was purified by Prep-HPLC with the following conditions: Column: XBridge Prep C18 OBD, 19.times.150 mm 5 .mu.m; Mobile Phase A: Water/0.05% Formic acid, Mobile Phase B: CH.sub.3CN; Gradient: 5% B up to 14% B in 7 min; Detector: UV 254 nm, 220 nm. The collected fractions were lyophilized to afford 6.5 mg (9% yield) of the title compound as an off-white solid. .sup.1H NMR (400 MHz, DMSO-d6) .delta. (ppm): 10.76 (s, 1H), 8.98 (s, 1H), 7.55-7.52 (t, J=6.2 Hz, 2H), 7.23-7.21 (d, J=8.0 Hz, 1H), 3.39-3.31 (m, 4H), 2.86-2.81 (m, 5H), 1.82-1.72 (m, 4H). MS: (ES, m/z): 275 [M+H].sup.+.
Step-7: Methyl (S)-1'-methyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-5-carboxyla- te
[0431] The procedure from Step 5 was followed using the second eluted isomer from Step 4, which was assigned as methyl (S)-2'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-5-carboxylate as described above, (50 mg, 0.19 mmol, 1 equiv) to afford 40 mg (crude) of the title compound as a yellow solid. MS: (ES, m/z): 274 [M+H].sup.+.
Step-8: (S)--N-Hydroxy-1'-methyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-piper- idine]-5-carboxamide
[0432] The procedure from Step 6 was followed using methyl (S)-1'-methyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-5-carboxyla- te (75 mg, 0.27 mmol, 1 equiv) to afford 13.4 mg (18% yield) of the title compound as a pink solid. .sup.1H NMR (400 MHz, DMSO-d6) .delta. (ppm): 11.08 (s, 1H), 8.96 (s, 1H), 7.55 (s, 1H), 7.54-7.52 (d, J=7.6 Hz, 1H), 7.23-7.21 (d, J=7.6 Hz, 1H), 3.40 (s, 1H), 3.38-3.35 (d, J=9.2 Hz, 2H), 3.31 (s, 1H), 2.86-2.81 (m, 5H), 1.82-1.72 (m, 4H). MS: (ES, m/z): 275 [M+H].sup.+.
Example 10--Preparation of (R)-1'-benzyl-N-hydroxy-2'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-5- -carboxamide (I-34)
##STR00040##
[0433] Step-1: (R)-1'-Benzyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-5-carboxyli- c acid
[0434] Into a 10-mL 4-necked round-bottom flask purged and maintained with an inert atmosphere of nitrogen, was placed a solution of the first eluted isomer from Example 9 Step 4, which was assigned as methyl (R)-2'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-5-carboxylate as described above, (50 mg, 0.19 mmol, 1 equiv) in DMF (2 mL). This was followed by the portionwise addition of NaH (60% in mineral oil, 31 mg, 0.78 mmol, 4 equiv) at 0.degree. C. The mixture was stirred for 30 min at 25.degree. C. (Bromomethyl)benzene (36 mg, 0.21 mmol, 1.09 equiv) was added. The resulting solution was stirred for 2 h at 25.degree. C. The reaction was then quenched by the addition of 10 mL of water and the reaction was extracted with 20 mL of EtOAc. The aqueous layer was adjusted to pH 4 with 6N HCl and was extracted with 3.times.50 mL of CH.sub.2Cl.sub.2. The combined organic layers were washed with 2.times.50 mL of brine, dried over Na.sub.2SO.sub.4, filtered and concentrated under vacuum to afford 75 mg (crude) of title compound as a yellow oil. MS: (ES, m/z): 336 [M+H].sup.+.
Step-2: (R)-1'-Benzyl-N-hydroxy-2'-oxo-1,3-dihydrospiro[indene-2,3'-piperi- dine]-5-carboxamide
[0435] Into a 25-mL round-bottom flask was placed (R)-1'-benzyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-5-carboxyli- c acid (75 mg, 0.22 mmol, 1 equiv) and DMA (2 mL). This was followed by the addition of isopropyl chloroformate (141 mg, 1.15 mmol, 5.14 equiv) dropwise with stirring at 0.degree. C. NMM (113 mg, 1.12 mmol, 5 equiv) was added dropwise with stirring at 0.degree. C. The mixture was stirred for 30 min at 0.degree. C. A solution of NH.sub.2OH.HCl (78 mg, 1.12 mmol, 5 equiv) in DMA (1 mL) was added dropwise with stirring at 0.degree. C. The resulting solution was stirred overnight at 25.degree. C. The solids were filtered out and the crude product was purified by Prep-HPLC with the following conditions: Column: XBridge Prep C18 OBD, 19.times.150 mm, 5 .mu.m; Mobile Phase A: Water/0.1% Formic acid, Mobile Phase B: CH.sub.3CN; Gradient: 15% B up to 55% B in 7 min, Detector: UV 254 nm, 220 nm. The collected fractions were lyophilized to afford 25.2 mg (32% yield) of title compound as an off-white solid. .sup.1H NMR (400 MHz, DMSO-d6) .delta. (ppm): 11.08 (s, 1H), 8.95 (s, 1H), 7.57-7.53 (m, 2H), 7.38-7.34 (m, 2H), 7.29-7.22 (m, 4H), 4.53 (s, 2H), 3.44 (d, J=16.0 Hz, 2H), 3.29-3.26 (m, 2H), 2.91 (d, J=15.6 Hz, 2H), 1.81 (d, J=17.6 Hz, 4H). MS: (ES, m/z): 351 [M+H].sup.+.
Example 11--Preparation of (S)-1'-benzyl-N-hydroxy-2'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-5- -carboxamide (I-35)
##STR00041##
[0436] Step-1: Methyl (S)-1'-benzyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-5-carboxyla- te
[0437] Into a 10-mL vial was placed a solution of the second eluted isomer from Example 9 Step 4, which was assigned as methyl (S)-2'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-5-carboxylate as described above, (50 mg, 0.19 mmol, 1 equiv) in DMF (2 mL). This was followed by the addition of NaH (60% in mineral oil, 8.47 mg, 0.18 mmol, 1.1 equiv) in portions at 0.degree. C. The resulting solution was stirred for 30 min at 0.degree. C. (Bromomethyl)benzene (35.7 mg, 0.21 mmol, 1.1 equiv) was added and the reaction was stirred for 2 h at 25.degree. C. The reaction was then quenched by the addition of 10 mL of ice water. The reaction was extracted with 2.times.15 mL of EtOAc. The combined organic layers were dried over Na.sub.2SO.sub.4, filtered and concentrated under vacuum to afford 50 mg (74% yield) of the title compound as a white solid. MS: (ES, m/z): 349 [M+H].sup.+.
Step-2: (S)-1'-Benzyl-N-hydroxy-2'-oxo-1,3-dihydrospiro[indene-2,3'-piperi- dine]-5-carboxamide
[0438] Into a 25-mL round-bottom flask was placed methyl (S)-1'-benzyl-2'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-5-carboxyla- te (56 mg, 0.16 mmol, 1 equiv), THF/MeOH (4:1, 3.0 mL), NH.sub.2OH (50% in water, 516.78 mg, 10.12 mmol, 60 equiv), aq. 1N NaOH (0.3 mL, 0.26 mmol, 2 equiv). The resulting solution was stirred for 2 h at 25.degree. C. The solids were filtered out. The crude product was purified by Prep-HPLC with the following conditions: Column: XBridge Prep C18 OBD, 19.times.150 mm, 5 m, Mobile Phase A: Water/0.1% Formic acid, Mobile Phase B: CH.sub.3CN; Gradient: 5% B up to 64% B in 7 min; Detector: UV 254 nm, 220 nm. The collected fractions were lyophilized to afford the title compound as a white solid. .sup.1H NMR (400 MHz, DMSO-d6) .delta. (ppm): 11.10 (s, 1H), 8.95 (s, 1H), 7.57 (s, 1H), 7.55-7.53 (d, J=8.0 Hz, 1H), 7.37-7.33 (m, 2H), 7.29-7.21 (m, 4H), 4.53 (s, 2H), 3.45-3.41 (d, J=12.0 Hz, 2H), 3.27 (s, 2H), 2.93-2.89 (d, J=15.6 Hz, 2H), 1.78 (s, 4H). MS: (ES, m/z): 351 [M+H].sup.+.
Example 12--Preparation of (R)--N-hydroxy-2'-oxo-1'-phenyl-1,3-dihydrospiro[indene-2,3'-piperidine]-- 5-carboxamide (I-36)
##STR00042##
[0439] Step-1: Methyl (R)-2'-oxo-1'-phenyl-1,3-dihydrospiro[indene-2,3'-piperidine]-5-carboxyla- te
[0440] Into a 25-mL round-bottom flask was placed the first eluted isomer from Example 9 Step 4, which was assigned as methyl (R)-2'-oxo-1,3-dihydrospiro[indene-2,3'-piperidine]-5-carboxylate as described above, (70 mg, 0.27 mmol, 1 equiv), THF (10 mL), Cu(OAc).sub.2 (49 mg, 0.27 mmol, 1 equiv), pyridine (32 mg, 0.40 mmol, 1.5 equiv), Et.sub.3N (82 mg, 0.81 mmol, 3 equiv) and phenylboronic acid (165 mg, 1.35 mmol, 5 equiv). O.sub.2 (g) was introduced to the flask and the reaction was stirred for 48 h at 60.degree. C. in an oil bath. The reaction was cooled to room temperature, filtered and concentrated under vacuum. The residue was purified by normal phase chromatography on silica gel using EtOAc/petroleum ether (1:2). The collected fractions were concentrated under vacuum to afford 40 mg (44% yield) of the title compound as a brown oil. MS: (ES, m/z): 336 [M+H].sup.+.
Step-2: (R)--N-Hydroxy-2'-oxo-1'-phenyl-1,3-dihydrospiro[indene-2,3'-piper- idine]-5-carboxamide
[0441] Into a 25-mL round-bottom flask was placed methyl (R)-2'-oxo-1'-phenyl-1,3-dihydrospiro[indene-2,3'-piperidine]-5-carboxyla- te (40 mg, 0.12 mmol, 1 equiv), THF/MeOH (4:1, 2.0 mL), NH.sub.2OH (50% in water, 946 mg, 120 equiv), aq. 1N NaOH (0.24 mL, 2 equiv). The resulting solution was stirred for 2 h at 25.degree. C. The solids were filtered out and the crude product was purified by Prep-HPLC with the following conditions: Column: XBridge Prep C18 OBD, 19.times.150 mm, 5 .mu.m; Mobile Phase A: Water/0.05% NH.sub.4OH, Mobile phase B: CH.sub.3CN; Gradient: 25% B up to 55% B in 7 min; Detector: UV 254 nm, 220 nm. The collected fractions were lyophilized to afford 8.0 mg (20% yield) of the title compound as a white solid. .sup.1H NMR (400 MHz, DMSO-d6) .delta. (ppm): 11.04 (s, 1H), 8.96 (s, 1H), 7.58-7.54 (m, 2H), 7.40-7.36 (m, 2H), 7.29-7.22 (m, 4H), 3.70-3.67 (m, 2H), 3.50-3.45 (m, 2H), 3.00-2.94 (m, 2H), 1.97-1.89 (m, 4H). MS: (ES, m/z): 337 [M+H].sup.+.
TABLE-US-00007 TABLE 6 The following compound was prepared according to the method of Example 12, with stereochemistry assigned as described. Found (ES, m/z) Ex. Structure .sup.1H-NMR .delta. (ppm) [M + H].sup.+ I-37 ##STR00043## (400 MHz, DMSO-d6): 11.09 (s, 1H), 8.97 (s, 1H), 7.58 (s, 1H), 7.55-7.53 (d, J = 7.6 Hz, 1H), 7.40-7.36 (m, 2H), 7.28-7.22 (m, 4H), 3.69-3.66 (m, 2H), 3.49-3.44 (m, 2H), 2.99-2.94 (m, 2H), 1.98-1.90 (m, 4H) 337
Example 13--In Vitro Histone Deacetylase Assay
[0442] The enzymatic HDAC6 assay was performed using electrophoretic mobility shift assay. Full length human recombinant HDAC6 protein was expressed in baculoviral system and purified by affinity chromatography. The enzymatic reactions were assembled in 384 well plates in a total volume of 25 .mu.L in a reaction buffer composing; 100 mM HEPES, pH 7.5, 25 mM KCl, 0.1% bovine serum albumin, 0.01% Triton X-100, 1% DMSO (from compounds) 2 .mu.M of the fluorescently labeled peptide substrate and enzyme. The enzyme was added at a final concentration of 1 nM. The peptide substrate RHKK(Ac)--NH.sub.2 was used. The compounds were tested at 12 concentrations spaced by 3.times. dilution intervals. Negative control samples (0%-inhibition in the absence of inhibitor) and positive control samples (100%-inhibition) were assembled in replicates of four in each assay plate. The reactions were incubated at 25.degree. C. and quenched by the addition of 45 .mu.L of termination buffer (100 mM HEPES, pH 7.5, 0.01% Triton X-100, 0.05% SDS).
[0443] The terminated assay plates were analyzed on LabChip.RTM. 3000 microfluidic electrophoresis instrument (Perkin Elmer/Caliper Life Sciences). The fluorescence intensity of the electrophoretically separated de-acetylated product and substrate peptide was measured. Activity in each sample was determined as the product to sum ratio (PSR): P/(S+P), where P is the peak height of the product peptide and S is the peak height of the substrate peptide. Percent inhibition (Pinh) is determined using the following equation:
[0444] Pinh=(PSR0%-PSRinh)/(PSR0%-PSR100%)*100, where PSRinh is the product sum ratio in the presence of inhibitor, PSR0% is the average product sum ration in the absence of inhibitor and PSR100% is the average product sum ratio in 100%-inhibition control samples. The IC.sub.50 values of inhibitors were determined by fitting the %-inhibition curves with 4 parameter dose-response model using XLfit 4 software.
[0445] As set forth in Table-7, below, IC.sub.50 values are defined as follows: IC.sub.50.ltoreq.0.01 .mu.M (+++); IC.sub.50>0.01 .mu.M and .ltoreq.0.05 .mu.M (++) IC.sub.50>0.05 .mu.M (+).
TABLE-US-00008 TABLE-7 Inhibitory Concentration (IC.sub.50) Values for Representative Compounds against HDAC6 HDAC6 activity IUPAC Name Number range (R)-N-hydroxy-1'-methyl-2'-oxo-1,3-dihydrospiro[indene-2,3'- (I-1) ++ pyrrolidine]-5-carboxamide (S)-N-hydroxy-1'-methyl-2'-oxo-1,3-dihydrospiro[indene-2,3'- (I-2) ++ pyrrolidine]-5-carboxamide (R)-N-hydroxy-1'-isopropyl-2'-oxo-1,3-dihydrospiro[indene-2,3'- (I-3) ++ pyrrolidine]-5-carboxamide (S)-N-hydroxy-1'-isopropyl-2'-oxo-1,3-dihydrospiro[indene-2,3'- (I-4) + pyrrolidine]-5-carboxamide (R)-1'-ethyl-N-hydroxy-2'-oxo-1,3-dihydrospiro[indene-2,3'- (I-5) ++ pyrrolidine]-5-carboxamide (S)-1'-ethyl-N-hydroxy-2'-oxo-1,3-dihydrospiro[indene-2,3'- (I-6) + pyrrolidine]-5-carboxamide (R)-N-hydroxy-1'-isobutyl-2'-oxo-1,3-dihydrospiro[indene-2,3'- (I-7) ++ pyrrolidine]-5-carboxamide (S)-N-hydroxy-1'-isobutyl-2'-oxo-1,3-dihydrospiro[indene-2,3'- (I-8) ++ pyrrolidine]-5-carboxamide (R)-N-hydroxy-1'-(2-methylbenzyl)-2'-oxo-1,3- (I-9) ++ dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxamide (S)-N-hydroxy-1'-(2-methylbenzyl)-2'-oxo-1,3- (I-10) ++ dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxamide (R)-1'-benzyl-N-hydroxy-2'-oxo-1,3-dihydrospiro[indene-2,3'- (I-11) +++ pyrrolidine]-5-carboxamide (S)-1'-benzyl-N-hydroxy-2'-oxo-1,3-dihydrospiro[indene-2,3'- (I-12) ++ pyrrolidine]-5-carboxamide (R)-N-hydroxy-2'-oxo-1'-phenyl-1,3-dihydrospiro[indene-2,3'- (I-13) ++ pyrrolidine]-5-carboxamide (S)-N-hydroxy-2'-oxo-1'-phenyl-1,3-dihydrospiro[indene-2,3'- (I-14) ++ pyrrolidine]-5-carboxamide (R)-1'-(4-fluorophenyl)-N-hydroxy-2'-oxo-1,3- (I-15) ++ dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxamide (S)-1'-(fluorophenyl)-N-hydroxy-2'-oxo-1,3- (I-16) ++ dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxamide (R)-N-hydroxy-2'-oxo-1'-(pyridin-3-yl)-1,3-dihydrospiro[indene- (I-17) +++ 2,3'-pyrrolidine]-5-carboxamide (S)-N-hydroxy-2'-oxo-1'-(pyridin-3-yl)-1,3-dihydrospiro[indene- (I-18) ++ 2,3'-pyrrolidine]-5-carboxamide (R)-N-hydroxy-2'-oxo-1'-(pyridin-2-yl)-1,3-dihydrospiro[indene- (I-19) ++ 2,3'-pyrrolidine]-5-carboxamide (S)-N-hydroxy-2'-oxo-1'-(pyridin-2-yl)-1,3-dihydrospiro[indene- (I-20) ++ 2,3'-pyrrolidine]-5-carboxamide (R)-N-hydroxy-2'-oxo-1'-(pyridin-4-yl)-1,3-dihydrospiro[indene- (I-21) ++ 2,3'-pyrrolidine]-5-carboxamide (S)-N-hydroxy-2'-oxo-1'-(pyridin-4-yl)-1,3-dihydrospiro[indene- (I-22) ++ 2,3'-pyrrolidine]-5-carboxamide (R)-N-hydroxy-2'-oxo-1'-(pyridin-3-ylmethyl)-1,3- (I-23) ++ dihydrospiro[indene-2,3'-pyrrolidine]-5-carboxamide (R)-N-hydroxy-1'-methyl-1,3-dihydrospiro[indene-2,3'- (I-24) ++ pyrrolidine]-5-carboxamide (S)-N-hydroxy-1'-methyl-1,3-dihydrospiro[indene-2,3'- (I-25) + pyrrolidine]-5-carboxamide (R)-1'-benzyl-N-hydroxy-1,3-dihydrospiro[indene-2,3'- (I-26) ++ pyrrolidine]-5-carboxamide (S)-1'-benzyl-N-hydroxy-1,3-dihydrospiro[indene-2,3'- (I-27) ++ pyrrolidine]-5-carboxamide (R)-1'-acetyl-N-hydroxy-1,3-dihydrospiro[indene-2,3'- (I-28) ++ pyrrolidine]-5-carboxamide (S)-1'-acetyl-N-hydroxy-1,3-dihydrospiro[indene-2,3'-pyrrolidine]- (I-29) ++ 5-carboxamide (R)-1'-benzoyl-N-hydroxy-1,3-dihydrospiro[indene-2,3 '- (I-30) +++ pyrrolidine]-5-carboxamide (S)-1'-benzoyl-N-hydroxy-1,3-dihydrospiro[indene-2,3'- (I-31) +++ pyrrolidine]-5-carboxamide (R)-N-hydroxy-1'-methyl-2-oxo-1,3-dihydrospiro[indene-2,3'- (I-32) ++ piperidine]-5-carboxamide (S)-N-hydroxy-1'-methyl-2'-oxo-1,3-dihydrospiro[indene-2,3'- (I-33) ++ piperidine]-5-carboxamide (R)-1'-benzyl-N-hydroxy-2'-oxo-1,3-dihydrospiro[indene-2,3'- (I-34) +++ piperidine]-5-carboxamide (S)-1'-benzyl-N-hydroxy-2'-oxo-1,3-dihydrospiro[indene-2,3'- (I-35) ++ piperidine]-5-carboxamide (R)-N-hydroxy-2'-oxo-1'-phenyl-1,3-dihydrospiro[indene-2,3'- (I-36) +++ piperidine]-5-carboxamide (S)-N-hydroxy-2'-oxo-1'-phenyl-1,3-dihydrospiro[indene-2,3'- (I-37) ++ piperidine]-5-carboxamide
EQUIVALENTS
[0446] While the present invention has been described in conjunction with the specific embodiments set forth above, many alternatives, modifications and other variations thereof will be apparent to those of ordinary skill in the art. All such alternatives, modifications and variations are intended to fall within the spirit and scope of the present invention.
* * * * *














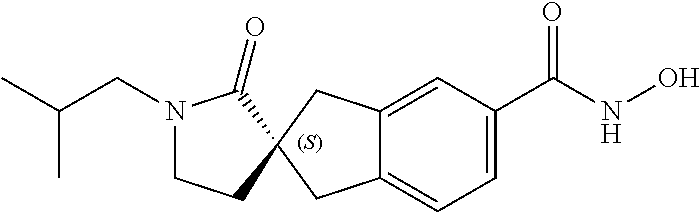





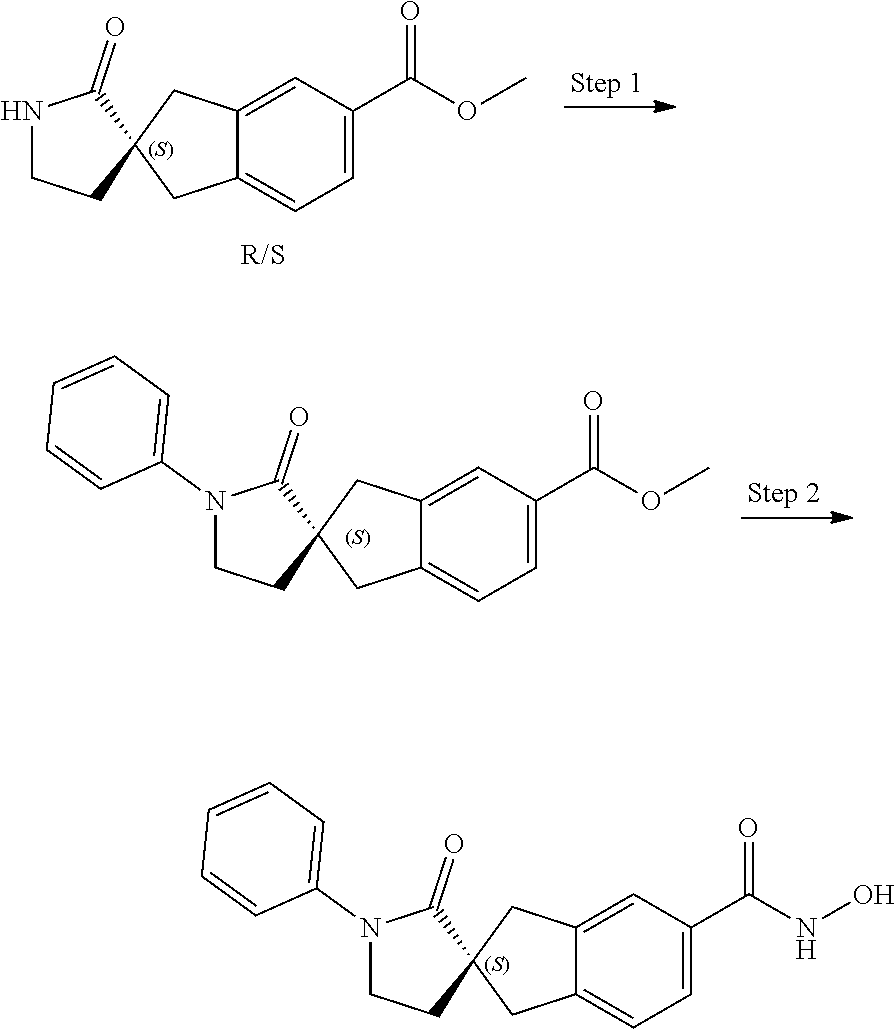
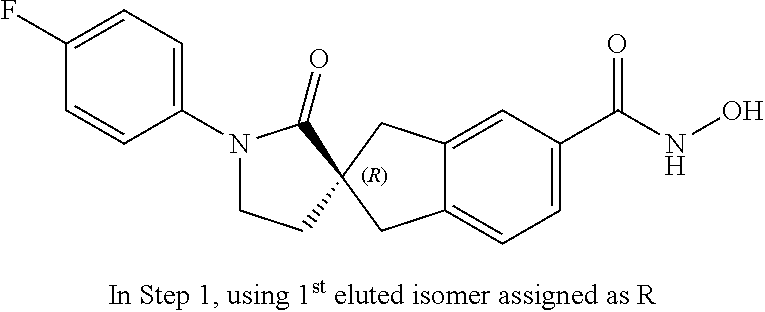



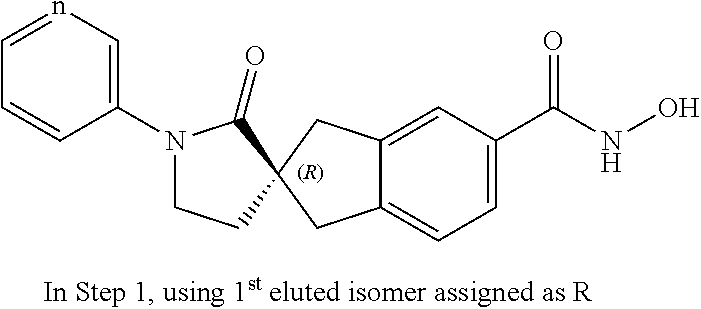


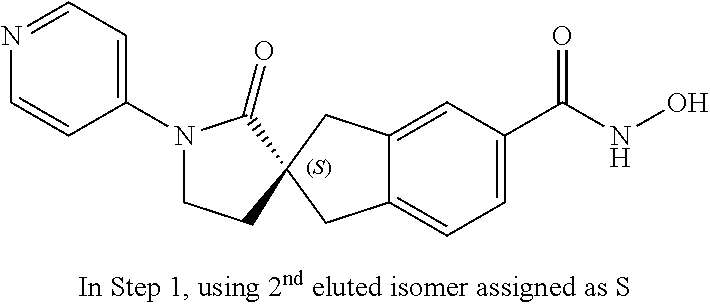


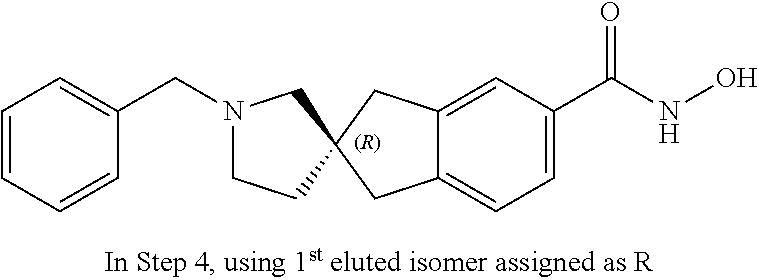
















XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.