Method For Preventing Or Reducing Low Speed Pre-ignition
Smith; Jordan C. ; et al.
U.S. patent application number 16/176161 was filed with the patent office on 2019-06-06 for method for preventing or reducing low speed pre-ignition. The applicant listed for this patent is ExxonMobil Research and Engineering Company. Invention is credited to Raymond G. Burns, III, Smruti A. Dance, Douglas E. Deckman, Jordan C. Smith.
| Application Number | 20190169524 16/176161 |
| Document ID | / |
| Family ID | 64427214 |
| Filed Date | 2019-06-06 |
| United States Patent Application | 20190169524 |
| Kind Code | A1 |
| Smith; Jordan C. ; et al. | June 6, 2019 |
METHOD FOR PREVENTING OR REDUCING LOW SPEED PRE-IGNITION
Abstract
A method for preventing or reducing low speed pre-ignition in an engine lubricated with a lubricating oil by using as the lubricating oil a formulated oil. The formulated oil has a composition including a lubricating oil base stock as a major component, and at least one lubricating oil additive, as a minor component. The lubricating oil contains at least one metal or metalloid in an amount sufficient that, in thermogravimetric measurements of the lubricating oil by a Thermogravimetric Engine Oil Simulation Test, carbon black decomposition temperature is increased as compared to the carbon black decomposition temperature in a lubricating oil containing calcium or magnesium. A lubricating oil useful for preventing or reducing low speed pre-ignition in an engine lubricated with the lubricating oil. The lubricating oils of this disclosure are particularly advantageous as passenger vehicle engine oil (PVEO) products.
| Inventors: | Smith; Jordan C.; (Marlton, NJ) ; Burns, III; Raymond G.; (Easton, PA) ; Dance; Smruti A.; (Robbinsville, NJ) ; Deckman; Douglas E.; (Easton, PA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 64427214 | ||||||||||
| Appl. No.: | 16/176161 | ||||||||||
| Filed: | October 31, 2018 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 62594233 | Dec 4, 2017 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C10M 107/02 20130101; C10N 2030/45 20200501; C10M 125/10 20130101; C10M 2201/041 20130101; C10N 2010/08 20130101; C10N 2030/52 20200501; C10M 2203/102 20130101; C10N 2030/00 20130101; C10N 2010/14 20130101; C10N 2030/08 20130101; C10N 2040/25 20130101; C10N 2040/255 20200501; C10M 2205/028 20130101; C10N 2030/10 20130101; C10M 169/04 20130101; C10M 2201/062 20130101; C10M 125/02 20130101; C10N 2010/02 20130101; C10N 2010/06 20130101; C10M 2205/0206 20130101 |
| International Class: | C10M 169/04 20060101 C10M169/04; C10M 107/02 20060101 C10M107/02; C10M 125/02 20060101 C10M125/02; C10M 125/10 20060101 C10M125/10 |
Claims
1. A method for preventing or reducing low speed pre-ignition in an engine lubricated with a lubricating oil by using as the lubricating oil a formulated oil, said formulated oil having a composition comprising a lubricating oil base stock as a major component; and at least one lubricating oil additive, as a minor component; wherein said lubricating oil contains at least one metal or metalloid in an amount sufficient that, in thermogravimetric measurements of the lubricating oil by a Thermogravimetric Engine Oil Simulation Test, carbon black decomposition temperature is increased as compared to the carbon black decomposition temperature in a lubricating oil containing calcium or magnesium.
2. The method of claim 1 wherein, in thermogravimetric measurements of the lubricating oil by a Thermogravimetric Engine Oil Simulation Test, the carbon black decomposition temperature is increased by at least 10.degree. C. as compared to the carbon black decomposition temperature in a lubricating oil containing calcium or magnesium.
3. The method of claim 1 wherein, in thermogravimetric measurements of the lubricating oil by a Thermogravimetric Engine Oil Simulation Test, the carbon black decomposition temperature is increased by at least 25.degree. C. as compared to the carbon black decomposition temperature in a lubricating oil containing calcium or magnesium.
4. The method of claim 1 wherein the engine exhibits greater than about 10% reduced low speed pre-ignition, based on normalized low speed pre-ignition (LSPI) counts per 25,000 engine cycles, engine operation at 2000 revolutions per minute (RPM) and brake mean effective pressure (BMEP) at 18 bar, as compared to low speed pre-ignition performance achieved in an engine using a lubricating oil containing calcium or magnesium.
5. The method of claim 1 wherein the at least one metal or metalloid comprises sodium, manganese, titanium, aluminum, or mixtures thereof.
6. The method of claim 1 wherein the at least one metal or metalloid is present in the lubricating oil in an amount from about 100 ppm to about 25,000 ppm.
7. The method of claim 1 wherein (i) the at least one metal or metalloid is present in the lubricating oil in an amount from about 100 ppm to about 1000 ppm; (ii) total base number (TBN), as measured by ASTM D2896, of the lubricating oil ranges from about 2 mg KOH to about 17 mg KOH; (iii) total sulfated ash of the lubricating oil ranges from about 0.01 to about 1 wt %.
8. The method of claim 1 wherein the at least one metal or metalloid is incorporated into the lubricating oil by maintaining constant ash, maintaining constant detergent metal, maintaining constant total base number (TBN), or top treating.
9. The method of claim 1 wherein the at least one metal or metalloid is incorporated into the lubricating oil through the at least one lubricating oil additive.
10. The method of claim 1 wherein the at least one lubricating oil additive is selected from the group consisting of a salicylate, a carboxylate, a sulfonate, a phenate, a stearate, and an oxide.
11. The method of claim 1 wherein the at least one lubricating oil additive has a total sulfated ash from about 0.02 to about 0.17 wt %.
12. The method of claim 1 wherein the at least one lubricating oil additive comprises one or more of a detergent, dispersant, viscosity index improver, antioxidant, pour point depressant, corrosion inhibitor, metal deactivator, seal compatibility additive, anti-foam agent, inhibitor, anti-rust additive, and friction modifier.
13. The method of claim 1 wherein the lubricating oil base stock comprises a Group II, Group III, or Group IV base oil.
14. The method of claim 1 wherein at least one lubricating oil additive concentration ranges from about 0.1 to about 20 weight percent, based on the total weight of the lubricating oil.
15. The method of claim 1 wherein the lubricating oil base stock concentration ranges from about 50 to about 99 weight percent, based on the total weight of the lubricating oil.
16. The method of claim 1 wherein the lubricating oil is a passenger vehicle engine oil (PVEO).
17. A lubricating engine oil having a composition comprising a lubricating oil base stock as a major component; and at least one lubricating oil additive, as a minor component; wherein said lubricating engine oil contains at least one metal or metalloid in an amount sufficient that, in thermogravimetric measurements of the lubricating engine oil by a Thermogravimetric Engine Oil Simulation Test, carbon black decomposition temperature is increased as compared to the carbon black decomposition temperature in a lubricating engine oil containing calcium or magnesium.
18. The lubricating engine oil of claim 17 wherein, in thermogravimetric measurements of the lubricating engine oil by a Thermogravimetric Engine Oil Simulation Test, the carbon black decomposition temperature is increased by at least 10.degree. C. as compared to the carbon black decomposition temperature in a lubricating engine oil containing calcium or magnesium.
19. The lubricating engine oil of claim 17 wherein, in thermogravimetric measurements of the lubricating engine oil by a Thermogravimetric Engine Oil Simulation Test, the carbon black decomposition temperature is increased by at least 25.degree. C. as compared to the carbon black decomposition temperature in a lubricating engine oil containing calcium or magnesium.
20. The lubricating engine oil of claim 17 wherein the engine exhibits greater than about 10% reduced low speed pre-ignition, based on normalized low speed pre-ignition (LSPI) counts per 25,000 engine cycles, engine operation at 2000 revolutions per minute (RPM) and brake mean effective pressure (BMEP) at 18 bar, as compared to low speed pre-ignition performance achieved in an engine using a lubricating engine oil containing calcium or magnesium.
21. The lubricating engine oil of claim 17 wherein the at least one metal or metalloid comprises sodium, manganese, titanium, aluminum, or mixtures thereof.
22. The lubricating engine oil of claim 17 wherein the at least one metal or metalloid is present in the lubricating engine oil in an amount from about 100 ppm to about 25,000 ppm.
23. The lubricating engine oil of claim 17 wherein (i) the at least one metal or metalloid is present in the lubricating engine oil in an amount from about 100 ppm to about 1000 ppm; (ii) total base number (TBN), as measured by ASTM D2896, of the lubricating engine oil ranges from about 2 mg KOH to about 17 mg KOH; (iii) total sulfated ash of the lubricating engine oil ranges from about 0.01 to about 1 wt %.
24. The lubricating engine oil of claim 17 wherein the at least one metal or metalloid is incorporated into the lubricating engine oil by maintaining constant ash, maintaining constant detergent metal, maintaining constant total base number (TBN), or top treating.
25. The lubricating engine oil of claim 17 wherein the at least one metal or metalloid is incorporated into the lubricating oil through the at least one lubricating oil additive.
26. The lubricating engine oil of claim 17 wherein the at least one lubricating oil additive is selected from the group consisting of a salicylate, a carboxylate, a sulfonate, a phenate, a stearate, and an oxide.
27. The lubricating engine oil of claim 17 wherein the at least one lubricating oil additive has a total sulfated ash from about 0.02 to about 0.17 wt %.
28. The lubricating engine oil of claim 17 wherein the at least one lubricating oil additive comprises one or more of a detergent, dispersant, viscosity index improver, antioxidant, pour point depressant, corrosion inhibitor, metal deactivator, seal compatibility additive, anti-foam agent, inhibitor, anti-rust additive, and friction modifier.
29. The lubricating engine oil of claim 17 wherein the lubricating oil base stock comprises a Group II, Group III, or Group IV base oil.
30. The lubricating engine oil of claim 17 wherein at least one lubricating oil additive concentration ranges from about 0.1 to about 20 weight percent, based on the total weight of the lubricating engine oil.
31. The lubricating engine oil of claim 17 wherein the lubricating oil base stock concentration ranges from about 50 to about 99 weight percent, based on the total weight of the lubricating engine oil.
32. The lubricating engine oil of claim 17 which is a passenger vehicle engine oil (PVEO).
33. An engine lubricated with the lubricating engine oil of claim 17.
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application claims the benefit of U.S. Provisional Application No. 62/594,233, filed on Dec. 4, 2017, the entire contents of which are incorporated herein by reference.
FIELD
[0002] This disclosure relates to a method for preventing or reducing low speed pre-ignition (LSPI) in an engine lubricated with a lubricating oil by using as the lubricating oil a formulated oil that has at least one metal or metalloid present in a particular amount in the formulated oil. The lubricating oils of this disclosure are useful as passenger vehicle engine oil (PVEO) products.
BACKGROUND
[0003] Pre-ignition in a flame propagation (or "spark-ignition") engine describes an event wherein the air/fuel mixture in the cylinder ignites before the spark plug fires. Pre-ignition is initiated by an ignition source other than the spark, such as hot spots in the combustion chamber, a spark plug that runs too hot for the application, or carbonaceous deposits in the combustion chamber heated to incandescence by previous engine combustion events.
[0004] Many passenger car manufacturers have observed intermittent pre-ignition in their production turbocharged gasoline engines, particularly at low speeds and medium-to-high loads. At these elevated loads, pre-ignition usually results in severe engine knock that can damage the engine. The cause of the pre-ignition is not fully understood, and may in fact be attributed to multiple phenomena such as hot deposits within the combustion chamber, elevated levels of lubricant vapor entering from the PCV system, oil seepage past the turbocharger compressor seals or oil and/or fuel droplet auto-ignition during the compression stroke.
[0005] Pre-ignition can sharply increase combustion chamber temperatures and lead to rough engine operation or loss of performance. Traditional methods of eliminating pre-ignition include, for example, proper spark plug selection, proper fuel/air mixture adjustment, and periodic cleaning of the combustion chambers. Hardware solutions such as cooled exhaust gas recirculation (EGR) are known, but these can be costly to implement and present packaging problems.
[0006] Low speed pre-ignition (LSPI) is a type of abnormal combustion affecting engines operating at high brake mean effective pressure (BMEP) and low engine speed (RPM). This includes internal combustion engines using a variety of fuels, including natural gas, gasoline, diesel, biofuels, and the like. Downsized, downspeeded, turbocharged engines are most susceptible to operating under these engine conditions and are thus more susceptible to LSPI. As the automobile industry continues to move towards further downsizing, downspeeding, and increased turbocharging to increase vehicle fuel economy and reduce carbon dioxide emissions, the concern over LSPI continues to grow.
[0007] The further development of downspeeded, turbocharged gasoline engines is being impeded by LSPI. A solution to this problem or even a mitigation of its occurrence would remove barriers for original equipment manufacturer (OEM) technology and efficiency improvement. A lubricant formulation solution would enable product differentiation with regard to LSPI.
[0008] Although pre-ignition problems can be and are being resolved by optimization of internal engine components and by the use of new component technology such as electronic controls, modification of the lubricating oil compositions used to lubricate such engines is desirable. For example, it would be desirable to develop new lubricating oil formulations which are particularly useful in internal combustion engines and, when used in internal combustion engines, will prevent or minimize the pre-ignition problems. It is desired that the lubricating oil composition be useful in lubricating gasoline-fueled, spark-ignited engines.
[0009] Despite the advances in lubricant oil formulation technology, there exists a need for an engine oil lubricant that effectively prevents or reduces low speed pre-ignition especially for downsized, downspeeded, turbocharged engines.
SUMMARY
[0010] This disclosure relates in part to new lubricating oil formulations which are particularly useful in internal combustion engines and, when used in internal combustion engines will prevent or minimize pre-ignition problems. The lubricating oil compositions of this disclosure are useful in lubricating gasoline-fueled, spark-ignited engines. The lubricant formulation chemistry of this disclosure can be used to prevent or control the detrimental effect of LSPI in engines which have already been designed or sold in the marketplace as well as future engine technology. The lubricant formulation chemistry of this disclosure removes barriers for OEM technology and efficiency improvement, and enables further development of downspeeded, turbocharged gasoline engines that is currently being impeded by LSPI. The lubricant formulation solution afforded by this disclosure for preventing or reducing LSPI enables product differentiation with regard to LSPI.
[0011] In addition to LSPI prevention, the lubricating oil formulations of this disclosure can also achieve desired performance characteristics in terms of wear, cleanliness, fuel economy, and the like. In accordance with this disclosure, formulators have increased flexibility to maximize other performance attributes, while minimizing LSPI activity.
[0012] This disclosure also relates in part to a method for preventing or reducing low speed pre-ignition in an engine lubricated with a lubricating oil by using as the lubricating oil a formulated oil. The formulated oil has a composition comprising a lubricating oil base stock as a major component, and at least one lubricating oil additive, as a minor component. The lubricating oil contains at least one metal or metalloid in an amount sufficient that, in thermogravimetric measurements of the lubricating oil by a Thermogravimetric Engine Oil Simulation Test, carbon black decomposition temperature is increased as compared to the carbon black decomposition temperature in a lubricating oil containing calcium or magnesium.
[0013] This disclosure further relates in part to a lubricating engine oil having a composition comprising a lubricating oil base stock as a major component, and at least one lubricating oil additive, as a minor component. The lubricating engine oil contains at least one metal or metalloid in an amount sufficient that, in thermogravimetric measurements of the lubricating engine oil by a Thermogravimetric Engine Oil Simulation Test, carbon black decomposition temperature is increased as compared to the carbon black decomposition temperature in a lubricating engine oil containing calcium or magnesium.
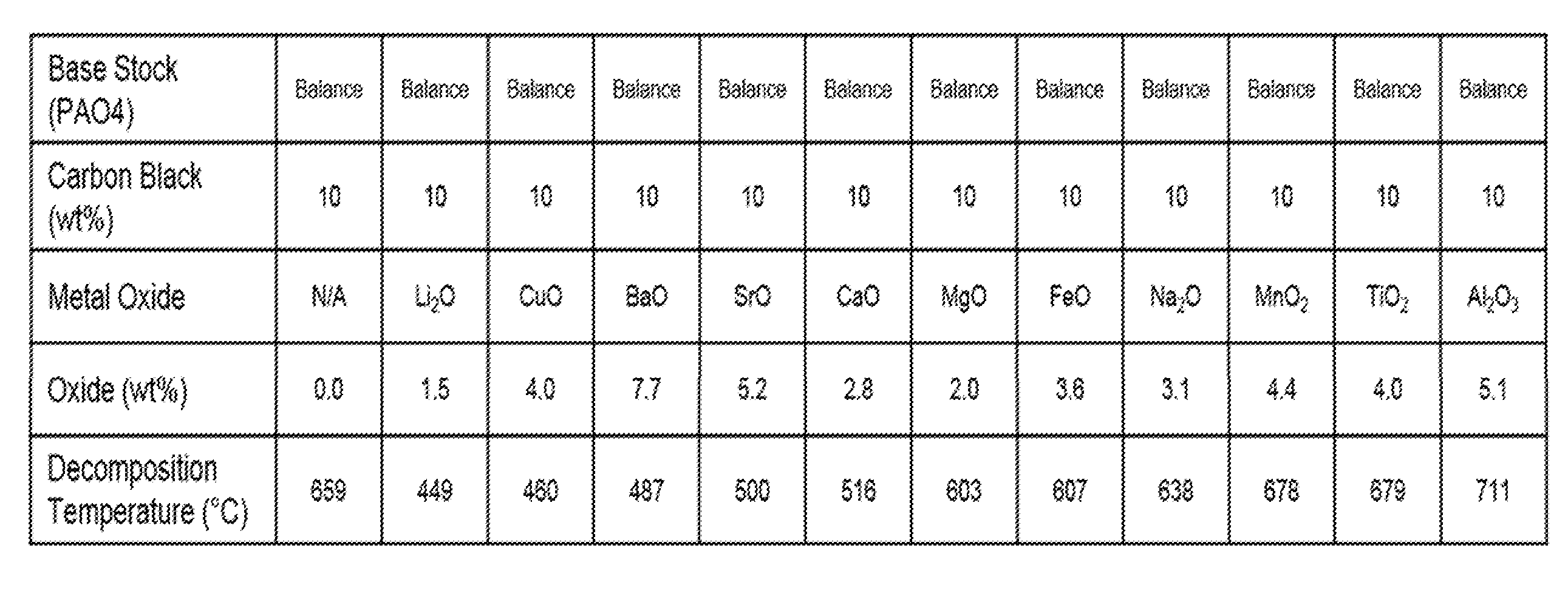
[0014] It has been surprisingly found that, in accordance with this disclosure, prevention or reduction of LSPI can be attained in an engine lubricated with a lubricating oil by using as the lubricating oil a formulated oil having at least one metal or metalloid in an amount sufficient that, in thermogravimetric measurements of the lubricating oil by a Thermogravimetric Engine Oil Simulation Test, the carbon black decomposition temperature is increased by at least 10.degree. C., or by at least 25.degree. C., as compared to the carbon black decomposition temperature in a lubricating oil containing calcium or magnesium. The carbon black decomposition temperature is an indicator of the catalytic activity of the metal oxide for promoting auto-ignition. An increase in the decomposition temperature of a lubricant corresponds to an increase of the auto-ignition temperature of the LSPI precursors resulting in less LSPI activity.
[0015] In addition, it has been surprisingly found that, in accordance with this disclosure, lubricant compositions containing at least one of sodium, manganese, titanium, aluminum, or mixtures thereof, in combination with other typical lubricating oil additives, and in amounts from about 100 ppm to about 25,000 ppm, provide improved LSPI performance.
[0016] Other objects and advantages of the present disclosure will become apparent from the detailed description that follows.
BRIEF DESCRIPTION OF THE DRAWINGS
[0017] FIG. 1 shows carbon decomposition temperatures of various metal oxides in a carbon black/PAO mixture, in accordance with the Examples.
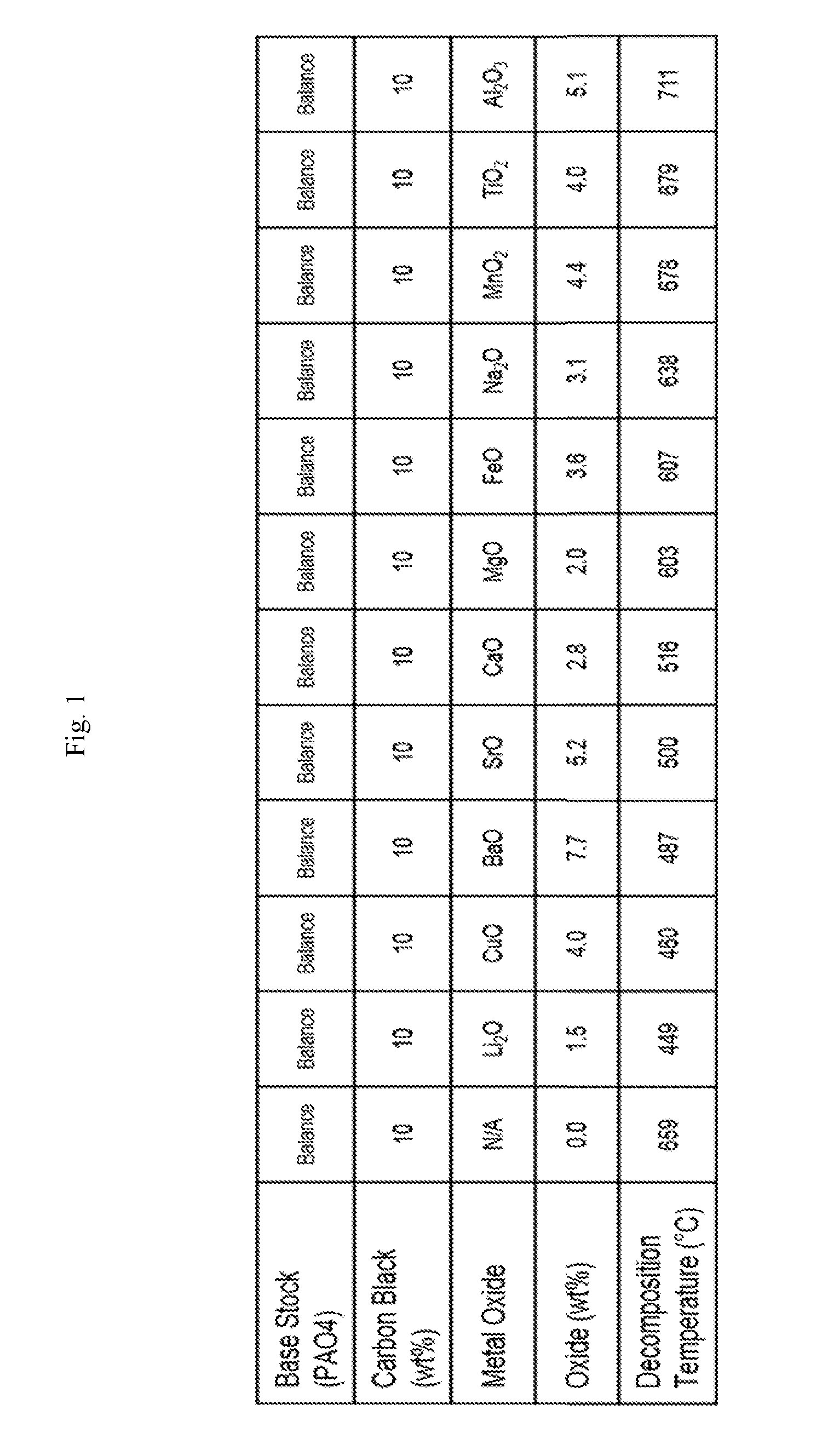
[0018] FIG. 2 shows carbon decomposition temperatures of various metal oxides in a carbon black/base stock mixture in which barium and titanium and various base stocks were used, in accordance with the Examples.
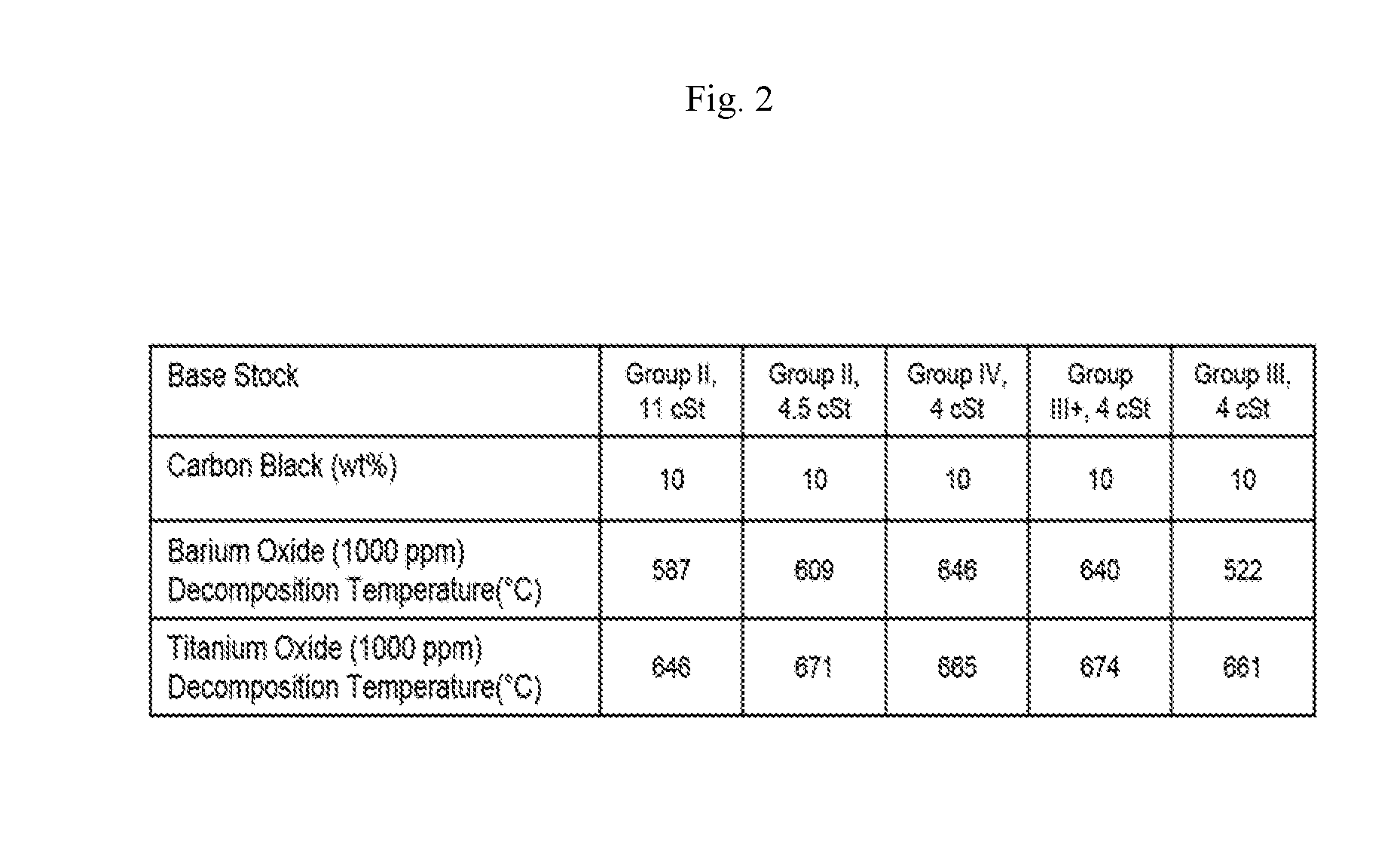
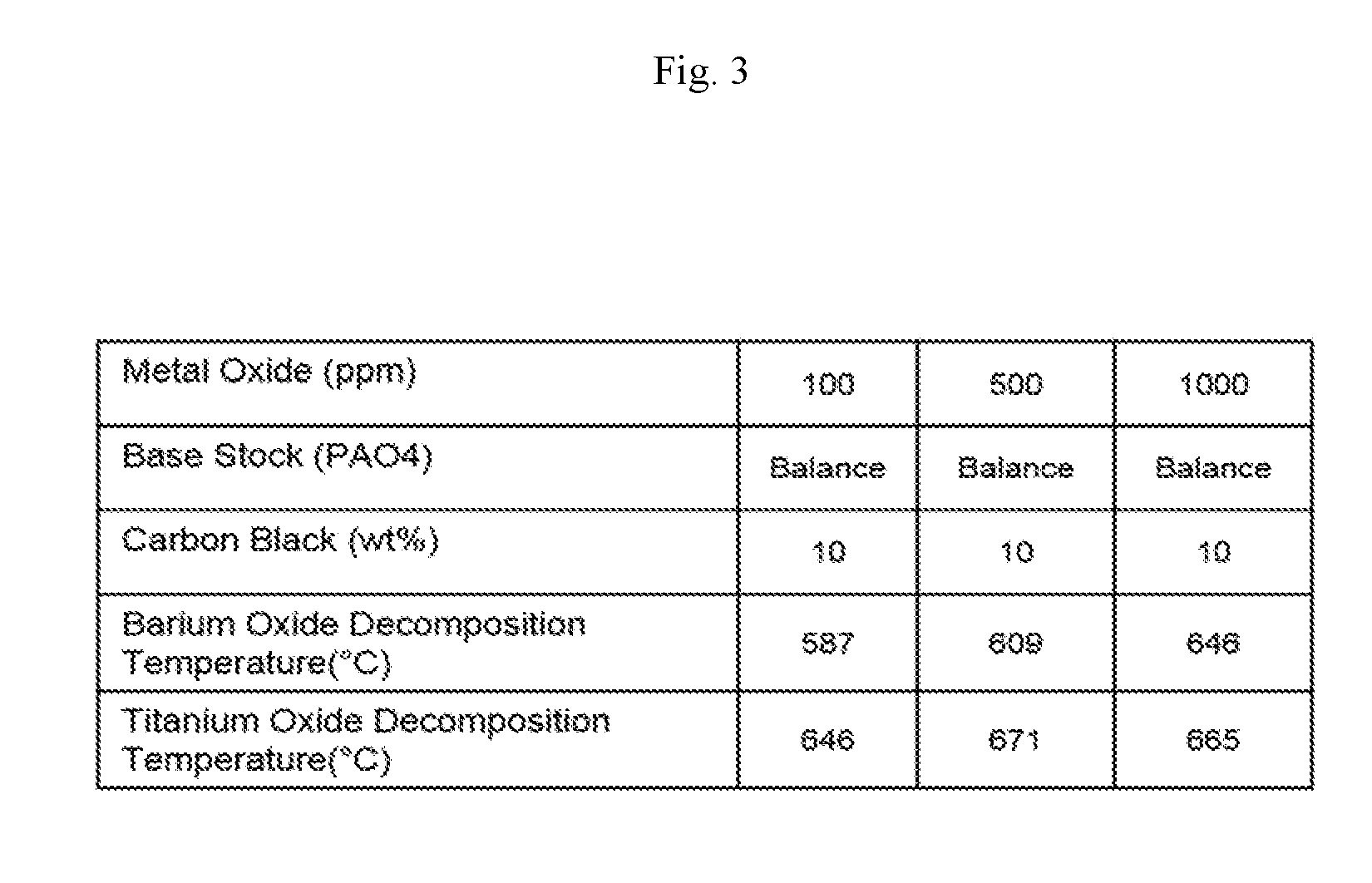
[0019] FIG. 3 shows carbon decomposition temperatures of various metal oxides in a carbon black mixture in which barium and titanium and PAO base stock were used with varying concentrations of metal oxide, in accordance with the Examples.
DETAILED DESCRIPTION
[0020] All numerical values within the detailed description and the claims herein are modified by "about" or "approximately" the indicated value, and take into account experimental error and variations that would be expected by a person having ordinary skill in the art. The phrase "major amount" or "major component" as it relates to components included within the lubricating oils of the specification and the claims means greater than or equal to 50 wt. %, or greater than or equal to 60 wt. %, or greater than or equal to 70 wt. %, or greater than or equal to 80 wt. %, or greater than or equal to 90 wt. % based on the total weight of the lubricating oil. The phrase "minor amount" or "minor component" as it relates to components included within the lubricating oils of the specification and the claims means less than 50 wt. %, or less than or equal to 40 wt. %, or less than or equal to 30 wt. %, or greater than or equal to 20 wt. %, or less than or equal to 10 wt. %, or less than or equal to 5 wt. %, or less than or equal to 2 wt. %, or less than or equal to 1 wt. %, based on the total weight of the lubricating oil. The phrase "essentially free" as it relates to components included within the lubricating oils of the specification and the claims means that the particular component is at 0 weight % within the lubricating oil, or alternatively is at impurity type levels within the lubricating oil (less than 100 ppm, or less than 20 ppm, or less than 10 ppm, or less than 1 ppm). The phrase "other lubricating oil additives" as used in the specification and the claims means other lubricating oil additives that are not specifically recited in the particular section of the specification or the claims. For example, other lubricating oil additives may include, but are not limited to, antioxidants, detergents, dispersants, antiwear additives, corrosion inhibitors, viscosity modifiers, metal passivators, pour point depressants, seal compatibility agents, antifoam agents, extreme pressure agents, friction modifiers and combinations thereof.
[0021] It has now been found that prevention or reduction of LSPI can be attained in an engine lubricated with a lubricating oil by using as the lubricating oil a formulated oil that includes at least one metal or metalloid in an amount sufficient that, in thermogravimetric measurements of the lubricating oil by a Thermogravimetric Engine Oil Simulation Test as described herein, carbon black decomposition temperature is increased as compared to the carbon black decomposition temperature in a lubricating oil containing calcium or magnesium.
[0022] In an embodiment, in thermogravimetric measurements of the lubricating oil by a Thermogravimetric Engine Oil Simulation Test, the carbon black decomposition temperature is increased by at least 10.degree. C. as compared to the carbon black decomposition temperature in a lubricating oil containing calcium or magnesium.
[0023] In another embodiment, in thermogravimetric measurements of the lubricating oil by a Thermogravimetric Engine Oil Simulation Test, the carbon black decomposition temperature is increased by at least 25.degree. C. as compared to the carbon black decomposition temperature in a lubricating oil containing calcium or magnesium.
[0024] Illustrative metals and metalloids useful in the lubricating oils of this disclosure include, for example, sodium, manganese, titanium, aluminum, or mixtures thereof.
[0025] The at least one metal or metalloid is present in the lubricating oils of this disclosure in an amount from about 10 ppm to about 25,000 ppm, preferably from about 50 ppm to about 10,000 ppm, and more preferably from about 100 ppm to about 5,000 ppm, and more preferably from about 100 ppm to about 1,000 ppm, and more preferably from about 100 ppm to about 500 ppm.
[0026] In an embodiment, the total base number (TBN), as measured by ASTM D2896, of the lubricating oil ranges from about 2 mg KOH to about 17 mg KOH, preferably from about 3 mg KOH to about 16 mg KOH, and more preferably from about 4 mg KOH to about 16 mg KOH.
[0027] In a further embodiment, the total sulfated ash of the lubricating oil ranges from about 0.01 to about 1 wt %, preferably from about 0.01 to about 0.8 wt %, more preferably from about 0.01 to about 0.6 wt %, more preferably from about 0.01 to about 0.4 wt %, more preferably from about 0.01 to about 0.17 wt %, more preferably from about 0.05 to about 0.16 wt %, and more preferably from about 0.1 to about 0.15 wt %.
[0028] The at least one metal or metalloid can be incorporated into the lubricating oil by several methods, for example, by maintaining constant ash, maintaining constant detergent metal, maintaining constant total base number (TBN), top treating, and the like.
[0029] Preferably, the at least one metal or metalloid can be incorporated into the lubricating oil through at least one lubricating oil additive (e.g., metal-containing detergent). The at least one lubricating oil additive is preferably selected from a salicylate, a carboxylate, a sulfonate, a phenate, a stearate, an oxide, and the like.
[0030] The at least one lubricating oil additive can have a total sulfated ash content from about 0.02 to about 0.17 wt %, preferably from about 0.05 to about 0.16 wt %, and more preferably from about 0.1 to about 0.15 wt %.
[0031] In addition, it has been found that reduction of LSPI can be attained in an engine lubricated with a lubricating oil by using as the lubricating oil a formulated oil that has a particular base stock (e.g., Group IV base stock) and at least one metal or metalloid in an amount from about 100 ppm to about 25,000 ppm. The lubricating oils of this disclosure are particularly advantageous in internal combustion engines using a variety of fuels including natural gas, gasoline, diesel, biofuels and the like, and for a variety of applications including passenger vehicle engine oils and natural gas engine oils.
[0032] The lubricating oils of this disclosure are particularly useful in internal combustion engines and, when used in internal combustion engines, will prevent or minimize pre-ignition problems. The lubricating oil compositions of this disclosure are useful in lubricating gasoline-fueled, spark-ignited engines.
[0033] As described herein, the lubricant formulation chemistry of this disclosure can be used to prevent or control the detrimental effect of LSPI in engines which have already been designed or sold in the marketplace as well as future engine technology. The lubricant formulation chemistry of this disclosure removes barriers for OEM technology and efficiency improvement, and enables further development of downspeeded, turbocharged gasoline engines that is currently being impeded by LSPI. The lubricant formulation solution afforded by this disclosure for preventing or reducing LSPI enables product differentiation with regard to LSPI.
[0034] Lubricating Oil Base Stocks
[0035] A wide range of lubricating base oils is known in the art. Lubricating base oils that are useful in the present disclosure are both natural oils, and synthetic oils, and unconventional oils (or mixtures thereof) can be used unrefined, refined, or rerefined (the latter is also known as reclaimed or reprocessed oil). Unrefined oils are those obtained directly from a natural or synthetic source and used without added purification. These include shale oil obtained directly from retorting operations, petroleum oil obtained directly from primary distillation, and ester oil obtained directly from an esterification process. Refined oils are similar to the oils discussed for unrefined oils except refined oils are subjected to one or more purification steps to improve at least one lubricating oil property. One skilled in the art is familiar with many purification processes. These processes include solvent extraction, secondary distillation, acid extraction, base extraction, filtration, and percolation. Rerefined oils are obtained by processes analogous to refined oils but using an oil that has been previously used as a feed stock.
[0036] Groups I, II, III, IV, and V are broad base oil stock categories developed and defined by the American Petroleum Institute (API Publication 1509; www.API.org) to create guidelines for lubricant base oils. Group I base stocks have a viscosity index of between 80 to 120 and contain greater than 0.03% sulfur and/or less than 90% saturates. Group II base stocks have a viscosity index of between about 80 to 120, and contain less than or equal to 0.03% sulfur and greater than or equal to 90% saturates. Group III stocks have a viscosity index greater than or equal to 120 and contain less than or equal to 0.03% sulfur and greater than or equal to 90% saturates. Group IV includes polyalphaolefins (PAO). Group V base stock includes base stocks not included in Groups I-IV. The table below summarizes properties of each of these five groups.
TABLE-US-00001 Base Oil Properties Saturates Sulfur Viscosity Index Group I <90 and/or >0.03% and .gtoreq.80 and <120 Group II .gtoreq.90 and .ltoreq.0.03% and .gtoreq.80 and <120 Group III .gtoreq.90 and .ltoreq.0.03% and .gtoreq.120 Group IV Includes polyalphaolefins (PAO) Group V All other base oil stocks not included in Groups I, II, III, or IV
[0037] Natural oils include animal oils, vegetable oils (castor oil and lard oil, for example), and mineral oils. Animal and vegetable oils possessing favorable thermal oxidative stability can be used. Of the natural oils, mineral oils are preferred. Mineral oils vary widely as to their crude source, for example, as to whether they are paraffinic, naphthenic, or mixed paraffinic-naphthenic. Oils derived from coal or shale are also useful. Natural oils vary also as to the method used for their production and purification, for example, their distillation range and whether they are straight run or cracked, hydrorefined, or solvent extracted.
[0038] Group II and/or Group III hydroprocessed or hydrocracked base stocks, including synthetic oils such as polyalphaolefins, alkyl aromatics and synthetic esters are also well known base stock oils.
[0039] Synthetic oils include hydrocarbon oil. Hydrocarbon oils include oils such as polymerized and interpolymerized olefins (polybutylenes, polypropylenes, propylene isobutylene copolymers, ethylene-olefin copolymers, and ethylene-alphaolefin copolymers, for example). Polyalphaolefin (PAO) oil base stocks are commonly used synthetic hydrocarbon oil. By way of example, PAOs derived from C.sub.6, C.sub.8, C.sub.10, C.sub.12, C.sub.14 olefins or mixtures thereof may be utilized. See U.S. Pat. Nos. 4,956,122; 4,827,064; and 4,827,073.
[0040] The number average molecular weights of the PAOs, which are known materials and generally available on a major commercial scale from suppliers such as ExxonMobil Chemical Company, Chevron Phillips Chemical Company, BP, and others, typically vary from about 250 to about 3,000, although PAO's may be made in viscosities up to about 150 cSt (100.degree. C.). The PAOs are typically comprised of relatively low molecular weight hydrogenated polymers or oligomers of alphaolefins which include, but are not limited to, C.sub.2 to about C.sub.32 alphaolefins with the C.sub.6 to about C.sub.16 alphaolefins, such as 1-hexene, 1-octene, 1-decene, 1-dodecene, 1-tetradocene, and the like, being preferred. The preferred polyalphaolefins are poly-1-hexene, poly-1-octene, poly-1-decene and poly-1-dodecene and mixtures thereof and mixed olefin-derived polyolefins. However, the dimers of higher olefins in the range of C.sub.14 to C.sub.18 may be used to provide low viscosity base stocks of acceptably low volatility. Depending on the viscosity grade and the starting oligomer, the PAOs may be predominantly trimers and tetramers of the starting olefins, with minor amounts of the higher oligomers, having a viscosity range of 1.5 to 12 cSt. PAO fluids of particular use may include 3.0 cSt, 3.4 cSt, and/or 3.6 cSt and combinations thereof. Bi-modal mixtures of PAO fluids having a viscosity range of 1.5 to 150 cSt may be used if desired.
[0041] The PAO fluids may be conveniently made by the polymerization of an alphaolefin in the presence of a polymerization catalyst such as the Friedel-Crafts catalysts including, for example, aluminum trichloride, boron trifluoride or complexes of boron trifluoride with water, alcohols such as ethanol, propanol or butanol, carboxylic acids or esters such as ethyl acetate or ethyl propionate. For example the methods disclosed by U.S. Pat. No. 4,149,178 or 3,382,291 may be conveniently used herein. Other descriptions of PAO synthesis are found in the following U.S. Pat. Nos. 3,742,082; 3,769,363; 3,876,720; 4,239,930; 4,367,352; 4,413,156; 4,434,408; 4,910,355; 4,956,122; and 5,068,487. The dimers of the C.sub.14 to C.sub.18 olefins are described in U.S. Pat. No. 4,218,330.
[0042] Other useful lubricant oil base stocks include wax isomerate base stocks and base oils, comprising hydroisomerized waxy stocks (e.g. waxy stocks such as gas oils, slack waxes, fuels hydrocracker bottoms, etc.), hydroisomerized Fischer-Tropsch waxes, Gas-to-Liquids (GTL) base stocks and base oils, and other wax isomerate hydroisomerized base stocks and base oils, or mixtures thereof Fischer-Tropsch waxes, the high boiling point residues of Fischer-Tropsch synthesis, are highly paraffinic hydrocarbons with very low sulfur content. The hydroprocessing used for the production of such base stocks may use an amorphous hydrocracking/hydroisomerization catalyst, such as one of the specialized lube hydrocracking (LHDC) catalysts or a crystalline hydrocracking/hydroisomerization catalyst, preferably a zeolitic catalyst. For example, one useful catalyst is ZSM-48 as described in U.S. Pat. No. 5,075,269, the disclosure of which is incorporated herein by reference in its entirety. Processes for making hydrocracked/hydroisomerized distillates and hydrocracked/hydroisomerized waxes are described, for example, in U.S. Pat. Nos. 2,817,693; 4,975,177; 4,921,594 and 4,897,178 as well as in British Patent Nos. 1,429,494; 1,350,257; 1,440,230 and 1,390,359. Each of the aforementioned patents is incorporated herein in their entirety. Particularly favorable processes are described in European Patent Application Nos. 464546 and 464547, also incorporated herein by reference. Processes using Fischer-Tropsch wax feeds are described in U.S. Pat. Nos. 4,594,172 and 4,943,672, the disclosures of which are incorporated herein by reference in their entirety.
[0043] Gas-to-Liquids (GTL) base oils, Fischer-Tropsch wax derived base oils, and other wax-derived hydroisomerized (wax isomerate) base oils be advantageously used in the instant disclosure, and may have useful kinematic viscosities at 100.degree. C. of about 3 cSt to about 50 cSt, preferably about 3 cSt to about 30 cSt, more preferably about 3.5 cSt to about 25 cSt, as exemplified by GTL 4 with kinematic viscosity of about 4.0 cSt at 100.degree. C. and a viscosity index of about 141. These Gas-to-Liquids (GTL) base oils, Fischer-Tropsch wax derived base oils, and other wax-derived hydroisomerized base oils may have useful pour points of about -20.degree. C. or lower, and under some conditions may have advantageous pour points of about -25.degree. C. or lower, with useful pour points of about -30.degree. C. to about -40.degree. C. or lower. Useful compositions of Gas-to-Liquids (GTL) base oils, Fischer-Tropsch wax derived base oils, and wax-derived hydroisomerized base oils are recited in U.S. Pat. Nos. 6,080,301; 6,090,989, and 6,165,949 for example, and are incorporated herein in their entirety by reference.
[0044] The hydrocarbyl aromatics can be used as base oil or base oil component and can be any hydrocarbyl molecule that contains at least about 5% of its weight derived from an aromatic moiety such as a benzenoid moiety or naphthenoid moiety, or their derivatives. These hydrocarbyl aromatics include alkyl benzenes, alkyl naphthalenes, alkyl diphenyl oxides, alkyl naphthols, alkyl diphenyl sulfides, alkylated bis-phenol A, alkylated thiodiphenol, and the like. The aromatic can be mono-alkylated, dialkylated, polyalkylated, and the like. The aromatic can be mono- or poly-functionalized. The hydrocarbyl groups can also be comprised of mixtures of alkyl groups, alkenyl groups, alkynyl, cycloalkyl groups, cycloalkenyl groups and other related hydrocarbyl groups. The hydrocarbyl groups can range from about C.sub.6 up to about C.sub.60 with a range of about C.sub.8 to about C.sub.20 often being preferred. A mixture of hydrocarbyl groups is often preferred, and up to about three such substituents may be present. The hydrocarbyl group can optionally contain sulfur, oxygen, and/or nitrogen containing substituents. The aromatic group can also be derived from natural (petroleum) sources, provided at least about 5% of the molecule is comprised of an above-type aromatic moiety. Viscosities at 100.degree. C. of approximately 3 cSt to about 50 cSt are preferred, with viscosities of approximately 3.4 cSt to about 20 cSt often being more preferred for the hydrocarbyl aromatic component. In one embodiment, an alkyl naphthalene where the alkyl group is primarily comprised of 1-hexadecene is used. Other alkylates of aromatics can be advantageously used. Naphthalene or methyl naphthalene, for example, can be alkylated with olefins such as octene, decene, dodecene, tetradecene or higher, mixtures of similar olefins, and the like. Useful concentrations of hydrocarbyl aromatic in a lubricant oil composition can be about 2% to about 25%, preferably about 4% to about 20%, and more preferably about 4% to about 15%, depending on the application.
[0045] Alkylated aromatics such as the hydrocarbyl aromatics of the present disclosure may be produced by well-known Friedel-Crafts alkylation of aromatic compounds. See Friedel-Crafts and Related Reactions, Olah, G. A. (ed.), Inter-science Publishers, New York, 1963. For example, an aromatic compound, such as benzene or naphthalene, is alkylated by an olefin, alkyl halide or alcohol in the presence of a Friedel-Crafts catalyst. See Friedel-Crafts and Related Reactions, Vol. 2, part 1, chapters 14, 17, and 18, See Olah, G. A. (ed.), Inter-science Publishers, New York, 1964. Many homogeneous or heterogeneous, solid catalysts are known to one skilled in the art. The choice of catalyst depends on the reactivity of the starting materials and product quality requirements. For example, strong acids such as AlCl.sub.3, BF.sub.3, or HF may be used. In some cases, milder catalysts such as FeCl.sub.3 or SnCl.sub.4 are preferred. Newer alkylation technology uses zeolites or solid super acids.
[0046] Esters comprise a useful base stock. Additive solvency and seal compatibility characteristics may be secured by the use of esters such as the esters of dibasic acids with monoalkanols and the polyol esters of mono-carboxylic acids. Esters of the former type include, for example, the esters of dicarboxylic acids such as phthalic acid, succinic acid, alkyl succinic acid, alkenyl succinic acid, maleic acid, azelaic acid, suberic acid, sebacic acid, fumaric acid, adipic acid, linoleic acid dimer, malonic acid, alkyl malonic acid, alkenyl malonic acid, etc., with a variety of alcohols such as butyl alcohol, hexyl alcohol, dodecyl alcohol, 2-ethylhexyl alcohol, etc. Specific examples of these types of esters include dibutyl adipate, di(2-ethylhexyl) sebacate, di-n-hexyl fumarate, dioctyl sebacate, diisooctyl azelate, diisodecyl azelate, dioctyl phthalate, didecyl phthalate, dieicosyl sebacate, etc.
[0047] Particularly useful synthetic esters are those which are obtained by reacting one or more polyhydric alcohols, preferably the hindered polyols (such as the neopentyl polyols, e.g., neopentyl glycol, trimethylol ethane, 2-methyl-2-propyl-1,3-propanediol, trimethylol propane, pentaerythritol and dipentaerythritol) with alkanoic acids containing at least about 4 carbon atoms, preferably C.sub.5 to C.sub.30 acids such as saturated straight chain fatty acids including caprylic acid, capric acid, lauric acid, myristic acid, palmitic acid, stearic acid, arachic acid, and behenic acid, or the corresponding branched chain fatty acids or unsaturated fatty acids such as oleic acid, or mixtures of any of these materials.
[0048] Suitable synthetic ester components include the esters of trimethylol propane, trimethylol butane, trimethylol ethane, pentaerythritol and/or dipentaerythritol with one or more monocarboxylic acids containing from about 5 to about 10 or more carbon atoms. These esters are widely available commercially, for example, the Mobil P-41 and P-51 esters of ExxonMobil Chemical Company.
[0049] Preferred synthetic esters useful in this disclosure have a kinematic viscosity at 100.degree. C. of about 3 cSt to about 50 cSt, preferably about 3 cSt to about 30 cSt, more preferably about 3.5 cSt to about 25 cSt, and even more preferably about 2 cSt to about 8 cSt. Group V base oils useful in this disclosure preferably comprise an ester at a concentration of about 2% to about 20%, preferably from about 5% to about 15%.
[0050] Also useful are esters derived from renewable material such as coconut, palm, rapeseed, soy, sunflower and the like. These esters may be monoesters, di-esters, polyol esters, complex esters, or mixtures thereof. These esters are widely available commercially, for example, the Mobil P-51 ester of ExxonMobil Chemical Company.
[0051] Engine oil formulations containing renewable esters are included in this disclosure. For such formulations, the renewable content of the ester is typically greater than about 70 weight percent, preferably more than about 80 weight percent and most preferably more than about 90 weight percent.
[0052] Other useful fluids of lubricating viscosity include non-conventional or unconventional base stocks that have been processed, preferably catalytically, or synthesized to provide high performance lubrication characteristics.
[0053] Non-conventional or unconventional base stocks/base oils include one or more of a mixture of base stock(s) derived from one or more Gas-to-Liquids (GTL) materials, as well as isomerate/isodewaxate base stock(s) derived from natural wax or waxy feeds, mineral and or non-mineral oil waxy feed stocks such as slack waxes, natural waxes, and waxy stocks such as gas oils, waxy fuels hydrocracker bottoms, waxy raffinate, hydrocrackate, thermal crackates, or other mineral, mineral oil, or even non-petroleum oil derived waxy materials such as waxy materials received from coal liquefaction or shale oil, and mixtures of such base stocks.
[0054] GTL materials are materials that are derived via one or more synthesis, combination, transformation, rearrangement, and/or degradation/deconstructive processes from gaseous carbon-containing compounds, hydrogen-containing compounds and/or elements as feed stocks such as hydrogen, carbon dioxide, carbon monoxide, water, methane, ethane, ethylene, acetylene, propane, propylene, propyne, butane, butylenes, and butynes. GTL base stocks and/or base oils are GTL materials of lubricating viscosity that are generally derived from hydrocarbons; for example, waxy synthesized hydrocarbons, that are themselves derived from simpler gaseous carbon-containing compounds, hydrogen-containing compounds and/or elements as feed stocks. GTL base stock(s) and/or base oil(s) include oils boiling in the lube oil boiling range (1) separated/fractionated from synthesized GTL materials such as, for example, by distillation and subsequently subjected to a final wax processing step which involves either or both of a catalytic dewaxing process, or a solvent dewaxing process, to produce lube oils of reduced/low pour point; (2) synthesized wax isomerates, comprising, for example, hydrodewaxed or hydroisomerized cat and/or solvent dewaxed synthesized wax or waxy hydrocarbons; (3) hydrodewaxed or hydroisomerized cat and/or solvent dewaxed Fischer-Tropsch (F-T) material (i.e., hydrocarbons, waxy hydrocarbons, waxes and possible analogous oxygenates); preferably hydrodewaxed or hydroisomerized/followed by cat and/or solvent dewaxing dewaxed F-T waxy hydrocarbons, or hydrodewaxed or hydroisomerized/followed by cat (or solvent) dewaxing dewaxed, F-T waxes, or mixtures thereof.
[0055] GTL base stock(s) and/or base oil(s) derived from GTL materials, especially, hydrodewaxed or hydroisomerized/followed by cat and/or solvent dewaxed wax or waxy feed, preferably F-T material derived base stock(s) and/or base oil(s), are characterized typically as having kinematic viscosities at 100.degree. C. of from about 2 mm.sup.2/s to about 50 mm.sup.2/s (ASTM D445). They are further characterized typically as having pour points of -5.degree. C. to about -40.degree. C. or lower (ASTM D97). They are also characterized typically as having viscosity indices of about 80 to about 140 or greater (ASTM D2270).
[0056] The term GTL base stock and/or base oil and/or wax isomerate base stock and/or base oil is to be understood as embracing individual fractions of such materials of wide viscosity range as recovered in the production process, mixtures of two or more of such fractions, as well as mixtures of one or two or more low viscosity fractions with one, two or more higher viscosity fractions to produce a blend wherein the blend exhibits a target kinematic viscosity.
[0057] The GTL material, from which the GTL base stock(s) and/or base oil(s) is/are derived is preferably an F-T material (i.e., hydrocarbons, waxy hydrocarbons, wax).
[0058] In addition, the GTL base stock(s) and/or base oil(s) are typically highly paraffinic (>90% saturates), and may contain mixtures of monocycloparaffins and multicycloparaffins in combination with non-cyclic isoparaffins. The ratio of the naphthenic (i.e., cycloparaffin) content in such combinations varies with the catalyst and temperature used. Further, GTL base stock(s) and/or base oil(s) and hydrodewaxed, or hydroisomerized/cat (and/or solvent) dewaxed base stock(s) and/or base oil(s) typically have very low sulfur and nitrogen content, generally containing less than about 10 ppm, and more typically less than about 5 ppm of each of these elements. The sulfur and nitrogen content of GTL base stock(s) and/or base oil(s) obtained from F-T material, especially F-T wax, is essentially nil. In addition, the absence of phosphorous and aromatics make this material especially suitable for the formulation of low sulfur, sulfated ash, and phosphorus (low SAP) products.
[0059] Base oils for use in the formulated lubricating oils useful in the present disclosure are any of the variety of oils corresponding to API Group I, Group II, Group III, Group IV, and Group V oils and mixtures thereof, preferably API Group II, Group III, Group IV, and Group V oils and mixtures thereof, more preferably the Group III to Group V base oils due to their exceptional volatility, stability, viscometric and cleanliness features.
[0060] The base oil constitutes the major component of the engine oil lubricant composition of the present disclosure and typically is present in an amount ranging from about 50 to about 99 weight percent, preferably from about 70 to about 95 weight percent, and more preferably from about 85 to about 95 weight percent, based on the total weight of the composition. The base oil may be selected from any of the synthetic or natural oils typically used as crankcase lubricating oils for spark-ignited and compression-ignited engines. The base oil conveniently has a kinematic viscosity, according to ASTM standards, of about 2.5 cSt to about 12 cSt (or mm.sup.2/s) at 100.degree. C. and preferably of about 2.5 cSt to about 9 cSt (or mm.sup.2/s) at 100.degree. C. Mixtures of synthetic and natural base oils may be used if desired. Mixtures of Group III, IV, and V may be preferably used if desired.
Lubricating Oil Additives
[0061] The formulated lubricating oil useful in the present disclosure may additionally contain one or more of the other commonly used lubricating oil performance additives including but not limited to antiwear agents, dispersants, detergents, corrosion inhibitors, rust inhibitors, metal deactivators, extreme pressure additives, anti-seizure agents, wax modifiers, viscosity index improvers, viscosity modifiers, fluid-loss additives, seal compatibility agents, friction modifiers, lubricity agents, anti-staining agents, chromophoric agents, defoamants, demulsifiers, emulsifiers, densifiers, wetting agents, gelling agents, tackiness agents, colorants, and others. For a review of many commonly used additives, see Klamann in Lubricants and Related Products, Verlag Chemie, Deerfield Beach, Fla.; ISBN 0-89573-177-0. Reference is also made to "Lubricant Additives" by M. W. Ranney, published by Noyes Data Corporation of Parkridge, N.J. (1973); see also U.S. Pat. No. 7,704,930, the disclosure of which is incorporated herein in its entirety. These additives are commonly delivered with varying amounts of diluent oil, that may range from 5 weight percent to 50 weight percent.
[0062] The types and quantities of performance additives used in combination with the instant disclosure in lubricant compositions are not limited by the examples shown herein as illustrations.
Antiwear Additives
[0063] A metal alkylthiophosphate and more particularly a metal dialkyl dithio phosphate in which the metal constituent is zinc, or zinc dialkyl dithio phosphate (ZDDP) can be a useful component of the lubricating oils of this disclosure. ZDDP can be derived from primary alcohols, secondary alcohols or mixtures thereof. ZDDP compounds generally are of the formula
Zn[SP(S)(OR.sup.1)(OR.sup.2)].sub.2
where R.sup.1 and R.sup.2 are C.sub.1-C.sub.18 alkyl groups, preferably C.sub.2-C.sub.12 alkyl groups. These alkyl groups may be straight chain or branched. Alcohols used in the ZDDP can be propanol, 2-propanol, butanol, secondary butanol, pentanols, hexanols such as 4-methyl-2-pentanol, n-hexanol, n-octanol, 2-ethyl hexanol, alkylated phenols, and the like. Mixtures of secondary alcohols or of primary and secondary alcohol can be preferred. Alkyl aryl groups may also be used.
[0064] Preferable zinc dithiophosphates which are commercially available include secondary zinc dithiophosphates such as those available from for example, The Lubrizol Corporation under the trade designations "LZ 677A", "LZ 1095" and "LZ 1371", from for example Chevron Oronite under the trade designation "OLOA 262" and from for example Afton Chemical under the trade designation "HITEC 7169".
[0065] The ZDDP is typically used in amounts of from about 0.3 weight percent to about 1.5 weight percent, preferably from about 0.4 weight percent to about 1.2 weight percent, more preferably from about 0.5 weight percent to about 1.0 weight percent, and even more preferably from about 0.6 weight percent to about 0.8 weight percent, based on the total weight of the lubricating oil, although more or less can often be used advantageously. Preferably, the ZDDP is a secondary ZDDP and present in an amount of from about 0.6 to 1.0 weight percent of the total weight of the lubricating oil.
Dispersants
[0066] During engine operation, oil-insoluble oxidation byproducts are produced. Dispersants help keep these byproducts in solution, thus diminishing their deposition on metal surfaces. Dispersants used in the formulation of the lubricating oil may be ashless or ash-forming in nature. Preferably, the dispersant is ashless. So called ashless dispersants are organic materials that form substantially no ash upon combustion. For example, non-metal-containing or borated metal-free dispersants are considered ashless. In contrast, metal-containing detergents discussed above form ash upon combustion.
[0067] Suitable dispersants typically contain a polar group attached to a relatively high molecular weight hydrocarbon chain. The polar group typically contains at least one element of nitrogen, oxygen, or phosphorus. Typical hydrocarbon chains contain 50 to 400 carbon atoms.
[0068] A particularly useful class of dispersants are the (poly)alkenylsuccinic derivatives, typically produced by the reaction of a long chain hydrocarbyl substituted succinic compound, usually a hydrocarbyl substituted succinic anhydride, with a polyhydroxy or polyamino compound. The long chain hydrocarbyl group constituting the oleophilic portion of the molecule which confers solubility in the oil, is normally a polyisobutylene group. Many examples of this type of dispersant are well known commercially and in the literature. Exemplary U.S. patents describing such dispersants are U.S. Pat. Nos. 3,172,892; 3,2145,707; 3,219,666; 3,316,177; 3,341,542; 3,444,170; 3,454,607; 3,541,012; 3,630,904; 3,632,511; 3,787,374 and 4,234,435. Other types of dispersant are described in U.S. Pat. Nos. 3,036,003; 3,200,107; 3,254,025; 3,275,554; 3,438,757; 3,454,555; 3,565,804; 3,413,347; 3,697,574; 3,725,277; 3,725,480; 3,726,882; 4,454,059; 3,329,658; 3,449,250; 3,519,565; 3,666,730; 3,687,849; 3,702,300; 4,100,082; 5,705,458. A further description of dispersants may be found, for example, in European Patent Application No. 471 071, to which reference is made for this purpose.
[0069] Hydrocarbyl-substituted succinic acid and hydrocarbyl-substituted succinic anhydride derivatives are useful dispersants. In particular, succinimide, succinate esters, or succinate ester amides prepared by the reaction of a hydrocarbon-substituted succinic acid compound preferably having at least 50 carbon atoms in the hydrocarbon substituent, with at least one equivalent of an alkylene amine are particularly useful.
[0070] Succinimides are formed by the condensation reaction between hydrocarbyl substituted succinic anhydrides and amines. Molar ratios can vary depending on the polyamine. For example, the molar ratio of hydrocarbyl substituted succinic anhydride to TEPA can vary from about 1:1 to about 5:1. Representative examples are shown in U.S. Pat. Nos. 3,087,936; 3,172,892; 3,219,666; 3,272,746; 3,322,670; and 3,652,616, 3,948,800; and Canada Patent No. 1,094,044.
[0071] Succinate esters are formed by the condensation reaction between hydrocarbyl substituted succinic anhydrides and alcohols or polyols. Molar ratios can vary depending on the alcohol or polyol used. For example, the condensation product of a hydrocarbyl substituted succinic anhydride and pentaerythritol is a useful dispersant.
[0072] Succinate ester amides are formed by condensation reaction between hydrocarbyl substituted succinic anhydrides and alkanol amines. For example, suitable alkanol amines include ethoxylated polyalkylpolyamines, propoxylated polyalkylpolyamines and polyalkenylpolyamines such as polyethylene polyamines. One example is propoxylated hexamethylenediamine. Representative examples are shown in U.S. Pat. No. 4,426,305.
[0073] The molecular weight of the hydrocarbyl substituted succinic anhydrides used in the preceding paragraphs will typically range between 800 and 2,500 or more. The above products can be post-reacted with various reagents such as sulfur, oxygen, formaldehyde, carboxylic acids such as oleic acid. The above products can also be post reacted with boron compounds such as boric acid, borate esters or highly borated dispersants, to form borated dispersants generally having from about 0.1 to about 5 moles of boron per mole of dispersant reaction product.
[0074] Mannich base dispersants are made from the reaction of alkylphenols, formaldehyde, and amines. See U.S. Pat. No. 4,767,551, which is incorporated herein by reference. Process aids and catalysts, such as oleic acid and sulfonic acids, can also be part of the reaction mixture. Molecular weights of the alkylphenols range from 800 to 2,500. Representative examples are shown in U.S. Pat. Nos. 3,697,574; 3,703,536; 3,704,308; 3,751,365; 3,756,953; 3,798,165; and 3,803,039.
[0075] Typical high molecular weight aliphatic acid modified Mannich condensation products useful in this disclosure can be prepared from high molecular weight alkyl-substituted hydroxyaromatics or HNR2 group-containing reactants.
[0076] Hydrocarbyl substituted amine ashless dispersant additives are well known to one skilled in the art; see, for example, U.S. Pat. Nos. 3,275,554; 3,438,757; 3,565,804; 3,755,433, 3,822,209, and 5,084,197.
[0077] Preferred dispersants include borated and non-borated succinimides, including those derivatives from mono-succinimides, bis-succinimides, and/or mixtures of mono- and bis-succinimides, wherein the hydrocarbyl succinimide is derived from a hydrocarbylene group such as polyisobutylene having a Mn of from about 500 to about 5000, or from about 1000 to about 3000, or about 1000 to about 2000, or a mixture of such hydrocarbylene groups, often with high terminal vinylic groups. Other preferred dispersants include succinic acid-esters and amides, alkylphenol-polyamine-coupled Mannich adducts, their capped derivatives, and other related components.
[0078] Polymethacrylate or polyacrylate derivatives are another class of dispersants. These dispersants are typically prepared by reacting a nitrogen containing monomer and a methacrylic or acrylic acid esters containing 5-25 carbon atoms in the ester group. Representative examples are shown in U.S. Pat. Nos. 2,100,993, and 6,323,164. Polymethacrylate and polyacrylate dispersants are normally used as multifunctional viscosity modifiers. The lower molecular weight versions can be used as lubricant dispersants or fuel detergents.
[0079] Illustrative preferred dispersants useful in this disclosure include those derived from polyalkenyl-substituted mono- or dicarboxylic acid, anhydride or ester, which dispersant has a polyalkenyl moiety with a number average molecular weight of at least 900 and from greater than 1.3 to 1.7, preferably from greater than 1.3 to 1.6, most preferably from greater than 1.3 to 1.5, functional groups (mono- or dicarboxylic acid producing moieties) per polyalkenyl moiety (a medium functionality dispersant). Functionality (F) can be determined according to the following formula:
F=(SAP.times.M.sub.n)/((112,200.times.A.I.)-(SAP.times.98))
wherein SAP is the saponification number (i.e., the number of milligrams of KOH consumed in the complete neutralization of the acid groups in one gram of the succinic-containing reaction product, as determined according to ASTM D94); M.sub.n is the number average molecular weight of the starting olefin polymer; and A.I, is the percent active ingredient of the succinic-containing reaction product (the remainder being unreacted olefin polymer, succinic anhydride and diluent).
[0080] The polyalkenyl moiety of the dispersant may have a number average molecular weight of at least 900, suitably at least 1500, preferably between 1800 and 3000, such as between 2000 and 2800, more preferably from about 2100 to 2500, and most preferably from about 2200 to about 2400. The molecular weight of a dispersant is generally expressed in terms of the molecular weight of the polyalkenyl moiety. This is because the precise molecular weight range of the dispersant depends on numerous parameters including the type of polymer used to derive the dispersant, the number of functional groups, and the type of nucleophilic group employed.
[0081] Polymer molecular weight, specifically M.sub.n, can be determined by various known techniques. One convenient method is gel permeation chromatography (GPC), which additionally provides molecular weight distribution information (see W. W. Yau, J. J. Kirkland and D. D. Bly, "Modern Size Exclusion Liquid Chromatography", John Wiley and Sons, New York, 1979). Another useful method for determining molecular weight, particularly for lower molecular weight polymers, is vapor pressure osmometry (e.g., ASTM D3592).
[0082] The polyalkenyl moiety in a dispersant preferably has a narrow molecular weight distribution (MWD), also referred to as polydispersity, as determined by the ratio of weight average molecular weight (M.sub.w) to number average molecular weight (M.sub.n). Polymers having a M.sub.w/M.sub.n of less than 2.2, preferably less than 2.0, are most desirable. Suitable polymers have a polydispersity of from about 1.5 to 2.1, preferably from about 1.6 to about 1.8.
[0083] Suitable polyalkenes employed in the formation of the dispersants include homopolymers, interpolymers or lower molecular weight hydrocarbons. One family of such polymers comprise polymers of ethylene and/or at least one C.sub.3 to C.sub.2 alpha-olefin having the formula H.sub.2C=CHR.sup.1 wherein R.sup.1 is a straight or branched chain alkyl radical comprising 1 to 26 carbon atoms and wherein the polymer contains carbon-to-carbon unsaturation, and a high degree of terminal ethenylidene unsaturation. Preferably, such polymers comprise interpolymers of ethylene and at least one alpha-olefin of the above formula, wherein R.sup.1 is alkyl of from 1 to 18 carbon atoms, and more preferably is alkyl of from 1 to 8 carbon atoms, and more preferably still of from 1 to 2 carbon atoms.
[0084] Another useful class of polymers is polymers prepared by cationic polymerization of monomers such as isobutene and styrene. Common polymers from this class include polyisobutenes obtained by polymerization of a C.sub.4 refinery stream having a butene content of 35 to 75% by wt., and an isobutene content of 30 to 60% by wt. A preferred source of monomer for making poly-n-butenes is petroleum feedstreams such as Raffinate II. These feed stocks are disclosed in the art such as in U.S. Pat. No. 4,952,739. A preferred embodiment utilizes polyisobutylene prepared from a pure isobutylene stream or a Raffinate I stream to prepare reactive isobutylene polymers with terminal vinylidene olefins. Polyisobutene polymers that may be employed are generally based on a polymer chain of from 1500 to 3000.
[0085] The dispersant(s) are preferably non-polymeric (e.g., mono- or bis-succinimides). Such dispersants can be prepared by conventional processes such as disclosed in U.S. Patent Application Publication No. 2008/0020950, the disclosure of which is incorporated herein by reference.
[0086] The dispersant(s) can be borated by conventional means, as generally disclosed in U.S. Pat. Nos. 3,087,936, 3,254,025 and 5,430,105.
[0087] Such dispersants may be used in an amount of about 0.01 to 20 weight percent or 0.01 to 10 weight percent, preferably about 0.5 to 8 weight percent, or more preferably 0.5 to 4 weight percent. Or such dispersants may be used in an amount of about 2 to 12 weight percent, preferably about 4 to 10 weight percent, or more preferably 6 to 9 weight percent. On an active ingredient basis, such additives may be used in an amount of about 0.06 to 14 weight percent, preferably about 0.3 to 6 weight percent. The hydrocarbon portion of the dispersant atoms can range from C.sub.60 to C.sub.1000, or from C.sub.70 to C.sub.300, or from C.sub.70 to C.sub.200. These dispersants may contain both neutral and basic nitrogen, and mixtures of both. Dispersants can be end-capped by borates and/or cyclic carbonates. Nitrogen content in the finished oil can vary from about 200 ppm by weight to about 2000 ppm by weight, preferably from about 200 ppm by weight to about 1200 ppm by weight. Basic nitrogen can vary from about 100 ppm by weight to about 1000 ppm by weight, preferably from about 100 ppm by weight to about 600 ppm by weight.
[0088] Dispersants as described herein are beneficially useful with the compositions of this disclosure and substitute for some or all of the surfactants of this disclosure. Further, in one embodiment, preparation of the compositions of this disclosure using one or more dispersants is achieved by combining ingredients of this disclosure, plus optional base stocks and lubricant additives, in a mixture at a temperature above the melting point of such ingredients, particularly that of the one or more M-carboxylates (M=H, metal, two or more metals, mixtures thereof).
[0089] As used herein, the dispersant concentrations are given on an "as delivered" basis. Typically, the active dispersant is delivered with a process oil. The "as delivered" dispersant typically contains from about 20 weight percent to about 80 weight percent, or from about 40 weight percent to about 60 weight percent, of active dispersant in the "as delivered" dispersant product.
Detergents
[0090] Illustrative detergents useful in this disclosure include, for example, alkali metal detergents, alkaline earth metal detergents, or mixtures of one or more alkali metal detergents and one or more alkaline earth metal detergents. A typical detergent is an anionic material that contains a long chain hydrophobic portion of the molecule and a smaller anionic or oleophobic hydrophilic portion of the molecule. The anionic portion of the detergent is typically derived from an organic acid such as a sulfur-containing acid, carboxylic acid (e.g., salicylic acid), phosphorus-containing acid, phenol, or mixtures thereof. The counterion is typically an alkaline earth or alkali metal. The detergent can be overbased as described herein.
[0091] The detergent is preferably a metal salt of an organic or inorganic acid, a metal salt of a phenol, or mixtures thereof. The metal is preferably selected from an alkali metal, an alkaline earth metal, and mixtures thereof. The organic or inorganic acid is selected from an aliphatic organic or inorganic acid, a cycloaliphatic organic or inorganic acid, an aromatic organic or inorganic acid, and mixtures thereof.
[0092] The metal is preferably selected from an alkali metal, an alkaline earth metal, and mixtures thereof. More preferably, the metal is selected from calcium (Ca), magnesium (Mg), and mixtures thereof.
[0093] The organic acid or inorganic acid is preferably selected from a sulfur-containing acid, a carboxylic acid, a phosphorus-containing acid, and mixtures thereof.
[0094] Preferably, the metal salt of an organic or inorganic acid or the metal salt of a phenol comprises calcium phenate, calcium sulfonate, calcium salicylate, magnesium phenate, magnesium sulfonate, magnesium salicylate, an overbased detergent, and mixtures thereof.
[0095] Salts that contain a substantially stochiometric amount of the metal are described as neutral salts and have a total base number (TBN, as measured by ASTM D2896) of from 0 to 80. Many compositions are overbased, containing large amounts of a metal base that is achieved by reacting an excess of a metal compound (a metal hydroxide or oxide, for example) with an acidic gas (such as carbon dioxide). Useful detergents can be neutral, mildly overbased, or highly overbased. These detergents can be used in mixtures of neutral, overbased, highly overbased calcium salicylate, sulfonates, phenates and/or magnesium salicylate, sulfonates, phenates. The TBN ranges can vary from low, medium to high TBN products, including as low as 0 to as high as 600. Preferably the TBN delivered by the detergent is between 1 and 20. More preferably between 1 and 12. Mixtures of low, medium, high TBN can be used, along with mixtures of calcium and magnesium metal based detergents, and including sulfonates, phenates, salicylates, and carboxylates. A detergent mixture with a metal ratio of 1, in conjunction of a detergent with a metal ratio of 2, and as high as a detergent with a metal ratio of 5, can be used. Borated detergents can also be used.
[0096] Alkaline earth phenates are another useful class of detergent. These detergents can be made by reacting alkaline earth metal hydroxide or oxide (CaO, Ca(OH).sub.2, BaO, Ba(OH).sub.2, MgO, Mg(OH).sub.2, for example) with an alkyl phenol or sulfurized alkylphenol. Useful alkyl groups include straight chain or branched C.sub.1-C.sub.30 alkyl groups, preferably, C.sub.4-C.sub.20 or mixtures thereof. Examples of suitable phenols include isobutylphenol, 2-ethylhexylphenol, nonylphenol, dodecyl phenol, and the like. It should be noted that starting alkylphenols may contain more than one alkyl substituent that are each independently straight chain or branched and can be used from 0.5 to 6 weight percent. When a non-sulfurized alkylphenol is used, the sulfurized product may be obtained by methods well known in the art. These methods include heating a mixture of alkylphenol and sulfurizing agent (including elemental sulfur, sulfur halides such as sulfur dichloride, and the like) and then reacting the sulfurized phenol with an alkaline earth metal base.
[0097] In accordance with this disclosure, metal salts of carboxylic acids are preferred detergents. These carboxylic acid detergents may be prepared by reacting a basic metal compound with at least one carboxylic acid and removing free water from the reaction product. These compounds may be overbased to produce the desired TBN level. Detergents made from salicylic acid are one preferred class of detergents derived from carboxylic acids. Useful salicylates include long chain alkyl salicylates. One useful family of compositions is of the formula
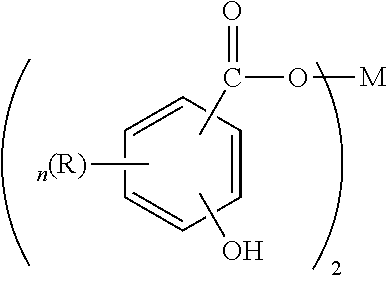
##STR00001##
where R is an alkyl group having 1 to about 30 carbon atoms, n is an integer from 1 to 4, and M is an alkaline earth metal. Preferred R groups are alkyl chains of at least C.sub.11, preferably C.sub.13 or greater. R may be optionally substituted with substituents that do not interfere with the detergent's function. M is preferably, calcium, magnesium, barium, or mixtures thereof. More preferably, M is calcium.
[0098] Hydrocarbyl-substituted salicylic acids may be prepared from phenols by the Kolbe reaction (see U.S. Pat. No. 3,595,791). The metal salts of the hydrocarbyl-substituted salicylic acids may be prepared by double decomposition of a metal salt in a polar solvent such as water or alcohol.
[0099] Alkaline earth metal phosphates are also used as detergents and are known in the art.
[0100] Detergents may be simple detergents or what is known as hybrid or complex detergents. The latter detergents can provide the properties of two detergents without the need to blend separate materials. See U.S. Pat. No. 6,034,039.
[0101] Preferred detergents include calcium sulfonates, magnesium sulfonates, calcium salicylates, magnesium salicylates, calcium phenates, magnesium phenates, and other related components (including borated detergents), and mixtures thereof. Preferred mixtures of detergents include magnesium sulfonate and calcium salicylate, magnesium sulfonate and calcium sulfonate, magnesium sulfonate and calcium phenate, calcium phenate and calcium salicylate, calcium phenate and calcium sulfonate, calcium phenate and magnesium salicylate, calcium phenate and magnesium phenate. Overbased detergents are also preferred.
[0102] The detergent concentration in the lubricating oils of this disclosure can range from about 0.5 to about 6.0 weight percent, preferably about 0.6 to 5.0 weight percent, and more preferably from about 0.8 weight percent to about 4.0 weight percent, based on the total weight of the lubricating oil.
[0103] As used herein, the detergent concentrations are given on an "as delivered" basis. Typically, the active detergent is delivered with a process oil. The "as delivered" detergent typically contains from about 20 weight percent to about 100 weight percent, or from about 40 weight percent to about 60 weight percent, of active detergent in the "as delivered" detergent product.
Viscosity Index Improvers
[0104] Viscosity index improvers (also known as VI improvers, viscosity modifiers, and viscosity improvers) can be included in the lubricant compositions of this disclosure.
[0105] Viscosity index improvers provide lubricants with high and low temperature operability. These additives impart shear stability at elevated temperatures and acceptable viscosity at low temperatures.
[0106] Suitable viscosity index improvers include high molecular weight hydrocarbons, polyesters and viscosity index improver dispersants that function as both a viscosity index improver and a dispersant. Typical molecular weights of these polymers are between about 10,000 to 1,500,000, more typically about 20,000 to 1,200,000, and even more typically between about 50,000 and 1,000,000.
[0107] Examples of suitable viscosity index improvers are linear or star-shaped polymers and copolymers of methacrylate, butadiene, olefins, or alkylated styrenes. Polyisobutylene is a commonly used viscosity index improver. Another suitable viscosity index improver is polymethacrylate (copolymers of various chain length alkyl methacrylates, for example), some formulations of which also serve as pour point depressants. Other suitable viscosity index improvers include copolymers of ethylene and propylene, hydrogenated block copolymers of styrene and isoprene, and polyacrylates (copolymers of various chain length acrylates, for example). Specific examples include styrene-isoprene or styrene-butadiene based polymers of 50,000 to 200,000 molecular weight.
[0108] Olefin copolymers, are commercially available from Chevron Oronite Company LLC under the trade designation "PARATONE.RTM." (such as "PARATONE.RTM. 8921" and "PARATONE.RTM. 8941"); from Afton Chemical Corporation under the trade designation "HiTEC.RTM." (such as "HiTEC.RTM. 5850B"; and from The Lubrizol Corporation under the trade designation "Lubrizol.RTM. 7067C". Polyisoprene polymers are commercially available from Infineum International Limited, e.g. under the trade designation "SV200"; diene-styrene copolymers are commercially available from Infineum International Limited, e.g. under the trade designation "SV 260".
[0109] In an embodiment of this disclosure, the viscosity index improvers may be used in an amount of less than about 2.0 weight percent, preferably less than about 1.0 weight percent, and more preferably less than about 0.5 weight percent, based on the total weight of the formulated oil or lubricating engine oil. Viscosity improvers are typically added as concentrates, in large amounts of diluent oil.
[0110] In another embodiment of this disclosure, the viscosity index improvers may be used in an amount of from 0.25 to about 2.0 weight percent, preferably 0.15 to about 1.0 weight percent, and more preferably 0.05 to about 0.5 weight percent, based on the total weight of the formulated oil or lubricating engine oil.
Antioxidants
[0111] Antioxidants retard the oxidative degradation of base oils during service. Such degradation may result in deposits on metal surfaces, the presence of sludge, or a viscosity increase in the lubricant. One skilled in the art knows a wide variety of oxidation inhibitors that are useful in lubricating oil compositions. See, Klamann in Lubricants and Related Products, op cite, and U.S. Pat. Nos. 4,798,684 and 5,084,197, for example.
[0112] Useful antioxidants include hindered phenols. These phenolic antioxidants may be ashless (metal-free) phenolic compounds or neutral or basic metal salts of certain phenolic compounds. Typical phenolic antioxidant compounds are the hindered phenolics which are the ones which contain a sterically hindered hydroxyl group, and these include those derivatives of dihydroxy aryl compounds in which the hydroxyl groups are in the o- or p-position to each other. Typical phenolic antioxidants include the hindered phenols substituted with C.sub.6+ alkyl groups and the alkylene coupled derivatives of these hindered phenols. Examples of phenolic materials of this type 2-t-butyl-4-heptyl phenol; 2-t-butyl-4-octyl phenol; 2-t-butyl-4-dodecyl phenol; 2,6-di-t-butyl-4-heptyl phenol; 2,6-di-t-butyl-4-dodecyl phenol; 2-methyl-6-t-butyl-4-heptyl phenol; and 2-methyl-6-t-butyl-4-dodecyl phenol. Other useful hindered mono-phenolic antioxidants may include for example hindered 2,6-di-alkyl-phenolic propionic ester derivatives. Bis-phenolic antioxidants may also be advantageously used in combination with the instant disclosure. Examples of ortho-coupled phenols include: 2,2'-bis(4-heptyl-6-t-butyl-phenol); 2,2'-bis(4-octyl-6-t-butyl-phenol); and 2,2'-bis(4-dodecyl-6-t-butyl-phenol). Para-coupled bisphenols include for example 4,4'-bis(2,6-di-t-butyl phenol) and 4,4'-methylene-bis(2,6-di-t-butyl phenol).
[0113] Effective amounts of one or more catalytic antioxidants may also be used. The catalytic antioxidants comprise an effective amount of a) one or more oil soluble polymetal organic compounds; and, effective amounts of b) one or more substituted N,N'-diaryl-o-phenylenediamine compounds or c) one or more hindered phenol compounds; or a combination of both b) and c). Catalytic antioxidants are more fully described in U.S. Pat. No. 8,048,833, herein incorporated by reference in its entirety.
[0114] Non-phenolic oxidation inhibitors which may be used include aromatic amine antioxidants and these may be used either as such or in combination with phenolics. Typical examples of non-phenolic antioxidants include: alkylated and non-alkylated aromatic amines such as aromatic monoamines of the formula R.sup.8R.sup.9R.sup.10N where R.sup.8 is an aliphatic, aromatic or substituted aromatic group, R.sup.9 is an aromatic or a substituted aromatic group, and R.sup.10 is H, alkyl, aryl or R.sup.11S(O).sub.xR.sup.12 where R.sup.11 is an alkylene, alkenylene, or aralkylene group, R.sup.12 is a higher alkyl group, or an alkenyl, aryl, or alkaryl group, and x is 0, 1 or 2. The aliphatic group R.sup.8 may contain from 1 to 20 carbon atoms, and preferably contains from 6 to 12 carbon atoms. The aliphatic group is a saturated aliphatic group. Preferably, both R.sup.8 and R.sup.9 are aromatic or substituted aromatic groups, and the aromatic group may be a fused ring aromatic group such as naphthyl. Aromatic groups R.sup.8 and R.sup.9 may be joined together with other groups such as S.
[0115] Typical aromatic amines antioxidants have alkyl substituent groups of at least 6 carbon atoms. Examples of aliphatic groups include hexyl, heptyl, octyl, nonyl, and decyl. Generally, the aliphatic groups will not contain more than 14 carbon atoms. The general types of amine antioxidants useful in the present compositions include diphenylamines, phenyl naphthylamines, phenothiazines, imidodibenzyls and diphenyl phenylene diamines. Mixtures of two or more aromatic amines are also useful. Polymeric amine antioxidants can also be used. Particular examples of aromatic amine antioxidants useful in the present disclosure include: p,p'-dioctyldiphenylamine; t-octylphenyl-alpha-naphthylamine; phenyl-alphanaphthylamine; and p-octylphenyl-alpha-naphthylamine.
[0116] Sulfurized alkyl phenols and alkali or alkaline earth metal salts thereof also are useful antioxidants.
[0117] Preferred antioxidants include hindered phenols, arylamines. These antioxidants may be used individually by type or in combination with one another. Such additives may be used in an amount of 0.01 to 5 weight percent, preferably 0.01 to 2 weight percent, more preferably zero to 1.5 weight percent, more preferably zero to less than 1 weight percent.
Pour Point Depressants (PPDs)
[0118] Conventional pour point depressants (also known as lube oil flow improvers) may be added to the compositions of the present disclosure if desired. These pour point depressant may be added to lubricating compositions of the present disclosure to lower the minimum temperature at which the fluid will flow or can be poured. Examples of suitable pour point depressants include polymethacrylates, polyacrylates, polyarylamides, condensation products of haloparaffin waxes and aromatic compounds, vinyl carboxylate polymers, and terpolymers of dialkylfumarates, vinyl esters of fatty acids and allyl vinyl ethers. U.S. Pat. Nos. 1,815,022; 2,015,748; 2,191,498; 2,387,501; 2,655,479; 2,666,746; 2,721,877; 2,721,878; and 3,250,715 describe useful pour point depressants and/or the preparation thereof. Such additives may be used in an amount of about 0.01 to 5 weight percent, preferably about 0.01 to 1.5 weight percent.
Seal Compatibility Agents
[0119] Seal compatibility agents help to swell elastomeric seals by causing a chemical reaction in the fluid or physical change in the elastomer. Suitable seal compatibility agents for lubricating oils include organic phosphates, alkoxysulfolanes, aromatic esters, aromatic hydrocarbons, esters (butylbenzyl phthalate, for example), and polybutenyl succinic anhydride. Such additives may be used in an amount of about 0.01 to 3 weight percent, preferably about 0.01 to 2 weight percent.
Antifoam Agents
[0120] Anti-foam agents may advantageously be added to lubricant compositions. These agents retard the formation of stable foams. Silicones and organic polymers are typical anti-foam agents. For example, polysiloxanes, such as silicon oil or polydimethyl siloxane, provide antifoam properties. Anti-foam agents are commercially available and may be used in conventional minor amounts along with other additives such as demulsifiers; usually the amount of these additives combined is less than 1 weight percent and often less than 0.1 weight percent.
Inhibitors and Antirust Additives
[0121] Antirust additives (or corrosion inhibitors) are additives that protect lubricated metal surfaces against chemical attack by water or other contaminants. A wide variety of these are commercially available.
[0122] One type of antirust additive is a polar compound that wets the metal surface preferentially, protecting it with a film of oil. Another type of antirust additive absorbs water by incorporating it in a water-in-oil emulsion so that only the oil touches the metal surface. Yet another type of antirust additive chemically adheres to the metal to produce a non-reactive surface. Examples of suitable additives include zinc dithiophosphates, metal phenolates, basic metal sulfonates, fatty acids and amines. Such additives may be used in an amount of about 0.01 to 5 weight percent, preferably about 0.01 to 1.5 weight percent.
Friction Modifiers
[0123] A friction modifier is any material or materials that can alter the coefficient of friction of a surface lubricated by any lubricant or fluid containing such material(s). Friction modifiers, also known as friction reducers, or lubricity agents or oiliness agents, and other such agents that change the ability of base oils, formulated lubricant compositions, or functional fluids, to modify the coefficient of friction of a lubricated surface may be effectively used in combination with the base oils or lubricant compositions of the present disclosure if desired. Friction modifiers that lower the coefficient of friction are particularly advantageous in combination with the base oils and lube compositions of this disclosure.
[0124] Illustrative friction modifiers may include, for example, organometallic compounds or materials, or mixtures thereof. Illustrative organometallic friction modifiers useful in the lubricating engine oil formulations of this disclosure include, for example, molybdenum amine, molybdenum diamine, an organotungstenate, a molybdenum dithiocarbamate, molybdenum dithiophosphates, molybdenum amine complexes, molybdenum carboxylates, and the like, and mixtures thereof. Similar tungsten based compounds may be preferable.
[0125] Other illustrative friction modifiers useful in the lubricating engine oil formulations of this disclosure include, for example, alkoxylated fatty acid esters, alkanolamides, polyol fatty acid esters, borated glycerol fatty acid esters, fatty alcohol ethers, and mixtures thereof.
[0126] Illustrative alkoxylated fatty acid esters include, for example, polyoxyethylene stearate, fatty acid polyglycol ester, and the like. These can include polyoxypropylene stearate, polyoxybutylene stearate, polyoxyethylene isosterate, polyoxypropylene isostearate, polyoxyethylene palmitate, and the like.
[0127] Illustrative alkanolamides include, for example, lauric acid diethylalkanolamide, palmic acid diethylalkanolamide, and the like. These can include oleic acid diethyalkanolamide, stearic acid diethylalkanolamide, oleic acid diethylalkanolamide, polyethoxylated hydrocarbylamides, polypropoxylated hydrocarbylamides, and the like.
[0128] Illustrative polyol fatty acid esters include, for example, glycerol mono-oleate, saturated mono-, di-, and tri-glyceride esters, glycerol mono-stearate, and the like. These can include polyol esters, hydroxyl-containing polyol esters, and the like.
[0129] Illustrative borated glycerol fatty acid esters include, for example, borated glycerol mono-oleate, borated saturated mono-, di-, and tri-glyceride esters, borated glycerol mono-sterate, and the like. In addition to glycerol polyols, these can include trimethylolpropane, pentaerythritol, sorbitan, and the like. These esters can be polyol monocarboxylate esters, polyol dicarboxylate esters, and on occasion polyoltricarboxylate esters. Preferred can be the glycerol mono-oleates, glycerol dioleates, glycerol trioleates, glycerol monostearates, glycerol distearates, and glycerol tristearates and the corresponding glycerol monopalmitates, glycerol dipalmitates, and glycerol tripalmitates, and the respective isostearates, linoleates, and the like. On occasion the glycerol esters can be preferred as well as mixtures containing any of these. Ethoxylated, propoxylated, butoxylated fatty acid esters of polyols, especially using glycerol as underlying polyol can be preferred.
[0130] Illustrative fatty alcohol ethers include, for example, stearyl ether, myristyl ether, and the like. Alcohols, including those that have carbon numbers from C.sub.3 to C.sub.50, can be ethoxylated, propoxylated, or butoxylated to form the corresponding fatty alkyl ethers. The underlying alcohol portion can preferably be stearyl, myristyl, C.sub.11-C.sub.13 hydrocarbon, oleyl, isosteryl, and the like.
[0131] Useful concentrations of friction modifiers may range from 0.01 weight percent to 5 weight percent, or about 0.1 weight percent to about 2.5 weight percent, or about 0.1 weight percent to about 1.5 weight percent, or about 0.1 weight percent to about 1 weight percent. Concentrations of molybdenum-containing materials are often described in terms of Mo metal concentration. Advantageous concentrations of Mo may range from 25 ppm to 700 ppm or more, and often with a preferred range of 50-200 ppm. Friction modifiers of all types may be used alone or in mixtures with the materials of this disclosure. Often mixtures of two or more friction modifiers, or mixtures of friction modifier(s) with alternate surface active material(s), are also desirable.
[0132] When lubricating oil compositions contain one or more of the additives discussed above, the additive(s) are blended into the composition in an amount sufficient for it to perform its intended function. Typical amounts of such additives useful in the present disclosure are shown in Table 1 below.
[0133] It is noted that many of the additives are shipped from the additive manufacturer as a concentrate, containing one or more additives together, with a certain amount of base oil diluents. Accordingly, the weight amounts in the table below, as well as other amounts mentioned herein, are directed to the amount of active ingredient (that is the non-diluent portion of the ingredient). The weight percent (wt %) indicated below is based on the total weight of the lubricating oil composition.
TABLE-US-00002 TABLE 1 Typical Amounts of Other Lubricating Oil Components Approximate Approximate Compound wt % (Useful) wt % (Preferred) Dispersant 0.1-20 0.1-8 Detergent 0.1-20 0.1-8 Friction Modifier 0.01-5 0.01-1.5 Antioxidant 0.1-5 0.1-1.5 Pour Point Depressant (PPD) 0.0-5 0.01-1.5 Anti-foam Agent 0.001-3 0.001-0.15 Viscosity Index Improver 0.1-2 0.1-1 (solid polymer basis) Anti-wear 0.1-2 0.5-1 Inhibitor and Antirust 0.01-5 0.01-1.5
[0134] The foregoing additives are all commercially available materials. These additives may be added independently but are usually precombined in packages which can be obtained from suppliers of lubricant oil additives. Additive packages with a variety of ingredients, proportions and characteristics are available and selection of the appropriate package will take the requisite use of the ultimate composition into account.
[0135] Different engine hardware and control schemes can significantly influence the occurrence of LSPI. See, for example, U.S. Patent Application Publication Nos. 2012/1866225 and 2003/0908070, and also SAE 2012-02-1148, which are all incorporated herein by reference. For example, a 2.0 L, 4-cylinder TGDI GM Ecotec engine can be used for LSPI testing. A six segment test procedure can be used to determine the number of LSPI events that occur at two different specified engine load and speed conditions. Each segment of the test procedure comprises 25,000 engine cycles, where one cycle corresponds to 720 degrees of crank shaft rotation. The first set of conditions is 2000 RPM and 18 bar BMEP, hereafter referred to as "High Load". The second set of conditions is 1500 RPM and 12.5 bar BMEP, hereafter referred to as "Low Load". The test procedure comprises two segments of High Load, followed by two segments of Low Load, followed by two segments of High Load. A 30 minute warm up at 2000 RPM and 4 bar BMEP is also conducted prior to commencing the test procedure. This test procedure is repeated four times for each of the lubricants tested. LSPI events are counted during the High Load segments only, using pressure transducers placed in each of the 4 cylinders to monitor the peak cylinder pressure. Peak pressures in the cylinder that are greater than 4.7 standard deviations above the mean peak cylinder pressure, or more than 4.7 standard deviations below the mean peak cylinder pressure are counted as an LSPI event.
[0136] In accordance with this disclosure, the engine can exhibit greater than about 10%, or greater than about 25%, reduced low speed pre-ignition, based on normalized low speed pre-ignition (LSPI) counts per 25,000 engine cycles, engine operation at 2000 revolutions per minute (RPM) and brake mean effective pressure (BMEP) at 18 bar, as compared to low speed pre-ignition performance achieved in an engine using a lubricating oil containing calcium or magnesium.
[0137] The following non-limiting examples are provided to illustrate the disclosure.
Examples
[0138] Formulations were prepared as simple blends with 10% carbon black (Cabot Vulcan XC72R), a metal oxide, and base stock (balance) as shown in FIGS. 1-3. All of the ingredients used herein are commercially available.
[0139] Testing was conducted for the formulations as described in Ko Onodera et al., Engine Oil Formulation Technology to Prevent Pre-ignition in Turbocharged Direct Injection Spark Ignition Engines, JSAE 20159071, SAE 2015-01-2027, incorporated herein by reference. The testing compared LSPI engine test results to thermogravimetric analysis (TGA) decomposition temperatures. The carbon black decomposition temperature was defined as the temperature at which 50% mass decrease between 400.degree. C. and 800.degree. C. was observed. The carbon black decomposition temperature is an indicator of the catalytic activity of the metal oxide for promoting auto-ignition. An increase in the decomposition temperature of a lubricant corresponds to an increase of the auto-ignition temperature of the LSPI precursors resulting in less LSPI activity. TGA was run according to the conditions listed in Table 2 below.
TABLE-US-00003 TABLE 2 Temperature 25 .degree.C.-800 .degree.C. Temperature increase rate 10 .degree.C./min Atmosphere Air Sample pan Pt
[0140] For purposes of this disclosure, the above testing is referred to as the Thermogravimetric Engine Oil Simulation Test.
[0141] For comparative purposes, results were generated that match those disclosed in Onodera et al.
[0142] In addition, various other metal oxides were tested. The results show that metal oxides can alter the carbon black decomposition temperatures by up to 250.degree. C. (see FIG. 1). FIG. 1 shows carbon decomposition temperatures of various metal oxides in a carbon black/PAO mixture. Most notable among the oxide results are sodium, manganese, titanium, and aluminum. These four metals show low catalytic activity, even below that of magnesium, which in the form of a detergent, is well known to have significantly lower LSPI activity than calcium detergents.
[0143] Simplified lubricating oil blends were prepared as shown in FIGS. 2 and 3. The blends contained only base stock, carbon black, and metal oxide. All of the ingredients used herein are commercially available. Group III, IV, and V base stocks were used in the formulations.
[0144] FIG. 2 shows carbon decomposition temperatures of various metal oxides in a carbon black/base stock mixture in which barium oxide and titanium(IV) oxide and various base stocks were used. Barium oxide was selected because of its poor performance while titanium(IV) oxide was selected for its strong performance. Base stock selection impacted the decomposition temperature with Visom (Group III) performing the worst, EHC (Group II) in the middle, and GTL and PAO (Group III+ and Group IV) having the highest decomposition temperatures. Titanium has higher decomposition temperatures than barium across the spectrum and also has less variability between the different base stocks.
[0145] FIG. 3 shows carbon decomposition temperatures of various metal oxides in a carbon black/formulated mixture in which barium and titanium and PAO base stock were used with varying concentrations of metal oxide. Titanium has higher decomposition temperatures than barium, and both sets of samples remain fairly constant regardless of concentration.
[0146] The forgoing examples have shown that various metal oxides demonstrate low catalytic activity which leads to improved LSPI performance in an engine.
PCT and EP Clauses:
[0147] 1. A method for preventing or reducing low speed pre-ignition in an engine lubricated with a lubricating oil by using as the lubricating oil a formulated oil, said formulated oil having a composition comprising a lubricating oil base stock as a major component; and at least one lubricating oil additive, as a minor component; wherein said lubricating oil contains at least one metal or metalloid in an amount sufficient that, in thermogravimetric measurements of the lubricating oil by a Thermogravimetric Engine Oil Simulation Test, carbon black decomposition temperature is increased as compared to the carbon black decomposition temperature in a lubricating oil containing calcium or magnesium.
[0148] 2. The method of clause 1 wherein, in thermogravimetric measurements of the lubricating oil by a Thermogravimetric Engine Oil Simulation Test, the carbon black decomposition temperature is increased by at least 10.degree. C. as compared to the carbon black decomposition temperature in a lubricating oil containing calcium or magnesium.
[0149] 3. The method of clause 1 wherein, in thermogravimetric measurements of the lubricating oil by a Thermogravimetric Engine Oil Simulation Test, the carbon black decomposition temperature is increased by at least 25.degree. C. as compared to the carbon black decomposition temperature in a lubricating oil containing calcium or magnesium.
[0150] 4. The method of clause 1 wherein the engine exhibits greater than 10% reduced low speed pre-ignition, based on normalized low speed pre-ignition (LSPI) counts per 25,000 engine cycles, engine operation at 2000 revolutions per minute (RPM) and brake mean effective pressure (BMEP) at 18 bar, as compared to low speed pre-ignition performance achieved in an engine using a lubricating oil containing calcium or magnesium.
[0151] 5. The method of clauses 1-4 wherein the at least one metal or metalloid comprises sodium, manganese, titanium, aluminum, or mixtures thereof.
[0152] 6. The method of clauses 1-5 wherein the at least one metal or metalloid is present in the lubricating oil in an amount from 100 ppm to 25,000 ppm.
[0153] 7. The method of clauses 1-6 wherein (i) the at least one metal or metalloid is present in the lubricating oil in an amount from 100 ppm to 1000 ppm; (ii) total base number (TBN), as measured by ASTM D2896, of the lubricating oil ranges from 2 mg KOH to 17 mg KOH; (iii) total sulfated ash of the lubricating oil ranges from 0.01 to 1 wt %.
[0154] 8. A lubricating engine oil having a composition comprising a lubricating oil base stock as a major component; and at least one lubricating oil additive, as a minor component; wherein said lubricating engine oil contains at least one metal or metalloid in an amount sufficient that, in thermogravimetric measurements of the lubricating engine oil by a Thermogravimetric Engine Oil Simulation Test, carbon black decomposition temperature is increased as compared to the carbon black decomposition temperature in a lubricating engine oil containing calcium or magnesium.
[0155] 9. The lubricating engine oil of clause 8 wherein, in thermogravimetric measurements of the lubricating engine oil by a Thermogravimetric Engine Oil Simulation Test, the carbon black decomposition temperature is increased by at least 10.degree. C. as compared to the carbon black decomposition temperature in a lubricating engine oil containing calcium or magnesium.
[0156] 10. The lubricating engine oil of clause 8 wherein, in thermogravimetric measurements of the lubricating engine oil by a Thermogravimetric Engine Oil Simulation Test, the carbon black decomposition temperature is increased by at least 25.degree. C. as compared to the carbon black decomposition temperature in a lubricating engine oil containing calcium or magnesium.
[0157] 11. The lubricating engine oil of clause 8 wherein the engine exhibits greater than 10% reduced low speed pre-ignition, based on normalized low speed pre-ignition (LSPI) counts per 25,000 engine cycles, engine operation at 2000 revolutions per minute (RPM) and brake mean effective pressure (BMEP) at 18 bar, as compared to low speed pre-ignition performance achieved in an engine using a lubricating engine oil containing calcium or magnesium.
[0158] 12. The lubricating engine oil of clauses 8-11 wherein the at least one metal or metalloid comprises sodium, manganese, titanium, aluminum, or mixtures thereof.
[0159] 13. The lubricating engine oil of clauses 8-12 wherein the at least one metal or metalloid is present in the lubricating engine oil in an amount from 100 ppm to 25,000 ppm.
[0160] 14. The lubricating engine oil of clauses 8-13 wherein (i) the at least one metal or metalloid is present in the lubricating engine oil in an amount from 100 ppm to 1000 ppm; (ii) total base number (TBN), as measured by ASTM D2896, of the lubricating engine oil ranges from 2 mg KOH to 17 mg KOH; (iii) total sulfated ash of the lubricating engine oil ranges from 0.01 to 0.17 wt %.
[0161] 15. The lubricating engine oil of clauses 8-14 wherein the at least one lubricating oil additive has a total sulfated ash from 0.02 to 0.17 wt %.
[0162] All patents and patent applications, test procedures (such as ASTM methods, UL methods, and the like), and other documents cited herein are fully incorporated by reference to the extent such disclosure is not inconsistent with this disclosure and for all jurisdictions in which such incorporation is permitted.
[0163] When numerical lower limits and numerical upper limits are listed herein, ranges from any lower limit to any upper limit are contemplated. While the illustrative embodiments of the disclosure have been described with particularity, it will be understood that various other modifications will be apparent to and can be readily made by those skilled in the art without departing from the spirit and scope of the disclosure. Accordingly, it is not intended that the scope of the claims appended hereto be limited to the examples and descriptions set forth herein but rather that the claims be construed as encompassing all the features of patentable novelty which reside in the present disclosure, including all features which would be treated as equivalents thereof by those skilled in the art to which the disclosure pertains.
[0164] The present disclosure has been described above with reference to numerous embodiments and specific examples. Many variations will suggest themselves to those skilled in this art in light of the above detailed description. All such obvious variations are within the full intended scope of the appended claims
* * * * *
References

D00000

D00001

D00002

D00003

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.