Polyisocyanurate Plastic Containing Siloxane Groups And Method For Production Thereof
KLIMMASCH; Thomas ; et al.
U.S. patent application number 16/095193 was filed with the patent office on 2019-05-16 for polyisocyanurate plastic containing siloxane groups and method for production thereof. The applicant listed for this patent is COVESTRO DEUTSCHLAND AG. Invention is credited to Thomas KLIMMASCH, Jurgen KOCHER, Hans-Josef LAAS, Jan WEIKARD.
| Application Number | 20190144598 16/095193 |
| Document ID | / |
| Family ID | 55752224 |
| Filed Date | 2019-05-16 |




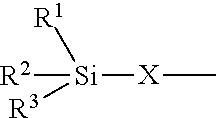





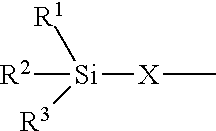
View All Diagrams
| United States Patent Application | 20190144598 |
| Kind Code | A1 |
| KLIMMASCH; Thomas ; et al. | May 16, 2019 |
POLYISOCYANURATE PLASTIC CONTAINING SILOXANE GROUPS AND METHOD FOR PRODUCTION THEREOF
Abstract
The invention relates to novel polyisocyanurate plastics containing siloxane groups, which are obtainable by a method comprising the following steps: al) providing a composition A), which contains i) oligomeric polyisocyanates B) and compounds containing silicon C) or ii) contains oligomeric silicon-modified polyisocyanates D); or iii) contains oligomeric silicon-modified polyisocyanates D) and oligomeric polyisocyanates B); or iv) contains oligomeric polyisocyanates B), compounds containing silicon C) and modified oligomeric polyisocyanates D); or v) contains compounds containing silicon C) and silicon-modified, oligomeric polyisocyanates D); a2) catalytic trimerization of the composition A); wherein the composition A) has a content of monomer diisocyanates of 20 wt % at maximum. The invention further relates to the method by which the polyisocyanurate plastics according to the invention are obtainable, to the use of the polyisocyanurate plastics according to the invention for producing coatings, films, semi-finished products, and molded parts, and to substrates coated by such a coating.
| Inventors: | KLIMMASCH; Thomas; (Leverkusen, DE) ; WEIKARD; Jan; (Leverkusen, DE) ; LAAS; Hans-Josef; (Odenthal, DE) ; KOCHER; Jurgen; (Langenfeld, DE) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 55752224 | ||||||||||
| Appl. No.: | 16/095193 | ||||||||||
| Filed: | October 25, 2016 | ||||||||||
| PCT Filed: | October 25, 2016 | ||||||||||
| PCT NO: | PCT/EP2016/075689 | ||||||||||
| 371 Date: | October 19, 2018 |
| Current U.S. Class: | 528/28 |
| Current CPC Class: | C08G 18/168 20130101; C08G 18/792 20130101; C09D 175/08 20130101; C08G 18/809 20130101; C08J 5/18 20130101; C08G 18/73 20130101; C08G 18/1816 20130101; C08G 18/225 20130101; C08G 18/283 20130101; C08G 18/289 20130101; C08G 18/8064 20130101; C08G 18/022 20130101; C08J 2375/08 20130101; C08G 18/4833 20130101 |
| International Class: | C08G 18/80 20060101 C08G018/80; C08G 18/22 20060101 C08G018/22; C08G 18/16 20060101 C08G018/16; C08G 18/73 20060101 C08G018/73 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Apr 21, 2016 | EP | 16166427.1 |
Claims
1.-20. (canceled)
21. A polyisocyanurate plastic containing siloxane groups, obtainable by a process comprising the steps of: a1) providing a composition A) which i) contains oligomeric polyisocyanates B) and silicon-containing compounds C); or ii) contains oligomeric silicon-modified polyisocyanates D); or iii) contains oligomeric silicon-modified polyisocyanates D) and oligomeric polyisocyanates B); or iv) contains oligomeric polyisocyanates B), silicon-containing compounds C) and modified oligomeric polyisocyanates D); or v) contains silicon-containing compounds C) and silicon-modified, oligomeric polyisocyanates D); a2) catalytic trimerization of the composition A); wherein the composition A) has a content of monomeric diisocyanates of not more than 20% by weight.
22. The polyisocyanurate plastic containing siloxane groups as claimed in claim 21, characterized in that the silicon-containing compounds C) has at least one isocyanate-reactive functional group.
23. The polyisocyanurate plastic containing siloxane groups as claimed in claim 21, characterized in that the silicon-containing compounds C) are selected from the group consisting of oligomeric silicon-modified polyisocyanates D), aminosilanes E), silane-functional aspartic esters F), silane-functional alkylamides G), mercaptosilanes H), isocyanatosilanes I) and mixtures thereof.
24. The polyisocyanurate plastic containing siloxane groups as claimed in claim 21, characterized in that the silicon-containing compounds C) and the oligomeric silicon-modified polyisocyanates D) comprise at least one structural unit selected from the group consisting of: a) a structural unit of formula (I) ##STR00017## where R.sup.1, R.sup.2 and R.sup.3 are identical or different radicals and are each a saturated or unsaturated, linear or branched, aliphatic or cycloaliphatic or an optionally substituted aromatic or araliphatic radical having 1 to 18 carbon atoms, which may optionally contain 1 to 3 heteroatoms from the series of oxygen, sulfur and nitrogen, X is a linear or branched organic radical which has at least 1 carbon atom and may optionally contain 1 to 2 imino groups (--NH--), and R.sup.4 is hydrogen, a saturated or unsaturated, linear or branched, aliphatic or cycloaliphatic or an optionally substituted aromatic or araliphatic radical having 1 to 18 carbon atoms or a radical of the formula ##STR00018## in which R.sup.1, R.sup.2, R.sup.3 and X are as defined above; b) a structural unit of formula (II) ##STR00019## wherein R.sup.1, R.sup.2 and R.sup.3 are as defined for formula (I), X is a linear or branched organic radical having at least 1 carbon atom and R.sup.5 and R.sup.6 are independently saturated or unsaturated, linear or branched, aliphatic or cycloaliphatic or aromatic organic radicals which have 1 to 18 carbon atoms, are substituted or unsubstituted and/or have heteroatoms in the chain; c) a structural unit of formula (III) ##STR00020## wherein R.sup.1, R.sup.2 and R.sup.3 are as defined for formula (I), X is a linear or branched organic radical having at least 1 carbon atom and R.sup.9 is a saturated linear or branched, aliphatic or cycloaliphatic organic radical having 1 to 8 carbon atoms; d) a structural unit of formula (IV) ##STR00021## wherein R.sup.1, R.sup.2 and R.sup.3 are as defined for formula (I) and Y is a linear or branched organic radical having at least 1 carbon atom; and e) a structural unit of formula (V) ##STR00022## wherein R.sup.1, R.sup.2 and R.sup.3 are as defined for formula (I), and Y is a linear or branched organic radical having at least 1 carbon atom.
25. The polyisocyanurate plastic containing siloxane groups as claimed in claim 21, characterized in that said plastic comprises alkoxysiloxane groups, the silicon-containing compounds C) are alkoxysilyl-containing compounds C) and the oligomeric silicon-modified polyisocyanates D) are oligomeric polyisocyanates D) modified with alkoxysilyl groups.
26. The polyisocyanurate plastic containing siloxane groups as claimed in claim 21, characterized in that the silicon-containing compounds C) and the oligomeric silicon-modified polyisocyanates D) comprise at least one structural unit selected from the group consisting of: a) a structural unit of formula (I) ##STR00023## where R.sup.1, R.sup.2 and R.sup.3 are each methyl, methoxy and/or ethoxy, with the proviso that at least one of the radicals R.sup.1, R.sup.2 and R.sup.3 is such a methoxy or ethoxy radical, X is a propylene radical (--CH.sub.2--CH.sub.2--CH.sub.2--), and R.sup.4 is hydrogen, a methyl radical or a radical of formula ##STR00024## in which R.sup.1, R.sup.2, R.sup.3 and X are as defined above; b) a structural unit of formula (II) ##STR00025## where R.sup.1, R.sup.2, R.sup.3 and X wherein R.sup.1, R.sup.2 and R.sup.3 are as defined for formula (I), R.sup.5 and R.sup.6 are identical or different radicals and are a methyl-, ethyl-, n-butyl- or 2-ethylhexyl radical; c) a structural unit of formula (III) ##STR00026## where R.sup.1, R.sup.2, R.sup.3 and X wherein R.sup.1, R.sup.2 and R.sup.3 are as defined for formula (I), R.sup.9 is hydrogen; d) structural unit of formula (IV) ##STR00027## where R.sup.1, R.sup.2 and R.sup.3 wherein R.sup.1, R.sup.2 and R.sup.3 are as defined for formula (I), Y is a propylene radical (--CH.sub.2--CH.sub.2--CH.sub.2--); and e) a structural unit of formula (V) ##STR00028## wherein R.sup.1, R.sup.2 and R.sup.3 are as defined for formula (I), and Y is a linear or branched organic radical having at least 1 carbon atom.
27. The polyisocyanurate plastic containing siloxane groups as claimed in claim 21, characterized in that the oligomeric polyisocyanates B) or the oligomeric, silicon-modified polyisocyanates D) are selected from at least one oligomeric polyisocyanate having uretdione, isocyanurate, allophanate, biuret, iminooxadiazinedione or oxadiazinetrione structure or mixtures thereof.
28. The polyisocyanurate plastic containing siloxane groups as claimed in claim 21, characterized in that the oligomeric polyisocyanates B) or the oligomeric, silicon-modified polyisocyanates D) consist of one or more oligomeric polyisocyanates formed from 1,5-diisocyanatopentane, 1,6-diisocyanatohexane, isophorone diisocyanate or 4,4'-diisocyanatodicyclohexylmethane or mixtures thereof.
29. The polyisocyanurate plastic containing siloxane groups as claimed in claim 21, characterized in that the catalytic trimerization is carried out in the presence of a trimerization catalyst L), wherein the trimerization catalyst L) preferably comprises at least one alkali metal or alkaline earth metal salt.
30. The polyisocyanurate plastic containing siloxane groups as claimed in claim 29, characterized in that the trimerization catalyst L) comprises potassium acetate as the alkali metal salt.
31. The polyisocyanurate plastic containing siloxane groups as claimed in claim 29, characterized in that the trimerization catalyst L) comprises a polyether, in particular a polyethylene glycol.
32. The polyisocyanurate plastic containing siloxane groups as claimed in claim 21, characterized in that the composition A) has a content of monomeric diisocyanates of not more than 15% by weight, not more than 10% by weight or not more than 5% by weight, based in each case on the weight of the composition A).
33. The polyisocyanurate plastic containing siloxane groups as claimed in claim 21, characterized in that it constitutes a highly converted polyisocyanurate plastic containing siloxane groups in which not more than 20% of the isocyanate groups originally contained in the composition A) have been preserved.
34. The use of a polyisocyanurate plastic containing siloxane groups as claimed in claim 21 for producing coatings, films, semifinished products and moldings.
35. coating, film, semifinished product or molding containing a polyisocyanurate plastic containing siloxane groups as claimed in claim 21.
36. A substrate coated with a coating as claimed in claim 35, wherein the substrate may in particular be selected from a vehicle body, in particular a goods vehicle or motor vehicle body, and electronic entertainment devices, such as laptops, tablets or mobile phones.
37. A process for producing a polyisocyanurate plastic containing siloxane groups comprising the steps of: a1) providing a composition A) which i) contains oligomeric polyisocyanates B) and silicon-containing compounds C); or ii) contains oligomeric silicon-modified polyisocyanates D); or iii) contains oligomeric silicon-modified polyisocyanates D) and oligomeric polyisocyanates B); or iv) contains oligomeric polyisocyanates B), silicon-containing compounds C) and modified oligomeric polyisocyanates D); or v) contains silicon-containing compounds C) and silicon-modified, oligomeric polyisocyanates D); a2) catalytic trimerization of the composition A); wherein the composition A) has a content of monomeric diisocyanates of not more than 20% by weight.
38. The process as claimed in claim 37, characterized in that the catalytic trimerization is carried out in the presence of a trimerization catalyst L) which is defined as in claim 29.
39. The process as claimed in claim 37, characterized in that the silicon-containing compounds C) and/or the oligomeric, silicon-modified polyisocyanates D) are defined as in claim 22 and/or in that the composition A) is defined as in claim 32.
40. The process as claimed in claim 37, characterized in that the catalytic trimerization is continued at least up to a degree of conversion at which only not more than 20% of the isocyanate groups originally contained in the composition A) remain present, so that a highly converted polyisocyanurate plastic containing siloxane groups is obtained.
Description
[0001] The invention relates to a siloxane-containing polyisocyanurate plastic, to a process for production thereof, to the use thereof for producing coatings, films, semifinished products or moldings and also to substrates coated with such a coating.
[0002] Siloxane-containing polyurethane plastics produced from alkoxysilyl-containing polyisocyanates and polyols have long been known. They are used in particular for producing chemicals-resistant and scratch-resistant coatings for OEM production line finishing and automotive refinishing. The curing of such coatings is effected by the urethanization reaction of the isocyanate groups of the alkoxysilyl-containing polyisocyanate with the hydroxyl groups of the polyol and also by hydrolysis and condensation of the alkoxysilyl groups to afford siloxane groups.
[0003] EP 1 273 640 A1 describes a solvent-containing thermally curable two-component polyurethane automotive clearcoat/topcoat having improved scratch resistance which is obtainable by using silicon-modified polyisocyanates as isocyanate crosslinker components. The silicon-modified polyisocyanates are obtained by reaction of aliphatic and/or cycloaliphatic polyisocyanates with N,N-bis(trialkoxysilylpropyl)amines. Alternatively, silicon-modified polyisocyanates may also be produced by partial reaction of monomeric diisocyanates, such as HDI or IPDI, with N,N-bis(trialkoxysilylpropyl)amines and subsequent oligomerization. In this latter method of production of the silicon-modified polyisocyanates the subsequent oligomerization specifically does not form highly converted, crosslinked siloxane-containing polyisocyanurate plastics but rather only oligomeric, low-viscosity, soluble products.
[0004] A series of further publications, for example WO 2008/074489 A1, WO 2008/074490 A1, WO 2010/149236 A1, WO 2014/086-530 A1 and WO 2009/156148 A1, likewise describe coating compositions for producing weather-resistant, scratch-resistant polyurethane clearcoat layers for OEM production line finishing and automotive refinishing in which as the isocyanate crosslinker component silicon-modified polyisocyanates are used.
[0005] The disadvantage in all of these cases is that not only isocyanate groups and hydroxyl groups but also alkoxysilyl groups and hydroxyl groups must be stored separately from one another in different components and typically mixed with one another only immediately before application since the recited groups can react with one another even under customary storage conditions.
[0006] The curing of highly scratch-resistant coatings by catalytic condensation of alkoxysilyl-containing compounds is described for example in S. Hofacker et al., Progress in Organic Coatings 45 (2002), 159-164.
[0007] Since the curing mechanism of the alkoxysilyl groups proceeds via a condensation reaction with the elimination of the corresponding alcohol a considerable volume shrinkage occurs.
[0008] Strains, tears and loss of adhesion can therefore result. Furthermore, in coatings technology, for example, coatings which undergo severe shrinking are known to telegraph roughness and surface structures of the substrate, which can lead to a loss of shine and other disruptions to the appearance of the coating.
[0009] The siloxane-containing polyurethane plastics known from the prior art thus have the essential disadvantage that to achieve high network densities either a high volume shrinkage has to be suffered due to excessive elimination of constituents during curing or when using polyaddition reactions (for example by urethane formation) the achievement of high network densities becomes difficult. However, a minimum requirement that results is that of handling two components during storage.
[0010] Polymers having polyisocyanurate structure are known for their high mechanical strength. The present processes known from the prior art for producing such highly converted polyisocyanurate plastics often proceed from liquid monomeric diisocyanates. On account of the high exothermicity of the trimerization reaction to afford polyisocyanurates (-75 kJ/mol NCO), a reaction proceeding from monomeric diisocyanates, particularly in the case of monomeric diisocyanates having a high isocyanate content (e.g. BDI, PDI, HDI, TIN), is typically not possible on a large scale but only in small amounts of substance under strict temperature control.
[0011] WO 2015/166983 describes the use of polyisocyanurates plastics for encapsulation of LEDs. The use of silicon-modified polyisocyanates is not disclosed.
[0012] While U.S. Pat. No. 6,133,397 describes the use of oligomeric polyisocyanates for forming coatings it does not disclose silicon-modified coatings.
[0013] Temperature control during the production of highly converted polyisocyanurates is of enormous importance since, due to the high isocyanate contents of the monomeric starting materials and the exothermic reaction, temperatures of more than 300.degree. C. can arise, which can result in direct decomposition of the products and even in in situ evaporation of the monomeric diisocyanates. In addition to the occupational hygiene disadvantages due to the liberated toxic monomeric diisocyanates or decomposition products, the formation of blisters and discolorations in the finished polyisocyanurate plastic is very disruptive here.
[0014] Consequently, polyisocyanurates have hitherto usually only found practical applications as crosslinking agents in paint chemistry, the production of which involves stopping the trimerization reaction at low conversions and removing excess unreacted monomeric diisocyanate. Thus, DE 31 00 263; GB 952 931, GB 966 338; U.S. Pat. Nos. 3,211,703, 3,330,828 envisage conducting the reaction either in dilution or only up to low conversion values with very precise temperature control during the production of crosslinking agents based on isocyanurates starting from aliphatic and mixed aliphatic and aromatic monomeric diisocyanates. Here too highly converted polyisocyanurate plastics are specifically not formed, but rather only oligomeric, low-viscosity, soluble products.
[0015] For a number of applications, for example for automotive refinishing and in particular OEM production line finishing and also for coating of electronic entertainment devices, in particular laptops, tablets and mobile phones, there is a constant demand in the market for coating compositions having further-improved scratch resistance.
[0016] The problem addressed by the present invention was accordingly that of providing novel siloxane-containing polyisocyanurate plastics having improved properties, in particular improved scratch resistance and chemicals resistance and wear resistance, which are suitable in particular for automotive finishing and for the coating of electronic entertainment devices and may be handled in one-component form.
[0017] This problem is solved in accordance with the invention by the polyisocyanurate plastic specified in claim 1, by the uses and articles of manufacture specified in claims 14 to 16 and by the process specified in claim 17.
[0018] Advantageous configurations of the invention are specified in the dependent claims and are elucidated in detail hereinafter, as is the general concept of the invention.
[0019] The invention provides a polyisocyanurate plastic containing siloxane groups, obtainable by a process comprising the steps of: [0020] a1) providing a composition A) which [0021] i) contains oligomeric polyisocyanates B) and silicon-containing compounds C); or [0022] ii) contains oligomeric silicon-modified polyisocyanates D); or [0023] iii) contains oligomeric silicon-modified polyisocyanates D) and oligomeric polyisocyanates B); or [0024] iv) contains oligomeric polyisocyanates B), silicon-containing compounds C) and modified oligomeric polyisocyanates D); or [0025] v) contains silicon-containing compounds C) and silicon-modified, oligomeric polyisocyanates D); [0026] a2) catalytic trimerization of the composition A); wherein the composition A) has a content of monomeric diisocyanates of not more than 20% by weight.
[0027] The invention also further provides the abovementioned process from which the polyisocyanurate plastics of the invention are obtainable. Also forming part of the subject matter of the invention, moreover, is the use of the polyisocyanurate plastics according to the invention for producing coatings, films, semifinished products and moldings and also substrates coated with the polyisocyanurate plastic according to the invention.
[0028] The invention more particularly elucidated hereinbelow is based on the surprising observation that a composition containing oligomeric polyisocyanates and silicon-containing compounds and/or oligomeric silicon-modified polyisocyanates may be cured by catalytic trimerization in such a way that simultaneously with or before hydrolysis/condensation of silicon-containing functional groups of the silicon-containing compounds and/or of the silicon-modified polyisocyanates to afford siloxane groups, isocyanate groups of the oligomeric polyisocyanates and/or of the silicon-modified polyisocyanates react to afford polyisocyanurates by trimerization, thus forming highly crosslinked, highly converted polyisocyanurate plastics having novel properties. This dual curing mechanism results in a novel crosslinking structure in the obtainable polyisocyanurate plastic, said plastic therefore differing structurally from the materials known from the prior art.
[0029] The use of compositions containing oligomeric polyisocyanates and silicon-containing compounds and/or oligomeric, silicon-modified polyisocyanates instead of monomeric diisocyanates as starting materials for producing polyisocyanurate plastics moreover has the advantage that on account of the low isocyanate contents of the oligomeric reactants a markedly lower heat of reaction requires removal during curing which in particular also facilitates the production of large-volume components.
[0030] A "polyisocyanurate plastic" as used herein is a plastic containing polyisocyanurate. It may also consist predominantly of a polyisocyanurate. Blends composed of polyisocyanurates and other plastics are likewise covered by the term "polyisocyanurate plastic" as used here.
[0031] When reference is made here to "plastic", this means a product which is very substantially dimensionally stable at room temperature--in contrast, for example, to gels or liquids. The term "plastic", as used here, comprises all customary classes of plastic, i.e. in particular including thermosets, thermoplastics and elastomers.
[0032] A "polyisocyanurate" as used here is any molecule, preferably a polymer, having a plurality of isocyanurate structural units, for example at least 10 isocyanurate structural units. A molecule having a single isocyanurate structural unit can be referred to as "isocyanurate".
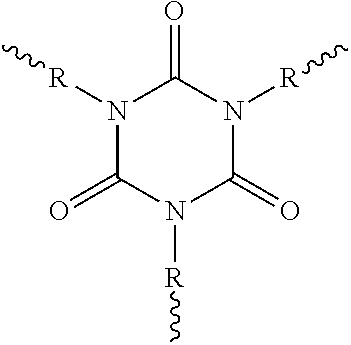
[0033] The characteristic cyclic isocyanurate structural unit is shown in the following structural formula:
##STR00001##
[0034] Isocyanurates and polyisocyanurates can be obtained by cyclotrimerization of polyisocyanates. The conventionally operated cyclotrimerization proceeding from monomeric diisocyanates is--as described above--a strongly exothermic reaction. This can considerably restrict the use options and the levels of trimerization that are still achievable industrially and efficiently.
[0035] The term "polyisocyanate" as used here is a collective term for compounds containing two or more isocyanate groups (this is understood by the person skilled in the art to mean free isocyanate groups of the general structure --N.dbd.C.dbd.O) in the molecule. The simplest and most important representatives of these polyisocyanates are the diisocyanates. These have the general structure O.dbd.C.dbd.N--R--N.dbd.C.dbd.O where R typically represents aliphatic, alicyclic and/or aromatic radicals.
[0036] Because of the polyfunctionality (.gtoreq.2 isocyanate groups), it is possible to use polyisocyanates to produce a multitude of polymers (e.g. polyurethanes, polyureas and polyisocyanurates) and low molecular weight compounds (for example those having uretdione, isocyanurate, allophanate, biuret, iminooxadiazinedione and/or oxadiazinetrione structure).
[0037] Where reference is made here to "polyisocyanates" in general terms, this means monomeric and/or oligomeric polyisocyanates alike. For the understanding of many aspects of the invention, however, it is important to distinguish between monomeric diisocyanates and oligomeric polyisocyanates. Where reference is made here to "oligomeric polyisocyanates", this means polyisocyanates formed from at least two monomeric diisocyanate molecules, i.e. compounds that constitute or contain a reaction product formed from at least two monomeric diisocyanate molecules.
[0038] The production of oligomeric polyisocyanates from monomeric diisocyanates is also referred to here as oligomerization of monomeric diisocyanates. This "oligomerization" as used here means the reaction of monomeric diisocyanates to give oligomeric polyisocyanates having uretdione, isocyanurate, allophanate, thioallophanate, biuret, iminooxadiazinedione and/or oxadiazinetrione structure.
[0039] For example, hexamethylene diisocyanate (HDI) is a "monomeric diisocyanate" since it contains two isocyanate groups and is not a reaction product formed from at least two polyisocyanate molecules:
##STR00002##
[0040] Reaction products which are formed from at least two HDI molecules and still have at least two isocyanate groups, by contrast, are "oligomeric polyisocyanates" within the context of the invention. Representatives of such "oligomeric polyisocyanates" are, proceeding from monomeric HDI, for example, HDI isocyanurate and HDI biuret, each of which are formed from three monomeric HDI units:
##STR00003##
[0041] In the context of the invention the term "siloxane group" describes a group formed by hydrolysis/condensation of silicon-containing functional groups of the silicon-containing compounds and/or of the silicon-modified polyisocyanates. The term "siloxane" as used here is a designation for compounds of general structure --[SiR.sub.2--O--SiR.sub.2].sub.n--, wherein R typically represents a hydrogen atom, an alkyl group, an alkoxy group or an --O--SiR.sub.2-- radical where R is as defined above.
[0042] The polyisocyanurate plastic according to the invention containing siloxane groups is obtainable by the process according to the invention. Described hereinbelow therefore are embodiments relevant both to the inventive polyisocyanurate plastic containing siloxane groups obtainable by the process according to the invention and to the process according to the invention as such.
[0043] According to the invention the composition A) used as reactant for producing the siloxane-containing polyisocyanurate plastic according to the invention is low in monomers (i.e. low in monomeric diisocyanates) and already contains oligomeric polyisocyanates B), silicon-containing compounds C) or oligomeric silicon-modified polyisocyanates D) or oligomeric silicon-modified polyisocyanates D) and oligomeric polyisocyanates B) or oligomeric polyisocyanates B), silicon-containing compounds C) and modified oligomeric polyisocyanates D) or silicon-containing compounds C) and silicon-modified, oligomeric polyisocyanates D).
[0044] In one embodiment of the invention the composition A) contains oligomeric polyisocyanates B), silicon-containing compounds C) and/or oligomeric silicon-modified polyisocyanates D) in amounts such that the composition A) has an NCO content of 5.0% to 23.5% by weight and a silicon content (calculated with an Si molecular weight of 28.g/mol) of 0.1% to 30% by weight.
[0045] The presence of oligomeric polyisocyanates B), silicon-containing compounds C) and/or oligomeric, silicon-modified polyisocyanates D) provided for in accordance with the invention and the resulting NCO and Si contents relate to the composition A) provided originally, i.e. before commencement of the catalytic trimerization.
[0046] The process according to the invention forms siloxane-containing polyisocyanurate plastics having a homogeneous distribution of the siloxane groups in the polyisocyanurate plastic, i.e. the product is not a polyisocyanurate plastic modified with silicon-containing compounds C) exclusively at the surface.
[0047] The terms "low in monomers" and "low in monomeric diisocyanates" are here used synonymously in relation to the composition A).
[0048] Results of particular practical relevance are established when the composition A) has a proportion of monomeric diisocyanates in the composition A) of not more than 20% by weight, especially not more than 15% by weight or not more than 10% by weight, based in each case on the weight of the composition A). It is preferable when the composition A) has a content of monomeric diisocyanates of not more than 5% by weight, preferably not more than 2.0% by weight, particularly preferably not more than 1.0% by weight, based in each case on the weight of the composition A). Particularly good results are established when the composition A) is essentially free of monomeric diisocyanates. "Essentially free" here means that the content of monomeric diisocyanates is not more than 0.5% by weight, based on the weight of the composition A).
[0049] It is essential to the invention that the composition A) used is low in monomers. In practice this can be achieved in particular when the composition A) contains oligomeric polyisocyanates B) and/or oligomeric silicon-modified polyisocyanates D) in whose production the actual oligomerization reaction is in each case followed by at least one further process step for removal of unconverted excess monomeric diisocyanates and/or monomeric, silicon-modified diisocyanates. In a manner of particular practical relevance, this monomer removal can be effected by processes known per se, preferably by thin-film distillation under high vacuum or by extraction with suitable solvents that are inert toward isocyanate groups, for example aliphatic or cycloaliphatic hydrocarbons such as pentane, hexane, heptane, cyclopentane or cyclohexane.
[0050] In a preferred embodiment of the invention the oligomeric polyisocyanates B) are obtained by oligomerization of monomeric diisocyanates with subsequent removal of unconverted monomers.
[0051] The processes for producing polyisocyanurate plastics described in the prior art use very substantially monomeric diisocyanates as reactants, i.e. pure monomeric diisocyanates or monomer-rich polyisocyanate compositions are catalytically trimerized. By contrast, the inventive use, or the "provision" of a low-monomer composition A) already containing oligomeric polyisocyanates B) and/or oligomeric, silicon-modified polyisocyanates D), surprisingly leads to markedly lower volume shrinkage. The relatively low exothermicity of the inventive reaction additionally allows polyisocyanurate plastics with a high conversion level to be obtained.
[0052] Preferably, no monomeric diisocyanate is used in the trimerization reaction of the invention. However, in one particular embodiment of the invention the composition A) may comprise an outside monomeric diisocyanate. In this context, "outside monomeric diisocyanate" means that said diisocyanate differs from the monomeric diisocyanates used for producing the oligomeric polyisocyanates B) and/or the oligomeric silicon-modified polyisocyanates D) present in the composition A). Addition of outside monomeric diisocyanate may be advantageous for achievement of special technical effects, for example an exceptional hardness. Results of particular practical relevance are established when the composition A) has a proportion of outside monomeric diisocyanate in the composition A) of not more than 20% by weight, especially not more than 15% by weight or not more than 10% by weight, based in each case on the weight of the composition A). It is preferable when the composition A) has a content of outside monomeric diisocyanate of not more than 5% by weight, preferably not more than 2.0% by weight, particularly preferably not more than 1.0% by weight, based in each case on the weight of the composition A).
[0053] In a further particular embodiment of the invention, the composition A) may contain monomeric monoisocyanates or monomeric isocyanates having an isocyanate functionality greater than two, i.e. having more than two isocyanate groups per molecule. The addition of monomeric monoisocyanates or monomeric isocyanates having an isocyanate functionality greater than two has been found to be advantageous in order to influence the network density of the polyisocyanurate plastic. Results of particular practical relevance are established when the composition A) has a proportion of monomeric monoisocyanates or monomeric isocyanates having an isocyanate functionality greater than two in the composition A) of not more than 20% by weight, especially not more than 15% by weight or not more than 10% by weight, based in each case on the weight of the composition A). It is preferable when the composition A) has a content of monomeric monoisocyanates or monomeric isocyanates having an isocyanate functionality greater than two of not more than 5% by weight, preferably not more than 2.0% by weight, particularly preferably not more than 1.0% by weight, based in each case on the weight of the composition A). Preferably, no monomeric monoisocyanate or monomeric isocyanate having an isocyanate functionality greater than two is used in the trimerization reaction of the invention.
[0054] The oligomeric polyisocyanates B) are typically obtained by oligomerization of simple aliphatic, cycloaliphatic, araliphatic and/or aromatic monomeric diisocyanates or mixtures of such monomeric diisocyanates.
[0055] The oligomeric polyisocyanates B) and/or oligomeric silicon-modified polyisocyanates D) may, in accordance with the invention, especially have uretdione, isocyanurate, allophanate, thioallophanate, biuret, iminooxadiazinedione and/or oxadiazinetrione structure. In one embodiment of the invention, the oligomeric polyisocyanates B) and/or oligomeric silicon-modified polyisocyanates D) have at least one of the following oligomeric structure types or mixtures thereof:
##STR00004##
[0056] In a particularly preferred embodiment of the invention the oligomeric polyisocyanates B) contain at least one structure selected from the group consisting of uretdione, isocyanurate, biuret, iminooxadiazinedione and oxadiazinetrione but are essentially allophanate-free. In this context the term "allophanate-free" means that the molar ratio of allophanate groups to all of the abovementioned groups present in the oligomeric polyisocyanate B) is preferably less than 1:99. It is more preferable when the molar ratio of allophanate groups to isocyanurate groups is below 1:99. It is particularly preferable when the oligomeric polyisocyanate B) contains no allophanate groups. All of the other definitions given hereinabove in relation to the oligomeric polyisocyanate B, also apply to this embodiment.
[0057] It has been found that, surprisingly, it can be advantageous to use oligomeric polyisocyanates B) and/or oligomeric silicon-modified polyisocyanates D) that constitute a mixture of at least two oligomeric polyisocyanates B) and/or at least two oligomeric silicon-modified polyisocyanates D) where the at least two oligomeric polyisocyanates B) and/or the at least two oligomeric silicon-modified polyisocyanates D) differ in terms of their structure. This structure is preferably selected from the group consisting of uretdione, isocyanurate, allophanate, thioallophanate, biuret, iminooxadiazinedione and oxadiazinetrione structures, and mixtures thereof. Particularly compared to trimerization reactions with oligomeric polyisocyanates B) and/or oligomeric silicon-modified polyisocyanates D) of just one defined structure, starting mixtures of this kind can lead to an effect on the Tg value, which is advantageous for many applications.
[0058] It is preferable to employ in accordance with the invention a composition A) containing oligomeric polyisocyanates B) and/or oligomeric, silicon-modified polyisocyanates D) having biuret, allophanate, thioallophanate, isocyanurate and/or iminooxadiazinedione structure and mixtures thereof.
[0059] In another embodiment, the composition A) is one containing oligomeric polyisocyanates B) and/or oligomeric silicon-modified polyisocyanates D) of only a single defined oligomeric structure, for example exclusively or for the most part isocyanurate structure. However, as a consequence of production the composition A) generally always contains oligomeric polyisocyanates B) and/or oligomeric silicon-modified polyisocyanates D) of a plurality of different oligomeric structures simultaneously.
[0060] According to the invention in a further embodiment oligomeric polyisocyanates B) and/or oligomeric silicon-modified polyisocyanates D) of a single defined oligomeric structure are used, the oligomeric structure being selected from uretdione, isocyanurate, allophanate, thioallophanate, biuret, iminooxadiazinedione and/or oxadiazinetrione structure.
[0061] In a further embodiment, the oligomeric polyisocyanates B) and/or oligomeric silicon-modified polyisocyanates D) are those which have mainly isocyanurate structure and which may contain the abovementioned uretdione, allophanate, thioallophanate, biuret, iminooxadiazinedione and/or oxadiazinetrione structure only as by-products.
[0062] It is likewise possible in accordance with the invention to use oligomeric polyisocyanates B) and/or oligomeric silicon-modified polyisocyanates D) having very substantially no isocyanurate structure, and containing mainly at least one of the abovementioned uretdione, allophanate, biuret, iminooxadiazinedione and/or oxadiazinetrione structure types. In a particular embodiment of the invention the composition A) contains oligomeric polyisocyanates B) and/or oligomeric, silicon-modified polyisocyanates D) having a structure type selected from the group consisting of uretdione, allophanate, thioallophanate, biuret, iminooxadiazinedione and/or oxadiazinetrione structure.
[0063] The uretdione, isocyanurate, allophanate, thioallophanate, biuret, iminooxadiazinedione and/or oxadiazinetrione structure in the oligomeric polyisocyanates B) and/or the oligomeric silicon-modified polyisocyanates D) can be determined, for example, by NMR spectroscopy.
[0064] It is possible here with preference to use .sup.13C NMR spectroscopy, preferably in proton-decoupled form, since the oligomeric structures mentioned give characteristic signals.
[0065] Irrespective of the underlying oligomeric structure type (uretdione, isocyanurate, allophanate, thioallophanate, biuret, iminooxadiazinedione and/or oxadiazinetrione structure) the oligomeric polyisocyanates B) present in accordance with the invention in the composition A) preferably have an (average) NCO functionality of 2.0 to 5.0, preferably of 2.3 to 4.5. Irrespective of the underlying oligomeric structure type (uretdione, isocyanurate, allophanate, thioallophanate biuret, iminooxadiazinedione and/or oxadiazinetrione structure) the oligomeric, silicon-modified polyisocyanates D) present according to the invention in the composition A) preferably have an (average) NCO functionality of 1.8 to 6.0, preferably of 2.0 to 5.0.
[0066] Results of particular practical relevance are established when the composition A) to be used in accordance with the invention has a content of isocyanate groups of 6.0% to 23.0% by weight. It has been found to be of particular practical relevance when the composition A) according to the invention has a content of isocyanate groups of 10.0% to 21.5% by weight, based in each case on the weight of the composition A).
[0067] Production processes for the oligomeric polyisocyanates B) having uretdione, isocyanurate, allophanate, thioallophanate, biuret, iminooxadiazinedione and/or oxadiazinetrione structure to be used according to the invention in the low-monomer composition A) are described, for example, in J. Prakt. Chem. 336 (1994) 185-200, in DE-A 1 670 666, DE-A 1 954 093, DE-A 2 414 413, DE-A 2 452 532, DE-A 2 641 380, DE-A 3 700 209, DE-A 3 900 053 and DE-A 3 928 503 or in EP-A 0 336 205, EP-A 0 339 396 and EP-A 0 798 299.
[0068] In an additional or alternative embodiment of the invention, the composition A) according to the invention is defined in that it contains oligomeric polyisocyanates B) which have been obtained from monomeric diisocyanates, irrespective of the nature of the oligomerization reaction used, with observation of an oligomerization level of 5% to 45%, preferably 10% to 40%, more preferably 15% to 30%. "Oligomerization level" is understood here to mean the percentage of isocyanate groups originally present in the starting mixture which are consumed during the production process to form uretdione, isocyanurate, allophanate, biuret, iminooxadiazinedione and/or oxadiazinetrione structures.
[0069] Suitable monomeric polyisocyanates for producing the oligomeric polyisocyanates B) are any desired polyisocyanates obtainable in various ways, for example by phosgenation in the liquid or gas phase or by a phosgene-free route, for example by thermal urethane cleavage.
[0070] Particularly good results are established when the polyisocyanates are monomeric diisocyanates. Preferred monomeric diisocyanates are those having a molecular weight in the range from 140 to 400 g/mol, having aliphatically, cycloaliphatically, araliphatically and/or aromatically bonded isocyanate groups, for example 1,4-diisocyanatobutane (BDI), 1,5-diisocyanatopentane (PDI), 1,6-diisocyanatohexane (HDI), 2-methyl-1,5-diisocyanatopentane, 1,5-diisocyanato-2,2-dimethylpentane, 2,2,4- or 2,4,4-trimethyl-1,6-diisocyanatohexane, 1,10-diisocyanatodecane, 1,3- and 1,4-diisocyanatocyclohexane, 1,4-diisocyanato-3,3,5-trimethylcyclohexane, 1,3-diisocyanato-2-methylcyclohexane, 1,3-diisocyanato-4-methylcyclohexane, I-isocyanato-3,3,5-trimethyl-5-isocyanatomethylcyclohexane (isophorone diisocyanate; IPDI), 1-isocyanato-1-methyl-4(3)-isocyanatomethylcyclohexane, 2,4'- and 4,4'-diisocyanatodicyclohexylmethane (HI2MDI), 1,3- and 1,4-bis(isocyanatomethyl)cyclohexane, bis(isocyanatomethyl)norbornane (NBDI), 4,4'-diisocyanato-3,3'-dimethyldicyclohexylmethane, 4,4'-diisocyanato-3,3',5,5'-tetramethyldicyclohexylmethane, 4,4'-diisocyanato-1,1'-bi(cyclohexyl), 4,4'-diisocyanato-3,3'-dimethyl-1,1'-bi(cyclohexyl), 4,4'-diisocyanato-2,2',5,5'-tetramethyl-1,1'-bi(cyclohexyl), 1,8-diisocyanato-p-menthane, 1,3-diisocyanatoadamantane, 1,3-dimethyl-5,7-diisocyanatoadamantane, 1,3- and 1,4-bis(isocyanatomethyl)benzene (xylylene diisocyanate; XDI), 1,3- and 1,4-bis(1-isocyanato-1-methylethyl)benzene (TMXDI) and bis(4-(l-isocyanato-1-methylethyl)phenyl) carbonate, 2,4- and 2,6-diisocyanatotoluene (TDI), 2,4'- and 4,4'-diisocyanatodiphenylmethane (MDI), 1,5-diisocyanatonaphthalene and any desired mixtures of such diisocyanates. Further diisocyanates that are likewise suitable can additionally be found, for example, in Justus Liebigs Annalen der Chemie, volume 562 (1949) p. 75-136.
[0071] In addition, it is also possible according to the invention to use conventional prepolymers bearing aliphatic or aromatic isocyanate end groups, for example polyether, polyester, polycarbonate prepolymers bearing aliphatic or aromatic isocyanate end groups, as mono- and polyisocyanates in the composition A).
[0072] Suitable monomeric monoisocyanates which can optionally be used in the composition A) are, for example, n-butyl isocyanate, n-amyl isocyanate, n-hexyl isocyanate, n-heptyl isocyanate, n-octyl isocyanate, undecyl isocyanate, dodecyl isocyanate, tetradecyl isocyanate, cetyl isocyanate, stearyl isocyanate, cyclopentyl isocyanate, cyclohexyl isocyanate, 3- or 4-methylcyclohexyl isocyanate or any desired mixtures of such monoisocyanates. An example of a monomeric isocyanate having an isocyanate functionality greater than two which can optionally be added to the composition A) is 4-isocyanatomethyloctane 1,8-diisocyanate (triisocyanatononane; TIN).
[0073] In one embodiment of the invention, the composition A) contains not more than 30% by weight, especially not more than 20% by weight, not more than 15% by weight, not more than 10% by weight, not more than 5% by weight or not more than 1% by weight, based in each case on the weight of the composition A), of aromatic polyisocyanates. As used here, "aromatic polyisocyanate" means a polyisocyanate having at least one aromatically bonded isocyanate group.
[0074] Aromatically bonded isocyanate groups are understood to mean isocyanate groups bonded to an aromatic hydrocarbyl radical.
[0075] In a preferred embodiment of the invention, a composition A) having exclusively aliphatically and/or cycloaliphatically bonded isocyanate groups is used.
[0076] Aliphatically and cycloaliphatically bonded isocyanate groups are understood to mean isocyanate groups bonded, respectively, to an aliphatic and cycloaliphatic hydrocarbyl radical.
[0077] In another preferred embodiment of the invention, a composition A) containing one or more oligomeric polyisocyanates B) is used, wherein the one or more oligomeric polyisocyanates B) comprise exclusively aliphatically and/or cycloaliphatically bonded isocyanate groups.
[0078] Practical experiments have shown that particularly good results can be achieved with compositions A) in which the oligomeric polyisocyanates present therein comprise exclusively aliphatically and/or cycloaliphatically bonded isocyanate groups.
[0079] In a particularly preferred embodiment of the invention, a composition A) is used which contains one or more oligomeric polyisocyanates B), where the one or more oligomeric polyisocyanates is/are based on 1,5-diisocyanatopentane, (PDI), 1,6-diisocyanatohexane (HDI), isophorone diisocyanate (IPDI) or 4,4'-diisocyanatodicyclohexylmethane (HI2MDI) or mixtures thereof.
[0080] The oligomeric polyisocyanates B) employed in the composition A) in the catalytic trimerization preferably have a low viscosity. When reference is made here to "low viscosity" this means that the oligomeric polyisocyanates B) have at room temperature a viscosity below the gel point of the oligomeric polyisocyanates B), i.e. the oligomeric polyisocyanates B) are in the form of a liquid and not a gel. The gel point is understood to mean the time at which the crosslinking density of the oligomeric polyisocyanates B) has progressed to an extent such that the viscosity of the oligomeric polyisocyanates B) abruptly increases and the oligomeric polyisocyanates B) gel to afford a polyisocyanurate bulk material, i.e. no longer undergo substantial deformation or flow.
[0081] The oligomeric silicon-modified polyisocyanates D) are typically obtained by oligomerization of simple aliphatic, cycloaliphatic, araliphatic and/or aromatic monomeric diisocyanates as previously described as starting diisocyanates for the production of the oligomeric polyisocyanates B) in admixture with silicon-modified diisocyanates and/or isocyanatoalkylsilanes or by partial reaction of oligomeric polyisocyanates B) with isocyanate-reactive silicon-containing compounds C). The terms "partial reaction with silicon-containing compounds" and "silicon-modified" are used interchangeably in the context of the invention and mean in particular that I to 99.9, preferably 5 to 80, particularly preferably 10 to 50, very particularly preferably 15 to 40, mol % of the isocyanate groups originally present in the oligomeric polyisocyanate or in the diisocyanate have been reacted with silicon-containing compounds C). In other words the oligomeric, silicon-modified polyisocyanates D) produced by partial reaction of oligomeric polyisocyanates B) with silicon-containing compounds C) have an isocyanate content of 99.0 to 0.1 mol %, preferably 95 to 20 mol %, particularly preferably 90 to 50 mol %, very particularly preferably 85 to 60 mol %, based on the originally present isocyanate groups of the oligomeric polyisocyanates B).
[0082] Production processes for the oligomeric, silicon-modified polyisocyanates D) having uretdione, isocyanurate, allophanate, biuret, iminooxadiazinedione and/or oxadiazinetrione structure to be used according to the invention in the low-monomer composition A) and obtainable by reaction of oligomeric polyisocyanates B) with isocyanate-reactive silicon-containing compounds C) are described for example in EP-A 1 273 640, WO-A 2008/074490, WO-A 2008/074489, WO-A 2014/086530, WO-A 2010/149236, WO-A 2009/156148.
[0083] Further oligomeric, silicon-modified polyisocyanates D) also include for example the allophanate-containing and silane-containing polyisocyanates described in EP-A 2 014 692 and EP-A 2 305 691 that are obtainable by reaction of silane-containing hydroxyurethanes and/or hydroxyamides with excess amounts of monomeric diisocyanates.
[0084] Further oligomeric, silicon-modified polyisocyanates D) also include for example the alkoxysilane-functional biuret polyisocyanates obtainable by reaction of amino silanes with HDI, according to the process of WO 2005/070942.
[0085] Also usable according to the invention as oligomeric, silicon-modified polyisocyanates D) in the low-monomer composition A) are the silane-containing thioallophanate polyisocyanates described in the present applicant's own as yet unpublished patent application BMS 141043-EP (application No: 14 172 295.9).
[0086] Silicon-containing compounds C) that may be used include organic and inorganic silicon-containing compounds C). "Inorganic silicon-containing compounds" is understood to mean silicon-containing compounds which do not comprise a silicon-carbon bond. Inorganic silicon-containing compounds C) suitable according to the invention are for example SiO.sub.2 nanoparticles or organosols or silicates.
[0087] The silicon-containing compound C) need not comprise isocyanate-reactive functional groups.
[0088] Silicon-containing compounds C) inert toward isocyanate groups are for example reaction products of diisocyanates or oligomeric polyisocyanates in which the originally present isocyanate groups have been completely reacted with isocyanate-reactive silicon-containing compounds C). Examples of such silicon-containing compounds C) are for example the isocyanate-free reaction products of monomeric diisocyanates and/or oligomeric polyisocyanates with secondary aminosilanes described in WO 2006/042584 and WO 2007/033786.
[0089] A further type of silicon-containing compound C) inert toward isocyanate groups is also reaction products of isocyanate-functional silane building blocks with at least equimolar amounts of hydroxyl-functional and/or amino-functional compounds. Examples of such silicon-containing compounds C) are for example the reaction products of isocyanatoalkylalkoxysilanes with diols or polyols described in WO 2008/034409 or WO 2013/189882 or the reaction products of diols and/or polyols with specific thiourethane-containing isocyanatosilanes described in WO 2014/037265.
[0090] However, in a preferred embodiment of the invention the silicon-containing compounds C) have at least one isocyanate-reactive functional group. This allows in situ modification of the oligomeric polyisocyanates B) optionally likewise present in the composition A), thus affording oligomeric silicon-modified polyisocyanates D). Usable isocyanate-reactive functional groups include in particular hydroxyl groups, thiol groups, amino groups, amide groups and isocyanate groups.
[0091] It has proven of particular practical relevance to use silicon-containing compounds C) selected from the group consisting of aminosilanes E), silane-functional aspartic esters F), silane-functional alkylamides G), mercaptosilanes H), isocyanatosilanes I) and mixtures thereof. These each contain at least one isocyanate-reactive functional group.
[0092] Suitable aminosilanes E) are for example 3-aminopropyltrimethoxysilane, 3-am inopropyltriethoxysilane, 3-aminopropylmethyldimethoxysilane, 3-aminopropylmethyldiethoxysilane, 3-aminopropylethyldiethoxysilane, 3-aminopropyldimethylethoxysilane, 3-aminopropyldiisopropylethoxysilane, 3-am inopropyltripropoxysilane, 3-aminopropyltributoxysilane, 5 3-aminopropyiphenyldiethoxysilane, 3-aminopropylphenyldimethoxysilane, 3-aminopropyltris(methoxyethoxyethoxy)silane, 2-aminoisopropyltrimethoxysilane, 4-aminobutyltrimethoxysilane, 4-aminobutyltriethoxysilane, 4-aminobutylmethyldimethoxysilane, 4-aminobutylmethyldiethoxysilane, 4-aminobutylethyldimethoxysilane, 4-aminobutylethyldiethoxysilane, 4-aminobutyldimethylmethoxysilane, 4-aminobutylphenyldimethoxysilane, 4-am inobutylphenyldiethoxysilane, 4-amino(3-methylbutyl)methyldimethoxysilane, 4-amino(3-methylbutyl)methyldiethoxysilane, 4-amino(3-methylbutyl)trimethoxysilane, 3-aminopropylphenylmethyl-n-propoxysilane, 3-aminopropylmethyldibutoxysilane, 3-aminopropyldiethylmethylsilane, 3-aminopropylmethylbis(trimethylsiloxy)silane, 11-aminoundecyltrimethoxysilane, N-methyl-3-aminopropyltrimethoxysilane, N-methyl-3-aminopropyltriethoxysilane, N-(n-butyl)-3-aminopropyltrimethoxysilane, N-(n-butyl)-3-aminopropyltriethoxysilane, N-(2-aminoethyl)-3-aminopropyltrimethoxysilane, N-(2-aminoethyl)-3-aminoisobutylmethyldimethoxysilane, N-(2-aminoethyl)-3-aminopropylmethyldimethoxysilane, N-(2-aminoethyl)-3-aminopropyltris(2-ethylhexoxy)silane, N-(6-aminohexyl)-3-aminopropyltrimethoxysilane, N-benzyl-N-(2-aminoethyl)-3-aminopropyltrimethoxysilane, bis(3-trimethoxysilylpropyl)amine, bis(3-triethoxysilylpropyl)amine, (aminoethylaminomethyl)phenethyltrimethoxysilane, N-vinylbenzyl-N-(2-aminoethyl)-3-aminopropylpolysiloxane, N-vinylbenzyl-N(2-aminoethyl)-3-aminopropylpolysiloxane, 3-ureidopropyltriethoxysilane, 3-(m-aminophenoxy)propyltrimethoxysilane, m- and/or p-aminophenyltrimethoxysilane, 3-(3-aminopropoxy)-3,3-dimethyl-1-propenyltrimethoxysilane, 3-aminopropylmethylbis(trimethylsiloxy)silane, 3-aminopropyltris(trimethylsiloxy)silane, 3-aminopropylpentamethyldisiloxane or any desired mixtures of such aminosilanes.
[0093] Very particularly preferred aminosilanes E) are N-methyl-3-aminopropyltrimethoxysilane, N-methyl-3-aminopropyltriethoxysilane, N-(n-Butyl)-3-aminopropyltrimethoxysilane, N-(n-butyl)-3-aminopropyltriethoxysilane, bis(3-trimethoxysilylpropyl)amine and/or bis(3-triethoxysilylpropyl)amine.
[0094] Suitable silane-functional aspartic esters F) are obtainable according to the teaching of EP-A 0 596 360 by reaction of aminosilanes bearing primary amino groups with fumaric esters and/or maleic esters.
[0095] Particularly preferred silane-functional aspartic esters F) are reaction products of 3-aminopropyltrimethoxysilane and/or 3-aminopropyltriethoxysilane with diethyl maleate.
[0096] Silane-functional alkylamides G) suitable according to the invention are obtainable, for example, by the processes disclosed in U.S. Pat. Nos. 4,788,310 and 4,826,915 by reaction of aminosilanes bearing primary amino groups with alkyl alkylcarboxylates with elimination of alcohol.
[0097] Particularly preferred silane-functional alkylamides G) are reaction products of 3-aminopropyltrimethoxysilane and/or 3-aminopropyltriethoxysilane with methyl formate and/or ethyl formate.
[0098] Examples of mercaptosilanes H) suitable according to the invention are for example 2-mercaptoethyltrimethylsilane, 2-mercaptoethylmethyldimethoxysilane, 2-mercaptoethyltrimethoxysilane, 2-mercaptoethyltriethoxysilane, 3-mercaptopropylmethyldimethoxysi lane, 3-mercaptopropyldimethylmethoxysilane, 3-mercaptopropyltrimethoxysilane, 3-mercaptopropylmethyldiethoxysilane, 3-mercaptopropyltriethoxysilane, 3-mercaptopropylethyldimethoxysilane, 3-mercaptopropylethyldiethoxysilane and/or 4-mercaptobutyltrimethoxysilane.
[0099] Very particularly preferred mercaptosilanes H) are in particular 3-mercaptopropyltrimethoxysilane and/or 3-mercaptopropyltriethoxysilane.
[0100] Isocyanatosilanes I) suitable according to the invention are for example 3-isocyanatopropyltrimethoxysilane, 3-isocyanatopropyltriethoxysilane, 3-isocyanatopropyltriisopropoxysilane, 2-isocyanatoethyltrimethoxysilane, 2-isocyanatoethyltriethoxysilane, 2-isocyanatoethyltriisopropoxysilane, 4-isocyanatobutyltrimethoxysilane, 4-isocyanatobutyltriethoxysilane, 4-isocyanatobutyltriisopropoxysilane, isocyanatomethyltrimethoxysilane, isocyanatomethyltriethoxysilane, isocyanatomethyltriisopropoxysilane or the low-monomer isocyanatosilanes having thiourethane structure obtainable by the process of WO 2014/037279.
[0101] Preferably used isocyanatosilanes I) are 3-isocyanatopropyltrimethoxysilane and/or 3-isocyanatopropyltriethoxysilane.
[0102] In a preferred embodiment of the invention, the silicon-containing compounds C) and the oligomeric silicon-modified polyisocyanates D) comprise at least one structural unit selected from the group consisting of:
a) a structural unit of formula (I)
##STR00005## [0103] wherein [0104] R.sup.1, R.sup.2 and R.sup.3 are identical or different radicals and are each a saturated or unsaturated, linear or branched, aliphatic or cycloaliphatic or an optionally substituted aromatic or araliphatic radical having 1 to 18 carbon atoms, which may optionally contain 1 to 3 heteroatoms from the series of oxygen, sulfur and nitrogen, [0105] X is a linear or branched organic radical which has at least 1 carbon atom and may optionally contain 1 to 2 imino groups (--NH--), and [0106] R.sup.4 is hydrogen, a saturated or unsaturated, linear or branched, aliphatic or cycloaliphatic or an optionally substituted aromatic or araliphatic radical having 1 to 18 carbon atoms or a radical of the formula
[0106] ##STR00006## in which R.sup.1, R.sup.2, R.sup.3 and X are as defined above; b) a structural unit of formula (II)
##STR00007## [0107] wherein R.sup.1, R.sup.2 and R.sup.3 are as defined for formula (I), [0108] X is a linear or branched organic radical having at least 1 carbon atom and [0109] R.sup.5 and R.sup.6 are independently saturated or unsaturated, linear or branched, aliphatic or cycloaliphatic or aromatic organic radicals which have 1 to 18 carbon atoms, are substituted or unsubstituted and/or have heteroatoms in the chain; c) a structural unit of formula (III)
[0109] ##STR00008## [0110] wherein R.sup.1, R.sup.2 and R.sup.3 are as defined for formula (I), [0111] X is a linear or branched organic radical having at least 1 carbon atom and [0112] R.sup.9 is a saturated linear or branched, aliphatic or cycloaliphatic organic radical having 1 to 8 carbon atoms; d) a structural unit of formula (IV)
[0112] ##STR00009## [0113] wherein R.sup.1, R.sup.2 and R.sup.3 are as defined for formula (I) [0114] and [0115] Y is a linear or branched organic radical having at least 1 carbon atom; [0116] and e) a structural unit of formula (V)
[0116] ##STR00010## [0117] wherein R.sup.1, R.sup.2 and R.sup.3 are as defined for formula (I), [0118] and [0119] Y is a linear or branched organic radical having at least 1 carbon atom.
[0120] In a further preferred embodiment of the invention, the silicon-containing compounds C) and the oligomeric silicon-modified polyisocyanates D) comprise at least one structural unit selected from the group consisting of:
a) a structural unit of formula (I)
##STR00011## [0121] wherein [0122] R.sup.1, R.sup.2 and R.sup.3 are each methyl, methoxy and/or ethoxy, with the proviso that at least one of the radicals R.sup.1, R.sup.2 and R.sup.3 is such a methoxy or ethoxy radical, [0123] X is a propylene radical (--CH.sub.2--CH.sub.2--CH.sub.2--), and [0124] R.sup.4 is hydrogen, a methyl radical or a radical of formula
[0124] ##STR00012## in which R.sup.1, R.sup.2, R.sup.3 and X are as defined above; b) a structural unit of formula (II)
##STR00013## [0125] wherein [0126] R.sup.1, R.sup.2, R.sup.3 and X wherein R.sup.1, R.sup.2 and R.sup.3 are as defined for formula (I), [0127] R.sup.5 and R.sup.6 are identical or different radicals and are a methyl-, ethyl-, n-butyl- or 2-ethylhexyl radical; c) a structural unit of formula (III)
[0127] ##STR00014## [0128] wherein [0129] R.sup.1, R.sup.2, R.sup.3 and X wherein R.sup.1, R.sup.2 and R.sup.3 are as defined for formula (I), [0130] R.sup.9 is hydrogen; d) a structural unit of formula (IV)
[0130] ##STR00015## [0131] wherein [0132] R.sup.1, R.sup.2 and R.sup.3 wherein R.sup.1, R.sup.2 and R.sup.3 are as defined for formula (I), Y is a propylene radical (--CH.sub.2--CH.sub.2--CH.sub.2--); and e) a structural unit of formula (V)
[0132] ##STR00016## [0133] wherein R.sup.1, R.sup.2 and R.sup.3 are as defined for formula (I), [0134] and [0135] Y is a linear or branched organic radical having at least 1 carbon atom.
[0136] The polyisocyanurates of the invention are obtainable by catalytic trimerization by the process of the invention. "Catalytic" here means in the presence of a suitable trimerization catalyst L).
[0137] To accelerate the hydrolysis and condensation of the alkoxysilane groups it is also possible to use further catalysts in addition to the trimerization catalysts provided that said further catalysts do not significantly retard or even inhibit the trimerization reaction. Catalysts of this kind are for example bases, for example N-substituted amidines such as 1,5-diazabicyclo[4.3.0]non-5-ene (DBN) and 1,5-diazabicyclo[5.4.0]undec-7-ene (DBU), but also metal salts or organometallic compounds, such as tetraisopropyl titanate, tetrabutyl titanate, titanium(IV) acetylacetonate, aluminum acetylacetonate, aluminum triflate or tin triflate, for example.
[0138] Suitable trimerization catalysts L) for the process according to the invention are in principle any compounds which accelerate the trimerization of isocyanate groups to isocyanurate structures. It is preferable to use a trimerization catalyst L) which does not accelerate or does not significantly accelerate the trimerization reaction below 25.degree. C., in particular below 30.degree. C., preferably below 40.degree. C., but significantly accelerates said reaction above 60.degree. C., in particular above 70.degree. C. "Does not significantly accelerate" here means that below 25.degree. C., in particular below 30.degree. C., preferably below 40.degree. C., the presence of the trimerization catalyst L) in the reaction mixture does not have any significant effect on the reaction rate of the reaction that proceeds in any case. A significant acceleration is understood to mean that above 60.degree. C., in particular above 70.degree. C., the presence of the thermally latent catalyst in the reaction mixture has a distinct effect on the reaction rate of the reaction that proceeds in any case.
[0139] Since isocyanurate formation, depending-on the catalyst used, is frequently accompanied by side reactions, for example dimerization to give uretdione structures or trimerization to form iminooxadiazinediones (called asymmetric trimers), and by allophanatization reactions in the case of presence of urethane groups in the starting polyisocyanate, the term "trimerization" shall also synonymously represent these reactions that proceed additionally in the context of the present invention.
[0140] In a particular embodiment, however, trimerization means that predominantly cyclotrimerizations of at least 50%, preferably at least 60%, particularly preferably at least 70%, in particular at least 80%, of the isocyanate groups present in the composition A) to give isocyanurate structural units are catalyzed. However, side reactions, especially those to give uretdione, allophanate and/or iminooxadiazinedione structures, typically occur and can even be used in a controlled manner in order to advantageously affect, for example, the Tg value of the polyisocyanurate plastic obtained.
[0141] Suitable catalysts L) for the process according to the invention are, for example, simple tertiary amines, for example triethylamine, tributylamine, N,N-dimethylaniline, N-ethylpiperidine or N,N'-dimethylpiperazine. Suitable catalysts are also the tertiary hydroxyalkylamines described in GB 2 221 465, for example triethanolamine, N-methyldiethanolamine, dimethylethanolamine, N-isopropyldiethanolamine and 1-(2-hydroxyethyl)pyrrolidine, or the catalyst systems that are known from GB 2 222 161 and consist of mixtures of tertiary bicyclic amines, for example DBU, with simple low molecular weight aliphatic alcohols.
[0142] Likewise suitable as trimerization catalysts L) for the process of the invention are a multitude of different metal compounds. Suitable examples are the octoates and naphthenates of manganese, iron, cobalt, nickel, copper, zinc, zirconium, cerium or lead that are described as catalysts in DE-A 3 240 613, or mixtures thereof with acetates of lithium, sodium, potassium, calcium or barium, the sodium and potassium salts of linear or branched alkanecarboxylic acids having up to 10 carbon atoms that are known from DE-A 3 219 608, for example of propionic acid, butyric acid, valeric acid, caproic acid, heptanoic acid, caprylic acid, pelargonic acid, capric acid and undecylic acid, the alkali metal or alkaline earth metal salts of aliphatic, cycloaliphatic or aromatic mono- and polycarboxylic acids having 2 to 20 carbon atoms that are known from EP-A 0 100 129, for example sodium or potassium benzoate, the alkali metal phenoxides known from GB-A 1 391 066 and GB-A 1 386 399, for example sodium or potassium phenoxide, the alkali metal and alkaline earth metal oxides, hydroxides, carbonates, aikoxides and phenoxides known from GB 809 809, alkali metal salts of enolizable compounds and metal salts of weak aliphatic or cycloaliphatic carboxylic acids, for example sodium methoxide, sodium acetate, potassium acetate, sodium acetoacetate, lead 2-ethylhexanoate and lead naphthenate, the basic alkali metal compounds complexed with crown ethers or polyether alcohols that are known from EP-A 0 056 158 and EP-A 0 056 159, for example complexed sodium or potassium carboxylates, the pyrrolidinone-potassium salt known from EP-A 0 033 581, the mono- or polynuclear complexs of titanium, zirconium and/or hafnium known from application EP 13196508.9, for example zirconium tetra-n butoxide, zirconium tetra-2-ethylhexanoate and zirconium tetra-2-ethylhexoxide, and tin compounds of the type described in European Polymer Journal, vol. 16, 147-148 (1979), for example dibutyltin dichloride, diphenyltin dichloride, triphenylstannanol, tributyltin acetate, tributyltin oxide, tin octoate, dibutyl(dimethoxy)stannane and tributyltin imidazolate.
[0143] Further trimerization catalysts L) suitable for the process according to the invention are, for example, the quaternary ammonium hydroxides known from DE-A 1 667 309, EP-A 0 013 880 and EP-A 0 047 452, for example tetraethylammonium hydroxide, trimethylbenzylammonium hydroxide, N,N-dimethyl-N-dodecyl-N-(2-hydroxyethyl)ammonium hydroxide, N-(2-hydroxyethyl)-N,N-dimethyl-N-(2,2'-dihydroxymethylbutyl)ammonium hydroxide and 1-(2-hydroxyethyl)-1,4-diazabicyclo[2.2.2]octane hydroxide (monoadduct of ethylene oxide and water with 1,4-diazabicyclo[2.2.2]octane), the quaternary hydroxyalkylammonium hydroxides known from EP-A 37 65 or EP-A 10 589, for example N,N,N-trimethyl-N-(2-hydroxyethyl)ammonium hydroxide, the trialkylhydroxylalkylammonium carboxylates that are known from DE-A 2631733, EP-A 0 671 426, EP-A 1 599 526 and U.S. Pat. No. 4,789,705, for example N,N,N-trimethyl-N-2-hydroxypropylammonium p-tert-butylbenzoate and N,N,N-trimethyl-N-2-hydroxypropylammonium 2-ethylhexanoate, the quaternary benzylammonium carboxylates known from EP-A 1 229 016, such as N-benzyl-N,N-dimethyl-N-ethylammonium pivalate, N-benzyl-N,N-dimethyl-N-ethylammonium 2-ethylhexanoate, N-benzyl-N,N,N-tributylammonium 2-ethylhexanoate, N,N-dimethyl-N-ethyl-N-(4-methoxybenzyl)ammonium 2-ethylhexanoate or N,N,N-tributyl-N-(4-methoxybenzyl)ammonium pivalate, the tetrasubstituted ammonium a-hydroxycarboxylates known from WO 2005/087828, for example tetramethylammonium lactate, the quaternary ammonium or phosphonium fluorides known from EP-A 0 339 396, EP-A 0 379 914 and EP-A 0 443 167, for example N-methyl-N,N,N-trialkylammonium fluorides with C.sub.8-C.sub.10-alkyl radicals, N,N,N,N-tetra-n-butylammonium fluoride, N,N,N-trimetbyl-N-benzylammonium fluoride, tetramethylphosphonium fluoride, tetraethylphosphonium fluoride or tetra-n-butylphosphonium fluoride, the quaternary ammonium and phosphonium polyfluorides known from EP-A 0 798 299, EP-A 0 896 009 and EP-A 0 962 455, for example benzyltrimethylammonium hydrogen polyfluoride, the tetraalkylammonium alkylcarbonates obtainable by reaction of tertiary amines with dialkyl carbonates, or betaine-structured quaternary ammonioalkyl carbonates, which are known from EP-A 0 668 271, the quaternary ammonium hydrogencarbonates known from WO 1999/023128, such as choline bicarbonate, the quaternary ammonium salts which are known from EP 0 102 482 and are obtainable from tertiary amines and alkylating esters of phosphorus acids, examples of such salts being reaction products of triethylamine, DABCO or N-methylmorpholine with dimethyl methanephosphonate, or the tetrasubstituted ammonium salts of lactams that are known from WO 2013/167404, for example trioctylammonium caprolactamate or dodecyltrimethylammonium caprolactamate.
[0144] Further trimerization catalysts L) suitable for the process of the invention can be found, for example, in J. H. Saunders and K. C. Frisch, Polyurethanes Chemistry and Technology, p. 94 ff. (1962) and the literature cited therein.
[0145] The catalysts L) can be used in the process of the invention either individually or in the form of any desired mixtures with one another.
[0146] Preferred catalysts L) are metal compounds of the aforementioned type, especially carboxylates and alkoxides of alkali metals, alkaline earth metals or zirconium, and organic tin compounds of the type mentioned.
[0147] Particularly preferred trimerization catalysts L) are sodium and potassium salts of aliphatic carboxylic acids having 2 to 20 carbon atoms and aliphatically substituted tin compounds.
[0148] Very particularly preferred trimerization catalysts L) for the process according to the invention are potassium acetate, tin octoate and/or tributyltin oxide.
[0149] In one embodiment of the invention catalytic trimerization is carried out in the presence of a trimerization catalyst L), wherein the trimerization catalyst L) preferably comprises at least one alkali metal or alkaline earth metal salt.
[0150] In a preferred embodiment of the invention the trimerization catalyst L) comprises potassium acetate as the alkali metal salt and/or a polyether, in particular a polyethylene glycol.
[0151] In the process according to the invention, the trimerization catalyst L) is generally used in a concentration based on the amount of the composition A) used of 0.0005% to 5.0% by weight, preferably of 0.10% to 2.0% by weight and particularly preferably of 0.25% to 1.0% by weight.
[0152] The trimerization catalysts L) that are used in the process of the invention generally have sufficient solubility in the composition A) in the amounts that are required for initiation of the oligomerization reaction. The catalyst L) is therefore preferably added to the composition A) in neat form.
[0153] Optionally, however, the catalysts L) can also be used dissolved in a suitable organic solvent to improve their incorporability. The dilution level of the catalyst solutions can be chosen freely within a very wide range. Such catalyst solutions are typically catalytically active from a concentration of about 0.01% by weight based on the total weight of catalyst and of the organic solvent.
[0154] Suitable catalyst solvents are, for example, solvents that are inert toward isocyanate groups, for example hexane, toluene, xylene, chlorobenzene, ethyl acetate, butyl acetate, diethylene glycol dimethyl ether, dipropylene glycol dimethyl ether, ethylene glycol monomethyl or monoethyl ether acetate, diethylene glycol ethyl and butyl ether acetate, propylene glycol monomethyl ether acetate, I-methoxyprop-2-yl acetate, 3-methoxy-n-butyl acetate, propylene glycol diacetate, acetone, methyl ethyl ketone, methyl isobutyl ketone, cyclohexanone, lactones such as P-propiolactone, y-butyrolactone, .epsilon.-caprolactone and .epsilon.-methylcaprolactone, but also solvents such as N-methylpyrrolidone and N-methylcaprolactam, 1,2-propylene carbonate, methylene chloride, dimethyl sulfoxide, triethyl phosphate or any desired mixtures of such solvents.
[0155] If catalyst solvents are used in the process of the invention, preference is given to using catalyst solvents which bear groups reactive toward isocyanates and can be incorporated into the polyisocyanurate plastic. Examples of such solvents are mono- or polyhydric simple alcohols, for example methanol, ethanol, n-propanol, isopropanol, n-butanol, n-hexanol, 2-ethyl-1-hexanol, ethylene glycol, propylene glycol, the isomeric butanediols, 2-ethylhexane-1,3-diol or glycerol; ether alcohols, for example 1-methoxy-2-propanol, 3-ethyl-3-hydroxymethyloxetane, tetrahydrofurfuryl alcohol, ethylene glycol monomethyl ether, ethylene glycol monoethyl ether, ethylene glycol monobutyl ether, diethylene glycol monomethyl ether, diethylene glycol monoethyl ether, diethylene glycol monobutyl ether, diethylene glycol, dipropylene glycol or else liquid higher molecular weight polyethylene glycols, polypropylene glycols, mixed polyethylene/polypropylene glycols and the monoalkyl ethers thereof; ester alcohols, for example ethylene glycol monoacetate, propylene glycol monolaurate, glycerol mono- and diacetate, glycerol monobutyrate or 2,2,4-trimethylpentane-1,3-diol monoisobutyrate; unsaturated alcohols, for example allyl alcohol, 1,1-dimethylallyl alcohol or oleyl alcohol; araliphatic alcohols, for example benzyl alcohol; N-monosubstituted amides, for example N-methylformamide, N-methylacetamide, cyanoacetamide or 2-pyrrolidinone, or any desired mixtures of such solvents.
[0156] The polyisocyanurate plastics obtainable by the process of the invention, even as such, i.e. without addition of appropriate auxiliaries and additives M), feature very good light stability. Nevertheless, it is optionally also possible to use in the production of said plastics standard auxiliaries and additives M), for example standard fillers, UV stabilizers, antioxidants, mold release agents, water scavengers, slip additives, defoamers, levelling agents, rheology additives, flame retardants and/or pigments. These auxiliaries and additives M), excluding fillers and flame retardants, are typically present in the polyisocyanurate plastic in an amount of less than 10% by weight, preferably less than 5% by weight, particularly preferably up to 3% by weight, based on the composition A). Flame retardants are typically present in the polyisocyanurate plastic in amounts of not more than 70% by weight, preferably not more than 50% by weight, particularly preferably not more than 30% by weight, calculated as the total amount of employed flame retardants, based on the total weight of the composition A).
[0157] Suitable fillers M.sub.w) are, for example AlOH.sub.3, CaCO.sub.3, metal pigments such as TiO.sub.2 and further known standard fillers. These fillers M.sub.w) are preferably used in amounts of not more than 70% by weight, preferably not more than 50% by weight, particularly preferably not more than 30% by weight, calculated as the total amount of employed fillers based on the total weight of the composition A).
[0158] Suitable UV stabilizers M.sub.x) may preferably be selected from the group consisting of piperidine derivatives, for example 4-benzoyloxy-2,2,6,6-tetramethylpiperidine, 4-benzoyloxy-1,2,2,6,6-pentamethylpiperidine, bis(2,2,6,6-tetramethyl-4-piperidyl) sebacate, bis(1,2,2,6,6-pentamethyl-1-4-piperidinyl) sebacate, bis(2,2,6,6-tetramethyl-4-piperidyl) suberate, bis(2,2,6,6-tetramethyl-4-piperidyl) dodecanedioate; benzophenone derivatives, for example 2,4-dihydroxy-, 2-hydroxy-4-methoxy-, 2-hydroxy-4-octoxy-, 2-hydroxy-4-dodecyloxy- or 2,2'-dihydroxy-4-dodecyloxybenzophenone; benzotriazole derivatives, for example 2-(2H-benzotriazol-2-yl)-4,6-di-tert-pentylphenol, 2-(2H-benzotriazol-2-yl)-6-dodecyl-4-methylphenol, 2-(2H-benzotriazol-2-yl)-4,6-bis(1-methyl-1-phenylethyl)phenol, 2-(5-chloro-2H-benzotriazol-2-yl)-6-(1,1-dimethylethyl)-4-methylphenol, 2-(2H-benzotriazol-2-yl)-4-(1,1,3,3-tetramethylbutyl)phenol, 2-(2H-benzotriazol-2-yl)-6-(I-methyl-1-phenylethyl)-4-(1,1,3,3-tetramethy- lbutyl)phenol, isooctyl 3-(3-(2H-benzotriazol-2-yl)-5-(1,1-dimethylethyl)-4-hydroxyphenylpropiona- te), 2-(2H-benzotriazol-2-yl)-4,6-bis(1,1-dimethylethyl)phenol, 2-(2H-benzotriazol-2-yl)-4,6-bis(1-methyl-1-phenylethyl)phenol, 2-(5-chloro-2H-benzotriazol-2-yl)-4,6-bis(1,1-dimethylethyl)phenol; oxalanilides, for example 2-ethyl-2'-ethoxy- or 4-methyl-4'-methoxyoxalanilide; salicylic esters, for example phenyl salicylate, 4-tert-butylphenyl salicylate, 4-tert-octylphenyl salicylate; cinnamic ester derivatives, for example methyl a-cyano-.beta.-methyl-4-methoxycinnamate, butyl a-cyano-.beta.-methyl-4-methoxycinnamate, ethyl a-cyano-.beta.-phenylcinnamate, isooctyl a-cyano-.beta.-phenylcinnamate; and malonic ester derivatives, such as dimethyl 4-methoxybenzylidenemalonate, diethyl 4-methoxybenzylidenemalonate, dimethyl 4 butoxybenzylidenemalonate. These preferred light stabilizers can be used either individually or in any desired combinations with one another.
[0159] Particularly preferred UV stabilizers M.sub.x) for the polyisocyanurate plastics producible according to the invention are those which fully absorb radiation of wavelength <400 nm. These include the recited benzotriazole derivatives for example. Very particularly preferred UV stabilizers are 2-(5-chloro-2H-benzotriazol-2-yl)-6-(1,1-dimethylethyl)-4-methylphenol, 2-(2H-benzotriazol-2-yl)-4-(1,1,3,3-tetramethylbutyl)phenol and/or 2-(5-chloro-2H-benzotriazol-2-yl)-4,6-bis(1,1-dimethylethyl)phenol.
[0160] One or more of the UV stabilizers M.sub.x) recited by way of example are optionally added to the composition A) preferably in amounts of 0.001 to 3.0 wt %, particularly preferably 0.01 to 2 wt %, calculated as the total amount of UV stabilizers used, based on the total weight of the composition A).
[0161] Suitable antioxidants M.sub.y) are preferably sterically hindered phenols, which may be selected preferably from the group consisting of 2,6-di-tert-butyl-4-methylphenol (ionol), pentaerythritol tetrakis(3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionate), octadecyl 3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionate, triethylene glycol bis(3-tert-butyl-4-hydroxy-5-methylphenyl)propionate, 2,2'-thio bis(4-methyl-6-tert-butylphenol) and 2,2'-thiodiethyl bis[3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionate]. These may be used either individually or in any desired combinations with one another as required.
[0162] These antioxidants M.sub.y) are preferably used in amounts of 0.01% to 3.0% by weight, particularly preferably 0.02% to 2.0% by weight, calculated as the total amount of antioxidants used, based on the total weight of the composition A).
[0163] Apart from the small amounts of any catalyst solvents to be used, the process according to the invention may be performed in a solvent-free manner. Especially in the case of the inventive use for production of coatings or films, the polyisocyanate component can optionally also be diluted with organic solvents to reduce the processing viscosity. Solvents suitable for this purpose are, for example, the catalyst solvents inert toward isocyanate groups that have already been described above.
[0164] In the case of the inventive use for production of films, semifinished products or moldings it is finally also possible to add internal mold release agents M.sub.z) as further auxiliaries and additives M).
[0165] These are preferably the nonionic surfactants that contain perfluoroalkyl or polysiloxane units and are known as mold release agents, quaternary alkyl ammonium salts, for example trimethylethylammonium chloride, trimethylstearylammonium chloride, dimethylethylcetylammonium chloride, triethyldodecylammonium chloride, trioctylmethylammonium chloride and diethylcyclohexyldodecylammonium chloride, acidic mono- and dialkyl phosphates having 2 to 18 carbon atoms in the alkyl radical, for example ethyl phosphate, diethyl phosphate, isopropyl phosphate, diisopropyl phosphate, butyl phosphate, dibutyl phosphate, octyl phosphate, dioctyl phosphate, isodecyl phosphate, diisodecyl phosphate, dodecyl phosphate, didodecyl phosphate, tridecanol phosphate, bis(tridecanol) phosphate, stearyl phosphate, distearyl phosphate, and any desired mixtures of such mold release agents.
[0166] Particularly preferred mold release agents M.sub.z) are the acidic mono- and dialkyl phosphates mentioned, very particularly preferably those having 8 to 12 carbon atoms in the alkyl radical.
[0167] Internal mold release agents M.sub.z) are optionally used in the process according to the invention preferably in amounts of 0.01% to 3.0% by weight, particularly preferably 0.02% to 2.0% by weight, calculated as the total amount of internal mold release agent used, based on the total weight of the composition A).
[0168] In one embodiment of the process according to the invention, a trimerization catalyst L) or a mixture of different trimerization catalysts L) is added to the composition A) described, optionally under inert gas, for example nitrogen, and optionally with use of the aforementioned solvents and auxiliaries and additives M), and mixed in homogeneously with the aid of a suitable mixing unit. The addition of catalyst L) and any solvent and auxiliaries and additives M) to be used can be carried out in any sequence, successively or in a mixture, in the above-specified amounts and generally at a temperature of 0 to 100.degree. C., preferably of 15 to 80.degree. C., particularly preferably of 20 to 60.degree. C.
[0169] The hereindescribed inventive polyisocyanurate plastic containing siloxane groups is suitable for producing coatings, films, semifinished products and moldings. Depending on the intended use the application of the reaction mixture may be carried out by different methods known per se.
[0170] For production of films or coatings, for example lacquers, a reaction mixture comprising catalyst L) and composition A) can be applied, for example by spraying, spreading, dipping, flow-coating, printing or with the aid of brushes, rollers or doctor blades, in one or more layers to any desired substrates, for example metal, wood, glass, stone, ceramic materials, concrete, hard and flexible plastics, textiles, leather and paper, which may optionally also be provided with standard primers prior to the coating.
[0171] The invention also provides a substrate coated with a coating containing the inventive polyisocyanurate plastic containing siloxane groups, wherein the substrate may in particular be selected from a vehicle body, in particular a goods vehicle or motor vehicle body, and electronic entertainment devices, such as laptops, tablets or mobile phones. The coating of the substrate with the coating containing the polyisocyanurate plastic according to the invention may be carried out by the abovementioned methods known per se. Either a reaction mixture comprising catalyst L) and composition A) may be applied to the substrate or a separate application of the catalyst L) and of the composition A) onto the substrate may be carried out.
[0172] The catalytic trimerization of the composition A) may in the former case be started either already before application onto the substrate by heating of the reaction mixture at temperatures as defined hereinbelow or by heating, for example, the substrate after application of the reaction mixture at temperatures as defined hereinbelow. The reaction mixture may also be applied to a substrate already heated before application at temperatures as defined hereinbelow.
[0173] For production of solid bodies, for example semifinished products or moldings, the mixture of catalyst L) and composition A) can be introduced into open or closed molds, for example, by simple manual pouring, or with the aid of suitable machinery, for example the low-pressure or high-pressure machinery which is standard in polyurethane technology.
[0174] Subsequently, the trimerization reaction can be started by heating the coated substrates or filled molds for example, wherein depending on the trimerization catalyst L) chosen in each case the optimum reaction temperature is from 45.degree. C. to 200.degree. C., particularly preferably from 60.degree. C. to 150.degree. C., very particularly preferably from 80.degree. C. to 140.degree. C. The reaction temperature can be kept constant within the range specified over the entire curing process to give the polyisocyanurate, or else can be heated, for example, in a linear or stepwise manner up to a temperature greater than 80.degree. C., preferably greater than 100.degree. C., for example up to 130.degree. C., over several hours. Where reference is made here to "reaction temperature", this means the ambient temperature.
[0175] Depending on the catalyst L) chosen and the reaction temperature chosen, the trimerization reaction is very substantially complete, as defined hereinbelow, after a period of less than one minute up to several hours or only after a number of days. The progress of the reaction can initially still be monitored by titrimetric determination of the NCO content, but gelation and solidification of the reaction mixture sets in rapidly as the reaction progresses, which makes wet-chemical analysis methods impossible. The further conversion of isocyanate groups can then be monitored only by spectroscopic methods, for example by IR spectroscopy with reference to the intensity of the isocyanate band at about 2270 cm.sup.-1.
[0176] The inventive polyisocyanurate plastics containing siloxane groups are preferably highly converted siloxane-containing polyisocyanurates, i.e. those in which the trimerization reaction to give polyisocyanurate structures is very substantially complete. A trimerization reaction to give polyisocyanurate structures can be regarded as "very substantially complete" in the context of the present invention when at least 80%, preferably at least 90%, particularly preferably at least 95%, of the free isocyanate groups originally present in the composition A) have reacted. In other words only not more than 20%, not more than 10%, particularly preferably not more than 5%, of the isocyanate groups originally contained in the composition A) preferably remain present in the siloxane-containing polyisocyanurate plastic according to the invention. This can be achieved when in the process according to the invention the catalytic trimerization is continued at least up to a degree of conversion at which only, for example, not more than 20% of the isocyanate groups originally contained in the composition A) remain present, so that a highly converted polyisocyanurate is obtained.
[0177] The percentage of isocyanate groups still present can be determined by comparison of the content of isocyanate groups in % by weight in the original composition A) with the content of isocyanate groups in % by weight in the reaction product, for example by the aforementioned comparison of the intensity of the isocyanate band at about 2270 cm.sup.-1 by means of IR spectroscopy.
[0178] In the inventive polyisocyanurate plastics containing siloxane groups the content of silicon (calculated from the value for silicon dioxide determined gravimetrically after oxidative digestion) based on the polyisocyanurate plastic is from 0.2% to 45%, preferably 1.0% to 25%, in particular from 4% to 15%.
[0179] Results of particular practical relevance are achievable when the polyisocyanurate plastic containing siloxane groups is one which comprises alkoxysiloxane groups. Such an alkoxysiloxane-containing polyisocyanurate plastic is obtainable by the process according to the invention, wherein the silicon-containing compounds C) are alkoxysilyl-containing compounds C) and the oligomeric silicon-modified polyisocyanates D) are oligomeric polyisocyanates D) modified with alkoxysilyl groups.
[0180] The process of the invention affords transparent, yellowing-resistant, siloxane-containing polyisocyanurate plastics which, depending on the nature of the starting polyisocyanate used, not only contain isocyanurate structures and siloxane groups but may also contain further oligomeric structures, such as uretdione, allophanate, biuret, iminooxadiazinedione and/or oxadiazinetrione structures and feature outstanding heat resistances.
[0181] The process according to the invention makes it possible to synthesize highly converted siloxane-containing polyisocyanurate plastics having different properties, for example different hardnesses, mechanical properties or glass transition temperatures in simple fashion by suitable selection of oligomeric polyisocyanates B) and silicon-containing compounds C) and/or oligomeric, silicon-modified polyisocyanates D).
[0182] In contrast to siloxane-containing polyurethane plastics produced by urethanization of the isocyanate groups and hydrolysis/condensation of the silicon-containing functional groups, the inventive siloxane-containing polyisocyanurates feature a significantly higher crosslinking density as a result of which they are suitable in particular for manufacturing coatings having high scratch resistance and chemicals resistance precision. The comparatively low heat of reaction liberated also allows for problem-free production of solid, large-volume moldings.
[0183] The invention is elucidated in greater detail hereinafter with reference to examples.
EXAMPLES
1. Methods
[0184] All reported percentages are based on weight unless otherwise stated.
[0185] The NCO contents were determined by titrimetry to DIN EN ISO 11909.
[0186] OH numbers were determined by titrimetry to DIN 53240-2: 2007-11, acid numbers to DIN 3682 5. The OH contents reported were calculated from the OH numbers determined by analysis.
[0187] The residual monomer contents were measured in accordance with DIN EN ISO 10283 by gas chromatography with an internal standard.
[0188] All viscosity measurements were taken with a Physica MCR 51 rheometer from Anton Paar Germany GmbH (DE) according to DIN EN ISO 3219/A3.
[0189] Pendulum damping: Pendulum damping is measured to DIN EN ISO 1522 on a glass plate and is determined according to Konig.
[0190] Solvent resistance: For this purpose, a small amount of the relevant solvents (xylene, 1-methoxyprop-2-yl acetate, ethyl acetate or acetone) was added to a test tube and provided with a cotton pad at the opening, so that an atmosphere saturated with solvent was formed inside the test tube. The test tubes were subsequently placed with the cotton pad on the lacquer surface where they remained for 5 minutes. Once the solvent had been wiped off, the film was examined for destruction/softening/loss of adhesion (0=no change, 5=film destroyed).
[0191] Hammer test with steel wool (dry scratching):
[0192] A hammer (weight: 800 g without handle) covered with 00 steel wool on its flat side was cautiously placed onto the coated surface at right angles and guided over the coating in a trace without tipping and without additional physical force. 50 back-and-forth strokes were conducted in each case. After being subjected to the scratching medium, the test surface was cleaned with a soft cloth and subsequently the haze in comparison to the value before the scratching was determined according to ASTM D1003 with a BYK-Gardner Haze Gard Plus instrument.
2. Materials
Example 1
[0193] Production of a Polyisocyanate Having Alkoxysilyl Groups:
[0194] In a flask fitted with a thermometer, KPG stirrer, reflux cooler and dropping funnel 536.2 g of Desmodur.RTM. N 3900 [Covestro AG, D, low-monomer polyisocyanurate polyisocyanate based on hexamethylene diisocyanate (HDI), NCO content: 23.5%; viscosity (23.degree. C.): 750 mPas, content of monomeric HDI.ltoreq.0.25%] and 91 mg of zinc trifluoromethanesulfonate were initially charged under a nitrogen atmosphere and heated to 100.degree. C. At this temperature, over the course of 1 h, 373.1 g of N-(3-trimethoxysilylpropyl)formamide (1.8 mol, prepared as per example 1 of WO 2015/113919 A1) were added dropwise. Stirring was continued at 100.degree. C. for a further 3 hours until the free NCO group content had dropped to 4.8%. The batch was admixed with 114 g of butyl acetate and 114 g of solvent naphtha 100 and cooled to room temperature.
[0195] A clear solution was obtained.
[0196] NCO content 4.4%
[0197] Si content: 4.4%
[0198] Solids content 80%
[0199] Viscosity (23.degree. C., shear gradient D=250 l/s) 1030 mPas.
Example 2
[0200] Production of a Polyisocyanate Having Alkoxysilyl Groups
[0201] Based on a working example in WO 2012/168079 (curing agent system VB2-1), by reaction of 100 parts by weight of Desmodur N 3300 (low-monomer polyisocyanurate polyisocyanate based on HDI, NCO content: 21.6%; average NCO functionality: 3.5; viscosity (23.degree. C.): 3200 mPas, content of monomeric HDI.ltoreq.0.25%) with a mixture of 30 parts by weight of bis-[3-(trimethoxysilyl)propyl]amine and 21 parts by weight of N-[3-(trimethoxysilyl)propyl]butylamine in 84 parts by weight of butyl acetate at 50.degree. C., a partially silanized HDI trimerizate was produced. After a reaction time of approximately four hours, the characteristics found for the present solution were as follows:
[0202] NCO content: 6.0%
[0203] Si content: 3.2%
[0204] Solids content: 64%
[0205] Viscosity (23.degree. C., shear gradient D=100 l/s): 70 mPas.
Example 3
[0206] Mixing of an Alkoxysilyl-Functional Diurethane with a Polyisocyanate
[0207] 100 g of VESTANAT.RTM. EP-M 95 [Evonik Industries, DE; solvent-free, trimethoxysilyl-containing crosslinker, produced according to the teaching of WO 2014/180623 (table 1) by reaction of 2 mol of 3-isocyanatopropyltrimethoxysilane with 1 mol of 1,9-nonanediol] were mixed with 200 g of Desmodur.RTM. N 3600 [Covestro AG, DE, low-monomer polyisocyanurate polyisocyanate based on hexamethylene diisocyanate (HDI), NCO content: 23.0%; viscosity (23.degree. C.): 1200 mPas, content of monomeric HDI .quadrature. 0.25%] with exclusion of moisture at room temperature.
[0208] NCO content: 15.3%
[0209] Si content: 3.3%
[0210] Solids content: 100%
[0211] Viscosity (23.degree. C., D=100 l/s): 1300 mPas
Example 4
[0212] Production of a Polyisocyanate Having a Alkoxysilyl Groups
[0213] 756 g (4.5 mol) of hexamethylene diisocyanate (HDI) were introduced under dry nitrogen with stirring at a temperature of 80.degree. C. and 0.1 g of zinc(II) 2-ethyl-1-hexanoate as catalyst were added. Over a period of about 30 minutes, 294 g (1.5 mol) of mercaptopropyltrimethoxysilane were added dropwise, the temperature of the mixture rising to up to 85.degree. C. owing to the exothermic reaction. Stirring of the reaction mixture was continued at 85.degree. C. until, after about 2 hours, the NCO content had dropped to 34.9%. The catalyst was deactivated by addition of 0.1 g of orthophosphoric acid and the unreacted monomeric HDI was removed in a thin-film evaporator at a temperature of 130.degree. C. and a pressure of 0.1 mbar. This gave 693 g of a virtually colorless, clear polyisocyanate mixture having the following characteristics and composition:
[0214] NCO content: 11.8%
[0215] Si content: 6.1%
[0216] Monomeric HDI: 0.06%
[0217] Viscosity (23.degree. C., shear gradient D=100 l/s): 452 mPas
[0218] Thiourethane: 0.0 mol %
[0219] Thioallophanate: 99.0 mol %
[0220] Isocyanurate groups: 1.0 mol %.
[0221] Catalysts
[0222] Cat.1: Production of a Catalytic Potassium Acetate 18-Crown-6 Solution in Diethylene Glycol
[0223] 1.77 g of potassium acetate (Sigma Aldrich), 4.75 g of the crown ether 18-crown-6 from Merck KGaA and 31.15 g of diethylene glycol from Sigma Aldrich were weighed in and stirred for several hours until a clear solution was formed.
[0224] Cat. 2: Tetrabutylammonium Benzoate (TBAB)
[0225] TBAB (tetrabutylammonium benzoate from Sigma Aldrich) was used as a 10% solution in MPA (1-methoxy-2-propyl acetate; 1,2-propylene glycol monomethyl ether acetate).
[0226] Use Example:
[0227] The coating compositions were produced by initially charging the polyisocyanates having alkoxysilyl groups/the mixture according to example 3 and subsequently adding the respective catalyst solutions. Stirring was carried out using a Speed Mixer (Hausschild Engineering, type DAC 150.1 FVZ) at 2750 rpm for 1 min. The clearcoats were subsequently applied to a glass sheet using a 100 .mu.m film casting frame (Byk Gardner) and dried in a recirculating air drying cabinet for 10 minutes at 140.degree. C. After cooling (10 min RT) the films were subjected to visual assessment and the first Konig pendulum damping measurements were taken.
TABLE-US-00001 Comparison Inventive Comparison Inventive Example 5 6 7 8 Product according to example 4 30.00 g 30.00 g 30.00 g 30.00 g Cat. 1 0.94 g 0.94 g Cat. 2 1.50 g 1.50 g Total amount of product + catalyst 30.00 g 30.94 g 31.50 g 32.44 g Curing: 10 min, 140.degree. C. Coating appearance clear, wet clear, dry clear, dry clear, dry but rubbery, scrapeable Pendulum immediately measurable 14 s not 13 s damping measurable after 1 d RT -- 17 s -- 20 s after 3 d RT -- 17 s -- 18 s after 7 d RT -- 45 s -- 22 s after 14 d RT -- 70 s -- 30 s Solvent xylene -- 1 -- 0 resistance 1-methoxyprop-2-yl -- 1 -- 0 after 5 min.; acetate sheets after ethyl acetate -- 2 -- 2 14 d RT acetone -- 2 -- 2 -- = not measured
[0228] To verify the completeness of the NCO reaction the NCO reduction was investigated by IR spectroscopy for the compositions according to examples 6 and 8.
[0229] The coating compositions were produced by weighing in the silane-modified polyisocyanates with the catalysts and their mixtures and subsequently stirring in the Speed Mixer (Hausschild Engineering, type DAC 150.1 FVZ) at 2750 rpm for 1 min.
TABLE-US-00002 Composition according to example 6 8 Integration or NCO (2380-2170) cm.sup.-1 Liquid 100% 100% Dry, immediately 1% 1% after curing
[0230] The test lacquers were analyzed directly (wet lacquer) with an FT-IR spectrometer (Tensor 11 with platinum ATR unit (diamond crystal) from Bruker). The clearcoat was subsequently applied to a glass sheet using a 100 .mu.m film casting frame and dried in a recirculating air drying cabinet for 10 minutes at 140.degree. C. and then immediately after the baking removed as a clearcoat film and reanalyzed.
[0231] The isocyanate reaction is characterized by tracking the intensity of the NCO peak (using an integration in the wavenumber range of 2380-2170 cm.sup.-1), wherein the first measurement of the wet lacquer material after mixing of the components is set to 100% as the starting value. All further measurements (after application, thermal treatment and/or storage) are then calculated relative to the starting value (ratio formation).
[0232] In the measurement on an ATR crystal the intensity of the spectrum is dependent on the penetration depth of the IR beam into the material to be measured and on the covering of the crystal surface. This penetration depth is in turn dependent on the refractive index of the material. Since the refractive index of the wet lacquer differs from the refractive index of the baked clearcoat film and a comparable covering of the crystal surface cannot be ensured for different measurements (on the wet lacquer/on the clearcoat) the ratio calculation must include a correction of both effects by virtue of all spectra being normalized to the CH stretching vibration peak (wavenumber range 3000-2800 cm.sup.-1).
[0233] Examples 6 and 8 (inventive) show, in contrast to the comparative examples 5 and 7 that, the trimerization reaction starts only when the catalyst cat. I is present. The IR measurements verify the virtually complete conversion of the NCO groups. Subsequent post-curing upon storage must then be ascribed to the second curing mechanism of the alkoxysilyl groups. Without catalyst (5) or only with a catalyst (7) which catalyzes the hydrolysis/condensation of alkoxysilyl groups at the chosen temperature, no cured films are produced.
TABLE-US-00003 Inventive Inventive Inventive Inventive Example 8 9 10 11 Product according to example 4 30.00 g Product according to example 1 30.00 g Product according to example 2 30.00 g Product according to example 3 30.00 g Cat. 1 0.94 g 0.94 g 0.94 g 0.94 g Cat. 2 1.50 g 1.50 g 1.50 g 1.50 g Total amount of product + catalyst 32.44 g 32.44 g 32.44 g 32.44 g Curing: 10 min, 140.degree. C. Coating appearance clear, dry clear, dry clear, dry clear, dry Pendulum damping immediately 13 s 24 s 24 s 63 s after 1 d RT 20 s 112 s 70 s 78 s after 3 d RT 18 s 166 s 155 s 161 s after 7 d RT 22 s 173 s 184 s 176 s after 14 d RT 30 s 180 s 195 s 184 s Solvent resistance xylene 0 0 1 1 after 5 min.; sheets 1-methoxyprop-2-yl 0 0 1 1 after 14 d RT acetate ethyl acetate 2 1 2 2 acetone 2 2 2 2
[0234] Examples 8 to 11 show that the properties of the inventive polyisocyanurate plastics can be varied with respect to hardness and solvent resistance.
[0235] The scratch resistance of the coated glass sheets stored for 21 days according to examples 6, 8, 9, 10 and 11 in comparison with a two component polyurethane lacquer was tested. As comparative example 12, 49.06 g of Setalux.RTM. D A 665 BA/X, 65% in butyl acetate/xylene (3:1) (polyacrylate polyol from Nuplex, DE) and 0.50 g of BYK-331, 10% in 1-methoxyprop-2-yl acetate (flow assistant from BYK, DE), were mixed with 16.56 g of Desmodur.RTM. N 3300 using a Speed Mixer (Hausschild Engineering, type DAC 150.1 FVZ) at 2750 rpm for 1 min. The clearcoat was subsequently applied to to a glass sheet using a 100 .mu.m film casting frame (Byk Gardner) and dried in a recirculating air drying cabinet for 30 minutes at 140.degree. C. The lacquer according to comparative example 12 was likewise stored for 21 days at RT.
[0236] The determination of scratch resistance by means of the hammer test gave the following values:
TABLE-US-00004 Example 6 8 9 10 11 12 In- In- In- In- In- Com- ventive ventive ventive ventive ventive parison Delta 122 92 60 81 139 216 haze
[0237] The inventive examples exhibit a smaller increase in haze upon scratching with steel wool than the comparison.
* * * * *





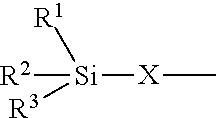





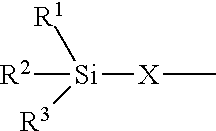
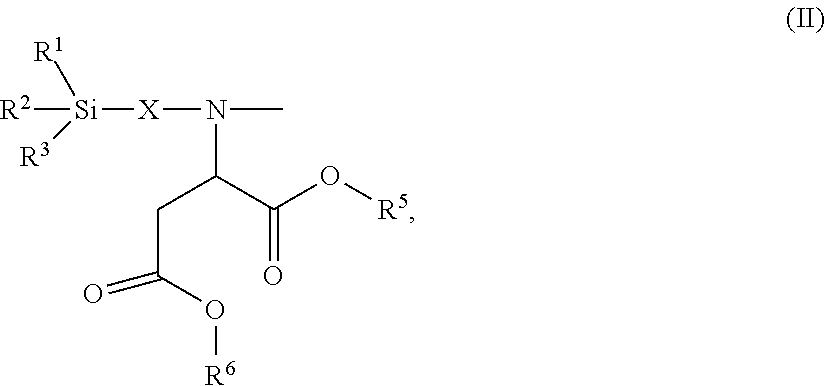




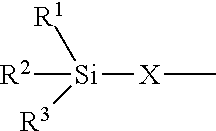
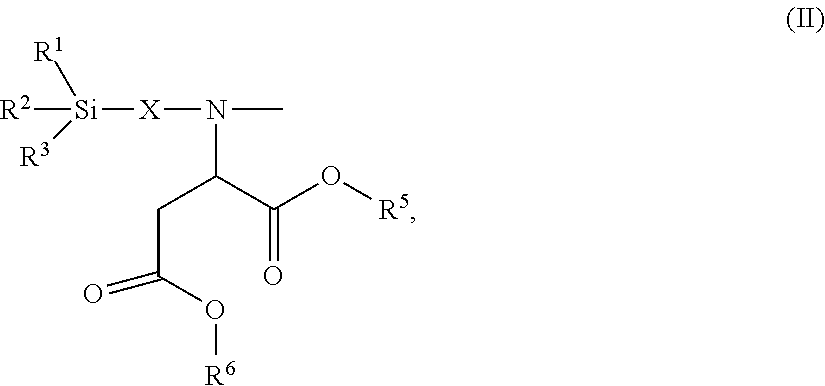




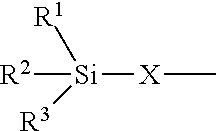
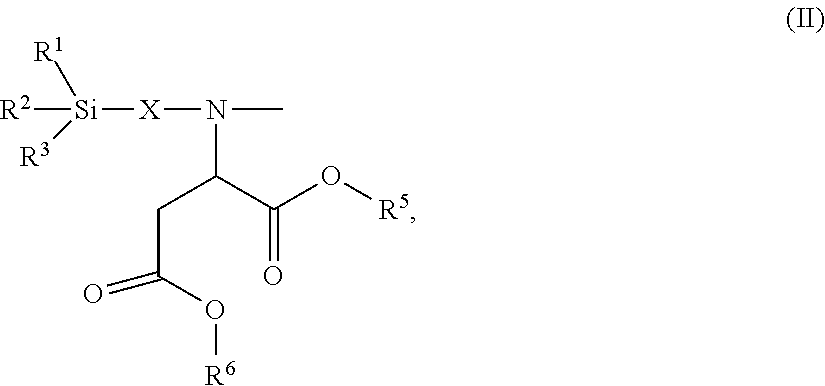



XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.