Crystalline Forms Of Dimethoxy Docetaxel And Methods For Preparing The Same
BILLOT; Pascal ; et al.
U.S. patent application number 16/188687 was filed with the patent office on 2019-05-16 for crystalline forms of dimethoxy docetaxel and methods for preparing the same. The applicant listed for this patent is Aventis Pharma S.A.. Invention is credited to Pascal BILLOT, Marielle DUFRAIGNE, Hagit ELMALEH, Alexandre GIULIANI, Fabrice MANGIN, Patricia RORTAIS, Lionel ZASKE.
| Application Number | 20190144406 16/188687 |
| Document ID | / |
| Family ID | 39712334 |
| Filed Date | 2019-05-16 |




| United States Patent Application | 20190144406 |
| Kind Code | A1 |
| BILLOT; Pascal ; et al. | May 16, 2019 |
CRYSTALLINE FORMS OF DIMETHOXY DOCETAXEL AND METHODS FOR PREPARING THE SAME
Abstract
The invention relates to anhydrides, solvates and ethanol hetero-solvates and hydrates of dimethoxy docetaxel or (2R,3S)-3-tert-butoxycarbonylamino-2-hydroxy-3-phenyl-propionate of 4-acetoxy-2.alpha.-benzoyloxy-5.beta.,20-epoxy-1-hydroxy-7.beta., 10.beta.-dimethoxy-9-oxo-tax-11-ene-13.alpha.-yle, and to the preparation thereof.
| Inventors: | BILLOT; Pascal; (Montreuil, FR) ; DUFRAIGNE; Marielle; (Brunoy, FR) ; ELMALEH; Hagit; (Ormesson sur Marne, FR) ; MANGIN; Fabrice; (Paris, FR) ; RORTAIS; Patricia; (Bourg La Reine, FR) ; ZASKE; Lionel; (Saint Maurice, FR) ; GIULIANI; Alexandre; (Boissy Saint Leger, FR) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 39712334 | ||||||||||
| Appl. No.: | 16/188687 | ||||||||||
| Filed: | November 13, 2018 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15646375 | Jul 11, 2017 | 10160739 | ||
| 16188687 | ||||
| 14453087 | Aug 6, 2014 | |||
| 15646375 | ||||
| 13767966 | Feb 15, 2013 | 8846959 | ||
| 14453087 | ||||
| 12837559 | Jul 16, 2010 | 8378128 | ||
| 13767966 | ||||
| PCT/FR2009/000042 | Jan 15, 2009 | |||
| 12837559 | ||||
| Current U.S. Class: | 549/510 ; 549/457 |
| Current CPC Class: | A61P 35/00 20180101; C07D 305/14 20130101; A61P 35/02 20180101 |
| International Class: | C07D 305/14 20060101 C07D305/14 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Jan 17, 2008 | FR | 0800243 |
Claims
1. Crystalline forms of 4-acetoxy-2a-benzoyloxy-5b,20-epoxy-1-hydroxy-7.beta.,10.beta.-dimethoxy-- 9-oxotax-11-en-13a-yl (2R,3S)-3-tert-butoxycarbonylamino-2-hydroxy-3-phenylpropionate, with the exception of the acetonate form.
2. Forms according to claim 1, characterized in that they are anhydrous forms of 4-acetoxy-2a-benzoyloxy-5b,20-epoxy-1-hydroxy-7.beta.,10.beta.-d- imethoxy-9-oxotax-11-en-13a-yl (2R,3S)-3-tert-butoxycarbonylamino-2-hydroxy-3-phenylpropionate.
3. (canceled)
4. Anhydrous form C of 4-acetoxy-2.alpha.-benzoyloxy-5b,20-epoxy-1-hydroxy-7.beta.,10.beta.-dime- thoxy-9-oxotax-11-en-13a-yl (2R,3S)-3-tert-butoxycarbonylamino-2-hydroxy-3-phenylpropionate according to claim 2, characterized by a PXRD diagram exhibiting characteristic lines located at 4.3, 6.8, 7.4, 8.7, 10.1, 11.1, 11.9, 12.3, 12.6 and 13.1.+-.0.2 degrees 2-theta.
5. (canceled)
6. Anhydrous form E of 4-acetoxy-2a-benzoyloxy-5b,20-epoxy-1-hydroxy-7.beta.,10.beta.-dimethoxy-- 9-oxotax-11-en-13a-yl (2R,3S)-3-tert-butoxycarbonylamino-2-hydroxy-3-phenylpropionate according to claim 2, characterized by a PXRD diagram exhibiting characteristic lines located at 7.1, 8.1, 8.9, 10.2, 10.8, 12.5, 12.7, 13.2, 13.4 and 13.9.+-.0.2 degrees 2-theta.
7. (canceled)
8. Forms according to claim 1, characterized in that they are ethanolic solvate or heterosolvate forms of 4-acetoxy-2a-benzoyloxy-5b,20-epoxy-1-hydroxy-7.beta.,10.beta.-dimethoxy-- 9-oxotax-11-en-13a-yl (2R,3S)-3-tert-butoxycarbonylamino-2-hydroxy-3-phenylpropionate.
9. Ethanolate form B of 4-acetoxy-2a-benzoyloxy-5b,20-epoxy-1-hydroxy-7.beta.,10.beta.-dimethoxy-- 9-oxotax-11-en-13a-yl (2R,3S)-3-tert-butoxycarbonylamino-2-hydroxy-3-phenylpropionate according to claim 8, characterized by a PXRD diagram exhibiting characteristic lines located at 7.3, 7.8, 8.8, 10.2, 12.6, 12.9, 13.4, 14.2, 14.7 and 15.1.+-.0.2 degrees 2-theta.
10. Ethanolate form D of 4-acetoxy-2a-benzoyloxy-5b,20-epoxy-1-hydroxy-7.beta.,10.beta.-dimethoxy-- 9-oxotax-11-en-13a-yl (2R,3S)-3-tert-butoxycarbonylamino-2-hydroxy-3-phenylpropionate according to claim 8, characterized by a PXRD diagram exhibiting characteristic lines located at 3.8, 7.5, 7.7, 8.4, 9.4, 10.3, 10.5, 11.1, 11.5 and 11.9.+-.0.2 degrees 2-theta.
11. (canceled)
12. Ethanol/water heterosolvate form F of 4-acetoxy-2a-benzoyloxy-5b,20-epoxy-1-hydroxy-7.beta.,10.beta.-dimethoxy-- 9-oxotax-11-en-13a-yl (2R,3S)-3-tert-butoxy-carbonylamino-2-hydroxy-3-phenylpropionate according to claim 8, characterized by a PXRD diagram exhibiting characteristic lines located at 4.4, 7.2, 8.2, 8.3, 8.8, 9.6, 10.3, 10.9, 11.2 and 12.2.+-.0.2 degrees 2-theta.
13. Forms according to claim 1, characterized in that they are hydrate forms of 4-acetoxy-2a-benzoyloxy-5b,20-epoxy-1-hydroxy-7p,10.beta.-dimeth- oxy-9-oxotax-11-en-13a-yl (2R,3S)-3-tert-butoxycarbonylamino-2-hydroxy-3-phenylpropionate.
14. Monohydrate form C of 4-acetoxy-2a-benzoyloxy-5b,20-epoxy-1-hydroxy-7.beta.,10.beta.-dimethoxy-- 9-oxotax-11-en-13a-yl (2R,3S)-3-tert-butoxycarbonylamino-2-hydroxy-3-phenylpropionate according to claim 13, characterized by a PXRD diagram exhibiting characteristic lines located at 4.3, 6.8, 7.4, 8.6, 10.1, 11.1, 11.9, 12.2, 12.6 and 13.3.+-.0.2 degrees 2-theta.
15. Dihydrate form C of 4-acetoxy-2a-benzoyloxy-5b,20-epoxy-1-hydroxy-7.beta.,10.beta.-dimethoxy-- 9-oxotax-11-en-13a-yl (2R,3S)-3-tert-butoxycarbonylamino-2-hydroxy-3-phenylpropionate according to claim 13, characterized by a PXRD diagram exhibiting characteristic lines located at 4.2, 6.9, 7.5, 8.4, 9.9, 10.9, 11.7, 12.3, 12.6 and 13.2.+-.0.2 degrees 2-theta.
16. Method for preparing the anhydrous form B according to claim 3, which consists in heating the acetone solvate form A between 100 and 110.degree. C. under vacuum or nitrogen sweeping, preferably for at least 9 hours, and then returning to ambient temperature.
17. Method for preparing the anhydrous form C according to claim 4, by maturation of the acetone solvate form A, or of the anhydrous form B, in water, followed by drying up to approximately 50.degree. C. and then maintaining at ambient temperature at a relative humidity of less than 5%.
18. Method for preparing the anhydrous form D according to claim 5, by maturation, at ambient temperature, of the acetone solvate form A in ethanol and drying under nitrogen or under vacuum.
19. Method for preparing the anhydrous form D according to claim 5, by crystallization, at ambient temperature, of the acetone solvate form A from an oil, followed by rinsing with an alkane.
20. Method for preparing the anhydrous form D according to claim 5, by crystallization of a solution of 4-acetoxy-2a-benzoyloxy-5b,20-epoxy-1-hydroxy-7.beta.,10.beta.-dimethoxy-- 9-oxotax-11-en-13a-yl (2R,3S)-3-tert-butoxycarbonylamino-2-hydroxy-3-phenylpropionate in a mixture of Polysorbate 80, pH 3.5, ethanol and water (preferably, a 25/25/50 mixture) for approximately 48 hours at ambient temperature.
21. Method for preparing the anhydrous form E according to claim 6, by maturation of the acetone solvate form A in ethanol so as to intermediately form an ethanolic form which is subsequently desolvated under nitrogen sweeping or by heating at approximately 100.degree. C. for 2 hours and then returning to ambient temperature.
22. Method for preparing the anhydrous form F according to claim 7, by desolvating the ethanol/water heterosolvate at 120.degree. C. under a nitrogen atmosphere for 24 hours and then maintaining at a relative humidity of 0% at ambient temperature.
Description
[0001] This application is a divisional of U.S. patent application Ser. No. 15/646,375, filed Jul. 11, 2017, which is a divisional application of U.S. patent application Ser. No. 14/453,087, filed Aug. 6, 2014, which is a divisional application of U.S. patent application Ser. No. 13/767,966, filed Feb. 15, 2013, now U.S. Pat. No. 8,846,959, which is a divisional application of U.S. patent application Ser. No. 12/837,559 filed Jul. 16, 2010, now U.S. Pat. No. 8,378,128, which is a continuation of International Patent Application PCT/FR2009/000042 filed Jan. 15, 2009, all of which are incorporated herein by reference; and which claim priority to French Patent Application No. 0800243 filed on Jan. 17, 2008.
[0002] The present invention relates to crystalline forms of dimethoxy docetaxel or 4-acetoxy-2.alpha.-benzoyloxy-5.beta.,20-epoxy-1-hydroxy-7.beta., 10.beta.-dimethoxy-9-oxotax-11-en-13.alpha.-yl (2R,3S)-3-tert-butoxycarbonylamino-2-hydroxy-3-phenylpropionate and to methods for the preparation thereof.
BACKGROUND OF THE INVENTION
[0003] 4-Acetoxy-2.alpha.-benzoyloxy-5.beta., 20-epoxy-1-hydroxy-7.beta., 10.beta.-dimethoxy-9-oxotax-11-en-13.alpha.-yl (2R,3S)-3-tert-butoxycarbonylamino-2-hydroxy-3-phenylpropionate exhibits notable anticancer and antileukaemic properties.
[0004] 4-Acetoxy-2.alpha.-benzoyloxy-5.beta., 20-epoxy-1-hydroxy-7.beta., 10.beta.-di methoxy-9-oxotax-11-en-13.alpha.-yl (2R,3S)-3-tert-butoxycarbonylamino-2-hydroxy-3-phenylpropionate is prepared according to the method which is described more particularly in PCT International Application WO 96/30355 or PCT International Application WO 99/25704. According to the method described in these applications, the product is not crystallized and is not characterized.
[0005] It was found that the acetone solvate of 4-acetoxy-2.alpha.-benzoyloxy-5.beta.,20-epoxy-1-hydroxy-7.beta., 10.beta.-dimethoxy-9-oxotax-11-en-13.alpha.-yl (2R,3S)-3-tert-butoxycarbonylamino-2-hydroxy-3-phenylpropionate (called form A) was completely determined and characterized according to the patent published under number WO2005/028462.
BRIEF SUMMARY OF THE INVENTION
[0006] The present invention relates to new crystalline forms, with the exclusion of the acetonate form, the only one known to date.
[0007] According to the present invention, it has now been found that certain anhydrous forms, certain ethanolic solvates or heterosolvates and hydrated forms have been completely characterized from a physical and chemical structure point of view.
BRIEF DESCRIPTION OF THE DRAWINGS
[0008] FIG. 1 depicts a Differential Scanning calorimetry (DSC) analysis for anhydrous form D.
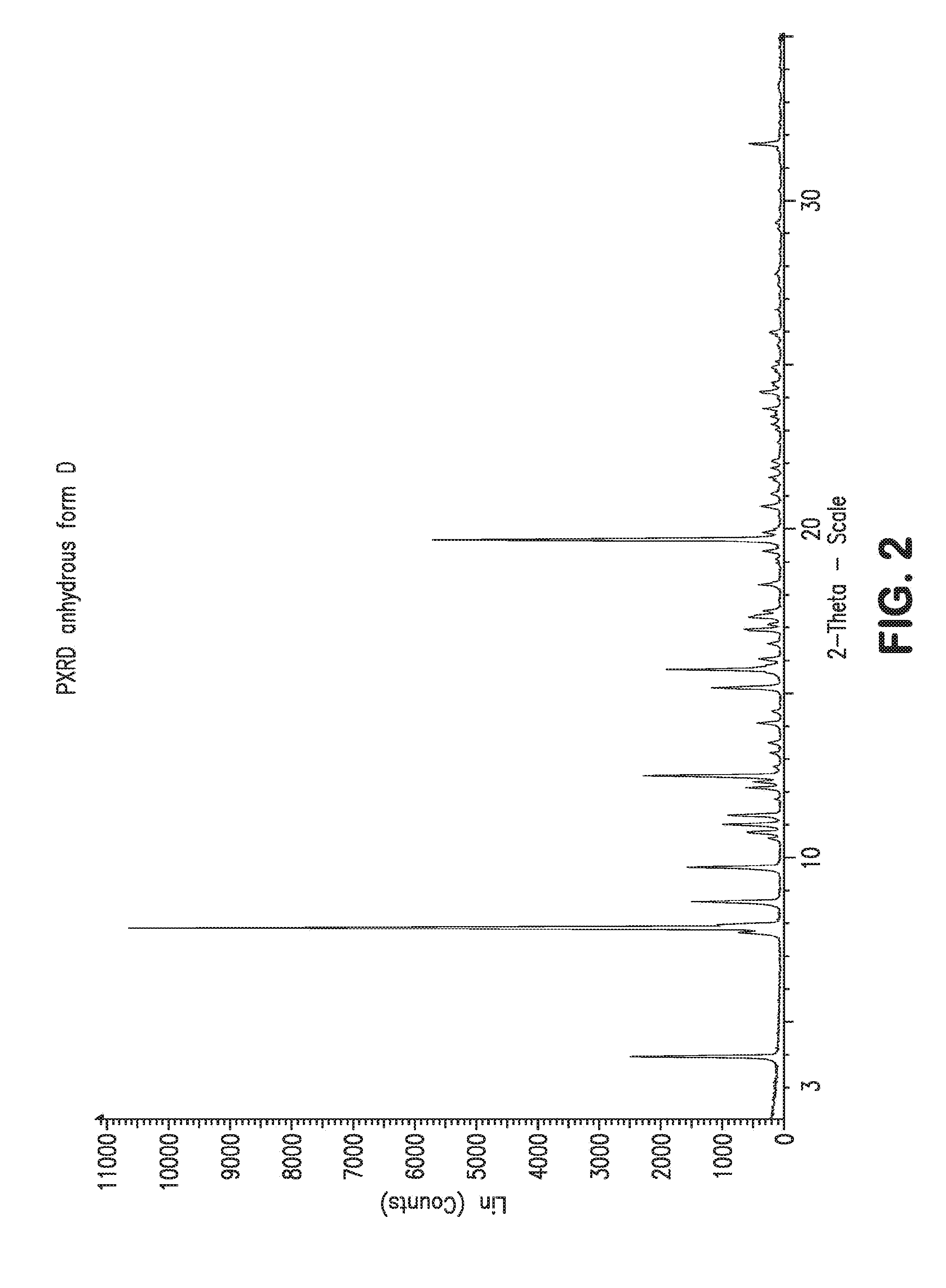
[0009] FIG. 2 depicts a Powder X-Ray Diffraction (PXRD) analysis for anhydrous form D.
[0010] FIG. 3 depicts Fourier Transform InfraRed (FTIR) spectrometry analysis for anhydrous form D.
[0011] FIG. 4 depicts a Powder X-Ray Diffraction (PXRD) analysis for ethanolate form D.
[0012] FIG. 5 depicts a Powder X-Ray Diffraction (PXRD) analysis for ethanolate form E.
DETAILED DESCRIPTION OF THE INVENTION
[0013] According to the invention, among the anhydrous forms of 4-acetoxy-2.alpha.-benzoyloxy-5.beta.,20-epoxy-1-hydroxy-7.beta.,10.beta.- -dimethoxy-9-oxotax-11-en-13.alpha.-yl(2R,3S)-3-tert-butoxycarbonylamino-2- -hydroxy-3-phenylpropionate, five different forms have been identified, among the ethanolic solvates or heterosolvates of 4-acetoxy-2.alpha.-benzoyloxy-5.beta.,20-epoxy-1-hydroxy-7.beta.,10.beta.- -dimethoxy-9-oxotax-11-en-13.alpha.-yl(2R,3S)-3-tert-butoxy c arb onyl amino-2-hydroxy-3-phenylpropionate, four different forms have been identified and among the hydrates of 4-acetoxy-2.alpha.-benzoyloxy-5.beta.,20-epoxy-1-hydroxy-7.beta.,10.beta.- -dimethoxy-9-oxotax-11-en-13.alpha.-yl (2R,3S)-3-tert-butoxycarbonylamino-2-hydroxy-3-phenylpropionate, two different forms have been identified.
[0014] The five anhydrous forms identified were obtained according to the following methods: [0015] The anhydrous form B by a method which consists in heating the acetone form or form A obtained according to the patent mentioned above, between 100 and 110.degree. C. under vacuum or nitrogen sweeping. This treatment is preferably carried out for at least 9 hours before a return to ambient temperature without inducing chemical decomposition. Its melting point by DSC is approximately 150.degree. C. The PXRD diagram of the anhydrous form B exhibits characteristic lines located at 7.3, 8.1, 9.8, 10.4, 11.1, 12.7, 13.1, 14.3, 15.4 and 15.9.+-.0.2 degrees 2-theta. [0016] The anhydrous form C is obtained by maturation of the acetone solvate form A, or of the anhydrous form B, in water followed by drying at up to 50.degree. C. and maintaining between 0 and 5% RH at ambient temperature. Its melting point by DSC is approximately 146.degree. C. The PXRD diagram of the anhydrous form C exhibits characteristic lines located at 4.3, 6.8, 7.4, 8.7, 10.1, 11.1, 11.9, 12.3, 12.6 and 13.1.+-.0.2 degrees 2-theta. It is, among the various anhydrous forms, the least stable of all the forms described in the present invention. In the presence of a relative humidity of greater than 5%, it changes to a hydrated form. [0017] The anhydrous form D is obtained according to a first method by crystallization of the form A in an oil (especially Miglyol), following by rinsing with an alkane, for example heptane; the second preparation method consists in leaving a solution of 4-acetoxy-2.alpha.-benzoyloxy-5.beta.,20-epoxy-1-hydroxy-7.beta.,10.beta.- -dimethoxy-9-oxotax-11-en-13.alpha.-yl (2R,3S)-3-tert-butoxycarbonylamino-2-hydroxy-3-phenylpropionate in a mixture of Polysorbate 80, pH 3.5, ethanol and water (preferably a 25/25/50 mixture) to crystallize for approximately 48 hours. Its boiling point by DSC is approximately 175.degree. C. (cf. FIG. 1) and is found to be the highest of all the anhydrous forms isolated. The PXRD diagram of the anhydrous form D (cf. FIG. 2) exhibits characteristic lines located at 3.9, 7.7, 7.8, 7.9, 8.6, 9.7, 10.6, 10.8, 11.1 and 12.3.+-.0.2 degrees 2-theta. The FTIR spectrum of the anhydrous form D exhibits characteristic bands located at 979, 1072, 1096, 1249, 1488, 1716, 1747, 3436.+-.1 cm.sup.-1 (cf. FIG. 3). Among all the forms described in the present invention, it is the most stable anhydrous form. [0018] The anhydrous form E is obtained at ambient temperature by maturation of the acetone form or form A in ethanol so as to intermediately form an ethanolic form which is subsequently desolvated under nitrogen sweeping or by heating at approximately 100.degree. C. for 2 hours. Its melting point by DSC is approximately 157.degree. C. The PXRD diagram of the anhydrous form E exhibits characteristic lines located at 7.1, 8.1, 8.9, 10.2, 10.8, 12.5, 12.7, 13.2, 13.4 and 13.9.+-.0.2 degrees 2-theta. [0019] The anhydrous form F is obtained by desolvating the ethanol/water heterosolvate at 120.degree. C. under a nitrogen atmosphere for 24 hours and then maintaining in a dry environment at 0% RH at ambient temperature. Its melting point by DSC is approximately 148.degree. C. The PXRD diagram of the anhydrous form F exhibits characteristic lines located at 4.4, 7.2, 8.2, 8.8, 9.6, 10.2, 10.9, 11.2, 12.1 and 12.3.+-.0.2 degrees 2-theta.
[0020] There are four crystalline forms identified in ethanolic solvate or heterosolvate form: [0021] The ethanolate form B is obtained at ambient temperature by maintaining the anhydrous form B in an ethanol-vapour-saturated environment. The PXRD diagram of the ethanolate form B exhibits characteristic lines located at 7.3, 7.8, 8.8, 10.2, 12.6, 12.9, 13.4, 14.2, 14.7 and 15.1.+-.0.2 degrees 2-theta. [0022] The ethanolate form D is obtained at ambient temperature by maintaining the anhydrous form D in an ethanol-vapour-saturated environment. The PXRD diagram of the ethanolate form D (cf. FIG. 4) exhibits characteristic lines located at 3.8, 7.5, 7.7, 8.4, 9.4, 10.3, 10.5, 11.1, 11.5 and 11.9.+-.0.2 degrees 2-theta. [0023] The ethanolate form E is obtained at ambient temperature by maturation of the acetonate form A in ethanol. The PXRD diagram of the ethanolate form E (cf. FIG. 5) exhibits characteristic lines located at 7.1, 8.1, 8.8, 10.2, 10.7, 12.5, 13.2, 13.4, 13.9 and 14.2.+-.0.2 degrees 2-theta. [0024] The ethanol/water heterosolvate form F is obtained by maintaining the form B in a minimum amount of ethanol at reflux, slow cooling and isolation at ambient temperature and ambient relative humidity. The PXRD diagram of the ethanol/water heterosolvate form F exhibits characteristic lines located at 4.4, 7.2, 8.2, 8.3, 8.8, 9.6, 10.3, 10.9, 11.2 and 12.2.+-.0.2 degrees 2-theta.
[0025] There are two crystalline forms identified in hydrate form: [0026] The monohydrated forms C are obtained at ambient temperature by maintaining the anhydrous form C in an atmosphere containing at least 10% relative humidity. The PXRD diagram of the monohydrate form C exhibits characteristic lines located at 4.3, 6.8, 7.4, 8.6, 10.1, 11.1, 11.9, 12.2, 12.6 and 13.3.+-.0.2 degrees 2-theta. [0027] The dihydrate form C is obtained at ambient temperature by maintaining the anhydrous form C in an atmosphere containing at least 60% relative humidity. The PXRD diagram of the dihydrate form C exhibits characteristic lines located at 4.2, 6.9, 7.5, 8.4, 9.9, 10.9, 11.7, 12.3, 12.6 and 13.2.+-.0.2 degrees 2-theta.
[0028] Other, nonethanolic, solvates of the form B were prepared, such as in particular those obtained with the following solvents: dichloromethane, diisopropyl ether, n-propanol, isopropanol, toluene, methyl isobutyl ketone, tetrahydrofuran, dimethylformamide, ethyl acetate, etc.
[0029] The present invention will be described more fully by means of the following examples which should not be considered to limit the invention.
[0030] Experimental Analysis Conditions:
[0031] Differential Scanning Calorimetry (DSC):
[0032] The measurements were carried out on a T.A. Instruments DSC2010 thermal analyser. The sample is subjected to temperature programming from 25.degree. C. to 225.degree. C. with a heating rate of 5.degree. C./min. The product is placed in a crimped aluminium capsule and the amount of product analysed is between 2 and 5 mg. Constant nitrogen sweeping at 55 mL/min is used in the oven chamber.
[0033] Powder X-Ray Diffraction (PXRD):
[0034] The analyses were carried out on a Panalytical X'Pert Pro diffractometer with a reflection-mode Bragg-Brentano focusing geometry (.theta.-2.theta.) assembly. The product analysed is deposited as a thin layer on a silicon single crystal. A copper anticathode tube (45 kV/40 mA) supplies an incident radiation Cu K.alpha..sub.1 (.lamda.=1.5406 .ANG.). The beam is collimated using Sollers slits which improve the parallelism and variable slits which limit scattering. An X'Celerator detector completes the device. The diagram recording characteristics are the following: sweeping from 2 to 30 degrees 2.theta., counting time from 100 to 500 seconds per step with a step of 0.017.degree..
[0035] Fourier Transform InfraRed (FTIR) Spectrometry:
[0036] The solid samples were analysed using a Nicolet Nexus spectrometer. The analysis is carried out by attenuated total reflectance (ATR) using a Smart Orbit accessory from the company Thermo (single reflection diamond crystal ATR accessory). The spectral range swept is between 4000 and 400 cm.sup.-1 with a resolution of 2 cm.sup.-1 and an accumulated scan number of 20.
Example 1
[0037] Two tests of dissolution of approximately 550 mg of 4-acetoxy-2.alpha.-benzoyloxy-5.beta.,20-epoxy-1-hydroxy-7.beta.,10.beta.- -dimethoxy-9-oxotax-11-en-13.alpha.-yl (2R,3S)-3-tert-butoxycarbonylamino-2-hydroxy-3-phenylpropionate in 14 g of Miglyol 812 Neutral oil, Sasol are carried out. Magnetic stirring is carried out at 500 rpm for 24 hours at ambient temperature.
[0038] After one week, the samples are vacuum-filtered and rinsed with heptane. Each sample is analysed by PXRD for confirmation of the form obtained. After filtration, between 300 and 350 mg of anhydrous form D are obtained.
Example 2
[0039] Approximately 3 g of 4-acetoxy-2.alpha.-benzoyloxy-5.beta.,20-epoxy-1-hydroxy-70,100-dimethoxy- -9-oxotax-11-en-13.alpha.-yl (2R,3S)-3-tert-butoxycarbonylamino-2-hydroxy-3-phenyl-propionate are dissolved in a mixture of 50 mL ethanol+50 mL Polysorbate 80, pH 3.5. 100 mL of water are added to the previous mixture and the whole is homogenized. After storage for 48 hours at ambient temperature, crystals of anhydrous form D appeared. The amount of crystallized product recovered by filtration is approximately 2.45 g.
[0040] A comparative stability study was carried out between the acetone solvate form A and the anhydrous form D. The comparison of the PXRD analyses carried out on the A and D forms immediately after production and after having maintained said forms at 40.degree. C. for one month gives the following results: [0041] Form A: partial desolvation resulting in a mixture of the acetone solvate form A and of the anhydrous form B being obtained. [0042] Form D: no change detected after maintaining at 40.degree. C. for one month.
* * * * *
D00001

D00002

D00003

D00004

D00005

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.