Method of Restoring the Incretin Effect
Clemens; Anton H.
U.S. patent application number 15/997965 was filed with the patent office on 2019-05-09 for method of restoring the incretin effect. The applicant listed for this patent is Neurendo Pharma LLC. Invention is credited to Anton H. Clemens.
| Application Number | 20190134023 15/997965 |
| Document ID | / |
| Family ID | 39325299 |
| Filed Date | 2019-05-09 |


| United States Patent Application | 20190134023 |
| Kind Code | A1 |
| Clemens; Anton H. | May 9, 2019 |
Method of Restoring the Incretin Effect
Abstract
The present invention relates to methods of treating metabolic syndrome, Type 2 diabetes mellitus, atherogenic dyslipidemia and/or obesity. The present invention a so relates to methods of restoring the incretin effect, to restoring physiologic control of glucagon levels, to restoring first-phase insulin secretion, and to restoring the physiologic glucose-dependent insulin secretion. The methods of the present invention comprise administration of a selective .kappa.-receptor antagonist, such as guanidinylated naltrindole (GNTI), or pharmaceutically acceptable derivatives thereof to a subject in need thereof.
| Inventors: | Clemens; Anton H.; (Madison, WI) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 39325299 | ||||||||||
| Appl. No.: | 15/997965 | ||||||||||
| Filed: | June 5, 2018 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 14479828 | Sep 8, 2014 | 9987268 | ||
| 15997965 | ||||
| 13859940 | Apr 10, 2013 | 8829018 | ||
| 14479828 | ||||
| 12906735 | Oct 18, 2010 | 8445508 | ||
| 13859940 | ||||
| 11876279 | Oct 22, 2007 | 7893080 | ||
| 12906735 | ||||
| 60862227 | Oct 20, 2006 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61P 3/10 20180101; A61K 38/45 20130101; A61P 3/00 20180101; A61K 31/4748 20130101; A61P 3/04 20180101; A61K 31/485 20130101; A61K 31/437 20130101; Y10S 514/866 20130101; A61K 45/06 20130101; A61P 3/08 20180101 |
| International Class: | A61K 31/485 20060101 A61K031/485; A61K 45/06 20060101 A61K045/06; A61K 38/45 20060101 A61K038/45; A61K 31/4748 20060101 A61K031/4748; A61K 31/437 20060101 A61K031/437 |
Claims
1. A method of treating type 2 diabetes mellitus comprising administering to a subject a therapeutically effective amount of a selective .kappa.-receptor antagonist, or a pharmaceutically acceptable derivative thereof.
2. The method of claim 1, wherein the selective .kappa.-receptor antagonist is GNTI.
3. The method of claim 1, wherein the selective .kappa.-receptor antagonist is administered weekly or daily.
4. The method of claim 3, wherein the selective .kappa.-receptor antagonist is administered weekly in an amount from about 30 ng to about 300 ng per kg of body weight weekly.
5. The method of claim 3, wherein the selective .kappa.-receptor antagonist is administered daily in an amount from about 8 ng to about 80 ng per kg of body weight daily.
6. The method of claim 1, wherein the selective .kappa.-receptor antagonist is administered sublingually, orally, enterally, parenterally, topically, systemically or injected intravascularly, subcutaneously, peritoneally.
7. The method of claim 1, further comprising co-administration of an effective amount of an insulinogenic agent.
8. The method of claim 7, wherein the insulinogenic agent is an extended release composition.
9. The method of claim 1, wherein a .mu.-agonist is not co-administered.
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application is a divisional patent application of U.S. patent application Ser. No. 14/479,828 filed Sep. 8, 2014, which is a continuation of U.S. patent application Ser. No. 13/859,940 filed Apr. 10, 2013 and issued as U.S. Pat. No. 8,829,018, which is a continuation patent application of U.S. patent application Ser. No. 12/906,735 filed Oct. 18, 2010 and issued as U.S. Pat. No. 8,445,508 on May 21, 2013, which is a divisional patent application of application Ser. No. 11/876,279 filed Oct. 22, 2007 and issued as U.S. Pat. No. 7,893,080 on Feb. 22, 2011, which is a non-provisional application of U.S. Provisional Application No. 60/862,227 filed Oct. 22, 2006. The Ser. No. 11/876,279 application claims priority to U.S. Provisional Application No. 60/862,227 filed Oct. 22, 2006. The above-referenced applications are incorporated herein in their entireties.
STATEMENT REGARDING FEDERALLY SPONSORED RESEARCH
[0002] Not applicable.
INTRODUCTION
[0003] Insulin secretion is stimulated to greater extent by oral intake of glucose, than by intravenous intake of glucose. This effect, which is called the incretin effect, is estimated to be responsible for more than half of the insulin response to glucose. The incretin effect is caused mainly by the two intestinal insulin-stimulating hormones, glucagon-like peptide-1 (GLP)-1) and glucose-dependent insulinotropic polypeptide (GIP). In patients with Type 2 diabetes mellitus, and other components of the metabolic syndrome, such as impaired glucose tolerance, atherogenic dyslipidemia, overweight and obesity, the incretin effect is either greatly impaired or absent.
[0004] The rapid and sizable increase in insulin release initiated by the incretin effect begins within two minutes of nutrient ingestion and continues for up to 15 minutes. This post-meal increase in insulin is referred to as first phase insulin secretion. A second phase of insulin secretion follows and is sustained until normal blood glucose levels are restored. Clinical observations reveal that patients with metabolic syndrome disorders such as Type 2 diabetes mellitus, impaired glucose tolerance, and obesity are characterized by progressive reductions in the magnitude of first-phase, insulin secretion, insulin resistance, and beta-cell dysfunction, creating a new pathogenic platform shared by all components of the metabolic syndrome. Beta-cell dysfunction is, in turn, characterized by its two contributing components: (1) the progressive impairment of insulin production, and (2) the progressive impairment of physiologic control of insulin release. It follows, that the metabolic disorders should be correctable by the treatment, or restoration, of the failing components of the underlying pathogenic platform of insulin resistance and beta-cell dysfunction.
[0005] Glucose intolerance includes a continuous range of impairments in carbohydrate metabolism. Type 2 diabetes mellitus, impaired glucose tolerance, and impaired fasting glucose, among other conditions associated with glucose intolerance, have been implicated as risk factors contributing to heart disease, stroke, overweight, obesity, hypertension, and atherogentic dyslipidemia.
[0006] The pathogenesis of obesity is associated with other components of the metabolic syndrome, like atherogenic dyslipidemia, and glucose intolerance, the magnitude of which may progress over time. Threshold levels for pharmacological treatment have, therefore, been revised downward on several occasions in order to intervene at an earlier stage in the epidemic expansion of the affected population, and the corresponding health care expenditures. Non-insulin therapies are available to reduce endogenous gluconeogenesis or improve peripheral insulin sensitivity, e.g., metformin, sulfonylureas or thiazolidinediones (TZD). However, these therapies fail to restore first-phase insulin release or the incretin effect. Importantly, enhanced early insulin release is associated with improved overall glucose tolerance. There is, currently, no treatment available to restore or reactivate the natural physiology of the native incretin system.
SUMMARY
[0007] The present invention relates to methods of treating metabolic syndrome, Type 2 diabetes atherogenic dyslipidemia and/or obesity. The present invention also relates to methods of restoring the incretin effect, to restoring physiologic control of glucagon levels, to restoring first-phase insulin secretion, and to restoring the physiologic glucose-dependent secretion.
[0008] The methods of the present invention comprise administration of a selective .kappa.-receptor antagonist such as guanidinylated naltrindole (GNTI), or a pharmaceutically acceptable derivative thereof, to a subject in need thereof. The selective .kappa.-receptor antagonist may be administered daily, weekly or at any suitable time interval. Administration may be sublingually, orally, enterally, parenterally, topically or systemically. The selective .kappa.-receptor antagonist may be co-administered with an insulinotropic agent. The insulinotropic agent may be an extended release formulation.
BRIEF DESCRIPTION OF FIGURES
[0009] FIG. 1 illustrates the blood glucose readings of a male subject treated with GNTI over an extended period of time as described in Example 1.
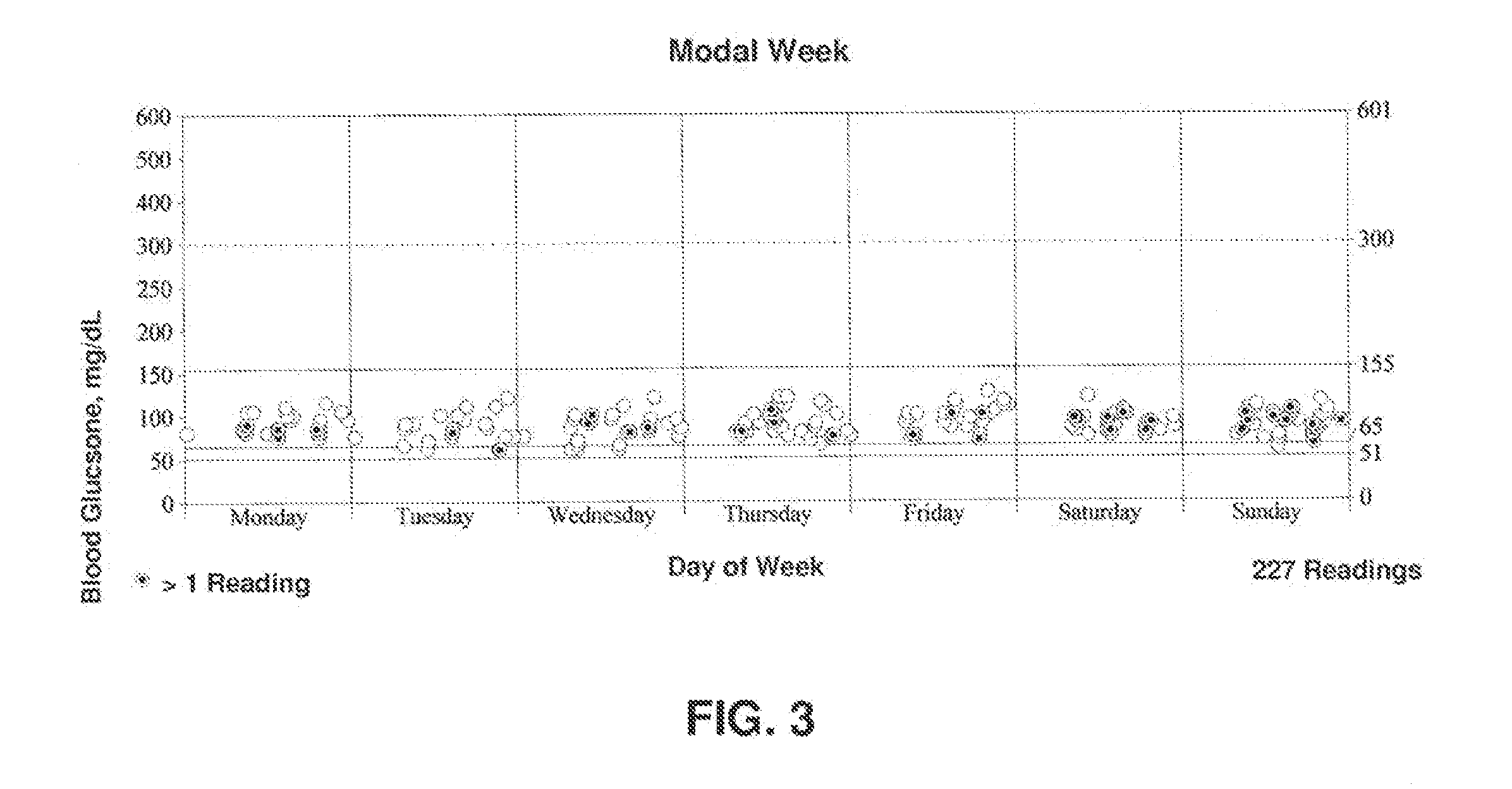
[0010] FIG. 2 illustrates the blood glucose readings of a male subject treated with GNTI over a modal week as described in Example 1.
[0011] FIG. 3 illustrates the blood glucose readings of a male subject treated with GNTI over a modal day as described in Example 1.
DETAILED DESCRIPTION
[0012] It has surprisingly been discovered that the administration of a selective .kappa.-receptor antagonist, such as GNTI, is useful in the treatment of metabolic syndrome by targeting the incretin effect. The term "selective .kappa.-receptor antagonist" means a .kappa.-receptor antagonist characterized by its .mu./.kappa. antagonism K.sub.c selectivity ratio, as defined in J. Med. Chem 43, 2759-2769 (2000), the ratio required for this method being <35. Suitably, the selective .kappa.-receptor antagonist acts peripherally. That is, it is substantially free of CNS activity.
[0013] The term "metabolic syndrome" may include, but is not limited to, atherogenic dyslipidemia, pre-diabetes, overweight/obesity, Type 2 diabetes mellitus and essential hypertension. The pathogenesis of obesity is associated with other components of the metabolic syndrome, e.g., atherogenic dyslipidemia, and glucose intolerance, the magnitude of which may progress, from its initial stages characterized by impaired fasting glucose, followed by impaired glucose tolerance and culminating in Type 2 diabetes mellitus. Administration of a selective .kappa.-receptor antagonist, or a pharmaceutically acceptable derivative thereof has been found to restore the incretin effect, restore physiological control of glucagon levels in response to ingested nutrition, restore first-phase insulin secretion, restore glucose-dependent insulin secretion, reduce weight gain and/or lower weight in a subject without co-administration of a .mu.-agonist.
[0014] As will be appreciated, the methods described herein may be useful in both research and clinical settings, suitably wherein treatment of certain disease states are implicated, including, but not limited to, impaired glucose tolerance, Type 2 diabetes mellitus, diminished or absent first-phase insulin secretion, and obesity.
[0015] The administration of a selective .kappa.-receptor antagonist to a subject in need thereof may treat metabolic syndrome. Administration of a selective .kappa.-receptor antagonist may treat metabolic syndrome by restoring the incretin effect, by restoring physiologic control of glucagon levels, by restoring the physiologic glucose dependent insulin secretion, and/or by restoring first-phase insulin secretion. Suitably, the administration of a selective .kappa.-receptor antagonist may also treat overweight, atherogenic dyslipidemia obesity or Type 2 diabetes mellitus by restoring the incretin effect, by restoring physiologic control of glucagon levels, by restoring the physiologic glucose dependent insulin secretion, and/or by restoring first-phase insulin secretion.
[0016] The administration of a selective .kappa.-receptor antagonist to a subject in need thereof restores the incretin effect. In a subject having a normal response to oral nutrient administration, the release of the insulinotropic hormones, GIP and GLP-1, results in an increase in insulin secretion. This is called the "incretin effect". As used herein, to "restore," for example, with respect to the incretin effect, suitably includes enhancing, potentiating, increasing, reestablishing, re-activating, or improving the physiological state. For example, a subject having Type 2 diabetes mellitus may exhibit diminished or even zero incretin effect, i.e., diminished or no activity of GIP or GLP-1, or diminished or no increase in insulin secretion upon nutrient administration. Consequently, to "restore" the incretin effect suitably increases, though does not necessarily normalize, GIP or GLP-1 activity or insulin secretion upon nutrient administration in a subject. The subject is suitably a mammal, such as a human, dog, cat, primate, etc.
[0017] The administration of a selective .kappa.-receptor antagonist may restore physiologic control of glucagon levels in a subject in need thereof. As used herein, to "restore," for example with respect to physiologic control of glucagon levels, suitably includes, decreasing, lowering, regulating, reestablishing, or improving the physiologic state. In a subject having a `normal` physiologic response to nutrient administration, physiologic control of glucagon primarily responds to blood glucose levels, i.e., as blood glucose levels decline, glucagon is released from the a cells of the islets of Langerhans in the pancreas, and act on the liver to induce gluconeogenesis, i.e., endogenous glucose production, and/or glycogenolysis. Conversely, glucagon release decreases in response to increasing blood glucose levels. Additionally, glucagon levels decrease in response to release of insulin by pancreatic .beta.-cells. Consequently, in a subject having abnormal insulin production or release in response to increasing blood glucose levels, glucagon release may remain abnormally high and result hyperglucagonemia which further exacerbates conditions such as Type 2 diabetes mellitus and impaired glucose tolerance.
[0018] Normal insulin secretion from the pancreatic .beta. cells is biphasic. The initial release of insulin that acts on the pancreatic .alpha. cells to decrease glucagon is referred to as the first-phase of insulin secretion. First-phase insulin secretion is characterized by a rapid and sizable increase in insulin, beginning within two minutes of nutrient ingestion, and continuing for 10-15 minutes. For example, in Examples 3 and 4, first-phase insulin secretion can be seen at the 5-minute time point coupled with a corresponding drop in glucagon levels. Two minutes later, blood glucose levels show an initial decrease. The second phase of insulin secretion follows and insulin secretion peaks approximately 1-2 hours following nutrient ingestion. Insulin secretion continues until normal blood glucose levels are restored. Often, in subjects having impaired glucose tolerance, first-phase insulin secretion is reduced and it is believed that the reduction in first-phase insulin secretion may be a preliminary sign in the progression of Type 2 diabetes mellitus.
[0019] The administration of a selective .kappa.-receptor antagonist restores first-phase insulin secretion in a subject in need thereof. The administration of a selective .kappa.-receptor antagonist may restore the physiologic glucose dependent insulin secretion in a subject in need thereof, and a selective .kappa.-receptor antagonist may restore the physiologic control of glucagon release.
[0020] The methods described herein restore the incretin effect, first-phase insulin secretion and/or physiologic insulin secretion through administration a selective .kappa.-receptor antagonist, Suitably, the present invention may also provide a method of treating Type 2 diabetes mellitus, atherogenic dyslipidemia, obesity/overweight or metabolic syndrome through administration of a selective .kappa.-receptor antagonist. The administration of an effective amount of a selective .kappa.-receptor antagonist may reduce weight gain or lower weight in a subject in need thereof.
[0021] Suitably, an insulinogenic agent may be used in combination with a selective .kappa.-receptor antagonist. An "insulinogenic agent" stimulates, participates in the stimulation of, or potentiates the biosynthesis of insulin by the pancreatic .beta.-cells. Examples of insulinogenic agents include sulfonylureas, repaglinide, nateglinide, mitiginide and BTS-67-582. Suitably, the insulinogenic agent is provided in an extended release composition, i.e., the insulinogenic agent is formulated such that it is released over a period of time. An extended release insulinogenic agent acts to potentiate the synthesis of insulin.
[0022] Pharmacologically equivalent derivatives of a selective .kappa.-receptor antagonist include any pharmaceutically acceptable salts, hydrates, esters, ethers, amides, or any other derivative which is not biologically or otherwise undesirable and induces the desired pharmacological and/or physiological effect.
[0023] The selective .kappa.-receptor antagonist and the insulinogenic agent (together referred to as "active agent") are suitably administered in a pharmaceutical composition, which include the active agent(s) and one or more pharmaceutically acceptable excipients such as stabilizers, anti-oxidants, binders, coloring agents, emulsifiers. The pharmaceutical composition may be administered as a solution, an emulsion, a suspension, a dispersion, a transdermal patch, a pill, a tablet or a capsule. One of ordinary skill in the art would be able to formulate the pharmaceutical composition using the appropriate solid, liquid or gel carriers. The selective .kappa.-receptor antagonist and the insulinogenic agent may be formulated separately or together.
[0024] Various methods for administration of the active agent(s) may be employed. For example, the active agent(s) may be given sublingually, orally, enterally, parenterally, topically, systemically or may be injected intravascularly, subcutaneously, peritoneally, and so forth. The active agent(s) may be administered weekly, semi-weekly, daily, or multiple times a day, such as twice a day or three times a day. The selective .kappa.-receptor antagonist and the insulinogenic agent may be administered concurrently. Alternatively, the selective .kappa.-receptor antagonist may be administered before or after administration of the insulinogenic agent.
[0025] The dosage of a selective .kappa.-receptor antagonist will vary widely, depending upon the frequency of administration, the manner of administration, and the clearance of a selective .kappa.-receptor antagonist from the subject. It will be appreciated that the specific dosage administered in any given case will be adjusted in accordance with the condition of the subject and other relevant medical factors that may modify the activity of the selective .kappa.-receptor antagonist. For example, the specific dose for a particular patient depends on age, body weight, general state of health, diet, the timing and mode of administration, the rate of excretion and medicaments used in combination. For example, a suitable weekly dose of a selective .kappa.-receptor antagonist may be less than about 300 ng per kg of body weight. Alternatively the weekly dose of a selective .kappa.-receptor antagonist may be less than about 200 ng per kg of body weight, less than about 150 ng per kg of body weight or less than about 100 ng per kg of body weight. The initial dose may be larger, followed by smaller maintenance doses. The dose may be administered as infrequently as weekly or biweekly, or fractionated into smaller doses and administered daily, semi-weekly, etc, to maintain an effective dosage level. A suitable daily dosage of a selective .kappa.-receptor antagonist is less than about 80 ng per kg of body weight, Alternatively the daily dosage of a selective .kappa.-receptor antagonist may be less than about 50 ng per kg of body weight, less than about 25 ng per kg of body weight, or less than about 20 ng per kg of body weight.
[0026] The dosage of the insulinogenic agent will vary depending on the condition of the subject or other relevant medical factors that modify the activity of the insulinogenic agent or the response of the subject. For example, the specific dose for a particular patient depends on the severity of glucose intolerance, age, body weight, general state of health, diet, the timing and mode of administration, the rate of excretion, and medicaments used in combination. The initial dose may be larger, followed by smaller maintenance doses. The dose may be formulated for extended release, and administered as infrequently as weekly or biweekly, or fractionated into smaller doses and administered daily, semi-weekly, weekly, etc. to maintain an effective dosage level. One of ordinary skill in the art would be able to determine the appropriate dose of the insulinogenic agent.
[0027] As used in this specification and the appended claims, the singular forms "a," "an," and "the" include plural referents unless the content clearly dictates otherwise. It should also be noted that the term "or" is generally employed in its sense including "and/or" unless the content clearly dictates otherwise. All publications, patents and patent applications referenced in this specification are indicative of the level of ordinary skill in the art to which this invention pertains. All publications, patents and patent applications are herein expressly incorporated by reference to the same extent as if each individual publication or patent application was specifically and individually indicated by reference. In case of conflict between the present disclosure and the incorporated patents, publications and references, the present disclosure should control.
[0028] It also is specifically understood that any numerical range recited herein includes all values from the lower value to the upper value, i.e., all possible combinations of numerical values between the lowest value and the highest value enumerated are to be considered to he expressly stated in this application. For example, if a concentration range is stated as 1% to 50%, it is intended that values such as 2% to 40%, 10% to 30%, or 1% to 3%, etc., are expressly enumerated in this specification. For further example, if a dosage is stated as less than about 250 ng/kg of body weight, it is intended that values such as 50 to 200 ng per kg of body weight, and 100 to 200 ng per kg of body weight are expressly enumerated in this specification. These are only examples of What is specifically intended.
[0029] The present invention is further explained by the following examples, which should not be construed by way of limiting the scope of the present invention.
EXAMPLE 1
Evaluation of Blood Glucose in a Human
[0030] The daily blood glucose profile of a male subject with early stage, [non-insulin dependent] Type 2 diabetes was monitored during extended periods of administration of guanidinylated naltrindole (GNTI). A summary of the blood glucose reading is shown in Table 1.
TABLE-US-00001 TABLE 1 Summary Average, mg/dL 92 Highest Blood Glucose, mg/dL 127 Lowest Blood Glucose, mg/dL 61 Standard Deviation, mg/dL 14 Number of Glucose Readings 227 Days Covered 15 Number of Days Without Tests 0 Average Readings Per Day 15.1 Deleted Glucose Readings 0 Control Readings 13 Deleted Control Readings 0 Average of Recorded Daily Insulin Shots with All Days Covered: 0.0 with Days with Insulin Records: 0.0
[0031] A weekly dose of about 70 ng/kg was administered. The subject's blood glucose (BG) levels were measured in mg/dL at time intervals as shown in Table 2.
TABLE-US-00002 TABLE 2 Early Overall AM Morning Midday Evening Night Time Range 00:00 05:00 11:00 15:00 19:00 04:59 10:59 14:59 18:59 23:59 Average, mg/dL 92 79 90 92 91 95 Std. Dev. mg/dL 14 3 12 14 13 15 Number of 227 4 65 56 44 58 Readings Very Highs, >300 Highs. 156-300 In Target, 65-155 98% 100% 97% 98% 100% 97% Lows, 51-64 2% 3% 2% 3% Very Lows, <51
[0032] FIGS. 1-3 illustrate the blood glucose readings over an extended period of time, a modal week and a modal day. The target blood glucose range was 65-155 mg/dL. Table 3 illustrates that 98% of the readings were within the target range. The other 2% of the readings fell below the target range and fell between 51-64 mg/dL. Note that none of these readings were in the hypoglycemic range.
TABLE-US-00003 TABLE 3 Reading/Range Glucose Ranges, mg/dL Number Percent Very High (301-601) 0 0% High (156-300) 0 0% Target (65-155) 222 98% Lows (51-64) 5 2% Very Low (0-50) 0 0%
[0033] FIGS. 1-3 illustrate the subject's blood glucose readings over an extended period of time, a modal week, and a modal day. Accordingly, 227 blood glucose readings were taken, averaging 92 mg/dL over a 15 day period. 98% of all readings were between 65 and 127 mg/dL, and 2% between 61 and 65, representing a very narrow overall spread. These data exemplify glucose dependent insulin secretion, one of the characteristics of GNTI mediated incretin targeted treatment of Type 2 diabetes mellitus subjects.
[0034] Table 4 illustrates the blood glucose statistics by day of week. These data illustrate that once weekly administration of GNTI aided in maintaining blood glucose levels within the target range.
TABLE-US-00004 TABLE 4 Blood Glucose Statistics by Day of Week, mg/dL Weekdays Weekends Mon Tue Wed Thur Fri Sat Sun Average, mg/dL 92 92 92 87 88 94 96 92 92 Std. Dev, mg/dL 15 12 11 17 15 14 15 10 13 Number of 148 79 30 23 28 36 31 32 47 Readings Very Highs, >300 Highs, 156-300 In Target, 65-155 97 100 100 87 93 100 100 100 100 (as %) Lows, 51-64 3 13 7 (as %) Very Lows, <51
[0035] The weight of the male subject with early stage Type 2 diabetes mellitus was also monitored during extended periods of administration on GNTI, decreasing from a baseline body mass index (BMI) of 25 to an average BMI of 22.8, corresponding to a reduction by about 9%.
EXAMPLE 2
Evaluation of Blood Glucose in Rhesus Monkeys
[0036] A cohort of rhesus monkeys having progressively increasing degrees of impaired glucose tolerance were monitored following an oral dose of GNTI of 86 ng/kg of body weight. Baseline readings of blood glucose (BG), high density lipoprotein C (HDL-C) and triglycerides (TG) were taken on Day 0. Table 5 shows the baseline readings on Day 0 and the results of the second reading on Day 8 of BG, HDL-C and TG.
TABLE-US-00005 TABLE 5 Oral Dose BG HDL-C TG Animal Sequence GNTI mg/dL mg/dL mg/dL r89163 Baseline 65 64 59 Day 0 86 ng/kg Day 8 63 84 60 Change (-3%) (+)31% (--) r98068 Baseline 71 81 <45 Day 0 86 ng/kg Day 8 51 85 <45 Change (-)29% (+)5% (--) r96027 Baseline 74 44 112 Day 0 86 ng/kg Day 8 53 46 89 Change (-)28% (+)10% (-)10% r00072 Baseline 80 48 <45 Day 0 86 ng/kg Day 8 67 61 <45 Change (-)16% (+)27% (--) r01078 Baseline 81 51 <45 Day 0 86 ng/kg Day 8 56 63 <45 Change (-)30% (+)24% (--)
[0037] The effect of GNTI was most readily seen in the change of blood glucose and blood lipid levels. Subject r89163 had a baseline blood glucose level of 65, i.e., a "normal" fasting blood glucose level for a rhesus monkey. The other 4 subjects of the cohort, i.e., r98060, r96027, r00072, r01078, had baseline blood glucose levels ranging from 71-81 and were considered to have impaired glucose tolerance. On day 8, the subjects having impaired glucose tolerance showed a decrease in blood glucose ranging from 16%-30% with an average of 26%. Comparatively, the glucose "normal" subject (r89163), receiving the same GNTI dose, exhibited a decrease in blood glucose levels of 3% only, and in a glucose dependent manner, a characteristic feature of physiological incretin action. The lipid "normal" subject (r98068) exhibited an increase in HDL-C lipid levels of only 5%, where the four dyslipidemic subjects (r89163, r96027, r00072, r01078) exhibited HDL-C increase in the range of 10-31%, with an average of 23%.
EXAMPLE 3
Development of a Meal Tolerance Test (MTT)
[0038] To test for the effect of physiological first phase insulin secretion, a meal tolerance test (MTT) was developed. The meal tolerance test involved oral administration of meal based nutrients within suitable ratios of carbohydrates, proteins and fats. The ratios used for the given example were 70% carbohydrates, 8% protein and 22% fats. Prior to administration of a MTT to subject r89163, baseline blood glucose, insulin and glucagon readings were taken as seen in Table 6. Following MTT administration, the same readings were taken at time points of 3-9 minutes to test for first-phase insulin secretion and the subsequent reduction in blood glucose. It was determined that a first-phase insulin secretion dependent slight increase in insulin concentration appeared, typically, at 5 minutes post MTT administration, followed by a corresponding blood glucose decrease at 7 minutes. A slight decrease in glucagon levels was also coupled with first-phase insulin secretion at 5 minutes. The MTT serves as a corollary to the intravenous glucose tolerance test (IVGTT) and better measures first-phase insulin secretion because oral administration of nutrients has a more direct effect on the incretin effect.
TABLE-US-00006 TABLE 6 Test Teste Glucagon Points Meal Cal BG Insulin GIP GLP-1 Glucagon Change T0- Animal Min Drug C/P/F (%) Mg/dL microU/mL Pg/mL Pmol/L Pg/mL T60/120 R89163 None 70/8/22 T-0 69.8 24.43 506.96 Test Meal Administration T-3 73.3 15.17 506.86 T-4 73.3 28.85 517.64 TP5/Ins. 76.7 34.63 480.54 T-6 76.1 32.8 456.43 TP7/BG 74.3 32.02 430.75 T-8 75.5 24.81 448.98 T-9 75.6 24.73 481.24
EXAMPLE 4
Application of the Meal Tolerance Test
[0039] Table 7 illustrates administration of the MTT to two rhesus monkeys categorized as metabolically "normal" based on "normal" fasting blood glucose values. The monkeys were given a diet of 55-60% carbohydrates, 15-25% protein, and 15-30% fat. Readings of blood glucose, insulin, and glucagon were taken at time points 0, 5, 7, 60 and 120 minutes. Additionally, at the same time points, the incretin hormones, GIP and GLP-1 were measured to more directly measure the incretin effect. In subject r98038, a significant increase in insulin and concurrent drop in glucagon were seen at time point 5, as would be expected. One hour after MTT administration very significant increases in insulin, GIP and GLP-1 were seen in subject r98038, again coupled with consecutive decreases in glucagon. Comparison of blood glucose levels at time points 0 and 120 minutes illustrates that normal metabolic functioning has returned the subject's blood glucose levels to normal within 120 minutes of MTT administration.
[0040] By contrast, subject r91081 appears from its baseline blood glucose level to be a "normal" metabolic subject. However, the characteristic first-phase insulin secretion at time point 5 following MTT administration is lacking as is the time point 7 blood glucose decrease. Further analysis showed that at 120 minutes following MTT administration the blood glucose and glucagon values were markedly higher than the baseline readings. These results contradict the results for "normal" subject r98038, and strongly indicate impaired metabolic control which was not discernable from the fasting blood glucose value.
TABLE-US-00007 TABLE 7 Test Glucagon Points TestMealCal BG Insulin GIP GLP-1 Glucagon Change T0- Animal Min Drug C/P/F (%) Mg/dL microU/mL Pg/mL pmol/L Pg/mL T60/120 r98038 None 74/6/20 TP 0 min 58.15 15.68 <8 12.8 180.79 TP 5 min 70 25.52 <8 13.6 145.5 TP 7 min 71.15 17.16 <8 11.3 243.26 TP 60 min 53.7 108.56 640 26.6 182.7 (+) 1% TP120 min 56.3 43.97 560 34.9 138.22 .sup. (-) 23% r91081 None 70/8/22 TP 0 min 56.15 24.71 175 196 99.8 TP 5 min 63.05 32.44 160 178 95.4 TP 7 min 64.5 59.48 150 146 90.51 TP 60 min 63.85 111.1 950 141 114.49 (+) 14.7% TP120 min 78.15 202.29 950 124 145.32 (+) 45.6%
EXAMPLE 5
[0041] Table 8 illustrates the effect of GNTI on physiologic control of glucagon levels. Baseline readings were taken on Day 1 of blood glucose, insulin, glucagon, GIP, and GLP-1 in metabolically impaired rhesus monkeys as determined through administration of the MTT. Cumulative in-situ doses of 45 ng/kg and 95 ng/kg were active on day 5 and day 7, respectively, during administration of the MTT. Readings of blood glucose, insulin, glucagon, GIP, and GLP-1 were taken at time points 0, 60 minutes and 120 minutes. As Table 8 illustrates, both dosages resulted in a marked decrease in glucagon at both time points 60 and 120 in all subjects tested. Comparison to untreated subject r91081 in Table 7 further demonstrates the marked decrease in glucagon.
TABLE-US-00008 TABLE 8 Test Points Glucagon 0/60/120 GNTI TestMealCal BG Insulin GIP GLP-1 Glucagon Change T0- Animal Minutes ng/kg C/P/F (%) mg/dL microU/mL pg/mL pmol/L pg/mL T60/120 r96022 Day 1 Baseline No Drug 64 16 25 13 394 Day 5 TP 0 min 45 ng/kg 73/9/18 51 14 8 11 411 TP 60 min 64 122 650 31 267 -35% TP120 min 67 200 n/a 31 261 -36% Day 7 TP 0 min 95 ng/kg 74/7/19 56 14 30 28 472 TP 60 min 70 100 675 35 240 -49% TP120 min 63 54 675 n/a 271 -43% rh2251 Day 1 Baseline No Drug 76 11 140 13 377 Day 5 TP 0 min 45 ng/kg 73/9/18 83 57 50 34 411 TP 60 min 98 396 640 83 366 -11% TP120 min 78 249 760 82 249 -39% Day 7 TP 0 min 95 ng/kg 75/6/19 82 71 25 60 487 TP 60 min 106 401 675 n/a 351 -28% TP120 min 95 307 775 223 267 -45% rh2258 Day 1 Baseline No Drug 74 15 n/a 10 316 Day 5 TP 0 min 45 ng/kg 74/8/18 69 23 n/a 19 302 TP 60 min 70 145 320 49 172 -43% TP120 min 76 137 775 79 122 -60% Day 7 TP 0 min 95 ng/kg 74/7/19 73 30 20 67 293 TP 60 min 69 54 75 60 107 -63% TP120 min 86 71 240 77 108 -63%
[0042] While the present invention has now been described and exemplified with some specificity, those skilled in the art will appreciate the various modifications, including variations, additions, and omissions that may be made in what has been described. Accordingly, it is intended that these modifications also be encompassed by the present invention and that the scope.
* * * * *
D00001

D00002

D00003

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.