Humanized Antibodies Specific To The Protofibrillar Form Of The Beta-amyloid Peptide
Baurin; Nicolas ; et al.
U.S. patent application number 16/142666 was filed with the patent office on 2019-04-25 for humanized antibodies specific to the protofibrillar form of the beta-amyloid peptide. The applicant listed for this patent is SANOFI. Invention is credited to Nicolas Baurin, Francis Blanche, Beatrice Cameron, Marc Duchesne, Vincent Mikol, Souad Naimi, Laurent Pradier, Yi Shi.
| Application Number | 20190119365 16/142666 |
| Document ID | / |
| Family ID | 41591699 |
| Filed Date | 2019-04-25 |







| United States Patent Application | 20190119365 |
| Kind Code | A1 |
| Baurin; Nicolas ; et al. | April 25, 2019 |
HUMANIZED ANTIBODIES SPECIFIC TO THE PROTOFIBRILLAR FORM OF THE BETA-AMYLOID PEPTIDE
Abstract
Humanized antibodies specific to the protofibrillar form of the beta-amyloid peptide are provided. Methods of using antibodies specific to the protofibrillar form of the beta-amyloid peptide in the field of Alzheimer's disease are also provided.
| Inventors: | Baurin; Nicolas; (Arpajon, FR) ; Blanche; Francis; (Paris, FR) ; Cameron; Beatrice; (Paris, FR) ; Duchesne; Marc; (Sucy En Brie, FR) ; Mikol; Vincent; (Charenton-le-Pont, FR) ; Naimi; Souad; (Saint Maur Des Fosses, FR) ; Pradier; Laurent; (Verrieres, FR) ; Shi; Yi; (Paris, FR) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 41591699 | ||||||||||
| Appl. No.: | 16/142666 | ||||||||||
| Filed: | September 26, 2018 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15171708 | Jun 2, 2016 | 10112991 | ||
| 16142666 | ||||
| 14042942 | Oct 1, 2013 | 9382312 | ||
| 15171708 | ||||
| 13319710 | Mar 26, 2012 | 8614299 | ||
| PCT/FR2010/050915 | May 11, 2010 | |||
| 14042942 | ||||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C07K 16/18 20130101; C07H 21/04 20130101; C07K 2317/52 20130101; C12N 2015/8518 20130101; C07K 2317/565 20130101; C12N 15/63 20130101; C07K 2317/33 20130101; A61K 39/3955 20130101; A61P 25/28 20180101; C07K 2317/24 20130101; C07K 2317/92 20130101; A61P 25/00 20180101; C07K 2317/56 20130101; C07K 2317/71 20130101 |
| International Class: | C07K 16/18 20060101 C07K016/18 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| May 12, 2009 | FR | 0953133 |
Claims
1. A humanized antibody specific for the protofibrillar form of the peptide A-.beta..
2. The antibody of claim 1, wherein: the antibody characterized in that it binds to aggregated A-.beta. peptide-senile plaques and not to diffuse deposits of A-.beta. peptides; the antibody induces a reduction in amyloid plaques; or affinity of the antibody for the protofibrillar form of A-.beta. peptide is at least 100 times greater than affinity of the antibody for other forms of A-.beta. peptide.
3. The antibody of claim 1, comprising at least one CDR encoded by a nucleotide sequence having a sequence identical to one of the sequences SEQ ID NO: 9: 11, 13, 15, 17 and 19 or by sequences respectively differing by 1, 2, 3, 4 or 5 nucleotides of these sequences.
4. The antibody of claim 1, comprising at least one CDR having an amino acid sequence identical to one of the sequences SEQ ID NO: 10, 12, 14, 16, 18 and 20 or by sequences respectively differing by one or two amino acids of these sequences.
5. (canceled)
6. (canceled)
7. The antibody of claim 1, comprising the CDR amino acid sequences of SEQ ID NO: 10, 12, 14, 16, 18 and 20; SEQ ID NO: 10, 12, 14, 32, 18 and 20; or SEQ ID NO: 10, 12, 30, 32, 18 and 20.
8. The antibody of claim 1, comprising the CDRs encoded by the nucleotide sequences: SEQ ID NO 9, 11, 13, 31, 17 and 19; SEQ ID NO 9, 11, 29, 31, 17 and 19; or sequences differing respectively by 1, 2, 3, 4 or 5 nucleotides of these sequences.
9-11. (canceled)
12. The antibody of claim 1, comprising a heavy chain variable region encoded by a sequence having at least 80% identity with a nucleic acid sequence selected from the group consisting of SEQ ID NO: 1, 5, 25, and 27.
13. The antibody of claim 1, comprising a heavy chain variable region having at least 80% identity with an amino acid sequence selected from the group consisting of SEQ ID NO: 2, 6, 26, and 28.
14. The antibody of claim 1, comprising a light chain variable region encoded by a sequence having at least 80% identity with a nucleic acid sequence selected from the group consisting of SEQ ID NO: 3, 7, 21, and 23.
15. The antibody of claim 1, comprising a light chain variable region having at least 80% identity with an amino acid sequence selected from the group consisting of SEQ ID NO: 4, 8, 22, and SEQ ID NO 24.
16. The antibody of claim 1, comprising a heavy chain variable region encoded by a nucleotide sequence selected from the group consisting of SEQ ID NO 5 and SEQ ID NO 27, or a heavy chain variable region amino acid sequence selected from the group consisting of SEQ ID NO 6 and SEQ ID NO 28.
17. (canceled)
18. The antibody of claim 1, comprising a light chain variable region encoded by a nucleotide sequence selected from the group consisting of SEQ ID NO 7 and SEQ ID NO 23, or a light chain variable region amino acid sequence selected from the group consisting of SEQ ID NO 8 or SEQ ID NO 24.
19. (canceled)
20. The antibody of claim 1, comprising sequences encoded by the nucleotide sequences selected from the group consisting of SEQ ID NO: 5 and 7, or amino acid sequences selected from the group consisting of SEQ ID NO: 6 and 8.
21-25. (canceled)
26. The antibody of claim 1, comprising a pair of nucleotide sequences selected from the group consisting of SEQ ID NO: 1 and 3, SEQ ID NO: 5 and 23, and SEQ ID NO: 27 and 23, or a pair of amino acid sequences selected from the group consisting of SEQ ID NO: 2 and 4, SEQ ID NO: 6 and 24, and SEQ ID NO: 28 and 24.
27-33. (canceled)
34. A method of treating a disease associated with a neurodegenerative disorder comprising administering to the patient the antibody of claim 1, wherein the neurodegenerative disease is optionally Alzheimer's disease.
35. (canceled)
36. A pharmaceutical composition comprising the antibody of claim 1 and one or more pharmaceutically acceptable excipients.
37-40. (canceled)
41. A polynucleotide encoding a polypeptide having at least 80% identity with one of SEQ ID NO:2, 4, 6, 8, 22, 24, 26 or 28, or a polynucleotide having at least 80% identity with one of SEQ ID NO: 1, 3, 5, 7, 21, 23, 25 or 27.
42. (canceled)
43. A recombinant vector comprising a nucleic acid according to claim 41.
44. A host cell comprising a vector according to claim 43.
45. A method of producing a humanized antibody specific for the protofibrillar form of the peptide A-.beta. comprising culturing the host cell of claim 44.
Description
[0001] The present invention relates to humanized antibodies specific for the protofibrillar form of .beta.-amyloid peptide. The present invention also relates to the therapeutic, diagnostic and/or preventive use of these antibodies, in particular associated with the induction and with the progression of neurodegenerative disorders and/or with diseases associated with the deposition of amyloid plaques, and notably Alzheimer's disease.
[0002] Alzheimer's disease (AD) is a progressive neurodegenerative disease that affects a high proportion of the older population. This disease is characterized clinically by memory loss and a decline in cognitive functions, and neuropathologically by the presence, in the brain, of intracellular neurofibrillar deposits and of extracellular deposits of the .beta.-amyloid peptide (A-.beta.) forming amyloid plaques. (Yanker et al. Nature Med. Vol. 2 No. 8 (1996)). As well as these signs, there are many other abnormal changes including a deterioration of the immune and inflammatory systems as well as a deterioration of mitochondrial function, which can lead to an increase in oxidative stress, activation of the mechanisms of apoptosis and ultimately to cell death.
[0003] Amyloid plaques are predominantly composed of A-.beta. peptides with 40 or 42 residues, which are generated during the proteolytic process of the .beta.-amyloid peptide precursor (APP) protein. The extracellular deposits of A-.beta. peptides represent the invariable early characteristic feature of all forms of AD, including the familial forms (FAD). FADs appear relatively early (between 40 and 60 years) and are due to mutations in the gene of APP in 5% of cases of FAD (>20 families) with six single or double missense mutations; in the gene of presenilin 1 (PS 1) in 50 to 70% of cases of FAD (>200 families) with more than 80 different mutations identified to date; and in the gene of presenilin 2 (PS 2) in fewer cases of FAD with 2 missense mutations described in 8 families. Mutations in these three genes have been shown to induce changes in the proteolysis of APP, which lead to overproduction of A-.beta. and to the early appearance of the pathology and of symptoms that are similar to those of the sporadic forms of AD.
[0004] The neuronal toxicity of the amyloid plaques might reside in the high molecular weight fibrils that are formed by aggregation of soluble A-.beta. peptides in fibrillar forms that are soluble initially (also called protofibrillar form) and are then converted to insoluble forms incorporated in the amyloid plaques. In fact, it was shown in vitro that the soluble A-.beta. peptide aggregates progressively to a fibrillar form (i.e. which can be labelled with agents such as Congo Red or thioflavin S which recognize the beta-sheet tertiary structures of the peptides/proteins), of high molecular weight (>200 kDa) but still soluble. Because this form is soluble, it is often called the protofibrillar form, whereas the fibrils result from even greater aggregation, leading to loss of solubility. The protofibrillar transitional forms are generally regarded as the precursors of the amyloid fibres and might be responsible for the cellular dysfunction and the neuronal loss in Alzheimer's disease and in other diseases associated with the aggregation of proteins.
[0005] It has been shown that the senile amyloid plaques (i.e. aggregated, also called mature plaques) are correlated with the cognitive status of Alzheimer's patients in contrast to the diffuse deposits of A-.beta. peptide which are also widely present in unaffected patients. (Duyckaerts et al., Neurobiol. Aging 1997; 18: 33-42 and Jellinger et al., 1998; 54:77-95). By targeting these senile amyloid plaques in particular, it is therefore possible to treat Alzheimer's disease more specifically and effectively.
[0006] A great many treatments have been tried for preventing the formation of the A-.beta. peptides, for example inhibitors of the proteolytic process of APP.
[0007] Immunotherapeutic strategies such as the administration of anti-A-.beta. antibody (to reduce the amyloid deposits) or immunization with antigens of the A-.beta. peptides (to promote a humoral response) have been tested in order to reduce the size and density of the plaques.
[0008] For example, a method of treatment against Alzheimer's disease has been described (U.S. Pat. No. 7,179,463), consisting of administering an antibody directed against a protofibril presenting an Arctic mutation in the region coding for the A-.beta. peptide.
[0009] No example of antibody has really been described. Moreover, no comparison of the affinity of the antibodies for the peptides as a function of the molecular weight of these peptides has been performed. Other patents (U.S. Pat. Nos. 6,761,888 and 6,750,324) have referred to antibodies recognizing various epitopes along the amino acid sequence of the peptide A-.beta..sub.42. An international application (WO2007/108756) has been filed concerning antibodies specific for the protofibrils but the antibodies described recognize both the high molecular weight A-.beta. peptides and the medium-weight oligomers. Furthermore, there is no mention of the affinity of the antibodies for the mature plaques relative to their affinity for the diffuse plaques.
[0010] Despite the current development of knowledge concerning Alzheimer's disease, there is still a need for compositions and methods of treatment and/or prevention of this pathology limiting the secondary effects to the maximum extent. Antibodies such as described in the present application, humanized and specific for the protofibrillar form of the A-.beta. peptides, aim to solve this problem. Permitting recognition of the senile amyloid plaques but not the diffuse plaques, the antibodies according to the invention recognize the pathological plaques much more effectively than antibodies recognizing all forms of Abeta, which will largely be attached to the diffuse deposits or attached to the soluble forms of monomeric or low-molecular-weight A-.beta. peptide.
[0011] Moreover, the fact that only the protofibrillar forms of the A-.beta. peptides are recognized and not the protofibrillar forms of other proteins not linked to Alzheimer's disease avoids useless binding that may reduce the concentration of antibodies that are effective against the disease.
[0012] The murine antibody that has been humanized will be called antibody 1303 throughout the present application.
[0013] The sequences that can code for or constitute the humanized antibodies according to the invention are shown in Table 2.
[0014] The present invention relates to a humanized antibody that binds specifically to the protofibrillar form of the A-.beta. peptide, i.e. a high molecular weight peptide.
[0015] In a more advantageous embodiment, the antibody binds to the A-.beta. peptide having a molecular weight greater than 200, 300, 400 or 500 kDa.
[0016] According to one embodiment, the antibody according to the invention binds to the A-.beta. peptides aggregated into senile plaques and not to the diffuse deposits of A-.beta. peptides.
[0017] In an advantageous embodiment, the antibody according to the invention binds specifically to the protofibrillar form of the A-.beta. peptide but not to the other proteins of amyloid structure (for example IAPP, Islet Amyloid Polypeptide).
[0018] The present invention also relates to a humanized antibody having reduced effector functions, making it possible to limit adverse effects such as the development of microhaemorrhages and vasogenic oedemas.
[0019] In an advantageous embodiment, the antibody according to the invention no longer possesses effector functions.
[0020] In an even more advantageous embodiment, the antibody is an immunoglobulin G 4 whose Fc domain has undergone mutations reducing the production of half-molecules.
[0021] In an even more advantageous embodiment, the antibody is an immunoglobulin G 4 whose Fc domain has undergone mutations reducing the effector activity.
[0022] The present invention relates to a humanized antibody comprising at least one CDR encoded by a nucleotide sequence having a sequence identical to one of the sequences SEQ ID NO: 9, 11, 13, 15, 17 and 19, or by sequences differing respectively by 1, 2, 3, 4 or 5 nucleotides from these sequences.
[0023] The present invention also relates to a humanized antibody comprising at least one CDR having a sequence identical to one of the sequences SEQ ID NO: 10, 12, 14, 16, 18 and 20.
[0024] In another embodiment, the antibody according to the invention comprises at least one CDR whose sequence differs by one to two amino acids relative to one of the sequences SEQ ID NO: 10, 12, 14, 16, 18, 20 and 32, inasmuch as the antibody maintains its binding specificity.
[0025] In an advantageous embodiment, the antibody comprises the CDRs encoded by the nucleotide sequences SEQ ID NO: 9, 11, 13, 15, 17 and 19, or by sequences differing respectively by 1, 2, 3, 4 or 5 nucleotides from these sequences.
[0026] In another advantageous embodiment, the antibody comprises the CDRs of sequence SEQ ID NO: 10, 12, 14, 16, 18 and 20.
[0027] The antibody according to the invention can also comprise the CDRs encoded by the nucleotide sequences SEQ ID NO: 9, 11, 13, 31, 17 and 19 or by sequences differing respectively by 1, 2, 3, 4 or 5 nucleotides from these sequences.
[0028] In an advantageous embodiment, the antibody according to the invention comprises the CDRs of sequence SEQ ID NO: 10, 12, 14, 32, 18 and 20.
[0029] One object of the invention is the humanized antibody comprising the CDRs encoded by the nucleotide sequences SEQ ID NO: 9, 11, 29, 31, 17 and 19 or by sequences differing respectively by 1, 2, 3, 4 or 5 nucleotides from these sequences.
[0030] The invention also relates to a humanized antibody comprising the CDRs of sequence SEQ ID NO: 10, 12, 30, 32, 18 and 20.
[0031] In an advantageous embodiment, the antibody according to the invention comprises a variable part of its heavy chain (VH) encoded by a sequence having at least 80%, 85%, 90%, 95% or 99% identity with the sequence SEQ ID NO: 5 or the sequence SEQ ID NO 27.
[0032] In an advantageous embodiment, the antibody according to the invention comprises a variable part of its heavy chain (VH) comprising a sequence having at least 80%, 85%, 90%, 95% or 99% identity with the sequence SEQ ID NO: 6 or the sequence SEQ ID NO 28.
[0033] In an advantageous embodiment, the antibody according to the invention comprises a variable part of its light chain (VL) encoded by a sequence having at least 80%, 85%, 90%, 95% or 99% identity with the sequence SEQ ID NO: 7 or the sequence SEQ ID NO 23.
[0034] In an advantageous embodiment, the antibody according to the invention comprises a variable part of its light chain (VL) comprising a sequence having at least 80%, 85%, 90%, 95% or 99% identity with the sequence SEQ ID NO: 8 or the sequence SEQ ID NO 24.
[0035] In an even more advantageous embodiment, the antibody comprises a heavy chain comprising a variable part (VH) encoded by one of the nucleotide sequences SEQ ID NO 5 and SEQ ID NO 27.
[0036] In an even more advantageous embodiment, the antibody comprises a heavy chain comprising a variable part (VH) of polypeptide sequence SEQ ID NO 6 or SEQ ID NO 28.
[0037] In another embodiment, the antibody comprises a light chain comprising a variable part (VL) encoded by one of the nucleotide sequences SEQ ID NO 7 and SEQ ID NO 23.
[0038] In another embodiment, the antibody comprises a light chain comprising a variable part (VL) of polypeptide sequence SEQ ID NO 8 or SEQ ID NO 24.
[0039] In an advantageous embodiment, the antibody comprises the sequences encoded by the nucleotide sequences SEQ ID NO: 5 and 7.
[0040] In an advantageous embodiment, the antibody comprises the polypeptide sequences SEQ ID NO: 6 and 8.
[0041] In another embodiment, the antibody comprises the sequences encoded by the nucleotide sequences SEQ ID NO: 5 and 23.
[0042] In another embodiment, the antibody comprises the polypeptide sequences SEQ ID NO: 6 and 24.
[0043] In another embodiment, the antibody comprises the sequences encoded by the nucleotide sequences SEQ ID NO: 27 and 23.
[0044] In another embodiment, the antibody comprises the polypeptide sequences SEQ ID NO: 28 and 24.
[0045] The present invention also relates to an antibody comprising a heavy chain encoded by a sequence having at least 80%, 85%, 90%, 95% or 99% identity with one of the nucleotide sequences SEQ ID NO 1 and SEQ ID NO 25.
[0046] The present invention also relates to an antibody comprising a heavy chain having at least 80%, 85%, 90%, 95% or 99% identity with the polypeptide sequence SEQ ID NO 2 or with the polypeptide sequence SEQ ID NO 26.
[0047] In an advantageous embodiment the antibody comprises a light chain encoded by a sequence having at least 80%, 85%, 90%, 95% or 99% identity with one of the nucleotide sequences SEQ ID NO 3 and SEQ ID NO 21.
[0048] In another embodiment the antibody comprises a light chain comprising a sequence having at least 80%, 85%, 90%, 95% or 99% identity with one of the polypeptide sequences SEQ ID NO 4 and SEQ ID NO 22.
[0049] One object of the invention is an antibody comprising the sequences encoded by the nucleotide sequences SEQ ID NO: 1 and 3.
[0050] Another object of the invention is an antibody whose sequence comprises the polypeptide sequences SEQ ID NO: 2 and 4.
[0051] One object of the invention is an antibody comprising the sequences encoded by the nucleotide sequences SEQ ID NO: 1 and 21.
[0052] Another object of the invention is an antibody whose sequence comprises the polypeptide sequences SEQ ID NO: 2 and 22.
[0053] One object of the invention is an antibody comprising the sequences encoded by the nucleotide sequences SEQ ID NO: 25 and 21.
[0054] Another object of the invention is an antibody whose sequence comprises the polypeptide sequences SEQ ID NO: 26 and 22.
[0055] Another object of the invention is a humanized anti-peptide A.beta. antibody having an affinity for the protofibrillar form of peptide A.beta. at least 100 times greater than its affinity for the other forms of this peptide.
[0056] Another object of the invention is an antibody, characterized in that it induces a reduction of amyloid plaques.
[0057] Another object of the invention is the use of a humanized anti-peptide A.beta. antibody in the treatment of diseases associated with neurodegenerative disorders, and in particular in the treatment of Alzheimer's disease.
[0058] Another object of the invention is a pharmaceutical composition comprising a humanized anti-peptide A.beta. antibody and excipients.
[0059] Another object of the invention is a method of treatment of Alzheimer's disease comprising the administration of a humanized anti-peptide-A.beta. antibody to the patient.
[0060] Another object of the invention is a cell or cells producing a humanized anti-peptide-A.beta. antibody, as well as the method of production of this antibody comprising the culturing of these cells. Said cells are derived advantageously from one cell line.
[0061] One object of the invention is a medicinal product comprising a humanized anti-peptide-A.beta. antibody.
[0062] One object of the invention is a polynucleotide coding for a polypeptide having at least 80%, 85%, 90%, 95% or 99% identity with one of the sequences SEQ ID NO: 2, 4, 6, 8, 22, 24, 26 or 28.
[0063] Another object of the invention is a polynucleotide with a sequence having at least 80%, 85%, 90%, 95% or 99% identity with one of the sequences SEQ ID NO: 1, 3, 5, 7, 21, 23, 25, or 27.
[0064] Another object of the invention is a recombinant vector comprising a nucleic acid having one of the sequences SEQ ID NO 1, 3, 5, 7, 21, 23, 25, or 27, as well as a host cell comprising this vector.
Definitions
[0065] Specific binding is understood as a difference by a factor of at least about 10, 20, 30, 40, 50, or 100 between the strength of binding to one receptor relative to another, here between binding to the protofibrillar form of the A-.beta. peptide and binding to the other forms of the peptide.
[0066] "Epitope" means the site of the antigen to which the antibody binds. If the antigen is a polymer, such as a protein or a polysaccharide, the epitope can be formed by contiguous or non-contiguous residues. Here the epitope is conformational, i.e. related to the three-dimensional structure of the protofibrillar A-.beta. peptide.
[0067] "Protofibrillar form" means an oligomeric form of A-.beta. peptides, soluble in vitro and which can be isolated as an entity of molecular weight greater than 200 kDa, 300 kDa, 400 kDa or 500 kDa and which can fix agents such as thioflavin-S or Congo Red.
[0068] "Senile plaque" means a plaque composed of an amyloid core (fixing thioflavin S or Congo Red) surrounded by dystrophic neurites and a reaction of glial cells. Senile plaques are found in particular in patients with Alzheimer's disease, in contrast to the diffuse amyloid deposits (which do not fix thioflavin S or Congo Red), which are far more numerous but are not associated with the disease.
[0069] An antibody, also called immunoglobulin, is composed of two identical heavy chains ("CH") and two identical light chains ("CL"), which are joined by a disulphide bridge.
[0070] Each chain contains a constant region and a variable region. Each variable region comprises three segments called "complementarity determining regions" ("CDRs") or "hypervariable regions", which are mainly responsible for binding to the epitope of an antigen.
[0071] The term "VH" refers to the variable regions of a heavy chain of immunoglobulin of an antibody, including the heavy chains of a fragment Fv, scFv, dsFv, Fab, Fab' or F(ab)'.
[0072] The term "VL" refers to the variable regions of a light chain of immunoglobulin of an antibody, including the light chains of a fragment Fv, scFv, dsFv, Fab, Fab' or F(ab)'.
[0073] "Antibody" also means any functional fragment of antibody: Fab (Fragment antigen binding), Fv, scFv (single chain Fv), Fc (Fragment, crystallizable). Preferably, these functional fragments will be fragments of type Fv, scFv, Fab, F(ab') 2, Fab', scFv-Fc, diabodies, multispecific antibodies (notably bispecific), synthetic polypeptides containing the sequences of one or more CDRs, which generally possess the same specificity of fixation as the humanized antibody from which they are derived. According to the present invention, fragments of antibodies of the invention can be obtained from the humanized antibodies by methods such as digestion by enzymes, such as pepsin or papain and/or by cleavage of the disulphide bridges by chemical reduction.
[0074] Nanobodies also come under this definition.
[0075] "CDR or CDRs" denotes the hypervariable regions of the heavy and light chains of the immunoglobulins as defined by Kabat et al. (Kabat et al., Sequences of proteins of immunological interest, 5th Ed., U.S. Department of Health and Human Services, NIH, 1991, and later editions). There are 3 heavy-chain CDRs and 3 light-chain CDRs. The term CDR or CDRs is used here to denote, as applicable, one or more, or even all, of these regions that contain the majority of the amino acid residues responsible for the affine binding of the antibody for the antigen or the epitope that it recognizes. The most conserved regions of the variable domains are called FR regions or sequences, for "framework regions".
[0076] The present invention relates to humanized antibodies.
[0077] "Humanized antibody" means an antibody that contains mainly human immunoglobulin sequences. This term generally refers to a non-human immunoglobulin that has been modified by incorporating human sequences or residues found in human sequences.
[0078] In general, humanized antibodies comprise one or typically two variable domains in which all or part of the CDR regions correspond to parts derived from the non-human parent sequence and in which all or part of the FR regions are derived from a human immunoglobulin sequence. The humanized antibody can then comprise at least one portion of a constant region of immunoglobulin (Fc), in particular that of the selected reference human immunoglobulin.
[0079] We thus try to obtain an antibody that is the least immunogenic in a human. Thus it is possible that one or two amino acids of one or more CDRs are modified by an amino acid that is less immunogenic for the human host, without substantially reducing the binding specificity of the antibody to the A-.beta. peptide of high molecular weight. Furthermore, the residues of the framework regions need not be human and it is possible that they are not modified, as they do not contribute to the immunogenic potential of the antibody.
[0080] Several methods of humanization are known by a person skilled in the art for modifying a non-human parent antibody to an antibody that is less immunogenic in humans. Complete identity of the sequences with a human antibody is not essential. In fact complete sequence identity is not necessarily a predictive indicator of reduced immunogenicity and modification of a limited number of residues can lead to humanized antibodies presenting a very attenuated immunogenic potential in humans (Molecular Immunology (2007) 44, 1986-1998).
[0081] Some methods are for example the inclusion of CDRs (grafting) (EPO 0 239 400; WO 91/09967; and U.S. Pat. Nos. 5,530,101 and 5,585,089), the resurfacing (EPO 0 592 106; EPO 0 519 596; Padlan, 1991, Molec Imm 28(4/5):489-498; Studnicka et al., 1994, Prot Eng 7(6):805-814; and Roguska et al., 1994, PNAS 91:969-973) or chain mixing (U.S. Pat. No. 5,565,332).
[0082] The present invention relates in particular to humanized antibodies whose variable parts are modified according to the technology explained in international patent application WO 2009/032661.
[0083] This technique notably uses dynamic molecular simulation based on three-dimensional models of antibodies, said models being constructed by homology.
[0084] The present invention also relates to any form of antibody having diminished effector functions, such as immunoglobulins bearing mutations of the Fc domain reducing its affinity for the receptors of the immune system or such as nanobodies.
[0085] "Effector functions" means any fixation of the Fc domain of the antibody to receptors or proteins inducing immune responses. Decreasing these effector functions makes it possible to reduce adverse effects such as the induction of microhaemorrhages (Racke et al. J Neurosci 2005, 25:629).
[0086] Affinity can be measured by any technique known by a person skilled in the art. It is advantageously measured by the Biostat Speed technique developed on the basis of the algorithms described by Ratkovsk DA and Reedy TJ (Biometrics, 1986, 42, 575-82).
[0087] In order to permit expression of heavy chains and/or light chains of the antibody according to the invention, the polynucleotides coding for said chains are inserted in expression vectors. These expression vectors can be plasmids, YACs, cosmids, retroviruses, episomes derived from EBV, and all the vectors that a person skilled in the art may judge to be suitable for expression of said chains.
[0088] These vectors can be used for transforming cells advantageously derived from one cell line. Said cell line is even more advantageously derived from a mammal. It is advantageously the CHO line or a line derived from this line, or the HEK293 line or a line derived from this line.
[0089] The transformation of the cells can be carried out by any method known by a person skilled in the art for introducing polynucleotides into a host cell. Said method can be transformation by means of dextran, precipitation by calcium phosphate, transfection by means of polybrene, protoplast fusion, electroporation, encapsulation of the polynucleotides in liposomes, biolistic injection and direct micro-injection of DNA into the nucleus.
[0090] The antibody according to the invention can be included in pharmaceutical compositions with a view to administration by the topical, oral, parenteral, intranasal, intravenous, intramuscular, subcutaneous, intraocular or other routes. Preferably, the pharmaceutical compositions contain pharmaceutically acceptable vehicles for an injectable formulation. These can be in particular sterile, isotonic saline solutions (monosodium or disodium phosphate, sodium, potassium, calcium or magnesium chloride, etc., or mixtures of said salts), or dry compositions, notably lyophilized, which, by adding sterilized water or physiological serum as appropriate, permit injectable solutes to be constituted.
[0091] As an example, a pharmaceutical composition comprises (1) a Dulbecco phosphate buffer (pH.about.7.4), optionally containing 1 mg/ml to 25 mg/ml of human serum albumin, (2) 0.9% w/v of sodium chloride (NaCl), and (3) 5% (w/v) of dextrose. It can also comprise an antioxidant such as tryptamine and a stabilizer such as Tween 20.
[0092] The pathologies in question can be any diseases associated with the deposition of amyloid plaques. In particular, the pathology in question is Alzheimer's disease.
[0093] The doses depend on the desired effect, the duration of the treatment and the route of administration used; they are generally between 5 mg and 1000 mg of antibody per day for an adult. Generally the doctor will determine the appropriate dosage in relation to the stage of the disease, the patient's age and weight, or any other patient-related factor that has to be taken into account.
[0094] The present invention is illustrated, but is not limited, by the examples given below.
BRIEF DESCRIPTION OF THE DRAWINGS
[0095] FIG. 1A: Map of the plasmid pXL4973 permitting expression of the light chain LC1 of the antiAbeta antibody 13C3-VH1VL1.
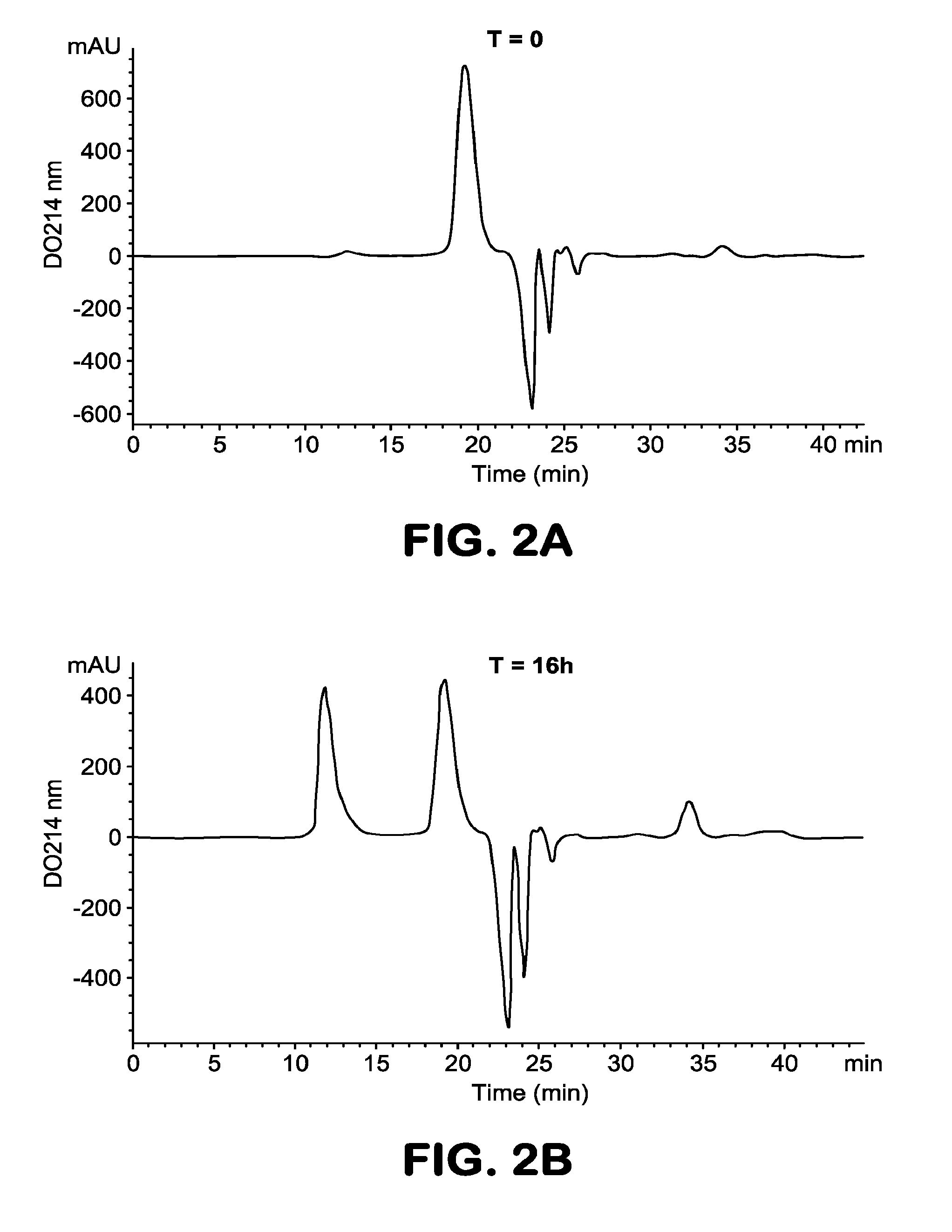
[0096] FIG. 1B: Map of the plasmid pXL4979 permitting expression of the heavy chain HCl of the antiAbeta antibody 13C3-VH1VL1.
[0097] FIGS. 2A and 2B: Separation of the protofibrils and of the low-molecular-weight oligomers by gel filtration on Superdex 75 (at t=0 and at t=16 h respectively).
[0098] FIG. 3: Determination of the molecular weight of the protofibrils.
[0099] FIGS. 4A, 4B and 4C: Determination of the affinities of the humanized antibodies (antibodies LP09027 (4A), LP09026 (4B) and LP09028 (4C) respectively) for the protofibrils (mean value from 3 experiments.+-.sem).
[0100] FIG. 5: Specificity of the humanized antibody LP09027 with respect to fibrils of A.beta..
[0101] FIGS. 6A and 6B: Specificity of the humanized antibody (LP09027) for the mature senile plaques respectively of the frontal cortex (6A) and of the hippocampus (6B) of a mouse. The arrows indicate the senile plaques.
EXAMPLES
Example 1: Obtaining Humanized Antibodies
[0102] A murine antibody 13C3 was humanized.
[0103] This example describes the sequence and the production of the humanized anti-peptide A.beta. antibody VH1VL1 (LP09027) by production by transient expression in the mammalian line HEK293 designated FreeStyle 293-F.
[0104] The cDNAs coding for the humanized variable chains VL1 and VH1 are fused with the cDNAs coding for the human constant regions Ckappa and IgG4 respectively. The sequence of the constant region IgG4 is that of the variant having the substitutions S241 P and L248E in Kabat's nomenclature, for a significant reduction in the production of half-molecules (Angla et al., 1993, Mol. Immunol., 30: 105-108) and the effector functions (WO 97/09351).
[0105] The nucleic acid sequences coding for CH1 (SEQ ID NO 1) and for CL1 (SEQ ID NO 3) were cloned independently in the expression vector to generate the plasmids pXL4973 (FIG. 1A) and pXL4979 (FIG. 1B), respectively.
[0106] A batch of the antibody is produced by production by transient expression in the line FreeStyle 293-F (Invitrogen) after co-transfection of the plasmids pXL4973 and pXL4979 according to the protocol described by Invitrogen (catalogue reference K9000-01). This batch (LP09027) is then purified by affinity chromatography on a column of MabSelect gel (Amersham) according to the supplier's recommendations and then formulated in PBS buffer (reference Dulbecco 14190-094) and submitted to sterile filtration (0.2 .mu.m). Starting from 1 L of culture, 33 mg of antibody is obtained at a purity of 97% by SDS-PAGE in denaturing conditions and by steric exclusion chromatography. The mass obtained by SDS-PAGE in denaturing conditions and by LC/MS is in agreement with the primary amino acid sequence and the presence of an N-glycan on the Fc domain, namely a mass of 23969 Da for LC1 and 49650 Da for HCl taking into account the N-glycan in the G0F form. The mass obtained by SDS-PAGE in non-denaturing conditions and by size exclusion chromatography is in agreement with the hetero-tetrameric structure of the antibody of 150 kDa (FIG. 4A).
[0107] According to the same method, batches of humanized antibodies LP09026 and LP09028 were produced starting from the nucleotide sequences SEQ ID NO 25 and SEQ ID NO 21 for LP09026 (FIG. 4B), and SEQ ID NO 1 and SEQ ID NO 21 for LP09028 (FIG. 4C).
Example 2: Preparation of Protofibrils from Peptide A.beta. (1-42)
[0108] The protofibrils were prepared from the synthetic peptide A.beta. (1-42) according to the method described by Johansson et al. (FEBS, 2006, 2618-2630). The lyophilized peptide (Anaspec reference 24224) is dissolved in 10 mM NaOH at a concentration of 100 .mu.M, then stirred for 1 min and incubated on ice for 10 min. The solution of peptide is then diluted in buffer of 100 mM sodium phosphate, 200 mM NaCl pH=7.4 to a concentration of 50 .mu.M, then stirred for 1 min. The preparation is incubated overnight at 37.degree. C. for formation of protofibrils and then centrifuged at 17900 g for 15 min at 16.degree. C. to remove the insoluble aggregates. To separate the protofibrils from the oligomeric forms of A.beta. of low molecular weight, the supernatant is loaded on a Superdex 75 gel filtration column equilibrated in 50 mM ammonium acetate buffer pH=8.5. The fractions corresponding to the protofibrils and to the low-molecular-weight oligomers are collected and stored at 4.degree. C. FIG. 2 shows a typical profile of separation of the protofibrils. The molecular weight of the protofibrils is determined by Superdex200 gel filtration using, as markers of molecular weight, the Biorad calibration kit (reference 150-1901). FIG. 3 shows that the molecular weight of the protofibrils is greater than 200 kDa.
Example 3: Specificity and Affinity of the Humanized Antibodies with Respect to the Protofibrils
[0109] 50 .mu.l of protofibrils and low-molecular-weight oligomers at a concentration of 1 .mu.g/ml in PBS (Gibco, reference 70011) are deposited in the wells of an ELISA plate (Nunc, reference 442404) and incubated overnight at 4.degree. C. After removing the excess antigen, 200 .mu.l of buffer PBS+5% milk powder (weight/volume) is deposited in each well to remove the non-specific adsorptions and incubated for 2 h at room temperature. The wells are then washed 4 times with 300 .mu.l of buffer PBS Tween 0.02%. 50 .mu.l of a primary antibody solution (dilution of 3 in 3 in PBS Tween starting from a concentration of 100 .mu.g/ml for the oligomers and from 25 .mu.g/ml for the protofibrils) is added to each well and incubated for 1 h at room temperature. The wells are washed 4 times with 300 .mu.l of buffer PBS Tween. The secondary anti-Fc human antibody coupled to peroxidase (Goat Anti Human IgG (Fc) peroxidase conjugated, Pierce, reference 31413) diluted to 1/10000 in buffer PBS Tween is added to each well and incubated for 1 h at room temperature. After 4 washings with 300 .mu.l of PBS Tween, 100 .mu.l of TMB (Interchim, reference UP664782) is added to each well and incubated for about 10 min, then the reaction is stopped with a solution of 1M HCl (Interchim, reference UPS29590) and the plates are read at an OD measured at a wavelength of 450 nm. The EC50 values are determined by BioStat Speed. The results obtained are presented in Table 1 and in FIG. 4 and show the very high specificity of the antibody for the protofibrils relative to the low-molecular-weight oligomers (factor of 184).
TABLE-US-00001 TABLE 1 EC50 (.mu.g/ml) LMW PF LMW/PF LP09026 41.4 .+-. 40.1 0.0587 .+-. 0.004 705.3 LP09027 14.7 .+-. 2.7 0.0798 .+-. 0.007 184.2 LP09028 21.8 .+-. 5.3 0.0892 .+-. 0.007 244.4
[0110] The lyophilized peptide A.beta.1-42 (Anaspec reference 24224) is dissolved according to the supplier's recommendations: 40 .mu.l of 1% NH.sub.4OH is added to 500 .mu.g of A.beta.1-42. After complete dissolution, 460 .mu.l of PBS is added to obtain a concentration of 1 mg/ml. Aliquots of 10 .mu.l are prepared and stored at -80.degree. C.
[0111] 50 .mu.l of a solution of peptide A.beta.1-42 at a concentration of 1 .mu.g/ml in carbonate buffer (NaHCO.sub.3 0.025 M (Acros Organics, reference 217120010), Na.sub.2CO.sub.3 0.025 M (Acros Organics, reference 207810010), pH 9.7 is deposited in the wells of an ELISA plate and incubated overnight at room temperature. As previously, the wells are washed with buffer PBS Tween, incubated in the presence of buffer PBS+5% milk powder (weight/volume) and washed with buffer PBS Tween. The humanized antibody at a concentration of 0.02 .mu.g/ml is incubated for 1 h at room temperature with a concentration range (starting from 1 .mu.g/ml) of peptides A.beta.1-28 (Bachem, reference H7865), A.beta.1-16 (Anaspec, reference 24225), A.beta.25-35 (Anaspec, reference 24227), low-molecular-weight oligomers or protofibrils prepared as described previously. The antibody/antigen mixture is then deposited in each well and the microtitration plate is incubated for 1 h at room temperature. The free, uncomplexed antibody is determined according to the same ELISA protocol as described previously. These competitive experiments show that only the protofibrils with a much higher affinity than the low-molecular-weight oligomers are capable of neutralizing the humanized antibody by preventing it from interacting with the peptide A.beta.1-42; none of the peptides is capable of neutralizing the antibody.
Example 4: Specificity of the Humanized Antibody LP09027 with Respect to the Fibrils of A.beta.1-42
[0112] The peptide A.beta.1-42 (Anaspec, 20276) is dissolved in 200 .mu.l of 10 mM NaOH to a concentration of 5 mg/ml. The peptide IAPP (Anaspec, 60804) is diluted in 200 .mu.l of 50% DMSO to a concentration of 5 mg/ml. 100 .mu.l of each preparation is diluted in 400 .mu.l of PBS 1.25.times.. The final concentration of the peptides is 1 mg/ml in 500 .mu.l. The samples are incubated for 72 h at 37.degree. C. After incubation, the samples are centrifuged at 17900 g for 30 minutes at 4.degree. C. The supernatant is removed and the pellet is washed 3 times with PBS 1.times.. After the last washing, the pellet of fibrils is taken up in 150 .mu.l of PBS. To check for the presence of fibrils of amyloid type, a thioflavin T fluorescence test (Anaspec, 88306) is carried out. 20 .mu.l of thioflavin T (20 .mu.M final), 10 .mu.l of the sample and 70 .mu.l of PBS 1.times. (final volume 100 .mu.l) are mixed in a well of a black plate (Corning, 3792). The thioflavin T is excited at 450 nm and, in the presence of a structure of amyloid type, emits fluorescence at 482 nm. 50 .mu.l of fibrils of A.beta.1-42 at 1 .mu.g/ml and IAPP at 0.5 .mu.g/ml are deposited in each well of a microtitration plate. The ELISA protocol is applied using serial dilutions of the humanized antibody starting from 10 .mu.g/ml. FIG. 5 shows that the humanized antibody LP09027 specifically recognizes the fibrils of A.beta.1-42 but not those of IAPP.
Example 5: Specificity of the Humanized Antibody LP09027 for the Mature Senile Plaques but not for the Diffuse Plaques
[0113] The humanized antibody (LP09027) conjugated with digoxigenin (digoxigenin-3-O-methylcarbonyl-.epsilon.-aminocaproic acid-N-hydroxysuccinimide ester: Roche 11333054001; 11418165001) was used in immunohistochemistry (Ventana Robot) on brain sections from mice APP PS1 (Alzheimer model described by Schmitz C. et al., Am. J. Pathol, 2004, 164, 1495-1502) as well as human brain sections (cerebral cortex) derived from patients with Alzheimer's disease. The samples had been fixed in formol and embedded in paraffin beforehand.
[0114] The results obtained in the mouse (FIGS. 6A and 6B) clearly show that the humanized antibody recognizes exclusively the dense, mature senile plaques, but not the diffuse deposits of peptide A.beta..
[0115] These data correlate with the properties of this antibody, which is specific for the protofibrillar Abeta form and so does not recognize the soluble, mono- or oligomeric forms of this peptide.
TABLE-US-00002 TABLE 2 Nucleotide sequences Protein sequences Antibody 1 VH1VL1 VH.sub.1 + CH.sub.1 SEQ ID NO 1 SEQ ID NO 2 VL.sub.1 + CL.sub.1 SEQ ID NO 3 SEQ ID NO 4 VH.sub.1 SEQ ID NO 5 SEQ ID NO 6 VL.sub.1 SEQ ID NO 7 SEQ ID NO 8 CDR VH.sub.1 SEQ ID NO 9, 11, 13 SEQ ID NO 10, 12, 14 CDR VL.sub.1 SEQ ID NO 15, 17, 19 SEQ ID NO 16, 18, 20 Antibody 2 VH1 VL2 VH.sub.1 + CH.sub.1 SEQ ID NO 1 SEQ ID NO 2 VL.sub.2 + CL.sub.2 SEQ ID NO 21 SEQ ID NO 22 VH.sub.1 SEQ ID NO 5 SEQ ID NO 6 VL.sub.2 SEQ ID NO 23 SEQ ID NO 24 CDR VH.sub.1 SEQ ID NO 9, 11, 13 SEQ ID NO 10, 12, 14 CDR VL.sub.2 SEQ ID NO 31, 17, 19 SEQ ID NO 32, 18, 20 Antibody 3 VH2 VL2 VH.sub.2 + CH.sub.2 SEQ ID NO 25 SEQ ID NO 26 VL.sub.2 + CL.sub.2 SEQ ID NO 21 SEQ ID NO 22 VH.sub.2 SEQ ID NO 27 SEQ ID NO 28 VL2 SEQ ID NO 23 SEQ ID NO 24 CDR VH.sub.2 SEQ ID NO 9, 11, 29 SEQ ID NO 10, 12, 30 CDR VL.sub.2 SEQ ID NO 31, 17, 19 SEQ ID NO 32, 18, 20
Sequence CWU 1
1
3211326DNAArtificial SequenceHumanized sequenceCDS(1)..(1326) 1gag
gtc cag ctg cag cag tct ggg cct gag gtg gtg aag cct ggg gtc 48Glu
Val Gln Leu Gln Gln Ser Gly Pro Glu Val Val Lys Pro Gly Val1 5 10
15tca gtg aag att tcc tgc aag ggt tcc ggc tac aca ttc act gat tat
96Ser Val Lys Ile Ser Cys Lys Gly Ser Gly Tyr Thr Phe Thr Asp Tyr
20 25 30gct atg cac tgg gtg aag cag agt cct ggc aag agt ctg gag tgg
att 144Ala Met His Trp Val Lys Gln Ser Pro Gly Lys Ser Leu Glu Trp
Ile 35 40 45gga gtt att agt act aag tat ggt aag aca aac tac aac ccc
agc ttt 192Gly Val Ile Ser Thr Lys Tyr Gly Lys Thr Asn Tyr Asn Pro
Ser Phe 50 55 60cag ggc cag gcc aca atg act gtt gac aaa tcc tcc agc
aca gcc tat 240Gln Gly Gln Ala Thr Met Thr Val Asp Lys Ser Ser Ser
Thr Ala Tyr65 70 75 80atg gag ctt gcc agc ttg aag gcc tcc gat tct
gcc atc tat tac tgt 288Met Glu Leu Ala Ser Leu Lys Ala Ser Asp Ser
Ala Ile Tyr Tyr Cys 85 90 95gca aga ggg gac gat ggt tat tcc tgg ggt
caa gga acc tca gtc acc 336Ala Arg Gly Asp Asp Gly Tyr Ser Trp Gly
Gln Gly Thr Ser Val Thr 100 105 110gtc tcc agc gct tct acc aag ggc
cct tcc gtg ttc cct ctg gcc cct 384Val Ser Ser Ala Ser Thr Lys Gly
Pro Ser Val Phe Pro Leu Ala Pro 115 120 125tgc tcc cgg tcc acc tcc
gag tcc acc gcc gct ctg ggc tgc ctg gtg 432Cys Ser Arg Ser Thr Ser
Glu Ser Thr Ala Ala Leu Gly Cys Leu Val 130 135 140aag gac tac ttc
cct gag cct gtg acc gtg tcc tgg aac tct ggc gcc 480Lys Asp Tyr Phe
Pro Glu Pro Val Thr Val Ser Trp Asn Ser Gly Ala145 150 155 160ctg
acc tcc ggc gtg cac acc ttc cct gcc gtg ctg cag tcc tcc ggc 528Leu
Thr Ser Gly Val His Thr Phe Pro Ala Val Leu Gln Ser Ser Gly 165 170
175ctg tac tcc ctg tcc tcc gtg gtg acc gtg cct tcc tcc tcc ctg ggc
576Leu Tyr Ser Leu Ser Ser Val Val Thr Val Pro Ser Ser Ser Leu Gly
180 185 190acc aag acc tac acc tgt aac gtg gac cac aag cct tcc aac
acc aag 624Thr Lys Thr Tyr Thr Cys Asn Val Asp His Lys Pro Ser Asn
Thr Lys 195 200 205gtg gac aag cgg gtg gag tcc aag tac ggc cct cct
tgc cct ccc tgc 672Val Asp Lys Arg Val Glu Ser Lys Tyr Gly Pro Pro
Cys Pro Pro Cys 210 215 220cct gcc cct gag ttc gag ggc gga cct agc
gtg ttc ctg ttc cct cct 720Pro Ala Pro Glu Phe Glu Gly Gly Pro Ser
Val Phe Leu Phe Pro Pro225 230 235 240aag cct aag gac acc ctg atg
atc tcc cgg acc cct gag gtg acc tgt 768Lys Pro Lys Asp Thr Leu Met
Ile Ser Arg Thr Pro Glu Val Thr Cys 245 250 255gtg gtg gtg gac gtg
tcc cag gag gac cct gag gtc cag ttc aac tgg 816Val Val Val Asp Val
Ser Gln Glu Asp Pro Glu Val Gln Phe Asn Trp 260 265 270tac gtg gac
ggc gtg gag gtg cac aac gcc aag acc aag cct cgg gag 864Tyr Val Asp
Gly Val Glu Val His Asn Ala Lys Thr Lys Pro Arg Glu 275 280 285gag
cag ttc aat tcc acc tac cgg gtg gtg tct gtg ctg acc gtg ctg 912Glu
Gln Phe Asn Ser Thr Tyr Arg Val Val Ser Val Leu Thr Val Leu 290 295
300cac cag gac tgg ctg aac ggc aaa gaa tac aag tgt aag gtc tcc aac
960His Gln Asp Trp Leu Asn Gly Lys Glu Tyr Lys Cys Lys Val Ser
Asn305 310 315 320aag ggc ctg ccc tcc tcc atc gag aaa acc atc tcc
aag gcc aag ggc 1008Lys Gly Leu Pro Ser Ser Ile Glu Lys Thr Ile Ser
Lys Ala Lys Gly 325 330 335cag cct agg gag cct cag gtg tac acc ctg
cct cct agc cag gaa gag 1056Gln Pro Arg Glu Pro Gln Val Tyr Thr Leu
Pro Pro Ser Gln Glu Glu 340 345 350atg acc aag aac cag gtg tcc ctg
acc tgt ctg gtg aag ggc ttc tac 1104Met Thr Lys Asn Gln Val Ser Leu
Thr Cys Leu Val Lys Gly Phe Tyr 355 360 365cct tcc gac atc gcc gtg
gag tgg gag tcc aac ggc cag cct gag aac 1152Pro Ser Asp Ile Ala Val
Glu Trp Glu Ser Asn Gly Gln Pro Glu Asn 370 375 380aac tac aag acc
acc cct cct gtg ctg gac tcc gac ggc tcc ttc ttc 1200Asn Tyr Lys Thr
Thr Pro Pro Val Leu Asp Ser Asp Gly Ser Phe Phe385 390 395 400ctg
tac tcc agg ctg acc gtg gac aag tcc cgg tgg cag gag ggc aac 1248Leu
Tyr Ser Arg Leu Thr Val Asp Lys Ser Arg Trp Gln Glu Gly Asn 405 410
415gtc ttt tcc tgc tcc gtg atg cac gag gcc ctg cac aac cac tac acc
1296Val Phe Ser Cys Ser Val Met His Glu Ala Leu His Asn His Tyr Thr
420 425 430cag aag tcc ctg tcc ctg tct ctg ggc tga 1326Gln Lys Ser
Leu Ser Leu Ser Leu Gly 435 4402441PRTArtificial SequenceSynthetic
Construct 2Glu Val Gln Leu Gln Gln Ser Gly Pro Glu Val Val Lys Pro
Gly Val1 5 10 15Ser Val Lys Ile Ser Cys Lys Gly Ser Gly Tyr Thr Phe
Thr Asp Tyr 20 25 30Ala Met His Trp Val Lys Gln Ser Pro Gly Lys Ser
Leu Glu Trp Ile 35 40 45Gly Val Ile Ser Thr Lys Tyr Gly Lys Thr Asn
Tyr Asn Pro Ser Phe 50 55 60Gln Gly Gln Ala Thr Met Thr Val Asp Lys
Ser Ser Ser Thr Ala Tyr65 70 75 80Met Glu Leu Ala Ser Leu Lys Ala
Ser Asp Ser Ala Ile Tyr Tyr Cys 85 90 95Ala Arg Gly Asp Asp Gly Tyr
Ser Trp Gly Gln Gly Thr Ser Val Thr 100 105 110Val Ser Ser Ala Ser
Thr Lys Gly Pro Ser Val Phe Pro Leu Ala Pro 115 120 125Cys Ser Arg
Ser Thr Ser Glu Ser Thr Ala Ala Leu Gly Cys Leu Val 130 135 140Lys
Asp Tyr Phe Pro Glu Pro Val Thr Val Ser Trp Asn Ser Gly Ala145 150
155 160Leu Thr Ser Gly Val His Thr Phe Pro Ala Val Leu Gln Ser Ser
Gly 165 170 175Leu Tyr Ser Leu Ser Ser Val Val Thr Val Pro Ser Ser
Ser Leu Gly 180 185 190Thr Lys Thr Tyr Thr Cys Asn Val Asp His Lys
Pro Ser Asn Thr Lys 195 200 205Val Asp Lys Arg Val Glu Ser Lys Tyr
Gly Pro Pro Cys Pro Pro Cys 210 215 220Pro Ala Pro Glu Phe Glu Gly
Gly Pro Ser Val Phe Leu Phe Pro Pro225 230 235 240Lys Pro Lys Asp
Thr Leu Met Ile Ser Arg Thr Pro Glu Val Thr Cys 245 250 255Val Val
Val Asp Val Ser Gln Glu Asp Pro Glu Val Gln Phe Asn Trp 260 265
270Tyr Val Asp Gly Val Glu Val His Asn Ala Lys Thr Lys Pro Arg Glu
275 280 285Glu Gln Phe Asn Ser Thr Tyr Arg Val Val Ser Val Leu Thr
Val Leu 290 295 300His Gln Asp Trp Leu Asn Gly Lys Glu Tyr Lys Cys
Lys Val Ser Asn305 310 315 320Lys Gly Leu Pro Ser Ser Ile Glu Lys
Thr Ile Ser Lys Ala Lys Gly 325 330 335Gln Pro Arg Glu Pro Gln Val
Tyr Thr Leu Pro Pro Ser Gln Glu Glu 340 345 350Met Thr Lys Asn Gln
Val Ser Leu Thr Cys Leu Val Lys Gly Phe Tyr 355 360 365Pro Ser Asp
Ile Ala Val Glu Trp Glu Ser Asn Gly Gln Pro Glu Asn 370 375 380Asn
Tyr Lys Thr Thr Pro Pro Val Leu Asp Ser Asp Gly Ser Phe Phe385 390
395 400Leu Tyr Ser Arg Leu Thr Val Asp Lys Ser Arg Trp Gln Glu Gly
Asn 405 410 415Val Phe Ser Cys Ser Val Met His Glu Ala Leu His Asn
His Tyr Thr 420 425 430Gln Lys Ser Leu Ser Leu Ser Leu Gly 435
4403660DNAArtificial SequenceHumanized sequenceCDS(1)..(660) 3gag
atc gtg atg acc caa act cca ctc tcc ctg cct gtc agt ctt gga 48Glu
Ile Val Met Thr Gln Thr Pro Leu Ser Leu Pro Val Ser Leu Gly1 5 10
15gat aga gcc tcc atc tct tgc aga tct ggt cag agc ctt gtg cac agt
96Asp Arg Ala Ser Ile Ser Cys Arg Ser Gly Gln Ser Leu Val His Ser
20 25 30aat gga aac acc tat ctg cat tgg tac ctg cag aag cca ggc cag
tct 144Asn Gly Asn Thr Tyr Leu His Trp Tyr Leu Gln Lys Pro Gly Gln
Ser 35 40 45cca aag ctc ctg atc tat aca gtt tcc aac cga ttt tct ggg
gtc ccg 192Pro Lys Leu Leu Ile Tyr Thr Val Ser Asn Arg Phe Ser Gly
Val Pro 50 55 60gac agg ttc agt ggc agt gga tca ggg tca gat ttc aca
ctc acc atc 240Asp Arg Phe Ser Gly Ser Gly Ser Gly Ser Asp Phe Thr
Leu Thr Ile65 70 75 80agc aga gtg gag gct gag gat ctg gga gtt tat
ttc tgc tct caa aat 288Ser Arg Val Glu Ala Glu Asp Leu Gly Val Tyr
Phe Cys Ser Gln Asn 85 90 95aca ttt gtt cct tgg acg ttc ggt gga ggc
acc aag ctg gaa atc aaa 336Thr Phe Val Pro Trp Thr Phe Gly Gly Gly
Thr Lys Leu Glu Ile Lys 100 105 110cgt acg gtg gct gca cca tct gtc
ttc atc ttc ccg cca tct gat gag 384Arg Thr Val Ala Ala Pro Ser Val
Phe Ile Phe Pro Pro Ser Asp Glu 115 120 125cag ttg aaa tct gga act
gcc tct gtt gtg tgc ctg ctg aat aac ttc 432Gln Leu Lys Ser Gly Thr
Ala Ser Val Val Cys Leu Leu Asn Asn Phe 130 135 140tat ccc aga gag
gcc aaa gta cag tgg aag gtg gat aac gcc ctc caa 480Tyr Pro Arg Glu
Ala Lys Val Gln Trp Lys Val Asp Asn Ala Leu Gln145 150 155 160tcg
ggt aac tcc cag gag agt gtc aca gag cag gac agc aag gac agc 528Ser
Gly Asn Ser Gln Glu Ser Val Thr Glu Gln Asp Ser Lys Asp Ser 165 170
175acc tac agc ctc agc agc acc ctg acg ctg agc aaa gca gac tac gag
576Thr Tyr Ser Leu Ser Ser Thr Leu Thr Leu Ser Lys Ala Asp Tyr Glu
180 185 190aaa cac aaa gtc tac gcc tgc gaa gtc acc cat cag ggc ctg
agc tcg 624Lys His Lys Val Tyr Ala Cys Glu Val Thr His Gln Gly Leu
Ser Ser 195 200 205ccc gtc aca aag agc ttc aac agg gga gag tgt tga
660Pro Val Thr Lys Ser Phe Asn Arg Gly Glu Cys 210
2154219PRTArtificial SequenceSynthetic Construct 4Glu Ile Val Met
Thr Gln Thr Pro Leu Ser Leu Pro Val Ser Leu Gly1 5 10 15Asp Arg Ala
Ser Ile Ser Cys Arg Ser Gly Gln Ser Leu Val His Ser 20 25 30Asn Gly
Asn Thr Tyr Leu His Trp Tyr Leu Gln Lys Pro Gly Gln Ser 35 40 45Pro
Lys Leu Leu Ile Tyr Thr Val Ser Asn Arg Phe Ser Gly Val Pro 50 55
60Asp Arg Phe Ser Gly Ser Gly Ser Gly Ser Asp Phe Thr Leu Thr Ile65
70 75 80Ser Arg Val Glu Ala Glu Asp Leu Gly Val Tyr Phe Cys Ser Gln
Asn 85 90 95Thr Phe Val Pro Trp Thr Phe Gly Gly Gly Thr Lys Leu Glu
Ile Lys 100 105 110Arg Thr Val Ala Ala Pro Ser Val Phe Ile Phe Pro
Pro Ser Asp Glu 115 120 125Gln Leu Lys Ser Gly Thr Ala Ser Val Val
Cys Leu Leu Asn Asn Phe 130 135 140Tyr Pro Arg Glu Ala Lys Val Gln
Trp Lys Val Asp Asn Ala Leu Gln145 150 155 160Ser Gly Asn Ser Gln
Glu Ser Val Thr Glu Gln Asp Ser Lys Asp Ser 165 170 175Thr Tyr Ser
Leu Ser Ser Thr Leu Thr Leu Ser Lys Ala Asp Tyr Glu 180 185 190Lys
His Lys Val Tyr Ala Cys Glu Val Thr His Gln Gly Leu Ser Ser 195 200
205Pro Val Thr Lys Ser Phe Asn Arg Gly Glu Cys 210
2155345DNAArtificial SequenceHumanized sequenceCDS(1)..(345) 5gag
gtc cag ctg cag cag tct ggg cct gag gtg gtg aag cct ggg gtc 48Glu
Val Gln Leu Gln Gln Ser Gly Pro Glu Val Val Lys Pro Gly Val1 5 10
15tca gtg aag att tcc tgc aag ggt tcc ggc tac aca ttc act gat tat
96Ser Val Lys Ile Ser Cys Lys Gly Ser Gly Tyr Thr Phe Thr Asp Tyr
20 25 30gct atg cac tgg gtg aag cag agt cct ggc aag agt ctg gag tgg
att 144Ala Met His Trp Val Lys Gln Ser Pro Gly Lys Ser Leu Glu Trp
Ile 35 40 45gga gtt att agt act aag tat ggt aag aca aac tac aac ccc
agc ttt 192Gly Val Ile Ser Thr Lys Tyr Gly Lys Thr Asn Tyr Asn Pro
Ser Phe 50 55 60cag ggc cag gcc aca atg act gtt gac aaa tcc tcc agc
aca gcc tat 240Gln Gly Gln Ala Thr Met Thr Val Asp Lys Ser Ser Ser
Thr Ala Tyr65 70 75 80atg gag ctt gcc agc ttg aag gcc tcc gat tct
gcc atc tat tac tgt 288Met Glu Leu Ala Ser Leu Lys Ala Ser Asp Ser
Ala Ile Tyr Tyr Cys 85 90 95gca aga ggg gac gat ggt tat tcc tgg ggt
caa gga acc tca gtc acc 336Ala Arg Gly Asp Asp Gly Tyr Ser Trp Gly
Gln Gly Thr Ser Val Thr 100 105 110gtc tcc agc 345Val Ser Ser
1156115PRTArtificial SequenceSynthetic Construct 6Glu Val Gln Leu
Gln Gln Ser Gly Pro Glu Val Val Lys Pro Gly Val1 5 10 15Ser Val Lys
Ile Ser Cys Lys Gly Ser Gly Tyr Thr Phe Thr Asp Tyr 20 25 30Ala Met
His Trp Val Lys Gln Ser Pro Gly Lys Ser Leu Glu Trp Ile 35 40 45Gly
Val Ile Ser Thr Lys Tyr Gly Lys Thr Asn Tyr Asn Pro Ser Phe 50 55
60Gln Gly Gln Ala Thr Met Thr Val Asp Lys Ser Ser Ser Thr Ala Tyr65
70 75 80Met Glu Leu Ala Ser Leu Lys Ala Ser Asp Ser Ala Ile Tyr Tyr
Cys 85 90 95Ala Arg Gly Asp Asp Gly Tyr Ser Trp Gly Gln Gly Thr Ser
Val Thr 100 105 110Val Ser Ser 1157339DNAArtificial
SequenceHumanized sequenceCDS(1)..(339) 7gag atc gtg atg acc caa
act cca ctc tcc ctg cct gtc agt ctt gga 48Glu Ile Val Met Thr Gln
Thr Pro Leu Ser Leu Pro Val Ser Leu Gly1 5 10 15gat aga gcc tcc atc
tct tgc aga tct ggt cag agc ctt gtg cac agt 96Asp Arg Ala Ser Ile
Ser Cys Arg Ser Gly Gln Ser Leu Val His Ser 20 25 30aat gga aac acc
tat ctg cat tgg tac ctg cag aag cca ggc cag tct 144Asn Gly Asn Thr
Tyr Leu His Trp Tyr Leu Gln Lys Pro Gly Gln Ser 35 40 45cca aag ctc
ctg atc tat aca gtt tcc aac cga ttt tct ggg gtc ccg 192Pro Lys Leu
Leu Ile Tyr Thr Val Ser Asn Arg Phe Ser Gly Val Pro 50 55 60gac agg
ttc agt ggc agt gga tca ggg tca gat ttc aca ctc acc atc 240Asp Arg
Phe Ser Gly Ser Gly Ser Gly Ser Asp Phe Thr Leu Thr Ile65 70 75
80agc aga gtg gag gct gag gat ctg gga gtt tat ttc tgc tct caa aat
288Ser Arg Val Glu Ala Glu Asp Leu Gly Val Tyr Phe Cys Ser Gln Asn
85 90 95aca ttt gtt cct tgg acg ttc ggt gga ggc acc aag ctg gaa atc
aaa 336Thr Phe Val Pro Trp Thr Phe Gly Gly Gly Thr Lys Leu Glu Ile
Lys 100 105 110cgt 339Arg8113PRTArtificial SequenceSynthetic
Construct 8Glu Ile Val Met Thr Gln Thr Pro Leu Ser Leu Pro Val Ser
Leu Gly1 5 10 15Asp Arg Ala Ser Ile Ser Cys Arg Ser Gly Gln Ser Leu
Val His Ser 20 25 30Asn Gly Asn Thr Tyr Leu His Trp Tyr Leu Gln Lys
Pro Gly Gln Ser 35 40 45Pro Lys Leu Leu Ile Tyr Thr Val Ser Asn Arg
Phe Ser Gly Val Pro 50 55 60Asp Arg Phe Ser Gly Ser Gly Ser Gly Ser
Asp Phe Thr Leu Thr Ile65 70 75 80Ser Arg Val Glu Ala Glu Asp Leu
Gly Val Tyr Phe Cys Ser Gln Asn 85 90 95Thr Phe Val Pro Trp Thr Phe
Gly Gly Gly Thr Lys Leu Glu Ile Lys 100 105 110Arg933DNAMus
musculusCDS(1)..(33) 9tcc ggc tac aca ttc act gat tat gct atg cac
33Ser Gly Tyr Thr Phe Thr Asp Tyr Ala Met His1 5 101011PRTMus
musculus 10Ser Gly Tyr Thr Phe Thr Asp Tyr Ala Met His1 5
101130DNAMus musculusCDS(1)..(30) 11gtt att agt act aag tat ggt aag
aca aac 30Val Ile Ser Thr Lys Tyr Gly Lys Thr Asn1 5 101210PRTMus
musculus 12Val Ile Ser Thr Lys Tyr Gly Lys Thr Asn1 5
101318DNAMus
musculusCDS(1)..(18) 13ggg gac gat ggt tat tcc 18Gly Asp Asp Gly
Tyr Ser1 5146PRTMus musculus 14Gly Asp Asp Gly Tyr Ser1 51548DNAMus
musculusCDS(1)..(48) 15aga tct ggt cag agc ctt gtg cac agt aat gga
aac acc tat ctg cat 48Arg Ser Gly Gln Ser Leu Val His Ser Asn Gly
Asn Thr Tyr Leu His1 5 10 151616PRTMus musculus 16Arg Ser Gly Gln
Ser Leu Val His Ser Asn Gly Asn Thr Tyr Leu His1 5 10 151724DNAMus
musculusCDS(1)..(24) 17aca gtt tcc aac cga ttt tct ggg 24Thr Val
Ser Asn Arg Phe Ser Gly1 5188PRTMus musculus 18Thr Val Ser Asn Arg
Phe Ser Gly1 51927DNAMus musculusCDS(1)..(27) 19tct caa aat aca ttt
gtt cct tgg acg 27Ser Gln Asn Thr Phe Val Pro Trp Thr1 5209PRTMus
musculus 20Ser Gln Asn Thr Phe Val Pro Trp Thr1 521660DNAArtificial
SequenceHumanized sequenceCDS(1)..(660) 21gag atc gtg atg acc caa
act cca ctc tcc ctg cct gtc agt ctt gga 48Glu Ile Val Met Thr Gln
Thr Pro Leu Ser Leu Pro Val Ser Leu Gly1 5 10 15gat aga gcc tcc atc
tct tgc aga tct ggt cag agc ctt gtg cac agt 96Asp Arg Ala Ser Ile
Ser Cys Arg Ser Gly Gln Ser Leu Val His Ser 20 25 30aat acc aac acc
tat ctg cat tgg tac ctg cag aag cca ggc cag tct 144Asn Thr Asn Thr
Tyr Leu His Trp Tyr Leu Gln Lys Pro Gly Gln Ser 35 40 45cca aag ctc
ctg atc tat aca gtt tcc aac cga ttt tct ggg gtc ccg 192Pro Lys Leu
Leu Ile Tyr Thr Val Ser Asn Arg Phe Ser Gly Val Pro 50 55 60gac agg
ttc agt ggc agt gga tca ggg tca gat ttc aca ctc acc atc 240Asp Arg
Phe Ser Gly Ser Gly Ser Gly Ser Asp Phe Thr Leu Thr Ile65 70 75
80agc aga gtg gag gct gag gat ctg gga gtt tat ttc tgc tct caa aat
288Ser Arg Val Glu Ala Glu Asp Leu Gly Val Tyr Phe Cys Ser Gln Asn
85 90 95aca ttt gtt cct tgg acg ttc ggt gga ggc acc aag ctg gaa atc
aaa 336Thr Phe Val Pro Trp Thr Phe Gly Gly Gly Thr Lys Leu Glu Ile
Lys 100 105 110cgt acg gtg gct gca cca tct gtc ttc atc ttc ccg cca
tct gat gag 384Arg Thr Val Ala Ala Pro Ser Val Phe Ile Phe Pro Pro
Ser Asp Glu 115 120 125cag ttg aaa tct gga act gcc tct gtt gtg tgc
ctg ctg aat aac ttc 432Gln Leu Lys Ser Gly Thr Ala Ser Val Val Cys
Leu Leu Asn Asn Phe 130 135 140tat ccc aga gag gcc aaa gta cag tgg
aag gtg gat aac gcc ctc caa 480Tyr Pro Arg Glu Ala Lys Val Gln Trp
Lys Val Asp Asn Ala Leu Gln145 150 155 160tcg ggt aac tcc cag gag
agt gtc aca gag cag gac agc aag gac agc 528Ser Gly Asn Ser Gln Glu
Ser Val Thr Glu Gln Asp Ser Lys Asp Ser 165 170 175acc tac agc ctc
agc agc acc ctg acg ctg agc aaa gca gac tac gag 576Thr Tyr Ser Leu
Ser Ser Thr Leu Thr Leu Ser Lys Ala Asp Tyr Glu 180 185 190aaa cac
aaa gtc tac gcc tgc gaa gtc acc cat cag ggc ctg agc tcg 624Lys His
Lys Val Tyr Ala Cys Glu Val Thr His Gln Gly Leu Ser Ser 195 200
205ccc gtc aca aag agc ttc aac agg gga gag tgt tga 660Pro Val Thr
Lys Ser Phe Asn Arg Gly Glu Cys 210 21522219PRTArtificial
SequenceSynthetic Construct 22Glu Ile Val Met Thr Gln Thr Pro Leu
Ser Leu Pro Val Ser Leu Gly1 5 10 15Asp Arg Ala Ser Ile Ser Cys Arg
Ser Gly Gln Ser Leu Val His Ser 20 25 30Asn Thr Asn Thr Tyr Leu His
Trp Tyr Leu Gln Lys Pro Gly Gln Ser 35 40 45Pro Lys Leu Leu Ile Tyr
Thr Val Ser Asn Arg Phe Ser Gly Val Pro 50 55 60Asp Arg Phe Ser Gly
Ser Gly Ser Gly Ser Asp Phe Thr Leu Thr Ile65 70 75 80Ser Arg Val
Glu Ala Glu Asp Leu Gly Val Tyr Phe Cys Ser Gln Asn 85 90 95Thr Phe
Val Pro Trp Thr Phe Gly Gly Gly Thr Lys Leu Glu Ile Lys 100 105
110Arg Thr Val Ala Ala Pro Ser Val Phe Ile Phe Pro Pro Ser Asp Glu
115 120 125Gln Leu Lys Ser Gly Thr Ala Ser Val Val Cys Leu Leu Asn
Asn Phe 130 135 140Tyr Pro Arg Glu Ala Lys Val Gln Trp Lys Val Asp
Asn Ala Leu Gln145 150 155 160Ser Gly Asn Ser Gln Glu Ser Val Thr
Glu Gln Asp Ser Lys Asp Ser 165 170 175Thr Tyr Ser Leu Ser Ser Thr
Leu Thr Leu Ser Lys Ala Asp Tyr Glu 180 185 190Lys His Lys Val Tyr
Ala Cys Glu Val Thr His Gln Gly Leu Ser Ser 195 200 205Pro Val Thr
Lys Ser Phe Asn Arg Gly Glu Cys 210 21523339DNAArtificial
SequenceHumanized sequenceCDS(1)..(339) 23gag atc gtg atg acc caa
act cca ctc tcc ctg cct gtc agt ctt gga 48Glu Ile Val Met Thr Gln
Thr Pro Leu Ser Leu Pro Val Ser Leu Gly1 5 10 15gat aga gcc tcc atc
tct tgc aga tct ggt cag agc ctt gtg cac agt 96Asp Arg Ala Ser Ile
Ser Cys Arg Ser Gly Gln Ser Leu Val His Ser 20 25 30aat acc aac acc
tat ctg cat tgg tac ctg cag aag cca ggc cag tct 144Asn Thr Asn Thr
Tyr Leu His Trp Tyr Leu Gln Lys Pro Gly Gln Ser 35 40 45cca aag ctc
ctg atc tat aca gtt tcc aac cga ttt tct ggg gtc ccg 192Pro Lys Leu
Leu Ile Tyr Thr Val Ser Asn Arg Phe Ser Gly Val Pro 50 55 60gac agg
ttc agt ggc agt gga tca ggg tca gat ttc aca ctc acc atc 240Asp Arg
Phe Ser Gly Ser Gly Ser Gly Ser Asp Phe Thr Leu Thr Ile65 70 75
80agc aga gtg gag gct gag gat ctg gga gtt tat ttc tgc tct caa aat
288Ser Arg Val Glu Ala Glu Asp Leu Gly Val Tyr Phe Cys Ser Gln Asn
85 90 95aca ttt gtt cct tgg acg ttc ggt gga ggc acc aag ctg gaa atc
aaa 336Thr Phe Val Pro Trp Thr Phe Gly Gly Gly Thr Lys Leu Glu Ile
Lys 100 105 110cgt 339Arg24113PRTArtificial SequenceSynthetic
Construct 24Glu Ile Val Met Thr Gln Thr Pro Leu Ser Leu Pro Val Ser
Leu Gly1 5 10 15Asp Arg Ala Ser Ile Ser Cys Arg Ser Gly Gln Ser Leu
Val His Ser 20 25 30Asn Thr Asn Thr Tyr Leu His Trp Tyr Leu Gln Lys
Pro Gly Gln Ser 35 40 45Pro Lys Leu Leu Ile Tyr Thr Val Ser Asn Arg
Phe Ser Gly Val Pro 50 55 60Asp Arg Phe Ser Gly Ser Gly Ser Gly Ser
Asp Phe Thr Leu Thr Ile65 70 75 80Ser Arg Val Glu Ala Glu Asp Leu
Gly Val Tyr Phe Cys Ser Gln Asn 85 90 95Thr Phe Val Pro Trp Thr Phe
Gly Gly Gly Thr Lys Leu Glu Ile Lys 100 105
110Arg251326DNAArtificial SequenceHumanized sequenceCDS(1)..(1326)
25gag gtc cag ctg cag cag tct ggg cct gag gtg gtg aag cct ggg gtc
48Glu Val Gln Leu Gln Gln Ser Gly Pro Glu Val Val Lys Pro Gly Val1
5 10 15tca gtg aag att tcc tgc aag ggt tcc ggc tac aca ttc act gat
tat 96Ser Val Lys Ile Ser Cys Lys Gly Ser Gly Tyr Thr Phe Thr Asp
Tyr 20 25 30gct atg cac tgg gtg aag cag agt cct ggc aag agt ctg gag
tgg att 144Ala Met His Trp Val Lys Gln Ser Pro Gly Lys Ser Leu Glu
Trp Ile 35 40 45gga gtt att agt act aag tat ggt aag aca aac tac aac
ccc agc ttt 192Gly Val Ile Ser Thr Lys Tyr Gly Lys Thr Asn Tyr Asn
Pro Ser Phe 50 55 60cag ggc cag gcc aca atg act gtt gac aaa tcc tcc
agc aca gcc tat 240Gln Gly Gln Ala Thr Met Thr Val Asp Lys Ser Ser
Ser Thr Ala Tyr65 70 75 80atg gag ctt gcc agc ttg aag gcc tcc gat
tct gcc atc tat tac tgt 288Met Glu Leu Ala Ser Leu Lys Ala Ser Asp
Ser Ala Ile Tyr Tyr Cys 85 90 95gca aga ggg gac gag ggt tat tcc tgg
ggt caa gga acc tca gtc acc 336Ala Arg Gly Asp Glu Gly Tyr Ser Trp
Gly Gln Gly Thr Ser Val Thr 100 105 110gtc tcc agc gct tct acc aag
ggc cct tcc gtg ttc cct ctg gcc cct 384Val Ser Ser Ala Ser Thr Lys
Gly Pro Ser Val Phe Pro Leu Ala Pro 115 120 125tgc tcc cgg tcc acc
tcc gag tcc acc gcc gct ctg ggc tgc ctg gtg 432Cys Ser Arg Ser Thr
Ser Glu Ser Thr Ala Ala Leu Gly Cys Leu Val 130 135 140aag gac tac
ttc cct gag cct gtg acc gtg tcc tgg aac tct ggc gcc 480Lys Asp Tyr
Phe Pro Glu Pro Val Thr Val Ser Trp Asn Ser Gly Ala145 150 155
160ctg acc tcc ggc gtg cac acc ttc cct gcc gtg ctg cag tcc tcc ggc
528Leu Thr Ser Gly Val His Thr Phe Pro Ala Val Leu Gln Ser Ser Gly
165 170 175ctg tac tcc ctg tcc tcc gtg gtg acc gtg cct tcc tcc tcc
ctg ggc 576Leu Tyr Ser Leu Ser Ser Val Val Thr Val Pro Ser Ser Ser
Leu Gly 180 185 190acc aag acc tac acc tgt aac gtg gac cac aag cct
tcc aac acc aag 624Thr Lys Thr Tyr Thr Cys Asn Val Asp His Lys Pro
Ser Asn Thr Lys 195 200 205gtg gac aag cgg gtg gag tcc aag tac ggc
cct cct tgc cct ccc tgc 672Val Asp Lys Arg Val Glu Ser Lys Tyr Gly
Pro Pro Cys Pro Pro Cys 210 215 220cct gcc cct gag ttc gag ggc gga
cct agc gtg ttc ctg ttc cct cct 720Pro Ala Pro Glu Phe Glu Gly Gly
Pro Ser Val Phe Leu Phe Pro Pro225 230 235 240aag cct aag gac acc
ctg atg atc tcc cgg acc cct gag gtg acc tgt 768Lys Pro Lys Asp Thr
Leu Met Ile Ser Arg Thr Pro Glu Val Thr Cys 245 250 255gtg gtg gtg
gac gtg tcc cag gag gac cct gag gtc cag ttc aac tgg 816Val Val Val
Asp Val Ser Gln Glu Asp Pro Glu Val Gln Phe Asn Trp 260 265 270tac
gtg gac ggc gtg gag gtg cac aac gcc aag acc aag cct cgg gag 864Tyr
Val Asp Gly Val Glu Val His Asn Ala Lys Thr Lys Pro Arg Glu 275 280
285gag cag ttc aat tcc acc tac cgg gtg gtg tct gtg ctg acc gtg ctg
912Glu Gln Phe Asn Ser Thr Tyr Arg Val Val Ser Val Leu Thr Val Leu
290 295 300cac cag gac tgg ctg aac ggc aaa gaa tac aag tgt aag gtc
tcc aac 960His Gln Asp Trp Leu Asn Gly Lys Glu Tyr Lys Cys Lys Val
Ser Asn305 310 315 320aag ggc ctg ccc tcc tcc atc gag aaa acc atc
tcc aag gcc aag ggc 1008Lys Gly Leu Pro Ser Ser Ile Glu Lys Thr Ile
Ser Lys Ala Lys Gly 325 330 335cag cct agg gag cct cag gtg tac acc
ctg cct cct agc cag gaa gag 1056Gln Pro Arg Glu Pro Gln Val Tyr Thr
Leu Pro Pro Ser Gln Glu Glu 340 345 350atg acc aag aac cag gtg tcc
ctg acc tgt ctg gtg aag ggc ttc tac 1104Met Thr Lys Asn Gln Val Ser
Leu Thr Cys Leu Val Lys Gly Phe Tyr 355 360 365cct tcc gac atc gcc
gtg gag tgg gag tcc aac ggc cag cct gag aac 1152Pro Ser Asp Ile Ala
Val Glu Trp Glu Ser Asn Gly Gln Pro Glu Asn 370 375 380aac tac aag
acc acc cct cct gtg ctg gac tcc gac ggc tcc ttc ttc 1200Asn Tyr Lys
Thr Thr Pro Pro Val Leu Asp Ser Asp Gly Ser Phe Phe385 390 395
400ctg tac tcc agg ctg acc gtg gac aag tcc cgg tgg cag gag ggc aac
1248Leu Tyr Ser Arg Leu Thr Val Asp Lys Ser Arg Trp Gln Glu Gly Asn
405 410 415gtc ttt tcc tgc tcc gtg atg cac gag gcc ctg cac aac cac
tac acc 1296Val Phe Ser Cys Ser Val Met His Glu Ala Leu His Asn His
Tyr Thr 420 425 430cag aag tcc ctg tcc ctg tct ctg ggc tga 1326Gln
Lys Ser Leu Ser Leu Ser Leu Gly 435 44026441PRTArtificial
SequenceSynthetic Construct 26Glu Val Gln Leu Gln Gln Ser Gly Pro
Glu Val Val Lys Pro Gly Val1 5 10 15Ser Val Lys Ile Ser Cys Lys Gly
Ser Gly Tyr Thr Phe Thr Asp Tyr 20 25 30Ala Met His Trp Val Lys Gln
Ser Pro Gly Lys Ser Leu Glu Trp Ile 35 40 45Gly Val Ile Ser Thr Lys
Tyr Gly Lys Thr Asn Tyr Asn Pro Ser Phe 50 55 60Gln Gly Gln Ala Thr
Met Thr Val Asp Lys Ser Ser Ser Thr Ala Tyr65 70 75 80Met Glu Leu
Ala Ser Leu Lys Ala Ser Asp Ser Ala Ile Tyr Tyr Cys 85 90 95Ala Arg
Gly Asp Glu Gly Tyr Ser Trp Gly Gln Gly Thr Ser Val Thr 100 105
110Val Ser Ser Ala Ser Thr Lys Gly Pro Ser Val Phe Pro Leu Ala Pro
115 120 125Cys Ser Arg Ser Thr Ser Glu Ser Thr Ala Ala Leu Gly Cys
Leu Val 130 135 140Lys Asp Tyr Phe Pro Glu Pro Val Thr Val Ser Trp
Asn Ser Gly Ala145 150 155 160Leu Thr Ser Gly Val His Thr Phe Pro
Ala Val Leu Gln Ser Ser Gly 165 170 175Leu Tyr Ser Leu Ser Ser Val
Val Thr Val Pro Ser Ser Ser Leu Gly 180 185 190Thr Lys Thr Tyr Thr
Cys Asn Val Asp His Lys Pro Ser Asn Thr Lys 195 200 205Val Asp Lys
Arg Val Glu Ser Lys Tyr Gly Pro Pro Cys Pro Pro Cys 210 215 220Pro
Ala Pro Glu Phe Glu Gly Gly Pro Ser Val Phe Leu Phe Pro Pro225 230
235 240Lys Pro Lys Asp Thr Leu Met Ile Ser Arg Thr Pro Glu Val Thr
Cys 245 250 255Val Val Val Asp Val Ser Gln Glu Asp Pro Glu Val Gln
Phe Asn Trp 260 265 270Tyr Val Asp Gly Val Glu Val His Asn Ala Lys
Thr Lys Pro Arg Glu 275 280 285Glu Gln Phe Asn Ser Thr Tyr Arg Val
Val Ser Val Leu Thr Val Leu 290 295 300His Gln Asp Trp Leu Asn Gly
Lys Glu Tyr Lys Cys Lys Val Ser Asn305 310 315 320Lys Gly Leu Pro
Ser Ser Ile Glu Lys Thr Ile Ser Lys Ala Lys Gly 325 330 335Gln Pro
Arg Glu Pro Gln Val Tyr Thr Leu Pro Pro Ser Gln Glu Glu 340 345
350Met Thr Lys Asn Gln Val Ser Leu Thr Cys Leu Val Lys Gly Phe Tyr
355 360 365Pro Ser Asp Ile Ala Val Glu Trp Glu Ser Asn Gly Gln Pro
Glu Asn 370 375 380Asn Tyr Lys Thr Thr Pro Pro Val Leu Asp Ser Asp
Gly Ser Phe Phe385 390 395 400Leu Tyr Ser Arg Leu Thr Val Asp Lys
Ser Arg Trp Gln Glu Gly Asn 405 410 415Val Phe Ser Cys Ser Val Met
His Glu Ala Leu His Asn His Tyr Thr 420 425 430Gln Lys Ser Leu Ser
Leu Ser Leu Gly 435 44027345DNAArtificial SequenceHumanized
sequenceCDS(1)..(345) 27gag gtc cag ctg cag cag tct ggg cct gag gtg
gtg aag cct ggg gtc 48Glu Val Gln Leu Gln Gln Ser Gly Pro Glu Val
Val Lys Pro Gly Val1 5 10 15tca gtg aag att tcc tgc aag ggt tcc ggc
tac aca ttc act gat tat 96Ser Val Lys Ile Ser Cys Lys Gly Ser Gly
Tyr Thr Phe Thr Asp Tyr 20 25 30gct atg cac tgg gtg aag cag agt cct
ggc aag agt ctg gag tgg att 144Ala Met His Trp Val Lys Gln Ser Pro
Gly Lys Ser Leu Glu Trp Ile 35 40 45gga gtt att agt act aag tat ggt
aag aca aac tac aac ccc agc ttt 192Gly Val Ile Ser Thr Lys Tyr Gly
Lys Thr Asn Tyr Asn Pro Ser Phe 50 55 60cag ggc cag gcc aca atg act
gtt gac aaa tcc tcc agc aca gcc tat 240Gln Gly Gln Ala Thr Met Thr
Val Asp Lys Ser Ser Ser Thr Ala Tyr65 70 75 80atg gag ctt gcc agc
ttg aag gcc tcc gat tct gcc atc tat tac tgt 288Met Glu Leu Ala Ser
Leu Lys Ala Ser Asp Ser Ala Ile Tyr Tyr Cys 85 90 95gca aga ggg gac
gag ggt tat tcc tgg ggt caa gga acc tca gtc acc 336Ala Arg Gly Asp
Glu Gly Tyr Ser Trp Gly Gln Gly Thr Ser Val Thr 100 105 110gtc tcc
agc 345Val Ser Ser 11528115PRTArtificial SequenceSynthetic
Construct 28Glu Val Gln Leu Gln Gln Ser Gly Pro Glu Val Val Lys Pro
Gly Val1 5 10 15Ser Val Lys Ile Ser Cys Lys Gly Ser Gly Tyr Thr Phe
Thr Asp Tyr 20 25 30Ala Met His Trp Val Lys Gln Ser Pro Gly Lys Ser
Leu Glu Trp Ile 35 40 45Gly Val Ile Ser Thr
Lys Tyr Gly Lys Thr Asn Tyr Asn Pro Ser Phe 50 55 60Gln Gly Gln Ala
Thr Met Thr Val Asp Lys Ser Ser Ser Thr Ala Tyr65 70 75 80Met Glu
Leu Ala Ser Leu Lys Ala Ser Asp Ser Ala Ile Tyr Tyr Cys 85 90 95Ala
Arg Gly Asp Glu Gly Tyr Ser Trp Gly Gln Gly Thr Ser Val Thr 100 105
110Val Ser Ser 1152918DNAMus musculusCDS(1)..(18) 29ggg gac gag ggt
tat tcc 18Gly Asp Glu Gly Tyr Ser1 5306PRTMus musculus 30Gly Asp
Glu Gly Tyr Ser1 53148DNAMus musculusCDS(1)..(48) 31aga tct ggt cag
agc ctt gtg cac agt aat acc aac acc tat ctg cat 48Arg Ser Gly Gln
Ser Leu Val His Ser Asn Thr Asn Thr Tyr Leu His1 5 10 153216PRTMus
musculus 32Arg Ser Gly Gln Ser Leu Val His Ser Asn Thr Asn Thr Tyr
Leu His1 5 10 15
D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

S00001
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.