Assistive Capnography Device
Al-Ali; Ammar
U.S. patent application number 16/222853 was filed with the patent office on 2019-04-25 for assistive capnography device. The applicant listed for this patent is MASIMO CORPORATION. Invention is credited to Ammar Al-Ali.
| Application Number | 20190117930 16/222853 |
| Document ID | / |
| Family ID | 53881221 |
| Filed Date | 2019-04-25 |











View All Diagrams
| United States Patent Application | 20190117930 |
| Kind Code | A1 |
| Al-Ali; Ammar | April 25, 2019 |
ASSISTIVE CAPNOGRAPHY DEVICE
Abstract
Systems and method for monitoring patient physiological data are presented herein. A gas analyzing measurement head can be provided to sample and analyze respiratory gases of a patient. In one embodiment, the gas analyzing measurement head can read information on an information element of an airway adapter or resuscitation bag. Such information can be used to generate instructions for manual ventilation using the gas analyzing measurement head, airway adapter, and resuscitation bag. Manual ventilation instructions can be displayed on the gas analyzing measurement head or can be transmitted for display on another device, such as a clinician's mobile computing device.
| Inventors: | Al-Ali; Ammar; (San Juan Capistrano, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 53881221 | ||||||||||
| Appl. No.: | 16/222853 | ||||||||||
| Filed: | December 17, 2018 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 14627500 | Feb 20, 2015 | |||
| 16222853 | ||||
| 61943263 | Feb 21, 2014 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61B 2560/0443 20130101; A61B 5/0836 20130101; A61B 5/097 20130101; A61M 16/1005 20140204; A61B 5/082 20130101; A61M 16/04 20130101; A61M 16/208 20130101; A61M 2230/432 20130101; A61M 16/0051 20130101; A61M 16/021 20170801; A61M 2205/35 20130101; A61B 2505/01 20130101; A61M 16/06 20130101; A61M 16/085 20140204; A61M 16/0003 20140204; A61M 16/01 20130101; A61B 5/7445 20130101; A61M 16/0084 20140204; A61B 5/4836 20130101; A61M 2205/505 20130101; A61M 2016/103 20130101 |
| International Class: | A61M 16/10 20060101 A61M016/10; A61M 16/00 20060101 A61M016/00; A61M 16/08 20060101 A61M016/08; A61B 5/083 20060101 A61B005/083 |
Claims
1.-24. (canceled)
25. (canceled)
26. A physiological monitor including an electronic display device, which provides respiration guidance to assist in timing respiration events, the physiological monitor comprising: at least one sensor configured to measure information responsive to at least one physiological parameter; at least one processor configured to provide guidance for respiration event sequences; and a display configured to provide a visual indication of the guidance, the visual indication comprising a graphic that expands and contracts to indicate a timing of a desired respiratory sequence.
27. The physiological monitor of claim 26, wherein the display device is a smartphone.
28. The physiological monitor of claim 26, further comprising a first housing for the sensor and a second housing for the display.
29. The physiological monitor of claim 26, wherein the graphic comprises a plurality of circles.
30. The physiological monitor of claim 26, wherein the at least one processor is further configured to detect a time of performance of the respiration event sequences; and generate feedback for the user based on a predetermined rate and the detected time of performance.
31. The physiological monitor of claim 30, wherein the predetermined rate is based on reminder settings corresponding to certain time or cycle.
32. The physiological monitor of claim 26, wherein the respiration event sequences correspond to cardiopulmonary resuscitation (CPR).
33. The physiological monitor of claim 26, wherein the physiological parameter comprises respiration rate and wherein the at least one processor is configured to determine that the respiration rate of the user is outside of threshold readings and select guidance based on the determination.
34. The physiological monitor of claim 26, wherein the guidance includes a rate of conducting the respiration event sequences and wherein the at least one processor is further configured to generate stimulatory feedback to guide the user to follow the rate.
35. The physiological monitor of claim 34, wherein the at least one processor is configured to update the rate based on the determined physiological parameter.
36. The physiological monitor of claim 34, wherein the stimulatory feedback includes a vibrational feedback.
37. The physiological monitor of claim 26, wherein the expansion and contraction is correlated with breathing of the user.
38. The physiological monitor of claim 26, wherein the at least one processor is configured to display heart rate after the performance of the respiration event sequences.
39. A method of providing respiration guidance to assist in timing respiration events, the method comprising: measuring, with at least one sensor, information responsive to at least one physiological parameter; generating guidance for respiration event sequences; and displaying a visual indication of the guidance, the visual indication comprising a graphic that expands and contracts to indicate volume of desired breathing in respiratory sequence.
Description
INCORPORATION BY REFERENCE TO ANY PRIORITY APPLICATIONS
[0001] The present application claims priority benefit under 35 U.S.C. .sctn. 119(e) to U.S. Provisional Application No. 61/943,263, titled "DIGITAL INSTRUCTIONS IN CAPNOGRAPHY DEVICE," filed Feb. 21, 2014.
[0002] The present application also claims priority to any and all applications for which a foreign or domestic priority claim is identified in the Application Data Sheet as filed with the present application, including without limitation the above-mentioned provisional application, are hereby incorporated by reference in their entirety under 37 C.F,R, .sctn. 1.57 and for all purposes.
FIELD OF THE DISCLOSURE
[0003] The invention relates generally to systems, devices, and methods for monitoring a patient's respiratory system and health, including capnographers and other devices that monitor patient respiratory gases. In particular, this disclosure relates to respiratory gas measuring devices capable of recognizing attached airway adapters and resuscitation bags and providing assistance for manual ventilation.
BACKGROUND
[0004] In respiratory care, it is often desirable to analyze and monitor the gas composition of a patient's exhaled and/or inhaled breathing gases. Various requirements for gas analyses exist in health care. For instance, measurement of respiratory CO.sub.2, O.sub.2, N.sub.2O and anesthetic agents, such as halothane, isoflurane, enflurane, sevoflurane or desflurane, is useful in the care of critically ill patients undergoing anesthesia or mechanical ventilation, while for emergency care such as manual ventilation it is typically sufficient to monitor breathing of a patient with a simple CO.sub.2 analysis.
[0005] Respiratory gases can be analyzed in accordance with many different measuring principles. The most common method of respiratory gas analysis, however, is through the medium of non-dispersive spectroscopy. This measuring principle is based on the fact that many gases absorb infrared energy at a wavelength specific to the substance concerned. Main flow gas analyzers based on non-dispersive spectroscopy measure light absorption at specific wavelengths directly in the patient's respiratory circuit. Capnography is the monitoring of the concentration or partial pressure of CO.sub.2 in respiratory gases, and provides real-time information regarding CO.sub.2 exhalation and respiratory rates as well as a rapid and reliable assessment of a patient's ventilatory, circulatory and metabolic function. Although the terms capnography and capnometry are sometimes considered synonymous, capnometry suggests measurement without a continuous written record or waveform. Typically in capnography and capnometry, a main flow measuring head is placed as close as possible to the patient's mouth or trachea to sample exhaled and/or inhaled breathing gases and calculate gas concentrations directly in the respiratory circuit of the patient.
[0006] Measurement of end tidal CO.sub.2 can also provide useful information such as regarding CO.sub.2 production, pulmonary (lung) perfusion, alveolar ventilation, respiratory patterns, and elimination of CO.sub.2 from an anesthesia breathing circuit or ventilator. The gas sample measured at the end of a person's exhalation is called the "end-tidal" gas sample. The amount of carbon dioxide in a person's breath can indicate the overall efficiency of the cardio-pulmonary system and quality of breathing. For example, the concentration of carbon dioxide can indicate shallow breathing and poor oxygen intake. Thus, capnographers are used in hospitals and other medical institutions for monitoring the condition of a patient's respiratory system, pulmonary perfusion, and metabolism, and are most often used for patients in intensive care and under anesthesia.
[0007] In many clinical and emergency settings, respiratory assistance is accomplished through use of bag-valve mask (BVM) ventilation systems. Main flow measuring heads can be useful for implementation in BVM ventilation systems and other manual ventilation systems to measure end tidal respiratory gases during respiratory assistance. BVM ventilation is a life-saving skill of an emergency physician or pre-hospital care provider that can easily be overlooked because of its apparent simplicity. However, BVM ventilation is a difficult skill to master, and poor BVM ventilation technique can lead to the need for more invasive means of airway management and their inherent complications. Implementing BVM ventilation with a low rate of bag compression can lead to hypoventilation and inadequate oxygen supply to the patient. Hyperventilation due to overzealous BVM ventilation can be harmful by increasing intra-thoracic pressure, which decreases venous blood to the heart and subsequently decreases cerebral and coronary perfusion pressures. The appropriate rate of bag compression for proper patient oxygenation differs based on factors such as the age of the patient and the size of the bag. Therefore, there is a need for measuring heads that are capable of providing instructions and feedback to manual ventilation providers.
SUMMARY
[0008] Advantageously, in certain embodiments, a physiological monitoring system can be designed to include a respiratory gas measurement head with a processing board or card as well as an airway adapter and resuscitation bag each including an information element that can identify the airway adapter and resuscitation bag to the measurement head. For example, an airway adapter information element can identify the airway adapter to the measurement head as an adult or infant adapter, and a resuscitation bag information element can identify a volume of the resuscitation bag to the measurement head. The system may be connectable to a mobile computing device, such as a smartphone, such that display of the instructions for manual ventilation based on monitored physiological data may occur on the computing device. The board or card may communicate the instructions and data for display with the mobile computing device wirelessly or through a physical and electrical connection with the cable assembly. Alternatively, the measurement head can include a display to provide instructions to the care giver.
[0009] Physiological monitoring systems such as are described herein advantageously enable adaptive display of manual ventilation instructions to a medical care provider. This improves patient care and provides a higher likelihood of a positive outcome for the patient. For instance, upon or after assembly of an airway adapter and resuscitation bag to a capnographic measurement head, the measurement head can identify a type of the airway adapter and a volume of the resuscitation bag. In one example, a processor of the measurement head can perform the identification by communicating with an information element located on one or both of the airway adapter and the resuscitation bag. The identification of the adapter and ventilation bag can provide many useful parameters. For example, the parameters can include, for example, the type of patient, such as an adult patient or an infant patient, the volume of the bag, the length of the airway adapter and any other useful parameters helpful in determining proper operation of the manual ventilation system. As a result of being able to identify the airway adapter and resuscitation bag, the measurement head provides appropriate instructions for the parameters of the manual ventilation system.
[0010] The present disclosure allows a medical care provider to receive real time (or near real time) feedback regarding their manual ventilation efforts through analysis of the patient's physiological data such as end tidal gas values taking into account the characteristics of the adapter airway and resuscitation bag. To illustrate, a resuscitation bag that is identified as having a high volume should be compressed more slowly and/or less frequently than a resuscitation bag identified as having a small volume in order for appropriate oxygen delivery to the patient. The present disclosure allows the ventilation system to provide feedback to pace the caregiver's efforts. Further, for an individual who is untrained in manual ventilation but is called to perform such techniques in an emergency setting, the present system provides critical manual ventilation instructions based on real time monitored conditions of the patient.
[0011] For purposes of summarizing the disclosure, certain aspects, advantages and novel features of the inventions have been described herein. It is to be understood that not necessarily all such advantages can be achieved in accordance with any particular embodiment of the inventions disclosed herein. Thus, the inventions disclosed herein can be embodied or carried out in a manner that achieves or optimizes one advantage or group of advantages as taught herein without necessarily achieving other advantages as can be taught or suggested herein.
BRIEF DESCRIPTION OF THE DRAWINGS
[0012] Throughout the drawings, reference numbers can be re-used to indicate correspondence between referenced elements. The drawings are provided to illustrate embodiments of the inventions described herein and not to limit the scope thereof.
[0013] FIG. 1A illustrates an embodiment of a physiological monitoring system.
[0014] FIG. 1B illustrates an embodiment of a measuring head.
[0015] FIG. 1C illustrates an embodiment of an airway adapter.
[0016] FIG. 1D illustrates another embodiment of an airway adapter.
[0017] FIGS. 2A-2C illustrate various embodiments of a ventilation assembly.
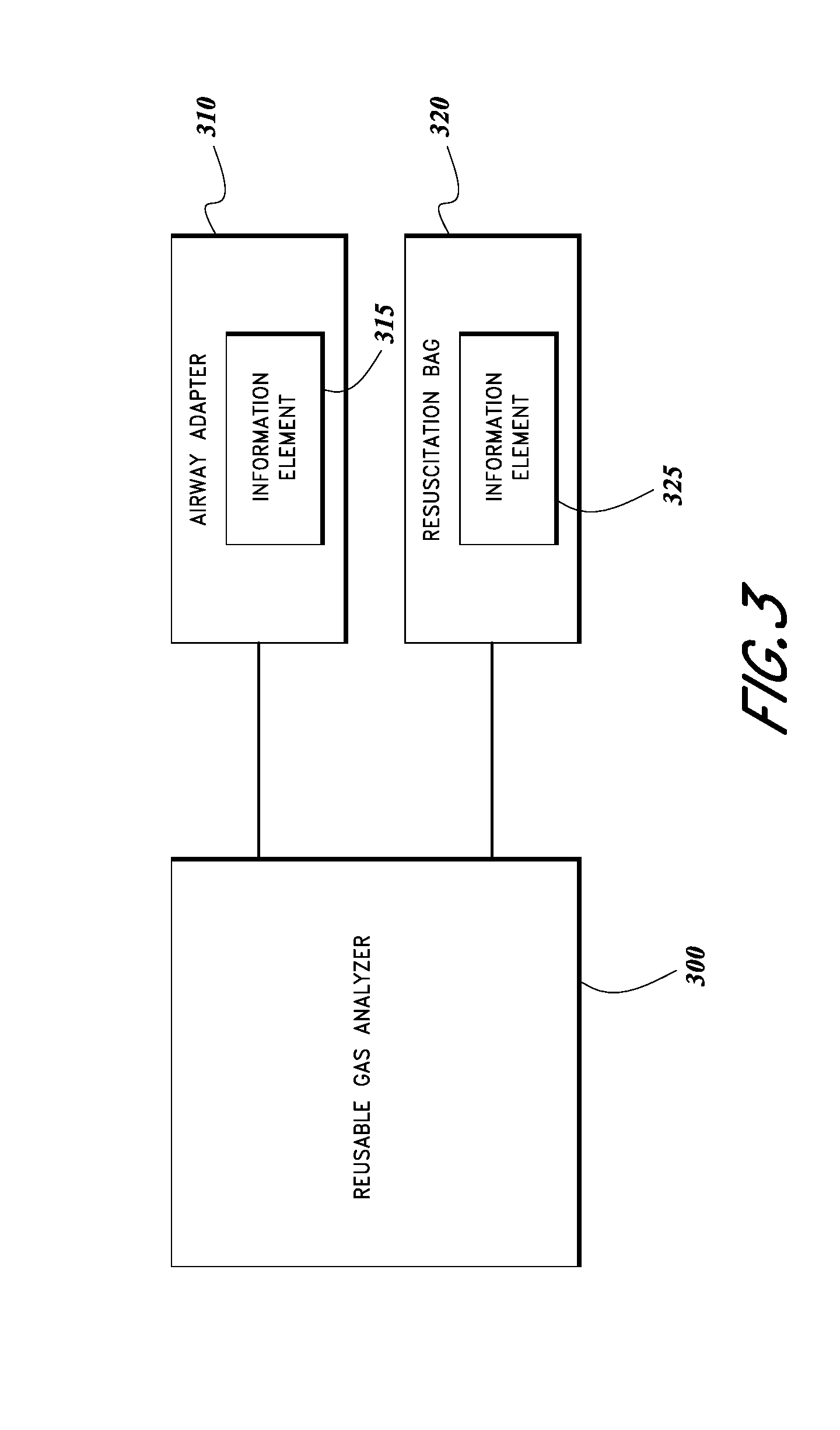
[0018] FIG. 3 illustrates a schematic block diagram of one embodiment of a physiological monitoring system.
[0019] FIG. 4A illustrates an embodiment of a software application for display of ventilation instructions.
[0020] FIG. 4B illustrates another embodiment of a software application for display of ventilation instructions.
[0021] FIGS. 5A-5D illustrate various embodiments of display interfaces on a measuring head.
[0022] FIG. 6 illustrates an embodiment of a manual ventilation kit.
[0023] FIG. 7 illustrates an embodiment of a manual ventilation process.
[0024] FIG. 8 illustrates an embodiment of a process for providing ventilation instructions.
DETAILED DESCRIPTION
I. Example Physiological Monitoring Systems
[0025] FIGS. 1A-1D illustrate embodiments of a physiological monitoring system 100. The physiological monitoring system 100 shown in FIG. 1A includes a capnographic measurement head 105 and an airway adapter 120. The measurement head 105 can include a display 110 having an indication 115 regarding when instructions are available and a connection port 120 for insertion of the airway adapter 130. FIG. 1B illustrates an example of the measurement head 105 illustrated without an airway adapter in place. FIG. 1C illustrates an example of an airway adapter 130a that can be used in adult or pediatric ventilation assistance, the airway adapter 130a includes an information element 135. FIG. 1D illustrates an example of an airway adapter 130b that can be used in infant or neonate ventilation assistance, the airway adapter 130b has an information element 135.
[0026] The physiological monitoring system 100 can be used to monitor physiological parameters such as end tidal respiratory gases including oxygen (O.sub.2), carbon dioxide (CO.sub.2), and nitrous oxide (N.sub.2O), among others, as well as patient respiratory rate. A system capable of measuring these parameters is commercially available from Masimo Corporation of Irvine, Calif., marketed under the name EMMA'. The physiological monitoring system 100 can also measure anesthetic agents and perform agent identification in some examples. The physiological monitoring system 100 can be used for proof of intubation (that is, to show that an endotracheal tube has been correctly placed in the trachea, and not the esophagus, of a critically ill patient), short-term CO.sub.2 monitoring for ventilation during emergency patient transport, CO.sub.2 monitoring during cardiopulmonary resuscitation (CPR), among other uses.
[0027] The measurement head 105 can be configured to analyze respiration rate and concentration of gases in a patient's exhaled respiratory gases, among other things. Some embodiments of the measurement head 105 can be compact, portable for flexible use at multiple points of care including pre-hospital, emergency medicine, operating rooms, intensive care units, and long-term acute care. The measurement head 105 can be provided with display 110 for illustrating monitored physiological parameters to a clinician. Although end tidal CO.sub.2 and respiratory rate in breaths per minute are illustrated in the example display 110, other embodiments can analyze and display these and/or the additional parameters discussed above. In addition, in some embodiments the measurement head 105 can be configured to communicate with an external display. In some embodiments, the display 110 may be omitted. Display 110 also includes an indicator 115 to highlight to a clinician when usage instructions are available for a connected airway adapter and/or resuscitation bag, as will be discussed in more detail below. The measurement head 105 can also be provided with visual and/or audible alarms, a capnograph waveform display, and various user interface features such as a power button. Some embodiments of the measurement head 105 may be battery operated and contain a power source housing.
[0028] As in the illustrated embodiment, the measurement head 105 can be a mainstream capnometer or capnographer placed directly into a patient's airway or coupled to an airway adapter 130 placed in the patient's airway. The measurement head 105 can house an infrared light source and photodetector in some embodiments, such as a non-dispersive infrared gas analyzer. The measurement head 105 can be, in other embodiments, a sidestream capnometer or capnographer sampling a patient's respiratory gases through a tube or lumen from the patient's airway to the measurement head 105.
[0029] The airway adapter 130 can be provided for insertion into or placement adjacent the patient's mouth. The airway adapter 130 is configured to transfer or guide gases exhaled from the patient to the measuring head 105. In some embodiments, the airway adapter 130 can be about 1 inch, about 2 inches, about 4 inches, or about six inches long. In other embodiments, the airway adapter 130 can be no longer than about 5 inches. As illustrated in FIGS. 1C and 1D, the airway adapter can have different configurations for patients of different ages. For example an airway adapter such as airway adapter 130a can be used in adult or pediatric ventilation assistance, while an airway adapter such as airway adapter 130b can be used in infant or neonate ventilation assistance. In some embodiments, the airway adapter 130 can be replaceable and/or disposable while the measurement head 105 is reusable. The airway adapter 130 may have a hydrophilic inner surface to create a film of water condensed from a patient's exhaled breath such that the film does not scatter an infrared beam used to measure gas concentrations. The inner surface can also be etched in some embodiments to further control the formation of condensation within the airway adapter 130.
[0030] The airway adapter 130 can be placed into connection port 120 in the measuring head 105 to provide fluid communication between a patient's respiratory circuit and a measuring chamber of the measurement head 105. Exhaled respiratory gases can pass into the airway adapter 130 through a breathing mask or a sampling line, and the exhaled gases can pass through an output opening in a side of the airway adapter into a measuring chamber of the measurement head 105. In one example, the measuring chamber of the measurement head 105 can be compressed to a size of 50 .mu.l to provide accurate measurements under extreme conditions, such as for young patients with very high breathing rates, delivering approximately a 50 mL/min sampling flow that can accommodate respiratory monitoring for patients of a wide range of ages, from adults to neonates.
[0031] The measurement head 105 can be configured to measure a physiological parameter of the patient by analyzing the respiratory gases in the measuring chamber. In one implementation of gas analysis, the light from an infrared emitter can pass through the gas mixture in the measuring chamber and the light can be filtered by a narrow-band optical band-pass filter. The gases can absorb the infrared light at known, gas-specific wavelengths during passage of the light through the gas mixture. The partially absorbed light can be detected by an infrared detector and the intensity of the detected light can be determined, for example by a processor of the measurement head 105 or by the processor of another computing device. By measuring the intensity of the light that was not absorbed into the gas mixture, a quantification of the concentration of a gas or gases in the gas mixture can be obtained. In this manner, an embodiment of the measurement head 105 can analyze an unknown gas mixture and identify which gases and/or agents are present in the mixture.
[0032] Accordingly, the measurement head 105 can include an emitter, an optical filter, and at least one sensor (not illustrated) for conducting analysis of gas concentrations in respiratory gas samples received from the airway adapter 130. The emitter, filter, and sensor can be positioned in the measuring chamber within the measurement head 105, for example near the output opening in the airway adapter. An emitter can be configured to emit light at one or more wavelengths into a measuring chamber containing a gas sample, and a sensor can be configured to detect the emitted light. An optical filter (not illustrated) can be included in the measurement head 105. For example, the filter can be a narrow band optical filter, and can be manufactured using a film deposition process that can balance out film layer thickness variations created by changes in temperature in order to reduce center wavelength drift with temperature.
[0033] A sensor, such as an infrared detector, can be included in the measurement head 105 to receive the light emitted by the emitter. In one embodiment, the sensor or sensors can be a spectrometer used to detect slight changes in infrared radiation to precisely determine gas concentrations in a mixture by measuring absorption caused by molecules in the gas sample. In some embodiments, the spectrometer can detect changes in multiple wavelengths of light, for example at nine different wavelengths in the long-wavelength infrared (LWIR) spectrum. The LWIR wavelength band contains strong absorption peaks for CO2, N2O, and various anesthetic agents, with negligible interference from alcohol, acetone, and other gases and vapors that could potentially degrade measurement accuracy. In another embodiment, the sensor(s) can be carbon dioxide sensors, for example nanotechnology carbon dioxide sensors, nanoelectric sensors, pyroelectric detectors, thermopile detectors, or infrared sensors. In multigas monitoring embodiments of the measurement head 105, the sensor(s) can be configured to trace gas sample compositions across multiple pre-selected narrow band optical filters. In some embodiments multiple sensors may be mounted in a thermally stable array, for example a block of aluminum.
[0034] The measurement head 105 can also include a processor (not illustrated) generally including circuitry that can process the physiological parameter signal(s) generated by the sensor(s) prior to display or storage of the parameters. The processor can include instructions to process analog pressure, temperature, and flow signals combined with data from the sensor(s). The processor can analyze the data received from the sensor(s) and determine a physiological parameter or parameters of the patient. For example, the processor can include any of a variety of front-end signal conditioners, such as filters, amplifiers, buffers, memories and/or analog-to-digital (A/D) converters known to those of skill in the art. The processor can extract one or more optical filter signals from the sensor data and can filter the signal(s) to remove noise in some embodiments, such as high and low frequency noise. The processor can analyze the sensor data and/or filtered data to determine gas and agent identification and measurement. In one example, the processor can be a 32-bit RISC microprocessor. In another example, the processor can be a 41-MIPS RISC DSP and can provide power to a spectrometer of the measurement head 105. The processor can be designed to be compact and power-efficient in some embodiments. The processor can be a digital signal processor (DSP) or analog processor or combination of both.
[0035] The processor can also include instructions to communicate with the information element 135 of an airway adapter 130, the information element of a resuscitation bag, and/or the information element of another respiratory assistance component. Such information elements can be placed at any location on the airway adapter or resuscitation bag that can be in electrical contact with the measurement head 105, and the measurement head 105 can have corresponding reading element(s) for contacting the information element(s). Though discussed primarily in the context of airway adapters and resuscitation bags, such information elements can be provided on any ventilation assistance component such as gas sampling lines and breathing masks, to name a few other examples.
[0036] In some embodiments, the processor of the measuring head 105 can read data stored on an information element upon or after physical and electrical connection to the corresponding reading element. The processor can use the data to identify an attached ventilation assistance component. The information element 133 can be an active circuit such as a transistor network, memory chip, EEPROM (electronically erasable programmable read-only memory), EPROM (erasable programmable read-only memory), or other identification device, such as multi-contact single wire memory devices or other devices, such as those commercially available from Dallas Semiconductor or the like. The information element can be, in some embodiments, a resistor, a capacitor, a microchip, a RAM, a ROM, or any other information storage element. In addition, the information element can include a combination of one or more of any of the above.
[0037] In other embodiments, the processor and information element may communicate wirelessly. For example, radio communications can be used to identify an attached ventilation assistance component and/or its characteristics. In one embodiment, radio-frequency identification (RFID) can enable communication between an information element and the processor. A passive RFID tag can be included in or on an airway adapter and/or resuscitation bag, the tag containing electronically stored information. The RFID tag can act as a passive transponder to emit radio waves that can be detected by an active reader element associated with the processor of the measurement head. In another embodiment, near field communication (NFC) technology can enable communications between an unpowered NFC chip on an airway adapter or resuscitation bag and an NFC reading component communicating with the measurement head processor. As another example, the measurement head can be equipped with an optical scanning means for scanning a barcode, matrix barcode, or other optical machine-readable representation of data on an airway adapter or resuscitation bag. In some embodiments, various ventilation assistance components can implement the same or different communication means as discussed above.
[0038] An information element can store information specific to the corresponding ventilation accessory component. For example, an airway adapter information element 135 can include data to identify an intended age group of the airway adapter, for example by identifying the airway adapter as one of an adult/pediatric airway adapter 130a or an infant/neonate airway adapter 130b. Such information can be used by the processor to provide age-specific ventilation instructions to a clinician. Though two examples of airway adapters are illustrated in FIGS. 1C and 1D, other sizes of airway adapters are possible in other embodiments. As another example, a resuscitation bag information element can include data to identify characteristics of the resuscitation bag such as volume, compressive resistance, or the like. Such information can be used by the processor to provide instructions regarding compression rate and compression depth, as well as other manual ventilation techniques. Such information elements can also be used to store instructions specific to the component to which they are attached.
[0039] In addition, such information elements can store information about the use of the airway adapter or resuscitation bag in order to prevent overuse or reuse. During use, airway adapters and resuscitation bags collect condensation from a patient's exhaled respiratory gases. As such, it can be unsanitary to reuse such components from patient to patient. Accordingly, an information element can store data indicating that the component has already been attached to a measurement head, and therefore has presumably been used. In some embodiments, the measurement head may not perform measurements when a used component is detected and/or may output an indication to replace the component with a new component.
[0040] The instructions can enable clinicians to assess the effectiveness of cardiopulmonary resuscitation (CPR) and can guide manual ventilation. For example, the measurement head 105 can use the physiological data to determine whether adequate ventilation is occurring, and the measurement head 105 can use communications with a resuscitation bag information element to provide feedback on the depth and effectiveness of compressions of the resuscitation bag. As another example, the measurement head 105 can use communications with an airway adapter information element 135 to provide ventilation instructions appropriate for the patient's age group based on whether the airway adapter is an adult airway adapter 130a or an infant airway adapter 130b.
[0041] Some embodiments of display 110 can be sized and configured to display the instructions on the measurement head 105. In other embodiments the processor may communicate the instructions to an external display, such as a medical terminal or a clinician's mobile device. The instructions can be a graphical representation of compression rate and compression depth in one example. As another example, an auditory signal can be provided to guide compression rate and compression depth. Indications of the quality of manual ventilation technique can be provided including an alarm, an icon, or a color that generally represents the quality of a measured physiological parameter. The instructions can include, in some embodiments, instructions for proper assembly of the physiological monitoring system 100 and/or proper placement on a patient. The measurement head 105 can update or alter the instructions during the course of manual ventilation assistance based at least partly on the physiological data of the patient in some embodiments.
[0042] The system 200a as illustrated in FIG. 2A shows a measuring head 205 coupled to an airway adapter 210, with the airway adapter 210 coupled to a breathing mask 215. The system 200a can be placed over a patient's mouth for safe delivery of rescue breaths during CPR, for example during cardiac arrest or respiratory arrest. The system 200a can also be attached to other manual or mechanical ventilation components.
[0043] The system 200b as illustrated in FIG. 2B shows a measuring head 205 coupled to an airway adapter 210, with the airway adapter 210 coupled to a breathing mask 215, similar to the system 200a of FIG. 2A. The system 200b also includes a resuscitation bag 225 coupled to the airway adapter 210 for operation by a care provider 240. The breathing mask 215 of system 200b is illustrated placed over a patient's 230 mouth for delivery of manual ventilation therapy. A care provider must ensure that the mask is substantially sealed around the patient's face such that pressure needed to force-inflate the lungs is not released into the environment. Though illustrated with a mask 215 over a patient's mouth, other implementations of the system 200b can be adapter for connection to an endotracheal tube or laryngeal mask airway.
[0044] System 200b can be used to provide positive pressure ventilation to patients who are not breathing or are not breathing adequately without assistance. The resuscitation bag 225 acts as a flexible air chamber that, when squeezed, forces air through a one-way valve into the patient's lungs. When released, the resuscitation bag 225 self-inflates through the end not coupled to the airway adapter 210, drawing in either ambient air or an oxygen flow supplied by a regulated cylinder, while also allowing the patient's lungs to deflate to the ambient environment.
[0045] The system 200b can be available in different sizes to fit infants, children, and adults in some embodiments. The sizes of the face mask 215, airway adapter 210, and bag 225 may vary independent of one another. For example, a pediatric sized bag might be used with different masks for multiple face sizes, or a pediatric mask might be used with an adult sized bag for patients with smaller faces. In order to be effective, a bag valve mask must generally deliver between 500 and 800 ml of air to a normal male adult patient's lungs, however if supplemental oxygen is provided 400 ml may still be adequate. This amount can vary for females, children, and infants. Generally, squeezing the bag once every 5-6 seconds for an adult or once every 3 seconds for an infant or child can provide adequate respiratory rate (determined as 10-12 respirations per minute in an adult and 20 per minute in a child or infant). As such, based on the patient as well as the size of the face mask 215, airway adapter 210, and bag 225, a care provider performing the manual ventilation is required to use different compression rates and depths of compression in order to provide suitable ventilation assistance. The presently described device can provide feedback to a user based on current patient parameters and device parameters in order to assist a user to provide optimal manual ventilation to a patient.
[0046] FIG. 3 illustrates a schematic block diagram of one embodiment of a physiological monitoring system including a reusable gas analyzer 300, an airway adapter 310, and a resuscitation bag 320. The airway adapter 310 includes an information element 315 and the resuscitation bag 320 includes an information element 325. As discussed above, the information element 315 can serve to identify characteristics of the airway adapter 310 to the reusable gas analyzer 300, and the information element 325 can serve to identify characteristics of the resuscitation bag 320 to the reusable gas analyzer 300.
[0047] Information element 315 can store data identifying characteristics of the airway adapter 310. For example, the data can include a size of the airway adapter 310 such as adult, child, infant, or neonatal. The data can also include a type of the airway adapter such as an airway adapter designed to connect to a breathing mask or an airway adapter designed to connect to an endotracheal tube. In some embodiments, the data can include information regarding a manufacturer of the airway adapter 310. This data can be used to determine whether to recommend instructions to a care provider and/or which instructions to recommend to a care provider. For example, an airway adapter 310 from a known manufacturer may be associated with a specific set of instructions, while an airway adapter 310 from an unknown manufacturer may not be associated with instructions. As another example, an infant airway adapter may be associated with instructions for a more rapid rate of compression relative to the instructions associated with an adult airway adapter. Information element 315 can store data representing instructions associated with use of the airway adapter 310 in some embodiments. In an embodiment, the information can store a formula or algorithm that the measurement head can use in addition to other data to determine proper compression rates. For example, the information from the airway adapter can be combined with end tidal CO.sub.2 to determine an adjusted compression rate depending on the patient's responsiveness.
[0048] Information element 325 can store data identifying characteristics of the resuscitation bag 320. For example, the data can include a size or volume of the resuscitation bag 320 corresponding to an adult, child, infant, or neonatal patient's lung volume and respiratory needs. Adult bags, in some embodiments, can deliver volumes of 240-2,000 ml of room air or oxygen with each compression. Child and infant bags can be designed to deliver smaller volumes of room air per compression. In some embodiments, the data can include information regarding a manufacturer of the resuscitation bag 320. This data can be used to determine whether to recommend instructions to a care provider and/or which instructions to recommend to a care provider. For example, a resuscitation bag 320 from a known manufacturer may be associated with a specific set of instructions, while a resuscitation bag 320 from an unknown manufacturer may not be associated with instructions. As another example, an infant resuscitation bag may be associated with instructions for a more rapid rate of compression relative to the instructions associated with an adult resuscitation bag. Compression rate and depth instructions can also be based on a comparison of a volume of the resuscitation bag as communicated by the information element 325 and an age or size of the patient as input by a care provider. Information element 325 can store data representing instructions associated with use of the resuscitation bag 320 in some embodiments.
[0049] Information elements 315, 325 can be provided through a circuit that contacts a corresponding portion in the reusable gas analyzer 300, for example a transistor network, memory chip, EEPROM (electronically erasable programmable read-only memory), EPROM (erasable programmable read-only memory), multi-contact single wire memory device, a resistor, a capacitor, a microchip, a RAM, or a ROM, or any other information storage element. Information elements 315, 325 can also be provided through a wireless communication means such as RFID or NFC, or through optical scanning technology. Information elements 315, 325 can be configured using the same or different technologies. For example, in some embodiments information element 315 of the airway adapter 310 may be in physical contact with the reusable gas analyzer 300, while information element 325 of the resuscitation bag 320 may communicate wirelessly with the reusable gas analyzer 300.
[0050] The reusable gas analyzer 300 may communicate with the information elements 315, 325 once in some embodiments, for example upon connection of the airway adapter 310 and resuscitation bag 320, or when the information elements 315, 325 are in wireless communication range. In some embodiments the reusable gas analyzer 300 may communicate with the information elements 315, 325 when powered on. In other embodiments reusable gas analyzer 300 may communicate with the information elements 315, 325 at various points during ventilation therapy.
[0051] FIG. 4A illustrates an embodiment of an application for display and management of manual ventilation instructions 405 and also, in some embodiments, physiological monitoring data. Some embodiments of the software application may be used with a mobile computing device of a clinician, illustrated here as smartphone 400. Although specific reference may be made to smartphones in this disclosure, persons skilled in the art will understand that a mobile computing device compatible with the physiological sensor system may be one of a wide range of mobile devices such as a laptop, tablet computer, netbook, PDA, media player, mobile game console, stationary or portable medical terminal, or other microprocessor based device configured to interface with a physiological sensor. Some embodiments of the mobile computing device may be used with the system for display of instructions and/or data as well as storage of data. Cables used to connect smartphone 400 to a reusable gas analyzer (for example, measurement head 105 of FIG. 1 discussed above) can be flex cables or other cables, including cables having triboelectric properties, and some devices may be configured to connect wirelessly to the reusable gas analyzer.
[0052] Smartphone 400 may include a display screen such as an LED or LCD screen, and may include touch sensitive technologies in combination with the display screen. Smartphone 400 may include software configured to display manual ventilation instructions 405 as well as some or all of the output measurement data on the display screen. The instructions can include steps for proper assembly or placement of a ventilation assistance system and/or steps for operation of the ventilation assistance system. The operation steps can be based on data read from information elements on components of the ventilation assistance system, as discussed above. A measurement data display may comprise numerical or graphical representations of end tidal O.sub.2, CO.sub.2, N.sub.2O, and/or patient respiratory rate and some embodiments may simultaneously display numerical and graphical data representations.
[0053] The smartphone 400 may include software such as an application configured to enable interaction with the instructions 405 as well as to manage output measurement data from the measurement head processing module. The instruction application functionality can include provision of assembly or operation instructions prior to and during ventilation assistance, allowing clinician input of patient and/or equipment characteristics, and provision of feedback during ventilation assistance based on physiological parameters. The data management functionality of the application can include trend analysis, current measurement information, alarms associated with above/below threshold readings, reminders to take measurement data at certain times or cycles, display customization, iconic data such as hearts beating, color coordination, bar graphs, gas bars, charts, graphs, or the like, all usable by a caregiver or smartphone user to enable helpful and directed medical monitoring of specified physiological parameters.
[0054] In some embodiments, software capable of analyzing the output measurement data received from the processing module and making the data available in an appropriate manner health management is installed on the smartphone 400. The smartphone 400 may also be able to alert the user to an abnormal data reading and to update the manual ventilation instructions 405 accordingly. For example, an abnormally low or high carbon dioxide reading may cause the smartphone 400 to buzz, vibrate or otherwise notify the user of an abnormal reading. The smartphone 400 can also issue a graphical warning. In some embodiments, the instructions can be updated based on the abnormal reading to provide an updated compression rate and/or compression depth for a resuscitation bag.
[0055] The smartphone 400 can include graphical instructions 405 for review by the clinician prior to manual ventilation therapy as well as a selectable option 410 to begin therapy. In some embodiments, the smartphone 400 can determine that therapy has begun based on input physiological parameter data and the option 410 can be omitted. The instructions 405 can be replaced or supplemented, upon commencement of ventilation therapy, with a compression rate and/or compression depth indicator providing visual or auditory feedback to a provider regarding depth and timing of resuscitation bag compressions.
[0056] For example, as illustrated in FIG. 4B, a visual indicator 415 can be accompanied by text 420 in one embodiment indicating compression rate and compression depth determined to be suitable for the patient based at least partly on the information elements. Some embodiments of the software application may also allow for input by a physician or other care provider and can use the input to determine the compression rate and compression depth. The visual indicator 415 can be, in one example, a plurality of concentric circles. In some embodiments, the circles can be sequentially illuminated from the outer circle toward the inner circle to simulate squeezing of the resuscitation bag in order to provide a physician with visual feedback regarding depth and timing of compressions. The text 420 can indicate to the physician compression rate in breaths per minute, a length of time for each compression, and a percentage of compression of the bag, among other things.
[0057] In certain embodiments, a software application for presenting resuscitation instructions may be downloadable from a computer network at a cost, by subscription, pay-per-use, or the like. Other embodiments may advantageously be incorporated into caregiver-specific applications which include reminders for timed measurements or protocols. For example, a caregiver for a surgical patient may desire measurement data at regular intervals to assess the presence and effects of anesthetic agents. A caregiver-specific application may be advantageously programmed to accomplish such a protocol. Other caregiver-specific applications may provide animated or textual instructions, links to online information regarding certain monitoring situations, ailments, or other useful patient research.
[0058] FIGS. 5A-5D illustrate various embodiments of display interfaces on a respiratory gas analyzing measuring head 500. The measuring head 500 comprises a display 505, which may include a plurality of display portions in which a plurality of physiological parameters may be displayed, such as end tidal carbon dioxide (ETCO.sub.2) and respiratory rate. The display 505 can also include a portion for manual ventilation compression instructions 510. The configuration of these various display portions is meant for illustrative purposes, and one skilled in the art would appreciate that the parameter and instruction displays could be rearranged relative to one another, displayed alone, or the user interface could be modified to include other parameter display portions. Further, although some of the parameter display portions employ numerical representations of the physiological data, some embodiments may employ graphical representations, for example a contracting/expanding lung icon may indicate respiratory rate.
[0059] As illustrated in FIG. 5A and FIG. 5B, manual ventilation compression instructions 510 can be graphically represented using a plurality of concentric circles in one embodiment. The circles can be sequentially illuminated or filled from the inner circle towards the outer circle in some embodiments to indicate compression rate and compression depth. To illustrate, in one embodiment the timing between the innermost circle first being illuminated, the expansion of the illumination toward the outer circle, and the contraction of the illumination toward the inner circle until no circle is illuminated can correspond to the length of time for one compression and decompression of a resuscitation bag. As the illumination begins expanding outward the clinician can slowly apply pressure on the resuscitation bag. When the illumination reaches its outermost point and begins contracting inward the clinician can slowly release pressure on the resuscitation bag. As another illustration, the amount of illumination of compression instructions circles can correspond to an amount of compression applied to the resuscitation bag in some embodiments. When no circles are illuminated, no compression should be applied to the bag. When the outermost circle is illuminated the bag should be fully compressed. When circles between the inner and outer circle are illuminated, the bag should be partially compressed corresponding to the positioning of the largest illuminated circle relative to the outer circle. According to one embodiment of the instructions, a bag may not need full compression for suitable patient ventilation so the outermost circle would not be illuminated. In other embodiments, the circles can be sequentially illuminated from the outer circle toward the inner circle to simulate squeezing of the resuscitation bag. Similarly, the circles can be sequentially un-illuminated from the inner circle to the outer circle as the bag is decompressed.
[0060] The compression rate and/or compression level can be set at the initialization of instructions based on data read from the information element of an attached airway adapter and/or resuscitation bag. The compression rate and/or compression level can be varied during therapy based on physiological parameters from the patient's respiratory gases or other physiological parameters from other physiological sensors.
[0061] The illustrated graphical example is just one means by which the measuring head 500 or an associated display can provide a clinician with instructions or feedback for manual ventilation therapy. Animated graphical representations of compression can be used, such as an animated graphic of a hand squeezing and releasing a bag, an icon of lungs inflating and deflating, or the like. Auditory representations can be used such as verbal instructions or a sound that increases or decreases in pitch or volume. These examples are meant to illustrate and not to limit the manual ventilation instruction capabilities of the systems discussed herein.
[0062] As illustrated in FIG. 5C and FIG. 5D, the measuring head 500 display can be used to provide feedback such as a warning to a care provider. One example of a warning can be an indication that ventilation is too slow 515, indicating that the care provider should increase a rate of compressing a resuscitation bag. Another example of a warning can be an indication 520 to apply more pressure to a resuscitation bag. Other warnings and indications are possible in other embodiments.
[0063] FIG. 6 illustrates an embodiment of a manual ventilation kit 600. In the illustrated embodiment, the kit 600 can include an airway adapter 605 and a resuscitation bag 610. In other embodiments the kit 600 can include multiple airway adapters and multiple resuscitation bags. The airway adapter 605 and resuscitation bag 610 can be matched according to patient age or size, for example an adult airway adapter and an adult resuscitation bag. Another example of a kit can include an infant airway adapter and an infant resuscitation bag. The kit can be packaged so as to keep the contents sterile. One or both of the airway adapter 605 and resuscitation bag 610 can include an information element as described above.
II. Example Physiological Monitoring Processes
[0064] FIG. 7 illustrates an embodiment of a manual ventilation process 700. The process 700 can be implemented using systems and components as are described above with respect to FIG. 1A through FIG. 6, or by any manual ventilation system having the component-recognizing and instruction-generating capabilities discussed herein.
[0065] The process 700 begins at block 705 when a clinician or other care provider connects a disposable airway adapter to a reusable respiratory gas measurement device. This can cause electrical or wireless connection of an information element located on the airway adapter with a processor of the measurement device, as discussed above. The measurement device can determine whether the airway adapter has an information element and, if so, whether data on the information element identifies a size, type, or manufacturer of the airway adapter.
[0066] At block 710, the clinician connects a resuscitation bag to the measurement device. This can cause electrical or wireless connection of an information element located on the resuscitation bag with a processor of the measurement device, as discussed above. The measurement device can read the information element as discussed above.
[0067] At block 715, the clinician can receive instructions for ventilation therapy performed using the connected airway adapter and resuscitation bag. The instructions can include one or more of assembly instructions, patient placement instructions, or therapy instructions. The therapy instructions can include one or both of compression rate and compression depth for the resuscitation bag. As discussed above, the clinician can receive the instructions on the measurement device or a connected display such as the clinician's smartphone.
[0068] At block 720, the clinician can perform therapy as needed according to the instructions. In some embodiments, the instructions can be updated based on physiological parameters of the patient as determined by the measurement device.
[0069] FIG. 8 illustrates an embodiment of a process 800 for providing active feedback ventilation instructions. The process 800 can be implemented using systems and components as are described above with respect to FIG. 1A through FIG. 6, or by any manual ventilation system having the component-recognizing and instruction-generating capabilities discussed herein.
[0070] The process 800 begins at block 805 when a respiratory gas measurement device detects a connected airway adapter. A measurement device can be the measurement head 105 of FIGS. 1A-1B in some embodiments. The airway adapter can be detected using an information element, pressure sensing, clinician input, or other known means. The measurement device can detect the airway adapter in preferred embodiments using electrical or wireless connection of an information element located on the airway adapter with a processor of the measurement device, as discussed above. The measurement device can determine whether the airway adapter has an information element in some embodiments or whether the airway adapter is an unknown adapter with no information element.
[0071] At block 810, the respiratory gas measurement device detects a connected resuscitation bag. The resuscitation bag can be detected using an information element, pressure sensing, clinician input, or other known means. The measurement device can detect the resuscitation bag in preferred embodiments using electrical or wireless connection of an information element located on the resuscitation bag with a processor of the measurement device, as discussed above. The measurement device can determine whether the resuscitation bag has an information element in some embodiments or whether the resuscitation bag is an unknown bag with no information element.
[0072] At decision block 815, the measurement device can determine whether the adapter and bag are recognized. A recognized adapter or bag in one example can be an adapter or bag associated with instructions. The instructions can be stored locally on the measurement device in some embodiments, or in other embodiments can be stored in another location such as on an information element read by the measurement device or on a server accessed by the measurement device through a network. In one embodiment, if one or both of the adapter and bag are not recognized, then the process 800 can end. In another embodiment, only if both of the adapter and bag are not recognized will the process 800 end.
[0073] If one or both of the adapter and bag are recognized, then the process transitions to block 820. At block 820, the measurement device provides instructions for ventilation therapy performed using the connected airway adapter and resuscitation bag. The instructions can include one or more of assembly instructions, patient placement instructions, or therapy instructions. The therapy instructions can include one or both of compression rate and compression depth for the resuscitation bag, and can be delivered graphically or through auditory devices. As discussed above, the clinician can receive the instructions on the measurement device or a connected display such as the clinician's smartphone.
[0074] Optionally, at block 825, the measurement device can determine whether the ventilation therapy is being performed correctly. For example, the measurement device can determine the concentration of a desired substance (such as carbon dioxide, oxygen, etc.), in exhaled gases of the patient. The concentration can be compared to a range or threshold indicating that adequate ventilation is being provided to the patient. If therapy is being performed correctly, then the process 800 can loop back to block 820 to continue providing instructions for ventilation to the clinician. The process 800 can periodically or continuously perform the determination of block 825.
[0075] If, at block 825, the measurement device determines that ventilation therapy is not being performed correctly, then the process 800 transitions to optional block 830. Optionally, at block 830, the measurement device can provide feedback or a warning to indicate that the patient is receiving inadequate ventilation. For example, the feedback can include a change to the instructions, such as an increase or decrease in compression rate. The feedback can also include textual or spoken instructions regarding changes in therapy technique. As another example, a warning can be issued indicating that ventilation is too fast or too slow.
[0076] The process 800 can then transition to optional block 845 to determine whether therapy is on-going or has ceased. This can be determined based on clinician input, prompting the clinician to indicate whether therapy is continuing, or by monitoring patient physiological parameters. If therapy is not continuing then the process 800 can end. If therapy is continuing then the process 800 can loop back to block 820 to continue providing instructions for ventilation to the clinician. In some embodiments, block 845 can be omitted and the process 800 can continue as long as the measurement device is powered on and/or connected to an airway adapter and ventilation bag.
III. Terminology
[0077] Many other variations than those described herein will be apparent from this disclosure. For example, depending on the embodiment, certain acts, events, or functions of any of the algorithms described herein can be performed in a different sequence, can be added, merged, or left out altogether (e.g., not all described acts or events are necessary for the practice of the algorithms). Moreover, in certain embodiments, acts or events can be performed concurrently, e.g., through multi-threaded processing, interrupt processing, or multiple processors or processor cores or on other parallel architectures, rather than sequentially. In addition, different tasks or processes can be performed by different machines and/or computing systems that can function together.
[0078] The various illustrative logical blocks, modules, and algorithm steps described in connection with the embodiments disclosed herein can be implemented as electronic hardware, computer software, or combinations of both. To clearly illustrate this interchangeability of hardware and software, various illustrative components, blocks, modules, and steps have been described above generally in terms of their functionality. Whether such functionality is implemented as hardware or software depends upon the particular application and design constraints imposed on the overall system. The described functionality can be implemented in varying ways for each particular application, but such implementation decisions should not be interpreted as causing a departure from the scope of the disclosure.
[0079] The various illustrative logical blocks and modules described in connection with the embodiments disclosed herein can be implemented or performed by a machine, such as a general purpose processor, a digital signal processor (DSP), an application specific integrated circuit (ASIC), a field programmable gate array (FPGA) or other programmable logic device, discrete gate or transistor logic, discrete hardware components, or any combination thereof designed to perform the functions described herein. A general purpose processor can be a microprocessor, but in the alternative, the processor can be a controller, microcontroller, or state machine, combinations of the same, or the like. A processor can also be implemented as a combination of computing devices, e.g., a combination of a DSP and a microprocessor, a plurality of microprocessors, one or more microprocessors in conjunction with a DSP core, or any other such configuration. Although described herein primarily with respect to digital technology, a processor may also include primarily analog components. For example, any of the signal processing algorithms described herein may be implemented in analog circuitry. A computing environment can include any type of computer system, including, but not limited to, a computer system based on a microprocessor, a mainframe computer, a digital signal processor, a portable computing device, a personal organizer, a device controller, and a computational engine within an appliance, to name a few.
[0080] The steps of a method, process, or algorithm described in connection with the embodiments disclosed herein can be embodied directly in hardware, in a software module executed by a processor, or in a combination of the two. A software module can reside in RAM memory, flash memory, ROM memory, EPROM memory, EEPROM memory, registers, hard disk, a removable disk, a CD-ROM, or any other form of non-transitory computer-readable storage medium, media, or physical computer storage known in the art. An exemplary storage medium can be coupled to the processor such that the processor can read information from, and write information to, the storage medium. In the alternative, the storage medium can be integral to the processor. The processor and the storage medium can reside in an ASIC. The ASIC can reside in a user terminal. In the alternative, the processor and the storage medium can reside as discrete components in a user terminal.
[0081] Conditional language used herein, such as, among others, "can," "might," "may," "e.g.," and the like, unless specifically stated otherwise, or otherwise understood within the context as used, is generally intended to convey that certain embodiments include, while other embodiments do not include, certain features, elements and/or states. Thus, such conditional language is not generally intended to imply that features, elements and/or states are in any way required for one or more embodiments or that one or more embodiments necessarily include logic for deciding, with or without author input or prompting, whether these features, elements and/or states are included or are to be performed in any particular embodiment. The terms "comprising," "including," "having," and the like are synonymous and are used inclusively, in an open-ended fashion, and do not exclude additional elements, features, acts, operations, and so forth. Also, the term "or" is used in its inclusive sense (and not in its exclusive sense) so that when used, for example, to connect a list of elements, the term "or" means one, some, or all of the elements in the list.
[0082] While the above detailed description has shown, described, and pointed out novel features as applied to various embodiments, it will be understood that various omissions, substitutions, and changes in the form and details of the devices or algorithms illustrated can be made without departing from the spirit of the disclosure. As will be recognized, certain embodiments of the inventions described herein can be embodied within a form that does not provide all of the features and benefits set forth herein, as some features can be used or practiced separately from others.
* * * * *
D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

D00013

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.