Remote Loading Of Sparingly Water-soluble Drugs Into Lipid Vesicles
HAYES; Mark E. ; et al.
U.S. patent application number 15/972012 was filed with the patent office on 2019-04-11 for remote loading of sparingly water-soluble drugs into lipid vesicles. The applicant listed for this patent is ZONEONE PHARMA, INC.. Invention is credited to Mark E. HAYES, Charles O. NOBLE, Francis C. SZOKA.
| Application Number | 20190105339 15/972012 |
| Document ID | / |
| Family ID | 55264423 |
| Filed Date | 2019-04-11 |












View All Diagrams
| United States Patent Application | 20190105339 |
| Kind Code | A1 |
| HAYES; Mark E. ; et al. | April 11, 2019 |
REMOTE LOADING OF SPARINGLY WATER-SOLUBLE DRUGS INTO LIPID VESICLES
Abstract
The present invention provides liposome compositions containing sparingly soluble drugs that are used to treat life-threatening diseases. A preferred method of encapsulating a drug inside a liposome is by remote or active loading. Remote loading of a drug into liposomes containing a transmembrane electrochemical gradient is initiated by co-mixing a liposome suspension with a solution of drug, whereby the neutral form of the compound freely enters the liposome and becomes electrostatically charged thereby preventing the reverse transfer out of the liposome. There is a continuous build-up of compound within the liposome interior until the electrochemical gradient is dissipated or all the drug is encapsulated in the liposome. However, this process as described in the literature has been limited to drugs that are freely soluble in aqueous solution or solubilized as a water-soluble complex. This invention describes compositions and methods for remote loading drugs with low water solubility (<2 mg/mL). In the preferred embodiment the drug in the solubilizing agent is mixed with the liposomes in aqueous suspension so that the concentration of solubilizing agent is lowered to below its capacity to completely solubilize the drug. This results in the drug precipitating but remote loading is capability retained. The process is scalable and the resulting drug-loaded liposomes are characterized by a high drug-to-lipid ratios and predictable drug retention when the liposome encapsulated drug is administered in patients.
| Inventors: | HAYES; Mark E.; (San Francisco, CA) ; NOBLE; Charles O.; (San Francisco, CA) ; SZOKA; Francis C.; (San Francisco, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 55264423 | ||||||||||
| Appl. No.: | 15/972012 | ||||||||||
| Filed: | May 4, 2018 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15502092 | Feb 6, 2017 | 10004759 | ||
| PCT/US2015/043594 | Aug 4, 2015 | |||
| 15972012 | ||||
| 62033073 | Aug 4, 2014 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61K 31/496 20130101; A61K 9/127 20130101; A61K 31/7048 20130101; A61K 9/19 20130101; A61K 31/4196 20130101; A61K 9/1277 20130101; A61K 9/1271 20130101; A61K 31/337 20130101; A61K 38/07 20130101; A61K 31/704 20130101; A61K 47/02 20130101; A61K 31/395 20130101 |
| International Class: | A61K 31/7048 20060101 A61K031/7048; A61K 31/704 20060101 A61K031/704; A61K 31/395 20060101 A61K031/395; A61K 9/127 20060101 A61K009/127; A61K 31/337 20060101 A61K031/337; A61K 31/4196 20060101 A61K031/4196; A61K 9/19 20060101 A61K009/19; A61K 38/07 20060101 A61K038/07; A61K 47/02 20060101 A61K047/02; A61K 31/496 20060101 A61K031/496 |
Claims
1. A pharmaceutical formulation comprising a sparingly water-soluble agent encapsulated within a liposome, said formulation manufactured by a method comprising: contacting an aqueous suspension of said liposome with said aqueous suspension of said agent under conditions appropriate to encapsulate said sparingly water-soluble agent in said liposome, wherein said liposome has an internal aqueous environment encapsulated by a lipid membrane and said aqueous suspension of said liposome comprises a gradient selected from a proton gradient, an ion gradient and a combination thereof across said membrane, and wherein said conditions are appropriate for said sparingly water-soluble agent to traverse said membrane and concentrate in said internal aqueous environment, thereby forming said pharmaceutical formulation.
2. The pharmaceutical formulation according to claim 1, wherein said aqueous suspension of said sparingly water-soluble agent is prepared by a method comprising: essentially completely dissolving the agent in an aprotic solvent, forming an agent solution and subsequently diluting said agent solution with an aqueous medium beyond the point of drug solubility.
3. The pharmaceutical formulation of claim 1 where said liposome is selected from multilamellar vesicles (MLV), large unilamellar vesicles (LUV) and small unilamellar vesicles (SUV), oligolamellar vesicles (OLV), paucilamellar vesicles (PLV) or reverse phase evaporation vesicles (REV).
4. The pharmaceutical formulation according to claim 1, wherein said agent is a low molecular weight therapeutic agent having a water solubility of less than or equal to about 2 mg/mL.
5. The pharmaceutical formulation of claim 1, wherein, the agent is selected from an anthracycline compound, a camptothecin compound, a vinca alkaloid, an ellipticine compound, a taxane compound, a wortmannin compound, a geldanamycin compound, a polyene antibiotic, a pyrazolopyrimidine compound, a steroid compound, a peptide-based compound, a taxane, a derivative of any of the foregoing, a pro-drug of any of the foregoing, and an analog of any of the foregoing.
6. The pharmaceutical formulation according to claim 1, wherein said agent is selected from carfilzomib, voriconazole, amiodarone, ziprasidone, aripiprazole, imatinib, lapatinib, oprozomib, cyclopamine, CUR-61414, PF-05212384, PF-4691502, toceranib, PF-477736, PF-337210, sunitinib, SU14813, axitinib, AG014699, veliparib, MK-4827, ABT-263, SU11274, PHA665752, Crizotinib, XL880, PF-04217903, XR5000, AG14361, veliparib, bosutunib, PD-0332991, PF-01367338, AG14361, NVP-ADW742, NVP-AUY922, NVP-LAQ824, NVP-TAE684, NVP-LBH589, erubulin, doxorubicin, daunorubicin, mitomycin C, epirubicin, pirarubicin, rubidomycin, carcinomycin, N-acetyladriamycin, rubidazone, 5-imido daunomycin, N-acetyl daunomycin, daunoryline, mitoxanthrone, camptothecin, 9-aminocamptothecin, 7-ethylcamptothecin, 7-Ethyl-10-hydroxy-camptothecin, 10-hydroxycamptothecin, 9-nitrocamptothecin,10,11-methylenedioxycamptothecin, 9-amino-1O,11-methylenedioxycamptothecin, 9-chloro-1 0, 11-methylenedioxycamptothecin, irinotecan, lurtotecan, silatecan, (7-(4-methylpiperazinomethylene)-10,ll-ethylenedioxy-20(S)-camptothecin, 7-(4-methylpiperazinomethylene)-10, II-methylenedioxy-20(S)-camptothecin, 7-(2-N-isopropylamino)ethyl)-(20S)-camptothecin, CKD-602, vincristine, vinblastine, vinorelbine, vinflunine, vinpocetine, vindesine, 2'succinylpaclitaxel, 2' succinyldocetaxel, 2' succinylcabazitaxel, 2'glutarylpaclitaxel, 2'glutaryldocetaxel, 2'glutarylcabazitaxel, 2'succinyldimethylaminopropylamide of paclitaxel, 2'succinyldimethylaminopropylamide of docetaxel, 2'succinyldimethylaminopropylamide of cabazitaxel, 2' morpholine-4-ylethyl amide of succinylcabazitaxel, 2' morpholine-4-ylethyl amide of succinylpaclitaxel, 2' morpholine-4-ylethyl amide of succinyldocetaxel, 2' morpholinopropylamide paclitaxel, 2' morpholinopropylamide docetaxel, 2' morpholinopropylcabazitaxel. ellipticine, 6-3-aminopropyl-ellipticine, 2-diethylaminoethyl-ellipticinium, datelliptium, retelliptine, paclitaxel, docetaxel, cabazitaxel, diclofenac, bupivacaine, 17-allylamino-geldanamycin, 17-dimethylaminoethylamino-17-demethoxygeldanamycin, cetirizine, fexofenadine, Onx 0912, Onx 0914, PD0332991, Axitinib, Lenvatinib, PHA665752, SU11274, PF-02341066, foretinib, XL880, PX-478, GDC-0349, PD0332991, AZD4547, Golotimod, SCH900776, TG02, UNC0638, ARRY-520, Elacridar hydrochloride, golvatinib, MK-1775, PF-03758309, AT13387, BAY 80-6946, cobicistat, GDC-0068, INNO-206, MLN0905, resminostat, tariquidar, primidone and other catecholamines, epinephrine, salts, prodrugs and derivatives of these medicinal compounds and mixtures thereof.
7. The pharmaceutical formulation according to claim 1, wherein said agent is selected from an antihistamine ethylenediamine derivative, bromphenifamine, diphenhydramine, an anti-protozoal drug, quinolone, iodoquinol, an amidine compound, pentamidine, an antihelmintic compound, pyrantel, an anti-schistosomal drug, oxaminiquine, an antifungal compound amphotericin B, 2'deoxyamphotericin B, triazole derivative, fliconazole, itraconazole, ketoconazole, miconazole, an antimicrobial cephalosporin, chelating agents, deferoxamine, deferasirox, deferiprone, FBS0701, cefazolin, cefonicid, cefotaxime, ceftazimide, cefuoxime, an antimicrobial beta-lactam derivative, aztreopam, cefmetazole, cefoxitin, an antimicrobial of erythromycin group, erythromycin, azithromycin, clarithromycin, oleandomycin, a penicillin compound, benzylpenicillin, phenoxymethylpenicillin, cloxacillin, methicillin, nafcillin, oxacillin, carbenicillin, a tetracycline compound, novobiocin, spectinomycin, vancomycin; an antimycobacterial drug, aminosalicycic acid, capreomycin, ethambutol, isoniazid, pyrazinamide, rifabutin, rifampin, clofazimine, an antiviral adamantane compound, amantadine, rimantadine, a quinidine compound, quinine, quinacrine, chloroquine, hydroxychloroquine, primaquine, amodiaquine, mefloquine, an antimicrobial, qionolone, ciprofloxacin, enoxacin, lomefloxacin, nalidixic acid, norfloxacin, ofloxacin, a sulfonamide; a urinary tract antimicrobial, nitrofurantoin, trimetoprim; anitroimidazoles derivative, metronidazole, a cholinergic quaternary ammonium compound, ambethinium, neostigmine, physostigmine, an anti-Alzheimer aminoacridine, tacrine, an anti-parkinsonal drug, benztropine, biperiden, procyclidine, trihexylhenidyl, an anti-muscarinic agent, atropine, hyoscyamine, scopolamine, propantheline, an adrenergic compound, dopamine, serotonin, a hedgehog inhibitor, albuterol, dobutamine, ephedrine, epinephrine, norepinephrine, isoproterenol, metaproperenol, salmetrol, terbutaline, a serotonin reuptake inhibitor, an ergotamine derivative, a myorelaxant, a curare series, a central action myorelaxant, baclophen, cyclobenzepine, dentrolene, nicotine, a nicotine receptor antagonist, a beta-adrenoblocker, acebutil, amiodarone, abenzodiazepine compound, ditiazem, an antiarrhythmic drug, diisopyramide, encaidine, a local anesthetic compound, procaine, procainamide, lidocaine, flecaimide, quinidine, an ACE inhibitor, captopril, enelaprilat, Hsp90 inhibitor, fosinoprol, quinapril, ramipril; an opiate derivative, codeine, meperidine, methadone, morphine, an antilipidemic, fluvastatin, gemfibrosil, an HMG-coA inhibitor, pravastatin, a hypotensive drug, clonidine, guanabenz, prazocin, guanethidine, granadril, hydralazine, a non-coronary vasodilator, dipyridamole, an acetylcholine esterase inhibitor, pilocarpine, an alkaloid, physostigmine, neostigmine, a derivative of any of the foregoing, a pro-drug of any of the foregoing, and ananalog of any of the foregoing.
8. The pharmaceutical formulation of claim 1, wherein the aprotic solvent is selected from dimethylsulfoxide, dioxane, tetrahydrofuran, dimethylformamide, acetonitrile, dimethylacetamide, sulfolane, gamma butyrolactone, pyrrolidones, 1-methyl-2-pyrrolidinone, methylpyrroline, ethylene glycol monomethyl ether, diethylene glycol monomethyl ether, polyethylene glycol.
9. The pharmaceutical formulation of claim 1, wherein the liposome is prepared from one or more lipids selected from egg phosphatidylcholine (EPC), egg phosphatidylglycerol (EPG), dipalmitoylphosphatidylcholine (DPPC), sphingomyelin (SM), cholesterol (Chol), cholesterol sulfate and its salts (CS), cholesterol hemisuccinate and its salts (Chems), cholesterol phosphate and its salts (CP), cholesterol phthalate, cholesterylphosphorylcholine, 3,6,9-trioxaoctan-ol-cholesteryl-3e-ol, dimyristoylphosphatidylglycerol (DMPG), dimyristoylphosphatidylglycerol (DMPG), dimyristoylphosphatidylcholine (DMPC), distearoylphosphatidylcholine (DSPC), hydrogenated soy phosphatidylcholine (HSPC), distearoylphosphatidylglycerol (DSPG), sterol modified lipids (SML), inverse-phosphocholine lipids, cationic lipids and zwitterlipids.
10. The pharmaceutical formulation of claim 1, wherein the percentage of agent from said aqueous suspension of said agent encapsulated in said internal aqueous medium of said liposome ranges from about 10% to about 100% of the total agent in said aqueous suspension of said agent.
11. The pharmaceutical formulation of claim 1, wherein said ion gradient is caused by a difference in concentrations across said membrane of a member selected from an amine salt and a metal salt of a member selected from a carboxylate, a sulfate, and a phosphate.
12. The pharmaceutical formulation according to claim 11, wherein said carboxylate is acetate.
13. The pharmaceutical formulation according to claim 11, wherein said amine salt and said metal salt are selected from a salt of a member selected from a monovalent carboxylate, a multivalent carboxylate, a sulfate and a phosphate.
14. The pharmaceutical formulation according to claim 11, wherein the cation in said salt is selected from sodium, calcium, magnesium, zinc, copper, potassium, primary, secondary, tertiary and quaternary ammonium species.
15. The pharmaceutical formulation according to claim 1, wherein said formulation is lyophilized.
16. The pharmaceutical formulation according to claim 1, wherein said agent is present in said internal aqueous medium as a unit dosage format.
17. A method of treating a disease state in a subject in need of said treatment, said method comprising administering to said patient a therapeutically effective amount of said formulation of claim.
Description
CROSS-REFERENCES TO RELATED APPLICATIONS
[0001] This application is a Continuation of U.S. application Ser. No. 15/502,092 filed Feb. 6, 2017, which is a 371 U.S. National Phase of PCT International Application No. PCT/US2015/043594 filed Aug. 4, 2015, which claims the benefit under 35 U.S.C. .sctn. 119(e) of U.S. Provisional Application No. 62/033,073 filed on Aug. 4, 2014, all of which are incorporated herein by reference in their entireties for all purposes.
BACKGROUND OF THE INVENTION
[0002] This invention relates to the fields of pharmaceutical compositions, methods for making them and the uses of the resulting compositions in drug therapy. The pharmaceutical compositions include the active therapeutic agent encapsulated within the aqueous interior of a liposome vesicle.
DESCRIPTION OF THE RELATED ART
[0003] The pharmaceutical industry, in its quest for improved drugs, has generated a large number of potent compounds that are sparingly soluble in water, the ubiquitous solvent that makes life possible. The low water solubility of these new drugs has made it difficult to deliver them in animals including humans. This has created the need for drug delivery systems that can solubilize sparingly water-soluble drugs to enable to their delivery in the body.
[0004] Liposomes are vesicle structures usually composed of a bilayer membrane of amphipathic molecules such as, phospholipids, entrapping an aqueous core. The diameters and morphology of various types of liposomes are illustrated in FIG. 1. Drugs are either encapsulated in the aqueous core or interdigitated in the bilayer membrane. Drugs interdigitated in the membrane transfer out of the liposome when it is diluted into the body. Importantly, drugs that are encapsulated in the aqueous core or held in complexes in the aqueous core are retained substantially longer than drugs in the bilayer. The use of liposomes with drugs encapsulated in the aqueous core for drug delivery is well established (D. Drummond et al., J. Pharm. Sci., (2008) 97(11):4696-4740, PMID 10581328).
[0005] A variety of loading methods for encapsulating functional compounds, particularly drugs, in liposomes is available. Hydrophilic compounds for example can be encapsulated in liposomes by hydrating a mixture of the functional compounds and vesicle-forming lipids. This technique is called passive loading. The functional compound is encapsulated in the liposome as the nanoparticle is formed. The available lipid vesicle (liposome) production procedures are satisfactory for most applications where water-soluble drugs are encapsulated (G. Gregoriadis, Ed., Liposome Technology, (2006) Liposome Preparation and Related Techniques, 3rd Ed.) However, the manufacture of lipid vesicles that encapsulate drugs sparing water-soluble (e.g., with a water solubility less than 2 mg/mL) in the aqueous inner compartment of the liposome is exceedingly difficult (D. Zucker et al., Journal of Controlled Release (2009) 139:73-80, PMID 19508880).
[0006] Passive loading of lipophilic and, to a lesser extent amphiphilic functional compounds, is somewhat more efficient than hydrophilic functional compounds because they partition in both the lipid bilayer and the intraliposomal (internal) aqueous medium. However, using passive loading, the final functional-compound-to-lipid ratio as well as the encapsulation efficiency are generally low. The concentration of drug in the liposome equals that of the surrounding fluid and drug not entrapped in the internal aqueous medium is washed away after encapsulation.
[0007] Certain hydrophilic or amphiphilic compounds can be loaded into preformed liposomes using transmembrane pH- or ion-gradients (D. Zucker et al., Journal of Controlled Release (2009) 139:73-80). This technique is called active or remote loading. Compounds amenable to active loading should be able to change from an uncharged form, which can diffuse across the liposomal membrane, to a charged form that is not capable thereof. Typically, the functional compound is loaded by adding it to a suspension of liposomes prepared to have a lower outside/higher inside pH- or ion-gradient. Via active loading, a high functional-compound-to-lipid mass ratio and a high loading efficiency (up to 100%) can be achieved. Examples are active loading of anticancer drugs doxorubicin, daunorubicin, and vincristine (P. R. Cullis et al., Biochimica et Biophysica Acta, (1997) 1331:187-211, and references therein).
[0008] Hydrophobic drugs are only considered capable of loading into liposomes through membrane intercalation via some passive loading/assembly mechanism. Wasan et al. states "Agents that have hydrophobic attributes can intercalate into the lipid bilayer and this can be achieved by adding the agent to the preformed liposomes." in a description of the use of micelles to transfer sparingly soluble agents to a liposome bilayer (US 2009/0028931).
[0009] Remote loading of a sparingly soluble drug into a liposome under conditions where the drug is above its solubility limit and is in the form of a precipitate is an unexpected event. D. Zucker et al., Journal of Controlled Release (2009) 139:73-80 states "Hydrophobic molecules may aggregate, and these aggregates have low permeability across the liposomal membrane. Thus, when the non-polar/polar surface area ratio is >2.31 (FIG. 4), it is necessary that the drug would have a reasonable solubility, >1.9 mM, in order to achieve high loading because only soluble uncharged molecules can enter the liposome." (D. Zucker et al., Journal of Controlled Release (2009) 139:73-80).
[0010] To date, a method has not been developed for the active loading of the aqueous core of a liposome with a sparingly water-soluble agent from a precipitate.
BRIEF DESCRIPTION OF THE DRAWINGS
[0011] FIG. 1 illustrates the diameters and morphology of various types of liposomes.
[0012] FIG. 2. Liposome formulations composed of HSPC/Chol/Peg-DSPE containing either sodium sulfate (light shade) or ammonium sulfate (dark shade) were incubated with carfilzomib at two input drug-to-lipid ratios using conditions described below. The liposomes were purified from unencapsulated drug and the amount of encapsulated carfilzomib within the liposomes is shown, expressed as .mu.g of carfilzomib per .mu.mol lipid.
[0013] FIG. 3 is a bar graph showing a trapping agent effect on liposome loading of carfilzomib.
[0014] FIG. 4 is a bar graph showing a method of drug introduction effect on liposome loading of carfilzomib.
[0015] FIG. 5 is a line graph showing carfilzomib loading from precipitate demonstrated by reduction of light scattering at 600 nm.
[0016] FIG. 6 is a HPLC Chromatogram of Carfilzomib before loading into liposomes (upper) and after loading into liposomes from a precipitate and being released from liposomes using a reverse ammonium sulfate gradient back to a precipitate in the extraliposomal solution (lower).
[0017] FIG. 7 is a line graph showing liposome encapsulation efficiency as a function of [DMSO]. The input drug-to-lipid ratio was 200 .mu.g/.mu.mol.
[0018] FIG. 8 is a line graph showing light scattering of carfilzomib solution as a function of DMSO concentration. The concentration of carfilzomib was 0.2 mg/mL.
[0019] FIG. 9 is a bar graph showing the effect of delay time between the formation of drug precipitate and liposome loading of the precipitate.
[0020] FIG. 10 is a line graph showing the effect of ammonium sulfate trapping agent concentration on liposome drug payload of carfilzomib Loaded from Precipitate.
[0021] FIG. 11 is a line graph showing effect of ammonium sulfate trapping agent concentration on liposome loading efficiency of carfilzomib from precipitate.
[0022] FIG. 12 is a line graph loading insoluble carfilzomib precipitate into liposomes using a triethylammonium sulfate gradient.
[0023] FIG. 13 is a line graph showing the transfer of insoluble carfilzomib precipitate into liposomes by remote loading.
[0024] FIG. 14 is a bar graph showing the comparison of liposome loading of aripiprazole when mixed with liposomes as a SBCD complex (Abilify) or when diluted from a stock DMSO solution directly into liposomes, creating a drug suspension.
[0025] FIG. 15 is a bar graph showing absorbance at 600 nm (scattering) of drug solutions (dark bars) and liposome drug mixtures (gray bars). The rectangle indicates the samples where a substantial decrease in scattering was measured upon incubation with liposomes indicating drug loading.
[0026] FIG. 16 is a bar graph showing the loading efficiency of DFX in calcium acetate liposomes.
[0027] FIG. 17 is a plot showing DFX loading capacity in liposomes containing calcium acetate as a trapping agent.
[0028] FIG. 18 is a plot showing DFX loading capacity in liposomes containing different acetate trapping agents.
[0029] FIG. 19A-FIG. 19B are illustrations of the structures of paclitaxel, docetaxel and cabazitaxel and modifications to them that enable the taxanes to be loaded from a precipitate into liposomes containing an ion gradient.
SUMMARY OF THE INVENTION
[0030] In utilizing liposomes for delivery of functional compounds, it is generally desirable to load the liposomes to high concentration, resulting in a high functional-compound-lipid mass ratio, since this reduces the amount of liposomes to be administered per treatment to attain the required therapeutic effect, all the more since several lipids used in liposomes have a dose-limiting toxicity by themselves. The loading percentage is also of importance for cost efficiency, since poor loading results in a great loss of the active compound.
[0031] In an exemplary embodiment, the invention provides a liposome comprising a liposomal lipid membrane encapsulating an internal aqueous medium. The internal aqueous medium comprises an aqueous solution of a complex between a trapping agent and a sparingly water-soluble therapeutic agent.
[0032] In a further exemplary embodiment, the invention provides pharmaceutical formulations comprising a liposome of the invention. The formulations include the liposome and a pharmaceutically acceptable diluent or excipient. In various embodiments, the pharmaceutical formulation is in a unit dosage format, providing a unit dosage of the therapeutic agent encapsulated in the liposome.
[0033] In another exemplary embodiment, the invention provides methods of making the liposomes of the invention. In various embodiments, there is provided a method of remotely loading a liposome with an agent that is sparingly water-soluble. The method comprises: a) incubating an aqueous mixture comprising: (i) a liposome suspension having a proton and/or ion gradient that exists across the liposomal membrane; (ii) with an aqueous suspension of a sparingly soluble drug (iii) wherein the drug suspension is made by completely dissolving the drug in an aprotic solvent or polyol and diluting it into the aqueous solution beyond the point of drug solubility where a precipitate is formed, wherein incubating the combined liposome drug precipitate mixture for a period of time results in the drug accumulating within the liposome interior in response to the proton/ion gradient. The mixture used to load the liposome with the agent is prepared such that a proton- and/or ion-gradient exists across the liposomal membrane between the internal aqueous membrane and the external aqueous medium. The incubating can be for any useful period but is preferably for a period of time sufficient to cause at least part of the insoluble drug precipitate to accumulate in the internal aqueous medium under the influence of the proton and/or ion gradient.
[0034] Other embodiments, objects and advantages are set forth in the Detailed Description that follows.
DESCRIPTION OF THE PREFERRED EMBODIMENTS
Introduction
[0035] In utilizing liposomes for delivery of functional compounds, it is generally desirable to load the liposomes to high concentration, resulting in a high agent-lipid mass ratio, since this reduces the amount of liposomes to be administered per treatment to attain the required therapeutic effect of the agent, all the more since several lipids used in liposomes have a dose-limiting toxicity by themselves. The loading percentage is also of importance for cost efficiency, since poor loading results in an increase loss of agent during the loading of the agent into the liposome.
[0036] The present invention provides liposomes encapsulating agents, e.g., sparingly water-soluble, methods of making such liposomes, formulations containing such liposomes and methods of making the liposomes and formulations of the invention.
[0037] In an exemplary embodiment, the invention provides a liposome having a membrane encapsulating an aqueous compartment. The liposome is prepared such that a proton- and/or ion-gradient exists across the liposomal membrane between the internal aqueous compartment and the external aqueous medium. The agent is dissolved in an aprotic solvent at a concentration that when diluted in the liposome suspension its solubility in the suspension is exceeded and the agent forms a precipitate. A portion of the agent precipitate is loaded into the liposome aqueous compartment using a proton- and/or ion-gradient exists across the liposomal membrane between the internal aqueous compartment and the external aqueous medium.
[0038] In some embodiments, essentially the entire amount of the insoluble agent precipitate is loaded into the aqueous compartment of the liposome. In an exemplary embodiment, at least about 95%, at least about 90%, at least about 85%, at least about 80% or at least about 70% of the insoluble drug precipitate is loaded into the aqueous compartment of the liposome.
Liposomes
[0039] The term liposome is used herein in accordance with its usual meaning, referring to microscopic lipid vesicles composed of a bilayer of phospholipids or any similar amphipathic lipids encapsulating an internal aqueous medium. The liposomes of the present invention can be unilamellar vesicles such as small unilamellar vesicles (SUVs) and large unilamellar vesicles (LUVs), and multilamellar vesicles (MLV), typically varying in size from 30 nm to 200 nm. No particular limitation is imposed on the liposomal membrane structure in the present invention. The term liposomal membrane refers to the bilayer of phospholipids separating the internal aqueous medium from the external aqueous medium.
[0040] Exemplary liposomal membranes useful in the current invention may be formed from a variety of vesicle-forming lipids, typically including dialiphatic chain lipids, such as phospholipids, diglycerides, dialiphatic glycolipids, single lipids such as sphingomyelin and glycosphingolipid, cholesterol and derivates thereof, and combinations thereof. As defined herein, phospholipids are amphiphilic agents having hydrophobic groups formed of long-chain alkyl chains, and a hydrophilic group containing a phosphate moiety. The group of phospholipids includes phosphatidic acid, phosphatidyl glycerols, phosphatidylcholines, phosphatidylethanolamines, phosphatidylinositols, phosphatidylserines, and mixtures thereof. Preferably, the phospholipids are chosen from 1,2-dipalmitoyl-sn-glycero-3-phosphocholine (DPPC), dimyristoyl-phosphatidylcholine (DMPC), hydrogenated soy phosphatidylcholine (HSPC), soy phosphatidylcholine (SPC), dimyristoylphosphatidylglycerol (DMPG), disrearoylphosphatidylglycerol (DSPG),1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine (POPC), 1,2-dioleoyl-sn-glycero-3-phosphocholine (DOPC)distearoyl phosphatidylcholine (DSPC), egg yolk phosphatidylcholine (EYPC) or hydrogenated egg yolk phosphatidylcholine (HEPC), sterol modified lipids (SML), cationic lipids and inverse-zwitterlipids.
[0041] Liposomal membranes according to the present invention may further comprise ionophores like nigericin and A23187.
[0042] In the method according to the present invention, an exemplary liposomal phase transition temperature is between -25.degree. C. and 100.degree. C., e.g., between 4.degree. C. and 65.degree. C. The phase transition temperature is the temperature required to induce a change in the physical state of the lipids constituting the liposome, from the ordered gel phase, where the hydrocarbon chains are fully extended and closely packed, to the disordered liquid crystalline phase, where the hydrocarbon chains are randomly oriented and fluid. Above the phase transition temperature of the liposome, the permeability of the liposomal membrane increases. Choosing a high transition temperature, where the liposome would always be in the gel state, could provide a non-leaking liposomal composition, i.e. the concentration of the sparingly water-soluble agent in the internal aqueous medium is maintained during exposure to the environment. Alternatively, a liposome with a transition temperature between the starting and ending temperature of the environment it is exposed to provides a means to release the sparingly water-soluble agent when the liposome passes through its transition temperature. Thus, the process temperature for the active-loading technique typically is above the liposomal phase transition temperature to facilitate the active-loading process. As is generally known in the art, phase transition temperatures of liposomes can, among other parameters, be influenced by the choice of phospholipids and by the addition of steroids like cholesterol, lanosterol, cholestanol, stigmasterol, ergosterol, and the like. Hence, in an embodiment of the invention, a method according to any of the foregoing is provided in which the liposomes comprise one or more components selected from different phospholipids and cholesterol in several molar ratios in order to modify the transition, the required process temperature and the liposome stability in plasma. Less cholesterol in the mixture will result in less stable liposomes in plasma. An exemplary phospholipid composition of use in the invention comprises between about 10 and about 50 mol % of steroids, preferably cholesterol.
[0043] In accordance with the invention, liposomes can be prepared by any of the techniques now known or subsequently developed for preparing liposomes. For example, the liposomes can be formed by the conventional technique for preparing multilamellar lipid vesicles (MLVs), that is, by depositing one or more selected lipids on the inside walls of a suitable vessel by dissolving the lipids in chloroform and then evaporating the chloroform, and by then adding the aqueous solution which is to be encapsulated to the vessel, allowing the aqueous solution to hydrate the lipid, and swirling or vortexing the resulting lipid suspension. This process engenders a mixture including the desired liposomes. Alternatively, techniques used for producing large unilamellar lipid vesicles (LUVs), such as reverse-phase evaporation, infusion procedures, and detergent dilution, can be used to produce the liposomes. A review of these and other methods for producing lipid vesicles can be found in the text Liposome Technology, Volume I, Gregory Gregoriadis Ed., CRC Press, Boca Raton, Fla., (1984), which is incorporated herein by reference. For example, the lipid-containing particles can be in the form of steroidal lipid vesicles, stable plurilamellar lipid vesicles (SPLVs), monophasic vesicles (MPVs), or lipid matrix carriers (LMCs). In the case of MLVs, if desired, the liposomes can be subjected to multiple (five or more) freeze-thaw cycles to enhance their trapped volumes and trapping efficiencies and to provide a more uniform interlamellar distribution of solute.
[0044] Following liposome preparation, the liposomes are optionally sized to achieve a desired size range and relatively narrow distribution of liposome sizes. A size range of about 20-200 nanometers allows the liposome suspension to be sterilized by filtration through a conventional filter, typically a 0.22 or 0.4 micron filter. The filter sterilization method can be carried out on a high through-put basis if the liposomes have been sized down to about 20-200 nanometers. Several techniques are available for sizing liposomes to a desired size. Sonicating a liposome suspension either by bath or probe sonication produces a progressive size reduction down to small unilamellar vesicles less than about 50 nanometer in size. Homogenization is another method which relies on shearing energy to fragment large liposomes into smaller ones. In a typical homogenization procedure, multilamellar vesicles are recirculated through a standard emulsion homogenizer until selected liposome sizes, typically between about 50 and 500 nanometers, are observed. In both methods, the particle size distribution can be monitored by conventional laser-beam particle size determination. Extrusion of liposome through a small-pore polycarbonate membrane or an asymmetric ceramic membrane is also an effective method for reducing liposome sizes to a relatively well-defined size distribution. Typically, the suspension is cycled through the membrane one or more times until the desired liposome size distribution is achieved. The liposomes may be extruded through successively smaller-pore membranes, to achieve a gradual reduction in liposome size. Alternatively controlled size liposomes can be prepared using microfluidic techniques wherein the lipid in an organic solvent such as ethanol or ethanol-aprotic solvent mixtures is rapidly mixed with the aqueous medium, so that the organic solvent/water ratio is less than 30%, in a microchannel with dimensions less than 300 microns and preferable less than 150 microns in wide and 50 microns in height. The organic solvent is then removed from the liposomes by dialysis. Other useful sizing methods such as sonication, solvent vaporization or reverse phase evaporation are known to those of skill in the art.
[0045] Exemplary liposomes for use in various embodiments of the invention have a size of from about 30 nanometers to about 40 microns.
[0046] The internal aqueous medium, as referred to herein, typically is the original medium in which the liposomes were prepared and which initially becomes encapsulated upon formation of the liposome. In accordance with the present invention, freshly prepared liposomes encapsulating the original aqueous medium can be used directly for active loading. Embodiments are also envisaged however wherein the liposomes, after preparation, are dehydrated, e.g. for storage. In such embodiments the present process may involve addition of the dehydrated liposomes directly to the external aqueous medium used to create the transmembrane gradients. However it is also possible to hydrate the liposomes in another external medium first, as will be understood by those skilled in the art. Liposomes are optionally dehydrated under reduced pressure using standard freeze-drying equipment or equivalent apparatus. In various embodiments, the liposomes and their surrounding medium are frozen in liquid nitrogen before being dehydrated and placed under reduced pressure. To ensure that the liposomes will survive the dehydration process without losing a substantial portion of their internal contents, one or more protective sugars are typically employed to interact with the lipid vesicle membranes and keep them intact as the water in the system is removed. A variety of sugars can be used, including such sugars as trehalose, maltose, sucrose, glucose, lactose, and dextran. In general, disaccharide sugars have been found to work better than monosaccharide sugars, with the disaccharide sugars trehalose and sucrose being most effective. Other more complicated sugars can also be used. For example, aminoglycosides, including streptomycin and dihydrostreptomycin, have been found to protect liposomes during dehydration. Typically, one or more sugars are included as part of either the internal or external media of the lipid vesicles. Most preferably, the sugars are included in both the internal and external media so that they can interact with both the inside and outside surfaces of the liposomes' membranes. Inclusion in the internal medium is accomplished by adding the sugar or sugars to the buffer which becomes encapsulated in the lipid vesicles during the liposome formation process. In these embodiments the external medium used during the active loading process should also preferably include one or more of the protective sugars
[0047] As is generally known to those skilled in the art, polyethylene glycol (PEG)-lipid conjugates have been used extensively to improve circulation times for liposome-encapsulated functional compounds, to avoid or reduce premature leakage of the functional compound from the liposomal composition and to avoid detection of liposomes by the body's immune system. Attachment of PEG-derived lipids onto liposomes is called PEGylation. Hence, in an exemplary embodiment of the invention, the liposomes are PEGylated liposomes. PEGylation can be accomplished by incubating a reactive derivative of PEG with the target liposomes. Suitable PEG-derived lipids according to the invention, include conjugates of DSPE-PEG, functionalized with one of carboxylic acids, glutathione (GSH), maleimides (MAL), 3-(2-pyridyldithio) propionic acid (PDP), cyanur, azides, amines, biotin or folate, in which the molecular weight of PEG is between 2000 and 5000 g/mol. Other suitable PEG-derived lipids are mPEGs conjugated with ceramide, having either C8- or C16-tails, in which the molecular weight of mPEG is between 750 and 5000 daltons. Still other appropriate ligands are mPEGs or functionalized PEGs conjugated with glycerophospholipds like 1,2-dimyristoyl-sn-glycero-3-phosphoethanolamine (DMPE), 1,2-dipalmitoyl-sn-glycero-3-phosphoethanolamine (DPPE), 1,2-dioleoyl-sn-glycero-3-phosphoethanolamine (DOPE) and 1,2-distearoyl-sn-glycero-3-phosphoethanolamine (DSPE), and the like. PEGylation of liposomes is a technique generally known by those skilled in the art.
[0048] In various embodiments, the liposomes are PEGylated with DSPE-PEG-GSH conjugates (up to 5 mol %) and/or DSPE-mPEG conjugates (wherein the molecular weight of PEG is typically within the range of 750-5000 daltons, e.g. 2000 daltons). The phospholipid composition of an exemplary PEGylated liposome of the invention may comprise up to 5-20 mol % of PEG-lipid conjugates.
[0049] Furthermore, in certain embodiments, one or more moieties that specifically target the liposome to a particular cell type, tissue or the like are incorporated into the membrane. Targeting of liposomes using a variety of targeting moieties (e.g., ligands, receptors and monoclonal antibodies) has been previously described. Suitable examples of such targeting moieties include hyaluronic acid, anti-ErbB family antibodies and antibody fragments, lipoprotein lipase (LPL), [.alpha.]2-macroglobulin ([.alpha.]2M), receptor associated protein (RAP), lactoferrin, desmoteplase, tissue- and urokinase-type plasminogen activator (tPA/uPA), plasminogen activator inhibitor (PAI-I), tPA/uPA:PAI-1 complexes, melanotransferrin (or P97), thrombospondin 1 and 2, hepatic lipase, factor Vila/tissue-factor pathway inhibitor (TFPI), factor Villa, factor IXa, A[.beta.]1-40, amyloid-[.beta.] precursor protein (APP), CI inhibitor, complement C3, apolipoproteinE (apoE), pseudomonas exotoxin A, CRM66, HIV-I Tat protein, rhinovirus, matrix metalloproteinase 9 (MMP-9), MMP-13 (collagenase-3), spingolipid activator protein (SAP), pregnancy zone protein, antithrombin III, heparin cofactor II, [.alpha.]1-antitrypsin, heat shock protein 96 (HSP-96), platelet-derived growth factor (PDGF), apolipoproteinJ (apoJ, or clusterin), A[.beta.] bound to apoJ and apoE, aprotinin, angiopep-2 (TFFYGGSRGKRNNFKTEEY), very-low-density lipoprotein (VLDL), transferrin, insulin, leptin, an insulin-like growth factor, epidermal growth factors, lectins, peptidomimetic and/or humanized monoclonal antibodies, dingle chain antibodies or peptides specific for said receptors (e.g., sequences HAIYPRH and THRPPMWSPVWP that bind to the human transferrin receptor, or anti-human transferrin receptor (TfR) monoclonal antibody A24), hemoglobin, non-toxic portion of a diphtheria toxin polypeptide chain, all or a portion of the diphtheria toxin B chain, all or a portion of a non-toxic mutant of diphtheria toxin CRM197, apolipoprotein B, apolipoprotein E (e.g., after binding to polysorb-80 coating), vitamin D-binding protein, vitamin A/retinol-binding protein, vitamin B12/cobalamin plasma carrier protein, glutathione and transcobalamin-B 12.
[0050] Targeting mechanisms generally require that the targeting agents be positioned on the surface of the liposome in such a manner that the target moieties are available for interaction with the target, for example, a cell surface receptor. In an exemplary embodiment, the liposome is manufactured to include a connector portion incorporated into the membrane at the time of forming the membrane. An exemplary connector portion has a lipophilic portion which is firmly embedded and anchored in the membrane. An exemplary connector portion also includes a hydrophilic portion which is chemically available on the aqueous surface of the liposome. The hydrophilic portion is selected so that it will be chemically suitable to form a stable chemical bond with the targeting agent, which is added later. Techniques for incorporating a targeting moiety in the liposomal membrane are generally known in the art.
Sparingly Water-Soluble Agent
[0051] As indicated above, the present invention provides liposomes encapsulating a a sparingly water-soluble agent. In the context of the present invention the term `sparingly water-soluble` means being insoluble or having a very limited solubility in water, more in particular having an aqueous solubility of less than 2 mg/mL, e.g., less than 1.9 mg/mL, e.g., having an aqueous solubility of less than 1 mg/mL. As used herein, water solubilities refer to solubilities measured at ambient temperature, which is typically about 20.degree. C., and pH=7.
[0052] According to an exemplary embodiment of the invention, the sparingly water-soluble agent is a therapeutic agent selected from the group of a therapeutic is selected from a group consisting of an amphotericin B compound, an anthracycline compound, a camptothecin compound, a vinca alkaloid, an ellipticine compound, a taxane compound, a wortmannin compound, a geldanamycin compound, a pyrazolopyrimidine compound, a peptide-based compound such as carfilzomib, a steroid compound, a derivative of any of the foregoing, a pro-drug of any of the foregoing, and an analog of any of the foregoing.
[0053] Exemplary small molecule compounds having a water solubility less than about 2 mg/mL include, but are not limited to, amphotericin B, 2'deoxyamphotericin B, carfilzomib, voriconazole, amiodarone, ziprasidone, aripiprazole, imatinib, lapatinib, cyclopamine, oprozomib, CUR-61414, PF-05212384, PF-4691502, toceranib, PF-477736, PF-337210, sunitinib, SU14813, axitinib, AG014699, veliparib, MK-4827, ABT-263, SU11274, PHA665752, Crizotinib, XL880, PF-04217903, XR5000, AG14361, veliparib, bosutunib, PD-0332991, PF-01367338, AG14361, NVP-ADW742, NVP-AUY922, NVP-LAQ824, NVP-TAE684, NVP-LBH589, erubulin, doxorubicin, daunorubicin, mitomycin C, epirubicin, pirarubicin, rubidomycin, carcinomycin, N-acetyladriamycin, rubidazone, 5-imido daunomycin, N-acetyl daunomycin, daunory line, mitoxanthrone, camptothecin, 9-aminocamptothecin, 7-ethylcamptothecin, 7-Ethyl-10-hydroxy-camptothecin, 10-hydroxycamptothecin, 9-nitrocamptothecin, 1O,11-methylenedioxycamptothecin, 9-amino-1O,11-methylenedioxycamptothecin, 9-chloro-1 0, 11-methylenedioxycamptothecin, ininotecan, lurtotecan, silatecan, (7-(4-methylpiperazinomethylene)-10,ll-ethylenedioxy-20(S)-camptothecin, 7-(4-methylpiperazinomethylene)-10, II-methylenedioxy-20(S)-camptothecin, 7-(2-N-isopropylamino)ethyl)-(20S)-camptothecin, CKD-602, vincristine, vinblastine, vinorelbine, vinflunine, vinpocetine, vindesine, ellipticine, 6-3-aminopropyl-ellipticine, 2-diethylaminoethyl-ellipticinium, datelliptium, retelliptine, paclitaxel, docetaxel, diclofenac, bupivacaine, 17-dimethylaminoethylamino-17-demethoxygeldanamycin, cetirizine, fexofenadine, primidone and other catecholamines, epinephrine, (S)-2-(2,4-dihydroxyphenyl)-4,5-dihydro-4-methyl-4-thiazolecarboxylic acid (deferitrin), (S)-4,5-dihydro-2-(3-hydroxy-2-pyridinyl)-4-methyl-4-thiazolecarboxylic acid (desferrithiocin), (S)-4,5-dihydro-2-[2-hydroxy-4-(3,6,9,12-tetraoxatridecyloxy)phenyl]-4-me- thyl-4-thiazolecarboxylic acid, (S)-4,5-dihydro-2-[2-hydroxy-4-(3,6-dioxaheptyloxy)phenyl]-4-methyl-4-thi- azolecarboxylic acid, ethyl (S)-4,5-dihydro-2-[2-hydroxy-4-(3,6-dioxaheptyloxy)phenyl]-4-methyl-4-thi- azolecarboxylate, (S)-4,5-dihydro-2-[2-hydroxy-3-(3,6,9-trioxadecyloxy)]-4-methyl-4-thiazol- ecarboxylic acid, desazadesferrithiocin salts, 2'-hydroxyl modified paclitaxel, 2'-hydroyxl modified docetaxel, 2'-hydroxy modified carbazitaxel and prodrugs and derivatives of these medicinal compounds and mixtures thereof.
[0054] An exemplary therapeutic agent is selected from: an antihistamine ethylenediamine derivative, bromphenifamine, diphenhydramine, an anti-protozoal drug, quinolone, iodoquinol, an amidine compound, pentamidine, an antihelmintic compound, pyrantel, an anti-schistosomal drug, oxaminiquine, an antifungal triazole derivative, fliconazole, itraconazole, ketoconazole, miconazole, an antimicrobial cephalosporin, chelating agents, deferoxamine, deferasirox, deferiprone, FBS0701, cefazolin, cefonicid, cefotaxime, ceftazimide, cefuoxime, an antimicrobial beta-lactam derivative, aztreopam, cefmetazole, cefoxitin, an antimicrobial of erythromycin group, erythromycin, azithromycin, clarithromycin, oleandomycin, a penicillin compound, benzylpenicillin, phenoxymethylpenicillin, cloxacillin, methicillin, nafcillin, oxacillin, carbenicillin, a tetracycline compound, novobiocin, spectinomycin, vancomycin; an antimycobacterial drug, aminosalicycic acid, capreomycin, ethambutol, isoniazid, pyrazinamide, rifabutin, rifampin, clofazimine, an antiviral adamantane compound, amantadine, rimantadine, a quinidine compound, quinine, quinacrine, chloroquine, hydroxychloroquine, primaquine, amodiaquine, mefloquine, an antimicrobial, qionolone, ciprofloxacin, enoxacin, lomefloxacin, nalidixic acid, norfloxacin, ofloxacin, a sulfonamide; a urinary tract antimicrobial, nitrofurantoin, trimetoprim; anitroimidazoles derivative, metronidazole, a cholinergic quaternary ammonium compound, ambethinium, neostigmine, physostigmine, an anti-Alzheimer aminoacridine, tacrine, an anti-parkinsonal drug, benztropine, biperiden, procyclidine, trihexylhenidyl, an anti-muscarinic agent, atropine, hyoscyamine, scopolamine, propantheline, an adrenergic compound, dopamine, serotonin, a hedgehog inhibitor, albuterol, dobutamine, ephedrine, epinephrine, norepinephrine, isoproterenol, metaproperenol, salmetrol, terbutaline, a serotonin reuptake inhibitor, an ergotamine derivative, a myorelaxant, a curare series, a central action myorelaxant, baclophen, cyclobenzepine, dentrolene, nicotine, a nicotine receptor antagonist, a beta-adrenoblocker, acebutil, amiodarone, abenzodiazepine compound, ditiazem, an antiarrhythmic drug, diisopyramide, encaidine, a local anesthetic compound, procaine, procainamide, lidocaine, flecaimide, quinidine, an ACE inhibitor, captopril, enelaprilat, Hsp90 inhibitor, fosinoprol, quinapril, ramipril; an opiate derivative, codeine, meperidine, methadone, morphine, an antilipidemic, fluvastatin, gemfibrosil, an HMG-coA inhibitor, pravastatin, a hypotensive drug, clonidine, guanabenz, prazocin, guanethidine, granadril, hydralazine, a non-coronary vasodilator, dipyridamole, an acetylcholine esterase inhibitor, pilocarpine, an alkaloid, physostigmine, neostigmine, a derivative of any of the foregoing, a pro-drug of any of the foregoing, and ananalog of any of the foregoing.
[0055] This list of agents, however, is not intended to limit the scope of the invention. In fact, the compound encapsulated within the liposome can be any sparingly water-soluble amphipathic weak base or amphipathic weak acid. As noted above, embodiments wherein the sparingly water-soluble agent is not a pharmaceutical or medicinal agent are also encompassed by the present invention.
[0056] Typically, within the context of the present invention, sparingly water-soluble amphipathic weak bases have an octanol-water distribution coefficient (log D) at pH 7 between -2.5 and 7.0 and pKa <11, while sparingly water-soluble amphipathic weak acids have a log D at pH 7 between -2.5 and 7.0 and pKa >3. The pKa is measured in water.
[0057] Typically, the terms weak base and weak acid, as used in the foregoing, respectively refer to compounds that are only partially protonated or deprotonated in water. Examples of protonable agents include compounds having an amino group, which can be protonated in acidic media, and compounds which are zwitterionic in neutral media and which can also be protonated in acidic environments. Examples of deprotonable agents include compounds having a carboxy group, which can be deprotonated in alkaline media, and compounds which are zwitterionic in neutral media and which can also be deprotonated in alkaline environments.
[0058] The term zwitterionic refers to compounds that can simultaneously carry a positive and a negative electrical charge on different atoms. The term amphipathic, as used in the foregoing is typically employed to refer to compounds having both lipophilic and hydrophilic moieties. The foregoing implies that aqueous solutions of compounds being weak amphipathic acids or bases simultaneously comprise charged and uncharged forms of said compounds. Only the uncharged forms may be able to cross the liposomal membrane.
[0059] When agents of use in the present invention contain relatively basic or acidic functionalities, salts of such compounds are included in the scope of the invention. Salts can be obtained by contacting the neutral form of such compounds with a sufficient amount of the desired acid or base, either neat or in a suitable inert solvent. Examples of salts for relative acidic compounds of the invention include sodium, potassium, calcium, ammonium, organic amino, or magnesium salts, or a similar salts. When compounds of the present invention contain relatively basic functionalities, acid addition salts can be obtained by contacting the neutral form of such compounds with a sufficient amount of the desired acid, either neat or in a suitable inert solvent. Examples of acid addition salts include those derived from inorganic acids like hydrochloric, hydrobromic, nitric, carbonic, monohydrogencarbonic, phosphoric, monohydrogenphosphoric, dihydrogenphosphoric, sulfuric, monohydrogensulfuric, hydriodic, or phosphorous acids and the like, as well as the salts derived from organic acids like acetic, propionic, isobutyric, maleic, malonic, benzoic, succinic, suberic, fumaric, lactic, mandelic, phthalic, benzenesulfonic, p-tolylsulfonic, citric, tartaric, methanesulfonic, and the like. Also included are salts of amino acids such as arginate and the like, and salts of organic acids like glucuronic or galactunoric acids and the like (see, for example, Berge et al., Journal of Pharmaceutical Science 1977, 66: 1-19). Certain specific compounds of the present invention contain both basic and acidic functionalities that allow the compounds to be converted into either base or acid addition salts.
[0060] The neutral forms of the compounds are preferably regenerated by contacting the salt with a base or acid and isolating the parent compound in the conventional manner. The parent form of the compound differs from the various salt forms in certain physical properties, such as solubility in polar solvents, but otherwise the salts are equivalent to the parent form of the compound for the purposes of the present invention.
Active Loading
[0061] The process of active loading, involves the use of transmembrane potentials. The principle of active loading, in general, has been described extensively in the art. The terms active-loading and remote-loading are synonymous and will be used interchangeably.
[0062] During active loading, the precipitate of the sparingly water-soluble agent is transferred from the external aqueous medium across the liposomal membrane to the internal aqueous medium by a transmembrane proton- or ion-gradient. The term gradient of a particular compound as used herein refers to a discontinuous increase of the concentration of said compound across the liposomal membrane from outside (external aqueous medium) to inside the liposome (internal aqueous medium).
[0063] To create the concentration gradient, the liposomes are typically formed in a first liquid, typically aqueous, phase, followed by replacing or diluting said first liquid phase. The diluted or new external medium has a different concentration of the charged species or a totally different charged species, thereby establishing the ion- or proton-gradient.
[0064] The replacement of the external medium can be accomplished by various techniques, such as, by passing the lipid vesicle preparation through a gel filtration column, e.g., a Sephadex or Sepharose column, which has been equilibrated with the new medium, or by centrifugation, dialysis, or related techniques.
[0065] The efficiency of active-loading into liposomes depends, among other aspects, on the chemical properties of the complex to be loaded and the type and magnitude of the gradient applied. In an embodiment of the invention, a method as defined in any of the foregoing is provided employing a gradient across the liposomal membrane, in which the gradient is chosen from a pH-gradient, a sulfate-, phosphate-, citrate-, or acetate-salt gradient, an EDTA-ion gradient, an ammonium-salt gradient, an alkylated, e.g methyl-, ethyl-, propyl- and amyl, ammonium-salt gradient, a Ca.sup.2+, Cu.sup.2+, Fe.sup.+2, Mg.sup.2+, Mn.sup.2+, Zn.sup.2+, Na.sup.+-, K.sup.+-gradient, with or without using ionophores, or a combination thereof. These loading techniques have been extensively described in the art.
[0066] Preferably, the internal aqueous medium of pre-formed, i.e., unloaded, liposomes comprises a so-called active-loading buffer which contains water and, dependent on the type of gradient employed during active loading, may further comprise a sulfate-, phosphate-, citrate-, or acetate-salt, an ammonium-salt, an alkylated, e.g., methyl-, ethyl-, propyl- and amyl, ammonium-salt, an Ca.sup.2+, Cu.sup.2+, Fe.sup.+2, Mg.sup.2+, Mn.sup.2+, Zn.sup.2+, Na.sup.+, and/or K.sup.+-salt, an EDTA-ion salt, and optionally a pH-buffer to maintain a pH-gradient. The salts may be polymeric such as dextran sulfate, polyethyleneimine, polyamidoamine dendrimers, the 1.5 carboxylate terminal version of polyamidoamines, polyphosphates, low molecular weight heparin, or hyaluronic acid. In an exemplary embodiment, the concentration of salts in the internal aqueous medium of unloaded liposomes is between 1 and 1000 mM.
[0067] The external aqueous medium, used to establish the transmembrane gradient for active loading, comprises water, the precipitate of the sparingly water-soluble agent(s) to be loaded, and optionally sucrose, saline or some other agent to adjust the osmolarity and/or a chelator like EDTA to aid ionophore activity, more preferably sucrose and/or EDTA.
[0068] In an exemplary embodiment the gradient is chosen from an amine or a metal salt of a member selected from a carboxylate, sulfate or phosphate or an acetate. As is generally known by those skilled in the art, transmembrane pH- (lower inside, higher outside pH) or cation acetate-gradients can be used to actively load amphiphilic weak acids. Amphipathic weak bases can also be actively loaded into liposomes using an ammonium sulfate- or triethylamine sulfate, triethylamine dextran sulfate or ammonium chloride-gradient.
[0069] Carboxylates of use in the invention include, without limitation, carboxylate, citrate, diethylenetriaminepentaaceetate, melletic acetate, 1,2,3,4-butanetetracarboxylate, benzoate, isophalate, phthalate, 3,4-bis(carboxymethyl)cyclopentanecarboxylate, the carboxylate generation of polyamidoamine dendrimers, benzenetricarboxylates, benzenetetracarboxylates, ascorbate, ascorbate phosphate, glucuronate, and ulosonate.
[0070] Sulfates of use in the invention include, but are not limited to, sulfate, 1,5-naphthalenedisulfonate, dextran sulfate, sulfobutlyether beta cyclodextrin, sucrose octasulfate benzene sulfonate, poly(4-styrenesulfonate) trans resveratrol-trisulfate.
[0071] Phosphates of use in the invention include, but are not limited to, phosphate, ascorbate phosphate, hexametaphosphate, phosphate glasses, polyphosphates, triphosphate, trimetaphosphate, bisphosphonates, ethanehydroxy bisphosphonate, inositol hexaphosphate
[0072] Exemplary salts of use in the invention include a mixture of carboxylate, sulfate or phosphate including but not limited to: 2-carboxybenensulfonate, creatine phosphate, phosphocholine, carnitine phosphate, the carboxyl generation of polyamidoamines.
[0073] Amines of use in the invention include, but are not limited to, monoamines, polyamines, trimethylammonium, triethylammonium, tributyl ammonium, diethylmethylammonium, diisopropylethyl ammonium, triisopropylammonium, N-methylmorpholinium, N-ethylmorpholinium, N-hydroxyethylpiperidinium, N-methylpyrrolidinium, N,N-dimethylpiperazinium, isopropylethylammonium, isopropylmethylammonium, diisopropylammonium, tert-butylethylammonium, dicychohexylammonium, protonized forms of morpholine, pyridine, piperidine, pyrrolidine, piperazine, imidazole, tert-butylamine, 2-amino-2-methylpropanol, 2-amino-2-methyl-propandiol, tris-(hydroxyethyl)-aminomethane, diethyl-(2-hydroxyethyl)amine, tris-(hydroxymethyl)-aminomethane tetramethylammonium, tetraethylammonium, N-methylglucamine and tetrabutylammonium, polyethyleneimine, and polyamidoamine dendrimers.
[0074] Depending upon the permeability of the lipid vesicle membranes, the full transmembrane potential corresponding to the concentration gradient will either form spontaneously or a permeability enhancing agent, e.g., a proton ionophore can be added to the medium. If desired, the permeability enhancing agent can be removed from the liposome preparation after loading with the complex is complete using chromatography or other techniques.
[0075] Typically the temperature of the medium during active loading is between about -25.degree. C. and about 100.degree. C., e.g., between about 0.degree. C. and about 70.degree. C., e.g., between about 4.degree. C. and 65.degree. C.
[0076] The encapsulation or loading efficiency, defined as encapsulated amount (e.g., as measured in grams of agent/moles of phospholipid or g of drug/g total lipid) of the sparingly water-soluble agent in the internal aqueous phase divided by the initial amount in the external aqueous phase multiplied by 100%, is at least 10%, preferably at least 50%, at least 90%.
[0077] In an exemplary embodiment, the invention provides a method of loading a sparingly water-soluble agent into a liposome. An exemplary method comprises, contacting an aqueous suspension of said liposome with said aqueous suspension of said agent under conditions appropriate to encapsulate said sparingly water-soluble agent in said liposome, wherein said liposome has an internal aqueous environment encapsulated by a lipid membrane and said aqueous suspension of said liposome comprises a gradient selected from a proton gradient, an ion gradient and a combination thereof across said membrane, and wherein said conditions are appropriate for said sparingly water-soluble agent to traverse said membrane and concentrate in said internal aqueous environment, thereby forming said pharmaceutical formulation.
[0078] In various embodiments, the reaction mixture above is incubated for a selected period of time and the pH gradient, sulfate gradient, phosphate gradient, carboxylate gradient (citrate gradient, acetate gradient), EDTA ion gradient, ammonium salt gradient, alkylated ammonium salt gradient, Ca.sup.2+, Cu.sup.2+, Fe.sup.+2, Mg.sup.2+, Mn.sup.2+, Zn.sup.2+, Na.sup.+ gradient, K.sup.+ gradient or a combination thereof, exists across the liposomal membrane during the incubating.
[0079] In exemplary embodiments of the invention, the sparingly water-soluble therapeutic agent is not covalently attached to a component of the liposome, nor is it covalently attached to any component of the pH or salt gradient used to form the liposomal preparation of the invention.
Aprotic Solvent
[0080] In an exemplary embodiment, the sparingly water-soluble agent is completely dissolved in an aprotic solvent that is miscible with water. The agent solution is added to the aqueous liposome suspension at a concentration that is greater than the solubility of the drug agent in either the liposome suspension or the liposome suspension/aprotic solvent mixture, thus a precipitate is formed. Exemplary aprotic solvents include dimethylsulfoxide, dioxane, tetrahydrofuran, dimethylformamide, acetonitrile, dimethylacetamide, sulfolane, gamma butyrolactone, pyrrolidones, 1-methyl-2-pyrrolidinone, methylpyrroline, ethylene glycol monomethyl ether, diethylene glycol monomethyl ether, PEG400 and polyethylene glycols.
Sparingly Water-Soluble Agent Precipitate
[0081] The invention describes loading of an insoluble precipitate. An exemplary precipitate is conceptualized as some insoluble portion of the agent in suspension. The insoluble portion is defined as a portion of the agent that is not solvated as indicated by any of the following: any appearance of cloudiness greater than that of the liposome suspension in the absence of the agent, any degree of increased light scattering at a wavelength where the contents do not absorb light, such at 600 nm greater than the liposome suspension alone, any portion of the drug than can be isolated (pelleted) through centrifugation at a rate below 12,000 RPM for 15 min, any portion of the drug agent than can be isolated by filtration through 0.2 um filter.
Kits
[0082] In an exemplary embodiment, the invention provides a kit containing one or more components of the liposomes or formulations of the invention and instructions on how to combine and use the components and the formulation resulting from the combination. In various embodiments, the kit includes a the sparingly water-soluble agent in one vessel and a liposome preparation in another vessel. An exemplary liposome preparation includes a distribution of salt on the outside and inside of the lipid membrane to establish and/or maintain an ion gradient, such as that described herein. Also included are instructions for combining the contents of the vessels to produce a liposome or a formulation thereof of the invention. In various embodiments, the amount of complex and liposome are sufficient to formulate a unit dosage formulation of the complexed agent.
[0083] In an exemplary embodiment, one vessel includes a liposome or liposome solution, which is used to convert at least part of the contents of a vessel of a sparingly water-soluble therapeutic agent formulation (e.g., of an approved therapeutic agent) into a liquid formulation of the liposome encapsulated therapeutic agent at the point of care for administration to a subject. In an exemplary embodiment, the contents of the vessels are sufficient to formulate a unit dosage formulation of the therapeutic agent.
[0084] In the embodiment in which a unit dosage format is formed, the vessel includes from about 1 mg to about 500 mg of the therapeutic agent, e.g, from about 1 mg to about 200 mg, e.g., from about 5 mg to about 100 mg, e.g., from about 10 mg to about 60 mg.
[0085] In an exemplary embodiment, the approved therapeutic agent is carfilzomib and it is present in the vessel in an amount of from about 40 mg to about 80 mg, e.g., from about 50 mg to about 70 mg. In an exemplary embodiment, the carfilzomib is present in about 60 mg.
Methods of Treatment
[0086] In one aspect, the invention provides a method of treating a proliferative disorder, e.g., a cancer, in a subject, e.g., a human, the method comprising administering a composition that comprises a pharmaceutical formulation of the invention to a subject in an amount effective to treat the disorder, thereby treating the proliferative disorder.
[0087] In one embodiment, the pharmaceutical formulation is administered in combination with one or more additional anticancer agent, e.g., chemotherapeutic agent, e.g., a chemotherapeutic agent or combination of chemotherapeutic agents described herein, and radiation.
[0088] In one embodiment, the cancer is a cancer described herein. For example, the cancer can be a cancer of the bladder (including accelerated and metastatic bladder cancer), breast (e.g., estrogen receptor positive breast cancer; estrogen receptor negative breast cancer; HER-2 positive breast cancer; HER-2 negative breast cancer; progesterone receptor positive breast cancer; progesterone receptor negative breast cancer; estrogen receptor negative, HER-2 negative and progesterone receptor negative breast cancer (i.e., triple negative breast cancer); inflammatory breast cancer), colon (including colorectal cancer), kidney (e.g., transitional cell carcinoma), liver, lung (including small and non-small cell lung cancer, lung adenocarcinoma and squamous cell cancer), genitourinary tract, e.g., ovary (including fallopian tube and peritoneal cancers), cervix, prostate, testes, kidney, and ureter, lymphatic system, rectum, larynx, pancreas (including exocrine pancreatic carcinoma), esophagus, stomach, gall bladder, thyroid, skin (including squamous cell carcinoma), brain (including glioblastoma multiforme), head and neck (e.g., occult primary), and soft tissue (e.g., Kaposi's sarcoma (e.g., AIDS related Kaposi's sarcoma), leiomyosarcoma, angiosarcoma, and histiocytoma).
[0089] In an exemplary embodiment, the cancer is multiple myeloma. In one embodiment, the pharmaceutical formulation of the invention includes carfilzomib as the sparingly water-soluble therapeutic agent.
[0090] In one aspect, the disclosure features a method of treating a disease or disorder associated with inflammation, e.g., an allergic reaction or an autoimmune disease, in a subject, e.g., a human, the method comprises: administering a composition that comprises a Pharmaceutical formulation of the invention to a subject in an amount effective to treat the disorder, to thereby treat the disease or disorder associated with inflammation.
[0091] In one embodiment, the disease or disorder associated with iron overload such as occurs when a patient receives multiple units of blood transfusion such as occurs in thalassemia, sickle anemia, traumatic injury or after a bone marrow transplant. The iron overload may be local, such as can occur in endometriosis due to the extravasation of red blood cells into the local tissue where the provoke an inflammatory immune response.
[0092] In one embodiment, the disease or disorder associated with inflammation is a disease or disorder described herein. For example, the disease or disorder associated with inflammation can be for example, multiple sclerosis, rheumatoid arthritis, psoriatic arthritis, degenerative joint disease, spondouloarthropathies, gouty arthritis, systemic lupus erythematosus, juvenile arthritis, rheumatoid arthritis, osteoarthritis, osteoporosis, diabetes (e.g., insulin dependent diabetes mellitus or juvenile onset diabetes), menstrual cramps, cystic fibrosis, inflammatory bowel disease, irritable bowel syndrome, Crohn's disease, mucous colitis, ulcerative colitis, gastritis, esophagitis, pancreatitis, peritonitis, Alzheimer's disease, shock, ankylosing spondylitis, gastritis, conjunctivitis, pancreatitis (acute or chronic), multiple organ injury syndrome (e.g., secondary to septicemia or trauma), myocardial infarction, atherosclerosis, stroke, reperfusion injury (e.g., due to cardiopulmonary bypass or kidney dialysis), acute glomerulonephritis, vasculitis, thermal injury (i.e., sunburn), necrotizing enterocolitis, granulocyte transfusion associated syndrome, and/or Sjogren's syndrome. Exemplary inflammatory conditions of the skin include, for example, eczema, atopic dermatitis, contact dermatitis, urticaria, scleroderma, psoriasis, and dermatosis with acute inflammatory components. In some embodiments, the autoimmune disease is an organ-tissue autoimmune diseases (e.g., Raynaud's syndrome), scleroderma, myasthenia gravis, transplant rejection, endotoxin shock, sepsis, psoriasis, eczema, dermatitis, multiple sclerosis, autoimmune thyroiditis, uveitis, systemic lupus erythematosis, Addison's disease, autoimmune polyglandular disease (also known as autoimmune polyglandular syndrome), or Grave's disease.
[0093] In another embodiment, a pharmaceutical formulation of the invention or method described herein may be used to treat or prevent allergies and respiratory conditions, including asthma, bronchitis, pulmonary fibrosis, allergic rhinitis, oxygen toxicity, emphysema, chronic bronchitis, acute respiratory distress syndrome, and any chronic obstructive pulmonary disease (COPD). The pharmaceutical formulation of the invention, particle or composition may be used to treat chronic hepatitis infection, including hepatitis B and hepatitis C.
[0094] In one aspect, the disclosure features a method of treating cardiovascular disease, e.g., heart disease, in a subject, e.g., a human, the method comprising administering a a pharmaceutical formulation of the invention to a subject in an amount effective to treat the disorder, thereby treating the cardiovascular disease.
[0095] In one embodiment, cardiovascular disease is a disease or disorder described herein. For example, the cardiovascular disease may be cardiomyopathy or myocarditis; such as idiopathic cardiomyopathy, metabolic cardiomyopathy, alcoholic cardiomyopathy, drug-induced cardiomyopathy, ischemic cardiomyopathy, and hypertensive cardiomyopathy. Also treatable or preventable using a pharmaceutical formulation of the inventions, particles, compositions and methods described herein are atheromatous disorders of the major blood vessels (macrovascular disease) such as the aorta, the coronary arteries, the carotid arteries, the cerebrovascular arteries, the renal arteries, the iliac arteries, the femoral arteries, and the popliteal arteries. Other vascular diseases that can be treated or prevented include those related to platelet aggregation, the retinal arterioles, the glomerular arterioles, the vasa nervorum, cardiac arterioles, and associated capillary beds of the eye, the kidney, the heart, and the central and peripheral nervous systems. Yet other disorders that may be treated with pharmaceutical formulation of the invention, include restenosis, e.g., following coronary intervention, and disorders relating to an abnormal level of high density and low density cholesterol.
[0096] In one embodiment, the pharmaceutical formulation of the invention can be administered to a subject undergoing or who has undergone angioplasty. In one embodiment, the Pharmaceutical formulation of the invention, particle or composition is administered to a subject undergoing or who has undergone angioplasty with a stent placement. In some embodiments, the pharmaceutical formulation of the invention, particle or composition can be used as a strut of a stent or a coating for a stent.
[0097] In one aspect, the invention provides a method of treating a disease or disorder associated with the kidney, e.g., renal disorders, in a subject, e.g., a human, the method comprises: administering a pharmaceutical formulation of the invention to a subject in an amount effective to treat the disorder, thereby treating the disease or disorder associated with kidney disease.
[0098] In one embodiment, the disease or disorder associated with the kidney is a disease or disorder described herein. For example, the disease or disorder associated with the kidney can be for example, acute kidney failure, acute nephritic syndrome, analgesic nephropathy, atheroembolic renal disease, chronic kidney failure, chronic nephritis, congenital nephrotic syndrome, end-stage renal disease, good pasture syndrome, interstitial nephritis, kidney damage, kidney infection, kidney injury, kidney stones, lupus nephritis, membranoproliferative GN I, membranoproliferative GN II, membranous nephropathy, minimal change disease, necrotizing glomerulonephritis, nephroblastoma, nephrocalcinosis, nephrogenic diabetes insipidus, nephrosis (nephrotic syndrome), polycystic kidney disease, post-streptococcal GN, reflux nephropathy, renal artery embolism, renal artery stenosis, renal papillary necrosis, renal tubular acidosis type I, renal tubular acidosis type II, renal underperfusion, renal vein thrombosis.
[0099] In one embodiment, the disease or disorder is caused by a microbe or virus. These infectious agents can be viruses such as HIV, fungi such as aspergillosis, bacteria such as staphylococcus, protist, such as malaria or multicellular infectious agents, such as schistosomyosis.
[0100] An "effective amount" or "an amount effective" refers to an amount of the pharmaceutical formulation of the invention which is effective, upon single or multiple dose administrations to a subject, in treating a cell, or curing, alleviating, relieving or improving a symptom of a disorder. An effective amount of the composition may vary according to factors such as the disease state, age, sex, and weight of the individual, and the ability of the compound to elicit a desired response in the individual. An effective amount is also one in which any toxic or detrimental effects of the composition are outweighed by the therapeutically beneficial effects.
[0101] As used herein, the term "prevent" or "preventing" as used in the context of the administration of an agent to a subject, refers to subjecting the subject to a regimen, e.g., the administration of a pharmaceutical formulation of the invention such that the onset of at least one symptom of the disorder is delayed as compared to what would be seen in the absence of the regimen.
[0102] As used herein, the term "subject" is intended to include human and non-human animals. Exemplary human subjects include a human patient having a disorder, e.g., a disorder described herein, or a normal subject. The term "non-human animals" includes all vertebrates, e.g., non-mammals (such as chickens, amphibians, reptiles) and mammals, such as non-human primates, domesticated and/or agriculturally useful animals, e.g., sheep, dog, cat, cow, pig, etc.
[0103] As used herein, the term "treat" or "treating" a subject having a disorder refers to subjecting the subject to a regimen, e.g., the administration of a pharmaceutical formulation of the invention such that at least one symptom of the disorder is cured, healed, alleviated, relieved, altered, remedied, ameliorated, or improved. Treating includes administering an amount effective to alleviate, relieve, alter, remedy, ameliorate, improve or affect the disorder or the symptoms of the disorder. The treatment may inhibit deterioration or worsening of a symptom of a disorder.
[0104] The following examples are provided to illustrate exemplary embodiments of the invention and are not to be construed as limiting the scope of the invention.
EXAMPLES
Example 1
Carfilzomib Liposome Entrapment by Remote Loading
Materials and Method
[0105] Ammonium sulfate solution was prepared by dissolving ammonium sulfate solid to a final concentration of 250 mM (500 mequivilents of anion/L) no pH adjustment was made to yield a final pH of 5.6. Sodium sulfate solution (250 mM) was prepared by adding 0.35 g sodium sulfate to 10 mL deionized water.
[0106] The liposomes were formed by extrusion. Lipids were dissolved in ethanol at a concentration of 500 mM HSPC (591 mg/mL total lipid) at 65.degree. C. and the 9 volumes of the trapping agent solution heated to 65.degree. C. was added to the ethanol/lipid solution also at 65.degree. C. The mixture was vortexed and transferred to a 10 mL thermostatically controlled (65.degree. C.) Lipex Extruder. The liposomes were formed by extruding 10 times through polycarbonate membranes having 0.1 um pores. After extrusion the liposomes were cooled on ice. The transmembrane electrochemical gradient was formed by purification of the liposomes by dialysis in dialysis tubing having a molecular weight cut off of 12,000-14,000. The samples are dialyzed against 5 mM HEPES, 10% sucrose pH 6.5 (stirring at 4.degree. C.) at volume that is 100 fold greater than the sample volume. The dialysate was changed after 2 h then 4 more times after 12 h each. The conductivity of the liposome solution was measured and was indistinguishable from the dialysis medium .about.40 .mu.S/cm.
[0107] The lipid concentration is determined by measuring the cholesterol by HPLC using an Agilent 1100 HPLC with and Agilent Zorbax 5 um, 4.6.times.150 mM, Eclipse XDB-C8 column and a mobile phase of A=0.1% TFA, B=0.1% TFA/MeOH with an isocratic elution of 99% B. The flow rate is 1.0 mL/min, column temperature is 50.degree. C., 10 .mu.L injection and detection by absorbance at 205 nm. The retention time of cholesterol is 4.5 min. The liposome size is measured by dynamic light scattering.
[0108] Carfilzomib (Selleck Chemicals) was dissolved in DMSO at a concentration of 10 mg/mL. The carfilzomib was introduced to the liposomes at a carfilzomib to HSPC ratio of 100 g drug/mol HSPC (drug to total lipid ratio (wt/wt) of 0.12). The liposomes were diluted with 50 mM citrate, 10% sucrose pH 4.0 to increase the volume to a point where after addition of the drug the final DMSO concentration is 2%. The carfilzomib/DMSO was added to the diluted liposomes, which were mixed at room temperature then transferred to a 65.degree. C. bath and swirled every 30 s for the first 3 min and then swirled every 5 min over a total heating time of 30 min. All samples were very cloudy when the drug was added and all became clear (same as liposomes with no drug added) after 15 min. After heating for 30 min all samples were placed on ice for 15 min. The loaded liposomes were vortexed and 100 .mu.L of sample was kept as the "before column" and the rest transferred to microcentrifuge tubes and spun at 10,000 RPM for 5 min. The supernatants were purified on a Sephadex G25 column collected and analyzed by HPLC. The HPLC analysis of carfilzomib was performed on the same system as described for analysis of cholesterol. The mobile phase consists of A=0.1% TFA, B=0.1% TFA/MeOH with a gradient elution starting at 50% B and increasing to 83% B in 13 min with 7 min equilibration back to 50% B. The flow rate is 1.0 mL/min, column temperature is 30 C, 10 .mu.l injection and detection by absorbance at 205 nm. The retention time of carfilzomib is 12.2 min. The lipid concentration is determined by analysis of the cholesterol by HPLC.
Results
[0109] The loading of liposomes containing 250 mM ammonium sulfate resulted in a final drug to lipid ratio of 95.26.+-.3.47 g drug/mol of HSPC liposomes when the drug was added at 100 g drug/mol of HSPC lipid (95.26.+-.3.47% efficient) and a final drug to lipid ratio of 136.9.+-.7.35 g drug/mol of HSPC liposomes when the drug was added at 200 g drug/mol of HSPC lipid (67.94.+-.3.67% efficient) (FIG. 2). This demonstrates that the loading capacity of these particular liposomes is between 100 and 200 g drug/mol phospholipid. The control liposomes containing 250 mM sodium sulfate which have no electrochemical gradient for remote loading resulted in a final drug load of 33.28.+-.0.79 and 29.01.+-.0.79 g drug/mol of HSPC when the drug was added at a ratio of 100 and 200 g drug/mol of HSPC respectively. This demonstrates that the capacity for loading these liposomes was saturated below 100 g drug/mol of HSPC and at this drug input ratio the remote loaded liposomes exhibit >3 fold higher loading capacity. Saturation of the drug loading capacity for sodium sulfate liposomes at a ratio at least 3 fold lower than the ammonium sulfate liposomes indicates that when no electrochemical gradient is present for remote loading the drug partitions into the lipid bilayer but does not form a salt with the interior trapping agent. FIG. 5 illustrates the precipitate is still present after the loading process with sodium sulfate liposomes but not with ammonium sulfate liposomes.
Conclusion
[0110] Liposomes of identical lipid matrix composition and size but varying in the composition of the sulfate salt internally trapped had very different capabilities to load carfilzomib. The liposome capable of generating an electrochemical gradient (ammonium sulfate) was able to load close to 100% of the drug at optimal conditions and the one incapable of creating a gradient had poor loading efficiency suggesting that remote or active loading was the primary mechanism for carfilizomib incorporation into the liposome.
Example 2
Comparison of Liposome Trapping Agents
Introduction
[0111] Liposomes to be used for remote loading are formed in an ionic solution that is intended to complex the loaded drug as a salt. Trapping agents can form complexes with loaded drugs and the stability of this complex is one factor that dictates liposome drug loading ability, stability and drug release rates. Comparison of different liposome trapping agents was made by evaluating the efficiency of carfilzomib loading.
Methods
[0112] Three liposome formulations were used, all at a molar ratio 3 HSPC/2 Chol/0.15 PEG-DSPE each with a different trapping agent: 1. mellitic acid; 2. ammonium sulfate; and 3. napthelene disulfononic acid.
[0113] Mellitic acid (MA) was dissolved in water and titrated with diethylamine to a final pH of 5.5 and concentration of 83 mM (500 mequivilents of anion/L). Ammonium sulfate was prepared by dissolving ammonium sulfate solid to a final concentration of 250 mM (500 mequivilents of anion/L) no pH adjustment was made to yield a final pH of 5.6.
[0114] Napthelenedisulfonic acid (NDS) was dissolved in water and titrated with diethylamine to a final pH of 8.0 and concentration of 250 mM (500 mequivilents of anion/L).
[0115] See Example 1, Carfilzomib liposome entrapment by remote loading for details on how the liposomes were made, purified and characterized.
TABLE-US-00001 TABLE 1 Sizes of Liposomes Loaded with Carfilzomib. before lyophilizing Z-ave Trapping agent (nm) PDI (NH4).sub.2SO.sub.4 108 0.062 (Drug added quickly) (NH4).sub.2SO.sub.4 109.2 0.035 (Drug added slowly) Napthelenedisulfonic acid 111.9 0.039 Mellitic Acid 105.5 0.08
[0116] To ensure complete removal of the DMSO added with carfilzomib, the liposomal carfilzomib samples were dialyzed in dialysis tubing having a molecular weight cut off of 12,000-14,000. The samples are dialyzed against 5 mM HEPES, 10% sucrose pH 6.5 (stirring at 4.degree. C.) at volume that is 100 fold greater than the sample volume. The dialysate was changed after 2 h then 2 more times after 12 h each. The carfilzomib liposomes were again analyzed for drug and lipid concentration as described above.
Results
[0117] The efficiency of carfilzomib remote loading into liposomes at 100 g drug/mol of HSPC lipid for liposomes with the trapping agents mellitic acid, ammonium sulfate, and napthelenedisulfonic acid were 37.4%.+-.2.01%, 97.0%.+-.2.38%, and 95.1%.+-.1.76% respectively (FIG. 3).
Conclusion
[0118] The invention described here enables remote loading of carfilzomib from an insoluble precipitate into liposomes can be accomplished with various using the electrochemical gradient generated by various trapping agents including mellitic acid, ammonium sulfate and napthelene disulfononic acid.
Example 3
Comparison of Method for Introducing Drug
Method
[0119] A comparison of the method used for addition of the drug to the liposomes during the loading procedure. The loading procedure was the same as described above in Example 1 with the exception of the drug being added to the liposome loading solution as a solid powder, as a 10 mg/mL DMSO solution quickly and as 10 mg/mL DMSO solution slowly.
Results
[0120] The efficiency of carfilzomib remote loading into liposomes at 100 g drug/mol of HSPC lipid for the drug which was added as the solid powder was 3.88%.+-.0.053% and 3.47%.+-.0.030% when heated to 65.degree. C. for 30 and 120 min respectively. The efficiency of loading the drug as a 10 mg/mL DMSO solution was 97.0%.+-.2.38% when the drug/DMSO was added quickly and 96.3%.+-.1.09% when the drug/DMSO was added in 5 increments over 1 min to a liposome solution while vortexing. The drug/liposome mixture that results from the slow drug addition is clearer than the drug/liposome mixture that results from rapid addition of the drug. However, both solutions have no visible precipitate (or centrifugal precipitate at 10,000 rpm for 5 min) after heating to 65.degree. C. for 30 min, which is a result of all of the drug being loaded into the liposomes regardless of the precipitate formed upon addition of the drug (FIG. 4).
Example 4
[0121] Carfilzomib Loading into Liposomes at Room Temperature
Introduction
[0122] The ability to load a drug into liposomes at room temperature is beneficial to reduce heat-induced drug degradation, simplify manufacturing and allow for bedside liposome loading. Efficient transport across the liposome membrane requires the membrane to be in a fluid phase. This is accomplished with saturated phospholipids having a high phase transition temperature (T.sub.m) such as HSPC (T.sub.m=55.degree. C.) by heating the liposomes above the T.sub.m during the loading process. An alternative to heating is to use lipids that are fluid phase at room temperature. The disadvantage of these lipids is that they are unstable in circulation and result in rapid drug release. Sterol modified lipids incorporate a novel lipid construction where cholesterol (sterol) is covalently attached to the phosphate headgroup. Sterol modified lipids have proven to render the sterol non-exchangable from the lipid bilayer in circulation. Sterol modified lipids are also fluid phase at room temperature, making them ideal for room temperature loading of drugs into liposomes that are to be used for in vivo delivery of therapeutics.
Method
[0123] The loading of carfilzomib into liposomes at room temperature was performed by using two liposome formulations composed of a molar ratio of 95 PChemsPC/5 PEG-DSPE and another with a molar ratio of 3 POPC/2 Chol/0.15 PEG-DSPE each containing 250 mM ammonium sulfate as the trapping agent. The liposomes were prepared using the procedure, drug/liposome ratio, buffers ad pH as described in Example 1. The liposomes were stirred at room temperature (20.degree. C.) and the carfilzomib was added as a 10 mg/mL DMSO solution in 5 increments over 1 min to result in a cloudy solution. The liposome/drug mixture was stirred at room temperature for a total of 30 min to yield a clear solution with the same appearance as the liposome solution before the drug was added.
Results
[0124] The efficiency of carfilzomib remote loading into liposomes at 100 g drug/mol of PChemsPC was 95.5%.+-.1.23% The efficiency of carfilzomib remote loading into liposomes at 187 g drug/mol of 3 POPC/2 Chol/0.15 PEG-DSPE was 100.52%.+-.1.01%
Conclusion
[0125] The invention described here was not able to load carfilzomib into liposomes by adding the crystal form of the drug directly to the loading solution. The drug requires solubilization in some solvent prior to addition to the loading solution at a concentration above the solubility of the drug. Liposome loading efficiency of the precipitate that is formed upon addition of the drug to the loading solution is not dependent upon the rate of addition in this case using carfilzomib.
Example 5
[0126] Drug Precipitate Loading into Liposomes as Determined by Light Scattering at 600 nm.
Introduction
[0127] Liposome loading of drug from a precipitate into liposomes is evidenced by the resulting drug to lipid ratio and clarifying of the solution as the drug precipitate transfers into the liposome. To get a quantitative measure liposome loading from a drug precipitate the light scattering was measured at 600 nm during the loading process.
Method
[0128] Liposomes containing 250 mM ammonium sulfate as trapping agent and 250 mM sodium sulfate as control liposomes which would not remote load drug. The liposomes were prepared and loaded using the procedure described in Example 1 except a disposable polystyrene cuvette was used as the reaction vessel. The scattering of light at 600 nm was measured with a UV/vis spectrophotometer during the loading process.
Results/Conclusion
[0129] The sodium sulfate liposomes do not show any clarification of the precipitate during the loading procedure indication that the drug is not remote loading into the liposomes. (see FIG. 5). The ammonium sulfate liposomes efficiently load the drug resulting in clarification of the solution within 15 min.
Example 6
[0130] Confirmation of Drug Release from Remote Loaded Liposomes
Introduction
[0131] A reverse gradient was used to attempt to release the active drug from within the liposome. The theory is that if a drug can be released from within a liposome with a reverse gradient there is a good chance the that drug release will be possible in vivo.
Method
[0132] Liposomes were loaded with carfilzomib as described in Example 1 and were purified into deionized water. The sample was divided into two aliquots. To the first aliquot, concentrated Hepes pH 7.4 and NaCl was added so that the final concentration was 5 mM Hepes, 145 mM NaCl (HBS). To the second aliquot, concentrated ammonium sulfate was added so that the final concentration was 250 mM. No obvious physical changes were initially observed. The samples were then heated at 65.degree. C. for 30 min. The samples were transferred to clean eppendorf tubes and centrifuged for 10,000 rpm for 5 min after which the supernatants and precipitates were separated and tested for carfilzomib content by HPLC assay. Released drug precipitated, liposome encapsulated drug remained in the supernatant. The % carfilzomib released was calculated by
% Release = amt . of drug in precipitate amt . of total drug ##EQU00001##
Results
TABLE-US-00002 [0133] TABLE 2 The reverse gradient-directed drug release from liposomes Solution Composition % Carfilzomib Released Hepes buffered saline 10.6 .+-. 0.28 Ammonium sulfate 68.5 .+-. 1.82
[0134] The drug released using the reverse gradient is 6.5-fold greater than the drug released from the control with no reverse gradient (Table 2). HPLC chromatogram of the released drug was identical to the starting material indicating that no degradation of carfilzomib had taken place (FIG. 7). HPLC retention time for the stock solution of carfilzomib was 12.15 min and the retention time for the carfilzomib that was released from the remote loaded liposome was 12.27 min, as shown in FIG. 6.
Conclusion
[0135] Carfilzomib was released from the liposome using a reverse gradient to yield the original molecule as indicated by HPLC analysis.
Example 7
Carfilzomib Loading as a Function of DMSO Content
Introduction
[0136] The physical form of the drug when added to liposomes is important for loading efficiency, i.e., when added as a dry powder almost no loading is observed but addition using a predissolved solution in an aprotic solvent can lead to high entrapment efficiency. This study looks at the effect of aprotic solvent concentration on drug loading efficiency of carfilzomib.
Method
[0137] Ammonium sulfate containing liposomes were diluted in 50 mM citric acid sucrose (10% wt/wt) buffer pH 4.0 to 1 mM phospholipid. Various amounts of DMSO were added so that when 200 .mu.g drug was added from a 10 mg/mL carfilzomib solution in DMSO the final DMSO concentration ranged from 1-10% v/v.
Results
[0138] DMSO had a dramatic effect on the ability of carfilzomib to remote load into liposomes. When absent, there is practically no loading. At concentrations 1% and above the loading efficiency ranges from 74-94%, with higher efficiencies observed at higher DMSO concentrations (FIG. 7). It should be noted that drug precipitates were observed in all samples before loading commenced, suggesting that the concentrations of DMSO used here are below the minimum concentration required to effectively solubilize carfilzomib at the drug concentration used (0.2 mg/mL).
Conclusions
[0139] The introduction of pre-solubilized carfilzomib is necessary for efficient remote loading. However, above 1% DMSO there is a relatively small change in loading efficiency, up as far as 10%.
Example 8
[0140] Carfilzomib Solubility as a Function of DMSO Content
Introduction
[0141] Under the conditions described above, carfilzomib is solubilized in DMSO before diluting in liposome buffer solution prior to loading. It then immediately precipitates before remote loading. This study is designed to determine the DMSO concentration that is required to effectively solubilize carfilzomib at room temperature and at the temperature required for liposome loading into liposomes composed of high T.sub.m lipids (65.degree. C.).
Method
[0142] Carfilzomib was added from a stock 10 mg/mL solution in DMSO to 1 mL of a citric acid/DMSO mixture so that the composition of DMSO was 2%, 25%, 50%, 75% and 100%. The final drug concentration was 0.2 mg/mL. The solutions were prepared and measured for optical density at 600 nm. The optical density at 600 nm is a good measure of how turbid or how much scattering material (such as drug precipitates) are in a solution, generally, the more precipitates the higher the absorbance. From FIG. 8 is apparent that at DMSO concentrations below 50% vol/vol (25.degree. C.) and 25% vol/vol (65.degree. C.) the drug remains in a precipitated form. Only when the concentration of DMSO is increased does it become effectively solubilized at this concentration of 0.2 mg/mL.
[0143] To test the integrity of the liposomes in 25% DMSO we attempted to remote load the water-soluble weak base drugs doxorubicin and 17-dimethylaminoethylamino-17-demethoxygeldanamycin (17-DMAG) and compared to the same loading without DMSO. We found that the loading efficiency was adversely affected (Table 3).
Results
[0144] At 0.2 mg/mL carfilzomib the drug precipitates and the aggregates are large enough to cause a light scattering signal at 600 nm. As the % DMSO is increased the signal is reduced and indicates solubilization of the drug. We observed that >25% vol/vol DMSO is required to completely dissolve the drug at 0.2 mg/mL at a temperature of 65.degree. C.
TABLE-US-00003 TABLE 3 Comparison of remote loading doxorubicin and 17-DMAG into ammonium sulfate containing liposomes in the presence and absence of 25% DMSO. % Efficiency compared to Drug % DMSO control of no DMSO doxorubicin 25 92.7 .+-. 0.43 17-DMAG 25 73.1 .+-. 1.4
Conclusions
[0145] Previous studies loading carfilzomib were done using 10% v/v DMSO or less and the light scattering results above show that the vast majority of the drug under these conditions is in a precipitated form at the concentrations used. Adding enough aprotic solvent to completely solubilize the drug (i.e., greater than 25% DMSO at 65.degree. C.) has a negative impact of the liposome loading of amphipathic weak base drugs indicating liposome instability caused by contents leakage or electrochemical gradient dissipation for example. Under conditions that maintain good liposome stability, we have not found a DMSO concentration that will solubilize carfilzomib completely or alternatively we have not found conditions using DMSO where simultaneously the drug is completely solubilized and the liposomes are not adversely destabilized.
Example 9
[0146] Effect of Delay on Liposome Loading of Carfilzomib after the Drug Precipitate is Formed
Introduction
[0147] The invention described in this application allows for loading of an insoluble drug precipitate into liposomes. Example 9 evaluates the effect of the time between the formation of the drug precipitate and the time it is loaded into liposomes.
Procedure
[0148] Liposomes were prepared from the same composition and methods as described in Example 1.
[0149] Carfilzomib was dissolved in DMSO at a concentration of 10 mg/mL and we added to a final concentration of 2% (v/v) to 50 mM citrate, 10% sucrose at pH 3.5 containing no liposomes. Upon addition of the drug to the citrate buffer a precipitate was formed. The liposomes for loading were added to the solution containing drug precipitate either immediately after formation, after a 1 h delay or after a 12 h delay and then the precipitate was loaded into the liposomes using the loading conditions described in Example 1.
Results/Conclusion
[0150] The time between the formation of the drug precipitate and the loading of the precipitate does not have a significant impact on the efficiency of the liposome loading procedure for carfilzomib even if the delay time is up to 12 h.
Example 10
[0151] Effect of Liposome Drug Payload on Efficiency of Carfilzomib Loaded from Precipitate Procedure
[0152] Liposomes were prepared from the same composition and methods as described in Comparison of Trapping Agents except the concentration of ammonium sulfate internal trapping agent was either 250 mM or 500 mM.
[0153] Carfilzomib was dissolved in DMSO at a concentration of 10 mg/mL. The carfilzomib was introduced to the liposomes at carfilzomib to HSPC ratios of 91.8, 167, 251, 338 and 433, g drug/mol HSPC for the liposomes having 250 mM ammonium sulfate as the trapping agent and 451, 546, 639, and 759 g drug/mol HSPC for the liposomes having 500 mM ammonium sulfate as the trapping agent. The liposomes were diluted with 50 mM citrate, 10% sucrose pH 4.0 to increase the volume to a point where after addition of the drug the final DMSO concentration is 10%. The carfilzomib/DMSO was added to the diluted liposomes, which were mixed at room temperature then transferred to a 65.degree. C. bath and swirled every 30 s for the first 3 min and then swirled every 5 min over a total heating time of 30 min. All samples were very cloudy when the drug was added and all became clear (same as liposomes with no drug added) after 15 min. After heating for 30 min all samples were placed on ice for 15 min. The loaded liposomes were purified and analyzed as described in Example 1.
Results/Conclusion
TABLE-US-00004 [0154] TABLE 4 Effect of Ammonium Sulfate Trapping Agent Concentration on Liposome Drug Payload of Carfilzomib Loaded from Precipitate Input Drug trapping payload/carrier agent Output weight ratio drug/HSPC SD [(NH.sub.4).sub.2SO.sub.4] drug/HSPC SD SD (g drug/g total (g/mol) (g/mol) (mM) (g/mol) (g/mol) efficiency % (g/mol) lipid) 91.8 0.3 250 83.5 2.7 90.9 3.0 0.07 167.3 4.2 250 127.2 1.5 76.0 2.1 0.11 251.8 5.8 250 174.1 3.9 69.1 2.2 0.15 338.1 4.1 250 210.7 2.5 62.3 1.1 0.18 432.8 14.4 250 240.5 4.4 55.6 2.1 0.20 450.6 9.6 500 345.2 7.1 76.6 2.3 0.29 545.9 17.1 500 380.9 21.4 69.8 4.5 0.32 639.2 42.5 500 438.9 10.2 68.7 4.8 0.37 758.7 12.4 500 468.2 4.9 61.7 1.2 0.40
[0155] The resulting drug payload increases as the drug to liposome input lipid ratios is increased in the loading solution. The efficiency is greatest at the lowest input ratio used for each different concentration of ammonium sulfate trapping agent. Using the conditions described in this example, carfilzomib can be loaded into liposomes from an insoluble precipitate up to a final drug payload of 469.+-.4.9 g drug/mol HSPC (drug/carrier total lipid weight ratio of 0.4) at an efficiency of 61.7.+-.1.2%.
Example 11
[0156] Loading of Insoluble Carfilzomib into Liposomes Using a Triethylammonium Sulfate Gradient
Introduction
[0157] Remote loading of drugs into liposomes is commonly accomplished using an ammonium sulfate gradient. Some drug molecules including the example carfilzomib used here have an epoxide group which is required for activity. The epoxide of these drugs is potentially susceptible to aminolysis from any remaining ammonia that is used in the ammonium sulfate remote loading. In this, Example 11, the ammonium sulfate is replaced with a triethylammonium sulfate remote loading agent to eliminate potential ammonia/epoxide reactions by replacement with nonreactive triethylamine.
Methods
[0158] The liposomes were prepared by using the same compositions and procedure as described in Carfilzomib Liposome Entrapment by Remote Loading with the following exception that 50 mM triethylammonium sulfate was used as the trapping agent. Triethylammonium Sulfate was prepared by titrating 1 M sulfuric acid with triethylamine to a final pH of 7.3 and sulfate concentration of 500 mM.
[0159] Carfilzomib was dissolved in DMSO at a concentration of 10 mg/mL. The carfilzomib was introduced to the liposomes at carfilzomib to HSPC ratios of 650 g drug/mol HSPC. The liposomes were diluted with 50 mM citrate, 10% sucrose pH 4.0 to increase the volume to a point where after addition of the drug the final DMSO concentration is 10%. The carfilzomib/DMSO was added to the diluted liposomes, which were mixed at room temperature then transferred to a 65.degree. C. bath and swirled every 30 s for the first 3 min and then swirled every 5 min. A sample of the loading mixture was removed at 10, 20, 30 and 40 min during the loading procedure and placed on ice for 15 min. The loaded liposomes were vortexed and 100 .mu.L of sample was kept as the "before column" and the rest transferred to microcentrifuge tubes and spun at 10,000 RPM for 5 min. The supernatants were purified on a Sephadex G25 column collected and analyzed by HPLC. The drug precipitate pellets were dissolved in DMSO/MeOH (10:1) and analyzed by HPLC.
Results/Conclusion
[0160] Loading an insoluble carfilzomib precipitate into liposomes using a triethylammonium sulfate gradient results in similar liposomes to those produced using an ammonium sulfate gradient (Example 1). FIG. 12 illustrates the time dependence on the liposome loading, which begins quickly by 10 min. The greatest payload achieved was 440.+-.12.6 g drug/mol HSPC (efficiency of 65.9.+-.1.98%) was achieved at 30 min. This result using 500 mM triethylamine as a trapping agent at drug to HSPC ratios of 650 g drug/mol HSPC is very similar to that using 500 mM ammonium sulfate as the trapping agent drug to HSPC ratios of 639 g drug/mol HSPC which resulted in a final drug to lipid ratio of 440.+-.10.2 g drug/mol HSPC (efficiency of 68.7.+-.4.80%).
[0161] The insoluble drug precipitate on the liposome exterior is transferred (remote loaded) to the liposome interior as indicated a reduction in the amount of precipitate in the mixture over the course of the loading process. FIG. 12 shows the greatest reduction in the extraliposomal precipitate happens between 0-10 min which corresponds to the loading of precipitate into liposomes as seen in FIG. 13.
Example 12
[0162] Loading Another Sparingly Soluble Drug from a Precipitate
Introduction
[0163] Another drug, aripiprazole is formulated with sulfobutyl cyclodextran (SBCD) and is used to treat bipolar disorders and schizophrenia (Abilify, Pfizer). The drug is very insoluble in water and when added to a liposome suspension, fine precipitates are immediately observed.
[0164] Whether aripiprazole would remote load under similar conditions outlined above for carfilzomib was tested.
Method
[0165] Liposomes (HSPC/Chol/PEG-DSPE 3/2/0.15 mol/mol/mol) containing 250 mM ammonium sulfate or 250 mM sodium sulfate were diluted in 1 mL of 50 mM citric acid, 10% (wt/wt) sucrose, pH 4.0 to a concentration of 6 mM phospholipid. 0.3 mg of aripiprazole was added from a stock solution of 15 mg/mL in DMSO, so that the final DMSO concentration was 2% (v/v). Fine precipitates were immediately observed after the drug was added to both liposome samples. The samples were heated at 65.degree. C. for 30 min, the cooled on ice. The samples were filtered through a 0.2 um polyethersulfone syringe filter to remove any drug precipitates, followed by purification on a Sephadex G25 column equilibrated with HBS, pH 6.5 to remove any soluble extraliposome drug. The turbid fraction was collected and analyzed for lipid and drug as described above.
Results
TABLE-US-00005 [0166] TABLE 5 Results of loading aripiprazole into liposomes containing ammonium and sodium sulfate. Input % Efficiency D/L Output D/L % NH4SO4/ Loading Agent ug/umol ug/umol Efficiency NaSO4 (NH.sub.4).sub.2SO.sub.4 50 42.28 .+-. 0.49 84.56 .+-. 0.99 49.3 (Na).sub.2SO.sub.4 50 0.86 .+-. 0.51 1.71 .+-. 0.10
[0167] The liposomes containing ammonium sulfate were found to load approximately 85% of the drug, while the loading into sodium sulfate liposomes was less than 2%, with about a 50-fold increase in loading attributable to the ability of ammonium sulfate liposomes to facilitate remote loading (Table 5).
[0168] Ariprazole, when introduced to the liposome solution in the form of a SBCD complex (from the pharmaceutical product Abilify) gave a loading efficiency of 68% under the same concentration and loading conditions (FIG. 14).
Conclusion
[0169] This is another example of a poorly soluble drug, that can be remote loaded into liposomes using the approach described above, and gives slightly better loading than if the drug was introduced as a SBDC complex.
Example 13
[0170] Loading Sparingly Soluble Drug from Precipitates Made by Diluting Various Drug Solvent Solutions into Liposome Solution
[0171] This Example describes a technique for remote loading poorly soluble drugs into liposomes that begins with dissolving the drug in a solubilizing agent that initially forms drug precipitates when added to an aqueous solution of liposomes. After some incubation time the drug enters the liposome in response to an electrochemical gradient, accumulating in the liposome core. Solvents that may be used include but not limited to dimethylsulfoxide, dioxane, tetrahydrofuran, dimethylformamide, acetonitrile, dimethylacetamide, sulfolane, gamma butyrolactone, pyrrolidones, 1-methyl-2-pyrrolidinone, methylpyrroline, ethylene glycol monomethyl ether, diethylene glycol monomethyl ether, polyethylene glycol.
Method
[0172] Aripiprazole was dissolved in a range of solvents indicated below at 4 mg/mL. Liposomes composed of HSPC/Chol/PEG-DSPE (3/2/0.15 mol/mol/mol) that were prepared in 250 mM ammonium sulfate were used and diluted to 6 mM in Hepes buffered sucrose 10% (wt/wt) (HBSuc pH 6.5). 0.3 mg of drug was introduced by slow addition of each solvent while vortexing. The final solvent concentration was 7.5% for all samples. As controls, the drug was added from each solvent to the same volume of HBSuc pH 6.5 without the liposomes. The samples were heated at 65.degree. C. for 30 min then cooled on ice. After reaching room temperature again, the samples were measured for absorbance at 600 nm (Cary 100 Bio UV-Vis spectrometer) and the values are displayed below (FIG. 15).
Results
[0173] All the solutions without liposomes became extremely turbid or there was gross precipitation and settling (especially in the case of methanol and 1-butanol). Some of the liposome samples were also very turbid, but some clarified the drug precipitate consistent with earlier results indicating drug loading of the drug precipitate had taken place (namely in the cases where the drug was initially dissolved in DMSO, 1-4-methylpyrrolidone, diethylenemonoethylether or polyethyleneglycol (MW400), see FIG. 15.
Example 14
[0174] Remote Loading of an Insoluble Precipitate of Deferasirox into Liposomes Using an Acetate Gradient
[0175] Remote loading of deferasirox into liposomes containing calcium acetate demonstrates the use of an acetate gradient for loading an iron chelating agent. Calcium acetate gradient remote loading differs from ammonium sulfate remote loading in that the drug molecule being loaded must have a carboxylate (or hydroxamate) rather than an amine. Deferasirox is known to have significant kidney toxicity and liposome delivery is a technique for reducing kidney toxicity.
Method
[0176] The remote loading of a deferasirox insoluble precipitate into iposomes using an acetate gradient is performed in the same manner as acetate loading of soluble carboxyfluoroscein and nalidixic acid by Clerc and Barenholtz 1995 (PMID 8541297). Liposomes are prepared as described in Example 1 but in this case the liposomes are extruded in a solution of 120 mM calcium acetate at pH 8. The acetate gradient is formed by exchanging the external media for 120 mM sodium sulfate at pH 6.0. Deferasirox is dissolved in DMSO at a concentration of 10 mg/ml and added to the liposome suspension where it forms a precipitate. The precipitate is loaded into the liposomes by heating to 65.degree. C. for 1 h and purification and analysis is performed as described in Example 1.
Results
[0177] Deferasirox forms a precipitate when diluted from a 10 mg/ml DMSO stock to a concentration of 1 mg/ml in the liposome loading suspension due to its poor water solubility (.about.0.038 mg/mL). The insoluble deferasirox precipitate is loaded into the liposomes using a calcium acetate gradient at an efficiency at least 5-fold greater than it is loaded into control liposomes which contain sodium sulfate and no acetate gradient.
[0178] Remote loading an insoluble precipitate of deferasirox into the liposome provides an example of the use of an acetate gradient to remote load a carboxylate drug from a precipitate. In this example the drug being loaded is a chelating agent, in particular an iron chelating agent. The 5-fold greater loading into the liposomes having an acetate gradient over control liposomes indicates that the majority of the deferasirox is remote loaded rather than intercalated in the lipid bilayer.
Example 15
Introduction
[0179] One goal of liposomal delivery of carfilzomib is to protect the drug from degradation and elimination which required the drug to be retained within the liposome. One technique for evaluating the drug retention within the liposome, and thus the benefits obtained from liposome delivery, is to measure the pharmacokinetics of the drug in mice. Stable formulations with greater drug retention within the liposome will result in a higher concentration of non-metabolized drug in mouse plasma compared to less stable formulations or unencapsulated drug.
Materials and Methods
[0180] 100 nm liposomes comprised of HSPC/Cholesterol/PEG-DSPE (60/40/5 mol/mol/mol) and sphingomyelin/cholesterol/PEG-DSPE (55/45/2.8, mol/mol/mol) were formed, purified and drug loaded with carfilzomib using the methods described in Example 1. The trapping agents used to remote load carfilzomib were triethylammonium dextran sulfate (1.0 M SO.sub.4) or triethylammonium sucroseoctasulfate (1.0 M SO.sub.4). The drug loaded liposomes were purified by tangential flow filtration with buffer exchange into HBS, pH 6.5. The liposomes were sterile filtered through 0.2 um polyethersulfone filters and assayed for carfilzomib and lipid content as described in Example 1. The drug-to-lipid ratio, drug concentration and loading efficiency were calculated and results shown in Table 6.
Results.
TABLE-US-00006 [0181] TABLE 6 Carfilzomib concentration in mouse plasma after IV. administration of liposome formulations. Drug Lipid Formulation Loading CFZ/PL # (mol/mol/mol) Trapping Agent Efficiency (.mu.g/.mu.mol) % ID @ 4 h 1 HSPC/Chol/PEG- Ammonium Sucrose 94.1 .+-. 0.43 329.2 .+-. 2.26 0.65 .+-. 0.29 DSPE (60/40/5) Octasulfate (1.0M SO.sub.4) 2 HSPC/Chol/PEG- Triethylammonium 94.6 .+-. 7.55 381 .+-. 12.1 4.48 .+-. 1.10 DSPE (60/40/5) Dextran Sulfate (1.0M SO.sub.4) 3 HSPC/Chol/PEG- Triethylammonium 94.6 .+-. 7.55 381 .+-. 12.1 5.53 .+-. 1.69.dagger. DSPE (60/40/5) Dextran Sulfate (1.0M SO.sub.4) 4 SM/Chol/PEG-DSPE Triethylammonium 80.4 .+-. 0.71 321.8 .+-. 7.98 66.3 .+-. 20.3 (55/45/2.8) Dextran Sulfate (1.0M SO.sub.4) .dagger.formulation #3 is the same as #2 except it was stored at 4.degree. C. for 30 days before PK analysis
[0182] In addition, we examined the pharmacokinetics of carfilzomib encapsulated in the liposome formulations in male CD1 mice. The mice were dosed by IV bolus injection through the tail vein at 5 mg/kg carfilzomib using 3 mice per formulation. At 4 h, the mice were sacrificed and plasma harvested by centrifugation of the blood. 0.1 mL of plasma was mixed with 0.2 mL methanol, mixed well and carfilzomib concentration measured by HPLC as described in Example 1. The results are shown in Table 6. While no effort was made to distinguish between non-liposome entrapped and liposome entrapped drug in the plasma as our analysis measures total drug content we presume that the majority of the measured carfilzomib is liposome entrapped because the drug is very rapidly eliminated in the blood stream (t.sub.1/2<20 min) (Yang et al 2011, Drug Metab Dispos. 2011 October; 39(10):1873-82). We observed a 100-fold range of drug retention from 0.65% to 66.3% ID depending on the liposome formulation composition. The most stable liposome tested was sphingomyelin based and contained an internal ammonium dextran sulfate solution. The liposomes described above increased the plasma retention of carfilzomib 46-to-4735 fold more than a SBCD formulation, or 5-to-510 fold higher than published liposome formulations at 4 h post administration. (Chu et al 2012 AAPS Meeting, Poster T2082).
Example 16
[0183] Remote Loading of an Insoluble Precipitate of Deferasirox into Liposomes Using an Acetate Gradient
Introduction
[0184] Remote loading of deferasirox (DFX) into liposomes containing calcium acetate demonstrates the use of an acetate gradient for loading an iron chelating agent. Calcium acetate gradient remote loading differs from ammonium sulfate remote loading in that the drug molecule being loaded must have a carboxylate (or hydroxamate) rather than an amine. Deferasirox is known to have significant kidney toxicity and liposome delivery is a technique for reducing kidney toxicity.
Method
[0185] Liposomes were prepared using the extrusion and purification method described in Example 1. The lipid composition was HSPC/Cholesterol (3/0.5, mol/mol) or POPC/cholesterol (3/0.5, mol/mol). The trapping agent consisted of calcium acetate or sodium sulfate each at a concentration of 120 mM. A solution of DFX in DMSO at 20 mg/mL was added to the liposome solution slowly over 30 seconds while vortexing to produce a drug precipitate in the liposome solution. The target drug to phospholipid ratio was 100 g DFX/mol phospholipid. The solution was heated for 30 min (at 45.degree. C. for POPC liposomes and 65.degree. C. for HSPC liposomes) and then cooled on ice. A sample was removed to determine the input drug to lipid ratio and the remaining solution was spun in a centrifuge at 12,000 RPM for 5 minutes to pellet any unloaded drug. The supernatant was further purified from unloaded drug using a Sephadex G25 size exclusion column eluted with 5 mM HEPES, 145 mM NaCl at pH 6.5. The purified liposomes are analyzed for drug and lipid content by HPLC using the system described in Example 1 and a program consisting of gradient elution of 65% B to 98% B in 6 min with 5 min equilibration back to 65% B (A=0.1% TFA, B=0.1% TFA/MeOH, 1.0 mL/min), column temperature held constant at 30.degree. C., 10 ul injection, and detection by absorbance at 254 nm.
Results
[0186] Upon addition of the drug in DMSO to the liposomes containing calcium acetate as the trapping agent, the solution of POPC liposomes were less cloudy than the solution of HSPC liposomes, both contained precipitated drug before loading. After heating, the solutions clarified and appeared like liposomes with no drug precipitate. Liposomes containing sodium sulfate as the control trapping agent never clarified during the heating process and a drug precipitate pellet was formed upon centrifugation. The loading of liposomes containing calcium acetate made from POPC and HSPC was very efficient. Both liposomes containing the calcium acetate trapping resulted in >90% loading efficiency. The liposomes containing sodium sulfate resulted in 3.3% loading efficiency, which indicates that the loading of DFX into calcium acetate liposomes is not passive but can be described as remote loading. The DFX loading results are shown in Table 7 (FIG. 16).
TABLE-US-00007 TABLE 7 Loading Efficiency of DFX in Calcium Acetate Liposomes 2 DFX loading Lipid composition Interior buffer efficiency 3 mol POPC/0.5 mol Chol 120 mM calcium acetate 94.8 .+-. 1.46% 3 mol HSPC/0.5 mol Chol 120 mM calcium acetate 92.5 .+-. 0.33% 3 mol HSPC/0.5 mol Chol 120 mM sodium sulfate 3.3 .+-. 0.14%
Conclusion
[0187] Remote loading an insoluble precipitate of deferasirox into the liposome provides an example of the use of an acetate gradient to remote load a carboxylate drug from a precipitate. In this example the drug loaded was a chelating agent, in particular an iron chelating agent. The 28-fold greater loading into the liposomes having an acetate gradient over control liposomes indicates that the majority of the deferasirox is remote loaded rather than intercalated in the lipid bilayer.
Example 17
[0188] Remote Loading of an Insoluble Precipitate of Deferasirox into Liposomes. Evaluation of Drug to Lipid Ratio and Calcium Acetate Trapping Agent Concentration.
Introduction
[0189] The remote loading capacity of DFX in liposomes containing calcium acetate was evaluated by using different concentrations calcium acetate on the liposome interior and loading a range of different DFX-to-lipid ratios.
Method
[0190] Liposomes were prepared using the extrusion and purification method described in Example 1. The lipid composition was POPC/cholesterol (3/0.5, mol/mol). The trapping agent consisted of calcium acetate 120 mM, 250 mM or 500 mM. A solution of DFX in DMSO at 20 mg/mL was added to the liposome solution slowly over 30 seconds while vortexing to produce a drug precipitate in the liposome solution. The target drug to phospholipid ratio was 100, 200 or 300 g DFX/mol phospholipid. The solution was heated for 30 min at 45.degree. C. and then cooled on ice. A sample was removed to determine the input drug to lipid ratio and the remaining solution was spun in a centrifuge at 12,000 RPM for 5 minutes to pellet any unloaded drug. The supernatant was further purified from unloaded drug using a Sephadex G25 size exclusion column eluted with 5 mM HEPES, 145 mM NaCl at pH 6.5. The purified liposomes are analyzed for drug and lipid content by HPLC as described in Example 16.
Results
[0191] Upon addition of the drug in DMSO to the liposomes containing calcium acetate as the trapping agent the DFX forms a precipitate before loading. After heating, the solutions clarify and look like liposomes with no drug precipitate. The maximum drug load was higher for liposomes containing 250 and 500 mM calcium acetate compared to 120 mM calcium acetate. The maximum drug load and efficiency was achieved at an input of 200 g DFX/mol phospholipid for liposomes containing either 250 mM calcium acetate or 500 mM calcium acetate. The efficiency of loading for a target of 100 g DFX/mol phospholipid ranged from 99.2 to 103% for all three concentrations of internal calcium acetate. When the target drug load was increased to 200 g DFX/mol phospholipid the efficiency of liposomes having 250 or 500 mM internal calcium acetate was at least two-fold greater than liposomes having an internal calcium acetate concentration of 120 mM. The capacity of all three liposomes was exceeded at input of 300 g DFX/mol phospholipid resulting in n efficiency <24%. The results are shown in FIG. 17.
Conclusion
[0192] The drug payload capacity of DFX when remote loaded into liposomes can be substantially increased by increasing the concentration of the trapping agent concentration inside the liposome. This example demonstrates the dependence of loading capacity on calcium acetate trapping agent concentration. This example also demonstrates DFX liposome loading can have an optimum drug to lipid ratio where the efficiency and drug load are both greatest. The achieved drug to lipid ratio allows for the DFX to be administered to an animal using a tolerated dose of lipid.
Example 18
[0193] Remote Loading of an Insoluble Precipitate of Deferasirox into Liposomes. Evaluation of Trapping Agent.
[0194] The remote loading capacity of DFX in liposomes containing calcium acetate, magnesium acetate and zinc acetate was evaluated by preparing liposomes with different trapping agents on the interior and loading a range of different DFX-to-lipid ratios.
Method
[0195] Liposomes were prepared using the extrusion and purification method described in Example 1. The lipid composition was POPC/cholesterol (3/0.5, mol/mol). The trapping agent consisted of calcium acetate, magnesium acetate or zinc acetate at 120 mM. A solution of DFX in DMSO at 20 mg/mL was added to the liposome solution slowly over 30 seconds while vortexing to produce a drug precipitate in the liposome solution. The target drug to phospholipid ratio was 100, 150 or 200 g DFX/mol phospholipid. The solution was heated for 30 min at 45.degree. C. and then cooled on ice. A sample was removed to determine the input drug to lipid ratio and the remaining solution was spun in a centrifuge at 12,000 RPM for 5 minutes to pellet any unloaded drug. The supernatant was further purified from unloaded drug using a Sephadex G25 size exclusion column eluted with 5 mM HEPES, 145 mM NaCl at pH 6.5. The purified liposomes are analyzed for drug and lipid content by HPLC as described in Example 16.
Results
[0196] Upon addition of the drug in DMSO to the liposomes, the DFX forms a precipitate before loading. After heating, the solutions containing liposomes with calcium acetate and magnesium acetate became much less turbid than the liposomes containing zinc acetate as the trapping agent. The maximum drug load was highest for the liposomes containing magnesium the second highest for the liposomes containing calcium acetate and the liposomes containing zinc acetate resulted in the lowest drug payload. The efficiency of loading for a target of 100 g DFX/mol phospholipid was 5.3.+-.0.07% but the efficiency using calcium acetate and magnesium acetate were 97.6.+-.0.41% and 99.2.+-.2.42% respectively. The results are shown in FIG. 18.
Conclusion
[0197] The drug payload capacity of DFX when remote loaded into liposomes can be dependent on the particular metal salt of acetate used for remote loading. This example demonstrates that magnesium acetate is a better trapping agent for DFX than calcium acetate or zinc acetate.
Example 19
[0198] Remote Loading of an Insoluble Precipitate of Amphotericin B into Liposomes Method
[0199] Liposomes were prepared with a lipid composition of HSPC/DSPG/Chol/PEG-DSPE in the ratio 2/0.6/2/0.3 containing 1.0 M (SO.sub.4) TEA dextran sulfate. The liposomes were separated form the non-entrapped TEA dextran sulfate by anion exchange and then by dialysis against 5 mM Hepes buffered 10% (wt/wt) sucrose pH 6.5. The liposomes were exchanged into 0.01 N HCL, 10% sucrose pH 2.0 before drug loading. Amphotericin B was dissolved in DMSO at 10 mg/ml.
[0200] In a typical preparation, the DMSO amphotericin B solution was added dropwise to the liposomes at room temperature while the liposomes suspension was rapidly mixed on a vortex mixer. The concentration of the liposomal lipid was 5 umol (phospholipid)/mL and 0.1 mL of the amphotericin B solution was added per mL of liposomes so that the final amphotericin B concentration was about 1.0 mg/ml AmB and the final DMSO concentration was about 10% (V/V). By adjusting the amount of liposomes in the preparation various AmB/PL ratios were tested, e.g., 200, 400, 800 g/mol.
[0201] The samples were heated at 65.degree. C. for 15 min. Then cooled on ice for 30 minutes. The preparation was then passed through a PD10 (Sephadex G25) gel filtration column to remove any precipitated amphotericin B that was not incorporated into the liposome. The buffer was also exchanged into 5 mM Hepes, 144 mM NaCL pH 6.5. to remove the DMSO from the liposomal amphotericin B preparation. The diameter of the drug loaded liposomes was not changed at the 200 and 400 g drug/mole lipid input ratio and were slighted larger circa 10% in the 800 g drug lipid ratio. The purified liposomes were analyzed for drug and lipid content by the HPLC method as described in Example 1.
Results
[0202] Amphotericin B was remote loaded into liposomes at 96% efficiency at a 200 g drug/mole lipid, at the input ratio of 400 g drug/mole of lipid the drug was about 90% encapsulated to provide a purified preparation of 360 g amphotericin B/mole lipid. At 800 g drug/mole lipid, amphotericin B was about 70% encapsulated, to provide 560 g amphotericin B/mole lipid. All of these values are substantially greater than the approximate value of 120 g amphotericin B/mole lipid that is contained in the drug product Ambisome.RTM.. The final liposome preparation was readily concentrated to a 10 mg/mL amphotericin B concentration in the 5 mM Hepes, 144 mM NaCl, pH 7.4 buffer.
Conclusion
[0203] The drug payload capacity of liposomes for amphotericin B when amphotericin B is remote loaded into pre-formed liposomes in containing TEA dextran sulfate from the precipitated amphotericin B greatly exceeds the amount of amphotericin B that can be loaded into the liposome membrane by the classical mixing of the drug with the lipid components and then forming the liposomes. This example demonstrates that the exceptionally insoluble compound amphotericin B can be remote loaded from the precipitate to provide a high concentration of liposome encapsulated amphotericin B, something that has not been previously possible with amphotericin B lipid formulations.
Example 20
[0204] Remote Loading of an Insoluble Precipitate of a Taxane Derivative, 2' Succinyl Cabazitaxel, into Liposome Method for the Synthesis of Sparingly Soluble Taxane Derivatives with pH Dependent Solubility
[0205] In order to take full advantage of the precipitate loading method, it is advantageous to prepare drugs with a pH adjustable solubility so one can optimize both the encapsulation and release of the encapsulated drug in the correct place in the body using the various remote-loading agents described in this application. To create sparingly soluble taxane derivatives with properties that enable them to be loaded into liposomes, we modified paclitaxel, docetaxel and carbazitaxel with either carboxylic, dimethylamino or morpolino containing moieties at the 2' position (FIG. 19) by standard chemical reactions, well known to those skilled in the art. The parent compounds are illustrated on the right hand side of FIG. 19 with the 2' position indicated by an R group. In the parent compound the R group is a hydrogen. R can be a succinate, glutarate, morpholino or a succinyl dimethylaminopropylamide, glutaryl dimethylamino propylamide, a succinyl-2-(morpholin-4-yl)ethanamide or glutaryl-2-(morpholin-4-yl)ethanamide.
[0206] The reaction of carbazitaxel with either succinic anhydride or glutaric anhydride in pyridine solution at room temperature will give the crystalline mono 2'-adducts succinyl and glutaryl, respectively. The contaminating diesters (about 5% or less) are easily removed by crystallization. This site selectivity is in agreement with published results, which indicates much higher reactivity of the 2'-hydroxyl as compared to the 7-hydroxyl group for acylation reactions. 2'-succinylcarbazitaxel is synthesized by reacting a solution of 0.050 g (0.060 mmol) of carbazitaxel and 0.090 g (0.076 mmol) of succinyl anhydride for three hours at room temperature in 3 mL of pyridine. The reaction mixture is evaporated to dryness in vacuo. The residue is treated with 10 mL of water, stirred for 20 min, and filtered. The precipitate is dissolved in acetone, water is slowly added, and the tiny crystals are collected. The crystals are recrystalled from chloroform/benzene to yield the product, 2'succinylcarbazitaxel.
[0207] The 2' succinyl ester of carbazitaxel is converted into a carbazitaxel with a basic group attached to the carboxylate of the 2' glutarylic acid by coupling with 3-(dimethylamino)-1-propylamine in the presence of CDI with acetonitrile as a solvent. To a well-stirred solution of 4 g (4.1 mmol) of 2'succinylcarbazitaxel in 40 mL of acetonitrile is added 0.88 g (5.43 mmol) of CDI, and the mixture is heated to 45.degree. C. for 5 min. After the mixture is cooled to room temperature, a solution of 0.47 g (4.61 mmol) of 3-(dimethylamino)-1-propylamine in 3 mL of acetonitrile is added over a period of 20 min. After 30 min, the solvent is evaporated, and the residue is treated with 150 mL of water and 40 mL chloroform. The organic layer is washed five times with 150 mL of water, dried with K.sub.2CO.sub.3, and evaporated to yield 3.6 g (83%) an oil. Recrystallization from methylene chloride/ethyl acetate will yield circa 3.6 g (83%) of the title compound. The hydrochloride salt is prepared by the addition of 1 equiv of hydrochloric acid, followed by freeze-drying of the aqueous solution.
[0208] The 2' succinyl ester of carbazitaxel was converted into a morpholin-4-ylethyl amide by reacting the 2' glutaryl ester of carbazitaxel in the presence of CDI with acetonitrile as a solvent with 2-(morpholin-4-yl)ethanamine. To a well-stirred solution of 4 g (4.1 mmol) of 2'-succinylcarbazitaxel in 40 mL of acetonitrile is added 0.88 g (5.43 mmol) of CDI, and the mixture is heated to 45.degree. C. for 5 min. After the mixture is cooled to room temperature, a solution of 0.6 g (4.61 mmol) of 2-(morpholin-4-yl)ethanamine in 3 mL of acetonitrile is added over a period of 20 min. After 30 min, the solvent is evaporated, and the residue is treated with 150 mL of water and 40 mL chloroform. The organic layer is washed five times with 150 mL of water, dried with K.sub.2CO.sub.3, and evaporated to yield circa 3.6 g of an oil of the title compound. The hydrochloride salt was prepared by the addition of 1 equiv of hydrochloric acid. The HCl salt of 2' Morpholine-4-ylethyl amide of succinylcabazitaxel was isolated in good yield by crystallization from a mixture of methylene chloride and ethyl acetate.
[0209] An alternative approach to the preparation of a basic derivative of carbazitaxel was to react (4-morpholinyl)propanoic acid with carbazitaxel via the activation of the carboxylate on the morpholinyl propanoic acid. A stirred solution of 4-morpholinylpropanic acid in pyridine (circa 0.1 g, 0.5 mM, 1.2 equivalents) in 4 mL pyridine containing DBU (1.4 mL, circa 3 equivalents in a 50 mL roundbottom flask was cooled on in a water ice bath and then 2.0 mL of acetonitrile was added. Then carbazitaxel (0.3 grams, 0.35 mmoles) was added. Then 2.5 equivalents (circa 1.8 grams) of 1-ethyl-3-(3'-dimethylaminopropyl)carbodiimide was added in portions over 30 minutes. The suspension was stirred and the ice cold solution was allowed to come to room temperature over the course of 24 hours. Analysis of the reaction mixture on TLC (ethylacetate/hexane/methanol/ammonium hydroxide: 60/30/6/4: V/V) indicated the cabazitaxel is substantially reacted over the course of the 24 hours. Ethanol, circa 4 mL was added to the reaction mixture and the material was concentrated to an oil by removal of the solvent under reduced pressure on a rotary evaporator. The oil was dissolved in circa 8 mL of ethanol and concentrated again. The dried residue was dissolved in about 6 mL of methylchloride and loaded on to a silica flash column that had been pre-equilibrated with ethylacetate/hexane/methanol/ammonium hydroxide: 60/30/2/4: V/V and was eluted with an increasing polar solvent from 2-10% methanol in ethylacetate/hexanes/methanol/ammonium hydroxide (60/30/2-10/4. The fractions containing the desired material were pooled and concentrated. Derivatives of paclitaxel and docetaxel are prepared in a similar manner to the above synthesis to provide sparingly soluble taxane derivatives whose solubility can be adjusted by altering the pH of the liposome solution in which the aprotic solution of the derivatives are added.
[0210] Liposomes were prepared using the extrusion and purification method described in Example 1. The lipid composition was POPC/cholesterol (3/0.5, mol/mol). The trapping agent consisted of zinc acetate or magnesium acetate at 120 mM. A solution of either unmodify carbazitaxel or 2'succinylcarbazitaxel in DMSO at 20 mg/mL was added to the liposome solution slowly over 30 seconds while vortexing to produce a drug precipitate in the liposome solution. The target drug to phospholipid ratio was 100 g/mole lipid for unmodified carbazitaxel and 100, 150 or 200 g for succinylcarbazitaxel/mol phospholipid. The solutions are heated for 30 min at 45.degree. C. and then cooled on ice. A sample was removed to determine the input drug to lipid ratio and the remaining solution was spun in a centrifuge at 12,000 RPM for 5 minutes to pellet any unloaded drug. The supernatant was further purified from unloaded drug using a Sephadex G25 size exclusion column eluted with 5 mM HEPES, 145 mM NaCl at pH 6.5. The purified liposomes were analyzed for drug and lipid content by HPLC as described in Example 16.
Results
[0211] Upon addition of the drug in DMSO to the liposomes containing the divalent acetate buffer, both the carbazitaxel and 2'succinylcarbazitaxel formed a white precipitate before loading. After heating, the white precipitate of the unmodified carbazitaxel solutions containing liposomes was unchanged. However, after heating the 2'-succinylcabazitaxel solution with zinc acetate or magnesium acetate liposomes, the turbidity was substantially less. The maximum 2'succinylcarbazitaxel load was highest for the liposomes containing magnesium acetate and second highest for the liposomes containing zinc acetate. The loading efficacy was greater than 85% in loading the 2'succinylcabazitaxel. The efficiency of loading unmodified carbazitaxel added at 100 g carbazitaxel/mol phospholipid was very low in both the calcium acetate and magnesium acetate liposomes.
[0212] To encapsulate the 2' morpholine-4-ylethyl amide of succinylcabazitaxel, liposomes were prepared with a lipid composition of HSPC/DSPG/Chol/PEG-DSPE in the ratio 2/0.6/2/0.3 containing 1.0 M (SO.sub.4) TEA dextran sulfate. The liposomes were separated form the non-entrapped TEA dextran sulfate by anion exchange and then by dialysis against 5 mM Hepes buffered 10% (wt/wt) sucrose, pH 6.5. 2' morpholine-4-ylethyl amide of succinylcabazitaxel was dissolved in DMSO at 10 mg/mL.
[0213] In a typical preparation, the DMSO 2' morpholine-4-ylethyl amide of succinylcabazitaxel solution was added dropwise to the liposomes at room temperature while the liposomes suspension was rapidly mixed on a vortex mixer. The concentration of the liposomal lipid was 5 umol (phospholipid)/mL and 0.1 mL of the 2' morpholine-4-ylethyl amide of succinylcabazitaxel solution was added per mL of liposomes so that the final 2' morpholine-4-ylethyl amide of succinylcabazitaxel concentration was about 1.0 mg/ml and the final DMSO concentration was about 10% (V/V). By adjusting the amount of liposomes in the preparation various 2' morpholine-4-ylethyl amide of succinylcabazitaxel/PL ratios were tested, e.g., 200, 400, 800 g/mol. To determine if the unmodified cabazitaxel was loaded into the TEA-dextran sulfate liposomes, cabazitaxel was dissolved in DMSO and added to the liposomes as described for the 2' morpholine-4-ylethyl amide of succinylcabazitaxel compound. The solutions were heated for 30 min at 45.degree. C. and then cooled on ice. A sample was removed to determine the input drug to lipid ratio and the remaining solution is spun in a centrifuge at 12,000 RPM for 5 minutes to pellet any unloaded drug. The supernatant was further purified from unloaded drug using a Sephadex G25 size exclusion column eluted with 5 mM HEPES, 145 mM NaCl at pH 6.5. The purified liposomes were analyzed for drug and lipid content by HPLC as described in Example 16. Using a similar method for precipitate loading the 2' morpholine-propyl ester cabazitaxel (FIG. 19) can be precipitate loaded into liposomes containing the TEA-dextran sulfate gradient.
Results
[0214] The unmodified carbazitaxel formed a white turbid precipitate when added to the liposome suspension in a DMSO solution but the precipitate did not clear up upon heating. The encapsulation of cabazitaxel was less then 5% of the added drug. The 2' morpholine-4-ylethyl amide of succinylcabazitaxel and 2' morpholinopropyl derivatives of cabazitaxel (FIG. 19) formed a white turbid precipitate when added to the liposomes that cleared upon heating. The precipitate was loaded into TEA-dextran sulfate liposomes at greater than 90% of the added drug at the 200 and 400 g drug/mole lipid ratios and greater than 70% at the 800 g drug/mole lipid.
Conclusion
[0215] pH titratable taxane derivatives can be remote loaded into liposomes from a precipitate formed when the taxane derivative is added in an aprotic solvent to a preformed liposome containing remote loading agent where the concentration of the mobile ion species is greater on the inside than on the outside of the liposome. Taxanes modified to contain a carboxylate on the 2'OH position can be loaded into liposomes that contain a divalent cation with a mobile anionic salt such as acetate. Taxanes modified at the 2'hydroxyl group to contain a titratable amine can be remote loaded in liposomes containing a mobile cation such as ammonium or triethylamine and an impermeable anion such as sulfate or dextran sulfate. Thus converting an uncharged taxane into a titratable taxane allows the taxane to be loaded into a liposome from a precipitate with high efficiency and retained in the liposome when the liposome is injected into an animal.
[0216] The foregoing descriptions of specific embodiments of the present invention have been presented for purposes of illustration and description. They are not intended to be exhaustive or to limit the invention to the precise forms disclosed, and obviously many modifications and variations are possible in light of the above teaching. The embodiments were chosen and described in order to best explain the principles of the invention and its practical application, to thereby enable others skilled in the art to best utilize the invention and various embodiments with various modifications as are suited to the particular use contemplated. It is intended that the scope of the invention be defined by the claims appended hereto and their equivalents.
[0217] All publications, patents, and patent applications cited herein are hereby incorporated by reference in their entirety for all purposes.
* * * * *
D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

D00013

D00014
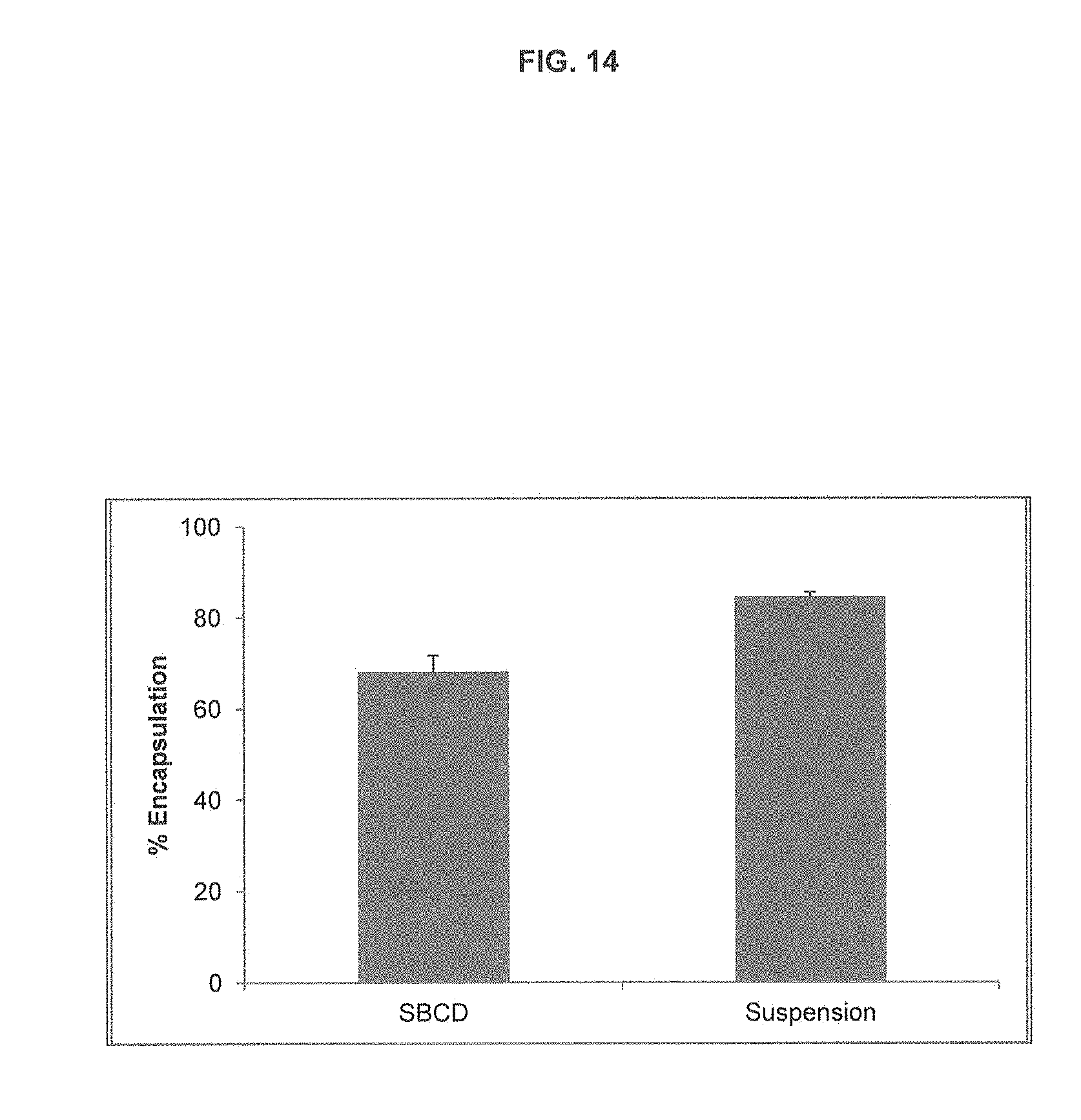
D00015
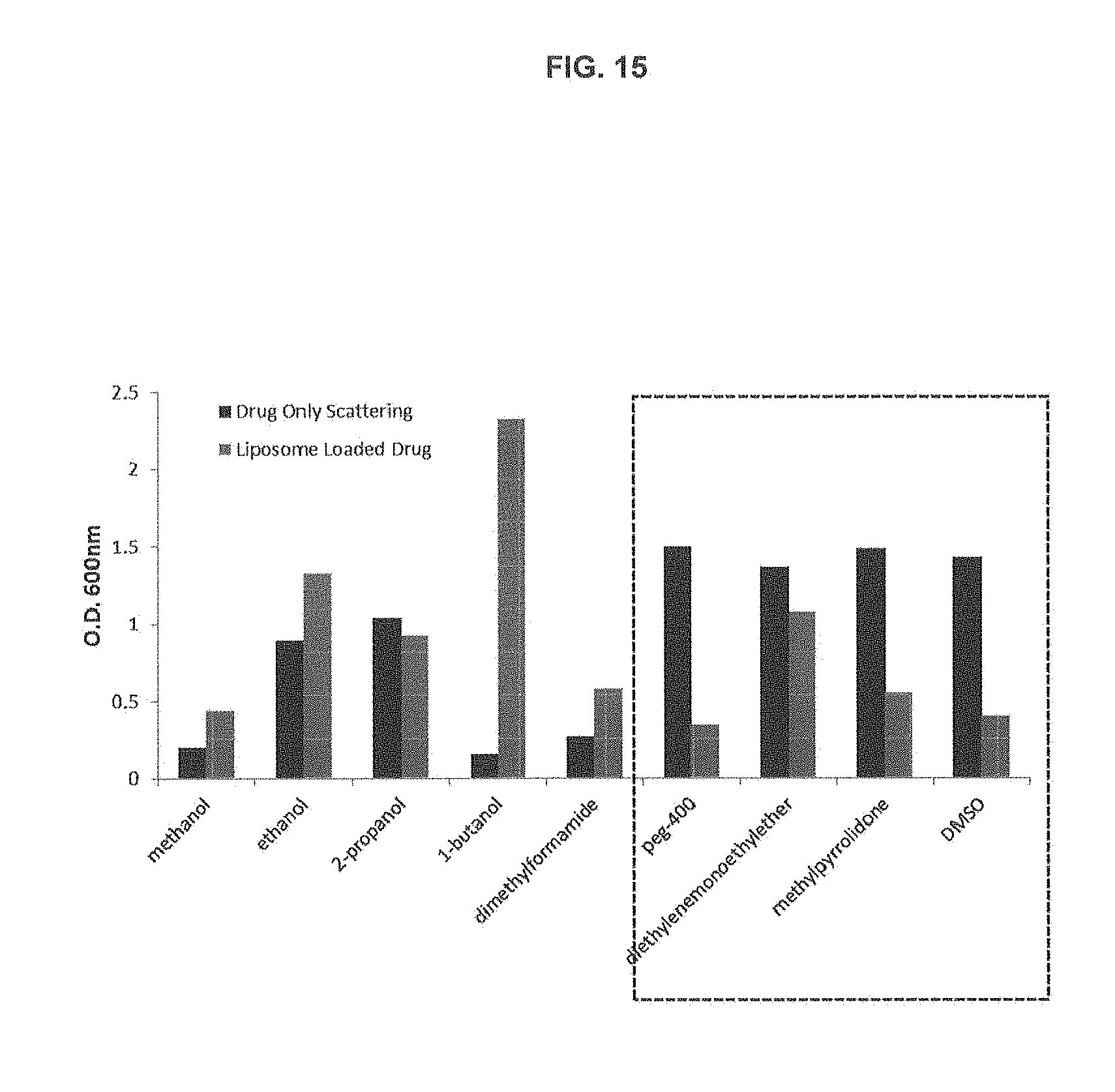
D00016

D00017

D00018

D00019

D00020


XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.