Nucleic Acid Detection Combining Amplification With Fragmentation
Sanders; Heather R. ; et al.
U.S. patent application number 16/165400 was filed with the patent office on 2019-04-04 for nucleic acid detection combining amplification with fragmentation. This patent application is currently assigned to Quest Diagnostics Investments LLC. The applicant listed for this patent is Quest Diagnostics Investments LLC. Invention is credited to Richard A. Bender, Kevin Z. Qu, Heather R. Sanders, Charles M. Strom.
| Application Number | 20190100808 16/165400 |
| Document ID | / |
| Family ID | 40850961 |
| Filed Date | 2019-04-04 |








View All Diagrams
| United States Patent Application | 20190100808 |
| Kind Code | A1 |
| Sanders; Heather R. ; et al. | April 4, 2019 |
NUCLEIC ACID DETECTION COMBINING AMPLIFICATION WITH FRAGMENTATION
Abstract
Provided herein are methods and compositions for detection of a nucleic acid target in a sample. The methods and compositions use primer directed amplification in conjunction with nucleic acid fragmentation. The methods have high sensitivity even in the presence of a large amount of non-target nucleic acid. Also provided are oligonucleotides and kits useful in the method. Exemplary nucleic acid targets are those with mutant gene sequence such as mutant sequence of the EGFR, APC, TMPRSS2, ERG and ETV1 genes.
| Inventors: | Sanders; Heather R.; (RSM, CA) ; Qu; Kevin Z.; (Lake Forest, CA) ; Strom; Charles M.; (San Clemente, CA) ; Bender; Richard A.; (Dana Point, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | Quest Diagnostics Investments
LLC Secaucus NJ |
||||||||||
| Family ID: | 40850961 | ||||||||||
| Appl. No.: | 16/165400 | ||||||||||
| Filed: | October 19, 2018 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15712448 | Sep 22, 2017 | 10106857 | ||
| 16165400 | ||||
| 14679403 | Apr 6, 2015 | 9783854 | ||
| 15712448 | ||||
| 12035356 | Feb 21, 2008 | 8999634 | ||
| 14679403 | ||||
| 61007928 | Jun 8, 2007 | |||
| 60926611 | Apr 27, 2007 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C12Q 1/6886 20130101; C12Q 1/6886 20130101; C12Q 2600/118 20130101; C12Q 2600/156 20130101; C12Q 2521/301 20130101; C12Q 2600/16 20130101; C12Q 1/6806 20130101; C12Q 2531/113 20130101 |
| International Class: | C12Q 1/6886 20060101 C12Q001/6886; C12Q 1/6806 20060101 C12Q001/6806 |
Claims
1-5. (canceled)
6. A kit comprising one or more oligonucleotide primer pairs, wherein at least one oligonucleotide of at least one primer pair comprises a sequence selected from the group consisting of SEQ ID NOs: 1-12, at least one oligonucleotide of the primer pair is labeled with a detectable moiety.
7. The kit of claim 6, wherein the oligonucleotide primer pair comprises a forward primer of SEQ NO:1 and a reverse primer of SEQ NO:2.
8. The kit of claim 6, wherein the oligonucleotide primer pair comprises a forward primer of SEQ NO:3 and a reverse primer of SEQ NO:4.
9. The kit of claim 6, wherein the oligonucleotide primer pair comprises a forward primer of SEQ NO:5 and a reverse primer of SEQ NO:6.
10. The kit of claim 6, wherein the oligonucleotide primer pair comprises a forward primer of SEQ NO:7 and a reverse primer of SEQ NO:8.
11. The kit of claim 6, wherein the oligonucleotide primer pair comprises a forward primer of SEQ NO:9 and a reverse primer of SEQ NO:10.
12. The kit of claim 6, wherein the oligonucleotide primer pair comprises a forward primer of SEQ NO:11 and a reverse primer of SEQ NO:12.
13. The kit of claim 6, wherein the detectable moiety is a fluorescent dye.
14. The kit of claim 6, wherein the detectable moiety is a fluorescent dye.
15. The kit of claim 6, wherein different pairs of primers in the kit are labeled with different distinguishable detectable moieties.
16. The kit of claim 6, wherein the at least one primer pair comprises a forward primer and a reverse primer labeled with different detectable moieties.
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] The present application is a Continuation of U.S. application Ser. No. 15/712,448, filed Sep. 22, 2017, which is a Divisional of U.S. application Ser. No. 14/679,403, filed Apr. 6, 2015, which is a Continuation of U.S. application Ser. No. 12/035,356, filed Feb. 21, 2008, which claims priority to U.S. Provisional Patent Application No. 60/926,611, Titled: Nucleic Acid Detection Combining Amplification With Fragmentation, filed Apr. 27, 2007 and U.S. Provisional Patent Application No. 61/007,928, Titled: Nucleic Acid Detection Combining Amplification With Fragmentation, filed Jun. 8, 2007, which are incorporated herein by reference.
[0002] The instant application contains a Sequence Listing which has been submitted in ASCII format via EFS-WEB and is hereby incorporated by reference in its entirety. Said ASCII copy, created on Sep. 18, 2018, is named sequence.txt and is 17 KB.
FIELD OF THE INVENTION
[0003] Provided herein are methods and compositions for detecting target nucleic acid such as mutant nucleic acid. The methods and compositions combine amplification with nucleic acid fragmentation, are useful for detecting very low amounts of target nucleic acids, even in the presence of large amounts of non-target nucleic acids.
BACKGROUND OF THE INVENTION
[0004] Although nucleic acid assays are known to offer a high degree of specificity, there are limits in the sensitivity of such assays, particularly when the target nucleic acid to be detected is present in relatively low quantities compared to non-target nucleic acid. In the case of cancer, the ability to detect the presence of a small amount of a cancer specific mutant nucleic acid allows for early cancer diagnosis and offers the possibility of more effective therapeutic intervention. However, detection can be challenging if the sample of nucleic acid being tested has very little of the mutant nucleic acid and if there is an excess of normal nucleic acid in the sample. Although a tumor biopsy may contain significant mutant nucleic acids, a plasma sample from a cancer patient may contain one or only a few copies of a mutant nucleic acid of interest. Amplification methods such as PCR may detect a few copies of a mutant nucleic acid, however, the abundance of normal nucleic acid in samples such as plasma can interfere.
[0005] Focus has been placed on identifying tumor-derived mutations in circulating DNA found in plasma or serum of solid tumor patients as a noninvasive and early diagnostic tool. Confirmed reports of the presence of solid tumor-derived mutations found in circulating DNA include, but are not limited to, patients with colorectal tumors, pancreatic cancer, breast cancer, head and neck squamous cell carcinoma, and lung cancer (Hibi et al. 1998, Chen et al. 1999, Diehl et al. 2005, Coulet et al. 2000, Hagiwara 2006, Kimura et al. 2006).
[0006] Reports have also demonstrated that cancer patients show elevated levels of circulating DNA and have proposed use of DNA quantification as prognostic and diagnostic factors (Gautschi et al. 2004, Goebel et al. 2005, Sozzi et al. 2001, Herrera et al. 2005, Pathak et al. 2006). This has led to efforts to describe the origin of such elevated levels of DNA. While still under investigation, the sharp increase in circulating DNA is not likely attributed to DNA released from tumor cells. In fact, analysis of mutations present in the plasma of patients with colorectal tumors revealed that the levels of mutations found in circulating DNA did not increase proportionally with the overall elevated levels of circulating DNA (Diehl et al. 2005). Thus while some cancer patients show elevated levels of plasma DNA, detection of tumor-derived mutations will require the ability to detect very few mutations in the presence of larger amounts of wild type DNA.
[0007] A number of strategies have been described for detecting low copy number nucleic acid targets. Methods including allele-specific PCR of p53 and ABL kinase domain mutations have demonstrated sensitivities ranging from 0.1-0.01% and in one mutation, 0.001% (Righetti et al. 1999, Coulet et al. 2000, and Kang et al 2006). Ohnishi, H., et al. reported a method of amplification using a mutation specific primer that spans a deletion site and does not anneal to the wild-type sequence. Ohnishi, H., et al., 15(2) Diagnostic Molecular Pathology 101-108 (2006). Mutation specific primers of the Scorpion type also have been reported. Kimura, H. et al., 12(13) Clinical Cancer Research 3915-3921 (2006); Newton, C. R., et al., 17(7) Nucleic Acids Research 2503-2516 (1989); and Whitcombe, D. et al., 17 Nature Biotechnology 804-807 (1999) (describing Scorpion ARMS primers and strategies for primer design). Methods that enrich mutant nucleic acid by digesting wild-type DNA with restriction enzymes prior to amplification have been reported. Asano, H., et al., 12(1) Clinical Cancer Research 43-48 (2006); Gocke, C., et al., U.S. Pat. No. 6,630,301. The Asano et al., method uses multiple PCR reactions. A first PCR reaction is used to remove an upstream restriction enzyme recognition site. Following the first PCR, a restriction digestion is performed. After digestion, a second PCR reaction is used to amplify the target sequence.
SUMMARY OF THE INVENTION
[0008] Provided herein are methods and compositions for detecting target nucleic acids at very low levels and in the presence of large amounts of non-target nucleic acids. Generally, a target and non-target nucleic acid are distinguished by the presence or absence of a fragmentation site, such as a restriction enzyme recognition site. By differentiating the target and non-target by a fragmentation site, the methods and compositions used herein can be used with various nucleic acid detection methods known in the art, such as PCR.
[0009] As used herein, the term "target" nucleic acid refers to a nucleic acid which contains an allele or a mutant nucleic acid sequence. A mutant nucleic acid sequence may be any mutant sequence including but not limited to substitution, insertion, deletion, and translocation.
[0010] As used herein, the term "non-target" or "other" nucleic acid used in reference to a target nucleic acid means a nucleic acid that does not contain the target sequence. For example, a non-target nucleic acid of a target nucleic acid encoding an allelic sequence encompasses nucleic acid that contains an alternative allele. The non-target of a nucleic acid containing a mutant sequence is a nucleic acid that contains normal or wild-type nucleic acid sequence with respect to the mutant sequence.
[0011] As used herein, the term "locked nucleic acid" or "LNA" refers to bicyclic nucleic acid analogs contain one or more 2'-O, 4'-C methylene linkage(s), which effectively locks the furanose ring in a C3'-endo conformation. This methylene linkage restricts the flexibility of the ribofuranose ring and locks the structure into a rigid bicyclic formation. Because of its structural conformation, locked nucleic acids demonstrate a much greater affinity and specificity to their complementary nucleic acids than do natural DNA counterparts and increases the thermal and chemical stability of a primer/target nucleic acid duplex. LNAs will hybridize to complementary nucleic acids even under adverse conditions, such as under low salt concentrations and in the presence of chaotropic agents. According to one aspect of the invention, locked nucleic acids increase the melting point of the primer/target nucleic acid duplex by about 3 to about 8.degree. Celsius per locked nucleic acid base incorporated in the primer. The basic structural and functional characteristics of LNAs and related analogues are disclosed in various publications and patents, including WO 99/14226, WO 00/56748, WO 00/66604, WO 98/39352, U.S. Pat. Nos. 6,043,060, and 6,268,490; see also, Braasch et al., "Locked nucleic acid (LNA): fine-tuning the recognition of DNA and RNA," Chem. Biol. 8(1):1
[0012] Locked nucleic acid bases may be interspersed throughout a strand of a primer, placed consecutively or placed singularly in predetermined locations. In one embodiment, the mutation specific primer comprises a locked nucleic acid base at its 3' terminus. In another embodiment, the mutation specific primer comprises a locked nucleic acid base at its N-1 (i.e., penultimate) base. The mutant base may be a locked nucleic acid.
[0013] In one aspect, provided herein is a method for detecting the presence or absence of a target nucleic acid by testing a sample that potentially contains the target nucleic acid in the presence of non-target nucleic acid, the method includes: a) fragmenting the sample nucleic acid under conditions such that a subsequent amplification directed to the target nucleic acid results in an increased detection of the target nucleic acid over the non-target nucleic acid as compared to amplification without fragmentation; b) amplifying the target nucleic acid with a pair of primers, where a first primer is specific for the target nucleic acid; and c) detecting the presence or absence of an amplification product, which indicates the presence or absence of the target nucleic acid in the sample.
[0014] In another aspect, provided herein is a method for diagnosing a cancer or detecting the presence of a tumor cell by determining if an individual has a mutant sequence associated with the cancer or tumor cell type, the method includes: a) obtaining a sample including nucleic acid from the individual; b) fragmenting the sample nucleic acid under conditions such that a subsequent amplification directed to the target nucleic acid results in an increased detection of the target nucleic acid over the non-target nucleic acid as compared to amplification without fragmentation; c) amplifying the target nucleic acid with a pair of primers, where a first primer is specific for the target nucleic acid; and d) detecting the presence or absence of an amplification product containing the mutant sequence, where diagnosis of cancer is determined by the presence absence or amount of amplification product containing the mutant sequence.
[0015] In yet another aspect, provided herein is a method for determining prognosis with cancer by determining if an individual has a mutant sequence associated with the cancer, the method includes: a) obtaining a sample containing nucleic acid from the individual; b) fragmenting the mutant nucleic acid under conditions such that a subsequent amplification directed to the mutant nucleic acid results in an increased detection of the mutant nucleic acid over the non-mutant nucleic acid as compared to amplification without fragmentation; c) amplifying the mutant nucleic acid with a pair of primers, where a first primer is specific for the mutant nucleic acid; and d) detecting the presence, absence and/or amount of an amplification product containing the mutant sequence, where the likelihood of an outcome in the individual is associated with the presence and or amount of mutant nucleic acid sequence.
[0016] In still yet another aspect, provided herein is a method for determining drug sensitivity of an individual diagnosed with cancer, the method includes: a) obtaining a sample comprising nucleic acid from the individual; b) fragmenting the mutant nucleic acid under conditions such that a subsequent amplification directed to the mutant nucleic acid results in an increased detection of the mutant nucleic acid over the non-mutant nucleic acid as compared to amplification without fragmentation; c) amplifying the mutant nucleic acid with a pair of primers, where a first primer is specific for the mutant nucleic acid; d) detecting the presence, absence and/or amount of an amplification product containing the mutant sequence; and e) relating the presence, absence and/or amount of an amplification product containing the mutant sequence to cancer drug sensitivity. Some examples of mutations that affect drug sensitivity which may be targeted by the assay methods described herein are described in Lynch, et al., 350(21) NEJM 2129-2139 (2004); Bell, et al., US Patent App. No. 20060147959 (2005) (determining tyrosine kinase inhibitor, i.e., gefitinib and erlotinib sensitivity by detecting EGFR mutations); and Sawyers, et al., U.S. Patent Appl. No. 2006/0269956 (describing mutations that affect drug resistance to BCR-ABL kinase activity inhibitors typically used to treat CML due to the T315I mutation in the Abl gene).
[0017] In certain embodiments of the aspects provided herein, the mutated nucleic acid sequence is due to a deletion, insertion, substitution and/or translocation or combinations thereof. In preferred embodiments, fragmentation of nucleic acid sequence in which cleavage of wild-type sequence is with a restriction enzyme. Such pre-amplification digestion treatment allows for fragmentation to destroy or substantially decrease the number of wild-type sequences that might be amplified. In yet more preferred embodiments, the fragmentation using a restriction enzyme is combined with the use of a mutation specific primer (or mutated sequence primer).
[0018] In preferred embodiments, a mutated sequence destroys or disrupts a restriction enzyme recognition site present in the corresponding wild-type sequence and that a mutation specific primer can be designed to bind to the mutated version of the sequence and not its wild-type counterpart. For example a mutation specific primer can overlap a border region, which is a region that contains portions of both a wild-type sequence adjacent to a portion of the mutated sequence. In further examples, if a mutation is the result of a deletion, such as the 15 bp deletion in exon 19 of the Epidermal Growth Factor Receptor (EGFR) gene (E746_A750del), a mutation specific primer could be designed, as illustrated in FIG. 1, so as to span a new site in the DNA which arises from the deletion. Other methods of detecting EGFR nucleic acid are described in U.S. Pat. No. 6,759,217 (which describes detecting EGFR nucleic acid in plasma or serum), U.S. Pat. Nos. 6,127,126 and 5,981,725 (both disclose detecting nucleic acid encoding an EGFR mutant protein type II for a mutation in which a portion of the extracellular domain of EGFR is deleted). If a mutation is due to an insertion, a mutation specific primer could be designed to span either or both junctions where the inserted sequence is adjacent to wild-type sequence. If a mutation is due to one or more substitutions, then a mutation specific primer could be designed to span any or all of the substitutions. If a mutation is due to a translocation, then a mutation specific primer could be designed to span one or both junctions of the translocated sequence, in any region where the sequence is altered by the translocation. These examples are merely exemplary and provide guidance to one of skill in the art to design various permutations of primers that would anneal to a mutated sequence and not a wild-type sequence which are appropriate for the methods and compositions provided herein.
[0019] In one approach, a sample is assayed for the presence or absence of a mutated sequence by amplification and detection of the resulting amplification products. In a preferred embodiment, amplification of target nucleic acids is accomplished by polymerase chain reaction (PCR).
[0020] Single or multiple mutant sequences can be assayed. Amplification of multiple mutant sequences can be performed simultaneously in a single reaction vessel, e.g., multiplex PCR. In this case, probes may be distinguishably labeled and/or amplicons may be distinguishable by size differentiation. Alternatively, the assay could be performed in parallel in separate reaction vessels. In such later case, the probes could have the same label.
[0021] In certain embodiments of the aspects provided herein, the methods further comprise a nucleic acid extraction step. Various extraction nucleic acid methods are known in the art which can be employed with the methods and compositions provided herein such as lysis methods (such as alkaline lysis), phenol:chloroform and isopropanol precipitation. Nucleic acid extraction kits can also be used. Preferably, the extraction method is Agencourt Genfind.TM., Roche Cobas.RTM. or phenol:chloroform extraction using Eppendorf Phase Lock Gels.RTM.. More preferably the nucleic acid is extracted using Agencourt Genfind.TM.
[0022] Also provided are exemplary oligonucleotides useful in the methods and kits described herein.
[0023] The following mutated sequences can be detected with the methods and compositions provided herein. The methods outlined for detection of specific deletion mutations, insertion mutations, point mutations and fusion transcripts can be applied to any biomarker which may be used with fragmentation, particularly when a restriction digestion recognition site is disrupted. Embodiments of specific primer designs are described below but the sequence will vary to fit the mutation to be detected. Restriction enzyme digestion sites will also depend on the sequence of the wild-type sequence as compared to the mutated nucleic acid or fusion transcript. The frequency of various restriction sites found in DNA virtually ensures that a site unique to the wild-type DNA of interest can be found for any mutation detection assay, thus this methodology is applicable to a wide array of cancer biomarkers.
[0024] In one approach, a mutation specific primer is designed for detecting a deletion mutation. Mutation specific primer can be designed to span the deleted region such that the primer contains wild-type sequence that lies 5' and 3' of the deleted region or the complement thereof. Thus, the mutation specific primer cannot bind to the wild-type sequence and cannot produce an amplicon.
[0025] In one approach, a mutation specific primer is designed for detecting an insertion mutation. A mutation specific primer can be designed to span all or a portion of the inserted region such that the primer includes all or a part of the inserted region. A primer could be designed to span the either or both junctions of the inserted sequence, for example, the primer sequence would include a portion of wild-type sequence that is adjacent to the inserted sequence or the complement thereof. Thus, the mutation specific primer is not complementary to the wild-type sequence and cannot produce an amplicon.
[0026] In one approach, a mutation specific primer is designed for detecting one or more substitution mutations. A mutation specific primer can be designed to include one or more substitutions or the complement thereof. For example, the 3' nucleotide of the primer can be designed such that it contains the mutated base pair and does not bind hybridize, or base pair, in the wild-type gene and thus cannot elongate.
[0027] In one approach, a mutation specific primer is designed for detecting one or more translocation mutations. A mutation specific primer can be designed to span the junction of the translocation or the complement thereof. A primer pair could be designed to so that one primer is upstream of the translocation junction and the second is downstream of the junction. Thus, when the primer pair is used on wild-type sequence, no amplification products will be produced because the locations of the primers relative to each other are cannot be amplified. However, when the translocation is present, the primers are in close enough proximity of each other such that an amplification product can be produced. For example, the primer can be designed to include a portion of the first gene and a portion of the second gene, where the genes are located on different chromosomes in wild-type form but are adjacent to one another in the mutated form.
[0028] In certain embodiments, at least one primer of each primer pair in the amplification reaction is labeled with a detectable moiety. Thus, following amplification, the various target segments can be identified by size and color. The detectable moiety is preferably a fluorescent dye. In some embodiments, different pairs of primers in a multiplex PCR may be labeled with different distinguishable detectable moieties. Thus, for example, HEX and FAM fluorescent dyes may be present on different primers in multiplex PCR and associated with the resulting amplicons. In other embodiments, the forward primer is be labeled with one detectable moiety, while the reverse primer is labeled with a different detectable moiety, e.g. FAM dye for a forward primer and HEX dye for a reverse primer. Use of different detectable moieties is useful for discriminating between amplified products which are of the same length or are very similar in length. Thus, in certain embodiments, at least two different fluorescent dyes are used to label different primers used in a single amplification. In still another embodiment, control primers can be labeled with one moiety, while the patient (or test sample) primers can be labeled with a different moiety, to allow for mixing of both samples (post PCR) and the simultaneous detection and comparison of signals of normal and test sample. In a modification of this embodiment, the primers used for control samples and patient samples can be switched to allow for further confirmation of results.
[0029] Analysis of amplified products from amplification reactions, such as multiplex PCR, can be performed using an automated DNA analyzer such as an automated DNA sequencer (e.g., ABI PRISM 3100 Genetic Analyzer) which can evaluate the amplified products based on size (determined by electrophoretic mobility) and/or respective fluorescent label.
[0030] The methods and compositions provided herein provide increased sensitivity for detection of a mutated nucleic acid. Preferably the methods can detect mutated nucleic acid that is present in 10% or less, 1% or less, 0.1% or less, 0.01% or less, 0.001% or less, 0.0005% or less, 0.0003% or less, or 0.0002% or less than the total nucleic acid of a sample.
[0031] Various other cancer biomarkers suitable for detection using the methods and compositions provided herein include, but are not limited to, breast cancer markers, such as, GSTP1, RASSF1A (both described in Papadopoulou, E. et al., 1075 Ann. N.Y. Acad. Sci. 235-243 (2006); and RASSF1A (Papadopoulou, E., et al., and Coyle, et al., 16(2) Cancer Epidemiol. Biomarkers Prev. 192-196 (2007)), ATM (Papadopoulou, E., et al.), APC (Coyle, et al.), RARbeta2 (Hogue, M O, et al., 24(26) J. Clin. Oncol. 4262-4269, Epub 2006 Aug. 14 (2006)), and TP53 (Silva, J. M., et al., 8(12) Clin. Cancer Res. 3761-3766 (2002)); ovarian cancer markers, such as p53 (Swisher, E. M., et al., 193(3) American Journal of Obstetrics and Gynecology 662-667 (2005)); hepatocellular carcinoma markers, such as, p53 mutations (Huang, X. H., et al., 9(4) World J Gastroenterol. 692-695 (2003)), and p16 (Le Roux, E., 53(3) Rev. Epidemiol. Sante Publique. 257-266 (2005)); and pancreatic cancer markers, such as K-ras (Castells, A., et al., 17(2) J. Clin. Oncol. 578-584 (1999)).
[0032] Oligonucleotides or combinations of oligonucleotides that are useful as primers or probes in the methods are also provided. These oligonucleotides are provided as substantially purified material.
[0033] Kits comprising oligonucleotides which may be primers for performing amplifications as described herein also are provided. Kits may further include oligonucleotides that may be used as probes to detect amplified nucleic acid. Kits may also include restriction enzymes for digesting non-target nucleic acid to increase detection of target nucleic acid by the oligonucleotide primers.
[0034] As used herein, the term "junction" refers to the position where target and non-target sequences are adjacent to one another due to a sequence change. For example, in the event of a translocation between sequence 1, ATGC and sequence 2, CGTA, the resulting mutated sequence or fusion sequence would be ATGCCGTA. The junction in this translocation example would be "CC." For example, in the even of an insertion of sequence 3, CCCC into sequence 4, ATGC, the resulting mutated sequence would be ATCCCCGC. The junctions in this event would be "TC" and "CG."
[0035] "Fragmentation" as used herein refers a process in which longer lengths of nucleic acid are broken up into shorter lengths of nucleic acid. Nucleic acids may be broken up or fragmented by chemical or biochemical means, preferably nucleic acids are fragmented in a manner that is reproducible, preferably nucleic acids are fragmented by one or more restriction endonucleases. The length of a fragment containing the nucleic acid segment of interest can depend on the length of the nucleic acid segment of interest as well as the restriction enzyme chosen to fragment the DNA.
[0036] A "restriction endonuclease" or "restriction enzyme" as used herein refers to an enzyme that cuts double-stranded DNA at a specific sequence (i.e., the recognition sequence or site). The frequency with which a given restriction endonuclease cuts DNA depends on the length of the recognition site of the enzyme. For example, some enzymes recognize sites that are four nucleotides long (referred to as "four cutters"). In general one can estimate how frequently an enzyme should cut a piece of DNA based the length of the recognition site and the assumption that the probability of any one nucleotide occurring at a given location is 1/4. In the case of a "four cutter" a specific sequence of four nucleotides must be present. Assuming that each nucleotide has an equal chance (i.e., 1/4) of occurring at any particular site within the four nucleotide sequence, then a four-cutter should on average cut once every 256 base pairs (i.e., 1/4.times.1/4.times.1/4.times.1/4= 1/256). A similar calculation can be applied to any restriction enzyme as long as the length of its recognition site is known, making it possible to predict the size and number of a DNA fragments that would be obtained by cutting a DNA molecule of known size. This allows one of skill in the art to produce DNA fragments of known size. Restriction endonucleases are obtained from bacteria or are produced through recombinant technology and are readily available through numerous commercial sources.
[0037] As used herein, the term "increased detection" refers to the ability to detect lower amounts of target nucleic acid in the presence of non-target nucleic acid. For example, as non-target nucleic acid increases, fragmentation of the non-target nucleic acid increases the ability to detect a smaller fraction of target nucleic acid in total nucleic acid.
[0038] As used herein, the term "sample" or "test sample" refers to any liquid or solid (or both) material can be used to test for the presence of nucleic acids. In preferred embodiments, a test sample is obtained from a biological source (i.e., a "biological sample"), such as cells in culture or tissue cells from an animal, preferably, a human. Preferred sample sources include, but are not limited to, sputum (processed or unprocessed), bronchial alveolar lavage (BAL), bronchial wash (BW), blood, bone marrow, bodily fluids, cerebrospinal fluid (CSF), urine, plasma, serum or tissue (e.g., biopsy material). A body fluid sample refers to fluid containing samples from an individual including sputum (processed or unprocessed), bronchial alveolar lavage (BAL), bronchial wash (BW), blood, plasma, serum, and cerebrospinal fluid (CSF). The term "patient sample" as used herein refers to a sample obtained from a human seeking diagnosis and/or treatment of a disease.
[0039] As used herein, the term "oligonucleotide" refers to a short polymer composed of deoxyribonucleotides, ribonucleotides or any combination thereof. Oligonucleotides are generally between about 10, 11, 12, 13, 14 or 15 to about 150 nucleotides (nt) in length, more preferably about 10, 11, 12, 13, 14 or 15 to about 150 nt, more preferably about 10, 11, 12, 13, 14, or 15 to about 70 nt, and most preferably between about 20 to about 26 nt in length. The single letter code for nucleotides is as described in the U.S. Patent Office Manual of Patent Examining Procedure, section 2422, table 1. In this regard, the nucleotide designation "R" means guanine or adenine, "Y" means thymine (uracil if RNA) or cytosine; and "M" means adenine or cytosine. An oligonucleotide may be used as a primer or as a probe.
[0040] As used herein, the term "detecting" used in context of detecting a signal from a detectable label to indicate the presence of a target nucleic acid in the sample does not require the method to provide 100% sensitivity and/or 100% specificity. As is well known, "sensitivity" is the probability that a test is positive, given that the person has a target nucleic acid sequence, while "specificity" is the probability that a test is negative, given that the person does not have the target nucleic acid sequence. A sensitivity of at least 50% is preferred, although sensitivities of at least 60%, at least 70%, at least 80%, at least 90% and at least 99% are clearly more preferred. A specificity of at least 50% is preferred, although sensitivities of at least 60%, at least 70%, at least 80%, at least 90% and at least 99% are clearly more preferred. Detecting also encompasses assays with false positives and false negatives. False negative rates may be 1%, 5%, 10%, 15%, 20% or even higher. False positive rates may be 1%, 5%, 10%, 15%, 20% or even higher.
[0041] As used herein, the term "substantially purified" in reference to oligonucleotides does not require absolute purity. Instead, it represents an indication that the specified oligonucleotide is relatively more pure than it is in the natural environment. Such oligonucleotides may be obtained by a number of methods including, for example, laboratory synthesis, restriction enzyme digestion or PCR. A "substantially purified" oligonucleotide is preferably greater than 50% pure, more preferably at least 75% pure, and most preferably at least 95% pure.
[0042] As used herein, an oligonucleotide is "specific" for a nucleic acid if the oligonucleotide has at least 50% sequence identity with a portion of the nucleic acid when the oligonucleotide and the nucleic acid are aligned. An oligonucleotide that is specific for a nucleic acid is one that, under the appropriate hybridization or washing conditions, is capable of hybridizing to the target of interest and not substantially hybridizing to nucleic acids which are not of interest. Higher levels of sequence identity are preferred and include at least 75%, at least 80%, at least 85%, at least 90%, at least 95% and more preferably at least 98% sequence identity. Sequence identity can be determined using a commercially available computer program with a default setting that employs algorithms well know in the art.
[0043] As used herein, the term "hybridize" or "specifically hybridize" refers to a process where two complementary nucleic acid strands anneal to each other under appropriately stringent conditions. Hybridizations are typically and preferably conducted with oligonucleotides Nucleic acid hybridization techniques are well known in the art. See, e.g., Sambrook, et al., 1989, Molecular Cloning: A Laboratory Manual, Second Edition, Cold Spring Harbor Press, Plainview, N.Y. Those skilled in the art understand how to estimate and adjust the stringency of hybridization conditions such that sequences having at least a desired level of complementarity will stably hybridize, while those having lower complementarity will not. For examples of hybridization conditions and parameters, see, e.g., Sambrook, et al., 1989, Molecular Cloning: A Laboratory Manual, Second Edition, Cold Spring Harbor Press, Plainview, N.Y.; Ausubel, F. M. et al. 1994, Current Protocols in Molecular Biology. John Wiley & Sons, Secaucus, N.J.
[0044] The terms "target nucleic acid" or "target sequence" as used herein refer to a sequence which includes an allele or mutation of interest to be amplified and detected. Copies of the target sequence which are generated during the amplification reaction are referred to as amplification products, amplimers, or amplicons. Target nucleic acid may be composed of segments of a chromosome, a complete gene with or without intergenic sequence, segments or portions of a gene with or without intergenic sequence, or sequence of nucleic acids which probes or primers are designed. Target nucleic acids may include a wild-type sequences, a mutation, deletion or duplication, tandem repeat regions, a gene of interest, a region of a gene of interest or any upstream or downstream region thereof. Target nucleic acids may represent alternative sequences or alleles of a particular gene. Target nucleic acids may be derived from genomic DNA, cDNA, or RNA. As used herein target nucleic acid may be DNA or RNA extracted from a cell or a nucleic acid copied or amplified therefrom.
[0045] "Genomic nucleic acid" or "genomic DNA" refers to some or all of the DNA from a chromosome. Genomic DNA may be intact or fragmented (e.g., digested with restriction endonucleases by methods known in the art). In some embodiments, genomic DNA may include sequence from all or a portion of a single gene or from multiple genes. In contrast, the term "total genomic nucleic acid" is used herein to refer to the full complement of DNA contained in the genome. Methods of purifying DNA and/or RNA from a variety of samples are well-known in the art.
[0046] The term "flanking" as used herein means that a primer hybridizes to a target nucleic acid adjoining a region of interest sought to be amplified on the target. The skilled artisan will understand that preferred primers are pairs of primers that hybridize 3' from a region of interest, one on each strand of a target double stranded DNA molecule, such that nucleotides may be add to the 3' end of the primer by a suitable DNA polymerase.
[0047] The term "complement" "complementary" or "complementarity" as used herein with reference to polynucleotides (i.e., a sequence of nucleotides such as an oligonucleotide or a target nucleic acid) refers to standard Watson/Crick pairing rules. The complement of a nucleic acid sequence such that the 5' end of one sequence is paired with the 3' end of the other, is in "antiparallel association." For example, the sequence "5'-A-G-T-3'" is complementary to the sequence "3'-T-C-A-5'." Certain bases not commonly found in natural nucleic acids may be included in the nucleic acids described herein; these include, for example, inosine, 7-deazaguanine, Locked Nucleic Acids (LNA), and Peptide Nucleic Acids (PNA). Complementary need not be perfect; stable duplexes may contain mismatched base pairs, degenerative, or unmatched bases. Those skilled in the art of nucleic acid technology can determine duplex stability empirically considering a number of variables including, for example, the length of the oligonucleotide, base composition and sequence of the oligonucleotide, ionic strength and incidence of mismatched base pairs. A complement sequence can also be a sequence of RNA complementary to the DNA sequence or its complement sequence, and can also be a cDNA.
[0048] The term "substantially complementary" as used herein means that two sequences hybridize under stringent hybridization conditions. The skilled artisan will understand that substantially complementary sequences need not hybridize along their entire length. In particular, substantially complementary sequences can comprise a contiguous sequence of bases that do not hybridize to a target sequence, positioned 3' or 5' to a contiguous sequence of bases that hybridize under stringent hybridization conditions to a target sequence.
[0049] The term "coding sequence" as used herein means a sequence of a nucleic acid or its complement, or a part thereof, that can be transcribed and/or translated to produce the mRNA for and/or the polypeptide or a fragment thereof. Coding sequences include exons in a genomic DNA or immature primary RNA transcripts, which are joined together by the cell's biochemical machinery to provide a mature mRNA. The anti-sense strand is the complement of such a nucleic acid, and the encoding sequence can be deduced from there.
[0050] The terms "amplification" or "amplify" as used herein includes methods for copying a target nucleic acid, thereby increasing the number of copies of a selected nucleic acid sequence. Amplification may be exponential or linear. A target nucleic acid may be either DNA or RNA. The sequences amplified in this manner form an "amplicon." While the exemplary methods described hereinafter relate to amplification using the polymerase chain reaction (PCR), numerous other methods are known in the art for amplification of nucleic acids (e.g., isothermal methods, rolling circle methods, etc.). The skilled artisan will understand that these other methods may be used either in place of, or together with, PCR methods. See, e.g., Saiki, "Amplification of Genomic DNA" in PCR Protocols, Innis et al., Eds., Academic Press, San Diego, Calif. 1990, pp 13-20; Wharam, et al., Nucleic Acids Res. 2001 Jun. 1; 29(11):E54-E54; Hafner, et al., Biotechniques 2001 April; 30(4):852-6, 858, 860 passim; Zhong, et al., Biotechniques 2001 April; 30(4):852-6, 858, 860.
[0051] The term "multiplex PCR" as used herein refers to simultaneous amplification of two or more products which are each primed using a distinct primer pair.
[0052] As used herein, a "primer" for amplification is an oligonucleotide that specifically anneals to a target nucleotide sequence and leads to addition of nucleotides to the 3' end of the primer in the presence of a DNA or RNA polymerase. The 3' nucleotide of the primer should generally be identical to the target sequence at a corresponding nucleotide position for optimal expression and amplification. The term "primer" as used herein includes all forms of primers that may be synthesized including peptide nucleic acid primers, locked nucleic acid primers, phosphorothioate modified primers, labeled primers, and the like.
[0053] "Sense strand" means the strand of double-stranded DNA (dsDNA) that includes at least a portion of a coding sequence of a functional protein. "Anti-sense strand" means the strand of dsDNA that is the reverse complement of the sense strand.
[0054] As used herein, a "forward primer" is a primer that anneals to the anti-sense strand of dsDNA. A "reverse primer" anneals to the sense-strand of dsDNA.
[0055] As used herein, sequences that have "high sequence identity" have identical nucleotides at least at about 50% of aligned nucleotide positions, preferably at least at about 58% of aligned nucleotide positions, and more preferably at least at about 76% of aligned nucleotide positions.
[0056] As used herein "TaqMan.RTM. PCR detection system" refers to a method for real time PCR. In this method, a TaqMan.RTM. probe which hybridizes to the nucleic acid segment amplified is included in the PCR reaction mix. The TaqMan.RTM. probe comprises a donor and a quencher fluorophore on either end of the probe and in close enough proximity to each other so that the fluorescence of the donor is taken up by the quencher. However, when the probe hybridizes to the amplified segment, the 5'-exonuclease activity of the Taq polymerase cleaves the probe thereby allowing the donor fluorophore to emit fluorescence which can be detected.
[0057] As used herein, "about" means plus or minus 10%.
BRIEF DESCRIPTION OF THE FIGURES
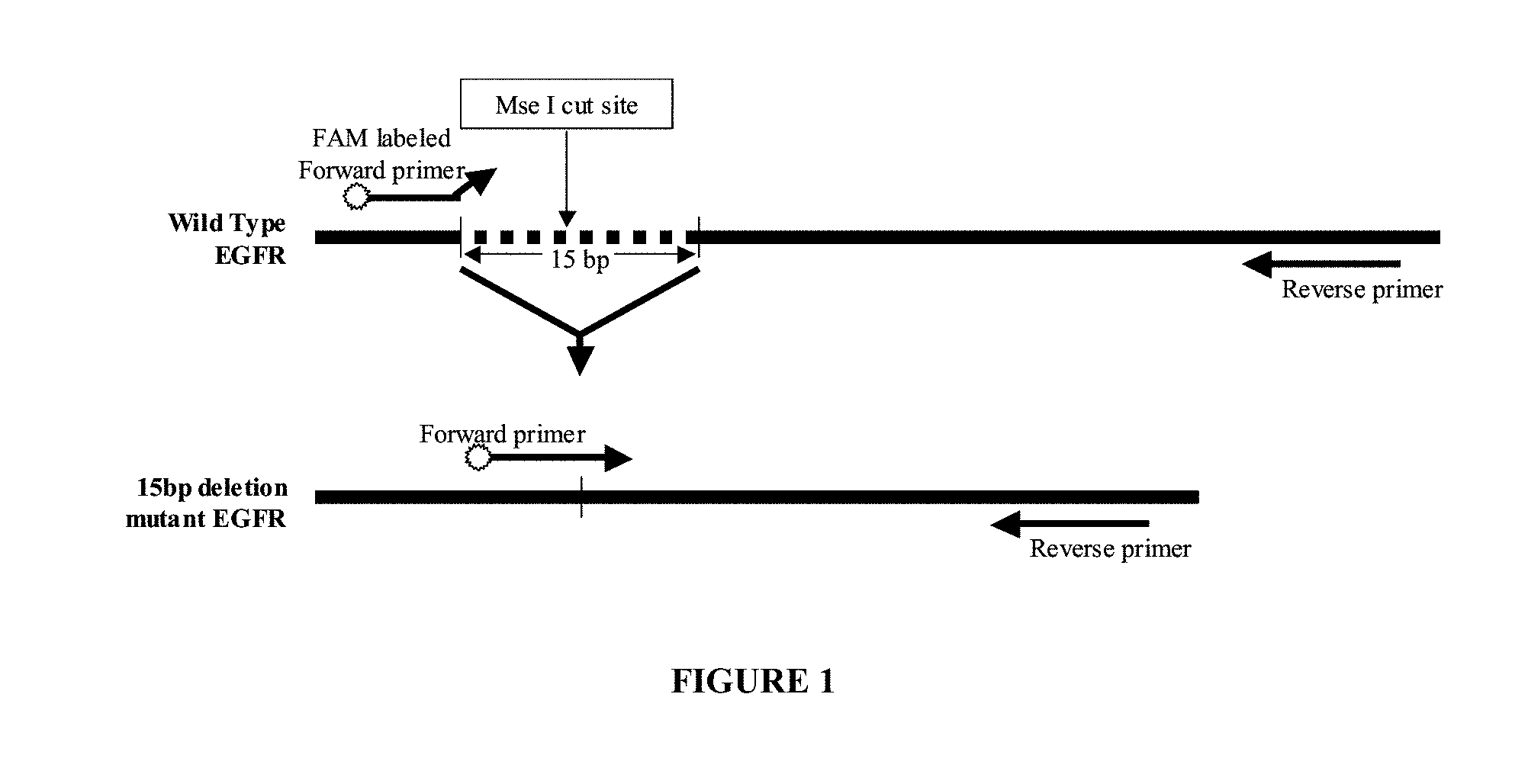
[0058] FIG. 1. Schematic diagram of primer placement for mutant specific PCR of a deletion mutation exemplified by E746_A750del in the EGFR gene. The deleted sequence is shown as a dashed line in the EGFR wild-type DNA. Horizontal arrows indicate primer placement for forward and reverse primers.
[0059] FIG. 2A. Nucleotide sequence of a portion of the sequence coding for the wild-type form of the EGFR (SEQ ID NO:22). The two segments of highlighted unbolded text together represent the sequence for a forward mutation specific PCR primer (SEQ ID NO:1) specific for the E746_A750del mutant EGFR gene. Only a portion of the forward mutation specific primer is complementary to a contiguous segment of the wild-type EGFR gene. Highlighted bolded text indicates sequence for a reverse PCR primer (SEQ ID NO:2). The 15 bp region that is deleted is located between the two portions of the forward primer in the E746_A750del mutant EGFR gene. Boxed TTAA regions indicate MseI restriction sites.
[0060] FIG. 2B. Nucleotide sequence of a portion of the coding region of the E746_A750del mutant of EGFR (SEQ ID NO:23. Highlighted unbolded text indicates sequence of a forward mutation specific PCR primer (SEQ ID NO:1) specific for the E746_A750del mutant EGFR gene. Highlighted bolded text indicates sequence for a reverse PCR primer (SEQ ID NO:2). Boxed TTAA regions indicate MseI restriction sites.
[0061] FIG. 3. Schematic diagram of primer placement for mutant specific PCR of an insertion mutation exemplified by the exon 16 mutation in the APC gene. Horizontal arrows indicate primer placement for forward and reverse primers. The gray region represents the inserted sequence. White stars indicate MnlI restriction sites.
[0062] FIG. 4. Nucleotide sequence of a portion of APC gene showing the exon 16 insertion sequence in unbolded text (SEQ ID NO:24). Highlighted bolded text indicates the sequence for a forward PCR primer (SEQ ID NO:3) and highlighted unbolded text indicates the sequence of a reverse PCR primer (SEQ ID NO:4). Boxed GAGG and CCTC regions indicate MnlI restriction sites.
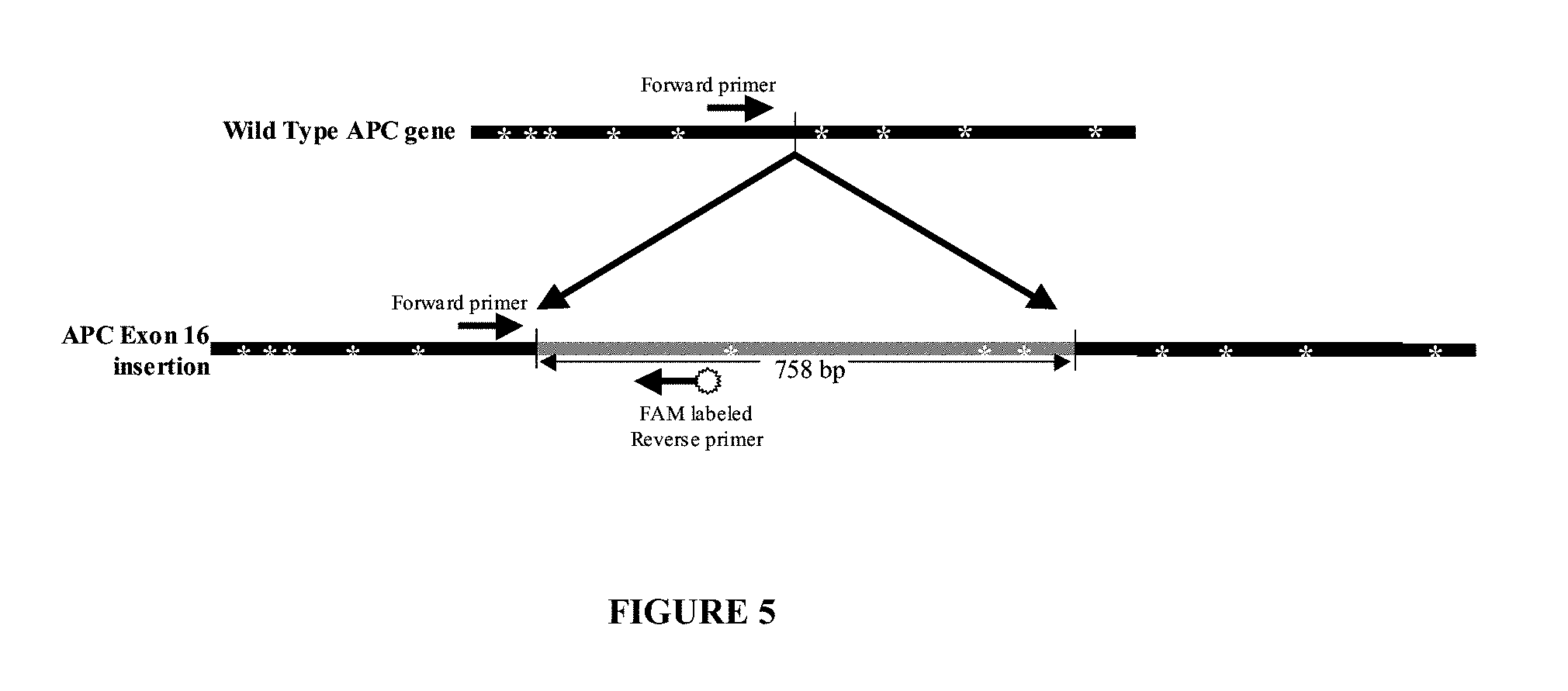
[0063] FIG. 5. Schematic diagram of alternative primer placement for mutant specific PCR of an insertion mutation exemplified by the exon 16 mutation in the APC gene. Horizontal arrows indicate primer placement for forward and reverse primers. The gray region represents the inserted sequence. White stars indicate MnlI restriction sites.
[0064] FIG. 6. Nucleotide sequence of a portion of APC gene showing the exon 16 insertion sequence in unbolded text (SEQ ID NO:25). Highlighted bolded text indicates the sequence for a forward PCR primer (SEQ ID NO:5). Highlighted unbolded text indicates the sequence of a reverse PCR primer (SEQ ID NO:6). Boxed GAGG and CCTC regions indicate MnlI restriction sites.
[0065] FIG. 7. Schematic diagram of primer placement for mutant specific PCR of a substitution mutation exemplified by point mutation L858R in the EGFR gene. Horizontal arrows indicate primer placement for forward and reverse primers. The boxed sequence indicates the EaeI restriction site. The bolded "G" base pair represents the substituted base (SEQ ID NO:26 and SEQ ID NO:27, respectively in order of appearance).
[0066] FIG. 8. Nucleotide sequences of portions of wild-type (SEQ ID NO:28) and mutant EGFR genes (SEQ ID NO:29). Highlighted unbolded text indicates the sequence for a forward mutation specific PCR primer (SEQ ID NO:7) specific for the L858R mutant EGFR gene. Only a portion of the forward mutation specific primer is complementary to a contiguous segment of the wild-type EGFR gene. Highlighted bolded text indicates the sequence for a reverse PCR primer (SEQ ID NO:8). The bolded boxed base pair "T" indicates where the point mutation occurs in the L858R mutant EGFR gene. The bolded "G" in the forward mutation specific primer is the location of the locked nucleic acid. Highlighted unbolded text in the wild-type EGFR gene sequence indicates where the forward mutation specific primer would hybridize, or base pair. Boxed TTAA regions indicate MseI restriction sites. Boxed YGGCCR region, where Y=C or T; and R=A or G, indicates an EaeI restriction site.
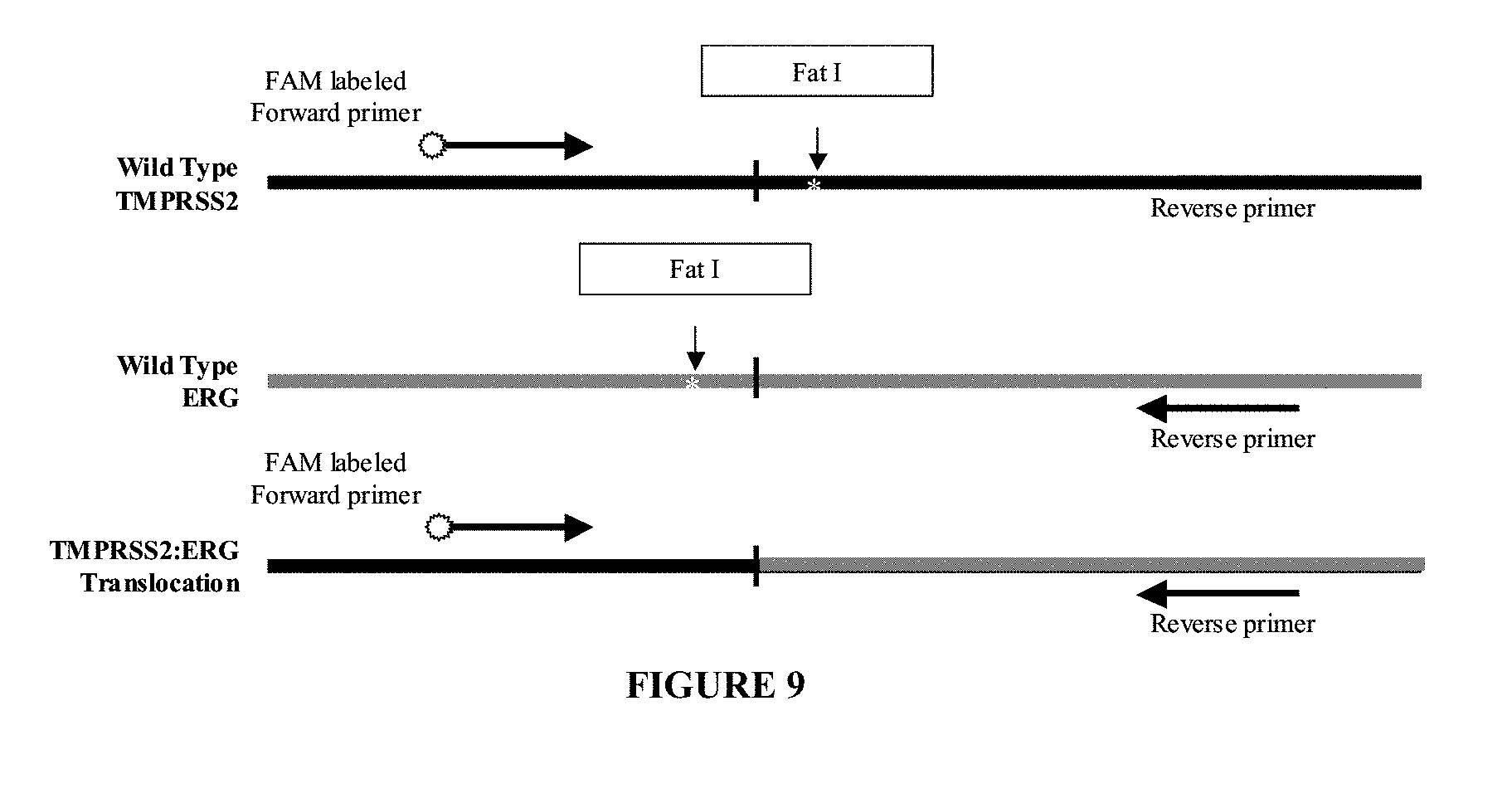
[0067] FIG. 9. Schematic diagram of primer placement for mutant specific PCR of a translocation mutation exemplified by the TMPRSS2:ERG fusion transcript. Horizontal arrows indicate primer placement for forward and reverse primers. White stars indicate FatI restriction sites.
[0068] FIG. 10. Nucleotide sequences of portions of wild-type TMPRSS2 (SEQ ID NO:30) and ERG (SEQ ID NO:31) and mutant fusion (SEQ ID NO:32) gene, TMPRSS2:ERG. Highlighted bolded text indicates the sequence for a forward mutation specific PCR primer (SEQ ID NO:9). Highlighted unbolded text indicates the sequence for a reverse mutation specific PCR primer (SEQ ID NO:10). Underlined regions in the wild-type sequences correspond to the depicted portion of the resulting fusion gene. Boxed CATG regions indicate FatI restriction sites.
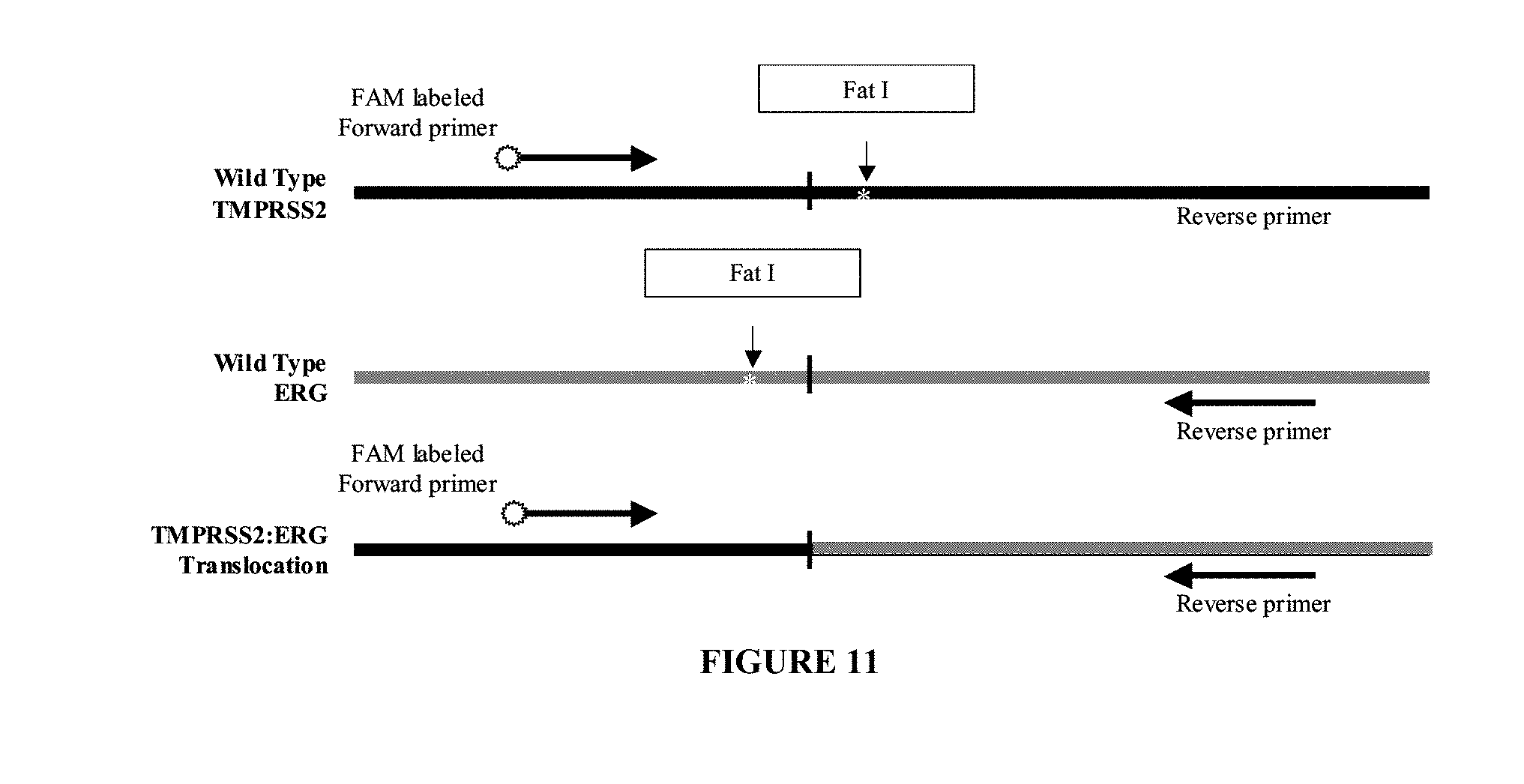
[0069] FIG. 11. Schematic diagram of primer placement for mutant specific PCR of a translocation mutation exemplified by the TMPRSS2:ETV1 fusion transcript. Horizontal arrows indicate primer placement for forward and reverse primers. White stars indicate FatI and HpyCH4V restriction sites.
[0070] FIG. 12. Nucleotide sequences of portions of wild-type TMPRSS2 (SEQ ID NO:33) and ETVI (SEQ ID NO:34) and mutant fusion (SEQ ID NO:35) gene, TMPRSS2:ETV1. Highlighted bolded text indicates sequence for a forward mutation specific PCR primer (SEQ ID NO:11). Highlighted unbolded text indicates sequence for a reverse mutation specific PCR primer (SEQ ID NO:12). Underlined regions in the wild-type sequences correspond to the depicted portion of the resulting fusion gene. Boxed CATG and TGCA regions indicate HpyCH4V restriction sites.
[0071] FIG. 13. Graphical depiction of results of sensitivity comparison assay between Sanders method and Asano method for detection of the E746_A750del mutation in EGFR.
[0072] FIG. 14. Comparison of eight DNA extraction methods. Columns indicate the mean percent recovery of six plasma samples as described in Example 5.
[0073] FIG. 15. Evaluation of detection between Agencourt Genfind.TM., Roche Cobas.RTM. and phenol:chloroform extraction using Eppendorf Phase Lock Gels.RTM. methods using spiked plasma samples as described in Example 5. Columns represent the mean peak intensity of E746_A750del PCR product from the six samples tested for each method obtained from an ABI 3100 Genetic Analyzer.
[0074] FIG. 16. Evaluation of nucleic acid spiking conditions as described in Example 5. Six nucleic acid carrier conditions include 1) no carrier, 2) no carrier plus 100 ng of normal DNA, 3) 1 .mu.g of RNA carrier, 4) 1 .mu.g of RNA carrier plus 100 ng of normal DNA, 5) 395 ng of normal DNA as a carrier, and 6) 778 ng of normal DNA as a carrier. Columns represent the mean peak intensity of E746_A750del PCR product.
DETAILED DESCRIPTION OF THE INVENTION
[0075] In accordance with the present invention, there are provided methods for determining whether a sample contains target nucleic acid. The methods outlined for detection of specific deletion mutations, insertion mutations, point mutations and translocation mutations can be applied to any biomarker in proximity to a restriction digestion recognition site, preferably a restriction digestion recognition site is disrupted by one or more mutations. A blueprint of the primer designs is depicted below but primer sequences will vary to fit the mutation to be detected. Restriction enzyme digestion sites will also depend on the sequence of the non-target sequence as compared to the target nucleic acid or fusion transcript but can follow the formats below. The frequency of various restriction sites found in DNA virtually ensures that a site unique to the non-target DNA of interest can be found for any target detection assay, thus these methodologies are applicable to a wide array of cancer biomarkers.
Primers
[0076] For the methods provided herein, a single primer could be used for detection, for example as in single nucleotide primer extension, or a second primer can be used which can be upstream or downstream of the mutation specific primer. One or more of the primers used may be mutation specific primers. Preferably, the mutation specific primer contains wild-type sequence, more preferably at least about 3-40 consecutive nucleotides of wild-type sequence.
Fragmentation
[0077] Fragmentation is preferably achieved by restriction enzyme treatment or one of other methods of fragmentation well known in the art. In order to reduce the likelihood of mis-priming or inability of the decreased ability for the primer to find a low copy target sequence among non-target sequences, a restriction enzyme recognition site is preferably present in the deleted sequence. Restriction digestion treatment prior to amplification will then cleave non-target sequences. Preferably, the mutation destroys a restriction enzyme recognition site such that the wild-type sequence will be digested, but the mutant sequence no longer contains the recognition site.
[0078] One of skill in the art would recognize that a restriction enzyme fragmentation method can be modified by using a restriction enzyme that cuts at a particular frequency or a particular site, or by using multiple restriction enzymes. The choice of enzyme or enzyme combinations is chosen to suit the target of interest in an assay. Enzymes for fragmentation can be chosen by using a restriction enzyme map of the region of interest. Such maps can be readily generated by software programs well-known to those of skill in the art.
[0079] Chemical fragmentation may include degradation by a nuclease such as DNase or RNase which generate fragments having 3'-OH, 5'-OH, 3'-phosphate and 5'-phosphate ends; depurination or depyrimidation with acid; the use of restriction enzymes; intron-encoded endonucleases; DNA-based cleavage methods, such as triplex and hybrid formation methods, that rely on the specific hybridization of a nucleic acid segment to localize a cleavage agent to a specific location in the nucleic acid molecule; or other enzymes or compounds which cleave DNA at known or unknown locations (see, for example, U.S. Pat. No. 6,495,320). It is possible to depurinate or depyrimidinate the DNA, which is then fragmented in the presence of a base (i.e., ".beta.-elimination") DNA can be fragmented by oxidation, alkylation or free radical addition mechanisms. Metal cations, which are often combined with organic molecules which may function as chemical catalysts, for example imidazole, are used for fragmenting RNA. This fragmentation is preferably carried out in an alkaline medium and generates fragments having 3'-phosphate ends. Chemical catalysts that may be used for nucleic acid fragmentation include MOPS, HEPES, PIPES, and bioorganic polyamines, such as spermine, spermidine and putrescine (Bibille et al., 27 Nucleic Acids Res. 3931-3937 (1999)).
[0080] Different nucleic acid fragmentation techniques have been described, for example, in Trawick et al., 98 Chem Rev. 939-960 (1998), Oivanen at al., 1998, 98 Chem Rev. 961-990 (1998) and Laayoon, et al. U.S. Pat. No. 6,902,891. A method for fragmenting and labeling RNA is described in WO88/04300A1, in which fragmentation is carried out using RNA which possesses enzymatic properties (ribozymes).
[0081] Physical fragmentation methods may involve subjecting the DNA to a high shear rate. High shear rates may be produced, for example, by moving DNA through a chamber or channel with pits or spikes, or forcing the DNA sample through a restricted size flow passage, e.g., an aperture having a cross sectional dimension in the micron or submicron scale. Other physical methods include sonication and nebulization. Combinations of physical and chemical fragmentation methods may likewise be employed such as fragmentation by heat and ion-mediated hydrolysis. See for example, Sambrook et al., "Molecular Cloning: A Laboratory Manual," 3rd Ed. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y. (2001).
[0082] Preferential cleavage can be achieved my other methods known in the art such as the Maxam-Gilbert method. This method involves degrading DNA at a specific base using chemical reagents. A. M. Maxim et al., 65(1) Meth. in Enzym. 499-560 (1980). In general, this method starts with end labeled DNA and cleaves by base specific reagents. For example with guanine bases (the same principle applies to all four bases), DNA of interest is end-labeled (can be 5'- or 3'-end labeling). Then one kind of base is modified, for example with dimethyl sulfate (DMS) to methylate guanines. Conditions can be adjusted to achieve various frequencies of methylation. Following methylation. a reagent such as piperidine is added which causes loss of a methylated base and then breaks the DNA backbone at the site of the lost base (the apurinic site).
Deletion Mutations
[0083] In one approach, a mutation specific primer is designed for detecting a deletion mutation. Mutation specific primer can be designed to span the deleted region such that the primer contains wild-type sequence that lies 5' and 3' of the deleted region or the complement thereof. Thus, the mutation specific primer cannot bind to the wild-type sequence and cannot produce an amplicon.
[0084] Oligonucleotide primers may be designed for amplifying regions of mutated nucleic acid. In one approach, a primer pair is designed for detecting a deletion mutation. In one embodiment, the primer pair is designed to hybridize to a specified segment of the EGFR gene. The sequence of exemplary oligo primers are shown as highlighted regions in FIGS. 2A and 2B (SEQ ID NOs:1 and 2). Exemplary primer pairs for amplifying a region of the EGFR sequence for the E746_A750del mutation use a forward primer (mutation specific primer) with SEQ ID NO:1 (5'-CCCGTCGCTATCAAAACATC-3') and a reverse primer with SEQ ID NO:2 (5'-ATGTGGAGATGAGCAGGGTCT-3'). In this example, the mutation specific primer spans sequence that is deleted in the mutated sequence. Thus, the primer cannot anneal to the wild-type sequence due to the presence of the 15 base pairs. Preferably, the primers with SEQ ID NOs:1 and 2 are each or both used in conjunction with a restriction enzyme digestion treatment with MseI which has a recognition site of TTAA. The mutation specific primer in that example lies 5' and 3' of the 15 bp deleted region, it cannot bind to the wild-type sequence, thus making the primer mutation specific for the deletion mutation. FIGS. 1 and 2 illustrate this example of detecting the E746_A750del mutation in the EGFR gene.
Insertion Mutations
[0085] In one approach, a mutation specific primer is designed for detecting an insertion mutation. A mutation specific primer can be designed to span all or a portion of the inserted region such that the primer includes all or a part of the inserted region. A primer could be designed to span the either or both junctions of the inserted sequence, for example, the primer sequence would include a portion of wild-type sequence that is adjacent to the inserted sequence or the complement thereof. Thus, the mutation specific primer is not complementary to the wild-type sequence and cannot produce an amplicon.
[0086] Preferably, the insertion destroys a restriction enzyme recognition site such that the wild-type sequence will be digested, but the mutant sequence no longer contains the recognition site. Restriction digestion treatment prior to amplification will then cleave non-target sequences. Restriction digestion can also enhance sensitivity by cleaving away sequence surrounding target nucleic acid and facilitate amplification.
[0087] In one embodiment, a primer pair is designed to detect the 758 base pair insertion in exon 16 insertion in the APC gene. The sequence of exemplary oligo primers are shown as highlighted regions in FIG. 4 (SEQ ID NOs:3 and 4). Exemplary primer pairs for amplifying a region of the APC sequence for an exon 16 (Miki, et al., 52(3) Cancer Research 643-645 (1992)) insertion mutation use a forward primer (mutation specific primer) with SEQ ID NO:3 (5'-CTTCCACAATGGTTGAACTAG-3') and a reverse primer (mutation specific primer) with SEQ ID NO:4 (5'-CATCCATGTCCCTACAAAGG-3'). In this example, both forward and reverse primers are mutation specific because they lie within the insertion sequence. Preferably, the primers with SEQ ID NOs:3 and 4 are each or both used in conjunction with a restriction enzyme digestion treatment with MnlI which has a recognition site of CCTC.
[0088] The mutation specific primer in that example lies within the inserted region. Because the primers lie within the insertion sequence, no amplification will occur unless the insertion is present. In addition, there are MnlI restriction sites upstream and downstream of the desired amplification product which will facilitate amplification subsequent to digestion by removing surrounding sequence. FIGS. 3 and 4 illustrate this example of detecting the exon 16 insertion mutation in the APC gene.
[0089] In another embodiment, a primer pair is designed to detect the 758 base pair insertion in exon 16 insertion in the APC gene. The sequence of exemplary oligo primers are shown as highlighted regions in FIG. 6 (SEQ ID NOs:5 and 6). Exemplary primer pairs for amplifying a region of the APC sequence for an exon 16 insertion mutation use a forward primer (mutation specific primer) with SEQ ID NO:5 (5'-GAGCCATTTATACAGAAAGATG-3') and a reverse primer (mutation specific primer) with SEQ ID NO:6 (5'-GAAATACCATTTGACCCAGC-3'). In this example, the forward primer lies outside the insertion sequence and reverse primer lies inside the insertion sequence. Both are mutation specific because an amplicon will not be produced in the absence of the insertion sequence. Preferably, the primers with SEQ ID NOs:5 and 6 are each or both used in conjunction with a restriction enzyme digestion treatment with MnlI which has a recognition site of CCTC.
[0090] One primer is upstream of the insertion site and the second is within the insertion sequence. In addition, there are MnlI restriction sites less than 20 bases upstream and downstream of the desired amplification product which will facilitate amplification of the target nucleic acid by removing surrounding sequence. The wild-type APC sequence is heavily targeted by MnlI restriction enzyme with a number of sites immediately downstream of the forward primer. The insertion sequence in the APC exon 16 insertion mutant contains a region devoid of MnlI restriction sites that is used as the template for PCR. Digestion with MnlI prior to PCR eliminates any linear amplification that may occur by forward primer binding to the wild type APC gene. Fluorescent PCR can be performed using one forward primer that binds to the APC gene just before the insertion and one insertion specific reverse primer (unlabeled forward, FAM labeled reverse) designed to specifically recognize the insertion sequence in the region not containing MnlI restriction sites. FIGS. 5 and 6 illustrate this example of detecting the exon 16 insertion mutation in the APC gene.
Substitution Mutations
[0091] In one approach, a mutation specific primer is designed for detecting one or more substitution mutations. A mutation specific primer can be designed to include one or more substitutions. In a preferred embodiment, the 3' nucleotide of the primer can be designed such that it contains the mutated base pair and does not bind, hybridize, or base pair, in the wild-type gene and thus cannot elongate. In another preferred embodiment, the mutated base pair is located at the -1 position at the 3'-end of a mutation specific primer (i.e., the penultimate base).
[0092] Preferably, the one or more substitutions destroys a restriction enzyme recognition site such that the wild-type sequence will be digested, but the mutant sequence no longer contains the recognition site. Restriction digestion treatment prior to amplification will then cleave non-target sequences.
[0093] In further preferred embodiments, the mutated base pair in the mutation specific primer is a locked nucleic acid (LNA). The locked nucleic acid provides increased specificity by increasing the melting temperature of the of a primer containing the substitution base. This allows for the use of an increased annealing temperature during amplification which decreases amplification of wild type sequences.
[0094] In one embodiment, a primer pair is designed to detect the L858R mutation in the EGFR gene. The sequence of exemplary oligo primers are shown as highlighted regions in FIG. 8 (SEQ ID NOs:7 and 8). Exemplary primer pairs for amplifying a region of the EGFR sequence for the L858R mutation use a forward primer (mutation specific primer) with SEQ ID NO:7 (5'-TCACAGATTTTGGGCGG-3') and a reverse primer with SEQ ID NO:8 (5'-CCTGGTGTCAGGAAAATGCT-3'). In this example, the mutation specific primer contains the mutated sequence at the terminal base. Thus, it will not properly anneal to the wild-type sequence because the last base is not complementary. Preferably, the primers with SEQ ID NOs:7 and 8 are each or both used in conjunction with a restriction enzyme digestion treatment with EaeI which has a recognition site of YGGCCR, where Y=C or T and R=A or G. As shown in FIG. 8, the boxed MseI restriction sites, TTAA, illustrate that a simultaneous reaction, such as a multiplex PCR reaction, can be used to detect either or both the E746_A750del and L858R mutations in the same reaction. Digestion with both MseI and EaeI does not disrupt the L858R sequence of interest.
[0095] The mutation specific primer in that example includes the mutated base pair sequence, a G, at its 3' end. Because the primer is not complementary to the wild-type sequence, which contains a T, elongation will not occur. The EaeI cut site allows cleavage of the wild-type EGFR gene but is destroyed by the T-*G conversion. Thus, when the L858R EGFR mutant is present, the recognition site is no longer present and can no longer be digested by EaeI. FIGS. 7 and 8 illustrate this example of detecting the L858R mutation in the EGFR gene.
Translocation Mutations
[0096] In one approach, a mutation specific primer is designed for detecting one or more translocation mutations. A mutation specific primer can be designed to span the junction of the translocation or the complement thereof. A primer pair could be designed to so that one primer is upstream of the translocation junction and the second is downstream of the junction. Thus, when the primer pair is used on wild-type sequence, no amplification products will be produced because the locations of the primers relative to each other are cannot be amplified. However, when the translocation is present, the primers are in close enough proximity of each other such that an amplification product can be produced. For example, the primer can be designed to include a portion of the first gene and a portion of the second gene, where the genes are located on different chromosomes in wild-type form but are adjacent to one another in the mutated form.
[0097] Preferably, one or more translocations destroys a restriction enzyme recognition site such that the wild-type sequence will be digested, but the mutant sequence no longer contains the recognition site. Restriction digestion treatment prior to amplification will then cleave non-target sequences.
[0098] In one embodiment, a primer pair is designed to detect the TMPRSS2:ERG or translocation mutation of the TMPRSS2 and ERG genes. The sequence of exemplary oligo primers are shown as higlighted regions in FIG. 10 (SEQ ID NOs:9 and 10). Exemplary primer pairs for amplifying a region of the TMPRSS2 and ERG sequences for the TMPRSS2:ERG translocation mutation use a forward primer (mutation specific primer) with SEQ ID NO:9 (5'-CGAGCTAAGCAGGAGGCGG-3') and a reverse primer (mutation specific primer) with SEQ ID NO:10 (5'-GTCCATAGTCGCTGGAGGAG-3'). In this example, while both primers anneal to wild-type sequences, they are mutation specific when used in conjunction with each other because they will not produce an amplification product unless the translocation is present in the nucleic acid sample. Preferably, the primers with SEQ ID NOs:9 and 10 are each or both used in conjunction with a restriction enzyme digestion treatment with FatI which has a recognition site of CATG.
[0099] In another embodiment, a primer pair is designed to detect the TMPRSS2:ETV1 translocation mutations of the TMPRSS2 and ETV1 genes. The sequence of exemplary oligo primers are shown as highlighted regions in FIG. 12 (SEQ ID NOs:11 and 12). Exemplary primer pairs for amplifying a region of the TMPRSS2 and ERG sequences for the TMPRSS2:ETV1 translocation mutation use a forward primer (mutation specific primer) with SEQ ID NO:11 (5'-CGAGCTAAGCAGGAGGCGG-3') and a reverse primer (mutation specific primer) with SEQ ID NO:12 (5'-ACTTTCAGCCTGATAGTCTGG-3'). In this example, while both primers anneal to wild-type sequences, they are mutation specific when used in conjunction with each other because they will not produce an amplification product unless the translocation is present in the nucleic acid sample. Preferably, the primers with SEQ ID NOs:11 and 12 are each or both used in conjunction with a restriction enzyme digestion treatment with HpyCH4VI which has a recognition site of TGCA.
[0100] In these embodiments, the Fat I and HpyCH4V cut sites allow cleavage of the wild-type TMPRSS2, ERG, and ETV1 in the regions that are absent in the fusion transcripts, essentially "decontaminating" the sample of wild-type TMPRSS2 and ERG or ETV1 translocations. Because the forward and reverse primer sequences are only both present in the fusion transcripts, only the nucleic acids representing a fusion transcript will yield PCR products. FIGS. 9 and 10 illustrate this example of detecting the TMPRSS2:ERG translocation mutation.
Sample Preparation
[0101] The method may be performed using any sample containing nucleic acid. Samples may be obtained by standard procedures and may be used immediately or stored (e.g., the sample may be frozen between about -15.degree. C. to about -100.degree. C.) for later use. Samples may be obtained from patients suspected of having a mutated nucleic acid sequence, for example from a tumor cell or cancer cells. The presence of mutated nucleic acids in a sample can be determined by amplifying cancer marker regions. Thus, any liquid or solid material believed to contain cancer marker nucleic acids can be an appropriate sample. Preferred sample tissues include plasma, blood, bone marrow, body fluids, cerebrospinal fluid, urine and others. Heparin is known to inhibit PCR (Beutler, et al. BioTechniques 9:166, 1990), so samples containing heparin are not ideal for the uses contemplated herein. Nucleic acid extraction techniques that remove heparin are known in the art. These techniques may be used to remove heparin from samples to make the samples more suitable for amplification.
[0102] The sample may be processed to release or otherwise make available a nucleic acid for detection as described herein. Such processing may include steps of nucleic acid manipulation, e.g., preparing a cDNA by reverse transcription of RNA from the biological sample. Thus, the nucleic acid to be amplified by the methods of the invention may be genomic DNA, cDNA, single stranded DNA or mRNA.
Oligonucleotides
[0103] Oligonucleotide primers may be approximately 15-100 nucleotides in length. Of the specific oligonucleotides provided herein, additional variations of the primers comprise all or a portion of the SEQ IDs described herein. Other preferred oligonucleotide primers include an oligonucleotide sequence that hybridizes to the complement of a 15-100 nucleotide sequence that comprises the complement of all or a portion of the SEQ IDs described herein. Such oligonucleotides may be substantially purified.
Amplification of Nucleic Acids
[0104] Nucleic acid samples or isolated nucleic acids may be amplified by various methods known to the skilled artisan. Preferably, PCR is used to amplify mutated nucleic acids of interest. In this method, two or more oligonucleotide primers that flank or include, and anneal to opposite strands of a nucleic acid of interest are repetitively annealed to their complementary sequences, extended by a DNA polymerase (e.g., AmpliTaq Gold polymerase), and heat denatured, resulting in exponential amplification of the target nucleic acid sequences. Cycling parameters can be varied, depending on the length of nucleic acids to be extended. The skilled artisan is capable of designing and preparing primers that are appropriate for amplifying a target sequence in view of this disclosure. The length of the amplification primers for use in the present invention depends on several factors including the nucleotide sequence identity and the temperature at which these nucleic acids are hybridized or used during in vitro nucleic acid amplification. The considerations necessary to determine a preferred length for an amplification primer of a particular sequence identity are well known to the person of ordinary skill. For example, the length of a short nucleic acid or oligonucleotide can relate to its hybridization specificity or selectivity.
[0105] Assay controls may be used in the assay for detecting a mutated nucleic acid sequence. An internal positive amplification control (IPC) can be included in the sample, utilizing oligonucleotide primers and/or probes.
Detection of Amplified Nucleic Acids
[0106] Amplification of nucleic acids can be detected by any of a number of methods well-known in the art such as gel electrophoresis, column chromatography, hybridization with a probe, or sequencing.
[0107] In one approach, sequences from two or more regions of interest are amplified in the same reaction vessel. In this case, the amplicon(s) could be detected by first size-separating the amplicons then detection of the size-separated amplicons. The separation of amplicons of different sizes can be accomplished by, for example, gel electrophoresis, column chromatography, or capillary electrophoresis. These and other separation methods are well-known in the art. In one example, amplicons of about 10 to about 150 base pairs whose sizes differ by 10 or more base pairs can be separated, for example, on a 4% to 5% agarose gel, (a 2% to 3% agarose gel for about 150 to about 300 base pair amplicons) or a 6% to 10% polyacrylamide gel. The separated nucleic acids can then be stained with a dye such as ethidium bromide and the size of the resulting stained band or bands can be compared to a standard DNA ladder.
[0108] In another embodiment, two or more regions of interest are amplified in separate reaction vessels. If the amplification is specific, that is, one primer pair amplifies for one region of interest but not the other, detection of amplification is sufficient to distinguish between the two types--size separation would not be required.
[0109] In some embodiments, amplified nucleic acids are detected by hybridization with a mutation-specific probe. Probe oligonucleotides, complementary to a portion of the amplified target sequence may be used to detect amplified fragments. Amplified nucleic acids for each of the target sequences may be detected simultaneously (i.e., in the same reaction vessel) or individually (i.e., in separate reaction vessels). In preferred embodiments, the amplified DNA is detected simultaneously, using two distinguishably-labeled, gene-specific oligonucleotide probes, one which hybridizes to the first target sequence and one which hybridizes to the second target sequence.
[0110] The probe may be detectably labeled by methods known in the art. Useful labels include, e.g., fluorescent dyes (e.g., Cy5.RTM., Cy3.RTM., FITC, rhodamine, lanthamide phosphors, Texas red), 32P, 35S, 3H, 14C, 125I, 131I, electron-dense reagents (e.g., gold), enzymes, e.g., as commonly used in an ELISA (e.g., horseradish peroxidase, beta-galactosidase, luciferase, alkaline phosphatase), colorimetric labels (e.g., colloidal gold), magnetic labels (e.g., Dynabeads.TM.), biotin, dioxigenin, or haptens and proteins for which antisera or monoclonal antibodies are available. Other labels include ligands or oligonucleotides capable of forming a complex with the corresponding receptor or oligonucleotide complement, respectively. The label can be directly incorporated into the nucleic acid to be detected, or it can be attached to a probe (e.g., an oligonucleotide) or antibody that hybridizes or binds to the nucleic acid to be detected.
[0111] A probe oligonucleotide, complementary to the amplified region of nucleic acid, is used to detect the amplification of mutated nucleic acids. The probe may be detectably labeled by methods known in the art. The binding of a probe to the amplified region of the mutated nucleic acid may be determined by hybridization as is well known in the art. Hybridization may be detected in real time or in non-real time.
[0112] One general method for real time PCR uses fluorescent probes such as the TaqMan.RTM. probes, molecular beacons and scorpions. Real-time reverse-transcriptase (RT) PCR quantitates the initial amount of the template with more specificity, sensitivity and reproducibility, than other forms of quantitative reverse transcriptase PCR, which detect the amount of final amplified product. Real-time RT-PCR does not detect the size of the amplicon. The probes employed in TaqMan.RTM. and molecular beacon technologies are based on the principle of fluorescence quenching and involve a donor fluorophore and a quenching moiety.
[0113] In a preferred embodiment, the detectable label is a fluorophore. The term "fluorophore" as used herein refers to a molecule that absorbs light at a particular wavelength (excitation frequency) and subsequently emits light of a longer wavelength (emission frequency). The term "donor fluorophore" as used herein means a fluorophore that, when in close proximity to a quencher moiety, donates or transfers emission energy to the quencher. As a result of donating energy to the quencher moiety, the donor fluorophore will itself emit less light at a particular emission frequency that it would have in the absence of a closely positioned quencher moiety.
[0114] The term "quencher moiety" as used herein means a molecule that, in close proximity to a donor fluorophore, takes up emission energy generated by the donor and either dissipates the energy as heat or emits light of a longer wavelength than the emission wavelength of the donor. In the latter case, the quencher is considered to be an acceptor fluorophore. The quenching moiety can act via proximal (i.e., collisional) quenching or by Forster or fluorescence resonance energy transfer ("FRET"). Quenching by FRET is generally used in TaqMan.RTM. probes while proximal quenching is used in molecular beacon and scorpion type probes.
[0115] In proximal quenching (a.k.a. "contact" or "collisional" quenching), the donor is in close proximity to the quencher moiety such that energy of the donor is transferred to the quencher, which dissipates the energy as heat as opposed to a fluorescence emission. In FRET quenching, the donor fluorophore transfers its energy to a quencher which releases the energy as fluorescence at a higher wavelength. Proximal quenching requires very close positioning of the donor and quencher moiety, while FRET quenching, also distance related, occurs over a greater distance (generally 1-10 nm, the energy transfer depending on R-6, where R is the distance between the donor and the acceptor). Thus, when FRET quenching is involved, the quenching moiety is an acceptor fluorophore that has an excitation frequency spectrum that overlaps with the donor emission frequency spectrum. When quenching by FRET is employed, the assay may detect an increase in donor fluorophore fluorescence resulting from increased distance between the donor and the quencher (acceptor fluorophore) or a decrease in acceptor fluorophore emission resulting from increased distance between the donor and the quencher (acceptor fluorophore).
[0116] Suitable fluorescent moieties include the following fluorophores known in the art: [0117] 4-acetamido-4'-isothiocyanatostilbene-2,2'disulfonic acid [0118] acridine and derivatives: [0119] acridine [0120] acridine isothiocyanate [0121] Alexa Fluor.RTM. 350, Alexa Fluor.RTM. 488, Alexa Fluor.RTM. 546, Alexa Fluor.RTM. 555, Alexa Fluor.RTM. 568, Alexa Fluor.RTM. 594, Alexa Fluor.RTM. 647 (Molecular Probes) [0122] 5-(2'-aminoethy)aminonaphthalene-1-sulfonic acid (EDANS) [0123] 4-amino-N-[3-vinylsulfonyl)phenyl]naphthalimide-3,5 disulfonate (Lucifer Yellow VS) [0124] N-(4-anilino-1-naphthyl)maleimide [0125] anthranilamide [0126] Black Hole Quencher.TM. (BHQ.TM.) dyes (biosearch Technologies) [0127] BODIPY.RTM. R-6G, BOPIPY.RTM. 530/550, BODIPY.RTM. FL [0128] Brilliant Yellow [0129] coumarin and derivatives: [0130] coumarin [0131] 7-amino-4-methylcoumarin (AMC, Coumarin 120) [0132] 7-amino-4-trifluoromethylcouluarin (Coumarin 151) [0133] Cy2.RTM., Cy3.RTM., Cy3.5.RTM., Cy5.RTM., Cy5.5.RTM. [0134] cyanosine [0135] 4',6-diaminidino-2-phenylindole (DAPI) [0136] 5', 5''-dibromopyrogallol-sulfonephthalein (Bromopyrogallol Red) [0137] 7-diethylamino-3-(4'-isothiocyanatophenyl)-4-methylcoumarin [0138] diethylenetriamine pentaacetate [0139] 4,4'-diisothiocyanatodihydro-stilbene-2,2'-disulfonic acid [0140] 4,4'-diisothiocyanatostilbene-2,2'-disulfonic acid [0141] 5-[dimethylamino]naphthalene-1-sulfonyl chloride (DNS, dansyl chloride) [0142] 4-(4'-dimethylaminophenylazo)benzoic acid (DABCYL) [0143] 4-dimethylaminophenylazophenyl-4'-isothiocyanate (DABITC) [0144] Eclipse.TM. (Epoch Biosciences Inc.) [0145] eosin and derivatives: [0146] eosin [0147] eosin isothiocyanate [0148] erythrosin and derivatives: [0149] erythrosin B [0150] erythrosin isothiocyanate [0151] ethidium [0152] fluorescein and derivatives: [0153] 5-carboxyfluorescein (FAM) [0154] 5-(4,6-dichlorotriazin-2-yl)aminofluorescein (DTAF) [0155] 2',7'-dimethoxy-4'5'-dichloro-6-carboxyfluorescein (JOE) [0156] fluorescein [0157] fluorescein isothiocyanate (FITC) [0158] hexachloro-6-carboxyfluorescein (HEX) [0159] QFITC (XRITC) [0160] tetrachlorofluorescein (TET) [0161] fluorescamine [0162] IR144 [0163] IR1446 [0164] Malachite Green isothiocyanate [0165] 4-methylumbelliferone [0166] ortho cresolphthalein [0167] nitrotyrosine [0168] pararosaniline [0169] Phenol Red [0170] B-phycoerythrin, R-phycoerythrin [0171] o-phthaldialdehyde [0172] Oregon Green.RTM. [0173] propidium iodide [0174] pyrene and derivatives: [0175] pyrene [0176] pyrene butyrate [0177] succinimidyl 1-pyrene butyrate [0178] QSY.RTM. 7, QSY.RTM. 9, QSY.RTM. 21, QSY.RTM. 35 (Molecular Probes) [0179] Reactive Red 4 (Cibacron.RTM. Brilliant Red 3B-A) [0180] rhodamine and derivatives: [0181] 6-carboxy-X-rhodamine (ROX) [0182] 6-carboxyrhodamine (R6G) [0183] lissamine rhodamine B sulfonyl chloride [0184] rhodamine (Rhod) [0185] rhodamine B [0186] rhodamine 123 [0187] rhodamine green [0188] rhodamine X isothiocyanate [0189] sulforhodamine B [0190] sulforhodamine 101 [0191] sulfonyl chloride derivative of sulforhodamine 101 (Texas Red) [0192] N,N,N',N'-tetramethyl-6-carboxyrhodamine (TAMRA) [0193] tetramethyl rhodamine [0194] tetramethyl rhodamine isothiocyanate (TRITC) [0195] riboflavin [0196] rosolic acid [0197] terbium chelate derivatives
[0198] Other fluorescent nucleotide analogs can be used, see, e.g., Jameson, 278 Meth. Enzymol. 363-390 (1997); Zhu, 22 Nucl. Acids Res. 3418-3422 (1994). U.S. Pat. Nos. 5,652,099 and 6,268,132 also describe nucleoside analogs for incorporation into nucleic acids, e.g., DNA and/or RNA, or oligonucleotides, via either enzymatic or chemical synthesis to produce fluorescent oligonucleotides. U.S. Pat. No. 5,135,717 describes phthalocyanine and tetrabenztriazaporphyrin reagents for use as fluorescent labels.
[0199] The detectable label can be incorporated into, associated with or conjugated to a nucleic acid. Label can be attached by spacer arms of various lengths to reduce potential steric hindrance or impact on other useful or desired properties. See, e.g., Mansfield, 9 Mol. Cell. Probes 145-156 (1995).
[0200] Detectable labels can be incorporated into nucleic acids by covalent or non-covalent means, e.g., by transcription, such as by random-primer labeling using Klenow polymerase, or nick translation, or amplification, or equivalent as is known in the art. For example, a nucleotide base is conjugated to a detectable moiety, such as a fluorescent dye, e.g., Cy3.RTM. or Cy50 and then incorporated into genomic nucleic acids during nucleic acid synthesis or amplification. Nucleic acids can thereby be labeled when synthesized using Cy3.RTM.- or Cy5.RTM.-dCTP conjugates mixed with unlabeled dCTP.
[0201] Nucleic acid probes can be labeled by using PCR or nick translation in the presence of labeled precursor nucleotides, for example, modified nucleotides synthesized by coupling allylamine-dUTP to the succinimidyl-ester derivatives of the fluorescent dyes or haptens (such as biotin or digoxigenin) can be used; this method allows custom preparation of most common fluorescent nucleotides, see, e.g., Henegariu, 18 Nat. Biotechnol. 345-348 (2000).
[0202] Nucleic acid probes may be labeled by non-covalent means known in the art. For example, Kreatech Biotechnology's Universal Linkage System.RTM. (ULS.RTM.) provides a non-enzymatic labeling technology, wherein a platinum group forms a co-ordinative bond with DNA, RNA or nucleotides by binding to the N7 position of guanosine. This technology may also be used to label proteins by binding to nitrogen and sulphur containing side chains of amino acids. See, e.g., U.S. Pat. Nos. 5,580,990; 5,714,327; and 5,985,566; and European Patent No. 0539466.
[0203] The binding of a probe to the marker sequence flanking the tandem repeat region may be determined by hybridization as is well known in the art. Hybridization may be detected in real time or in non-real time.
[0204] TaqMan.RTM. probes (Heid, et al., 1996) use the fluorogenic 5' exonuclease activity of Taq polymerase to measure the amount of target sequences in cDNA samples. TaqMan.RTM. probes are oligonucleotides that contain a donor fluorophore usually at or near the 5' base, and a quenching moiety typically at or near the 3' base. The quencher moiety may be a dye such as TAMRA or may be a non-fluorescent molecule such as 4-(4-dimethylaminophenylazo) benzoic acid (DABCYL). See Tyagi, et al., 16 Nature Biotechnology 49-53 (1998). When irradiated, the excited fluorescent donor transfers energy to the nearby quenching moiety by FRET rather than fluorescing. Thus, the close proximity of the donor and quencher prevents emission of donor fluorescence while the probe is intact.
[0205] TaqMan.RTM. probes are designed to anneal to an internal region of a PCR product. When the polymerase (e.g., reverse transcriptase) replicates a template on which a TaqMan.RTM. probe is bound, its 5' exonuclease activity cleaves the probe. This ends the activity of quencher (no FRET) and the donor fluorophore starts to emit fluorescence which increases in each cycle proportional to the rate of probe cleavage. Accumulation of PCR product is detected by monitoring the increase in fluorescence of the reporter dye (note that primers are not labeled). If the quencher is an acceptor fluorophore, then accumulation of PCR product can be detected by monitoring the decrease in fluorescence of the acceptor fluorophore.
[0206] TaqMan.RTM. assay uses universal thermal cycling parameters and PCR reaction conditions. Because the cleavage occurs only if the probe hybridizes to the target, the fluorescence detected originates from specific amplification. The process of hybridization and cleavage does not interfere with the exponential accumulation of the product. One specific requirement for fluorogenic probes is that there be no G at the 5' end. A `G` adjacent to the reporter dye quenches reporter fluorescence even after cleavage.
[0207] Other methods of probe hybridization detected in real time can be used for detecting amplification of mutated nucleic acids. For example, the commercially available MGB Eclipse.TM. probes (Epoch Biosciences), which do not rely on a probe degradation can be used. MGB Eclipse.TM. probes work by a hybridization-triggered fluorescence mechanism. MGB Eclipse.TM. probes have the Eclipse.TM. Dark Quencher and the MGB positioned at the 5'-end of the probe. The fluorophore is located on the 3'-end of the probe. When the probe is in solution and not hybridized, the three dimensional conformation brings the quencher into close proximity of the fluorophore, and the fluorescence is quenched. However, when the probe anneals to a target sequence, the probe is unfolded, the quencher is moved from the fluorophore, and the resultant fluorescence can be detected.
[0208] Suitable donor fluorophores include 6-carboxyfluorescein (FAM), tetrachloro-6-carboxyfluorescein (TET), 2'-chloro-7'-phenyl-1,4-dichloro-6-carboxyfluorescein (VIC) and the like. Suitable quenchers include tetra-methylcarboxyrhodamine (TAMRA) 4-(4-dimethylaminophenylazo) benzoic acid ("DABCYL" or a DABCYL analog) and the like. Tetramethylrhodamine (TMR) or 5-carboxyrhodamine 6G (RHD) may be combined as donor fluorophores with DABCYL as quencher. Multiplex TaqMan.RTM. assays can be performed using multiple detectable labels each comprising a different donor and quencher combination. Probes for detecting amplified sequence in real time may be stored frozen (-10.degree. to -30.degree. C.) as 100 M stocks. TaqMan.RTM. probes are available from Applied BioSystems (4316032).
[0209] In a preferred embodiment, real time PCR is performed using TaqMan.RTM. probes in combination with a suitable amplification/analyzer such as the ABI Prism 7900HT Sequence Detection System. The ABI PRISM.RTM. 7900HT Sequence Detection System is a high-throughput real-time PCR system that detects and quantitates nucleic acid sequences. Briefly, TaqMan.RTM. probes specific for each allele are included in the PCR assay. These probes contain a reporter dye at the 5' end and a quencher dye at the 3' end. Each allele specific probe is conjugated with a different fluorescent reporter dye. During PCR, the fluorescently labeled probes bind specifically to their respective target sequences; the 5' nuclease activity of Taq polymerase cleaves the reporter dye from the probe and a fluorescent signal is generated. The increase in fluorescence signal is detected only if the target sequence is complementary to the probe and is amplified during PCR. A mismatch between probe and target greatly reduces the efficiency of probe hybridization and cleavage. The ABI Prism 7700HT or 7900HT Sequence detection System measures the increase in fluorescence during PCR thermal cycling, providing "real time" detection of PCR product accumulation.
[0210] Real Time detection on the ABI Prism 7900HT or 7900HT Sequence Detector monitors fluorescence and calculates the measure of reporter signal, or Rn value, during each PCR cycle. The threshold cycle, or Ct value, is the cycle at which fluorescence intersects the threshold value. The threshold value is determined by the sequence detection system software or manually.
[0211] To minimize the potential for cross contamination, reagent and mastermix preparation, specimen processing and PCR setup, and amplification and detection are all carried out in physically separated areas. In addition, Uracil-N-Glycosylase is utilized (along with the incorporation of Uracil into PCR amplicons) to eliminate carry over contamination.
[0212] The following examples serve to illustrate the present invention. These examples are in no way intended to limit the scope of the invention.
Example 1
E746_A750 Deletion
[0213] Background:
[0214] Somatic mutations in the tyrosine kinase (TK) domain of the EGFR gene are associated with clinical response to TK inhibitors in patients with non-small cell lung cancer (NSCLC). An assay that detects such mutations in plasma provides a noninvasive procedure to assess suitability for TK inhibitor therapy. Described below are 1) the development of a sensitivity assay to detect the E746_A750 deletion (E746_A750del) in the TK domain of EGFR in plasma; and 2) optimization of the assay for plasma DNA extraction.
[0215] Methods:
[0216] The assay uses MseI to specifically digest wild-type (WT) genomic DNA (gDNA) to reduce non-specific amplification. After digestion, samples are PCR-amplified using one unlabeled primer and one FAM-labeled primer spanning the E746_A750 deletion region. The fluorescence signal is detected with an automated genetic analyzer. Serial dilution studies were conducted using H1650 gDNA, which is a E746_A750 deletion cell line) diluted in WT gDNA after MseI digestion. To assess detection of the deletion in plasma, 3-4 mL of whole blood was spiked with 1-37 ng H1650 gDNA; gDNA from the separated plasma was then extracted by silica column/2-propanol precipitation, digested with MseI, and amplified as above. Several extraction methods (silica column, magnetic bead, phenol:chloroform, and 2-propanol precipitation) were evaluated using pooled plasma samples and phosphate buffer solution (PBS) spiked with 10-350 ng of gDNA.
[0217] Results:
[0218] Using a combined approach of digesting the WT EGFR allele followed by deletion-specific fluorescent PCR, the equivalent of circa 1 copy of the E746_A750 deletion (10 pg gDNA) diluted to 0.001% could be detected. Furthermore, the E746_A750 deletion was successfully detected in 1/5 the final DNA volume (5 .mu.l) in all spiked blood samples. In the DNA extraction method evaluation, the magnetic bead-based method yielded the highest percent recovery of gDNA from PBS (69% recovery of the 10 ng sample). Phenol:chloroform extraction gave the highest yield with pooled plasma samples.
[0219] Conclusions:
[0220] The combination of an optimized DNA extraction method, clearing the plasma DNA sample of amplifiable WT DNA by restriction digestion, and mutation-specific fluorescent PCR provides a highly sensitive assay for detection of somatic mutations in plasma.
Example 2
Preparation of and Sensitivity Studies for Detection of EGFR Mutations
[0221] Serial dilution studies were conducted using H1650 gDNA (E746_A750 deletion mutation cell line) diluted in wild-type gDNA with no treatment of after MseI digestion. For each dilution, 10 pg of H1650 gDNA. H1650 is a heterozygous cell line, thus, 50% of the DNA contributes to the deletion allele and 50% to the wild-type allele) was spiked into wild-type gDNA at varying concentrations to yield 0.001%-10% of the exon 19 deletion in the background of wild-type allele. Nucleic acid is spiked into a sample in order to control the amount of target nucleic acid in a sample and test the sensitivity of the assay to detect various amounts, in particular low amounts, of target nucleic acid. The results show that peaks were detected corresponding to the deletion mutant at levels of 0.01% in non-treated samples and at least 0.001% when digested with MseI. These results are presented in Table 1.
TABLE-US-00001 TABLE 1 Exon 19 deletion tumor DNA spiked into purified DNA wild-type DNA % Exon 19 DNA Amount per Peak Detected deletion reaction Concentration No MseI allele Wild-Type Exon 19 Deletion Treatment Treated 10.00% 45 pg 10 pg (50% E746_A750del) Yes Yes 1.000% 495 pg 10 pg (50% E746_A750del) Yes No 0.100% 5 ng 10 pg (50% E746_A750del) Yes Yes 0.010% 50 ng 10 pg (50% E746_A750del) Yes Yes 0.001% 500 ng 10 pg (50% E746_A750del) No Yes 0.000% 500 ng 0 pg No No *10 pg of tumor DNA was spiked into wild-type DNA at varying concentrations, but since 1 allele is wild-type, only half of the tumor DNA contributes to exon 19 deletion DNA and the other half to the wild-type allele
[0222] To assess detection of the deletion in plasma, 3-4 mL whole blood was spiked with 1-37 ng H1650 gDNA; gDNA from the separated plasma was then extracted (QiaAmp DNA Blood Midi Kit), further concentrated by ethanol or isopropanol precipitation, and digested with MseI. The digested DNA was then subjected to fluorescent PCR using deletion-specific primers.
TABLE-US-00002 TABLE 2 Exon 19 deletion tumor DNA spiked into blood Peak Detected Amt of Spiked No Precipitation Method H1650 gDNA Treatment Mse I Treated Ethanol Precipitation 37 ng No Yes 1 ng No No Isopropanol Precipitation 37 ng NT Yes 1 ng NT Yes *All samples were first extracted using the QiaAmp DNA Blood Midi kit, then further precipitated by ethanol or isopropanol precipitation NT, Not tested
Example 3
Method Comparison of EGFR Mutation Detection
[0223] A method comparison was performed to demonstrate the sensitivity of the EGFR mutation detection assay disclosed herein (also referred to as the Sanders method) which is designed to be able to detect mutations in plasma from NSCLC patients. In this study, the method disclosed herein was compared with two other methods (Asano, 2006 and Ohnishi, 2006) that claim high sensitivity for detecting E746_A750del. The Asano and Ohnishi methods were performed as described in the respective publications but included a fluorescent label on the forward primer. Amplification products were then analyzed by capillary electrophoresis for fluorescent detection of the PCR fragments using an ABI 3100 Genetic Analyzer. Table 3 shows the expected and observed fragment sizes for each fragment.
TABLE-US-00003 TABLE 3 Expected and observed fragment sizes Method Fragment Expected Size Observed Size Sanders Wild-Type EGFR 197 194 E746_A750del 153 151 Asano Wild-Type EGFR 138 135 E746_A750del 123 120 Ohnishi E746_A750del 133 138 *Due to the mobility shift of PCR primer/products fluorescently labeled with FAM, some of the amplicons are slightly shifted from the expected size in the ABI 3100 Genetic Analyzer.
[0224] The assay disclosed herein has been previously detected as little as 10 pg of H1650 cell line DNA (circa 1 copy of the E746_A750del mutation) at as low as 0.001% in the background of the wild-type EGFR gene. Therefore, all three methods (Sanders, Asano and Ohnishi) were tested for their ability to detect 10 pg of H1650 cell line DNA at levels ranging from 0.0005% to 10% in the background of the wild-type EGFR gene (Table 4).
TABLE-US-00004 TABLE 4 Sensitivity of Sanders, Asano, and Ohnishi methods for detecting the E746_A750del mutation. Sanders Method Asano Method Ohnishi Method Wild-Type Deletion Peak Wild-Type Deletion Peak Deletion Peak Peak Intensity Intensity Peak Intensity Intensity % H1650 DNA Intensity (1/5) (120 bp)* (135 bp)* (138 bp)* 10% 937 0 5811 0 0 1% 989 166 7209 195 0 0.10% 1395 152 2685 1746 0 0.01% 520 1395 1004 3682 0 0.001% 820 796 97 7087 0 0.0005% 1184 2321 0 7338 167 0.0003% 699 3942 NT NT NT 0.0002% 803 3324 NT NT NT 0.0001% 0 3798 0 4603 NT 0% (5 .mu.g) 0 2287 0 3836 NT 0% (50 ng) 0 0 0 5280 0 *Sizes indicated are the actual size NT = Not Tested
[0225] The results of this comparison indicate that the Sanders method detects circa 1 copy of the E746_A750del mutation in as little as 0.0005% in the background of the wild-type EGFR gene. (6.6 picograms (pg) is equivalent to one copy, so 10 pg is 1.5 copies, thus, circa is used to indicate the approximate copy number.) The Asano method demonstrated strong peaks at 0.01-10% levels and a weak peak at 0.001%, but no detectable amplification at 0.0005%. The Ohnishi method was unable to detect circa 1 copy of the E746_A750del mutation as determined by the absence of any peak from 0.001%-10% levels. However, a weak peak was observed in the 0.0005% sample, although this is most likely attributed to background amplification of the wild-type EGFR gene due to the very high levels of wild-type DNA present in the sample.
[0226] The Asano and Ohnishi methods were also tested for their ability to detect the E746_A750del mutation at high and low copy numbers without interfering wild-type DNA spike into the sample. Table 5 shows that the Asano method was successful at detecting both a high copy numbers (850 pg=130 copies) and low copy numbers (10 pg=.about.1 copy) of the mutation, as also demonstrated above. However, while the Ohnishi method successfully detected high copy numbers of the mutation, it failed at detecting low copy numbers.
TABLE-US-00005 TABLE 5 Detection of low and high copy numbers of the E746_A750del mutation. Asano Method Ohnishi Method Deletion Peak Wild Type Deletion Peak H1650 DNA Intensity Peak Intensity Intensity (pg/rxn) (120 bp)* (135 bp)* (138 bp)* 850 7345 0 2008 10 2422 0 0 *Sizes indicated are the actual size
[0227] The results presented in this study confirm that the Sanders assay for detection of the exon 19 EGFR deletion (E746_A750del) utilizing restriction digestion followed by deletion specific fluorescent PCR, demonstrates superior sensitivity over methods in the prior art, in particular, the methods described by Asano and Ohnishi.
Example 4
[0228] Detection of E746_A750del Mutation in Plasma of a NSCLC Patient
[0229] The assay tested two separate DNA extractions from plasma of a single NSCLC patient. Each extraction was digested with MseI and subsequently split into 11 separate PCR reactions. Two reactions yielded positive results as shown in Table 6 below.
TABLE-US-00006 TABLE 6 E746_A750del Mutation in the Plasma of a NSCLC Patient Peak Intensity 500 .mu.l Plasma 350 .mu.l Plasma Primer set Aliquot# 1:10 Undiluted 1:10 Undiluted E746_A750del 1 0 0 53 163 2 0 0 0 0 3 3568 7952 0 0 4 0 0 0 0 5 0 0 0 0 6 0 0 0 0 7 0 0 0 0 8 0 0 0 0 9 3971 7359 0 0 10 0 0 0 0 11 0 0 2518 6469 WT 1 0 -- 0 --
Example 5
DNA Extraction Comparison
[0230] Detection of rare mutations in plasma is a difficult feat and is dependant on both the ability to detect very low amounts of the mutation among large amounts of normal DNA and the ability to successfully recover the mutation from the patient plasma sample. Using a high quality DNA extraction method in conjunction with the methods and compositions provided herein, particularly combining non-target fragmentation with mutation specific primers further increases the ability to detect low copy target nucleic acid in patient samples, particularly plasma.
[0231] To determine an optimal method of DNA extraction, eight DNA extraction methods were evaluated to find a method that is superior for obtaining high yield DNA as well as providing DNA that is amenable to the detection methods provided herein. Six plasma samples for each extraction method were spiked with various nucleic acids including every plasma sample being spiked with the EGFR exon 19 deletion (H1650 cell line gDNA) for subsequent evaluation of detection. All six plasma samples were extracted using the eight methods that included two lysis methods using magnetic bead based methods (Agencourt Genfind.TM. and Ambion MagMax.TM.), two column based methods (Qiagen QIAmp DNA Blood Mini Kit and the automated Corbett DNA Xtractor.TM.), two isopropanol precipitation based methods (Gentra Puregene.TM. and Roche Cobas.RTM.), and two phenol:chloroform based methods (standard method and with Eppendorf Phase Lock Gels.RTM.). The resulting yield was determined by picogreen fluorescent assay (Invitrogen Quant-iT.TM. PicoGreen.RTM. dsDNA Quantitation Assay) and the mean percent recovery of the six plasma samples was calculated for each method. Comparison of these eight DNA extraction methods revealed that three methods were clearly more efficient at DNA recovery than the other five (FIG. 14). These methods included Agencourt Genfind.TM., Roche Cobas.RTM. and phenol:chloroform extraction using Eppendorf Phase Lock Gels.RTM..
[0232] Ambion MagMax.TM. is an isolation kit which can be used to isolate RNA or DNA from serum, plasma, or any other biofluid. Agencourt Genfind.TM. is a DNA isolation for blood, serum, or plasma samples. Both involve sample disruption with a lysis reagent followed by binding of the nucleic acid to magnetic beads (proprietary chemistry). The beads are then washed with a series of buffers to reduce and/or eliminate proteins and other contaminants from the sample in order to purify the nucleic acid. The nucleic acid is then eluted off the beads to yield the final DNA sample.
[0233] QIAamp DNA Blood Mini Kit and Corbett DNA Xtractor.TM. are both column based methods. The QIAamp procedure used in these studies was manual while the Corbett was an automated system. Both involve disruption of the sample with a lysis reagent followed by binding to a silica column. The column containing bound nucleic acid is washed with a series of wash buffers and the nucleic acid is then eluted in the last step with elution buffer to yield the final sample.
[0234] Gentra Puregene.TM. and Roche Cobas.RTM. methods are crude extractions and both incorporate treatment of plasma with a lysis reagent to disrupt the sample followed by isopropanol precipitation of DNA.
[0235] Phenol:Chloroform extraction involves separation of organic and aqueous phases of the plasma. The aqueous phase containing nucleic acid is isolated and re-extracted once more with phenol:chloroform. The aqueous phase is again isolated and the DNA is purified from this phase by isopropanol precipitation.
[0236] Phenol:Chloroform extraction using Phase Lock Gels.RTM. employs the phenol:chloroform procedure as described above, but once separated the aqueous and organic phases are separated by a solid gel. This allows increased recovery of the aqueous phase without contamination from the organic phase which can lead to inhibition of PCR.
[0237] Further evaluations were performed to identify the best method from the three yielding the highest amounts of DNA. For this comparison, 18% of the final DNA sample from each extraction was subjected to our EGFR mutation detection assay as described in the Examples above. The mean peak intensity of the PCR products obtained corresponding to the E746_A750del EGFR mutation were calculated as an indicator of DNA quality and recovery of the spiked mutation (FIG. 15). Peak intensity values indicated that of the three methods tested, the Agencourt Genfind.TM. method provided the most robust amplification of spiked mutation.
[0238] Once the superior method was identified, further evaluation of the six nucleic acid carrier conditions was performed to determine if either condition facilitated recovery of the spiked mutation. These nucleic acid carrier conditions included use of RNA carrier or no carrier at low and high (100 nanograms (ng) spiked normal DNA) plasma DNA levels and the use of normal DNA as a carrier (395 ng and 778 ng). To determine optimal nucleic acid carrier conditions, dilutions of each sample were analyzed using our detection method. The concentration of mutant DNA in the extracted samples was first estimated based on the calculated percent recovery for each sample. The equivalent of 15 pg of H1650 DNA was used as the starting point and was further diluted 1:2 and 1:4. All three dilutions were analyzed for the presence of detectable spiked EGFR mutation. The samples employing RNA carrier were the only ones that detected the mutation in the 1:4 dilution indicating that spiking RNA carrier during the extraction facilitates recovery of the mutation present in the plasma sample (FIG. 16). FIG. 16 provides the results from evaluation of nucleic acid spiking conditions. The six nucleic acid carrier conditions include 1) no carrier, 2) no carrier plus 100 ng of normal DNA, 3) 1 .mu.g of RNA carrier, 4) 1 .mu.g of RNA carrier plus 100 ng of normal DNA, 5) 395 ng of normal DNA as a carrier, and 6) 778 ng of normal DNA as a carrier. Plasma samples spiked with 100 ng of normal DNA were included to represent patient plasma samples containing high amounts of DNA and were spiked immediately after thawing of the plasma sample. The two samples containing 395 ng and 778 ng of normal DNA were spiked following sample lysis during the extraction, the point at which nucleic acid carrier is to be added. Columns represent the mean peak intensity of E746_A750del PCR product.
[0239] The experiments performed in this study identified an optimal method for obtaining high yield DNA from plasma samples that is amenable to detection of rare mutations. This method was further improved by identifying nucleic acid spiking conditions that facilitate the recovery of mutations from the sample. Thus to improve the ability to recover rare mutations in the plasma and to successfully detect such mutations, it was determined that the Agencourt Genfind.TM. method with a modification to include addition of RNA carrier to the sample lysate is the choice method.
[0240] While Agencourt Genfind.TM. was the optimal extraction method in these experiments, any of the extraction methods tested can be used and are provide as exemplary extraction methods that can be used in conjunction with the methods and compositions provided herein. In different hands, results may vary, but all are acceptable methods.
Example 6
Detection Sensitivity of L858R in Wild-Type Background
[0241] DNA samples were initially digested with restriction enzymes (New England Biolabs) to cleave TTAA (MseI) and TGGCCA (MscI) recognition sites targeting wild type EGFR sequences (Mse I for singleplex E746_A750del; Msc I for singleplex L858R; Mse I and Msc I for multiplex reactions) for 2 hours at 37.degree. C. followed by 20 minute inactivation at 65.degree. C. Following restriction digestion, samples were amplified with AccuPrime.TM. Taq DNA Polymerase (Invitrogen) using E746_A750del and/or L858R mutation-specific primers (E746_A750del forward: 5'-[6FAM] CCC GTC GCT ATC AAA ACA TC-3' (SEQ ID NO:1); E746_A750del reverse: 5'-ATG TGG AGA TGA GCA GGG TCT-3' (SEQ ID NO:2); L858R forward: [6FAM] TCA CAG ATT TTG GGC GG-3' (SEQ ID NO:7); L858R reverse: CCT GGT GTC AGG AAA ATG CT-3' (SEQ ID NO:8)). In each set, the forward primer was labeled with 5'-6FAM. For added specificity, the L858R forward primer contained a locked nucleic acid (LNA) in the -1 position corresponding to the mutated base. Thermocycling conditions were as follows: denatured at 95.degree. C. for 5 min; amplified with 40 cycles of 94.degree. C. for 40 seconds, 55.degree. C. (E746_A750del singleplex PCR) or 61.7.degree. C. (L858R singleplex PCR and multiplex PCR) for 1 minute, 72.degree. C. for 1 minute; final extension at 72.degree. C. for 7 minutes. E746_A750del PCR product yielded an expected size of 153 bp and L858R PCR product yielded an expected size of 113 bp.
TABLE-US-00007 TABLE 7 Detection of L858R Mutation in the Background of Wild type EGFR. Mean Peak Intensity.sup.1 % L858R (110 bp).sup.2 0.001% 463 0.0005% 412 0.0003% -- 0.0002% -- 0% (2.5 ug/rxn) -- .sup.1Peak intensities <200 RFU are not reported .sup.2Size represents observed L858R peak size on ABI 3100 Genetic Analyzer
[0242] Several other mutation specific primers for detecting the L858R mutation were tested but were not effective in determining the presence of the mutant nucleic acid. The primers are provided in the table below:
TABLE-US-00008 TABLE 8 L858R Mutation Specific Primers. Mutant Base as a Locked SEQ Nucleic Acid ID NO: Primer Sequence (Yes or No) 13 ATCACAGATTTTGGGCG Yes 14 CAAGATCACAGATTTTGGGCG No 15 ATGTCAAGATCACAGATTTTGGGCG No 16 AGATCACAGATTTTGGGCG No 17 ATCACAGATTTTGGGCGG No 18 TCACAGATTTTGGGCGGG No 19 ATTTTGGGCGGGCCAAAC No 20 AGATTTTGGGCGGGCCA No 21 ATCACAGATTTTGGGCGG Yes * The bolded underlined base is the location of the L858R mutant base.
Example 7
EGFR Mutation Detection in Paired Tissue and Plasma Samples
[0243] Paired FFPE tissue and plasma were obtained from 11 NSCLC donors with informed consent (10 from Indivumed, Hamburg, Germany; 1 from Good Samaritan Hospital, Kearney, Nebr.). DNA from FFPE tissue was analyzed by PCR followed by direct sequencing to determine mutation status of the paired tissue/plasma donors. One donor provided two plasma samples (856 and 3107) 1 year apart for analysis. In addition, 6 normal plasma samples and 5 plasma samples spiked with E746_A750del or L858R mutations were analyzed by the methods described herein.
[0244] Results of direct sequencing of FFPE tissue and multiplex fluorescent RF-PCR of plasma samples are presented in Table 3 along with tumor size and overall plasma DNA concentration. Overall 3/11 (27%) NSCLC donors had mutation positive FFPE tissue, while the remaining 8 were negative for the two mutations. Of the paired NSCLC samples, 83.3% (10/12) of plasma samples demonstrated identical mutation status to the matched FFPE tissue specimens. The two E746_A750del positive plasma samples were drawn from the same donor 1 year apart, thus 81.8% (9/11) unique donors had identical mutation status between paired samples. Notably, although the overall plasma DNA concentration had decreased by nearly half, the latter plasma specimen had twice as many positive wells as the specimen drawn 1 year previous (data not shown), suggesting the circulating mutation concentration had increased in that time.
[0245] For the mutation spiked plasma samples, 100% of samples spiked with 100, 200, and 300 pg of E746_A750del positive DNA (H1650 cell line) and 100% of samples spiked with 100 and 300 pg of L858R positive DNA (H1975 cell line) tested positive for their respective mutation. Furthermore, 6 normal plasma specimens tested negative for either mutation as expected.
TABLE-US-00009 TABLE 9 EGFR Mutation Detection in Paired Tissue and Plasma Samples Tumor EGFR Mutation Detected Sample Diameter Plasma DNA Tissue Plasma ID (cm) Conc. (ng/mL) PCR/Direct Seq RF-PCR NSCLC 856.sup.1 6.5.sup.2 15.6 E746_A750del E746_A750del 3107.sup.1 6.5.sup.2 8.1 E746_A750del E746_A750del 378 8.5 144 -- -- 401 5 20.6 -- -- 455 3.4 212 -- -- 477 3.5 54.0 -- -- 497 2.5 26.5 L858R -- 516 5.7 11.1 -- -- 532 3.5 41.5 -- -- 563 1.9 265 L858R -- 631 2.9 128 -- -- 662 10.5 386 -- -- E746_A750del Spiked 200 pg N/A 23.3 N/A E746_A750del 300 pg N/A 9.9 N/A E746_A750del 100 pg N/A 7.6 N/A E746_A750del L858R spiked 300 pg N/A 8.0 N/A L858R 100 pg N/A 6.4 N/A L858R Normal 4207 N/A 4.3 N/A -- 2011 N/A 5.8 N/A -- 2258 N/A 8.7 N/A -- 3391 N/A 7.4 N/A -- 975 N/A 4.4 N/A -- 725 N/A 7.1 N/A -- .sup.1Sample IDs 856 and 3107 are from the same patient. Sample ID 3107 was drawn 1 year after Sample ID 856. .sup.2Tumor size measured 4.7 years prior to initial draw and 5.7 years prior to second draw.
[0246] Unless otherwise defined, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which this invention belongs.
[0247] The inventions illustratively described herein may suitably be practiced in the absence of any element or elements, limitation or limitations, not specifically disclosed herein. Thus, for example, the terms "comprising," "including," "containing," etc. shall be read expansively and without limitation. Additionally, the terms and expressions employed herein have been used as terms of description and not of limitation, and there is no intention in the use of such terms and expressions of excluding any equivalents of the features shown and described or portions thereof, but it is recognized that various modifications are possible within the scope of the invention claimed.
[0248] Thus, it should be understood that although the present invention has been specifically disclosed by preferred embodiments and optional features, modification, improvement and variation of the inventions embodied therein herein disclosed may be resorted to by those skilled in the art, and that such modifications, improvements and variations are considered to be within the scope of this invention. The materials, methods, and examples provided here are representative of preferred embodiments, are exemplary, and are not intended as limitations on the scope of the invention.
[0249] The invention has been described broadly and generically herein. Each of the narrower species and subgeneric groupings falling within the generic disclosure also form part of the invention. This includes the generic description of the invention with a proviso or negative limitation removing any subject matter from the genus, regardless of whether or not the excised material is specifically recited herein.
[0250] In addition, where features or aspects of the invention are described in terms of Markush groups, those skilled in the art will recognize that the invention is also thereby described in terms of any individual member or subgroup of members of the Markush group.
[0251] All publications, patent applications, patents, and other references mentioned herein are expressly incorporated by reference in their entirety, to the same extent as if each were incorporated by reference individually. In case of conflict, the present specification, including definitions, will control.
[0252] Other embodiments are set forth within the following claims.
Sequence CWU 1
1
35120DNAArtificial SequenceDescription of Artificial Sequence
Synthetic primer 1cccgtcgcta tcaaaacatc 20221DNAArtificial
SequenceDescription of Artificial Sequence Synthetic primer
2atgtggagat gagcagggtc t 21321DNAArtificial SequenceDescription of
Artificial Sequence Synthetic primer 3cttccacaat ggttgaacta g
21420DNAArtificial SequenceDescription of Artificial Sequence
Synthetic primer 4catccatgtc cctacaaagg 20522DNAArtificial
SequenceDescription of Artificial Sequence Synthetic primer
5gagccattta tacagaaaga tg 22620DNAArtificial SequenceDescription of
Artificial Sequence Synthetic primer 6gaaataccat ttgacccagc
20717DNAArtificial SequenceDescription of Artificial Sequence
Synthetic primer 7tcacagattt tgggcgg 17820DNAArtificial
SequenceDescription of Artificial Sequence Synthetic primer
8cctggtgtca ggaaaatgct 20919DNAArtificial SequenceDescription of
Artificial Sequence Synthetic primer 9cgagctaagc aggaggcgg
191020DNAArtificial SequenceDescription of Artificial Sequence
Synthetic primer 10gtccatagtc gctggaggag 201119DNAArtificial
SequenceDescription of Artificial Sequence Synthetic primer
11cgagctaagc aggaggcgg 191221DNAArtificial SequenceDescription of
Artificial Sequence Synthetic primer 12actttcagcc tgatagtctg g
211317DNAArtificial SequenceDescription of Artificial Sequence
Synthetic primer 13atcacagatt ttgggcg 171421DNAArtificial
SequenceDescription of Artificial Sequence Synthetic primer
14caagatcaca gattttgggc g 211525DNAArtificial SequenceDescription
of Artificial Sequence Synthetic primer 15atgtcaagat cacagatttt
gggcg 251619DNAArtificial SequenceDescription of Artificial
Sequence Synthetic primer 16agatcacaga ttttgggcg
191718DNAArtificial SequenceDescription of Artificial Sequence
Synthetic primer 17atcacagatt ttgggcgg 181818DNAArtificial
SequenceDescription of Artificial Sequence Synthetic primer
18tcacagattt tgggcggg 181918DNAArtificial SequenceDescription of
Artificial Sequence Synthetic primer 19attttgggcg ggccaaac
182017DNAArtificial SequenceDescription of Artificial Sequence
Synthetic primer 20agattttggg cgggcca 172118DNAArtificial
SequenceDescription of Artificial Sequence Synthetic primer
21atcacagatt ttgggcgg 1822770DNAHomo sapiens 22ccacacggac
tttataacag gctttacaag cttgagattc ttttatctaa ataatcagtg 60tgattcgtgg
agcccaacag ctgcagggct gcgggggcgt cacagccccc agcaatatca
120gccttaggtg cggctccaca gccccagtgt ccctcacctt cggggtgcat
cgctggtaac 180atccacccag atcactgggc agcatgtggc accatctcac
aattgccagt taacgtcttc 240cttctctctc tgtcataggg actctggatc
ccagaaggtg agaaagttaa aattcccgtc 300gctatcaagg aattaagaga
agcaacatct ccgaaagcca acaaggaaat cctcgatgtg 360agtttctgct
ttgctgtgtg ggggtccatg gctctgaacc tcaggcccac cttttctcat
420gtctggcagc tgctctgctc tagaccctgc tcatctccac atcctaaatg
ttcactttct 480atgtctttcc ctttctagct ctagtgggta taactccctc
cccttagaga cagcactggc 540ctctcccatg ctggtatcca ccccaaaagg
ctggaaacag gcaattactg gcatctaccc 600agcactagtt tcttgacacg
catgatgagt gagtgctctt ggtgagcctg gagcatgggt 660attgtttttg
gtattttttg gatgaagaaa tggaggcata aagaaattgg ctgaccctta
720tatggctggg atagggttta agccccttgt tatttctgac tctgaaactt
77023755DNAHomo sapiens 23ccacacggac tttataacag gctttacaag
cttgagattc ttttatctaa ataatcagtg 60tgattcgtgg agcccaacag ctgcagggct
gcgggggcgt cacagccccc agcaatatca 120gccttaggtg cggctccaca
gccccagtgt ccctcacctt cggggtgcat cgctggtaac 180atccacccag
atcactgggc agcatgtggc accatctcac aattgccagt taacgtcttc
240cttctctctc tgtcataggg actctggatc ccagaaggtg agaaagttaa
aattcccgtc 300gctatcaaaa catctccgaa agccaacaag gaaatcctcg
atgtgagttt ctgctttgct 360gtgtgggggt ccatggctct gaacctcagg
cccacctttt ctcatgtctg gcagctgctc 420tgctctagac cctgctcatc
tccacatcct aaatgttcac tttctatgtc tttccctttc 480tagctctagt
gggtataact ccctcccctt agagacagca ctggcctctc ccatgctggt
540atccacccca aaaggctgga aacaggcaat tactggcatc tacccagcac
tagtttcttg 600acacgcatga tgagtgagtg ctcttggtga gcctggagca
tgggtattgt ttttggtatt 660ttttggatga agaaatggag gcataaagaa
attggctgac ccttatatgg ctgggatagg 720gtttaagccc cttgttattt
ctgactctga aactt 755241562DNAArtificial SequenceDescription of
Artificial Sequence Synthetic construct 24ccagctccgt tcagagtgaa
ccatgcagtg gaatggtaag tggcattata agccccagtg 60atcttccaga tagccctgga
caaaccatgc caccaagcag aagtaaaaca cctccaccac 120ctcctcaaac
agctcaaacc aagcgagaag tacctaaaaa taaagcacct actgctgaaa
180agagagagag tggacctaag caagctgcag taaatgctgc agttcagagg
gtccaggttc 240ttccagatgc tgatacttta ttacattttg ccacggaaag
tactccagat ggattttctt 300gttcatccag cctgagtgct ctgagcctcg
atgagccatt tatacagaaa gatgtggaat 360taagaataat gtgcatgtgt
ctttatagca gcatgattta tactcatttg ggtatatacc 420cagtaatggg
atggctgggt caaatggtat ttctagttct agatccctga ggaatcgcca
480cactgacttc cacaatggtt gaactagttt acagtcccac caagaaaatg
tggcacatat 540acaccatgga atactatgca gccataaaaa atgatgagtt
catatccttt gtagggacat 600ggatgaaatt ggaaaccatc attctcagta
aactatcgca agaacaaaaa accaaacacc 660gcatattctc acttataggt
gggaattgaa caatgagatc acatggacac aggaagggga 720atatcacact
ctggggactg tggtggggtc gggggagggg ggagggatag cattgggaga
780tatacctaat gctagatgac acattagtgg gtgcagcgca gcatggcaca
tgtatacata 840tgtaactaac ctgcacaatg tgcacatgta ccctaaaact
tagagtataa taaaaaaaaa 900aaaaaaaaaa ataacaataa atgagataaa
atctaaaaaa aaaaaaaaaa aaaaaaaaaa 960aaaaaaaaaa aaaaaaaaaa
aaaaaaaaaa aaaaaaaaaa aaaaaaaaaa aaaaaaaaaa 1020aaaaaaaaaa
aaaaaaaaaa aaaaaaaaaa aaaaaaaaaa aaaaaaaaaa aaaaaaaaaa
1080aaaaaaaaaa aaaaaaaaaa aaaaaaaaaa aaaaaaaaaa agaataatgc
ctccagttca 1140ggaaaatgac aatgggaatg aaacagaatc agagcagcct
aaagaatcaa atgaaaacca 1200agagaaagag gcagaaaaaa ctattgattc
tgaaaaggac ctattagatg attcagatga 1260tgatgatatt gaaatactag
aagaatgtat tatttctgcc atgccaacaa agtcatcacg 1320taaagcaaaa
aagccagccc agactgcttc aaaattacct ccacctgtgg caaggaaacc
1380aagtcagctg cctgtgtaca aacttctacc atcacaaaac aggttgcaac
cccaaaagca 1440tgttagtttt acaccggggg atgatatgcc acgggtgtat
tgtgttgaag ggacacctat 1500aaacttttcc acagctacat ctctaagtga
tctaacaatc gaatcccctc caaatgagtt 1560ag 1562251562DNAArtificial
SequenceDescription of Artificial Sequence Synthetic construct
25ccagctccgt tcagagtgaa ccatgcagtg gaatggtaag tggcattata agccccagtg
60atcttccaga tagccctgga caaaccatgc caccaagcag aagtaaaaca cctccaccac
120ctcctcaaac agctcaaacc aagcgagaag tacctaaaaa taaagcacct
actgctgaaa 180agagagagag tggacctaag caagctgcag taaatgctgc
agttcagagg gtccaggttc 240ttccagatgc tgatacttta ttacattttg
ccacggaaag tactccagat ggattttctt 300gttcatccag cctgagtgct
ctgagcctcg atgagccatt tatacagaaa gatgtggaat 360taagaataat
gtgcatgtgt ctttatagca gcatgattta tactcatttg ggtatatacc
420cagtaatggg atggctgggt caaatggtat ttctagttct agatccctga
ggaatcgcca 480cactgacttc cacaatggtt gaactagttt acagtcccac
caagaaaatg tggcacatat 540acaccatgga atactatgca gccataaaaa
atgatgagtt catatccttt gtagggacat 600ggatgaaatt ggaaaccatc
attctcagta aactatcgca agaacaaaaa accaaacacc 660gcatattctc
acttataggt gggaattgaa caatgagatc acatggacac aggaagggga
720atatcacact ctggggactg tggtggggtc gggggagggg ggagggatag
cattgggaga 780tatacctaat gctagatgac acattagtgg gtgcagcgca
gcatggcaca tgtatacata 840tgtaactaac ctgcacaatg tgcacatgta
ccctaaaact tagagtataa taaaaaaaaa 900aaaaaaaaaa ataacaataa
atgagataaa atctaaaaaa aaaaaaaaaa aaaaaaaaaa 960aaaaaaaaaa
aaaaaaaaaa aaaaaaaaaa aaaaaaaaaa aaaaaaaaaa aaaaaaaaaa
1020aaaaaaaaaa aaaaaaaaaa aaaaaaaaaa aaaaaaaaaa aaaaaaaaaa
aaaaaaaaaa 1080aaaaaaaaaa aaaaaaaaaa aaaaaaaaaa aaaaaaaaaa
agaataatgc ctccagttca 1140ggaaaatgac aatgggaatg aaacagaatc
agagcagcct aaagaatcaa atgaaaacca 1200agagaaagag gcagaaaaaa
ctattgattc tgaaaaggac ctattagatg attcagatga 1260tgatgatatt
gaaatactag aagaatgtat tatttctgcc atgccaacaa agtcatcacg
1320taaagcaaaa aagccagccc agactgcttc aaaattacct ccacctgtgg
caaggaaacc 1380aagtcagctg cctgtgtaca aacttctacc atcacaaaac
aggttgcaac cccaaaagca 1440tgttagtttt acaccggggg atgatatgcc
acgggtgtat tgtgttgaag ggacacctat 1500aaacttttcc acagctacat
ctctaagtga tctaacaatc gaatcccctc caaatgagtt 1560ag 15622639DNAHomo
sapiens 26cagcatgtca agatcacaga ttttgggctg gccaaactg 392739DNAHomo
sapiens 27cagcatgtca agatcacaga ttttgggcgg gccaaactg 3928524DNAHomo
sapiens 28tgaccctgaa ttcggatgca gagcttcttc ccatgatgat ctgtccctca
cagcagggtc 60ttctctgttt cagggcatga actacttgga ggaccgtcgc ttggtgcacc
gcgacctggc 120agccaggaac gtactggtga aaacaccgca gcatgtcaag
atcacagatt ttgggctggc 180caaactgctg ggtgcggaag agaaagaata
ccatgcagaa ggaggcaaag taaggaggtg 240gctttaggtc agccagcatt
ttcctgacac cagggaccag gctgccttcc cactagctgt 300attgtttaac
acatgcaggg gaggatgctc tccagacatt ctgggtgagc tcgcagcagc
360tgctgctggc agctgggtcc agccagggtc tcctggtagt gtgagccaga
gctctgaggt 420ttcactctgg cctgctgggc tcctagcagc caccaaccca
tgatgctggg ccctgaaaac 480acacgcagac ctggatgagt gaggccactg
ggcacaacca gggc 52429413DNAHomo sapiens 29tgaccctgaa ttcggatgca
gagcttcttc ccatgatgat ctgtccctca cagcagggtc 60ttctctgttt cagggcatga
actacttgga ggaccgtcgc ttggtgcacc gcgacctggc 120agccaggaac
gtactggtga aaacaccgca gcatgtcaag atcacagatt ttgggcgggc
180caaactgctg ggtgcggaag agaaagaata ccatgcagaa ggaggcaaag
taaggaggtg 240gctttaggtc agccagcatt ttcctgacac cagggaccag
gctgccttcc cactagctgt 300attgtttaac acatgcaggg gaggatgctc
tccagacatt ctgggtgagc tcgcagcagc 360tgctgctggc agctgggtcc
agccagggtc tcctggtagt gtgagccaga gct 41330316DNAHomo sapiens
30tgtcgccctg gaccctggga caccgcctcc tgagattaaa gcgagagcca gggcgggccg
60ggccgagtag gcgcgagcta agcaggaggc ggaggcggag gcggagggcg aggggcgggg
120agcgccgcct ggagcgcggc aggtcatatt gaacattcca gatacctatc
attactcgat 180gctgttgata acagcaagat ggctttgaac tcagggtcac
caccagctat tggaccttac 240tatgaaaacc atggatacca accggaaaac
ccctatcccg cacagcccac tgtggtcccc 300actgtctacg aggtgc
31631364DNAHomo sapiens 31ccaaaagcaa gacaaatgac tcacagagaa
aaaagatggc agaaccaagg gcaactaaag 60ccgtcaggtt ctgaacagct ggtagatggg
ctggcttact gaaggacatg attcagactg 120tcccggaccc agcagctcat
atcaaggaag ccttatcagt tgtgagtgag gaccagtcgt 180tgtttgagtg
tgcctacgga acgccacacc tggctaagac agagatgacc gcgtcctcct
240ccagcgacta tggacagact tccaagatga gcccacgcgt ccctcagcag
gattggctgt 300ctcaaccccc agccagggtc accatcaaaa tggaatgtaa
ccctagccag gtgaatggct 360caag 36432360DNAArtificial
SequenceDescription of Artificial Sequence Synthetic construct
32tgtcgccctg gaccctggga caccgcctcc tgagattaaa gcgagagcca gggcgggccg
60ggccgagtag gcgcgagcta agcaggaggc ggaggcggag gcggagggcg aggggcgggg
120agcgccgcct ggagcgcggc aggaagcctt atcagttgtg agtgaggacc
agtcgttgtt 180tgagtgtgcc tacggaacgc cacacctggc taagacagag
atgaccgcgt cctcctccag 240cgactatgga cagacttcca agatgagccc
acgcgtccct cagcaggatt ggctgtctca 300acccccagcc agggtcacca
tcaaaatgga atgtaaccct agccaggtga atggctcaag 36033316DNAHomo sapiens
33tgtcgccctg gaccctggga caccgcctcc tgagattaaa gcgagagcca gggcgggccg
60ggccgagtag gcgcgagcta agcaggaggc ggaggcggag gcggagggcg aggggcgggg
120agcgccgcct ggagcgcggc aggtcatatt gaacattcca gatacctatc
attactcgat 180gctgttgata acagcaagat ggctttgaac tcagggtcac
caccagctat tggaccttac 240tatgaaaacc atggatacca accggaaaac
ccctatcccg cacagcccac tgtggtcccc 300actgtctacg aggtgc
31634355DNAHomo sapiens 34tgcgaagagc agcagcatgg atggatttta
tgaccagcaa gtgccttaca tggtcaccaa 60tagtcagcgt gggagaaatt gtaacgagaa
accaacaaat gtcaggaaaa gaaaattcat 120taacagagat ctggctcatg
attcagaaga actctttcaa gatctaagtc aattacagga 180aacatggctt
gcagaagctc aggtacctga caatgatgag cagtttgtac cagactatca
240ggctgaaagt ttggcttttc atggcctgcc actgaaaatc aagaaagaac
cccacagtcc 300atgttcagaa atcagctctg cctgcagtca agaacagccc
tttaaattca gctat 35535300DNAArtificial SequenceDescription of
Artificial Sequence Synthetic construct 35tgtcgccctg gaccctggga
caccgcctcc tgagattaaa gcgagagcca gggcgggccg 60ggccgagtag gcgcgagcta
agcaggaggc ggaggcggag gcggagggcg aggggcgggg 120agcgccgcct
ggagcgcggc agctcaggta cctgacaatg atgagcagtt tgtaccagac
180tatcaggctg aaagtttggc ttttcatggc ctgccactga aaatcaagaa
agaaccccac 240agtccatgtt cagaaatcag ctctgcctgc agtcaagaac
agccctttaa attcagctat 300
D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

D00013

D00014
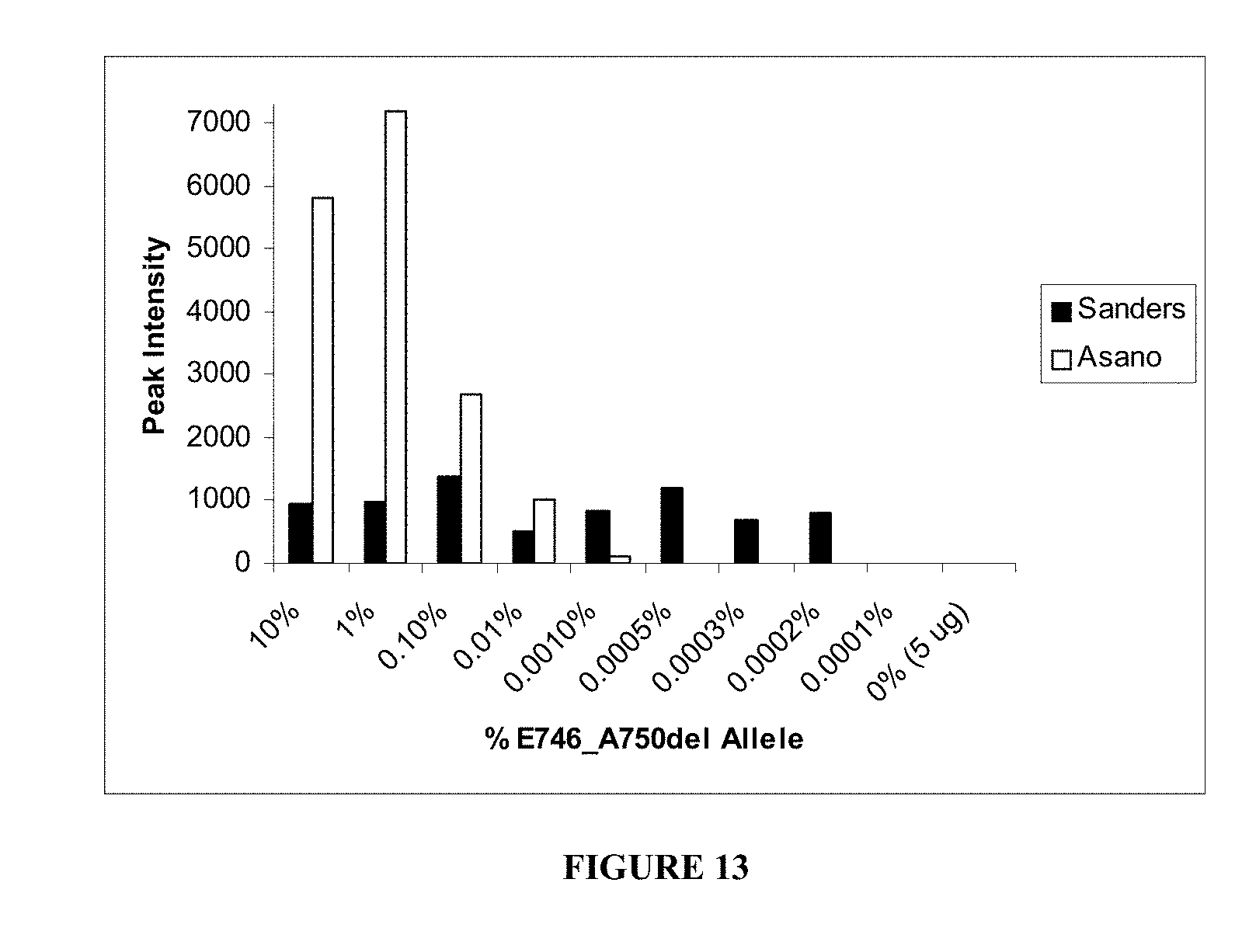
D00015
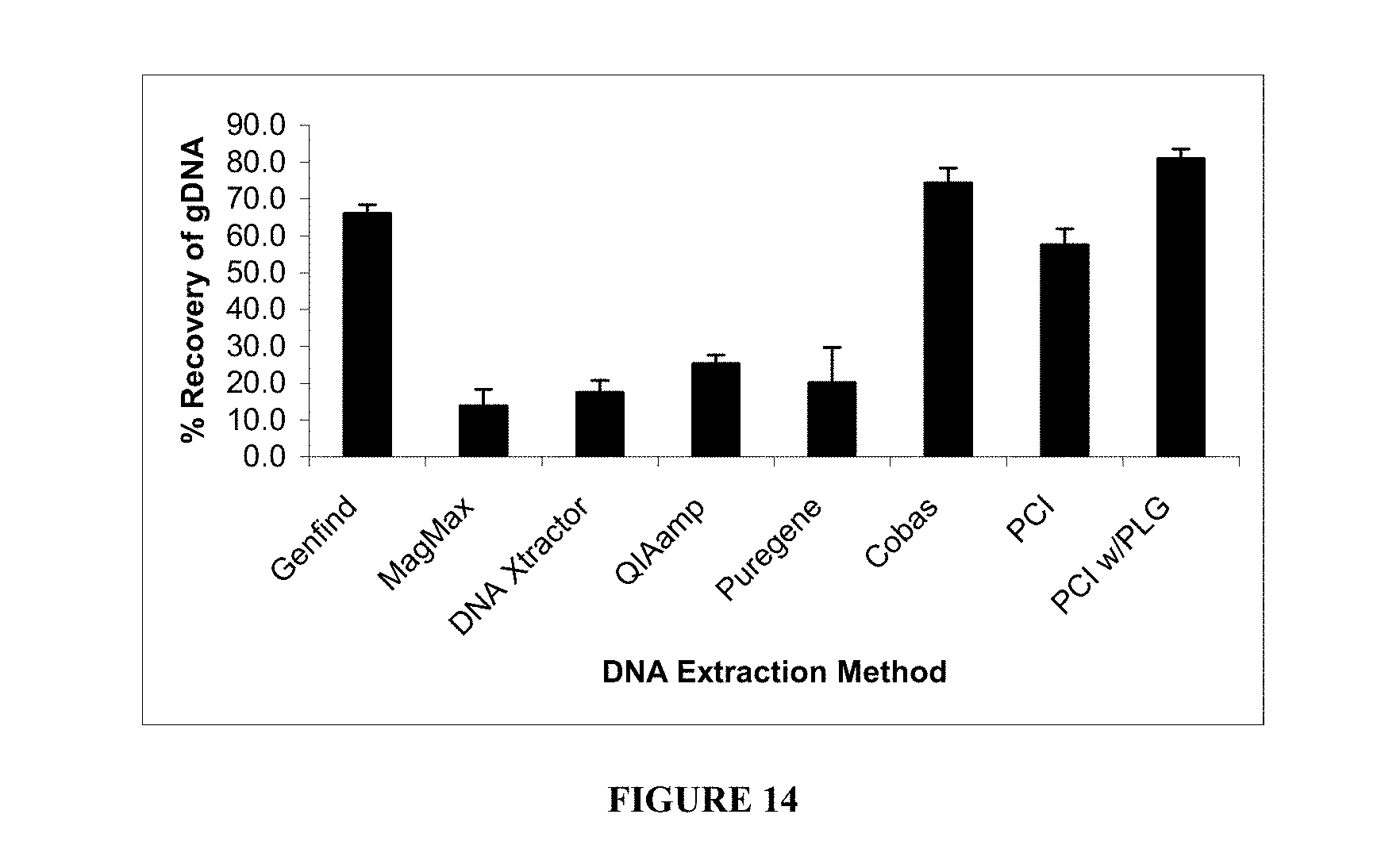
D00016

D00017
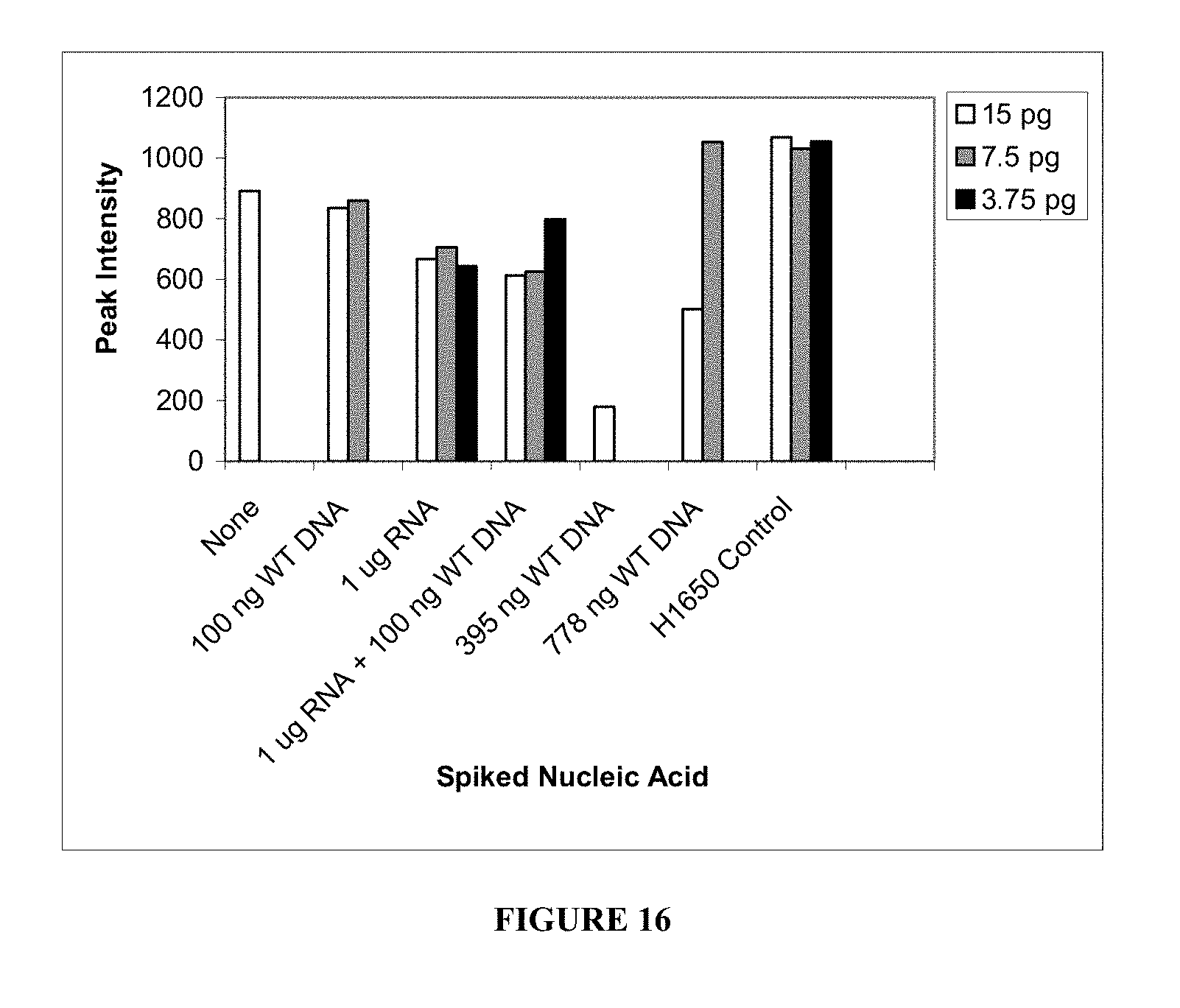
S00001
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.