Process For Controlling The Malodor "sweat", Using Bacterial Spores Capable Of Inhibiting Or Preventing The Production Of Such Malodor
Kandzia; Michael ; et al.
U.S. patent application number 16/084314 was filed with the patent office on 2019-03-28 for process for controlling the malodor "sweat", using bacterial spores capable of inhibiting or preventing the production of such malodor. This patent application is currently assigned to Henkel AG & Co. KGaA. The applicant listed for this patent is Henkel AG & Co. KGaA. Invention is credited to Michael Kandzia, Rainer Simmering, Mirko Weide.
| Application Number | 20190093049 16/084314 |
| Document ID | / |
| Family ID | 55527449 |
| Filed Date | 2019-03-28 |


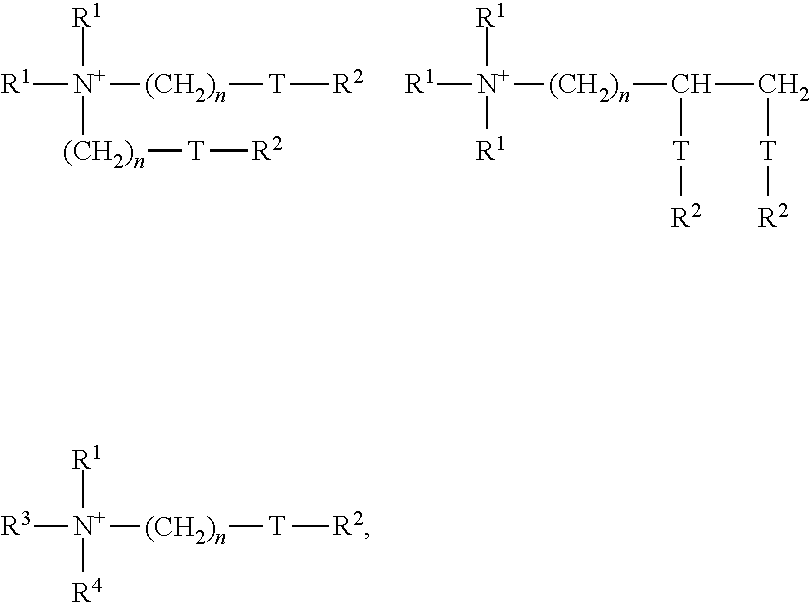
| United States Patent Application | 20190093049 |
| Kind Code | A1 |
| Kandzia; Michael ; et al. | March 28, 2019 |
PROCESS FOR CONTROLLING THE MALODOR "SWEAT", USING BACTERIAL SPORES CAPABLE OF INHIBITING OR PREVENTING THE PRODUCTION OF SUCH MALODOR
Abstract
The present disclosure generally relates to a method for degrading laundry malodor "sweat" preferably with regard to the treatment of hard and/or soft surfaces, and more particularly relates to the degradation of the laundry malodor "sweat" in the context of a textile treatment method using bacterial spores.
| Inventors: | Kandzia; Michael; (Solingen, DE) ; Simmering; Rainer; (Grevenbroich, DE) ; Weide; Mirko; (Duesseldorf, DE) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | Henkel AG & Co. KGaA Duesseldorf DE |
||||||||||
| Family ID: | 55527449 | ||||||||||
| Appl. No.: | 16/084314 | ||||||||||
| Filed: | March 10, 2017 | ||||||||||
| PCT Filed: | March 10, 2017 | ||||||||||
| PCT NO: | PCT/EP2017/055632 | ||||||||||
| 371 Date: | September 12, 2018 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C11D 3/381 20130101; C11D 11/0023 20130101; A61L 9/013 20130101; D06F 35/008 20130101; C11D 11/0017 20130101; C11D 3/0068 20130101 |
| International Class: | C11D 3/00 20060101 C11D003/00; C11D 3/38 20060101 C11D003/38; C11D 11/00 20060101 C11D011/00; D06F 35/00 20060101 D06F035/00; A61L 9/013 20060101 A61L009/013 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Mar 14, 2016 | EP | 16160054.9 |
Claims
1. A method of inhibiting or preventing the production of laundry malodor "sweat", comprising contacting a fabric or a laundry washing machine with bacterial spores of at least one species of Bacillus, which is selected from the group of Bacillus amyloliquefaciens, Bacillus tequilensis, Bacillus subtilis, Bacillus atrophaeus, Bacillus vallismortis, Bacillus mojavensis, and combinations thereof.
2. The method according to claim 1, wherein the method comprises contacting the fabric or the laundry washing machine with combinations of bacterial spores of one or more of the species of claim 1.
3. The method according to claim 1, wherein the laundry malodor is caused by Alphaproteobacteria.
4. A use of bacterial spores comprising using bacterial spores of at least one species of Bacillus selected from the group of Bacillus amyloliquefaciens, Bacillus tequilensis, Bacillus subtilis, Bacillus atrophaeus, Bacillus vallismortis, Bacillus mojavensis, and combinations thereof, for inhibiting or preventing the production of the laundry malodor "sweat".
5. A use of bacterial spores comprising using bacterial spores of at least one species of Bacillus, which is selected from the group of Bacillus amyloliquefaciens, Bacillus tequilensis, Bacillus subtilis, Bacillus atrophaeus, Bacillus vallismortis, Bacillus mojavensis, and combinations thereof, in washing or cleaning agents, fabric softeners, hygienic rinsers or post wash additives for inhibiting or preventing the production of the laundry malodor "sweat".
6. The method of claim 1 further comprising: combining the bacterial spores with a detergent prior to contacting the fabric or laundry washing machine with the bacterial spores.
7. The method of claim 1 further comprising: combining the bacterial spores with a fabric softener prior to contacting the fabric or laundry washing machine with the bacterial spores.
8. The method of claim 1 further comprising: combining the bacterial spores with a hygienic rinser prior to contacting the fabric or laundry washing machine with the bacterial spores.
9. The method of claim 1 further comprising: combining the bacterial spores with a post wash additive prior to contacting the fabric or laundry washing machine with the bacterial spores.
10. The use of claim 4 further comprising: using two or more of the spores of the species of Bacillus selected from the group of Bacillus amyloliquefaciens, Bacillus tequilensis, Bacillus subtilis, Bacillus atrophaeus, Bacillus vallismortis and/or Bacillus mojavensis.
11. The use of claim 4 further comprising: combining the spores with a detergent.
12. The use of claim 11 further comprising: washing laundry with the detergent and the spores.
13. The use of claim 5 further comprising: combining the bacterial spores with the washing or cleaning agents, fabric softeners, hygienic rinsers or post wash additives prior to inhibiting or preventing the production of the laundry malodor.
Description
CROSS-REFERENCE TO RELATED APPLICATION
[0001] This application is a U.S. National-Stage entry under 35 U.S.C. .sctn. 371 based on International Application No PCT/EP2017/055632, filed Mar. 10, 2017 which was published under PCT Article 21(2) and which claims priority to European Application No. 16160054.9, filed Mar. 14, 2016, which are all hereby incorporated in their entirety by reference.
TECHNICAL FIELD
[0002] The present disclosure generally relates to a method for degrading malodors preferably with regard to the treatment of hard and/or soft surfaces, and more particularly relates to the degradation of malodors in the context of a textile treatment method.
BACKGROUND
[0003] Malodor is a growing problem, particularly in laundry, with the changed habits of lower temperature washing, front load wash machines that save water but leave behind residual water between loads allowing bacterial biofilms to flourish, line drying clothes to save energy rather than appliance drying, and the increased popularity of manmade fabrics, such as athletic wear, that appear to retain odors more than natural fabrics.
[0004] An important consumer requirement, which also plays a role e.g. in the utilization of washing, cleaning, or care-providing agents, therefore includes in the elimination or at least diminution of malodors (i.e. off-odors) or undesired odors. Off-odors derive from specific olfactorily active compounds that are also referred to as "malodorants." Malodorants are foul-smelling compounds having so-called kakosmophoric groups, e.g. amine derivatives and sulfur derivatives. The presence of such off-odors generally results in a negative effect on human comfort, and for that reason the consumer makes an effort to extinguish these odors. Often, however, the off-odors are not extinguished but merely masked. It is usual to use for this purpose products that contain volatile, usually pleasant-smelling substances, and that even in small quantities can mask foul odors.
[0005] These solutions, however, are not completely effective as they are short-term. There is a need in the art for new solutions for controling the problem of malodor.
[0006] It is therefore the object of the present disclosure to provide the consumer with a further capability for bringing about an inhibition, degradation or prevention of malodors.
BRIEF SUMMARY
[0007] Methods and uses of bacterial spores for controlling malodor are provided. In an exemplary embodiment, a method includes inhibiting or preventing laundry malodor by contacting a fabric or a laundry washing machine with bacterial spores. The bacterial spores are at least one species of Bacillus, and are selected from the group of Bacillus amyloliquefaciens, Bacillus tequilensis, Bacillus subtilis, Bacillus atrophaeus, Bacillus vallismortis, Bacillus mojavensis, and combinations thereof.
[0008] A use of a bacterial spore is provided in another embodiment. The use includes inhibiting or preventing laundry malodor with at least one species of Bacillus. The at least one species of Bacillus is selected from the group of Bacillus amyloliquefaciens, Bacillus tequilensis, Bacillus subtilis, Bacillus atrophaeus, Bacillus vallismortis, Bacillus mojavensis, and combinations thereof.
[0009] A use of bacterial spores is provided in yet another embodiment. The use includes inhibiting or preventing laundry malodor with bacterial spores in washing or cleaning agents, fabric softeners, hygienic rinsers, or post wash additives. The bacterial spores are selected from the group of Bacillus amyloliquefaciens, Bacillus tequilensis, Bacillus subtilis, Bacillus atrophaeus, Bacillus vallismortis, Bacillus mojavensis, and combinations thereof.
DETAILED DESCRIPTION
[0010] The following detailed description is merely exemplary in nature and is not intended to limit the disclosure or the application and uses of the subject matter as described herein. Furthermore, there is no intention to be bound by any theory presented in the preceding background or the following detailed description.
[0011] The present disclosure provides a method of inhibiting or preventing the production of the laundry malodor "sweat", comprising contacting a fabric or a laundry washing machine with bacterial spores of at least one species of Bacillus, which is selected from the group including Bacillus amyloliquefaciens, Bacillus tequilensis, Bacillus subtilis, Bacillus atrophaeus, Bacillus vallismortis and/or Bacillus mojavensis.
[0012] The Bacillus species mentioned above or mixtures of those are commercially available as Freshen Herbal.RTM. and Drain Ease Open.RTM. from Novozymes A/S, Denmark, and UBFE Kultur.RTM. and WC Kultur.RTM. from Julius Hoesch GmbH & Co. KG, 52353 Duren-Hoven, Germany.
[0013] The contacting can occur before, during, or after the washing process. Fabrics preferably are contacted with the bacterial spores during the washing process; washing machines may alternatively or additionally be contacted with the bacterial spores in between two washing processes.
[0014] Combinations of bacterial spores of such species and/or isolates may also be used, such as blends of two or more species and/or isolates, three or more species and/or isolates, etc. Preferred are combinations comprising spores of Bacillus subtilis and Bacillus mojavensis. Also preferred are combinations comprising spores of Bacillus subtilis and Bacillus atrophaeus and/or Bacillus vallismortis.
[0015] The present disclosure also provides the use of said bacterial spores in inhibiting the malodor "sweat", in particular in washing or cleaning agents, fabric softeners, hygienic rinsers or post wash additives.
[0016] The methods and compositions of the present disclosure may be used to treat an existing odor problem and/or as a preventative treatment to prevent a potential odor problem. The present disclosure may be used, for example, to inhibit malodor in laundry washing machines/processes, dry cleaning machines/processes, steam cleaning machines/processes, carpet cleaning machines/processes, dish washing machines/processes, and other cleaning machines/processes.
[0017] Malodor may be generated from a number of sources, mostly microbial and in particular bacterial sources (including compounds derived or produced therefrom). Sources of malodor causing bacteria, include bacterium species selected from the group including of Bacillus amyloliquefaciens, Acinetobacter junii, Bacillus subtilis, Janibacter melois, Sphingobium ummariense, Sphingomonas panni, Sphingomonadaceae, Actinobacter tandoii, Junibacter melonis, Curtobacterium flaccumfaciens subsp. flaccumfaciens, Flavobacterium denitrificans, Staphylococcus epidermidis, Escherichia coli, Leclercia adecarboxylata, Enterobacter sp., Cronobacter sakazakii, Bacillus megaterium, Sphingobacterium faecium, Enterobacter cloacae, Pseudomonas veronii, Microbacterium luteolum, Morganella morganii, Bacillus cereus, Pseudomonas sp., Pseudomonas-marginalis, Citrobacter sp., Escherichia coli strain JCLys5, Roseomonas aquatic, Pseudomonas panipatensis, Brevibacillus subtilis subtilis, Micrococcus luteus, Bacillus pumilus, Ralstonia eutropha, Caulobacter fusiformis, Stenotrophomonas maltophilia, Rhodococcus opacus, Breviundimonas intermedia, Agrobacterium tumefaciens and in particular Alphaproteobacteria (a class of bacteria in the phylum Proteobacteria), and/or a combination thereof, and/or substances derived therefrom.
[0018] The methods and compositions may also be applied directly to an article treated (e.g., cleaned) in the cleaning machine or cleaning process, such as, to a laundry treated in the machine. The article may be treated before cleaning, during the cleaning process, after the cleaning processes and any combination thereof. Examples of such articles to be treated include laundry, carpets, and fabrics.
[0019] The term "fabrics" encompasses all kind of fabrics, textiles, fibers, clothes garments, and fabrics used on, e.g., furniture and cars. The term "laundry" refers to already used and/or stained/soiled clothes in need of washing, and is in contrast to newly manufactured fabrics. Washing laundry may be carried out in private households and in commercial and institutional facilities, such as, hospitals, prisons, uniform service companies. Washing of newly manufactured fabrics is mainly done in the textile industry. The fabric or laundry may be made from any suitable material. In preferred embodiments the fabrics and/or laundry are made from cellulosic materials, synthetic materials and/or man-made fibers, or blends thereof. Examples of contemplated cellulosic materials include cotton, viscose, rayon, ramie, linen, lyocell (e.g., TENCEL.TM., produced by Courtaulds Fibers), or blends thereof, or blends of any of these fibers together with synthetic or man-made fibers (e.g., polyester, polyamid, nylon) or other natural fibers such as wool and silk, such as viscose/cotton blends, lyocell/cotton blends, viscose/wool blends, lyocell/wool blends, cotton/wool blends; flax (linen), ramie and other fabrics and/or laundry based on cellulose fibers, including all blends of cellulosic fibers with other fibers such as wool, polyamide, acrylic and polyester fibers, e.g., viscose/cotton/polyester blends, wool/cotton/polyester blends, flax/cotton blends etc. The fabric and/or laundry may also be a synthetic materials, e.g., including essentially 100% polyester, polyamid, nylon, respectively. The term "wool," means any commercially useful animal hair product, for example, wool from sheep, camel, rabbit, goat, llama, and known as merino wool, Shetland wool, cashmere wool, alpaca wool, mohair etc. and includes wool fibers and animal hair. The method of the present disclosure can be used on wool or animal hair material in the form of top, fiber, yarn, or woven or knitted fabrics.
[0020] The treating may include contacting the odor-generating organism(s) or odor-generating compound(s) present in the cleaning machine or cleaning process with the bacterial spores. Such contacting may include contacting a surface of a machine with the bacterial spores and/or contacting a process water or cleaning composition used in the cleaning machine with the bacterial spores.
[0021] Contacting means contacting the odor-causing organism and/or odor causing compound with the bacterial spores.
[0022] The ability to prepare spores and vegetative cells is considered routine in the art. See Tzeng, Y. M., Y. K. Rao, et al. (2008). "Effect of cultivation conditions on spore production from Bacillus amyloliquefaciens B128 and its antagonism to Botrytis elliptica." Journal of Applied Microbiology 104(5): 1275-1282.
[0023] Compositions of the present disclosure comprise bacterial spores as described herein. The bacterial spores should be present in effective amounts. The terms "effective amount", "effective concentration" or "effective dosage" are defined herein as the amount, concentration or dosage of odor-controling bacterial spores that can inhibit the malodor caused by the odor causing organism or substances derived therefrom on articles, articles subjected to a cleaning machine or cleaning process, and/or cleaning machines. The actual effective dosage in absolute numbers depends on factors including: the odor causing organisms(s) in question; whether the aim is prevention or reduction of malodor; other ingredients present in the composition, and also the articles and/or cleaning machine in question.
[0024] In an embodiment an effective dosage of the bacterial spores as described herein would be introduced to the detergent at a final concentration of about 1.times.10.sup.2-1.times.10.sup.9 CFU/g of detergent, with a preferred range of about 1.times.10.sup.3-1.times.10.sup.7 CFU/g of detergent.
[0025] In another embodiment an effective dosage of the bacterial spores as described herein would be introduced to a fabric softener or hygienic rinser or at a final concentration of about 1.times.10.sup.1-1.times.10.sup.9 CFU/g of the fabric softener or hygienic rinser, with a preferred range of about 1.times.10.sup.2-1.times.10.sup.7 CFU/g of the fabric softener or hygienic rinser.
[0026] In yet another embodiment an effective dosage of the bacterial spores as described herein would be introduced to a post wash additive at a final concentration of about 1.times.10.sup.1-1.times.10.sup.9 CFU/g of the post wash additive, with a preferred range of about 1.times.10.sup.1-1.times.10.sup.6 CFU/g of the post wash additive.
[0027] Effective amounts can be determined by one skilled in the art using routine assays.
[0028] The bacterial spores of the present disclosure can be used in combination with or as an ingredient of a washing product, such as detergents and/or fabric softeners in particular, including but not limited to aerosols, powders, solids, creams, etc., for use, e.g., in cleaning machines, cleaning processes and/or articles treated in cleaning machines or cleaning processes, such as, fabrics.
[0029] The compositions of the present disclosure may in an embodiment have a pH in the range of from about 5 to about 10 and may further include water and/or one or more preservatives. For preservation of compositions comprising bacterial spores of Bacillus amyloliquefaciens, for example, the following preservatives can be useful: chloromethylisothiazolinone/methylisothiazolinone (CMIT/MIT) (Kathon.RTM. or others); MIT (Neolone.RTM. or others); 1,2-benzisothiazolin-3-one (BIT) (if allowed in personal care); CMIT/MIT+EDTA; CMIT/MIT+Biodegradable Chelator; MIT+EDTA; MIT+Biodegradable Chelator; BIT+EDTA; BIT+Biodegradable Chelator; Bronopol; 2-Phenoxyethanol; 2-Phenoxyethanol+Biodegradable Chelator; Potassium sorbate (used at low pH); Sodium benzoate (used at low pH); Salt; Glycerol; Propylene Glycol; Essential Oils; Dichlorobenzyl alcohol; Triclosan.RTM.; Parabens; and 1-Phenoxy-2-propanol and 2-Phenoxy-1-propanol. In an embodiment, the preservative is 2-Phenoxyethanol; 2-Phenoxyethanol+Biodegradable Chelator; Potassium Sorbate (used at low pH); Sodium Benzoate (used at low pH); Salt; Glycerol; Propylene Glycol; or one of more Essential Oils--e.g., white mustard seed, tea tree, rosewood, or some citrus oils. In another embodiment, the preservative is 2-Phenoxyethanol; 2-Phenoxyethanol+Biodegradable Chelator; or Glycerol. Accordingly, an embodiment of the present disclosure is directed to a composition comprising bacterial spores as described herein and a preservative selected from the group including chloromethylisothiazolinone/methylisothiazolinone (CMIT/MIT) (Kathon.RTM. or others); MIT (Neolone.RTM. or others); 1,2-benzisothiazolin-3-one (BIT) (if allowed in personal care); CMIT/MIT+EDTA; CMIT/MIT+Biodegradable Chelator; MIT+EDTA; MIT+Biodegradable Chelator; BIT+EDTA; BIT+Biodegradable Chelator; Bronopol; 2-Phenoxyethanol; 2-Phenoxyethanol+Biodegradable Chelator; Potassium sorbate (used at low pH); Sodium benzoate (used at low pH); Salt; Glycerol; Propylene Glycol; Essential Oils; Dichlorobenzyl alcohol; Triclosan.RTM.; Parabens; and 1-Phenoxy-2-propanol and 2-Phenoxy-1-propanol. In an embodiment, the preservative is 2-Phenoxyethanol; 2-Phenoxyethanol+Biodegradable Chelator; Potassium Sorbate (used at low pH); Sodium Benzoate (used at low pH); Salt; Glycerol; Propylene Glycol; or one of more Essential Oils--e.g., white mustard seed, tea tree, rosewood, or some citrus oils, 2-Phenoxyethanol; 2-Phenoxyethanol+Biodegradable Chelator; or Glycerol, and wherein the composition is a liquid, solid or gel composition.
[0030] A composition of the present disclosure may be in solid or liquid form. The composition may be a concentrate to be diluted, rehydrated and/or dissolved in a solvent, including water, before use. The composition may also be a ready-to-use (in-use) composition. The composition may furthermore be an active cleaning base ingredient to be incorporated into other cleaning or washing compositions.
[0031] In one embodiment, the composition is adapted for delivery to a washing machine to prevent fouling by bacterial species capable of causing the laundry malodor "sweat". In another embodiment, the composition is further adapted for delivery to a washing machine by applications which include, but are not limited to, solid, semi-solid, gel, liquid, aerosol, emulsion, and/or powder applications alone and/or in combination with liquid, solid, semisolid, aerosol, emulsion, and/or gel detergents, alone and/or in combination with liquid, solid, semi-solid, aerosol, emulsion, and/or gel fabric softeners, and/or alone and/or in combination with any other laundry and/or washing maching additive.
[0032] In one aspect, the present disclosure provides a composition adapted for application to a fabric. The composition adapted for delivery to a fabric may be in the form of a solid, semi-solid, gel, liquid, aerosol, emulsion, and/or powder, as a treatment for fabrics to prevent fouling by bacterial species capable of causing laundry malodor. In another embodiment, the composition is adapted for delivery to a fabric by applications which include, but are not limited to, solid, semi-solid, gel, liquid, aerosol, emulsion, and/or powder applications alone and/or in combination with liquid, solid, semi-solid, aerosol, emulsion, and/or gel detergents, alone and/or in combination with liquid, solid, semi-solid, aerosol, emulsion, and/or gel fabric softeners, and/or alone and/or in combination with any other laundry and/or washing maching additive.
[0033] When used in washing and cleaning agents for household applications, such as detergents, fabric softeners, fabric finishers, laundry performance enhancers, laundry care products, automatic and hand dishwashing products, toilet care products, hard surface cleaners such as cleaners for bathrooms, glass, floors and kitchens, the composition can furthermore contain other usual constituents of washing or cleaning agents, in particular textile washing agents, selected in particular from the group of builders, surfactants, polymers, enzymes, disintegration adjuvants, scents, and perfume carriers.
[0034] Included among the builders are in particular zeolites, silicates, carbonates, organic cobuilders, and--provided no environmental prejudices against their use exist--also phosphates.
[0035] The finely crystalline synthetic zeolite containing bound water that is preferably used is zeolite A and/or zeolite P. Zeolite MAP.RTM. (commercial product of the Crosfield Co.), for example, is appropriate as zeolite P. Also suitable, however, are zeolite X as well as mixtures of A, X, and/or P. Also commercially available and usable in the context of the present disclosure is, for example, a co-crystal of zeolite X and zeolite A (approx. 80 wt % zeolite X) that can be described by the formula
nNa.sub.2O.(1-n)K.sub.2O.Al.sub.2O.sub.3.(2-2.5)SiO.sub.2.(3.5-5.5)H.sub- .2O.
[0036] The zeolite can be used both as a builder in a granular compound and as a kind of "dusting" on a granular mixture, preferably a mixture to be compressed, both approaches to incorporation of the zeolite into the pre-mixture usually being used. Zeolites can exhibit an average particle size of less than about 10 .mu.m (volume distribution; measurement method: Coulter Counter), and preferably contain from about 18 wt % to about 22 wt %, in particular from, about 20 wt % to about 22 wt %, bound water.
[0037] Crystalline sheet silicates of the general formula NaMSi.sub.xO.sub.2x+1.y H.sub.2O can also be used, where M represents sodium or hydrogen, x is a number from about 1.9 to about 22, preferably from about 1.9 to about 4, particularly preferred values for x being 2, 3, or 4, and y denotes a number from about 0 to about 33, preferably from about 0 to about 20. The crystalline sheet silicates of the formula NaMSi.sub.xO.sub.2x+1.y H.sub.2O are marketed, for example, by Clariant GmbH (Germany) under the trade name Na-SKS. Examples of these silicates are Na-SKS-1 (Na.sub.2Si.sub.22O.sub.45.x H.sub.2O, kenyaite), Na-SKS-2 (Na.sub.2Si.sub.14O.sub.29.x H.sub.2O, magadiite), Na-SKS-3 (Na.sub.2Si.sub.8O.sub.17.x H.sub.2O), or Na-SKS-4 (Na.sub.2Si.sub.4O.sub.9.x H.sub.2O, makatite).
[0038] Crystalline sheet silicates of the formula NaMSi.sub.xO.sub.2x+1.y H.sub.2O in which x denotes 2 are preferred. Both .beta.- and .delta.-sodium disilicates Na.sub.2Si.sub.2O.sub.5.y H.sub.2O, as well as also principally Na-SKS-5 (.alpha.-Na.sub.2Si.sub.2O.sub.5), Na-SKS-7 (.beta.-Na.sub.2Si.sub.2O.sub.5, natrosilite), Na-SKS-9 (NaHSi.sub.2O.sub.5.H.sub.2O), Na-SKS-10 (NaHSi.sub.2O.sub.5.3 H.sub.2O, kanemite), Na-SKS-11 (t-Na.sub.2Si.sub.2O.sub.5), and Na-SKS-13 (NaHSi.sub.2O.sub.5), but in particular Na-SKS-6 (6-Na.sub.2Si.sub.2O.sub.5), are particularly preferred. Washing or cleaning agents preferably contain a weight proportion of the crystalline sheet silicates of the formula NaMSi.sub.xO.sub.2x+1. yH.sub.2O from about 0.1 wt % to about 20 wt %, preferably from about 0.2 wt % to about 15 wt %, and in particular from about 0.4 wt % to about 10 wt %.
[0039] Also usable are amorphous sodium silicates having a Na.sub.2O:SiO.sub.2 modulus from about 1:2 to about 1:3.3, preferably from about 1:2 to about 1:2.8, and in particular from about 1:2 to about 1:2.6, which are preferably dissolution-delayed and exhibit secondary washing properties. The dissolution delay as compared with conventional amorphous sodium silicates can have been brought about in various ways, for example by surface treatment, compounding, compacting/densification, or overdrying. The term "amorphous" is understood to mean that in X-ray diffraction experiments the silicates do not yield the sharp X-ray reflections that are typical of crystalline substances, but produce at most one or more maxima in the scattered X radiation that have a width of several degree units of the diffraction angle.
[0040] Alternatively or in combination with the aforesaid amorphous sodium silicates, it is possible to use X-amorphous silicates whose silicate particles yield blurred or even sharp diffraction maxima in electron beam diffraction experiments. This is to be interpreted to mean that the products comprise microcrystalline regions from about 10 to several hundred nm in size, values of up to a maximum of about 50 nm, and in particular up to a maximum of about 20 nm, being preferred. X-amorphous silicates of this kind likewise exhibit a dissolution delay as compared with conventional water glasses. Densified/compacted amorphous silicates, compounded amorphous silicates, and overdried X-amorphous silicates are particularly preferred.
[0041] This/these silicate(s), preferably alkali silicates, particularly preferably crystalline or amorphous alkali disilicates, if present, are contained in washing and cleaning agents in quantities from about 3 wt % to about 60 wt %, preferably from about 8 wt % to about 50 wt %, and in particular from about 20 wt % to about 40 wt %.
[0042] Utilization of the commonly known phosphates as builder substances is also possible, provided such use is not to be avoided for environmental reasons. Among the plurality of commercially obtainable phosphates, the alkali-metal phosphates have the greatest significance in the washing- and cleaning-agent industry, with particular preference for pentasodium resp. pentapotassium triphosphate (sodium resp. potassium tripolyphosphate).
[0043] "Alkali-metal phosphates" is the summary designation for the alkali-metal (in particular sodium and potassium) salts of the various phosphoric acids, in which context a distinction can be made between metaphosphoric acids (HPO.sub.3)n and orthophosphoric acid H.sub.3PO.sub.4, in addition to higher-molecular-weight representatives. The phosphates embody a combination of advantages: they act as alkali carriers, prevent lime deposits on machine parts resp. lime incrustations in fabrics, and furthermore contribute to cleaning performance. Phosphates that are technically especially important are pentasodium triphosphate Na.sub.5P.sub.3O.sub.10 (sodium tripolyphosphate) and the corresponding potassium salt pentapotassium triphosphate K.sub.5P.sub.3O.sub.10 (potassium tripolyphosphate). Sodium potassium tripolyphosphates are also used with preference. If phosphates are employed in washing or cleaning agents, preferred agents then contain that/those phosphate(s), preferably alkali metal phosphate(s), particularly preferably pentasodium resp. pentapotassium triphosphate (sodium resp. potassium tripolyphosphate), in quantities from about 5 wt % to about 80 wt %, preferably from about 15 wt % to about 75 wt %, and in particular from about 20 wt % to about 70 wt %.
[0044] Alkali carriers are also usable. Alkali carriers are considered to be, for example, alkali-metal hydroxides, alkali-metal carbonates, alkali-metal hydrogen carbonates, alkali-metal sesquicarbonates, the aforesaid alkali silicates, alkali metasilicates, and mixtures of the aforesaid substances; the alkali carbonates, in particular sodium carbonate, sodium hydrogen carbonate, or sodium sesquicarbonate, are preferably used. A builder system containing a mixture of tripolyphosphate and sodium carbonate can be particularly preferred. Because of their low chemical compatibility with the other ingredients of washing or cleaning agents as compared with other builder substances, the alkali-metal hydroxides are preferably used only in small quantities, preferably in quantities below about 10 wt %, preferably below about 6 wt %, particularly preferably below about 4 wt %, and in particular below about 2 wt %. Agents that contain, based on their total weight, less than about 0.5 wt % and in particular no alkali-metal hydroxides are particularly preferred. It is preferred to use carbonate(s) and/or hydrogen carbonate(s), preferably alkali carbonate(s), particularly preferably sodium carbonate, in quantities from about 2 wt % to about 50 wt %, preferably from about 5 wt % to about 40 wt %, and in particular from about 7.5 wt % to about 30 wt %.
[0045] Organic builders that are to be recited are in particular polycarboxylates/polycarboxylic acids, polymeric polycarboxylates, aspartic acid, polyacetals, dextrins, as well as phosphonates. Polycarboxylic acids are usable, for example, in the form of the free acid and/or sodium salts thereof, "polycarboxylic acids" being understood as those carboxylic acids which carry more than one acid function. These are, for example, citric acid, adipic acid, succinic acid, glutaric acid, malic acid, tartaric acid, maleic acid, fumaric acid, sugar acids, aminocarboxylic acids, nitrilotriacetic acid (NTA), provided such use is not objectionable for environmental reasons, as well as mixtures thereof. The free acids typically also possess, besides their builder effect, the property of an acidifying component, and thus also serve to establish a lower and milder pH for washing or cleaning agents. To be recited in this context are, in particular, citric acid, succinic acid, glutaric acid, adipic acid, gluconic acid, and any mixtures thereof. Also suitable as builders are polymeric polycarboxylates; these are, for example, the alkali metal salts of polyacrylic acid or of polymethacrylic acid, for example those having a relative molecular weight from about 500 to about 70,000 g/mol. Polyacrylates that preferably have a molecular weight from about 2000 to about 20,000 g/mol are particularly suitable. Of this group in turn, the short-chain polyacrylates, which have molar masses from about 2000 to about 10,000 g/mol and particularly preferably from about 3000 to about 5000 g/mol, can be preferred because of their superior solubility. Also suitable are copolymeric polycarboxylates, in particular those of acrylic acid with methacrylic acid and of acrylic acid or methacrylic acid with maleic acid. Copolymers of acrylic acid with maleic acid that contain from about 50 wt % to about 90 wt % acrylic acid and from about 50 wt % to about 10 wt % maleic acid have proven particularly suitable. Their relative molecular weight, based on free acids, is generally from about 2000 g/mol to about 70,000 g/mol, preferably from about 20,000 g/mol to about 50,000 g/mol, and in particular from about 30,000 gmol to about 40,000 g/mol. To improve water solubility, the polymers can also contain allylsulfonic acids, for example allyloxybenzenesulfonic acid and methallylsulfonic acid, as monomers. The (co)polymeric polycarboxylates can be employed as a solid or in aqueous solution. The concentration of (co)polymeric polycarboxylates in washing or cleaning agents is preferably from about 0.5 wt % to about 20 wt %, and in particular from about 3 wt % to about 10 wt %.
[0046] Also particularly preferred are biodegradable polymers made up of more than two different monomer units, for example those that contain as monomers salts of acrylic acid and of maleic acid as well as vinyl alcohol resp. vinyl alcohol derivatives, or that contain as monomers salts of acrylic acid and of 2-alkylallylsulfonic acid, as well as sugar derivatives. Further preferred copolymers are those that comprise acrolein and acrylic acid/acrylic acid salts, resp. acrolein and vinyl acetate, as monomers. Also to be mentioned as further preferred builder substances are polymeric aminodicarboxylic acids, salts thereof, or precursor substances thereof. Polyaspartic acids and/or salts thereof are particularly preferred.
[0047] A further substance class having builder properties is represented by phosphonates. These are the salts of, in particular, hydroxyalkane- or aminoalkanephosphonic acids. Among the hydroxyalkanephosphonic acids, 1-hydroxyethane-1,1-diphosphonate (HEDP) is of particular importance. It is employed in particular as a sodium salt, the disodium salt reacting neutrally and the tetrasodium salt in alkaline fashion. Suitable aminoalkanephosphonic acids are, in particular, ethylenediaminetetramethylenephosphonic acid (EDTMP), diethylenetriaminepentamethylenephosphonic acid (DTPMP), and their higher homologs. They are used in particular in the form of the neutrally reacting sodium salts, e.g. as the hexasodium salt of EDTMP or as the hepta- and octasodium salt of DTPMP. Mixtures of the aforesaid phosphonates can also be used as organic builders. Aminoalkanephosphonates in particular moreover possess a pronounced heavy-metal binding capability.
[0048] Further suitable builder substances are polyacetals, which can be obtained by reacting dialdehydes with polyolcarboxylic acids that comprise from about 5 to about 7 carbon atoms and at least three hydroxyl groups. Preferred polyacetals are obtained from dialdehydes such as glyoxal, glutaraldehyde, terephthalaldehyde and mixtures thereof, and from polyolcarboxylic acids such as gluconic acid and/or glucoheptonic acid.
[0049] Further suitable organic builder substances are dextrins, for example oligomers resp. polymers of carbohydrates, which can be obtained by partial hydrolysis of starches. Hydrolysis can be carried out in accordance with usual, e.g. acid- or enzyme-catalyzed, methods. These are preferably hydrolysis products having average molar weights in the range from about 400 g/mol to about 500,000 g/mol. A polysaccharide having a dextrose equivalent (DE) in the range from about 0.5 to about 40, in particular from about 2 to about 30, is preferred, DE being a common indicator of the reducing effect of a polysaccharide as compared with dextrose, which possesses a DE of about 100. Both maltodextrins having a DE between from about 3 and about 20 and dry glucose syrups having a DE between from about 20 and about 37, as well as so-called yellow dextrins and white dextrins having higher molar weights in the range from about 2000 to about 30,000 g/mol, are usable. The oxidized derivatives of such dextrins are their reaction products with oxidizing agents that are capable of oxidizing at least one alcohol function of the saccharide ring to the carboxylic acid function.
[0050] Oxydisuccinates and other derivatives of disuccinates, preferably ethylenediamine disuccinate, are additional suitable cobuilders. Ethylenediamine-N,N'-disuccinate (EDDS) is used here, preferably in the form of its sodium or magnesium salts. Also preferred in this context are glycerol disuccinates and glycerol trisuccinates. If desired, suitable utilization quantities in particular in zeolite-containing and/or silicate-containing formulations are from about 3 wt % to about 15 wt %.
[0051] Other usable organic cobuilders are, for example, acetylated hydroxycarboxylic acids resp. salts thereof, which can optionally also be present in lactone form and which contain at least about 4 carbon atoms and at least one hydroxy group, as well as a maximum of two acid groups.
[0052] All compounds that are capable of forming complexes with alkaline earth ions can also be used as builders.
[0053] Washing and cleaning agents can contain nonionic, anionic, cationic, and/or amphoteric surfactants.
[0054] All nonionic surfactants known to one skilled in the art can be used as nonionic surfactants. With particular preference, washing or cleaning agents contain nonionic surfactants from the group of the alkoxylated alcohols. The nonionic surfactants used are preferably alkoxylated, advantageously ethoxylated, in particular primary alcohols having preferably from about 8 to about 18 carbon atoms and an average of from about 1 to about 12 mol ethylene oxide (EO) per mol of alcohol, in which the alcohol residue can be linear or preferably methyl-branched in the 2-position, resp. can contain mixed linear and methyl-branched residues, such as those that are usually present in oxo alcohol residues. Particularly preferred, however, are alcohol ethoxylates having linear residues made up of alcohols of natural origin having from about 12 to about 18 carbon atoms, e.g. from coconut, palm, tallow, or oleyl alcohol, and an average of from about 2 to about 8 EO per mol of alcohol. The preferred ethoxylated alcohols include, for example, C.sub.12-14 alcohols with 3 EO or 4 EO, C.sub.9-11 alcohols with 7 EO, C.sub.13-15 alcohols with 3 EO, 5 EO, 7 EO, or 8 EO, C.sub.12-18 alcohols with 3 EO, 5 EO, or 7 EO, and mixtures thereof, such as mixtures of C.sub.12-14 alcohol with 3 EO and C.sub.12-18 alcohol with 5 EO. The degrees of ethoxylation indicated represent statistical averages that can correspond to an integral or a fractional number for a specific product. Preferred alcohol ethoxylates exhibit a restricted distribution of homologs (narrow range ethoxylates, NRE).
[0055] Alternatively or in addition to these nonionic surfactants, fatty alcohols with more than about 12 EO can also be used. Examples of these are tallow fatty alcohol with 14 EO, 25 EO, 30 EO, or 40 EO. Also usable as further nonionic surfactants are alkylglycosides of the general formula RO(G).sub.x in which R corresponds to a primary straight-chain or methyl-branched aliphatic residue, in particular methyl-branched in the 2-position, having from about 8 to about 22, preferably from about 12 to about 18 carbon atoms, and G is the symbol that denotes a glycose unit having 5 or 6 carbon atoms, preferably glucose. The degree of oligomerization x, which indicates the distribution of monoglycosides and oligoglycosides, is any number between about 1 and about 10; x is preferably from about 1.2 to about 1.4.
[0056] A further class of nonionic surfactants used in preferred fashion, which are used either as the only nonionic surfactant or in combination with other nonionic surfactants, are alkoxylated, preferably ethoxylated or ethoxylated and propoxylated, fatty acid alkyl esters, preferably having 1 to 4 carbon atoms in the alkyl chain.
[0057] Nonionic surfactants of the amine oxide type, for example N-cocalkyl-N,N-dimethylamine oxide and N-tallowalkyl-N,N-dihydroxyethylamine oxide, and the fatty acid alkanolamides, can also be used. The quantity of these nonionic surfactants is preferably equal to no more than that of the ethoxylated fatty alcohols, in particular no more than half thereof.
[0058] Further suitable surfactants are polyhydroxy fatty acid amides of the formula
##STR00001##
in which R denotes an aliphatic acyl residue having from about 6 to about 22 carbon atoms; R.sup.1 denotes hydrogen, an alkyl or hydroxyalkyl residue having 1 to 4 carbon atoms; and [Z] denotes a linear or branched polyhydroxyalkyl residue having from about 3 to about 10 carbon atoms and from about 3 to about 10 hydroxyl groups. Polyhydroxy fatty acid amides are known substances that can usually be obtained by reductive amination of a reducing sugar with ammonia, an alkylamine, or an alkanolamine, and subsequent acylation with a fatty acid, a fatty acid alkyl ester, or a fatty acid chloride. Also belonging to the group of the polyhydroxy fatty acid amides are compounds of the formula
##STR00002##
in which R denotes a linear or branched alkyl or alkenyl residue having from about 7 to about 12 carbon atoms; R.sup.1 denotes a linear, branched, or cyclic alkyl residue or an aryl residue having from about 2 to about 8 carbon atoms; and R.sup.2 denotes a linear, branched, or cyclic alkyl residue or an aryl residue or an oxyalkyl residue having from about 1 to about 8 carbon atoms, C.sub.1-4 alkyl or phenyl residues being preferred; and [Z] denotes a linear polyhydroxyalkyl residue whose alkyl chain is substituted with at least two hydroxyl groups, or alkoxylated, preferably ethoxylated or propoxylated, derivatives of that residue. [Z] is preferably obtained by reductive amination of a reduced sugar, for example glucose, fructose, maltose, lactose, galactose, mannose, or xylose. The N-alkoxy- or N-aryloxy-substituted compounds can be converted into the desired polyhydroxy fatty acid amides by reaction with fatty acid methyl esters in the presence of an alkoxide as catalyst.
[0059] Nonionic surfactants from the group of alkoxylated alcohols, particularly preferably from the group of mixed alkoxylated alcohols and in particular from the group of EO/AO/EO nonionic surfactants or PO/AO/PO nonionic surfactants, especially PO/EO/PO nonionic surfactants, are particularly preferred. These PO/EO/PO nonionic surfactants are notable for good foam control.
[0060] Anionic surfactants used are, for example, those of the sulfonate and sulfate types. Possibilities as surfactants of the sulfonate type are, for example, preferably C.sub.9-13 alkylbenzenesulfonates, olefinsulfonates, i.e. mixtures of alkene- and hydroxyalkanesulfonates, and disulfonates, for example such as those obtained from C.sub.12-18 monoolefins having a terminal or internal double bond, by sulfonation with gaseous sulfur trioxide and subsequent alkaline or acid hydrolysis of the sulfonation products. Also suitable are alkanesulfonates that are obtained from C.sub.12-18 alkanes, for example by sulfochlorination or sulfoxidation with subsequent hydrolysis resp. neutralization. Also suitable are the esters of .alpha.-sulfo fatty acids (estersulfonates), for example the .alpha.-sulfonated methyl esters of hydrogenated coconut, palm kernel, or tallow fatty acids.
[0061] Further suitable anionic surfactants are sulfonated fatty acid glycerol esters. "Fatty acid glycerol esters" are to be understood as the mono-, di- and triesters, and mixtures thereof, that are obtained in the context of manufacture by esterification of a monoglycerol with from about 1 to about 3 mol fatty acid, or upon transesterification of triglycerides with from about 0.3 to about 2 mol glycerol. Preferred sulfonated fatty acid glycerol esters are the sulfonation products of saturated fatty acids having from about 6 to about 22 carbon atoms, for example hexanoic acid, octanoic acid, decanoic acid, myristic acid, lauric acid, palmitic acid, stearic acid, or behenic acid.
[0062] Preferred alk(en)yl sulfates are the alkali, and in particular sodium salts of the sulfuric acid semi-esters of C.sub.12-18 fatty alcohols, for example from coconut fatty alcohol, tallow fatty alcohol, lauryl, myristyl, cetyl, or stearyl alcohol, or C.sub.10 to C.sub.20 oxo alcohols, and those semi-esters of secondary alcohols of those chain lengths. Also preferred are alk(en)yl sulfates of the aforesaid chain length that contain a synthetic straight-chain alkyl residue produced on a petrochemical basis, which possess a breakdown behavior analogous to those appropriate compounds based on fat-chemistry raw materials. For purposes of washing technology, the C.sub.12 to C.sub.16 alkyl sulfates and C.sub.12 to C.sub.15 alkyl sulfates, as well as C.sub.14 to C.sub.15 alkyl sulfates, are preferred. 2,3-Alkyl sulfates that can be obtained, for example, as commercial products of the Shell Oil Company under the name DAN.RTM., are also suitable anionic surfactants.
[0063] Sulfuric acid monoesters of straight-chain or branched C.sub.7-21 alcohols ethoxylated with from about 1 to about 6 mol ethylene oxide, such as 2-methyl-branched C.sub.9-11 alcohols with an average of 3.5 mol ethylene oxide (EO) or C.sub.12-18 fatty alcohols with from about 1 to about 4 EO, are also suitable. Because of their high-foaming behavior they are used in cleaning agents only in relatively small quantities, for example in quantities from about 1 wt % to about 5 wt %.
[0064] Other suitable anionic surfactants are also the salts of alkylsulfosuccinic acid, which are also referred to as sulfosuccinates or as sulfosuccinic acid esters and represent the monoesters and/or diesters of sulfosuccinic acid with alcohols, preferably fatty alcohols, and in particular ethoxylated fatty alcohols. Preferred sulfosuccinates contain C.sub.8-18 fatty alcohol residues or mixtures thereof. Particularly preferred sulfosuccinates contain a fatty alcohol residue that derives from ethoxylated fatty alcohols that, considered per se, represent nonionic surfactants. Sulfosuccinates whose fatty alcohol residues derive from ethoxylated fatty alcohols having a restricted homolog distribution are, in turn, particularly preferred. It is likewise also possible to use alk(en)ylsuccinic acid having preferably from about 8 to about 18 carbon atoms in the alk(en)yl chain, or salts thereof.
[0065] Soaps are particularly appropriate as further anionic surfactants. Saturated fatty acid soaps, such as salts of lauric acid, myristic acid, palmitic acid, stearic acid, hydrogenated erucic acid and behenic acid, are suitable, as are soap mixtures derived in particular from natural fatty acids, e.g. coconut, palm-kernel, or tallow fatty acids.
[0066] The anionic surfactants, including soaps, can be present in the form of their sodium, potassium, or ammonium salts and as soluble salts of organic bases such as mono-, di-, or triethanolamine. The anionic surfactants are preferably present in the form of their sodium or potassium salts, in particular in the form of sodium salts.
[0067] Instead of or in combination with the aforesaid surfactants, cationic and/or amphoteric surfactants can also be used.
[0068] Cationic active substances that can be used are, for example, cationic compounds of the following formulas:
##STR00003##
in which each group R.sup.1 is selected mutually independently from C.sub.1-6 alkyl, alkenyl, or hydroxyalkyl groups; each group R.sup.2 is selected mutually independently from C.sub.8-28 alkyl or alkenyl groups; R.sup.3=R.sup.1 or (CH.sub.2).sub.n-T-R.sup.2; R.sup.4=R.sup.1 or R.sup.2 or (CH.sub.2).sub.n-T-R.sup.2; T=--CH.sub.2--, --O--CO--, or --CO--O--, and n is an integer from 0 to 5.
[0069] Textile-softening compounds can be used for textile care and in order to improve textile properties, such as a softer "hand" (avivage) and decreased electrostatic charge (increased wearing comfort). The active agents in these formulations are quaternary ammonium compounds having two hydrophobic residues, for example distearyldimethylammonium chloride, although because of its insufficient biodegradability the latter is increasingly being replaced by quaternary ammonium compounds that contain ester groups in their hydrophobic residues as defined break points for biodegradation.
[0070] "Esterquats" of this kind having improved biodegradability are obtainable, for example, by esterifying mixtures of methyl diethanolamine and/or triethanolamine with fatty acids and then quaternizing the reaction products in known fashion with alkylating agents. Dimethylolethylene urea is additionally suitable as a finish.
[0071] Enzymes can be used to increase the performance of washing or cleaning agents. These include in particular proteases, amylases, lipases, hemicellulases, cellulases, perhydrolases, or oxidoreductases, as well as preferably mixtures thereof. These enzymes are in principle of natural origin; proceeding from the natural molecules, improved variants are available for use in washing or cleaning agents and are used in correspondingly preferred fashion. Washing or cleaning agents contain enzymes preferably in total quantities from about 1.times.10.sup.-6 to about 5 wt %, based on active protein. The protein concentration can be determined with the aid of known methods, for example the BCA method or the biuret method.
[0072] Among the proteases, those of the subtilisin type are preferred. Examples thereof are subtilisins BPN' and Carlsberg and further developed forms thereof, protease PB92, subtilisins 147 and 309, the alkaline protease from Bacillus lentus, subtilisin DY, and the enzymes (to be classified, however, as subtilases and no longer as subtilisins in the strict sense) thermitase, proteinase K, and proteases TW3 and TW7.
[0073] Examples of usable amylases are the .alpha.-amylases from Bacillus licheniformis, from B. amyloliquefaciens, from B. stearothermophilus, from Aspergillus niger and A. oryzae, and the further developments of the aforementioned amylases improved for use in washing and cleaning agents. Additionally to be highlighted for this purpose are the .alpha.-amylase from Bacillus sp. A 7-7 (DSM 12368) and the cyclodextrin-glucanotransferase (CGTase) from B. agaradherens (DSM 9948).
[0074] Lipases or cutinases are usable because of their triglyceride-cleaving activity. Included thereamong are, for example, the lipases obtainable originally from Humicola lanuginosa (Thermomyces lanuginosus) or lipases further developed therefrom, in particular those having the D96L amino acid exchange. Also usable, for example, are the cutinases that were originally isolated from Fusarium solani pisi and Humicola insolens. Lipases and/or cutinases whose starting enzymes were originally isolated from Pseudomonas mendocina and Fusarium solanii are furthermore usable.
[0075] Enzymes that are grouped under the term "hemicellulases" can also be used. These include, for example, mannanases, xanthanlyases, pectinlyases (=pectinases), pectinesterases, pectatelyases, xyloglucanases (=xylanases), pullulanases, and .beta.-glucanases.
[0076] Oxidoreductases, for example oxidases, oxygenases, catalases, peroxidases such as halo-, chloro-, bromo-, lignin, glucose, or manganese peroxidases, dioxygenases, or laccases (phenoloxidases, polyphenoloxidases), can be used if desired to intensify the bleaching effect. Advantageously, preferably organic, particularly preferably aromatic compounds that interact with the enzymes are additionally added in order to enhance the activity of the relevant oxidoreductases (enhancers) or, if there is a large difference in redox potential between the oxidizing enzymes and the stains, to ensure electron flow (mediators).
[0077] Enzymes can be used in any form established according to the existing art. This includes, for example, the solid preparations obtained by granulation, extrusion, or lyophilization or, in particular in the case of liquid or gelled agents, solutions of the enzymes, advantageously as concentrated as possible, low in water and/or with added stabilizers. Alternatively, the enzymes can be encapsulated for both the solid and the liquid administration form, for example by spray drying or extrusion of the enzyme solution together with a preferably natural polymer, or in the form of capsules, for example those in which the enzymes are enclosed e.g. in a solidified gel, or in those of the core-shell type, in which an enzyme-containing core is coated with a water-, air-, and/or chemical-impermeable protective layer. Further active agents, for example stabilizers, emulsifiers, pigments, bleaches, or dyes, can additionally be applied in superimposed layers. Such capsules are applied using methods known per se, for example by vibratory or roll granulation or in fluidized bed processes. Advantageously, such granulates are low in dust, for example as a result of the application of polymeric film-formers, and are shelf-stable because of the coating. It is furthermore possible to package two or more enzymes together, so that a single granulate exhibits multiple enzyme activities.
[0078] One or more enzymes and/or enzyme preparations, preferably protease preparations and/or amylase preparations, are preferably used, in quantities from about 0.1 wt % to about 5 wt %, preferably from about 0.2 wt % to about 4.5 wt %, and in particular from about 0.4 wt % to about 4 wt %.
[0079] Individual fragrance compounds, e.g. synthetic products of the ester, ether, aldehyde, ketone, alcohol, and hydrocarbon types, can be used as perfume oils resp. scents. It is preferred, however, to use mixtures of different fragrances that together generate an attractive scent note. Such perfume oils can also contain natural fragrance mixtures such as those accessible from plant sources, for example pine, citrus, jasmine, patchouli, rose, or ylang-ylang oil. In order to be perceptible, a fragrance must be volatile; in addition to the nature of the functional groups and the structure of the chemical compound, the molecular weight also plays an important part. Most fragrances, for example, possess molar weights of up to approximately 200 g/mol, while molar weights of about 300 g/mol and above represent something of an exception. Because of the differing volatility of fragrances, the odor of a perfume or fragrance made up of multiple fragrances changes during volatilization, the odor impressions being subdivided into a "top note," "middle note" or "body," and "end note" or "dry out." Because the perception of an odor also depends a great deal on the odor intensity, the top note of a perfume or scent is not made up only of highly volatile compounds, while the end note comprises for the most part less-volatile, i.e. adherent fragrances. In the compounding of perfumes, more-volatile fragrances can, for example, be bound to specific fixatives, thereby preventing them from volatilizing too quickly. The division below of fragrances into "more-volatile" and "adherent" fragrances therefore makes no statement with regard to the odor impression, or as to whether the corresponding fragrance is perceived as a top or middle note. The scents can be processed directly, but it can also be advantageous to apply the scents onto carriers that ensure a slower scent release for a lasting scent. Cyclodextrins, for example, have proven successful as such carrier materials; the cyclodextrin-perfume complexes can additionally be coated with further adjuvants.
[0080] In selecting the coloring agent, care must be taken that the coloring agents exhibit excellent shelf stability and insensitivity to light, and they cannot have too strong an affinity with respect to textile surfaces and, particularly in this case, toward synthetic fibers. At the same time, it must also be considered that coloring agents have differing levels of stability with respect to oxidation. It is generally the case that water-insoluble coloring agents are more stable with respect to oxidation than water-soluble coloring agents. The concentration of the coloring agent in the washing or cleaning agents varies as a function of solubility and thus also of oxidation sensitivity. For readily water-soluble coloring agents, coloring-agent concentrations in the range of from about 10.sup.-2 wt % to about 10.sup.-2 wt % are typically selected. In the case of pigment dyes, on the other hand, which are particularly preferred because of their brilliance but are less readily water-soluble, the appropriate concentration of the coloring agent in washing or cleaning agents is typically from about 10.sup.-3 wt % to about 10.sup.-4 wt %. Coloring agents that can be oxidatively destroyed in a washing process, as well as mixtures thereof with suitable blue dyes, so-called bluing agents, are preferred. It has proven advantageous to use coloring agents that are soluble in water or at room temperature in liquid organic substances. Anionic coloring agents, e.g. anionic nitroso dyes, are suitable, for example.
[0081] In addition to the components recited hitherto, the washing or cleaning agents can contain further ingredients that further improve the applications-engineering and/or aesthetic properties of said agents. Preferred agents contain one or more substances from the group of electrolytes, pH adjusting agents, fluorescing agents, hydrotopes, foam inhibitors, silicone oils, anti-redeposition agents, optical brighteners, anti-gray agents, shrinkage preventers, crease prevention agents, color transfer inhibitors, antimicrobial active agents, germicides, fungicides, antioxidants, antistatic agents, ironing adjuvants, proofing and impregnation agents, swelling and anti-slip agents, and UV absorbers.
[0082] A large number of very varied salts from the group of the inorganic salts can be used as electrolytes. Preferred cations are the alkali and alkaline-earth metals; preferred anions are the halides and sulfates. From a production-engineering standpoint, the use of NaCl or MgCl.sub.2 in the washing or cleaning agents is preferred.
[0083] In order to bring the pH of washing or cleaning agents into the desired range, the use of pH adjusting agents may be indicated. All known acids resp. bases are usable here, provided their use is not prohibited for environmental or applications-engineering reasons, resp. for reasons of consumer safety. The quantity of these adjusting agents usually does not exceed 1 wt % of the total formulation.
[0084] Appropriate foam inhibitors are soaps, oils, fats, paraffins, or silicone oils, which optionally can be applied onto carrier materials. Suitable carrier materials are, for example, inorganic salts such as carbonates or sulfates, cellulose derivatives, or silicates, as well as mixtures of the aforesaid materials. Agents preferred in the context of the present application contain paraffins, preferably unbranched paraffins (n-paraffins), and/or silicones, preferably linear-polymer silicones, which are constructed according to the (R.sub.2SiO).sub.x pattern and are also referred to as silicone oils. These silicone oils usually represent clear, colorless, neutral, odorless, hydrophobic liquids having a molecular weight between from about 1000 g/mol and about 150,000 g/mol and viscosities between about 10 mPas and about 1,000,000 mPas.
[0085] Suitable anti-redeposition agents are, for example, nonionic cellulose ethers such as methyl cellulose and methylhydroxypropyl cellulose having a from about 15 to about 30 wt % proportion of methoxy groups and a from about 1 to about 15 wt % proportion of hydroxypropyl groups, based in each case on the nonionic cellulose ether.
[0086] Suitable soil repellents are polymers, known from the existing art, of phthalic acid and/or terephthalic acid resp. derivatives thereof, in particular polymers of ethylene terephthalate and/or polyethylene glycol terephthalate or anionically and/or nonionically modified derivatives thereof. Of these, the sulfonated derivatives of phthalic acid polymers and terephthalic acid polymers are particularly preferred.
[0087] Optical brighteners can be added in particular to washing agents in order to eliminate graying and yellowing of the treated textiles. These substances absorb onto the fibers and cause brightening and a simulated bleaching effect by converting invisible ultraviolet radiation into longer-wave visible light, the ultraviolet light absorbed from sunlight being emitted as slightly bluish fluorescence and resulting, with the yellow tone of the grayed or yellowed laundry, in pure white. Suitable compounds derive, for example, from the substance classes of 4,4'-diamino-2,2'-stilbenedisulfonic acids (flavonic acids), 4,4'-distyrylbiphenyls, methylumbelliferones, cumarins, dihydroquinolinones, 1,3-diarylpyrazolines, naphthalic acid imides, benzoxazole, benzisoxazole, and benzimidazole systems, and pyrene derivatives substituted with heterocycles.
[0088] The purpose of anti-gray agents is to keep dirt that has been detached from fibers suspended in the bath, and thus to prevent redeposition of the dirt. Water-soluble colloids, usually organic in nature, are suitable for this, for example water-soluble salts of polymeric carboxylic acids, size, gelatin, salts of ethersulfonic acids of starch or of cellulose, or salts of acidic sulfuric-acid esters of cellulose or of starch. Water-soluble polyamides containing acid groups are also suitable for this purpose. Soluble starch preparations can furthermore be used, for example degraded starch, aldehyde starches, etc. Polyvinylpyrrolidone is also usable. Cellulose ethers such as carboxymethyl cellulose (sodium salt), methyl cellulose, hydroxyalkyl cellulose, and mixed ethers such as methylhydroxyethyl cellulose, methylhydroxypropyl cellulose, methylcarboxymethyl cellulose, and mixtures thereof, are also usable as anti-gray agents.
[0089] Because textile fabrics, in particular those made of rayon, viscose, cotton, and mixtures thereof, can tend to wrinkle because the individual fibers are sensitive to bending, kinking, compression, and squeezing perpendicularly to the fiber direction, synthetic crease-prevention agents can be used. These include, for example, synthetic products based on fatty acids, fatty acid esters, fatty acid amides, fatty acid alkylol esters, fatty acid alkylolamides, or fatty alcohols that are usually reacted with ethylene oxide, or products based on lecithin or modified phosphoric acid esters.
[0090] The purpose of proofing and impregnation methods is to finish textiles with substances that prevent the deposition of dirt or make it easier to wash out. Preferred proofing and impregnation agents are perfluorinated fatty acids, including in the form of their aluminum and zirconium salts, organic silicates, silicones, polyacrylic acid esters having perfluorinated alcohol components, or polymerizable compounds coupled to a perfluorinated acyl or sulfonyl residue. Antistatic agents can also be contained. Dirt-repellent finishing with proofing and impregnation agents is often categorized as an "easy-care" finish. Penetration of the impregnation agents, in the form of solutions or emulsions of the relevant active agents, can be facilitated by the addition of wetting agents that reduce surface tension. A further area of use of proofing and impregnation agents is water-repellent finishing of textile materials, tents, awnings, leather, etc. in which, in contrast to waterproofing, the fabric pores are not sealed, i.e. the material is still able to "breathe" (hydrophobizing). The hydrophobizing agents used for hydrophobizing cover the textiles, leather, paper, wood, etc. with a very thin layer of hydrophobic groups such as longer alkyl chains or siloxane groups. Suitable hydrophobizing agents are, for example, paraffins, waxes, metal soaps, etc. having added portions of aluminum or zirconium salts, quaternary ammonium compounds with long-chain alkyl residues, urea derivatives, fatty acid-modified melamine resins, chromium-complex salts, silicones, organo-tin compounds, and glutaric dialdehyde, as well as perfluorinated compounds. The hydrophobized materials are not oily to the touch, but water droplets bead up on them (similarly to oiled fabrics) without wetting them. Silicone-impregnated textiles, for example, have a soft hand and are water- and dirt-repellent; drops of ink, wine, fruit juice, and the like are easier to remove.
[0091] Antimicrobial active substances can be used in order to counteract microorganisms, if they do not inhibit the function of the bacterial spores of the present disclosure. A distinction is made here, in terms of the antimicrobial spectrum and mechanism of action, between bacteriostatics and bactericides, fungistatics and fungicides, etc. Substances from these groups are, for example, benzalkonium chlorides, alkylarylsulfonates, halogen phenols, and phenol mercuric acetate; these compounds can also be entirely omitted.
[0092] The agents can contain antioxidants in order to prevent undesirable changes to the washing and cleaning agents and/or to the treated textiles caused by the action of oxygen and other oxidative processes. This class of compounds includes, for example, substituted phenols, hydroquinones, catechols, and aromatic amines, as well as organic sulfides, polysulfides, dithiocarbamates, phosphites, and phosphonates.
[0093] Increased wearing comfort can result from the additional use of antistatic agents. Antistatic agents increase the surface conductivity and thus make possible improved dissipation of charges that have formed. External antistatic agents are usually substances having at least one hydrophilic molecule ligand, and yield a more or less hygroscopic film on the surfaces. These usually surface-active antistatic agents can be subdivided into nitrogen-containing (amines, amides, quaternary ammonium compounds), phosphorus-containing (phosphoric acid esters), and sulfur-containing antistatic agents (alkylsulfonates, alkyl sulfates). Lauryl-(resp. stearyl)dimethylbenzylammonium chlorides are likewise suitable as antistatic agents for textile fabrics resp. as an additive to washing agents, an avivage effect additionally being achieved.
[0094] Silicone derivatives can be used in textile washing agents in order to improve the water absorption capability and rewettability of the treated textile fabrics and to facilitate ironing of the treated textiles. These additionally improve the rinsing behavior of washing or cleaning agents thanks to their foam-inhibiting properties. Preferred silicone derivatives are, for example, polydialkyl- or alkylarylsiloxanes in which the alkyl groups comprise one to five carbon atoms and are entirely or partly fluorinated. Preferred silicones are polydimethylsiloxanes, which optionally can be derivatized and are then aminofunctional or quaternized resp. comprise Si--OH, Si--H, and/or Si--Cl bonds. Further preferred silicones are the polyalkylene oxide-modified polysiloxanes, i.e. polysiloxanes that comprise, for example, polyethylene glycols, as well as polyalkylene oxide-modified dimethylpolysiloxanes.
[0095] Lastly, UV absorbers, which are absorbed onto the treated textiles and improve the light-fastness of the fibers, can also be used. Compounds that exhibit these desired properties are, for example, the compounds that act by radiationless deactivation, and derivatives of benzophenone having substituents in the 2- and/or 4-position. Also suitable are substituted benzotriazoles, acrylates phenyl-substituted in the 3-position (cinnamic acid derivatives) optionally having cyano groups in the 2-position, salicylates, organic nickel complexes, and natural substances such as umbelliferone and endogenous urocanic acid.
[0096] Protein hydrolysates are further suitable active substances because of their fiber-care-providing effect. Protein hydrolysates are product mixtures that are obtained by acid-, base-, or enzyme-catalyzed breakdown of proteins. Protein hydrolysates of both vegetable and animal origin can be used. Animal protein hydrolysates are, for example, elastin, collagen, keratin, silk, and milk protein hydrolysates, which can also be present in the form of salts. It is preferred to use protein hydrolysates of vegetable origin, e.g. soy, almond, rice, pea, potato, and wheat protein hydrolysates. Although the use of protein hydrolysates as such is preferred, amino acid mixtures obtained in other ways, or individual amino acids such as arginine, lysine, histidine, or pyroglutamic acid, can also optionally be used instead of them. It is also possible to employ derivatives of protein hydrolysates, for example in the form of their fatty acid condensation products.
EXAMPLES
Example 1: Prevention of Odor in Test Tubes
[0097] In test tubes, sterile human sweat was fermented with various microorganisms, known to produce the malodor "sweat". Bacterial spores of Bacillus tequilensis were added and the mixture incubated for up to 27 hours.
[0098] Persons trained in olfaction smelled the tubes and rated the intensity of the malodor on a scale of 0 to 4 (0 being no odor, 1=weak odor, 2=moderate odor, 3=strong odor, 4=extremely strong odor).
[0099] The results are presented in Table 1:
TABLE-US-00001 TABLE 1 Malodor scores With spores, Incubation time Control Bacillus tequilensis, [hours] (without spores) dosage 2.5 .times. 10.sup.2 cfu/ml 19 1.7 1.0 27 2.5 2.3
[0100] The results show the significantly improved performance of the use of bacterial spores for the elimination of the malodor "sweat".
[0101] While at least one exemplary embodiment has been presented in the foregoing detailed description of the present disclosure, it should be appreciated that a vast number of variations exist. It should also be appreciated that the exemplary embodiment or exemplary embodiments are only examples, and are not intended to limit the scope, applicability, or configuration of the present disclosure in any way. Rather, the foregoing detailed description will provide those skilled in the art with a convenient road map for implementing an exemplary embodiment of the present disclosure, it being understood that various changes may be made in the function and arrangement of elements described in an exemplary embodiment without departing from the scope of the present disclosure as set forth in the appended claims and their legal equivalents.
* * * * *
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.