Drug Delivery System and Methods of Treating Open Angle Glaucoma and Ocular Hypertension
Cadden; Suzanne ; et al.
U.S. patent application number 15/985699 was filed with the patent office on 2019-03-28 for drug delivery system and methods of treating open angle glaucoma and ocular hypertension. This patent application is currently assigned to Mati Therapeutics Inc.. The applicant listed for this patent is Mati Therapeutics Inc.. Invention is credited to Suzanne Cadden, Yong Hao, Deepank Utkhede.
| Application Number | 20190091066 15/985699 |
| Document ID | / |
| Family ID | 49955680 |
| Filed Date | 2019-03-28 |





View All Diagrams
| United States Patent Application | 20190091066 |
| Kind Code | A1 |
| Cadden; Suzanne ; et al. | March 28, 2019 |
Drug Delivery System and Methods of Treating Open Angle Glaucoma and Ocular Hypertension
Abstract
A method of decreasing intraocular pressure (IOP) in an eye of a patient in need thereof includes implanting a first lacrimal implant through an upper punctum and into an upper lacrimal canaliculus of the eye of the patient. The method may further comprise implanting a second lacrimal implant through a lower punctum and into a lower lacrimal canaliculus of the eye of the patient, and releasing, on a sustained basis a therapeutically effective amount of an intraocular pressure-reducing therapeutic agent.
| Inventors: | Cadden; Suzanne; (North Vancouver, CA) ; Hao; Yong; (Vancouver, CA) ; Utkhede; Deepank; (Surrey, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | Mati Therapeutics Inc. Austin TX |
||||||||||
| Family ID: | 49955680 | ||||||||||
| Appl. No.: | 15/985699 | ||||||||||
| Filed: | May 22, 2018 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 13779628 | Feb 27, 2013 | 9974685 | ||
| 15985699 | ||||
| 13598573 | Aug 29, 2012 | |||
| 13779628 | ||||
| 61644397 | May 8, 2012 | |||
| 61528736 | Aug 29, 2011 | |||
| 61717615 | Oct 23, 2012 | |||
| 61680641 | Aug 7, 2012 | |||
| 61659921 | Jun 14, 2012 | |||
| 61644401 | May 8, 2012 | |||
| 61642287 | May 3, 2012 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61K 9/0051 20130101; A61K 9/0092 20130101; A61F 9/00781 20130101; A61F 9/0017 20130101; A61F 9/00772 20130101; A61K 31/5575 20130101 |
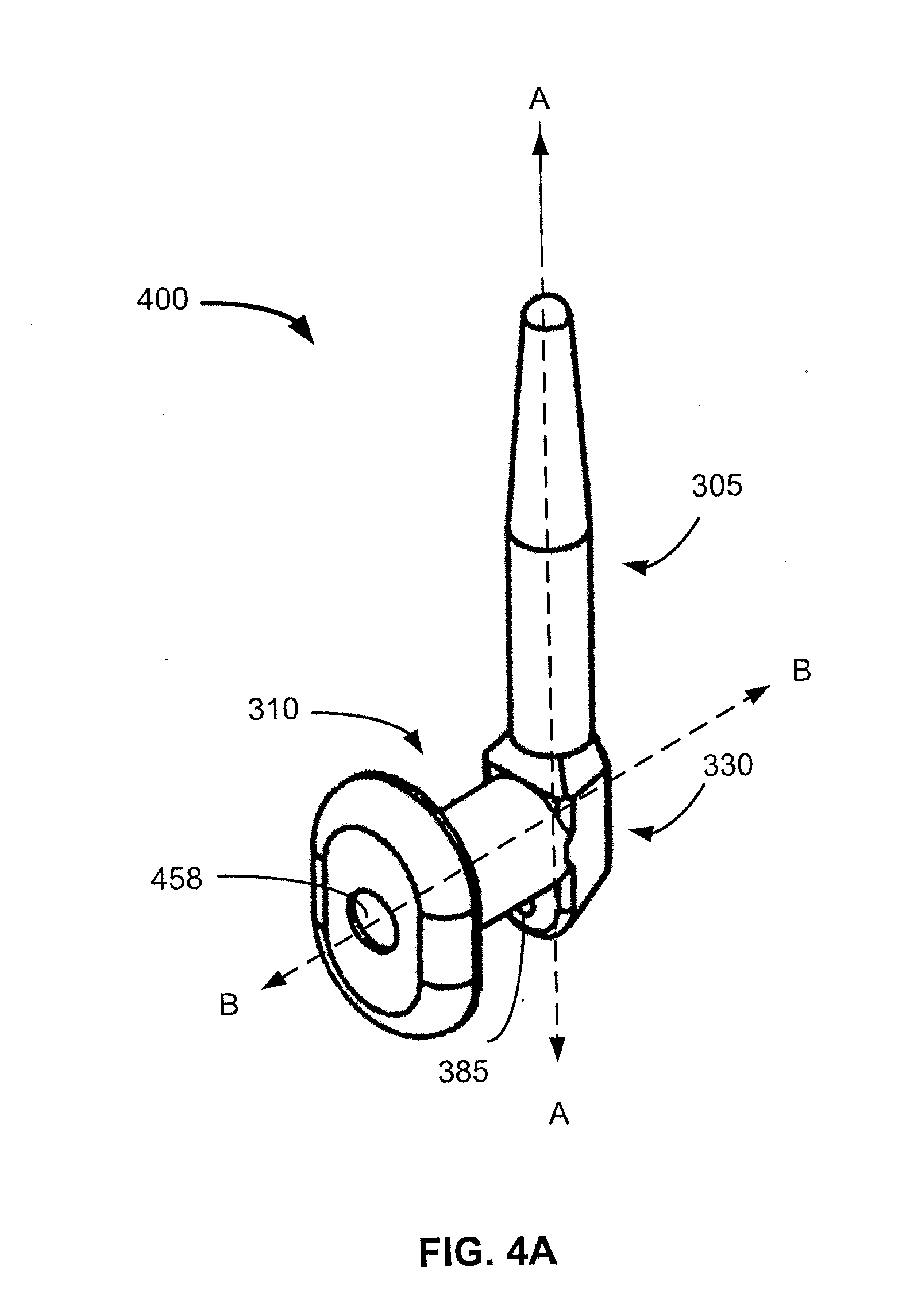
| International Class: | A61F 9/00 20060101 A61F009/00; A61F 9/007 20060101 A61F009/007; A61K 31/5575 20060101 A61K031/5575; A61K 9/00 20060101 A61K009/00 |
Claims
1. A kit for treating a patient with Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye, comprising: a unit dosage format per eye of a prostaglandin analog comprising a first lacrimal implant and a second lacrimal implant, wherein the first lacrimal implant comprises a sustained release formulation of a prostaglandin analog configured for release in a therapeutically effective dose from the first lacrimal implant over a treatment period; and, wherein the second lacrimal implant is a blank lacrimal implant that does not comprise a prostaglandin analog.
2. The kit of claim 1, wherein the first or second lacrimal implant is a punctual plug.
3. The kit of claim 1, wherein the first or second lacrimal implant comprises a first member defining a first axis and having a first end along the first axis; a second member defining a second axis and having a second end along the second axis; and a third member connecting the first end of the first member and the second end of the second member at a first angle to form an angled intersection.
4. The kit of claim 3, wherein in the second member of the first or second lacrimal implant further comprises a cavity for insertion of a drug core comprising the prostaglandin analog.
5. The kit of claim 3, wherein the third member of the first or second lacrimal implant further comprises a bore that is characterized by a third axis and a second angle, wherein the first angle is defined by the first axis with respect to the second axis, the second angle is defined by the first axis with respective to the third axis, and the bore is configured to be accessible to an insertion tool for facilitating insertion of the implant.
6. The kit of claim 3, wherein the lacrimal implant further comprises the prostaglandin analog dispersed throughout the implant.
7. The kit of claim 1, wherein the first or second lacrimal implant is configured with a retention rate of about 90% or greater at week 12 in a lower punctum.
8. The kit of claim 1, wherein the first or second lacrimal implant is configured with a retention rate of about 85% or greater at week 8 in the upper punctum.
9. The kit of claim 1, wherein the first or second lacrimal implant is made of a material that comprises a plastic, a rubber, a polymer, a composite or a material that comprises a liquid silicone rubber, or a mixture including a liquid silicone rubber.
10. The kit of claim 9, wherein the first or second lacrimal implant further comprises a green colorant.
11. The kit of claim 1, wherein the first or second lacrimal implant comprises a first member, a second member and a heel that is at least partially fabricated with silicone
12. (canceled)
13. (canceled)
14. The kit of claim 1, wherein the first lacrimal implant is configured for insertion into an upper punctum of the eye.
15. The kit of claim 1, wherein the second lacrimal implant is configured for insertion into a lower punctum of the eye.
16-24. (canceled)
25. The kit of claim 1, wherein the sustained release formulation is configured to provide a dosage of the prostaglandin analog sufficient to reduce the intraocular pressure of the eye by at least 5 mm Hg from baseline for a continuous period of time selected from at least 12 weeks after implantation of the first lacrimal implant and second lacrimal implant.
26. The kit of claim 1, wherein the treatment period is at least 4 weeks.
27. The kit of claim 1, wherein the treatment period is at least 8 weeks.
28. The kit of claim 1, wherein the treatment period is at least 12 weeks.
29. The kit of claim 1, further comprising an insertion tool.
30. The kit of claim 1, wherein the patient has a baseline TOP between about 22 mm Hg and about 33 mm Hg.
31. The kit of claim 1, wherein the first or second lacrimal implant comprises a first member defining a first axis and having a first end along the first axis; a second member defining a second axis and having a second end along the second axis; and a third member connecting the first end of the first member and the second end of the second member at a first angle to form an angled intersection and wherein the third member of the lacrimal implant further comprises a bore defining a third axis and a second angle having an upper surface, wherein the first angle is defined by the first axis with respect to the second axis, the second angle is defined by the first axis with respect to the third axis, and the bore is configured to be accessible to an insertion tool for facilitating insertion of the implant and extended from the upper surface into the third member.
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application is a continuation-in-part of U.S. Ser. No. 13/598,573, filed Aug. 29, 2012, which claims priority to U.S. Provisional Patent Application No. 61/528,736, filed on Aug. 29, 2011, and U.S. Provisional Patent Application No. 61/644,397, filed on May 8, 2012, the disclosures of each of which are incorporated herein by reference in their entirety for all purposes. This application claims the benefit under 35 U.S.C. 1.19(e) of U.S. Provisional Patent Application No. 61/642,287, filed May 3, 2012; U.S. Provisional Patent Application No. 61/644,401 filed May 8, 2012; U.S. Provisional Patent Application No. 61/644,397 filed May 8, 2012; U.S. Provisional Patent Application No. 61/659,921 filed Jun. 14, 2012; 61/680,641 filed Aug. 7, 2012 and U.S. Provisional Patent Application No. 61/717,615 filed Oct. 23, 2012, the disclosures of each of which are incorporated herein by reference in their entirety for all purposes.
FIELD OF THE INVENTION
[0002] This application pertains generally to methods of treating ocular diseases, particularly those with elevated intraocular hypertension.
BACKGROUND OF THE INVENTION
[0003] Glaucoma is a collection of disorders characterized by progressive visual field loss due to optic nerve damage. It is the leading cause of blindness in the United States, affecting 1-2% of individuals aged 60 and over. Although there are many risk factors associated with the development of glaucoma (age, race, myopia, family history, and injury), elevated intraocular pressure (IOP), also known as ocular hypertension (OH), is the only risk factor successfully manipulated and correlated with the reduction of glaucomatous optic neuropathy. Public health figures estimate that 2.5 million Americans manifest ocular hypertension.
[0004] In glaucoma associated with an elevation in eye pressure the source of resistance to outflow is in the trabecular meshwork. The tissue of the trabecular meshwork allows the "aqueous" to enter Schlemm's canal, which then empties into aqueous collector channels in the posterior wall of Schlemm's canal and then into aqueous veins. The aqueous or aqueous humor is a transparent liquid that fills the region between the cornea at the front of the eye and the lens. The aqueous humor is constantly secreted by the ciliary body around the lens, so there is a continuous flow of the aqueous humor from the ciliary body to the eye's front chamber. The eye's pressure is determined by a balance between the production of aqueous and its exit through the trabecular meshwork (major route) or via uveal scleral outflow (minor route). The trabecular meshwork is located between the outer rim of the iris and the internal periphery of the cornea. The portion of the trabecular meshwork adjacent to Schlemm's canal causes most of the resistance to aqueous outflow (juxtacanalicular meshwork).
[0005] Glaucoma is grossly classified into two categories: closed-angle glaucoma and open-angle glaucoma. Closed-angle glaucoma is caused by closure of the anterior angle by contact between the iris and the inner surface of the trabecular meshwork. Closure of this anatomical angle prevents normal drainage of aqueous humor from the anterior chamber of the eye. Open-angle glaucoma (OAG) is any glaucoma in which the angle of the anterior chamber remains open, but the exit of aqueous through the trabecular meshwork is diminished. The exact cause for diminished filtration is unknown for most cases of open-angle glaucoma. However, there are secondary open-angle glaucomas that may include edema or swelling of the trabecular spaces (from steroid use), abnormal pigment dispersion, or diseases such as hyperthyroidism that produce vascular congestion.
[0006] Although there is no known cure, the principal objective in treating patients with OAG or OH is to preserve visual function by the reduction and maintenance of IOP. As such all current therapies for glaucoma are directed at decreasing intraocular pressure. Self-administered topical agents or pills are usually the first-line choice of therapy for reducing IOP. This therapy reduces the production of aqueous humor or increases the outflow of aqueous. Other means to treat glaucoma and ocular hypertension, involve surgical therapy for open-angle glaucoma such as laser (trabeculoplasty), trabeculectomy and aqueous shunting implants after failure of trabeculectomy or if trabeculectomy is unlikely to succeed. Trabeculectomy is a major surgery that is most widely used and is augmented with topically applied anticancer drugs such as 5-flurouracil or mitomycin-c to decrease scarring and increase surgical success.
[0007] Topical eye drops, though effective, can be inefficient. For instance, when an eye drop is instilled in an eye, it often overfills the conjunctival sac (i.e., the pocket between the eye and the lids) causing a substantial portion of the drop to be lost due to overflow of the lid margin and spillage onto the cheek. In addition, a large portion of the drop remaining on the ocular surface can be washed away into and through a lacrimal canaliculus, thereby diluting the concentration of the drug before it can treat the eye. Further, in many cases, topically applied medications have a peak ocular effect within about two hours, after which additional applications of the medications should be performed to maintain the therapeutic benefit. PCT Publication WO 06/014434 (Lazar), which is incorporated herein by reference in its entirety, may be relevant to these or other issues associated with eye drops.
[0008] Compounding ocular management difficulty, patients often do not use their eye drops as prescribed. Noncompliance rates of at least 25% are reported. This poor compliance can be due to discomfort and the normal reflex to protect the eye. Therefore, one or more drops may miss the eye. Older patients may have additional problems instilling drops due to arthritis, unsteadiness, and decreased vision. Pediatric and psychiatric populations pose difficulties as well.
[0009] Prostaglandins are one group of drugs administered as eye drops to patients diagnosed with glaucoma. Latanoprost is an ester analogue of prostaglandin F.sub.2.alpha. that reduces IOP by increasing uveoscleral outflow. Latanoprost is marketed as Xalatan.RTM. (latanoprost ophthalmic solution) 0.005% (50 .mu.g/mL) (Xalatan PI 2011). The IOP-lowering efficacy of Xalatan lasts for up to 24 hours after a single topical dose, which allows for a once daily dosage regimen.
[0010] Lacrimal implants are devices that are inserted into a punctum and an associated lacrimal canaliculus of an eye, either to block drainage of tears (to prevent conditions such as dry eye), or to contain a quantity of drug for release into the eye.
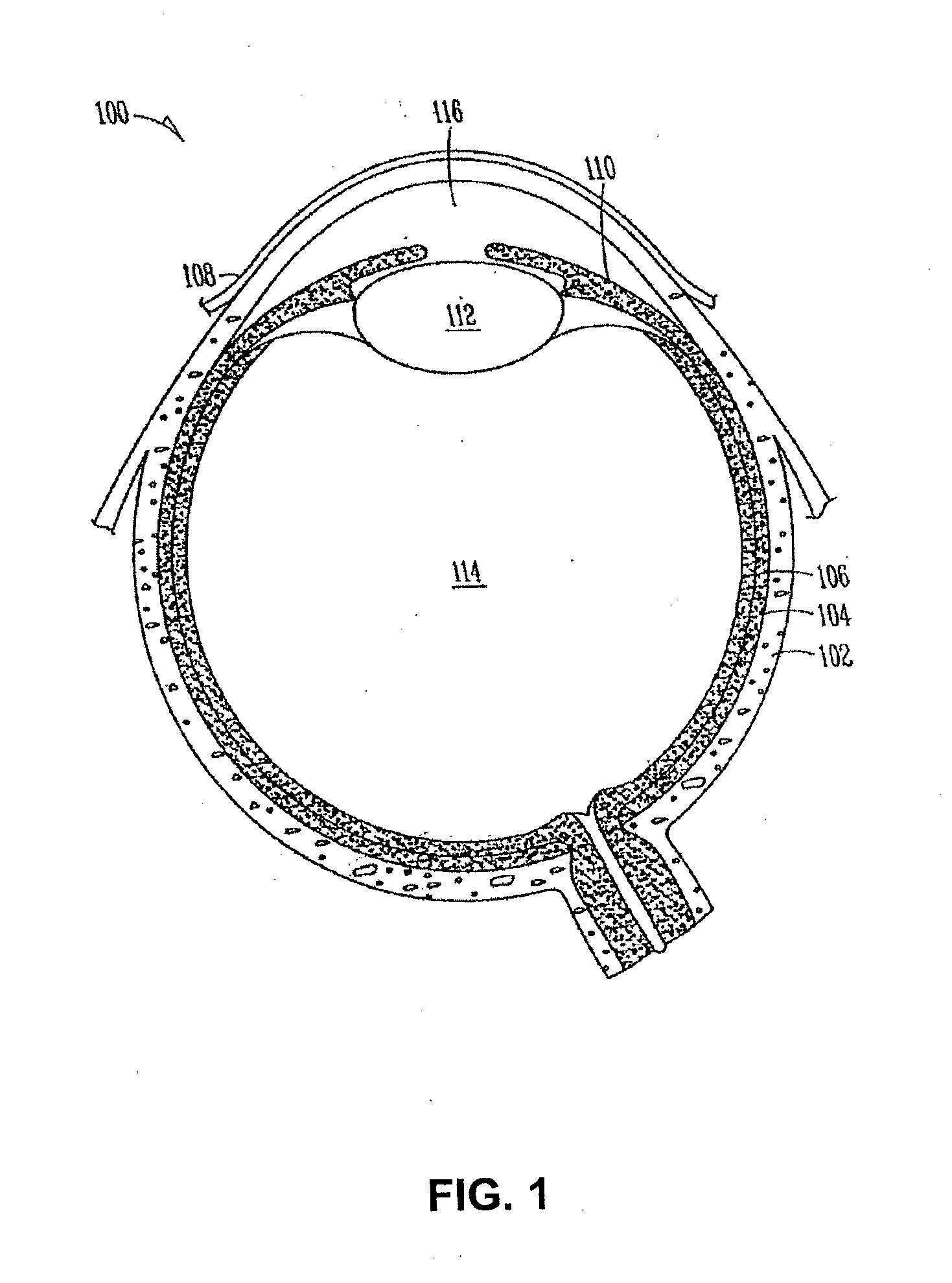
[0011] FIGS. 1-2 illustrate example views of anatomical tissue structures associated with an eye 100. Certain of the anatomical tissue structures shown may be suitable for treatment using the various lacrimal implants and methods discussed herein. The eye 100 is a spherical structure including a wall having three layers: an outer sclera 102, a middle choroid layer 104 and an inner retina 106. The sclera 102 includes a tough fibrous coating that protects the inner layers. It is mostly white except for the transparent area at the front, commonly known as the cornea 108, which allows light to enter the eye 100.
[0012] The choroid layer 104, situated inside the sclera 102, contains many blood vessels and is modified at the front of the eye 100 as a pigmented iris 110. A biconvex lens 112 is situated just behind the pupil. A chamber 114 behind the lens 112 is filled with vitreous humor, a gelatinous substance. Anterior and posterior chambers 116 are situated between the cornea 108 and iris 110, respectively and filled with aqueous humor. At the back of the eye 100 is the light-detecting retina 106.
[0013] The cornea 108 is an optically transparent tissue that conveys images to the back of the eye 100. It includes a vascular tissue to which nutrients and oxygen are supplied via bathing with lacrimal fluid and aqueous humor as well as from blood vessels that line the junction between the cornea 108 and sclera 102. The cornea 108 includes a pathway for the permeation of drugs into the eye 100.
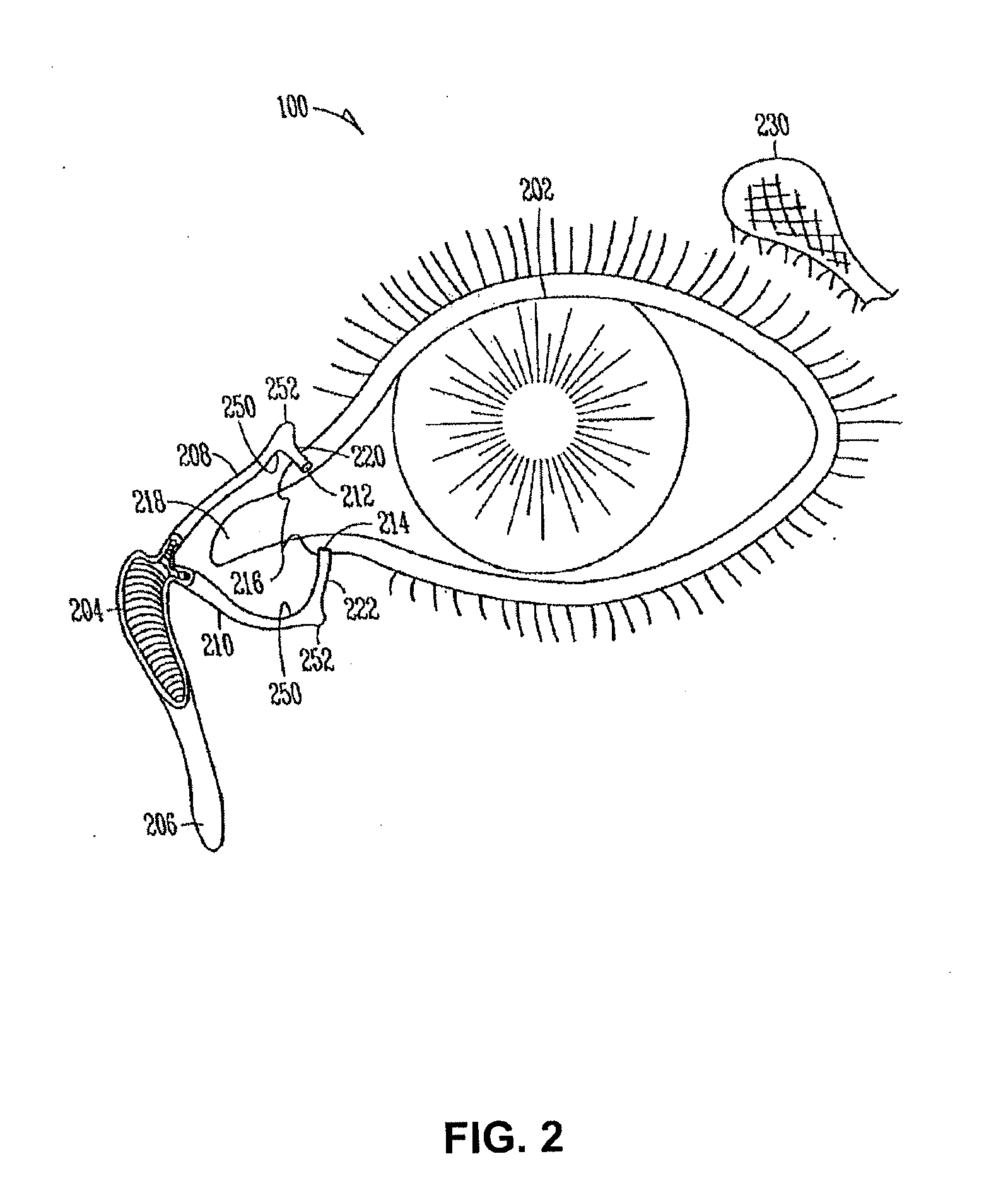
[0014] Turning to FIG. 2, other anatomical tissue structures associated with the eye 100 including the lacrimal drainage system, which includes a secretory system 230, a distributive system and an excretory system, are shown. The secretory system 230 comprises secretors that are stimulated by blinking and temperature change due to tear evaporation and reflex secretors that have an efferent parasympathetic nerve supply and secrete tears in response to physical or emotional stimulation. The distributive system includes the eyelids 202 and the tear meniscus around the lid edges of an open eye, which spread tears over the ocular surface by blinking, thus reducing dry areas from developing.
[0015] The excretory system of the lacrimal drainage system includes, in order of flow, drainage, the lacrimal puncta, the lacrimal canaliculi, the lacrimal sac 204 and the lacrimal duct 206. From the lacrimal duct 206, tears and other flowable materials drain into a passage of the nasolacrimal system. The lacrimal canaliculi include an upper (superior) lacrimal canaliculus 208 and a lower (inferior) lacrimal canaliculus 210, which respectively terminate in an upper 212 and lower 214 lacrimal punctum. The upper 212 and lower 214 punctum are slightly elevated at the medial end of a lid margin at the junction 216 of the ciliary and lacrimal portions near a conjunctival sac 218. The upper 212 and lower 214 punctum are generally round or slightly ovoid openings surrounded by a connective ring of tissue. Each of puncta 212, 214 leads into a vertical portion 220, 222 of their respective canaliculus before turning more horizontal at a canaliculus curvature 250 to join one another at the entrance of the lacrimal sac 204. The canaliculi 208, 210 are generally tubular in shape and lined by stratified squamous epithelium surrounded by elastic tissue, which permits them to be dilated. As shown, a lacrimal canaliculus ampulla 252 exists near an outer edge of each canaliculus curvature 250.
[0016] For numerous reasons (e.g., the size, shape, positioning, and materials of some conventional lacrimal implants, and variability in punctum size and shape), retention of the implants in the punctum and associated lacrimal canaliculus has been inconsistent. Users of lacrimal implants may inadvertently dislodge the lacrimal implant by wiping their eye. Further, some configurations of lacrimal implants may dislodge themselves, such as when a user sneezes, or tears excessively.
[0017] Accordingly, it is desirable to have a lacrimal implant that solves these problems and provides improved retention across different sizes of puncta, while providing efficient administration of a therapeutic agent for treatment of open angle glaucoma (OAG) and/or ocular hypertension (OH).
SUMMARY OF THE INVENTION
[0018] In an exemplary embodiment, the present invention provides methods of reducing intraocular pressure (IOP) in an eye. In an exemplary embodiment, the method of the invention utilizes a latanoprost-eluting lacrimal implant inserted into at least the upper punctum of an eye. Previous methods of delivering latanoprost to the eye using a latanoprost-eluting lacrimal implant have met with varied and minimal success. For example, as show in FIG. 16, in an eye implanted with a single latanoprost-eluting plug in the lower punctum, the reduction in IOP is minimal, and is substantially identical across a range of latanoprost loadings: from 3.5 .mu.g to 95 .mu.g, the IOP does not decrease even though more latanoprost is being delivered by the plugs with higher latanoprost loading. See, FIG. 17. Thus, it is surprising that the methods of the present invention, in which an eye has a latanoprost-eluting punctal implant in at least the upper punctum yields a statistically significant reduction in IOP after about two weeks.
[0019] In an exemplary embodiment, the methods of the invention provide a reduction in IOP of at least about 4 mm Hg, at least about 5 mm Hg, at least about 6 mm Hg or at least about 7 mm Hg from baseline during the treatment period during which the lacrimal implant is inserted into at least the upper punctum,
[0020] In various embodiments, the method of the invention includes implanting a first lacrimal implant through a first lacrimal punctum and into a first lacrimal canaliculus of the eye of the patient. The first lacrimal implant is configured to release an intraocular pressure-reducing therapeutic agent to the eye of the patient on a sustained basis. In an exemplary embodiment, the first implant contains approximately 0 .mu.g (blank), 46 .mu.g or 95 .mu.g of latanoprost and a second implant contains about 95 .mu.g of latanoprost or is a "blank" implant and does not comprise latanoprost. In an exemplary embodiment, the first implant is installed in the upper punctum and the second implant is installed in the lower punctum. In various embodiments, the location of the implants is reversed. In other embodiments, only the first lacrimal implant is installed in the upper punctum and no implant is placed in the lower punctum.
[0021] In certain embodiments, the method of the invention can include implanting more than one implant in more than one punctum of one or more eye. Thus, in various embodiments, the method also includes implanting a second lacrimal implant through a second punctum and into a second lacrimal canaliculus of the eye of the patient, the second lacrimal implant being configured to release the intraocular pressure-reducing therapeutic agent to the eye of the patent on a sustained basis. In an alternative embodiment, the second lacrimal implant is a blank.
[0022] In various embodiments, the implant is configured to release, on a sustained basis over a selected time course to the eye, a total amount of the intraocular pressure-reducing therapeutic agent from a combination of the first lacrimal implant and the second lacrimal implant greater than or equal to a recommended daily total dose of the intraocular pressure-reducing therapeutic agent in eye drop form to reduce intraocular pressure of the eye by at least 4 mm Hg from baseline for a continuous period of time of at least 4 weeks after implantation of the first lacrimal implant and the second lacrimal implant.
[0023] In an exemplary embodiment, the invention provides a method for reducing intraocular pressure in an eye of a subject in need thereof. An exemplary method includes implanting a first lacrimal implant through a first punctum and into a first lacrimal canaliculus of an eye of the subject. The first lacrimal implant is configured to release a therapeutically effective amount of an intraocular pressure-reducing therapeutic agent to the eye of the patient on a sustained basis. In various embodiments a second implant is installed in a second punctum or in a second eye. Thus, there is provided a method as set forth above, further comprising implanting a second lacrimal implant through a second punctum and into a second lacrimal canaliculus of the eye of the subject. The second lacrimal implant is configured to release the intraocular pressure-reducing therapeutic agent to the eye of the patent on a sustained basis. The method also includes, once the one or more implant is installed in an eye, releasing, on a sustained basis over a selected time course to the eye, a total amount of the intraocular pressure-reducing therapeutic agent from a combination of the first lacrimal implant and the second lacrimal implant. The total amount of therapeutic agent released is sufficient to reduce the intraocular pressure.
[0024] The implant can be of any useful form, structure or composition. In an exemplary embodiment, the implant includes, a first member defining a first axis and having a first end along the first axis. The implant also includes a second member defining a second axis and having a second end along the second axis; and a third member connecting the first end of the first member and the second end of the second member at a first angle to form an angled intersection, and the third member comprising a bore that is characterized by a third axis and a second angle. In general, the first angle is defined by the first axis with respect to the second axis, the second angle is defined by the first axis with respective to the third axis, and the bore is configured to be accessible to an insertion tool for facilitating insertion of the implant.
[0025] Also provided are kits that include at least one implant. An exemplary kit includes one or more implant operatively engaged to an implanting tool of use in implanting the device in the punctum of a subject's eye.
[0026] The devices and methods described herein include a removable, and optionally drug releasing, lacrimal implant, which can be implanted in the lacrimal canaliculus through a lacrimal punctum. In various embodiments, the lacrimal implants described herein utilize the features of the nasolacrimal drainage system (e.g., by mimicking the shape of the lacrimal canaliculus) to provide improved patient comfort and implant retention in the ocular anatomy. In this way, exemplary lacrimal implants described herein overcome drawbacks associated with current implants. The lacrimal implants described herein are easily implanted and removed without much biasing of the lacrimal punctum or associated canaliculus, and are securely retained in the lacrimal canaliculus upon implantation, optionally without being pre-sized to a particular lacrimal punctum or canaliculus diameter. In various embodiments, the implants are drug delivery system, providing sustained, localized release of one or more drugs or other therapeutic agents at a desired therapeutic level for an extended period of time.
[0027] In an exemplary embodiment, the invention provides an implant for insertion into a lacrimal canaliculus. An exemplary implant includes, a first member defining a first axis and having a first end along the first axis. The implant also includes a second member defining a second axis and having a second end along the second axis. The implant further includes a third member connecting the first end of the first member and the second end of the second member at a first angle to form an angled intersection. The third member includes a bore that is characterized by a third axis and a second angle. The bore is configured to be accessible to an insertion tool for facilitating insertion of the implant. In various embodiments, the first angle is defined by the first axis with respect to the second axis and the second angle is defined by the first axis with respective to the third axis.
[0028] In various embodiments, the invention includes a kit having an implant of the invention and an insertion tool for inserting the implant into the punctum.
[0029] Also provided is a method of treating an ocular disease using one or more punctal implant.
[0030] These and other embodiments, advantages, and aspects of the methods disclosed herein are set forth in part in following detailed description.
BRIEF DESCRIPTION OF THE DRAWINGS
[0031] In the drawings, like numerals describe similar components throughout the several views. Like numerals having different letter suffixes represent different instances of similar components. The drawings illustrate generally, by way of example, but not by way of limitation, various embodiments disclosed herein.
[0032] FIG. 1 illustrates an example of anatomical tissue structures associated with an eye, certain of these tissue structures providing a suitable environment in which a lacrimal implant can be used.
[0033] FIG. 2 illustrates another example of anatomical tissue structures associated with an eye, certain of these tissue structures providing a suitable environment in which a lacrimal implant can be used.
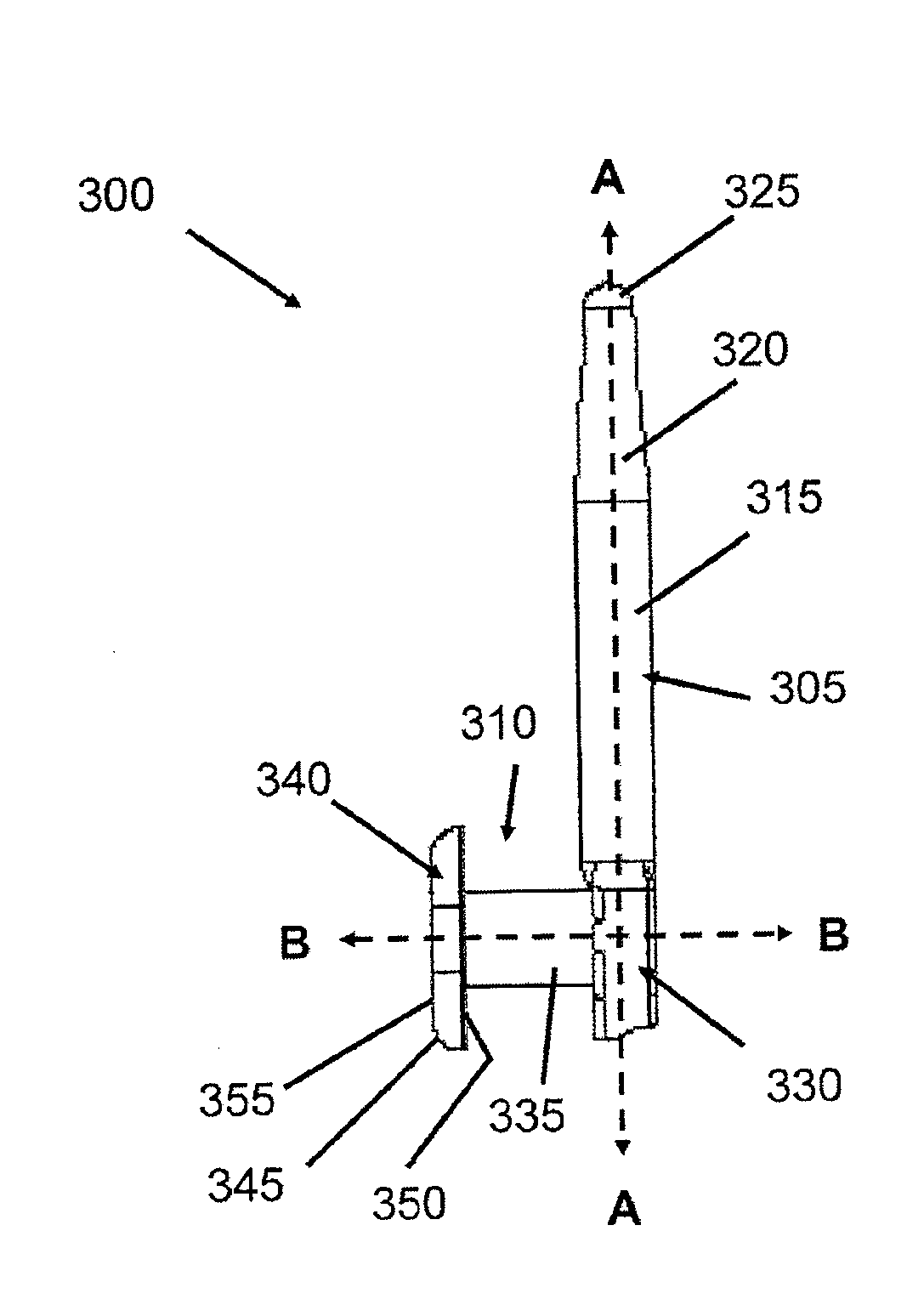
[0034] FIG. 3A provides a perspective view of an implant in accordance with an embodiment of the present invention.
[0035] FIG. 3B is a side view of an implant in accordance with an embodiment of the present invention.
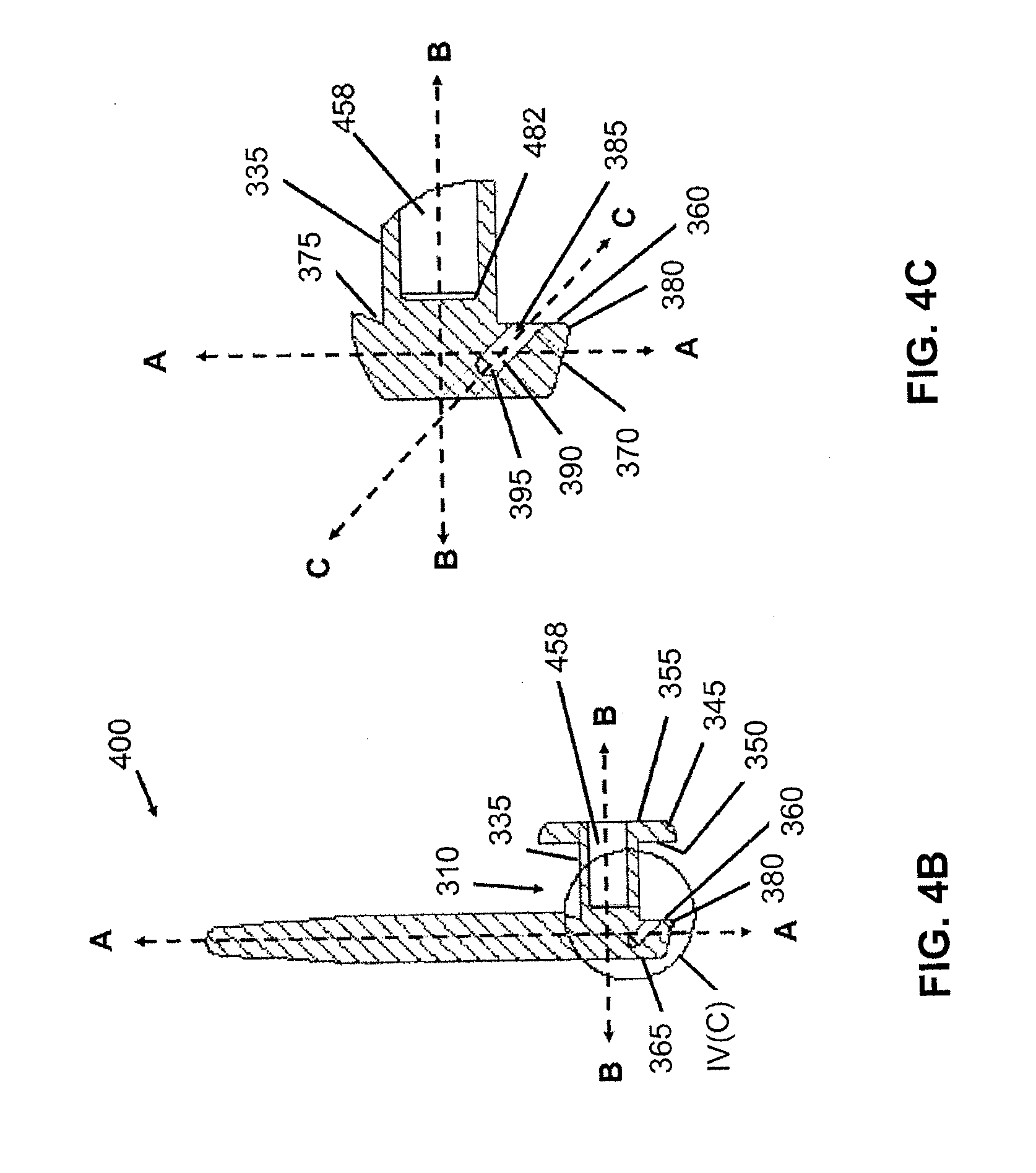
[0036] FIG. 3C is a side view illustrating the second member and the third member of an implant in accordance with an embodiment of the present invention.
[0037] FIG. 3D is a back view of an implant in accordance with an embodiment of the present invention.
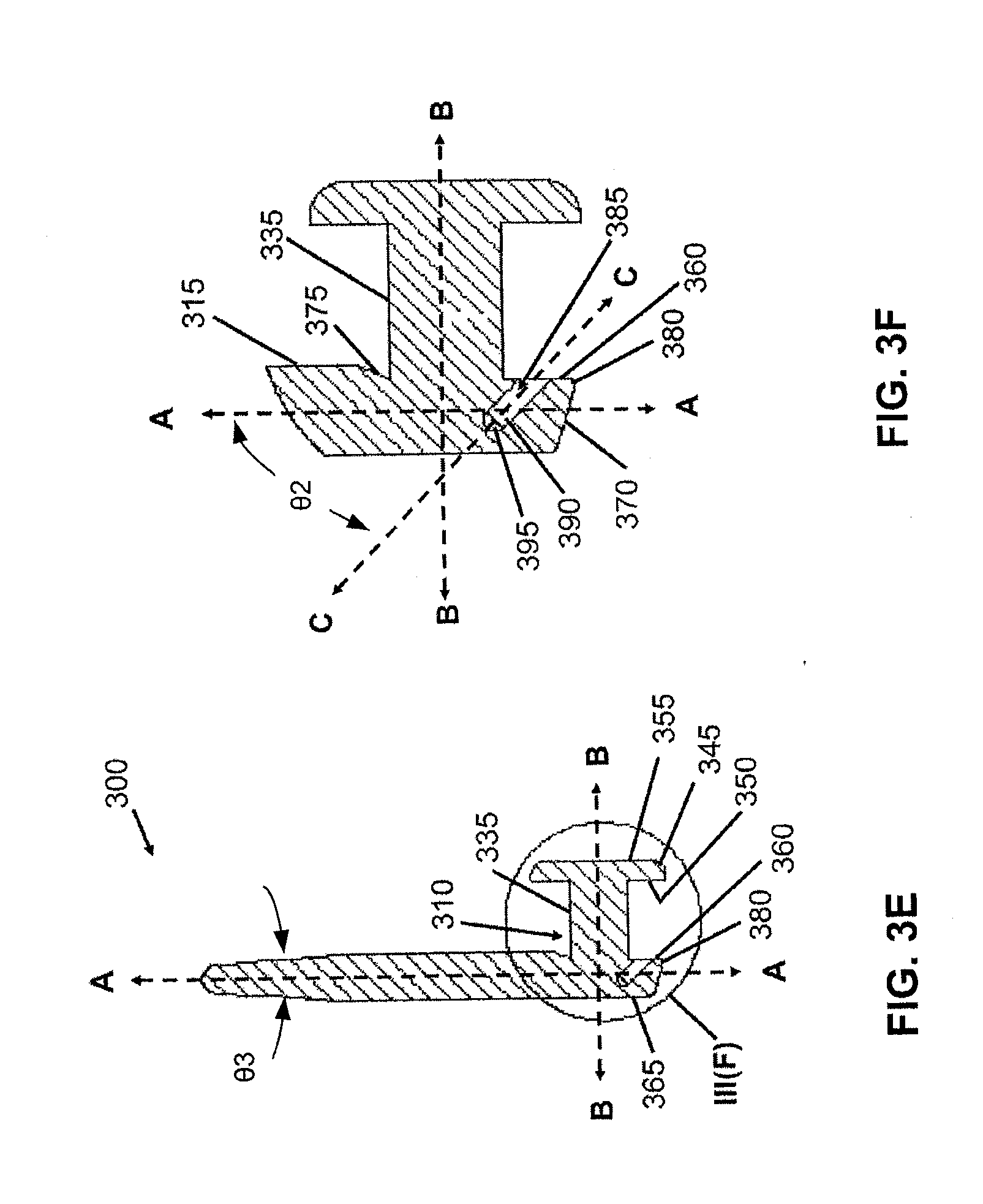
[0038] FIG. 3E is a cross-sectional view taken about line III(E)-III(E) of FIG. 3D depicting an implant with a bore, in accordance with an embodiment of the present invention.
[0039] FIG. 3F is a partially enlarged view of FIG. 3E taken about circle III(F) depicting the second member, the third member and a bore formed in the third member of an implant, in accordance with an embodiment of the present invention.
[0040] FIG. 4A provides a perspective view of an implant in accordance with an embodiment of the present invention.
[0041] FIG. 4B is a cross-sectional view depicting an implant having a cavity formed in the second member, in accordance with an embodiment of the present invention.
[0042] FIG. 4C is a partially enlarged view taken about circle IV(C) of FIG. 4B depicting a cavity in the second member and a bore in the third member of an implant, in accordance with an embodiment of the present invention.
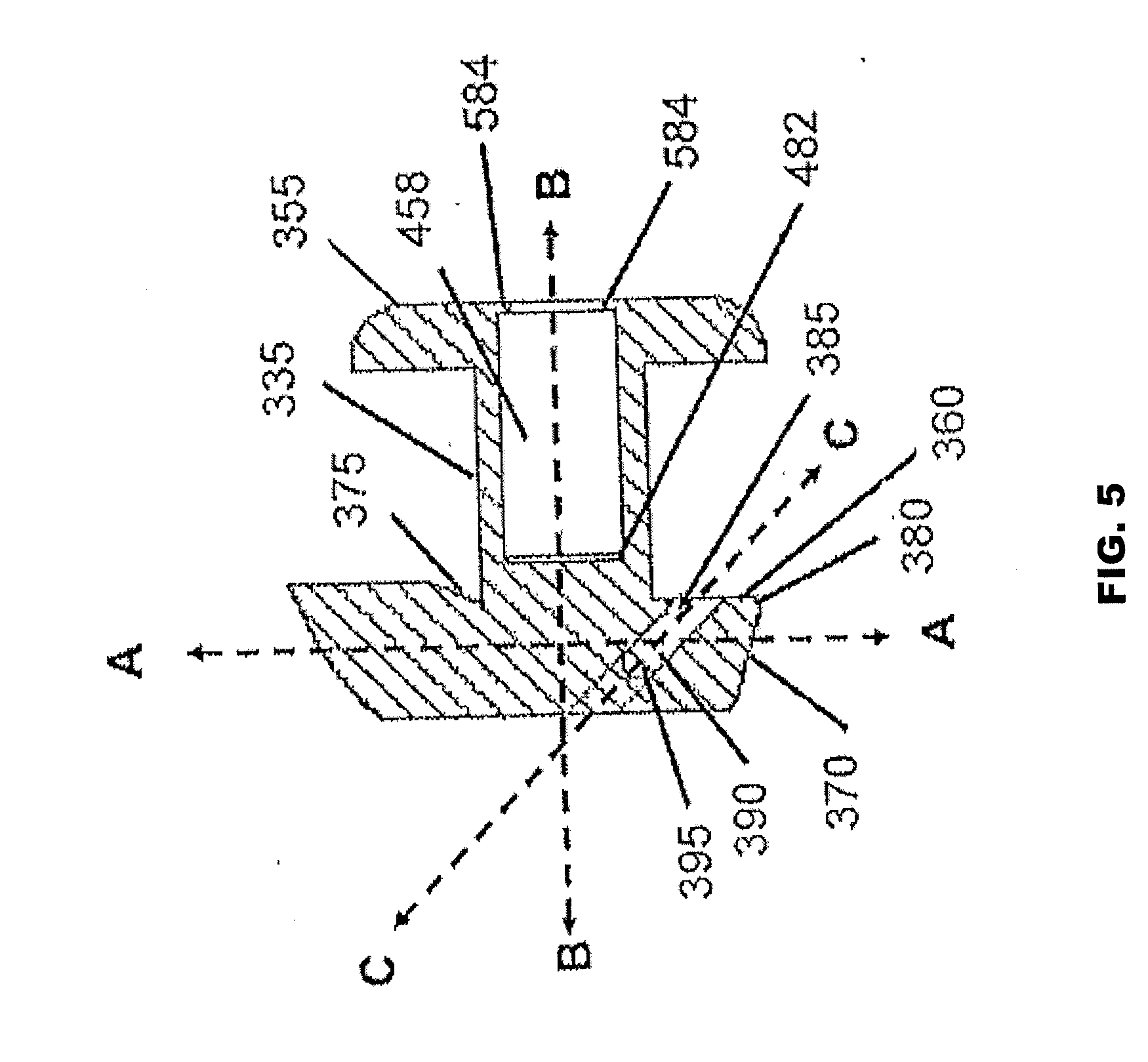
[0043] FIG. 5 provides a partial cross-sectional view of an implant in accordance with one embodiment of the present invention.
[0044] FIG. 6 provides a partial cross-section view of an implant in accordance with another embodiment of the present invention.
[0045] FIG. 7 depicts engagement of an insertion tool with an implant in accordance with an embodiment of the present invention.
[0046] FIG. 8A provides initial retention data for various exemplary implants in accordance with various embodiments of the present invention.
[0047] FIG. 8B lists retention data for various exemplary implants over one day, in accordance with various embodiments of the present invention.
[0048] FIG. 8C lists retention data for various exemplary implants over one week, in accordance with various embodiments of the present invention.
[0049] FIG. 8D lists retention data for various exemplary implants over two weeks, in accordance with various embodiments of the present invention.
[0050] FIG. 8E lists retention data for various exemplary implants over four weeks, in accordance with various embodiments of the present invention.
[0051] FIG. 8F lists retention data for various exemplary implants over eight weeks, in accordance with various embodiments of the present invention.
[0052] FIG. 8G lists retention data for various exemplary implants over twelve weeks, in accordance with various embodiments of the present invention.
[0053] FIG. 9 is a plot comparing retention rates of an implant of the invention (lower punctum) with a commercial implant (upper punctum). The two implants are implanted in the same eye of the patient.
[0054] FIG. 10A illustrates a side view of a commercial implant used for the comparison studies herein.
[0055] FIG. 10B illustrates a top view of the commercial implant used for the comparison studies herein.
[0056] FIG. 10C is a cross-sectional view taken about line A-A of FIG. 10A illustrating a modified cavity formed in the commercial implant for the comparison studies herein.
[0057] FIG. 10D is a partially enlarged view taken about circle B of FIG. 10C illustrating a lip at an opening of the modified cavity in the commercial implant for the comparison studies herein.
[0058] FIG. 11 provides the baseline demographics for the comparison studies herein.
[0059] FIG. 12 lists retention data for various exemplary implants over four weeks, in accordance with various embodiments of the present invention.
[0060] FIG. 13 illustrates mean intraocular pressure (IOP) change from baseline during treatment with a sustained release ophthalmic drug delivery system according to an embodiment of the present invention over a four-week period.
[0061] FIG. 14 illustrates percentage of subjects achieving categorical absolute intraocular pressure (IOP) reduction from baseline during treatment with the sustained release ophthalmic drug delivery system according to an embodiment of the present invention over the four-week period.
[0062] FIG. 15 illustrates percent change in IOP from baseline during treatment with a sustained release ophthalmic drug delivery system according to an embodiment of the present invention over a four-week period.
[0063] FIG. 16 illustrates the lack of dose dependency of intraocular pressure reduction when latanoprost is administered from a single punctal implant.
[0064] FIG. 17 illustrates the dosages of latanoprost delivered by the punctal implants of FIG. 16.
[0065] FIG. 18 is a graphical illustration comparing change in IOP from baseline during treatment in the GLAU 11 and GLAU 12 Studies with a sustained release opthalmic drug delivery system according to an embodiment of the present invention. The data marked (.box-solid.) are for a punctal plug with an unoptimized drug core. 141 .mu.g, the maximum dosage, is administered by upper (46 .mu.g) and lower (95 .mu.g) plugs. The data marked (.diamond-solid.) are for a punctal plug modified to enhance insertion and retention. The maximum dosage, 141 .mu.g, is administered by upper (46 .mu.g) and lower (95 .mu.g) plugs. The results show a comparable, in two studies, sustained reduction in IOP at week 4 of more than 5 mmHg. N=number of eyes.
[0066] FIG. 19 is a graphical illustration comparing change in IOP from baseline during treatment with a sustained release ophthalmic drug delivery system according to an embodiment of the present invention. The data marked (.diamond-solid.) are for a punctal plug modified to enhance insertion and retention. The maximum dosage, 95 .mu.g, is administered by an upper (95 .mu.g) plug. The lower plug is a blank. The data marked (.box-solid.) are for a punctal plug modified to enhance insertion and retention. The maximum dosage, 95 .mu.g, is administered by an upper (95 .mu.g) plug. The lower plug is open. The data marked (.tangle-solidup.) are for a punctal plug modified to enhance insertion and retention. The maximum dosage, 95 .mu.g, is administered by lower (95 .mu.g) plug. The upper plug is a blank
[0067] FIG. 20 is a graphical illustration comparing change in IOP from baseline during treatment with a sustained release ophthalmic drug delivery system according to an embodiment of the present invention. The data marked (.diamond-solid.) are for a punctal plug modified to enhance insertion and retention. The maximum dosage administered is 95 .mu.g. The data marked (.box-solid.) are for a punctal plug modified to enhance insertion and retention. The maximum dosage adminstered is 141 .mu.g. The data marked (.tangle-solidup.) are for a punctal plug modified to enhance insertion and retention. The maximum dosage administered is 190 .mu.g.
[0068] FIG. 21 is a graphical illustration of the five treatment arms of GLAU 12 and GLAU 13 (Ex. 5 and 6). N=number of subjects
[0069] FIG. 22 is a graphical illustration of the two treatment arms for GLAU 12 Addendum exploring the effect of repeat plug placement. N=number of subjects
[0070] FIG. 23 lists a summary of change in IOP from baseline (mmHg) in the GLAU 12 and GLAU 13 studies for both intent to treat (ITT) groups. N=number of eyes
[0071] FIG. 24 is a graphical illustration of the reduction in IOP (mmHg) for the All IOP ITT group of the GLAU 12 study from day 1 to week 12. N=number of eyes.
[0072] FIG. 25 is a graphical illustration of the reduction in IOP (mmHg) for the second ITT group (IOP excluded after first plug loss/removal) of the GLAU 12 study from day 1 to week 12. N=number of eyes.
[0073] FIG. 26 is a graphical illustration of the reduction in IOP (mmHg) for both ITT groups in the GLAU 12 addendum study during course 2 (an additional 8 weeks after the 12 week main study). N=number of eyes.
[0074] FIG. 27 is a graphical illustration of the reduction in IOP (mmHg) for the All IOP ITT group of the GLAU 13 study from day 1 to week 12. The data indicates that the effect of latanoprost and the reduction in IOP may be influenced by the plug position. N=number of eyes.
[0075] FIG. 28 is a graphical illustration of the reduction in IOP (mmHg) for the second ITT group (IOP excluded after first plug loss/removal) of the GLAU 13 study from day 1 to week 12. The data indicates that the effect of latanoprost and the reduction in IOP may be influenced by the plug position. N=number of eyes.
[0076] FIG. 29 is a graphical illustration of the change in IOP (mmHg) from baseline over 12 weeks showings the percentage of eyes with a better than 5 mmHg decrease in IOP for the All IOP ITT group of GLAU 12. N=number of eyes.
[0077] FIG. 30 is a graphical illustration of the change in IOP (mmHg) from baseline over 12 weeks showings the percentage of eyes with a better than 5 mmHg decrease in IOP for the second ITT group (IOP excluded after first plug loss/removal) of GLAU 12. N=number of eyes.
[0078] FIG. 31 is a graphical illustration of the change in IOP (mmHg) from baseline over 12 weeks showings the percentage of eyes with a better than 5 mmHg decrease in IOP for the All IOP ITT group of GLAU 13. N=number of eyes.
[0079] FIG. 32 is a graphical illustration of the change in IOP (mmHg) from baseline over 12 weeks showings the percentage of eyes with a better than 5 mmHg decrease in IOP for the second ITT group (IOP excluded after first plug loss/removal) of GLAU 13. N=number of eyes.
[0080] FIG. 33 is a list and description of the plug designs used in the GLAU 11, 12 and 13 studies.
[0081] FIG. 34 is a graphical illustration of the upper and lower plug retention by eye represented as a percentage at each time point from day 1 to week 12 for the plugs used in the GLAU 12 study.
[0082] FIG. 35 is a table listing the upper and lower plug retention by eye represented as a percentage at each time point from day 1 to week 12 for the plugs used in the GLAU 12 study. N=number of eyes.
[0083] FIG. 36 is a graphical illustration of the upper and lower plug retention by eye represented as a percentage at each time point for Course 2 (8 weeks) and Course 3 (4 weeks) for the plugs used in the GLAU 12 addendum study.
[0084] FIG. 37 is a graphical illustration of the upper and lower plug retention by eye represented as a percentage at each time point from day 1 to week 12 for the plugs used in the GLAU 13 study.
[0085] FIG. 38 is a table listing the upper and lower plug retention by eye represented as a percentage at each time point from day 1 to week 12 for the plugs used in the GLAU 13 study. N=number of eyes.
[0086] FIG. 39 is a table representing the upper plug retention by eye represented as a percentage in 4 week blocks (0-4 weeks, 4-8 weeks and 8-12 weeks) for the plugs used in the GLAU 12 and GLAU 13 studies
DETAILED DESCRIPTION OF THE INVENTION
A) Introduction
[0087] In various embodiments, the present invention is directed to the treatment of ocular diseases such as Glaucoma or ocular hypertension. In certain embodiments, the invention includes the use of an implant that comprises a sustained release formulation of a therapeutic agent of use in treating the disease. The implant is configured to deliver a therapeutically effective amount of the therapeutic agent to the eye during the period that it is implanted in the eye (e.g. the treatment period). In an exemplary embodiment, the disease is glaucoma and the therapeutic agent is a prostaglandin or derivative thereof. In certain embodiments, the implant is a punctual plug configured for insertion through a human lacrimal punctum into a corresponding lacrimal canaliculus and retention in the canaliculus. In an exemplary embodiment, the sustained release formulation of the therapeutic agent is released over a period of from about 4 weeks to about 12 weeks in a therapeutic dose sufficient to reduce intraocular pressure of the eye. In various embodiments, the therapeutic dose of the agent is sufficient to decrease intraocular pressure by at least 4 mm Hg from baseline (e.g., "normal").
[0088] In certain embodiments is provided a method for treating a patient diagnosed with Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye. In this instance lacrimal implants are provided for insertion into the upper and/or lower punctum of the eye. Each lacrimal implant comprises a sustained release formulation of a therapeutic agent for treating OAG and/or OH, wherein the sustained release formulation can be released in a therapeutically effective amount for at least 4 weeks and up to 12 weeks or longer. In one embodiment, the lacrimal implant is inserted at least in the upper punctum. In one aspect this therapeutically effective agent is latanoprost. In this instance of treating OAG and/or OH the IOP is reduced. In an exemplary embodiment, the methods of the invention provide a reduction in IOP of at least about 4 mm Hg, at least about 5 mm Hg, at least about 6 mm Hg or at least about 7 mm Hg from baseline during the treatment period.
[0089] In certain embodiments, a method for treating a patient diagnosed with Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye is provided wherein a first lacrimal implant comprising a sustained release formulation of the therapeutic agent is inserted into a upper or lower punctum and a second lacrimal implant that does not comprise the therapeutic agent is inserted into the open punctum of the eye (i.e. the upper or lower punctum that does not contain the first lacrimal implant). The second lacrimal implant is also referred to herein as a "blank" implant. In one embodiment the therapeutic agent is released in a therapeutically effective dose from the first lacrimal implant on a sustained release basis over at least four (4) weeks. In another aspect, the therapeutic agent is released in a therapeutically effective dose from the first lacrimal implant on a sustained release basis over at least twelve (12) weeks.
[0090] In certain other embodiments, a method for treating a patient diagnosed with Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye is provided wherein the IOP of the eye is measured to obtain a baseline IOP before treatment and wherein a lacrimal implant comprising a sustained release formulation is inserted into a punctum. In exemplary embodiments the IOP is reduced by at least 5.5 mm Hg from baseline at week 6, reduced by at least 4.0 mm Hg from baseline at week 12, or reduced by at least 5.0 mm Hg from baseline at week 12.
[0091] In an exemplary embodiment, the method of the invention utilizes latanoprost-eluting punctal implants. Previous methods of delivering latanoprost to the eye using a latanoprost-eluting punctal implant have met with varied and minimal success. For example, as show in FIG. 16, in an eye implanted with a single latanoprost-eluting plug in the lower punctum, the reduction in IOP is minimal, and is substantially identical across a range of latanoprost loadings: from 3.5 .mu.g to 95 .mu.g, the IOP does not decrease even though more latanoprost is being delivered by the plugs with higher latanoprost loading. See, FIG. 17. Thus, it is surprising that the methods of the present invention, in which either an eye has a latanoprost-eluting punctal implant in both the upper and lower punctum, a blank and latanoprost-eluting punctual implant in either the upper and lower punctum, or in certain instances a latanoprost-eluting punctal implant in the upper punctum and no implant in the bottom punctum, would yield a statistically significant reduction in IOP after about two weeks. See Example 6 and Table 8
[0092] Also disclosed herein are exemplary structures of ocular implants of use in the methods of the invention for treating various diseases and disorders. Exemplary structures include lacrimal implants for at least partial insertion through the lacrimal punctum and into its associated canaliculus. Various embodiments further provide an insertion tool for placing a lacrimal implant into a lacrimal punctum. Also disclosed herein are exemplary implants including therapeutic agents incorporated throughout the device, within one or more section of the device, or in a therapeutic agent core, e.g., a localized therapeutic agent core. The devices of the invention are of use for treating various diseases.
[0093] In the various embodiments of methods of the invention, implanting a lacrimal implant of the invention through the lacrimal punctum and into its associated canaliculus, in various embodiments, inhibits or blocks tear flow therethrough. In various embodiments, a device inhibiting or blocking tear flow is of use to treat dry eye. In an exemplary embodiment, the insertion of the lacrimal implant allows for the delivery of a therapeutic agent. In various embodiments, the delivery is sustained delivery. Exemplary therapeutic agents incorporated into the implants of the invention are of use to treat the eye, or they can be of use more broadly systemic therapies. For example, using a device of the invention, the therapeutic agent can be delivered to a nasal passage, to an inner ear system, or to other passages or systems for treatment of various diseases including, but not limited to, eye infection, eye inflammation, glaucoma, other ocular disease, other ocular disorder, a sinus or allergy disorder, dizziness or a migraine. The devices of the invention are of use for systemic delivery of one or more therapeutic agents in an amount having therapeutic efficacy.
[0094] Those of ordinary skill in the art will understand that the following detailed description of the present invention is illustrative only and is not intended to be in any way limiting. Other embodiments of the present invention will readily suggest themselves to such skilled persons having benefit of this disclosure. Reference will now be made in detail to implementations of the present invention as illustrated in the accompanying drawings. The same reference indicators will be used throughout the drawings and the following detailed description to refer to the same or like parts.
B) Definitions
[0095] As used herein, the terms "a" or "an" are used, as is common in patent documents, to include one or more than one, independent of any other instances or usages of "at least one" or "one or more."
[0096] As used herein, the term "or" is used to refer to a nonexclusive or, such that "A or B" includes "A but not B," "B but not A," and "A and B," unless otherwise indicated.
[0097] As used herein, the term "about" is used to refer to an amount that is approximately, nearly, almost, or in the vicinity of being equal to or is equal to a stated amount, e.g., the state amount plus/minus about 5%, about 4%, about 3%, about 2% or about 1%.
[0098] As used herein, an "axis" refers to a general direction along which a member extends. According to this definition, the member is not required to be entirely or partially symmetric with respect to the axis or to be straight along the direction of the axis. Thus, in the context of this definition, any member disclosed in the present application characterized by an axis is not limited to a symmetric or a straight structure.
[0099] In this document, the term "proximal" refers to a location relatively closer to the cornea of an eye, and the term "distal" refers to a location relatively further from the cornea and inserted deeper into a lacrimal canaliculus.
[0100] In the appended claims, the terms "including" and "in which" are used as the plain-English equivalents of the respective terms "comprising" and "wherein." Also, in the following claims, the terms "including" and "comprising" are open-ended, that is, a system, assembly, device, article, or process that includes elements in addition to those listed after such a term in a claim are still deemed to fall within the scope of that claim. Moreover, in the following claims, the terms "first," "second," and "third," etc. are used merely as labels, and are not intended to impose numerical requirements on their objects.
[0101] As used herein, the term "adverse event" refers to any undesirable clinical event experienced by a patient undergoing a therapeutic treatment including a drug and/or a medical device, whether in a clinical trial or a clinical practice. Adverse events include a change in the patient's condition or laboratory results, which has or could have a deleterious effect on the patient's health or well-being. For example, adverse events include but are not limited to: device malfunction identified prior to placement, device malposition, device malfunction after placement, persistent inflammation, endophthalmitis, corneal complications (corneal edema, opacification, or graft decompensation), chronic pain, iris pigmentation changes, conjunctival hyperemia, eyelash growth (increased length, thickness, pigmentation, and number of lashes), eyelid skin darkening, intraocular inflammation (iritis/uveitis), macular edema including cystoid macular edema, blurred vision, burning and stinging, foreign body sensation, itching, punctate epithelial keratopathy, dry eye, excessive tearing, eye pain, lid crusting, lid discomfort/pain, lid edema, lid erythema, photophobia, VA decrease, conjunctivitis, diplopia, discharge from the eye, retinal artery embolus, retinal detachment, vitreous hemorrhage from diabetic retinopathy, upper respiratory tract infection/cold/flu, chest pain/angina pectoris, muscle/joint/back pain, and rash/allergic skin reaction, eye pruritus, increase in lacrimation, ocular hyperemia and punctate keratitis. In an exemplary embodiment, use of the device and method of the invention results in one or more of: (i) occurrence of fewer adverse events; or (ii) adverse events of less severity, than those occurring with the use of a therapeutic agent in drop form, e.g., when the therapeutic agent is administered via drops in essentially the same unit dosage as that delivered by a device as set forth herein.
[0102] As used herein, the phrase "consisting essentially of" limits a composition to the specified materials or steps and those additional, undefined components that do not materially affect the basic and novel characteristic(s) of the composition.
[0103] As used herein, the term "continuous" or "continuously" means essentially unbroken or uninterrupted. For example, continuously administered active agents are administered over a period of time essentially without interruption.
[0104] As used herein, the term "diameter" encompasses a broad meaning. For example, with respect to a member having a circular cross section, the term "diameter" has the conventional meaning and refers to a straight line through the center of the circle connecting two points on the circumference. When the cross section is not a circle, the term "diameter" in the present disclosure refers to the characteristic diameter of the cross section. The "characteristic diameter" refers to the diameter of a circle that has the same surface area as the cross section of the element. In the present application, "diameter" is interchangeable with "characteristic diameter."
[0105] As used herein, the term "eye" refers to any and all anatomical tissues and structures associated with an eye. The eye is a spherical structure with a wall having three layers: the outer sclera, the middle choroid layer and the inner retina. The sclera includes a tough fibrous coating that protects the inner layers. It is mostly white except for the transparent area at the front, the cornea, which allows light to enter the eye. The choroid layer, situated inside the sclera, contains many blood vessels and is modified at the front of the eye as the pigmented iris. The biconvex lens is situated just behind the pupil. The chamber behind the lens is filled with vitreous humour, a gelatinous substance. The anterior and posterior chambers are situated between the cornea and iris, respectively and filled with aqueous humour. At the back of the eye is the light-detecting retina. The cornea is an optically transparent tissue that conveys images to the back of the eye. It includes avascular tissue to which nutrients and oxygen are supplied via bathing with lacrimal fluid and aqueous humour as well as from blood vessels that line the junction between the cornea and sclera. The cornea includes one pathway from the permeation of drugs into the eye. Other anatomical tissue structures associated with the eye include the lacrimal drainage system, which includes a secretory system, a distributive system and an excretory system. The secretory system comprises secretors that are stimulated by blinking and temperature change due to tear evaporation and reflex secretors that have an efferent parasympathetic nerve supply and secrete tears in response to physical or emotional stimulation. The distributive system includes the eyelids and the tear meniscus around the lid edges of an open eye, which spread tears over the ocular surface by blinking, thus reducing dry areas from developing.
[0106] As used herein, the term "implant" refers to a structure that can be configured to contain or be impregnated with a drug, for example via a drug core or a drug matrix, such as those as disclosed in this patent document and in WO 07/115,261, which is herein incorporated by reference in its entirety, and which is capable of releasing a quantity of active agent, such as latanoprost or other intraocular pressure-reducing therapeutic agent(s), into tear fluid for a sustained release period of time when the structure is implanted at a target location along the path of the tear fluid in the patient. The terms "implant," "plug," "punctal plug," and "punctal implant" are meant herein to refer to similar structures. Likewise, the terms "implant body" and "plug body" are meant herein to refer to similar structures. The implants described herein may be inserted into the punctum of a subject, or through the punctum into the canaliculus. The implant may be also the drug core or drug matrix itself, which is configured for insertion into the punctum without being housed in a carrier such as a punctal implant occluder, for example having a polymeric component and a latanoprost or other intraocular pressure-reducing therapeutic agent(s) component with no additional structure surrounding the polymeric component and latanoprost or other intraocular pressure-reducing therapeutic agent(s) component.
[0107] As used in exemplary embodiments herein, "loss of efficacy" (LoE) is defined as an IOP increase to baseline (post-washout) IOP in either or both eyes while wearing a latanoprost punctal plug delivery system (L-PPDS) continuously from Day 0. Subjects were followed for at least 4 weeks before the subject could complete the study due to LoE and LoE was confirmed at 2 sequential visits.
[0108] As used herein, a "pharmaceutically acceptable vehicle" is any physiologically acceptable vehicle known to those of ordinary skill in the art useful in formulating pharmaceutical compositions. Suitable vehicles include polymeric matrices, sterile distilled or purified water, isotonic solutions such as isotonic sodium chloride or boric acid solutions, phosphate buffered saline (PBS), propylene glycol and butylene glycol. Other suitable vehicular constituents include phenylmercuric nitrate, sodium sulfate, sodium sulfite, sodium phosphate and monosodium phosphate. Additional examples of other suitable vehicle ingredients include alcohols, fats and oils, polymers, surfactants, fatty acids, silicone oils, humectants, moisturizers, viscosity modifiers, emulsifiers and stabilizers. The compositions may also contain auxiliary substances, i.e. antimicrobial agents such as chlorobutanol, parabans or organic mercurial compounds; pH adjusting agents such as sodium hydroxide, hydrochloric acid or sulfuric acid; and viscosity increasing agents such as methylcellulose. An exemplary final composition is sterile, essentially free of foreign particles, and has a pH that allows for patient comfort and acceptability balanced with a pH that is desirable for optimum drug stability. An exemplary "pharmaceutically acceptable vehicle is an "ophthalmically acceptable vehicle" as used herein refers to any substance or combination of substances which are non-reactive with the compounds and suitable for administration to patient. In an exemplary embodiment, the vehicle is an aqueous vehicle suitable for topical application to the patient's eyes. In various embodiments, the vehicle further includes other ingredients which may be desirable to use in the ophthalmic compositions of the present invention include antimicrobials, preservatives, co-solvents, surfactants and viscosity building agents.
[0109] In various embodiments, the "pharmaceutically acceptable vehicle" includes more than one therapeutic agent.
[0110] As used herein, the term "punctum" refers to the orifice at the terminus of the lacrimal canaliculus, seen on the margins of the eyelids at the lateral extremity of the lacus lacrimalis. Puncta (plural of punctum) function to reabsorb tears produced by the lacrimal glands. The excretory part of the lacrimal drainage system includes, in flow order of drainage, the lacrimal puncta, the lacrimal canaliculi, the lacrimal sac and the lacrimal duct. From the lacrimal duct, tears and other flowable materials drain into a passage of the nasal system. The lacrimal canaliculi include an upper (superior) lacrimal canaliculus and a lower (inferior) lacrimal canaliculus, which respectively terminate in an upper and lower lacrimal punctum. The upper and lower punctum are slightly elevated at the medial end of a lid margin at the junction of the ciliary and lacrimal portions near a conjunctival sac. The upper and lower punctum are generally round or slightly ovoid openings surrounded by a connective ring of tissue. Each of the puncta leads into a vertical portion of their respective canaliculus before turning more horizontal at a canaliculus curvature to join one another at the entrance of the lacrimal sac. The canaliculi are generally tubular in shape and lined by stratified squamous epithelium surrounded by elastic tissue, which permits them to be dilated.
[0111] The terms "subject" and "patient" refer to animals such as mammals, including, but not limited to, primates (e.g., humans), cows, sheep, goats, horses, dogs, cats, rabbits, rats, mice and the like. In many embodiments, the subject or patient is a human.
[0112] An "intraocular pressure-reducing therapeutic agent" can comprise a drug and may be any of the following or their equivalents, derivatives or analogs, including anti-glaucoma medications (e.g. adrenergic agonists, adrenergic antagonists (beta blockers), carbonic anhydrase inhibitors (CAIs, systemic and topical), therapeutic agent(s) such as prostaglandins, antiprostaglandins, prostaglandin precursors, including antiglaucoma drugs including beta-blockers such as timolol, betaxolol, levobunolol, atenolol (see U.S. Pat. No. 4,952,581); adrenergic agonists including clonidine derivatives, such as apraclonidine or brimonidine (see U.S. Pat. No. 5,811,443); and prostaglandin analogues such as bimatoprost, travoprost, tafluprost, latanoprost, etc. In an exemplary embodiment, the therapeutic agent is already marketed for glaucoma, and commercially available preparations thereof can be used. Further therapeutic agents include carbonic anhydrase inhibitors such as acetazolamide, dorzolamide, brinzolamide, methazolamide, dichlorphenamide, diamox; and the like.
[0113] The term "topical" refers to any surface of a body tissue or organ. A topical formulation is one that is applied to a body surface, such as an eye, to treat that surface or organ. Topical formulations as used herein also include formulations that can release therapeutic agents into the tears to result in topical administration to the eye.
[0114] As used herein, the term "treating" or "treatment" of a state, disease, disorder, injury or condition as used herein is understood to mean one or more of (1) preventing or delaying the appearance of clinical symptoms of the state, disease, disorder, injury or condition developing in a mammal that may be afflicted with or predisposed to the state, disease, disorder, injury or condition but does not yet experience or display clinical or subclinical symptoms of the state, disease, disorder, injury or condition, (2) inhibiting the state, disease, disorder, injury or condition, i.e., arresting or reducing the development of the disease or at least one clinical or subclinical symptom thereof, or (3) relieving the state, disease, disorder, injury or condition, i.e., causing regression of the state, disease, disorder, injury or condition or at least one of its clinical or subclinical symptoms. In an exemplary embodiment, the present invention provides a method of treating glaucoma or ocular hypertension including contacting an effective intraocular pressure reducing amount of a composition with the eye in order to reduce eye pressure and to maintain the pressure on a reduced level for a sustained period, e.g., at least about 1, 2, 3, 4, 5, 6, 7 8, 9, 10, 11 or 12 weeks.
[0115] The term "delivering", as used herein, shall be understood to mean providing a therapeutically effective amount of a pharmaceutically active agent to a particular location within a host causing a therapeutically effective concentration of the pharmaceutically active agent at the particular location.
[0116] As used herein, the term "diameter" encompasses a broad meaning. For example, with respect to a member having a circular cross section, the term "diameter" has the conventional meaning and refers to a straight line through the center of the circle connecting two points on the circumference. When the cross section is not a circle, the term "diameter" in the present disclosure refers to the characteristic diameter of the cross section. The "characteristic diameter" refers to the diameter of a circle that has the same surface area as the cross section of the element. In the present application, "diameter" is interchangeable with "characteristic diameter."
[0117] Some embodiments of the invention provide the use of latanoprost or another active agent or agents for treatment of diabetic retinopathy, uveitis, intraocular inflammation, keratitis, dry eye, macular edema including cystoid macular edema, infection, macular degeneration, blurred vision, herpetic conjunctivitis, blepharitis, retinal or choroidal neovascularizaton, and other proliferative eye diseases. In some embodiments, the invention provides the use of an anti-glaucoma drug for treatment of the above diseases. In certain embodiments, the use of a prostaglandin or prostaglandin analogue for treatment of the above diseases is provided.
[0118] "Prostaglandin derivatives", as used herein refers to compounds having the basic prostaglandin structure of 20 carbon atoms and a 5-carbon ring. Exemplary prostaglandin derivatives of use in the present invention are of the PGI.sub.2, PGE.sub.2 and PGF.sub.2.alpha. types. The structure can be augmented by incorporating or eliminating functional groups (e.g., HO, carbonyl, ether, ester, carboxylic acid, halide) or by adding carbon atom-based radicals (e.g., Me, Et, i-Pr, etc.). See for example, U.S. Pat. No. 7,910,767. In some embodiments, the prostaglandin derivative is a derivative of PGA, PGB, PGD, PGE and PGF, in which the omega chain has been modified with the common feature of containing a ring structure. See, U.S. Pat. No. 5,296,504. The prostaglandin derivatives of use in the invention are synthesized de novo or derived from modification of naturally occurring prostaglandins.
C) Drug Delivery System
[0119] Applicants herein disclose a method for treating open angle glaucoma (OAG) and/or ocular hypertension (OH) in an eye of a patient utilizing a lacrimal implant comprising a sustained release formulation to deliver the therapeutic agent to the eye. The treatment of these eye diseases relies on a drug delivery system for administering the therapeutic agent, wherein the therapeutic agent may be a known drug for reducing IOP or a newly developed drug. The drug delivery system comprises 1) the therapeutic agent, 2) the lacrimal implant and 3) sustained release formulations while taking into account the specific disease being treated.
[0120] Applicants provide herein, for the first time, methods for treating OAG and/or OH wherein a therapeutically effective dose of the therapeutic agent (e.g. latanoprost) is administered from the present lacrimal implants over the treatment period (e.g. 4-12 weeks) wherein the IOP is reduced over the treatment period by a clinically meaningful amount (e.g. about 5 mm Hg from baseline.) In this instance, no additional treatment, except for the therapeutic agent eluted from the implants, was needed to reduce IOP by a clinically meaningful amount.
[0121] For ease of understanding the invention, the drug delivery system and each of the components will be described in detail followed by methods and clinical applications for treating OAG and/or OH wherein intraocular pressure (IOP) is reduced.
1) Therapeutic Agents
[0122] Generally, pharmaceutically active agents or drugs useful in the methods of the present invention can be any compound, composition of matter, or mixtures thereof that can be delivered from an implant, such as those described herein, to produce a beneficial and useful result to, for example, the eye, especially an agent effective in obtaining a desired local or systemic physiological or pharmacological effect.
[0123] Examples of such agents include, but are not limited to, anesthetics and pain killing agents such as lidocaine and related compounds, benzodiazepam and related compounds and the like; anti-cancer agents such as 5-fluorouracil, adriamycin and related compounds and the like; anti-fungal agents such as fluconazole and related compounds and the like; anti-viral agents such as trisodium phosphomonoformate, trifluorothymidine, acyclovir, ganciclovir, DDI, AZT and the like; cell transport/mobility impending agents such as colchicine, vincristine, cytochalasin B and related compounds and the like; antiglaucoma drugs (e.g. adrenergic agonists, adrenergic antagonists (beta blockers), carbonic anhydrase inhibitors (CAIs, systemic and topical), parasympathomimetics, prostaglandins and hypotensive lipids, and combinations thereof), antimicrobial agent (e.g., antibiotic, antiviral, antiparacytic, antifungal, etc.), a corticosteroid or other anti-inflammatory (e.g., an NSAID or other analgesic and pain management compounds), a decongestant (e.g., vasoconstrictor), an agent that prevents of modifies an allergic response (e.g., an antihistamine, cytokine inhibitor, leucotriene inhibitor, IgE inhibitor, immunomodulator), a mast cell stabilizer, cycloplegic, mydriatic or the like.
[0124] Other agents that can be incorporated into implants of use in the invention include antihypertensives; decongestants such as phenylephrine, naphazoline, tetrahydrazoline and the like; immunological response modifiers such as muramyl dipeptide and related compounds and the like; peptides and proteins such as cyclosporin, insulin, growth hormones, insulin related growth factor, heat shock proteins and related compounds and the like; steroidal compounds such as dexamethasone, prednisolone and related compounds and the like; low solubility steroids such as fluocinolone acetonide and related compounds and the like; carbonic anhydrase inhibitors; diagnostic agents; antiapoptosis agents; gene therapy agents; sequestering agents; reductants such as glutathione and the like; antipermeability agents; antisense compounds; antiproliferative agents; antibody conjugates; antidepressants; blood flow enhancers; antiasthmatic drugs; antiparasiticagents; non-steroidal anti inflammatory agents such as ibuprofen and the like; nutrients and vitamins: enzyme inhibitors: antioxidants; anticataract drugs; aldose reductase inhibitors; cytoprotectants; cytokines, cytokine inhibitors, and cytokin protectants; uv blockers; mast cell stabilizers; anti neovascular agents such as antiangiogenic agents, e.g., matrix metalloprotease inhibitors and the like.
[0125] Representative examples of additional pharmaceutically active agent for use herein include, but are not limited to, neuroprotectants such as nimodipine and related compounds and the like; antibiotics such as tetracycline, chlortetracycline, bacitracin, neomycin, polymyxin, gramicidin, oxytetracycline, chloramphenicol, gentamycin, erythromycin and the like; anti-infectives; antibacterials such as sulfonamides, sulfacetamide, sulfamethizole, sulfisoxazole; nitrofurazone, sodium propionate and the like; antiallergenics such as antazoline, methapyriline, chlorpheniramine, pyrilamine, prophenpyridamine and the like; anti-inflammatories such as hydrocortisone, hydrocortisone acetate, dexamethasone 21-phosphate, fluocinolone, medrysone, methylprednisolone, prednisolone 21-phosphate, prednisolone acetate, fluoromethalone, betamethasone, triminolone and the like; miotics; anti-cholinesterase such as pilocarpine, eserine salicylate, carbachol, di-isopropyl fluorophosphate, phospholine iodine, demecarium bromide and the like; miotic agents; mydriatics such as atropine sulfate, cyclopentolate, homatropine, scopolamine, tropicamide, eucatropine, hydroxyamphetamine and the like; svmpathomimetics such as epinephrine and the like; and prodrugs such as, for example, those described in Design of Prodrugs, edited by Hans Bundgaard, Elsevier Scientific Publishing Co., Amsterdam, 1985. In addition to the foregoing agents, other agents suitable for treating, managing, or diagnosing conditions in a mammalian organism may be entrapped in the copolymer and administered using the drug delivery systems of the current invention. Once again, reference may be made to any standard pharmaceutical textbook such as, for example, Remington's Pharmaceutical Sciences for pharmaceutically active agents.
[0126] Any pharmaceutically acceptable form of the foregoing therapeutically active agent may be employed in the practice of the present invention, e.g., the free base; free acid; pharmaceutically acceptable salts, esters or amides thereof, e.g., acid additions salts such as the hydrochloride, hydrobromide, sulfate, bisulfate, acetate, oxalate, valerate, oleate, palmitate, stearate, laurate, borate, benzoate, lactate, phosphate, tosylate, mesylate, citrate, maleate, fumarate, succinate, tartrate, ascorbate, glucoheptonate, lactobionate, and lauryl sulfate salts and the like; alkali or alkaline earth metal salts such as the sodium, calcium, potassium and magnesium salts and the like; hydrates; enantiomers; isomers; stereoisomers; diastereoisomers; tautomers; polymorphs, mixtures thereof, prodrugs thereof or racemates or racemic mixtures thereof.
[0127] Additional agents that can be used with the present methods utilizing lacrimal implants include, but are not limited to, drugs that have been approved under Section 505 of the United States Federal Food, Drug, and Cosmetic Act or under the Public Health Service Act, some of which can be found at the U.S. Food and Drug Administration (FDA) website http://www.accessdata.fda.gov/scripts/cder/drugsatfda/index. The present lacrimal implants can also be used with drugs listed in the Orange Book, either in paper or in electronic form, which can be found at the FDA Orange Book website (http://www.fda.gov/cder/ob/)), that has or records the same date as, earlier date than, or later date than, the filing date of this patent document. For example, these drugs can include, among others, dorzolamide, olopatadine, travoprost, bimatoprost, latanoprost, cyclosporin, brimonidine, moxifloxacin, tobramycin, brinzolamide, aciclovir timolol maleate, ketorolac tromethamine, prednisolone acetate, sodium hyaluronate, nepafenac, bromfenac, diclofenac, flurbiprofen, suprofenac, binoxan, patanol, dexamethasone/tobramycin combination, moxifloxacin, or acyclovir.
[0128] Further discussion of drugs or other agents can be found in commonly-owned U.S. Patent Application Publication No. 2009/0104248, U.S. Patent Application Publication No. 2010/0274204, and U.S. Patent Application Publication No. 2009/0105749, which are herein incorporated by reference in its entirety.
Prostaglandins
[0129] Prostaglandins are regarded as potent ocular hypertensives; however, evidence accumulated in the last decade shows that some prostaglandins are highly effective ocular hypotensive agents and are ideally suited for the long-term medical management of glaucoma (see, for example, Bito, L. Z. Biological Protection with Prostaglandins Cohen, M. M., ed., Boca Raton, Fla., CRC Press Inc., 1985, pp. 231-252; and Bito, L. Z., Applied Pharmacology in the Medical Treatment of Glaucomas Drance, S. M. and Neufeld, A. H. eds., New York, Grune & Stratton, 1984, pp. 477-505). Such prostaglandins include PGF2.alpha., PGF.sub.1.alpha., PGE.sub.2, and certain lipid-soluble esters, such as C.sub.1 to C.sub.5 alkyl esters, e.g. 1-isopropyl ester, of such compounds.
[0130] Thus, in certain embodiments, the therapeutic agent is a prostaglandin, including derivatives thereof. Prostaglandins are derivatives of prostanoic acid. Various types of prostaglandins are known, depending on the structure and substituents carried on the alicyclic ring of the prostanoic acid skeleton. Further classification is based on the number of unsaturated bonds in the side chains indicated by numerical subscripts after the generic type of prostaglandin (e.g., prostaglandin E.sub.1 (PGE.sub.1), prostaglandin E.sub.2 (PGE.sub.2)), and on the configuration of the substituents on the alicyclic ring indicated by .alpha. or .beta. (e.g. prostaglandin F.sub.2.alpha. (PGF.sub.2.alpha.)). Any of these prostaglandins are of use in the present invention.
[0131] An exemplary therapeutic agent for use in the methods described herein is latanoprost. Latanoprost is a prostaglandin F.sub.2.alpha. analogue. Its chemical name is isopropyl-(Z)-7 [(1R,2R,3R,5S)3,5-dihydroxy-2-[(3R)-3-hydroxy-5-phenylpentyl]cyclopentyl]- -5-heptenoate. Its molecular formula is C.sub.26H.sub.40O.sub.5 and its chemical structure is:
##STR00001##
[0132] Latanoprost is a colorless to slightly yellow oil that is very soluble in acetonitrile and freely soluble in acetone, ethanol, ethyl acetate, isopropanol, methanol and octanol. It is practically insoluble in water.
[0133] Latanoprost is believed to reduce intraocular pressure (IOP) by increasing the outflow of aqueous humor. Studies in animals and man suggest that the main mechanism of action is increased uveoscleral outflow of aqueous fluid from the eyes. Latanoprost is absorbed through the cornea where the isopropyl ester prodrug is hydrolyzed to the acid form to become biologically active. Studies in man indicate that the peak concentration in the aqueous humor is reached about two hours after topical administration.
[0134] Xalatan.RTM. latanoprost ophthalmic solution is a commercially available product indicated for the reduction of elevated IOP in patients with open-angle glaucoma or ocular hypertension. The amount of latanoprost in the commercially available product Xalatan.RTM. is approximately 1.5 micrograms/drop, which is the recommended daily total dose of latanoprost to one eye. As described above, eye drops, though effective, can be inefficient and require multiple applications to maintain the therapeutic benefit. Low patient compliance compounds these effects.
[0135] In various embodiments, the prostaglandin is latanoprost. In an illustrative embodiment, the unit dosage format includes from 40 .mu.g to 100 .mu.g of the therapeutic agent. In an exemplary embodiment, the implant includes about 46 .mu.g or about 95 .mu.g of latanoprost.
[0136] In an exemplary embodiment, the implant of the invention is a member of a pair of implants. In various embodiments, the pair of implants is configured as a unit dosage. In various embodiments, the implant is formatted as a unit dosage of an antiglaucoma agent. In an exemplary embodiment, the antiglaucoma agent is a prostaglandin. In various embodiments, the prostaglandin is latanoprost. In an illustrative embodiment, the unit dosage format includes from 40 .mu.g to 100 .mu.g of the therapeutic agent. In an exemplary embodiment, the unit dosage is 141 .mu.g of latanoprost. In an exemplary embodiment, one implant includes about 46 .mu.g of latanoprost and the other includes about 95 .mu.g of latanoprost. In an exemplary embodiment, the unit dosage is a unit dosage for both eyes, including four implants as described herein.
[0137] In an exemplary embodiment, the implant of the invention is a member of a pair of implants. In various embodiments, the pair of implants is configured as a unit dosage. In various embodiments, the implant is formatted as a unit dosage of an antiglaucoma agent. In an exemplary embodiment, the antiglaucoma agent is a prostaglandin. In various embodiments, the prostaglandin is latanoprost. In an illustrative embodiment, the unit dosage format includes from 40 .mu.g to 100 .mu.g of the therapeutic agent. In an exemplary embodiment, the unit dosage is 190 .mu.g of latanoprost. In an exemplary embodiment, each implant includes about 95 .mu.g of latanoprost. In an exemplary embodiment, the unit dosage is a unit dosage for both eyes, including four implants as described herein.
[0138] In an exemplary embodiment, the implant of the invention is a member of a pair of implants. In various embodiments, the pair of implants is configured as a unit dosage. In various embodiments, the implant is formatted as a unit dosage of an antiglaucoma agent. In an exemplary embodiment, the antiglaucoma agent is a prostaglandin. In various embodiments, the prostaglandin is latanoprost. In an illustrative embodiment, the unit dosage format includes from 40 .mu.g to 100 .mu.g of the therapeutic agent. In an exemplary embodiment, the unit dosage is 95 .mu.g of latanoprost. In an exemplary embodiment, a first implant includes about 95 .mu.g of latanoprost and a second implant does not include latanoprost (e.g. a blank implant). In an exemplary embodiment, the unit dosage is a unit dosage for both eyes, including four implants as described herein.
[0139] In an alternative embodiment, the implant of the invention is a single implant configured as a unit dosage. In various embodiments, the implant is formatted as a unit dosage of an antiglaucoma agent. In an exemplary embodiment, the antiglaucoma agent is a prostaglandin. In various embodiments, the prostaglandin is latanoprost. In an illustrative embodiment, the unit dosage format includes from 40 .mu.g to 100 .mu.g of the therapeutic agent. In an exemplary embodiment, the unit dosage is 95 .mu.g of latanoprost. In an exemplary embodiment, a first implant includes about 95 .mu.g of latanoprost and is inserted into the upper punctum while no implant is inserted into the lower punctum. In an exemplary embodiment, the unit dosage is a unit dosage for both eyes, including two implants as described herein.
[0140] Actual dosage levels of the pharmaceutically active agent(s) in the drug delivery systems of use in the present invention may be varied to obtain an amount of the pharmaceutically active agent(s) that is effective to obtain a desired therapeutic response for a particular system and method of administration. The selected dosage level therefore depends upon such factors as, for example, the desired therapeutic effect, the route of administration, the desired duration of treatment, and other factors. The total daily dose of the pharmaceutically active agent(s) administered to a host in single or divided doses can vary widely depending upon a variety of factors including, for example, the body weight, general health, sex, diet, time and route of administration, rates of absorption and excretion, combination with other drugs, the severity of the particular condition being treated, etc. In certain embodiments, the amounts of pharmaceutically active agent(s) present in the drug delivery systems of the present invention can range from about 0.1% w/w to about 60% w/w. In one embodiment, the amounts of pharmaceutically active agent(s) present in the present drug delivery systems can range from about 1% w/w to about 50% w/w.
[0141] The therapeutic agents are formulated as a sustained release formulation and incorporated into the lacrimal implants. This sustained release formulation may either be in the form of a drug core or dispersed throughout the lacrimal implant. In this instance the lacrimal implant may be saturated and/or impregnated with the therapeutic agent. However, before describing the sustained release formulations the lacrimal implants will first be described in detail.
2) Lacrimal Implants
[0142] The implant can be one of any number of different designs that releases latanoprost or other intraocular pressure-reducing therapeutic agent(s) for a sustained period of time. The disclosures of the following patent documents, which describe example implant structure or processing embodiments for use in the methods of embodiments of the current invention and methods of making those implants, are incorporated herein by reference in their entirety: U.S. Application Ser. No. 60/871,864 (filed Dec. 26, 2006 and entitled Nasolacrimal Drainage System Implants for Drug Therapy); U.S. application Ser. No. 11/695,537 (filed Apr. 2, 2007 and entitled Drug Delivery Methods, Structures, and Compositions for Nasolacrimal System); U.S. U.S. application Ser. No. 12/332,219 (filed Dec. 10, 2008 and entitled Drug Delivery Methods, Structures, and Compositions for Nasolacrimal System); U.S. Application Ser. No. 60/787,775 (filed Mar. 31, 2006 and entitled Nasolacrimal Drainage System Implants for Drug Therapy); U.S. application Ser. No. 11/695,545 (filed Apr. 2, 2007 and entitled Nasolacrimal Drainage System Implants for Drug Therapy); U.S. Application Ser. No. 60/585,287 (filed Jul. 2, 2004 and entitled Treatment Medium Delivery Device and Methods for Delivery of Such Treatment Mediums to the Eye Using Such a Delivery Device); U.S. application Ser. No. 11/571,147 (filed Dec. 21, 2006 and entitled Treatment Medium Delivery Device and Methods for Delivery of Such Treatment Mediums to the Eye Using Such a Delivery Device); U.S. Application Ser. No. 60/970,696 (filed Sep. 7, 2007 and entitled Expandable Nasolacrimal Drainage System Implants); U.S. Application Ser. No. 60/974,367 (filed Sep. 21, 2007 and entitled Expandable Nasolacrimal Drainage System Implants); U.S. Application Ser. No. 60/970,699 (filed Sep. 7, 2007 and entitled Manufacture of Drug Cores for Sustained Release of Therapeutic Agents); U.S. Application Ser. No. 60/970,709 (filed Sep. 7, 2007 and entitled Nasolacrimal Drainage System Implants for Drug Delivery); U.S. Application Ser. No. 60/970,720 (filed Sep. 7, 2007 and entitled Manufacture of Expandable Nasolacrimal Drainage System Implants); U.S. Application Ser. No. 60/970,755 (filed Sep. 7, 2007 and entitled Prostaglandin Analogues for Implant Devices and Methods); U.S. Application Ser. No. 60/970,820 (filed Sep. 7, 2007 and entitled Multiple Drug Delivery Systems and Combinations of Drugs with Punctal Implants); U.S. Application Ser. No. 61/066,223 (filed Feb. 18, 2008 and entitled Lacrimal Implants and Related Methods); U.S. Application Ser. No. 61/049,347 (filed Apr. 30, 2008 and entitled Lacrimal Implants and Related Methods); U.S. Application Ser. No. 61/033,211 (filed Mar. 3, 2008 and entitled Lacrimal Implants and Related Methods); U.S. Application Ser. No. 61/049,360 (filed Apr. 30, 2008 and entitled Lacrimal Implants and Related Methods); U.S. Application Ser. No. 61/052,595 (filed May 12, 2008 and entitled Lacrimal Implants and Related Methods); U.S. Application Ser. No. 61/075,309 (filed Jun. 24, 2008 and entitled Lacrimal Implants and Related Methods); U.S. Application Ser. No. 61/154,693 (filed Feb. 23, 2009 and entitled Lacrimal Implants and Related Methods); U.S. Application Ser. No. 61/209,036 (filed Mar. 2, 2009 and entitled Lacrimal Implants and Related Methods); U.S. Application Ser. No. 61/209,630 (filed Mar. 9, 2009 and entitled Lacrimal Implants and Related Methods); U.S. Application Ser. No. 61/036,816 (filed Mar. 14, 2008 and entitled Lacrimal Implants and Related Methods); U.S. Application Ser. No. 61/271,862 (filed Jul. 27, 2009 and entitled Lacrimal Implants and Related Methods); U.S. Application Ser. No. 61/252,057 (filed Oct. 15, 2009 and entitled Lacrimal Implants and Related Methods); U.S. application Ser. No. 12/710,855 (filed Feb. 23, 2010 and entitled Lacrimal Implants and Related Methods); U.S. Application Ser. No. 60/871,867 (filed Dec. 26, 2006 and entitled Drug Delivery Implants for Inhibition of Optical Defects); U.S. application Ser. No. 14/521,543 (filed Dec. 31, 2009 and entitled Drug Delivery Implants for Inhibition of Optical Defects); U.S. Application Ser. No. 61/052,068 (filed May 9, 2008 and entitled Sustained Release Delivery of Latanoprost to Treat Glaucoma); U.S. Application Ser. No. 61/052,113 (filed May 9, 2008 and entitled Sustained Release Delivery of Latanoprost to Treat Glaucoma); U.S. Application Ser. No. 61/108,777 (filed Oct. 27, 2008 and entitled Sustained Release Delivery of Latanoprost to Treat Glaucoma); U.S. application Ser. No. 12/463,279 (filed May 8, 2009 and entitled Sustained Release Delivery of Active Agents to Treat Glaucoma and Ocular Hypertension); U.S. Application Ser. No. 61/049,337 (filed Apr. 30, 2008 and entitled Lacrimal Implants and Related Methods); U.S. application Ser. No. 12/432,553 (filed Apr. 29, 2009 and entitled Composite Lacrimal Insert and Related Methods); U.S. Application Ser. No. 61/049,317 (filed Apr. 30, 2008 and entitled Drug-Releasing Polyurethane Lacrimal Insert); U.S. application Ser. No. 12/378,710 (filed Feb. 17, 2009 and entitled Lacrimal Implants and Related Methods); U.S. Application Ser. No. 61/075,284 (filed Jun. 24, 2008 and entitled Combination Treatment of Glaucoma); U.S. application Ser. No. 12/490,923 (filed Jun. 24, 2009 and entitled Combination Treatment of Glaucoma); U.S. Application Ser. No. 61/134,271 (filed Jul. 8, 2008 and entitled Lacrimal Implant Body Including Comforting Agent); U.S. application Ser. No. 12/499,605 (filed Jul. 8, 2009 and entitled Lacrimal Implant Body Including Comforting Agent); U.S. Application Ser. No. 61/057,246 (filed May 30, 2008 and entitled Surface Treatment of Implants and Related Methods); U.S. Application Ser. No. 61/132,927 (filed Jun. 24, 2008 and entitled Surface Treated Implantable Articles and Related Methods); U.S. application Ser. No. 12/283,002 (filed Sep. 5, 2008 and entitled Surface Treated Implantable Articles and Related Methods); U.S. application Ser. No. 12/231,989 (filed Sep. 5, 2008 and entitled Lacrimal Implants and Related Methods); U.S. Application Ser. No. 61/049,317 (filed Apr. 30, 2008 and entitled Drug-Releasing Polyurethane Lacrimal Insert); U.S. application Ser. No. 12/231,986 (filed Sep. 5, 2008 and entitled Drug Cores for Sustained Release of Therapeutic Agents); U.S. Application Ser. No. 61/050,901 (filed May 6, 2008 and entitled Punctum Plug Detection); U.S. application Ser. No. 12/231,987 (filed Sep. 5, 2008 and entitled Lacrimal Implant Detection); U.S. Application Ser. No. 61/146,860 (filed Jan. 23, 2009 and entitled Sustained Release Delivery of One or More Anti-Glaucoma Agents); U.S. Application Ser. No. 61/152,909 (filed Feb. 16, 2009 and entitled Sustained Release Delivery of One or More Anti-Glaucoma Agents); U.S. Application Ser. No. 61/228,894 (filed Jul. 27, 2009 and entitled Sustained Release Delivery of One or More Anti-Glaucoma Agents); U.S. Application Ser. No. 61/277,000 (filed Sep. 18, 2009 and entitled Drug Cores for Sustained Ocular Release of Therapeutic Agents); U.S. application Ser. No. 12/692,452 (filed Jan. 22, 2010 and entitled Sustained Release Delivery of One or More Agents); U.S. Application Ser. No. 61/283,100 (filed Nov. 27, 2009 and entitled Lacrimal Implants Including Split and Insertable Drug Core); International Application Serial No. PCT/US2010/058129 (filed Nov. 26, 2010, published as WO 2011/066479 and entitled Lacrimal Implants Including Split and Insertable Drug Core); U.S. Application Ser. No. 61/139,456 (filed Dec. 19, 2008 and entitled Substance Delivering Punctum Implants and Methods); U.S. application Ser. No. 12/643,502 (filed Dec. 21, 2009 and entitled Substance Delivering Punctum Implants and Methods); U.S. application Ser. No. 10/825,047 (filed Apr. 15, 2004 and entitled Drug Delivery via Punctal Plug); U.S. application Ser. No. 12/604,202 (filed Oct. 22, 2009 and entitled Drug Delivery via Ocular Implant); International Application Serial No. PCT/US2005/023848 (filed Jul. 1, 2005, published as WO 2006/014434 and entitled Treatment Medium Delivery Device and Methods for Delivery); International Application Serial No. PCT/US2007/065792 (filed Apr. 2, 2007, published as WO 2007/115261 and entitled Drug Delivery Methods, Structures, and Compositions for Nasolacrimal System); and International Application Serial No. PCT/US2007/065789 (filed Apr. 2, 2007, published as WO 2007/115259 and entitled Nasolacrimal Drainage System Implants for Drug Therapy).
Occlusive Element
[0143] In an exemplary embodiment, the methods of the invention use an implant having an occlusive element. An occlusive element can be mounted to and expandable with the retention structure, described below, to inhibit tear flow. An occlusive element may inhibit tear flow through the lumen, and the occlusive element may cover at least a portion of the retention structure to protect the lumen from the retention structure. The occlusive element comprises an appropriate material that is sized and shaped so that the implant can at least partially inhibit, even block, the flow of fluid through the hollow tissue structure, for example lacrimal fluid through the canaliculus. The occlusive material may be a thin walled membrane of a biocompatible material, for example silicone, that can expand and contract with the retention structure. The occlusive element is formed as a separate thin tube of material that is slid over the end of the retention structure and anchored to one end of the retention structure as described above. Alternatively, the occlusive element can be formed by dip coating the retention structure in a biocompatible polymer, for example silicone polymer. The thickness of the occlusive element can be in a range from about 0.01 mm to about 0.15 mm, and often from about 0.05 mm to 0.1 mm.
Retention
[0144] In various embodiments of the methods of the invention, an implant including a retention structure is employed to retain the implant in the punctum or canaliculus. The retention structure is attached to or integral with the implant body. The retention structure comprises an appropriate material that is sized and shaped so that the implant can be easily positioned in the desired tissue location, for example, the punctum or canaliculus. In some embodiments, the drug core may be attached to the retention structure via, at least in part, the sheath. In some embodiments, the retention structure comprises a hydrogel configured to expand when the retention structure is placed in the punctum. The retention structure can comprise an attachment member having an axially oriented surface. In some embodiments, expansion of the hydrogel can urge against the axially oriented surface to retain the hydrogel while the hydrogel is hydrated. In some embodiments, the attachment member can comprise at least one of a protrusion, a flange, a rim, or an opening through a portion of the retention structure. In some embodiments, the retention structure includes an implant body portion size and shape to substantially match an anatomy of the punctum and canaliculus.
[0145] The retention structure may have a size suitable to fit at least partially within the canalicular lumen. The retention structure can be expandable between a small profile configuration suitable for insertion and a large profile configuration to anchor the retention structure in the lumen, and the retention structure can be attached near the distal end of the drug core. In specific embodiments, the retention structure can slide along the drug core near the proximal end when the retention structure expands from the small profile configuration to the large profile configuration. A length of the retention structure along the drug core can be shorter in the large profile configuration than the small profile configuration.
[0146] In some embodiments, the retention structure is resiliently expandable. The small profile may have a cross section of no more than about 0.2 mm, and the large profile may have a cross section of no more than about 2.0 mm. The retention structure may comprise a tubular body having arms separated by slots. The retention structure can be disposed at least partially over the drug core.
[0147] In some embodiments, the retention structure is mechanically deployable and typically expands to a desired cross sectional shape, for example with the retention structure comprising a super elastic shape memory alloy such as Nitinol.TM.. Other materials in addition to Nitinol.TM. can be used, for example resilient metals or polymers, plastically deformable metals or polymers, shape memory polymers, and the like, to provide the desired expansion. In some embodiments polymers and coated fibers available from Biogeneral, Inc. of San Diego, Calif. may be used. Many metals such as stainless steels and non-shape memory alloys can be used and provide the desired expansion. This expansion capability permits the implant to fit in hollow tissue structures of varying sizes, for example canaliculae ranging from 0.3 mm to 1.2 mm (i.e. one size fits all). Although a single retention structure can be made to fit canaliculae from 0.3 to 1.2 mm across, a plurality of alternatively selectable retention structures can be used to fit this range if desired, for example a first retention structure for canaliculae from 0.3 to about 0.9 mm and a second retention structure for canaliculae from about 0.9 to 1.2 mm. The retention structure has a length appropriate to the anatomical structure to which the retention structure attaches, for example a length of about 3 mm for a retention structure positioned near the punctum of the canaliculus. For different anatomical structures, the length can be appropriate to provide adequate retention force, e.g. 1 mm to 15 mm lengths as appropriate.
[0148] Although the implant body may be attached to one end of the retention structure as described above, in many embodiments the other end of the retention structure is not attached to the implant body so that the retention structure can slide over the implant body including the sheath body and drug core while the retention structure expands. This sliding capability on one end is desirable as the retention structure may shrink in length as the retention structure expands in width to assume the desired cross sectional width. However, it should be noted that many embodiments may employ a sheath body that does not slide in relative to the core.
[0149] In many embodiments, the retention structure can be retrieved from tissue. A projection, for example a hook, a loop, or a ring, can extend from a portion of the implant body to facilitate removal of the retention structure.
[0150] In some embodiments the sheath and retention structure can comprise two parts.
[0151] In certain embodiments, the lacrimal implants used with the present methods have exceptional retention properties, and are retained in the punctum and canaliculus for a period that is enhanced relative to a commercially available plug (FIG. 9) based upon the percentage of eyes in which an implant was implanted retaining the implant over a selected time period. In other embodiments, the retention properties of the present lacrimal implants of FIG. 33 were evaluated demonstrating superior retention rates over a period of weeks. See, FIGS. 34-39.
[0152] In an exemplary embodiment, the method of the invention uses a lacrimal implant configured to remain implanted in a punctum for at least about 1 week, 2 weeks, 3 weeks, 4 weeks, 5 weeks, 6 weeks, 7 weeks, 8, weeks, 9 weeks 10 weeks, 11 weeks, or at least about 12 weeks or more. In an exemplary embodiment, the lacrimal implant is configured to be retained by the puncta for the duration of the intended sustained release of the therapeutic agent. In various embodiments, the duration of the intended sustained release of the therapeutic agent is at least about 1 week, 2 weeks, 3 weeks, 4 weeks, 5 weeks, 6 weeks, 7 weeks, 8, weeks, 9 weeks 10 weeks, 11 weeks, or at least about 12 weeks or more. In various embodiments at least about 95%, at least about 90%, at least about 85% or at least about 80% of the implanted implants are retained for the duration of the intended controlled release of the therapeutic agent. In an exemplary embodiment, the implant is retained by the puncta for a length of time to show therapeutic efficacy.
[0153] In various embodiments, the present invention provides for the use of implants having structural features that enhance the retention of the implant in a puncta. Amongst other features, the heel of the present implant (e.g., 330) is configured to come to rest in the lacrimal canaliculus ampulla (e.g., 252), effectively locking the implant into place. However, the Applicants have recognized that to prevent rotation and relative movement of the implanted device, which plays a role in the displacement of the device, a first member was needed to maintain the heel in the ampula. Thus, the first member, e.g., 305, is configured to stabilize the punctal plug within the lacrimal canaliculus, prevent rotation and maintain positioning of the plug when the surrounding tissue moves.
[0154] FIGS. 3-6 illustrate exemplary embodiments of lacrimal implants of use in the methods of the invention. The exemplary implants are insertable through a lacrimal punctum 212, 214 and into its associated canaliculus 208, 210. Exemplary lacrimal implants of use in the present invention comprise a first member, a second member and a heel, such as the first member 305, the second member 310 and the third member or heel 330 depicted in FIG. 3A. Exemplary lacrimal implants further comprise a bore that is formed in the heel, for example, the bore 385 formed in the third member or heel 330 in FIG. 3A. In some embodiments, exemplary lacrimal implants further comprise a cavity 458 (e.g., lacrimal implants illustrated in FIG. 4A).
[0155] Referring to FIG. 3A, where a perspective view of an exemplary lacrimal implant 300 of use in the present methods is depicted, the first member 305 is characterized by a first axis A and the second member 310 is characterized by a second axis B.
[0156] The third member or heel 330 is configured to connect the first member 305 and the second member 310 at a first angle .theta..sub.1, where .theta..sub.1 is defined by the first axis A with respect to the second axis B. For instance, in FIG. 3A, the first angle .theta..sub.1 refers to the angle originating at the first axis A and turning counterclockwise from the first axis A to the second axis B. In some embodiments, the first axis A and the second axis B are in the same plane and intersect each other. In some embodiments, the first axis A is in a plane other than the plane of the second axis B, and the first axis A and the second axis B do not intersect. In such embodiments, the first angle .theta..sub.1 refers to the angle defined by a parallel line of the first axis A with respect to the second axis B. This parallel line of the first axis A lies in the same plane as the second axis and intersects with the second axis.
[0157] In some embodiments, the first angle .theta..sub.1 is from about 30 degrees to about 150 degrees, from about 45 degrees to about 135 degrees, or from about 75 degrees to about 105 degrees. For example, in some embodiments, the first angle .theta..sub.1 is approximately 90 degrees.
[0158] In some embodiments, the overall dimension of the implant along the first axis is from about 4 mm to about 8 mm. In an exemplary embodiment, the overall dimension along the first axis is about 5 mm to about 7 mm. In various embodiments, the overall dimension along the first axis is about 6.3 mm.
[0159] In various embodiments, the overall dimension along the second axis B is from about 1 mm to about 3 mm, e.g., from about 1.2 mm to about 1.9 mm.
[0160] In some embodiments, the overall dimension along the first axis is approximately 6.3 mm and the overall dimension along the second axis is approximately 1.2 mm. In various embodiments, the overall dimension along the first axis is approximately 6.3 mm and the overall dimension along the second axis is approximately 1.9 mm. In some embodiments, the overall dimension along the first axis is approximately 4.8 mm and the overall dimension along the second axis is approximately 1.9 mm.
First Member 305
[0161] In some embodiments, the first member 305 is configured to extend into a canaliculus, while the second member 310 is configured to reside in the vertical portion 220, 222 of the canaliculus and to extend to the opening of, or out of the opening of, the associated puncta. When a lacrimal implant 300 of such configuration is inserted into a canaliculus, the intersection of the first axis A and the second axis B resides generally at a curvature of the canaliculus, such as the canaliculus curvature 250 in FIG. 2. In some embodiments, the first member 305 and the second member 310 are connected at the first angle, and that angle is at least about 45 degree, thereby forming an angled intersection between the first member and the second member. In various embodiments, when the lacrimal implant 300 is positioned in the lacrimal canaliculus, at least a portion of the angled intersection is biased against a canaliculus curvature of the lacrimal canaliculus. In this embodiment, the lacrimal implant 300 uses anatomical structures to facilitate the retention of the implanted lacrimal implant 300.
[0162] FIG. 3B depicts a side view of an exemplary lacrimal implant 300 of the invention. In some embodiments, the first member 305 includes an intermediate segment 315, a tip segment or tip 325, and a forward segment 320 in between the forward segment and tip segment. While the intermediate segment 315 is configured to be connected to the second member 310 by the third member or heel 330, the tip segment or tip 325 is configured to be inserted through a punctum prior to the other two segments of the first member 305 and prior to the other members of the lacrimal implant 300.
[0163] In some embodiments, the intermediate segment 315, the forward segment 320 and the tip segment or tip 325 are distinguishable from each other in general by their shapes. For example, in some embodiments, the intermediate segment 315 has a generally cylindrical shape with a diameter that is larger than the diameter of the tip segment or tip 325. In various embodiments, the forward segment 320 is tapered and has a conical shape, such that the forward segment 320 connects the intermediate segment 315 at one end and the tip segment or tip 325 at the other end. In some embodiments, the transition from the intermediate segment 315 to the forward segment 320 or the transition from the forward segment 320 to the tip segment or tip 325 is gradual and smooth such that no distinguishable edge exists at the transition.
[0164] In some embodiments, the intermediate segment 315 has a cylindrical shape. In various embodiments, the intermediate segment has a circular cross section, an elliptic cross section, or a polygonal cross section. The intermediate segment 315 is of any useful combination of length and diameter.
[0165] In some embodiments, the intermediate segment 315 has a diameter that is from about 0.4 mm to about 0.8 mm. For example, in some embodiments the diameter of the intermediate segment 315 is from about 0.53 mm to about 0.63 mm. In some embodiments, the intermediate segment 315 has a length along the first axis A that is from about 0.5 mm to about 3.5 mm. For example, in some embodiments the length of the intermediate segment 315 is from about 1 mm to about 2.8 mm.
[0166] In some embodiments, the tip segment or tip 325 is substantially a semi-sphere, or a portion of a semi-sphere. In exemplary embodiments, the semi-sphere, or portion therapy, has a radius that is from about 0.05 mm to about 0.3 mm. For example, in some embodiments, the radius of the tip segment or tip 325 is approximately 0.20 mm.
[0167] In some embodiments, the forward segment 320 has a conical configuration, tapering from the diameter of the intermediate segment 315 as it approaches the tip segment or tip 325. In some embodiments, the forward segment 320 is short and is tapered steeply, thus forming a wider taper angle. The forward segment 320 can also be long and tapered more gradually, thus forming a narrower taper angle. The tapering angle .theta..sub.3 is illustrated in FIG. 3E. In some embodiments, the tapering angle .theta..sub.3 is from about 2.degree. to about 10.degree.. For example, in some embodiments the tapering angle .theta..sub.3 is from about 3.8.degree. to about 7.8.degree.. In some embodiments, .theta..sub.3 is about 7.8.degree.. In some embodiments, the forward segment 320 has a length along the first axis A that is from about 1 mm to about 5 mm. For example in some embodiments the length of forward segment 320 is from about 1.7 mm to about 3.5 mm.
Second Member 310
[0168] Referring to FIG. 3B, in some embodiments of implants of use in the present method, the second member 310 includes an upright segment 335 that extends from the third member or heel 330 generally along the direction of the second axis B. In various embodiments, the second member 310 further includes a head segment 340 that attaches to the upright segment 335 at an end opposite to the third member or heel 330. In some embodiments, the second member 310 is configured such that the upright segment 335 resides in the vertical portion of the canaliculus while the head segment 340 contacts the tissue surrounding the exterior of the punctum when the lacrimal implant 300 is positioned in the lacrimal canaliculus. In an exemplary embodiment, illustrated in FIGS. 3A-3F, the upright segment 335 has a cylindrical shape and the head segment 340 has an oval or oblong configuration. However, it will be appreciated that any other suitable shapes or configurations can be used and are within the scope of the present invention. For example, in various embodiments, the upright segment 335 is configured to be a conical; the head segment 340 is configured to have a circular, elliptical or polygonal cross section.
[0169] In some embodiments, the upright segment 335 has a characteristic diameter that is from about 0.7 mm to about 0.9 mm. For example, in some embodiments, the characteristic diameter of the upright segment 335 is about 0.8 mm.
[0170] In some embodiments, the upright segment 335 has a length in the direction of the second axis B that is from about 0.7 mm to about 1.5 mm. For example, in some embodiments the length of upright segment 335 along the direction of the second axis B is about 0.9 mm.
[0171] Generally, the head segment 340 has a cross section characterized by a minor axis and a major axis. The minor axis and the major axis refer to the shortest characteristic diameter and the longest characteristic diameter of the cross section, respectively. As such, the minor axis is equal to or less than the major axis. For instance, in some embodiments where the head segment 340 has a circular cross section, the minor axis and the major axis are of equal length. In various embodiments, the head segment 340 has an oval or oblong cross section, and the minor axis is shorter than the major axis. In some embodiments, the head segment 340 is elongated in a direction that is parallel to the first axis A. The major axis indicates the extension of the first member 305 and facilitates positioning of the lacrimal implant 300 in the punctum and canalinculus. In some embodiments, the major axis is from about 1.5 mm to about 2.5 mm. In various embodiments, the minor axis is from about 1 mm to about 1.5 mm. For example, in some embodiments, the major axis and the minor axis head segment 340 are approximately 1.9 mm and 1.3 mm respectively. In some embodiments, the head segment 340 has a thickness in the direction of the second axis that is from about 0.2 mm to about 0.4 mm. For example, in some embodiments, the thickness of the head segment 340 in the direction of the second axis is approximately 0.3 mm.
[0172] Referring still to FIG. 3B, exemplary head segment 340 comprises an under-surface 350 facing towards the third member or heel 330 and an outer-surface 355 that faces away from the third member or heel 330. Exemplary head segment 340 further comprises an edge surface 345 that couples the under-surface 350 and the outer-surface 355. The distance between the under-surface 350 and the outer-surface 355 can be readily varied. In some embodiments, the distance is from about 0.2 mm to about 0.4 mm.
[0173] In some embodiments, the outer-surface 355 is smaller than the under-surface 350 and is substantially flat. In various embodiments, the edge surface 345 is tapered, curved, angular, or multifaceted. In some embodiments, the edge surface 345 has a radius of curvature that is from about 0.2 mm to about 0.7 mm. In some embodiments, the under-surface 350 is in general flat and is configured to contact the exterior tissue surrounding the punctum when the lacrimal implant 300 is positioned in the lacrimal canaliculus.
Third Member or Heel 330
[0174] In some embodiments, the third member or heel 330 includes an upper surface 360, a lower surface 365, and side surfaces 370. In the illustrated embodiments, the bore 385 extends from the upper surface 360 into the third member or heel 330. In some embodiments, the upper surface 360 and the lower surface 365 are substantially flat and separated from each other by a distance. Such distance is readily variable and is typically about 0.3 mm to about 0.7 mm. For instance, in some embodiments, the upper surface 360 and the lower surface 365 are separated by a distance that is from about 0.4 mm to 0.6 mm (e.g., about 0.53 mm). In some embodiments, the upper surface 360 extends beyond the intersection with the second member 310. In some embodiments, the upper surface 360 extends beyond the intersection with the second member 310 for a distance that is from about 0.3 to about 0.6 mm. The upper surface 360 can also be joined with the side surfaces 370. In various embodiments, upper surface 360 and side surfaces 370 are joined by a curved intersection 380. In some embodiments, the curved intersection 380 has a radius of curvature that is from about 0.04 mm to about 0.08 mm.
[0175] Referring now to FIGS. 3D and 3F, in some embodiments, the third member or heel 330 includes a heel connecting segment 375 configured to couple the third member or heel 330 to the first member 305, or to the intermediate segment 315 of the first member 305. The heel connecting segment 375 is of readily variable shape, including flat or curved structures. In FIG. 3F, a width of the heel connecting segment 375 in the direction of the second axis B varies along the direction of the first axis A. For example, the heel connecting segment 375 has a smaller width at or near the side surfaces 370 than the diameter of the intermediate segment 315 of the first member 305. In some embodiments, at or near the intersection with the intermediate segment 315, the heel connecting segment 375 increases the width and thus forms a notch as depicted in FIG. 3F. It will be appreciated that the notch can be either deeper or shallower along both the first axis A and the second axis B before it meets the first member 305 or the second member 310.
[0176] A notch is not a required feature in the implants of the present invention. In some embodiments, the heel connecting segment 375 has the same dimension as the diameter of the intermediate segment 315. For example, the thickness of the third member or heel 330 along the second axis B is equal to the diameter of the intermediate segment 315 of the first member 305. For example, in some embodiments, both the thickness of the third member or heel 330 in the direction of the second axis B and the diameter of the intermediate segment 315 are from about 0.53 mm to about 0.63 mm. In such configurations, the third member or heel 330 couples with the intermediate segment 315 without forming a notch, as illustrated by the alternative heel connecting segment 675 in FIG. 6.
[0177] By way of illustration, the third member or heel 330 depicted in FIGS. 3A-3F is substantially parallel to the first axis A of the first member 305. It would be appreciated that this is unnecessary. In some embodiments, the third member or heel 330 can form an angle with relation to the first axis A.
Bore 385
[0178] Exemplary structures of the bore 385 are detailed in FIGS. 3E and 3F, where a cross sectional view and a partial enlarged cross sectional view of the lacrimal implant 300 are provided. The bore 385 is configured to receive a tip or other protrusion of an external insertion tool for facilitating insertion of the lacrimal implant 300 into a lacrimal punctum. See FIG. 7. The configuration, including size, shape, angle (O.sub.2) and position of the bore in the heel are readily adjustable to facilitate the mating of the insertion tool with the bore, the flexibility of the heel, or the retention of the lacrimal implants. Depending on the purpose or use of the implant and the materials used for making the heel, the characteristics of the bore noted above are readily varied. Configurations of the bore 385 disclosed herein are illustrative and any other suitable configurations are within the scope of the present invention.
[0179] In FIG. 3F, an exemplary bore 385 is characterized by a third axis C and a second angle .theta..sub.2 that is defined by the first axis with respect to the third axis A in a similar way as the first angle .theta..sub.1. In some embodiments, the second angle .theta..sub.2 is from about 15.degree. to about 90.degree.. For example, in some embodiments, the second angle .theta..sub.2 is about 45.degree..
[0180] In some embodiments, the bore 385 has a depth along the direction of the third axis C that is from about 0.3 mm to about 0.7 mm. For example, in some embodiments the depth of the bore 385 is approximately 0.4 mm and in some embodiments is approximately 0.6 mm. The bore 385 may include a bore shaft 390 that is generally cylindrical, with a circular, elliptical, oval, or polygonal cross section. The bore 385 may further include a bore tip 395 at which the bore shaft 390 terminates. An exemplary bore tip 395 generally has a semispherical configuration. In some embodiments, the bore shaft 390 has a characteristic diameter that is from about 0.1 mm to about 0.3 mm. In some embodiments, the characteristic diameter of the bore is approximately 0.17 mm. As will be appreciated, the shapes, sizes, orientations disclosed in the present application are illustrative, and any other suitable shapes, sizes, or orientations are within the scope of the present application. In addition, it will be appreciated that the opening of the bore can be positioned closer to the second member or closer to the edge of the heel.
Cavity 458
[0181] FIG. 4A-4C illustrates an exemplary lacrimal implant 400 that is insertable through a lacrimal punctum 212, 214 and into its associated canaliculus 208, 210. In FIG. 4A, the lacrimal implant 400 comprises a cavity 458 that is configured to house a therapeutic agent core, also referred to herein as a drug core, or other materials for release into an eye or surrounding tissues for treatment of various ocular, sinus or other diseases.
[0182] In the illustrated exemplary embodiment, the cavity 458 is formed in the head segment 340 and has an opening through the outer-surface 355. The cavity 458 can be shallow such that it stays within the head segment 340. The cavity 458 can be also deeper and extend beyond the head segment 340 and into the upright segment 335. Illustrated exemplary cavity 458 is in general substantially cylindrical with a circular cross section. Any other suitable configuration is within the scope of the present application. For example, in some embodiments, the cavity 458 has a truncated spherical configuration, or has a cylindrical configuration with an oblong or a polygonal cross section.
[0183] In some embodiments, the cavity 458 has a depth in the direction of the second axis B that is about from 0.2 mm to about 1.4 mm. For example, in some embodiments, the depth of the cavity 458 is approximately 1.2 mm. In some embodiments, the cavity 458 has a diameter that is from about 0.3 mm to about 0.7 mm. For example, in some embodiments the diameter of the cavity 458 is from about 0.42 mm to about 0.55 mm. In an exemplary embodiment, the cavity 458 extends into the upright segment 335, and the diameter of the cavity 458 is smaller than the diameter of the upright segment 335.
[0184] Referring to FIG. 4C, the cavity 458 includes a bottom 482. In various embodiments, the bottom 482 is rounded. In various embodiments, the rounded bottom has a radius of curvature that is from about 0.03 mm to about 0.07 mm.
[0185] FIG. 5 depicts exemplary configurations of the cavity 458. In FIG. 5, the cavity 458 includes a lip 584 or other retaining structure positioned at the opening of the cavity 458. The lip 584 or the other retaining structure are optionally configured to partially enclose the cavity 458, e.g, prevent a therapeutic agent core or other materials from moving out of the cavity 458. In some embodiments, the lip 584 is a square cross sectional annulus that extends down from the outer-surface 355 into the cavity 458 and extends inwardly towards the center of the opening of the cavity 458. In some embodiments, the lip 584 is of a tab configuration and includes a plurality of spaced lips that extend inwardly into the opening of the cavity 458. The lip 584 may extend downwardly from about 0.02 mm to about 0.1 mm and inwardly from about 0.02 mm to about 0.1 mm. For example, in some embodiments, the lip 584 extends about 0.05 mm downwardly or inwardly.
Formation of Lacrimal Implants
[0186] Exemplary lacrimal implants of use in methods of the present invention are made of various materials including plastic, rubber, polymer, or composite. Exemplary lacrimal implants of the present invention formed from one or more material including plastic, rubber, polymer, composites, or other appropriate materials. In some embodiments, the lacrimal implants are formed from liquid silicone rubber. For instance, in exemplary embodiments, lacrimal implants are formed from a material marketed as NuSil 4840 liquid silicone rubber, NuSil 4870, or a mixture including such a liquid silicone rubber. Examples of such a mixture include a material marketed as 6-4800, which comprises NuSil 4840 with from about 1% to about 5%, e.g., from about 2% to about 4% 6-4800.
[0187] In some embodiments, the lacrimal implant is formed from biodegradable materials, for instance, biodegradable elastic materials including cross-linked polymers, such as poly (vinyl alcohol). In some embodiments, the lacrimal implant can comprise a co-polymer, such as silicone/polyurethane co-polymer, silicone/urethane, silicone/poly (ethylene glycol) (PEG), and silicone/2hydroxyethyl methacrylate (HEMA). As discussed in commonly-owned Utkhede et al., U.S. Patent Publication No. 2009/0104243, entitled "DRUG CORES FOR SUSTAINED RELEASE OF THERAPEUTIC AGENTS," filed Sep. 5, 2008, which is herein incorporated by reference in its entirety, urethane-based polymer and copolymer materials allow for a variety of processing methods and bond well to one another.
[0188] The hardness of the material is selected to facilitate or alter the retention of the lacrimal implant within the lacrimal punctum and its associated canaliculus. Accordingly, in some embodiments, a material having a durometer rating of from about 20D to about 80D, e.g., about 30D to about 70D, e.g., from about 40D to about 60D is of use to adjust parameters such as patient comfort and retention. For example, in some embodiments, the durometer rating of the material used to form the lacrimal implants is approximately 40D. Materials other than those exemplified above providing a durometer rating for the lacrimal implants within the stated ranges, and particularly that is about 40D are also of use. In some embodiments, a harder material or softer material is utilized for the entire lacrimal implant or for portions thereof. In such case, the lacrimal implants are formed from the materials that provide a durometer rating of about 70D.
[0189] In some embodiments, the lacrimal implants of use in the present methods are formed of multiple materials, where certain members or portions of the lacrimal implants are formed with materials having different properties. For example, in some embodiments the first member 305 is formed of a harder durometer rated material while the second member 310 is formed of a softer durometer rated material. In some embodiments, the first member 305 is formed of a softer durometer rated material while the second member 310 is formed of a harder durometer rated material. In some embodiments the third member or heel 330 is formed of a harder durometer rated material than one or more parts of the remainder of the second member 310. In various embodiments, the third member or heel 330 is formed of a softer durometer rated material than the remainder of the second member 310.
[0190] In certain embodiments, the implant comprises a contrast agent to aid in detection of the inserted lacrimal implant. See, US Patent Publication No. 2009/0099626, filed Sep. 5, 2008 entitled LACRIMAL IMPLANT DETECTION. In one embodiment, the contrast agent is a dye or pigment. In another embodiment, a green colorant is added during the manufacturing process of the lacrimal implant. In certain embodiments, this green colorant is premixed with the NuSil liquid silicone rubber to form a green lacrimal implant.
[0191] Exemplary implants of use in the invention can be formed by methods known in the art, including, but not limited to, machining a blank to the desired shape and size and molding the material forming the implant.
Insertion Tools
[0192] Installing the lacrimal implant of use in the invention can be facilitated by the use of an insertion tool. For example, in some embodiments the lacrimal implants and/or the inserter tool may include features or components that are found in U.S. Patent Application Publication No. 2009/0104248, U.S. Patent Application Publication No. 2010/0274204, U.S. Patent Application Publication No. 2009/0105749 and International Patent Application Publication No. WO 2011/066479, both of which are incorporated herein by reference in their entirety.
[0193] Turning to FIG. 7, an exemplary insertion tool is shown engaged with an implant of the invention through meeting of pin 760 and insertion of the lacrimal implants into a lacrimal punctum. The lacrimal implants include the exemplary embodiments disclosed above, variations thereof, or any similar structures.
3) Sustained Release Formulations
[0194] Conventional drug delivery involving frequent periodic dosing is not ideal or practical in many instances. For example, with more toxic drugs, conventional periodic dosing can result in unfavorably high initial drug levels at the time of dosing, followed by low drug levels between doses often times below levels of therapeutic value. Likewise, conventional periodic dosing may not be practical or therapeutically effective in certain instances such as with pharmaceutical therapies targeting areas of the inner eye or brain in need of treatment such as the retina. Accordingly, in certain embodiments, the lacrimal implant further comprises one or more therapeutic agents within its structure. In certain embodiments, the therapeutic agent is dispersed throughout the device (e.g. providing a saturated or impregnated implant). In other certain embodiments, the therapeutic agent is located at one or more distinct locations or zones of the implant. In an exemplary embodiment, the therapeutic agent is located in a cavity of the device and the component holding the therapeutic agent is referred to as a drug core. This drug core may comprise additional component such as an impermeable sheath to prevent migration of the therapeutic drug through the lacrimal implant and/or provide direction for the drug migration.
[0195] In certain embodiments, in which the agent is dispersed throughout the device, the rate and location of release of the agent is controlled by coating at least a component of the device with a material that is impermeable to the drug. In an exemplary embodiment, essentially the entire device is coated with the material with the exception of one or more gaps in the material through which the agent can elute into the eye or surrounding tissue. An exemplary coating is a Parylene coating (See, US Patent Publication No. 2008/0181930, herein incorporated by reference).
[0196] In one embodiment, the lacrimal implant of the invention is configured as a sustained release device, releasing the incorporated therapeutic agent in a therapeutically effective manner, e.g., at a rate that provides a therapeutically effective dosage for at least about 1 week, 2 weeks, 3 weeks, 4 weeks, 5 weeks, 6 weeks, 7 weeks, 8, weeks, 9 weeks 10 weeks, 11 weeks, or at least about 12 weeks or more. In an exemplary embodiment, the lacrimal implant is configured to be retained by the puncta for the duration of the intended controlled release of the therapeutic agent. In various embodiments, the duration of the intended controlled release of the therapeutic agent is at least about 1 week, 2 weeks, 3 weeks, 4 weeks, 5 weeks, 6 weeks, 7 weeks, 8, weeks, 9 weeks 10 weeks, 11 weeks, or at least about 12 weeks or more. In various embodiments at least 95% of the implanted implants are retained for the duration of the intended controlled release of the therapeutic agent. In an exemplary embodiment, the implant is retained by the puncta for a length of time to show therapeutic efficacy.
[0197] In an exemplary embodiment, the implant is formatted as a unit dosage of the therapeutic agent. In various embodiments, the implant is formatted as a unit dosage of an antiglaucoma agent. In an exemplary embodiment, the antiglaucoma agent is a prostaglandin.
Therapeutic Agent (Drug) Core
[0198] In an exemplary embodiment, the methods of the invention utilize an implant including a distinct therapeutic agent core or integrated drug or other agent disposed in at least one of the first member 305 or the second member 310 of the implant body, to provide a sustained release of a therapeutic agent. For instance, the drug core or integrated drug or other agent disposed may be disposed in the cavity 458 of the lacrimal implant 400 to provide a sustained drug or other therapeutic agent release.
[0199] An exemplary implant of use in the methods of the invention is configured to deliver a therapeutic agent to one or more of an eye, nasal passage or inner ear system. In various embodiments, the drug is delivered systemically to the subject through the eye. A therapeutic agent core can comprise one or more therapeutic agents, and in some examples, one or more matrix materials to provide sustained release of the drug or other agents.
[0200] In various embodiments, the therapeutic agent core is inserted into cavity 458.
[0201] In various examples, the distinct drug core or integrated drug or other agent includes at least about 20 micrograms, at least about 40 micrograms, at least about 45 micrograms, at least 80 micrograms, or at least 95 micrograms of a drug (e.g., latanoprost), such as is further discussed in commonly-owned Butuner et al., U.S. Patent Publication No. 2009/0280158, entitled "SUSTAINED RELEASE DELIVERY OF ACTIVE AGENTS TO TREAT GLAUCOMA AND OCULAR HYPERTENSION," filed May 8, 2009, and commonly-owned Butuner, U.S. Patent Publication No. US 2010/0209477, entitled "SUSTAINED RELEASE DELIVERY OF ONE OR MORE AGENTS," filed Jan. 22, 2010, both of which are incorporated by reference in their entirety, including their descriptions of drug or other agent concentration and formulations.
[0202] The drug core can comprise one or more biocompatible materials capable of providing a sustained release of the one or more drugs or agents. The drug core can comprise a matrix including a substantially non-biodegradable silicone matrix with dissolvable inclusions of the drugs or agents located therein. The drug core can include other structures that provide sustained release of the drugs or agents, for example a biodegradable matrix, a porous drug core, a liquid drug core or a solid drug core. A matrix that includes the drugs or agents can be formed from either biodegradable or non-biodegradable polymers. In some examples, a non-biodegradable drug core can include silicone, acrylates, polyethylenes, polyurethane, polyurethane, hydrogel, polyester (e.g., DACRON.TM. from E.I. Du Pont de Nemours and Company, Wilmington, Del.), polypropylene, polytetrafluoroethylene (PTFE), expanded PTFE (ePTFE), polyether ether ketone (PEEK), nylon, extruded collagen, polymer foam, silicone rubber, polyethylene terephthalate, ultra high molecular weight polyethylene, polycarbonate urethane, polyurethane, polyimides, stainless steel, nickel-titanium alloy (e.g., Nitinol), titanium, stainless steel, cobalt-chrome alloy (e.g., ELGILOY.TM. from Elgin Specialty Metals, Elgin, Ill.; CONICHROME.TM. from Carpenter Metals Corp., Wyomissing, Pa.). In some examples, a biodegradable drug core can comprise one or more biodegradable polymers, such as protein, hydrogel, polyglycolic acid (PGA), polylactic acid (PLA), poly(L-lactic acid) (PLLA), poly(L-glycolic acid) (PLGA), polyglycolide, poly-L-lactide, poly-D-lactide, poly(amino acids), polydioxanone, polycaprolactone, polygluconate, polylactic acid-polyethylene oxide copolymers, modified cellulose, collagen, polyorthoesters, polyhydroxybutyrate, polyanhydride, polyphosphoester, poly(alpha-hydroxy acid) and combinations thereof. In some examples, the drug core can comprise a hydrogel polymer.
[0203] The therapeutic agent can be present in the device in a formulation with a pharmaceutically acceptable carrier, e.g., excipients, suspending agents, diluents, fillers, salts, buffers, stabilizers, solubilizers, solvents, dispersion media, coatings, isotonic agents, and other materials known in the art. The pharmaceutical formulation optionally includes potentiators, complexing agents, targeting agents, stabilizing agents, cosolvents, pressurized gases, or solubilizing conjugates.
[0204] Exemplary excipients include sugars such as lactose, sucrose, mannitol, or sorbitol; cellulose preparations such as, maize starch, wheat starch, rice starch, potato starch, gelatin, gum tragacanth, methyl cellulose, hydroxypropylmethylcellulose, sodium caroxymethylcellulose, and/or polyvinylpyrrolidone (PVP). Preferred excipients include lactose, gelatin, sodium carboxymethyl cellulose, and low molecular weight starch products.
[0205] Exemplary suspending agents that can serve as valve lubricants in pressurized pack inhaler systems are desirable. Such agents include oleic acid, simple carboxylic acid derivatives, and sorbitan trioleate.
[0206] Exemplary diluents include water, saline, phosphate-buffered citrate or saline solution, and mucolytic preparations. Other diluents that can be considered include alcohol, propylene glycol, and ethanol; these solvents or diluents are more common in oral aerosol formulations. Physiologically acceptable diluents that have a tonicity and pH compatible with the alveolar apparatus are desirable. Preferred diluents include isotonic saline, phosphate buffered isotonic solutions whose tonicity have been adjusted with sodium chloride or sucrose or dextrose or mannitol.
[0207] Exemplary fillers include glycerin, propylene glycol, ethanol in liquid or fluid preparations. Suitable fillers for dry powder inhalation systems include lactose, sucrose, dextrose, suitable amino acids, and derivatives of lactose. Preferred fillers include glycerin, propylene glycol, lactose and certain amino acids.
[0208] Exemplary salts include those that are physiologically compatible and provide the desired tonicity adjustment. Monovalent and divalent salts of strong or weak acids are desirable. Preferred salts include sodium chloride, sodium citrate, ascorbates, sodium phosphates.
[0209] Exemplary buffers include phosphate or citrate buffers or mixed buffer systems of low buffering capacity. Preferred buffers include phosphate or citrate buffers.
[0210] Table 1 shows exemplary drug insert silicones that may be used and associated cure properties, according to embodiments of the present invention. The drug core insert matrix material can include a base polymer comprising dimethyl siloxane, such as MED-4011, MED 6385 and MED 6380, each of which is commercially available from NuSil. The base polymer can be cured with a cure system such as a platinum-vinyl hydride cure system or a tin-alkoxy cure system, both commercially available from NuSil. In many embodiments, the cure system may comprise a known cure system commercially available for a known material, for example a known platinum vinyl hydride cure system with known MED-4011. In a specific embodiment shown in Table 1, 90 parts of MED-4011 can be combined with 10 parts of the crosslinker, such that the crosslinker comprises 10% of the mixture. A mixture with MED-6385 may comprise 2.5% of the crosslinker, and mixtures of MED-6380 may comprise 2.5% or 5% of the crosslinker.
TABLE-US-00001 TABLE 1 Drug Insert Silicone Selections Crosslinker Material Base Polymer Cure System Percent MED-4011 Dimethyl siloxane Platinum vinyl 10% Silica filler hydride system material 10% MED-6385 Dimethyl siloxane Tin-Alkoxy 2.5% 2.5% Diatomaceous earth filler material MED-6380 Dimethyl siloxane Tin-Alkoxy 2.5 to 5% without filler material
[0211] It has been determined according to the present invention that the cure system and type of silicone material can affect the curing properties of the solid drug core insert, and may potentially affect the yield of therapeutic agent from the drug core matrix material. In specific embodiments, curing of MED-4011 with the platinum vinyl hydride system can be inhibited with high concentrations of drug/prodrug, for example over 20% drug, such that a solid drug core may not be formed. In specific embodiments, curing of MED-6385 or MED 6380 with the tin alkoxy system can be slightly inhibited with high concentrations, e.g. 20%, of drug/prodrug. This slight inhibition of curing can be compensated by increasing the time or temperature of the curing process. For example, embodiments of the present invention can make drug cores comprising 40% drug and 60% MED-6385 with the tin alkoxy system using appropriate cure times and temperatures. Similar results can be obtained with the MED-6380 system the tin-alkoxy system and an appropriate curing time or temperature. Even with the excellent results for the tin alkoxy cure system, it has been determined according to the present invention that there may be an upper limit, for example 50% drug/prodrug or more, at which the tin-alkoxy cure system may not produce a solid drug core. In many embodiments, the latanoprost or other intraocular pressure-reducing therapeutic agent(s) in the solid drug core may be at least about 5%, for example a range from about 5% to 50%, and can be from about 20% to about 40% by weight of the drug core.
[0212] In a specific embodiment, drug cores with two different concentrations of Latanoprost are utilized in the method of the invention.
[0213] In one embodiment of the present methods an implant with a drug core with 46 .mu.g of Latanoprost was inserted into a lacrimal implant, See Table 2.
TABLE-US-00002 TABLE 2 Latanoprost Punctal Plug Delivery System (PPDS) Composition (46 .mu.g) L-PPDS, 46 .mu.g Material Specific Formulation or ID Description with DMPC Latanoprost Chirogate International GMP grade, neat oil 46.0 .mu.g Everlight Chemical Industrial Corporation Silicone NuSil MED-6385 (MAF 970) Two part medical grade formulation Part A - proprietary silicone 60.1 .mu.g formulation Part B - stannous octoate 0.70 nL Crosslinker NuSil MED5-6382 (MAF 1289) Only crosslinker is used from kit 2.1 nL Dimyristoyl Nippon Fine Chemical GMP grade, white solid 8.6 .mu.g Phosphatidylcholine (DMPC) Tubing Polyimide Polyimide tube length (0.0155'' 0.95 mm inner diameter, with 0.0010'' wall - medical grade) Cyanoacrylate Loctite .RTM. 4305 .TM. Medical grade ethyl cyanoacrylate ~0.3 .mu.g adhesive with photoinitiator
[0214] In another embodiment, a drug core with 95 .mu.g of Latanoprost was made and inserted into a lacrimal implant, see Table 3.
TABLE-US-00003 TABLE 3 Latanoprost Punctal Plug Delivery System (PPDS) Composition (95 .mu.g) L-PPDS, 95 .mu.g Material Specific Formulation or ID Description with DMPC Latanoprost Everlight Chemical Industrial GMP grade, neat oil 95.0 .mu.g Corporation Silicone NuSil MED-6385 (MAF 970) Two part medical grade formulation Part A - proprietary silicone 124.7 .mu.g formulation Part B - stannous octoate 0.70 nL Crosslinker NuSil MED5-6382 (MAF 1289) Only crosslinker is used from kit 4.8 nL Dimyristoyl Nippon Fine Chemical GMP grade, white solid 17.8 .mu.g Phosphatidylcholine (DMPC) Tubing Polyimide Polyimide tube length (0.0220'' 0.95 mm inner diameter, with 0.0010'' wall - medical grade) Cyanoacrylate Loctite .RTM. 4305 .TM. Medical grade ethyl cyanoacrylate ~0.3 .mu.g adhesive with photoinitiator
[0215] Further discussion of drug-releasing or other agent-releasing drug cores can be found in commonly-owned Utkhede et al., U.S. Patent Publication No. 2009/0104243, entitled "DRUG CORES. FOR SUSTAINED RELEASE OF THERAPEUTIC AGENTS," filed Sep. 5, 2008, which is herein incorporated by reference in its entirety.
Sheath Body
[0216] In certain embodiments, the implant of use in the methods of the invention includes a therapeutic agent core which is encased in a sheath body. The sheath body can comprise appropriate shapes and materials to control the migration of latanoprost or other anti-glaucoma agents from the drug core. In some embodiments, the sheath body houses the drug core and can fit snugly against the core. The sheath body is made from a material that is substantially impermeable to the therapeutic agent so that the rate of migration of the agent may be largely controlled by the exposed surface area of the drug core that is not covered by the sheath body. In certain embodiments, migration of the therapeutic agent through the sheath body can be about one tenth of the migration of the therapeutic agent through the exposed surface of the drug core, or less, often being one hundredth or less. In other words, the migration of the therapeutic agent through the sheath body is at least about an order of magnitude less that the migration of the therapeutic agent through the exposed surface of the drug core. Suitable sheath body materials include polyimide, polyethylene terephthalate (hereinafter "PET"). The sheath body has a thickness, as defined from the sheath surface adjacent the core to the opposing sheath surface away from the core, from about 0.00025'' to about 0.0015''. The total diameter of the sheath that extends across the core ranges from about 0.2 mm to about 1.2 mm. The core may be formed by dip coating the core in the sheath material. Alternatively or in combination, the sheath body can comprise a tube and the core introduced into the sheath, for example as a liquid or solid that can be slid, injected or extruded into the sheath body tube. The sheath body can also be dip coated around the core, for example dip coated around a pre-formed core.
[0217] The sheath body can be provided with additional features to facilitate clinical use of the implant. For example, the sheath may receive a drug core that is exchangeable while the implant body, retention structure and sheath body remain implanted in the subject. The sheath body is often rigidly attached to the retention structure as described above, and the core is exchangeable while the retention structure retains the sheath body. In specific embodiments, the sheath body can be provided with external protrusions that apply force to the sheath body when squeezed and eject the core from the sheath body. Another drug core can then be positioned in the sheath body. In many embodiments, the sheath body or retention structure may have a distinguishing feature, for example a distinguishing color, to show placement such that the placement of the sheath body or retention structure in the canaliculus or other body tissue structure can be readily detected by the subject. The retention element or sheath body may comprise at least one mark to indicate the depth of placement in the canaliculus such that the retention element or sheath body can be positioned to a desired depth in the canaliculus based on the at least one mark.
Formation of the Therapeutic Agent Cores
[0218] Those of skill in the art will be familiar with various methods useful for making the drug cores and inserting into the lacrimal implant to complete the present drug delivery system described as being of use in the methods disclosed herein. Particular methods are described in the above-identified patent documents, the disclosures of which are incorporated herein by reference in their entirety.
[0219] For example, drug cores as described above may be fabricated with different cross sectional sizes of between about 0.006 inches and 0.025 inches. Drug concentrations in the core may be about 5%, 10%, 20%, 30%, 40% or 50% in a silicone matrix. These drug cores can be made with a syringe tube and cartridge assembly, mixing the therapeutic agent(s) with silicone, and injecting the mixture into a polyimide tube which is cut to desired lengths and sealed. The length of the drug cores can be approximately 0.80 to 0.95 mm, or any length designed to fit within the cavity of the present lacrimal implants.
[0220] Syringe Tube and Cartridge Assembly: 1. Polyimide tubing of various diameters (for example 0.006 inches, 0.0125 inches and 0.025 inches) can be cut to 15 cm length. 2. The polyimide tubes can be inserted into a Syringe Adapter. 3. The polyimide tube can be adhesive bonded into luer adapter (Loctite, low viscosity UV cure). 4. The end of the assembly can then be trimmed. 5. The cartridge assembly can be cleaned using distilled water and then with methanol and dried in oven at 60 degrees C.
[0221] The therapeutic agent can be mixed with silicone. Therapeutic agent(s) may be provided as a 1% solution in methylacetate. The appropriate amount of solution can be placed into a dish and using a nitrogen stream, the solution can be evaporated until only the therapeutic agent(s) remains. The dish with the therapeutic agent(s) oil can be placed under vacuum for 30 minutes. This therapeutic agent(s) can then be combined with silicone, with three different concentrations of therapeutic agent(s) (5%, 10% and 20%) in silicone NuSil 6385 being injected into tubing of different diameters (0.006 in, 0.012 in and 0.025 inches) to generate 3.times.3 matrixes. The tube can then be injected: 1. The cartridge and polyimide tubes assembly can be inserted into a 1 ml syringe. 2. One drop of catalyst (MED-6385 Curing Agent) can be added in the syringe. 3. Excess catalyst can be forced out of the polyimide tube with clean air. 4. The syringe can then be filled with silicone drug matrix. 5. The tube can then be injected with drug matrix until the tube is filled or the syringe plunger becomes too difficult to push. 6. The distal end of the polyimide tube can be closed off and pressure can be maintained until the silicone begins to solidify. 7. Allow to cure at room temperature for 12 hours. 8. Place under vacuum for 30 minutes. 9. The tube can then be place in the correct size trim fixture (prepared in house to hold different size tubing) and drug inserts can be cut to length (0.80-0.95 mm).
[0222] In certain embodiments, the drug core formulations of Table 2 and 3 are made using a cold extrusion method described in US Patent Publication No. 2009/0104243 entitled DRUG CORES FOR SUSTAINED RELEASE OF THERAPEUTIC AGENTS. Filed Sep. 5, 2008, the entirety of which is incorporated herein by reference. See, Example 3.
[0223] In this instance, the silicone and latanoprost are prepared as described above. When latanoprost, which is in a liquid physical state at about room temperature (22.degree. C.), and thus is also in a liquid physical state at human body temperature (37.degree. C.), is used, the agent and the matrix material can be mixed by techniques that bring about a high degree of dispersion of the liquid latanoprost droplets in the matrix material in which it can be substantially insoluble. Mixing techniques should provide for a dispersion of the droplet within the matrix material, such that when curing takes place, the liquid therapeutic agent is present as relatively small, relatively homogeneously dispersed discrete droplets within the matrix of solid silicone material.
[0224] In this cold extrusion method, the mixture of latanoprost and silicone can be injected into the tubing (e.g. sheath body) wherein the mixture is at a subambient temperature. A syringe, for example a 1 ml syringe, can be connected to the syringe tube and cartridge assembly. A drop of catalyst appropriate for the silicone, for example MED-6385 curing agent, can be placed into the syringe and the syringe is then filled with the uncured mixture of silicone and latanoprost. The mixture, i.e., mixture of the uncured silicone and latanoprost still liquid enough to flow or pump, can be chilled to subambient temperatures. For example, the mixture can be chilled to temperatures of less than 20.degree. C. For example, the mixtures can be chilled to 0.degree. C., or to -25.degree. C. In a particular embodiment, the mixture is chilled to between about zero and 5.degree. C.
[0225] The polyimide tube is injected with the drug/matrix mixture until the tube is filled. The tube and associated apparatus can also be chilled to maintain the subambient temperature of the mixture throughout the process of filling or injecting the sheath with the mixture. In various embodiments, the polyimide tube, or sheath, is filled with the drug matrix mixture under pressure, for example through use of a high pressure pump. For instance, the drug/matrix mixture, such as can be obtained in mixtures of latanoprost with MED-6385 Part A to which amounts of catalyst Part B have been added, can be pumped into the tube under at least about 40 psi pressure. The tube can be filled at any suitable rate, but preferably, at rates of less than about 0.5 linear cm/sec. Without wishing to be bound by a theory, it is believed that filling the tube relatively rapidly under a relatively high head of pressure can reduce the degree of phase separation of the substantially immiscible latanoprost oil and silicone monomer material, such that upon polymerization ("curing") to provide the final silicone polymeric product, the latanoprost droplets are finely dispersed in the solid matrix in which they are only slightly soluble.
[0226] Curing takes place in the presence of the catalyst ("Part B") of the NuSil MED-6385, and can be carried out at temperatures of at least about 40.degree. C., at relative humidity (RH) of at least about 80%, or both. Curing can be initiated directly after filling the tube and clamping the ends of the filled tube to prevent the formation of voids and loss of the precursor material from the tube ends.
[0227] After curing, which can be complete in about 16-24 hours at 40.degree. C. and 80% RH, the clamps can be removed from the ends of the tubing, as the silicone is fully set up. The tubing can then be cut into sections of suitable length for use as drug cores, for example, lengths of about 1 mm.
[0228] When the extrusion is carried out at subambient temperatures, small and more uniform inclusions of the therapeutic agent can result. For example, when the agent is latanoprost, a liquid at room temperature, extrusion at -5.degree. C. provides significantly smaller and more uniform inclusion droplets. In an example, cold extrusion yielded a drug core comprising a silicone matrix with latanoprost droplets of average diameter of 6 .mu.m, with a standard deviation of diameter of 2 .mu.m. In comparison, an extrusion carried out at room temperature yielded a drug core comprising a silicone matrix with latanoprost droplets of average diameter of 19 .mu.m, with a standard deviation of droplet diameter of 19 .mu.m. It is apparent that the cold extrusion technique provides smaller, more uniform inclusions than does extrusion at room temperature. This in turn results in a more uniform concentration of drug throughout the core, or the insert containing the core.
[0229] The final step in making the present lacrimal implants comprises inserting the drug core, cut to an appropriate length and sealed on one end, into the cavity of the lacrimal implant. This can be done manually or with the aid of a machine.
D) Release of Latanoprost or Other Intraocular Pressure-Reducing Therapeutic Agent(s) at Effective Levels
[0230] The rate of release of latanoprost or other intraocular pressure-reducing therapeutic agent(s) can be related to the concentration of latanoprost or other intraocular pressure-reducing therapeutic agent(s) dissolved in the drug core. In some embodiments, the drug core comprises non-therapeutic agents that are selected to provide a desired solubility of the latanoprost or other intraocular pressure-reducing therapeutic agent(s) in the drug core. The non-therapeutic agent of the drug core can comprise polymers as described herein, and additives. A polymer of the core can be selected to provide the desired solubility of the latanoprost or other intraocular pressure-reducing therapeutic agent(s) in the matrix. For example, the core can comprise hydrogel that may promote solubility of hydrophilic treatment agent. In some embodiments, functional groups can be added to the polymer to provide the desired solubility of the latanoprost or other intraocular pressure-reducing therapeutic agent(s) in the matrix. For example, functional groups can be attached to silicone polymer.
[0231] Additives may be used to control the concentration of latanoprost or other intraocular pressure-reducing therapeutic agent(s) by increasing or decreasing solubility of the latanoprost or other intraocular pressure-reducing therapeutic agent(s) in the drug core so as to control the release kinetics of the latanoprost or other intraocular pressure-reducing therapeutic agent(s). The solubility may be controlled by providing appropriate molecules or substances that increase or decrease the content of latanoprost or other intraocular pressure-reducing therapeutic agent(s) in the matrix. The latanoprost or other intraocular pressure-reducing therapeutic agent(s) content may be related to the hydrophobic or hydrophilic properties of the matrix and latanoprost or other intraocular pressure-reducing therapeutic agent(s). For example, surfactants and salts can be added to the matrix and may increase the content of hydrophobic latanoprost in the matrix. In addition, oils and hydrophobic molecules can be added to the matrix and may increase the solubility of hydrophobic treatment agent in the matrix.
[0232] Instead of or in addition to controlling the rate of migration based on the concentration of latanoprost or other intraocular pressure-reducing therapeutic agent(s) dissolved in the matrix, the surface area of the drug core can also be controlled to attain the desired rate of drug migration from the core to the target site. For example, a larger exposed surface area of the core will increase the rate of migration of the treatment agent from the drug core to the target site, and a smaller exposed surface area of the drug core will decrease the rate of migration of the latanoprost or other intraocular pressure-reducing therapeutic agent(s) from the drug core to the target site. The exposed surface area of the drug core can be increased in any number of ways, for example by any of castellation of the exposed surface, a porous surface having exposed channels connected with the tear or tear film, indentation of the exposed surface, protrusion of the exposed surface. The exposed surface can be made porous by the addition of salts that dissolve and leave a porous cavity once the salt dissolves. Hydrogels may also be used, and can swell in size to provide a larger exposed surface area. Such hydrogels can also be made porous to further increase the rate of migration of the latanoprost or other intraocular pressure-reducing therapeutic agent(s).
[0233] Further, an implant may be used that includes the ability to release two or more drugs in combination, such as the structure disclosed in U.S. Pat. No. 4,281,654. For example, in the case of glaucoma treatment, it may be desirable to treat a patient with multiple prostaglandins or a prostaglandin and a cholinergic agent or an adrenergic antagonist (beta blocker), such as Alphagan.RTM., or latanoprost and a carbonic anhydrase inhibitor.
[0234] In addition, drug impregnated meshes may be used such as those disclosed in U.S. Patent Publication No. 2002/0055701 or layering of biostable polymers as described in U.S. Patent Publication No. 2005/0129731, the disclosures of which are incorporated herein in their entirety. Certain polymer processes may be used to incorporate latanoprost or other intraocular pressure-reducing therapeutic agent(s) into the devices of the present invention; such as so-called "self-delivering drugs" or PolymerDrugs (Polymerix Corporation, Piscataway, N.J.) are designed to degrade only into therapeutically useful compounds and physiologically inert linker molecules, further detailed in U.S. Patent Publication No. 2005/0048121, hereby incorporated by reference in its entirety. Such delivery polymers may be employed in the devices of the present invention to provide a release rate that is equal to the rate of polymer erosion and degradation and is constant throughout the course of therapy. Such delivery polymers may be used as device coatings or in the form of microspheres for a drug depot injectable (such as a reservoir of the present invention). A further polymer delivery technology may also be configured to the devices of the present invention such as that described in U.S. Patent Publication No. 2004/0170685, and technologies available from Medivas (San Diego, Calif.).
[0235] In specific embodiments, the drug core matrix comprises a solid material, for example silicone, that encapsulates inclusions of the latanoprost or other intraocular pressure-reducing therapeutic agent(s). The drug comprises molecules which are very insoluble in water and slightly soluble in the encapsulating drug core matrix. The inclusions encapsulated by the drug core can be micro-particles having dimensions from about 1 micrometer to about 100 micrometers across. The drug inclusions can comprise droplets of oil, for example latanoprost oil. The drug inclusions can dissolve into the solid drug core matrix and substantially saturate the drug core matrix with the drug, for example dissolution of latanoprost oil into the solid drug core matrix. The drug dissolved in the drug core matrix is transported, often by diffusion, from the exposed surface of the drug core into the tear film. As the drug core is substantially saturated with the drug, in many embodiments the rate limiting step of drug delivery is transport of the drug from the surface of the drug core matrix exposed to the tear film. As the drug core matrix is substantially saturated with the drug, gradients in drug concentration within the matrix are minimal and do not contribute significantly to the rate of drug delivery. As surface area of the drug core exposed to the tear film is nearly constant, the rate of drug transport from the drug core into the tear film can be substantially constant. Naturally occurring surfactants may affect the solubility of the latanoprost or other intraocular pressure-reducing therapeutic agent(s) in water and molecular weight of the drug can affect transport of the drug from the solid matrix to the tear. In many embodiments, the latanoprost or other intraocular pressure-reducing therapeutic agent(s) is nearly insoluble in water and has a solubility in water of about 0.03% to 0.002% by weight and a molecular weight from about 400 grams/mol. to about 1200 grams/mol.
[0236] In many embodiments the latanoprost or other intraocular pressure-reducing therapeutic agent(s) has a very low solubility in water, for example from about 0.03% by weight to about 0.002% by weight, a molecular weight from about 400 grams per mole (g/mol) to about 1200 g/mol, and is readily soluble in an organic solvent. Latanoprost is a liquid oil at room temperature, and has an aqueous solubility of 50 micrograms/mL in water at 25 degrees C., or about 0.005% by weight and a M.W. of 432.6 g/mol.
[0237] Naturally occurring surfactants in the tear film, for example surfactant D and phospholipids, may affect transport of the drug dissolved in the solid matrix from the core to the tear film. In some embodiments the drug core can be configured in response to the surfactant in the tear film to provide sustained delivery of latanoprost or other intraocular pressure-reducing therapeutic agent(s) into the tear film at therapeutic levels. For example, empirical data can be generated from a patient population, for example 10 patients whose tears are collected and analyzed for surfactant content. Elution profiles in the collected tears for a drug that is sparingly soluble in water can also be measured and compared with elution profiles in buffer and surfactant such that an in vitro model of tear surfactant is developed. An in vitro solution with surfactant based on this empirical data can be used to adjust the drug core in response to the surfactant of the tear film.
[0238] The drug cores may also be modified to utilize carrier vehicles such as nanoparticles or microparticles depending on the size of the molecule to be delivered such as latent-reactive nanofiber compositions for composites and nanotextured surfaces (Innovative Surface Technologies, LLC, St. Paul, Minn.), nanostructured porous silicon, known as BioSilicon.RTM., including micron sized particles, membranes, woven fivers or micromachined implant devices (pSividia, Limited, UK) and protein nanocage systems that target selective cells to deliver a drug (Chimeracore).
[0239] In many embodiments, the drug insert comprises of a thin-walled polyimide tube sheath with a drug core comprising latanoprost dispersed in Nusil 6385 (MAF 970), a medical grade solid silicone that serves as the matrix for drug delivery. The distal end of the drug insert is sealed with a cured film of solid Loctite 4305 medical grade adhesive. The drug insert may be placed within the bore of the punctal implant, the Loctite 4305 adhesive does not come into contact with either tissue or the tear film. The inner diameter of the drug insert can be 0.32 mm; and the length can be 0.95 mm. At least four latanoprost concentrations in the finished drug product can be employed: Drug cores can comprise 3.5, 7, 14 or 21 micrograms latanoprost, with percent by weight concentrations of 5, 10, 20, or 30% respectively. Assuming an overall elution rate of approximately 100 ng/day, the drug core comprising 14 micrograms of latanoprost is configured to deliver drug for approximately at least 100 days, for example 120 days. The overall weight of the drug core, including latanoprost or other intraocular pressure-reducing therapeutic agent(s), can be about 70 micrograms. The weight of the drug insert including the polyimide sleeve can be approximately 100 micrograms. In an embodiment, the drug core can comprise 46 micrograms of latanoprost, and in another embodiment, the drug core can comprise 95 micrograms of latanoprost.
[0240] In many embodiments, the drug core may elute with an initial elevated level of latanoprost or other intraocular pressure-reducing therapeutic agent(s) followed by substantially constant elution of the latanoprost or other intraocular pressure-reducing therapeutic agent(s). In many instances, an amount of latanoprost or other intraocular pressure-reducing therapeutic agent(s) released daily from the core may be below the therapeutic levels and still provide a benefit to the patient. An elevated level of eluted latanoprost or other intraocular pressure-reducing therapeutic agent(s) can result in a residual amount of latanoprost or other intraocular pressure-reducing therapeutic agent(s) or residual effect of the latanoprost or other intraocular pressure-reducing therapeutic agent(s) that is combined with a sub-therapeutic amount of latanoprost or other intraocular pressure-reducing therapeutic agent(s) to provide relief to the patient. In embodiments where therapeutic level is about 80 ng per day, the device may deliver about 100 ng per day for an initial delivery period. The extra 20 ng delivered per day can have a beneficial effect when latanoprost or other intraocular pressure-reducing therapeutic agent(s) is released at levels below the therapeutic level, for example at 60 ng per day. As the amount of drug delivered can be precisely controlled, an initial elevated dose may not result in complications or adverse events to the patient.
E) Clinical Use of the Drug Delivery System to Treat Glaucoma and/or Ocular Hypertension
[0241] Ocular hypertension (OH) and primary open angle glaucoma (POAG) are caused by a build-up of aqueous humor in the anterior chamber primarily due to the eye's inability to properly drain aqueous fluid. The ciliary body, situated at the root of the iris, continuously produces aqueous humor. It flows into the anterior chamber and then drains via the angle between the cornea and iris through the trabecular meshwork and into a channel in the sclera. In the normal eye, the amount of aqueous humor being produced is equal to the amount that is draining out. However, in an eye in which this mechanism is compromised, intraocular pressure (IOP) rises. Elevated IOP represents a major risk factor for glaucomatous field loss. Results from several studies indicate that early intervention targeted at lowering intraocular pressure retards the progression of optic nerve damage and loss of visual fields that lead to decreased vision and blindness.
[0242] As described above, first line treatment for treating OAG and/or OH is the use of eye drops, such as Xalatan. However, numerous studies have been published showing high noncompliance by patients using eye drops for treatment of various ocular disorders. One study showed only 64% of patients used the eye drops as directed (Winfield A J, et al. A study of the causes of non-compliance by patients prescribed eyedrops. Br J Ophthalmol. 1990 August; 74(8):477-80). Another study showed that 41% of patients using eye drops for glaucoma missed six or more doses over a 30-day period (Norell S E, Granstrom P A. Self-medication with pilocarpine among outpatients in a glaucoma clinic. Br J Ophthalmol. 1980 February; 64(2):137-41).
[0243] In certain embodiments, the invention described herein provides methods to treat glaucoma that avoid the problem of noncompliance associated with eye drop administration. In some embodiments, the methods of the invention reduce patient noncompliance significantly compared to eye drop administration, e.g., by at least about 10%, at least about 20%, at least about 30%, at least about 40%, at least about 50%, at least about 60%, at least about 70%, at least about 80%, or at least about 90%. In some embodiments, overall patient noncompliance with the methods described herein is about 5%, about 10%, about 15%, about 20%, or about 25%.
[0244] Patient noncompliance may occur if an implant of the invention is intentionally removed by a patient or if the patient does not seek reinsertion of the implant after such implant has been unintentionally lost from the punctum of the patient. Patient compliance is considered to be met if the implant is intentionally removed and the patient seeks reinsertion within less than about 48 hours. Patient compliance is also considered to be met if the implant is intentionally removed and the patient seeks reinsertion within less than about 24 hours of removal or loss of the implant.
[0245] Implicit in the methods to treat OAG and/or OH to avoid patient non-compliance is the comparable efficacy of the present drug delivery system comprising lacrimal implants to the use of eye drops. Lacrimal implants to treat ocular disease have been in development for many years by the applicants and others with limited success. However, applicants demonstrate for the first time herein a clinically meaningful reduction in IOP over the treatment period (e.g. between 4 weeks and 12 weeks) using the present lacrimal implants to administer latanoprost.
[0246] In certain embodiments, the invention described herein provides methods to treat glaucoma, elevated intraocular pressure, and glaucoma-associated elevated intraocular pressure with a therapeutic agent. Examples of glaucoma treatable according to the present invention include primary open angle glaucoma, normal intraocular tension glaucoma, hypersecretion glaucoma, ocular hypertension, acute angle-closure glaucoma, chronic closed angle glaucoma, combined-mechanism glaucoma, corticosteroid glaucoma, amyloid glaucoma, neovascular glaucoma, malignant glaucoma, capsular glaucoma, plateau iris syndrome and the like.
[0247] In one embodiment, the present disclosure provides methods of treating a patient with open angle glaucoma (OAG) and/or ocular hypertension (OH) in an eye. In a further embodiment, the present disclosure provides methods of treating a patient with Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye by reducing intraocular pressure (IOP) in the eye.
[0248] In certain embodiments the treatment period is at least four (4) weeks, and can be up to twelve (12) week or longer, wherein the therapeutic agent is released in a therapeutically effective dose from a lacrimal implant on a sustained basis over the treatment period.
[0249] In one embodiment, the implants and methods of the invention provide a 90-day course of treatment. In other embodiments, the implants and methods of the invention provide a 60-day course of treatment. In still other embodiments, the implants and methods of the invention provide a 45-day course of treatment. In still other embodiments, the implants and methods of the invention provide a 30-day course of treatment, depending upon the disease to be treated and the therapeutic agent to be delivered. Other embodiments include four weeks, five weeks, six weeks, seven weeks, eight weeks, nine weeks, ten weeks, eleven weeks, or twelve weeks of treatment.
[0250] In certain embodiments, the methods comprise reducing the intraocular pressure (IOP) during the treatment period. In one embodiment, reduction in intraocular pressure (IOP) is between about 10% and 24% from baseline over the treatment period. In particular embodiments, the percentage reduction or decrease in intraocular pressure (IOP) is approximately 23%, approximately 22%, approximately 21%, or approximately 20% from baseline. In other particular embodiments, the present methods result in a percentage reduction or decrease in intraocular pressure (IOP) of at least 24%, at least 23%, at least 22%, at least 21%, at least 20%, at least 15%, or at least 10% from baseline.
[0251] In certain embodiments, the methods of the invention result in a reduction in intraocular pressure from baseline over a treatment period of about 4, mm Hg, about 5 mm Hg, about 6 mm Hg, or about 7 mm Hg. In certain embodiments, the methods of the invention result in a reduction in intraocular pressure from baseline of at least 4 mm Hg, at least 5 mm Hg, at least 6 mm Hg, or at least 7 mm Hg. In some embodiments, intraocular pressure is reduced to less than or equal to 24 mm Hg, less than or equal to 23 mm Hg, less than or equal to 22 mm Hg, less than or equal to 21 mm Hg, less than or equal to 20 mm Hg, less than or equal to 19 mm Hg, less than or equal to 18 mm Hg, or less than or equal to 17 mm Hg, or less than or equal to 16 mm Hg, less than or equal to 15 mm Hg, less than or equal to 14 mm Hg, or less than or equal to 13 mm Hg.
[0252] In one embodiment, the invention provides a method of treating glaucoma and/or ocular hypertension with a punctal plug delivering a therapeutic agent effective against these conditions in a sustained release manner. The release occurs at a rate and in an amount sufficient to be therapeutically useful. In various embodiments, the therapeutic agent is a prostaglandin, e.g., latanoprost.
[0253] In an exemplary embodiment, the condition treated is primary open-angle glaucoma (POAG) and ocular hypertension (OH) with a punctual plug of the invention in which a sustained release formulation of a prostaglandin derivative is provided. In this method one to four punctual plugs may be inserted per patient. Exemplary punctal plugs are formulated with from about 40 .mu.g to about 115 .mu.g of prostaglandin. In various embodiments, the prostaglandin is latanoprost. In various embodiments, the plugs are formulated with either 46 .mu.g or 95 .mu.g of latanoprost (See, Tables 2 and 3) so that a dosage which is a member selected from 46 .mu.g, 92 .mu.g, 95 .mu.g, 141 .mu.g or 190 .mu.g is administered to each eye. In one embodiment 141 .mu.g of latanoprost was administered to an individual eye. In another embodiment, 190 .mu.g of latanoprost was administered to an individual eye. In another embodiment, 95 .mu.g of latanoprost was administered to an individual eye. A patient may have the same amount of latanoprost administered to each eye or the patient may have a different amount administered to each eye.
[0254] The implants described herein may be inserted into the superior (upper) punctum, the inferior (lower) punctum, or both, and may be inserted into one or both eyes of the subject. Without wishing to be bound by a theory, the data presented in the figures and examples appear to demonstrate that the placement and configuration of the lacrimal implants may contribute to the reduction of IOP over the treatment period. In previously published studies, the lacrimal implants were only inserted into the lower punctum, and those studies, despite dose escalation (FIG. 16) and with a constant elution of drug over the treatment period (FIG. 17) were unable to show significant reduction in IOP over the treatment period. See Example 2. Thus, in certain embodiments at least the upper punctum is inserted with a present lacrimal implant. Surprisingly though, this lacrimal implant need not comprise a therapeutic agent (See Example 6 and FIGS. 19 and 27). In this instance a blank lacrimal implant is inserted into the upper punctum and a lacrimal implant comprising a therapeutic agent (e.g. 95 .mu.g of latanoprost) is inserted into the lower punctum. This configuration demonstrated a mean reduction in IOP from baseline of 5.17 mm Hg at week twelve (FIG. 23 and Table 8), while previous studies with no lacrimal implant in the upper punctum and the same concentration of latanoprost in the lower puntum lacrimal implant demonstrated only a reduction of less than about 4.0 mm Hg from baseline at week 2 (FIG. 16).
[0255] In certain embodiments, the method for treating OAG and/or OH comprises inserting a lacrimal implant into at least the upper punctum. In one embodiment, the lacrimal implant comprises a therapeutic agent (e.g. latanoprost). In another embodiment, the lacrimal implant does not comprise a therapeutic agent for treating OAG and/or OH. In this instance, a lacrimal implant is inserted into the lower punctum comprising a therapeutic agent (e.g. latanoprost).
[0256] Thus, the present methods comprise inserting at least a lacrimal implant into an upper punctum wherein a number of different configurations are contemplated resulting in significant reduction of IOP over the treatment period. In one embodiment, the method for treating OAG and/or OH comprises inserting a lacrimal implant into the upper punctum comprising a therapeutic agent and inserting a lacrimal implant into the lower punctum comprising a therapeutic agent. In another embodiment, the method of treating OAG and/or OH comprises inserting a lacrimal implant into the upper punctum that does not comprise a therapeutic agent for lowering IOP and inserting a lacrimal implant into the lower punctum that comprises a therapeutic agent for treating OAG and/or OH. In yet another embodiment, the present method for treating OAG and/or OH comprises inserting a lacrimal implant into the upper punctum comprising a therapeutic agent wherein no lacrimal implant is inserted into the lower punctum. In each of the above embodiments, reference to an upper and lower punctum is referring to the same eye. Each eye may have the same configuration of lacrimal implant inserted or different; each eye is treated separately for OAG and/or OH.
[0257] In a particular embodiment, the method of treating OAG and/or OH in an eye comprises providing a first lacrimal implant comprising a sustained release formulation of a therapeutic agent for treating OAG or OH; providing a second lacrimal implant that does not comprise the therapeutic agent; and inserting the first and second lacrimal implant through an upper and lower punctum into a lacrimal canaliculus of the same eye wherein the therapeutic agent is released in a therapeutically effective dose from the first lacrimal implant on a sustained basis over at least four (4) weeks. In one aspect, the therapeutic agent is latanoprost. In another aspect, the dose of latanoprost administered to the eye is about 95 .mu.g. In yet another aspect, the therapeutic agent is released in a therapeutically effective dose from the first lacrimal implant on a sustained basis over at least twelve (12) weeks. In this particular embodiment, it was surprisingly found that the IOP was reduced by about 5.0 mm Hg at week 4 and about at least 4.0 mm Hg at week 12.
[0258] In another particular embodiment, the method of treating OAG and/or OH in an eye comprises providing a lacrimal implant comprising a sustained release formulation of a therapeutic agent for treating OAG or OH; and inserting the lacrimal implant through an upper punctum into a lacrimal canaliculus of the eye wherein the therapeutic agent is released in a therapeutically effective dose from the first lacrimal implant on a sustained basis over at least four (4) weeks. In one aspect, the therapeutic agent is latanoprost. In another aspect, the dose of latanoprost administered to the eye is about 95 .mu.g. In yet another aspect, the therapeutic agent is released in a therapeutically effective dose from the first lacrimal implant on a sustained basis over at least twelve (12) weeks. In this particular embodiment, it was surprisingly found that the IOP was reduced by about at least 4.0 mm Hg at week 4 and about at least 4.0 mm Hg at week 12.
[0259] In some embodiments, the therapeutic agent is released to the eye over a sustained period of time. In an embodiment, the sustained period of time is at least about 28 days, about 45 days, about 60 days or at least about 90 days. In some embodiments, the method comprises inserting through a punctum an implant having a body and a drug core so that the drug core is retained near the punctum. In some embodiments, the method comprises inserting through a punctum an implant having a body dispersed throughout with a therapeutic agent. In some embodiments, an exposed surface of the drug core or agent dispersed body located near the proximal end of the implant contacts the tear or tear film fluid and the latanoprost or other intraocular pressure-reducing therapeutic agent(s) migrates from the exposed surface to the eye over a sustained period of time while the drug core and body is at least partially retained within the punctum. In an exemplary embodiment, a method of treating an eye with latanoprost or other intraocular pressure-reducing therapeutic agent(s) is provided, the method comprising inserting through a punctum into a canalicular lumen an implant having an optional retention structure so that the implant body is anchored to a wall of the lumen by the retention structure. The implant releases effective amounts of latanoprost or other intraocular pressure-reducing therapeutic agent(s) from a drug core or other agent supply into a tear or tear film fluid of the eye. In some embodiments, the drug core may be removed from the retention structure while the retention structure remains anchored within the lumen. A replacement drug core can then be attached to the retention structure while the retention structure remains anchored. At least one exposed surface of the replacement drug core releases latanoprost or other intraocular pressure-reducing therapeutic agent(s) at therapeutic levels over a sustained period.
[0260] A replacement implant, or in other embodiments, a replacement drug core which can in some embodiments be attached to or include its own retention structure, can be attached to the retention structure approximately every 30 days, approximately every 60 days or approximately every 90 days to result in continuous release of the drug to the eye for a period of time of approximately 180 days, approximately 270 days, approximately 360 days, approximately 450 days, approximately 540 days, approximately 630 days, approximately 720 days, approximately 810 days or approximately 900 days. In some embodiments, a replacement implant can be inserted into the punctum approximately every 30 days, approximately every 60 days or approximately every 90 days to achieve release of the drug to the eye for extended periods of time, including up to about 180 days, about 270 days, about 360 days, about 450 days, about 540 days, about 630 days, about 720 days, about 810 days or about 900 days.
[0261] In other embodiments, a method for treating an eye with latanoprost or other intraocular pressure-reducing therapeutic agent(s) is provided, the method comprising inserting a drug core or other implant body at least partially into at least one punctum of the eye. The drug core may or may not be associated with a separate implant body structure. The drug core or agent-impregnated implant body provides sustained release delivery of latanoprost or other intraocular pressure-reducing therapeutic agent(s) at therapeutic levels. In some embodiments, the sustained release delivery of latanoprost or other intraocular pressure-reducing therapeutic agent(s) continues for up to 90 days.
[0262] In exemplary embodiments, a method for treating an eye with latanoprost or other intraocular pressure-reducing therapeutic agent(s) is provided, the method comprising inserting a distal end of an implant into at least one punctum and into at least one lacrimal canaliculus of the eye. In some embodiment, a retention structure of the implant is fitted so as to inhibit expulsion of the implant. The expansion of the retention structure can help to occlude a flow of tear fluid through the punctum. In some embodiments, the implant is configured such that, when implanted, at least 45 degree angled intersection exists between a first axis, defined by a proximal end of the implant, and a second axis, defined by the distal end of the implant, to inhibit expulsion of the implant. Latanoprost or other intraocular pressure-reducing therapeutic agent(s) is delivered from a proximal end of the implant to the tear fluid adjacent the eye. Delivery of the latanoprost or other intraocular pressure-reducing therapeutic agent(s) is inhibited distally of the proximal end.
[0263] The methods of the invention provide sustained release of latanoprost or other intraocular pressure-reducing therapeutic agent(s). In some embodiments, the active agent is released from the implant for at least four weeks, at least five weeks, at least six weeks, at least seven weeks, at least eight weeks, at least nine weeks, at least ten weeks, at least eleven weeks, at least twelve weeks, at least thirteen weeks, at least fourteen weeks, at least fifteen weeks, or at least sixteen weeks. In some embodiments, the therapeutic agent is latanoprost. In an embodiment, the latanoprost or other intraocular pressure-reducing therapeutic agent(s) is released for at least twelve weeks. In an exemplary embodiment, the methods of treatment according to the present invention comprise an adjunctive therapy with a latanoprost-delivering eye drop solution, for example, Xalatan.RTM..
[0264] The amount of latanoprost or other intraocular pressure-reducing therapeutic agent(s) associated with the implant may vary depending on the desired therapeutic benefit and the time during which the device is intended to deliver the therapy. Since the devices of the present invention present a variety of shapes, sizes and delivery mechanisms, the amount of drug associated with the device will depend on the particular disease or condition to be treated, and the dosage and duration that is desired to achieve the therapeutic effect. Generally, the amount of latanoprost or other intraocular pressure-reducing therapeutic agent(s) is at least the amount of drug that, upon release from the device, is effective to achieve the desired physiological or pharmacological local or systemic effects.
[0265] Certain embodiments of the implants of the present invention can be configured to provide, in combination with each other or separately, delivery of latanoprost or other intraocular pressure-reducing therapeutic agent(s) at daily rates that are greater than or equivalent to the therapeutically effective drop form of treatment. Other embodiments of the implants of the present invention can be configured to provide, in combination with each other or separately, delivery of latanoprost or other intraocular pressure-reducing therapeutic agent(s) at daily rates that enable comparable clinical outcomes to that of daily administered eye drops. Other embodiments of the implants of the present invention can be configured to provide delivery of latanoprost or other intraocular pressure-reducing therapeutic agent(s) at daily rates that exceed the therapeutically effective drop form of treatment.
[0266] For comparison purposes, standard treatment, i.e., the recommended daily total dose, with drops, such as Xalatan.RTM. drops, delivers about 1.5 micrograms of latanoprost to the eye all at once, assuming a 35 microliter drop volume. In embodiments of the present invention, the sustained release of at least 1.5 micrograms of latanoprost per day can be administered. For example, in an embodiment, a sustained release ophthalmic drug delivery system is configured to release, on a sustained basis over the course of 24 hours to the eye, a total amount of latanoprost from a combination of a first lacrimal implant, located in a lower punctum of the eye, and a second lacrimal implant, located in an upper punctum of the same eye, that is greater than or equal to the recommended daily total dose of latanoprost that is in Xalatan.RTM. drops (i.e., eye drop form). In other embodiments, at least two times the recommended daily total dose of latanoprost that is in Xalatan.RTM. drops may be release by a combination of the first lacrimal implant and the second lacrimal implant that are in the lower punctum and the upper punctum, respectively, of the same eye. In an embodiment, both eyes of the patient may be treated with two lacrimal implants at the same time.
[0267] Methods of inserting and removing the implant are known to those of skill in the art. For instance, tools for insertion and removal/extraction of implants are described in U.S. Patent Publication No. 2009/0105749 (filed Sep. 5, 2008 and entitled Insertion and Extraction Tools for Lacrimal Implants), the disclosure of which is incorporated herein in its entirety. Generally, for placement, the size of a punctal implant to be used may be determined by using suitable magnification or, if provided, using a sizing tool that accompanies the punctal implant. The patient's punctum may be dilated if necessary to fit the punctal implant. A drop of lubricant may be applied if necessary to facilitate placement of the implant into the punctum. Using an appropriate placement instrument, the implant may be inserted into the superior or inferior punctum of the eye. After placement, the cap of the implant may be visible. This process may be repeated for the patient's other eye. For removal of the implant, small surgical forceps may be used to securely grasp the implant at the tube section below the cap. Using a gentle tugging motion the implant may be gently retrieved.
F) Kits
[0268] The present invention also provides methods that utilize kits that, in an exemplary embodiment, include one, two, three or four implants of use in the methods of the invention. In an exemplary embodiment, the implants are sterilized. In various embodiments, there is also provided an insertion tool. An exemplary insertion tool of use in this embodiment is set forth herein. In various embodiments, at least one implant is engaged with the insertion tool by engaging the pin of the tool (760) with the bore of the implant (385). In various embodiments, the tool is sterilized. In an exemplary embodiment, the elements of the kit are packaged together with one or more of a set of instructions for installing the implant in the punctum, a topical anesthetic, an administration device for the topical anesthetic or another component of use in installing the implant in the punctum.
G) Specific Embodiments
[0269] E1. A method of treating a patient with Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye, comprising: providing a unit dosage of about 95 .mu.g of latanoprost to an eye over a treatment period, wherein the latanoprost is administered from a lacrimal implant comprising a sustained release formulation of the 95 ug of latanoprost and the latanoprost is released in a therapeutically effective dose from the lacrimal implant over the treatment period, with the proviso that the lacrimal implant is inserted into an upper punctum of the eye and a lower punctum of the eye is open or has inserted a blank lacrimal implant that does not comprise latanoprost.
[0270] E2. A method of treating a patient with Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye, comprising: providing a unit dosage of about 95 .mu.g of latanoprost to an eye over a treatment period, wherein the latanoprost is administered from a lacrimal implant comprising a sustained release formulation of the 95 .mu.g of latanoprost and the latanoprost is released in a therapeutically effective dose from the lacrimal implant over the treatment period, with the proviso that the lacrimal implant is inserted into a lower punctum and a blank lacrimal implant that does not comprise latanoprost is inserted in an upper punctum.
[0271] E3. A method of treating a patient with Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye by reducing intraocular pressure (IOP) in the eye, comprising: providing a unit dosage of about 95 .mu.g of latanoprost to an eye over a treatment period of at least 4 weeks, wherein the latanoprost is administered from a lacrimal implant comprising a sustained release formulation of the 95 .mu.g of latanoprost and the latanoprost is released in a therapeutically effective dose from the lacrimal implant over the treatment period, wherein the IOP is reduced by at least 4 mm Hg from a baseline at week 4.
[0272] E4. A method of treating a patient with Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye by reducing intraocular pressure (IOP) in the eye, comprising: providing a unit dosage of about 95 .mu.g of latanoprost to an eye over a treatment period of at least 8 weeks, wherein the latanoprost is administered from a lacrimal implant comprising a sustained release formulation of the 95 .mu.g of latanoprost and the latanoprost is released in a therapeutically effective dose from the lacrimal implant over the treatment period, wherein the IOP is reduced by at least 4 mm Hg from a baseline at week 8.
[0273] E5. A method of treating a patient with Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye by reducing intraocular pressure (IOP) in the eye, comprising: providing a unit dosage of about 95 .mu.g of latanoprost to an eye over a treatment period of at least 12 weeks, wherein the latanoprost is administered from a lacrimal implant comprising a sustained release formulation of the 95 .mu.g of latanoprost and the latanoprost is released in a therapeutically effective dose from the lacrimal implant over the treatment period, wherein the IOP is reduced by at least 4 mm Hg from a baseline at week 12.
[0274] E6. A method of treating a patient with Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye by reducing intraocular pressure (IOP) in the eye, comprising: providing a unit dosage of about between 140 and 200 .mu.g of latanoprost to an eye over a treatment period of at least 4 weeks, wherein the latanoprost is administered from a lacrimal implant comprising a sustained release formulation of the latanoprost and the latanoprost is released in a therapeutically effective dose from the lacrimal implant over the treatment period, wherein the IOP is reduced by at least 5 mm Hg from a baseline at week 4.
[0275] E7. A method of treating a patient with Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye by reducing intraocular pressure (IOP) in the eye, comprising: providing a unit dosage of about between 140 and 200 .mu.g of latanoprost to an eye over a treatment period of at least 8 weeks, wherein the latanoprost is administered from a lacrimal implant comprising a sustained release formulation of the latanoprost and the latanoprost is released in a therapeutically effective dose from the lacrimal implant over the treatment period, wherein the IOP is reduced by at least 5 mm Hg from a baseline at week 8.
[0276] E8. A method of treating a patient with Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye by reducing intraocular pressure (IOP) in the eye, comprising: providing a unit dosage of about between 140 and 200 .mu.g of latanoprost to an eye over a treatment period of at least 12 weeks, wherein the latanoprost is administered from a lacrimal implant comprising a sustained release formulation of the latanoprost and the latanoprost is released in a therapeutically effective dose from the lacrimal implant over the treatment period, wherein the IOP is reduced by at least 5 mm Hg from a baseline at week 12.
[0277] E9. A method of treating a patient with Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye by reducing intraocular pressure (IOP) in the eye, comprising: providing a unit dosage of about 190 .mu.g of latanoprost to an eye over a treatment period of at least 4 weeks, wherein the latanoprost is administered from a lacrimal implant comprising a sustained release formulation of the latanoprost and the latanoprost is released in a therapeutically effective dose from the lacrimal implant over the treatment period, wherein the IOP is reduced by at least 5 mm Hg from a baseline at week 4.
[0278] E10. A method of treating a patient with Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye by reducing intraocular pressure (IOP) in the eye, comprising: providing a unit dosage of about 190 .mu.g of latanoprost to an eye over a treatment period of at least 8 weeks, wherein the latanoprost is administered from a lacrimal implant comprising a sustained release formulation of the latanoprost and the latanoprost is released in a therapeutically effective dose from the lacrimal implant over the treatment period, wherein the IOP is reduced by at least 5 mm Hg from a baseline at week 8.
[0279] E11. A method of treating a patient with Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye by reducing intraocular pressure (IOP) in the eye, comprising: providing a unit dosage of about 190 .mu.g of latanoprost to an eye over a treatment period of at least 12 weeks, wherein the latanoprost is administered from a lacrimal implant comprising a sustained release formulation of the latanoprost and the latanoprost is released in a therapeutically effective dose from the lacrimal implant over the treatment period, wherein the IOP is reduced by at least 4 mm Hg from a baseline at week 12.
[0280] E12. A method of treating a patient with Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye by reducing intraocular pressure (IOP) in the eye, comprising: providing a unit dosage of about 141 .mu.g of latanoprost to an eye over a treatment period of at least 8 weeks, wherein the latanoprost is administered from a lacrimal implant comprising a sustained release formulation of the latanoprost and the latanoprost is released in a therapeutically effective dose from the lacrimal implant over the treatment period, wherein the IOP is reduced by at least 5 mm Hg from a baseline at week 8.
[0281] E13. A method of treating a patient with Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye, comprising: (a) providing a first lacrimal implant comprising a sustained release formulation of a therapeutic agent for treating OAG or OH; (b) providing a second lacrimal implant that does not comprise the therapeutic agent; and (c) inserting the first and second lacrimal implant through an upper and lower punctum into a lacrimal canaliculus of the same eye wherein the therapeutic agent is released in a therapeutically effective dose from the first lacrimal implant on a sustained basis over at least four (4) weeks.
[0282] E14. A method of treating a patient with Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye, comprising: (a) providing a first lacrimal implant comprising a sustained release formulation of a therapeutic agent for treating OAG or OH; (b) providing a second lacrimal implant that does not comprise the therapeutic agent; and (c) inserting the first and second lacrimal implant through an upper and lower punctum into a lacrimal canaliculus of the same eye wherein the therapeutic agent is released in a therapeutically effective dose from the first lacrimal implant on a sustained basis over at least eight (8) weeks.
[0283] E15. A method of treating a patient with Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye, comprising: (a) providing a first lacrimal implant comprising a sustained release formulation of a therapeutic agent for treating OAG or OH; (b) providing a second lacrimal implant that does not comprise the therapeutic agent; (c) inserting the first and second lacrimal implant through an upper and lower punctum into a lacrimal canaliculus of the same eye; and (d) releasing the therapeutic agent from the first lacrimal implant as a therapeutically effective dose on a sustained basis over at least twelve (12) weeks.
[0284] E16. A method of treating a patient with Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye by reducing intraocular pressure (IOP) in the eye, comprising: (a) measuring the IOP of the patient to obtain a baseline IOP before treatment; (b) providing a therapeutic agent for treating OAG or OH as a sustained release formulation; (c) delivering the sustained release formulation to the eye using a lacrimal implant comprising the sustained release formulation; and (d) releasing the therapeutic agent to the eye on a sustained basis over at least 8 weeks wherein the IOP is reduced by at least 4 mmHg from baseline at week 8.
[0285] E17. A method of treating a patient with Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye by reducing intraocular pressure (IOP) in the eye, comprising: (a) measuring the IOP of the patient to obtain a baseline IOP before treatment; (b) providing a therapeutic agent for treating OAG or OH as a sustained release formulation; (c) delivering the sustained release formulation to the eye using a lacrimal implant comprising the sustained release formulation; and (d) releasing the therapeutic agent to the eye on a sustained basis over at least 12 weeks wherein the IOP is reduced by at least 4.0 mmHg from baseline at week 12.
[0286] E18. A method of treating a patient with Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye by reducing intraocular pressure (IOP) in the eye, comprising: (a) measuring the IOP of the patient to obtain a baseline IOP before treatment; (b) providing a therapeutic agent for treating OAG or OH as a sustained release formulation; (c) delivering the sustained release formulation to the eye using a lacrimal implant comprising the sustained release formulation; and (d) releasing the therapeutic agent to the eye on a sustained basis over at least 12 weeks wherein the IOP is reduced by at least 5.0 mmHg from baseline at week 12.
[0287] E19. A lacrimal implant comprising a unit dosage of about 95 .mu.g of latanoprost for use in the treatment of Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye, wherein the latanoprost is administered from the lacrimal implant comprising a sustained release formulation of the 95 ug of latanoprost and the latanoprost is released in a therapeutically effective dose from the lacrimal implant over a treatment period, with the proviso that the lacrimal implant is inserted into an upper punctum of the eye and a lower punctum of the eye is open or has inserted a blank lacrimal implant that does not comprise latanoprost.
[0288] E20. A lacrimal implant comprising a unit dosage of about 95 .mu.g of latanoprost for use in the treatment of Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye, wherein the latanoprost is administered from the lacrimal implant comprising a sustained release formulation of the 95 .mu.g of latanoprost and the latanoprost is released in a therapeutically effective dose from the lacrimal implant over the treatment period, with the proviso that the lacrimal implant is inserted into a lower punctum and a blank lacrimal implant that does not comprise latanoprost is inserted in an upper punctum.
[0289] E21. A lacrimal implant comprising a unit dosage of about 95 .mu.g of latanoprost for use in the treatment of Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye by reducing intraocular pressure (IOP) in the eye, wherein the latanoprost is administered from a lacrimal implant comprising a sustained release formulation of the 95 .mu.g of latanoprost and the latanoprost is released in a therapeutically effective dose from the lacrimal implant over a treatment period of at least 4 weeks, wherein the IOP is reduced by at least 4 mm Hg from a baseline at week 4.
[0290] E22. A lacrimal implant comprising a unit dosage of about 95 .mu.g of latanoprost for use in the treatment of Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye by reducing intraocular pressure (IOP) in the eye, wherein the latanoprost is administered from a lacrimal implant comprising a sustained release formulation of the 95 .mu.g of latanoprost and the latanoprost is released in a therapeutically effective dose from the lacrimal implant over a treatment period of at least 8 weeks, wherein the IOP is reduced by at least 4 mm Hg from a baseline at week 8.
[0291] E23. A lacrimal implant comprising a unit dosage of about 95 .mu.g of latanoprost for use in the treatment of Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye by reducing intraocular pressure (IOP) in the eye, wherein the latanoprost is administered from a lacrimal implant comprising a sustained release formulation of the 95 .mu.g of latanoprost and the latanoprost is released in a therapeutically effective dose from the lacrimal implant over a treatment period of at least 12 weeks, wherein the IOP is reduced by at least 4 mm Hg from a baseline at week 12.
[0292] E24. A lacrimal implant drug delivery system comprising a unit dosage of about between 140 and 200 .mu.g of latanoprost for use in the treatment of Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye by reducing intraocular pressure (IOP) in the eye, comprising: providing the unit dosage of about between 140 and 200 .mu.g of latanoprost to an eye over a treatment period of at least 4 weeks, wherein the latanoprost is administered from a lacrimal implant comprising a sustained release formulation of the latanoprost and the latanoprost is released in a therapeutically effective dose from the lacrimal implant over the treatment period, wherein the IOP is reduced by at least 5 mm Hg from a baseline at week 4.
[0293] E25. A lacrimal implant drug delivery system comprising a unit dosage of about between 140 and 200 .mu.g of latanoprost for use in the treatment of Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye by reducing intraocular pressure (IOP) in the eye, comprising: providing the unit dosage of about between 140 and 200 .mu.g of latanoprost to an eye over a treatment period of at least 8 weeks, wherein the latanoprost is administered from a lacrimal implant comprising a sustained release formulation of the latanoprost and the latanoprost is released in a therapeutically effective dose from the lacrimal implant over the treatment period, wherein the IOP is reduced by at least 5 mm Hg from a baseline at week 8.
[0294] E26. A lacrimal implant drug delivery system comprising a unit dosage of about between 140 and 200 .mu.g of latanoprost for use in the treatment of Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye by reducing intraocular pressure (IOP) in the eye, comprising: providing the unit dosage of about between 140 and 200 .mu.g of latanoprost to an eye over a treatment period of at least 12 weeks, wherein the latanoprost is administered from a lacrimal implant comprising a sustained release formulation of the latanoprost and the latanoprost is released in a therapeutically effective dose from the lacrimal implant over the treatment period, wherein the IOP is reduced by at least 5 mm Hg from a baseline at week 12.
[0295] E27. A lacrimal implant drug delivery system comprising a unit dosage of about 190 .mu.g of latanoprost for use in the treatment of Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye by reducing intraocular pressure (IOP) in the eye, comprising: providing the unit dosage of about 190 .mu.g of latanoprost to an eye over a treatment period of at least 4 weeks, wherein the latanoprost is administered from a lacrimal implant comprising a sustained release formulation of the latanoprost and the latanoprost is released in a therapeutically effective dose from the lacrimal implant over the treatment period, wherein the IOP is reduced by at least 5 mm Hg from a baseline at week 4.
[0296] E28. A lacrimal implant drug delivery system comprising a unit dosage of about 190 .mu.g of latanoprost for use in the treatment of Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye by reducing intraocular pressure (IOP) in the eye, comprising: providing the unit dosage of about 190 .mu.g of latanoprost to an eye over a treatment period of at least 8 weeks, wherein the latanoprost is administered from a lacrimal implant comprising a sustained release formulation of the latanoprost and the latanoprost is released in a therapeutically effective dose from the lacrimal implant over the treatment period, wherein the IOP is reduced by at least 5 mm Hg from a baseline at week 8.
[0297] E29. A lacrimal implant drug delivery system comprising a unit dosage of about 190 .mu.g of latanoprost for use in the treatment of Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye by reducing intraocular pressure (IOP) in the eye, comprising: providing the unit dosage of about 190 .mu.g of latanoprost to an eye over a treatment period of at least 12 weeks, wherein the latanoprost is administered from a lacrimal implant comprising a sustained release formulation of the latanoprost and the latanoprost is released in a therapeutically effective dose from the lacrimal implant over the treatment period, wherein the IOP is reduced by at least 4 mm Hg from a baseline at week 12.
[0298] E30. A lacrimal implant drug delivery system comprising a unit dosage of about 141 .mu.g of latanoprost for use in the treatment of Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye by reducing intraocular pressure (IOP) in the eye, comprising: providing the unit dosage of about 141 .mu.g of latanoprost to an eye over a treatment period of at least 8 weeks, wherein the latanoprost is administered from a lacrimal implant comprising a sustained release formulation of the latanoprost and the latanoprost is released in a therapeutically effective dose from the lacrimal implant over the treatment period, wherein the IOP is reduced by at least 5 mm Hg from a baseline at week 8.
[0299] E31. A lacrimal implant drug delivery system comprising a sustained release formulation of a therapeutic agent for use in the treatment of Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye, comprising: (a) providing a first lacrimal implant comprising the sustained release formulation of a therapeutic agent for treating OAG or OH; (b) providing a second lacrimal implant that does not comprise the therapeutic agent; and (c) inserting the first and second lacrimal implant through an upper and lower punctum into a lacrimal canaliculus of the same eye wherein the therapeutic agent is released in a therapeutically effective dose from the first lacrimal implant on a sustained basis over at least four (4) weeks.
[0300] E32. A lacrimal implant drug delivery system comprising a sustained release formulation of a therapeutic agent for use in the treatment of Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye, comprising: (a) providing a first lacrimal implant comprising the sustained release formulation of a therapeutic agent for treating OAG or OH; (b) providing a second lacrimal implant that does not comprise the therapeutic agent; and (c) inserting the first and second lacrimal implant through an upper and lower punctum into a lacrimal canaliculus of the same eye wherein the therapeutic agent is released in a therapeutically effective dose from the first lacrimal implant on a sustained basis over at least eight (8) weeks.
[0301] E33. A lacrimal implant drug delivery system comprising a sustained release formulation of a therapeutic agent for use in the treatment of Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye, comprising: (a) providing a first lacrimal implant comprising the sustained release formulation of a therapeutic agent for treating OAG or OH; (b) providing a second lacrimal implant that does not comprise the therapeutic agent; (c) inserting the first and second lacrimal implant through an upper and lower punctum into a lacrimal canaliculus of the same eye; and (d) releasing the therapeutic agent from the first lacrimal implant as a therapeutically effective dose on a sustained basis over at least twelve (12) weeks.
[0302] E34. A lacrimal implant drug delivery system comprising a sustained release formulation of a therapeutic agent for use in the treatment of Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye by reducing intraocular pressure (IOP) in the eye, comprising: (a) measuring the IOP of the patient to obtain a baseline IOP before treatment; (b) providing the therapeutic agent for treating OAG or OH as a sustained release formulation; (c) delivering the sustained release formulation to the eye using the lacrimal implant comprising the sustained release formulation; and (d) releasing the therapeutic agent to the eye on a sustained basis over at least 8 weeks wherein the IOP is reduced by at least 4 mmHg from baseline at week 8.
[0303] E35. A lacrimal implant drug delivery system comprising a sustained release formulation of a therapeutic agent for use in the treatment of Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye by reducing intraocular pressure (IOP) in the eye, comprising: (a) measuring the IOP of the patient to obtain a baseline IOP before treatment; (b) providing the therapeutic agent for treating OAG or OH as a sustained release formulation; (c) delivering the sustained release formulation to the eye using a lacrimal implant comprising the sustained release formulation; and (d) releasing the therapeutic agent to the eye on a sustained basis over at least 12 weeks wherein the IOP is reduced by at least 4.0 mmHg from baseline at week 12.
[0304] E36. A lacrimal implant drug delivery system comprising a sustained release formulation of a therapeutic agent for use in the treatment of Open Angle Glaucoma (OAG) or Ocular Hypertension (OH) in an eye by reducing intraocular pressure (IOP) in the eye, comprising: (a) measuring the IOP of the patient to obtain a baseline IOP before treatment; (b) providing the therapeutic agent for treating OAG or OH as a sustained release formulation; (c) delivering the sustained release formulation to the eye using a lacrimal implant comprising the sustained release formulation; and (d) releasing the therapeutic agent to the eye on a sustained basis over at least 12 weeks wherein the IOP is reduced by at least 5.0 mmHg from baseline at week 12.
[0305] The following Examples are provided to illustrate exemplary embodiments of the invention and are not to be construed as limiting the scope of the present invention.
EXAMPLES
Example 1--Evaluation of Safety and Efficacy of the Latanoprost Punctal Plug Delivery System (L-PPDS) Containing Latanoprost
[0306] A Phase II, open-label, clinical study was conducted in human subjects with ocular hypertension (OH) or open-angle glaucoma (OAG) to evaluate safety and efficacy of the latanoprost punctal plug delivery system (L-PPDS).
[0307] The Phase II trial featured simultaneous placement of punctal plugs in both the upper and lower puncta for delivery of a daily drug load with a goal of enabling comparable clinical outcomes to that of daily administered Xalatan.RTM. eye drops. The overall objective was a mean reduction in IOP of 5 mm Hg or greater. The primary endpoint in this Phase II study was a mean change in IOP from baseline (measured as mm Hg) at 2 weeks. Secondary endpoints were the mean change in IOP from baseline at 4 weeks and mean percentage change in IOP at 2 weeks and 4 weeks. A total of 95 ITT (Intent to Treat) subjects were included in the L-PPDS treatments in this study. The mean IOP as baseline was 25.8 mm HG for this group (with a range of baseline of 22.5 mm Hg to 33 mm Hg).
[0308] After 2 weeks of L-PPDS treatment, IOP showed a statistically significant mean change from baseline of -6.2 mm Hg (95% C.I. -6.8, -5.6). At the end of week 2, 73% of subjects showed an IOP reduction vs. baseline of 5 mm Hg or greater and 51% of subjects showed a reduction of 6 mm Hg or greater. The mean percentage change in IOP from baseline at 2 weeks was -24.3%, which was statistically significant (95% C.I. -26.7, -21.9).
[0309] After 4 weeks of L-PPDS treatment, IOP showed a statistically significant mean change from baseline of -5.7 mm Hg (95% C.I. -6.5, -4.9). At the end of 4 weeks, 60% of subjects showed an IOP reduction vs. baseline of 5 mm Hg or greater and 47% of subjects showed a reduction of 6 mm or greater. The mean percentage change in IOP from baseline at 4 weeks was also statistically significant at 22.3% (95% C.I. -25.4, -19.2).
[0310] Subjects were fitted with L-PPDS containing specified latanoprost concentrations in the upper L-PPDS (46 .mu.g) and the lower L-PPDS (95 .mu.g).
Study Procedures
[0311] During the screening visit, the subjects were fitted with the trial punctal plugs for approximately 15 minutes to 2 hours to assess fitting and eligibility. Pre-washout IOP measurements were determined in each eye of the patient, and adverse device events (ADEs) were also monitored. After the trial fitting, subjects began the washout period in which subjects were discontinued from topical prostaglandin therapy to assess IOP eligibility for a minimum of 4 weeks and to a maximum of 6 weeks.
[0312] After the washout, baseline IOP was measured in each eye of a patient on two separate visits that were 2 to 4 days apart (Day -2 and Day 0 study visits), after the patient had washed out of previous topical prostaglandin therapy.
[0313] At the start of the study (Day 0), each patient had an L-PPDS inserted bilaterally into each puncta of each eyes and inspected thereafter at each visit. If an L-PPDS was spontaneously extruded, one replacement L-PPDS per patient was allowed. The L-PPDS were removed at the Week 4 visit.
[0314] After placement of the L-PPDS, subjects were monitored for any treatment-emergent or adverse events (Aes) during the 4-week treatment period. Subjects had weekly follow-up visits with the last study visit at Week 4. Tear volume was measured by a Schirmer test with anesthesia over 5 minutes at Day 0 and at the last visit. Visual acuity was measured with best correction using a Snellen chart at every visit. Biomicroscopy examinations were performed in each eye at every visit, including an inspection of the L-PPDS placement. Treatment-emergent ocular and systemic Aes and concomitant medications were monitored at every visit with standardized questioning techniques. Automated perimetry was performed to measure visual fields at the last visit. A funduscopy examination was performed at the last visit.
[0315] Goldmann IOP measurements (the average of 3 measurements) were measured in each eye at every visit. The baseline IOP was taken on two separate days, at least 48 hours apart. Specifically, IOP measurements were taken at 8:30 am (.+-.30 minutes) at each visit.
L-PPDS
[0316] Each L-PPDS for the upper puncta was of a proprietary punctal plug design and had a latanoprost strength of 46 .mu.g. Inactive components were medical grade silicone, polyimide tubing, DMPC, cyanoacrylate medical grade adhesive, and 2% green colorant. Each L-PPDS was supplied in a separate sterilized mylar foil pouch.
[0317] Each L-PPDS for the lower puncta was of a proprietary punctal plug design and had a latanoprost strength of 95 .mu.g. Inactive components are medical grade silicone, polyimide tubing, DMPC, cyanoacrylate medical grade adhesive, and 2% green colorant. L-PPDS for the lower puncta were preloaded on an insertion tool. Each L-PPDS and insertion tool were supplied in a tray contained in a sterilized mylar foil pouch.
[0318] FIG. 13 shows the mean reduction in intraocular pressure (IOP) from baseline in weeks during the treatment period. FIG. 14 shows the percent of subjects that recorded an IOP reduction of .gtoreq.5 mm Hg from baseline, .gtoreq.6 mm Hg from baseline, and .gtoreq.7 mm Hg from baseline, in weeks.
[0319] A secondary endpoint in the study was the percentage change in average IOP from baseline at 2 weeks and 4 weeks for L-PPDS treatment. FIG. 15 shows the mean reduction in IOP from baseline in weeks during the treatment period. At 2 weeks, the percentage change from baseline of -24.3% (95% C.I. -26.7, -21.9) was statistically significant, and at 4 weeks, the percentage change from baseline of -22.3% (95% C.I. -25.4, -19.2) was also statistically significant.
[0320] The IOP reduction results are summarized in the Table 4:
TABLE-US-00004 TABLE 4 Mean change and % change in IOP in weeks for L-PPDS containing latanoprost concentration of 141 .mu.g Mean Change in IOP from % Change in IOP Baseline (95% C.I.) from Baseline (95% C.I.) 2 weeks -6.2 (-6.8, -5.6) -24.3% (-26.7%, -21.9%) (n = 70) 4 weeks -5.7 (-6.5, -4.9) -22.3% (-25.4%, -19.2%) (n = 53)
Plug Retention Results
[0321] The lower punctal plug achieved 94% or greater retention per subject across the duration of the 4 week study. The upper punctal plug showed a retention rate of 40% per subject across the duration of the study.
Assessment of Treatment Safety and Tolerability
[0322] The L-PPDS was well tolerated over the testing period. The majority of AEs were ocular in nature, however, none were serious AEs. The most frequently reported AE was mild to moderate tearing. Few subjects experienced any discomfort related to the punctal plugs with most subjects having either no awareness or mild awareness of the punctal plugs by week 4.
Example 2--Comparison of Clinical Studies of the L-PPDS at Different Dosages
[0323] A number of clinical studies have been conducted to assess the safety, efficacy, and dosing for L-PPDS treatment in more than 300 human subjects with OH or OAG. These studies have investigated the preliminary safety and efficacy of L-PPDS over the dose range of 3.5 to 95 .mu.g per eye, primarily delivered via an L-PPDS positioned in the lower puncta. See, for example, ClinicalTrials.gov Identifier: NCT00967811, "An Open-Label, Phase 2 Study of Different Formulations (E1 and E2) of the Latanoprost Punctal Plug Delivery System (L-PPDS) in Subjects With Ocular Hypertension (OH) or Open-Angle Glaucoma (OAG)," Study Start Date: August 2009, (other study ID number: PPL GLAU 07), incorporated herein by reference; and ClinicalTrials.gov Identifier: NCT01037036, "An Open-Label, Phase 2 Study of the Latanoprost Punctal Plug Delivery System (L-PPDS) With Adjunctive Xalatan.RTM. Eye Drops in Subjects With Ocular Hypertension (OH) or Open-Angle Glaucoma (OAG)," Study Start Date: Dec. 17, 2009, (other study ID number: PPL GLAU 08), incorporated herein by reference. Based on the published clinical results for IOP reduction associated with Xalatan.RTM. eye drops, the magnitude of mean IOP reductions has been less than expected; however, results for L-PPDS show that some subjects had IOP reductions that would be expected with Xalatan.RTM.. All studies have occluded only one punctum per eye. The overall mean IOP reduction for the L-PPDS was similar among most of the studies (range -3 to -5 mmHg), regardless of latanoprost concentration from 3.5 .mu.g to 95 .mu.g per eye. Specifically, the mean IOP reduction according to the clinical study described in Example 1 and the clinical studies of the L-PPDS containing latanoprost concentrations of 44 .mu.g and 81 .mu.g, see ClinicalTrials.gov Identifier: NCT00820300, "An Open-Label, Phase 2 Study of the Latanoprost Punctal Plug Delivery System (L-PPDS) in Subjects With Ocular Hypertension (OH) or Open Angle Glaucoma (OAG)," Study Start Date: January 2009, (other study ID number: PPL GLAU 03), incorporated herein by reference, and two different 95 .mu.g formulations were compared, where the two 95 .mu.g formulations (E1 and E2) were developed to deliver different average daily doses, see ClinicalTrials.gov Identifier: NCT00967811, "An Open-Label, Phase 2 Study of Different Formulations (E1 and E2) of the Latanoprost Punctal Plug Delivery System (L-PPDS) in Subjects With Ocular Hypertension (OH) or Open-Angle Glaucoma (OAG)," Study Start Date: August 2009, (other study ID number: PPL GLAU 07), incorporated herein by reference. Table 5 summarizes the range of mean IOP change from baseline during the first 4 weeks period of the L-PPDS treatments in the clinical studies. Table 6 summarizes the IOP change (mm Hg) in number (percent) of subjects in weeks 2 and 4.
TABLE-US-00005 TABLE 5 Mean IOP change from baseline in clinical studies Study ID GLAU 11 GLAU 03 GLAU 07 (Example 1) L-PPDS 44 .mu.g 81 .mu.g 95 .mu.g E1 95 .mu.g E2 141 .mu.g Formulation (N = 57) (N = 53) (N = 42) (N = 41) (N = 95) Range of Mean -3.5 to -3.6 -3.0 to -3.4 -3.5 to -4.2 -3.9 to -4.7 -5.7 to -6.8 IOP Change from Baseline: Weeks 1 to 4 (mmHg)
TABLE-US-00006 TABLE 6 IOP Changes in % Subjects Study ID GLAU 03 GLAU 07 GLAU 11 L-PPDS 44 .mu.g 81 .mu.g 95 .mu.g E1 95 .mu.g E2 141 .mu.g Formulation (N = 57) (N = 53) (N = 42) (N = 41) (N = 95) Week 2 .gtoreq.5 31% 25% 31% 22% 73% .gtoreq.6 24% 16% 17% 17% 51% .gtoreq.7 16% 4% 12% 15% 36% Week 4 .gtoreq.5 35% 29% 38% 31% 60% .gtoreq.6 23% 22% 15% 21% 47% .gtoreq.7 8% 12% 13% 10% 28%
[0324] No mean IOP change of .gtoreq.5 mm Hg was observed within the 4 weeks duration of treatments of L-PPDS containing latanoprost concentrations of 44 .mu.g, 81 .mu.g, and 95 .mu.g. The mean IOP reductions were significantly greater with L-PPDS containing a combined latanoprost concentration of 141 .mu.g recorded in the clinical study described in Example 1, compared with L-PPDS containing lower lantanoprost doses. The mean IOP change from baseline for L-PPDS with a combined latanoprost concentration of 141 .mu.g was substantial, from -5.7 mm Hg to -6.8 mm Hg.
Example 3--Method of Preparation L-PPDS (95 .mu.g) Cores
[0325] NuSil Silicone MED6385 part A was stirred for a minimum of 5 minutes, and 63 mg of which was weighed and transferred onto a glass slide. To the same glass slide was added Latanoprost (obtained from Everlight Chemical, Taipei, Taiwan) (48 mg), dimyristoylphosphatidylcholine (DMPC) (9 mg) and NuSil Med6382 crosslinker (2.4 mL). Using a 0.5 .mu.L Hamilton Syringe, Nusil MED6385 part B (0.348 .mu.L) was transferred directly onto a mini spatula. The latanoprost, NuSil MED6382 crosslinker, NuSil Silicone MED-6385 part B and DMPC were mixed together for 2-5 minutes to form a homogenous mixture. The resulting mixture was combined with NuSil Silicone MED6385 part A and was mixed for another 2 minutes to form a homogenous mixture, which was immediately transferred into a previously prepared syringe assembly. The syringe was then attached to polyimide tubing (0.024'' OD) by way of an adapter. The polyimide tubing was kept at a temperature of 4.degree. C. by way of a cooling jacket. After 2 minutes, the mixture was injected into the polyimide tubing by increasing the pressure of the system to 40 psi over 2.5 minutes. Once the mixture had extruded through the polyimide tubing to the end, the tubing was cut and both ends were clamped. The tube was placed into a humidity chamber at 40.degree. C. and 80% relative humidity for 24-96 hours to cure the silicone, and the tubing was cut into 1.0 mm lengths. Loctite 4305 (Henkel Adhesives Technologies, Ltd.) was applied to the bottom end of the cut tubing and cured for 20 seconds in a 100 W UV curing chamber. These cores were then inserted into the cavity of punctal plugs with the glued end positioned in the bottom of the cavity.
Example 4--PP DEV 05: A Device Evaluation Study to Further Assess the Physical and Clinical Characteristics of Prototype Punctal Plug Design Iterations
Study Objective
[0326] To evaluate the physical and clinical performance characteristics of punctal plug design iterations.
Study Design
[0327] This is a multicenter, device assessment, feasibility study to assess the physical and clinical performance characteristics of prototype punctal plugs. Up to approximately 500 subjects will be enrolled at 5-15 sites in the US. No drug treatment will be administered. The study will evaluate the physical (handling) and clinical (comfort, tearing, retention) characteristics of punctal plug prototypes. The study will be iterative, with data monitored on an ongoing basis and design modifications to the punctal plugs made if further improvements are indicated. An investigational plug detection aid may also be evaluated.
[0328] Subjects will be enrolled into 1 of 2 groups. Group 1 will undergo two 12-week plug placement periods. Group 2 will undergo two 2-week plug placement periods followed by one 12-week plug placement period. Plug placement will be attempted in the lower and upper puncta of both eyes. Placement must be successful in both the upper and lower puncta of at least 1 eye for the subject to be eligible for the study. The Sponsor will inform the sites in advance in writing which plug iterations to use for each subject for each placement period. For Group 1, study visits will occur at Day 0, and Weeks 4, 8, 12, 16, 20 and 24, with plug placement at the Day 0 and Week 12 visits. For Group 2, study visits will occur at Day 0 and Weeks 1, 2, 3, 4, 8, 12 and 16, with plug placement at Day 0, Week 2, and Week 4.
[0329] A subject who completes or is withdrawn from the Group 1 or Group 2 treatment schedule may be re-enrolled into the study (to either Group 1 or Group 2). A re-enrolled subject will be assigned a new subject number and undergo screening procedures again.
[0330] Safety will be assessed throughout the study.
Study Population
[0331] Subjects will be male and female volunteers, age 50 years or older. Main exclusion criteria will include: [0332] History of, or active, lid disease requiring lid scrubs (e.g., moderate or severe blepharitis, dacryocystitis, meibomianitis) [0333] Structural lid abnormalities (e.g., ectropion, entropion) [0334] Active anterior segment inflammatory disease [0335] Ocular allergies [0336] Habitual eye rubbing [0337] Previous intolerance of punctal plugs (e.g., inflammatory reaction, granuloma, dacryocystitis, etc. due to punctal plug wear) [0338] Laser eye surgery within the last 3 months or incisional eye surgery within the last 6 months.
Study Devices
[0339] The punctal plugs will be placed bilaterally into the upper and lower puncta using an investigational insertion tool provided with the plug or ophthalmic forceps. The techniques for insertion and removal are similar to the procedures for other commercial punctal plugs.
Study Variables
[0340] Device Performance: [0341] Retention rates [0342] Insertion success [0343] Ease of use
[0344] Tolerability [0345] Comfort [0346] Tearing
Safety
[0346] [0347] Adverse device events (ADEs) [0348] Biomicroscopy
Study Procedures and Assessments:
[0349] Device Performance: [0350] For subjects who provide additional consent, photographs of the punctal plugs may be taken after their placement to observe their location in the lid margin; videography of punctal plug placement and removal procedures may be performed for future physician training. In-person observational physician training of punctal plug placement and removal procedures may also occur.
[0351] Tolerability: [0352] Subjects will rate the acceptability of tearing and comfort according to a visual analog scale at every visit.
[0353] Safety: [0354] Biomicroscopy will be performed in each eye and ADEs will be collected at every study visit.
Sample Size and Statistical Analyses:
[0354] [0355] The sample size is based on clinical judgment and is believed to be sufficient to meet the study objectives. All study variables will be summarized descriptively. ADEs will be coded using the Medical Dictionary for Regulatory Activities (MedDRA) and summarized descriptively by system organ class and preferred term.
Example 5: GLAU 12: A Phase 2 Dose Evaluation Study for the Latanoprost Punctal Plug Delivery System (L-PPDS) in Subjects With Ocular Hypertension (OH) or Open-Angle Glaucoma (OAG)
[0356] The Phase II trial featured simultaneous placement of punctal plugs in both the upper and lower puncta for delivery of a daily drug load with a goal of enabling comparable clinical outcomes to that of daily administered Xalatan.RTM. eyedrops. The objective of this study was to evaluate the efficacy, safety and duration of effect of the L-PPDS at two dose levels (141 .mu.g and 190 .mu.g).
[0357] Study PPL GLAU 11 (Example 1 and FIGS. 13-15) showed that occlusion of both puncta with the L-PPDS at a total latanoprost dose per eye of 141 .mu.g significantly reduced IOP for up to 4 weeks. This study, in which the left eye has the same dose as PPL GLAU 11 and the right eye has a 95 .mu.g L-PPDS in both the upper and lower puncta, is designed to replicate the results of PPL GLAU 11 and to assess the effect of a higher dose. In addition, this study will run for 12 weeks to determine whether the effect observed in PPL GLAU 11 is durable for a longer time period.
[0358] The latanoprost dose for the left eye (46 .mu.g L-PPDS in the upper punctum and 95 .mu.g L-PPDS in the lower punctum) was chosen in order to replicate the dose used in Study PPL GLAU 11. The latanoprost dose for the right eye (95 .mu.g L-PPDS in both the upper and lower puncta) was chosen because it is the highest dose currently attainable.
[0359] The highest latanoprost dose in this study (190 .mu.g delivered over 3 months) is equivalent to the amount in approximately 127 drops of Xalatan. The prescribed Xalatan dose over 3 months is 90 drops (1 drop/day), so 190 .mu.g latanoprost delivered over 3 months represents a dose about one-third higher than that for Xalatan drops.
Main Study Design
[0360] Subjects diagnosed with bilateral OH or OAG who are treatment naive or managed with up to 2 glaucoma medications were eligible for study screening. IOP eligibility was established at baseline prior to enrollment in the study.
[0361] After eligibility was established, subjects received treatment for 12 weeks as follows: [0362] Right eye: 95 .mu.g L-PPDS inserted in the lower puncta and 95 .mu.g L-PPDS in the upper puncta. [0363] Left eye: 95 .mu.g L-PPDS inserted in the lower puncta and a 46 .mu.g L-PPDS in the upper puncta.
Addendum Study Design
[0364] The addendum study included subjects enrolled in the main study who had a decrease in IOP of >5 mmHg from baseline at Day 7 in response to two 95 .mu.g L-PPDS and who retained both plugs in at least 1 eye for at least 4 weeks in the first treatment cycle. Eligible subjects started L-PPDS Cycle 2 (C2) within 30 days of removal of L-PPDS in the main study. On C2 Day 0, subjects had 95 .mu.g L-PPDS inserted in the upper and lower puncta of both eyes for 8 weeks (C2). At the end of C2, another set of 95 .mu.g L-PPDS were inserted in the upper and lower puncta of both eyes for 4 weeks (Cycle 3 [C3]). Subjects were followed up for assessment of safety and IOP effect with visits at 1, 2, 4, 6 and 8 weeks of C2, and 1, 2 and 4 weeks of C3. Safety was monitored as in the main study. Analysis of IOP effect was primarily based on change from baseline in IOP measurements (the baseline value from the main study was used to determine IOP change from baseline in subsequent cycles) and between-cycle comparisons.
[0365] Treatment
[0366] Main Study
[0367] Investigational L-PPDS for the lower puncta of both eyes was the L67 design with a latanoprost dose of 95 .mu.g. See, FIG. 33. Investigational L-PPDS for the upper puncta was the L69 design with a latanoprost dose of 95 .mu.g for the right eye, and the L72 design with a latanoprost dose of 46 .mu.g for the left eye.
[0368] The total latanoprost dose was 190 .mu.g for the right eye and 141 .mu.g for the left eye.
[0369] At the Day 0 visit, subjects had the plugs inserted bilaterally into the upper and lower puncta. If any plug spontaneously extruded, it was to be replaced with an L67 95-.mu.g L-PPDS, if possible. The number of replacements was limited to 2, and once a subject lost plugs from both eyes, the subject was withdrawn from the study. The LPPDS was removed at the Week 12 visit.
Addendum Study
[0370] Investigational L-PPDS for all puncta had a latanoprost dose of 95 .mu.g. The total latanoprost dose was 190 .mu.g/eye. Subjects underwent an 8-week L-PPDS treatment cycle (C2) followed by a 4-week L-PPDS treatment cycle (C3). If any plug spontaneously extruded, it was to be replaced; however, once a subject lost a plug from each eye in C2, the remaining L-PPDS were removed, and the subject started C3. Once a subject lost a plug from each eye in C3 the subject was withdrawn from the study.
[0371] Subjects were followed up for assessment of safety and IOP effect. Safety was monitored with adverse events (AEs), IOP, Snellen best-corrected visual acuity (BCVA) or pinhole visual acuity (method should be consistent for a given subject throughout the study), biomicroscopy, subject tearing and comfort assessments, automated perimetry, and funduscopy. Analysis of IOP effect was primarily based on change from baseline in IOP measurements.
[0372] To address the study objective of evaluating the safety and IOP lowering effects of the L-PPDS, IOP results were compared to baseline IOP. The IOP entry criteria included precautions to ensure that washout (if applicable) is complete and baseline IOP is stable (e.g., minimum 5 mmHg change from pre-screening and less than 3 mmHg difference in IOP between 2 baseline visits 2 days apart).
[0373] Providing different treatments to each of a subject's eyes necessitated that the eyes be independent of each other for the results to be valid.
Number of Subjects and Statistical Analyses
[0374] Approximately 55 subjects were enrolled to have 35 eyes available for each treatment for the evaluable analysis. With a sample size of 35 evaluable eyes in each treatment group, a 2-sided 95.0% confidence interval (CI) for the mean IOP change from baseline extended 1.0 mm Hg from the observed mean, assuming that the standard deviation was known to be 3.0 mm Hg and the CI is based on the large sample z statistic. The standard deviation of 3.0 mm Hg used in the above sample size calculation was based on results of the L-PPDS clinical studies conducted to date.
[0375] The primary efficacy variable was the change from baseline in IOP measurements and the primary analysis time point will be at 4 weeks. Other IOP variables listed above were secondary efficacy variables. For analyses using the intent-to-treat (ITT) data set, all data from all subjects with at least 1 follow-up IOP measurement were included. For analyses using the evaluable (EVAL) data set, data from subjects or visits with significant protocol deviations were excluded.
Inclusion Criteria
[0376] To be eligible for the study, subjects must fulfill all of the following criteria:
[0377] Subjects who are men or women >18 years old.
[0378] Subjects diagnosed with bilateral OAG or OH. Subjects may be treatment naive or managed with up to 2 medications (combination products such as Cosopt.RTM. will be considered 2 medications). [0379] For subjects on a topical prostaglandin treatment (as monotherapy or in combination): Screening IOP is .ltoreq.21.0 mmHg. [0380] Subjects who can be fitted with L-PPDS in all 4 puncta at Day 0. [0381] Subjects whose baseline IOP measured at 2 baseline visits (i.e., average of IOP values obtained at 2 baseline visits) meets the following criteria in each eye after the screening period: [0382] a. .gtoreq.22.0 mmHg [0383] b. .ltoreq.34.0 mmHg [0384] For subjects on topical prostaglandin therapy at screening: Is increased .gtoreq.5.0 mmHg from screening. [0385] Subjects whose baseline IOP measurements in each eye are .ltoreq.3 mmHg apart between 2 sequential baseline visits. [0386] Subjects who have central corneal thickness in each eye .gtoreq.500 .mu.m and .gtoreq.600 .mu.m. [0387] Subjects who have Snellen BCVA 20/100 or better in each eye. [0388] Subjects who are women of child-bearing potential must not be pregnant or lactating, must have a negative pregnancy test at screening and must be practicing an adequate method of birth control. Acceptable methods of birth control include intrauterine device (IUD); oral, dermal ("patch"), implanted or injected contraceptives; tubal ligation; and barrier methods with spermicide. [0389] Subjects who sign an approved informed consent form for the study. [0390] Subjects who are willing to comply with the protocol.
Study Procedures and Assessments
[0391] IOP measurements were determined in each eye at the screening visit. The duration of the screening period depended on the time required for washout of topical ocular hypotensive therapy. Washout was not required for treatment-naive subjects.
[0392] After the screening period, IOP measurements were determined in each eye on 2 separate visits, 2 to 4 days apart. For treatment-naive subjects the initial screening visit was considered as the first visit for determination of baseline IOP.
[0393] The L-PPDS treatment period was 12 weeks. Study visits occurred at Days 1, 3, 7, and 14, and Weeks 3, 4, 6, 8, 10 and 12. (Subjects who were prostaglandin-naive at study entry had a visit at Week 14 after the Xalatan run-out period is complete.) There was a follow-up telephone call 3 days after the last visit. The following tests and procedures were performed during study follow-up. [0394] Goldmann IOP measurements (the average of 3 measurements) will be measured in each eye at every visit. The baseline IOP will be taken on 2 separate days, at least 48 hours apart. IOP measurements must be taken at 8:30 am (.+-.30 minutes) at each visit. [0395] Visual acuity will be measured with best correction or pinhole using a Snellen chart at every visit; the method (best correction or pinhole) should be consistent for a given subject throughout the study. [0396] Biomicroscopy examinations, including an inspection of L-PPDS placement, will be performed in each eye at every visit. [0397] Subjects will rate the acceptability of the tearing and comfort level of the plugs, and the frequency of tearing, on a visual analog scale at each visit starting at Day 0. Subjects will also be asked at the end of the study which they prefer: punctal plugs or eye drops. [0398] Treatment-emergent ocular and systemic AEs and concomitant medications will be monitored at every visit with standardized questioning techniques. [0399] Investigators will rate the ease of insertion of the plugs. [0400] Automated perimetry will be performed to measure visual fields at screening and Week 12. [0401] A funduscopy examination will be performed at screening and Week 12.
[0402] All ocular procedures were performed by an experienced and appropriately qualified individual(s). Subjects who discontinue the study treatment prematurely underwent the tests and procedures for the last visit.
[0403] It is unexpected for the L-PPDS to malfunction and release all or a major portion of its contents in a short period of time. The expected exposure to latanoprost (190 .mu.g delivered over 3 months) is within the safety profile established in a series of ocular toxicity studies conducted to support the FDA approval of Xalatan. Specifically, no adverse effects were observed in rabbits with twice-daily ocular instillation of latanoprost doses up to 50 .mu.g per eye for 52 weeks (100 .mu.g/eye/day). In a similar 52-week ocular study in cynomolgus monkeys, the only effects observed at doses up to 50 .mu.g/eye given twice daily (100 .mu.g/eye/day) were a reversible change in the aspect of the palpebral fissure and a non-reversible increase in iris pigmentation, which were not judged to be deleterious (Xalatan Product Monograph 2011). In a study of 28 healthy volunteers, in which 1 drop of latanoprost 50 .mu.g/mL was administered once daily in 1 eye and 4 times daily in the other eye for 2 weeks, transient photophobia, cells, and mild flare were common during the 4-dose regimen, but these effects resolved spontaneously without cessation of treatment (Linden and Alm 2001).
[0404] Results: Interim analysis (n=83) showed sustained mean IOP decreases from baseline at Week 8 greater than 5 mmHg for the 190 .mu.g dose, with somewhat lower levels for the 141 .mu.g dose.
[0405] Final Study Results: A total of 57 subjects enrolled in the study. Men and women were represented approximately evenly (51% and 49%, respectively). Subjects were Caucasian (37%), Hispanic (32%), Black (26%), and Asian (5%), with an average age of 65 years. Most eyes were assessed to have primary open angle glaucoma (74% right eyes, 69% left eyes), while the other eyes had ocular hypertension (26% right eyes, 31% left eyes). Mean IOP at screening was 18.79 mmHg for right eyes and 18.97 mmHg for left eyes; by baseline (after the washout period) mean IOP had increased to 24.75 mmHg for right eyes and 24.66 mmHg for left eyes.
[0406] At Week 4, 47 subjects (82%) had IOP measured; at Week 12, 35 subjects (61%) had IOP measured. A total of 44 subjects (77%) completed the main study (Week 12). Twelve subjects (21%) participated in the Xalatan run-out. Nineteen subjects (100%) entered the addendum study, and 12 (62%) completed C2 (Week 8) and had IOP measured at that visit. Six subjects (100%) began C3, and 5 subjects (83%) completed C3 (Week 4) had IOP measured at that visit.
[0407] The ITT data set included 109 eyes, EVAL included 101 eyes, and safety included all 57 subjects. Five subjects received 190 .mu.g latanoprost in both eyes (95 .mu.g L-PPDS in upper and lower puncta of both eyes) because the 46 .mu.g L-PPDS was not available when they started treatment. Consequently, although the ITT data set included 57 subjects (114 eyes), the average IOP from the eyes of the 5 subjects who received 190 .mu.g latanoprost in both eyes was used for the analysis, so ITT results are based on 109 data points (eyes).
[0408] Treatment with L-PPDS resulted in significant mean IOP decreases from baseline across all time points (Table 7 and FIG. 23) in the main study.
TABLE-US-00007 TABLE 7 Summary of IOP (mmHg) Results ITT (N = 109 eyes) EVAL (N = 101 eyes) Visit (Main Study) Observed IOP Excl.sup.b Observed IOP Excl.sup.b Day 14 n 99 83 91 76 Mean IOP 18.9 18.8 19.0 18.8 Mean IOP .dwnarw. -5.8 -6.0 -5.8 -5.9 CI (-6.40, -5.26) (-6.58, -5.42) (-6.37, -5.23) (-6.51, -5.36) Week 4 n 89 73 81 67 Mean IOP 19.3 19.3 19.2 19.2 Mean IOP .dwnarw. -5.6 -5.6 -5.7 -5.7 CI (-6.12, -5.05) (-6.13, -5.09) (-6.27, -5.16) (-6.21, -5.16) Week 8 n 70 54 62 48 Mean IOP 20.6 20.3 20.5 20.2 Mean IOP .dwnarw. -4.3 -4.7 -4.3 -4.7 CI (-4.96, -3.64) (-5.29, -4.00) (-5.06, -3.59) (-5.37, -3.95) Week 12 n 67 48 61 44 Mean IOP 20.8 21.1 20.8 21.2 Mean IOP .dwnarw. -4.0 -3.9 -4.1 -3.8 CI (-4.77, -3.28) (-4.77, -3.01) (-4.86, -3.28) (-4.75, -2.89)
[0409] Significant reduction in mean IOP was observed at Weeks 4 and 6 (-5.4 and -5.8 mmHg, respectively, in the ITT group. No meaningful difference in IOP results was observed between eyes (ie, 190 .mu.g and 141 .mu.g latanoprost).
[0410] In the main study, retention rate of L-PPDS in the lower puncta was .gtoreq.96% through Week 12. Retention of upper L-PPDS was 69%, 53%, and 48% at Weeks 4, 8, and 12, respectively. In C2, upper L-PPDS retention was notably higher than in the main study (90% and 88% at Weeks 4 and 8, respectively).
[0411] Study Conclusions: Treatment with L-PPDS in both puncta (total latanoprost dose of either 190 or 141 .mu.g/eye) resulted in a clinically meaningful and statistically significant reduction in IOP from baseline of approximately 6 mmHg after 4 weeks (primary endpoint).
Example 6: GLAU 13: A Randomized Phase 2 Study of the Effect of Plug Placement on Efficacy and Safety of the Latanoprost Punctal Plug Delivery System (L-PPDS) in Subjects With Ocular Hypertension (OH) or Open Angle Glaucoma (OAG)
[0412] Study PPL GLAU 11 (Example 1 and FIGS. 13-15) showed that occlusion of both puncta with the L-PPDS at a total latanoprost dose per eye of 141 .mu.g significantly reduced IOP for up to 4 weeks. This effect could have been due to the dose, double occlusion of the puncta, placement of the L-PPDS in the upper punctum, or a combination thereof. This study (GLAU 13), in which the L-PPDS will be placed in either the upper or lower puncta, with the other punctum either left open or blocked by a punctal plug that does not contain latanoprost, will assess whether the effect observed in Study PPL GLAU 11 was due to delivery of latanoprost from the upper punctum, or was a result of having both puncta blocked, thus increasing the residence time of latanoprost in the tear film and making more drug available to the cornea. In addition, this study will run for 12 weeks to determine the duration of effect.
[0413] Each eye in this study had one 95 .mu.g L-PPDS. This dose was chosen because it is the highest dose available when only one lacrimal implant is utilized. The dose of 95 .mu.g (delivered over 3 months) is equivalent to the amount in approximately 63 drops of Xalatan. The prescribed Xalatan dose over 3 months is 90 drops (1 drop/day), so 95 .mu.g latanoprost delivered over 3 months represents a dose about one-third lower than that for Xalatan drops.
[0414] Subjects diagnosed with bilateral OH or OAG who were treatment naive or managed with up to 2 glaucoma medications were eligible for study screening. During the washout period subjects were discontinued from glaucoma therapy (if applicable). Intraocular pressure (IOP) eligibility was established at baseline prior to enrollment in the study. Successful plug insertion on Day 0 was required for study enrollment.
[0415] After eligibility was established, subjects received treatment for 12 weeks. There were 3 different treatments studied, as follows:
TABLE-US-00008 Upper Punctum Lower Punctum Treatment A 95 .mu.g L-PPDS Punctal plug (no latanoprost) Treatment B 95 .mu.g L-PPDS No plug Treatment C Punctal plug 95 .mu.g L-PPDS (no latanoprost)
[0416] The total dose of Latanoprost per eye was 95 .mu.g.
[0417] Subjects received different treatments in each eye. Because there are 3 different treatments, subjects were randomly assigned to 1 of 3 groups to determine what treatment combination they would receive, as follows:
TABLE-US-00009 Eye Treatment Upper Punctum Lower Punctum Group 1 Right A 95 .mu.g L-PPDS Punctal plug Left B 95 .mu.g L-PPDS No plug Group 2 Right A 95 .mu.g L-PPDS Punctal plug Left C Punctal plug 95 .mu.g L-PPDS Group 3 Right B 95 .mu.g L-PPDS No plug Left C Punctal plug 95 .mu.g L-PPDS
[0418] Subjects were followed up for assessment of safety and IOP effect. Safety was monitored with adverse events (AEs), IOP, Snellen best-corrected visual acuity (BCVA), biomicroscopy, subject tearing and comfort assessments, automated perimetry, and funduscopy. Analysis of IOP effect was primarily based on change from baseline in IOP measurements.
Discussion of Study Design
[0419] To address the study objective of evaluating the safety and IOP lowering effects of the L-PPDS, IOP results were compared to baseline IOP. The IOP entry criteria included precautions to ensure that washout (if applicable) was complete and baseline IOP was stable (e.g., minimum 5 mmHg change from the start of screening and less than 3 mmHg difference in IOP between 2 baseline visits 2 days apart).
[0420] Providing different treatments to each of a subject's eyes necessitates that the eyes be independent of each other for the results to be valid. Studies have shown that the contralateral effect of prostaglandins is minimal, due to their rapid systemic metabolism, and using different treatments on each of a subject's eyes will produce valid and independent results (Ziai et al., The effects on aqueous dynamics of PhXA41, a new prostaglandin F2 .alpha. analogue, after topical application in normal and ocular hypertensive human eyes. Arch Ophthalmol. 111:1351-1358 (1993); Realini et al., The uniocular drug trial and second-eye response to glaucoma medications. Ophthalmol. 111:421-426 (2004)). Contralateral treatment is also a more efficient study design as it requires fewer subjects to be treated. The decision to use contralateral treatment for this study was made in consultation with glaucoma experts.
Study Population
[0421] Approximately 80 subjects were enrolled in the study to have 35 eyes available for the evaluable analysis for each treatment. With a sample size of 35 evaluable eyes for each treatment, a 2-sided 95.0% confidence interval (CI) for the mean IOP change from baseline will extend 1.0 mmHg from the observed mean, assuming that the standard deviation is known to be 3.0 mmHg and the CI is based on the large sample z statistic. The standard deviation of 3.0 mmHg used in the above sample size calculation was based on results of the L-PPDS clinical studies conducted to date.
Main Inclusion Criteria:
[0422] Diagnosed with bilateral OH or OAG and either treatment-naive or currently managed with up to 2 medications. Combination products such as Cosopt.RTM. will be considered 2 medications. [0423] For subjects on a topical prostaglandin treatment either as monotherapy or in combination: Screening IOP is .gtoreq.21.0 mmHg. [0424] Can be fitted with punctal plugs and L-PPDS on Day 0. [0425] Baseline IOP measured at 2 baseline visits meets the following criteria in each eye after the screening period: [0426] c. .gtoreq.22.0 mmHg [0427] d. .ltoreq.34.0 mmHg [0428] e. For subjects on topical prostaglandin therapy at screening: Is increased .gtoreq.5.0 mmHg from screening [0429] Baseline IOP measurements are .ltoreq.3 mmHg apart between 2 sequential baseline visits in each eye. [0430] Central corneal thickness .gtoreq.500 .mu.m and .ltoreq.600 .mu.m in each eye.
Study Treatments
L-PPDS Treatment
[0431] At the Day 0 visit, investigational L-PPDS with 95 .mu.g of Latanoprost was inserted into either the upper or lower puncta of each eye (depending on the treatment group). The other punctum of each eye had either a solid punctal plug (containing no latanoprost), or did not have a plug inserted, depending on the treatment group. The total latanoprost dose for each eye was 95 .mu.g. The plugs were removed at the Week 12 visit.
Study Variables
[0432] IOP, IOP change from baseline, and percentage IOP change from baseline; BCVA and change from baseline BCVA; Biomicroscopy examination variables; Subject tearing and comfort scores; Funduscopy variables; Automated perimetry variables; Adverse events (AEs); Concomitant medications; Proportion of subjects who lose an L-PPDS/punctal plug; Proportion of eyes that lose an L-PPDS/punctal plug; and Investigator assessment of L-PPDS insertion
Study Procedures and Assessments
[0433] IOP measurements were determined in each eye at the screening visit. The duration of the washout period depended on the time required for washout of topical ocular hypotensive therapy. Washout was not required for treatment-naive subjects. After the washout period, IOP measurements were determined in each eye on 2 separate visits, 2 to 4 days apart. For treatment-naive subjects the initial screening visit was considered as the first visit for determination of baseline IOP.
[0434] The L-PPDS treatment period was 12 weeks. Study visits occurred at Days 1, 3, 7, and 14, and Weeks 3, 4, 6, 8, 10 and 12. (Subjects who were prostaglandin-naive at study entry will also have a visit at Week 14 after the Xalatan run-out period is complete.) There was a follow-up telephone call 3 days after the last visit. The following tests and procedures were performed during study follow-up.
[0435] Goldmann IOP measurements (the average of 3 measurements) were measured in each eye at every visit. The baseline IOP was taken on 2 separate days, at least 48 hours apart. IOP measurements must be taken at 8:30 am (.+-.30 minutes) at each visit.
[0436] Visual acuity was measured with best correction or pinhole using a Snellen chart at every visit; the method (best correction or pinhole) were consistent for a given subject throughout the study.
[0437] Biomicroscopy examinations, including an inspection of L-PPDS placement was performed in each eye at every visit.
[0438] Subjects will rate the acceptability of the tearing and comfort level of the plugs, and the frequency of tearing, on a visual analog scale at each visit starting at Day 0. Subjects were also asked at the end of the study which they prefer: punctal plugs or eye drops.
[0439] Treatment-emergent ocular and systemic AEs and concomitant medications were monitored at every visit with standardized questioning techniques.
[0440] Investigators rated the ease of insertion of the plugs.
[0441] Automated perimetry was performed to measure visual fields at screening and Week 12.
[0442] A funduscopy examination was performed at screening and Week 12.
[0443] All ocular procedures are performed by an experienced and appropriately qualified individual(s). Subjects who discontinued the study treatment prematurely underwent the tests and procedures for the last visit.
[0444] It is unexpected for the L-PPDS to malfunction and release all or a major portion of its contents in a short period of time. The expected exposure to latanoprost (95 .mu.g delivered over 3 months) was within the safety profile established in a series of ocular toxicity studies conducted to support the FDA approval of Xalatan. Specifically, no adverse effects were observed in rabbits with twice-daily ocular instillation of latanoprost doses up to 50 .mu.g per eye for 52 weeks (100 .mu.g/eye/day). In a similar 52-week ocular study in cynomolgus monkeys, the only effects observed at doses up to 50 .mu.g/eye given twice daily (100 .mu.g/eye/day) were a reversible change in the aspect of the palpebral fissure and a non-reversible increase in iris pigmentation, which were not judged to be deleterious (Xalatan Product Monograph 2011). In a study of 28 healthy volunteers, in which 1 drop of latanoprost 50 .mu.g/mL was administered once daily in 1 eye and 4 times daily in the other eye for 2 weeks, transient photophobia, cells, and mild flare were common during the 4-dose regimen, but these effects resolved spontaneously without cessation of treatment (Linden and Alm, the effect on intraocular pressure of latanoprost once or four times daily. Br. J. Ophthalmol. 85:1163-1166 (2001)).
IOP Variables and Analyses
[0445] The primary efficacy variable was IOP change from baseline and the primary analysis time point will be at Week 4. The secondary efficacy variables were IOP and percentage IOP change from baseline.
[0446] Baseline IOP was defined as the average of 6 measurements: 3 measurements taken on Day -2 (end of screening period) and 3 measurements taken on Day 0 (before L-PPDS insertion).
[0447] The analyses of IOP variables were performed based on the values from each eye. For analyses using the ITT data set, all data from all eyes with at least 1 follow-up IOP measurement was included. For analyses using the evaluable (EVAL) data set, data from subjects, eyes or visits with significant protocol deviations were excluded.
[0448] The primary and secondary IOP variables were summarized for each treatment at each study visit using means with 95% CIs, standard deviations, minimums, medians and maximums. The primary and secondary IOP variables were summarized similarly based on the difference between treatments within a subject. The calculations of mean and 95% CI of the IOP change from baseline for each treatment at Week 4 is considered the primary analysis. All the other analyses are secondary analyses.
[0449] Interim results at week 8 (where n is the number of eyes), with the 3 different plug placement configurations, showed IOP decreases from baseline of -4.95 mmHg in Treatment group A (n=14), -4.31 mm Hg in treatment group B (n=14) and -6.07 mmHg in treatment group C (n=12). With the 95 .mu.g dose comparing 3 different plug placement configurations (n=40), IOP decreases from baseline at Week 8 ranged from -4.31 mmHg to -6.07 mmHg.
[0450] Final Study Results: A total of 77 subjects enrolled in the study. Men and women were represented approximately evenly (47% and 53%, respectively). Most subjects were Caucasian (70%) or Black (25%), with an average age of 66 years. Most eyes were assessed to have open angle glaucoma (74%; of those, most were primary [98%]), while the other eyes had ocular hypertension (26%). Mean IOP at screening was 18.02 mmHg overall (18.3, 17.9, and 17.9 mmHg for eyes who received Treatment A, B, and C, respectively); by baseline (after the washout period) mean IOP had increased to 25.39 mmHg overall (25.56, 25.00, and 25.61 mmHg for eyes who received Treatment A, B, and C, respectively).
[0451] At Week 4, 66 subjects (86%) had IOP measured; at Week 12, 49 subjects (64%) had IOP measured. A total of 53 subjects (69%) completed the study (Week 12),
[0452] The ITT data set included 154 eyes (53, 51, and 50 for Treatments A, B, and C, respectively), EVAL included 148 eyes (49, 51, and 48 for Treatments A, B, and C, respectively), and safety included all 77 subjects. Six eyes were completely excluded from the EVAL analysis (both eyes of 3 subjects, 3 eyes from Treatment A and 3 from Treatment C), and 2 additional eyes were excluded from Week 8 only (both eyes of 1 subject, Treatments A and B); all eyes were excluded because of concomitant medication.
[0453] Treatment with L-PPDS resulted in significant mean IOP decreases from baseline with all treatments across all time points (Table 8 and FIG. 23)
TABLE-US-00010 TABLE 8 Summary of IOP (mmHg) Results (ITT Observed) Treatment A Treatment B Treatment Total Visit (N = 53) (N = 51) C (N = 50) (N = 154) Day 14 n 51 48 45 144 Mean IOP 20.16 19.98 20.04 20.06 Mean IOP .dwnarw. -5.29 -4.86 -5.35 -5.17 CI (-6.29, -4.28) (-5.67, -4.05) (-6.17, -4.53) (-5.67, -4.66) Week 4 n 45 46 41 132 Mean IOP 20.44 20.51 19.96 20.32 Mean IOP .dwnarw. -4.84 -4.38 -5.07 -4.75 CI (-6.01, -3.66) (-5.33, -3.43) (-6.11, -4.04) (-5.35, -4.15) Week 8 n 63 41 35 112 Mean IOP 20.53 20.63 20.14 20.45 Mean IOP .dwnarw. -4.65 -4.22 -4.88 -4.56 CI (-5.72, -3.58) (-5.18, -3.25) (-5.77, -3.99) -5.11, -4.01) Week 12 n 31 37 30 98 Mean IOP 20.42 20.45 19.83 20.25 Mean IOP .dwnarw. -4.34 -4.21 -5.06 -4.51 CI (-5.32, -3.37) (-5.33, -3.09) (-6.20, -3.93) (-5.12, -3.90)
[0454] Across time points after Day 1, Treatment C (95 .mu.g L-PPDS lower punctum, blank plug upper punctum) resulted in the best mean IOP reduction compared with the other treatment groups. Results for ITT observed data including IOP after plug loss or removal (Table 8) were similar both to the results for ITT observed data excluding IOP after plug loss or removal and to EVAL data.
[0455] Retention rate of plugs in the lower puncta was .gtoreq.96% through Week 10 and 92% at Week 12. Retention of upper plugs was 76%, 65%, and 58% at Weeks 4, 8, and 12, respectively.
[0456] Study Conclusions: Treatment with L-PPDS (latanoprost dose of 95 .mu.g/eye) resulted in a clinically meaningful and statistically significant reduction in IOP from baseline of approximately 5 mmHg after 4 weeks (primary endpoint). The configuration of a 95 .mu.g L-PPDS in the lower punctum and a blank plug in the upper punctum (Treatment C) showef the best IOP reduction of the three configurations investigated.
Example 7: Discussion and Final Analysis of GLAU 12 and GLAU 13 Studies
[0457] The primary endpoint in the Phase II studies was the mean change in IOP from baseline (measured as mmHg) at 4 weeks. Secondary endpoints were the IOP change from baseline at other time points as well as the IOP and percentage IOP change from baseline at all time points in the 12-week study period.
[0458] A total of 57 ITT (Intent to Treat) subjects were included in the L-PPDS treatments in PPL GLAU 12, and a total of 77 ITT subjects were included in L-PPDS treatments in PPL GLAU 13. See, FIG. 21. Two ITT datasets were analyzed, one including all IOP values regardless of plug loss, and the other, or second group, with IOP excluded after first plug loss/removal. FIG. 23 summarizes IOP changes from baseline at 4, 8 and 12 weeks for the two studies for both ITT datasets. For both studies, mean IOP changes from baseline were statistically significant at all time points. Across the 5 treatment arms of both studies, 3 arms showed clinically significant IOP lowering of 5 mmHg or greater at 4 weeks for both datasets, and 2 arms showed clinically significant lowering of 5 mmHg or greater at 4 and 6 weeks for one ITT dataset (IOP excluded after plug loss). One arm (the 95 .mu.g lower/blank plug upper configuration (Treatment C)) showed a clinically significant IOP lowering of approximately 5.0 mmHg at 4, 8 and 12 weeks for both ITT datasets.
[0459] In GLAU 12, at the end of 4 weeks, the percentage of eyes with an IOP reduction vs baseline of 5 mmHg or greater was 70% for the 190 .mu.g dose and 58% for the 141 .mu.g dose. At 12 weeks these values were 45% of eyes at the 190 .mu.g dose and 27% of eyes at the 146 .mu.g dose (ITT observed data with IOP excluded after first plug loss/removal). See FIG. 30.
[0460] In GLAU 13, at the end of 4 weeks, the percentage of eyes with an IOP reduction vs. baseline of 5 mmHg or greater was 58% for the blank plug lower/95 .mu.g L-PPDS upper configuration (Treatment A), 50% for the open lower punctum/95 .mu.g L-PPDS upper configuration (Treatment B), and 57% for the 95 .mu.g L-PPDS lower/blank plug upper configuration (Treatment C); at 12 weeks these values were 52%, 59% and 66% for the three configurations, respectively (ITT observed data with IOP excluded after first plug loss/removal). See, FIG. 32.
[0461] During the 8-week second treatment course in GLAU 12 (n=38 eyes), the L-PPDS (190 .mu.g) produced a statistically and clinically significant reduction in mean IOP at 4 and 6 weeks of 5.4 and 5.8 mmHg, respectively for the all-observed IOP ITT dataset and 5.7 and 5.8 mm Hg, respectively for the IOP excluded after plug loss ITT dataset. See FIG. 26. Upper plug retention was notably higher compared to the main study, achieving values of 90 and 88% at 4 and 8 weeks, respectively, for this shorter re-treatment course. See, FIG. 36
[0462] The 95 .mu.g lower/blank upper configuration (Treatment C) demonstrated the most sustained IOP reduction (12 weeks) across all plug configurations and doses, suggesting IOP lowering with the L-PPDS as currently designed may be affected by the plug position (and tearing effects) of these designs. See, FIGS. 27 and 28. Results of these studies also suggest that double-plugging (simultaneous placement of both an upper and lower plug) may be necessary to achieve a minimum IOP lowering effect using the current design configurations.
[0463] Data from the high dose study (PPL GLAU 12) demonstrated that higher dose levels of the current designs produced the largest mean IOP change from baseline at 4 weeks of all configurations across these two studies. However, higher doses alone did not result in a sustained effect beyond 4 weeks, suggesting potentially different dose delivery mechanics with the higher dose plugs. In addition, the high dose effects observed in the repeat treatment phase of GLAU 12 (8 weeks at 190 ug for 19 subjects) show differing effects between the first course (12 weeks) and second course (8 weeks). With the second treatment course of L-PPDS at the 190 .mu.g dose over 8 weeks, the IOP lowering effect was sustained longer, until 6 weeks (both ITT datasets)
[0464] In PPL GLAU 12, 2 subjects discontinued from the study due to AEs, 17 discontinued due to plug loss, and 2 withdrew.
[0465] In PPL GLAU 13, 5 subjects discontinued from the study due to AEs, 14 discontinued due to plug loss, 8 discontinued due to inadequate IOP control and 1 withdrew.
Example 8: Retention Study (GLAU 11, 12 and 13 Studies)
[0466] A clinical study (Glau 11) was conducted to evaluate exemplary embodiments of the present invention in comparison with a modified commercial implant. The commercial implant 1000 is illustrated in FIGS. 10A and 10B, where a side view and a top view are depicted respectively along with corresponding major dimensions. The commercial implant 1000 has no cavity. For the purpose of comparison study, the commercial implant 1000 is modified by constructing a cavity 1002 in the implant 1000, as shown in FIGS. 10C and 10D. The cavity 1002 is configured such that it has essentially the same shape and the same size as of the cavity 458 in the exemplary embodiments of the present invention selected for the comparison study.
[0467] The comparison study involves ninety six subjects, baseline demographics of which are provided in FIG. 11. Each subject is fitted with two modified commercial implants indicated and two selected exemplary embodiments of the present invention. The modified commercial implants are referred as upper implants and the selected exemplary embodiments of the present application as lower implants. Both upper and lower implants contain 141 .mu.g of total latanoprotst drug stored in their respective cavities. The 141 .mu.g of total latanoprotst drug is consistent with three months of Xalatan drops.
[0468] The study was conducted over four weeks. During the study, the subjects were examined and the intraocular pressure was checked weekly. The observed retention rate is plotted and illustrated in FIG. 9. In the study as well as in the present invention, the retention rate is defined as the percentage of eyes that retains implants over a certain period of time. As indicated by the plot in FIG. 9, the selected exemplary embodiments of the present invention achieves higher retention rates than the modified commercial implants. For example, while the retention rate of the modified commercial implants declines to a rate below 60% in week three, the retention rate of the selected exemplary embodiments of the present invention maintains at a relatively higher rate, approximately 95% or more, over the entire clinical trial. Having higher retention rate over a longer period of time is one of various advantages of embodiments of the present invention.
[0469] Retention rates of different plug designs (FIG. 33) were also evaluated in the GLAU 12 and 13 studies.
[0470] Retention rate by eye of plugs in the lower puncta was >95% through week 12 in PPL GLAU 12 (FIGS. 34 and 35) and through week 10 in PPL GLAU 13 (week 12 retention was 92%) (FIGS. 37 and 38). Retention of upper plugs by eye was 69%, 53% and 48% at weeks 4, 8 and 12, respectively, in PPL GLAU 12. In PPL GLAU 13, retention of upper plugs was 76%, 65% and 58% at weeks 4, 8 and 12, respectively. In addition, for eyes that retained the plugs past 4 weeks the rates the plugs were lost slowed, See FIG. 39.
[0471] Upper plug retention with the proprietary punctal plugs was notably improved (approximately 19-33%) over the commercial plugs used in Study PPL GLAU 11. At 4 weeks, the upper plug retention by eye had increased from 48% in GLAU 11 to 67-81% in PPL GLAU 12 and PPL GLAU 13.
[0472] Upper plug retention was notably improved (by approximately 26%) over the commercial plugs used in GLAU 11:
a. At 4 weeks: i. The upper plug retention increased from 45% in GLAU 11 to 71% for the 141 .mu.g total dose in GLAU 12 ii. The upper plug retention for the higher 190 .mu.g total dose was similar to improvements for the 141 .mu.g dose, at 67% iii. The upper plug retention for the lower dose plug combinations (95 .mu.g) ranged from 77%-81% across the 3 treatment arms
[0473] At 8 weeks, 48%-69% upper plug retention across both studies
i. Upper plug retention for the 190 and 141 .mu.g combinations ranged from 48%-58% respectively ii. Upper plug retention for the lower dose (95 .mu.g) combinations ranged from 62%-69% across the 3 combinations
[0474] At 12 weeks, 42%-64% upper plug retention across both studies:
i. Upper plug retention for the 190 and 141 .mu.g combinations ranged from 42%-55% respectively ii. Upper plug retention for the lower dose (95 .mu.g) combinations ranged from 52%-64% across the three combinations
[0475] The above detailed description is intended to be illustrative, and not restrictive. For example, the above-described examples (or one or more features thereof) may be used in combination with each other. As an example, one or more dimensions from the various implant embodiments shown or described may be grouped together to form an implant embodiment capable of providing a desired drug concentration. Other embodiments may be used, such as by one of ordinary skill in the art upon reviewing the above description. Also, in the above Detailed Description, various features may be grouped together to streamline the disclosure. This should not be interpreted as intending that an unclaimed disclosed feature is essential to any claim. Rather, inventive subject matter may be in less than all features of a particular disclosed embodiment. Thus, the following claims are hereby incorporated into the Detailed Description, with each claim standing on its own as a separate embodiment. The scope of the invention should be determined with reference to the appended claims, along with the full scope of equivalents to which such claims are entitled.
[0476] The Abstract is provided to comply with 37 C.F.R. .sctn. 1.72(b), to allow the reader to quickly ascertain the nature of the technical disclosure. It is submitted with the understanding that it will not be used to interpret or limit the scope or meaning of the claims.
* * * * *
References

D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

D00013
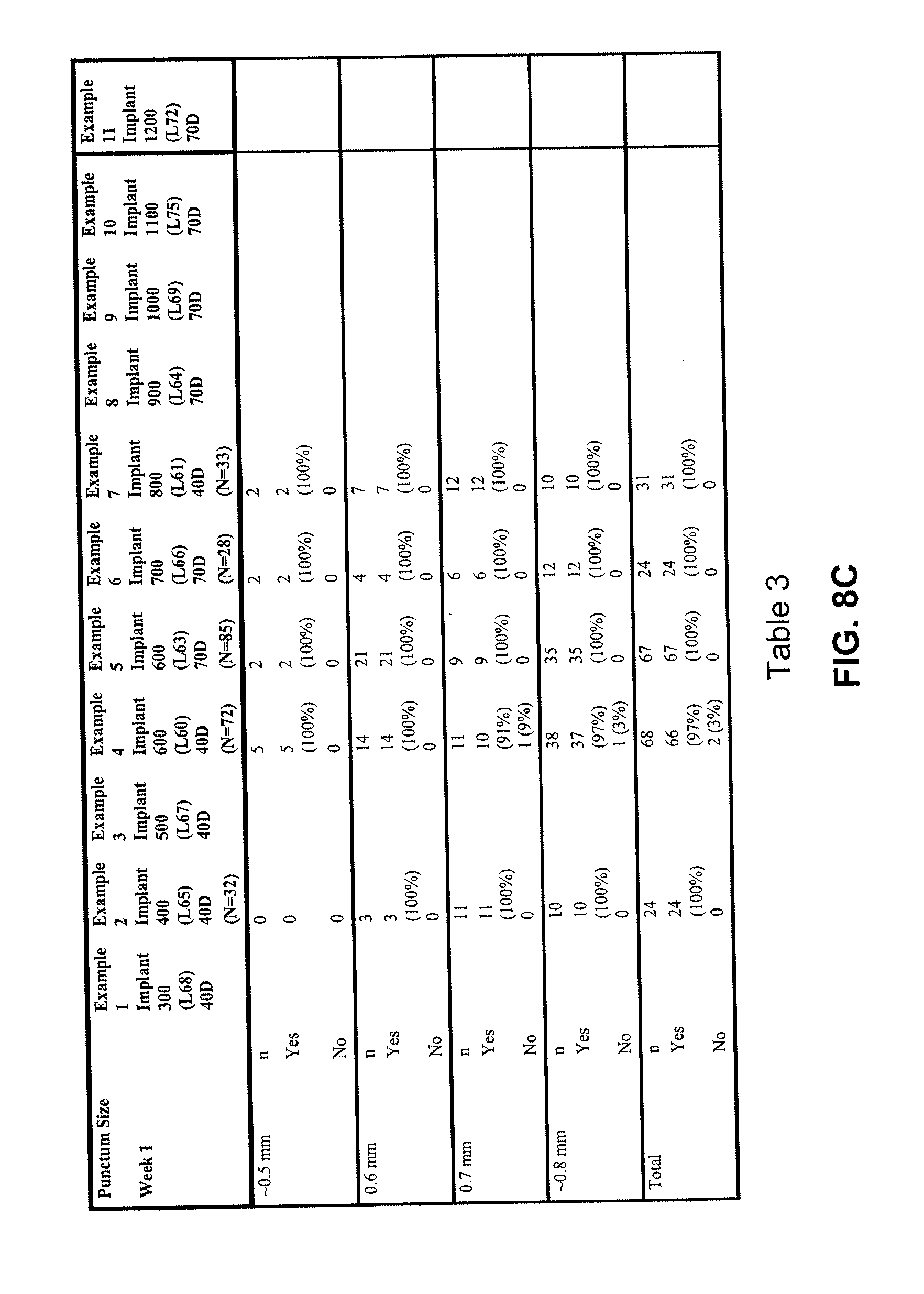
D00014

D00015
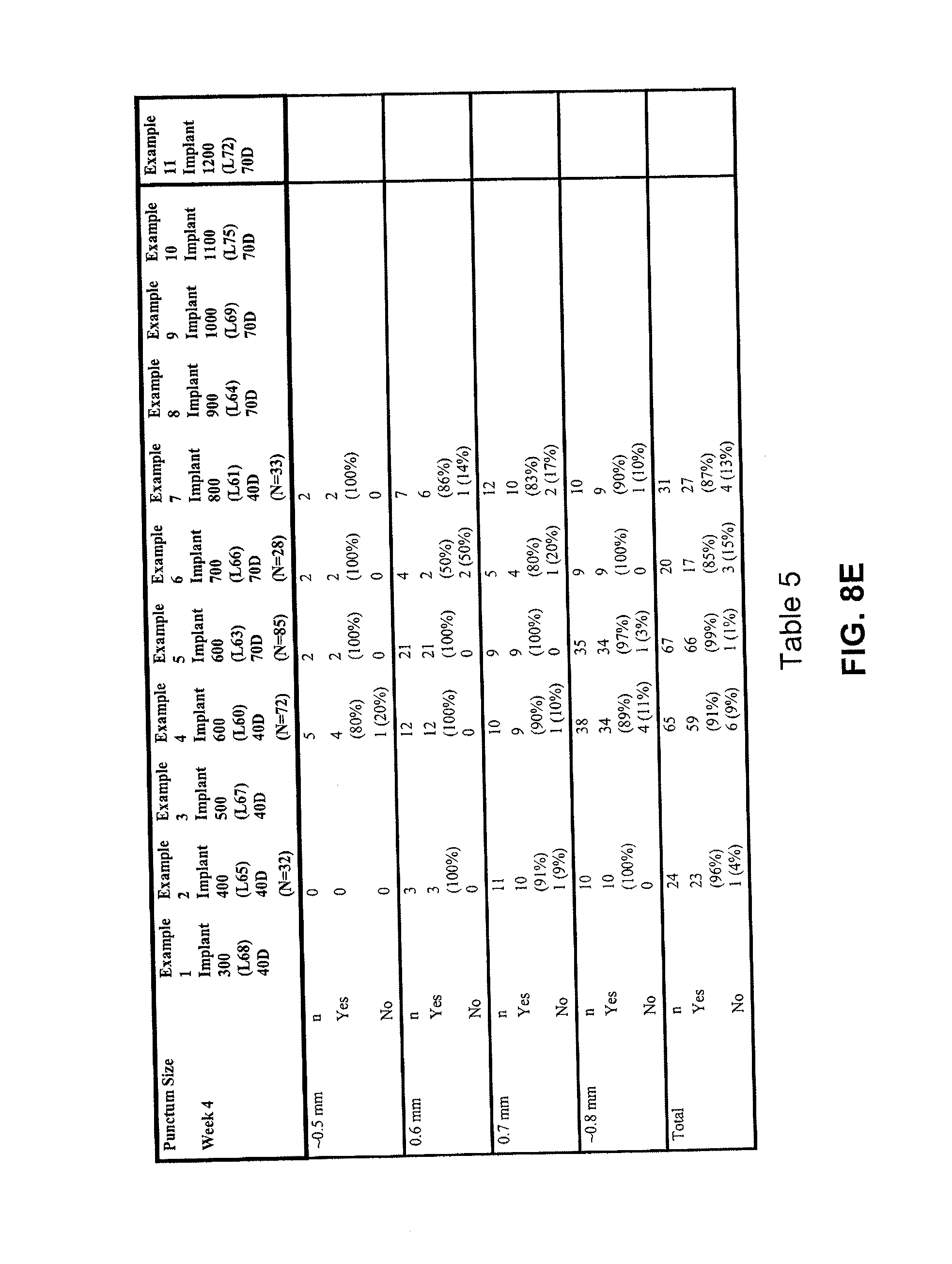
D00016
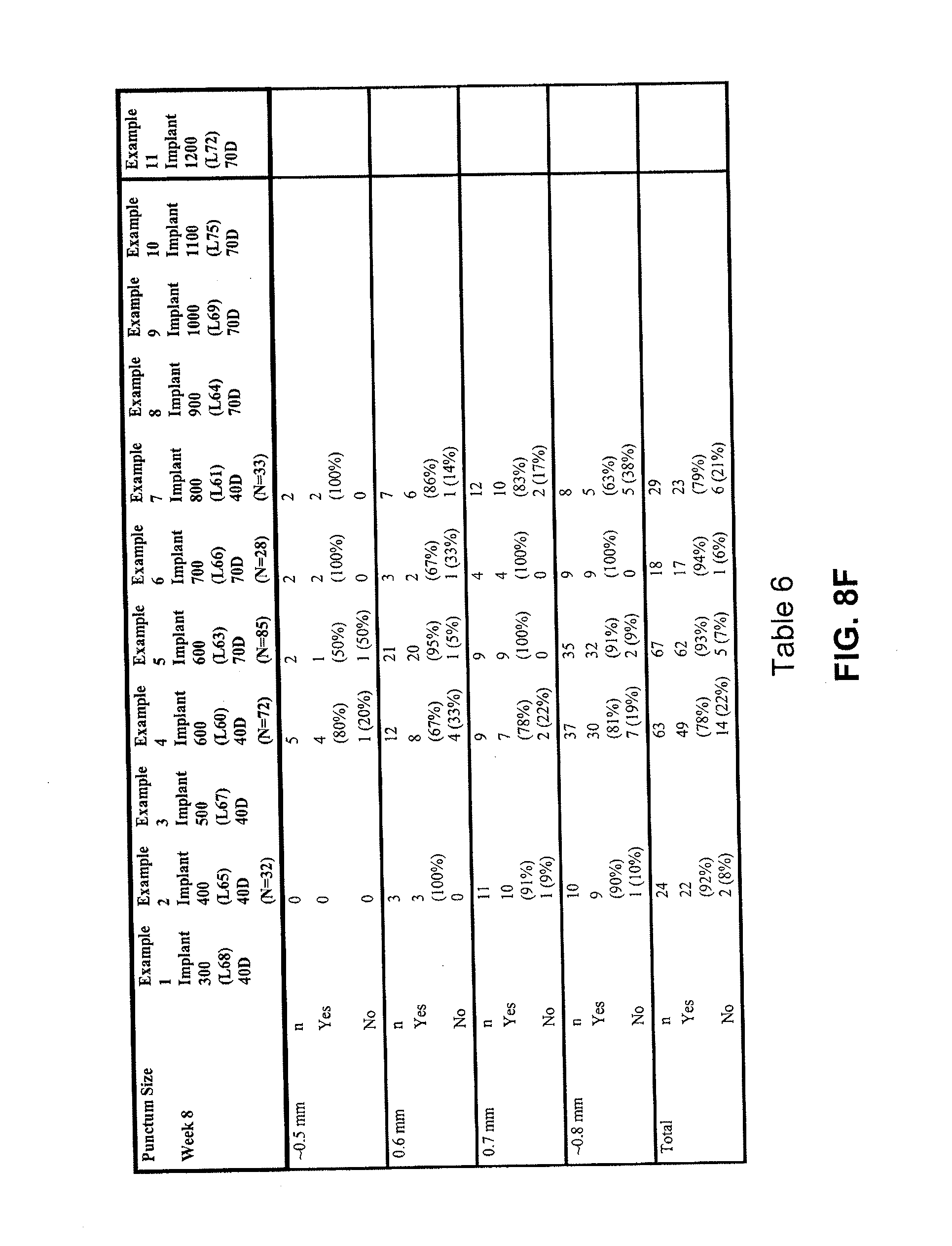
D00017

D00018

D00019

D00020
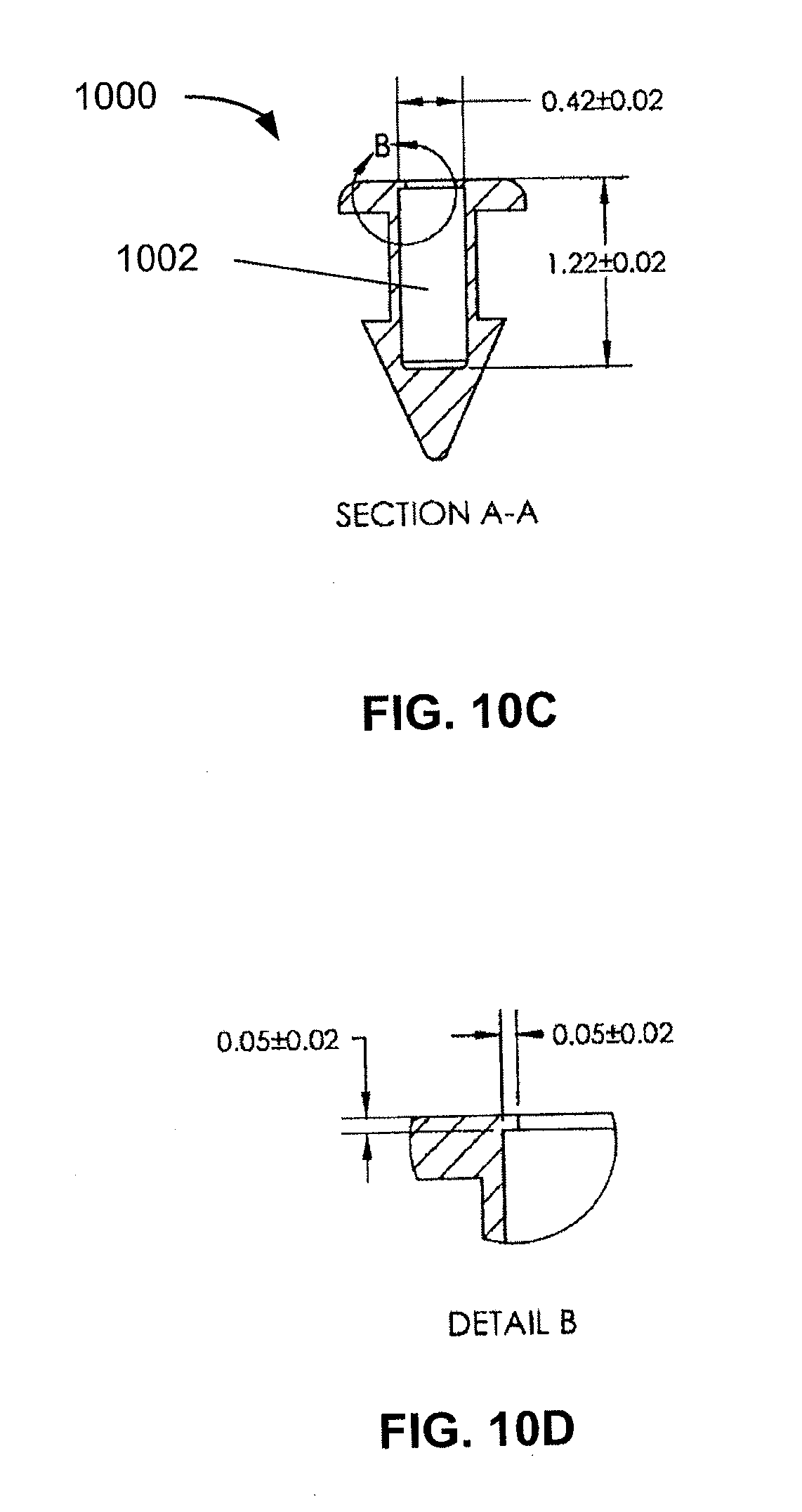
D00021

D00022

D00023

D00024

D00025

D00026
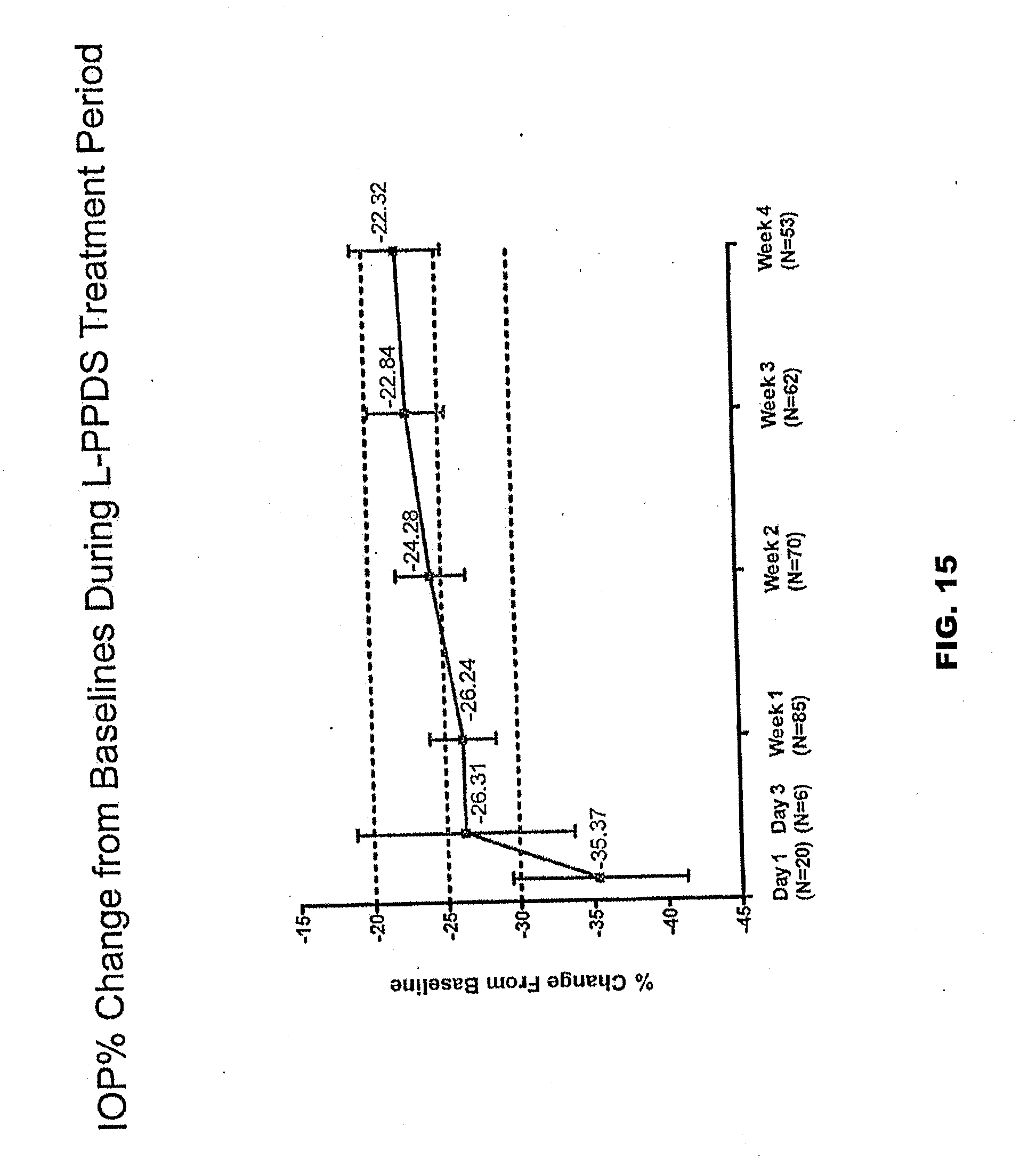
D00027

D00028
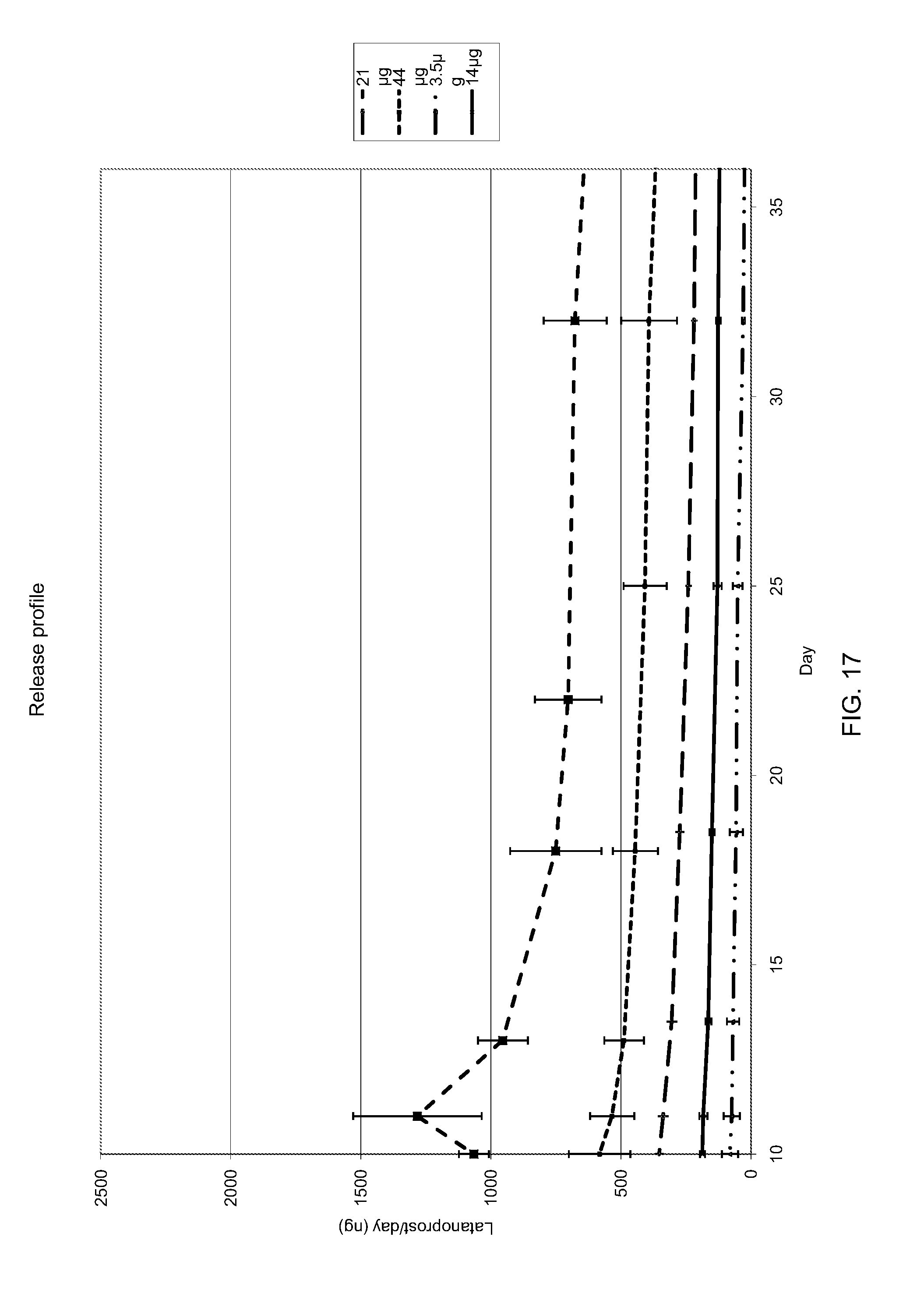
D00029

D00030

D00031
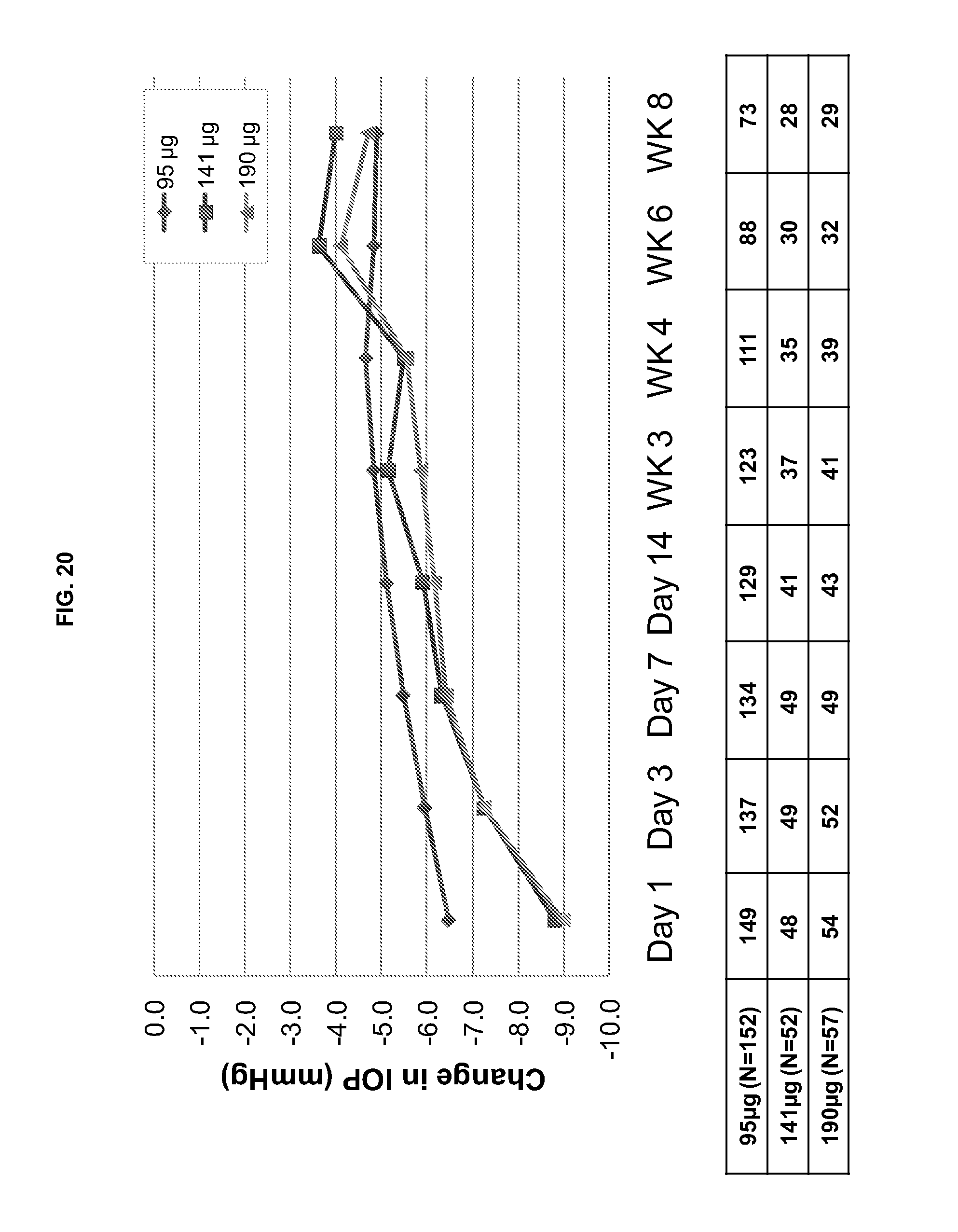
D00032

D00033

D00034
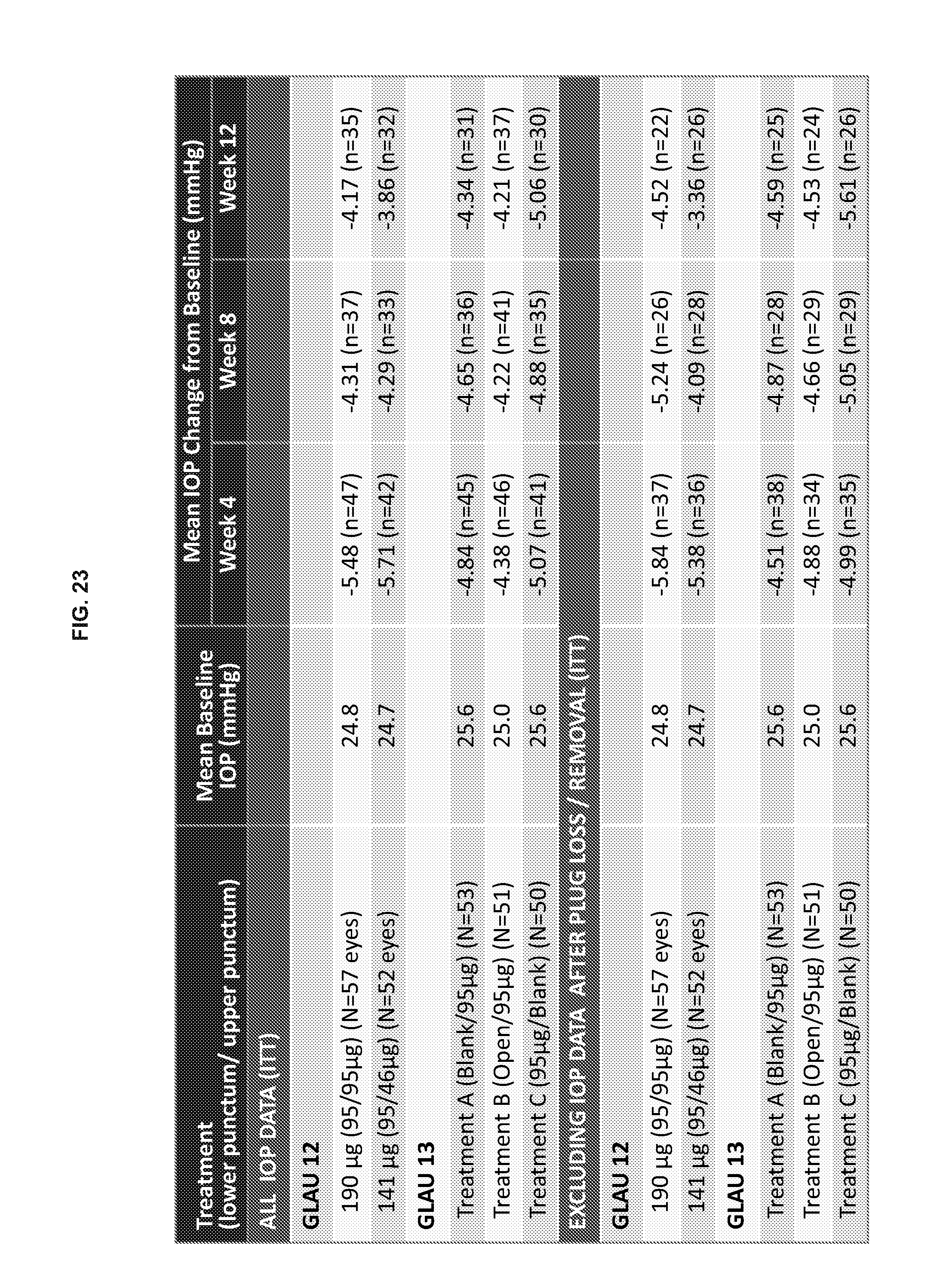
D00035

D00036
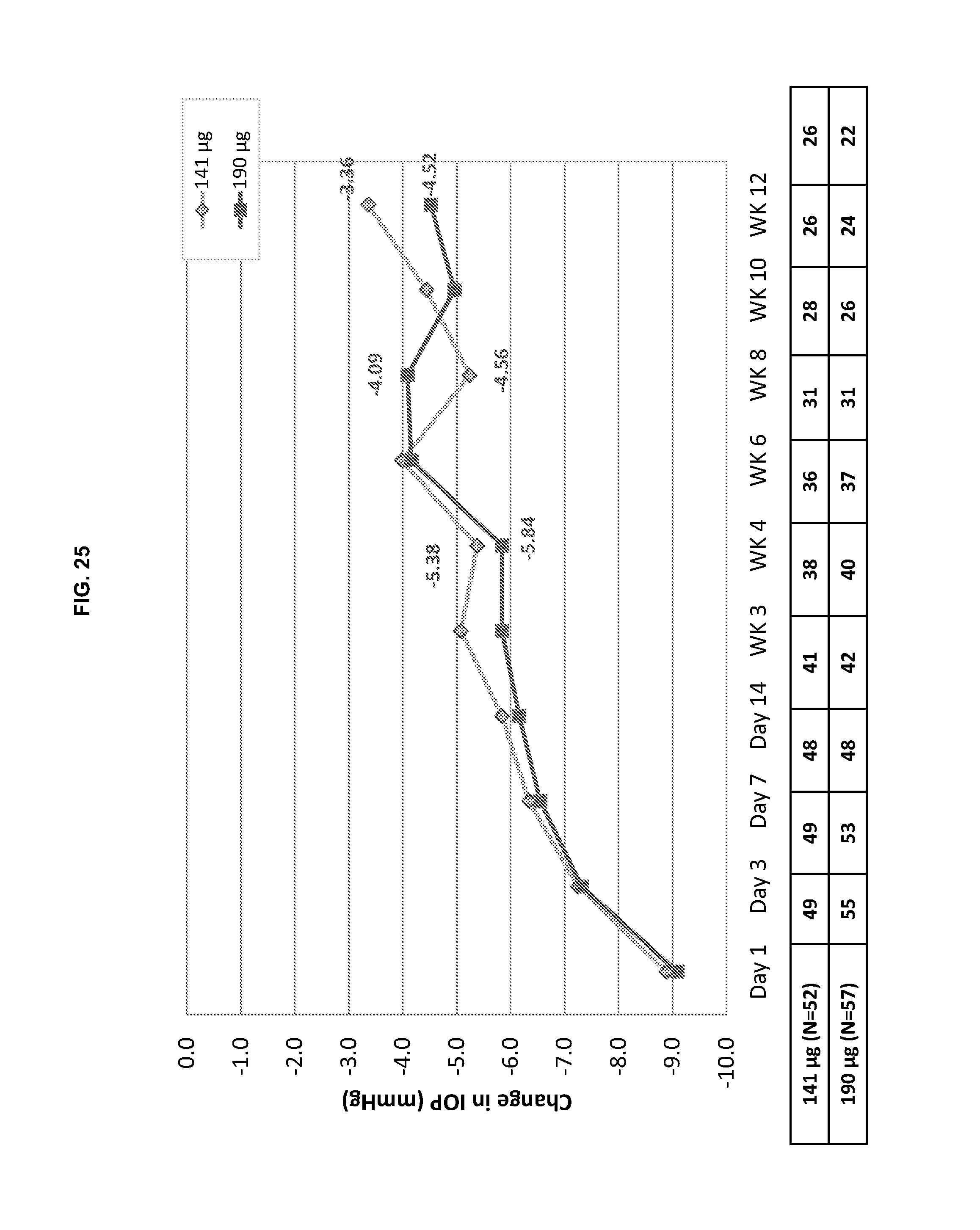
D00037

D00038

D00039

D00040

D00041

D00042
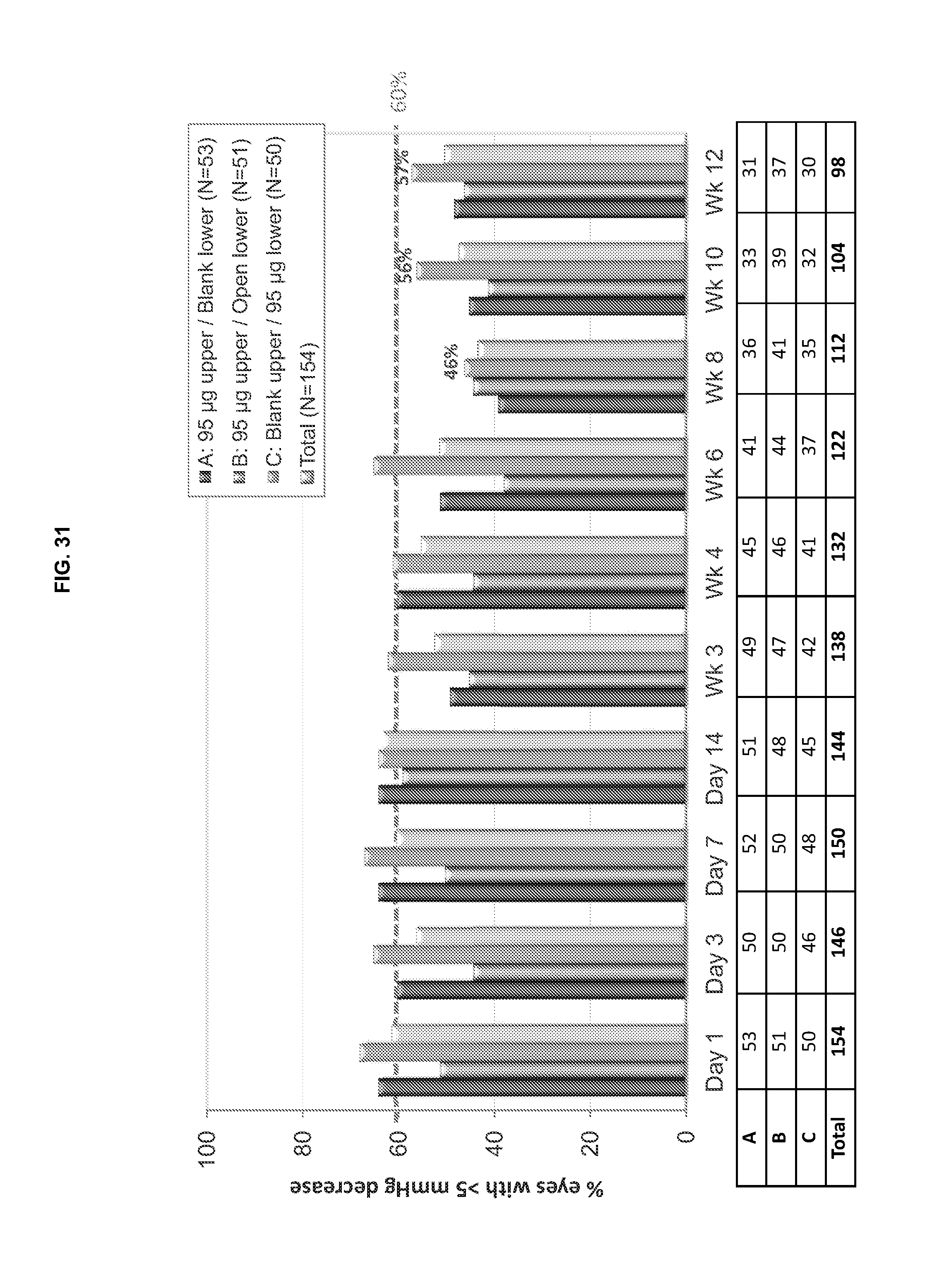
D00043
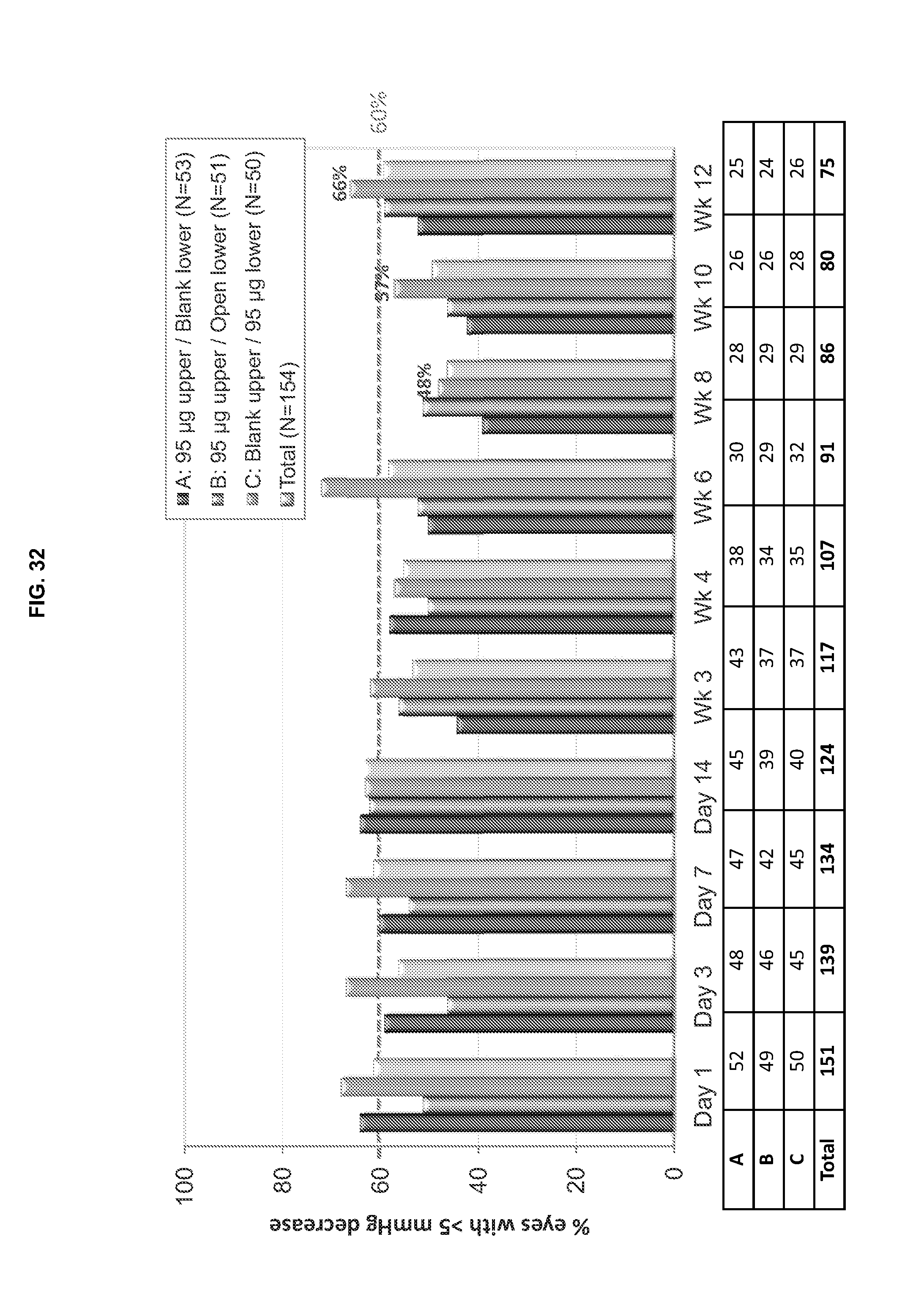
D00044

D00045
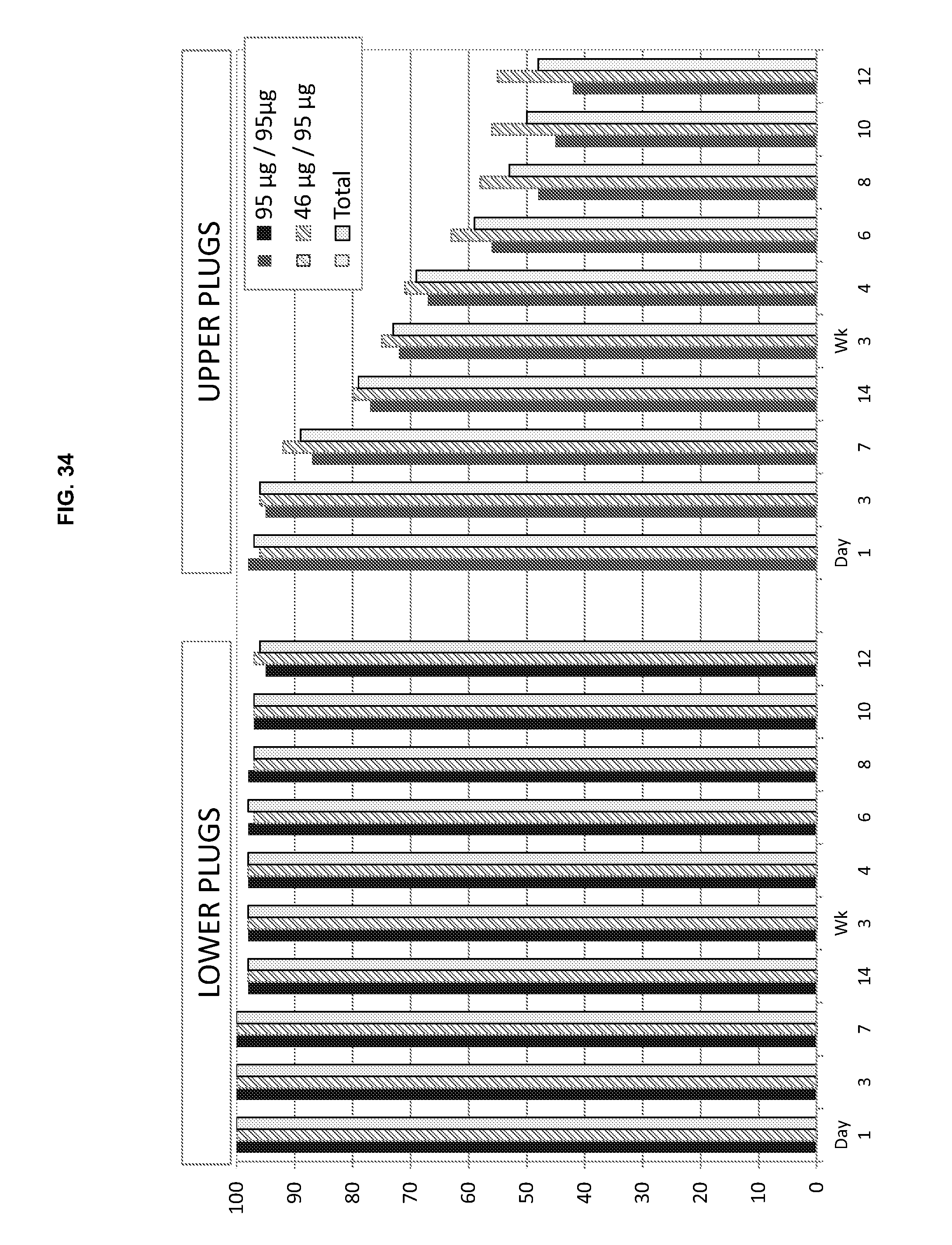
D00046

D00047
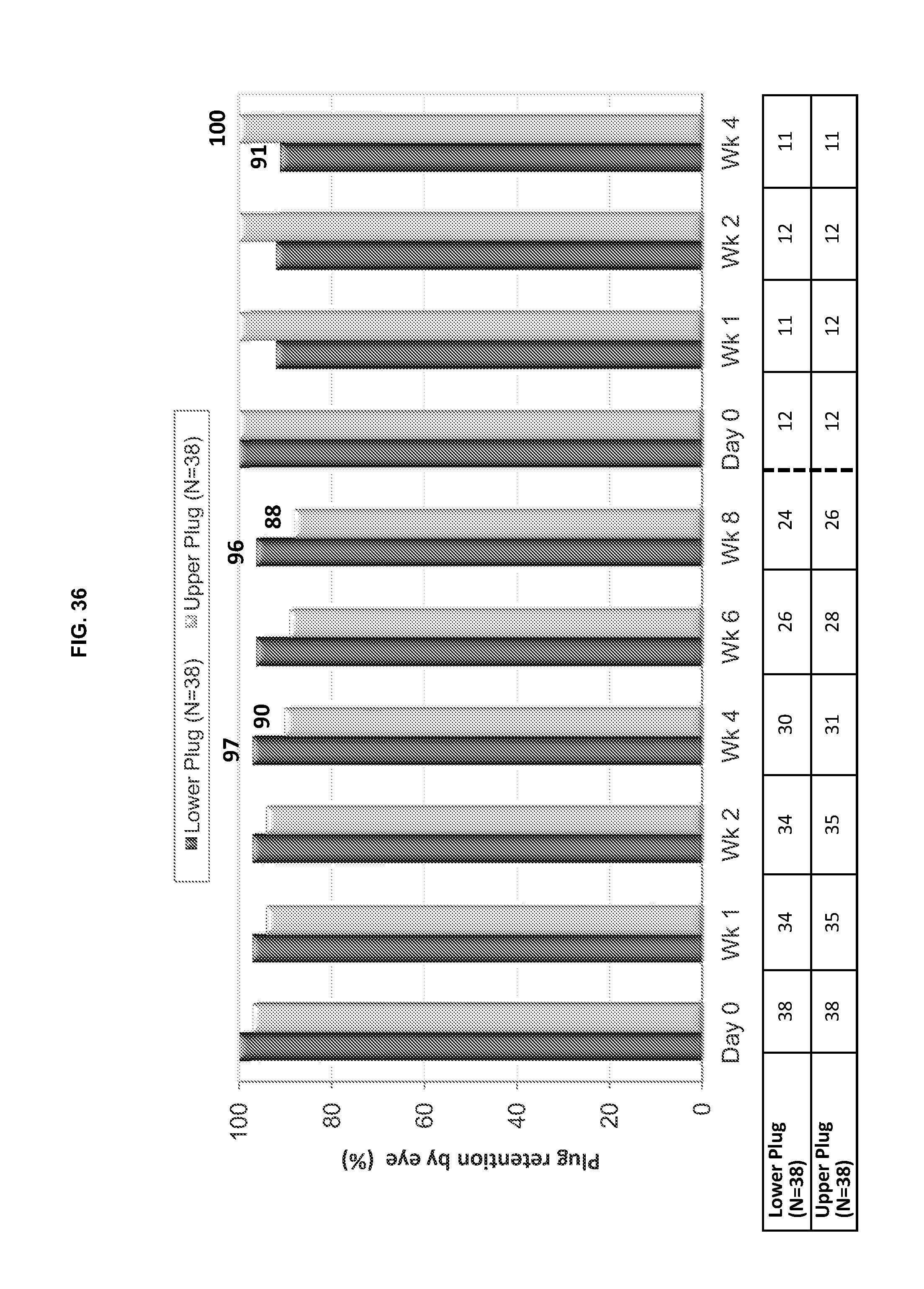
D00048

D00049

D00050
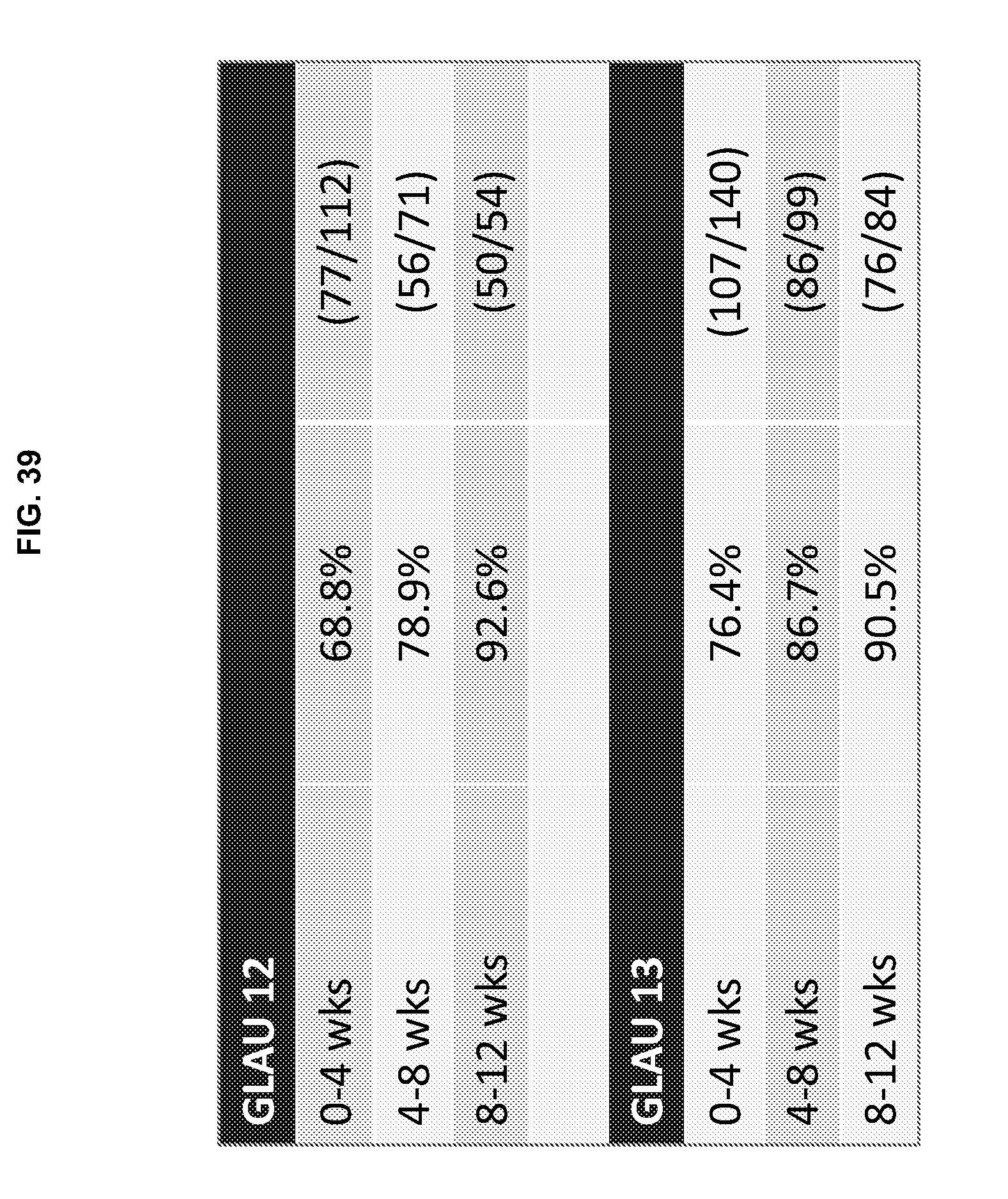
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.