Method For Treating Male Sexual Dysfunction By Using Underpants Comprising Far-infrared Fibers
Liang; Tien-Show
U.S. patent application number 15/709516 was filed with the patent office on 2019-03-21 for method for treating male sexual dysfunction by using underpants comprising far-infrared fibers. The applicant listed for this patent is GREEN ENERGY NANO TECHNOLOGY CO., LTD.. Invention is credited to Tien-Show Liang.
| Application Number | 20190083807 15/709516 |
| Document ID | / |
| Family ID | 65719724 |
| Filed Date | 2019-03-21 |








View All Diagrams
| United States Patent Application | 20190083807 |
| Kind Code | A1 |
| Liang; Tien-Show | March 21, 2019 |
METHOD FOR TREATING MALE SEXUAL DYSFUNCTION BY USING UNDERPANTS COMPRISING FAR-INFRARED FIBERS
Abstract
Differing from conventional approaches for improving sexual dysfunction of an adult man being commonly carried out by letting the adult man administer a dosage of synthetic drug or a composition of Chinese herbal medicines, the present invention particularly discloses a method for treating male sexual dysfunction by using underpants comprising far-infrared fibers. A variety of clinical data have proved that, after wearing this underpants for 3 months, both the men diagnosed with sexual dysfunction and the men not sure to suffer from sexual dysfunction have an apparent progress on improvements of their erectile ability, erectile quality, ejaculation control ability, and lower urinary tract symptoms (LUTS). Moreover, clinical data have also proved that, despite the fact that a male person wears the underpants provided by the present invention for three months, there is no adverse effect induced by the underpants for causing the physiological condition of the men be abnormal.
| Inventors: | Liang; Tien-Show; (Taipei City, TW) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 65719724 | ||||||||||
| Appl. No.: | 15/709516 | ||||||||||
| Filed: | September 20, 2017 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | G09B 3/10 20130101; A61N 2005/066 20130101; A41B 9/02 20130101; A41B 2400/32 20130101; A61N 2005/0645 20130101; G09B 3/06 20130101; A61N 5/0613 20130101; A61N 2005/063 20130101; A41B 17/00 20130101; A41B 2500/20 20130101 |
| International Class: | A61N 5/06 20060101 A61N005/06; A41B 9/02 20060101 A41B009/02; A41B 17/00 20060101 A41B017/00; G09B 3/10 20060101 G09B003/10; G09B 3/06 20060101 G09B003/06 |
Claims
1. A method for treating male sexual dysfunction by using underpants comprising far-infrared fibers, comprising following steps: (1) providing an underpants weaved from a plurality of far-infrared fibers and a plurality of fibers, wherein each of the far-infrared fibers comprises a polymer sheath and far-infrared powders doped in or enclosed by the polymer sheath; moreover, the far-infrared fibers having a fiber amount occupying 20-50 percent of a total fiber amount of the underpants; and (2) letting adult men diagnosed with sexual dysfunction or not sure to suffer from sexual dysfunction wear the underpants for at least one month.
2. The method of claim 1, wherein the type of the underpants is selected from the group consisting of briefs, boxer briefs and boxer shorts.
3. The method of claim 1, wherein the manufacturing material of the polymer sheath is selected from the group consisting of polyester (PET), polyurethane (PU), Poly(vinyl chloride) (PVC), poly propylene (PP), and polyamide (PA).
4. The method of claim 1, wherein the polymer sheath is made of a polymer compound comprising silica of at least 50 wt %, and the chemical structure of the polymer compound is presented by following chemical formula 1: ##STR00002## wherein n in the chemical formula 1 is in a range from 50 to 100.
5. The method of claim 1, wherein the far-infrared powders comprises powdered titanium (Ti) with a first metal weight percent of 11-18 wt %, powdered germanium (Ge) with a second metal weight percent of 0.1-0.2 wt % and powdered zinc (Zn) with a third metal weight percent of 0.05-0.15 wt %.
6. The method of claim 5, wherein the far-infrared fibers are able to emit a far-infrared ray with a wavelength in a range from 2 .mu.m to 22 .mu.m, and the emissivity of the far-infrared ray is above 90%.
7. The method of claim 1, wherein the far-infrared powders further comprises powdered oxide and powdered carbide for increasing the emissivity of the far-infrared ray.
8. The method of claim 7, wherein the powdered oxide is selected from the group consisting of: Al.sub.2O.sub.3, MgO, NiO.sub.2, SiO.sub.2, ZrO.sub.2, and a mixture made by any two or more aforesaid materials.
9. The method of claim 7, wherein the powdered carbide is selected from the group consisting of: TaC, ZrC, SiC, and a mixture made by any two or more aforesaid materials.
10. A method for treating male sexual dysfunction by using underpants comprising far-infrared fibers, comprising following steps: (1) providing an underpants weaved from a plurality of far-infrared fibers, wherein each of the far-infrared fibers comprises a polymer sheath and far-infrared powders doped in or enclosed by the polymer sheath; and (2) letting adult men diagnosed with sexual dysfunction or not sure to suffer from sexual dysfunction wear the underpants for at least one month.
11. The method of claim 10, wherein the type of the underpants is selected from the group consisting of briefs, boxer briefs and boxer shorts.
12. The method of claim 10, wherein the manufacturing material of the polymer sheath is selected from the group consisting of polyester (PET), polyurethane (PU), Poly(vinyl chloride) (PVC), poly propylene (PP), and polyamide (PA).
13. The method of claim 10, wherein the polymer sheath is made of a polymer compound comprising silica of at least 50 wt %, and the chemical structure of the polymer compound is presented by following chemical formula 1: ##STR00003## wherein n in the chemical formula 1 is in a range from 50 to 100.
14. The method of claim 10, wherein the far-infrared powders comprises powdered titanium (Ti) with a first metal weight percent of 11-18 wt %, powdered germanium (Ge) with a second metal weight percent of 0.1-0.2 wt % and powdered zinc (Zn) with a third metal weight percent of 0.05-0.15 wt %.
15. The method of claim 14, wherein the far-infrared fibers are able to emit a far-infrared ray with a wavelength in a range from 2 .mu.m to 22 .mu.m, and the emissivity of the far-infrared ray is above 90%.
16. The method of claim 14, wherein the far-infrared powders further comprises powdered oxide and powdered carbide for increasing the emissivity of the far-infrared ray.
17. The method of claim 16, wherein the powdered oxide is selected from the group consisting of: Al.sub.2O.sub.3, MgO, NiO.sub.2, SiO.sub.2, ZrO.sub.2, and a mixture made by any two or more aforesaid materials.
18. The method of claim 16, wherein the powdered carbide is selected from the group consisting of: TaC, ZrC, SiC, and a mixture made by any two or more aforesaid materials.
Description
BACKGROUND OF THE INVENTION
1. Field of the Invention
[0001] The present invention relates to the technology field of far-infrared fibers, and more particularly to a method for treating male sexual dysfunction by using underpants comprising far-infrared fibers.
2. Description of the Prior Art
[0002] Male sexual dysfunction includes erectile dysfunction (ED), premature ejaculation (PE), delayed ejaculation, and low libido, wherein mental factors, physiological factors and drug factors are known able to cause the male sexual dysfunction. Moreover, clinical research reports that mental factor may formed by melancholia, low self-confidence, and anxiety of sexual performance, and the physiological factor is commonly resulted from cardiovascular diseases, endocrine system diseases and disorders, or injury or fracture of penile corpus cavernosum.
[0003] Statistical data indicate that, about half of all men between the ages of 40 and 70 suffer from erectile dysfunction (ED) to some degree. ED is one of the most common chronic diseases affecting men and its prevalence increases with aging.
[0004] Phosphodiesterase-5 inhibitors (PDE5-I) are commonly used for on-demand or chronic treatment of ED. Viagra (sildenafil), Levitra (vardenafil) and Cialis (tadalafil) are currently known prescription ED drugs comprising PDE5-I. By dilating blood vessels and relaxing smooth muscle tissues in penis, these drugs successfully improve or treat erectile dysfunction. However, this is the result of a series of conditions occurring simultaneously. When ED drug is taken correctly, it is no longer a concern. Nevertheless, in the mind of the public, use of the ED drug is at this time invariably considered dangerous for patients with heart disease. Moreover, it is well known that these ED drugs may cause unwanted side effects that require medical attention.
[0005] In view of the fact that most of ED drugs induce unwanted side effects, U.S. Pat. No. 7,147,874 discloses a pharmaceutical composition for prevention and treatment of premature ejaculation. The pharmaceutical composition comprises an extract of Bufonis Venenum and an extract of Ginseng. To carry out treatment of premature ejaculation, 0.2 g of gel pharmaceutical composition is applied to an adult man's penis glans, such that the contraction of penile smooth muscle is reduced so as to extend erection time of the adult man. However, the pharmaceutical composition is just used to prevent men from premature ejaculation during his sexual intercourse, but fail to provide a real improvement on solving sexual dysfunction to the men.
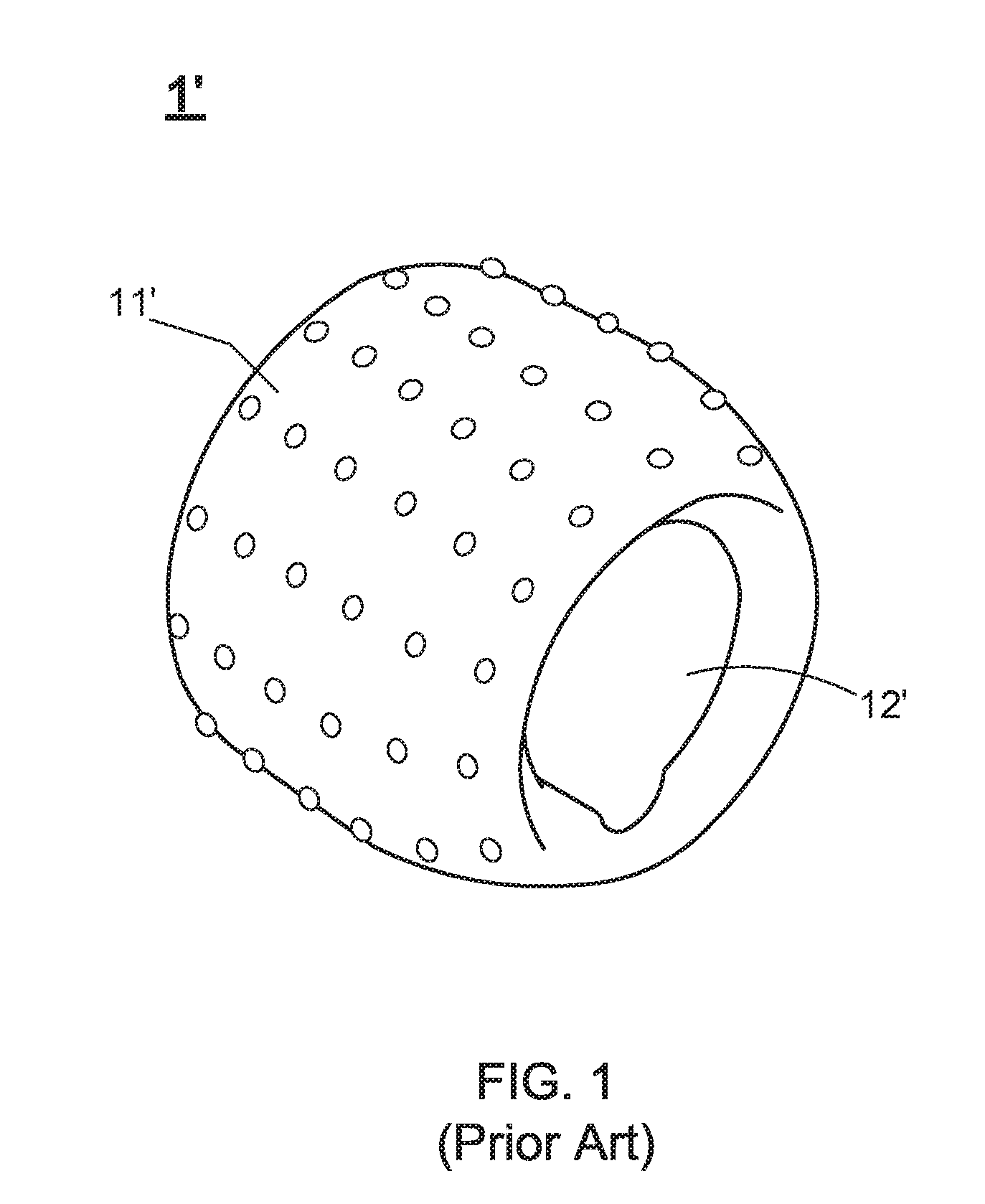
[0006] Accordingly, U.S. Pat. No. 8,211,006 discloses a penis erection stabilizer. FIG. 1 shows a stereo diagram of the penis erection stabilizer. From FIG. 1, it is understood that the penis erection stabilizer 1' comprises a stabilizer body 11' and a through-hole 12' defined through a center portion of the stabilizer body 11'. It is well know that, the blood supplied to the spongy tissue of an adult man's penis would flow back via vein when the adult man ejaculates or loses tension due to fatigue under sexual intercourse. In the meantime, because the adult man's penis is sleeved with the penis erection stabilizer 1', the blood in the expanded penis should first pass through a small diameter portion of the through-hole 12', such that the small diameter portion keeps the blood so as to retard the discharge of the blood. Eventually, the erected state of the penis can be maintained for a time sufficient for the completion of the sexual intercourse. It is worth noting that, owing to the fact that the use way of the penis erection stabilizer 1' is to sleeve on user's penis, it is presumably that frequently-repeating use of the penis erection stabilizer 1' would cause the injury of the penis.
[0007] From above descriptions, it is clear that both the pharmaceutical composition for prevention of premature ejaculation and the penis erection stabilizer 1' show practical-application drawbacks. In view of that, inventors of the present application have made great efforts to make inventive research thereon and eventually provided a method for treating male sexual dysfunction by using underpants comprising far-infrared fibers.
SUMMARY OF THE INVENTION
[0008] Differing from the fact that conventional approaches for improving sexual dysfunction of an adult man are commonly carried out by letting the adult man administer a dosage of synthetic drug or a composition of Chinese herbal medicines, the present invention particularly discloses a method for treating male sexual dysfunction by using underpants comprising far-infrared fibers. A variety of clinical data have proved that, after wearing this underpants for 3 months, both the men diagnosed with sexual dysfunction and the men not sure to suffer from sexual dysfunction have an apparent progress on improvements of their erectile ability, erectile quality, ejaculation control ability, and lower urinary tract symptoms (LUTS). Moreover, clinical data have also proved that, despite the fact that a male person wears the underpants provided by the present invention for three months, there is no any adverse effect induced by the underpants so as to cause the physiological condition of the men be abnormal.
[0009] In order to achieve the primary objective of the present invention, the inventor of the present invention provides an embodiment of the method for treating male sexual dysfunction by using underpants, comprising following steps: [0010] (1) providing an underpants weaved from a plurality of far-infrared fibers and a plurality of fibers, wherein each of the far-infrared fibers comprises a polymer sheath and far-infrared metal powders doped in or enclosed by the polymer sheath; moreover, the far-infrared fibers having a fiber amount occupying 40-60 percent of a total fiber amount of the underpants; and [0011] (2) letting adult men diagnosed with sexual dysfunction or not sure to suffer from sexual dysfunction wear the underpants for at least one month.
[0012] Moreover, for achieving the primary objective of the present invention, the inventor of the present invention further provides another one embodiment of the method for treating male sexual dysfunction by using underpants, comprising following steps: [0013] (1) providing an underpants weaved from a plurality of far-infrared fibers, wherein each of the far-infrared fibers comprises a polymer sheath and far-infrared powders doped in or enclosed by the polymer sheath; and [0014] (2) letting adult men diagnosed with sexual dysfunction or not sure to suffer from sexual dysfunction wear the underpants for at least one month.
[0015] In the two embodiments of the method, wherein the type of the underpants is selected from the group consisting of briefs, boxer briefs and boxer shorts.
[0016] In the two embodiments of the method, wherein the far-infrared powders comprises powdered titanium (Ti) with a first metal weight percent of 11-18 wt %, powdered germanium (Ge) with a second metal weight percent of 0.1-0.2 wt % and powdered zinc (Zn) with a third metal weight percent of 0.05-0.15 wt %.
[0017] In the two embodiments of the method, wherein the far-infrared fibers are able to emit a far-infrared ray with a wavelength in a range from 2 .mu.m to 22 .mu.m, and the emissivity of the far-infrared ray is above 90%.
[0018] In the two embodiments of the method, wherein the far-infrared powders further comprises powdered oxide and powdered carbide for increasing the emissivity of the far-infrared ray.
[0019] In the two embodiments of the method, wherein the far-infrared powders further comprises other powdered metal selected from the group consisting of powdered copper (Cu), powdered silver (Ag), powdered aluminum (Al), powdered magnesium (Mg), a mixture made by any two or more aforesaid materials.
BRIEF DESCRIPTION OF THE DRAWINGS
[0020] The invention as well as a preferred mode of use and advantages thereof will be best understood by referring to the following detailed description of an illustrative embodiment in conjunction with the accompanying drawings, wherein:
[0021] FIG. 1 shows a stereo diagram of a penis erection stabilizer;
[0022] FIG. 2A and FIG. 2B show stereo diagrams of a first embodiment of an underpants capable of improving male sexual dysfunction;
[0023] FIG. 3 shows a cross-sectional view of one far-infrared fiber;
[0024] FIG. 4A, FIG. 4B and FIG. 4C show a schematic production processing flow of the underpants capable of improving male sexual dysfunction;
[0025] FIG. 5 shows a stereo diagram of a second embodiment of the underpants capable of improving male sexual dysfunction;
[0026] FIG. 6 shows a stereo diagram of a third embodiment of the underpants capable of improving male sexual dysfunction;
[0027] FIG. 7 shows a diagram for introducing an IIEF-5 questionnaire;
[0028] FIG. 8 shows a diagram for introducing a QEQ questionnaire;
[0029] FIG. 9 shows a diagram for introducing a PEDT questionnaire;
[0030] FIG. 10 shows a diagram for introducing an IPSS questionnaire;
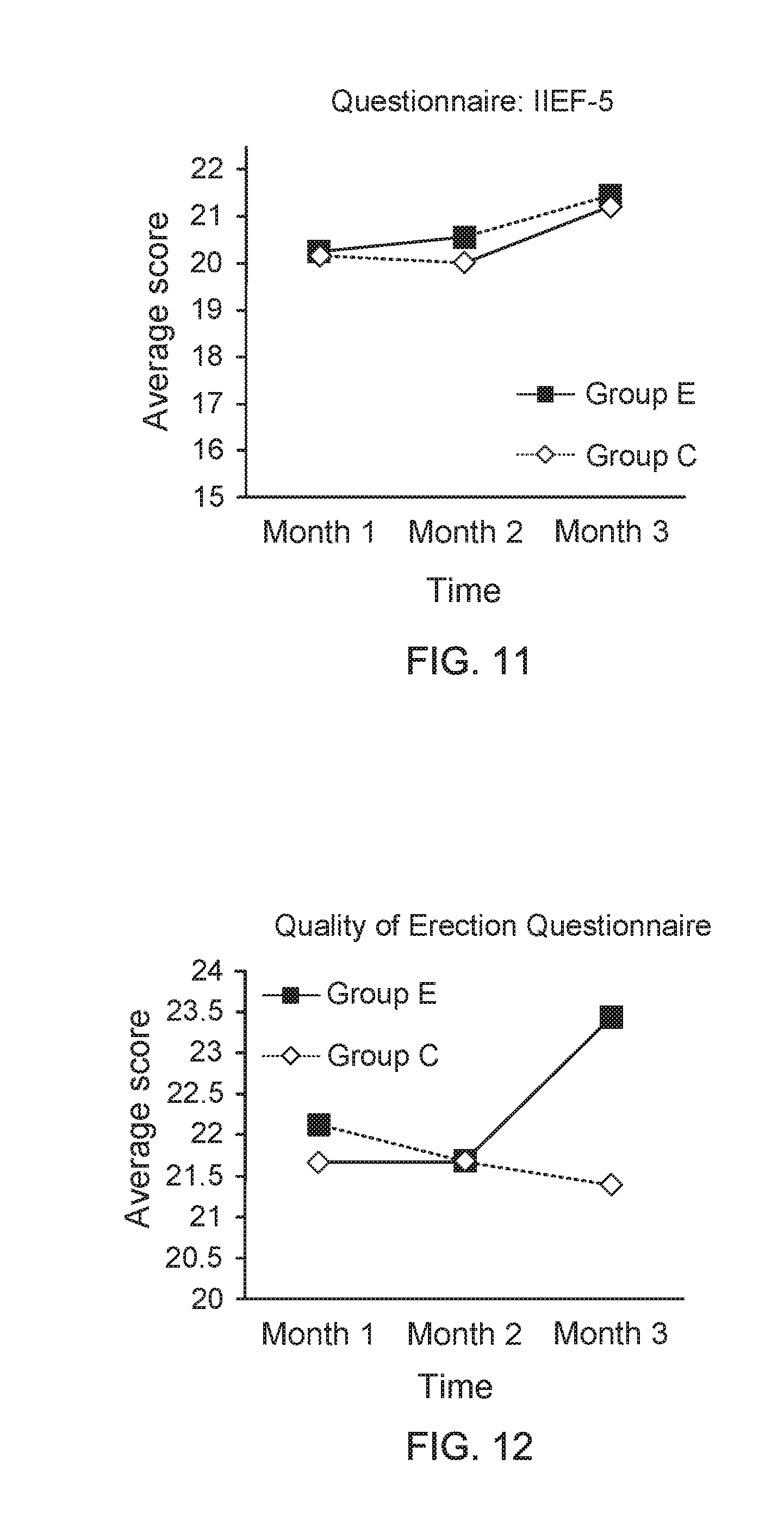
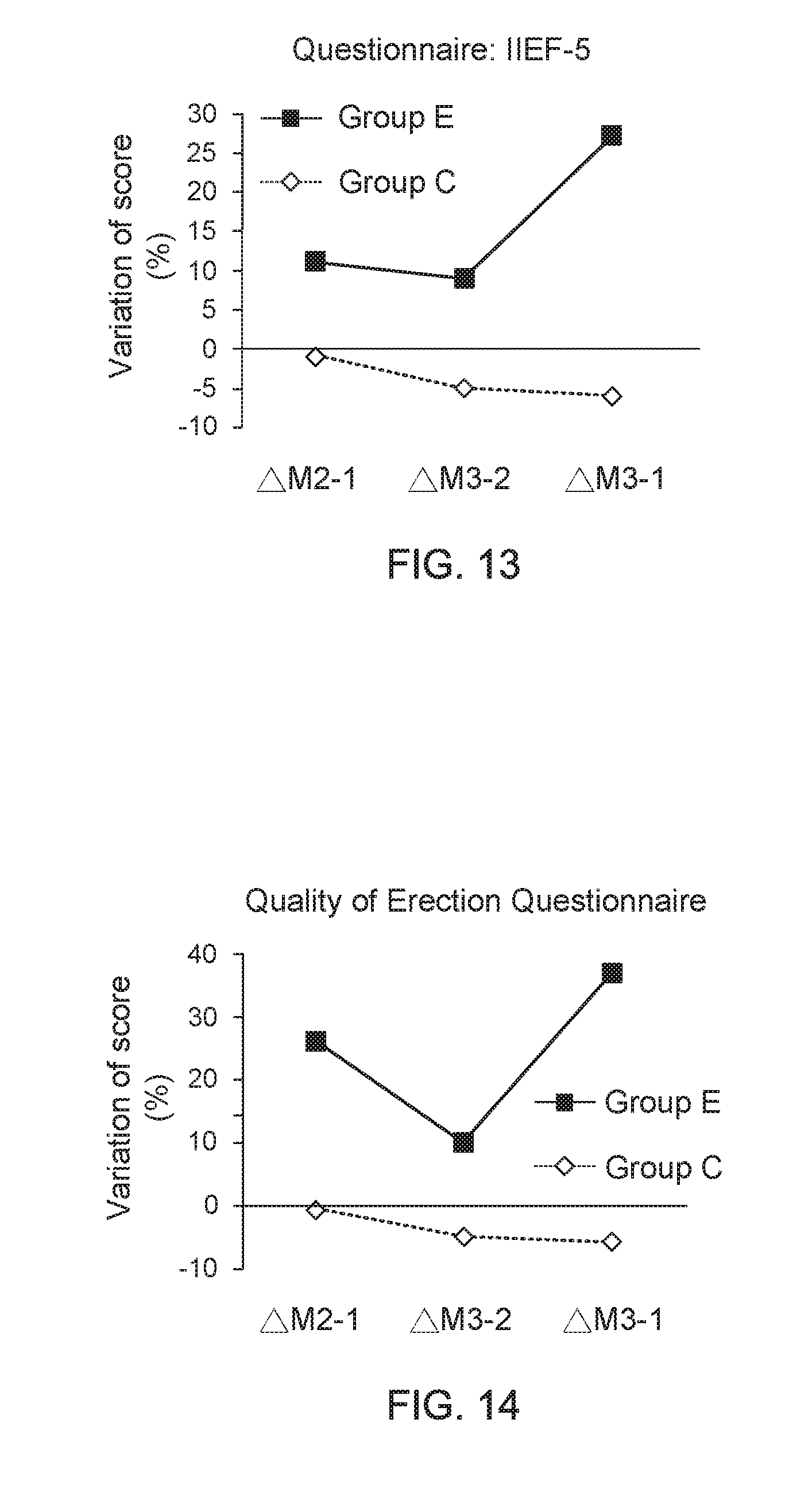
[0031] FIG. 11 shows a data graph of time of clinical experiment versus average score of questionnaire;
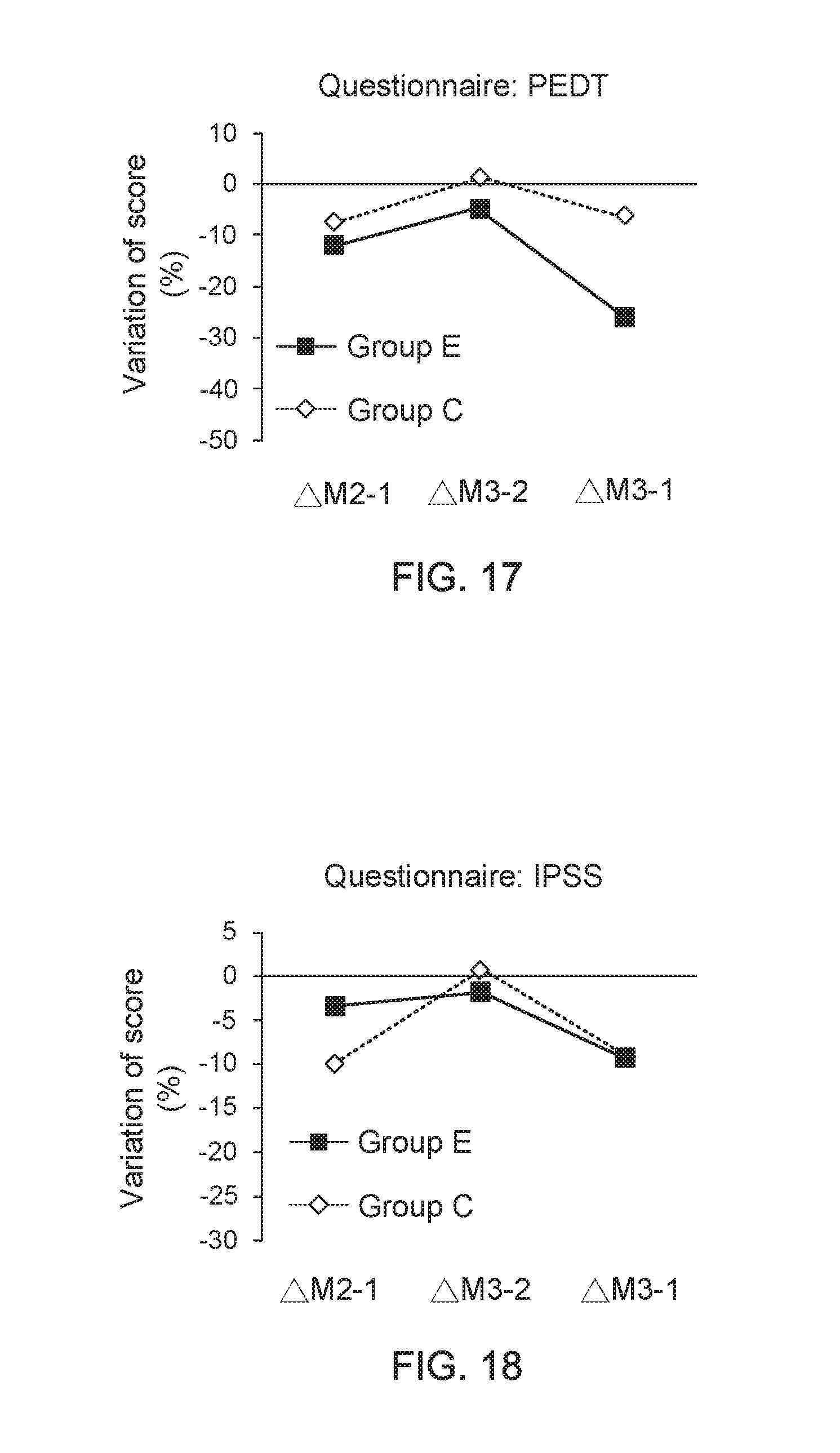
[0032] FIG. 12 shows a data graph of time of clinical experiment versus average score of questionnaire;
[0033] FIG. 13 shows a data graph for describing questionnaire score variation between adjacent two months;
[0034] FIG. 14 shows a data graph for describing questionnaire score variation between adjacent two months;
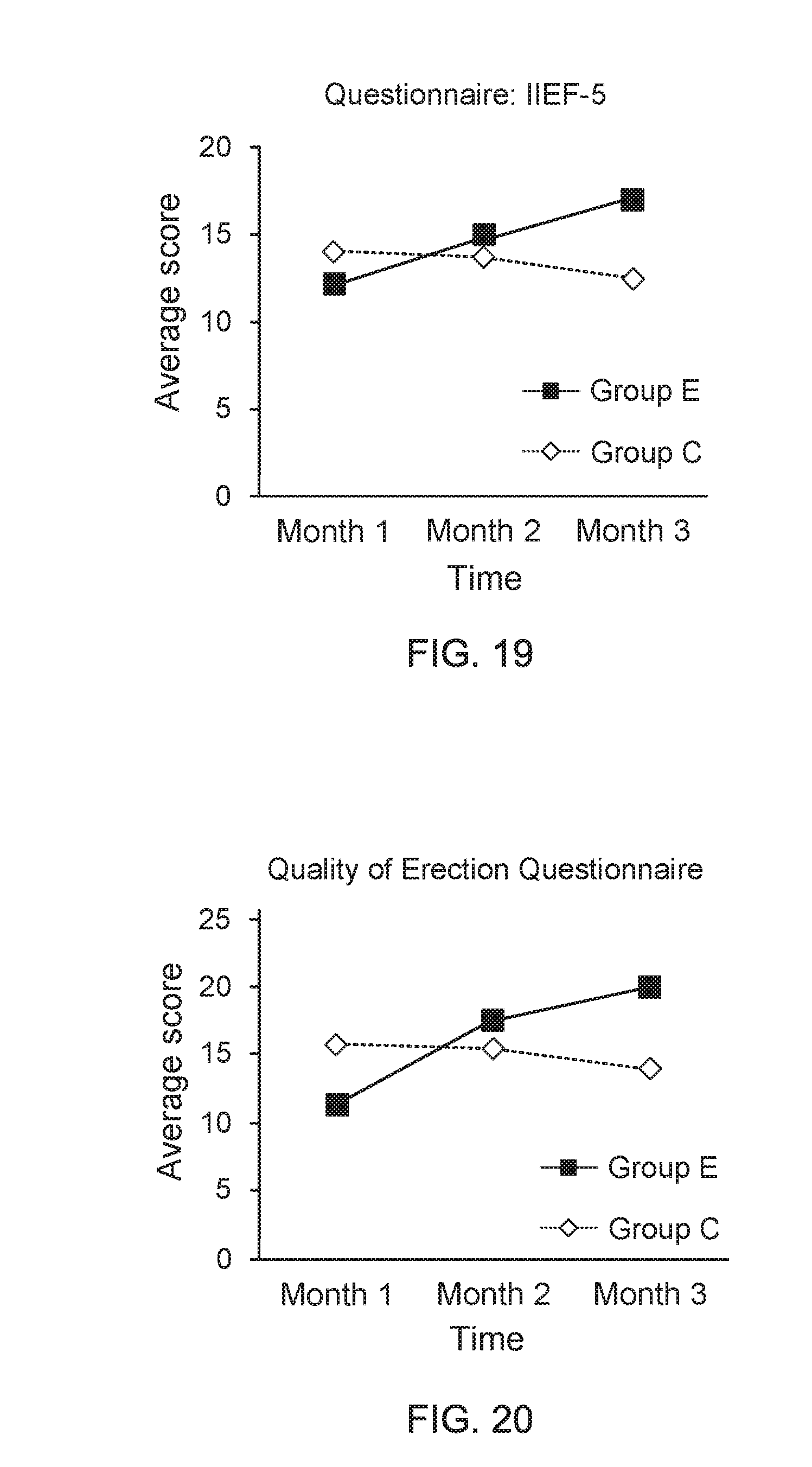
[0035] FIG. 15 shows a data graph of time of clinical experiment versus average score of questionnaire;
[0036] FIG. 16 shows a data graph of time of clinical experiment versus average score of questionnaire;
[0037] FIG. 17 shows a data graph for describing questionnaire score variation between adjacent two months;
[0038] FIG. 18 shows a data graph for describing questionnaire score variation between adjacent two months;
[0039] FIG. 19 shows a data graph of time of clinical experiment versus average score of questionnaire;
[0040] FIG. 20 shows a data graph of time of clinical experiment versus average score of questionnaire;
[0041] FIG. 21 shows a data graph for describing questionnaire score variation between adjacent two months;
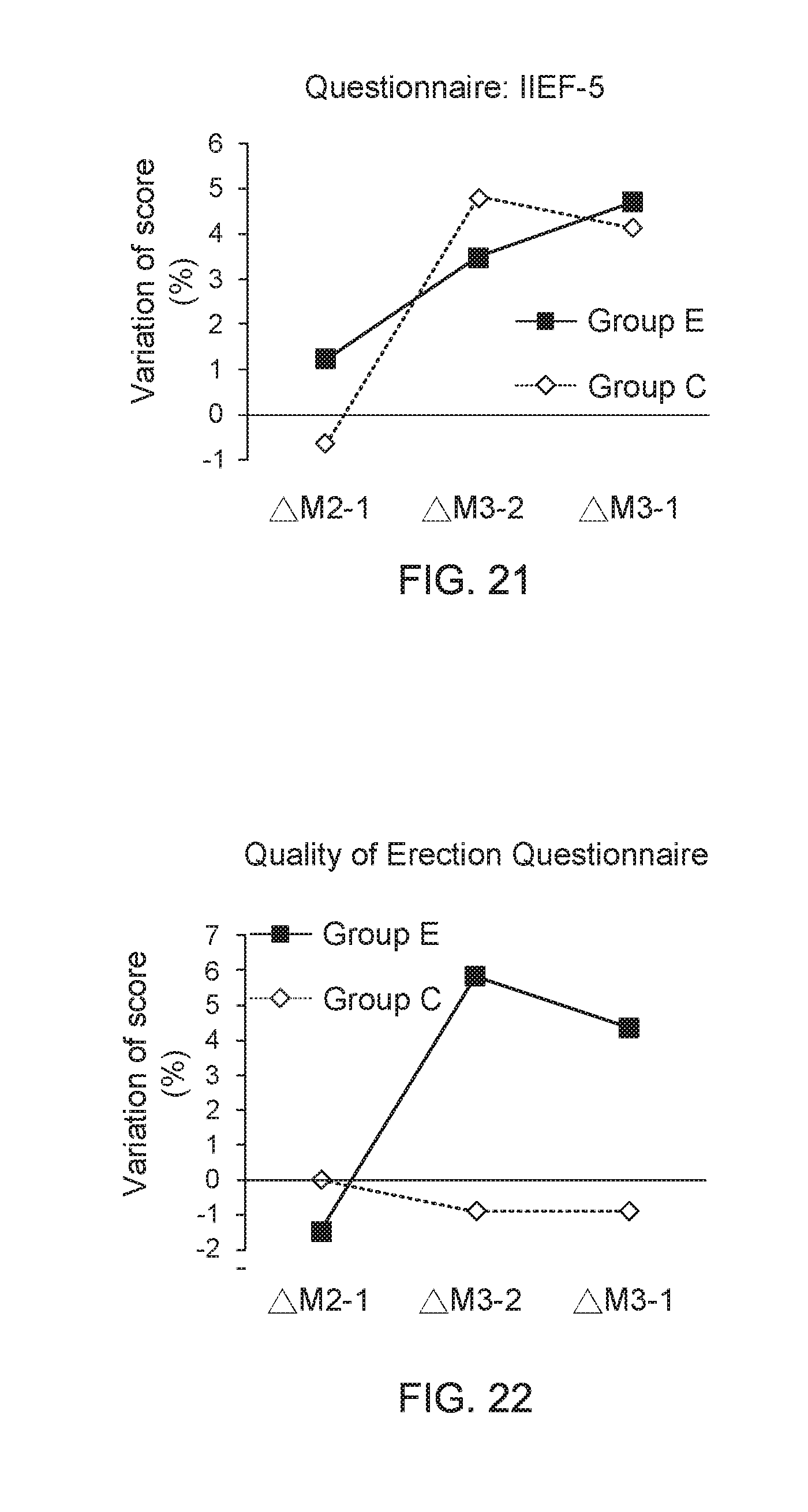
[0042] FIG. 22 shows a data graph for describing questionnaire score variation between adjacent two months;
[0043] FIG. 23 shows a data graph of time of clinical experiment versus average score of questionnaire;
[0044] FIG. 24 shows a data graph of time of clinical experiment versus average score of questionnaire;
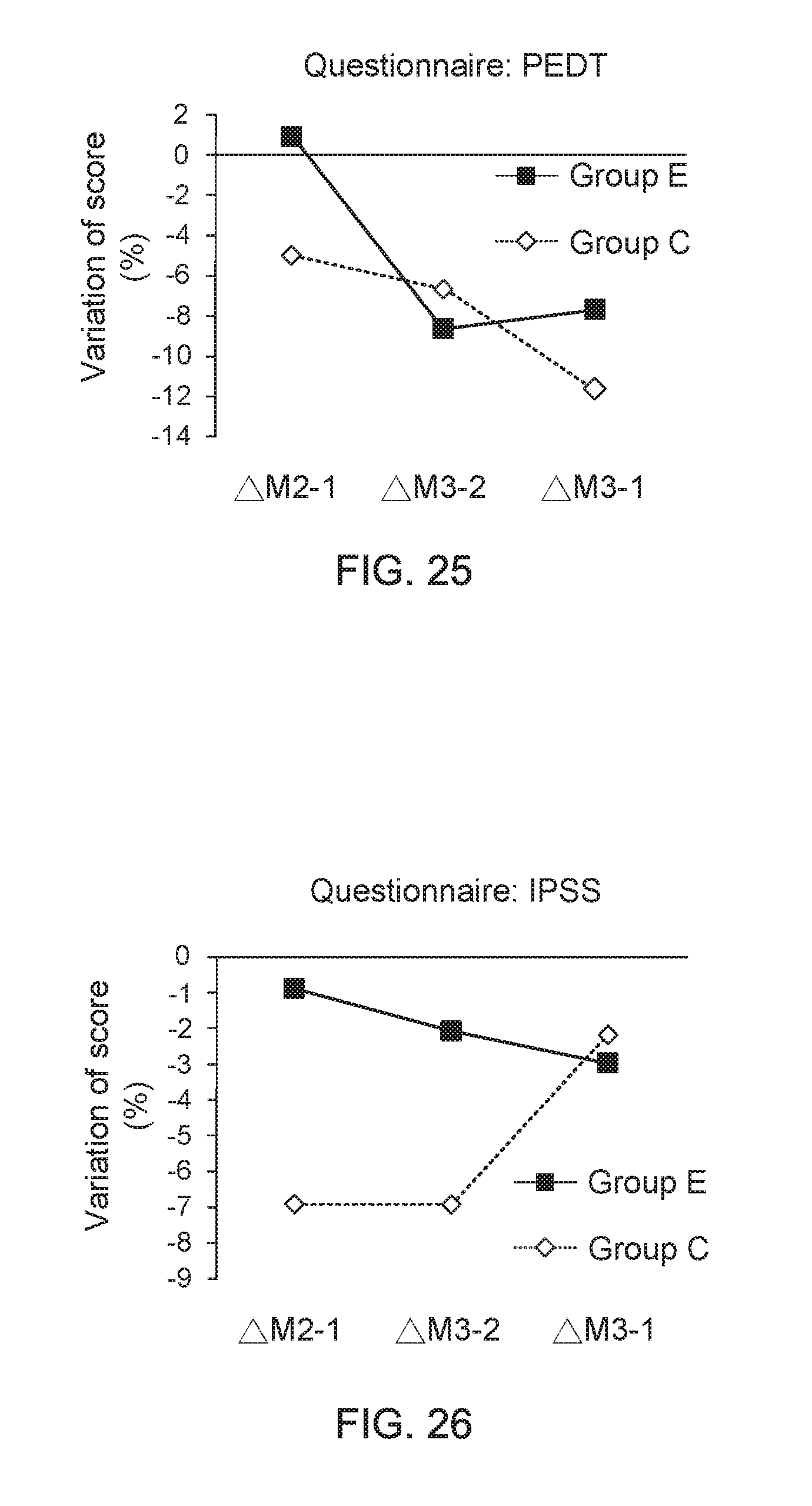
[0045] FIG. 25 shows a data graph for describing questionnaire score variation between adjacent two months; and
[0046] FIG. 26 shows a data graph for describing questionnaire score variation between adjacent two months.
DETAILED DESCRIPTION OF THE PREFERRED EMBODIMENTS
[0047] To more clearly describe a method for treating male sexual dysfunction by using underpants comprising far-infrared fibers according to the present invention, embodiments of the present invention will be described in detail with reference to the attached drawings hereinafter.
First Embodiment
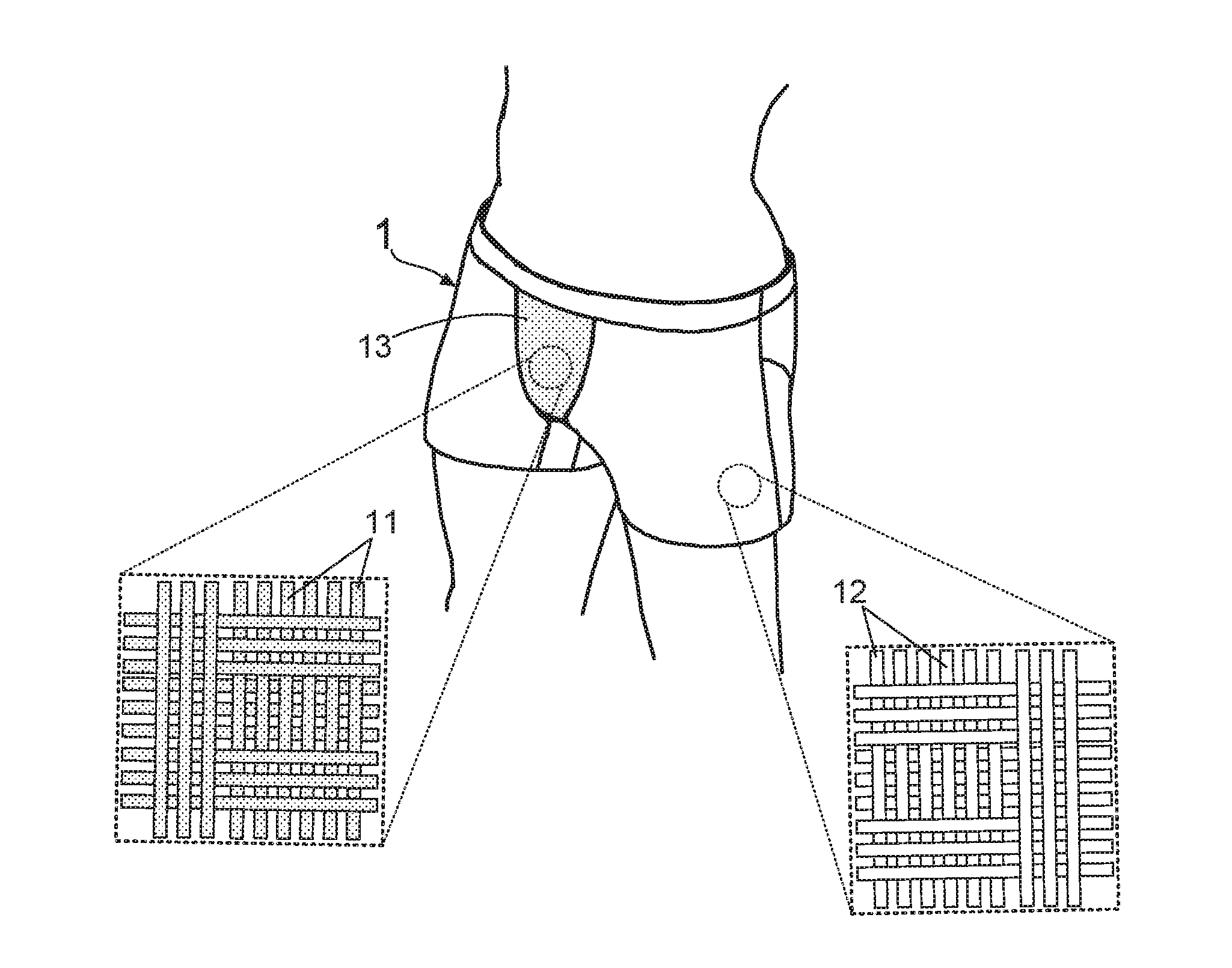
[0048] The inventors of the present invention find that the underpants comprising far-infrared fibers has a particular function to improve male sexual dysfunction. Please refer to FIG. 2A and FIG. 2B, where stereo diagrams of a first embodiment of an underpants capable of improving male sexual dysfunction are provided. According to FIG. 2A and FIG. 2B, it is understood that the underpants 1 capable of improving male sexual dysfunction is weaved from a plurality of far-infrared fibers 11 and a plurality of fibers 12, wherein the far-infrared fibers 11 has a fiber amount occupying 20-50 percent of a total fiber amount of the underpants 1. For instance, FIG. 2A and FIG. 2B indicate that the front rise portion 13 and the crotch portion 14 of the underpants 1 is constituted by the far-infrared fibers 11, and other portions of the underpants 1 is weaved from the fibers 12.
[0049] FIG. 3 shows a cross-sectional view of one far-infrared fiber. In the present invention, the far-infrared fiber 11 comprises a polymer sheath 111 and far-infrared powders 112 doped in or enclosed by the polymer sheath 111. Moreover, the manufacturing material of the polymer sheath 111 is selected from the group consisting of polyester (PET), polyurethane (PU), Poly(vinyl chloride) (PVC), poly propylene (PP), and polyamide (PA). It is worth explaining that, vinyl contains phthalate chemicals or similar plasticizers that are used to turn hard polyvinyl chloride (PVC) into soft and pliable vinyl. Related research reports that phthalates are animal carcinogens and can cause fetal death, malformations, and reproductive toxicity in laboratory animals. Based on the reasons, the polymer sheath 11 can also be made of a polymer compound comprising silica of at least 50 wt %, wherein the chemical structure of the polymer compound is presented by following chemical formula 1.
##STR00001##
[0050] In chemical formula 1, n is an integer in a range from 50 to 100. On the other hand, the far-infrared powders comprises powdered titanium (Ti) with a first metal weight percent of 11-18 wt %, powdered germanium (Ge) with a second metal weight percent of 0.1-0.2 wt % and powdered zinc (Zn) with a third metal weight percent of 0.05-0.15 wt %. By such arrangement, when an adult man wear this underpants 1, the far-infrared ray emitted from the front rise portion 13 and the crotch portion 14 of the underpants 1 would make the dilation of penile arteries of the adult man, and simultaneously urge blood to flow into the penile arteries so as to make the erection of the adult man's penis.
[0051] It needs further explain that, the far-infrared powders 112 further comprises powdered oxide and powdered carbide for increasing the emissivity of the far-infrared ray of the far-infrared fibers 11, wherein the powdered oxide is selected from the group consisting of: Al.sub.2O.sub.3, MgO, NiO.sub.2, SiO.sub.2, ZrO.sub.2, and a mixture made by any two or more aforesaid materials. Moreover, the powdered carbide is selected from the group consisting of: TaC, ZrC, SiC, and a mixture made by any two or more aforesaid materials. By such arrangement, the front rise portion 13 and the crotch portion 14 of the underpants 1 are able to emit the far-infrared ray having an emissivity greater than 90% and a wavelength of 2-22 .mu.m. In addition, other powdered metal such as powdered copper (Cu), powdered silver (Ag), powdered aluminum (Al), or powdered magnesium (Mg) can also be mixed into the far-infrared powders 112.
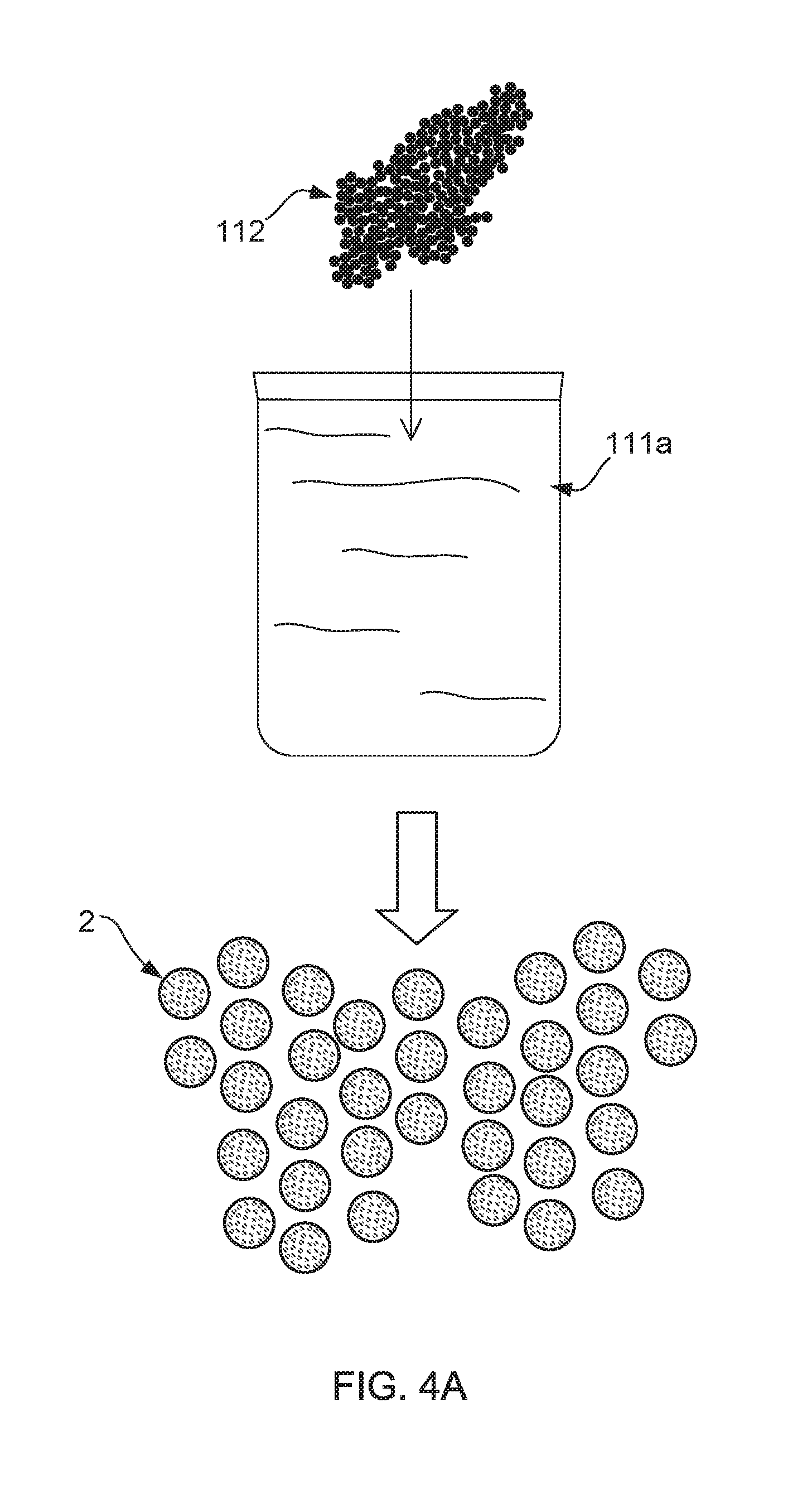
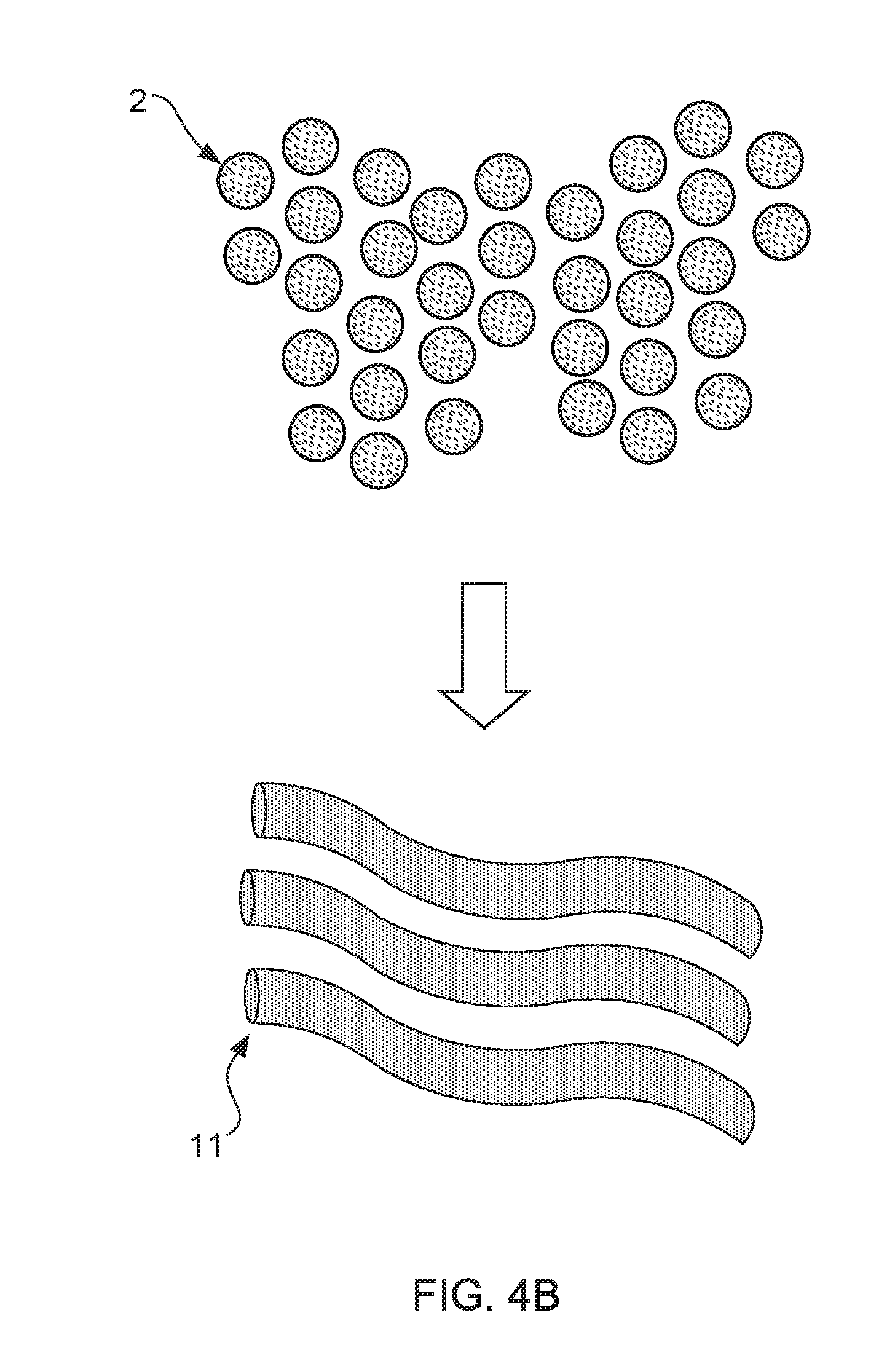
[0052] FIG. 4A, FIG. 4B and FIG. 4C show a schematic production processing flow of the underpants capable of improving male sexual dysfunction. The underpants 1 capable of improving male sexual dysfunction is manufactured through following process flow: [0053] step (1): as FIG. 4A shows, to add far-infrared powders 112 into a polymer solution 111a, and then convert the polymer solution 111a to a plurality of masterbatches 2 through polymerization and granulation process; [0054] step (2): as FIG. 4B shows, executing a spinning process for drawing the masterbatches 2 to a plurality of far-infrared fibers 11; and [0055] step (3): as FIG. 4C shows, the underpants 1 capable of improving male sexual dysfunction is weaved from the far-infrared fibers 11 and a plurality of fibers 12.
Second Embodiment
[0056] It needs to emphasize that, in spite of the fact that the fiber content of the far-infrared fibers 11 of the underpants 1 is in a range from 20% to 50%, that does not used for limiting the constitution of the underpants 1. In the first embodiment, what is weaved from the far-infrared fibers 11 are only the front rise portion 13 and the crotch portion 14 of the underpants 1, with the purpose of cost saving. Please refer to FIG. 5, which illustrates a stereo diagram of a second embodiment of the underpants capable of improving male sexual dysfunction. After comparing FIG. 2B with FIG. 5, it is found that the underpants 1 shown in FIG. 5 is a blend fabric of the far-infrared fibers 11 and the fibers 12, wherein the far-infrared fibers 11 has a maximum fiber amount occupying 50 percent of a total fiber amount of the underpants 1. However, for business benefit consideration, the far-infrared fibers 11 content of the underpants 1 must be limited to lower than 50% because the cost of the far-infrared fibers 11 is higher than the general fibers' 12.
Third Embodiment
[0057] Of course, the far-infrared fibers 11 content of the underpants 1 may be greater than 50% without the consideration of business benefit. FIG. 6 shows a stereo diagram of a third embodiment of the underpants capable of improving male sexual dysfunction. Compared FIG. 5 with FIG. 6, it is understood that the underpants 1 shown in FIG. 6 is weaved merely from the far-infrared fibers 11, such that the far-infrared fibers 11 content of the underpants 1 is 100%.
[0058] First Clinical Trials
[0059] A clinical trial is, essentially, any form of planned experiment that involves patients and is designed to elucidate the most effective treatment for patients with the underpants 1 weaved from the far-infrared fibers 11 and the fibers 12. More particularly, first clinical trials are typically utilized to evaluate the effectiveness of the underpants 1 on improving sexual dysfunction of adult men. According to following Table (1), adult men diagnosed with sexual dysfunction are chosen as test subjects, and further divided into control group (abbreviated to group C) and experimental group (abbreviated to group E) for completing 3-month clinical trials. It needs to further explain that, both the adult men of the group C and the group E wear the underpants 1 provided by the present invention. Moreover, the primary difference between the group C and the group E is that the adult men of the group C has been informed that the underpants they wore is a commercial underpants. On the other hand, in Table (1), physiology examination reports to be "abnormal" means the adult men suffer from hypertension.
TABLE-US-00001 TABLE (1) Physiology Age BMI examination Standard Over Normal Abnormal Average .+-. deviation Normal weight Obese Group C 3 1 49 .+-. 8.18 2 2 0 (n = 4) Group E 4 1 47 .+-. 6.45 2 3 0 (n = 5)
[0060] What is needed to emphasize is that the adult men are chosen as the test subjects after passing the following exclusion criteria: [0061] (1) ever using sex hormone agents or PDE-5 inhibitors like Sildenafil, Vardenafil, or Tadalafil administered in the 7 days previously for the treatment of male sexual dysfunction; [0062] (2) a long user of antihypertensive drugs, antidepressants, sedatives, anti-androgens, or medicines for treating peptic ulcer; [0063] (3) ever being subject to serious injuries of central system, for example, stroke or spinal cord injury; [0064] (4) ever suffering from erectile disorder caused by non-vascular diseases factors, such as nervous factors or hormone factors; [0065] (5) being an HIV patient or the patient with liver disease; [0066] (6) diagnosis of urethral stricture, urinary tract infection, prostatitis, prostate cancer, or bladder cancer; [0067] (7) sexual partner is pregnant or in a breast-feeding period; [0068] (8) being a patient with Peyronie's disease; [0069] (9) suffering from alcoholism or tobacco addiction; [0070] (10) suffering from cancer of prostate gland or other malignant neoplasms; [0071] (11) will have an operation (surgical treatment) during clinical trials period; [0072] (12) having an unhealthy mental status which will affect evaluations of clinical trials; [0073] (13) ever completing at least one sexual performance test which will affect evaluations of clinical trials; and [0074] (14) being ineligibility for the current study judged by investigators.
[0075] During 3-month clinical trials, the test subjects are requested to come back for follow up at the hospital every 1 month for completing the filling of four questionnaires of IIEF-5, QEQ, PEDT, and IPSS. Moreover, physiological data of the test subjects are also measured and recorded by nursing personnel. Diagrams of IIEF-5 questionnaire, QEQ questionnaire, PEDT questionnaire, and IPSS questionnaire are displayed in FIG. 7, FIG. 8, FIG. 9, and FIG. 10, respectively.
[0076] Moreover, FIG. 11 shows a data graph of time of clinical experiment versus average score of questionnaire, and FIG. 12 shows a data graph of time of clinical experiment versus average score of questionnaire. From the data of clinical trials provided in FIG. 11 and FIG. 12, it is found that, compared to the adult men wearing the underpants provided by the present invention for one month, the adult men wearing the underpants provided by the present invention for three months have an apparent progress on improvements of their erectile ability and erectile quality. On the other hand, FIG. 13 shows a data graph for describing questionnaire score variation between adjacent two months and FIG. 14 shows a data graph for describing questionnaire score variation between adjacent two months. From the data of clinical trials provided in FIG. 13 and FIG. 14, it is further understood that, the adult men wearing the underpants provided by the present invention for three months have an apparent progress on improvements of their erectile ability and erectile quality, compared to the adult men wearing the underpants provided by the present invention for one month.
[0077] In addition, FIG. 15 shows a data graph of time of clinical experiment versus average score of questionnaire, and FIG. 16 shows a data graph of time of clinical experiment versus average score of questionnaire. From the data of clinical trials provided in FIG. 15 and FIG. 16, it is found that, compared to the adult men wearing the underpants provided by the present invention for one month, the adult men wearing the underpants provided by the present invention for three months have an apparent progress on improvements of their ejaculation control ability and lower urinary tract symptoms (LUTS). On the other hand, FIG. 17 shows a data graph for describing questionnaire score variation between adjacent two months and FIG. 18 shows a data graph for describing questionnaire score variation between adjacent two months. From the data of clinical trials provided in FIG. 17 and FIG. 18, it is further understood that, the adult men wearing the underpants provided by the present invention for three months have an apparent progress on improvements of their ejaculation control ability and lower urinary tract symptoms (LUTS), compared to the adult men wearing the underpants provided by the present invention for one month.
[0078] Second Clinical Trials
[0079] Second clinical trials are planned for verifying the accuracy of the data corrected from the first clinical trials. According to following Table (2), adult men not sure to suffer from sexual dysfunction are chosen as test subjects, and further divided into control group (abbreviated to group C) and experimental group (abbreviated to group E) for completing 3-month clinical trials. It needs to further explain that, both the adult men of the group C and the group E wear the underpants 1 provided by the present invention. Moreover, the primary difference between the group C and the group E is that the adult men of the group C has been informed that the underpants they wore is a commercial underpants. On the other hand, in Table (2), physiology examination reports to be "abnormal" means the adult men suffer from hypertension.
TABLE-US-00002 TABLE (2) Physiology Age BMI examination Standard Over Normal Abnormal Avg. .+-. deviation Normal weight Obese Group C 4 1 46.66 .+-. 5.36 2 3 0 (n = 5) Group E 13 1 48.93 .+-. 7.45 2 8 4 (n = 14)
[0080] During 3-month clinical trials, the test subjects are requested to come back for follow up at the hospital every 1 month for completing the filling of four questionnaires of IIEF-5, QEQ, PEDT, and IPSS. Moreover, physiological data of the test subjects are also measured and recorded by nursing personnel. Diagrams of IIEF-5 questionnaire, QEQ questionnaire, PEDT questionnaire, and IPSS questionnaire are displayed in FIG. 19, FIG. 20, FIG. 21, and FIG. 22, respectively. FIG. 19 shows a data graph of time of clinical experiment versus average score of questionnaire, and FIG. 20 shows a data graph of time of clinical experiment versus average score of questionnaire. From the data of clinical trials provided in FIG. 19 and FIG. 20, it is found that, compared to the adult men wearing the underpants provided by the present invention for one month, the adult men wearing the underpants provided by the present invention for three months have an apparent progress on improvements of their erectile ability and erectile quality. On the other hand, FIG. 21 shows a data graph for describing questionnaire score variation between adjacent two months and FIG. 22 shows a data graph for describing questionnaire score variation between adjacent two months. From the data of clinical trials provided in FIG. 21 and FIG. 22, it is further understood that, the adult men wearing the underpants provided by the present invention for three months have an apparent progress on improvements of their erectile ability and erectile quality, compared to the adult men wearing the underpants provided by the present invention for one month.
[0081] In addition, FIG. 23 shows a data graph of time of clinical experiment versus average score of questionnaire, and FIG. 24 shows a data graph of time of clinical experiment versus average score of questionnaire. From the data of clinical trials provided in FIG. 23 and FIG. 24, it is found that, compared to the adult men wearing the underpants provided by the present invention for one month, the adult men wearing the underpants provided by the present invention for three months have an apparent progress on improvements of their ejaculation control ability and lower urinary tract symptoms (LUTS). On the other hand, FIG. 25 shows a data graph for describing questionnaire score variation between adjacent two months and FIG. 26 shows a data graph for describing questionnaire score variation between adjacent two months. From the data of clinical trials provided in FIG. 25 and FIG. 26, it is further understood that, the adult men wearing the underpants provided by the present invention for three months have an apparent progress on improvements of their ejaculation control ability and lower urinary tract symptoms (LUTS), compared to the adult men wearing the underpants provided by the present invention for one month.
[0082] Furthermore, data of FIG. 11-FIG. 26 are integrated in following Table (3) and Table (4).
TABLE-US-00003 TABLE (3) First month Third month Avg. Avg. Improvement status Group C of Standard of Standard P Questionnaire score .+-. deviation score .+-. deviation unimproved unimproved value IIEF-5 17.7 .+-. 8.53 17.33 .+-. 8.51 0.92 QEQ 19.30 .+-. 8.46 18.11 .+-. 8.34 0.76 PEDT 6.3 .+-. 5.21 4.44 .+-. 3.71 0.38 IPSS 8.00 .+-. 10.72 6.00 .+-. 8.00 0.64
TABLE-US-00004 TABLE (4) First month Third month Avg. Avg. Improvement status Group C of Standard of Standard P Questionnaire score .+-. deviation score .+-. deviation unimproved unimproved value IIEF-5 18.33 .+-. 6.54 20 .+-. 5.69 0.33 26 QEQ 19.57 .+-. 7.53 22.53 .+-. 8.02 0.24 PEDT 5.14 .+-. 4.22 3.16 .+-. 3.76 0.12 IPSS 3.86 .+-. 6.40 2.58 .+-. 4.06 0.45
[0083] Therefore, data of clinical trials have proved that, after wearing the underpants provided by the present invention for three months, both the men diagnosed with sexual dysfunction and the men not sure to suffer from sexual dysfunction have an apparent progress on improvements of their erectile ability, erectile quality, ejaculation control ability, and lower urinary tract symptoms (LUTS).
[0084] It is worth explaining that, the far-infrared fibers 11 contained by the underpants 1 (as FIG. 5 shows) would emit electromagnetic radiation in the far infrared region (wavelength 4-14 microns). Such far-infrared rays have been examined for effects on human leukocyte activity and on lipid peroxidation of unsaturated fatty acids. Moreover, related research also report that far-infrared rays significantly increase intracellular calcium ion concentration, phagocytosis, and generation of reactive oxygen species in neutrophils, and the blastogenetic response of lymphocytes to mitogens. Despite the increase in reactive oxygen species generated by neutrophils, lipid peroxidation from unsaturated fatty acid may be markedly inhibited by far-infrared rays, related research suggest that the far-infrared rays can be widely used for cosmetic, therapeutic, and preservative purposes, appear capable of potentiating leukocyte functions without promoting oxidative injury. On the other hand, far-infrared ray (FIR) radiation has been proved to be beneficial to human health. Particularly, related research have investigated the effect carried out by FIR on the expression of calmodulin (Cam) protein and nitric oxide (NO) production in human body. Experimental data indicated a significant increase in Cam protein in FIR-treated RAW 264.7 macrophages with or without the addition of lipopolysaccharide (LPS). In addition, the amount of NO was slightly higher but increased significantly in FIR plus LPS-treated RAW 264.7 macrophages.
[0085] It is worth explaining that, the physiologic mechanism of erection of the penis involves release of nitric oxide (NO) in the corpus cavernosum during sexual stimulation. Related research indicates that NO would activate the enzyme guanylate cyclase, which results in increased levels of cyclic guanosine monophosphate (cGMP), so as to produce smooth muscle relaxation in the corpus cavernosum and allowing inflow of blood. NO in vascular regulation couples endothelial and smooth muscle cells. In blood vessels, vascular dilation is initiated by acetylcholine acting at muscarinic receptors on endothelial cells. This initiates IP3 production, Ca.sup.2+ release from endoplasmic reticulum and activation of NO synthase by Ca.sup.2+/calmodulin. Nitric oxide diffuses to smooth muscle cells and activates guanylyl cyclase. cGMP activates a cGMP-dependent protein kinase which phosphorylates myosin light chain and causes vascular relaxation.
[0086] It is repeated that, when an adult man wear this underpants 1 (as shown in FIG. 5), the far-infrared rays emitted from the front rise portion 13 and the crotch portion 14 of the underpants 1 would make the dilation of penile arteries of the adult man, and simultaneously urge blood to flow into the penile arteries so as to make the erection of the adult man's penis. Accordingly, physiological data of the test subjects measured by nursing personnel are integrated in following Table (5), Table (6) and Table (7). Therefore, the recorded physiological data have proved that, despite the fact that the adult men wear the underpants provided by the present invention for three months, there is no any adverse effect induced by the underpants so as to cause the physiological condition of the men be abnormal.
TABLE-US-00005 TABLE 5 First month Standard physiological data Average .+-. deviation Maximum Minimum SBP (mmHg) 126.64 .+-. 9.92 147.00 108.00 DBP (mmHg) 80.86 .+-. 9.13 98.00 64.00 Body Temp. (.degree. C.) 36.37 .+-. 0.56 37.2 35.00 Pulse (bit/min) 73.29 .+-. 7.25 93.00 64.00
TABLE-US-00006 TABLE 6 Second month Standard physiological data Average .+-. deviation Maximum Minimum SBP (mmHg) 123.33 .+-. 8.50 140.00 110.00 DBP (mmHg) 83.44 .+-. 6.64 92.00 71.00 Body Temp. (.degree. C.) 36.64 .+-. 0.29 37.00 36.20 Pulse (bit/min) 74.44 .+-. 8.23 96.00 64.00
TABLE-US-00007 TABLE 7 Third month Standard physiological data Average .+-. deviation Maximum Minimum SBP (mmHg) 126.00 .+-. 8.29 135.00 115.00 DBP (mmHg) 84.00 .+-. 7.79 93.00 74.00 Body Temp. (.degree. C.) 36.20 .+-. 0.28 36.60 36.00 Pulse (bit/min) 73.67 .+-. 12.12 90.00 61.00
[0087] Therefore, through above descriptions, the method for treating male sexual dysfunction by using underpants comprising far-infrared fibers proposed by the present invention have been introduced completely and clearly; in summary, the present invention includes the advantages of:
[0088] (1) Differing from conventional approaches for improving sexual dysfunction of an adult man being commonly carried out by letting the adult man administer a dosage of synthetic drug or a composition of Chinese herbal medicines, the present invention particularly discloses a method for treating male sexual dysfunction by using underpants comprising far-infrared fibers. A variety of clinical data have proved that, after wearing this underpants for three months, both the men diagnosed with sexual dysfunction and the men not sure to suffer from sexual dysfunction have an apparent progress on improvements of their erectile ability, erectile quality, ejaculation control ability, and lower urinary tract symptoms (LUTS).
[0089] (2) Moreover, clinical data have also proved that, despite the fact that a male person wears the underpants provided by the present invention for three months, there is no adverse effect induced by the underpants for causing the physiological condition of the men be abnormal.
[0090] The above description is made on embodiments of the present invention. However, the embodiments are not intended to limit scope of the present invention, and all equivalent implementations or alterations within the spirit of the present invention still fall within the scope of the present invention.
* * * * *



D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

D00013

D00014
D00015
D00016

D00017
D00018
D00019
D00020

D00021
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.