Feeding Deterrence In Agricultural Pests Such As Hemiptera, Lepidoptera And Coleoptera
BEDOUKIAN; Robert H.
U.S. patent application number 16/131412 was filed with the patent office on 2019-03-21 for feeding deterrence in agricultural pests such as hemiptera, lepidoptera and coleoptera. The applicant listed for this patent is BEDOUKIAN RESEARCH, INC.. Invention is credited to Robert H. BEDOUKIAN.
| Application Number | 20190082690 16/131412 |
| Document ID | / |
| Family ID | 49514699 |
| Filed Date | 2019-03-21 |











View All Diagrams
| United States Patent Application | 20190082690 |
| Kind Code | A1 |
| BEDOUKIAN; Robert H. | March 21, 2019 |
FEEDING DETERRENCE IN AGRICULTURAL PESTS SUCH AS HEMIPTERA, LEPIDOPTERA AND COLEOPTERA
Abstract
Compounds are used as agents that deter feeding on crops by agricultural pests, such as hemiptera, lepidoptera and coleoptera, including, but not limited to, stink bugs, codling moth larvae and granary weevils. Feeding deterrence is obtained by contact of the insects with at least one of the compounds of the structure (I) wherein ##STR00001## R is --OH, .dbd.O, --OC(O)R.sub.4, --OR.sub.6 or --(OR.sub.6).sub.2, wherein each R.sub.6 is independently an alkyl group containing from 1 to 4 carbon atoms and R.sub.4 is a branched or straight chain, saturated or unsaturated, hydrocarbyl group with zero to two double bonds and from 1 to 15 carbon atoms; X is O or CH.sub.2 with the proviso that when X is O, R can only be .dbd.O; each Z is independently (CH) or (CH.sub.2); y is a numeral selected from 1 and 2; R.sub.1 is H or a branched or straight chain, saturated or unsaturated hydrocarbyl group with zero to two double bonds and from 1 to 15 carbon atoms; R.sub.2 is H or a branched or straight chain, saturated or unsaturated hydrocarbyl group with zero to three double bonds and from 1 to 15 carbon atoms; R.sub.3 is selected from H, a branched or straight chain, saturated or unsaturated hydrocarbyl group with zero to three double bonds and from 1 to 15 carbon atoms, --(CH.sub.2).sub.nOH, --C(O)OR.sub.5, --CH.sub.2C(O)OR.sub.7, --CH.sub.2C(O)R.sub.8, --C(O)NR.sub.9R.sub.10, and --CH.sub.2C(O)NR.sub.11R.sub.12 where each of R.sub.5, R.sub.7, R.sub.8, R.sub.9, R.sub.10, R.sub.11 and R.sub.12 is independently selected from H and a branched or straight chain, saturated or unsaturated hydrocarbyl group with zero to three double bonds and from 1 to 15 carbon atoms and n is an integer of from 1 to 12; the bond between the 2 and 3 positions in the ring structure may be a single or a double bond; and wherein the compounds of structure (I) contain from 9 to 20 total carbon atoms in the compounds.
| Inventors: | BEDOUKIAN; Robert H.; (West Redding, CT) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 49514699 | ||||||||||
| Appl. No.: | 16/131412 | ||||||||||
| Filed: | September 14, 2018 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 14390826 | Oct 6, 2014 | 10111429 | ||
| PCT/US2013/000120 | Apr 30, 2013 | |||
| 16131412 | ||||
| 61687920 | May 2, 2012 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A01N 37/02 20130101; A01N 31/06 20130101; A01N 37/42 20130101; A01N 35/06 20130101; A01N 43/16 20130101; A01N 43/08 20130101; A01N 37/18 20130101; A01N 49/00 20130101; A01N 37/06 20130101; A01N 37/36 20130101 |
| International Class: | A01N 43/16 20060101 A01N043/16; A01N 37/02 20060101 A01N037/02; A01N 37/36 20060101 A01N037/36; A01N 37/42 20060101 A01N037/42; A01N 43/08 20060101 A01N043/08; A01N 31/06 20060101 A01N031/06; A01N 37/18 20060101 A01N037/18; A01N 35/06 20060101 A01N035/06; A01N 49/00 20060101 A01N049/00; A01N 37/06 20060101 A01N037/06 |
Claims
1. A method for deterring the feeding of one or more agricultural pests selected from the group consisting of hemiptera, lepidoptera and coleoptera on food or crops is obtained by: contacting of the pests with an effective amount of a formulation comprising at least one of the compounds of the structure (I) ##STR00013## wherein R is selected from the group consisting of --OH, .dbd.O, --OC(O)R.sub.4, --OR.sub.6, and --(OR.sub.6).sub.2, wherein each R.sub.6 is independently selected from an alkyl group containing from 1 to 4 carbon atoms and R.sub.4 is a branched or straight chain, saturated or unsaturated hydrocarbyl group with zero to two double bonds and from 1 to 15 carbon atoms; X is O or CH.sub.2 with the proviso that when X is O, R can only be .dbd.O; each Z is independently selected from the group consisting of (CH) and (CH.sub.2) y is a numeral selected from 1 and 2; R.sub.1 is selected from the group consisting of H and a branched or straight chain, saturated or unsaturated hydrocarbyl group with zero to two double bonds and from 1 to 15 carbon atoms; R.sub.2 is selected from the group consisting of H and a branched or straight chain, saturated or unsaturated hydrocarbyl group with zero to three double bonds and from 1 to 15 carbon atoms; R.sub.3 is selected from H, a branched or straight chain, saturated or unsaturated hydrocarbyl group with zero to three double bonds and from 1 to 15 carbon atoms, --(CH.sub.2).sub.nOH, --C(O)OR.sub.5, --CH.sub.2C(O)OR.sub.7, --CH.sub.2C(O)R.sub.8, --C(O)NR.sub.9R.sub.10, and --CH.sub.2C(O)NR.sub.11R.sub.12 where each of R.sub.5, R.sub.7, R.sub.8, R.sub.9, R.sub.10, R.sub.11 and R.sub.12 is independently selected from H and a branched or straight chain, saturated or unsaturated hydrocarbyl group with zero to three double bonds and from 1 to 15 carbon atoms, and n is n integer of from 1 to 12; the bond between the 2 and 3 positions in the ring structure may be a single or a double bond, and wherein the compounds of structure (I) contain from 9 to 20 total carbon atoms in the compounds.
2. The method according to claim 1 wherein the agricultural pest is selected from the group consisting of: cicadas, aphids, stink bugs, butterflies, moths and beetles.
3. The method according to claim 1 wherein the compounds of structure (I) have from 10 to 16 carbon atoms in the compound.
4. The method according to claim 1 wherein the at least one compound of structure (I) is a compound wherein: R is selected from the group consisting of --OH and .dbd.O, X is CH.sub.2, y is 1 or 2, each Z is selected from the group consisting of (CH) and (CH.sub.2), the bond between positions 2 and 3 in the ring is a single bond, one of R.sub.1 and R.sub.2 is H or --CH.sub.3 and the other of R.sub.1 and R.sub.2 is a hydrocarbyl group containing from 9 to 15 carbon atoms and 0 to 3 double bonds, and R.sub.3 is H.
5. The method of claim 1 wherein the at least one compound of structure (I) is a compound wherein: R is selected from the group consisting of --OH and .dbd.O, X is CH.sub.2, y is 1 or 2, each Z is selected from (CH) and (CH.sub.2), the bond between positions 2 and 3 in the ring is a single or double bond, one of R.sub.1 and R.sub.2 is H and the other of R.sub.1 and R.sub.2 is a hydrocarbyl group containing from 9 to 15 carbon atoms and 0 to 3 double bonds, and R.sub.3 is selected from the group consisting of --C(O)OR.sub.5 and --CH.sub.2C(O)R.sub.8 where R.sub.5 and R.sub.8 are each selected from a hydrocarbyl group containing from 1 to 6 carbon atoms.
6. The method according to claim 5 wherein R is .dbd.O, y is 1, the bond between positions 2 and 3 in the ring is a single bond, and R.sub.2 and R.sub.5 are each --CH.sub.3.
7. The method according to claim 1 wherein the at least one compound of structure (I) is a compounds wherein: R is .dbd.O, X is O, y is 1 or 2, each Z is selected from the group consisting of (CH) and (CH.sub.2), the bond between positions 2 and 3 of the rings is a single or double bond, one of R.sub.1 and R.sub.2 is H and the other of R.sub.1 and R.sub.2 is a hydrocarbyl group containing group containing from 9 to 15 carbon atoms and 0 to 3 double bonds, and R.sub.3 is selected from the group consisting of --C(O)OR.sub.5 and --CH.sub.2C(O)R.sub.8 where R.sub.5 and R.sub.8 are each selected from a hydrocarbyl group containing from 1 to 6 carbon atoms and wherein the total number of carbon atoms in the compounds of structure (I) is from 11 to 17.
8. The method according to claim 7 wherein the bond between positions 2 and 3 of the rings is a single bond and R.sub.5 and R.sub.7 are each selected from a hydrocarbyl group containing from 3 to 5 carbon atoms.
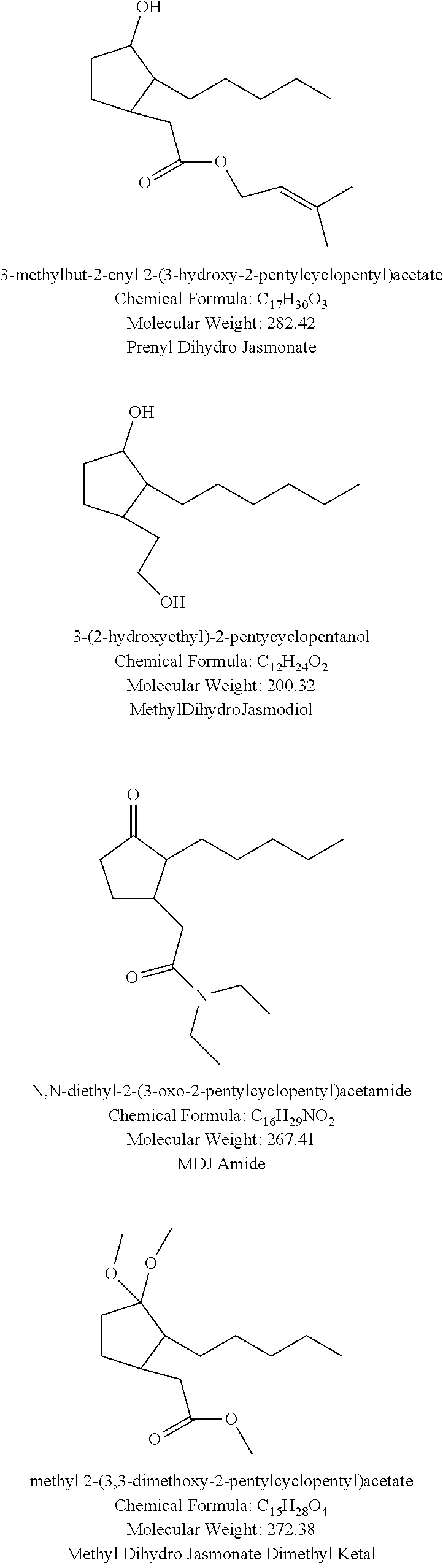
9. The method according to claim 7 wherein the bond between positions 2 and 3 of the rings is a single bond and R.sub.5 and R.sub.7 are each --CH.sub.3.
10. The method according to claim 1 wherein the at least one compound of structure (I) is a compound wherein: R is .dbd.O, X is O, y is 1 or 2, each Z is selected from (CH) and (CH.sub.2), the bond between positions 2 and 3 in the ring is a single bond, R.sub.1 is an alkyl group containing from 5 to 13 carbon atoms, R.sub.2 is selected from the group consisting of H or --CH.sub.3, and R.sub.3 is H.
11. The method according to claim 10 wherein: R.sub.1 is an alkyl group of from 5 to 10 carbon atoms such that the compound of structure (I) contains from 11 to 14 total carbon atoms.
12. The method according to claim I wherein the at least one compound of structure (I) is selected from the group consisting of: ##STR00014## ##STR00015## ##STR00016##
13. The method according to claim 1 wherein the at least one compound of structure (I) is selected from the group consisting of: ##STR00017## ##STR00018##
14. The method according to claim 1 wherein the at least one compound of structure (I) is selected from the group consisting of ##STR00019## ##STR00020##
15. The method according to claim 1 wherein the at east one compound of structure (I) is selected from the group consisting of: ##STR00021## ##STR00022## ##STR00023##
16. The method according to claim 1 wherein the at least one compound of structure (I) is selected from the group consisting of ##STR00024##
17. The method according to claim I wherein the at least one compound of structure (I) is applied to crops, agricultural or ornamental trees, plants, vegetation, produce or packaging materials for plants or crops by way of a formulation selected from the group consisting of direct spray formulations, fogger formulations, microencapsulated formulations, soil treatment formulations, seed treatment formulations, injectable formulations for injection into or onto plants or crops, and formulations for evaporative devices.
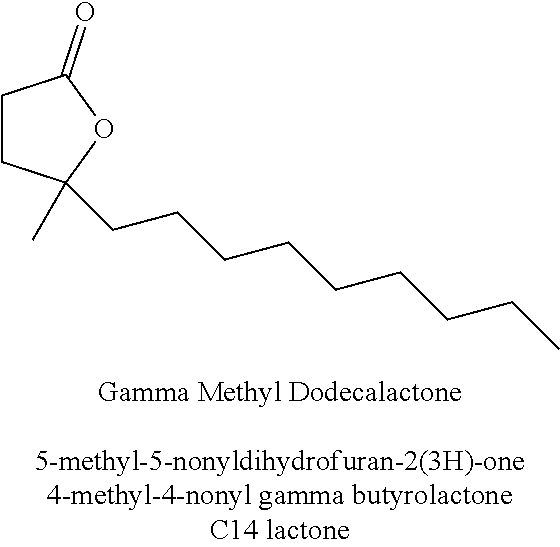
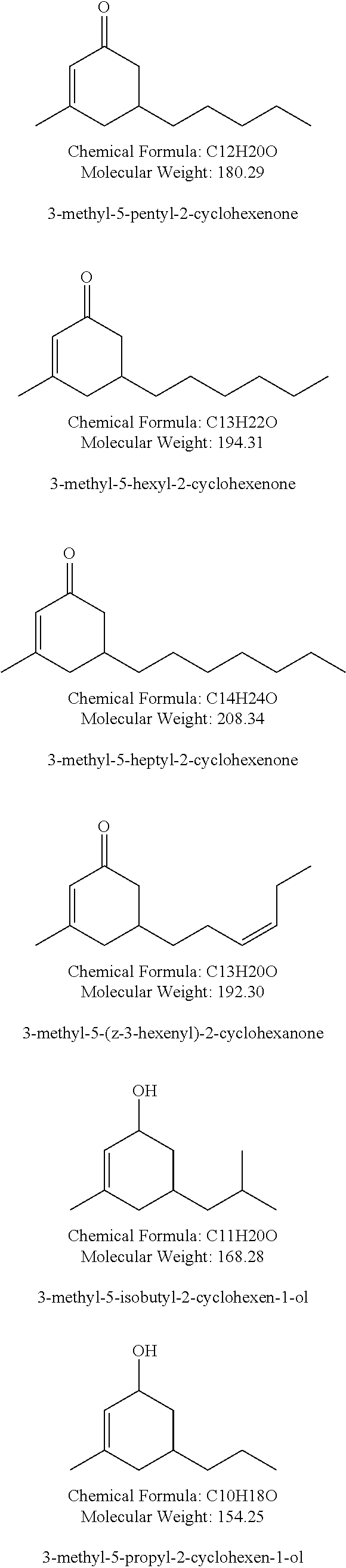
18. The method of claim 1 wherein the at least one compound of structure (I) is selected from the group consisting of : methyl apritone, methyl dihydrojasmonate, propyl dihydrojasmonate, methyl dihydrojasmolate, gamma-dodecalactone, gamma-tridecalactone, gamma methyl dodecalactone, gamma methyl tridecalactone. 3-methyl-5-propyl-2-cyclohexenone, 3-methyl-5-isobutyl-2-cyclohexenone, 3-methyl-5-isobutyl-2-cyclohexenol, 3-methyl-5-pentyl-2-cyclohexenone, 3-methyl-5-hexyl-2-cyclohexenone, and 3-methyl-5-heptyl-2-cyclohexenone.
19. The method according to claim 1 wherein the at least one compound of the structure (I) is present in the formulation in an amount sufficient to provide increased pest repellency or mortality over control pest repellency or mortality of at least 1%.
20. The method of claim 1 wherein the formulation is used to treat food or crops such that the treated food or crops have from about 0.05 to about 250 mg/sq. in. of at least one of the compounds thereon.
Description
CROSS-REFERENCED APPLICATIONS
[0001] This application is a continuation of U.S. patent application Ser. No. 14/390,826, filed Oct. 6, 2014, which claims priority to international application PCT/US13/00120, filed Apr. 30, 2013, and U.S. Application Ser. No. 61/687,920, filed May 2, 2012, all of which are incorporated herein by reference.
BACKGROUND
1. Field of the Disclosure
[0002] This disclosure relates to the use of compounds as agents that deter feeding on crops by agricultural pests such as hemiptera, lepidoptera and coleoptera including, but not limited to, stink bugs, codling moth larvae and granary weevils.
[0003] The active feeding deterrence agents of this disclosure are an effective control agent against agricultural pests such as hemiptera, lepidoptera and coleoptera. Hemiptera includes cicadas, aphids and stink bugs such as the Brown Marmorated Stink Bug. Lepidoptera includes butterflies and moths such as the Codling Moth. Coleoptera are beetles and the order contains granary weevils.
2. Discussion of the Background Art
[0004] Agricultural pests including but not limited to the brown marmorated stink bug, codling moth and granary weevil are known to feed on various crops causing damage to fruits, vegetables and other plant life. The cost of this damage is extensive and leads to hardship among growers.
[0005] In the Mid-Atlantic region, where brown marmorated stink bugs are well established, they caused an estimated $37 million in damage in apple crops alone in 2010, the most recent year for which data are available. [Darryl Fears, "Stink bugs migrating to deep south", Washington Post, Mar. 16, 2012]. Aside from apples, the bug will feed on nearly anything, including cherries, tomatoes, grapes, lima beans, soybeans, green peppers and peaches.
[0006] Codling moth larvae penetrate into apples and pears and tunnel to the core, leaving holes in the fruit that are filled with reddish-brown, crumbly droppings. If left uncontrolled, larvae can cause substantial damage, often infesting 20 to 90% of the fruit, depending on the variety and location. Late maturing varieties are more likely to suffer severe damage than early varieties. [J. L. Caprile and P. M. Vossen, Pest Notes: Codling Moth UC ANR Publication 7412, May 2011].
[0007] In walnuts, codling moth larvae feed on the kernels. Nuts damaged early in the season when the nuts are quite small will drop off trees soon after damage occurs. Nuts damaged later in the season will remain on trees, but their kernels are inedible. Walnuts aren't as favored a host as apples and pears, and untreated trees might incur very little to modest damage (10 to 15% of the nuts), depending on the variety and location. [Pest Notes, May 2011]
[0008] Phosmet, a phthalimide-derived, non-systemic, organophosphate insecticide is the primary means of controlling codling moth damage to apples. This material is on the US Emergency Planning list of extremely hazardous substances and is highly toxic to bees. The materials covered in this patent application would be a much gentler means of deterring the larvae and controlling moth infestation.
[0009] Granary weevils are attracted to and will attack all kinds of grains and grain products. These products will include such materials as wheat, corn, barley, or rice. In stored food products, they may be found in materials like macaroni or spaghetti. Adult weevils will feed on the same foods as the larvae. However, because they do not need to develop in the inside of whole grains like larvae do, they are not limited to just one grain or in other words, they are not restricted in their diets. [http://www.pestmall.com/blog/pest-info/other-pests/granary-weevil-biolog- y]
[0010] Control of weevils is quite difficult as they live and breed in our food sources. Use of pyrethrins is an option, but food shouldn't be sprayed directly. Use of our materials may be sprayed directly on crops, seeds, agricultural or ornamental trees, plants, vegetation, produce or packaging materials so they provide a better alternative to traditional insecticides.
SUMMARY
[0011] In accordance with this disclosure, feeding deterrence of crops by agricultural pests such as hemiptera, lepidoptera and coleoptera is obtained by contact of the insects with at least one of the compounds of the structure (I)
##STR00002##
wherein: [0012] R is selected from --OH, .dbd.O, --OC(O)R.sub.4, --OR.sub.6, and --(OR.sub.6).sub.2, wherein each R.sub.6 is independently selected from an alkyl group containing from 1 to 4 carbon atoms and R.sub.4 is a branched or straight chain, saturated or unsaturated, hydrocarbyl group with zero to two double bonds and from 1 to 15 carbon atoms; [0013] X is O or CH.sub.2 with the proviso that when X is O, R can only be .dbd.O; [0014] each Z is independently selected from (CH) and (CH.sub.2); [0015] y is a numeral selected from 1 and 2; [0016] R.sub.1 is selected from H or a branched or straight chain, saturated or unsaturated hydrocarbyl group with zero to two double bonds and from 1 to 1.5 carbon atoms; [0017] R.sub.2 is selected from H and a branched or straight chain, saturated or unsaturated hydrocarbyl group with zero to three double bonds and from 1 to 15 carbon atoms; [0018] R.sub.3 is selected from H, a branched or straight chain, saturated or unsaturated hydrocarbyl group with zero to three double bonds and from 1 to 15 carbon atoms, --(CH.sub.2).sub.nOH, --C(O)OR.sub.5, --CH.sub.2C(O)OR.sub.7, --CH.sub.2C(O)R.sub.8, --C(O)NR.sub.9R.sub.10, and --CH.sub.2C(O)NR.sub.11R.sub.12 where each of R.sub.5, R.sub.7, R.sub.8, R.sub.9, R.sub.10, R.sub.11 and R.sub.12 is independently selected from H and a branched or straight chain, saturated or unsaturated hydrocarbyl group with zero to three double bonds and from 1 to 15 carbon atoms and n is an integer of from 1 to 12; [0019] the bond between the 2 and 3 positions in the ring structure may be a single or a double bond; and [0020] wherein the compounds of structure (I) contain from 9 to 20 total carbon atoms in the compounds. The disclosure also includes optical isomers, diastereomers and enantiomers of the compounds of structure (I). Thus, at all stereocenters where stereochemistry is not explicitly defined, all possible epimers are envisioned.
[0021] The feeding deterrence compounds of this disclosure, which could be applied directly to crops, seeds, agricultural or ornamental trees, plants, vegetation, produce or packaging materials for crops, have low mammalian toxicity and are similar to naturally occurring materials used in flavor/fragrance applications. Therefore, these provide a better method of control for farmers and home owners. Additionally, the feeding deterrence compounds of this disclosure have the potential to be used on organic crops.
[0022] The compounds of structure (I) may be employed to defer feeding by agricultural pests, such as hemiptera, lepidoptera and coleoptera which include, but are not limited to, stink bugs, codling moth larvae and granary weevils. The active compounds of structure (I) may be employed in any suitable formulation, such as, but not limited to, direct spray formulations, fogger formulations, microencapsulated formulations, soil treatment formulations, seed treatment formulations, injectable formulations for injection into the plant and formulations for use in evaporative devices.
DETAILED DESCRIPTION OF EMBODIMENTS
[0023] Deterrence of feeding by agricultural pests, such as hemiptera, lepidoptera and coleoptera including, but not limited to, stink bugs, codling moth larvae and granary weevils is obtained by contact of the insects with at least one of the compounds of the structure (I)
##STR00003##
wherein [0024] R is selected from --OH, .dbd.O, --OC(O)R.sub.4, --OR.sub.6, and --(OR.sub.6).sub.2, wherein each R.sub.6 is independently selected from an alkyl group containing from 1 to 4 carbon atoms and R.sub.4 is a branched or straight chain, saturated or unsaturated hydrocarbyl group with zero to two double bonds and from 1 to 15 carbon atoms. [0025] X is O or CH.sub.2 with the proviso that when X is O, R can only be .dbd.O; [0026] each Z is independently selected from (CH) and (CH.sub.2); [0027] y is a numeral selected from 1 and 2; [0028] R.sub.1 is selected from H or a branched or straight chain, saturated or unsaturated hydrocarbyl group with zero to two double bonds and from 1 to 15 carbon atoms; [0029] R.sub.2 is selected from H and a branched or straight chain, saturated or unsaturated hydrocarbyl group with zero to three double bonds and from 1 to 15 carbon atoms; [0030] R.sub.3 is selected from H, a branched or straight chain, saturated or unsaturated hydrocarbyl group with zero to three double bonds and from 1 to 15 carbon atoms --(CH.sub.2).sub.nOH, --C(O)OR.sub.5, --CH.sub.2C(O)OR.sub.7, --CH.sub.2C(O)R.sub.8, --C(O)NR.sub.9R.sub.10, and --CH.sub.2C(O)NR.sub.11R.sub.12 where each of R.sub.5, R.sub.7, R.sub.8, R.sub.9, R.sub.10, R.sub.11 and R.sub.12 is independently selected from H and a branched or straight chain, saturated or unsaturated hydrocarbyl group with zero to three double bonds and from Ito 15 carbon atoms, and n is n integer of from 1 to 12; [0031] the bond between the 2 and 3 positions in the ring structure may be a single or a double bond; and [0032] wherein the compounds of structure (I ) contain from 11 to 20 total carbon atoms in the compounds. The disclosure also includes optical isomers, diastereomers and enantiomers of the named structures. Thus, at all stereocenters where stereochemistry is not explicitly defined, all possible epimers are envisioned.
[0033] A preferred group of feeding deterrence compounds are those compounds of structure (I) wherein; [0034] R is selected from --OH and .dbd.O, X is CH.sub.2, y is 1 or 2, each Z is selected from (CH) and (CH.sub.2), the bond between positions 2 and 3 in the ring is a single bond, one of R.sub.1 and R.sub.2 is H or --CH.sub.3 and the other of R.sub.1 and R.sub.2 is a branched or straight chain, saturated or unsaturated hydrocarbyl group containing from 9 to 15 carbon atoms and 0 to 3 double bonds, and R.sub.3 is H.
[0035] Another preferred group of feeding deterrence compounds are those compounds of structure (I) wherein; [0036] R is selected from --OH and .dbd.O, more preferably .dbd.O, X is CH.sub.2, y is 1 or 2, more preferably 1, each Z is selected from (CH) and (CH.sub.2), the bond between positions 2 and 3 in the ring is a single or double bond, more preferably a single bond, one of R.sub.1 and R.sub.2 is H and the other of R.sub.1 and R.sub.2 is a branched or straight chain, saturated or unsaturated hydrocarbyl group containing from 9 to 15 carbon atoms and 0 to 3 double bonds, and R.sub.3 is selected from --C(O)OR.sub.5 and --CH.sub.2C(O)R.sub.8 where R.sub.5 and R.sub.8 are each selected from a straight chain or branched, saturated or unsaturated hydrocarbyl group containing from 1 to 6 carbon atoms, and more preferably 3 to 5 carbon atoms and still more preferably --CH.sub.3.
[0037] Another preferred group of feeding deterrence compounds are those compounds of structure (I) wherein [0038] R is .dbd.O, X is O, y is 1 or 2, each Z is selected from (CH) and (CH.sub.2), the bond between positions 2 and 3 of the rings is a single or double bond, more preferably a single bond, one of R.sub.1 and R.sub.2 is H and the other of R.sub.1 and R.sub.2 is a branched or straight chain, saturated or unsaturated hydrocarbyl group containing from 9 to 15 carbon atoms and 0 to 3 double bonds, and R.sub.3 is selected from --C(O)OR.sub.5 and --CH.sub.2C(O)R.sub.8 where R.sub.5 and R.sub.7 are each selected from a hydrocarbyl group containing from 1 to 6 carbon atoms, and more preferably 3 to 5 carbon atoms and still more preferably --CH.sub.3 and wherein the total number of carbon atoms in the compounds of structure (I) is from 9 to 20, more preferably from 9 to 14 total carbon atoms.
[0039] Another preferred group of feeding deterrence compounds are those compounds of structure (I) wherein R is .dbd.O, X is O, y is 1 or 2, each Z is selected from (CH) and (CH.sub.2), the bond between positions 2 and 3 in the ring is a single bond, R.sub.1 is a branched or straight chain, saturated or unsaturated alkyl group containing from 5 to 13 carbon atoms, R.sub.2 is H or --CH.sub.3, R.sub.3 is H, and more preferably where R.sub.1 is an alkyl group of from 5 to 10 carbon atoms such that the compound of structure (I) contains from 11 to 14 total carbon atoms.
[0040] The active compounds of structure (I) may be employed in any suitable formulation, such as, but not limited to, direct spray formulations, fogger formulations, microencapsulated formulations, soil treatment formulations, seed treatment formulations, injectable formulation for injection into the plant and formulations for use in evaporative devices. The formulations of the active compounds will be such that the food or crops being treated will have from about 0.05 to about 250 mg/sq. in., preferably about 0.25 to about 50 mg/sq. in., of the active compounds thereon.
[0041] Representative examples of compounds of structure (I) include, but are not limited to,
##STR00004## ##STR00005## ##STR00006## ##STR00007## ##STR00008## ##STR00009## ##STR00010## ##STR00011## ##STR00012##
[0042] Preferred compounds of structure (I) include methyl apritone, methyl dihydrojasmonate, propyl dihydrojasmonate, gamma-dodecalactone, gamma-tridecalactone, gamma methyl dodecalactone, gamma methyl tridecalactone, 3-methyl-5-propyl-2-cyclohexenone, 3-methyl-5-isobutyl-2-cyclohexenone, 3-methyl-5-isobutyl-2-cyclohexenol, 3-methyl-5-pentyl-2-cyclohexenone, 3-methyl-5-hexyl-2-cyclohexenone, and 3-methyl-5-heptyl-2-cyclohexenone.
[0043] A test was designed to measure the repellency as feeding deterrence in brown marmorated stink bugs (BMSB). Five replicates of 5 BMSBs were introduced into test arenas, containing one 60 mm.times.15 mm Petri dish with treated filter papers in which a food source (green bean) was attached, at the start of the test.
[0044] The small Petri dishes containing the treated filter paper and food source were covered with a fitted Petri dish lid in between observation times, and were removed 5 minutes before each observation time to prevent stink bugs from feeding until sated. The distribution of the BMSBs was observed and documented hourly for 5 hours.
TABLE-US-00001 TABLE 1 Compound in a 57% Ethanol:43% Repellency averaged water solution over 5 hours Control (57% Ethanol:43% water solution) 38% Methyl Dihydrojasmonate (3.5%) 90% Methyl Dihydrojasmonate - Low epi (2.5%) 58% Methyl Dihydrojasmonate - High epi (2.5%) 39% Propyl Dihydrojasmonate (3.5%) 100% Propyl Dihydrojasmonate - Low epi (2.5%) 74% Propyl Dihydrojasmonate - High epi (2.5%) 49% Para-Menthanediol (3.5%) 91% Gamma-Dodecalactone (3.5%) 82% Gamma-Methyl Tridecalactone (3.5%) 67%
[0045] Another test was designed to measure the repellency as feeding deterrence in Codling Moth larvae. The treatment was applied to the surface of a proprietary laboratory media in a tray with wells for each compound. The media was allowed to dry for 30-60 minutes so that larvae were not exposed to it as a liquid. One first instar codling moth larva was then placed on the treated media. The cover film was placed on the tray to prevent escapes. Ten additional replicates treated with a 57% ethanol solution were prepared to serve as controls. The larvae were observed at 24 hours to determine if they burrowed into the media. After the 24 hour reading, the larvae were extracted from the media to record any mortality.
TABLE-US-00002 TABLE 2 Compound at 3.5% in a 57% Ethanol:43% Repellency at water solution 24 hours Control (57% Ethanol:43% water solution) 8% Para-Menthanediol 10% Propyl Dihydrojasmonate 59% Prenyl Dihydrojasmonate 79% Gamma-Dodecalactone 46% Methyl Apritone 100% 3-Methyl-5-Propyl-2-Cyclohexenone 21% 3-Methyl-5-Propyl-2-Cyclohexenol 30% 3-Methyl-5-Heptyl-2-Cyclohexenone 95%
[0046] Mortality observations were also made for the above protocol. The control was the 57% ethanol:43% water solution.
TABLE-US-00003 TABLE 3 Increased Mortality over Compound at 3.5% in a 57% Ethanol:43% Control Mortality water solution at 24 hours Para-Menthanediol 3% Propyl Dihydrojasmonate 18% Prenyl Dihydrojasmonate 34% Gamma-Dodecalactone 37% Methyl Apritone 95% 3-Methyl-5-Propyl-2-Cyclohexenone 1% 3-Methyl-5-Propyl-2-Cyclohexenol 9% 3-Methyl-5-Heptyl-2-Cyclohexenone 93%
[0047] Ten replicates of 30 kernels of wheat were immersed into the test compound and allowed to dry for 24 hours. The grains were then transferred to a test container, and 10 granary weevils were released. Test containers were secured and left for a 3 week period. At the end of this time, the wheat grains were examined for developing larvae and damage by larvae. The number of grains that contained developing larvae or that showed damage was recorded. Ten additional replicates treated with isopropyl alcohol were prepared to serve as controls.
TABLE-US-00004 TABLE 4 Increased Increased Repellency Mortality over Test compound, diluted in isopropyl vs. Control Control alcohol Repellency Mortality Methyl Apritone (5%) 11% N/A Propyl Dihydrojasmonate (5%) 8% N/A Gamma Tridecalactone (5%) 40% N/A 3-Methyl-5-Propyl-2-Cyclohexenone (5%) 66% 57% 3-Methyl-5-Isobutyl-2-Cyclohexenol (5%) 86% 93%
[0048] The feeding deterrent compounds of this disclosure may be blended with active repellents or toxicants including, but not limited to, NN-Diethyl-m-toluamide (DEET.RTM.) and p-Menthane-3,8-diol (PMD).
[0049] While the disclosure has been described herein with reference to the specific embodiments thereof, it will be appreciated that changes, modification and variations can be made without departing from the spirit and scope of the inventive concept disclosed herein. Accordingly, it is intended to embrace all such changes, modification and variations that fall with the spirit and scope of the appended claims.
* * * * *
References






















XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.