Diagnostic Method For Urinary Tract Infection
BJORCK; Lars ; et al.
U.S. patent application number 16/138555 was filed with the patent office on 2019-03-14 for diagnostic method for urinary tract infection. The applicant listed for this patent is Hansa Medical AB. Invention is credited to Per AKESSON, Lars BJORCK, Bertil CHRISTENSSON, Heiko HERWALD, Adam LINDER.
| Application Number | 20190079104 16/138555 |
| Document ID | / |
| Family ID | 43836342 |
| Filed Date | 2019-03-14 |



| United States Patent Application | 20190079104 |
| Kind Code | A1 |
| BJORCK; Lars ; et al. | March 14, 2019 |
DIAGNOSTIC METHOD FOR URINARY TRACT INFECTION
Abstract
It has been demonstrated that the urinary level of HBP increases in individuals that have a urinary tract infection. Accordingly, the urinary level of HBP in an individual can be used to determine whether or not an individual has a urinary tract infection.
| Inventors: | BJORCK; Lars; (Lund, SE) ; CHRISTENSSON; Bertil; (Lund, SE) ; HERWALD; Heiko; (Veberod, SE) ; LINDER; Adam; (Lund, SE) ; AKESSON; Per; (Lund, SE) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 43836342 | ||||||||||
| Appl. No.: | 16/138555 | ||||||||||
| Filed: | September 21, 2018 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 15153570 | May 12, 2016 | 10126312 | ||
| 16138555 | ||||
| 13983224 | Nov 19, 2013 | |||
| PCT/EP2012/052059 | Feb 7, 2012 | |||
| 15153570 | ||||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C07K 2317/33 20130101; G01N 33/6893 20130101; A61K 45/06 20130101; G01N 2400/40 20130101; G01N 33/569 20130101; G01N 2800/348 20130101; C07K 16/18 20130101; A61P 13/02 20180101 |
| International Class: | G01N 33/68 20060101 G01N033/68; C07K 16/18 20060101 C07K016/18; A61K 45/06 20060101 A61K045/06; G01N 33/569 20060101 G01N033/569 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Feb 7, 2011 | GB | 1102108.6 |
Claims
1-14. (canceled)
15. A method of identifying whether an individual has a urinary tract infection (UTI), comprising: measuring the level of Heparin Binding Protein (HBP, CAP37, Azuricidin) in a urine sample obtained from an individual by; (i) contacting the sample with an antibody which binds specifically to HBP; and (ii) quantitating the amount of antibody-HBP complex to measure the HBP level in the urine sample; and detecting the presence of UTI in the individual at about 93% Sensitivity or greater and at about 90% Specificity or greater based on the measured HBP level in the urine sample.
16. The method of claim 15, wherein the individual is a mammal.
17. The method of claim 16, wherein the mammal is a human.
18. The method of claim 17, wherein the detecting of UTI at about 93% Sensitivity or greater and at about 90% Specificity or greater is determined by a Receiver Operating Characteristic (ROC).
19. The method of claim 17, wherein the 93.3% Sensitivity and the 90.3% Specificity for detecting UTI is a threshold value of about 32 ng/ml in the urine sample.
20. The method of claim 15, wherein the binding of the antibody to HBP is measured by enzyme linked immunosorbent assay (ELISA).
21. The method of claim 15, wherein the binding of the antibody to HBP is measured using a dipstick assay.
22. The method of claim 15, wherein the urinary tract infection is lower urinary tract infection or pyelonephritis.
23. The method of claim 15, further comprising determining the white blood cell count or the level of IL6 in a urine sample from the individual, or conducting the nitrite test on a urine sample from the individual.
Description
FIELD OF THE INVENTION
[0001] The invention relates to the diagnosis and treatment of urinary tract infection.
BACKGROUND OF THE INVENTION
[0002] Urinary tract infection (UTI) is a common diagnosis that can result in serious complications in both children and adults. Efficient diagnosis and treatment is important to reduce the risk of renal failure and overuse of antibiotics. Rapid and safe diagnostic methods are required as an alternative to bacterial cultures. Early treatment improves prognosis of urinary tract infection, and therefore early diagnosis is vital. A reliable biological or clinical marker to determine as early as possible whether or not an individual has a urinary tract infection is needed.
SUMMARY OF THE INVENTION
[0003] Heparin-binding protein (HBP, CAP37, Azurocidin) is a glycosylated, single chain, positively charged 37 kDa inactive serine protease homologue exhibiting 44% sequence identity with human neutrophil elastase. The three dimensional structure of HBP has been published (Iversen et al Nat Struct Biol. 1997 April; 4(4):265-8). It is contained in the azurophilic granulae and secretory vesicles of human neutrophils (Lindmark et al, J Leukoc Biol 1999; 66(4):634-43 and Tapper et al, Blood 2000; 96:2329-2337). It is a multifunctional protein that has been shown to induce vascular leakage by altering the Ca.sup.2+ balance of the blood vessel cytoskeleton (Gautam et al, Nature Medicine 2001; 7(10):1123-7). The M-protein of group A streptococci (GAS) in complex with fibrinogen has been shown to induce HBP release by stimulation of the B2-integrin receptor of neutrophils (Herwald et al, Cell 2004; 116(3):367-79). LPS can also induce HBP release by an unknown mechanism (Rasmussen et al, FEBS Lett 1996; 390(1):109 12). The sequence of HBP is publically available (for example as NCBI accession no. NP_001691 REGION: 27 . . . 248) and is reproduced below as SEQ ID NO.1
TABLE-US-00001 SEQ ID NO: 1 IVGGRKARPRQFPFLASIQNQGRHFCGGALIHARFVMTAASCFQSQNPGV STVVLGAYDLRRRERQSRQTFSISSMSENGYDPQQNLNDLMLLQLDREAN LTSSVTILPLPLQNATVEAGTRCQVAGWGSQRSGGRLSRFPRFVNVTVTP EDQCRPNNVCTGVLTRRGGICNGDGGTPLVCEGLAHGVASFSLGPCGRGP DFFTRVALFRDWIDGVLNNPGP
[0004] Urinary HBP levels in patients suspected of having a urinary tract infection have not previously been investigated. The inventors have shown for the first time that levels of HBP are increased in individuals with urinary tract infection. According to the invention there is thus provided a method of identifying whether or not an individual has urinary tract infection, which method comprises measuring HBP in the individual and thereby determining whether or not the individual has urinary tract infection.
[0005] The invention further provides: [0006] an agent for the detection of HBP for use in determining whether or not an individual has a urinary tract infection; [0007] a test kit for use in a method for determining whether or not an individual has a urinary tract infection, which test kit comprises an agent for the detection of HBP in an individual; [0008] a method of treating an individual for urinary tract infection comprising: [0009] (i) determining whether or not an individual has a urinary tract infection using a method of the invention; and [0010] (ii) administering to an individual identified in (i) as at risk, a therapeutically effective amount of at least one agent suitable for the treatment of urinary tract infection.
DESCRIPTION OF THE FIGURES
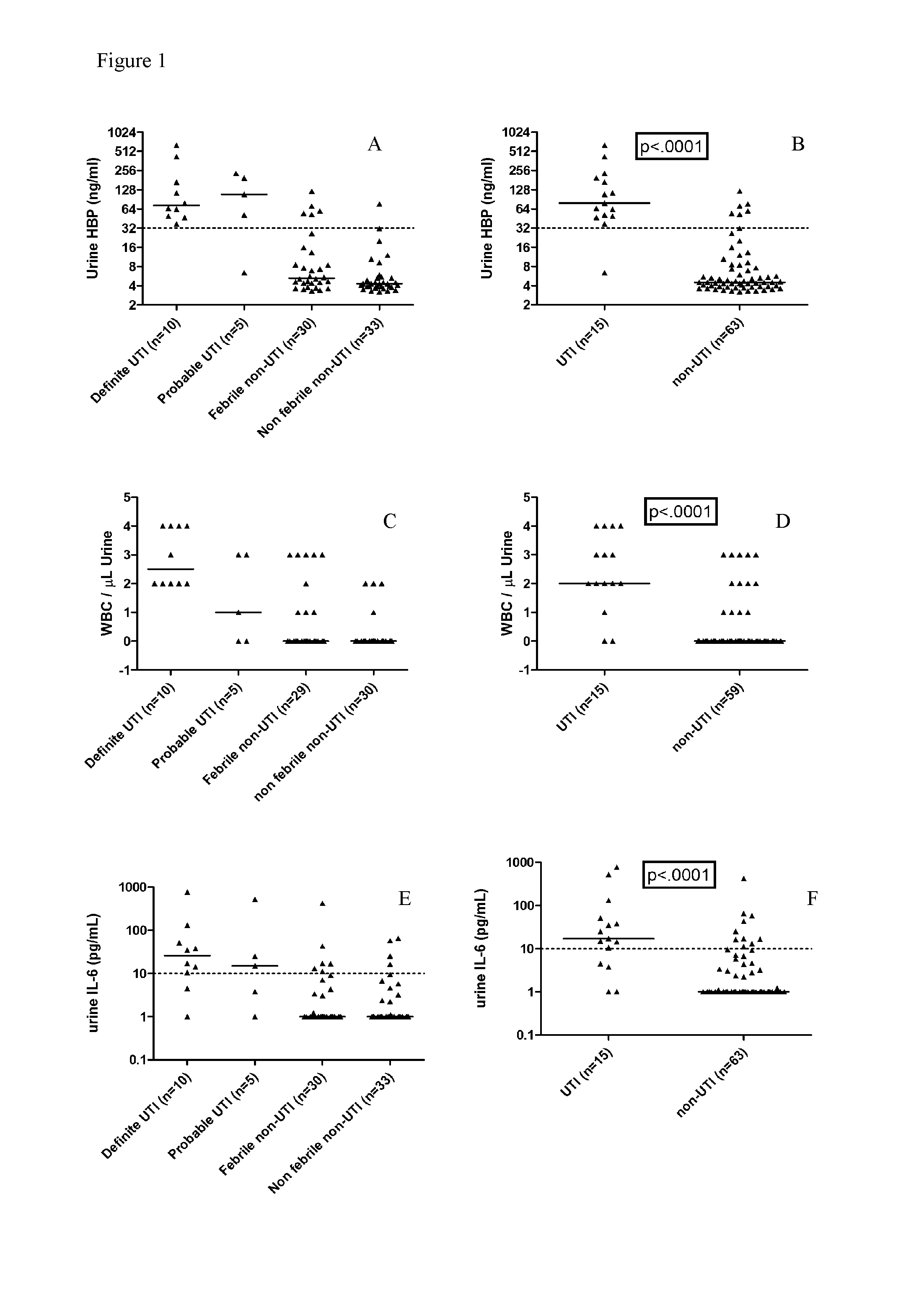
[0011] FIG. 1 shows Urine levels of HBP, U-WBC and IL-6. Each dot represents the concentration in an individual urine sample of HBP (A and B), WBC (C and D) and IL-6 (E and F). The four patients groups are described in the Methods section. Bars represent median of values. The suggested cut-off value for HBP is marked at 32 ng/ml, for WBC at 2 WBC/.mu.L and for IL-6 10 pg/mL.
[0012] FIG. 2 shows Urinary levels of HBP and IL-6 correlated to concentrations of bacteria in urine Each dot represents the concentration in an individual urine sample of HBP (A) and IL-6 (B). Bars represent median of values.
[0013] FIG. 3 shows correlation between HBP and WBC in urine, and between IL-6 and U-WBC in urine. Spearman's rank correlation coefficient (rho)=0.79 indicate that there is a correlation between levels of HBP and WBC in urine (A). Spearman's rank correlation coefficient for IL-6 is 0.338. Each dot represents the concentration in an individual urine sample of HBP, WBC and IL-6.
[0014] FIG. 4 shows Receiver-operating characteristics curves (ROC) of HBP and WBC in urine differentiating between UTI and no UTI. Areas under the ROC curves (AUC) were 0.942 (95% confidence interval, 0.889-0.996) for HBP, 0.808 (95% confidence interval, 0.675-0.941) for IL-6, 0.858 (95% confidence interval, 0.742-0.974) for WBC.
DETAILED DESCRIPTION OF THE INVENTION
Diagnosis
[0015] The present invention relates to a method of identifying whether or not a subject has a urinary tract infection. The invention therefore relates to the diagnosis of urinary tract infection.
[0016] The present inventors have for the first time investigated the level of urinary HBP in patients suspected of having urinary tract infection. The inventors have demonstrated that urinary HBP levels are elevated in patients with urinary tract infection relative to those who do not have urinary tract infection.
[0017] The individual under test is typically suspected of having a urinary tract infection. The individual is typically a mammal. The mammal is typically a human or a domestic mammal such as a horse, a cow, a sheep, a dog or a cat. The individual is preferably a human.
[0018] The individual under test may have one or more symptoms associated urinary tract infections including for example fever, typically greater than 37.5.degree. C., abdominal or back pain, and/or a positive nitrite test.
[0019] The level of HBP is typically measured in vitro in a sample obtained from an individual. The sample typically comprises a body fluid of an individual. A fluid sample may be a sample of blood, plasma, serum, urine, cerebrospinal fluid or joint fluid. The sample is preferably a urine sample.
[0020] According to the present invention, an increased level or concentration of HBP compared with the baseline level or concentration indicates that the individual has a urinary tract infection. The baseline level is typically the level of HBP in an individual who does not have a urinary tract infection, or who is suspected of having a urinary tract infection, but is subsequently confirmed to not have a urinary tract infection. Thus the method according to the present invention may include comparison of the level of HBP in the sample to that of a baseline level.
[0021] In accordance with the present invention the concentration of HBP is greater than 15 ng/ml, preferably greater than 25 ng/ml, more preferably greater than 30 ng/ml, more preferably greater than 32 ng/ml, more preferably greater than 35 ng/ml, and most preferably greater than 50 ng/ml in the sample. Such levels can be used to indicate that an individual has a urinary tract infection.
[0022] In an alternative aspect, the method may comprise assessing the relative level or concentration of HBP in the sample compared to a baseline level or concentration. Typically, a method wherein the level or concentration of HBP in the sample is increased by at least 3 fold or 4 fold relative to the baseline level or concentration of HBP is indicative of urinary tract infection.
[0023] Other markers may also be included in the analysis such as the white blood cell count (WBC), particularly the urine WBC, and/or IL-6. HBP may also be used in combination with the nitrite test.
Detection of HBP
[0024] The invention is typically carried out by measuring the level of HBP in vitro in a sample obtained from the individual. The sample typically comprises a body fluid of the individual. A fluid sample may be a sample of blood, plasma, serum, urine, cerebrospinal fluid or joint fluid. The sample is preferably a urine sample. The sample may be processed prior to being assayed, for example by centrifugation. The sample may also be typically stored prior to assay, preferably below -70.degree. C.
[0025] Standard methods known in the art may be used to assay the level of HBP. These methods typically involve using an agent for the detection of HBP. The agent typically binds specifically to HBP. The agent may be an antibody specific for HBP, an aptamer that binds to HBP, a serine proteinase inhibitor such as aprotinin, for example as described in Petersen et al, Eur J Biochem 1993; 271-9, or a soluble fragment of an integrin, for example as described in Cai and Wright, S. D. J Exp Med 1996; 184:213-23. By specific, it will be understood that the agent or antibody binds to HBP with no significant cross-reactivity to any other molecule, particularly any other protein. For example, an agent or antibody specific for HBP will show no significant cross-reactivity with human neutrophil elastase. Cross-reactivity may be assessed by any suitable method.
[0026] An antibody used in the method of the invention may either be a whole antibody or a fragment thereof which is capable of binding to HBP. The antibody may be monoclonal. Such a whole antibody is typically an antibody which is produced by any suitable method known in the art. For example, polyclonal antibodies may be obtained by immunising a mammal, typically a rabbit or a mouse, with HBP under suitable conditions and isolating antibody molecules from, for example, the serum of said mammal. Monoclonal antibodies may be obtained by hybridoma or recombinant methods.
[0027] Hybridoma methods involve immunising a mammal, typically a rabbit, a rat or a mouse, with HBP under suitable conditions, then harvesting the spleen cells of said mammal and fusing them with myeloma cells. The mixture of fused cells is then diluted and clones are grown from single parent cells. The antibodies secreted by the different clones are then tested for their ability to bind to HBP, and the most productive and stable clone is then grown in culture medium to a high volume. The secreted antibody is collected and purified.
[0028] Recombinant methods involve the cloning into phage or yeast of different immunoglobulin gene segments to create libraries of antibodies with slightly different amino acid sequences. Those sequences which give rise to antibodies which bind to HBP may be selected and the sequences cloned into, for example, a bacterial cell line, for production.
[0029] Typically the antibody is a mammalian antibody, such as a primate, human, rodent (e.g. mouse or rat), rabbit, ovine, porcine, equine or camel antibody. The antibody may be a camelid antibody or shark antibody. The antibody may be a nanobody. The antibody can be any class or isotype of antibody, for example IgM, but is preferably IgG.
[0030] The fragment of whole antibody that can be used in the method comprises an antigen binding site, e.g. Fab or F(ab)2 fragments or ScFV. The whole antibody or fragment may be associated with other moieties, such as linkers which may be used to join together 2 or more fragments or antibodies. Such linkers may be chemical linkers or can be present in the form of a fusion protein with the fragment or whole antibody. The linkers may thus be used to join together whole antibodies or fragments which have the same or different binding specificities, e.g. that can bind the same or different polymorphisms. The antibody may be a bispecific antibody which is able to bind to two different antigens, typically any two of the polymorphisms mentioned herein. The antibody may be a `diabody` formed by joining two variable domains back to back. In the case where the antibodies used in the method are present in any of the above forms which have different antigen binding sites of different specificities then these different specificities are typically to polymorphisms at different positions or on different proteins. In one embodiment the antibody is a chimeric antibody comprising sequence from different natural antibodies, for example a humanised antibody.
[0031] Methods to assess HBP level typically involve contacting a sample with an agent or antibody capable of binding specifically to HBP. Such methods may include dipstick assays and Enzyme-linked Immunosorbant Assay (ELISA). Typically dipsticks comprise one or more antibodies or proteins that specifically bind HBP. If more than one antibody is present, the antibodies preferably have different non-overlapping determinants such that they may bind to HBP simultaneously.
[0032] ELISA is a heterogeneous, solid phase assay that requires the separation of reagents. ELISA is typically carried out using the sandwich technique or the competitive technique. The sandwich technique requires two antibodies. The first specifically binds HBP and is bound to a solid support. The second antibody is bound to a marker, typically an enzyme conjugate. A substrate for the enzyme is used to quantify the HBP-antibody complex and hence the amount of HBP in a sample. The antigen competitive inhibition assay also typically requires an HBP-specific antibody bound to a support. An HBP-enzyme conjugate is added to the sample (containing HBP) to be assayed. Competitive inhibition between the HBP-enzyme conjugate and unlabeled HBP allows quantification of the amount of HBP in a sample. The solid supports for ELISA reactions preferably contain wells.
[0033] The present invention may also employ antibodies to HBP in direct sensing techniques including but not limited to those based upon surface plasmon resonance, surface acoustic wave, quartz crystal microbalance, microcalorimetry or electrochemical impedance spectroscopy.
[0034] The present invention may also employ methods of measuring HBP that do not comprise antibodies. High Performance Liquid Chromatography (HPLC) separation and fluorescence detection is preferably used as a method of determining the HBP level. HPLC apparatus and methods as described previously may be used (Tsikas D et al. J Chromatogr B Biomed Sci Appl 1998; 705:174-6) Separation during HPLC is typically carried out on the basis of size or charge. Prior to HPLC, endogenous amino acids and an internal standard L-homoarginine are typically added to assay samples and these are phase extracted on CBA cartridges (Varian, Harbor City, Calif.). Amino acids within the samples are preferably derivatized with o-phthalaldehyde (OPA). The accuracy and precision of the assay is preferably determined within quality control samples for all amino acids.
[0035] The invention further provides a diagnostic kit that comprises means for measuring the HBP level in an individual and thereby determining whether or not the individual has a urinary tract infection. The kit typically contains one or more antibodies that specifically bind HBP. For example, the kit may comprise a monoclonal antibody, a polyclonal antibody, a single chain antibody, a chimeric antibody, a CDR-grafted antibody or a humanized antibody. The antibody may be an intact immunoglobulin molecule or a fragment thereof such as a Fab, F(ab').sub.2 or Fv fragment. If more than one antibody is present, the antibodies preferably have different non-overlapping determinants such that they may bind to HBP simultaneously.
[0036] The kit may additionally comprise means for the measurement of other laboratory or clinical parameters. For example the kit may comprise means for measuring the WBC count in an individual, and/or the level or concentration of one or more of urinary nitrite, IL-6, glucose, protein and plasma C-reactive protein.
[0037] The kit may additionally comprise one or more other reagents or instruments which enable any of the embodiments of the method mentioned above to be carried out. Such reagents or instruments include one or more of the following: suitable buffer(s) (aqueous solutions), means to isolate HBP from sample, means to obtain a sample from the individual (such as a vessel or an instrument comprising a needle) or a support comprising wells on which quantitative reactions can be done. The kit may, optionally, comprise instructions to enable the kit to be used in the method of the invention or details regarding which individuals the method may be carried out upon.
Therapy
[0038] The present invention also relates to the treatment of an individual identified by a method of the invention as having a urinary tract infection. Thus, a substance for use in the treatment of a urinary tract infection may be used in the manufacture of a medicament for use in the treatment of an individual identified by a method of the invention as having a urinary tract infection. The condition of an individual identified by a method of the invention as having a urinary tract infection can therefore be improved by administration of such a substance. A therapeutically effective amount of a substance useful for the treatment of a urinary tract infection may be given to an individual identified by a method of the invention as in need thereof. Substances suitable for the treatment of a urinary tract infection typically include one or more antibiotics.
[0039] The following Example illustrates the invention:
Example
Methods
Study Population
[0040] This prospective study included 78 individuals (26 male and 52 female), who were admitted to the Department of Paediatric Diseases, the Hospital of Helsingborg, Sweden, between March and June 2009. The criteria for inclusion of patients in the study were fever (temperature .gtoreq.37.5.degree. C.) or symptoms suggesting urinary tract infection, abdominal or back pain in older individuals and nonspecific signs such as irritability or difficulties in feeding in younger children. Urinary tract infection included both lower urinary tract infection and pyelonephritis. Patients with neutropenia (neutrophils .ltoreq.0.5.times.10.sup.9/L) or patients who had received antibiotics in the previous 72 hours were excluded. The project protocol was approved by the ethics committee of Lund University Hospital and consent was obtained from all patients by their parents. Patients were divided into the following groups: 1) Patients with a final diagnosis of urinary tract infection, based on bacterial culture, a urine sample with a single microorganism at .gtoreq.10.sup.5 colony-forming-units (CFU)/ml and typical clinical symptoms. 2) Patients with suspected urinary tract infection, based on clinical characteristics or a urine sample with a single microorganism .gtoreq.10.sup.3 CFU/ml, or a positive nitrite test. The lower cut off value for bacteriuria is based on recommendations from Swedish Institute for Infectious Disease Control. 3) Patients with fever, but non-urinary tract infection 4) Patients without fever, and no urinary tract infection.
Clinical and Laboratory Evaluations
[0041] Body temperature was recorded and CRP was analysed in plasma. Urine was collected with midstream (75 samples) samples or suprapubic aspiration (2 samples). In one patient the technique was not registered. Analyses of urine samples were done with bacterial cultures and dipstick tests, containing tests for nitrite, white blood cells (WBC), albumin, red blood cells and glucose. Dipsticks (Multisticks.RTM. 7, Siemens) were analysed on a Clinitek Status (Bayer Health Care). Nitrite was presented as either positive or negative, and WBC as semi-quantitative values in a graded scale: 0<15 leukocytes/.mu.L urine, 1.gtoreq.15 leukocytes/.mu.L urine, 2.gtoreq.70 leukocytes/.mu.L urine, 3.gtoreq.125 leukocytes/.mu.L urine, 4.gtoreq.500 leukocytes/.mu.L urine. For determination of HBP levels, urine was centrifuged within one hour of the sampling, and aliquots of the supernatants were stored at -70.degree. C. until analysis. The concentration of HBP was determined by enzyme-linked immunosorbent assay (ELISA). Microtiter plates (NUNC) were coated with a mouse monoclonal antibody directed against HBP in coating buffer (0.05M NaHCO.sub.3, pH 9.6). Plates were washed with phosphate-buffered saline and blocked with bovine serum albumin (Sigma). Urine samples were diluted 1/40 in sample buffer (1M NaCl), added to the wells in duplicate, and incubated for 30 min at 37.degree. C. Each plate also contained calibration samples of known concentration of recombinant HBP. After washing, plates were incubated with a polyclonal rabbit antiserum towards HBP diluted 1/7000. Bound antibodies were detected by incubation with peroxidase-conjugated antibody against rabbit IgG, Bio-Rad (1/3000). The level of HBP in each patient sample was determined by calculating the mean optical densities of duplicates which were correlated to the results from the standard curve. Levels of IL-6 in urine were analysed with a sandwich-ELISA, (ELISA MAX.TM. Deluxe Sets), according to the manufacture's description. Urine samples were diluted 1/2 in incubation buffer, and analysed in duplicates.
Statistical Analysis
[0042] Comparisons between groups were made by the non-parametric Kruskal-Wallis test and Mann-Whitney-U-test for continuous variables with skewed distributions. Correlations were made by Spearman's non-parametric test. Areas under receiver-operator characteristic curves were used to illustrate the power of HBP, IL-6, U-WBC and nitrite. Sensitivity, specificity, positive and negative predictive value were calculated for HBP, IL-6 and U-WBC. The level of statistical significance was defined as two-tailed p-value<0.05. The SPSS 17.0 software system (SPSS) was used for statistical analyzes.
Results
Characteristics of the Patients and Laboratory Signs
[0043] Seventy eight individuals were enrolled in the study. The age of the patients ranged from 1 month to 18 years (mean 5.96 years) and 75% were older than 1 year. Ten patients were diagnosed with urinary tract infection (2 with lower urinary tract infection and 8 with pyelonephritis), and all had Escherichia coli in the urine culture (group 1). Five patients had a suspected urinary tract infection based on characteristic symptoms or laboratory results indicating urinary tract infection (group 2). Three of these 5 patients had only a moderate growth of Escherichia coli or beta-hemolytic Streptococci group B in urine (.gtoreq.10.sup.3-.ltoreq.10.sup.5 CFU/ml), one patient had typical clinical symptoms of cystitis but were not cultured and had a negative dipstick test, and one patient had characteristic signs of pyelonephritis with a negative urine culture and a dipstick test that was positive for nitrite. Thirty patients had fever but no laboratory or clinical signs of urinary tract infection (group 3). Their urine cultures were negative or had mixed strains of bacteria (indicative of sample contamination). Thirty-three patients had no fever and no laboratory signs of urinary tract infection (group 4). Their urine cultures were negative. Some of these had diagnoses (known before entering the study) like enuresis, Alport syndrome, Henoch-Schonlein-syndrome and nephrotic syndrome. Statistical Analysis of Results from Plasma and Urine There were no significant differences in median age, gender, temperature, CRP in plasma, HBP in urine, IL-6 in urine, WBC or nitrite in urine between group 1 (urinary tract infection) and 2 (suspected urinary tract infection). Plasma CRP and urinary glucose did not result in any significant differences between any groups. Urinary albumin had significant differences between group 1 and 3 (fever, non-urinary tract infection), 1 and 4 (no fever, non infection), 2 and 3, but not between the others. Urinary red blood cells had significant differences between group 1 and 3, 1 and 4 but not between the others. There were significant differences in median levels of HBP, IL-6 and WBC in urine between group 1 and 3. Also comparison of group 1 and 4 had significantly different median levels of HBP, IL-6 and WBC in urine. There were significant differences in the urine levels of HBP, IL-6 and WBC between group 1 combined with group 2 as compared to group 3 combined with group 4. (FIG. 1 A-F). An interesting implication was that urine with high concentrations of bacteria (.gtoreq.10.sup.5 CFU/ml) seemed to have higher levels of HBP and WBC compared to urine with less or no bacteria (FIG. 2 A-B). Spearman's test with a rank correlation coefficient (rho)=0.79 indicated a relatively strong correlation between levels of HBP and WBC in urine (FIG. 3). IL-6 did not have this strong correlation with WBC in urine, (rho=0.38).
[0044] For calculation of sensitivity, specificity, positive predictive value and negative predictive value group 1 and 2 was combined, referred to urinary tract infection-group and compared to the combined groups 3 and 4, referred to non-urinary tract infection. Using a cut-off level of HBP of .gtoreq.32 ng/ml, the sensitivity and specificity in diagnosing urinary tract infection were 93.3% and 90.3% respectively. HBP in urine had the highest sensitivity compared to WBC, nitrite and IL-6. HBP in urine also had the highest specificity compared to WBC and IL-6 (Table 2). Nitrite had the best specificity compared to the other markers. Based on the 19.2% prevalence of urinary tract infection in this study the positive and negative predictive values of HBP for prediction of urinary tract infection were 70.0% and 98.3%, respectively (Table 2). ROC curves demonstrated that HBP in urine was the best predictor of urinary tract infection with an area under curve (AUC) value of 0.94 compared to U-WBC and IL-6 (FIG. 4).
Discussion
[0045] Urinary tract infection can have serious consequences and antibiotic resistance is a growing issue. Therefore, tests with higher sensitivity and specificity are needed. Several previous studies have investigated biomarkers in urine for the detection of urinary tract infection, like IL-6, lactoferrin, IL-8, TNF-.alpha., elastase and Clara cell protein. HBP in plasma has been studied as a biomarker for sepsis in adults, and data suggested that a normal plasma level of HBP in febrile patients with suspected infection could with high probability (negative predictive value 94.5%) rule out the risk of developing severe sepsis. In the present study, concentrations of HBP in urine was compared with urinary IL-6, nitrite, WBC, red blood cells, albumin, glucose and plasma CRP. HBP in urine had a higher AUC value (0.94) than IL-6, nitrite and WBC, and was thus the best predictor for urinary tract infection. HBP in urine had a high negative predictive value (98.3%), and could thus rule out disease with a high probability. The nitrite test has its limitations with a low sensitivity. Some bacterial species can not transform nitrate to nitrite, for example Staphylococcus saprophyticus and Enterococcus. Other reasons for false negative nitrite are short incubation time of urine in the bladder and intake of high doses of vitamin C. Outliers in the study exist. In group 2 (suspected urinary tract infection), one patient with symptoms of pyelonephritis and beta-hemolytic Streptococci group-B in the urine culture (10.sup.4-10.sup.5 CFU/ml) had a low level of HBP (6.3 ng/ml). This patient was also negative in the rapid test for urinary nitrite and WBC. Urine HBP levels were higher in patients with urinary tract infection than in patients with other kinds of infections, which indicates that HBP release in urine is dependent on the activation of white blood cells by for instance bacterial infection in urine.
TABLE-US-00002 TABLE 1 Patient Characteristics. Clinical and laboratory data. Median, range and P-values for age, gender, CRP, HBP, temperature, U-WBC and nitrite. Group 1 Group 2 Group 3 Group 4 P-value P-value P-value (n = 10) (n = 5) (n = 30) (n = 33) x xx xxx Age 4.5 (0-17) 7.36 (0-16) 2.0 (0-17) 8.1 (0-18) 0.322 0.225 0.299 (years) Gender 9/1 4/1 23/7 16/17 0.604 0.367 0.021 (female/male) HBP 72.1 (37.0-632.2) 108.3 (6.4-230.9) 5.3 (3.3-121.2) 4.3 (3.2-76.5) 1.00 0.000 0.000 (ng/ml) U-WBC.sup.1 3 (2-4) 1 (0-3) 0 (0-3) 0 (0-2) 0.077 0.000 0.000 (WBC/.mu.L) Nitrite 1 (0-1) 0 (0-1) 0 (0-0) 0 (0-0) 0.077 0.000 0.000 (pos/neg).sup.2 Temperature 37.9 (36.7-40.0) 38.5 (37.3-41.1) 38.2 (37.0-41.1) 37.0 (36.9-39.4) 0.297 0.171 0.000 (.degree. C.) CRP 146 (0-201) 165 (10-184) 56 (0-190) 16 (0-169) 0.322 0.152 0.013 (ng/ml) NOTE: P-values analyzed with the Mann-Whitney test. x = Group 1 vs 2, xx = Group 1 vs 3, xxx = Group 1 vs 4 .sup.1Semi-quantitative scale: 0 < 15 leukocytes/.mu.L, 1 .gtoreq. 15 leukocytes/.mu.L, 2 .gtoreq. 70 leukocytes/.mu.L, 3 .gtoreq. 125 leukocytes/.mu.L, 4 .gtoreq. 500 leukocytes/.mu.L .sup.2Positive nitrite was defined as 1 and negative nitrite was defined as 0.
TABLE-US-00003 TABLE 2 Sensitivity, Specificity, PPV and NPV of HBP, U-WBC and nitrite for predicition of UTI. (Cutoff Values of 32 ng/mL for HBP, 10 pg/mL for IL-6, 2 for U-WBC, 100 ng/ml for CRP, 0.5 for nitrite, 38.degree. C. for temperature). Sensitivity, % Specificity, % PPV, % NPV, % HBP 93.3 90.3 70.0 98.3 IL-6 73.3 84.1 52.4 93.0 U-WBC 80.0 84.8 57.1 94.3 Nitrite 53.3 100 100 89.4 CRP 64.3 72.7 50 82.8 Temperature 53.3 63.3 23.7 84.4 PPV, positive predictive value, NPV, negative predictive value.
Sequence CWU 1
1
11222PRThomo sapiens 1Ile Val Gly Gly Arg Lys Ala Arg Pro Arg Gln
Phe Pro Phe Leu Ala 1 5 10 15 Ser Ile Gln Asn Gln Gly Arg His Phe
Cys Gly Gly Ala Leu Ile His 20 25 30 Ala Arg Phe Val Met Thr Ala
Ala Ser Cys Phe Gln Ser Gln Asn Pro 35 40 45 Gly Val Ser Thr Val
Val Leu Gly Ala Tyr Asp Leu Arg Arg Arg Glu 50 55 60 Arg Gln Ser
Arg Gln Thr Phe Ser Ile Ser Ser Met Ser Glu Asn Gly 65 70 75 80 Tyr
Asp Pro Gln Gln Asn Leu Asn Asp Leu Met Leu Leu Gln Leu Asp 85 90
95 Arg Glu Ala Asn Leu Thr Ser Ser Val Thr Ile Leu Pro Leu Pro Leu
100 105 110 Gln Asn Ala Thr Val Glu Ala Gly Thr Arg Cys Gln Val Ala
Gly Trp 115 120 125 Gly Ser Gln Arg Ser Gly Gly Arg Leu Ser Arg Phe
Pro Arg Phe Val 130 135 140 Asn Val Thr Val Thr Pro Glu Asp Gln Cys
Arg Pro Asn Asn Val Cys 145 150 155 160 Thr Gly Val Leu Thr Arg Arg
Gly Gly Ile Cys Asn Gly Asp Gly Gly 165 170 175 Thr Pro Leu Val Cys
Glu Gly Leu Ala His Gly Val Ala Ser Phe Ser 180 185 190 Leu Gly Pro
Cys Gly Arg Gly Pro Asp Phe Phe Thr Arg Val Ala Leu 195 200 205 Phe
Arg Asp Trp Ile Asp Gly Val Leu Asn Asn Pro Gly Pro 210 215 220
D00001

D00002

D00003

D00004

S00001
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.