Reactor Staging For Slurry Hydroconversion Of Polycyclic Aromatic Hydrocarbon Feeds
Johnson; Gregory R. ; et al.
U.S. patent application number 16/115737 was filed with the patent office on 2019-03-14 for reactor staging for slurry hydroconversion of polycyclic aromatic hydrocarbon feeds. The applicant listed for this patent is ExxonMobil Research and Engineering Company. Invention is credited to John P. Greeley, Gregory R. Johnson, Paul Podsiadlo, Kevin Sutowski.
| Application Number | 20190078029 16/115737 |
| Document ID | / |
| Family ID | 63684441 |
| Filed Date | 2019-03-14 |



| United States Patent Application | 20190078029 |
| Kind Code | A1 |
| Johnson; Gregory R. ; et al. | March 14, 2019 |
REACTOR STAGING FOR SLURRY HYDROCONVERSION OF POLYCYCLIC AROMATIC HYDROCARBON FEEDS
Abstract
Methods for processing heavy oil feeds are provided comprising first and second hydroconversion reactors at differing hydroconversion conditions.
| Inventors: | Johnson; Gregory R.; (Bound Brook, NJ) ; Greeley; John P.; (Manasquan, NJ) ; Podsiadlo; Paul; (Humble, TX) ; Sutowski; Kevin; (Basking Ridge, NJ) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 63684441 | ||||||||||
| Appl. No.: | 16/115737 | ||||||||||
| Filed: | August 29, 2018 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 62555734 | Sep 8, 2017 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C10G 67/02 20130101; C10G 65/12 20130101; C10G 65/10 20130101 |
| International Class: | C10G 65/10 20060101 C10G065/10; C10G 67/02 20060101 C10G067/02 |
Claims
1. A method for processing a heavy oil feedstock, comprising: providing a heavy oil feedstock having a 10% distillation point of at least 650.degree. F. (343.degree. C.); exposing the heavy oil feedstock to a catalyst under first effective slurry hydroconversion conditions in a first hydroconversion reactor to form a first effluent, the first effective slurry hydroconversion conditions comprising a temperature of 840.degree. F. (449.degree. C.) to 1000.degree. F. (538.degree. C.) and a pressure of 1500 psig to 3000 psig; exposing at least a portion of the first effluent to a catalyst under second effective slurry hydroconversion conditions in a second hydroconversion reactor to form a second effluent; the second effective slurry hydroconversion conditions comprising a temperature of 750.degree. F. (399.degree. C.) to 840.degree. F. (449.degree. C.) and a pressure of 1500 psig to 3000 psig; wherein the temperature of the second effective slurry hydroconversion conditions is lower than the temperature of the first effective slurry hydroconversion conditions.
2. The method of claim 1, further comprising exposing the first effluent to a separator; wherein the separator removes naphtha and distillate fractions from the first effluent prior to a exposing the first effluent to the second hydroconversion reactor.
3. The method of claim 2, wherein the separator comprises a first separator and a second separator; further comprising exposing the first effluent to the first separator, thereby forming a first separator bottoms fraction and a first separator light fraction; wherein the first separator bottoms fraction comprises 650.degree. F.+ (343.degree. C.+) hydrocarbons and the first separator light fraction comprises 650.degree. F.- (343.degree. C.-) hydrocarbons and treat gas; exposing at least a portion of the first separator bottoms fraction to the second hydroconversion reactor; exposing the first separator light fraction to the second separator, thereby forming a second separator bottoms fraction comprising liquid 160.degree. F.+ (71.degree. C.+) hydrocarbons and a second separator light fraction comprising 160.degree. F.- (71.degree. C.-) hydrocarbons and treat gas; and exposing at least a portion of the second separator light fraction to the second hydroconversion reactor.
4. The method of claim 2, wherein the separator comprises a first separator and a second separator; further comprising exposing the first effluent to the first separator, thereby forming a first separator bottoms fraction comprising liquid 160.degree. F.+ (71.degree. C.+) hydrocarbons and a first separator light fraction comprising 160.degree. F.- (71.degree. C.-) hydrocarbons and treat gas; exposing at least a portion of the first separator light fraction to the second hydroconversion reactor; exposing at least a portion of the first separator bottoms fraction to the second separator; thereby forming a second separator bottoms fraction and a second separator light fraction; wherein the second separator bottoms fraction comprises 650.degree. F.+ (343.degree. C.+) hydrocarbons and the second separator light fraction comprises 650.degree. F.- (343.degree. C.-) hydrocarbons and treat gas; and exposing at least a portion of the second separator bottoms fraction to the second hydroconversion reactor.
5. The method of claim 1, wherein the first effective slurry hydroconversion conditions and the second slurry hydroconversion conditions are effective for a combined conversion of at least 70 wt % of the heavy oil feedstock relative to a conversion temperature of at least 700.degree. F. (371.degree. C.).
6. The method of claim 1, wherein the second effluent has an API gravity of at least 12.
7. The method of claim 5, wherein the second effluent has an API gravity of at least 12.
8. The method of claim 1, wherein the heavy oil feedstock comprises at least one of fluid catalytic cracker main column bottoms, steam cracker tar, and coker gas oil.
9. The method of claim 1, wherein the catalyst comprises MoS.sub.2.
10. The method of claim 1, wherein the catalyst is present in the heavy oil feedstock at a concentration of 50 wppm to 500 wppm.
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application claims priority to U.S. Provisional Application Ser. No. 62/555,734 filed on Sep. 8, 2017, which is herein incorporated by reference in its entirety.
FIELD
[0002] This invention provides methods for slurry hydroprocessing of polyaromatic hydrocarbon feeds, such as fluid catalytic cracker (FCC) main column bottoms (MCB), steam cracker tar, and coker gas oil.
BACKGROUND
[0003] Slurry hydroprocesssing provides a method for conversion of high boiling, low value petroleum fractions into higher value liquid products. Slurry hydroconversion technology can process difficult feeds, such as feeds with high concentrations of polycyclic aromatic hydrocarbons (PAH), while still maintaining high liquid yields. Slurry hydroconversion units have been used to process challenging streams present in refinery/petrochemical complexes such as FCC MCB, steam cracked tar, and coker gas oil. Slurry hydroprocessing offers a means for converting low-value heavy feedstocks into fuels using a technology that is not as susceptible to fouling as emerging fixed-bed technologies. In a fixed-bed reactor, there is a risk that feeds containing polycyclic aromatic hydrocarbons will lead to deposits forming on the reactor internals and catalyst bed, resulting in a build-up of pressure that hinders process operability. Because a slurry hydroconversion reactor does not have an internal catalyst bed, these risks are largely avoided. Unfortunately, slurry hydroprocessing is also an expensive refinery process from both a capital investment standpoint and a hydrogen consumption standpoint.
[0004] Various slurry hydroprocessing configurations have previously been described. For example, U.S. Pat. No. 5,755,955 and U.S. Patent Application Publication 2010/0122939 provide examples of configurations for performing slurry hydroprocessing. U.S. Patent Application Publication 2011/0210045 also describes examples of configurations for slurry hydroconversion, including examples of configurations where the heavy oil feed is diluted with a stream having a lower boiling point range, such as a vacuum gas oil stream and/or catalytic cracking slurry oil stream, and examples of configurations where a bottoms portion of the product from slurry hydroconversion is recycled to the slurry hydroconversion reactor.
[0005] U.S. Pat. No. 9,605,217 relates to a multi-stage slurry hydroconversion process using multiple reactor stages in series at different pressures.
[0006] U.S. Patent Application Publication 2013/0075303 describes a reaction system for combining slurry hydroconversion with a coking process. An unconverted portion of the feed after slurry hydroconversion is passed into a coker for further processing. The resulting coke is described as being high in metals.
[0007] U.S. Patent Application Publication 2013/0112593 describes a reaction system for performing slurry hydroconversion on a deasphalted heavy oil feed. The asphalt from a deasphalting process and a portion of the unconverted material from the slurry hydroconversion can be gasified to form hydrogen and carbon oxides.
[0008] Current slurry hydroconversion technology converts high PAH feeds into lighter to molecules suitable for higher value dispositions such as gasoline or diesel. In a slurry hydroconversion process, high temperatures (>830.degree. F.) are used to thermally crack heavy hydrocarbons in the presence of hydrogen. With higher reactor temperature, the rate of the cracking reaction is accelerated, reducing the reactor volume required to achieve a target conversion at a given feed rate. A negative side effect of operating at higher temperature is that the desaturation of aromatics becomes more thermodynamically favored. This phenomenon results in higher liquid product densities having lower economic value than would be achieved in the absence of desaturation. It would be beneficial to have a process that can both achieve high rates of conversion, but also maintain a lower liquid product density to increase economic value.
[0009] This application provides a process for enabling high feed conversion without sacrificing product density by using multiple slurry hydroconversion reactor stages operated at different temperatures. The first stage is operated at high temperature to efficiently crack the feed to lighter boiling point components and at least one additional reactor stage is operated at lower temperature to reverse the aromatic desaturation that occurred in the first stage. In lieu of using a second slurry hydroconversion stage, the liquid product could be hydrogenated using a conventional fixed-bed catalyst hydrogenation process. However, the presence of slurry catalyst, demetallization products, and coke in the liquid effluent from the first reactor stage could create fouling risks for the fixed-bed hydrogenation reactor. By using a slurry reactor as hydrogenation step, these risks are avoided. In general, a slurry reactor system will be more robust to poor feed quality than a fixed-bed process.
SUMMARY
[0010] In various aspects, methods for processing a heavy oil feedstock are provided. In certain aspects, the methods comprise providing a heavy oil feedstock having a 10% distillation point of at least about 650.degree. F. (343.degree. C.); exposing the heavy oil feedstock to a catalyst under first effective slurry hydroconversion conditions in a first hydroconversion reactor to form a first effluent, the first effective slurry hydroconversion conditions comprising a temperature of about 840.degree. F. (449.degree. C.) to about 1000.degree. F. (538.degree. C.) and a pressure of about 1500 psig to 3000 psig; exposing at least a portion of the first effluent to a catalyst under second effective slurry hydroconversion conditions in a second hydroconversion reactor to form a second effluent; the second effective slurry hydroconversion conditions comprising a temperature of about 750.degree. F. (399.degree. C.) to about 840.degree. F. (449.degree. C.) and a pressure of about 1500 psig to about 3000 psig; wherein the temperature of the second effective slurry hydroconversion conditions is lower than the temperature of the first effective slurry hydroconversion conditions.
[0011] In another aspect, the method further comprises exposing the first effluent to a separator; wherein the separator removes naphtha and distillate fractions from the first effluent prior to a exposing the first effluent to the second hydroconversion reactor. In certain aspects the separator comprises a first separator and a second separator; further comprising exposing the first effluent to the first separator, thereby forming a first separator bottoms fraction and a first separator light fraction; wherein the first separator bottoms fraction comprises 650.degree. F.+ (343.degree. C.+) hydrocarbons and the first separator light fraction comprises 650.degree. F.- (343.degree. C.-) hydrocarbons and treat gas; exposing at least a portion of the first separator bottoms fraction to the second hydroconversion reactor; exposing the first separator light fraction to the second separator, thereby forming a second separator bottoms fraction comprising liquid 160.degree. F.+ (71.degree. C.+) hydrocarbons and a second separator light fraction comprising 160.degree. F.- (71.degree. C.-) hydrocarbons and treat gas; and exposing at least a portion of the second separator light fraction to the second hydroconversion reactor. In an alternative aspect, the separator comprises a first separator and a second separator; further comprising exposing the first effluent to the first separator, thereby forming a first separator bottoms fraction comprising liquid 160.degree. F.+ (71.degree. C.+) hydrocarbons and a first separator light fraction comprising 160.degree. F.- (71.degree. C.-) hydrocarbons and treat gas; exposing at least a portion of the first separator light fraction to the second hydroconversion reactor; exposing at least a portion of the first separator bottoms fraction to the second separator; thereby forming a second separator bottoms fraction and a second separator light fraction; wherein the second separator bottoms fraction comprises 650.degree. F.+ (343.degree. C.+) hydrocarbons and the second separator light fraction comprises 650.degree. F.- (343.degree. C.-) hydrocarbons and treat gas; and exposing at least a portion of the second separator bottoms fraction to the second hydroconversion reactor.
[0012] In yet another aspect, the first effective slurry hydroconversion conditions and the second slurry hydroconversion conditions are effective for a combined conversion of at least about 70 wt % of the heavy oil feedstock relative to a conversion temperature of at least about 700.degree. F. (371.degree. C.). Additionally or alternatively, the second effluent may have an API gravity of at least 12. In another aspect, the heavy oil feedstock comprises at least one of fluid catalytic cracker main column bottoms, steam cracker tar, and coker gas oil. The catalyst may comprise MoS.sub.2 in a concentration of 50 wppm to 500 wppm.
BRIEF DESCRIPTION OF THE FIGURES
[0013] FIG. 1 shows an example of a slurry hydroconversion reaction system according to the present disclosure.
[0014] FIG. 2 shows an example of a slurry hydroconversion reaction system according to the present disclosure.
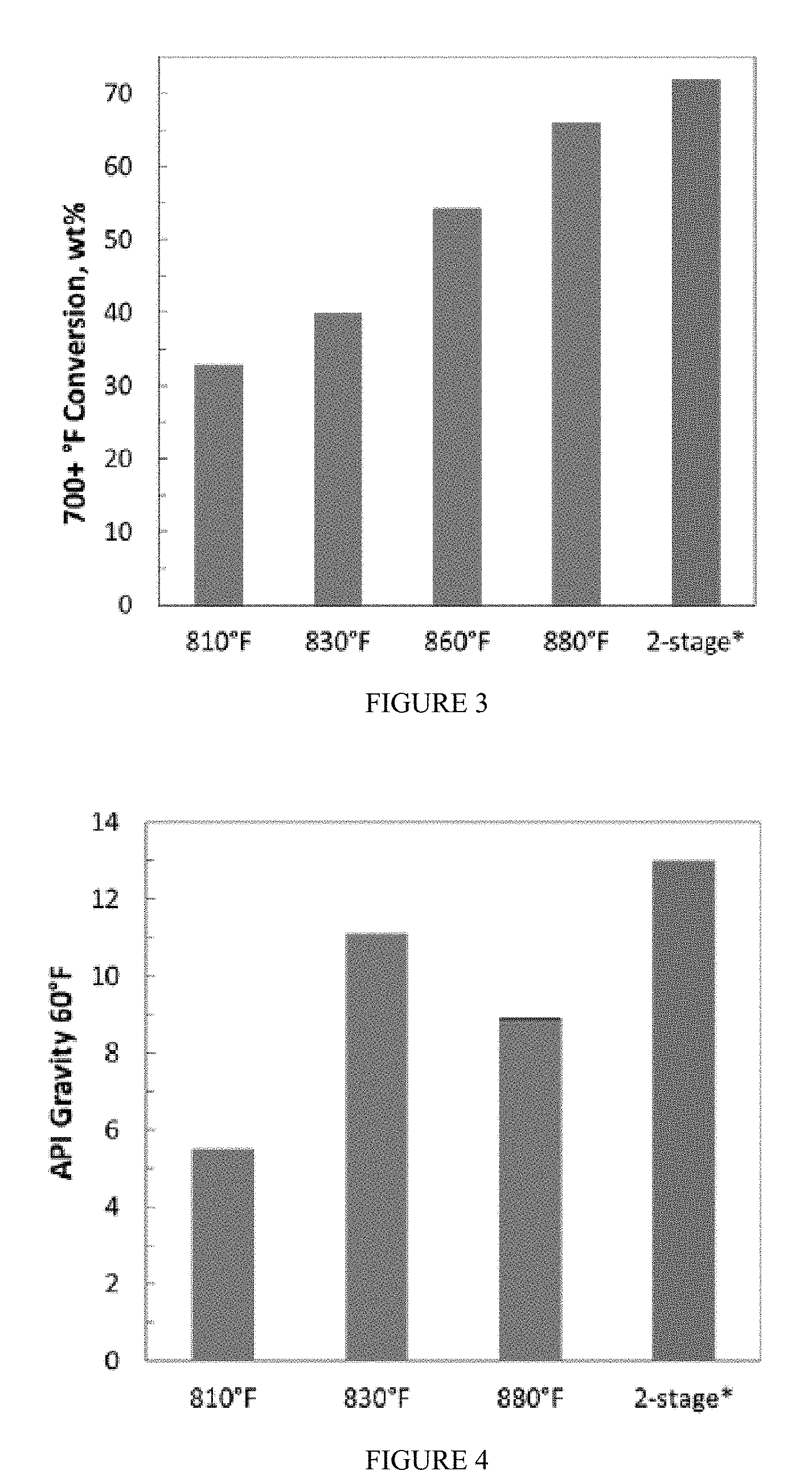
[0015] FIG. 3 provides a graphical depiction of conversion of a PAH feed hydroprocessed using a reaction system according to the present disclosure.
[0016] FIG. 4 provides a graphical depiction of liquid product API of a PAH feed hydroprocessed using a reaction system according to the present disclosure.
DETAILED DESCRIPTION OF THE EMBODIMENTS
Overview
[0017] In various aspects, a process for hydroconverting heavy feeds rich in polycyclic aromatic hydrocarbons, such as bottoms from a fluid catalytic cracking (FCC) process, using a multistage slurry reactor system is provided. The feed enters a slurry reactor operated at a high temperature to initiate thermally-driven hydrocracking reactions. The operating temperature and pressure of this stage are maximized to drive the conversion of multi-ring aromatic compounds having boiling points greater than 650 F..degree. into lighter compounds. The process involves at least one additional reactor stage operated at lower temperature to promote the saturation of aromatic and olefinic compounds, increasing the API gravity of the hydrocracked liquid product. An optional interstage, highpressure separator system allows for recovery of naphtha and distillate after the first reactor stage to prevent over-cracking of these desired products. With respect to existing slurry hydroconversion process configurations, the present disclosure allows one to achieve both high boiling point conversion and a high product API gravity, which tend to be mutually exclusive.
[0018] In an embodiment, a feed high in polycyclic aromatic hydrocarbons, such as FCC MCB, steam cracker tar, or coker gas oil, is passed into an initial slurry hydroconversion reactor operated a temperature of at least about 840.degree. F. (449.degree. C.)--e.g. at least about 850.degree. F. (454.degree. C.), at least about 860.degree. F. (460.degree. C.), at least about 870.degree. F. (465.degree. C.), at least about 880.degree. F. (471.degree. C.)--or from about 840.degree. F. (449.degree. C.) to about 1000.degree. F. (538.degree. C.)--e.g. from about 850.degree. F. (454.degree. C.) to about 980.degree. F. (527.degree. C.), from about 860.degree. F. (460.degree. C.) to about 950.degree. F. (510.degree. C.). The high temperature in the initial reaction stage results in a preferred conversion of the feed, but also in increased aromatic desaturation (i.e. increased density). The conversion products from the initial slurry hydroconversion reactor can optionally be separated by an interstage separator to pull out desired naphtha and distillate boiling range products.
[0019] The bottoms portion from the initial low pressure hydroconversion reactor is then passed into a second slurry hydroconversion reactor operated at a temperature of about 840.degree. F. (449.degree. C.) or less--e.g. about 830.degree. F. (443.degree. C.) or less, about 820.degree. F. (438.degree. C.) or less, about 810.degree. F. (432.degree. C.) or less, about 800.degree. F. (427.degree. C.) or less--or from about 700.degree. F. (371.degree. C.) to about 840.degree. F. (449.degree. C.)--e.g. from about 720.degree. F. (382.degree. C.) to about 830.degree. F. (443.degree. C.), from about 750.degree. F. (399.degree. C.) to about 820.degree. F. (438.degree. C.). The lower temperature in the second reaction stage promotes aromatic saturation of portions of the feed desaturated in the first, high-temperature reaction stage, which in turn lowers the gravity of the final hydroprocessed products.
Feedstocks
[0020] In various aspects, a hydroprocessed product is produced from a heavy oil feed component. Examples of heavy oils include, but are not limited to, heavy crude oils, distillation residues, heavy oils coming from catalytic treatment (such as heavy cycle bottom slurry oils from fluid catalytic cracking), thermal tars (such as oils from visbreaking, steam cracking, or similar thermal or non-catalytic processes), oils (such as bitumen) from oil sands and heavy oils derived from coal.
[0021] Heavy oil feedstocks can be liquid or semi-solid. Examples of heavy oils that can be hydroprocessed, treated or upgraded according to this invention include bitumens and residuum from refinery distillation processes, including atmospheric and vacuum distillation processes. Such heavy oils can have an initial boiling point of 650.degree. F. (343.degree. C.) or greater. Preferably, the heavy oils will have a 10% distillation point of at least 650.degree. F. (343.degree. C.), alternatively at least 660.degree. F. (349.degree. C.) or at least 750.degree. F. (399.degree. C.). In some aspects the 10% distillation point can be still greater, such as at least 900.degree. F. (482.degree. C.), or at least 950.degree. F. (510.degree. C.), or at least 975.degree. F. (524.degree. C.), or at least 1020.degree. F. (549.degree. C.) or at least 1050.degree. F. (566.degree. C.). In this discussion, boiling points can be determined by a convenient method, such as ASTM D86, ASTM D2887, or another suitable standard method.
[0022] Steam cracker tar (SCT) as used herein is also referred to in the art as "pyrolysis fuel oil". The terms can be used interchangeably herein. The tar will typically be obtained from the first fractionator downstream from a steam cracker (pyrolysis furnace) as the bottoms product of the fractionator, nominally having a boiling point of at least about 550.degree. F.+ (.about.288.degree. C.+). Boiling points and/or fractional weight distillation points can be determined by, for example, ASTM D2892. Alternatively, SCT can have a T5 boiling point (temperature at which 5 wt % will boil off) of at least about 550.degree. F. (.about.288.degree. C.). The final boiling point of SCT can be dependent on the nature of the initial pyrolysis feed and/or the pyrolysis conditions, and typically can be about 1450.degree. F. (.about.788.degree. C.) or less.
[0023] SCT can have a relatively low hydrogen content compared to heavy oil fractions that are typically processed in a refinery setting. In some aspects, SCT can have a hydrogen content of about 8.0 wt % or less, about 7.5 wt % or less, or about 7.0 wt % or less, or about 6.5 wt % or less. In particular, SCT can have a hydrogen content of about 5.5 wt % to about 8.0 wt %, or about 6.0 wt % to about 7.5 wt %. Additionally or alternately, SCT can have a micro carbon residue (or alternatively Conradson Carbon Residue) of at least about 10 wt %, or at least about 15 wt %, or at least about 20 wt %, such as up to about 40 wt % or more.
[0024] SCT can also be highly aromatic in nature. The paraffin content of SCT can be about 2.0 wt % or less, or about 1.0 wt % or less, such as having substantially no paraffin content. The naphthene content of SCT can also be about 2.0 wt % or less or about 1.0 wt % or less, such as having substantially no naphthene content. In some aspects, the combined paraffin and naphthene content of SCT can be about 1.0 wt % or less. With regard to aromatics, at least about 30 wt % of SCT can correspond to 3-ring aromatics, or at least 40 wt %. In particular, the 3-ring aromatics content can be about 30 wt % to about 60 wt %, or about 40 wt % to about 55 wt %, or about 40 wt % to about 50 wt %. Additionally or alternately, at least about 30 wt % of SCT can correspond to 4-ring aromatics, or at least 40 wt %. In particular, the 4-ring aromatics content can be about 30 wt % to about 60 wt %, or about 40 wt % to about 55 wt %, or about 40 wt % to about 50 wt %. Additionally or alternately, the 1-ring aromatic content can be about 15 wt % or less, or about 10 wt % or less, or about 5 wt % or less, such as down to about 0.1 wt %.
[0025] SCT can also have a higher density than many types of crude or refinery fractions. In various aspects, SCT can have a density at 15.degree. C. of about 1.08 g/cm.sup.3 to about 1.20 g/cm.sup.3, or 1.10 g/cm.sup.3 to 1.18 g/cm.sup.3. By contrast, many types of vacuum resid fractions can have a density of about 1.05 g/cm.sup.3 or less. Additionally or alternately, density (or weight per volume) of the heavy hydrocarbon can be determined according to ASTM D287-92 (2006) Standard Test Method for API Gravity of Crude Petroleum and Petroleum Products (Hydrometer Method), which characterizes density in terms of API gravity. In general, the higher the API gravity, the less dense the oil. API gravity can be 5.degree. or less, or 0.degree. or less, such as down to about -10.degree. or lower.
[0026] Contaminants such as nitrogen and sulfur are typically found in SCT, often in organically-bound form. Nitrogen content can range from about 50 wppm to about 10,000 wppm elemental nitrogen or more, based on total weight of the SCT. Sulfur content can range from about 0.1 wt % to about 10 wt %, based on total weight of the SCT.
[0027] Coker bottoms represent another type of cracked feed suitable for hydroprocessing, optionally in combination with a catalytic slurry oil and/or steam cracker tar and/or other cracked fractions. Coking is a thermal cracking process that is suitable for conversion of heavy feeds into fuels boiling range products. The feedstock to a coker typically also includes 5 wt % to 25 wt % recycled product from the coker, which can be referred to as coker bottoms. This recycle fraction allows metals, asphaltenes, micro-carbon residue, and/or other solids to be returned to the coker, as opposed to being incorporated into a coker gas oil product. This can maintain a desired product quality for the coker gas oil product, but results in a net increase in the amount of light ends and coke that are generated by a coking process. The coker bottoms can correspond to a fraction with a T10 distillation point of at least 550.degree. F. (288.degree. C.), or at least 300.degree. C., or at least 316.degree. C., and a T90 distillation point of 566.degree. C. or less, or 550.degree. C. or less, or 538.degree. C. or less. The coker recycle fraction can have an aromatic carbon content of about 20 wt % to about 50 wt %, or about 30 wt % to about 45 wt %, and a micro carbon residue content of about 4.0 wt % to about 15 wt %, or about 6.0 wt % to about 15 wt %, or about 4.0 wt % to about 10 wt %, or about 6.0 wt % to about 12 wt %.
[0028] In addition to initial boiling points and/or 10% distillation points, other distillation points may also be useful in characterizing a feedstock. For example, a feedstock can be characterized based on the portion of the feedstock that boils above 1050.degree. F. (566.degree. C.). In some aspects, a feedstock can have a 70% distillation point of 1050.degree. F. or greater, or a 60% distillation point of 1050.degree. F. or greater, or a 50% distillation point of 1050.degree. F. or greater, or a 40% distillation point of 1050.degree. F. or greater.
[0029] Density, or weight per volume, of the heavy hydrocarbon can be determined according to ASTM D287-92 (2006) Standard Test Method for API Gravity of Crude Petroleum and Petroleum Products (Hydrometer Method), and is provided in terms of API gravity. In general, the higher the API gravity, the less dense the oil. API gravity is 20.degree. or less in one aspect, 15.degree. or less in another aspect, and 10.degree. or less in another aspect.
[0030] Heavy oil feedstocks (also referred to as heavy oils) can be high in metals. For example, the heavy oil can be high in total nickel, vanadium and iron contents. In one embodiment, the heavy oil will contain at least 0.00005 grams of Ni/V/Fe (50 ppm) or at least 0.0002 grams of Ni/V/Fe (200 ppm) per gram of heavy oil, on a total elemental basis of nickel, vanadium and iron. In other aspects, the heavy oil can contain at least about 500 wppm of nickel, vanadium, and iron, such as at least about 1000 wppm.
[0031] Contaminants such as nitrogen and sulfur are typically found in heavy oils, often in organically-bound form. Nitrogen content can range from about 50 wppm to about 10,000 wppm elemental nitrogen or more, based on total weight of the heavy hydrocarbon component. The nitrogen containing compounds can be present as basic or non-basic nitrogen species. Examples of basic nitrogen species include quinolines and substituted quinolines. Examples of non-basic nitrogen species include carbazoles and substituted carbazoles.
Slurry Hydroprocessing
[0032] FIG. 1 shows an example of a reaction system according to the present disclosure. In
[0033] FIG. 1, a feed mixture 105, which is a mixture of heavy feed, catalyst slurry, and Hz-rich treat gas, is admitted to slurry hydroprocessing reactor 101 operated at temperature of at least about 840.degree. F. (449.degree. C.)--e.g. at least about 850.degree. F. (454.degree. C.), at least about 860.degree. F. (460.degree. C.), at least about 870.degree. F. (465.degree. C.), at least about 880.degree. F. (471.degree. C.)--or from about 840.degree. F. (449.degree. C.) to about 1000.degree. F. (538.degree. C.)--e.g. from about 850.degree. F. (454.degree. C.) to about 980.degree. F. (527.degree. C.), from about 860.degree. F. (460.degree. C.) to about 950.degree. F. (510.degree. C.). Feed mixture 105 can be heated prior to entering reactor 101 in order to achieve a desired temperature for the slurry hydroprocessing reaction.
[0034] The effluent 106 from slurry hydroprocessing reactor 101 is passed into one or more separation stages--two are shown in FIG. 1. For example, an initial separation stage can be a high pressure separator 102. A lower boiling point portion 107--e.g. treat gas and 650.degree. F.- (343.degree. C.-) hydrocarbons--leaving as vapor from separator 102 can be passed to a second high pressure separator 103. In separator 103, distillate and naphtha are separated from light ends (C.sub.1 to C.sub.4 hydrocarbons) and treat gas. Liquid product 110 comprising naphtha and distillate exit separator 103 from the bottom and light ends stream 109 is transported to slurry hydroprocessing reactor 104. As used herein, naphtha and distillate fractions include naphtha fractions, kerosene fractions, diesel fractions, and other heavier (gas oil) fractions. Each of these types of fractions can be defined based on a boiling range, such as a boiling range that includes at least .about.90 wt % of the fraction, or at least .about.95 wt % of the fraction. For example, for many types of naphtha fractions, at least .about.90 wt % of the fraction, or at least .about.95 wt %, can have a boiling point in the range of .about.85.degree. F. (.about.29.degree. C.) to .about.350.degree. F. (.about.177.degree. C.). For some heavier naphtha fractions, at least .about.90 wt % of the fraction, and preferably at least .about.95 wt %, can have a boiling point in the range of .about.85.degree. F. (.about.29.degree. C.) to .about.400.degree. F. (.about.204.degree. C.). For a kerosene fraction, at least .about.90 wt % of the fraction, or at least .about.95 wt %, can have a boiling point in the range of .about.300.degree. F. (.about.149.degree. C.) to .about.600.degree. F. (.about.288.degree. C.). For a kerosene fraction targeted for some uses, such as jet fuel production, at least .about.90 wt % of the fraction, or at least .about.95 wt %, can have a boiling point in the range of .about.300.degree. F. (.about.149.degree. C.) to .about.550.degree. F. (.about.288.degree. C.). For a diesel fraction, at least .about.90 wt % of the fraction, and preferably at least .about.95 wt %, can have a boiling point in the range of .about.350.degree. F. (.about.177.degree. C.) to .about.700.degree. F. (.about.371.degree. C.). For a (vacuum) gas oil fraction, at least .about.90 wt % of the fraction, and preferably at least .about.95 wt %, can have a boiling point in the range of .about.650.degree. F. (.about.343.degree. C.) to .about.1100.degree. F. (.about.593.degree. C.). Optionally, for some gas oil fractions, a narrower boiling range may be desirable. For such gas oil fractions, at least .about.90 wt % of the fraction, or at least .about.95 wt %, can have a boiling point in the range of .about.650.degree. F. (.about.343.degree. C.) to .about.1000.degree. F. (.about.538.degree. C.), or .about.650.degree. F. (.about.343.degree. C.) to .about.900.degree. F. (.about.482.degree. C.).
[0035] Unconverted feed 108 from separator 102 is also sent to reactor 104. Reactor 104 is operated at lower temperature than reactor 101 to reverse the aromatic desaturation that occurred in separator 102. Typical operating temperature of reactor 104 include about 840.degree. F. (449.degree. C.) or less--e.g. about 830.degree. F. (443.degree. C.) or less, about 820.degree. F. (438.degree. C.) or less, about 810.degree. F. (432.degree. C.) or less, about 800.degree. F. (427.degree. C.) or less--or from about 700.degree. F. (371.degree. C.) to about 840.degree. F. (449.degree. C.)--e.g. from about 720.degree. F. (382.degree. C.) to about 830.degree. F. (443.degree. C.), from about 750.degree. F. (399.degree. C.) to about 830.degree. F. (443.degree. C.). Effluent 111 from reactor 104 achieves greater conversion of feed mixture 105 at a lower density than conventional slurry hydroprocessing systems.
[0036] FIG. 2 shows an alternative example of a reaction system according to the present disclosure. In FIG. 2, a feed mixture 205, which is a mixture of heavy feed, catalyst slurry, and Hz-rich treat gas, is admitted to slurry hydroprocessing reactor 201 operated at temperature of at least about 840.degree. F. (449.degree. C.)--e.g. at least about 850.degree. F. (454.degree. C.), at least about 860.degree. F. (460.degree. C.), at least about 870.degree. F. (465.degree. C.), at least about 880.degree. F. (471.degree. C.)--or from about 840.degree. F. (449.degree. C.) to about 1000.degree. F. (538.degree. C.)--e.g. from about 850.degree. F. (454.degree. C.) to about 980.degree. F. (527.degree. C.), from about 860.degree. F. (460.degree. C.) to about 950.degree. F. (510.degree. C.). Feed mixture 205 can be heated prior to entering reactor 201 in order to achieve a desired temperature for the slurry hydroprocessing reaction.
[0037] The effluent 206 from slurry hydroprocessing reactor 201 is passed into one or more separation stages--two are shown in FIG. 2. For example, an initial separation stage can be a high pressure separator 202. A higher boiling point portion 107--e.g. 160.degree. F.+(71.degree. C.+) hydrocarbons--leaving as liquid from separator 202 can be passed to a second high pressure separator 203. In separator 203, distillate and naphtha 210 are separated from unconverted feed 209 of feed mixture 205. Light ends (C.sub.1 to C.sub.4 hydrocarbons) and treat gas 208 are sent to second slurry hydroprocessing reactor 204 along with unconverted feed 209.
[0038] Reactor 204 is operated at lower temperature than reactor 201 to reverse the aromatic desaturation that occurred in separator 202. Typical operating temperature of reactor 104 include about 840.degree. F. (449.degree. C.) or less--e.g. about 830.degree. F. (443.degree. C.) or less, about 820.degree. F. (438.degree. C.) or less, about 810.degree. F. (432.degree. C.) or less, about 800.degree. F. (427.degree. C.) or less--or from about 700.degree. F. (371.degree. C.) to about 840.degree. F. (449.degree. C.)--e.g. from about 720.degree. F. (382.degree. C.) to about 830.degree. F. (443.degree. C.), from about 750.degree. F. (399.degree. C.) to about 830.degree. F. (443.degree. C.). Effluent 211 from reactor 204 achieves greater conversion of feed mixture 205 at a lower density than conventional slurry hydroprocessing systems.
[0039] The reaction conditions in a slurry hydroprocessing reactor can vary based on the nature of the catalyst, the nature of the feed, the desired products, and/or the desired amount of conversion. With regard to catalyst, suitable catalyst concentrations can range from about 50 wppm to about 20,000 wppm (or about 2 wt %), e.g. about 50 wppm to about 10,000 wppm, about 50 wppm to about 1,000 wppm, about 50 wppm to about 500 wppm, depending on the nature of the catalyst. Catalyst can be incorporated into a hydrocarbon feedstock directly, or the catalyst can be incorporated into a side or slip stream of feed and then combined with the main flow of feedstock. Still another option is to form catalyst in-situ by introducing a catalyst precursor into a feed (or a side/slip stream of feed) and forming catalyst by a subsequent reaction.
[0040] Catalytically active metals for use in hydroprocessing can include those from Group IVB, Group VB, Group VIB, Group VIIB, or Group VIII of the Periodic Table. Examples of suitable metals include iron, nickel, molybdenum, vanadium, tungsten, cobalt, ruthenium, and mixtures thereof. The catalytically active metal may be present as a solid particulate in elemental form or as an organic compound or an inorganic compound such as a sulfide (e.g., molybdenum sulfide) or other ionic compound. Metal or metal compound nanoaggregates may also be used to form the solid particulates.
[0041] A catalyst in the form of a solid particulate is generally a compound of a catalytically active metal, or a metal in elemental form, either alone or supported on a refractory material such as an inorganic metal oxide (e.g., alumina, silica, titania, zirconia, and mixtures thereof). Other suitable refractory materials can include carbon, coal, and clays. Zeolites and non-zeolitic molecular sieves are also useful as solid supports. One advantage of using a support is its ability to act as a "coke getter" or adsorbent of asphaltene precursors that might otherwise lead to fouling of process equipment.
[0042] In some aspects, it can be desirable to form catalyst for slurry hydroprocessing in situ, such as forming catalyst from a metal sulfate (e.g., iron sulfate monohydrate) catalyst precursor or another type of catalyst precursor that decomposes or reacts in the hydroprocessing reaction zone environment, or in a pretreatment step, to form a desired, well-dispersed and catalytically active solid particulate (e.g., as iron sulfide). Precursors also include oil-soluble organometallic compounds containing the catalytically active metal of interest that thermally decompose to form the solid particulate (e.g., iron sulfide) having catalytic activity. Other suitable precursors include metal oxides that may be converted to catalytically active (or more catalytically active) compounds such as metal sulfides. In a particular embodiment, a metal oxide containing mineral may be used as a precursor of a solid particulate comprising the catalytically active metal (e.g., iron sulfide) on an inorganic refractory metal oxide support (e.g., alumina).
[0043] The reaction conditions within the slurry hydroconversion reactors can include the temperatures described above and pressures of about 1200 psig (8.3 MPag) to about 3400 psig (23.4 MPag), e.g. about 1500 psig (10.3 MPag) to about 3000 psig (20.7 MPag). Because the catalyst is in slurry form within the feedstock, the space velocity for a slurry hydroconversion reactor can be characterized based on the volume of feed processed relative to the volume of the reactor used for processing the feed. Suitable space velocities for slurry hydroconversion can range, for example, from about 0.05 v/v/hr.sup.-1 to about 5 v/v/hr.sup.-1, such as about 0.1 v/v/hr.sup.-1 to about 2 v/v/hr.sup.-1.
[0044] The reaction conditions for slurry hydroprocessing can be selected so that the net conversion of feed across all slurry hydroprocessing reactors (if there is more than one arranged in series) is at least about 60%, such as at least about 70%, or at least about 75%. For slurry hydroprocessing, conversion is defined as conversion of compounds with boiling points greater than a conversion temperature, such as 700.degree. F. (371.degree. C.), to compounds with boiling points below the conversion temperature. The portion of a heavy feed that is unconverted after slurry hydroprocessing can be referred to as pitch or a bottoms fraction from the slurry hydroprocessing.
[0045] In some alternative aspects, multiple slurry hydroconversion stages and/or reactors can be used for conversion of a feed. In such aspects, the effluent from a first slurry hydroconversion stage can be fractionated to separate out one or more product fractions. For example, the feed can be fractionated to separate out one or more naphtha fractions and/or distillate fuel (such as diesel) fractions. Such a fractionation can also separate out lower boiling compounds, such as compounds containing 4 carbons or less and contaminant gases such as H.sub.2S or NH.sub.3. The remaining higher boiling fraction of the feed can have a boiling range roughly corresponding to an atmospheric resid, such as a 10 wt % boiling point of at least about 650.degree. F. (343.degree. C.) or at least about 700.degree. F. (371.degree. C.). At least a portion of this higher boiling fraction can be passed into a second (or later) slurry hydroconversion stage for additional conversion of the 975.degree. F.+ (524.degree. C.) portion, or optionally the 1050.degree. F.+ (566.degree. C.) portion of the feed. By separating out the lower boiling portions after performing an intermediate level of conversion, the amount of "overcracking" of desirable products can be reduced or minimized.
[0046] Using multiple stages of slurry hydroconversion reactors can allow for selection of different processing conditions in the stages and/or reactors. As described herein, the temperature in the first slurry hydroconversion reactor can be higher than the temperature in a second reactor. In such an aspect, the first effective hydroprocessing conditions for use in the first slurry hydroconversion reactor can include a temperature that is at least about 5.degree. C. greater than a temperature for the second effective slurry hydroprocessing conditions in the second reactor, or at least about 10.degree. C. greater, or at least about 15.degree. C. greater, or at least about 20.degree. C. greater, or at least about 30.degree. C. greater, or at least about 40.degree. C. greater, or at least about 50.degree. C. greater.
[0047] The benefits of the present disclosure can be seen clearly with respect to the examples.
Example: Multi-Stage Slurry Hydroprocessing with a Lower Temperature Second Stage
[0048] An experiment was conducted documenting the conversion of a 700.degree. F.+ cut of an FCC MCB feed into lighter hydrocarbons as a function of process temperature. The FCC MCB feed had the properties shown in Table 1. The reactions were carried out in an autoclave operated in semi-batch mode with flowing H.sub.2 treat gas at 2100 psig for 3 hours. The autoclave was charged at the beginning of the reaction with the MCB FCC feed and a catalyst slurry comprising MoS.sub.2 blended with heavy oil. The charge weight of catalyst slurry was such that the liquid contents of the autoclave had a Mo concentration of 500 wppm at the start of the reaction. Single stage reactions were performed at 810.degree. F. (432.degree. C.), 830.degree. F. (443.degree. C.), 860.degree. F. (460.degree. C.), and 880.degree. F. (471.degree. C.) for comparative data. A two stage reaction was performed by flowing H.sub.2 treat gas at 2100 psig for 3 hours at a first temperature of 860.degree. F. (460.degree. C.) followed by flowing H.sub.2 treat gas at 2100 psig for 3 hours at a second temperature of 810.degree. F. (432.degree. C.). Each reaction was carried out with a fresh charge of feed and catalyst slurry. All products and unreacted feedstock were extracted from the autoclave after completion of each reaction.
TABLE-US-00001 TABLE 1 FCC MCB Feed Properties Density at 60.degree. F. (g/cm.sup.3) 1.115 API Gravity -4.56 Sulfur (wt %) 3.07 Nitrogen (wt %) 0.18 n-heptane insolubles (wt %) 4.4 hydrogen content (wt %) 7.31 SIMDIST T5 (.degree. F./.degree. C.) 626/330 T50 (.degree. F./.degree. C.) 797/425 T95 (.degree. F./.degree. C.) 1129/609
[0049] FIGS. 3 and 4 provide graphical representation of the results. FIG. 3 provides evidence that the rate of the hydrocracking reaction can be enhanced by operating the slurry reactor at higher temperature. In this series of experiments, higher conversions of the feed were obtained at higher reaction temperatures over the range of 810.degree. F. (432.degree. C.) to 880.degree. F. (471.degree. C.), and notably, the two-stage reaction provides comparable wt. % conversion to the 880.degree. F. (471.degree. C.) case despite the high temperature reaction occurring at 860.degree. F. (460.degree. C.). As described above, however, at high temperatures, thermodynamics favors desaturation of aromatic compounds, which is evidenced by API gravities shown in FIG. 4. As shown, API gravity of the the liquid product obtained from hydrocracking of an FCC MCB feed passes through a maximum as the reaction temperature varies from 810.degree. F. (432.degree. C.) to 880.degree. F. (471.degree. C.). This results from an interplay between kinetics and thermodynamics. As temperature increases, the hydrocracking reaction becomes faster, which leads to a less dense product on account of the molecules having been broken down into smaller molecules to a greater extent. At too high of a temperature (>830.degree. F.), aromatic desaturation begins to dominate, which causes the product API gravity to decrease. An intermediate temperature balances these two competing effects, resulting in the highest product API gravity for a single stage process being attained at about 830.degree. F.
[0050] The two stage configuration exploits these trends with temperature variation to achieve both high boiling point conversion and high product API gravity. The first stage, operated at high temperature, maximizes the boiling point conversion due to the hydrocracking reaction, which is irreversible. The second stage is operated at lower temperature to enable aromatic saturation, a reversible reaction for which equilibrium is sensitive to temperature. The benefit of this strategy is seen in the right most columns in FIGS. 3 and 4. The product API gravity from the two-stage process is higher than that from the single-stage process at intermediate-temperature (830.degree. F.). Additionally, the boiling point conversion from the two-stage process is comparable to that from the single-stage process at high-temperature (880.degree. F.).
ADDITIONAL EMBODIMENTS
Embodiment 1
[0051] A method for processing a heavy oil feedstock, comprising: providing a heavy oil feedstock having a 10% distillation point of at least about 650.degree. F. (343.degree. C.); exposing the heavy oil feedstock to a catalyst under first effective slurry hydroconversion conditions in a first hydroconversion reactor to form a first effluent, the first effective slurry hydroconversion conditions comprising a temperature of about 840.degree. F. (449.degree. C.) to about 1000.degree. F. (538.degree. C.) and a pressure of about 1500 psig to 3000 psig; exposing at least a portion of the first effluent to a catalyst under second effective slurry hydroconversion conditions in a second hydroconversion reactor to form a second effluent; the second effective slurry hydroconversion conditions comprising a temperature of about 750.degree. F. (399.degree. C.) to about 840.degree. F. (449.degree. C.) and a pressure of about 1500 psig to about 3000 psig; wherein the temperature of the second effective slurry hydroconversion conditions is lower than the temperature of the first effective slurry hydroconversion conditions.
Embodiment 2
[0052] The method of embodiment 1, further comprising exposing the first effluent to a separator; wherein the separator removes naphtha and distillate fractions from the first effluent prior to a exposing the first effluent to the second hydroconversion reactor.
Embodiment 3
[0053] The method of embodiment 2, wherein the separator comprises a first separator and a second separator; further comprising exposing the first effluent to the first separator, thereby forming a first separator bottoms fraction and a first separator light fraction; wherein the first separator bottoms fraction comprises 650.degree. F.+ (343.degree. C.+) hydrocarbons and the first separator light fraction comprises 650.degree. F.- (343.degree. C.-) hydrocarbons and treat gas; exposing at least a portion of the first separator bottoms fraction to the second hydroconversion reactor; exposing the first separator light fraction to the second separator, thereby forming a second separator bottoms fraction comprising liquid 160.degree. F.+ (71.degree. C.+) hydrocarbons and a second separator light fraction comprising 160.degree. F.- (71.degree. C.-) hydrocarbons and treat gas; and exposing at least a portion of the second separator light fraction to the second hydroconversion reactor.
Embodiment 4
[0054] The method of embodiment 2, wherein the separator comprises a first separator and a second separator; further comprising exposing the first effluent to the first separator, thereby forming a first separator bottoms fraction comprising liquid 160.degree. F.+ (71.degree. C.+) hydrocarbons and a first separator light fraction comprising 160.degree. F.- (71.degree. C.-) hydrocarbons and treat gas; exposing at least a portion of the first separator light fraction to the second hydroconversion reactor; exposing at least a portion of the first separator bottoms fraction to the second separator; thereby forming a second separator bottoms fraction and a second separator light fraction; wherein the second separator bottoms fraction comprises 650.degree. F.+ (343.degree. C.+) hydrocarbons and the second separator light fraction comprises 650.degree. F.- (343.degree. C.-) hydrocarbons and treat gas; and exposing at least a portion of the second separator bottoms fraction to the second hydroconversion reactor.
Embodiment 5
[0055] The method of any of the previous embodiments, wherein the first effective slurry hydroconversion conditions and the second slurry hydroconversion conditions are effective for a combined conversion of at least about 70 wt % of the heavy oil feedstock relative to a conversion temperature of at least about 700.degree. F. (371.degree. C.).
Embodiment 6
[0056] The method of any of the previous embodiments, wherein the second effluent has an API gravity of at least 12.
Embodiment 7
[0057] The method of any of the previous embodiments, wherein the heavy oil feedstock comprises at least one of fluid catalytic cracker main column bottoms, steam cracker tar, and coker gas oil.
Embodiment 8
[0058] The method of any of the previous embodiments, wherein the catalyst comprises MoS.sub.2.
Embodiment 9
[0059] The method of any of the previous embodiments, wherein the catalyst is present in the heavy oil feedstock at a concentration of 50 wppm to 500 wppm.
* * * * *
D00000

D00001

D00002

D00003

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.