Hydroprocessing Of High Density Cracked Fractions
Deimund; Mark A. ; et al.
U.S. patent application number 16/115775 was filed with the patent office on 2019-03-14 for hydroprocessing of high density cracked fractions. The applicant listed for this patent is ExxonMobil Research and Engineering Company. Invention is credited to Ajit B. Dandekar, Mark A. Deimund, Samia Ilias, Darryl D. Lacy, Randolph J. Smiley, Scott J. Weigel.
| Application Number | 20190078027 16/115775 |
| Document ID | / |
| Family ID | 63684442 |
| Filed Date | 2019-03-14 |








| United States Patent Application | 20190078027 |
| Kind Code | A1 |
| Deimund; Mark A. ; et al. | March 14, 2019 |
HYDROPROCESSING OF HIGH DENSITY CRACKED FRACTIONS
Abstract
Systems and methods are provided for upgrading a heavy cracked feedstock in a single reaction stage under fixed bed hydroprocessing conditions, including exposing the feedstock to a first bulk or supported mixed metal catalyst comprising Ni and Mo; exposing the feedstock to a second bulk or supported mixed metal catalyst comprising Ni and W; and exposing the feedstock to a third catalyst comprising a zeolite-based hydrocracking catalyst.
| Inventors: | Deimund; Mark A.; (Jersey City, NJ) ; Ilias; Samia; (Bridgewater, NJ) ; Smiley; Randolph J.; (Hellertown, PA) ; Dandekar; Ajit B.; (The Woodlands, TX) ; Weigel; Scott J.; (Allentown, PA) ; Lacy; Darryl D.; (Easton, PA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 63684442 | ||||||||||
| Appl. No.: | 16/115775 | ||||||||||
| Filed: | August 29, 2018 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 62555748 | Sep 8, 2017 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C10G 45/50 20130101; C10G 45/52 20130101; C10G 47/02 20130101; C10G 65/12 20130101; C10G 2300/301 20130101; C10G 47/16 20130101; C10G 47/18 20130101; C10G 45/08 20130101; C10G 2300/308 20130101 |
| International Class: | C10G 45/08 20060101 C10G045/08; C10G 45/52 20060101 C10G045/52; C10G 47/18 20060101 C10G047/18 |
Claims
1. A process for upgrading a heavy cracked feedstock, comprising: providing a feedstock comprising a density at 15.degree. C. of 1.06 g/cm.sup.3 or more, at least 50 wt % of one or more 343.degree. C.+ cracked fractions, and a sulfur content of 0.8 to 5.0 wt % sulfur; in a single reaction stage under fixed bed hydroprocessing conditions, exposing the feedstock to a first bulk or supported mixed metal catalyst comprising Ni and Mo; exposing the feedstock to a second bulk or supported mixed metal catalyst comprising Ni and W; and exposing the feedstock to a third catalyst comprising a zeolite-based hydrocracking catalyst.
2. The process of claim 1, wherein the zeolite-based hydrocracking catalyst comprises a Group VIII noble metal.
3. The process of claim 2, wherein the zeolite-based hydrocracking catalyst comprises 0.5 to 2.5 wt % Pt.
4. The process of claim 1, wherein the third catalyst further comprises an aromatic saturation catalyst comprising Pd, Pt, or a combination thereof on an aluminosilicate support.
5. The process of claim 4, wherein the aromatic saturation catalyst comprises Pd and Pt in a wt % ratio of about 3:1 Pd to Pt.
6. The process of claim 4, wherein the aromatic saturation catalyst comprises 2.0 wt % Pt.
7. The process of claim 1, wherein the zeolite exhibits a faujasite (FAU) framework type.
8. The process of claim 1, wherein the feedstock comprises a density at 15.degree. C. of 1.1 g/cm.sup.3 or more, a T.sub.10 of at least 343.degree. C. and a T.sub.90 of at least 475.degree. C.; and a sulfur content of 3.0 wt % to 5.0 wt %.
9. The process of claim 8, wherein the feedstock comprises a de-asphalted heavy cracked feedstock.
10. The process of claim 1, wherein the fixed bed hydroprocessing conditions include a temperature of 300.degree. C. to 400.degree. C., a pressure of 1500 psig to 3000 psig, a hydrogen treat gas rate of 2,000 scf/bbl to 12,000 scf/bbl, and a LHSV of 0.2 h.sup.-1 to 1.5 h.sup.-1.
11. A system for processing a cracked feedstock comprising: a hydroprocessing reactor comprising a hydroprocessing inlet, and a hydroprocessing outlet, and a fixed bed comprising a first bulk or supported mixed metal catalyst comprising Ni and Mo; a second bulk or supported mixed metal catalyst comprising Ni and W; and a third catalyst comprising a zeolite-based hydrocracking catalyst; wherein the hydroprocessing inlet is designed to receive a feedstock comprising a density at 15.degree. C. of 1.06 g/cm.sup.3 or more, at least 50 wt % of one or more 343.degree. C.+ cracked fractions, and a sulfur content of 1.0 to 5.0 wt % sulfur; and wherein the fixed bed is oriented such that the feedstock contacts the first catalyst, second catalyst, and third catalyst sequentially.
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application claims priority to U.S. Provisional Application Ser. No. 62/555,748 filed Sep. 8, 2017, which is herein incorporated by reference in its entirety.
FIELD
[0002] Systems and methods are provided for hydroprocessing of heavy cracked petroleum fractions to form low sulfur hydroprocessed product fractions.
BACKGROUND
[0003] High density cracked fractions are particularly challenging in refining operations. For example, the main column bottoms (MCB, also called catalytic slurry oil or cat tar) of an FCC unit requires significant treatment to convert it to useful products. One product, heavy aromatic fuel oil (HAFO), is mainly MCB, combined with minor amounts of steam cracker tar and coal tar. All three of these components are highly aromatic (65 to 85% of the carbon atoms in these streams are aromatic). Further, MCB has high sulfur content (.about.3 wt %), making such processing challenging. At present, disposal of MCB is feasible as HAFO, but future regulations on sulfur content could make this disposal method infeasible.
[0004] It is desirable to remove sulfur from fuel oil to levels of 0.5 wt % or lower. Thus, there is an incentive to process high-sulfur fuel oil streams (such as MCB) into low sulfur fuel products such as low sulfur fuel oil (LSFO), gasoline, and ultra-low sulfur diesel (ULSD) via hydrotreating or other chemistries. MCB is, however, prone to hydrotreating reactor fouling caused by feed incompatibility. When MCB is blended with virgin crude oil streams, fouling occurs in the hydrotreater, and it ages rapidly and/or plugs the reactor. Thus, separate hydrotreating and other means of upgrading the MCB feed are required to allow for conversion to more useful low sulfur products, as well as to ensure compatibility of blending with other refinery streams.
[0005] It has been discovered that a sequential combination of chemistries can be effective in upgrading this MCB stream into ULSD and LSFO, with minimal naphtha and gas make. This combination of hydrotreating, followed by a hydrocracking catalyst, mixed hydrocracking-arosat catalyst, mixed noble-metal catalyst, or mixtures thereof, has proven effective at converting much of the 700.degree. F.+ material in MCB into ULSD blendstock-range molecules while also greatly reducing API gravity and sulfur and nitrogen content of the stream.
[0006] At present, disposal of MCB is feasible as HAFO, but future regulations on sulfur content could make this disposal method infeasible. Accordingly, hydrotreating and hydrocracking and/or saturating aromatics in the stream to upgrade the material to ULSD blendstock or other useful fuel streams would be quite valuable and preferable. There is a need to dispose of MCB (namely 700.degree. F.+ components), as it can be no longer used as-is for HAFO. Simultaneously reducing the sulfur content and boiling point range can allow for formation of clean fuel streams.
SUMMARY
[0007] In various aspects, systems and methods are provided for upgrading a heavy cracked feedstock. Although most references within this application will be to processes for simplicity, it would be well understood to a person of skill in the art where similar attributes would be equally applicable to systems. The process comprises providing a feedstock comprising a density at 15.degree. C. of 1.06 g/cm.sup.3 or more, at least 50 wt % of one or more 343.degree. C.+ cracked fractions, and a sulfur content of 0.8 to 5.0 wt % sulfur, and, in a single reaction stage under fixed bed hydroprocessing conditions, exposing the feedstock to a first bulk or supported mixed metal catalyst comprising Ni and Mo; exposing the feedstock to a second bulk or supported mixed metal catalyst comprising Ni and W; and exposing the feedstock to a third catalyst comprising a zeolite-based hydrocracking catalyst.
[0008] In certain aspects, the zeolite-based hydrocracking catalyst comprises a Group VIII noble metal such as Pt. The Pt will typically comprises 0.5 to 2.5 wt % of the hydrocracking catalyst. In other aspects, the zeolite exhibits a faujasite (FAU) framework type.
[0009] Additionally or alternatively, the third catalyst may comprise an aromatic saturation catalyst on an aluminosilicate support. The arosat catalyst may include Pd, Pt, or a combination thereof. When the arosat catalyst comprises both Pd and Pt, it would typically do so in a wt % ration of about 3:1 Pd to Pt.
[0010] In yet another aspect, the feedstock is a deasphalted heavy cracked feed. Additionally or alternatively, the fixed bed hydroprocessing conditions include a temperature of 300.degree. C. to 400.degree. C., a pressure of 1500 psig to 3000 psig, a hydrogen treat gas rate of 2,000 scf/bbl to 12,000 scf/bbl, and a LHSV of 0.2 h.sup.-1 to 1.5 h.sup.-1.
[0011] Also provided is a system for processing a cracked feedstock comprising: a hydroprocessing reactor comprising a hydroprocessing inlet, and a hydroprocessing outlet, and a fixed bed comprising a first bulk or supported mixed metal catalyst comprising Ni and Mo; a second bulk or supported mixed metal catalyst comprising Ni and W; and a third catalyst comprising a zeolite-based hydrocracking catalyst; wherein the hydroprocessing inlet is designed to receive a feedstock comprising a density at 15.degree. C. of 1.06 g/cm.sup.3 or more, at least 50 wt % of one or more 343.degree. C.+ cracked fractions, and a sulfur content of 1.0 to 5.0 wt % sulfur; and wherein the fixed bed is oriented such that the feedstock contacts the first catalyst, second catalyst, and third catalyst sequentially.
BRIEF DESCRIPTION OF THE FIGURES
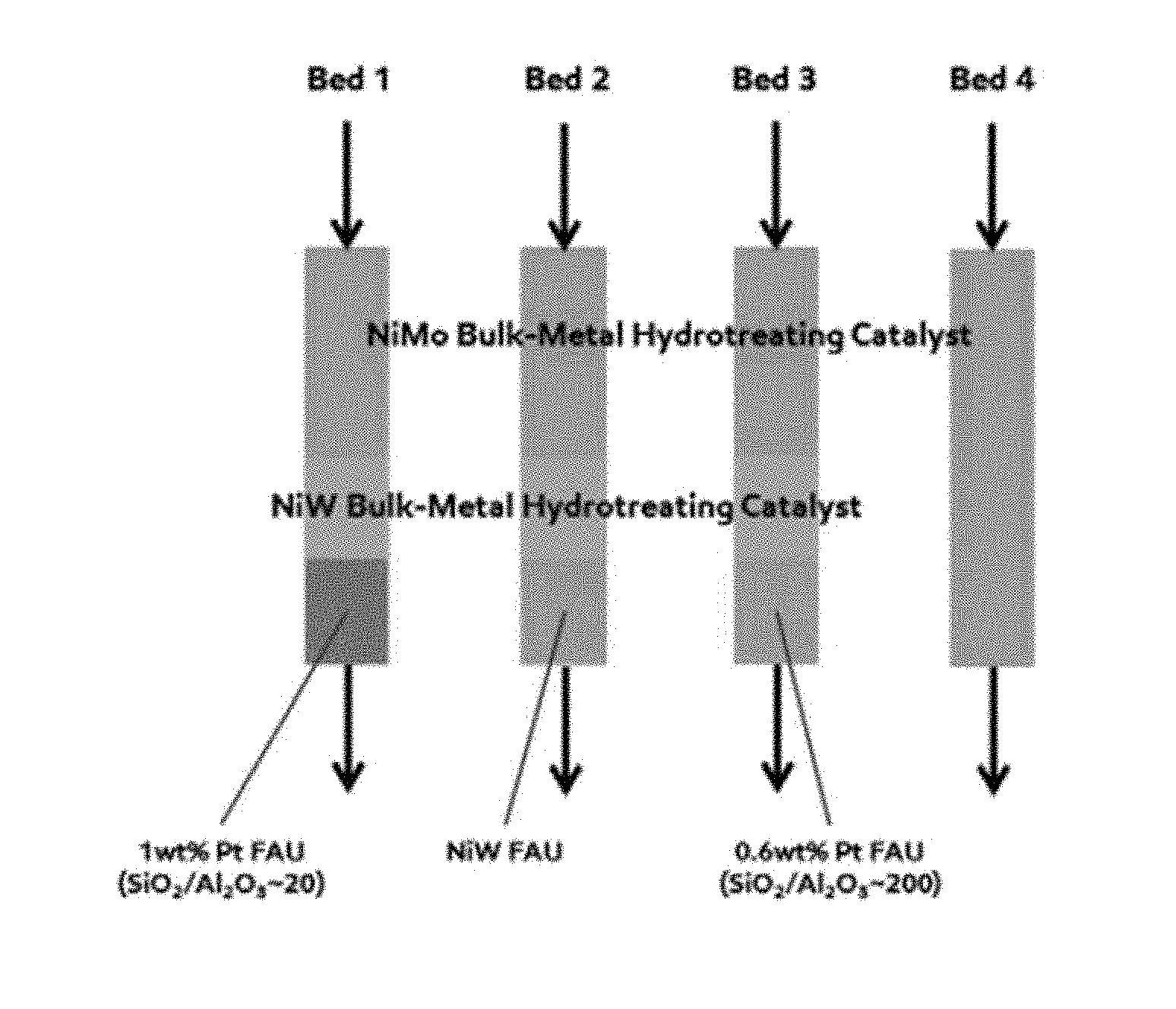
[0012] FIG. 1 depicts catalyst bed configurations according the present disclosure and those used in Examples 1 and 2.
[0013] FIGS. 2A-2F provide graphical data obtained in Example 1.
[0014] FIGS. 3A-3F provide graphical data obtained in Example 2.
[0015] FIG. 4 depicts catalyst bed configurations according the present disclosure and those used in Example 3.
[0016] FIGS. 5A-5F provide graphical data obtained in Example 3.
[0017] FIG. 6 depicts catalyst bed configurations according the present disclosure and those used in Examples 4 and 5.
[0018] FIGS. 7A-7F provide graphical data obtained in Example 4.
[0019] FIGS. 8A-8F provide graphical data obtained in Example 5.
DETAILED DESCRIPTION
[0020] In various aspects, systems and methods are provided for upgrading a high density cracked feedstock, such as a catalytic slurry oil, by hydroprocessing. In this discussion, reference may be made to catalytic slurry oil, FCC bottoms, and main column bottoms. These terms can be used interchangeably herein. Difficulties in processing heavy cracked feeds can be related to difficulties in performing distillation on the feeds. Conventionally, one of the strategies for processing a challenging feedstock can be to use distillation to separate a more favorable portion of a feed from a typically higher boiling less favorable portion. Under such a conventional strategy, an atmospheric distillation can be used to separate a feed into lower boiling portions and a higher boiling portion at a distillation cut point between about 600.degree. F. (.about.316.degree. C.) and about 700.degree. F. (.about.371.degree. C.). The higher boiling portion can then correspond to a roughly 316.degree. C.+ portion, or a roughly 343.degree. C.+ portion, or a roughly 371.degree. C.+ portion. Conventionally, a further distillation can be performed on this higher boiling portion under reduced pressure or vacuum distillation conditions. This can produce one or more vacuum distillate fractions and a bottoms fraction. Unfortunately, heavy cracked feeds such as catalytic slurry oils can often have a density of about 1.04 g/cm.sup.3 or more, or about 1.06 g/cm.sup.3 or more, or about 1.08 g/cm.sup.3 or more, such as up to 1.14 g/cm.sup.3 or possibly still higher. At such higher density values, performing a vacuum distillation under conventional vacuum distillation conditions becomes increasingly difficult and/or inefficient. In particular, such high density fractions can tend to have poor separation characteristics under conventional vacuum distillation conditions. As a result, either substantial amounts of undesirable components can remain in the "desired" distillate fraction(s), and/or substantial amounts of the desired components can remain in the bottoms fraction.
[0021] It has been unexpectedly discovered that heavy cracked fractions, such as catalytic slurry oils, can be hydroprocessed with reduced or minimized amounts of coking by exposing the fraction, in sequence, to a first mixed metal catalyst comprising Ni and Mo, a second mixed metal catalyst comprising Ni and W, and a third catalyst comprising a zeolite-based hydrocracking catalyst in a single reaction stage. In first and second catalysts may be bulk metal or supported catalysts. As used herein, the term "single reaction stage" means that no intermediate separation is performed between exposing the feed to the catalysts. In other words, the reactions described herein may take place in a single reactor or multiple reactors. So long as no intermediate separation is performed between exposures to the different catalysts, then it can be said that the process takes place in a single reaction stage.
[0022] This third zeolite-based hydrocracking catalyst can be any number of commercially available hydrocracking catalysts. Additionally or alternatively, the third catalyst can include an aromatic saturation ("arosat") catalyst. Additionally or alternatively, the third catalyst can include a noble metal. Use of a zeolite-based hydrocracking catalyst with additional aromatic saturation and/or noble metal catalyst would conventionally be considered undesirable with feeds having high aromatic, nitrogen, and sulfur content because of rapid poisoning of the catalyst. Particularly, the organosulfur and organonitrogen species present in the feed are thought to interact with noble metals and acid sites on hydrocracking, arosat, and/or noble-metal catalysts, respectively, causing this catalyst poisoning and deactivation. It is conventionally believed widely in the industry that hydrogen sulfide and ammonia are also involved. Thus, these types of catalysts are generally not employed industrially in single-stage and/or sour service operations (in the presence of high concentrations of S- and N-containing species). In the present disclosure, the initial hydrotreating catalysts are present to help convert these feed organosulfur and organonitrogen species into hydrogen sulfide and ammonia, respectively, before reaching the third catalyst bed. Surprisingly, this also translates to less deactivation and poisoning of the hydrocracking, arosat, and/or noble-metal catalysts, enabling single-stage sour service operation for catalysts in the third bed. It is believed here that the much smaller hydrogen sulfide and ammonia molecules are less preferentially adsorbed onto catalytic sites in the hydrocracking, arosat, and/or noble-metal catalysts than the larger organosulfur and organonitrogen species, and thus, they cause reduced catalytic poisoning. This discovery enables cost and complexity savings due to the lack of a requirement for separations of hydrogen sulfide and ammonia from streams between catalyst beds, as well as any additional feed treatments, in industrial processes. In this application, the first hydrotreating catalyst bed (Ni and Mo, in this instance) is intended to do hydrotreating on the "easier" S- and N-containing species to reduce the organosulfur and organonitrogen content dramatically from the levels found in the feed. Use of a second intermediate bed of higher activity hydrotreating catalyst (such as a Ni and W containing material) can further reduce organic S- and N-content to ppm levels by converting the more refractory molecules that are thought to act as stronger catalyst poisons. At such low levels, the organosulfur and organonitrogen species cause significantly less poisoning and deactivation relative to their feed levels, and furthermore the hydrogen sulfide and ammonia present surprisingly do not appear to have any significant effect on catalyst activity in the third bed containing hydrocracking, arosat, and/or noble-metal catalysts.
[0023] In some aspects, reference may be made to conversion of a feedstock relative to a conversion temperature. Conversion relative to a temperature can be defined based on the portion of the feedstock that boils at greater than the conversion temperature. The amount of conversion during a process (or optionally across multiple processes) can correspond to the weight percentage of the feedstock converted from boiling above the conversion temperature to boiling below the conversion temperature. As an illustrative hypothetical example, consider a feedstock that includes 40 wt % of components that boil at 700.degree. F. (.about.371.degree. C.) or greater. By definition, the remaining 60 wt % of the feedstock boils at less than 700.degree. F. (.about.371.degree. C.). For such a feedstock, the amount of conversion relative to a conversion temperature of .about.371.degree. C. would be based only on the 40 wt % that initially boils at .about.371.degree. C. or greater. If such a feedstock could be exposed to a process with 30% conversion relative to a .about.371.degree. C. conversion temperature, the resulting product would include 72 wt % of .about.371.degree. C.- components and 28 wt % of .about.371.degree. C.+ components.
[0024] In various aspects, reference may be made to one or more types of fractions generated during distillation of a feedstock or effluent. Such fractions may include naphtha fractions, kerosene fractions, diesel fractions, and other heavier (gas oil) fractions. Each of these types of fractions can be defined based on a boiling range, such as a boiling range that includes at least .about.90 wt % of the fraction, or at least .about.95 wt % of the fraction. For example, for many types of naphtha fractions, at least .about.90 wt % of the fraction, or at least .about.95 wt %, can have a boiling point in the range of .about.85.degree. F. (.about.29.degree. C.) to .about.350.degree. F. (.about.177.degree. C.). For some heavier naphtha fractions, at least .about.90 wt % of the fraction, and preferably at least .about.95 wt %, can have a boiling point in the range of .about.85.degree. F. (.about.29.degree. C.) to .about.400.degree. F. (.about.204.degree. C.). For a kerosene fraction, at least .about.90 wt % of the fraction, or at least .about.95 wt %, can have a boiling point in the range of .about.300.degree. F. (.about.149.degree. C.) to .about.600.degree. F. (.about.288.degree. C.). For a kerosene fraction targeted for some uses, such as jet fuel production, at least .about.90 wt % of the fraction, or at least .about.95 wt %, can have a boiling point in the range of .about.300.degree. F. (.about.149.degree. C.) to .about.550.degree. F. (.about.288.degree. C.). For a diesel fraction, at least .about.90 wt % of the fraction, and preferably at least .about.95 wt %, can have a boiling point in the range of .about.350.degree. F. (.about.177.degree. C.) to .about.700.degree. F. (.about.371.degree. C.). For a (vacuum) gas oil fraction, at least .about.90 wt % of the fraction, and preferably at least .about.95 wt %, can have a boiling point in the range of .about.650.degree. F. (.about.343.degree. C.) to .about.1100.degree. F. (.about.593.degree. C.). Optionally, for some gas oil fractions, a narrower boiling range may be desirable. For such gas oil fractions, at least .about.90 wt % of the fraction, or at least .about.95 wt %, can have a boiling point in the range of .about.650.degree. F. (.about.343.degree. C.) to .about.1000.degree. F. (.about.538.degree. C.), or .about.650.degree. F. (.about.343.degree. C.) to .about.900.degree. F. (.about.482.degree. C.). A residual fuel product can have a boiling range that may vary and/or overlap with one or more of the above boiling ranges. A residual marine fuel product can satisfy the requirements specified in ISO 8217, Table 2.
[0025] In this discussion, a low sulfur fuel oil can correspond to a fuel oil containing about 0.5 wt % or less of sulfur. An ultra low sulfur fuel oil, which can also be referred to as an Emission Control Area fuel, can correspond to a fuel oil containing about 0.1 wt % or less of sulfur. A low sulfur diesel can correspond to a diesel fuel containing about 500 wppm or less of sulfur. An ultra low sulfur diesel can correspond to a diesel fuel containing about 15 wppm or less of sulfur, or about 10 wppm or less.
[0026] It is noted that when initially formed, a catalytic slurry oil can include several weight percent of catalyst fines. Any such catalyst fines can be removed prior to incorporating a fraction derived from a catalytic slurry oil into a product pool, such as a naphtha fuel pool or a diesel fuel pool. In this discussion, unless otherwise explicitly noted, references to a catalytic slurry oil are defined to include catalytic slurry oil either prior to or after such a process for reducing the content of catalyst fines within the catalytic slurry oil.
Feedstocks for Hydroprocessing--Cracked Fractions
[0027] A catalytic slurry oil is an example of a suitable cracked fraction for incorporation into a feedstock. It is conventionally understood that conversion of .about.1050.degree. F.+(.about.566.degree. C.+) vacuum resid fractions by hydroprocessing and/or hydrocracking can be limited by incompatibility. Under conventional understanding, at somewhere between .about.30 wt % and .about.55 wt % conversion of the .about.1050.degree. F.+(.about.566.degree. C.+) portion, the reaction product during hydroprocessing can become incompatible with the feed. For example, as the .about.566.degree. C.+ feedstock converts to .about.1050.degree. F.- (.about.566.degree. C.-) products, hydrogen transfer, oligomerization, and dealkylation reactions can occur which create molecules that are increasingly difficult to keep in solution. Somewhere between .about.30 wt % and .about.55 wt % .about.566.degree. C.+ conversion, a second liquid hydrocarbon phase separates. This new incompatible phase, under conventional understanding, can correspond to mostly polynuclear aromatics rich in N, S, and metals. The new incompatible phase can potentially be high in micro carbon residue (MCR). The new incompatible phase can stick to surfaces in the unit where it cokes and then can foul the equipment. Based on this conventional understanding, catalytic slurry oil can conventionally be expected to exhibit properties similar to a vacuum resid fraction during hydroprocessing. Based on the above conventional understanding, it can be expected that hydroprocessing of a catalytic slurry oil would cause incompatibility as the asphaltenes and/or .about.566.degree. C.+ material converts.
[0028] In contrast to conventional understanding, it has been discovered that hydroprocessing can be performed while reducing or minimizing the above difficulties by exposing the heavy cracked feed, in sequence, to a first mixed metal catalyst comprising Ni and Mo, a second mixed metal catalyst comprising Ni and W, and a third catalyst comprising a zeolite-based hydrocracking catalyst in a single reaction stage. A heavy cracked feed can be processed as part of a feed where the heavy cracked feed corresponds to at least about 25 wt % of the feed to a process for forming fuels, such as at least about 50 wt %, at least about 75 wt %, at least about 90 wt %, or at least about 95 wt %. Optionally, the feed can correspond to at least about 99 wt % of a heavy cracked feed, therefore corresponding to a feed that consists essentially of heavy cracked feed. In particular, a feed can comprise about 25 wt % to about 100 wt % heavy cracked feed, or about 25 wt % to about 99 wt %, or about 50 wt % to about 90 wt %. It has been unexpectedly discovered that this sequential exposure of the heavy cracked feed to the catalysts at effective hydroprocessing conditions results in substantial conversion of the feed without causing excessive coking of the catalyst or differential pressure build across the hydroprocessing reactor, which would be indicative of asphaltene precipitation.
[0029] Typically the cut point for forming a heavy cracked feed can be at least about 650.degree. F. (.about.343.degree. C.). As a result, a heavy cracked feed can have a T5 distillation (boiling) point or a T10 distillation point of at least about 288.degree. C., or at least about 316.degree. C., or at least about 650.degree. F. (.about.343.degree. C.), as measured according to ASTM D2887. In some aspects the D2887 10% distillation point (T10) can be greater, such as at least about 675.degree. F. (.about.357.degree. C.), or at least about 700.degree. F. (.about.371.degree. C.). In some aspects, a broader boiling range portion of FCC products can be used as a feed (e.g., a 350.degree. F.+/.about.177.degree. C.+ boiling range fraction of FCC liquid product), where the broader boiling range portion includes a 650.degree. F.+(.about.343.degree. C.+) fraction that corresponds to a heavy cracked feed. The heavy cracked feed (650.degree. F.+/.about.343.degree. C.+) fraction of the feed does not necessarily have to represent a "bottoms" fraction from an FCC process, so long as the heavy cracked feed portion comprises one or more of the other feed characteristics described herein.
[0030] In addition to and/or as an alternative to initial boiling points, T5 distillation point, and/or T10 distillation points, other distillation points may be useful in characterizing a feedstock. For example, a feedstock can be characterized based on the portion of the feedstock that boils above 1050.degree. F. (.about.566.degree. C.). In some aspects, a feedstock (or alternatively a 650.degree. F.+/.about.343.degree. C.+ portion of a feedstock) can have an ASTM D2887 T95 distillation point of 1050.degree. F. (.about.566.degree. C.) or greater, or a T90 distillation point of 1050.degree. F. (.about.566.degree. C.) or greater. If a feedstock or other sample contains components that are not suitable for characterization using D2887, ASTM D1160 may be used instead for such components.
[0031] In various aspects, density, or weight per volume, of the heavy cracked feed can be characterized. The density of the heavy cracked feed (or alternatively a 650.degree. F.+/.about.343.degree. C.+ portion of a feedstock) can be at least about 1.06 g/cc, or at least about 1.08 g/cc, or at least about 1.10 g/cc, such as up to about 1.20 g/cc. The density of the heavy cracked feed can provide an indication of the amount of heavy aromatic cores that are present within the heavy cracked feed.
[0032] Contaminants such as organic nitrogen and organic sulfur are typically found in heavy cracked feeds, often in organically-bound form. Nitrogen content can range from about 50 wppm to about 5000 wppm elemental nitrogen, or about 100 wppm to about 2000 wppm elemental nitrogen, or about 250 wppm to about 1000 wppm, based on total weight of the heavy cracked feed. The nitrogen containing compounds can be present as basic or non-basic nitrogen species. Examples of nitrogen species can include quinolones, substituted quinolones, carbazoles, and substituted carbazoles.
[0033] The sulfur content of a heavy cracked feed can be at least about 500 wppm elemental sulfur, based on total weight of the heavy cracked feed. Generally, the sulfur content of a heavy cracked feed can range from about 500 wppm to about 100,000 wppm elemental sulfur, or from about 1000 wppm to about 50,000 wppm, or from about 1000 wppm to about 30,000 wppm, based on total weight of the heavy component. Sulfur may also be expressed as weight percent and can range from about 0.5 wt % to about 6 wt %, or from about 1 wt % to about 5 wt %, or about 2 wt % to about 4 wt %. Sulfur can usually be present as organically bound sulfur. Examples of such sulfur compounds include the class of heterocyclic sulfur compounds such as thiophenes, tetrahydrothiophenes, benzothiophenes and their higher homologs and analogs. Other organically bound sulfur compounds include aliphatic, naphthenic, and aromatic mercaptans, sulfides, di- and polysulfides.
[0034] A favorable feature of hydroprocessing a heavy cracked feed can be the increase in product volume that can be achieved. Due to the high percentage of aromatic cores in a heavy cracked feed, hydroprocessing of heavy cracked feed can result in substantial consumption of hydrogen. The additional hydrogen added to a heavy cracked feed can result in an increase in volume for the hydroprocessed heavy cracked feed or volume swell. For example, the amount of C.sub.3+ liquid products generated from hydrotreatment and FCC processing of catalytic slurry oil can be greater than .about.100% of the volume of the initial catalytic slurry oil. (A similar proportional increase in volume can be achieved for feeds that include only a portion of deasphalted catalytic slurry oil.) Hydroprocessing within the normal range of commercial hydrotreater operations can enable .about.2000-4000 SCF/bbl (.about.340 Nm.sup.3/m.sup.3 to .about.680 m.sup.3/m.sup.3) of hydrogen to be added to a feed corresponding to a deasphalted heavy cracked feed. This can result in substantial conversion of a deasphalted heavy cracked feed to .about.700.degree. F.- (.about.371.degree. C.-) products, such as at least about 40 wt % conversion to .about.371.degree. C.- products, or at least about 50 wt %, or at least about 60 wt %, and up to about 90 wt % or more. In some aspects, the .about.371.degree. C.- product can meet the requirements for a low sulfur diesel fuel blendstock in the U.S. Additionally or alternately, the .about.371.degree. C.- product(s) can be upgraded by further hydroprocessing to a low sulfur diesel fuel or blendstock. The remaining .about.700.degree. F.+(.about.371.degree. C.+) product can meet the normal specifications for a <.about.0.5 wt % S bunker fuel or a <.about.0.1 wt % S bunker fuel, and/or may be blended with a distillate range blendstock to produce a finished blend that can meet the specifications for a <.about.0.1 wt % S bunker fuel. Additionally or alternately, a .about.343.degree. C.+ product can be formed that can be suitable for use as a <.about.0.1 wt % S bunker fuel without additional blending. The additional hydrogen for the hydrotreatment of the heavy cracked feed can be provided from any convenient source.
[0035] Additionally or alternately, the remaining .about.371.degree. C.+ product (and/or portions of the .about.371.degree. C.+ product) can be used as feedstock to an FCC unit and cracked to generate additional LPG, gasoline, and diesel fuel, so that the yield of .about.371.degree. C.- products relative to the total liquid product yield can be at least about 60 wt %, or at least about 70 wt %, or at least about 80 wt %. Relative to the feed, the yield of C.sub.3+ liquid products can be at least about 100 vol %, such as at least about 105 vol %, at least about 110 vol %, at least about 115 vol %, or at least about 120 vol %. In particular, the yield of C.sub.3+ liquid products can be about 100 vol % to about 150 vol %, or about 110 vol % to about 150 vol %, or about 120 vol % to about 150 vol %.
[0036] More generally, the systems and methods described herein can be used for processing feedstocks containing one or more types of cracked feeds that have a high density prior to hydroprocessing, such as a density of 1.04 g/cm.sup.3 or more, or 1.06 g/cm.sup.3 or more, or 1.08 g/cm.sup.3 or more, such as up to 1.20 g/cm.sup.3 or possibly still higher. Additionally or alternately, the feedstock including one or more cracked feeds can have an aromatics content of about 40 wt % to about 80 wt %, or about 40 wt % to about 70 wt %, or about 50 wt % to about 80 wt %. In addition to catalytic slurry oils, other types of cracked stocks include, but are not limited to, heavy coker gas oils (such coker bottoms), steam cracker tars, coal tars, and visbreaker gas oils.
[0037] For example, steam cracker tar (SCT) as used herein is also referred to in the art as "pyrolysis fuel oil". The terms can be used interchangeably herein. The tar will typically be obtained from the first fractionator downstream from a steam cracker (pyrolysis furnace) as the bottoms product of the fractionator, nominally having a boiling point of at least about 550.degree. F.+(.about.288.degree. C.+). Boiling points and/or fractional weight distillation points can be determined by, for example, ASTM D2892. Alternatively, SCT can have a T5 boiling point (temperature at which 5 wt % will boil off) of at least about 550.degree. F. (.about.288.degree. C.). The final boiling point of SCT can be dependent on the nature of the initial pyrolysis feed and/or the pyrolysis conditions, and typically can be about 1450.degree. F. (.about.788.degree. C.) or less.
[0038] SCT can have a relatively low hydrogen content compared to heavy oil fractions that are typically processed in a refinery setting. In some aspects, SCT can have a hydrogen content of about 8.0 wt % or less, about 7.5 wt % or less, or about 7.0 wt % or less, or about 6.5 wt % or less. In particular, SCT can have a hydrogen content of about 5.5 wt % to about 8.0 wt %, or about 6.0 wt % to about 7.5 wt %. Additionally or alternately, SCT can have a micro carbon residue (or alternatively Conradson Carbon Residue) of at least about 10 wt %, or at least about 15 wt %, or at least about 20 wt %, such as up to about 40 wt % or more.
[0039] SCT can also be highly aromatic in nature. The paraffin content of SCT can be about 2.0 wt % or less, or about 1.0 wt % or less, such as having substantially no paraffin content. The naphthene content of SCT can also be about 2.0 wt % or less or about 1.0 wt % or less, such as having substantially no naphthene content. In some aspects, the combined paraffin and naphthene content of SCT can be about 1.0 wt % or less. With regard to aromatics, at least about 30 wt % of SCT can correspond to 3-ring aromatics, or at least 40 wt %. In particular, the 3-ring aromatics content can be about 30 wt % to about 60 wt %, or about 40 wt % to about 55 wt %, or about 40 wt % to about 50 wt %. Additionally or alternately, at least about 30 wt % of SCT can correspond to 4-ring aromatics, or at least 40 wt %. In particular, the 4-ring aromatics content can be about 30 wt % to about 60 wt %, or about 40 wt % to about 55 wt %, or about 40 wt % to about 50 wt %. Additionally or alternately, the 1-ring aromatic content can be about 15 wt % or less, or about 10 wt % or less, or about 5 wt % or less, such as down to about 0.1 wt %.
[0040] SCT can also have a higher density than many types of crude or refinery fractions. In various aspects, SCT can have a density at 15.degree. C. of about 1.08 g/cm.sup.3 to about 1.20 g/cm.sup.3, or 1.10 g/cm.sup.3 to 1.18 g/cm.sup.3. By contrast, many types of vacuum resid fractions can have a density of about 1.05 g/cm.sup.3 or less. Additionally or alternately, density (or weight per volume) of the heavy hydrocarbon can be determined according to ASTM D287-92 (2006) Standard Test Method for API Gravity of Crude Petroleum and Petroleum Products (Hydrometer Method), which characterizes density in terms of API gravity. In general, the higher the API gravity, the less dense the oil. API gravity can be 5.degree. or less, or 0.degree. or less, such as down to about -10.degree. or lower.
[0041] Contaminants such as nitrogen and sulfur are typically found in SCT, often in organically-bound form. Nitrogen content can range from about 50 wppm to about 10,000 wppm elemental nitrogen or more, based on total weight of the SCT. Sulfur content can range from about 0.1 wt % to about 10 wt %, based on total weight of the SCT.
[0042] Coker bottoms represent another type of cracked feed suitable for hydroprocessing, optionally in combination with a catalytic slurry oil and/or steam cracker tar and/or other cracked fractions. Coking is a thermal cracking process that is suitable for conversion of heavy feeds into fuels boiling range products. The feedstock to a coker typically also includes 5 wt % to 25 wt % recycled product from the coker, which can be referred to as coker bottoms. This recycle fraction allows metals, asphaltenes, micro-carbon residue, and/or other solids to be returned to the coker, as opposed to being incorporated into a coker gas oil product. This can maintain a desired product quality for the coker gas oil product, but results in a net increase in the amount of light ends and coke that are generated by a coking process. The coker bottoms can correspond to a fraction with a T10 distillation point of at least 550.degree. F. (288.degree. C.), or at least 300.degree. C., or at least 316.degree. C., and a T90 distillation point of 566.degree. C. or less, or 550.degree. C. or less, or 538.degree. C. or less. The coker recycle fraction can have an aromatic carbon content of about 20 wt % to about 50 wt %, or about 30 wt % to about 45 wt %, and a micro carbon residue content of about 4.0 wt % to about 15 wt %, or about 6.0 wt % to about 15 wt %, or about 4.0 wt % to about 10 wt %, or about 6.0 wt % to about 12 wt %.
Additional Feedstocks
[0043] In some aspects, at least a portion of a feedstock for processing as described herein can correspond to a vacuum resid fraction or another type 950.degree. F.+(510.degree. C.+) or 1000.degree. F.+(538.degree. C.+) fraction. Another example of a method for forming a 950.degree. F.+(510.degree. C.+) or 1000.degree. F.+(538.degree. C.+) fraction is to perform a high temperature flash separation. The 950.degree. F.+(510.degree. C.+) or 1000.degree. F.+(538.degree. C.+) fraction formed from the high temperature flash can be processed in a manner similar to a vacuum resid.
[0044] A vacuum resid fraction or a 950.degree. F.+(510.degree. C.+) fraction formed by another process (such as a flash fractionation bottoms or a bitumen fraction) can be deasphalted at low severity to form a deasphalted oil. Optionally, the feedstock can also include a portion of a conventional feed for lubricant base stock production, such as a vacuum gas oil.
[0045] A vacuum resid (or other 510.degree. C.+) fraction can correspond to a fraction with a T5 distillation point (ASTM D2892, or ASTM D7169 if the fraction will not completely elute from a chromatographic system) of at least about 900.degree. F. (482.degree. C.), or at least 950.degree. F. (510.degree. C.), or at least 1000.degree. F. (538.degree. C.). Alternatively, a vacuum resid fraction can be characterized based on a T10 distillation point (ASTM D2892/D7169) of at least about 900.degree. F. (482.degree. C.), or at least 950.degree. F. (510.degree. C.), or at least 1000.degree. F. (538.degree. C.).
[0046] Resid (or other 510.degree. C.+) fractions can be high in metals. For example, a resid fraction can be high in total nickel, vanadium and iron contents. In an aspect, a resid fraction can contain at least 0.00005 grams of Ni/V/Fe (50 wppm) or at least 0.0002 grams of Ni/V/Fe (200 wppm) per gram of resid, on a total elemental basis of nickel, vanadium and iron. In other aspects, the heavy oil can contain at least 500 wppm of nickel, vanadium, and iron, such as up to 1000 wppm or more.
[0047] Contaminants such as nitrogen and sulfur are typically found in resid (or other 510.degree. C.+) fractions, often in organically-bound form. Nitrogen content can range from about 50 wppm to about 10,000 wppm elemental nitrogen or more, based on total weight of the resid fraction. Sulfur content can range from 500 wppm to 100,000 wppm elemental sulfur or more, based on total weight of the resid fraction, or from 1000 wppm to 50,000 wppm, or from 1000 wppm to 30,000 wppm.
[0048] Still another method for characterizing a resid (or other 510.degree. C.+) fraction is based on the Conradson carbon residue (CCR) of the feedstock. The Conradson carbon residue of a resid fraction can be at least about 5 wt %, such as at least about 10 wt % or at least about 20 wt %. Additionally or alternately, the Conradson carbon residue of a resid fraction can be about 50 wt % or less, such as about 40 wt % or less or about 30 wt % or less.
Hydroprocessing of Feedstock Including One or More Cracked Fractions
[0049] A feedstock including one or more cracked fractions can be hydroprocessed to form a hydroprocessed effluent. This can include hydrotreatment and/or hydrocracking to remove heteroatoms (such as sulfur and/or nitrogen) to desired levels, reduce Conradson Carbon content, and/or provide viscosity index (VI) uplift. Additionally or alternately, the hydroprocessing can be performed to achieve a desired level of conversion of higher boiling compounds in the feed to fuels boiling range compounds. Depending on the aspect, a feedstock can be hydroprocessed by demetallization, aromatics saturation, hydrotreating, hydrocracking, or a combination thereof.
[0050] In various aspects, the aromatics content of the feedstock can be at least 50 wt %, or at least 55 wt %, or at least 60 wt %, or at least 65 wt %, or at least 70 wt %, or at least 75 wt %, such as up to 90 wt % or more. Additionally or alternately, the saturates content of the feedstock can be 50 wt % or less, or 45 wt % or less, or 40 wt % or less, or 35 wt % or less, or 30 wt % or less, or 25 wt % or less, such as down to 10 wt % or less. In this discussion and the claims below, the aromatics content and/or the saturates content of a fraction can be determined based on ASTM D7419.
[0051] Depending on the aspect, the hydroprocessing can be performed in a configuration including a single reaction stage. The reaction conditions during demetallization and/or hydrotreatment and/or hydrocracking of the feedstock can be selected to generate a desired level of conversion of a feed. Any convenient type of reactor, such as fixed bed (for example trickle bed) reactors can be used. Conversion of the feed can be defined in terms of conversion of molecules that boil above a temperature threshold to molecules below that threshold. The conversion temperature can be any convenient temperature, such as .about.700.degree. F. (371.degree. C.) or 1050.degree. F. (566.degree. C.). The amount of conversion can correspond to the total conversion of molecules within the combined hydrotreatment and hydrocracking stages. Suitable amounts of conversion of molecules boiling above 1050.degree. F. (566.degree. C.) to molecules boiling below 566.degree. C. include 30 wt % to 100 wt % conversion relative to 566.degree. C., or 30 wt % to 90 wt %, or 30 wt % to 70 wt %, or 40 wt % to 90 wt %, or 40 wt % to 80 wt %, or 40 wt % to 70 wt %, or 50 wt % to 100 wt %, or 50 wt % to 90 wt %, or 50 wt % to 70 wt %. In particular, the amount of conversion relative to 566.degree. C. can be 30 wt % to 100 wt %, or 50 wt % to 100 wt %, or 40 wt % to 90 wt %. Additionally or alternately, suitable amounts of conversion of molecules boiling above .about.700.degree. F. (371.degree. C.) to molecules boiling below 371.degree. C. include 10 wt % to 70 wt % conversion relative to 371.degree. C., or 10 wt % to 60 wt %, or 10 wt % to 50 wt %, or 20 wt % to 70 wt %, or 20 wt % to 60 wt %, or 20 wt % to 50 wt %, or 30 wt % to 70 wt %, or 30 wt % to 60 wt %, or 30 wt % to 50 wt %. In particular, the amount of conversion relative to 371.degree. C. can be 10 wt % to 70 wt %, or 20 wt % to 50 wt %, or 30 wt % to 60 wt %.
[0052] The hydroprocessed effluent can also be characterized based on the product quality. After hydroprocessing (hydrotreating and/or hydrocracking), the liquid (C.sub.3+) portion of the hydroprocessed deasphalted oil/hydroprocessed effluent can have a sulfur content of about 1000 wppm or less, or about 500 wppm or less, or about 100 wppm or less (such as down to .about.0 wppm). Additionally or alternately, the hydroprocessed deasphalted oil/hydroprocessed effluent can have a nitrogen content of 200 wppm or less, or 100 wppm or less, or 50 wppm or less (such as down to .about.0 wppm). Additionally or alternately, the liquid (C.sub.3+) portion of the hydroprocessed deasphalted oil/hydroprocessed effluent can have a MCR content and/or Conradson Carbon residue content of 2.5 wt % or less, or 1.5 wt % or less, or 1.0 wt % or less, or 0.7 wt % or less, or 0.1 wt % or less, or 0.02 wt % or less (such as down to .about.0 wt %). MCR content and/or Conradson Carbon residue content can be determined according to ASTM D4530.
[0053] In some aspects, the portion of the hydroprocessed effluent having a boiling range/distillation point of less than about 700.degree. F. (.about.371.degree. C.) can be used as a low sulfur fuel oil or blendstock for low sulfur fuel oil. In other aspects, such a portion of the hydroprocessed effluent can be used (optionally with other distillate streams) to form ultra-low sulfur naphtha and/or distillate (such as diesel) fuel products, such as ultra-low sulfur fuels or blendstocks for ultra-low sulfur fuels. The portion having a boiling range/distillation point of at least about 700.degree. F. (.about.371.degree. C.) can be used as an ultra-low sulfur fuel oil having a sulfur content of about 0.1 wt % or less or optionally blended with other distillate or fuel oil streams to form an ultra-low sulfur fuel oil or a low sulfur fuel oil. In some aspects, at least a portion of the liquid hydrotreated effluent having a distillation point of at least about .about.371.degree. C. can be used as a feed for FCC processing. In still other aspects, the portion having a boiling range/distillation point of at least about 371.degree. C. can be used as a feedstock for lubricant base oil production.
[0054] In various aspects, the feedstock can be exposed to uniquely oriented stacked beds of hydrotreating catalyst under effective hydrotreating conditions in a single reaction stage. The catalysts used can include conventional hydroprocessing catalysts, such as those comprising at least one Group VIII non-noble metal (Columns 8-10 of IUPAC periodic table), preferably Fe, Co, and/or Ni, such as Co and/or Ni; and at least one Group VI metal (Column 6 of IUPAC periodic table), preferably Mo and/or W. Such hydroprocessing catalysts optionally include transition metal sulfides that are impregnated or dispersed on a refractory support or carrier such as alumina and/or silica. The support or carrier itself typically has no significant/measurable catalytic activity. Substantially carrier- or support-free catalysts, commonly referred to as bulk catalysts, generally have higher volumetric activities than their supported counterparts. In a preferred embodiment, the heavy cracked feed is exposed in order to a first bulk or supported mixed metal catalyst comprising Ni and Mo, a second bulk or supported mixed metal catalyst comprising Ni and W, and a third catalyst comprising a zeolite-based hydrocracking catalyst.
[0055] The catalysts can either be in bulk form or in supported form. In addition to alumina and/or silica, other suitable support/carrier materials can include, but are not limited to, zeolites, titania, silica-titania, and titania-alumina. Suitable aluminas are porous aluminas such as gamma or eta having average pore sizes from 50 to 200 .ANG., or 75 to 150 .ANG. (as determined by ASTM D4284); a surface area (as measured by the BET method) from 100 to 300 m.sup.2/g, or 150 to 250 m.sup.2/g; and a pore volume of from 0.25 to 1.0 cm.sup.3/g, or 0.35 to 0.8 cm.sup.3/g. More generally, any convenient size, shape, and/or pore size distribution for a catalyst suitable for hydrotreatment of a distillate (including lubricant base stock) boiling range feed in a conventional manner may be used. Preferably, the support or carrier material is an amorphous support, such as a refractory oxide. Preferably, the support or carrier material can be free or substantially free of the presence of molecular sieve, where substantially free of molecular sieve is defined as having a content of molecular sieve of less than about 0.01 wt %.
[0056] The at least one Group VIII non-noble metal, in oxide form, can typically be present in an amount ranging from about 2 wt % to about 40 wt %, preferably from about 4 wt % to about 15 wt %. The at least one Group VI metal, in oxide form, can typically be present in an amount ranging from about 2 wt % to about 70 wt %, preferably for supported catalysts from about 6 wt % to about 40 wt % or from about 10 wt % to about 30 wt %. These weight percents are based on the total weight of the catalyst. Suitable metal catalysts include nickel/molybdenum (1-10% Ni as oxide, 10-40% Mo as oxide), or nickel/tungsten (1-10% Ni as oxide, 10-40% W as oxide) on alumina, silica, silica-alumina, or titania.
[0057] The hydroprocessing is carried out in the presence of hydrogen. A hydrogen stream is, therefore, fed or injected into a vessel or reaction zone or hydroprocessing zone in which the hydroprocessing catalyst is located. Hydrogen, which is contained in a hydrogen "treat gas," is provided to the reaction zone. Treat gas, as referred to herein, can be either pure hydrogen or a hydrogen-containing gas, which is a gas stream containing hydrogen in an amount that is sufficient for the intended reaction(s), optionally including one or more other gasses (e.g., nitrogen and light hydrocarbons such as methane). The treat gas stream introduced into a reaction stage will preferably contain at least about 50 vol. % and more preferably at least about 75 vol. % hydrogen. Optionally, the hydrogen treat gas can be substantially free (less than 1 vol %) of impurities such as H.sub.2S and NH.sub.3 and/or such impurities can be substantially removed from a treat gas prior to use.
[0058] Hydrogen can be supplied at a rate of from about 100 SCF/B (standard cubic feet of hydrogen per barrel of feed) (17 Nm.sup.3/m.sup.3) to about 15000 SCF/B (1700 Nm.sup.3/m.sup.3). Preferably, the hydrogen is provided in a range of from about 2000 SCF/B (340 Nm.sup.3/m.sup.3) to about 12000 SCF/B (2040 Nm.sup.3/m.sup.3). Hydrogen can be supplied co-currently with the input feed to the hydrotreatment reactor and/or reaction zone or separately via a separate gas conduit to the hydrotreatment zone.
[0059] Hydrotreating conditions can include temperatures of 200.degree. C. to 450.degree. C., or 315.degree. C. to 425.degree. C.; pressures of 250 psig (1.8 MPag) to 5000 psig (34.6 MPag) or 300 psig (2.1 MPag) to 3000 psig (20.8 MPag), or about 2.9 MPag to about 13.9 MPag (.about.400 to .about.2000 psig); liquid hourly space velocities (LHSV) of 0.1 hr.sup.-1 to 10 hr.sup.-1, or 0.1 hr.sup.-1 to 5.0 hr'; and a hydrogen treat gas rate of from about 430 to about 2600 Nm.sup.3/m.sup.3 (.about.2500 to .about.15000 SCF/bbl), or about 850 to about 1700 Nm.sup.3/m.sup.3 (.about.5000 to .about.10000 SCF/bbl).
[0060] In various aspects, the feedstock can be exposed to a hydrocracking catalyst under effective hydrocracking conditions. Hydrocracking catalysts typically contain sulfided base metals on acidic supports, such as amorphous silica alumina, cracking zeolites such as USY, or acidified alumina. Often these acidic supports are mixed or bound with other metal oxides such as alumina, titania or silica. Examples of suitable acidic supports include acidic molecular sieves, such as zeolites or silicoaluminophophates. One example of suitable zeolite is USY, such as a USY zeolite with cell size of 24.30 Angstroms or less. Additionally or alternately, the catalyst can be a low acidity molecular sieve, such as a USY zeolite with a Si to Al ratio of at least about 20, and preferably at least about 40 or 50. ZSM-48, such as ZSM-48 with a SiO.sub.2 to Al.sub.2O.sub.3 ratio of about 110 or less, such as about 90 or less, is another example of a potentially suitable hydrocracking catalyst. Still another option is to use a combination of USY and ZSM-48. Still other options include using one or more of zeolite Beta, ZSM-5, ZSM-35, or ZSM-23, either alone or in combination with a USY catalyst. Non-limiting examples of metals for hydrocracking catalysts include metals or combinations of metals that include at least one Group VIII metal, such as nickel, nickel-cobalt-molybdenum, cobalt-molybdenum, nickel-tungsten, nickel-molybdenum, and/or nickel-molybdenum-tungsten. Additionally or alternately, hydrocracking catalysts with noble metals can also be used. Non-limiting examples of noble metal catalysts include those based on platinum and/or palladium. Support materials which may be used for both the noble and non-noble metal catalysts can comprise a refractory oxide material such as alumina, silica, alumina-silica, kieselguhr, diatomaceous earth, magnesia, zirconia, or combinations thereof, with alumina, silica, alumina-silica being the most common (and preferred, in one embodiment).
[0061] When only one hydrogenation metal is present on a hydrocracking catalyst, the amount of that hydrogenation metal can be at least about 0.1 wt % based on the total weight of the catalyst, for example at least about 0.5 wt % or at least about 0.6 wt %. Additionally or alternately when only one hydrogenation metal is present, the amount of that hydrogenation metal can be about 5.0 wt % or less based on the total weight of the catalyst, for example about 3.5 wt % or less, about 2.5 wt % or less, about 1.5 wt % or less, about 1.0 wt % or less, about 0.9 wt % or less, about 0.75 wt % or less, or about 0.6 wt % or less. Further additionally or alternately when more than one hydrogenation metal is present, the collective amount of hydrogenation metals can be at least about 0.1 wt % based on the total weight of the catalyst, for example at least about 0.25 wt %, at least about 0.5 wt %, at least about 0.6 wt %, at least about 0.75 wt %, or at least about 1 wt %. Still further additionally or alternately when more than one hydrogenation metal is present, the collective amount of hydrogenation metals can be about 35 wt % or less based on the total weight of the catalyst, for example about 30 wt % or less, about 25 wt % or less, about 20 wt % or less, about 15 wt % or less, about 10 wt % or less, or about 5 wt % or less. In embodiments wherein the supported metal comprises a noble metal, the amount of noble metal(s) is typically less than about 2 wt %, for example less than about 1 wt %, about 0.9 wt % or less, about 0.75 wt % or less, or about 0.6 wt % or less. It is noted that hydrocracking under sour conditions is typically performed using a base metal (or metals) as the hydrogenation metal.
[0062] A hydrocracking process under sour conditions can be carried out at temperatures of about 550.degree. F. (288.degree. C.) to about 840.degree. F. (449.degree. C.), hydrogen partial pressures of from about 1500 psig to about 5000 psig (10.3 MPag to 34.6 MPag), liquid hourly space velocities of from 0.05 h.sup.-1 to 10 h.sup.-1, and hydrogen treat gas rates of from 35.6 m.sup.3/m.sup.3 to 2670 m.sup.3/m.sup.3 (200 SCF/B to 15,000 SCF/B). In other embodiments, the conditions can include temperatures in the range of about 600.degree. F. (343.degree. C.) to about 815.degree. F. (435.degree. C.), hydrogen partial pressures of from about 1500 psig to about 3000 psig (10.3 MPag-20.9 MPag), and hydrogen treat gas rates of from about 213 m.sup.3/m.sup.3 to about 1780 m.sup.3/m.sup.3 (1200 SCF/B to 10,000 SCF/B). The LHSV can be from about 0.25 h.sup.-1 to about 50 h.sup.-1, or from about 0.5 h.sup.-1 to about 20 preferably from about 0.25 h.sup.-1 to about 4.0 h.sup.-1.
Deasphalting Heavy Cracked Feeds
[0063] In various aspects, the heavy cracked feed can be a deasphalted heavy cracked feed. Deasphalting of heavy hydrocarbons, such as vacuum resids, is known in the art and practiced commercially. A deasphalting process typically corresponds to contacting a heavy hydrocarbon with an alkane solvent (propane, butane, pentane, hexane, heptane etc. and their isomers), either in pure form or as mixtures, to produce two types of product streams. One type of product stream can be a deasphalted oil extracted by the alkane, which is further separated to produce deasphalted oil stream. A second type of product stream can be a residual portion of the feed not soluble in the solvent, often referred to as rock or asphaltene fraction. The deasphalted oil fraction can be further processed into make fuels or lubricants. The rock fraction can be further used as blend component to produce asphalt, fuel oil, and/or other products. The rock fraction can also be used as feed to gasification processes such as partial oxidation, fluid bed combustion or coking processes. The rock can be delivered to these processes as a liquid (with or without additional components) or solid (either as pellets or lumps).
[0064] During solvent deasphalting, the input feed to the solvent deasphalting unit can be mixed with a solvent. Portions of the feed that are soluble in the solvent are then extracted, leaving behind a residue with little or no solubility in the solvent. The portion of the deasphalted feedstock that is extracted with the solvent is often referred to as deasphalted oil. Typical solvent deasphalting conditions include mixing a feedstock fraction with a solvent in a weight ratio of from about 1:2 to about 1:10, such as about 1:8 or less. Typical solvent deasphalting temperatures range from 40.degree. C. to 200.degree. C., or 40.degree. C. to 150.degree. C., depending on the nature of the feed and the solvent. The pressure during solvent deasphalting can be from about 50 psig (.about.345 kPag) to about 1000 psig (.about.6900 kPag).
Examples of Reactor Configurations
[0065] Provided herein is a unique multi-layered reactor for treatment of heavy cracked feeds. Exemplary beds are provided in the in the figures. In preferred cases, the reactor comprises a first bulk or supported mixed metal catalyst comprising Ni and Mo, a second bulk or supported mixed metal catalyst comprising Ni and W, and a third catalyst comprising a zeolite-based hydrocracking catalyst. As shown in the figures, the third catalyst may comprise a hydrocracking catalyst (FIG. 1), a mixture of hydrocracking catalyst and arosat catalyst (FIG. 4), and/or a noble metal containing catalyst (FIG. 6). The reactor configurations and benefits of the present disclosure can be better understood with reference to the examples.
Example 1--Hydrocracking Catalysts for Heavy Cracked Feeds
[0066] A raw fluid catalytic cracking (FCC) main column bottoms (MCB) was obtained for catalytic testing of combined hydrotreating/hydrocracking catalysts systems. The raw MCB feedstock had the following properties
TABLE-US-00001 TABLE 1 Raw MCB Feed Properties Property Value T.sub.10 674.degree. F. (356.degree. C.) T.sub.50 792.degree. F. (426.degree. C.) T.sub.90 984.degree. F. (528.degree. C.) Density 1.11 g/cc Sulfur 2.8 wt % Nitrogen 1750 ppm
[0067] The raw MCB stream from Table 1 was used as a feedstock for a pilot scale processing plant. The MCB was exposed to four different catalyst beds, which are shown in FIG. 1. From left to right, the first, second, and third beds include a stacked bed configurations including an initial layer bulk metal hydrotreating catalyst comprising Ni and Mo followed by a second layer of hydrotreating catalyst comprising Ni and W, which in turn is followed by a third and final layer of zeolite-based hydrocracking catalyst. The only variance between these stacked beds occurs in the third and final layer. The first bed contains a 1 wt % Pt hydrocracking catalyst exhibiting a FAU framework on a silica-alumina support with a silica to alumina ratio of about 20. The second bed contains a commercially available hydrocracking catalyst comprising Ni and W exhibiting a FAU framework. The third bed contains a 0.6 wt % Pt hydrocracking catalyst exhibiting a FAU framework on a silica-alumina support with a silica to alumina ratio of about 200. The fourth bed is a control containing only a single layer of bulk metal hydrotreating catalyst comprising Ni and Mo.
[0068] The conditions for the run included a constant pressure of about 2175 psig (15 MPa) and about 10,000 SCF/B of hydrogen treat gas. Temperature and liquid hourly space velocity (LHSV) were varied between 680.degree. F.-700.degree. F. (360.degree. C.-371.degree. C.) and 0.25 h.sup.--0.5 h.sup.-1, respectively. The results are shown in FIGS. 2A-F. The feed was processed in the pilot plant for about 47 days with an initial temperature of 680.degree. F. (360.degree. C.) and LHSV of 0.25 h.sup.-1. At about Day 37, temperature was increased to 700.degree. F. (371.degree. C.). At about Day 43, LHSV was increased to 0.5 h.sup.-1. As shown in FIGS. 2A and 2B, the stacked configuration generally results in comparable or better conversion of 700.degree. F.+ molecules and lower density than using bulk metal hydrotreating catalyst comprising Ni and Mo alone. Remaining data related to naphtha yield, distillate yield, sulfur content, and nitrogen content are generally comparable and within acceptable ranges.
Example 2--Hydrocracking Catalysts for Deasphalted Heavy Cracked Feeds
[0069] A deasphalted fluid catalytic cracking (FCC) main column bottoms (MCB) was obtained for catalytic testing of combined hydrotreating/hydrocracking catalysts systems. The deasphalted MCB feedstock had the following properties
TABLE-US-00002 TABLE 2 Deasphalted MCB Feed Properties Property Value T.sub.10 687.degree. F. (363.degree. C.) T.sub.50 779.degree. F. (415.degree. C.) T.sub.90 916.degree. F. (491.degree. C.) Density 1.13 g/cc Sulfur 4.6 wt % Nitrogen 2390 ppm
[0070] The MCB was exposed to the same four catalyst beds as Example 1, which are shown in FIG. 1. The conditions for the run included a constant pressure of about 2175 psig (15 MPa) and about 10,000 SCF/B of hydrogen treat gas. Temperature and liquid hourly space velocity (LHSV) were varied between 680.degree. F.-700.degree. F. (360.degree. C.-371.degree. C.) and 0.25 h.sup.-1-0.5 h.sup.-1, respectively. The results are shown in FIGS. 3A-F. The feed was processed in the pilot plant for about 25 days with an initial temperature of 700.degree. F. (371.degree. C.) and LHSV of 0.5 h.sup.-1. At about Day 13, LHSV was decreased to 0.25 h.sup.-1. At about Day 18, temperature was decreased to 680.degree. F. (360.degree. C.). The advantages of the stacked bed configurations are particularly telling in the context of the deasphalted MCB. As shown in FIGS. 3A and 3B, the stacked configuration results in better conversion of 700.degree. F.+ molecules and lower density than using bulk metal hydrotreating catalyst comprising Ni and Mo alone in all cases. Remaining data related to naphtha yield, distillate yield, sulfur content, and nitrogen content are generally comparable and within acceptable ranges. Examples 1 and 2 prove the discovery that a combination of bulk-metal hydrotreating catalyst followed by zeolitic, metal-containing, hydrocracking catalyst can achieve high yields of ULSD blendstock and reduce 700.degree. F.+ range material. This is non-intuitive for a single reaction stage because conventional wisdom would dictate that the use of such zeolitic, metal-containing, hydrocracking catalysts would be ineffective due to rapid poisoning of the catalyst.
Example 3--Hydrocracking/Arosat Catalysts for Deasphalted Heavy Cracked Feeds
[0071] A deasphalted FCC MCB was obtained for catalytic testing, comparing several combination of NiMo and NiW bulk-metal hydrotreating catalysts followed by mixtures of hydrocracking and aromatic saturation ("arosat") catalysts. The deasphalted MCB feedstock had the same properties as the deasphalted MCB feedstock in Table 2 above.
[0072] The deasphalted MCB from Table 2 was used as a feedstock for a pilot scale processing plant. The deasphalted MCB was exposed to four different catalyst beds, which are shown in FIG. 4. From left to right, the first, second, and third beds include a stacked bed configurations including an initial layer bulk metal hydrotreating catalyst comprising Ni and Mo followed by a second layer of hydrotreating catalyst comprising Ni and W, which in turn is followed by a third and final layer of mixed zeolite-based hydrocracking catalyst and noble metal arosat catalyst. The final layer of Bed 1 contains an equal mixture (by weight) of approximately 1.0 wt % Pt impregnated on a FAU-type zeolite with an SiO.sub.2/Al.sub.2O.sub.3 ratio of .about.20 in an extrudate with commercially available alumina, and an arosat catalyst containing both Pd and Pt (0.77 wt % and 0.25 wt %, respectively) on a mesoporous aluminosilicate support with an SiO.sub.2/Al.sub.2O.sub.3 ratio of .about.50. The final layer of Bed 2 contains an equal mixture (by weight) of approximately 1.0 wt % Pt impregnated on a FAU-type zeolite with an SiO.sub.2/Al.sub.2O.sub.3 ratio of .about.60 in an extrudate with commercially available alumina, and an arosat catalyst containing both Pd and Pt (0.77 wt % and 0.25 wt %, respectively) on a mesoporous aluminosilicate support with an SiO.sub.2/Al.sub.2O.sub.3 ratio of .about.50. The final layer of Bed 3 contains an equal mixture (by weight) of approximately 1.0 wt % Pt impregnated on a FAU-type zeolite with an SiO.sub.2/Al.sub.2O.sub.3 ratio of .about.20 in an extrudate with commercially available alumina, and an arosat catalyst containing 2.0 wt % Pt on a 40 .ANG.-pore mesoporous aluminosilicate support.
[0073] The conditions for the run included a constant pressure of about 2175 prig (15 MPa) and about 10,000 SCF/B of hydrogen treat gas. Temperature and liquid hourly space velocity (LHSV) were varied between 680.degree. F.-700.degree. F. (360.degree. C.-371.degree. C.) and 0.25 h.sup.-1-0.5 h.sup.-1, respectively. The results are shown in FIGS. 5A-F. The feed was processed in the pilot plant for about 25 days with an initial temperature of 700.degree. F. (371.degree. C.) and LHSV of 0.5 h.sup.-1. At about Day 13, LHSV was decreased to 0.25 h.sup.-1. At about Day 18, temperature was decreased to 680.degree. F. (360.degree. C.). The advantages of the stacked bed configurations are particularly telling in the context of the deasphalted MCB. As shown in FIGS. 5A and 5B, the stacked configuration results in better conversion of 700.degree. F.+ molecules and lower density than using bulk metal hydrotreating catalyst comprising Ni and Mo alone in all cases. Remaining data related to naphtha yield, distillate yield, sulfur content, and nitrogen content are generally comparable or better and within acceptable ranges. Example 3 proves the discovery that a combination of bulk-metal hydrotreating catalyst followed by a hydrocracking/arosat mixed catalyst can achieve high yields of ULSD blendstock and reduce 700.degree. F.+ range material. This is non-intuitive for a single reaction stage because conventional wisdom would dictate that the use of such a catalyst mixture would be ineffective due to rapid poisoning of the catalyst.
Example 4--Noble Metal Catalysts and Mixed Catalysts for Heavy Cracked Feeds
[0074] A raw FCC MCB was obtained for catalytic testing of combined noble metal catalysts systems. The raw MCB feedstock had the same properties as the raw MCB feedstock in Table 1 above.
[0075] The raw MCB from Table 1 was used as a feedstock for a pilot scale processing plant. The MCB was exposed to seven different catalyst beds, which are shown in FIG. 6. The stacked beds, beds 1-6, include an initial layer bulk metal hydrotreating catalyst comprising Ni and Mo followed by a second layer of hydrotreating catalyst comprising Ni and W, which in turn is followed by a third and final layer containing one of six different noble-metal hydrocracking, arosat, or combined hydrocracking/arosat catalysts.
[0076] The final layer of Bed 1 is mesoporous aluminosilicate with an SiO.sub.2/Al.sub.2O.sub.3 ratio of .about.50 containing a mixture of Pd and Pt (0.77 wt % and 0.25 wt %, respectively). The final layer of Bed 2 is zeolite-based (FAU), with an SiO.sub.2/Al.sub.2O.sub.3 ratio of .about.200 and a Pt content of 0.6 wt % bound in an extrudate with commercially available alumina. The final layer of Bed 3 is FAU-based, having an SiO.sub.2/Al.sub.2O.sub.3 ratio of .about.20 and Pt content of 1.0 wt % bound in an extrudate with commercially available alumina. The final layer of Bed 4 contains an equal mixture (by weight) of approximately 1.0 wt % Pt impregnated on a FAU-type zeolite with an SiO.sub.2/Al.sub.2O.sub.3 ratio of .about.20 in an extrudate with commercially available alumina, and an arosat catalyst containing both Pd and Pt (0.77 wt % and 0.25 wt %, respectively) on a mesoporous aluminosilicate support with an SiO.sub.2/Al.sub.2O.sub.3 ratio of .about.50. The final layer of Bed 5 contains an equal mixture (by weight) of approximately 1.0 wt % Pt impregnated on a FAU-type zeolite with an SiO.sub.2/Al.sub.2O.sub.3 ratio of .about.60 in an extrudate with commercially available alumina, and an arosat catalyst containing both Pd and Pt (0.77 wt % and 0.25 wt %, respectively) on a mesoporous aluminosilicate support with an SiO.sub.2/Al.sub.2O.sub.3 ratio of .about.50. The final layer of Bed 6 contains an equal mixture (by weight) of approximately 1.0 wt % Pt impregnated on a FAU-type zeolite with an SiO.sub.2/Al.sub.2O.sub.3 ratio of .about.20 in an extrudate with commercially available alumina, and an arosat catalyst containing 2.0 wt % Pt on a 40 .ANG.-pore mesoporous aluminosilicate support. Bed 7 is a bulk-metal NiMo hydrotreating catalyst used for comparison to these stacked beds.
[0077] The conditions for the run included a constant pressure of about 2175 psig (15 MPa) and about 10,000 SCF/B of hydrogen treat gas. Temperature and liquid hourly space velocity (LHSV) were varied between 680.degree. F.-700.degree. F. (360.degree. C.-371.degree. C.) and 0.25 h.sup.-1-0.5 h.sup.-1, respectively. The results are shown in FIGS. 7A-F. The feed was processed in the pilot plant for about 47 days with an initial temperature of 680.degree. F. (360.degree. C.) and LHSV of 0.25 h.sup.-1. At about Day 37, temperature was increased to 700.degree. F. (371.degree. C.). At about Day 43, LHSV was increased to 0.5 h.sup.-1. As shown in FIGS. 7A and 7B, the stacked configuration generally results in comparable or better conversion of 700.degree. F.+ molecules and lower density than using bulk metal hydrotreating catalyst comprising Ni and Mo alone. Remaining data related to naphtha yield, distillate yield, sulfur content, and nitrogen content are generally comparable and within acceptable ranges.
Example 5--Noble Metal Catalysts and Mixed Catalysts for Deasphalted Heavy Cracked Feeds
[0078] A deasphalted FCC MCB was obtained for catalytic testing, comparing several combination of NiMo and NiW bulk-metal hydrotreating catalysts followed by one of six different noble-metal hydrocracking, arosat, or combined hydrocracking/arosat catalysts. The deasphalted MCB feedstock had the same properties as the deasphalted MCB feedstock in Table 2 above.
[0079] The deasphalted MCB was used as a feedstock for a pilot scale processing plant. The deasphalted MCB was exposed to the same seven catalyst beds as Example 4, which are shown in FIG. 6. The conditions for the run included a constant pressure of about 2175 psig (15 MPa) and about 10,000 SCF/B of hydrogen treat gas. Temperature and liquid hourly space velocity (LHSV) were varied between 680.degree. F.-700.degree. F. (360.degree. C.-371.degree. C.) and 0.25 h.sup.-1-0.5 h.sup.-1, respectively. The results are shown in FIGS. 8A-F. The feed was processed in the pilot plant for about 25 days with an initial temperature of 700.degree. F. (371.degree. C.) and LHSV of 0.5 h.sup.-1. At about Day 13, LHSV was decreased to 0.25 h.sup.-1. At about Day 18, temperature was decreased to 680.degree. F. (360.degree. C.). The advantages of the stacked bed configurations are particularly telling in the context of the deasphalted MCB. As shown in FIGS. 8A and 8B, the stacked configuration results in better conversion of 700.degree. F.+ molecules and lower density than using bulk metal hydrotreating catalyst comprising Ni and Mo alone in nearly all cases. Remaining data related to naphtha yield, distillate yield, sulfur content, and nitrogen content are generally comparable or better and within acceptable ranges. Example 3 proves the discovery that a combination of bulk-metal hydrotreating catalyst followed by a mixture of noble metal micro- and/or mesoporous hydrocracking and arosat catalysts can achieve high yields of ULSD blendstock and reduce 700.degree. F.+ range material. This is non-intuitive for a single reaction stage because conventional wisdom would dictate that the use of such a catalyst mixture would be ineffective due to rapid poisoning of the catalyst.
Additional Embodiments
Embodiment 1
[0080] A process for upgrading a heavy cracked feedstock, comprising: providing a feedstock comprising a density at 15.degree. C. of 1.06 g/cm.sup.3 or more, at least 50 wt % of one or more 343.degree. C.+ cracked fractions, and a sulfur content of 0.8 to 5.0 wt % sulfur; in a single reaction stage under fixed bed hydroprocessing conditions, exposing the feedstock to a first bulk or supported mixed metal catalyst comprising Ni and Mo; exposing the feedstock to a second bulk or supported mixed metal catalyst comprising Ni and W; and exposing the feedstock to a third catalyst comprising a zeolite-based hydrocracking catalyst.
Embodiment 2
[0081] The process of embodiment 1, wherein the zeolite-based hydrocracking catalyst comprises a Group VIII noble metal.
Embodiment 3
[0082] The process of embodiment 2, wherein the zeolite-based hydrocracking catalyst comprises 0.5 to 2.5 wt % Pt.
Embodiment 4
[0083] The process of any of the previous embodiments, wherein the third catalyst further comprises an aromatic saturation catalyst comprising Pd, Pt, or a combination thereof on an aluminosilicate support.
Embodiment 5
[0084] The process of embodiment 4, wherein the aromatic saturation catalyst comprises Pd and Pt in a wt % ratio of about 3:1 Pd to Pt.
Embodiment 6
[0085] The process of embodiment 4 or 5, wherein the aromatic saturation catalyst comprises 2.0 wt % Pt.
Embodiment 7
[0086] The process of any of the previous embodiments, wherein the zeolite exhibits a faujasite (FAU) framework type.
Embodiment 8
[0087] The process of any of the previous embodiments, wherein the feedstock comprises a density at 15.degree. C. of 1.1 g/cm.sup.3 or more, a T.sub.10 of at least 343.degree. C. and a T90 of at least 475.degree. C.; and a sulfur content of 3.0 wt % to 5.0 wt %.
Embodiment 9
[0088] The process of any of the previous embodiments, wherein the feedstock comprises a de-asphalted heavy cracked feedstock.
Embodiment 10
[0089] The process of any of the previous embodiments, wherein the fixed bed hydroprocessing conditions include a temperature of 300.degree. C. to 400.degree. C., a pressure of 1500 psig to 3000 psig, a hydrogen treat gas rate of 2,000 scf/bbl to 12,000 scf/bbl, and a LHSV of 0.2 h.sup.-1 to 1.5 h.sup.-1.
Embodiment 11
[0090] A system for processing a cracked feedstock comprising: a hydroprocessing reactor comprising a hydroprocessing inlet, and a hydroprocessing outlet, and a fixed bed comprising a first bulk or supported mixed metal catalyst comprising Ni and Mo; a second bulk or supported mixed metal catalyst comprising Ni and W; and a third catalyst comprising a zeolite-based hydrocracking catalyst; wherein the hydroprocessing inlet is designed to receive a feedstock comprising a density at 15.degree. C. of 1.06 g/cm.sup.3 or more, at least 50 wt % of one or more 343.degree. C.+ cracked fractions, and a sulfur content of 1.0 to 5.0 wt % sulfur; and wherein the fixed bed is oriented such that the feedstock contacts the first catalyst, second catalyst, and third catalyst sequentially.
[0091] When numerical lower limits and numerical upper limits are listed herein, ranges from any lower limit to any upper limit are contemplated. While the illustrative embodiments of the invention have been described with particularity, it will be understood that various other modifications will be apparent to and can be readily made by those skilled in the art without departing from the spirit and scope of the invention. Accordingly, it is not intended that the scope of the claims appended hereto be limited to the examples and descriptions set forth herein but rather that the claims be construed as encompassing all the features of patentable novelty which reside in the present invention, including all features which would be treated as equivalents thereof by those skilled in the art to which the invention pertains.
[0092] The present invention has been described above with reference to numerous embodiments and specific examples. Many variations will suggest themselves to those skilled in this art in light of the above detailed description. All such obvious variations are within the full intended scope of the appended claims.
* * * * *
D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.