Methods And Compositions For Prevention Of Fouling In Caustic Towers
TANG; Xiaofeng ; et al.
U.S. patent application number 16/084613 was filed with the patent office on 2019-03-07 for methods and compositions for prevention of fouling in caustic towers. The applicant listed for this patent is General Electric Company. Invention is credited to Mike HONG, Yongtao SHI, Xiaofeng TANG, Chun XU, Dengchao YAN, Guixi ZHANG.
| Application Number | 20190071610 16/084613 |
| Document ID | / |
| Family ID | 59850243 |
| Filed Date | 2019-03-07 |











View All Diagrams
| United States Patent Application | 20190071610 |
| Kind Code | A1 |
| TANG; Xiaofeng ; et al. | March 7, 2019 |
METHODS AND COMPOSITIONS FOR PREVENTION OF FOULING IN CAUSTIC TOWERS
Abstract
Provided are methods and compositions for inhibiting carbonyl based fouling materials of basic wash systems. Said methods comprise contacting the hydrocarbon stream that is or will be subjected to said wash systems with water soluble or water dispersible copolymers. Said copolymers comprise repeat units of ethylenically unsaturated monomers such as acrylic acid with other repeat units such as alkyl acrylates, allyl ethers, ethoxylated allyl repeat units, etc. In other embodiments, a third repeat unit is present and may comprise a hydrophobic moiety such as a styrene repeat unit.
| Inventors: | TANG; Xiaofeng; (Shanghai, CN) ; ZHANG; Guixi; (Shanghai, CN) ; HONG; Mike; (Shanghai, CN) ; SHI; Yongtao; (Shanghai, CN) ; XU; Chun; (Shanghai, CN) ; YAN; Dengchao; (Shanghai, CN) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 59850243 | ||||||||||
| Appl. No.: | 16/084613 | ||||||||||
| Filed: | March 18, 2016 | ||||||||||
| PCT Filed: | March 18, 2016 | ||||||||||
| PCT NO: | PCT/CN2016/076767 | ||||||||||
| 371 Date: | September 13, 2018 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C10G 19/02 20130101; C10G 2300/202 20130101; C09K 15/28 20130101; C08F 220/382 20200201; C10G 75/04 20130101; C08F 228/02 20130101; C10G 19/04 20130101; C08F 220/06 20130101; C08F 220/58 20130101; C09K 15/12 20130101; C08F 220/38 20130101; C08F 220/585 20200201; C08F 220/06 20130101; C08F 220/585 20200201; C08F 220/06 20130101; C08F 220/585 20200201 |
| International Class: | C10G 75/04 20060101 C10G075/04; C09K 15/12 20060101 C09K015/12; C09K 15/28 20060101 C09K015/28; C08F 220/06 20060101 C08F220/06; C08F 220/58 20060101 C08F220/58; C08F 220/38 20060101 C08F220/38; C10G 19/02 20060101 C10G019/02 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Mar 18, 2016 | CN | PCT/CN2016/076767 |
Claims
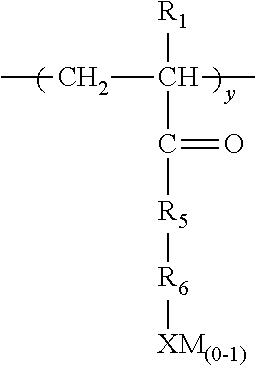
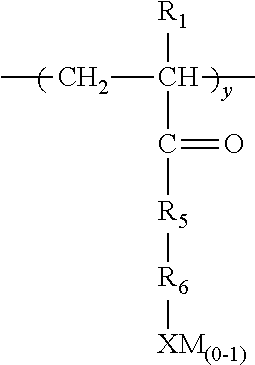
1. A method for inhibiting the formation of fouling materials comprising contacting a hydrocarbon stream containing carbonyl compounds with an antifoulant and treating said hydrocarbon stream with a basic wash, wherein said antifoulant comprises a polymer having repeat units characterized by the formula ##STR00015## wherein a must be present, b or c or b+c is present and d may or may not be present; E is a repeat unit remaining after polymerization of an ethylenically unsaturated compound; each R.sub.1 is independently chosen from H or lower (C.sub.1-C.sub.4) alkyl; R.sub.2 is a hydroxy substituted alkyl or alkylene moiety having from about 1-6 carbon atoms, X is an anionic radical selected from the group consisting of SO.sub.3, OSO.sub.3, PO.sub.3OPO.sub.3 or COO; M is H or hydrogens or any water soluble cationic moiety that counterbalances the valence of the anionic radical X; F is an ethylenically unsaturated hydrophobic moiety; Q in repeat unit c is chosen from C.sub.1-C.sub.3 alkylene or carbonyl, m is 0 or 1, R.sub.3 is CH.sub.2--CH--O .sub.n or CH.sub.2--CH--CH.sub.3--O .sub.n wherein n is from 1 to about 100 or R.sub.3 is CH.sub.2--CHOH or CH.sub.2--CH--(OH)--CH.sub.2; R.sub.4 is H, OH, SO.sub.3M, OSO.sub.3M, PO.sub.3M, OPO.sub.3M, or CO.sub.2M; with the proviso that when d is present it is present in an amount of 0.01-0.8 moles based on 1 mole of a; either b or c, or both b+c (when both are present) are present in a molar ratio of a:b or a:c or a:(b+c) of 0.1-100.
2. The method of claim 1, wherein about 1-2,000 ppm of said antifoulant is brought into contact with said hydrocarbon stream, based upon 1 million parts of said stream by weight.
3. The method of claim 1, wherein said polymer is a copolymer of acrylic acid (AA) acid/allyl ether or (AA)/allyl hydroxylated alkyl ether.
4. (canceled)
5. The method of claim 1, wherein said polymer is a terpolymer of AA/AHP SE/styrene.
6. The method of claim 1, wherein said polymer is a copolymer of AA/allylpolyethyleneglycol ether or AA/ethoxylated allyl ether.
7. The method of claim 6, wherein said ethoxylated allyl ether is allylpolyethyoxy (10) sulfate.
8. The method of claim 1, wherein said polymer is a terpolymer of AA/AHPSE/ammonium allylpolyethoxy (10) sulfate or a copolymer of AA/lower alkyl (C.sub.1-C.sub.4) acrylate.
9. The method of claim 8, wherein said lower alkyl (C.sub.1-C.sub.4) acrylate is an hydroxy alkyl acrylate.
10. The method of claim 9, wherein said hydroxy alkyl acrylate is 2 hydroxypropyl acrylate (HPA).
11. The method of claim 1, wherein said polymer is a terpolymer of AA/HPA/AHP SE/styrene.
12. (canceled)
13. The method of claim 1, wherein the antifoulant is added to the hydrocarbon stream simultaneously with the base wash or is added to the hydrocarbon stream in a hydrocarbon charge line to a basic wash tower or is added to the hydrocarbon stream in an input line to or recycle line from a basic wash tower, or a recycle line from a basic wash tower or to a caustic solution pipeline line.
14. The method of claim 1, wherein said hydrocarbon stream is a methanol to olefin (MTO) process stream or a cracked hydrocarbon stream from the pyrolysis of hydrocarbons.
15. The method of claim 14, wherein the cracked hydrocarbon stream is from the pyrolysis of ethane, propane, butane, naphtha, or mixtures thereof.
16. The method of claim 1, wherein the basic wash has a pH greater than 7.0 and comprises sodium hydroxide, potassium hydroxide, sodium carbonate, sodium hydrocarbonate, potassium hydrocarbonate, organic amine, or an alkanolamine.
17. The method according to claim 1, wherein the antifoulant further comprises another carbonyl scavenger, said another carbonyl scavenger comprises at least one compound selected from the group consisting of alcohol amines, alkyl amines, keto amines, amino acids, hydrazide compounds, hydroxylamines, reducing sugars, hydroxybenzenes, acetoacetate ester compounds, lactams, oxidizers, and reducers.
18. (canceled)
19. A water soluble or water dispersible polymer composition having the structure E .sub.a G .sub.z F .sub.d Formula VI wherein a, z, and d are all present; E is a repeat unit remaining after polymerization of an ethylenically unsaturated compound, F is a repeat unit remaining after polymerization of an ethylenically unsaturated hydrophobic moiety; wherein the molar ratio of d:a is about 0.1-0.8 moles of d:1 mole a; z is present in an amount of a:z of 0.1-100 moles a per 1 mole z; G is a repeat unit chosen from VIa, VIb, VIc, or VId or mixtures thereof, wherein VIa is ##STR00016## VIb is ##STR00017## wherein R.sub.1 is H or lower (C.sub.1-C.sub.4) alkyl, R.sub.2 is a hydroxy substituted alkyl or alkylene moiety having from about 1-6 carbon atoms, X is an anionic radical selected from the group consisting of SO.sub.3, OSO.sub.3, PO.sub.3, OPO.sub.3, or COO; M is H or hydrogens or any water soluble cationic moiety that counterbalances the valence of the anionic radical X; Q is chosen from C.sub.1-C.sub.3 alkylene or carbonyl, m is 0 or 1; R.sub.3 is CH.sub.2--CH.sub.2--O .sub.n; --CH.sub.2CHCH.sub.3O .sub.n wherein n=1 to 100; or R.sub.3 is hydroxylated lower (C.sub.1-C.sub.4) alkylene; and R.sub.4, is H, OH, SO.sub.3M, OSO.sub.3M, PO.sub.3M, OPO.sub.3M, or CO.sub.2M; VIc is ##STR00018## wherein R.sub.1 is as defined above, R.sub.5 is NH or O; R.sub.6 is lower (C.sub.1-C.sub.4) alkyl or alkylene or lower (C.sub.1-C.sub.4) hydroxy substituted alkyl or alkylene; X and M are as defined above; and VId is ##STR00019## wherein R.sub.7 is CH.sub.2 or benzyl, and X and M are defined above.
20. The polymer composition as recited in claim 19, wherein E is acrylic acid or water soluble salt form thereof (AA).
21. The polymer composition of claim 20, wherein F is styrene.
22. The polymer composition of claim 21, wherein G is VIc and R.sub.5 is NH, or O; R.sub.6 is 2-methyl-1-propane or 2 hydroxypropyl, and X is SO.sub.3.
23. (canceled)
24. The polymer composition of claim 21, wherein G is VIa and R.sub.2 is 2 hydroxypropyl and X is SO.sub.3.
25. The polymer composition of claim 21, wherein G is VIb and Q is CH.sub.2, m=1; R.sub.3 is CH.sub.2--CH.sub.2--O .sub.n and R.sub.4 is SO.sub.3M.
26. The polymer composition of claim 21, wherein G is VIb and wherein Q=CH.sub.2, m=O; R.sub.3 is CH.sub.2CH.sub.2--O .sub.n and R.sub.4 is OSO.sub.3M.
27. The polymer composition of claim 21, wherein G comprises both VIa and VIb and wherein R.sub.2 is hydroxypropyl; X is SO.sub.3; Q is CH.sub.2; m=O; R.sub.3 is CH.sub.2CH.sub.2--O .sub.n and R.sub.4 is OSO.sub.3M.
28. The polymer composition of claim 21, wherein G is VId; wherein R.sub.7 is benzyl or CH.sub.2, and X is SO.sub.3.
29. (canceled)
30. A water soluble terpolymer composition comprising acrylic acid or acrylic acid salt (AA) repeat units, hydrophobic monomeric repeat units, and a third repeat unit selected from the group consisting of acrylamide repeat units, allyl ether repeat units, lower alkyl (C.sub.1-C.sub.4) acrylate repeat units, ethoxylated or propoxylated allyl repeat units, allyl polyethylene glycol ether repeat units, sulfonated styrene repeat units, and allyl sulfonic acid repeat units.
31. The water soluble terpolymer composition as recited in claim 30, wherein said hydrophobic monomeric repeat units comprise styrene.
32. The water soluble terpolymer composition as recited in claim 31, wherein said third repeat unit is 2-acrylamido-2-methyl-1 propane sulfonic acid (AMPS), allyl hydroxy propyl sulfonate ether (AHPSE), allylpolyethoxy sulfonate (APES), or hydroxypropyl acrylate (HPA).
33-36. (canceled)
Description
FIELD OF INVENTION
[0001] The invention relates to methods and compositions for inhibiting the formation of fouling deposits in basic wash systems of the type adapted to scrub impurities, such as those that may be formed via aldol condensation reactions, from liquid or gas phase hydrocarbonaceous streams.
BACKGROUND OF THE INVENTION
[0002] Olefinic compounds such as ethylene, propylene, butylene, and amylene can be formed from methanol to olefin (MTO) or various pyrolytic cracking processes. In these processes, a variety of carbonyl compounds such as aldehydes and ketones are often formed. Typically, the carbonyl compounds are found in the gas stream in about 1 to 200 parts per million (ppm) by weight relative to the hydrocarbon stream with concentrations of more than 1,000 ppm sometimes encountered due to the particular feedstock and reactor operation parameter employed for the reactions.
[0003] The hydrocarbon product stream formed via cracking or MTO processes is cooled and sometimes compressed. The product gas stream may be passed through a basic wash system (pH>7) to remove acidic components such as hydrogen sulfide and carbon dioxide. In many cases, the carbonyl compounds present, such as the aldehydes, will undergo polymerization to form condensation polymers known as aldol polymers or red oil. These aldol polymers or red oil possess low solubility in the alkaline wash and the hydrocarbon media and can deposit on wash tower tray conduits and other internal surfaces of the process equipment leading to fouling and eventual plugging. These deposits can restrict flow through the equipment and can cause undesirable pressure drops, resulting in decreased process throughput, increased operating costs, and unit shut down for periodic cleaning.
[0004] The basic wash systems in which treatment is required to inhibit such polymer based fouling include amine acid gas scrubber, such as MEA, DEA, isopropyl amine, butyl amine, etc., and caustic wash systems.
[0005] Generally, the basic washing entails contacting the gaseous olefins with an aqueous basic solution in a wash tower to remove hydrogen sulfide, carbon dioxide, and other oxygenated compounds therefrom. The basic washing is particularly appropriate for the basic washing process which follows the pyrolytic cracking of such hydrocarbons as ethane, propane, butane, naphtha, and mixtures thereof to produce the corresponding gaseous ethylene, propylene, butadiene, and the like, or follows the MTO production process containing the carbonyl and other contaminants.
SUMMARY OF THE INVENTION
[0006] In one embodiment of the invention, methods are provided for inhibiting the formation of fouling materials comprising contacting a hydrocarbon media containing aldehyde compounds with an antifoulant. The hydrocarbon media is treated in a basic wash system. The antifoulant may comprise a polymer having repeat units characterized by the formula
##STR00001##
wherein a must be present, b or c or b+c is present and d may or may not be present; E is a repeat unit remaining after polymerization of an ethylenically unsaturated compound and can be, for example, (meth) acrylic acid or (meth) acrylamide; each R.sub.1 is independently chosen from H or lower (C.sub.1-C.sub.4) alkyl; R.sub.2 is a hydroxy substituted alkyl or alkylene moiety having from about 1-6 carbon atoms, X is an anionic radical selected from the group consisting of SO.sub.3, OSO.sub.3, PO.sub.3OPO.sub.3 or COO; M is one or more hydrogens or any water soluble cationic moiety that counterbalances the valence of the anionic radical X and can be Ca, Na, K, NH.sub.4, etc.; F is an ethylenically unsaturated hydrophobic moiety such as styrene and its derivatives, acrylonitrile, olefin with (C.sub.1-C.sub.18) alkyl group, alkyl (meth) acrylate; Q in repeat unit c is chosen from C.sub.1-C.sub.3 alkylene or carbonyl, m is 0 or 1, R.sub.3 is CH.sub.2--CH--O .sub.n or CH.sub.2--CHCH.sub.3--O .sub.n wherein n is from 1 to about 100 or R.sub.3 is CH.sub.2--CHOH or CH.sub.2--CH--(OH)--CH.sub.2; R.sub.4 is H, OH, SO.sub.3M, OSO.sub.3M, PO.sub.3M, OPO.sub.3M, or CO.sub.2M; with the proviso that when d is present it is present in an amount of 0.01-0.8 moles based on 1 mole of a; either b or c, or both b+c (when both are present) are present in a molar ratio of a:b or a:c or a:(b+c) of 0.1-100, or in some exemplary embodiments 1-10.
[0007] In certain embodiments, the polymeric antifoulant may comprise a copolymer of acrylic acid (AA) and allyl ether. In other embodiments of the invention, the polymeric foulant may be an acrylic acid (AA) allylhydroxylated alkyl ether, also referred to as 1-propane sulfonic acid, 2-hydroxy-3 (2-propenyl oxy) mono sodium salt (AHPSE).
[0008] The polymeric antifoulant may also be a terpolymer of AA/AHPSE/styrene or it may be, in certain embodiments, a copolymer of acrylic acid and an allyl polyethylene glycol ether. In some cases, the polymer may comprise a copolymer of acrylic acid with an ethoxylated allyl ether. In other embodiments, the copolymer may comprise acrylic acid and lower alkyl acrylates such as hydroxy substituted alkyl acrylates.
[0009] In another aspect of the invention, novel water soluble or water dispersible polymer compositions are provided having the structure
E .sub.a G .sub.z F .sub.d Formula VI
wherein a, z, and d are all present; E is a repeat unit remaining after polymerization of an ethylenically unsaturated compound, F is a repeat unit remaining after polymerization of an ethylenically unsaturated hydrophobic moiety; wherein the molar ratio of d:a is about 0.1-0.8 moles of d:1 mole a; z is present in an amount of a:z of 0.1-100, 1-10 moles a per 1 mole z and in some embodiments is present in an amount of 1-10 moles of a per mole z; G is a repeat unit chosen from VIa, VIb, VIc, or VId or mixtures thereof, wherein VIa is
##STR00002##
VIb is
##STR00003##
[0010] wherein R.sub.1 is H or lower (C.sub.1-C.sub.4) alkyl, R.sub.2 is a hydroxy substituted alkyl or alkylene moiety having from about 1-6 carbon atoms, X is an anionic radical selected from the group consisting of SO.sub.3, OSO.sub.3, PO.sub.3, OPO.sub.3, or COO; M is H or hydrogens or any water soluble cationic moiety that counterbalances the valence of the anionic radical X; Q is chosen from C.sub.1-C.sub.3 alkylene or carbonyl, m is 0 or 1; R.sub.3 is CH.sub.2--CH.sub.2--O .sub.n; --CH.sub.2CHCH.sub.3O .sub.n wherein n=1 to 100; or R.sub.3 is hydroxylated lower (C.sub.1-C.sub.4) alkylene; and R.sub.4i, is H, OH, SO.sub.3M, OSO.sub.3M, PO.sub.3M, OPO.sub.3M, or CO.sub.2M;
VIc is
##STR00004##
[0011] wherein R.sub.1 is as defined above, R.sub.5 is NH or O; R.sub.6 is lower (C.sub.1-C.sub.4) alkyl or alkylene or lower (C.sub.1-C.sub.4) hydroxy substituted alkyl or alkylene; X and M are as defined above; and VId is
##STR00005##
wherein R.sub.7 is CH.sub.2 or benzyl, and X and M are defined above.
[0012] In further aspects of the invention, novel water soluble terpolymer compositions are provided that comprise acrylic acid or acrylic acid salt repeat units, a hydrophobic repeat unit such as styrene and its derivatives, acrylonitrile, olefin with (C.sub.1-C.sub.18) alkyl group, alkyl (meth) acrylate, and a third repeat unit selected from the group consisting acrylamide repeat units, allyl ether repeat units, lower alkyl (C.sub.1-C.sub.4) acrylate repeat units, ethoxylated or propoxylated allyl repeat units, allyl polyethylene glycol ether repeat units, sulfonated styrene repeat units, and allyl sulfonic acid repeat units. Terpolymers wherein the hydrophobic monomeric repeat unit comprises styrene may be mentioned as exemplary.
BRIEF DESCRIPTION OF THE DRAWING
[0013] The invention is further described in connection with the drawings wherein:
[0014] FIG. 1 is a microphotograph of filter cakes resulting from candidate antifoulant treatments as referred to in Example 3.
DETAILED DESCRIPTION
[0015] In one aspect of the invention, methods and compositions are provided to inhibit the formation of polymeric based fouling deposits during the basic washing of hydrocarbons contaminated with carbonyl compounds which lead to the formation of undesirable insoluble polymer contaminants. In one embodiment, the antifoulant compound is a polymer having the Formula I
##STR00006##
wherein a must be present and either b or c or both b and c are present; d may or may not be present. In one embodiment, d is present. E is a repeat unit remaining after polymerization of an ethylenically unsaturated compound including carboxylic acids such as acrylic acid, sulfonic acid, phosphonic acid, or amide of such acid or mixtures thereof; E can be for example (meth) acrylic acid or (meth) acrylamide; each R.sub.1 is independently chosen from H or lower (C.sub.1-C.sub.4) alkyl. R.sub.2 is a hydroxy substituted alkyl or alkylene moiety having from about 1-6 carbon atoms, X is an anionic radical selected from the group consisting of SO.sub.3, OSO.sub.3, PO.sub.3, OPO.sub.3, or COO; M is one or more hydrogens or any water soluble cationic moiety that counterbalances the valence of the anionic radical X including but not limited to Na, K, Ca, or NH.sub.4; F is an ethylenically unsaturated hydrophobic moiety such as styrene, and its derivatives, acrylonitrile, olefin with (C.sub.1-C.sub.18) alkyl group, alkyl (meth) acrylate.
[0016] Q in repeat unit c is chosen from C.sub.1-C.sub.3 alkylene or carbonyl; m is 0 or 1 meaning that O may or may not be present, R.sub.3 is CH.sub.2--CH.sub.2--O .sub.n CH.sub.2--CHCH.sub.3--O .sub.n wherein n ranges from about 1 to 100, including 1 to 20, or R.sub.3 is hydroxylated lower (C.sub.1-C.sub.4) alkylene such as CH.sub.2--CH(OH) or CH.sub.2--CH(OH)--CH.sub.2; R.sub.4 is H, OH, SO.sub.3M, OSO.sub.3M, PO.sub.3M, OPO.sub.3M, CO.sub.2M or mixtures thereof with M being previously defined.
[0017] In Formula I above, when d is present, it is present in an amount of about 0.01-0.8 moles based on 1 mole of a. Either b or c, or both b or c if both are present, are present in a monomer ratio of a:b or a:c or a:(b+c) of 0.1-100, including 1-10. The molecular weight of polymers as set forth in Formula I is not critical as long as the polymer is water soluble or water dispersible. In some embodiments, the molecular weight can range from about 500-50,000 (Mn).
[0018] Exemplary polymers that may be used to inhibit fouling in hydrocarbonaceous media containing carbonyl compounds such as aldehydes include acrylic acid/allyl ether copolymers such as acrylic acid/allyl hydroxylated alkyl copolymers and water soluble salt forms thereof such as acrylic acid/1-propane sulfonic acid, 2 hydroxy-3(2-propenyl oxy) mono sodium salt also referred sometimes to as acrylic acid/allyl hydroxypropyl sulfonate ether (AHPSE). Additionally, terpolymers comprising acrylic acid/AHPSE/and styrene repeat units can also be mentioned.
[0019] Acrylic acid/ethoxylated allyl ethers such as those enumerated in U.S. Pat. No. 7,094,852 can also be mentioned as exemplary. These include acrylic acid/allylpolyethoxylated copolymers such as acrylic acid/allylpolyethoxy (10) sulfate (APES) and others. Also, acrylic acid/allyl polyethylene glycol ethers such as those set forth in U.S. Pat. No. 6,641,754 are noteworthy. One particular terpolymer of interest is a terpolymer of acrylic acid/AHPSE/and ammonium allyl polyethoxy (10) sulfate.
[0020] Other exemplary polymers can include water soluble or water dispersible acrylic acid/hydroxylated alkyl acrylates such as acrylic acid/2 hydroxypropylacrylate copolymers. Certain of the exemplary polymers are shown in Formula II-V following:
##STR00007##
[0021] The copolymer shown in Formula II may be referred to as AA/AHPSE (as herein used AA denotes acrylic acid and/or its various water soluble salt forms), and AHPSE has been previously referred to.
##STR00008##
The terpolymer shown in Formula III may be referred to as AA/AHPSE/styrene.
##STR00009##
This may be referred to as AA/AHPSE/allylpolyethoxy(10) sulfate (APES).
##STR00010##
AA/AHP SE/APES/Styrene
[0022] The antifoulant polymers may be fed to the basic (pH>7) wash tower itself or to input or recycle lines in communication with the wash tower. In some cases, the antifoulant is dosed into the caustic solution feed or recycle lines that are in fluid communication with the wash tower. Typically, the antifoulant polymers are fed to the hydrocarbon stream (charge gas) in an amount of 1-2,000 ppm by weight relative to the hydrocarbon stream. In other embodiments, the antifoulants are fed in an amount of about 1-1,000 ppm. In one embodiment, the feed rate may be from about (0.01-100)X of the antifoulant wherein X is the molar concentration of aldehyde or ketone in the charge gas.
[0023] In Formulae III and IV, a, b, and c are, independently, zero or a positive integer such that the molecular weight of the molecule is less than about 500,000 Daltons, such as from 500-500,000 Daltons. In Formula V, a, b, c, and d, are independently zero or positive integers such that the molecular weight of the molecule is less than about 500,000 Daltons.
[0024] In some aspects of the invention, the antifoulant is conjointly used with other carbonyl scavengers which can include alcohol amines such as triisopropanolamine, diglycolamine, aminomethylpropanol, N, N-diethylethanolamine, monoisopropanolamine, monoethanolamine, diethanolamine, triethanolamine, dimethylaminoethanol, and etc.; alkyl amines, such as phenothiazine, diazacyclohexane, N--N-dimethyldodecylamine, N,N'-bis(1-methylpropyl)-1,4-phenylenediamine, aminoethylpiperazine, 1,2-dianilinoethane, diethylenetriamine and etc.; keto-amines, such as triacetonamine; amino acids, such as 6 amino caproic acid; hydrazide compounds, such as 1,2-diformylhydrazine, carbohydrazide, N-methyl-hydrazide, oxalyl dihydrazide, chlorobenzhydrazide, aminobenzhydrazide, benzoic hydrazide, and etc.; hydroxylamine compounds, such as N,N-diethylhydroxylamine, isopropyl hydroxylamine, hydroxylamine sulfate, N,N-dialkylhydroxylamine, and etc.; reducing sugars, hydroxybenzenes, acetoacetate ester compounds, lactams, oxidizers, such as hydroperoxide, peroxyester, percarbonate compounds and etc.; and reducer, such as sodium borohydride, sodium (bi)sulfite and etc. These additional compounds may be present in an amount of about 1 to about 2,000 ppm by weight relative to the hydrocarbon stream.
[0025] In another aspect of the invention, novel water soluble or water dispersible polymers are provided that are useful as deposit control, scale inhibition and anti-foulant treatments in hydrocarbon media. As an example, these polymers may be used to inhibit carbonyl based polymer deposits that may otherwise form in basic washing systems employed in MTO and olefin cracking processes. The antifoulant polymers generally have the Formula VI
E .sub.a G .sub.z F .sub.d Formula VI
wherein a, z, and d are all present. E and F are as previously defined in conjunction with Formula I. G is VIa, VIb, VIc, or VId or mixtures thereof, or G is either or both of the repeat unit moieties of b and c as set forth in Formula I, wherein
VIa is
##STR00011##
[0026] VIb is
##STR00012##
[0027] wherein R.sub.1 is H or lower (C.sub.1-C.sub.4) alkyl, R.sub.2 is a hydroxy substituted alkyl or alkylene moiety having from about 1-6 carbon atoms, X is an anionic radical selected from the group consisting of SO.sub.3, OSO.sub.3, PO.sub.3, OPO.sub.3, or COO; M is H or hydrogens or any water soluble cationic moiety that counterbalances the valence of the anionic radical X; Q is chosen from C.sub.1-C.sub.3 alkylene or carbonyl, m is 0 or 1; R.sub.3 is CH.sub.2--CH.sub.2--O .sub.n; --CH.sub.2CHCH.sub.3O .sub.n wherein n=1 to 100; or R.sub.3 is hydroxylated lower (C.sub.1-C.sub.4) alkylene; and R.sub.4 is H, OH, SO.sub.3M, OSO.sub.3M, PO.sub.3M, OPO.sub.3M, or CO.sub.2M or mixtures thereof with M being as previously defined;
VIc is
##STR00013##
[0028] wherein R.sub.1 is as defined above, R.sub.5 is NH or O; R.sub.6 is lower (C.sub.1-C.sub.4) alkyl or alkylene or lower (C.sub.1-C.sub.4) hydroxy substituted alkyl or alkylene; X and M are as defined above; and VId is
##STR00014##
wherein R.sub.7 is CH.sub.2 or benzyl, and X and M are defined above.
[0029] In Formula VI, the molar ratio of d:a is about 0.1-0.8 moles of d per 1 mole a; z is present in an amount of a:z of 0.1-100 moles a per 1 mole z with certain embodiments having 1-10 moles a per 1 mole z.
[0030] Exemplary polymers in accordance with Formula VI include acrylic acid (AA)/2-acryloylamino-2-methyl-1-propanesulfonic acid (AMPS)/styrene terpolymers (i.e., Formulae VI and VIc) wherein E is AA, R.sub.5 is NH, R.sub.6=2-methylpropane, and X is SO.sub.3.sup.-; F is styrene. Additionally, Formula VI terpolymers of AA/allysulfonic acid/styrene can be noted wherein R.sub.7 in Formula VId is CH.sub.2 with X being SO.sub.3.sup.-. Also, in some embodiments, R.sub.7 can comprise a benzyl moiety with X being SO.sub.3.sup.-, namely AA/sulfonated styrene/styrene terpolymers.
[0031] Further, other terpolymeric combinations within the ambit of Figure VI include AA/AHPSE/styrene terpolymers, AA/lower alkyl (C.sub.1-C.sub.4) acrylate/styrene terpolymers, AA/hydroxylated alkyl (C.sub.1-C.sub.4) acrylate/styrene terpolymers, AA/allyl polyethylene glycol (PEG) ether/styrene terpolymers; AA/allyl polyethyoxy sulfate (APES)/styrene terpolymers and AA/PEG allyl ether/APES/styrene polymers.
[0032] The polymers of the invention can be prepared via radical chain addition polymerization of the requisite monomers. The reaction may proceed, for example, under conventional solution polymerization techniques. The requisite monomers may be mixed with water and alcohol. Polymerization initiators such as the persulfate initiators, peroxide initiators, etc., may be employed. The resulting copolymers, terpolymers, quad polymers, etc. (at least four monomeric repeat units) may be isolated by well known techniques such as distillation, etc., or the polymer may simply be used in aqueous solution.
[0033] For example, a terpolymer in accordance with Formula III can be prepared as follows:
[0034] 1. Charge initial 97.7 g (sodium 1-allyloxy 2-hydroxy propyl sulfonate) and 50 g DI water into the reactor and set up the reactor
[0035] 2. Record reactor weight
[0036] 3. Sparge with N.sub.2 for 10 minutes
[0037] 4. Switch to a nitrogen blanket and heat to 90.degree. C.
[0038] 5. Start a simultaneous co-feed of the following reagents [0039] 39 g Acrylic Acid over 150 minutes [0040] 5 g Styrene over 60 minutes [0041] 25 g Sodium persulfate (5%) over 150 minutes
[0042] 6. After feed, hold at 90.degree. C. for 90 minutes
[0043] 7. Cool to room temperature and add caustic solution (50% NaOH solution) and dilution water
[0044] 8. Measure reactor weight and solid content.
[0045] In some aspects of the invention, a water soluble terpolymer composition is provided that comprises: i) acrylic acid or acrylic acid salt repeat units, ii) a hydrophobic repeat unit such as styrene repeat units, and a third repeat unit iii). The third repeat unit may be selected from the group consisting of acrylamide repeat units, allyl ether repeat units, lower alkyl (C.sub.1-C.sub.4) acrylate repeat units, ethoxylated or protoxylated allyl repeat units, allyl polyethylene glycol ether repeat units, sulfonated styrene repeat units, and allyl sulfonic acid repeat units.
[0046] In some cases, the third repeat unit may be 2-acrylamido-2-methyl-1 propane sulfonic acid (AMPS). In other cases, the third repeat unit may be allyl hydroxypropyl sulfonate ether (AHPSE). Still, in other aspects, the third repeat unit may comprise allylpolyethoxy sulfonate (APES). Additionally, the third repeat unit may, in some aspects of the invention, comprise hydroxypropyl acrylate (HPA). In other embodiments, this third repeat unit may comprise AHPSE, and a fourth repeat unit may be present. This fourth repeat unit may comprise APES.
[0047] These terpolymer comprising repeat units i), ii), and iii) may comprise monomeric repeat units of about 0.01-0.8 moles ii) per mole of i). The repeat unit iii) may be present in an amount of about 0.1-100, including 1-10 moles i) per mole of iii). Molecular weight of these polymers may range from about 500-500,000 (Mn).
[0048] The invention will be described in conjunction with the following specific examples which are to be regarded as illustrative and not as restricting the scope of the invention.
EXAMPLES
Example 1
[0049] In order to simulate carbonyl compound fouling in caustic towers and to evaluate the dispersion capability and fouling inhibition of candidate compounds, the following procedure was employed.
TABLE-US-00001 TABLE 1 Components of simulated carbonyl compound fouling and treatment Components Concentration NaOH 10% (w/w) Dispersant/inhibitor 0-1000 ppm Acetaldehyde 2000 ppm
25 ml 10% NaOH and no dispersant (blank)/1000 ppm dispersant were charged into a 30 ml glass bottle. The bottle was capped and the solution was mixed. Then 2000 ppm aldehyde was dosed into the bottles and mixed after the bottle had been capped tightly. After that, the mixture was incubated in a water bath at 50.degree. C. for 24 hours. Finally, the mixture appearance of the bottle was recorded immediately when being taken out of the water bath without shaking. Thus, the sample untreated and samples tested with the candidates listed in Table 2 were tested. Results are shown in Table 3.
[0050] Dispersant/inhibitor candidates were selected. Some of these included known dispersant/inhibitors that are commonly used to control carbonyl based fouling. These are designated in Table 2 with the prefix "C" (comparative). Dispersant inhibitor compounds in accordance with the invention are denoted by the prefix "N".
TABLE-US-00002 TABLE 2 Candidates of inhibitor and dispersant Candidate Chemical C1 30% Hydroxylamine sulfate C2 45% Naphthalene sulfonate formaldehyde condensate C3 Poly(AA/AMPS) (43% solid) C4 PAA (50% solid) C5 PMA (50% solid) C6 Sodium Ligninsulfonate (45% solid) N1 Chemical with Formula II (37% solid) N2 Chemical with Formula III (37% solid) N3 Chemical with Formula IV (50% solid) N4 Chemical with Formula V (51% solid)
AA=acrylic acid or salt thereof; poly (AA/AMPS)=poly (acrylic acid/2-acrylamido-2-methylpropane sulfonic acid); PAA=polyacrylic acid, PMA=poly maleic acid; N.sub.1=poly (acrylic acid/allyl 2-hydroxypropyl sulfonate ether (AA/AHPSE)); N.sub.2=poly (acrylic acid/AHPSE/styrene) terpolymer; N3=poly (AA/AHPSE/allylpolyethoxy (10) sulfate (APES)); N4=poly (AA/AHPSE/APES/styrene).
[0051] Carbonyl compound fouling dispersion capability of the common dispersants in water system was studied. The carbonyl compound fouling was simulated and treated as Example 1. The dispersion performance of the candidates in Table 2 was studied under 1000 ppm product dosage by weight relative to the total solution. Table 3 shows the test results. From the appearance, we can conclude that the C1, C3, C4 and C5 did not show any dispersion capability to the carbonyl compound fouling at 1000 ppm dosage. C2 and C6 possessed some dispersion capability to the formed fouling. The sample treated with N1 resulted in a homogeneous suspension with a little precipitation, no flocs observed. This indicates that N1 possesses good dispersion capability to the polymeric fouling caused by carbonyl compound, such as aldehyde in caustic tower. N2 showed the best dispersion performance without any flocs or precipitation. The hydrophobic monomer in N2 enhances its dispersion capability.
TABLE-US-00003 TABLE 3 Carbonyl compound fouling dispersion test result Candidate Result Blank Severe flocs C1 Severe flocs C2 Flocs C3 Severe flocs C4 Severe flocs C5 Severe flocs C6 Moderate Flocs N1 Homogeneous suspension with a little precipation N2 Homogeneous suspension N3 Homogenous suspension with a little precipitation N4 Homogenous suspension
Example 2
[0052] The carbonyl compound fouling dispersion performance of C2, N1 and N2 was studied under dosage from 500 to 1000 ppm. The carbonyl compound fouling was simulated and treated as Example 1. Table 4 lists the test result. From the table, it can be observed that N2 showed the best dispersion capability, 800 ppm dosage was enough to keep the fouling suspension stable under lab static test condition.
TABLE-US-00004 TABLE 4 Carbonyl compound fouling dispersion V.S. dosage Treatment reagent 500 ppm 800 ppm 1000 ppm C2 Severe flocs Flocs Flocs N1 Severe flocs Flocs Homogeneous suspension with a little precipation N2 Floc Homogeneous Homogeneous suspension suspension
Example 3
[0053] The dispersion capability was evaluated with a filtration method. The carbonyl compound fouling was simulated and treated by the procedures similar with Example 1. 200 ml 10% NaOH was charged into 500 ml glass bottle and no treatment (blank) and 1000 ppm treatment reagents, including C1, C2 and N2 were dosed into the bottles. The bottles were capped and shaken. Then, 2000 ppm aldehyde was dosed into the above solution and mixed. After that, the mixture was incubated in water bath at 50.degree. C. for 24 hours immediately. Finally, the bottle was taken out, mixed and then the suspension was filtrated with 0.8 .mu.m fiberglass filter. FIG. 1 shows the filter cakes of the fouling treated with corresponding chemicals. It can be observed that there was no fouling substance kept on the surface of the filter after being treated with N2. It shows that the fouling particle size after being treated with N2 is smaller than 0.8 .mu.m. This indicates N2 possesses excellent capability to disperse carbonyl compound fouling into small particles. This kind of capability can prevent the fouling to flocculate or precipitate, so to eliminate the jamming or blockage in the caustic tower tray or pipelines during MTO, ethylene or propylene production process.
[0054] While this invention has been described with respect to particular embodiments thereof, it is apparent that numerous other forms and modifications of this invention will be obvious to those skilled in the art. The appended claims an this invention generally should be construed to cover all such obvious forms and modifications which are within the true spirit and scope of the present invention.
* * * * *












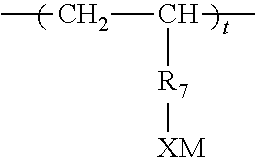





D00001

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.