Corrosion Mitigation Of Magnesium And Magnesium Alloys
Abd Elhamid; Mahmoud ; et al.
U.S. patent application number 15/690329 was filed with the patent office on 2019-02-28 for corrosion mitigation of magnesium and magnesium alloys. The applicant listed for this patent is GM GLOBAL TECHNOLOGY OPERATIONS LLC. Invention is credited to Mahmoud Abd Elhamid, Yang Guo, Ming Liu, Anil K. Sachdev.
| Application Number | 20190062926 15/690329 |
| Document ID | / |
| Family ID | 65321525 |
| Filed Date | 2019-02-28 |

| United States Patent Application | 20190062926 |
| Kind Code | A1 |
| Abd Elhamid; Mahmoud ; et al. | February 28, 2019 |
CORROSION MITIGATION OF MAGNESIUM AND MAGNESIUM ALLOYS
Abstract
A method is provided for reducing the corrosion rate of surfaces of formed magnesium or magnesium alloy articles in which the formed surface contains small embedded particles of iron. By exposing the iron particle-containing formed surface to an acidic, aqueous solution comprising alkali metal fluoride ions at a temperature of between 20.degree. C. and 30.degree. C., an adherent passivating layer of MgF.sub.2 is formed. Further, such exposure to the acidified, aqueous, fluoride ion-containing solution reduces or eliminates the concentration of cathodic, corrosion-promoting, iron-containing particles on the article surface as the magnesium fluoride layer is being formed. The development of the passivating layer reduces corrosion in a water-containing environment, and even if the passivating MgF.sub.2 layer is breached, the reduction in surface iron-containing particles reduces the inherent corrosion rate of the article.
| Inventors: | Abd Elhamid; Mahmoud; (Troy, MI) ; Guo; Yang; (Shanghai, CN) ; Liu; Ming; (Shanghai, CN) ; Sachdev; Anil K.; (Rochester Hills, MI) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 65321525 | ||||||||||
| Appl. No.: | 15/690329 | ||||||||||
| Filed: | August 30, 2017 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C23F 13/02 20130101; C23C 22/34 20130101 |
| International Class: | C23F 13/02 20060101 C23F013/02 |
Claims
1. A method of reducing the corrosion rate of a formed magnesium or magnesium-based alloy article, the formed article having a formed surface which may be exposed to an aqueous environment in use, the formed surface having embedded iron-containing particles; the method comprising reacting the formed surface of the article with an acidified aqueous solution of one or more of an alkali metal fluoride compound and ammonium fluoride so as to form a coextensive layer of MgF.sub.2 on the formed surface of the article, the embedded iron-containing particles being removed from the formed surface during the reaction, the acidified fluoride ion-containing solution having a pH of less than 2.
2. The method of reducing the corrosion rate of a formed magnesium or magnesium-based article as stated in claim 1 in which an acidified solution of one or more alkali metal fluoride compounds is reacted with the surface of the formed article.
3. The method of reducing the corrosion rate of a formed magnesium or magnesium-based article as stated in claim 1 in which the reaction is conducted with the formed part and the aqueous solution being at a temperature in the range of 20.degree. C. to 30.degree. C.
4. The method of reducing the corrosion rate of a formed magnesium or magnesium-based article as stated in claim 1 in which the thickness of the formed MgF.sub.2 layer is in the range of 0.1 micrometer to 1000 micrometers
5. The method of reducing the corrosion rate of a formed magnesium or magnesium-based article as stated in claim 1 in which the fluoride ion-containing solution comprises hydrogen fluoride (HF).
6. The method of reducing the corrosion rate of a formed magnesium or magnesium-based article as stated in claim 1 in which the pH value of the aqueous solution is obtained with the addition of one of the group consisting of H.sub.2SO.sub.4 and HNO.sub.3.
7. The method of reducing the corrosion rate of a formed magnesium or magnesium-based article as stated in claim 1 in which the alkali metal earth compound is at least one of sodium fluoride or potassium fluoride.
8. The method of reducing the corrosion rate of a formed magnesium or magnesium-based article as stated in claim 1 in which the fluoride ion-containing solution comprises between 0.1 mole and 28.9 moles of fluoride ion per liter.
9. The method of reducing the corrosion rate of a formed magnesium or magnesium-based article as stated in claim 1 in which the formed surface of the magnesium article is exposed to the fluoride ion-containing solution for between 30 and 300 seconds.
10. The method of reducing the corrosion rate of a formed magnesium or magnesium-based article as stated in claim 1 in which the formed magnesium article is exposed to the fluoride ion-containing solution by dipping the magnesium article into a bath of the fluoride-containing solution.
11. The method of reducing the corrosion rate of a formed magnesium or magnesium-based article as stated in claim 8 in which the bath of the fluoride-containing solution is agitated.
12. The method of reducing the corrosion rate of a formed magnesium or magnesium-based article as stated in claim 8 further comprising agitating the article during exposure of the article to the fluoride ion-containing solution.
13. The method of reducing the corrosion rate of a formed magnesium or magnesium-based article as stated in claim 1 in which the magnesium article is exposed to the fluoride ion-containing solution by spraying the fluoride-containing solution on the magnesium article.
14. The method of reducing the corrosion rate of a formed magnesium or magnesium-based article as stated in claim 1 further comprising rinsing, drying and optionally baking the article, after terminating exposure of the magnesium article to the fluoride ion-containing solution.
15. A formed magnesium article, the formed article having a formed surface which may be exposed to an aqueous environment in use, the formed magnesium article having on its surface, a co-extensive layer of MgF.sub.2 with a thickness ranging from 0.1 micrometer to 1000 micrometers, the article having a surface concentration of iron-containing particles which is less than 50% of the volume concentration of iron-containing particles in the body of the article.
16. A formed magnesium article as stated in claim 15 in which the layer of magnesium fluoride was formed by reacting the formed surface of the article with an acidified aqueous solution of one or more of an alkali metal fluoride compound and ammonium fluoride.
17. A formed magnesium article as stated in claim 15 in which the layer of magnesium fluoride was formed by reacting the formed surface of the article with an acidified aqueous solution of an alkali metal fluoride compound.
18. A formed magnesium article as stated in claim 15 in which the layer of magnesium fluoride was formed by reacting the formed surface of the article with an acidified aqueous solution of at least one of sodium fluoride and potassium fluoride.
Description
TECHNICAL FIELD
[0001] This disclosure describes methods of reducing the rate of corrosion at formed surfaces of manufactured articles composed of magnesium and its alloys. Magnesium alloy articles which are exposed to water, particularly salt-containing water, tend to be vulnerable to reactions on their surfaces which lead to unattractive and debilitating corrosion of the magnesium.
BACKGROUND OF THE INVENTION
[0002] Magnesium and magnesium-based alloys offer attractive strength-to-weight ratios when compared to other structural alloys such as steels and aluminum alloys. For this reason, magnesium is favorably considered when substitution of higher strength-to-weight materials is considered, for example, in aerospace applications and for weight reduction in automotive vehicles and other consumer goods.
[0003] Cars, vans, trucks, and motorcycles may have components formed of magnesium-based alloys (typically containing 85-90% by weight or more magnesium) such as door inner panels, wheels, control arms, oil pans, and engine blocks. The surfaces of such manufactured components on vehicles may be exposed to environmental conditions, especially water-containing conditions, which may promote corrosion. Magnesium is highly reactive, and, unlike aluminum and its alloys, does not form a protective oxide coating. In addition, magnesium and its commercial alloys commonly contain metal impurities which may set up galvanic corrosion cells within a magnesium article and accelerate its corrosion. Corrosion of magnesium may be exacerbated when any aqueous environment to which the magnesium is exposed incorporates high concentrations of ions, such when the road de-icing salts, typically employed in snow-prone and ice-prone regions, are used. These road de-icing salts, intended to promote melting of ice and snow at temperatures of less than 0.degree. C., dissolve into the water formed from the melted snow and ice.
[0004] Thus, there a continuing need to fortify formed articles of magnesium and its alloys against corrosion, especially in aggressively-corrosive environments, so that its full potential for reduction of weight in vehicle components may be realized.
SUMMARY OF THE INVENTION
[0005] This disclosure describes a method of enhancing the corrosion resistance of surfaces of formed articles of magnesium or magnesium alloys, hereinafter typically referred to as a formed magnesium alloy article. The enhanced corrosion resistance is particularly helpful when the magnesium alloy article may be exposed to an aqueous environment, possibly with a high ion concentration from the dissolution of road de-icing salts. An underlying aspect of the subject method is our recognition that iron, when present as elemental iron or as an iron-containing phase or compound on the surface of a magnesium alloy article (or embedded in the surface of the article), presents a local electrochemical environment that renders the surrounding magnesium susceptible to galvanic corrosion. The iron or iron-containing material, even as a micro-scale particle on the surface of a magnesium alloy article, may function as a cathode in an electrochemical corrosion reaction that leads to oxidation of nearby anodic magnesium. The iron may be present in the original alloy composition, although the specifications of most magnesium alloys limit iron content to less than fifty parts per million of total magnesium alloy content. More commonly, the iron may have been deposited into or onto the article as the magnesium alloy was being cast into the shape of the article or when the article was being machined or otherwise formed with iron-containing tools. Molten magnesium alloy may also dissolve iron from casting dies.
[0006] In accordance with the methods of this invention, surfaces of a manufactured (formed) magnesium alloy article are suitably cleaned of residual material from the tools and processes by which it was formed. The cleaning methods are conducted to suitably expose the surface of the magnesium alloy article, including regions in which small (sometimes micrometer-size) particles of iron (or containing iron) are firmly attached or embedded. When iron in such forms may be present in the surface of a magnesium alloy article, the whole surface or selected surface regions of the article are subjected to the following processing.
[0007] A reactive aqueous solution is formed of acidified fluoride ions that is composed to remove iron-based cathode sites from the surface(s) of the magnesium alloy article while concurrently forming a thin protective, water-resistant layer of magnesium fluoride (MgF.sub.2) on the article surface. For example, the aqueous solution may contain up to about ten percent by weight of sodium fluoride and/or potassium fluoride at a pH<2. Ammonium fluoride may also be used in combination with or in place of an alkali metal fluoride compound. The solution of the alkali metal fluoride and/or ammonium fluoride salts may be acidified by the addition of a suitable amount of hydrogen fluoride or of nitric acid or sulfuric acid. The solution may be applied to the surface of the magnesium alloy article, for example, at a temperature in the range of 20.degree. C. to 30.degree. C., such as a typical ambient or room temperature. The aqueous solution reacts with the article surface to progressively form a thin protective layer of magnesium fluoride and to dissolve iron material as the protective layer is formed around the iron-containing site on the surface of the article. The fluoride salt-containing aqueous solution is maintained in contact with the surface for a period of minutes (e.g., thirty seconds to five minutes) to form the conforming magnesium fluoride surface layer having a desired thickness, typically in the range of 0.1 micrometer to 1000 micrometers.
[0008] Following the formation of the MgF.sub.2 protective layer and the removal of the iron sites, the treated surface may be rinsed with water, dried, and, optionally, baked to consolidate the protective layer.
[0009] The above-described process promotes enhanced corrosion resistance of the formed magnesium article for two reasons. First, the passivating MgF.sub.2 layer will inhibit magnesium corrosion. Second, even if the passivating MgF.sub.2 layer is breached, the thus-exposed treated magnesium surface will be depleted in iron-containing particles resulting in a treated magnesium surface which is inherently less corrodible.
BRIEF DESCRIPTION OF THE DRAWINGS
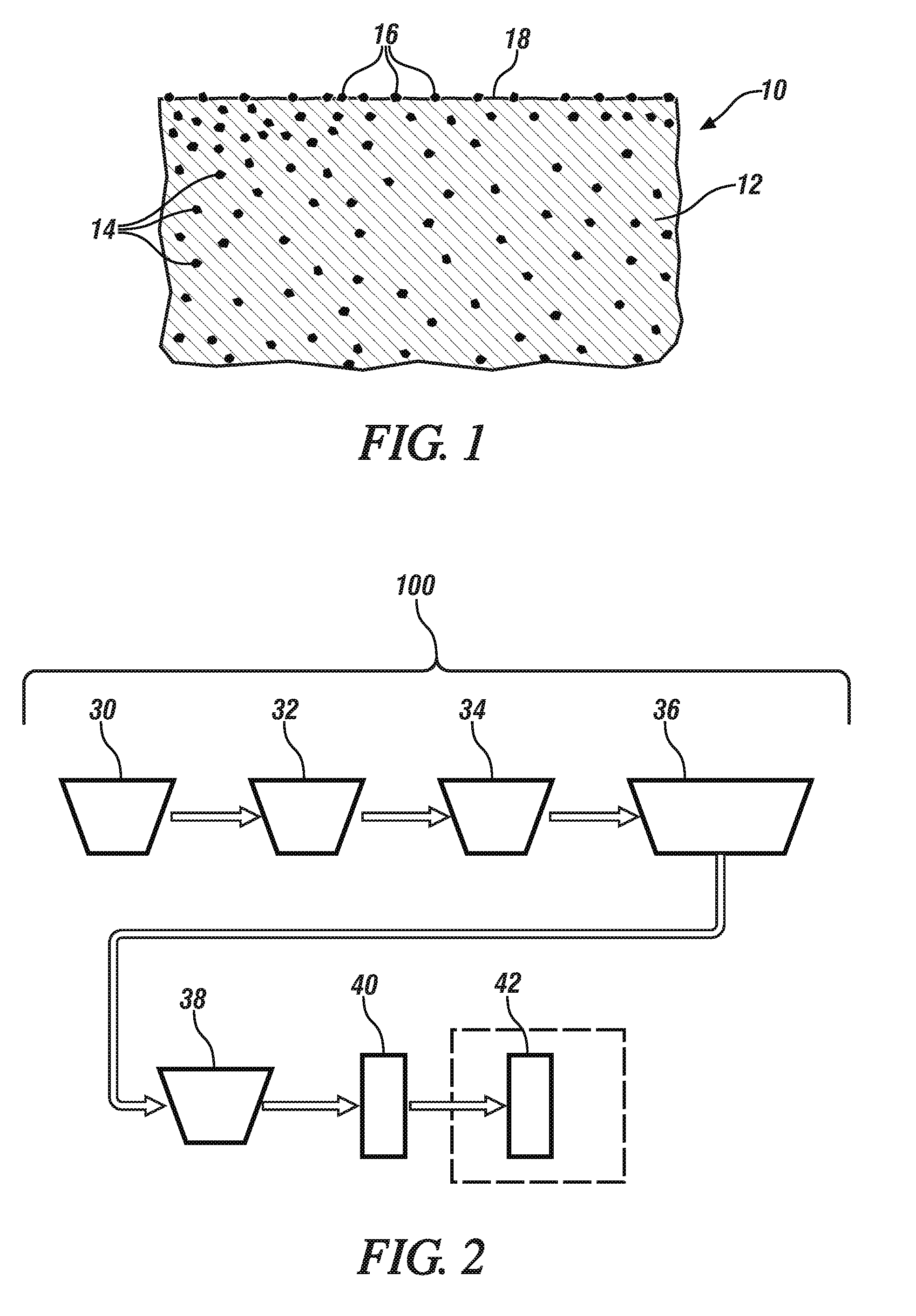
[0010] FIG. 1 shows, in schematic enlarged cross-section, a portion of a magnesium alloy article, such as a portion of a die cast transfer case housing or an inner door panel for an automobile. The formed magnesium alloy article or part has iron-containing particles. Some of the iron-containing particles are located within the microstructural cross-section of the formed article, while other particles are embedded into a surface of the article and are at least partially exposed to an environment. The iron particles in the surface of the formed article present cathode sites, potentially leading to electrochemical conversion (corrosion) of adjacent magnesium atoms.
[0011] FIG. 2 illustrates a flow diagram for a process which improves the corrosion resistance of a magnesium alloy article such as that shown at FIG. 1. The process comprises (i) cleaning and removing surface contaminants from the formed magnesium alloy panel, (ii) exposing the surface of the panel to an acidic, aqueous, fluoride ion-containing solution followed by (iii) rinsing, drying and, optionally, baking the solution-treated article.
[0012] FIGS. 3A, 3B, 3C illustrate a suggested sequence of changes to the surface of the magnesium alloy panel of FIG. 1 resulting from exposure of the surface shown in FIG. 1 to the aqueous, acidic, fluoride ion-containing solution during the process illustrated in FIG. 2. Initially, as shown at FIG. 3A, local regions of the magnesium alloy surface react with the fluoride ion-containing solution to form `patches` of a passivating layer of MgF.sub.2 on the surface of the shaped and formed magnesium alloy article. On continued exposure of the surface to the fluoride ion-containing solution, the `patches` of MgF.sub.2 expand and spread across the entire surface of the article, except for those regions where the surface-located iron-containing particles occur as shown at FIG. 3B. On yet further exposure of the surface of the formed magnesium alloy article to the fluoride ion-containing solution, the iron-containing particles dissolve into the acidic solution and the MgF.sub.2 passivating layer becomes thicker, of variable thickness, and extends across the entire treated article surface as shown at FIG. 3C.
DESCRIPTION OF PREFERRED EMBODIMENTS
[0013] This disclosure aims at enhancing the corrosion behavior of magnesium in formed magnesium and magnesium alloy articles of manufacture by the treatment of vulnerable surfaces of the sized and shaped article with acidic aqueous solutions of alkali metal fluorides. For example, vulnerable surfaces of the magnesium article are those surfaces which are exposed to water, especially salty water, in the use of the article. The treatment process is practiced to eliminate the iron particulates in the surface of the article, which particles otherwise present cathodic sites that electrochemically accelerate the oxidation and dissolution (corrosion) of adjacent magnesium atoms in the surface. The removal of the iron particulates is achieved as a passivating magnesium fluoride (MgF.sub.2) layer is deposited on the article surface being treated with the alkali metal fluorides.
[0014] It is known that commercial magnesium alloys contain metal impurities distributed as dispersed metal-containing phases such as particles or precipitates in a magnesium matrix. These metal-containing particles are cathodic to magnesium and so, in the presence of a suitable electrolyte, a galvanic reaction is likely to occur which leads to the anodic oxidation (corrosion) of nearby magnesium atoms. Iron is a particularly problematic metal in magnesium alloys, and it is generally accepted that, for acceptable corrosion performance, the iron content of pure magnesium should be maintained below 170 parts per million (ppm) by weight and, for magnesium alloys, below 40 ppm by weight. Exceeding these limits may lead to a dramatic increase in corrosion rate of magnesium. Iron is substantially insoluble in magnesium, unlike the common magnesium alloying agents, aluminum and zinc. Manganese, frequently used in concentrations of up to 0.6 weight percent in zinc alloys, is soluble in magnesium as are some other impurities commonly found in magnesium alloys such as nickel, but generally, any iron present exists as a dispersion of highly iron-rich, iron containing particles in the magnesium-rich matrix.
[0015] These restrictions on iron concentrations result from the requirement that, for galvanic corrosion to occur, any suitable electrolyte must be able to simultaneously access the magnesium matrix and any iron-containing phases. Thus, since a magnesium alloy article may have an arbitrary surface, any section cut through a magnesium body must expose sufficient iron-containing phase to promote corrosion.
[0016] As an example, consider a die-cast transfer case housing, which is a container for a driveline mechanism in an automotive vehicle. Such a transfer case housing may be die-cast from a magnesium alloy containing less than 0.004 percent by weight of iron. The die-cast housing may then be further processed by machining, for example by introduction of holes by drilling, possibly followed by honing or threading, or the creation of bearing support surfaces or sealing faces by milling. Thus, the housing, when ready to receive the transfer gearing/mechanism, will typically exhibit iron on its surfaces shaped by contact with the die casting die and surfaces shaped by subtractive machining processes. Any of these exterior surfaces may be expected to carry exposed iron-containing particles, and any of these exterior surfaces may be exposed to potentially corrosion-inducing aqueous road splash in service.
[0017] A representative, schematic, cross-sectional view of a portion of an article, such as transfer case housing, incorporating iron-containing particles is shown at FIG. 1 which shows, in fragmentary, cross-sectional view, a portion of a magnesium alloy article 10 comprising a magnesium matrix or microstructure 12. Iron-containing particles 14 are shown internal to the magnesium alloy matrix 12 with additional particles 16 shown located (typically at least partially embedded) at article surface 18 which may be exposed to a corrosion-promoting environment. As the surface of the magnesium alloy article corrodes, releasing surface-located, iron-containing particles 16 and removing them from contact with the magnesium matrix 12, previously internally-located, iron-containing particles 14 will be exposed to maintain the progress of the corrosion. It will be appreciated that other particles, out of the plane of the illustrated section in FIG. 1, that is, either above or below the plane of section, will also contribute to the corrosion of the magnesium even though not visible in the figure.
[0018] Although corrosion of magnesium promoted by iron-containing phases or particles is largely dependent on the overall iron content of the magnesium, any such corrosion may be exacerbated by pick-up of iron during manufacture of a magnesium article. Examples include the pick-up of iron particles from dies, such a stamping or die-casting dies. Any iron particles picked up during article processing tend to be particularly problematic since the iron is typically located on the finished article surface. Thus, as illustrated in FIG. 1, the concentration of iron-containing particles 16 on surface 18 may be substantially greater than the concentration of iron-containing particles 14 embedded in magnesium matrix 12.
[0019] Most magnesium articles of commerce are formed of selected magnesium based alloys. A few examples of such alloys include AZ91D die cast or wrought (extruded or sheet) magnesium alloy, AZ31B die cast or extruded (extruded or sheet) magnesium alloy, and AM60B die cast magnesium alloy. As those of skill in the art appreciate, these magnesium alloys vary in composition and microstructure, and may incorporate, in their microstructure, a plurality of metallurgical phases. The representation of the magnesium matrix and its absence of any microstructural detail in FIG. 1 and also in FIGS. 3A, 3B, and 3C is neither intended to, nor should be interpreted as, representative of, or suggestive of, any specific magnesium composition or alloy.
[0020] In general, corrosion of the surfaces of a magnesium alloy article may be inhibited in two ways. The surface of the article may be coated with a passivating, or non-corroding layer, thereby denying any aqueous, corrosion-promoting composition access to the magnesium surface. In a second approach, the sources of magnesium corrosion such as iron-containing particles may be removed from the surface of the magnesium alloy article. In accordance with practices of this invention, an acidic aqueous solution of alkali metal fluorides is used to react with the surface of the magnesium-based alloy article to progressively form a thin layer of magnesium fluoride (MgF.sub.2). Ammonium fluoride may also be used to form the magnesium fluoride layer. And as magnesium fluoride is formed around iron-containing particles, the particles are dissolved in the acidic solution. Suitable alkali metal fluorides include sodium fluoride and potassium fluoride. An inorganic acid such as hydrogen fluoride or a mineral acid such as nitric acid and/or sulfuric acid is added to the fluoride salt solution to reduce the pH of the solution to 2 or lower. Preferably this reactive, acidic, metal fluoride, and/or ammonium fluoride solution is suitably applied to the surface(s) of a formed magnesium alloy article after the surfaces have been cleaned of processing lubricants or aids used during the forming of the article or otherwise found on the surface of the article which could interfere with the chemical action of the acidic metal fluoride solution.
[0021] A suitable procedure for effectuating both the development of a passivating surface layer on a magnesium article and substantially eliminating cathodic iron-containing particles from the surface of a magnesium article is shown in FIG. 2. An as-fabricated magnesium article to be rendered more corrosion resistant is subjected to a process 100 which encompasses a plurality of sequential operations 30, 32, 34, 36, 38, 40 and (optionally 42). The process 100 may be conducted in a number of different vessels, vats, pots, enclosures, containers or the like, each having a unique environment with the article being transported from one vat or like container to another. Alternatively, the article may be placed in a single vat, pot, enclosure or container and remain in that vat etc. as the environment in the vat is successively modified to conduct the individual operations 30, 32 etc. Any such vat etc. may be sized and shaped to fully contain the article to be processed.
[0022] Several of the steps of process 100 comprise exposing the surface of the magnesium article to a liquid reactant or cleaning solution. This exposure may result from immersing the article in quiescent bath of liquid or may include agitating either or both of the liquid and the article. Alternatively, liquid, dispensed at a suitable pressure generally ranging from 50 to 2000 psi, may be sprayed on the surface of the article. To assure substantially uniform coverage of the surface if spray application is selected, the liquid may be dispensed through either a plurality of spray-heads arranged to ensure uniform or near-uniform coverage of the surface or some means of moving the article relative to the spray-head to achieve near-uniform coverage may be employed. Any combination of these liquid application processes may be used to accomplish the plurality of operations comprising process 100.
[0023] Operations 30, 32, and 34 clean and prepare the article surface and are intended to remove, among others, any loose debris, water-soluble contaminants and grease from the article surface. Operation 30 is a water rinse suitably conducted at a temperature of from about 20.degree. C. and to about 50.degree. C. Suitably operation 30 may be conducted for between 30 and 120 seconds, which rinse period can be reduced using pressurized water. Operation 32 is a degreasing step conducted at between about 20.degree. C. and 60.degree. C. for a period of between 30 and 300 seconds using, for example, trichloroethylene or tetrachloroethylene as the degreasing agent. This can be followed by the use of an alkaline cleaner, such as an aqueous solution of sodium carbonate and trisodium phosphate, at 60-80.degree. C. for 60 to 180 seconds Operation 34 is a final rinse, using de-ionized water at a temperature of between about 20.degree. C. and no greater than 50.degree. C., and carried out for between 30 and 300 seconds.
[0024] Operation 36 is the operation which endows the magnesium alloy article with its corrosion-resisting characteristics resulting from forming a passivating MgF.sub.2 layer on the article surface and eliminating any iron-containing particles on the article surface. Operation 36 entails exposing the magnesium article surface to an acidified, fluoride ion-containing, aqueous solution with a pH of less than 2. Preferably, the fluoride ions are provided by an alkali metal fluoride such as sodium fluoride and/or potassium fluoride. Ammonium fluoride may be used alone or in combination with one or more alkali metal fluorides. The fluoride ion-containing solution is suitably maintained in an ambient temperature range of, for example, between 20.degree. C. and 30.degree. C. The magnesium alloy article is suitably exposed to the solution for a period of between 30 and 300 seconds. Such exposure is sufficient to generate, on the formed article surface, an adherent, passivating, layer of MgF.sub.2 ranging in thickness from 0.1 micrometer to about 1000 nanometers. A magnesium fluoride layer thickness in the range of two to one hundred micrometers is generally suitable. The acidification of the fluoride ion-containing solution may be accomplished using HF, which serves both as a source of fluoride ions as well as an acidifying agent, or with a mineral acid such as H.sub.2SO.sub.4 and H.sub.2NO.sub.3, added in sufficient concentration to generate the required acidity. As stated, one or more alkali metal fluoride(s) is a suitable and preferred source of the fluoride ions used to form the magnesium fluoride passivation coating. Suitably the molar concentration of fluoride ions should range from 0.1M to 28.9M.
[0025] Operation 38 is a rinse to remove remnant fluoride ion-containing solution and employs water at a temperature of between about 20.degree. C. and 30.degree. C. Suitably the article should be exposed to the rinse water for a period 30-120 seconds.
[0026] Operation 40 is a drying operation which may be conducted by exposing the magnesium article to a heated air flow at a temperature of 100.degree. C. for a period of from 30 to 120 seconds. Alternatively, the rinsed parts may be simply dried in ambient air.
[0027] Operation 42, which is optional, is a bake operation, generally conducted at a higher temperature of up to 200.degree. C. to more rapidly remove all retained or absorbed water from the MgF.sub.2 layer. Operation 42 will commonly employ a heat source such as an oven, furnace or the like maintained at a temperature of 300.degree. C. But forced hot air or heat lamps may also be used to elevate the part temperature. Generally, exposure of the article to the heat source for a period of 30-120 seconds will be sufficient to raise the article temperature to between 40-200.degree. C. and remove all water from the MgF.sub.2 layer. Alternatively, the removal of residual water may be part of a subsequent heat treatment of the magnesium alloy part that could also be used to remove absorbed water.
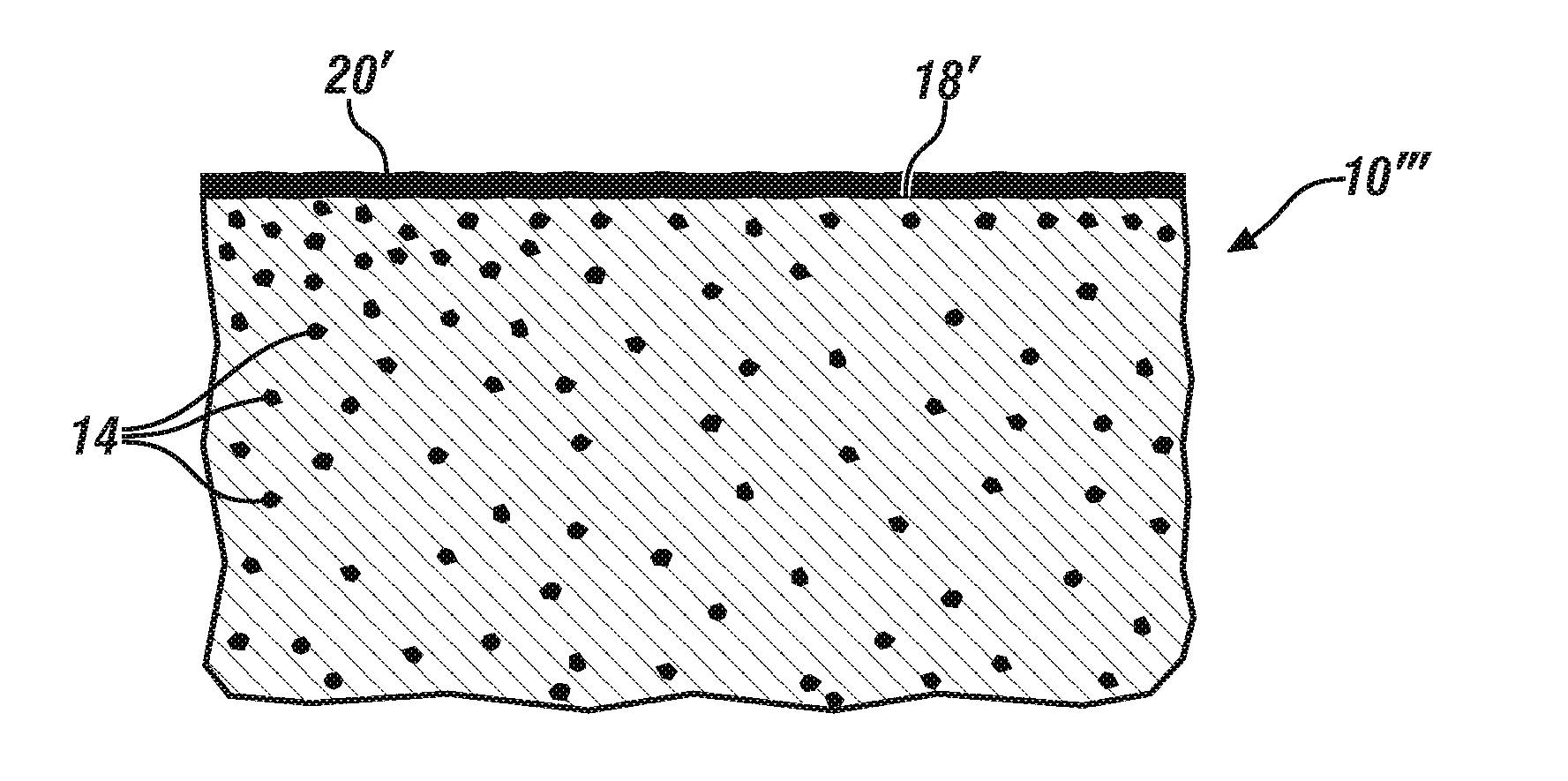
[0028] Although not relied upon, it is believed that the development of the passivating MgF.sub.2 layer and the reduction or elimination of surface-located iron-containing particles occurs by the mechanism shown in FIGS. 3A, 3B, and 3C. FIG. 3A illustrates a representative portion of an outer surface 18 of a formed magnesium alloy article 10' shortly after the magnesium particle 16 containing surface 18 was exposed to the acidic, fluoride ion-containing solution employed in operation 36 of FIG. 2. Magnesium particles 14 within the body of the article 10' are not affected by the treatment of the surface 18. As shown at FIG. 3A, localized regions or `islands` of MgF.sub.2 20 have formed on the most reactive portions of the magnesium surface 18. With continued exposure to the fluoride ion-containing solution, the extent of the magnesium surface 18 which has reacted and is now covered by an increased number of layers 20 of MgF.sub.2 continues to increase as indicated in the changed article 10''. At the completion of the layer forming process, as shown at FIG. 3C, layer 20' covers substantially the entirety of the treated surface 18' of the treated article 10''. The iron particles 14 within the microstructure of the article 10''' are not affected by the formation of the magnesium fluoride layer 18'.
[0029] When such nearly full coverage of the original article surface 18 is achieved, the iron-containing particles which were cathodic to magnesium, become anodic with respect to the MgF.sub.2 layer now covering the surface of the magnesium alloy article. The iron-containing particles which did not initially react with the fluoride ion-containing solution, because of the preference of the solution to react with magnesium, now begin to react with, and be dissolved by, the solution. Upon complete dissolution of the iron-containing particles fresh magnesium surface is exposed below the (now-removed) particles and additional MgF.sub.2 forms on the newly-exposed magnesium surface. This, in combination with some growth (thickening) of the MgF.sub.2 layer results in the formation of the thickened MgF.sub.2 layer 20' overlying a slightly roughened magnesium article surface 18'.
[0030] By the precepts of quantitative metallography, the area fraction of iron-containing particles on any arbitrary plane is equal to the volume fraction of iron-containing particles in the bulk. Thus, dissolution of surface iron-containing particles will impact the corrosion rate of the article analogously to reducing the bulk iron concentration in the magnesium.
[0031] The above mechanism relies upon MgF.sub.2 initially completely covering the magnesium article surface except for the iron-containing particles, and then, after dissolution of the iron-containing particles, the entirety of the magnesium surface. With particular reference to magnesium alloys, a plurality of phases, in addition to substantially pure magnesium, may be present, including magnesium-rich phases such as Mg.sub.17Al.sub.12 as well as magnesium-free phases such as MnAl resulting from the occurrence of manganese as an impurity in, for example, magnesium AZ91 alloys.
[0032] In general, the magnesium-rich phases in a magnesium alloy respond to the fluoride-ion containing solution analogously to substantially pure magnesium and form MgF.sub.2. The behavior of other, magnesium-free, phases on the surface is less clear, but it will be appreciated that, provided coverage of any magnesium-containing regions of the surface with MgF.sub.2 is complete, any corrosion-inducing liquid will be denied access to the underlying magnesium. Thus, even in the presence of highly-corrosive liquids, corrosion of the magnesium will be inhibited. In general, substantially complete coverage of the surface is observed as a result of the formation of the thermodynamically favored MgF.sub.2 layer thus formed on the surface of the magnesium or magnesium alloy article.
[0033] Although such theory is not relied upon, it is supported by the results of the following experiment. A 99.9% Mg electrode was connected to a 99.95% Fe electrode, and the electrode pair was immersed in a 2 wt. % HF aqueous solution (1M of fluoride ions) prepared from de-ionized water and containing a trace quantity of potassium ferricyanide as an indicator. Upon immersion, initially, the Mg electrode turned black due to the formation of MgF.sub.2, while H.sub.2 evolved on the Fe electrode. After the Mg electrode was fully covered with MgF.sub.2, and thus passivated, the Fe started to dissolve as indicated by the development of a blue color in the solution--potassium ferricyanide reacts to form ferrous ferricyanide (Prussian blue) in the presence of Fe.sup.++ ions. Continued immersion of the electrodes resulted in the dissolution of yet more iron as indicated by the deepening of the blue color of the solution as a result of the formation of more ferrous ferricyanide.
[0034] The above detailed description and the associated drawings or figures are presented for illustration of suitable exemplary embodiments and not for limitation of the following claims.
* * * * *
D00000

D00001

D00002

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.