Copper Alloy Material
YANO; Shoichiro ; et al.
U.S. patent application number 15/771847 was filed with the patent office on 2019-02-28 for copper alloy material. This patent application is currently assigned to MITSUBISHI MATERIALS CORPORATION. The applicant listed for this patent is MITSUBISHI MATERIALS CORPORATION. Invention is credited to Kanta DAIRAKU, Toshio SAKAMOTO, Shoichiro YANO.
| Application Number | 20190062874 15/771847 |
| Document ID | / |
| Family ID | 58695049 |
| Filed Date | 2019-02-28 |




| United States Patent Application | 20190062874 |
| Kind Code | A1 |
| YANO; Shoichiro ; et al. | February 28, 2019 |
COPPER ALLOY MATERIAL
Abstract
A copper alloy material has a composition including: 0.3 mass % or more and less than 0.5 mass % of Cr; 0.01 mass % or more and 0.15 mass % or less of Zr; and a Cu balance including inevitable impurities, wherein an average of crystal grain sizes is in a range of 0.1 mm or more and 2.0 mm or less, and a standard deviation of the crystal grain sizes is 0.6 or less.
| Inventors: | YANO; Shoichiro; (Iwaki-shi, JP) ; DAIRAKU; Kanta; (Iwaki-shi, JP) ; SAKAMOTO; Toshio; (Kitamoto-shi, JP) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | MITSUBISHI MATERIALS
CORPORATION Tokyo JP |
||||||||||
| Family ID: | 58695049 | ||||||||||
| Appl. No.: | 15/771847 | ||||||||||
| Filed: | October 11, 2016 | ||||||||||
| PCT Filed: | October 11, 2016 | ||||||||||
| PCT NO: | PCT/JP2016/080125 | ||||||||||
| 371 Date: | April 27, 2018 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C22F 1/08 20130101; B22C 9/061 20130101; C22C 9/00 20130101; C22C 9/01 20130101 |
| International Class: | C22C 9/01 20060101 C22C009/01; C22F 1/08 20060101 C22F001/08 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Nov 9, 2015 | JP | 2015-219852 |
Claims
1. A copper alloy material having a composition including: 0.3 mass % or more and less than 0.5 mass % of Cr; 0.01 mass % or more and 0.15 mass % or less of Zr; and a Cu balance including inevitable impurities, wherein an average of crystal grain sizes is in a range of 0.1 mm or more and 2.0 mm or less, and a standard deviation of the crystal grain sizes is 0.6 or less.
2. The copper alloy material according to claim 1, wherein an area ratio of crystallized Cr in cross section observation is 0.5% or less.
3. The copper alloy material according to claim 1, wherein, after performing a heat treatment at 1000.degree. C. for 1 hour retention, an average of crystal grain sizes is in a range of 0.1 mm or more and 3.0 mm or less, and a standard deviation of the crystal grain sizes is 1.5 or less.
4. The copper alloy material according to claim 1, wherein the composition of the copper alloy material further includes Al in a range of 0.1 mass % or more and 2.0 mass % or less.
5. The copper alloy material according to claim 1, wherein the composition of the copper alloy material further includes one or more elements selected from Fe, Co, Sn, Zn, P, Si and Mg in a range of 0.005 mass % or more and 0.1 mass % or less as a total.
Description
CROSS-REFERENCE TO RELATED PATENT APPLICATIONS
[0001] This application is a U.S. National Phase Application under 35 U.S.C. .sctn. 371 of International Patent Application No. PCT/JP2016/080125 filed on Oct. 11, 2016 and claims the benefit of Japanese Patent Application No. 2015-219852, filed on Nov. 9, 2015, all of which are incorporated herein by reference in their entireties. The International Application was published in Japanese on May 18, 2017 as International Publication No. WO/2017/081972 under PCT Article 21(2).
FIELD OF THE INVENTION
[0002] The present invention relates to a copper alloy material suitable for a part used in a high temperature environment such as a molding material for casting and a welding part such as a contact tip.
BACKGROUND OF THE INVENTION
[0003] Conventionally, Cu--Cr--Zr-based alloys such as C18150 are used as a material for casting mold materials and welding members which are used at high temperatures, since they have excellent heat resistance and electrical conductivity as shown in Japanese Examined Patent Application, Second Publication No. S62-097748 and Japanese Unexamined Patent Application, First Publication No. H05-339688.
[0004] These Cu--Cr--Zr-based alloy are usually produced by a production process, in which a cast made of Cu--Cr--Zr-based alloy is subjected to a plastic working; a solution treatment, for example in a condition of 950-1050.degree. C. of a retention temperature and 0.5-1.5 hours of a retention time, and an aging treatment, for example in a condition of 400-500.degree. C. of a retention temperature and 2-4 hours of a retention time, are performed on the plastically worked material; and then the material subjected to the solution and aging treatments is finished into a predetermined shape by machine working in the end.
[0005] In the above-described methods, Cr and Zr are dissolved in the matrix of Cu by the solution treatment and fine precipitates of Cr and Zr are dispersed by the aging treatment to improve strength and conductivity.
Technical Problem
[0006] Although the above-described Cu--Cr--Zr-based alloy has excellent heat resistance, when it is exposed to a use environment with a peak temperature of 500.degree. C. or more, occasionally, re-solution of precipitate starts for the strength and the conductivity to be reduced and for coarsening of the crystal grain to occur.
[0007] When coarsening of the crystal grains occurs, the propagation speed of the crack increases and the service life of the product may be shortened. In addition, there has been a problem that mechanical properties such as strength and elongation are remarkably deteriorated due to local occurrence of coarsening of crystal grains.
[0008] The present invention has been made in view of the above-described circumstances. An object of the present invention to provide a copper alloy material which is stable in characteristics and excellent in service life even when it is used in a high temperature environment of 500.degree. C. or more.
SUMMARY OF THE INVENTION
Solution to Problem
[0009] To solve the above-described technical problem, an aspect of the present invention is directed to a copper alloy material having a composition including: 0.3 mass % or more and less than 0.5 mass % of Cr; 0.01 mass % or more and 0.15 mass % or less of Zr; and a Cu balance including inevitable impurities, wherein an average of crystal grain sizes is in a range of 0.1 mm or more and 2.0 mm or less, and a standard deviation of the crystal grain sizes is 0.6 or less (hereinafter, referred as "a copper alloy material of the present invention").
[0010] In the copper alloy material as configured above, the composition includes 0.3 mass % or more and less than 0.5 mass % of Cr; 0.01 mass % or more and 0.15 mass % or less of Zr; and a Cu balance including inevitable impurities. Thus, by precipitating fine precipitates by the aging treatment, strength (hardness) and conductivity can be improved. In addition, crystallized Cr is reduced since the Cr content is set to a relatively low level at 0.3 mass % or more and less than 0.5 mass %. Thus, occurrence of unevenly-sized re-crystallized grains because of accumulation of local strain due to these crystallized Cr can be suppressed. Therefore, local coarsening of crystal grains can be suppressed even in a case of being used under high temperature condition.
[0011] In addition, the average of the crystal grain sizes is set in the range of 0.1 mm or more and 2.0 mm or less in the copper alloy material of the present invention. Thus, accumulation of strain is relatively low for the copper alloy material to be re-crystallized. In addition, the standard deviation of the crystal grain sizes is set to 0.6 or less. Thus, the crystal grain sizes are uniform, accumulation of local strain is less. Accordingly, local coarsening of crystal grains can be suppressed even in a case of being used under high temperature condition.
[0012] In the copper alloy material of the present invention, it is preferable that an area ratio of crystallized Cr in cross section observation is 0.5% or less.
[0013] In this case, since the area ratio of the crystallized Cr in cross section observation is set to 0.5% or less, accumulation of local strain is less. Accordingly, local coarsening of crystal grains can be reliably suppressed even in a case of being used under high temperature condition.
[0014] In the copper alloy material of the present invention, it is preferable that after performing a heat treatment at 1000.degree. C. for 1 hour retention, an average of crystal grain sizes is in a range of 0.1 mm or more and 3.0 mm or less, and a standard deviation of the crystal grain sizes is 1.5 or less.
[0015] In this case, even after performing the heat treatment at 1000.degree. C. for 1 hour retention, crystal grains are not coarsened and the crystal grain sizes are kept relatively uniform. Accordingly, mechanical properties and conductivity are stable even in a case of being used under high temperature condition at 500.degree. C. or more.
[0016] In the copper alloy material of the present invention, the composition of the copper alloy material may further include Al in a range of 0.1 mass % or more and 2.0 mass % or less.
[0017] In this case, since the composition of the copper alloy material further includes Al in a range of 0.1 mass % or more and 2.0 mass % or less, conductivity can be adjusted to about 30-60% IACS. The copper alloy material having such a conductivity is particularly suitable as a casting mold material for electromagnetic stirring applications.
[0018] In the copper alloy material of the present invention, the composition of the copper alloy material may further include one or more elements selected from Fe, Co, Sn, Zn, P, Si and Mg in a range of 0.005 mass % or more and 0.1 mass % or less as a total.
[0019] In this case, since the composition of the copper alloy material further includes one or more elements selected from Fe, Co, Sn, Zn, P, Si and Mg in the above-described range, coarsening of crystal grain sizes can be suppressed more reliably because of the grain boundary pinning effect by a compound including these elements.
Advantageous Effects of Invention
[0020] According to the present invention, it is possible to provide a copper alloy material having stable characteristics and excellent service life even when it is used in a high temperature environment of 500.degree. C. or more.
BRIEF DESCRIPTION OF THE DRAWINGS
[0021] FIG. 1 is a flowchart of a method of manufacturing a copper alloy material that is an embodiment of the present invention.
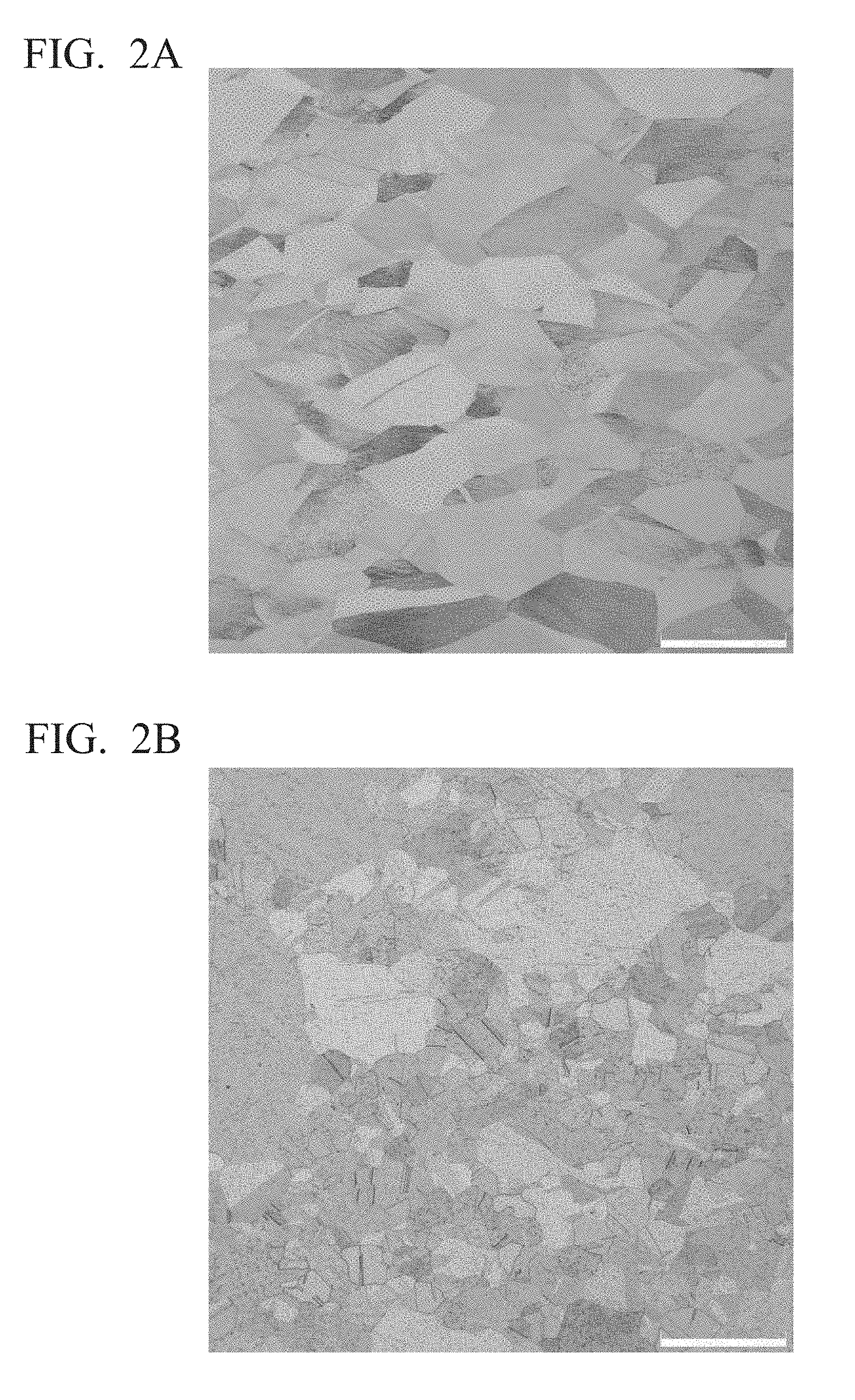
[0022] FIG. 2A is a structural observation photograph of Example 1 of the present invention.
[0023] FIG. 2B is a structural observation photograph of Comparative Example 4.
[0024] FIG. 3A is a structure observation photograph of Example 1 of the present invention after performing the heat treatment at 1000.degree. C. for 1 hour retention time.
[0025] FIG. 3B is a structure observation photograph of Comparative Example 4 after performing the heat treatment at 1000.degree. C. for 1 hour retention time.
[0026] FIG. 4A is an observation photograph of crystallized Cr and an SEM image in Example 1 of the present invention.
[0027] FIG. 4B is an EPMA (Cr) image in Example 1 of the present invention.
[0028] FIG. 4C is an observation photograph of crystallized Cr and an SEM image in Comparative Example 4.
[0029] FIG. 4D is an EPMA (Cr) image in Comparative Example 4.
DETAILED DESCRIPTION OF THE INVENTION
[0030] Hereinafter, a copper alloy material is an embodiment of the present invention will be described.
[0031] The copper alloy material of the present embodiment is a material for a part used in a high temperature environment such as a molding material for casting and a welding part.
[0032] The copper alloy material according to the present embodiment has a composition including: 0.3 mass % or more and less than 0.5 mass % of Cr; 0.01 mass % or more and 0.15 mass % or less of Zr; and a Cu balance including inevitable impurities. In the copper alloy material of the present embodiment, Al may be contained in the range of 0.1 mass % or more and 2.0 mass % or less, if necessary. Further, one or more elements selected from Fe, Co, Sn, Zn, P, Si and Mg may be contained in a total amount of 0.005 mass % or more and 0.1 mass % or less.
[0033] In the copper alloy material of the present embodiment, the average of crystal grain sizes is in a range of 0.1 mm or more and 2.0 mm or less, and the standard deviation of the crystal grain sizes is 0.6 or less.
[0034] In addition, in the copper alloy material of the present embodiment, the area ratio of crystallized Cr in cross section observation is 0.5% or less.
[0035] The area ratio of the crystallized Cr is determined by observing the structure of an arbitrary cross section of the copper alloy material (for example, a cross section parallel to the rolling direction) with microscopic etching, observing the structure with SEM or the like, and analyzing it.
[0036] Furthermore, in the copper alloy material of the present embodiment, after performing a heat treatment at 1000.degree. C. for 1 hour retention, the average of crystal grain sizes is in a range of 0.1 mm or more and 3.0 mm or less, and the standard deviation of the crystal grain sizes is 1.5 or less.
[0037] The reasons why the composition of the copper alloy material according to the present embodiment, the crystal structure and the like are defined as described above will be described below.
[0038] (Cr: 0.3 Mass % or More and Less than 0.5 Mass %)
[0039] Cr is an element having an action effect that improves strength (hardness) and electrical conductivity by finely precipitating Cr-based precipitates in crystal grains of the parent phase by means of an aging treatment.
[0040] Here, in a case where the content of Cr is less than 0.3 mass %, the precipitation amount during the aging treatment becomes insufficient, and there is a concern that the strength (hardness) improvement effect cannot be sufficiently obtained. In addition, in a case where the content of Cr is 0.5 mass % or more, a relatively large amount of crystallized Cr exist even after the solution treatment; local strain is accumulated due to these crystallized Cr; and sizes of recrystallized grains becomes uneven. When it is used under a high temperature environment, it is possible that the crystal grains are coarsened.
[0041] On the basis of what has been described above, in the present embodiment, the content of Cr is set in a range of 0.3 mass % or more and less than 0.5 mass %. Meanwhile, in order to reliably exhibit the above-described action effect, the lower limit of the content of Cr is preferably set to 0.35 mass % or more, and the upper limit of the content of Cr is preferably set to 0.45 mass % or less.
[0042] (Zr: 0.01 Mass % or More and 0.15 Mass % or Less)
[0043] Zr is an element having an action effect that improves strength (hardness) and electrical conductivity by finely precipitating Zr-based precipitates in the crystal grain boundaries of the parent phase by means of the aging treatment.
[0044] Here, in a case where the content of Zr is less than 0.01 mass %, the precipitation amount during the aging treatment becomes insufficient, and there is a concern that the strength (hardness) improvement effect cannot be sufficiently obtained. In addition, in a case where the content of Zr exceeds 0.15 mass %, there is a concern that electrical conductivity and thermal conductivity may decrease. In addition, even when more than 0.15 mass % of Zr is included, there is a concern that an additional strength improvement effect cannot be obtained.
[0045] On the basis of what has been described above, in the present embodiment, the content of Zr is set in a range of 0.01 mass % or more and 0.15 mass % or less. Meanwhile, in order to reliably exhibit the above-described action effect, the lower limit of the content of Zr is preferably set to 0.05 mass % or more, and the upper limit of the content of Zr is preferably set to 0.13 mass % or less.
[0046] Al: 0.1 mass % or more and less than 2.0 mass %
[0047] Al is an element having an action effect that decreases electrical conductivity by forming a solid solution in copper alloys. Therefore, it is possible to adjust the electrical conductivity of the copper alloy material to approximately 30% to 60% IACS by controlling the amount of Al added.
[0048] Here, in a case where the content of Al is less than 0.1 mass %, it becomes difficult to suppress the electrical conductivity at a low level. In addition, in a case where the content of Al is 2.0 mass % or more, there is a concern that the electrical conductivity may significantly decrease and the thermal conductivity may become insufficient.
[0049] On the basis of what has been described above, in the present embodiment, the content of Al is set in a range of 0.1 mass % or more and less than 2.0 mass % in the case of adding Al. Meanwhile, in order to reliably exhibit the above-described action effect, the lower limit of the content of Al is preferably set to 0.3 mass % or more, and the upper limit of the content of Al is preferably set to 1.5 mass % or less. In the case where Al is not intentionally added, Al may be included in the inevitable impurities at less than 0.1 mass %.
[0050] (One or More Elements Selected from Fe, Co, Sn, Zn, P, Si and Mg: Total of 0.005 Mass % or More and 0.1 Mass % or Less)
[0051] Elements of Fe, Co, Sn, Zn, P, Si and Mg are elements that form fine compounds and exhibit a pinning effect to suppress crystal growth.
[0052] Here, in a case where the total content of one or more elements selected from Fe, Co, Sn, Zn, P, Si and Mg is less than 0.005 mass %, there is a concern that the above-described pinning effect cannot be exhibited sufficiently. On the other hand, in a case where the total content of one or more elements selected from Fe, Co, Sn, Zn, P, Si and Mg exceeds 0.1 mass %, there is a concern that the electrical conductivity and the thermal conductivity may decrease.
[0053] On the basis of what has been described above, in the present embodiment, the total content of one or more elements selected from Fe, Co, Sn, Zn, P, Si and Mg is set in a range of 0.005 mass % or more and 0.1 mass % or less. Meanwhile, in order to reliably exhibit the above-described action effect, the lower limit of the total content of one or more elements selected from Fe, Co, Sn, Zn, P, Si and Mg P is preferably set to 0.02 mass % or more, and the upper limit of the total content of one or more elements selected from Fe, Co, Sn, Zn, P, Si and Mg is preferably set to 0.07 mass % or less. In addition, when elements such as Fe, Co, Sn, Zn, P, Si and Mg are not intentionally added, these elements may be contained as impurities in a total amount of less than 0.005 mass %.
[0054] (Other Inevitable Impurities: 0.05 Mass % or Less)
[0055] Meanwhile, examples of the inevitable impurities other than Cr, Zr, Al, Fe, Co, Sn, Zn, P, Si, and Mg described above include B, Ag, Ca, Te, Mn, Ni, Sr, Ba, Sc, Y, Ti, Hf, V, Nb, Ta, Mo, W, Re, Ru, Os, Se, Rh, Ir, Pd, Pt, Au, Cd, Ga, In, Li, Ge, As, Sb, Tl, Pb, Be, N, H, Hg, Tc, Na, K, Rb, Cs, Po, Bi, lanthanoids, O, S, C and the like. Since there is a concern that these inevitable impurities may decrease the electrical conductivity and the thermal conductivity, the total amount thereof is preferably set to 0.05 mass % or less.
[0056] (Average Crystal Grain Size: 0.1 mm or More and 2.0 mm or Less/Standard Deviation of Grain Size: 0.6 or Less)
[0057] In the case of having a fine crystal structure with an average crystal grain size of less than 0.1 mm, the driving force in recrystallization increases and there is a possibility that high strain is introduced locally. Therefore, when used under a high temperature environment, the crystal grains may be coarsened. On the other hand, when the average crystal grain size exceeds 2.0 mm, workability becomes insufficient and it is difficult to use industrially. Specifically, since the grain boundary strength decreases, the tensile strength and elongation are lowered and the crack propagation speed also increases, which makes it difficult to use it industrially.
[0058] In addition, when the standard deviation of the crystal grain size exceeds 0.6, the deviation of the crystal grain sizes becomes large and strain is locally accumulated. Thus, when used under a high temperature environment, the crystal grains become coarse. In addition, the mechanical properties may be deteriorated.
[0059] On the basis of what has been described above, in the present embodiment, the average crystal grain size is set in the range of 0.1 mm or more and 2.0 mm or less, and the standard deviation of the crystal grain size is set to be 0.6 or less. The lower limit of the average crystal grain size is preferably 0.15 mm or more, and the upper limit of the average crystal grain size is preferably 1.0 mm or less. In addition, it is preferable to set the upper limit of the standard deviation of crystal grain size to 0.5 or less.
[0060] (Area Ratio of Cr Crystallized Material in Section Observation: 0.5% or Less)
[0061] When the area ratio of the crystallized Cr exceeds 0.5%, the strain is locally accumulated, so that the size of the recrystallized grains is difficult to be uniform. Thus, when used in a high temperature environment, there is a possibility that crystal grains becomes coarse.
[0062] From the above, in the present embodiment, the area ratio of the crystallized Cr in the cross sectional observation is set to be 0.5% or less. The upper limit of the area ratio of the crystallized Cr is preferably 0.3% or less.
[0063] (Average Crystal Grain Size after Heat Treatment Maintained at 1000.degree. C. for 1 Hour: 0.1 mm or More and 3.0 mm or Less/Standard Deviation of Grain Size: 1.5 or Less)
[0064] By setting the average crystal grain size after the heat treatment maintained at 1000.degree. C. for 1 hour within the above range, coarsening of crystal grains when used in a high temperature environment is reliably suppressed. In addition, by setting the standard deviation after heat treatment held at 1000.degree. C. for 1 to 1.5 hours or less, occurrence of unevenly sized crystal grains when used under a high temperature environment is reliably suppressed.
[0065] From the above description, in the present embodiment, the average crystal grain size after the heat treatment maintained at 1000.degree. C. for 1 hour is set in the range of 0.1 mm to 3.0 mm, the standard deviation of the crystal grain size is 1.5 or less. The lower limit of the average crystal grain size is preferably 0.2 mm or more, and the upper limit of the average crystal grain size is preferably 0.5 mm or less. In addition, it is preferable to set the upper limit of the standard deviation of crystal grain size to 1.3 or less.
[0066] Next, a method for manufacturing the casting mold material according to the embodiment of the present invention will be described with reference to a flowchart of FIG. 1.
[0067] (Melting and Casting Step S01)
[0068] First, a copper raw material made of oxygen-free copper having a copper purity of 99.99 mass % or higher is loaded into a carbon crucible and is melted using a vacuum melting furnace, thereby obtaining molten copper. Next, the above-described additive elements are added to the obtained molten metal so as to obtain a predetermined concentration, and components are formulated, thereby obtaining a molten copper alloy.
[0069] Here, as raw materials of Cr, and Zr which are the additive elements, Cr, and Zr having a high purity are used, and, for example, Cr having a purity of 99.99 mass % or higher is used as a raw material of Cr, and Zr having a purity of 99.95 mass % or higher is used as a raw material of Zr. In addition, Al, Fe, Co, Sn, Zn, P, Si and Mg are added thereto as necessary. Meanwhile, as raw materials of Al, Fe, Co, Sn, Zn, P, Si and Mg, parent alloys with Cu may also be used.
[0070] In addition, the component-formulated molten copper alloy is injected into a die, thereby obtaining an ingot.
[0071] (Homogenization Treatment Step S02)
[0072] Next, a thermal treatment is carried out in order for the homogenization of the obtained ingot.
[0073] Specifically, a homogenization treatment is carried out on the ingot in the atmosphere under conditions of 950.degree. C. or higher and 1,050.degree. C. or lower for one hour or longer.
[0074] (Hot Working Step S03)
[0075] Next, hot rolling with a working percentage of 50% or higher and 99% or lower is carried out on the ingot in a temperature range of 900.degree. C. or higher and 1,000.degree. C. or lower, thereby obtaining a rolled material. Meanwhile, the method of the hot working may be hot forging. After this hot working, the rolled material is immediately cooled by means of water cooling.
[0076] (Solution Treatment Step S04)
[0077] Next, a heating treatment is carried out on the rolled material obtained in the hot working step S03 under conditions of 920.degree. C. or higher and 1,050.degree. C. or lower for 0.5 hours or longer and five hours or shorter, thereby carrying out a solution treatment. The heating treatment is carried out, for example, in the atmosphere or an inert gas atmosphere, and as cooling after the heating, water cooling is carried out.
[0078] (Aging Treatment Step S05)
[0079] Next, after the solution treatment step S04, an aging treatment is carried out, and precipitates such as Cr-based precipitates and Zr-based precipitates are finely precipitated, thereby obtaining a first aging treatment material.
[0080] Here, the first aging treatment is carried out under conditions of, for example, 400.degree. C. or higher and 530.degree. C. or lower for 0.5 hours or longer and five hours or shorter.
[0081] Meanwhile, the thermal treatment method during the aging treatment is not particularly limited, but the thermal treatment is preferably carried out in an inert gas atmosphere. In addition, the cooling method after the heating treatment is not particularly limited, but water cooling is preferably carried out.
[0082] By means of the above-described steps, the copper alloy material that is the present embodiment is manufactured.
[0083] According to the copper alloy material of the present invention provided with the above-described constitution, since the casting mold material is provided with a composition including 0.3 mass % or more and less than 0.5 mass % of Cr, 0.01 mass % or more and 0.15 mass % or less of Zr, and a Cu balance including inevitable impurities, fine precipitates can be precipitated by performing the solution and aging treatments. Thus, strength and electrical conductivity can be improved.
[0084] In addition, since the Cr content is set to a relatively low content of 0.3 mass % or more and less than 0.5 mass %, crystallized Cr hardly exists after the solution treatment. Specifically, the area ratio of crystallized Cr in cross section observation is 0.5% or less. Therefore, occurrence of unevenly sized recrystallized grains by accumulation of local strain due to the crystallized Cr can be suppressed. Accordingly, local coarsening of crystal grains can be reliably suppressed even when it is used under a high temperature environment.
[0085] In the present embodiment, the average crystal grain size is set in the range of 0.1 mm or more and 2.0 mm or less, and the standard deviation of the crystal grain size is set to 0.6 or less, so that there is less accumulation of local strain. Accordingly, local coarsening of crystal grains can be reliably suppressed even when it is used under a high temperature environment.
[0086] Furthermore, the average crystal grain size after the heat treatment maintained at 1000.degree. C. for 1 hour is set in the range of 0.1 mm or more and 3.0 mm or less, and the standard deviation of the crystal grain size is set to 1.5 or less. Thus, even after heat treatment at 1000.degree. C. for 1 hour retention, crystal grains are not coarsened locally and even when used under a high temperature environment of 500.degree. C. or more, mechanical properties and conductivity are stable.
[0087] Further, in this embodiment, when Al is contained in the range of 0.1 mass % or more and 2.0 mass % or less, the conductivity can be adjusted to about 30 to 60% IACS.
[0088] As a result, it is possible to obtain a copper alloy material particularly suitable as a casting mold material for electromagnetic stirring applications.
[0089] In addition, in the present embodiment, when the copper alloy material further includes one or more elements selected from Fe, Co, Sn, Zn, P, Si and Mg in a range of 0.005 mass % or more and 0.1 mass % or less as a total, coarsening of crystal grains can be more reliably suppressed by the pinning effect of a compound containing these elements.
[0090] Hitherto, the embodiment of the present invention has been described, but the present invention is not limited thereto and can be appropriately modified in the scope of the technical concept of the invention.
Examples
[0091] Hereinafter, the results of confirmation tests carried out in order to confirm the effects of the present invention will be described.
[0092] A copper raw material made of oxygen-free copper having a copper purity of 99.99 mass % or higher was prepared, was loaded into a carbon crucible, and was melted using a vacuum melting furnace (with a degree of vacuum of 10.sup.-2 Pa or lower), thereby obtaining molten copper. A variety of additive elements were added to the obtained molten copper so as to formulate a component composition shown in Table 1, the component composition was maintained for five minutes, and then the molten copper alloy was injected into a cast iron die, thereby obtaining an ingot. The sizes of the ingot were set to a width of approximately 80 mm, a thickness of approximately 50 mm, and a length of approximately 130 mm.
[0093] Meanwhile, as a raw material of Cr which was an additive element, Cr having a purity of 99.99 mass % or higher was used, and as a raw material of Zr, Zr having a purity of 99.95 mass % or higher was used.
[0094] Next, a homogenization treatment was carried out in the atmosphere under conditions of 1,000.degree. C. for one hour, and then hot rolling was carried out. The rolling reduction in the hot rolling was set to 80%, thereby obtaining a hot-rolled material having a width of approximately 100 mm, a thickness of approximately 10 mm, and a length of approximately 520 mm.
[0095] A solution treatment was carried out on this hot-rolled material under conditions of 1,000.degree. C. for 1.5 hours, and then cooled at the cooling rates shown in Table 2.
[0096] Next, a aging treatment was carried out under conditions of 500 (.+-.15).degree. C. for three hours. By following above-described procedures, the copper alloy materials were obtained.
[0097] For the obtained copper alloy material, the structure of the copper alloy material after the aging treatment was observed, and the standard deviation of the average crystal grain size and crystal grain size was measured.
[0098] In addition, the average crystal grain size and the standard deviation of the crystal grain diameter after the heat treatment for 1 hour at 1000.degree. C. retention were measured for this copper alloy material.
[0099] Further, cross-section observation was performed on the material after the solution treatment, and the area ratio of crystallized Cr was measured.
[0100] FIGS. 2A and 2B show the structure observation pictures of the copper alloy materials of Example 1 of the present invention and Comparative Example 4, respectively, after the above-described aging treatment and before the heat treatment at 1000.degree. C. for 1 hour retention.
[0101] Similarly, FIG. 3A and FIG. 3B show the structure observation pictures of the copper alloy materials of Example 1 of the present invention and Comparative Example 4, respectively, after the above-described aging treatment and the heat treatment at 1000.degree. C. for 1 hour retention.
[0102] (Composition Analysis)
[0103] The component composition of the obtained copper alloy material was measured by ICP-MS analysis. The measurement results are shown in Table 1.
[0104] (Average Crystal Grain Size and Standard Deviation of Grain Size)
[0105] A sample of 10 mm.times.15 mm from the central portion of the plate width was cut out from the obtained thickness of the copper alloy material and the surface in the rolling direction (RD direction) was polished and then micro etching was performed.
[0106] This sample was observed and the average crystal grain size was measured by the intercept method prescribed in JIS H 0501.
[0107] (Area Ratio of Cr Crystallization)
[0108] A sample of 10 mm.times.15 mm from the central portion of the plate width was cut out from the thickness of the copper alloy material and the surface in the rolling direction (RD direction) was polished and then micro etching was performed.
[0109] This sample was observed by SEM, and in a SEM-EPMA image of 1500 times magnification (field of view of approximately 70 .mu.m.times.70 .mu.m), a region where the Cr concentration was higher than that of the parent phase was judged to be "crystallized Cr", and ratios of crystallized Cr were determined by the following formula.
Area ratio=(area occupied by crystallized Cr)/(70 .mu.m.times.70 .mu.m)
[0110] FIGS. 4A to 4D show SEM-EPMA images of Example 1 of the present invention and Comparative Example 4.
[0111] (Tensile Strength)
[0112] JIS Z 2241 No. 2 test piece was taken with the rolling direction as the pulling direction and subjected to the test using a 100 kN tensile tester.
TABLE-US-00001 TABLE 1 Composition (mass %) Cr Zr Al Fe Co Sn Zn P Si Mg Cu Examples of 1 0.35 0.14 -- -- -- -- -- -- -- -- balance the present 2 0.35 0.14 0.02 -- -- 0.02 0.02 -- -- -- balance invention 3 0.39 0.14 0.93 -- -- -- -- -- -- -- balance 4 0.45 0.08 -- 0.01 -- -- -- 0.01 -- -- balance 5 0.45 0.07 -- -- -- -- -- -- 0.02 0.01 balance 6 0.50 0.02 1.90 -- 0.07 -- -- 0.02 -- -- balance Comparative 1 0.20 0.10 -- -- -- -- -- -- -- -- balance Examples 2 0.70 0.14 -- -- -- -- -- -- -- -- balance 3 0.70 0.02 -- -- -- -- -- -- 0.01 -- balance 4 0.90 0.08 -- -- -- -- -- -- -- 0.20 balance
TABLE-US-00002 TABLE 2 After heat treatment at Area ratio After aging treatment 1000.degree. C. for 1 hour of Cooling Average Average crystallized rate after crystal crystal Cr after solution grain Tensile grain Tensile solution treatment size Standard strength size Standard strength treatment (.degree. C./min) (mm) deviation (MPa) (mm) deviation (MPa) (%) Examples of 1 1000 0.144 0.58 433 0.243 1.21 413 0.3 the present 2 1000 0.162 0.55 430 0.451 1.25 407 0.3 invention 3 1000 0.169 0.56 423 0.331 1.34 420 0.3 4 1000 0.110 0.49 417 0.207 1.29 419 0.5 5 1000 0.161 0.52 433 0.303 1.30 401 0.4 6 27 0.142 0.55 427 0.213 1.45 389 0.5 Comparative 1 9 0.120 0.62 383 0.270 1.53 357 0.7 Examples 2 1000 0.078 0.70 432 0.292 1.56 339 1.8 3 1000 0.081 0.65 422 0.346 1.52 331 1.8 4 1000 0.096 0.68 434 0.261 1.63 353 2.0
[0113] FIGS. 2A and 3A, in Examples 1 to 6 of the present invention, local grain coarsening was suppressed even after being placed in a high-temperature environment.
[0114] On the other hand, as shown in FIGS. 2B and 3B, in Comparative Examples 1 to 4, the crystal grains coarsened locally after being placed in a high-temperature environment.
[0115] In Comparative Example 1 in which the Cr content was less than the range of the scope of the present invention and the standard deviation of the crystal grain size was more than the range of the scope of the present invention, the tensile strength after the aging heat treatment and after the heat treatment at 1000.degree. C. for 1 hour was insufficient.
[0116] In Comparative Example 2-4 in which the content of Cr was more than the range of the scope of the present invention and the average crystal grain size was less than the scope of the present invention and the standard deviation of crystal grain size was more than the range of the scope of the present invention, the tensile strength greatly decreased after the heat treatment at 1000.degree. C. for 1 hour.
[0117] In contrast, in Examples 1-6 of the present invention, the tensile strength after aging heat treatment was high and the tensile strength did not decrease greatly after the heat treatment at 1000.degree. C. for 1 hour.
[0118] From the above-described results, according to the example of the present invention, it is confirmed that a copper alloy material having stable properties and excellent service life even when it is used under a high temperature environment of 500.degree. C. or more was provided.
INDUSTRIAL APPLICABILITY
[0119] It is possible to suppress property deterioration of a part made of the Cu--Cr--Zr-based alloy in a high temperature environment and prolong the service life of the casting molding material, the welding part and the like.
* * * * *
D00001

D00002

D00003

D00004

D00005

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.