Ashless Engine Lubricants For High Temperature Applications
GAO; Zhisheng ; et al.
U.S. patent application number 16/108352 was filed with the patent office on 2019-02-28 for ashless engine lubricants for high temperature applications. The applicant listed for this patent is ExxonMobil Research and Engineering Company. Invention is credited to Matthew W. BOLAND, Eugine CHOI, Zhisheng GAO, Luca SALVI.
| Application Number | 20190062667 16/108352 |
| Document ID | / |
| Family ID | 63490706 |
| Filed Date | 2019-02-28 |


| United States Patent Application | 20190062667 |
| Kind Code | A1 |
| GAO; Zhisheng ; et al. | February 28, 2019 |
ASHLESS ENGINE LUBRICANTS FOR HIGH TEMPERATURE APPLICATIONS
Abstract
An ashless lubricating oil having a lubricating oil base stock as a major component, and a mixture of (i) at least one ashless antiwear additive, (ii) at least one ashless detergent, and (iii) at least one aminic antioxidant, as minor components. The lubricating oil base stock is a branched polyol ester. The lubricating oil base stock is present in an amount from about 30 to about 99.8 mass percent, based on the total mass of the ashless lubricating oil. A method for improving oxidative stability and high temperature stability of a lubricating oil in an engine or other mechanical component lubricated with the lubricating oil by using the ashless lubricating oil. The ashless lubricating oil is useful as a passenger vehicle engine oil (PVEO).
| Inventors: | GAO; Zhisheng; (Rose Valley, PA) ; SALVI; Luca; (Haddonfield, NJ) ; CHOI; Eugine; (Marlton, NJ) ; BOLAND; Matthew W.; (Philadelphia, PA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 63490706 | ||||||||||
| Appl. No.: | 16/108352 | ||||||||||
| Filed: | August 22, 2018 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 62550117 | Aug 25, 2017 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C10M 133/12 20130101; C10M 2223/065 20130101; C10M 2215/065 20130101; C10M 2207/2835 20130101; C10N 2040/255 20200501; C10M 2207/026 20130101; C10N 2030/45 20200501; C10N 2030/10 20130101; C10M 137/14 20130101; C10M 2209/104 20130101; C10M 2209/105 20130101; C10M 2215/064 20130101; C10N 2040/252 20200501; C10M 161/00 20130101; C10M 169/044 20130101; C10M 2207/2805 20130101; C10M 2290/04 20130101; C10M 2209/106 20130101; C10N 2030/04 20130101; C10M 135/06 20130101; C10M 2219/024 20130101; C10M 2223/063 20130101; C10N 2020/071 20200501; C10N 2030/08 20130101; C10M 169/04 20130101; C10M 2201/085 20130101; C10M 2223/047 20130101; C10M 105/38 20130101; C10M 145/36 20130101; C10M 2209/105 20130101; C10M 2209/108 20130101; C10M 2209/104 20130101; C10M 2209/108 20130101; C10M 2209/106 20130101; C10M 2209/108 20130101 |
| International Class: | C10M 169/04 20060101 C10M169/04; C10M 137/14 20060101 C10M137/14; C10M 135/06 20060101 C10M135/06; C10M 145/36 20060101 C10M145/36; C10M 133/12 20060101 C10M133/12; C10M 161/00 20060101 C10M161/00 |
Claims
1. A method for improving oxidative stability of a lubricating oil in an engine or other mechanical component lubricated with the lubricating oil by using as the lubricating oil an ashless formulated oil, said ashless formulated oil having a composition comprising a lubricating oil base stock as a major component; and a mixture of (i) at least one ashless antiwear additive, (ii) at least one ashless detergent, and (iii) at least one aminic antioxidant, as minor components; wherein the lubricating oil base stock comprises at least one branched polyol ester, which is obtained by reacting one or more polyhydric alcohols with one or more branched mono-carboxylic acids containing at least 4 carbon atoms, and wherein the lubricating oil base stock is present in an amount from 30 to 99.8 mass percent, based on the total mass of the lubricating oil.
2. The method of claim 1 wherein oxidative stability is improved, as compared to oxidative stability achieved using a lubricating oil other than the ashless formulated oil.
3. The method of claim 1 wherein the one or more polyhydric alcohols are selected from the group consisting of trimethylol propane, pentaerythritol, neopentyl glycol, trimethylol ethane, 2-methyl-2-propyl-1,3-propanediol, and dipentaerythritol.
4. The method of claim 1 wherein the one or more branched mono-carboxylic acids containing at least 4 carbon atoms are selected from the group consisting of 3,5,5-trimethyl hexanoic acid (TMH), 2,2-dimethyl propionic acid (neopentanoic acid), neoheptanoic acid, neooctanoic acid, neononanoic acid, iso-hexanoic acid, neodecanoic acid, 2-ethyl hexanoic acid (2EH), isoheptanoic acid, isooctanoic acid, isononanoic acid, and isodecanoic acid.
5. The method of claim 1 wherein the at least one branched polyol ester is selected from the group consisting of trimethylol propane ester of 3,5,5-trimethyl hexanoic acid (TMH), trimethylol propane ester of 2,2-dimethyl propionic acid (neopentanoic acid), trimethylol propane ester of neoheptanoic acid, trimethylol propane ester of neooctanoic acid, trimethylol propane ester of neononanoic acid, trimethylol propane ester of iso-hexanoic acid, trimethylol propane ester of neodecanoic acid, trimethylol propane ester of 2-ethyl hexanoic acid (2EH), trimethylol propane ester of isoheptanoic acid, trimethylol propane ester of isooctanoic acid, trimethylol propane ester of isononanoic acid, and trimethylol propane ester of isodecanoic acid.
6. The method of claim 1 wherein the at least one branched polyol ester is selected from the group consisting of pentaerythritol ester of 3,5,5-trimethyl hexanoic acid (TMH), pentaerythritol ester of 2,2-dimethyl propionic acid (neopentanoic acid), pentaerythritol ester of neoheptanoic acid, pentaerythritol ester of neooctanoic acid, pentaerythritol ester of neononanoic acid, pentaerythritol ester of iso-hexanoic acid, pentaerythritol ester of neodecanoic acid, pentaerythritol ester of 2-ethyl hexanoic acid (2EH), pentaerythritol ester of isoheptanoic acid, pentaerythritol ester of isooctanoic acid, pentaerythritol ester of isononanoic acid, and pentaerythritol ester of isodecanoic acid.
7. The method of claim 1 wherein the at least one ashless antiwear additive is selected from the group consisting of an amine phosphate, a thiophosphate, a dithiophosphate, an amine salt of sulfurized phosphate, an alkylated triphenyl phosphorothionate, a sulfurized triglyceride, and mixtures thereof.
8. The method of claim 1 wherein the at least one ashless detergent is selected from the group consisting of a polyoxyethylene alkyl ether, a polyoxypropylene alkyl ether, and a polyoxybutylene alkyl ether.
9. The method of claim 1 wherein the at least one aminic antioxidant is selected from the group consisting of p,p'-dioctyldiphenylamine, octylated phenyl-alpha-naphthylamine, octylated/butylated diphenylamine, and a polymeric aminic antioxidant.
10. The method of claim 9 wherein the polymeric aminic antioxidant is the polymerization reaction product of one or more substituted or hydrocarbyl-substituted diphenyl amines, one or more unsubstituted or hydrocarbyl-substituted phenyl naphthyl amines, or a combination thereof.
11. The method of claim 1 wherein the at least one ashless antiwear additive is present in an amount from 0.01 to 1.2 mass percent, based on the total mass of the lubricating oil.
12. The method of claim 1 wherein the at least one ashless detergent is present in an amount from 0.01 to 6 mass percent, based on the total mass of the lubricating oil.
13. The method of claim 1 wherein the at least one aminic antioxidant is present in an amount from 0.01 to 15 mass percent, based on the total mass of the lubricating oil.
14. The method of claim 1 wherein the ashless formulated oil further comprises one or more of a viscosity modifier, dispersant, pour point depressant, corrosion inhibitor, metal deactivator, seal compatibility additive, anti-foam agent, inhibitor, and anti-rust additive.
15. The method of claim 1 wherein the engine or other mechanical component comprises a high energy density gasoline engine equipped with turbo chargers, or gasoline particular filters (GPFs).
16. The method of claim 1 wherein the lubricating oil is a passenger vehicle engine oil (PVEO).
17. An ashless lubricating oil having a composition comprising a lubricating oil base stock as a major component; and a mixture of (i) at least one ashless antiwear additive, (ii) at least one ashless detergent, and (iii) at least one aminic antioxidant, as minor components; wherein the lubricating oil base stock comprises at least one branched polyol ester, which is obtained by reacting one or more polyhydric alcohols with one or more branched mono-carboxylic acids containing at least 4 carbon atoms; and wherein the lubricating oil base stock is present in an amount from 30 to 99.8 mass percent, based on the total mass of the ashless lubricating oil.
18. The ashless lubricating oil of claim 17 wherein, in an engine or other mechanical component lubricated with the ashless lubricating oil, oxidative stability is improved achieved using a lubricating oil other than the ashless lubricating oil.
19. The ashless lubricating oil of claim 17 wherein the one or more polyhydric alcohols are selected from the group consisting of trimethylol propane, pentaerythritol, neopentyl glycol, trimethylol ethane, 2-methyl-2-propyl-1,3-propanediol, and dipentaerythritol.
20. The ashless lubricating oil of claim 17 wherein the one or more branched mono-carboxylic acids containing at least 4 carbon atoms are selected from the group consisting of 3,5,5-trimethyl hexanoic acid (TMH), 2,2-dimethyl propionic acid (neopentanoic acid), neoheptanoic acid, neooctanoic acid, neononanoic acid, iso-hexanoic acid, neodecanoic acid, 2-ethyl hexanoic acid (2EH), isoheptanoic acid, isooctanoic acid, isononanoic acid, and isodecanoic acid.
21. The ashless lubricating oil of claim 17 wherein the at least one branched polyol ester is selected from the group consisting of trimethylol propane ester of 3,5,5-trimethyl hexanoic acid (TMH), trimethylol propane ester of 2,2-dimethyl propionic acid (neopentanoic acid), trimethylol propane ester of neoheptanoic acid, trimethylol propane ester of neooctanoic acid, trimethylol propane ester of neononanoic acid, trimethylol propane ester of iso-hexanoic acid, trimethylol propane ester of neodecanoic acid, trimethylol propane ester of 2-ethyl hexanoic acid (2EH), trimethylol propane ester of isoheptanoic acid, trimethylol propane ester of isooctanoic acid, trimethylol propane ester of isononanoic acid, and trimethylol propane ester of isodecanoic acid.
22. The ashless lubricating oil of claim 17 wherein the at least one branched polyol ester is selected from the group consisting of pentaerythritol ester of 3,5,5-trimethyl hexanoic acid (TMH), pentaerythritol ester of 2,2-dimethyl propionic acid (neopentanoic acid), pentaerythritol ester of neoheptanoic acid, pentaerythritol ester of neooctanoic acid, pentaerythritol ester of neononanoic acid, pentaerythritol ester of iso-hexanoic acid, pentaerythritol ester of neodecanoic acid, pentaerythritol ester of 2-ethyl hexanoic acid (2EH), pentaerythritol ester of isoheptanoic acid, pentaerythritol ester of isooctanoic acid, pentaerythritol ester of isononanoic acid, and pentaerythritol ester of isodecanoic acid.
23. The ashless lubricating oil of claim 17 wherein the at least one ashless antiwear additive is selected from the group consisting of an amine phosphate, a thiophosphate, a dithiophosphate, an amine salt of sulfurized phosphate, an alkylated triphenyl phosphorothionate, a sulfurized triglyceride, and mixtures thereof.
24. The ashless lubricating oil of claim 17 wherein the at least one ashless detergent is selected from the group consisting of a polyoxyethylene alkyl ether, a polyoxypropylene alkyl ether, and a polyoxybutylene alkyl ether.
25. The ashless lubricating oil of claim 17 wherein the at least one aminic antioxidant is selected from the group consisting of p,p'-dioctyldiphenylamine, octylated phenyl-alpha-naphthylamine, octylated/butylated diphenylamine, and a polymeric aminic antioxidant.
26. The ashless lubricating oil of claim 25 wherein the polymeric aminic antioxidant is the polymerization reaction product of one or more substituted or hydrocarbyl-substituted diphenyl amines, one or more unsubstituted or hydrocarbyl-substituted phenyl naphthyl amines, or a combination thereof.
27. The ashless lubricating oil of claim 17 wherein the at least one ashless antiwear additive is present in an amount from 0.01 to 1.2 mass percent, based on the total mass of the lubricating oil.
28. The ashless lubricating oil of claim 17 wherein the at least one ashless detergent is present in an amount from 0.01 to 6 mass percent, based on the total mass of the lubricating oil.
29. The ashless lubricating oil of claim 17 wherein the at least one aminic antioxidant is present in an amount from 0.01 to 15 mass percent, based on the total mass of the lubricating oil.
30. The ashless lubricating oil of claim 17 further comprising one or more of a viscosity modifier, dispersant, pour point depressant, corrosion inhibitor, metal deactivator, seal compatibility additive, anti-foam agent, inhibitor, and anti-rust additive.
31. The ashless lubricating oil of claim 17 wherein the engine or other mechanical component comprises a high energy density gasoline engine equipped with turbo chargers, or gasoline particular filters (GPFs).
32. The ashless lubricating oil of claim 17 which is a passenger vehicle engine oil (PVEO).
33. A method for improving high temperature stability of a lubricating oil in an engine or other mechanical component lubricated with the lubricating oil by using as the lubricating oil an ashless formulated oil, said ashless formulated oil having a composition comprising a lubricating oil base stock as a major component; and a mixture of (i) at least one ashless antiwear additive, (ii) at least one ashless detergent, and (iii) at least one aminic antioxidant, as minor components; wherein the lubricating oil base stock comprises at least one branched polyol ester, which is obtained by reacting one or more polyhydric alcohols with one or more branched mono-carboxylic acids containing at least 4 carbon atoms, and at least one alkylated naphthalene; and wherein the lubricating oil base stock is present in an amount from 30 to 99.8 mass percent, based on the total mass of the lubricating oil.
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application claims the benefit of U.S. Provisional Application No. 62/550,117, filed on Aug. 25, 2017, the entire contents of which are incorporated herein by reference.
FIELD
[0002] This disclosure relates to ashless engine lubricating oils for high temperature applications. In particular, this disclosure relates to ashless lubricating oils, methods for improving oxidative stability in an engine or other mechanical component lubricated with an ashless lubricating oil, and methods for improving high temperature stability of a lubricating oil in an engine or other mechanical component lubricated with an ashless lubricating oil. The ashless lubricating oils of this disclosure are useful as passenger vehicle engine oil (PVEO) products, especially for engines equipped with turbo chargers, or diesel or gasoline particular filters (DPFs or GPFs).
BACKGROUND
[0003] In today's high energy density engines, lubricants experience much higher temperatures than in the past and in some parts of the engines (e.g., the turbo chargers), the temperature can reach 200.degree. C. or higher. In addition, there are advantages in running an engine hotter while maintaining a low air intake temperature. One such advantage is to reduce the need for cooling.
[0004] General Motors has disclosed the use of an automated air shutter system that reduces the wind drag at high vehicle speed when the temperature is not too high, resulting in a fuel efficiency of nearly half a mile per gallon in combined city and highway driving for the 2011 Chevrolet Cruze Eco (see GM Corporate Newsroom, Unique Air Shutter Helps Morph Chevrolet Cruze into 40 MPG Leader, 2010-08-10).
[0005] Further, GPFs are being used in direct injection gasoline engines to meet the particular emission requirements. Studies have shown that engine oil ash level could have impacts on GPF performance and service life (N. C. Custer "Ash Impacts on Gasoline Particular Filter Performance and Service Life", Master Thesis, MIT, June 2015). What is needed is an ashless engine oil that is beneficial to GPFs.
SUMMARY
[0006] This disclosure relates in part to new ashless lubricating oil formulations which are useful in high temperature applications such as high energy density engines equipped with turbo chargers. This disclosure also relates in part to new ashless lubricating oil formulations having high temperature stability sufficient to reduce engine cooling needs (smaller radiator or less wind drag), and sufficient to reduce water contamination (for engines equipped with water injection technology).
[0007] This disclosure also relates in part to a method for improving oxidative stability of a lubricating oil, in an engine or other mechanical component lubricated with the lubricating oil by using as the lubricating oil an ashless formulated oil. The ashless formulated oil has a composition comprising a lubricating oil base stock as a major component, and a mixture of (i) at least one ashless antiwear additive, (ii) at least one ashless detergent, and (iii) at least one aminic antioxidant, as minor components. The lubricating oil base stock is at least one branched polyol ester, which is obtained by reacting one or more polyhydric alcohols with one or more branched mono-carboxylic acids containing at least about 4 carbon atoms, or at least one alkylated naphthalene. The lubricating oil base stock is present in an amount from about 30 to about 99.8 mass percent, based on the total mass of the lubricating oil.
[0008] This disclosure further relates in part to a method for improving oxidative stability of a lubricating oil in an engine or other mechanical component lubricated with the lubricating oil by using as the lubricating oil an ashless formulated oil. The ashless formulated oil has a composition comprising a lubricating oil base stock as a major component, and at least one ashless antiwear additive, as a minor component. The lubricating oil base stock is at least one branched polyol ester, which is obtained by reacting one or more polyhydric alcohols with one or more branched mono-carboxylic acids containing at least about 4 carbon atoms. The lubricating oil base stock is present in an amount from about 30 to about 99.8 mass percent, based on the total mass of the lubricating oil.
[0009] This disclosure yet further relates in part to an ashless lubricating oil having a composition comprising a lubricating oil base stock as a major component, and a mixture of (i) at least one ashless antiwear additive, (ii) at least one ashless detergent, and (iii) at least one aminic antioxidant, as minor components. The lubricating oil base stock is at least one branched polyol ester, which is obtained by reacting one or more polyhydric alcohols with one or more branched mono-carboxylic acids containing at least about 4 carbon atoms. The lubricating oil base stock is present in an amount from about 30 to about 99.8 mass percent, based on the total mass of the ashless lubricating oil.
[0010] This disclosure also relates in part to an ashless lubricating oil having a composition comprising a lubricating oil base stock. The lubricating oil base stock is at least one branched polyol ester, which is obtained by reacting one or more polyhydric alcohols with one or more branched mono-carboxylic acids containing at least about 4 carbon atoms. The lubricating oil base stock is present in an amount from about 30 to about 99.8 mass percent, based on the total mass of the ashless lubricating oil.
[0011] This disclosure further relates in part to an ashless lubricating oil having a composition comprising a lubricating oil base stock as a major component; and at least one ashless antiwear additive, as a minor component. The lubricating oil base stock is at least one branched polyol ester, which is obtained by reacting one or more polyhydric alcohols with one or more branched mono-carboxylic acids containing at least about 4 carbon atoms. The lubricating oil base stock is present in an amount from about 30 to about 99.8 mass percent, based on the total mass of the ashless lubricating oil.
[0012] This disclosure yet further relates in part to a method for improving high temperature stability of a lubricating oil in an engine or other mechanical component lubricated with the lubricating oil by using as the lubricating oil an ashless formulated oil. The ashless formulated oil has a composition comprising a lubricating oil base stock as a major component, and a mixture of (i) at least one ashless antiwear additive, (ii) at least one ashless detergent, and (iii) at least one aminic antioxidant, as minor components. The lubricating oil base stock is at least one branched polyol ester, which is obtained by reacting one or more polyhydric alcohols with one or more branched mono-carboxylic acids containing at least about 4 carbon atoms. The lubricating oil base stock is present in an amount from about 30 to about 99.8 mass percent, based on the total mass of the lubricating oil.
[0013] This disclosure also relates in part to a method for improving high temperature stability of a lubricating oil in an engine or other mechanical component lubricated with the lubricating oil by using as the lubricating oil an ashless formulated oil. The ashless formulated oil has a composition comprising a lubricating oil base stock as a major component, and at least one ashless antiwear additive, as a minor component. The lubricating oil base stock is at least one branched polyol ester, which is obtained by reacting one or more polyhydric alcohols with one or more branched mono-carboxylic acids containing at least about 4 carbon atoms. The lubricating oil base stock is present in an amount from about 30 to about 99.8 mass percent, based on the total mass of the lubricating oil.
[0014] It has been surprisingly found that, in accordance with this disclosure, high temperature stability of a lubricating oil can be attained in an engine or other mechanical component lubricated with a lubricating oil by using as the lubricating oil an ashless formulated oil having at least one branched polyol ester base stock, in which the base stock is present in an amount from about 30 to about 99.8 mass percent, based on the total mass of the lubricating oil. Such high temperature stability affords a number of advantages including, for example, more compatibility with high energy density engines equipped with turbo chargers, or DPFs or GPFs, reducing engine cooling needs (smaller radiator or less wind drag), and reducing water contamination (for engines equipped with water injection technology).
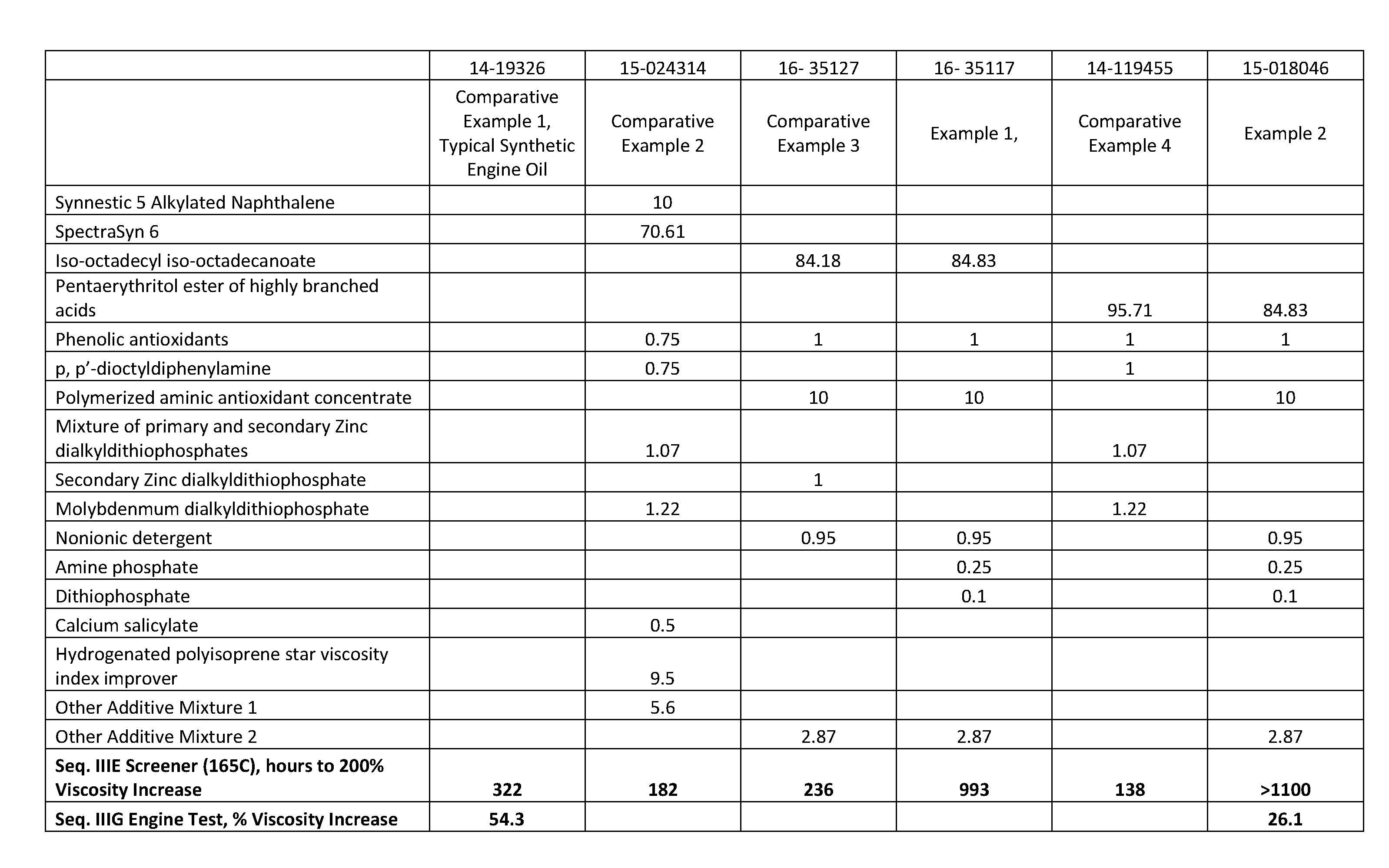
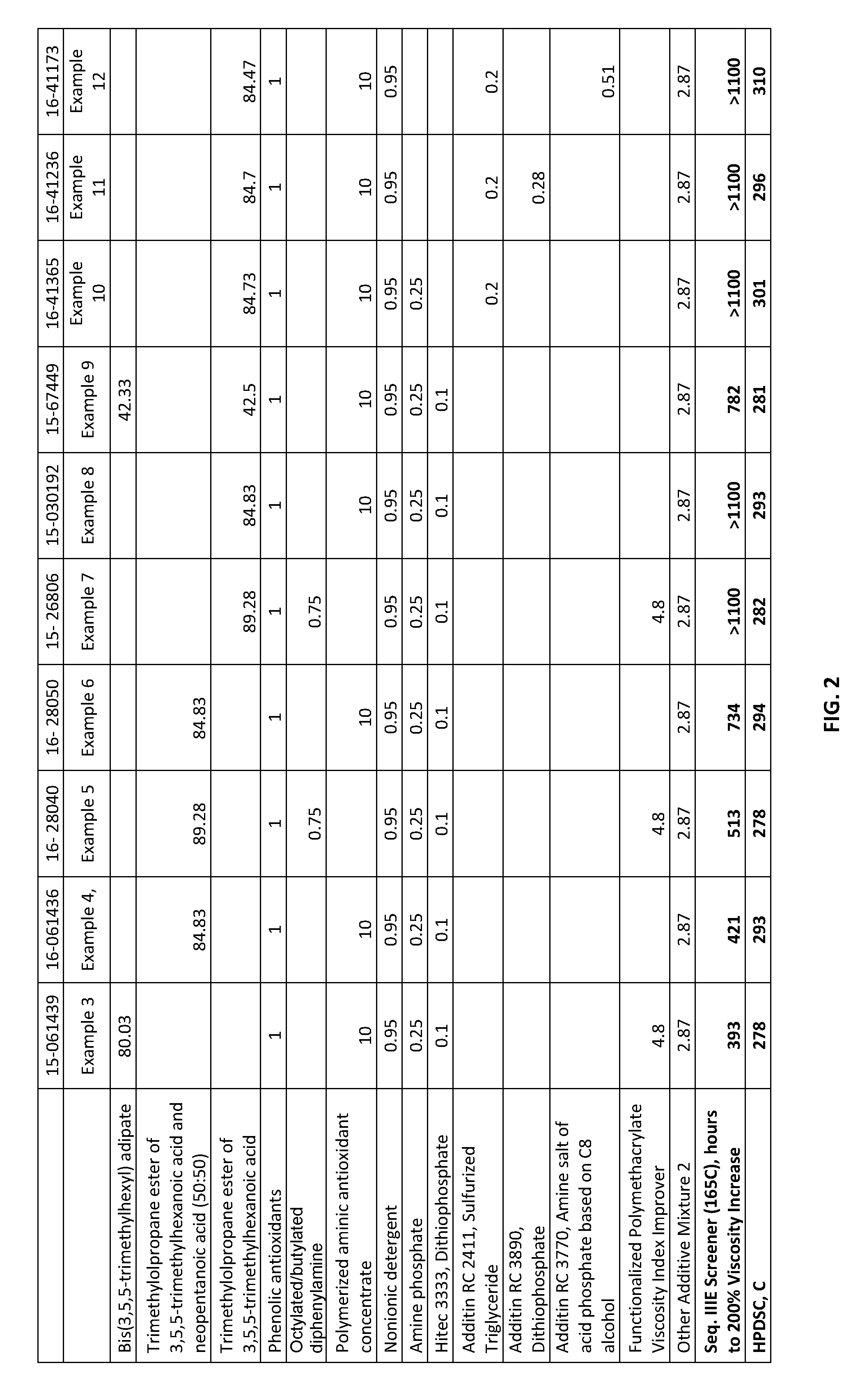
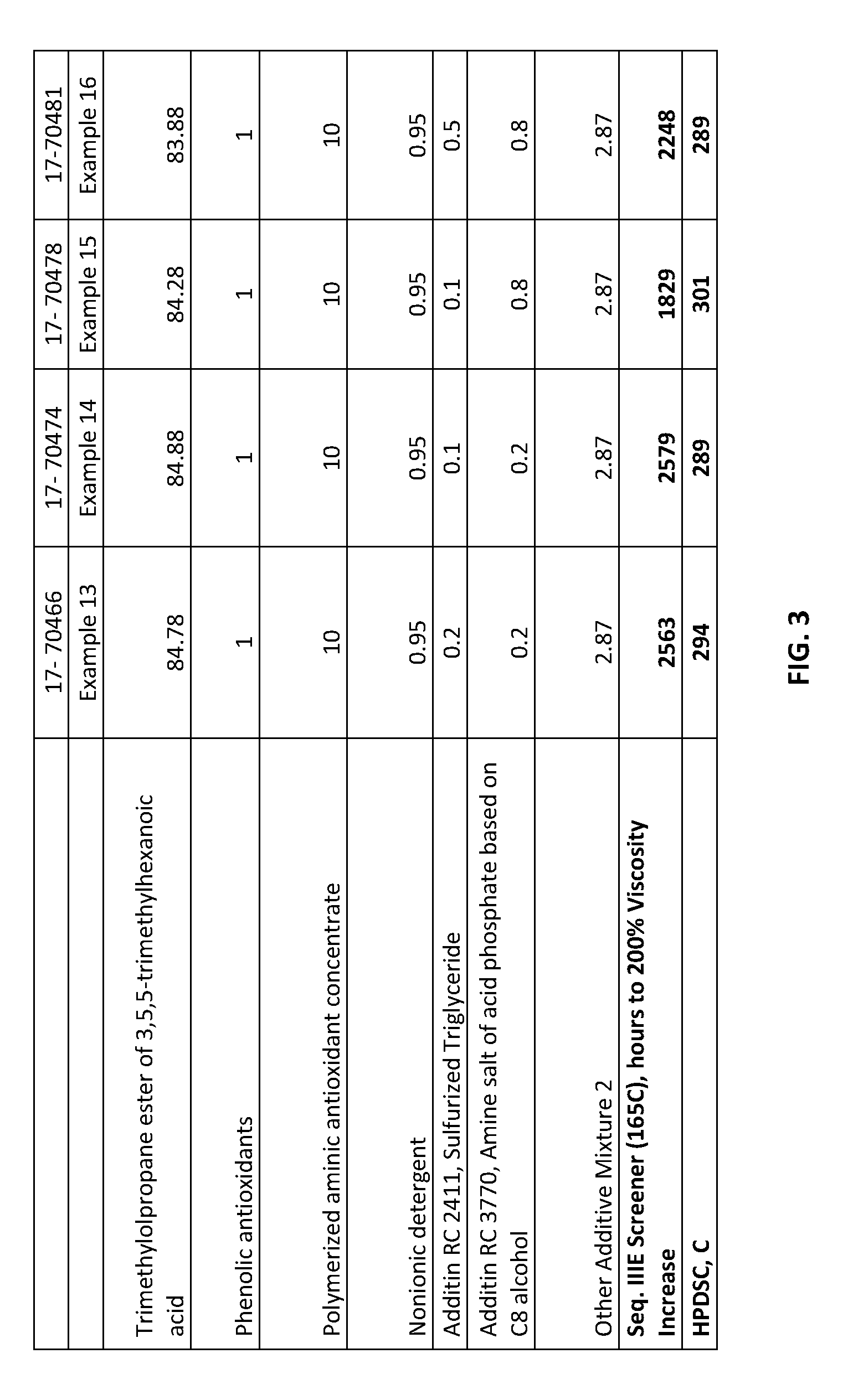
[0015] In particular, it has been surprisingly found that the ashless lubricating oils of this disclosure, in a bench Sequence IIIE Screener test, reach 200% viscosity increase after 600 hours, or after 800 hours, or after 1000 hours, or after 1100 hours (versus about 300 hours for a typical synthetic engine oil as shown in the Examples hereinbelow).
[0016] Other objects and advantages of the present disclosure will become apparent from the detailed description that follows.
BRIEF DESCRIPTION OF THE DRAWINGS
[0017] FIG. 1 shows lubricating oil formulations prepared in accordance with the Examples, and also testing results from the formulations.
[0018] FIG. 2 shows lubricating oil formulations prepared in accordance with the Examples, and also testing results from the formulations.
[0019] FIG. 3 shows lubricating oil formulations prepared in accordance with Examples 13-16 and also testing results from the formulations.
DETAILED DESCRIPTION
[0020] All numerical values within the detailed description and the claims herein are modified by "about" or "approximately" the indicated value, and take into account experimental error and variations that would be expected by a person having ordinary skill in the art.
[0021] In accordance with this disclosure, it has been found that engine lubricants with step-out oxidation stability can be formulated. By improving the oxidation stability of the lubricants, the deposit formation tendency on a metal surface is also reduced significantly, enabling the use of ashless detergent additives. Ashless antiwear additives, such as amine phosphates, have been widely used in many industrial lubricants.
[0022] It has now been found that high temperature stability of a lubricating oil can be attained in an engine or other mechanical component lubricated with a lubricating oil by using as the lubricating oil an ashless formulated oil having at least one branched polyol ester base stock, in which the base stock is present in an amount from about 30 to about 99.8 mass percent, based on the total mass of the lubricating oil. The lubricating oil preferably has a mixture of at least one ashless antiwear additive, at least one ashless detergent, and at least one aminic antioxidant. Such high temperature stability affords a number of advantages including, for example, more compatibility with high energy density engines equipped with turbo chargers, reducing engine cooling needs (smaller radiator or less wind drag), and reducing water contamination (for engines equipped with water injection technology).
[0023] In addition, it has been found that improved oxidative stability and deposit control can be attained in an engine or other mechanical component lubricated with a lubricating oil by using as the lubricating oil an ashless formulated oil having at least one branched polyol ester base stock, in which the base stock is present in an amount from about 30 to about 99.8 mass percent, based on the total mass of the lubricating oil. The lubricating oil preferably has a mixture of at least one ashless antiwear additive, at least one ashless detergent, and at least one aminic antioxidant.
Branched Polyol Ester Lubricating Oil Base Stocks
[0024] Branched polyol esters comprise a useful base stock of this disclosure. The branched polyol esters are obtained by reacting one or more polyhydric alcohols, preferably the hindered polyols (such as the neopentyl polyols, e.g., neopentyl glycol, trimethylol ethane, 2-methyl-2-propyl-1,3-propanediol, trimethylol propane, pentaerythritol and dipentaerythritol) with single or mixed branched mono-carboxylic acids containing at least about 4 carbon atoms, preferably C.sub.5 to C.sub.30 branched mono-carboxylic acids including 2,2-dimethyl propionic acid (neopentanoic acid), neoheptanoic acid, neooctanoic acid, neononanoic acid, iso-hexanoic acid, neodecanoic acid, 2-ethyl hexanoic acid (2EH), 3,5,5-trimethyl hexanoic acid (TMH), isoheptanoic acid, isooctanoic acid, isononanoic acid, isodecanoic acid, or mixtures of any of these materials. These branched polyol esters include fully converted and partially converted polyol esters.
[0025] Particularly useful polyols include, for example, neopentyl glycol, 2,2-dimethylol butane, trimethylol ethane, trimethylol propane, trimethylol butane, mono-pentaerythritol, technical grade pentaerythritol, di-pentaerythritol, tri-pentaerythritol, ethylene glycol, propylene glycol and polyalkylene glycols (e.g., polyethylene glycols, polypropylene glycols, 1,4-butanediol, sorbitol and the like, 2-methylpropanediol, polybutylene glycols, etc., and blends thereof such as a polymerized mixture of ethylene glycol and propylene glycol). The most preferred alcohols are technical grade (e.g., approximately 88% mono-, 10% di- and 1-2% tri-pentaerythritol) pentaerythritol, mono-pentaerythritol, di-pentaerythritol, neopentyl glycol and trimethylol propane.
[0026] Particularly useful branched mono-carboxylic acids include, for example, 2,2-dimethyl propionic acid (neopentanoic acid), neoheptanoic acid, neooctanoic acid, neononanoic acid, iso-hexanoic acid, neodecanoic acid, 2-ethyl hexanoic acid (2EH), 3,5,5-trimethyl hexanoic acid (TMH), isoheptanoic acid, isooctanoic acid, isononanoic acid, isodecanoic acid, or mixtures of any of these materials. One especially preferred branched acid is 3,5,5-trimethyl hexanoic acid. The term "neo" as used herein refers to a trialkyl acetic acid, i.e., an acid which is triply substituted at the alpha carbon with alkyl groups.
[0027] Preferably, the branched polyol ester is derived from a polyhydric alcohol and a branched mono-carboxylic acid. In particular, the branched polyol ester is obtained by reacting one or more polyhydric alcohols with one or more branched mono-carboxylic acids containing at least about 4 carbon atoms.
[0028] Preferred branched polyol esters useful in this disclosure include, for example, mono-pentaerythritol ester of branched mono-carboxylic acids, di-pentaerythritol ester of branched mono-carboxylic acids, trimethylolpropane ester of C8-C10 acids, and the like.
[0029] Other synthetic esters that can be useful in this disclosure are those which are obtained by reacting one or more polyhydric alcohols, preferably the hindered polyols (such as the neopentyl polyols, e.g., neopentyl glycol, trimethylol ethane, 2-methyl-2-propyl-1,3-propanediol, trimethylol propane, pentaerythritol and dipentaerythritol) with mono caboxylic acids containing at least about 4 carbon atoms, preferably branched C.sub.5 to C.sub.30 acids including caprylic acid, capric acid, lauric acid, myristic acid, palmitic acid, stearic acid, arachic acid, and behenic acid, or the corresponding branched chain fatty acids or unsaturated fatty acids such as oleic acid, or mixtures of any of these materials.
[0030] Other ester base oils useful in this disclosure include adipate esters. The dialkyl adipate ester is derived from adipic acid and a branched alkyl alcohol.
[0031] Mixtures of branched polyol ester base stocks with other lubricating oil base stocks (e.g., Groups I, II, III, IV and V base stocks) may be useful in the lubricating oil formulations of this disclosure.
[0032] The branched polyol ester can be present in an amount of from about 30 to about 99.8 weight percent, or from about 35 to about 95 weight percent, or from about 40 to about 90 weight percent, or from about 45 to about 85 weight percent, or from about 50 to about 80 weight percent, or from about 55 to about 75 weight percent, or from about 60 to about 70 weight percent, based on the total weight of the formulated oil.
Ashless Antiwear Agents
[0033] In accordance with this disclosure, the lubricating engine oils have at least one ashless antiwear additive selected from a phosphorus-containing ashless antiwear additive, a sulfur-containing ashless antiwear additive, and a phosphorus/sulfur-containing ashless antiwear additive. Illustrative ashless antiwear additives useful in this disclosure include, for example, amine phosphates, thiophosphates, dithiophosphates, amine salts of sulfurized phosphates, alkylated triphenyl phosphorothionates (e.g., butylated triphenyl phosphorothionate), and mixtures thereof, and the like. These ashless antiwear additives can be obtained commercially from suppliers such as BASF under the trade name Irgalube 353, Irgalube 349, Irgalube 875, Irgalube 232, and from Vanderbilt Chemicals, LLC under the trade name Vanlube 9123, and from Dorf Ketal under the trade name PX 3844.
[0034] In particular, the ashless antiwear additive can be a phosphate ester or salt, in which the phosphate ester or salt may be a monohydrocarbyl, dihydrocarbyl or a trihydrocarbyl phosphate, wherein each hydrocarbyl group is saturated. In one embodiment, each hydrocarbyl group independently contains from about 6 to about 30, or from about 8 up to about 20, or from about 8 up to about 12 carbons atoms.
[0035] A phosphate ester or salt is a phosphorus acid ester prepared by reacting one or more phosphorus acid or anhydride with a saturated alcohol. The phosphorus acid or anhydride is generally an inorganic phosphorus reagent, such as phosphorus pentoxide, phosphorus trioxide, phosphorus tetroxide, phosphorous acid, phosphoric acid, phosphorus halide, lower phosphorus esters, or a phosphorus sulfide, including phosphorus pentasulfide, and the like. Lower phosphorus acid esters generally contain from 1 to about 7 carbon atoms in each ester group. Alcohols used to prepare the phosphorus acid esters or salts include saturated alcohols. Examples of commercially available alcohols including hexanol, heptanol, octanol, nonanol, decanol, and undecanol, and dodecanol, and alcohol mixtures include Alfol 1218 (a mixture of synthetic, primary, straight-chain alcohols containing 12 to 18 carbon atoms); Alfol 20+ alcohols (mixtures of C18-C28 primary alcohols having mostly C20 alcohols as determined by GLC (gas-liquid-chromatography)); and Alfol22+ alcohols (C18-C28 primary alcohols containing primarily C22 alcohols). Alfol alcohols are available from Continental Oil Company. Another example of a commercially available alcohol mixture is Adol 60 (about 75% by weight of a straight chain C22 primary alcohol, about 15% of a C20 primary alcohol and about 8% of C18 and C24 alcohols). The Adol alcohols are marketed by Ashland Chemical. The preferred alcohol is octanol or those with more carbon atoms.
[0036] A variety of mixtures of monohydric fatty alcohols derived from naturally occurring triglycerides and ranging in chain length from C8 to C18 are available from Procter & Gamble Company. These mixtures contain various amounts of fatty alcohols containing 12, 14, 16, or 18 carbon atoms. For example, CO-1214 is a fatty alcohol mixture containing 0.5% of C10 alcohol, 66.0% of C12 alcohol, 26.0% of C14 alcohol and 6.5% of C16 alcohol.
[0037] Another group of commercially available mixtures include the "Neodol" products available from Shell Chemical Co. For example, Neodol 23 is a mixture of C12 and C13 alcohols; Neodol 25 is a mixture of C12 to C15 alcohols; and Neodol 45 is a mixture of C14 to C15 linear alcohols. The phosphate contains from about 14 to about 18 carbon atoms in each hydrocarbyl group. The hydrocarbyl groups of the phosphate are generally derived from a mixture of fatty alcohols having from about 14 up to about 18 carbon atoms. The hydrocarbyl phosphate may also be derived from a fatty vicinal diol. Fatty vicinal diols include those available from Ashland Oil under the general trade designation Adol 114 and Adol 158. The former is derived from a straight chain alpha olefin fraction of C11-C14, and the latter is derived from a C15-C18 fraction.
[0038] The phosphate salts may be prepared by reacting an acidic phosphate ester with an amine compound or a metallic base to form an amine or a metal salt. The amines may be monoamines or polyamines. Useful amines include those amines disclosed in U.S. Pat. No. 4,234,435.
[0039] The monoamines generally contain a hydrocarbyl group which contains from 1 to about 30 carbon atoms, or from 1 to about 12, or from 1 to about 6. Examples of primary monoamines useful in the present disclosure include methylamine, ethylamine, propylamine, butylamine, cyclopentylamine, cyclohexylamine, octylamine, dodecylamine, allylamine, cocoamine, stearylamine, and laurylamine. Examples of secondary monoamines include dimethylamine, diethylamine, dipropylamine, dibutylamine, dicyclopentylamine, dicyclohexylamine, methylbutylamine, ethylhexylamine, and the like.
[0040] A useful amine is a fatty (C8-30) amine which includes n-octylamine, n-decylamine, n-dodecylamine, n-tetradecylamine, n-hexadecylamine, n-octadecylamine, oleyamine, etc. Also useful fatty amines include commercially available fatty amines such as "Armeen" amines (products available from Akzo Chemicals, Chicago, Ill.), such Armeen C, Armeen O, Armeen OL, Armeen T, Armeen HT, Armeen S and Armeen S D, wherein the letter designation relates to the fatty group, such as coco, oleyl, tallow, or stearyl groups.
[0041] Other useful amines include primary ether amines, such as those represented by the formula, R''(OR') x NH2, wherein R' is a divalent alkylene group having about 2 to about 6 carbon atoms; x is a number from one to about 150, or from about one to about five, or one; and R'' is a hydrocarbyl group of about 5 to about 150 carbon atoms. An example of an ether amine is available under the name SURFAM.RTM. amines produced and marketed by Mars Chemical Company, Atlanta, Ga. Preferred ether amines are exemplified by those identified as SURFAM P14B (decyloxypropylamine), SURFAM P16A (linear C16), SURFAM P17B (tridecyloxypropylamine). The carbon chain lengths (i.e., C14, etc.) of the SURFAMS described above and used hereinafter are approximate and include the oxygen ether linkage.
[0042] An illustrative amine is a tertiary-aliphatic primary amine. Generally, the aliphatic group, preferably an alkyl group, contains from about 4 to about 30, or from about 6 to about 24, or from about 8 to about 22 carbon atoms. Usually the tertiary alkyl primary amines are monoamines the alkyl group is a hydrocarbyl group containing from one to about 27 carbon atoms and R6 is a hydrocarbyl group containing from 1 to about 12 carbon atoms. Such amines are illustrated by tert-butylamine, tert-hexylamine, 1-methyl-1-amino-cyclohexane, tert-octylamine, tert-decylamine, tert-dodecylamine, tert-tetradecylamine, tert-hexadecylamine, tert-octadecylamine, tert-tetracosanylamine, and tert-octacosanylamine. Mixtures of tertiary aliphatic amines may also be used in preparing the phosphate salt. Illustrative of amine mixtures of this type are "Primene 81R" which is a mixture of C11-C14 tertiary alkyl primary amines and "Primene JMT" which is a similar mixture of C18-C22 tertiary alkyl primary amines (both are available from Rohm and Haas Company). The tertiary aliphatic primary amines and methods for their preparation are known to those of ordinary skill in the art.
[0043] Another illustrative amine is a heterocyclic polyamine. The heterocyclic polyamines include aziridines, azetidines, azolidines, tetra- and dihydropyridines, pyrroles, indoles, piperidines, imidazoles, di- and tetra-hydroimidazoles, piperazines, isoindoles, purines, morpholines, thiomorpholines, N-aminoalkylmorpholines, N-aminoalkylthiomorpholines, N-aminoalkyl-piperazines, N,N'-diaminoalkylpiperazines, azepines, azocines, azonines, azecines and tetra-, di- and perhydro derivatives of each of the above and mixtures of two or more of these heterocyclic amines. Preferred heterocyclic amines are the saturated 5- and 6-membered heterocyclic amines containing only nitrogen, oxygen and/or sulfur in the hetero ring, especially the piperidines, piperazines, thiomorpholines, morpholines, pyrrolidines, and the like. Piperidine, aminoalkyl substituted piperidines, piperazine, aminoalkyl substituted piperazines, morpholine, aminoalkyl substituted morpholines, pyrrolidine, and aminoalkyl-substituted pyrrolidines, are especially preferred. Usually the aminoalkyl substituents are substituted on a nitrogen atom forming part of the hetero ring. Specific examples of such heterocyclic amines include N-aminopropylmorpholine, N-aminoethylpiperazine, and N,N'-diaminoethylpiperazine. Hydroxy heterocyclic polyamines are also useful. Examples include N-(2-hydroxyethyl)cyclohexylamine, 3-hydroxycyclopentylamine, parahydroxyaniline, N-hydroxyethylpiperazine, and the like.
[0044] Ashless antiwear additives also may include a fatty imidazoline or a reaction product of a fatty carboxylic acid and at least one polyamine. The fatty imidazoline has fatty substituents containing from 8 to about 30, or from about 12 to about 24 carbon atoms. The substituent may be saturated or unsaturated, heptadeceneyl derived oleyl groups, preferably saturated. In one aspect, the fatty imidazoline may be prepared by reacting a fatty carboxylic acid with a polyalkylenepolyamine, such as those discussed above. The fatty carboxylic acids are generally mixtures of straight and branched chain fatty carboxylic acids containing about 8 to about 30 carbon atoms, or from about 12 to about 24, or from about 16 to about 18. Carboxylic acids include the polycarboxylic acids or carboxylic acids or anhydrides having from 2 to about 4 carbonyl groups, preferably 2. The polycarboxylic acids include succinic acids and anhydrides and Diels-Alder reaction products of unsaturated monocarboxylic acids with unsaturated carboxylic acids (such as acrylic, methacrylic, maleic, fumaric, crotonic and itaconic acids). Preferably, the fatty carboxylic acids are fatty monocarboxylic acids, having from about 8 to about 30, preferably about 12 to about 24 carbon atoms, such as octanoic, oleic, stearic, linoleic, dodecanoic, and tall oil acids, preferably stearic acid. The fatty carboxylic acid is reacted with at least one polyamine. The polyamines may be aliphatic, cycloaliphatic, heterocyclic or aromatic. Examples of the polyamines include alkylene polyamines and heterocyclic polyamines.
[0045] Hydroxyalkyl groups are to be understood as meaning, for example, monoethanolamine, diethanolamine or triethanolamine, and the term amine also includes diamine. The amine used for the neutralization depends on the phosphoric esters used. The ashless antiwear additive according to the disclosure has the following advantages. It very high effectiveness when used in low concentrations and it is free of chlorine. For the neutralization of the phosphoric esters, the latter are taken and the corresponding amine slowly added with stirring. The resulting heat of neutralization is removed by cooling. The ashless antiwear additive according to the disclosure can be incorporated into the respective base liquid with the aid of fatty substances (e.g. tall oil fatty acid, oleic acid, etc.) as solubilizers. The base liquids used are napthenic or paraffinic base oils, synthetic oils (e.g. polyglycols, mixed polyglycols), polyolefins, carboxylic esters, and the like.
[0046] The ashless antiwear additive can include at least one straight and/or branched chain saturated or unsaturated monocarboxylic acid which is optionally sulphurized in an amount which may be up to 35% by weight; and/or an ester of such an acid. At least one triazole or alkyl derivatives thereof, or short chain alkyl of up to 5 carbon atoms and is hydrogen, morphilino, alkyl, amido, amino, hydroxy or alkyl or aryl substituted derivatives thereof; or a triazole selected from 1,2,4 triazole, 1,2,3 triazole, 3-amino-1,2,4 triazole, 1-H-benzotriazole-1-yl-methylisocyanide, methylene-bis-benzotriazole and naphthotriazole. The neutral organic phosphate which forms a component of the formulation may be present in an amount of 0.01 to 4%, preferably 1.5 to 2.5% by weight of the composition. The above amine phosphates and any of the aforementioned benzo- or tolyltriazoles can be mixed together to form a single component capable of delivering antiwear performance. The neutral organic phosphate is also a conventional ingredient of lubricating compositions and any such neutral organic phosphate falling within the formula as previously defined may be employed.
[0047] In an embodiment, phosphates for use in the present disclosure include phosphates, acid phosphates, phosphites and acid phosphites. The phosphates include triaryl phosphates, trialkyl phosphates, trialkylaryl phosphates, triarylalkyl phosphates and trialkenyl phosphates. As specific examples of these, referred to are triphenyl phosphate, tricresyl phosphate, benzyldiphenyl phosphate, ethyldiphenyl phosphate, tributyl phosphate, ethyldibutyl phosphate, cresyldiphenyl phosphate, dicresylphenyl phosphate, ethylphenyldiphenyl phosphate, diethylphenylphenyl phosphate, propylphenyldiphenyl phosphate, dipropylphenylphenyl phosphate, triethylphenyl phosphate, tripropylphenyl phosphate, butylphenyldiphenyl phosphate, dibutylphenylphenyl phosphate, tributylphenyl phosphate, trihexyl phosphate, tri(2-ethylhexyl) phosphate, tridecyl phosphate, trilauryl phosphate, trimyristyl phosphate, tripalmityl phosphate, tristearyl phosphate, and trioleyl phosphate. The acid phosphates include, for example, 2-ethylhexyl acid phosphate, ethyl acid phosphate, butyl acid phosphate, oleyl acid phosphate, tetracosyl acid phosphate, isodecyl acid phosphate, lauryl acid phosphate, tridecyl acid phosphate, stearyl acid phosphate, and isostearyl acid phosphate.
[0048] The phosphites include, for example, triethyl phosphite, tributyl phosphite, triphenyl phosphite, tricresyl phosphite, tri(nonylphenyl) phosphite, tri(2-ethylhexyl) phosphite, tridecyl phosphite, trilauryl phosphite, triisooctyl phosphite, diphenylisodecyl phosphite, tristearyl phosphite, and trioleyl phosphite.
[0049] The acid phosphites include, for example, dibutyl hydrogenphosphite, dilauryl hydrogenphosphite, dioleyl hydrogenphosphite, distearyl hydrogenphosphite, and diphenyl hydrogenphosphite.
[0050] Amines that form amine salts with such phosphates include, for example, mono-substituted amines, di-substituted amines and tri-substituted amines.
[0051] Examples of the mono-substituted amines include butylamine, pentylamine, hexylamine, cyclohexylamine, octylamine, laurylamine, stearylamine, oleylamine and benzylamine; and those of the di-substituted amines include dibutylamine, dipentylamine, dihexylamine, dicyclohexylamine, dioctylamine, dilaurylamine, distearylamine, dioleylamine, dibenzylamine, stearyl monoethanolamine, decyl monoethanolamine, hexyl monopropanolamine, benzyl monoethanolamine, phenyl monoethanolamine, and tolyl monopropanolamine. Examples of tri-substituted amines include tributylamine, tripentylamine, trihexylamine, tricyclohexylamine, trioctylamine, trilaurylamine, tristearylamine, trioleylamine, tribenzylamine, dioleyl monoethanolamine, dilauryl monopropanolamine, dioctyl monoethanolamine, dihexyl monopropanolamine, dibutyl monopropanolamine, oleyl diethanolamine, stearyl dipropanolamine, lauryl diethanolamine, octyl dipropanolamine, butyl diethanolamine, benzyl diethanolamine, phenyl diethanolamine, tolyl dipropanolamine, xylyl diethanolamine, triethanolamine, and tripropanolamine.
[0052] As used herein, the term "hydrocarbyl substituent" or "hydrocarbyl group" is used in its ordinary sense, which is well-known to those skilled in the art. Specifically, it refers to a group having a carbon atom directly attached to the remainder of the molecule and having predominantly hydrocarbon character. Examples of hydrocarbyl groups include: hydrocarbon substituents, that is, aliphatic (e.g., alkyl or alkenyl), alicyclic (e.g., cycloalkyl, cycloalkenyl) substituents, and aromatic-, aliphatic-, and alicyclic-substituted aromatic substituents, as well as cyclic substituents wherein the ring is completed through another portion of the molecule (e.g., two substituents together form an alicyclic radical); the substituted hydrocarbon substituents, that is, substituents containing non-hydrocarbon groups which, in the context of this disclosure, do not alter the predominantly hydrocarbon substituent, hydroxy, alkoxy, nitro); hetero-atom containing substituents, that is, substituents which, while having a predominantly hydrocarbon character, in the context of this disclosure, contain other than carbon in a ring or chain otherwise composed of carbon atoms. Heteroatoms include sulfur, oxygen, nitrogen, and encompass substituents as pyridyl, furyl, thienyl and imidazolyl. In general, no more than two, preferably no more than one, non-hydrocarbon substituent will be present for every ten carbon atoms in the hydrocarbyl group; typically, there will be no non-hydrocarbon substituents in the hydrocarbyl group.
[0053] Examples of straight chain hydrocarbyl groups include methyl, ethyl, n-propyl, n-butyl, n-hexyl, n-octyl, n-decyl, n-dodecyl, n-tetradecyl, stearyl, n-hexadecyl, n-octadecyl, oleyl, and cetyl. Examples of branched-chain hydrocarbon groups include isopropyl, isobutyl, secondary butyl, tertiary butyl, neopentyl, 2-ethylhexyl, and 2,6-dimethylheptyl. Examples of cyclic groups include cyclobutyl, cyclopentyl, methylcyclopentyl, cyclohexyl, methylcyclohexyl, cycloheptyl, and cyclooctyl. A few examples of aromatic hydrocarbyl groups and mixed aromatic-aliphatic hydrocarbyl groups include phenyl, methylphenyl, tolyl, and naphthyl.
[0054] The substituent groups can also comprise a mixture of hydrocarbyl groups derived from commercial alcohols. Examples of some monohydric alcohols and alcohol mixtures include the commercially available "Alfol.TM." alcohols marketed by Continental Oil Corporation. Alfol.TM. 810, for instance, is a mixture containing alcohols consisting essentially of straight chain, primary alcohols having from 8 to 12 carbon atoms. Alfol.TM. 12 is a mixture of mostly C12 fatty alcohols; Alfol.TM. 22+ comprises C 18-28 primary alcohols having mostly C 22 alcohols, and so on. Various mixtures of monohydric fatty alcohols derived from naturally occurring triglycerides and ranging in chain length from C8 to C18 are available from Procter & Gamble Company. "Neodol.TM." alcohols are available from Shell Chemical Co., where, for instance, Neodol.TM. 25 is a mixture of C 12 to C 15 alcohols.
[0055] In an embodiment, the phosphorus compounds useful in the present disclosure are prepared by well known reactions. One route the reaction of an alcohol or a phenol with phosphorus trichloride or by a transesterification reaction. Alcohols and phenols can be reacted with phosphorus pentoxide to provide a mixture of an alkyl or aryl phosphoric acid and a dialkyl or diaryl phosphoric acid. Alkyl phosphates can also be prepared by the oxidation of the corresponding phosphites. In any case, the reaction can be conducted with moderate heating. Moreover, various phosphorus esters can be prepared by reaction using other phosphorus esters as starting materials. Thus, medium chain (C8 to C22) phosphorus esters have been prepared by reaction of dimethylphosphite with a mixture of medium-chain alcohols by means of a thermal transesterification or an acid- or base-catalyzed transesterification; see for example U.S. Pat. No. 4,652,416. Most such materials are also commercially available; for instance, triphenyl phosphite is available from Albright and Wilson as Duraphos TPP.TM.; di-n-butyl hydrogen phosphite from Albright and Wilson as Duraphos DBHP.TM.; and triphenylthiophosphate from BASF as Irgalube TPPT.TM..
[0056] Organic triesters of phosphorus acids are also employed as ashless antiwear additives. Typical esters include triarylphosphates, trialkyl phosphates, neutral alkylaryl phosphates, alkoxyalkyl phosphates, triaryl phosphite, trialkylphosphite, neutral alkyl aryl phosphites, neutral phosphonate esters and neutral phosphine oxide esters. In one embodiment, the long chain dialkyl phosphonate esters are used. More preferentially, the dimethyl-, diethyl-, and dipropyl-oleyl phophonates can be used. Neutral acids of phosphorus acids are the triesters rather than an acid (HO--P) or a salt of an acid.
[0057] Any C4 to C8 alkyl or higher phosphate ester may be employed in the disclosure. For example, tributyl phosphate (TBP) and tri isooctal phosphate (TOF) can be used. The specific triphosphate ester or combination of esters can easily be selected by one skilled in the art to adjust the density, viscosity etc. of the formulated fluid. Mixed esters, such as dibutyl octyl phosphate or the like may be employed rather than a mixture of two or more trialkyl phosphates.
[0058] A trialkyl phosphate is useful as an ashless antiwear additive and to adjust the specific gravity of the formulation, but it is desirable that the specific trialkyl phosphate be a liquid at low temperatures. Consequently, a mixed ester containing at least one partially alkylated with a C3 to C4 alkyl group is very desirable, for example, 4-isopropylphenyl diphenyl phosphate or 3-butylphenyl diphenyl phosphate. Even more desirable is a triaryl phosphate produced by partially alkylating phenol with butylene or propylene to form a mixed phenol which is then reacted with phosphorus oxychloride as taught in U.S. Pat. No. 3,576,923.
[0059] Any mixed triaryl phosphate (TAP) esters may be used as ashless antiwear additives including cresyl diphenyl phosphate, tricresyl phosphate, mixed xylyl cresyl phosphates, lower alkylphenyl/phenyl phosphates, such as mixed isopropylphenyl/phenyl phosphates, t-butylphenyl phenyl phosphates.
[0060] The phosphoric acid ester, thiophosphoric acid ester, and amine salt thereof functions to enhance the lubricating performances, and can be selected from known compounds conventionally employed as ashless antiwear additives. Generally employed are phosphoric acid esters, or an amine salt thereof which has an alkyl group, an alkenyl group, an alkylaryl group, or an aralkyl group, any of which contains approximately 3 to 30 carbon atoms.
[0061] In an embodiment, examples of the phosphoric acid esters include aliphatic phosphoric acid esters such as triisopropyl phosphate, tributyl phosphate, ethyl dibutyl phosphate, trihexyl phosphate, tri-2-ethylhexyl phosphate, trilauryl phosphate, tristearyl phosphate, and trioleyl phosphate; and aromatic phosphoric acid esters such as benzyl phenyl phosphate, allyl diphenyl phosphate, triphenyl phosphate, tricresyl phosphate, ethyl diphenyl phosphate, cresyl diphenyl phosphate, dicresyl phenyl phosphate, ethylphenyl diphenyl phosphate, diethylphenyl phenyl phosphate, propylphenyl diphenyl phosphate, dipropylphenyl phenyl phosphate, triethylphenyl phosphate, tripropylphenyl phosphate, butylphenyl diphenyl phosphate, dibutylphenyl phenyl phosphate, and tributylphenyl phosphate. Preferably, the phosphoric acid ester is a trialkylphenyl phosphate.
[0062] Also employable are amine salts of the above-mentioned phosphates. Amine salts of acidic alkyl or aryl esters of the phosphoric acid and thiophosphoric acid are also employable. Preferably, the amine salt is an amine salt of trialkylphenyl phosphate or an amine salt of alkyl phosphate.
[0063] One or any combination of the compounds selected from the group consisting of a phosphoric acid ester, and an amine salt thereof may be used.
[0064] The phosphorus acid ester and/or its amine salt function as ashless antiwear additives and to enhance the lubricating performances, and can be selected from known compounds conventionally employed as ashless antiwear additives. Generally employed are a phosphorus acid ester or an amine salt thereof which has an alkyl group, an alkenyl group, an alkylaryl group, or an aralkyl group, any of which contains approximately 3 to 30 carbon atoms.
[0065] In another embodiment, examples of the phosphorus acid esters include aliphatic phosphorus acid esters such as triisopropyl phosphite, tributyl phosphite, ethyl dibutyl phosphite, trihexyl phosphite, tri-2-ethylhexylphosphite, trilauryl phosphite, tristearyl phosphite, and trioleyl phosphite; and aromatic phosphorus acid esters such as benzyl phenyl phosphite, allyl diphenylphosphite, triphenyl phosphite, tricresyl phosphite, ethyl diphenyl phosphite, tributyl phosphite, ethyl dibutyl phosphite, cresyl diphenyl phosphite, dicresyl phenyl phosphite, ethylphenyl diphenyl phosphite, diethylphenyl phenyl phosphite, propylphenyl diphenyl phosphite, dipropylphenyl phenyl phosphite, triethylphenyl phosphite, tripropylphenyl phosphite, butylphenyl diphenyl phosphite, dibutylphenyl phenyl phosphite, and tributylphenyl phosphite. Also favorably employed are dilauryl phosphite, dioleyl phosphite, dialkyl phosphites, and diphenyl phosphite. Preferably, the phosphorus acid ester is a dialkyl phosphite or a trialkyl phosphite.
[0066] In an embodiment, the phosphate salt may be derived from a polyamine. The polyamines include alkoxylated diamines, fatty polyamine diamines, alkylenepolyamines, hydroxy containing polyamines, condensed polyamines arylpolyamines, and heterocyclic polyamines. Commercially available examples of alkoxylated diamines include those amine where y in the above formula is one. Examples of these amines include Ethoduomeen T/13 and T/20 which are ethylene oxide condensation products of N-tallowtrimethylenediamine containing 3 and 10 moles of ethylene oxide per mole of diamine, respectively.
[0067] In another embodiment, the polyamine is a fatty diamine. The fatty diamines include mono- or dialkyl, symmetrical or asymmetrical ethylene diamines, propane diamines (1,2, or 1,3), and polyamine analogs of the above. Suitable commercial fatty polyamines are Duomeen C. (N-coco-1,3-diaminopropane), Duomeen S (N-soya-1,3-diaminopropane), Duomeen T (N-tallow-1,3-diaminopropane), and Duomeen O (N-oleyl-1,3-diaminopropane). "Duomeens" are commercially available from Armak Chemical Co., Chicago, Ill.
[0068] Such alkylenepolyamines include methylenepolyamines, ethylenepolyamines, butylenepolyamines, propylenepolyamines, pentylenepolyamines, etc. The higher homologs and related heterocyclic amines such as piperazines and N-amino alkyl-substituted piperazines are also included. Specific examples of such polyamines are ethylenediamine, triethylenetetramine, tris-(2-aminoethyl)amine, propylenediamine, trimethylenediamine, tripropylenetetramine, tetraethylenepentamine, hexaethyleneheptamine, pentaethylenehexamine, etc. Higher homologs obtained by condensing two or more of the above-noted alkyleneamines are similarly useful as are mixtures of two or more of the aforedescribed polyamines.
[0069] In one embodiment, the polyamine is an ethylenepolyamine. Such polyamines are described in detail under the heading Ethylene Amines in Kirk Othmer's "Encyclopedia of Chemical Technology", 2d Edition, Vol. 7, pages 22-37, Interscience Publishers, New York (1965). Ethylenepolyamines are often a complex mixture of polyalkylenepolyamines including cyclic condensation products.
[0070] Other useful types of polyamine mixtures are those resulting from stripping of the above-described polyamine mixtures to leave, as residue, what is often termed "polyamine bottoms". In general, alkylenepolyamine bottoms can be characterized as having less than 2%, usually less than 1% (by weight) material boiling below about 200 C. A typical sample of such ethylene polyamine bottoms obtained from the Dow Chemical Company of Freeport, Tex. designated "E-100". These alkylenepolyamine bottoms include cyclic condensation products such as piperazine and higher analogs of diethylenetriamine, triethylenetetramine and the like. These alkylenepolyamine bottoms can be reacted solely with the acylating agent or they can be used with other amines, polyamines, or mixtures thereof.
[0071] Another useful polyamine is a condensation reaction between at least one hydroxy compound with at least one polyamine reactant containing at least one primary or secondary amino group. The hydroxy compounds are preferably polyhydric alcohols and amines. The polyhydric alcohols are described below. (See carboxylic ester dispersants.) In one embodiment, the hydroxy compounds are polyhydric amines. Polyhydric amines include any of the above-described monoamines reacted with an alkylene oxide (e.g., ethylene oxide, propylene oxide, butylene oxide, etc.) having from two to about 20 carbon atoms, or from two to about four. Examples of polyhydric amines include tri-(hydroxypropyl)amine, tris-(hydroxymethyl)amino methane, 2-amino-2-methyl-1,3-propanediol, N,N,N',N'-tetrakis(2-hydroxypropyl)ethylenediamine, and N,N,N',N'-tetrakis(2-hydroxyethyl)ethylenediamine, preferably tris(hydroxymethyl)aminomethane (THAM).
[0072] Polyamines which react with the polyhydric alcohol or amine to form the condensation products or condensed amines, are described above. Preferred polyamines include triethylenetetramine (TETA), tetraethylenepentamine (TEPA), pentaethylenehexamine (PEHA), and mixtures of polyamines such as the above-described "amine bottoms".
[0073] Preferred ashless antiwear additives selected from phosphorus-containing ashless antiwear additives, sulfur-containing ashless antiwear additives, and phosphorus/sulfur-containing ashless antiwear additives useful in this disclosure include, for example, amine phosphates, thiophosphates, dithiophosphates, amine salts of sulfurized phosphates, and mixtures thereof, and the like.
[0074] The concentration of ashless antiwear additive selected from a phosphorus-containing ashless antiwear additive, a sulfur-containing ashless antiwear additive, and a phosphorus/sulfur-containing ashless antiwear additive in the lubricating oils of this disclosure can range from 0.05 to 4.0 weight percent, preferably 0.1 to 2.0 weight percent, and more preferably from 0.2 weight percent to 1.0 weight percent, based on the total weight of the lubricating oil.
Ashless Detergents
[0075] Illustrative ashless detergents useful in this disclosure include, for example, nonionic detergents such as polyoxyethylene, polyoxypropylene, or polyoxybutylene alkyl ethers. For reference, see "Nonionic Surfactants: Physical Chemistry" Martin J. Schick, CRC Press; 2 edition (Mar. 27, 1987). These ashless detergents are less common in engine lubricant formulations, but offer a number of advantages such as improved solubility in ester base oils.
[0076] The preferred ashless detergents of this disclosure include ashless detergents soluble in a branched polyol ester, preferably a mono- or dipentaerythritol ester of at least one branched mono carboxylic acid, and more preferably the nonionic detergents.
[0077] The ashless detergent concentration in the lubricating oils of this disclosure can range from 0.5 to 6.0 weight percent, preferably 0.6 to 5.0 weight percent, and more preferably from 0.8 weight percent to 4.0 weight percent, based on the total weight of the lubricating oil.
[0078] As used herein, the detergent concentrations are given on an "as delivered" basis. Typically, the active detergent is delivered with a process oil. The "as delivered" detergent typically contains from 20 weight percent to 100 weight percent, or from 40 weight percent to 60 weight percent, of active detergent in the "as delivered" detergent product.
Amine Antioxidants
[0079] Antioxidants retard the oxidative degradation of base oils during service. Such degradation may result in deposits on metal surfaces, the presence of sludge, or a viscosity increase in the lubricant. One skilled in the art knows a wide variety of oxidation inhibitors that are useful in lubricating oil compositions. See, Klamann in Lubricants and Related Products, op cite, and U.S. Pat. Nos. 4,798,684 and 5,084,197, for example.
[0080] Useful antioxidants include amine antioxidants, preferably aromatic amine antioxidants. Aromatic amine antioxidants may be used alone or in combination with phenolic antioxidants. Typical examples of amine antioxidants include: alkylated and non-alkylated aromatic amines such as aromatic monoamines of the formula R.sup.8R.sup.9R.sup.10N where R.sup.8 is an aliphatic, aromatic or substituted aromatic group, R.sup.9 is an aromatic or a substituted aromatic group, and R.sup.10 is H, alkyl, aryl or R.sup.11S(O)xR.sup.12 where R.sup.11 is an alkylene, alkenylene, or aralkylene group, R.sup.12 is a higher alkyl group, or an alkenyl, aryl, or alkaryl group, and x is 0, 1 or 2. The aliphatic group R.sup.8 may contain from 1 to 20 carbon atoms, and preferably contains from 6 to 12 carbon atoms. The aliphatic group is an aliphatic group. Preferably, both R.sup.8 and R.sup.9 are aromatic or substituted aromatic groups, and the aromatic group may be a fused ring aromatic group such as naphthyl. Aromatic groups R.sup.8 and R.sup.9 may be joined together with other groups such as S.
[0081] Typical aromatic amine antioxidants have alkyl substituent groups of at least 6 carbon atoms. Examples of aliphatic groups include hexyl, heptyl, octyl, nonyl, and decyl. Generally, the aliphatic groups will not contain more than 14 carbon atoms. The general types of amine antioxidants useful in the present compositions include diphenylamines, phenyl naphthylamines, phenothiazines, imidodibenzyls and diphenyl phenylene diamines. Mixtures of two or more aromatic amines are also useful. Polymeric amine antioxidants can also be used. Particular examples of aromatic amine antioxidants useful in the present disclosure include: p,p'-dioctyldiphenylamine; t-octylphenyl-alpha-naphthylamine; phenyl-alpha-naphthylamine; and p-octylphenyl-alpha-naphthylamine. Polymeric aminic antioxidants derived from these diphenylamines, phenyl naphthylamines, and their mixtures can also be used. The polymeric aminic antioxidants may be available in a concentrate from with active polymeric amines in the 10 to 40 weight %. These polymeric aminic antioxidant concentrates are commercially available from, for example, Nyco S. A., under the trade name of Nycoperf AO 337, which is a concentrate containing approximately 3 weight percent active polymeric aminic antioxidant
[0082] Preferred antioxidants include arylamines and polymeric aminic antioxidants. The arylamines antioxidants may be used individually or in combination. Such additives may be used in an amount of 0.01 to 5 weight percent, preferably 0.01 to 1.5 weight percent, more preferably zero to less than 1.5 weight percent, more preferably zero to less than 1 weight percent. The polymeric aminic antioxidant concentrates may be used in an amount of 1 to 15 weight percent, preferably 7 to 13 weight percent.
[0083] The preferred amine antioxidants in this disclosure include polymeric or oligomeric amines which are the polymerization reaction products of one or more substituted or hydrocarbyl-substituted diphenyl amities, one or more unsubstituted or hydrocarbyl-substituted phenyl naphthyl amines, or both one or more of unsubstituted or hydrocarbyl-substituted diphenylamine with one or more unsubstituted or hydrocarbyl-substituted phenyl naphthylamine. A representative schematic is presented below:
wherein (a) and (b) each range from zero to 10, preferably zero to 5, more preferably zero to 3, most preferably 1 to 3, provided (a)+(b) is at least 2; for example:
##STR00001##
wherein R.sup.2 is a styrene or C.sub.1 to C.sub.30 alkyl, R.sup.3 is a styrene or C.sub.1 to C.sub.30 alkyl, R.sup.4 is a styrene or C.sub.1 to C.sub.30 alkyl, preferably R2 is a C.sub.1 to C.sub.30 alkyl, R3 is a C.sub.1 to C.sub.30 alkyl, R4 is a C.sub.1 to C.sub.30 alkyl, more preferably R2 is a C.sub.4 to C.sub.10 alkyl, R3 is a C.sub.4 to C.sub.10 alkyl and R4 is a C.sub.4 to C.sub.10 alkyl, p, q and y individually range from 0 to up to the valence of the aryl group to which the respective R groups are attached, preferably at least one of p, q and y range from 1 to up to the valence of the aryl group to which the respective R group(s) are attached, more preferably p, q and y each individually range from at least 1 to up to the valence of the aryl group to which the respective R groups are attached.
[0084] Other more extensive oligomers are within the scope of this disclosure, but materials of formulae A, B, C and D are preferred. Examples can be also found in U.S. Pat. No. 8,492,321, which is herein incorporated by reference.
Other Lubricating Oil Base Stocks
[0085] A wide range of lubricating base oils is known in the art. Lubricating base oils that are useful in the present disclosure are both natural oils, and synthetic oils, and unconventional oils (or mixtures thereof) can be used unrefined, refined, or rerefined (the latter is also known as reclaimed or reprocessed oil). Unrefined oils are those obtained directly from a natural or synthetic source and used without added purification. These include shale oil obtained directly from retorting operations, petroleum oil obtained directly from primary distillation, and ester oil obtained directly from an esterification process. Refined oils are similar to the oils discussed for unrefined oils except refined oils are subjected to one or more purification steps to improve at least one lubricating oil property. One skilled in the art is familiar with many purification processes. These processes include solvent extraction, secondary distillation, acid extraction, base extraction, filtration, and percolation. Rerefined oils are obtained by processes analogous to refined oils but using an oil that has been previously used as a feed stock.
[0086] Groups I, II, III, IV and V are broad base oil stock categories developed and defined by the American Petroleum Institute (API Publication 1509; www.API.org) to create guidelines for lubricant base oils. Group I base stocks have a viscosity index of between about 80 to 120 and contain greater than about 0.03% sulfur and/or less than about 90% saturates. Group II base stocks have a viscosity index of between about 80 to 120, and contain less than or equal to about 0.03% sulfur and greater than or equal to about 90% saturates. Group III stocks have a viscosity index greater than about 120 and contain less than or equal to about 0.03% sulfur and greater than about 90% saturates. Group IV includes polyalphaolefins (PAO). Group V base stock includes base stocks not included in Groups I-IV. The table below summarizes properties of each of these five groups.
TABLE-US-00001 TABLE 1 Base Oil Properties Saturates Sulfur Viscosity Index Group I <90 and/or >0.03% and .gtoreq.80 and <120 Group II .gtoreq.90 and .ltoreq.0.03% and .gtoreq.80 and <120 Group III .gtoreq.90 and .ltoreq.0.03% and .gtoreq.120 Group IV Polyalphaolefins (PAO) Group V All other base oil stocks not included in Groups I, II, III or IV
[0087] Natural oils include animal oils, vegetable oils (castor oil and lard oil, for example), and mineral oils. Animal and vegetable oils possessing favorable thermal oxidative stability can be used. Of the natural oils, mineral oils are preferred. Mineral oils vary widely as to their crude source, for example, as to whether they are paraffinic, naphthenic, or mixed paraffinic-naphthenic. Oils derived from coal or shale are also useful. Natural oils vary also as to the method used for their production and purification, for example, their distillation range and whether they are straight run or cracked, hydrorefined, or solvent extracted.
[0088] Group II and/or Group III hydroprocessed or hydrocracked base stocks, including synthetic oils such as polyalphaolefins, alkyl aromatics and synthetic esters are also well known base stock oils.
[0089] Synthetic oils include hydrocarbon oil. Hydrocarbon oils include oils such as polymerized and interpolymerized olefins (polybutylenes, polypropylenes, propylene isobutylene copolymers, ethylene-olefin copolymers, and ethylene-alphaolefin copolymers, for example). Polyalphaolefin (PAO) oil base stocks are commonly used synthetic hydrocarbon oil. By way of example, PAOs derived from C.sub.8, C.sub.10, C.sub.12, C.sub.14 olefins or mixtures thereof may be utilized. See U.S. Pat. Nos. 4,956,122; 4,827,064; and 4,827,073.
[0090] The number average molecular weights of the PAOs, which are known materials and generally available on a major commercial scale from suppliers such as ExxonMobil Chemical Company, Chevron Phillips Chemical Company, BP, and others, typically vary from about 250 to about 3,000, although PAO's may be made in viscosities up to about 150 cSt (100.degree. C.). The PAOs are typically comprised of relatively low molecular weight hydrogenated polymers or oligomers of alphaolefins which include, but are not limited to, C.sub.2 to about C.sub.32 alphaolefins with the C.sub.8 to about C.sub.16 alphaolefins, such as 1-hexene, 1-octene, 1-decene, 1-dodecene and the like, being preferred. The preferred polyalphaolefins are poly-1-hexene, poly-1-octene, poly-1-decene and poly-1-dodecene and mixtures thereof and mixed olefin-derived polyolefins. However, the dimers of higher olefins in the range of C.sub.14 to C.sub.18 may be used to provide low viscosity base stocks of acceptably low volatility. Depending on the viscosity grade and the starting oligomer, the PAOs may be predominantly trimers and tetramers of the starting olefins, with minor amounts of the higher oligomers, having a viscosity range of 1.5 to 12 cSt. PAO fluids of particular use may include 3.0 cSt, 3.4 cSt, and/or 3.6 cSt and combinations thereof. Bi-modal mixtures of PAO fluids having a viscosity range of 1.5 to 150 cSt may be used if desired.
[0091] The PAO fluids may be conveniently made by the polymerization of an alphaolefin in the presence of a polymerization catalyst such as the Friedel-Crafts catalysts including, for example, aluminum trichloride, boron trifluoride or complexes of boron trifluoride with water, alcohols such as ethanol, propanol or butanol, carboxylic acids or esters such as ethyl acetate or ethyl propionate. For example the methods disclosed by U.S. Pat. No. 4,149,178 or 3,382,291 may be conveniently used herein. Other descriptions of PAO synthesis are found in the following U.S. Pat. Nos. 3,742,082; 3,769,363; 3,876,720; 4,239,930; 4,367,352; 4,413,156; 4,434,408; 4,910,355; 4,956,122; and 5,068,487. The dimers of the C.sub.14 to Cis olefins are described in U.S. Pat. No. 4,218,330.
[0092] The alkylated naphthalene can be used as base oil or base oil component and can be any hydrocarbyl molecule that contains at least about 5% of its weight derived from a naphthenoid moiety, or its derivatives. These alkylated naphthalenes include alkyl naphthalenes, alkyl naphthols, and the like. The naphthenoid group can be mono-alkylated, dialkylated, polyalkylated, and the like. The naphthenoid group can be mono- or poly-functionalized. The naphthenoid group can also be derived from natural (petroleum) sources, provided at least about 5% of the molecule is comprised of the naphthenoid moiety. Viscosities at 100.degree. C. of approximately 3 cSt to about 50 cSt are preferred, with viscosities of approximately 3.4 cSt to about 20 cSt often being more preferred for the naphthylene component. In one embodiment, an alkyl naphthalene where the alkyl group is primarily comprised of 1-hexadecene is used. Other alkylates of naphthalene can be advantageously used. Naphthalene or methyl naphthalene, for example, can be alkylated with olefins such as octene, decene, dodecene, tetradecene or higher, mixtures of similar olefins, and the like.
[0093] Alkylated naphthalenes of the present disclosure may be produced by well-known Friedel-Crafts alkylation of aromatic compounds. See Friedel-Crafts and Related Reactions, Olah, G. A. (ed.), Inter-science Publishers, New York, 1963. For example, an aromatic compound, such as naphthalene, is alkylated by an olefin, alkyl halide or alcohol in the presence of a Friedel-Crafts catalyst. See Friedel-Crafts and Related Reactions, Vol. 2, part 1, chapters 14, 17, and 18, See Olah, G. A. (ed.), Inter-science Publishers, New York, 1964. Many homogeneous or heterogeneous, solid catalysts are known to one skilled in the art. The choice of catalyst depends on the reactivity of the starting materials and product quality requirements. For example, strong acids such as AlCl.sub.3, BF.sub.3, or HF may be used. In some cases, milder catalysts such as FeCl.sub.3 or SnCl.sub.4 are preferred. Newer alkylation technology uses zeolites or solid super acids.
[0094] Mixtures of alkylated naphthalene base stocks with other lubricating oil base stocks (e.g., Groups I, II, III, IV and V base stocks) may be useful in the lubricating oil formulations of this disclosure.
[0095] The alkylated naphthalene can be present in an amount of from about 30 to about 99.8 weight percent, or from about 35 to about 95 weight percent, or from about 40 to about 90 weight percent, or from about 45 to about 85 weight percent, or from about 50 to about 80 weight percent, or from about 55 to about 75 weight percent, or from about 60 to about 70 weight percent, based on the total weight of the formulated oil.
[0096] Other useful lubricant oil base stocks include wax isomerate base stocks and base oils, comprising hydroisomerized waxy stocks (e.g. waxy stocks such as gas oils, slack waxes, fuels hydrocracker bottoms, etc.), hydroisomerized Fischer-Tropsch waxes, Gas-to-Liquids (GTL) base stocks and base oils, and other wax isomerate hydroisomerized base stocks and base oils, or mixtures thereof. Fischer-Tropsch waxes, the high boiling point residues of Fischer-Tropsch synthesis, are highly paraffinic hydrocarbons with very low sulfur content. The hydroprocessing used for the production of such base stocks may use an amorphous hydrocracking/hydroisomerization catalyst, such as one of the specialized lube hydrocracking (LHDC) catalysts or a crystalline hydrocracking/hydroisomerization catalyst, preferably a zeolitic catalyst. For example, one useful catalyst is ZSM-48 as described in U.S. Pat. No. 5,075,269, the disclosure of which is incorporated herein by reference in its entirety. Processes for making hydrocracked/hydroisomerized distillates and hydrocracked/hydroisomerized waxes are described, for example, in U.S. Pat. Nos. 2,817,693; 4,975,177; 4,921,594 and 4,897,178 as well as in British Patent Nos. 1,429,494; 1,350,257; 1,440,230 and 1,390,359. Each of the aforementioned patents is incorporated herein in their entirety. Particularly favorable processes are described in European Patent Application Nos. 464546 and 464547, also incorporated herein by reference. Processes using Fischer-Tropsch wax feeds are described in U.S. Pat. Nos. 4,594,172 and 4,943,672, the disclosures of which are incorporated herein by reference in their entirety.
[0097] Gas-to-Liquids (GTL) base oils, Fischer-Tropsch wax derived base oils, and other wax-derived hydroisomerized (wax isomerate) base oils be advantageously used in the instant disclosure, and may have useful kinematic viscosities at 100.degree. C. of about 3 cSt to about 50 cSt, preferably about 3 cSt to about 30 cSt, more preferably about 3.5 cSt to about 25 cSt, as exemplified by GTL 4 with kinematic viscosity of about 4.0 cSt at 100.degree. C. and a viscosity index of about 141. These Gas-to-Liquids (GTL) base oils, Fischer-Tropsch wax derived base oils, and other wax-derived hydroisomerized base oils may have useful pour points of about -20.degree. C. or lower, and under some conditions may have advantageous pour points of about -25.degree. C. or lower, with useful pour points of about -30.degree. C. to about -40.degree. C. or lower. Useful compositions of Gas-to-Liquids (GTL) base oils, Fischer-Tropsch wax derived base oils, and wax-derived hydroisomerized base oils are recited in U.S. Pat. Nos. 6,080,301; 6,090,989, and 6,165,949 for example, and are incorporated herein in their entirety by reference.
[0098] The hydrocarbyl aromatics can be used as base oil or base oil component and can be any hydrocarbyl molecule that contains at least about 5% of its weight derived from an aromatic moiety such as a benzenoid moiety or naphthenoid moiety, or their derivatives. These hydrocarbyl aromatics include alkyl benzenes, alkyl naphthalenes, alkyl diphenyl oxides, alkyl naphthols, alkyl diphenyl sulfides, alkylated bis-phenol A, alkylated thiodiphenol, and the like. The aromatic can be mono-alkylated, dialkylated, polyalkylated, and the like. The aromatic can be mono- or poly-functionalized. The hydrocarbyl groups can also be comprised of mixtures of alkyl groups, alkenyl groups, alkynyl, cycloalkyl groups, cycloalkenyl groups and other related hydrocarbyl groups. The hydrocarbyl groups can range from about C.sub.6 up to about C.sub.60 with a range of about C.sub.8 to about C.sub.20 often being preferred. A mixture of hydrocarbyl groups is often preferred, and up to about three such substituents may be present. The hydrocarbyl group can optionally contain sulfur, oxygen, and/or nitrogen containing substituents. The aromatic group can also be derived from natural (petroleum) sources, provided at least about 5% of the molecule is comprised of an above-type aromatic moiety. Viscosities at 100.degree. C. of approximately 3 cSt to about 50 cSt are preferred, with viscosities of approximately 3.4 cSt to about 20 cSt often being more preferred for the hydrocarbyl aromatic component. In one embodiment, an alkyl naphthalene where the alkyl group is primarily comprised of 1-hexadecene is used. Other alkylates of aromatics can be advantageously used. Naphthalene or methyl naphthalene, for example, can be alkylated with olefins such as octene, decene, dodecene, tetradecene or higher, mixtures of similar olefins, and the like. Useful concentrations of hydrocarbyl aromatic in a lubricant oil composition can be about 2% to about 25%, preferably about 4% to about 20%, and more preferably about 4% to about 15%, depending on the application.
[0099] Alkylated aromatics such as the hydrocarbyl aromatics of the present disclosure may be produced by well-known Friedel-Crafts alkylation of aromatic compounds. See Friedel-Crafts and Related Reactions, Olah, G. A. (ed.), Inter-science Publishers, New York, 1963. For example, an aromatic compound, such as benzene or naphthalene, is alkylated by an olefin, alkyl halide or alcohol in the presence of a Friedel-Crafts catalyst. See Friedel-Crafts and Related Reactions, Vol. 2, part 1, chapters 14, 17, and 18, See Olah, G. A. (ed.), Inter-science Publishers, New York, 1964. Many homogeneous or heterogeneous, solid catalysts are known to one skilled in the art. The choice of catalyst depends on the reactivity of the starting materials and product quality requirements. For example, strong acids such as AlCl.sub.3, BF.sub.3, or HF may be used. In some cases, milder catalysts such as FeCl.sub.3 or SnCl.sub.4 are preferred. Newer alkylation technology uses zeolites or solid super acids.
[0100] Other useful fluids of lubricating viscosity include non-conventional or unconventional base stocks that have been processed, preferably catalytically, or synthesized to provide high performance lubrication characteristics.
[0101] Non-conventional or unconventional base stocks/base oils include one or more of a mixture of base stock(s) derived from one or more Gas-to-Liquids (GTL) materials, as well as isomerate/isodewaxate base stock(s) derived from natural wax or waxy feeds, mineral and or non-mineral oil waxy feed stocks such as slack waxes, natural waxes, and waxy stocks such as gas oils, waxy fuels hydrocracker bottoms, waxy raffinate, hydrocrackate, thermal crackates, or other mineral, mineral oil, or even non-petroleum oil derived waxy materials such as waxy materials received from coal liquefaction or shale oil, and mixtures of such base stocks.
[0102] GTL materials are materials that are derived via one or more synthesis, combination, transformation, rearrangement, and/or degradation/deconstructive processes from gaseous carbon-containing compounds, hydrogen-containing compounds and/or elements as feed stocks such as hydrogen, carbon dioxide, carbon monoxide, water, methane, ethane, ethylene, acetylene, propane, propylene, propyne, butane, butylenes, and butynes. GTL base stocks and/or base oils are GTL materials of lubricating viscosity that are generally derived from hydrocarbons; for example, waxy synthesized hydrocarbons, that are themselves derived from simpler gaseous carbon-containing compounds, hydrogen-containing compounds and/or elements as feed stocks. GTL base stock(s) and/or base oil(s) include oils boiling in the lube oil boiling range (1) separated/fractionated from synthesized GTL materials such as, for example, by distillation and subsequently subjected to a final wax processing step which involves either or both of a catalytic dewaxing process, or a solvent dewaxing process, to produce lube oils of reduced/low pour point; (2) synthesized wax isomerates, comprising, for example, hydrodewaxed or hydroisomerized cat and/or solvent dewaxed synthesized wax or waxy hydrocarbons; (3) hydrodewaxed or hydroisomerized cat and/or solvent dewaxed Fischer-Tropsch (F-T) material (i.e., hydrocarbons, waxy hydrocarbons, waxes and possible analogous oxygenates); preferably hydrodewaxed or hydroisomerized/followed by cat and/or solvent dewaxing dewaxed F-T waxy hydrocarbons, or hydrodewaxed or hydroisomerized/followed by cat (or solvent) dewaxing dewaxed, F-T waxes, or mixtures thereof.
[0103] GTL base stock(s) and/or base oil(s) derived from GTL materials, especially, hydrodewaxed or hydroisomerized/followed by cat and/or solvent dewaxed wax or waxy feed, preferably F-T material derived base stock(s) and/or base oil(s), are characterized typically as having kinematic viscosities at 100.degree. C. of from about 2 mm.sup.2/s to about 50 mm.sup.2/s (ASTM D445). They are further characterized typically as having pour points of -5.degree. C. to about -40.degree. C. or lower (ASTM D97). They are also characterized typically as having viscosity indices of about 80 to about 140 or greater (ASTM D2270).
[0104] In addition, the GTL base stock(s) and/or base oil(s) are typically highly paraffinic (>90% saturates), and may contain mixtures of monocycloparaffins and multicycloparaffins in combination with non-cyclic isoparaffins. The ratio of the naphthenic (i.e., cycloparaffin) content in such combinations varies with the catalyst and temperature used. Further, GTL base stock(s) and/or base oil(s) typically have very low sulfur and nitrogen content, generally containing less than about 10 ppm, and more typically less than about 5 ppm of each of these elements. The sulfur and nitrogen content of GTL base stock(s) and/or base oil(s) obtained from F-T material, especially F-T wax, is essentially nil. In addition, the absence of phosphorous and aromatics make this materially especially suitable for the formulation of low SAP products.
[0105] The term GTL base stock and/or base oil and/or wax isomerate base stock and/or base oil is to be understood as embracing individual fractions of such materials of wide viscosity range as recovered in the production process, mixtures of two or more of such fractions, as well as mixtures of one or two or more low viscosity fractions with one, two or more higher viscosity fractions to produce a blend wherein the blend exhibits a target kinematic viscosity.
[0106] The GTL material, from which the GTL base stock(s) and/or base oil(s) is/are derived is preferably an F-T material (i.e., hydrocarbons, waxy hydrocarbons, wax).
[0107] Base oils for use in the formulated lubricating oils useful in the present disclosure are any of the variety of oils corresponding to API Group I, Group II, Group III, Group IV, and Group V oils and mixtures thereof, preferably API Group II, Group III, Group IV, and Group V oils and mixtures thereof, more preferably the Group III to Group V base oils due to their exceptional volatility, stability, viscometric and cleanliness features.
[0108] This other base oil typically is present in an amount ranging from about 0.1 to about 70 weight percent, or from about 1 to about 60 weight percent, or from about 1 to about 50 weight percent, or from about 1 to about 40 weight percent, or from about 1 to about 30 weight percent, or from about 1 to about 20 weight percent, or from about 1 to about 10 weight percent, based on the total weight of the composition. The base oil may be selected from any of the synthetic or natural oils typically used as crankcase lubricating oils for spark ignition and compression-ignited engines. The base oil conveniently has a kinematic viscosity, according to ASTM standards, of about 2.5 cSt to about 12 cSt (or mm.sup.2/s) at 100.degree. C. and preferably of about 2.5 cSt to about 9 cSt (or mm.sup.2/s) at 100.degree. C. Mixtures of synthetic and natural base oils may be used if desired. Mixtures of Group III, IV, and V may be preferable.
Other Additives
[0109] The formulated lubricating oil useful in the present disclosure may additionally contain one or more of the other commonly used lubricating oil performance additives including but not limited to dispersants, corrosion inhibitors, rust inhibitors, metal deactivators, extreme pressure additives, anti-seizure agents, wax modifiers, viscosity index improvers, viscosity modifiers, fluid-loss additives, seal compatibility agents, friction modifiers, lubricity agents, anti-staining agents, chromophoric agents, defoamants, demulsifiers, emulsifiers, densifiers, wetting agents, gelling agents, tackiness agents, colorants, and others. For a review of many commonly used additives, see Klamann in Lubricants and Related Products, Verlag Chemie, Deerfield Beach, Fla.; ISBN 0-89573-177-0. Reference is also made to "Lubricant Additives" by M. W. Ranney, published by Noyes Data Corporation of Parkridge, N J (1973); see also U.S. Pat. No. 7,704,930, the disclosure of which is incorporated herein in its entirety. These additives are commonly delivered with varying amounts of diluent oil, that may range from 5 weight percent to 50 weight percent.
[0110] The types and quantities of performance additives used in combination with the instant disclosure in lubricant compositions are not limited by the examples shown herein as illustrations.
[0111] When lubricating oil compositions contain one or more of the additives discussed above, the additive(s) are blended into the composition in an amount sufficient for it to perform its intended function. Typical amounts of such additives useful in the present disclosure are shown in Table 1 below.
[0112] It is noted that many of the additives are shipped from the additive manufacturer as a concentrate, containing one or more additives together, with a certain amount of base oil diluents. Accordingly, the weight amounts in the table below, as well as other amounts mentioned herein, are directed to the amount of active ingredient (that is the non-diluent portion of the ingredient). The weight percent (wt %) indicated below is based on the total weight of the lubricating oil composition.
TABLE-US-00002 TABLE 2 Typical Amounts of Other Lubricating Oil Components Approximate Approximate Compound wt % (Useful) wt % (Preferred) Antiwear 0.1-2 0.5-1 Dispersant 0.1-20 0.1-8 Detergent 0.1-20 0.1-8 Antioxidant 0.1-5 0.1-1.5 Friction Modifier 0.01-5 0.01-1.5 Pour Point Depressant 0.0-5 0.01-1.5 (PPD) Anti-foam Agent 0.001-3 0.001-0.15 Viscosity Index Improver 0.0-8 0.1-6 (pure polymer basis) Inhibitor and Antirust 0.01-5 0.01-1.5
[0113] The foregoing additives are all commercially available materials. These additives may be added independently but are usually precombined in packages which can be obtained from suppliers of lubricant oil additives. Additive packages with a variety of ingredients, proportions and characteristics are available and selection of the appropriate package will take the requisite use of the ultimate composition into account.
[0114] The following non-limiting examples are provided to illustrate the disclosure.
EXAMPLES
[0115] Formulations and the oxidation test results are set forth in FIGS. 1 and 2. All of the ingredients used herein are commercially available.
[0116] In the testing, a bench oxidation test (Sequence IIIE screener) was conducted at 165.degree. C., under a flow of 500 ml/min of air, with 40 ppm of iron from added ferric acetylacetonate as catalyst. Oil samples were taken periodically and their viscosities at 40.degree. C. were measured with a Houillon viscometer. The time (hours) to reach 200% viscosity increase was recorded for each oil. The Sequence IIIG test was conducted in accordance with ASTM D7320.
[0117] In the High Pressure Differential Scanning calorimetry (HPDSC) test, the oxidation onset temperature is measured with a Universal V4.5A TA instrument equipped with a Q20 Tzero Pressure DSC Cell. The test method is similar to ASTM E2009-08 Test Method B except that 500 psig of air is used instead of 500 psig of oxygen and the sample size is 3.0+/-0.2 mg. The pressurized sample is first equilibrated at 50 C and then ramped up to 400 C at 10 C/min.
[0118] In FIG. 1, comparative Example 1 is a typical synthetic engine oil with conventional synthetic base oil, viscosity index improver, dispersant, detergent, antioxidant, pour point depressant, antifoam additive, etc. Its Sequence IIIE screener bench oxidation test result was 322 hours and its Seq. IIIG engine test was 54.3% increase in viscosity. Comparative Example 2 is a PAO based engine oil formulation and its components are shown in FIG. 1. It contains a mixture of primary and secondary zinc dialkyldithiophosphate, a molybdenum dialkyldithiophosphate, a calcium salicylate detergent, and other additives. Its Sequence IIIE screener bench oxidation test result was 182 hours. Comparative Example 3 contains a monoester iso-octadecyl iso-octadecanoate (2-(4,4-dimethylpentan-2-yl)-5,7,7-trimethyloctyl 2-(4,4-dimethylpentan-2-yl)-5,7,7-trimethyloctanoate) base oil, a secondary zinc dialkyldithiophosphate, a non-ionic detergent, and other additives. Its Sequence IIIE screener bench oxidation result was 236 hours. Example 1 contains the same base oil and additives except the 1.0 wt % secondary zinc dialkyldithiophosphate was replaced by 0.25 wt % of an amine phosphate and 0.1 wt % of a dithiophosphate. Unexpectedly, the Sequence IIIE screener bench oxidation test result improved significantly to 993 hours.
[0119] Further, in FIG. 1, Comparative Example 4 contains a pentaerythritol ester of highly branched acids (Nycoperf 1040X), a phenolic antioxidant, a p, p'-dioctyldiphenylamine antioxidant, a secondary zinc dialkyldithiophosphate, and a molybdenum dialkyldithiophosphate. Its Sequence IIIE screener bench oxidation test result was 128 hours. In Example 2, the p, p'-dioctyldiphenylamine antioxidant in Comparative Example 4 was replaced by an polymerized aminic antioxidant concentrate and the secondary zinc dialkyldithiophosphate and molybdenum dialkyldithiophosphate in Comparative Example 4 were replaced by an amine phosphate and Hitec 3333, a dithiophosphate, the Sequence IIIE screener result was improved significantly to over 1100 hours. The Sequence IIIG engine test also showed only 26.1% increase in viscosity.
[0120] In FIG. 2, Example 3 contains polymerized aminic antioxidant concentrate, amine phosphate, dithiophosphate, and is based on bis (3,5,5-trimethylhexyl) adipate ester. Its Sequence IIIE screener result was 393 hours. In Example 4, TMP ester of 3,5,5-trimethylhexanoic acid and isobutanoic acid (50/50) was used. Its Sequence IIIE screener result was 421 hours. Examples 5 and 6 were based on TMP ester of 3,5,5-trimethylhexanoic acid and neopentanoic acid (50/50). The only differences were that Example 5 used an octylated/butylated diphenylamine antioxidant and a viscosity index improver while Example 6 used a polymerized aminic antioxidant concentrate and no viscosity index improver. The Sequence IIIE screener results for Examples 5 and 6 were 513 and 734 hours, respectively, indicating the benefit of polymerized aminic antioxidant concentrate. Examples 7 and 8 were based on TMP ester of 3,5,5-trimethylhexanoic acid. The only differences were that Example 7 used an octylated/butylated diphenylamine antioxidant and a viscosity index improver while Example 8 used a polymerized aminic antioxidant concentrate and no viscosity index improver. The Sequence IIIE screener results for Examples 7 and 8 were both higher than 1100, respectively.
[0121] By comparing Examples 7 and 8 with Examples 5 and 6, it is clear that the longer alkyl chain length is beneficial in oxidation stability in these ashless formulations. Example 9 is based on a mixture of bis (3,5,5-trimethylhexyl) adipate and TMP ester of 3,5,5-trimethylhexanoic acid. Its Sequence IIIE screener result was 782 hours. Examples 10-12 are the same as Example 8 but with different combination of ashless antiwear additives. Example 10 contains 0.25 wt % of amine phosphate and 0.2 wt % of Additin RC 2411, a sulfurized triglyceride. Example 11 contains 0.2 wt % of Additin RC 2411, a sulfurized triglyceride and 0.28 wt % of Additin RC3890, and a dithiophosphate. Example 12 contains 0.2 wt % of Additin RC 2411, sulfurized triglyceride and 0.51 wt % of Additin RC 3770, an amine salt of acid phosphate based on C8 alcohol. The Sequence IIIE screener results of Examples 10-12 are all higher than 1100 hours. It is also surprising to observe that the HPDSC results of Example 12 are the best.
[0122] In FIG. 3, Examples 13-16 showed additional combinations of Additin RC 2411, a sulfurized triglyceride, and Additin RC 3770, an amine salt of acid phosphate based on C8 alcohol and the Sequence IIIE screener and HPDSC results.
PCT and EP Clauses:
[0123] 1. A method for improving oxidative stability of a lubricating oil in an engine or other mechanical component lubricated with the lubricating oil by using as the lubricating oil an ashless formulated oil, said ashless formulated oil having a composition comprising a lubricating oil base stock as a major component; and a mixture of (i) at least one ashless antiwear additive, (ii) at least one ashless detergent, and (iii) at least one aminic antioxidant, as minor components; wherein the lubricating oil base stock comprises at least one branched polyol ester, which is obtained by reacting one or more polyhydric alcohols with one or more branched mono-carboxylic acids containing at least 4 carbon atoms, and wherein the lubricating oil base stock is present in an amount from 30 to 99.8 mass percent, based on the total mass of the lubricating oil.
[0124] 2. The method of clause 1 wherein oxidative stability is improved, as compared to oxidative stability achieved using a lubricating oil other than the ashless formulated oil.
[0125] 3. The method of clauses 1 and 2 wherein the one or more polyhydric alcohols are selected from the group consisting of trimethylol propane, pentaerythritol, neopentyl glycol, trimethylol ethane, 2-methyl-2-propyl-1,3-propanediol, and dipentaerythritol.
[0126] 4. The method of clauses 1-3 wherein the one or more branched mono-carboxylic acids containing at least 4 carbon atoms are selected from the group consisting of 3,5,5-trimethyl hexanoic acid (TMH), 2,2-dimethyl propionic acid (neopentanoic acid), neoheptanoic acid, neooctanoic acid, neononanoic acid, iso-hexanoic acid, neodecanoic acid, 2-ethyl hexanoic acid (2EH), isoheptanoic acid, isooctanoic acid, isononanoic acid, and isodecanoic acid.
[0127] 5. The method of clauses 1-4 wherein the at least one branched polyol ester is selected from the group consisting of trimethylol propane ester of 3,5,5-trimethyl hexanoic acid (TMH), trimethylol propane ester of 2,2-dimethyl propionic acid (neopentanoic acid), trimethylol propane ester of neoheptanoic acid, trimethylol propane ester of neooctanoic acid, trimethylol propane ester of neononanoic acid, trimethylol propane ester of iso-hexanoic acid, trimethylol propane ester of neodecanoic acid, trimethylol propane ester of 2-ethyl hexanoic acid (2EH), trimethylol propane ester of isoheptanoic acid, trimethylol propane ester of isooctanoic acid, trimethylol propane ester of isononanoic acid, and trimethylol propane ester of isodecanoic acid.
[0128] 6. The method of clauses 1-5 wherein the at least one branched polyol ester is selected from the group consisting of pentaerythritol ester of 3,5,5-trimethyl hexanoic acid (TMH), pentaerythritol ester of 2,2-dimethyl propionic acid (neopentanoic acid), pentaerythritol ester of neoheptanoic acid, pentaerythritol ester of neooctanoic acid, pentaerythritol ester of neononanoic acid, pentaerythritol ester of iso-hexanoic acid, pentaerythritol ester of neodecanoic acid, pentaerythritol ester of 2-ethyl hexanoic acid (2EH), pentaerythritol ester of isoheptanoic acid, pentaerythritol ester of isooctanoic acid, pentaerythritol ester of isononanoic acid, and pentaerythritol ester of isodecanoic acid.
[0129] 7. The method of clauses 1-6 wherein the at least one ashless antiwear additive is selected from the group consisting of an amine phosphate, a thiophosphate, a dithiophosphate, an amine salt of sulfurized phosphate, an alkylated triphenyl phosphorothionate, a sulfurized triglyceride, and mixtures thereof.
[0130] 8. The method of clauses 1-7 wherein the at least one ashless detergent is selected from the group consisting of a polyoxyethylene alkyl ether, a polyoxypropylene alkyl ether, and a polyoxybutylene alkyl ether.
[0131] 9. The method of clauses 1-8 wherein the at least one aminic antioxidant is selected from the group consisting of p,p'-dioctyldiphenylamine, octylated phenyl-alpha-naphthylamine, octylated/butylated diphenylamine, and a polymeric aminic antioxidant.
[0132] 10. The method of clauses 1-9 wherein the at least one ashless antiwear additive is present in an amount from 0.01 to 1.2 mass percent, based on the total mass of the lubricating oil, the at least one ashless detergent is present in an amount from 0.01 to 6 mass percent, based on the total mass of the lubricating oil, and the at least one aminic antioxidant is present in an amount from 0.01 to 5 mass percent, based on the total mass of the lubricating oil.
[0133] 11. The method of clauses 1-10 wherein the ashless formulated oil further comprises one or more of a viscosity modifier, dispersant, pour point depressant, corrosion inhibitor, metal deactivator, seal compatibility additive, anti-foam agent, inhibitor, and anti-rust additive.
[0134] 12. The method of clauses 1-11 wherein the engine or other mechanical component comprises a high energy density gasoline engine equipped with turbo chargers, or gasoline particular filters (GPFs).
[0135] 13. The method of clauses 1-12 wherein the lubricating oil is a passenger vehicle engine oil (PVEO).
[0136] 14. An ashless lubricating oil having a composition comprising a lubricating oil base stock as a major component; and a mixture of (i) at least one ashless antiwear additive, (ii) at least one ashless detergent, and (iii) at least one aminic antioxidant, as minor components; wherein the lubricating oil base stock comprises at least one branched polyol ester, which is obtained by reacting one or more polyhydric alcohols with one or more branched mono-carboxylic acids containing at least 4 carbon atoms; and wherein the lubricating oil base stock is present in an amount from 30 to 99.8 mass percent, based on the total mass of the ashless lubricating oil.
[0137] 15. A method for improving high temperature stability of a lubricating oil in an engine or other mechanical component lubricated with the lubricating oil by using as the lubricating oil an ashless formulated oil, said ashless formulated oil having a composition comprising a lubricating oil base stock as a major component; and a mixture of (i) at least one ashless antiwear additive, (ii) at least one ashless detergent, and (iii) at least one aminic antioxidant, as minor components; wherein the lubricating oil base stock comprises at least one branched polyol ester, which is obtained by reacting one or more polyhydric alcohols with one or more branched mono-carboxylic acids containing at least 4 carbon atoms, and at least one alkylated naphthalene; and wherein the lubricating oil base stock is present in an amount from 30 to 99.8 mass percent, based on the total mass of the lubricating oil.
[0138] All patents and patent applications, test procedures (such as ASTM methods, UL methods, and the like), and other documents cited herein are fully incorporated by reference to the extent such disclosure is not inconsistent with this disclosure and for all jurisdictions in which such incorporation is permitted.
[0139] When numerical lower limits and numerical upper limits are listed herein, ranges from any lower limit to any upper limit are contemplated. While the illustrative embodiments of the disclosure have been described with particularity, it will be understood that various other modifications will be apparent to and can be readily made by those skilled in the art without departing from the spirit and scope of the disclosure. Accordingly, it is not intended that the scope of the claims appended hereto be limited to the examples and descriptions set forth herein but rather that the claims be construed as encompassing all the features of patentable novelty which reside in the present disclosure, including all features which would be treated as equivalents thereof by those skilled in the art to which the disclosure pertains.
[0140] The present disclosure has been described above with reference to numerous embodiments and specific examples. Many variations will suggest themselves to those skilled in this art in light of the above detailed description. All such obvious variations are within the full intended scope of the appended claims.
* * * * *
References

D00000

D00001

D00002

D00003

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.