Alkali-free Boroalumino Silicate Glasses
Ellison; Adam James ; et al.
U.S. patent application number 16/078425 was filed with the patent office on 2019-02-14 for alkali-free boroalumino silicate glasses. The applicant listed for this patent is Corning Incorporated. Invention is credited to Adam James Ellison, Sinue Gomez, Yoshiaki Kato.
| Application Number | 20190047898 16/078425 |
| Document ID | / |
| Family ID | 58108754 |
| Filed Date | 2019-02-14 |


| United States Patent Application | 20190047898 |
| Kind Code | A1 |
| Ellison; Adam James ; et al. | February 14, 2019 |
ALKALI-FREE BOROALUMINO SILICATE GLASSES
Abstract
Described herein are alkali-free, boroalumino silicate glasses exhibiting desirable physical and chemical properties for use as substrates in flat panel display devices, such as, active matrix liquid crystal displays (AMLCDs) and active matrix organic light emitting diode displays (AMOLEDs). In accordance with certain of its aspects, the glasses possess good dimensional stability as a function of temperature.
| Inventors: | Ellison; Adam James; (Corning, NY) ; Gomez; Sinue; (Corning, NY) ; Kato; Yoshiaki; (Tokyo, JP) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 58108754 | ||||||||||
| Appl. No.: | 16/078425 | ||||||||||
| Filed: | February 13, 2017 | ||||||||||
| PCT Filed: | February 13, 2017 | ||||||||||
| PCT NO: | PCT/US2017/017617 | ||||||||||
| 371 Date: | August 21, 2018 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 62298246 | Feb 22, 2016 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C03C 3/091 20130101; C03C 3/093 20130101; C03C 2203/10 20130101 |
| International Class: | C03C 3/091 20060101 C03C003/091 |
Claims
1. A glass comprising, in mole percent on an oxide basis in the ranges: SiO.sub.2 65-72, Al.sub.2O.sub.3 10-14, B.sub.2O.sub.3 3-8, MgO 2-6, CaO 3-9, SrO 0-6, and BaO 0-<1, wherein the glass exhibits a strain point >650.degree. C., an annealing point greater than or equal to about 730.degree. C., a Young's modulus >78 GPa, T.sub.35kP-T.sub.liq>30.degree. C., a density <about 2.55 g/cm.sup.3, and a CTE less than about 39.times.10.sup.-7/.degree. C.
2. The glass of claim 1, wherein the glass has an annealing point greater than about 735.degree. C., 740.degree. C., or 750.degree. C.
3. The glass of claim 1, wherein the glass has a strain point greater than or equal to about 680.degree. C., 690.degree. C., or 700.degree. C.
4. The glass of claim 1, wherein the glass has a softening point greater than or equal to about 950.degree. C., 970.degree. C., 980.degree. C., or 990.degree. C.
5. The glass of claim 1, wherein the glass has a T.sub.200P less than about 1650.degree. C., 1625.degree. C., or 1610.degree. C.
6. The glass of claim 1, wherein the glass has a T.sub.35kP less than about 1300.degree. C., 1260.degree. C., or 1250.degree. C.
7. The glass of claim 1, wherein the glass has a T.sub.35kP-T.sub.liq>40.degree. C., >50.degree. C., >60.degree. C., >65.degree. C., or >70.degree. C.
8. The glass of claim 1, wherein the glass has a specific modulus greater than 31, 31.5, or 32.
9. The glass of claim 1, wherein the glass has a CTE less than about 38.times.10.sup.-7/.degree. C. or less than about 36.times.10.sup.-7/.degree. C.
10. The glass of claim 1, wherein the glass has a Young's Modulus greater than or equal to 79 GPa, 80 GPa, or 81 GPa.
11. A glass comprising, in mole percent on an oxide basis in the ranges: SiO.sub.2 67-70, Al.sub.2O.sub.3 11-13, B.sub.2O.sub.3 4-6, MgO 3-5.5, CaO 5.5-7, SrO 3-5, and BaO 0-<1, wherein the glass exhibits a strain point >700.degree. C., an annealing point greater than or equal to about 750.degree. C., a Young's modulus >79 GPa, T.sub.35kP-T.sub.liq>65.degree. C., a density <about 2.55 g/cm.sup.3, and a CTE less than about 36.times.10.sup.-7/.degree. C.
12. The glass of claim 11, wherein the glass has a softening point greater than or equal to about 950.degree. C., 970.degree. C., 980.degree. C., or 990.degree. C.
13. The glass of claim 11, wherein the glass has a T.sub.200P less than about 1650.degree. C., 1625.degree. C., or 1610.degree. C.
14. The glass of claim 11, wherein the glass has a T.sub.35kP less than about 1300.degree. C., 1260.degree. C., or 1250.degree. C.
15. The glass of claim 11, wherein the glass has a T.sub.35kP-T.sub.liq>70.degree. C.
16. The glass of claim 11, wherein the glass has a specific modulus greater than 31, 31.5, or 32.
17. The glass of claim 11, wherein the glass has a Young's Modulus greater than or equal to 80 GPa or 81 GPa.
18. A glass comprising, in mole percent on an oxide basis in the ranges: SiO.sub.2>60, Al.sub.2O.sub.3>10, B.sub.2O.sub.3>0.1, and (MgO+CaO+SrO+BaO)/Al.sub.2O.sub.3>1.0, wherein the glass exhibits an annealing point greater than or equal to about 730.degree. C., a strain point greater than or equal to about 650.degree. C., a softening point greater than or equal to about 950.degree. C., a T.sub.200P less than about 1650.degree. C., a T.sub.35kP less than about 1300.degree. C., a T.sub.35kP-T.sub.liq>30.degree. C., a specific modulus greater than 31, and a Young's Modulus greater than or equal to 78 GPa.
19. The glass of claim 18, wherein (MgO+CaO+SrO+BaO)/Al.sub.2O.sub.3.gtoreq.1.05, .gtoreq.1.08, or .gtoreq.1.10.
20. The glass of claim 18, wherein the glass exhibits an annealing point greater than or equal to about 735.degree. C., 740.degree. C., or 750.degree. C.
21. The glass of claim 18, wherein the glass exhibits a strain point greater than or equal to about 680.degree. C., 690.degree. C., or 700.degree. C.
22. The glass of claim 18, wherein the glass exhibits a softening point greater than or equal to about 970.degree. C., 980.degree. C., or 990.degree. C.
23. The glass of claim 18, wherein the glass exhibits a T.sub.200P less than about 1625.degree. C., or 1610.degree. C.
24. The glass of claim 18, wherein the glass exhibits a T.sub.35kP less than about 1260.degree. C. or 1250.degree. C.
25. The glass of claim 18, wherein the glass exhibits a T.sub.35kP-T.sub.liq>40.degree. C., >50.degree. C., >60.degree. C., >65.degree. C., or >70.degree. C.
26. The glass of claim 18, wherein the glass exhibits a specific modulus greater than 31.5 or 32.
27. The glass of claim 18, wherein the glass exhibits a density less than or equal to 2.55 g/cm.sup.3.
28. The glass of claim 18, wherein the glass exhibits a CTE less than about 39.times.10.sup.-7/.degree. C., less than about 38.times.10.sup.-7/.degree. C., or less than about 36.times.10.sup.-7/.degree. C.
29. The glass of claim 18, wherein the glass exhibits a Young's Modulus greater than or equal to 79 GPa, 80 GPa, or 81 GPa.
30. An object comprising the glass of any one of claims 1, 11 and 18 wherein the object is produced by a downdraw sheet fabrication process.
31. An object comprising the glass of any one of claims 1, 11 and 18 wherein the object is produced by the fusion process.
32. A liquid crystal display substrate comprising the glass of any one of claims 1, 11 and 18.
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application claims the benefit of priority under 35 U.S.C. .sctn. 119 of U.S. Provisional Application Ser. No. 62/298,246 filed on Feb. 22, 2016, the content of which is relied upon and incorporated herein by reference in its entirety.
TECHNICAL FIELD
[0002] Embodiments of the present disclosure relate to display glass for liquid crystal and other suitable displays.
BACKGROUND
[0003] The production of flat panel display devices, for example, active matrix liquid crystal display devices (AMLCDs) and active matrix organic light emitting diode displays (AMOLEDs), is very complex, and the properties of the substrate glass are extremely important. First and foremost, the glass substrates used in the production of AMLCD and AMOLED devices need to have their physical dimensions tightly controlled. The downdraw sheet drawing processes and, in particular, the fusion process described in U.S. Pat. Nos. 3,338,696 and 3,682,609, both to Dockerty, are capable of producing glass sheets that can be used as substrates without requiring costly post-forming finishing operations such as lapping and polishing.
[0004] In the liquid crystal display field, there are various methods of producing the thin film transistors (TFTs) necessary for AMLCDs. Historically, panel makers have produced either large, low resolution displays utilizing amorphous-silicon (a-Si) based transistors, or small, high resolution displays utilizing poly-crystalline (p-Si) based and oxide thin film (Ox) based transistors. Although it was once thought that a-Si TFTs would be replaced by p-Si TFTs, consumer demand for low cost, large, high resolution displays, and the cost of manufacturing such large scale displays with p-Si TFTs, is driving AMLCD manufacturers to extend their use of a-Si TFTs to higher and higher resolution. These resolution increases necessitate stricter standards for the glass substrate used in the TFT manufacturing process. During a-Si, oxide or low temperature p-Si TFT fabrication, the glass substrate is held at a process temperature ranging from 350.degree. C. to 450.degree. C., whilst the thin film transistor is created. At these temperatures most AMLCD and AMOLED glass substrates undergo a process called compaction. Compaction, also referred to as thermal stability or dimensional change, is an irreversible dimensional change (shrinkage) in the glass substrate due to changes in the glass' fictive temperature. "Fictive temperature" is a concept used to indicate the structural state of a glass. Glass that is cooled quickly from a high temperature is said to have a higher fictive temperature because of the "frozen in" higher temperature structure. Glass that is cooled more slowly, or that is annealed by holding for a time near its annealing point, is said to have a lower fictive temperature.
[0005] The magnitude of compaction depends both on the process by which a glass is made and the viscoelastic properties of the glass. In the float process for producing sheet products from glass, the glass sheet is cooled relatively slowly from the melt and, thus, "freezes in" a comparatively low temperature structure into the glass. The fusion process, by contrast, results in very rapid quenching of the glass sheet from the melt, and freezes in a comparatively high temperature structure. As a result, a glass produced by the float process may undergo less compaction when compared to glass produced by the fusion process, since the driving force for compaction is the difference between the fictive temperature and the process temperature experienced by the glass during compaction. Thus, it would be desirable to minimize the level of compaction in a glass substrate that is produced by a downdraw process.
[0006] There are two approaches to minimize compaction in glass. The first is to thermally pretreat the glass to create a fictive temperature similar to the one the glass will experience during TFT manufacture. There are several difficulties with this approach. First, the multiple heating steps employed during the TFT manufacturing process create slightly different fictive temperatures in the glass that cannot be fully compensated for by this pretreatment. Second, the thermal stability of the glass becomes closely linked to the details of the TFT manufacture, which could mean different pretreatments for different end-users. Finally, pretreatment adds to processing costs and complexity.
[0007] Another approach is to slow the rate of strain at the process temperature by increasing the viscosity of the glass. This can be accomplished by raising the annealing point of the glass. The annealing point represents the temperature corresponding to a fixed viscosity for a glass, and thus an increase in annealing point equates to an increase in viscosity at fixed temperature. The challenge with this approach, however, is the production of high annealing point glass that is cost effective. The main factors impacting cost include defects and asset lifetime. In a modern continuous unit (CU) melter comprising a refractory premelt, a precious metal finer and a precious metal glass delivery stem--coupled to a fusion draw machine, four types of defects are commonly encountered: (1) gaseous inclusions (bubbles or blisters); (2) solid inclusions from refractories or from failure to properly melt the batch; (3) metallic defects consisting largely of platinum; and (4) devitrification products resulting from low liquidus viscosity or excessive devitrification at either end of the forming device, mandrel or isopipe. Glass composition has a disproportionate impact on the rate of melting, and hence on the tendency of a glass to form gaseous or solid defects and the oxidation state of the glass impacts the tendency to incorporate platinum defects. Devitrification of the glass on the forming mandrel, or isopipe, is best managed by selecting compositions with high liquidus viscosities.
[0008] Dimensional stability in the glass substrate during the TFT manufacturing process is also affected by elastic strain. There are two main causes of elastic strain experienced by the substrate. During the fusion process elastic strain, and therefore stress, can be introduced into the glass sheet upon cooling via small thermal gradients. These stresses undergo stress relaxation during the TFT manufacturing process resulting in dimensional instability. Such instability can be minimized, in the same way as compaction, via a decrease in the strain rate at the TFT process temperature by increasing the annealing point of the glass substrate. The second type of elastic strain is related to the stress imposed on the substrate when the transistor itself is deposited on glass surface. Such elastic strain is minimized by increasing the Young's modulus of the glass, such that the stress imposed on the substrate surface produces a minimal amount of strain and reduces dimensional changes seen in the glass sheet.
[0009] In addition to the problems with dimensional stability mentioned above, AMLCD and AMOLED manufacturers are finding that both demand for larger display sizes and the economics of scale are driving them to process larger sized pieces of glass in excess of 2 meters on each side. This raises several difficulties. First is simply the weight of the glass. The increase in glass weight when going to larger sized pieces of glass has implications for the robotic handlers used to move the glass into and through process stations. In addition, elastic sag, which is dependent upon glass density and Young's modulus, becomes an issue with larger sheet sizes, impacting the ability to load, retrieve, and space the glass in the cassettes used to transport the glass between process stations. Accordingly, it is desirable to identify compositions that reduce issues associated with sag in addition to minimizing compaction, stress relaxation and elastic strain, while maintaining the defect-limiting attributes described above.
SUMMARY
[0010] Some embodiments pertain to compounds, compositions, articles, devices, and methods for the manufacture of alkali-free, boroalumino silicate glasses exhibiting desirable physical and chemical properties. In one or more embodiments, the glasses are suitable for use as substrates in flat panel display devices, such as, active matrix liquid crystal displays (AMLCDs) and active matrix organic light emitting diode displays (AMOLEDs). In accordance with one or more embodiments, glasses are provided that possess densities less than 2.55 g/cm.sup.3 and good dimensional stability (i.e., low compaction). Additionally, one or more embodiments of the disclosed compositions have strain points in excess of 680.degree. C. which, when subjected to the thermal history of the fusion process, have acceptable thermal stability for a-Si thin film transistor processes.
[0011] Also described herein are glasses that are substantially free of alkalis that possess high annealing points and high Young's modulus, thus, good dimensional stability (i.e., low compaction, elastic strain and stress relaxation) for use as TFT backplane substrates in amorphous silicon and oxide TFT processes. A high annealing point glass can prevent panel distortion due to compaction/shrinkage or stress relaxation during thermal processing subsequent to manufacturing of the glass. Embodiments of the inventive glasses also possess a high elastic modulus and relatively low density, thereby increasing the specific modulus of the glass and greatly reducing the risk of elastic sag in the glass sheet. Additionally, the inventive glasses possess unusually high liquidus viscosity, and thus a significantly reduced risk to devitrification at cold places in the forming apparatus. It is to be understood that while low alkali concentrations are generally desirable, in practice it may be difficult or impossible to economically manufacture glasses that are entirely free of alkalis. The alkalis in question arise as contaminants in raw materials, as minor components in refractories, etc., and can be very difficult to eliminate entirely. Therefore, the inventive glasses are considered substantially free of alkalis if the total concentration of the alkali elements Li.sub.2O, Na.sub.2O, and K.sub.2O is less than about 0.1 mole percent (mol %).
[0012] In some embodiments, a glass is provided comprising in mole percent on an oxide basis in the ranges: SiO.sub.2 65-72, Al.sub.2O.sub.3 10-14, B.sub.2O.sub.3 3-8, MgO 2-6, CaO 3-9, SrO 0-6, and BaO 0-<1, wherein the glass exhibits a strain point >650.degree. C., an annealing point greater than or equal to about 730.degree. C., a Young's modulus >78 GPa, T.sub.35kP-T.sub.liq>30.degree. C., a density <about 2.55 g/cm.sup.3, a CTE less than about 39.times.10.sup.-7/.degree. C.
[0013] In further embodiments, a glass is provided comprising, in mole percent on an oxide basis in the ranges: SiO.sub.2 67-70, Al.sub.2O.sub.3 11-13, B.sub.2O.sub.3 4-6, MgO 3-5.5, CaO 5.5-7, SrO 3-5, and BaO 0-<1, wherein the glass exhibits a strain point >700.degree. C., an annealing point greater than or equal to about 750.degree. C., a Young's modulus >79 GPa, T.sub.35kP-T.sub.liq>65.degree. C., a density <about 2.55 g/cm.sup.3, a CTE less than about 36.times.10.sup.-7/.degree. C.
[0014] In other embodiments, a glass is provided comprising in mole percent on an oxide basis in the ranges: SiO.sub.2>60, Al.sub.2O.sub.3>10, B.sub.2O.sub.3>0.1, (MgO+CaO+SrO+BaO)/Al.sub.2O.sub.3>1.0, wherein the glass exhibits an annealing point greater than or equal to about 730.degree. C., a strain point greater than or equal to about 650.degree. C., a softening point greater than or equal to about 950.degree. C., a T.sub.200P less than about 1650.degree. C., a T.sub.35kP less than about 1300.degree. C., a T.sub.35kP-T.sub.liq>30.degree. C., a specific modulus greater than 31, and a Young's Modulus greater than or equal to 78 GPa.
[0015] Additional features and advantages of the disclosure will be set forth in the detailed description which follows, and in part will be readily apparent to those skilled in the art from that description or recognized by practicing the methods as described herein, including the detailed description which follows, the claims, as well as the appended drawings.
[0016] It is to be understood that both the foregoing general description and the following detailed description present various embodiments of the disclosure, and are intended to provide an overview or framework for understanding the nature and character of the claims. The accompanying drawings are included to provide a further understanding of the disclosure, and are incorporated into and constitute a part of this specification. The drawings illustrate various embodiments of the disclosure and together with the description serve to explain the principles and operations of the disclosure.
BRIEF DESCRIPTION OF THE DRAWINGS
[0017] The following detailed description can be further understood when read in conjunction with the following drawings.
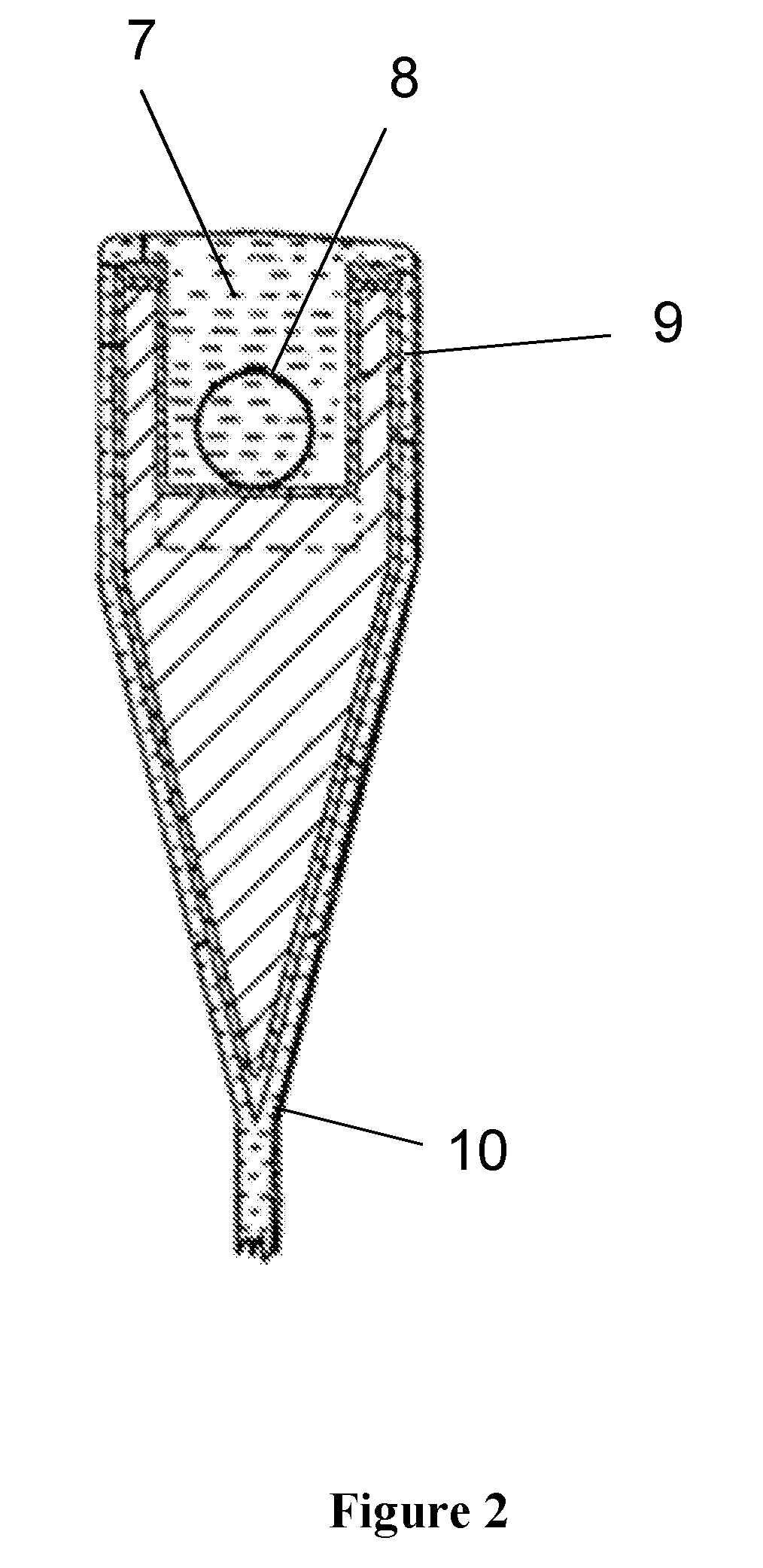
[0018] FIG. 1 is a schematic representation of an isopipe, the forming mandrel used to make precision sheet in the fusion draw process; and
[0019] FIG. 2 is a cross section of the isopipe of FIG. 1 at section line 6.
DETAILED DESCRIPTION
[0020] Described herein are glasses that are substantially free of alkalis that possess densities less than 2.55 g/cm.sup.3 and good dimensional stability (i.e., low compaction, reduced stress relaxation and elastic strain). Additionally, one or more embodiments of the disclosed compositions have strain points in excess of 680.degree. C. which, when subjected to the thermal history of the fusion process, have acceptable thermal stability for a-Si thin film transistor processes. Embodiments of the present subject matter also provide glasses with high annealing points. A high annealing point glass can prevent panel distortion due to compaction/shrinkage and stress relaxation during thermal processing subsequent to manufacturing of the glass. It is to be understood that while low alkali concentrations are generally desirable, in practice it may be difficult or impossible to economically manufacture glasses that are entirely free of alkalis. The alkalis in question arise as contaminants in raw materials, as minor components in refractories, etc., and can be very difficult to eliminate entirely. Therefore, the disclosed glasses are considered substantially free of alkalis if the total concentration of the alkali elements Li.sub.2O, Na.sub.2O, and K.sub.2O is less than about 0.1 weight percent (mol %).
[0021] In one or more embodiments, the substantially alkali-free glasses have annealing points greater than about 730.degree. C., greater than 735.degree. C., greater than 740.degree. C., or greater than 750.degree. C. In further embodiments, the substantially alkali-free glasses have strain points greater than about 650.degree. C., greater than 680.degree. C., greater than 690.degree. C., or greater than 700 OC. In yet additional embodiments, the substantially alkali-free glasses have softening points greater than about 950.degree. C., greater than 970.degree. C., greater than 980.degree. C., or greater than 990.degree. C. According to one or more embodiments, the temperature of the disclosed glasses at a viscosity of about 200 poise (T.sub.200P) is less than about 1650.degree. C., less than about 1625.degree. C., or less than about 1610.degree. C. According to one or more embodiments, the temperature of the disclosed glasses at a viscosity of about 35,000 poise (T.sub.35kP) is less than about 1300.degree. C., less than about 1260.degree. C., or less than about 1250.degree. C. The liquidus temperature of a glass (T.sub.liq) is the highest temperature above which no crystalline phases can coexist in equilibrium with the glass. According to one or more embodiments, T.sub.35k-T.sub.liq>30.degree. C., T.sub.35kP-T.sub.liq>40.degree. C., T.sub.35kP-T.sub.liq>50.degree. C., T.sub.35kP-T.sub.liq>60.degree. C., T.sub.35kP-T.sub.liq>65.degree. C., or T.sub.35kP-T.sub.liq>70.degree. C. The float process delivers glass at a viscosity between 3000 and 10,000 poise. If the glass devitrifies anywhere near the temperature corresponding to this viscosity, then devitrification products would show up in the finished product. In one or more embodiments, the glasses exhibit a specific modulus greater than 31, greater than 31.5, or greater than 32. The specific modulus of the glass, defined as E/.rho., (elastic or Young's modulus in GPa divided by density in g/cm.sup.3) is indicative of the magnitude of elastic sag which a glass sheet will experience during processing. For this reason glass compositions with a specific modulus of 31 or greater are desirable. It will be understood that the disclosed glasses can exhibit one or more of the disclosed properties provided above. Thus, the disclosed glasses may exhibit one of the above disclosed properties, two of the above disclosed properties, three of the disclosed properties, four of the disclosed properties, five of the disclosed properties, six of the disclosed properties and/or seven of the disclosed properties, in any combination of the disclosed properties.
[0022] In some embodiments, exemplary substantially alkali-free glass comprises in mole percent on an oxide basis in the following ranges: [0023] SiO.sub.2 65-72 [0024] Al.sub.2O.sub.3 10-14 [0025] B.sub.2O.sub.3 3-8 [0026] MgO 2-6 [0027] CaO 3-9 [0028] SrO 0-6 [0029] BaO 0-<1.
[0030] In other embodiments, exemplary substantially alkali-free glass comprises in mole percent on an oxide basis in the following ranges: [0031] SiO.sub.2 67-70 [0032] Al.sub.2O.sub.311-13 [0033] B.sub.2O.sub.3 4-6 [0034] MgO 3-5.5 [0035] CaO 5.5-7 [0036] SrO 3-5 [0037] BaO 0-<1.
[0038] Any of the aforementioned embodiments can include one or more of the following properties: (MgO+CaO+SrO+BaO)/Al.sub.2O.sub.3.gtoreq.1.05, .gtoreq.1.08, .gtoreq.1.10, an annealing point greater than or equal to about 730.degree. C., 735.degree. C., 740.degree. C., or 750.degree. C., a strain point greater than or equal to about 650.degree. C., 680.degree. C., 690.degree. C., or 700.degree. C., and/or a softening point greater than or equal to about 950.degree. C., 970.degree. C., 980.degree. C., or 990.degree. C. Any of these embodiments may also include one or more of the following properties: a T.sub.200P less than about 1650.degree. C., 1625.degree. C., or 1610.degree. C., a T.sub.35kP less than about 1300.degree. C., 1260.degree. C., or 1250.degree. C., a T.sub.35kP-T.sub.liq>40.degree. C., >30.degree. C., >50.degree. C., >60.degree. C., >65.degree. C., or >70.degree. C., and/or a specific modulus greater than 31, 31.5, or 32. Any of these embodiments may further include one or more of the following properties: a density less than or equal to 2.55 g/cm.sup.3, a CTE less than about 39.times.10.sup.-7/.degree. C., less than about 38.times.10.sup.-7/.degree. C., or less than about 36.times.10.sup.-7/.degree. C., a Young's Modulus greater than or equal to 78 GPa, 79 GPa, 80 GPa, or 81 GPa.
[0039] In some embodiments, the disclosed glass includes a chemical fining agent. Such fining agents include, but are not limited to, SnO.sub.2, As.sub.2O.sub.3, Sb.sub.2O.sub.3, F, Cl and Br, and in which the concentrations of the chemical fining agents are kept at a level of 0.5 mol % or less. Chemical fining agents may also include CeO.sub.2, Fe.sub.2O.sub.3, and other oxides of transition metals, such as MnO.sub.2. These oxides may introduce color to the glass via visible absorptions in their final valence state(s) in the glass, and thus their concentration is specifically kept at a level of 0.2 mol % or less.
[0040] In some embodiments, the disclosed glasses are manufactured into sheets via the fusion process. The fusion draw process results in a pristine, fire-polished glass surface that reduces surface-mediated distortion to high resolution TFT backplanes and color filters. FIG. 1 is a schematic drawing of the fusion draw process at the position of the forming mandrel, or isopipe, so called because its gradient trough design produces the same (hence "iso") flow at all points along the length of the isopipe (from left to right). FIG. 2 is a schematic cross-section of the isopipe near position 6 in FIG. 1. Glass is introduced from the inlet 1, flows along the bottom of the trough 2 formed by the weir walls 3 to the compression end 4. Glass overflows the weir walls 4 on either side of the isopipe (see FIG. 2), and the two streams of glass join or fuse at the root 6. Edge directors 7 at either end of the isopipe serve to cool the glass and create a thicker strip at the edge called a bead. The bead is pulled down by pulling rolls, hence enabling sheet formation at high viscosity. By adjusting the rate at which sheet is pulled off the isopipe, it is possible to use the fusion draw process to produce a very wide range of thicknesses at a fixed melting rate.
[0041] The downdraw sheet drawing processes and, in particular, the fusion process described in U.S. Pat. Nos. 3,338,696 and 3,682,609 (both to Dockerty), which are incorporated by reference, can be used herein. Compared to other forming processes, such as the float process, the fusion process is preferred for several reasons. First, glass substrates made from the fusion process do not require polishing. Current glass substrate polishing is capable of producing glass substrates having an average surface roughness greater than about 0.5 nm (Ra), as measured by atomic force microscopy. The glass substrates produced by the fusion process have an average surface roughness as measured by atomic force microscopy of less than 0.5 nm. The substrates also have an average internal stress as measured by optical retardation which is less than or equal to 150 psi.
[0042] In some embodiments, the disclosed glasses are manufactured into sheet form using the fusion process. While the disclosed glasses are compatible with the fusion process, they may also be manufactured into sheets or other ware through less demanding manufacturing processes. Such processes include but are not limited to slot draw, float, rolling, and other sheet-forming processes known to those skilled in the art.
[0043] Relative to these alternative methods for creating sheets of glass, the fusion process as discussed above is capable of creating very thin, very flat, very uniform sheets with a pristine surface. Slot draw also can result in a pristine surface, but due to change in orifice shape over time, accumulation of volatile debris at the orifice-glass interface, and the challenge of creating an orifice to deliver truly flat glass, the dimensional uniformity and surface quality of slot-drawn glass are generally inferior to fusion-drawn glass. The float process is capable of delivering very large, uniform sheets, but the surface is substantially compromised by contact with the float bath on one side, and by exposure to condensation products from the float bath on the other side. This means that float glass must be polished for use in high performance display applications.
[0044] Unlike the float process, the fusion process results in rapid cooling of the glass from high temperature. This rapid cooling results in a high fictive temperature T.sub.f. The fictive temperature can be thought of as representing the discrepancy between the structural state of the glass and the state it would assume if fully relaxed at the temperature of interest. We consider now the consequences of reheating a glass with a glass transition temperature T.sub.g to a process temperature T.sub.p such that T.sub.p<T.sub.g.ltoreq.T.sub.f. Since T.sub.p<T.sub.f, the structural state of the glass is out of equilibrium at T.sub.p, and the glass will spontaneously relax toward a structural state that is in equilibrium at T.sub.p. The rate of this relaxation scales inversely with the effective viscosity of the glass at T.sub.p, such that high viscosity results in a slow rate of relaxation, and a low viscosity results in a fast rate of relaxation. The effective viscosity varies inversely with the fictive temperature of the glass, such that a low fictive temperature results in a high viscosity, and a high fictive temperature results in a comparatively low viscosity. Therefore, the rate of relaxation at T.sub.p scales directly with the fictive temperature of the glass. A process that introduces a high fictive temperature results in a comparatively high rate of relaxation when the glass is reheated at T.sub.p.
[0045] One means to reduce the rate of relaxation at T.sub.p is to increase the viscosity of the glass at that temperature. The annealing point of a glass represents the temperature at which the glass has a viscosity of 10.sup.13.2 poise. As temperature decreases below the annealing point, the viscosity of the supercooled melt increases. At a fixed temperature below T.sub.g, a glass with a higher annealing point has a higher viscosity than a glass with a lower annealing point. Therefore, to increase the viscosity of a substrate glass at T.sub.p, one might choose to increase its annealing point. Unfortunately, it is generally the case that the composition changes necessary to increase the annealing point also increase viscosity at all other temperatures. In particular, the fictive temperature of a glass made by the fusion process corresponds to a viscosity of about 10.sup.11-10.sup.12 poise, so an increase in annealing point for a fusion-compatible glass generally increases its fictive temperature as well. For a given glass, higher fictive temperature results in lower viscosity at temperature below T.sub.g, and thus increasing fictive temperature works against the viscosity increase that would otherwise be obtained by increasing the annealing point. To see a substantial change in the rate of relaxation at T.sub.p, it is generally necessary to make relatively large changes in annealing point. An aspect of the disclosed glass is that it has an annealing point greater than or equal to about 730.degree. C., 735.degree. C., 740.degree. C., or 750.degree. C. Without being bound by any particular theory of operation, it is believed that such high annealing points result in acceptably low rates of thermal relaxation during low-temperature TFT processing, e.g., typical low-temperature polysilicon rapid thermal anneal cycles.
[0046] In addition to its impact on fictive temperature, increasing annealing point also increases temperatures throughout the melting and forming system, particularly the temperatures on the isopipe. For example, Eagle XG.RTM. glass and Lotus.TM. glass (Corning Incorporated, Corning, N.Y.) have annealing points that differ by about 50.degree. C., and the temperature at which they are delivered to the isopipe also differ by about 50.degree. C. When held for extended periods of time above about 1310.degree. C., zircon refractory forming the isopipe shows thermal creep, which can be accelerated by the weight of the isopipe itself plus the weight of the glass on the isopipe. A second aspect of exemplary glasses is that their delivery temperatures are less than or equal to about 1350.degree. C., or 1345.degree. C., or 1340.degree. C., or 1335.degree. C., or 1330.degree. C., or 1325.degree. C., or 1320.degree. C., or 1315.degree. C. or 1310.degree. C. Such delivery temperatures may permit extended manufacturing campaigns without a need to replace the isopipe or extend the time between isopipe replacements.
[0047] In manufacturing trials of glasses with high annealing points and delivery temperatures below 1350.degree. C. and below 1310.degree. C., it has been found that the glass showed a greater tendency toward devitrification on the root of the isopipe and--especially--the edge directors relative to glasses with lower annealing points. Careful measurement of the temperature profile on the isoipe showed that the edge director temperatures were much lower relative to the center root temperature than had been anticipated and is believed to be due to radiative heat loss. The edge directors typically are maintained at a temperature below the center root temperature to ensure that the glass is viscous enough as it leaves the root to put the sheet in between the edge directors under tension, thus maintaining a flat shape. As edge directors are located at either end of the isopipe, the edge directors are difficult to heat, and thus the temperature difference between the center of the root and the edge directors may differ by 50.degree. C. or more.
[0048] While not wishing to be held to theory, it is believed that the increased tendency toward devitirication in the fusion process can be understood in terms of the radiative heat loss of glass as a function of temperature. Fusion is substantially an isothermal process, so glass exits the inlet at a particular viscosity and exits the root at a much higher viscosity, but the actual values for the viscosity are not strongly dependent on the identity of the glass or the temperature of the process. Thus, a glass with a higher annealing point generally requires much higher isopipe temperatures than a glass with a lower annealing point just to match the delivery and exit viscosities. Again, without being bound by any particular theory of operation, it is believed that since radiative heat loss increases with temperature, and since high annealing point glasses generally are formed at higher temperatures than lower annealing point glasses, the temperature difference between the center root and the edge director generally increases with the annealing point of the glass. This may have a direct relationship to the tendency of a glass to form devitrification products on the isopipe or edge directors.
[0049] The liquidus temperature of a glass is defined as the highest temperature at which a crystalline phase would appear if a glass were held indefinitely at that temperature. The liquidus viscosity is the viscosity of a glass at the liquidus temperature. To completely avoid devitrification on an isopipe, it may be helpful for the liquidus viscosity to be high enough to ensure that glass is no longer on the isopipe refractory or edge director material at or near the liquidus temperature.
[0050] In practice, few alkali-free glasses have liquidus viscosities of the desired magnitude. Experience with substrate glasses suitable for amorphous silicon applications (e.g., Eagle XG.RTM. glass) indicated that edge directors could be held continuously at temperatures up to 60.degree. C. below the liquidus temperature of certain alkali-free glasses. While it was understood that glasses with higher annealing points would require higher forming temperatures, it was not anticipated that the edge directors would be so much cooler relative to the center root temperature. A useful metric for keeping track of this effect is the difference between the delivery temperature onto the isopipe and the liquidus temperature of the glass, T.sub.liq. In the fusion process, it is generally desirable to deliver glass at about 35,000 poise (T.sub.35kP). For a particular delivery temperature, it may be useful to make T.sub.35kP-T.sub.liq as large possible, but for an amorphous silicon substrate such as Eagle XG.RTM. glass, it is found that extended manufacturing campaigns can be conducted if T.sub.35kP-T.sub.liq is about 80.degree. C. or more. As temperature increases, T.sub.35kP-T.sub.liq must increase as well, such that for T.sub.35k near 1300.degree. C., it may be helpful to have T.sub.35kP-T.sub.liq equal to or greater than about 100.degree. C. The minimum useful value for T.sub.35kP-T.sub.liq varies approximately linearly with temperature from about 1200.degree. C. to about 1320.degree. C., and can be expressed according the relationship below:
Minimum T.sub.35kP-T.sub.liq=0.25T.sub.35k-225 (1)
where all temperatures are in .degree. C. Thus, one or more embodiments of exemplary glasses has a T.sub.35kP-T.sub.liq>30.degree. C., T.sub.35kP-T.sub.liq>40.degree. C., T.sub.35kP-T.sub.liq>50.degree. C., T.sub.35kP-T.sub.liq>60.degree. C., T.sub.35kP-T.sub.liq>65.degree. C., or T.sub.35kP-T.sub.liq>70.degree. C.
[0051] In addition, the forming process may require glass with a high liquidus viscosity. This is necessary so as to avoid devitrification products at interfaces with glass and to minimize visible devitrification products in the final glass. Thus, for a given glass compatible with fusion for a particular sheet size and thickness, adjusting the process so as to manufacture wider sheet or thicker sheet generally results in lower temperatures at either end of the isopipe. Some embodiments have higher liquidus viscosities to provide greater flexibility for manufacturing via the fusion process. In some embodiments, the liquidus viscosity is greater than or equal to about 150 kP.
[0052] In tests of the relationship between liquidus viscosity and subsequent devitrification tendencies in the fusion process, the inventors have surprisingly found that high delivery temperatures, such as those of exemplary glasses, generally require higher liquidus viscosities for long-term production than would be the case for typical AMLCD substrate compositions with lower annealing points. While not wishing to be bound by theory, it is believed that this arises from accelerated rates of crystal growth as temperature increases. Fusion is essentially an isoviscous process, so a more viscous glass at some fixed temperature may be formed by fusion at higher temperature than a less viscous glass. While some degree of undercooling (cooling below the liquidus temperature) can be sustained for extended periods in a glass at lower temperature, crystal growth rates increase with temperature, and thus more viscous glasses grow an equivalent, unacceptable amount of devitrification products in a shorter period of time than less viscous glasses. Depending on where formed, devitrification products can compromise forming stability and introduce visible defects into the final glass.
[0053] To be formed by the fusion process, one or more embodiments of the glass compositions have a liquidus viscosity greater than or equal to about 150,000 poises, or 175,000 poises, or 200,000 poises. A surprising result is that throughout the range of exemplary glasses, it is possible to obtain a liquidus temperature low enough, and a viscosity high enough, such that the liquidus viscosity of the glass is unusually high compared to other compositions.
[0054] In the glass compositions described herein, SiO.sub.2 serves as the basic glass former. In certain embodiments, the concentration of SiO.sub.2 can be greater than 60 mole percent to provide the glass with a density and chemical durability suitable for a flat panel display glass (e.g., an AMLCD or AMOLED glass), and a liquidus temperature (liquidus viscosity), which allows the glass to be formed by a downdraw process (e.g., a fusion process). In terms of an upper limit, in general, the SiO.sub.2 concentration can be less than or equal to about 80 mole percent to allow batch materials to be melted using conventional, high volume, melting techniques, e.g., Joule melting in a refractory melter. As the concentration of SiO.sub.2 increases, the 200 poise temperature (melting temperature) generally rises. In various applications, the SiO.sub.2 concentration is adjusted so that the glass composition has a melting temperature less than or equal to 1,750.degree. C. In some embodiments, the SiO.sub.2 concentration is in the range of about 60.0 mol % to about 80.0 mol %, or in the range of about 62.0 mol % to about 78.0 mol %, or in the range of about 65.0 mol % to about 72 mol % or in the range of about 67 mol % and 70 mol %.
[0055] Al.sub.2O.sub.3 is another glass former used to make the glasses described herein. An Al.sub.2O.sub.3 concentration greater than or equal to 10 mole percent provides the glass with a low liquidus temperature and high viscosity, resulting in a high liquidus viscosity. The use of at least 10 mole percent Al.sub.2O.sub.3 also improves the glass's annealing point and modulus. In order that the ratio (MgO+CaO+SrO+BaO)/Al.sub.2O.sub.3 is greater than or equal to 1.0, the Al.sub.2O.sub.3 concentration may be below about 15 mole percent. In some embodiments, the Al.sub.2O.sub.3 concentration is in the range of about 10.0 to 14.0 mole percent, or in the range of about 11.0 to about 13.0 mol % while maintaining a ratio of (MgO+CaO+SrO+BaO)/Al.sub.2O.sub.3 greater than or equal to about 1.0.
[0056] Some embodiments of the disclosure have a modulus greater than about 79 GPa, or 80 GPa, or 80.5 GPa, or 81 GPa, or 81.5 GPa, or 82 GPa, or 82.5 GPa, or 83 GPa, or 83.5 GPa, or 84 GPa, or 84.5 GPa or 85 GPa. In various embodiments, an aluminosilicate glass article has a Young's modulus in the range of about 81 GPa to about 88 GPa, or in the range of about 81.5 GPa to about 85 GPa, or in the range of about 82 GPa to about 84.5 GPa.
[0057] The density of some embodiments of aluminosilicate glass articles is less than about 2.7 g/cc, or 2.65 g/cc, or 2.61 g/cc, or 2.6 g/cc, or 2.55 g/cc. In various embodiments, the density is in the range of about 2.49 g/cc to about 2.53 g/cc, or in the range of about 2.50 g/cc to about 2.60 g/cc.
[0058] B.sub.2O.sub.3 is both a glass former and a flux that aids melting and lowers the melting temperature. It has an impact on both liquidus temperature and viscosity. Increasing B.sub.2O.sub.3 can be used to increase the liquidus viscosity of a glass. To achieve these effects, the glass compositions of one or more embodiments may have B.sub.2O.sub.3 concentrations that are equal to or greater than 0.1 mole percent. As discussed above with regard to SiO.sub.2, glass durability is very important for flat panel display applications. Durability can be controlled somewhat by elevated concentrations of alkaline earth oxides, and significantly reduced by elevated B.sub.2O.sub.3 content. Annealing point decreases as B.sub.2O.sub.3 increases, so it may be helpful to keep B.sub.2O.sub.3 content low relative to its typical concentration in amorphous silicon substrates. Thus in some embodiments, the glass composition has B.sub.2O.sub.3 concentrations that are in the range of about 3 to about 8.0 mole percent, or greater than 0 to about 10.0 mol %, or about 2.0 to about 9.0 mol %, or greater than 0 to about 9.0 mol %, or about 3 to about 7 mol %, or in the range of about 4.0 to about 6.0 mol %.
[0059] The Al.sub.2O.sub.3 and B.sub.2O.sub.3 concentrations can be selected as a pair to increase annealing point, increase modulus, improve durability, reduce density, and reduce the coefficient of thermal expansion (CTE), while maintaining the melting and forming properties of the glass. For example, an increase in B.sub.2O.sub.3 and a corresponding decrease in Al.sub.2O.sub.3 can be helpful in obtaining a lower density and CTE, while an increase in Al.sub.2O.sub.3 and a corresponding decrease in B.sub.2O.sub.3 can be helpful in increasing annealing point, modulus, and durability, provided that the increase in Al.sub.2O.sub.3 does not reduce the (MgO+CaO+SrO+BaO)/Al.sub.2O.sub.3 ratio below about 1.0. For (MgO+CaO+SrO+BaO)/Al.sub.2O.sub.3 ratios below about 1.0, it may be difficult or impossible to remove gaseous inclusions from the glass due to late-stage melting of the silica raw material. Furthermore, when (MgO+CaO+SrO+BaO)/Al.sub.2O.sub.3.ltoreq.1.05, mullite, an aluminosilicate crystal, can appear as a liquidus phase. Once mullite is present as a liquidus phase, the composition sensitivity of liquidus increases considerably, and mullite devitrification products both grow very quickly and are very difficult to remove once established. Thus, in some embodiments, the glass composition has (MgO+CaO+SrO+BaO)/Al.sub.2O.sub.3.gtoreq.1.0 (or greater than or equal to about 1.0). In various embodiments, the glass has (MgO+CaO+SrO+BaO)/Al.sub.2O.sub.3.gtoreq.1.05 (or greater than or equal to about 1.05), or in the range of about 1 to about 1.25.
[0060] In one or more embodiments, glasses for use in flat panel display applications (such as AMLCD and AMOLED displays) have coefficients of thermal expansion (CTEs) (22-300.degree. C.) in the range of about 28.times.10.sup.-7/.degree. C. to about 42.times.10.sup.-7/.degree. C., or in the range of about 30.times.10.sup.-7/.degree. C. to about 40.times.10.sup.-7/.degree. C., or in the range of about 32.times.10.sup.-7/.degree. C. to about 38.times.10.sup.-7/.degree. C.
[0061] In addition to the glass formers (SiO.sub.2, Al.sub.2O.sub.3, and B.sub.2O.sub.3), the glasses described herein also include alkaline earth oxides. In one embodiment, at least three alkaline earth oxides are part of the glass composition, e.g., MgO, CaO, and BaO, and, optionally, SrO. The alkaline earth oxides provide the glass with various properties important to melting, fining, forming, and ultimate use. Accordingly, to improve glass performance in these regards, in one embodiment, the (MgO+CaO+SrO+BaO)/Al.sub.2O.sub.3 ratio is greater than or equal to about 1.0. As this ratio increases, viscosity tends to increase more strongly than liquidus temperature, and thus it is increasingly difficult to obtain suitably high values for T.sub.35k-T.sub.liq. Thus in another embodiment, ratio (MgO+CaO+SrO+BaO)/Al.sub.2O.sub.3 is less than or equal to about 2. In some embodiments, the (MgO+CaO+SrO+BaO)/Al.sub.2O.sub.3 ratio is in the range of about 1 to about 1.25, or in the range of about 1 to about 1.18, or in the range of about 1.1 to about 1.8.
[0062] For certain embodiments of this disclosure, the alkaline earth oxides may be treated as what is in effect a single compositional component. This is because their impact upon viscoelastic properties, liquidus temperatures and liquidus phase relationships are qualitatively more similar to one another than they are to the glass forming oxides SiO.sub.2, Al.sub.2O.sub.3 and B.sub.2O.sub.3. However, the alkaline earth oxides CaO, SrO and BaO can form feldspar minerals, notably anorthite (CaAl.sub.2Si.sub.2O.sub.8) and celsian (BaAl.sub.2Si.sub.2O.sub.8) and strontium-bearing solid solutions of same, but MgO does not participate in these crystals to a significant degree. Therefore, when a feldspar crystal is already the liquidus phase, a superaddition of MgO may serves to stabilize the liquid relative to the crystal and thus lower the liquidus temperature. At the same time, the viscosity curve typically becomes steeper, reducing melting temperatures while having little or no impact on low-temperature viscosities.
[0063] The inventors have found that the addition of small amounts of MgO may benefit melting by reducing melting temperatures, forming by reducing liquidus temperatures and increasing liquidus viscosity, while preserving high annealing point and, thus, low compaction. In various embodiments, the glass composition comprises MgO in an amount in the range of about 0.1 mol % to about 6 mol %, or in the range of about 1.0 mol % to about 6.0 mol %, or in the range of about 2.0 mol % to about 6.0 mol %, or in the range of about 3.0 mol % to about 5.5 mol %.
[0064] The inventors have surprisingly found that glasses with suitably high values of T.sub.35kP-T.sub.liq, the ratio of MgO to the other alkaline earths, MgO/(MgO+CaO+SrO+BaO), falls within a relatively narrow range. As noted above, additions of MgO can destabilize feldspar minerals, and thus stabilize the liquid and lower liquidus temperature. However, once MgO reaches a certain level, mullite, Al.sub.6Si.sub.2O.sub.13, may be stabilized, thus increasing the liquidus temperature and reducing the liquidus viscosity. Moreover, higher concentrations of MgO tend to decrease the viscosity of the liquid, and thus even if the liquidus viscosity remains unchanged by addition of MgO, it will eventually be the case that the liquidus viscosity will decrease. Thus in another embodiment, 0.20.ltoreq.MgO/(MgO+CaO+SrO+BaO).ltoreq.0.40 or in some embodiments, 0.22.ltoreq.MgO/(MgO+CaO+SrO+BaO).ltoreq.0.37. Within these ranges, MgO may be varied relative to the glass formers and the other alkaline earth oxides to maximize the value of T.sub.35k-T.sub.liq consistent with obtaining other desired properties.
[0065] Without being bound by any particular theory of operation, it is believed that calcium oxide present in the glass composition can produce low liquidus temperatures (high liquidus viscosities), high annealing points and moduli, and CTE's in the most desired ranges for flat panel applications, specifically, AMLCD or AMOLED applications. It also contributes favorably to chemical durability, and compared to other alkaline earth oxides, it is relatively inexpensive as a batch material. However, at high concentrations, CaO increases the density and CTE. Furthermore, at sufficiently low SiO.sub.2 concentrations, CaO may stabilize anorthite, thus decreasing liquidus viscosity. Accordingly, in one or more embodiment, the CaO concentration can be greater than or equal to 2 mole percent. In various embodiments, the CaO concentration of the glass composition is in the range of about 2.0 mol % to about 10.0 mol %, or in the range of about 3 mol % to about 9.0 mol %, or in the range of about 4 mol % to about 8 mol %, or in the range of about 5 mol % to about 7 mol %, or in the range of about 5.5 mol % to about 7 mol %.
[0066] SrO and BaO can both contribute to low liquidus temperatures (high liquidus viscosities) and, thus, the glasses described herein will typically contain at least both of these oxides. However, the selection and concentration of these oxides are selected to avoid an increase in CTE and density and a decrease in modulus and annealing point. The relative proportions of SrO and BaO can be balanced so as to obtain a suitable combination of physical properties and liquidus viscosity such that the glass can be formed by a downdraw process. In various embodiments, the glass comprises SrO in the range of about 0 to about 6.0 mol %, or greater than 0 to about 6.0 mol %, or about 0 to about 5.5 mol %, or in the range of about 3 mol % to about 5 mol %. In one or more embodiments, the glass comprises BaO in the range of about 0 to less than about 1.0 mol %, or greater than 0 to about 0.5 mol %, or about 0 to about 0.09 mol %, or about 0 mol % to about 0.03 mol %.
[0067] To summarize the effects/roles of the central components of the glasses of the disclosure, SiO.sub.2 is the basic glass former. Al.sub.2O.sub.3 and B.sub.2O.sub.3 are also glass formers and can be selected as a pair with, for example, an increase in B.sub.2O.sub.3 and a corresponding decrease in Al.sub.2O.sub.3 being used to obtain a lower density and CTE, while an increase in Al.sub.2O.sub.3 and a corresponding decrease in B.sub.2O.sub.3 being used in increasing annealing point, modulus, and durability, provided that the increase in Al.sub.2O.sub.3 does not reduce the RO/Al.sub.2O.sub.3 ratio below about 1.0, where RO=(MgO+CaO+SrO+BaO). If the ratio goes too low, meltability is compromised, i.e., the melting temperature becomes too high. B.sub.2O.sub.3 can be used to bring the melting temperature down, but high levels of B.sub.2O.sub.3 compromise annealing point.
[0068] In addition to meltability and annealing point considerations, for AMLCD and AMOLED applications, the CTE of the glass should be compatible with that of silicon. To achieve such CTE values, exemplary glasses can control the RO content of the glass. For a given Al.sub.2O.sub.3 content, controlling the RO content corresponds to controlling the RO/Al.sub.2O.sub.3 ratio. In practice, glasses having suitable CTE's are produced if the RO/Al.sub.2O.sub.3 ratio is below about 1.6.
[0069] On top of these considerations, the glasses are preferably formable by a downdraw process, e.g., a fusion process, which means that the glass' liquidus viscosity needs to be relatively high. Individual alkaline earths play an important role in this regard since they can destabilize the crystalline phases that would otherwise form. BaO and SrO are particularly effective in controlling the liquidus viscosity and are included in exemplary glasses for at least this purpose. As illustrated in the examples presented below, various combinations of the alkaline earths will produce glasses having high liquidus viscosities, with the total of the alkaline earths satisfying the RO/Al.sub.2O.sub.3 ratio constraints needed to achieve low melting temperatures, high annealing points, and suitable CTE's. In some embodiments, the liquidus viscosity is greater than or equal to about 150 kP.
[0070] In addition to the above components, the glass compositions described herein can include various other oxides to adjust various physical, melting, fining, and forming attributes of the glasses. Examples of such other oxides include, but are not limited to, TiO.sub.2, MnO, Fe.sub.2O.sub.3, ZnO, Nb.sub.2O.sub.5, MoO.sub.3, Ta.sub.2O.sub.5, WO.sub.3, Y.sub.2O.sub.3, La.sub.2O.sub.3 and CeO.sub.2 as well as other rare earth oxides and phosphates. In one embodiment, the amount of each of these oxides can be less than or equal to 2.0 mole percent, and their total combined concentration can be less than or equal to 5.0 mole percent. In some embodiments, the glass composition comprises ZnO in an amount in the range of about 0 to about 1.5 mol %, or about 0 to about 1.0 mol %. The glass compositions described herein can also include various contaminants associated with batch materials and/or introduced into the glass by the melting, fining, and/or forming equipment used to produce the glass, particularly Fe.sub.2O.sub.3 and ZrO.sub.2. The glasses can also contain SnO.sub.2 either as a result of Joule melting using tin-oxide electrodes and/or through the batching of tin containing materials, e.g., SnO.sub.2, SnO, SnCO.sub.3, SnC.sub.2O.sub.2, etc.
[0071] The glass compositions are generally alkali free; however, the glasses can contain some alkali contaminants. In the case of AMLCD and AMOLED applications, it is desirable to keep the alkali levels below 0.1 mole percent to avoid having a negative impact on thin film transistor (TFT) performance through diffusion of alkali ions from the glass into the silicon of the TFT. As used herein, an "alkali-free glass" is a glass having a total alkali concentration which is less than or equal to 0.1 mole percent, where the total alkali concentration is the sum of the Na.sub.2O, K.sub.2O, and Li.sub.2O concentrations. In one embodiment, the total alkali concentration is less than or equal to 0.1 mole percent.
[0072] As discussed above, (MgO+CaO+SrO+BaO)/Al.sub.2O.sub.3 ratios greater than or equal to 1.0 improve fining, i.e., the removal of gaseous inclusions from the melted batch materials. This improvement allows for the use of more environmentally friendly fining packages. For example, on an oxide basis, the glass compositions described herein can have one or more or all of the following compositional characteristics: (i) an As.sub.2O.sub.3 concentration of at most 0.05 mole percent; (ii) an Sb.sub.2O.sub.3 concentration of at most 0.05 mole percent; (iii) a SnO.sub.2 concentration of at most 0.25 mole percent.
[0073] As.sub.2O.sub.3 is an effective high temperature fining agent for AMLCD and AMOLED glasses, and in some embodiments described herein, As.sub.2O.sub.3 is used for fining because of its superior fining properties. However, As.sub.2O.sub.3 is poisonous and requires special handling during the glass manufacturing process. Accordingly, in certain embodiments, fining is performed without the use of substantial amounts of As.sub.2O.sub.3, i.e., the finished glass has at most 0.05 mole percent As.sub.2O.sub.3. In one embodiment, no As.sub.2O.sub.3 is purposely used in the fining of the glass. In such cases, the finished glass will typically have at most 0.005 mole percent As.sub.2O.sub.3 as a result of contaminants present in the batch materials and/or the equipment used to melt the batch materials.
[0074] Although not as toxic as As.sub.2O.sub.3, Sb.sub.2O.sub.3 is also poisonous and requires special handling. In addition, Sb.sub.2O.sub.3 raises the density, raises the CTE, and lowers the annealing point in comparison to glasses that use As.sub.2O.sub.3 or SnO.sub.2 as a fining agent. Accordingly, in certain embodiments, fining is performed without the use of substantial amounts of Sb.sub.2O.sub.3, i.e., the finished glass has at most 0.05 mole percent Sb.sub.2O.sub.3. In another embodiment, no Sb.sub.2O.sub.3 is purposely used in the fining of the glass. In such cases, the finished glass will typically have at most 0.005 mole percent Sb.sub.2O.sub.3 as a result of contaminants present in the batch materials and/or the equipment used to melt the batch materials.
[0075] Compared to As.sub.2O.sub.3 and Sb.sub.2O.sub.3 fining, tin fining (i.e., SnO.sub.2 fining) is less effective, but SnO.sub.2 is a ubiquitous material that has no known hazardous properties. Also, for many years, SnO.sub.2 has been a component of AMLCD and AMOLED glasses through the use of tin oxide electrodes in the Joule melting of the batch materials for such glasses. The presence of SnO.sub.2 in AMLCD and AMOLED glasses has not resulted in any known adverse effects in the use of these glasses in the manufacture of liquid crystal displays. However, high concentrations of SnO.sub.2 are not preferred as this can result in the formation of crystalline defects in AMLCD and AMOLED glasses. In one embodiment, the concentration of SnO.sub.2 in the finished glass is less than or equal to 0.25 mole percent.
[0076] Tin fining can be used alone or in combination with other fining techniques if desired. For example, tin fining can be combined with halide fining, e.g., bromine fining. Other possible combinations include, but are not limited to, tin fining plus sulfate, sulfide, cerium oxide, mechanical bubbling, and/or vacuum fining. It is contemplated that these other fining techniques can be used alone. In certain embodiments, maintaining the (MgO+CaO+SrO+BaO)/Al.sub.2O.sub.3 ratio and individual alkaline earth concentrations within the ranges discussed above makes the fining process easier to perform and more effective.
[0077] The glasses described herein can be manufactured using various techniques known in the art. In one embodiment, the glasses are made using a downdraw process such as, for example, a fusion downdraw process. In one embodiment, described herein is a method for producing an alkali-free glass sheet by a downdraw process comprising selecting, melting, and fining batch materials so that the glass making up the sheets comprises SiO.sub.2, Al.sub.2O.sub.3, B.sub.2O.sub.3, MgO, CaO and BaO, and, on an oxide basis, comprises: (i) a (MgO+CaO+SrO+BaO)/Al.sub.2O.sub.3 ratio greater than or equal to 1.0; (ii) a MgO content greater than or equal to 3.0 mole percent; (iii) a CaO content greater than or equal to 4.0 mole percent; and (iv) a BaO content greater than or equal to 1.0 mole percent, wherein: (a) the fining is performed without the use of substantial amounts of arsenic (and, optionally, without the use of substantial amounts of antimony); and (b) a population of 50 sequential glass sheets produced by the downdraw process from the melted and fined batch materials has an average gaseous inclusion level of less than 0.10 gaseous inclusions/cubic centimeter, where each sheet in the population has a volume of at least 500 cubic centimeters.
[0078] According to one embodiment, a population of 50 sequential glass sheets produced by the downdraw process from the melted and fined batch materials has an average gaseous inclusion level of less than 0.05 gaseous inclusions/cubic centimeter, where each sheet in the population has a volume of at least 500 cubic centimeters
EXAMPLES
[0079] The following examples are set forth below to illustrate exemplary embodiments. These examples are not intended to be inclusive of all embodiments of the subject matter disclosed herein, but rather to illustrate representative methods and results. These examples are not intended to exclude equivalents and variations of the present invention which are apparent to one skilled in the art.
[0080] Efforts have been made to ensure accuracy with respect to numbers (e.g., amounts, temperature, etc.) but some errors and deviations should be accounted for. Unless indicated otherwise, temperature is in OC or is at ambient temperature, and pressure is at or near atmospheric. The compositions themselves are given in weight percent on an oxide basis and have been normalized to 100%. There are numerous variations and combinations of reaction conditions, e.g., component concentrations, temperatures, pressures and other reaction ranges and conditions that can be used to optimize the product purity and yield obtained from the described process. Only reasonable and routine experimentation will be required to optimize such process conditions.
[0081] The glass properties set forth in Table 1 were determined in accordance with techniques conventional in the glass art. Thus, the linear coefficient of thermal expansion (CTE) over the temperature range 25-300.degree. C. is expressed in terms of x 10.sup.-7/.degree. C. and the annealing point is expressed in terms of .degree. C. These were determined from fiber elongation techniques (ASTM references E228-85 and C336, respectively). The density in terms of grams/cm.sup.3 was measured via the Archimedes method (ASTM C693). The melting temperature in terms of .degree. C. (defined as the temperature at which the glass melt demonstrates a viscosity of 200 poises) was calculated employing a Fulcher equation fit to high temperature viscosity data measured via rotating cylinders viscometry (ASTM C965-81).
[0082] The liquidus temperature of the glass in terms of OC was measured using the standard gradient boat liquidus method of ASTM C829-81. This involves placing crushed glass particles in a platinum boat, placing the boat in a furnace having a region of gradient temperatures, heating the boat in an appropriate temperature region for 24 hours, and determining by means of microscopic examination the highest temperature at which crystals appear in the interior of the glass. More particularly, the glass sample is removed from the Pt boat in one piece, and examined using polarized light microscopy to identify the location and nature of crystals which have formed against the Pt and air interfaces, and in the interior of the sample. Because the gradient of the furnace is very well known, temperature vs. location can be well estimated, within 5-10.degree. C. The temperature at which crystals are observed in the internal portion of the sample is taken to represent the liquidus of the glass (for the corresponding test period). Testing is sometimes carried out at longer times (e.g. 72 hours), to observe slower growing phases. The temperature corresponding to 200 poise and the viscosity at the liquidus (in poises) were determined from fits to high viscosity data using the Vogel-Fulcher-Tammann equation,
log(.eta.)=A+B/(T-T.sub.o)
in which T is temperature and A, B and T.sub.o are fitting parameters. To determine liquidus viscosity, the liquidus temperature is used as the value for T. Young's modulus values in terms of GPa were determined using a resonant ultrasonic spectroscopy technique of the general type set forth in ASTM E1875-00e1.
[0083] As can be seen in Table 1, the exemplary glasses have density, CTE, annealing point and Young's modulus values that make the glasses suitable for display applications, such as AMLCD and AMOLED substrate applications, and more particularly for low-temperature polysilicon and oxide thin film transistor applications. Although not shown in Table 1, the glasses have durabilities in acid and base media that are similar to those obtained from commercial AMLCD and AMOLED substrates, and thus are appropriate for AMLCD and AMOLED applications. The exemplary glasses can be formed using downdraw techniques, and in particular are compatible with the fusion process, via the aforementioned criteria.
[0084] The exemplary glasses of Table 1 were prepared using a commercial sand as a silica source, milled such that 90% by weight passed through a standard U.S. 100 mesh sieve. Alumina was the alumina source, periclase was the source for MgO, limestone the source for CaO, strontium carbonate, strontium nitrate or a mix thereof was the source for SrO, barium carbonate was the source for BaO, and tin (IV) oxide was the source for SnO.sub.2. The raw materials were thoroughly mixed, loaded into a platinum vessel suspended in a furnace heated by silicon carbide glowbars, melted and stirred for several hours at temperatures in the range of 1600 and 1650.degree. C. to ensure homogeneity, and delivered through an orifice at the base of the platinum vessel. The resulting patties of glass were annealed at or near the annealing point, and then subjected to various experimental methods to determine physical, viscous and liquidus attributes.
[0085] These methods are not unique, and the glasses of Table 1 can be prepared using standard methods well-known to those skilled in the art. Such methods include a continuous melting process, such as would be performed in a continuous melting process, wherein the melter used in the continuous melting process is heated by gas, by electric power, or combinations thereof.
[0086] Raw materials appropriate for producing the disclosed glass include commercially available sands as sources for SiO.sub.2; alumina, aluminum hydroxide, hydrated forms of alumina, and various aluminosilicates, nitrates and halides as sources for Al.sub.2O.sub.3; boric acid, anhydrous boric acid and boric oxide as sources for B.sub.2O.sub.3; periclase, dolomite (also a source of CaO), magnesia, magnesium carbonate, magnesium hydroxide, and various forms of magnesium silicates, aluminosilicates, nitrates and halides as sources for MgO; limestone, aragonite, dolomite (also a source of MgO), wolastonite, and various forms of calcium silicates, aluminosilicates, nitrates and halides as sources for CaO; and oxides, carbonates, nitrates and halides of strontium and barium. If a chemical fining agent is desired, tin can be added as SnO.sub.2, as a mixed oxide with another major glass component (e.g., CaSnO.sub.3), or in oxidizing conditions as SnO, tin oxalate, tin halide, or other compounds of tin known to those skilled in the art.
[0087] The glasses in Table 1 contain SnO.sub.2 as a fining agent, but other chemical fining agents could also be employed to obtain glass of sufficient quality for TFT substrate applications. For example, the disclosed glasses could employ any one or combinations of As.sub.2O.sub.3, Sb.sub.2O.sub.3, CeO.sub.2, Fe.sub.2O.sub.3, and halides as deliberate additions to facilitate fining, and any of these could be used in conjunction with the SnO.sub.2 chemical fining agent shown in the examples. Of these, As.sub.2O.sub.3 and Sb.sub.2O.sub.3 are generally recognized as hazardous materials, subject to control in waste streams such as might be generated in the course of glass manufacture or in the processing of TFT panels. It is therefore desirable to limit the concentration of As.sub.2O.sub.3 and Sb.sub.2O.sub.3 individually or in combination to no more than 0.005 mol %.
[0088] In addition to the elements deliberately incorporated into the disclosed glasses, nearly all stable elements in the periodic table are present in glasses at some level, either through low levels of contamination in the raw materials, through high-temperature erosion of refractories and precious metals in the manufacturing process, or through deliberate introduction at low levels to fine tune the attributes of the final glass. For example, zirconium may be introduced as a contaminant via interaction with zirconium-rich refractories. As a further example, platinum and rhodium may be introduced via interactions with precious metals. As a further example, iron may be introduced as a tramp in raw materials, or deliberately added to enhance control of gaseous inclusions. As a further example, manganese may be introduced to control color or to enhance control of gaseous inclusions. As a further example, alkalis may be present as a tramp component at levels up to about 0.1 mol % for the combined concentration of Li.sub.2O, Na.sub.2O and K.sub.2O.
[0089] Hydrogen is inevitably present in the form of the hydroxyl anion, OH.sup.-, and its presence can be ascertained via standard infrared spectroscopy techniques. Dissolved hydroxyl ions significantly and nonlinearly impact the annealing point of the disclosed glasses, and thus to obtain the desired annealing point it may be necessary to adjust the concentrations of major oxide components so as to compensate. Hydroxyl ion concentration can be controlled to some extent through choice of raw materials or choice of melting system. For example, boric acid is a major source of hydroxyls, and replacing boric acid with boric oxide can be a useful means to control hydroxyl concentration in the final glass. The same reasoning applies to other potential raw materials comprising hydroxyl ions, hydrates, or compounds comprising physisorbed or chemisorbed water molecules. If burners are used in the melting process, then hydroxyl ions can also be introduced through the combustion products from combustion of natural gas and related hydrocarbons, and thus it may be desirable to shift the energy used in melting from burners to electrodes to compensate. Alternatively, one might instead employ an iterative process of adjusting major oxide components so as to compensate for the deleterious impact of dissolved hydroxyl ions.
[0090] Sulfur is often present in natural gas, and likewise is a tramp component in many carbonate, nitrate, halide, and oxide raw materials. In the form of SO.sub.2, sulfur can be a troublesome source of gaseous inclusions. The tendency to form SO.sub.2-rich defects can be managed to a significant degree by controlling sulfur levels in the raw materials, and by incorporating low levels of comparatively reduced multivalent cations into the glass matrix. While not wishing to be bound by theory, it appears that SO.sub.2-rich gaseous inclusions arise primarily through reduction of sulfate (SO.sub.4.sup.=) dissolved in the glass. The elevated barium concentrations of the disclosed glasses appear to increase sulfur retention in the glass in early stages of melting, but as noted above, barium is required to obtain low liquidus temperature, and hence high T.sub.35k-T.sub.liq and high liquidus viscosity. Deliberately controlling sulfur levels in raw materials to a low level is a useful means of reducing dissolved sulfur (presumably as sulfate) in the glass. In particular, sulfur is specifically less than 200 ppm by weight in the batch materials, and more specifically less than 100 ppm by weight in the batch materials.
[0091] Reduced multivalents can also be used to control the tendency of the disclosed glasses to form SO.sub.2 blisters. While not wishing to be bound to theory, these elements behave as potential electron donors that suppress the electromotive force for sulfate reduction. Sulfate reduction can be written in terms of a half reaction such as
SO.sub.4.sup.=.fwdarw.SO.sub.2+O.sub.2+2e-
where e- denotes an electron. The "equilibrium constant" for the half reaction is
K.sub.eq=[SO.sub.2][O.sub.2][e-].sup.2/[SO.sub.4.sup.=]
where the brackets denote chemical activities. Ideally one would like to force the reaction so as to create sulfate from SO.sub.2, O.sub.2 and 2e-. Adding nitrates, peroxides, or other oxygen-rich raw materials may help, but also may work against sulfate reduction in the early stages of melting, which may counteract the benefits of adding them in the first place. SO.sub.2 has very low solubility in most glasses, and so is impractical to add to the glass melting process. Electrons may be "added" through reduced multivalents. For example, an appropriate electron-donating half reaction for ferrous iron (Fe.sup.2+) is expressed as
2Fe.sup.2+.fwdarw.2Fe.sup.3+2e-
[0092] This "activity" of electrons can force the sulfate reduction reaction to the left, stabilizing SO.sub.4.sup.= in the glass. Suitable reduced multivalents include, but are not limited to, Fe.sup.2+, Mn.sup.2+, Sn.sup.2+, Sb.sup.3+, As.sup.3+, V.sup.3+, Ti.sup.3+, and others familiar to those skilled in the art. In each case, it may be important to minimize the concentrations of such components so as to avoid deleterious impact on color of the glass, or in the case of As and Sb, to avoid adding such components at a high enough level so as to complication of waste management in an end-user's process.
[0093] In addition to the major oxides components of the disclosed glasses, and the minor or tramp constituents noted above, halides may be present at various levels, either as contaminants introduced through the choice of raw materials, or as deliberate components used to eliminate gaseous inclusions in the glass. As a fining agent, halides may be incorporated at a level of about 0.4 mol % or less, though it is generally desirable to use lower amounts if possible to avoid corrosion of off-gas handling equipment. In a preferred embodiment, the concentration of individual halide elements are below about 200 ppm by weight for each individual halide, or below about 800 ppm by weight for the sum of all halide elements.
[0094] In addition to these major oxide components, minor and tramp components, multivalents and halide fining agents, it may be useful to incorporate low concentrations of other colorless oxide components to achieve desired physical, optical or viscoelastic properties. Such oxides include, but are not limited to, TiO.sub.2, ZrO.sub.2, HfO.sub.2, Nb.sub.2O.sub.5, Ta.sub.2O.sub.5, MoO.sub.3, WO.sub.3, ZnO, In.sub.2O.sub.3, Ga.sub.2O.sub.3, Bi.sub.2O.sub.3, GeO.sub.2, PbO, SeO.sub.3, TeO.sub.2, Y.sub.2O.sub.3, La.sub.2O.sub.3, Gd.sub.2O.sub.3, and others known to those skilled in the art. Through an iterative process of adjusting the relative proportions of the major oxide components of the disclosed glasses, such colorless oxides can be added to a level of up to about 2 mol % without unacceptable impact to annealing point, T.sub.10k-T.sub.liq or liquidus viscosity.
[0095] Examples of glass compositions according to some embodiments are disclosed below in Tables 1 and 2.
TABLE-US-00001 TABLE 1 1 2 3 4 5 6 7 weight % SiO.sub.2 60.79 60.82 60.71 60.79 61.74 62.31 61.83 Al.sub.2O.sub.3 19.36 19.33 19.26 19.36 17.94 18.59 18.06 B.sub.2O.sub.3 6.10 5.67 5.56 6.10 5.68 5.60 5.92 MgO 2.46 3.26 2.80 2.46 2.08 1.92 2.02 CaO 5.27 5.21 5.48 5.27 5.43 4.96 5.28 SrO 5.60 5.33 5.78 5.60 6.73 6.12 6.50 BaO 0.06 0.06 0.05 0.06 0.08 0.20 0.07 SnO.sub.2 0.23 0.23 0.22 0.23 0.19 0.18 0.19 Fe.sub.2O.sub.3 0.05 0.05 0.05 0.05 0.02 0.03 0.03 ZrO.sub.2 0.04 0.02 0.04 0.04 0.06 0.06 0.06 As.sub.2O.sub.3 0 0 0 0 0 0 0 Sb.sub.2O.sub.3 0 0 0 0 0 0 0 RO 17.27 13.86 14.11 13.39 14.32 13.2 13.87 mol % SiO.sub.2 67.93 67.32 67.15 67.43 68.47 69.22 68.58 Al.sub.2O.sub.3 12.51 12.52 12.56 12.62 11.72 12.17 11.8 B.sub.2O.sub.3 4.55 5.19 5.31 5.82 5.44 5.37 5.67 MgO 4.7 5.22 4.62 4.03 3.43 3.18 3.34 CaO 6.25 6.14 6.49 6.31 6.45 5.9 6.27 SrO 3.9 3.46 3.71 3.63 4.33 3.94 4.18 BaO 0.03 0.03 0.02 0.02 0.03 0.09 0.03 SnO.sub.2 0.09 0.09 0.09 0.09 0.08 0.08 0.08 Fe.sub.2O.sub.3 0.01 0.02 0.02 0.02 0.01 0.01 0.01 ZrO.sub.2 0.03 0.01 0.02 0.02 0.03 0.03 0.03 RO 14.88 14.85 14.84 13.99 14.24 13.11 13.82 Properties RO/Al.sub.2O.sub.3 1.19 1.18 1.18 1.11 1.22 1.08 1.17 Strain point 713 708 704 702 705 714 706 (.degree. C.) Anneal point 767 760 758 756 757 769 758 (.degree. C.) Softening 996 991 988 995 994 1007 995 Point (.degree. C.) CTE (10.sup.-7/.degree. C.) 35 35.1 35.7 34.1 37.7 33.5 38.2 Density 2.521 2.510 2.512 2.501 2.506 2.494 2.501 (g/cm.sup.3) Young's 81.8 81.6 81.1 79.8 79.1 79.0 78.9 mod (GPa) Specific 32.4 32.5 32.3 31.9 31.6 31.7 31.6 modulus (GPa/g/cm.sup.3) T.sub.200P (.degree. C.) 1610 1593 1596 1605 1623 1644 1627 T.sub.35kP (.degree. C.) 1243 1233 1233 1239 1245 1260 1247 Liquidus 72 h air 1175 1170 1175 scum to 1195 scum to 1185 1190 int 1200 1165 1160 1160 1160 1180 1165 Pt 1180 1150 1150 1140 1150 1170 1150 Phase An crist/an Cris + An, Cris + An, crist/an An/Crist crist/an air scum air scum (anor) to (anor) to 1240 1220 Liq 8.57E+04 1.63E+05 1.78E+05 1.94E+05 2.18E+05 2.14E+05 2.03E+05 Viscosity (P) T.sub.35kP - T.sub.liq 43 68 73 79 85 84 82
TABLE-US-00002 TABLE 2 8 9 10 11 12 13 14 weight % SiO2 (analyzed) 58.89 59.09 59.35 59.78 60.11 60.29 59.6 Al2O3 18.74 18.74 18.81 18.94 19.04 19.12 18.51 B2O3 8 7.31 6.88 6.22 5.78 5.28 5.78 MgO 3.169 3.034 3.038 3.051 3.059 3.07 3.585 CaO 5.149 5.171 5.188 5.235 5.245 5.278 6.079 SrO 6.1 6.47 6.54 6.6 6.62 6.66 5.98 BaO 0.08 0.08 0.08 0.09 0.09 0.08 0.09 SnO2 0.15 0.15 0.149 0.148 0.148 0.148 0.149 Fe2O3 0.054 0.053 0.054 0.053 0.053 0.055 0.053 ZrO2 0.013 0.009 0.008 0.008 0.008 0.007 0.007 As2O3 Sb2O3 mol % SiO2 64.75 65.33 65.62 66.05 66.38 66.74 65.59 Al2O3 12.22 12.24 12.29 12.37 12.43 12.48 11.98 B2O3 7.64 6.99 6.58 5.95 5.53 5.05 5.48 MgO 5.23 5.01 5.02 5.04 5.05 5.07 5.87 CaO 6.11 6.14 6.16 6.22 6.23 6.26 7.15 SrO 3.92 4.16 4.2 4.24 4.25 4.28 3.81 BaO 0.03 0.03 0.03 0.04 0.04 0.03 0.04 SnO2 0.07 0.07 0.07 0.07 0.07 0.07 0.07 Fe2O3 0.02 0.02 0.02 0.02 0.02 0.02 0.02 ZrO2 0.01 0 0 0 0 0 0 Properties RO/Al2O3 1.25 1.25 1.25 1.25 1.25 1.25 1.40 RO - Al2O3 3.07 3.10 3.12 3.17 3.14 3.16 4.89 RO/(Al2O3 + B2O3) 0.77 0.80 0.82 0.85 0.87 0.89 0.97 RO - Al2O3 - -4.57 -3.89 -3.46 -2.78 -2.39 -1.89 -0.59 B2O3 MgO/RO 0.34 0.33 0.33 0.33 0.33 0.32 0.35 CaO/RO 0.40 0.40 0.40 0.40 0.40 0.40 0.42 SrO/RO 0.26 0.27 0.27 0.27 0.27 0.27 0.23 (CaO + SrO)/ 0.82 0.84 0.84 0.85 0.84 0.84 0.91 Al2O3 (CaO + SrO + 0.82 0.84 0.85 0.85 0.85 0.85 0.92 BaO)/Al2O3 CaO/(CaO + SrO) 0.61 0.60 0.59 0.59 0.59 0.59 0.65 density 2.505 2.513 2.517 2.523 2.526 2.531 2.531 CTE pred 36 36.2 36.2 36.3 36.2 36.3 37.6 Anneal pred 729 736 740 747 752 757 742 Youngs mod 79.2 79.6 80.1 80.7 81.2 81.7 81.9 (pred) specific modulus 31.6 31.7 31.8 32 32.1 32.3 32.4 (pred) 200p prediciton 1555 1566 1571 1579 1584 1590 1565 35kp prediciton 1194 1204 1208 1215 1221 1226 1201 SOC pred 30.51 30.24 30.07 29.78 29.61 29.42 29.32 liquidus 1136 1152 1161 1174 1182 1192 1180 prediction strain 682 685 688 702 706 710 690 anneal 734 737 740 747 751.3 756 741 soft 963.5 965.2 969.3 975.2 980.7 983.9 965.7 CTE 36.1 39.9 37.7 36 36.5 36.6 37.3 density 2.5057 2.5138 2.5178 2.5228 2.5251 2.532 2.5322 Poisson's ratio Shear modulus (Mpsi) Young's modulus (Mpsi) Youngs mod (GPa) Specific modulus (Gpa/density) 200p 1566 1567 1576 1595 1586 1595 1566 35kp 1203 1206 1210 1225 1222 1225 1204 Liquidus 72 hr air 1125 1160 1165 1195 1175 1205 1155 internal 1120 1150 1155 1170 1170 1200 1150 Pt 1120 1120 1130 1150 1150 1180 1135 phase An An An second phase 72 h liquidus 0 120951 117209 116176 0 0 0 viscosity (int) T35k - Tliq 83 56 55 55 52 25 54 15 16 17 18 19 20 21 weight % SiO2 (analyzed) 60.12 60.58 59.87 59.8 60.38 60.8 60.2 Al2O3 18.85 19.08 19.054 19.3 19.17 19.29 19.1 B2O3 5.72 5.7 5.71 5.72 5.73 5.79 5.89 MgO 3.207 2.619 3.341 3.08 3.354 4.076 3.053 CaO 5.975 5.905 5.104 6.17 5.601 4.943 5.82 SrO 5.88 5.82 6.25 5.02 5.38 4.87 5.7 BaO 0.08 0.08 0.105 0.069 0.07 0.06 0.1 SnO2 0.149 0.149 0.152 0.16 0.149 0.151 0.151 Fe2O3 0.055 0.054 0.055 0.052 0.057 0.057 0.058 ZrO2 0.008 0.007 0.039 0.051 0.048 0.049 0.048 As2O3 Sb2O3 mol % SiO2 66.13 66.95 66.45 66.37 66.45 66.41 66.24 Al2O3 12.24 12.43 12.4 12.52 12.43 12.44 12.43 B2O3 5.44 5.44 5.44 5.43 5.44 5.47 5.61 MgO 5.27 4.32 5.5 5.05 5.5 6.65 5.02 CaO 7.05 7 6.04 7.28 6.6 5.8 6.88 SrO 3.76 3.73 4 3.2 3.43 3.09 3.65 BaO 0.03 0.03 0.05 0.03 0.03 0.03 0.04 SnO2 0.07 0.07 0.07 0.07 0.07 0.07 0.07 Fe2O3 0.02 0.02 0.02 0.02 0.02 0.02 0.02 ZrO2 0 0 0.02 0.03 0.03 0.03 0.03 Properties RO/Al2O3 1.31 1.21 1.25 1.24 1.25 1.25 1.25 RO - Al2O3 3.87 2.65 3.19 3.04 3.13 3.13 3.16 RO/(Al2O3 + B2O3) 0.91 0.84 0.87 0.87 0.87 0.87 0.86 RO - Al2O3 - -1.57 -2.79 -2.25 -2.39 -2.31 -2.34 -2.45 B2O3 MgO/RO 0.33 0.29 0.35 0.33 0.35 0.43 0.32 CaO/RO 0.44 0.47 0.39 0.47 0.42 0.37 0.44 SrO/RO 0.23 0.25 0.26 0.21 0.22 0.20 0.23 (CaO + SrO)/ 0.88 0.86 0.81 0.84 0.81 0.71 0.85 Al2O3 (CaO + SrO + 0.89 0.87 0.81 0.84 0.81 0.72 0.85 BaO)/Al2O3 CaO/(CaO + SrO) 0.65 0.65 0.60 0.69 0.66 0.65 0.65 density 2.525 2.515 2.522 2.513 2.514 2.506 2.518 CTE pred 36.8 35.9 35.8 35.8 35.6 34.5 36.1 Anneal pred 749 756 753 754 753 753 751 Youngs mod 81.6 80.9 81.5 81.7 81.7 82.3 81.3 (pred) specific modulus 32.3 32.2 32.3 32.5 32.5 32.8 32.3 (pred) 200p prediciton 1577 1594 1584 1581 1582 1578 1580 35kp prediciton 1212 1228 1220 1217 1218 1215 1217 SOC pred 29.47 29.74 29.64 29.59 29.67 29.8 29.65 liquidus 1177 1177 1179 1177 1175 1179 1175 prediction strain 693 699 708 707 710 711 705 anneal 746 752 753 752 754 756 751 soft 972.5 984.3 980.9 981.7 983.1 977.9 978.1 CTE 37.2 36.4 35.7 35.6 35.9 34.6 36.1 density 2.5254 2.5125 2.5208 2.5108 2.5134 2.5051 2.517 Poisson's ratio 0.24 0.243 Shear modulus 4.759 4.757 (Mpsi) Young's modulus 11.804 11.831 (Mpsi) Youngs mod 81.385 81.57187 (GPa) Specific modulus 32.285 32.40837 (Gpa/density) 200p 1579 1595 1588 1578 1582 1581 1582 35kp 1213 1226 1222 1219 1218 1219 1218 Liquidus 72 hr air 1160 1175 1180 1210 1170/1145 1180/1165 1210 internal 1150 1165 1170 1190 1165/1140 1170/1160 1185 Pt 1140 1150 1140 1165 1140/1135 1160/1155 1165 phase An + Cr Anorthite 1o Anorthite/ 1o cordierite/ Anorthite 2o cristobalite 2o cristobalite second phase 72 h liquidus 0 0 109631 64566 71743.94 viscosity (int) T35k - Tliq 63 61 52 29 53 49 33 22 23 24 25 26 27 28 weight % SiO2 (analyzed) 60.32 60.95 60.66 60.56 60.71 61.05 61.32 Al2O3 19.17 19.38 19.44 19.64 19.37 19.56 19.72 B2O3 5.75 5.7 5.71 5.87 5.59 5.78 5.89 MgO 3.378 3.934 2.963 2.807 2.993 3.022 2.966 CaO 6.048 5.553 6.327 6.31 6.397 6.666 6.942 SrO 4.58 4.19 4.78 4.76 4.5 3.87 2.93 BaO 0.06 0.06 0.07 0.06 0.06 0.05 0.04 SnO2 0.156 0.155 0.154 0.155 0.154 0.155 0.157 Fe2O3 0.057 0.056 0.057 0.057 0.057 0.058 0.056 ZrO2 0.043 0 0.042 0.045 0.048 0.046 0.045 As2O3 Sb2O3 mol % SiO2 66.51 66.5 66.45 66.4 66.73 66.54 66.82 Al2O3 12.38 12.47 12.61 12.76 12.54 12.63 12.69 B2O3 5.44 5.37 5.42 5.59 5.3 5.47 5.55 MgO 5.52 6.4 4.86 4.61 4.9 4.94 4.83 CaO 7.1 6.5 7.46 7.45 7.53 7.83 8.12 SrO 2.91 2.65 3.05 3.04 2.87 2.46 1.86 BaO 0.03 0.03 0.03 0.03 0.03 0.02 0.02 SnO2 0.07 0.07 0.07 0.07 0.07 0.07 0.07 Fe2O3 0.02 0.02 0.02 0.02 0.02 0.02 0.02 ZrO2 0.02 0 0.02 0.02 0.03 0.02 0.02 Properties RO/Al2O3 1.25 1.25 1.22 1.18 1.22 1.21 1.17 RO - Al2O3 3.18 3.11 2.79 2.37 2.79 2.62 2.14 RO/(Al2O3 + B2O3) 0.87 0.87 0.85 0.82 0.86 0.84 0.81 RO - Al2O3 - -2.26 -2.26 -2.63 -3.22 -2.51 -2.85 -3.41 B2O3 MgO/RO 0.36 0.41 0.32 0.31 0.32 0.32 0.33 CaO/RO 0.46 0.42 0.49 0.49 0.49 0.51 0.55 SrO/RO 0.19 0.17 0.20 0.20 0.19 0.16 0.13 (CaO + SrO)/ 0.81 0.73 0.83 0.82 0.83 0.81 0.79 Al2O3 (CaO + SrO + 0.81 0.74 0.84 0.82 0.83 0.82 0.79 BaO)/Al2O3 CaO/(CaO + SrO) 0.71 0.71 0.71 0.71 0.72 0.76 0.81 density 2.506 2.502 2.51 2.507 2.506 2.499 2.486 CTE pred 35.4 34.6 35.7 35.4 35.5 35.2 34.4 Anneal pred 753 755 756 756 757 756 759 Youngs mod 81.8 82.5 81.6 81.4 81.7 81.8 81.7 (pred) specific modulus 32.6 33 32.5 32.5 32.6 32.7 32.9 (pred) 200p prediciton 1581 1578 1582 1582 1586 1581 1585 35kp prediciton 1217 1215 1219 1220 1222 1218 1221 SOC pred 29.7 29.74 29.6 29.69 29.63 29.7 29.89 liquidus 1183 1189 1178 1176 1184 1185 1193 prediction strain 701 704 702 705 704 703 705 anneal 753 755 754 757 756 755 758 soft 979.4 981.9 984.1 986.6 984.2 986.4 988.3 CTE 32.4 34.8 35.2 35.6 36.2 36.2 34.7 density 2.5038 2.499 2.508 2.507 2.505 2.4952 2.471 Poisson's ratio 0.237 Shear modulus 4.811 (Mpsi) Young's modulus 11.901 (Mpsi) Youngs mod 82.05451 (GPa) Specific modulus 32.83494 (Gpa/density) 200p 1585 1586 1595 1595 1587 1587 1593 35kp 1217 1217 1221 1225 1221 1220 1225 Liquidus 72 hr air 1175 1195 1210 1225 1210 1180 1190 internal 1170 1185 1185 1200 1175 1175 1180 Pt 1155 1160 1160 1170 1160 1150 1170 phase Celsian + Cristobalite Anorthite Anorthite Anorthite Anorthite + Anorthite + Cristobalite Cristobalite Cristobalite second phase 72 h liquidus 98634.86 69323.89 74560.81 58863.62 96239.69 92830.54 92496.67 viscosity (int) T35k - Tliq 47 32 36 25 46 45 45 29 30 31 32 33 34 35 weight % SiO2 (analyzed) 60.55 60.99 61.3 60.72 60.86 60.77 61.2 Al2O3 19.48 19.45 19.35 19.38 19.4 19.2 18.96 B2O3 5.86 5.82 5.8 5.75 5.73 5.75 5.71 MgO 2.989 3.384 3.29 3.407 3.348 3.218 3.112 CaO 6.315 5.855 5.66 5.073 4.769 5.978 6.163 SrO 4.79 4.43 4.29 5.66 5.88 5.06 4.76 BaO 0.07 0.07 0.067 0.09 0.09 0.07 0.06
SnO2 0.158 0.154 0.16 0.151 0.153 0.154 0.153 Fe2O3 0.057 0.056 0.052 0.058 0.056 0.056 0.057 ZrO2 0.044 0.044 0.048 0 0.046 0.047 0.044 As2O3 Sb2O3 mol % SiO2 66.23 66.54 67.13 66.58 66.81 66.41 66.83 Al2O3 12.64 12.57 12.49 12.6 12.63 12.44 12.26 B2O3 5.57 5.51 5.48 5.47 5.47 5.46 5.41 MgO 4.91 5.53 5.37 5.6 5.52 5.28 5.09 CaO 7.45 6.88 6.64 6 5.65 7.04 7.25 SrO 3.06 2.82 2.73 3.62 3.77 3.23 3.03 BaO 0.03 0.03 0.03 0.04 0.04 0.03 0.03 SnO2 0.07 0.07 0.07 0.07 0.07 0.07 0.07 Fe2O3 0.02 0.02 0.02 0.02 0.02 0.02 0.02 ZrO2 0.02 0.02 0.03 0 0.02 0.03 0.02 Properties RO/Al2O3 1.22 1.21 1.18 1.21 1.18 1.25 1.25 RO - Al2O3 2.81 2.69 2.28 2.66 2.35 3.14 3.14 RO/(Al2O3 + B2O3) 0.85 0.84 0.82 0.84 0.83 0.87 0.87 RO - Al2O3 - -2.76 -2.82 -3.2 -2.81 -3.12 -2.32 -2.27 B2O3 MgO/RO 0.32 0.36 0.36 0.37 0.37 0.34 0.33 CaO/RO 0.48 0.45 0.45 0.39 0.38 0.45 0.47 SrO/RO 0.20 0.19 0.19 0.24 0.25 0.21 0.20 (CaO + SrO)/ 0.83 0.77 0.75 0.76 0.75 0.83 0.84 Al2O3 (CaO + SrO + 0.83 0.77 0.75 0.77 0.75 0.83 0.84 BaO)/Al2O3 CaO/(CaO + SrO) 0.71 0.71 0.71 0.62 0.60 0.69 0.71 density 2.51 2.502 2.494 2.514 2.513 2.512 2.506 CTE pred 35.7 34.8 34.2 35 34.7 35.7 35.6 Anneal pred 754 756 759 756 757 753 754 Youngs mod 81.6 81.8 81.6 81.7 81.5 81.7 81.5 (pred) specific modulus 32.5 32.7 32.7 32.5 32.4 32.5 32.5 (pred) 200p prediciton 1578 1582 1593 1585 1590 1581 1588 35kp prediciton 1216 1219 1227 1222 1227 1217 1221 SOC pred 29.62 29.79 30 29.74 29.84 29.64 29.76 liquidus 1176 1180 1185 1179 1181 1175 1184 prediction strain 704 703 706 706 705 700 700 anneal 755 755 758 757 757 753 753 soft 985 983.2 987.6 986.5 984.9 983.3 981.5 CTE 36.1 34.8 34.3 35.7 34 36 35.5 density 2.508 2.5013 2.498 2.5126 2.5131 2.5096 2.505 Poisson's ratio Shear modulus (Mpsi) Young's modulus (Mpsi) Youngs mod (GPa) Specific modulus (Gpa/density) 200p 1593 1590 1591 1599 1597 1589 1592 35kp 1220 1219 1225 1224 1227 1223 1224 Liquidus 72 hr air 1190 1170 1175 1180 1190 1185 1180 internal 1180 1170 1170 1170 1175 1165 1175 Pt 1160 1155 1160 1150 1160 1145 1150 phase Anorthite Anorthite + Anorthite + Cristobalite + Cristobalite + Cristobalite + Cristobalite Cristobalite Cristobalite Cordierite- Cordierite- Anorthite like crystals like crystals second phase 72 h liquidus 83109.07 99911.1 115749.1 114019.5 107797.9 123487.5 101088.6 viscosity (int) T35k - Tliq 40 49 55 54 52 58 49 36 37 38 39 40 41 42 weight % SiO2 (analyzed) 61.07 61.16 61.02 61.14 61.14 60.95 60.84 Al2O3 19.19 19.1 19.47 19.4 19.69 18.77 18.82 B2O3 5.88 5.69 5.72 5.73 6.07 6.15 6.17 MgO 3.129 3.116 2.885 2.821 2.801 2.556 2.571 CaO 6.364 6.363 6.273 6.187 5.578 5.546 5.551 SrO 4.48 4.42 4.65 4.68 4.36 5.51 5.52 BaO 0.06 0.06 0.07 0.07 0 0.05 0.04 SnO2 0.154 0.155 0.154 0.155 0.15 0.148 0.15 Fe2O3 0.057 0.056 0.057 0.057 0.056 0.057 0.057 ZrO2 0.044 0.042 0.046 0.047 0.046 0.064 0.064 As2O3 Sb2O3 mol % SiO2 66.44 66.77 66.7 66.91 67.39 67.48 67.39 Al2O3 12.4 12.34 12.63 12.59 12.77 12.22 12.25 B2O3 5.57 5.38 5.43 5.44 5.77 5.86 5.88 MgO 5.12 5.09 4.73 4.63 4.6 4.21 4.23 CaO 7.48 7.47 7.4 7.3 6.58 6.56 6.57 SrO 2.85 2.81 2.97 2.99 2.78 3.53 3.54 BaO 0.03 0.03 0.03 0.03 0 0.02 0.02 SnO2 0.07 0.07 0.07 0.07 0.07 0.07 0.07 Fe2O3 0.02 0.02 0.02 0.02 0.02 0.02 0.02 ZrO2 0.02 0.02 0.02 0.03 0.02 0.03 0.03 Properties RO/Al2O3 1.25 1.25 1.20 1.19 1.09 1.17 1.17 RO - Al2O3 3.08 3.06 2.50 2.36 1.19 2.10 2.11 RO/(Al2O3 + B2O3) 0.86 0.87 0.84 0.83 0.75 0.79 0.79 RO - Al2O3 - -2.49 -2.32 -2.93 -3.08 -4.58 -3.76 -3.77 B2O3 MgO/RO 0.33 0.33 0.31 0.31 0.33 0.29 0.29 CaO/RO 0.48 0.49 0.49 0.49 0.47 0.46 0.46 SrO/RO 0.18 0.18 0.20 0.20 0.20 0.25 0.25 (CaO + SrO)/ 0.83 0.83 0.82 0.82 0.73 0.83 0.83 Al2O3 (CaO + SrO + 0.84 0.84 0.82 0.82 0.73 0.83 0.83 BaO)/Al2O3 CaO/(CaO + SrO) 0.72 0.73 0.71 0.71 0.70 0.65 0.65 density 2.505 2.503 2.506 2.503 2.488 2.498 2.499 CTE pred 35.6 35.5 35.3 35.1 33.6 34.9 34.9 Anneal pred 753 755 757 758 761 755 755 Youngs mod 81.5 81.6 81.5 81.3 80.8 80 80.1 (pred) specific modulus 32.5 32.6 32.5 32.5 32.5 32 32.1 (pred) 200p prediciton 1580 1586 1587 1591 1599 1604 1603 35kp prediciton 1216 1220 1223 1226 1234 1234 1233 SOC pred 29.73 29.73 29.7 29.79 30.25 30.27 30.25 liquidus 1180 1186 1178 1178 1175 1167 1166 prediction strain 704 704 705 705 704 701 700 anneal 755 755 757 757 757 754 753 soft 985.2 985.7 986.4 987.3 990.9 985.5 986.3 CTE 35.7 36.1 35.8 35.4 33.9 34.5 34.6 density 2.502 2.501 2.5036 2.5027 2.486 2.498744 2.501356 Poisson's ratio 0.234 0.239 Shear modulus 4.688 4.695 (Mpsi) Young's modulus 11.572 11.638 (Mpsi) Youngs mod 79.78613 80.24119 (GPa) Specific modulus 31.9305 32.07907 (Gpa/density) 200p 1592 1596 1595 1601 1600 1600 1604 35kp 1223 1225 1224 1229 1233 1232 1233 Liquidus 72 hr air 1190 1185 1195 1200 1220 1180 1175 internal 1180 1180 1190 1185 1200 1165 1165 Pt 1150 1150 1160 1155 1160 1145 1140 phase Cristobalite + Cristobalite + Cristobalite + Cristobalite + Cristobalite + Cristobalite + Cristobalite + Anorthite Anorthite Anorthite Anorthite Mullite Anorthite Anorthite second phase 72 h liquidus 87524.31 91576.45 69910.47 88707.02 69863.45 149127.7 152147 viscosity (int) T35k - Tliq 43 45 34 44 33 67 68 43 44 45 46 47 48 49 weight % SiO2 (analyzed) 60.8 60.76 60.711 60.87 61.88 60.86 60.13 Al2O3 19.31 19.24 19.264 19.02 18.99 19.55 19.05 B2O3 6.08 5.94 5.56 5.79 7.34 7.43 6.71 MgO 2.438 2.604 2.802 3.048 2.655 2.741 2.631 CaO 5.311 5.361 5.479 5.44 6.787 7.005 5.353 SrO 5.65 5.69 5.783 5.48 1.97 2.05 5.67 BaO 0.05 0.05 0.047 0.04 0.02 0.01 0.05 SnO2 0.209 0.212 0.215 0.167 0.175 0.175 0.213 Fe2O3 0.054 0.053 0.053 0.037 0.039 0.038 0.035 ZrO2 0.044 0.042 0.043 0.041 0.041 0.043 0.061 As2O3 Sb2O3 mol % SiO2 67.43 67.29 67.15 67.09 67.36 66.4 66.66 Al2O3 12.62 12.56 12.56 12.35 12.17 12.56 12.43 B2O3 5.82 5.68 5.31 5.51 6.89 6.99 6.41 MgO 4.03 4.3 4.62 5.01 4.3 4.45 4.34 CaO 6.31 6.36 6.49 6.42 7.91 8.18 6.35 SrO 3.63 3.65 3.71 3.5 1.24 1.3 3.64 BaO 0.02 0.02 0.02 0.02 0.01 0 0.02 SnO2 0.09 0.09 0.09 0.07 0.08 0.08 0.09 Fe2O3 0.02 0.02 0.02 0.02 0.02 0.02 0.01 ZrO2 0.02 0.02 0.02 0.02 0.02 0.02 0.03 Properties RO/Al2O3 1.11 1.14 1.18 1.21 1.11 1.11 1.15 RO - Al2O3 1.37 1.77 2.28 2.60 1.29 1.37 1.92 RO/(Al2O3 + B2O3) 0.76 0.79 0.83 0.84 0.71 0.71 0.76 RO - Al2O3 - -4.45 -3.91 -3.03 -2.91 -5.6 -5.62 -4.49 B2O3 MgO/RO 0.29 0.30 0.31 0.34 0.32 0.32 0.30 CaO/RO 0.45 0.44 0.44 0.43 0.59 0.59 0.44 SrO/RO 0.26 0.26 0.25 0.23 0.09 0.09 0.25 (CaO + SrO)/ 0.79 0.80 0.81 0.80 0.75 0.75 0.80 Al2O3 (CaO + SrO + 0.79 0.80 0.81 0.80 0.75 0.75 0.81 BaO)/Al2O3 CaO/(CaO + SrO) 0.63 0.64 0.64 0.65 0.86 0.86 0.64 density 2.5 2.504 2.513 2.507 2.448 2.458 2.499 CTE pred 34.4 34.7 35.2 35.1 32.9 33.4 34.9 Anneal pred 760 759 760 756 750 748 750 Youngs mod 80.2 80.5 81.2 81.1 79.5 80 79.8 (pred) specific modulus 32.1 32.1 32.3 32.3 32.5 32.5 31.9 (pred) 200p prediciton 1605 1602 1598 1594 1595 1578 1591 35kp prediciton 1238 1235 1232 1228 1224 1213 1225 SOC pred 30.22 30.08 29.78 29.93 31.07 30.76 30.36 liquidus 1166 1169 1179 1173 1186 1174 1155 prediction strain 702 706 704 699 698 697 702 anneal 756 759 758 752 749 748 754 soft 994.8 988.4 987.7 982.1 981.8 979.1 983.5 CTE 34.1 34.7 35.7 35.1 33.2 33.7 34.9 density 2.500631 2.506029 2.512237 2.508 2.448 2.457 2.502 Poisson's ratio 0.229 0.231 0.236 0.235 0.233 0.237 0.224 Shear modulus 4.708 4.733 4.76 4.757 4.663 4.685 4.693 (Mpsi) Young's modulus 11.571 11.655 11.768 11.75 11.501 11.589 11.49 (Mpsi) Youngs mod 79.77924 80.3584 81.1375 81.0134 79.2966 79.90334 79.22076 (GPa) Specific modulus 31.90364 32.06603 32.29692 32.30199 32.3924 32.52069 31.66297 (Gpa/density) 200p 1605 1599 1596 1589 1593 1580 1593 35kp 1239 1237 1233 1224 1224 1215 1228 Liquidus 72 hr air 1175 1185 1170 1190 1200 1200 1175 internal 1160 1170 1160 1180 1180 1185 1155 Pt 1140 1150 1150 1150 1160 1155 1140 phase Cristobalite + Cristobalite + Cristobalite + Anorthite Cristobalite Cristobalite + Anorthite + Anorthite, air Anorthite, air Anorthite, air Mullite Cristobalite scum (anor) scum (anor) scum (anor) to 1220 to 1220 to 1240 second phase 72 h liquidus 199044.6 151378.2 174238.2 90955.1 89597.49 67118.68 174031.1 viscosity (int) T35k - Tliq 79 67 73 44 44 30 73 50 51 52 53 54 55 56 weight % SiO2 (analyzed) 60.44 60.74 61.12 61.41 61.79 62.25 62.55 Al2O3 19.11 19.26 19.46 19.5 19.6 19.78 19.84 B2O3 6.28 5.75 5.23 4.71 4.27 3.66 3.16 MgO 2.619 2.643 2.68 2.656 2.703 2.711 2.723 CaO 5.363 5.42 5.38 5.428 5.48 5.532 5.542 SrO 5.69 5.73 5.71 5.76 5.82 5.89 5.88 BaO 0.05 0.05 0.06 0.05 0.05 0.05 0.05 SnO2 0.214 0.215 0.23 0.214 0.211 0.213 0.212 Fe2O3 0.034 0.035 0.03 0.035 0.035 0.036 0.036 ZrO2 0.061 0.062 0.06 0.063 0.062 0.064 0.06 As2O3
Sb2O3 mol % SiO2 67.03 67.31 67.66 68.11 68.29 68.63 69.07 Al2O3 12.47 12.57 12.7 12.72 12.78 12.9 12.93 B2O3 6 5.49 5 4.5 4.08 3.5 3.02 MgO 4.32 4.36 4.42 4.38 4.46 4.47 4.49 CaO 6.36 6.43 6.38 6.44 6.5 6.56 6.57 SrO 3.65 3.68 3.67 3.7 3.73 3.78 3.77 BaO 0.02 0.02 0.03 0.02 0.02 0.02 0.02 SnO2 0.09 0.09 0.1 0.09 0.09 0.09 0.09 Fe2O3 0.01 0.01 0.01 0.01 0.01 0.01 0.01 ZrO2 0.03 0.03 0.03 0.03 0.03 0.03 0.03 Properties RO/Al2O3 1.15 1.15 1.14 1.14 1.15 1.15 1.15 RO - Al2O3 1.88 1.92 1.80 1.82 1.93 1.93 1.92 RO/(Al2O3 + B2O3) 0.78 0.80 0.82 0.84 0.87 0.90 0.93 RO - Al2O3 - -4.12 -3.57 -3.2 -2.68 -2.15 -1.57 -1.1 B2O3 MgO/RO 0.30 0.30 0.31 0.30 0.30 0.30 0.30 CaO/RO 0.44 0.44 0.44 0.44 0.44 0.44 0.44 SrO/RO 0.25 0.25 0.25 0.25 0.25 0.26 0.25 (CaO + SrO)/ 0.80 0.80 0.79 0.80 0.80 0.80 0.80 Al2O3 (CaO + SrO + 0.80 0.81 0.79 0.80 0.80 0.80 0.80 BaO)/Al2O3 CaO/(CaO + SrO) 0.64 0.64 0.63 0.64 0.64 0.63 0.64 density 2.502 2.507 2.511 2.515 2.52 2.527 2.529 CTE pred 34.8 34.9 34.7 34.8 34.9 34.9 34.9 Anneal pred 755 760 766 771 775 781 787 Youngs mod 80.1 80.7 81.3 81.8 82.3 83 83.4 (pred) specific modulus 32 32.2 32.4 32.5 32.7 32.8 33 (pred) 200p prediciton 1597 1602 1608 1615 1618 1624 1631 35kp prediciton 1231 1235 1241 1248 1251 1257 1263 SOC pred 30.21 29.96 29.75 29.57 29.35 29.07 28.9 liquidus 1162 1174 1185 1194 1204 1217 1226 prediction strain 704 710 714 720 725 730 735 anneal 756 762 766 771 777 782 788 soft 986.5 991.5 1000.5 1005.6 1008.7 1014 1022.5 CTE 35.1 34.9 35.2 34.7 35.4 34.9 35 density 2.505 2.507 2.514 2.517 2.521 2.528 2.529 Poisson's ratio 0.242 0.241 0.233 0.234 0.227 0.235 0.231 Shear modulus 4.721 4.747 4.831 4.805 4.804 4.882 4.914 (Mpsi) Young's modulus 11.726 11.779 11.91 11.86 11.804 12.058 12.096 (Mpsi) Youngs mod 80.84792 81.21335 82.11656 81.77182 81.38572 83.13698 83.39898 (GPa) Specific modulus 32.27462 32.39463 32.66371 32.48781 32.28311 32.88647 32.97706 (Gpa/density) 200p 1588 1599 1602 1613 1617 1624 1632 35kp 1224 1232 1237 1243 1250 1252 1259 Liquidus 72 hr air 1170 1180 1190 1215 1210 1230 1240 internal 1165 1170 1180 1185 1195 1215 1225 Pt 1140 1150 1160 1175 1180 1210 1220 phase Anorthite + Anorthite + anorthite anorthite anorthite Anorthite Anorthite Cristobalie Cristobalite second phase 72 h liquidus 128970.3 135681.9 117914.4 122199 115025.9 75109.91 69790.97 viscosity (int) T35k - Tliq 59 62 57 58 55 37 34 57 58 59 60 61 62 63 weight % SiO2 (analyzed) 62.61 63.09 60.65 60.55 59.77 61.34 60.76 Al2O3 18.47 18.37 19.25 19.17 19.63 18.75 19.19 B2O3 5.02 4.16 5.62 5.52 5.63 5.4 5.52 MgO 2.746 2.085 3.25 2.67 3.34 2.54 2.83 CaO 4.737 6.254 4.97 5.71 5.82 5.33 5.88 SrO 6.05 5.98 5.77 5.75 5.28 6.11 5.18 BaO 0.05 0.06 0.07 0.07 0.06 0.07 0.05 SnO2 0.313 0.257 0.208 0.207 0.203 0.207 0.209 Fe2O3 0.033 0.037 0.05 0.05 0.051 0.049 0.05 ZrO2 0.001 0.02 0.04 0.04 0.03 0.03 As2O3 Sb2O3 mol % SiO2 69.03 69.27 67.06 67.22 66.01 68.03 67.24 Al2O3 12.02 11.95 12.52 12.49 12.75 12.23 12.46 B2O3 4.78 3.96 5.35 5.27 5.35 5.16 5.25 MgO 4.52 3.43 5.35 4.4 5.49 4.19 4.65 CaO 5.6 7.4 5.88 6.77 6.87 6.32 6.94 SrO 3.87 3.83 3.69 3.69 3.37 3.92 3.31 BaO 0.02 0.03 0.03 0.03 0.03 0.03 0.02 SnO2 0.14 0.11 0.09 0.09 0.09 0.09 0.09 Fe2O3 0.01 0.02 0.02 0.02 0.02 0.02 0.02 ZrO2 0 0 0.01 0.02 0.02 0.02 0.02 Properties RO/Al2O3 1.16 1.23 1.19 1.19 1.23 1.18 1.20 RO - Al2O3 1.99 2.74 2.43 2.40 3.01 2.23 2.46 RO/(Al2O3 + B2O3) 0.83 0.92 0.84 0.84 0.87 0.83 0.84 RO - Al2O3 - -2.79 -1.22 -2.92 -2.87 -2.34 -2.93 -2.79 B2O3 MgO/RO 0.32 0.23 0.36 0.30 0.35 0.29 0.31 CaO/RO 0.40 0.50 0.39 0.46 0.44 0.44 0.47 SrO/RO 0.28 0.26 0.25 0.25 0.21 0.27 0.22 (CaO + SrO)/ 0.79 0.94 0.76 0.84 0.80 0.84 0.82 Al2O3 (CaO + SrO + 0.79 0.94 0.77 0.84 0.81 0.84 0.82 BaO)/Al2O3 CaO/(CaO + SrO) 0.59 0.66 0.61 0.65 0.67 0.62 0.68 density 2.502 2.516 2.511 2.513 2.519 2.509 2.507 CTE pred 34.1 35.9 34.8 35.5 35.8 35.1 35.2 Anneal pred 768 773 758 759 755 762 760 Youngs mod 80.7 81.2 81.5 81.1 82.1 80.6 81.3 (pred) specific modulus 32.3 32.3 32.5 32.3 32.6 32.1 32.4 (pred) 200p prediciton 1632 1637 1594 1599 1574 1615 1598 35kp prediciton 1257 1259 1230 1233 1214 1244 1231 SOC pred 30.16 29.52 29.84 29.75 29.43 29.97 29.78 liquidus 1195 1209 1178 1179 1186 1179 1180 prediction strain 711 719 706 709 704 709 707 anneal 765 771 759 760 755 762 759 soft 1006.7 1010.8 986 991.6 981.1 993.3 988.8 CTE 34 35.9 34.6 35.6 35.6 35.5 35.4 density 2.502 2.515 2.512 2.515 2.521 2.512 2.508 Poisson's ratio 0.223 0.226 0.234 0.235 0.234 0.233 0.243 Shear modulus 4.791 4.799 4.767 4.753 4.812 4.743 4.763 (Mpsi) Young's modulus 11.72 11.771 11.762 11.736 11.874 11.7 11.837 (Mpsi) Youngs mod 80.80656 81.15819 81.09614 80.91687 81.86835 80.66866 81.61324 (GPa) Specific modulus 32.29678 32.26966 32.28349 32.17371 32.47455 32.11332 32.54117 (Gpa/density) 200p 1637 1639 1588 1590 1567 1599 1589 35kp 1253 1259 1228 1236 1217 1243 1230 Liquidus 72 hr air -- 1220 1190 1190 1195 1190 1200 internal 1220 1220 1180 1185 1185 1180 1190 Pt 1200 1210 1160 1170 1170 1160 1175 phase cristobalite: Anorthite Cristobalite Cristobalite Anorthite Anorthite Anorthite Air-scum with Cristob. up to 1240 second phase Anorthite Cristobalite 72 h liquidus 68178.02 77873.11 97272.46 104315.3 69532.35 136418.1 81498 viscosity (int) T35k - Tliq 33 39 48 51 32 63 40 64 65 66 67 68 69 weight % SiO2 (analyzed) 60.57 60.83 60.46 60.48 60.59 60.36 Al2O3 19.18 19.28 19.18 19.21 19.22 19.15 B2O3 5.44 5.46 5.47 5.47 5.29 5.49 MgO 2.86 3.18 2.69 2.71 2.94 2.74 CaO 5.22 5.2 5.54 5.89 5.19 5.19 SrO 6.25 5.41 5.95 5.52 6.03 6.52 BaO 0.07 0.06 0.07 0.07 0.07 0.07 SnO2 0.207 0.205 0.205 0.207 0.206 0.204 Fe2O3 0.05 0.05 0.05 0.05 0.05 0.05 ZrO2 0.02 0.02 0.02 0.02 0.02 0.02 As2O3 Sb2O3 mol % SiO2 67.2 67.32 67.29 67.17 67.41 67.17 Al2O3 12.52 12.52 12.51 12.5 12.53 12.52 B2O3 5.2 5.19 5.23 5.21 5.05 5.26 MgO 4.72 5.22 4.44 4.46 4.85 4.53 CaO 6.19 6.14 6.57 6.97 6.15 6.17 SrO 4.01 3.46 3.82 3.53 3.87 4.2 BaO 0.03 0.03 0.03 0.03 0.03 0.03 SnO2 0.09 0.09 0.09 0.09 0.09 0.09 Fe2O3 0.02 0.02 0.02 0.02 0.02 0.02 ZrO2 0.01 0.01 0.01 0.01 0.01 0.01 Properties RO/Al2O3 1.19 1.18 1.19 1.20 1.19 1.19 RO - Al2O3 2.43 2.33 2.35 2.49 2.37 2.41 RO/(Al2O3 + B2O3) 0.84 0.84 0.84 0.85 0.85 0.84 RO - Al2O3 - -2.77 -2.86 -2.88 -2.72 -2.68 -2.85 B2O3 MgO/RO 0.32 0.35 0.30 0.30 0.33 0.30 CaO/RO 0.41 0.41 0.44 0.47 0.41 0.41 SrO/RO 0.27 0.23 0.26 0.24 0.26 0.28 (CaO + SrO)/ 0.81 0.77 0.83 0.84 0.80 0.83 Al2O3 (CaO + SrO + 0.82 0.77 0.83 0.84 0.80 0.83 BaO)/Al2O3 CaO/(CaO+ SrO) 0.61 0.64 0.63 0.66 0.61 0.59 density 2.518 2.508 2.515 2.512 2.516 2.521 CTE pred 35.4 34.6 35.4 35.5 35.1 35.6 Anneal pred 760 761 760 760 762 759 Youngs mod 81.2 81.6 81.1 81.2 81.4 81 (pred) specific modulus 32.2 32.5 32.2 32.3 32.4 32.1 (pred) 200p prediciton 1599 1599 1601 1598 1602 1600 35kp prediciton 1234 1233 1234 1231 1236 1234 SOC pred 29.71 29.83 29.74 29.7 29.7 29.72 liquidus 1184 1179 1180 1180 1184 1186 prediction strain 708 708 707 707 707 707 anneal 760 760 759 760 759 758 soft 992.1 991 990.1 990.2 989.6 990.7 CTE 35.2 35.1 35.8 34.6 34.6 35.5 density 2.519 2.51 2.517 2.512 2.518 2.523 Poisson's ratio 0.236 0.237 0.232 0.237 0.24 0.233 Shear modulus 4.756 4.786 4.751 4.753 4.76 4.751 (Mpsi) Young's modulus 11.756 11.84 11.709 11.761 11.805 11.715 (Mpsi) Youngs mod 81.05477 81.63393 80.73071 81.08924 81.39261 80.77208 (GPa) Specific modulus 32.17736 32.52348 32.07418 32.28075 32.32431 32.0143 (Gpa/density) 200p 1601 1593 1594 1594 1592 1582 35kp 1236 1233 1230 1235 1229 1233 Liquidus 72 hr air 1190 1175 1190 1190 1180 1190 internal 1180 1165 1175 1180 1170 1180 Pt 1160 1150 1160 1170 1150 1165 phase Anorthite Cristobalite Anorthite Anorthite Anorthite Anorthite second phase Cristobalite Anorthite Primary Cristobalite 72 h liquidus 116392.3 152728.1 113708.3 113653.4 126769.8 111997.5 viscosity (int) T35k - Tliq 56 68 55 55 59 53
[0096] It will be appreciated that the various disclosed embodiments may involve particular features, elements or steps that are described in connection with that particular embodiment. It will also be appreciated that a particular feature, element or step, although described in relation to one particular embodiment, may be interchanged or combined with alternate embodiments in various non-illustrated combinations or permutations.
[0097] It is also to be understood that, as used herein the terms "the," "a," or "an," mean "at least one," and should not be limited to "only one" unless explicitly indicated to the contrary. Thus, for example, reference to "a ring" includes examples having two or more such rings unless the context clearly indicates otherwise. Likewise, a "plurality" or an "array" is intended to denote "more than one." As such, a "plurality of droplets" includes two or more such droplets, such as three or more such droplets, etc., and an "array of rings" comprises two or more such droplets, such as three or more such rings, etc.
[0098] Ranges can be expressed herein as from "about" one particular value, and/or to "about" another particular value. When such a range is expressed, examples include from the one particular value and/or to the other particular value. Similarly, when values are expressed as approximations, by use of the antecedent "about," it will be understood that the particular value forms another aspect. It will be further understood that the endpoints of each of the ranges are significant both in relation to the other endpoint, and independently of the other endpoint.
[0099] While various features, elements or steps of particular embodiments may be disclosed using the transitional phrase "comprising," it is to be understood that alternative embodiments, including those that may be described using the transitional phrases "consisting" or "consisting essentially of," are implied. Thus, for example, implied alternative embodiments to a device that comprises A+B+C include embodiments where a device consists of A+B+C and embodiments where a device consists essentially of A+B+C.
[0100] It will be apparent to those skilled in the art that various modifications and variations can be made to the present disclosure without departing from the spirit and scope of the disclosure. Since modifications combinations, sub-combinations and variations of the disclosed embodiments incorporating the spirit and substance of the disclosure may occur to persons skilled in the art, the disclosure should be construed to include everything within the scope of the appended claims and their equivalents.
* * * * *
D00000

D00001

D00002

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.