Aglycosylated Enzyme And Uses Thereof
Medoff; Marshall ; et al.
U.S. patent application number 16/151690 was filed with the patent office on 2019-01-24 for aglycosylated enzyme and uses thereof. The applicant listed for this patent is XYLECO, INC.. Invention is credited to Natasha Kreder, Sean Landry, James J. Lynch, Thomas Craig Masterman, Marshall Medoff, Desiree Pangilinan, Aiichiro Yoshida.
| Application Number | 20190024068 16/151690 |
| Document ID | / |
| Family ID | 55264604 |
| Filed Date | 2019-01-24 |




| United States Patent Application | 20190024068 |
| Kind Code | A1 |
| Medoff; Marshall ; et al. | January 24, 2019 |
AGLYCOSYLATED ENZYME AND USES THEREOF
Abstract
The present invention relates to compositions comprising an aglycosylated polypeptide having cellobiase activity, and methods for producing and using the same.
| Inventors: | Medoff; Marshall; (Wakefield, MA) ; Kreder; Natasha; (Wakefield, MA) ; Lynch; James J.; (Woburn, MA) ; Landry; Sean; (Essex, MA) ; Yoshida; Aiichiro; (Canton, MA) ; Pangilinan; Desiree; (Waltham, MA) ; Masterman; Thomas Craig; (Rockport, MA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 55264604 | ||||||||||
| Appl. No.: | 16/151690 | ||||||||||
| Filed: | October 4, 2018 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 14768126 | Aug 14, 2015 | 10131894 | ||
| PCT/US15/44136 | Aug 7, 2015 | |||
| 16151690 | ||||
| 62035346 | Aug 8, 2014 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C12N 9/2445 20130101; C12P 19/14 20130101; C12P 19/02 20130101; C12Y 302/01021 20130101 |
| International Class: | C12N 9/42 20060101 C12N009/42; C12P 19/02 20060101 C12P019/02; C12P 19/14 20060101 C12P019/14 |
Claims
1. An aglycosylated polypeptide having cellobiase activity comprising at least 90% identity to SEQ ID NO: 1, or a functional fragment thereof.
2. The aglycosylated polypeptide of claim 1, wherein the polypeptide comprises a Cel3A enzyme from wild-type T. reesei, or a functional variant or fragment thereof.
3. The aglycosylated polypeptide of claim 2, wherein the Cel3A enzyme comprises (e.g., consists of) the amino acid sequence SEQ ID NO: 1.
4. The aglycosylated polypeptide of any one of claims 1-3, wherein the polypeptide is encoded by a nucleic acid sequence comprising (e.g., consisting of) SEQ ID NO: 2.
5. The aglycosylated polypeptide of any one of claims 1-4, wherein the polypeptide comprises a mutation proximal to or at one or more glycosylation sites, wherein the mutation prevents glycosylation at the one or more glycosylation sites.
6. The aglycosylated polypeptide of claim 5, wherein the mutation is at one or more of the threonine at amino acid position 78, the threonine at amino acid position 241, the serine at amino acid position 343, the serine at amino acid position 450, the threonine at amino acid position 599, the serine at amino acid position 616, the threonine at amino acid position 691, the serine at amino acid position 21, the threonine at amino acid position 24, the serine at amino acid position 25, the serine at amino acid position 28, the threonine at amino acid position 38, the threonine at amino acid position 42, the threonine at amino acid position 303, the serine at amino acid position at 398, the at serine amino acid position 435, the serine at amino acid position 436, the threonine at amino acid position 439, threonine at amino acid position 442, the threonine at amino acid position 446, the serine at amino acid position 451, the serine at amino acid position 619, the serine at amino acid position 622, the threonine at amino acid position 623, the serine at amino acid position 626, or the threonine at amino acid position 630 of SEQ ID NO: 1.
7. The aglycosylated polypeptide of any one of the preceding claims, wherein the aglycosylated polypeptide has increased cellobiase activity as compared to glycosylated Cel3A enzyme from wild-type T. reesei.
8. The aglycosylated polypeptide of any one of the preceding claims, wherein the aglycosylated polypeptide has increased substrate recognition, a more active substrate recognition site, or reduced steric hindrance as compared to glycosylated Cel3A enzyme from wild-type T. reesei.
9. The aglycosylated polypeptide of any one of the preceding claims, wherein the aglycosylated polypeptide hydrolyzes a carbohydrate such as cellobiose into one or more monosaccharides, e.g., glucose.
10. The aglycosylated polypeptide of any one of the preceding claims, wherein the cellobiase activity comprises hydrolysis of a beta 1,4 glycosidic linkage of cellobiose.
11. A nucleic acid sequence encoding the polypeptide of claims 1-10.
12. A nucleic acid sequence encoding a Cel3A enzyme or functional variant thereof, wherein the nucleic acid sequence comprises (e.g., consists of) at least 90% identity to SEQ ID NO: 2 or SEQ ID NO: 3.
13. A nucleic acid molecule comprising a nucleic acid sequence of any of claims 11 and 12.
14. The nucleic acid molecule of claim 13, further comprising a promoter, e.g., a promoter for prokaryotic cell expression, e.g., bacterial cell expression, e.g., expression in E. coli.
15. The nucleic acid molecule of claim 14, wherein the promoter sequence is a constitutive promoter sequence, inducible promoter sequence, or a repressible promoter sequence.
16. The nucleic acid molecule of claim 14 or 15, wherein the promoter is a T7 promoter.
17. The nucleic acid molecule of any one of claims 13-16, further comprising a nucleic acid sequence encoding a tag, e.g., a tag for detection and/or purification and/or for linkage to another molecule, e.g., a His tag.
18. The nucleic acid molecule of any one of claims 13-17, further comprising a nucleic acid encoding one or more signal sequences, e.g., a secretion signal sequence.
19. An expression vector comprising the nucleic acid sequence of any of claims 11-18.
20. The expression vector of claim 18, further comprising a nucleic acid sequence encoding a selection marker, e.g., a kanamycin or an ampicillin marker.
21. A cell comprising the vector of any one of claims 13-20.
22. A prokaryotic cell or bacterial cell comprising the vector of any one of claims 13-20.
23. A cell expressing a polypeptide of any one of claims 1-10.
24. A prokaryotic cell or bacterial cell expressing a polypeptide of any one of claims 1-10.
25. The bacterial cell of claim 22 or 24 wherein the bacterial cell is impaired for glycosylation.
26. The bacterial cell of claim 25, wherein the bacterial cell is an E. coli cell.
27. The bacterial cell of claim 26, wherein the E. coli cell is an origami E. coli cell.
28. A method for producing the aglycosylated polypeptide of any one of claim 1, 2, 3, 4, 7, 8, 9 or 10, comprising culturing a cell expressing a polypeptide of any one of claim 1, 2, 3, 4, 7, 8, 9 or 10, under conditions suitable for the expression of the polypeptide, wherein the cell does not glycosylate the polypeptide, e.g., a bacterial cell, e.g., an E. coli cell, e.g., an origami E. coli cell.
29. A method for producing the aglycosylated polypeptide of any of claim 1, 2, 3, 4, 7, 8, 9 or 10, comprising treating a polypeptide comprising an amino acid with at least 90% identity to SEQ ID NO: 1 with a deglycosylating enzyme.
30. The method of claim 29, wherein the deglycosylating enzyme is selected from PGNase and EndoH.
31. A method for producing the aglycosylated polypeptide of any one of claim 1, 2, 3, 4, 7, 8, 9 or 10, comprising culturing a cell that comprises a nucleic acid sequence encoding a polypeptide of any one of claim 1, 2, 3, 4, 7, 8, 9 or 10, wherein the nucleic acid sequence has one or more mutations which prevent glycosylation of the encoded polypeptide.
32. A method for culturing a cell expressing the aglycosylated polypeptide of any one of claim 1, 2, 3, 4, 7, 8, 9 or 10 in the presence of a glycosylation inhibitor, e.g., tunicamycin.
33. An enzyme mixture comprising a glycosylated polypeptide comprising an amino acid sequence with at least 90% identity to SEQ ID NO: 1 and an aglycosylated polypeptide comprising an amino acid sequence with at least 90% identity to SEQ ID NO: 1, wherein both of the glycosylated polypeptide and the aglycosylated peptide have cellobiase activity.
34. The enzyme mixture of claim 33, wherein the aglycosylated polypeptide is the aglycosylated polypeptide of any one of claims 1-10.
35. The enzyme mixture of claim 33 or 34, wherein the glycosylated polypeptide and the aglycosylated polypeptide both comprise Cel3A enzyme from wild-type T. reesei.
36. The enzyme mixture of any one of claims 33-35, further comprising at least one additional enzyme derived from a microorganism, wherein the additional enzyme has a biomass-degrading activity of a cellulose based material.
37. The enzyme mixture of claim 36, wherein the additional enzyme is selected from a ligninase, an endoglucanase, a cellobiohydrolase, xylanase, and a cellobiase.
38. The enzyme mixture of claim 33 or 34, wherein the mixture further comprises one or more ligninase, one or more endoglucanase, one or more cellobiohydrolase, one or more xylanase.
39. The enzyme mixture of any one of claims 33-38, wherein the ratio between the aglycosylated polypeptide to the remaining enzymes in the mixture is at least 1:32, e.g., 1:32 to 1:300.
40. The enzyme mixture of any of claims 33-38, wherein the ratio of the aglycosylated polypeptide to a glycosylated polypeptide is at least 1:32, e.g., 1:32 to 1:300.
41. A method of producing a product (e.g., a hydrogen, sugar, alcohol, etc.) from a biomass (or converting a biomass to a product) comprising contacting a biomass, e.g., by treatment with an electron beam, with an aglycosylated polypeptide of any one of claims 1-10 and a microorganism (mixture) that produces one or more biomass-degrading enzyme or an enzyme mixture comprising biomass-degrading enzymes under conditions suitable for the production of the sugar product.
42. A method of producing a product (e.g., a hydrogen, a sugar, an alcohol) from a biomass comprising contacting a biomass with an enzyme mixture of any one of claims 33-40 under conditions suitable for the production of the product.
43. The method of claim 41 or 42, wherein said product is a sugar product.
44. The method of any one of claims 41-43, further comprising isolating the sugar product.
45. The method of claim 44, wherein the isolating of the sugar product comprises precipitation, crystallization, chromatography, centrifugation, and/or extraction.
46. The method of any one of claims 43-45, wherein the sugar product is glucose and/or xylose.
47. The method of any one of claims 41-46, wherein the enzyme mixture comprises at least two of the enzymes selected from the group consisting of B2AF03, CIP1, CIP2, Cel1a, Cel3a, Cel5a, Cel6a, Cel7a, Cel7b, Cel12a, Cel45a, Cel74a, paMan5a, paMan26a, Swollenin, and the enzymes listed in Table 1.
48. The method of any one of claims 41-47, wherein the biomass comprises one or more of an agricultural product or waste, a paper product or waste, a forestry product, or a general waste, or any combination thereof; wherein: a) an agricultural product or waste comprises sugar cane jute, hemp, flax, bamboo, sisal, alfalfa, hay, arracacha, buckwheat, banana, barley, cassava, kudzu, oca, sago, sorghum, potato, sweet potato, taro, yams, beans, favas, lentils, peas, grasses, switchgrass, miscanthus, cord grass, reed canary grass, grain residues, canola straw, wheat straw, barley straw, oat straw, rice straw, corn cobs, corn stover, corn fiber, coconut hair, beet pulp, bagasse, soybean stover, grain residues, rice hulls, oat hulls, wheat chaff, barley hulls, or beeswing, or a combination thereof; b) a paper product or waste comprises paper, pigmented papers, loaded papers, coated papers, filled papers, magazines, printed matter, printer paper, polycoated paper, cardstock, cardboard, paperboard, or paper pulp, or a combination thereof; c) a forestry product comprises aspen wood, particle board, wood chips, or sawdust, or a combination thereof; and d) a general waste comprises manure, sewage, or offal, or a combination thereof.
49. The method of any one of claims 41-48, further comprising a step of treating the biomass prior to introducing the microorganism or the enzyme mixture to reduce the recalcitrance of the biomass, wherein the treating comprises bombardment with electrons, sonication, oxidation, pyrolysis, steam explosion, chemical treatment, mechanical treatment, or freeze grinding.
50. The method of any one of claims 41-49 wherein the microorganism that produces a biomass-degrading enzyme is from species in the genera selected from Bacillus, Coprinus, Myceliophthora, Cephalosporium, Scytalidium, Penicillium, Aspergillus, Pseudomonas, Humicola, Fusarium, Thielavia, Acremonium, Chrysosporium or Trichoderma.
51. The method of any one of claims 41-50, wherein the microorganism that produces a biomass-degrading enzyme is selected from Aspergillus, Humicola insolens (Scytalidium thermophilum) Coprinus cinereus, Fusarium oxysporum, Myceliophthora thermophila, Meripilus giganteus, Thielavia terrestris, Acremonium persicinum, Acremonium acremonium, Acremonium brachypenium, Acremonium dichromosporum, Acremonium obclavatum, Acremonium pinkertoniae, Acremonium roseogriseum, Acremonium incoloratum, Acremonium furatum, Chrysosporium lucknowense, Trichoderma viride, Trichoderma reesei, or Trichoderma koningii.
52. The method of any one of claims 41-51, wherein the microorganism has been induced to produce a biomass-degrading enzyme by combining the microorganism with an induction biomass sample under conditions suitable for increasing production of a biomass-degrading enzyme compared to an uninduced microorganism.
53. The method of any of claim 52, wherein said induction biomass sample comprises paper, paper products, paper waste, paper pulp, pigmented papers, loaded papers, coated papers, filled papers, magazines, printed matter, printer paper, polycoated paper, card stock, cardboard, paperboard, cotton, wood, particle board, forestry wastes, sawdust, aspen wood, wood chips, grasses, switchgrass, miscanthus, cord grass, reed canary grass, grain residues, rice hulls, oat hulls, wheat chaff, barley hulls, agricultural waste, silage, canola straw, wheat straw, barley straw, oat straw, rice straw, jute, hemp, flax, bamboo, sisal, abaca, corn cobs, corn stover, soybean stover, corn fiber, alfalfa, hay, coconut hair, sugar processing residues, bagasse, beet pulp, agave bagasse, algae, seaweed, manure, sewage, offal, agricultural or industrial waste, arracacha, buckwheat, banana, barley, cassava, kudzu, oca, sago, sorghum, potato, sweet potato, taro, yams, beans, favas, lentils, peas, or any combination thereof.
Description
RELATED APPLICATIONS
[0001] This application is a continuation of U.S. patent application Ser. No. 14/768,126, filed Aug. 14, 2015, which is a national stage application under 35 U.S.C. .sctn. 371 of International Application No. PCT/US2015/044136, filed Aug. 7, 2015, which claims the benefit of U.S. Provisional Application No. 62/035,346, filed Aug. 8, 2014; the entire contents of each of which are hereby incorporated by reference.
SEQUENCE LISTING
[0002] The instant application contains a Sequence Listing which has been submitted electronically in ASCII format and is hereby incorporated by reference in its entirety. Said ASCII copy, created on Aug. 5, 2015, is named X2002-7000WO_SL.txt and is 76,949 bytes in size.
FIELD OF THE INVENTION
[0003] The present invention relates generally to compositions having cellobiase activity and methods for producing the compositions described herein. The present invention also provides methods for using such compositions, e.g., to process biomass materials.
BACKGROUND OF THE INVENTION
[0004] Biomass-degrading enzymes, such as cellulases, xylanases, and ligninases, are important for the degradation of biomass, such as feedstock. Cellulosic and lignocellulosic materials are produced, processed, and used in large quantities in a number of applications. Often such materials are used once, and then discarded as waste, or are simply considered to be wasted materials, e.g., sewage, bagasse, sawdust, and stover
SUMMARY OF THE INVENTION
[0005] The present invention is based, at least in part, on the surprising discovery that a cellobiase from T. reesei that was expressed in a non-fungal cell line and isolated from a host cell that does not significantly glycosylate the enzyme had higher specific activity on pure substrate than the endogenous cellobiase (glycosylated and secreted) from T. reesei. Furthermore, when the aglycosylated cellobiase was used in a saccharification reaction with an enzyme mixture containing other saccharifying enzymes, a substantial increase in yield of sugar products was observed compared to the reaction without the aglycosylated cellobiase. Therefore, provided herein are, an aglycosylated polypeptide having enzymatic activity, e.g., cellobiase activity, compositions comprising the aglycosylated polypeptide and methods for producing and using the compositions described herein.
[0006] Accordingly, in one aspect, the disclosure features an aglycosylated polypeptide having cellobiase activity. In one embodiment, the aglycosylated polypeptide has increased cellobiase activity as compared to glycosylated Cel3A enzyme from wild-type T. reesei or a mutant thereof, such as T. reesei RUTC30. For example, the aglycosylated polypeptide can have increased substrate recognition or a more active substrate recognition site as compared to glycosylated Cel3A enzyme from wild-type T. reesei. In one embodiment, the aglycosylated polypeptide has reduced steric hindrance as compared to glycosylated Cel3A enzyme from wild-type T. reesei.
[0007] In one embodiment, the aglycosylated polypeptide comprises at least 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% identity to SEQ ID NO: 1, or a functional fragment thereof. In one embodiment, the aglycosylated polypeptide comprises (e.g., consists of) the amino acid sequence SEQ ID NO: 1. In another embodiment, the aglycosylated polypeptide differs by no more than 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 15, or 20 amino acids from the amino acid sequence of SEQ ID NO: 1.
[0008] In one embodiment, the polypeptide comprises a Cel3A enzyme from wild-type T. reesei, or a mutant thereof, such as T. reesei RUTC30, or a functional variant or fragment thereof.
[0009] In one embodiment, the aglycosylated polypeptide is encoded by a nucleic acid sequence, wherein the nucleic acid sequence comprises (e.g., consists of) at least 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% identity to SEQ ID NO: 2 or SEQ ID NO: 3. In one embodiment, the aglycosylated polypeptide is encoded by a nucleic acid sequence comprising (e.g., consisting of) SEQ ID NO: 2 or SEQ ID NO: 3.
[0010] In one embodiment, the aglcyosylated polypeptide is expressed by a gene that comprises a mutation proximal to or a mutation at one or more glycosylation site, wherein the mutation prevents glycosylation at the glycosylation site. In one embodiment, the mutation is at one or more of the threonine at amino acid position 78, the threonine at amino acid position 241, the serine at amino acid position 343, the serine at amino acid position 450, the threonine at amino acid position 599, the serine at amino acid position 616, the threonine at amino acid position 691, the serine at amino acid position 21, the threonine at amino acid position 24, the serine at amino acid position 25, the serine at amino acid position 28, the threonine at amino acid position 38, the threonine at amino acid position 42, the threonine at amino acid position 303, the serine at amino acid position at 398, the at serine amino acid position 435, the serine at amino acid position 436, the threonine at amino acid position 439, threonine at amino acid position 442, the threonine at amino acid position 446, the serine at amino acid position 451, the serine at amino acid position 619, the serine at amino acid position 622, the threonine at amino acid position 623, the serine at amino acid position 626, or the threonine at amino acid position 630 of SEQ ID NO: 1. A mutation proximal to one or more glycosylation site can prevent glycosylation at that site, e.g., by changing the conformation of the polypeptide or changing the consensus site recognized by the glycosylating enzyme such that glycosylation would not occur at the glycosylation site.
[0011] In one embodiment, the aglycosylated polypeptide hydrolyzes a carbohydrate, such as a dimer, a trimer, a tetramer, a pentamer, a hexamer, a heptamer, an octamer, or an oligomer of glucose; or an oligomer of glucose and xylose, into one or more monosaccharide, e.g., glucose. In one embodiment, the cellobiase activity comprises hydrolysis of a beta 1,4 glycosidic linkage of cellobiose.
[0012] In another aspect, the disclosure features a nucleic acid sequence encoding an aglycosylated polypeptide described herein.
[0013] In one embodiment, the nucleic acid sequence encodes a Cel3A enzyme or functional fragment thereof, wherein the nucleic acid sequence comprises (e.g., consists of) at least 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% identity to SEQ ID NO: 2 or SEQ ID NO: 3. In one embodiment, the nucleic acid sequence encodes a Cel3A enzyme, wherein the nucleic acid sequence comprises (e.g., consists of) SEQ ID NO: 2 or SEQ ID NO: 3.
[0014] In one aspect, the disclosure features a nucleic acid molecule that includes a nucleic acid sequence described herein.
[0015] In one embodiment, the nucleic acid molecule further includes a promoter, e.g., a promoter for prokaryotic cell expression, e.g., bacterial cell expression, e.g., expression in E. coli. In one embodiment, the promoter sequence is a constitutive promoter sequence, inducible promoter sequence, or a repressible promoter sequence. In one embodiment, the promoter is a T7 promoter.
[0016] In one embodiment, the nucleic acid molecule further comprises a nucleic acid sequence encoding a tag, e.g., a tag for detection and/or purification and/or for linkage to another molecule, e.g., a His tag.
[0017] In one embodiment, the nucleic acid molecule further comprises a nucleic acid encoding one or more signal sequences, e.g., a secretion signal sequence.
[0018] In one aspect, the disclosure features an expression vector comprising the nucleic acid sequence described herein or a nucleic acid molecule described herein.
[0019] In one embodiment, the vector further comprises a nucleic acid sequence encoding a selection marker, e.g., a kanamycin or an ampicillin marker.
[0020] In one aspect, the disclosure features a cell comprising a vector described herein.
[0021] In one embodiment, the cell is a prokaryotic cell/bacterial cell, e.g., an E. coli cell, e.g., an Origami E. coli cell.
[0022] In one embodiment, the cell expresses an aglycosylated polypeptide described herein.
[0023] In one embodiment, the cell is impaired for glycosylation.
[0024] In one aspect, the disclosure features a method for producing the aglycosylated polypeptide described herein, comprising culturing a cell expressing a polypeptide described herein, under conditions suitable for the expression of the polypeptide, wherein the cell does not glycosylate the polypeptide, e.g., a bacterial cell, e.g., an E. coli cell, e.g., an origami E. coli cell.
[0025] In one aspect, the disclosure features a method for producing the aglycosylated polypeptide described herein, comprising treating a polypeptide comprising an amino acid sequence of SEQ ID NO:1, or an amino acid sequence with at least 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% identity to SEQ ID NO: 1 with a deglycosylating enzyme. For example, a deglycosylating enzyme can be PGNase or EndoH.
[0026] In one aspect, the disclosure features a method for producing the aglycosylated polypeptide described herein, comprising culturing a cell that comprises a nucleic acid sequence encoding a polypeptide having at least 75%, 80%, 85%, 90%, 95%, 96%, 97%, 98%, 99% identity to SEQ ID NO: 1, wherein the nucleic acid has one or more mutations which prevent glycosylation of the encoded polypeptide, and optionally obtaining the aglycosylated polypeptide from the culture.
[0027] In one aspect, the disclosure features a method for culturing a cell expressing the aglycosylated polypeptide described herein in the presence of a glycosylation inhibitor. For example, the glycosylation inhibitor is tunicamycin.
[0028] In one aspect, the disclosure features an enzyme mixture comprising an aglycosylated polypeptide described herein and one or more additional enzyme, such as one or more glycosylated enzymes, e.g., cellulases from fungal cells. In one embodiment, the enzyme mixture comprises a glycosylated polypeptide comprising an amino acid sequence with at least 75%, 80%, 95%, 90%, 95%, 96%, 97%, 98%, 99% identity to SEQ ID NO: 1 and an aglycosylated polypeptide described herein, wherein both of the glycosylated polypeptide and the aglycosylated peptide have cellobiase activity.
[0029] In one embodiment, the enzyme mixture comprises a glycosylated polypeptide and an aglycosylated polypeptide and both the glycosylated polypeptide and the aglycosylated polypeptide both comprise Cel3A enzyme from wild-type T. reesei or a mutant thereof, such as T. reesei RUTC30.
[0030] In one embodiment, the enzyme mixture further comprises at least one additional enzyme derived from a microorganism, wherein the additional enzyme has a biomass-degrading activity. In one embodiment, the additional enzyme is selected from a ligninase, an endoglucanase, a cellobiohydrolase, xylanase, and a cellobiase.
[0031] In one embodiment, the reaction mixture has a ratio between the aglycosylated polypeptide and the remaining enzymes of at least 1:32, e.g., 1:32 to 1:300. In one embodiment, the reaction mixture has a ratio of the aglycosylated polypeptide to a glycosylated polypeptide is at least 1:32, e.g., 1:32 to 1:300.
[0032] In one aspect, the disclosure features a method of producing a product (e.g., hydrogen, a sugar, an alcohol, etc.) from a biomass (or converting a biomass to a product) comprising contacting a biomass, e.g., a biomass that has been treated with radiation, e.g., radiation from an electron beam, with an aglycosylated polypeptide described herein, under condiaitions suitable for the production of a sugar product. In one embodiment, the method further comprises contacting the biomass with a microorganism that produces one or more biomass-degrading enzyme or an enzyme mixture comprising biomass-degrading enzymes, e.g., an enzyme mixture described herein.
[0033] In one aspect, the disclosure features a method of producing a product (e.g., hydrogen, sugar, alcohol, etc.) from a biomass (or converting a biomass to a product) comprising contacting a biomass with an enzyme mixture described herein under conditions suitable for the production of the product.
[0034] In one embodiment, the product is a sugar product, e.g., a sugar product described herein. In one embodiment, the sugar product is glucose and/or xylose, or other sugar products, such as fructose, arabinose, galactose, and cellobiose.
[0035] In one embodiment, the method further comprises isolating the sugar product. In one embodiment, the isolating of the sugar product comprises precipitation, crystallization, chromatography, centrifugation, and/or extraction.
[0036] In one embodiment, the enzyme mixture comprises at least two of the enzymes selected from B2AF03, CIP1, CIP2, Cel1a, Cel3a, Cel5a, Cel6a, Cel7a, Cel7b, Cel12a, Cel45a, Cel74a, paMan5a, paMan26a, Swollenin, or any of the enzymes listed in Table 1. In an embodiment, the enzymes listed above are isolated from a cell that expresses the enzyme heterologously or endogenously, e.g., Trichoderma reesei or Podospora anserina.
[0037] In one embodiment, the biomass comprises a starchy material or a starchy material that includes a cellulosic component. In some embodiments, the biomass comprises one or more of an agricultural product or waste, a paper product or waste, a forestry product, or a general waste, or any combination thereof; wherein: a) an agricultural product or waste comprises sugar cane jute, hemp, flax, bamboo, sisal, alfalfa, hay, arracacha, buckwheat, banana, barley, cassava, kudzu, oca, sago, sorghum, potato, sweet potato, taro, yams, beans, favas, lentils, peas, grasses, switchgrass, miscanthus, cord grass, reed canary grass, grain residues, canola straw, wheat straw, barley straw, oat straw, rice straw, corn cobs, corn stover, corn fiber, coconut hair, beet pulp, bagasse, soybean stover, grain residues, rice hulls, oat hulls, wheat chaff, barley hulls, or beeswing, or a combination thereof; b) a paper product or waste comprises paper, pigmented papers, loaded papers, coated papers, filled papers, magazines, printed matter, printer paper, polycoated paper, cardstock, cardboard, paperboard, or paper pulp, or a combination thereof; c) a forestry product comprises aspen wood, particle board, wood chips, or sawdust, or a combination thereof; and d) a general waste comprises manure, sewage, or offal, or a combination thereof. In one embodiment, the method further comprises a step of treating the biomass prior to introducing the microorganism or the enzyme mixture to reduce the recalcitrance of the biomass, e.g., by treating the biomass with bombardment with electrons, sonication, oxidation, pyrolysis, steam explosion, chemical treatment, mechanical treatment, and/or freeze grinding.
[0038] In one embodiment, the microorganism that produces a biomass-degrading enzyme is from species in the genera selected from Bacillus, Coprinus, Myceliophthora, Cephalosporium, Scytalidium, Penicillium, Aspergillus, Pseudomonas, Humicola, Fusarium, Thielavia, Acremonium, Chrysosporium or Trichoderma. In one embodiment, the microorganism that produces a biomass-degrading enzyme is selected from Aspergillus, Humicola insolens (Scytalidium thermophilum), Coprinus cinereus, Fusarium oxysporum, Myceliophthora thermophila, Meripilus giganteus, Thielavia terrestris, Acremonium persicinum, Acremonium acremonium, Acremonium brachypenium, Acremonium dichromosporum, Acremonium obclavatum, Acremonium pinkertoniae, Acremonium roseogriseum, Acremonium incoloratum, Acremonium furatum, Chrysosporium lucknowense, Trichoderma viride, Trichoderma reesei, or Trichoderma koningii.
[0039] In one embodiment, the microorganism has been induced to produce biomass-degrading enzymes by combining the microorganism with an induction biomass sample under conditions suitable for increasing production of biomass-degrading enzymes compared to an uninduced microorganism. In one embodiment, the induction biomass sample comprises paper, paper products, paper waste, paper pulp, pigmented papers, loaded papers, coated papers, filled papers, magazines, printed matter, printer paper, polycoated paper, card stock, cardboard, paperboard, cotton, wood, particle board, forestry wastes, sawdust, aspen wood, wood chips, grasses, switchgrass, miscanthus, cord grass, reed canary grass, grain residues, rice hulls, oat hulls, wheat chaff, barley hulls, agricultural waste, silage, canola straw, wheat straw, barley straw, oat straw, rice straw, jute, hemp, flax, bamboo, sisal, abaca, corn cobs, corn stover, soybean stover, corn fiber, alfalfa, hay, coconut hair, sugar processing residues, bagasse, beet pulp, agave bagasse, algae, seaweed, manure, sewage, offal, agricultural or industrial waste, arracacha, buckwheat, banana, barley, cassava, kudzu, oca, sago, sorghum, potato, sweet potato, taro, yams, beans, favas, lentils, peas, or any combination thereof.
BRIEF DESCRIPTION OF THE DRAWINGS
[0040] FIG. 1 is a picture of purified Cel3a that was expressed in E. coli. Lane 1 represents molecular weight marker (Precision Plus All Blue Protein marker, Biorad); lane 2 represents purified Cel3a-His protein.
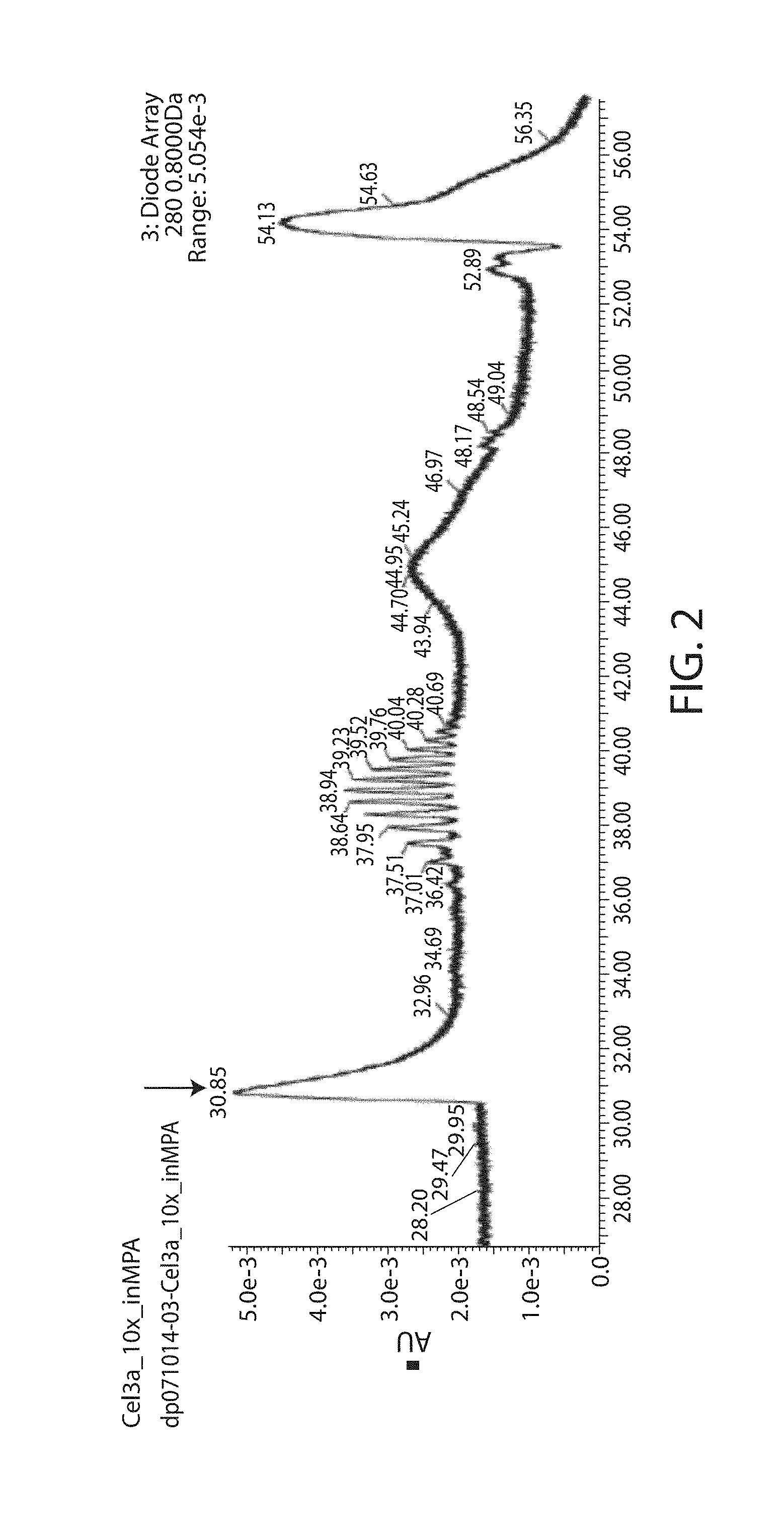
[0041] FIG. 2 is a chromatographic profile of the Cel3a-N'His sample, showing the detection of Cel3a-N'His at 31 minutes.
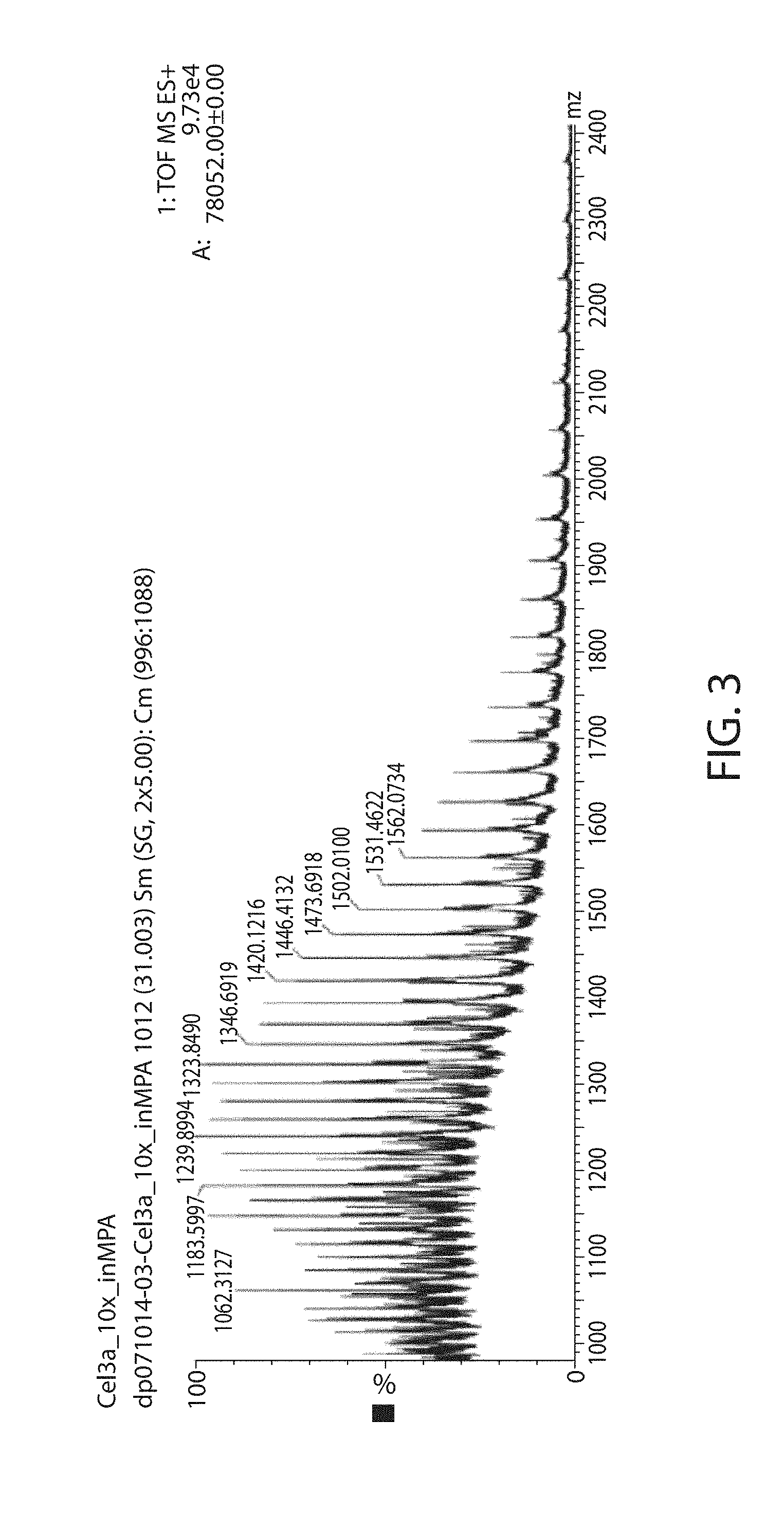
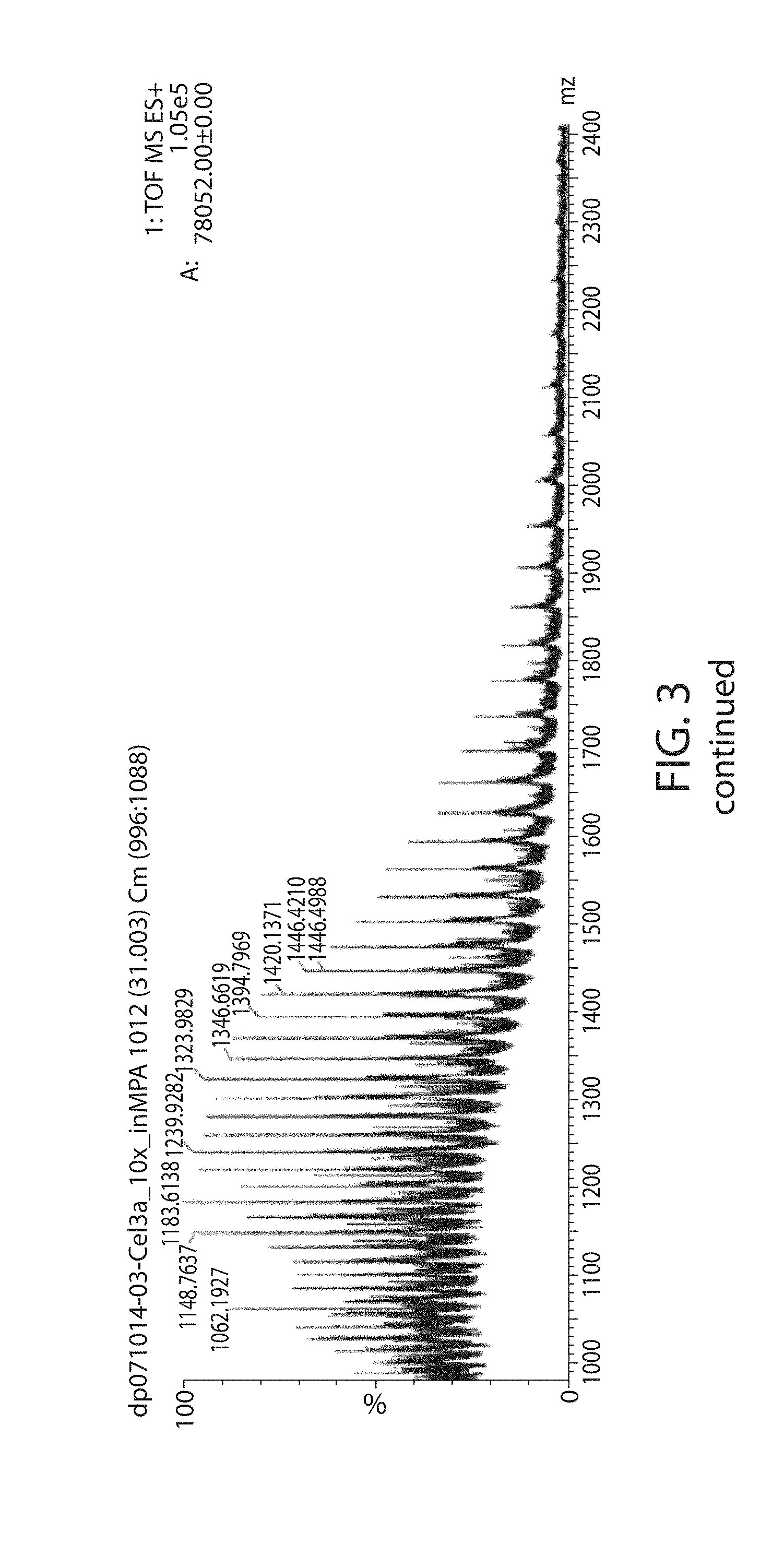
[0042] FIG. 3 is a profile showing no evidence of glycosylation in the mass spectral region for the peak in the chromatographic profile of the Cel3a-N'His at 31 minutes.
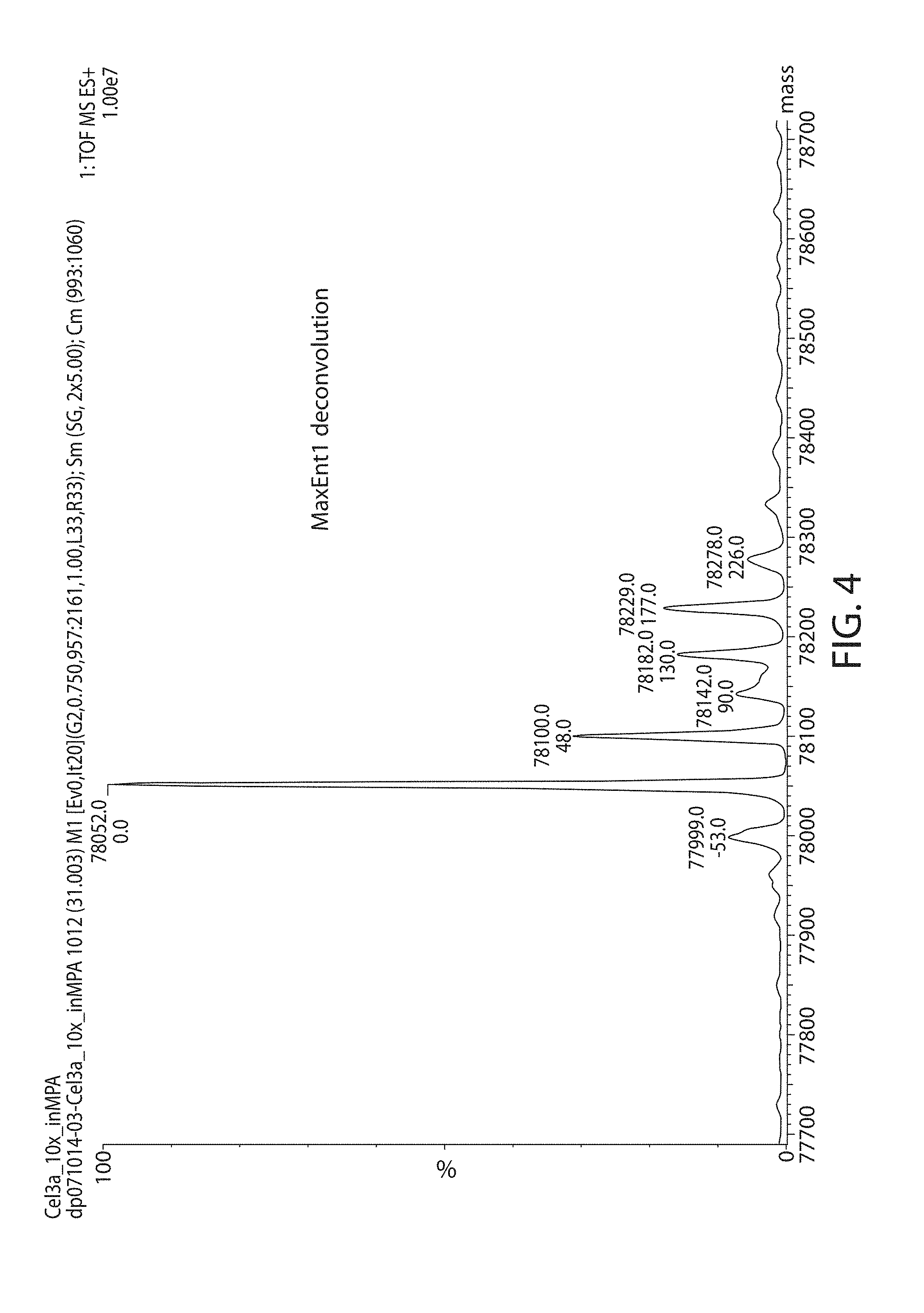
[0043] FIG. 4 is a profile showing the deconvolution of the charge state envelope, identifying the molecular weight of the major component (aglycosylated Cel3a-N'His) and the minor components (modified Cel3a-N'His).
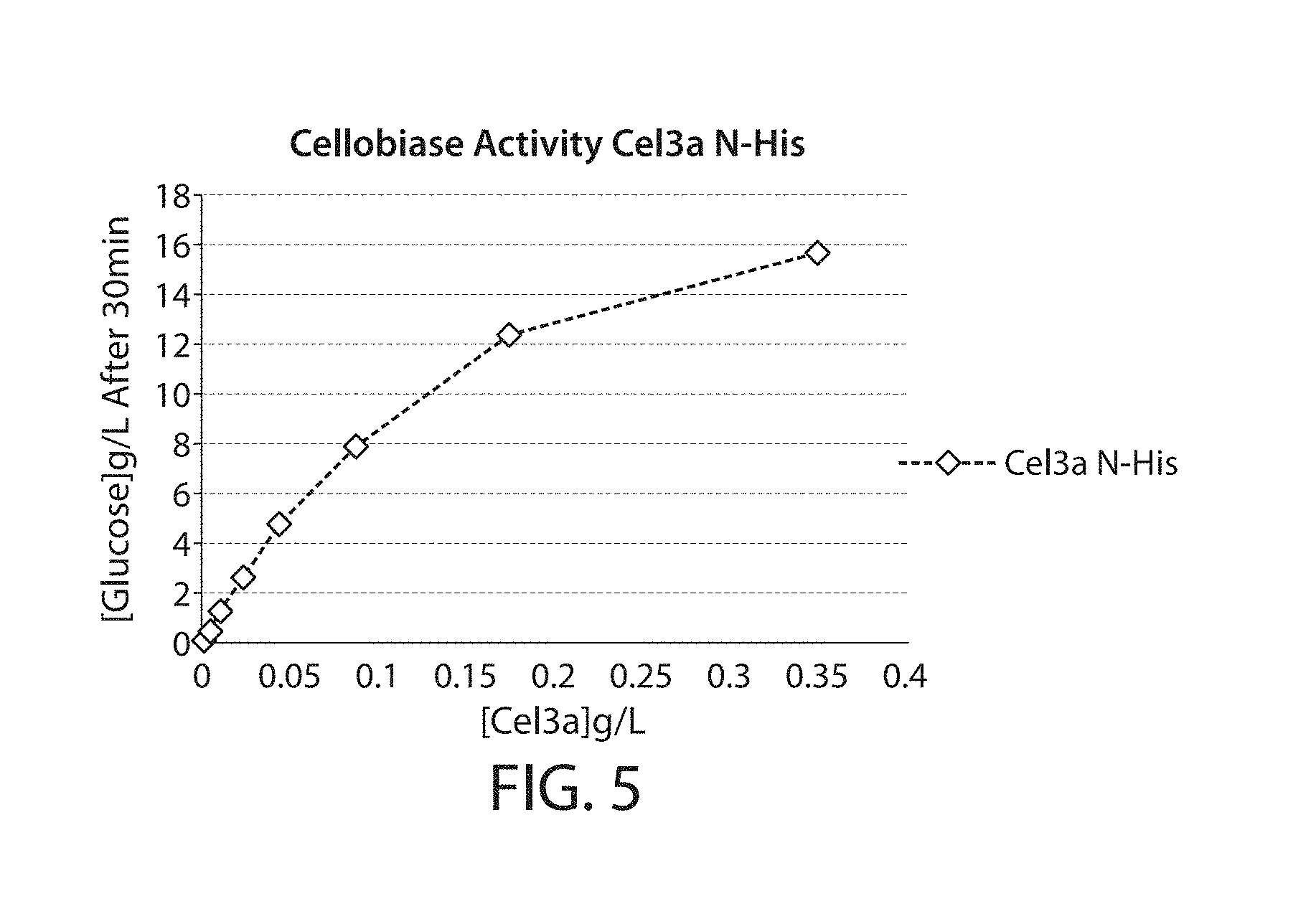
[0044] FIG. 5 is a graph showing the cellobiase activity, as determined by cellobiase assay, of a purified Cel3a-N'His.
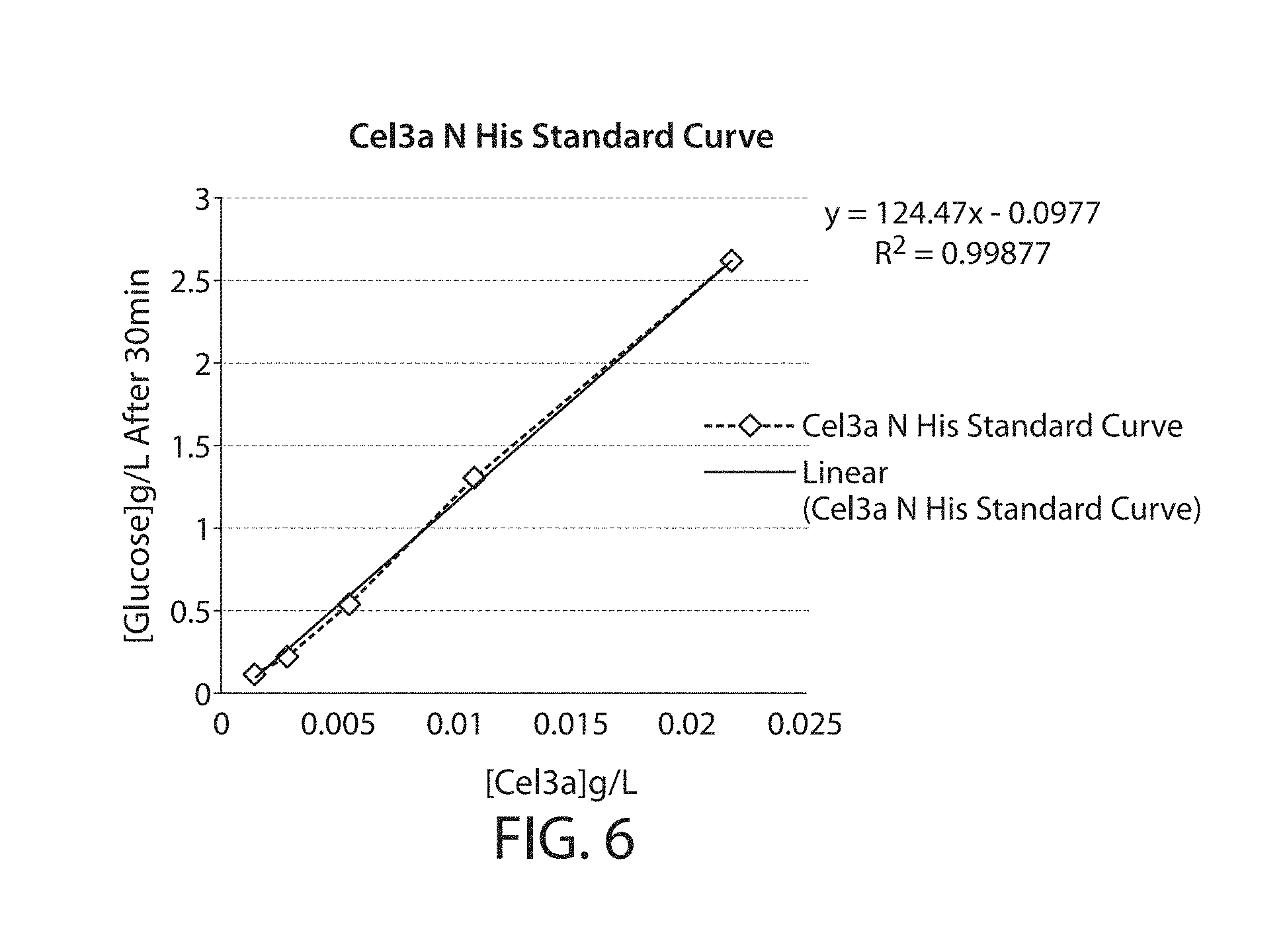
[0045] FIG. 6 is a graph showing a standard curve for cellobiase activity generated for a known concentration of Cel3a-N'His that can be used to determine the concentration of Cel3a of a sample with an unknown concentration of Cel3a.
[0046] FIG. 7 is a graph showing that specific activity of recombinant Cel3a compared to endogenous cellobiase from T. Reesei (L4196).
[0047] FIG. 8 is a graph showing the yield of glucose product from a saccharification reaction performed with a standard enzyme mix compared to the standard enzyme mix (L331 control) with the addition of aglycosylated cellobiase Cel3a (L331 (0.8 mg/ml Cel3a).
DETAILED DESCRIPTION
Definitions
[0048] Unless defined otherwise, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which the invention pertains.
[0049] The term "a" and "an" refers to one or to more than one (i.e., to at least one) of the grammatical object of the article. By way of example, "an element" means one element or more than one element.
[0050] The term "aglycosylated", as used herein, refers to a molecule, e.g., a polypeptide, that is not glycosylated (i.e., it comprises a hydroxyl group or other functional group that is not attached to a glycosylate group) at one or more sites which has a glycan attached when the molecule is produced in its native environment. For example, a Cel3A enzyme is aglycosylated when one or more site in the protein that normally has a glycan group attached to it when the Cel3A enzyme is produced in T. reesei does not have a glycan attached at that site. In some embodiments, the aglycosylated molecule does not have any attached glycans. In one embodiment, the molecule has been altered or mutated such that the molecule cannot be glycosylated, e.g., one or more glycosylation site is mutated such that a glycan cannot be attached to the glycosylation site. In another embodiment, an attached glycan can be removed from the molecule, e.g., by an enzymatic process, e.g., by incubating with enzymes that remove glycans or have deglycosylating activity. In yet another embodiment, glycosylation of the molecule can be inhibited, e.g., by use of a glycosylation inhibitor (that inhibits a glycosylating enzyme). In another embodiment, the molecule, e.g., the polypeptide, can be produced by a host cell that does not glycosylate, e.g., E. coli.
[0051] The term "biomass", as used herein, refers to any non-fossilized, organic matter. Biomass can be a starchy material and/or a cellulosic, hemicellulosic, or lignocellulosic material. For example, the biomass can be an agricultural product, a paper product, forestry product, or any intermediate, byproduct, residue or waste thereof, or a general waste. The biomass may be a combination of such materials. In an embodiment, the biomass is processed, e.g., by a saccharification and/or a fermentation reaction described herein, to produce products, such as sugars, alcohols, organic acids, or biofuels.
[0052] The term "biomass degrading enzymes", as used herein, refers to enzymes that break down components of the biomass matter described herein into intermediates or final products. For example, biomass-degrading enzymes include at least amylases, e.g., alpha, beta or gamma amylases, cellulases, hemicellulases, ligninases, endoglucancases, cellobiases, xylanases, and cellobiohydrolases. Biomass-degrading enzymes are produced by a wide variety of microorganisms, and can be isolated from the microorganisms, such as T. reesei. The biomass degrading enzyme can be endogenously expressed or heterologously expressed.
[0053] The term "cellobiase", as used herein, refers to an enzyme that catalyzes the hydrolysis of a dimer, trimer, tetramer, pentamer, hexamer, heptamer, octamer, or an oligomer of glucose, or an oligomer of glucose and xylose, to glucose and/or xylose. For example, the cellobiase is beta-glucosidase, which catalyzes beta-1,4 bonds in cellobiose to release two glucose molecules.
[0054] The term "cellobiase activity", as used herein, refers to activity of a category of cellulases that catalyze the hydrolysis of cellobiose to glucose, e.g., catalyzes the hydrolysis of beta-D-glucose residues to release beta-D-glucose. Cellobiase activity can be determined according to the assays described herein, e.g., in Example 6. One unit of cellobiase activity can be defined as [glucose] g/L/[Cel3a] g/L/30 minutes.
[0055] The term "cellobiohydrolase" as used herein, refers to an enzyme that hydrolyzes glycosidic bonds in cellulose. For example, the cellobiohydrolase is 1,4-beta-D-glucan cellobiohydrolase, which catalyzes the hydrolysis of 1,4-beta-D-glucosidic linkages in cellulose, cellooligosaccharides, or any beta-1,4-linked glucose containing polymer, releasing oligosaccharides from the polymer chain.
[0056] The term "endoglucanase" as used herein, refers to an enzyme that catalyzes the hydrolysis of internal .beta.-1,4 glucosidic bonds of cellulose. For example, the endoglucanase is endo-1,4-(1,3; 1,4)-beta-D-glucan 4-glucanohydrolase, which catalyses endohydrolysis of 1,4-beta-D-glycosidic linkages in cellulose, cellulose derivatives (such as carboxymethyl cellulose and hydroxyethyl cellulose), lichenan, beta-1,4 bonds in mixed beta-1, 3 glucans such as cereal beta-D-glucans or xyloglucans, and other plant material containing cellulosic components.
[0057] The term "enzyme mixture" as used herein, refers to a combination of at least two different enzymes, or at least two different variants of an enzyme (e.g., a glycosylated and an aglycosylated version of an enzyme). The enzyme mixture referred to herein includes at least the aglycosylated polypeptide having cellobiase activity described herein. In one embodiment, the enzyme mixture includes one or more of a cellobiase, an endoglucanase, a cellobiohydrolase, a ligninase, and/or a xylanase. In some embodiments, the enzyme mixture includes a cell, e.g., a microorganism, which expresses and, e.g., secretes, one or more of the enzymes. For example, the enzyme mixture can include an aglycosylated polypeptide described herein and a cell, e.g., a microorganism, which expresses and, e.g., secretes, one or more additional enzymes and/or variants of the polypeptide.
[0058] The term "ligninase" as used herein, refers to an enzyme that catalyzes the breakdown of lignin, commonly found in the cell walls of plants, such as by an oxidation reaction.
[0059] The terms "nucleic acid" or "polynucleotide" are used interchangeable, and refer to deoxyribonucleic acids (DNA) or ribonucleic acids (RNA) and polymers thereof in either single- or double-stranded form. Unless specifically limited, the term encompasses nucleic acids containing known analogues of natural nucleotides that have similar binding properties as the reference nucleic acid and are metabolized in a manner similar to naturally occurring nucleotides. Unless otherwise indicated, a particular nucleic acid sequence also implicitly encompasses conservatively modified variants thereof (e.g., degenerate codon substitutions), alleles, orthologs, SNPs, and complementary sequences as well as the sequence explicitly indicated. Specifically, degenerate codon substitutions may be achieved by generating sequences in which the third position of one or more selected (or all) codons is substituted with mixed-base and/or deoxyinosine residues (Batzer et al., Nucleic Acid Res. 19:5081 (1991); Ohtsuka et al., J. Biol. Chem. 260:2605-2608 (1985); and Rossolini et al., Mol. Cell. Probes 8:91-98 (1994)).
[0060] The term "operably linked", as used herein, refers to a configuration in which a control or regulatory sequence is placed at a position relative to a nucleic acid sequence that encodes a polypeptide, such that the control sequence influences the expression of a polypeptide (encoded by the DNA sequence). In an embodiment, the control or regulatory sequence is upstream of a nucleic acid sequence that encodes a polypeptide with cellobiase activity. In an embodiment, the control or regulatory sequence is downstream of a nucleic acid sequence that encodes a polypeptide with cellobiase activity.
[0061] The terms "peptide," "polypeptide," and "protein" are used interchangeably, and refer to a compound comprised of amino acid residues covalently linked by peptide bonds. A protein or peptide must contain at least two amino acids, and no limitation is placed on the maximum number of amino acids that can comprise a protein's or peptide's sequence. Polypeptides include any peptide or protein comprising two or more amino acids joined to each other by peptide bonds. "Polypeptides" include, for example, biologically active fragments, substantially homologous polypeptides, oligopeptides, homodimers, heterodimers, variants of polypeptides, modified polypeptides, derivatives, analogs, fusion proteins, among others. A polypeptide includes a natural peptide, a recombinant peptide, or a combination thereof.
[0062] The term "promoter", as used herein, refers to a DNA sequence recognized by the synthetic machinery of the cell, or introduced synthetic machinery, required to initiate the specific transcription of a polynucleotide sequence.
[0063] The term "regulatory sequence" or "control sequence", as used interchangeably herein, refers to a nucleic acid sequence which is required for expression of a nucleic acid product. In some instances, this sequence may be a promoter sequence and in other instances, this sequence may also include an enhancer sequence and other regulatory elements which are required for expression of the gene product. The regulatory/control sequence may, for example, be one which expresses the nucleic acid product in a regulated manner, e.g., inducible manner.
[0064] The term "constitutive" promoter refers to a nucleotide sequence which, when operably linked with a polynucleotide which encodes a polypeptide, causes the polypeptide to be produced in a cell under most or all physiological conditions of the cell. In an embodiment, the polypeptide is a polypeptide having cellobiase activity.
[0065] The term "inducible" promoter refers to a nucleotide sequence which, when operably linked with a polynucleotide which encodes a polypeptide, causes the polypeptide to be produced in a cell substantially only when an inducer which corresponds to the promoter is present in the cell. In an embodiment, the polypeptide is a polypeptide having cellobiase activity.
[0066] The term "repressible" promoter refers to a nucleotide sequence, which when operably linked with a polynucleotide which encodes a polypeptide, causes the polypeptide to be produced in a cell substantially only until a repressor which corresponds to the promoter is present in the cell. In an embodiment, the polypeptide is a polypeptide having cellobiase activity.
[0067] The term "xylanase" as used herein, refers to enzymes that hydrolyze xylan-containing material. Xylan is polysaccharide comprising units of xylose. A xylanase can be an endoxylanase, a beta-xylosidase, an arabinofuranosidase, an alpha-glucuronidase, an acetylxylan esterase, a feruloyl esterase, or an alpha-glucuronyl esterase.
Description
[0068] Glycosylation is thought to play a critical role in enzyme structure and function, such as enzyme activity, solubility, stability, folding, and/or secretion. Accordingly, processes for converting biomass into biofuels and other products have focused on producing and utilizing glycosylated enzymes, e.g., cellobiases, for use in saccharification of cellulosic and/or lignocellulosic materials. Enzymes for use in saccharification are typically produced in eukaryote host cell lines that properly glycosylate, fold, and secrete the proteins, such as Pichia pastoris.
[0069] The present invention is based, at least in part, on the surprising discovery that a cellobiase from T. reesei that was expressed in a non-fungal cell line and isolated from a host cell that does not significantly glycosylate the enzyme had higher specific activity on pure substrate than the endogenous cellobiase (glycosylated and secreted) from T. reesei. Furthermore, when the aglycosylated cellobiase was used in a saccharification reaction with an enzyme mixture containing other saccharifying enzymes, a substantial increase in yield of sugar products was observed compared to the reaction without the aglycosylated cellobiase. Therefore, provided herein are, an aglycosylated polypeptide having enzymatic activity, e.g., cellobiase activity, compositions comprising the aglycosylated polypeptide and methods for producing and using the compositions described herein.
Polypeptides and Variants
[0070] The present disclosure provides an aglycosylated polypeptide with cellobiase activity. In an embodiment, the aglycosylated polypeptide is a cellobiase. A cellobiase is an enzyme that hydrolyzes beta-1,4 bonds in its substrate, cellobiose, to release two glucose molecules. Cellobiose is a water soluble 1,4-linked dimer of glucose.
[0071] Cel3a (also known as BglI) is a cellobiase that was identified in Trichoderma reesei. The amino acid sequence for Cel3a (GenBank Accession No. NW_006711153) is provided below:
TABLE-US-00001 (SEQ ID NO: 1) MGDSHSTSGASAEAVVPPAGTPWGTAYDKAKAALAKLNLQDKVGIVSGVG WNGGPCVGNTSPASKISYPSLCLQDGPLGVRYSTGSTAFTPGVQAASTWD VNLIRERGQFIGEEVKASGIHVILGPVAGPLGKTPQGGRNWEGFGVDPYL TGIAMGQTINGIQSVGVQATAKHYILNEQELNRETISSNPDDRTLHELYT WPFADAVQANVASVMCSYNKVNTTWACEDQYTLQTVLKDQLGFPGYVMTD WNAQHTTVQSANSGLDMSMPGTDFNGNNRLWGPALTNAVNSNQVPTSRVD DMVTRILAAWYLTGQDQAGYPSFNISRNVQGNHKTNVRAIARDGIVLLKN DANILPLKKPASIAVVGSAAIIGNHARNSPSCNDKGCDDGALGMGWGSGA VNYPYFVAPYDAINTRASSQGTQVTLSNTDNTSSGASAARGKDVAIVFIT ADSGEGYITVEGNAGDRNNLDPWHNGNALVQAVAGANSNVIVVVHSVGAI ILEQILALPQVKAVVWAGLPSQESGNALVDVLWGDVSPSGKLVYTIAKSP NDYNTRIVSGGSDSFSEGLFIDYKHFDDANITPRYEFGYGLSYTKFNYSR LSVLSTAKSGPATGAVVPGGPSDLFQNVATVTVDIANSGQVTGAEVAQLY ITYPSSAPRTPPKQLRGFAKLNLTPGQSGTATFNIRRRDLSYWDTASQKW VVPSGSFGISVGASSRDIRLTSTLSVAGSGS
[0072] The present disclosure also provides functional variants of an aglycosylated polypeptide having cellobiase activity described herein, e.g., Cel3a. In an embodiment, a functional variant has an amino acid sequence with at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity to a Cel3a described herein, or a functional fragment thereof, e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity to a Cel3a described herein, or a functional fragment thereof. In an embodiment, a functional variant has an amino acid sequence with at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 91% identity, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity to SEQ ID NO: 1, or a functional fragment thereof.
[0073] Percent identity in the context of two or more amino acid or nucleic acid sequences, refers to two or more sequences that are the same. Two sequences are "substantially identical" if two sequences have a specified percentage of amino acid residues or nucleotides that are the same (e.g., 60% identity, optionally 70%, 71%, 72%, 73%, 74%, 75%, 76%, 77%, 78%, 79%, 80%, 81%, 82%, 83%, 84%, 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity over a specified region, or, when not specified, over the entire sequence), when compared and aligned for maximum correspondence over a comparison window, or designated region as measured using one of the following sequence comparison algorithms or by manual alignment and visual inspection. Optionally, the identity exists over a region that is at least about 50 nucleotides, 100 nucleotides, 150 nucleotides, in length. More preferably, the identity exists over a region that is at least about 200 or more amino acids, or at least about 500 or 1000 or more nucleotides, in length.
[0074] For sequence comparison, one sequence typically acts as a reference sequence, to which one or more test sequences are compared. When using a sequence comparison algorithm, test and reference sequences are entered into a computer, subsequence coordinates are designated, if necessary, and sequence algorithm program parameters are designated. Default program parameters can be used, or alternative parameters can be designated. The sequence comparison algorithm then calculates the percent sequence identities for the test sequences relative to the reference sequence, based on the program parameters. Methods of alignment of sequences for comparison are well known in the art. Optimal alignment of sequences for comparison can be conducted, e.g., by the local homology algorithm of Smith and Waterman, (1970) Adv. Appl. Math. 2:482c, by the homology alignment algorithm of Needleman and Wunsch, (1970) J. Mol. Biol. 48:443, by the search for similarity method of Pearson and Lipman, (1988) Proc. Nat'l. Acad. Sci. USA 85:2444, by computerized implementations of these algorithms (GAP, BESTFIT, FASTA, and TFASTA in the Wisconsin Genetics Software Package, Genetics Computer Group, 575 Science Dr., Madison, Wis.), or by manual alignment and visual inspection (see, e.g., Brent et al., (2003) Current Protocols in Molecular Biology).
[0075] Two examples of algorithms that are suitable for determining percent sequence identity and sequence similarity are the BLAST and BLAST 2.0 algorithms, which are described in Altschul et al., (1977) Nuc. Acids Res. 25:3389-3402; and Altschul et al., (1990) J. Mol. Biol. 215:403-410, respectively. Software for performing BLAST analyses is publicly available through the National Center for Biotechnology Information.
[0076] Functional variants may comprise one or more mutations, such that the variant retains cellobiase activity that is better, e.g., increased in comparison to, than the cellobiase activity of an ezyme of SEQ ID NO:1 produced by T. reesei. In an embodiment, the functional variant has at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, or at least 99% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, or at least 99%) of the cellobiase activity as an aglycosylated version of SEQ ID NO: 1 as produced by E. coli. Cellobiase activity can be tested using the functional assays described herein.
[0077] In another embodiment, the aglycosylated polypeptide differs by no more than 1, no more than 2, no more than 3, no more than 4, no more than 5, no more than 6, no more than 7, no more than 8, no more than 9, no more than 10, no more than 15, no more than 20, no more than 30, no more than 40, or no more than 50 amino acids from a reference amino acid sequence, e.g., the amino acid sequence of SEQ ID NO: 1.
[0078] The mutations present in a functional variant include amino acid substitutions, additions, and deletions. Mutations can be introduced by standard techniques known in the art, such as site-directed mutagenesis and PCR-mediated mutagenesis. The mutation may be a conservative amino acid substitution, in which the amino acid residue is replaced with an amino acid residue having a similar side chain. Families of amino acid residues having similar side chains have been defined in the art. These families include amino acids with basic side chains (e.g., lysine, arginine, histidine), acidic side chains (e.g., aspartic acid, glutamic acid), uncharged polar side chains (e.g., glycine, asparagine, glutamine, serine, threonine, tyrosine, cysteine, tryptophan), nonpolar side chains (e.g., alanine, valine, leucine, isoleucine, proline, phenylalanine, methionine), beta-branched side chains (e.g., threonine, valine, isoleucine) and aromatic side chains (e.g., tyrosine, phenylalanine, tryptophan, histidine). Thus, one or more amino acid residues within the polypeptide having cellobiase activity of the disclosure can be replaced with other amino acids from the same side chain family, and the resultant polypeptide retains cellobiase activity comparable (e.g., at least 80%, 85%, 90%, 95%, or 99% of the cellobiase activity) to that of the wild-type polypeptide. Alternatively, the mutation may be an amino acid substitution in which an amino acid residue is replaced with an amino acid residue having a different side chain.
[0079] Such mutations may alter or affect various enzymatic characteristics of the cellobiase. For example, such mutations may alter or affect the cellobiase activity, thermostability, optimal pH for reaction, enzyme kinetics, or substrate recognition of the cellobiase. In some embodiments, a mutation increases the cellobiase activity of the variant in comparison to the cellobiase produced by T. reesei and/or SEQ ID NO:1 produced in E.coli. In some embodiments, a mutation increases or decreases the thermostability of the variant in comparison to wild-type cellobiase and/or SEQ ID NO:1 produced in E.coli. In an embodiment, a mutation changes the pH range at which the variant optimally performs the cellobiase reaction in comparison to wild-type cellobiase and/or SEQ ID NO:1 produced in E.coli. In an embodiment, a mutation increases or decreases the kinetics of the cellobiase reaction (e.g., k.sub.cat, K.sub.M or K.sub.D) in comparison to wild-type cellobiase and/or SEQ ID NO:1 produced in E.coli. In an embodiment, a mutation increases or decreases the ability of the cellobiase to recognize or bind to the substrate (e.g., cellobiose) in comparison to wild-type cellobiase and/or SEQ ID NO:1 produced in E.coli.
[0080] The present invention also provides functional fragments of a polypeptide having cellobiase activity as described herein, e.g., Cel3a or SEQ ID NO: 1. One of ordinary skill in the art could readily envision that a fragment of a polypeptide having cellobiase activity as described herein that contains the functional domains responsible for enzymatic activity would retain functional activity, e.g., cellobiase activity, and therefore, such fragments are encompassed in the present invention. In an embodiment, the functional fragment is at least 700 amino acids, at least 650 amino acids, at least 600 amino acids, at least 550 amino acids, at least 500 amino acids, at least 450 amino acids, at least 400 amino acids, at least 350 amino acids, at least 300 amino acids, at least 250 amino acids, at least 200 amino acids, at least 150 amino acids, at least 100 amino acids, or at least 50 amino acids in length. In an embodiment, the functional fragment is 700 to 744 amino acids, 650 to 699 amino acids, 600 to 649 amino acids, 550 to 599 amino acids, 500 to 549 amino acids, 450 to 499 amino acids, 400 to 449 amino acids, 350 to 399 amino acids, 300 to 349 amino acids, 250 to 299 amino acids, 200 to 249 amino acids, 150 to 199 amino acids, 100 to 149 amino acids, or 50 to 99 amino acids. With regard to the ranges of amino acid length described above, the lowest and highest values of amino acid length are included within each disclosed range. In an embodiment, the functional fragment has at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, or at least 99% of the cellobiase activity as wild-type Cel3a or the polypeptide comprising SEQ ID NO: 1 produced in E.coli.
[0081] Assays for detecting cellobiase activity are known in the art. For example, detection of the amount of glucose released from cellobiose can be determined by incubating purified cellobiase with substrate, e.g., cellobiose, D-(+)-cellobiose, and detecting the resultant amount of free glucose after completion of the reaction. The amount of free glucose can be determined using a variety of methods known in the art. For example, dilutions of purified cellobiase are prepared in a buffer containing 50 mM sodium citrate monobasic, pH 5.0 NaOH. The cellobiose substrate is added to the purified cellobiase in an amount such that the final concentration of cellobiose in the reaction mixture is 30 mM. The reaction mixture is incubated under conditions suitable for the reaction to occur, e.g., in a shaker (700 rpm) at 48.degree. C. for 30 minutes. To stop the reaction, the reaction mixture is heated for 5 minutes at 100.degree. C. The reaction mixture is filtered through a 0.45 .mu.m filter and the filtrate is analyzed to quantify the amount of glucose and/or cellobiase. A YSI instrument that measures analytes such as glucose can be used to determine the concentration of glucose produced from the reaction. Alternatively, UPLC (Ultra Performance Liquid Chromatography) can be used to determine the concentration of glucose and cellobiose from the reaction. This assay can be formatted in a single reaction or in multiple reaction formats, e.g., 96 well format. In some embodiments, the multiple reaction format may be preferred to generate an activity curve representing cellobiase activity with respect to different concentrations of the purified cellobiase. The concentration of the purified cellobiase can be determined using a standard Bradford assay. Dilutions of the purified cellobiase assay are prepared, e.g., 2-fold dilutions, and are aliquoted into a 96 well plate, e.g., 12 wells of 2-fold dilutions. Cellobiose substrate is added as previously described, such that the final concentration of cellobiase in the reaction is 30 mM. The plate is sealed and treated under conditions sufficient for the cellobiase reaction to occur, and then under conditions to stop the reaction. The reaction is then filtered through a 96 well format 0.45 .mu.m membrane (e.g., Durapore) and analyzed by YSI and/or HPLC methods, e.g., UPLC.
[0082] This activity assay can also be used to determine the concentration, or titer, of a Cel3a in a sample by generating a standard curve of activity of dilutions of a Cel3a sample with a known concentration. The activity of dilutions of the sample with unknown concentration is determined and compared with the standard curve to identify an approximate concentration based on the standard curve. This method is described in further detail in Example 6.
[0083] In other embodiments, a colorimetric/fluorometric assay can be used. The purified cellobiase is incubated with substrate cellobiose under conditions for the reaction to occur. Detection of the product glucose is as follows. Glucose oxidase is added to the mixture, which oxidizes glucose (the product) to gluconic acid and hydrogen peroxide. Peroxidase and o-dianisidine is then added. O-dianisidine reacts with the hydrogen peroxide in the presence of peroxidase to form a colored product. Sulfuric acid is added, which reacts with the oxidized o-dianisidine reacts to form a more stable colored product. The intensity of the color when measured, e.g., by spectrophotometer or colorimeter, e.g., at 540 nm, is directly proportional to the glucose concentration. Such colorimetric/fluorometric glucose assays are commercially available, for example from Sigma Aldrich, Catalog No. GAGO-20.
[0084] For all of the polypeptides having cellobiase activity described above, the polypeptides are aglycosylated using the methods for producing aglycosylated polypeptides described herein.
[0085] Glycosylation is the enzymatic process by which a carbohydrate is attached to a glycosyl acceptor, e.g., the nitrogen of arginine or asparginine side chains or the hydroxyl oxygen of serine, threonine, or tyrosine side chains. There are two types of glycosylation: N-linked and O-linked glycosylation. N-linked glycosylation occurs at consensus site Asn-X-Ser/Thr, wherein the X can be any amino acid except a proline. O-linked glycosylation occurs at Ser/Thr residues. Glycosylation sites can be predicted using various algorithms known in the art, such as Prosite, publicly available by the Swiss Institute of Bioinformatics, and NetNGlyc 1.0 or NetOGlyc 4.0, publicly available by the Center for Biological Sequence Analysis.
[0086] In embodiments, the functional variant contains one or more mutations wherein one or more glycoslyation sites present in the Cel3a polypeptide expressed by a nucleic acid sequence described herein that has been mutated such that a glycan can no longer be attached or linked to the glycosylation site. In another embodiment, the functional variant contains one or more mutations proximal to one or more glycosylation sites present in the Cel3a polypeptide expressed by a nucleic acid sequence described herein that has been mutated such that a glycan can no longer be attached or linked to the glycosylation site. For example, the mutation proximal to a glycosylation site mutates the consensus motif recognized by the glycosylating enzyme, or changes the conformation of the polypeptide such that the polypeptide cannot be glycosylated, e.g., the glycoslation site is hidden or steric hindrance due to the new conformation prevents the glycosylating enzymes from accessing the glycosylation site. A mutation proximal to a glycosylation site in the Cel3a polypeptide is directly adjacent to, or at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 15, at least 20, at least 30 or at least 40 amino acids from the glycosylation site that, as a result of the proximal mutation, will not be glycosylated.
[0087] In an embodiment, one or more of the following glycosylation sites of Cel3a, or SEQ ID NO: 1, are mutated: the threonine at amino acid position 78, the threonine at amino acid position 241, the serine at amino acid position 343, the serine at amino acid position 450, the threonine at amino acid position 599, the serine at amino acid position 616, the threonine at amino acid position 691, the serine at amino acid position 21, the threonine at amino acid position 24, the serine at amino acid position 25, the serine at amino acid position 28, the threonine at amino acid position 38, the threonine at amino acid position 42, the threonine at amino acid position 303, the serine at amino acid position at 398, the at serine amino acid position 435, the serine at amino acid position 436, the threonine at amino acid position 439, threonine at amino acid position 442, the threonine at amino acid position 446, the serine at amino acid position 451, the serine at amino acid position 619, the serine at amino acid position 622, the threonine at amino acid position 623, the serine at amino acid position 626, or the threonine at amino acid position 630, or any combination thereof. In embodiments, the glycosylation site is mutated from a serine or threonine to an alanine. For example, the aglycosylated polypeptide described herein has one or more of the following mutations: T78A, T241A, S343A, S450A, T599A, S616A, T691A, S21A, T24A, S25A, S28A, T38A, T42A, T303A, T398A, S435A, S436A, T439A, T442A, T446A, S451A, S619A, S622A, T623A, S626A, or T630A, or any combination thereof. Alternatively, one or more amino acids proximal to the glycosylation sites described above is mutated.
[0088] Assays to detect whether a polypeptide is modified by a glycan (e.g., whether the polypeptide is glycosylated or aglycosylated) are known in the art. The polypeptide can be purified or isolated and can be stained for detection and quantification of glycan moieties, or the polypeptide can be analyzed by mass spectrometry, and compared to a corresponding reference polypeptide. The reference polypeptide has the same primary sequence as the test polypeptide (of which the glycosylation state is to be determined), but is either glycosylated or aglycosylated.
[0089] The aglycosylated polypeptides described herein have increased cellobiase activity compared to a corresponding glycosylated polypeptide, e.g., glycosylated Cel3a polypeptide. For example, the aglycosylated polypeptide having cellobiase activity has at least 1%, 2%, 5%, 10%, 15%, 20%, 25%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 100%, or 200% cellobiase activity compared to the glyocyosylated polypeptide.
Nucleic Acids
[0090] The present invention also provides a nucleic acid sequence encoding a polypeptide having cellobiase activity of the present invention. In an embodiment, the nucleic acid sequence encodes a Cel3a enzyme or a functional fragment thereof with the amino acid sequence described herein.
[0091] In an embodiment, the nucleic acid sequence that encodes Cel3a is provided below:
TABLE-US-00002 (SEQ ID NO: 2) ATGCGTTACCGAACAGCAGCTGCGCTGGCACTTGCCAC TGGGCCCTTTGCTAGGGCAGACAGTCACTCAACATCGG GGGCCTCGGCTGAGGCAGTTGTACCTCCTGCAGGGACT CCATGGGGAACCGCGTACGACAAGGCGAAGGCCGCATT GGCAAAGCTCAATCTCCAAGATAAGGTCGGCATCGTGA GCGGTGTCGGCTGGAACGGCGGTCCTTGCGTTGGAAAC ACATCTCCGGCCTCCAAGATCAGCTATCCATCGCTATG CCTTCAAGACGGACCCCTCGGTGTTCGATACTCGACAG GCAGCACAGCCTTTACGCCGGGCGTTCAAGCGGCCTCG ACGTGGGATGTCAATTTGATCCGCGAACGTGGACAGTT CATCGGTGAGGAGGTGAAGGCCTCGGGGATTCATGTCA TACTTGGTCCTGTGGCTGGGCCGCTGGGAAAGACTCCG CAGGGCGGTCGCAACTGGGAGGGCTTCGGTGTCGATCC ATATCTCACGGGCATTGCCATGGGTCAAACCATCAACG GCATCCAGTCGGTAGGCGTGCAGGCGACAGCGAAGCAC TATATCCTCAACGAGCAGGAGCTCAATCGAGAAACCAT TTCGAGCAACCCAGATGACCGAACTCTCCATGAGCTGT ATACTTGGCCATTTGCCGACGCGGTTCAGGCCAATGTC GCTTCTGTCATGTGCTCGTACAACAAGGTCAATACCAC CTGGGCCTGCGAGGATCAGTACACGCTGCAGACTGTGC TGAAAGACCAGCTGGGGTTCCCAGGCTATGTCATGACG GACTGGAACGCACAGCACACGACTGTCCAAAGCGCGAA TTCTGGGCTTGACATGTCAATGCCTGGCACAGACTTCA ACGGTAACAATCGGCTCTGGGGTCCAGCTCTCACCAAT GCGGTAAATAGCAATCAGGTCCCCACGAGCAGAGTCGA CGATATGGTGACTCGTATCCTCGCCGCATGGTACTTGA CAGGCCAGGACCAGGCAGGCTATCCGTCGTTCAACATC AGCAGAAATGTTCAAGGAAACCACAAGACCAATGTCAG GGCAATTGCCAGGGACGGCATCGTTCTGCTCAAGAATG ACGCCAACATCCTGCCGCTCAAGAAGCCCGCTAGCATT GCCGTCGTTGGATCTGCCGCAATCATTGGTAACCACGC CAGAAACTCGCCCTCGTGCAACGACAAAGGCTGCGACG ACGGGGCCTTGGGCATGGGTTGGGGTTCCGGCGCCGTC AACTATCCGTACTTCGTCGCGCCCTACGATGCCATCAA TACCAGAGCGTCTTCGCAGGGCACCCAGGTTACCTTGA GCAACACCGACAACACGTCCTCAGGCGCATCTGCAGCA AGAGGAAAGGACGTCGCCATCGTCTTCATCACCGCCGA CTCGGGTGAAGGCTACATCACCGTGGAGGGCAACGCGG GCGATCGCAACAACCTGGATCCGTGGCACAACGGCAAT GCCCTGGTCCAGGCGGTGGCCGGTGCCAACAGCAACGT CATTGTTGTTGTCCACTCCGTTGGCGCCATCATTCTGG AGCAGATTCTTGCTCTTCCGCAGGTCAAGGCCGTTGTC TGGGCGGGTCTTCCTTCTCAGGAGAGCGGCAATGCGCT CGTCGACGTGCTGTGGGGAGATGTCAGCCCTTCTGGCA AGCTGGTGTACACCATTGCGAAGAGCCCCAATGACTAT AACACTCGCATCGTTTCCGGCGGCAGTGACAGCTTCAG CGAGGGACTGTTCATCGACTATAAGCACTTCGACGACG CCAATATCACGCCGCGGTACGAGTTCGGCTATGGACTG TCTTACACCAAGTTCAACTACTCACGCCTCTCCGTCTT GTCGACCGCCAAGTCTGGTCCTGCGACTGGGGCCGTTG TGCCGGGAGGCCCGAGTGATCTGTTCCAGAATGTCGCG ACAGTCACCGTTGACATCGCAAACTCTGGCCAAGTGAC TGGTGCCGAGGTAGCCCAGCTGTACATCACCTACCCAT CTTCAGCACCCAGGACCCCTCCGAAGCAGCTGCGAGGC TTTGCCAAGCTGAACCTCACGCCTGGTCAGAGCGGAAC AGCAACGTTCAACATCCGACGACGAGATCTCAGCTACT GGGACACGGCTTCGCAGAAATGGGTGGTGCCGTCGGGG TCGTTTGGCATCAGCGTGGGAGCGAGCAGCCGGGATAT CAGGCTGACGAGCACTCTGTCGGTAGCG
[0092] The nucleic acid sequence encoding the polypeptide with cellobiase activity described herein can be codon-optimized for increased expression in host cells. Codon optimization includes changing the nucleic acid sequence to take into consideration factors including codon usage bias, cryptic splicing sites, mRNA secondary structure, premature polyA sites, interaction of codon and anti-codon, and RNA instability motifs, to increase expression of the encoded polypeptide in the host. Various algorithms and commercial services for codon-optimization are known and available in the art.
[0093] The codon-optimized nucleic acid sequence that encodes Cel3a is provided below:
TABLE-US-00003 (SEQ ID NO: 3) ATGCGTTATCGTACAGCCGCAGCCCTGGCACTGGCCAC AGGTCCGTTCGCACGTGCCGATAGTCACAGTACCAGCG GTGCCAGCGCAGAAGCCGTGGTTCCGCCGGCAGGCACA CCGTGGGGCACAGCCTATGATAAAGCCAAAGCCGCCCT GGCCAAGCTGAATCTGCAGGATAAAGTGGGCATCGTGA GTGGCGTGGGCTGGAACGGTGGTCCGTGCGTTGGCAAC ACCAGCCCGGCAAGCAAGATCAGCTATCCGAGCTTATG CCTGCAGGATGGTCCGCTGGGCGTGCGCTATAGCACCG GTAGTACCGCCTTTACACCTGGTGTGCAGGCCGCCAGT ACCTGGGACGTTAACCTGATCCGCGAACGTGGCCAATT TATCGGCGAAGAAGTTAAAGCCAGCGGCATTCATGTTA TTCTGGGTCCGGTGGCCGGTCCTCTGGGTAAAACCCCG CAGGGCGGCCGTAATTGGGAAGGCTTCGGCGTTGATCC GTATTTAACCGGCATCGCAATGGGCCAGACCATTAATG GCATCCAGAGCGTGGGTGTTCAAGCCACCGCCAAACAC TACATATTAAACGAACAGGAACTGAATCGTGAAACCAT CAGCAGCAATCCGGATGATCGCACCCTGCATGAGCTGT ATACATGGCCTTTTGCCGACGCAGTTCAGGCCAACGTG GCAAGTGTGATGTGTAGCTATAACAAGGTGAACACCAC CTGGGCCTGCGAAGACCAGTACACCCTGCAGACCGTTT TAAAAGACCAACTGGGCTTCCCTGGTTACGTGATGACA GATTGGAATGCCCAGCACACAACCGTTCAGAGCGCAAA CAGTGGCCTGGATATGAGCATGCCGGGCACCGACTTCA ACGGCAATAATCGTCTGTGGGGTCCGGCACTGACCAAT GCCGTTAACAGCAACCAGGTGCCGACCAGTCGTGTGGA CGATATGGTTACCCGTATTCTGGCCGCCTGGTACCTGA CAGGTCAAGACCAGGCCGGCTACCCGAGCTTCAACATC AGCCGCAACGTGCAGGGTAATCACAAGACCAACGTTCG CGCAATCGCACGCGATGGTATCGTGCTGTTAAAGAACG ATGCCAACATTCTGCCGCTGAAAAAACCGGCCAGCATC GCCGTTGTTGGTAGCGCAGCCATCATTGGCAACCACGC CCGTAACAGTCCGAGCTGCAATGATAAAGGCTGTGACG ACGGTGCCCTGGGCATGGGTTGGGGTAGTGGTGCCGTG AACTACCCGTATTTCGTGGCCCCGTACGACGCCATTAA CACCCGTGCAAGTAGCCAGGGTACCCAGGTTACCCTGA GCAACACCGACAACACAAGCAGCGGTGCCAGTGCAGCA CGTGGTAAGGATGTGGCCATCGTGTTCATCACCGCCGA CAGCGGCGAAGGCTACATTACCGTGGAGGGTAATGCCG GTGATCGCAATAATCTGGACCCGTGGCATAACGGCAAC GCCCTGGTTCAGGCAGTGGCAGGCGCAAATAGCAACGT GATCGTTGTGGTGCATAGCGTGGGTGCCATCATTCTGG AGCAGATCCTGGCCCTGCCGCAAGTTAAGGCAGTTGTG TGGGCAGGTCTGCCGAGCCAAGAAAGTGGCAATGCCCT GGTGGACGTTCTGTGGGGCGATGTTAGTCCGAGCGGCA AGCTGGTGTATACAATCGCCAAGAGCCCGAACGACTAT AACACCCGCATCGTTAGCGGCGGCAGTGATAGCTTCAG CGAGGGCCTGTTTATCGACTACAAGCATTTCGATGATG CCAATATTACCCCGCGCTACGAATTTGGTTATGGCCTG AGCTATACCAAGTTCAACTACAGCCGCCTGAGCGTTTT AAGTACCGCCAAGAGTGGTCCGGCAACAGGTGCCGTGG TTCCTGGTGGTCCGAGTGATCTGTTTCAGAATGTGGCC ACCGTGACCGTGGATATCGCCAACAGTGGTCAGGTTAC CGGCGCCGAAGTGGCACAGCTGTACATCACCTATCCGA GCAGTGCACCGCGCACCCCGCCGAAACAGCTGCGTGGC TTCGCCAAATTAAACCTGACCCCGGGCCAGAGCGGTAC AGCAACCTTCAATATTCGCCGCCGTGATCTGAGCTATT GGGACACCGCCAGCCAAAAATGGGTGGTGCCGAGCGGC AGCTTTGGCATTAGTGTGGGTGCAAGTAGCCGCGACAT TCGCTTAACAAGCACCCTGAGTGTTGCC
[0094] In an embodiment, the nucleic acid sequence encoding a Cel3a enzyme or functional variant thereof comprises at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity to SEQ ID NO: 2. In an embodiment, the nucleic acid sequence encoding a Cel3a enzyme or functional variant thereof comprises at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity to SEQ ID NO:2 or SEQ ID NO: 3.
[0095] Provided herein is a nucleic acid sequence encoding an aglycosylated polypeptide, e.g., Cel3a polypeptide, as described above, in which one or more glycoslyation sites present in the polypeptide has been mutated such that a glycan can no longer be attached or linked to the glycosylation site. In another embodiment, the nucleic acid sequence described herein encoding an aglycosylated polypeptide, e.g., a Cel3a polypeptide, as described above, in which one or more mutations proximal to one or more glycosylation sites present in the polypeptide has been mutated such that a glycan can no longer be attached or linked to the glycosylation site, as previously described. In an embodiment, the nucleic acid sequence encodes a polypeptide comprising one or more mutations at one or more of the following glycosylation sites of Cel3a, or SEQ ID NO: 1: the threonine at amino acid position 78, the threonine at amino acid position 241, the serine at amino acid position 343, the serine at amino acid position 450, the threonine at amino acid position 599, the serine at amino acid position 616, the threonine at amino acid position 691, the serine at amino acid position 21, the threonine at amino acid position 24, the serine at amino acid position 25, the serine at amino acid position 28, the threonine at amino acid position 38, the threonine at amino acid position 42, the threonine at amino acid position 303, the serine at amino acid position at 398, the at serine amino acid position 435, the serine at amino acid position 436, the threonine at amino acid position 439, threonine at amino acid position 442, the threonine at amino acid position 446, the serine at amino acid position 451, the serine at amino acid position 619, the serine at amino acid position 622, the threonine at amino acid position 623, the serine at amino acid position 626, or the threonine at amino acid position 630, or any combination thereof. In embodiments, the glycosylation site is mutated from a serine or threonine to an alanine. For example, the nucleic acid sequence of the invention encodes an aglycosylated polypeptide comprising one or more of the following mutations: T78A, T241A, S343A, S450A, T599A, S616A, T691A, S21A, T24A, S25A, S28A, T38A, T42A, T303A, T398A, S435A, S436A, T439A, T442A, T446A, S451A, S619A, S622A, T623A, S626A, or T630A, or any combination thereof. The ordinarily skilled artisan could readily modify the nucleic acid sequence of wild-type Cel3a (SEQ ID NO: 2) to encode a polypeptide with one or more glycosylation site mutation by using methods known in the art, such as site-directed mutagenesis.
[0096] The techniques used to isolate or clone a nucleic acid sequence encoding a polypeptide are known in the art and include isolation from genomic DNA, preparation from cDNA, or a combination thereof. The cloning of the nucleic acid sequences of the present invention from such genomic DNA can be effected, e.g., by using the well known polymerase chain reaction (PCR) or antibody screening of expression libraries to detect cloned DNA fragments with shared structural features. See, e.g., Innis et al., 1990, PCR: A Guide to Methods and Application, Academic Press, New York. Other amplification procedures such as ligase chain reaction (LCR), ligated activated transcription (LAT) and nucleotide sequence-based amplification (NASBA) may be used. The nucleic acid sequence may be cloned from a strain of Trichoderma reesei, e.g., wild-type T. reesei, or T. reesei RUTC30, or another or related organism and thus, for example, may be an allelic or species variant of the polypeptide encoding region of the nucleic acid sequence.
[0097] The nucleic acid sequence may be obtained by standard cloning procedures used in genetic engineering to relocate the nucleic acid sequence from its natural location to a different site where it will be reproduced. The cloning procedures may involve excision and isolation of a desired fragment comprising the nucleotide sequence encoding the polypeptide, insertion of the fragment into a vector molecule, and incorporation of the recombinant vector into a host cell where multiple copies or clones of the nucleotide sequence will be replicated. The nucleotide sequence may be of genomic, cDNA, RNA, semisynthetic, synthetic origin, or any combinations thereof.
Expression Vectors and Host Cells
[0098] The present invention also provides nucleic acid constructs comprising a nucleic acid sequence encoding the polypeptide having cellobiase activity described herein operably linked to one or more control sequences that direct the expression, secretion, and/or isolation of the expressed polypeptide.
[0099] As used herein, an "expression vector" is a nucleic acid construct for introducing and expressing a nucleic acid sequence of interest into a host cell. In some embodiments, the vector comprises a suitable control sequence operably linked to and capable of effecting the expression of the polypeptide encoded in the nucleic acid sequence of interest. The control sequence may be an appropriate promoter sequence, recognized by a host cell for expression of the nucleic acid sequence. In an embodiment, the nucleic acid sequence of interest is a nucleic acid sequence encoding a polypeptide having cellobiase activity as described herein.
[0100] A promoter in the expression vector of the invention can include promoters obtained from genes encoding extracellular or intracellular polypeptides either homologous or heterologous to the host cell, mutant promoters, truncated promoters, and hybrid promoters.
[0101] Examples of suitable promoters for directing transcription of the nucleic acid constructs of the present invention in a bacterial host cell are the promoters obtained from the E. coli lac operon, E. coli tac promoter (hybrid promoter, DeBoer et al, PNAS, 1983, 80:21-25), E. coli rec A, E. coli araBAD, E. coli tetA, and prokaryotic beta-lactamase. Other examples of suitable promoters include viral promoters, such as promoters from bacteriophages, including a T7 promoter, a T5 promoter, a T3 promoter, an M13 promoter, and a SP6 promoter. In some embodiments, more than one promoter controls the expression of the nucleic acid sequence of interest, e.g., an E. coli lac promoter and a T7 promoter. Further promoters that may be suitable for use in the present invention are described in "Useful proteins from recombinant bacteria" in Scientific American, 1980, 242:74-94, and Sambrook et al., Molecular Cloning: A Laboratory Manual, 1989. In some preferred embodiments, the promoter is inducible, where the addition of a molecule stimulates the transcription and expression of the downstream reading frame.
[0102] Examples of suitable promoters for directing transcription of the nucleic acid constructs of the present invention in a eukaryotic host cell, e.g., in a fungal or yeast cell are promoters obtained from the genes of Trichoderma Reesei, methanol-inducible alcohol oxidase (AOX promoter), Aspergillus nidulans tryptophan biosynthesis (trpC promoter), Aspergillus niger var. awamori flucoamylase (glaA), Saccharomyces cerevisiae galactokinase (GAL1), or Kluyveromyces lactis Plac4-PBI promoter.
[0103] A control sequence present in the expression vector of the present invention may also be a signal sequence that codes for an amino acid sequence linked to the amino terminus of a polypeptide and directs the encoded polypeptide into the cell's secretory pathway, e.g., a secretion signal sequence. The signal sequence may be an endogenous signal sequence, e.g., where the signal sequence is present at the N-terminus of the wild-type polypeptide when endogenously expressed by the organism from which the polypeptide of interest originates from. The signal sequence may be a foreign, or heterologous, signal peptide, in which the signal sequence is from a different organism or a different polypeptide than that of the polypeptide of interest being expressed. Any signal sequence which directs the expressed polypeptide into the secretory pathway of a host cell may be used in the present invention. Typically, signal sequences are composed of between 6 and 136 basic and/or hycrophobic amino acids.
[0104] Examples of signal sequences suitable for the present invention include the signal sequence from Saccharomyces cerevisiae alpha-factor.
[0105] Fusion tags may also be used in the expression vector of the present invention to facilitate the detection and purification of the expressed polypeptide. Examples of suitable fusion tags include His-tag (e.g., 3.times. His, 6.times. His (SEQ ID NO: 6), or 8.times. His (SEQ ID NO: 7)), GST-tag, HSV-tag, S-tag, T7 tag. Other suitable fusion tags include myc tag, hemagglutinin (HA) tag, and fluorescent protein tags (e.g., green fluorescent protein). The fusion tag is typically operably linked to the N or C terminus of the polypeptide to be expressed. In some embodiments, there may be a linker region between the fusion tag sequence and the N-terminus or C-terminus of the polypeptide to be expressed. In an embodiment, the linker region comprises a sequence between 1 to 20 amino acids, that does not affect or alter the expression or function of the expressed polypeptide.
[0106] Utilization of the fusion tags described herein allows detection of the expressed protein, e.g., by western blot by using antibodies that specifically recognize the tag. The tags also allows for purification of the expressed polypeptide from the host cell, e.g., by affinity chromatography. For example, an expressed polypeptide fused to a His-tag can be purified by using nickel affinity chromatography. The His tag has affinity for the Nickel ions, and a nickel column will retain the his-tagged polypeptide, while allowing all other proteins and cell debris to flow through the column. Elution of the His-tagged polypeptide using an elution buffer, e.g., containing imidazole, releases the His-tagged polypeptide from the column, resulting in substantially purified polypeptide.
[0107] The expression vector of the invention may further comprise a selectable marker gene to enable isolation of a genetically modified microbe transformed with the construct as is commonly known to those of skill in the art. The selectable marker gene may confer resistance to an antibiotic or the ability to grow on medium lacking a specific nutrient to the host organism that otherwise could not grow under these conditions. The present invention is not limited by the choice of selectable marker gene, and one of skill in the art may readily determine an appropriate gene. For example, the selectable marker gene may confer resistance to ampicillin, chloramphenicol, tetracycline, kanamycin, hygromycin, phleomycin, geneticin, or G418, or may complement a deficiency of the host microbe in one of the trp, arg, leu, pyr4, pyr, ura3, ura5, his, or ade genes or may confer the ability to grow on acetamide as a sole nitrogen source.
[0108] The expression vector of the invention may further comprise other nucleic acid sequences, e.g., additional control sequences, as is commonly known to those of skill in the art, for example, transcriptional terminators, synthetic sequences to link the various other nucleic acid sequences together, origins of replication, ribosome binding sites, a multiple cloning site (or polylinker site), a polyadenylation signal and the like. The ribosomal binding site suitable for the expression vector depends on the host cell used, for example, for expression in a prokaryotic host cell, a prokaryotic RBS, e.g., a T7 phage RBS can be used. A multiple cloning site, or polylinker site, contains one or more restriction enzyme sites that are preferably not present in the remaining sequence of the expression vector. The restriction enzyme sites are utilized for the insertion of a nucleic acid sequence encoding a polypeptide having cellobiase activity or other desired control sequences. The practice of the present invention is not limited by the presence of any one or more of these other nucleic acid sequences, e.g., other control sequences.
[0109] Examples of suitable expression vectors for use in the present invention include vectors for expression in prokaryotes, e.g., bacterial expression vectors. A bacterial expression vector suitable for use in the present invention in the pET vector (Novagen), which contains the following: a viral T7 promoter which is specific to only T7 RNA polymerase (not bacterial RNA polymerase) and also does not occur anywhere in the prokaryotic genome, a lac operator comprising a lac promoter and coding sequence for the lac repressor protein (lacI gene), a polylinker, an f1 origin of replication (so that a single-stranded plasmid can be produced when co-infected with M13 helper phage), an ampicillin resistance gene, and a ColE1 origin of replication (Blaber, 1998). Both the promoter and the lac operator are located 5', or upstream, of the polylinker in which the nucleic acid sequence encoding a polypeptide described herein is inserted. The lac operator confers inducible expression of the nucleic acid sequence encoding a polypeptide having cellobiase activity. Addition of IPTG (Isopropyl .beta.-D-1-thiogalactopyranoside), a lactose metabolite, triggers transcription of the lac operon and induces protein expression of the nucleic acid sequence under control of the lac operator. Use of this system requires the addition of T7 RNA polymerase to the host cell for vector expression. The T7 RNA polymerase can be introduced via a second expression vector, or a host cell strain that is genetically engineered to express T7 RNA polymerase can be used.
[0110] An exemplary expression vector for use with the invention is a pET vector, commercially available from Novagen. The pET expression system is described in U.S. Pat. Nos. 4,952,496; 5,693,489; and 5,869,320. In one embodiment, the pET vector is a pET-DUET vector, e.g., pET-Duet1, commercially available from Novagen. Other vectors suitable for use in the present invention include vectors containing His-tag sequences, such as those described in U.S. Pat. Nos. 5,310,663 and 5,284,933; and European Patent No. 282042.
[0111] The present invention also relates to a host cell comprising the nucleic acid sequence or expression vector of the invention, which are used in the recombinant production of the polypeptides having cellobiase activity.
[0112] An expression vector comprising a nucleic acid sequence of the present invention is sintroduced into a host cell so that the vector is maintained (e.g., by chromosomal integration or as a self-replicating extra-chromosomal vector) such that the polypeptide is expressed.
[0113] The host cell may be a prokaryote or a eukaryote. The host cell may be a bacteria, such as an E. coli strain, e.g., K12 strains NovaBlue, NovaBlue T1R, JM109, and DH5.alpha.. Preferably, the bacteria cell has the capability to fold, or partially fold, exogenously expressed proteins, such as E. coli Origami strains, e.g., Origami B, Origami B (DE3), Origami 2, and Origami 2(DE3) strains. In some embodiments, it may be preferred to use a host cell that is deficient for glycosylation, or has an impaired glycosylation pathway such that proteins expressed by the host cell are not significantly glycosylated.
[0114] The host cell may be a yeast or a filamentous fungus, particularly those classified as Ascomycota. Genera of yeasts useful as host microbes for the expression of modified TrCel3A beta-glucosidases of the present invention include Saccharomyces, Pichia, Hansenula, Kluyveromyces, Yarrowia, and Arxula. Genera of fungi useful as microbes for the expression of the polypeptides of the present invention include Trichoderma, Hypocrea, Aspergillus, Fusarium, Humicola, Neurospora, Chrysosporium, Myceliophthora, Thielavia, Sporotrichum and Penicillium. For example, the host cell may be Pichia pastoris. For example, the host cell may be an industrial strain of Trichoderma reesei, or a mutant thereof, e.g., T. reesei RUTC30. Typically, the host cell is one which does not express a parental cellobiase or Cel3a.
[0115] The selection of the particular host cell, e.g., bacterial cell or a fungal cell, depends on the expression vector (e.g., the control sequences) and/or the method utilized for producing an aglycosylated polypeptide of the invention, as described in further detail below.
[0116] The expression vector of the invention may be introduced into the host cell by any number of methods known by one skilled in the art of microbial transformation, including but not limited to, transformation, treatment of cells with CaCl.sub.2, electroporation, biolistic bombardment, lipofection, and PEG-mediated fusion of protoplasts (e.g. White et al., WO 2005/093072, which is incorporated herein by reference). After selecting the recombinant host cells containing the expression vector (e.g., by selection utilizing the selectable marker of the expression vector), the recombinant host cells may be cultured under conditions that induce the expression of the polypeptide having cellobiase activity of the invention.
Methods for Producing Aglycosylated Polypeptides
[0117] The present invention further provides methods for producing an aglycosylated polypeptide having cellobiase activity, as described herein. The method comprises culturing the recombinant host cell expressing the polypeptide of the present invention under conditions suitable for the expression of the polypeptide. The method may also comprise recovering the aglycosylated polypeptide from the recombinant host cell.
[0118] Methods for recovering polypeptides expressed from prokaryote and eukaryote cells are known in the art. In embodiments, the method for recovering the polypeptide comprises lysing the cells, e.g., by mechanical, chemical, or enzymatic means. For example, cells can be physically broken apart, e.g., by sonication, milling (shaking with beads), or shear forces. Cell membranes can be treated such that they are permeabilized such that the contents of the cells are released, such as treatment with detergents, e.g., Triton, NP-40, or SDS. Cells with cell walls, e.g., bacterial cells, can be permeabilized using enzymes, such as a lysozyme or lysonase. Any combination of the mechanical, chemical, and enzymatic techniques described above are also suitable for recovering expressed polypeptides of interest from the host cell in the context of this invention. For example, when expressing an aglycosylated polypeptide having cellobiase activity described herein in a bacterial cell, e.g., an E. coli cell, the cell is typically lysed by centrifuging and pelleting the cell culture, and resuspending in a lysis buffer containing lysozyme. To ensure complete lysis, the resuspended cells are subjected to one of the following methods: sonication, milling, or homogenization.
[0119] In one embodiment, the expressed aglycosylated polypeptides having cellobiase activity described herein are not lysed before addition to the biomass for the saccharification reaction. In some instances, the methods for lysing host cells can result in protein denaturation and/or decreased enzyme activity, which leads to increased cost of downstream processing. Thus, the present invention also provides methods for directly adding the host cells expressing an aglycosylated polypeptide having cellobiase activity described herein to the biomass prior to the saccharification step.
[0120] In an embodiment, the host cell, e.g., the E. coli cell, expressing the aglycosylated polypeptide having cellobiase activity described herein is isolated, e.g., by centrifugation, and added to the saccharification reaction, e.g., the saccharification reactor containing biomass. The cells are lysed by a combination of shear from the biomass, the impellers, and the increased temperature. In an embodiment, the culture of host cell, e.g., the E. coli cell, expressing the aglycosylated polypeptide having cellobiase activity described herein is added directly from the fermentation tank directly to the saccharification tank and eliminating the need to pellet cells by centrifugation.
[0121] Using a Host Cell Deficient for Glycosylation
[0122] In embodiments, the expression vector comprises a nucleic acid sequence encoding a polypeptide having cellobiase activity described herein operably linked to a fusion tag is introduced to and expressed in a cell that does not significantly glycosylate proteins expressed in the cell, e.g., a bacterial host cell. The recombinant host cell is cultured under conditions for expression of the polypeptide, resulting in the production of an aglycosylated polypeptide having cellobiase activity. The aglycosylated polypeptide can be purified or isolated from the host cell using affinity chromatography methods for the fusion tag as described herein.
[0123] For example, in this embodiment, the expression vector contains a lac operator and a T7 promoter upstream of the nucleic acid sequence encoding a polypeptide having cellobiase activity, and the host cell has the capacity to express T7 RNA polymerase. Expression of the polypeptide having cellobiase activity is induced by addition of IPTG. Preferably, the host cell is an E. coli cell, preferably an E. coli Origami cell. In this embodiment, the fusion tag is a His-tag, and the purification of the expressed aglycosylated polypeptide comprises nickel affinity chromatography.
[0124] Using a Host Cell with the Capacity for Glycosylation
[0125] In another embodiment, an expression vector comprising a nucleic acid sequence encoding a polypeptide comprising one or more glycosylation site mutations such that the polypeptide is not glyscosylated, as described herein, is expressed in a host cell, wherein the host cell is capable of glycosylating proteins expressed within the cell, e.g., a yeast or fungal host cell. Alternatively, the host cell is not capable of glycosylating proteins expressed within the cell, e.g., a bacterial host cell. In this embodiment, the polypeptide is operably linked to a fusion tag. The aglycosylated polypeptide can be purified or isolated from the bacterial host cell using affinity chromatography methods for the fusion tag as described herein.
[0126] In yet another embodiment, an expression vector comprising a nucleic acid sequence encoding a polypeptide having cellobiase activity described herein is expressed in a host cell, wherein the host cell is capable of glycosylating proteins expressed within the cell. The cells are cultured under conditions sufficient for expression and glycosylation of the polypeptide. In this embodiment, the polypeptide is operably linked to a fusion tag. The glycosylated polypeptide can be purified or isolated from the bacterial host cell using affinity chromatography methods for the fusion tag as described herein. After purification from the host cells and other endogenous host enzymes, e.g., glycosylation enzymes, the glycans of the isolated glycosylated polypeptide can be removed by incubation with deglycosylating enzymes. Deglycosylating enzymes include PNGase F, PNGase A, EndoH (endoglycosidase H), EndoS (endoglycosidase S), EndoD (endoglycosidase D), EndoF (endoglycosidase F), EndoF1 (endoglycosidase F1), or EndoF2 (endoglycosidase F2). Protein deglycosylation mixes containing enzymes sufficient for the complete removal of glycans are commercially available, e.g., from New England Biolabs. The isolated polypeptide is incubated with one or more deglycosylating enzyme under conditions sufficient for the removal of all of the glycans from the polypeptide. Other methods are known in the art for removing glycans from a polypeptide, e.g., .beta.-elimination with mild alkali or mild hydrazinolysis. Assessment of the glycosylation state of the polypeptide can be determined using methods for staining and visualization of glycans known in the art, or mass spectrometry.
[0127] In yet another embodiment, an expression vector comprising a nucleic acid sequence encoding a polypeptide having cellobiase activity described herein is expressed in a host cell, wherein the host cell is capable of glycosylating proteins expressed within the cell. The cells are cultured under conditions sufficient for expression of the polypeptide, but in the presence of glycosylation inhibitors. The glycosylation inhibitors are present at a concentration and for a sufficient time such that the expressed polypeptides are not glycosylated. In this embodiment, the polypeptide is operably linked to a fusion tag. The resulting aglycosylated polypeptide can be purified or isolated from the bacterial host cell using affinity chromatography methods for the fusion tag as described herein.
[0128] Examples of suitable glycosylation inhibitors for use in this embodiment include tunicamycin, Benzyl-GalNAc (Benzyl 2-acetamido-2-deoxy-.alpha.-D-galactopyranoside), 2-Fluoro-2-deoxy-D-glucose, and 5'CDP (5' cytidylate diphosphate). In some embodiments, a combination of glycosylation inhibitors is used. Preferably, the concentration of glycosylation inhibitors used in this embodiment is sufficient to inhibit glycosylation of the polypeptide, but do not cause cytotoxicity or inhibition of protein expression of the host cell.
Methods of Producing Products Using Aglycosylated Polypeptides
[0129] The present invention provides methods and compositions for converting or processing a biomass into products, using an aglycosylated polypeptide having cellobiase activity, as described herein. Methods for converting a biomass to products, such as sugar products, are known in the art, for example, as described in US Patent Application 2014/0011258, the contents of which are incorporated by reference in its entirety. Briefly, a biomass is optimally pretreated, e.g., to reduce the recalcitrance, and saccharified by a saccharification process that involves incubating the treated biomass with biomass-degrading, or cellulolytic, enzymes to produce sugars (e.g., glucose and/or xylose). The sugar products can then be further processed to produce a final product, e.g., by fermentation or distillation. Final products include alcohols (e.g., ethanol, isobutanol, or n-butanol), sugar alcohols (e.g., erythritol, xylitol, or sorbitol), or organic acids (e.g., lactic acid, pyurvic acid, succinic acid).
[0130] Using the processes described herein, the biomass material can be converted to one or more products, such as energy, fuels, foods and materials. Specific examples of products include, but are not limited to, hydrogen, sugars (e.g., glucose, xylose, arabinose, mannose, galactose, fructose, cellobiose, disaccharides, oligosaccharides and polysaccharides), alcohols (e.g., monohydric alcohols or dihydric alcohols, such as ethanol, n-propanol, isobutanol, sec-butanol, tert-butanol or n-butanol), hydrated or hydrous alcohols (e.g., containing greater than 10%, 20%, 30% or even greater than 40% water), biodiesel, organic acids, hydrocarbons (e.g., methane, ethane, propane, isobutene, pentane, n-hexane, biodiesel, bio-gasoline and mixtures thereof), co-products (e.g., proteins, such as cellulolytic proteins (enzymes) or single cell proteins), and mixtures of any of these in any combination or relative concentration, and optionally in combination with any additives (e.g., fuel additives). Other examples include carboxylic acids, salts of a carboxylic acid, a mixture of carboxylic acids and salts of carboxylic acids and esters of carboxylic acids (e.g., methyl, ethyl and n-propyl esters ketones (e.g., acetone), aldehydes (e.g., acetaldehyde), alpha and beta unsaturated acids (e.g., acrylic acid) and olefins (e.g., ethylene). Other alcohols and alcohol derivatives include propanol, propylene glycol, 1,4-butanediol, 1,3-propanediol, sugar alcohols and polyols (e.g., glycol, glycerol, erythritol, threitol, arabitol, xylitol, ribitol, mannitol, sorbitol, galactitol, iditol, inositol, volemitol, isomalt, maltitol, lactitol, maltotriitol, maltotetraitol, and polyglycitol and other polyols), and methyl or ethyl esters of any of these alcohols. Other products include methyl acrylate, methylmethacrylate, lactic acid, citric acid, formic acid, acetic acid, propionic acid, butyric acid, succinic acid, valeric acid, caproic acid, 3-hydroxypropionic acid, palmitic acid, stearic acid, oxalic acid, malonic acid, glutaric acid, oleic acid, linoleic acid, glycolic acid, gamma-hydroxybutric acid, and mixtures thereof, salts of any of these acids, mixtures of any of the acids and their respective salts.
[0131] Biomass
[0132] The biomass to be processed using the methods described herein is a starchy material and/or a cellulosic material comprising cellulose, e.g., a lignocellulosic material. The biomass may also comprise hemicellulose and/or lignin. The biomass can comprise one or more of an agricultural product or waste, a paper product or waste, a forestry product, or a general waste, or any combination thereof. An agricultural product or waste comprises material that can be cultivated, harvested, or processed for use or consumption, e.g., by humans or animals, or any intermediate, byproduct, or waste that is generated from the cultivation, harvest, or processing methods. Agricultural products or waste include, but are not limited to, sugar cane, jute, hemp, flax, bamboo, sisal, alfalfa, hay, arracacha, buckwheat, banana, barley, cassava, kudzu, oca, sago, sorghum, potato, sweet potato, taro, yams, beans, favas, lentils, peas, grasses, switchgrass, miscanthus, cord grass, reed canary grass, grain residues, canola straw, wheat straw, barley straw, oat straw, rice straw, corn cobs, corn stover, corn fiber, coconut hair, beet pulp, bagasse, soybean stover, grain residues, rice hulls, oat hulls, wheat chaff, barley hulls, or beeswing, or a combination thereof. A paper product or waste comprises material that is used to make a paper product, any paper product, or any intermediate, byproduct or waste that is generated from making or breaking down the paper product. Paper products or waste include, but are not limited to, paper, pigmented papers, loaded papers, coated papers, corrugated paper, filled papers, magazines, printed matter, printer paper, polycoated paper, cardstock, cardboard, paperboard, or paper pulp, or a combination thereof. A forestry product or waste comprises material that is produced by cultivating, harvesting, or processing of wood, or any intermediate, byproduct, or waste that is generated from the cultivation, harvest, or processing of the wood. Forestry products or waste include, but are not limited to, aspen wood, wood from any genus or species of tree, particle board, wood chips, or sawdust, or a combination thereof. A general waste includes, but is not limited to, manure, sewage, or offal, or a combination thereof.
[0133] In an embodiment, the biomass comprises agriculture waste, such as corn cobs, e.g., corn stover. In another embodiment, the biomass comprises grasses.
[0134] In one embodiment, the biomass is treated prior to contact with the compositions described herein. For example, the biomass is treated to reduce the recalcitrance of the biomass, to reduce its bulk density, and/or increase its surface area. Suitable biomass treatment process may include, but are not limited to: bombardment with electrons, sonication, oxidation, pyrolysis, steam explosion, chemical treatment, mechanical treatment, and freeze grinding. Preferably, the treatment method is bombardment with electrons.
[0135] In some embodiments, electron bombardment is performed until the biomass receives a total dose of at least 0.5 Mrad, e.g. at least 5, 10, 20, 30, or at least 40 Mrad. In some embodiments, the treatment is performed until the biomass receives a dose a of from about 0.5 Mrad to about 150 Mrad, about 1 Mrad to about 100 Mrad, about 5 Mrad to about 75 Mrad, about 2 Mrad to about 75 Mrad, about 10 Mrad to about 50 Mrad, e.g., about 5 Mrad to about 50 Mrad, about 20 Mrad to about 40 Mrad, about 10 Mrad to about 35 Mrad, or from about 20 Mrad to about 30 Mrad. In some implementations, a total dose of 25 to 35 Mrad is preferred, applied ideally over a couple of seconds, e.g., at 5 Mrad/pass with each pass being applied for about one second. Applying a dose of greater than 7 to 9 Mrad/pass can in some cases cause thermal degradation of the feedstock material.
[0136] The biomass material (e.g., plant biomass, animal biomass, paper, and municipal waste biomass) can be used as feedstock to produce useful intermediates and products such as organic acids, salts of organic acids, anhydrides, esters of organic acids and fuels, e.g., fuels for internal combustion engines or feedstocks for fuel cells. Systems and processes are described herein that can use as feedstock cellulosic and/or lignocellulosic materials that are readily available, but often can be difficult to process, e.g., municipal waste streams and waste paper streams, such as streams that include newspaper, kraft paper, corrugated paper or mixtures of these.
[0137] In order to convert the biomass to a form that can be readily processed, the glucan- or xylan-containing cellulose in the feedstock can be hydrolyzed to low molecular weight carbohydrates, such as sugars, by a saccharifying agent, e.g., an enzyme or acid, a process referred to as saccharification. The low molecular weight carbohydrates can then be used, for example, in an existing manufacturing plant, such as a single cell protein plant, an enzyme manufacturing plant, or a fuel plant, e.g., an ethanol manufacturing facility.
[0138] The biomass can be hydrolyzed using an enzyme, e.g., a biomass degrading enzyme, by combining the materials and the enzyme in a solvent, e.g., in an aqueous solution. The enzymes can be made/induced according to the methods described herein.
[0139] Specifically, the biomass degrading enzyme can be supplied by organisms, e.g., a microorganism, that are capable of breaking down biomass (such as the cellulose and/or the lignin portions of the biomass), or that contain or manufacture various cellulolytic enzymes (cellulases), ligninases or various small molecule biomass-degrading metabolites. These enzymes may be a complex of enzymes that act synergistically to degrade crystalline cellulose or the lignin portions of biomass. Examples of cellulolytic enzymes include: endoglucanases, cellobiohydrolases, and cellobiases (beta-glucosidases).
[0140] During saccharification a cellulosic substrate can be initially hydrolyzed by endoglucanases at random locations producing oligomeric intermediates. These intermediates are then substrates for exo-splitting glucanases such as cellobiohydrolase to produce cellobiose from the ends of the cellulose polymer. Cellobiose is a water-soluble 1,4-linked dimer of glucose. Finally, cellobiase cleaves cellobiose to yield glucose. The efficiency (e.g., time to hydrolyze and/or completeness of hydrolysis) of this process depends on the recalcitrance of the cellulosic material.
[0141] Saccharification
[0142] The reduced-recalcitrance biomass is treated with the biomass-degrading enzymes discussed above, generally by combining the reduced-recalcitrance biomass and the biomass-degrading enzymes in a fluid medium, e.g., an aqueous solution. In some cases, the feedstock is boiled, steeped, or cooked in hot water prior to saccharification, as described in U.S. Pat. App. Pub. 2012/0100577 A1 by Medoff and Masterman, published on Apr. 26, 2012, the entire contents of which are incorporated herein.
[0143] Provided herein are mixtures of enzymes that are capable of degrading the biomass, e.g., an enzyme mixture of biomass-degrading enzymes, for use in the saccharification process described herein.
[0144] The saccharification process can be partially or completely performed in a tank (e.g., a tank having a volume of at least 4000, 40,000, or 500,000 L) in a manufacturing plant, and/or can be partially or completely performed in transit, e.g., in a rail car, tanker truck, or in a supertanker or the hold of a ship. The time required for complete saccharification will depend on the process conditions and the biomass material and enzyme used. If saccharification is performed in a manufacturing plant under controlled conditions, the cellulose may be substantially entirely converted to sugar, e.g., glucose in about 12-96 hours. If saccharification is performed partially or completely in transit, saccharification may take longer.
[0145] In a preferred embodiment, the saccharification reaction occurs at a pH optimal for the enzymatic reactions to occur, e.g., at the pH optimal for the activity of the biomass-degrading enzymes. Preferably, the pH of the saccharification reaction is at pH 4-4.5. In a preferred embodiment, the saccharification reaction occurs at a temperature optimal for the enzymatic reactions to occur, e.g., at the temperature optimal for the activity of the biomass-degrading enzymes. Preferably, the temperature of the saccharification reaction is at 42.degree. C.-52.degree. C.
[0146] It is generally preferred that the tank contents be mixed during saccharification, e.g., using jet mixing as described in International App. No. PCT/US2010/035331, filed May 18, 2010, which was published in English as WO 2010/135380 and designated the United States, the full disclosure of which is incorporated by reference herein.
[0147] The addition of surfactants can enhance the rate of saccharification. Examples of surfactants include non-ionic surfactants, such as a Tween.RTM. 20 or Tween.RTM. 80 polyethylene glycol surfactants, ionic surfactants, or amphoteric surfactants.
[0148] It is generally preferred that the concentration of the sugar solution resulting from saccharification be relatively high, e.g., greater than 40%, or greater than 50, 60, 70, 80, 90 or even greater than 95% by weight. Water may be removed, e.g., by evaporation, to increase the concentration of the sugar solution. This reduces the volume to be shipped, and also inhibits microbial growth in the solution.
[0149] Alternatively, sugar solutions of lower concentrations may be used, in which case it may be desirable to add an antimicrobial additive, e.g., a broad spectrum antibiotic, in a low concentration, e.g., 50 to 150 ppm. Other suitable antibiotics include amphotericin B, ampicillin, chloramphenicol, ciprofloxacin, gentamicin, hygromycin B, kanamycin, neomycin, penicillin, puromycin, streptomycin. Antibiotics will inhibit growth of microorganisms during transport and storage, and can be used at appropriate concentrations, e.g., between 15 and 1000 ppm by weight, e.g., between 25 and 500 ppm, or between 50 and 150 ppm. If desired, an antibiotic can be included even if the sugar concentration is relatively high. Alternatively, other additives with anti-microbial of preservative properties may be used. Preferably the antimicrobial additive(s) are food-grade.
[0150] A relatively high concentration solution can be obtained by limiting the amount of water added to the biomass material with the enzyme. The concentration can be controlled, e.g., by controlling how much saccharification takes place. For example, concentration can be increased by adding more biomass material to the solution. In order to keep the sugar that is being produced in solution, a surfactant can be added, e.g., one of those discussed above. Solubility can also be increased by increasing the temperature of the solution. For example, the solution can be maintained at a temperature of 40-50.degree. C., 60-80.degree. C., or even higher.
[0151] In the processes described herein, for example after saccharification, sugars (e.g., glucose and xylose) can be isolated. For example, sugars can be isolated by precipitation, crystallization, chromatography (e.g., simulated moving bed chromatography, high pressure chromatography), centrifugation, extraction,any other isolation method known in the art, and combinations thereof.
[0152] Enzyme Mixtures for Saccharification
[0153] The present invention provides an enzyme mixture comprising a glycosylated polypeptide comprising at least 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 1, and an aglycosylated polypeptide comprising at least 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 1, wherein both the glycosylated polypeptide and the aglyscosylated polypeptide have cellobiase activity. The aglycosylated polypeptide having cellobiase activity is any of the aglycosylated polypeptides described herein, e.g., produced using the methods described herein. The glycosylated polypeptide comprising at least 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% identity to SEQ ID NO: 1 can be isolated or obtained from a microorganism that endogenously expresses the polypeptide.
[0154] In an embodiment, the glysocylated polypeptide and the aglycosylated polypeptide are both the Cel3A enzyme from wild-type T. reesei, e.g., comprising SEQ ID NO: 1.
[0155] In embodiments, the enzyme mixture further comprises at least one additional enzyme derived from a microorganism, wherein the additional enzyme has biomass or cellulose-based material-degrading activity, e.g., the additional enzyme is a cellulolytic enzyme, e.g., a cellulase. For example, the additional enzyme is a ligninase, an endoglucanase, a cellobiohydrolase, a xylanase, and a cellobiase. In an embodiment, the mixture further comprises one or more ligninase, one or more endogluconase, one or more cellobiohydrolase, and one or more xylanase. In embodiments, the additional biomass-degrading enzyme is glycosylated. In embodiments, the enzyme mixture further comprises at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, or at least 10 additional biomass-degrading enzymes described herein. For example, the enzyme mixture further comprises at least 1, at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, at least 15 or all of the enzymes listed in Table 1.
[0156] For example, the enzyme mixture further comprises a mixture of additional biomass-degrading enzymes produced by a microorganism, e.g., a fungal cell, such as wild-type T. reesei, or a mutant thereof, e.g., T. Reesei RUTC30. In an embodiment, the additional biomass-degrading enzymes are isolated from the microorganisms. In an embodiment, the mixture comprises one or more of the following biomass-degrading enzymes: B2AF03, CIP1, CIP2, Cel1a, Cel3a, Cel5a, Cel6a, Cel7a, Cel7b, Cel12a, Cel45a, Cel74a, paMan5a, paMan26a, or Swollenin, or any combination thereof. The additional biomass-degrading enzymes, e.g., listed above, can be endogenously expressed and isolated from the microorganism, e.g., fungal cell, from which the enzyme originates from (listed below in Table 1). Alternatively, the additional biomass-degrading enzymes, e.g., listed above, can be heterologously expressed using similar methods of expression in a host cell described herein, and isolated from the host cells. In an embodiment, the heterologously expressed additional biomass-degrading enzymes are tagged with a His tag at the C or N terminus of the enzyme and are isolated using nickel affinity chromatography techniques known in the art. For example, the additional biomass-degrading enzymes can be selected from Table 1 below.
TABLE-US-00004 TABLE 1 Examples of Additional Biomass-Degrading Enzymes Protein MW, kDa no AA's th. pl no. Cysteines Organism B2AF03-C'His 88.6 813 6.3 10 Podospora anserina CIP1-C'His 32.5 311 5.6 8 Trichoderma reesei CIP2-C'His 48.0 457 7.0 12 Trichoderma reesei Cel1a-C'His 53.6 478 5.8 5 Trichoderma reesei Cel3a-C'His 78.0 739 6.3 6 Trichoderma reesei Cel3a-N'His 78.0 739 6.3 6 Trichoderma reesei Cel5a-N'His 43.7 411 5.7 12 Trichoderma reesei Cel6a-C'His 48.8 461 6.0 12 Trichoderma reesei Cel7a-C'His 53.8 511 4.8 24 Trichoderma reesei Cel7b-C'His 47.6 451 5.3 22 Trichoderma reesei Cel12a-C'His 25.1 232 6.9 2 Trichoderma reesei Cel45a-C'His 24.4 239 5.4 16 Trichoderma reesei Cel74a-C'His 86.7 832 5.7 4 Trichoderma reesei paMan5a-C'His 41.0 370 7.0 6 Podospora anserina paMan26a-C'His 51.4 463 5.2 1 Podospora anserina Swollenin-N'His 51.5 491 5.3 28 Trichoderma reesei
[0157] The amino acid sequences for the biomass-degrading enzymes listed in Table 1 are provided below.
TABLE-US-00005 B2AF03 (Podospora anserina) (SEQ ID NO: 9) MKSSVFWGASLTSAVVRAIDLPFQFYPNCVDDLLSTNQVCNTTLSPPERAAALVAALTPEE KLQNIVSKSLGAPRIGLPAYNWWSEALHGVAYAPGTQFWQGDGPFNSSTSFPMPLLMAATFDDELLEKI AEVIGIEGRAFGNAGFSGLDYWTPNVNPFKDPRWGRGSETPGEDVLLVKRYAAAMIKGLEGPVPEKERR VVATCKHYAANDFEDWNGATRHNFNAKISLQDMAEYYFMPFQQCVRDSRVGSIMCAYNAVNGVPSCASP YLLQTILREHWNWTEHNNYITSDCEAVLDVSLNHKYAATNAEGTAISFEAGMDTSCEYEGSSDIPGAWS QGLLKESTVDRALLRLYEGIVRAGYFDGKQSLYSSLGWADVNKPSAQKLSLQAAVDGTVLLKNDGTLPL SDLLDKSRPKKVAMIGFWSDAKDKLRGGYSGTAAYLHTPAYAASQLGIPFSTASGPILHSDLASNQSWT DNAMAAAKDADYILYFGGIDTSAAGETKDRYDLDWPGAQLSLINLLTTLSKPLIVLQMGDQLDNTPLLS NPKINAILWANWPGQDGGTAVMELVTGLKSPAGRLPVTQYPSNFTELVPMTDMALRPSAGNSQLGRTYR WYKTPVQAFGFGLHYTTFSPKFGKKFPAVIDVDEVLEGCDDKYLDTCPLPDLPVVVENRGNRTSDYVAL AFVSAPGVGPGPWPIKTLGAFTRLRGVKGGEKREGGLKWNLGNLARHDEEGNTVVYPGKYEVSLDEPPK ARLRFEIVRGGKGKGKVKGKGKAAQKGGVVLDRWPKPPKGQEPPAIERV CIP1 (Trichoderma reesei) (SEQ ID NO: 10) MVRRTALLALGALSTLSMAQISDDFESGWDQTKWPISAPDCNQGGTVSLDTTVAHSGSNSM KVVGGPNGYCGHIFFGTTQVPTGDVYVRAWIRLQTALGSNHVTFIIMPDTAQGGKHLRIGGQSQVLDYN RESDDATLPDLSPNGIASTVTLPTGAFQCFEYHLGTDGTIETWLNGSLIPGMTVGPGVDNPNDAGWTRA SYIPEITGVNFGWEAYSGDVNTVWFDDISIASTRVGCGPGSPGGPGSSTTGRSSTSGPTSTSRPSTTIP PPTSRTTTATGPTQTHYGQCGGIGYSGPTVCASGTTCQVLNPYYSQCL CIP2 (Trichoderma reesei) (SEQ ID NO: 11) MASRFFALLLLAIPIQAQSPVWGQCGGIGWSGPTTCVGGATCVSYNPYYSQCIPSTQASSS IASTTLVTSFTTTTATRTSASTPPASSTGAGGATCSALPGSITLRSNAKLNDLFTMFNGDKVTTKDKFS CRQAEMSELIQRYELGTLPGRPSTLTASFSGNTLTINCGEAGKSISFTVTITYPSSGTAPYPAIIGYGG GSLPAPAGVAMINFNNDNIAAQVNTGSRGQGKFYDLYGSSHSAGAMTAWAWGVSRVIDALELVPGARID TTKIGVTGCSRNGKGAMVAGAFEKRIVLTLPQESGAGGSACWRISDYLKSQGANIQTASEIIGEDPWFS TTFNSYVNQVPVLPFDHHSLAALIAPRGLFVIDNNIDWLGPQSCFGCMTAAHMAWQALGVSDHMGYSQI GAHAHCAFPSNQQSQLTAFVQKFLLGQSTNTAIFQSDFSANQSQWIDWTTPTLS Cel1a (Trichoderma reesei) (SEQ ID NO: 12) MLPKDFQWGFATAAYQIEGAVDQDGRGPSIWDTFCAQPGKIADGSSGVTACDSYNRTAEDI ALLKSLGAKSYRFSISWSRIIPEGGRGDAVNQAGIDHYVKFVDDLLDAGITPFITLFHWDLPEGLHQRY GGLLNRTEFPLDFENYARVMFRALPKVRNWITFNEPLCSAIPGYGSGTFAPGRQSTSEPWTVGHNILVA HGRAVKAYRDDFKPASGDGQIGIVLNGDFTYPWDAADPADKEAAERRLEFFTAWFADPIYLGDYPASMR KQLGDRLPTFTPEERALVHGSNDFYGMNHYTSNYIRHRSSPASADDTVGNVDVLFTNKQGNCIGPETQS PWLRPCAAGFRDFLVWISKRYGYPPIYVTENGTSIKGESDLPKEKILEDDFRVKYYNEYIRAMVTAVEL DGVNVKGYFAWSLMDNFEWADGYVTRFGVTYVDYENGQKRFPKKSAKSLKPLFDELIAAA Cel3a (Trichoderma reesei) (SEQ ID NO: 13) MRYRTAAALALATGPFARADSHSTSGASAEAVVPPAGTPWGTAYDKAKAALAKLNLQDKVG IVSGVGWNGGPCVGNTSPASKISYPSLCLQDGPLGVRYSTGSTAFTPGVQAASTWDVNLIRERGQFIGE EVKASGIHVILGPVAGPLGKTPQGGRNWEGFGVDPYLTGIAMGQTINGIQSVGVQATAKHYILNEQELN RETISSNPDDRTLHELYTWPFADAVQANVASVMCSYNKVNTTWACEDQYTLQTVLKDQLGFPGYVMTDW NAQHTTVQSANSGLDMSMPGTDFNGNNRLWGPALTNAVNSNQVPTSRVDDMVTRILAAWYLTGQDQAGY PSFNISRNVQGNHKTNVRAIARDGIVLLKNDANILPLKKPASIAVVGSAAIIGNHARNSPSCNDKGCDD GALGMGWGSGAVNYPYFVAPYDAINTRASSQGTQVTLSNTDNTSSGASAARGKDVAIVFITADSGEGYI TVEGNAGDRNNLDPWHNGNALVQAVAGANSNVIVVVHSVGAIILEQILALPQVKAVVWAGLPSQESGNA LVDVLWGDVSPSGKLVYTIAKSPNDYNTRIVSGGSDSFSEGLFIDYKHFDDANITPRYEFGYGLSYTKF NYSRLSVLSTAKSGPATGAVVPGGPSDLFQNVATVTVDIANSGQVTGAEVAQLYITYPSSAPRTPPKQL RGFAKLNLTPGQSGTATFNIRRRDLSYWDTASQKWVVPSGSFGISVGASSRDIRLTSTLSVA Cel5a (Trichoderma reesei) (SEQ ID NO: 14) MNKSVAPLLLAASILYGGAAAQQTVWGQCGGIGWSGPTNCAPGSACSTLNPYYAQCIPGAT TITTSTRPPSGPTTTTRATSTSSSTPPTSSGVRFAGVNIAGFDFGCTTDGTCVTSKVYPPLKNFTGSNN YPDGIGQMQHFVNDDGMTIFRLPVGWQYLVNNNLGGNLDSTSISKYDQLVQGCLSLGAYCIVDIHNYAR WNGGIIGQGGPTNAQFTSLWSQLASKYASQSRVWFGIMNEPHDVNINTWAATVQEVVTAIRNAGATSQF ISLPGNDWQSAGAFISDGSAAALSQVTNPDGSTTNLIFDVHKYLDSDNSGTHAECTTNNIDGAFSPLAT WLRQNNRQAILTETGGGNVQSCIQDMCQQIQYLNQNSDVYLGYVGWGAGSFDSTYVLTETPTGSGNSWT DTSLVSSCLARK Cel6a (Trichoderma reesei) (SEQ ID NO: 15) MIVGILTTLATLATLAASVPLEERQACSSVWGQCGGQNWSGPTCCASGSTCVYSNDYYSQC LPGAASSSSSTRAASTTSRVSPTTSRSSSATPPPGSTTTRVPPVGSGTATYSGNPFVGVTPWANAYYAS EVSSLAIPSLTGAMATAAAAVAKVPSFMWLDTLDKTPLMEQTLADIRTANKNGGNYAGQFVVYDLPDRD CAALASNGEYSIADGGVAKYKNYIDTIRQIVVEYSDIRTLLVIEPDSLANLVTNLGTPKCANAQSAYLE CINYAVTQLNLPNVAMYLDAGHAGWLGWPANQDPAAQLFANVYKNASSPRALRGLATNVANYNGWNITS PPSYTQGNAVYNEKLYIHAIGPLLANHGWSNAFFITDQGRSGKQPTGQQQWGDWCNVIGTGFGIRPSAN TGDSLLDSFVWVKPGGECDGTSDSSAPRFDSHCALPDALQPAPQAGAWFQAYFVQLLTNANPSFL Cel7a(Trichoderma reesei) (SEQ ID NO: 16) MYRKLAVISAFLATARAQSACTLQSETHPPLTWQKCSSGGTCTQQTGSVVIDANWRWTHAT NSSTNCYDGNTWSSTLCPDNETCAKNCCLDGAAYASTYGVTTSGNSLSIGFVTQSAQKNVGARLYLMAS DTTYQEFTLLGNEFSFDVDVSQLPCGLNGALYFVSMDADGGVSKYPTNTAGAKYGTGYCDSQCPRDLKF INGQANVEGWEPSSNNANTGIGGHGSCCSEMDIWEANSISEALTPHPCTTVGQEICEGDGCGGTYSDNR YGGTCDPDGCDWNPYRLGNTSFYGPGSSFTLDTTKKLTVVTQFETSGAINRYYVQNGVTFQQPNAELGS YSGNELNDDYCTAEEAEFGGSSFSDKGGLTQFKKATSGGMVLVMSLWDDYYANMLWLDSTYPTNETSST PGAVRGSCSTSSGVPAQVESQSPNAKVTFSNIKFGPIGSTGNPSGGNPPGGNPPGTTTTRRPATTTGSS PGPTQSHYGQCGGIGYSGPTVCASGTTCQVLNPYYSQCL Cel7b (Trichoderma reesei) (SEQ ID NO: 17) MAPSVTLPLTTAILAIARLVAAQQPGTSTPEVHPKLTTYKCTKSGGCVAQDTSVVLDWNYR WMHDANYNSCTVNGGVNTTLCPDEATCGKNCFIEGVDYAASGVTTSGSSLTMNQYMPSSSGGYSSVSPR LYLLDSDGEYVMLKLNGQELSFDVDLSALPCGENGSLYLSQMDENGGANQYNTAGANYGSGYCDAQCPV QTWRNGTLNTSHQGFCCNEMDILEGNSRANALTPHSCTATACDSAGCGFNPYGSGYKSYYGPGDTVDTS KTFTIITQFNTDNGSPSGNLVSITRKYQQNGVDIPSAQPGGDTISSCPSASAYGGLATMGKALSSGMVL VFSIWNDNSQYMNWLDSGNAGPCSSTEGNPSNILANNPNTHVVFSNIRWGDIGSTTNSTAPPPPPASST TFSTTRRSSTTSSSPSCTQTHWGQCGGIGYSGCKTCTSGTTCQYSNDYYSQCL Cel12a (Trichoderma reesei) (SEQ ID NO: 18) MKFLQVLPALIPAALAQTSCDQWATFTGNGYTVSNNLWGASAGSGFGCVTAVSLSGGASWH ADWQWSGGQNNVKSYQNSQIAIPQKRTVNSISSMPTTASWSYSGSNIRANVAYDLFTAANPNHVTYSGD YELMIWLGKYGDIGPIGSSQGTVNVGGQSWTLYYGYNGAMQVYSFVAQTNTTNYSGDVKNFFNYLRDNK GYNAAGQYVLSYQFGTEPFTGSGTLNVASWTASIN Cel45a (Trichoderma reesei) (SEQ ID NO: 19) MKATLVLGSLIVGAVSAYKATTTRYYDGQEGACGCGSSSGAFPWQLGIGNGVYTAAGSQAL FDTAGASWCGAGCGKCYQLTSTGQAPCSSCGTGGAAGQSIIVMVTNLCPNNGNAQWCPVVGGTNQYGYS YHFDIMAQNEIFGDNVVVDFEPIACPGQAASDWGTCLCVGQQETDPTPVLGNDTGSTPPGSSPPATSSS PPSGGGQQTLYGQCGGAGWTGPTTCQAPGTCKVQNQWYSQCLP Cel74a (Trichoderma reesei) (SEQ ID NO: 20) MKVSRVLALVLGAVIPAHAAFSWKNVKLGGGGGFVPGIIFHPKTKGVAYARTDIGGLYRLN ADDSWTAVTDGIADNAGWHNWGIDAVALDPQDDQKVYAAVGMYTNSWDPSNGAIIRSSDRGATWSFTNL PFKVGGNMPGRGAGERLAVDPANSNIIYFGARSGNGLWKSTDGGVTFSKVSSFTATGTYIPDPSDSNGY NSDKQGLMWVTFDSTSSTTGGATSRIFVGTADNITASVYVSTNAGSTWSAVPGQPGKYFPHKAKLQPAE KALYLTYSDGTGPYDGTLGSVWRYDIAGGTWKDITPVSGSDLYFGFGGLGLDLQKPGTLVVASLNSWWP DAQLFRSTDSGTTWSPIWAWASYPTETYYYSISTPKAPWIKNNFIDVTSESPSDGLIKRLGWMIESLEI DPTDSNHWLYGTGMTIFGGHDLTNWDTRHNVSIQSLADGIEEFSVQDLASAPGGSELLAAVGDDNGFTF ASRNDLGTSPQTVWATPTWATSTSVDYAGNSVKSVVRVGNTAGTQQVAISSDGGATWSIDYAADTSMNG GTVAYSADGDTILWSTASSGVQRSQFQGSFASVSSLPAGAVIASDKKTNSVFYAGSGSTFYVSKDTGSS FTRGPKLGSAGTIRDIAAHPTTAGTLYVSTDVGIFRSTDSGTTFGQVSTALTNTYQIALGVGSGSNWNL YAFGTGPSGARLYASGDSGASWTDIQGSQGFGSIDSTKVAGSGSTAGQVYVGTNGRGVFYAQGTVGGGT GGTSSSTKQSSSSTSSASSSTTLRSSVVSTTRASTVTSSRTSSAAGPTGSGVAGHYAQCGGIGWTGPTQ CVAPYVCQKQNDYYYQCV paMan5a (Podospora anserina) (SEQ ID NO: 21) MKGLFAFGLGLLSLVNALPQAQGGGAAASAKVSGTRFVIDGKTGYFAGTNSYWIGFLTNNR DVDTTLDHIASSGLKILRVWGFNDVNNQPSGNTVWFQRLASSGSQINTGPNGLQRLDYLVRSAETRGIK LIIALVNYWDDFGGMKAYVNAFGGTKESWYTNARAQEQYKRYIQAVVSRYVNSPAIFAWELANEPRCKG CNTNVIFNWATQISDYIRSLDKDHLITLGDEGFGLPGQTTYPYQYGEGTDFVKNLQIKNLDFGTFHMYP GHWGVPTSFGPGWIKDHAAACRAAGKPCLLEEYGYESDRCNVQKGWQQASRELSRDGMSGDLFWQWGDQ LSTGQTHNDGFTIYYGSSLATCLVTDHVRAINALPA paMan26a (Podospora anserina) (SEQ ID NO: 22) MVKLLDIGLFALALASSAVAKPCKPRDGPVTYEAEDAILTGTTVDTAQVGYTGRGYVTGFD EGSDKITFQISSATTKLYDLSIRYAAIYGDKRTNVVLNNGAVSEVFFPAGDSFTSVAAGQVLLNAGQNT IDIVNNWGWYLIDSITLTPSAPRPPHDINPNLNNPNADTNAKKLYSYLRSVYGNKIISGQQELHHAEWI RQQTGKTPALVAVDLMDYSPSRVERGTTSHAVEDAIAHHNAGGIVSVLWHWNAPVGLYDTEENKWWSGF YTRATDFDIAATLANPQGANYTLLIRDIDAIAVQLKRLEAAGVPVLWRPLHEAEGGWFWWGAKGPEPAK QLWDILYERLTVHHGLDNLIWVWNSILEDWYPGDDTVDILSADVYAQGNGPMSTQYNELIALGRDKKMI AAAEVGAAPLPGLLQAYQANWLWFAVWGDDFINNPSWNTVAVLNEIYNSDYVLTLDEIQGWRS Swollenin (Trichoderma reesei) (SEQ ID NO: 23) MAGKLILVALASLVSLSIQQNCAALFGQCGGIGWSGTTCCVAGAQCSFVNDWYSQCLASTGGNPPNGTT SSSLVSRTSSASSSVGSSSPGGNSPTGSASTYTTTDTATVAPHSQSPYPSIAASSCGSWTLVDNVCCPS YCANDDTSESCSGCGTCTTPPSADCKSGTMYPEVHHVSSNESWHYSRSTHFGLTSGGACGFGLYGLCTK GSVTASWTDPMLGATCDAFCTAYPLLCKDPTGTTLRGNFAAPNGDYYTQFWSSLPGALDNYLSCGECIE LIQTKPDGTDYAVGEAGYTDPITLEIVDSCPCSANSKWCCGPGADHCGEIDFKYGCPLPADSIHLDLSD
IAMGRLQGNGSLTNGVIPTRYRRVQCPKVGNAYIWLRNGGGPYYFALTAVNTNGPGSVTKIEIKGADTD NWVALVHDPNYTSSRPQERYGSWVIPQGSGPFNLPVGIRLTSPTGEQIVNEQAIKTFTPPATGDPNFYY IDIGVQFSQN
[0158] The ratio between the aglycosylated polypeptide described herein to the other biomass-degrading enzymes in the mixture can be, for example, at least 1:1; 1:2; 1:4; 1:8; 1:16; 1:32; 1:50; 1:75; 1:100, 1:150; 1:200; 1:300, 1:400 or 1:500. In an embodiment, the ratio of aglycosylated polypeptide described herein to the other enzymes in the mixture is 1:32. The ratio between the aglycosylated polypeptide described herein to the glycosylated polypeptide is, for example, at least 1:1; 1:2; 1:4; 1:8; 1:16; 1:32; 1:50; 1:75; 1:100, 1:150; 1:200; 1:300, 1:400, or 1:500.
[0159] Other examples of suitable biomass-degrading enzymes for use in the enzyme mixture of the present invention include the enzymes from species in the genera Bacillus, Coprinus, Myceliophthora, Cephalosporium, Scytalidium, Penicillium, Aspergillus, Pseudomonas, Humicola, Fusarium, Thielavia, Acremonium, Chrysosporium and Trichoderma, especially those produced by a strain selected from the species Aspergillus (see, e.g., EP Pub. No. 0 458 162), Humicola insolens (reclassified as Scytalidium thermophilum, see, e.g., U.S. Pat. No. 4,435,307), Coprinus cinereus, Fusarium oxysporum, Myceliophthora thermophila, Meripilus giganteus, Thielavia terrestris, Acremonium sp. (including, but not limited to, A. persicinum, A. acremonium, A. brachypenium, A. dichromosporum, A. obclavatum, A. pinkertoniae, A. roseogriseum, A. incoloratum, and A. furatum). Preferred strains include Humicola insolens DSM 1800, Fusarium oxysporum DSM 2672, Myceliophthora thermophila CBS 117.65, Cephalosporium sp. RYM-202, Acremonium sp. CBS 478.94, Acremonium sp. CBS 265.95, Acremonium persicinum CBS 169.65, Acremonium acremonium AHU 9519, Cephalosporium sp. CBS 535.71, Acremonium brachypenium CBS 866.73, Acremonium dichromosporum CBS 683.73, Acremonium obclavatum CBS 311.74, Acremonium pinkertoniae CBS 157.70, Acremonium roseogriseum CBS 134.56, Acremonium incoloratum CBS 146.62, and Acremonium furatum CBS 299.70H. Biomass-degrading enzymes may also be obtained from Chrysosporium, preferably a strain of Chrysosporium lucknowense. Additional strains that can be used include, but are not limited to, Trichoderma (particularly T. viride, T. reesei, and T. koningii), alkalophilic Bacillus (see, for example, U.S. Pat. No. 3,844,890 and EP Pub. No. 0 458 162), and Streptomyces (see, e.g., EP Pub. No. 0 458 162).
[0160] In embodiments, the microorganism is induced to produce the biomass-degrading enzymes described herein under conditions suitable for increasing production of biomass-degrading enzymes compared to an uninduced microorganism. For example, an induction biomass sample comprising biomass as described herein is incubated with the microorganism to increase production of the biomass-degrading enzymes. Further description of the induction process can be found in US 2014/0011258, the contents of which are hereby incorporated by reference in its entirety.
[0161] The biomass-degrading enzymes produced and/or secreted by the aforementioned microorganisms can be isolated and added to the enzyme mixture of the present invention. Alternatively, in one embodiment, the aforementioned microorganisms or host cells expressing the biomass-degrading enzymes described herein and above are not lysed before addition to the saccharification reaction.
[0162] In an embodiment, the enzyme mixture comprises the host cell expressing an aglycosylated polypeptide having cellobiase activity as described herein, and one or more additional biomass-degrading enzymes described herein. In an embodiment, the enzyme mixture comprises a host cell expressing an aglycosylated polypeptide having cellobiase activity as described herein, and one or more host cells expressing one or more additional biomass-degrading enzymes described herein. For example,
[0163] Use of the enzyme mixture described herein comprising an aglycosylated polypeptide having cellobiase activity results in increased yield of sugar products from saccharification compared to the yield of sugar products from saccharification using the standard mixture of biomass-degrading enzymes (e.g., RUTC30 cocktail, e.g., without addition of an aglycosylated polypeptide having cellobiase activity). The yield of sugar products increases at least 5%, at least 10%, at least 15%, at least 20%, at least 25%, at least 30%, at least 35%, at least 40%, at least 45%, at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, at least 100% when an aglycosylated polypeptide having cellobiase activity is added to the saccharification process.
[0164] Further Processing
[0165] Further processing steps may be performed on the sugars produced by saccharification to produce alternative products. For example, the sugars can be hydrogenated, fermented, or treated with other chemicals to produce other products.
[0166] Glucose can be hydrogenated to sorbitol. Xylose can be hydrogenated to xylitol. Hydrogenation can be accomplished by use of a catalyst (e.g., Pt/gamma-Al.sub.2O.sub.3, Ru/C, Raney Nickel, or other catalysts know in the art) in combination with H.sub.2 under high pressure (e.g., 10 to 12000 psi). The sorbitol and/or xylitol products can be isolated and purified using methods known in the art.
[0167] Sugar products from saccharification can also be fermented to produce alcohols, sugar alcohols, such as erythritol, or organic acids, e.g., lactic, glutamic or citric acids or amino acids.
[0168] Yeast and Zymomonas bacteria, for example, can be used for fermentation or conversion of sugar(s) to alcohol(s). Other microorganisms are discussed below. The optimum pH for fermentations is about pH 4 to 7. For example, the optimum pH for yeast is from about pH 4 to 5, while the optimum pH for Zymomonas is from about pH 5 to 6. Typical fermentation times are about 24 to 168 hours (e.g., 24 to 96 hrs) with temperatures in the range of 20.degree. C. to 40.degree. C. (e.g., 26.degree. C. to 40.degree. C.), however thermophilic microorganisms prefer higher temperatures.
[0169] In some embodiments, e.g., when anaerobic organisms are used, at least a portion of the fermentation is conducted in the absence of oxygen, e.g., under a blanket of an inert gas such as N.sub.2, Ar, He, CO.sub.2 or mixtures thereof. Additionally, the mixture may have a constant purge of an inert gas flowing through the tank during part of or all of the fermentation. In some cases, anaerobic conditions can be achieved or maintained by carbon dioxide production during the fermentation and no additional inert gas is needed.
[0170] In some embodiments, all or a portion of the fermentation process can be interrupted before the low molecular weight sugar is completely converted to a product (e.g., ethanol). The intermediate fermentation products include sugar and carbohydrates in high concentrations. The sugars and carbohydrates can be isolated via any means known in the art. These intermediate fermentation products can be used in preparation of food for human or animal consumption. Additionally or alternatively, the intermediate fermentation products can be ground to a fine particle size in a stainless-steel laboratory mill to produce a flour-like substance.
[0171] Jet mixing may be used during fermentation, and in some cases saccharification and fermentation are performed in the same tank.
[0172] Nutrients for the microorganisms may be added during saccharification and/or fermentation, for example the food-based nutrient packages described in U.S. Pat. App. Pub. 2012/0052536, filed Jul. 15, 2011, the complete disclosure of which is incorporated herein by reference.
[0173] "Fermentation" includes the methods and products that are disclosed in U.S. Prov. App. No. 61/579,559, filed Dec. 22, 2012, and U.S. Prov. App. No. 61/579,576, filed Dec. 22, 2012, the contents of both of which are incorporated by reference herein in their entirety.
[0174] Mobile fermenters can be utilized, as described in International App. No. PCT/US2007/074028 (which was filed Jul. 20, 2007, was published in English as WO 2008/011598 and designated the United States), the contents of which is incorporated herein in its entirety. Similarly, the saccharification equipment can be mobile. Further, saccharification and/or fermentation may be performed in part or entirely during transit.
[0175] The microorganism(s) used in fermentation can be naturally-occurring microorganisms and/or engineered microorganisms. For example, the microorganism can be a bacterium (including, but not limited to, e.g., a cellulolytic bacterium), a fungus, (including, but not limited to, e.g., a yeast), a plant, a protist, e.g., a protozoa or a fungus-like protest (including, but not limited to, e.g., a slime mold), or an algae. When the organisms are compatible, mixtures of organisms can be utilized.
[0176] Suitable fermenting microorganisms have the ability to convert carbohydrates, such as glucose, fructose, xylose, arabinose, mannose, galactose, oligosaccharides or polysaccharides into fermentation products. Fermenting microorganisms include strains of the genus Saccharomyces spp. (including, but not limited to, S. cerevisiae (baker's yeast), S. distaticus, S. uvarum), the genus Kluyveromyces, (including, but not limited to, K. marxianus, K. fragilis), the genus Candida (including, but not limited to, C. pseudotropicalis, and C. brassicae), Pichia stipitis (a relative of Candida shehatae), the genus Clavispora (including, but not limited to, C. lusitaniae and C. opuntiae), the genus Pachysolen (including, but not limited to, P. tannophilus), the genus Bretannomyces (including, but not limited to, e.g., B. clausenii (Philippidis, G. P., 1996, Cellulose bioconversion technology, in Handbook on Bioethanol: Production and Utilization, Wyman, C. E., ed., Taylor & Francis, Washington, D.C., 179-212)). Other suitable microorganisms include, for example, Zymomonas mobilis, Clostridium spp. (including, but not limited to, C. thermocellum (Philippidis, 1996, supra), C. saccharobutylacetonicum, C. saccharobutylicum, C. Puniceum, C. beijernckii, and C. acetobutylicum), Moniliella pollinis, Moniliella megachiliensis, Lactobacillus spp. Yarrowia lipolytica, Aureobasidium sp., Trichosporonoides sp., Trigonopsis variabilis, Trichosporon sp., Moniliellaacetoabutans sp., Typhula variabilis, Candida magnoliae, Ustilaginomycetes sp., Pseudozyma tsukubaensis, yeast species of genera Zygosaccharomyces, Debaryomyces, Hansenula and Pichia, and fungi of the dematioid genus Torula.
[0177] For instance, Clostridium spp. can be used to produce ethanol, butanol, butyric acid, acetic acid, and acetone. Lactobacillus spp. can be used to produce lactic acid.
[0178] Many such microbial strains are publicly available, either commercially or through depositories such as the ATCC (American Type Culture Collection, Manassas, Va., USA), the NRRL (Agricultural Research Sevice Culture Collection, Peoria, Ill., USA), or the DSMZ (Deutsche Sammlung von Mikroorganismen and Zellkulturen GmbH, Braunschweig, Germany), to name a few.
[0179] Commercially available yeasts include, for example, Red Star.RTM./Lesaffre Ethanol Red (available from Red Star/Lesaffre, USA), FALI.RTM. (available from Fleischmann's Yeast, a division of Burns Philip Food Inc., USA), SUPERSTART.RTM. (available from Alltech, now Lalemand), GERT STRAND.RTM. (available from Gert Strand AB, Sweden) and FERMOL.RTM. (available from DSM Specialties).
[0180] Many microorganisms that can be used to saccharify biomass material and produce sugars can also be used to ferment and convert those sugars to useful products.
[0181] After fermentation, the resulting fluids can be distilled using, for example, a "beer column" to separate ethanol and other alcohols from the majority of water and residual solids. The vapor exiting the beer column can be, e.g., 35% by weight ethanol and can be fed to a rectification column. A mixture of nearly azeotropic (92.5%) ethanol and water from the rectification column can be purified to pure (99.5%) ethanol using vapor-phase molecular sieves. The beer column bottoms can be sent to the first effect of a three-effect evaporator. The rectification column reflux condenser can provide heat for this first effect. After the first effect, solids can be separated using a centrifuge and dried in a rotary dryer. A portion (25%) of the centrifuge effluent can be recycled to fermentation and the rest sent to the second and third evaporator effects. Most of the evaporator condensate can be returned to the process as fairly clean condensate with a small portion split off to waste water treatment to prevent build-up of low-boiling compounds.
[0182] Other types of chemical transformation of the products from the processes described herein can be used, for example, production of organic sugar derived products such (e.g., furfural and furfural-derived products). Chemical transformations of sugar derived products are described in U.S. Prov. App. No. 61/667,481, filed Jul. 3, 2012, the disclosure of which is incorporated herein by reference in its entirety.
EXAMPLES
[0183] The invention is further described in detail by reference to the following experimental examples. These examples are provided for purposes of illustration only, and are not intended to be limiting unless otherwise specified. Thus, the invention should in no way be construed as being limited to the following examples, but rather, should be construed to encompass any and all variations which become evident as a result of the teaching provided herein.
[0184] Without further description, it is believed that one of ordinary skill in the art can, using the preceding description and the following illustrative examples, make and utilize the compounds of the present invention and practice the claimed methods. The following working examples specifically point out various aspects of the present invention, and are not to be construed as limiting in any way the remainder of the disclosure.
Example 1
Cloning of Cel3a-C'His into an Expression Vector
[0185] The mature sequence for Cel3a (amino acids 20-744) was synthesized and codon-optimized for E. coli expression by Genewiz. The Cel3a-C'His referred to in the following examples refers to the codon-optimized mature sequence for Cel3a (aas 20-744) with an 8.times. His (SEQ ID NO: 7) tag at the C-terminus. The below primers were used to clone the Cel3a-C'His into pET-Duet (Novagen, Catalog No. 71146):
TABLE-US-00006 Forward (SEQ ID NO: 4) 5'-CATGCCATGGGCGATAGTCACAGTACCAGC Reverse (SEQ ID NO: 5) 3'-CCCAAGCTTTCATTAGTGATGATGATGATGATGATGATGGCTGCCGC TGCCGGCAACACTCAGGGTGC
(NcoI and HindIII sites are underlined; start and stop codons are in bold; the polyhistidine (8-His (SEQ ID NO: 7)) tag; and glycine-serine (GSGS) linker (SEQ ID NO: 8) are italized.) The Amplification reaction was performed using PfuUltra II Fusion HS Polymerase (Agilent, Catalog No. 600672).
[0186] The amplified DNA was cloned by restriction digestion using NcoI restriction enzyme (New England Biolabs, R3193) and HindIII restriction enzyme (New England Biolabs, R3104) under conditions suggested by the manufacturer. The digested amplified DNA was ligated into the NcoI-HindIII sites in the pETDuet vector using T4 DNA ligase (New England Biolabs, M0202), followed by transformation of E. coli cloning host Top10 One Shot (Invitrogen). Plasmid purification was carried out using Qiagen's plasmid purification kit.
Example 2
Expression and Purification of Cel3a-C'His
[0187] The Cel3A-C'His constructs were transformed into the E. coli expression host Origami B (DE3) (EMD Millipore, Catalog No. 70837) and streaked on plates containing LB medium and 100 .mu.g/ml ampicillin (Fisher Scientific, Catalog No. BP1760), 15 .mu.g/ml kanamysin (Fisher Scientific, Catalog No. BP906) and 12.5 .mu.g/ml tetracycline (Fisher Scientified, Catalog No. BP912). Colonies carrying the recombinant DNA were picked from plates for the inoculation of 2 ml starter cultures, and grown overnight at 37.degree. C., then subsequently used to inoculate 100 ml of LB media containing the appropriate antibiotics. Cultures were grown at 37.degree. C. until OD600 reached 0.8.
[0188] To induce protein expression, 500 .mu.M IPTG (Isopropyl-b-D-thiogalactopyranoside; Fisher Scientific, Catalog No. BP1755) was added. The expression culture was further grown for another 4 hours at 37.degree. C. The cells were harvested by centrifugation at 4000 g at room temperature (RT) for 20 minutes using the Sorvall St16 rotor TX400; and the pellet was stored at -80.degree. C.
[0189] For protein extraction, the cell pellet was thawed on ice and resuspended in 2 ml of native lysis buffer containing 50 mM Tris-HCl pH 7.5, 0.1% Triton X-100, 5 mM imidazole (Fisher Scientific, Catalog No. 03196), and 1 mg/ml lysozyme (Fisher Scientific, Catalog No. BP535), then incubated on ice for 20 minutes. To digest the DNA in the sample, 2 .mu.l of lysonase (EMD Millipore, Catalog No. 71230) was added and incubated for another 10 minutes. The sample was then spun in a microcentrifuge at maximum speed for 10 minutes at 4.degree. C. The clarified lysate was transferred to a fresh tube containing 100 .mu.l of pre-equilibrated Profinity (Biorad) Ni-charged IMAC resin slurry (Biorad, Catalog No. 732-4614). The native binding buffer consisted of 50 mM Tris HCl pH 7.5, 150 mM NaCl, 0.1% Triton X-100, and 5 .mu.M imidazole. The protein was batch-bound for 1 hour at RT, and then washed with native buffer containing 25 .mu.M imidazole. The protein was eluted in 300 ml of native buffer containing 200 .mu.M imidazole. Purified Cel3a protein from the expression and purification process described above is shown in FIG. 1.
Example 3
Expression of Cel3a-C'His in Bioreactors
[0190] In this example, the expression and purification of Cel3a is scaled up to 3 liter and 5 liter bioreactors. The skilled artisan could readily use the process described herein to scale the production further, for example, to 3 L and 7 L bioreactors.
[0191] For each bioreactor run, the pET Duet-1 Cel3a-C'His construct is transformed into E. Coli Origami B (DE3) host cell line, and streaked on plates containing LB medium and 100 .mu.g/ml ampicillin (Fisher Scientific, Catalog No. BP1760), 15 .mu.g/ml kanamysin (Fisher Scientific, Catalog No. BP906) and 12.5 .mu.g/ml tetracycline (Fisher Scientific, Catalog No. BP912).
[0192] On Day 1, colonies carrying the recombinant DNA were picked from plates for the inoculation of 200 ml starter cultures, and grown overnight at 37.degree. C. The bioreactors were prepared, e.g., 1.5 L of LB media was added to each 3 L reactor, and 4 L of LB media was added to each 5 L reactor, with the appropriate antibiotics. Each reactor has a pH probe, a dissolved oxygen (DO) probe, and a condenser.
[0193] On Day 2, the OD of the overnight inoculums was measured by a spectrophotometer at 600 nM. The spectrophotometer can be blanked by using LB media. The bioreactors were set to 37.degree. C., 300 rpm, and 2.5 vvm. Once the bioreactors reach 37.degree. C., the appropriate mls of overnight inoculums was added to each 1.5 L and to each 4 L culture for a target starting OD value of 0.05. The OD of the reactors were measured occasionally by sampling out of the top with ethanol and a sterile pipet. As the OD approaches 0.8, samples were taken more frequently.
[0194] When the OD of the reactors reached 0.7-0.8 (approximately 2-4 hours after inoculation), the temperature of the bioreactors was reduced to 20.degree. C., and the cultures were induced with IPTG for Cel3a-C'His protein expression. 750 ml of IPTG was added to each 1.5 L reactor and 1.5 ml of IPTG was added to each 5 L reactor. The reactors were run overnight.
[0195] On Day 3, in the morning, the cells were harvested into clean 2 L centrifuge bottles and spun at 4200 rpm for 45 minutes. The supernatant was discarded, and the pellet was saved and the weight of the pellet was recorded. Using PBS buffer, the pellet was transferred to 50 mL conical centrifuge tubes. The 50 mL tubes were spun at 4500 rpm for 45 minutes in the Sorvall St-16 tabletop centrifuge. Supernatant was discarded and the pellet was frozen at -20.degree. C. until ready for purification, or processed immediately for purification.
Example 4
Protein Extraction from Cells
[0196] Lysis Buffer with EDTA was prepared, containing the following: 50 mM Tris-HCl pH 7.5, 0.1% Triton X-100, 1% Glycerol, 5 mM imidazole, 1 mg/mL Lysozyme, and 1 mM EDTA in de-ionized water. The cell pellet was resuspended in 5 mL volume of Lysis Buffer with EDTA. 10 .mu.l of Lysonase (DNAse) was added to the sample. Samples were then incubated for 1 hour at room temperature.
[0197] Then, samples were sonicated on ice for three 1 minute intervals (e.g., for 3 minutes total, Sorvall sonicator at setting 17).
[0198] At this point, 1000 .mu.l aliquots were e reserved from each fraction of the extraction process to analyze the yield and activity of the Cel3a-C'His protein from the extraction and purification process. An aliquot was taken from sonicated sample (sample A) to represent the total protein isolated. The sample was then centrifuged at 13000 rpm for 5 minutes, and the supernatant was removed. An aliquot was taken from the supernatant (sample D), representing the soluble fraction. The remaining pellet was resuspended in 1000 .mu.l of Lysis Buffer+EDTA, and then centrifuged at 13000 rpm for 5 minutes. The supernatant was removed, and an aliquot was taken from the supernatant (sample E), representing the soluble wash fraction. The remaining pellet was reserved (sample C), representing the insoluble fraction. All samples were stored in the refrigerator (e.g., 4.degree. C.) until analysis.
[0199] A cellobiase activity assay was performed using the total protein aliquot (sample A), for example, as described in Example 6.
[0200] The samples A, C, D, and E were also analyzed by SDS-PAGE and western blotting. Samples were diluted to 2.00 OD to normalize. 17.5 .mu.l of LDS sample buffer (375 .mu.l LDS buffer and 150 .mu.l of NuPAGE reducing agent were mixed to prepare 525 .mu.l of LDS sample buffer) was added to 32.5 .mu.l of each sample. Samples were boiled for 5 minutes and 20 .mu.l of each sample was loaded into each gel well. A standard (molecular weight ladder) was also loaded into a gel well.
Example 5
Mass Spectrometry Analysis of Purified Cel3a-N'His
[0201] Cel3a with a His-tag at the N-terminus (Cel3a-N'His) was cloned, expressed in E. coli, and harvested using methods similar to those described above in Examples 1-4. Cel3a-N'His was cloned into pET-Duet1 vector. The expressed Cel3a-N'His was purified with an affinity column containing Profinity Ni-charge IMAC resin slurry and eluted using elution buffer containing 200 mM imidazole in the lysis buffer.
[0202] The purified Cel3a-N'His was dialyzed in buffer containing 50mM Tris-HCl and 0.5M EDTA. The dialyzed protein was then submitted for liquid chromatography-mass spectrometry (LCMS) intact mass analysis.
[0203] Sample concentration was determined, e.g., by Bradford assay and by Nanodrop assay. Sample concentration was at about 4 mg/mL. A 10.times. and 100.times. dilution sample was prepared for LCMS, with 0.1% formic acid in de-ionized water to provide 0.4 mg/mL and 0.04 mg/mL samples.
[0204] LC conditions were as follows: [0205] Acquity UPLC H-Class Bio System: BEH300 C4 column, 1.7 m, 2.1.times.150 mm (p/n: 186004497) [0206] Column Temp: 45.degree. C. [0207] MPA: 0.1% FA in Water [0208] MPB: 0.1% FA in ACN
[0209] Mass Spectrometer conditions were as follows: [0210] Source Temp: 125.degree. C. [0211] Desolvation Temp: 450.degree. C. [0212] Cone Voltage: 40V [0213] Calibration: Csl (500-5000 Da) [0214] Lockmass: Csl
[0215] The chromatographic profile of the sample is shown in FIG. 2. The profile shows three areas of interest: a large peak at 31 minutes, a cluster of peaks between 38 and 40 minutes, and a smaller peak at about 45 minutes. Manual deconvolution of the mass spectrum of peak at about 45 minutes and the peak cluster at about 39 minutes shows that the main peaks are not part of a charge state envelope, but rather a polymer series (with 44 amu as the repeating unit), and were thus attributed to possibly being from the Triton-X used in the lysis buffer.
[0216] Analysis of the mass spectral region for the peak detected at 31 minutes indicates a protein charge state envelope, and importantly, with no evidence of glycosylation (FIG. 3). Expansion of the charge state envelope indicates the presence of 2-3 minor proteins slightly larger than the major component. Deconvolution of the charge state envelope indicated that the major component had a molecular weight of 78,052 (FIG. 4), which corresponds to the expected molecular weight of Cel3a-N'His. The minor components had the following molecular weights: 78,100, 78,182, and 78,229 (FIG. 4), therefore indicating minor modifications to the Cel3a-N'His protein.
[0217] Among the charge states for the main component were several signals corresponding to smaller peptide fragments. These are distinguished by the more widely spaced isotope peaks. Some of the fragments identified have molecular weights of 8,491, 9,261, and 11,629. These fragments may be generated in the MS source, or may originate from the sample.
[0218] The results from this experiment demonstrate that an aglycosylated cellobiase, Cel3a-N'His was cloned, expressed, and isolated using the methods encompassed by the present invention.
Example 6
Cellobiase Activity Assay
[0219] His-tagged Cel3a (e.g., Cel3a with a His-tag at the N or C-terminus) were purified using IMAC techniques. The amount (titer) of purified His-tagged Cel3a was determined using Bradford assay and/or the nanodrop. For nanodrop quantification, the molar extinction coefficient was estimated by inserting the amino acid sequence of the target form of Cel3a into the ExPASy ProtParam online tool.
[0220] For the activity assay, two fold serial dilutions of samples containing Cel3a-N'His were performed in a 96 well plate format. Dilutions were incubated with a D-(+)-Cellobiose (Fluka) substrate solution in 50 mM sodium citrate monobasic buffer at pH 5.0, at 48.degree. C. for 30 minutes. After 30 minutes, the samples were heated to 100.degree. C. for 10 minutes to stop the reaction. Samples were analysed for glucose and cellobiose using the YSI Biochemistry analyser (YSI Life Sciences) and/or HPLC methods. The protein activity of the purified Cel3a-N'His was recorded as percent conversion of cellobiose to glucose per 30 minutes (FIG. 5).
[0221] For samples where the amount of Cel3a cannot be assayed by using a Bradford assay and/or the nanodrop, e.g., crude lysate sample, the activity assay can be used to determine the titer of Cel3a. A standard curve of cellobiase activity of a sample of purified Cel3a-N'His with a known concentration is generated. Two-fold serial dilutions of Cel3a-N'His with a known concentration were prepared in one row of a 96 well plate, e.g., 12 two-fold serial dilutions. The other rows contained two-fold serial dilutions of other remaining samples whose titer is to be determined, e.g., the crude lysate sample. The dilutions were incubated with a D-(+)-Cellobiose (Fluka) substrate solution in 50 mM sodium citrate monobasic buffer at pH 5.0, at 48.degree. C. for 30 minutes. After 30 minutes, the samples were heated to 100.degree. C. for 10 minutes to stop the reaction. Samples were analysed for glucose and cellobiose using the YSI Biochemistry analyser (YSI Life Sciences) and/or HPLC methods. Using Cel3a-N'His samples of known concentration, a standard curve is generated using the data points within the linear range of the assay (FIG. 6). The cellobiase activity detected from the samples with unknown Cel3a titer can be compared to the standard curve to determine the titer of Cel3a in the sample.
[0222] Units of activity are only relative if calculated using values within the linear range of the assay. The linear range of the assay is defined as using glucose values that are less than 30% of the original soluble substrate load. In addition, glucose values lower than 0.05 g/L are omitted due to instrumentation reporting levels. One unit of cellobiase activity is defined as the amount of glucose per the amount of Cel3a per 30 minutes: [Glucose]g/L/[Cel3a]g/L/30 min.
[0223] A comparison of the activity between aglycosylated Cel3a purified from E. coli and endogenous (glycosylated) cellobiase Cel3a from T. reesei was performed, using the cellobiase activity described herein. The results are shown in FIG. 7, which show that the recombinant Cel3a (aglycosylated Cel3a purified from E. coli; labeled Cel3a in FIG. 7) demonstrated higher specific activity for converting cellobiose to glucose compared to the endogenous Cel3a (labeled L4196 in FIG. 7). Specifically, at concentrations lower than 0.2 mg/mL of cellobiase, the recombinant Cel3a was able to produce a larger amount of glucose after 30 minutes than the endogenous Cel3a.
Example 7
Addition of Aglycosylated Cellobiase Increases Product from Saccharification Process
[0224] In this example, the glucose yield was compared between two saccharification reactions. In one saccharification reaction, an enzyme mixture comprising the enzymes described in Table 1 was added to a biomass. In a second saccharification reaction, 0.8 mg/ml of aglycosylated Cel3a purified from E. coli was added to the enzyme mixture comprising the enzymes described in Table 1, and was added to a biomass. The saccharification reactions were run under the conditions as described herein, with samples taken at multiple timepoints between 0 and 80 minutes of the reaction. The resulting glucose was measured and quantified using methods in the art, such as by YSI instrument or HPLC, and plotted over time, as shown in FIG. 8. These results show that addition of aglycosylated Cel3a increased the yield of sugar product, e.g., glucose, in a saccharification reaction.
EQUIVALENTS
[0225] The disclosures of each and every patent, patent application, and publication cited herein are hereby incorporated herein by reference in their entirety. While this invention has been disclosed with reference to specific aspects, it is apparent that other aspects and variations of this invention may be devised by others skilled in the art without departing from the true spirit and scope of the invention. The appended claims are intended to be construed to include all such aspects and equivalent variations.
Sequence CWU 1
1
231731PRTTrichoderma reesei 1Met Gly Asp Ser His Ser Thr Ser Gly
Ala Ser Ala Glu Ala Val Val 1 5 10 15 Pro Pro Ala Gly Thr Pro Trp
Gly Thr Ala Tyr Asp Lys Ala Lys Ala 20 25 30 Ala Leu Ala Lys Leu
Asn Leu Gln Asp Lys Val Gly Ile Val Ser Gly 35 40 45 Val Gly Trp
Asn Gly Gly Pro Cys Val Gly Asn Thr Ser Pro Ala Ser 50 55 60 Lys
Ile Ser Tyr Pro Ser Leu Cys Leu Gln Asp Gly Pro Leu Gly Val 65 70
75 80 Arg Tyr Ser Thr Gly Ser Thr Ala Phe Thr Pro Gly Val Gln Ala
Ala 85 90 95 Ser Thr Trp Asp Val Asn Leu Ile Arg Glu Arg Gly Gln
Phe Ile Gly 100 105 110 Glu Glu Val Lys Ala Ser Gly Ile His Val Ile
Leu Gly Pro Val Ala 115 120 125 Gly Pro Leu Gly Lys Thr Pro Gln Gly
Gly Arg Asn Trp Glu Gly Phe 130 135 140 Gly Val Asp Pro Tyr Leu Thr
Gly Ile Ala Met Gly Gln Thr Ile Asn 145 150 155 160 Gly Ile Gln Ser
Val Gly Val Gln Ala Thr Ala Lys His Tyr Ile Leu 165 170 175 Asn Glu
Gln Glu Leu Asn Arg Glu Thr Ile Ser Ser Asn Pro Asp Asp 180 185 190
Arg Thr Leu His Glu Leu Tyr Thr Trp Pro Phe Ala Asp Ala Val Gln 195
200 205 Ala Asn Val Ala Ser Val Met Cys Ser Tyr Asn Lys Val Asn Thr
Thr 210 215 220 Trp Ala Cys Glu Asp Gln Tyr Thr Leu Gln Thr Val Leu
Lys Asp Gln 225 230 235 240 Leu Gly Phe Pro Gly Tyr Val Met Thr Asp
Trp Asn Ala Gln His Thr 245 250 255 Thr Val Gln Ser Ala Asn Ser Gly
Leu Asp Met Ser Met Pro Gly Thr 260 265 270 Asp Phe Asn Gly Asn Asn
Arg Leu Trp Gly Pro Ala Leu Thr Asn Ala 275 280 285 Val Asn Ser Asn
Gln Val Pro Thr Ser Arg Val Asp Asp Met Val Thr 290 295 300 Arg Ile
Leu Ala Ala Trp Tyr Leu Thr Gly Gln Asp Gln Ala Gly Tyr 305 310 315
320 Pro Ser Phe Asn Ile Ser Arg Asn Val Gln Gly Asn His Lys Thr Asn
325 330 335 Val Arg Ala Ile Ala Arg Asp Gly Ile Val Leu Leu Lys Asn
Asp Ala 340 345 350 Asn Ile Leu Pro Leu Lys Lys Pro Ala Ser Ile Ala
Val Val Gly Ser 355 360 365 Ala Ala Ile Ile Gly Asn His Ala Arg Asn
Ser Pro Ser Cys Asn Asp 370 375 380 Lys Gly Cys Asp Asp Gly Ala Leu
Gly Met Gly Trp Gly Ser Gly Ala 385 390 395 400 Val Asn Tyr Pro Tyr
Phe Val Ala Pro Tyr Asp Ala Ile Asn Thr Arg 405 410 415 Ala Ser Ser
Gln Gly Thr Gln Val Thr Leu Ser Asn Thr Asp Asn Thr 420 425 430 Ser
Ser Gly Ala Ser Ala Ala Arg Gly Lys Asp Val Ala Ile Val Phe 435 440
445 Ile Thr Ala Asp Ser Gly Glu Gly Tyr Ile Thr Val Glu Gly Asn Ala
450 455 460 Gly Asp Arg Asn Asn Leu Asp Pro Trp His Asn Gly Asn Ala
Leu Val 465 470 475 480 Gln Ala Val Ala Gly Ala Asn Ser Asn Val Ile
Val Val Val His Ser 485 490 495 Val Gly Ala Ile Ile Leu Glu Gln Ile
Leu Ala Leu Pro Gln Val Lys 500 505 510 Ala Val Val Trp Ala Gly Leu
Pro Ser Gln Glu Ser Gly Asn Ala Leu 515 520 525 Val Asp Val Leu Trp
Gly Asp Val Ser Pro Ser Gly Lys Leu Val Tyr 530 535 540 Thr Ile Ala
Lys Ser Pro Asn Asp Tyr Asn Thr Arg Ile Val Ser Gly 545 550 555 560
Gly Ser Asp Ser Phe Ser Glu Gly Leu Phe Ile Asp Tyr Lys His Phe 565
570 575 Asp Asp Ala Asn Ile Thr Pro Arg Tyr Glu Phe Gly Tyr Gly Leu
Ser 580 585 590 Tyr Thr Lys Phe Asn Tyr Ser Arg Leu Ser Val Leu Ser
Thr Ala Lys 595 600 605 Ser Gly Pro Ala Thr Gly Ala Val Val Pro Gly
Gly Pro Ser Asp Leu 610 615 620 Phe Gln Asn Val Ala Thr Val Thr Val
Asp Ile Ala Asn Ser Gly Gln 625 630 635 640 Val Thr Gly Ala Glu Val
Ala Gln Leu Tyr Ile Thr Tyr Pro Ser Ser 645 650 655 Ala Pro Arg Thr
Pro Pro Lys Gln Leu Arg Gly Phe Ala Lys Leu Asn 660 665 670 Leu Thr
Pro Gly Gln Ser Gly Thr Ala Thr Phe Asn Ile Arg Arg Arg 675 680 685
Asp Leu Ser Tyr Trp Asp Thr Ala Ser Gln Lys Trp Val Val Pro Ser 690
695 700 Gly Ser Phe Gly Ile Ser Val Gly Ala Ser Ser Arg Asp Ile Arg
Leu 705 710 715 720 Thr Ser Thr Leu Ser Val Ala Gly Ser Gly Ser 725
730 22232DNATrichoderma reesei 2atgcgttacc gaacagcagc tgcgctggca
cttgccactg ggccctttgc tagggcagac 60agtcactcaa catcgggggc ctcggctgag
gcagttgtac ctcctgcagg gactccatgg 120ggaaccgcgt acgacaaggc
gaaggccgca ttggcaaagc tcaatctcca agataaggtc 180ggcatcgtga
gcggtgtcgg ctggaacggc ggtccttgcg ttggaaacac atctccggcc
240tccaagatca gctatccatc gctatgcctt caagacggac ccctcggtgt
tcgatactcg 300acaggcagca cagcctttac gccgggcgtt caagcggcct
cgacgtggga tgtcaatttg 360atccgcgaac gtggacagtt catcggtgag
gaggtgaagg cctcggggat tcatgtcata 420cttggtcctg tggctgggcc
gctgggaaag actccgcagg gcggtcgcaa ctgggagggc 480ttcggtgtcg
atccatatct cacgggcatt gccatgggtc aaaccatcaa cggcatccag
540tcggtaggcg tgcaggcgac agcgaagcac tatatcctca acgagcagga
gctcaatcga 600gaaaccattt cgagcaaccc agatgaccga actctccatg
agctgtatac ttggccattt 660gccgacgcgg ttcaggccaa tgtcgcttct
gtcatgtgct cgtacaacaa ggtcaatacc 720acctgggcct gcgaggatca
gtacacgctg cagactgtgc tgaaagacca gctggggttc 780ccaggctatg
tcatgacgga ctggaacgca cagcacacga ctgtccaaag cgcgaattct
840gggcttgaca tgtcaatgcc tggcacagac ttcaacggta acaatcggct
ctggggtcca 900gctctcacca atgcggtaaa tagcaatcag gtccccacga
gcagagtcga cgatatggtg 960actcgtatcc tcgccgcatg gtacttgaca
ggccaggacc aggcaggcta tccgtcgttc 1020aacatcagca gaaatgttca
aggaaaccac aagaccaatg tcagggcaat tgccagggac 1080ggcatcgttc
tgctcaagaa tgacgccaac atcctgccgc tcaagaagcc cgctagcatt
1140gccgtcgttg gatctgccgc aatcattggt aaccacgcca gaaactcgcc
ctcgtgcaac 1200gacaaaggct gcgacgacgg ggccttgggc atgggttggg
gttccggcgc cgtcaactat 1260ccgtacttcg tcgcgcccta cgatgccatc
aataccagag cgtcttcgca gggcacccag 1320gttaccttga gcaacaccga
caacacgtcc tcaggcgcat ctgcagcaag aggaaaggac 1380gtcgccatcg
tcttcatcac cgccgactcg ggtgaaggct acatcaccgt ggagggcaac
1440gcgggcgatc gcaacaacct ggatccgtgg cacaacggca atgccctggt
ccaggcggtg 1500gccggtgcca acagcaacgt cattgttgtt gtccactccg
ttggcgccat cattctggag 1560cagattcttg ctcttccgca ggtcaaggcc
gttgtctggg cgggtcttcc ttctcaggag 1620agcggcaatg cgctcgtcga
cgtgctgtgg ggagatgtca gcccttctgg caagctggtg 1680tacaccattg
cgaagagccc caatgactat aacactcgca tcgtttccgg cggcagtgac
1740agcttcagcg agggactgtt catcgactat aagcacttcg acgacgccaa
tatcacgccg 1800cggtacgagt tcggctatgg actgtcttac accaagttca
actactcacg cctctccgtc 1860ttgtcgaccg ccaagtctgg tcctgcgact
ggggccgttg tgccgggagg cccgagtgat 1920ctgttccaga atgtcgcgac
agtcaccgtt gacatcgcaa actctggcca agtgactggt 1980gccgaggtag
cccagctgta catcacctac ccatcttcag cacccaggac ccctccgaag
2040cagctgcgag gctttgccaa gctgaacctc acgcctggtc agagcggaac
agcaacgttc 2100aacatccgac gacgagatct cagctactgg gacacggctt
cgcagaaatg ggtggtgccg 2160tcggggtcgt ttggcatcag cgtgggagcg
agcagccggg atatcaggct gacgagcact 2220ctgtcggtag cg
223232232DNAArtificial SequenceDescription of Artificial Sequence
Synthetic polynucleotide 3atgcgttatc gtacagccgc agccctggca
ctggccacag gtccgttcgc acgtgccgat 60agtcacagta ccagcggtgc cagcgcagaa
gccgtggttc cgccggcagg cacaccgtgg 120ggcacagcct atgataaagc
caaagccgcc ctggccaagc tgaatctgca ggataaagtg 180ggcatcgtga
gtggcgtggg ctggaacggt ggtccgtgcg ttggcaacac cagcccggca
240agcaagatca gctatccgag cttatgcctg caggatggtc cgctgggcgt
gcgctatagc 300accggtagta ccgcctttac acctggtgtg caggccgcca
gtacctggga cgttaacctg 360atccgcgaac gtggccaatt tatcggcgaa
gaagttaaag ccagcggcat tcatgttatt 420ctgggtccgg tggccggtcc
tctgggtaaa accccgcagg gcggccgtaa ttgggaaggc 480ttcggcgttg
atccgtattt aaccggcatc gcaatgggcc agaccattaa tggcatccag
540agcgtgggtg ttcaagccac cgccaaacac tacatattaa acgaacagga
actgaatcgt 600gaaaccatca gcagcaatcc ggatgatcgc accctgcatg
agctgtatac atggcctttt 660gccgacgcag ttcaggccaa cgtggcaagt
gtgatgtgta gctataacaa ggtgaacacc 720acctgggcct gcgaagacca
gtacaccctg cagaccgttt taaaagacca actgggcttc 780cctggttacg
tgatgacaga ttggaatgcc cagcacacaa ccgttcagag cgcaaacagt
840ggcctggata tgagcatgcc gggcaccgac ttcaacggca ataatcgtct
gtggggtccg 900gcactgacca atgccgttaa cagcaaccag gtgccgacca
gtcgtgtgga cgatatggtt 960acccgtattc tggccgcctg gtacctgaca
ggtcaagacc aggccggcta cccgagcttc 1020aacatcagcc gcaacgtgca
gggtaatcac aagaccaacg ttcgcgcaat cgcacgcgat 1080ggtatcgtgc
tgttaaagaa cgatgccaac attctgccgc tgaaaaaacc ggccagcatc
1140gccgttgttg gtagcgcagc catcattggc aaccacgccc gtaacagtcc
gagctgcaat 1200gataaaggct gtgacgacgg tgccctgggc atgggttggg
gtagtggtgc cgtgaactac 1260ccgtatttcg tggccccgta cgacgccatt
aacacccgtg caagtagcca gggtacccag 1320gttaccctga gcaacaccga
caacacaagc agcggtgcca gtgcagcacg tggtaaggat 1380gtggccatcg
tgttcatcac cgccgacagc ggcgaaggct acattaccgt ggagggtaat
1440gccggtgatc gcaataatct ggacccgtgg cataacggca acgccctggt
tcaggcagtg 1500gcaggcgcaa atagcaacgt gatcgttgtg gtgcatagcg
tgggtgccat cattctggag 1560cagatcctgg ccctgccgca agttaaggca
gttgtgtggg caggtctgcc gagccaagaa 1620agtggcaatg ccctggtgga
cgttctgtgg ggcgatgtta gtccgagcgg caagctggtg 1680tatacaatcg
ccaagagccc gaacgactat aacacccgca tcgttagcgg cggcagtgat
1740agcttcagcg agggcctgtt tatcgactac aagcatttcg atgatgccaa
tattaccccg 1800cgctacgaat ttggttatgg cctgagctat accaagttca
actacagccg cctgagcgtt 1860ttaagtaccg ccaagagtgg tccggcaaca
ggtgccgtgg ttcctggtgg tccgagtgat 1920ctgtttcaga atgtggccac
cgtgaccgtg gatatcgcca acagtggtca ggttaccggc 1980gccgaagtgg
cacagctgta catcacctat ccgagcagtg caccgcgcac cccgccgaaa
2040cagctgcgtg gcttcgccaa attaaacctg accccgggcc agagcggtac
agcaaccttc 2100aatattcgcc gccgtgatct gagctattgg gacaccgcca
gccaaaaatg ggtggtgccg 2160agcggcagct ttggcattag tgtgggtgca
agtagccgcg acattcgctt aacaagcacc 2220ctgagtgttg cc
2232430DNAArtificial SequenceDescription of Artificial Sequence
Synthetic primer 4catgccatgg gcgatagtca cagtaccagc
30568DNAArtificial SequenceDescription of Artificial Sequence
Synthetic primer 5cgtgggactc acaacggccg tcgccgtcgg tagtagtagt
agtagtagta gtgattactt 60tcgaaccc 6866PRTArtificial
SequenceDescription of Artificial Sequence Synthetic 6xHis tag 6His
His His His His His 1 5 78PRTArtificial SequenceDescription of
Artificial Sequence Synthetic 8xHis tag 7His His His His His His
His His 1 5 84PRTArtificial SequenceDescription of Artificial
Sequence Synthetic peptide 8Gly Ser Gly Ser 1 9800PRTPodospora
anserina 9Met Lys Ser Ser Val Phe Trp Gly Ala Ser Leu Thr Ser Ala
Val Val 1 5 10 15 Arg Ala Ile Asp Leu Pro Phe Gln Phe Tyr Pro Asn
Cys Val Asp Asp 20 25 30 Leu Leu Ser Thr Asn Gln Val Cys Asn Thr
Thr Leu Ser Pro Pro Glu 35 40 45 Arg Ala Ala Ala Leu Val Ala Ala
Leu Thr Pro Glu Glu Lys Leu Gln 50 55 60 Asn Ile Val Ser Lys Ser
Leu Gly Ala Pro Arg Ile Gly Leu Pro Ala 65 70 75 80 Tyr Asn Trp Trp
Ser Glu Ala Leu His Gly Val Ala Tyr Ala Pro Gly 85 90 95 Thr Gln
Phe Trp Gln Gly Asp Gly Pro Phe Asn Ser Ser Thr Ser Phe 100 105 110
Pro Met Pro Leu Leu Met Ala Ala Thr Phe Asp Asp Glu Leu Leu Glu 115
120 125 Lys Ile Ala Glu Val Ile Gly Ile Glu Gly Arg Ala Phe Gly Asn
Ala 130 135 140 Gly Phe Ser Gly Leu Asp Tyr Trp Thr Pro Asn Val Asn
Pro Phe Lys 145 150 155 160 Asp Pro Arg Trp Gly Arg Gly Ser Glu Thr
Pro Gly Glu Asp Val Leu 165 170 175 Leu Val Lys Arg Tyr Ala Ala Ala
Met Ile Lys Gly Leu Glu Gly Pro 180 185 190 Val Pro Glu Lys Glu Arg
Arg Val Val Ala Thr Cys Lys His Tyr Ala 195 200 205 Ala Asn Asp Phe
Glu Asp Trp Asn Gly Ala Thr Arg His Asn Phe Asn 210 215 220 Ala Lys
Ile Ser Leu Gln Asp Met Ala Glu Tyr Tyr Phe Met Pro Phe 225 230 235
240 Gln Gln Cys Val Arg Asp Ser Arg Val Gly Ser Ile Met Cys Ala Tyr
245 250 255 Asn Ala Val Asn Gly Val Pro Ser Cys Ala Ser Pro Tyr Leu
Leu Gln 260 265 270 Thr Ile Leu Arg Glu His Trp Asn Trp Thr Glu His
Asn Asn Tyr Ile 275 280 285 Thr Ser Asp Cys Glu Ala Val Leu Asp Val
Ser Leu Asn His Lys Tyr 290 295 300 Ala Ala Thr Asn Ala Glu Gly Thr
Ala Ile Ser Phe Glu Ala Gly Met 305 310 315 320 Asp Thr Ser Cys Glu
Tyr Glu Gly Ser Ser Asp Ile Pro Gly Ala Trp 325 330 335 Ser Gln Gly
Leu Leu Lys Glu Ser Thr Val Asp Arg Ala Leu Leu Arg 340 345 350 Leu
Tyr Glu Gly Ile Val Arg Ala Gly Tyr Phe Asp Gly Lys Gln Ser 355 360
365 Leu Tyr Ser Ser Leu Gly Trp Ala Asp Val Asn Lys Pro Ser Ala Gln
370 375 380 Lys Leu Ser Leu Gln Ala Ala Val Asp Gly Thr Val Leu Leu
Lys Asn 385 390 395 400 Asp Gly Thr Leu Pro Leu Ser Asp Leu Leu Asp
Lys Ser Arg Pro Lys 405 410 415 Lys Val Ala Met Ile Gly Phe Trp Ser
Asp Ala Lys Asp Lys Leu Arg 420 425 430 Gly Gly Tyr Ser Gly Thr Ala
Ala Tyr Leu His Thr Pro Ala Tyr Ala 435 440 445 Ala Ser Gln Leu Gly
Ile Pro Phe Ser Thr Ala Ser Gly Pro Ile Leu 450 455 460 His Ser Asp
Leu Ala Ser Asn Gln Ser Trp Thr Asp Asn Ala Met Ala 465 470 475 480
Ala Ala Lys Asp Ala Asp Tyr Ile Leu Tyr Phe Gly Gly Ile Asp Thr 485
490 495 Ser Ala Ala Gly Glu Thr Lys Asp Arg Tyr Asp Leu Asp Trp Pro
Gly 500 505 510 Ala Gln Leu Ser Leu Ile Asn Leu Leu Thr Thr Leu Ser
Lys Pro Leu 515 520 525 Ile Val Leu Gln Met Gly Asp Gln Leu Asp Asn
Thr Pro Leu Leu Ser 530 535 540 Asn Pro Lys Ile Asn Ala Ile Leu Trp
Ala Asn Trp Pro Gly Gln Asp 545 550 555 560 Gly Gly Thr Ala Val Met
Glu Leu Val Thr Gly Leu Lys Ser Pro Ala 565 570 575 Gly Arg Leu Pro
Val Thr Gln Tyr Pro Ser Asn Phe Thr Glu Leu Val 580 585 590 Pro Met
Thr Asp Met Ala Leu Arg Pro Ser Ala Gly Asn Ser Gln Leu 595 600 605
Gly Arg Thr Tyr Arg Trp Tyr Lys Thr Pro Val Gln Ala Phe Gly Phe 610
615 620 Gly Leu His Tyr Thr Thr Phe Ser Pro Lys Phe Gly Lys Lys Phe
Pro 625 630 635 640 Ala Val Ile Asp Val Asp Glu Val Leu Glu Gly Cys
Asp Asp Lys Tyr 645 650 655 Leu Asp Thr Cys Pro Leu Pro Asp Leu Pro
Val Val Val Glu Asn Arg 660 665 670 Gly Asn Arg Thr Ser Asp Tyr Val
Ala Leu Ala Phe Val Ser Ala Pro 675 680 685 Gly Val Gly Pro Gly Pro
Trp Pro Ile Lys Thr Leu Gly Ala Phe Thr 690 695 700 Arg Leu Arg Gly
Val Lys Gly Gly Glu Lys Arg Glu Gly Gly Leu Lys 705 710 715 720 Trp
Asn Leu Gly Asn Leu Ala Arg His Asp Glu Glu Gly Asn Thr Val 725 730
735 Val Tyr Pro Gly Lys Tyr Glu Val Ser Leu Asp Glu Pro Pro Lys Ala
740 745 750 Arg Leu Arg
Phe Glu Ile Val Arg Gly Gly Lys Gly Lys Gly Lys Val 755 760 765 Lys
Gly Lys Gly Lys Ala Ala Gln Lys Gly Gly Val Val Leu Asp Arg 770 775
780 Trp Pro Lys Pro Pro Lys Gly Gln Glu Pro Pro Ala Ile Glu Arg Val
785 790 795 800 10316PRTTrichoderma reesei 10Met Val Arg Arg Thr
Ala Leu Leu Ala Leu Gly Ala Leu Ser Thr Leu 1 5 10 15 Ser Met Ala
Gln Ile Ser Asp Asp Phe Glu Ser Gly Trp Asp Gln Thr 20 25 30 Lys
Trp Pro Ile Ser Ala Pro Asp Cys Asn Gln Gly Gly Thr Val Ser 35 40
45 Leu Asp Thr Thr Val Ala His Ser Gly Ser Asn Ser Met Lys Val Val
50 55 60 Gly Gly Pro Asn Gly Tyr Cys Gly His Ile Phe Phe Gly Thr
Thr Gln 65 70 75 80 Val Pro Thr Gly Asp Val Tyr Val Arg Ala Trp Ile
Arg Leu Gln Thr 85 90 95 Ala Leu Gly Ser Asn His Val Thr Phe Ile
Ile Met Pro Asp Thr Ala 100 105 110 Gln Gly Gly Lys His Leu Arg Ile
Gly Gly Gln Ser Gln Val Leu Asp 115 120 125 Tyr Asn Arg Glu Ser Asp
Asp Ala Thr Leu Pro Asp Leu Ser Pro Asn 130 135 140 Gly Ile Ala Ser
Thr Val Thr Leu Pro Thr Gly Ala Phe Gln Cys Phe 145 150 155 160 Glu
Tyr His Leu Gly Thr Asp Gly Thr Ile Glu Thr Trp Leu Asn Gly 165 170
175 Ser Leu Ile Pro Gly Met Thr Val Gly Pro Gly Val Asp Asn Pro Asn
180 185 190 Asp Ala Gly Trp Thr Arg Ala Ser Tyr Ile Pro Glu Ile Thr
Gly Val 195 200 205 Asn Phe Gly Trp Glu Ala Tyr Ser Gly Asp Val Asn
Thr Val Trp Phe 210 215 220 Asp Asp Ile Ser Ile Ala Ser Thr Arg Val
Gly Cys Gly Pro Gly Ser 225 230 235 240 Pro Gly Gly Pro Gly Ser Ser
Thr Thr Gly Arg Ser Ser Thr Ser Gly 245 250 255 Pro Thr Ser Thr Ser
Arg Pro Ser Thr Thr Ile Pro Pro Pro Thr Ser 260 265 270 Arg Thr Thr
Thr Ala Thr Gly Pro Thr Gln Thr His Tyr Gly Gln Cys 275 280 285 Gly
Gly Ile Gly Tyr Ser Gly Pro Thr Val Cys Ala Ser Gly Thr Thr 290 295
300 Cys Gln Val Leu Asn Pro Tyr Tyr Ser Gln Cys Leu 305 310 315
11460PRTTrichoderma reesei 11Met Ala Ser Arg Phe Phe Ala Leu Leu
Leu Leu Ala Ile Pro Ile Gln 1 5 10 15 Ala Gln Ser Pro Val Trp Gly
Gln Cys Gly Gly Ile Gly Trp Ser Gly 20 25 30 Pro Thr Thr Cys Val
Gly Gly Ala Thr Cys Val Ser Tyr Asn Pro Tyr 35 40 45 Tyr Ser Gln
Cys Ile Pro Ser Thr Gln Ala Ser Ser Ser Ile Ala Ser 50 55 60 Thr
Thr Leu Val Thr Ser Phe Thr Thr Thr Thr Ala Thr Arg Thr Ser 65 70
75 80 Ala Ser Thr Pro Pro Ala Ser Ser Thr Gly Ala Gly Gly Ala Thr
Cys 85 90 95 Ser Ala Leu Pro Gly Ser Ile Thr Leu Arg Ser Asn Ala
Lys Leu Asn 100 105 110 Asp Leu Phe Thr Met Phe Asn Gly Asp Lys Val
Thr Thr Lys Asp Lys 115 120 125 Phe Ser Cys Arg Gln Ala Glu Met Ser
Glu Leu Ile Gln Arg Tyr Glu 130 135 140 Leu Gly Thr Leu Pro Gly Arg
Pro Ser Thr Leu Thr Ala Ser Phe Ser 145 150 155 160 Gly Asn Thr Leu
Thr Ile Asn Cys Gly Glu Ala Gly Lys Ser Ile Ser 165 170 175 Phe Thr
Val Thr Ile Thr Tyr Pro Ser Ser Gly Thr Ala Pro Tyr Pro 180 185 190
Ala Ile Ile Gly Tyr Gly Gly Gly Ser Leu Pro Ala Pro Ala Gly Val 195
200 205 Ala Met Ile Asn Phe Asn Asn Asp Asn Ile Ala Ala Gln Val Asn
Thr 210 215 220 Gly Ser Arg Gly Gln Gly Lys Phe Tyr Asp Leu Tyr Gly
Ser Ser His 225 230 235 240 Ser Ala Gly Ala Met Thr Ala Trp Ala Trp
Gly Val Ser Arg Val Ile 245 250 255 Asp Ala Leu Glu Leu Val Pro Gly
Ala Arg Ile Asp Thr Thr Lys Ile 260 265 270 Gly Val Thr Gly Cys Ser
Arg Asn Gly Lys Gly Ala Met Val Ala Gly 275 280 285 Ala Phe Glu Lys
Arg Ile Val Leu Thr Leu Pro Gln Glu Ser Gly Ala 290 295 300 Gly Gly
Ser Ala Cys Trp Arg Ile Ser Asp Tyr Leu Lys Ser Gln Gly 305 310 315
320 Ala Asn Ile Gln Thr Ala Ser Glu Ile Ile Gly Glu Asp Pro Trp Phe
325 330 335 Ser Thr Thr Phe Asn Ser Tyr Val Asn Gln Val Pro Val Leu
Pro Phe 340 345 350 Asp His His Ser Leu Ala Ala Leu Ile Ala Pro Arg
Gly Leu Phe Val 355 360 365 Ile Asp Asn Asn Ile Asp Trp Leu Gly Pro
Gln Ser Cys Phe Gly Cys 370 375 380 Met Thr Ala Ala His Met Ala Trp
Gln Ala Leu Gly Val Ser Asp His 385 390 395 400 Met Gly Tyr Ser Gln
Ile Gly Ala His Ala His Cys Ala Phe Pro Ser 405 410 415 Asn Gln Gln
Ser Gln Leu Thr Ala Phe Val Gln Lys Phe Leu Leu Gly 420 425 430 Gln
Ser Thr Asn Thr Ala Ile Phe Gln Ser Asp Phe Ser Ala Asn Gln 435 440
445 Ser Gln Trp Ile Asp Trp Thr Thr Pro Thr Leu Ser 450 455 460
12466PRTTrichoderma reesei 12Met Leu Pro Lys Asp Phe Gln Trp Gly
Phe Ala Thr Ala Ala Tyr Gln 1 5 10 15 Ile Glu Gly Ala Val Asp Gln
Asp Gly Arg Gly Pro Ser Ile Trp Asp 20 25 30 Thr Phe Cys Ala Gln
Pro Gly Lys Ile Ala Asp Gly Ser Ser Gly Val 35 40 45 Thr Ala Cys
Asp Ser Tyr Asn Arg Thr Ala Glu Asp Ile Ala Leu Leu 50 55 60 Lys
Ser Leu Gly Ala Lys Ser Tyr Arg Phe Ser Ile Ser Trp Ser Arg 65 70
75 80 Ile Ile Pro Glu Gly Gly Arg Gly Asp Ala Val Asn Gln Ala Gly
Ile 85 90 95 Asp His Tyr Val Lys Phe Val Asp Asp Leu Leu Asp Ala
Gly Ile Thr 100 105 110 Pro Phe Ile Thr Leu Phe His Trp Asp Leu Pro
Glu Gly Leu His Gln 115 120 125 Arg Tyr Gly Gly Leu Leu Asn Arg Thr
Glu Phe Pro Leu Asp Phe Glu 130 135 140 Asn Tyr Ala Arg Val Met Phe
Arg Ala Leu Pro Lys Val Arg Asn Trp 145 150 155 160 Ile Thr Phe Asn
Glu Pro Leu Cys Ser Ala Ile Pro Gly Tyr Gly Ser 165 170 175 Gly Thr
Phe Ala Pro Gly Arg Gln Ser Thr Ser Glu Pro Trp Thr Val 180 185 190
Gly His Asn Ile Leu Val Ala His Gly Arg Ala Val Lys Ala Tyr Arg 195
200 205 Asp Asp Phe Lys Pro Ala Ser Gly Asp Gly Gln Ile Gly Ile Val
Leu 210 215 220 Asn Gly Asp Phe Thr Tyr Pro Trp Asp Ala Ala Asp Pro
Ala Asp Lys 225 230 235 240 Glu Ala Ala Glu Arg Arg Leu Glu Phe Phe
Thr Ala Trp Phe Ala Asp 245 250 255 Pro Ile Tyr Leu Gly Asp Tyr Pro
Ala Ser Met Arg Lys Gln Leu Gly 260 265 270 Asp Arg Leu Pro Thr Phe
Thr Pro Glu Glu Arg Ala Leu Val His Gly 275 280 285 Ser Asn Asp Phe
Tyr Gly Met Asn His Tyr Thr Ser Asn Tyr Ile Arg 290 295 300 His Arg
Ser Ser Pro Ala Ser Ala Asp Asp Thr Val Gly Asn Val Asp 305 310 315
320 Val Leu Phe Thr Asn Lys Gln Gly Asn Cys Ile Gly Pro Glu Thr Gln
325 330 335 Ser Pro Trp Leu Arg Pro Cys Ala Ala Gly Phe Arg Asp Phe
Leu Val 340 345 350 Trp Ile Ser Lys Arg Tyr Gly Tyr Pro Pro Ile Tyr
Val Thr Glu Asn 355 360 365 Gly Thr Ser Ile Lys Gly Glu Ser Asp Leu
Pro Lys Glu Lys Ile Leu 370 375 380 Glu Asp Asp Phe Arg Val Lys Tyr
Tyr Asn Glu Tyr Ile Arg Ala Met 385 390 395 400 Val Thr Ala Val Glu
Leu Asp Gly Val Asn Val Lys Gly Tyr Phe Ala 405 410 415 Trp Ser Leu
Met Asp Asn Phe Glu Trp Ala Asp Gly Tyr Val Thr Arg 420 425 430 Phe
Gly Val Thr Tyr Val Asp Tyr Glu Asn Gly Gln Lys Arg Phe Pro 435 440
445 Lys Lys Ser Ala Lys Ser Leu Lys Pro Leu Phe Asp Glu Leu Ile Ala
450 455 460 Ala Ala 465 13744PRTTrichoderma reesei 13Met Arg Tyr
Arg Thr Ala Ala Ala Leu Ala Leu Ala Thr Gly Pro Phe 1 5 10 15 Ala
Arg Ala Asp Ser His Ser Thr Ser Gly Ala Ser Ala Glu Ala Val 20 25
30 Val Pro Pro Ala Gly Thr Pro Trp Gly Thr Ala Tyr Asp Lys Ala Lys
35 40 45 Ala Ala Leu Ala Lys Leu Asn Leu Gln Asp Lys Val Gly Ile
Val Ser 50 55 60 Gly Val Gly Trp Asn Gly Gly Pro Cys Val Gly Asn
Thr Ser Pro Ala 65 70 75 80 Ser Lys Ile Ser Tyr Pro Ser Leu Cys Leu
Gln Asp Gly Pro Leu Gly 85 90 95 Val Arg Tyr Ser Thr Gly Ser Thr
Ala Phe Thr Pro Gly Val Gln Ala 100 105 110 Ala Ser Thr Trp Asp Val
Asn Leu Ile Arg Glu Arg Gly Gln Phe Ile 115 120 125 Gly Glu Glu Val
Lys Ala Ser Gly Ile His Val Ile Leu Gly Pro Val 130 135 140 Ala Gly
Pro Leu Gly Lys Thr Pro Gln Gly Gly Arg Asn Trp Glu Gly 145 150 155
160 Phe Gly Val Asp Pro Tyr Leu Thr Gly Ile Ala Met Gly Gln Thr Ile
165 170 175 Asn Gly Ile Gln Ser Val Gly Val Gln Ala Thr Ala Lys His
Tyr Ile 180 185 190 Leu Asn Glu Gln Glu Leu Asn Arg Glu Thr Ile Ser
Ser Asn Pro Asp 195 200 205 Asp Arg Thr Leu His Glu Leu Tyr Thr Trp
Pro Phe Ala Asp Ala Val 210 215 220 Gln Ala Asn Val Ala Ser Val Met
Cys Ser Tyr Asn Lys Val Asn Thr 225 230 235 240 Thr Trp Ala Cys Glu
Asp Gln Tyr Thr Leu Gln Thr Val Leu Lys Asp 245 250 255 Gln Leu Gly
Phe Pro Gly Tyr Val Met Thr Asp Trp Asn Ala Gln His 260 265 270 Thr
Thr Val Gln Ser Ala Asn Ser Gly Leu Asp Met Ser Met Pro Gly 275 280
285 Thr Asp Phe Asn Gly Asn Asn Arg Leu Trp Gly Pro Ala Leu Thr Asn
290 295 300 Ala Val Asn Ser Asn Gln Val Pro Thr Ser Arg Val Asp Asp
Met Val 305 310 315 320 Thr Arg Ile Leu Ala Ala Trp Tyr Leu Thr Gly
Gln Asp Gln Ala Gly 325 330 335 Tyr Pro Ser Phe Asn Ile Ser Arg Asn
Val Gln Gly Asn His Lys Thr 340 345 350 Asn Val Arg Ala Ile Ala Arg
Asp Gly Ile Val Leu Leu Lys Asn Asp 355 360 365 Ala Asn Ile Leu Pro
Leu Lys Lys Pro Ala Ser Ile Ala Val Val Gly 370 375 380 Ser Ala Ala
Ile Ile Gly Asn His Ala Arg Asn Ser Pro Ser Cys Asn 385 390 395 400
Asp Lys Gly Cys Asp Asp Gly Ala Leu Gly Met Gly Trp Gly Ser Gly 405
410 415 Ala Val Asn Tyr Pro Tyr Phe Val Ala Pro Tyr Asp Ala Ile Asn
Thr 420 425 430 Arg Ala Ser Ser Gln Gly Thr Gln Val Thr Leu Ser Asn
Thr Asp Asn 435 440 445 Thr Ser Ser Gly Ala Ser Ala Ala Arg Gly Lys
Asp Val Ala Ile Val 450 455 460 Phe Ile Thr Ala Asp Ser Gly Glu Gly
Tyr Ile Thr Val Glu Gly Asn 465 470 475 480 Ala Gly Asp Arg Asn Asn
Leu Asp Pro Trp His Asn Gly Asn Ala Leu 485 490 495 Val Gln Ala Val
Ala Gly Ala Asn Ser Asn Val Ile Val Val Val His 500 505 510 Ser Val
Gly Ala Ile Ile Leu Glu Gln Ile Leu Ala Leu Pro Gln Val 515 520 525
Lys Ala Val Val Trp Ala Gly Leu Pro Ser Gln Glu Ser Gly Asn Ala 530
535 540 Leu Val Asp Val Leu Trp Gly Asp Val Ser Pro Ser Gly Lys Leu
Val 545 550 555 560 Tyr Thr Ile Ala Lys Ser Pro Asn Asp Tyr Asn Thr
Arg Ile Val Ser 565 570 575 Gly Gly Ser Asp Ser Phe Ser Glu Gly Leu
Phe Ile Asp Tyr Lys His 580 585 590 Phe Asp Asp Ala Asn Ile Thr Pro
Arg Tyr Glu Phe Gly Tyr Gly Leu 595 600 605 Ser Tyr Thr Lys Phe Asn
Tyr Ser Arg Leu Ser Val Leu Ser Thr Ala 610 615 620 Lys Ser Gly Pro
Ala Thr Gly Ala Val Val Pro Gly Gly Pro Ser Asp 625 630 635 640 Leu
Phe Gln Asn Val Ala Thr Val Thr Val Asp Ile Ala Asn Ser Gly 645 650
655 Gln Val Thr Gly Ala Glu Val Ala Gln Leu Tyr Ile Thr Tyr Pro Ser
660 665 670 Ser Ala Pro Arg Thr Pro Pro Lys Gln Leu Arg Gly Phe Ala
Lys Leu 675 680 685 Asn Leu Thr Pro Gly Gln Ser Gly Thr Ala Thr Phe
Asn Ile Arg Arg 690 695 700 Arg Asp Leu Ser Tyr Trp Asp Thr Ala Ser
Gln Lys Trp Val Val Pro 705 710 715 720 Ser Gly Ser Phe Gly Ile Ser
Val Gly Ala Ser Ser Arg Asp Ile Arg 725 730 735 Leu Thr Ser Thr Leu
Ser Val Ala 740 14418PRTTrichoderma reesei 14Met Asn Lys Ser Val
Ala Pro Leu Leu Leu Ala Ala Ser Ile Leu Tyr 1 5 10 15 Gly Gly Ala
Ala Ala Gln Gln Thr Val Trp Gly Gln Cys Gly Gly Ile 20 25 30 Gly
Trp Ser Gly Pro Thr Asn Cys Ala Pro Gly Ser Ala Cys Ser Thr 35 40
45 Leu Asn Pro Tyr Tyr Ala Gln Cys Ile Pro Gly Ala Thr Thr Ile Thr
50 55 60 Thr Ser Thr Arg Pro Pro Ser Gly Pro Thr Thr Thr Thr Arg
Ala Thr 65 70 75 80 Ser Thr Ser Ser Ser Thr Pro Pro Thr Ser Ser Gly
Val Arg Phe Ala 85 90 95 Gly Val Asn Ile Ala Gly Phe Asp Phe Gly
Cys Thr Thr Asp Gly Thr 100 105 110 Cys Val Thr Ser Lys Val Tyr Pro
Pro Leu Lys Asn Phe Thr Gly Ser 115 120 125 Asn Asn Tyr Pro Asp Gly
Ile Gly Gln Met Gln His Phe Val Asn Asp 130 135 140 Asp Gly Met Thr
Ile Phe Arg Leu Pro Val Gly Trp Gln Tyr Leu Val 145 150 155 160 Asn
Asn Asn Leu Gly Gly Asn Leu Asp Ser Thr Ser Ile Ser Lys Tyr 165 170
175 Asp Gln Leu Val Gln Gly Cys Leu Ser Leu Gly Ala Tyr Cys Ile Val
180 185 190 Asp Ile His Asn Tyr Ala Arg Trp Asn Gly Gly Ile Ile Gly
Gln Gly 195 200 205 Gly Pro Thr Asn Ala Gln Phe Thr Ser Leu Trp Ser
Gln Leu Ala Ser 210 215 220 Lys Tyr Ala Ser Gln Ser Arg Val Trp Phe
Gly Ile Met Asn Glu Pro 225 230 235 240 His Asp Val Asn Ile Asn Thr
Trp Ala Ala Thr Val Gln Glu Val Val 245 250
255 Thr Ala Ile Arg Asn Ala Gly Ala Thr Ser Gln Phe Ile Ser Leu Pro
260 265 270 Gly Asn Asp Trp Gln Ser Ala Gly Ala Phe Ile Ser Asp Gly
Ser Ala 275 280 285 Ala Ala Leu Ser Gln Val Thr Asn Pro Asp Gly Ser
Thr Thr Asn Leu 290 295 300 Ile Phe Asp Val His Lys Tyr Leu Asp Ser
Asp Asn Ser Gly Thr His 305 310 315 320 Ala Glu Cys Thr Thr Asn Asn
Ile Asp Gly Ala Phe Ser Pro Leu Ala 325 330 335 Thr Trp Leu Arg Gln
Asn Asn Arg Gln Ala Ile Leu Thr Glu Thr Gly 340 345 350 Gly Gly Asn
Val Gln Ser Cys Ile Gln Asp Met Cys Gln Gln Ile Gln 355 360 365 Tyr
Leu Asn Gln Asn Ser Asp Val Tyr Leu Gly Tyr Val Gly Trp Gly 370 375
380 Ala Gly Ser Phe Asp Ser Thr Tyr Val Leu Thr Glu Thr Pro Thr Gly
385 390 395 400 Ser Gly Asn Ser Trp Thr Asp Thr Ser Leu Val Ser Ser
Cys Leu Ala 405 410 415 Arg Lys 15471PRTTrichoderma reesei 15Met
Ile Val Gly Ile Leu Thr Thr Leu Ala Thr Leu Ala Thr Leu Ala 1 5 10
15 Ala Ser Val Pro Leu Glu Glu Arg Gln Ala Cys Ser Ser Val Trp Gly
20 25 30 Gln Cys Gly Gly Gln Asn Trp Ser Gly Pro Thr Cys Cys Ala
Ser Gly 35 40 45 Ser Thr Cys Val Tyr Ser Asn Asp Tyr Tyr Ser Gln
Cys Leu Pro Gly 50 55 60 Ala Ala Ser Ser Ser Ser Ser Thr Arg Ala
Ala Ser Thr Thr Ser Arg 65 70 75 80 Val Ser Pro Thr Thr Ser Arg Ser
Ser Ser Ala Thr Pro Pro Pro Gly 85 90 95 Ser Thr Thr Thr Arg Val
Pro Pro Val Gly Ser Gly Thr Ala Thr Tyr 100 105 110 Ser Gly Asn Pro
Phe Val Gly Val Thr Pro Trp Ala Asn Ala Tyr Tyr 115 120 125 Ala Ser
Glu Val Ser Ser Leu Ala Ile Pro Ser Leu Thr Gly Ala Met 130 135 140
Ala Thr Ala Ala Ala Ala Val Ala Lys Val Pro Ser Phe Met Trp Leu 145
150 155 160 Asp Thr Leu Asp Lys Thr Pro Leu Met Glu Gln Thr Leu Ala
Asp Ile 165 170 175 Arg Thr Ala Asn Lys Asn Gly Gly Asn Tyr Ala Gly
Gln Phe Val Val 180 185 190 Tyr Asp Leu Pro Asp Arg Asp Cys Ala Ala
Leu Ala Ser Asn Gly Glu 195 200 205 Tyr Ser Ile Ala Asp Gly Gly Val
Ala Lys Tyr Lys Asn Tyr Ile Asp 210 215 220 Thr Ile Arg Gln Ile Val
Val Glu Tyr Ser Asp Ile Arg Thr Leu Leu 225 230 235 240 Val Ile Glu
Pro Asp Ser Leu Ala Asn Leu Val Thr Asn Leu Gly Thr 245 250 255 Pro
Lys Cys Ala Asn Ala Gln Ser Ala Tyr Leu Glu Cys Ile Asn Tyr 260 265
270 Ala Val Thr Gln Leu Asn Leu Pro Asn Val Ala Met Tyr Leu Asp Ala
275 280 285 Gly His Ala Gly Trp Leu Gly Trp Pro Ala Asn Gln Asp Pro
Ala Ala 290 295 300 Gln Leu Phe Ala Asn Val Tyr Lys Asn Ala Ser Ser
Pro Arg Ala Leu 305 310 315 320 Arg Gly Leu Ala Thr Asn Val Ala Asn
Tyr Asn Gly Trp Asn Ile Thr 325 330 335 Ser Pro Pro Ser Tyr Thr Gln
Gly Asn Ala Val Tyr Asn Glu Lys Leu 340 345 350 Tyr Ile His Ala Ile
Gly Pro Leu Leu Ala Asn His Gly Trp Ser Asn 355 360 365 Ala Phe Phe
Ile Thr Asp Gln Gly Arg Ser Gly Lys Gln Pro Thr Gly 370 375 380 Gln
Gln Gln Trp Gly Asp Trp Cys Asn Val Ile Gly Thr Gly Phe Gly 385 390
395 400 Ile Arg Pro Ser Ala Asn Thr Gly Asp Ser Leu Leu Asp Ser Phe
Val 405 410 415 Trp Val Lys Pro Gly Gly Glu Cys Asp Gly Thr Ser Asp
Ser Ser Ala 420 425 430 Pro Arg Phe Asp Ser His Cys Ala Leu Pro Asp
Ala Leu Gln Pro Ala 435 440 445 Pro Gln Ala Gly Ala Trp Phe Gln Ala
Tyr Phe Val Gln Leu Leu Thr 450 455 460 Asn Ala Asn Pro Ser Phe Leu
465 470 16514PRTTrichoderma reesei 16Met Tyr Arg Lys Leu Ala Val
Ile Ser Ala Phe Leu Ala Thr Ala Arg 1 5 10 15 Ala Gln Ser Ala Cys
Thr Leu Gln Ser Glu Thr His Pro Pro Leu Thr 20 25 30 Trp Gln Lys
Cys Ser Ser Gly Gly Thr Cys Thr Gln Gln Thr Gly Ser 35 40 45 Val
Val Ile Asp Ala Asn Trp Arg Trp Thr His Ala Thr Asn Ser Ser 50 55
60 Thr Asn Cys Tyr Asp Gly Asn Thr Trp Ser Ser Thr Leu Cys Pro Asp
65 70 75 80 Asn Glu Thr Cys Ala Lys Asn Cys Cys Leu Asp Gly Ala Ala
Tyr Ala 85 90 95 Ser Thr Tyr Gly Val Thr Thr Ser Gly Asn Ser Leu
Ser Ile Gly Phe 100 105 110 Val Thr Gln Ser Ala Gln Lys Asn Val Gly
Ala Arg Leu Tyr Leu Met 115 120 125 Ala Ser Asp Thr Thr Tyr Gln Glu
Phe Thr Leu Leu Gly Asn Glu Phe 130 135 140 Ser Phe Asp Val Asp Val
Ser Gln Leu Pro Cys Gly Leu Asn Gly Ala 145 150 155 160 Leu Tyr Phe
Val Ser Met Asp Ala Asp Gly Gly Val Ser Lys Tyr Pro 165 170 175 Thr
Asn Thr Ala Gly Ala Lys Tyr Gly Thr Gly Tyr Cys Asp Ser Gln 180 185
190 Cys Pro Arg Asp Leu Lys Phe Ile Asn Gly Gln Ala Asn Val Glu Gly
195 200 205 Trp Glu Pro Ser Ser Asn Asn Ala Asn Thr Gly Ile Gly Gly
His Gly 210 215 220 Ser Cys Cys Ser Glu Met Asp Ile Trp Glu Ala Asn
Ser Ile Ser Glu 225 230 235 240 Ala Leu Thr Pro His Pro Cys Thr Thr
Val Gly Gln Glu Ile Cys Glu 245 250 255 Gly Asp Gly Cys Gly Gly Thr
Tyr Ser Asp Asn Arg Tyr Gly Gly Thr 260 265 270 Cys Asp Pro Asp Gly
Cys Asp Trp Asn Pro Tyr Arg Leu Gly Asn Thr 275 280 285 Ser Phe Tyr
Gly Pro Gly Ser Ser Phe Thr Leu Asp Thr Thr Lys Lys 290 295 300 Leu
Thr Val Val Thr Gln Phe Glu Thr Ser Gly Ala Ile Asn Arg Tyr 305 310
315 320 Tyr Val Gln Asn Gly Val Thr Phe Gln Gln Pro Asn Ala Glu Leu
Gly 325 330 335 Ser Tyr Ser Gly Asn Glu Leu Asn Asp Asp Tyr Cys Thr
Ala Glu Glu 340 345 350 Ala Glu Phe Gly Gly Ser Ser Phe Ser Asp Lys
Gly Gly Leu Thr Gln 355 360 365 Phe Lys Lys Ala Thr Ser Gly Gly Met
Val Leu Val Met Ser Leu Trp 370 375 380 Asp Asp Tyr Tyr Ala Asn Met
Leu Trp Leu Asp Ser Thr Tyr Pro Thr 385 390 395 400 Asn Glu Thr Ser
Ser Thr Pro Gly Ala Val Arg Gly Ser Cys Ser Thr 405 410 415 Ser Ser
Gly Val Pro Ala Gln Val Glu Ser Gln Ser Pro Asn Ala Lys 420 425 430
Val Thr Phe Ser Asn Ile Lys Phe Gly Pro Ile Gly Ser Thr Gly Asn 435
440 445 Pro Ser Gly Gly Asn Pro Pro Gly Gly Asn Pro Pro Gly Thr Thr
Thr 450 455 460 Thr Arg Arg Pro Ala Thr Thr Thr Gly Ser Ser Pro Gly
Pro Thr Gln 465 470 475 480 Ser His Tyr Gly Gln Cys Gly Gly Ile Gly
Tyr Ser Gly Pro Thr Val 485 490 495 Cys Ala Ser Gly Thr Thr Cys Gln
Val Leu Asn Pro Tyr Tyr Ser Gln 500 505 510 Cys Leu
17459PRTTrichoderma reesei 17Met Ala Pro Ser Val Thr Leu Pro Leu
Thr Thr Ala Ile Leu Ala Ile 1 5 10 15 Ala Arg Leu Val Ala Ala Gln
Gln Pro Gly Thr Ser Thr Pro Glu Val 20 25 30 His Pro Lys Leu Thr
Thr Tyr Lys Cys Thr Lys Ser Gly Gly Cys Val 35 40 45 Ala Gln Asp
Thr Ser Val Val Leu Asp Trp Asn Tyr Arg Trp Met His 50 55 60 Asp
Ala Asn Tyr Asn Ser Cys Thr Val Asn Gly Gly Val Asn Thr Thr 65 70
75 80 Leu Cys Pro Asp Glu Ala Thr Cys Gly Lys Asn Cys Phe Ile Glu
Gly 85 90 95 Val Asp Tyr Ala Ala Ser Gly Val Thr Thr Ser Gly Ser
Ser Leu Thr 100 105 110 Met Asn Gln Tyr Met Pro Ser Ser Ser Gly Gly
Tyr Ser Ser Val Ser 115 120 125 Pro Arg Leu Tyr Leu Leu Asp Ser Asp
Gly Glu Tyr Val Met Leu Lys 130 135 140 Leu Asn Gly Gln Glu Leu Ser
Phe Asp Val Asp Leu Ser Ala Leu Pro 145 150 155 160 Cys Gly Glu Asn
Gly Ser Leu Tyr Leu Ser Gln Met Asp Glu Asn Gly 165 170 175 Gly Ala
Asn Gln Tyr Asn Thr Ala Gly Ala Asn Tyr Gly Ser Gly Tyr 180 185 190
Cys Asp Ala Gln Cys Pro Val Gln Thr Trp Arg Asn Gly Thr Leu Asn 195
200 205 Thr Ser His Gln Gly Phe Cys Cys Asn Glu Met Asp Ile Leu Glu
Gly 210 215 220 Asn Ser Arg Ala Asn Ala Leu Thr Pro His Ser Cys Thr
Ala Thr Ala 225 230 235 240 Cys Asp Ser Ala Gly Cys Gly Phe Asn Pro
Tyr Gly Ser Gly Tyr Lys 245 250 255 Ser Tyr Tyr Gly Pro Gly Asp Thr
Val Asp Thr Ser Lys Thr Phe Thr 260 265 270 Ile Ile Thr Gln Phe Asn
Thr Asp Asn Gly Ser Pro Ser Gly Asn Leu 275 280 285 Val Ser Ile Thr
Arg Lys Tyr Gln Gln Asn Gly Val Asp Ile Pro Ser 290 295 300 Ala Gln
Pro Gly Gly Asp Thr Ile Ser Ser Cys Pro Ser Ala Ser Ala 305 310 315
320 Tyr Gly Gly Leu Ala Thr Met Gly Lys Ala Leu Ser Ser Gly Met Val
325 330 335 Leu Val Phe Ser Ile Trp Asn Asp Asn Ser Gln Tyr Met Asn
Trp Leu 340 345 350 Asp Ser Gly Asn Ala Gly Pro Cys Ser Ser Thr Glu
Gly Asn Pro Ser 355 360 365 Asn Ile Leu Ala Asn Asn Pro Asn Thr His
Val Val Phe Ser Asn Ile 370 375 380 Arg Trp Gly Asp Ile Gly Ser Thr
Thr Asn Ser Thr Ala Pro Pro Pro 385 390 395 400 Pro Pro Ala Ser Ser
Thr Thr Phe Ser Thr Thr Arg Arg Ser Ser Thr 405 410 415 Thr Ser Ser
Ser Pro Ser Cys Thr Gln Thr His Trp Gly Gln Cys Gly 420 425 430 Gly
Ile Gly Tyr Ser Gly Cys Lys Thr Cys Thr Ser Gly Thr Thr Cys 435 440
445 Gln Tyr Ser Asn Asp Tyr Tyr Ser Gln Cys Leu 450 455
18234PRTTrichoderma reesei 18Met Lys Phe Leu Gln Val Leu Pro Ala
Leu Ile Pro Ala Ala Leu Ala 1 5 10 15 Gln Thr Ser Cys Asp Gln Trp
Ala Thr Phe Thr Gly Asn Gly Tyr Thr 20 25 30 Val Ser Asn Asn Leu
Trp Gly Ala Ser Ala Gly Ser Gly Phe Gly Cys 35 40 45 Val Thr Ala
Val Ser Leu Ser Gly Gly Ala Ser Trp His Ala Asp Trp 50 55 60 Gln
Trp Ser Gly Gly Gln Asn Asn Val Lys Ser Tyr Gln Asn Ser Gln 65 70
75 80 Ile Ala Ile Pro Gln Lys Arg Thr Val Asn Ser Ile Ser Ser Met
Pro 85 90 95 Thr Thr Ala Ser Trp Ser Tyr Ser Gly Ser Asn Ile Arg
Ala Asn Val 100 105 110 Ala Tyr Asp Leu Phe Thr Ala Ala Asn Pro Asn
His Val Thr Tyr Ser 115 120 125 Gly Asp Tyr Glu Leu Met Ile Trp Leu
Gly Lys Tyr Gly Asp Ile Gly 130 135 140 Pro Ile Gly Ser Ser Gln Gly
Thr Val Asn Val Gly Gly Gln Ser Trp 145 150 155 160 Thr Leu Tyr Tyr
Gly Tyr Asn Gly Ala Met Gln Val Tyr Ser Phe Val 165 170 175 Ala Gln
Thr Asn Thr Thr Asn Tyr Ser Gly Asp Val Lys Asn Phe Phe 180 185 190
Asn Tyr Leu Arg Asp Asn Lys Gly Tyr Asn Ala Ala Gly Gln Tyr Val 195
200 205 Leu Ser Tyr Gln Phe Gly Thr Glu Pro Phe Thr Gly Ser Gly Thr
Leu 210 215 220 Asn Val Ala Ser Trp Thr Ala Ser Ile Asn 225 230
19242PRTTrichoderma reesei 19Met Lys Ala Thr Leu Val Leu Gly Ser
Leu Ile Val Gly Ala Val Ser 1 5 10 15 Ala Tyr Lys Ala Thr Thr Thr
Arg Tyr Tyr Asp Gly Gln Glu Gly Ala 20 25 30 Cys Gly Cys Gly Ser
Ser Ser Gly Ala Phe Pro Trp Gln Leu Gly Ile 35 40 45 Gly Asn Gly
Val Tyr Thr Ala Ala Gly Ser Gln Ala Leu Phe Asp Thr 50 55 60 Ala
Gly Ala Ser Trp Cys Gly Ala Gly Cys Gly Lys Cys Tyr Gln Leu 65 70
75 80 Thr Ser Thr Gly Gln Ala Pro Cys Ser Ser Cys Gly Thr Gly Gly
Ala 85 90 95 Ala Gly Gln Ser Ile Ile Val Met Val Thr Asn Leu Cys
Pro Asn Asn 100 105 110 Gly Asn Ala Gln Trp Cys Pro Val Val Gly Gly
Thr Asn Gln Tyr Gly 115 120 125 Tyr Ser Tyr His Phe Asp Ile Met Ala
Gln Asn Glu Ile Phe Gly Asp 130 135 140 Asn Val Val Val Asp Phe Glu
Pro Ile Ala Cys Pro Gly Gln Ala Ala 145 150 155 160 Ser Asp Trp Gly
Thr Cys Leu Cys Val Gly Gln Gln Glu Thr Asp Pro 165 170 175 Thr Pro
Val Leu Gly Asn Asp Thr Gly Ser Thr Pro Pro Gly Ser Ser 180 185 190
Pro Pro Ala Thr Ser Ser Ser Pro Pro Ser Gly Gly Gly Gln Gln Thr 195
200 205 Leu Tyr Gly Gln Cys Gly Gly Ala Gly Trp Thr Gly Pro Thr Thr
Cys 210 215 220 Gln Ala Pro Gly Thr Cys Lys Val Gln Asn Gln Trp Tyr
Ser Gln Cys 225 230 235 240 Leu Pro 20838PRTTrichoderma reesei
20Met Lys Val Ser Arg Val Leu Ala Leu Val Leu Gly Ala Val Ile Pro 1
5 10 15 Ala His Ala Ala Phe Ser Trp Lys Asn Val Lys Leu Gly Gly Gly
Gly 20 25 30 Gly Phe Val Pro Gly Ile Ile Phe His Pro Lys Thr Lys
Gly Val Ala 35 40 45 Tyr Ala Arg Thr Asp Ile Gly Gly Leu Tyr Arg
Leu Asn Ala Asp Asp 50 55 60 Ser Trp Thr Ala Val Thr Asp Gly Ile
Ala Asp Asn Ala Gly Trp His 65 70 75 80 Asn Trp Gly Ile Asp Ala Val
Ala Leu Asp Pro Gln Asp Asp Gln Lys 85 90 95 Val Tyr Ala Ala Val
Gly Met Tyr Thr Asn Ser Trp Asp Pro Ser Asn 100 105 110 Gly Ala Ile
Ile Arg Ser Ser Asp Arg Gly Ala Thr Trp Ser Phe Thr 115 120 125 Asn
Leu Pro Phe Lys Val Gly Gly Asn Met Pro Gly Arg Gly Ala Gly 130 135
140 Glu Arg Leu Ala Val Asp Pro Ala Asn Ser Asn Ile Ile Tyr Phe Gly
145 150 155 160 Ala Arg Ser Gly Asn Gly Leu Trp Lys Ser Thr Asp Gly
Gly Val Thr 165 170 175 Phe Ser Lys Val Ser Ser Phe Thr Ala Thr Gly
Thr Tyr Ile Pro Asp 180 185 190 Pro Ser Asp Ser Asn Gly Tyr Asn Ser
Asp Lys Gln Gly Leu Met Trp 195
200 205 Val Thr Phe Asp Ser Thr Ser Ser Thr Thr Gly Gly Ala Thr Ser
Arg 210 215 220 Ile Phe Val Gly Thr Ala Asp Asn Ile Thr Ala Ser Val
Tyr Val Ser 225 230 235 240 Thr Asn Ala Gly Ser Thr Trp Ser Ala Val
Pro Gly Gln Pro Gly Lys 245 250 255 Tyr Phe Pro His Lys Ala Lys Leu
Gln Pro Ala Glu Lys Ala Leu Tyr 260 265 270 Leu Thr Tyr Ser Asp Gly
Thr Gly Pro Tyr Asp Gly Thr Leu Gly Ser 275 280 285 Val Trp Arg Tyr
Asp Ile Ala Gly Gly Thr Trp Lys Asp Ile Thr Pro 290 295 300 Val Ser
Gly Ser Asp Leu Tyr Phe Gly Phe Gly Gly Leu Gly Leu Asp 305 310 315
320 Leu Gln Lys Pro Gly Thr Leu Val Val Ala Ser Leu Asn Ser Trp Trp
325 330 335 Pro Asp Ala Gln Leu Phe Arg Ser Thr Asp Ser Gly Thr Thr
Trp Ser 340 345 350 Pro Ile Trp Ala Trp Ala Ser Tyr Pro Thr Glu Thr
Tyr Tyr Tyr Ser 355 360 365 Ile Ser Thr Pro Lys Ala Pro Trp Ile Lys
Asn Asn Phe Ile Asp Val 370 375 380 Thr Ser Glu Ser Pro Ser Asp Gly
Leu Ile Lys Arg Leu Gly Trp Met 385 390 395 400 Ile Glu Ser Leu Glu
Ile Asp Pro Thr Asp Ser Asn His Trp Leu Tyr 405 410 415 Gly Thr Gly
Met Thr Ile Phe Gly Gly His Asp Leu Thr Asn Trp Asp 420 425 430 Thr
Arg His Asn Val Ser Ile Gln Ser Leu Ala Asp Gly Ile Glu Glu 435 440
445 Phe Ser Val Gln Asp Leu Ala Ser Ala Pro Gly Gly Ser Glu Leu Leu
450 455 460 Ala Ala Val Gly Asp Asp Asn Gly Phe Thr Phe Ala Ser Arg
Asn Asp 465 470 475 480 Leu Gly Thr Ser Pro Gln Thr Val Trp Ala Thr
Pro Thr Trp Ala Thr 485 490 495 Ser Thr Ser Val Asp Tyr Ala Gly Asn
Ser Val Lys Ser Val Val Arg 500 505 510 Val Gly Asn Thr Ala Gly Thr
Gln Gln Val Ala Ile Ser Ser Asp Gly 515 520 525 Gly Ala Thr Trp Ser
Ile Asp Tyr Ala Ala Asp Thr Ser Met Asn Gly 530 535 540 Gly Thr Val
Ala Tyr Ser Ala Asp Gly Asp Thr Ile Leu Trp Ser Thr 545 550 555 560
Ala Ser Ser Gly Val Gln Arg Ser Gln Phe Gln Gly Ser Phe Ala Ser 565
570 575 Val Ser Ser Leu Pro Ala Gly Ala Val Ile Ala Ser Asp Lys Lys
Thr 580 585 590 Asn Ser Val Phe Tyr Ala Gly Ser Gly Ser Thr Phe Tyr
Val Ser Lys 595 600 605 Asp Thr Gly Ser Ser Phe Thr Arg Gly Pro Lys
Leu Gly Ser Ala Gly 610 615 620 Thr Ile Arg Asp Ile Ala Ala His Pro
Thr Thr Ala Gly Thr Leu Tyr 625 630 635 640 Val Ser Thr Asp Val Gly
Ile Phe Arg Ser Thr Asp Ser Gly Thr Thr 645 650 655 Phe Gly Gln Val
Ser Thr Ala Leu Thr Asn Thr Tyr Gln Ile Ala Leu 660 665 670 Gly Val
Gly Ser Gly Ser Asn Trp Asn Leu Tyr Ala Phe Gly Thr Gly 675 680 685
Pro Ser Gly Ala Arg Leu Tyr Ala Ser Gly Asp Ser Gly Ala Ser Trp 690
695 700 Thr Asp Ile Gln Gly Ser Gln Gly Phe Gly Ser Ile Asp Ser Thr
Lys 705 710 715 720 Val Ala Gly Ser Gly Ser Thr Ala Gly Gln Val Tyr
Val Gly Thr Asn 725 730 735 Gly Arg Gly Val Phe Tyr Ala Gln Gly Thr
Val Gly Gly Gly Thr Gly 740 745 750 Gly Thr Ser Ser Ser Thr Lys Gln
Ser Ser Ser Ser Thr Ser Ser Ala 755 760 765 Ser Ser Ser Thr Thr Leu
Arg Ser Ser Val Val Ser Thr Thr Arg Ala 770 775 780 Ser Thr Val Thr
Ser Ser Arg Thr Ser Ser Ala Ala Gly Pro Thr Gly 785 790 795 800 Ser
Gly Val Ala Gly His Tyr Ala Gln Cys Gly Gly Ile Gly Trp Thr 805 810
815 Gly Pro Thr Gln Cys Val Ala Pro Tyr Val Cys Gln Lys Gln Asn Asp
820 825 830 Tyr Tyr Tyr Gln Cys Val 835 21373PRTPodospora anserina
21Met Lys Gly Leu Phe Ala Phe Gly Leu Gly Leu Leu Ser Leu Val Asn 1
5 10 15 Ala Leu Pro Gln Ala Gln Gly Gly Gly Ala Ala Ala Ser Ala Lys
Val 20 25 30 Ser Gly Thr Arg Phe Val Ile Asp Gly Lys Thr Gly Tyr
Phe Ala Gly 35 40 45 Thr Asn Ser Tyr Trp Ile Gly Phe Leu Thr Asn
Asn Arg Asp Val Asp 50 55 60 Thr Thr Leu Asp His Ile Ala Ser Ser
Gly Leu Lys Ile Leu Arg Val 65 70 75 80 Trp Gly Phe Asn Asp Val Asn
Asn Gln Pro Ser Gly Asn Thr Val Trp 85 90 95 Phe Gln Arg Leu Ala
Ser Ser Gly Ser Gln Ile Asn Thr Gly Pro Asn 100 105 110 Gly Leu Gln
Arg Leu Asp Tyr Leu Val Arg Ser Ala Glu Thr Arg Gly 115 120 125 Ile
Lys Leu Ile Ile Ala Leu Val Asn Tyr Trp Asp Asp Phe Gly Gly 130 135
140 Met Lys Ala Tyr Val Asn Ala Phe Gly Gly Thr Lys Glu Ser Trp Tyr
145 150 155 160 Thr Asn Ala Arg Ala Gln Glu Gln Tyr Lys Arg Tyr Ile
Gln Ala Val 165 170 175 Val Ser Arg Tyr Val Asn Ser Pro Ala Ile Phe
Ala Trp Glu Leu Ala 180 185 190 Asn Glu Pro Arg Cys Lys Gly Cys Asn
Thr Asn Val Ile Phe Asn Trp 195 200 205 Ala Thr Gln Ile Ser Asp Tyr
Ile Arg Ser Leu Asp Lys Asp His Leu 210 215 220 Ile Thr Leu Gly Asp
Glu Gly Phe Gly Leu Pro Gly Gln Thr Thr Tyr 225 230 235 240 Pro Tyr
Gln Tyr Gly Glu Gly Thr Asp Phe Val Lys Asn Leu Gln Ile 245 250 255
Lys Asn Leu Asp Phe Gly Thr Phe His Met Tyr Pro Gly His Trp Gly 260
265 270 Val Pro Thr Ser Phe Gly Pro Gly Trp Ile Lys Asp His Ala Ala
Ala 275 280 285 Cys Arg Ala Ala Gly Lys Pro Cys Leu Leu Glu Glu Tyr
Gly Tyr Glu 290 295 300 Ser Asp Arg Cys Asn Val Gln Lys Gly Trp Gln
Gln Ala Ser Arg Glu 305 310 315 320 Leu Ser Arg Asp Gly Met Ser Gly
Asp Leu Phe Trp Gln Trp Gly Asp 325 330 335 Gln Leu Ser Thr Gly Gln
Thr His Asn Asp Gly Phe Thr Ile Tyr Tyr 340 345 350 Gly Ser Ser Leu
Ala Thr Cys Leu Val Thr Asp His Val Arg Ala Ile 355 360 365 Asn Ala
Leu Pro Ala 370 22469PRTPodospora anserina 22Met Val Lys Leu Leu
Asp Ile Gly Leu Phe Ala Leu Ala Leu Ala Ser 1 5 10 15 Ser Ala Val
Ala Lys Pro Cys Lys Pro Arg Asp Gly Pro Val Thr Tyr 20 25 30 Glu
Ala Glu Asp Ala Ile Leu Thr Gly Thr Thr Val Asp Thr Ala Gln 35 40
45 Val Gly Tyr Thr Gly Arg Gly Tyr Val Thr Gly Phe Asp Glu Gly Ser
50 55 60 Asp Lys Ile Thr Phe Gln Ile Ser Ser Ala Thr Thr Lys Leu
Tyr Asp 65 70 75 80 Leu Ser Ile Arg Tyr Ala Ala Ile Tyr Gly Asp Lys
Arg Thr Asn Val 85 90 95 Val Leu Asn Asn Gly Ala Val Ser Glu Val
Phe Phe Pro Ala Gly Asp 100 105 110 Ser Phe Thr Ser Val Ala Ala Gly
Gln Val Leu Leu Asn Ala Gly Gln 115 120 125 Asn Thr Ile Asp Ile Val
Asn Asn Trp Gly Trp Tyr Leu Ile Asp Ser 130 135 140 Ile Thr Leu Thr
Pro Ser Ala Pro Arg Pro Pro His Asp Ile Asn Pro 145 150 155 160 Asn
Leu Asn Asn Pro Asn Ala Asp Thr Asn Ala Lys Lys Leu Tyr Ser 165 170
175 Tyr Leu Arg Ser Val Tyr Gly Asn Lys Ile Ile Ser Gly Gln Gln Glu
180 185 190 Leu His His Ala Glu Trp Ile Arg Gln Gln Thr Gly Lys Thr
Pro Ala 195 200 205 Leu Val Ala Val Asp Leu Met Asp Tyr Ser Pro Ser
Arg Val Glu Arg 210 215 220 Gly Thr Thr Ser His Ala Val Glu Asp Ala
Ile Ala His His Asn Ala 225 230 235 240 Gly Gly Ile Val Ser Val Leu
Trp His Trp Asn Ala Pro Val Gly Leu 245 250 255 Tyr Asp Thr Glu Glu
Asn Lys Trp Trp Ser Gly Phe Tyr Thr Arg Ala 260 265 270 Thr Asp Phe
Asp Ile Ala Ala Thr Leu Ala Asn Pro Gln Gly Ala Asn 275 280 285 Tyr
Thr Leu Leu Ile Arg Asp Ile Asp Ala Ile Ala Val Gln Leu Lys 290 295
300 Arg Leu Glu Ala Ala Gly Val Pro Val Leu Trp Arg Pro Leu His Glu
305 310 315 320 Ala Glu Gly Gly Trp Phe Trp Trp Gly Ala Lys Gly Pro
Glu Pro Ala 325 330 335 Lys Gln Leu Trp Asp Ile Leu Tyr Glu Arg Leu
Thr Val His His Gly 340 345 350 Leu Asp Asn Leu Ile Trp Val Trp Asn
Ser Ile Leu Glu Asp Trp Tyr 355 360 365 Pro Gly Asp Asp Thr Val Asp
Ile Leu Ser Ala Asp Val Tyr Ala Gln 370 375 380 Gly Asn Gly Pro Met
Ser Thr Gln Tyr Asn Glu Leu Ile Ala Leu Gly 385 390 395 400 Arg Asp
Lys Lys Met Ile Ala Ala Ala Glu Val Gly Ala Ala Pro Leu 405 410 415
Pro Gly Leu Leu Gln Ala Tyr Gln Ala Asn Trp Leu Trp Phe Ala Val 420
425 430 Trp Gly Asp Asp Phe Ile Asn Asn Pro Ser Trp Asn Thr Val Ala
Val 435 440 445 Leu Asn Glu Ile Tyr Asn Ser Asp Tyr Val Leu Thr Leu
Asp Glu Ile 450 455 460 Gln Gly Trp Arg Ser 465 23493PRTTrichoderma
reesei 23Met Ala Gly Lys Leu Ile Leu Val Ala Leu Ala Ser Leu Val
Ser Leu 1 5 10 15 Ser Ile Gln Gln Asn Cys Ala Ala Leu Phe Gly Gln
Cys Gly Gly Ile 20 25 30 Gly Trp Ser Gly Thr Thr Cys Cys Val Ala
Gly Ala Gln Cys Ser Phe 35 40 45 Val Asn Asp Trp Tyr Ser Gln Cys
Leu Ala Ser Thr Gly Gly Asn Pro 50 55 60 Pro Asn Gly Thr Thr Ser
Ser Ser Leu Val Ser Arg Thr Ser Ser Ala 65 70 75 80 Ser Ser Ser Val
Gly Ser Ser Ser Pro Gly Gly Asn Ser Pro Thr Gly 85 90 95 Ser Ala
Ser Thr Tyr Thr Thr Thr Asp Thr Ala Thr Val Ala Pro His 100 105 110
Ser Gln Ser Pro Tyr Pro Ser Ile Ala Ala Ser Ser Cys Gly Ser Trp 115
120 125 Thr Leu Val Asp Asn Val Cys Cys Pro Ser Tyr Cys Ala Asn Asp
Asp 130 135 140 Thr Ser Glu Ser Cys Ser Gly Cys Gly Thr Cys Thr Thr
Pro Pro Ser 145 150 155 160 Ala Asp Cys Lys Ser Gly Thr Met Tyr Pro
Glu Val His His Val Ser 165 170 175 Ser Asn Glu Ser Trp His Tyr Ser
Arg Ser Thr His Phe Gly Leu Thr 180 185 190 Ser Gly Gly Ala Cys Gly
Phe Gly Leu Tyr Gly Leu Cys Thr Lys Gly 195 200 205 Ser Val Thr Ala
Ser Trp Thr Asp Pro Met Leu Gly Ala Thr Cys Asp 210 215 220 Ala Phe
Cys Thr Ala Tyr Pro Leu Leu Cys Lys Asp Pro Thr Gly Thr 225 230 235
240 Thr Leu Arg Gly Asn Phe Ala Ala Pro Asn Gly Asp Tyr Tyr Thr Gln
245 250 255 Phe Trp Ser Ser Leu Pro Gly Ala Leu Asp Asn Tyr Leu Ser
Cys Gly 260 265 270 Glu Cys Ile Glu Leu Ile Gln Thr Lys Pro Asp Gly
Thr Asp Tyr Ala 275 280 285 Val Gly Glu Ala Gly Tyr Thr Asp Pro Ile
Thr Leu Glu Ile Val Asp 290 295 300 Ser Cys Pro Cys Ser Ala Asn Ser
Lys Trp Cys Cys Gly Pro Gly Ala 305 310 315 320 Asp His Cys Gly Glu
Ile Asp Phe Lys Tyr Gly Cys Pro Leu Pro Ala 325 330 335 Asp Ser Ile
His Leu Asp Leu Ser Asp Ile Ala Met Gly Arg Leu Gln 340 345 350 Gly
Asn Gly Ser Leu Thr Asn Gly Val Ile Pro Thr Arg Tyr Arg Arg 355 360
365 Val Gln Cys Pro Lys Val Gly Asn Ala Tyr Ile Trp Leu Arg Asn Gly
370 375 380 Gly Gly Pro Tyr Tyr Phe Ala Leu Thr Ala Val Asn Thr Asn
Gly Pro 385 390 395 400 Gly Ser Val Thr Lys Ile Glu Ile Lys Gly Ala
Asp Thr Asp Asn Trp 405 410 415 Val Ala Leu Val His Asp Pro Asn Tyr
Thr Ser Ser Arg Pro Gln Glu 420 425 430 Arg Tyr Gly Ser Trp Val Ile
Pro Gln Gly Ser Gly Pro Phe Asn Leu 435 440 445 Pro Val Gly Ile Arg
Leu Thr Ser Pro Thr Gly Glu Gln Ile Val Asn 450 455 460 Glu Gln Ala
Ile Lys Thr Phe Thr Pro Pro Ala Thr Gly Asp Pro Asn 465 470 475 480
Phe Tyr Tyr Ile Asp Ile Gly Val Gln Phe Ser Gln Asn 485 490
D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

S00001
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.