Analogues Of Hepcidin Mimetics With Improved In Vivo Half Lives
Bourne; Gregory Thomas ; et al.
U.S. patent application number 16/067568 was filed with the patent office on 2019-01-03 for analogues of hepcidin mimetics with improved in vivo half lives. The applicant listed for this patent is Protagonist Therapeutics, Inc.. Invention is credited to Ashok Bhandari, Gregory Thomas Bourne, Brian Troy Frederick, Mark Leslie Smythe.
| Application Number | 20190002503 16/067568 |
| Document ID | / |
| Family ID | 59225940 |
| Filed Date | 2019-01-03 |










View All Diagrams
| United States Patent Application | 20190002503 |
| Kind Code | A1 |
| Bourne; Gregory Thomas ; et al. | January 3, 2019 |
ANALOGUES OF HEPCIDIN MIMETICS WITH IMPROVED IN VIVO HALF LIVES
Abstract
The present invention provides hepcidin analogues with improved in vivo half-lives, and related pharmaceutical compositions and methods of use thereof.
| Inventors: | Bourne; Gregory Thomas; (Jindalee, AU) ; Smythe; Mark Leslie; (Bardon, Queensland, AU) ; Frederick; Brian Troy; (Ben Lomond, CA) ; Bhandari; Ashok; (Pleasanton, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 59225940 | ||||||||||
| Appl. No.: | 16/067568 | ||||||||||
| Filed: | December 29, 2016 | ||||||||||
| PCT Filed: | December 29, 2016 | ||||||||||
| PCT NO: | PCT/US16/69255 | ||||||||||
| 371 Date: | June 29, 2018 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 62273265 | Dec 30, 2015 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C07K 14/575 20130101; A61K 38/00 20130101; A61P 3/02 20180101; C07K 7/08 20130101; C07K 14/00 20130101 |
| International Class: | C07K 7/08 20060101 C07K007/08; C07K 14/575 20060101 C07K014/575; A61P 3/02 20060101 A61P003/02 |
Claims
1. A hepcidin analogue comprising a polypeptide sequence of Formula (I): X--Y (I) or a pharmaceutically acceptable salt or solvate thereof, wherein: X is a peptide sequence having the formula Xa: TABLE-US-00022 (Xa) (SEQ ID NO: 1) X1-Thr-His-X4-Pro-X6-X7-X8-Phe-X10
wherein X1 is Asp, isoGlu or Ida; X4 is Phe, Phe(4-F), Phe(4-CN), 4-BIP, Phe(4-OCH.sub.3), Tyr, Phe(2,3-(OCH.sub.3).sub.2), Phe(2,3-Cl.sub.2), or Dpa; X6 is Cys or Pen; X7 is any amino acid; X8 is Ile, Leu, Val, nLeu, Lys or Arg; and X10 is Lys, Glu or absent; and Y is absent or present; wherein if Y is present, Y is a peptide sequence having the formula Ya: TABLE-US-00023 (Ya) (SEQ ID NO: 2) Y1-Y2-Y3-Y4-Y5-Y6-Y7
wherein Y1 is amino acid Y2 is any amino acid Y3 is any amino acid Y4 is any amino acid Y5 is any amino acid; Y6 is Cys or Pen; and Y7 is Lys or absent; and wherein the hepcidin analogue comprises a conjugated half-life extension moiety, wherein the half-life extension moiety is optionally conjugated via a linker moiety.
2. The hepcidin analogue of claim 1, comprising one of more of the following: X1 is Asp; X4 is Phe or Dpa; X7 is Ile, Leu, Val, nLeu, or Lys; X7 is Ile or Lys; X8 is Lys or Arg; Y1 is Pro or hPro; Y1 is Pro; Y2 is Arg or Lys; Y2 is Arg; Y3 is Ser; Y4 is Lys, Arg or His; Y4 is Lys; or Y5 is Gly or Sar.
3. The hepcidin analogue of claim 1, wherein: X1 is Asp; X4 is Phe or Dpa; X7 is Ile, Leu, Val, nLeu, or Lys; X8 is Lys or Arg; Y1 is Pro or hPro; Y2 is Arg or Lys; Y3 is Ser; Y4 is Lys, Arg or His; and Y5 is Gly or Sar.
4. The hepcidin analogue of claim 3, wherein: X7 is Ile or Lys; Y1 is Pro; Y2 is Arg; and Y4 is Lys.
5. The hepcidin analogue of any one of claims 1-4, comprising a structure of Formula II: R.sup.1--X-L-Y--R.sup.2 (II) or a pharmaceutically acceptable salt or solvate thereof, wherein: R.sup.1 is hydrogen, a C1-C6 alkyl, a C6-C12 aryl, a C6-C12 aryl C1-C6 alkyl, or a C1-C20 alkanoyl, and including PEGylated versions alone or as spacers of any of the foregoing; R.sup.2 is OH or NH.sub.2; X is a peptide sequence having the formula Xa; L is absent, a bond, or a linker moiety; and Y is absent or present; provided that if Y is present, Y is a peptide having the formula Ya.
6. The hepcidin analogue of any one of claims 1-5, wherein X or Y further comprise one to three additional amino acids at the N-terminus or C-terminus.
7. The hepcidin analogue of any one of claims 1-6, wherein Y is present.
8. The hepcidin analogue of claim 7, comprising a disulfide bond between X6 and Y6.
9. The hepcidin analogue of claim 7 or claim 8, wherein L is a bond.
10. The hepcidin analogue of any one of claims 7-9, comprising a half-life extension moiety conjugated to a Lys at X8 or X10.
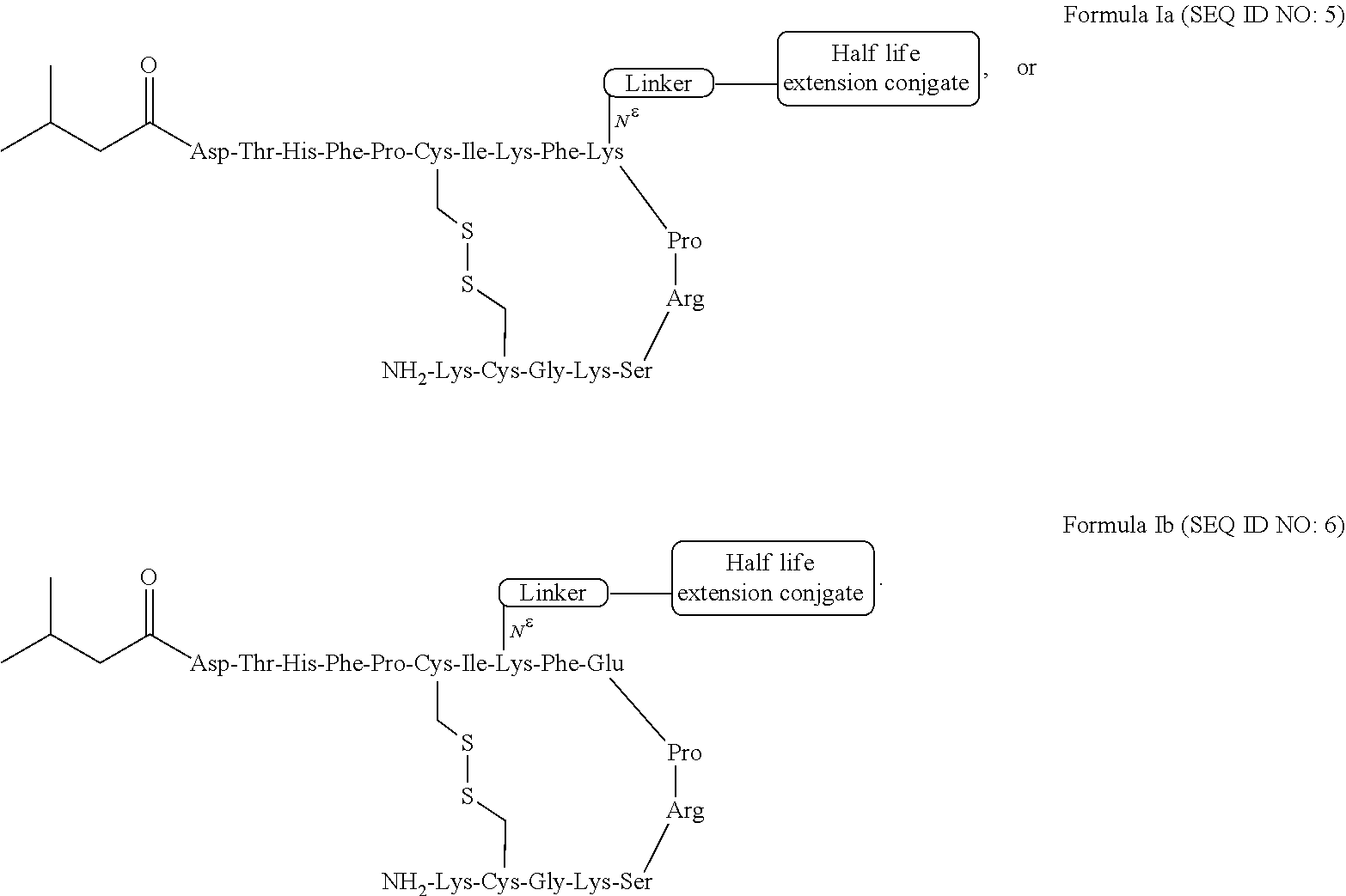
11. The hepcidin analogue of any one of claims 1-10, wherein the hepcidin analogue comprises one of the following structures: ##STR00048##
12. The hepcidin analogue of any one of claims 1-6, wherein Y is absent.
13. The hepcidin analogue of claim 12, wherein X10 is absent.
14. A dimer comprising two hepcidin analogues of claim 12 or claim 13, wherein the two polypeptide sequence of Formula I or two structures of Formula II are dimerized via a linker moiety.
15. The dimer of claim 14, wherein the linker moiety is bound to the C-terminus of each hepcidin analogue.
16. The dimer of claim 14 or claim 15, wherein the half-life extension moiety is conjugated to the linker moiety.
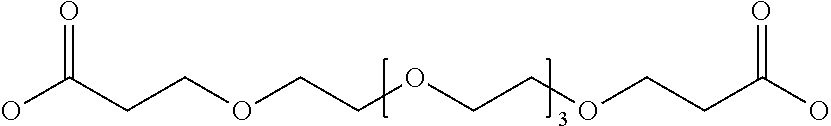
17. The hepcidin analogue of any one of claims 1-13 or the dimer of any one of claims 14-16, wherein the linker moiety is selected from IsoGlu, Dapa, PEGn where n=1 to 25, PEG11(40 atoms), OEG, IsoGlu-Ahx, IsoGlu-OEG-OEG, IsoGlu-PEG5, IsoGlu-PEGn where n=1 to 25 .beta.Ala-PEG2, and .beta.Ala-PEG11(40 atoms).
18. The hepcidin analogue or dimer of any one of claims 1-17, wherein the half-life extension moiety is selected from C12 (Lauric acid), C14 (Mysteric acid), C16 (Palmitic acid), C18 (Stearic acid, C20, C12 diacid, C14 diacid, C16 diacid, C18 diacid, C20 diacid, biotin, and isovaleric acid.
19. The hepcidin analogue or dimer of any one of claims 1-18, wherein the half-life extension moiety is attached to a linker moiety that is attached to the peptide.
20. The hepcidin analogue or dimer of any one of claims 1-19, wherein the half-life extension moiety increases the molecular weight of the hepcidin analogue by about 50 D to about 2 KD.
21. The hepcidin analogue or dimer of any one of claims 1-20, wherein the half-life extension moiety increases serum half-life, enhances solubility, and/or improves bioavailability of the hepcidin analogue.
22. The hepcidin analogue or dimer of any one of claims 1-21, comprising an isovaleric acid moiety conjugated to the N-terminal Asp residue.
23. The hepcidin analogue or dimer of any one of claims 1-22, comprising an amidated C-terminal residue.
24. The hepcidin analogue or dimer of any one of claims 1-23, comprises the sequence: TABLE-US-00024 (SEQ ID NO: 6) Asp-Thr-His-Phe-Pro-Cys-Ile-Lys-Phe-Glu-Pro-Arg- Ser-Lys-Gly-Cys-Lys.
25. The hepcidin analogue or dimer of any one of claims 1-23, comprising the sequence: TABLE-US-00025 (SEQ ID NO: 5) Asp-Thr-His-Phe-Pro-Cys-Ile-Lys-Phe-Lys-Pro-Arg- Ser-Lys-Gly-Cys-Lys.
26. A polynucleotide encoding a peptide of the hepcidin analogue of any one of claims 1-25.
27. A vector comprising the polynucleotide of claim 26.
28. A pharmaceutical composition comprising the hepcidin analogue or dimer of any one of claims 1-25, the polynucleotide of claim 26, or the vector of claim 27, and a pharmaceutically acceptable carrier, excipient or vehicle.
29. A method of binding a ferroportin or inducing ferroportin internalization and degradation, comprising contacting the ferroportin with at least one hepcidin analogue or dimer of any one of claims 1-25 or a composition of claim 28.
30. A method for treating a disease of iron metabolism in a subject in need thereof comprising providing to the subject an effective amount of the pharmaceutical composition of claim 28.
31. The method of claim 30, wherein the pharmaceutical composition is provided to the subject by an oral, intravenous, peritoneal, intradermal, subcutaneous, intramuscular, intrathecal, inhalation, vaporization, nebulization, sublingual, buccal, parenteral, rectal, vaginal, or topical route of administration.
32. The method of claim 31, wherein the pharmaceutical composition is provided to the subject by an oral or subcutaneous route of administration.
33. The method of any one of claims 30-32, wherein the disease of iron metabolism is an iron overload disease.
34. The method of any one of claims 30-33, wherein the pharmaceutical composition is provided to the subject at most twice daily, at most once daily, at most once every two days, at most once a week, or at most once a month.
35. The method of any one of claims 30-34, wherein the hepcidin analogue is provided to the subject at a dosage of about 1 mg to about 100 mg.
36. A device comprising the pharmaceutical composition of claim 28, for delivery of the hepcidin analogue to a subject, optionally orally or subcutaneously.
37. A kit comprising the pharmaceutical composition of claim 28, packaged with a reagent, a device, or an instructional material, or a combination thereof.
Description
CROSS REFERENCE TO RELATED APPLICATIONS
[0001] This application claims priority to U.S. Provisional Application No. 62/273,265, filed on Dec. 30, 2015, which is incorporated by reference herein in its entirety.
STATEMENT REGARDING SEQUENCE LISTING
[0002] The Sequence Listing associated with this application is provided in text format in lieu of a paper copy, and is hereby incorporated by reference into the specification. The name of the text file containing the Sequence Listing is PRTH_022_01WO_ST25.txt. The text file is 17 KB, was created on Dec. 29, 2016, and is being submitted electronically via EFS-Web.
FIELD OF THE INVENTION
[0003] The present invention relates, inter alia, to certain hepcidin peptide analogues, including both peptide monomers and peptide dimers, and conjugates and derivatives thereof, as well as compositions comprising the peptide analogues, and to the use of the peptide analogues in the treatment and/or prevention of a variety of diseases, conditions or disorders, including treatment and/or prevention of iron overload diseases such as hereditary hemochromatosis, iron-loading anemias, and other conditions and disorders described herein.
BACKGROUND
[0004] Hepcidin (also referred to as LEAP-1), a peptide hormone produced by the liver, is a regulator of iron homeostasis in humans and other mammals. Hepcidin acts by binding to its receptor, the iron export channel ferroportin, causing its internalization and degradation. Human hepcidin is a 25-amino acid peptide (Hep25). See Krause et al. (2000) FEBS Lett 480:147-150, and Park et al. (2001) J Biol Chem 276:7806-7810. The structure of the bioactive 25-amino acid form of hepcidin is a simple hairpin with 8 cysteines that form 4 disulfide bonds as described by Jordan et al. J Biol Chem 284:24155-67. The N terminal region is required for iron-regulatory function, and deletion of 5 N-terminal amino acid residues results in a loss of iron-regulatory function. See Nemeth et al. (2006) Blood 107:328-33.
[0005] Abnormal hepcidin activity is associated with iron overload diseases, including hereditary hemochromatosis (HH) and iron-loading anemias. Hereditary hemochromatosis is a genetic iron overload disease that is mainly caused by hepcidin deficiency or in some cases by hepcidin resistance. This allows excessive absorption of iron from the diet and development of iron overload. Clinical manifestations of HH may include liver disease (e.g., hepatic cirrhosis and hepatocellular carcinoma), diabetes, and heart failure. Currently, the only treatment for HH is regular phlebotomy, which is very burdensome for the patients. Iron-loading anemias are hereditary anemias with ineffective erythropoiesis such as .beta.-thalassemia, which are accompanied by severe iron overload. Complications from iron overload are the main cause of morbidity and mortality for these patients. Hepcidin deficiency is the main cause of iron overload in non-transfused patients, and contributes to iron overload in transfused patients. The current treatment for iron overload in these patients is iron chelation which is very burdensome, sometimes ineffective, and accompanied by frequent side effects.
[0006] Hepcidin has a number of limitations which restrict its use as a drug, including a difficult synthesis process due in part to aggregation and precipitation of the protein during folding, which in turn leads to high cost of goods. What are needed in the art are compounds having hepcidin activity and also possessing other beneficial physical properties such as improved solubility, stability, and/or potency, so that hepcidin-like biologics might be produced affordably, and used to treat hepcidin-related diseases and disorders such as, e.g., those described herein.
[0007] The present invention addresses such needs, providing novel peptide analogues, including both peptide monomer analogues and peptide dimer analogues, having hepcidin activity and also having other beneficial properties making the peptides of the present invention suitable alternatives to hepcidin.
BRIEF SUMMARY OF THE INVENTION
[0008] The present invention generally relates to peptide analogues, including both monomer and dimers, exhibiting hepcidin activity and methods of using the same.
[0009] In one embodiment, the present invention includes a hepcidin analogue comprising a polypeptide sequence of Formula (I):
X--Y (I)
or a pharmaceutically acceptable salt or solvate thereof, wherein: X is a peptide comprising the sequence Xa:
TABLE-US-00001 (Xa) (SEQ ID NO: 1) X1-Thr-His-X4-Pro-X6-X7-X8-Phe-X10
[0010] wherein [0011] X1 is Asp, isoGlu or Ida; [0012] X4 is Phe, Phe(4-F), Phe(4-CN), 4-BIP, Phe(4-OCH.sub.3), Tyr, Phe(2,3-(OCH.sub.3).sub.2), Phe(2,3-Cl.sub.2), or Dpa; [0013] X6 is Cys or Pen; [0014] X7 is any amino acid; [0015] X8 is Ile, Leu, Val, nLeu, Lys or Arg; and [0016] X10 is Lys, Glu or absent; and Y is absent or present; wherein if Y is present, Y is a peptide comprising the sequence Ya:
TABLE-US-00002 [0016] (Ya) (SEQ ID NO: 2) Y1-Y2-Y3-Y4-Y5-Y6-Y7
[0017] wherein [0018] Y1 is amino acid [0019] Y2 is any amino acid [0020] Y3 is any amino acid [0021] Y4 is any amino acid [0022] Y5 is any amino acid; [0023] Y6 is Cys or Pen; and [0024] Y7 is Lys or absent; and wherein the hepcidin analogue comprises a conjugated half-life extension moiety, wherein the half-life extension moiety is optionally conjugated via a linker moiety. The peptides Xa and Ya may be linked via a peptide bond to form the polypeptide of Formula (I).
[0025] In particular embodiments of any of the hepcidin analogues, the hepcidin analogue comprises one of more of the following: X1 is Asp; X4 is Phe or Dpa; X7 is Ile, Leu, Val, nLeu, or Lys; X7 is Ile or Lys; X8 is Lys or Arg; Y1 is Pro or hPro; Y1 is Pro; Y2 is Arg or Lys; Y2 is Arg; Y3 is Ser; Y4 is Lys, Arg or His; Y4 is Lys; or Y5 is Gly or Sar. In certain embodiments: X1 is Asp; X4 is Phe or Dpa; X7 is Ile, Leu, Val, nLeu, or Lys; X8 is Lys or Arg; Y1 is Pro or hPro; Y2 is Arg or Lys; Y3 is Ser; Y4 is Lys, Arg or His; and Y5 is Gly or Sar. In certain embodiments, X1 is Asp; X4 is Phe or Dpa; X7 is Ile or Lys; X8 is Lys or Arg; Y1 is Pro; Y2 is Arg; Y3 is Ser; and Y4 is Lys; and Y5 is Gly or Sar.
[0026] In a related embodiments, the present invention includes a hepcidin analogue comprising a polypeptide sequence of Formula (V):
X--Y (V)
or a pharmaceutically acceptable salt or solvate thereof, wherein: X is a peptide sequence having the formula Xv:
TABLE-US-00003 (Xv) (SEQ ID NO: 3) Asp-Thr-His-X4-Pro-X6-X7-X8-Phe-X10
[0027] wherein [0028] X4 is Phe or Dpa; [0029] X6 is Cys or Pen; [0030] X7 is Ile or Lys; [0031] X8 is Lys or Arg; and [0032] X10 is Lys, Glu or absent; and Y is absent or present; wherein if Y is present, Y is a peptide sequence having the formula Yv:
TABLE-US-00004 [0032] (Yv) (SEQ ID NO: 4) Pro-Arg-Ser-Lys-Y5-Y6-Y7
[0033] wherein [0034] Y5 is Gly or Sar; [0035] Y6 is Cys or Pen; and [0036] Y7 is Lys or absent; and wherein the hepcidin analogue comprises a conjugated half-life extension moiety, wherein the half-life extension moiety is optionally conjugated via a linker moiety. The peptides Xv and Yv may be linked via a peptide bond to form the polypeptide of Formula (V).
[0037] In certain embodiment, any of the hepcidin analogues comprises a structure of Formula II:
R.sup.1--X-L-Y--R.sup.2 (II)
or a pharmaceutically acceptable salt or solvate thereof, wherein: R.sup.1 is hydrogen, a C1-C6 alkyl, a C6-C12 aryl, a C6-C12 aryl C1-C6 alkyl, or a C1-C20 alkanoyl, and including PEGylated versions alone or as spacers of any of the foregoing; R.sup.2 is OH or NH.sub.2; X is a peptide sequence having the formula Xa (SEQ ID NO:1) or Xv (SEQ ID NO:3); L is absent, a bond, or a linker moiety; and Y is absent or present; provided that if Y is present, Y is a peptide having the formula Ya (SEQ ID NO:2) or Yv (SEQ ID NO:4).
[0038] In particular embodiments of any of the hepcidin analogues, X or Y further comprise one to three additional amino acids at the N-terminus or C-terminus.
[0039] In certain embodiments, Y is present. In particular embodiments, the hepcidin analogue comprises a disulfide bond between X6 and Y6.
[0040] In particular embodiments, L is a bond.
[0041] In various embodiments, a hepcidin analogue comprises a half-life extension moiety conjugated to a Lys at X8 or X10.
[0042] In certain embodiments, a hepcidin analogue comprises one of the following structures:
##STR00001##
[0043] In particular embodiments of the hepcidin analogues, Y is absent. In certain embodiments, X10 is absent.
[0044] In a related embodiment, the present invention includes a dimer comprising two hepcidin analogues of the present invention, e.g., two polypeptide sequence of Formula I or Formula V, or two structures of Formula II, dimerized via a linker moiety. In particular embodiments, the linker moiety is bound to the C-terminus of each hepcidin analogue. In certain embodiments, the half-life extension moiety is conjugated to the linker moiety.
[0045] In particular embodiments of any of the hepcidin analogues or dimers of the present invention, the linker moiety is selected from IsoGlu, Dapa, PEGn where n=1 to 25, PEG11 (40 atoms), OEG, IsoGlu-Ahx, IsoGlu-OEG-OEG, IsoGlu-PEG5, IsoGlu-PEGn where n=1 to 25 .beta.Ala-PEG2, and .beta.Ala-PEG11(40 atoms). In certain embodiments, more than one linker moiety is conjugated to a peptide of the hepcidin analogue or dimer.
[0046] In particular embodiments of any of the hepcidin analogues or dimers of the present invention, the half-life extension moiety is selected from C12 (Lauric acid), C14 (Mysteric acid), C16 (Palmitic acid), C18 (Stearic acid), C20, C12 diacid, C14 diacid, C16 diacid, C18 diacid, C20 diacid, biotin, and isovaleric acid. In certain embodiments, the half-life extension moiety is attached to a linker moiety that is attached to the peptide. In certain embodiments, the half-life extension moiety increases the molecular weight of the hepcidin analogue by about 50 D to about 2 KD. In various embodiments, the half-life extension moiety increases serum half-life, enhances solubility, and/or improves bioavailability of the hepcidin analogue.
[0047] In certain embodiments, a peptide analogue or dimer of the present invention comprises an isovaleric acid moiety conjugated to an N-terminal Asp residue.
[0048] In certain embodiments, a peptide analogue of the present invention comprises an amidated C-terminal residue.
[0049] In certain embodiments, a hepcidin analogue or dimer of the present invention comprises the sequence: Asp-Thr-His-Phe-Pro-Cys-Ile-Lys-Phe-Glu-Pro-Arg-Ser-Lys-Gly-Cys-Lys (SEQ ID NO:6), or comprises a sequence having at least 80%, at least 90%, or at least 94% identity to this sequence.
[0050] In certain embodiments, a hepcidin analogue or dimer of the present invention comprises the sequence: Asp-Thr-His-Phe-Pro-Cys-Ile-Lys-Phe-Lys-Pro-Arg-Ser-Lys-Gly-Cys-Lys (SEQ ID NO:5), or comprises a sequence having at least 80%, at least 90%, or at least 94% identity to this sequence.
[0051] In a related embodiment, the present invention includes a polynucleotide that encodes a peptide of a hepcidin analogue or dimer (or monomer subunit of a dimer) of the present invention.
[0052] In a further related embodiment, the present invention includes a vector comprising a polynucleotide of the invention.
[0053] In another embodiment, the present invention includes a pharmaceutical composition, comprising a hepcidin analogue, dimer, polynucleotide, or vector of the present invention, and a pharmaceutically acceptable carrier, excipient or vehicle.
[0054] In another embodiments, the present invention provides a method of binding a ferroportin or inducing ferroportin internalization and degradation, comprising contacting the ferroportin with at least one hepcidin analogue, dimer or composition of the present invention.
[0055] In a further embodiment, the present invention includes a method for treating a disease of iron metabolism in a subject in need thereof comprising providing to the subject an effective amount of a pharmaceutical composition of the present invention. In certain embodiments, the pharmaceutical composition is provided to the subject by an oral, intravenous, peritoneal, intradermal, subcutaneous, intramuscular, intrathecal, inhalation, vaporization, nebulization, sublingual, buccal, parenteral, rectal, vaginal, or topical route of administration. In certain embodiments, the pharmaceutical composition is provided to the subject by an oral or subcutaneous route of administration. In certain embodiments, the disease of iron metabolism is an iron overload disease. In certain embodiments, the pharmaceutical composition is provided to the subject at most or about twice daily, at most or about once daily, at most or about once every two days, at most or about once a week, or at most or about once a month.
[0056] In particular embodiments, the hepcidin analogue is provided to the subject at a dosage of about 1 mg to about 100 mg or about 1 mg to about 5 mg.
[0057] In another embodiment, the present invention provides a device comprising pharmaceutical composition of the present invention, for delivery of a hepcidin analogue or dimer of the invention to a subject, optionally orally or subcutaneously.
[0058] In yet another embodiment, the present invention includes a kit comprising a pharmaceutical composition of the invention, packaged with a reagent, a device, or an instructional material, or a combination thereof.
BRIEF DESCRIPTION OF THE DRAWINGS
[0059] FIGS. 1A and 1B are graphs showing serum iron levels at the indicated times following administration to mice of 1 uM Compound 1 either intravenously (FIG. 1A) or subcutaneously (FIG. 1B).
[0060] FIG. 2 is a graph showing serum iron levels at 30 hours or 36 hours following subcutaneous administration to healthy mice of 1 umol/kg of the indicated hepcidin analogues.
DETAILED DESCRIPTION OF THE INVENTION
[0061] The present invention relates generally to hepcidin analogue peptides and methods of making and using the same. In certain embodiments, the hepcidin analogues exhibit one or more hepcidin activity. In certain embodiments, the present invention relates to hepcidin peptide analogues comprising one or more peptide subunit that forms a cyclized structures through an intramolecular bond, e.g., an intramolecular disulfide bond. In particular embodiments, the cyclized structure has increased potency and selectivity as compared to non-cyclized hepcidin peptides and analogies thereof. In particular embodiments, hepcidin analogue peptides of the present invention exhibit increased half-lives, e.g., when delivered orally, as compared to hepcidin or previous hepcidin analogues.
Definitions and Nomenclature
[0062] Unless otherwise defined herein, scientific and technical terms used in this application shall have the meanings that are commonly understood by those of ordinary skill in the art. Generally, nomenclature used in connection with, and techniques of, chemistry, molecular biology, cell and cancer biology, immunology, microbiology, pharmacology, and protein and nucleic acid chemistry, described herein, are those well-known and commonly used in the art.
[0063] As used herein, the following terms have the meanings ascribed to them unless specified otherwise.
[0064] Throughout this specification, the word "comprise" or variations such as "comprises" or "comprising" will be understood to imply the inclusion of a stated integer (or components) or group of integers (or components), but not the exclusion of any other integer (or components) or group of integers (or components).
[0065] The singular forms "a," "an," and "the" include the plurals unless the context clearly dictates otherwise.
[0066] The term "including" is used to mean "including but not limited to." "Including" and "including but not limited to" are used interchangeably.
[0067] The terms "patient," "subject," and "individual" may be used interchangeably and refer to either a human or a non-human animal. These terms include mammals such as humans, primates, livestock animals (e.g., bovines, porcines), companion animals (e.g., canines, felines) and rodents (e.g., mice and rats). The term "mammal" refers to any mammalian species such as a human, mouse, rat, dog, cat, hamster, guinea pig, rabbit, livestock, and the like.
[0068] The term "peptide," as used herein, refers broadly to a sequence of two or more amino acids joined together by peptide bonds. It should be understood that this term does not connote a specific length of a polymer of amino acids, nor is it intended to imply or distinguish whether the polypeptide is produced using recombinant techniques, chemical or enzymatic synthesis, or is naturally occurring.
[0069] The term "peptide analogue," as used herein, refers broadly to peptide monomers and peptide dimers comprising one or more structural features and/or functional activities in common with hepcidin, or a functional region thereof. In certain embodiments, a peptide analogue includes peptides sharing substantial amino acid sequence identity with hepcidin, e.g., peptides that comprise one or more amino acid insertions, deletions, or substitutions as compared to a wild-type hepcidin, e.g., human hepcidin, amino acid sequence. In certain embodiments, a peptide analogue comprises one or more additional modification, such as, e.g., conjugation to another compound. Encompassed by the term "peptide analogue" is any peptide monomer or peptide dimer of the present invention. In certain instances, a "peptide analog" may also or alternatively be referred to herein as a "hepcidin analogue," "hepcidin peptide analogue," or a "hepcidin analogue peptide."
[0070] The recitations "sequence identity", "percent identity", "percent homology", or, for example, comprising a "sequence 50% identical to," as used herein, refer to the extent that sequences are identical on a nucleotide-by-nucleotide basis or an amino acid-by-amino acid basis over a window of comparison. Thus, a "percentage of sequence identity" may be calculated by comparing two optimally aligned sequences over the window of comparison, determining the number of positions at which the identical nucleic acid base (e.g., A, T, C, G, I) or the identical amino acid residue (e.g., Ala, Pro, Ser, Thr, Gly, Val, Leu, Ile, Phe, Tyr, Trp, Lys, Arg, His, Asp, Glu, Asn, Gln, Cys and Met) occurs in both sequences to yield the number of matched positions, dividing the number of matched positions by the total number of positions in the window of comparison (i.e., the window size), and multiplying the result by 100 to yield the percentage of sequence identity.
[0071] Calculations of sequence similarity or sequence identity between sequences (the terms are used interchangeably herein) can be performed as follows. To determine the percent identity of two amino acid sequences, or of two nucleic acid sequences, the sequences can be aligned for optimal comparison purposes (e.g., gaps can be introduced in one or both of a first and a second amino acid or nucleic acid sequence for optimal alignment and non-homologous sequences can be disregarded for comparison purposes). In certain embodiments, the length of a reference sequence aligned for comparison purposes is at least 30%, preferably at least 40%, more preferably at least 50%, 60%, and even more preferably at least 70%, 80%, 90%, 100% of the length of the reference sequence. The amino acid residues or nucleotides at corresponding amino acid positions or nucleotide positions are then compared. When a position in the first sequence is occupied by the same amino acid residue or nucleotide as the corresponding position in the second sequence, then the molecules are identical at that position.
[0072] The percent identity between the two sequences is a function of the number of identical positions shared by the sequences, taking into account the number of gaps, and the length of each gap, which need to be introduced for optimal alignment of the two sequences.
[0073] The comparison of sequences and determination of percent identity between two sequences can be accomplished using a mathematical algorithm. In some embodiments, the percent identity between two amino acid sequences is determined using the Needleman and Wunsch, (1970, J. Mol. Biol. 48: 444-453) algorithm which has been incorporated into the GAP program in the GCG software package, using either a Blossum 62 matrix or a PAM250 matrix, and a gap weight of 16, 14, 12, 10, 8, 6, or 4 and a length weight of 1, 2, 3, 4, 5, or 6. In yet another preferred embodiment, the percent identity between two nucleotide sequences is determined using the GAP program in the GCG software package, using an NWSgapdna.CMP matrix and a gap weight of 40, 50, 60, 70, or 80 and a length weight of 1, 2, 3, 4, 5, or 6. Another exemplary set of parameters includes a Blossum 62 scoring matrix with a gap penalty of 12, a gap extend penalty of 4, and a frameshift gap penalty of 5. The percent identity between two amino acid or nucleotide sequences can also be determined using the algorithm of E. Meyers and W. Miller (1989, Cabios, 4: 11-17) which has been incorporated into the ALIGN program (version 2.0), using a PAM120 weight residue table, a gap length penalty of 12 and a gap penalty of 4.
[0074] The peptide sequences described herein can be used as a "query sequence" to perform a search against public databases to, for example, identify other family members or related sequences. Such searches can be performed using the NBLAST and XBLAST programs (version 2.0) of Altschul, et al., (1990, J. Mol. Biol, 215: 403-10). BLAST nucleotide searches can be performed with the NBLAST program, score=100, wordlength=12 to obtain nucleotide sequences homologous to nucleic acid molecules of the invention. BLAST protein searches can be performed with the XBLAST program, score=50, wordlength=3 to obtain amino acid sequences homologous to protein molecules of the invention. To obtain gapped alignments for comparison purposes, Gapped BLAST can be utilized as described in Altschul et al. (Nucleic Acids Res. 25:3389-3402, 1997). When utilizing BLAST and Gapped BLAST programs, the default parameters of the respective programs (e.g., XBLAST and NBLAST) can be used.
[0075] The term "conservative substitution" as used herein denotes that one or more amino acids are replaced by another, biologically similar residue. Examples include substitution of amino acid residues with similar characteristics, e.g., small amino acids, acidic amino acids, polar amino acids, basic amino acids, hydrophobic amino acids and aromatic amino acids. See, for example, the table below. In some embodiments of the invention, one or more Met residues are substituted with norleucine (Nle) which is a bioisostere for Met, but which, as opposed to Met, is not readily oxidized. In some embodiments, one or more Trp residues are substituted with Phe, or one or more Phe residues are substituted with Trp, while in some embodiments, one or more Pro residues are substituted with L-isonipecotic acid (NPC), or one or more NPC residues are substituted with Pro. Another example of a conservative substitution with a residue normally not found in endogenous, mammalian peptides and proteins is the conservative substitution of Arg or Lys with, for example, ornithine, canavanine, aminoethylcysteine or another basic amino acid. In some embodiments, another conservative substitution is the substitution of one or more Pro residues with bhPro or Leu or D-isonipecotic acid (D-NPC).
[0076] For further information concerning phenotypically silent substitutions in peptides and proteins, see, for example, Bowie et. al. Science 247, 1306-1310, 1990. In the scheme below, conservative substitutions of amino acids are grouped by physicochemical properties. I: neutral, hydrophilic, II: acids and amides, III: basic, IV: hydrophobic, V: aromatic, bulky amino acids.
TABLE-US-00005 I II III IV V A N H M F S D R L Y T E K I W P Q V G C
[0077] In the scheme below, conservative substitutions of amino acids are grouped by physicochemical properties. VI: neutral or hydrophobic, VII: acidic, VIII: basic, IX: polar, X: aromatic.
TABLE-US-00006 VI VII VIII IX X A E H M F L D R S Y I K T W P C G N V Q
[0078] The term "amino acid" or "any amino acid" as used here refers to any and all amino acids, including naturally occurring amino acids (e.g., a-amino acids), unnatural amino acids, modified amino acids, and non-natural amino acids. It includes both D- and L-amino acids. Natural amino acids include those found in nature, such as, e.g., the 23 amino acids that combine into peptide chains to form the building-blocks of a vast array of proteins. These are primarily L stereoisomers, although a few D-amino acids occur in bacterial envelopes and some antibiotics. The 20 "standard," natural amino acids are listed in the above tables. The "non-standard," natural amino acids are pyrrolysine (found in methanogenic organisms and other eukaryotes), selenocysteine (present in many noneukaryotes as well as most eukaryotes), and N-formylmethionine (encoded by the start codon AUG in bacteria, mitochondria and chloroplasts). "Unnatural" or "non-natural" amino acids are non-proteinogenic amino acids (i.e., those not naturally encoded or found in the genetic code) that either occur naturally or are chemically synthesized. Over 140 natural amino acids are known and thousands of more combinations are possible. Examples of "unnatural" amino acids include .beta.-amino acids (.beta..sup.3 and .beta..sup.2), homo-amino acids, proline and pyruvic acid derivatives, 3-substituted alanine derivatives, glycine derivatives, ring-substituted phenylalanine and tyrosine derivatives, linear core amino acids, diamino acids, D-amino acids, and N-methyl amino acids. Unnatural or non-natural amino acids also include modified amino acids. "Modified" amino acids include amino acids (e.g., natural amino acids) that have been chemically modified to include a group, groups, or chemical moiety not naturally present on the amino acid.
[0079] As is clear to the skilled artisan, the peptide sequences disclosed herein are shown proceeding from left to right, with the left end of the sequence being the N-terminus of the peptide and the right end of the sequence being the C-terminus of the peptide. Among sequences disclosed herein are sequences incorporating a "Hy-" moiety at the amino terminus (N-terminus) of the sequence, and either an "--OH" moiety or an "--NH.sub.2" moiety at the carboxy terminus (C-terminus) of the sequence. In such cases, and unless otherwise indicated, a "Hy-" moiety at the N-terminus of the sequence in question indicates a hydrogen atom, corresponding to the presence of a free primary or secondary amino group at the N-terminus, while an "--OH" or an "--NH.sub.2" moiety at the C-terminus of the sequence indicates a hydroxy group or an amino group, corresponding to the presence of an amido (CONH.sub.2) group at the C-terminus, respectively. In each sequence of the invention, a C-terminal "--OH" moiety may be substituted for a C-terminal "--NH.sub.2" moiety, and vice-versa. It is further understood that the moiety at the amino terminus or carboxy terminus may be a bond, e.g., a covalent bond, particularly in situations where the amino terminus or carboxy terminus is bound to a linker or to another chemical moiety, e.g., a PEG moiety.
[0080] The term "NH.sub.2," as used herein, refers to the free amino group present at the amino terminus of a polypeptide. The term "OH," as used herein, refers to the free carboxy group present at the carboxy terminus of a peptide. Further, the term "Ac," as used herein, refers to Acetyl protection through acylation of the C- or N-terminus of a polypeptide.
[0081] The term "carboxy," as used herein, refers to --CO.sub.2H.
[0082] For the most part, the names of naturally occurring and non-naturally occurring aminoacyl residues used herein follow the naming conventions suggested by the IUPAC Commission on the Nomenclature of Organic Chemistry and the IUPAC-IUB Commission on Biochemical Nomenclature as set out in "Nomenclature of .alpha.-Amino Acids (Recommendations, 1974)" Biochemistry, 14(2), (1975). To the extent that the names and abbreviations of amino acids and aminoacyl residues employed in this specification and appended claims differ from those suggestions, they will be made clear to the reader. Some abbreviations useful in describing the invention are defined below in the following Table 2.
TABLE-US-00007 TABLE 2 Abbreviations of Non-Natural Amino Acids and Chemical Moieties Abbreviation Definition DIG Diglycolic acid Dapa Diaminopropionic acid Daba Diaminobutyric acid Pen Penicillamine Sarc or Sar Sarcosine Cit Citroline Cav Cavanine NMe-Arg N-Methyl-Arginine NMe-Trp N-Methyl-Tryptophan NMe-Phe N-Methyl-Phenylalanine Ac- Acetyl 2-Nal 2-Napthylalanine 1-Nal 1-Napthylalanine Bip Biphenylalanine .beta.Ala beta-Alanine Aib 2-aminoisobutyric acid Azt azetidine-2-carboxylic acid Tic (3S)-1,2,3,4-Tetrahydroisoquinoline- hydroxy-3-carboxylic acid Phe(OMe) Tyrosine (4-Methyl) N-MeLys N-Methyl-Lysine N-MeLys(Ac) N-e-Acetyl-D-lysine Dpa .beta.,.beta. diphenylalanine NH.sub.2 Free Amine CONH.sub.2 Amide COOH Acid Phe(4-F) 4-Fluoro-Phenylalanine PEG3 NH.sub.2CH.sub.2CH.sub.2(OCH.sub.2CH.sub.2).sub.3CH.sub.2CH.sub.2CO.s- ub.2H m-PEG3 CH.sub.3OCH.sub.2CH.sub.2(OCH.sub.2CH.sub.2).sub.2CH.sub.2CH.sub.2C- O.sub.2H m-PEG4 CH.sub.3OCH.sub.2CH.sub.2(OCH.sub.2CH.sub.2).sub.3CH.sub.2CH.sub.2C- O.sub.2H m-PEG8 CH.sub.3OCH.sub.2CH.sub.2(OCH.sub.2CH.sub.2).sub.7CH.sub.2CH.sub.2C- O.sub.2H PEG11 O-(2-aminoethyl)-O'-(2-carboxyethyl)- undecaethyleneglycol NH.sub.2CH.sub.2CH.sub.2(OCH.sub.2CH.sub.2).sub.11CH.sub.2CH.sub.2CO.sub.- 2H PEG13 Bifunctional PEG linker with 13 PolyEthylene Glycol units PEG25 Bifunctional PEG linker with 25 PolyEthylene Glycol units PEG1K Bifunctional PEG linker with PolyEthylene Glycol Mol wt of 1000 Da PEG2K Bifunctional PEG linker with PolyEthylene Glycol Mol wt of 2000 Da PEG3.4K Bifunctional PEG linker with PolyEthylene Glycol Mol wt of 3400 Da PEG5K Bifunctional PEG linker with PolyEthylene Glycol Mol wt of 5000 Da IDA or Ida Iminodiacetic acid IDA-Palm (Palmityl)-Iminodiacetic acid hPhe homoPhenylalanine Ahx Aminohexanoic acid DIG-OH Glycolic monoacid Triazine Amino propyl Triazine di-acid Boc-Triazine Boc-Triazine di-acid Trifluorobutyric acid 4,4,4-Trifluorobutyric acid 2-Methylltrifluorobutyric 2-methyl-4,4,4-Butyric acid acid Trifluorpentanoic acid 5,5,5-Trifluoropentanoic acid 1,4-Phenylenediacetic para-Phenylenediacetic acid acid 1,3-Phenylenediacetic meta-Phenylenediacetic acid acid DTT Dithiothreotol Nle Norleucine .beta.hTrp or bhTrp .beta.-homoTryptophane .beta.hPhe or bhPhe .beta.-homophenylalanine Phe(4-CF.sub.3) 4-TrifluoromethylPhenylalanine .beta.Glu or bGlu .beta.-Glutamic acid .beta.Glu or bhGlu .beta.-homoglutamic acid 2-2-Indane 2-Aminoindane-2-carboxylic acid 1-1-Indane 1-Aminoindane-1-carboxylic acid hCha homocyclohexylalanine Cyclobutyl Cyclobutylalanine hLeu Homoleucine Gla .gamma.-Carboxy-glutamic acid Aep 3-(2-aminoethoxy)propanoic acid Aea (2-aminoethoxy)acetic acid IsoGlu-octanoic acid octanoyl-.gamma.-Glu K-octanoic acid octanoyl-.epsilon.-Lys Dapa(Palm) Hexadecanoyl-.beta.-Diaminopropionic acid IsoGlu-Palm hexadecanoyl-.gamma.-Glu C-StBu S-tert-butylthio-cysteine C-tBu S-tert-butyl-cysteine Dapa(AcBr) NY-(bromoacetyl)-2,3-diaminopropionic acid Tle tert-Leucine Phg phenylglycine Oic octahydroindole-2-carboxylic acid Chg .alpha.-cyclohexylglycine GP-(Hyp) Gly-Pro-HydroxyPro Inp isonipecotic acid Amc 4-(aminomethyl)cyclohexane carboxylic acid Betaine (CH.sub.3).sub.3NCH.sub.2CH.sub.2CO2H NPC Isonipecotic acid D-NPC D-Isonipecotic acid
[0083] Throughout the present specification, unless naturally occurring amino acids are referred to by their full name (e.g. alanine, arginine, etc.), they are designated by their conventional three-letter or single-letter abbreviations (e.g. Ala or A for alanine, Arg or R for arginine, etc.). In the case of less common or non-naturally occurring amino acids, unless they are referred to by their full name (e.g. sarcosine, ornithine, etc.), frequently employed three- or four-character codes are employed for residues thereof, including, Sar or Sarc (sarcosine, i.e. N-methylglycine), Aib (.alpha.-aminoisobutyric acid), Daba (2,4-diaminobutanoic acid), Dapa (2,3-diaminopropanoic acid), .gamma.-Glu (.gamma.-glutamic acid), pGlu (pyroglutamic acid), Gaba (.gamma.-aminobutanoic acid), .beta.-Pro (pyrrolidine-3-carboxylic acid), 8Ado (8-amino-3,6-dioxaoctanoic acid), Abu (4-aminobutyric acid), bhPro (.beta.-homo-proline), bhPhe (.beta.-homo-L-phenylalanine), bhAsp (.beta.-homo-aspartic acid]), Dpa (.beta.,.beta. diphenylalanine), Ida (Iminodiacetic acid), hCys (homocysteine), bhDpa (.beta.-homo-.beta.,.beta.-diphenylalanine).
[0084] Furthermore, R.sup.1 can in all sequences be substituted with isovaleric acids or equivalent. In some embodiments, wherein a peptide of the present invention is conjugated to an acidic compound such as, e.g., isovaleric acid, isobutyric acid, valeric acid, and the like, the presence of such a conjugation is referenced in the acid form. So, for example, but not to be limited in any way, instead of indicating a conjugation of isovaleric acid to a peptide by referencing isovaleroyl, in some embodiments, the present application may reference such a conjugation as isovaleric acid.
[0085] The term "L-amino acid," as used herein, refers to the "L" isomeric form of a peptide, and conversely the term "D-amino acid" refers to the "D" isomeric form of a peptide. In certain embodiments, the amino acid residues described herein are in the "L" isomeric form, however, residues in the "D" isomeric form can be substituted for any L-amino acid residue, as long as the desired functional is retained by the peptide.
[0086] Unless otherwise indicated, reference is made to the L-isomeric forms of the natural and unnatural amino acids in question possessing a chiral center. Where appropriate, the D-isomeric form of an amino acid is indicated in the conventional manner by the prefix "D" before the conventional three-letter code (e.g. Dasp, (D)Asp or D-Asp; Dphe, (D)Phe or D-Phe).
[0087] The term "DRP," as used herein, refers to disulfide rich peptides.
[0088] The term "dimer," as used herein, refers broadly to a peptide comprising two or more monomer subunits. Certain dimers comprise two DRPs. Dimers of the present invention include homodimers and heterodimers. A monomer subunit of a dimer may be linked at its C- or N-terminus, or it may be linked via internal amino acid residues. Each monomer subunit of a dimer may be linked through the same site, or each may be linked through a different site (e.g., C-terminus, N-terminus, or internal site).
[0089] As used herein, in the context of certain peptide sequences disclosed herein, parentheticals, e.g., (______) represent side chain conjugations and brackets, e.g., [______], represent unnatural amino acid substitutions or amino acids and conjugated side chains. Generally, where a linker is shown at the N-terminus of a peptide sequence, it indicates that the peptide is dimerized with another peptide, wherein the linker is attached to the N-terminus of the two peptides. Generally, where a linker is shown at the C-terminus of a peptide sequence or structure, it indicates that the peptide is dimerized with another peptide, wherein the linker is attached to the C-terminus of the two peptides.
[0090] The term "isostere replacement" or "isostere substitution" are used interchangeably herein to refer to any amino acid or other analog moiety having chemical and/or structural properties similar to a specified amino acid. In certain embodiments, an isostere replacement is a conservative substitution with a natural or unnatural amino acid.
[0091] The term "cyclized," as used herein, refers to a reaction in which one part of a polypeptide molecule becomes linked to another part of the polypeptide molecule to form a closed ring, such as by forming a disulfide bridge or other similar bond.
[0092] The term "subunit," as used herein, refers to one of a pair of polypeptide monomers that are joined to form a dimer peptide composition.
[0093] The term "linker moiety," as used herein, refers broadly to a chemical structure that is capable of linking or joining together two peptide monomer subunits to form a dimer.
[0094] The term "solvate" in the context of the present invention refers to a complex of defined stoichiometry formed between a solute (e.g., a hepcidin analogue or pharmaceutically acceptable salt thereof according to the invention) and a solvent. The solvent in this connection may, for example, be water, ethanol or another pharmaceutically acceptable, typically small-molecular organic species, such as, but not limited to, acetic acid or lactic acid. When the solvent in question is water, such a solvate is normally referred to as a hydrate.
[0095] As used herein, a "disease of iron metabolism" includes diseases where aberrant iron metabolism directly causes the disease, or where iron blood levels are dysregulated causing disease, or where iron dysregulation is a consequence of another disease, or where diseases can be treated by modulating iron levels, and the like. More specifically, a disease of iron metabolism according to this disclosure includes iron overload diseases, iron deficiency disorders, disorders of iron biodistribution, other disorders of iron metabolism and other disorders potentially related to iron metabolism, etc. Diseases of iron metabolism include hemochromatosis, HFE mutation hemochromatosis, ferroportin mutation hemochromatosis, transferrin receptor 2 mutation hemochromatosis, hemojuvelin mutation hemochromatosis, hepcidin mutation hemochromatosis, juvenile hemochromatosis, neonatal hemochromatosis, hepcidin deficiency, transfusional iron overload, thalassemia, thalassemia intermedia, alpha thalassemia, sideroblastic anemia, porphyria, porphyria cutanea tarda, African iron overload, hyperferritinemia, ceruloplasmin deficiency, atransferrinemia, congenital dyserythropoietic anemia, anemia of chronic disease, anemia of inflammation, anemia of infection, hypochromic microcytic anemia, sickle cell disease, polycythemia vera (primary and secondary), myelodysplasia, pyruvate kinase deficiency, iron-deficiency anemia, iron-refractory iron deficiency anemia, anemia of chronic kidney disease, erythropoietin resistance, iron deficiency of obesity, other anemias, benign or malignant tumors that overproduce hepcidin or induce its overproduction, conditions with hepcidin excess, Friedreich ataxia, gracile syndrome, Hallervorden-Spatz disease, Wilson's disease, pulmonary hemosiderosis, hepatocellular carcinoma, cancer, hepatitis, cirrhosis of liver, pica, chronic renal failure, insulin resistance, diabetes, atherosclerosis, neurodegenerative disorders, multiple sclerosis, Parkinson's disease, Huntington's disease, and Alzheimer's disease.
[0096] In some embodiments, the disease and disorders are related to iron overload diseases such as iron hemochromatosis, HFE mutation hemochromatosis, ferroportin mutation hemochromatosis, transferrin receptor 2 mutation hemochromatosis, hemojuvelin mutation hemochromatosis, hepcidin mutation hemochromatosis, juvenile hemochromatosis, neonatal hemochromatosis, hepcidin deficiency, transfusional iron overload, thalassemia, thalassemia intermedia, alpha thalassemia, sickle cell disease, polycythemia vera (primary and secondary), mylodysplasia, and pyruvate kinase deficiency.
[0097] In some embodiments, the hepcidin analogues of the invention are used to treat diseases and disorders that are not typically identified as being iron related. For example, hepcidin is highly expressed in the murine pancreas suggesting that diabetes (Type I or Type II), insulin resistance, glucose intolerance and other disorders may be ameliorated by treating underlying iron metabolism disorders. See Ilyin, G. et al. (2003) FEBS Lett. 542 22-26, which is herein incorporated by reference. As such, peptides of the invention may be used to treat these diseases and conditions. Those skilled in the art are readily able to determine whether a given disease can be treated with a peptide according to the present invention using methods known in the art, including the assays of WO 2004092405, which is herein incorporated by reference, and assays which monitor hepcidin, hemojuvelin, or iron levels and expression, which are known in the art such as those described in U.S. Pat. No. 7,534,764, which is herein incorporated by reference.
[0098] In certain embodiments of the present invention, the diseases of iron metabolism are iron overload diseases, which include hereditary hemochromatosis, iron-loading anemias, alcoholic liver diseases and chronic hepatitis C.
[0099] The term "pharmaceutically acceptable salt," as used herein, represents salts or zwitterionic forms of the peptides or compounds of the present invention which are water or oil-soluble or dispersible, which are suitable for treatment of diseases without undue toxicity, irritation, and allergic response; which are commensurate with a reasonable benefit/risk ratio, and which are effective for their intended use. The salts can be prepared during the final isolation and purification of the compounds or separately by reacting an amino group with a suitable acid. Representative acid addition salts include acetate, adipate, alginate, citrate, aspartate, benzoate, benzenesulfonate, bisulfate, butyrate, camphorate, camphorsulfonate, digluconate, glycerophosphate, hemisulfate, heptanoate, hexanoate, formate, fumarate, hydrochloride, hydrobromide, hydroiodide, 2-hydroxyethansulfonate (isethionate), lactate, maleate, mesitylenesulfonate, methanesulfonate, naphthylenesulfonate, nicotinate, 2-naphthalenesulfonate, oxalate, pamoate, pectinate, persulfate, 3-phenylproprionate, picrate, pivalate, propionate, succinate, tartrate, trichloroacetate, trifluoroacetate, phosphate, glutamate, bicarbonate, para-toluenesulfonate, and undecanoate. Also, amino groups in the compounds of the present invention can be quaternized with methyl, ethyl, propyl, and butyl chlorides, bromides, and iodides; dimethyl, diethyl, dibutyl, and diamyl sulfates; decyl, lauryl, myristyl, and steryl chlorides, bromides, and iodides; and benzyl and phenethyl bromides. Examples of acids which can be employed to form therapeutically acceptable addition salts include inorganic acids such as hydrochloric, hydrobromic, sulfuric, and phosphoric, and organic acids such as oxalic, maleic, succinic, and citric. A pharmaceutically acceptable salt may suitably be a salt chosen, e.g., among acid addition salts and basic salts. Examples of acid addition salts include chloride salts, citrate salts and acetate salts. Examples of basic salts include salts where the cation is selected among alkali metal cations, such as sodium or potassium ions, alkaline earth metal cations, such as calcium or magnesium ions, as well as substituted ammonium ions, such as ions of the type N(R1)(R2)(R3)(R4)+, where R1, R2, R3 and R4 independently will typically designate hydrogen, optionally substituted C1-6-alkyl or optionally substituted C2-6-alkenyl. Examples of relevant C1-6-alkyl groups include methyl, ethyl, 1-propyl and 2-propyl groups. Examples of C2-6-alkenyl groups of possible relevance include ethenyl, 1-propenyl and 2-propenyl. Other examples of pharmaceutically acceptable salts are described in "Remington's Pharmaceutical Sciences", 17th edition, Alfonso R. Gennaro (Ed.), Mark Publishing Company, Easton, Pa., USA, 1985 (and more recent editions thereof), in the "Encyclopaedia of Pharmaceutical Technology", 3rd edition, James Swarbrick (Ed.), Informa Healthcare USA (Inc.), NY, USA, 2007, and in J. Pharm. Sci. 66: 2 (1977). Also, for a review on suitable salts, see Handbook of Pharmaceutical Salts: Properties, Selection, and Use by Stahl and Wermuth (Wiley-VCH, 2002). Other suitable base salts are formed from bases which form non-toxic salts. Representative examples include the aluminum, arginine, benzathine, calcium, choline, diethylamine, diolamine, glycine, lysine, magnesium, meglumine, olamine, potassium, sodium, tromethamine, and zinc salts. Hemisalts of acids and bases may also be formed, e.g., hemisulphate and hemicalcium salts.
[0100] The term "N(alpha)Methylation", as used herein, describes the methylation of the alpha amine of an amino acid, also generally termed as an N-methylation.
[0101] The term "sym methylation" or "Arg-Me-sym", as used herein, describes the symmetrical methylation of the two nitrogens of the guanidine group of arginine. Further, the term "asym methylation" or "Arg-Me-asym" describes the methylation of a single nitrogen of the guanidine group of arginine.
[0102] The term "acylating organic compounds", as used herein refers to various compounds with carboxylic acid functionality that are used to acylate the N-terminus of an amino acid subunit prior to forming a C-terminal dimer. Non-limiting examples of acylating organic compounds include cyclopropylacetic acid, 4-Fluorobenzoic acid, 4-fluorophenylacetic acid, 3-Phenylpropionic acid, Succinic acid, Glutaric acid, Cyclopentane carboxylic acid, 3,3,3-trifluoropropeonic acid, 3-Fluoromethylbutyric acid, Tetrahedro-2H-Pyran-4-carboxylic acid.
[0103] The term "alkyl" includes a straight chain or branched, noncyclic or cyclic, saturated aliphatic hydrocarbon containing from 1 to 24 carbon atoms. Representative saturated straight chain alkyls include, but are not limited to, methyl, ethyl, n-propyl, n-butyl, n-pentyl, n-hexyl, and the like, while saturated branched alkyls include, without limitation, isopropyl, sec-butyl, isobutyl, tert-butyl, isopentyl, and the like. Representative saturated cyclic alkyls include, but are not limited to, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, and the like, while unsaturated cyclic alkyls include, without limitation, cyclopentenyl, cyclohexenyl, and the like.
[0104] As used herein, a "therapeutically effective amount" of the peptide agonists of the invention is meant to describe a sufficient amount of the peptide agonist to treat an hepcidin-related disease, including but not limited to any of the diseases and disorders described herein (for example, a disease of iron metabolism). In particular embodiments, the therapeutically effective amount will achieve a desired benefit/risk ratio applicable to any medical treatment.
[0105] Peptide Analogues of Hepcidin
[0106] The present invention provides peptide analogues of hepcidin, which may be monomers or dimers (collectively "hepcidin analogues").
[0107] In some embodiments, a hepcidin analogue of the present invention binds ferroportin, e.g., human ferroportin. In certain embodiments, hepcidin analogues of the present invention specifically bind human ferroportin. As used herein, "specifically binds" refers to a specific binding agent's preferential interaction with a given ligand over other agents in a sample. For example, a specific binding agent that specifically binds a given ligand, binds the given ligand, under suitable conditions, in an amount or a degree that is observable over that of any nonspecific interaction with other components in the sample. Suitable conditions are those that allow interaction between a given specific binding agent and a given ligand. These conditions include pH, temperature, concentration, solvent, time of incubation, and the like, and may differ among given specific binding agent and ligand pairs, but may be readily determined by those skilled in the art. In some embodiments, a hepcidin analogue of the present invention binds ferroportin with greater specificity than a hepcidin reference compound (e.g., any one of the hepcidin reference compounds provided herein). In some embodiments, a hepcidin analogue of the present invention exhibits ferroportin specificity that is at least about 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 100%, 200%, 300%, 400%, 500%, 700%, 1000%, or 10,000% higher than a hepcidin reference compound (e.g., any one of the hepcidin reference compounds provided herein. In some embodiments, a hepcidin analogue of the present invention exhibits ferroportin specificity that is at least about 5 fold, or at least about 10, 20, 50, or 100 fold higher than a hepcidin reference compound (e.g., any one of the hepcidin reference compounds provided herein.
[0108] In certain embodiments, a hepcidin analogue of the present invention exhibits a hepcidin activity. In some embodiments, the activity is an in vitro or an in vivo activity, e.g., an in vivo or an in vitro activity described herein. In some embodiments, a hepcidin analogue of the present invention exhibits at least about 50%, 60%, 70%, 80%, 90%, 95%, 97%, 98%, 99%, or greater than 99% of the activity exhibited by a hepcidin reference compound (e.g., any one of the hepcidin reference compounds provided herein.
[0109] In some embodiments, a hepcidin analogue of the present invention exhibits at least about 50%, 60%, 70%, 80%, 90%, 95%, 97%, 98%, 99%, or greater than 99% of the ferroportin binding ability that is exhibited by a reference hepcidin. In some embodiments, a hepcidin analogue of the present invention has a lower IC.sub.50 (i.e., higher binding affinity) for binding to ferroportin, (e.g., human ferroportin) compared to a reference hepcidin. In some embodiments, a hepcidin analogue the present invention has an IC.sub.50 in a ferroportin competitive binding assay which is at least about 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 100%, 200%, 300%, 400%, 500%, 700%, or 1000% lower than a reference hepcidin.
[0110] In certain embodiments, a hepcidin analogue of the present invention exhibits increased hepcidin activity as compared to a hepcidin reference peptide. In some embodiments, the activity is an in vitro or an in vivo activity, e.g., an in vivo or an in vitro activity described herein. In certain embodiments, the hepcidin analogue of the present invention exhibits 1.5, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 30, 40, 50, 60, 70, 80, 90, 100, 120, 140, 160, 180, or 200-fold greater hepcidin activity than a reference hepcidin. In certain embodiments, the hepcidin analogue of the present invention exhibits at least about 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, 97%, 98%, 99% or greater than 99%, 100%, 200% 300%, 400%, 500%, 700%, or 1000% greater activity than a reference hepcidin.
[0111] In some embodiments, a peptide analogue of the present invention exhibits at least about 50%, 60%, 70%, 80%, 90%, 95%, 97%, 98%, 99%, or greater than 99%, 100%, 200% 300%, 400%, 500%, 700%, or 1000% greater in vitro activity for inducing the degradation of human ferroportin protein as that of a reference hepcidin, wherein the activity is measured according to a method described herein.
[0112] In some embodiments, a peptide or a peptide dimer of the present invention exhibits at least about 50%, 60%, 70%, 80%, 90%, 95%, 97%, 98%, 99%, or greater than 99%, 100%, 200% 300%, 400%, 500%, 700%, or 1000% greater in vivo activity for inducing the reduction of free plasma iron in an individual as does a reference hepcidin, wherein the activity is measured according to a method described herein.
[0113] In some embodiments, the activity is an in vitro or an in vivo activity, e.g., an in vivo or an in vitro activity described herein. In certain embodiments, a hepcidin analogue of the present invention exhibits 1.5, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 30, 40, 50, 60, 70, 80, 90, 100, 120, 140, 160, 180, or 200-fold greater or at least about 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 100%, 200%, 300%, 400%, 500%, 700%, or 1000% greater activity than a reference hepcidin, wherein the activity is an in vitro activity for inducing the degradation of ferroportin, e.g., as measured according to the Examples herein; or wherein the activity is an in vivo activity for reducing free plasma iron, e.g., as measured according to the Examples herein.
[0114] In some embodiments, the hepcidin analogues of the present invention mimic the hepcidin activity of Hep25, the bioactive human 25-amino acid form, are herein referred to as "mini-hepcidins". As used herein, in certain embodiments, a compound (e.g., a hepcidin analogue) having a "hepcidin activity" means that the compound has the ability to lower plasma iron concentrations in subjects (e.g. mice or humans), when administered thereto (e.g. parenterally injected or orally administered), in a dose-dependent and time-dependent manner. See e.g. as demonstrated in Rivera et al. (2005), Blood 106:2196-9. In some embodiments, the peptides of the present invention lower the plasma iron concentration in a subject by at least about 1.2, 1.5, 2, 3, 4, 5, 6, 7, 8, 9, or 10-fold, or at least about 5%, 10%, 20%, 25%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or about 99%.
[0115] In some embodiments, the hepcidin analogues of the present invention have in vitro activity as assayed by the ability to cause the internalization and degradation of ferroportin in a ferroportin-expressing cell line as taught in Nemeth et al. (2006) Blood 107:328-33. In some embodiments, in vitro activity is measured by the dose-dependent loss of fluorescence of cells engineered to display ferroportin fused to green fluorescent protein as in Nemeth et al. (2006) Blood 107:328-33. Aliquots of cells are incubated for 24 hours with graded concentrations of a reference preparation of Hep25 or a mini-hepcidin. As provided herein, the EC.sub.50 values are provided as the concentration of a given compound (e.g. a hepcidin analogue peptide or peptide dimer of the present invention) that elicits 50% of the maximal loss of fluorescence generated by a reference compound. The EC.sub.50 of the Hep25 preparations in this assay range from 5 to 15 nM and in certain embodiments, preferred hepcidin analogues of the present invention have EC.sub.50 values in in vitro activity assays of about 1,000 nM or less. In certain embodiments, a hepcidin analogue of the present invention has an EC.sub.50 in an in vitro activity assay (e.g., as described in Nemeth et al. (2006) Blood 107:328-33 or the Example herein) of less than about any one of 0.01, 0.05, 0.1, 0.2, 0.3, 0.4, 0.5, 0.6, 0.7, 0.8, 0.9, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 25, 30, 40, 50, 60, 70, 80, 90, 100, 200 or 500 nM. In some embodiments, a hepcidin analogue or biotherapeutic composition (e.g., any one of the pharmaceutical compositions described herein) has an EC.sub.50 value of about 1 nM or less.
[0116] Other methods known in the art for calculating the hepcidin activity and in vitro activity of the hepcidin analogues according to the present invention may be used. For example, in certain embodiments, the in vitro activity of the hepcidin analogues or the reference peptides is measured by their ability to internalize cellular ferroportin, which is determined by immunohistochemistry or flow cytometry using antibodies which recognizes extracellular epitopes of ferroportin. Alternatively, in certain embodiments, the in vitro activity of the hepcidin analogues or the reference peptides is measured by their dose-dependent ability to inhibit the efflux of iron from ferroportin-expressing cells that are preloaded with radioisotopes or stable isotopes of iron, as in Nemeth et al. (2006) Blood 107:328-33.
[0117] In some embodiments, the hepcidin analogues of the present invention exhibit increased stability (e.g., as measured by half-life, rate of protein degradation) as compared to a reference hepcidin. In certain embodiments, the stability of a hepcidin analogue of the present invention is increased at least about 1.5, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 30, 40, 50, 60, 70, 80, 90, 100, 120, 140, 160, 180, or 200-fold greater or at least about 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 100%, 200%, 300%, 400%, or 500% greater than a reference hepcidin. In some embodiments, the stability is a stability that is described herein. In some embodiments, the stability is a plasma stability, e.g., as optionally measured according to the method described herein. In some embodiments, the stability is stability when delivered orally.
[0118] In particular embodiments, a hepcidin analogue of the present invention exhibits a longer half-life than a reference hepcidin. In particular embodiments, a hepcidin analogue of the present invention has a half-life under a given set of conditions (e.g., temperature, pH) of at least about 5 minutes, at least about 10 minutes, at least about 20 minutes, at least about 30 minutes, at least about 45 minutes, at least about 1 hour, at least about 2 hour, at least about 3 hours, at least about 4 hours, at least about 5 hours, at least about 6 hours, at least about 12 hours, at least about 18 hours, at least about 1 day, at least about 2 days, at least about 4 days, at least about 7 days, at least about 10 days, at least about two weeks, at least about three weeks, at least about 1 month, at least about 2 months, at least about 3 months, or more, or any intervening half-life or range in between, about 5 minutes, about 10 minutes, about 20 minutes, about 30 minutes, about 45 minutes, about 1 hour, about 2 hour, about 3 hours, about 4 hours, about 5 hours, about 6 hours, about 12 hours, about 18 hours, about 1 day, about 2 days, about 4 days, about 7 days, about 10 days, about two weeks, about three weeks, about 1 month, about 2 months, about 3 months, or more, or any intervening half-life or range in between. In some embodiments, the half-life of a hepcidin analogue of the present invention is extended due to its conjugation to one or more lipophilic substituent or half-life extension moiety, e.g., any of the lipophilic substituents or half-life extension moieties disclosed herein. In some embodiments, the half-life of a hepcidin analogue of the present invention is extended due to its conjugation to one or more polymeric moieties, e.g., any of the polymeric moieties or half-life extension moieties disclosed herein. In certain embodiments, a hepcidin analogue of the present invention has a half-life as described above under the given set of conditions wherein the temperature is about 25.degree. C., about 4.degree. C., or about 37.degree. C., and the pH is a physiological pH, or a pH about 7.4.
[0119] In certain embodiments, a hepcidin analogue of the present invention, comprising a conjugated half-life extension moiety, has an increased serum half-life following oral, intravenous or subcutaneous administration as compared to the same analogue but lacking the conjugated half-life extension moiety. In particular embodiments, the serum half-life of a hepcidin analogue of the present invention following any of oral, intravenous or subcutaneous administration is at least 12 hours, at least 24 hours, at least 30 hours, at least 36 hours, at least 48 hours, at least 72 hours or at least 168 h. In particular embodiments, it is between 12 and 168 hours, between 24 and 168 hours, between 36 and 168 hours, or between 48 and 168 hours.
[0120] In certain embodiments, a hepcidin analogue of the present invention, comprising a conjugated half-life extension moiety, results in decreased concentration of serum iron following oral, intravenous or subcutaneous administration to a subject. In particular embodiments, the subject's serum iron concentration is decreased to less than 10%, less than 20%, less than 25%, less than 30%, less than 40%, less than 50%, less than 60%, less than 70%, less than 80%, or less than 90% of the serum iron concentration in the absence of administration of the hepcidin analogue to the subject. In particular embodiments, the decreased serum iron concentration remains for a least 1 hour, at least 4 hours, at least 10 hours, at least 12 hours, at least 24 hours, at least 36 hours, at least 48 hours, or at least 72 hours following administration to the subject. In particular embodiments, it remains for between 12 and 168 hours, between 24 and 168 hours, between 36 and 168 hours, or between 48 and 168 hours. In one embodiment, the serum iron concentration of the subject is reduced to less than 20% at about 4 hours or about 10 hours following administration to the subject, e.g., intravenously, orally, or subcutaneously. In one embodiment, the serum iron concentration of the subject is reduced to less than 50% or less than 60% for about 24 to about 30 hours following administration, e.g., intravenously, orally, or subcutaneously.
[0121] In some embodiments, the half-life is measured in vitro using any suitable method known in the art, e.g., in some embodiments, the stability of a hepcidin analogue of the present invention is determined by incubating the hepcidin analogue with pre-warmed human serum (Sigma) at 37.degree. C. Samples are taken at various time points, typically up to 24 hours, and the stability of the sample is analyzed by separating the hepcidin analogue from the serum proteins and then analyzing for the presence of the hepcidin analogue of interest using LC-MS.
[0122] In some embodiments, the stability of the hepcidin analogue is measured in vivo using any suitable method known in the art, e.g., in some embodiments, the stability of a hepcidin analogue is determined in vivo by administering the peptide or peptide dimer to a subject such as a human or any mammal (e.g., mouse) and then samples are taken from the subject via blood draw at various time points, typically up to 24 hours. Samples are then analyzed as described above in regard to the in vitro method of measuring half-life. In some embodiments, in vivo stability of a hepcidin analogue of the present invention is determined via the method disclosed in the Examples herein.
[0123] In some embodiments, the present invention provides a hepcidin analogue as described herein, wherein the hepcidin analogue exhibits improved solubility or improved aggregation characteristics as compared to a reference hepcidin. Solubility may be determined via any suitable method known in the art. In some embodiments, suitable methods known in the art for determining solubility include incubating peptides (e.g., a hepcidin analogue of the present invention) in various buffers (Acetate pH4.0, Acetate pH5.0, Phos/Citrate pH5.0, Phos Citrate pH6.0, Phos pH 6.0, Phos pH 7.0, Phos pH7.5, Strong PBS pH 7.5, Tris pH7.5, Tris pH 8.0, Glycine pH 9.0, Water, Acetic acid (pH 5.0 and other known in the art) and testing for aggregation or solubility using standard techniques. These include, but are not limited to, visual precipitation, dynamic light scattering, Circular Dichroism and fluorescent dyes to measure surface hydrophobicity, and detect aggregation or fibrillation, for example. In some embodiments, improved solubility means the peptide (e.g., the hepcidin analogue of the present invention) is more soluble in a given liquid than is a reference hepcidin.
[0124] In certain embodiments, the present invention provides a hepcidin analogue as described herein, wherein the hepcidin analogue exhibits a solubility that is increased at least about 1.5, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 30, 40, 50, 60, 70, 80, 90, 100, 120, 140, 160, 180, or 200-fold greater or at least about 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 100%, 200%, 300%, 400%, or 500% greater than a reference hepcidin in a particular solution or buffer, e.g., in water or in a buffer known in the art or disclosed herein.
[0125] In certain embodiments, the present invention provides a hepcidin analogue as described herein, wherein the hepcidin analogue exhibits decreased aggregation, wherein the aggregation of the peptide in a solution is at least about 1.5, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 30, 40, 50, 60, 70, 80, 90, 100, 120, 140, 160, 180, or 200-fold less or at least about 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 100%, 200%, 300%, 400%, or 500% less than a reference hepcidin in a particular solution or buffer, e.g., in water or in a buffer known in the art or disclosed herein.
[0126] In some embodiments, the present invention provides a hepcidin analogue, as described herein, wherein the hepcidin analogue exhibits less degradation (i.e., more degradation stability), e.g., greater than or about 10% less, greater than or about 20% less, greater than or about 30% less, greater than or about 40 less, or greater than or about 50% less than a reference hepcidin. In some embodiments, degradation stability is determined via any suitable method known in the art. In some embodiments, suitable methods known in the art for determining degradation stability include the method described in Hawe et al J Pharm Sci, VOL. 101, NO. 3, 2012, p 895-913, incorporated herein in its entirety. Such methods are in some embodiments used to select potent sequences with enhanced shelf lives.
[0127] In some embodiments, the hepcidin analogue of the present invention is synthetically manufactured. In other embodiments, the hepcidin analogue of the present invention is recombinantly manufactured.
[0128] The various hepcidin analogue monomer and dimer peptides of the invention may be constructed solely of natural amino acids. Alternatively, these hepcidin analogues may include unnatural or non-natural amino acids including, but not limited to, modified amino acids. In certain embodiments, modified amino acids include natural amino acids that have been chemically modified to include a group, groups, or chemical moiety not naturally present on the amino acid. The hepcidin analogues of the invention may additionally include D-amino acids. Still further, the hepcidin analogue peptide monomers and dimers of the invention may include amino acid analogs. In particular embodiments, a peptide analogue of the present invention comprises any of those described herein, wherein one or more natural amino acid residues of the peptide analogue is substituted with an unnatural or non-natural amino acid, or a D-amino acid.
[0129] In certain embodiments, the hepcidin analogues of the present invention include one or more modified or unnatural amino acids. For example, in certain embodiments, a hepcidin analogue includes one or more of Daba, Dapa, Pen, Sar, Cit, Cav, HLeu, 2-Nal, 1-Nal, d-1-Nal, d-2-Nal, Bip, Phe(4-OMe), Tyr(4-OMe), .beta.hTrp, .beta.hPhe, Phe(4-CF.sub.3), 2-2-Indane, 1-1-Indane, Cyclobutyl, .beta.hPhe, hLeu, Gla, Phe(4-NH.sub.2), hPhe, 1-Nal, Nle, 3-3-diPhe, cyclobutyl-Ala, Cha, Bip, .beta.-Glu, Phe(4-Guan), homo amino acids, D-amino acids, and various N-methylated amino acids. One having skill in the art will appreciate that other modified or unnatural amino acids, and various other substitutions of natural amino acids with modified or unnatural amino acids, may be made to achieve similar desired results, and that such substitutions are within the teaching and spirit of the present invention.
[0130] The present invention includes any of the hepcidin analogues described herein, e.g., in a free or a salt form.
[0131] The hepcidin analogues of the present invention include any of the peptide monomers or dimers described herein linked to a linker moiety, including any of the specific linker moieties described herein.
[0132] The hepcidin analogues of the present invention include peptides, e.g., monomers or dimers, comprising a peptide monomer subunit having at least 85%, at least 90%, at least 92%, at least 94%, at least 95%, at least 98%, or at least 99% amino acid sequence identity to a hepcidin analogue peptide sequence described herein (e.g., any one of the peptides disclosed herein), including but not limited to any of the amino acid sequences shown in Tables 3 and 4.
[0133] In certain embodiments, a peptide analogue of the present invention, or a monomer subunit of a dimer peptide analogue of the present invention, comprises or consists of 7 to 35 amino acid residues, 8 to 35 amino acid residues, 9 to 35 amino acid residues, 10 to 35 amino acid residues, 7 to 25 amino acid residues, 8 to 25 amino acid residues, 9 to 25 amino acid residues, 10 to 25 amino acid residues, 7 to 18 amino acid residues, 8 to 18 amino acid residues, 9 to 18 amino acid residues, or 10 to 18 amino acid residues, and, optionally, one or more additional non-amino acid moieties, such as a conjugated chemical moiety, e.g., a half-life extension moiety, a PEG or linker moiety. In particular embodiments, a monomer subunit of a hepcidin analogue comprises or consists of 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, or 35 amino acid residues. In particular embodiments, a monomer subunit of a hepcidin analogue of the present invention comprises or consists of 10 to 18 amino acid residues and, optionally, one or more additional non-amino acid moieties, such as a conjugated chemical moiety, e.g., a PEG or linker moiety. In various embodiments, the monomer subunit comprises or consists of 7 to 35 amino acid residues, 9 to 18 amino acid residues, or 10 to 18 amino acid residues. In particular embodiments of any of the various Formulas described herein, X comprises or consists of 7 to 35 amino acid residues, 8 to 35 amino acid residues, 9 to 35 amino acid residues, 10 to 35 amino acid residues, 7 to 25 amino acid residues, 8 to 25 amino acid residues, 9 to 25 amino acid residues, 10 to 25 amino acid residues, 7 to 18 amino acid residues, 8 to 18 amino acid residues, 9 to 18 amino acid residues, or 10 to 18 amino acid residues.
[0134] In particular embodiments, a hepcidin analogue or dimer of the present invention does not include any of the compounds described in PCT/US2014/030352 or PCT/US2015/038370.
[0135] Peptide Monomer Hepcidin Analogues
[0136] In certain embodiments, hepcidin analogues of the present invention comprise a single peptide subunit, optionally conjugated to a half-life extension moiety. In certain embodiments, these hepcidin analogues form cyclized structures through intramolecular disulfide or other bonds.
[0137] In one embodiment, the present invention includes a hepcidin analogue comprising a polypeptide sequence of Formula (I'):
X--Y (I')
or a pharmaceutically acceptable salt or solvate thereof, wherein: X is a peptide comprising the sequence Xa':
TABLE-US-00008 (Xa) (SEQ ID NO: 1) X1-Thr-His-X4-Pro-X6-X7-X8-Phe-X10
[0138] wherein [0139] X1 is Asp, isoGlu or Ida; [0140] X4 is Phe, Phe(4-F), Phe(4-CN), 4-BIP, Phe(4-OCH.sub.3), Tyr, Phe(2,3-(OCH.sub.3).sub.2), Phe(2,3-Cl.sub.2), or Dpa; [0141] X6 is Cys or Pen; [0142] X7 is any amino acid; [0143] X8 is Ile, Leu, Val, nLeu, Lys or Arg; and [0144] X10 is Lys, Glu or absent; and Y is a peptide comprising the sequence Ya:
TABLE-US-00009 [0144] (Ya) (SEQ ID NO: 2) Y1-Y2-Y3-Y4-Y5-Y6-Y7
[0145] wherein [0146] Y1 is amino acid [0147] Y2 is any amino acid [0148] Y3 is any amino acid [0149] Y4 is any amino acid [0150] Y5 is any amino acid; [0151] Y6 is Cys or Pen; and [0152] Y7 is Lys or absent; and wherein the hepcidin analogue comprises a conjugated half-life extension moiety, wherein the half-life extension moiety is optionally conjugated via a linker moiety. The peptides Xa and Ya may be linked via a peptide bond to form the polypeptide of Formula (I').
[0153] In particular embodiments of any of the hepcidin analogues, the hepcidin analogue comprises one of more of the following: X1 is Asp; X4 is Phe or Dpa; X7 is Ile, Leu, Val, nLeu, or Lys; X7 is Ile or Lys; X8 is Lys or Arg; Y1 is Pro or hPro; Y1 is Pro; Y2 is Arg or Lys; Y2 is Arg; Y3 is Ser; Y4 is Lys, Arg or His; Y4 is Lys; or Y5 is Gly or Sar. In certain embodiments: X1 is Asp; X4 is Phe or Dpa; X7 is Ile, Leu, Val, nLeu, or Lys; X8 is Lys or Arg; Y1 is Pro or hPro; Y2 is Arg or Lys; Y3 is Ser; Y4 is Lys, Arg or His; and Y5 is Gly or Sar. In certain embodiments, X1 is Asp; X4 is Phe or Dpa; X7 is Ile or Lys; X8 is Lys or Arg; Y1 is Pro; Y2 is Arg; Y3 is Ser; and Y4 is Lys; and Y5 is Gly or Sar.
[0154] In certain embodiments, a hepcidin analogue comprises a polypeptide sequence of Formula (V'):
X--Y (V')
or a pharmaceutically acceptable salt or solvate thereof, wherein: X is a peptide sequence having the formula Xv:
TABLE-US-00010 (Xv) (SEQ ID NO: 3) Asp-Thr-His-X4-Pro-X6-X7-X8-Phe-X10
[0155] wherein [0156] X4 is Phe or Dpa; [0157] X6 is Cys or Pen; [0158] X7 is Ile or Lys; [0159] X8 is Lys or Arg; and [0160] X10 is Lys, Glu or absent; and Y is a peptide sequence having the formula Yv:
TABLE-US-00011 [0160] (Yv) (SEQ ID NO: 4) Pro-Arg-Ser-Lys-Y5-Y6-Y7
[0161] wherein [0162] Y5 is Gly or Sar; [0163] Y6 is Cys or Pen; and
[0164] Y7 is Lys or absent; and
wherein the hepcidin analogue comprises a conjugated half-life extension moiety, and wherein the half-life extension moiety is optionally conjugated via a linker moiety.
[0165] In particular embodiments, the hepcidin analogue comprises a structure of Formula II':
R.sup.1--X-L-Y--R.sup.2 (II')
or a pharmaceutically acceptable salt or solvate thereof, wherein: R.sup.1 is hydrogen, a C1-C6 alkyl, a C6-C12 aryl, a C6-C12 aryl C1-C6 alkyl, or a C1-C20 alkanoyl, and including PEGylated versions alone or as spacers of any of the foregoing; R.sup.2 is OH or NH.sub.2; X is a peptide sequence having the formula Xa or Xv as described herein; L is absent, a bond, or a linker moiety; and Y is a peptide having the formula Ya or Yv as described herein.
[0166] In particular embodiments of hepcidin analogues of Formulaes I', V' or II', the hepcidin analogue comprises one or more additional amino acid residues. For example either X or Y may further comprise one to three additional amino acids at the N-terminus or C-terminus.
[0167] In particular embodiments of hepcidin analogues of Formulaes I', V' or II', Y5 is Sar.
[0168] In various embodiments, the hepcidin analogues comprise a disulfide bond between X6 and Y6.
[0169] In particular embodiments of hepcidin analogues of Formula II', L is a bond, e.g., a peptide bond.
[0170] In particular embodiments, a half-life extension moiety is conjugated to a Lys at X8 or X10.
[0171] Particular embodiments of hepcidin analogues comprises one of the following illustrative structures:
##STR00002##
[0172] In certain embodiments of any of the peptide analogues having any of the various Formulae set forth herein, R.sup.1 is selected from methyl, acetyl, formyl, benzoyl, trifluoroacetyl, isovaleryl, isobutyryl, octanyl, and conjugated amides of lauric acid, hexadecanoic acid, and .gamma.-Glu-hexadecanoic acid.
[0173] In certain embodiments, the linker between the peptide and the half-life extension moiety is PEG11, Ahx, or any of the others described herein.
[0174] In certain embodiments, the half-life extension moiety is Palm.
[0175] In certain embodiment, the present invention includes a polypeptide comprising an amino acid sequence set forth in Table 3 (with or without the indicated linker moieties and half-life extension moieties), or having any amino acid sequence with at least 85%, at least 90%, at least 92%, at least 94%, or at least 95% identity to any of these amino acid sequences. In certain embodiment, the present invention provides a cyclized form of any one of the hepcidin analogues listed in Table 3, comprising a disulfide bond between the two Cys and/or Pen residues. The conjugated half-life extension moiety and the amino acid residue to which it is conjugated are indicated by parentheses and brackets, respectively.
TABLE-US-00012 TABLE 3 Illustrative Monomer Hepcidin Analogues HEK- GFP cell based Cmpd # Sequence assay 1 Isovaleric acid-DTHFPCI-[Lys(IsoGlu-Palm)]-FEPRSKGCK-NH.sub.2 (SEQ ID NO: 7) 5 (reference compound) 2 Isovaleric acid-DTHFPCIKF-[Lys(IsoGlu-Palm)]-PRSKGCK-NH.sub.2 (SEQ ID NO: 8) 12 (reference compound) 3 Isovaleric acid-DTHFP-[Pen]-IKF-[Lys(IsoGlu-Palm)]-PRSKG-[Pen]-K-NH.sub.2 (SEQ 418 ID NO: 9) 4 Isovaleric acid-DTHFP-[Pen]-IKF-[Lys(IsoGlu-Palm)]-PRSKGCK-NH.sub.2 (SEQ ID 68 NO: 10) 5 Isovaleric acid-DTHFPCIKF-[Lys(IsoGlu-Palm)]-PRSKG-[Pen]-K-NH.sub.2 (SEQ ID 13 NO: 11) 6 Isovaleric acid-DTHFPCIKF-[LysIsoGlu-Palm)]-PRSK-[Sar]-CK-NH.sub.2 (SEQ ID 11 NO: 12) 7 Isovaleric acid-DTH-[DPA]-PCIKF-[Lys(IsoGlu-Palm)]-PRSKGCK-NH.sub.2 SEQ ID 12 NO: 13) 8 Isovaleric acid-DTH-[DPA]-PCIKF-[Lys(IsoGlu-Palm)]-PRSK[Sar]-CK-NH.sub.2 (SEQ 15 ID NO: 14) 9 Isovaleric acid-DTHFPCIKF-[Lys(IsoGlu-Palm)]-PRSKGC-NH.sub.2 (SEQ ID NO: 15) 11 10 Isovaleric acid-DTHFPCIKF-[Lys(IsoGlu-Palm)]-PRSK-[Sar]-C-NH.sub.2 (SEQ ID 10 NO: 16) 11 Isovaleric acid-DTHFPCKKF-[Lys(IsoGlu-Palm)]-PRSKGCK-NH.sub.2 (SEQ ID NO: 17) 22 12 Isovaleric acid-DTHFPCIKF-[Lys(isoGlu-Lauric acid)]-PRSKGCK-NH.sub.2 (SEQ ID 14 NO: 18) 13 Isovaleric acid-DTHFPCIKF-Lys(isoGlu-Myristic acid)-PRSKGCK-NH.sub.2 (SEQ ID 10 NO: 19) 14 Isovaleric acid-DTHFPCIKF-[Lys(isoGlu-Bioitin)]-PRSKGCK-NH.sub.2 (SEQ ID NO: 20) 29 15 Isovaleric acid-DTHFPCIKF-[Lys(isoGlu-Isovaleric acid)]-PRSKGCK-NH.sub.2 (SEQ 26 ID NO: 21) 16 Isovaleric acid-DTHFPCIKF-[Lys(PEG2-Palm)]-PRSKGCK-NH.sub.2 (SEQ ID NO: 22) 36 17 Isovaleric acid-DTHFPCIKF-[Lys(PEG11-Palm)]-PRSKGCK-NH.sub.2 (SEQ ID NO: 23) 30 18 Isovaleric acid-DTHFPCIKF-[Lys(PEG11-Lauric acid)]-PRSKGCK-NH.sub.2 (SEQ ID 36 NO: 24) 19 Isovaleric acid-DTHFPCIKF-[Lys(PEG11-Myristic acid)]-PRSKGCK-NH.sub.2 (SEQ ID 30 NO: 25) 20 Isovaleric acid-DTHFPCIKF-[Lys(Palm)]-PRSKGCK-NH.sub.2 (SEQ ID NO: 26) 19 21 Isovaleric acid-DTHFPCIKF-[Lys(PEG8)]-PRSKGCK-NH.sub.2 (SEQ ID NO: 27) 32 22 Isovaleric acid-DTHFPCIKF-[Lys(Ahx-Palm)]-PRSKGCK-NH.sub.2 (SEQ ID NO: 28) 18 23 Isovaleric acid-DTHFPCI-[Lys(Ahx-Palm)]-FEPRSK-[Sar]-CK-NH.sub.2 (SEQ ID 6 NO: 29) 24 Isovaleric acid-DTHFPCIKF-[Lys(Ahx-Palm)]-PRSK-[Sar]-CK-NH.sub.2 (SEQ ID 10 NO: 30) 25 Isovaleric acid-DTHFPCIKF-[Lys(Ac)]-PRSKGCK-NH.sub.2 (SEQ ID NO: 31) 14 26 Isovaleric acid-DTHFPCI-[Lys(PEG11-Palm)]-FEPRSK-[Sar]-CK-NH.sub.2 (SEQ ID 16 NO: 32) 27 Isovaleric acid-DTHFPCIKF-[Lys(PEG11-Palm)]-PRSK-[Sar]-CK-NH.sub.2 (SEQ ID 13 NO: 33) 28 Isovaleric acid-DTHFPCI-[Lys(isoGlu-Ahx-Palm)]-FEPRSK-[Sar]-CK-NH.sub.2 (SEQ 23 ID NO: 34 29 Isovaleric acid-DTHFPCI-[Lys(OEG-OEG-isoGlu-(C18-diacid))]-FEPRSK-[Sar]- 98 CK-NH.sub.2 (SEQ ID NO: 35)
[0176] Peptide Dimer Hepcidin Analogues
[0177] In certain embodiments, the present invention includes dimer hepcidin analogues, which include dimers of any of the monomer hepcidin analogues described herein, including dimers comprising any of the monomer peptides sequences or structures set forth in the formulaes described herein, e.g., various embodiments of Formulaes I, I', V, V', II, and II', and certain dimers of sequences or structures set forth in Table 3. These dimers fall within the scope of the general term "hepcidin analogues" as used herein. The term "dimers," as in peptide dimers, refers to compounds in which two peptide monomer subunits are linked. A peptide dimer of the present invention may comprise two identical monomer subunits, resulting in a homodimer, or two non-identical monomer subunits, resulting in a heterodimer. A cysteine dimer comprises two peptide monomer subunits linked through a disulfide bond between a cysteine residue in one monomer subunit and a cysteine residue in the other monomer subunit.
[0178] In certain embodiments, a dimer hepcidin analogue comprises two polypeptide sequences of Formula (I''):
X--Y (I'')
or a pharmaceutically acceptable salt or solvate thereof, wherein: X is a peptide comprising the sequence Xa:
TABLE-US-00013 (Xa) (SEQ ID NO: 1) X1-Thr-His-X4-Pro-X6-X7-X8-Phe-X10
[0179] wherein [0180] X1 is Asp, isoGlu or Ida; [0181] X4 is Phe, Phe(4-F), Phe(4-CN), 4-BIP, Phe(4-OCH.sub.3), Tyr, Phe(2,3-(OCH.sub.3).sub.2), Phe(2,3-Cl.sub.2), or Dpa; [0182] X6 is Cys or Pen; [0183] X7 is any amino acid; [0184] X8 is Ile, Leu, Val, nLeu, Lys or Arg; and [0185] X10 is Lys, Glu or absent; and Y is absent, wherein the dimer hepcidin analogue comprises a conjugated half-life extension moiety, wherein the half-life extension moiety is optionally conjugated via a linker moiety.
[0186] In particular embodiments of any of the hepcidin analogues, the hepcidin analogue comprises one of more of the following: X1 is Asp; X4 is Phe or Dpa; X7 is Ile, Leu, Val, nLeu, or Lys; X7 is Ile or Lys; or X8 is Lys or Arg. In certain embodiments: X1 is Asp; X4 is Phe or Dpa; X7 is Ile, Leu, Val, nLeu, or Lys; and X8 is Lys or Arg. In certain embodiments, X1 is Asp; X4 is Phe or Dpa; X7 is Ile or Lys; and X8 is Lys or Arg.
[0187] In a related embodiments, the present invention includes a dimer hepcidin analogue comprising two polypeptide sequences of Formula (V''):
X--Y (V'')
or a pharmaceutically acceptable salt or solvate thereof, wherein: X is a peptide sequence having the formula Xv:
TABLE-US-00014 (Xv) (SEQ ID NO: 3) Asp-Thr-His-X4-Pro-X6-X7-X8-Phe-X10
[0188] wherein [0189] X4 is Phe or Dpa; [0190] X6 is Cys or Pen; [0191] X7 is Ile or Lys; [0192] X8 is Lys or Arg; and [0193] X10 is Lys, Glu or absent; and Y is absent, wherein the hepcidin analogue comprises a conjugated half-life extension moiety, wherein the half-life extension moiety is optionally conjugated via a linker moiety.
[0194] In particular embodiments, the dimer hepcidin analogue comprises a structure of Formula II'':
R.sup.1--X-L-Y--R.sup.2 (II'')
or a pharmaceutically acceptable salt or solvate thereof, wherein: R.sup.1 is hydrogen, a C1-C6 alkyl, a C6-C12 aryl, a C6-C12 aryl C1-C6 alkyl, or a C1-C20 alkanoyl, and including PEGylated versions alone or as spacers of any of the foregoing; R.sup.2 is OH or NH.sub.2; X is a peptide sequence having the formula Xa or Xv as described herein; L is absent, a bond, or a linker moiety; and Y is absent.
[0195] In particular embodiments of dimer hepcidin analogues of Formulaes I'', V'' or II'', the hepcidin analogue comprises one or more additional amino acid residues. For example, X may further comprise one to three additional amino acids at the N-terminus or C-terminus.
[0196] In particular embodiments, a dimer hepcidin analogues comprises two polypeptide sequence of Formula I'' or Formula V'', or two structures of Formula II'', dimerized via a linker moiety. In particular embodiments, the linker moiety is bound to the C-terminus of each hepcidin analogue. In particular embodiments, the linker moiety is bound to the N-terminus of each hepcidin analogue. In particular embodiments, the linker moiety is bound to the N-terminus of one hepcidin analogue and the C-terminus of the other hepcidin analogue present in the dimer.
[0197] In certain embodiments, the half-life extension moiety is conjugated to the linker moiety.
[0198] In some embodiments, the hepcidin analogues of the present invention are active in a dimer conformation, in particular when free cysteine residues are present in the peptide. In certain embodiments, this occurs either as a synthesized dimer or, in particular, when a free cysteine monomer peptide is present and under oxidizing conditions, dimerizes. In some embodiments, the dimer is a homodimer. In other embodiments, the dimer is a heterodimer.
[0199] In certain embodiments, a hepcidin analogue dimer of the present invention is a peptide dimer comprising two hepcidin analogue peptide monomers of the invention.
[0200] In certain embodiment, the present invention includes a polypeptide comprising an amino acid sequence set forth in Table 4 (with or without the indicated linker moieties and half-life extension moieties), or having any amino acid sequence with at least 85%, at least 90%, at least 92%, at least 94%, or at least 95% identity to any of these amino acid sequences. In a related embodiments, the present invention includes a dimer comprising two polypeptides, each comprising an amino acid sequence set forth in Table 4 (with or without the indicated linker moieties and half-life extension moieties), or having any amino acid sequence with at least 85%, at least 90%, at least 92%, at least 94%, or at least 95% identity to any of these amino acid sequences. In particular embodiments, a peptide dimer hepcidin analogue comprises one or more, e.g., two, peptide monomer subunits shown in Table 4. The conjugated half-life extension moiety and the amino acid residue to which it is conjugated are indicated by parentheses and brackets, respectively. Table 4 shows dimer hepcidin analogues, each comprising a dimer of the sequences in parentheses followed by a lower case "2", which are linked by the indicated one or more linkers, e.g., Lys or IDA, and conjugated to the indicated half-life extension moiety, e.g., octanoic acid or Palm.
TABLE-US-00015 TABLE 4 Illustrative Dimer Hepcidin Analogues HEK-GFP cell based Cmpd # Sequence assay 30 (Isovaleric acid-DTHFPCIRF).sub.2-[Lys(IsoGlu-Octanoic acid)]-NH.sub.2 18 (SEQ ID NO: 36) 31 (Isovaleric acid-DTHFPCIKF).sub.2-IDA-[.beta.ala]-(Peg2-Palm) 8 (SEQ ID NO: 37) 32 (Isovaleric acid-DTHFPCIKF).sub.2-IDA-[.beta.ala(Peg11-Palm)] 13 (SEQ ID NO: 38)
[0201] In particular embodiments, the monomer subunits may be dimerized by a disulfide bridge between two cysteine residues, one in each peptide monomer subunit, or they may be dimerized by another suitable linker moiety, including those described herein. Some of the monomer subunits are shown having C- and/or N-termini that both comprise free amine. Thus, to produce a peptide dimer inhibitor, the monomer subunit may be modified to eliminate either the C- or N-terminal free amine, thereby permitting dimerization at the remaining free amine. Further, in some instances, a terminal end of one or more monomer subunits is acylated with an acylating organic compound selected from the group consisting of 2-me-Trifluorobutyl, Trifluoropentyl, Acetyl, Octonyl, Butyl, Pentyl, Hexyl, Palmityl, Trifluoromethyl butyric, cyclopentane carboxylic, cyclopropylacetic, 4-fluorobenzoic, 4-fluorophenyl acetic, 3-Phenylpropionic, tetrahedro-2H-pyran-4carboxylic, succinic acid, and glutaric acid. In some instances, monomer subunits comprise both a free carboxy terminal and a free amino terminal, whereby a user may selectively modify the subunit to achieve dimerization at a desired terminus. One having skill in the art will, therefore, appreciate that the monomer subunits of the instant invention may be selectively modified to achieve a single, specific amine for a desired dimerization.
[0202] It is further understood that the C-terminal residues of the monomer subunits disclosed herein may be amides, unless otherwise indicated. Further, it is understood that, in certain embodiments, dimerization at the C-terminus is facilitated by using a suitable amino acid with a side chain having amine functionality, as is generally understood in the art. Regarding the N-terminal residues, it is generally understood that dimerization may be achieved through the free amine of the terminal residue, or may be achieved by using a suitable amino acid side chain having a free amine, as is generally understood in the art.
[0203] Moreover, it is understood that the side chains of one or more internal residue comprised in the hepcidin analogue peptide monomers of the present invention can be utilized for the purpose of dimerization. In such embodiments, the side chain is in some embodiments a suitable natural amino acid (e.g., Lys), or alternatively it is an unnatural amino acid comprising a side chain suitable for conjugation, e.g., to a suitable linker moiety, as defined herein.
[0204] The linker moieties connecting monomer subunits may include any structure, length, and/or size that is compatible with the teachings herein. In at least one embodiment, a linker moiety is selected from the non-limiting group consisting of: cysteine, lysine, DIG, PEG4, PEG4-biotin, PEG13, PEG25, PEG1K, PEG2K, PEG3.4K, PEG4K, PEG5K, IDA, IDA-Palm, ADA, Boc-IDA, Glutaric acid, Isophthalic acid, 1,3-phenylenediacetic acid, 1,4-phenylenediacetic acid, 1,2-phenylenediacetic acid, Triazine, Boc-Triazine, IDA-biotin, PEG4-Biotin, AADA, suitable aliphatics, aromatics, heteroaromatics, and polyethylene glycol based linkers having a molecular weight from approximately 400 Da to approximately 40,000 Da. Non-limiting examples of suitable linker moieties are provided in Table 5. In particular embodiment, any of these linker moieties may alternatively link a half-life extension moiety to a hepcidin analogue.
TABLE-US-00016 TABLE 5 Illustrative Linker Moieties Abbreviation Description Structure DIG DIGlycolic acid ##STR00003## PEG4 Bifunctional PEG linker with 4 PolyEthylene Glycol units ##STR00004## PEG13 Bifunctional PEG linker with 13 PolyEthylene Glycol units ##STR00005## PEG25 Bifunctional PEG linker with 25 PolyEthylene Glycol units ##STR00006## PEG1K Bifunctional PEG linker with PolyEthylene Glycol Mol wt of 1000 Da PEG2K Bifunctional PEG linker with PolyEthylene Glycol Mol wt of 2000 Da PEG3.4K Bifunctional PEG linker with PolyEthylene Glycol Mol wt of 3400 Da PEG5K Bifunctional PEG linker with PolyEthylene Glycol Mol wt of 5000 Da DIG Diglycolic acid ##STR00007## .beta.-Ala-IDA .beta.-Ala-Iminodiacetic acid ##STR00008## Boc-.beta.-Ala- IDA Boc-.beta.-Ala-Iminodiacetic acid ##STR00009## Ac-.beta.-Ala- IDA Ac-.beta.-Ala-Iminodiacetic acid ##STR00010## Palm-.beta.-Ala- IDA- Palmityl-.beta.-Ala-Iminodiacetic acid ##STR00011## GTA Glutaric acid ##STR00012## PMA Pemilic acid ##STR00013## AZA Azelaic acid ##STR00014## DDA Dodecanedioic acid ##STR00015## IPA Isopthalic acid ##STR00016## 1,3-PDA 1,3-Phenylenediacetic acid ##STR00017## 1,4-PDA 1,4-Phenylenediacetic acid ##STR00018## 1,2-PDA 1,2-Phenylenediacetic acid ##STR00019## Triazine Amino propyl Triazine di-acid ##STR00020## Boc-Triazine Boc-Triazine di-acid ##STR00021## IDA Iminodiacetic acid ##STR00022## AIDA n-Acetyl imino acetic acid ##STR00023## Biotin-.beta.-ala- IDA- N-Biotin-.beta.-Ala-Iminodiacetic acid ##STR00024## Lys Lysine ##STR00025##
[0205] One having skill in the art will appreciate that the C- and N-terminal and internal linker moieties disclosed herein are non-limiting examples of suitable linker moieties, and that the present invention may include any suitable linker moiety.
[0206] In certain embodiments of any of the hepcidin analogue peptide dimers, the N-terminus of each peptide monomer subunit is connected by a linker moiety.
[0207] In certain embodiments of any of the hepcidin analogue peptide dimers, the C-terminus of each peptide monomer subunit is connected by a linker moiety.
[0208] In certain embodiments, the side chains of one or more internal amino acid residues (e.g., Lys residues) comprised in each peptide monomer subunit of a hepcidin analogue peptide dimer are connected by a linker moiety.
[0209] In certain embodiments of any of the hepcidin analogue peptide dimers, the C-terminus, the N terminus, or an internal amino acid (e.g., a lysine sidechain) of each peptide monomer subunit is connected by a linker moiety and at least two cysteine or Pen residues of the hepcidin analogue peptide dimers are linked by a disulfide bridge. In some embodiments, a peptide dimer has a general structure shown below. Non-limiting schematic examples of such hepcidin analogues are shown in the following illustration:
##STR00026## ##STR00027##
[0210] Peptide Analogue Conjugates
[0211] In certain embodiments, hepcidin analogues of the present invention, including both monomers and dimers, comprise one or more conjugated chemical substituents, such as lipophilic substituents and polymeric moieties, collectively referred to herein as half-life extension moieties. Without wishing to be bound by any particular theory, it is believed that the lipophilic substituent binds to albumin in the bloodstream, thereby shielding the hepcidin analogue from enzymatic degradation, and thus enhancing its half-life. In addition, it is believed that polymeric moieties enhance half-life and reduce clearance in the bloodstream, and in some cases enhance permeability through the epithelium and retention in the lamina propria. Moreover, it is also surmised that these substituents in some cases may enhance permeability through the epithelium and retention in the lamina propria. The skilled person will be well aware of suitable techniques for preparing the compounds employed in the context of the invention. For examples of non-limiting suitable chemistry, see, e.g., WO98/08871, WO00/55184, WO00/55119, Madsen et al (J. Med. Chem. 2007, 50, 6126-32), and Knudsen et al. 2000 (J. Med Chem. 43, 1664-1669).
[0212] In one embodiment, the side chains of one or more amino acid residues (e.g., Lys residues) in a hepcidin analogue of the invention is further conjugated (e.g., covalently attached) to a lipophilic substituent or other half-life extension moiety. The lipophilic substituent may be covalently bonded to an atom in the amino acid side chain, or alternatively may be conjugated to the amino acid side chain via one or more spacers or linker moieties. The spacer or linker moiety, when present, may provide spacing between the hepcidin analogue and the lipophilic substituent. In particular embodiments, the half-life extension moiety is conjugated to the hepcidin analogue via a linker moiety, which in certain embodiments is a linker moiety shown in Table 5 or Table 7, or depicted in any of the illustrative compounds shown in Tables 3 and 4.
[0213] In certain embodiments, the lipophilic substituent or half-life extension moiety comprises a hydrocarbon chain having from 4 to 30 C atoms, for example at least 8 or 12 C atoms, and preferably 24 C atoms or fewer, or 20 C atoms or fewer. The hydrocarbon chain may be linear or branched and may be saturated or unsaturated. In certain embodiments, the hydrocarbon chain is substituted with a moiety which forms part of the attachment to the amino acid side chain or the spacer, for example an acyl group, a sulfonyl group, an N atom, an O atom or an S atom. In some embodiments, the hydrocarbon chain is substituted with an acyl group, and accordingly the hydrocarbon chain may form part of an alkanoyl group, for example palmitoyl, caproyl, lauroyl, myristoyl or stearoyl.
[0214] A lipophilic substituent may be conjugated to any amino acid side chain in a hepcidin analogue of the invention. In certain embodiment, the amino acid side chain includes a carboxy, hydroxyl, thiol, amide or amine group, for forming an ester, a sulphonyl ester, a thioester, an amide or a sulphonamide with the spacer or lipophilic substituent. For example, the lipophilic substituent may be conjugated to Asn, Asp, Glu, Gln, His, Lys, Arg, Ser, Thr, Tyr, Trp, Cys or Dbu, Dpr or Orn. In certain embodiments, the lipophilic substituent is conjugated to Lys. An amino acid shown as Lys in any of the formula provided herein may be replaced by, e.g., Dbu, Dpr or Orn where a lipophilic substituent is added.
[0215] In further embodiments of the present invention, alternatively or additionally, the side-chains of one or more amino acid residues in a hepcidin analogue of the invention may be conjugated to a polymeric moiety or other half-life extension moiety, for example, in order to increase solubility and/or half-life in vivo (e.g., in plasma) and/or bioavailability. Such modifications are also known to reduce clearance (e.g. renal clearance) of therapeutic proteins and peptides.
[0216] As used herein, "Polyethylene glycol" or "PEG" is a polyether compound of general formula H--(O--CH2-CH2)n-OH. PEGs are also known as polyethylene oxides (PEOs) or polyoxyethylenes (POEs), depending on their molecular weight PEO, PEE, or POG, as used herein, refers to an oligomer or polymer of ethylene oxide. The three names are chemically synonymous, but PEG has tended to refer to oligomers and polymers with a molecular mass below 20,000 g/mol, PEO to polymers with a molecular mass above 20,000 g/mol, and POE to a polymer of any molecular mass. PEG and PEO are liquids or low-melting solids, depending on their molecular weights. Throughout this disclosure, the 3 names are used indistinguishably. PEGs are prepared by polymerization of ethylene oxide and are commercially available over a wide range of molecular weights from 300 g/mol to 10,000,000 g/mol. While PEG and PEO with different molecular weights find use in different applications, and have different physical properties (e.g. viscosity) due to chain length effects, their chemical properties are nearly identical. The polymeric moiety is preferably water-soluble (amphiphilic or hydrophilic), non-toxic, and pharmaceutically inert. Suitable polymeric moieties include polyethylene glycols (PEG), homo- or co-polymers of PEG, a monomethyl-substituted polymer of PEG (mPEG), or polyoxyethylene glycerol (POG). See, for example, Int. J. Hematology 68:1 (1998); Bioconjugate Chem. 6:150 (1995); and Crit. Rev. Therap. Drug Carrier Sys. 9:249 (1992). Also encompassed are PEGs that are prepared for purpose of half-life extension, for example, mono-activated, alkoxy-terminated polyalkylene oxides (POA's) such as mono-methoxy-terminated polyethyelene glycols (mPEG's); bis activated polyethylene oxides (glycols) or other PEG derivatives are also contemplated. Suitable polymers will vary substantially by weights ranging from about 200 to about 40,000 are usually selected for the purposes of the present invention. In certain embodiments, PEGs having molecular weights from 200 to 2,000 daltons or from 200 to 500 daltons are used. Different forms of PEG may also be used, depending on the initiator used for the polymerization process, e.g., a common initiator is a monofunctional methyl ether PEG, or methoxypoly(ethylene glycol), abbreviated mPEG. Other suitable initiators are known in the art and are suitable for use in the present invention.
[0217] Lower-molecular-weight PEGs are also available as pure oligomers, referred to as monodisperse, uniform, or discrete. These are used in certain embodiments of the present invention.
[0218] PEGs are also available with different geometries: branched PEGs have three to ten PEG chains emanating from a central core group; star PEGs have 10 to 100 PEG chains emanating from a central core group; and comb PEGs have multiple PEG chains normally grafted onto a polymer backbone. PEGs can also be linear. The numbers that are often included in the names of PEGs indicate their average molecular weights (e.g. a PEG with n=9 would have an average molecular weight of approximately 400 daltons, and would be labeled PEG 400.
[0219] As used herein, "PEGylation" is the act of coupling (e.g., covalently) a PEG structure to the hepcidin analogue of the invention, which is in certain embodiments referred to as a "PEGylated hepcidin analogue". In certain embodiments, the PEG of the PEGylated side chain is a PEG with a molecular weight from about 200 to about 40,000. In certain embodiments, the PEG portion of the conjugated half-life extension moiety is PEG3, PEG4, PEG5, PEG6, PEG7, PEG8, PEG9, PEG10, or PEG11. In particular embodiments, it is PEG11. In certain embodiments, the PEG of a PEGylated spacer is PEG3 or PEG8. In some embodiments, a spacer is PEGylated. In certain embodiments, the PEG of a PEGylated spacer is PEG3, PEG4, PEG5, PEG6, PEG7, PEG8, PEG9, PEG10, or PEG11. In certain embodiments, the PEG of a PEGylated spacer is PEG3 or PEG8.
[0220] In some embodiments, the present invention includes a hepcidin analogue peptide (or a dimer thereof) conjugated with a PEG that is attached covalently, e.g., through an amide, a thiol, via click chemistry, or via any other suitable means known in the art. In particular embodiments PEG is attached through an amide bond and, as such, certain PEG derivatives used will be appropriately functionalized. For example, in certain embodiments, PEG11, which is O-(2-aminoethyl)-O'-(2-carboxyethyl)-undecaethyleneglycol, has both an amine and carboxylic acid for attachment to a peptide of the present invention. In certain embodiments, PEG25 contains a diacid and 25 glycol moieties.
[0221] Other suitable polymeric moieties include poly-amino acids such as poly-lysine, poly-aspartic acid and poly-glutamic acid (see for example Gombotz, et al. (1995), Bioconjugate Chem., vol. 6: 332-351; Hudecz, et al. (1992), Bioconjugate Chem., vol. 3, 49-57 and Tsukada, et al. (1984), J. Natl. Cancer Inst., vol. 73: 721-729. The polymeric moiety may be straight-chain or branched. In some embodiments, it has a molecular weight of 500-40,000 Da, for example 500-10,000 Da, 1000-5000 Da, 10,000-20,000 Da, or 20,000-40,000 Da.
[0222] In some embodiments, a hepcidin analogue of the invention may comprise two or more such polymeric moieties, in which case the total molecular weight of all such moieties will generally fall within the ranges provided above.
[0223] In some embodiments, the polymeric moiety may be coupled (by covalent linkage) to an amino, carboxyl or thiol group of an amino acid side chain. Certain examples are the thiol group of Cys residues and the epsilon amino group of Lys residues, and the carboxyl groups of Asp and Glu residues may also be involved.
[0224] The skilled worker will be well aware of suitable techniques which can be used to perform the coupling reaction. For example, a PEG moiety bearing a methoxy group can be coupled to a Cys thiol group by a maleimido linkage using reagents commercially available from Nektar Therapeutics AL. See also WO 2008/101017, and the references cited above, for details of suitable chemistry. A maleimide-functionalised PEG may also be conjugated to the side-chain sulfhydryl group of a Cys residue.
[0225] As used herein, disulfide bond oxidation can occur within a single step or is a two-step process. As used herein, for a single oxidation step, the trityl protecting group is often employed during assembly, allowing deprotection during cleavage, followed by solution oxidation. When a second disulfide bond is required, one has the option of native or selective oxidation. For selective oxidation requiring orthogonal protecting groups, Acm and Trityl is used as the protecting groups for cysteine. Cleavage results in the removal of one protecting pair of cysteine allowing oxidation of this pair. The second oxidative deprotection step of the cysteine protected Acm group is then performed. For native oxidation, the trityl protecting group is used for all cysteines, allowing for natural folding of the peptide.
[0226] A skilled worker will be well aware of suitable techniques which can be used to perform the oxidation step.
[0227] In particular embodiments, a hepcidin analogue of the present invention comprises a half-life extension moiety, which may be selected from but is not limited to the following: Ahx-Palm, PEG2-Palm, PEG11-Palm, isoGlu-Palm, dapa-Palm, isoGlu-Lauric acid, isoGlu-Mysteric acid, and isoGlu-Isovaleric acid.
[0228] In particular embodiments, a hepcidin analogue comprises a half-life extension moiety having the structure shown below, wherein n=0 to 24 or n=14 to 24:
##STR00028##
[0229] In certain embodiments, a hepcidin analogue of the present invention comprises a conjugated half-life extension moiety shown in Table 6.
TABLE-US-00017 TABLE 6 Illustrative Half-Life Extension Moieties # Conjugates C1 ##STR00029## C12 (Lauric acid) C2 ##STR00030## C14 (Mysteric acid) C3 ##STR00031## C16 (Palm or Palmitic acid) C4 C5 C6 C7 C8 C9 C10 C11 ##STR00032## Biotin C12
[0230] In certain embodiments, a half-life extension moiety is conjugated directly to a hepcidin analogue, while in other embodiments, a half-life extension moiety is conjugated to a hepcidin analogue peptide via a linker moiety, e.g., any of those depicted in Tables 5 or 7.
TABLE-US-00018 TABLE 7 Illustrative Linker Moieties # Linker Moiety L1 ##STR00033## IsoGlu L2 ##STR00034## Dapa L3 ##STR00035## Ahx L4 Lipdic based linkers: ##STR00036## n = 1 to 24 L5 ##STR00037## PEG1 L6 ##STR00038## PEG2 L7 ##STR00039## PEG11 (40 atoms) L8 ##STR00040## n = 1 to 25 PEG based linkers L9 ##STR00041## OEG L10 ##STR00042## IsoGlu-Ahx L11 ##STR00043## IsoGlu-OEG-OEG L12 ##STR00044## IsoGlu-PEG5 L13 ##STR00045## IsoGlu-PEGn L14 ##STR00046## .beta.Ala-PEG2 L15 ##STR00047## .beta.Ala-PEG11 (40 atoms)
[0231] With reference to linker structures shown in Table 7, reference to n=1 to 24 or n=1 to 25, or the like, (e.g., in L4, L8 or L13) indicates that n may be any integer within the recited range. For example, for L4 shown in Table 7, n could be 1, 2, 3, etc., wherein when n=5, L4 has the structure shown in L3 (Ahx).
[0232] In particular embodiments, a hepcidin analogue of the present invention comprises any of the linker moieties shown in Table 7 and any of the half-life extension moieties shown in Table 6, including any of the following combinations shown in Table 8.
TABLE-US-00019 TABLE 8 Illustrative Combinations of Linkers and Half- Life Extension Moieties in Hepcidin Analogues Half-Life Linker Extension Moiety L1 C1 L2 C1 L3 C1 L4 C1 L5 C1 L6 C1 L7 C1 L8 C1 L9 C1 L10 C1 L11 C1 L12 C1 L13 C1 L14 C1 L15 C1 L1 C2 L2 C2 L3 C2 L4 C2 L5 C2 L6 C2 L7 C2 L8 C2 L9 C2 L10 C2 L11 C2 L12 C2 L13 C2 L14 C2 L15 C2 L1 C3 L2 C3 L3 C3 L4 C3 L5 C3 L6 C3 L7 C3 L8 C3 L9 C3 L10 C3 L11 C3 L12 C3 L13 C3 L14 C3 L15 C3 L1 C4 L2 C4 L3 C4 L4 C4 L5 C4 L6 C4 L7 C4 L8 C4 L9 C4 L10 C4 L11 C4 L12 C4 L13 C4 L14 C4 L15 C4 L1 C5 L2 C5 L3 C5 L4 C5 L5 C5 L6 C5 L7 C5 L8 C5 L9 C5 L10 C5 L11 C5 L12 C5 L13 C5 L14 C5 L15 C5 L1 C6 L2 C6 L3 C6 L4 C6 L5 C6 L6 C6 L7 C6 L8 C6 L9 C6 L10 C6 L11 C6 L12 C6 L13 C6 L14 C6 L15 C6 L1 C7 L2 C7 L3 C7 L4 C7 L5 C7 L6 C7 L7 C7 L8 C7 L9 C7 L10 C7 L11 C7 L12 C7 L13 C7 L14 C7 L15 C7 L1 C8 L2 C8 L3 C8 L4 C8 L5 C8 L6 C8 L7 C8 L8 C8 L9 C8 L10 C8 L11 C8 L12 C8 L13 C8 L14 C8 L15 C8 L1 C9 L2 C9 L3 C9 L4 C9 L5 C9 L6 C9 L7 C9 L8 C9 L9 C9 L10 C9 L11 C9 L12 C9 L13 C9 L14 C9 L15 C9 L1 C10 L2 C10 L3 C10 L4 C10 L5 C10 L6 C10 L7 C10 L8 C10 L9 C10 L10 C10 L11 C10 L12 C10 L13 C10 L14 C10 L15 C10 L1 C11 L2 C11 L3 C11 L4 C11 L5 C11 L6 C11 L7 C11 L8 C11 L9 C11 L10 C11 L11 C11 L12 C11 L13 C11 L14 C11 L15 C11 L1 C12 L2 C12 L3 C12 L4 C12 L5 C12 L6 C12 L7 C12 L8 C12 L9 C12 L10 C12 L11 C12 L12 C12 L13 C12 L14 C12 L15 C12
[0233] In certain embodiments, a hepcidin analogue comprises two or more linkers. In particular embodiments, the two or more linkers are concatamerized, i.e., bound to each other.
[0234] In related embodiments, the present invention includes polynucleotides that encode a polypeptide having a peptide sequence present in any of the hepcidin analogues described herein.
[0235] In addition, the present invention includes vectors, e.g., expression vectors, comprising a polynucleotide of the present invention.
[0236] Methods of Treatment
[0237] In some embodiments, the present invention provides methods for treating a subject afflicted with a disease or disorder associated with dysregulated hepcidin signaling, wherein the method comprises administering to the subject a hepcidin analogue of the present invention. In some embodiments, the hepcidin analogue that is administered to the subject is present in a composition (e.g., a pharmaceutical composition). In one embodiment, a method is provided for treating a subject afflicted with a disease or disorder characterized by increased activity or expression of ferroportin, wherein the method comprises administering to the individual a hepcidin analogue or composition of the present invention in an amount sufficient to (partially or fully) bind to and agonize ferroportin in the subject. In one embodiment, a method is provided for treating a subject afflicted with a disease or disorder characterized by dysregulated iron metabolism, wherein the method comprises administering to the subject a hepcidin analogue or composition of the present invention.
[0238] In some embodiments, methods of the present invention comprise providing a hepcidin analogue or a composition of the present invention to a subject in need thereof. In particular embodiments, the subject in need thereof has been diagnosed with or has been determined to be at risk of developing a disease or disorder characterized by dysregulated iron levels (e.g., diseases or disorders of iron metabolism; diseases or disorders related to iron overload; and diseases or disorders related to abnormal hepcidin activity or expression). In particular embodiments, the subject is a mammal (e.g., a human).
[0239] In certain embodiments, the disease or disorder is a disease of iron metabolism, such as, e.g., an iron overload disease, iron deficiency disorder, disorder of iron biodistribution, or another disorder of iron metabolism and other disorder potentially related to iron metabolism, etc. In particular embodiments, the disease of iron metabolism is hemochromatosis, HFE mutation hemochromatosis, ferroportin mutation hemochromatosis, transferrin receptor 2 mutation hemochromatosis, hemojuvelin mutation hemochromatosis, hepcidin mutation hemochromatosis, juvenile hemochromatosis, neonatal hemochromatosis, hepcidin deficiency, transfusional iron overload, thalassemia, thalassemia intermedia, alpha thalassemia, beta thalassemia, sideroblastic anemia, porphyria, porphyria cutanea tarda, African iron overload, hyperferritinemia, ceruloplasmin deficiency, atransferrinemia, congenital dyserythropoietic anemia, anemia of chronic disease, anemia of inflammation, anemia of infection, hypochromic microcytic anemia, iron-deficiency anemia, iron-refractory iron deficiency anemia, anemia of chronic kidney disease, transfusion-dependent anemia, hemolytic anemia, erythropoietin resistance, iron deficiency of obesity, other anemias, benign or malignant tumors that overproduce hepcidin or induce its overproduction, conditions with hepcidin excess, Friedreich ataxia, gracile syndrome, Hallervorden-Spatz disease, Wilson's disease, pulmonary hemosiderosis, hepatocellular carcinoma, cancer (e.g., liver cancer), hepatitis, cirrhosis of liver, pica, chronic renal failure, insulin resistance, diabetes, atherosclerosis, neurodegenerative disorders, dementia, multiple sclerosis, Parkinson's disease, Huntington's disease, or Alzheimer's disease.
[0240] In certain embodiments, the disease or disorder is related to iron overload diseases such as iron hemochromatosis, HFE mutation hemochromatosis, ferroportin mutation hemochromatosis, transferrin receptor 2 mutation hemochromatosis, hemojuvelin mutation hemochromatosis, hepcidin mutation hemochromatosis, juvenile hemochromatosis, neonatal hemochromatosis, hepcidin deficiency, transfusional iron overload, thalassemia, thalassemia intermedia, alpha thalassemia.
[0241] In certain embodiments, the disease or disorder is one that is not typically identified as being iron related. For example, hepcidin is highly expressed in the murine pancreas suggesting that diabetes (Type I or Type II), insulin resistance, glucose intolerance and other disorders may be ameliorated by treating underlying iron metabolism disorders. See Ilyin, G. et al. (2003) FEBS Lett. 542 22-26, which is herein incorporated by reference. As such, peptides of the invention may be used to treat these diseases and conditions. Those skilled in the art are readily able to determine whether a given disease can be treated with a peptide according to the present invention using methods known in the art, including the assays of WO 2004092405, which is herein incorporated by reference, and assays which monitor hepcidin, hemojuvelin, or iron levels and expression, which are known in the art such as those described in U.S. Pat. No. 7,534,764, which is herein incorporated by reference.
[0242] In certain embodiments, the disease or disorder is postmenopausal osteoporosis.
[0243] In certain embodiments of the present invention, the diseases of iron metabolism are iron overload diseases, which include hereditary hemochromatosis, iron-loading anemias, alcoholic liver diseases, heart disease and/or failure, cardiomyopathy, and chronic hepatitis C.
[0244] In particular embodiments, any of these diseases, disorders, or indications are caused by or associated with a deficiency of hepcidin or iron overload.
[0245] In some embodiments, methods of the present invention comprise providing a hepcidin analogue of the present invention (i.e., a first therapeutic agent) to a subject in need thereof in combination with a second therapeutic agent. In certain embodiments, the second therapeutic agent is provided to the subject before and/or simultaneously with and/or after the pharmaceutical composition is administered to the subject. In particular embodiments, the second therapeutic agent is iron chelator. In certain embodiments, the second therapeutic agent is selected from the iron chelators Deferoxamine and Deferasirox (Exjade.TM.) In another embodiment, the method comprises administering to the subject a third therapeutic agent.
[0246] The present invention provides compositions (for example pharmaceutical compositions) comprising one or more hepcidin analogues of the present invention and a pharmaceutically acceptable carrier, excipient or diluent. A pharmaceutically acceptable carrier, diluent or excipient refers to a non-toxic solid, semi-solid or liquid filler, diluent, encapsulating material or formulation auxiliary of any type. Prevention of the action of microorganisms may be ensured by the inclusion of various antibacterial and antifungal agents, for example, paraben, chlorobutanol, phenol sorbic acid, and the like. It may also be desirable to include isotonic agents such as sugars, sodium chloride, and the like.
[0247] The term "pharmaceutically acceptable carrier" includes any of the standard pharmaceutical carriers. Pharmaceutically acceptable carriers for therapeutic use are well known in the pharmaceutical art and are described, for example, in "Remington's Pharmaceutical Sciences", 17th edition, Alfonso R. Gennaro (Ed.), Mark Publishing Company, Easton, Pa., USA, 1985. For example, sterile saline and phosphate-buffered saline at slightly acidic or physiological pH may be used. Suitable pH-buffering agents may, e.g., be phosphate, citrate, acetate, tris(hydroxymethyl)aminomethane (TRIS), N-tris(hydroxymethyl)methyl-3-aminopropanesulfonic acid (TAPS), ammonium bicarbonate, diethanolamine, histidine, arginine, lysine or acetate (e.g. as sodium acetate), or mixtures thereof. The term further encompasses any carrier agents listed in the US Pharmacopeia for use in animals, including humans.
[0248] In certain embodiments, the compositions comprise two or more hepcidin analogues disclosed herein. In certain embodiments, the combination is selected from one of the following: (i) any two or more of the hepcidin analogue peptide monomers shown therein; (ii) any two or more of the hepcidin analogue peptide dimers disclosed herein; (iii) any one or more of the hepcidin analogue peptide monomers disclosed herein, and any one or more of the hepcidin analogue peptide dimers disclosed herein.
[0249] It is to be understood that the inclusion of a hepcidin analogue of the invention (i.e., one or more hepcidin analogue peptide monomers of the invention or one or more hepcidin analogue peptide dimers of the present invention) in a pharmaceutical composition also encompasses inclusion of a pharmaceutically acceptable salt or solvate of a hepcidin analogue of the invention. In particular embodiments, the pharmaceutical compositions further comprise one or more pharmaceutically acceptable carrier, excipient, or vehicle.
[0250] In certain embodiments, the invention provides a pharmaceutical composition comprising a hepcidin analogue, or a pharmaceutically acceptable salt or solvate thereof, for treating a variety of conditions, diseases, or disorders as disclosed herein or elsewhere (see, e.g., Methods of Treatment, herein). In particular embodiments, the invention provides a pharmaceutical composition comprising a hepcidin analogue peptide monomer, or a pharmaceutically acceptable salt or solvate thereof, for treating a variety of conditions, diseases, or disorders as disclosed herein elsewhere (see, e.g., Methods of Treatment, herein). In particular embodiments, the invention provides a pharmaceutical composition comprising a hepcidin analogue peptide dimer, or a pharmaceutically acceptable salt or solvate thereof, for treating a variety of conditions, diseases, or disorders as disclosed herein.
[0251] The hepcidin analogues of the present invention may be formulated as pharmaceutical compositions which are suited for administration with or without storage, and which typically comprise a therapeutically effective amount of at least one hepcidin analogue of the invention, together with a pharmaceutically acceptable carrier, excipient or vehicle.
[0252] In some embodiments, the hepcidin analogue pharmaceutical compositions of the invention are in unit dosage form. In such forms, the composition is divided into unit doses containing appropriate quantities of the active component or components. The unit dosage form may be presented as a packaged preparation, the package containing discrete quantities of the preparation, for example, packaged tablets, capsules or powders in vials or ampoules. The unit dosage form may also be, e.g., a capsule, cachet or tablet in itself, or it may be an appropriate number of any of these packaged forms. A unit dosage form may also be provided in single-dose injectable form, for example in the form of a pen device containing a liquid-phase (typically aqueous) composition. Compositions may be formulated for any suitable route and means of administration, e.g., any one of the routes and means of administration disclosed herein.
[0253] In particular embodiments, the hepcidin analogue, or the pharmaceutical composition comprising a hepcidin analogue, is suspended in a sustained-release matrix. A sustained-release matrix, as used herein, is a matrix made of materials, usually polymers, which are degradable by enzymatic or acid-base hydrolysis or by dissolution. Once inserted into the body, the matrix is acted upon by enzymes and body fluids. A sustained-release matrix desirably is chosen from biocompatible materials such as liposomes, polylactides (polylactic acid), polyglycolide (polymer of glycolic acid), polylactide co-glycolide (copolymers of lactic acid and glycolic acid) polyanhydrides, poly(ortho)esters, polypeptides, hyaluronic acid, collagen, chondroitin sulfate, carboxylic acids, fatty acids, phospholipids, polysaccharides, nucleic acids, polyamino acids, amino acids such as phenylalanine, tyrosine, isoleucine, polynucleotides, polyvinyl propylene, polyvinylpyrrolidone and silicone. One embodiment of a biodegradable matrix is a matrix of one of either polylactide, polyglycolide, or polylactide co-glycolide (co-polymers of lactic acid and glycolic acid).
[0254] In certain embodiments, the compositions are administered parenterally, subcutaneously or orally. In particular embodiments, the compositions are administered orally, intracisternally, intravaginally, intraperitoneally, intrarectally, topically (as by powders, ointments, drops, suppository, or transdermal patch, including delivery intravitreally, intranasally, and via inhalation) or buccally. The term "parenteral" as used herein refers to modes of administration which include intravenous, intramuscular, intraperitoneal, intrasternal, subcutaneous, intradermal and intra-articular injection and infusion. Accordingly, in certain embodiments, the compositions are formulated for delivery by any of these routes of administration.
[0255] In certain embodiments, pharmaceutical compositions for parenteral injection comprise pharmaceutically acceptable sterile aqueous or nonaqueous solutions, dispersions, suspensions or emulsions, or sterile powders, for reconstitution into sterile injectable solutions or dispersions just prior to use. Examples of suitable aqueous and nonaqueous carriers, diluents, solvents or vehicles include water, ethanol, polyols (such as glycerol, propylene glycol, polyethylene glycol, and the like), carboxymethylcellulose and suitable mixtures thereof, beta-cyclodextrin, vegetable oils (such as olive oil), and injectable organic esters such as ethyl oleate. Proper fluidity may be maintained, for example, by the use of coating materials such as lecithin, by the maintenance of the required particle size in the case of dispersions, and by the use of surfactants. These compositions may also contain adjuvants such as preservative, wetting agents, emulsifying agents, and dispersing agents. Prolonged absorption of an injectable pharmaceutical form may be brought about by the inclusion of agents which delay absorption, such as aluminum monostearate and gelatin.
[0256] Injectable depot forms include those made by forming microencapsule matrices of the hepcidin analogue in one or more biodegradable polymers such as polylactide-polyglycolide, poly(orthoesters), poly(anhydrides), and (poly)glycols, such as PEG. Depending upon the ratio of peptide to polymer and the nature of the particular polymer employed, the rate of release of the hepcidin analogue can be controlled. Depot injectable formulations are also prepared by entrapping the hepcidin analogue in liposomes or microemulsions compatible with body tissues.
[0257] The injectable formulations may be sterilized, for example, by filtration through a bacterial-retaining filter, or by incorporating sterilizing agents in the form of sterile solid compositions which can be dissolved or dispersed in sterile water or other sterile injectable medium just prior to use.
[0258] Hepcidin analogues of the present invention may also be administered in liposomes or other lipid-based carriers. As is known in the art, liposomes are generally derived from phospholipids or other lipid substances. Liposomes are formed by mono- or multi-lamellar hydrated liquid crystals that are dispersed in an aqueous medium. Any non-toxic, physiologically acceptable and metabolizable lipid capable of forming liposomes can be used. The present compositions in liposome form can contain, in addition to a hepcidin analogue of the present invention, stabilizers, preservatives, excipients, and the like. In certain embodiments, the lipids comprise phospholipids, including the phosphatidyl cholines (lecithins) and serines, both natural and synthetic. Methods to form liposomes are known in the art.
[0259] Pharmaceutical compositions to be used in the invention suitable for parenteral administration may comprise sterile aqueous solutions and/or suspensions of the peptide inhibitors made isotonic with the blood of the recipient, generally using sodium chloride, glycerin, glucose, mannitol, sorbitol, and the like.
[0260] In some aspects, the invention provides a pharmaceutical composition for oral delivery. Compositions and hepcidin analogues of the instant invention may be prepared for oral administration according to any of the methods, techniques, and/or delivery vehicles described herein. Further, one having skill in the art will appreciate that the hepcidin analogues of the instant invention may be modified or integrated into a system or delivery vehicle that is not disclosed herein, yet is well known in the art and compatible for use in oral delivery of peptides.
[0261] In certain embodiments, formulations for oral administration may comprise adjuvants (e.g. resorcinols and/or nonionic surfactants such as polyoxyethylene oleyl ether and n-hexadecylpolyethylene ether) to artificially increase the permeability of the intestinal walls, and/or enzymatic inhibitors (e.g. pancreatic trypsin inhibitors, diisopropylfluorophosphate (DFF) or trasylol) to inhibit enzymatic degradation. In certain embodiments, the hepcidin analogue of a solid-type dosage form for oral administration can be mixed with at least one additive, such as sucrose, lactose, cellulose, mannitol, trehalose, raffinose, maltitol, dextran, starches, agar, alginates, chitins, chitosans, pectins, gum tragacanth, gum arabic, gelatin, collagen, casein, albumin, synthetic or semisynthetic polymer, or glyceride. These dosage forms can also contain other type(s) of additives, e.g., inactive diluting agent, lubricant such as magnesium stearate, paraben, preserving agent such as sorbic acid, ascorbic acid, alpha-tocopherol, antioxidants such as cysteine, disintegrators, binders, thickeners, buffering agents, pH adjusting agents, sweetening agents, flavoring agents or perfuming agents.
[0262] In particular embodiments, oral dosage forms or unit doses compatible for use with the hepcidin analogues of the present invention may include a mixture of hepcidin analogue and nondrug components or excipients, as well as other non-reusable materials that may be considered either as an ingredient or packaging. Oral compositions may include at least one of a liquid, a solid, and a semi-solid dosage forms. In some embodiments, an oral dosage form is provided comprising an effective amount of hepcidin analogue, wherein the dosage form comprises at least one of a pill, a tablet, a capsule, a gel, a paste, a drink, a syrup, ointment, and suppository. In some instances, an oral dosage form is provided that is designed and configured to achieve delayed release of the hepcidin analogue in the subject's small intestine and/or colon.
[0263] In one embodiment, an oral pharmaceutical composition comprising a hepcidin analogue of the present invention comprises an enteric coating that is designed to delay release of the hepcidin analogue in the small intestine. In at least some embodiments, a pharmaceutical composition is provided which comprises a hepcidin analogue of the present invention and a protease inhibitor, such as aprotinin, in a delayed release pharmaceutical formulation. In some instances, pharmaceutical compositions of the instant invention comprise an enteric coat that is soluble in gastric juice at a pH of about 5.0 or higher. In at least one embodiment, a pharmaceutical composition is provided comprising an enteric coating comprising a polymer having dissociable carboxylic groups, such as derivatives of cellulose, including hydroxypropylmethyl cellulose phthalate, cellulose acetate phthalate and cellulose acetate trimellitate and similar derivatives of cellulose and other carbohydrate polymers.
[0264] In one embodiment, a pharmaceutical composition comprising a hepcidin analogue of the present invention is provided in an enteric coating, the enteric coating being designed to protect and release the pharmaceutical composition in a controlled manner within the subject's lower gastrointestinal system, and to avoid systemic side effects. In addition to enteric coatings, the hepcidin analogues of the instant invention may be encapsulated, coated, engaged or otherwise associated within any compatible oral drug delivery system or component. For example, in some embodiments a hepcidin analogue of the present invention is provided in a lipid carrier system comprising at least one of polymeric hydrogels, nanoparticles, microspheres, micelles, and other lipid systems.
[0265] To overcome peptide degradation in the small intestine, some embodiments of the present invention comprise a hydrogel polymer carrier system in which a hepcidin analogue of the present invention is contained, whereby the hydrogel polymer protects the hepcidin analogue from proteolysis in the small intestine and/or colon. The hepcidin analogues of the present invention may further be formulated for compatible use with a carrier system that is designed to increase the dissolution kinetics and enhance intestinal absorption of the peptide. These methods include the use of liposomes, micelles and nanoparticles to increase GI tract permeation of peptides.
[0266] Various bioresponsive systems may also be combined with one or more hepcidin analogue of the present invention to provide a pharmaceutical agent for oral delivery. In some embodiments, a hepcidin analogue of the instant invention is used in combination with a bioresponsive system, such as hydrogels and mucoadhesive polymers with hydrogen bonding groups (e.g., PEG, poly(methacrylic) acid [PMAA], cellulose, Eudragit.RTM., chitosan and alginate) to provide a therapeutic agent for oral administration. Other embodiments include a method for optimizing or prolonging drug residence time for a hepcidin analogue disclosed herein, wherein the surface of the hepcidin analogue surface is modified to comprise mucoadhesive properties through hydrogen bonds, polymers with linked mucins or/and hydrophobic interactions. These modified peptide molecules may demonstrate increase drug residence time within the subject, in accordance with a desired feature of the invention. Moreover, targeted mucoadhesive systems may specifically bind to receptors at the enterocytes and M-cell surfaces, thereby further increasing the uptake of particles containing the hepcidin analogue.
[0267] Other embodiments comprise a method for oral delivery of a hepcidin analogue of the present invention, wherein the hepcidin analogue is provided to a subject in combination with permeation enhancers that promote the transport of the peptides across the intestinal mucosa by increasing paracellular or transcellular permeation. For example, in one embodiment, a permeation enhancer is combined with a hepcidin analogue, wherein the permeation enhancer comprises at least one of a long-chain fatty acid, a bile salt, an amphiphilic surfactant, and a chelating agent. In one embodiment, a permeation enhancer comprising sodium N-[hydroxybenzoyl)amino] caprylate is used to form a weak noncovalent association with the hepcidin analogue of the instant invention, wherein the permeation enhancer favors membrane transport and further dissociation once reaching the blood circulation. In another embodiment, a hepcidin analogue of the present invention is conjugated to oligoarginine, thereby increasing cellular penetration of the peptide into various cell types. Further, in at least one embodiment a noncovalent bond is provided between a peptide inhibitor of the present invention and a permeation enhancer selected from the group consisting of a cyclodextrin (CD) and a dendrimers, wherein the permeation enhancer reduces peptide aggregation and increasing stability and solubility for the hepcidin analogue molecule.
[0268] Other embodiments of the invention provide a method for treating a subject with a hepcidin analogue of the present invention having an increased half-life. In one aspect, the present invention provides a hepcidin analogue having a half-life of at least several hours to one day in vitro or in vivo (e.g., when administered to a human subject) sufficient for daily (q.d.) or twice daily (b.i.d.) dosing of a therapeutically effective amount. In another embodiment, the hepcidin analogue has a half-life of three days or longer sufficient for weekly (q.w.) dosing of a therapeutically effective amount. Further, in another embodiment, the hepcidin analogue has a half-life of eight days or longer sufficient for bi-weekly (b.i.w.) or monthly dosing of a therapeutically effective amount. In another embodiment, the hepcidin analogue is derivatized or modified such that is has a longer half-life as compared to the underivatized or unmodified hepcidin analogue. In another embodiment, the hepcidin analogue contains one or more chemical modifications to increase serum half-life.
[0269] When used in at least one of the treatments or delivery systems described herein, a hepcidin analogue of the present invention may be employed in pure form or, where such forms exist, in pharmaceutically acceptable salt form.
[0270] The total daily usage of the hepcidin analogues and compositions of the present invention can be decided by the attending physician within the scope of sound medical judgment. The specific therapeutically effective dose level for any particular subject will depend upon a variety of factors including: a) the disorder being treated and the severity of the disorder; b) activity of the specific compound employed; c) the specific composition employed, the age, body weight, general health, sex and diet of the patient; d) the time of administration, route of administration, and rate of excretion of the specific hepcidin analogue employed; e) the duration of the treatment; 0 drugs used in combination or coincidental with the specific hepcidin analogue employed, and like factors well known in the medical arts.
[0271] In particular embodiments, the total daily dose of the hepcidin analogues of the invention to be administered to a human or other mammal host in single or divided doses may be in amounts, for example, from 0.0001 to 300 mg/kg body weight daily or 1 to 300 mg/kg body weight daily. In certain embodiments, a dosage of a hepcidin analogue of the present invention is in the range from about 0.0001 to about 100 mg/kg body weight per day, such as from about 0.0005 to about 50 mg/kg body weight per day, such as from about 0.001 to about 10 mg/kg body weight per day, e.g. from about 0.01 to about 1 mg/kg body weight per day, administered in one or more doses, such as from one to three doses. In particular embodiments, a total dosage is about 1 mg, about 2 mg, about 3 mg, about 4 mg, about 5 mg, about 6 mg, about 7 mg, about 8 mg, about 9 mg, or about 10 mg about once or twice weekly, e.g., for a human patient. In particular embodiments, the total dosage is in the range of about 1 mg to about 5 mg, or about 1 mg to about 3 mg, or about 2 mg to about 3 mg per human patient, e.g., about once weekly.
[0272] In various embodiments, a hepcidin analogue of the invention may be administered continuously (e.g. by intravenous administration or another continuous drug administration method), or may be administered to a subject at intervals, typically at regular time intervals, depending on the desired dosage and the pharmaceutical composition selected by the skilled practitioner for the particular subject. Regular administration dosing intervals include, e.g., once daily, twice daily, once every two, three, four, five or six days, once or twice weekly, once or twice monthly, and the like.
[0273] Such regular hepcidin analogue administration regimens of the invention may, in certain circumstances such as, e.g., during chronic long-term administration, be advantageously interrupted for a period of time so that the medicated subject reduces the level of or stops taking the medication, often referred to as taking a "drug holiday." Drug holidays are useful for, e.g., maintaining or regaining sensitivity to a drug especially during long-term chronic treatment, or to reduce unwanted side-effects of long-term chronic treatment of the subject with the drug. The timing of a drug holiday depends on the timing of the regular dosing regimen and the purpose for taking the drug holiday (e.g., to regain drug sensitivity and/or to reduce unwanted side effects of continuous, long-term administration). In some embodiments, the drug holiday may be a reduction in the dosage of the drug (e.g. to below the therapeutically effective amount for a certain interval of time). In other embodiments, administration of the drug is stopped for a certain interval of time before administration is started again using the same or a different dosing regimen (e.g. at a lower or higher dose and/or frequency of administration). A drug holiday of the invention may thus be selected from a wide range of time-periods and dosage regimens. An exemplary drug holiday is two or more days, one or more weeks, or one or more months, up to about 24 months of drug holiday. So, for example, a regular daily dosing regimen with a peptide, a peptide analogue, or a dimer of the invention may, for example, be interrupted by a drug holiday of a week, or two weeks, or four weeks, after which time the preceding, regular dosage regimen (e.g. a daily or a weekly dosing regimen) is resumed. A variety of other drug holiday regimens are envisioned to be useful for administering the hepcidin analogues of the invention.
[0274] Thus, the hepcidin analogues may be delivered via an administration regime which comprises two or more administration phases separated by respective drug holiday phases.
[0275] During each administration phase, the hepcidin analogue is administered to the recipient subject in a therapeutically effective amount according to a pre-determined administration pattern. The administration pattern may comprise continuous administration of the drug to the recipient subject over the duration of the administration phase. Alternatively, the administration pattern may comprise administration of a plurality of doses of the hepcidin analogue to the recipient subject, wherein said doses are spaced by dosing intervals.
[0276] A dosing pattern may comprise at least two doses per administration phase, at least five doses per administration phase, at least 10 doses per administration phase, at least 20 doses per administration phase, at least 30 doses per administration phase, or more.
[0277] Said dosing intervals may be regular dosing intervals, which may be as set out above, including once daily, twice daily, once every two, three, four, five or six days, once or twice weekly, once or twice monthly, or a regular and even less frequent dosing interval, depending on the particular dosage formulation, bioavailability, and pharmacokinetic profile of the hepcidin analogue of the present invention.
[0278] An administration phase may have a duration of at least two days, at least a week, at least 2 weeks, at least 4 weeks, at least a month, at least 2 months, at least 3 months, at least 6 months, or more.
[0279] Where an administration pattern comprises a plurality of doses, the duration of the following drug holiday phase is longer than the dosing interval used in that administration pattern. Where the dosing interval is irregular, the duration of the drug holiday phase may be greater than the mean interval between doses over the course of the administration phase. Alternatively the duration of the drug holiday may be longer than the longest interval between consecutive doses during the administration phase.
[0280] The duration of the drug holiday phase may be at least twice that of the relevant dosing interval (or mean thereof), at least 3 times, at least 4 times, at least 5 times, at least 10 times, or at least 20 times that of the relevant dosing interval or mean thereof.
[0281] Within these constraints, a drug holiday phase may have a duration of at least two days, at least a week, at least 2 weeks, at least 4 weeks, at least a month, at least 2 months, at least 3 months, at least 6 months, or more, depending on the administration pattern during the previous administration phase.
[0282] An administration regime comprises at least 2 administration phases. Consecutive administration phases are separated by respective drug holiday phases. Thus the administration regime may comprise at least 3, at least 4, at least 5, at least 10, at least 15, at least 20, at least 25, or at least 30 administration phases, or more, each separated by respective drug holiday phases.
[0283] Consecutive administration phases may utilise the same administration pattern, although this may not always be desirable or necessary. However, if other drugs or active agents are administered in combination with a hepcidin analogue of the invention, then typically the same combination of drugs or active agents is given in consecutive administration phases. In certain embodiments, the recipient subject is human.
[0284] In some embodiments, the present invention provides compositions and medicaments comprising at least one hepcidin analogue as disclosed herein. In some embodiments, the present invention provides a method of manufacturing medicaments comprising at least one hepcidin analogue as disclosed herein for the treatment of diseases of iron metabolism, such as iron overload diseases. In some embodiments, the present invention provides a method of manufacturing medicaments comprising at least one hepcidin analogue as disclosed herein for the treatment of diabetes (Type I or Type II), insulin resistance, or glucose intolerance. Also provided are methods of treating a disease of iron metabolism in a subject, such as a mammalian subject, and preferably a human subject, comprising administering at least one hepcidin analogue, or composition as disclosed herein to the subject. In some embodiments, the hepcidin analogue or the composition is administered in a therapeutically effective amount. Also provided are methods of treating diabetes (Type I or Type II), insulin resistance, or glucose intolerance in a subject, such as a mammalian subject, and preferably a human subject, comprising administering at least one hepcidin analogue or composition as disclosed herein to the subject. In some embodiments, the hepcidin analogue or composition is administered in a therapeutically effective amount.
[0285] In some embodiments, the invention provides a process for manufacturing a hepcidin analogue or a hepcidin analogue composition (e.g., a pharmaceutical composition), as disclosed herein.
[0286] In some embodiments, the invention provides a device comprising at least one hepcidin analogue of the present invention, or pharmaceutically acceptable salt or solvate thereof for delivery of the hepcidin analogue to a subject.
[0287] In some embodiments, the present invention provides methods of binding a ferroportin or inducing ferroportin internalization and degradation which comprises contacting the ferroportin with at least one hepcidin analogue, or hepcidin analogue composition as disclosed herein.
[0288] In some embodiments, the present invention provides kits comprising at least one hepcidin analogue, or hepcidin analogue composition (e.g., pharmaceutical composition) as disclosed herein packaged together with a reagent, a device, instructional material, or a combination thereof.
[0289] In some embodiments, the present invention provides a method of administering a hepcidin analogue or hepcidin analogue composition (e.g., pharmaceutical composition) of the present invention to a subject via implant or osmotic pump, by cartridge or micro pump, or by other means appreciated by the skilled artisan, as well-known in the art.
[0290] In some embodiments, the present invention provides complexes which comprise at least one hepcidin analogue as disclosed herein bound to a ferroportin, preferably a human ferroportin, or an antibody, such as an antibody which specifically binds a hepcidin analogue as disclosed herein, Hep25, or a combination thereof.
[0291] In some embodiments, the hepcidin analogue of the present invention has a measurement (e.g., an EC50) of less than 500 nM within the Fpn internalization assay. As a skilled person will realize, the function of the hepcidin analogue is dependent on the tertiary structure of the hepcidin analogue and the binding surface presented. It is therefore possible to make minor changes to the sequence encoding the hepcidin analogue that do not affect the fold or are not on the binding surface and maintain function. In other embodiments, the present invention provides a hepcidin analogue having 85% or higher (e.g., 85%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 99.5%) identity or homology to an amino acid sequence of any hepcidin analogue described herein that exhibits an activity (e.g., hepcidin activity), or lessens a symptom of a disease or indication for which hepcidin is involved.
[0292] In other embodiments, the present invention provides a hepcidin analogue having 85% or higher (e.g., 85%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 99.5%) identity or homology to an amino acid sequence of any hepcidin analogue presented herein, or a peptide according to any one of the formulae or hepcidin analogues described herein.
[0293] In some embodiments, a hepcidin analogue of the present invention may comprise functional fragments or variants thereof that have at most 10, 9, 8, 7, 6, 5, 4, 3, 2, or 1 amino acid substitutions compared to one or more of the specific peptide analogue sequences recited herein.
[0294] In addition to the methods described in the Examples herein, the hepcidin analogues of the present invention may be produced using methods known in the art including chemical synthesis, biosynthesis or in vitro synthesis using recombinant DNA methods, and solid phase synthesis. See e.g. Kelly & Winkler (1990) Genetic Engineering Principles and Methods, vol. 12, J. K. Setlow ed., Plenum Press, NY, pp. 1-19; Merrifield (1964) J Amer Chem Soc 85:2149; Houghten (1985) PNAS USA 82:5131-5135; and Stewart & Young (1984) Solid Phase Peptide Synthesis, 2ed. Pierce, Rockford, Ill., which are herein incorporated by reference. The hepcidin analogues of the present invention may be purified using protein purification techniques known in the art such as reverse phase high-performance liquid chromatography (HPLC), ion-exchange or immunoaffinity chromatography, filtration or size exclusion, or electrophoresis. See Olsnes, S. and A. Pihl (1973) Biochem. 12(16):3121-3126; and Scopes (1982) Protein Purification, Springer-Verlag, NY, which are herein incorporated by reference. Alternatively, the hepcidin analogues of the present invention may be made by recombinant DNA techniques known in the art. Thus, polynucleotides that encode the polypeptides of the present invention are contemplated herein. In certain preferred embodiments, the polynucleotides are isolated. As used herein "isolated polynucleotides" refers to polynucleotides that are in an environment different from that in which the polynucleotide naturally occurs.
EXAMPLES
[0295] The following examples demonstrate certain specific embodiments of the present invention. The following examples were carried out using standard techniques that are well known and routine to those of skill in the art, except where otherwise described in detail. It is to be understood that these examples are for illustrative purposes only and do not purport to be wholly definitive as to conditions or scope of the invention. As such, they should not be construed in any way as limiting the scope of the present invention.
Abbreviations
[0296] DCM: dichloromethane
DMF: N,N-dimethylformamide
NMP: N-methylpyrolidone
[0297] HBTU: O-(Benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium hexafluorophosphate HATU: 2-(7-aza-1H-benzotriazole-1-yl)-1,1,3,3-tetramethyluronium hexafluorophosphate
DCC: Dicyclohexylcarbodiimide
NHS: N-hydoxysuccinimide
[0298] DIPEA: diisopropylethylamine EtOH: ethanol Et2O: diethyl ether Hy: hydrogen TFA: trifluoroacetic acid TIS: triisopropylsilane ACN: acetonitrile HPLC: high performance liquid chromatography ESI-MS: electron spray ionization mass spectrometry PBS: phosphate-buffered saline Boc: t-butoxycarbonyl
Fmoc: Fluorenylmethyloxycarbonyl
[0299] Acm: acetamidomethyl IVA: Isovaleric acid (or Isovaleryl) K( ): In the peptide sequences provided herein, wherein a compound or chemical group is presented in parentheses directly after a Lysine residue, it is to be understood that the compound or chemical group in the parentheses is a side chain conjugated to the Lysine residue. So, e.g., but not to be limited in any way, K-[(PEG8)]- indicates that a PEG8 moiety is conjugated to a side chain of this Lysine. Palm: Indicates conjugation of a palmitic acid (palmitoyl).
[0300] As used herein "C( )" refers to a cysteine residue involved in a particular disulfide bridge. For example, in Hepcidin, there are four disulfide bridges: the first between the two C(1) residues; the second between the two C(2) residues; the third between the two C(3) residues; and the fourth between the two C(4) residues. Accordingly, in some embodiments, the sequence for Hepcidin is written as follows:
TABLE-US-00020 (SEQ ID NO: 39) Hy-DTHFPIC(1)IFC(2)C(3)GC(2)C(4)HRSKC(3)GMC(4)C(1) KT-OH;
and the sequence for other peptides may also optionally be written in the same manner.
Example 1
Synthesis of Peptide Analogues
[0301] Unless otherwise specified, reagents and solvents employed in the following were available commercially in standard laboratory reagent or analytical grade, and were used without further purification.
[0302] Procedure for Solid-Phase Synthesis of Peptides
[0303] Peptide analogues of the invention were chemically synthesized using optimized 9-fluorenylmethoxy carbonyl (Fmoc) solid phase peptide synthesis protocols. For C-terminal amides, rink-amide resin was used, although wang and trityl resins were also used to produce C-terminal acids. The side chain protecting groups were as follows: Glu, Thr and Tyr: 0-tButyl; Trp and Lys: t-Boc (t-butyloxycarbonyl); Arg: N-gamma-2,2,4,6,7-pentamethyldihydrobenzofuran-5-sulfonyl; His, Gln, Asn, Cys: Trityl. For selective disulfide bridge formation, Acm (acetamidomethyl) was also used as a Cys protecting group. For coupling, a four to ten-fold excess of a solution containing Fmoc amino acid, HBTU and DIPEA (1:1:1.1) in DMF was added to swelled resin [HBTU: 0-(Benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium hexafluorophosphate; DIPEA: diisopropylethylamine; DMF: dimethylformamide]. HATU (O-(7-azabenzotriazol-1-yl)-1,1,3,3,-tetramethyluronium hexafluorophosphate) was used instead of HBTU to improve coupling efficiency in difficult regions. Fmoc protecting group removal was achieved by treatment with a DMF, piperidine (2:1) solution.
[0304] Procedure for Cleavage of Peptides Off Resin
[0305] Side chain deprotection and cleavage of the peptide analogues of the invention (e.g., Compound No. 2) was achieved by stirring dry resin in a solution containing trifluoroacetic acid, water, ethanedithiol and tri-isopropylsilane (90:5:2.5:2.5) for 2 to 4 hours. Following TFA removal, peptide was precipitated using ice-cold diethyl ether. The solution was centrifuged and the ether was decanted, followed by a second diethyl ether wash. The peptide was dissolved in an acetonitrile, water solution (1:1) containing 0.1% TFA (trifluoroacetic acid) and the resulting solution was filtered. The linear peptide quality was assessed using electrospray ionization mass spectrometry (ESI-MS).
[0306] Procedure for Purification of Peptides
[0307] Purification of the peptides of the invention (e.g., Compound No. 2) was achieved using reverse-phase high performance liquid chromatography (RP-HPLC). Analysis was performed using a C18 column (3 .mu.m, 50.times.2 mm) with a flow rate of 1 mL/min. Purification of the linear peptides was achieved using preparative RP-HPLC with a C18 column (5 .mu.m, 250.times.21.2 mm) with a flow rate of 20 mL/min. Separation was achieved using linear gradients of buffer B in A (Buffer A: Aqueous 0.05% TFA; Buffer B: 0.043% TFA, 90% acetonitrile in water).
[0308] Procedure for Oxidation of Peptides
[0309] Method a (Single Disulfide Oxidation).
[0310] Oxidation of the unprotected peptides of the invention was achieved by adding drop-wise iodine in MeOH (1 mg per 1 mL) to the peptide in a solution (ACN: H.sub.2O, 7: 3, 0.5% TFA). After stirring for 2 min, ascorbic acid portion wise was added until the solution was clear and the sample was immediately loaded onto the HPLC for purification.
[0311] Method B (Selective Oxidation of Two Disulfides).
[0312] When more than one disulfide was present, selective oxidation was often performed. Oxidation of the free cysteines was achieved at pH 7.6 NH.sub.4CO.sub.3 solution at 1 mg/10 mL of peptide. After 24 h stirring and prior to purification the solution was acidified to pH 3 with TFA followed by lyophilization. The resulting single oxidized peptides (with ACM protected cysteines) were then oxidized/selective deprotection using iodine solution. The peptide (1 mg per 2 mL) was dissolved in MeOH/H.sub.2O, 80:20 iodine dissolved in the reaction solvent was added to the reaction (final concentration: 5 mg/mL) at room temperature. The solution was stirred for 7 minutes before ascorbic acid was added portion wise until the solution is clear. The solution was then loaded directly onto the HPLC.
[0313] Method C (Native Oxidation).
[0314] When more than one disulfide was present and when not performing selective oxidations, native oxidation was performed. Native oxidation was achieved with 100 mM NH4CO3 (pH7.4) solution in the presence of oxidized and reduced glutathione (peptide/GSH/GSSG, 1:100:10 molar ratio) of (peptide: GSSG: GSH, 1:10, 100). After 24 h stirring and prior to RP-HPLC purification the solution was acidified to pH 3 with TFA followed by lyophilization.
[0315] Procedure of Cysteine Oxidation to Produce Dimers.
[0316] Oxidation of the unprotected peptides of the invention was achieved by adding drop-wise iodine in MeOH (1 mg per 1 mL) to the peptide in a solution (ACN: H2O, 7: 3, 0.5% TFA). After stirring for 2 min, ascorbic acid portion wise was added until the solution was clear and the sample was immediately loaded onto the HPLC for purification.
[0317] Procedure for Dimerization.
[0318] Glyoxylic acid (DIG), IDA, or Fmoc-.beta.-Ala-IDA was pre-activated as the N-hydoxysuccinimide ester by treating 1 equivalent (abbreviated "eq") of the acid with 2.2 eq of both N-hydoxysuccinimide (NHS) and dicyclohexyl carbodiimide (DCC) in NMP (N-methyl pyrolidone) at a 0.1 M final concentration. For the PEG13 and PEG25 linkers, these chemical entities were purchased pre-formed as the activated succinimide ester. The activated ester .about.0.4 eq was added slowly to the peptide in NMP (1 mg/mL) portionwise. The solution was left stirring for 10 min before 2-3 additional aliquots of the linker .about.0.05 eq were slowly added. The solution was left stirring for a further 3 h before the solvent was removed under vacuo and the residue was purified by reverse phase HPLC. An additional step of stirring the peptide in 20% piperidine in DMF (2.times.10 min) before an additional reverse phase HPLC purification was performed.
[0319] One of skill in the art will appreciate that standard methods of peptide synthesis may be used to generate the compounds of the invention.
[0320] Linker Activation and Dimerization
[0321] Peptide monomer subunits were linked to form hepcidin analogue peptide dimers as described below.
[0322] Small Scale DIG Linker Activation Procedure:
[0323] 5 mL of NMP was added to a glass vial containing IDA diacid (304.2 mg, 1 mmol), N-hydroxysuccinimide (NHS, 253.2 mg, 2.2 eq. 2.2 mmol) and a stirring bar. The mixture was stirred at room temperature to completely dissolve the solid starting materials. N, N'-Dicyclohexylcarbodiimide (DCC, 453.9 mg, 2.2 eq., 2.2 mmol) was then added to the mixture. Precipitation appeared within 10 min and the reaction mixture was further stirred at room temperature overnight. The reaction mixture was then filtered to remove the precipitated dicyclohexylurea (DCU). The activated linker was kept in a closed vial prior to use for dimerization. The nominal concentration of the activated linker was approximately 0.20 M.
[0324] For dimerization using PEG linkers, there was no pre-activation step involved. Commercially available pre-activated bi-functional PEG linkers were used.
[0325] Dimerization Procedure:
[0326] 2 mL of anhydrous DMF was added to a vial containing peptide monomer (0.1 mmol). The pH of the peptide was the adjusted to 8-9 with DIEA. Activated linker (IDA or PEG13, PEG 25) (0.48 eq relative to monomer, 0.048 mmol) was then added to the monomer solution. The reaction mixture was stirred at room temperature for one hour. Completion of the dimerization reaction was monitored using analytical HPLC. The time for completion of dimerization reaction varied depending upon the linker. After completion of reaction, the peptide was precipitated in cold ether and centrifuged. The supernatant ether layer was discarded. The precipitation step was repeated twice. The crude dimer was then purified using reverse phase HPLC (Luna C18 support, 10 u, 100 A, Mobile phase A: water containing 0.1% TFA, mobile phase B: Acetonitrile (ACN) containing 0.1% TFA, gradient of 15% B and change to 45% B over 60 min, flow rate 15 ml/min). Fractions containing pure product were then freeze-dried on a lyophilizer.
[0327] Conjugation of Half-Life Extension Moieties
[0328] Conjugation of peptides were performed on resin. Lys(ivDde) was used as the key amino acid. After assembly of the peptide on resin, selective deprotection of the ivDde group occurred using 3.times.5 min 2% hydrazine in DMF for 5 min. Activation and acylation of the linker using HBTU, DIEA 1-2 equivalents for 3 h, and Fmoc removal followed by a second acylation with the lipidic acid gave the conjugated peptide.
Example 2
Activity of Peptide Analogues
[0329] Peptide analogues were tested in vitro for induction of internalization of the human ferroportin protein. Following internalization, the peptides are degraded. The assay measures a decrease in fluorescence of the receptor.
[0330] The cDNA encoding the human ferroportin (SLC40 A1) was cloned from a cDNA clone from Origene (NM_014585). The DNA encoding the ferroportin was amplified by PCR using primers also encoding terminal restriction sites for subcloning, but without the termination codon. The ferroportin receptor was subcloned into a mammalian GFP expression vector containing a neomycin (G418) resistance marker in such that the reading frame of the ferroportin was fused in frame with the GFP protein. The fidelity of the DNA encoding the protein was confirmed by DNA sequencing. HEK293 cells were used for transfection of the ferroportin-GFP receptor expression plasmid. The cells were grown according to standard protocol in growth medium and transfected with the plasmids using Lipofectamine (manufacturer's protocol, Invitrogen). The cells stably expressing ferroportin-GFP were selected using G418 in the growth medium (in that only cells that have taken up and incorporated the cDNA expression plasmid survive) and sorted several times on a Cytomation MoFlo.TM. cell sorter to obtain the GFP-positive cells (488 nm/530 nm). The cells were propagated and frozen in aliquots.
[0331] To determine activity of the hepcidin analogues (compounds) on the human ferroportin, the cells were incubated in 96 well plates in standard media, without phenol red. Compound was added to desired final concentration for at least 18 hours in the incubator. Following incubation, the remaining GFP-fluorescence was determined either by whole cell GFP fluorescence (Envision plate reader, 485/535 filter pair), or by Beckman Coulter Quanta.TM. flow cytometer (express as Geometric mean of fluorescence intensity at 485 nm/525 nm). Compound was added to desired final concentration for at least 18 hours but no more than 24 hours in the incubator.
[0332] In certain experiments, reference compounds included native Hepcidin, Mini-Hepcidin, and R1-Mini-Hepcidin, which is an analog of mini-hepcidin. The "RI" in RI-Mini-Hepcidin refers to Retro Inverse. A retro inverse peptide is a peptide with a reversed sequence in all D amino acids. An example is that Hy-Glu-Thr-His-NH2 becomes Hy-DHis-DThr-DGlu-NH2. The EC.sub.50 of these reference compounds for ferroportin degradation was determined according to the activity assay described above. These peptides served as control standards.
TABLE-US-00021 TABLE 9 Reference compounds Potency EC50 Name Sequence (nM) Hepcidin Hy-DTHFPIC(1)IFC(2)C(3)GC(2)C(4)HRSKC(3)GMC(4)C(1) 34 KT-OH (SEQ ID NO: 39) Mini- Hy-DTHFPICIF-NH.sub.2 (SEQ ID NO: 40) 712 Hepcidin 1-9 RI-Mini Hy-DPhe-DIle-DCys-DIle-DPro-DPhe-DHis-DThr-DAsp- >10 .mu.M Hepcidin NH.sub.2 (SEQ ID NO: 41)
[0333] The potency EC.sub.50 values (nM) determined for various peptide analogues of the present invention are provided in Tables 3 and 4. These values were determined as described herein.
Example 3
In Vivo Validation of Peptide Analogues
[0334] Hepcidin analogues of the present invention were tested for in vivo activity, to determine their ability to decrease free Fe2+ in serum.
[0335] A hepcidin analogue (Compound 1) or vehicle control were administered to mice (n=3/group) at 1000 nmol/kg either intravenously or subcutaneously. Serum samples were taken from groups of mice administered with the hepcidin analog at 30 min, 1 h, 2 h, 4 h, 10 h, 24 h, 30 h, 36 h, and 48 h post-administration. Iron content in plasma/serum was measured using a colorimetric assay on the Cobas c 111 according to instructions from the manufacturer of the assay (assay: IRON2: ACN 661). The data obtained from the cobas Iron2 analysis is presented in FIG. 1A (intravenous administration) and FIG. 1B (subcutaneous administration) as mean values+/-SEM.
[0336] In another experiment, various hepcidin analogues or vehicle control were administered to mice (n=3/group) at 1000 nmol/kg subcutaneously. Serum samples were taken from groups of mice administered with vehicle or hepcidin analog at 30 h and 36 h post-administration. Iron content in plasma/serum was measured using a colorimetric assay on the Cobas c 111 according to instructions from the manufacturer of the assay (assay: IRON2: ACN 661). The data obtained from the cobas Iron2 analysis is presented in FIG. 2 as mean values+/-SEM.
[0337] These studies demonstrate that hepcidin analogues of the present invention reduce serum iron levels for at least 30 hours, thus demonstrating their increased serum stability.
[0338] All of the above U.S. patents, U.S. patent application publications, U.S. patent applications, foreign patents, foreign patent applications and non-patent publications referred to in this specification and/or listed in the Application Data Sheet, are incorporated herein by reference, in their entirety.
[0339] From the foregoing it will be appreciated that, although specific embodiments of the invention have been described herein for purposes of illustration, various modifications may be made without deviating from the spirit and scope of the invention.
Sequence CWU 1
1
41110PRTArtificial SequenceHepcidin analogue peptide
formulaMOD_RES(1)..(1)Xaa is Asp, isoGlu or IdaMOD_RES(4)..(4)Xaa
is Phe, Phe(4-F), Phe(4-CN), 4-BIP, Phe(4-OCH3), Tyr,
Phe(2,3-(OCH3)2), Phe(2,3-Cl2), or DpaMOD_RES(6)..(6)Xaa is Cys or
PenMOD_RES(7)..(7)Xaa is any amino acidmisc_feature(8)..(8)Xaa can
be any naturally occurring amino acidMOD_RES(10)..(10)Xaa is Lys,
Glu or absent 1Xaa Thr His Xaa Pro Xaa Xaa Xaa Phe Xaa 1 5 10
27PRTArtificial SequenceHepcidin analogue peptide
formulaMOD_RES(1)..(5)Xaa is any amino acidMOD_RES(6)..(6)Xaa is
Cys or PenMOD_RES(7)..(7)Xaa is Lys or absent 2Xaa Xaa Xaa Xaa Xaa
Xaa Xaa 1 5 310PRTArtificial SequenceHepcidin analogue peptide
formulaMOD_RES(4)..(4)Phe or DpaMOD_RES(6)..(6)Cys or
PenMOD_RES(7)..(7)Ile or LysMOD_RES(8)..(8)Lys or
ArgMOD_RES(10)..(10)Lys, Glu or absent 3Asp Thr His Xaa Pro Xaa Xaa
Xaa Phe Xaa 1 5 10 47PRTArtificial SequenceHepcidin analogue
peptide formulaMOD_RES(5)..(5)Gly or SarMOD_RES(6)..(6)Cys or
PenMOD_RES(7)..(7)Lys or absent 4Pro Arg Ser Lys Xaa Xaa Xaa 1 5
517PRTArtificial SequenceSynthesized hepcidin analogue 5Asp Thr His
Phe Pro Cys Ile Lys Phe Lys Pro Arg Ser Lys Gly Cys 1 5 10 15 Lys
617PRTArtificial SequenceSynthesized hepcidin analogue 6Asp Thr His
Phe Pro Cys Ile Lys Phe Glu Pro Arg Ser Lys Gly Cys 1 5 10 15 Lys
717PRTArtificial SequenceSynthesized hepcidin
analogueMOD_RES(8)..(8)IsoGlu-Palm conjugated half-life extension
moiety 7Asp Thr His Phe Pro Cys Ile Lys Phe Glu Pro Arg Ser Lys Gly
Cys 1 5 10 15 Lys 817PRTArtificial SequenceSynthesized hepcidin
analogueMOD_RES(10)..(10)IsoGlu-Palm conjugated half-life extension
moiety 8Asp Thr His Phe Pro Cys Ile Lys Phe Lys Pro Arg Ser Lys Gly
Cys 1 5 10 15 Lys 917PRTArtificial SequenceSynthesized hepcidin
analogueMOD_RES(6)..(6)PenMOD_RES(10)..(10)IsoGlu-Palm conjugated
half-life extension moietyMOD_RES(16)..(16)Pen 9Asp Thr His Phe Pro
Xaa Ile Lys Phe Lys Pro Arg Ser Lys Gly Xaa 1 5 10 15 Lys
1017PRTArtificial SequenceSynthesized hepcidin
analogueMOD_RES(6)..(6)PenMOD_RES(10)..(10)IsoGlu-Palm conjugated
half-life extension moiety 10Asp Thr His Phe Pro Xaa Ile Lys Phe
Lys Pro Arg Ser Lys Gly Cys 1 5 10 15 Lys 1117PRTArtificial
SequenceSynthesized hepcidin analogueMOD_RES(10)..(10)IsoGlu-Palm
conjugated half-life extension moietyMOD_RES(16)..(16)Pen 11Asp Thr
His Phe Pro Cys Ile Lys Phe Lys Pro Arg Ser Lys Gly Xaa 1 5 10 15
Lys 1217PRTArtificial SequenceSynthesized hepcidin
analogueMOD_RES(10)..(10)IsoGlu-Palm conjugated half-life extension
moietyMOD_RES(15)..(15)Sar (sarcosine0 12Asp Thr His Phe Pro Cys
Ile Lys Phe Lys Pro Arg Ser Lys Xaa Cys 1 5 10 15 Lys
1317PRTArtificial SequenceSynthesized hepcidin
analogueMOD_RES(4)..(4)beta, beta
diphenylalanineMOD_RES(10)..(10)IsoGlu-Palm conjugated half-life
extension moiety 13Asp Thr His Xaa Pro Cys Ile Lys Phe Lys Pro Arg
Ser Lys Gly Cys 1 5 10 15 Lys 1417PRTArtificial SequenceSynthesized
hepcidin analogueMOD_RES(4)..(4)beta, beta
diphenylalanineMOD_RES(10)..(10)IsoGlu-Palm conjugated half-life
extension moietyMOD_RES(15)..(15)Sar (sarcosine) 14Asp Thr His Xaa
Pro Cys Ile Lys Phe Lys Pro Arg Ser Lys Xaa Cys 1 5 10 15 Lys
1516PRTArtificial SequenceSynthesized hepcidin
analogueMOD_RES(10)..(10)IsoGlu-Palm conjugated half-life extension
moiety 15Asp Thr His Phe Pro Cys Ile Lys Phe Lys Pro Arg Ser Lys
Gly Cys 1 5 10 15 1616PRTArtificial SequenceSynthesized hepcidin
analogueMOD_RES(10)..(10)IsoGlu-Palm conjugated half-life extension
moietyMOD_RES(15)..(15)Sar (sarcosine0 16Asp Thr His Phe Pro Cys
Ile Lys Phe Lys Pro Arg Ser Lys Xaa Cys 1 5 10 15 1717PRTArtificial
SequenceSynthesized hepcidin analogueMOD_RES(10)..(10)IsoGlu-Palm
conjugated half-life extension moiety 17Asp Thr His Phe Pro Cys Lys
Lys Phe Lys Pro Arg Ser Lys Gly Cys 1 5 10 15 Lys 1817PRTArtificial
SequenceSynthesized hepcidin analogueMOD_RES(10)..(10)IsoGlu-Lauric
acid conjugated half-life extension moiety 18Asp Thr His Phe Pro
Cys Ile Lys Phe Lys Pro Arg Ser Lys Gly Cys 1 5 10 15 Lys
1917PRTArtificial SequenceSynthesized hepcidin
analogueMOD_RES(10)..(10)IsoGlu-Myristic acid conjugated half-life
extension moiety 19Asp Thr His Phe Pro Cys Ile Lys Phe Lys Pro Arg
Ser Lys Gly Cys 1 5 10 15 Lys 2017PRTArtificial SequenceSynthesized
hepcidin analogueMOD_RES(10)..(10)IsoGlu-Bioitin conjugated
half-life extension moiety 20Asp Thr His Phe Pro Cys Ile Lys Phe
Lys Pro Arg Ser Lys Gly Cys 1 5 10 15 Lys 2117PRTArtificial
SequenceSynthesized hepcidin
analogueMOD_RES(10)..(10)IsoGlu-Isovaleric acid conjugated
half-life extension moiety 21Asp Thr His Phe Pro Cys Ile Lys Phe
Lys Pro Arg Ser Lys Gly Cys 1 5 10 15 Lys 2217PRTArtificial
SequenceSynthesized hepcidin analogueMOD_RES(10)..(10)PEG2-Palm
conjugated half-life extension moiety 22Asp Thr His Phe Pro Cys Ile
Lys Phe Lys Pro Arg Ser Lys Gly Cys 1 5 10 15 Lys 2317PRTArtificial
SequenceSynthesized hepcidin analogueMOD_RES(10)..(10)PEG11-Palm
conjugated half-life extension moiety 23Asp Thr His Phe Pro Cys Ile
Lys Phe Lys Pro Arg Ser Lys Gly Cys 1 5 10 15 Lys 2417PRTArtificial
SequenceSynthesized hepcidin analogueMOD_RES(10)..(10)PEG11-Lauric
acid conjugated half-life extension moiety 24Asp Thr His Phe Pro
Cys Ile Lys Phe Lys Pro Arg Ser Lys Gly Cys 1 5 10 15 Lys
2517PRTArtificial SequenceSynthesized hepcidin
analogueMOD_RES(10)..(10)PEG11-Myristic acid conjugated half-life
extension moiety 25Asp Thr His Phe Pro Cys Ile Lys Phe Lys Pro Arg
Ser Lys Gly Cys 1 5 10 15 Lys 2617PRTArtificial SequenceSynthesized
hepcidin analogueMOD_RES(10)..(10)Palm conjugated half-life
extension moiety 26Asp Thr His Phe Pro Cys Ile Lys Phe Lys Pro Arg
Ser Lys Gly Cys 1 5 10 15 Lys 2717PRTArtificial SequenceSynthesized
hepcidin analogueMOD_RES(10)..(10)PEG8 conjugated half-life
extension moiety 27Asp Thr His Phe Pro Cys Ile Lys Phe Lys Pro Arg
Ser Lys Gly Cys 1 5 10 15 Lys 2817PRTArtificial SequenceSynthesized
hepcidin analogueMOD_RES(10)..(10)Ahx-Palm conjugated half-life
extension moiety 28Asp Thr His Phe Pro Cys Ile Lys Phe Lys Pro Arg
Ser Lys Gly Cys 1 5 10 15 Lys 2917PRTArtificial SequenceSynthesized
hepcidin analogueMOD_RES(8)..(8)Ahx-Palm conjugated half-life
extension moietyMOD_RES(15)..(15)Sar (sarcosine) 29Asp Thr His Phe
Pro Cys Ile Lys Phe Glu Pro Arg Ser Lys Xaa Cys 1 5 10 15 Lys
3017PRTArtificial SequenceSynthesized hepcidin
analogueMOD_RES(10)..(10)Ahx-Palm conjugated half-life extension
moietyMOD_RES(15)..(15)Sar (sarcosine) 30Asp Thr His Phe Pro Cys
Ile Lys Phe Lys Pro Arg Ser Lys Xaa Cys 1 5 10 15 Lys
3117PRTArtificial SequenceSynthesized hepcidin
analogueMOD_RES(10)..(10)Ac conjugated half-life extension moiety
31Asp Thr His Phe Pro Cys Ile Lys Phe Lys Pro Arg Ser Lys Gly Cys 1
5 10 15 Lys 3217PRTArtificial SequenceSynthesized hepcidin
analogueMOD_RES(8)..(8)PEG11-Palm conjugated half-life extension
moietyMOD_RES(15)..(15)Sar (sarcosine) 32Asp Thr His Phe Pro Cys
Ile Lys Phe Glu Pro Arg Ser Lys Xaa Cys 1 5 10 15 Lys
3317PRTArtificial SequenceSynthesized hepcidin
analogueMOD_RES(10)..(10)PEG11-Palm conjugated half-life extension
moietyMOD_RES(15)..(15)Sar (sarcosine) 33Asp Thr His Phe Pro Cys
Ile Lys Phe Lys Pro Arg Ser Lys Xaa Cys 1 5 10 15 Lys
3417PRTArtificial SequenceSynthesized hepcidin
analogueMOD_RES(8)..(8)isoGlu-Ahx-Palm conjugated half-life
extension moietyMOD_RES(15)..(15)Sar (sarcosine) 34Asp Thr His Phe
Pro Cys Ile Lys Phe Glu Pro Arg Ser Lys Xaa Cys 1 5 10 15 Lys
3517PRTArtificial SequenceSynthesized hepcidin
analogueMOD_RES(8)..(8)OEG-OEG-isoGlu-(C18-diacid) conjugated
half-life extension moietyMOD_RES(15)..(15)Sar (sarcosine) 35Asp
Thr His Phe Pro Cys Ile Lys Phe Glu Pro Arg Ser Lys Xaa Cys 1 5 10
15 Lys 3610PRTArtificial SequenceSynthesized hepcidin
analogueMOD_RES(10)..(10)IsoGlu-Octanoic acid conjugated half-life
extension moiety 36Asp Thr His Phe Pro Cys Ile Arg Phe Lys 1 5 10
3711PRTArtificial SequenceSynthesized hepcidin
analogueMOD_RES(10)..(10)IDA (Iminodiacetic
acid)MOD_RES(11)..(11)beta alanine with Peg2-Palm conjugated
half-life extension moiety 37Asp Thr His Phe Pro Cys Ile Lys Phe
Xaa Xaa 1 5 10 3811PRTArtificial SequenceSynthesized hepcidin
analogueMOD_RES(10)..(10)IDA (Iminodiacetic
acid)MOD_RES(11)..(11)beta alanine with Peg11-Palm conjugated
half-life extension moiety 38Asp Thr His Phe Pro Cys Ile Lys Phe
Xaa Xaa 1 5 10 3925PRTHomo sapiens 39Asp Thr His Phe Pro Ile Cys
Ile Phe Cys Cys Gly Cys Cys His Arg 1 5 10 15 Ser Lys Cys Gly Met
Cys Cys Lys Thr 20 25 409PRTHomo sapiens 40Asp Thr His Phe Pro Ile
Cys Ile Phe 1 5 419PRTArtificial SequenceSynthesized retro inverse
peptide with D amino acidsMOD_RES(1)..(9)All amino acids are D
amino acids 41Phe Ile Cys Ile Pro Phe His Thr Asp 1 5































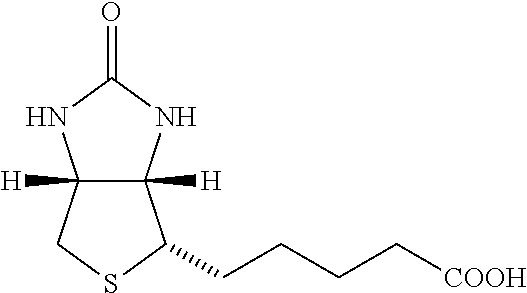
















D00001

D00002

D00003

S00001
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.