An Eye Drop Solution For Enhanced Hyaluronic Acid Retention And Delivery
Singh; Anirudha ; et al.
U.S. patent application number 15/753674 was filed with the patent office on 2019-01-03 for an eye drop solution for enhanced hyaluronic acid retention and delivery. The applicant listed for this patent is THE JOHNS HOPKINS UNIVERSITY. Invention is credited to Jennifer H. Elisseeff, David Lee, Nicole Lu, Anirudha Singh.
| Application Number | 20190000983 15/753674 |
| Document ID | / |
| Family ID | 58051345 |
| Filed Date | 2019-01-03 |


| United States Patent Application | 20190000983 |
| Kind Code | A1 |
| Singh; Anirudha ; et al. | January 3, 2019 |
AN EYE DROP SOLUTION FOR ENHANCED HYALURONIC ACID RETENTION AND DELIVERY
Abstract
The present invention provides biomaterial compositions that can immobilize HA to the ocular surfaces through an HABpep and transmembrane mucins and collagen of ocular tissues can act as anchoring sites. These biomaterial compositions provide prolonged HA binding and retention in both ex vivo and in vivo animal models. HA eye drop solutions with these the inventive biomaterials can prolong the biological and physical benefits to the ocular surface and potentially be more effective in treating eye disorders including dry eye than standard HA-eye drop presently available. Methods of making and use of the compositions are also provided.
| Inventors: | Singh; Anirudha; (Baltimore, MD) ; Elisseeff; Jennifer H.; (Baltimore, MD) ; Lee; David; (Baltimore, MD) ; Lu; Nicole; (Baltimore, MD) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 58051345 | ||||||||||
| Appl. No.: | 15/753674 | ||||||||||
| Filed: | August 17, 2016 | ||||||||||
| PCT Filed: | August 17, 2016 | ||||||||||
| PCT NO: | PCT/US2016/047248 | ||||||||||
| 371 Date: | February 20, 2018 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 62207417 | Aug 20, 2015 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | A61K 38/08 20130101; H01L 2224/75745 20130101; A61P 27/04 20180101; H01L 21/67092 20130101; H01L 24/00 20130101; A61K 47/60 20170801; H01L 2224/75821 20130101; H01L 21/67132 20130101; A61K 31/728 20130101; H01L 24/75 20130101 |
| International Class: | A61K 47/60 20060101 A61K047/60; A61K 38/08 20060101 A61K038/08; A61K 31/728 20060101 A61K031/728; A61P 27/04 20060101 A61P027/04 |
Claims
1. A biomaterial comprising at least one biologically compatible polymer having one or more HA binding peptides (HABPep) covalently linked to the biologically compatible polymer, and one or more sialic acid binding peptides (SABPep) covalently linked to the biologically compatible polymer.
2. (canceled)
3. A biomaterial comprising at least one biologically compatible polymer having one or more HA binding peptides (HABPep) covalently linked to the biologically compatible polymer, and one or more sialic acid binding peptides (SABPep) and one or more collagen binding peptides (ColBPep) covalently linked to the biologically compatible polymer.
4. The biomaterial of claim 1, wherein HA is mixed into the biologically compatible polymer.
5. The biomaterial of claim 1, further comprising HA bound to the HABPep.
6. The biomaterial of claim 5, wherein the HA has a molecular weight of between 5000 Da and 20,000,000 Da.
7. The biomaterial of claim 6, wherein the HA is crosslinked.
8. The biomaterial of claim 1, wherein the HABPep is a peptide comprising the following amino acid sequence: TABLE-US-00002 (SEQ ID NO: 1) RRDDGAHWQFNALTVR, (SEQ ID NO: 2) CRRDDGAHWQFNALTVR, (SEQ ID NO: 3) GAAWQFNALTVR, (SEQ ID NO: 4) GAHWQFAALTVR, (SEQ ID NO: 5) GAHWQFNALTVA, (SEQ ID NO: 6) GAHWQFNALTVR, (SEQ ID NO: 7) STMMSRSHKTRSHHV, (SEQ ID NO: 8) RYPISRPRKRC, (SEQ ID NO: 9) TAGHGRRWS, or (SEQ ID NO: 10) LKQKIKHVVKLKVVVKLRSQLVKRKQN.
9. The biomaterial of claim 1, wherein the ColBpep is selected from the group consisting of RRANAALKAGELYKSILYGC (SEQ ID NO: 11), SYIRIADTNIT (SEQ ID NO: 12), YSFYSDESLQ (SEQ ID NO: 13) and WYRGRL (SEQ ID NO: 14).
10. The biomaterial of claim 1, wherein the SABpep is selected from the group consisting of GGSPYGRC (SEQ ID NO: 15), and GGPQEQITQHGSPYGRC (SEQ ID NO: 16).
11. The biomaterial of claim 1, wherein the biocompatible polymer is hydrophilic.
12. The biomaterial of claim 11, wherein the biocompatible polymer is selected from the group consisting of: Poly(ethylene glycol), Poly(propylene glycol), Poly(methyl vinyl ether), Oligoethylene, Poly(isobutylene) Poly(tetrahydrofuran) Poly(oxytrimethylene), Poly(dimethylsiloxsane), Poly(dimethylsilane), Nylon 6, Nylon 11, Poly(acrylonitrile), Squalane, Poly(1,3-dioxolane), Poly(iminooligomethylene), Poly(l-lysine), Polyethyleneimine, Poly(adipate), Poly(l-caprolactone), Poly(L-lactic acid), or derivatives thereof.
13. The biomaterial of claim 11, wherein the one or more biocompatible polymers are mono, disubstituted, or multisubstituted with at least one functional group.
14. The biomaterial of claim 1, comprising SABpep linked to Poly(ethylene glycol) linked to HABpep (SABpep-PEG-HABpep).
15. The biomaterial of claim 1, comprising ColBpep linked to Poly(ethylene glycol) linked to HABpep (ColBpep-PEG-HABpep).
16. A biomaterial comprising at least one biologically compatible polymer having HA conjugated to a linker molecule covalently linked to the biologically compatible polymer, and one or more sialic acid binding peptides (SABPep) covalently linked to the biologically compatible polymer.
17. The biomaterial of claim 16, wherein HA is mixed into the biologically compatible polymer.
18. The biomaterial of claim 16, wherein HA is conjugated to a linker molecule and conjugated to a detectable moiety which is one or more sialic acid binding peptides (SABPep) covalently linked to the biologically compatible polymer.
19. The biomaterial of claim 18, wherein the detectable moiety is selected from the group consisting of: radioactive isotopes, magnetic beads, metallic beads, colloidal particles, fluorescent dyes, electron-dense reagents, enzymes, biotin, digoxigenin, or haptens.
20. The biomaterial of claim 19, wherein the detectable moiety is fluorescein.
21. A pharmaceutical composition comprising the biomaterial of claim 1, and a pharmaceutically acceptable carrier.
22. The pharmaceutical composition of claim 21, wherein the pharmaceutically acceptable carrier is suitable for ophthalmic use.
23. The pharmaceutical composition of either of claim 22, further comprising an additional therapeutic agent.
24. A method for treatment of a disease or condition of the eye of a subject in need of treatment, comprising administering to the subject an effective amount of the biomaterial composition of claim 1, or the pharmaceutical composition of claim 21.
25. The method of claim 24, wherein the disease or condition is Dry Eye.
26. A method for treating eye diseases by means of an eye surgery treatment in a subject in need of treatment comprising administering to the subject an effective amount of the biomaterial composition of claim 1 or the pharmaceutical composition of claim 21.
27. The method of claim 26, wherein the surgery treatment is selected from the group consisting of corneal transplantation, cataract surgery, glaucoma surgery, and surgery to repair retinal detachment.
28. The biomaterial of claim 3 wherein HA is mixed into the biologically compatible polymer.
29. The biomaterial of claim 3, further comprising HA bound to the HABPep.
30. The biomaterial of claim 29, wherein the HA has a molecular weight of between 5000 Da and 20,000,000 Da.
31. The biomaterial of claim 30, wherein the HA is crosslinked.
32. The biomaterial of claim 3, wherein the HABPep is a peptide comprising the following amino acid sequence: RRDDGAHWQFNALTVR (SEQ ID NO: 1), CRRDDGAHWQFNALTVR (SEQ ID NO: 2), GAAWQFNALTVR (SEQ ID NO: 3), GAHWQFAALTVR (SEQ ID NO: 4), GAHWQFNALTVA (SEQ ID NO: 5), GAHWQFNALTVR (SEQ ID NO: 6), STMMSRSHKTRSHHV (SEQ ID NO: 7), RYPISRPRKRC (SEQ ID NO: 8), TAGHGRRWS (SEQ ID NO: 9), or LKQKIKHVVKLKVVVKLRSQLVKRKQN (SEQ ID NO: 10).
33. The biomaterial of claim 3, wherein the ColBpep is selected from the group consisting of RRANAALKAGELYKSILYGC (SEQ ID NO: 11), SYIRIADTNIT (SEQ ID NO: 12), YSFYSDESLQ (SEQ ID NO: 13) and WYRGRL (SEQ ID NO: 14).
34. The biomaterial of claim 3, wherein the SABpep is selected from the group consisting of GGSPYGRC (SEQ ID NO: 15), and GGPQEQITQHGSPYGRC (SEQ ID NO: 16).
35. The biomaterial of claim 3, wherein the biocompatible polymer is hydrophilic.
36. The biomaterial of claim 35, wherein the biocompatible polymer is selected from the group consisting of: Poly(ethylene glycol), Poly(propylene glycol), Poly(methyl vinyl ether), Oligoethylene, Poly(isobutylene) Poly(tetrahydrofuran) Poly(oxytrimethylene), Poly(dimethylsiloxsane), Poly(dimethylsilane), Nylon 6, Nylon 11, Poly(acrylonitrile), Squalane, Poly(1,3-dioxolane), Poly(iminooligomethylene), Poly(l-lysine), Polyethyleneimine, Poly(adipate), Poly(l-caprolactone), Poly(L-lactic acid), or derivatives thereof.
37. The biomaterial of claim 35, wherein the one or more biocompatible polymers are mono, disubstituted, or multisubstituted with at least one functional group.
Description
REFERENCE TO RELATED APPLICATIONS
[0001] This application claims the benefit of International Patent Application No. 62/207,419, filed Aug. 20, 2015, which is hereby incorporated by reference for all purposes as if fully set forth herein.
INCORPORATION-BY-REFERENCE OF MATERIAL SUBMITTED ELECTRONICALLY
[0002] The instant application contains a Sequence Listing which has been submitted in ASCII format via EFS-Web and is hereby incorporated by reference in its entirety. Said ASCII copy, created on Aug. 12, 2016, is named P13662-02_ST25.txt and is 4,807 bytes in size.
BACKGROUND OF THE INVENTION
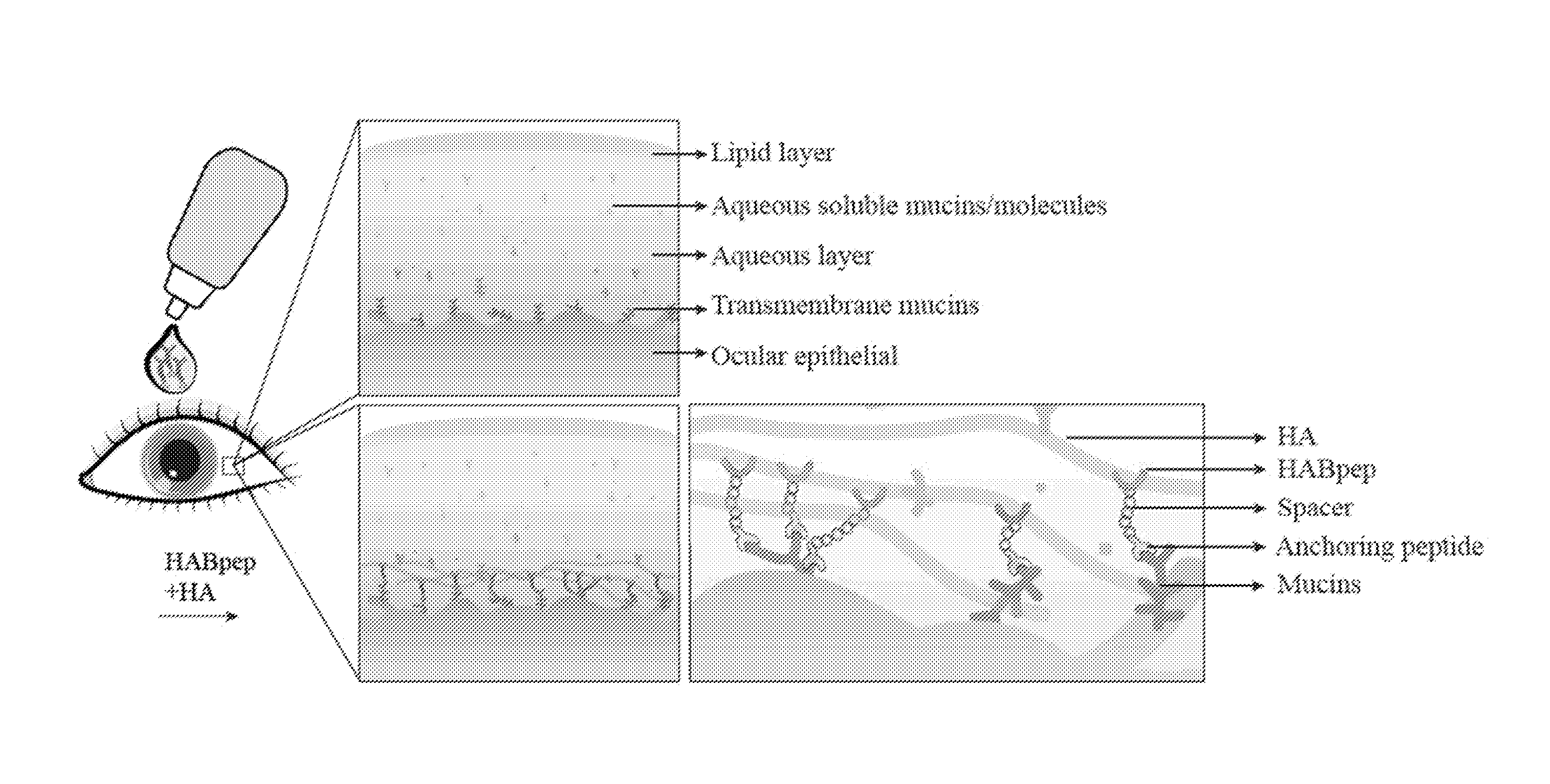
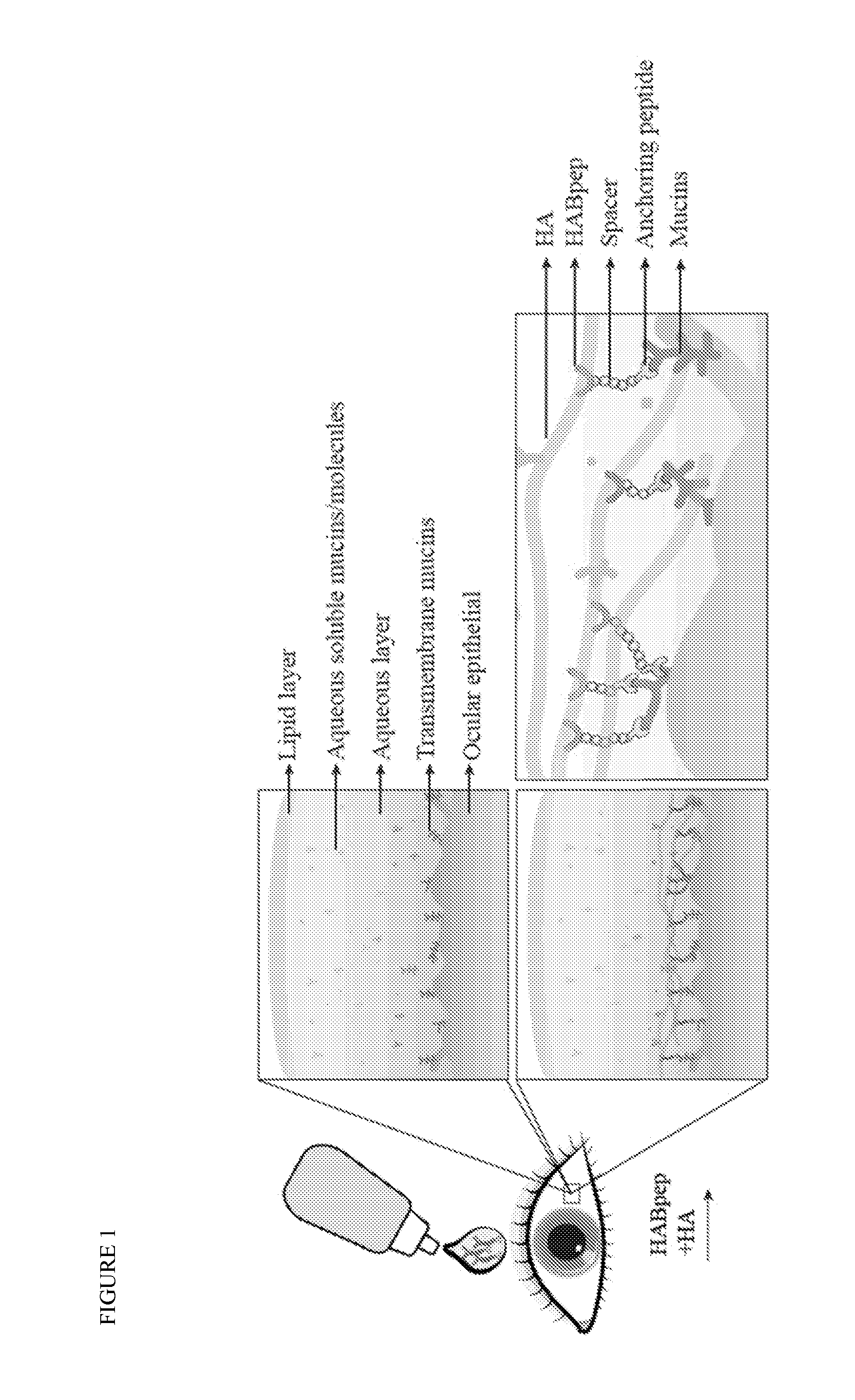
[0003] Dry eye is a prevalent ophthalmic condition especially to elderly population, affecting over 15% of the US population (Ding and Sullivan 2012). Symptoms include discomfort, dryness or lack of hydration, visual disturbances and pain in the eye, which can hinder the performance of everyday activities (Abetz, Rajagopalan et al. 2011; Pouyeh, Viteri et al. 2012; Pili, Kastelan et al. 2014). Although dry eye is a multifactorial disease, it occurs mostly due to any alterations in the tear film composition and stability with potential damage to the ocular surface. There are three tear film layers that have unique functions. All together, they prevent tear evaporation, protect the eye from foreign objects, lubricate and hydrate the ocular surface (Gipson 2007) (FIG. 1). Tear film-related dry eye disease is further categorized into two major conditions: aqueous-deficient and evaporative. Aqueous-deficient dry eye is caused by lacrimal gland's inability to produce sufficient tear fluid, whereas evaporative dry eye is caused by increased tear film evaporation rate due to a compromised protective lipid layer (Horwath-Winter, Berghold et al. 2003; Sharma and Hindman 2014). To treat dry eye disorders, many different approaches have been employed (Young, Veys et al. 2002; Johnson, Murphy et al. 2006; Ali and Byrne 2009; Papas, Ciolino et al. 2013); however, these strategies are temporary, involving the application of a saline or an emulsion-based eye drop to reduce discomfort and increase water retention at the ocular surface (Young, Veys et al. 2002; Johnson, Murphy et al. 2006; Prabhasawat, Tesavibul et al. 2007; Ali and Byrne 2009; Papas, Ciolino et al. 2013; Labetoulle 2015; Simmons, Carlisle-Wilcox et al. 2015). These artificial tears must be reapplied frequently owing to their short residence time in the eye.
[0004] Currently, commercially available eye drops containing 0.1-0.2% hyaluronic acid (HA) solutions, such as Opticalm.TM., Scope.TM., and Oxyal.TM. are commonly used as an active ingredient in Europe to relieve dry eye symptoms, while in USA as an inactive ingredient of the eye drop formulation (e.g. in Blink.TM.).
[0005] HA is an anionic glycosaminoglycan that is present in many tissues and organs as a major component of the extracellular matrix, has been shown to ameliorate dry eye because it retains water through hydrogen bonding (Hargittai and Hargittai 2008), stabilizes the tear film and lubricates the surface (Tonge, Jones et al. 2001; Read, Morgan et al. 2009). HA has also been shown to slow tear removal and permit uninterrupted blinking (Tsubota and Yamada 1992; Nakamura, Hikida et al. 1993; Hamano, Horimoto et al. 1996). In addition, HA has several desirable therapeutic properties; for example, it encourages corneal wound healing by promoting epithelial cell migration (Stuart and Linn 1985; Shimmura, Ono et al. 1995; Gomes, Amankwah et al. 2004; Johnson, Murphy et al. 2006; Rah 2011), reduces inflammation (Pauloin, Dutot et al.) and protects cells from free-radical damage (Presti and Scott). Recent studies (Bray, J. Theor. Biol, 2001; Georgiev et al., Soft Matter, 2013; Cerretani et al., Adv. Colloid Interface Sci. 2013) further indicate that polyanionic polysaccharide moieties, including HA can enhance the spreading of the tear film lipid layer, which may be an added clinical benefit of HA for ocular application. HA eye drop solutions effectively wet and lubricate the contact lens and ocular surfaces, however similar to saline, poly(vinyl alcohol)/PVA, methyl cellulose (MC) and hydroxypropyl methylcellulose (HPMC)-based eye drops, they suffer from low ocular residence time (<10 min) due to limited adherence to ocular surface (Snibson, Greaves et al. 1992; Mochizuki, Yamada et al. 2008; Papas, Ciolino et al. 2013). Conversely, highly concentrated HA-based eye drops are too viscous and interfere with vision and blinking (Marner, Mooller et al. 1996; Reddy, Grad et al. 2004; Ali and Byrne 2009; Papas, Tilia et al. 2014).
[0006] There still exists, however a need to develop an improved HA binding eye drop technology that prolongs HA retention and delivery on ocular surfaces providing biological and physical benefits of HA.
SUMMARY OF THE INVENTION
[0007] The present invention provides biomaterial compositions developed to enhance the availability of HA that can retain a thin film of moisture to mimic the smooth, hydrated and lubricated ocular surface.
[0008] In accordance with one or more embodiments, the present inventors have accomplished this by, in an embodiment, immobilizing HA through a HA binding peptide that localizes HA to the ocular surfaces. In accordance with another embodiment, the HA is immobilized through conjugation of the HA to a hydrophilic linker which is also conjugated to a binding peptide which binds transmembrane mucins of the epithelium that contain sialic acid and collagen of ocular matrices and act as anchoring sites for HA immobilization.
[0009] In accordance with a further embodiment, the present invention comprises, in an embodiment, an eye drop technology based on a heterobifunctional HA-binding peptide (HABpep) polymer-peptide system that binds and retains HA, from tear fluid or exogenous application, for longer periods of time at the surface of the eye. The key is to anchor HABpep polymer-peptide system on either sialic acid-containing glycosylated transmembrane molecules, such as mucins, the peripheral extracellular ocular epithelium protecting and lubricating surface, or the ocular tissue matrix enriched in collagen type I.
[0010] In some embodiments the HA can be provided within the biologically compatible polymer matrix, or added separately.
[0011] In accordance with an embodiment, the present invention provides a biomaterial comprising at least one biologically compatible polymer having one or more HA binding peptides (HABPep) covalently linked to the biologically compatible polymer, and one or more sialic acid binding peptides (SABPep) covalently linked to the biologically compatible polymer.
[0012] In accordance with another embodiment, the present invention provides a biomaterial comprising at least one biologically compatible polymer having one or more HA binding peptides (HABPep) covalently linked to the biologically compatible polymer, and one or more collagen binding peptides (ColBPep) covalently linked to the biologically compatible polymer.
[0013] In accordance with a further embodiment, the present invention provides a biomaterial comprising at least one biologically compatible polymer having one or more HA binding peptides (HABPep) covalently linked to the biologically compatible polymer, and one or more sialic acid binding peptides (SABPep) and one or more collagen binding peptides (ColBPep) covalently linked to the biologically compatible polymer.
[0014] In accordance with an embodiment, the present invention provides a biomaterial comprising SABpep linked to Poly(ethylene glycol) linked to HABpep (SABpep-PEG-HABpep).
[0015] In accordance with another embodiment, the present invention provides a biomaterial comprising ColBpep linked to Poly(ethylene glycol) linked to HABpep (ColBpep-PEG-HABpep).
[0016] In accordance with an embodiment, the present invention provides a biomaterial comprising HA covalently linked to one or more sialic acid binding peptides (SABPep) via a linker.
[0017] In accordance with another embodiment, the present invention provides a biomaterial comprising HA covalently linked to a detectable moiety which is covalently linked to one or more sialic acid binding peptides (SABPep) (for example, with a fluorescent moiety (FL), HA-FL-SABPep) via a linker.
[0018] In accordance with an embodiment, the present invention provides a biomaterial a pharmaceutical composition comprising the biomaterial as described above, and pharmaceutically acceptable carrier.
[0019] In accordance with another embodiment, the present invention provides a biomaterial a pharmaceutical composition comprising the biomaterial as described above, an additional therapeutic agent, and pharmaceutically acceptable carrier.
[0020] In accordance with an embodiment, the present invention provides a method of treatment of an ophthalmic disease or condition of the eye comprising administering to the eye of a subject an effective amount of the biomaterial described above or a pharmaceutical composition comprising the biomaterial.
BRIEF DESCRIPTION OF THE DRAWINGS
[0021] FIGS. 1A-1B: Schematic of an HA binding eye drop technology. 1A) Exterior ocular surface has three layers: lipid, aqueous and mucin layers. 1B) On application of HA binding eye drop, HABpep binds HA through an anchoring peptide on the ocular surface. Specifically, sialic acid present in extracellular domains, such as transmembrane mucins is targeted (magnified); alternatively, collagen of the ocular tissue present in conjunctiva or cornea tissues can also be targeted to immobilize HA.
[0022] FIGS. 2A-2C: Selecting ocular surface binding peptides: targeting sialic acid and collagen as sites for binding SABpep and ColBpep on the conjunctival tissue ex vivo. 2A) A HABA assay quantifying the amount of peptide bound to conjunctiva tissue (normalized to the dry weight) indicated that out of the six peptides tested (as listed in Table 1), peptide 3 (ColBpep) and peptide 6 (ColBpep) bound the most to conjunctiva tissue compared to control (p<0.5). 2B) Immunohistochemistry showed that both SABpep and anti-mucin 1 antibody stained positively for the transmembrane mucin-1 on the epithelial surface, while ColBpep binds on ocular matrices throughout cellular and non-cellular regions (scale bar=100 .mu.m). 2C) A magnified immunostaining image showing that SABpep specifically binds to the cellular periphery throughout the epithelium (scale bar=100 .mu.m).
[0023] FIGS. 3A-3D QCM-D measurements of in vitro binding of SABpep and HABpep to sialic acid and HA, respectively. QCM-D curves indicate successful deposition of substrates and binding peptides: 3A) Sialic acid-containing mucin adsorbed onto gold substrate followed by attachment of SABpep. 3B) Thiolated HA is covalently immobilized onto pristine gold, subsequent HABpep biding to HA. 3C) Deposition of the substrate layers of mucin and HA on pristine gold QCM-D surfaces quantified by the visco-elastic Voigt model, respectively. 3D) Attachment of binding peptides to mucin and HA substrates quantified by Sauerbrey relationship (overtone n=5). Conditions: All concentrations cw=0.1 mg/ml, T=37.degree. C., flow rate=25 .mu.L/min in PBS buffer (pH=7.4) (n=3).
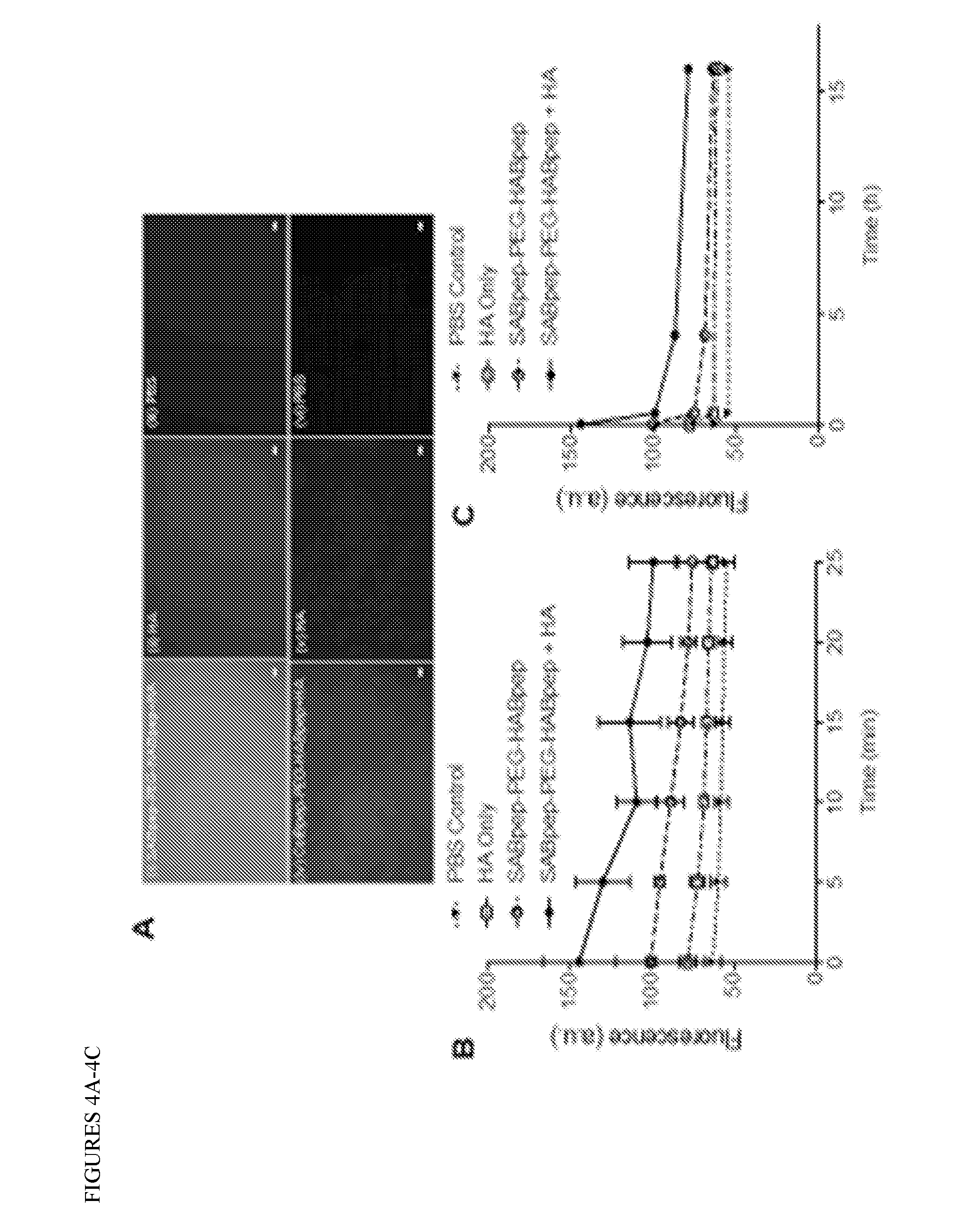
[0024] FIGS. 4A-4C: HA binding through HABpep and overtime HA release profile. SABpep-PEG-HABpep treated eyes SABpep-PEG-HABpep and ColBpep-PEG-HABpep bind HA ex vivo. 4A) SABpep-PEG-HABpep treated rabbit eyes (corneas were dissected and imaged after treatments) (i) showed intense fluorescence compared to the eyes treated with only HA (ii) and PBS (iii). Similarly, ColBpep-PEG-HABpep treated rabbit eyes (iv) showed a higher fluorescence compared to the eyes treated with only HA (v) and PBS (vi). Scale bar 100 .mu.m. 4B) Short-time HA release profile for SABpep-PEG-HABpep (-/+HA) treated eyes showed a higher amount of HA was releasing at each time point compared to HA without the peptide. SABpep-PEG-HABpep treated eyes bind to .about.1.8 times more HA in the beginning as indicated by the greater initial fluorescence value and retain HA longer, showing signal significantly greater (.about.x1.6) than the control after 25 minutes. 4C) Long-term HA release profile for SABpep-PEG-HABpep (+/-HA) treated eyes showed that compared to the controls, a higher amount of HA was bound to the ocular surface even after 15 h.
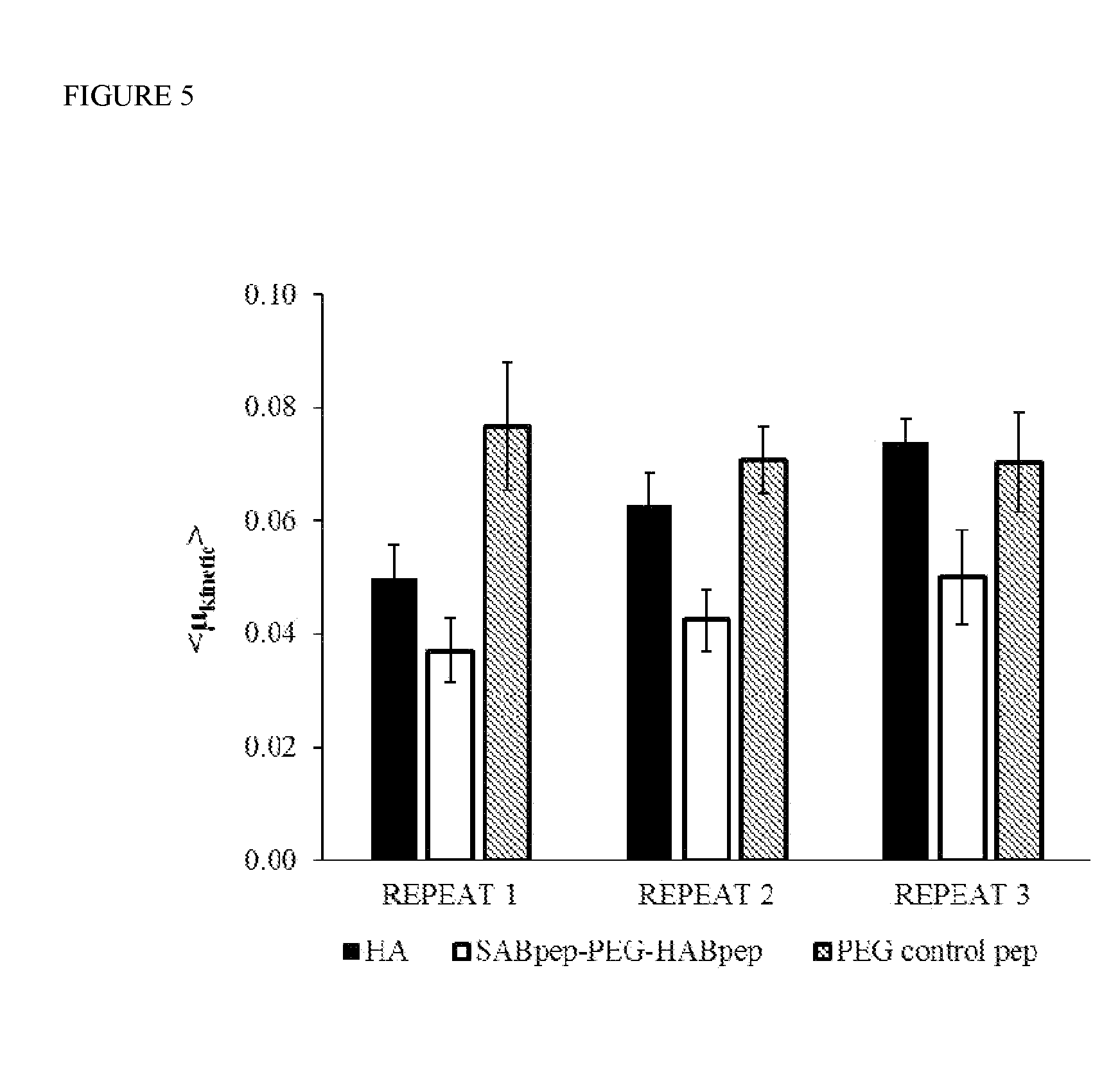
[0025] FIG. 5: Friction measurement of rabbit ocular tissues treated with HA-only compared with tissues treated with SABpep-PEG-HABpep then HA and a non-HA-binding PEG control peptide. Kinetic friction values (mean.+-.SEM) for three sequential times (repeat 1, 2, 3) at the rabbit eyelid-cornea biointerface in a PBS bath showed enhanced lubrication for tissues treated with peptide and HA compared to HA-only (Cohen's d effect size, d=1.25, 2.13, and 2.10) and the PEG control peptide (d=2.02, 2.70 and 1.34) as indicated by the strong effect sizes in all three repeats in both controls, respectively.
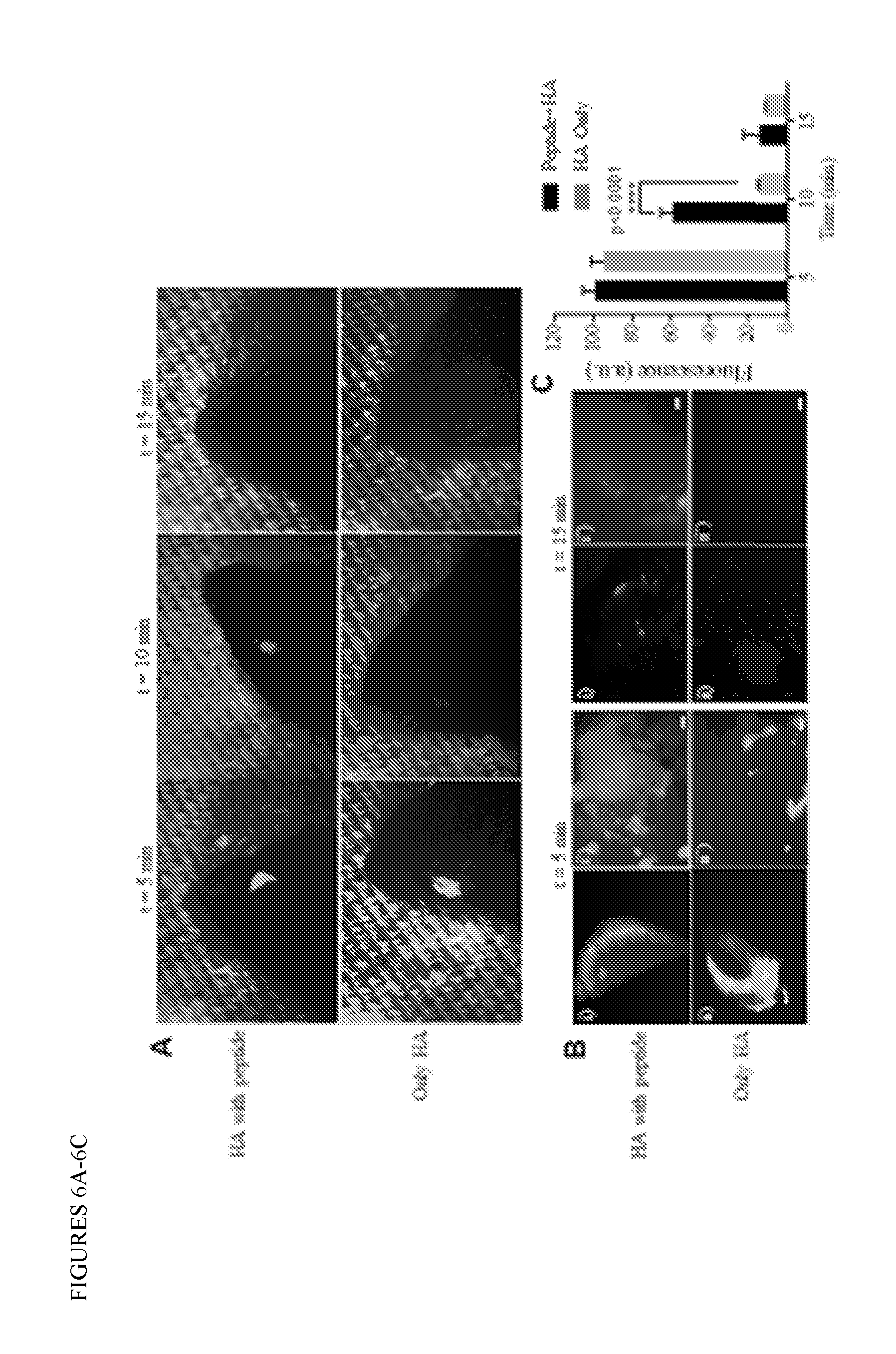
[0026] FIGS. 6A-6B: HA retention on in vivo mice eyes. 6A) Harvested mice eyes applied with SABpep-PEG-HABpep+HA eye drops (top row) retained HA for at least three fold time period longer than those applied with control eye drops (only HA, bottom row) (n=2). Even after 15 minutes, HA was present on the samples treated with the binding peptide. 6B) Magnified images showed that both at beginning and after 15 min, SABpep-PEG-HABpep+HA (i- only eye, i'-magnified image of i) was bound homogenously to the ocular epithelial cells, while samples treated with only HA showed relatively less homogenous distribution. (Scale bar=100 .mu.m)
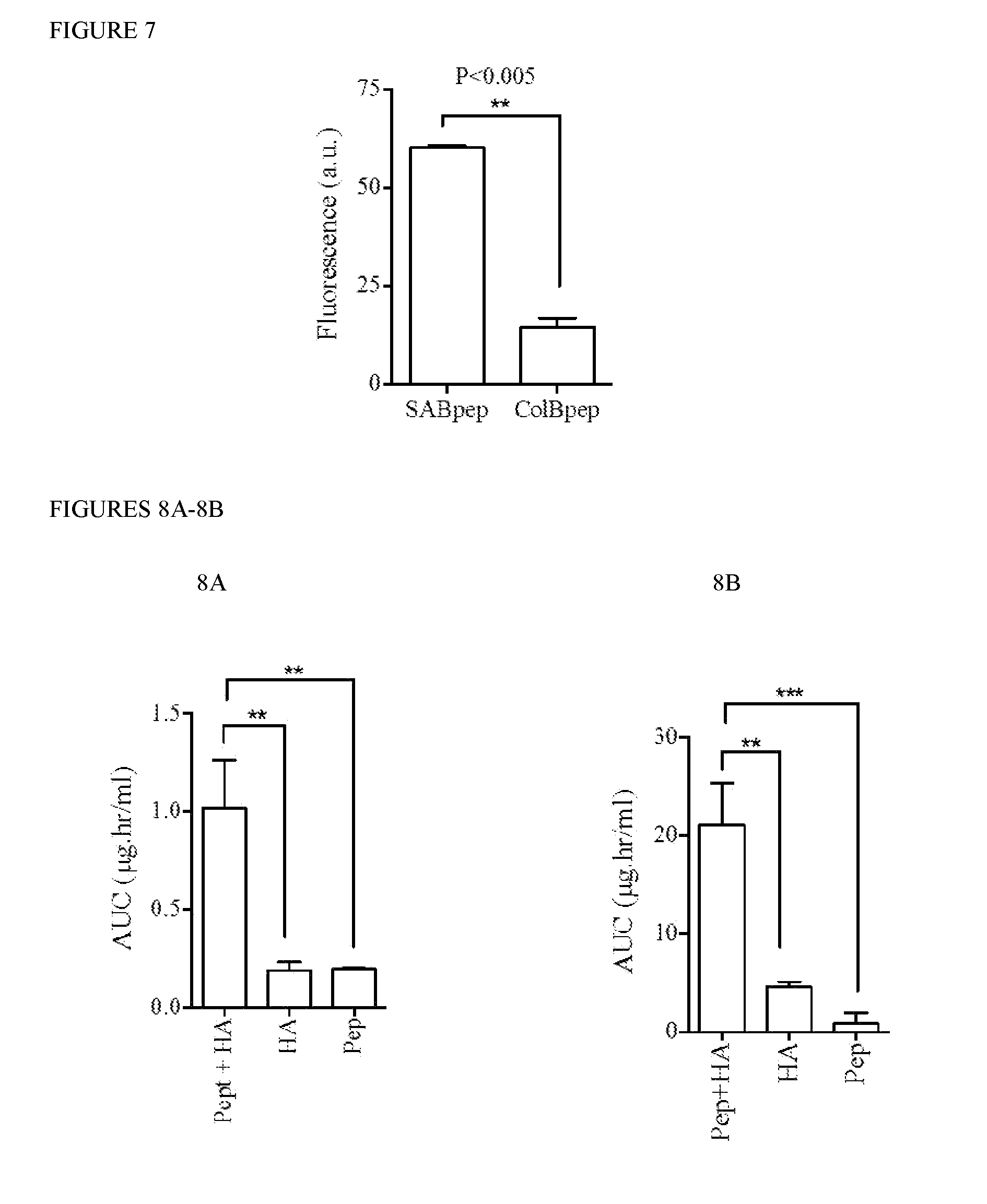
[0027] FIG. 7: HA binding on ocular surfaces through SABpep and ColBpep. Images from FIG. 4A were analyzed using ImageJ to quantify the relative amount of fluorescence between SABpep+HA and ColBpep+HA treated samples. Background signal from the PBS controls were subtracted from the images.
[0028] FIGS. 8A-8B: Estimated HA release over time by area under curve calculation (AUC). Area under curve (AUC) was calculated for each experimental group. The background from PBS control was then subtracted from each AUC to quantify total amount of HA retained for each experimental condition. A standard curve of fluorescence versus HA was used to convert AUC values into HA (.mu.g). AUC for release time of A) 25 minutes, and B) 16 h.
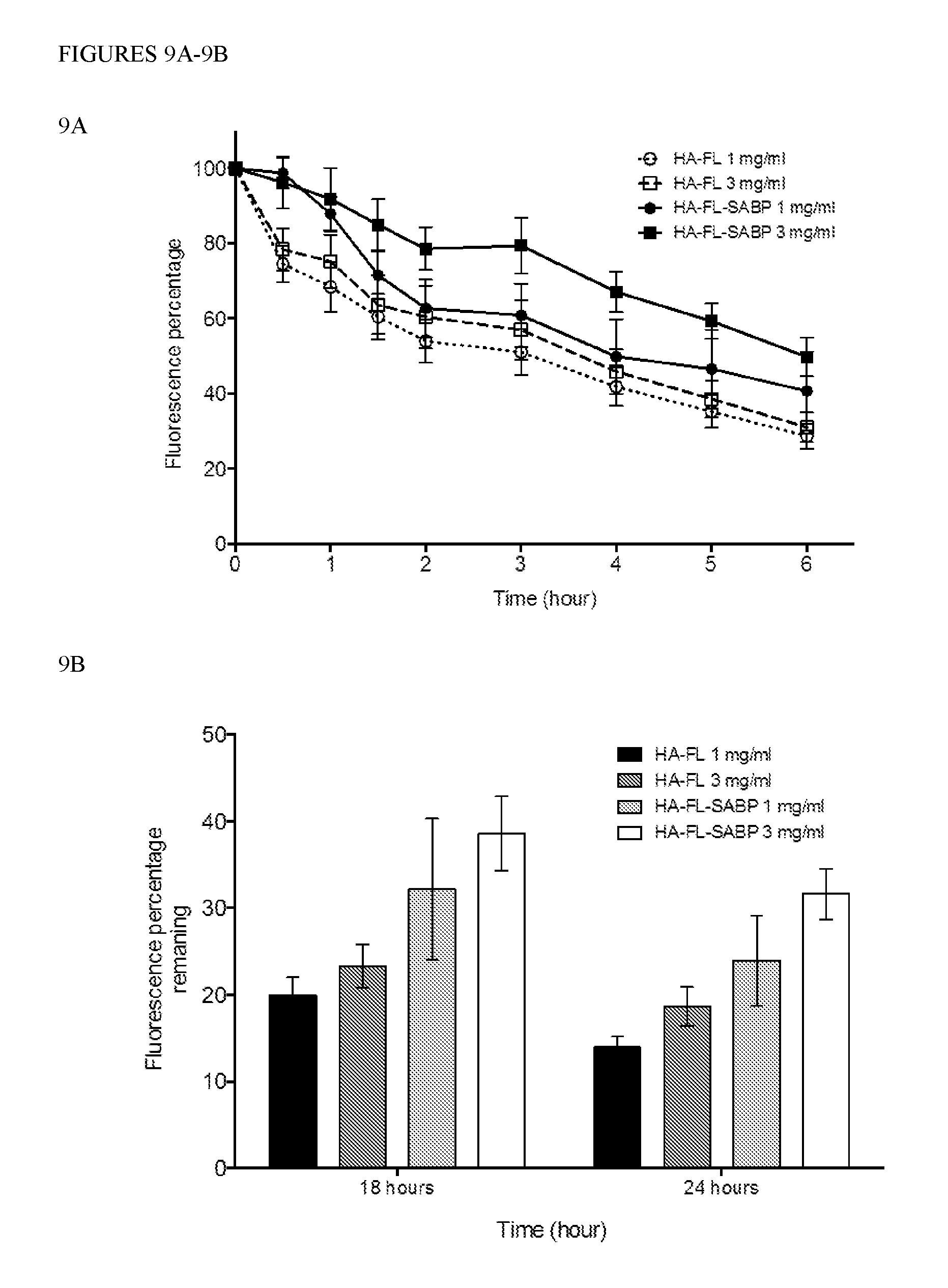
[0029] FIGS. 9A-9B: HA binding through covalently conjugated SABpep and overtime bound HA profile ex vivo. 9A) Overtime bound HA profile for tissues treated with SABpep-conjugated HA-FL showed a higher amount of retained HA at each time point compared to HA without the peptide. It is more obvious in samples treated with 3.0 mg/mL or 0.3% w/v HA-FL, probably due to the higher overall amount of SABpep that bound to the tissue. At beginning, tissues treated with SABpep conjugated to HA-FL showed similar fluorescence values as for HA-FL with no peptide. This is due to the same amount of FITC concentration in starting solutions. However, the rate of drop of fluorescence (%) was slower in samples treated with HA-SABpep compared to no peptide. As expected, the rates of drop in fluorescence values remain similar for HA-FL at concentrations of 1 mg/mL and 3 mg/mL. The initial drop in fluorescence rate after 1st wash (within 30 min) is .about.24% vs .about.4% while after 2nd wash (within 60 min) is -28% vs 8% for tissues treated with HA-FL but no peptide vs SABpep conjugated HA-FL. After 6 h, fluorescence values for HA-FL-SABpep treated tissue samples were .about.1.7 times more than that of tissues treated with HA-FL but no peptide. 9B) Similarly, bound HA for SABpep-HA treated eyes showed that compared to the control, a higher amount of HA (.about.1.7 times for 3.0 mg/mL solution) was still bound to the ocular surface even at 24 h time point.
DETAILED DESCRIPTION OF THE INVENTION
[0030] A current challenge in HA-based eye drop technology is finding a way for HA to remain on the ocular surface for a prolonged time. Increased HA retention can benefit dry eye patients by reducing symptoms of dry eye and by increasing corneal wettability and reducing tear evaporation. Recently, an in vitro study demonstrated that HA is superior to two other common lubricants, carboxymethylcellulose (CMC) and hydroxypropyl methylcelluolose (HPMC) tested for its water retention ability and its protective effectiveness of human corneal epithelial cells against dehydration (Cornea 2013; 32:1260-4). Moreover, HA is demonstrated to be beneficial for both, patients with aqueous tear- and lipid-deficient dry eye because it increases tear volume and tear film stability (Br J Ophthalmol 2007; 91:47-50). The present inventors have been able to concentrate and retain HA using the inventive compositions and methods in order to provide many physical and biological benefits to parts of the ocular surfaces, such as cornea and conjunctiva.
[0031] The goal of the present invention is to enhance the binding of HA to the eye surface, which would retain a thin film of moisture to mimic the smooth, hydrated and lubricated ocular surface. The present invention utilizes a HA binding peptide (HABpep) (Tolg, Hamilton et al. 2012; Amemiya, Nakatani et al. 2005) which can localize HA to the ocular surface through anchoring sites on the epithelium. Sialic acid is usually found at the outermost of the glycan chains on the cell surface proteins and lipids (Varki 2008), and it is widely distributed through different types of tissues, including ocular epithelium. Membrane-bound mucins (e.g. mucin-1, mucin-16 and mucin-20) known as the glycocalyx contain sialic acid in 0-glycosylated extracellular domains and are present in the peripheral extracellular epithelium layer of the ocular tissues (FIG. 1). The inventors chose sialic acid as the potential anchoring site for immobilizing HA on the ocular tissue surface. Moreover, in case of severe dry eye, sight-threatening complications, such as persistent corneal epithelial defect or sterile stromal ulceration, allow the stromal tissue to be exposed to the external environment (Pflugfelder 2004). At the same time, the ocular surface epithelium is damaged and membrane-bound mucins in the glycocalyx decrease significantly (Corrales, Narayanan et al. 2011). Therefore, in an alternative embodiment, the present inventors also utilize collagen as an alternative anchoring site, as stromal collagen will be exposed when the epithelium is absent.
[0032] In accordance with some embodiments, the present inventors developed an eye drop composition comprising a HABpep that noncovalently binds HA. The inventors have also developed a chemical spacer or linker with HABpep to facilitate interactions of HABpep with HA. In some embodiments the linker is poly(ethylene glycol) (PEG). The other end of the PEG linker is modified by a peptide that can bind to tissues or proteins on the ocular surface. A number of candidate peptides that were capable of binding to the ocular surface through different mechanisms were screened to determine which ocular binding-peptides were the most effective (Table 1).
TABLE-US-00001 TABLE 1 Peptides Screened Peptide Sequence (#1-6) Description Reference K-K(Palm)-K-K-K-K(Palm) Cell membrane binding (Matsuda, Hatanaka et al. (SEQ ID NO: 18) 2014) GWQPPRARI Epithelial binding- (Mooradian, McCarthy et al. (SEQ ID NO: 19) Fibronectin derived 1993) GGSPYGR Sialic acid binding (Heerze, Chong et al. 1992) (SEQ ID NO: 15) GGPQEQITQHGSPYGRC Sialic acid binding (Heerze, Smith et al. 1995) (SEQ ID NO: 20) RRANAALKAGELYKSILYGC Collagen I binding peptide (Paderi, Sistiabudi et al.) (SEQ ID NO: 11) derived from platelet rich plasma SYIRIADTNIT Collagen I binding peptide (Kalamajski, Aspberg et al. (SEQ ID NO: 12) derived from decorin 2007)
[0033] Out of these peptides, the inventors found that SABpep GGSPYGRC (SEQ ID NO: 15) and ColBpep SYIRIADTNIT (SEQ ID NO: 12) were the most promising candidates (FIG. 2A). SABpep is especially promising, because it can directly target epithelial cells, which consist of sialic-acid rich Mucin-1. Transmembrane mucins are the first layer on the epithelial layer that can be targeted to localize HA, therefore, we investigated SABpep for HA binding in depth.
[0034] The inventors later confirmed by immunochemistry and fluorescent cell imaging that SABpep-biotin binds selectively to epithelial lining of conjunctiva while ColBpep-biotin binds to any collagen I on the tissue (FIG. 2A). This selectivity can be of importance when treating dry eye since the disease can be caused by abnormality of any of the tear film's three layers (lipid, aqueous, and mucin). Therefore, different ocular surface-binding peptides can be used for different dry eye causes. Damage to a certain region of the eye, such as where epithelium is removed, can be selectively treated for by using ColBpep-PEG-HABpep, while in conditions where epithelium is still partially or fully present can be treated with SABpep.
[0035] The polymer-peptide system with collagen-binding peptide is most useful for treating conditions related to severe dry eye that can lead to blindness, such as Sjogren syndrome, in which the stromal collagen is exposed as the epithelium is missing (Pflugfelder 2004). In conditions where epithelium is still partially or fully present (Gipson 2004), the eyes can be treated with the polymer-peptide system containing SABpep. The binding sites of SABpep was specifically localized on the cell membrane throughout the ocular epithelium (FIG. 2C). It is well known that transmembrane mucins, such as mucin-1, mucin-4 and mucin-16 are localized primarily within apical cell membranes of the stratified corneal and conjunctival epithelia, while transmembrane mucin-20 is uniquely distributed throughout the conjunctival and corneal epithelia (Gipson 2014, Woodard 2014). Since, mucins are rich in sialic acid, SABpep should bind to these molecules.
[0036] In accordance with an embodiment, the present invention provides novel biomaterial compositions which bind to tissue surfaces, such as the eye, and bind HA. The embodiments of the present invention provide biomaterials that allow tissue modification with HA binding polymer coatings, and utilize a synthetic peptide to target and locally concentrate hyaluronic acid to those tissue surfaces.
[0037] The HA-binding biomaterials of the present invention can be powerful tools for effective modification of the lubrication environment of the eye and for other tissues where lubrication or the presence of HA is critical for homeostasis and health.
[0038] In accordance with an embodiment, the present invention provides a biomaterial comprising at least one biologically compatible polymer having one or more HA binding peptides (HABPep) covalently linked to the biologically compatible polymer, and one or more sialic acid binding peptides (SABPep) covalently linked to the biologically compatible polymer.
[0039] In accordance with another embodiment, the present invention provides a biomaterial comprising at least one biologically compatible polymer having one or more HA binding peptides (HABPep) covalently linked to the biologically compatible polymer, and one or more collagen binding peptides (ColBPep) covalently linked to the biologically compatible polymer.
[0040] In accordance with a further embodiment the present invention provides a biomaterial comprising at least one biologically compatible polymer having one or more HA binding peptides (HABPep) covalently linked to the biologically compatible polymer, and one or more sialic acid binding peptides (SABPep) and one or more collagen binding peptides (ColBPep) covalently linked to the biologically compatible polymer.
[0041] In accordance with an embodiment, the present invention provides a biomaterial comprising SABpep linked to Poly(ethylene glycol) linked to HABpep (SABpep-PEG-HABpep).
[0042] In accordance with another embodiment, the present invention provides a biomaterial comprising ColBpep linked to Poly(ethylene glycol) linked to HABpep (ColBpep-PEG-HABpep).
[0043] In accordance with an embodiment, the present invention provides a biomaterial comprising HA covalently linked to one or more sialic acid binding peptides (SABPep) via a linker.
[0044] In accordance with another embodiment, the present invention provides a biomaterial comprising HA covalently linked to a detectable moiety (such as a fluorescent dye FL) and to one or more sialic acid binding peptides (SABPep) (HA-FL-SABPep) via a linker.
[0045] The HA is linked to the polymer and/or the detectable moiety by a linker molecule. For instance linking groups having alkyl, aryl, combination of alkyl and aryl, or alkyl and aryl groups having heteroatoms may be present. For example, the linker can be a C.sub.1-C.sub.20 alkyl, C.sub.2-C.sub.20 alkenyl, C.sub.2-C.sub.20 alkynyl, C.sub.1-C.sub.20 hydroxyalkyl, C.sub.1-C.sub.20 alkoxy, C.sub.1-C.sub.20 alkoxy C.sub.1-C.sub.20 alkyl, C.sub.1-C.sub.20 alkylamino, di-C.sub.1-C.sub.20 alkylamino, C.sub.1-C.sub.20 dialkylamino C.sub.1-C.sub.20 alkyl, C.sub.1-C.sub.20 thioalkyl, C.sub.2-C.sub.20 thioalkenyl, C.sub.2-C.sub.20 thioalkynyl, C.sub.6-C.sub.22 acyloxy, C.sub.6-C.sub.22 arylamino C.sub.2-C.sub.20 acyloxy, C.sub.2-C.sub.20 thioacyl, C.sub.1-C.sub.20 amido, and C.sub.1-C.sub.20 sulphonamido.
[0046] In some embodiments the HA can be provided within the biologically compatible polymer matrix, or added separately.
[0047] As used herein, the term "biocompatible biomaterial" means materials that can be used for binding to tissues, and which are acceptable for use in a mammal, preferably in a human subject.
[0048] A biologically compatible polymer refers to a polymer which is functionalized to serve as a composition for applying to a biological surface. The polymer is one that is a naturally occurring polymer or one that is not toxic to the host. The polymer can, e.g., contain at least an imide. The polymer may be a homopolymer where all monomers are the same or a hetereopolymer containing two or more kinds of monomers. The terms "biocompatible polymer," "biocompatible cross-linked polymer matrix" and "biocompatibility" when used in relation to the instant polymers are art-recognized are considered equivalent to one another, including to biologically compatible polymer. For example, biocompatible polymers include polymers that are neither toxic to the host (e.g., an animal or human), nor degrade (if the polymer degrades at a rate that produces monomeric or oligomeric subunits or other byproducts at toxic concentrations in the host).
[0049] As used herein, the term "crosslinked" refers to a composition containing intermolecular links and, optionally, intramolecular links, arising from the formation of covalent bonds. Covalent bonding between two crosslinkable components may be direct, in which case, an atom in one component is directly bound to an atom in the other component, or it may be indirect, that is, for example, through a linking group. A crosslinked gel or polymer matrix may, in addition to covalent bonds, also include intermolecular and/or intramolecular noncovalent bonds such as hydrogen bonds and electrostatic (ionic) bonds.
[0050] By "detectable label(s) or moieties" is meant a composition that when linked to a molecule of interest renders the latter detectable, via spectroscopic, photochemical, biochemical, immunochemical, or chemical means. For example, useful labels include radioactive isotopes, magnetic beads, metallic beads, colloidal particles, fluorescent dyes, electron-dense reagents, enzymes (for example, as commonly used in an ELISA), biotin, digoxigenin, or haptens. Specific radioactive labels include most common commercially available isotopes including, for example, .sup.3H, .sup.11C, .sup.13C, .sup.15N, .sup.18F, .sup.19F, .sup.123I, .sup.124I, .sup.125I, .sup.131I, .sup.86Y, .sup.89Zr, .sup.111In, .sup.94mTc, .sup.99mTc, .sup.64Cu and .sup.68Ga. Suitable dyes include any commercially available dyes such as, for example, 5(6)-carboxyfluorescein, IRDye 680RD maleimide or IRDye 800CW, ruthenium polypyridyl dyes, and the like.
[0051] Gel refers to a state of matter between liquid and solid, and is generally defined as a polymer network swollen in a liquid medium. Typically, a gel is a two-phase colloidal dispersion containing both solid and liquid, wherein the amount of solid is greater than that in the two-phase colloidal dispersion referred to as a "sol." As such, a "gel" has some of the properties of a liquid (i.e., the shape is resilient and deformable) and some of the properties of a solid (i.e., the shape is discrete enough to maintain three dimensions on a two-dimensional surface). "Gelation time," also referred to herein as "gel time," refers to the time it takes for a composition to become non-flowable under modest stress. This is generally exhibited as reaching a physical state in which the elastic modulus, G', equals or exceeds the viscous modulus, G'', i.e., when tan(A) becomes 1 (as may be determined using conventional rheological techniques).
[0052] The term "polymer" is used to refer to molecules composed of repeating monomer units, including homopolymers, block copolymers, heteropolymers, random copolymers, graft copolymers and so on. "Polymers" also include linear polymers as well as branched polymers, with branched polymers including highly branched, dendritic, and star polymers.
[0053] In accordance with a further embodiment, the biocompatible polymer can be hydrophilic.
[0054] A hydrogel is a water-swellable polymeric matrix that can absorb water to form elastic gels. Hydrogels consist of hydrophilic polymers crosslinked to from a water-swollen, insoluble polymer network. Crosslinking can be initiated by many physical or chemical mechanisms, for example, such as, a light-induced reaction.
[0055] A "matrix" is a three-dimensional network of macromolecules held together by covalent or noncovalent crosslinks. On placement in an aqueous environment, dry hydrogels swell to the extent allowed by the viscosity, the gel state and/or degree of crosslinking in the polymer or network. A matrix can be a network.
[0056] Hydrogels are semi-interpenetrating networks that promote cell, tissue and organ repair, and in some instances, discourage scar formation. Hydrogels can be derivatized to contain a reactive group to facilitate polymerization and linking. Hydrogels can also carry a reactive group or a functional group reactive with a biological surface, an artificial surface and/or a second polymer or network. The latter form of reactivity also can anchor a gel of interest at and to a site of interest. Hydrogels of interest also are configured to have a viscosity that will enable the gelled hydrogel to remain or reside in place for longer periods of time. Viscosity can be controlled by the monomers and polymers used, the degree of crosslinking, by the level of water trapped in the hydrogel and by incorporated thickeners, such as biopolymers, such as proteins, lipids, saccharides and the like. An example of such a hydrogel is HA, whether crosslinked or not.
[0057] The term "functionalized" as used herein, refers to a modification of an existing molecular segment to generate or introduce a new reactive or more reactive group (e.g., an amine, ester or imide group) that is capable of undergoing reaction with another molecule, polymer or functional group (e.g., an amine, an ester or a carboxyl group) to form a covalent bond. For example, carboxylic acid groups can be functionalized by reaction with a carbodiimide and an imide reagent using known procedures to provide a new reactive functional group in the form of an imide group substituting for the hydrogen in the hydroxyl group of the carboxyl function.
[0058] The terms "substituted," "functional group" and "reactive group" are contemplated to include all permissible substituents of organic compounds on the monomers, polymers and networks of interest. In a broad aspect, the permissible substituents include acyclic and cyclic, branched and unbranched, carbocyclic and heterocyclic, aromatic and nonaromatic substituents of organic compounds. Illustrative substituents include, for example, carboxy groups, amine groups, amide groups, hydroxyl groups and so on, as known in the art. The permissible substituents may be one or more and the same or different for appropriate organic compounds.
[0059] A functional group or a moiety capable of mediating formation of a polymer or network can be added to a naturally occurring molecule or a synthetic molecule practicing methods known in the art. Functional groups include the various radicals and chemical entities taught herein, and include alkenyl moieties such as acrylates, methacrylates, dimethacrylates, oligoacrylates, oligomethacrylates, ethacrylates, itaconates or acrylamides.
[0060] Further functional groups include aldehydes. Other functional groups may include ethylenically unsaturated monomers including, for example, alkyl esters of acrylic or methacrylic acid such as methyl methacrylate, ethyl methacrylate, butyl methacrylate, ethyl acrylate, butyl acrylate, hexyl acrylate, n-octyl acrylate, lauryl methacrylate, 2-ethylhexyl methacrylate, nonyl acrylate, benzyl methacrylate, the hydroxyalkyl esters of the same acids such as 2-hydroxyethyl acrylate, 2-hydroxyethyl methacrylate, and 2-hydroxypropyl methacrylate, the nitrite and amides of the same acids such as acrylonitrile, methacrylonitrile, and methacrylamide, vinyl acetate, vinyl propionate, vinylidene chloride, vinyl chloride, and vinyl aromatic compounds such as styrene, t-butyl styrene and vinyl toluene, dialkyl maleates, dialkyl itaconates, dialkyl methylene-malonates, isoprene and butadiene. Suitable ethylenically unsaturated monomers containing carboxylic acid groups include acrylic monomers such as acrylic acid, methacrylic acid, ethacrylic acid, itaconic acid, maleic acid, fumaric acid, monoalkyl itaconate including monomethyl itaconate, monoethyl itaconate, and monobutyl itaconate, monoalkyl maleate including monomethyl maleate, monoethyl maleate, and monobutyl maleate, citraconic acid and styrene carboxylic acid. Suitable polyethylenically unsaturated monomers include butadiene, isoprene, allylmethacrylate, diacrylates of alkyl dials such as butanediol diacrylate and hexanediol diacrylate, divinyl benzene and the like.
[0061] In some embodiments, other suitable hydrophilic polymers which can serve as the biocompatible polymer include synthetic polymers such as poly(ethylene glycol), poly(ethylene oxide), partially or fully hydrolyzed poly(vinyl alcohol), poly(vinylpyrrolidone), poly(ethyloxazoline), poly(ethylene oxide)-co-poly(propylene oxide) block copolymers (poloxamers and meroxapols), poloxamines, carboxymethyl cellulose, and hydroxyalkylated celluloses such as hydroxyethyl cellulose and methylhydroxypropyl cellulose, and natural polymers, such as, polysaccharides or carbohydrates such as Ficoll.TM. polysucrose, dextran, heparan sulfate, chondroitin sulfate or alginate, and polypeptides or proteins such as gelatin, collagen, albumin or ovalbumin, or copolymers or blends thereof.
[0062] In accordance with still another embodiment, the biocompatible polymers are selected from the group consisting of: Poly(ethylene glycol), Poly(propylene glycol), Poly(methyl vinyl ether), Oligoethylene, Poly(isobutylene) Poly(tetrahydrofuran) Poly(oxytrimethylene), Poly(dimethylsiloxsane), Poly(dimethylsilane), Nylon 6, Nylon 11, Poly(acrylonitrile), Squalane, Poly(1,3-dioxolane), Poly(iminooligomethylene), Poly(1-lysine), Polyethyleneimine, Poly(adipate), Poly(1-caprolactone), Poly(L-lactic acid), or derivatives thereof.
[0063] By way of example, and not limitation, and in particular embodiments, the polymer can comprise synthetic reactants and comprises poly(ethylene glycol) (PEG) or a derivative thereof.
[0064] Polysaccharides that are very viscous liquids or that are thixotropic, and form a gel over time by the slow evolution of structure, are also useful. For example, HA, which can form an injectable gel with a consistency like a hair gel, may be utilized. Modified hyaluronic acid can also be useful. As used herein, the term "modified hyaluronic acids" refers to chemically modified hyaluronic acids. Modified hyaluronic acids may be designed and synthesized with preselected chemical modifications to adjust the rate and degree of linking and biodegradation. For example, modified hyaluronic acids may be designed and synthesized to be esterified with a relatively hydrophobic group such as propionic acid or benzylic acid to render the polymer more hydrophobic and gel-forming, or which are grafted with amines to promote electrostatic self-assembly. Modified hyaluronic acids thus, may be synthesized which are injectable, to flow under stress, but maintain a gel-like structure when not under stress. Hyaluronic acid and hyaluronic derivatives are available from Genzyme, Cambridge, Mass. and Fidia, Italy. Other commercially available HA useful in the invention are Restylane, comprising a crosslinked HA, and Juvederm (Allergan) comprising HA.
[0065] HA is a polymer of disaccharides, themselves composed of D-glucuronic acid and D-N-acetylglucosamine, linked via alternating (3-1,4 and (3-1,3 glycosidic bonds. HA can have 25,000 disaccharide repeats in length. Polymers of HA can range in size from 5,000 to 20,000,000 Da in vivo.
[0066] In accordance with one or more embodiments, the HA that can be used with the inventive compositions and methods can have molecular weights in the range of 5 kDa to about 20.times.10.sup.4 kDa, in some embodiments, HA can have a molecular weight of between about 10 kDa to about 2.times.10.sup.4 kDa.
[0067] In accordance with one or more embodiments, the HA that can be used with the inventive compositions and methods can be crosslinked.
[0068] Photopolymerization is a method to covalently crosslink polymer chains, whereby a photoinitiator and polymer solution (termed "pre-gel" or monomer solution) are exposed to a light source specific to the photoinitiator. On activation, the photoinitiator reacts with specific functional groups in the polymer chains, linking the functional groups to form the hydrogel. The reaction generally is rapid (3-5 minutes) and can proceed at room or body temperature. Photoinduced gelation enables spatial and temporal control of gel formation, permitting shape manipulation after injection and during gelation in vivo. Cells and bioactive factors can be incorporated into the hydrogel scaffold by simply mixing same in and with the polymer solution prior to gelation
[0069] In accordance with an embodiment, the HABPep is a peptide which is capable of specifically binding HA. Many such HA binding peptides are known in the art. See for example WO/2006/130974, which describes many such peptides which have at least one repetition of the ammo acid residue sequence B.sub.rX.sub.7-B.sub.2 where B is any basic amino acid residue and X.sub.7 are any 7 non-acidic amino acid residues. The binding of the peptide to HA may be enhanced by the addition of basic ammo acid residues between B1 and B2 or flanking either end of motif (non-conservative substitutions).
[0070] The HABP52 family of HA binding peptides includes peptides with an amino acid sequence selected from the group consisting of i) (RRDDGAHWQFNALTVR) (SEQ ID NO: 1) or (CRRDDGAHWQFNALTVR) (SEQ ID NO: 2) or a conservative amino acid substitution thereof at a residue position other than 4, 5, 6, 9, 10 or 11, ii) GAAWQFNALTVR, (SEQ ID NO: 3) or a conservative amino acid substitution thereof at a residue position other than 4, 5, 6, 9, 10 or 11, iii) GAHWQFAALTVR, (SEQ ID NO: 4) or a conservative amino acid substitution thereof at a residue position other than 4, 5, 6, 9, 10 or 11, and iv) GAHWQFNALTVA (SEQ ID NO: 5) or a conservative amino acid substitution thereof at a residue position other than 4, 5, 6, 9, 10 or 11.
[0071] In accordance with some embodiments the HABpep comprises the peptide GAHWQFNALTVR (SEQ ID NO: 6) or a conservative amino acid substitution thereof at a residue position other than 4, 5, 6, 9, 10 or 11.
[0072] In accordance with some embodiments the HABpep comprises the peptide STMMSRSHKTRSHHV (SEQ ID NO: 7); or RYPISRPRKRC (SEQ ID NO: 8); or TAGHGRRWS (SEQ ID NO: 9); or LKQKIKHVVKLKVVVKLRSQLVKRKQN (SEQ ID NO: 10) or a peptide derivative thereof with conservative amino acid substitutions.
[0073] As used herein, the term "collagen binding peptide (ColBPep)" means a protein, peptide or fragment which is capable of specifically binding extracellular matrix proteins, such as collagen I or collagen II. In accordance with one or more embodiments, the ColBpep can comprise a peptide having the following sequences: RRANAALKAGELYKSILYGC (SEQ ID NO: 11), SYIRIADTNIT (SEQ ID NO: 12), YSFYSDESLQ (SEQ ID NO: 13) and WYRGRL (SEQ ID NO: 14) or a peptide derivative thereof with conservative amino acid substitutions.
[0074] As used herein, the term "sialic acid binding peptide (SABPep)" means a protein, peptide or fragment which is capable of specifically binding a glycoprotein having sialic acid sugar moieties. In accordance with one or more embodiments, the SABpep can comprise a peptide having the following sequences: GGSPYGRC (SEQ ID NO: 15), and GGPQEQITQHGSPYGRC (SEQ ID NO: 16) or a peptide derivative thereof with conservative amino acid substitutions.
[0075] It will be understood by those of ordinary skill in the art that any known conjugation method which can be used to attach both peptides, HABpep, ColBpep or SABpep to the PEG spacer or linker, or any biocompatible spacer through functional reactive groups can be used in the compositions and methods of the present invention.
[0076] In accordance with an embodiment, the present invention provides pharmaceutical compositions comprising the biomaterials described herein, and a pharmaceutically acceptable carrier.
[0077] Therapeutic formulations of the product may be prepared for storage as lyophilized formulations or aqueous solutions by mixing the biomaterials described herein, having the desired degree of purity with optional pharmaceutically acceptable carriers, diluents, excipients or stabilizers typically employed in the art, i.e., buffering agents, stabilizing agents, preservatives, isotonifiers, non-ionic detergents, antioxidants and other miscellaneous additives, see Remington's Pharmaceutical Sciences, 16th ed., Osol, ed. (1980). Such additives are generally nontoxic to the recipients at the dosages and concentrations employed, hence, the excipients, diluents, carriers and so on are pharmaceutically acceptable.
[0078] The compositions can take the form of solutions, suspensions, emulsions, powders, sustained-release formulations, depots and the like. Examples of suitable carriers are described in "Remington's Pharmaceutical Sciences," Martin. Such compositions will contain an effective amount of the biopolymer of interest, preferably in purified form, together with a suitable amount of carrier so as to provide the form for proper administration to the patient. As known in the art, the formulation will be constructed to suit the mode of administration.
[0079] Buffering agents help to maintain the pH in the range which approximates physiological conditions. Buffers are preferably present at a concentration ranging from about 2 mM to about 50 mM. Suitable buffering agents for use with the instant invention include both organic and inorganic acids, and salts thereof, such as citrate buffers (e.g., monosodium citrate-disodium citrate mixture, citric acid-trisodium citrate mixture, citric acid-monosodium citrate mixture etc.), succinate buffers (e.g., succinic acid monosodium succinate mixture, succinic acid-sodium hydroxide mixture, succinic acid-disodium succinate mixture etc.), tartrate buffers (e.g., tartaric acid-sodium tartrate mixture, tartaric acid-potassium tartrate mixture, tartaric acid-sodium hydroxide mixture etc.), fumarate buffers (e.g., fumaric acid-monosodium fumarate mixture, fumaric acid-disodium fumarate mixture, monosodium fumarate-disodium fumarate mixture etc.), gluconate buffers (e.g., gluconic acid-sodium glyconate mixture, gluconic acid-sodium hydroxide mixture, gluconic acid-potassium gluconate mixture etc.), oxalate buffers (e.g., oxalic acid-sodium oxalate mixture, oxalic acid-sodium hydroxide mixture, oxalic acid-potassium oxalate mixture etc.), lactate buffers (e.g., lactic acid-sodium lactate mixture, lactic acid-sodium hydroxide mixture, lactic acid-potassium lactate mixture etc.) and acetate buffers (e.g., acetic acid-sodium acetate mixture, acetic acid-sodium hydroxide mixture etc.). Phosphate buffers, carbonate buffers, histidine buffers, trimethylamine salts, such as Tris, HEPES and other such known buffers can be used.
[0080] Preservatives may be added to retard microbial growth, and may be added in amounts ranging from 0.2%-1% (w/v). Suitable preservatives for use with the present invention include phenol, benzyl alcohol, m-cresol, octadecyldimethylbenzyl ammonium chloride, benzyaconium halides (e.g., chloride, bromide and iodide), hexamethonium chloride, alkyl parabens, such as, methyl or propyl paraben, catechol, resorcinol, cyclohexanol and 3-pentanol.
[0081] Isotonicifiers are present to ensure physiological isotonicity of liquid compositions of the instant invention and include polhydric sugar alcohols, preferably trihydric or higher sugar alcohols, such as glycerin, erythritol, arabitol, xylitol, sorbitol and mannitol. Polyhydric alcohols can be present in an amount of between about 0.1% to about 25%, by weight, preferably 1% to 5% taking into account the relative amounts of the other ingredients.
[0082] Stabilizers refer to a broad category of excipients which can range in function from a bulking agent to an additive which solubilizes the therapeutic agent or helps to prevent denaturation or adherence to the container wall. Typical stabilizers can be polyhydric sugar alcohols; amino acids, such as arginine, lysine, glycine, glutamine, asparagine, histidine, alanine, ornithine, L-leucine, 2-phenylalanine, glutamic acid, threonine etc.; organic sugars or sugar alcohols, such as lactose, trehalose, stachyose, arabitol, erythritol, mannitol, sorbitol, xylitol, ribitol, myoinisitol, galactitol, glycerol and the like, including cyclitols such as inositol; polyethylene glycol; amino acid polymers; sulfur containing reducing agents, such as urea, glutathione, thioctic acid, sodium thioglycolate, thioglycerol, a-monothioglycerol and sodium thiosulfate; low molecular weight polypeptides (i.e., <10 residues); proteins, such as human serum albumin, bovine serum albumin, gelatin or immunoglobulins; hydrophilic polymers, such as polyvinylpyrrolidone, saccharides, monosaccharides, such as xylose, mannose, fructose or glucose; disaccharides, such as lactose, maltose and sucrose; trisaccharides, such as raffinose; polysaccharides, such as, dextran and so on. Stabilizers can be present in the range from 0.1 to 10,000 w/w per part of biopolymer.
[0083] Additional miscellaneous excipients include bulking agents, (e.g., starch), chelating agents (e.g., EDTA), antioxidants (e.g., ascorbic acid, methionine or vitamin E) and cosolvents.
[0084] Non-ionic surfactants or detergents (also known as "wetting agents") may be added to help solubilize the therapeutic agent, as well as to protect the therapeutic protein against agitation-induced aggregation, which also permits the formulation to be exposed to shear surface stresses without causing denaturation of the protein. Suitable non-ionic surfactants include polysorbates (20, 80 etc.), polyoxamers (184, 188 etc.), Pluronic.RTM. polyols and polyoxyethylene sorbitan monoethers (TWEEN-20.RTM., TWEEN-80.RTM. etc.). Non-ionic surfactants may be present in a range of about 0.05 mg/ml to about 1.0 mg/ml, preferably about 0.07 mg/ml to about 0.2 mg/ml.
[0085] The present invention provides liquid formulations of a biopolymer having a pH ranging from about 5.0 to about 7.0, or about 5.5 to about 6.5, or about 5.8 to about 6.2, or about 6.0, or about 6.0 to about 7.5, or about 6.5 to about 7.0.
[0086] The instant invention encompasses formulations, such as, liquid formulations having stability at temperatures found in a commercial refrigerator and freezer found in the office of a physician or laboratory, such as from about 20.degree. C. to about 5.degree. C., said stability assessed, for example, by microscopic analysis, for storage purposes, such as for about 60 days, for about 120 days, for about 180 days, for about a year, for about 2 years or more. The liquid formulations of the present invention also exhibit stability, as assessed, for example, by particle analysis, at room temperatures, for at least a few hours, such as one hour, two hours or about three hours prior to use.
[0087] Examples of diluents include a phosphate buffered saline, buffer for buffering against gastric acid in the bladder, such as citrate buffer (pH 7.4) containing sucrose, bicarbonate buffer (pH 7.4) alone, or bicarbonate buffer (pH 7.4) containing ascorbic acid, lactose, or aspartame. Examples of carriers include proteins, e.g., as found in skim milk, sugars, e.g., sucrose, or polyvinylpyrrolidone. Typically these carriers would be used at a concentration of about 0.1-90% (w/v) but preferably at a range of 1-10%.
[0088] The formulations to be used for in vivo administration must be sterile. That can be accomplished, for example, by filtration through sterile filtration membranes. For example, the formulations of the present invention may be sterilized by filtration.
[0089] The biomaterial compositions of the present invention will be formulated, dosed and administered in a manner consistent with good medical practice. Factors for consideration in this context include the particular disorder being treated, the particular mammal being treated, the clinical condition of the individual patient, the cause of the disorder, the site of delivery of the agent, the method of administration, the scheduling of administration, and other factors known to medical practitioners. The "therapeutically effective amount" of the biomaterial to be administered will be governed by such considerations, and can be the minimum amount necessary to prevent, ameliorate or treat a disorder of interest. As used herein, the term "effective amount" is an equivalent phrase refers to the amount of a therapy (e.g., a prophylactic or therapeutic agent), which is sufficient to reduce the severity and/or duration of a disease, ameliorate one or more symptoms thereof, prevent the advancement of a disease or cause regression of a disease, or which is sufficient to result in the prevention of the development, recurrence, onset, or progression of a disease or one or more symptoms thereof, or enhance or improve the prophylactic and/or therapeutic effect(s) of another therapy (e.g., another therapeutic agent) useful for treating a disease.
[0090] Generally, the ingredients are supplied either separately or mixed together in unit dosage form, for example, as a dry lyophilized powder or water-free concentrate in a sealed container, such as an ampule or sachet indicating the quantity of active agent. Where the composition is to be administered by ophthalmic solution, it can be dispensed with a bottle containing sterile pharmaceutical grade water or saline.
[0091] An "active agent" and a "biologically active agent" are phrases used interchangeably herein to refer a chemical or biological compound that induces a desired pharmacological or physiological effect, wherein the effect may be prophylactic or therapeutic. The terms also encompass pharmaceutically acceptable, pharmacologically active derivatives of those active agents specifically mentioned herein, including, but not limited to, salts, esters, amides, prodrugs, active metabolites, analogs and the like.
[0092] When the terms "active agent," "pharmacologically active agent" and "drug" are used, it is to be understood that the invention includes the active agent per se, as well as pharmaceutically acceptable, pharmacologically active salts, esters, amides, prodrugs, metabolites, analogs etc. The active agent can be a biological entity, such as a virus or cell, whether naturally occurring or manipulated, such as transformed.
[0093] In accordance with an embodiment, the present invention provides the use of the biomaterial compositions disclosed herein, for treating eye diseases by means of an eye surgery treatment in a subject, characterized in that an effective amount of the biomaterial composition is administered to the tissue of the subject in need of treatment.
[0094] In accordance with an embodiment, the present invention provides a therapeutic method for the treatment of eye diseases by means of an eye surgery treatment, comprising applying to the eye of a subject in need of such treatment a therapeutically effective amount of the biomaterial compositions described herein. Such surgical procedures include, but are not limited to, corneal transplantation, cataract surgery, glaucoma surgery, and surgery to repair retinal detachment.
[0095] In accordance with another embodiment, the present invention provides a therapeutic method for the treatment of dry eye or keratoconjunctivitis sicca (KCS) which can be the result of a number of disorders, including, for example, Sjogren's syndrome. The inventive methods comprise applying an effective amount of the biomaterial compositions of the present invention on the cornea of the eye, and which may include other therapeutic agents, such as estrogens, or cyclosporine.
[0096] In accordance with an embodiment, the present invention provides the use of the biomaterial compositions disclosed herein, for treating dry eye in a subject, characterized in that an effective amount of the biomaterial composition is applied to the eye of a subject in need of such treatment.
[0097] In accordance with an embodiment, the present invention provides the use of a biomaterial comprising HA covalently linked to a detectable moiety (such as, for example, a fluorescent dye FL) and to one or more sialic acid binding peptides (SABPep) (HA-FL-SABPep) via a linker for diagnosis or confirmation or grading the severity of dry eye or keratoconjunctivitis sicca (KCS) in a subject, comprising administering to the eye of the subject suspected of having dry eye or KCS an effective amount of the biomaterial and detecting the amount of binding of the biomaterial to the cornea of the eye. If the amount of binding of the composition is less than the amount of binding in a control eye, the subject is diagnosed or confirmed as having dry eye or KCS, or in the case of comparing the binding amount to the amounts in cases of known severity, the severity of the disease can be graded or assessed.
[0098] One of ordinary skill in the art would understand that the type of detectable moiety can be other than a fluorescent moiety, including for example, colloidal particles, fluorescent dyes, electron-dense reagents, enzymes, and dyes such as, for example, 5(6)-carboxyfluorescein, IRDye 680RD maleimide or IRDye 800CW. It will be understood that the uses provided herein can also include administration of at least one additional therapeutic agent with the compositions disclosed herein.
[0099] Examples of biologically active agents include, without limitation, enzymes, receptor antagonists or agonists, hormones, growth factors, antibiotics, antimicrobial agents, and antibodies. The term "biologically active agent" is also intended to encompass various cell types and genes that can be incorporated into the compositions of the invention.
[0100] In certain embodiments, the subject compositions comprise about 1% to about 75% or more by weight of the total composition, alternatively about 2.5%, 5%, 10%, 20%, 30%, 40%, 50%, 60% or 70%, of a biologically active agent.
[0101] Non-limiting examples of biologically active agents include following: adrenergic blocking agents, androgenic steroids, anti-allergenic materials, anti-cholinergics and sympathomimetics, anti-infective agents, anti-inflammatory agents such as steroids, non-steroidal anti-inflammatory agents, anti-pyretic and analgesic agents, antihistamines, biologicals, decongestants, estrogens, ion exchange resins, growth factors, neuromuscular drugs, nutritional substances, peripheral vasodilators, progestational agents, prostaglandins, vitamins, antigenic materials, and prodrugs.
[0102] In accordance with one or more embodiments, there is provided ophthalmic formulations comprising the inventive biomaterials, wherein the formulation is suitable for administration to the eye of a subject. The ophthalmic formulation may have a pH between 5.5 and 7. In some embodiments the ophthalmic formulation is an aqueous formulation. In some embodiments the ophthalmic formulation is in the form of a single dose unit. In some embodiments the ophthalmic formulation does not comprise a preservative. The ophthalmic formulation may further comprise one or more additional therapeutic agents, such as antioxidants. The ophthalmic formulation may further comprise one or more tear substitutes. In some embodiments, at least one of the tear substitutes contains an ophthalmic lubricant (e.g., hydroxypropylmethylcellulose).
[0103] A variety of tear substitutes are known in the art and include, but are not limited to: monomeric polyols, such as, glycerol, propylene glycol, and ethylene glycol; polymeric polyols such as polyethylene glycol; cellulose esters such hydroxypropylmethyl cellulose, carboxy methylcellulose sodium and hydroxy propylcellulose; dextrans such as dextran 70; water soluble proteins such as gelatin; vinyl polymers, such as polyvinyl alcohol, polyvinylpyrrolidone, and povidone; and carbomers, such as carbomer 934P, carbomer 941, carbomer 940 and carbomer 974P. Many such tear substitutes are commercially available, which include, but are not limited to cellulose esters such as Bion Tears.RTM., Celluvisc.RTM., Genteal.RTM., OccuCoat.RTM., Refresh.RTM., Teargen II.RTM., Tears Naturale.RTM., Tears Natural II.RTM., Tears Naturale Free.RTM., and TheraTears.RTM.; and polyvinyl alcohols such as Akwa Tears.RTM., HypoTears.RTM., Moisture Eyes.RTM., Murine Lubricating.RTM., and Visine Tears.RTM.. Tear substitutes may also be comprised of paraffins, such as the commercially available Lacri-Lube.RTM. ointments. Other commercially available ointments that are used as tear substitutes include Lubrifresh PM.RTM., Moisture Eyes PM.RTM. and Refresh PM.RTM.. Preservatives and other additives may also be present such as, for example, antimicrobials, antioxidants, chelating agents, and inert gases and the like.
[0104] It will be understood by those of ordinary skill that a dosing regimen used in the inventive compositions and methods can be any length of time sufficient to provide enhanced HA concentration in the eyes of the subject. The term "chronic" as used herein, means that the length of time of the dosage regimen can be hours, days, weeks, months, or possibly years. In accordance with an embodiment, the composition is administered for at least 48 hours.
[0105] Typically, an attending physician will decide the dosage of the composition with which to treat each individual subject, taking into consideration a variety of factors, such as age, body weight, general health, diet, sex, compound to be administered, route of administration, and the severity of the condition being treated. By way of example, and not intending to limit the invention, the dose of the compositions of the present invention can be at a concentration from about 0.1 mg/ml of biomaterial composition to about 100 mg/ml, preferably from about 1 mg/ml to about 10 mg/ml. In some embodiments, solutions comprising HA can have HA in concentrations of between about 0.01% to 1% by weight, preferably between about 0.1% to about 0.5% by weight of solution.
[0106] In accordance with an embodiment, the present invention provides a method for making the biomaterial composition as described herein, comprising: a) obtaining a sufficient amount of one or more biocompatible polymers conjugated to at least one or more N-succinimide groups and one or more maleimide groups in a suitable solution; b) adding to the solution of a) a sufficient amount of one or more ColBPep or SABpep and allowing it to react with the one or more N-succinimide groups to produce one or more biocompatible polymers having one or more ColBPep or SABpep which are covalently linked to the biocompatible polymers; c) obtaining a sufficient amount of having one or more thiolated HA binding peptides (C-HABPep) in a suitable solution; d) adding the solution of b) to the solution of c) and mixing for a sufficient period of time to produce one or more biocompatible polymers having one or more HA binding peptides (HABPep) which are covalently linked to the biocompatible polymers which are covalently linked to one or more ColBPep or SABpep; e) adding to the solution of d) a sufficient amount of hyaluronic acid (HA) in a suitable solvent for a sufficient time to allow HA to bind to the HABPep in the solution; f) removing the unreacted reagents of b), c) and e) and purifying the remaining product.
[0107] It will be understood by those of ordinary skill in the art that any biocompatible polymer can be used in the inventive methods. In a preferred embodiment, the biocompatible polymer is poly(ethylene glycol).
[0108] In accordance with an embodiment, the present invention provides a bifunctional biopolymer composition comprising a biologically compatible polymer having at least one amine reactive moiety and at least one thiol reactive moiety.
[0109] The term "bifunctional biopolymer composition" means a biocompatible polymer which has been chemically modified to have at least one amine reactive moiety and at least one thiol reactive moiety covalently linked to the polymer either directly or via a linking moiety.
[0110] In one or more embodiments, the amine reactive moiety can include N-hydroxysuccinimide or N-hydroxysulfosuccinimide. Other bifunctional biopolymer compositions can include, for example, maleimide-PEG-N-hydroxysuccinimide; iodoacetamide-PEG-hydroxysuccinimide/sulfsuccinimide; and acrylate-PEG-N-hydroxysuccinimide. Other molecules such as azlactones, imidoesters, epoxides, fluorophenyl ester, anhydride, caronate, acyl azide, isothiocyanate, isocyanate, aldehyde, etc. can also be used.
[0111] In one or more embodiments, the thiol reactive moiety can include maleimide or iodoacetamide.
[0112] One of ordinary skill in the art will understand that the chemical modifications to the biopolymers to incorporate the amine reactive moieties and thiol reactive moieties are known in the art and can be accomplished using known methods.
[0113] In some embodiments, the biomaterial composition described herein can be administered to the site in the subject along with HA in a one-step process. In other embodiments the biomaterial composition described herein can be administered to the site either before or after administering HA to the site.
[0114] Therefore, in an embodiment, the inventive SABpep-PEG-HABpep and/or ColBpep-PEG-HABpep compositions can be applied to existing eye drop technology by simply mixing the composition to a standard HA-eye drop solution. The advantages of this technology are that it is easy to prepare and does not require any modifications to standard HA eye drop solution--the peptide construct can simply be added to the HA-eye drop solution. The practical implication is that HABpep technology may overcome frequent HA-containing eye drop instillation and problems associated with dry eyes by localizing HA on ocular surface that retains water for prolonged time and enhances wettability.
EXAMPLES
[0115] Materials
[0116] Six different binding peptide candidates that could potentially bind ocular tissues were custom synthesized (NeoBioLab, MA, USA) (Table 1) (36-41). The peptides were biotinylated with a linker, N-hydroxysuccinimde (NHS)-PEG-biotin (Life Technologies, NY) with a standard method as recommended by the manufacturer's protocol. HA, HA-biotin and HA-fluorescein (HA-FL) (each with Mw 800 kDa) with a degree of substitution .about.5 mol %, were purchased from Creative PEGWorks (Chapel Hill, N.C.). Heterofunctional poly(ethylene glycol) (PEG, Mw 1000 Da) with SABpep (GGSPYGRC) (SEQ ID NO: 15) and HABpep (STMMSRSHKTRSHHVGC) (SEQ ID NO: 17) (Amemiya, Nakatani et al. 2005; Tolg, Hamilton et al. 2012) were custom synthesized from SynPeptide (China).
[0117] Screening of ocular surface-binding peptides.
[0118] Conjunctival tissue from the ocular surface was targeted to test the binding affinity of the multiple peptides. Conjunctiva was harvested from rabbit eyes (Pel-Freez Biologicals, Rogers, Ariz.) with the underlying tenon's capsule removed, and then cut into 6 mm pieces with a biopsy punch. The conjunctival epithelium was kept intact for testing SABpep, while the epithelium was scrapped off with a cell scraper for testing type I collagen-binding peptide (ColBpep). The tissue pieces were merged in the biotin-conjugated peptide solutions (1.0 mg/mL in PBS, pH 7.4; Life Technologies, NY) for 20 minutes and then washed three times with PBS to remove any unbound peptides. A HABA (4'-hydroxyazobenzene-2-carboxylic acid) assay (Life Technologies, NY) was used to measure the amount of bound peptide (biotin), per manufacturer's instructions. An aqueous solution of biotin (1.0 mg/mL) without peptides was used as a negative control. Peptides with the highest binding to conjunctiva corresponding to their fluorescence values were chosen for further experiments.
[0119] Immunohistochemistry.
[0120] To visualize binding of SABpep and ColBpep to the ocular surface, we performed immunohistochemistry with biotin-conjugated peptides on tissue section of rabbit conjunctiva. Conjunctival tissue dissected from rabbit eyes were fixed with 4% paraformaldehyde (PFA) solution (Electron Microscopy Sciences, Hatfield, Pa.), embedded and sectioned (thickness-5 .mu.m) in paraffin. The sections were first incubated with biotin-conjugated peptides (1.0 mg/ml in PBS pH 7.4) for 1 h at room temperature, washed thrice with PBS and then stained with streptavidin-Texas Red conjugate (Vector Laboratories, CA) in accordance to manufacturer's protocol. The samples were then washed again three times, mounted with DAPI mounting medium (Vector Laboratories) and imaged with Zeiss Axio Imager 2 microscope (Carl Zeiss, Jena, Germany). Sections stained with only secondary antibody served as negative control. Mouse anti-mucin-1 primary antibody (Novus Biologicals, Littleton, Colo.) and mouse anti-collagen type I antibody (Abcam, Cambridge, Mass.) were used as replacements to SABpep and ColBpep in the positive controls, respectively, and stained with goat anti-mouse IgG secondary antibody (Life Technologies, NY).
[0121] Quartz Crystal microbalance with dissipation (QCM-D) binding studies.
[0122] QCM-D studies were conducted using a Qsense E4 system and gold-coated sensor crystals QSX 301 (Biolin Scientific, Linthicum Heights, Md.) to directly evaluate the binding polymer-peptide constructs of SABpep and HABpep to mucin and HA, respectively. At a flow rate of 25 .mu.l/min in PBS (pH=7.4), mucin from the bovine submaxillary glands (type I-S, Sigma-Aldrich, St. Louis, Mo.) or HA-thiol (Creative PEGWorks, Chapel Hill, N.C.) were applied on pristine surfaces at cw=0.1 mg/mL in PBS at 37.degree. C. After a PBS rinse, either SABpep (0.1 mg/mL) or SABPep-PEG-HABpep (Mw 3698.43 Da, 0.1 mg/mL) was applied to the respective substrates. Changes of frequency (.DELTA.f) and dissipation (.DELTA.D) were recorded, and the mass per surface area (.DELTA.m) for immobilized SABpep and HABpep was calculated using the Sauerbrey relationship on the fifth overtone (.DELTA.m/C=-.DELTA.f, C=17.7 ng/cm.sup.2) and for HA-thiol and Mucin substrates using the visco-elastic Voigt model (Reviakine, Johannsmann et al. 2011; Majd, Kuijer et al. 2014).
[0123] Ex vivo imaging for studying HA binding.
[0124] SABpep-PEG-HABpep and ColBpep-PEG-HABpep (1.0 mg/mL) solutions were applied on the corneal surface of the rabbit eyes for 20 minutes. The epithelium was scrapped off with a cell scraper for testing ColBpep-PEG-HABpep. After washing twice with PBS, 10 .mu.g/ml of HA-FL was applied to the same area for 20 minutes. Positive controls received HA-FL with no peptides and negative controls were treated with only PBS. For imaging, the eyes were washed again with PBS and corneas were excised and imaged under Axio Imager 2 microscope (Carl Zeiss). ImageJ was used to process and quantify the amount fluorescence in each image. We further performed overtime HA release studies on SABpep-PEG-HABpep treated samples by measuring fluorescence values with full area scan periodically every 5 minutes for a total of 25 minutes and also 2, 4 and 16 hours after the initial fluorescence measurement. The value of fluorescence was compared to positive and negative controls. All groups were performed in triplicate.
[0125] Friction Measurement
[0126] Ocular biomechanical friction tests were conducted on freshly excised rabbit ocular tissues mounted on a Bose ELF3200 instrument using a custom eyelid-cornea friction testing procedure that was previously reported (Morrison, Sullivan et al. 2012; Schmidt, Sullivan et al. 2013; Samsom, Chan et al. 2015). To assess the lubricating ability of eye drops, the tissues were soaked in either an HA-only solution (1.0 mg/mL), or a PEG non-HA-binding peptide control solution (SABpep-PEG, 1.0 mg/mL), or an SABpep-PEG-HABpep solution (1.0 mg/mL) followed by a 1.0 mg/mL HA solution for 1 h in each solution and washed three times in saline for two minutes each between peptide and HA solutions and before mounting the samples for friction testing. During the friction testing, the tissues were articulated against each other at an effective sliding velocity of 0.3 mm per second and under three different normal loads of approximately 4-20 kPa, mimicking the blinking of a human eye. Each test sequence was repeated 3 times in a 0.25 mL saline lubricant bath.
[0127] HA release profile studies for SABpep-PEG-HABpep treated eyes.
[0128] Rabbit eyes (n=3) were placed in a 24-well plate and were incubated in 0.5 mg/ml SABpep-PEG-HABpep solution for 20 minutes. They were then washed with PBS three times and emerged in HA-FITC (10 .mu.g/ml) solution. Similar to the ex vivo method, positive controls were treated with only HA-FITC and negative controls were treated with only PBS. After twenty minutes, the eyes were washed with PBS three times and fluorescence was measured using an area scan from a microwell plate reader. Change in fluorescence was recorded periodically every 5 minutes for a total of 25 minutes and also after 2 hours, 4 hours and 16 hours after the initial fluorescence measurement.
[0129] In Vivo Studies.
[0130] In vivo studies were performed on mice treated with an HA eye drop solution containing SABpep-PEG-HABpep. SABpep eye drop solution (total volume 5 .mu.l) consisting of SABpep-PEG-HABpep and HA-FL was prepared by combining a 1:1 volume ratio of 5 mg/ml SABpep-PEG-HABpep and 1 mg/ml HA-FL solution (corresponds to 0.1% HA eye drop solution). Five mg/ml HA-FL solution mixed with an equal volume of PBS served as a control eye drop. Mice (C57BL/6, Charles River) were anesthetized with isoflurane (Baxter, Ill.) and SABpep-PEG-HABpep/HA eye drop solution was applied to the right eye while the control eye drop was applied to the left one. Isoflurane gas was then removed and the mice were allowed to regain consciousness. Mice were sacrificed approximately 5, 10 or 15 minutes after regaining consciousness. Whole mice were imaged using a fluorescent dissecting microscope (Nikon, Melville, N.Y.), allowing us to take images of the eye without harvesting the tissues. However, we also harvested eyes and imaged with Zeiss Axio Imager 2 microscope to obtain magnified images.
[0131] Live/dead staining and AlamarBlue assay for the cytotoxicity assessment of SABpep and ColBpep.
[0132] Rabbit conjunctival primary epithelial cells were isolated as described previously in the main text. Cells were seeded in 96 well microplates for AlamarBlue assays (Life Technologies, NY) and live/dead staining at a density of 10,000 cells/cm.sup.2. Cells were allowed to grow to about 90% confluence before experiments were carried out. Three different concentrations of HA-FL were tested: 1.0 mg/ml, 100 .mu.g/ml and 10 .mu.g/ml. SABpep and ColBpep were added to the medium for 24 h with a concentration of 1.0 mg/ml. Calcein AM and ethidium homodimer-1 (EthD-1; Life Technologies, NY) were used as live/dead staining agents according manufacture's manual, and cells were imaged using Zeiss Axio Imager 2 microscope. AlamarBlue assay was performed with quadruplets for every group. After 2 h incubation, the absorbance values at 570 nm on a Synergy 2 microplate reader were recorded.
[0133] Binding specificity of SABpep.
[0134] To examine if SABpep binds specifically to sialic acid segment on the oligosaccharide chains, paraffin embedded and 4% PFA fixed rabbit conjunctiva tissue sections were stained with three different groups were selected (SABpep, sialidase treated+SABpep, SA+SABpep). Sections in the first group were stained with SABpep as described in the main text; sections in the second group were treated with sialidase (.alpha.2-3,6,8 Neuraminidase, 60 U/ml; New England Biolabs, Ipswich, Mass.) first for one hour at 37.degree. C. before the addition of SABpep; and finally, sections in the third group were stained with SABpep pre-incubated with sialic acid (N-Acetylneuraminic acid, 100 mM; Carbosynth, Compton, Berkshire, UK) for one hour at room temperature. The staining procedure was the same in all three groups (streptavidin-fluorescein was used for detecting biotin conjugated SABpep; DAPI was used as counterstain for nuclei). The sections were imaged using Zeiss Axio Imager 2 microscope.
[0135] Statistical Analysis
[0136] One-way ANOVA was performed among groups to determine any statistically significance in mean values of HA retention on ex vivo rabbit ocular tissues (statistically significant values with p.ltoreq.0.05 was marked as *, p<0.0001 marked as ****, p<0.002 marked as ***, and p<0.005 marked as **). The measured kinetic coefficient of friction values between the treatment groups for each repeat of the test were analyzed by Cohen's d effect size. Cohen's d measure is defined as the difference between two means divided by the pooled standard deviation for the data and classifies effect sizes as small (d=0.2), medium (d=0.5), and large (d.gtoreq.0.8) (Cohen 1988). The effect size was calculated instead of significance as it allowed for the determination the magnitude of the difference between the two treatment groups independent from sample size (n=3).
[0137] In the following examples, SABpep-PEG-HABpep of the present invention was investigated for the amount and duration of HA bound on ocular surfaces and found that SABpep-PEG-HABpep bound HA that is immobilized to the ocular surfaces with the sialic acid as the anchoring sites prolongs HA retention in both ex vivo and in vivo animal models. In the ex vivo rabbit eye model, HA was bound 1.8 times more in the beginning and .about.1.2 times more at 24 h through SABpep-PEG-HABpep compared to control. In addition, the lubricating ability of the eye drops was assessed which showed a strong magnitude of difference in kinetic coefficient friction values for rabbit ocular tissues treated with SABpep-PEG-HABpep and HA solution compared to the tissues treated with HA-only solution (.about.20% in repeat 1 and .about.30% in repeat 2 and 3). The kinetic coefficient of friction measurements suggest that SABpep-PEG-HABpep was effective in immobilizing HA onto the ocular surface, and the surface bound HA without binding peptide sheared off from the tissue to a greater extent, which is consistent with the longer retention time of HA bound with SABpep-PEG-HABpep. Kinetic friction coefficient results from the SABpep-PEG-HABpep and HA solution showed a strong magnitude of difference compared to the tissues treated with a non-HA-binding PEG control peptide. This suggests that the PEG present in the SABpep-PEG-HABpep solution does not play a significant role in the kinetic coefficient of friction measurements obtained.
[0138] While these results were performed on a rabbit model and not on human tissue, the rabbit animal model has often been used in ophthalmic research due to their similarities to human ocular anatomy. Although rabbit ocular tissues have differences in tear film stability and sialic acid-containing membrane-bound mucin structures compared to human ocular tissue (Tsonis 2011), the rabbit model is a better alternative than testing against synthetic materials. Lastly, while increased in vitro friction of commercial contact lenses has been correlated with increased discomfort of wears, there are no `clinically relevant changes in tear friction` to reference from the literature. As such, the reported friction values are simply an in vitro measurement yet still provide a valuable information related to lubricating function of the peptide studied here.
[0139] In an in vivo mouse model, HA immobilized through the peptide was observed even at 15 min compared to 5 min for the control HA that was not bound with the peptide. Although HA bound was not quantified in vivo, the amount of HA-FL decreased more dramatically over the course of 30 min. However, this difference between in vivo and ex vivo retention times is expected because in a live animal the tear film is active and is constantly flushing fluid.
[0140] From a combination of in vivo, ex vivo, and in vitro studies we posit using animal models that peptide-binding agents included in HA eye-drops can prolong the physical and biological benefits of HA. Although glycan compositions on the ocular surface are different across species, human ocular surface has a higher occurrence of sialic acid than other species such as rabbits and canine, and we expect that SABpep eye drop technology will be extrapolated to human tissue too.
Example 1
[0141] Peptide screening: selecting peptide that binds to the ocular tissues.
[0142] We screened multiple peptides that are supposed to bind different sites of the ocular tissues and cells (data not shown). Out of the 6 screened peptides, a sialic acid binding peptide GGSPYGRC (SEQ ID NO: 15) hereafter respectively referred to as SABpep, and a collagen binding peptide SYIRIADTNIT (SEQ ID NO: 12) hereafter respectively referred to as ColBpep were chosen for further experiments (FIG. 2A). The chosen SABpep and ColBpep exhibited little to no cytotoxicity to epithelial cells as well (data not shown)
Example 2
[0143] SABpep and ColBpep binding on conjunctiva tissue.
[0144] Rabbit conjunctival sections bound SABpep specifically on the epithelial lining of the conjunctiva (FIG. 2B), indicating the strong presence of sialic acid on mucin-1 as compared to mucin-1 antibody staining. A higher magnification confirmed that SABpep staining was localized to only on cell membranes (FIG. 2C). Conversely, as expected, ColBpep staining was localized on conjunctiva tissue more indiscriminately, which is similar to type I collagen antibody staining (FIG. 2B). The binding specificity of SABpep was further proven by sialidase digestion and pre-incubation with sialic acid. SABpep could not bind to conjunctival epithelium if the section was digested by sialiadase before staining. However, SABpep still stained the epithelium even after preincubation with sialic acid solution (data not shown).
Example 3
[0145] Mucin and HA binding studies by Quartz Crystal Microbalance with Dissipation (QCM-D) monitoring.
[0146] Surface binding of polymer-peptide conjugates to mucin and HA was directly measured using QCM-D. First, sialic acid bearing mucin was adsorbed on gold substrates to mimic the interface at the corneal epithelium (FIG. 3A), as native mucin from bovine submaxillary glands is abundant with O-linked oligosaccharide chains, and can be used as a source of sialic acids. A visco-elastic layer of highly hydrated mucin was formed as shown by a frequency change of .DELTA.f=-32 Hz and dissipation change of .DELTA.D=7. Then SABpep-PEG-HABpep was applied to the mucin layer and specific binding was indicated with a frequency change of .DELTA.f=-5 Hz. In turn, SABpep-PEG-HABpep was immobilized onto HA substrates with a .DELTA.f=-3 Hz (FIG. 3B). The Voigt mass quantified the consistent formation of visco-elastic substrates Mucin and HA (FIG. 3C). In turn, HApep binding showed a frequency but not a dissipation change. Considering the relatively small molecular weight of SABpep-PEG-HABpep (Mw=3698.43 Da) we observed a considerable binding to mucin and HA with Sauerbrey mass of .DELTA.m=68.+-.28 and 54.+-.5 ng/cm.sup.2. For example, the drop in frequency directly corresponds to binding of the polymer-peptide to mucin and can be attributed to sialic acid chains branching out from the proteoglycan (Gipson 2004) with binding sites available in a 3-dimensional network.
Example 4
[0147] Ex vivo HA binding and release profile.
[0148] To confirm the binding of HA on SABpep-PEG-HABpep and ColBpep-PEG-HABpep treated ocular tissue samples, we applied HA-FL solution and monitored the fluorescence overtime. The peptide treated samples fluoresced with higher intensity compared to both positive (only HA) and negative controls (PBS) (FIG. 4A). The positive control had minimal fluorescence, and it was significantly lower than the peptide-treated groups, indicating that the ocular surface is poor at retaining HA without an aid from the peptide constructs. Fluorescent HA release studies performed on SABpep-PEG-HABpep treated samples showed .about.1.8-fold more bound HA in within the first 25 min (FIG. 4B), and .about.1.3-fold more after incubating overnight compared with HA only-treated controls (FIG. 4C). Although a detailed stoichiometric and control study to compare the binding ability of SABpep vs. ColBpep is required for an accurate comparison, our initial experiments with conjunctiva (physically abraded to expose collagen) showed a greater relative fluorescence value (only as an estimate) for SABpep+HA treated samples than ColBpep+HA treated samples (Fig. S3 add fig). We further calculated Area under curve (AUC) for HA release overtime (Fig. S4 add fig) that showed the total released amount of HA on ocular surface bound through SABpep was significantly higher than that for samples with only HA after 25 minutes and after 16 h.
Example 5
[0149] Lubrication of ocular tissues with eye drop solutions.
[0150] Rabbit ocular tissues treated with SABpep-PEG-HABpep and HA solution had reduced coefficient of friction values, and therefore improved tissue lubrication, compared to HA-only treated tissues as control. Kinetic coefficient of friction values of <.mu..sub.kinetic, N.sub.eq>, expressed as the mean.+-.standard error of the mean (SEM), decreased from 0.05.+-.0.01, to 0.04.+-.0.01 (d=1.25), 0.06.+-.0.01 to 0.04.+-.0.01 (d=2.13) and 0.07.+-.0.00 to 0.05.+-.0.01 (d=2.10) during the first, second and third repeats of the HA treated tissues compared to the SABpep-PEG-HABpep followed by HA treated tissues, respectively (FIG. 5). A strong effect size was observed in all three repeats, indicating that there is a strong magnitude of difference between the SABpep-PEG-HABpep+HA group compared to the HA control group. Kinetic friction coefficient values for SABpep-PEG-HABpep followed by HA treated tissues were also compared to tissues treated with a PEG non-HA-binding peptide (SABpep-PEG) solution, which acted as a polymer control. <.mu..sub.kinetic, N.sub.eq> values during the first, second and third repeats of the PEG non-HA-binding peptide solution were 0.08.+-.0.01, 0.07.+-.0.01 and 0.07.+-.0.01, exhibiting strong effect sizes (d=2.02, 2.70 and 1.34) when compared to the kinetic friction coefficient values of the tissues treated with SABpep-PEG-HABpep+HA (FIG. 5).
Example 6
[0151] In Vivo HA Retention Studies
[0152] An eye drop formulation with equal volume mixture of SABpep-PEG-HABpep and 0.1% HA (as in commercial HA eye drops) was applied in vivo to one of the mice eyes, while the other eye was applied with only 0.1% HA, and monitored overtime till 30 min. Eyes treated with SABpep eye drops retained HA longer than eyes treated with regular HA eye drops (15 minutes versus 10 minutes, respectively) (FIGS. 6A&B). The fluorescence from eyes treated with only HA was quickly vanishing and lasted till 10 min, while the fluorescence from peptide and HA treated eyes was still intense at 10 min and lasted even at 15 min (FIG. 6C). We further harvested mice eyes and imaged them at a higher magnification, which, as expected, showed more bound HA on ocular surface at epithelial cell level at 5 min and lasted till 15 min (FIGS. 6B&C). Initial and over time fluorescence of standard HA-FL eye drop solution with and without peptide solutions remained approximately the same that ruled out the difference in fluorescence values due to the pipetting errors (data not shown).
Example 7
[0153] SABpep with thiol end group was conjugated to HA (750K Da) with Maleimide functional group and FITC (fluorescent tag) in a standard Maleimide-thiol Michael addition chemistry. This HA with SABpep was used as an eye drop for the ex vivo applications in FIG. 9.
[0154] All references, including publications, patent applications, and patents, cited herein are hereby incorporated by reference to the same extent as if each reference were individually and specifically indicated to be incorporated by reference and were set forth in its entirety herein.
[0155] The use of the terms "a" and "an" and "the" and similar referents in the context of describing the invention (especially in the context of the following claims) are to be construed to cover both the singular and the plural, unless otherwise indicated herein or clearly contradicted by context. The terms "comprising," "having," "including," and "containing" are to be construed as open-ended terms (i.e., meaning "including, but not limited to,") unless otherwise noted. Recitation of ranges of values herein are merely intended to serve as a shorthand method of referring individually to each separate value falling within the range, unless otherwise indicated herein, and each separate value is incorporated into the specification as if it were individually recited herein. All methods described herein can be performed in any suitable order unless otherwise indicated herein or otherwise clearly contradicted by context. The use of any and all examples, or exemplary language (e.g., "such as") provided herein, is intended merely to better illuminate the invention and does not pose a limitation on the scope of the invention unless otherwise claimed. No language in the specification should be construed as indicating any non-claimed element as essential to the practice of the invention.
[0156] Preferred embodiments of this invention are described herein, including the best mode known to the inventors for carrying out the invention. Variations of those preferred embodiments may become apparent to those of ordinary skill in the art upon reading the foregoing description. The inventors expect skilled artisans to employ such variations as appropriate, and the inventors intend for the invention to be practiced otherwise than as specifically described herein. Accordingly, this invention includes all modifications and equivalents of the subject matter recited in the claims appended hereto as permitted by applicable law. Moreover, any combination of the above-described elements in all possible variations thereof is encompassed by the invention unless otherwise indicated herein or otherwise clearly contradicted by context.
REFERENCES
[0157] Abetz, L., K. Rajagopalan, et al. (2011). "Development and validation of the impact of dry eye on everyday life (IDEEL) questionnaire, a patient-reported outcomes (PRO) measure for the assessment of the burden of dry eye on patients." Health Qual Life Outcomes 9: 111. [0158] Ali, M. and M. E. Byrne (2009). "Controlled release of high molecular weight hyaluronic Acid from molecularly imprinted hydrogel contact lenses." Pharmaceut res 26(3): 714-726. [0159] Amemiya, K., T. Nakatani, et al. (2005). "Hyaluronan-binding motif identified by panning a random peptide display library." Biochim Biophys Acta 1724(1-2): 94-99. [0160] Bray, B. A. (2001). "The role of hyaluronan in the pulmonary alveolus." J Theor Biol 210(1):121-130. [0161] Cerretani, C. F., N. H. Ho, et al. (2013). "Water-evaporation reduction by duplex films: application to the human tear film." Adv Colloid Interface Sci 197-198:33-57. [0162] Cohen, J. (1988). "Statistical Power Analysis for the Behavioral Sciences, 2nd edn. Hillsdale, N.J.: Laurence Erlbaum Associates." Inc CIT0006. [0163] Corrales, R. M., S. Narayanan, et al. (2011). "Ocular Mucin Gene Expression Levels as Biomarkers for the Diagnosis of Dry Eye Syndrome." Invest Ophthalmol Vis Sci 52(11): 8363-8369. [0164] Ding, J. and D. A. Sullivan (2012). "Aging and dry eye disease." Exp Gerontol 47(7): 483-490. [0165] Georgiev, G. A., Y. Norihiko, et al. (2013). "Surface chemistry study of the interactions of hyaluronic acid and benzalkonium chloride with meibomian and corneal cell lipids." Soft Matter 9: 10841-10856. [0166] Gipson, I. K. (2004). "Distribution of mucins at the ocular surface." Exp Eye Res 78(3): 379-388. [0167] Gipson, I. K. (2007). "The ocular surface: the challenge to enable and protect vision: the Friedenwald lecture." Invest Ophthalmol Vis Sci 48(10): 4390-4398. [0168] Gipson, I. K., S. Spurr-Michaud, et al. (2014). "Comparison of the transmembrane mucins muc1 and muc16 in epithelial barrier function." PLOS One 9(6):e100393. [0169] Gomes, J. A. P., R. Amankwah, et al. (2004). "Sodium hyaluronate (hyaluronic acid) promotes migration of human corneal epithelial cells in vitro." Br J Ophthalmol 88(6): 821-825. [0170] Hamano, T., K. Horimoto, et al. (1996). "Sodium hyaluronate eyedrops enhance tear film stability." Jpn J Ophthalmol 40(1): 62-65. [0171] Hargittai, I. and M. Hargittai (2008). "Molecular structure of hyaluronan: an introduction." Struct Chem 19(5): 697-717. [0172] Heerze, L. D., P. C. Chong, et al. (1992). "Investigation of the lectin-like binding domains in pertussis toxin using synthetic peptide sequences. Identification of a sialic acid binding site in the S2 subunit of the toxin." J Biol Chem 267(36): 25810-25815. [0173] Heerze, L. D., R. H. Smith, et al. (1995). "Utilization of sialic acid-binding synthetic peptide sequences derived from pertussis toxin as novel anti-inflammatory agents." Glycobiology 5(4): 427-433. [0174] Horwath-Winter, J., A. Berghold, et al. (2003). "Evaluation of the clinical course of dry eye syndrome." Arch Ophthalmol (Chicago, Ill.: 1960) 121(10): 1364-1368. [0175] Johnson, M. E., P. J. Murphy, et al. (2006). "Effectiveness of sodium hyaluronate eyedrops in the treatment of dry eye." Graefes Archive for Clinical and Experimental Ophthalmology 244(1): 109-112. [0176] Kalamajski, S., A. Aspberg, et al. (2007). "The decorin sequence SYIRIADTNIT binds collagen type I." J Biol Chem 282(22): 16062-16067. [0177] Korb, D. R., J. P. Herman, et al. (2005). "Lid wiper epitheliopathy and dry eye symptoms." Eye Contact Lens 31(1): 2-8. [0178] Labetoulle, M., et al. (2015). "Clinical Efficacy of an Oil-based Lubricant Eye Drop in Dry Eye Patients with Lipid Deficiency." Acta Ophthalmol 93(S255). [0179] Majd, S. E., R. Kuijer, et al. (2014). "Both hyaluronan and collagen type II keep proteoglycan 4 (lubricin) at the cartilage surface in a condition that provides low friction during boundary lubrication." Langmuir 30(48): 14566-14572. [0180] Marner, K., P. M. Mooller, et al. (1996). "Viscous carbomer eye drops in patients with dry eyes. Efficacy and safety. A randomized, open, cross-over, multicentre study." Acta ophthalmol Scand 74(3): 249-252. [0181] Matsuda, M., W. Hatanaka, et al. (2014). "Short peptide motifs for long-lasting anchoring to the cell surface." Bioconjugate Chem 25(12): 2134-2143. [0182] Mochizuki, H., M. Yamada, et al. (2008). "Fluorophotometric measurement of the precorneal residence time of topically applied hyaluronic acid." Brit J Ophthalmol 92(1): 108-111. [0183] Mooradian, D. L., J. B. McCarthy, et al. (1993). "Characterization of FN-C/H-V, a novel synthetic peptide from fibronectin that promotes rabbit corneal epithelial cell adhesion, spreading, and motility." Invest Ophthalmol Vis Sci 34(1): 153-164. [0184] Morrison, S., D. A. Sullivan, et al. (2012). "Dose-Dependent and Synergistic Effects of Proteoglycan 4 on Boundary Lubrication at a Human Cornea-Polydimethylsiloxane Biointerface." Eye Contact Lens 38(1): 27-35. [0185] Nakamura, M., M. Hikida, et al. (1993). "Characterization of water retentive properties of hyaluronan." Cornea 12(5): 433-436. [0186] Paderi, J. E., R. Sistiabudi, et al. (2009). "Collagen-binding peptidoglycans: a biomimetic approach to modulate collagen fibrillogenesis for tissue engineering applications." Tissue Eng Part A 15(10): 2991-2999. [0187] Papas, E. B., J. B. Ciolino, et al. (2013). "The TFOS International Workshop on Contact Lens Discomfort: report of the management and therapy subcommittee." Invest Ophthalmol Vis Sci 54(11): TFOS183-203. [0188] Papas, E. B., D. Tilia, et al. (2014). "Consequences of wear interruption for discomfort with contact lenses." Optom Vis Sci 91(1): 24-31. [0189] Pauloin, T., M. Dutot, et al. (2009). "High molecular weight hyaluronan decreases UVB-induced apoptosis and inflammation in human epithelial corneal cells." Mol Vis 15: 577-583. [0190] Pflugfelder S. C. (2004). "Antiinflammatory therapy for dry eye." Am J Ophthalmol 137(2):337-342. [0191] Pili, K., S. Kastelan, et al. (2014). "Dry eye in contact lens wearers as a growing public health problem." Psychiatr Danub 26 Suppl 3: 528-532. [0192] Pouyeh, B., E. Viteri, et al. (2012). "Impact of ocular surface symptoms on quality of life in a United States veterans affairs population." Am J Ophthalmol 153(6): 1061-1066 e1063. [0193] Prabhasawat, P., N. Tesavibul, et al. (2007). "Performance profile of sodium hyaluronate in patients with lipid tear deficiency: randomised, double-blind, controlled, exploratory study." Brit J Ophthalmol 91(1): 47-50. [0194] Presti, D. and J. E. Scott (1994). "Hyaluronan-mediated protective effect against cell damage caused by enzymatically produced hydroxyl (OH.) radicals is dependent on hyaluronan molecular mass." Cell Biochem Funct 12(4): 281-288. [0195] Rah, M. J. (2011). "A review of hyaluronan and its ophthalmic applications." Optometry 82(1): 38-43. [0196] Read, M. L., P. B. Morgan, et al. (2009). "Measurement errors related to contact angle analysis of hydrogel and silicone hydrogel contact lenses." J Biomed Mater Res Part B. Appl Biomater 91(2): 662-668. [0197] Reddy, P., O. Grad, et al. (2004). "The economic burden of dry eye: a conceptual framework and preliminary assessment." Cornea 23(8): 751-761. [0198] Reviakine, I., D. Johannsmann, et al. (2011). "Hearing what you cannot see and visualizing what you hear: interpreting quartz crystal microbalance data from solvated interfaces." Anal Chem 83(23): 8838-8848. [0199] Royle, L., E. Matthews, et al. (2008) "Glycan structures of ocular surface mucins in man, rabbit and dog display species differences." Glycoconj J 25:763-773. [0200] Samsom, M., A. Chan, et al. (2015). "In vitro friction testing of contact lenses and human ocular tissues: Effect of proteoglycan 4 (PRG4)." Tribol Int 89: 27-33. [0201] Schmidt, T. A., D. A. Sullivan, et al. (2013). "Transcription, Translation, and Function of Lubricin, a Boundary Lubricant, at the Ocular Surface." JAMA Ophthalmol 131(6): 766-776. [0202] Sharma, A. and H. B. Hindman (2014). "Aging: a predisposition to dry eyes." J Ophthalmol 2014: 781683. [0203] Shimmura, S., M. Ono, et al. (1995). "Sodium hyaluronate eyedrops in the treatment of dry eyes." Brit J Ophthalmol 79(11): 1007-1011. [0204] Simmons, P. A., C. Carlisle-Wilcox, et al. (2015). "Comparison of novel lipid-based eye drops with aqueous eye drops for dry eye: a multicenter, randomized controlled trial." Clin Ophthalmol 9: 657-664. [0205] Singh, A., P. Li, et al. (2015). "A hyaluronic acid-binding contact lens with enhanced water retention." Cont Lens Anterior eye 38(2): 79-84. [0206] Snibson, G. R., J. L. Greaves, et al. (1992). "Ocular surface residence times of artificial tear solutions." Cornea 11(4): 288-293. [0207] Stuart, J. C. and J. G. Linn (1985). "Dilute sodium hyaluronate (Healon) in the treatment of ocular surface disorders." Ann Ophthalmol 17(3): 190-192. [0208] Tolg, C., S. R. Hamilton, et al. (2012). "A RHAMM mimetic peptide blocks hyaluronan signaling and reduces inflammation and fibrogenesis in excisional skin wounds." Am J Pathol 181(4): 1250-1270. [0209] Tonge, S., L. Jones, et al. (2001). "The ex vivo wettability of soft contact lenses." Curr Eye Res 23(1): 51-59. [0210] Tsonis, P. A. (2011). Animal Models in Eye Research. Academic Press. Tsubota, K. and M. Yamada (1992). "Tear evaporation from the ocular surface." Invest Ophthalmol Vis Sci 33(10): 2942-2950. [0211] Woodard, A. M., P. Argueso (2014). "Expression analysis of the transmembrane mucin muc20 in human corneal and conjuctival epithelia." Invest Ophthalmol Vis Sci 55 (10): 613-6138. [0212] Wei, X. E., M. Markoulli, et al. (2013). "Tear film break-up time in rabbits." Clin Exp Optom 96: 70-75. [0213] Varki, A. (2008). "Sialic acids in human health and disease." Trends Mol Med 14(8): 351-360. [0214] Young, G., J. Veys, et al. (2002). "A multi-centre study of lapsed contact lens wearers." Ophthalmic Physiol Opt 22(6): 516-527. [0215] Zheng, X., T. Goto, et al. (2013). "In vitro efficacy of ocular surface lubricants against dehydration." Cornea 32(9): 1260-1264.
Sequence CWU 1
1
20116PRTArtificial Sequencesynthetic sequence 1Arg Arg Asp Asp Gly
Ala His Trp Gln Phe Asn Ala Leu Thr Val Arg 1 5 10 15
217PRTArtificial Sequencesynthetic sequence 2Cys Arg Arg Asp Asp
Gly Ala His Trp Gln Phe Asn Ala Leu Thr Val 1 5 10 15 Arg
312PRTArtificial Sequencesynthetic sequence 3Gly Ala Ala Trp Gln
Phe Asn Ala Leu Thr Val Arg 1 5 10 412PRTArtificial
Sequencesynthetic sequence 4Gly Ala His Trp Gln Phe Ala Ala Leu Thr
Val Arg 1 5 10 512PRTArtificial Sequencesynthetic sequence 5Gly Ala
His Trp Gln Phe Asn Ala Leu Thr Val Ala 1 5 10 612PRTArtificial
Sequencesynthetic sequence 6Gly Ala His Trp Gln Phe Asn Ala Leu Thr
Val Arg 1 5 10 715PRTArtificial Sequencesynthetic sequence 7Ser Thr
Met Met Ser Arg Ser His Lys Thr Arg Ser His His Val 1 5 10 15
811PRTArtificial Sequencesynthetic sequence 8Arg Tyr Pro Ile Ser
Arg Pro Arg Lys Arg Cys 1 5 10 99PRTArtificial Sequencesynthetic
sequence 9Thr Ala Gly His Gly Arg Arg Trp Ser 1 5 1027PRTArtificial
Sequencesynthetic sequence 10Leu Lys Gln Lys Ile Lys His Val Val
Lys Leu Lys Val Val Val Lys 1 5 10 15 Leu Arg Ser Gln Leu Val Lys
Arg Lys Gln Asn 20 25 1120PRTArtificial Sequencesynthetic sequence
11Arg Arg Ala Asn Ala Ala Leu Lys Ala Gly Glu Leu Tyr Lys Ser Ile 1
5 10 15 Leu Tyr Gly Cys 20 1211PRTArtificial Sequencesynthetic
sequence 12Ser Tyr Ile Arg Ile Ala Asp Thr Asn Ile Thr 1 5 10
1310PRTArtificial Sequencesynthetic sequence 13Tyr Ser Phe Tyr Ser
Asp Glu Ser Leu Gln 1 5 10 146PRTArtificial Sequencesynthetic
sequence 14Trp Tyr Arg Gly Arg Leu 1 5 158PRTArtificial
Sequencesynthetic sequence 15Gly Gly Ser Pro Tyr Gly Arg Cys 1 5
1617PRTArtificial Sequencesynthetic sequence 16Gly Gly Pro Gln Glu
Gln Ile Thr Gln His Gly Ser Pro Tyr Gly Arg 1 5 10 15 Cys
1717PRTArtificial Sequencesynthetic sequence 17Ser Thr Met Met Ser
Arg Ser His Lys Thr Arg Ser His His Val Gly 1 5 10 15 Cys
186PRTArtificial Sequencesynthetic sequence 18Lys Lys Lys Lys Lys
Lys 1 5 199PRTArtificial Sequencesynthetic sequence 19Gly Trp Gln
Pro Pro Arg Ala Arg Ile 1 5 2017PRTArtificial Sequencesynthetic
sequence 20Gly Gly Pro Gln Glu Gln Ile Thr Gln His Gly Ser Pro Tyr
Gly Arg 1 5 10 15 Cys
D00000

D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

S00001
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.