Low Viscosity Lubricants Based On Methyl Paraffin Containing Hydrocarbon Fluids
OUMAR-MAHAMAT; Halou ; et al.
U.S. patent application number 16/002109 was filed with the patent office on 2018-12-27 for low viscosity lubricants based on methyl paraffin containing hydrocarbon fluids. The applicant listed for this patent is ExxonMobil Research and Engineering Company. Invention is credited to Mark P. HAGEMEISTER, Halou OUMAR-MAHAMAT.
| Application Number | 20180371348 16/002109 |
| Document ID | / |
| Family ID | 62751583 |
| Filed Date | 2018-12-27 |











View All Diagrams
| United States Patent Application | 20180371348 |
| Kind Code | A1 |
| OUMAR-MAHAMAT; Halou ; et al. | December 27, 2018 |
LOW VISCOSITY LUBRICANTS BASED ON METHYL PARAFFIN CONTAINING HYDROCARBON FLUIDS
Abstract
A lubricating oil base stock including a lubricating oil base stock including from 5 to 50 wt % of 9-methylnonadecane and from 95 to 50 wt % of 9-methyl-11-octylheneicosane. The lubricating oil base stock has a relationship between Noack volatility at 250.degree. C. as measured by ASTM D5800 (y) and kinematic viscosity at 40.degree. C. as measured by ASTM D445 (x) that is less than y=2.15-0.765*ln(x). Also provided is a lubricating oil containing the lubricating oil base stock and one or more lubricating oil additives. A method for improving one or more of thermal and oxidative stability, deposit control and traction control in a lubricating oil by using as the lubricating oil a formulated oil containing the lubricating oil base stock and one or more lubricating oil additives is also provided.
| Inventors: | OUMAR-MAHAMAT; Halou; (Mullica Hill, NJ) ; HAGEMEISTER; Mark P.; (Mullica Hill, NJ) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 62751583 | ||||||||||
| Appl. No.: | 16/002109 | ||||||||||
| Filed: | June 7, 2018 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 62523398 | Jun 22, 2017 | |||
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C10M 105/04 20130101; C10M 107/10 20130101; C10N 2040/25 20130101; C10N 2020/017 20200501; C10N 2030/02 20130101; C10N 2030/08 20130101; C10N 2030/54 20200501; C10N 2030/06 20130101; C10N 2030/74 20200501; C10N 2020/071 20200501; C10M 2209/084 20130101; C10N 2020/083 20200501; C10N 2030/10 20130101; C10M 2203/0206 20130101; C10M 2203/022 20130101; C10M 2205/0285 20130101; C10N 2020/02 20130101; C10N 2020/065 20200501 |
| International Class: | C10M 105/04 20060101 C10M105/04 |
Claims
1. A lubricating oil base stock comprising from 5 to 50 wt % of 9-methylnonadecane and from 95 to 50 wt % of 9-methyl-11-octylheneicosane, wherein the base stock has a relationship between Noack volatility at 250.degree. C. as measured by ASTM D5800 (y) and kinematic viscosity at 40.degree. C. as measured by ASTM D445 (x) that is less than y=2.15-0.765*ln(x).
2. The lubricating oil base stock of claim 1, wherein the base stock has a kinematic viscosity at 100.degree. C. as measured by ASTM D445 of from 1.5 to 3.5 cSt.
3. The lubricating oil base stock of claim 1, wherein the base stock has a kinematic viscosity at 40.degree. C. as measured by ASTM D445 of from 4.0 to 14.0 cSt.
4. The lubricating oil base stock of claim 1 comprising about 30 wt % of 9-methylnonadecane and about 70 wt % of 9-methyl-11-octylheneicosane.
5. The lubricating oil base stock of claim 1 comprising about 10 wt % of 9-methylnonadecane and about 90 wt % of 9-methyl-11-octylheneicosane.
6. The lubricating oil base stock of claim 1, wherein the base stock has a Noack volatility at 250.degree. C. as measured by ASTM D5800 of 10 to 90%.
7. The lubricating oil base stock of claim 1, wherein the base stock has a Viscosity Index from about 100 to 170 as determined by ASTM D2270.
8. The lubricating oil base stock of claim 1, wherein the base stock has a pour point of from about -10 to -80.degree. C. as determined by ASTM D5950.
9. The lubricating oil base stock of claim 1, wherein the base stock has a MTM average traction coefficient (at 100 deg. C, 1 GPa, 2 m/s and 0-100% SRR) of from about 0.0060 to 0.0090.
10. The lubricating oil base stock of claim 1, wherein the base stock has a MTM average traction coefficient (at 100 deg. C, 1 GPa, 2 m/s and 0-100% SRR) that is about 20 to 180% lower than a Group II or Group III base stock of comparable KV100.degree. C. viscosity.
11. The lubricating oil base stock of claim 1, wherein the base stock has a high temperature high shear (HTHS) viscosity of less than about 1.6 cP as determined by ASTM D4683, and a Noack volatility from about 16 to about 30 percent as determined by ASTM D5800.
12. A lubricating oil comprising a major amount of a lubricating oil base stock and a minor amount of one or more additives, said lubricating oil base stock comprising from 5 to 50 wt % of 9-methylnonadecane and from 95 to 50 wt % of 9-methyl-11-octylheneicosane, wherein the base stock has a relationship between Noack volatility at 250.degree. C. as measured by ASTM D5800 (y) and kinematic viscosity at 40.degree. C. as measured by ASTM D445 (x) that is less than y=2.15-0.765*ln(x).
13. The lubricating oil of claim 12 wherein the one or more additives are selected from the group consisting of a viscosity improver or modifier, antioxidant, detergent, dispersant, pour point depressant, corrosion inhibitor, metal deactivator, seal compatibility additive, anti-foam agent, inhibitor, anti-rust additive and combinations thereof.
14. The lubricating oil of claim 12, wherein the major amount of the lubricating oil base stock is from about 55 to 99 wt % of the lubricating oil and the minor amount of the one or more additives is from about 45 to 1 wt % of the lubricating oil.
15. The lubricating oil of claim 12, wherein the oil has a kinematic viscosity at 100.degree. C. as measured by ASTM D445 of from 2.0 to 6.0 cSt.
16. The lubricating oil of claim 12, wherein the oil has a kinematic viscosity at 40.degree. C. as measured by ASTM D445 of from 5.0 to 25.0 cSt.
17. The lubricating oil of claim 12 wherein the lubricating oil base stock comprises about 30 wt % of 9-methylnonadecane and about 70 wt % of 9-methyl-11-octylheneicosane.
18. The lubricating oil of claim 12 wherein the lubricating oil base stock comprises about 10 wt % of 9-methylnonadecane and about 90 wt % of 9-methyl-11-octylheneicosane.
19. The lubricating oil of claim 12, wherein the oil has a Noack volatility at 250.degree. C. as measured by ASTM D5800 of 10 to 90%.
20. The lubricating oil of claim 12, wherein the oil has a Viscosity Index from about 100 to 200 as determined by ASTM D2270.
21. The lubricating oil of claim 12, wherein the oil has a pour point of from about -10 to -60.degree. C. as determined by ASTM D5950.
22. The lubricating oil base stock of claim 12, wherein the oil has a MTM average traction coefficient (at 100 deg. C, 1 GPa, 2 m/s and 0-100% SRR) of from about 0.0050 to 0.0090.
23. The lubricating oil of claim 12, wherein the oil has a MTM average traction coefficient (at 100 deg. C, 1 GPa, 2 m/s and 0-100% SRR) that is about 20 to 180% lower than a comparable lubricating oil including a Group II or a Group III base stock of comparable KV100.degree. C. viscosity.
24. The lubricating oil of claim 12, wherein the oil has a high temperature high shear (HTHS) viscosity of less than about 2.3 cP as determined by ASTM D4683.
25. The lubricating oil of claim 12, wherein the oil has an oxidation stability (210 hour test with time to 200% KV40 increase) of from about 60 to 150 hours.
26. The lubricating oil of claim 12, wherein the oil has resistance to deposit formation as measured by TEOST 33C test for 2 hours at 200 to 480 deg. C per ASTM D6335 of from 10 to 30 mg.
27. The lubricating oil of claim 12, wherein the oil has a Cold Crank Simulator (CC S) viscosity at -35 deg. C per ASTM D5293 of from 700 to 1000 mPas.
28. The lubricating oil of claim 13 wherein the one or more additives include a viscosity modifier selected from a polymethacrylate, a hydrocarbon hydrogenated polyisoprene star polymer and combinations thereof.
29. The lubricating oil of claim 28 wherein the oil has a high temperature high shear (HTHS) viscosity of less than about 2.3 cP as determined by ASTM D4683.
30. The lubricating oil of claim 28 wherein the oil has a Viscosity Index from about 220 to 340 as determined by ASTM D2270.
31. The lubricating oil of claim 12 further including a cobase stock at from 5 to 40 wt %, wherein the cobase stock is selected from the group consisting of a Group I base stock, a Group II base stock, a Group III base stock, a Group IV base stock, a Group V base stock and combinations thereof.
32. The lubricating oil of claim 12, wherein the oil has a high temperature high shear (HTHS) viscosity of less than about 2.3 cP as determined by ASTM D4683, and a Noack volatility from about 15 to about 90 percent as determined by ASTM D5800.
33. A method for improving one or more of thermal and oxidative stability, deposit control and traction control in a lubricating oil comprising: providing a lubricating oil including a major amount of a lubricating oil base stock and a minor amount of one or more additives, said lubricating oil base stock comprising from 5 to 50 wt % of 9-methylnonadecane and from 95 to 50 wt % of 9-methyl-11-octylheneicosane, wherein the base stock has a relationship between Noack volatility at 250.degree. C. as measured by ASTM D5800 (y) and kinematic viscosity at 40.degree. C. as measured by ASTM D445 (x) that is less than y=2.15-0.765*ln(x), and using the lubricating oil in a formulated oil to improve one or more of thermal and oxidative stability, deposit control and traction control.
34. The method of claim 33 wherein the one or more additives are selected from the group consisting of a viscosity improver or modifier, antioxidant, detergent, dispersant, pour point depressant, corrosion inhibitor, metal deactivator, seal compatibility additive, anti-foam agent, inhibitor, anti-rust additive and combinations thereof.
35. The method of claim 33, wherein the major amount of the lubricating oil base stock is from about 55 to 99 wt % of the lubricating oil and the minor amount of the one or more additives is from about 45 to 1 wt % of the lubricating oil.
36. The method of claim 33, wherein the oil has a kinematic viscosity at 100.degree. C. as measured by ASTM D445 of from 2.0 to 6.0 cSt.
37. The method of claim 33, wherein the oil has a kinematic viscosity at 40.degree. C. as measured by ASTM D445 of from 5.0 to 25.0 cSt.
38. The method of claim 33, wherein the lubricating oil base stock comprises about 30 wt % of 9-methylnonadecane and about 70 wt % of 9-methyl-11-octylheneicosane.
39. The method of claim 33, wherein the lubricating oil base stock comprises about 10 wt % of 9-methylnonadecane and about 90 wt % of 9-methyl-11-octylheneicosane.
40. The method of claim 33, wherein the oil has a Noack volatility at 250.degree. C. as measured by ASTM D5800 of 10 to 90%.
41. The method of claim 33, wherein the oil has a Viscosity Index from about 100 to 200 as determined by ASTM D2270.
42. The method of claim 33, wherein the oil has a pour point of from about -10 to -60.degree. C. as determined by ASTM D5950.
43. The method of claim 33, wherein the oil has a MTM average traction coefficient (at 100 deg. C, 1 GPa, 2 m/s and 0-100% SRR) of from about 0.0050 to 0.0090.
44. The method of claim 33, wherein the oil has a MTM average traction coefficient (at 100 deg. C, 1 GPa, 2 m/s and 0-100% SRR) that is about 20 to 180% lower than a comparable lubricating oil including a Group II or a Group III base stock of comparable KV100.degree. C. viscosity.
45. The method of claim 33, wherein the oil has a high temperature high shear (HTHS) viscosity of less than about 2.3 cP as determined by ASTM D4683.
46. The method of claim 33, wherein the oil has an oxidation stability (210 hour test with time to 200% KV40 increase) of from about 60 to 150 hours.
47. The method of claim 33, wherein the oil has resistance to deposit formation as measured by TEOST 33C test for 2 hours at 200 to 480 deg. C per ASTM D6335 of from 10 to 30 mg.
48. The method of claim 33, wherein the oil has a Cold Crank Simulator (CCS) viscosity at -35 deg. C per ASTM D5293 of from 700 to 1000 mPas.
49. The method of claim 34, wherein the one or more additives include a viscosity modifier selected from a polymethacrylate, a hydrocarbon hydrogenated polyisoprene star polymer and combinations thereof.
50. The method of claim 49 wherein the oil has a high temperature high shear (HTHS) viscosity of less than about 2.3 cP as determined by ASTM D4683.
51. The method of claim 49 wherein the oil has a Viscosity Index from about 220 to 340 as determined by ASTM D2270.
52. The method of claim 33 wherein the oil further includes a cobase stock at from 5 to 40 wt % of the formulated oil, wherein the cobase stock is selected from the group consisting of a Group I base stock, a Group II base stock, a Group III base stock, a Group IV base stock, a Group V base stock and combinations thereof.
53. The method of claim 33, wherein the oil has a high temperature high shear (HTHS) viscosity of less than about 2.3 cP as determined by ASTM D4683, and a Noack volatility from about 15 to about 90 percent as determined by ASTM D5800.
54. A method of making a low viscosity low volatility lubricating oil comprising: providing a lubricating oil base stock and one or more additives, wherein the lubricating oil base stock comprises from 5 to 50 wt % of 9-methylnonadecane and from 95 to 50 wt % of 9-methyl-11-octylheneicosane, and blending a major amount of the lubricating oil base stock and a minor amount of the one or more additives to form the lubricating oil, wherein the lubricating oil base has a relationship between Noack volatility at 250.degree. C. as measured by ASTM D5800 (y) and kinematic viscosity at 40.degree. C. as measured by ASTM D445 (x) that is less than y=2.15-0.765 ln(x).
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application claims the benefit of U.S. Provisional Application No. 62/523,398, filed on Jun. 22, 2017, the entire contents of which are incorporated herein by reference.
FIELD
[0002] This disclosure relates to low viscosity, low volatility lubricating oil base stocks, and lubricating oils containing the lubricating oil base stocks. This disclosure also relates to a method for improving one or more of thermal and oxidative stability, volatility, viscosity index, deposit control and traction control in a lubricating oil by using as the lubricating oil a formulated oil containing the lubricating oil base stock.
BACKGROUND
[0003] Lubricants in commercial use today are prepared from a variety of natural and synthetic base stocks admixed with various additive packages and solvents depending upon their intended application. The base stocks typically include mineral oils, polyalphaolefins (PAO), gas-to-liquid base oils (GTL), silicone oils, phosphate esters, monoesters, diesters, polyol esters, and the like.
[0004] A major trend for passenger car engine oils (PCEOs) is an overall improvement in quality as higher quality base stocks become more readily available. Typically the highest quality PCEO products are formulated with base stocks such as PAOs or GTL stocks admixed with various additive packages.
[0005] For improving fuel economy, base oil viscosity is very important. Substantial improved fuel economy (>2%) requires breakthrough in: (1) base oil volatility (2) durability and (3) friction. Friction losses occur between the moving components within the engine. Models developed to date indicate that fuel economy is heavily influenced by the lubricant properties at high shear. The base stock contributes a greater proportion of the total viscosity under high shear conditions than under low shear. Lowering base stock viscosity is likely to have the largest impact on future fuel economy gains.
[0006] Current commercial PAO fluids (e.g., SpectraSyn.TM. 2) based on hydrocarbon and commercial esters (e.g., 2-ethylhexyl adipate, di-2-ethylhexyl azelate, Esterex.TM. A32, Esterex.TM. A34) do not adequately allow formulation of ultra-low viscosity lubricant while still meeting API specification (e.g., Noack volatility of 15% or less) and/or other OEM (original equipment manufacturers) set specifications or requirements. In order to formulate ultra-low viscosity lubricant for fuel economy benefit, it is desirable to have low viscosity and low volatility properties co-exist in the same base stock, for meeting volatility requirement. In addition, the base stock should also possess adequate thermal and oxidative stability at high temperature to prevent or minimize deposit formation. Good compatibility with additives commonly used in lubricant formulations (PVL, Passenger Vehicle Lubricants, CVL, Commercial Vehicle Lubricants, and industrial lubricants), good low temperature properties, and acceptable viscosity indices are also necessary for the base stocks.
[0007] Poly-.alpha.-olefins (PAOs) are important lube base stocks with many excellent lubricant properties, including high viscosity index (VI), low volatility and are available in various viscosity range (Kv.sub.100 2-300 cSt). However, PAOs are paraffinic hydrocarbons with low polarity. This low polarity leads to low solubility and dispersancy for polar additives or sludge generated during service. To compensate for this low polarity, lube formulators usually add one or multiple polar cobase stocks. Ester or alkylated naphthalene (AN) is usually present at 1 wt. % to 50 wt. % levels in many finished lubricant formulations to increase the fluid polarity which improves the solubility of polar additives and sludge.
[0008] Future automotive and industrial trend suggest that there will be a need for advanced additive technology and synthetic base stocks with substantially better thermal and oxidative stability. This is primarily because of smaller sump sizes that will have more thermal and oxidative stresses on the lubricants. Performance requirements have become more stringent in the past 10 to 20 years and the demand for longer drain intervals has grown steadily. Also, the use of Group II, III and IV base oils is becoming more widespread. Such base oils have very little sulfur content since natural sulfur-containing antioxidants are either absent or removed during the severe refining process.
[0009] It is known that lubricant oils used in internal combustion engines and transmission of automobile engines or trucks are subjected to demanding environments during use. These environments result in the lubricant suffering oxidation catalyzed by the presence of impurities in the oil, such as iron (wear) compounds and elevated temperatures. The oxidation manifests itself by increase in acid or viscosity and deposit formation or any combination of these symptoms. These are controlled to some extent by the use of antioxidants which can extend the useful life of the lubricating oil, particularly by reducing or preventing unacceptable viscosity increases. Besides oxidation inhibition, other parameters such as rust and wear control are also important.
[0010] A major challenge in engine oil formulation is simultaneously achieving improved fuel economy while also achieving appropriate low temperature properties, and oxidative stability.
[0011] Therefore, there is need for better additive and base stock technology for lubricant compositions that will meet ever more stringent requirements of lubricant users. In particular, there is a need for additive technology and synthetic base stocks with improved fuel economy, viscosity indices, and oxidative stability.
[0012] The present disclosure also provides many additional advantages, which shall become apparent as described below.
SUMMARY
[0013] This disclosure provides lubricating oil base stocks that include one or more methyl substituted hydrocarbon fluids that have desirable low viscosity/low volatility properties while exhibiting good high-temperature thermal-oxidative stability. Thus, the lubricating oil base stocks of this disclosure provide a solution to achieve enhanced fuel economy and energy efficiency. In addition, thermal, oxidative, and inherent hydrolytic stability, deposit control and traction control are other advantages of these base stocks.
[0014] This disclosure relates in part to a lubricating oil base stock comprising from 5 to 50 wt % of 9-methylnonadecane and from 95 to 50 wt % of 9-methyl-11-octylheneicosane, wherein the base stock has a relationship between Noack volatility at 250.degree. C. as measured by ASTM D5800 (y) and kinematic viscosity at 40.degree. C. as measured by ASTM D445 (x) that is less than y=2.15-0.765*ln(x). The chemical structures of the 9-methylnonadecane and 9-methyl-11-octylheneicosane base stocks for blending are as follows:
##STR00001##
[0015] This disclosure also relates in part to a lubricating oil comprising a major amount of a lubricating oil base stock and a minor amount of one or more additives. The lubricating oil base stock comprising from 5 to 50 wt % of 9-methylnonadecane and from 95 to 50 wt % of 9-methyl-11-octylheneicosane, wherein the base stock has a relationship between Noack volatility at 250.degree. C. as measured by ASTM D5800 (y) and kinematic viscosity at 40.degree. C. as measured by ASTM D445 (x) that is less than y=2.15-0.7651n(x).
[0016] This disclosure also relates in part to a method for improving one or more of thermal and oxidative stability, deposit control and traction control in a lubricating oil comprising: providing a lubricating oil including a major amount of a lubricating oil base stock and a minor amount of one or more additives, said lubricating oil base stock comprising from 5 to 50 wt % of 9-methylnonadecane and from 95 to 50 wt % of 9-methyl-11-octylheneicosane, wherein the base stock has a relationship between Noack volatility at 250.degree. C. as measured by ASTM D5800 (y) and kinematic viscosity at 40.degree. C. as measured by ASTM D445 (x) that is less than y=2.15-0.765*ln(x), and using the lubricating oil in a formulated oil to improve one or more of thermal and oxidative stability, deposit control and traction control.
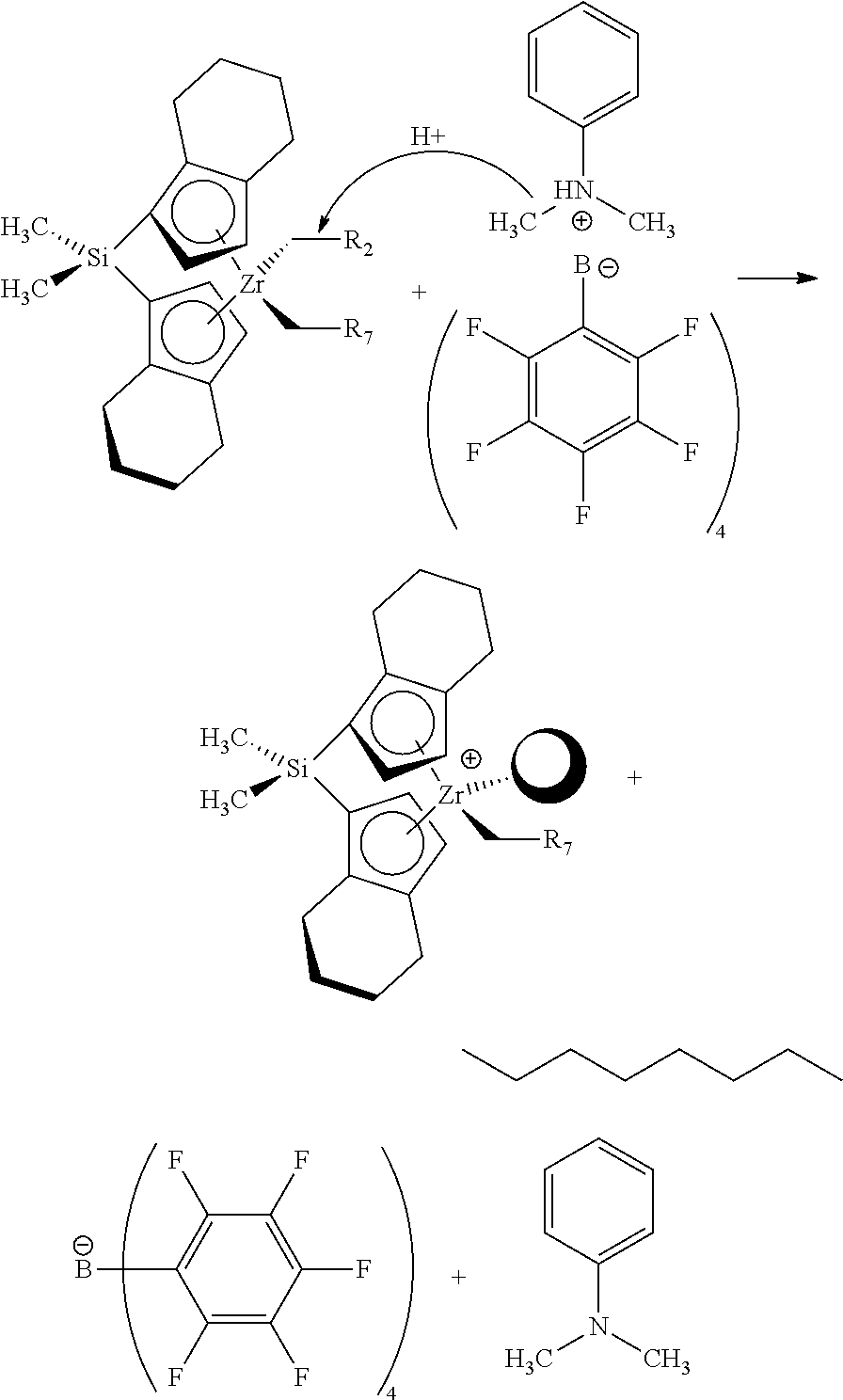
[0017] This disclosure also relates in part to a method for making a low viscosity low volatility lubricating oil comprising: providing a lubricating oil base stock and one or more additives, wherein the lubricating oil base stock comprises from 5 to 50 wt % of 9-methylnonadecane and from 95 to 50 wt % of 9-methyl-11-octylheneicosane, and blending a major amount of the lubricating oil base stock and a minor amount of the one or more additives to form the lubricating oil, wherein the lubricating oil base has a relationship between Noack volatility at 250.degree. C. as measured by ASTM D5800 (y) and kinematic viscosity at 40.degree. C. as measured by ASTM D445 (x) that is less than y=2.15-0.765*ln(x).
[0018] It has been surprisingly found that outstanding low viscosity low volatility properties, good high-temperature thermal and oxidative stability, deposit control, and traction benefits, can be attained in an engine lubricated with a lubricating oil by using as the lubricating oil a formulated oil in accordance with this disclosure. In particular, a lubricating oil base stock comprising low viscosity methyl paraffins exhibits low viscosity, low volatility, superior oxidative stability, desired deposit control and traction benefits, which helps to prolong the useful life of lubricants and significantly improve the durability and resistance of lubricants when exposed to high temperatures. The lubricating oils of this disclosure are particularly advantageous as passenger vehicle (PVL) or commercial vehicle (CVL) engine oils and/or driveline oil products.
[0019] The lubricating oils of this disclosure are particularly advantageous as passenger vehicle engine oil (PVEO) products, more specifically SAE 0WX, SAE SWX, or SAE 10WX, (where X=4, 8, 12, 16, 20, 30, 40, or 50), and similar oil formulations, especially oil formulations exhibiting lowered volatility when blended with the components of this invention.
[0020] Further objects, features and advantages of the present disclosure will be understood by reference to the following drawings and detailed description.
BRIEF DESCRIPTION OF THE DRAWINGS
[0021] FIG. 1 shows the physical characteristics of inventive low viscosity methyl paraffin based base stocks and comparative low viscosity base stocks.
[0022] FIG. 2 shows the physical characteristics and performance test results of engine oils formulations including the inventive methyl paraffin based and comparative low viscosity base stocks.
[0023] FIG. 3 shows kinematic viscosity and viscosity index of engine oils formulations including the inventive methyl paraffin based and comparative low viscosity base stocks which also include viscosity modifier.
[0024] FIG. 4 is a graph of the relationship between Noack volatility and kinematic viscosity at 40 deg. C and shows that the inventive methyl paraffin based low viscosity base stock blends have a relationship between Noack volatility (y) and KV40 (x) that is less than y=2.15-0.765*ln(x).
[0025] FIG. 5 shows the base stock blend ratios for the inventive mixtures of C10 dimer and C10 trimer of the inventive methyl paraffin based low viscosity base stock blends and the comparative mixtures of 2 cSt and 3.6 cSt conventional PAOs of the comparative PAO low viscosity base stock blends of FIG. 4.
DETAILED DESCRIPTION
[0026] All numerical values within the detailed description and the claims herein are modified by "about" or "approximately" the indicated value, and take into account experimental error and variations that would be expected by a person having ordinary skill in the art.
Lubricating Oil Base Stocks
[0027] The inventive base stocks of this disclosure are blends of C10 dimer and C10 trimer. In particular, the inventive base stocks of this disclosure are blends of 9-methylnonadecane (also referred to herein as "C10 dimer") and 9-methyl-11-octylheneicosane (also referred to herein as "C10 trimer"). The chemical structures of the 9-methylnonadecane and 9-methyl-11-octylheneicosane base stocks for blending are as follows:
##STR00002##
[0028] Also shown below is the chemical structure of a comparative 11-methyltricosane basestock (also referred to herein as "C12 dimer").
##STR00003##
[0029] These base stocks exhibit (1) outstanding low viscosity low volatility properties, (2) good high-temperature thermal and oxidative stability, (3) good low temperature properties and in particular high viscosity indices, (4) good deposit control, and (4) traction benefits, which make them attractive as Group IV synthetic base stocks in high performance, fuel economy lubricant applications.
[0030] Low viscosity base stocks (e.g., kinematic viscosity at 100.degree. C., 2-3 cSt) currently available in the marketplace are too volatile to be used for formulating next-generation ultra-low viscosity engine oils (i.e., xxW-4.fwdarw.xxW-16). These base stocks (e.g., SpectraSyn.TM. 2, QHVI.TM. 3, bis-(2-ethylhexyl) adipate, di-2-ethylhexyl azelate, Esterex.TM. A32) are unable to provide formulated engine oils that also meet current volatility API specification or other OEM volatility requirements. The present disclosure provides blends of methyl paraffins that have desirable low viscosity and low volatility properties while exhibiting traction benefits, good deposit control behavior and good high-temperature thermal-oxidative stability, and hence provide a solution to achieve enhanced fuel economy and energy efficiency.
[0031] As indicated above, the methyl paraffin base stock components useful in this disclosure include, a blend of a C10 dimer compound and a C10 trimer compound represented by the formula below.
##STR00004##
[0032] The methyl paraffin lubricating oil base stock may include from 10 to 60 wt % of the C10 dimer and from 40 to 90 wt % of the C10 trimer or may include from 5 to 50 wt % of the C10 dimer and from 50 to 95 wt % of the C10 trimer. More particularly, the C10 dimer in the inventive methyl paraffin base stock blend may be 5 wt %, 10 wt %, or 15 wt %, or 20 wt %, or 25 wt %, or 30 wt %, or 35 wt %, or 40 wt %, or 45 wt %, or 50 wt %, or 55 wt %, or 60 wt % of the total base stock blend. The C10 trimer in the inventive methyl paraffin base stock blend may be 95 wt %, 90 wt %, or 85 wt %, or 80 wt %, or 75 wt %, or 70 wt %, or 65 wt %, or 60 wt %, or 55 wt %, or 50 wt %, or 45 wt %, or 40 wt % of the total base stock blend. In one preferred form of the inventive methyl paraffin base stock blend, the C10 dimer constitutes 30 wt % of the total base stock blend and the C10 trimer constitutes 70 wt % of the total base stock blend. In another preferred form of the inventive methyl paraffin base stock blend, the C10 dimer constitutes 10 wt % of the total base stock blend and the C10 trimer constitutes 90 wt % of the total base stock blend.
[0033] The methyl paraffin lubricating oil base stocks of the instant disclosure have a viscosity (Kv.sub.100) from 1.5 cSt to 3.5 cSt, or 1.7 to 3.3 cSt, or 1.9 to 3.1 cSt, or 2.1 to 2.9 cSt, or 2.3 to 2.7 cSt at 100.degree. C., as determined by ASTM D445 or ASTM D7042. The methyl paraffin lubricating oil base stocks of the instant disclosure have a viscosity (Kv.sub.40) from 4.0 cSt to 14.0 cSt, or 4.5 to 13.5 cSt, or 5.0 to 13.0 cSt, or 5.5 to 12.5 cSt, or 6.0 to 12.0 cSt, 6.5 cSt to 11.5 cSt, or 7.0 to 11.0 cSt, or 7.5 to 10.5 cSt, or 8.0 to 10.0 cSt, or 8.5 to 9.5 cSt, or 8.7 to 9.3 cSt, as determined by ASTM D445 or ASTM D7042.
[0034] The methyl paraffin lubricating oil base stocks of the instant disclosure may have a Noack volatility at 250.degree. C. from about 10 to about 90 percent as determined by ASTM D5800. Alternatively, the methyl paraffin lubricating oil base stocks blends may have a Noack volatility at 250.degree. C. of from 12 to 85%, or 16 to 80%, or 20 to 75%, or 24 to 70%, or 28 to 65%, or 32 to 60%, or 36 to 55% as determined by ASTM D5800.
[0035] The methyl paraffin lubricating oil base stocks of the instant disclosure may have a Viscosity Index from about 100 to about 170 as determined by ASTM D2270. Alternatively, the methyl paraffin lubricating oil base stocks blends may have a Viscosity Index of from 105 to 165, or 110 to 160, or 115 to 155, or 120 to 150, or 125 to 145, or 130 to 140 as determined by ASTM D2270.
[0036] The methyl paraffin lubricating oil base stocks of the instant disclosure may have a pour point of from about -10 to -80.degree. C. as determined by ASTM D5950. Alternatively, the methyl paraffin lubricating oil base stocks blends may have a pour point of from -15 to -70.degree. C., or -17 to -65.degree. C., or -19 to -60.degree. C., or -21 to -55.degree. C., or -23 to -50.degree. C., or -25 to -40.degree. C. as determined by ASTM D5950.
[0037] The methyl paraffin lubricating oil base stocks of the instant disclosure may have a MTM average traction coefficient (at 100 deg. C, 1 GPa, 2 m/s and 0-100% SRR) ranging from 0.0060 to 0.0090. Alternatively, the methyl paraffin lubricating oil base stocks blends may have a MTM average traction coefficient ranging from 0.0055 to 0.0090, or 0.0060 to 0.0085, or 0.0065 to 0.0080. The MTM average traction coefficient may correlate with fuel efficiency with lower values providing improved fuel economy. The methyl paraffin lubricating oil base stocks blends of the instant disclosure have MTM average traction coefficient values that are from 20 to 180% lower, or from 40 to 160% lower, or from 60 to 140% lower, or from 80 to 120% lower, or from 90 to 110% lower than conventional Group II, Group III and Group III (GTL) base stocks of comparable KV100.degree. C. viscosity.
Method of Making Lubricating Oil Base Stocks
[0038] The methyl paraffin lubricating oil base stock components of the present disclosure can be prepared by a process that involves the oligomerization of linear alpha olefins using a metallocene catalyst followed by hydrogenation. More specifically, 9-methylnonadecane (C10 Dimer) and 9-methyl-11-octylheneicosane (C10 trimer), may be manufactured by oligomerization of 1-decene, [1-dodecene for 11-methyltricosane (C12 Dimer)], using a metallocene catalyst followed by hydrogenation. Pure compounds may then be obtained for the C10 dimer, C10 trimer, and C12 dimer by fractionation.
[0039] The methyl paraffin lubricating oil base stock components of the present disclosure can be prepared by the same process used to produce low viscosity polyalphaolefin ("PAO") base stocks. In particular, the low viscosity polyalphaolefin ("PAO") base stocks and the methyl paraffin lubricating oil base stock may be made by the metallocene catalyzed process or the two-step process described herein.
[0040] In a preferred embodiment, the first step involves oligomerizing low molecular weight linear alpha olefins in the presence of a single site catalyst and the second step involves oligomerization of at least a portion of the product from the first step in the presence of an oligomerization catalyst.
[0041] This invention is also directed to the PAO composition formed in the first oligomerization, wherein at least portions of the PAO have properties that make them highly desirable for subsequent oligomerization. A preferred process for the first oligomerization uses a single site catalyst at high temperatures without adding hydrogen to produce a low viscosity PAO with excellent Noack volatility at high conversion rates. This PAO comprises a dimer product with at least 25 wt % tri-substituted vinylene olefins wherein said dimer product is highly desirable as a feedstock for a subsequent oligomerization. This PAO also comprises trimer and optionally tetramer and higher oligomer products with outstanding properties that make these products useful as lubricant basestocks following hydrogenation. The hydrogenated trimer portion can be used as a methyl paraffin lubricating oil base stock component in the inventive lubricating oil base stock and engine oil compositions.
[0042] This invention also is directed to improved methyl paraffin lubricating oil base stock components characterized by very low viscosity and excellent Noack volatility that are obtained following the two-step process.
[0043] The methyl paraffin lubricating oil base stock components formed in the invention, both intermediate and final methyl paraffins, are liquids. For the purposes of this invention, a term "liquid" is defined to be a fluid that has no distinct melting point above 0.degree. C., preferably no distinct melting point above -10.degree. C., and has a kinematic viscosity at 100.degree. C. of 3000 cSt or less--though all of the liquid PAOs of the present invention have a kinematic viscosity at 100.degree. C. of 20 cSt or less as further disclosed.
[0044] When used in the present invention, in accordance with conventional terminology in the art, the following terms are defined for the sake of clarity. The term "vinyl" is used to designate groups of formula RCH.dbd.CH2. The term "vinylidene" is used to designate groups of formula RR'.dbd.CH2. The term "disubstituted vinylene" is used to designate groups of formula RCH.dbd.CHR'. The term "trisubstituted vinylene" is used to designate groups of formula RR' C.dbd.CHR''. The term "tetrasubstituted vinylene" is used to designate groups of formula RR'C.dbd.CR''R'''. For all of these formulas, R, R', R'', and R''' are alkyl groups which may be identical or different from each other.
[0045] The monomer feed used in both the first oligomerization and optionally contacted with the recycled intermediate PAO dimer and light olefin fractions in the subsequent oligomerization is at least one linear alpha olefin (LAO) typically comprised of monomers of 6 to 24 carbon atoms, usually 6 to 20, and preferably 6 to 14 carbon atoms, such as 1-hexene, 1-octene, 1-nonene, 1-decene, 1-dodecene, and 1-tetradecene. Olefins with even carbon numbers are preferred LAOS. Additionally, these olefins are preferably treated to remove catalyst poisons, such as peroxides, oxygen, sulfur, nitrogen-containing organic compounds, and/or acetylenic compounds as described in WO 2007/011973.
Catalyst
[0046] Useful catalysts in the first oligomerization include single site catalysts. In a preferred embodiment, the first oligomerization uses a metallocene catalyst. In this disclosure, the terms "metallocene catalyst" and "transition metal compound" are used interchangeably. Preferred classes of catalysts give high catalyst productivity and result in low product viscosity and low molecular weight. Useful metallocene catalysts may be bridged or un-bridged and substituted or un-substituted. They may have leaving groups including dihalides or dialkyls. When the leaving groups are dihalides, tri-alkylaluminum may be used to promote the reaction. In general, useful transition metal compounds may be represented by the following formula:
X.sub.1X.sub.2M.sub.1(CpCp*)M.sub.2X.sub.3X.sub.4
wherein: [0047] M.sub.1 is an optional bridging element, preferably selected from silicon or carbon; [0048] M.sub.2 is a Group 4 metal; [0049] Cp and Cp* are the same or different substituted or unsubstituted cyclopentadienyl ligand systems wherein, if substituted, the substitutions may be independent or linked to form multicyclic structures; [0050] X.sub.1 and X.sub.2 are independently hydrogen, hydride radicals, hydrocarbyl radicals, substituted hydrocarbyl radicals, silylcarbyl radicals, substituted silylcarbyl radicals, germylcarbyl radicals, or substituted germylcarbyl radicals or are preferably independently selected from hydrogen, branched or unbranched C.sub.1 to C.sub.20 hydrocarbyl radicals, or branched or unbranched substituted C.sub.1 to C.sub.20 hydrocarbyl radicals; and [0051] X.sub.3 and X.sub.4 are independently hydrogen, halogen, hydride radicals, hydrocarbyl radicals, substituted hydrocarbyl radicals, halocarbyl radicals, substituted halocarbyl radicals, silylcarbyl radicals, substituted silylcarbyl radicals, germylcarbyl radicals, or substituted germylcarbyl radicals; or both X.sub.3 and X.sub.4 are joined and bound to the metal atom to form a metallacycle ring containing from about 3 to about 20 carbon atoms, or are preferably independently selected from hydrogen, branched or unbranched C.sub.1 to C.sub.20 hydrocarbyl radicals, or branched or unbranched substituted C.sub.1 to C.sub.20 hydrocarbyl radicals.
[0052] For this disclosure, a hydrocarbyl radical is C1-C100 radical and may be linear, branched, or cyclic. A substituted hydrocarbyl radical includes halocarbyl radicals, substituted halocarbyl radicals, silylcarbyl radicals, and germylcarbyl radicals as these terms are defined below.
[0053] Substituted hydrocarbyl radicals are radicals in which at least one hydrogen atom has been substituted with at least one functional group such as NR*2, OR*, SeR*, TeR*, PR*2, AsR*2, SbR*2, SR*, BR*2, SiR*3, GeR*3, SnR*3, PbR*3 and the like or where at least one non-hydrocarbon atom or group has been inserted within the hydrocarbyl radical, such as --O--, --S--, --Se--, --Te--, --N(R*)--, .dbd.N--, --P(R*)--, .dbd.P--, --As(R*)--, .dbd.As--, --Sb(R*)--, .dbd.Sb--, --B(R*)--, .dbd.B--, --Si(R*)2-, --Ge(R*)2-, --Sn(R*)2-, --Pb(R*)2- and the like, where R* is independently a hydrocarbyl or halocarbyl radical, and two or more R* may join together to form a substituted or unsubstituted saturated, partially unsaturated or aromatic cyclic or polycyclic ring structure.
[0054] Halocarbyl radicals are radicals in which one or more hydrocarbyl hydrogen atoms have been substituted with at least one halogen (e.g., F, C1, Br, I) or halogen-containing group (e.g., CF3).
[0055] Substituted halocarbyl radicals are radicals in which at least one halocarbyl hydrogen or halogen atom has been substituted with at least one functional group such as NR*2, OR*, SeR*, TeR*, PR*2, AsR*2, SbR*2, SR*, BR*2, SiR*3, GeR*3, SnR*3, PbR*3 and the like or where at least one non-carbon atom or group has been inserted within the halocarbyl radical such as --O--, --S--, --Se--, --Te--, --N(R*)--, .dbd.N--, --P(R*)--, .dbd.P--, --As(R*)--, .dbd.As--, --Sb(R*)--, .dbd.Sb--, --B(R*)--, .dbd.B--, --Si(R*)2-, --Ge(R*)2-, --Sn(R*)2-, --Pb(R*)2- and the like, where R* is independently a hydrocarbyl or halocarbyl radical provided that at least one halogen atom remains on the original halocarbyl radical. Additionally, two or more R* may join together to form a substituted or unsubstituted saturated, partially unsaturated or aromatic cyclic or polycyclic ring structure.
[0056] Silylcarbyl radicals (also called silylcarbyls) are groups in which the silyl functionality is bonded directly to the indicated atom or atoms. Examples include SiH3, SiH2R*, SiHR*2, SiR*3, SiH2(OR*), SiH(OR*)2, Si(OR*)3, SiH2(NR*2), SiH(NR*2)2, Si(NR*2)3, and the like where R* is independently a hydrocarbyl or halocarbyl radical and two or more R* may join together to form a substituted or unsubstituted saturated, partially unsaturated or aromatic cyclic or polycyclic ring structure.
[0057] Germylcarbyl radicals (also called germylcarbyls) are groups in which the germyl functionality is bonded directly to the indicated atom or atoms. Examples include GeH3, GeH2R*, GeHR*2, GeR53, GeH2(OR*), GeH(OR*)2, Ge(OR*)3, GeH2(NR*2), GeH(NR*2)2, Ge(NR*2)3, and the like where R* is independently a hydrocarbyl or halocarbyl radical and two or more R* may join together to form a substituted or unsubstituted saturated, partially unsaturated or aromatic cyclic or polycyclic ring structure.
[0058] In an embodiment, the transition metal compound may be represented by the following formula:
X.sub.1X.sub.2M.sub.1(CpCp*)M.sub.2X.sub.3X.sub.4
wherein: [0059] M.sub.1 is a bridging element, and preferably silicon; [0060] M.sub.2 is a Group 4 metal, and preferably titanium, zirconium or hafnium; [0061] Cp and Cp* are the same or different substituted or unsubstituted indenyl or tetrahydroindenyl rings that are each bonded to both M.sub.1 and M.sub.2; [0062] X.sub.1 and X.sub.2 are independently hydrogen, hydride radicals, hydrocarbyl radicals, substituted hydrocarbyl radicals, silylcarbyl radicals, substituted silylcarbyl radicals, germylcarbyl radicals, or substituted germylcarbyl radicals; and [0063] X.sub.3 and X.sub.4 are independently hydrogen, halogen, hydride radicals, hydrocarbyl radicals, substituted hydrocarbyl radicals, halocarbyl radicals, substituted halocarbyl radicals, silylcarbyl radicals, substituted silylcarbyl radicals, germylcarbyl radicals, or substituted germylcarbyl radicals; or both X.sub.3 and X.sub.4 are joined and bound to the metal atom to form a metallacycle ring containing from about 3 to about 20 carbon atoms.
[0064] In using the terms "substituted or unsubstituted tetrahydroindenyl," "substituted or unsubstituted tetrahydroindenyl ligand," and the like, the substitution to the aforementioned ligand may be hydrocarbyl, substituted hydrocarbyl, halocarbyl, substituted halocarbyl, silylcarbyl, or germylcarbyl. The substitution may also be within the ring giving heteroindenyl ligands or heterotetrahydroindenyl ligands, either of which can additionally be substituted or unsubstituted.
[0065] In another embodiment, useful transition metal compounds may be represented by the following formula:
L.sup.AL.sup.BL.sup.C.sub.iMDE
wherein: [0066] L.sup.A is a substituted cyclopentadienyl or heterocyclopentadienyl ancillary ligand .pi.-bonded to M; [0067] L.sup.B is a member of the class of ancillary ligands defined for L.sup.A, or is J, a heteroatom ancillary ligand .sigma.-bonded to M; the L.sup.A and L.sup.B ligands may be covalently bridged together through a Group 14 element linking group; [0068] L.sup.C.sub.i is an optional neutral, non-oxidizing ligand having a dative bond to M (i equals 0 to 3); [0069] M is a Group 4 or 5 transition metal; and [0070] D and E are independently monoanionic labile ligands, each having a .pi.-bond to M, optionally bridged to each other or L.sup.A or L.sup.B. The mono-anionic ligands are displaceable by a suitable activator to permit insertion of a polymerizable monomer or a macromonomer can insert for coordination polymerization on the vacant coordination site of the transition metal compound.
[0071] One embodiment of this invention uses a highly active metallocene catalyst. In this embodiment, the catalyst productivity is greater than 15,000,
g PAO g catalyst , ##EQU00001##
preferably greater man 20,000
g PAO g catalyst , ##EQU00002##
preferably greater than 25,000
g PAO g catalyst , ##EQU00003##
and more preferably greater than 30,000
g PAO g catalyst , ##EQU00004##
wherein
g PAO g catalyst ##EQU00005##
represents grams of PAO formed per grams of catalyst used in the oligomerization reaction.
[0072] High productivity rates are also achieved. In an embodiment, the productivity rate in the first oligomerization is greater than 4,000
g PAO g catalyst * hour , ##EQU00006##
preferably greater than 6,000
g PAO g catalyst * hour , ##EQU00007##
preferably greater than 8,000
g PAO g catalyst * hour , ##EQU00008##
preferably greater than 10,000
g PAO g catalyst * hour , ##EQU00009##
wherein
g PAO g catalyst ##EQU00010##
represents grams of PAO formed per grams of catalyst used in the oligomerization reaction.
Activator
[0073] The catalyst may be activated by a commonly known activator such as non-coordinating anion (NCA) activator. An NCA is an anion which either does not coordinate to the catalyst metal cation or that coordinates only weakly to the metal cation. An NCA coordinates weakly enough that a neutral Lewis base, such as an olefinically or acetylenically unsaturated monomer, can displace it from the catalyst center. Any metal or metalloid that can form a compatible, weakly coordinating complex with the catalyst metal cation may be used or contained in the NCA. Suitable metals include, but are not limited to, aluminum, gold, and platinum. Suitable metalloids include, but are not limited to, boron, aluminum, phosphorus, and silicon.
[0074] Lewis acid and ionic activators may also be used. Useful but non-limiting examples of Lewis acid activators include triphenylboron, tris-perfluorophenylboron, tris-perfluorophenylaluminum, and the like. Useful but non-limiting examples of ionic activators include dimethylanilinium tetrakisperfluorophenylborate, triphenyl carb onium tetrakisperfluorophenylborate, dimethylanilinium tetrakisperfluorophenylaluminate, and the like.
[0075] An additional subclass of useful NCAs comprises stoichiometric activators, which can be either neutral or ionic. Examples of neutral stoichiometric activators include tri-substituted boron, tellurium, aluminum, gallium and indium or mixtures thereof. The three substituent groups are each independently selected from alkyls, alkenyls, halogen, substituted alkyls, aryls, arylhalides, alkoxy and halides. Preferably, the three groups are independently selected from halogen, mono or multicyclic (including halosubstituted) aryls, alkyls, and alkenyl compounds and mixtures thereof, preferred are alkenyl groups having 1 to 20 carbon atoms, alkyl groups having 1 to 20 carbon atoms, alkoxy groups having 1 to 20 carbon atoms and aryl groups having 3 to 20 carbon atoms (including substituted aryls). More preferably, the three groups are alkyls having 1 to 4 carbon groups, phenyl, naphthyl or mixtures thereof. Even more preferably, the three groups are halogenated, preferably fluorinated, aryl groups. Ionic stoichiometric activator compounds may contain an active proton, or some other cation associated with, but not coordinated to, or only loosely coordinated to, the remaining ion of the ionizing compound.
[0076] Ionic catalysts can be prepared by reacting a transition metal compound with an activator, such as B(C6F6)3, which upon reaction with the hydrolyzable ligand (X') of the transition metal compound forms an anion, such as ([B(C6F5)3(X')]--), which stabilizes the cationic transition metal species generated by the reaction. The catalysts can be, and preferably are, prepared with activator components which are ionic compounds or compositions. However preparation of activators utilizing neutral compounds is also contemplated by this invention.
[0077] Compounds useful as an activator component in the preparation of the ionic catalyst systems used in the process of this invention comprise a cation, which is preferably a Bronsted acid capable of donating a proton, and a compatible NCA which anion is relatively large (bulky), capable of stabilizing the active catalyst species which is formed when the two compounds are combined and said anion will be sufficiently labile to be displaced by olefinic diolefinic and acetylenically unsaturated substrates or other neutral Lewis bases such as ethers, nitriles and the like.
[0078] In an embodiment, the ionic stoichiometric activators include a cation and an anion component, and may be represented by the following formula:
(L**-H).sub.d.sup.+(A.sup.d-)
wherein: L** is an neutral Lewis base; H is hydrogen; (L**-H).sup.+ is a Bronsted acid or a reducible Lewis Acid; and A.sup.d- is an NCA having the charge d-, and d is an integer from 1 to 3.
[0079] The cation component, (L**-H).sub.d.sup.+ may include Bronsted acids such as protons or protonated Lewis bases or reducible Lewis acids capable of protonating or abstracting a moiety, such as an alkyl or aryl, from the catalyst after alkylation.
[0080] The activating cation (L**-H)d+ may be a Bronsted acid, capable of donating a proton to the alkylated transition metal catalytic precursor resulting in a transition metal cation, including ammoniums, oxoniums, phosphoniums, silyliums, and mixtures thereof, preferably ammoniums of methylamine, aniline, dimethylamine, diethylamine, N-methylaniline, diphenylamine, trimethylamine, triethylamine, N,N-dimethylaniline, methyldiphenylamine, pyridine, p-bromo N,N-dimethylaniline, p-nitro-N,N-dimethylaniline, phosphoniums from triethylphosphine, triphenylphosphine, and diphenylphosphine, oxomiuns from ethers such as dimethyl ether, diethyl ether, tetrahydrofuran and dioxane, sulfoniums from thioethers, such as diethyl thioethers and tetrahydrothiophene, and mixtures thereof. The activating cation (L**-H)d+ may also be a moiety such as silver, tropylium, carbeniums, ferroceniums and mixtures, preferably carboniums and ferroceniums; most preferably triphenyl carbonium. The anion component Ad--include those having the formula [Mk+Qn]d- wherein k is an integer from 1 to 3; n is an integer from 2-6; n-k=d; M is an element selected from Group 13 of the Periodic Table of the Elements, preferably boron or aluminum, and Q is independently a hydride, bridged or unbridged dialkylamido, halide, alkoxide, aryloxide, hydrocarbyl, substituted hydrocarbyl, halocarbyl, substituted halocarbyl, and halosubstituted-hydrocarbyl radicals, said Q having up to 20 carbon atoms with the proviso that in not more than one occurrence is Q a halide. Preferably, each Q is a fluorinated hydrocarbyl group having 1 to 20 carbon atoms, more preferably each Q is a fluorinated aryl group, and most preferably each Q is a pentafluoryl aryl group. Examples of suitable A.sup.d- also include diboron compounds as disclosed in U.S. Pat. No. 5,447,895, which is incorporated herein by reference.
[0081] Illustrative but non-limiting examples of boron compounds which may be used as an NCA activator in combination with a co-activator are tri-substituted ammonium salts such as: trimethylammonium tetraphenylborate, triethyl ammonium tetraphenylborate, tripropylammonium tetraphenylborate, tri(n-butyl)ammonium tetraphenylborate, tri(tert-butyl)ammonium tetraphenylborate, N,N-dimethylanilinium tetraphenylborate, N,N-diethylanilinium tetraphenylborate, N,N-dimethyl-(2,4,6-trimethylanilinium) tetraphenylborate, trimethylammonium tetrakis(pentafluorophenyl)borate, triethylammonium tetrakis(pentafluorophenyl)borate, tripropylammonium tetrakis(pentafluorophenyl)borate, tri(n-butyl)ammonium tetrakis(pentafluorophenyl)borate, tri(sec-butyl)ammonium tetrakis(pentafluorophenyl)borate, N,N-dimethylanilinium tetrakis(pentafluorophenyl)borate, N,N-diethylanilinium tetrakis(pentafluorophenyl)borate, N,N-dimethyl-(2,4,6-trimethylanilinium) tetrakis(pentafluorophenyl)borate, trimethylammonium tetrakis-(2,3,4,6-tetrafluorophenyl) borate, triethylammonium tetrakis-(2,3,4,6-tetrafluorophenyl)borate, tripropylammonium tetrakis-(2,3,4,6-tetrafluorophenyl)borate, tri(n-butyl)ammonium tetrakis-(2,3,4,6-tetrafluorophenyl)borate, dimethyl(tert-butyl)ammonium tetrakis-(2,3,4,6-tetrafluorophenyl)borate, N,N-dimethylanilinium tetrakis-(2,3,4,6-tetrafluorophenyl)borate, N,N-diethylanilinium tetrakis-(2,3,4,6-tetrafluorophenyl)borate, N,N-dimethyl-(2,4,6-trimethylanilinium) tetrakis-(2,3,4,6-tetrafluorophenyl)borate, trimethylammonium tetrakis(perfluoronaphthyl)borate, triethylammonium tetrakis(perfluoronaphthyl)borate, tripropylammonium tetrakis(perfluoronaphthyl)borate, tri(n-butyl)ammonium tetrakis(perfluoronaphthyl)borate, tri(tert-butyl)ammonium tetrakis(perfluoronaphthyl)borate, N,N-dimethylanilinium tetrakis(perfluoronaphthyl)borate, N,N-diethylanilinium tetrakis(perfluoronaphthyl)borate, N,N-dimethyl-(2,4,6-trimethylanilinium) tetrakis(perfluoronaphthyl)borate, trimethylammonium tetrakis(perfluorobiphenyl)borate, triethylammonium tetrakis(perfluorobiphenyl)borate, tripropylammonium tetrakis(perfluorobiphenyl)borate, tri(n-butyl)ammonium tetrakis(perfluorobiphenyl)borate, tri(tert-butyl)ammonium tetrakis(perfluorobiphenyl)borate, N,N-dimethylanilinium tetrakis(perfluorobiphenyl)borate, N,N-diethylanilinium tetrakis(perfluorobiphenyl)borate, N,N-dimethyl-(2,4,6-trimethylanilinium) tetrakis(perfluorobiphenyl)borate, trimethylammonium tetrakis(3,5-bis(trifluoromethyl)phenyl)borate, triethylammonium tetrakis(3,5-bis(trifluoromethyl)phenyl)borate, tripropylammonium tetrakis(3,5-bis(trifluoromethyl)phenyl)borate, tri(n-butyl)ammonium tetrakis(3,5-bis(trifluoromethyl)phenyl)borate, tri(tert-butyl)ammonium tetrakis(3,5-bis(trifluoromethyl)phenyl)borate, N,N-dimethylanilinium tetrakis(3,5-bis(trifluoromethyl)phenyl)borate, N,N-diethylanilinium tetrakis(3,5-bis(trifluoromethyl)phenyl)borate, N,N-dimethyl-(2,4,6-trimethylanilinium) tetrakis(3,5-bis(trifluoromethyl)phenyl)borate, and dialkyl ammonium salts such as: di-(iso-propyl)ammonium tetrakis(pentafluorophenyl)borate, and dicyclohexylammonium tetrakis(pentafluorophenyl)borate; and other salts such as tri(o-tolyl)phosphonium tetrakis(pentafluorophenyl)borate, tri(2,6-dimethylphenyl)phosphonium tetrakis(pentafluorophenyl)borate, tropillium tetraphenylborate, triphenylcarbenium tetraphenylborate, triphenylphosphonium tetraphenylborate, triethylsilylium tetraphenylborate, benzene(diazonium)tetraphenylborate, tropillium tetrakis(pentafluorophenyl)borate, triphenylcarbenium tetrakis(pentafluorophenyl)borate, triphenylphosphonium tetrakis(pentafluorophenyl)borate, triethyl silylium tetrakis(pentafluorophenyl)borate, benzene(diazonium) tetrakis(pentafluorophenyl)borate, tropillium tetrakis-(2,3,4,6-tetrafluorophenyl)borate, triphenylcarbenium tetrakis-(2,3,4,6-tetrafluorophenyl)borate, triphenylphosphonium tetrakis-(2,3,4,6-tetrafluorophenyl)borate, triethyl silylium tetrakis-(2,3,4,6-tetrafluorophenyl)borate, benzene(diazonium) tetrakis-(2,3,4,6-tetrafluorophenyl)borate, tropillium tetrakis(perfluoronaphthyl)borate, triphenylcarbenium tetrakis(perfluoronaphthyl)borate, triphenylphosphonium tetrakis(perfluoronaphthyl)borate, triethyl silylium tetrakis(perfluoronaphthyl)borate, benzene(diazonium) tetrakis(perfluoronaphthyl)borate, tropillium tetrakis(perfluorobiphenyl)borate, triphenylcarbenium tetrakis (perfluorobiphenyl)borate, triphenylphosphonium tetrakis(perfluorobiphenyl)borate, triethyl silylium tetrakis(perfluorobiphenyl)borate, benzene(diazonium) tetrakis(perfluorobiphenyl)borate, tropillium tetrakis(3,5-bis(trifluoromethyl)phenyl)borate, triphenylcarbenium tetrakis(3,5-bis(trifluoromethyl)phenyl)borate, triphenylphosphonium tetrakis(3,5-bis(trifluoromethyl)phenyl)borate, triethyl silylium tetrakis(3,5-bis (trifluoromethyl)phenyl)borate, and benzene(diazonium) tetrakis(3,5-bis(trifluoromethyl)phenyl)borate.
[0082] In an embodiment, the NCA activator, (L**-H)d.sup.+ (A.sup.d-), is N,N-dimethylanilinium tetrakis(perfluorophenyl)borate, N,N-dimethylanilinium tetrakis(perfluoronaphthyl)borate, N,N-dimethyl anilinium tetrakis(perfluorobiphenyl)borate, N,N-dimethylanilinium tetrakis(3,5-bis(trifluoromethyl)phenyl)borate, triphenylcarbenium tetrakis (perfluoronaphthyl)borate, triphenylcarbenium tetrakis (perfluorobiphenyl)borate, triphenylcarbenium tetrakis(3,5-bis(trifluoromethyl)phenyl)borate, or triphenylcarbenium tetra(perfluorophenyl)borate.
[0083] Pehlert et al., U.S. Pat. No. 7,511,104 provides additional details on NCA activators that may be useful in this invention, and these details are hereby fully incorporated by reference.
[0084] Additional activators that may be used include alumoxanes or alumoxanes in combination with an NCA. In one embodiment, alumoxane activators are utilized as an activator. Alumoxanes are generally oligomeric compounds containing --Al(R1)-O-- sub-units, where R1 is an alkyl group. Examples of alumoxanes include methylalumoxane (MAO), modified methylalumoxane (MMAO), ethylalumoxane and isobutylalumoxane. Alkylalumoxanes and modified alkylalumoxanes are suitable as catalyst activators, particularly when the abstractable ligand is an alkyl, halide, alkoxide or amide. Mixtures of different alumoxanes and modified alumoxanes may also be used.
[0085] A catalyst co-activator is a compound capable of alkylating the catalyst, such that when used in combination with an activator, an active catalyst is formed. Co-activators may include alumoxanes such as methylalumoxane, modified alumoxanes such as modified methylalumoxane, and aluminum alkyls such trimethylaluminum, tri-isobutylaluminum, triethylaluminum, and tri-isopropylaluminum, tri-n-hexylaluminum, tri-n-octylaluminum, tri-n-decylaluminum or tri-n-dodecylaluminum. Co-activators are typically used in combination with Lewis acid activators and ionic activators when the catalyst is not a dihydrocarbyl or dihydride complex. Preferred activators are non-oxygen containing compounds such as the aluminum alkyls, and are preferably tri-alkylaluminums.
[0086] The co-activator may also be used as a scavenger to deactivate impurities in feed or reactors. A scavenger is a compound that is sufficiently Lewis acidic to coordinate with polar contaminates and impurities adventitiously occurring in the polymerization feedstocks or reaction medium. Such impurities can be inadvertently introduced with any of the reaction components, and adversely affect catalyst activity and stability. Useful scavenging compounds may be organometallic compounds such as triethyl aluminum, triethyl borane, tri-isobutyl aluminum, methylalumoxane, isobutyl aluminumoxane, tri-n-hexyl aluminum, tri-n-octyl aluminum, and those having bulky substituents covalently bound to the metal or metalloid center being preferred to minimize adverse interaction with the active catalyst. Other useful scavenger compounds may include those mentioned in U.S. Pat. No. 5,241,025, EP-A 0426638, and WO 97/22635, which are hereby incorporated by reference for such details.
[0087] The reaction time or reactor residence time is usually dependent on the type of catalyst used, the amount of catalyst used, and the desired conversion level. Different transition metal compounds (also referred to as metallocene) have different activities. High amount of catalyst loading tends to gives high conversion at short reaction time. However, high amount of catalyst usage make the production process uneconomical and difficult to manage the reaction heat or to control the reaction temperature. Therefore, it is useful to choose a catalyst with maximum catalyst productivity to minimize the amount of metallocene and the amount of activators needed. For the preferred catalyst system of metallocene plus a Lewis Acid or an ionic promoter with NCA component, the transition metal compound use is typically in the range of 0.01 microgram to 500 micrograms of metallocene component/gram of alpha-olefin feed. Usually the preferred range is from 0.1 microgram to 100 microgram of metallocene component per gram of alpha-olefin feed. Furthermore, the molar ratio of the NCA activator to metallocene is in the range from 0.1 to 10, preferably 0.5 to 5, preferably 0.5 to 3. For the co-activators of alkylaluminums, the molar ratio of the co-activator to metallocene is in the range from 1 to 1000, preferably 2 to 500, preferably 4 to 400.
[0088] In selecting oligomerization conditions, to obtain the desired first reactor effluent, the system uses the transition metal compound (also referred to as the catalyst), activator, and co-activator.
[0089] US 2007/0043248 and US 2010/029242 provides additional details of metallocene catalysts, activators, co-activators, and appropriate ratios of such compounds in the feedstock that may be useful in this invention, and these additional details are hereby incorporated by reference.
Oligomerization Process
[0090] Many oligomerization processes and reactor types used for single site- or metallocene-catalyzed oligomerizations such as solution, slurry, and bulk oligomerization processes may be used in this invention. In some embodiments, if a solid catalyst is used, a slurry or continuous fixed bed or plug flow process is suitable. In a preferred embodiment, the monomers are contacted with the metallocene compound and the activator in the solution phase, bulk phase, or slurry phase, preferably in a continuous stirred tank reactor or a continuous tubular reactor. In a preferred embodiment, the temperature in any reactor used herein is from -10.degree. C. to 250.degree. C., preferably from 30.degree. C. to 220.degree. C., preferably from 50.degree. C. to 180.degree. C., preferably from 80.degree. C. to 150.degree. C. In a preferred embodiment, the pressure in any reactor used herein is from 10.13 to 10132.5 kPa (0.1 to 100 atm/1.5 to 1500 psi), preferably from 50.66 to 7600 kPa (0.5 to 75 atm/8 to 1125 psi), and most preferably from 101.3 to 5066.25 kPa (1 to 50 atm/15 to 750 psi). In another embodiment, the pressure in any reactor used herein is from 101.3 to 5,066,250 kPa (1 to 50,000 atm), preferably 101.3 to 2,533,125 kPa (1 to 25,000 atm). In another embodiment, the residence time in any reactor is 1 second to 100 hours, preferably 30 seconds to 50 hours, preferably 2 minutes to 6 hours, preferably 1 to 6 hours. In another embodiment, solvent or diluent is present in the reactor. These solvents or diluents are usually pre-treated in same manners as the feed olefins.
[0091] The oligomerization can be run in batch mode, where all the components are added into a reactor and allowed to react to a degree of conversion, either partial or full conversion. Subsequently, the catalyst is deactivated by any possible means, such as exposure to air or water, or by addition of alcohols or solvents containing deactivating agents. The oligomerization can also be carried out in a semi-continuous operation, where feeds and catalyst system components are continuously and simultaneously added to the reactor so as to maintain a constant ratio of catalyst system components to feed olefin(s). When all feeds and catalyst components are added, the reaction is allowed to proceed to a pre-determined stage. The reaction is then discontinued by catalyst deactivation in the same manner as described for batch operation. The oligomerization can also be carried out in a continuous operation, where feeds and catalyst system components are continuously and simultaneously added to the reactor so to maintain a constant ratio of catalyst system and feeds. The reaction product is continuously withdrawn from the reactor, as in a typical continuous stirred tank reactor (CSTR) operation. The residence times of the reactants are controlled by a pre-determined degree of conversion. The withdrawn product is then typically quenched in the separate reactor in a similar manner as other operation. In a preferred embodiment, any of the processes to prepare PAOs described herein are continuous processes.
[0092] A production facility may have one single reactor or several reactors arranged in series or in parallel, or both, to maximize productivity, product properties, and general process efficiency. The catalyst, activator, and co-activator may be delivered as a solution or slurry in a solvent or in the LAO feed stream, either separately to the reactor, activated in-line just prior to the reactor, or pre-activated and pumped as an activated solution or slurry to the reactor. Oligomerizations are carried out in either single reactor operation, in which the monomer, or several monomers, catalyst/activator/co-activator, optional scavenger, and optional modifiers are added continuously to a single reactor or in series reactor operation, in which the above components are added to each of two or more reactors connected in series. The catalyst components can be added to the first reactor in the series. The catalyst component may also be added to both reactors, with one component being added to first reaction and another component to other reactors.
[0093] The reactors and associated equipment are usually pre-treated to ensure proper reaction rates and catalyst performance. The reaction is usually conducted under inert atmosphere, where the catalyst system and feed components will not be in contact with any catalyst deactivator or poison which is usually polar oxygen, nitrogen, sulfur or acetylenic compounds. Additionally, in one embodiment of any of the process described herein, the feed olefins and or solvents are treated to remove catalyst poisons, such as peroxides, oxygen or nitrogen-containing organic compounds or acetylenic compounds. Such treatment will increase catalyst productivity 2- to 10-fold or more.
[0094] The reaction time or reactor residence time is usually dependent on the type of catalyst used, the amount of catalyst used, and the desired conversion level. When the catalyst is a metallocene, different metallocenes have different activities. Usually, a higher degree of alkyl substitution on the cyclopentadienyl ring, or bridging improves catalyst productivity. High catalyst loading tends to gives high conversion in short reaction time. However, high catalyst usage makes the process uneconomical and difficult to manage the reaction heat or to control the reaction temperature. Therefore, it is useful to choose a catalyst with maximum catalyst productivity to minimize the amount of metallocene and the amount of activators needed.
[0095] US 2007/0043248 and US 2010/0292424 provide significant additional details on acceptable oligomerization processes using metallocene catalysts, and the details of these processes, process conditions, catalysts, activators, co-activators, etc. are hereby incorporated by reference to the extent that they are not inconsistent with anything described in this disclosure.
[0096] Due to the low activity of some metallocene catalysts at high temperatures, low viscosity PAOs are typically oligomerized in the presence of added hydrogen at lower temperatures. The advantage is that hydrogen acts as a chain terminator, effectively decreasing molecular weight and viscosity of the PAO. Hydrogen can also hydrogenate the olefin, however, saturating the LAO feedstock and PAO. This would prevent LAO or the PAO dimer from being usefully recycled or used as feedstock into a further oligomerization process. Thus it is an improvement over prior art to be able to make an intermediate PAO without having to add hydrogen for chain termination because the unreacted LAO feedstock and intermediate PAO dimer maintain their unsaturation, and thus their reactivity, for a subsequent recycle step or use as a feedstock in a further oligomerization process.
[0097] The intermediate PAO produced is a mixture of dimers, trimers, and optionally tetramer and higher oligomers of the respective alpha olefin feedstocks. This intermediate PAO and portions thereof is referred to interchangeably as the "first reactor effluent" from which unreacted monomers have optionally been removed. In an embodiment, the dimer portion of the intermediate PAO may be a reactor effluent that has not been subject to a distillation process. In another embodiment, the dimer portion of the intermediate PAO may be subjected to a distillation process to separate it from the trimer and optional higher oligomer portion prior to feeding the at least dimer portion of the first reactor to a second reactor. In another embodiment, the dimer portion of the intermediate PAO may be a distillate effluent. In another embodiment, the at least dimer portion of the intermediate PAO is fed directly into the second reactor. In a further embodiment, the trimer portion of the intermediate PAO and the tetramer and higher oligomer portion of the intermediate PAO can be isolated from the first effluent by distillation. In another embodiment, the intermediate PAO is not subjected to a separate isomerization process following oligomerization.
[0098] In the invention, the intermediate PAO product has a kinematic viscosity at 100.degree. C. (KV100) of less than 20 cSt, preferably less than 15 cSt, preferably less than 12 cSt, more preferably less than 10 cSt. In the invention, the intermediate PAO trimer portion after a hydrogenation step has a KV100 of less than 4 cSt, preferably less than 3.6 cSt. In an embodiment, the tetramers and higher oligomer portion of the intermediate PAO after a hydrogenation step has a KV100 of less than 30 cSt. In an embodiment, the intermediate PAO oligomer portion remaining after the intermediate PAO dimer portion is removed has a KV100 of less than 25 cSt.
[0099] The intermediate PAO trimer portion has a VI of greater than 125, preferably greater than 130. In an embodiment, the trimer and higher oligomer portion of the intermediate PAO has a VI of greater than 130, preferably greater than 135. In an embodiment, the tetramer and higher oligomer portion of the intermediate PAO has a VI of greater than 150, preferably greater than 155.
[0100] The intermediate PAO trimer portion has a Noack volatility that is less than 15 wt %, preferably less than 14 wt %, preferably less than 13 wt %, preferably less than 12 wt %. In an embodiment, the intermediate PAO tetramers and higher oligomer portion has a Noack volatility that is less than 8 wt %, preferably less than 7 wt %, preferably less than 6 wt %.
[0101] The intermediate PAO dimer portion has a number average molecular weight in the range of 120 to 600.
[0102] The intermediate PAO dimer portion possesses at least one carbon-carbon unsaturated double bond. A portion of this intermediate PAO dimer comprises tri-substituted vinylene. This tri-substituted vinylene has two possible isomer structures that may coexist and differ regarding where the unsaturated double bond is located, as represented by the following structure:
##STR00005##
wherein the dashed line represents the two possible locations where the unsaturated double bond may be located and Rx and Ry are independently selected from a C.sub.3 to C.sub.21 alkyl group, preferably from linear C.sub.3 to C.sub.21 alkyl group.
[0103] In any embodiment, the intermediate PAO dimer contains greater than 20 wt %, preferably greater than 25 wt %, preferably greater than 30 wt %, preferably greater than 40 wt %, preferably greater than 50 wt %, preferably greater than 60 wt %, preferably greater than 70 wt %, preferably greater than 80 wt % of tri-substituted vinylene olefins represented by the general structure above.
[0104] In a preferred embodiment, Rx and Ry are independently C3 to C11 alkyl groups. In a preferred embodiment, Rx and Ry are both C7. In a preferred embodiment, the intermediate PAO dimer comprises a portion of tri-substituted vinylene dimer that is represented by the following structure:
##STR00006##
wherein the dashed line represents the two possible locations where the unsaturated double bond may be located.
[0105] In any embodiment, the intermediate PAO contains less than 70 wt %, preferably less than 60 wt %, preferably less than 50 wt %, preferably less than 40 wt %, preferably less than 30 wt %, preferably less than 20 wt % of di-substituted vinylidene represented by the formula:
RqRzC.dbd.CH2
wherein Rq and Rz are independently selected from alkyl groups, preferably linear alkyl groups, or preferably C3 to C21 linear alkyl groups.
[0106] One embodiment of the first oligomerization is illustrated and explained below as a non-limiting example. First, the following reactions show alkylation of a metallocene catalyst with tri n-octyl aluminum followed by activation of the catalyst with N,N-Dimethylanilinium tetrakis (penta-fluorophenyl) borate (1-):
[0107] Catalyst Alkylation
##STR00007##
[0108] Catalyst Activation
##STR00008##
[0109] Following catalyst activation, a 1,2 insertion process may take place as shown below:
##STR00009##
[0110] Both vinyl and vinylidene chain ends may be formed as a result of elimination from 1,2 terminated chains, as shown below. This chain termination mechanism shown below competes with propagation during this reaction phase.
##STR00010##
[0111] Alternatively following catalyst activation, a 2,1 insertion process may take place as shown below:
##STR00011##
[0112] Elimination is favored over propagation after 2,1 insertions due to the proximity of the alpha alkyl branch to the active center (see the area identified with the letter "A" in the reaction above). In other words, the more crowded active site hinders propagation and enhances elimination. 2,1 insertions are detected by nuclear magnetic resonance (NMR) using signals from the unique methylene-methylene unit (see the area identified with the letter "B" in the reaction above).
[0113] Certain metallocene catalysts result in a higher occurrence of 2,1 insertions, and elimination from 2,1 terminated chains preferentially forms vinylene chain ends, as shown below.
##STR00012##
Subsequent Oligomerization
[0114] The intermediate PAO dimer from the first oligomerization may be used as the sole olefin feedstock to the subsequent oligomerization or it may be used together with an alpha olefin feedstock of the type used as the olefin starting material for the first oligomerization. Other portions of the effluent from the first oligomerization may also be used as a feedstock to the subsequent oligomerization, including unreacted LAO. The intermediate PAO dimer may suitably be separated from the overall intermediate PAO product by distillation, with the cut point set at a value dependent upon the fraction to be used as lube base stock or the fraction to be used as feed for the subsequent oligomerization. Alpha olefins with the same attributes as those preferred for the first oligomerization are preferred for the subsequent oligomerization. Typically ratios for the intermediate PAO dimer fraction to the alpha olefins fraction in the feedstock are from 90:10 to 10:90 and more usually 80:20 to 20:80 by weight. But preferably the intermediate PAO dimer will make up around 50 mole % of the olefinic feed material since the properties and distribution of the final product, dependent in part upon the starting material, are favorably affected by feeding the intermediate PAO dimer at an equimolar ratio with the alpha olefins. Temperatures for the subsequent oligomerization in the second reactor range from 15 to 60.degree. C.
[0115] Any oligomerization process and catalyst may be used for the subsequent oligomerization. A preferred catalyst for the subsequent oligomerization is a non-transition metal catalyst, and preferably a Lewis acid catalyst. Patent applications US 2009/0156874 and US 2009/0240012 describe a preferred process for the subsequent oligomerization, to which reference is made for details of feedstocks, compositions, catalysts and co-catalysts, and process conditions. The Lewis acid catalysts of US 2009/0156874 and US 2009/0240012 include the metal and metalloid halides conventionally used as Friedel-Crafts catalysts, examples include AlCl.sub.3, BF.sub.3, AlBr.sub.3, TiCl.sub.3, and TiCl.sub.4 either alone or with a protic promoter/activator. Boron trifluoride is commonly used but not particularly suitable unless it is used with a protic promoter. Useful co-catalysts are well known and described in detail in US 2009/0156874 and US 2009/0240012. Solid Lewis acid catalysts, such as synthetic or natural zeolites, acid clays, polymeric acidic resins, amorphous solid catalysts such as silica-alumina, and heteropoly acids such as the tungsten zirconates, tungsten molybdates, tungsten vanadates, phosphotungstates and molybdotungstovanadogermanates (e.g., WOx/ZrO.sub.2, WOx/MoO.sub.3) may also be used although these are not generally as favored economically. Additional process conditions and other details are described in detail in US 2009/0156874 and US 2009/0240012, and incorporated herein by reference.
[0116] In a preferred embodiment, the subsequent oligomerization occurs in the presence of BF.sub.3 and at least two different activators selected from alcohols and alkyl acetates. The alcohols are C.sub.1 to C.sub.10 alcohols and the alkyl acetates are C.sub.1 to C.sub.10 alkyl acetates. Preferably, both co-activators are C.sub.1 to C.sub.6 based compounds. Two most preferred combination of co-activators are i) ethanol and ethyl acetate and ii) n-butanol and n-butyl acetate. The ratio of alcohol to alkyl acetate range from 0.2 to 15, or preferably 0.5 to 7.
[0117] The structure of the invented intermediate PAO is such that, when reacted in a subsequent oligomerization, the intermediate PAO reacts preferentially with the optional LAO to form a co-dimer of the dimer and LAO at high yields. This allows for high conversion and yield rates of the desired PAO products. In an embodiment, the PAO product from the subsequent oligomerization comprises primarily a co-dimer of the dimer and the respective LAO feedstock. In an embodiment, where the LAO feedstock for both oligomerization steps is 1-decene, the incorporation of intermediate C.sub.20 PAO dimer into higher oligomers is greater than 80%, the conversion of the LAO is greater than 95%, and the yield % of C30 product in the overall product mix is greater than 75%. In another embodiment, where the LAO feedstock is 1-octene, the incorporation of the intermediate PAO dimer into higher oligomers is greater than 85%, the conversion of the LAO is greater than 90%, and the yield % of C28 product in the overall product mix is greater than 70%. In another embodiment, where the feedstock is 1-dodecene, the incorporation of the intermediate PAO dimer into higher oligomers is greater than 90%, the conversion of the LAO is greater than 75%, and the yield % of C32 product in the overall product mix is greater than 70%.
[0118] In an embodiment, the monomer is optional as a feedstock in the second reactor. In another embodiment, the first reactor effluent comprises unreacted monomer, and the unreacted monomer is fed to the second reactor. In another embodiment, monomer is fed into the second reactor, and the monomer is an LAO selected from the group including 1-hexene, 1-octene, 1-nonene, 1-decene, 1-dodecene, and 1-tetradecene. In another embodiment, the PAO produced in the subsequent oligomerization is derived from the intermediate PAO dimer plus only one monomer. In another embodiment, the PAO produced in the subsequent oligomerization is derived from the intermediate PAO dimer plus two or more monomers, or three or more monomers, or four or more monomers, or even five or more monomers. For example, the intermediate PAO dimer plus a C.sub.8, C.sub.10, C.sub.12-LAO mixture, or a C.sub.6, C.sub.7, C.sub.8, C.sub.9, C.sub.10, C.sub.11, C.sub.12, C.sub.13, C.sub.14-LAO mixture, or a C.sub.4, C.sub.6, C.sub.8, C.sub.10, C.sub.12, C.sub.14, C.sub.16, C.sub.18-LAO mixture can be used as a feed. In another embodiment, the PAO produced in the subsequent oligomerization comprises less than 30 mole % of C2, C3 and C4 monomers, preferably less than 20 mole %, preferably less than 10 mole %, preferably less than 5 mole %, preferably less than 3 mole %, and preferably 0 mole %. Specifically, in another embodiment, the PAO produced in the subsequent oligomerization comprises less than 30 mole % of ethylene, propylene and butene, preferably less than 20 mole %, preferably less than 10 mole %, preferably less than 5 mole %, preferably less than 3 mole %, preferably 0 mole %.
[0119] The PAOs produced in the subsequent oligomerization may be a mixture of dimers, trimers, and optionally tetramer and higher oligomers. This PAO is referred to interchangeably as the "second reactor effluent" from which unreacted monomer may be optionally removed and recycled back to the second reactor. The desirable properties of the intermediate PAO dimer enable a high yield of a co-dimer of intermediate PAO dimer and LAO in the second reactor effluent. The PAOs in the second reactor effluent are especially notable because very low viscosity PAOs are achieved at very high yields and these PAOs have excellent rheological properties, including low pour point, outstanding Noack volatility, and very high viscosity indexes.
[0120] In an embodiment, this PAO may contain trace amounts of transition metal compound if the catalyst in the intermediate or subsequent oligomerization is a metallocene catalyst. A trace amount of transition metal compound is defined for purposes of this disclosure as any amount of transition metal compound or Group 4 metal present in the PAO. Presence of Group 4 metal may be detected at the ppm or ppb level by ASTM 5185 or other methods known in the art.
[0121] Preferably, the second reactor effluent PAO has a portion having a carbon count of C28-C32, wherein the C28-C32 portion is at least 65 wt %, preferably at least 70 wt %, preferably at least 75 wt %, more preferably at least 80 wt % of the second reactor effluent.
[0122] The kinematic viscosity at 100.degree. C. of the PAO is less than 10 cSt, preferably less than 6 cSt, preferably less than 4.5 cSt, preferably less than 3.2 cSt, or preferably in the range of 2.8 to 4.5 cSt. The kinematic viscosity at 100.degree. C. of the C28 portion of the PAO is less than 3.2 cSt. In an embodiment, the kinematic viscosity at 100.degree. C. of the C28 to C32 portion of the PAO is less than 10 cSt, preferably less than 6 cSt, preferably less than 4.5 cSt, and preferably in the range of 2.8 to 4.5 cSt.
[0123] In an embodiment, the pour point of the PAO is below -40.degree. C., preferably below -50.degree. C., preferably below -60.degree. C., preferably below -70.degree. C., or preferably below -80.degree. C. The pour point of the C28 to C32 portion of the PAO is below -30.degree. C., preferably below -40.degree. C., preferably below -50.degree. C., preferably below -60.degree. C., preferably below -70.degree. C., or preferably below -80.degree. C.
[0124] The Noack volatility of the PAO is not more than 9.0 wt %, preferably not more than 8.5 wt %, preferably not more than 8.0 wt %, or preferably not more than 7.5 wt %. The Noack volatility of the C28 to C32 portion of the PAO is less than 19 wt %, preferably less than 14 wt %, preferably less than 12 wt %, preferably less than 10 wt %, or more preferably less than 9 wt %.
[0125] The viscosity index of the PAO is more than 121, preferably more than 125, preferably more than 130, or preferably more than 136. The viscosity index of the trimer or C28 to C32 portion of the PAO is above 120, preferably above 125, preferably above 130, or more preferably at least 135.
[0126] The cold crank simulator value (CCS) at -25.degree. C. of the PAO or a portion of the PAO is not more than 500 cP, preferably not more than 450 cP, preferably not more than 350 cP, preferably not more than 250 cP, preferably in the range of 200 to 450 cP, or preferably in the range of 100 to 250 cP.
[0127] In an embodiment, the PAO has a kinematic viscosity at 100.degree. C. of not more than 3.2 cSt and a Noack volatility of not more than 19 wt %. In another embodiment, the PAO has a kinematic viscosity at 100.degree. C. of not more than 4.1 cSt and a Noack volatility of not more than 9 wt %.
[0128] The ability to achieve such low viscosity PAOs with such low Noack volatility at such high yields is especially remarkable, and highly attributable to the intermediate PAO tri-substituted vinylene dimer having properties that make it especially desirable in the subsequent oligomerization process.
[0129] The overall reaction scheme enabled by the present invention may be represented as shown below, starting from the original LAO feed and passing through the intermediate PAO dimer used as the feed for the subsequent oligomerization.
##STR00013##
[0130] The lube range oligomer product from the subsequent oligomerization is desirably hydrogenated prior to use as a lubricant basestock to remove any residual unsaturation and stabilize the product. Optional hydrogenation may be carried out in the manner conventional to the hydrotreating of conventional PAOs. Prior to any hydrogenation, the PAO is comprised of at least 10 wt % of tetra-substituted olefins; as determined via carbon NMR (described later herein); in other embodiments, the amount of tetra-substitution is at least 15 wt %, or at least 20 wt % as determined by carbon NMR. The tetra-substituted olefin has the following structure:
##STR00014##
Additionally, prior to any hydrogenation, the PAO is comprised of at least 60 wt % tri-substituted olefins, preferably at least 70 wt % tri-substituted olefins.
[0131] The intermediate PAOs and second reactor PAOs produced, particularly those of ultra-low viscosity, are especially suitable for high performance automotive engine oil formulations either by themselves or by blending with other fluids, such as Group II, Group II+, Group III, Group III+ or lube basestocks derived from hydroisomerization of wax fractions from Fisher-Tropsch hydrocarbon synthesis from CO/H2 syn gas, or other Group IV or Group V basestocks. They are also preferred grades for high performance industrial oil formulations that call for ultra-low and low viscosity oils. Additionally, they are also suitable for use in personal care applications, such as soaps, detergents, creams, lotions, sticks, shampoos, detergents, etc.
Lubricating Oils
[0132] The methyl paraffin lubricating oil base stock blends of the instant disclosure described above may be formulated into lubricating oils by the addition of a minor amount of one or more lubricating oil additives to a major amount of the methyl paraffin lubricating oil base stock blend. The methyl paraffin lubricating oil base stock can be present as the major base stock in the lubricating oils of this disclosure. Accordingly, the methyl paraffin lubricating oil base stock can be present in an amount from 55 to 99 wt %, or 60 to 97 wt %, or 65 to 95 wt %, or 70 to 90 wt %, or 75 to 85 wt %, or 77 to 83 wt % of the fully formulated lubricating oil. The one or more lubricating oil additives are present as a minor amount of the lubricating oils of this disclosure. Accordingly, the lubricating oil additives can be present in an amount from 1 to 45 wt %, or 3 to 40 wt %, or 5 to 35 wt %, or 10 to 30 wt %, or 15 to 25 wt %, or 17 to 23 wt % of the fully formulated lubricating oil. The lubricating oils include a major amount of the methyl paraffin lubricating oil base stock blend
[0133] The methyl paraffin lubricating oils of the present disclosure have a viscosity (Kv.sub.100) from 2.0 to 6.0 cSt, or 2.5 to 5.5 cSt, or 3.0 to 5.0 cSt, or 3.5 to 4.5 cSt, or 3.7 to 4.3 cSt at 100.degree. C., as determined by ASTM D445 or ASTM D7042. The methyl paraffin lubricating oils of the instant disclosure have a viscosity (K.sub.40) from 5.0 cSt to 25.0 cSt, or 7.0 to 23.0 cSt, or 9.0 to 21.0 cSt, or 10.0 to 20.0 cSt, or 11.0 to 19.0 cSt, or 12.0 to 18.0 cSt, or 13.0 to 17.0 cSt, or 14.0 to 16.0 cSt, as determined by ASTM D445 or ASTM D7042.
[0134] The methyl paraffin lubricating oils of the present disclosure have a high temperature high shear (HTHS) viscosity at 150 deg. C of less than about 2.3 cP, or less than about 2.2 cP, or less than about 2.1 cP, or less than about 2.0 cP, or less than about 1.9 cP, or less than about 1.8 cP, or less than about 1.7 cP, or less than 1.6 cP, or less than 1.5 cP, or less than 1.4 cP, or less than 1.3 cP, or less than 1.2 cP, or less than 1.1 cP, or less than 1.0 cP, as determined by ASTM D4683.
[0135] The methyl paraffin lubricating oils of the present disclosure may have a Noack volatility at 250.degree. C. from about 10 to about 90 percent as determined by ASTM D5800. Alternatively, the methyl paraffin lubricating oils may have a Noack volatility at 250.degree. C. of from 15 to 85%, or 20 to 80%, or 25 to 75%, or 30 to 70%, or 35 to 65%, or 40 to 60%, or 45 to 55% as determined by ASTM D5800.
[0136] The viscosity-temperature relationship of a lubricating oil is one of the critical criteria which must be considered when selecting a lubricant for a particular application. Viscosity Index (VI) is an empirical, unitless number which indicates the rate of change in the viscosity of an oil within a given temperature range. Fluids exhibiting a relatively large change in viscosity with temperature are said to have a low viscosity index. A low VI oil, for example, will thin out at elevated temperatures faster than a high VI oil. Usually, the high VI oil is more desirable because it has higher viscosity at higher temperature, which translates into better or thicker lubrication film and better protection of the contacting machine elements.
[0137] The methyl paraffin lubricating oils of the present disclosure may have a Viscosity Index from about 100 to about 200 as determined by ASTM D2270. Alternatively, the methyl paraffin lubricating oils may have a Viscosity Index of from 100 to 190, or 120 to 180, or 130 to 170, or 140 to 160, or 145 to 155, as determined by ASTM D2270.
[0138] Surprisingly, as shown in the Examples herein, it has been discovered that formulated engine oils employing these methyl paraffin base stock blends as a major component possess unexpectedly high thermal and oxidative stability along with good deposit control benefit, as confirmed by their consistently low deposit formation from the Thermo-Oxidation Engine Oil Simulation Test (TEOST 33C ASTM D6335) conducted at high temperature.
[0139] The methyl paraffin lubricating oils of the present disclosure in particular yield a synergistic improvement in oxidative stability relative to the oxidative stabilities of the C10 dimer and C10 trimer that are incorporated into the methyl paraffin base stock blend. In particular, the methyl paraffin lubricating oils of the present disclosure have an oxidation stability (210 hour test with time to 200% KV40 increase) of from 60 to 150 hours, or 70 to 140 hours, or 80 to 130 hours, or 90 to 120 hours, or 100 to 110 hours.
[0140] The methyl paraffin lubricating oils of the present disclosure also provide for outstanding resistance to deposit formation as measured by the TEOST 33C test for 2 hours at 200 to 480 deg. C per ASTM D6335. In particular, the methyl paraffin lubricating oils of the present disclosure yield a synergistic improvement in deposit formation relative to the deposit formation of the C10 dimer and C10 trimer that are incorporated into the methyl paraffin base stock blend. The methyl paraffin lubricating oils of the present disclosure provide total deposits ranging from 10 to 30 mg, or 12 to 28 mg, or 14 to 26 mg, or 16 to 24 mg, or 18 to 22 mg, or 19 to 21 mg.
[0141] The methyl paraffin lubricating oils of the instant disclosure may have a MTM average traction coefficient (at 100 deg. C, 1 GPa, 2 m/s and 0-100% SRR) ranging from 0.0050 to 0.0090. Alternatively, the methyl paraffin lubricating oils may have a MTM average traction coefficient ranging from 0.0055 to 0.0085, or 0.0060 to 0.0080, or 0.0065 to 0.0075. The MTM average traction coefficient correlates with fuel efficiency with lower values providing improved fuel economy. The methyl paraffin lubricating oils of the instant disclosure have MTM average traction coefficient values that are from 20 to 180% lower, or from 40 to 120% lower, or from 60 to 100% lower, or from 70 to 90% lower, or from 75 to 85% lower than conventional lubricating oils utilizing Group II, Group III or Group III (GTL) base stocks as the major component of the lubricating oil.
[0142] The methyl paraffin lubricating oils of the instant disclosure may have a pour point of from about -10 to -60.degree. C. as determined by ASTM D5950. Alternatively, the methyl paraffin lubricating oils may have a pour point of from -15 to -55.degree. C., or -20 to -50.degree. C., or -25 to -45.degree. C., or -30 to -40.degree. C., or -32 to -47.degree. C., as determined by ASTM D5950.
[0143] The methyl paraffin lubricating oils of the instant disclosure may also have a Cold Crank Simulator (CCS) viscosity (-35 deg. C) that ranges from 700 to 1000 mPas, or 750 to 950 mPas, or 800 to 900 mPas, or 830 to 880 mPas, as determined by ASTM D5293.
[0144] One particularly preferred lubricating oil additive to be incorporated into the methyl paraffin lubricating oils of the present disclosure is a viscosity modifier. A polymethacrylate (PMA) viscosity modifier (e.g. Viscoplex 3-200 by Evnoik Industries) and a hydrocarbon (HC) hydrogenated polyisoprene star polymer type viscosity modifier (SV600 by Infineum) are two particularly preferred viscosity modifiers. The methyl paraffin lubricating oils of the instant disclosure with a viscosity modifier may have a Viscosity Index from about 200 to about 350 as determined by ASTM D2270. Alternatively, the methyl paraffin lubricating oils of the instant disclosure with a viscosity modifier may have a Viscosity Index of from 220 to 340, or 240 to 330, or 260 to 320, or 270 to 310, or 280 to 300, as determined by ASTM D2270.
[0145] The methyl paraffin lubricating oils of the present disclosure may also include a cobase stock as a minor component. The cobase stock may be included in the lubricating oil at from 5 to 40 wt %, or 10 to 35 wt %, or 15 to 30 wt %, or 20 to 25 wt %.
[0146] The methyl paraffin lubricating oils of the present disclosure have a high temperature high shear (HTHS) viscosity of less than about 2.3 cP as determined by ASTM D4683, and a Noack volatility from about 15 to about 90 percent as determined by ASTM D5800.
[0147] Illustrative methyl paraffin lubricating oils of the instant disclosure have a viscosity (Kv.sub.100) from about 1 cSt to about 8 cSt, more preferably from about 2 cSt to about 6 cSt, even more preferably from about 1 cSt to about 4 cSt, still even more preferably from about 2 cSt to about 3 cSt, at 100.degree. C. as determined by ASTM D445 or ASTM D7042, a viscosity index (VI) from about -100 to about 300, more preferably from about 0 to about 200, even more preferably from about 25 to about 150, still even more preferably from about 100 to about 170, as determined by ASTM D2270, a Noack volatility of no greater than 90 percent, more preferably no greater than 75 percent, still more preferably no greater than 50 percent, even still more preferably no greater than 40 percent, yet more even more preferably no greater than 30 percent, as determined by ASTM D5800, and a high temperature high shear (HTHS) viscosity of less than about 2.5 cP, more preferably less than about 2.25 cP, even more preferably less than about 2.0 cP, as determined by ASTM D4683.
[0148] Preferred methyl paraffin base stocks of the instant disclosure have a high temperature high shear (HTHS) viscosity of less than about 1.7 cP as determined by ASTM D4683, and a Noack volatility from about 15 to about 90 percent as determined by ASTM D5800.
[0149] The methyl paraffin base stocks of the instant disclosure have more desirable viscosity-volatility characteristics when compared to commercially available low viscosity Group IV PAO synthetic base stocks (e.g., SpectraSyn.TM. 2, SpectraSyn.TM. 4) or Group V ester base stocks (e.g., 2-ethylhexyl oleate, 2-ethylhexyl adipate, isodecyl adipate, 2-ethylhexyl phthalate, nC8/nC10 neopentyl glycol esters, nC7 trimethyolpropane ester, and the like). The methyl paraffin base stocks of the instant disclosure have lower viscosities than commercially available esters at similar volatility. Additionally, the methyl paraffin base stocks of the instant disclosure have lower volatility than commercially available esters at comparable viscosities.
[0150] Surprisingly, as shown in the Examples herein, it has been discovered that formulated engine oils employing these methyl paraffin base stocks of the instant disclosure as a major component possess unexpectedly high thermal and oxidative stability along with good deposit control benefit, as confirmed by their consistently low deposit formation from the Thermo-Oxidation Engine Oil Simulation Test (TEOST 33C ASTM D6335) conducted at high temperature. In comparison, the same formulation based on commercially available ester base stocks (e.g., diisooctyl adipate) show high deposit formation in the same test.
[0151] As shown in the Examples herein, the methyl paraffin base stocks of the instant disclosure have more desirable viscosity-volatility characteristics when compared to commercially available low viscosity Group IV PAO synthetic base stocks (e.g., SpectraSyn.TM. 2, SpectraSyn.TM. 4) or commercially available low viscosity Group II and Group III base stocks.
[0152] Surprisingly, as shown in the Examples herein, it has been discovered that formulated engine oils employing these methyl paraffin base stocks of the instant disclosure as a major component possess unexpectedly high thermal and oxidative stability along with good deposit control benefit, as confirmed by their consistently low deposit formation from the Thermo-Oxidation Engine Oil Simulation Test (TEOST 33C ASTM D6335) conducted at high temperature. In comparison, the same formulation based on commercially available ester base stocks (e.g., diisooctyl adipate) show high deposit formation in the same test.
[0153] The methyl paraffin base stocks of the instant disclosure have more desirable viscosity-volatility characteristics when compared to commercially available low viscosity Group IV PAO synthetic base stocks (e.g., SpectraSyn.TM. 2, SpectraSyn.TM. 4)) or Group V ester base stocks (e.g., 2-ethylhexyl oleate, 2-ethylhexyl adipate, isodecyl adipate, 2-ethylhexyl phthalate, nC8/nC10 neopentyl glycol esters, nC7 trimethyolpropane ester, and the like) or commercially available low viscosity Group II and Group III base stocks.
[0154] Surprisingly, as shown in the Examples herein, it has been discovered that formulated engine oils employing these methyl paraffin base stocks of the instant disclosure as a major component possess unexpectedly good traction properties.
[0155] As shown in the Examples herein, the methyl paraffin base stocks of the instant disclosure have more desirable viscosity-volatility characteristics when compared to commercially available low viscosity Group IV PAO synthetic base stocks (e.g., SpectraSyn.TM. 2, SpectraSyn.TM. 4) or Group II base stocks or Group III base stocks
[0156] Examples of techniques that can be employed to characterize the compositions formed by the process described above include, but are not limited to, analytical gas chromatography, nuclear magnetic resonance, thermogravimetric analysis (TGA), inductively coupled plasma mass spectrometry, differential scanning calorimetry (DSC), volatility and viscosity measurements.
[0157] This disclosure provides lubricating oils useful as engine oils, driveline oils, and in other applications characterized by excellent oxidative stability. The lubricating oils are based on methyl paraffin base stocks of the instant disclosure. In the present specification and claims, the terms base oil(s) and base stock(s) are used interchangeably.
[0158] In another aspect, as the oil operating temperature decreases, the viscosity of a high VI oil will not increase as much as the viscosity of a low VI oil. This is advantageous because the excessive high viscosity of the low VI oil will decrease the efficiency of the operating machine. Thus high VI (HVI) oil has performance advantages in both high and low temperature operation. VI is determined according to ASTM D2270. VI is related to kinematic viscosities measured at 40.degree. C. and 100.degree. C. using ASTM D445.
Lubricating Oil Co-Base Stocks
[0159] The methyl paraffin lubricating oils of the instant disclosure may also include co-base stock as a minor component. The co-base stock component of the present lubricating oils will typically be from 1 to 50 percent, or more preferably from 5 to 45 percent, or more preferably from 10 to 40 percent, or more preferably from 20 to 30 percent.
[0160] A wide range of lubricating oils is known in the art that can function as a co-base stock component of the instant application. Lubricating oils that are useful as a co-base stock component in the present disclosure are both natural oils and synthetic oils. Natural and synthetic oils (or mixtures thereof) can be used unrefined, refined, or re-refined (the latter is also known as reclaimed or reprocessed oil). Unrefined oils are those obtained directly from a natural or synthetic source and used without added purification. These include shale oil obtained directly from retorting operations, petroleum oil obtained directly from primary distillation, and ester oil obtained directly from an esterification process. Refined oils are similar to the oils discussed for unrefined oils except refined oils are subjected to one or more purification steps to improve the at least one lubricating oil property. One skilled in the art is familiar with many purification processes. These processes include solvent extraction, secondary distillation, acid extraction, base extraction, filtration, and percolation. Re-refined oils are obtained by processes analogous to refined oils but using an oil that has been previously used as a feed stock.
[0161] Groups I, II, III, IV and V are broad categories of base oil stocks developed and defined by the American Petroleum Institute (API Publication 1509; www.API.org) to create guidelines for lubricant base oils. Group I base stocks generally have a viscosity index of between about 80 to 120 and contain greater than about 0.03% sulfur and less than about 90% saturates. Group II base stocks generally have a viscosity index of between about 80 to 120, and contain less than or equal to about 0.03% sulfur and greater than or equal to about 90% saturates. Group III stock generally has a viscosity index greater than about 120 and contains less than or equal to about 0.03% sulfur and greater than about 90% saturates. Group IV includes polyalphaolefins (PAO). Group V base stocks include base stocks not included in Groups I-IV. The table below summarizes properties of each of these five groups.
TABLE-US-00001 Base Oil Properties Saturates Sulfur Viscosity Index Group I <90 and/or >0.03% and .gtoreq.80 and <120 Group II .gtoreq.90 and .ltoreq.0.03% and .gtoreq.80 and <120 Group III .gtoreq.90 and .ltoreq.0.03% and .gtoreq.120 Group IV Includes polyalphaolefins (PAO) products Group V All other base oil stocks not included in Groups I, II, III or IV
[0162] Natural oils include animal oils, vegetable oils (castor oil and lard oil, for example), and mineral oils. Animal and vegetable oils possessing favorable thermal oxidative stability can be used. Of the natural oils, mineral oils are preferred. Mineral oils vary widely as to their crude source, for example, as to whether they are paraffinic, naphthenic, or mixed paraffinic-naphthenic.
[0163] Oils derived from coal or shale are also useful in the present disclosure. Natural oils vary also as to the method used for their production and purification, for example, their distillation range and whether they are straight run or cracked, hydrorefined, or solvent extracted.
[0164] Group II and/or Group III hydroprocessed or hydrocracked base stocks, as well as synthetic oils such as polyalphaolefins, alkyl aromatics and synthetic esters, i.e. Group IV and Group V oils are also well known base stock oils.
[0165] Synthetic oils include hydrocarbon oil such as polymerized and interpolymerized olefins (polybutylenes, polypropylenes, propylene isobutylene copolymers, ethylene-olefin copolymers, and ethylene-alphaolefin copolymers, for example). Polyalphaolefin (PAO) oil base stocks, the Group IV API base stocks, are a commonly used synthetic hydrocarbon oil. By way of example, PAOs derived from C.sub.8, C.sub.10, C.sub.12, C.sub.14 olefins or mixtures thereof may be utilized. See U.S. Pat. Nos. 4,956,122; 4,827,064; and 4,827,073, which are incorporated herein by reference in their entirety. Group IV oils, that is, the PAO base stocks have viscosity indices preferably greater than 130, more preferably greater than 135, still more preferably greater than 140.
[0166] Esters may be useful in the lubricating oils of this disclosure. Additive solvency and seal compatibility characteristics may be secured by the use of esters such as the esters of dibasic acids with monoalkanols and the polyol esters of monocarboxylic acids. Esters of the former type include, for example, the esters of dicarboxylic acids such as phthalic acid, succinic acid, sebacic acid, fumaric acid, adipic acid, linoleic acid dimer, malonic acid, alkyl malonic acid, alkenyl malonic acid, etc., with a variety of alcohols such as butyl alcohol, hexyl alcohol, dodecyl alcohol, 2-ethylhexyl alcohol, etc. Specific examples of these types of esters include dibutyl adipate, di-(2-ethylhexyl) sebacate, di-n-hexyl fumarate, dioctyl sebacate, diisooctyl azelate, diisodecyl azelate, dioctyl phthalate, didecyl phthalate, dieicosyl sebacate, etc.
[0167] Particularly useful synthetic esters are those which are obtained by reacting one or more polyhydric alcohols, preferably the hindered polyols such as the neopentyl polyols; e.g., neopentyl glycol, trimethylol ethane, 2-methyl-2-propyl-1,3-propanediol, trimethylol propane, pentaerythritol and dipentaerythritol with alkanoic acids containing at least about 4 carbon atoms, preferably C.sub.8 to C.sub.30 acids such as saturated straight chain fatty acids including caprylic acid, capric acids, lauric acid, myristic acid, palmitic acid, stearic acid, arachic acid, and behenic acid, or the corresponding branched chain fatty acids or unsaturated fatty acids such as oleic acid, or mixtures of any of these materials.
[0168] Esters should be used in an amount such that the improved wear and corrosion resistance provided by the lubricating oils of this disclosure are not adversely affected.
[0169] Non-conventional or unconventional base stocks and/or base oils include one or a mixture of base stock(s) and/or base oil(s) derived from: (1) one or more Gas-to-Liquids (GTL) materials, as well as (2) hydrodewaxed, or hydroisomerized/cat (and/or solvent) dewaxed base stock(s) and/or base oils derived from synthetic wax, natural wax or waxy feeds, mineral and/or non-mineral oil waxy feed stocks such as gas oils, slack waxes (derived from the solvent dewaxing of natural oils, mineral oils or synthetic oils; e.g., Fischer-Tropsch feed stocks), natural waxes, and waxy stocks such as gas oils, waxy fuels hydrocracker bottoms, waxy raffinate, hydrocrackate, thermal crackates, foots oil or other mineral, mineral oil, or even non-petroleum oil derived waxy materials such as waxy materials recovered from coal liquefaction or shale oil, linear or branched hydrocarbyl compounds with carbon number of about 20 or greater, preferably about 30 or greater and mixtures of such base stocks and/or base oils.
[0170] GTL materials are materials that are derived via one or more synthesis, combination, transformation, rearrangement, and/or degradation/deconstructive processes from gaseous carbon-containing compounds, hydrogen-containing compounds and/or elements as feed stocks such as hydrogen, carbon dioxide, carbon monoxide, water, methane, ethane, ethylene, acetylene, propane, propylene, propyne, butane, butylenes, and butynes. GTL base stocks and/or base oils are GTL materials of lubricating viscosity that are generally derived from hydrocarbons; for example, waxy synthesized hydrocarbons, that are themselves derived from simpler gaseous carbon-containing compounds, hydrogen-containing compounds and/or elements as feed stocks. GTL base stock(s) and/or base oil(s) include oils boiling in the lube oil boiling range (1) separated/fractionated from synthesized GTL materials such as, for example, by distillation and subsequently subjected to a final wax processing step which involves either or both of a catalytic dewaxing process, or a solvent dewaxing process, to produce lube oils of reduced/low pour point; (2) synthesized wax isomerates, comprising, for example, hydrodewaxed or hydroisomerized cat and/or solvent dewaxed synthesized wax or waxy hydrocarbons; (3) hydrodewaxed or hydroisomerized cat and/or solvent dewaxed Fischer-Tropsch (F-T) material (i.e., hydrocarbons, waxy hydrocarbons, waxes and possible analogous oxygenates); preferably hydrodewaxed or hydroisomerized/followed by cat and/or solvent dewaxing dewaxed F-T waxy hydrocarbons, or hydrodewaxed or hydroisomerized/followed by cat (or solvent) dewaxing dewaxed, F-T waxes, or mixtures thereof.
[0171] GTL base stock(s) and/or base oil(s) derived from GTL materials, especially, hydrodewaxed or hydroisomerized/followed by cat and/or solvent dewaxed wax or waxy feed, preferably F-T material derived base stock(s) and/or base oil(s), are characterized typically as having kinematic viscosities at 100.degree. C. of from about 2 mm.sup.2/s to about 50 mm.sup.2/s (ASTM D445). They are further characterized typically as having pour points of -5.degree. C. to about -40.degree. C. or lower (ASTM D97). They are also characterized typically as having viscosity indices of about 80 to about 140 or greater (ASTM D2270).
[0172] In addition, the GTL base stock(s) and/or base oil(s) are typically highly paraffinic (>90% saturates), and may contain mixtures of monocycloparaffins and multicycloparaffins in combination with non-cyclic isoparaffins. The ratio of the naphthenic (i.e., cycloparaffin) content in such combinations varies with the catalyst and temperature used. Further, GTL base stock(s) and/or base oil(s) typically have very low sulfur and nitrogen content, generally containing less than about 10 ppm, and more typically less than about 5 ppm of each of these elements. The sulfur and nitrogen content of GTL base stock(s) and/or base oil(s) obtained from F-T material, especially F-T wax, is essentially nil. In addition, the absence of phosphorous and aromatics make this materially especially suitable for the formulation of low SAP products.
[0173] The term GTL base stock and/or base oil and/or wax isomerate base stock and/or base oil is to be understood as embracing individual fractions of such materials of wide viscosity range as recovered in the production process, mixtures of two or more of such fractions, as well as mixtures of one or two or more low viscosity fractions with one, two or more higher viscosity fractions to produce a blend wherein the blend exhibits a target kinematic viscosity.
[0174] The GTL material, from which the GTL base stock(s) and/or base oil(s) is/are derived, is preferably an F-T material (i.e., hydrocarbons, waxy hydrocarbons, wax).
[0175] Base oils for use in the formulated lubricating oils useful in the present disclosure are any of the variety of oils corresponding to API Group I, Group II, Group III, Group IV, Group V and Group VI oils and mixtures thereof, preferably API Group II, Group III, Group IV, Group V and Group VI oils and mixtures thereof, more preferably the Group III to Group VI base oils due to their exceptional volatility, stability, viscometric and cleanliness features. Minor quantities of Group I stock, such as the amount used to dilute additives for blending into formulated lube oil products, can be tolerated but should be kept to a minimum, i.e. amounts only associated with their use as diluent/carrier oil for additives used on an "as received" basis. Even in regard to the Group II stocks, it is preferred that the Group II stock be in the higher quality range associated with that stock, i.e. a Group II stock having a viscosity index in the range 100<VI<120.
[0176] In addition, the GTL base stock(s) and/or base oil(s) are typically highly paraffinic (>90% saturates), and may contain mixtures of monocycloparaffins and multicycloparaffins in combination with non-cyclic isoparaffins. The ratio of the naphthenic (i.e., cycloparaffin) content in such combinations varies with the catalyst and temperature used. Further, GTL base stock(s) and/or base oil(s) and hydrodewaxed, or hydroisomerized/cat (and/or solvent) dewaxed base stock(s) and/or base oil(s) typically have very low sulfur and nitrogen content, generally containing less than about 10 ppm, and more typically less than about 5 ppm of each of these elements. The sulfur and nitrogen content of GTL base stock(s) and/or base oil(s) obtained from F-T material, especially F-T wax, is essentially nil. In addition, the absence of phosphorous and aromatics make this material especially suitable for the formulation of low sulfur, sulfated ash, and phosphorus (low SAP) products.
Other Additives
[0177] The formulated methyl paraffin lubricating oil useful in the present disclosure may additionally contain one or more of the other commonly used lubricating oil performance additives including but not limited to dispersants, other detergents, corrosion inhibitors, rust inhibitors, metal deactivators, other anti-wear agents and/or extreme pressure additives, anti-seizure agents, wax modifiers, viscosity index improvers, viscosity modifiers, fluid-loss additives, seal compatibility agents, other friction modifiers, lubricity agents, anti-staining agents, chromophoric agents, defoamants, demulsifiers, emulsifiers, densifiers, wetting agents, gelling agents, tackiness agents, colorants, and others. For a review of many commonly used additives, see Klamann in Lubricants and Related Products, Verlag Chemie, Deerfield Beach, Fla.; ISBN 0-89573-177-0. Reference is also made to "Lubricant Additives Chemistry and Applications" edited by Leslie R. Rudnick, Marcel Dekker, Inc. New York, 2003 ISBN: 0-8247-0857-1.
[0178] All of the lubricating oil additives described below can be used alone or in combination. The total treat rates for the additives can range from 1 to 30 weight percent, or more preferably from 2 to 25 weight percent, or more preferably from 3 to 20 weight percent, or more preferably from 4 to 15 weight percent, or more preferably from 5 to 10 weight percent. Particularly preferred compositions have additive levels between 15 and 20 weight percent.
[0179] When used in lubricating oils, the inventive methyl paraffin base stocks disclosed herein may be included in the lubricating oil at from 70 to 99 weight percent, or more preferably from 75 to 98 weight percent, or more preferably from 80 to 97 weight percent, or more preferably from 85 to 96 weight percent, or more preferably from 90 to 95 weight percent of the total lubricating oil composition. Particularly preferred oil compositions have base stock loadings between 80 and 85 weight percent.
[0180] The types and quantities of performance additives used in combination with the instant disclosure in lubricant compositions are not limited by the examples shown herein as illustrations.
Viscosity Modifiers
[0181] Viscosity modifiers (also known as viscosity index improvers (VI improvers), and viscosity improvers) can be included in the lubricant compositions of this disclosure.
[0182] Viscosity modifiers provide lubricants with high and low temperature operability. These additives impart shear stability at elevated temperatures and acceptable viscosity at low temperatures.
[0183] Suitable viscosity modifiers include high molecular weight hydrocarbons, polyesters and viscosity modifier dispersants that function as both a viscosity modifier and a dispersant. Typical molecular weights of these polymers are between about 10,000 to 1,500,000, more typically about 20,000 to 1,200,000, and even more typically between about 50,000 and 1,000,000.
[0184] Examples of suitable viscosity modifiers are linear or star-shaped polymers and copolymers of methacrylate, butadiene, olefins, or alkylated styrenes. Polyisobutylene is a commonly used viscosity modifier. Another suitable viscosity modifier is polymethacrylate (copolymers of various chain length alkyl methacrylates, for example), some formulations of which also serve as pour point depressants. Other suitable viscosity modifiers include copolymers of ethylene and propylene, hydrogenated block copolymers of styrene and isoprene, and polyacrylates (copolymers of various chain length acrylates, for example). Specific examples include styrene-isoprene or styrene-butadiene based polymers of 50,000 to 200,000 molecular weight.
[0185] Olefin copolymers are commercially available from Chevron Oronite Company LLC under the trade designation "PARATONE.RTM." (such as "PARATONE.RTM. 8921" and "PARATONE.RTM. 8941"); from Afton Chemical Corporation under the trade designation "HiTEC.RTM." (such as "HiTEC.RTM. 5850B"; and from The Lubrizol Corporation under the trade designation "Lubrizol.RTM. 7067C". Hydrogenated polyisoprene star polymers are commercially available from Infineum International Limited, e.g., under the trade designation "SV200" and "SV600". Hydrogenated diene-styrene block copolymers are commercially available from Infineum International Limited, e.g., under the trade designation "SV 50".
[0186] The polymethacrylate or polyacrylate polymers can be linear polymers which are available from Evonik Industries under the trade designation "Viscoplex.RTM." (e.g., Viscoplex 6-954, Viscoplex 3-200) or star polymers which are available from Lubrizol Corporation under the trade designation Asteric.TM. (e.g., Lubrizol 87708 and Lubrizol 87725).
[0187] Illustrative vinyl aromatic-containing polymers useful in this disclosure may be derived predominantly from vinyl aromatic hydrocarbon monomer. Illustrative vinyl aromatic-containing copolymers useful in this disclosure may be represented by the following general formula:
A-B
wherein A is a polymeric block derived predominantly from vinyl aromatic hydrocarbon monomer, and B is a polymeric block derived predominantly from conjugated diene monomer.
[0188] The vinyl aromatic-containing polymers or copolymers useful in this disclosure have a weight average molecular weight greater than about 80,000, and a number average molecular weight greater than about 40,000; preferably a weight average molecular weight greater than about 90,000, and a number average molecular weight greater than about 75,000; and more preferably a weight average molecular weight greater than about 100,000 and less than 1,000,000, and a number average molecular weight greater than about 100,000 and less than 1,000,000. The vinyl aromatic-containing polymers or copolymers have an amount of vinyl aromatic content greater than about 10% by weight, or greater than about 20% by weight, or greater than about 30% by weight, of the vinyl aromatic-containing polymer or copolymer. The vinyl aromatic-containing polymers or copolymers have an amount of vinyl aromatic content preferably between about 10% and about 50% by weight, more preferably between about 15% and about 40% by weight, and even more preferably between about 20% and about 35% by weight, of the vinyl aromatic-containing polymer or copolymer.
[0189] In an embodiment of this disclosure, the viscosity modifiers may be used in an amount of less than about 2.0 weight percent, preferably less than about 1.0 weight percent, and more preferably less than about 0.5 weight percent, based on the total weight of the formulated oil or lubricating engine oil. Viscosity modifiers are typically added as concentrates, in large amounts of diluent oil.
[0190] In another embodiment of this disclosure, the viscosity modifiers may be used in an amount of from 0.05 to about 2.0 weight percent, preferably 0.15 to about 1.0 weight percent, and more preferably 0.25 to about 0.5 weight percent, based on the total weight of the formulated oil or lubricating engine oil. Or the viscosity modifiers may be used in an amount (total solid polymer content) of from 0.5 to about 2.0 weight percent, preferably 0.8 to about 1.5 weight percent, and more preferably 1.0 to about 1.3 weight percent, based on the total weight of the formulated oil or lubricating engine oil.
[0191] As used herein, the viscosity modifier concentrations are given on an "as delivered" basis. Typically, the active polymer is delivered with a diluent oil. The "as delivered" viscosity modifier typically contains from 20 weight percent to 75 weight percent of an active polymer for polymethacrylate or polyacrylate polymers, or from 8 weight percent to 20 weight percent of an active polymer for olefin copolymers, hydrogenated polyisoprene star polymers, or hydrogenated diene-styrene block copolymers, in the "as delivered" polymer concentrate.
Antioxidants
[0192] Typical anti-oxidant include phenolic anti-oxidants, aminic anti-oxidants and oil-soluble copper complexes.
[0193] The phenolic antioxidants include sulfurized and non-sulfurized phenolic antioxidants. The terms "phenolic type" or "phenolic antioxidant" used herein includes compounds having one or more than one hydroxyl group bound to an aromatic ring which may itself be mononuclear, e.g., benzyl, or poly-nuclear, e.g., naphthyl and spiro aromatic compounds. Thus "phenol type" includes phenol per se, catechol, resorcinol, hydroquinone, naphthol, etc., as well as alkyl or alkenyl and sulfurized alkyl or alkenyl derivatives thereof, and bisphenol type compounds including such bi-phenol compounds linked by alkylene bridges sulfuric bridges or oxygen bridges. Alkyl phenols include mono- and poly-alkyl or alkenyl phenols, the alkyl or alkenyl group containing from about 3-100 carbons, preferably 4 to 50 carbons and sulfurized derivatives thereof, the number of alkyl or alkenyl groups present in the aromatic ring ranging from 1 to up to the available unsatisfied valences of the aromatic ring remaining after counting the number of hydroxyl groups bound to the aromatic ring.
[0194] Generally, therefore, the phenolic anti-oxidant may be represented by the general formula:
(R).sub.x--Ar.sub.x(OH).sub.y
where Ar is selected from the group consisting of:
##STR00015##
wherein R is a C.sub.3-C.sub.100 alkyl or alkenyl group, a sulfur substituted alkyl or alkenyl group, preferably a C.sub.4-C.sub.50 alkyl or alkenyl group or sulfur substituted alkyl or alkenyl group, more preferably C.sub.3-C.sub.100 alkyl or sulfur substituted alkyl group, most preferably a C.sub.4-C.sub.50 alkyl group, R.sup.G is a C.sub.1-C.sub.100 alkylene or sulfur substituted alkylene group, preferably a C.sub.2-C.sub.50 alkylene or sulfur substituted alkylene group, more preferably a C.sub.2-C.sub.2 alkylene or sulfur substituted alkylene group, y is at least 1 to up to the available valences of Ar, x ranges from 0 to up to the available valances of Ar-y, z ranges from 1 to 10, n ranges from 0 to 20, and m is 0 to 4 and p is 0 or 1, preferably y ranges from 1 to 3, x ranges from 0 to 3, z ranges from 1 to 4 and n ranges from 0 to 5, and p is 0.
[0195] Preferred phenolic anti-oxidant compounds are the hindered phenolics and phenolic esters which contain a sterically hindered hydroxyl group, and these include those derivatives of dihydroxy aryl compounds in which the hydroxyl groups are in the o- or p-position to each other. Typical phenolic anti-oxidants include the hindered phenols substituted with C.sub.1+ alkyl groups and the alkylene coupled derivatives of these hindered phenols. Examples of phenolic materials of this type 2-t-butyl-4-heptyl phenol; 2-t-butyl-4-octyl phenol; 2-t-butyl-4-dodecyl phenol; 2,6-di-t-butyl-4-heptyl phenol; 2,6-di-t-butyl-4-dodecyl phenol; 2-methyl-6-t-butyl-4-heptyl phenol; 2-methyl-6-t-butyl-4-dodecyl phenol; 2,6-di-t-butyl-4 methyl phenol; 2,6-di-t-butyl-4-ethyl phenol; and 2,6-di-t-butyl 4 alkoxy phenol; and
##STR00016##
[0196] Phenolic type anti-oxidants are well known in the lubricating industry and commercial examples such as Ethanox.RTM. 4710, Irganox.RTM. 1076, Irganox.RTM. L1035, Irganox.RTM. 1010, Irganox.RTM. L109, Irganox.RTM. L118, Irganox.RTM. L135 and the like are familiar to those skilled in the art. The above is presented only by way of exemplification, not limitation on the type of phenolic anti-oxidants which can be used.
[0197] The phenolic anti-oxidant can be employed in an amount in the range of about 0.1 to 3 wt %, preferably about 1 to 3 wt %, more preferably 1.5 to 3 wt % on an active ingredient basis.
[0198] Aromatic amine anti-oxidants include phenyl-.alpha.-naphthyl amine which is described by the following molecular structure:
##STR00017##
wherein R.sup.z is hydrogen or a C.sub.1 to C.sub.14 linear or C.sub.3 to C.sub.14 branched alkyl group, preferably C.sub.1 to C.sub.10 linear or C.sub.3 to C.sub.10 branched alkyl group, more preferably linear or branched C.sub.6 to C.sub.8 and n is an integer ranging from 1 to 5 preferably 1. A particular example is Irganox L06.
[0199] Other aromatic amine anti-oxidants include other alkylated and non-alkylated aromatic amines such as aromatic monoamines of the formula R.sup.8R.sup.9R.sup.10N where R.sup.8 is an aliphatic, aromatic or substituted aromatic group, R.sup.9 is an aromatic or a substituted aromatic group, and R.sup.10 is H, alkyl, aryl or R.sup.11S(O).sub.xR.sup.12 where R.sup.11 is an alkylene, alkenylene, or aralkylene group, R.sup.12 is a higher alkyl group, or an alkenyl, aryl, or alkaryl group, and x is 0, 1 or 2. The aliphatic group R.sup.8 may contain from 1 to about 20 carbon atoms, and preferably contains from about 6 to 12 carbon atoms. The aliphatic group is a saturated aliphatic group. Preferably, both R.sup.8 and R.sup.9 are aromatic or substituted aromatic groups, and the aromatic group may be a fused ring aromatic group such as naphthyl. Aromatic groups R.sup.8 and R.sup.9 may be joined together with other groups such as S.
[0200] Typical aromatic amines anti-oxidants have alkyl substituent groups of at least about 6 carbon atoms. Examples of aliphatic groups include hexyl, heptyl, octyl, nonyl, and decyl. Generally, the aliphatic groups will not contain more than about 14 carbon atoms. The general types of such other additional amine anti-oxidants which may be present include diphenylamines, phenothiazines, imidodibenzyls and diphenyl phenylene diamines. Mixtures of two or more of such other additional aromatic amines may also be present. Polymeric amine antioxidants can also be used.
[0201] Aromatic amines anti-oxidants can be employed in an amount in the range of about 0.1 to 5 wt %, preferably about 0.5 to 3 wt %, more preferably 1 to 3 wt % on an active ingredient basis.
[0202] Another class of anti-oxidant used in lubricating oil compositions and which may also be present are oil-soluble copper compounds. Any oil-soluble suitable copper compound may be blended into the lubricating oil. Examples of suitable copper antioxidants include copper dihydrocarbyl thio- or dithio-phosphates and copper salts of carboxylic acid (naturally occurring or synthetic). Other suitable copper salts include copper dithiacarbamates, sulphonates, phenates, and acetylacetonates. Basic, neutral, or acidic copper Cu(I) and or Cu(II) salts derived from alkenyl succinic acids or anhydrides are known to be particularly useful.
[0203] Such antioxidants may be used individually or as mixtures of one or more types of antioxidants, the total amount employed being an amount of about 0.50 to 5 wt %, preferably about 0.75 to 3 wt % (on an as-received basis).
Detergents
[0204] Illustrative detergents useful in this disclosure include, for example, alkali metal detergents, alkaline earth metal detergents, or mixtures of one or more alkali metal detergents and one or more alkaline earth metal detergents. Oils formulated with low concentrations of detergents and/or low ash detergents can be preferred as low ash, low metals, low phosphorus oils. A typical detergent is an anionic material that contains a long chain hydrophobic portion of the molecule and a smaller anionic or oleophobic hydrophilic portion of the molecule. The anionic portion of the detergent is typically derived from an organic acid such as a sulfur acid, carboxylic acid, phosphorous acid, phenol, or mixtures thereof. The counterion is typically an alkaline earth or alkali metal.
[0205] Salts that contain a substantially stochiometric amount of the metal are described as neutral salts and have a total base number (TBN, as measured by ASTM D2896) of from 0 to 80. Many compositions are overbased, containing large amounts of a metal base that is achieved by reacting an excess of a metal compound (a metal hydroxide or oxide, for example) with an acidic gas (such as carbon dioxide). Useful detergents can be neutral, mildly overbased, or highly overbased. These detergents can be used in mixtures of neutral, overbased, highly overbased calcium salicylate, sulfonates, phenates and/or magnesium salicylate, sulfonates, phenates. The TBN ranges can vary from low, medium to high TBN products, including as low as 0 to as high as 600. Mixtures of low, medium, high TBN can be used, along with mixtures of calcium and magnesium metal based detergents, and including sulfonates, phenates, salicylates, and carboxylates. A detergent mixture with a metal ratio of 1, in conjunction of a detergent with a metal ratio of 2, and as high as a detergent with a metal ratio of 5, can be used. Borated detergents can also be used.
[0206] Alkaline earth phenates are another useful class of detergent. These detergents can be made by reacting alkaline earth metal hydroxide or oxide (CaO, Ca(OH)2, BaO, Ba(OH)2, MgO, Mg(OH)2, for example) with an alkyl phenol or sulfurized alkylphenol. Useful alkyl groups include straight chain or branched C1-C30 alkyl groups, preferably, C4-C20 or mixtures thereof. Examples of suitable phenols include isobutylphenol, 2-ethylhexylphenol, nonylphenol, dodecyl phenol, and the like. It should be noted that starting alkylphenols may contain more than one alkyl substituent that are each independently straight chain or branched and can be used from 0.5 to 6 weight percent. When a non-sulfurized alkylphenol is used, the sulfurized product may be obtained by methods well known in the art. These methods include heating a mixture of alkylphenol and sulfurizing agent (including elemental sulfur, sulfur halides such as sulfur dichloride, and the like) and then reacting the sulfurized phenol with an alkaline earth metal base.
[0207] Metal salts of carboxylic acids are also useful as detergents. These carboxylic acid detergents may be prepared by reacting a basic metal compound with at least one carboxylic acid and removing free water from the reaction product. These compounds may be overbased to produce the desired TBN level. Detergents made from salicylic acid are one preferred class of detergents derived from carboxylic acids. Useful salicylates include long chain alkyl salicylates. One useful family of compositions is of the formula
##STR00018##
where R is an alkyl group having 1 to about 30 carbon atoms, n is an integer from 1 to 4, and M is an alkaline earth metal. Preferred R groups are alkyl chains of at least C11, preferably C13 or greater. R may be optionally substituted with substituents that do not interfere with the detergent's function. M is preferably, calcium, magnesium, or barium. More preferably, M is calcium or magnesium.
[0208] Hydrocarbyl-substituted salicylic acids may be prepared from phenols by the Kolbe reaction (see U.S. Pat. No. 3,595,791). The metal salts of the hydrocarbyl-substituted salicylic acids may be prepared by double decomposition of a metal salt in a polar solvent such as water or alcohol.
[0209] Alkaline earth metal phosphates are also used as detergents and are known in the art.
[0210] Detergents may be simple detergents or what is known as hybrid or complex detergents. The latter detergents can provide the properties of two detergents without the need to blend separate materials. See U.S. Pat. No. 6,034,039.
[0211] Preferred detergents include calcium phenates, calcium sulfonates, calcium salicylates, magnesium phenates, magnesium sulfonates, magnesium salicylates and other related components (including borated detergents), and mixtures thereof. Preferred mixtures of detergents include magnesium sulfonate and calcium salicylate, magnesium sulfonate and calcium sulfonate, magnesium sulfonate and calcium phenate, calcium phenate and calcium salicylate, calcium phenate and calcium sulfonate, calcium phenate and magnesium salicylate, calcium phenate and magnesium phenate.
[0212] The lubricating oils of this disclosure exhibit desired properties, e.g., wear control, deposit control and fuel efficiency, in the presence or absence of a detergent, in particular, the presence or absence of a salicylate detergent or a sulfonate detergent.
[0213] The detergent concentration in the lubricating oils of this disclosure can range from about 0.5 to about 20 weight percent or more, preferably about 0.6 to 5.0 weight percent, and more preferably from about 0.8 weight percent to about 4.0 weight percent, based on the total weight of the lubricating oil.
[0214] As used herein, the detergent concentrations are given on an "as delivered" basis.
[0215] Typically, the active detergent is delivered with a process oil. The "as delivered" detergent typically contains from about 20 weight percent to about 100 weight percent, or from about 40 weight percent to about 60 weight percent, of active detergent in the "as delivered" detergent product.
Dispersants
[0216] During engine operation, oil-insoluble oxidation byproducts are produced. Dispersants help keep these byproducts in solution, thus diminishing their deposition on metal surfaces. Dispersants may be ashless or ash-forming in nature. Preferably, the dispersant is ashless. So called ashless dispersants are organic materials that form substantially no ash upon combustion. For example, non-metal-containing or borated metal-free dispersants are considered ashless. In contrast, metal-containing detergents discussed above form ash upon combustion.
[0217] Suitable dispersants typically contain a polar group attached to a relatively high molecular weight hydrocarbon chain. The polar group typically contains at least one element of nitrogen, oxygen, or phosphorus. Typical hydrocarbon chains contain 50 to 400 carbon atoms.
[0218] A particularly useful class of dispersants are the alkenylsuccinic derivatives, typically produced by the reaction of a long chain substituted alkenyl succinic compound, usually a substituted succinic anhydride, with a polyhydroxy or polyamino compound. The long chain group constituting the oleophilic portion of the molecule which confers solubility in the oil, is normally a polyisobutylene group. Many examples of this type of dispersant are well known commercially and in the literature. Exemplary patents describing such dispersants are U.S. Pat. Nos. 3,172,892; 3,219,666; 3,316,177 and 4,234,435. Other types of dispersants are described in U.S. Pat. Nos. 3,036,003; and 5,705,458.
[0219] Hydrocarbyl-substituted succinic acid compounds are popular dispersants. In particular, succinimide, succinate esters, or succinate ester amides prepared by the reaction of a hydrocarbon-substituted succinic acid compound preferably having at least 50 carbon atoms in the hydrocarbon substituent, with at least one equivalent of an alkylene amine are particularly useful.
[0220] Succinimides are formed by the condensation reaction between alkenyl succinic anhydrides and amines. Molar ratios can vary depending on the amine or polyamine. For example, the molar ratio of alkenyl succinic anhydride to TEPA can vary from about 1:1 to about 5:1.
[0221] Succinate esters are formed by the condensation reaction between alkenyl succinic anhydrides and alcohols or polyols. Molar ratios can vary depending on the alcohol or polyol used. For example, the condensation product of an alkenyl succinic anhydride and pentaerythritol is a useful dispersant.
[0222] Succinate ester amides are formed by condensation reaction between alkenyl succinic anhydrides and alkanol amines. For example, suitable alkanol amines include ethoxylated polyalkylpolyamines, propoxylated polyalkylpolyamines and polyalkenylpolyamines such as polyethylene polyamines. One example is propoxylated hexamethylenediamine.
[0223] The molecular weight of the alkenyl succinic anhydrides will typically range between 800 and 2,500. The above products can be post-reacted with various reagents such as sulfur, oxygen, formaldehyde, carboxylic acids such as oleic acid, and boron compounds such as borate esters or highly borated dispersants. The dispersants can be borated with from about 0.1 to about 5 moles of boron per mole of dispersant reaction product.
[0224] Mannich base dispersants are made from the reaction of alkylphenols, formaldehyde, and amines. Process aids and catalysts, such as oleic acid and sulfonic acids, can also be part of the reaction mixture. Molecular weights of the alkylphenols range from 800 to 2,500. Mannich base dispersants can also be borated and mixtures of Mannich base dispersant can be used.
[0225] Typical high molecular weight aliphatic acid modified Mannich condensation products can be prepared from high molecular weight alkyl-substituted hydroxyaromatics or HN(R)2 group-containing reactants.
[0226] Examples of high molecular weight alkyl-substituted hydroxyaromatic compounds are polypropylphenol, polybutylphenol, and other polyalkylphenols. These polyalkylphenols can be obtained by the alkylation, in the presence of an alkylating catalyst, such as BF.sub.3, of phenol with high molecular weight polypropylene, polybutylene, and other polyalkylene compounds to give alkyl substituents on the benzene ring of phenol having an average 600-100,000 molecular weight.
[0227] Examples of HN(R).sub.2 group-containing reactants are alkylene polyamines, principally polyethylene polyamines. Other representative organic compounds containing at least one HN(R).sub.2 group suitable for use in the preparation of Mannich condensation products are well known and include the mono- and di-amino alkanes and their substituted analogs, e.g., ethylamine and diethanol amine; aromatic diamines, e.g., phenylene diamine, diamino naphthalenes; heterocyclic amines, e.g., morpholine, pyrrole, pyrrolidine, imidazole, imidazolidine, and piperidine; melamine and their substituted analogs.
[0228] Examples of alkylene polyamine reactants include ethylenediamine, diethylene triamine, triethylene tetraamine, tetraethylene pentaamine, pentaethylene hexamine, hexaethylene heptaamine, heptaethylene octaamine, octaethylene nonaamine, nonaethylene decamine, and decaethylene undecamine and mixture of such amines having nitrogen contents corresponding to the alkylene polyamines, in the formula H.sub.2N--(Z--NH--).sub.nH, mentioned before, Z is a divalent ethylene and n is 1 to 10 of the foregoing formula. Corresponding propylene polyamines such as propylene diamine and di-, tri-, tetra-, pentapropylene tri-, tetra-, penta- and hexaamines are also suitable reactants. The alkylene polyamines are usually obtained by the reaction of ammonia and dihalo alkanes, such as dichloro alkanes. Thus the alkylene polyamines obtained from the reaction of 2 to 11 moles of ammonia with 1 to 10 moles of dichloroalkanes having 2 to 6 carbon atoms and the chlorines on different carbons are suitable alkylene polyamine reactants.
[0229] Aldehyde reactants useful in the preparation of the high molecular products useful in this disclosure include the aliphatic aldehydes such as formaldehyde (also as paraformaldehyde and formalin), acetaldehyde and aldol (.beta.-hydroxybutyraldehyde). Formaldehyde or a formaldehyde-yielding reactant is preferred.
[0230] Preferred dispersants include borated and non-borated succinimides, including those derivatives from mono-succinimides, bis-succinimides, and/or mixtures of mono- and bis-succinimides, wherein the hydrocarbyl succinimide is derived from a hydrocarbylene group such as polyisobutylene having a Mn of from about 500 to about 5000 or a mixture of such hydrocarbylene groups. Other preferred dispersants include succinic acid-esters and amides, alkylphenol-polyamine-coupled Mannich adducts, their capped derivatives, and other related components. Such additives may be used in an amount of about 0.1 to 20 wt %, preferably about 0.1 to 8 wt %, more preferably about 1 to 6 wt % (on an as-received basis or 0-10 wt % on an active ingredient basis) based on the weight of the total lubricant.
Pour Point Depressants
[0231] Conventional pour point depressants (also known as lube oil flow improvers) may also be present. Pour point depressant may be added to lower the minimum temperature at which the fluid will flow or can be poured. Examples of suitable pour point depressants include alkylated naphthalenes polymethacrylates, polyacrylates, polyarylamides, condensation products of haloparaffin waxes and aromatic compounds, vinyl carboxylate polymers, and terpolymers of dialkylfumarates, vinyl esters of fatty acids and allyl vinyl ethers. Such additives may be used in amount of about 0.0 to 0.5 wt %, preferably about 0 to 0.3 wt %, more preferably about 0.001 to 0.1 wt % on an as-received basis.
Corrosion Inhibitors/Metal Deactivators
[0232] Corrosion inhibitors are used to reduce the degradation of metallic parts that are in contact with the lubricating oil composition. Suitable corrosion inhibitors include aryl thiazines, alkyl substituted dimercapto thiodiazoles thiadiazoles and mixtures thereof. Such additives may be used in an amount of about 0.01 to 5 wt %, preferably about 0.01 to 1.5 wt %, more preferably about 0.01 to 0.2 wt %, still more preferably about 0.01 to 0.1 wt % (on an as-received basis) based on the total weight of the lubricating oil composition.
Seal Compatibility Additives
[0233] Seal compatibility agents help to swell elastomeric seals by causing a chemical reaction in the fluid or physical change in the elastomer. Suitable seal compatibility agents for lubricating oils include organic phosphates, aromatic esters, aromatic hydrocarbons, esters (butylbenzyl phthalate, for example), and polybutenyl succinic anhydride and sulfolane-type seal swell agents such as Lubrizol 730-type seal swell additives. Such additives may be used in an amount of about 0.01 to 3 wt %, preferably about 0.01 to 2 wt % on an as-received basis.
Anti-Foam Agents
[0234] Anti-foam agents may advantageously be added to lubricant compositions. These agents retard the formation of stable foams. Silicones and organic polymers are typical anti-foam agents. For example, polysiloxanes, such as silicon oil or polydimethyl siloxane, provide antifoam properties. Anti-foam agents are commercially available and may be used in conventional minor amounts along with other additives such as demulsifiers; usually the amount of these additives combined is less than 1 percent, preferably 0.001 to about 0.5 wt %, more preferably about 0.001 to about 0.2 wt %, still more preferably about 0.0001 to 0.15 wt % (on an as-received basis) based on the total weight of the lubricating oil composition.
Inhibitors and Antirust Additives
[0235] Anti-rust additives (or corrosion inhibitors) are additives that protect lubricated metal surfaces against chemical attack by water or other contaminants. One type of anti-rust additive is a polar compound that wets the metal surface preferentially, protecting it with a film of oil. Another type of anti-rust additive absorbs water by incorporating it in a water-in-oil emulsion so that only the oil touches the surface. Yet another type of anti-rust additive chemically adheres to the metal to produce a non-reactive surface. Examples of suitable additives include zinc dithiophosphates, metal phenolates, basic metal sulfonates, fatty acids and amines. Such additives may be used in an amount of about 0.01 to 5 wt %, preferably about 0.01 to 1.5 wt % on an as-received basis.
[0236] ZDDP anti-wear additives are essential components of the present disclosure. ZDDP derived from C8 to C18 primary or secondary alcohols and preferably derived from C4, C5, and/or C7 primary or secondary alcohols and mixtures thereof are often preferred. In some applications, low phosphorus ZDDP additives with <0.10% by weight phosphorus, leading to about from 0.02% to 0.08% phosphorus in finished oils can be preferred. In addition to ZDDP, other anti-wear additives can be present, including zinc dithiocarbamates, molybdenum dialkyldithiophosphates, molybdenum dithiocarbamates, other organo molybdenum-nitrogen complexes, sulfurized olefins, etc.
[0237] The term "organo molybdenum-nitrogen complexes" embraces the organo molybdenum-nitrogen complexes described in U.S. Pat. No. 4,889,647. The complexes are reaction products of a fatty oil, dithanolamine and a molybdenum source. Specific chemical structures have not been assigned to the complexes. U.S. Pat. No. 4,889,647 reports an infrared spectrum for a typical reaction product of that disclosure; the spectrum identifies an ester carbonyl band at 1740 cm.sup.-1 and an amide carbonyl band at 1620 cm.sup.-1. The fatty oils are glyceryl esters of higher fatty acids containing at least 12 carbon atoms up to 22 carbon atoms or more. The molybdenum source is an oxygen-containing compound such as ammonium molybdates, molybdenum oxides and mixtures.
[0238] Other organo molybdenum complexes which can be used in the present disclosure are tri-nuclear molybdenum-sulfur compounds described in EP 1 040 115 and WO 99/31113 and the molybdenum complexes described in U.S. Pat. No. 4,978,464.
Performance and Uses
[0239] The methyl paraffin lubricant compositions of this disclosure give advantaged performance in the lubrication of internal combustion engines, power trains, drivelines, transmissions, gears, gear trains, gear sets, compressors, pumps, hydraulic systems, bearings, bushings, turbines, and the like,
[0240] Also, the methyl paraffin lubricant compositions of this disclosure also give advantaged friction, wear, and other lubricant performances in the lubrication of mechanical components, which comprise, for example, pistons, piston rings, cylinder liners, cylinders, cams, tappets, lifters, bearings (journal, roller, tapered, needle, ball, and the like), gears, valves, and the like.
[0241] Further, the methyl paraffin lubricant compositions of this disclosure give advantaged friction, wear, and other lubricant performances under a range of lubrication contact pressures, from 1 MPas to greater than 10 GPas, preferably greater than 10 MPas, more preferably greater than 100 MPas, even more preferable greater than 300 MPas. Under certain circumstances, the methyl paraffin lubricant compositions of this disclosure give advantaged wear and friction performance at greater than 0.5 GPas, often at greater than 1 GPas, sometimes greater than 2 GPas, under selected circumstances greater than 5 GPas.
[0242] Yet further, the methyl paraffin lubricant compositions of this disclosure give advantaged friction, wear, and other lubricant performances when used in combination with lubricated surfaces comprising: metals, metal alloys, non-metals, non-metal alloys, mixed carbon-metal composites and alloys, mixed carbon-nonmetal composites and alloys, ferrous metals, ferrous composites and alloys, non-ferrous metals, non-ferrous composites and alloys, titanium, titanium composites and alloys, aluminum, aluminum composites and alloys, magnesium, magnesium composites and alloys, ion-implanted metals and alloys, plasma modified surfaces; surface modified materials; coatings; mono-layer, multi-layer, and gradient layered coatings; honed surfaces; polished surfaces; etched surfaces; textured surfaces; micro and nano structures on textured surfaces; super-finished surfaces; diamond-like carbon (DLC), DLC with high-hydrogen content, DLC with moderate hydrogen content, DLC with low-hydrogen content, DLC with zero hydrogen content, DLC composites, DLC-metal compositions and composites, DLC-nonmetal compositions and composites; glasses, metallic glasses; ceramics, cermets, ceramic oxides, ceramic nitrides, FeN, CrN, ceramic carbides, mixed ceramic compositions, and the like; polymers, plastics, thermoplastic polymers, engineered polymers, polymer blends, polymer alloys, polymer composites; elastomers; materials compositions and composites containing dry lubricants, comprising for example graphite, carbon, molybdenum, molybdenum disulfide, polytetrafluoroethylene, polyperfluoropropylene, polyperfluoroalkylethers, and the like.
[0243] The viscometric properties of the methyl paraffin lubricants of this disclosure can be measured according to standard practices. A low viscosity can be advantageous for lubricants in modern equipment. A low high temperature high shear (HTHS) viscosity, in accordance with ASTM D4683, can indicate performance of a lubricant in a modern engine. In particular, the methyl paraffin lubricants of this disclosure can have an HTHS of less than 2.0 cP, or more preferably less than 1.9 cP, or more preferably less than 1.8 cP, or more preferably less than 1.7 cP.
[0244] The methyl paraffin lubricants of this disclosure can have lower volatility, as determined by the Noack volatility test ASTM D5800, or as predicted by a TGA test that simulates the Noack volatility. In particular, the lubricants of this disclosure can have a Noack between 1% and 50%, or more preferably between 10% and 50%, or more preferably between 15% and 45%, or more preferably between 20% and 35%. Particularly preferred compositions have a Noack between 15% and 30%.
[0245] The methyl paraffin lubricants of this disclosure can have lower deposition tendancy, as determined by the TEOST 33C deposition test ASTM D6335. In particular, the lubricants of this disclosure can have a TEOST 33C of less than 30 mg, or more preferably less than 20 mg, or more preferably less than 15 mg.
[0246] The methyl paraffin lubricants of this disclosure can have reduced traction as determined by the MTM (Mini Traction Machine) traction test. Traction is most easily assessed by comparison to a reference fluid, in this case a suitable reference fluid is an engine oil formulated with commercial GTL QHVI-3. Accordingly, the lubricants of this disclosure can have an MTM traction reduction of 5% versus a reference, or more preferably a reduction of 10% versus a reference, or more preferably a reduction of 20% versus a reference, or more preferably a reduction of 30% versus a reference, or more preferably a reduction of 40% versus a reference.
[0247] In the above detailed description, the specific embodiments of this disclosure have been described in connection with its preferred embodiments. However, to the extent that the above description is specific to a particular embodiment or a particular use of this disclosure, this is intended to be illustrative only and merely provides a concise description of the exemplary embodiments. Accordingly, the disclosure is not limited to the specific embodiments described above, but rather, the disclosure includes all alternatives, modifications, and equivalents falling within the true scope of the appended claims. Various modifications and variations of this disclosure will be obvious to a worker skilled in the art and it is to be understood that such modifications and variations are to be included within the purview of this application and the spirit and scope of the claims.
EXAMPLES
Example 1
Physical Characteristics of Inventive and Comparative Base Stocks
[0248] Low viscosity inventive C10 dimer, C10 trimer, blends of C10 dimer and trimer and comparative base stocks (C12 dimer, Group II, GTL and Group III) were prepared using the processes described in the detailed description and characterized for viscometric properties, volatility, density, pour point and MTM average traction coefficient. The inventive base stocks of this disclosure are blends of 9-methylnonadecane (also referred to herein as "C10 dimer") and 9-methyl-11-octylheneicosane (also referred to herein as "C10 trimer"). The chemical structures of the 9-methylnonadecane and 9-methyl-11-octylheneicosane base stocks for blending were as follows:
##STR00019##
[0249] Also shown below is the chemical structure of the comparative 11-methyltricosane basestock (also referred to herein as "C12 dimer").
##STR00020##
[0250] FIG. 1 shows the physical characteristics of inventive methyl paraffin based low viscosity base stocks and the comparative low viscosity base stocks. From FIG. 1, it can be seen that the 30/70 blend of C10 dimer and C10 trimer has a higher VI than GTL, Group III and Group II basestocks and lower Noack volatility than GTL and Group II of the same kinematic viscosity at 100.degree. C. The 30/70 blend of C10 dimer and C10 trimer has also improved low temperature properties than the neat C10 dimer and C12 dimer as illustrated by the lower pour point. In addition, the 30/70 blend of C10 dimer and C10 trimer provided a 26% to 158% improvement relative to low viscosity Group II, Group III and GTL comparative base stocks in terms of MTM traction coefficient, which may correlate directly with fuel and energy efficiency. This improvement in MTM traction coefficient for the 30/70 blend of C10 dimer and C10 trimer low viscosity base stock blend was surprising and unexpected relative to the low viscosity Group II, Group III and GTL comparative base stocks.
Example 2
Physical Characteristics and Performance Results of Inventive and Comparative Engine Oils
[0251] FIG. 2 shows the physical characteristics and performance test results of engine oils formulations including the inventive methyl paraffin low viscosity based base stocks and comparative low viscosity base stocks. From FIG. 2, it can be seen that engine oil formulations including the 30/70 blend of C10 dimer and C10 trimer as the low viscosity base stock provided a 45 to 105% improvement relative to engine oils formulations including low viscosity Group II, Group III and GTL comparative base stocks in terms of MTM traction coefficient, which correlates directly with fuel efficiency. This improvement in MTM traction coefficient for the engine oil with the 30/70 blend of C10 dimer and C10 trimer low viscosity base stock was surprising and unexpected relative to the low viscosity engine oils including comparative Group II, Group III and GTL base stocks. From FIG. 2, it can also be seen that the engine oil formulations including the 30/70 blend of C10 dimer and C10 trimer as the low viscosity base stock also provided lower Noack volatility, lower TEOST 33C deposits, higher 210 hour oxidation stability, higher Viscosity Index, lower HTHS and lower CCS viscosity relative to the low viscosity engine oils including comparative Group II, Group III and GTL base stocks. In fact, the TEOST 33C deposits of the engine oil formulations including the 30/70 blend of C10 dimer and C10 trimer as the low viscosity base stock was significantly lower than the TEOST 33C deposits of low viscosity engine oils including either C10 dimer or C10 trimer as the low viscosity base stock. Thus there are synergies in engine oil physical characteristics and performance upon blending of the C10 dimer and C10 trimer. The same synergistic improvement in blending also applies for the CCS viscosity as the engine oil formulations including the 30/70 blend of C10 dimer and C10 trimer was lower in CCS viscosity than engine oils including either C10 dimer or C10 trimer as the low viscosity base stock. Hence the blend of the C10 dimer and C10 trimer as the low viscosity base stock in engine oil formulations also provides unexpected improvements in engine oil physical and performance characteristics. It should be noted that for all the inventive and comparative engine oil formulations in FIG. 2, there was no viscosity modifier included in the formulations.
Example 3
[0252] Viscometric Characteristics of Inventive and Comparative Engine Oils with Viscosity Modifier
[0253] FIG. 3 shows kinematic viscosity and viscosity index of engine oils formulations including the inventive methyl paraffin based low viscosity base stock (30/70 blend of C10 dimer and C10 trimer) and comparative low viscosity base stocks (GTL and Yubase 3) which also include viscosity modifier. The two viscosity modifiers tested were a PMA (Viscoplex 3-200 VM) and HC (Infineum SV600 hydrocarbonVM). The treat rate of each of the viscosity modifiers in the engine oil formulations was adjusted to achieve a measured HTHS (high Temperature and High Shear) viscosity of 1.9 cP for the PMA and 2.2 cP for the HC, using test method ASTM D4663. Comparing FIG. 3 to FIG. 2, it can be seen that the viscosity modifier dramatically increases the Viscosity Index of all engine oil formulations, however, higher viscosity index was observed for the C10 dimer and C10 trimer mixture of the invention, resulting in lower KV40, indicating a potential fuel economy advantage.
Example 4
Noack Volatility Versus KV40 of Inventive and Comparative Base Stocks
[0254] FIG. 4 is a graph of the relationship between Noack volatility and kinematic viscosity at 40 deg. C for various mixtures of the C10 dimer and the C10 trimer (low viscosity inventive methyl paraffin base stocks) and also various mixtures of 2 cSt and 3.6 cSt comparative conventional PAOs. Also indicated on the graph are the relationship between Noack volatility and kinematic viscosity at 40 deg. C for the neat C10 dimer, C10 trimer, conventional 2 cSt PAO, conventional 3.6 cSt PAO, GTL (Shell QHVI 3), Group II (ExxonMobil EHC 20), and Group III (SK Oil Yubase 3). FIG. 5 shows the base stock blend ratios for the inventive mixtures of C10 dimer and C10 trimer of the inventive methyl paraffin based low viscosity base stock blends and the comparative mixtures of 2 cSt and 3.6 cSt conventional PAOs of the comparative PAO low viscosity base stock blends of FIG. 4.
[0255] FIG. 4 shows that the inventive methyl paraffin based low viscosity base stock blends of C10 dimer and C10 trimer have a relationship between Noack volatility (y) and KV40 (x) that is less than y=2.15-0.765*ln(x). In contrast, the comparative blends of 2 cSt and 3.6 cSt conventional PAOs have a relationship between Noack volatility (y) and KV40 (x) that is greater than y=2.15-0.765*ln(x). Therefore, for a given KV40, a base stock blend of C10 dimer and C10 trimer has a lower Noack Volatility than a conventional blend of a conventional 2 cSt PAO and a conventional 3.6 cSt PAO. This improvement and relationship between Noack volatility and kinematic viscosity at 40 deg. C for inventive base stock blends of C10 dimer and C10 trimer is surprising and unexpected.
PCT and EP Clauses:
[0256] 1. A lubricating oil base stock comprising from 5 to 50 wt % of 9-methylnonadecane and from 95 to 50 wt % of 9-methyl-11-octylheneicosane, wherein the base stock has a relationship between Noack volatility at 250.degree. C. as measured by ASTM D5800 (y) and kinematic viscosity at 40.degree. C. as measured by ASTM D445 (x) that is less than y=2.15-0.765*ln(x).
[0257] 2. The lubricating oil base stock of clause 1, wherein the base stock has a kinematic viscosity at 100.degree. C. as measured by ASTM D445 of from 1.5 to 3.5 cSt.
[0258] 3. The lubricating oil base stock of clauses 1-2, wherein the base stock has a kinematic viscosity at 40.degree. C. as measured by ASTM D445 of from 4.0 to 14.0 cSt.
[0259] 4. The lubricating oil base stock of clauses 1-3, wherein the base stock has a Noack volatility at 250.degree. C. as measured by ASTM D5800 of 10 to 90%.
[0260] 5. The lubricating oil base stock of clauses 1-4, wherein the base stock has a Viscosity Index from about 100 to 170 as determined by ASTM D2270.
[0261] 6. The lubricating oil base stock of clauses 1-5, wherein the base stock has a pour point of from about -10 to -80.degree. C. as determined by ASTM D5950.
[0262] 7. The lubricating oil base stock of clauses 1-6, wherein the base stock has a MTM average traction coefficient (at 100 deg. C, 1 GPa, 2 m/s and 0-100% SRR) of from about 0.0060 to 0.0090.
[0263] 8. The lubricating oil base stock of clauses 1-7, wherein the base stock has a MTM average traction coefficient (at 100 deg. C, 1 GPa, 2 m/s and 0-100% SRR) that is about 20 to 180% lower than a Group II or Group III base stock of comparable KV100.degree. C. viscosity.
[0264] 9. The lubricating oil base stock of clauses 1-8, wherein the base stock has a high temperature high shear (HTHS) viscosity of less than about 1.6 cP as determined by ASTM D4683, and a Noack volatility from about 16 to about 30 percent as determined by ASTM D5800.
[0265] 10. The lubricating oil base stock of clauses 1-9 further including a minor amount of one or more additives to form a lubricating oil.
[0266] 11. The lubricating oil base stock of clause 10, wherein the one or more additives are selected from the group consisting of a viscosity improver or modifier, antioxidant, detergent, dispersant, pour point depressant, corrosion inhibitor, metal deactivator, seal compatibility additive, anti-foam agent, inhibitor, anti-rust additive and combinations thereof.
[0267] 12. The lubricating oil base stock of clauses 10-11 wherein the lubricating oil has an oxidation stability (210 hour test with time to 200% KV40 increase) of from about 60 to 150 hours.
[0268] 13. The lubricating oil base stock of clauses 10-12 wherein the lubricating oil has resistance to deposit formation as measured by TEOST 33C test for 2 hours at 200 to 480 deg. C per ASTM D6335 of from 10 to 30 mg.
[0269] 14. The lubricating oil base stock of clauses 10-13 wherein the lubricating oil has a Cold Crank Simulator (CCS) viscosity at -35 deg. C per ASTM D5293 of from 700 to 1000 mPas.
[0270] 15. The lubricating oil base stock of clauses 10-14 wherein the one or more additives include a viscosity modifier selected from a polymethacrylate, a hydrocarbon hydrogenated polyisoprene star polymer and combinations thereof.
[0271] 16. The lubricating oil base stock of clause 15 wherein the lubricating oil has a high temperature high shear (HTHS) viscosity of less than about 2.3 cP as determined by ASTM D4683.
[0272] 17. The lubricating oil base stock of clauses 15-16 wherein the lubricating oil has a Viscosity Index from about 220 to 340 as determined by ASTM D2270.
[0273] 18. The lubricating oil base stock of clauses 10-17 wherein the lubricating oil further includes a cobase stock at from 5 to 40 wt %, wherein the cobase stock is selected from the group consisting of a Group I base stock, a Group II base stock, a Group III base stock, a Group IV base stock, a Group V base stock and combinations thereof.
[0274] 19. A method for improving one or more of thermal and oxidative stability, deposit control and traction control in a lubricating oil comprising: providing a lubricating oil including a major amount of a lubricating oil base stock and a minor amount of one or more additives, said lubricating oil base stock comprising from 5 to 50 wt % of 9-methylnonadecane and from 95 to 50 wt % of 9-methyl-11-octylheneicosane, wherein the base stock has a relationship between Noack volatility at 250.degree. C. as measured by ASTM D5800 (y) and kinematic viscosity at 40.degree. C. as measured by ASTM D445 (x) that is less than y=2.15-0.765*ln(x), and using the lubricating oil in a formulated oil to improve one or more of thermal and oxidative stability, deposit control and traction control.
[0275] All patents and patent applications, test procedures (such as ASTM methods, UL methods, and the like), and other documents cited herein are fully incorporated by reference to the extent such disclosure is not inconsistent with this disclosure and for all jurisdictions in which such incorporation is permitted.
[0276] When numerical lower limits and numerical upper limits are listed herein, ranges from any lower limit to any upper limit are contemplated. While the illustrative embodiments of the disclosure have been described with particularity, it will be understood that various other modifications will be apparent to and can be readily made by those skilled in the art without departing from the spirit and scope of the disclosure. Accordingly, it is not intended that the scope of the claims appended hereto be limited to the examples and descriptions set forth herein but rather that the claims be construed as encompassing all the features of patentable novelty which reside in the present disclosure, including all features which would be treated as equivalents thereof by those skilled in the art to which the disclosure pertains.
[0277] The present disclosure has been described above with reference to numerous embodiments and specific examples. Many variations will suggest themselves to those skilled in this art in light of the above detailed description. All such obvious variations are within the full intended scope of the appended claims.
* * * * *
References




















D00001

D00002

D00003

D00004

D00005











XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.