Process For Continuously Converting Mixed Waste Plastic Into Waxes And Liquid Fuels By Cracking
STREIFF; Stephane ; et al.
U.S. patent application number 16/062681 was filed with the patent office on 2018-12-27 for process for continuously converting mixed waste plastic into waxes and liquid fuels by cracking. The applicant listed for this patent is SOLVAY SA. Invention is credited to Dominique BALTHASART, Miriam CERRO-ALARCON, Avelino CORMA, Michel GARRAIT, Philippe MARION, Jes s MENGUAL, Marco PICCININI, Stephane STREIFF.
| Application Number | 20180371325 16/062681 |
| Document ID | / |
| Family ID | 54936879 |
| Filed Date | 2018-12-27 |





| United States Patent Application | 20180371325 |
| Kind Code | A1 |
| STREIFF; Stephane ; et al. | December 27, 2018 |
PROCESS FOR CONTINUOUSLY CONVERTING MIXED WASTE PLASTIC INTO WAXES AND LIQUID FUELS BY CRACKING
Abstract
The present invention relates to a process for continuously converting mixed waste plastic into waxes and liquid fuels by cracking. The process comprises the steps of feeding a mixed waste plastic stream to a cracking reactor where the mixed waste plastic is catalytically cracked in the presence of a catalyst and circulating the catalyst between the cracking reactor and a regenerator where the catalyst received from the cracking reactor is regenerated and heated by burning coke and/or other combustible material deposited on or mixed with the catalyst.
| Inventors: | STREIFF; Stephane; (Shanghai, CN) ; BALTHASART; Dominique; (Brussels, BE) ; PICCININI; Marco; (Brussels, BE) ; MARION; Philippe; (Vernaison, FR) ; GARRAIT; Michel; (Charly, FR) ; CORMA; Avelino; (Valencia, ES) ; CERRO-ALARCON; Miriam; (Valencia, ES) ; MENGUAL; Jes s; (Carcaixent, ES) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 54936879 | ||||||||||
| Appl. No.: | 16/062681 | ||||||||||
| Filed: | December 15, 2016 | ||||||||||
| PCT Filed: | December 15, 2016 | ||||||||||
| PCT NO: | PCT/EP2016/081310 | ||||||||||
| 371 Date: | June 15, 2018 |
| Current U.S. Class: | 1/1 |
| Current CPC Class: | C10G 1/10 20130101; C10G 2300/4081 20130101; C10G 11/187 20130101; C10G 65/12 20130101; C10G 1/086 20130101; C10G 1/083 20130101; C10G 2300/708 20130101; C10G 47/36 20130101 |
| International Class: | C10G 1/08 20060101 C10G001/08; C10G 11/18 20060101 C10G011/18; C10G 47/36 20060101 C10G047/36; C10G 65/12 20060101 C10G065/12 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Dec 18, 2015 | EP | 15201142.5 |
Claims
1. A process for continuously converting mixed waste plastic into waxes and liquid fuels by cracking, the process comprising the steps of: feeding a mixed waste plastic stream to a cracking reactor where the mixed waste plastic is catalytically cracked in the presence of a catalyst; circulating the catalyst between the cracking reactor and a regenerator where the catalyst received from the cracking reactor is regenerated and heated by burning coke and/or other combustible material deposited on or mixed with the catalyst; wherein the heat balance between the cracking reactor and the regenerator is adjusted depending on changes in the chemical composition of the mixed waste plastic.
2. The process according to claim 1 wherein the catalyst is a mixture of at least two different catalysts having different catalytic activity and the heat balance between the cracking reactor and the regenerator is adjusted by adjusting the ratio of the at least two different catalysts in the mixture of catalysts.
3. The process according to claim 1 wherein the heat balance between the cracking reactor and the regenerator is adjusted by diluting the mixed waste plastic with a diluent.
4. The process according to claim 3 wherein the mixed waste plastic in the feed stream is diluted with waxes and/or liquid fuels.
5. The process according to claim 4 wherein the waxes and liquid fuels are obtained from the cracking process.
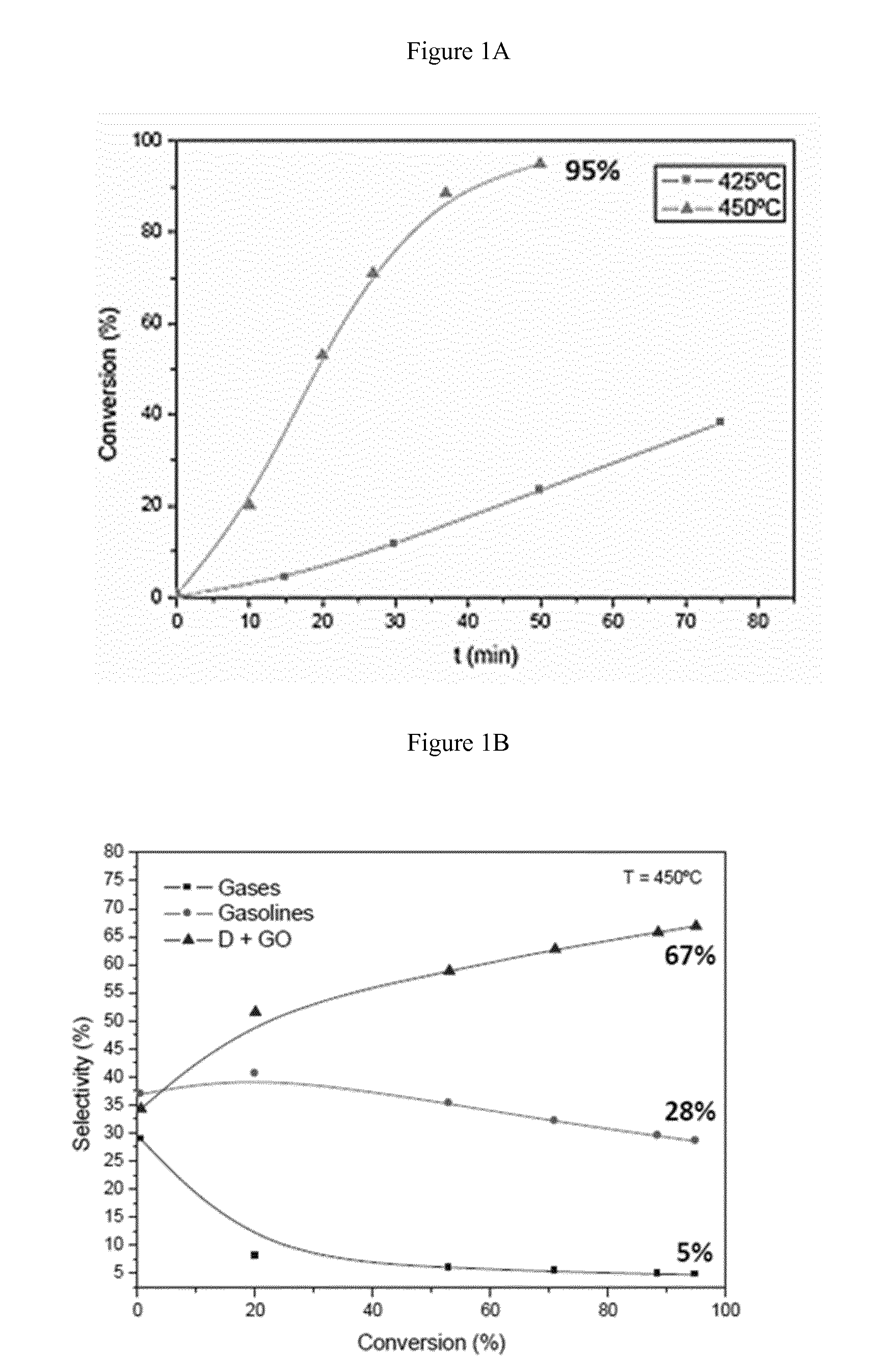
6. The process according to claim 1 wherein the heat balance between the cracking reactor and the regenerator is adjusted by introducing a combustible stream into the regenerator.
7. The process according to claim 1 wherein the heat balance between the cracking reactor and the regenerator is adjusted such that the cracking reactor and the regenerator are maintained in a steady state heat balance.
8. The process according to claim 1 wherein the heat balance between the cracking reactor and the regenerator is adjusted such that a predetermined ratio of waxes to liquid fuels is obtained from the cracking reactor.
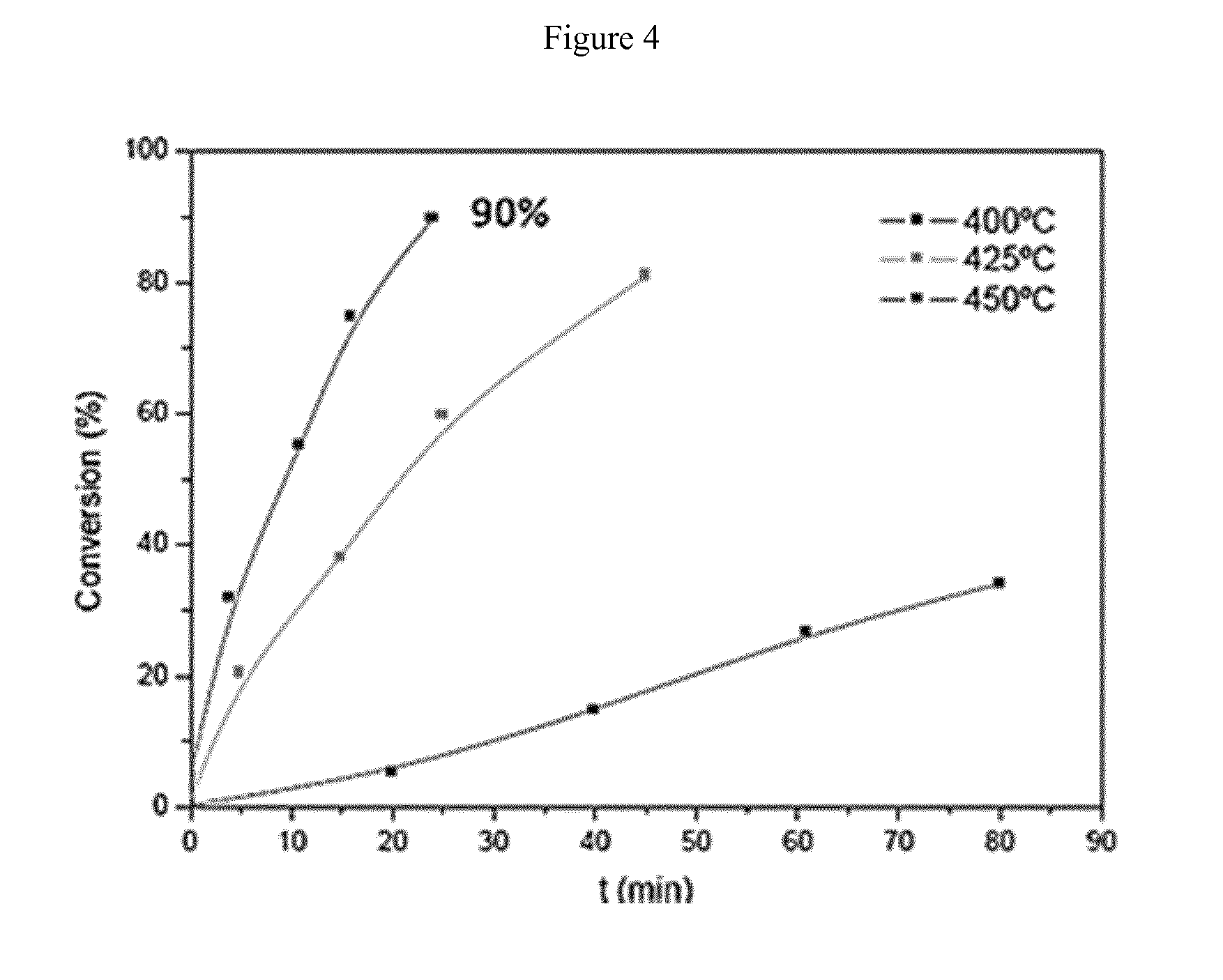
9. The process according to claim 1 wherein temperature and flow of the circulating catalyst are adjusted to maintain a pre-selected temperature in the cracking reactor.
10. The process according to claim 1 wherein the chemical composition of the mixed waste plastic changes over time.
11. The process according to claim 1 wherein the mixed waste plastic comprises more than 50% by weight of polyolefin and polystyrene based on the total weight of the mixed waste plastic.
12. The process according to claim 1 wherein the mixed waste plastic is subjected to a dry pretreatment prior to subjecting it to the cracking reactor.
13. The process according to claim 1 wherein the mixed waste plastic prior to feeding it to the cracking reactor has a water content of less than 20% by weight, based on the total weight of the mixed waste plastic.
14. The process according to claim 1, further comprising the step of reducing the content of air and/or oxygen in the mixed waste plastic prior to feeding it to the cracking reactor.
15. The process according to claim 1 wherein the oxygen content in the cracking reactor is lower than 10 vol. % of the gas phase in the reactor.
16. The process according to claim 13 wherein the mixed waste plastic prior to feeding it to the cracking reactor has a water content of less than 10% by weight, based on the total weight of the mixed waste plastic.
17. The process according to claim 15 wherein the oxygen content in the cracking reactor is lower than 5 vol. % of the gas phase in the reactor.
Description
[0001] This application claims priority to European application No. 15201142.5--filed on Dec. 18, 2015--, the whole content of this application being incorporated herein by reference for all purposes.
[0002] The present invention relates to a process for continuously converting mixed waste plastic into waxes and liquid fuels by cracking. The process comprises the steps of feeding a mixed waste plastic stream to a cracking reactor where the mixed waste plastic is catalytically cracked in the presence of a catalyst and circulating the catalyst between the cracking reactor and a regenerator where the catalyst received from the cracking reactor is regenerated and heated by burning coke and/or other combustible material deposited on or mixed with the catalyst.
[0003] In view of the increasing importance of polymers as substitutes for conventional materials of construction such as glass, metal, paper, and wood, the perceived need to convert non-renewable resources such as petroleum and dwindling amounts of landfill capacity available for the disposal of waste products, considerable attention has been devoted in recent years to the problem of recovering, reclaiming, recycling or in some way reusing waste plastic.
[0004] It has been proposed to pyrolyze or catalytically crack the waste plastic so as to convert high molecular weight polymers into volatile compounds having a much lower molecular weight. The volatile compounds, depending on the process employed, can be either relatively high boiling liquid hydrocarbons useful as fuel oils or fuel oil supplements or light to medium boiling hydrocarbons useful as gasoline-type fuels or as other chemicals.
[0005] The cracking reaction is endothermic. At the same time, the catalyst used in the cracking reaction is contaminated with coke and other combustible material. It is therefore common to circulate the catalyst between the cracking reactor and a regenerator where the catalyst is regenerated and heated by burning the coke and other combustible material. This burning reaction is exothermic and when the thus heated catalyst is recycled into the cracking reactor provides the energy for the cracking reaction. For that reason, catalytic cracking/regenerator units are often referred to as being "heat balanced".
[0006] It is for example common to operate the reactor at constant raw material feed modulating the catalyst to feed ratio in order to keep the temperature constant. In an alternative approach, for example US 2014/0228204 suggests periodically purging a small portion of the used catalyst and make that up with fresh catalyst in order to maintain the catalyst activity at a constant level.
[0007] U.S. Pat. No. 5,216,149 discloses a method for controlling the pyrolysis of a complex waste stream of plastics to convert thus stream into useful high value monomers or other chemicals, by identifying catalyst and temperature conditions that permit decomposition of a given polymer in the presence of others, without substantially decomposition of the other polymers. This method makes it easier to purify the monomer from the easier to decompose plastic. This process does, however, not allow to react on changes in the waste stream of plastics over time.
[0008] U.S. Pat. No. 5,904,879 suggests recycling portions of the hot cracked oil stream to a melting vessel to serve as the melting medium for the waste plastic materials. The hot oil stream is recycled at a set weight ratio of hot oil stream to bulk waste plastic materials.
[0009] The known processes intend to maintain a balance between the endothermic cracking reaction in the cracking reactor and the exothermic combustion reaction in the catalyst regenerator. These methods are effective as long as the waste plastic feed has a constant chemical composition, i.e. the waste plastic consists of only one type of polymer or a mixture of polymers which is constant over time. However, the known actions are not effective if mixed waste plastic is employed in a continuous process. This is because mixed waste plastic composition varies over time according to the market. Moreover, the exact plastic content of mixed waste plastic varies with the geographic origin of the waste and also the time of origin. For example, the composition of post-consumer waste plastic during summer time is different to the composition of the waste plastic in winter time. However, a varying composition of the mixed waste plastic creates a heat imbalance in the cracking reactor/regenerator system because different polymers require different amounts of energy for cracking and thus varying compositions of mixed waste plastic will require increasing or decreasing amounts of energy in the cracking reactor. As consequence thereof, the prior art processes which for example aim at maintaining the catalyst activity at a constant level, require an auxiliary heat sink and/or an auxiliary heat supply if mixed waste plastic is to be cracked in a continuous process. This decreases the energy yield of the process, increases the CO.sub.2 footprint and increases the overall cost.
[0010] There is therefore still a need for a process for cracking mixed waste plastic which does not have the above disadvantages. In particular, it would be desirable to have a process which can continuously crack mixed waste plastic which changes its composition over time without the requirement of any auxiliary heat sink or auxiliary heat supply. Furthermore, it would be desirable to have a process for continuously cracking mixed waste plastic which can balance the changes in energy required for the endothermic cracking reaction which are due to the changes in the composition of the mixed waste plastic.
[0011] It has now been found that the above problems can be solved by adjusting the heat balance between the cracking reactor and the regenerator depending on changes in the chemical composition of the mixed waste plastic. The present invention therefore relates to a process for continuously converting mixed waste plastic into waxes and liquid fuels by cracking, the process comprising the steps of [0012] feeding a mixed waste plastic stream to a cracking reactor where the mixed waste plastic is catalytically cracked in the presence of a catalyst; [0013] circulating the catalyst between the cracking reactor and a regenerator where the catalyst received from the cracking reactor is regenerated and heated by burning coke and/or other combustible material deposited on or mixed with the catalyst; characterized in that the heat balance between the cracking reactor and the regenerator is adjusted depending on changes in the chemical composition of the mixed waste plastic.
[0014] The present process has the following advantages: [0015] be less selective on the plastic sorting process. By accepting a lower quality of plastic materials, more recoverable materials can be recycled on the sorting steps. [0016] Currently the remaining wastes, after the sorting process, are mainly composed of plastics, paper, board, glass, food residue, etc. This fraction called solid recovered fuel (SRF) can only be valorized as energy in the cement furnace due to their high calorific value. Removing more plastic from the incoming feed allows an access to a higher volume of raw material, but also, to decrease the calorific value of the SRF and thus valorized as energy in the conventional incinerators. [0017] Less plastics will end up in landfill sites [0018] Less CO.sub.2 is emitted per unit of fuel and Capex of the unit is lower.
[0019] In the context of the present invention, waxes are to be understood as a mixture of hydrocarbons optionally comprising heteroatoms, such as O, N, etc., being solid at room temperature (23.degree. C.) and having a softening point of generally above 45.degree. C. Liquid fuels are to be understood as combustible liquid hydrocarbons optionally comprising heteroatoms, such as O, N, etc., being liquid at room temperature, such as gasoline, kerosene and diesel oil.
[0020] The invention therefore allows producing valuable chemicals from mixed waste plastic, such as post-consumer waste plastic, off spec plastic, industrial scrap plastic and the like.
[0021] A plastic is mostly constituted of a particular polymer and the plastic is generally named by this particular polymer. Preferably, a plastic contains more than 25% by weight of its total weight of the particular polymer, preferably more than 40% by weight and more preferably more than 50% by weight.
[0022] Other components in plastic are for example additives, such as fillers, reinforcers, processing aids, plasticizers, pigments, light stabilizers, lubricants, impact modifiers, antistatic agents, inks, antioxidants, etc. Generally, a plastic comprises more than one additive.
[0023] Plastics suitable in the process of the present invention are for example polyolefins and polystyrene, such as high density polyethylene (HDPE), low density polyethylene (LDPE), polypropylene (PP) and polystyrene. Mixed plastics mostly constituted of polyolefin and polystyrene are preferred. In this context "mostly constituted" is to be understood such that the concentration of the polyolefin and the polystyrene in the mixed plastic is above 50% by weight, more preferably above 75% by weight, each based on the total weight of the dry mixed plastic. The mixed plastic may be constituted of polyolefin and polystyrene. Preferably, the mixed plastic contains less than 99.5% by weight, more preferably less than 99% by weight of polyolefin and polystyrene, based on the total weight of the dry mixed plastic.
[0024] Other plastics, such as polyvinylchloride, polyvinylidene chloride, polyethylene terephthalate, polyurethane (PU), acrylonitrile-butadiene-styrene (ABS), nylon and fluorinated polymers are less desirable. If present in the waste plastic, they are preferably present in a minor amount of less than 50% by weight, preferably less than 30% by weight, more preferably less than 20% by weight, even more preferably less than 10% by weight of the total weight of the dry waste plastic. Preferably, the individual content of any less desirable plastic is less than 5% by weight, more preferably less than 2% by weight based on the total weight of the dry waste plastic.
[0025] Usually, waste plastic contains other non-desired components, namely foreign material, such as paper, glass, stone, metal, etc.
[0026] In the process of the present invention the chemical composition of the mixed waste plastic will change over time. In this context, "chemical composition" refers to the polymers present in the waste plastic. The above described additives and non-desired components in this context do not add to the chemical composition of the waste plastic. In other words, the chemical composition changes if the polymers constituting the waste plastic or their ratios to each other in the waste plastic change.
[0027] As different polymers require different amounts of energy for cracking, a change in the chemical composition of the mixed waste plastic results in a heat imbalance in the cracking reactor/regenerator system. In this regard, "cracking reactor" is to be understood as that part of the system where heat is consumed (mainly due to the endothermic cracking reaction but also for other parts of the process, such as the evaporation of the resulting products). "Regenerator" is to be understood as that part of the system where heat is produced by burning coke and/or other combustible material deposited on or mixed with the used catalyst received from the cracking reactor.
[0028] According to the invention the heat balance between the cracking reactor and the regenerator is adjusted, preferably automatically adjusted, depending on changes in the chemical composition of the mixed waste plastic. Thus, the invention allows to avoid heat imbalances in the cracking reactor/regenerator system.
[0029] The temperature obtained in the reactor in case of adiabatic operation is the result of balance between the hot catalyst (flow and temperature) and the cold fluxes (plastic mix heating, cracking and vaporization of the products). The overall cracking reaction is endothermic and its extend results from the kinetic mainly influence by the catalyst nature, catalyst amount (catalyst/plastic ratio) and the temperature. The catalyst nature influence the selectivity and activity of the cracking (in term of gaseous, gasoline, diesel, kerosene, wax and coke and unconverted plastic fractions) the catalyst amount and the temperature.
[0030] In the process of the present invention the heat balance between the cracking reactor and the regenerator can be adjusted by adjusting the above parameters. The present inventors have, however, found that in preferred embodiments the heat balance can be adjusted by using a mixture of at least two different catalysts having different catalytic activity and adjusting the ratio of the at least two different catalysts in the mixture. Thus, the overall catalytic activity of the mixture can be increased or decreased depending on the changes in the chemical composition of the mixed waste plastic.
[0031] Adjusting the heat balance between the cracking reactor and the regenerator can be accomplished by adjusting the ratio of at least two different catalysts having different catalytic activity in a mixture of catalysts being circulated between the cracking reactor and the regenerator. Such mixture of at least two different catalysts can comprise highly active FCC catalysts known to the person skilled in the art and being commercially available. Examples for highly active FCC catalysts are crystalline microporous zeolites like ZSM-5, ZSM-11, ZSM-22, ZSM-23, ZSM-35, ZSM-48, ZSM-50, TS-1, TS-2, SSZ-46, MCM-22, MCM-49, FU-9, PSH-3, ITQ-1, EU-1, NU-10, silicalite-1, silicalite-2, boralite-C, boralite-D, BCA and mixtures thereof. Less active FCC catalysts are also known to the person skilled in the art. Examples for less active FCC catalysts are SiO.sub.2 (sand) and kaolin. Preferred materials comprise at least 60% by weight, preferably at least 70% by weight and even more preferably at least 80% by weight of silica-equivalent of an oxidic compound based of silicon like Silica (SiO.sub.2), kaolin, etc. FCC catalysts can be fresh catalysts, equilibrated catalysts (such as spent catalysts), or mixtures thereof.
[0032] The influence of different catalytic activities of different catalysts on the cracking of different polymers is now explained in more detail with reference to the attached figures which show in
[0033] FIGS. 1A and 1B the effect of the reaction temperature on the cracking of LDPE in the presence of SiO.sub.2,
[0034] FIGS. 2A and 2B the effect of the reaction temperature on the cracking of HDPE in the presence of SiO.sub.2,
[0035] FIGS. 3A and 3B the effect of the reaction temperature on the cracking of HDPE in the presence of ECAT-1C, and
[0036] FIGS. 4, 5A, 5B and 5C the effect of the reaction temperature on the cracking of PP in the presence of SiO.sub.2.
[0037] FIG. 1A shows the conversion rate of LDPE over time at 425.degree. C. and 450.degree. C., respectively. It is apparent that at 450.degree. C. the conversion is nearly complete after about 50 minutes. FIG. 1B depicts the selectivity of the conversion at 450.degree. C. Surprisingly, with increasing conversion rate the gas fraction decreases, the gasoline fraction slightly decreases and the wax fraction (diesel (D) and gasoil (GO)) increases.
[0038] Basically, the same effects are observed when cracking HDPE in the presence of SiO.sub.2 (see FIGS. 2A and 2B).
[0039] Interestingly, the above effects are not observed when using a catalyst having higher catalytic activity than SiO.sub.2. This is demonstrated by using the highly active equilibrated FCC catalyst ECAT-1C for cracking HDPE. The results are shown in FIGS. 3A and 3B. FIG. 3A shows that even at low temperature the conversion is fast. However, as can be seen in FIG. 3B, at all temperatures the selectivity between gases, gasolines and waxes is about the same and in particular the gasoline fraction is much higher than the wax fraction which is contrary to the effect observed with the less reactive catalyst SiO.sub.2.
[0040] Finally, FIGS. 4, 5A, 5B and 5C relate to the cracking of PP using SiO.sub.2 as catalyst. FIG. 4 demonstrates that the conversion is fast and nearly complete at 425.degree. C. already. However, as shown in FIG. 5A, at 400.degree. C. the main fraction is the gasoline fraction while at 450.degree. C. (FIG. 5C) the main fraction is the wax fraction.
[0041] Thus, catalysts of a different catalytic activity have different effects on the cracking of different polymers. As a consequence thereof, it is possible to adjust the heat balance between the cracking reactor and the regenerator by adjusting the ratio of different catalysts having different catalytic activity in a mixture of catalysts circulated between the cracking reactor and the regenerator.
[0042] For example the plastic mix is introduced at a defined temperature and flow in a reaction chamber operating adiabatically. A hot catalyst stream is introduced in the reaction chamber at a relative flow rate versus the plastic mix in such a way to obtain the fixed temperature. The catalyst, the coke and the unconverted material are sent to a regenerator where air is introduced and the coke and unconverted material are burnt rising the temperature of the catalyst. A quantity of the catalyst is extracted from the regenerator and replaced by an amount of lower activity or higher activity catalyst in such a way to keep the temperature level in the reaction chamber at the desired value. Without willing to be limited by a theory, increasing the low activity catalyst content will induce an increase of the catalyst flow to keep the average activity resulting in an increase of the heat supplied and increase the reaction chamber temperature while increasing the high activity catalyst fraction will induce a decrease of the catalyst flow resulting in a decrease of the heat supplied and decrease the temperature in the reaction chamber allowing to control the temperature at the desired level.
[0043] In an alternative embodiment the inventors found that the heat balance between the cracking reactor and the regenerator can be adjusted by diluting the mixed waste plastic with a diluent. Such diluent participates also to the heat balance of the system and therefore is a suitable parameter for adjusting the heat balance between the cracking reactor and the regenerator. Preferred diluents are waxes, liquid fuels and mixtures thereof. Most preferred the waxes and liquid fuels are obtained from the cracking process. Diluting can take place in the feed stream i.e. prior to feeding the mixed waste plastic into the cracking reactor, or in the cracking reactor, i.e. after feeding the mixed waste plastic into the cracking reactor.
[0044] Preferred diluents are liquid products from the cracking process having an atmospheric boiling point in the range of 25 to 250.degree. C., more preferably in the range of 50 to 150.degree. C. The diluent is generally used at a ratio of 0.1 to 10 kg per kg of mixed waste plastic, preferably at a ratio of 0.5 to 5 kg per kg of mixed waste plastic and more preferably at a ratio of 0.7 to 2 kg per kg of mixed waste plastic.
[0045] The diluent is introduced preferably at a temperature below its flash point. When the mixed waste plastic is not melted, the diluent and the mixed waste plastic may form a slurry. When the mixed waste plastic is melted, the diluent allows reducing the viscosity of the melt, easing by this way its cleaning and processing. By cleaning removal of foreign materials is understood.
[0046] For example the plastic mix is introduced at a defined temperature and flow in a reaction chamber operating adiabatically. A hot catalyst stream is introduced in the reaction chamber at a defined relative flow rate versus the plastic mix. An auxiliary hydrocarbon liquid at a defined temperature is introduced in the reaction chamber at a flowrate such that the temperature in the reaction chamber reaches a desired value. The catalyst, the coke and the unconverted material are sent to a regenerator where air is introduced and the coke and unconverted material are burnt rising the temperature of the catalyst. Without willing to be limited by a theory, it is thought that the excess heat is absorbed mostly by the evaporation of the auxiliary liquid allowing to control the temperature in the reaction chamber.
[0047] Alternatively, a mixture of the plastic mix with a recycled hydrocarbon liquid is introduced in a reaction chamber operating adiabatically. A hot catalyst stream is introduced in the reaction chamber at a defined relative flow rate versus the plastic mix. The catalyst, the coke and the unconverted material are sent to a regenerator where air is introduced and the coke and unconverted material are burnt rising the temperature of the catalyst.
[0048] In a further alternative embodiment the inventors found that the heat balance between the cracking reactor and the regenerator can be adjusted by introducing a combustible stream into the regenerator. Such combustible stream participates also to the heat balance of the system and therefore is a suitable parameter for adjusting the heat balance between the cracking reactor and the regenerator.
[0049] Preferred combustible streams are hydrocarbon stream from the cracking process having an atmospheric boiling point lower than 250.degree. C., more preferably lower than 50.degree. C. The gaseous stream produced after the condensation of the liquid fraction of the cracked gas is particularly preferred. The combustible stream is generally used at a ratio of 0.001 to 1 kg per kg of mixed waste plastic, preferably at a ratio of 0.002 to 0.25 kg per kg of mixed waste plastic and more preferably at a ratio of 0.005 to 0.15 kg per kg of mixed waste plastic.
[0050] The combustible stream is introduced in the regenerator by any way known in the art. Examples of introduction are dedicated dip pipe and spray nozzle. Alternatively, the combustible stream could be mixed with the coked catalyst stream before introduction in the regenerator. A gaseous combustible gas stream recovered at the end of the condensation used as fluidizing medium to transport the coked catalyst is particularly preferred.
[0051] All three measures (mixing catalysts, using a diluent and introducing a combustible stream) may be taken alone or in combination of two or three of these measures.
[0052] In the process of the present invention, the waste plastic can be subjected to a pretreatment. The pretreatment includes for example size reduction and foreign material removal. In the prior art processes in particular foreign material removal is often conducted in the presence of or using water. The thus obtained pretreated waste plastic is rather wet and requires either time and energy consuming drying or results in potential problems during further processing, such as corrosion. The present inventors additionally found that also the heat balance and coke formation in the cracking reactor can be impaired by the presence of water which vaporizes thereby cooling the contents of the cracking reactor in addition to the endothermic cracking reaction. Thus, water entering the cracking reactor together with the waste plastic requires additional energy or may even lead to an un-uniform cooling within the reactor which may result in undesired coke formation on the catalyst particles. This may result in an increase in temperature when burning the coke in the regenerator which in turn may have an adverse influence on the overall energy balance of the system. Therefore, in a preferred embodiment of the present invention the pretreatment of the waste plastic is conducted as dry pretreatment.
[0053] Dry pretreatment is to be understood as a pretreatment in the absence of additional water. In this context, "additional water" is to be understood as water being present in addition to the water or moisture in the waste plastic before pretreatment. Thus, as pretreatment the waste plastic is for example not washed with water or separated from foreign material by floatation in water or an aqueous liquid.
[0054] Furthermore, dry pretreatment in the context of the present invention excludes any liquefying (e.g. melting or dissolving) of the waste plastic. Any liquefying step can optionally be conducted in addition to the dry pretreatment for example after the dry pretreatment and prior to subjecting the waste plastic to cracking.
[0055] Suitable dry pretreatment steps are for example size reduction by grinding or shredding and foreign material removal by separation by cycloning, air or gas elutriation, sieving and magnetic separation.
[0056] For example, during pretreatment the size of the mixed plastic pieces can be reduced to a suitable value to be handled. Waste plastic is available as "volume particles" having significant length in three directions, as "surface particles" having significant length in two directions and a much lower thickness or "line particles" having one major length and two minor dimensions. Examples of "volume particles" are pieces of shoe soles, car bumpers, residual plastic pieces from extrusion, etc. Examples of "surface particles" are pieces of bottles, bags, etc. Examples of "line particles" are wires, filaments, etc. For the volume particles the size if to be understood as the two larger dimensions of the particles, for surface particles, the larger of the two larger dimensions and for line particles, the larger dimension. Preferably, the waste plastic particles after size reduction have a maximum size of less than 100 mm, preferably less than 50 mm. Typical minimum size is 0.05 mm, preferably 0.1 mm. Suitable apparatuses for size reduction are known in the art.
[0057] Usually, the waste plastic includes some free water. "Free water" is to be understood as non-chemically bonded water. Usually, the water content of waste plastic is less than 20% by weight, preferably less than 10% by weight, each based on the total weight of the waste plastic. Since the pretreatment in the process of the present invention is a dry pretreatment, the water content of the waste plastic before and after pretreatment can be the same or the dry pretreatment can even reduce the water content of the waste plastic. The above preferred water contents of the waste plastic are preferably the water contents of the pretreated waste plastic.
[0058] Usually, the waste plastic is used in bulk with air enclosed in the bulk plastic. The present inventors found that also the presence of air and in particular oxygen in the waste plastic can adversely affect the cracking process. In particular, the presence of oxygen can be dangerous due to the risk of uncontrolled temperature increase or even explosion in the crack reactor. Therefore, it is preferred that the process comprises the further step of reducing the content of air and/or oxygen in the waste plastic prior to subjecting it to cracking. The content of air and/or oxygen in the waste plastic can for example be reduced by mechanical compression, applying vacuum, diluting the air by an inert gas, purging the waste plastic with an inert gas, and/or contacting the waste plastic with an oxygen scavenger. Suitable inert gases are nitrogen, carbon dioxide or combustion gases, combustion gases being preferred.
[0059] Furthermore, when a pneumatic transportation is used, which is preferred, as a transportation gas a suitable inert gas may be used. Suitable inert gases are nitrogen, carbon dioxide or combustion gases, combustion gases being preferred.
[0060] Preferably, the content of air in the waste plastic prior to subjecting it to cracking is lower than 10 g/kg of dry waste plastic, preferably lower than 5 g/kg of dry waste plastic.
[0061] In a further embodiment, the cracking is conducted at an oxygen content in the cracking reactor which is lower than 10 vol. % of the gas phase in the reactor, preferably lower than 5 vol. % of the gas phase in the reactor. The amount of oxygen in the reactor can be reduced by reducing the amount of air and/or oxygen in the supplied waste plastic.
[0062] After pretreatment, the plastic waste can be introduced in the cracking reactor by any suitable means known in the art. In one embodiment, the waste plastic is in solid state. For introducing solid waste plastic into the cracking reactor suitable means are screw conveyer, belt conveyer, pneumatic transportation, bucket elevator and flexiscrew (transitube). Screw conveyer and pneumatic transportation are preferred. Pneumatic transportation is preferably made using the inert gas as defined above. Preferably, by pneumatic transportation using an inert gas the oxygen content of the atmosphere surrounding the particles is reduced.
[0063] Before reacting, the waste plastic can be liquefied. Liquefaction can be conducted by any known means. Suitable means are heating, dissolving with a suitable solvent or a combination of heating and dissolving. Heating can be direct heating, indirect heating or a combination of both. Suitable direct heating are steaming, contact with hot gas, contact with hot liquid, and contact with hot solid. Suitable indirect heating are heat transfer through a surface, mechanical friction, etc. Heat transfer through a surface is the preferred indirect heating method.
[0064] During heating the waste plastic, if desired, can be azeotropically further dried with a suitable liquid. Examples of suitable liquids are hydrocarbons and in particular hydrocarbon mixes. Particularly suitable is a hydrocarbon mix produced by the pyrolysis of the waste plastic. Preferably, the light fraction of the fuel produced by the pyrolysis can be used to azeotropically further dry the waste plastic.
[0065] The liquefied waste plastic is usually a viscous liquid. In certain embodiments it can be convenient to reduce the viscosity of the liquefied waste plastic by adding a suitable diluent. A suitable diluent is a hydrocarbon mix, such as a hydrocarbon cut. A hydrocarbon cut is a mixture of hydrocarbons of various molecular weight mostly constituted with H and C optionally with minor amount of heteroatoms. Hydrocarbon cuts of any origin are suitable. Preferred is a hydrocarbon cut from the pyrolysis of plastic. Gasoline, kerosene, diesel or wax cut from pyrolysis of plastic or mixtures thereof are particularly preferred. Even more suitable are gasoline or wax cut from catalytic pyrolysis of mixed waste plastic. Gasoline cut is to be understood as a mixture mostly constituted of hydrocarbons having an atmospheric boiling point in the range of from 25 to 250.degree. C., preferably of from 40 to 250.degree. C., more preferably of from 50 to 150.degree. C. Kerosene cut is to be understood as a mixture mostly constituted of hydrocarbons having an atmospheric boiling point in the range of from 100 to 350.degree. C., preferably of from 150 to 250.degree. C. Diesel cut is to be understood as a mixture mostly constituted of hydrocarbons having an atmospheric boiling point in the range of from 250 to 500.degree. C., preferably of from 250 to 350.degree. C. Wax cut is to be understood as a mixture mostly constituted of hydrocarbons having an atmospheric boiling point over 300.degree. C., preferably over 350.degree. C. In this context, "mostly constituted" means that the cut is constituted of at least 95% by weight of said hydrocarbons, preferably by more than 99% by weight of said hydrocarbons.
[0066] Hydrocarbon cut is an organic phase which may contain water either dissolved, separated and/or in the form of an emulsion. The water content is preferably less than 5% by weight, more preferably less than 2% by weight. Particularly suitable is a hydrocarbon mix produced by the pyrolysis of waste plastic, in particular mixed waste plastic. During the melting of the waste plastic some decomposition may occur. For example, small molecules may be released during the polymer decomposition. Such small molecules often contain heteroatoms. Heteroatoms are atoms other than hydrogen and carbon. Examples of heteroatoms are O, Cl, Br, F, S and N. Addition of a scavenger for the heteroatoms can be useful to avoid corrosion induced by such heteroatoms and/or avoid fuel contamination. Examples of scavengers for heteroatoms are minerals, such as lime, soda lime, magnesia, silico alumina, alumina, silica.
[0067] During the liquefaction of the waste plastic some solid may remain. Such solid may be material having a melting temperature higher than the set temperature, may result from the decomposition of the plastic material or the foreign material, or may be a reaction product of an additive with the above heteroatoms. Examples of solid material are char resulting from the decomposition of paper or thermoplastics, such as ABS or PU, or foreign material, such as glass and metal. These solids are conveniently removed by filtration of the melted plastic. Any filtration apparatus may be used, such as plan filters, cartridge filters, etc. Magnetic separation can also be used.
[0068] Subsequently, the pretreated waste plastic is subjected to catalytic cracking.
[0069] In the cracking reactor, the pretreated waste plastic is contacted with a hot catalyst in a reaction chamber in order to pyrolise the plastic material. The hot catalyst provides at least part of the energy required to bring the plastic material to the reaction temperature, to supply the heat required for the endothermic cracking reaction and to bring the reaction products in their state after reaction. Preferably, the hot catalyst provides at least 60%, more preferably at least 90% of the heat required. Adiabatic operation is particularly preferred. Optionally, a heat exchanger can be introduced in the reaction chamber in order to remove any excess heat. Preferably, no more than 10% of the heat is removed. Preferably, the heat is removed by overheating low pressure steam. Low pressure steam is a steam pressure between 1 bar absolute and 10 bar absolute, preferably between 1.5 bar absolute and 4 bar absolute, more preferably between 2 bar absolute and 3 bar absolute.
[0070] The pressure in the reaction chamber is usually between 50 kPa absolute and 1500 kPa absolute, preferably between 80 kPa absolute and 1000 kPa absolute, more preferably between 100 kPa absolute and 500 kPa absolute. A pressure above atmospheric pressure is most preferred.
[0071] The hot catalyst is introduced into the reaction chamber in the form of heated particles or a mixture of heated particles which may comprise inert particles. These particles including inert particles are designated as "hot solid". Usually, the weight amount of hot solid is between 0.2 and 20 times the weight amount of plastic material, preferably between 0.5 and 10 times, more preferably between 1 and 12 times. Particularly preferred the amount of hot solid is between 3 and 9 times the amount of plastic material in the reaction chamber.
[0072] The residence time of the solid in the chamber may be between 0.1 and 6000 seconds, preferably between 1 and 3600 seconds, more preferably between 3 and 1800 seconds.
[0073] In one embodiment the temperature of the hot solid introduced in the reaction chamber is higher than the temperature in the reaction chamber. Usually, the temperature of the hot solid when introduced into the reaction chamber is between 100 and 500.degree. C. above the temperature in the reaction chamber, preferably between 150 and 400.degree. C. The temperature of the plastic material introduced into the reaction chamber is lower than the temperature in the reaction chamber. Usually, the temperature of the plastic material introduced into the reaction chamber is between 100 and 350.degree. C. lower than the temperature in the reaction chamber, preferably between 150 to 300.degree. C. lower.
[0074] The reaction chamber ensures the contact between the plastic feed and the hot solid and allows extracting a gaseous stream and a condensed stream. "Condensed stream" is to be understood as solid or liquid. Preferably, the condensed stream is a mixture of solid and liquid.
[0075] The reaction chamber can be any type known by the skilled person. Preferably, the reaction chamber has a continuous gas phase. The reaction chamber may be constituted of one or several zones having specific flow. Examples of reaction chambers and reaction zones are fluidized bed, bubbling, bed, spouted bed, entrained bed, etc. Fluidized bed and entrained bed are preferred. Fluidized bed is particularly preferred.
[0076] A fluidized bed can be operated using a gas flow from the bottom to the top in a riser or from the top to the bottom in a downer, a downer being preferred.
[0077] The reaction chamber can also include a condensed phase-gas separation zone. Examples of condensed phase-gas separation zones are decantation zone, sedimentation zone, elutriation zone, filtration zone and cyclone. Preferably, the reaction chamber is made of at least two combined zones, more preferably, at least three combined zones, even more preferable at least four combined zones. One of these zones should be the reaction zone.
[0078] In a preferred embodiment, the reaction chamber is constituted of a downer, a decantation zone, a sedimentation zone and a cyclone zone.
[0079] Optionally, an auxiliary gas may be introduced into the reaction chamber. The auxiliary gas may be introduced in any zone, in particular in the reaction zone. Examples of auxiliary gases are steam, inert gas and recycled gases. Recycled gases are preferred. More preferably, recycled gases are mainly constituted of hydrocarbon gases having less than 6 carbon atoms, hydrogen, nitrogen, carbon oxide, steam, oxygen and/or noble gas. Preferably, recycled gas contains mainly hydrocarbon gases having less than 6 carbon atoms, hydrogen and nitrogen. Also preferred, the recycled gas contains less than 5 vol. % of oxygen, more preferably less than 2 vol. %. Recycling gases obtained after condensation of the gas stream extracted from the reaction chamber are particularly preferred. Optionally, a gas stream exiting from a regeneration chamber may be used as auxiliary gas introduced into the reaction chamber. Optionally, the auxiliary gas may be preheated. Preferably, the auxiliary gas is heated up to the temperature at the bottom of the reactor. Preferably, the preheating is conducted using the gases from the regenerator. Preferably, the gas leaving the regenerator. In a preferred embodiment, a gas-gas heater using the gas leaving the regenerator to preheat the auxiliary gas is used. Preferably, the auxiliary gas is introduced at the bottom of the decantation zone so as to flash the condensed phase from the residual gas.
[0080] A gaseous stream is extracted from the reaction chamber by any means known in the art. Preferably, the gaseous stream is extracted in a condensed phase-gas separation zone of the reaction chamber, preferably from a cyclone zone.
[0081] The gaseous flow leaving the reaction chamber is directed to a condenser where the heavier hydrocarbons are condensed. The condensation can be induced by any means, for instance indirect cooling in a heat exchanger, aerocondenser, or by direct contact with a quench. Direct contact is preferred. Condensation can be made in one or in several steps in series. Condensation in one or two steps in series is preferred. Condensation made by direct contact of the gaseous stream with a subcooled liquid is preferred. Particularly in two steps. The first condensation step can be conducted at a temperature sufficient to avoid the solidification of the condensed stream. As the molecular weight of the hydrocarbons produced by the cracking is dispersed, the use of a direct contact condensation with the circulation of a suitable hydrocarbon cut is preferred. Suitable hydrocarbon cuts are kerosene, diesel, mix of kerosene and diesel and the like. The contact can be conducted by any means known in the art. Examples of quench means are quench tee, venturi, vessel and column. Quench tee, vessel and venturi are preferred. A combination of quench tee and vessel is particularly preferred.
[0082] The liquid vapor mixture obtained in the quench may be separated by any means known in the art. Such means are gravity liquid vapor separator, cyclone, demister, filter, etc. Gravity separator and cyclone being preferred. A combination of a gravity separator and a cyclone is particularly preferred. Optionally, a fractionating column where final cooling and condensation of liquid pyrolysis products take place, may be employed.
[0083] The uncondensed gas may be used as a fluidizing or transporting gas or may be burnt in a combustor.
[0084] Optionally, a stripping of the liquid solid mixture extracted at the bottom of the reaction chamber is conducted in a fluidized bed or in an entrained bed by contacting the product with a suitable gas stream, preferably using an entrained bed. Suitable gas streams are overheated steam, inert gas, recycled gases from the production, recycled gases from the regeneration, etc. Overheated steam is preferred.
[0085] The stripped hydrocarbon and the gas are separated in a cyclone from the entrained particles and they are supplied through a transfer line to a quench where the waxes are condensed and separated. The stripped solid enters in a transporting line and it is transported to the regenerator, where coke and unconverted plastic material are burnt, for example in a fluidized bed.
[0086] In a preferred embodiment the solid comprising the catalyst is circulated between the cracking reactor and the regenerator. Most preferably, the temperature and flow of the circulating catalyst are adjusted to obtain a pre-selected temperature in the cracking reactor.
[0087] The temperature in the regenerator usually is from 600 to 1000.degree. C., preferably from 650 to 800.degree. C. The pressure in the regenerator may slightly exceed the pressure in the reaction chamber. Flue gas is separated from particles entrained from the fluidized bed in a cyclone.
[0088] The catalyst recovered as the regenerator may include unburnt material. Unburnt material includes the mineral impurities introduced with the plastic material. It also includes the products of the reaction of those impurities with the impurities produced by the cracking reaction or in the regeneration reaction. Examples of impurities produced by the cracking reaction are HCl, HBr, HF, SO.sub.2, H.sub.2S, CO.sub.2, etc. Preferably, the mineral impurities are in the form of condensed matter, such as liquid or solid. More preferably, they are solid of low dimension. By low dimension one means less than 50 micron, preferably less than 20 micron. Fine particles obtained by abrasion of the catalyst are included in those mineral impurities. The gases leaving the regenerator are sent to a device allowing the separation of the condensed phase matter. Examples of such a device are cyclone, filter, electrostatic precipitator, quench vessel, etc. A cyclone is preferred. The condensed phase separated includes the ashes introduced with the plastic material, the reaction products formed in the reactor or in the regenerator, and the fine from the catalyst.
[0089] A more complete appreciation of the invention and many of the attendant advantages thereof will be readily understood by reference to the following detailed description when considered in connection with the accompanying drawing shown in FIG. 6.
[0090] According to FIG. 6, the mixed plastic feed 1 is introduced in the pretreatment 2 where the plastic pieces are shrunked, part of the foreign material 3 is removed by elutriation and optionally at least part of the free water 4 is removed. Air 5 is optionally used for these operations. The mixed product leaving the pretreatment 6 is introduced in the melting device 8. An auxiliary liquid 7 is added. The product is liquefied by heating to a predefined temperature. The gases produced by the increase of temperature and/or by the decomposition of some components of the plastic and/or by the reaction of decomposition products are vented by 9. The air introduced with the mixed plastic feed is also vented. The foreign impurities not soluble are separated by decantation and optionally filtration giving low density 10 and high density 11 impurities. The liquefied product 12 is sent to the reaction chamber 14 with the hot catalyst 13 coming from the regenerator. An auxiliary gas 12a is introduced in the reaction chamber in order to purge the condensed matter flux 16 produced in the reaction chamber. The vapor flux 15 is sent to the condensation area not shown in the present figure. The condensed matter flux 16 is sent to the regenerator 19 where air 17 is injected. The regeneration increases the temperature of the catalyst 13 which is recycled to the reaction chamber. The gases produced by the reaction and the ashes 18 are extracted and sent to the effluent gas treatment not shown in the figure.
[0091] Should the disclosure of any patents, patent applications, and publications which are incorporated herein by reference conflict with the description of the present application to the extent that it may render a term unclear, the present description shall take precedence.
* * * * *
D00000

D00001

D00002

D00003

D00004

D00005

D00006

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.