3.4 Kb Mitochondrial Dna Deletion For Use In The Detection Of Cancer
Parr; Ryan L. ; et al.
U.S. patent application number 14/874155 was filed with the patent office on 2016-12-29 for 3.4 kb mitochondrial dna deletion for use in the detection of cancer. The applicant listed for this patent is Mitomics Inc.. Invention is credited to Jennifer Creed, Gabriel Dakubo, Andrea Maggrah, Ryan L. Parr, Brian Reguly, Kerry Robinson, Robert Thayer.
| Application Number | 20160376660 14/874155 |
| Document ID | / |
| Family ID | 70285335 |
| Filed Date | 2016-12-29 |






| United States Patent Application | 20160376660 |
| Kind Code | A1 |
| Parr; Ryan L. ; et al. | December 29, 2016 |
3.4 KB MITOCHONDRIAL DNA DELETION FOR USE IN THE DETECTION OF CANCER
Abstract
A method for detecting cancer in an individual comprising detecting a deletion in the nucleic acid sequence between residues 10743 and 12125 in mitochondrial DNA, obtaining a biological sample from the individual, extracting the mitochondrial DNA (mtDNA) from the sample, quantifying the amount of mtDNA in the sample having a deletion in the nucleic acid sequence between residues 10743 and 14125 of the mtDNA genome, and comparing the amount of mtDNA in the sample having the deletion to at least one known reference sample.
| Inventors: | Parr; Ryan L.; (Thunder Bay, CA) ; Thayer; Robert; (Thunder Bay, CA) ; Dakubo; Gabriel; (Thunder Bay, CA) ; Creed; Jennifer; (Broomfield, CO) ; Robinson; Kerry; (Thunder Bay, CA) ; Maggrah; Andrea; (Thunder Bay, CA) ; Reguly; Brian; (Thunder Bay, CA) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Family ID: | 70285335 | ||||||||||
| Appl. No.: | 14/874155 | ||||||||||
| Filed: | October 2, 2015 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 14507027 | Oct 6, 2014 | |||
| 14874155 | ||||
| 12748120 | Mar 26, 2010 | |||
| 14507027 | ||||
| 11975390 | Oct 18, 2007 | 8008008 | ||
| 12748120 | ||||
| PCT/CA2006/000652 | Apr 18, 2006 | |||
| 11975390 | ||||
| PCT/CA2007/001711 | Sep 26, 2007 | |||
| 12748120 | ||||
| 60672016 | Apr 18, 2005 | |||
| 60721522 | Sep 29, 2005 | |||
| 60789872 | Apr 7, 2006 | |||
| Current U.S. Class: | 506/9 |
| Current CPC Class: | C12Q 2600/112 20130101; C12Q 1/6886 20130101; G01N 33/57434 20130101; C12Q 2600/156 20130101; C12Q 2600/118 20130101; C12Q 1/686 20130101; G01N 33/57415 20130101 |
| International Class: | C12Q 1/68 20060101 C12Q001/68 |
Claims
1. A method of detecting a cancer in an individual, the method comprising: a) obtaining a biological sample from the individual; b) quantifying the amount of mtDNA in the sample having a deletion in the nucleic acid sequence spanning approximately residues 10744 and 14124 of the mtDNA genome, wherein the step of quantifying comprises contacting the sample with a pair of amplification primers and amplifying a target region of mtDNA that is indicative of the deletion, wherein a first primer of the pair of primers is adapted to bind to a region of the mtDNA comprising a splice joining opposite ends of the mtDNA sequence after removal of the deletion; c) comparing the amount of mtDNA in the sample having the deletion to the amount of the deletion in a reference sample of mtDNA wherein the reference sample is from a known non-cancerous tissue or body fluid or from a known cancerous tissue or body fluid; wherein if the reference sample is from a known non-cancerous tissue or body fluid, then an elevated level of the deletion in the biological sample compared to the non-cancerous reference sample is indicative of cancer and if the reference sample is from a known cancerous tissue or body fluid sample then an equivalent or elevated level of the deletion in the biological sample compared to the cancerous reference sample is indicative of cancer.
2. The method of claim 1 wherein the deletion has a nucleic acid sequence set forth in SEQ ID NO: 1.
3. (canceled)
4. The method of claim 1, wherein the first primer has the nucleic acid sequence set forth in SEQ ID NO: 2.
5. The method of claim 1 wherein the step of quantifying is conducted using real-time PCR.
6. The method of claim 1 wherein the cancer is prostate cancer.
7. The method of claim 1 wherein the cancer is breast cancer.
8. The method of claim 1 wherein the biological sample is a body tissue or body fluid.
9. The method of claim 8 wherein the biological sample is breast tissue, prostate tissue, prostate massage fluid, or urine.
10. (canceled)
11. A method of monitoring an individual for the development of a cancer comprising; a) obtaining a biological sample from the individual; b) quantifying the amount of mtDNA in the sample having a deletion in the nucleic acid sequence spanning approximately residues 10744 and 14124 of the mtDNA genome, wherein the step of quantifying comprises contacting the sample with a pair of amplification primers and amplifying a target region of mtDNA that is indicative of the deletion, wherein a first primer of the pair of primers is adapted to bind to a region of the mtDNA comprising a splice joining opposite ends of the mtDNA sequence after removal of the deletion; c) repeating steps a) to b) over a duration of time; and d) wherein an increasing level of the deletion over the duration of time is indicative of cancer.
12. The method of claim 11 wherein the deletion has a nucleic acid sequence set forth in SEQ ID NO: 1.
13. The method of claim 11 further comprising at least one step selected from the group consisting of: (a) comparing the amount of mtDNA in the sample having the deletion to the amount of the deletion in a reference sample of mtDNA from known non-cancerous tissue or body fluid; and (b) comparing the amount of mtDNA in the sample having the deletion to the amount of the deletion in a reference sample of mtDNA from known cancerous tissue or body fluid.
14. (canceled)
15. The method of claim 11 wherein the step of quantifying is conducted using real-time PCR.
16. The method of claim 11, wherein the first primer has the nucleic acid sequence set forth in SEQ ID NO: 2.
17. The method of claim 11 wherein the cancer is prostate cancer.
18. The method of claim 11 wherein the cancer is breast cancer.
19. The method of claim 11 wherein the biological sample is a body tissue or body fluid.
20. The method of claim 19 wherein the biological sample is breast tissue, prostate tissue, prostate massage fluid, or urine.
21-35. (canceled)
Description
CROSS REFERENCE TO RELATED APPLICATIONS
[0001] This application is a continuation of U.S. patent application Ser. No. 14/507,027, filed Oct. 6, 2014, which is a continuation of U.S. patent application Ser. No. 12/748,120, filed Mar. 26, 2010, which is a continuation-in-part of U.S. patent application Ser. No. 11/975,390, filed Oct. 18, 2007, which is a continuation of PCT/CA2006/000652, filed Apr. 18, 2006, which PCT application claims the benefit under 35 U.S.C. .sctn.119(e) of U.S. Provisional Patent Application No. 60/672,016, filed Apr. 18, 2005; 60/721,522, filed Sep. 29, 2005; and 60/789,872, filed Apr. 7, 2006, which applications are hereby incorporated by reference in their entireties. Additionally, this application is a continuation of U.S. patent application Ser. No. 14/507,027, filed Oct. 6, 2014, which is a continuation of U.S. patent application Ser. No. 12/748,120, filed Mar. 26, 2010, which is a continuation of PCT/CA2007/001711, filed Sep. 26, 2007, which applications are hereby incorporated by reference in their entireties.
REFERENCE TO SEQUENCE IDENTIFICATION LISTING
[0002] The present application includes a sequence identification listing in .txt format as follows: [0003] Filename: Sequence_Listing re PCT International Patent Appl. No. PCT_CA2007_001711.txt [0004] Size: 26.8 KB [0005] Date Created: Mar. 22, 2010 This sequence identification listing is hereby expressly incorporated by reference in its entirety in the present application.
FIELD OF THE INVENTION
[0006] This invention is related to the field of mitochondrial genomics. In particular it is related to a 3.4 kb deletion in the mitochondrial genome and its utility as an indicator of cancer.
BACKGROUND OF THE INVENTION
[0007] Mitochondrial DNA (MtDNA) as a Diagnostic Tool
[0008] MtDNA sequence dynamics are important diagnostic tools. Mutations in mtDNA are often preliminary indicators of developing disease, often associated with nuclear mutations, and act as biomarkers specifically related to: disease, such as but not limited to, tissue damage and cancer from smoking and exposure to second hand tobacco smoke (Lee et al., 1998; Wei, 1998); longevity, based on accumulation of mitochondrial genome mutations beginning around 20 years of age and increasing thereafter (von Wurmb, 1998); metastatic disease caused by mutation or exposure to carcinogens, mutagens, ultraviolet radiation (Birch-Machin, 2000); osteoarthritis; cardiovascular, Alzheimer, Parkinson disease (Shoffner et al., 1993; Sherratt et al., 1997; Zhang et al, 1998); age associated hearing loss (Seidman et al., 1997); optic nerve degeneration and cardiac dysrhythmia (Brown et al., 1997; Wallace et al., 1988); chronic progressive external exophthalmoplegia (Taniike et al., 1992); atherosclerosis (Bogliolo et al., 1999); papillary thyroid carcinomas and thyroid tumours (Yeh et al., 2000); as well as others (e.g. Naviaux, 1997; Chinnery and Turnbull, 1999).
[0009] Mutations at specific sites of the mitochondrial genome can be associated with certain diseases. For example, mutations at positions 4216, 4217 and 4917 are associated with Leber's Hereditary Optic Neuropathy (LHON) (Mitochondrial Research Society; Huoponen (2001); MitoMap). A mutation at 15452 was found in 5/5 patients to be associated with ubiquinol cytochrome c reductase (complex III) deficiency (Valnot et al. 1999).
[0010] Specifically, these mutations or alterations include point mutations (transitions, transversions), deletions (one base to thousands of bases), inversions, duplications, (one base to thousands of bases), recombinations and insertions (one base to thousands of bases). In addition, specific base pair alterations, deletions, or combinations thereof have been found to be associated with early onset of prostate, skin, and lung cancer, as well as aging (e.g. Polyak et al., 1998), premature aging, exposure to carcinogens (Lee et al., 1998), etc.
[0011] Prostate Cancer
[0012] Prostate cancer is a frequently diagnosed solid tumour that most likely originates in the prostate epithelium (Huang et al. 1999). In 1997, nearly 10 million American men were screened for prostate specific antigen (PSA), the presence of which suggests prostate cancer (Woodwell, 1999). Indeed, this indicates an even higher number of men screened by an initial digital rectal exam (DRE). In the same year, 31 million men had a DRE (Woodwell, 1999). Moreover, the annual number of newly diagnosed cases of prostate cancer in the United States is estimated at 179,000 (Landis et al., 1999). It is the second most commonly diagnosed cancer and second leading cause of cancer mortality in Canadian men. In 1997 prostate cancer accounted for 19,800 of newly diagnosed cancers in Canadian men (28%) (National Cancer Institute of Canada). It is estimated that 30% to 40% of all men over the age of forty-nine (49) have some cancerous prostate cells, yet only 20% to 25% of these men have a clinically significant form of prostate cancer (SpringNet--CE Connection, internet, www.springnet.com/ce/j803a.htm). Prostate cancer exhibits a wide variety of histological behaviour involving both endogenous and exogenous factors, i.e. socio-economic situations, diet, geography, hormonal imbalance, family history and genetic constitution (Konishi et al. 1997; Hayward et al. 1998). Although certain mtDNA alterations have been previously associated with prostate cancer, the need exists for further markers for the detection of prostate cancer.
[0013] 3.4 kb MtDNA Deletion and the Detection of Prostate Cancer.
[0014] In the applicant's pending PCT application bearing publication no. WO/06/111029 (the entire contents of which are incorporated herein by reference) a deletion of a 3379 bp segment of mtDNA was identified through full mitochondrial genome amplification of prostate tissue. The 3379 bp deletion (referred to as the 3.4 kb deletion) was determined to be located between nucleotides 10744-14124 of the mitochondrial genome. It was determined that the detection of this deletion could be used in the diagnosis of prostrate cancer when tissue samples are tested.
[0015] The 3.4 kb deletion removes all or part of the following genes from the mtDNA genome: (i) NADH dehydrogenase subunit 4L, (ii) NADH dehydrogenase subunit 4, (iii) NADH dehydrogenase subunit 5, (iv) tRNA histidine, (v) tRNA serine2, and (vi) tRNA leucine2.
[0016] Breast Cancer
[0017] Breast cancer is a cancer of the glandular breast tissue and is the fifth most common cause of cancer death. In 2005, breast cancer caused 502,000 deaths (7% of cancer deaths; almost 1% of all deaths) worldwide (World Health Organization Cancer Fact Sheet No. 297). Among women worldwide, breast cancer is the most common cancer and the most common cause of cancer death (World Health Organization Cancer Fact Sheet No. 297). Although certain mtDNA alterations have been previously associated with breast cancer, for example, in Parrella et al. (Cancer Research: 61, 2001), the need exists for further markers for the detection of breast cancer.
BRIEF SUMMARY OF THE INVENTION
[0018] In one embodiment, the present invention provides a method of detecting a cancer in an individual comprising;
[0019] a) obtaining a biological sample from the individual;
[0020] b) extracting mitochondrial DNA, mtDNA, from the sample;
[0021] c) quantifying the amount of mtDNA in the sample having a deletion in the nucleic acid sequence spanning approximately residues 10744 and 14124 of the mtDNA genome;
[0022] d) comparing the amount of mtDNA in the sample having the deletion to at least one known reference value.
[0023] In one embodiment, the present invention provides a method of detecting a cancer in an individual comprising;
[0024] a) obtaining a biological sample from the individual;
[0025] b) extracting mitochondrial DNA, mtDNA, from the sample;
[0026] c) quantifying the amount of mtDNA in the sample having a deletion in the nucleic acid sequence spanning approximately residues 10744 and 14124 of the mtDNA genome;
[0027] d) comparing the amount of mtDNA in the sample having the deletion to the amount of the deletion in a reference sample of mtDNA from known non-cancerous tissue or body fluid;
[0028] wherein an elevated amount of the deletion in the biological sample compared to the reference sample is indicative of cancer.
[0029] In one embodiment, the present invention provides a method of detecting a cancer in an individual comprising;
[0030] a) obtaining a biological sample from the individual;
[0031] b) extracting mitochondrial DNA, mtDNA, from the sample;
[0032] c) quantifying the amount of mtDNA in the sample having a deletion in the nucleic acid sequence spanning approximately residues 10744 and 14124 of the mtDNA genome;
[0033] d) comparing the amount of mtDNA in the sample having the deletion to the amount of the deletion in a reference sample of mtDNA from known cancerous tissue or body fluid;
[0034] wherein a similar level of the deletion in the biological sample compared to the reference sample is indicative of cancer.
[0035] In one embodiment, the present invention provides a method of monitoring an individual for the development of a cancer comprising;
[0036] a) obtaining a biological sample;
[0037] b) extracting mtDNA from the sample;
[0038] c) quantifying the amount of mtDNA in the sample having a deletion in the nucleic acid sequence spanning approximately residues 10744 and 14124 of the mtDNA genome;
[0039] d) repeating steps a) to c) over a duration of time;
[0040] e) wherein an increasing level of the deletion over the duration of time is indicative of cancer.
[0041] In one embodiment, the present invention provides a method of detecting a cancer in an individual comprising;
[0042] a) obtaining a biological sample from the individual;
[0043] b) extracting mitochondrial DNA, mtDNA, from the sample;
[0044] c) quantifying the amount of mtDNA in the sample having a sequence corresponding to the sequence identified in SEQ ID NO: 1;
[0045] d) comparing the amount of mtDNA in the sample corresponding to SEQ ID NO: 1 to at least one known reference value.
BRIEF DESCRIPTION OF THE SEVERAL VIEWS OF THE DRAWINGS
[0046] An embodiment of the invention will now be described by way of example only with reference to the appended drawings wherein:
[0047] FIG. 1 is a schematic diagram showing the design and sequence of a primer useful for the detection of the 3.4 kb deletion. The primer (SEQ ID NO: 2) binds to bases 10729-10743/14125-14139 of the mtDNA genome, wherein the portion of the primer that binds to bases 10729-10743 is depicted in gray in the upper left corner of the figure (nucleotides 1-15 of SEQ ID NO: 2) and the portion of the primer that binds to bases 14125-14139 is depicted in white in the lower right corner of the figure (nucleotides 16-30 of SEQ ID NO: 2);
[0048] FIG. 2 is a graph showing a comparison of cycle threshold between malignant and symptomatic benign participants in the 3.4 kb study;
[0049] FIG. 3 is a graph showing cycle threshold as related to Example 1;
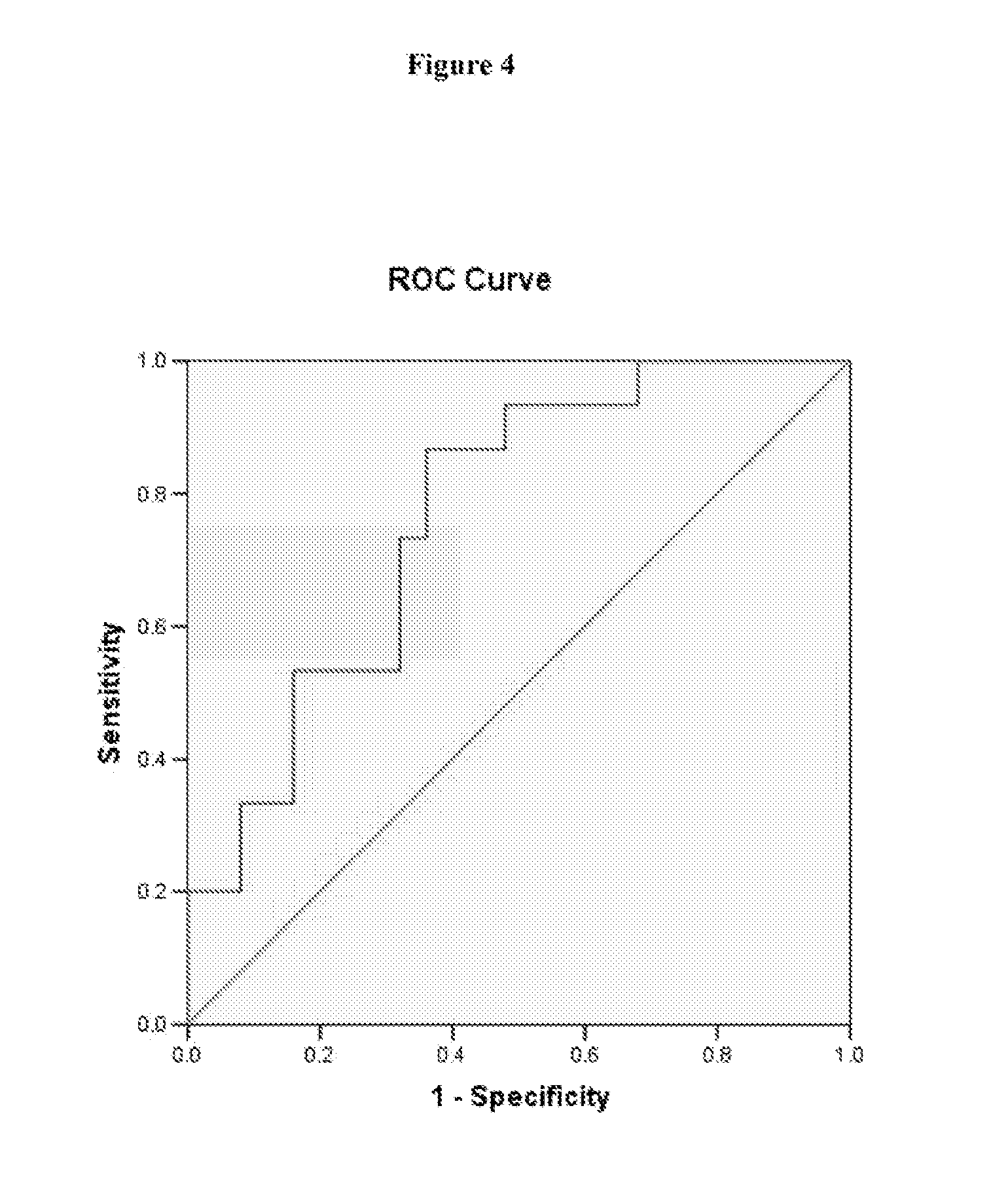
[0050] FIG. 4 shows a ROC curve illustrating the specificity and sensitivity of one embodiment of the present invention;
[0051] FIG. 5 shows a ROC curve illustrating the specificity and sensitivity of another embodiment of the present invention;
[0052] FIG. 6 shows real-time PCR data relating to 3.4 kb mtDNA deletion levels associated with breast cancer; and,
[0053] FIG. 7 shows a ROC curve illustrating the specificity and sensitivity of another embodiment of the present invention.
DETAILED DESCRIPTION OF THE INVENTION
[0054] As used herein, "cycle threshold" (C.sub.T) is the point at which target amplification using real-time PCR rises above background, as indicated by a signal such as a fluorescence signal. The C.sub.T is inversely related to the quantity of the sequence being investigated.
[0055] As defined herein, "sensitivity" refers to the fraction of true positives (true positive rate) results obtained using the method of the present invention.
[0056] As defined herein, "specificity" refers to the fraction of false positives (false positive rate) results obtained using the method of the present invention.
[0057] In one embodiment of the present invention, methods are provided for monitoring and diagnosing cancer through the detection and quantification of the aforementioned 3.4 kb mtDNA deletion. For example, the present invention may be used for detecting the presence of pre-neoplasia, neoplasia and progression towards potential malignancy of prostate cancer and breast cancer. In one aspect, the present invention involves the detection and quantification of the 3.4 kb mtDNA deletion (SEQ ID NO:1) for the detection, diagnosis, and/or monitoring of cancer. In this method, mtDNA is extracted from a biological sample (for example body tissue, or body fluids such as urine, prostate massage fluid). The extracted mtDNA is then tested in order to determine the levels (i.e. quantity) of the 3.4 kb deletion in the sample. In tests conducted by the present inventors, the levels of the deletion were found to be elevated in samples obtained from subjects with cancer when compared to samples obtained from subjects without cancer. Based on the information and data supplied below, the inventors have concluded that elevated levels of the 3.4 kb deletion in the mtDNA is indicative of cancer.
[0058] As disclosed in PCT WO/06/111029, the 3.4 kb deletion spans approximately nucleotides 10744 to 14124 of the mtDNA genome. The mtDNA genome is listed as SEQ ID NO:8 (Genbank accession no. AC_000021). The inventors have determined, as provided by example below, that this deletion is also associated with cancer and in particular prostate and breast cancer. Therefore, such deletion provides an accurate biomarker and, therefore, a valuable tool for the detection, diagnosis, or monitoring of cancer in at least these tissues.
[0059] The deletion results in the creation of two deletion monomers, one of 3.4 kb in size (small sublimon) and one of approximately 12.6 kb in size (large sublimon). The occurrence of the deletion may be detected by either identifying the presence of the small sublimon, or by determining that the 3.4 kb sequence has been deleted from the large sublimon.
[0060] As discussed above, the deletion is approximately 3379 bp, and comprises genes encoding NADH dehydrogenase subunit 4L, NADH dehydrogenase subunit 4, NADH dehydrogenase subunit 5, tRNA histidine, tRNAserine2, and tRNA leucine2.
[0061] In one embodiment, samples of, for example prostate tissue, prostate massage fluid, urine or breast tissue, are obtained from an individual and tested over a period of time (e.g. years) in order to monitor the genesis or progression of cancer. Increasing levels of the 3.4 kb deletion over time could be indicative of the beginning or progression of cancer.
[0062] Age related accumulation of the 3.4 kb mtDNA deletion may predispose an individual to, for example, prostate cancer or breast cancer, which is prevalent in middle aged and older men, and middle aged and older women, respectively. According to one aspect of the invention, a method is provided wherein regular cancer screening may take place by monitoring over time the amount of the 3.4 kb deletion in body tissues such as breast tissue or body fluids such as prostate massage fluid, or urine.
[0063] The system and method of the present invention may be used to detect cancer at an early stage, and before any histological abnormalities. For example, the system and method of the present invention may be used to detect pre-neoplasia in breast tissue.
[0064] The following primer sequences are preferred for the detection of the 3.4 kb deletion:
TABLE-US-00001 3.4 forward (binds to bases 10729-10743/ 14125-14139 of the mtDNA genome) (SEQ ID NO: 2) 5'-TAGACTACGTACATACTAACCCTACTCCTA-3'; 3.4 reverse (binds to bases 14361-14379 of the mtDNA genome) (SEQ ID NO: 3) 5'-GAGGTAGGATTGGTGCTGT-3'.
[0065] In one embodiment of the present invention, a pair of amplification primers are used to amplify a target region indicative of the presence of the 3.4 kb deletion. In this embodiment, one of the pair of amplification primers overlaps a spliced region of mtDNA after deletion of the 3.4 kb sequence has occurred (i.e. a splice at a position spanning approximately residues 10744 and 14124 of the mtDNA genome). Therefore, extension of the overlapping primer can only occur if the 3.4 kb section is deleted.
[0066] In another embodiment of the present invention, a pair of amplification primers are used to amplify a target region associated with the deleted 3.4 kb sequence. The deleted 3.4 kb sequence, upon deletion, may reform as a circular mtDNA molecule. In this embodiment, one of the pair of amplification primers overlaps the rejoining site of the ends of the 3.4 kb sequence. Thus, an increase in the amount of the 3.4 kb molecule detected in a sample is indicative of cancer. The below primer pair is preferred for the detection of the deleted 3.4 kb nucleic acid.
TABLE-US-00002 Forward 14115/10755 (SEQ ID NO: 9) 5'-CCCACTCATCACCTAAACCTAC-3' Reverse 10980R (SEQ ID NO: 10) 5'-GGTAGGAGTCAGGTAGTTAG-3'.
[0067] In one aspect of the invention, a kit for diagnosing cancer, for example prostate or breast cancer, comprising means for extraction of mtDNA, primers having the nucleic acid sequences recited in SEQ ID NOS: 2 and 3, or SEQ ID NOS: 9 and 10, reagents and instructions, is provided.
[0068] Another aspect of the invention provides methods for confirming or refuting the presence of a cancer biopsy test from a biopsy sample (e.g. prostate or breast cancer), comprising: obtaining non-cancerous tissue from a biopsy sample; and detecting and quantifying the amount of the 3.4 kb mtDNA deletion in the non-diseased tissue.
[0069] In one embodiment the present invention provides a method for screening individuals for prostate or breast cancer from a body fluid sample comprising; obtaining a body fluid sample, and detecting and quantifying the level of the 3.4 kb mtDNA deletion in the body fluid.
[0070] Although real-time quantitative PCR methods, as described in the examples below, represent the preferred means for detecting and quantifying the presence or absence of the 3.4 kb deletion, other methods that would be well known to an individual of skill in the art could also be utilized. For example quantification of the deletion could be made using Bio-Rad's Bioplex.TM. System and Suspension Array technology. Generally, the method requires amplification and quantification of sequences using any known methods.
[0071] The examples provided below illustrate that not only can this deletion be used for the detection of prostate cancer in prostate tissue, but can also be used to detect the presence of cancer in other biological samples, for example prostate massage fluid, urine, and breast tissue. Based on the findings in these examples, the 3.4 kb mtDNA deletion may be used as a biomarker for cancer.
[0072] The various examples provided illustrate a difference in the amount of mtDNA having the 3.4 kb deletion between samples obtained from subjects having cancer, and subjects without cancer. The amount of the 3.4 kb deletion was found to be higher in the samples obtained from subjects having cancer. This determination was made by comparing the amount of the 3.4 kb deletion in the test samples with amounts from known cancer cells and/or known non-cancer cells.
EXAMPLE 1
3.4 kb Deletion in the MtDNA of Prostate Tissue
[0073] A deletion of approximately 3.4 kilobases (kb) was identified through full mitochondrial genome amplification of fresh frozen prostate tissue. Using linear regression, the size of the deletion was estimated to be between 3000 base pairs (bp) and 3500 bp. Two possible candidate deletions were identified using Mitomap.TM. (Brandon, M. C., Lott, M. T., Nguyen, K. C., Spolim, S., Navathe, S. B., Baldi, P. & Wallace, D. C., MITOMAP: a human mitochondrial genome database--2004 update. Nucleic Acids Research 33 (Database Issue): D611-613, 2005; www.mitomap.org), the 3397 bp deletion at 9574-12972, and the 3379 bp deletion at 10744-14124. In order to determine which of the two deletions was associated with prostate cancer, if either, a forward primer which bridged the deletion junction was developed for each of the two candidates, ensuring that the primer extended further than the repeat regions that flank the deletions. FIG. 1 is a schematic diagram showing the design and sequence of the primer (i.e. SEQ ID NO: 2). Positive amplification results for the amplicon corresponding to the 3379 bp deletion (referred to as the 3.4 kb deletion) at 10744-14124 were obtained.
[0074] As indicated above, the 3.4 kb deletion removes all or part of the following genes: (i) NADH dehydrogenase subunit 4L, (ii) NADH dehydrogenase subunit 4, (iii) NADH dehydrogenase subunit 5, (iv) tRNA histidine, (v) tRNA serine2, and (vi) tRNA leucine2.
[0075] The 3.4 kb deletion was determined to be present in 91% of 33 fresh frozen prostate samples. With the specific deletion primers, formalin fixed tissues were tested in order increase the n value.
[0076] The present investigators sequenced entire mitochondrial genomes from 32 tissue samples microdissected by laser capture microdisection and 12 needle biopsies from histologically normal prostates. Archived tissue sections from each of these samples were used for the following study. 1-2 serial sections were removed from each sample. DNA was extracted from each sample in its entirety rather than as a microdissection. Thus, each sample consisted of a mixture of glandular prostate tissue as well as stromal prostate tissue. This extraction was performed using Qiagen's QIAamp.TM. DNA Mini Kit (Cat #51304). Following extraction the samples were quantified using a Nano-Drop.TM. spectrophotometer and the concentrations were subsequently normalized to 2 ng/ul. Each sample was amplified using 20 ng input DNA and an iQ.TM. SYBR Green Supermix.TM. kit (Bio-Rad Laboratories Inc.) Reactions were run on an Opticon.RTM. 2 two colour real-time PCR system (MJ Research).
[0077] As shown in FIG. 2, a distinct difference was observed in cycle threshold and, by extension, quantity of the deletion between the malignant prostate samples and the symptomatic benign prostate samples. Malignant samples exhibited a consistently earlier cycle threshold than the benign samples.
EXAMPLE 2
3.4 kb Deletion Blinded Study--Comparison of Cycle Threshold
[0078] An additional 21 prostate tissue samples were selected, 10 of which were benign and 11 of which were malignant. The pathological status was determined by needle biopsies conducted by a qualified pathologist. The samples were blinded such that the present investigators were unaware of their pathological status when they conducted this test. The present investigators were able to predict pathological status correctly in 81% of the cases by examining the cycle threshold. Of the 4 incorrect calls, two were malignant samples that were determined to be benign and 2 were benign samples that were determined to be malignant. Follow-up clinical information for the 2 individuals in the latter scenario was requested from the physician to determine if they had been diagnosed with prostate cancer subsequent to the needle biopsy results used for this study. One of the individuals who originally produced a benign sample but was predicted by this study to have a malignancy subsequently produced a malignant sample. As a result, one of the false positives became a true positive. Therefore, pathological status was predicted correctly in 86% of the cases examined in this study. The ultimate positive predictive value (PPV, where PPV=true positives/(true positives+false positives)) for this study was 91% and the negative predictive value (NPV, where NPV=true negatives/(true negatives+false negatives)) was 80%.
EXAMPLE 3
3.4 kb Deletion Study--Methods (n=76)
[0079] Seventy-six prostate tissue samples were examined for the 3.4 kb deletion in this study. All tissue samples were formalin-fixed, 25 being malignant, 12 being normal, and 39 having benign prostatic disease as shown histologically. Of the latter group more then half had hyperplasia. All specimens were needle biopsies taken from the investigators' tissue archives.
[0080] Prostate Specimens
[0081] A tapelift was performed on each slide using Prep-Strips (Catalogue Number LCM0207) from Arcturus Bioscience Inc. This allowed the removal of any particulate matter or non-adhering tissue from the slide prior to DNA extraction. With the tissue still on the slides, the slides were rinsed with PBS (Phosphate Buffered Saline Solution) to remove as much fixative as possible. The 1-2 needle biopsy sections on the slides were scraped into sterile microcentrifuge tubes using individually wrapped, sterilized surgical razor blades. DNA was then isolated and purified using a QIAamp.RTM. DNA Mini Kit (Qiagen, Cat. #51304) according to manufacturer's specifications. A negative extract control was processed in parallel with the slide extractions as a quality control checkpoint. The total concentration of DNA and purity ratio for each sample was determined by spectrophotometry (Nano-Drop.TM. ND-1000) and dilutions of 2 ng/.mu.l were prepared for the purpose of Quantitative Polymerase Chain Reaction (qPCR).
[0082] Primers (Oligonucleotides)
[0083] Purified oligonucleotide primers were chemically synthesized by Invitrogen (California, USA). The sequences of the primers and the expected sizes of the PCR products amplified are listed in Table 1. In addition, PCR analysis for mtDNA deletions included positive controls (DNA from a source known to carry the mutant mtDNA). Each primer set with the exception of TNF (tumor necrosis factor) were checked against a mitochondria-free rho 0 cell line to confirm the absence of pseudogene coamplification.
TABLE-US-00003 TABLE 1 Amplification Primers. Length of Position amplified Amplified product Primer Pair 5'-3' (base pairs) 3.4 Deletion 10729-14379 273 Real-Time (less 3379 bp at 10744-14124) 12 s mtDNA 708-945 238 TNF 3756-3886 131 3.4 forward (10729-10743-14125-14139) SEQ ID NO: 2 5'TAGACTACGTACATACTAACCCTACTCCTA-3' 3.4 reverse (14361-14379) SEQ ID NO: 3 5'-GAGGTAGGATTGGTGCTGT-3' 12 s forward (708-728) SEQ ID NO: 4 5'-CGTTCCAGTGAGTTCACCCTC-3'' 12 s reverse (923-945) SEQ ID NO: 5 5'-CACTCTTTACGCCGGCTTCTATT-3' TNF forward (3756-3775) SEQ ID NO: 6 5'-CCTGCCCCAATCCCTTTATT-3' TNF reverse (3866-3886) SEQ ID NO: 7 5'-GGTTTCGAAGTGGTGGTCTTG-3'
[0084] Real-Time Polymerase Chain Reaction
[0085] Three separate PCRs were performed on each sample. Each reaction was 25 .mu.l total volume and included template DNA, one pair of primers (12 s or 3.4 Deletion or TNF), an iQ.TM. SYBR Green Supermix.TM. kit (Catalogue Number 170-8882, Bio-Rad Laboratories Inc.) and distilled deionized water (ddH.sub.2O). The TNF (tumor necrosis factor) comprised single copy nuclear gene primers, and 12 s comprised total mitochondrial genome primers. The volume and concentrations for template DNA, primers, and reaction buffer are listed below.
TABLE-US-00004 TABLE 2 qPCR Components. Concentration per Volume per Reagent Reaction Reaction Reaction Buffer 1X 12.5 .mu.l Primer (forward 250 nM 0.0625 .mu.l of each and reverse) 100 umole stock ddH.sub.2O N/A 2.375. .mu.l Template DNA 20 ng 10.0 .mu.l Total 25 .mu.l
[0086] The cycling parameters for each amplicon are listed in Table 3.
TABLE-US-00005 TABLE 3 Cycling Parameters. Step Temperature (.degree. C.) Duration 1 95 3 min 2 95 30 sec 3 66 (3.4 deletion primers) or 30 sec 61.5 (12 s primers) or 61.5 (TNF primers) 4 72 30 sec 5 Plate Read 6 72 10 min 7 Melting Curve 50.degree. C.-110.degree. C. reading every 1.degree. C. 3 sec Repeat steps 2-5, 44 times for a total of 45 cycles.
[0087] Thermal cycling, real-time detection and analysis of the reactions was carried out using a DNA Engine Opticon.RTM. 2 Continuous Fluorescence Detection System equipped with Intuitive Opticon Monitor.TM. software (MJ Research Inc.). The standard curve method was utilized for DNA quantification. A set of serial dilutions (10.sup.6, 10.sup.5, 10.sup.4, 10.sup.3, 10.sup.2, 10.sup.1) of three purified PCR generated templates, one product for the 3.4 deletion, one for the 12 s primers, and one for TNF. From this, three different standard curves were generated showing the number of copies of total mtDNA (12 s amplicon-total mitochondrial genome primers), the amount of mtDNA having the 3.4 kb deletion, or total nuclear DNA (TNF-single copy nuclear gene primers). The C.sub.T values of the samples were then converted to the number of DNA copies by comparing the sample C.sub.T to that of the standards. The 3.4 deletion was considered to be absent or at low levels if the deletion was not detected within 37 cycles.
[0088] The determination of malignancy is based upon the quantity of the 3.4 kb deletion present in the normalized sample as indicated by the location of the cycle threshold. This location may be either absolute, as in greater than 25 cycles but less than 35 cycles, or more likely a ratio between the total mitochondrial DNA present as indicated by the 12 s amplicon, and the 3.4 kb deletion. This may be expressed as a per cent of the total mitochondrial DNA. The number of cells, as represented by the TNF amplicon, may be incorporated to refine the distinction between benign and malignant tissues.
[0089] In order to automate the analyses of these samples, bioinformatics tools were employed. The three variables that were considered for these analyses were the cycle threshold C.sub.T of Tumour Necrosis Factor (TNF), total pieces of mitochondria that contain those specific primer sites, and those mitochondria that harbour the deletion of interest.
[0090] Cluster Analysis
[0091] The clustering was not normalized nor were logarithmic functions used due to the similar and small range of data.
[0092] FIG. 3 shows the actual movement and trends of the data. The x-axis is the patient number and the y-axis is the cycle threshold obtained from real time PCR.
[0093] It is important to note that the higher the cycle threshold is, the lower amount of the deletion is present.
[0094] The general trend shown in FIG. 3 is based upon the differences/ratios between the variables of Deletion, Total, and TNF. The deletion is low to absent for the benign/normal samples (right side) and increases (toward the left) with abnormal benign and malignant samples. The abnormal benign and malignant samples begin to differentiate themselves from each other based on the cycle threshold ratio of Deletion to TNF.
[0095] Supervised Learning
[0096] Supervised learning is based on the system trying to predict outcomes for known samples. Half of the data was used to train and the other half to test the algorithm. Supervised learning compares its predictions to the target answer and "learns" from its mistakes. But, if the predicted output is higher or lower than the actual outcome in the data, the error is propagated back through the system and the weights are adjusted accordingly.
[0097] Data SET: 5% to 35%--Benign [0098] 35% to 65%--Hyperplasia [0099] 65% to 95%--Malignant
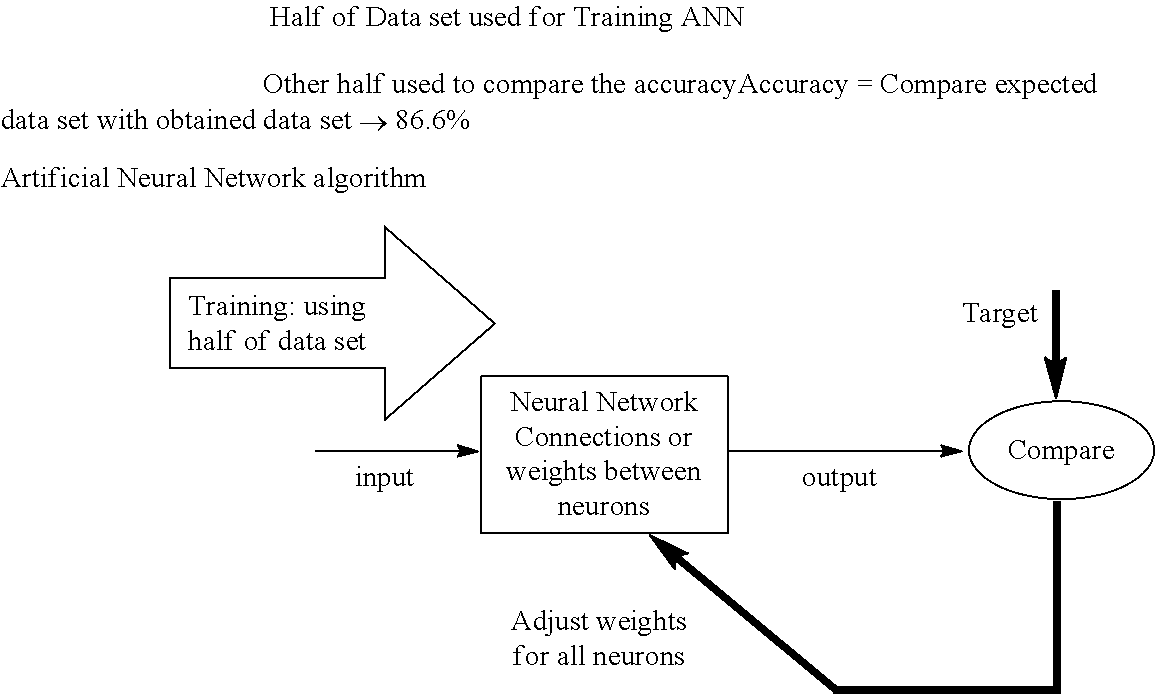
[0100] Artificial Neural Network (ANN) Algorithm (shown schematically below):
##STR00001##
[0101] Supervised Learning of Deletion Data using Artificial Neural Network (ANN)
[0102] Three Classifications: [0103] Benign [0104] Hyperplasia [0105] Malignant
[0106] Three variables for each classification were used based on Real Time PCR Cycle Threshold C.sub.T:
[0107] Tumour Necrosis Factor (TNF)--Nuclear copy control.
[0108] Total Mitochondria--Mitochondria copy control
[0109] Deletion--Mitochondria in the deleted state.
[0110] Results:
[0111] Half of data set is used to train the ANN, and the remaining half is used to compare the accuracy.
[0112] Three Classification Accuracy=86.6%
[0113] Positive Predictive Value (PPV);
[0114] Benign to Malignant=88.2%
[0115] Negative Predictive Value (NPV)
[0116] Benign to Malignant=76.5%
EXAMPLE 4
3.4 kb Deletion in MtDNA Associated with Breast Cancer
[0117] 18 samples were tested from malignant and benign breast tissue, 9 being malignant and 9 being benign, for the presence of the aforementioned 3.4 kb deletion. Samples were classified as either malignant or benign using conventional histopathological analysis.
[0118] DNA was isolated and purified from the samples using a QIAamp.RTM. DNA Mini Kit (Qiagen, Cat. #51304) according to manufacturer's specifications.
[0119] Purified oligonucleotide primers were chemically synthesized by Invitrogen (California, USA). The sequences of the primers and the expected sizes of the PCR products amplified are listed in Table 1 above.
[0120] Real-Time Polymerase Chain Reaction
[0121] Three separate PCRs were performed on each sample. Each reaction was 25 .mu.l total volume and included template DNA, one pair of primers (12 s or 3.4 Deletion or TNF), an iQ.TM. SYBR Green Supermix kit (Catalogue Number 170-8882, Bio-Rad Laboratories Inc.) and distilled deionized water (ddH.sub.2O). The TNF (tumor necrosis factor) comprised single copy nuclear gene primers, and 12 s comprised total mitochondrial genome primers. The volume and concentrations for template DNA, primers, and reaction buffer are listed below:
TABLE-US-00006 TABLE 4 qPCR Components. Concentration per Volume per Reagent Reaction Reaction Reaction Buffer 1X 12.5 .mu.l Primer (forward 250 nM 0.0625 .mu.l of each and reverse) 100 .mu.mole stock ddH.sub.2O N/A 2.375. .mu.l Template DNA 20 ng 10.0 .mu.l Total 25 .mu.l
[0122] The cycling parameters for each amplicon are listed in Table 5.
TABLE-US-00007 TABLE 5 Cycling Parameters. Step Temperature (.degree. C.) Duration 1 95 3 min 2 95 30 sec 3 66 (3.4 deletion primers) or 30 sec 61.5 (12 s primers) or 61.5 (TNF primers) 4 72 30 sec 5 Plate Read 6 72 10 min 7 Melting Curve 50.degree. C.-110.degree. C. reading every 1.degree. C. 3 sec Repeat steps 2-5, 44 times for a total of 45 cycles.
[0123] Thermal cycling, real-time detection and analysis of the reactions was carried out using a DNA Engine Opticon.RTM. 2 Continuous Fluorescence Detection System equipped with Intuitive Opticon Monitor.TM. software (MJ Research Inc.). The standard curve method was utilized for DNA quantification. A set of serial dilutions (10.sup.6, 10.sup.5, 10.sup.4, 10.sup.3, 10.sup.2, 10.sup.1) of three purified PCR generated templates were performed, one product for the 3.4 deletion, one for the 12 s primers, and one for TNF. From this, three different standard curves were generated showing the number of copies of total mtDNA (12 s amplicon-total mitochondrial genome primers), 3.4 deletion or total nuclear DNA (TNF-single copy nuclear gene primers). The C.sub.T values of the samples were then converted to the number of DNA copies by comparing the sample C.sub.T to that of the standards.
[0124] The determination of malignancy was based upon the quantity of the 3.4 kb deletion present in the normalized sample as indicated by the location of the cycle threshold. This location may be either absolute, as in greater than 25 cycles but less than 30 cycles, or more likely a ratio between the total mitochondrial DNA present as indicated by the 12 s amplicon, and the 3.4 kb deletion. This may be expressed as a percent of the total mitochondrial DNA.
[0125] In order to automate the analyses of these samples, bioinformatics tools were employed. The three variables that were considered for these analyses were the cycle threshold C.sub.T of Tumour Necrosis Factor (TNF), total species of mitochondria that contain those specific primer sites, and those mitochondria that harbour the deletion of interest.
[0126] Table 6 and FIG. 7 show the difference in the mean C.sub.T scores for samples from malignant tissue and benign tissue. The mean C.sub.T value for normal tissue was 30.5889, while the mean C.sub.T for malignant tissue was 27.8533 thereby illustrating a difference in the quantity of mtDNA having the 3.4 kb deletion in malignant breast tissue compared to normal breast tissue.
TABLE-US-00008 TABLE 6 Mean values for C.sub.T scores Group Statistics Std. Error GRP N Mean Std. Deviation Mean del3.4 normal 9 30.5889 2.53897 .84632 malignant 9 27.8533 2.52253 .84084
[0127] FIG. 8 is an ROC curve illustrating the specificity and sensitivity of the 3.4 kb mtDNA deletion as a marker for breast cancer when testing breast tissue. These results were obtained using a cutoff C.sub.T of 29.1900. The sensitivity of the marker at this C.sub.T was 77.8%, while the specificity was 77.8%.
[0128] Table 7 shows the calculation of the area under the curve for the present example. As a measure of the accuracy of the test.
TABLE-US-00009 TABLE 7 Results Showing Area Under the Curve Area Under the Curve Test Result Variable(s): del3.4 Asymptotic 95% Confidence Asymptotic Interval Area Std. Error.sup.a Sig..sup.b Lower Bound Upper Bound .790 .112 .038 .570 1.010 .sup.aUnder the nonparametric assumption .sup.bNull hypothesis: true area = 0.5
[0129] The determination of the cutoff C.sub.T of 29.1900 is shown in table 8 below. The results listed in table 8 show that a cutoff C.sub.T of 29.1900 provided the highest sensitivity and specificity at 78% and 78% respectively.
TABLE-US-00010 TABLE 8 Determination of C.sub.Tcutoff. Coordinates of the Curve Test Result Variable(s): del3.4 Positive if Less Than or Equal To.sup.a Sensitivity 1 - Specificity 24.6000 .000 .000 25.6800 .111 .000 25.7700 .222 .000 25.9250 .333 .000 26.2050 .444 .000 26.8400 .556 .000 27.4800 .556 .111 28.1600 .556 .222 28.8800 .667 .222 29.1900 .778 .222 29.4600 .778 .333 29.8750 .778 .444 30.5850 .778 .556 31.2200 .778 .667 31.5000 .889 .667 31.7650 .889 .778 32.9900 1.000 .778 34.3350 1.000 .889 35.6400 1.000 1.000 .sup.aThe smallest cutoff value is the minimum observed test value minus 1, and the largest cutoff value is the maximum observed test value plus 1. All the other cutoff values are the averages of two consecutive ordered observed test values.
EXAMPLE 5
The 3.4 kb Deletion in the Prostate Massage Fluid of Individuals with Prostate Cancer as Compared to the Fluid from those without Histological Evidence of Prostate Cancer
[0130] Forty prostate massage fluid samples were collected by urologists from patients who were either subsequently diagnosed with prostate cancer or showed no histological evidence of prostate cancer following a prostate needle biopsy procedure. The sample was deposited on a IsoCode Card.TM. (Schleicher & Shuell), dried, and then extracted according to the manufacturer's protocol. All DNA extracts were quantified using a NanoDrop.TM. ND-1000 Spectrophotometer and the DNA concentration normalized to 2 ng/ul. Each sample was then amplified according to the following parameters:
TABLE-US-00011 1X iQ SYBR Green Supermix .TM. (Bio-Rad P/N 170-8880) 150 nmol forward primer (SEQ ID NO: 2) (5'-TAGACTACGTACATACTAACCCTACTCCTA-3'). 150 nmol reverse primer (SEQ ID NO: 3) (5'-GAGGTAGGATTGGTGCTGT-3') 20 ng template DNA in a 25 ul reaction.
[0131] Reactions were cycled on an Opticon.TM. 2 DNA Engine (Bio-Rad Canada) according to the following protocol: [0132] 1. 95.degree. C. for 3 minutes [0133] 2. 95.degree. C. for 30 seconds [0134] 3. 66.degree. C. for 30 seconds [0135] 4. 72.degree. C. for 30 seconds [0136] 5. Plate Read [0137] 6. Repeat steps 2-5 44 times [0138] 7. 72.degree. C. for 10 minutes [0139] 8. Melting Curve from 50.degree. C. to 105.degree. C., read every 1.degree. C., hold for 3 seconds [0140] 9. 10.degree. C. Hold
TABLE-US-00012 [0140] TABLE 9 Results showing the mean C.sub.T Values for Prostate Massage Fluid Test Group Statistics Std. Error Group N Mean Std. Deviation Mean DEL3.4 benign 25 37.1869 3.18495 .63699 malignant 15 33.7712 3.98056 1.02778
[0141] Tables 9 and 10 show a significant difference between the mean C.sub.T values obtained for the benign sample and the malignant sample groups (p=0.005).
TABLE-US-00013 TABLE 10 Results Showing Difference (p = 0.005) for C.sub.T values of samples. Independent Samples Test Levene's t-test for Equality of Means Test for 95% Confidence Equality Interval of the of Variances Mean Std. Error Difference F Sig. t df Sig. (2-tailed) Difference Difference Lower Upper DEL3.4 Equal variances 1.251 .270 2.989 38 .005 3.41570 1.14283 1.10217 5.72923 assumed Equal variances 2.825 24.696 .009 3.41570 1.20917 .92382 5.90758 not assumed
[0142] FIG. 5 is a Receiver Operating Characteristic (ROC) curve illustrating the specificity and sensitivity of the 3.4 kb mtDNA deletion as a marker for prostate cancer when testing prostate massage fluid. These results were obtained using a cutoff C.sub.T of 37.3683. The sensitivity of the marker at this C.sub.T is 87%, while the specificity is 64%.
[0143] The accuracy of the test depends on how well the test separates the group being tested into those with and without the prostate cancer. Accuracy is measured by the area under the ROC curve. Table 11 shows the calculation of the area under the curve for the present example.
TABLE-US-00014 TABLE 11 Results Showing Area Under the ROC Curve Area Under the Curve Test Result Variable(s): DEL3.4 Asymptotic 95% Confidence Asymptotic Interval Area Std. Error.sup.a Sig..sup.b Lower Bound Upper Bound .768 .074 .005 .622 .914 .sup.aUnder the nonparametric assumption .sup.bNull hypothesis: true area = 0.5
TABLE-US-00015 TABLE 12 Determination of Specificity and Sensitivity Coordinates of the Curve Test Result Variable(s): DEL3.4 Positive if Less Than or Equal To.sup.a Sensitivity 1 - Specificity 26.2992 .000 .000 27.3786 .067 .000 28.2484 .133 .000 29.5193 .200 .000 30.1757 .200 .040 30.4580 .200 .080 30.5980 .267 .080 31.5709 .333 .080 32.5712 .333 .120 32.9500 .333 .160 33.3314 .400 .160 33.6547 .467 .160 33.9247 .533 .160 34.3554 .533 .200 34.9056 .533 .240 35.4650 .533 .280 35.9172 .533 .320 36.0648 .600 .320 36.3616 .667 .320 36.6421 .733 .320 36.8531 .733 .360 37.1188 .800 .360 37.3683 .867 .360 37.5200 .867 .400 37.8341 .867 .440 38.2533 .867 .480 38.5198 .933 .480 38.6519 .933 .520 38.8552 .933 .560 39.1258 .933 .600 39.2734 .933 .640 39.4952 .933 .680 39.7323 1.000 .680 39.6956 1.000 .720 41.0000 1.000 1.000
The smallest cutoff value is the minimum observed test value-1, and the largest cutoff value is the maximum observed test value plus 1. All the other cutoff values are the average of two consecutive ordered, observed test values.
[0144] The determination of the cutoff C.sub.T of 37.3683 is shown in table 12 above. The results listed in table 12 illustrate that a cutoff C.sub.T of 37.3683 provided the highest sensitivity and specificity.
EXAMPLE 6
The 3.4 kb Deletion in the Urine of Individuals with Prostate Cancer as Compared to the fluid from those without Histological Evidence of Prostate Cancer
[0145] Urine samples were collected from 5 patients who were diagnosed with prostate cancer and 5 who have had a needle biopsy procedure which was unable to detect prostate malignancy. These samples were collected following a digital rectal exam (DRE) to facilitate the collection of prostate cells.
[0146] Upon receipt of the samples a 5 ml aliquot was removed and then 2 mls were centrifuged at 14,000.times.g to form a pellet. The supernatant was removed and discarded. Pellets were resuspended in 200 ul phosphate buffered saline solution. Both the resuspended pellet and the whole urine sample were subjected to a DNA extraction procedure using the QiaAMP.TM. DNA Mini Kit (Qiagen P/N 51304) according to the manufacturer's directions. The resulting DNA extracts were then quantified using a NanoDrop.TM. ND-1000 Spectrophotometer and normalized to a concentration of 0.1 ng/ul.
[0147] Samples were analyzed by quantitative real-time PCR with the 3.4 kb deletion specific primers according to the following:
TABLE-US-00016 1X iQ SYBR Green Supermix .TM. (Bio-Rad P/N 170-8880) 100 nmol forward primer (SEQ ID NO: 2) (5'-TAGACTACGTACATACTAACCCTACTCCTA-3') 100 nmol reverse primer (SEQ ID NO: 3) (5'-GAGGTAGGATTGGTGCTGT-3') 1 ng template DNA in a 25 ul reaction.
[0148] Reactions were cycled on an Opticon.TM. 2 DNA Engine (Bio-Rad Canada) according to the following protocol:
[0149] 1. 95.degree. C. for 3 minutes
[0150] 2. 95.degree. C. for 30 seconds
[0151] 3. 69.degree. C. for 30 seconds
[0152] 4. 72.degree. C. for 30 seconds
[0153] 5. Plate Read
[0154] 6. Repeat steps 2-5 44 times
[0155] 7. 72.degree. C. for 10 minutes
[0156] 8. Melting Curve from 50.degree. C. to 105.degree. C., read every 1.degree. C., hold for 3 seconds
[0157] 9. 10.degree. C. Hold
TABLE-US-00017 TABLE 13 Mean values for C.sub.T scores Group Statistics Std. Error GRP fluid 38 N Mean Std. Deviation Mean CTf Benign 5 33.2780 1.10900 .49596 Malignant 5 30.6980 2.55767 1.14382
[0158] Tables 13 and 14 show a significant difference between the mean C.sub.T values obtained for benign sample and the malignant sample groups (p=0.005).
TABLE-US-00018 TABLE 14 Results Showing Difference (p = 0.005) for C.sub.T values of samples. Independent Samples Test Levene's t-test for Equality of Means Test for 95% Confidence Equality of Interval of the Variances Mean Std. Error Difference F Sig. t df Sig. (2-tailed) Difference Difference Lower Upper CTf Equal variances 1.272 .292 2.069 8 .072 2.58000 1.24672 -.29494 5.45494 assumed Equal variances 2.069 5.453 .089 2.58000 1.24672 -.54639 5.70639 not assumed
[0159] FIG. 6 is a Receiver Operating Characteristic (ROC) curve illustrating the specificity and sensitivity of the 3.4 kb mtDNA deletion as a marker for prostate cancer when testing urine. These results were obtained using a cutoff C.sub.T of 31.575. The sensitivity of the marker at this C.sub.T is 80%, while the specificity is 100%.
[0160] The determination of the cutoff C.sub.T of 31.575 is shown in table 15. The results listed in table 15 show that a cutoff C.sub.T of 31.575 provided the highest sensitivity and specificity.
TABLE-US-00019 TABLE 15 Determination of C.sub.Tcutoff. Coordinates of the Curve Test Result Variable(s): CTf Positive if Less Than or Equal To.sup.a Sensitivity 1 - Specificity 26.2900 .000 .000 28.4950 .200 .000 30.3850 .400 .000 31.0800 .600 .000 31.5750 .800 .000 32.1400 .800 .200 32.8150 .800 .400 33.8700 .800 .600 34.3350 .800 .800 34.3550 1.000 .800 35.3700 1.000 1.000 .sup.aThe smallest cutoff value is the minimum observed test value minus 1, and the largest cutoff value is the maximum observed test value plus 1. All the other cutoff values are the averages of two consecutive ordered observed test values.
EXAMPLE 7
Detection of Re-Circularized 3.4 kb Deleted Sequence in Prostate Malignant and Benign Tissue
[0161] In this example, the amount of re-circularized 3.4 kb deleted mtDNA molecules in samples was tested as an indicator for prostate cancer. As mentioned above, the 3.4 kb sequence, upon deletion, may reform as a circular mtDNA molecule. Amplification of a target region from the deleted 3.4 kb mtDNA sublimon was conducted using a primer pair (SEQ ID NOS: 9 and 10). The forward primer (SEQ ID NO: 9), overlaps the rejoining site of the ends of the 3.4 kb sequence.
[0162] Prostate tissue was formalin-fixed paraffin embedded prostate tissue needle biopsies.
[0163] The reagent setup used for this example was as follows:
[0164] 250 nmol each primer
[0165] 12.5 ul of 2.times. reaction mix,
[0166] 20 ng (10 ul of 2 ng/ul) template in 25 ul reaction volume.
[0167] The cycling parameters were as follows: [0168] 1. 95 degrees Celsius for 3 minutes [0169] 2. 95 degrees Celsius for 30 seconds [0170] 3. 62 degrees Celsius for 30 seconds [0171] 4. 72 degrees Celsius for 30 seconds [0172] 5. Plate Read [0173] 6. Repeat steps 2-5 44 times [0174] 7. 72 degrees for 10 minutes [0175] 8. Melting Curve from 50-100 degrees, reading every 1 degree for 3 seconds [0176] 9. 4 degrees HOLD.
[0177] Amplification of a target region from the deleted 3.4 kb mtDNA sublimon was conducted using a primer pair (SEQ ID NOS: 9 and 10).
[0178] Table 16 below provides a summary of testing conducted for the detection of the actual 3.4 kb deleted in mtDNA obtained from malignant and benign prostate tissue. Using a C.sub.T score of 30.0, a clear identification of malignant and benign tissue was possible. As such, an increase in the amount of the 3.4 kb molecule present in a sample was indicative of cancer.
TABLE-US-00020 TABLE 16 C.sub.T scores for Detection of Cancer in Prostate Tissue Description C.sub.T Benign sample 1 33.75 Malignant sample 1 28.79 Benign sample 2 30.96 Malignant sample 2 28.4 Benign sample 3 32.19 Malignant sample 3 27.38
[0179] Although the invention has been described with reference to certain specific embodiments, various modifications thereof will be apparent to those skilled in the art without departing from the spirit and scope of the invention as outlined in the claims appended hereto.
REFERENCES
[0180] Birch-Machin M A, Online Conference Report (Sunburnt DNA), International Congress of Biochemistry and Molecular Biology, New Scientist, 2000(a)
[0181] Birch-Machin M A, Taylor R W, Cochran B, Ackrell B A C, Tumbull D M. Ann Neurol 48: 330-335, 2000(b)
[0182] Birch-Machin, M. A. (2000). Mitochondria and skin disease. Clin Exp Dermatol, 25, 141-6.
[0183] Brown, M. D., et al., Am J. Humn Genet, 60: 381-387, 1997
[0184] Bogliolo, M, et al., Mutagenesis, 14: 77-82, 1999
[0185] Chinnery P F and Turnbull D M., Lancet 354 (supplement 1): 17-21, 1999
[0186] Huoponen, Kirsi, Leber hereditary optic neuropathy: clinical and molecular genetic findings, Neurogenetics (2001) 3: 119-125.
[0187] Hayward S W, Grossfeld G D, Tlsty T D, Cunha G R., Int J Oncol 13:35-47, 1998
[0188] Huang G M, Ng W L, Farkas J, He L, Liang H A, Gordon D, Hood R., Genomics 59(2):178-86, 1999
[0189] Konishi N, Cho M, Yamamoto K, Hiasa Y. Pathol. Int. 47:735-747, 1997
[0190] Landis S H, Murray T, Bolden S, Wingo P A. Cancer J. Clin. 49:8-31
[0191] Lee H C, Lu C Y, Fahn H J, Wei Y Hu. Federation of European Biochemical Societies, 441:292-296, 1998
[0192] Mitochondrial Research Society http://www.mitoresearch.org/diseases.html.
[0193] MITOMAP: A human mt genome database (www.gen.emory.edu/mitomap.html)
[0194] Naviaux, R K., Mitochondrial Disease--Primary Care Physician's Guide. Psy-Ed. Corp D/B/A Exceptional Parents Guide: 3-10, 1997
[0195] Parrella P, Xiao Y, Fliss M, Sanchez-Cespedes M, Mazzarelli P, Rinaldi M, Nicol T, Gabrielson E, Cuomo C, Cohen D, Pandit S, Spencer M, Rabitti C, Fazio V M, Sidransky D: Detection of mitochondrial DNA mutations in primary breast cancer and fine-needle aspirates. Cancer Res 2001, 61:7623-7626
[0196] Polyak Y, et al., Nature Genet. 20 (3):291-293, 1998
[0197] Seidman, M. D. et al., Arch. Otolaryngol Head Neck Surg., 123: 1039-1045, 1997
[0198] Sherrat E J, Thomas A W, Alcolado J C., Clin. Sci. 92:225-235, 1997
[0199] Shoffner J M, Brown M D, Torroni A, Lott M T, Cabell M F, Mirra S S, Beal M F, Yang C, Gearing M, Salvo R, Watts R L, Juncos J L, Hansen L A, Crain B J, Fayad M, Reckford C L, and Wallace D C., Genomics 17: 171-184, 1993
[0200] SpringNet--CE Connection: Screening, Diagnosis: Improving Primary Care Outcomes. Website: http://www.springnet.com/ce/j803a.htm
[0201] Taniike, M. et al., BioChem BioPhys Res Comun, 186: 47-53, 1992
[0202] Valnot, Isabelle, et al., A mitochondrial cytochrome b mutation but no mutations of nuclearly encoded subunits in ubiquinol cytochrome c reductase (complex III) deficiency, Human Genetics (1999) 104: 460-466
[0203] von Wurmb, N, Oehmichen, M, Meissner, C., Mutat Res. 422:247-254, 1998
[0204] Wallace et al., Mitochondrial DNA MUtatio Associated with Leber's Hereditary Optic Neuropathy, Science, 1427-1429
[0205] Wei Y H. Proceedings of the Nat. Sci. Council of the Republic of China April 22(2):5567, 1998
[0206] Woodwell D A. National Ambulatory Medical Care Survey: 1997 Summary. Advance data from vital and health statistics; no. 305. Hyattsville, Md.: National Center for Health Statistics. 1999
[0207] Yeh, J. J., et al., Oncogene Journal, 19: 2060-2066, 2000
[0208] Zhang et al., Multiple mitochondrial DNA deletions in an elderly human individual, FEBS Lett, 297, 34-38 1992
[0209] Zhang, C., et al., BioChem. BioPhys. Res. Comun., 195: 1104-1110, 1993
Sequence CWU 1
1
1013379DNAHomo sapiens 1cctaaaccta ctccaatgct aaaactaatc gtcccaacaa
ttatattact accactgaca 60tgactttcca aaaaacacat aatttgaatc aacacaacca
cccacagcct aattattagc 120atcatccctc tactattttt taaccaaatc
aacaacaacc tatttagctg ttccccaacc 180ttttcctccg accccctaac
aacccccctc ctaatactaa ctacctgact cctacccctc 240acaatcatgg
caagccaacg ccacttatcc agtgaaccac tatcacgaaa aaaactctac
300ctctctatac taatctccct acaaatctcc ttaattataa cattcacagc
cacagaacta 360atcatatttt atatcttctt cgaaaccaca cttatcccca
ccttggctat catcacccga 420tgaggcaacc agccagaacg cctgaacgca
ggcacatact tcctattcta caccctagta 480ggctcccttc ccctactcat
cgcactaatt tacactcaca acaccctagg ctcactaaac 540attctactac
tcactctcac tgcccaagaa ctatcaaact cctgagccaa caacttaata
600tgactagctt acacaatagc ttttatagta aagatacctc tttacggact
ccacttatga 660ctccctaaag cccatgtcga agcccccatc gctgggtcaa
tagtacttgc cgcagtactc 720ttaaaactag gcggctatgg tataatacgc
ctcacactca ttctcaaccc cctgacaaaa 780cacatagcct accccttcct
tgtactatcc ctatgaggca taattataac aagctccatc 840tgcctacgac
aaacagacct aaaatcgctc attgcatact cttcaatcag ccacatagcc
900ctcgtagtaa cagccattct catccaaacc ccctgaagct tcaccggcgc
agtcattctc 960ataatcgccc acgggcttac atcctcatta ctattctgcc
tagcaaactc aaactacgaa 1020cgcactcaca gtcgcatcat aatcctctct
caaggacttc aaactctact cccactaata 1080gctttttgat gacttctagc
aagcctcgct aacctcgcct taccccccac tattaaccta 1140ctgggagaac
tctctgtgct agtaaccacg ttctcctgat caaatatcac tctcctactt
1200acaggactca acatactagt cacagcccta tactccctct acatatttac
cacaacacaa 1260tggggctcac tcacccacca cattaacaac ataaaaccct
cattcacacg agaaaacacc 1320ctcatgttca tacacctatc ccccattctc
ctcctatccc tcaaccccga catcattacc 1380gggttttcct cttgtaaata
tagtttaacc aaaacatcag attgtgaatc tgacaacaga 1440ggcttacgac
cccttattta ccgagaaagc tcacaagaac tgctaactca tgcccccatg
1500tctaacaaca tggctttctc aacttttaaa ggataacagc tatccattgg
tcttaggccc 1560caaaaatttt ggtgcaactc caaataaaag taataaccat
gcacactact ataaccaccc 1620taaccctgac ttccctaatt ccccccatcc
ttaccaccct cgttaaccct aacaaaaaaa 1680actcataccc ccattatgta
aaatccattg tcgcatccac ctttattatc agtctcttcc 1740ccacaacaat
attcatgtgc ctagaccaag aagttattat ctcgaactga cactgagcca
1800caacccaaac aacccagctc tccctaagct tcaaactaga ctacttctcc
ataatattca 1860tccctgtagc attgttcgtt acatggtcca tcatagaatt
ctcactgtga tatataaact 1920cagacccaaa cattaatcag ttcttcaaat
atctactcat cttcctaatt accatactaa 1980tcttagttac cgctaacaac
ctattccaac tgttcatcgg ctgagagggc gtaggaatta 2040tatccttctt
gctcatcagt tgatgatacg cccgagcaga tgccaacaca gcagccattc
2100aagcaatcct atacaaccgt atcggcgata tcggtttcat cctcgcctta
gcatgattta 2160tcctacactc caactcatga gacccacaac aaatagccct
tctaaacgct aatccaagcc 2220tcaccccact actaggcctc ctcctagcag
cagcaggcaa atcagcccaa ttaggtctcc 2280acccctgact cccctcagcc
atagaaggcc ccaccccagt ctcagcccta ctccactcaa 2340gcactatagt
tgtagcagga atcttcttac tcatccgctt ccacccccta gcagaaaata
2400gcccactaat ccaaactcta acactatgct taggcgctat caccactctg
ttcgcagcag 2460tctgcgccct tacacaaaat gacatcaaaa aaatcgtagc
cttctccact tcaagtcaac 2520taggactcat aatagttaca atcggcatca
accaaccaca cctagcattc ctgcacatct 2580gtacccacgc cttcttcaaa
gccatactat ttatgtgctc cgggtccatc atccacaacc 2640ttaacaatga
acaagatatt cgaaaaatag gaggactact caaaaccata cctctcactt
2700caacctccct caccattggc agcctagcat tagcaggaat acctttcctc
acaggtttct 2760actccaaaga ccacatcatc gaaaccgcaa acatatcata
cacaaacgcc tgagccctat 2820ctattactct catcgctacc tccctgacaa
gcgcctatag cactcgaata attcttctca 2880ccctaacagg tcaacctcgc
ttccccaccc ttactaacat taacgaaaat aaccccaccc 2940tactaaaccc
cattaaacgc ctggcagccg gaagcctatt cgcaggattt ctcattacta
3000acaacatttc ccccgcatcc cccttccaaa caacaatccc cctctaccta
aaactcacag 3060ccctcgctgt cactttccta ggacttctaa cagccctaga
cctcaactac ctaaccaaca 3120aacttaaaat aaaatcccca ctatgcacat
tttatttctc caacatactc ggattctacc 3180ctagcatcac acaccgcaca
atcccctatc taggccttct tacgagccaa aacctgcccc 3240tactcctcct
agacctaacc tgactagaaa agctattacc taaaacaatt tcacagcacc
3300aaatctccac ctccatcatc acctcaaccc aaaaaggcat aattaaactt
tacttcctct 3360ctttcttctt cccactcat 3379230DNAArtificial
Sequencesynthetic primer 3.4 kb deletion forward 2tagactacgt
acatactaac cctactccta 30319DNAArtificial Sequencesynthetic primer
3.4 kb deletion reverse 3gaggtaggat tggtgctgt 19421DNAArtificial
Sequencesynthetic primer mtDNA genome forward 4cgttccagtg
agttcaccct c 21523DNAArtificial Sequencesynthetic primer mtDNA
genome reverse 5cactctttac gccggcttct att 23620DNAArtificial
Sequencesynthetic primer TNF nuclear gene forward 6cctgccccaa
tccctttatt 20721DNAArtificial Sequencesynthetic primer TNF nuclear
gene reverse 7ggtttcgaag tggtggtctt g 21816569DNAHomo
sapiensmisc_feature(3107)..(3107)n is a, c, g or t 8gatcacaggt
ctatcaccct attaaccact cacgggagct ctccatgcat ttggtatttt 60cgtctggggg
gtatgcacgc gatagcattg cgagacgctg gagccggagc accctatgtc
120gcagtatctg tctttgattc ctgcctcatc ctattattta tcgcacctac
gttcaatatt 180acaggcgaac atacttacta aagtgtgtta attaattaat
gcttgtagga cataataata 240acaattgaat gtctgcacag ccactttcca
cacagacatc ataacaaaaa atttccacca 300aaccccccct cccccgcttc
tggccacagc acttaaacac atctctgcca aaccccaaaa 360acaaagaacc
ctaacaccag cctaaccaga tttcaaattt tatcttttgg cggtatgcac
420ttttaacagt caccccccaa ctaacacatt attttcccct cccactccca
tactactaat 480ctcatcaata caacccccgc ccatcctacc cagcacacac
acaccgctgc taaccccata 540ccccgaacca accaaacccc aaagacaccc
cccacagttt atgtagctta cctcctcaaa 600gcaatacact gaaaatgttt
agacgggctc acatcacccc ataaacaaat aggtttggtc 660ctagcctttc
tattagctct tagtaagatt acacatgcaa gcatccccgt tccagtgagt
720tcaccctcta aatcaccacg atcaaaagga acaagcatca agcacgcagc
aatgcagctc 780aaaacgctta gcctagccac acccccacgg gaaacagcag
tgattaacct ttagcaataa 840acgaaagttt aactaagcta tactaacccc
agggttggtc aatttcgtgc cagccaccgc 900ggtcacacga ttaacccaag
tcaatagaag ccggcgtaaa gagtgtttta gatcaccccc 960tccccaataa
agctaaaact cacctgagtt gtaaaaaact ccagttgaca caaaatagac
1020tacgaaagtg gctttaacat atctgaacac acaatagcta agacccaaac
tgggattaga 1080taccccacta tgcttagccc taaacctcaa cagttaaatc
aacaaaactg ctcgccagaa 1140cactacgagc cacagcttaa aactcaaagg
acctggcggt gcttcatatc cctctagagg 1200agcctgttct gtaatcgata
aaccccgatc aacctcacca cctcttgctc agcctatata 1260ccgccatctt
cagcaaaccc tgatgaaggc tacaaagtaa gcgcaagtac ccacgtaaag
1320acgttaggtc aaggtgtagc ccatgaggtg gcaagaaatg ggctacattt
tctaccccag 1380aaaactacga tagcccttat gaaacttaag ggtcgaaggt
ggatttagca gtaaactaag 1440agtagagtgc ttagttgaac agggccctga
agcgcgtaca caccgcccgt caccctcctc 1500aagtatactt caaaggacat
ttaactaaaa cccctacgca tttatataga ggagacaagt 1560cgtaacatgg
taagtgtact ggaaagtgca cttggacgaa ccagagtgta gcttaacaca
1620aagcacccaa cttacactta ggagatttca acttaacttg accgctctga
gctaaaccta 1680gccccaaacc cactccacct tactaccaga caaccttagc
caaaccattt acccaaataa 1740agtataggcg atagaaattg aaacctggcg
caatagatat agtaccgcaa gggaaagatg 1800aaaaattata accaagcata
atatagcaag gactaacccc tataccttct gcataatgaa 1860ttaactagaa
ataactttgc aaggagagcc aaagctaaga cccccgaaac cagacgagct
1920acctaagaac agctaaaaga gcacacccgt ctatgtagca aaatagtggg
aagatttata 1980ggtagaggcg acaaacctac cgagcctggt gatagctggt
tgtccaagat agaatcttag 2040ttcaacttta aatttgccca cagaaccctc
taaatcccct tgtaaattta actgttagtc 2100caaagaggaa cagctctttg
gacactagga aaaaaccttg tagagagagt aaaaaattta 2160acacccatag
taggcctaaa agcagccacc aattaagaaa gcgttcaagc tcaacaccca
2220ctacctaaaa aatcccaaac atataactga actcctcaca cccaattgga
ccaatctatc 2280accctataga agaactaatg ttagtataag taacatgaaa
acattctcct ccgcataagc 2340ctgcgtcaga ttaaaacact gaactgacaa
ttaacagccc aatatctaca atcaaccaac 2400aagtcattat taccctcact
gtcaacccaa cacaggcatg ctcataagga aaggttaaaa 2460aaagtaaaag
gaactcggca aatcttaccc cgcctgttta ccaaaaacat cacctctagc
2520atcaccagta ttagaggcac cgcctgccca gtgacacatg tttaacggcc
gcggtaccct 2580aaccgtgcaa aggtagcata atcacttgtt ccttaaatag
ggacctgtat gaatggctcc 2640acgagggttc agctgtctct tacttttaac
cagtgaaatt gacctgcccg tgaagaggcg 2700ggcataacac agcaagacga
gaagacccta tggagcttta atttattaat gcaaacagta 2760cctaacaaac
ccacaggtcc taaactacca aacctgcatt aaaaatttcg gttggggcga
2820cctcggagca gaacccaacc tccgagcagt acatgctaag acttcaccag
tcaaagcgaa 2880ctactatact caattgatcc aataacttga ccaacggaac
aagttaccct agggataaca 2940gcgcaatcct attctagagt ccatatcaac
aatagggttt acgacctcga tgttggatca 3000ggacatcccg atggtgcagc
cgctattaaa ggttcgtttg ttcaacgatt aaagtcctac 3060gtgatctgag
ttcagaccgg agtaatccag gtcggtttct atctacnttc aaattcctcc
3120ctgtacgaaa ggacaagaga aataaggcct acttcacaaa gcgccttccc
ccgtaaatga 3180tatcatctca acttagtatt atacccacac ccacccaaga
acagggtttg ttaagatggc 3240agagcccggt aatcgcataa aacttaaaac
tttacagtca gaggttcaat tcctcttctt 3300aacaacatac ccatggccaa
cctcctactc ctcattgtac ccattctaat cgcaatggca 3360ttcctaatgc
ttaccgaacg aaaaattcta ggctatatac aactacgcaa aggccccaac
3420gttgtaggcc cctacgggct actacaaccc ttcgctgacg ccataaaact
cttcaccaaa 3480gagcccctaa aacccgccac atctaccatc accctctaca
tcaccgcccc gaccttagct 3540ctcaccatcg ctcttctact atgaaccccc
ctccccatac ccaaccccct ggtcaacctc 3600aacctaggcc tcctatttat
tctagccacc tctagcctag ccgtttactc aatcctctga 3660tcagggtgag
catcaaactc aaactacgcc ctgatcggcg cactgcgagc agtagcccaa
3720acaatctcat atgaagtcac cctagccatc attctactat caacattact
aataagtggc 3780tcctttaacc tctccaccct tatcacaaca caagaacacc
tctgattact cctgccatca 3840tgacccttgg ccataatatg atttatctcc
acactagcag agaccaaccg aacccccttc 3900gaccttgccg aaggggagtc
cgaactagtc tcaggcttca acatcgaata cgccgcaggc 3960cccttcgccc
tattcttcat agccgaatac acaaacatta ttataataaa caccctcacc
4020actacaatct tcctaggaac aacatatgac gcactctccc ctgaactcta
cacaacatat 4080tttgtcacca agaccctact tctaacctcc ctgttcttat
gaattcgaac agcatacccc 4140cgattccgct acgaccaact catacacctc
ctatgaaaaa acttcctacc actcacccta 4200gcattactta tatgatatgt
ctccataccc attacaatct ccagcattcc ccctcaaacc 4260taagaaatat
gtctgataaa agagttactt tgatagagta aataatagga gcttaaaccc
4320ccttatttct aggactatga gaatcgaacc catccctgag aatccaaaat
tctccgtgcc 4380acctatcaca ccccatccta aagtaaggtc agctaaataa
gctatcgggc ccataccccg 4440aaaatgttgg ttataccctt cccgtactaa
ttaatcccct ggcccaaccc gtcatctact 4500ctaccatctt tgcaggcaca
ctcatcacag cgctaagctc gcactgattt tttacctgag 4560taggcctaga
aataaacatg ctagctttta ttccagttct aaccaaaaaa ataaaccctc
4620gttccacaga agctgccatc aagtatttcc tcacgcaagc aaccgcatcc
ataatccttc 4680taatagctat cctcttcaac aatatactct ccggacaatg
aaccataacc aatactacca 4740atcaatactc atcattaata atcataatag
ctatagcaat aaaactagga atagccccct 4800ttcacttctg agtcccagag
gttacccaag gcacccctct gacatccggc ctgcttcttc 4860tcacatgaca
aaaactagcc cccatctcaa tcatatacca aatctctccc tcactaaacg
4920taagccttct cctcactctc tcaatcttat ccatcatagc aggcagttga
ggtggattaa 4980accaaaccca gctacgcaaa atcttagcat actcctcaat
tacccacata ggatgaataa 5040tagcagttct accgtacaac cctaacataa
ccattcttaa tttaactatt tatattatcc 5100taactactac cgcattccta
ctactcaact taaactccag caccacgacc ctactactat 5160ctcgcacctg
aaacaagcta acatgactaa cacccttaat tccatccacc ctcctctccc
5220taggaggcct gcccccgcta accggctttt tgcccaaatg ggccattatc
gaagaattca 5280caaaaaacaa tagcctcatc atccccacca tcatagccac
catcaccctc cttaacctct 5340acttctacct acgcctaatc tactccacct
caatcacact actccccata tctaacaacg 5400taaaaataaa atgacagttt
gaacatacaa aacccacccc attcctcccc acactcatcg 5460cccttaccac
gctactccta cctatctccc cttttatact aataatctta tagaaattta
5520ggttaaatac agaccaagag ccttcaaagc cctcagtaag ttgcaatact
taatttctgt 5580aacagctaag gactgcaaaa ccccactctg catcaactga
acgcaaatca gccactttaa 5640ttaagctaag cccttactag accaatggga
cttaaaccca caaacactta gttaacagct 5700aagcacccta atcaactggc
ttcaatctac ttctcccgcc gccgggaaaa aaggcgggag 5760aagccccggc
aggtttgaag ctgcttcttc gaatttgcaa ttcaatatga aaatcacctc
5820ggagctggta aaaagaggcc taacccctgt ctttagattt acagtccaat
gcttcactca 5880gccattttac ctcaccccca ctgatgttcg ccgaccgttg
actattctct acaaaccaca 5940aagacattgg aacactatac ctattattcg
gcgcatgagc tggagtccta ggcacagctc 6000taagcctcct tattcgagcc
gagctgggcc agccaggcaa ccttctaggt aacgaccaca 6060tctacaacgt
tatcgtcaca gcccatgcat ttgtaataat cttcttcata gtaataccca
6120tcataatcgg aggctttggc aactgactag ttcccctaat aatcggtgcc
cccgatatgg 6180cgtttccccg cataaacaac ataagcttct gactcttacc
tccctctctc ctactcctgc 6240tcgcatctgc tatagtggag gccggagcag
gaacaggttg aacagtctac cctcccttag 6300cagggaacta ctcccaccct
ggagcctccg tagacctaac catcttctcc ttacacctag 6360caggtgtctc
ctctatctta ggggccatca atttcatcac aacaattatc aatataaaac
6420cccctgccat aacccaatac caaacgcccc tcttcgtctg atccgtccta
atcacagcag 6480tcctacttct cctatctctc ccagtcctag ctgctggcat
cactatacta ctaacagacc 6540gcaacctcaa caccaccttc ttcgaccccg
ccggaggagg agaccccatt ctataccaac 6600acctattctg atttttcggt
caccctgaag tttatattct tatcctacca ggcttcggaa 6660taatctccca
tattgtaact tactactccg gaaaaaaaga accatttgga tacataggta
6720tggtctgagc tatgatatca attggcttcc tagggtttat cgtgtgagca
caccatatat 6780ttacagtagg aatagacgta gacacacgag catatttcac
ctccgctacc ataatcatcg 6840ctatccccac cggcgtcaaa gtatttagct
gactcgccac actccacgga agcaatatga 6900aatgatctgc tgcagtgctc
tgagccctag gattcatctt tcttttcacc gtaggtggcc 6960tgactggcat
tgtattagca aactcatcac tagacatcgt actacacgac acgtactacg
7020ttgtagccca cttccactat gtcctatcaa taggagctgt atttgccatc
ataggaggct 7080tcattcactg atttccccta ttctcaggct acaccctaga
ccaaacctac gccaaaatcc 7140atttcactat catattcatc ggcgtaaatc
taactttctt cccacaacac tttctcggcc 7200tatccggaat gccccgacgt
tactcggact accccgatgc atacaccaca tgaaacatcc 7260tatcatctgt
aggctcattc atttctctaa cagcagtaat attaataatt ttcatgattt
7320gagaagcctt cgcttcgaag cgaaaagtcc taatagtaga agaaccctcc
ataaacctgg 7380agtgactata tggatgcccc ccaccctacc acacattcga
agaacccgta tacataaaat 7440ctagacaaaa aaggaaggaa tcgaaccccc
caaagctggt ttcaagccaa ccccatggcc 7500tccatgactt tttcaaaaag
gtattagaaa aaccatttca taactttgtc aaagttaaat 7560tataggctaa
atcctatata tcttaatggc acatgcagcg caagtaggtc tacaagacgc
7620tacttcccct atcatagaag agcttatcac ctttcatgat cacgccctca
taatcatttt 7680ccttatctgc ttcctagtcc tgtatgccct tttcctaaca
ctcacaacaa aactaactaa 7740tactaacatc tcagacgctc aggaaataga
aaccgtctga actatcctgc ccgccatcat 7800cctagtcctc atcgccctcc
catccctacg catcctttac ataacagacg aggtcaacga 7860tccctccctt
accatcaaat caattggcca ccaatggtac tgaacctacg agtacaccga
7920ctacggcgga ctaatcttca actcctacat acttccccca ttattcctag
aaccaggcga 7980cctgcgactc cttgacgttg acaatcgagt agtactcccg
attgaagccc ccattcgtat 8040aataattaca tcacaagacg tcttgcactc
atgagctgtc cccacattag gcttaaaaac 8100agatgcaatt cccggacgtc
taaaccaaac cactttcacc gctacacgac cgggggtata 8160ctacggtcaa
tgctctgaaa tctgtggagc aaaccacagt ttcatgccca tcgtcctaga
8220attaattccc ctaaaaatct ttgaaatagg gcccgtattt accctatagc
accccctcta 8280ccccctctag agcccactgt aaagctaact tagcattaac
cttttaagtt aaagattaag 8340agaaccaaca cctctttaca gtgaaatgcc
ccaactaaat actaccgtat ggcccaccat 8400aattaccccc atactcctta
cactattcct catcacccaa ctaaaaatat taaacacaaa 8460ctaccaccta
cctccctcac caaagcccat aaaaataaaa aattataaca aaccctgaga
8520accaaaatga acgaaaatct gttcgcttca ttcattgccc ccacaatcct
aggcctaccc 8580gccgcagtac tgatcattct atttccccct ctattgatcc
ccacctccaa atatctcatc 8640aacaaccgac taatcaccac ccaacaatga
ctaatcaaac taacctcaaa acaaatgata 8700accatacaca acactaaagg
acgaacctga tctcttatac tagtatcctt aatcattttt 8760attgccacaa
ctaacctcct cggactcctg cctcactcat ttacaccaac cacccaacta
8820tctataaacc tagccatggc catcccctta tgagcgggca cagtgattat
aggctttcgc 8880tctaagatta aaaatgccct agcccacttc ttaccacaag
gcacacctac accccttatc 8940cccatactag ttattatcga aaccatcagc
ctactcattc aaccaatagc cctggccgta 9000cgcctaaccg ctaacattac
tgcaggccac ctactcatgc acctaattgg aagcgccacc 9060ctagcaatat
caaccattaa ccttccctct acacttatca tcttcacaat tctaattcta
9120ctgactatcc tagaaatcgc tgtcgcctta atccaagcct acgttttcac
acttctagta 9180agcctctacc tgcacgacaa cacataatga cccaccaatc
acatgcctat catatagtaa 9240aacccagccc atgaccccta acaggggccc
tctcagccct cctaatgacc tccggcctag 9300ccatgtgatt tcacttccac
tccataacgc tcctcatact aggcctacta accaacacac 9360taaccatata
ccaatgatgg cgcgatgtaa cacgagaaag cacataccaa ggccaccaca
9420caccacctgt ccaaaaaggc cttcgatacg ggataatcct atttattacc
tcagaagttt 9480ttttcttcgc aggatttttc tgagcctttt accactccag
cctagcccct accccccaat 9540taggagggca ctggccccca acaggcatca
ccccgctaaa tcccctagaa gtcccactcc 9600taaacacatc cgtattactc
gcatcaggag tatcaatcac ctgagctcac catagtctaa 9660tagaaaacaa
ccgaaaccaa ataattcaag cactgcttat tacaatttta ctgggtctct
9720attttaccct cctacaagcc tcagagtact tcgagtctcc cttcaccatt
tccgacggca 9780tctacggctc aacatttttt gtagccacag gcttccacgg
acttcacgtc attattggct 9840caactttcct cactatctgc ttcatccgcc
aactaatatt tcactttaca tccaaacatc 9900actttggctt cgaagccgcc
gcctgatact ggcattttgt agatgtggtt tgactatttc 9960tgtatgtctc
catctattga tgagggtctt actcttttag tataaatagt accgttaact
10020tccaattaac tagttttgac aacattcaaa aaagagtaat aaacttcgcc
ttaattttaa 10080taatcaacac cctcctagcc ttactactaa taattattac
attttgacta ccacaactca 10140acggctacat agaaaaatcc accccttacg
agtgcggctt cgaccctata tcccccgccc 10200gcgtcccttt ctccataaaa
ttcttcttag tagctattac cttcttatta tttgatctag 10260aaattgccct
ccttttaccc ctaccatgag ccctacaaac aactaacctg ccactaatag
10320ttatgtcatc cctcttatta atcatcatcc tagccctaag tctggcctat
gagtgactac 10380aaaaaggatt agactgaacc gaattggtat atagtttaaa
caaaacgaat gatttcgact 10440cattaaatta tgataatcat atttaccaaa
tgcccctcat ttacataaat attatactag 10500catttaccat ctcacttcta
ggaatactag tatatcgctc acacctcata tcctccctac 10560tatgcctaga
aggaataata ctatcgctgt tcattatagc tactctcata accctcaaca
10620cccactccct cttagccaat attgtgccta ttgccatact agtctttgcc
gcctgcgaag 10680cagcggtggg cctagcccta ctagtctcaa tctccaacac
atatggccta gactacgtac 10740ataacctaaa cctactccaa tgctaaaact
aatcgtccca acaattatat tactaccact 10800gacatgactt tccaaaaaac
acataatttg aatcaacaca accacccaca gcctaattat
10860tagcatcatc cctctactat tttttaacca aatcaacaac aacctattta
gctgttcccc 10920aaccttttcc tccgaccccc taacaacccc cctcctaata
ctaactacct gactcctacc 10980cctcacaatc atggcaagcc aacgccactt
atccagtgaa ccactatcac gaaaaaaact 11040ctacctctct atactaatct
ccctacaaat ctccttaatt ataacattca cagccacaga 11100actaatcata
ttttatatct tcttcgaaac cacacttatc cccaccttgg ctatcatcac
11160ccgatgaggc aaccagccag aacgcctgaa cgcaggcaca tacttcctat
tctacaccct 11220agtaggctcc cttcccctac tcatcgcact aatttacact
cacaacaccc taggctcact 11280aaacattcta ctactcactc tcactgccca
agaactatca aactcctgag ccaacaactt 11340aatatgacta gcttacacaa
tagcttttat agtaaagata cctctttacg gactccactt 11400atgactccct
aaagcccatg tcgaagcccc catcgctggg tcaatagtac ttgccgcagt
11460actcttaaaa ctaggcggct atggtataat acgcctcaca ctcattctca
accccctgac 11520aaaacacata gcctacccct tccttgtact atccctatga
ggcataatta taacaagctc 11580catctgccta cgacaaacag acctaaaatc
gctcattgca tactcttcaa tcagccacat 11640agccctcgta gtaacagcca
ttctcatcca aaccccctga agcttcaccg gcgcagtcat 11700tctcataatc
gcccacgggc ttacatcctc attactattc tgcctagcaa actcaaacta
11760cgaacgcact cacagtcgca tcataatcct ctctcaagga cttcaaactc
tactcccact 11820aatagctttt tgatgacttc tagcaagcct cgctaacctc
gccttacccc ccactattaa 11880cctactggga gaactctctg tgctagtaac
cacgttctcc tgatcaaata tcactctcct 11940acttacagga ctcaacatac
tagtcacagc cctatactcc ctctacatat ttaccacaac 12000acaatggggc
tcactcaccc accacattaa caacataaaa ccctcattca cacgagaaaa
12060caccctcatg ttcatacacc tatcccccat tctcctccta tccctcaacc
ccgacatcat 12120taccgggttt tcctcttgta aatatagttt aaccaaaaca
tcagattgtg aatctgacaa 12180cagaggctta cgacccctta tttaccgaga
aagctcacaa gaactgctaa ctcatgcccc 12240catgtctaac aacatggctt
tctcaacttt taaaggataa cagctatcca ttggtcttag 12300gccccaaaaa
ttttggtgca actccaaata aaagtaataa ccatgcacac tactataacc
12360accctaaccc tgacttccct aattcccccc atccttacca ccctcgttaa
ccctaacaaa 12420aaaaactcat acccccatta tgtaaaatcc attgtcgcat
ccacctttat tatcagtctc 12480ttccccacaa caatattcat gtgcctagac
caagaagtta ttatctcgaa ctgacactga 12540gccacaaccc aaacaaccca
gctctcccta agcttcaaac tagactactt ctccataata 12600ttcatccctg
tagcattgtt cgttacatgg tccatcatag aattctcact gtgatatata
12660aactcagacc caaacattaa tcagttcttc aaatatctac tcatcttcct
aattaccata 12720ctaatcttag ttaccgctaa caacctattc caactgttca
tcggctgaga gggcgtagga 12780attatatcct tcttgctcat cagttgatga
tacgcccgag cagatgccaa cacagcagcc 12840attcaagcaa tcctatacaa
ccgtatcggc gatatcggtt tcatcctcgc cttagcatga 12900tttatcctac
actccaactc atgagaccca caacaaatag cccttctaaa cgctaatcca
12960agcctcaccc cactactagg cctcctccta gcagcagcag gcaaatcagc
ccaattaggt 13020ctccacccct gactcccctc agccatagaa ggccccaccc
cagtctcagc cctactccac 13080tcaagcacta tagttgtagc aggaatcttc
ttactcatcc gcttccaccc cctagcagaa 13140aatagcccac taatccaaac
tctaacacta tgcttaggcg ctatcaccac tctgttcgca 13200gcagtctgcg
cccttacaca aaatgacatc aaaaaaatcg tagccttctc cacttcaagt
13260caactaggac tcataatagt tacaatcggc atcaaccaac cacacctagc
attcctgcac 13320atctgtaccc acgccttctt caaagccata ctatttatgt
gctccgggtc catcatccac 13380aaccttaaca atgaacaaga tattcgaaaa
ataggaggac tactcaaaac catacctctc 13440acttcaacct ccctcaccat
tggcagccta gcattagcag gaataccttt cctcacaggt 13500ttctactcca
aagaccacat catcgaaacc gcaaacatat catacacaaa cgcctgagcc
13560ctatctatta ctctcatcgc tacctccctg acaagcgcct atagcactcg
aataattctt 13620ctcaccctaa caggtcaacc tcgcttcccc acccttacta
acattaacga aaataacccc 13680accctactaa accccattaa acgcctggca
gccggaagcc tattcgcagg atttctcatt 13740actaacaaca tttcccccgc
atcccccttc caaacaacaa tccccctcta cctaaaactc 13800acagccctcg
ctgtcacttt cctaggactt ctaacagccc tagacctcaa ctacctaacc
13860aacaaactta aaataaaatc cccactatgc acattttatt tctccaacat
actcggattc 13920taccctagca tcacacaccg cacaatcccc tatctaggcc
ttcttacgag ccaaaacctg 13980cccctactcc tcctagacct aacctgacta
gaaaagctat tacctaaaac aatttcacag 14040caccaaatct ccacctccat
catcacctca acccaaaaag gcataattaa actttacttc 14100ctctctttct
tcttcccact catcctaacc ctactcctaa tcacataacc tattcccccg
14160agcaatctca attacaatat atacaccaac aaacaatgtt caaccagtaa
ctactactaa 14220tcaacgccca taatcataca aagcccccgc accaatagga
tcctcccgaa tcaaccctga 14280cccctctcct tcataaatta ttcagcttcc
tacactatta aagtttacca caaccaccac 14340cccatcatac tctttcaccc
acagcaccaa tcctacctcc atcgctaacc ccactaaaac 14400actcaccaag
acctcaaccc ctgaccccca tgcctcagga tactcctcaa tagccatcgc
14460tgtagtatat ccaaagacaa ccatcattcc ccctaaataa attaaaaaaa
ctattaaacc 14520catataacct cccccaaaat tcagaataat aacacacccg
accacaccgc taacaatcaa 14580tactaaaccc ccataaatag gagaaggctt
agaagaaaac cccacaaacc ccattactaa 14640acccacactc aacagaaaca
aagcatacat cattattctc gcacggacta caaccacgac 14700caatgatatg
aaaaaccatc gttgtatttc aactacaaga acaccaatga ccccaatacg
14760caaaactaac cccctaataa aattaattaa ccactcattc atcgacctcc
ccaccccatc 14820caacatctcc gcatgatgaa acttcggctc actccttggc
gcctgcctga tcctccaaat 14880caccacagga ctattcctag ccatgcacta
ctcaccagac gcctcaaccg ccttttcatc 14940aatcgcccac atcactcgag
acgtaaatta tggctgaatc atccgctacc ttcacgccaa 15000tggcgcctca
atattcttta tctgcctctt cctacacatc gggcgaggcc tatattacgg
15060atcatttctc tactcagaaa cctgaaacat cggcattatc ctcctgcttg
caactatagc 15120aacagccttc ataggctatg tcctcccgtg aggccaaata
tcattctgag gggccacagt 15180aattacaaac ttactatccg ccatcccata
cattgggaca gacctagttc aatgaatctg 15240aggaggctac tcagtagaca
gtcccaccct cacacgattc tttacctttc acttcatctt 15300gcccttcatt
attgcagccc tagcaacact ccacctccta ttcttgcacg aaacgggatc
15360aaacaacccc ctaggaatca cctcccattc cgataaaatc accttccacc
cttactacac 15420aatcaaagac gccctcggct tacttctctt ccttctctcc
ttaatgacat taacactatt 15480ctcaccagac ctcctaggcg acccagacaa
ttatacccta gccaacccct taaacacccc 15540tccccacatc aagcccgaat
gatatttcct attcgcctac acaattctcc gatccgtccc 15600taacaaacta
ggaggcgtcc ttgccctatt actatccatc ctcatcctag caataatccc
15660catcctccat atatccaaac aacaaagcat aatatttcgc ccactaagcc
aatcacttta 15720ttgactccta gccgcagacc tcctcattct aacctgaatc
ggaggacaac cagtaagcta 15780cccttttacc atcattggac aagtagcatc
cgtactatac ttcacaacaa tcctaatcct 15840aataccaact atctccctaa
ttgaaaacaa aatactcaaa tgggcctgtc cttgtagtat 15900aaactaatac
accagtcttg taaaccggag atgaaaacct ttttccaagg acaaatcaga
15960gaaaaagtct ttaactccac cattagcacc caaagctaag attctaattt
aaactattct 16020ctgttctttc atggggaagc agatttgggt accacccaag
tattgactca cccatcaaca 16080accgctatgt atttcgtaca ttactgccag
ccaccatgaa tattgtacgg taccataaat 16140acttgaccac ctgtagtaca
taaaaaccca atccacatca aaaccccctc cccatgctta 16200caagcaagta
cagcaatcaa ccctcaacta tcacacatca actgcaactc caaagccacc
16260cctcacccac taggatacca acaaacctac ccacccttaa cagtacatag
tacataaagc 16320catttaccgt acatagcaca ttacagtcaa atcccttctc
gtccccatgg atgacccccc 16380tcagataggg gtcccttgac caccatcctc
cgtgaaatca atatcccgca caagagtgct 16440actctcctcg ctccgggccc
ataacacttg ggggtagcta aagtgaactg tatccgacat 16500ctggttccta
cttcagggtc ataaagccta aatagcccac acgttcccct taaataagac
16560atcacgatg 16569922DNAArtificial Sequencesynthetic primer
forward 3.4 kb deleted sequence 9cccactcatc acctaaacct ac
221020DNAArtificial Sequencesynthetic primer reverse 3.4 kb deleted
sequence 10ggtaggagtc aggtagttag 20
References

D00001

D00002

D00003

D00004

D00005

D00006

D00007

S00001
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.