Composition For Low Temperature
RETTEMEYER; DIRK ; et al.
U.S. patent application number 15/039690 was filed with the patent office on 2016-12-29 for composition for low temperature. This patent application is currently assigned to BASF SE. The applicant listed for this patent is BASF SE. Invention is credited to DIRK RETTEMEYER, Ronny RINKLIEB, Markus SHERER.
| Application Number | 20160376518 15/039690 |
| Document ID | / |
| Family ID | 49667064 |
| Filed Date | 2016-12-29 |
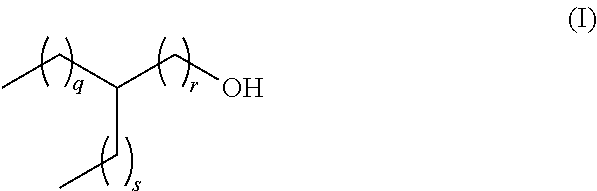
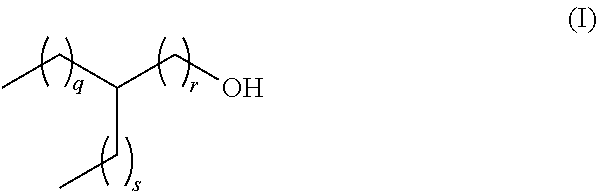
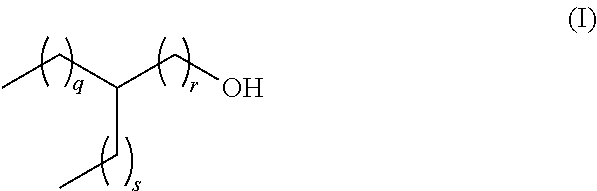
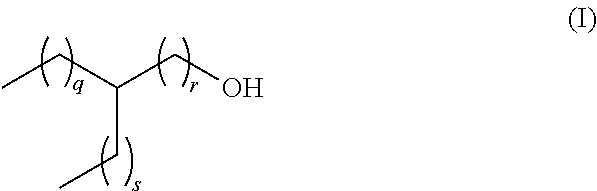

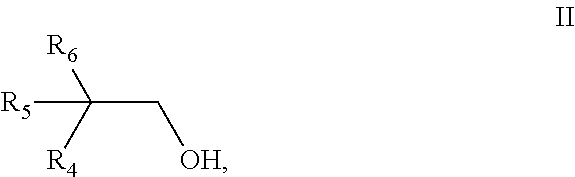
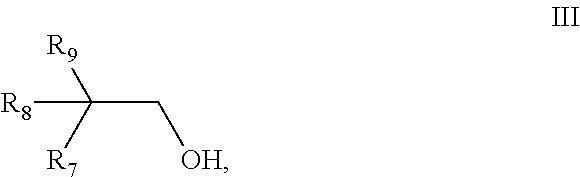
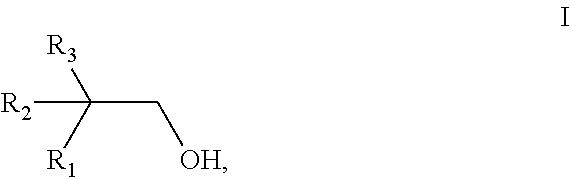
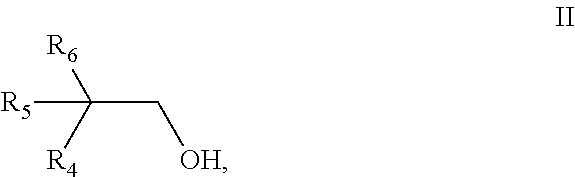
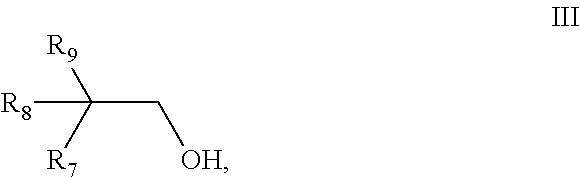
| United States Patent Application | 20160376518 |
| Kind Code | A1 |
| RETTEMEYER; DIRK ; et al. | December 29, 2016 |
COMPOSITION FOR LOW TEMPERATURE
Abstract
Lubricant composition comprising a dicarboxylic acid ester component which is formed from a dicarboxylic acid selected from the list consisting of adipic acid, phthalic acid, pimilic acid, suberic acid, azelaic acid and sebacic acid, and mixtures thereof and a branched aliphatic alcohol R--OH which is defined according to the following formula (I) ##STR00001## whereas q, r and s are defined as follows, q+r=4 to 9, s=0 to 5, q=1 to 8, and r=1 to 6, and an ethylene-propylene copolymer.
| Inventors: | RETTEMEYER; DIRK; (Hueckelhoven, DE) ; RINKLIEB; Ronny; (Dusseldorf, DE) ; SHERER; Markus; (Mannheim, DE) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | BASF SE Ludwigshafen DE |
||||||||||
| Family ID: | 49667064 | ||||||||||
| Appl. No.: | 15/039690 | ||||||||||
| Filed: | November 27, 2014 | ||||||||||
| PCT Filed: | November 27, 2014 | ||||||||||
| PCT NO: | PCT/EP2014/075816 | ||||||||||
| 371 Date: | May 26, 2016 |
| Current U.S. Class: | 508/499 |
| Current CPC Class: | C10M 105/36 20130101; C10M 2207/282 20130101; C10N 2040/04 20130101; C10N 2030/68 20200501; C10M 2207/30 20130101; C10M 2205/022 20130101; C10M 2207/2825 20130101; C10M 2205/024 20130101; C10N 2040/08 20130101; C10N 2040/25 20130101; C10M 2205/028 20130101; C10M 111/04 20130101; C10N 2030/02 20130101; C10M 2207/281 20130101; C10M 2205/0245 20130101; C10N 2030/10 20130101; C10N 2020/02 20130101; C10M 2205/0285 20130101; C10N 2040/02 20130101; C10N 2040/135 20200501; C10N 2040/30 20130101; C10M 169/041 20130101; C10M 2205/0225 20130101; C10M 2205/022 20130101; C10M 2205/024 20130101; C10M 2205/0225 20130101; C10M 2205/0245 20130101 |
| International Class: | C10M 111/04 20060101 C10M111/04; C10M 169/04 20060101 C10M169/04 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Nov 28, 2013 | EP | 13194911.7 |
Claims
1.-10. (canceled)
11. A lubricant composition comprising based on the total weight of the lubricant composition a) 35 to 95 wt.-% of a dicarboxylic acid ester component which is selected from diisononyladipate (DNA) and di-(2-ethylhexyl)adipate (DOA), b) 5 to 30 wt.-% of an ethylene-propylene copolymer, and c) 5 to 25 wt.-% of a monocarboxylic acid ester.
12. The lubricant composition according to claim 11, wherein the lubricant composition has a kinematic viscosity according to industrial standard DIN 51562-1 of not more than 1600 mm.sup.2/s at -30.degree. C. and of at least 7.5 mm.sup.2/s at 100.degree. C.
13. The lubricant composition according to claim 11, having a viscosity index according to the industrial standard DIN ISO 2909 of at least 160.
14. The lubricant composition according to claim 11, wherein the dicarboxylic acid ester has a kinematic viscosity according to DIN 51562-1 in the range of 2 to 15 mm.sup.2/s at 100.degree. C.
15. The lubricant composition according to claim 11, wherein the ethylene-propylene copolymer has a kinematic viscosity according to JIS K 2283 at 100.degree. C. in the range of from 1000 to 2200 mm.sup.2/s.
16. The lubricant composition according to claim 11, further comprising a base oil component having a kinematic viscosity according to DIN 51562-1 at 100.degree. C. in the range of from 1 to 5 mm.sup.2/s.
17. The lubricant composition according to claim 11, wherein the monocarboxylic acid ester is selected from the list consisting of 2-ethylhexyloleate, 2-ethylhexylcocoate, 2-ethylhexylpalmitate, 2-ethylhexylstearate, 2-ethylhexyltallowate, and mixtures thereof.
18. The lubricant composition according to claim 11, wherein the ratio of the dicarboxylic acid ester component to the oligomeric copolymers in the lubricant compositions according to the present invention is in the range of from 2:1 to 19:1 based on the relative weight of these components in the lubricant compositions according to the present invention.
19. The lubricant composition according to claim 11, further comprising an additive component which is present in an amount of 0.1 to 20 wt % of the total lubricant composition.
20. A vehicle transmission oil, axle oil, industrial transmission oil, industrial gear oil, compressor oil, turbine oil, hydraulic oil or motor oil which comprises the lubricant composition according to claim 11.
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application is a national stage application (under 35 U.S.C. .sctn.371) of PCT/EP2014/075816, filed Nov. 27, 2014, which claims benefit of European Application No. 13194911.7, filed Nov. 28, 2013, both of which are incorporated herein by reference in their entirety.
DESCRIPTION
[0002] The present invention relates to the field of lubricants. The lubricant compositions contain a di-carboxylic acid ester and an ethylene-propylene copolymer. The lubricant compositions can be used in a variety of different oil formulations required in motor vehicles.
TECHNICAL BACKGROUND
[0003] Commercially available lubricant compositions are based on a multitude of different natural or synthetic components. The resulting properties of the various existing lubricant compositions are tailored to the specific technical requirements by the addition of further components and selected combinations thereof. In this way, lubricant compositions are obtained which can fulfill the complex requirements associated with the various special technical applications in the field of motor vehicles, automotive engines and other machinery.
[0004] Typically, lubricant compositions are needed that provide high shear stability, improved low-temperature viscosity, minimum degree of evaporation loss, good fuel efficiency, acceptable seal compatibility and excellent wear protection.
[0005] One especially desired set of properties in high performance lubrication applications is an excellent low temperature profile indicated by favorable low temperature viscosity in combination with excellent dynamic behavior at high temperatures as indicated by high shear stability and high viscosity index.
[0006] Known lubricants which are able to fulfill such performance characteristics have been developed in the prior art by the addition of special thickening agents (viscosity index improving agents). Preferably, polyalphaolefin (PAO)-type base components have been modified with thickeners like polyisobutenes (PIB), oligomeric co-polymers (OCPs), polymethacrylates (PMAs) or high viscosity esters (complex esters) for achieving the desired set of properties.
[0007] U.S. Pat. No. 5,451,630 describes the general dilemma when using thickening agents in lubricant compositions because the increase of viscosity is directly related to the molecular weight of the polymeric thickening agent while on the other hand the shear stability decreases due to the greater tendency of breakage under shear and high temperature conditions.
[0008] U.S. Pat. No. 5,451,630 further suggests oligomeric copolymers which are demonstrated to provide good shear stability to lubricant compositions.
[0009] In WO 2007/144079 A2, a larger number of lubricant compositions have been described including a variety of different thickening agents like PIBs, OCPs, PMAs and high viscosity esters which have been demonstrated to be generally applicable as viscosity index improvers.
[0010] In addition, dicarboxylic acid esters like DIDA (diisodecyl adipate), DITA (diisotridecyl adipate) or TMTC (trimethylolpropane caprylate) have also been added to such lubricant compositions as solubilizers for polar additive types.
[0011] However, there is a continued need for new lubricant compositions which exceed the performance characteristics of the already existing lubricant compositions. This is particularly important for lubricant compositions that are designed for the use under extreme conditions. It is particularly difficult to develop lubricant compositions that offer the desired excellent rheological properties at very low temperatures as well as at elevated temperatures.
[0012] Surprisingly, lubricant compositions are provided by the present invention comprising the combination of a dicarboxylic acid ester component with an ethylene-propylene copolymer having excellent dynamic behavior at high temperature and unusual high viscosity index while the corresponding viscosity at very low temperatures still remains only moderate. Another important property is the high permanent shear stability of the lubricant compositions according to the present invention.
DESCRIPTION OF THE INVENTION
[0013] The present invention relates to a lubricant composition comprising [0014] a) a dicarboxylic acid ester component which is formed from a dicarboxylic acid selected from the list consisting of adipic acid, phthalic acid, pimilic acid, suberic acid, azelaic acid and sebacic acid, and mixtures thereof and a branched aliphatic alcohol R--OH which is defined according to the following formula (I)
[0014] ##STR00002## [0015] whereas q, r and s are defined as follows, [0016] q+r=4 to 9, [0017] s=0 to 5, [0018] q=1 to 8, and [0019] r=1 to 6, [0020] and [0021] b) an ethylene-propylene copolymer.
[0022] In another preferred embodiment, in the lubricant composition from above, the residue R in the branched aliphatic alcohol R--OH is selected from ethylhexyl, methyloctyl, propylheptyl, butyloctyl and mixtures thereof whereas q, r and s are defined as follows
TABLE-US-00001 R in R--OH Ethylhexyl q + r = 4 s = 1 q = 1 r = 3 q = 2 r = 2 q = 3 r = 1 Methyloctyl q + r = 6 s = 0 q = 1 r = 5 q = 2 r = 4 q = 3 r = 3 q = 4 r = 2 q = 5 r = 1 Propylheptyl q + r = 5 s = 2 q = 1 r = 4 q = 2 r = 3 q = 3 r = 2 q = 4 r = 1 Butyloctyl q + r = 6 s = 3 q = 1 r = 5 q = 2 r = 4 q = 3 r = 3 q = 4 r = 2 q = 5 r = 1
[0023] In another preferred embodiment, the residue R in the branched aliphatic alcohol R--OH from above according to formula (I)
##STR00003##
[0024] is defined by q, r and s as follows, [0025] q+r=4 to 9, [0026] s=0 to 5, [0027] q=3 to 8, and [0028] r=1.
[0029] In another preferred embodiment, the lubricant compositions as defined above additionally have a kinematic viscosity according to industrial standard DIN 51562-1 of not more than 1600 mm.sup.2/s at -30.degree. C. and of at least 7.5 mm.sup.2/s at 100.degree. C.
[0030] In another preferred embodiment, the lubricant composition comprises [0031] a) a dicarboxylic acid ester component [0032] b) an ethylene-propylene copolymer, wherein the lubricant composition has a kinematic viscosity according to industrial standard DIN 51562-1 of not more than 1600 mm.sup.2/s at -30.degree. C. and of at least 7.5 mm.sup.2/s at 100.degree. C.
[0033] In another preferred embodiment, the lubricant composition has a viscosity index according to the industrial standard DIN ISO 2909 of at least 160, preferably of at least 180.
[0034] In another preferred embodiment, the dicarboxylic acid ester has a kinematic viscosity according to DIN 51562-1 in the range of from 2 to 15 mm.sup.2/s at 100.degree. C.
[0035] In another preferred embodiment, the dicarboxylic acid ester is selected from the list consisting of di-(2-propylheptyl)adipate (DPHA), di-isononyladipate (DNA), di-(2-ethylhexyl)adipate (DOA), dipropylheptylphthalate (DPHP) and mixtures thereof.
[0036] In another preferred embodiment, the ethylene-propylene copolymer has a kinematic viscosity according to ASTM D445 at 100.degree. C. in the range of from 1000 to 2200 mm.sup.2/s.
[0037] In another preferred embodiment, the lubricant composition further comprises a base oil component having a kinematic viscosity according to DIN 51562-1 of from about 1 to 5 mm.sup.2/s at 100.degree. C., preferably a PAO-2 component.
[0038] In another preferred embodiment, the lubricant composition further comprises a monocarboxylic acid ester.
[0039] In another preferred embodiment, the monocarboxylic acid ester is selected from the list consisting of 2-ethylhexyloleate, 2-ethylhexylcocoate, 2-ethylhexylpalmitate, 2-ethylhexylstearate, and 2-ethylhexyltallowate, and mixtures thereof.
[0040] In another preferred embodiment, the ratio of the dicarboxylic acid ester component to the oligomeric copolymers in the lubricant compositions according to the present invention is in the range of from 2:1 to 19:1, preferably 3:1 to 10:1 based on the relative weight of these components in the lubricant compositions according to the present invention.
[0041] In another preferred embodiment, the lubricant composition further comprises an additive component which is present in an amount of 0.1 to 20 wt % of the total lubricant composition.
[0042] In another preferred embodiment, the lubricant compositions are used as vehicle transmission oil, axle oil, industrial transmission oil, compressor oil, turbine oil, hydraulic oil or motor oil.
[0043] The lubricant composition according to the present invention comprises [0044] a) a dicarboxylic acid ester component which is formed from a dicarboxylic acid selected from the list consisting of adipic acid, phthalic acid, pimilic acid, suberic acid, azelaic acid and sebacic acid and mixtures thereof, and a branched aliphatic alcohol R--OH which is defined according to the following formula (I)
[0044] ##STR00004## [0045] whereas q, r and s are defined as follows, [0046] q+r=4 to 9, [0047] s=0 to 5, [0048] q=1 to 8, and [0049] r=1 to 6, [0050] and [0051] b) an ethylene-propylene copolymer.
[0052] Preferably, the branched aliphatic alcohol R--OH according to formula (I) can be a primary C.sub.7 to C.sub.12 alcohol, wherein the alkyl side chain is C.sub.1 to C.sub.6 alkyl (s=0 to 5). The alkyl side chain can be linear or branched alkyl group while linear alkyl group is preferred for the alkyl side chain.
[0053] Accordingly, the main alkyl chain in residue R is C.sub.6 to C.sub.11. Accordingly, the residue R in R--OH of formula (I) includes methylhexyl, ethylhexyl, propylhexyl, butylhexyl, pentylhexyl and hexylhexyl, methylheptyl, ethylheptyl, propylheptyl, butylheptyl and pentylheptyl, methyloctyl, ethyloctyl, propyloctyl, and butyloctyl, methylnonyl, ethylnonyl, and propylnonyl, methyldecyl and ethyldecyl, and methylundecyl.
[0054] Especially preferred alcohols R--OH from the above list include residue R being ethylhexyl, methyloctyl, propylheptyl and butyloctyl.
[0055] Most preferably, the residue R in R--OH in the above lubricant composition is selected from ethylhexyl, methyloctyl, propylheptyl, butyloctyl and mixtures thereof defined by q, r and s as follows
TABLE-US-00002 R in R--OH Ethylhexyl q + r = 4 s = 1 q = 1 r = 3 q = 2 r = 2 q = 3 r = 1 Methyloctyl q + r = 6 s = 0 q = 1 r = 5 q = 2 r = 4 q = 3 r = 3 q = 4 r = 2 q = 5 r = 1 Propylheptyl q + r = 5 s = 2 q = 1 r = 4 q = 2 r = 3 q = 3 r = 2 q = 4 r = 1 Butyloctyl q + r = 6 s = 3 q = 1 r = 5 q = 2 r = 4 q = 3 r = 3 q = 4 r = 2 q = 5 r = 1
[0056] Especially preferred are the alcohols having q+r=4 to 9, r=1 and q=3 to 8, i.e. the primary aliphatic C.sub.7 to C.sub.12 alcohols in which the linear (which is preferred) or branched alkyl side chain C.sub.1 to C.sub.6 alkyl (s=0 to 5) is located at the 2-position of the primary alcohol. Such alcohols are typically named Guerbet alcohols as further explained below.
[0057] It is additionally preferred that the kinematic viscosity of the lubricant compositions from above based on the industrial standard DIN 51562-1 at a temperature of -30.degree. C. is not higher than 1600 mm.sup.2/s, preferably not higher than 1550 mm.sup.2/s, and even not higher than 1500 mm.sup.2/s, and/or, preferably, the kinematic viscosity based on the industrial standard DIN 51562-1 at a temperature of 100.degree. C. is at least 7.0 mm.sup.2/s, preferably at least 7.5 mm.sup.2/s, and even more preferably at least 8.0 mm.sup.2/s.
[0058] From a rheological perspective, the kinematic viscosity of the lubricant compositions according to the present invention based on the industrial standard DIN 51562-1 at a temperature of -30.degree. C. is, in its most generic definition, not higher than 1600 mm.sup.2/s, preferably not higher than 1550 mm.sup.2/s, and even not higher than 1500 mm.sup.2/s, even more preferably not higher than 1400 mm.sup.2/s and most preferably not higher than 1300 mm.sup.2/s. The kinematic viscosity of the lubricant compositions according to the present invention based on the industrial standard DIN 51562-1 at a temperature of -30.degree. C. is at least 500 mm.sup.2/s, more preferably at least 700 mm.sup.2/s, and even more preferably at least 900 mm.sup.2/s.
[0059] That is, the lubricant composition according to the present invention, when most generically defined, comprises a) a dicarboxylic acid ester component and b) an ethylene-propylene copolymer, while the resulting kinematic viscosity of the lubricant composition based on the industrial standard DIN 51562-1 at a temperature of -30.degree. C. is, in its most generic definition, not higher than 1600 mm.sup.2/s, preferably not higher than 1550 mm.sup.2/s, and even not higher than 1500 mm.sup.2/s.
[0060] The kinematic viscosity based on the industrial standard DIN 51562-1 at a temperature of 40.degree. C. is at least 25 mm.sup.2/s, preferably at least 30 mm.sup.2/s, and even more preferably at least 35 mm.sup.2/s.
[0061] The kinematic viscosity based on the industrial standard DIN 51562-1 at a temperature of 100.degree. C. is at least 7.0 mm.sup.2/s, preferably at least 7.5 mm.sup.2/s, and even more preferably at least 8.0 mm.sup.2/s.
[0062] The rheological profile of the lubricant compositions according to the present invention is especially defined by a kinematic viscosity based on the industrial standard DIN 51562-1 a temperature of -30.degree. C. which is not higher than 1600 mm.sup.2/s, preferably not higher than 1550 mm.sup.2/s, and even more preferably not higher than 1500 mm.sup.2/s and a kinematic viscosity based on the industrial standard DIN 51562-1 at a temperature of 100.degree. C. which is at least 7.0 mm.sup.2/s, preferably at least 7.5 mm.sup.2/s, and even more preferably at least 8.0 mm.sup.2/s.
[0063] Further, the rheological profile of the lubricant compositions according to the present invention is characterized by a viscosity index based on the industrial standard DIN ISO 2909 of at least 160, more preferably of at least 190, and even more preferably of at least 200.
[0064] The lubricant compositions according to the present invention have a pour point according to DIN ISO 3016 of not higher than -50.degree. C., preferably not higher than -60.degree. C., and even more preferably not higher than -70.degree. C.
[0065] Preferably, the lubricant compositions according to the present invention have a kinematic viscosity based on the industrial standard DIN 51562-1 at a temperature of -30.degree. C. which is not higher than 1600 mm.sup.2/s, preferably not higher than 1550 mm.sup.2/s, and even not higher than 1500 mm.sup.2/s, and a kinematic viscosity based on the industrial standard DIN 51562-1 at a temperature of 100.degree. C. which is at least 7.0 mm.sup.2/s, preferably at least 7.5 mm.sup.2/s, and even more preferably at least 8.0 mm.sup.2/s, and a viscosity index based on the industrial standard DIN ISO 2909 of at least 160, more preferably of at least 180, and even more preferably of at least 200.
[0066] The lubricant compositions according to the present invention include the following components which are described below in more detail.
[0067] The lubricant compositions according to the present invention include as the first essential component a dicarboxylic acid ester component.
[0068] In its most generic definition as understood in the present invention, the dicarboxylic ester component as used in the lubricant compositions according to the present invention is defined as a dicarboxylic ester component having kinematic viscosity at 100.degree. C. (DIN 51562-1) in the range of 2 to 15 mm.sup.2/s, preferably 3 to 10 mm.sup.2/s, or even more preferably 4 to 8 mm.sup.2/s.
[0069] The dicarboxylic ester component has a pour point (DIN ISO 3016) in the range of from -20.degree. C. to -75.degree. C., preferably in the range of from -40.degree. C. to -60.degree. C. and most preferably in the range of from -48.degree. C. to -60.degree. C.
[0070] The viscosity index (DIN ISO 2909) of the dicarboxylic ester component is in the range of from 120 to 170, preferably of from 130 to 160, and most preferably of from 135 to 145.
[0071] The kinematic viscosity of the dicarboxylic ester component at 40.degree. C. (DIN 51562-1) is in the range of 7 to 50 mm.sup.2/s, preferably 10 to 30 mm.sup.2/s, and most preferably 12 to 28 mm.sup.2/s.
[0072] The amount of the dicarboxylic acid ester component in the lubricant compositions according to the present invention is in the range of from 35 to 95 wt %, preferably in the range of from 55 to 85 wt %, and even more preferably in the range of from 65 to 80 wt % based on the total weight of the lubricant composition.
[0073] The dicarboxylic ester preferably is derived from the reaction of a dicarboxylic acid with an aliphatic alcohol.
[0074] Preferred dicarboxylic acids are adipic acid, pimelic acid, suberic acid, azelaic acid and sebacic acid and mixtures thereof. The dicarboxylic acid ester component is preferably formed from such dicarboxylic acids by esterification with medium-size aliphatic alcohols, which can be linear or branched, preferably C.sub.5 to C.sub.20 alcohol, more preferably C.sub.9 to C.sub.15 aliphatic alcohol and most preferably nonanol, isodecanol, isotridecanol and 2-propyl heptanol.
[0075] Another preferred group of alcohols is derived from so-called Guerbet alcohols or mixtures thereof. The trivial name of Guerbet alcohol is used for 2-alkyl-substituted 1-alkanols whose industrial synthesis is described inter alia in H. Machemer, Angewandte Chemie, Vol. 64, pages 213-220 (1952) and in G. Dieckelmann and H. J. Heinz in "The Basics of Industrial Oleochemistry", pages 145-145 (1988). In one preferred embodiment the Guerbet alcohol is derived at least partly from 2-hexyl decanol, 2-hexyl dodecanol, 2-octyl decanol and/or 2-octyl dodecanol.
[0076] Alternatively, the dicarboxylic acid ester component can be a trimethylolpropane-type ester, preferably formed with C.sub.8-C.sub.10 aliphatic alcohol, e.g. trimethylolpropane caprylate (TMTC), which is commercially available as Synative ES TMTC.RTM. (from BASF SE).
[0077] An additionally preferred dicarboxylic ester component is diisotridecyl adipate (DITA). Such ester is for example commercially available under the trademark Synative ES DIDA.RTM. from BASF SE.
[0078] The dicarboxylic acid ester may include one of the following four particularly preferred embodiments.
[0079] In a first particularly preferred embodiment, the dicarboxylic ester component according to the present invention is obtainable by reacting a mixture comprising [0080] a) at least one acid selected from the group consisting of aliphatic dicarboxylic acids and aliphatic dicarboxylic acid anhydrides, [0081] b1) a monoalcohol having 10 carbon atoms and a structure of the general formula I,
[0081] ##STR00005## [0082] wherein [0083] R.sub.1 is selected from the group consisting of pentyl, iso-pentyl, 2-methyl-butyl, 3-methyl-butyl and 2,2-dimethyl-propyl, [0084] R.sub.2 is H or methyl, [0085] R.sub.3 is selected from the group consisting of ethyl, propyl and iso-propyl, and [0086] b2) a monoalcohol having 10 carbon atoms and a structure of the general formula II,
[0086] ##STR00006## [0087] wherein [0088] R.sub.4 is selected from the group consisting of pentyl, iso-pentyl, 2-methyl-butyl, 3-methyl-butyl and 2,2-dimethyl-propyl, [0089] R.sub.5 is H or methyl, [0090] R.sub.6 is selected from the group consisting of ethyl, propyl and iso-propyl, with the proviso that the monoalcohol b1) and the monoalcohol b2) have a different structure.
[0091] Preferably, the aliphatic dicarboxylic acid is selected from the group consisting of glutaric acid, diglycolic acid, succinic acid, azelaic acid, sebacic acid, 1,4-cyclohexanedicarboxylic acid, adipic acid, 2,6-decahydronaphthalenedicarboxylic acid, 1,3-cyclohexanedicarboxylic acid, and 2,5-norbornanedicarboxylic acid. More preferably, the aliphatic dicarboxylic acid is adipic acid.
[0092] The acids can be used either in pure form or in the form of mixtures with monocarboxylic acids. Instead of the acids, their anhydrides can also be used. Representative monocarboxylic acids include n-butanoic acid, n-pentanoic acid, n-hexanoic acid, n-heptanoic acid, n-octanoic acid, n-nonanoic acid, n-decanoic acid, isobutanoic acid, isopentanoic acid, isohexanoic acid, isoheptanoic acid, isooctanoic acid, 2-ethylhexanoic acid, isononanoic acid, 3,5,5-trimethylhexanoic acid, and isodecanoic acid.
[0093] Preferably the monoalcohol b1) is selected from the group consisting of 2-propylheptanol, 2-propyl-4-methyl-hexanol, 2-propyl-5-methyl-hexanol, 2-isopropyl-4-methyl-hexanol, 2-isopropyl-5-methyl-hexanol, 2-propyl-4,4-dimethylpentanol, 2-ethyl-2,4-dimethylhexanol, 2-ethyl-2-methyl-heptanol, 2-ethyl-2,5-dimethylhexanol and 2-isopropyl-heptanol. More preferably the monoalcohol b1) is 2-propyl-heptanol.
[0094] Preferably the monoalcohol b2) is selected from the group consisting of 2-propylheptanol, 2-propyl-4-methyl-hexanol, 2-propyl-5-methyl-hexanol, 2-isopropyl-4-methyl-hexanol, 2-isopropyl-5-methyl-hexanol, 2-propyl-4,4-dimethylpentanol, 2-ethyl-2,4-dimethylhexanol, 2-ethyl-2-methyl-heptanol, 2-ethyl-2,5-dimethylhexanol and 2-isopropyl-heptanol. More preferably the monoalcohol b2) is 2-propyl-4-methyl-hexanol.
[0095] Preferably the weight ratio of monoalcohol b1) to monoalcohol b2) is in the range of 5:1 to 95:1, more preferably in the range of 6:1 to 50:1, even more preferably in the range of 10:1 to 40:1, most preferably in the range of 20:1 to 35:1.
[0096] Preferably the mixture further comprises a monoalcohol b3) having 10 carbon atoms and a structure of the general formula III,
##STR00007##
wherein [0097] R.sub.7 is selected from the group consisting of pentyl, iso-pentyl, 2-methyl-butyl, 3-methyl-butyl and 2,2-dimethyl-propyl, [0098] R.sub.8 is H or methyl, [0099] R.sub.9 is selected from the group consisting of ethyl, propyl and iso-propyl, with the proviso that monoalcohol b3) has a different structure from both the monoalcohol b1) and the monoalcohol b2).
[0100] Preferably the monoalcohol b3) is selected from the group consisting of 2-propylheptanol, 2-propyl-4-methyl-hexanol, 2-propyl-5-methyl-hexanol, 2-isopropyl-4-methyl-hexanol, 2-isopropyl-5-methyl-hexanol, 2-propyl-4,4-dimethylpentanol, 2-ethyl-2,4-dimethylhexanol, 2-ethyl-2-methyl-heptanol, 2-ethyl-2,5-dimethylhexanol and 2-isopropyl-heptanol. More preferably the monoalcohol b3) is 2-propyl-5-methyl-hexanol.
[0101] Preferably the mixture comprises 80 to 95 weight-% of 2-n-propyl-heptanol as component b1), 1.0 to 10 weight. % of 2-propyl-4-methyl-hexanol as component b2), 1.0 to 10 weight-% of 2-propyl-5-methyl-hexanol as component b3) and 0.1 to 2.0 weight-% of 2-isopropyl-heptanol, whereby the weight of each component is related to the total weight of the monoalcohols. More preferably the mixture comprises 91.0 to 95.0 weight-% of 2-n-propyl-heptanol as component b1), 2.0 to 5.0 weight-% of 2-propyl-4-methyl-hexanol as component b2), 3.0 to 5.0 weight-% of 2-propyl-5-methyl-hexanol as component b3) and 0.1 to 0.8 weight-% of 2-isopropyl-heptanol, whereby the weight of each component is related to the total weight of the monoalcohols.
[0102] The dicarboxylic ester component according to the present invention also includes dicarboxylic acid esters which are obtained by reacting a mixture comprising adipic acid, 2-propyl-heptanol, 2-propyl-4-methyl-hexanol and 2-propyl-5-methyl-hexanol.
[0103] The dicarboxylic ester component according to the present invention further includes dicarboxylic acid esters which are obtained by reacting a mixture comprising adipic acid and 80 to 95 weight-% of 2-n-propyl-heptanol, 1.0 to 10 weight. % of 2-propyl-4-methyl-hexanol, 1.0 to 10 weight-% of 2-propyl-5-methyl-hexanol and 0.1 to 2.0 weight-% of 2-isopropyl-heptanol, whereby the weight of each component is related to the total weight of the monoalcohols.
[0104] The most preferred dicarboxylic acid ester according to the first particularly preferred embodiment is di-(2-propylheptyl)-adipate (DPHA). This compound also is commercially available as DPHA-XPB 115 (BASF SE).
[0105] In a second particularly preferred embodiment, the dicarboxylic ester component according to the present invention is diisononyl adipate (DNA).
[0106] The diisononyl adipate (DNA) is obtainable by reacting a mixture comprising adipic acid and an alcohol mixture comprising 1-nonanol, monomethyloctanols, dimethylheptanols and monoethylheptanols whereby the polyester has a viscosity at 40.degree. C. in the range of 5 to 15 mm.sup.2/s determined according to DIN 51562-1. The viscosity of the polyester at 40.degree. C. is preferably from 6 to 14 mm.sup.2/s, more preferably from 7 to 13 mm.sup.2/s, and most preferably from 8 to 12 mm.sup.2/s determined according to DIN 51562-1.
[0107] The polyesters prepared by reacting a mixture comprising adipic acid and an alcohol mixture comprising 1-nonanol, monomethyloctanols, dimethylheptanols and monoethylheptanols preferably have a density at 20.degree. C. according to DIN 51757 of from 0.85 to 1.00 g/cm.sup.3, more preferably from 0.88 to 0.95 g/cm.sup.3 and most preferably from 0.90 to 0.94 g/cm.sup.3. The refractive index n.sub.D.sup.20 according to DIN 51423 is preferably from 1.400 to 1.500, more preferably from 1.420 to 1.480, and most preferably from 1.440 to 1.460.
[0108] The alcohol mixture comprising 1-nonanol, monomethyloctanols, dimethylheptanols and monoethylheptanols is particularly advantageously obtainable in a process involving two or more stages and starting from a hydrocarbon mixture comprising butenes. In a first step, the butenes are dimerized to give a mixture of isomeric octenes. The octene mixture is then hydroformylated to give C.sub.9 aldehydes and then hydrogenated to give the alcohol mixture. In this reaction sequence, specific, defined parameters have to be adhered to, at least during the butene dimerization, preferably during the butene dimerization and the hydroformylation.
[0109] It is preferable, therefore, that the isomeric octenes mixture is obtained by bringing a hydrocarbon mixture comprising butenes into contact with a heterogeneous catalyst comprising nickel oxide. The isobutene content of the hydrocarbon mixture is preferably 5% by weight or less, in particular 3% by weight or less, particularly preferably 2% by weight or less, and most preferably 1.5% by weight or less, based in each case on the total butene content. A suitable hydrocarbon stream is that known as the C.sub.4 cut, a mixture of butenes and butanes, available in large quantities from FCC plants or from steam crackers. A starting material used with particular preference is that known as raffinate II, which is an isobutene-depleted C.sub.4 cut.
[0110] A preferred starting material comprises from 50 to 100% by weight, preferably from 80 to 95% by weight, of butenes and from 0 to 50% by weight, preferably from 5 to 20% by weight, of butanes. The following makeup of the butenes can be given as a general guide to quantities:
TABLE-US-00003 1-butene from 1 to 98% by weight, cis-2-butene from 1 to 50% by weight, trans-2-butene from 1 to 98% by weight, and isobutene up to 5% by weight.
[0111] Possible catalysts are catalysts known per se and comprising nickel oxide, as described, for example, by O'Connor et al. in Catalysis Today, 6, (1990) p. 329. Supported nickel oxide catalysts may be used, and possible support materials are silica, alumina, aluminosilicates, aluminosilicates having a layer structure and zeolites. Particularly suitable catalysts are precipitation catalysts obtainable by mixing aqueous solutions of nickel salts and of silicates, e.g. of sodium silicate and sodium nitrate, and, where appropriate, of other constituents, such as aluminum salts, e. g. aluminum nitrate, and calcining.
[0112] Particular preference is given to catalysts which essentially consist of NiO, SiO.sub.2, TiO.sub.2 and/or ZrO.sub.2, and also, where appropriate, Al.sub.2O.sub.3. A most preferred catalyst comprises, as significant active constituents, from 10 to 70% by weight of nickel oxide, from 5 to 30% by weight of titanium dioxide and/or zirconium dioxide and from 0 to 20% by weight of aluminum oxide, the remainder being silicon dioxide. A catalyst of this type is obtainable by precipitating the catalyst composition at pH from 5 to 9 by adding an aqueous solution comprising nickel nitrate to an aqueous alkali metal water glass solution which comprises titanium dioxide and/or zirconium dioxide, filtering, drying and annealing at from 350 to 650.degree. C. For details of preparation of these catalysts reference may be made to DE-A 4339713. The entire content of the disclosure of that publication is incorporated herein by way of reference.
[0113] The hydrocarbon mixture comprising butenes is brought into contact with the catalyst, preferably at temperatures of from 30 to 280.degree. C., in particular from 30 to 140.degree. C. and particularly preferably from 40 to 130.degree. C. This preferably takes place at a pressure of from 10 to 300 bar, in particular from 15 to 100 bar and particularly preferably from 20 to 80 bar. The pressure here is usefully set in such a way that the olefin-rich hydrocarbon mixture is liquid or in the supercritical state at the temperature selected.
[0114] Examples of reactors suitable for bringing the hydrocarbon mixture into contact with the heterogeneous catalyst are tube-bundle reactors and shaft furnaces. Shaft furnaces are preferred because the capital expenditure costs are lower. The dimerization may be carried out in a single reactor, where the oligomerization catalyst may have been arranged in one or more fixed beds.
[0115] Another way is to use a reactor cascade composed of two or more, preferably two, reactors arranged in series, where the butene dimerization in the reaction mixture is driven to only partial conversion on passing through the reactor(s) preceding the last reactor of the cascade, and the desired final conversion is not achieved until the reaction mixture passes through the last reactor of the cascade. The butene dimerization preferably takes place in an adiabatic reactor or in an adiabatic reactor cascade.
[0116] After leaving the reactor or, respectively, the last reactor of a cascade, the octenes formed and, where appropriate, higher oligomers, are separated off from the unconverted butenes and butanes in the reactor discharge. The oligomers formed may be purified in a subsequent vacuum fractionation step, giving a pure octene fraction. During the butene dimerization, small amounts of dodecenes are generally also obtained. These are preferably separated off from the octenes prior to the subsequent reaction.
[0117] In a preferred embodiment, some or all of the reactor discharge, freed from the oligomers formed and essentially consisting of unconverted butenes and butanes, is returned. It is preferable to select the return ratio such that the concentration of oligomers in the reaction mixture does not exceed 35% by weight, preferably 20% by weight, based on the hydrocarbon mixture of the reaction. This measure increases the selectivity of the butene dimerization in relation to those octenes which, after hydroformylation, hydrogenation and esterification, give a particularly preferred alcohol mixture.
[0118] The octenes obtained are converted, in the second process step, by hydroformylation using synthesis gas in a manner known per se, into aldehydes having one additional carbon atom. The hydroformylation of olefins to prepare aldehydes is known per se and is described, for example, in J. Falbe, (ed.): New Synthesis with Carbon monoxide, Springer, Berlin, 1980. The hydroformylation takes place in the presence of catalysts homogeneously dissolved in the reaction medium. The catalysts generally used here are compounds or complexes of metals of transition group VIII, specifically Co, Rh, Ir, Pd, Pt or Ru compounds, or complexes of these metals, either unmodified or modified, for example, using amine-containing or phosphine-containing compounds.
[0119] The hydroformylation preferably takes place in the presence of a cobalt catalyst, in particular dicobaltoctacarbonyl [CO.sub.2(CO).sub.8]. It preferably takes place at from 120 to 240.degree. C., in particular from 160 to 200.degree. C., and under a synthesis gas pressure of from 150 to 400 bar, in particular from 250 to 350 bar. The hydroformylation preferably takes place in the presence of water. The ratio of hydrogen to carbon monoxide in the synthesis gas mixture used is preferably in the range from 70:30 to 50:50, in particular from 65:35 to 55:45.
[0120] The cobalt-catalyzed hydroformylation process may be carried out as a multistage process which comprises the following 4 stages: the preparation of the catalyst (precarbonylation), the catalyst extraction, the olefin hydroformylation and the removal of the catalyst from the reaction product (decobaltization). In the first stage of the process, the precarbonylation, an aqueous cobalt salt solution, e.g. cobalt formate or cobalt acetate, as starting material is reacted with carbon monoxide and hydrogen to prepare the catalyst complex needed for the hydroformylation. In the second stage of the process, the catalyst extraction, the cobalt catalyst prepared in the first stage of the process is extracted from the aqueous phase using an organic phase, preferably using the olefin to be hydroformylated. Besides the olefin, it is occasionally advantageous to use the reaction products and byproducts of the hydroformylation for catalyst extraction, as long as these are insoluble in water and liquid under the reaction conditions selected. After the phase separation, the organic phase loaded with the cobalt catalyst is fed to the third stage of the process, the hydroformylation. In the fourth stage of the process, the decobaltization, the organic phase of the reactor discharge is freed from the cobalt carbonyl complexes in the presence of process water, which may comprise formic acid or acetic acid, by treatment with oxygen or air. During this, the cobalt catalyst is destroyed by oxidation and the resultant cobalt salts are extracted back into the aqueous phase. The aqueous cobalt salt solution obtained from the decobaltization is returned to the first stage of the process, the precarbonylation. The raw hydroformylation product obtained may be fed directly to the hydrogenation. Another way is to isolate a C.sub.9 fraction from this in a usual manner, e.g. by distillation, and feed this to the hydrogenation.
[0121] The formation of the cobalt catalyst, the extraction of the cobalt catalyst into the organic phase and the hydroformylation of the olefins can also be carried out in a single-stage process in the hydroformylation reactor.
[0122] Examples of cobalt compounds which can be used are cobalt(II) chloride, cobalt(II) nitrate, the amine complexes or hydrate complexes of these, cobalt carboxylates, such as cobalt formate, cobalt acetate, cobalt ethylhexanoate and cobalt naphthenate (Co salts of naphthenic acid), and also the cobalt caprolactamate complex. Under the conditions of the hydroformylation, the catalytically active cobalt compounds form in situ as cobalt carbonyls. It is also possible to use carbonyl complexes of cobalt such as dicobalt octacarbonyl, tetracobalt dodecacarbonyl and hexacobalt hexadecacarbonyl.
[0123] The aldehyde mixture obtained during the hydroformylation is reduced to give primary alcohols. A partial reduction generally takes place straight away under the conditions of the hydroformylation, and it is also possible to control the hydroformylation in such a way as to give essentially complete reduction. However, the hydroformylation product obtained is generally hydrogenated in a further process step using hydrogen gas or a hydrogen-containing gas mixture. The hydrogenation generally takes place in the presence of a heterogeneous hydrogenation catalyst. The hydrogenation catalyst used may comprise any desired catalyst suitable for hydrogenating aldehydes to give primary alcohols. Examples of suitable commercially available catalysts are copper chromite, cobalt, cobalt compounds, nickel, nickel compounds, which, where appropriate, comprise small amounts of chromium or of other promoters, and mixtures of copper, nickel and/or chromium. The nickel compounds are generally in a form supported on support materials, such as alumina or kieselgur. It is also possible to use catalysts comprising noble metals, such as platinum or palladium.
[0124] A suitable method of carrying out the hydrogenation is a trickle-flow method, where the mixture to be hydrogenated and the hydrogen gas or, respectively, the hydrogen-containing gas mixture are passed, for example concurrently, over a fixed bed of the hydrogenation catalyst.
[0125] The hydrogenation preferably takes place at from 50 to 250.degree. C., in particular from 100 to 150.degree. C., and at a hydrogen pressure of from 50 to 350 bar, in particular from 150 to 300 bar. The desired isononanol fraction in the reaction discharge obtained during the hydrogenation can be separated off by fractional distillation from the C.sub.8 hydrocarbons and higher-boiling products.
[0126] Gas-chromatographic analysis of the resultant alcohol mixture can give the relative amounts of the individual compounds (the percentages given being percentages by gas chromatogram area):
[0127] The proportion of 1-nonanol in the alcohol mixture is preferably from 6 to 16% by weight, more preferably from 8 to 14% by weight, related to the overall weight of the alcohol mixture.
[0128] The proportion of the monomethyloctanols is preferably from 25 to 55% by weight, more preferably from 35 to 55% by weight, and it is particularly preferable for 6-methyl-1-octanol and 4-methyl-1-octanol together to make up at least 25% by weight, very particularly preferably at least 35% by weight, related to the overall weight of the alcohol mixture.
[0129] The proportion of the dimethylheptanols and monoethylheptanols is preferably from 15 to 60% by weight, more preferably from 20 to 55% by weight, and it is preferable for 2,5-dimethyl-1-heptanol, 3-ethyl-1-heptanol and 4,5-dimethyl-1-heptanol together to make up at least 15% and in particular 20% by weight, related to the overall weight of the alcohol mixture. The proportion of the hexanols is preferably from 4 to 10% by weight and more preferably from 5 to 10% by weight, related to the overall weight of the alcohol mixture.
[0130] The alcohol mixture is preferably composed of from 70 to 100%, more preferably from 70 to 99%, most preferably from 80 to 98%, and even more preferably from 85 to 95%, of a mixture of 1-nonanol, monomethyloctanols, dimethylheptanols and monoethylheptanols, related to the overall weight of the alcohol mixture.
[0131] Preferably the alcohol mixture contains a proportion of 6% by weight to 16% by weight 1-nonanol, 25% by weight to 55% by weight monomethyloctanols, 10% by weight to 30% by weight dimethylheptanols and 7% by weight to 15% by weight monoethylheptanols, related to the overall weight of the alcohol mixture.
[0132] Preferably the alcohol mixture is present in a molar ratio in the range of 1:1 to 2:1, more preferably in a molar ratio in the range of 1:1 to 1.3:1, in relation to the adipic acid.
[0133] The density of the alcohol mixture of the invention at 20.degree. C. is preferably from 0.75 to 0.9 g/cm.sup.3, more preferably from 0.8 to 0.88 g/cm.sup.3, and most preferably from 0.82 to 0.84 g/cm.sup.3, according to DIN 51757. The refractive index n.sub.D.sup.20 is preferably from 1.425 to 1.445, more preferably from 1.43 to 1.44 and most preferably from 1.432 to 1.438. The boiling range at atmospheric pressure is preferably from 190 to 220.degree. C., more preferably from 195 to 215.degree. C. and most preferably from 200 to 210.degree. C.
[0134] In a third particularly preferred embodiment, the dicarboxylic ester component according to the present invention is a di-(2-ethylhexyl)-adipate (DEHA or DOA).
[0135] The di-(2-ethylhexyl)-adipate preferably has a dynamic viscosity at 20.degree. C. in the range of 12 to 16 mPas according to DIN 51562 as calculated from the measured kinematic viscosity and multiplication of the measured kinematic viscosity with the density. The di-(2-ethylhexyl)-adipate has a density at 20.degree. C. in the range of 0.920 to 0.930 g/cm.sup.3 determined according to DIN 51757 and a pourpoint <-50.degree. C. determined according to DIN ISO 3016 as lubricant.
[0136] The dynamic viscosity of di-(2-ethylhexyl)-adipate at 20.degree. C. is preferably from 13 to 15 mm.sup.2/s determined according to DIN 51562.
[0137] Preferably, di-(2-ethylhexyl)-adipate has a density in the range of 0.922 to 0.928 g/cm.sup.3, more preferably in the range of 0.924 to 0.926 g/cm.sup.3. The density is determined according to DIN 51757.
[0138] One preferred example for the di-(2-ethylhexyl)-adipate component is Plastomoll.RTM. DOA which is commercially available from BASF SE.
[0139] In a fourth particularly preferred embodiment, the dicarboxylic ester component according to the present invention is obtainable by reacting a mixture comprising [0140] a) phthalic acid, optionally in form of its esters or its anhydride, [0141] b1) a monoalcohol having 10 carbon atoms and a structure of the general formula I,
[0141] ##STR00008## [0142] wherein [0143] R.sub.1 is selected from the group consisting of pentyl, iso-pentyl, 2-methyl-butyl, 3-methyl-butyl and 2,2-dimethyl-propyl, [0144] R.sub.2 is H or methyl, [0145] R.sub.3 is selected from the group consisting of ethyl, propyl and iso-propyl, [0146] and [0147] b2) a monoalcohol having 10 carbon atoms and a structure of the general formula II,
[0147] ##STR00009## [0148] wherein [0149] R.sub.4 is selected from the group consisting of pentyl, iso-pentyl, 2-methyl-butyl, 3-methyl-butyl and 2,2-dimethyl-propyl, [0150] R.sub.5 is H or methyl, [0151] R.sub.6 is selected from the group consisting of ethyl, propyl and iso-propyl, with the proviso that the monoalcohol b1) and the monoalcohol b2) have a different structure as a lubricant.
[0152] The viscosity of the polyester at 20.degree. C. is preferably from 80 to 150 mm.sup.2/s, more preferably from 100 to 140 mm.sup.2/s and most preferably from 115 to 130 mm.sup.2/s determined according to DIN 51562-1.
[0153] The esters preferably have densities at 20.degree. C. according to DIN 51757 of from 0.90 to 1.00 g/cm.sup.3, more preferably from 0.95 to 0.98 g/cm.sup.3 and most preferably from 0.96 to 0.97 g/cm.sup.3. The refractive index n.sub.D.sup.20 according to DIN 51423 is preferably from 1,450 to 1,550, more preferably from 1,470 to 1,500, and most preferably from 1,480 to 1,485.
[0154] Preferably the ester is a methyl ester or ethyl ester.
[0155] Preferably the monoalcohol b1) is selected from the group consisting of 2-propylheptanol, 2-propyl-4-methyl-hexanol, 2-propyl-5-methyl-hexanol, 2-isopropyl-4-methyl-hexanol, 2-isopropyl-5-methyl-hexanol, 2-propyl-4,4-dimethylpentanol, 2-ethyl-2,4-dimethylhexanol, 2-ethyl-2-methyl-heptanol, 2-ethyl-2,5-dimethylhexanol and 2-isopropyl-heptanol. More preferably the monoalcohol b1) is 2-propyl-heptanol.
[0156] Preferably the monoalcohol b2) is selected from the group consisting of 2-propylheptanol, 2-propyl-4-methyl-hexanol, 2-propyl-5-methyl-hexanol, 2-isopropyl-4-methyl-hexanol, 2-isopropyl-5-methyl-hexanol, 2-propyl-4,4-dimethylpentanol, 2-ethyl-2,4-dimethylhexanol, 2-ethyl-2-methyl-heptanol, 2-ethyl-2,5-dimethylhexanol and 2-isopropyl-heptanol. More preferably the monoalcohol b2) is 2-propyl-4-methyl-hexanol.
[0157] Preferably the monoalcohols b1) and b2) are present in a molar ratio in the range of 1.05:1 to 2.0:1, more preferably in the range of 1.05:1 to 1.5:1, in relation to the phthalic acid a)
[0158] Preferably the weight ratio of monoalcohol b1) to monoalcohol b2) is in the range of 5:1 to 95:1, more preferably in the range of 6:1 to 50:1, even more preferably in the range of 10:1 to 40:1, most preferably in the range of 20:1 to 35:1.
[0159] Preferably the mixture further comprises a monoalcohol b3) having 10 carbon atoms and a structure of the general formula III,
##STR00010##
wherein [0160] R.sub.7 is selected from the group consisting of pentyl, iso-pentyl, 2-methyl-butyl, 3-methyl-butyl and 2,2-dimethyl-propyl, [0161] R.sub.8 is H or methyl, [0162] R.sub.9 is selected from the group consisting of ethyl, propyl and iso-propyl, with the proviso that monoalcohol b3) has a different structure from both the monoalcohol b1) and the monoalcohol b2).
[0163] Preferably the monoalcohol b3) is selected from the group consisting of 2-propylheptanol, 2-propyl-4-methyl-hexanol, 2-propyl-5-methyl-hexanol, 2-isopropyl-4-methyl-hexanol, 2-isopropyl-5-methyl-hexanol, 2-propyl-4,4-dimethylpentanol, 2-ethyl-2,4-dimethylhexanol, 2-ethyl-2-methyl-heptanol, 2-ethyl-2,5-dimethylhexanol and 2-isopropyl-heptanol. More preferably the monoalcohol b3) is 2-propyl-5-methyl-hexanol.
[0164] Preferably the mixture comprises 80 to 95 weight-% of 2-n-propyl-heptanol as component b1), 1.0 to 10 weight-% of 2-propyl-4-methyl-hexanol as component b2), 1.0 to 10 weight-% of 2-propyl-5-methyl-hexanol as component b3) and 0.1 to 2.0 weight-% of 2-isopropyl-heptanol, whereby the weight of each component is related to the total weight of the monoalcohols. More preferably the mixture comprises 91.0 to 95.0 weight-% of 2-n-propyl-heptanol as component b1), 2.0 to 5.0 weight-% of 2-propyl-4-methyl-hexanol as component b2), 3.0 to 5.0 weight-% of 2-propyl-5-methyl-hexanol as component b3) and 0.1 to 0.8 weight-% of 2-isopropyl-heptanol, whereby the weight of each component is related to the total weight of the monoalcohols.
[0165] In another aspect, the presently claimed invention is also directed to dicarboxylic acid esters which are obtained by reacting a mixture comprising phthalic acid, optionally in form of its esters or its anhydride, 2-propyl-heptanol, 2-propyl-4-methyl-hexanol and 2-propyl-5-methyl-hexanol. The most preferred dicarboxylic acid ester according to the fourth particularly preferred embodiment is di-(propylheptyl)-phthalate (DPHP). This compound is also commercially available as Palatinol.RTM. (BASF SE).
[0166] In another aspect, the dicarboxylic acid esters are obtained by reacting a mixture comprising phthalic acid, optionally in form of its esters or its anhydride, and 80 to 95 weight-% of 2-n-propyl-heptanol, 1.0 to 10 weight. % of 2-propyl-4-methyl-hexanol, 1.0 to 10 weight-% of 2-propyl-5-methyl-hexanol and 0.1 to 2.0 weight-% of 2-isopropyl-heptanol, whereby the weight of each component is related to the total weight of the monoalcohols.
[0167] In another particularly preferred embodiment of the present invention, the lubricant composition according to the present invention includes at least one of the four above particularly preferred dicarboxylic acid esters while further comprising an additional monocarboxylic acid ester component.
[0168] The additional monocarboxylic acid ester component is preferably obtained by reacting one or more monoalcohols with a monocarboxylic acid.
[0169] The monocarboxylic acids preferably contain at least 4 carbons, preferably C.sub.6 to C.sub.30, more preferably C.sub.8 to C.sub.20, acids such as saturated straight chain fatty acids including caprylic acid, capric acid, lauric acid, myristic acid, palmitic acid, stearic acid, arachic acid, and behenic acid, or the corresponding branched chain fatty acids or unsaturated fatty acids such as oleic acid, or mixtures thereof.
[0170] The monoalcohol preferably is 2-ethylhexanol, propylheptanol, or the like.
[0171] Other typical monocarboxylic acid ester components are represented by 2-ethylhexyl oleate, e.g. commercially available as Synative ES EHO.RTM. (BASF SE), 2-ethylhexyl cocoate, e.g. commercially available as Synative ES EHK.RTM. (BASF SE), 2-ethylhexyl palmitate, e.g. commercially available as Synative ES EHPA.RTM. (BASF SE), 2-ethylhexylstearate, e.g. commercially available as Synative ES EHS.RTM. (BASF SE), 2-ethylhexyl tallowate, e.g. commercially available as Synative ES EHTI.RTM. (BASF SE).
[0172] The amount of additional monocarboxylic acid ester in the lubricant compositions according to the present invention is in the range of from 0 to 25 wt %, preferably in the range of from 5 to 20 wt %, or even more preferably in the range of from 10 to 18 wt % based on the total weight of the lubricant composition.
[0173] In another particularly preferred embodiment of the present invention, the lubricant composition according to the present invention includes at least one of the four above particularly preferred dicarboxylic acid esters, optionally an additional monocarboxylic acid ester, while further comprising an additional complex carboxylic acid ester component.
[0174] A complex carboxylic acid ester according to the present invention is defined as an ester which is formed from polyols with dicarboxylic acids and/or monocarboxylic acids.
[0175] The complex monocarboxylic acid ester component is preferably obtained by reacting one or more polyhydric alcohols, preferably the hindered polyols such as the neopentyl polyols, e.g. neopentyl glycol, trimethylol ethane, 2-methyl-2-propyl-1,3-propanediol, trimethylol propane, trimethylol butane, pentaerythritol and dipentaerythritol with monocarboxylic acids containing at least 4 carbons, normally the C.sub.5 to C.sub.30 acids such as saturated straight chain fatty acids including caprylic acid, capric acid, lauric acid, myristic acid, palmitic acid, stearic acid, arachic acid, and behenic acid, or the corresponding branched chain fatty acids or unsaturated fatty acids such as oleic acid, or mixtures thereof, with polycarboxylic acids.
[0176] For example, a neopentyl glycol ester of at least one monocarboxylic acid having from 7 to 10 carbon atoms and of at least one other ester of a different hindered polyol with a monocarboxylic acid having from 5 to 10 carbon atoms is preferred. Other preferred polyols are trimethylolpropane, pentaerythritol, or dipentaerythritol.
[0177] Another typical complex carboxylic acid ester that is preferably used in the lubricant compositions according to the present invention is a complex carboxylic acid ester commercially available as Synative ES 3345.RTM. (BASF SE).
[0178] The amount of additional complex carboxylic acid ester in the lubricant compositions according to the present invention is in the range of from 0 to 20 wt %, preferably in the range of from 2 to 15 wt %, or even more preferably in the range of from 5 to 10 wt % based on the total weight of the lubricant composition.
[0179] In another particularly preferred embodiment of the present invention, the lubricant composition according to the present invention comprises at least one of the four above particularly preferred dicarboxylic acid esters, optionally an additional monocarboxylic acid ester and/or an additional complex carboxylic acid ester component, further comprising an additional Guerbet alcohol component.
[0180] The term Guerbet alcohols is used for 2-alkyl-substituted 1-alkanols whose industrial synthesis is described inter alia in H. Machemer, Angewandte Chemie, Vol. 64, pages 213-220 (1952) and in G. Dieckelmann and H. J. Heinz in "The Basics of Industrial Oleochemistry", pages 145-145 (1988). In one preferred embodiment the Guerbet alcohol is derived at least partly from 2-hexyl decanol, 2-hexyl dodecanol, 2-octyl decanol and/or 2-octyl dodecanol.
[0181] Particularly preferred Guerbet alcohols are 2-hexyldecyl alcohol, e.g. commercially available as Synative AL G 16.RTM. (BASF SE), or 2-octyldodecyl alcohol, e.g. commercially available as Synative AL G 20.RTM. (BASF SE).
[0182] The amount of additional Guerbet alcohol component in the lubricant compositions according to the present invention is in the range of from 0 to 20 wt %, preferably in the range of from 2 to 15 wt %, or even more preferably in the range of from 5 to 10 wt % based on the total weight of the lubricant composition.
[0183] The base oil (or base stock) to be used in the lubricant compositions according to the present invention is an optional component.
[0184] The base oil to be used in the lubricant compositions according to the present invention is an inert, solvent-type oil component in the lubricant compositions according to the present invention.
[0185] Preferably, the lubricant compositions according to the present invention further comprise base oils selected from the group consisting of mineral oils (Group I, II or III oils), polyalphaolefins (Group IV oils), polymerized and interpolymerized olefins, alkyl naphthalenes, alkylene oxide polymers, silicone oils and phosphate esters (Group V oils).
[0186] Definitions for the base oils according to the present invention are the same as those found in the American Petroleum Institute (API) publication "Engine Oil Licensing and Certification System", Industry Services Department, Fourteenth Edition, December 1996, Addendum 1, December 1998. Said publication categorizes base stocks as follows:
[0187] a) Group I base oils contain less than 90 percent saturates and/or greater than 0.03 percent sulfur and have a viscosity index greater than or equal to 80 and less than 120 using the test methods specified in the following table.
[0188] b) Group II base oils contain greater than or equal to 90 percent saturates and less than or equal to 0.03 percent sulfur and have a viscosity index greater than or equal to 80 and less than 120 using the test methods specified in the following table.
[0189] c) Group III base oils contain greater than or equal to 90 percent saturates and less than or equal to 0.03 percent sulfur and have a viscosity index greater than or equal to 120 using the test methods specified in the following table
[0190] Analytical Methods for Base Stock:
TABLE-US-00004 Property Test Method Saturates ASTM D 2007 Viscosity index ASTM D 2270 Sulfur ASTM D 2622 ASTM D 4294 ASTM D 4927 ASTM D 3120
[0191] d) Group IV base oils contain polyalphaolefins. Synthetic lower viscosity fluids suitable for the present invention include the polyalphaolefins (PAOs) and the synthetic oils from the hydrocracking or hydro-isomerization of Fischer Tropsch high boiling fractions including waxes. These are both base oils comprised of saturates with low impurity levels consistent with their synthetic origin. The hydro-isomerized Fischer Tropsch waxes are highly suitable base oils, comprising saturated components of iso-paraffinic character (resulting from the isomerization of the predominantly n-paraffins of the Fischer Tropsch waxes) which give a good blend of high viscosity index and low pour point. Processes for the hydro-isomerization of Fischer Tropsch waxes are described in U.S. Pat. Nos. 5,362,378; 5,565,086; 5,246,566 and 5,135,638, as well in EP 710710, EP 321302 and EP 321304.
[0192] Polyalphaolefins suitable for the lubricant compositions according to the present invention, include known PAO materials which typically comprise relatively low molecular weight hydrogenated polymers or oligomers of alphaolefins which include but are not limited to C.sub.2 to about C.sub.32 alphaolefins with the C.sub.8 to about C.sub.16 alphaolefins, such as 1-octene, 1-decene, 1-dodecene and the like being preferred. The preferred polyalphaolefins are poly-1-octene, poly-1-decene, and poly-1-dodecene, although the dimers of higher olefins in the range of C.sub.14 to C.sub.18 provide low viscosity base stocks.
[0193] Terms like PAO-2, PAO 4, PAO 6 or PAO 8 represent preferred polyalphaolefins while these terms are commonly used specifications for different classes of polyalphaolefins characterized by their respective viscosity. For instance, PAO 2 refers to a particularly preferred class of polyalphaolefins according to the present invention which typically has a viscosity in the range of 2 mm.sup.2/s at 100.degree. C. A variety of commercially available compositions are available for these specifications.
[0194] Low viscosity PAO fluids suitable for the lubricant compositions according to the present invention, may be conveniently made by the polymerization of an alphaolefin in the presence of a polymerization catalyst such as the Friedel-Crafts catalysts including, for example, aluminum trichloride, boron trifluoride or complexes of boron trifluoride with water, alcohols such as ethanol, propanol or butanol, carboxylic acids or esters such as ethyl acetate or ethyl propionate. For example, the methods disclosed by U.S. Pat. Nos. 3,149,178 or 3,382,291 may be conveniently used herein. Other descriptions of PAO synthesis are found in the following U.S. Pat. No. 3,742,082 (Brennan); U.S. Pat. No. 3,769,363 (Brennan); U.S. Pat. No. 3,876,720 (Heilman); U.S. Pat. No. 4,239,930 (Allphin); U.S. Pat. No. 4,367,352 (Watts); U.S. Pat. No. 4,413,156 (Watts); U.S. Pat. No. 4,434,308 (Larkin); U.S. Pat. No. 4,910,355 (Shubkin); U.S. Pat. No. 4,956,122 (Watts); and U.S. Pat. No. 5,068,487 (Theriot).
[0195] e) Group V base oils contain any base stocks not described by Groups I to IV. Examples of Group V base oils include alkyl naphthalenes, alkylene oxide polymers, silicone oils and phosphate esters.
[0196] Carboxylic acid esters which are widely considered in the literature to belong to the Group V base oils are not understood according to the present invention as base oils (base stocks) or even group V base oils but are separately classified or defined as the dicarboxylic acid ester or monocarboxylic acid ester component being either essential or at least optional to the present invention, respectively.
[0197] Synthetic base oils include hydrocarbon oils and halo-substituted hydrocarbon oils such as polymerized and interpolymerized olefins (e.g., polypropylenes, propylene-isobutylene copolymers, chlorinated polybutylenes, poly(1-hexenes), poly(1-octenes), poly(1-decenes)); alkylbenzenes (e.g., dodecylbenzenes, tetradecylbenzenes, dinonylbenzenes, di(2-ethylhexyl)benzenes); polyphenyls (e.g., biphenyls, terphenyls, alkylated polyphenols); and alkylated diphenyl ethers and alkylated diphenyl sulfides and derivative, analogs and homologs thereof.
[0198] Alkylene oxide polymers and interpolymers and derivatives thereof where the terminal hydroxyl groups have been modified by esterification, etherification, etc, constitute another class of known synthetic base oils. These are exemplified by polyoxyalkylene polymers prepared by polymerization of ethylene oxide or propylene oxide, and the alkyl and aryl ethers of polyoxyalkylene polymers (e.g., methyl-polyiso-propylene glycol ether having a molecular weight of 1000 or diphenyl ether of polyethylene glycol having a molecular weight of 1000 to 1500); and mono- and polycarboxylic esters thereof, for example, the acetic acid esters, mixed C.sub.3-C.sub.8 fatty acid esters and C.sub.13 Oxo acid diester of tetraethylene glycol.
[0199] Silicon-based oils such as the polyalkyl-, polyaryl-, polyalkoxy- or polyaryloxysilicone oils and silicate oils comprise another useful class of synthetic base oils; such base oils include tetraethyl silicate, tetraisopropyl silicate, tetra-(2- ethylhexyl)silicate, tetra-(4-methyl-2-ethylhexyl)silicate, tetra-(p-tert-butyl-phenyl) silicate, hexa-(4-methyl-2-ethylhexyl)disiloxane, oly(methyl)siloxanes and poly(methylphenyl)siloxanes. Other synthetic base oils include liquid esters of phosphorous-containing acids (e.g., tricresyl phosphate, trioctyl phosphate, diethyl ester of decylphosphonic acid) and polymeric tetrahydrofurans.
[0200] The base oil may also include so-called gas-to-liquid (GTL) base stocks. Suitable GTL base stocks that can be used in the present invention are for instance described in WO 2010/021751 A1 and are herewith incorporated by reference.
[0201] The base oil component has a kinematic viscosity according to DIN 51562-1 at 100.degree. C. in the range of from 1 to 8 mm.sup.2/s, preferably of from 1 to 5 mm.sup.2/s, or even more preferably of from 1 to 3 mm.sup.2/s. PAO-2 is the most preferred base oil to be used in the lubricant compositions according to the present invention.
[0202] The relative amount of base oil in the lubricant compositions according to the present invention is in the range of 0 to 50 wt %, preferably in the range of from 5 to 35, or even more preferably in the range of 10 to 25 wt % based on the total amount of lubricant composition.
[0203] Oligomeric copolymers are preferably ethylene-propylene copolymers having a number average molecular weight Mn according to industrial standard DIN 55672 within the range of about 20000 to about 150000 kg/mol, preferably of from about 40000 to about 120000 kg/mol, or even more preferably of from 60000 to 100000 kg/mol. Such oligomeric copolymers are for instance described in U.S. Pat. No. 5,451,630. Oligomeric copolymers are typically used in the art as viscosity modifying agents in lubricant compositions with improved shear stability.
[0204] The ethylene propylene copolymers according to the present invention have a kinematic viscosity according to JIS K 2283 at 100.degree. C. in the range of from 500 to 3000 mm.sup.2/s, preferably of from 900 to 2500 mm.sup.2/s, or even more preferably of from 1000 to 2200 mm.sup.2/s.
[0205] The ethylene propylene copolymers according to the present invention have a kinematic viscosity according to JIS K 2283 at 40.degree. C. in the range of from 10000 to 50000 mm.sup.2/s, preferably of from 15000 to 40000 mm.sup.2/s, or even more preferably of from 18000 to 38000 mm.sup.2/s.
[0206] The ethylene propylene copolymers according to the present invention have a pourpoint according to JIS K 2269 in the range of from -20.degree. C. to -5.degree. C., preferably of from -15.degree. C. to -10.degree. C., or even more preferably of from -12.5 to -10.degree. C.
[0207] Another preferred type of ethylene propylene copolymers is commercially available as LUCANT.TM. (Mitsui Chemicals), preferably LUCANT.TM.2000 and LUCANT.TM.1100.
[0208] The amount of oligomeric copolymer, preferably ethylene propylene copolymer, is in the range of about 5 to 30 wt %. preferably 10 to 25 wt %, or even more preferably 12 to 20 wt % based on the total weight of the lubricant composition.
[0209] The ratio of the dicarboxylic acid ester component to the oligomeric copolymers in the lubricant compositions according to the present invention is in the range of from 2:1 to 19:1, preferably 3:1 to 10:1, even more preferably in the range of from 4:1 to 6:1 based on the relative weight of these components in the lubricant compositions according to the present invention.
[0210] The lubricant composition according to the present invention comprising oligomeric copolymers as viscosity index improving agents can further comprise additional viscosity index improving agents. Viscosity index improving agents are thickener components that are able to increase the viscosity of a lubricant composition when added to it.
[0211] Additional suitable viscosity index improving agents typically include conventional polyisobutenes (PIBs) having no terminal double bonds, highly reactive polyisobutenes having terminal double bonds, polymethacrylates (PMAs) or the like.
[0212] However, in a preferred embodiment of the present invention, the just mentioned, additional viscosity index improving agents apart from oligomeric copolymers are absent from the lubricant compositions according to the present invention, either individually or in combination.
[0213] Accordingly, in a preferred embodiment of the present invention, conventional polyisobutenes (PlBs) without terminal double bonds and/or highly reactive polyisobutenes are absent from the lubricant compositions according to the present invention.
[0214] The term "conventional polyisobutenes" as used in the present application relates to polyisobutenes which do not have terminal double bonds. Conventional polyisobutenes therefore differ in the latter aspect from the highly reactive polyisobutenes which have high degree of terminal double bonds. One preferred conventional polyisobutene is Lubrizol 8406.RTM..
[0215] Highly reactive polyisobutene polymers are understood in their most generic manner in the context of the present invention as a polyisobutene polymer having at least 60 mol % terminal double bonds based on the total number of double bonds in the polymer. The amount of terminal double bonds in the highly reactive polyisobutenes can be determined following the method mentioned in U.S. Pat. No. 5,962,604 using .sup.13C-NMR spectroscopy based on the relative peak areas corresponding to the signals for the C-alpha and C-beta carbon atom (chemical shift of 114.4 ppm and 143.6 ppm), respectively.
[0216] In another preferred embodiment of the present invention, poly(meth)acrylates (PMAs) are absent from the lubricant compositions according to the present invention.
[0217] Poly(meth)acrylates (PMAs) are esters of (meth)acrylic acid that are able to provide improved shear stability in lubricant compositions. Such poly(meth)acrylates are for instance described in DE 3544061. Typical PMAs used in the art are those from the commercial Viscoplex.RTM. series of additives. Preferred PMAs are alkylmethacrylate (Viscoplex 0-101), alkylmaleate-alpha-olefin copolymer I (Gear-Lube 7930), alkylfumarate-alpha-olefin-copolymer I (Gear-Lube 7960) and the like.
[0218] The lubricant compositions according to the present invention may also comprise an additive component.
[0219] The additive component as used in the present invention may include an additive package and/or performance additives.
[0220] The additive package as used in the present invention as well as the compounds relating to performance additives are considered mixtures of additives that are typically used in lubricant compositions in limited amounts for mechanically, physically or chemically stabilizing the lubricant compositions while special performance characteristics can be further established by the individual or combined presence of such selected additives.
[0221] Additive packages are separately defined in the present invention since a variety of such additive packages are commercially available and typically used in lubricant compositions. One such preferred additive package that is commercially available is marketed under the name Anglamol6004J.RTM..
[0222] However, the individual components contained in the additive packages and/or the compounds further defined in the present invention as so-called performance additives include a larger number of different types of additives including dispersants, metal deactivators, detergents, extreme pressure agents (typically boron- and/or sulfur- and/or phosphorus-containing), antiwear agents, antioxidants (such as hindered phenols, aminic antioxidants or molybdenum compounds), corrosion inhibitors, foam inhibitors, demulsifiers, pour point depressants, seal swelling agents, friction modifiers and mixtures thereof.
[0223] The additive component as the sum of all additives contained in the lubricant compositions according to the present invention also including all additives contained in an additive package or added separately is present in the lubricant compositions of the present invention in an amount of 0 to 20 wt %, preferably 0.1 to 15 wt %, more preferably 2 to 12 wt %, and most preferably in an amount of 3 to 10 wt %.
[0224] Extreme pressure agents include compounds containing boron and/or sulfur and/or phosphorus. The extreme pressure agent may be present in the lubricant compositions at 0% by weight to 20% by weight, or 0.05% by weight to 10% by weight, or 0.1% by weight to 8% by weight of the lubricant composition.
[0225] In one embodiment according to the present invention, the extreme pressure agent is a sulfur-containing compound. In one embodiment, the sulfur-containing compound may be a sulfurised olefin, a polysulfide, or mixtures thereof. Examples of the sulfurised olefin include a sulfurised olefin derived from propylene, isobutylene, pentene; an organic sulfide and/or polysulfide including benzyldisulfide; bis-(chlorobenzyl) disulfide; dibutyl tetrasulfide; di-tertiary butyl polysulfide; and sulfurised methyl ester of oleic acid, a sulfurised alkylphenol, a sulfurised dipentene, a sulfurised terpene, a sulfurised Diels-Alder adduct, an alkyl sulphenyl N'N- dialkyl dithiocarbamates; or mixtures thereof.
[0226] In one embodiment the sulfurised olefin includes a sulfurised olefin derived from propylene, isobutylene, pentene or mixtures thereof.
[0227] In one embodiment according to the present invention, the extreme pressure agent sulfur-containing compound includes a dimercaptothiadiazole or derivative, or mixtures thereof. Examples of the dimercaptothiadiazole include compounds such as 2,5-dimercapto-1,3,4-thiadiazole or a hydrocarbyl-substituted 2,5-dimercapto-1,3,4-thiadiazole, or oligomers thereof. The oligomers of hydrocarbyl-substituted 2,5-dimercapto-1,3,4-thiadiazole typically form by forming a sulfur-sulfur bond between 2,5-dimercapto-1,3,4-thiadiazole units to form derivatives or oligomers of two or more of said thiadiazole units. Suitable 2,5-dimercapto-1,3,4-thiadiazole derived compounds include for example 2,5-bis(tert-nonyldithio)-1,3,4-thiadiazole or 2-tert-nonyldithio-5-mercapto-1,3,4-thiadiazole. The number of carbon atoms on the hydrocarbyl substituents of the hydrocarbyl-substituted 2,5-dimercapto-1,3,4-thiadiazole typically include 1 to 30, or 2 to 20, or 3 to 16.
[0228] In one embodiment, the dimercaptothiadiazole may be a thiadiazole-functionalised dispersant. A detailed description of the thiadiazole-functionalised dispersant is described is paragraphs [0028] to [0052] of International Publication WO 2008/014315.
[0229] The thiadiazole-functionalised dispersant may be prepared by a method including heating, reacting or complexing a thiadiazole compound with a dispersant substrate. The thiadiazole compound may be covalently bonded, salted, complexed or otherwise solubilised with a dispersant, or mixtures thereof.
[0230] The relative amounts of the dispersant substrate and the thiadiazole used to prepare the thiadiazole-functionalised dispersant may vary. In one embodiment the thiadiazole compound is present at 0.1 to 10 parts by weight relative to 100 parts by weight of the dispersant substrate. In different embodiments the thiadiazole compound is present at greater than 0.1 to 9, or greater than 0.1 to less than 5, or 0.2 to less than 5: to 100 parts by weight of the dispersant substrate. The relative amounts of the thiadiazole compound to the dispersant substrate may also be expressed as (0.1-10):100, or (>0.1-9):100, (such as (>0.5-9):100), or (0.1 to less than 5): 100, or (0.2 to less than 5): 100.
[0231] In one embodiment the dispersant substrate is present at 0.1 to 10 parts by weight relative to 1 part by weight of the thiadiazole compound. In different embodiments the dispersant substrate is present at greater than 0.1 to 9, or greater than 0.1 to less than 5, or about 0.2 to less than 5: to 1 part by weight of the thiadiazole compound. The relative amounts of the dispersant substrate to the thiadiazole compound may also be expressed as (0.1-10):1, or (>0.1-9):1, (such as (>0.5-9):1), or (0.1 to less than 5): 1, or (0.2 to less than 5): 1.
[0232] The thiadiazole-functionalised dispersant may be derived from a substrate that includes a succinimide dispersant (for example, N-substituted long chain alkenyl succinimides, typically a polyisobutylene succinimide), a Mannich dispersant, an ester-containing dispersant, a condensation product of a fatty hydrocarbyl monocarboxylic acylating agent with an amine or ammonia, an alkyl amino phenol dispersant, a hydrocarbyl-amine dispersant, a polyether dispersant, a polyetheramine dispersant, a viscosity modifier containing dispersant functionality (for example polymeric viscosity index modifiers containing dispersant functionality), or mixtures thereof. In one embodiment the dispersant substrate includes a succinimide dispersant, an ester-containing dispersant or a Mannich dispersant.
[0233] In one embodiment according to the present invention, the extreme pressure agent includes a boron-containing compound. The boron-containing compound includes a borate ester (which in some embodiments may also be referred to as a borated epoxide), a borated alcohol, a borated dispersant, a borated phospholipid or mixtures thereof. In one embodiment the boron-containing compound may be a borate ester or a borated alcohol.
[0234] The borate ester may be prepared by the reaction of a boron compound and at least one compound selected from epoxy compounds, halohydrin compounds, epihalohydrin compounds, alcohols and mixtures thereof. The alcohols include dihydric alcohols, trihydric alcohols or higher alcohols, with the proviso for one embodiment that hydroxyl groups are on adjacent carbon atoms, i.e., vicinal.
[0235] Boron compounds suitable for preparing the borate ester include the various forms selected from the group consisting of boric acid (including metaboric acid, orthoboric acid and tetraboric acid), boric oxide, boron trioxide and alkyl borates. The borate ester may also be prepared from boron halides.
[0236] In one embodiment suitable borate ester compounds include tripropyl borate, tributyl borate, tripentyl borate, trihexyl borate, triheptyl borate, trioctyl borate, trinonyl borate and tridecyl borate. In one embodiment the borate ester compounds include tributyl borate, tri-2-ethylhexyl borate or mixtures thereof.
[0237] In one embodiment, the boron-containing compound is a borated dispersant, typically derived from an N-substituted long chain alkenyl succinimide. In one embodiment the borated dispersant includes a polyisobutylene succinimide. Borated dispersants are described in more detail in U.S. Pat. Nos. 3,087,936; and 3,254,025.
[0238] In one embodiment the borated dispersant may be used in combination with a sulfur-containing compound or a borate ester.
[0239] In one embodiment the extreme pressure agent is other than a borated dispersant.
[0240] The number average molecular weight Mn (GPC; kg/mol) of the hydrocarbon from which the long chain alkenyl group was derived includes ranges of 350 to 5000, or 500 to 3000, or 550 to 1500. The long chain alkenyl group may have a number average molecular weight Mn of 550, or 750, or 950 to 1000.
[0241] The N-substituted long chain alkenyl succinimides are borated using a variety of agents including boric acid (for example, metaboric acid, orthoboric acid and tetraboric acid), boric oxide, boron trioxide, and alkyl borates. In one embodiment the borating agent is boric acid which may be used alone or in combination with other borating agents.
[0242] The borated dispersant may be prepared by blending the boron compound and the N-substituted long chain alkenyl succinimides and heating them at a suitable temperature, such as, 80.degree. C. to 250.degree. C., or 90.degree. C. to 230.degree. C., or 100.degree. C. to 210.degree. C., until the desired reaction has occurred. The molar ratio of the boron compounds to the N-substituted long chain alkenyl succinimides may have ranges including 10:1 to 1:4, or 4:1 to 1:3; or the molar ratio of the boron compounds to the N-substituted long chain alkenyl succinimides may be 1:2. Alternatively, the ratio of moles B:moles N (that is, atoms of B:atoms of N) in the borated dispersant may be 0.25:1 to 10:1 or 0.33:1 to 4:1 or 0.2:1 to 1.5:1, or 0.25:1 to 1.3:1 or 0.8:1 to 1.2:1 or about 0.5:1 An inert liquid may be used in performing the reaction. The liquid may include toluene, xylene, chlorobenzene, dimethylformamide or mixtures thereof.
[0243] In one embodiment, the additive component in the lubricant composition according to the present invention further includes a borated phospholipid. The borated phospholipid may be derived from boronation of a phospholipid (for example boronation may be carried out with boric acid). Phospholipids and lecithins are described in detail in Encyclopedia of Chemical Technology, Kirk and Othmer, 3rd Edition, in "Fats and Fatty Oils", Volume 9, pages 795-831 and in "Lecithins", Volume 14, pages 250-269.
[0244] The phospholipid may be any lipid containing a phosphoric acid, such as lecithin or cephalin, or derivatives thereof. Examples of phospholipids include phosphatidylcholine, phosphatidylserine, phosphatidylinositol, phosphatidyl-ethanolamine, phosphotidic acid and mixtures thereof. The phospholipids may be glycerophospholipids, glycerol derivatives of the above list of phospholipids. Typically, the glycerophospholipids have one or two acyl, alkyl or alkenyl groups on a glycerol residue. The alkyl or alkenyl groups may contain 8 to 30, or 8 to 25, or 12 to 24 carbon atoms. Examples of suitable alkyl or alkenyl groups include octyl, dodecyl, hexadecyl, octadecyl, docosanyl, octenyl, dodecenyl, hexadecenyl and octadecenyl.
[0245] Phospholipids may be prepared synthetically or derived from natural sources. Synthetic phospholipids may be prepared by methods known to those in the art. Naturally derived phospholipids are often extracted by procedures known to those in the art. Phospholipids may be derived from animal or vegetable sources. A useful phospholipid is derived from sunflower seeds. The phospholipid typically contains 35% to 60% phosphatidylcholine, 20% to 35% phosphatidylinositol, 1% to 25% phosphatidic acid, and 10% to 25% phosphatidylethanolamine, wherein the percentages are by weight based on the total phospholipids. The fatty acid content may be 20% by weight to 30% by weight palmitic acid, 2% by weight to 10% by weight stearic acid, 15% by weight to 25% by weight oleic acid, and 40% by weight to 55% by weight linoleic acid.
[0246] In another embodiment, the performance additive in the lubricant compositions according to the present invention may include a friction modifier. A friction modifier is any material or materials that can alter the coefficient of friction of a surface lubricated by any lubricant or fluid containing such material(s). Friction modifiers, also known as friction reducers, or lubricity agents or oiliness agents, and other such agents that change the ability of base oils, formulated lubricant compositions, or functional fluids, to modify the coefficient of friction of a lubricated surface may be effectively used in combination with the base oils or lubricant compositions of the present invention if desired. Friction modifiers may include metal-containing compounds or materials as well as ashless compounds or materials, or mixtures thereof. Metal-containing friction modifiers may include metal salts or metal-ligand complexes where the metals may include alkali, alkaline earth, or transition group metals. Such metal-containing friction modifiers may also have low-ash characteristics. Transition metals may include Mo, Sb, Sn, Fe, Cu, Zn, and others. Ligands may include hydrocarbyl derivative of alcohols, polyols, glycerols, partial ester glycerols, thiols, carboxylates, carbamates, thiocarbamates, dithiocarbamates, phosphates, thiophosphates, dithiophosphates, amides, imides, amines, thiazoles, thiadiazoles, dithiazoles, diazoles, triazoles, and other polar molecular functional groups containing effective amounts of O, N, S, or P, individually or in combination. In particular, Mo-containing compounds can be particularly effective such as for example Mo-dithiocarbamates, Mo(DTC), Mo-dithiophosphates, Mo(DTP), Mo-amines, Mo (Am), Mo-alcoholates, Mo-alcohol-amides, and the like.
[0247] Ashless friction modifiers may also include lubricant materials that contain effective amounts of polar groups, for example, hydroxyl-containing hydrocarbyl base oils, glycerides, partial glycerides, glyceride derivatives, and the like. Polar groups in friction modifiers may include hydrocarbyl groups containing effective amounts of O, N, S, or P, individually or in combination. Other friction modifiers that may be particularly effective include, for example, salts (both ash-containing and ashless derivatives) of fatty acids, fatty alcohols, fatty amides, fatty esters, hydroxyl-containing carboxylates, and comparable synthetic long-chain hydrocarbyl acids, alcohols, amides, esters, hydroxy carboxylates, and the like. In some instances fatty organic acids, fatty amines, and sulfurized fatty acids may be used as suitable friction modifiers.
[0248] In one embodiment, the performance additive in the lubricant compositions according to the present invention may include phosphorus- or sulfur-containing anti-wear agents other than compounds described as an extreme pressure agent of the amine salt of a phosphoric acid ester described above. Examples of the anti-wear agent may include a non-ionic phosphorus compound (typically compounds having phosphorus atoms with an oxidation state of +3 or +5), a metal dialkyldithiophosphate (typically zinc dialkyldithiophosphates), amine dithiophosphate, ashless dithiophosphates and a metal mono- or di-alkylphosphate (typically zinc phosphates), or mixtures thereof.
[0249] The non-ionic phosphorus compound includes a phosphite ester, a phosphate ester, or mixtures thereof.
[0250] In one embodiment, the performance additive in the lubricant composition according to the present invention may further include at least one antioxidant. Antioxidants retard the oxidative degradation of base stocks during service. Such degradation may result in deposits on metal surfaces, the presence of sludge, or a viscosity increase in the lubricant. One skilled in the art knows a wide variety of oxidation inhibitors that are useful in lubricating oil compositions.
[0251] Useful antioxidants include hindered phenols. These phenolic antioxidants may be ashless (metal-free) phenolic compounds or neutral or basic metal salts of certain phenolic compounds. Typical phenolic antioxidant compounds are the hindered phenolics which are the ones which contain a sterically hindered hydroxyl group, and these include those derivatives of dihydroxy aryl compounds in which the hydroxyl groups are in the o- or p-position to each other. Typical phenolic antioxidants include the hindered phenols substituted with C.sub.6+ alkyl groups and the alkylene coupled derivatives of these hindered phenols. Examples of phenolic materials of this type 2-t-butyl-4-heptyl phenol; 2-t-butyl-4-octyl phenol; 2-t-butyl-4-dodecyl phenol; 2,6-di-t-butyl-4-heptyl phenol; 2,6-di-t-butyl-4-dodecyl phenol; 2-methyl-6-t-butyl-4-heptyl phenol; and 2-methyl-6-t-butyl-4-dodecyl phenol. Other useful hindered mono-phenolic antioxidants may include for example hindered 2,6-di-alkyl-phenolic propionic ester derivatives. Bis-phenolic antioxidants may also be advantageously used in combination with the instant invention. Examples of ortho-coupled phenols include: 2,2'-bis(4-heptyl-6-t-butyl-phenol); 2,2'-bis(4- octyl-6-t-butyl-phenol); and 2,2'-bis(4-dodecyl-6-t-butyl-phenol). Para-coupled bisphenols include for example 4,4'-bis(2,6-di-t-butyl phenol) and 4,4'- methylene-bis(2,6-di-t-butyl phenol).
[0252] Non-phenolic oxidation inhibitors which may be used include aromatic amine antioxidants and these may be used either as such or in combination with phenolics. Typical examples of non-phenolic antioxidants include: alkylated and non-alkylated aromatic amines such as aromatic monoamines of the formula R.sup.8R.sup.9R.sup.10N, where R.sup.8 is an aliphatic, aromatic or substituted aromatic group, R.sup.9 is an aromatic or a substituted aromatic group, and R.sup.10 is H, alkyl, aryl or R.sup.11S(O).sub.xR.sup.12, where R.sup.11 is an alkylene, alkenylene, or aralkylene group, R.sup.12 is a higher alkyl group, or an alkenyl, aryl, or alkaryl group, and x is 0, 1 or 2. The aliphatic group R.sup.8 may contain from 1 to about 20 carbon atoms, and preferably contains from about 6 to 12 carbon atoms. The aliphatic group is a saturated aliphatic group. Preferably, both R.sup.8 and R.sup.9 are aromatic or substituted aromatic groups, and the aromatic group may be a fused ring aromatic group such as naphthyl. Aromatic groups R.sup.8 and R.sup.9 may be joined together with other groups such as S.
[0253] Typical aromatic amines antioxidants have alkyl substituent groups of at least about 6 carbon atoms. Examples of aliphatic groups include hexyl, heptyl, octyl, nonyl, and decyl. Generally, the aliphatic groups will not contain more than about 14 carbon atoms. The general types of amine antioxidants useful in the present compositions include diphenylamines, phenyl naphthylamines, phenothiazines, imidodibenzyls and diphenyl phenylene diamines. Mixtures of two or more aromatic amines are also useful. Polymeric amine antioxidants can also be used. Particular examples of aromatic amine antioxidants useful in the present invention include: p,p'-dioctyldiphenylamine; t-octylphenyl-alpha-naphthylamine; phenyl-alphanaphthylamine; and p-octylphenyl-alpha-naphthylamine. Sulfurized alkyl phenols and alkali or alkaline earth metal salts thereof also are useful antioxidants.
[0254] In one embodiment, the performance additive in the lubricant compositions according to the present invention further includes a dispersant. The dispersant may be a succinimide dispersant (for example N-substituted long chain alkenyl succinimides), a Mannich dispersant, an ester-containing dispersant, a condensation product of a fatty hydrocarbyl monocarboxylic acylating agent with an amine or ammonia, an alkyl amino phenol dispersant, a hydrocarbylamine dispersant, a polyether dispersant or a polyetheramine dispersant.
[0255] In one embodiment the succinimide dispersant includes a polyisobutylene-substituted succinimide, wherein the polyisobutylene from which the dispersant is derived may have a number average molecular weight of 400 to 5000, or 950 to 1600. Succinimide dispersants and their methods of preparation are more fully described in U.S. Pat. Nos. 4,234,435 and 3,172,892. Suitable ester-containing dispersants are typically high molecular weight esters. These materials are described in more detail in U.S. Pat. No. 3,381,022.
[0256] In one embodiment the dispersant includes a borated dispersant. Typically the borated dispersant includes a succinimide dispersant including a polyisobutylene succinimide, wherein the polyisobutylene from which the dispersant is derived may have a number average molecular weight of 400 to 5000. Borated dispersants are described in more detail above within the extreme pressure agent description.
[0257] Dispersant viscosity modifiers (often referred to as DVMs) are considered additives in the context of the present invention due to their additional functionalisation and are therefore not considered viscosity improving agents according to the present invention. Dispersant viscosity modifiers include functionalised polyolefins, for example, ethylene-propylene copolymers that have been functionalised with the reaction product of maleic anhydride and an amine, a polymethacrylate functionalised with an amine, or esterified styrene-maleic anhydride copolymers reacted with an amine.
[0258] As another type of performance additives, corrosion inhibitors can be described as any materials (additives, functionalised fluids, etc.) that form a protective film on a surface that prevents corrosion agents from reacting or attacking that surface with a resulting loss of surface material. Protective films may be absorbed on the surface or chemically bonded to the surface. Protective films may be constituted from mono-molecular species, oligomeric species, polymeric species, or mixtures thereof. Protective films may derive from the intact corrosion inhibitors, from their combination products, or their degradation products, or mixtures thereof. Surfaces that may benefit from the action of corrosion inhibitors may include metals and their alloys (both ferrous and non-ferrous types) and non-metals.
[0259] Corrosion inhibitors may include various oxygen-, nitrogen-, sulfur-, and phosphorus-containing materials, and may include metal-containing compounds (salts, organometallics, etc.) and nonmetal-containing or ashless materials. Corrosion inhibitors may include, but are not limited to, additive types such as, for example, hydrocarbyl-, aryl-, alkyl-, arylalkyl-, and alkylaryl-versions of detergents (neutral, overbased), sulfonates, phenates, salicylates, alcoholates, carboxylates, salixarates, phosphites, phosphates, thiophosphates, amines, amine salts, amine phosphoric acid salts, amine sulfonic acid salts, alkoxylated amines, etheramines, polyetheramines, amides, imides, azoles, diazoles, triazoles, benzotriazoles, benzothiadoles, mercaptobenzothiazoles, tolyltriazoles (TTZ-type), heterocyclic amines, heterocyclic sulfides, thiazoles, thiadiazoles, mercaptothiadiazoles, dimercaptothiadiazoles (DMTD-type), imidazoles, benzimidazoles, dithiobenzimidazoles, imidazolines, oxazolines, Mannich reactions products, glycidyl ethers, anhydrides, carbamates, thiocarbamates, dithiocarbamates, polyglycols, etc., or mixtures thereof.
[0260] Corrosion inhibitors are used to reduce the degradation of metallic parts that are in contact with the lubricant composition. Suitable corrosion inhibitors include thiadiazoles. Aromatic triazoles, such as tolyltriazole, are suitable corrosion inhibitors for non-ferrous metals, such as copper.
[0261] Metal deactivators include derivatives of benzotriazoles (typically tolyltriazole), 1,2,4-triazoles, benzimidazoles, 2-alkyldithiobenzimidazoles, thiadiazoles or 2-alkyldithiobenzothiazoles.
[0262] Foam inhibitors may also advantageously be added as a performance additive to the lubricant compositions according to the present invention. These agents retard the formation of stable foams. Silicones and organic polymers are typical foam inhibitors. For example, polysiloxanes, such as silicon oil, or polydimethylsiloxane, provide foam inhibiting properties. Further foam inhibitors include copolymers of ethyl acrylate and 2-ethylhexyl acrylate and optionally vinyl acetate.
[0263] Demulsifiers include trialkyl phosphates, and various polymers and copolymers of ethylene glycol, ethylene oxide, propylene oxide, or mixtures thereof.
[0264] As pour point depressants, esters of maleic anhydride-styrene, or polyacrylamides are included.
[0265] As a further performance additive to be used in the lubricant compositions according to the present invention, seal compatibility agents help to swell elastomeric seals by causing a chemical reaction in the fluid or physical change in the elastomer. Suitable seal compatibility agents for lubricant compositions include organic phosphates, aromatic esters, aromatic hydrocarbons, esters (butylbenzyl phthalate, for example), and polybutenyl succinic anhydride. Such additives may preferably be used in an amount of 0.01 to 3% by weight, more preferably 0.01 to 2% by weight of the total amount of the lubricant composition.
[0266] The present invention provides lubricant compositions which have excellent low temperature viscosity and very good rheological properties including shear stability over a broad temperature range. Particularly, the lubricant compositions according to the present invention have high permanent shear stability. The lubricant compositions according to the present invention also have very good oxidation stability.
[0267] One preferred lubricant composition according to the present invention as further illustrated by inventive example 1 (IE-1) is as follows:
TABLE-US-00005 Lubricant composition Dicarboxylic acid ester component having kinematic 70 to 95 wt % viscosity according to DIN 51562-1 at 100.degree. C. in the range of from 2 to 5 mm.sup.2/s Ethylene-propylene copolymer having kinematic 5 to 30 wt % viscosity according to JIS K 2283 at 100.degree. C. in the range of from 1900 to 2100 mm.sup.2/s
[0268] Another preferred lubricant composition according to the present invention is defined as follows:
TABLE-US-00006 Lubricant composition Di-(2-propylheptyl)adipate (DPHA) 70 to 95 wt % Ethylene-propylene copolymer having kinematic 5 to 30 wt % viscosity according to JIS K 2283 at 100.degree. C. in the range of from 1900 to 2100 mm.sup.2/s
[0269] Another preferred lubricant composition according to the present invention as further illustrated by inventive example 2 (IE-2) is defined as follows:
TABLE-US-00007 Lubricant composition Dicarboxylic acid ester component having dynamic 60-80 wt % viscosity according to DIN 51562-1 at 100.degree. C. in the range of from 12 to 16 mm.sup.2/s Ethylene-propylene copolymer having kinematic 10 to 25 wt % viscosity according to JIS K 2283 at 100.degree. C. in the range of from 1000 to 1200 mm.sup.2/s Monocarboxylic acid ester having kinematic viscosity 10 to 25 wt % according to DIN 51562-1 at 100.degree. C. in the range of from 2 to 4 mm.sup.2/s
[0270] Another preferred lubricant composition according to the present invention as further illustrated by inventive examples 3 (IE-3) and 4 (IE-4) is defined as follows:
TABLE-US-00008 Lubricant composition Dicarboxylic acid ester component having dynamic 55-75 wt % viscosity according to DIN 51562-1 at 100.degree. C. in the range of from 12 to 16 mm.sup.2/s Ethylene-propylene copolymer having kinematic viscosity 5 to 20 wt % according to JIS K 2283 at 100.degree. C. in the range of from 1000 to 1200 mm.sup.2/s Monocarboxylic acid ester having kinematic viscosity 5 to 20 wt % according to DIN 51562-1 at 100.degree. C. in the range of from 2 to 4 mm.sup.2/s Polyalphaolefin 2 5 to 20 wt %
[0271] Another preferred lubricant composition according to the present invention as further illustrated by inventive example 5 (IE-5) is defined as follows:
TABLE-US-00009 Lubricant composition Dicarboxylic acid ester component having dynamic 55-75 wt % viscosity according to DIN 51562-1 at 100.degree. C. in the range of from 12 to 16 mm.sup.2/s Ethylene-propylene copolymer having kinematic 5 to 20 wt % viscosity according to JIS K 2283 at 100.degree. C. in the range of from 1000 to 1200 mm.sup.2/s Monocarboxylic acid ester having kinematic viscosity 3 to 15 wt % according to DIN 51562-1 at 100.degree. C. in the range of from 2 to 4 mm.sup.2/s Polyalphaolefin 2 10 to 25 wt % Complex carboxylic acid ester having kinematic 3 to 15 wt % viscosity according to DIN 51562-1 at 100.degree. C. in the range of from 10 to 20 mm.sup.2/s
[0272] The lubricant compositions according to the present invention can be used in a variety of different applications. Preferred embodiments include the use of the lubricant compositions according to the present invention in light, medium and heavy duty engine oils, industrial engine oils, marine engine oils, automotive engine oils, crankshaft oils, compressor oils, refrigerator oils, hydrocarbon compressor oils, very low-temperature lubricating oils and fats, high temperature lubricating oils and fats, wire rope lubricants, textile machine oils, refrigerator oils, aviation and aerospace lubricants, aviation turbine oils, transmission oils, gas turbine oils, spindle oils, spin oils, traction fluids, transmission oils, plastic transmission oils, passenger car transmission oils, truck transmission oils, industrial transmission oils, industrial gear oils, insulating oils, instrument oils, brake fluids, transmission liquids, shock absorber oils, heat distribution medium oils, transformer oils, fats, chain oils, minimum quantity lubricants for metalworking operations, oil to the warm and cold working, oil for water-based metalworking liquids, oil for neat oil metalworking fluids, oil for semi-synthetic metalworking fluids, oil for synthetic metalworking fluids, drilling detergents for the soil exploration, hydraulic oils, in biodegradable lubricants or lubricating greases or waxes, chain saw oils, release agents, moulding fluids, gun, pistol and rifle lubricants or watch lubricants and food grade approved lubricants.
EXAMPLES
[0273] Methods
[0274] Measurement of the number average molecular weight Mn of polymers mentioned in the present invention has been carried out using the industrial standard DIN 55672.
[0275] The pour point of the lubricant compositions according to the present invention has been determined according to the established industrial standard DIN ISO 3016 (if not indicated otherwise).
[0276] The various kinematic viscosities of the lubricant compositions according to the present invention have been determined following established industry standards.
[0277] The kinematic viscosity at -30.degree. C., 40.degree. C. and 100.degree. C., respectively, is determined according to the established industrial standard DIN 51562-1 (unless indicated otherwise).
[0278] The dynamic viscosity is determined based on DIN 51562-1 and further calculated by multiplication of the measured kinematic viscosity with the corresponding density.
[0279] The viscosity index has been determined according to the industrial standard DIN ISO 2909 (if not indicated otherwise).
[0280] Shear stability testing has been carried out according to the industrial standard DIN 51350/KRL/C.
[0281] Preparation of Carboxylic Acid Esters
[0282] Preparation of a Diisononyl Adipate (DNA)
[0283] a) Butene Dimerization
[0284] The butene dimerization was carried out continuously in an adiabatic reactor, composed of two subreactors (length: in each case 4 m, diameter: in each case 80 cm) with intermediate cooling at 30 bar. The starting product used was a raffinate II with the following makeup:
TABLE-US-00010 isobutane 2% by weight n-butane 10% by weight isobutene 2% by weight 1-butene 32% by weight trans-2-butene 37% by weight and cis-2-butene 17% by weight.
[0285] The catalyst used was a material prepared in accordance with DE-A 4339713, composed of 50% by weight of NiO, 12.5% by weight of TiO.sub.2, 33.5% by weight of SiO.sub.2 and 4% by weight of Al.sub.2O.sub.3, in the form of 5.times.5 mm tablets. The reaction was carried out with a throughput of 0.375 kg of raffinate II per I of catalyst and hour, with a return ratio of unreacted C.sub.4 hydrocarbons returned to fresh raffinate II of 3, an inlet temperature at the 1st subreactor of 38.degree. C. and an inlet temperature at the 2nd subreactor of 60.degree. C. The conversion, based on the butenes present in the raffinate II, was 83.1%, and the octene selectivity was 83.3%. Fractional distillation of the reactor discharge was used to separate off the octene fraction from unreacted raffinate II and from the high-boilers.
[0286] b) Hydroformylation and Hydrogenation
[0287] 750 g of the octene mixture prepared according to section A.1 of the examples were reacted for 5 hours discontinuously, in an autoclave, with 0.13% by weight of dicobalt octacarbonyl Co.sub.2(CO).sub.8 as catalyst, with addition of 75 g of water, at 185.degree. C. and with a synthesis gas pressure of 280 bar at a ratio of H.sub.2 to CO in the mixture of 60/40. Further material was injected to make up for the consumption of synthesis gas, seen in a fall-off of pressure in the autoclave. After releasing the pressure in the autoclave, the reaction discharge, with 10% strength by weight acetic acid, was freed oxidatively from the cobalt catalyst by introducing air, and the organic product phase was hydrogenated using Raney nickel at 125.degree. C. and with a hydrogen pressure of 280 bar for 10 h. The isononanol fraction was separated off from the C.sub.8 paraffins and the highboilers by fractional distillation of the reaction discharge.
[0288] The composition of the isononanol fraction was analyzed by gas chromatography. A specimen was trimethylsilylated in advance using 1 ml of N-methyl-N-trimethylsilyltrifluoracetamide per 100 .mu.l of specimen for 60 minutes at 80.degree. C. Use was made of a Hewlett Packard Ultra 1 separating column of length 50 m and internal diameter of 0.32 mm, with a film thickness of 0.2 .mu.m. Injector temperature and detector temperature were 250.degree. C., and the oven temperature was 120.degree. C. The split was 110 ml/min. The carrier gas used was nitrogen. The admission pressure was set at 200 kPa. 1 .mu.l of the specimen was injected and detected by FID. The compositions determined for specimens by this method (percentage by gas chromatogram area) were as follows:
TABLE-US-00011 11.0% 1-nonanol 20.8% 6-methyl-1-octanol 20.5% 4-methyl-1-octanol 5.3% 2-methyl-1-octanol 11.0% 2,5-dimethyl-1-heptanol 8.7% 3-ethyl-1-heptanol 6.2% 4,5-dimethyl-1-heptanol 2.9% 2-ethyl-1-heptanol 2.8% 2,3-dimethyl-1-heptanol 3.0% 2-ethyl-4-methyl-1-hexanol 2.7% 2-propyl-1-hexanol 1.6% 3-ethyl-4-methyl-1-hexanol
[0289] The density of this isononanol mixture was measured at 20.degree. C. as 0.8326, and the refractive index n.sub.D.sup.20 as 1.4353. The boiling range at atmospheric pressure was from 204 to 209.degree. C.
[0290] c) Esterification
[0291] 865.74 g of the isononanol fraction obtained in process step 2 (20% molar excess based on adipic acid) were reacted with 365.25 g of adipic acid and 0.42 g of isopropyl butyl titanate catalyst in a 2 I autoclave into which nitrogen was bubbled (10 l/h) with a stirrer speed of 500 rpm and a reaction temperature of 230.degree. C. The water formed in the reaction was removed progressively from the reaction mixture with the nitrogen stream. The reaction time was 180 min. The nonanol excess was then distilled off at a reduced pressure of 50 mbar. 1000 g of the crude diisononyl adipate were neutralized by stirring for 10 minutes at 80.degree. C. with 150 ml of 0.5% strength aqueous sodium hydroxide. This gave a two-phase mixture with an upper organic phase and a lower aqueous phase (waste liquor with hydrolyzed catalyst). The aqueous phase was separated off, and the organic phase subjected to two further washings with 200 ml of H.sub.2O. For further purification, the neutralized and washed diisononyl adipate was stripped using steam at 180.degree. C. and a reduced pressure of 50 mbar for two hours. The purified diisononyl adipate was then dried for 30 min at 150.degree. C./50 mbar by passing a nitrogen stream (2 l/h) through the material, then mixed with activated carbon for 5 min and filtered off with suction via a suction filter using Supra-Theorit 5 filtration aid (temperature 80.degree. C.).
[0292] The resultant diisononyl adipate has a density of 0.920 g/cm.sup.3 and a refractive index n.sub.D.sup.20 of 1.4500.
[0293] Preparation of di-(2-propylheptyl)-phthalate (DPHP)
[0294] Propylheptanol is commercially available from BASF SE, Ludwigshafen [93.0% by weight 2-propyl-heptanol; 2.9% by weight 2-propyl-4-methyl-hexanol; 3.9% by weight 2-propyl-5-methylhexanol and 0.2 Gew.-% 2-isopropylheptanol].
[0295] A mixture of structural isomers of an alcohol with 10 carbon atoms which is available by BASF SE as "propylheptanol" (2.4 mol) and phthalic acid (1.0 mol) is reacted in the present of isopropyl-butyl-titanate (0.001 mol) in an autoclave under inert gas (N.sub.2) at a reaction temperature of 230.degree. C. Water which is formed during the reaction is removed from the reaction mixture through an inert gas stream (N.sub.2-stream). After 180 minutes the excess alcohol is removed from the mixture by distillation at a pressure of 50 mbar. The thus obtained ester is then neutralized with 0.5% NaOH at 80.degree. C. Afterwards the organic phase and the aqueous phase are separated, followed by washing the organic phase two times with water. In a further step the organic phase is purified by treating the crude ester with steam at 180.degree. C. and 50 mbar. Then the ester is dried by subjecting it to a N.sub.2 stream at 150.degree. C. and 50 mbar. Finally the ester is mixed with activated carbon and is filtered using as a rheological agent supra-theorit at 80.degree. C. under reduced pressure. The ester shows a density of 0.962 at 20.degree. C., measured according to DIN 51757, respectively ASTM D 4052.
[0296] Experimental Tests
Test Example 1
[0297] Shear Stability (KRH Test)
TABLE-US-00012 1E-1 Dicarboxylic ester component DPHA XPB 115 .RTM. 90 wt % Ethylene-propylene copolymer Lucant .TM.HC 2000 10 wt % 1E-1 KRH testing Kinematic Viscosity After 20 h After 100 h at -30.degree. C. [mm.sup.2/s] 1540 1711 1880 at 40.degree. C. [mm.sup.2/s] 37.54 36.78 34.44 at 100.degree. C. [mm.sup.2/s] 7.96 7.79 7.32 Viscosity index 192 190 186 Pour point [.degree. C.] -72 -72 -72
[0298] DPHA XPB 115.RTM. (BASF SE) is a di-propylheptyl adipate (a dicarboxylic acid ester component) having a pourpoint according to ASTM D97 of <-75.degree. C. and a kinematic viscosity according to ASTM D445 at 40.degree. C. of 11.56 mm.sup.2/s, at 100.degree. C. of 3.04 mm.sup.2/s and a viscosity index according to ASTM D2270 of 123;
[0299] Lucant.TM.HC-2000 is an oligomeric ethylene-propylene copolymer having pour point according to JIS K 2269 of -10.0.degree. C., kinematic viscosity according to JIS K 2283 at 40.degree. C. of 37500 mm.sup.2/s and at 100.degree. C. of 2000 mm.sup.2/s and viscosity index according to JIS K 2283 of 300.
Test Example 2
[0300] Rheological Profile of Lubricant Compositions at Different Temperatures
TABLE-US-00013 IE-2 1E-3 1E-4 1E-5 Dicarboxylic ester Plastomoll .RTM. DOA Plastomoll .RTM. DOA Plastomoll .RTM. DOA Plastomoll .RTM. DOA component 70.00 wt % 68.00 wt % 63.50 wt % 63.00 wt % Ethylene- Lucant .TM.HC 1100 Lucant .TM.HC 1100 Lucant .TM.HC 1100 Lucant .TM.HC 1100 propylene copolymer 12.00 wt % 12.00 wt % 12.50 wt % 12.00 wt % Monoester component Synative ES EHO Synative ES EHO Synative ES EHO Synative ES EHO 18.00 wt % 8.00 wt % 9.00 wt % 7.00 wt % Base oil PAO-2 PAO-2 PAO-2 12.00 wt % 15.00 wt % 13.00 wt % Complex ester Synative ES 3345 5.00 wt % Kinematic Viscosity at -30.degree. C. [mm.sup.2/s] 903 1006 1535 1200 at 40.degree. C. [mm.sup.2/s] 37.67 31.90 32.7 33.76 at 100.degree. C. [mm.sup.2/s] 8.73 7.62 7.81 7.90 Viscosity index 222 221 222 219 Pour point [.degree. C.] n.d. -72 n.d. n.d.
[0301] Plastomoll.RTM. DOA (BASF SE) is a di-(2-ethylhexyl)adipate (a dicarboxylic acid ester component) having a pourpoint according to DIN ISO 3016 of <-60.degree. C. and a dynamic viscosity according to DIN 51562 at 20.degree. C. of 13-15 mPas;
[0302] Lucant.TM.HC-1100 is an oligomeric ethylene-propylene copolymer having pour point according to JIS K 2269 of -12.5.degree. C., kinematic viscosity according to JIS K 2283 at 40.degree. C. of 18900 mm.sup.2/s and at 100.degree. C. of 1100 mm.sup.2/s and viscosity index according to JIS K 2283 of 270;
[0303] Synative ES EHO.RTM. (BASF SE) is 2-ethylhexyloleate (a monocarboxylic acid ester) having a pourpoint according to DIN ISO 3016 of not higher than -30.degree. C.;
[0304] Synative ES 3345.RTM. (BASF SE) is a complex carboxylic acid ester component having a pourpoint according to DIN ISO 3016 of not higher than -40.degree. C., a kinematic viscosity according to DIN 51562.1 at 40.degree. C. in the range of 105 to 120 mm.sup.2/s, at 100.degree. C. in the range of 13 to 18 mm.sup.2/s and a viscosity index according to DIN ISO 2090 in the range of from 140 to 160 mm.sup.2/s.
* * * * *
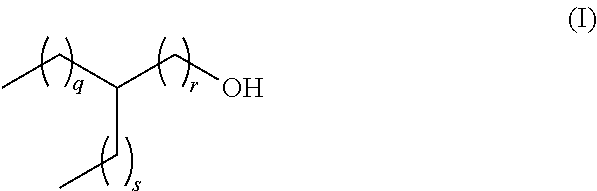
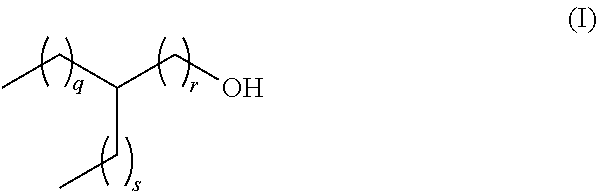
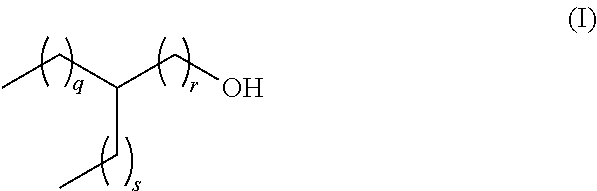
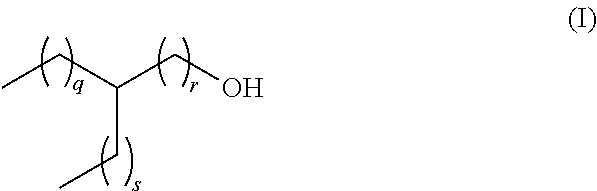

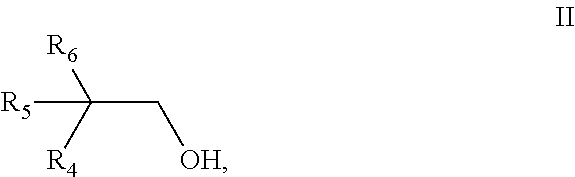
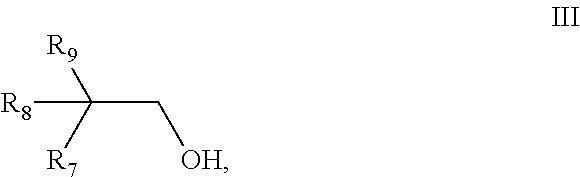
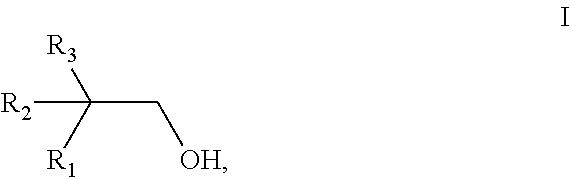
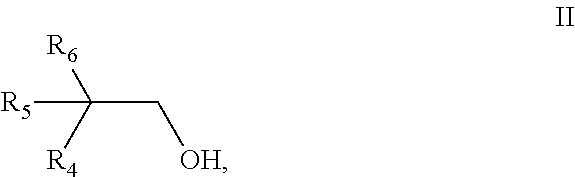
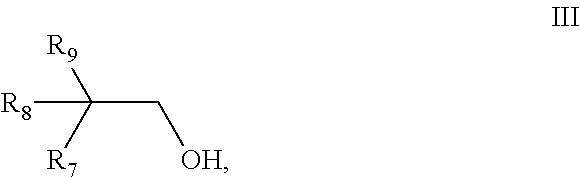
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.