Water-based Metal Working Oil Agent
KAWASAKI; Hiroshi ; et al.
U.S. patent application number 14/769174 was filed with the patent office on 2015-12-31 for water-based metal working oil agent. This patent application is currently assigned to IDEMITSU KOSAN CO., LTD.. The applicant listed for this patent is IDEMITSU KOSAN CO., LTD.. Invention is credited to Yosuke JIBIKI, Hiroshi KAWASAKI, Fumiaki TAKAGI.
| Application Number | 20150376536 14/769174 |
| Document ID | / |
| Family ID | 51391285 |
| Filed Date | 2015-12-31 |
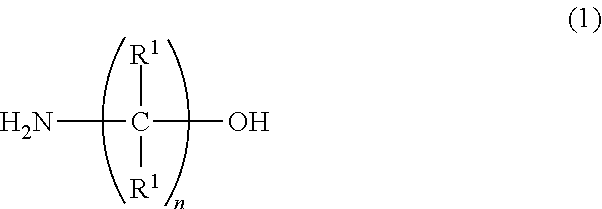


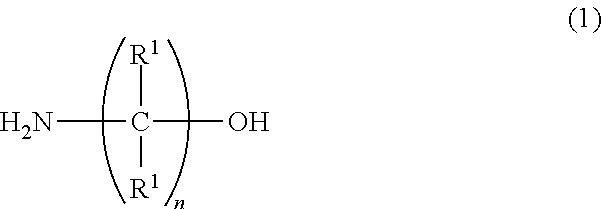

| United States Patent Application | 20150376536 |
| Kind Code | A1 |
| KAWASAKI; Hiroshi ; et al. | December 31, 2015 |
WATER-BASED METAL WORKING OIL AGENT
Abstract
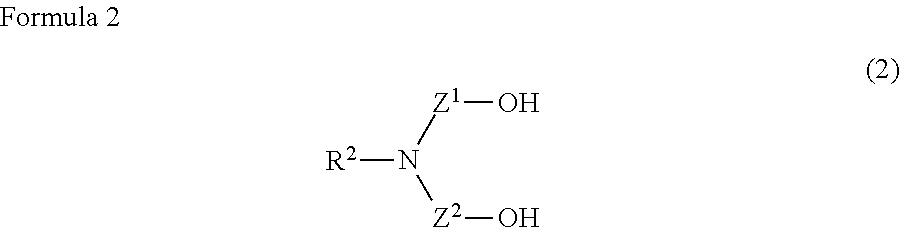
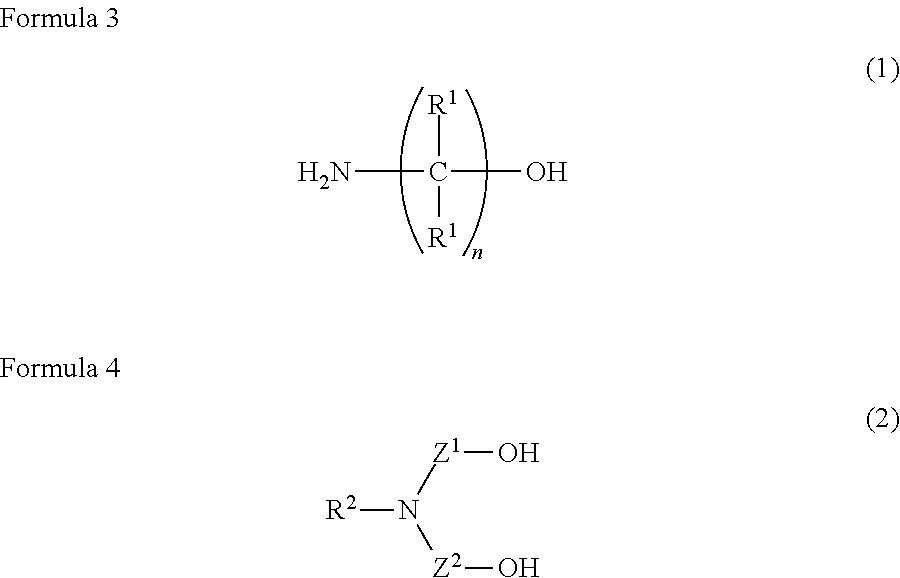
A water-based metalworking fluid contains a component (A) of alkanolamine represented by a formula (1) below as an amine component and a component (B) of alkanolamine represented by a formula (2) below as an amine component, a molar ratio of an amine component having a molecular weight of 90 or less to a total of the amine components (the amine component having a molecular weight of 90 or less/the total of the amine components) is 0.67 or less. ##STR00001## In the formula, R.sup.1 represents hydrogen or an alkyl group having 1 to 3 carbon atoms; n is 1 to 3; and R.sup.1 may be mutually the same or different. ##STR00002## In the formula: R.sup.2 represents an alkyl group having 1 to 10 carbon atoms; and Z.sup.1 and Z.sup.2 each independently represent an alkylene group having 2 to 8 carbon atoms.
| Inventors: | KAWASAKI; Hiroshi; (Ichihara-shi, JP) ; TAKAGI; Fumiaki; (Ichihara-shi, JP) ; JIBIKI; Yosuke; (Ichihara-shi, JP) | ||||||||||
| Applicant: |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assignee: | IDEMITSU KOSAN CO., LTD. Chiyoda-ku JP |
||||||||||
| Family ID: | 51391285 | ||||||||||
| Appl. No.: | 14/769174 | ||||||||||
| Filed: | February 19, 2014 | ||||||||||
| PCT Filed: | February 19, 2014 | ||||||||||
| PCT NO: | PCT/JP2014/053917 | ||||||||||
| 371 Date: | August 20, 2015 |
| Current U.S. Class: | 508/511 ; 508/562 |
| Current CPC Class: | C10M 173/02 20130101; C10M 2229/02 20130101; C10N 2040/22 20130101; C10M 2219/09 20130101; C10M 2219/104 20130101; C10N 2030/64 20200501; C10N 2030/12 20130101; C10M 2217/041 20130101; C10N 2040/24 20130101; C10M 133/08 20130101; C10M 2215/042 20130101; C10M 2215/042 20130101; C10M 2215/042 20130101 |
| International Class: | C10M 133/08 20060101 C10M133/08 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Feb 20, 2013 | JP | 2013-031026 |
Claims
1. A water-based metalworking fluid comprising: a component (A) of alkanolamine represented by formula (1) below as a first amine component; and a component (B) of alkanolamine represented by formula (2) below as a second amine component, wherein the water-based metalworking fluid has a molar ratio of an amine component having a molecular weight of 90 or less to a total of amine components of 0.67 or less. ##STR00006## where each R.sup.1 represents hydrogen or an alkyl group having 1 to 3 carbon atoms; n is 1, 2 or 3; and the R.sup.1's are mutually the same or different, ##STR00007## where R.sup.2 represents an alkyl group having 1 to 10 carbon atoms; and Z.sup.1 and Z.sup.2 each independently represent an alkylene group having 2 to 8 carbon atoms.
2. The water-based metalworking fluid according to claim 1, wherein n is 2 or 3.
3. The water-based metalworking fluid according to claim 1, wherein the component (A) comprises 1-amino-2-propanol.
4. The water-based metalworking fluid according to claim 1, wherein one or both of Z.sup.1 and Z.sup.2 are each an alkylene group having 2 carbon atoms.
5. The water-based metalworking fluid according to claim 1, wherein the component (B) comprises N-methyldiethanolamine and cyclohexyldiethanolamine.
6. The water-based metalworking fluid according to claim 1, further comprising an aliphatic carboxylic acid.
7. The water-based metalworking fluid according to claim 1, being diluted with water, wherein the water has an amount that is 200 or less times of the amount of the water-based metalworking fluid by mass.
Description
TECHNICAL FIELD
[0001] The present invention relates to a water-based metalworking fluid used in metalworking such as cutting or grinding.
BACKGROUND ART
[0002] A metalworking fluid used in metalworking is categorized into an oil-type (oil-based) fluid and a water-type (water-based) fluid, the latter of which is more frequently used because such a water-based fluid is excellent in cooling capabilities and infiltration capabilities and free from a risk of causing a fire. Such a water-based metalworking fluid, which is to be diluted with water in use, is required to have rust resistance. As such a rust resistant component, it is generally known to use alkanolamine such as monoethanolamine, diethanolamine and triethanolamine (see Patent Literature 1).
CITATION LIST
Patent Literature(s)
[0003] Patent Literature 1: JP-A-11-209774
SUMMARY OF THE INVENTION
Problem(s) to be Solved by the Invention
[0004] The water-based metalworking fluid generally exhibits more favorable rust resistance at a larger content of a rust resistant component. However, when an amine-based rust resistant component (e.g., alkanolamine) is contained, odor is derived from amines to occasionally have an adverse influence on human bodies and working environments. Accordingly, a type and a content of the rust resistant component still need to be studied.
[0005] An object of the present invention is to provide a water-based metalworking fluid that exhibits an excellent rust resistance and causes less odor to be more environmentally friendly and less harmful to human bodies.
Means for Solving the Problem(s)
[0006] In order to solve the above problems, the invention provides a water-based metalworking fluid as follows.
(1) According to an aspect of the invention, a water-based metalworking fluid contains a component (A) of alkanolamine represented by a formula (1) below as an amine component and a component (B) of alkanolamine represented by a formula (2) below as an amine component, a molar ratio of an amine component having a molecular weight of 90 or less to a total of the amine components (the amine component having a molecular weight of 90 or less/the total of the amine components) is 0.67 or less.
##STR00003##
[0007] In the formula, R' represents hydrogen or an alkyl group having 1 to 3 carbon atoms; n is 1 to 3; and R' may be mutually the same or different.
##STR00004##
[0008] In the formula: R.sup.2 represents an alkyl group having 1 to 10 carbon atoms; and Z.sup.1 and Z.sup.2 each independently represent an alkylene group having 2 to 8 carbon atoms.
(2) In the water-based metalworking fluid in the above aspect of the invention, n in the formula (1) representing the component (A) of alkanolamine is 2 or 3. (3) In the water-based metalworking fluid in the above aspect of the invention, the component (A) of alkanolamine comprises 1-amino-2-propanol. (4) In the water-based metalworking fluid in the above aspect of the invention, one or both of Z.sup.1 and Z.sup.2 in the formula (2) representing the component (B) of alkanolamine are an alkylene group(s) having 2 carbon atoms. (5) In the water-based metalworking fluid in the above aspect of the invention, the component (B) of alkanolamine comprises N-methyldiethanolamine and cyclohexyldiethanolamine. (6) The water-based metalworking fluid in the above aspect of the invention further contains an aliphatic carboxylic acid. (7) A water-based metalworking fluid according to another aspect of the invention is prepared by diluting the water-based metalworking fluid according to the above aspect of the invention with water, an amount of the water being 200 or less times as much as an amount of the water-based metalworking fluid according to the above aspect of the invention by a mass ratio.
[0009] According to the above aspect of the invention, since alkanolamine having a specific structure is contained as the amine component and the molar ratio of the amine component having a molecular weight of 90 or less is defined as being equal to or less than a specific ratio, the water-based metalworking fluid that exhibits an excellent rust resistance and causes less odor to be more environmentally friendly and less harmful to human bodies can be provided.
DESCRIPTION OF EMBODIMENT(S)
[0010] Exemplary embodiment(s) of the invention will be described below.
[0011] In an exemplary embodiment, a water-based metalworking fluid contains a component (A) of alkanolamine represented by a formula (1) below and a component (B) of alkanolamine represented by a formula (2) as amine components, a molar ratio of an amine component having a molecular weight of 90 or less to a total of amine components (i.e., an amine component having a molecular weight of 90 or less/a total of amine components) is 0.67 or less.
##STR00005##
[0012] First of all, the component (A) will be described. The component (A) is used for particularly providing rust resistance to the water-based metalworking fluid. In the above formula (1), R.sup.1 represents hydrogen or an alkyl group having 1 to 3 carbon atoms. n is 1 to 3. R.sup.1 may be mutually the same or different. When n is 4 or more, solubility of the component (A) is unfavorably reduced. n is more preferably 2 or 3, most preferably 2. In addition, when any one of R' has 4 or more carbon atoms, the solubility of the component (A) and rust resistance for iron are unfavorably deteriorated.
[0013] An amine compound having a molecular weight of 90 or less is preferably included in the component (A) of alkanolamine in order to improve rust resistance.
[0014] Examples of the component (A) are 1-amino-2-propanol, 2-amino-2-methyl-1-propanol, 1-amino-2-butanol, 2-amino-1-propanol, and 3-amino-2-butanol. Among the above, in view of the rust resistance for iron, 1-amino-2-propanol and 2-amino-2-methyl-1-propanol are preferable.
[0015] In the exemplary embodiment, the component (A) may be provided by a single one or a plurality of the above substances.
[0016] Next, alkanolamine as the component (B) will be described. The component (B) contributes to improving rust resistance and reducing odor.
[0017] In the formula (2), which represents the component (B), R.sup.2 represents an alkyl group having 1 to 10 carbon atoms. When R.sup.2 has a non-cyclic structure, R.sup.2 preferably has 1 to 4 carbon atoms, more preferably 1 carbon atom. When R.sup.2 has 11 or more carbon atoms, solubility and rust resistance of the component (B) are unfavorably deteriorated. Z.sup.1 and Z.sup.2 each independently represent an alkylene group having 2 to 8 carbon atoms. One or both of Z' and Z.sup.2 are preferably an alkylene group having 2 carbon atoms. When at least one of Z' and Z.sup.2 has 1 carbon atom, the component (B) is degraded to generate formaldehyde, which is environmentally unfavorable. When at least one of Z' and Z.sup.2 has 9 or more carbon atoms, the solubility of the component (B) is unfavorably deteriorated.
[0018] Examples of the component (B) are N-methyldiethanolamine, N-ethyldiethanolamine, cyclohexyldiethanolamine, N-n-propyldiethanolamine, N-i-propyldiethanolamine, N-n-butyldiethanolamine, N-i-butyldiethanolamine, and N-t-butyldiethanolamine. Among the above, it is particularly preferable to use N-methyldiethanolamine and cyclohexyldiethanolamine in combination.
[0019] In the exemplary embodiment, the component (B) may be provided by a single one or a plurality of the above substances.
[0020] When contents of the amine components used as the rust resistant component in the water-based metalworking fluid are the same, the amine component having a smaller molecular weight exhibits more favorable rust resistance. This means that the amine component having a smaller molecular weight can provide rust resistance equivalent to those of the amine component having a larger molecular weight with a content lower than a content of the amine component having a larger molecular weight. However, the amine component having a smaller molecular weight, particularly a molecular weight of 90 or less, is highly volatile, so that the amine component intensifies odor as the content thereof is increased in the fluid. Accordingly, it is generally difficult to achieve both of less odor and rust resistance by only adjusting the content of the rust resistant component.
[0021] In the water-based metalworking fluid in the exemplary embodiment, the molar ratio of the amine component having a molecular weight of 90 or less to the total of the amine components (i.e., the amine component having a molecular weight of 90 or less/the total of the amine components) is 0.67 or less, preferably in a range of 0.6 to 0.4, more preferably in a range of 0.55 to 0.5. When the molar ratio exceeds 0.67, odor of the water-based metalworking fluid is unfavorably intensified to have an adverse influence on human bodies and working environments.
[0022] In view of handling ability, the water-based metalworking fluid in the exemplary embodiment is preferably prepared as a stock solution having a high concentration, so that a user dilutes the stock solution as necessary with water to use the diluted stock solution as metalworking fluid.
[0023] A solvent for the stock solution is the most preferably water, but may be mineral oil or synthetic oil.
[0024] Such mineral oil or synthetic oil used as the solvent for preparing the stock solution is not specifically limited but may be any base oil generally used for the metalworking fluid. Such mineral oil or synthetic oil preferably has kinematic viscosity at 40 degrees C. in a range of 1 to 50 mm.sup.2/s, more preferably in a range of 2 to 30 mm.sup.2/s. When the kinematic viscosity of the base oil is too high, the fluid may adhere to a workpiece, whereby a large amount of the fluid may be carried together with the workpiece, which may be economically unfavorable. In contrast, when the kinematic viscosity of the base oil is too low, mist generation may unfavorably deteriorate workability. A pour point (i.e., the index of low-temperature fluidity) of the base oil is not subject to any limitations, but preferably -10 degrees C. or less.
[0025] As the mineral oil or the synthetic oil, various kinds of oil are available. The mineral oil or the synthetic oil may be suitably selected therefrom, depending on the usage.
[0026] Examples of the mineral oil are oil fraction obtained by atmospherically distilling paraffin-based crude oil, intermediate-based crude oil or naphthene-based crude oil or by vacuum-distilling residual oil formed by atmospheric distilling, and purified oil obtained by refining the oil fraction in accordance with an ordinary method. Examples of the purified oil are solvent-refined oil, hydrogenerated refined oil, dewaxing-processed oil, and white clay-processed oil.
[0027] On the other hand, examples of the synthetic oil are poly-.alpha.-olefin, .alpha.-olefin copolymer, polybutene, alkylbenzene, polyolester, diacid ester, polyoxyalkylene glycol, polyoxyalkylene glycol ester, polyoxyalkylene glycol ether, and silicone oil. Among the above synthetic oil, poly-.alpha.-olefin and .alpha.-olefin copolymer are preferable. The base oil may contain single one or a plurality of the above mineral oil or synthetic oil, or may contain both the mineral oil and the synthetic oil.
[0028] The component (A) is preferably contained in the stock solution at a ratio of 1 to 20 mass % of the total amount of the stock solution, more preferably 5 to 14 mass %, further preferably 9 to 13 mass %. When the ratio of the component (A) exceeds 20 mass %, odor is unfavorably intensified.
[0029] The component (B) is preferably contained in the stock solution at a ratio of 5 to 30 mass % of the total amount of the stock solution, more preferably 10 to 30 mass %, further preferably 22 to 29 mass %. When the ratio of the component (B) is less than 5 mass %, odor is unfavorably intensified. Even when the ratio of the component (B) exceeds 30 mass %, rust resistance for iron is not further enhanced, resulting in high cost, which is economically unfavorable.
[0030] When the water-based metalworking fluid in the exemplary embodiment is used, the above-described stock solution is preferably diluted with water an amount of which is 200 times or less as much as that of the stock solution by mass ratio. The stock solution is more preferably diluted to 10 to 100 times, further preferably 20 to 50 times. When the stock solution is diluted with water the amount of which exceeds 200 times as much as that of the stock solution, the prepared fluid unfavorably exhibits insufficient rust resistance.
[0031] The water-based metalworking fluid may be blended as necessary with various known additives as long as an object of the present invention is not hampered. Examples of the additives are a fungicide, aliphatic carboxylic acid, metal deactivator (anticorrosion agent), extreme pressure agent, oiliness agent, surfactant and antifoaming agent.
[0032] The fungicide is exemplified by 2-pyridylthio-1-oxide salt. Examples of the fungicide are 2-pyridylthio-1-oxide sodium, zinc bis(2-pyridylthio-1-oxide), and bis(2-sulfidepyridine-1-olato) copper. Among the above, 2-pyridylthio-1-oxide sodium is particularly preferable because the substance is effective on general bacteria and molds in a wide range even when contained therein with a low concentration. In view of blending effects, the fungicide is contained in the stock solution with a content of approximately 0.01 to 5 mass % of the total amount of the final diluted fluid.
[0033] The aliphatic carboxylic acid is added to the fluid in order to further enhance cutting capabilities, grinding capabilities and rust resistance of the fluid. The aliphatic carboxylic acid is exemplified by carboxylic acid having 6 to 60 carbon atoms and/or dicarboxylic acid. Specific examples of the aliphatic carboxylic acid are caproic acid, caprylic acid, nonane acid, lauric acid, stearic acid, olein acid, ricinolein acid, hydroxyfatty acid (such as recinoleic acid or 12-hydroxystearic acid), arachidic acid, behenic acid, melissic acid, isononane acid (3,5,5-trimethylhexane acid), neo-decane acid, isostearic acid, fatty acid extracted from fat and oil such as soy oil fatty acid, coconut oil fatty acid or rape-seed oil fatty acid, acid extracted from petroleum such as naphthene acid, adipic acid, sebacic acid (decanedioic acid), dodecanoic diacid, monohydroxy arachidic acid or dihydroxy arachidic acid, and synthetic fatty acid such as dimer or trimer of olein acid, recinoleic acid, ricinolein acid, 12-hydroxystearic acid. In view of antifoaming of the fluid and stability of hard water, particularly preferable examples of monocarboxylic acid are caproic acid having 8 to 10 carbon atoms, nonane acid having 8 to 10 carbon atoms, decane acid having 8 to 10 carbon atoms, isononane acid having 8 to 10 carbon atoms, and neodecane acid having 8 to 10 carbon atoms while examples of particularly preferable dicarboxylic acid are nonane diacid, undecanoic diacid, sebacic acid (decanedioic acid), and dodecanoic diacid. In view of blending effects, the aliphatic carboxylic acid is contained in the stock solution with a content of approximately 0.1 to 1.5 mass % of the total amount of the final diluted fluid.
[0034] The aliphatic carboxylic acid reacts with the amine components to form an aliphatic carboxylic acid amine salt, thereby enhancing processability or emulsion stability serving as an emulsifier to further enhance rust resistance.
[0035] A sum of the aliphatic carboxylic acid component and the amine components preferably accounts for 40 to 60 mass % of the total amount of the stock solution, more preferably 47 to 55 mass %, further preferably 50 to 53 mass %.
[0036] Examples of the metal deactivator are benzotriazole, benzotriazole derivative, imidazoline, pyrimidine derivative, thiadiazole and thiadiazole. The metal deactivator may be provided by a single one or a plurality of the above substances. In view of blending effects, the metal deactivator is contained in the stock solution with a content of approximately 0.01 to 3 mass % of the total amount of the final diluted fluid.
[0037] Examples of the extreme pressure agent are a sulfur-based extreme pressure agent, a phosphorus-based extreme pressure agent, an extreme pressure agent containing sulfur and metal and an extreme pressure agent containing phosphorus and metal. The extreme pressure agent may be provided by a single one or a plurality of the above substances. The extreme pressure agent may be any extreme pressure agent, as long as the extreme pressure agent contains sulfur atoms and/or phosphorus atoms in its molecule and as long as the extreme pressure agent can provide load bearing effects and wear resistance. Examples of the extreme pressure agent containing sulfur in its molecule are sulfurized fat and oil, sulfurized fatty acid, ester sulfide, olefin sulfide, dihydrocarbyl polysulfide, a thiadiazole compound, an alkylthiocarbamoyl compound, a triazine compound, a thioterpene compound, a dialkylthiodipropionate compound and the like. In view of blending effects, the extreme pressure agent is contained in the stock solution with a content of approximately 0.05 to 0.5 mass % of the total amount of the final diluted fluid.
[0038] Examples of the oiliness agent are a fatty acid compound such as fatty alcohol, fatty acid or fatty acid metal salt, an ester compound such as polyol ester, sorbitan ester or glyceride, an amine compound such as fatty amine and the like. In view of blending effects, the oiliness agent is contained in the stock solution with a content of approximately 0.2 to 2 mass % of the total amount of the final diluted fluid.
[0039] Examples of the antifoaming agent are methyl silicone oil, fluorosilicone oil, and polyacrylate. In view of blending effects, the antifoaming agent is contained in the stock solution with a content of approximately 0.004 to 0.02 mass % of the total amount of the final diluted fluid.
[0040] The surfactant is not subject to any limitations. A nonionic surfactant, an anionic surfactant, a cationic surfactant, an ampholytic surfactant or a mixture thereof is usable as the surfactant. Preferable examples of the surfactant are a nonionic surfactant, an anionic surfactant, or a mixture thereof.
[0041] Examples of the nonionic surfactant include a polyoxyalkylene surfactant such as: polyoxyalkyleneglycol, a monoether compound thereof or a diether compound thereof; and glycerin, an adduct of glycerin with alkylene oxide or an ether compound of glycerin, esters of carboxylic acid and alcohol, and an adduct of alkylamine with alkylene oxide.
[0042] Examples of the anionic surfactant include a salt of carboxylic acid (e.g., a saturated or unsaturated fatty acid or hydroxy fatty acid having 7 to 22 carbon atoms) or sulfonic acid with amine or metal, an ester of a polycondensation product of a hydroxy fatty acid (e.g., ricinoleic acid) with fatty acid or a salt of the ester with amine or metal, phosphoric ester salts such as sulfates (e.g., dialkyl sodium sulfosuccinate), a polymerized polymer surfactant provided by partially saponifying olefin (e.g., styrene) with a maleic anhydride copolymerized product, and a naphthalenesulfonate-formalin fused polymer surfactant.
[0043] The water-based metalworking fluid in the exemplary embodiment, which is diluted as necessary with water so that its concentration is adjusted suitably for the usage, is preferably applied in various metalworking fields such as cutting, grinding, polishing, squeezing, drawing, flatting and the like. Further, because of an excellent rust resistance for metal products and less odor, the water-based metalworking fluid in the exemplary embodiment is less harmful to environments and human bodies.
EXAMPLES
[0044] Next, the invention will be described in more detail with reference to Examples. However, the invention is not limited at all by the Examples.
Examples 1 to 4, Comparatives 1 to 3
[0045] A water-based metalworking fluid (stock solution) according to each of Examples 1 to 4 and Comparatives 1 to 3 was prepared by blending components shown in Table 1. After the prepared stock solution was diluted with water to a certain concentration, Examples 1 to 4 and Comparatives 1 to 3 each were evaluated in terms of the following characteristics. Evaluation results are shown in Table 1.
(1) Rust Resistance (in Accordance with DIN51360-02-A)
[0046] Test for rust resistance was conducted at room temperature for two hours on diluted solutions prepared by diluting the stock solution with tap water to the concentration of 1.0 mass %, 1.1 mass %, 1.2 mass %, 1.3 mass %, 1.4 mass %, 1.5 mass % and 2.0 mass % respectively. The minimum concentration at which a rust value had become 0 was set as limit concentration (mass %) in terms of rust resistance.
(2) Odor
[0047] An aqueous solution was prepared by diluting the stock solution with an ion-exchange water so that the concentration of the stock solution was 10.0 mass %. 100 mL of the aqueous solution was hermetically sealed in a 300 mL conical flask and heated at 100 degrees C. for three minutes. The heated aqueous solution was measured in terms of odor intensity using an odor sensor (e-nose mobile manufactured by Karumoa Inc.) A value indicates an odor index (relative value) representing an odor intensity. The larger value indicates the intenser odor.
TABLE-US-00001 TABLE 1 Compar- Compar- Comparative Example 1 Example 2 Example 3 Example 4 ative 1 ative 2 3 Blending Decanedioic acid 2.9 2.9 2.9 2.9 -- 2.9 2.9 Composition Dodecanedioic acid 2.2 2.2 2.2 2.2 12.3 2.2 2.2 (mass %) Neodecanoic acid 9.9 9.9 9.9 9.9 -- 9.9 9.9 3,5,5-trimethyl hexanoic acid -- -- -- -- 1.0 -- -- N-methyldiethanolamine (Component B) 16.8 22.1 13.3 9.8 -- -- -- 1-amino-2-propanol (Component A) 12.2 9.2 14.3 16.3 24.6 22.0 24.5 cyclohexyldiethanolamine (Component B) 6.2 6.2 6.2 6.2 10.0 6.2 0.0 1,2,3-benzotriazole 0.5 0.5 0.5 0.5 1.0 0.5 0.5 Other components.sup.1) 0.9 0.9 0.9 0.9 0.9 0.9 0.9 Water 48.4 46.1 49.8 51.3 50.2 55.4 59.1 Total 100.0 100.0 100.0 100.0 100.0 100.0 100.0 Amine component having a molecular weight of 0.50 0.37 0.58 0.67 0.86 0.90 1.00 90 or less/Total amine components (mol/mol) Total of Component A (mass %) 12.2 9.2 14.3 16.3 24.6 22.0 24.5 Total of Component B (mass %) 23.0 28.3 19.5 16.0 10.0 6.2 0.0 Acid component + Amine component 50.7 52.9 49.2 47.8 48.9 43.7 39.9 Evaluation Limit concentration of rust resistance (mass %) 1.1 1.1 1.1 1.1 1.1 1.2 1.3 Item Odor (Intensity) 3365 3225 3805 3924 4310 4417 4781
[0048] As is obvious from Table 1, the water-based metalworking fluid in each of Examples 1 to 4 exhibits an excellent rust resistance and causes less odor. In contrast, since the water-based metalworking fluid in each of Comparatives 1 to 3 does not contain the essential components required in the invention, the odor intensity is high although the rust resistance is excellent.
INDUSTRIAL APPLICABILITY
[0049] A water-based metalworking fluid of the invention is usable in a metalworking field such as cutting or grinding.
* * * * *
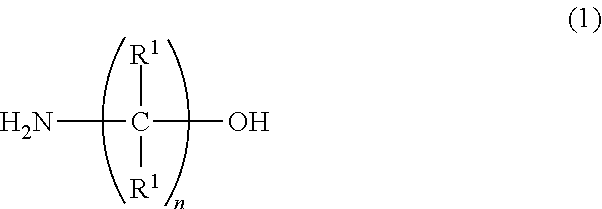




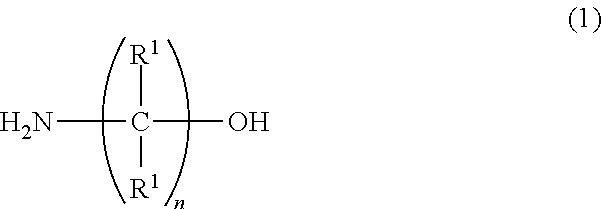

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.