Process For The Preparation Of Substituted Prolyl Peptides And Similar Peptidomimetics
Ruijter; Eelco ; et al.
U.S. patent application number 13/581179 was filed with the patent office on 2012-12-27 for process for the preparation of substituted prolyl peptides and similar peptidomimetics. This patent application is currently assigned to Vereniging voor Christelijk hoger onderwijs wetenschappelijk onderzoek en patieentenzorg. Invention is credited to Romano Orru, Marloes Polak, Eelco Ruijter, Nicholas Turner, Anass Znabet.
| Application Number | 20120329704 13/581179 |
| Document ID | / |
| Family ID | 43088069 |
| Filed Date | 2012-12-27 |












View All Diagrams
| United States Patent Application | 20120329704 |
| Kind Code | A1 |
| Ruijter; Eelco ; et al. | December 27, 2012 |
PROCESS FOR THE PREPARATION OF SUBSTITUTED PROLYL PEPTIDES AND SIMILAR PEPTIDOMIMETICS
Abstract
The present invention relates to a process for the stereoselective preparation of a compound having the general formula (I) or its respective diastereomers: comprising reacting a compound having the general formula (II) or its diastereomers: with a compound of the general formula III: R.sup.3--COOH and a compound of the general formula IV: R.sup.4--NC wherein R.sup.1 represents each independently, or jointly a substituted or unsubstituted alkyl, alkenyl, alkynyl, aromatic or non-aromatic, mono-, di- or tricyclic, or heterocyclic structure, and R.sup.2 represents a hydrogen atom, a substituted or unsubstituted alkyl, alkenyl, alkynyl, aromatic or non-aromatic, mono-, di- or tricyclic, or heterocyclic structure, and R.sup.3 represents a substituted or unsubstituted alkyl, alkenyl, or alkynyl, or an aromatic or non-aromatic aromatic or non-aromatic, mono-, di- or tricyclic, or heterocyclic structure. ##STR00001## R.sup.3--COOH (III) R.sup.4--NC (IV)
| Inventors: | Ruijter; Eelco; (Woerden, NL) ; Orru; Romano; (Haarlem, NL) ; Znabet; Anass; (Amsterdam, NL) ; Polak; Marloes; (Voorhout, NL) ; Turner; Nicholas; (Manchester, GB) |
| Assignee: | Vereniging voor Christelijk hoger
onderwijs wetenschappelijk onderzoek en patieentenzorg Amsterdam NL |
| Family ID: | 43088069 |
| Appl. No.: | 13/581179 |
| Filed: | September 16, 2010 |
| PCT Filed: | September 16, 2010 |
| PCT NO: | PCT/EP10/63656 |
| 371 Date: | September 13, 2012 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 61307873 | Feb 25, 2010 | |||
| Current U.S. Class: | 514/3.7 ; 435/68.1; 514/21.9; 530/330 |
| Current CPC Class: | C07D 209/58 20130101; C07D 403/06 20130101; C07D 209/94 20130101; C07C 291/10 20130101; C07C 2601/04 20170501; C07D 241/24 20130101; C07C 231/06 20130101; C07C 2601/02 20170501; A61P 31/12 20180101; C07D 209/52 20130101; C07C 237/14 20130101; C07D 403/12 20130101 |
| Class at Publication: | 514/3.7 ; 435/68.1; 530/330; 514/21.9 |
| International Class: | A61K 38/06 20060101 A61K038/06; C07K 5/00 20060101 C07K005/00; A61P 31/12 20060101 A61P031/12; C12P 21/02 20060101 C12P021/02 |
Claims
1. A process for stereo-selectively preparing a compound of formula I or diastereomer thereof: ##STR00054## comprising reacting a compound of formula II or a diastereomer thereof: ##STR00055## with a compound of formula III: R.sup.3--COOH (III) and a compound of formula IV: R.sup.4--NC (IV) wherein R.sup.1 represents each independently, or jointly a substituted or unsubstituted alkyl, alkenyl, alkynyl, aromatic or non-aromatic, mono-, di- or tricyclic, or heterocyclic group, R.sup.2 represents a hydrogen atom, a substituted or unsubstituted alkyl, alkenyl, alkynyl, aromatic or non-aromatic, mono-, di- or tricyclic, or heterocyclic group, and R.sup.3 represents a substituted or unsubstituted alkyl, alkenyl, or alkynyl, or an aromatic or non-aromatic aromatic or non-aromatic, mono-, di- or tricyclic, or heterocyclic group.
2. The process according to claim 1, wherein both substituents R.sup.1 jointly form a substituted or unsubstituted 3-, 4-, 5-, 6-, 7- or 8-membered ring structure.
3. The process according to claim 2, wherein R.sup.1 is chosen such that the compound of formula I has a structure of formula V: ##STR00056##
4. The process according to claim 2, wherein R.sup.1 is chosen such that the compound of formula I has a structure of formula VI: ##STR00057##
5. The process according to claim 2, wherein R.sup.1 is chosen such that the compound of formula I has a structure of formula VII: ##STR00058##
6. The process according to claim 1, wherein R.sup.2 represents a dipeptide of formula VIII: ##STR00059## wherein R.sup.a and R.sup.b each independently represents a hydrogen atom, a halogen atom, C.sub.1-14 alkyl optionally substituted by halogen, a cycloalkyl group, an aryl group, a lower alkoxy group, a lower thioalkyl group, a cycloalkyloxy group, an aralkyloxy group or an alkanoyl group; a hydroxyl group, a nitro group, a formyl group, an amino group which may be protected or substituted, a cycloalkyloxy, aralkyloxy, alkanoyl, ureido or mono-, di- or tricyclic heterocyclic group, all of which groups may optionally be substituted.
7. The process according to claim 1, wherein the compound of formula IV has a structure of formula IX: ##STR00060## wherein R.sup.d, R.sup.e and R.sup.f each independently represents a hydrogen atom, a halogen atom, a substituted or unsubstituted alkyl, alkenyl, alkynyl, aromatic or non-aromatic, mono-, di- or tricyclic and/or a heterocyclic group.
8. The process according to claim 1, further comprising: preparing the compound of formula IIa or IIb by desymmetrization of 3,4-substituted meso-pyrrolidine.
9. The process according to claim 8, wherein the desymmetrization comprises treating the meso-pyrrolidine with an enzyme capable of catalysing oxidation of an amine in an enantio selective manner.
10. The method according to claim 9, wherein the enzyme is a monoamine oxidase N derived from Aspergillus niger.
11. The process according to claim 1, wherein R.sup.2 is chosen such that the compound of formula III has a structure of formula XV: ##STR00061##
12. The process according to claim 1, wherein R.sup.3 is chosen such that the compound of formula IV has a structure of formula XVI: ##STR00062##
13. The process according to claim 12, further comprising: isolating from a reaction product a compound of formula XVII: ##STR00063##
14. The process according to claim 13, further comprising: subjecting the compound of formula XVII to a saponification, followed by a selective oxidation to form a compound of formula XVIII: ##STR00064##
15. The process according to claim 14, wherein the saponification is carried out by contacting the compound of formula XVII with an alkaline metal carbonate in a suitable solvent, to obtain a saponified alcohol product.
16. The process according to claim 15, wherein the selective oxidation is carried out by contacting the saponified alcohol product with a suitable oxidant in a suitable solvent.
17. A compound obtained by the process according to claim 1, or an enantiomer, stereoisomer, rotamer, tautomer, racemate, pharmaceutically acceptable salt or solvate thereof.
18-24. (canceled)
25. A method for catalyzing an organochemical reaction, comprising adding a compound obtained from the process of claim 1 to the organochemical reaction.
26. (canceled)
27. A composition comprising a compound of formula I obtained from the process of claim 1 and a pharmaceutically acceptable carrier, diluent or excipient.
28. The process according to claim 1, further comprising: formulating a compound of formula (I) and optionally a utilizable carrier to a pharmaceutical composition.
29. A method for treating a condition associated with viral infections in a subject, comprising: administering to a subject in need thereof at least one compound of formula (I) obtained from the process of claim 1 or a pharmaceutically acceptable salt or N-oxide thereof.
30-31. (canceled)
Description
FIELD OF THE INVENTION
[0001] The present invention relates to substituted prolyl peptides and similar peptidomimetics, methods for their preparation, and a variety of uses including as inhibitors of disease-associated targets as well as an organocatalyst component.
BACKGROUND TO THE INVENTION
[0002] Optically pure 3,4-substituted prolyl peptides and related peptidomimetic compounds are of considerable interest in organocatalysis and medicinal chemistry, specifically since they form key structural elements of the hepatitis C virus NS3 protease inhibitors telaprevir and boceprevir as disclosed in for instance WO2003/062265.
[0003] Multicomponent reactions (MCRs) offer the ability to rapidly and efficiently generate collections of structurally and functionally diverse organic compounds. Although MCRs are very efficient by their nature, the stereocontrol in these reactions is mostly not trivial.
[0004] The Ugi reaction is undoubtedly one of the most widely applied MCRs. It is of considerable interest owing to its exceptional synthetic efficiency and is widely used in the field of modern combinatorial and medicinal chemistry. The Ugi reaction involves a one-pot condensation of an aldehyde, an amine, a carboxylic acid and an isocyanide to produce chiral .alpha.-acylaminoamides. In 1982, Nutt and Joullie reported a variation on the Ugi reaction (further referred to herein as Joullie-Ugi reaction, or JU-3CR), which employed substituted 1-pyrrolines to produce substituted prolyl peptides. However, as in most MCRs, controlling the newly formed stereocenter proves highly complex, and therefore the reaction suffers from poor and/or unpredictable (dia)stereoselectivity, as illustrated for instance by WO2006/061585. This document discloses a JU-3CR employing dihydroxypyrolline compounds to form peptidomimetic compounds comprising dihydroxyproline structures. The reported products are formed in only limited yields and mostly unpredictable diastereoselectivity, while requiring the use of protecting groups that are often difficult to remove, such as benzyl groups.
[0005] Accordingly, the known multicomponent reactions for the preparation of proline derivative comprising peptides and peptidomimetics suffer from poor and/or unpredictable (dia)stereoselectivity. Alternative process schemes are tedious, require numerous steps and hence suffer from low yields.
[0006] Notwithstanding the state of the art it would be desirable to provide an enantioselective Joullie-Ugi reaction, or JU-3CR for preparing substituted prolyl peptide structures.
SUMMARY OF THE INVENTION
[0007] The present invention relates to a stereoselective process for the preparation of a compound having the general formula Ia or Ib:
##STR00002##
comprising reacting a compound having the general formula II or its diastereomers:
##STR00003##
with a compound of the general formula III:
R.sup.3--COOH (III)
and a compound of the general formula IV:
R.sup.4---NC (IV)
[0008] wherein R.sup.1 represents each independently, or jointly a substituted or unsubstituted lower alkyl, alkenyl, alkynyl, aromatic or non-aromatic, or heterocyclic structure, and
[0009] wherein R.sup.2 represents each independently a hydrogen, or each independently or jointly a substituted or unsubstituted alkyl, alkenyl, alkynyl, aromatic or non-aromatic, mono-, di- or tricyclic, or heterocyclic structure, and
[0010] R.sup.3 represents a substituted or unsubstituted alkyl, alkenyl, alkynyl, aromatic or non-aromatic, mono-, di- or tricyclic, heterocyclic, alkyloxy, alkanoyl, amino, ureido or a peptide structure, and
[0011] R.sup.4 represents a substituted or unsubstituted alkyl, alkenyl, or alkynyl, or an aromatic or non-aromatic aromatic or non-aromatic, mono-, di- or tricyclic, or heterocyclic alkyl, alkenyl, alkynyl, aromatic or non-aromatic, mono-, di- or tricyclic, heterocyclic, alkyloxy, alkanoyl, amino, ureido or a peptide structure.
[0012] In the subject process, stereoselectivity refers to enantioselectivity or diastereoselectivity, depending on the substrates.
[0013] As used herein, the term "alkyl" denotes a saturated straight or branched hydrocarbon chain comprising carbon and hydrogen atoms, for example, methyl, ethyl, propyl, isopropyl, n-butyl, i-butyl, 2-butyl, t-butyl and the like. Preferred alkyl groups are groups with 1-10 carbon atoms.
[0014] The term "alkyloxy" denotes an alkyl group as defined above, which is attached via an oxygen atom.
[0015] The term "alkyl substituted by halogen" denotes an alkyl group as defined above, wherein at least one hydrogen atom is replaced by halogen, for example CF.sub.3, CHF.sub.2, CH.sub.2F, CH.sub.2CF.sub.3, CH.sub.2CH.sub.2CF.sub.3, CH.sub.2CF.sub.2CF.sub.3 and the like. The term "halogen" denotes chlorine, iodine, fluorine and bromine.
[0016] The term "cycloalkyl" denotes a saturated carbocyclic ring, preferably containing from 3 to 10 carbon atoms, more preferably 3 to 8 carbon atoms, yet more preferably from 3 to 6 carbon atoms, for example, cyclopropyl, cyclobutyl, cyclopentyl or cyclohexyl.
[0017] The term "cycloalkenyl" denotes a saturated carbocyclic ring, preferably containing from 3 to 10 carbon atoms, more preferably 3 to 8 carbon atoms, yet more preferably from 3 to 6 carbon atoms, for example, cyclopropyl, cyclobutyl, cyclopentyl or cyclohexyl.
[0018] The term cyclocalkyl preferably comprises (C.sub.1-C.sub.10) alkyl. The term "(C.sub.1-C.sub.10) alkyl" means a straight chain or branched non cyclic hydrocarbon having from 1 to 10 carbon atoms. Representative straight chain --(C.sub.1-C.sub.10) alkyls include (C.sub.1, C.sub.2, C.sub.3, C.sub.4, C.sub.5, C.sub.6, C.sub.7, C.sub.8, C.sub.9 and C.sub.10 alyls, such as -methyl, -ethyl, -n propyl, -n-butyl, -n-pentyl, -n-hexyl, -n-heptyl, -n-octyl, -n-nonyl, and -n-decyl.
[0019] A branched alkyl means that one or more straight chain --(C.sub.1-C.sub.8) alkyl groups, such as -methyl, -ethyl or -propyl, replace one or both hydrogens in a --CH.sub.2-- group of a straight chain alkyl. A branched non cyclic hydrocarbon means that one or more straight chain (C.sub.1-C.sub.10) alkyl groups, such as -methyl, -ethyl or -propyl, replace one or both hydrogens in a --CH.sub.2-- group of a straight chain non cyclic hydrocarbon.
[0020] The term "--(C.sub.1-C.sub.2)alkyl" means a straight chain non cyclic hydrocarbon having 1 or 2 carbon atoms. Representative straight chain "--(C.sub.1-C.sub.2)alkyl groups include -methyl and -ethyl.
[0021] The term "(C.sub.1-C.sub.3)alkyl" means a straight chain or branched non cyclic hydrocarbon having from 1 to 3 carbon atoms. Representative straight chain (C.sub.1-C.sub.3)alkyl groups include -methyl, -ethyl, and -n-propyl. Representative branched --(C.sub.1-C.sub.3)alkyl groups include -iso-propyl.
[0022] The term "(C.sub.1-C.sub.4)alkyl" means a straight chain or branched non cyclic hydrocarbon having from 1 to 4 carbon atoms. Representative straight chain --(C.sub.1-C.sub.4)alkyl groups include -methyl, -ethyl, -n-propyl, and -n-butyl. Representative branched --(C.sub.1-C.sub.4)alkyls include -iso-propyl, -sec-butyl, -iso-butyl, and -tert-butyl. The term "(C.sub.1-C.sub.6)alkyl" means a straight chain or branched non cyclic hydrocarbon having from 1 to 6 carbon atoms. Representative straight chain --(C.sub.1-C.sub.6)alkyls include -methyl, -ethyl, -n-propyl, -n-butyl, -n-pentyl, and -n-hexyl. Representative branched (C.sub.1-C.sub.6)alkyls include iso-propyl, -sec-butyl, -iso-butyl, -tert-butyl, -iso-pentyl, -neopentyl, 1-methylbutyl, 2-methylbutyl, 3-methylbutyl, 1,1-dimethylpropyl, 1,2-dimethylpropyl, 1-methylpentyl, 2-methylpentyl, 3-methylpentyl, 4-methylpentyl, 1-ethylbutyl, 2-ethylbutyl, 3-ethylbutyl, 1,1-dimethylbutyl, 1,2-dimethylbutyl, 1,3-dimethylbutyl, 2,2-dimethylbutyl, 2,3-dimethylbutyl, and 3,3-dimethylbutyl.
[0023] Representative branched --(C.sub.1-C.sub.10) alkyl groups include iso-propyl, sec-butyl, iso-butyl, tert-butyl, iso-pentyl, neopentyl, 1-methylbutyl, 2-methylbutyl, 3-methylbutyl, 1,1-dimethylpropyl, 1,2-dimethylpropyl, 1-methylpentyl, 2-methylpentyl, 3-methylpentyl, 4-methylpentyl, 1-ethylbutyl, 2-ethylbutyl, 3-ethylbutyl, 1,1-dimethylbutyl, 1,2-dimethylbutyl, 1,3-dimethylbutyl, 2,2-dimethylbutyl, 2,3-dimethylbutyl, 3,3-dimethylbutyl, 1-methylhexyl, 2-methylhexyl, 3-methylhexyl, 4-methylhexyl, 5-methylhexyl, 1,2-dimethylpentyl, 1,3-dimethylpentyl, 1,2-dimethylhexyl, 1,3-dimethylhexyl, 3,3-dimethylhexyl, 1,2-dimethylheptyl, 1,3-dimethylheptyl, and 3,3-dimethylheptyl.
[0024] The term "(C.sub.2-C.sub.10)alkenyl" means a straight chain or branched non cyclic hydrocarbon having from 2 to 10 carbon atoms and including at least one carbon-carbon double bond. A branched alkenyl means that one or more straight chain --(C.sub.1-C.sub.8)alkyl groups, such as -methyl, -ethyl or -propyl, replace one or both hydrogens in a --CH.sub.2-- or --CH.dbd. group of a straight chain alkenyl. Representative straight chain and branched (C.sub.2-C.sub.10)alkenyl groups include -vinyl, -allyl, 1-butenyl, -2-butenyl, -iso-butylenyl, -1-pentenyl, -2-pentenyl, -3-methyl-1-butenyl, 2-methyl-2-butenyl, -2,3-dimethyl-2-butenyl, -1-hexenyl, -2-hexenyl, -3-hexenyl, -1-heptenyl, -2-heptenyl, -3-heptenyl, -1-octenyl, -2-octenyl, -3-octenyl, -1-nonenyl, -2-nonenyl, -3-nonenyl, -1-decenyl, -2-decenyl, -3-decenyl, and the like.
[0025] The term "C.sub.1-C.sub.6)alkoxy" represents a straight chain or branched non cyclic hydrocarbon having one or more ether groups and from 1 to 6 carbon atoms. Representative straight chain and branched C.sub.1-C.sub.6)alkoxy groups include -methoxy, -ethoxy, -methoxymethyl, -2-methoxyethyl, -5-methoxypentyl, -3-ethoxybutyl and the like.
[0026] The term "C.sub.3-C.sub.12 cycloalkyl" groups refers to a saturated monocyclic hydrocarbon having from 3 to 12 carbon atoms. Representative C.sub.3-C.sub.12 cycloalkyl groups are -cyclopropyl, -cyclobutyl, -cyclopentyl, -cyclohexyl, -cycloheptyl, -cyclooctyl, -cyclononyl, -cyclodecyl, and -cyclododecyl.
[0027] "C.sub.4-C.sub.8 cycloalkyl" groups refers to "4- to 8-member cycloalkyl rings", meaning a saturated monocyclic hydrocarbon having from 4 to 8 carbon atoms. Representative --C.sub.4-C.sub.8 cycloalkyl groups are -cyclobutyl, -cyclopentyl, -cyclohexyl, -cycloheptyl, and -cyclooctyl.
[0028] C.sub.3-C.sub.8 cycloalkyl groups mean a saturated monocyclic hydrocarbon having from 3 to 8 carbon atoms. Representative C.sub.3-C.sub.8 cycloalkyl groups include -cyclopropyl, -cyclobutyl, -cyclopentyl, -cyclohexyl, -cycloheptyl, and -cyclooctyl.
[0029] C.sub.3-C.sub.7 cycloalkyl groups means a saturated monocyclic hydrocarbon having from 3 to 7 carbon atoms. Representative C.sub.3-C.sub.7 cycloalkyl groups include -cyclopropyl, -cyclobutyl, -cyclopentyl, -cyclohexyl, and -cycloheptyl.
[0030] "-(6- to 10-membered) heterobicyclic" or "-(6- to 10-membered) bicycloheterocyclo" group refers to a 6 to 10 membered bicyclic, heterocyclic ring which is either saturated, unsaturated non-aromatic, or aromatic. A -(6- to 10-membered)heterobicyclic group contains from 1 to 4 heteroatoms independently selected from nitrogen, which can be quaternized; oxygen; and sulfur, including sulfoxide and sulfone. The -(6- to 10-membered)heterobicyclic group can be attached via a nitrogen or carbon atom. Representative -(6- to 10-membered)heterobicyclic groups include-3-azabicyclo[3.1.0]hexane, -quinolinyl, -isoquinolinyl, -chromonyl, -coumarinyl, -indolyl, -indolizinyl, benzo[b]furanyl, benzo[b]thiophenyl, -indazolyl, -purinyl, -4H-quinolizinyl, isoquinolyl, -quinolyl, -phthalazinyl, -naphthyridinyl, -carbazolyl, -[beta]-carbolinyl, -indolinyl, -isoindolinyl, -1,2,3,4-tetrahydroquinolinyl, -1,2,3,4-tetrahydroisoquinolinyl, pyrrolopyrrolyl and the like.
[0031] The term "CH.sub.2(halo)" group means a methyl group where one of the hydrogens of the methyl group has been replaced with a halogen. Representative --CH.sub.2(halo) groups include --CH.sub.2F, --CH.sub.2Cl, --CH.sub.2Br, and --CH.sub.2I.
[0032] The term "--CH(halo).sub.2" means a methyl group where two of the hydrogens of the methyl group have been replaced with a halogen. Representative --CH(halo).sub.2 groups include --CHF.sub.2, --CHCl.sub.2, --CHBr.sub.2, --CHBrCl, --CHClI, and --CHI.sub.2.
[0033] The term "--C(halo).sub.3" means a methyl group where each of the hydrogens of the methyl group has been replaced with a halogen. Representative --C(halo).sub.3 groups include --CF.sub.3, --CCl.sub.3, --CBr.sub.3, and --Cl.sub.3.
[0034] "-Halogen" or "-halo" means --F, --Cl, --Br, or --I.
[0035] "Oxo", ".dbd.O", and the like as used herein mean an oxygen atom doubly bonded to carbon or another element.
[0036] When a first group is "substituted with one or more" second groups, one or more hydrogen atoms of the first group are replaced with a corresponding number of second groups. When the number of second groups is two or greater, each second group can be the same or different.
[0037] The term "aryl" as used herein is a carbocyclic ring system, containing from 6 to 10 carbon atoms forming one or more rings, and wherein at least one ring is aromatic in nature, for example phenyl, naphthyl or 5,6,7,8-tetrahydronaphthalen-1-yl. The most preferred aryl group is phenyl.
[0038] The term "enantiomeric excess" refers to a difference between the amount of one enantiomer and the amount of the other enantiomer that is present in the product mixture. Thus for example, enantiomeric excess of 96% refers to a product mixture having 98% of one enantiomer and 2% of the other enantiomer.
[0039] The terms "enantiomeric excess" and "diastereomeric excess" are used interchangeably herein. Compounds with a single stereocenter are referred to as being present in "enantiomeric excess," those with at least two stereocenters are referred to as being present in "diastereomeric excess." In the graphic representations of racemic or enantiomerically pure compounds used herein, solid and broken wedges are used to denote the absolute configuration of a chiral element; wavy lines indicate disavowal of any stereochemical implication which the bond it represents could generate; solid and broken bold lines are geometric descriptors indicating the relative configuration shown but not implying any absolute stereochemistry; and wedge outlines and dotted or broken lines denote enantiomerically pure compounds of indeterminate absolute configuration.
[0040] The term "heterocyclic" embraces both "heteroaryl" and "heterocycloalkyl" groups. The term "heteroaryl" as used herein is an aromatic ring system, containing from 5 to 10 ring atoms forming one or more rings, wherein at least one ring atom is a heteroatom selected from the group consisting of O, N and S, and wherein at least one ring is aromatic in nature, for example oxazolyl, pyridyl, thiophenyl, quinolinyl, pyrrolyl, furyl, benzoimidazolyl, imidazolyl and the like. The most preferred group is pyridyl.
[0041] The term "heterocycloalkyl" denotes a fully saturated ring system, wherein one or two ring atoms are N, O or S, for example piperazinyl, pyrrolidinyl, morpholinyl or piperidinyl.
[0042] The term "monoamine oxidase" refers to a polypeptide having an enzymatic capability of oxidizing a compound of structural Formula I, supra to the corresponding product of structural Formula II, supra. The polypeptide typically utilizes an oxidized cofactor, such as but not limited to flavin adenine dinucleotide (FAD), flavin adenine mononucleotide (FMN), nicotinamide adenine dinucleotide (NAD), or nicotinamide adenine dinucleotide phosphate (NADP). In a particular embodiment, the oxidized cofactor is FAD. Monoamine oxidases as preferably used herein include naturally occurring (wild type) monoamine oxidases as well as non-naturally occurring engineered polypeptides generated by human manipulation.
[0043] The term "naturally occurring" or "wild type" refers to a polypeptide occurring in nature. For example, a naturally occurring or wild type polypeptide or polynucleotide sequence is a sequence present in an organism that can be isolated from a source in nature and which has not been intentionally modified by human manipulation.
[0044] The term "peptide" denotes polymers of amino acids linked by peptide bonds.
[0045] The term "peptidomimetics" denotes structures that resemble polymers of amino acids linked by peptide bonds, either comprising non-naturally occurring .alpha.-, .beta.- or similar amino acids, or using structurally different building blocks.
[0046] "Pharmaceutically acceptable" such as pharmaceutically acceptable salt, carrier, excipient, etc., means pharmacologically acceptable and substantially non-toxic to the subject to which the particular compound is administered.
[0047] The term "pharmaceutically acceptable salt" embraces salts with inorganic and organic acids, such as hydrochloric acid, nitric acid, sulfuric acid, phosphoric acid, citric acid, formic acid, fumaric acid, maleic acid, acetic acid, succinic acid, tartaric acid, methane-sulfonic acid, p-toluenesulfonic acid and the like.
[0048] The term "Pharmaceutically acceptable N-oxide" refers to N-oxides of tertiary nitrogen atoms in a molecule, which may be more potent than their corresponding tertiary amine, or less. N-oxides may or may not be reduced to their corresponding tertiary amines after indigestion. When N-oxides are converted to their corresponding tertiary amines, the conversion may be in mere trace amounts or nearly quantitative. Further, once formed, N-oxides may be more active than their corresponding tertiary amines, less active or even completely inactive.
[0049] The term "prodrug" refers to a precursor form of the compound that is metabolized to form the active ingredient.
[0050] The term "stereoselective" refers to the preferential formation in a chemical or enzymatic reaction of one stereoisomer over another. Stereoselectivity can be partial, where the formation of one stereoisomer is favoured over the other, or it may be complete where only one stereoisomer is formed. When the stereoisomers are enantiomers, the stereoselectivity is referred to as enantioselectivity, the fraction reported as a percentage of one enantiomer in the sum of both. It is commonly alternatively reported in the art (typically as a percentage) as the enantiomeric excess (e.e.) calculated therefrom according to the formula [major enantiomer-minor enantiomer]/[major enantiomer+minor enantiomer]. Where the stereoisomers are diastereoisomers, the stereoselectivity is referred to as diastereoselectivity, the fraction (typically reported as a percentage) of one diastereomer in a mixture of two diasteromers, commonly alternatively reported as the diastereomeric excess (d.e.). Enantiomeric excess and diastereomeric excess are types of stereomeric excess. The present process allows a stereoselective preparation of the desired compounds in a simple an convergent manner, yielding the desired enantiomers--or diastereomers based on easily available chiral information preferably derived from 3R,4S- or 3S,4R-configured pyrrolidine compounds.
[0051] The term "stereospecificity" refers to the preferential conversion in a chemical or enzymatic reaction of one stereoisomer over another. Stereospecificity can be partial, where the conversion of one stereoisomer is favored over the other, or it may be complete where only one stereoisomer is converted.
[0052] The term "chemoselectivity" refers to the preferential formation in a chemical or enzymatic reaction of one product over another.
[0053] "Therapeutically effective amount" means an amount that is effective to prevent, alleviate or ameliorate symptoms of disease or prolong the survival of the subject being treated.
[0054] As used herein, the terms "stereoisomer", "stereoisomeric form" and the like are general terms for all isomers of individual molecules that differ only in the orientation of their atoms in space. It includes enantiomers and isomers of compounds with more than one chiral center that are not mirror images of one another ("diastereomers").
[0055] The term "chiral centre" refers to a carbon atom to which four different groups are attached.
[0056] The term "enantiomer" or "enantiomeric" refers to a molecule that is non-superimposeable on its mirror image and hence optically active where the enantiomer rotates the plane of polarized light in one direction and its mirror image rotates the plane of polarized light in the opposite direction.
[0057] The term "racemic" refers to a mixture of equal parts of enantiomers which is optically inactive.
[0058] The term "resolution" refers to the separation or concentration or depletion of one of the two enantiomeric forms of a molecule.
[0059] "Substantially enantiomerically pure" as used herein means that the indicated enantiomer of a compound is present to a greater extent or degree than another enantiomer of the same compound. Accordingly, in particular embodiments, a substantially enantiomerically pure compound is present in 80%, 85%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% enantiomeric excess over another enantiomer of the same compound.
[0060] "Substantially stereomerically pure" as used herein means that the indicated enantiomer or diastereomer of a compound is present to a greater extent or degree than another enantiomer or diastereomer of the same compound. As noted above with respect to "stereoselectivity", enantiomeric excess and diastereomeric excess are types of stereomeric excess. Accordingly, in particular embodiments, a substantially stereomerically pure compound is present in 80%, 85%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% stereomeric excess over another enantiomer or diastereomer of the same compound.
[0061] Unlike prior art processes for the enantioselective preparation of compounds according to formula (I), the process of the present invention allows for the synthesis of either enantiomer of according to formula (I) in excellent yield and enantiomeric excess at mild conditions, and in a very small number of steps. In addition, the inventive process of the present invention allows for very effective use of readily available chiral starting information. Insofar, the process of the present invention is highly efficient as it does not produce 50% of the unwanted enantiomer. These advantages combine to make the process of the present invention very economic and amenable to industrial scale up.
[0062] The process according to the invention advantageously allows for the enantioselective formation of products, i.e., may produce products having a high enantiomeric excess. An "enantioselective" process according to the invention hence results in the formation of a product with an enantiomeric excess and/or diastereomeric excess of the desired respective enantiomer or diastereomers.
[0063] In an exemplary embodiment, the method produces the product with an enantiomeric excess of a product from 80% ee (de) to >99.9% ee (de), more preferably from 90% ee (de) to 99.9% ee (de), yet more preferably from 95% ee (de) to >99.7% ee (de), still more preferably from 98% ee (de) to >99.5% ee (de), still more preferably from 99.0% ee (de) to more than 99.3% ee(de).
[0064] For analysis of yield, diastereomeric and/or enantiomeric excess, the product may be analyzed by NMR (e.g., .sup.1H NMR, .sup.13C NMR, etc.), HPLC, GLC, or the like. In some cases, more than one analysis may be performed. For example, a product may be analyzed by NMR, wherein the presence of different enantiomers may be indicated by NMR peaks characteristic of a particular enantiomer upon addition of a chiral shift reagent. In some embodiments, the product may be analyzed using chromatography (e.g., HPLC or GLC), where different enantiomers or diastereomers may exhibit distinct retention times. Yet further, crystallographic evidence may be employed where a product of intermediate can be crystallized to such form that supports a sutibale analysis. A person skilled in the art will be able to determine the appropriate method, or combination of methods, to utilize based upon the product to be analyzed.
DETAILED DESCRIPTION OF THE INVENTION
[0065] In the process according to the invention, the use of the optically active 1-pyrroline compounds of formula II and the diastereomers in a reaction with compounds of general formula (III) and (IV) resulted in the formation of compounds (I) with an unprecedentedly high (dia)stereoselectivity and yield. Without wishing the bound to any particular theory, it is believed that the steric bulk of the substituents at the 3 and 4 position of the pyrroline compounds according to formula IIa or b directs the addition of nucleophiles to the imine with high diastereoselectivity.
[0066] The 1-pyrroline compound according to formula II may be conveniently prepared by the desymmetrization of 3,4-substituted meso-pyrrolidines. This may advantageously be performed in a biocatalytic process, such as a process comprising treating the meso-pyrrolidines with an enzyme capable of catalysing oxidation of the amine in an enantioselective manner to form compound II.
[0067] In the case of R.sup.2 being different from hydrogen, the diastereomers of compound II referred to above include the following compounds of general formula IIa and IIb:
##STR00004##
[0068] which will result predominantly in the formation of the respective stereoisomers of compound I according to general formula Ia and Ib:
##STR00005##
[0069] The (3R,7S)-diastereomers IIc and IId, i.e. the diastereomers having the opposite configuration of the substituents R.sup.2 can also be employed, yielding the equivalent (3R,7S)-configured proline derivatives Ic and Id.
[0070] Monoamine oxidase enzymes suitable for use in biocatalytic process have been used to resolve and deracemize racemic chiral amines via the stereospecific oxidation of one enantiomer to the corresponding imine using oxygen. Derivatives of the flavin dependent monoamine oxidase of Aspergillus niger (MAO N) (Shilling et al. et al. (1995) Biochim. Biophys. Acta. 1243: 529 37) have been reported as useful, in combination with non specific chemical reducing agents, for the deracemization of (d/1) .alpha.-methylbenzylamine to provide enantiomerically pure (93% ee) (d/l) .alpha.-methylbenzylamine (Alexeeva et al. (2002), Angew. Chem. Int. Ed. 41: 3177-3180). Derivatives of the flavin dependent monoamine oxidase of Aspergillus niger were also used for deracemization of (R/S)-2-phenypyrrolidine to provide enantiomerically pure (98% ee) (R)-2-phenypyrrolidine (Carr et al. (2005), ChemBioChem 6: 637 39; Gotor et al. "Enantioselective Enzymatic Desymmetrization in Organic Synthesis," Chem. Rev. (2005) 105: 313.
[0071] Preferably the biocatalytic desymmetrization comprises treating subsequently or simultaneously in situ the obtained oxidised amine with a chemical reducing agent, more preferably a non-enantioselective reducing agent, yet more preferably a reducing agent selected from sodium borohydride, sodium cyanoborohydride, an amine-borane complex or a transfer hydrogenation agent.
[0072] More preferably, the enzyme is a microbial monoamine oxidase, preferably a monoamine oxidase N derived from naturally occurring, selectively bred or genetically modified Aspergillus species, preferably A. niger.
[0073] Most preferably, the biocatalytic desymmetrization is performed using the monoamine oxidase N (MAO-N) from Aspergillus niger according to the method disclosed in WO03080855, and in J. Turner et al., Angew. Chem. Int. Ed. 2002, 41, 3177-3180; and Turner et al., Angew. Chem. Int. Ed. 2003, 42, 4807-4810.
[0074] R.sup.1 according to the invention may each independently or jointly be the same group, and preferably represents a hydrogen atom, a halogen atom, a hydroxyl group, a nitro group, a formyl group, an amino group which may be protected or substituted, a lower alkyl, cycloalkyl, aryl, lower alkoxy, cycloalkyloxy, aralkyloxy, alkanoyl, ureido or monocyclic heterocyclic group. Suitable imino compounds according to formula II are those disclosed in WO-A-2010/008828, more advantageously n paragraph [27] and [29] of this publication.
[0075] More preferably, both substituents R.sup.1 jointly form an optionally substituted 3-, 4-, 5-, 6-, 7- or 8 membered ring structure. This ring structure jointly formed by the substituents R.sup.1 may preferably be a saturated or unsaturated, mono-, bi- or tricyclic, (C.sub.1-C.sub.10) alkyl, (C.sub.2-C.sub.10)alkenyl, C.sub.1-C.sub.6)alkoxy C.sub.3-C.sub.12 cycloalkyl CH.sub.2(halo), --CH(halo).sub.2 or --C(halo).sub.3, heterocyclic such as heterocycloalkyl structure. The proline ring together with the ring structure formed by the substituents R.sup.1 may advantageously be bi- or tri-cyclic or of a higher annealed order.
[0076] Preferred embodiments of the process according to the invention employ compounds according to formula II with the structure according to general formula V, or the opposite enantiomer:
##STR00006##
according to formula VI, or the opposite enantiomer:
##STR00007##
according to formula VII, or the opposite enantiomer:
##STR00008##
[0077] R.sup.2 preferably represents a hydrogen atom, a (lower) alkyl group, preferably comprising from 1 to 4 carbon atoms, a lower alkyl group substituted by halogen, a cycloalkyl group, a (lower) alkoxy group, a (lower) thioalkyl group, a cycloalkyloxy group, an aralkyloxy group or an alkanoyl group; a hydroxyl group which may be protected or substituted, a nitro group, a formyl group, an amino group which may be protected or substituted, a cycloalkyloxy, aralkyloxy, alkanoyl, ureido or mono-, di- or tricyclic heterocyclic group, all of which groups may optionally be substituted.
[0078] R.sup.3 preferably represents a hydrogen atom, a lower alkyl group comprising from 1 to 4 carbon atoms, a lower alkyl group substituted by halogen, a cycloalkyl group, an aryl group, a lower alkoxy group, a lower thioalkyl group, a cycloalkyloxy group, an aralkyloxy group or an alkanoyl group; a hydroxyl group, a nitro group, a formyl group, an amino group which may be protected or substituted, a cycloalkyloxy, aralkyloxy, alkanoyl, ureido or mono-, di- or tricyclic heterocyclic group, all of which groups may optionally be substituted.
[0079] More preferably, R.sup.3 represents a compound according to general formula VIII:
##STR00009##
wherein R.sup.a and R.sup.b each independently represents a hydrogen atom, a (lower) alkyl group preferably comprising from 1 to 4 carbon atoms, a (lower) alkyl group substituted by halogen such as a --CH.sub.2(halo), a --CH(halo).sub.2 or a --C(halo).sub.3 group, a cycloalkyl group, an aryl group, a lower alkoxy group, a lower thioalkyl group, a cycloalkyloxy group, an aralkyloxy group or an alkanoyl group; a hydroxyl group, a nitro group, a formyl group, an amino group which may be protected or substituted, a cycloalkyloxy, aralkyloxy, alkanoyl, ureido or mono-, di- or tricyclic heterocyclic group, all of which groups may optionally be substituted.
[0080] R.sup.a preferably represents a branched alkyl group, more preferably a C.sub.3 or C.sub.4 alkyl group, and most preferably a tertiary butyl group.
[0081] R.sup.b preferably represents a (C.sub.1-C.sub.10) alkyl, (C.sub.2-C.sub.10)alkenyl, --(C.sub.1-C.sub.6)alkoxy, --(C.sub.3-C.sub.12)cycloalkyl, --CH.sub.2(halo), --CH(halo).sub.2 or --C(halo).sub.3, heterocyclic such as heterocycloalkyl group, or more preferably a N-tert-butyl amino group or a cyclohexyl-2-(pyrazine-2-carboxamido)acetamido) group, in particular for the synthesis of (S)-2-((S)-2-cyclohexyl-2-(pyrazine-2-carboxamido)acetamido)-3,3-dimethyl- butanoic acid.
[0082] R.sup.4 preferably represents a lower alkyl group comprising from 1 to 4 carbon atoms, a lower alkyl group substituted by halogen, a cycloalkyl group, an aryl group, a lower alkoxy group, a lower thioalkyl group, a cycloalkyloxy group, an aralkyloxy group or an alkanoyl group; a hydroxyl group, a nitro group, a formyl group, an amino group which may be protected or substituted, a cycloalkyloxy, aralkyloxy, alkanoyl, ureido or mono-, di- or tricyclic heterocyclic group, all of which groups may optionally be substituted.
[0083] In a preferred embodiment of the subject process, the compound according to general formula IV preferably has a structure according to general formula IX
##STR00010##
wherein R.sup.d, R.sup.e and R.sup.f each independently represents a hydrogen atom, a halogen atom, a substituted or unsubstituted alkyl, alkenyl, alkynyl, aromatic or non-aromatic, mono-, di- or tricyclic and/or a heterocyclic group.
[0084] Such compounds may advantageously be prepared from precursor compounds of the general formula X by dehydration under suitable conditions.
##STR00011##
Accordingly the present process further comprises:
[0085] A1) reacting a compound of the general formula XI:
##STR00012##
with a compound of the formula XII:
R.sup.e--COOH (XII),
and a compound of the general formula XIII
R.sup.f--NC (XIII)
under such conditions that compound X is formed,
[0086] wherein R.sup.d represents a hydrogen atom, a substituted or unsubstituted alkyl, alkenyl, alkynyl, aromatic or non-aromatic, mono-, di-polycyclic or alkylcycloalkyl, or a heterocyclic structure,
[0087] R.sup.e represents a substituted or unsubstituted alkyl, alkenyl, alkynyl, aromatic or non-aromatic, mono-, di- or tricyclic, or heterocyclic structure, and
[0088] R.sup.f represents a hydrogen atom, a substituted or unsubstituted alkyl, alkenyl, or alkynyl structure.
[0089] Preferably, compound XIII thus obtained is subsequently isolated from the reaction mixture.
[0090] R.sup.d preferably represents a hydrogen, a straight chain alkyl, a branched chain alkyl, a cycloalkyl, an alkylene-cycloalkyl, an aryl, alkylene-aryl, SO.sub.2-alkyl, SO.sub.2-aryl, alkylene-SO.sub.2-aryl, -alkylene-SO.sub.2-alkyl, heterocyclyl or alkylene-heterocyclyl; CH.sub.2CO--X--H, --CH.sub.2CO--X-straight chain alkyl, --CH.sub.2CO--X-branched chain alkyl, --CH.sub.2CO--X-cycloalkyl, --CH.sub.2CO--X-alkylene-cycloalkyl, --CH.sub.2CO--X-aryl, --CH.sub.2CO--X-alkylene-aryl, --CH.sub.2CO--X-heterocyclyl, --CH.sub.2CO--X-alkylene-heterocyclyl or --CH.sub.2CO-aryl; wherein X represents O or NH.
[0091] R.sup.e preferably represents hydrogen, a straight chain alkyl, a branched chain alkyl, a cycloalkyl, an alkylene-cycloalkyl, an aryl, and/or alkylene-aryl.
[0092] R.sup.f preferably represents hydrogen, a straight chain alkyl, a branched chain alkyl, a cycloalkyl, an alkylene-cycloalkyl, an aryl, and/or alkylene-aryl.
[0093] In the present invention, the substituents R.sup.d to R.sup.f according to general formula (I) are defined as follows:
[0094] In a preferred embodiment of the subject invention, R.sup.d represents an alkyl group such as ethyl, or an alkylcycloalkyl group, such as ethylcyclopropyl or ethlycyclobutyl.
[0095] R.sup.e preferably is an acetate group, and R.sup.f preferably is a cyclopropyl group. The process according to the present invention further advantageously comprises a step c) of subjecting compound X to dehydrating conditions to obtain an isocyanate compound according to general formula IX as set out herein above.
[0096] This may advantageously be achieved for instance by treatment of the formamido compound (X) with phosgene, diphosgene (trichloromethylchloroformate) and/or triphosgene [bis(trichloromethyl) carbonate], under suitable conditions as known to a skilled person.
[0097] Preferably, the aldehyde according to formula XI is derived from an optionally substantially enantiomerically pure 2-substituted 2-amino-1-ethanol according to general formula XIV
##STR00013##
wherein R.sup.1 represents R.sup.d as defined herein above.
[0098] The aldehyde compound XI may advantageously be prepared from an substituted 2-amino-1-ethanol according to general formula XIV by A) N-formylation, and B) by a selective oxidation of the primary alcohol of the obtained N-formylated alcohol intermediate to an aldehyde.
[0099] This oxidation is advantageously performed by employing a Dess-Martin oxidation. In this way, the stereogenic centre and various substituents R.sup.d can be introduced from often commercially or synthetically readily available 2-aminoethanols. A so-called Dess-Martin oxidation employs the Dess-Martin Periodinane (DMP), a hypervalent iodine compound for the selective and very mild oxidation of alcohols to aldehydes or ketones, as disclosed for instance in Y. Yip, F. Victor, J. Lamar, R. Johnson, Q. M. Wang, J. I. Glass, N. Yumibe, M. Wakulchik, J. Munroe, S.-H Chen, Bioorg. Med. Chem. Lett. 2004, 14, 5007-5011.
[0100] The oxidation preferably may be performed in dichloromethane or chloroform at room temperature, and is usually complete within 0.5-2 hours. Products are easily separated from the iodo-compound by-product after basic work-up.
[0101] Preferably, the Dess-Martin oxidation according to the invention is performed in the presence of compound IV, in such a way that the aldehyde II that is formed during the Dess-Martin oxidation immediately reacts in a Passerini reaction with the acetic acid that is formed as a by-product of the Dess-Martin oxidation as carboxylic acid III and isocyanide IV. This has the tremendous benefit that the atomic efficiency of the reaction is increased, since the Dess-Martin Periodinane (DMP) also provides a reactant for the second stage of the reaction, i.e., the Passerini three-component reaction. In addition, the combination of two reaction steps in one pot is advantageous in terms of both time and resources (less solvent and manpower required, one workup and chromatography less, etc.).
In a preferred embodiment of the subject process, the compound according to formula V
##STR00014##
is reacted with a compound according to general formula XV:
##STR00015##
and a compound according to general formula XVI:
##STR00016##
Under conditions that allow formation of a compound according to formula XVII:
##STR00017##
After the reaction, compound XVII could be advantageously isolated from the reaction mixture.
[0102] The subject process further preferably comprises subjecting the compound according to formula XVII to a saponification reaction to remove the acetate from the secondary alcohol at the .alpha.-hydroxy-.beta.-amino acid structure.
[0103] The saponification preferably is carried out by contacting the compound according to formula XVII with a alkaline metal carbonate, preferably K.sub.2CO.sub.3 in a suitable solvent, to obtain a saponified alcohol product according to formula XIIa.
[0104] The released intermediate compound comprising the secondary alcohol is the subjected to a selective oxidation of the secondary alcohol to form compound XVIII,
##STR00018##
This compound, which also known as Telaprevir, could be prepared in higher yields and with higher efficiency than any previously disclosed processes. Furthermore, the chiral information used for the preparation was derived from readily available simple building blocks, making the process a highly effective approach to such prolyl dipeptides and similar peptidomimetics.
[0105] In a further preferred embodiment of the subject process, a compound according to general formula VII as defined herein above is reacted with an acid compound according to general formula XVII:
##STR00019##
and an isocyanide compound according to general formula XX:
##STR00020##
to obtain a compound according to general formula XXI
##STR00021##
which may advantageously be saponified to a secondary alcohol and subsequently oxidized to a ketone, thereby yielding, after removal under suitable conditions of the R.sup.2 group, a compound according to formula XXII:
##STR00022##
also known as Boceprevir.
[0106] The process according to the present invention advantageously permits to selectively produce the two diastereomers according to the general formula XXIIa:
##STR00023##
[0107] and according to the general formula XXIIb, respectively,
##STR00024##
The subject invention therefore also relates to a process wherein XXIIa or XXIIb are selectively prepared, and to the thus obtained compounds XXIIa or XXIIb.
[0108] Suitable solvents for the subject reaction are polar protic and aprotic organic solvents, including methanol, ethanol, 2-propanol and other alcohol solvent, tetrahydrofuran, 1,4-dioxane, acetonitrile, and/or mixtures of these solvents with water or less polar organic solvents, such as dichloromethane or chloroform.
[0109] The saponification or removal of the ester group through hydrolysis may be performed by any suitable method known to a skilled person. Preferably, it is carried out by contacting the obtained reaction product according to formula I with an alkaline metal carbonate, more preferably K.sub.2CO.sub.3 in a suitable solvent, to obtain a saponified alcohol product. The saponified alcohol product may then advantageously be oxidised selectively at the secondary alcohol function, preferably without affecting the other structures on the compound, to yield a ketone compound.
The selective oxidation is preferably carried out by contacting the saponified alcohol product with a suitable oxidant in a suitable solvent. Suitable solvents include dichloromethane, THF, ethyl acetate, DMSO. Suitable oxidants include hypervalent iodine reagents such as IBX, Dess-Martin periodinane, etc., or a combination of TEMPO and PhI(OAc).sub.2 or related reagents.
[0110] The compounds obtainable by the subject process may further advantageously be used as (asymmetric) organocatalysts for the addition of enolizable aldehydes to electrophiles such as (among others) nitroalkenes, .alpha.,.beta.-unsaturated carbonyl compounds (aldehydes, esters, amides), vinyl sulfones, and the like. The subject invention also advantageously relates to compounds XVII, XVIIa, XXIIa and XXIIb, asc curucal building blocks for prolyl dipeptides.
EXPERIMENTAL SECTION
[0111] The following non-limiting experiments illustrate the process according to the subject invention.
[0112] General Information
Starting materials and solvents were purchased from ABCR and Sigma-Aldrich and were used without treatment. 3-Azabicylo[3,3,0]octane hydrochloride was purchased from AK Scientific. (1R,2S,6R,7S)-4-methyl-4-azatricyclo[5.2.1.0.sup.2,6]dec-8-ene was prepared according to literature procedure..sup.1 Column chromatography was performed on silica gel. .sup.1H and .sup.13C NMR spectra were recorded on a Bruker Avance 400 (400.13 MHz for .sup.1H and 100.61 MHz for .sup.13C) or Bruker Avance 500 (500.23 MHz for .sup.1H and 125.78 MHz for .sup.13C) in CDCl.sub.3 and DMSO-d.sub.6. Chemical shifts are reported in 6 values (ppm) downfield from tetramethylsilane.
[0113] Electrospray Ionization (ESI) mass spectrometry was carried out using a Bruker micrOTOF-Q instrument in positive ion mode (capillary potential of 4500 V). GC-MS spectra were recorded on a Hewlett Packard HP 6890 equipped with a J & W Scientific; HP-1MS; 30 m.times.0.32 mm.times.0.25 .mu.m column (injector temp. 300.degree. C., oven temp. 100.degree. C. to 280.degree. C. at 5.degree. C./min, hold for 10 min., He 1.6 ml/min. and detector temp. 275.degree. C.) and a HP 5973 Mass Selective Detector. GC-FID analyses were performed on Agilent 6850 GC with a J & W Scientific; HP-1; 30 m.times.0.32 mm.times.0.25 .mu.m (injector temp. 300.degree. C., oven temp. 100.degree. C. to 280.degree. C. at 5.degree. C./min, hold for 10 min., He 1.6 ml/min. and detector temp. 275.degree. C.) and a Varian Chirasil-Dex CB; 25 m.times.0.25 mm.times.0.26 .mu.m column (inj. 250.degree. C., oven temp. 100.degree. C. to 180.degree. C. at 5.degree. C./min, hold for 10 min., He 1.6 ml/min. and detector temp. 275.degree. C.) equipped with a Gerstel Multipurpose sampler MPS2L. Normal phase HPLC was performed on Agilent systems. Normal phase HPLC system was equipped with a G1322A degasser, a G1311A quaternary pump, a G1329 autosampler unit, a G1315B diode array detector and a G1316A temperature controlled column compartment. Infrared (IR) spectra were recorded neat, and wavelengths are reported in cm.sup.-1. Optical rotations were measured with a sodium lamp and are reported as follows: [.alpha.].sub.D.sup.20 (c=g/100 mL, solvent). Methyl 3-isocyanopropionate was synthesized as reported previously..sup.3
[0114] General Procedure 1: Preparation of Optically Active Imines (3S,7R)-4 and 6.
Unless stated otherwise: imines were synthesised according to literature procedure.sup.2 with minor adjustments. 2.5 g of freeze-dried MAO-N D5 E. Coli were rehydrated for 30 min. in 20 ml of KPO.sub.4 buffer (100 mM, pH=8.0) at 37.degree. C. Subsequently 1 mmol amine ((3S,7R)-4 or 6) in 30 ml of KPO.sub.4 buffer (100 mM, pH=8.0) was prepared.
[0115] The pH of the solution was adjusted to 8.0 by addition of NaOH and then added to the rehydrated cells. After 16-17 h The reaction was stopped (conversions were >95%) and worked up. For workup the reaction mixture was centrifuged at 4000 rpm and 4.degree. C. until the supernatant had clarified (40-60 min.). The pH of the supernatant was then adjusted to 10-11 by addition of aq. NaOH and the supernatant was subsequently extracted with t-butyl methyl ether or dichloromethane (4.times.70 mL). The combined organic phases were dried with Na.sub.2SO.sub.4 and concentrated at the rotary evaporator.
[0116] General Procedure 2: Preparation of Optically Active Ugi-Type Products 5a-g & 7a-g
Unless stated otherwise: Imine (0.70 mmol) was dissolved in 2 ml of CH.sub.2Cl.sub.2 followed by the addition of carboxylic acid (0.93 mmol) and isocyanide (0.93 mmol). The reaction mixture was stirred for 24 h at RT. CH.sub.2Cl.sub.2 (8 mL) was added and the resulting mixture was washed with Na.sub.2CO.sub.3 (2.times.10 mL), dried (MgSO.sub.4), filtered, and concentrated. Note: rotamers could be observed in the NMR data.
General Procedure 3: Determination of Enantiomeric Excess (ee) and Diastereomeric Ratio (d.r.)
[0117] Racemic imines were synthesised according to literature procedure.sup.2. Racemic Ugi-type products were prepared according to general procedure 3. Diastereomers could be separated by GC-MS and GC-FID. Enantiomers could be separated by normal phase HPLC and GC-FID.
Example 1
##STR00025##
[0118] Compound 5a:
[0119] General procedure 2 was followed using 3-azabicyclo[3,3,0]oct-2-ene ((3S,7R)-4, 76 mg, 0.70 mmol), acetic acid (55 mg, 52 .mu.l, 0.91 mmol) and t-butyl isocyanide (76 mg, 103 .mu.l, 0.91 mmol) giving 5a as a white solid, yield 73%.
[0120] 93:7 d.r. [HP-1, t (major)=14.852 min, t (minor)=16.773 min]; 95% ee [CP Chirasil-DEX CB, t (minor)=20.449 min, t (major)=20.860 min]; [.alpha.].sub.D.sup.20=-47.8.degree. (c=0.34, MeCN). .sup.1H NMR (400.1 MHz, CDCl.sub.3): .delta. 6.58 (bs, 1H), 4.28 (d, J=2.1 Hz, 1H), 3.70 (dd, J=8.3, 10.6 Hz, 1H), 3.24 (dd, J=4.5, 10.6 Hz, 1H), 2.96-2.93 (m, 1H), 2.91-2.82 (m, 1H), 2.01 (s, 3H), 1.93-1.78 (m, 2H), 1.71-1.42 (m, 2H), 1.41-1.31 (m, 2H), 1.25 (s, 9H); .sup.13C NMR (100.6 MHz, CDCl.sub.3) .delta. 170.5, 170.0, 66.8, 54.4, 51.0, 45.0, 42.7, 32.5, 32.3, 28.7, 25.7, 22.6; IR (neat): .nu..sub.max (cm.sup.-1)=3277 (m), 2957 (m), 1668 (s), 1630 (s), 1549 (s), 1447 (s), 1420 (s), 1223 (s), 667 (m), 606 (m); HRMS (ESI+) calcd for C.sub.14H.sub.24N.sub.2O.sub.2 ([M+H].sup.+) 253.1916. found 253.1925.
Example 2
##STR00026##
[0121] Compound 5b:
[0122] General procedure 2 was followed using 3-azabicyclo[3,3,0]oct-2-ene ((3S,7R)-4, 76 mg, 0.70 mmol), benzoic acid (111 mg, 0.91 mmol) and t-butyl isocyanide (76 mg, 103 .mu.l, 0.91 mmol) giving 5b as a white solid, yield 73%.
[0123] 93:7 d.r. [HP-1, t (major)=23.672 min, t (minor)=25.601 min]; 95% ee [Daicel Chiralpak AD-H, hexane/2-propanol=96/4, v=1.0 mL/min.sup.1, .lamda.=254 nm, t (minor)=10.698 min, t (major)=11.620 min]; [.alpha.].sub.D.sup.20=-53.7.degree. (c=0.34, MeCN). .sup.1H NMR (400.1 MHz, CDCl.sub.3): .delta. 7.49-7.38 (m, 5H), 6.66 (bs, 1H), 4.54 (d, J=2.8 Hz), 3.72 (dd, J=11.4, 7.8 Hz, 1H), 3.23 (d, J=11.0, 1H), 3.15-3.10 (m, 1H), 2.73-2.58 (m, 1H), 1.96-1.82 (m, 1H), 1.82-1.69 (m, 1H), 1.68-1.41 (m, 3H), 3.15-3.10 (m, 1H), 1.28 (s, 9H), 1.24-1.06 (m, 1H); .sup.13C NMR (100.6 MHz, CDCl.sub.3) .delta.170.3, 170.1, 136.3, 130.1, 128.4, 126.9, 67.1, 60.4, 55.9, 51.1, 44.2, 43.3, 33.0, 32.7, 28.7, 26.2; IR (neat): .nu..sub.max (cm.sup.-1)=3310 (m), 2961 (m), 1674 (s), 1618 (s), 1416 (s), 1223 (s), 698 (s); HRMS (ESI+) calcd for C.sub.19H.sub.26N.sub.2O.sub.2 ([M+H].sup.+) 315.2073. found 315.2077.
Example 3
##STR00027##
[0124] Compound 5c:
[0125] General procedure 2 was followed using 3-azabicyclo[3,3,0]oct-2-ene ((3S,7R)-4, 76 mg, 0.70 mmol), 3-furoic acid (102 mg, 0.91 mmol) and isopropyl isocyanide (63 mg, 86 .mu.l, 0.91 mmol) giving 5c as a white solid, yield 75%.
[0126] 92:8 d.r. [HP-1, t (major)=21.290 min, t (minor)=23.012 min] 94% ee [Daicel Chiralpak AD-H, hexane/2-propanol=90/10, v=1.0 mLmin.sup.1, .lamda.=254 nm, t (minor)=7.417 min, t (major)=12.039 min]; [.alpha.].sub.D.sup.20=-33.3.degree. (c=0.30, MeCN). .sup.1H NMR (400.1 MHz, CDCl.sub.3): .delta. 7.80 (bs, 1H), 7.43 (bs, 1H), 6.72 (bs, 1H), 6.51 (d, J=6.3 Hz, 1H), 4.56 (d, J=2.3 Hz, 1H), 4.03 (oct, J=7.1 1H), 3.88 (dd, J=10.4, 8.3 Hz, 1H), 3.53 (dd, J=10.4, 3.8 Hz, 1H), 3.09-3.01 (m, 1H), 2.95-2.84 (m, 1H), 2.00-1.84 (m, 2H), 1.74-1.65 (m, 1H), 1.64-1.54 (m, 1H), 1.53-1.43 (m, 1H), 1.43-1.33 (m, 1H), 1.17 (d, J=6.3 Hz, 3H) 1.13 (d, J=6.3 Hz, 3H); .sup.13C NMR (100.6 MHz, CDCl.sub.3) .delta. 170.1, 163.2, 144.3, 142.8, 121.8, 110.4, 66.8, 54.8, 44.4, 43.3, 41.3, 32.4, 32.2, 25.6, 22.5, 22.4; IR (neat): .nu..sub.max(cm.sup.-1)=3281 (w), 2949 (w), 1647 (m), 1609 (s), 1547 (s), 1427 (s), 1159 (s), 737 (s), 598 (s); HRMS (ESI+) calcd for C.sub.16H.sub.22N.sub.2O.sub.3 ([M+H].sup.+) 291.1709. found 291.1721.
Example 4
##STR00028##
[0127] Compound 5d:
[0128] General procedure 2 was followed using 3-azabicyclo[3,3,0]oct-2-ene ((3S,7R)-4, 76 mg, 0.70 mmol), benzoic acid (111 mg, 0.91 mmol) and isopropyl isocyanide (63 mg, 86 .mu.l, 0.91 mmol) giving 5d as a white solid, yield 78%.
[0129] 92:8 d.r. [HP-1, t (major)=23.809 min, t (minor)=25.563 min]; 94% ee [Daicel Chiralpak AD-H, hexane/2-propanol=96/4, v=1.0 mL/min.sup.1, .lamda.=254 nm, t (minor)=16.613 min, t (major)=21.363 min]; [.alpha.].sub.D.sup.20=-52.4.degree. (c=0.42, MeCN). .sup.1H NMR (400.1 MHz, CDCl.sub.3): .delta. 7.46-7.36 (m, 1H), 6.63 (bs, 1H), 4.59 (d, J=2.0 Hz, 1H), 4.10-4.01 (m, 1H), 3.73 (dd, J=11.4, 7.8 Hz, 1H), 3.73 (dd, J=11.4, 7.8 Hz, 1H), 3.32-3.29 (m, 1H), 3.23-3.17 (m, 1H), 2.76-2.71 (m, 1H), 2.02-1.94 (m, 1H), 1.88-1.78 (m, 1H), 1.75-1.63 (m, 1H), 1.63-1.50 (m, 1H), 1.16 (d, J=6.6 Hz, 3H) 1.13 (d, J=6.6 Hz, 3H); .sup.13C NMR (100.6 MHz, CDCl.sub.3) .delta. 170.4, 170.0, 136.2, 130.1, 128.4, 126.9, 126.6, 66.5, 55.9, 44.3, 43.3, 41.5, 32.9, 32.6, 26.1, 22.7, 22.6; IR (neat): .nu..sub.max(cm.sup.-1)=3300 (m), 2959 (m), 1615 (s), 1545 (s), 1416 (s), 1229 (m), 700 (m); HRMS (ESI+) calcd for C.sub.18H.sub.24N.sub.2O.sub.2 ([M+H].sup.+) 301.1916. found 301.1914.
Example 6
##STR00029##
[0130] Compound 5e:
[0131] General procedure 2 was followed using 3-azabicyclo[3,3,0]oct-2-ene ((3S,7R)-4, 76 mg, 0.70 mmol), acetic acid (55 mg, 52 .mu.l, 0.91 mmol) and benzyl isocyanide (107 mg, 111 .mu.l, 0.91 mmol) giving 5e as a white solid, yield 71%.
[0132] 92:8 d.r. [HP-1, t (major)=25.098 min, t (minor)=26.457 min]; 94% ee [Daicel Chiralpak OJ-H, hexane/2-propanol=93/7, v=1.0 mLmin.sup.1, .lamda.=254 nm, t (minor)=9.948 min, t (major)=10.718 min]; [.alpha.].sub.D.sup.20=-18.8.degree. (c=0.32, MeCN). .sup.1H NMR (400.1 MHz, CDCl.sub.3): .delta. 7.36-7.25 (m, 5H), 7.16 (bs, 1H), 4.47 (d, J=2.0 Hz, 1H), 4.25 (dd, J=15.2, 5.8 Hz, 2H), 3.73 (dd, J=10.6, 8.3 Hz, 1H), 3.28 (dd, J=10.5, 4.5 Hz, 1H), 3.10-3.03 (m, 1H), 2.96-2.88 (m, 1H), 2.10 (s, 3H), 2.01-1.85 (m, 1H), 1.82-1.55 (m, 2H), 1.52-1.39 (m, 2H); .sup.13C NMR (100.6 MHz, CDCl.sub.3) .delta. 171.3, 170.1, 138.3, 128.5, 128.4, 127.9, 127.4, 127.0, 65.9, 54.3, 45.4, 43.2, 42.7, 32.6, 32.2, 25.5, 22.1; IR (neat): .nu..sub.max(cm.sup.-1)=3267 (m), 2951 (w), 1626 (s), 1537 (m), 1418 (s), 1231 (s), 1030 (w), 748 (s); HRMS (ESI+) calcd for C.sub.17H.sub.22N.sub.2O.sub.2 ([M+H].sup.+) 287.1760. found 281.1765.
Example 7
##STR00030##
[0133] Compound 5f:
[0134] General procedure 2 was followed using 3-azabicyclo[3,3,0]oct-2-ene ((3S,7R)-4, 76 mg, 0.70 mmol), benzoic acid (111 mg, 0.91 mmol) and benzyl isocyanide (107 mg, 111 .mu.l, 0.91 mmol) giving 5f as a white solid, yield 81%.
[0135] 92:8 d.r. [HP-1, t (major)=33.333 min, t (minor)=35.085 min]; 97% ee [Daicel Chiralpak AD-H, hexane/2-propanol=96/4, v=1.0 mL/min.sup.1, .lamda.=254 nm, t (minor)=18.134 min, t (major)=23.440 min]; [.alpha.].sub.D.sup.20=-52.6.degree. (c=0.38, MeCN). .sup.1H NMR (400.1 MHz, CDCl.sub.3): .delta. 7.34-7.11 (m, 10H), 6.73 (bs, 1H), 4.61 (d, J=2.8 Hz, 1H). 4.37 (dd, J=5.3, 2.8 Hz, 2H), 3.66 (dd, J=11.1, 7.6 Hz, 1H), 3.24 (dd, J=10.9, 1.8 Hz, 1H), 3.18-3.11 (m, 1H), 2.72-2.64 (m, 1H), 1.92-1.82 (m, 1H), 1.82-1.62 (m, 1H), 1.25-1.13 (m, 1H); .sup.13C NMR (100.6 MHz, CDCl.sub.3) .delta.171.1, 170.3, 138.3, 135.9, 132.6, 130.0, 129.7, 128.4, 128.21, 128.0, 127.7, 127.3, 127.0, 126.9, 126.4, 66.3, 55.8, 44.9, 43.2, 43.1, 32.8, 32.4, 25.9; IR (neat): .nu..sub.max(cm.sup.-1)=3262 (m), 2928 (m), 1674 (s), 1613 (s), 1545 (s), 1423 (s), 1223 (m), 698 (s), 669 (s); HRMS (ESI+) calcd for C.sub.22H.sub.24N.sub.2O.sub.2 ([M+H].sup.+) 349.1916. found 349.1924.
Example 8
##STR00031##
[0136] Compound 5g:
[0137] General procedure 2 was followed using 3-azabicyclo[3,3,0]oct-2-ene ((3S,7R)-4, 76 mg, 0.70 mmol), isobutyric acid (80 mg, 84 .mu.l, 0.91 mmol) and t-butyl isocyanide (76 mg, 103 .mu.l, 0.91 mmol) giving 5g as a white solid, yield 83%.
[0138] 93:7 d.r. [HP-1, t (major)=17.165 min, t (minor)=18.750 min]; 97% ee [CP Chirasil-DEX CB, t (minor)=21.439 min, t (major)=21.846 min]; [.alpha.].sub.D.sup.20=-47.8.degree. (c=0.34, MeCN). .sup.1H NMR (400.1 MHz, CDCl.sub.3): .delta. 6.71 (bs, 1H), 4.37 (d, J=1.8 Hz, 1H), 3.65 (dd, J=10.6, 8.3 Hz, 1H), 3.34 (dd, J=4.3, 10.9 Hz, 1H), 3.04-2.98 (m, 1H), 2.89-2.81 (m, 1H), 2.65 (sep, J=6.8 Hz, 1H), 2.01-1.83 (m, 2H), 1.70-1.48 (m, 2H), 1.45-1.35 (m, 2H), 1.29 (s, 9H), 1.13 (d, J=6.6 Hz, 3H), 1.10 (d, J=6.8 Hz, 3H); .sup.13C NMR (100.6 MHz, CDCl.sub.3) .delta.176.5, 170.4, 66.5, 53.0, 43.7, 42.7, 32.8, 32.3, 32.0, 30.8, 24.9, 18.9, 18.5; IR (neat): .nu..sub.max (cm.sup.-1)=3291 (m), 2963 (m), 2870 (w), 1684 (s), 1618 (s), 1551 (s), 1433 (s), 1225 (s), 1090 (m), 658 (m); HRMS (ESI+) calcd for C.sub.14H.sub.24N.sub.2O.sub.2 ([M+H].sup.+) 281.2229. found 281.2235.
Example 9
##STR00032##
[0139] Compound 7a:
[0140] General procedure 2 was followed using (1R,2S,6R,7S)-4-methyl-4-azatricyclo[5.2.1.0.sup.2,6]dec-8-ene (6, 93 mg, 0.70 mmol), acetic acid (55 mg, 52 .mu.l, 0.91 mmol) and t-butyl isocyanide (76 mg, 103 .mu.l, 0.91 mmol) giving 7a as a white solid, yield 83%.
[0141] >99:1 d.r. (t (major)=18.179 min); >99% ee [Daicel Chiralpak AD-H, hexane/2-propanol=92/8, v=1.0 mLmin.sup.1, .lamda.=220 nm, t (major)=5.319 min, t (minor)=6.587 min]; [.alpha.].sub.D.sup.20=-24.0.degree. (c=0.25, MeCN). .sup.1H NMR (400.1 MHz, CDCl.sub.3): .delta. 6.65 (bs, 1H), 6.14-6.13 (m, 2H), 4.09 (d, J=2.0 Hz, 1H), 3.47 (dd, J=11.4, 8.6 Hz, 1H), 3.36-3.32 (m, 1H), 3.15 (dd, J=11.4, 2.0 Hz, 1H), 2.98-2.92 (m, 3H), 1.95 (s, 3H), 1.51-1.41 (m, 2H), 1.30 (s, 9H); .sup.13C NMR (100.6 MHz, CDCl.sub.3) .delta.170.6, 169.0, 135.4, 134.0, 62.9, 51.7, 51.0, 50.3, 47.0, 46.6, 46.0, 45.1, 28.7, 22.8; IR (neat): .nu..sub.max(cm.sup.-1)=3283 (w), 2970 (w), 2942 (w), 1647 (s), 1634 (s), 1553 (s), 1414 (s), 1223 (s), 733 (s); HRMS (ESI+) calcd for C.sub.16H.sub.24N.sub.2O.sub.2 ([M+H].sup.+) 277.1916. found 277.1922.
Example 10
##STR00033##
[0142] Compound 7b:
[0143] General procedure 2 was followed using 3-azabicyclo[3,3,0]oct-2-ene (6, 76 mg, 0.70 mmol), benzoic acid (111 mg, 0.91 mmol) and t-butyl isocyanide (76 mg, 103 .mu.l, 0.91 mmol) giving 7b as a white solid, yield 82%.
[0144] >99:1 d.r. (t (major)=26.830 min); HP-1, >99% ee [Daicel Chiralpak AD-H, hexane/2-propanol=96/4, v=1.0 mLmin.sup.1, .lamda.=220 nm, t (minor)=9.712 min, t (major)=11.741 min]; [.alpha.].sub.D.sup.20=-43.1.degree. (c=0.33, MeCN). .sup.1H NMR (400.1 MHz, CDCl.sub.3): .delta. 7.43-7.34 (m, 1H), 6.63 (bs, 1H), 6.20 (dd, J=5.8, 3.0 Hz, 1H), 5.91 (dd, J=5.6, 2.6 Hz, 1H), 4.43 (d, J=2.0 Hz, 1H), 3.52 (dd, J=11.9, 8.6 Hz, 1H), 3.44-3.39 (m, 1H), 3.05-3.00 (m, 2H), 2.91-2.85 (m, 1H), 2.78-2.76 (m, 1H), 1.48-1.45 (m, 1H), 1.40-1.37 (m, 1H), 1.32 (s, 9H); .sup.13C NMR (100.6 MHz, CDCl.sub.3) .delta.168.6, 168.0, 135.0, 133.2, 132.7, 128.4, 126.8, 124.9, 61.3, 50.4, 49.9, 49.5, 45.4, 44.9, 43.9, 43.3, 27.1; IR (neat): .nu..sub.max(cm.sup.-1)=3283 (m), 2970 (m), 2942 (m0, 1647 (s), 1634 (s), 1553 (s), 1414 (s), 1223 (s), 733 (s); HRMS (ESI+) calcd for C.sub.21H.sub.26N.sub.2O.sub.2 ([M+H].sup.+) 339.2073. found 339.2082.
Example 11
##STR00034##
[0145] Compound 7c:
[0146] General procedure 2 was followed using 1R,2S,6R,7S)-4-methyl-4-azatricyclo[5.2.1.0.sup.2,6]dec-8-ene (6, 93 mg, 0.70 mmol), 3-furoic acid (102 mg, 0.91 mmol) and isopropyl isocyanide (63 mg, 86 .mu.l, 0.91 mmol) giving 7c as a white solid, yield 75%.
[0147] >99:1 d.r. (t (major)=24.364 min); >99% ee [Daicel Chiralpak AD-H, hexane/2-propanol=90/10, v=1.0 mLmin.sup.1, .lamda.=220 nm, t (minor)=8.404 min, t (major)=9.968 min]; [.alpha.].sub.D.sup.20=-35.7.degree. (c=0.28, MeCN). .sup.1H NMR (400.1 MHz, CDCl.sub.3): .delta. 7.71 (dd, J=1.5, 0.8 Hz, 1H), 7.42 (dd, J=2.0, 1.5 Hz, 1H), 6.65 (dd, J=1.8, 0.8 Hz, 1H), 6.50 (d, J=6.6 Hz, 1H), 6.19-6.17 (m, 1H), 5.98-5.96 (m, 1H), 4.42 (d, J=2.0 Hz, 1H), 4.06-3.94 (m, 1H), 3.63 (dd, J=11.4, 8.8 Hz, 1H), 3.43-3.39 (m, 2H), 3-06-3.02 (m, 2H), 2.90-2.88 (m, 1H), 1.51-1.43 (m, 2H), 1.13 (d, J=6.6 Hz, 3H), 1.10 (d, J=6.6 Hz, 3H); .sup.13C NMR (100.6 MHz, CDCl.sub.3) .delta. 170.2, 162.7, 144.1, 142.9, 135.1, 134.4, 121.9, 110.4, 62.8, 51.6, 51.2, 47.1, 46.5, 45.8, 45.2, 41.5, 22.6, 22.7, 22.6; IR (neat): .nu..sub.max(cm.sup.-1)=3275 (m), 2970 (m), 2934 (m), 1678 (s), 1594 (s), 1545 (s), 1437 (s), 1219 (s), 1153 (m), 1018 (m) 874 (m0, 754 (s); HRMS (ESI+) calcd for C.sub.18H.sub.22N.sub.2O.sub.2 ([M+H].sup.+) 315.1709. found 315.1725.
Example 12
##STR00035##
[0148] Compound 7d:
[0149] General procedure 2 was followed using 1R,2S,6R,7S)-4-methyl-4-azatricyclo[5.2.1.0.sup.2,6]dec-8-ene (6, 93 mg, 0.70 mmol), benzoic acid (111 mg, 0.91 mmol) and isopropyl isocyanide (63 mg, 86 .mu.l, 0.91 mmol) giving 7d as a white solid, yield 78%.
[0150] >99:1 d.r. (t (major)=27.054 min); >99% ee [Daicel Chiralpak AD-H, hexane/2-propanol=96/4, v=1.0 mLmin.sup.1, .lamda.=220 nm, t (minor)=17.354 min, t (major)=29.404 min]; [.alpha.].sub.D.sup.20=-38.7.degree. (c=0.31, MeCN). .sup.1H NMR (400.1 MHz, CDCl.sub.3): .delta. 7.43-7.35 (m, 5H), 6.59 (d, J=7.6 Hz, 1H), 6.23-6.21 (m, 1H), 5.93-5.91 (m, 1H), 4.48 (d, J=1.7 Hz, 1H), 4.11-3.98 (m, 1H), 3.55 (dd, J=11.9, 8.8 Hz, 1H), 3.48-3.45 (m, 1H), 3.07-3.00 (m, 2H), 2.92-2.87 (m, 1H), 2.81-2.77 (m, 1H), 1.48-1.39 (m, 2H), 1.14 (d, J=6.6 Hz, 3H), 1.10 (d, J=6.6 Hz, 3H); .sup.13C NMR (100.6 MHz, CDCl.sub.3) .delta. 170.1, 169.8, 136.5, 134.8, 134.4, 130.1, 128.4, 126.6, 62.3, 52.0, 51.5, 47.0, 46.5, 45.6, 44.9, 41.5, 22.7, 22.6; IR (neat): .nu..sub.max(cm.sup.-1)=3287 (m), 2967 (m), 2940 (m), 1682 (s), 1601 (s), 1539 (s), 1424 (s), 1217 (s), 739 (s), 664 (m); HRMS (ESI+) calcd for C.sub.20H.sub.24N.sub.2O.sub.2 ([M+H].sup.+) 325.1916. found 325.1919.
Example 13
##STR00036##
[0151] Compound 7e:
[0152] General procedure 2 was followed using 1R,2S,6R,7S)-4-methyl-4-azatricyclo[5.2.1.0.sup.2,6]dec-8-ene (6, 93 mg, 0.70 mmol), acetic acid (55 mg, 52 .mu.l, 0.91 mmol) and benzyl isocyanide (107 mg, 111 .mu.l, 0.91 mmol) giving 7e as a white solid, yield 78%.
[0153] >99:1 d.r. (t (major)=28.213 min); >99% ee [Daicel Chiralpak OJ-H, hexane/2-propanol=92/8, v=1.0 mLmin.sup.1, .lamda.=220 nm, t (minor)=9.100 min, t (major)=10.760 min]; [.alpha.].sub.D.sup.20=-21.4.degree. (c=0.28, MeCN). .sup.1H NMR (400.1 MHz, CDCl.sub.3): .delta. 7.31-7.21 (m, 5H), 6.13-6.08 (m, 2H), 4.46 (dd, J=14.9, 6.1 Hz, 1H), 4.32 (dd, J=15.2, 5.8 Hz, 1H), 4.25 (d. J=2.0 Hz, 1H), 3.47-3.43 (m, 2H), 3.18 (dd, J=11.4, 2.0 Hz, 1H), 3.01-2.95 (m, 3H), 2.0 (s, 3H), 1.54-1.43 (m, 2H); .sup.13C NMR (100.6 MHz, CDCl.sub.3) 171.3, 169.3, 138.3, 135.4, 134.1, 128.6, 127.4, 127.3, 62.2, 51.7, 50.3, 47.1, 46.7, 46.0, 45.1, 43.4, 22.7; IR (neat): .nu..sub.max(cm.sup.-1)=3314 (w), 3082 (w), 2970 (w), 2932 (w), 1553 (s), 1433 (s), 1360 (m), 1317 (m), 1233 (m), 745 (s), 696 (s); HRMS (ESI+) calcd for C.sub.19H.sub.22N.sub.2O.sub.2 ([M+H].sup.+) 311.1760. found 311.1745.
Example 14
##STR00037##
[0154] Compound 7f:
[0155] General procedure 2 was followed using 31R,2S,6R,7S)-4-methyl-4-azatricyclo[5.2.1.0.sup.2,6]dec-8-ene (6, 93 mg, 0.70 mmol), benzoic acid (111 mg, 0.91 mmol) and benzyl isocyanide (107 mg, 111 .mu.l, 0.91 mmol) giving 7f as a white solid, yield 80%.
[0156] >99:1 d.r. (t (major)=36.331 min); >99% ee [Daicel Chiralpak OD-H, hexane/2-propanol=92/8, v=1.0 mLmin.sup.1, .lamda.=220 nm, t (major)=11.489 min, t (minor)=13.626 min]; [.alpha.].sub.D.sup.20=-35.1.degree. (c=0.29, MeCN). .sup.1H NMR (400.1 MHz, CDCl.sub.3): .delta. 7.44-7.17 (m, 10H), 6.24-6.18 (m, 1H), 5.95-5.93 (m, 1H), 4.60 (d, J=1.8 Hz, 1H), 4.45 (d, J=5.8 Hz, 2H), 3.58-3.50 (m, 2H), 3.10-3.04 (m, 2H), 2.95-2.89 (m, 1H), 2.83-2.79 (m, 1H), 1.50-1.41 (m, 2H); .sup.13C NMR (100.6 MHz, CDCl.sub.3) .delta. 171.1, 169.9, 138.4, 136.4, 134.4, 130.1, 128.6, 128.4, 127.4, 127.3, 126.4, 62.2, 52.1, 51.6, 47.0, 46.6, 45.6, 45.0, 43.5; IR (neat): .nu..sub.max (cm.sup.-1)=3268 (m), 3077 (w), 2972 (w), 2872 (w), 1684 (s), 1597 (s), 1560 (s), 1495 (m), 1431 (s), 1221 (s), 731 (s), 696 (s); HRMS (ESI+) calcd for C.sub.24H.sub.24N.sub.2O.sub.2 ([M+H].sup.+) 373.1916. found 373.1901.
Example 15
##STR00038##
[0157] Compound 7g:
[0158] General procedure 2 was followed using 1R,2S,6R,7S)-4-methyl-4-azatricyclo[5.2.1.0.sup.2,6]dec-8-ene (6, 93 mg, 0.70 mmol), isobutyric acid (80 mg, 84 .mu.l, 0.91 mmol) and t-butyl isocyanide (76 mg, 103 .mu.l, 0.91 mmol) giving 7g as a white solid, yield 81%.
[0159] >99:1 d.r. (t (major)=19.912 min); >99% ee [Daicel Chiralpak AD-H, hexane/2-propanol=95/5, v=1.0 mLmin.sup.1, .lamda.=220 nm, t (minor)=5.037 min, t (major)=6.877 min]; [.alpha.].sub.D.sup.20=-35.3.degree. (c=0.34, MeCN). .sup.1H NMR (400.1 MHz, CDCl.sub.3): .delta. 6.13-6.08 (m, 2H), 4.15 (d, J=1.8 Hz, 1H), 3.43 (dd, J=11.4, 8.6 Hz, 1H), 3.37-3.33 (m, 1H), 3.26 (dd, J=11.4, 2.0 Hz, 1H), 2.99-2.91 (m, 3H), 2.50 (sep, J=6.8 Hz, 1H), 1.54-1.38 (m, 2H), 1.27 (s, 9H), 1.06 (d, J=6.8 Hz, 3H), 1.03 (d, J=6.8 Hz, 3H); .sup.13C NMR (100.6 MHz, CDCl.sub.3) .delta. 175.6, 170.7, 135.3, 134.2, 62.7, 51.8, 50.8, 49.1, 47.01, 46.5, 45.2, 32.2, 28.7, 19.2, 18.3; IR (neat): .nu..sub.max (cm.sup.-1)=3325 (m), 2966 (m), 1678 (m), 1624 (s), 1553 (s), 1435 (s), 1315 (w), 1231 (m), 1088 (w); HRMS (ESI+) calcd for C.sub.18H.sub.28N.sub.2O.sub.2 ([M+H].sup.+) 305.2229. found 305.2224.
Example 16
##STR00039##
[0160] Compound 8a:
[0161] General procedure 2 was followed using 1R,2S,6R,7S)-4-methyl-4-azatricyclo[5.2.1.0.sup.2,6]dec-8-ene (6, 93 mg, 0.70 mmol), Fmoc-D-Pro-OH (307 mg, 0.91 mmol) and t-butyl isocyanide (76 mg, 103 .mu.l, 0.91 mmol). The crude product 8 was subjected using column chromatography (SiO.sub.2, EtOAc (1): cyclohexane (2)). Fmoc deprotection using 25% piperidine in DMF followed by column chromatography (CH.sub.2Cl.sub.2/MeOH 9:1) gave 8a as a white solid in 66% yield over two steps. [.alpha.].sub.D.sup.20=-75.0.degree. (c=0.16, MeCN). .sup.1H NMR (500.2 MHz, CDCl.sub.3): .delta. 6.75 (bs, 1H), 6.11 (d, J=5.7 Hz, 2H), 5.01 (bs, 1H), 4.20 (bs, 1H), 3.92-3.83 (m, 1H), 3.45-3.41 (m, 1H), 3.24-3.22 (m, 1H), 3.20-3.10 (m, 2H), 3.03-2.98 (m, 2H), 2.95-2.89 (m, 1H), 2.19-2.09 (m, 1H), 1.94-1.57 (m, 3H), 1.57-1.39 (m, 2H), 1.28 (s, 9H); .sup.13C NMR (125.8 MHz, CDCl.sub.3) .delta. 170.5, 135.4, 134.4, 63.7, 61.4, 51.6, 51.0, 49.01, 47.2, 46.9, 46.5, 46.3, 45.4, 30.0, 28.7, 25.9; IR (neat): .nu..sub.max (cm.sup.-1)=2960 (w), 1668 (s), 1622 (s), 1566 (m), 1414 (s), 1234 (m), 1094 (w), 853 (s); HRMS (ESI+) calcd for C.sub.19H.sub.29N.sub.3O.sub.2 ([M+H].sup.+) 332.2338. found 332.2342.
Example 18
##STR00040##
[0162] Compound 9:
[0163] General procedure 2 was followed using 1R,2S,6R,7S)-4-methyl-4-azatricyclo[5.2.1.0.sup.2,6]dec-8-ene (6, 93 mg, 0.70 mmol), Fmoc-L-Pro-OH (307 mg, 0.91 mmol) and t-butyl isocyanide (76 mg, 103 .mu.l, 0.91 mmol) giving 9 as a white solid, yield 76%.
[0164] [.alpha.].sub.D.sup.20=-6.7.degree. (c=0.60, MeCN). .sup.1H NMR (500.2 MHz, CDCl.sub.3): .delta. 7.76 (d, J=7.6 Hz, 2H), .delta. 7.59 (d, J=7.5 Hz, 1H), 7.55 (d, J=7.5 Hz, 1H), 7.40 (d, J=7.4 Hz, 2H), 7.40 (d, J=7.4 Hz, 2H), 7.31 (d, J=7.4 Hz, 2H),), 6.62 (bs, 1H), 6.19 (dd, J=5.6, 2.9 Hz, 1H), 6.11 (dd, J=5.6, 2.5 Hz, 1H), 4.35-4.33 (m, 1H), 4.30-4.28 (m, 2H), 4.24-4.22 (m, 1H), 4.20 (d, J=1.9 Hz, 1H), 3.72-3.58 (m, 2H), 3.35-3.32 (m, 1H), 3.25-3.22 (m, 2H), 2.93-2.90 (m, 2H), 2.18-2.13 (m, 2H), 1.95-1.91 (m, 2H), 1.52-1.40 (m, 2H), 1.29 (s, 9H);); .sup.13C NMR (125.8 MHz, CDCl.sub.3) .delta. 170.6, 170.4, 154.8, 143.9, 141.3, 135.9, 134.2, 127.8, 127.7, 127.1, 127.0, 125.1, 125.0, 120.0, 67.5, 63.3, 58.3, 51.8, 51.2, 49.4, 47.7, 47.4, 47.2, 47.1, 46.9, 44.4, 28.7, 28.6, 25.0; IR (neat): .nu..sub.max(cm.sup.-1)=2965 (w), 1643 (s), 1520 (w), 1449 (s), 1418 (s), 1358 (m), 1123 (m), 758 (m), 739 (s); HRMS (ESI+) calcd for C.sub.34H.sub.39N.sub.3O.sub.4 ([M+H].sup.+) 554.3019. found 554.3019.
Example 19
##STR00041##
[0165] Compound 10:
[0166] General procedure 2 was followed using 31R,2S,6R,7S)-4-methyl-4-azatricyclo[5.2.1.0.sup.2,6]dec-8-ene (6, 93 mg, 0.70 mmol), benzoic acid (111 mg, 0.91 mmol) and (R)-(+)-methylbenzyl isocyanide (119 mg, 123 .mu.l, 0.91 mmol) giving 10 as a white solid, yield 73%.
[0167] [.alpha.].sub.D.sup.20=-24.0.degree. (c=0.25, MeCN). .sup.1H NMR (500.2 MHz, CDCl.sub.3): .delta. 7.45-7.19 (m, 11H), 6.24 dd, J=5.7, 2.9 Hz, 1H), 5.93 dd, J=5.7, 2.9 Hz, 1H), 5.11-5.03 (m, 1H), 4.59 d, J=1.8 Hz, 1H), 3.53-3.49 (m, 1H), 3.40-3.35 (m, 1H), 3.02-2.97 (m, 2H), 2.88-2.81 (m, 1H), 2.77-2.75 (m, 1H), 1.48-1.39 (m, 2H), 1.45 (d, J=7.0 Hz, 2H); .sup.13C NMR (125.8 MHz, CDCl.sub.3): .delta. 169.8, 169.8, 144.0, 136.4, 134.8, 134.3, 130.0, 128.5, 128.4, 126.9, 126.4, 125.7, 62.0, 51.8, 51.5, 49.0, 47.0, 46.4, 45.5, 44.4, 22.7; IR (neat): .nu..sub.max (cm.sup.-1)=3302 (w), 3239 (w), 3059 (w), 2665 (w), 2929 (w), 1664 (s), 1559 (s), 1558 (s), 1427 (s), 1248 (m), 1020 (m), 698 (s), 667 (m); HRMS (ESI+) calcd for C.sub.25H.sub.26N.sub.2O.sub.2 ([M+H].sup.+) 387.2073. found 387.2067.
Example 20
##STR00042##
[0168] Compound 11:
[0169] General procedure 2 was followed using 31R,2S,6R,7S)-4-methyl-4-azatricyclo[5.2.1.0.sup.2,6]dec-8-ene (6, 93 mg, 0.70 mmol), benzoic acid (111 mg, 0.91 mmol) and (S)-(-)-methylbenzyl isocyanide (119 mg, 123 .mu.l, 0.91 mmol) giving 11 as a white solid, yield 70%.
[0170] [.alpha.].sub.D.sup.20=-78.6.degree. (c=0.28, MeCN). .sup.1H NMR (400.1 MHz, CDCl.sub.3): .delta. 7.43-7.16 (m, 11H), 6.24-6.19 (m, 1H), 5.97-5.90 (m, 1H), 5.10-5.01 (m, 1H), 4.52 (bs, 1H), 3.61-3.56 (m, 1H), 3.47-3.44 (m, 1H), 3.09-3.06 (m, 1H), 2.99 (bs, 1H), 2.93-2.87 (m, 1H), 2.80 (bs, 1H), 1.50-1.39 (m, 2H), 1.43 (d, J=6.8 Hz, 3H); .sup.13C NMR (125.8 MHz, CDCl.sub.3) 170.1, 169.9, 143.3, 136.5, 134.8, 134.4, 130.1, 128.7, 128.5, 127.2, 126.5, 126.1, 62.4, 52.1, 51.6, 49.1, 47.0, 46.5, 45.6, 44.9, 22.4; IR (neat): .nu..sub.max(cm.sup.-1)=3281 (m), 3281 (m), 2955 (m), 1638 (s), 1528 (s), 1397 (s), 1343 (m), 1227 (m), 1117 (m), 700 (s); HRMS (ESI+) calcd for C.sub.25H.sub.26N.sub.2O.sub.2 ([M+H].sup.+) 387.2073. found 387.2067.
Example 21
##STR00043##
[0171] Compound 12:
[0172] General procedure 2 was followed using 1R,2S,6R,7S)-4-methyl-4-azatricyclo[5.2.1.0.sup.2,6]dec-8-ene (6, 93 mg, 0.70 mmol), Fmoc-L-Pro-OH (307 mg, 0.91 mmol) and t-butyl isocyanide (76 mg, 103 .mu.l, 0.91 mmol). The crude product was purified by column chromatography (SiO.sub.2, EtOAc (1): cyclohexane (2)). Simultaneous Fmoc deprotection and saponification according to literature procedure.sup.4 followed by addition of 1.1 eq. TFA and purification using reversed phase chromatography (C.sub.18, H.sub.2O (1): EtOH (1)) gave 12 as a colorless solid, in 62% yield over two steps.
[0173] [.alpha.].sub.D.sup.20=-6.8.degree. (c=0.30, MeCN). .sup.1H NMR (500.2 MHz, DMSO): .delta. 12.43 (bs, 1H), 9.68-9.44 (m, 1H), 8.54 (bs, 1H), 8.19-8.14 (m, 1H), 6.29-6.25 (m, 1H), 6.13 (dd, J=5.7, 2.9 Hz, 1H), 3.95 (bs, 1H), 3.66-3.59 (m, 1H), 3.37-3.18 (m, 5H), 2.96 (bs, 2H), 2.79-2.73 (m, 1H), 2.50-2.38 (m, 4H), 1.97-1.85 (m, 2H), 1.69-1.57 (m, 2H), 1.46-1.35 (m, 2H); .sup.13C NMR (125.8 MHz, CDCl.sub.3) .delta. 172.7, 171.2, 165.8, 158.1, 157.9, 135.6, 134.8, 117.9, 115.5, 62.9, 58.1, 50.9, 49.6, 49.0, 46.5, 46.4, 45.8, 44.1, 34.6, 33.8, 27.9, 23.6; IR (neat): .nu..sub.max (cm.sup.-1)=2949 (w), 1640 (s), 1175 (s) 1130 (s), 833 (m), 719 (m); HRMS (ESI+) calcd for C.sub.20H.sub.26F.sub.3N.sub.3O.sub.3 ([M+H].sup.+) 347.1845. found 348.1929.
##STR00044##
Example 22
(S)-Methyl 2-cyclohexyl-2-(pyrazine-2-carboxamido)acetate (9)
[0174] Pyrazinecarboxylic acid (2.72 g, 21.9 mmol) was added to a solution of L-cyclohexylglycine methyl ester (4.13 g, 19.9 mmol) in CH.sub.2Cl.sub.2 (100 ml) at room temperature under N.sub.2, forming a white suspension. Triethylamine (6.33 ml, 4.62 g, 45.8 mmol) was added, followed by benzotriazol-1-yloxy-tris-(dimethylamino)-phosphonium hexafluorophosphate (BOP; 9.69 g, 21.9 mmol), which turned the reaction mixture from purple to an orange solution. After two days of stirring at room temperature the reaction mixture was washed two times with 50 ml saturated Na.sub.2CO.sub.3, followed by the washing of the aqueous layers with CH.sub.2Cl.sub.2 (2.times.50 ml). The organic layers were collected and dried with MgSO.sub.4, followed by concentration in vacuo. Purification by silica gel flash chromatography (c-Hex:EtOAc=2:1 with 0.5% triethylamine) afforded 9 (5.28 g, 19.03 mmol, 96%) as a yellow oil that solidified upon standing to give a white solid.
[0175] [.alpha.].sub.D.sup.20=+42.5 (c=1.13, CHCl.sub.3); .sup.1H NMR (250.13 MHz, CDCl.sub.3) .delta.=9.39 (d, J=1.25 Hz, 1H), 8.76 (d, J=2.5 Hz, 1H), 8.57 (t, J=1.5 Hz, 1H), 8.25 (d, J=8.8 Hz, 1H), 4.74 (dd, J=5.5, 9.3 Hz, 1H), 3.78 (s, 3H), 1.96 (m, 1H), 1.77 (m, 5H), 1.24 (m, 5H); .sup.13C NMR (62.90 MHz, CDCl.sub.3): .delta.=172.0 (C), 162.8 (C), 147.4 (CH), 144.5 (CH), 144.1 (C), 142.7 (CH), 57.0 (CH), 52.3 (CH.sub.3), 41.2 (CH), 29.7 (CH.sub.2), 28.4 (CH.sub.2), 26.0 (CH.sub.2); IR (neat): .nu..sub.max (cm.sup.-1)=3374 (m), 2920 (s), 2845 (w), 1740 (s), 1665 (s); HRMS (ESI, 4500 V): m/z calcd. for C.sub.14H.sub.19N.sub.3O.sub.3Na.sup.+ ([M+Na].sup.+) 300.1319. found 300.1319.
Example 23
##STR00045##
[0176] (S)-2-cyclohexyl-2-(pyrazine-2-carboxamido)acetic acid (10)
[0177] A solution of 1 M NaOH (12 ml, 12 mmol) was added to a solution of 9 (2.77 g, 10 mmol) in THF (25 ml) at 0.degree. C. MeOH was added to the formed suspension, to give a clear, colorless solution. The reaction mixture was stirred overnight at room temperature, followed by concentration in vacuo. The pH of the aqueous layer was set on 3.5 with a 1 M KHSO.sub.4 solution and was extracted with EtOAc (2.times.25 ml). The mixture was dried with Na.sub.2SO.sub.4, filtered, and concentrated in vacuo, to give 10 (2.49 g, 9.45 mmol, 95%) as a white solid.
[0178] [.alpha.].sub.D.sup.20=+50.9 (c=1.06, CHCl.sub.3); .sup.1H NMR (250.13 MHz, CDCl.sub.3): .delta.=9.38 (d, J=1.5 Hz, 1H), 8.78 (d, J=2.5 Hz, 1H), 8.58 (dd, J=1.5, 2.5 Hz, 1H), 8.27 (d, J=9.0, 1H), 4.77 (dd, J=4.3, 5.0 Hz, 1H), 2.00 (m, 1H), 1.76 (m, 5H), 1.37 (m, 5H); .sup.13C NMR (62.90 MHz, CDCl.sub.3): .delta.=175.7 (C), 163.0 (C), 147.2 (CH), 144.3 (CH), 144.2 (C), 142.0 (CH), 56.9 (CH), 40.9 (CH), 29.7 (CH.sub.2), 28.1 (CH.sub.2), 25.9 (CH.sub.2); IR (neat): .nu..sub.max (cm.sup.-1)=3383 (m), 2928 (s), 2852 (w), 1713 (m), 1676 (s), 1518 (s); HRMS (ESI, 4500 V): m/z calcd. For C.sub.13H.sub.17N.sub.3O.sub.3Na.sup.+ ([M+Na].sup.+) 286.1162. found 286.1158.
Example 23
##STR00046##
[0179] (S)-methyl 2-((S)-2-cyclohexyl-2-(pyrazine-2-carboxamido)-acetamido)-3,3-dimethylbut- anoate (11)
[0180] 10 (0.653 g, 4.5 mmol) was added to a solution of H-Tle-OMe (0.653 g, 4.5 mmol) in DMF (40 ml). 1-Ethyl-3-(3-dimethylaminopropyl)-carbodiimide-HCl (EDC-HCl; 0.919 g, 6.75 mmol) was added to this colorless solution followed by 1-hydroxy-7-azabenzotriazole (HOAt; 1.035 g, 5.4 mmol) giving a bright yellow solution. The reaction mixture was stirred for 3 days and afterwards concentrated in vacuo. The formed yellow solid was dissolved in EtOAc, washed with 40 ml saturated aqueous ammonium chloride solution and 40 ml of saturated aqueous NaHCO.sub.3 solution. The organic layers were collected, dried with MgSO.sub.4 and concentrated in vacuo to give 11 (1.48 g, 3.78 mmol, 84%) as a white solid.
[0181] [.alpha.].sub.D.sup.20=-2.0 (c=1.0, CHCl.sub.3); .sup.1H NMR (250.13 MHz, CDCl.sub.3): .delta.=9.39 (d, J=1.5 Hz, 1H), 8.76 (d, J=2.3 Hz, 1H), 8.55 (dd, J=2.4, 1.8 Hz, 1H), 8.29 (d, J=8.1, 1H), 6.40 (d, J=9.3 Hz, 1H), 4.46 (m, 2H), 3.74 (s, 3H), 1.81 (m, 1H), 1.76 (m, 4H), 1.24 (m, 6H), 0.96 (s, 12H); .sup.13C NMR (62.90 MHz, CDCl.sub.3): .delta.=171.7 (C), 170.4 (C), 163.0 (C), 147.5 (CH), 144.5 (CH), 144.2 (C), 142.7 (CH), 60.2 (CH.sub.3), 58.4 (CH), 51.9 (CH), 40.5 (CH), 31.7 (C), 29.7 (CH.sub.2), 28.7 (CH.sub.2), 26.6 (CH.sub.3), 25.9 (CH.sub.2); IR (neat): .nu..sub.max (cm.sup.-1)=3350 (m), 2928 (m), 2853 (w), 1738 (s), 1686 (s), 1640 (s), 1520 (s); HRMS (ESI, 4500 V): m/z calcd. for C.sub.20H.sub.30N.sub.4O.sub.4Na.sup.+ ([M+Na].sup.+) 413.2159. found 413.2169.
##STR00047##
Example 24
(S)-2-((S)-2-cyclohexyl-2-(pyrazine-2-carboxamido)acetamido)-3,3-dimethylb- utanoic acid (2)
[0182] A solution of 1 M NaOH (0.94 ml, 0.94 mmol) was added to a solution of 11 (0.31 g, 0.78 mmol) in THF (3 ml) at 0.degree. C. MeOH was added to the formed suspension, to give a clear and colourless solution. The reaction mixture was stirred overnight at room temperature, followed by concentration in vacuo. The pH of this aqueous layer was set to 3.5 with 1 M KHSO.sub.4 and subsequently extracted with EtOAc (2.times.10 ml). The mixture was dried with Na.sub.2SO.sub.4, filtered, and concentrated in vacuo, to give 2 (0.28 g, 0.75 mmol, 95%) as a white solid.
[0183] [.alpha.].sub.D.sup.20=+21.7 (c=1.015, CHCl.sub.3); .sup.1H NMR (250.13 MHz, CDCl.sub.3): .delta.=9.39 (d, J=1.3 Hz, 1H), 8.77 (d, J=2.5 Hz, 1H), 8.57 (dd, J=1.5, 2.5 Hz, 1H), 8.35 (d, J=9 Hz, 1H), 6.70 (d, J=9.0 Hz, 1H), 4.45 (t, J=8.8 Hz, 1H), 4.42 (d, J=9.2 Hz, 1H), 1.94 (m, 1H), 1.71 (m, 5H), 1.20 (m, 5H), 1.01 (s, 9H); .sup.13C NMR (62.90 MHz, CDCl.sub.3): .delta.=173.4 (C), 170.5 (C), 163.3 (C), 147.4 (CH), 144.4 (CH), 144.2 (C), 142.8 (CH), 58.4 (CH), 51.9 (CH), 40.4 (CH), 34.7 (C), 29.8 (CH.sub.2), 28.6 (CH.sub.2), 26.6 (CH.sub.3), 25.8 (CH.sub.2); IR (neat): .nu..sub.max (cm.sup.-1)=3335 (w), 2930 (m), 1726 (m), 1663 (s), 1514 (s); HRMS (ESI, 4500 V): m/z calc. for C.sub.19H.sub.29N.sub.4O.sub.4Na.sup.+ ([M+Na].sup.+) 399.2003. found 399.2013.
Example 25
##STR00048##
[0184] (S)-2-formamido-1-pentanol (12)
[0185] (S)-2-amino-1-pentanol (1.00 g, 9.7 mmol) was dissolved in ethylformate (7.84 ml, 7.19 g, 97 mmol). This reaction mixture was refluxed at 80.degree. C. for 4 hours, followed by stirring overnight at room temperature. The colourless solution was concentrated in vacuo and stirred for 1 hour in a 10 mol % K.sub.2CO.sub.3 in MeOH (25 ml). Afterwards, the pH was set to 7 with DOWEX 50wx8, followed by filtration and concentration in vacuo to give 12 (1.26 g, 9.61 mmol, 99%).
[0186] [.alpha.].sub.D.sup.20=-29.6 (c=1.15, CHCl.sub.3); .sup.1H NMR (250.13 MHz, CDCl.sub.3): .delta.=8.20 (s, 1H), 5.81 (bs, 1H), 4.04 (m, 1H), 2.11 (b, 1H), 1.47 (m, 4H), 0.94 (t, J=7.0 Hz, 3H); .sup.13C NMR (62.90 MHz, CDCl.sub.3): 161.8 (C), 65.1 (CH.sub.2), 50.6 (CH), 33.2 (CH.sub.2), 19.2 (CH.sub.2), 13.9 (CH.sub.3); IR (neat): .nu..sub.max (cm.sup.-1)=3248 (s), 2957 (m), 1651 (s), 1528 (m), 1381 (m); HRMS (ESI, 4500 V): m/z calcd. for C.sub.6H.sub.13NO.sub.2Na.sup.+ ([M+Na].sup.+) 154.0838. found 154.0835.
Example 26
##STR00049##
[0187] (S)-2-formamidopentanal
[0188] (7). Dess-Martin periodinane (5.514 g, 13 mmol) was added to a solution of (S)-2-formamido-1-pentanol (12, 1.31 g, 10 mmol) in CH.sub.2Cl.sub.2 (100 ml) at room temperature. The white suspension was stirred for 2 days and subsequently 35 ml MeOH was added and stirred for 30 minutes. The resulting suspension was filtrated and the filtrate was concentrated in vacuo. The crude product was purified by silica gel flash chromatography (cHex:EtOAc=1:4) to give 7 (1.08 g, 8.29 mmol, 83%) as a white solid. NMR analysis indicates that 7 is in equilibrium with its cyclic dimer.
[0189] [.alpha.].sub.D.sup.20=+37.6 (c=0.745, CHCl.sub.3); .sup.1H NMR assigned to the monomer (250.13 MHz, CDCl.sub.3): .delta.=8.22 (s, 1H), 7.84 (s, 1H), 7.10 (m, 1H), 5.31 (m, 1H), 1.52 (m, 4H), 0.95 (m, 3H); .sup.13C NMR assigned to the monomer (100.61 MHz, CDCl.sub.3): 198.8 (CH), 161.7 (CH), 57.4 (CH), 30.8 (CH.sub.2), 18.4 (CH.sub.2), 13.7 (CH.sub.3); .sup.1H NMR assigned to the dimer (400.13 MHz, CDCl.sub.3) 8.22 (s, 2H), 5.26 (m, 2H), 3.72 (m, 2H) 1.52 (m, 8H), 0.95 (m, 6H;) .sup.13C NMR (100.61 MHz, CDCl.sub.3) assigned to the dimer: 161.7 (CH), 89.8 (CH), 63.1 (CH), 30.8 (CH2), 18.4 (CH2), 13.7 (CH3); IR (neat): .nu..sub.max (cm.sup.-1): 3325 (s), 2959 (s), 1649 (s), 1530 (s), 1381 (m), 1123 (w); HRMS (ESI, 4500 V): m/z calc. for C.sub.6H.sub.12NO.sub.2.sup.+ ([M+H].sup.+) 130.0863. found 130.0858.
[0190] It was noted that the dimer exists as a mixture of diastereomers.
Example 27
##STR00050##
[0191] (3S)-2-acetoxy-N-cyclopropyl-3-formamidohexanoyl amide (13)
From 7:
[0192] Aldehyde 7 (0.892 g, 6.91 mmol) was added to a solution of cyclopropyl isocyanide (0.410 g, 6.12 mmol) in CH.sub.2Cl.sub.2 (110 ml) and stirred for 5 minutes at room temperature. Acetic acid (0.711 ml, 0.747 g, 12.44 mmol) was added and the yellow reaction mixture was stirred for 3 days at room temperature. The reaction mixture was washed twice with 100 ml saturated Na.sub.2CO.sub.3, followed by drying with Na.sub.2SO.sub.4 and concentration in vacuo. The crude was purified by silica gel flash chromatography (5% MeOH in CH.sub.2Cl.sub.2, 1% triethylamine). (3S)-2-acetoxy-N-cyclopropyl-3-formamidohexanoyl amide (0.99 g, 3.87 mmol, 56%) was obtained as a white solid as a 78:22 mixture of diastereomers.
From 12:
[0193] Dess Martin periodinane (5.66 g, 12.3 mmol) was added to a solution of (S)--N-(1 hydroxypentan-2-yl)formamide (1.15 g, 8.8 mmol) in CH.sub.2Cl.sub.2 (12 ml) at room temperature. The white suspension was stirred for 60 minutes and subsequently cyclopropyl isocyanide (0.74 g, 10.0 mmol) was added and stirred for 48 hours. The resulting suspension was filtrated and washed twice with 10 ml saturated Na.sub.2CO.sub.3, followed by drying with Na.sub.2SO.sub.4 and concentration in vacuo. The crude product was purified by silica gel flash chromatography (5% MeOH in CH.sub.2Cl.sub.2, 1% triethylamine) to give 13 (1.34 g, 5.22 mmol, 60%) as a pale yellow solid as a 78:22 mixture of diastereomers.
[0194] .sup.1H NMR (130.degree. C., 400.13 MHz, DMSO-d.sub.6): .delta.=8.03 (s, 1H), 7.52 (m, 1H), 7.30 (m, 1H), 4.89 (d, J=4.4, 1H), 4.28 (m, 1H), 2.65 (m, 1H), 2.17 (s, 3H), 1.27-1.47 (m, 4H), 0.89 (t, J=7.2, 3H), 0.63 (m, 2H), 0.48 (m, 2H); .sup.13C NMR (125.78 MHz, DMSO-d.sub.6): .delta.=169.8 (C), 168.5 (C), 160.6 (CH), 74.4 (CH), 47.5 (CH), 22.2 (CH), 18.4 (CH.sub.3), 13.6 (CH.sub.3), 5.7 (CH.sub.2); IR (neat): .nu..sub.max (cm.sup.-1) 3283 (s), 2961 (w), 1744 (m), 1661 (s), 1530 (s), 1238 (s); HRMS (ESI, 4500 V): m/z calcd. for C.sub.12H.sub.20N.sub.2O.sub.4Na.sup.+ ([M+Na].sup.+) 279.1315. found 279.1325.
Example 28
##STR00051##
[0195] (3S)-2-acetoxy-N-cyclopropyl-3-isocyano-hexanoyl amide (4)
[0196] N-methylmorpholine (0.57 ml, 0.562 g, 5.56 mmol) was added to a solution of (S)-1-(cyclopropylamino)-3-formamido-1-oxohexan-2-yl acetate (0.713 g, 2.78 mmol) in CH.sub.2Cl.sub.2 (40 ml) at room temperature. The reaction mixture was cooled to -78.degree. C. and triphosgene (0.289 g, 0.97 mmol) was quickly added and stirred for 5 minutes at this temperature. The resulting yellow solution was warmed up to -30.degree. C. and was stirred for another 3 h. Subsequently, the reaction was quenched with water and extracted twice with CH.sub.2Cl.sub.2 (40 ml). The organic layers were collected, dried with Na.sub.2SO.sub.4 and concentrated in vacuo. The crude product was purified by silica gel flash chromatography (2% MeOH in CH.sub.2Cl.sub.2) to give 4 (0.578 g, 2.42 mmol, 87%) as a white solid.
[0197] .sup.1H NMR (250.13 MHz, CDCl.sub.3): .delta.=6.28 (s, 1H), 5.25 (d, J=2.5 Hz, 1H), 4.2 (m, 1H), 2.74 (m, 1H), 2.24 (s, 3H), 1.55 (m, 4H), 0.96 (m, 3H), 0.84 (m, 2H), 0.60 (m, 2H); .sup.13C NMR (62.90 MHz, CDCl.sub.3): .delta.=169.7 (C), 168.3 (C), 74.4 (CH), 47.5 (CH), 22.0 (CH), 20.6 (CH.sub.3), 18.5 (CH.sub.2), 13.5 (CH.sub.3), 5.5 (CH.sub.2); IR (neat): .nu..sub.max (cm.sup.-1): 3267 (s), 2959 (m), 1745 (m), 1643 (s), 1512 (m), 1221 (s); HRMS (ESI, 4500 V): m/z calcd. for C.sub.12H.sub.18N.sub.2O.sub.3Na.sup.+ ([M+Na].sup.+) 261.1210. found 261.1214.
Example 29
##STR00052##
[0198] Compound 14.
[0199] Isocyanide 4 (0.549 g, 2.3 mmol) was dropwise added to a solution of imine 3 (0.252 g, 2.3 mmol) and carboxylic acid 2 (0.602 g, 1.60 mmol) in CH.sub.2Cl.sub.2 (5 ml) at room temperature. This yellow solution was stirred for 72 hours and afterwards diluted with 5 ml CH.sub.2Cl.sub.2. The reaction mixture was washed twice with saturated Na.sub.2CO.sub.3 solution (10 ml) and twice with saturated NH.sub.4Cl. The organic layers were collected, dried with MgSO.sub.4 and concentrated in vacuo. The crude product was purified by silica gel flash chromatography (5% MeOH in CH.sub.2Cl.sub.2) to give 14 (0.876 g, 1.21 mmol, 76%) as a mixture of diastereomers.
[0200] .sup.1H NMR (500.23 MHz, CDCl.sub.3): .delta.=9.50 (s, 1H), 8.75 (d, J=2.5, 1H), 8.59 (s, 1H), 8.35 (d, J=9.0, 1H), 6.84 (d, J=9.0, 1H), 6.44 (s, 1H), 5.20 (d, J=3.0, 1H), 4.74 (d, J=9.5, 1H), 4.58 (t, J=7.5, 1H), 4.38 (m, 1H), 3.37 (d, J=6.0, 1H), 2.82 (m, 1H), 2.69 (m, 1H), 2.11 (s, 3H), 1.26 (s, 2H), 0.97 (s, 9H), 0.86 (m, 3H), 0.84-2.00 (m, 21H), 0.76 (m, 2H), 0.51 (m, 2H); .sup.13C NMR (125.78 MHz, CDCl.sub.3): .delta.=170.5 (C), 169.3 (C), 162.9 (C), 147.4 (CH), 144.6 (CH), 144.2 (C), 142.8 (CH), 74.4 (CH), 66.6 (CH), 58.3 (CH), 56.6 (CH), 54.5 (CH.sub.2), 44.9 (CH), 43.0 (CH), 41.3 (CH), 35.5 (C), 26.4 (CH.sub.3), 20.8 (CH.sub.3), 19.1 (CH.sub.2), 13.8 (CH.sub.3), 6.6 (CH.sub.2); .nu..sub.max (cm.sup.-1): 3306 (m), 2928 (m), 2931 (m), 1743 (w), 1655 (s), 1520 (m), 1219 (m); HRMS (ESI, 4500 V): m/z calcd. for C.sub.38H.sub.57N.sub.7O.sub.7Na.sup.+ ([M+Na].sup.+) 746.4212. found 746.4107.
Example 30
##STR00053##
[0201] Telaprevir (1).
[0202] 250 .mu.l of saturated K.sub.2CO.sub.3 was added to a solution of 14 (0.514 g, 0.75 mmol) in MeOH (20 ml) at room temperature. The reaction mixture was stirred for 30 minutes at room temperature resulting in a pale yellow suspension. After full conversion (as judged by TLC analysis), the reaction mixture was washed with 20 ml brine, the aqueous layer was washed again with 10 ml CH.sub.2Cl.sub.2 (2.times.). The organic layers were collected, dried with MgSO.sub.4 and concentrated in vacuo, to yield a pale yellow solid. The yellow solid was dissolved in CH.sub.2Cl.sub.2 (10 ml) and Dess-Martin periodinane (0.650 g, 1.532 mmol) was added at room temperature. The reaction mixture was stirred overnight before adding saturated NaHCO.sub.3 solution (10 ml) and saturated Na.sub.2S.sub.2O.sub.3 solution (10 ml). This mixture was stirred for 10 minutes, separated and the aqueous layers were washed with EtOAc (2.times.10 ml). The organic layers were collected, dried with MgSO.sub.4 and concentrated in vacuo to give the crude product as an 83:13:4 mixture of diastereomers. After silica gel flash chromatography (1% MeOH in CH.sub.2Cl.sub.2), 1 (0.412 mg, 0.61 mmol, 80%) was obtained as a white solid.
[0203] .sup.1H NMR (500.23 MHz, DMSO-d.sub.6): .delta.=9.19 (d, J=1.4 Hz, 1H), 8.91 (d, J=24.5 Hz, 1H), 8.76 (dd, J=1.5, 2.5 Hz, 1H), 8.71 (d, J=5.3 Hz, 1H), 8.49 (d, J=9.2 Hz, 1H), 8.25 (d, J=6.8 Hz, 1H), 8.21 (d, J=8.9 Hz, 1H), 4.94 (m, 1H), 4.68 (dd, J=6.5, 9.0 Hz, 1H), 4.53 (d, J=9.0 Hz, 1H), 4.27 (d, J=3.5 Hz, 1H), 3.74 (dd, J=8.0, 10 Hz, 1H), 2.74 (m, 1H), 3.64 (d, J=3.5 Hz, 1H), 0.92 (s, 9H), 0.87 (t, 3H), 0.84-1.40 (m, 23H), 0.65 (m, 2H), 0.56 (m, 2H); .sup.13C NMR (125.78 MHz, CDCl.sub.3): .delta.=197.0 (C), 171.8 (C), 170.4 (C), 169.0 (C), 162.1 (C), 161.9 (C), 147.9 (CH), 144.0 (C), 143.4 (CH), 56.4 (CH), 56.3 (CH), 54.2 (CH), 53.4 (CH), 42.3 (CH), 41.3 (CH), 32.1 (CH), 31.8 (CH), 31.6 (CH), 29.1 (CH), 28.0 (CH), 26.4 (CH.sub.3); .nu..sub.max (cm.sup.-1): 3302 (m), 2928 (m), 2858 (w), 1658 (s), 1620 (s), 1561 (s), 1442 (m); HRMS (ESI, 4500 V): m/z calcd. for C.sub.36H.sub.53N.sub.7O.sub.6Na.sup.+ ([M+Na].sup.+) 702.3950. found 702.3941.
REFERENCES
[0204] 1. S. Michaelis, S. Blechert, Chem. Eur. J. 2007, 13, 2358-2368 [0205] 2. V. Kohler, K. R. Bailey, A. Znabet, J. Raftery, M. Helliwell, N. J. Turner, Angew. Chem. Int. Ed. 2010, 49, 2182-2184. [0206] 3. N. Elders, E. Ruijter, F. J. J. de Kanter, E. Janssen, M. Lutz, A. L. Spek, R. V. A. Orru Chem. Eur. J. 2009, 6096-6099. [0207] 4. V. Theodorou, K. Skobridis, A. G. Tzakos, V. Ragoussis Tetrahedron Lett. 2007, 48, 8230-8233.
[0208] While the present invention has been described with reference to specific preferred embodiments, it should be appreciated that variations are possible without departing from the scope of the invention. Therefore, the invention is not intended to be limited by the description in the specification but only by the language of the claims and equivalents thereof.
* * * * *






























































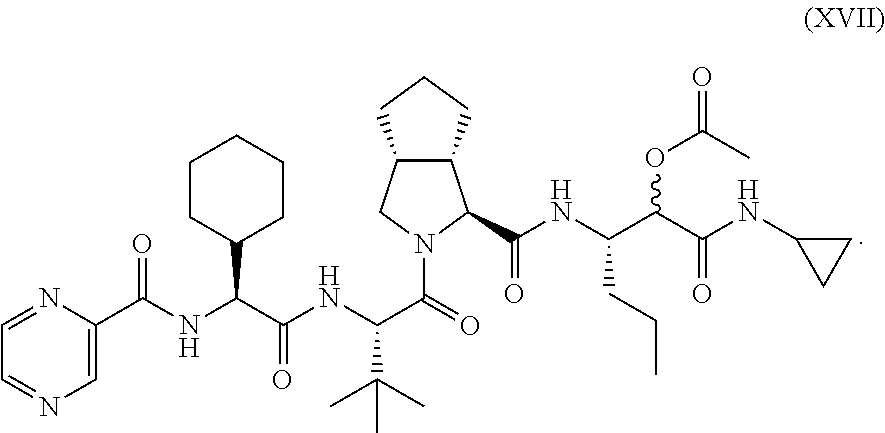

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.