Microorganisms for Producing 1,3-Butanediol and Methods Related Thereto
BURGARD; ANTHONY P. ; et al.
U.S. patent application number 13/528593 was filed with the patent office on 2012-12-27 for microorganisms for producing 1,3-butanediol and methods related thereto. This patent application is currently assigned to GENOMATICA, INC.. Invention is credited to ANTHONY P. BURGARD, MARK J. BURK, ROBIN E. OSTERHOUT, PRITI PHARKYA, JUN SUN.
| Application Number | 20120329113 13/528593 |
| Document ID | / |
| Family ID | 47362202 |
| Filed Date | 2012-12-27 |





View All Diagrams
| United States Patent Application | 20120329113 |
| Kind Code | A1 |
| BURGARD; ANTHONY P. ; et al. | December 27, 2012 |
Microorganisms for Producing 1,3-Butanediol and Methods Related Thereto
Abstract
Provided herein is a non-naturally occurring microbial organism having a 1,3-butanediol (1,3-BDO) pathway and comprising at least one exogenous nucleic acid encoding a 1,3-BDO pathway enzyme expressed in a sufficient amount to produce 1,3-BDO. In some embodiments, the pathway includes reducing equivalents from CO or hydrogen. In certain embodiments, a 1,3-BDO pathway proceeds by way of central metabolites pyruvate, succinate or alpha-ketoglutarate. Also provided herein is a method for producing 1,3-BDO, includes culturing such microbial organisms under conditions and for a sufficient period of time to produce 1,3-BDO.
| Inventors: | BURGARD; ANTHONY P.; (BELLEFONTE, PA) ; BURK; MARK J.; (SAN DIEGO, CA) ; OSTERHOUT; ROBIN E.; (SAN DIEGO, CA) ; SUN; JUN; (SAN DIEGO, CA) ; PHARKYA; PRITI; (SAN DIEGO, CA) |
| Assignee: | GENOMATICA, INC. SAN DIEGO CA |
| Family ID: | 47362202 |
| Appl. No.: | 13/528593 |
| Filed: | June 20, 2012 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 61500131 | Jun 22, 2011 | |||
| 61502702 | Jun 29, 2011 | |||
| Current U.S. Class: | 435/158 ; 435/252.33 |
| Current CPC Class: | C12N 15/52 20130101; C12P 7/18 20130101 |
| Class at Publication: | 435/158 ; 435/252.33 |
| International Class: | C12N 1/21 20060101 C12N001/21; C12P 7/18 20060101 C12P007/18 |
Claims
1. A non-naturally occurring microbial organism having a 1,3-butanediol pathway, wherein said microbial organism comprises at least one exogenous nucleic acid encoding a 1,3-butanediol pathway enzyme expressed in a sufficient amount to produce 1,3-butanediol; said non-naturally occurring microbial organism further comprising: (i) a reductive TCA pathway, wherein said microbial organism comprises at least one exogenous nucleic acid encoding a reductive TCA pathway enzyme selected from the group consisting of an ATP-citrate lyase, citrate lyase, a fumarate reductase, and an alpha-ketoglutarate:ferredoxin oxidoreductase; (ii) a reductive TCA pathway, wherein said microbial organism comprises at least one exogenous nucleic acid encoding a reductive TCA pathway enzyme selected from the group consisting of a pyruvate:ferredoxin oxidoreductase, a phosphoenolpyruvate carboxylase, a phosphoenolpyruvate carboxykinase, a CO dehydrogenase, and an H.sub.2 hydrogenase; or (iii) at least one exogenous nucleic acid encoding an enzyme selected from the group consisting of a CO dehydrogenase, an H.sub.2 hydrogenase, and combinations thereof; wherein said 1,3-butanediol pathway comprises a pathway selected from the group consisting of: (a) (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP dehydrogenase; (3) a 2-amino-4-hydroxypentanoate aminotransferase or oxidoreductase (deaminating); (4) a 2-oxo-4-hydroxypentanoate decarboxylase; and (5) a 3-hydroxybutyraldehyde reductase; (b) (1) an AKP thiolase; (2) an AKP aminotransferase or oxidoreductase (deaminating); (3) a 2,4-dioxopentanoate decarboxylase; (4) a 3-oxobutyraldehyde reductase (ketone reducing); and (5) a 3-hydroxybutyraldehyde reductase; (c) (1) an AKP thiolase; (2) an AKP aminotransferase or oxidoreductase (deaminating); (3) a 2,4-dioxopentanoate decarboxylase; (4) a 3-oxobutyraldehyde reductase (aldehyde reducing); and (5) a 4-hydroxy-2-butanone reductase; (d) (1) an AKP thiolase; (2) an AKP decarboxylase; (3) a 4-aminobutan-2-one aminotransferase or oxidoreductase (deaminating); (4) a 3-oxobutyraldehyde reductase (ketone reducing); and (5) a 3-hydroxybutyraldehyde reductase; (e) (1) an AKP thiolase; (2) an AKP decarboxylase; (3) a 4-aminobutan-2-one aminotransferase or oxidoreductase (deaminating); (4) a 3-oxobutyraldehyde reductase (aldehyde reducing); and (5) a 4-hydroxy-2-butanone reductase; (f) (1) an AKP thiolase; (2) an AKP decarboxylase; (3) a 4-aminobutan-2-one ammonia-lyase; (4) a butenone hydratase; and (5) a 4-hydroxy-2-butanone reductase; (g) (1) an AKP thiolase; (2) an AKP ammonia-lyase; (3) an acetylacrylate decarboxylase; (4) a butenone hydratase; and (5) a 4-hydroxy-2-butanone reductase; (h) (1) an acetoacetyl-CoA reductase (CoA-dependent, aldehyde forming); (2) a 3-oxobutyraldehyde reductase (ketone reducing); and (3) a 3-hydroxybutyraldehyde reductase; (i) an acetoacetyl-CoA reductase (CoA dependent, alcohol forming) and (2) a 4-hydroxy-2-butanone reductase; (j) (1) an acetoacetyl-CoA reductase (CoA-dependent, aldehyde forming); (2) a 3-oxobutyraldehyde reductase (aldehyde reducing); and (3) a 4-hydroxy-2-butanone reductase; (k) (1) an acetoacetyl-CoA reductase (ketone reducing) and (2) a 3-hydroxybutyryl-CoA reductase (alcohol forming); (l) (1) an acetoacetyl-CoA reductase (ketone reducing); (2) a 3-hydroxybutyryl-CoA reductase (aldehyde forming); and (3) a 3-hydroxybutyraldehyde reductase; (m) (1) a 4-hydroxybutyryl-CoA dehydratase; (2) a crotonase; and (3) a 3-hydroxybutyryl-CoA reductase (alcohol forming); and (n) (1) a 4-hydroxybutyryl-CoA dehydratase; (2) a crotonase; (3) a 3-hydroxybutyryl-CoA reductase (aldehyde forming); and (4) a 3-hydroxybutyraldehyde reductase; (o) (1) a succinyl-CoA transferase, a succinyl-CoA synthetase or a succinyl-CoA ligase, (2) a succinyl-CoA reductase (aldehyde forming), (3) a 4-hydroxybutyrate dehydrogenase, (4) a 4-hydroxybutyrate kinase, (5) a phosphotrans-4-hydroxybutyrylase, (6) a 4-hydroxybutyryl-CoA dehydratase, (7) a crotonase, (8) a 3-hydroxybutyryl-CoA reductase (aldehyde forming), and (9) a 3-hydroxybutanal reductase; (p) (1) (i) an alpha-ketoglutarate decarboxylase, or (ii) (a) a glutamate dehydrogenase and/or a glutamate transaminase, (b) a glutamate decarboxylase, and (c) a 4-aminobutyrate dehydrogenase and/or a 4-aminobutyrate transaminase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyrate kinase, (4) a phosphotrans-4-hydroxybutyrylase, (5) a 4-hydroxybutyryl-CoA dehydratase; (6) a crotonase, (7) a 3-hydroxybutyryl-CoA reductase (aldehyde forming), and (8) a 3-hydroxybutanal reductase; (q) (1) (i) an alpha-ketoglutarate decarboxylase, or (ii) (a) a glutamate dehydrogenase and/or a glutamate transaminase, (b) a glutamate decarboxylase, and (c) a 4-aminobutyrate dehydrogenase and/or a 4-aminobutyrate transaminase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (4) a 4-hydroxybutyryl-CoA dehydratase, (5) a crotonase, (6) a 3-hydroxybutyryl-CoA reductase (aldehyde forming), and (7) a 3-hydroxybutanal reductase; (r) (1) (i) an alpha-ketoglutarate decarboxylase, or (ii) (a) a glutamate dehydrogenase and/or a glutamate transaminase, (b) a glutamate decarboxylase, and (c) a 4-aminobutyrate dehydrogenase and/or a 4-aminobutyrate transaminase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyrate kinase, (4) a phosphotrans-4-hydroxybutyrylase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, and (7) a 3-hydroxybutyryl-CoA reductase (alcohol forming); (s) (1) (i) an alpha-ketoglutarate decarboxylase, or (ii) (a) a glutamate dehydrogenase and/or a glutamate transaminase, (b) a glutamate decarboxylase, and (c) a 4-aminobutyrate dehydrogenase and/or a 4-aminobutyrate transaminase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (4) a 4-hydroxybutyryl-CoA dehydratase, (5) a crotonase, and (6) a 3-hydroxybutyryl-CoA reductase (alcohol forming); (t) (1) (i) an alpha-ketoglutarate decarboxylase, or (ii) (a) a glutamate dehydrogenase and/or a glutamate transaminase, (b) a glutamate decarboxylase, and (c) a 4-aminobutyrate dehydrogenase and/or a 4-aminobutyrate transaminase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyrate kinase, (4) a phosphotrans-4-hydroxybutyrylase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, (7) a 3-hydroxybutyryl-CoA hydrolase, transferase or synthetase, and (8) a 3-hydroxybutyrate reductase; (u) (1) (i) an alpha-ketoglutarate decarboxylase, or (ii) (a) a glutamate dehydrogenase and/or a glutamate transaminase, (b) a glutamate decarboxylase, and (c) a 4-aminobutyrate dehydrogenase and/or a 4-aminobutyrate transaminase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (4) a 4-hydroxybutyryl-CoA dehydratase, (5) a crotonase, (6) a 3-hydroxybutyryl-CoA hydrolase, transferase or synthetase, and (7) a 3-hydroxybutyrate reductase. (v) (1) a succinate reductase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyrate kinase, (4) a phosphotrans-4-hydroxybutyrylase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, (7) a 3-hydroxybutyryl-CoA reductase (aldehyde forming), and (8) a 3-hydroxybutanal reductase; (w) (1) a succinate reductase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (4) a 4-hydroxybutyryl-CoA dehydratase, (5) a crotonase, (6) a 3-hydroxybutyryl-CoA reductase (aldehyde forming), (7) a 3-hydroxybutanal reductase; (x) (1) a succinate reductase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyrate kinase, (4) a phosphotrans-4-hydroxybutyrylase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, and (7) a 3-hydroxybutyryl-CoA reductase (alcohol forming); (y) (1) a succinate reductase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (4) a 4-hydroxybutyryl-CoA dehydratase, (5) a crotonase, and (6) a 3-hydroxybutyryl-CoA reductase (alcohol forming); (z) (1) a succinate reductase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyrate kinase, (4) a phosphotrans-4-hydroxybutyrylase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, (7) a 3-hydroxybutyryl-CoA hydrolase, transferase or synthetase, and (8) a 3-hydroxybutyrate reductase; (aa) (1) a succinate reductase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (4) a 4-hydroxybutyryl-CoA dehydratase, (5) a crotonase, (6) a 3-hydroxybutyryl-CoA hydrolase, transferase or synthetase, and (7) a 3-hydroxybutyrate reductase; (bb) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (aldehyde forming), (3) a 4-hydroxybutyrate dehydrogenase, (5) a 4-hydroxybutyrate kinase, (6) a pPhosphotrans-4-hydroxybutyrylase, (7) a 4-hydroxybutyryl-CoA dehydratase, (8) a crotonase, and (9) a 3-hydroxybutyryl-CoA reductase (alcohol forming); (cc) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (aldehyde forming), (3) a 4-hydroxybutyrate dehydrogenase, (4) a 4-hydroxybutyrate kinase, (5) a phosphotrans-4-hydroxybutyrylase, (6) a 4-hydroxybutyryl-CoA dehydratase, (7) a crotonase, (8) a 3-hydroxybutyryl-CoA hydrolase, transferase or synthetase, and (9) a 3-hydroxybutyrate reductase; (dd) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (alcohol forming), (3) a 4-hydroxybutyrate kinase, (4) a phosphotrans-4-hydroxybutyrylase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, (7) a 3-hydroxybutyryl-CoA reductase (aldehyde forming), and (8) a 3-hydroxybutanal reductase; (ee) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (alcohol forming), (3) a 4-hydroxybutyrate kinase, (4) a phosphotrans-4-hydroxybutyrylase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, and (7) a 3-hydroxybutyryl-CoA reductase (alcohol forming); (ff) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (alcohol forming), (3) a 4-hydroxybutyrate kinase, (4) a phosphotrans-4-hydroxybutyrylase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, (7) a 3-hydroxybutyryl-CoA hydrolase, transferase or synthetase, and (8) a 3-hydroxybutyrate reductase; (gg) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (aldehyde forming), (3) a 4-hydroxybutyrate dehydrogenase, (4) a 4-hydroxybutyryl-CoA transferase, or 4-hydroxybutyryl-CoA synthetase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, (7) a 3-hydroxybutyryl-CoA reductase (aldehyde forming), and (8) a 3-hydroxybutanal reductase; (hh) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (aldehyde forming), (3) a 4-hydroxybutyrate dehydrogenase, (4) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, and (7) a 3-hydroxybutyryl-CoA reductase (alcohol forming); (ii) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (aldehyde forming), (3) a 4-hydroxybutyrate dehydrogenase, (4) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, (7) a 3-hydroxybutyryl-CoA hydrolase, transferase or synthetase, (8) a 3-hydroxybutyrate reductase; (jj) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (alcohol forming), (3) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (4) a 4-hydroxybutyryl-CoA dehydratase, (5) a crotonase, (6) a 3-hydroxybutyryl-CoA reductase (aldehyde faulting), and (7) a 3-hydroxybutanal reductase; (kk) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (alcohol forming), (3) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (4) a 4-hydroxybutyryl-CoA dehydratase, (5) a crotonase, and (6) a 3-hydroxybutyryl-CoA reductase (alcohol forming); and (ll) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (alcohol forming), (3) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, (7) a 3-hydroxybutyryl-CoA hydrolase, transferase or synthetase, and (8) a 3-hydroxybutyrate reductase.
2. The non-naturally occurring microbial organism of claim 1, wherein said microbial organism further comprises an exogenous nucleic acid encoding an enzyme selected from the group consisting of a pyruvate:ferredoxin oxidoreductase, an aconitase, an isocitrate dehydrogenase, a succinyl-CoA synthetase, a succinyl-CoA transferase, a fumarase, a malate dehydrogenase, an acetate kinase, a phosphotransacetylase, an acetyl-CoA synthetase, an NAD(P)H:ferredoxin oxidoreductase, a ferredoxin, and combinations thereof.
3. The non-naturally occurring microbial organism of claim 1, wherein said microbial organism further comprises an exogenous nucleic acid encoding an enzyme selected from the group consisting of a succinyl-CoA synthetase, a succinyl-CoA transferase, a fumarase, a malate dehydrogenase, and combinations thereof.
4. The non-naturally occurring microbial organism of claim 1, wherein said microbial organism comprises two, three, four, five, six, seven, eight or nine exogenous nucleic acids, each encoding a 1,3-BDO pathway enzyme.
5. The non-naturally occurring microbial organism of claim 1, wherein said microbial organism comprises exogenous nucleic acids encoding each of the enzymes of at least one of the 1,3-butanediol pathways selected from the group consisting of (a)-(ll).
6. The non-naturally occurring microbial organism of claim 1, wherein said at least one exogenous nucleic acid is a heterologous nucleic acid.
7. The non-naturally occurring microbial organism of claim 1, wherein said non-naturally occurring microbial organism is in a substantially anaerobic culture medium.
8. A method for producing 1,3-BDO, comprising culturing a non-naturally occurring microbial organism according to claim 1, under conditions and for a sufficient period of time to produce 1,3-BDO.
9. The method of claim 8, wherein said microbial organism further comprises an exogenous nucleic acid encoding an enzyme selected from the group consisting of a pyruvate:ferredoxin oxidoreductase, an aconitase, an isocitrate dehydrogenase, a succinyl-CoA synthetase, a succinyl-CoA transferase, a fumarase, a malate dehydrogenase, an acetate kinase, a phosphotransacetylase, an acetyl-CoA synthetase, an NAD(P)H:ferredoxin oxidoreductase, ferredoxin, and combinations thereof.
10. The method of claim 8, wherein said microbial organism further comprises an exogenous nucleic acid encoding an enzyme selected from the group consisting of an aconitase, an isocitrate dehydrogenase, a succinyl-CoA synthetase, a succinyl-CoA transferase, a fumarase, a malate dehydrogenase, and combinations thereof.
11. The method of claim 8, wherein said microbial organism comprises two, three, four, five, six, seven, eight or nine exogenous nucleic acids, each encoding a 1,3-BDO pathway enzyme.
12. The method of claim 8, wherein said microbial organism comprises exogenous nucleic acids encoding each of the enzymes of at least one of the 1,3-butanediol pathways selected from the group consisting of (a)-(ll).
13. The method of claim 8, wherein said at least one exogenous nucleic acid is a heterologous nucleic acid.
14. The method of claim 8, wherein said non-naturally occurring microbial organism is in a substantially anaerobic culture medium.
Description
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] The present application claims the benefit of priority to U.S. Ser. Nos. 61/500,131, filed Jun. 22, 2011, and 61/502,702, filed Jun. 29, 2011, the contents of which are herein incorporated by reference in their entirety.
BACKGROUND
[0002] The present invention relates generally to biosynthetic processes and organisms capable of producing organic compounds. More specifically, the invention relates to non-naturally occurring organisms that can produce the commodity chemical 1,3-butanediol.
[0003] 1,3-butanediol (1,3-BDO) is a four carbon diol traditionally produced from acetylene via its hydration. The resulting acetaldehyde is then converted to 3-hydroxybutyraldehdye which is subsequently reduced to form 1,3-BDO. In more recent years, acetylene has been replaced by the less expensive ethylene as a source of acetaldehyde. 1,3-BDO is commonly used as an organic solvent for food flavoring agents. It is also used as a co-monomer for polyurethane and polyester resins and is widely employed as a hypoglycaemic agent. Optically active 1,3-BDO is a useful starting material for the synthesis of biologically active compounds and liquid crystals. A substantial commercial use of 1,3-butanediol is subsequent dehydration to afford 1,3-butadiene (Ichikawa et al., J. of Molecular Catalysis A-Chemical, 256:106-112 (2006); Ichikawa et al., J. of Molecular Catalysis A-Chemical, 231:181-189 (2005)), a 25 billion lb/yr petrochemical used to manufacture synthetic rubbers (e.g., tires), latex, and resins. The reliance on petroleum based feedstocks for either acetylene or ethylene warrants the development of a renewable feedstock based route to 1,3-butanediol and to butadiene.
[0004] Thus, there exists a need to develop microorganisms and methods of their use to produce 1,3-BDO. The present invention satisfies this need and provides related advantages as well.
SUMMARY
[0005] In some embodiments, the present invention is directed to a non-naturally occurring microbial organism that includes a microbial organism having a 1,3-butanediol (1,3-BDO) pathway having at least one exogenous nucleic acid encoding a 1,3-BDO pathway enzyme expressed in a sufficient amount to produce 1,3-BDO. The 1,3-BDO pathway includes an enzyme selected from the group consisting of a 2-amino-4-ketopentanoate (AKP) thiolase, an AKP dehydrogenase, a 2-amino-4-hydroxypentanoate aminotransferase, a 2-amino-4-hydroxypentanoate oxidoreductase (deaminating), a 2-oxo-4-hydroxypentanoate decarboxylase, a 3-hydroxybutyraldehyde reductase, an AKP aminotransferase, an AKP oxidoreductase (deaminating), a 2,4-dioxopentanoate decarboxylase, a 3-oxobutyraldehyde reductase (ketone reducing), a 3-oxobutyraldehyde reductase (aldehyde reducing), a 4-hydroxy-2-butanone reductase, an AKP decarboxylase, a 4-aminobutan-2-one aminotransferase, a 4-aminobutan-2-one oxidoreductase (deaminating), a 4-aminobutan-2-one ammonia-lyase, a butenone hydratase, an AKP ammonia-lyase, an acetylacrylate decarboxylase, an acetoacetyl-CoA reductase (CoA-dependent, aldehyde forming), an acetoacetyl-CoA reductase (CoA-dependent, alcohol forming), an acetoacetyl-CoA reductase (ketone reducing), a 3-hydroxybutyryl-CoA reductase (aldehyde forming), a 3-hydroxybutyryl-CoA reductase (alcohol forming), a 4-hydroxybutyryl-CoA dehydratase, and a crotonase.
[0006] In some embodiments, the present invention is directed to a method for producing 1,3-BDO that includes culturing such a non-naturally occurring microbial organism, under conditions and for a sufficient period of time to produce 1,3-BDO.
BRIEF DESCRIPTION OF THE DRAWINGS
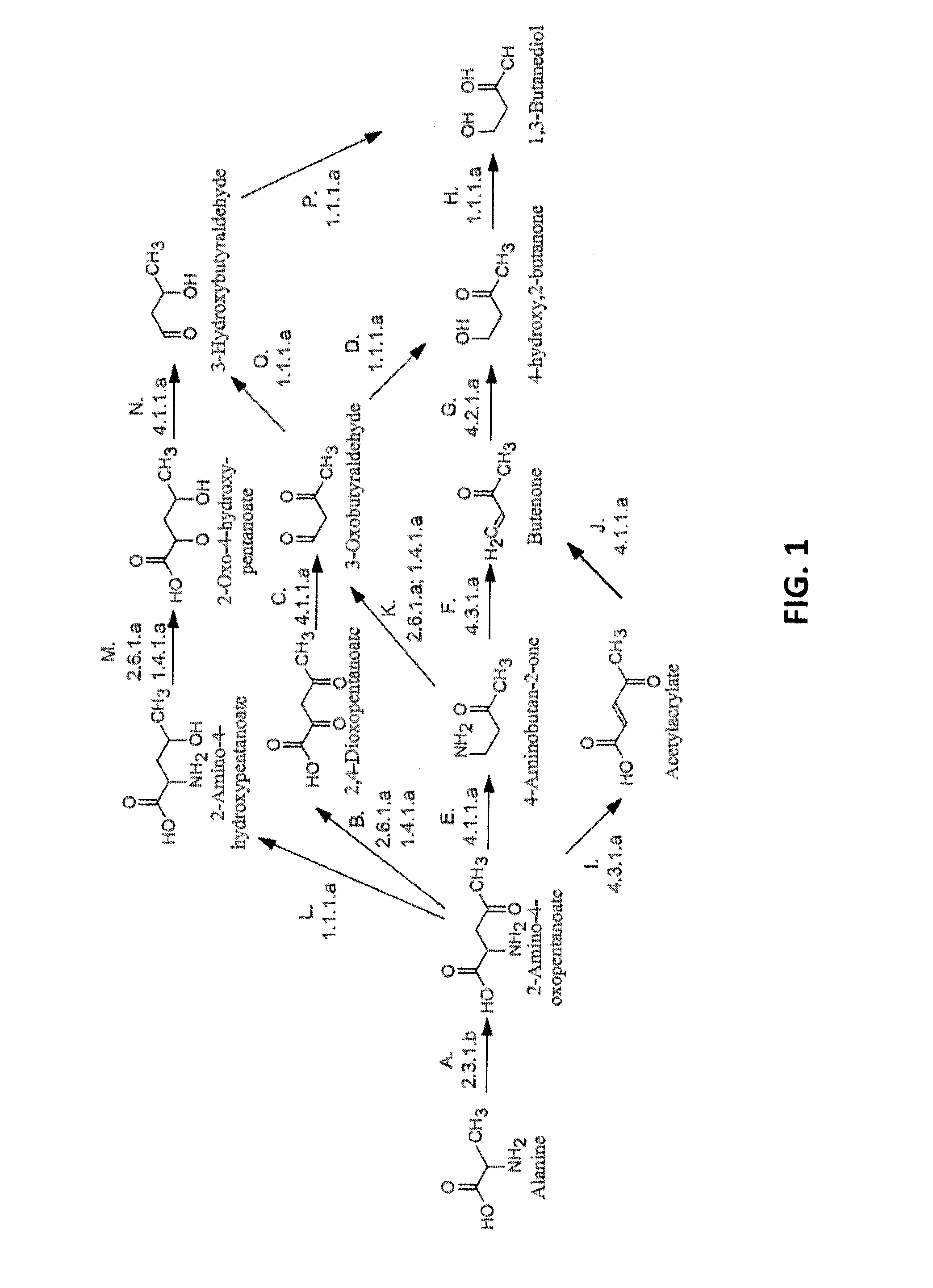
[0007] FIG. 1 shows pathways to 1,3-BDO from alanine. Enzymes are: A) AKP thiolase, B) AKP aminotransferase or AKP oxidoreductase (deaminating), C) 2,4-dioxopentanoate decarboxylase, D) 3-oxobutyraldehyde reductase (aldehyde reducing), E) AKP decarboxylase, F) 4-aminobutan-2-one ammonia-lyase, G) Butenone hydratase, H) 4-hydroxy,2-butanone reductase, I) AKP ammonia-lyase, J) acetylacrylate decarboxylase, K) 4-aminobutan-2-one aminotransferase or 4-aminobutan-2-one oxidoreductase (deaminating), L) AKP dehydrogenase, M) 2-amino-4-hydroxypentanoate aminotransferase or 2-amino-4-hydroxypentanoate oxidoreductase (deaminating), N) 2-oxo-4-hydroxypentanoate decarboxylase, O) 3-oxobutyraldehyde reductase (ketone reducing), and P) 3-hydroxybutyraldehdye reductase.
[0008] FIG. 2 shows pathways from acetoacetyl-CoA to 1,3-butanediol. Enzymes are: A) acetoacetyl-CoA reductase (CoA-dependent, aldehyde forming), B) 3-oxobutyraldehyde reductase (ketone reducing), C) 3-hydroxybutyraldehyde reductase, D) acetoacetyl-CoA reductase (CoA-dependent, alcohol forming), E) 3-oxobutyraldehyde reductase (aldehyde reducing), F) 4-hydroxy,2-butanone reductase, G) acetoacetyl-CoA reductase (ketone reducing), H) 3-hydroxybutyryl-CoA reductase (aldehyde forming), and I) 3-hydroxybutyryl-CoA reductase (alcohol forming).
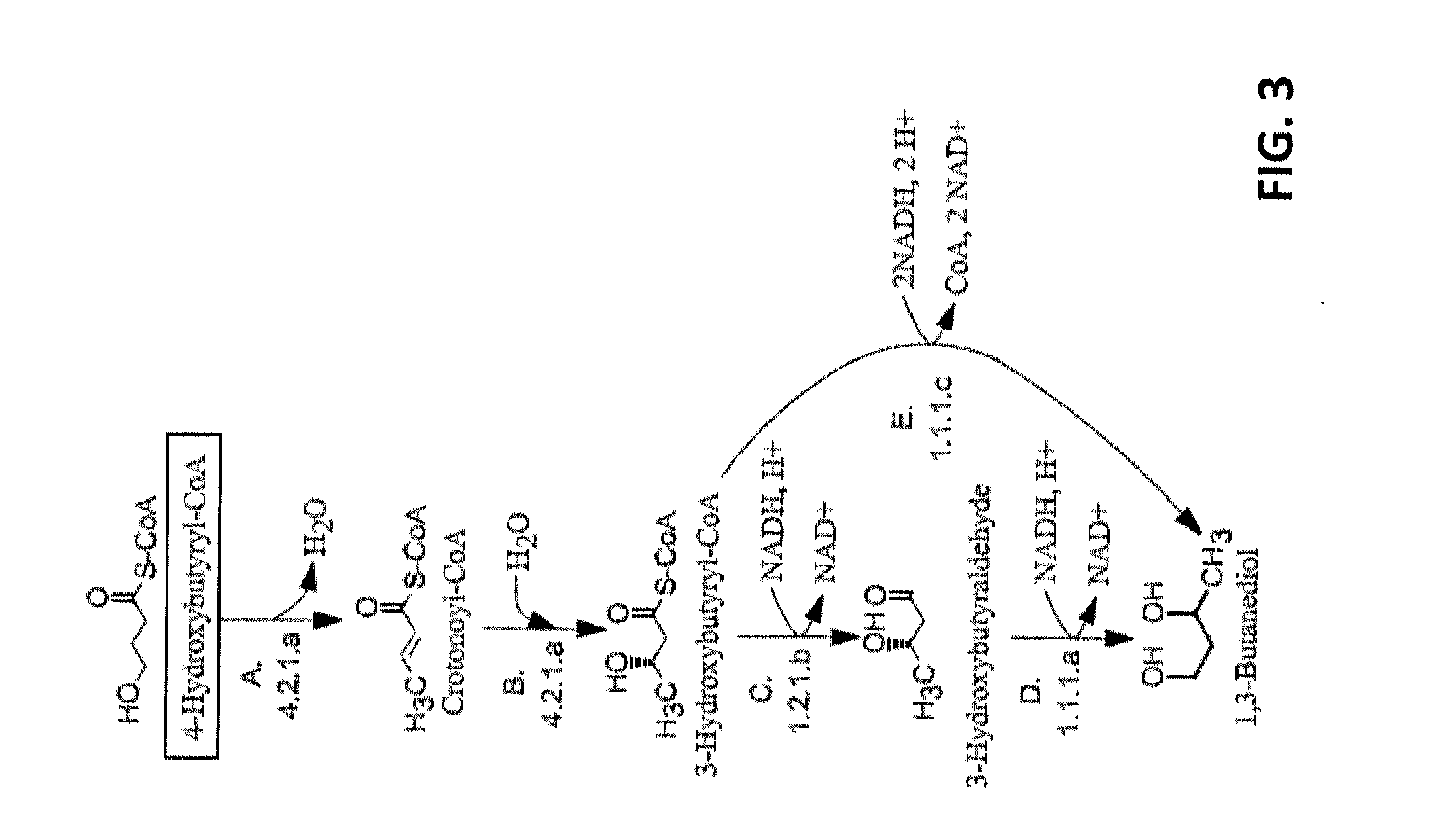
[0009] FIG. 3 shows pathways from 4-hydroxybutyryl-CoA to 1,3-butanediol. Enzymes are: A) 4-hydroxybutyryl-CoA dehydratase, B) crotonase, C) 3-hydroxybutyryl-CoA reductase (aldehyde forming), D) 3-hydroxybutyraldehyde reductase, and E) 3-hydroxybutyryl-CoA reductase (alcohol forming).
[0010] FIG. 4 shows aldehyde dehydrogenases showing significant activity on 3-hydroxybutyl-CoA.
[0011] FIG. 5 shows the specific activity of bld from Clostridium saccharoperbutylacetonicum on 3-Hydroxybutyryl-CoA before and after dialysis.
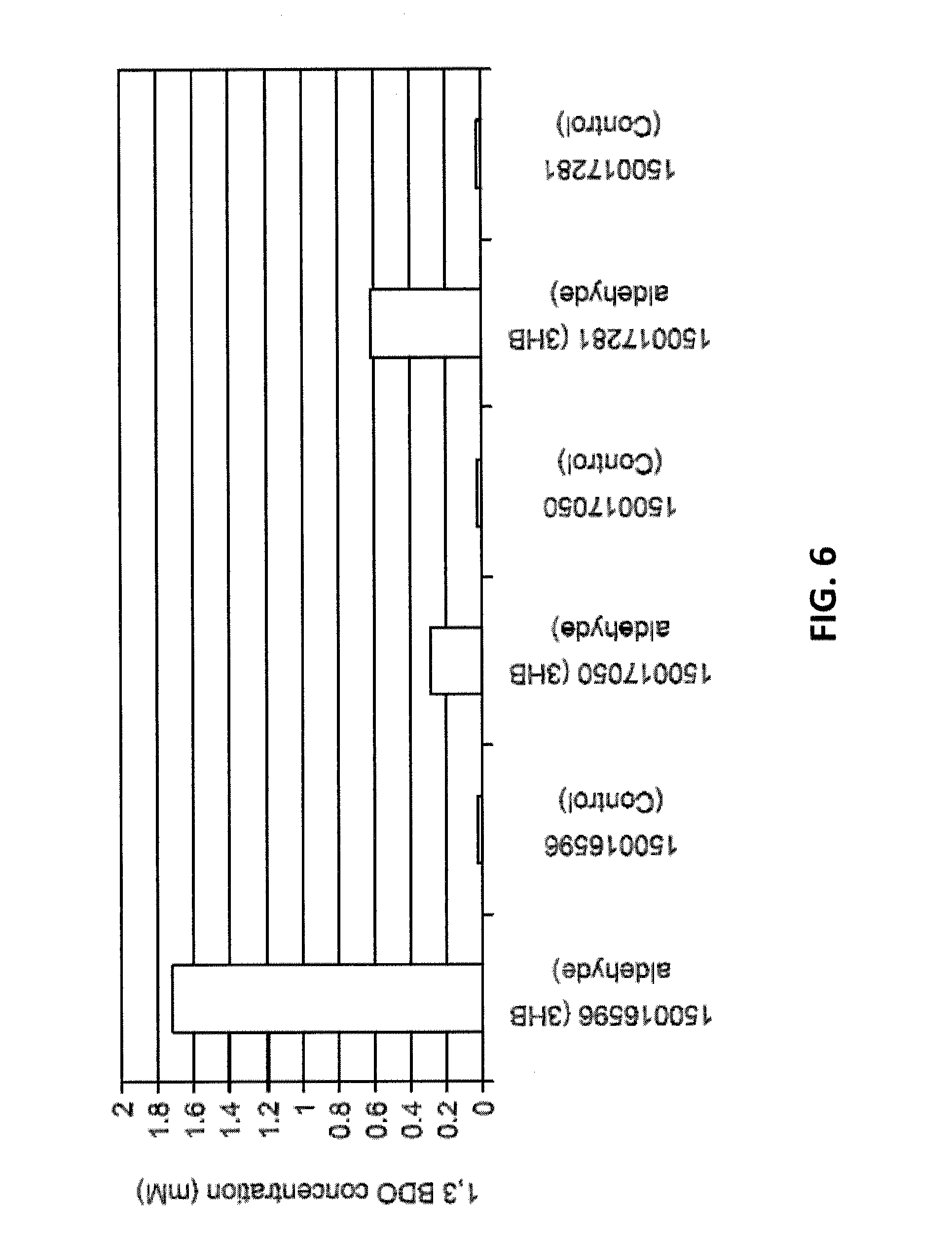
[0012] FIG. 6 shows 1,3-BDO concentrations when 3-hydroxybutyraldehyde was added as a substrate and in the control samples with no substrate. The GI numbers for the alcohol dehydrogenases are shown.
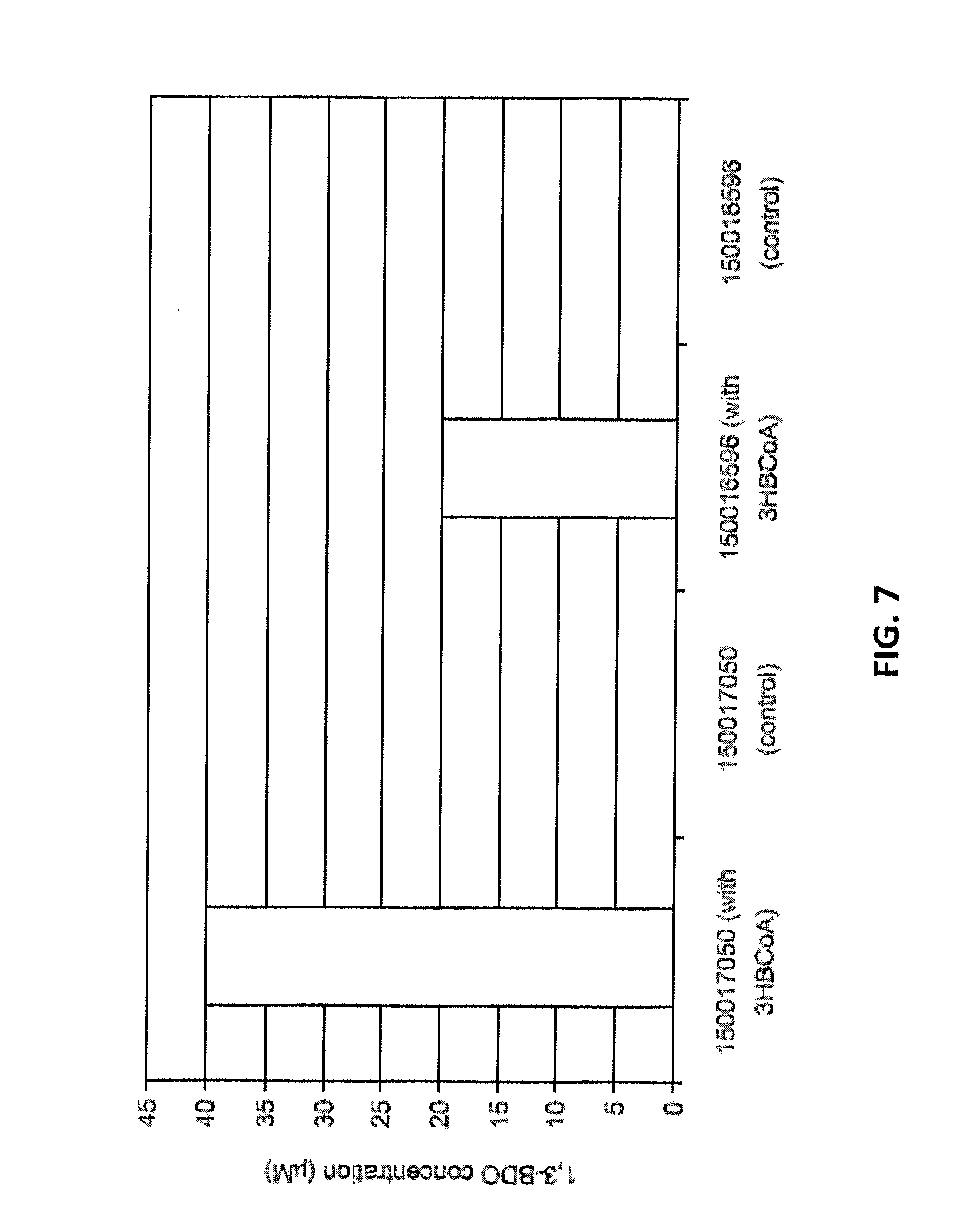
[0013] FIG. 7 shows 1,3-BDO concentrations when 3-hydroxybutyryl-CoA was added as a substrate and in the control samples with no substrate. The GI numbers for the alcohol dehydrogenases are shown. The GI number for the aldehyde dehydrogenase tested in conjunction is 163762382.
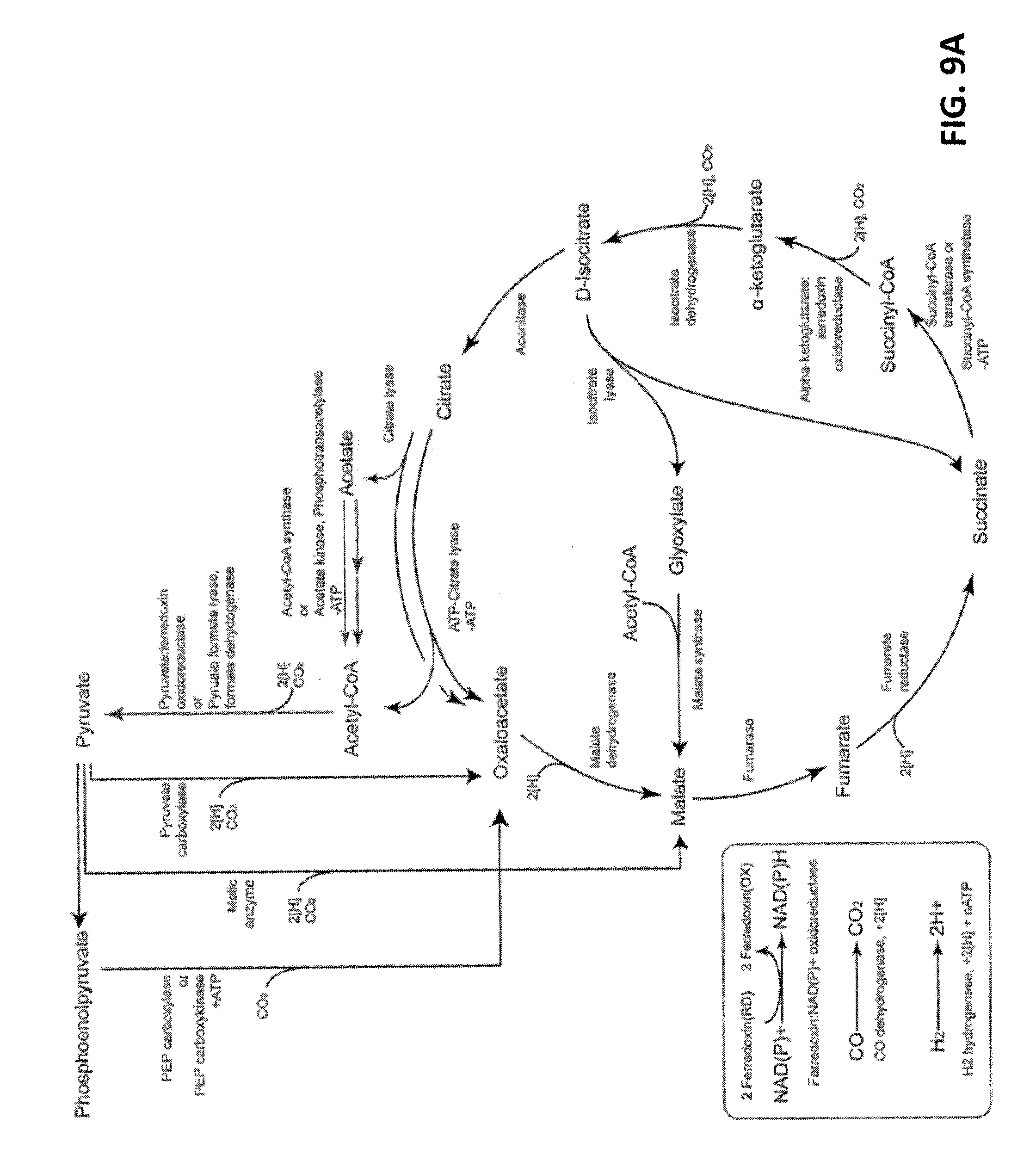
[0014] FIG. 8A shows the pathways for fixation of CO.sub.2 to pyruvate using the reductive TCA cycle.
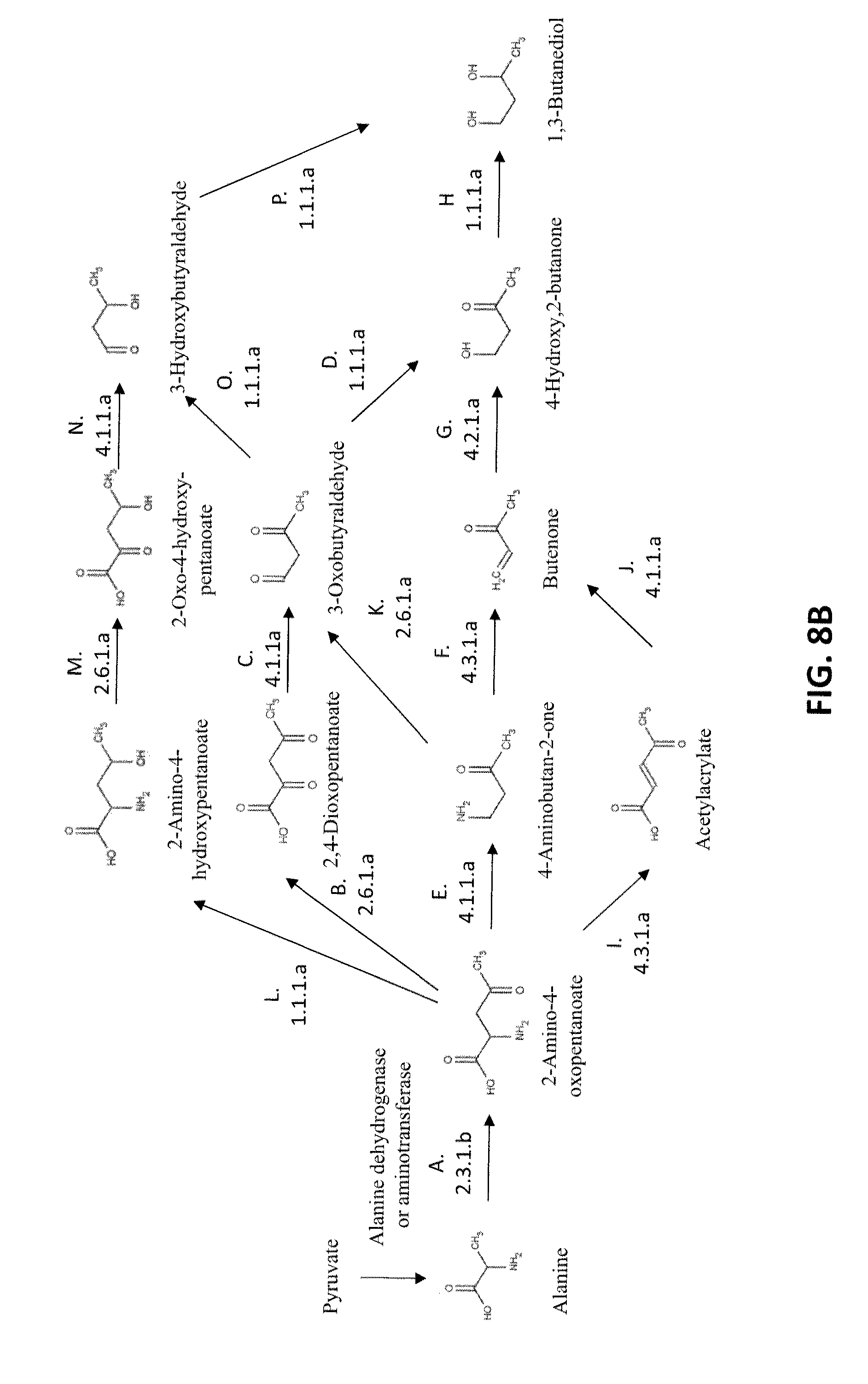
[0015] FIG. 8B shows exemplary pathways for the biosynthesis of 1,3-butanediol from pyruvate; pyruvate is converted to alanine by alanine dehydrogenase alanine aminotransferase; the remaining enzymatic transformations shown are carried out by the following enzymes: A) AKP thiolase, B) AKP aminotransferase or AKP oxidoreductase (deaminating), C) 2,4-dioxopentanoate decarboxylase, D) 3-oxobutyraldehyde reductase (aldehyde reducing), E) AKP decarboxylase, F) 4-aminobutan-2-one ammonia-lyase, G) Butenone hydratase, H) 4-hydroxy,2-butanone reductase, I) AKP ammonia-lyase, J) acetylacrylate decarboxylase, K) 4-aminobutan-2-one aminotransferase or 4-aminobutan-2-one oxidoreductase (deaminating), L) AKP dehydrogenase, M) 2-amino-4-hydroxypentanoate aminotransferase or 2-amino-4-hydroxypentanoate oxidoreductase (deaminating), N) 2-oxo-4-hydroxypentanoate decarboxylase, O) 3-oxobutyraldehyde reductase (ketone reducing), and P) 3-hydroxybutyraldehdye reductase.
[0016] FIG. 9A shows the pathways for fixation of CO.sub.2 to alpha-ketoglutarate, succinate and succinyl-CoA using the reductive TCA cycle.
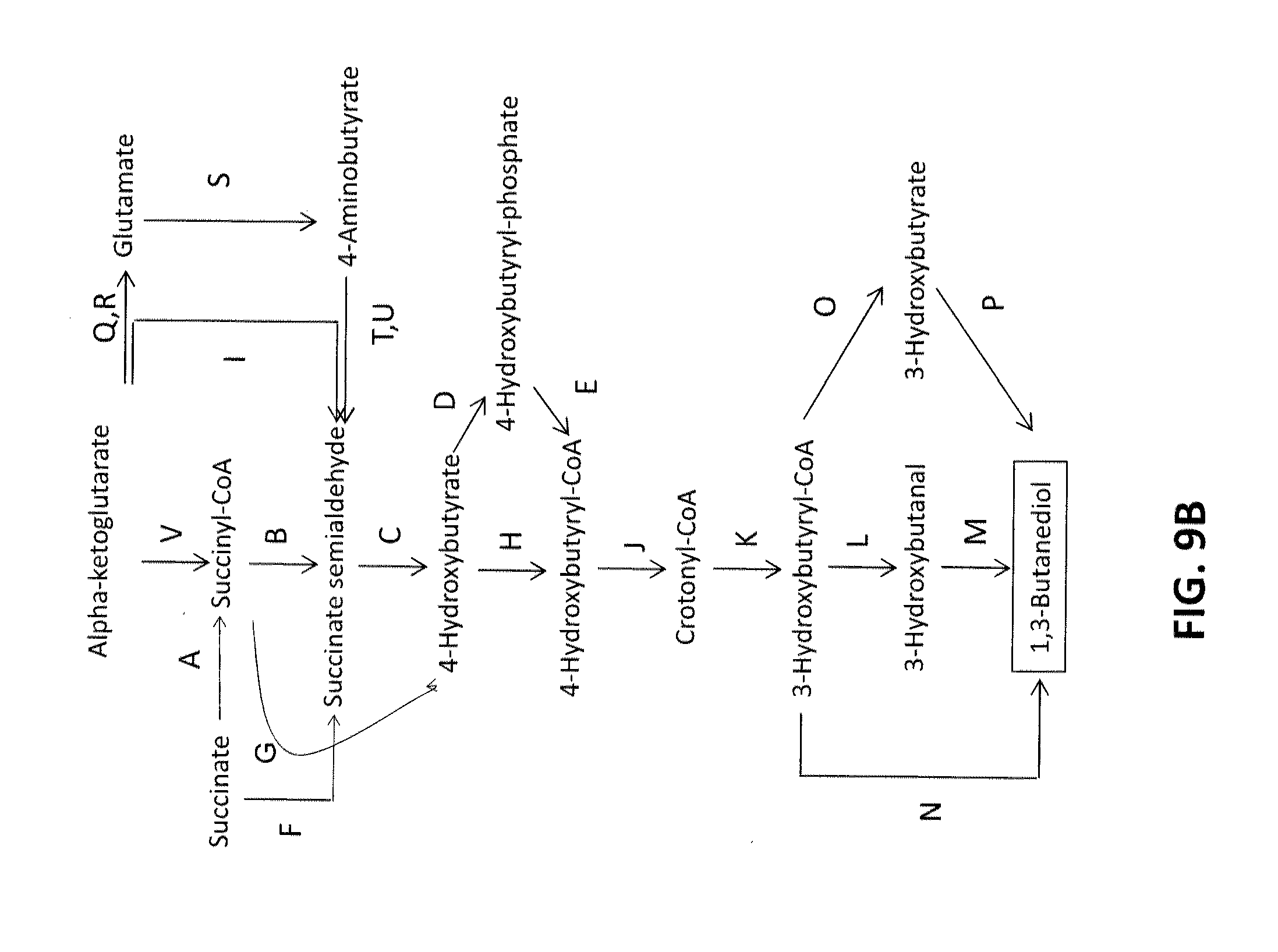
[0017] FIG. 9B shows exemplary pathways for the biosynthesis of 1,3-butanediol from alpha-ketoglutarate, succinate and succinyl-CoA; the enzymatic transformations shown are carried out by the following enzymes: A. Succinyl-CoA transferase, or Succinyl-CoA synthetase (or succinyl-CoA ligase), B. Succinyl-CoA reductase (aldehyde forming), C. 4-Hydroxybutyrate dehydrogenase, D. 4-Hydroxybutyrate kinase, E. Phosphotrans-4-hydroxybutyrylase, F. Succinate reductase, G. Succinyl-CoA reductase (alcohol forming), H. 4-Hydroxybutyryl-CoA transferase, or 4-Hydroxybutyryl-CoA synthetase, or 4-Hydroxybutyryl-CoA ligase I. Alpha-ketoglutarate decarboxylase, J. 4-hydroxybutyryl-CoA dehydratase, K. crotonase, L. 3-hydroxybutyryl-CoA reductase (aldehyde forming), M. 3-hydroxybutanal reductase, N. 3-hydroxybutyryl-CoA reductase (alcohol forming), O. 3-hydroxybutyryl-CoA hydrolase, transferase, or synthetase, P. 3-hydroxybutyrate reductase, Q. Glutamate dehydrogenase and/or R. Glutamate transaminase; S. Glutamate decarboxylase; T. 4-aminobutyrate dehydrogenase and/or U. 4-aminobutyrate transaminase and V. Alpha-ketoglutarate dehydrogenase.
[0018] FIG. 10 shows Western blots of 10 micrograms ACS90 (lane 1), ACS91 (lane2), Mta98/99 (lanes 3 and 4) cell extracts with size standards (lane 5) and controls of M. thermoacetica CODH (Moth.sub.--1202/1203) or Mtr (Moth.sub.--1197) proteins (50, 150, 250, 350, 450, 500, 750, 900, and 1000 ng).
[0019] FIG. 11 shows CO oxidation assay results. Cells (M. thermoacetica or E. coli with the CODH/ACS operon; ACS90 or ACS91 or empty vector: pZA33S) were grown and extracts prepared. Assays were performed at 55.degree. C. at various times on the day the extracts were prepared. Reduction of methylviologen was followed at 578 nm over a 120 sec time course.
[0020] FIG. 12A shows the nucleotide sequence (SEQ ID NO:1) of carboxylic acid reductase from Nocardia iowensis (GNM.sub.--720), and FIG. 12B shows the encoded amino acid sequence (SEQ ID NO:2).
[0021] FIG. 13A shows the nucleotide sequence (SEQ ID NO:3) of phosphpantetheine transferase, which was codon optimized, and FIG. 13B shows the encoded amino acid sequence (SEQ ID NO:4).
[0022] FIG. 14A shows the nucleotide sequence (SEQ ID NO:5) of carboxylic acid reductase from Mycobacterium smegmatis mc(2)155 (designated 890), and FIG. 14B shows the encoded amino acid sequence (SEQ ID NO:6).
[0023] FIG. 15A shows the nucleotide sequence (SEQ ID NO:7) of carboxylic acid reductase from Mycobacterium avium subspecies paratuberculosis K-10 (designated 891), and FIG. 15B shows the encoded amino acid sequence (SEQ ID NO:8).
[0024] FIG. 16A shows the nucleotide sequence (SEQ ID NO:9) of carboxylic acid reductase from Mycobacterium marinum M (designated 892), and FIG. 16B shows the encoded amino acid sequence (SEQ ID NO:10).
[0025] FIG. 17A shows the nucleotide sequence (SEQ ID NO:11) of carboxylic acid reductase designated 891GA, and FIG. 17B shows the encoded amino acid sequence (SEQ ID NO:12).
DETAILED DESCRIPTION
[0026] This invention is directed, in part, to non-naturally occurring microorganisms that express genes encoding enzymes that catalyze 1,3-butanediol (1,3-BDO) production. Pathways for the production of 1,3-butanediol disclosed herein are based on three precursors: (i) D-alanine, (ii) acetoacetyl-CoA, and (iii) 4-hydroxybutyryl-CoA. Successfully engineering these pathways entails identifying an appropriate set of enzymes with sufficient activity and specificity, cloning their corresponding genes into a production host, optimizing fermentation conditions, and assaying for product formation following fermentation.
[0027] The conversion of alanine to 1,3-BDO can be accomplished by a number of pathways in about five enzymatic steps as shown in FIG. 1. In the first step of all pathways (Step A), alanine and acetyl-CoA are combined by 2-amino-4-ketopentanoate thiolase, a highly selective enzyme. The product of this reaction, 2-amino-4-oxopentanoate (AKP) can then be transaminated, reduced, decarboxylated or deaminated as shown in FIG. 1. Further synthetic steps for the production of 1,3-BDO are discussed in detail below. The theoretical yield of 1,3-BDO from each of these pathways is calculated to be about 1.09 mole/mole of glucose consumed.
[0028] FIG. 2 outlines multiple routes for producing 1,3-BDO from acetoacetyl-CoA. Each of these pathways from acetoacetyl-CoA to 1,3-BDO utilizes three reducing equivalents and provides a theoretical yield of 1 mole of 1,3-BDO per mole of glucose consumed. Other carbon substrates such as syngas can also be used for the production of acetoacetyl-CoA. Gasification of glucose to form syngas will result in the maximum theoretical yield of 1.09 moles of 1,3-BDO per mole of glucose consumed, assuming that 6 moles of CO and 6 moles of H.sub.2 are obtained from glucose
6CO+6H.sub.2.fwdarw.1.091C.sub.4H.sub.10O.sub.2+1.636CO.sub.2+0.545H.sub- .2
[0029] 4-Hydroxybutyryl-CoA is an important starting metabolite from which a number of industrially useful compounds can be made, including 1,3-BDO as shown in FIG. 3. Although 4-hydroxybutyryl-CoA is not a highly common central metabolite, methods for engineering strains that synthesize 4-hydroxybutyryl-CoA have been described previously by Applicants in U.S. Patent Application No. 2009/0075351. The 4-hydroxybutyryl-CoA to 1,3-butanediol pathway has a theoretical yield of 1.09 mol/mol product yield assuming glucose as the carbohydrate feedstock.
[0030] This invention is also directed, in part, to methods for producing 1,3-BDO through culturing of these non-naturally occurring microbial organisms. Dehydration of 1,3-BDO produced by the organisms and methods described herein, provides an opportunity to produce renewable butadiene in small end-use facilities obviating the need to transport this flammable and reactive chemical.
[0031] As used herein, the term "non-naturally occurring" when used in reference to a microbial organism or microorganism of the invention is intended to mean that the microbial organism has at least one genetic alteration not normally found in a naturally occurring strain of the referenced species, including wild-type strains of the referenced species. Genetic alterations include, for example, modifications introducing expressible nucleic acids encoding metabolic polypeptides, other nucleic acid additions, nucleic acid deletions and/or other functional disruption of the microbial organism's genetic material. Such modifications include, for example, coding regions and functional fragments thereof, for heterologous, homologous or both heterologous and homologous polypeptides for the referenced species. Additional modifications include, for example, non-coding regulatory regions in which the modifications alter expression of a gene or operon. Exemplary metabolic polypeptides include enzymes or proteins within a 1,3-butanediol biosynthetic pathway.
[0032] A metabolic modification refers to a biochemical reaction that is altered from its naturally occurring state. Therefore, non-naturally occurring microorganisms can have genetic modifications to nucleic acids encoding metabolic polypeptides or, functional fragments thereof. Exemplary metabolic modifications are disclosed herein.
[0033] As used herein, the term "isolated" when used in reference to a microbial organism is intended to mean an organism that is substantially free of at least one component as the referenced microbial organism is found in nature. The term includes a microbial organism that is removed from some or all components as it is found in its natural environment. The term also includes a microbial organism that is removed from some or all components as the microbial organism is found in non-naturally occurring environments. Therefore, an isolated microbial organism is partly or completely separated from other substances as it is found in nature or as it is grown, stored or subsisted in non-naturally occurring environments. Specific examples of isolated microbial organisms include partially pure microbes, substantially pure microbes and microbes cultured in a medium that is non-naturally occurring.
[0034] As used herein, the terms "microbial," "microbial organism" or "microorganism" are intended to mean any organism that exists as a microscopic cell that is included within the domains of archaea, bacteria or eukarya. Therefore, the term is intended to encompass prokaryotic or eukaryotic cells or organisms having a microscopic size and includes bacteria, archaea and eubacteria of all species as well as eukaryotic microorganisms such as yeast and fungi. The term also includes cell cultures of any species that can be cultured for the production of a biochemical.
[0035] As used herein, the term "CoA" or "coenzyme A" is intended to mean an organic cofactor or prosthetic group (nonprotein portion of an enzyme) whose presence is required for the activity of many enzymes (the apoenzyme) to form an active enzyme system. Coenzyme A functions in certain condensing enzymes, acts in acetyl or other acyl group transfer and in fatty acid synthesis and oxidation, pyruvate oxidation and in other acetylation.
[0036] As used herein, the term "substantially anaerobic" when used in reference to a culture or growth condition is intended to mean that the amount of oxygen is less than about 10% of saturation for dissolved oxygen in liquid media. The term also is intended to include sealed chambers of liquid or solid medium maintained with an atmosphere of less than about 1% oxygen.
[0037] "Exogenous" as it is used herein is intended to mean that the referenced molecule or the referenced activity is introduced into the host microbial organism. The molecule can be introduced, for example, by introduction of an encoding nucleic acid into the host genetic material such as by integration into a host chromosome or as non-chromosomal genetic material such as a plasmid. Therefore, the term as it is used in reference to expression of an encoding nucleic acid refers to introduction of the encoding nucleic acid in an expressible form into the microbial organism. When used in reference to a biosynthetic activity, the term refers to an activity that is introduced into the host reference organism. The source can be, for example, a homologous or heterologous encoding nucleic acid that expresses the referenced activity following introduction into the host microbial organism. Therefore, the term "endogenous" refers to a referenced molecule or activity that is present in the host. Similarly, the term when used in reference to expression of an encoding nucleic acid refers to expression of an encoding nucleic acid contained within the microbial organism. The term "heterologous" refers to a molecule or activity derived from a source other than the referenced species whereas "homologous" refers to a molecule or activity derived from the host microbial organism. Accordingly, exogenous expression of an encoding nucleic acid of the invention can utilize either or both a heterologous or homologous encoding nucleic acid.
[0038] It is understood that when more than one exogenous nucleic acid is included in a microbial organism that the more than one exogenous nucleic acids refers to the referenced encoding nucleic acid or biosynthetic activity, as discussed above. It is further understood, as disclosed herein, that such more than one exogenous nucleic acids can be introduced into the host microbial organism on separate nucleic acid molecules, on polycistronic nucleic acid molecules, or a combination thereof, and still be considered as more than one exogenous nucleic acid. For example, as disclosed herein a microbial organism can be engineered to express two or more exogenous nucleic acids encoding a desired pathway enzyme or protein. In the case where two exogenous nucleic acids encoding a desired activity are introduced into a host microbial organism, it is understood that the two exogenous nucleic acids can be introduced as a single nucleic acid, for example, on a single plasmid, on separate plasmids, can be integrated into the host chromosome at a single site or multiple sites, and still be considered as two exogenous nucleic acids. Similarly, it is understood that more than two exogenous nucleic acids can be introduced into a host organism in any desired combination, for example, on a single plasmid, on separate plasmids, can be integrated into the host chromosome at a single site or multiple sites, and still be considered as two or more exogenous nucleic acids, for example three exogenous nucleic acids. Thus, the number of referenced exogenous nucleic acids or biosynthetic activities refers to the number of encoding nucleic acids or the number of biosynthetic activities, not the number of separate nucleic acids introduced into the host organism.
[0039] The non-naturally occurring microbal organisms of the invention can contain stable genetic alterations, which refers to microorganisms that can be cultured for greater than five generations without loss of the alteration. Generally, stable genetic alterations include modifications that persist greater than 10 generations, particularly stable modifications will persist more than about 25 generations, and more particularly, stable genetic modifications will be greater than 50 generations, including indefinitely.
[0040] Those skilled in the art will understand that the genetic alterations, including metabolic modifications exemplified herein, are described with reference to a suitable host organism such as E. coli and their corresponding metabolic reactions or a suitable source organism for desired genetic material such as genes for a desired metabolic pathway. However, given the complete genome sequencing of a wide variety of organisms and the high level of skill in the area of genomics, those skilled in the art will readily be able to apply the teachings and guidance provided herein to essentially all other organisms. For example, the E. coli metabolic alterations exemplified herein can readily be applied to other species by incorporating the same or analogous encoding nucleic acid from species other than the referenced species. Such genetic alterations include, for example, genetic alterations of species homologs, in general, and in particular, orthologs, paralogs or nonorthologous gene displacements.
[0041] An ortholog is a gene or genes that are related by vertical descent and are responsible for substantially the same or identical functions in different organisms. For example, mouse epoxide hydrolase and human epoxide hydrolase can be considered orthologs for the biological function of hydrolysis of epoxides. Genes are related by vertical descent when, for example, they share sequence similarity of sufficient amount to indicate they are homologous, or related by evolution from a common ancestor. Genes can also be considered orthologs if they share three-dimensional structure but not necessarily sequence similarity, of a sufficient amount to indicate that they have evolved from a common ancestor to the extent that the primary sequence similarity is not identifiable. Genes that are orthologous can encode proteins with sequence similarity of about 25% to 100% amino acid sequence identity. Genes encoding proteins sharing an amino acid similarity less that 25% can also be considered to have arisen by vertical descent if their three-dimensional structure also shows similarities. Members of the serine protease family of enzymes, including tissue plasminogen activator and elastase, are considered to have arisen by vertical descent from a common ancestor.
[0042] Orthologs include genes or their encoded gene products that through, for example, evolution, have diverged in structure or overall activity. For example, where one species encodes a gene product exhibiting two functions and where such functions have been separated into distinct genes in a second species, the three genes and their corresponding products are considered to be orthologs. For the production of a biochemical product, those skilled in the art will understand that the orthologous gene harboring the metabolic activity to be introduced or disrupted is to be chosen for construction of the non-naturally occurring microorganism. An example of orthologs exhibiting separable activities is where distinct activities have been separated into distinct gene products between two or more species or within a single species. A specific example is the separation of elastase proteolysis and plasminogen proteolysis, two types of serine protease activity, into distinct molecules as plasminogen activator and elastase. A second example is the separation of mycoplasma 5'-3' exonuclease and Drosophila DNA polymerase III activity. The DNA polymerase from the first species can be considered an ortholog to either or both of the exonuclease or the polymerase from the second species and vice versa.
[0043] In contrast, paralogs are homologs related by, for example, duplication followed by evolutionary divergence and have similar or common, but not identical functions. Paralogs can originate or derive from, for example, the same species or from a different species. For example, microsomal epoxide hydrolase (epoxide hydrolase I) and soluble epoxide hydrolase (epoxide hydrolase II) can be considered paralogs because they represent two distinct enzymes, co-evolved from a common ancestor, that catalyze distinct reactions and have distinct functions in the same species. Paralogs are proteins from the same species with significant sequence similarity to each other suggesting that they are homologous, or related through co-evolution from a common ancestor. Groups of paralogous protein families include HipA homologs, luciferase genes, peptidases, and others.
[0044] A nonorthologous gene displacement is a nonorthologous gene from one species that can substitute for a referenced gene function in a different species. Substitution includes, for example, being able to perform substantially the same or a similar function in the species of origin compared to the referenced function in the different species. Although generally, a nonorthologous gene displacement will be identifiable as structurally related to a known gene encoding the referenced function, less structurally related but functionally similar genes and their corresponding gene products nevertheless will still fall within the meaning of the term as it is used herein. Functional similarity requires, for example, at least some structural similarity in the active site or binding region of a nonorthologous gene product compared to a gene encoding the function sought to be substituted. Therefore, a nonorthologous gene includes, for example, a paralog or an unrelated gene.
[0045] Therefore, in identifying and constructing the non-naturally occurring microbial organisms of the invention having 1,3-BDO biosynthetic capability, those skilled in the art will understand with applying the teaching and guidance provided herein to a particular species that the identification of metabolic modifications can include identification and inclusion or inactivation of orthologs. To the extent that paralogs and/or nonorthologous gene displacements are present in the referenced microorganism that encode an enzyme catalyzing a similar or substantially similar metabolic reaction, those skilled in the art also can utilize these evolutionally related genes.
[0046] Orthologs, paralogs and nonorthologous gene displacements can be determined by methods well known to those skilled in the art. For example, inspection of nucleic acid or amino acid sequences for two polypeptides will reveal sequence identity and similarities between the compared sequences. Based on such similarities, one skilled in the art can determine if the similarity is sufficiently high to indicate the proteins are related through evolution from a common ancestor. Algorithms well known to those skilled in the art, such as Align, BLAST, Clustal W and others compare and determine a raw sequence similarity or identity, and also determine the presence or significance of gaps in the sequence which can be assigned a weight or score. Such algorithms also are known in the art and are similarly applicable for determining nucleotide sequence similarity or identity. Parameters for sufficient similarity to determine relatedness are computed based on well known methods for calculating statistical similarity, or the chance of finding a similar match in a random polypeptide, and the significance of the match determined. A computer comparison of two or more sequences can, if desired, also be optimized visually by those skilled in the art. Related gene products or proteins can be expected to have a high similarity, for example, 25% to 100% sequence identity. Proteins that are unrelated can have an identity which is essentially the same as would be expected to occur by chance, if a database of sufficient size is scanned (about 5%). Sequences between 5% and 24% may or may not represent sufficient homology to conclude that the compared sequences are related. Additional statistical analysis to determine the significance of such matches given the size of the data set can be carried out to determine the relevance of these sequences.
[0047] Exemplary parameters for determining relatedness of two or more sequences using the BLAST algorithm, for example, can be as set forth below. Briefly, amino acid sequence alignments can be performed using BLASTP version 2.0.8 (Jan. 5, 1999) and the following parameters: Matrix: 0 BLOSUM62; gap open: 11; gap extension: 1; x_dropoff: 50; expect: 10.0; wordsize: 3; filter: on. Nucleic acid sequence alignments can be performed using BLASTN version 2.0.6 (Sep. 16, 1998) and the following parameters: Match: 1; mismatch: -2; gap open: 5; gap extension: 2; x_dropoff: 50; expect: 10.0; wordsize: 11; filter: off. Those skilled in the art will know what modifications can be made to the above parameters to either increase or decrease the stringency of the comparison, for example, and determine the relatedness of two or more sequences.
[0048] In some embodiments, the present invention provides a non-naturally occurring microbial organism that includes a microbial organism having a 1,3-butanediol (1,3-BDO) pathway with at least one exogenous nucleic acid encoding a 1,3-BDO pathway enzyme expressed in a sufficient amount to produce 1,3-BDO. The 1,3-BDO pathway includes an enzyme selected from the group consisting of a 2-amino-4-ketopentanoate (AKP) thiolase, an AKP dehydrogenase, a 2-amino-4-hydroxypentanoate aminotransferase, a 2-amino-4-hydroxypentanoate oxidoreductase (deaminating), a 2-oxo-4-hydroxypentanoate decarboxylase, a 3-hydroxybutyraldehyde reductase, an AKP aminotransferase, an AKP oxidoreductase (deaminating), a 2,4-dioxopentanoate decarboxylase, a 3-oxobutyraldehyde reductase (ketone reducing), a 3-oxobutyraldehyde reductase (aldehyde reducing), a 4-hydroxy-2-butanone reductase, an AKP decarboxylase, a 4-aminobutan-2-one aminotransferase, a 4-aminobutan-2-one oxidoreductase (deaminating), a 4-aminobutan-2-one ammonia-lyase, a butenone hydratase, an AKP ammonia-lyase, an acetylacrylate decarboxylase, an acetoacetyl-CoA reductase (CoA-dependent, aldehyde forming), an acetoacetyl-CoA reductase (CoA-dependent, alcohol forming), an acetoacetyl-CoA reductase (ketone reducing), a 3-hydroxybutyryl-CoA reductase (aldehyde forming), a 3-hydroxybutyryl-CoA reductase (alcohol forming), a 4-hydroxybutyryl-CoA dehydratase, and a crotonase.
[0049] Any combination and any number of the aforementioned enzymes can be introduced into a host microbial organism to complete a 1,3-BDO pathway, as exemplified in FIGS. 1-3. For example, the non-naturally occurring microbial organism can include one, two, three, four, five, up to all of the nucleic acids in a 1,3-BDO pathway, each nucleic acid encoding a 1,3-BDO pathway enzyme. Such nucleic acids can include heterologous nucleic acids, additional copies of existing genes, and gene regulatory elements, as explained further below. The pathways of the non-naturally occurring microbial organisms of the invention are also suitably engineered to be cultured in a substantially anaerobic culture medium.
[0050] In some embodiments, the non-naturally occurring microbial organisms having a 1,3-BDO pathway include a set of 1,3-BDO pathway enzymes. A set of 1,3-BDO pathway enzymes represents a group of enzymes that can convert alanine, acetoacetyl-CoA, or 4-hydroxybutyryl-CoA to 1,3-BDO, as show in FIGS. 1-3. Exemplary sets of 1,3-BDO pathway enzymes to convert alanine to 1,3-BDO, according to FIG. 1 include (a) (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP dehydrogenase; (3) a 2-amino-4-hydroxypentanoate aminotransferase or oxidoreductase (deaminating); (4) a 2-oxo-4-hydroxypentanoate decarboxylase; and (5) a 3-hydroxybutyraldehyde reductase; (b) (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP aminotransferase or oxidoreductase (deaminating); (3) a 2,4-dioxopentanoate decarboxylase; (4) a 3-oxobutyraldehyde reductase (ketone reducing); and (5) a 3-hydroxybutyraldehyde reductase; (c) (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP aminotransferase or oxidoreductase (deaminating); (3) a 2,4-dioxopentanoate decarboxylase; (4) a 3-oxobutyraldehyde reductase (aldehyde reducing); and (5) a 4-hydroxy-2-butanone reductase; (d) (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP decarboxylase; (3) a 4-aminobutan-2-one aminotransferase or oxidoreductase (deaminating); (4) a 3-oxobutyraldehyde reductase (ketone reducing); and (5) a 3-hydroxybutyraldehyde reductase; (e) (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP decarboxylase; (3) a 4-aminobutan-2-one aminotransferase or oxidoreductase (deaminating); (4) a 3-oxobutyraldehyde reductase (aldehyde reducing); and (5) a 4-hydroxy-2-butanone reductase; (f) (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP decarboxylase; (3) a 4-aminobutan-2-one ammonia-lyase; (4) a butenone hydratase; and (5) a 4-hydroxy-2-butanone reductase; and (g) (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP ammonia-lyase; (3) an acetylacrylate decarboxylase; (4) a butenone hydratase; and (5) a 4-hydroxy-2-butanone reductase;
[0051] Exemplary sets of 1,3-BDO pathway enzymes to convert acetoacetyl-CoA to 1,3-BDO, according to FIG. 2 include (h) (1) an acetoacetyl-CoA reductase (CoA-dependent, aldehyde forming); (2) a 3-oxobutyraldehyde reductase (ketone reducing); and (3) a 3-hydroxybutyraldehyde reductase; (i) (1) an acetoacetyl-CoA reductase (CoA dependent, alcohol forming) and (2) a 4-hydroxy-2-butanone reductase; (j) (1) an acetoacetyl-CoA reductase (CoA-dependent, aldehyde forming); (2) a 3-oxobutyraldehyde reductase (aldehyde reducing); and (3) a 4-hydroxy-2-butanone reductase; (k) (1) an acetoacetyl-CoA reductase (ketone reducing) and (2) a 3-hydroxybutyryl-CoA reductase (alcohol forming); and (l) (1) an acetoacetyl-CoA reductase (ketone reducing); (2) a 3-hydroxybutyryl-CoA reductase (aldehyde forming); and (3) a 3-hydroxybutyraldehyde reductase;
[0052] Exemplary sets of 1,3-BDO pathway enzymes to convert 4-hydroxybutyryl-CoA to 1,3-BDO, according to FIG. 3 include (m) (1) a 4-hydroxybutyryl-CoA dehydratase; (2) a crotonase; and (3) a 3-hydroxybutyryl-CoA reductase (alcohol forming); and (n) (1) a 4-hydroxybutyryl-CoA dehydratase; (2) a crotonase; (3) a 3-hydroxybutyryl-CoA reductase (aldehyde forming); and (4) a 3-hydroxybutyraldehyde reductase.
[0053] The conversion of alanine to 1,3-BDO can be accomplished by a number of pathways involving about five enzymatic steps as shown in FIG. 1. In the first step of all pathways (Step A), alanine and acetyl-CoA are combined by 2-amino-4-ketopentanoate thiolase, a highly selective enzyme. The product of this reaction, 2-amino-4-oxopentanoate (AKP) can then be transaminated, reduced, decarboxylated or deaminated as shown in FIG. 1.
[0054] In one route, AKP converted to 2,4-dioxopentanoate, a 2-keto acid similar in structure to alpha-ketoglutarate, by an aminotransferase or deaminating oxidoreductase (Step B). 2,4-Dioxopentanoate is then converted to 3-oxobutyraldehyde by a 2-ketoacid decarboxylase (Step C). Reduction of the ketone and aldehyde groups to their corresponding alcohols yields 1,3-butanediol. These reductions can occur in either order to form the intermediates 3-hydroxybutyraldehyde (Steps O and P) or 4-hydroxy,2-butanone (Steps D and H).
[0055] In another route, the 4-oxo group of AKP is first reduced to a secondary alcohol by AKP dehydrogenase (Step L). The product, 2-amino-4-hydroxypentanoate, is then converted to 2-oxo-4-hydroxypentanoate (Step M). The resulting 2-ketoacid is decarboxylated to 3-hydroxybutyraldehyde (Step N). In the final step of this route, the aldehyde of 3-hydroxybutyraldehyde is reduced to a primary alcohol by 3-hydroxybutyraldehyde reductase, forming 1,3-butanediol (Step P).
[0056] Yet another route involves decarboxylation of AKP by an amino acid decarboxylase (Step E). The decarboxylation product, 4-aminobutan-2-one, can either be transaminated or oxidatively deaminated to 3-oxobutyraldehyde (Step K) or deaminated to butenone (Step F). When 3-oxobutyraldehyde is formed, two alcohol-forming reduction steps are used to form 1,3-butanediol, as described previously (Steps O and P, or Steps D and H). The deamination product, butenone, is then hydrolyzed to 4-hydroxy,2-butanone (Step G), which is reduced to 1,3-butanediol by 4-hydroxy-2-butanone reductase (Step H).
[0057] Yet another route involves the deamination of AKP to acetylacrylate (Step I). Acetylacrylate is decarboxylated to butenone (Step J), which is then converted to 1,3-butandiol by butenone hydratase (Step G) and 4-hydroxy,2-butanone reductase (Step H).
[0058] Based on the routes described above for the production 1,3-BDO from alanine, in some embodiments, the non-naturally occurring microbial organism has a set of 1,3-BDO pathway enzymes that includes (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP dehydrogenase; (3) a 2-amino-4-hydroxypentanoate aminotransferase or oxidoreductase (deaminating); (4) a 2-oxo-4-hydroxypentanoate decarboxylase; and (5) a 3-hydroxybutyraldehyde reductase. Any number of nucleic acids encoding these enzymes can be introduced into a host microbial organism including one, two, three, four, up to all five of the nucleic acids that encode these enzymes. Where one, two, three, or four exogenous nucleic acids are introduced, such nucleic acids can be any permutation of the five nucleic acids.
[0059] In other embodiments non-naturally occurring microbial organism has a set of 1,3-BDO pathway enzymes that includes (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP aminotransferase or oxidoreductase (deaminating); (3) a 2,4-dioxopentanoate decarboxylase; (4) a 3-oxobutyraldehyde reductase (ketone reducing); and (5) a 3-hydroxybutyraldehyde reductase. Any number of nucleic acids encoding these enzymes can be introduced into a host microbial organism including one, two, three, four, up to all five of the nucleic acids that encode these enzymes. Where one, two, three, or four exogenous nucleic acids are introduced, such nucleic acids can be any permutation of the five nucleic acids.
[0060] In still other embodiments, the non-naturally occurring microbial organism has a set of 1,3-BDO pathway enzymes that includes (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP aminotransferase or oxidoreductase (deaminating); (3) a 2,4-dioxopentanoate decarboxylase; (4) a 3-oxobutyraldehyde reductase (aldehyde reducing); and (5) a 4-hydroxy-2-butanone reductase. Any number of nucleic acids encoding these enzymes can be introduced into a host microbial organism including one, two, three, four, up to all five of the nucleic acids that encode these enzymes. Where one, two, three, or four exogenous nucleic acids are introduced, such nucleic acids can be any permutation of the five nucleic acids.
[0061] In yet further embodiments, the non-naturally occurring microbial organism has a set of 1,3-BDO pathway enzymes that includes (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP decarboxylase; (3) a 4-aminobutan-2-one aminotransferase or oxidoreductase (deaminating); (4) a 3-oxobutyraldehyde reductase (ketone reducing); and (5) a 3-hydroxybutyraldehyde reductase. Any number of nucleic acids encoding these enzymes can be introduced into a host microbial organism including one, two, three, four, up to all five of the nucleic acids that encode these enzymes. Where one, two, three, or four exogenous nucleic acids are introduced, such nucleic acids can be any permutation of the five nucleic acids.
[0062] In yet still further embodiments, the non-naturally occurring microbial organism has a set of 1,3-BDO pathway enzymes that includes (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP decarboxylase; (3) a 4-aminobutan-2-one aminotransferase or oxidoreductase (deaminating); (4) a 3-oxobutyraldehyde reductase (aldehyde reducing); and (5) a 4-hydroxy-2-butanone reductase. Any number of nucleic acids encoding these enzymes can be introduced into a host microbial organism including one, two, three, four, up to all five of the nucleic acids that encode these enzymes. Where one, two, three, or four exogenous nucleic acids are introduced, such nucleic acids can be any permutation of the five nucleic acids.
[0063] In still further embodiments, the non-naturally occurring microbial organism has a set of 1,3-BDO pathway enzymes that includes (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP decarboxylase; (3) a 4-aminobutan-2-one ammonia-lyase; (4) a butenone hydratase; and (5) a 4-hydroxy-2-butanone reductase. Any number of nucleic acids encoding these enzymes can be introduced into a host microbial organism including one, two, three, four, up to all five of the nucleic acids that encode these enzymes. Where one, two, three, or four exogenous nucleic acids are introduced, such nucleic acids can be any permutation of the five nucleic acids.
[0064] In yet still further embodiments, the non-naturally occurring microbial organism has a set of 1,3-BDO pathway enzymes that includes (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP ammonia-lyase; (3) an acetylacrylate decarboxylase; (4) a butenone hydratase; and (5) a 4-hydroxy-2-butanone reductase. Any number of nucleic acids encoding these enzymes can be introduced into a host microbial organism including one, two, three, four, up to all five of the nucleic acids that encode these enzymes. Where one, two, three, or four exogenous nucleic acids are introduced, such nucleic acids can be any permutation of the five nucleic acids.
[0065] FIG. 2 outlines multiple routes for producing 1,3-butanediol from acetoacetyl-CoA. One route through steps A, B and C utilizes (i) CoA-dependent, aldehyde forming acetoacetyl-CoA reductase to convert acetoacetyl-CoA into 3-oxobutyraldehyde (FIG. 2, Step A), (ii) 3-oxobutyraldehyde reductase to reduce 3-oxobutyraldehyde to 3-hydroxybutyraldehyde (FIG. 2, Step B), and (iii) finally, 3-hydroxybutyraldehyde reductase to foam 1,3-butanediol (FIG. 2, Step C).
[0066] Alternatively, acetoacetyl-CoA can be reduced via the aldehyde forming acetoacetyl-CoA reductase to form 4-hydroxy,2-butanone (FIG. 2, Step D). 4-hydroxy,2-butanone can also be formed by the reduction of 3-oxobutyraldehyde by the aldehyde reducing 3-oxobutyraldehyde reductase (FIG. 2, Step E). Eventually, 4-hydroxy,2-butanone can be reduced to form 1,3-BDO by 4-hydroxy-2-butanone reductase (FIG. 2, Step F).
[0067] Yet another set of 1,3-BDO forming routes rely on the reduction of acetoacetyl-CoA to 3-hydroxybutyryl-CoA by the ketone reducing acetoacetyl-CoA reductase (FIG. 2, Step G). This enzyme reduces the ketone function in acetoacetyl-CoA to a hydroxyl group. 3-hydroxybutyryl-CoA can be reduced by the bifunctional alcohol-forming 3-hydroxybutyryl-CoA reductase to form 1,3-butanediol (FIG. 2, Step I). Alternatively, it can first be reduced to 3-hydroxybutyraldehyde via the aldehyde forming 3-hydroxybutyryl-CoA reductase (Step H) and 3-hydroxybutyraldehyde can then be reduced as shown in Step C.
[0068] Based on the routes described above for the production 1,3-BDO from acetoacetyl-CoA, in some embodiments, the non-naturally occurring microbial organism has a set of 1,3-BDO pathway enzymes that includes (1) an acetoacetyl-CoA reductase (CoA-dependent, aldehyde forming); (2) a 3-oxobutyraldehyde reductase (ketone reducing); and (3) a 3-hydroxybutyraldehyde reductase. Any number of nucleic acids encoding these enzymes can be introduced into a host microbial organism including one, two up to all three of the nucleic acids that encode these enzymes. Where one or two exogenous nucleic acids are introduced, such nucleic acids can be any permutation of the three nucleic acids.
[0069] In other embodiments, the non-naturally occurring microbial organism has a set of 1,3-BDO pathway enzymes that includes (1) an acetoacetyl-CoA reductase (CoA dependent, alcohol forming) and (2) a 4-hydroxy-2-butanone reductase. Any number of nucleic acids encoding these enzymes can be introduced into a host microbial organism including one or both of the nucleic acids that encode these enzymes. Where one exogenous nucleic acid is introduced, such a nucleic acid can be either of the two nucleic acids.
[0070] In further embodiments, the non-naturally occurring microbial organism has a set of 1,3-BDO pathway enzymes that includes (1) an acetoacetyl-CoA reductase (CoA-dependent, aldehyde forming); (2) a 3-oxobutyraldehyde reductase (aldehyde reducing); and (3) a 4-hydroxy-2-butanone reductase. Any number of nucleic acids encoding these enzymes can be introduced into a host microbial organism including one, two up to all three of the nucleic acids that encode these enzymes. Where one or two exogenous nucleic acids are introduced, such nucleic acids can be any permutation of the three nucleic acids.
[0071] In yet further embodiments, the non-naturally occurring microbial organism has a set of 1,3-BDO pathway enzymes that includes (1) an acetoacetyl-CoA reductase (ketone reducing) and (2) a 3-hydroxybutyryl-CoA reductase (alcohol forming). Any number of nucleic acids encoding these enzymes can be introduced into a host microbial organism including one or both of the nucleic acids that encode these enzymes. Where one exogenous nucleic acid is introduced, such a nucleic acid can be either of the two nucleic acids.
[0072] In still further embodiments, the non-naturally occurring microbial organism has a set of 1,3-BDO pathway enzymes that includes (1) an acetoacetyl-CoA reductase (ketone reducing); (2) a 3-hydroxybutyryl-CoA reductase (aldehyde forming); and (3) a 3-hydroxybutyraldehyde reductase. Any number of nucleic acids encoding these enzymes can be introduced into a host microbial organism including one, two up to all three of the nucleic acids that encode these enzymes. Where one or two exogenous nucleic acids are introduced, such nucleic acids can be any permutation of the three nucleic acids.
[0073] 4-hydroxybutyryl-CoA is an important starting metabolite from which a number of industrially useful compounds can be made. Although 4-hydroxybutyryl-CoA is not a highly common central metabolite, methods for engineering strains that synthesize 4-hydroxybutyryl-CoA have been described in Burk et al. (US 20090075351). An exemplary method involves synthesizing 4-hydroxybutyryl-CoA from succinyl-CoA by employing genes encoding succinic semialdehyde dehydrogenase (CoA-dependent), 4-hydroxybutyrate dehydrogenase, 4-hydroxybutyrate kinase, and phosphotransbutyrylase activities.
[0074] The first step in the pathway involves the dehydration of 4-hydroxybutyryl-CoA (Step A, FIG. 3) followed by the hydration of crotonoyl-CoA to form 3-hydroxybutyryl-CoA (Step B). 3-hydroxybutyryl-CoA then undergoes two reduction steps to form 1,3-butanediol carried out by either two enzymes (Steps C and D) or a single dual-function enzyme (Step E).
[0075] Thus, in some embodiments, the non-naturally occurring microbial organism has a set of 1,3-BDO pathway enzymes that includes (1) a 4-hydroxybutyryl-CoA dehydratase; (2) a crotonase; and (3) a 3-hydroxybutyryl-CoA reductase (alcohol forming). Any number of nucleic acids encoding these enzymes can be introduced into a host microbial organism including one, two up to all three of the nucleic acids that encode these enzymes. Where one or two exogenous nucleic acids are introduced, such nucleic acids can be any permutation of the three nucleic acids.
[0076] In other embodiments, the non-naturally occurring microbial organism has a set of 1,3-BDO pathway enzymes that includes (1) a 4-hydroxybutyryl-CoA dehydratase; (2) a crotonase; (3) a 3-hydroxybutyryl-CoA reductase (aldehyde forming); and (4) a 3-hydroxybutyraldehyde reductase. Any number of nucleic acids encoding these enzymes can be introduced into a host microbial organism including one, two, three up to all four of the nucleic acids that encode these enzymes. Where one, two, or three exogenous nucleic acids are introduced, such nucleic acids can be any permutation of the four nucleic acids.
[0077] In an additional embodiment, the invention provides a non-naturally occurring microbial organism having a 1,3-BDO pathway, wherein the non-naturally occurring microbial organism comprises at least one exogenous nucleic acid encoding an enzyme or protein that converts a substrate to a product selected from the group consisting of alanine to 2-amino-4-oxopentanoate, 2-amino-4-oxopentanoate to 2-amino-4-hydroxypentanoate, 2-amino-4-hydroxypentanoate to 2-oxo-4-hydroxypentanoate, 2-oxo-4-hydroxypentanoate to 3-hydroxybutyraldehyde, and 3-hydroxybutyraldehyde to 1,3-BDO.
[0078] In an additional embodiment, the invention provides a non-naturally occurring microbial organism having a 1,3-BDO pathway, wherein the non-naturally occurring microbial organism comprises at least one exogenous nucleic acid encoding an enzyme or protein that converts a substrate to a product selected from the group consisting of alanine to 2-amino-4-oxopentanoate, 2-amino-4-oxopentanoate to 2,4-dioxopentanoate, 2,4-dioxopentanoate to 3-oxobutyraldehyde, 3-oxobutyraldehyde to 3-hydroxybutyraldehyde, and 3-hydroxybutyraldehyde to 1,3-BDO.
[0079] In an additional embodiment, the invention provides a non-naturally occurring microbial organism having a 1,3-BDO pathway, wherein the non-naturally occurring microbial organism comprises at least one exogenous nucleic acid encoding an enzyme or protein that converts a substrate to a product selected from the group consisting of alanine to 2-amino-4-oxopentanoate, 2-amino-4-oxopentanoate to 2,4-dioxopentanoate, 2,4-dioxopentanoate to 3-oxobutyraldehyde, 3-oxobutyraldehyde to 4-hydroxy-2-butanone, and 4-hydroxy-2-butanone to 1,3-BDO.
[0080] In an additional embodiment, the invention provides a non-naturally occurring microbial organism having a 1,3-BDO pathway, wherein the non-naturally occurring microbial organism comprises at least one exogenous nucleic acid encoding an enzyme or protein that converts a substrate to a product selected from the group consisting of alanine to 2-amino-4-oxopentanoate, 2-amino-4-oxopentanoate to 4-aminobutan-2-one, 4-aminobutan-2-one to 3-oxobutyraldehyde, 3-oxobutyraldehyde to 3-hydroxybutyraldehyde, and 3-hydroxybutyraldehyde to 1,3-BDO.
[0081] In an additional embodiment, the invention provides a non-naturally occurring microbial organism having a 1,3-BDO pathway, wherein the non-naturally occurring microbial organism comprises at least one exogenous nucleic acid encoding an enzyme or protein that converts a substrate to a product selected from the group consisting of alanine to 2-amino-4-oxopentanoate, 2-amino-4-oxopentanoate to 4-aminobutan-2-one, 4-aminobutan-2-one to 3-oxobutyraldehyde, 3-oxobutyraldehyde to 4-hydroxy-2-butanone, and 4-hydroxy-2-butanone to 1,3-BDO.
[0082] In an additional embodiment, the invention provides a non-naturally occurring microbial organism having a 1,3-BDO pathway, wherein the non-naturally occurring microbial organism comprises at least one exogenous nucleic acid encoding an enzyme or protein that converts a substrate to a product selected from the group consisting of alanine to 2-amino-4-oxopentanoate, 2-amino-4-oxopentanoate to 4-aminobutan-2-one, 4-aminobutan-2-one to butenone, butenone to 4-hydroxy-2-butanone, and 4-hydroxy-2-butanone to 1,3-BDO.
[0083] In an additional embodiment, the invention provides a non-naturally occurring microbial organism having a 1,3-BDO pathway, wherein the non-naturally occurring microbial organism comprises at least one exogenous nucleic acid encoding an enzyme or protein that converts a substrate to a product selected from the group consisting of alanine to 2-amino-4-oxopentanoate, 2-amino-4-oxopentanoate to acetylacrylate, acetylacrylate to butenone, butenone to 4-hydroxy-2-butanone, and 4-hydroxy-2-butanone to 1,3-BDO.
[0084] Thus, the invention provides a non-naturally occurring microbial organism containing at least one exogenous nucleic acid encoding an enzyme or protein, where the enzyme or protein converts the substrates and products of a 1,3-BDO pathway converting alanine to 1,3-BDO, as exemplified by the pathways shown in FIG. 1.
[0085] In an additional embodiment, the invention provides a non-naturally occurring microbial organism having a 1,3-BDO pathway, wherein the non-naturally occurring microbial organism comprises at least one exogenous nucleic acid encoding an enzyme or protein that converts a substrate to a product selected from the group consisting of acetoacetyl-CoA to 4-hydroxy-2-butanone, and 4-hydroxy-2-butanone to 1,3-BDO.
[0086] In an additional embodiment, the invention provides a non-naturally occurring microbial organism having a 1,3-BDO pathway, wherein the non-naturally occurring microbial organism comprises at least one exogenous nucleic acid encoding an enzyme or protein that converts a substrate to a product selected from the group consisting of acetoacetyl-CoA to 3-oxobutyraldehyde, 3-oxobutyraldehyde to 4-hydroxy-2-butanone, and 4-hydroxy-2-butanone to 1,3-BDO.
[0087] In an additional embodiment, the invention provides a non-naturally occurring microbial organism having a 1,3 BDO pathway, wherein the non-naturally occurring microbial organism comprises at least one exogenous nucleic acid encoding an enzyme or protein that converts a substrate to a product selected from the group consisting of acetoacetyl-CoA to 3-oxobutyraldehyde, 3-oxobutyraldehyde to 3-hydroxybutryaldehyde, and 3-hydroxybutryaldehyde to 1,3-BDO.
[0088] In an additional embodiment, the invention provides a non-naturally occurring microbial organism having a 1,3-BDO pathway, wherein the non-naturally occurring microbial organism comprises at least one exogenous nucleic acid encoding an enzyme or protein that converts a substrate to a product selected from the group consisting of acetoacetyl-CoA to 3-hydroxybutyryl-CoA, 3-hydroxybutyryl-CoA to 3-hydroxybutryaldehyde, and 3-hydroxybutryaldehyde to 1,3-BDO.
[0089] In an additional embodiment, the invention provides a non-naturally occurring microbial organism having a 1,3-BDO pathway, wherein the non-naturally occurring microbial organism comprises at least one exogenous nucleic acid encoding an enzyme or protein that converts a substrate to a product selected from the group consisting of acetoacetyl-CoA to 3-hydroxybutyryl-CoA, and 3-hydroxybutyryl-CoA to 1,3-BDO.
[0090] Thus, the invention provides a non-naturally occurring microbial organism containing at least one exogenous nucleic acid encoding an enzyme or protein, where the enzyme or protein converts the substrates and products of a 1,3-BDO pathway converting acetoacetyl-CoA to 1,3-BDO, as exemplified by the pathways shown in FIG. 2.
[0091] In an additional embodiment, the invention provides a non-naturally occurring microbial organism having a 1,3-BDO pathway, wherein the non-naturally occurring microbial organism comprises at least one exogenous nucleic acid encoding an enzyme or protein that converts a substrate to a product selected from the group consisting of 4-hydroxybutyryl-CoA to crotonoyl-CoA, crotonoyl-CoA to 3-hydroxybutyryl-CoA, 3-hydroxybutyryl-CoA to 3-hydroxybutryaldehyde, and 3-hydroxybutryaldehyde to 1,3-BDO.
[0092] In an additional embodiment, the invention provides a non-naturally occurring microbial organism having a 1,3-BDO pathway, wherein the non-naturally occurring microbial organism comprises at least one exogenous nucleic acid encoding an enzyme or protein that converts a substrate to a product selected from the group consisting of 4-hydroxybutyryl-CoA to crotonoyl-CoA, crotonoyl-CoA to 3-hydroxybutyryl-CoA, and 3-hydroxybutyryl-CoA to 1,3-BDO.
[0093] Thus, the invention provides a non-naturally occurring microbial organism containing at least one exogenous nucleic acid encoding an enzyme or protein, where the enzyme or protein converts the substrates and products of a 1,3-BDO pathway, the pathway converting 4-hydroxybutyryl-CoA to 1,3-BDO, as exemplified by the pathways shown in FIG. 3.
[0094] This invention is also directed, in part to engineered biosynthetic pathways to improve carbon flux through a central metabolism intermediate en route to 1,3-butanediol. The present invention provides non-naturally occurring microbial organisms having one or more exogenous genes encoding enzymes that can catalyze various enzymatic transformations en route to 1,3-butanediol. In some embodiments, these enzymatic transformations are part of the reductive tricarboxylic acid (RTCA) cycle and are used to improve product yields, including but not limited to, from carbohydrate-based carbon feedstock.
[0095] In numerous engineered pathways, realization of maximum product yields based on carbohydrate feedstock is hampered by insufficient reducing equivalents or by loss of reducing equivalents and/or carbon to byproducts. In accordance with some embodiments, the present invention increases the yields of 1,3-butanediol by (i) enhancing carbon fixation via the reductive TCA cycle, and/or (ii) accessing additional reducing equivalents from gaseous carbon sources and/or syngas components such as CO, CO.sub.2, and/or H.sub.2. In addition to syngas, other sources of such gases include, but are not limited to, the atmosphere, either as found in nature or generated.
[0096] The CO.sub.2-fixing reductive tricarboxylic acid (RTCA) cycle is an endergenic anabolic pathway of CO.sub.2 assimilation which uses reducing equivalents and ATP (FIGS. 8a and 9a). One turn of the RTCA cycle assimilates two moles of CO.sub.2 into one mole of acetyl-CoA, or four moles of CO.sub.2 into one mole of oxaloacetate. This additional availability of acetyl-CoA improves the maximum theoretical yield of product molecules derived from carbohydrate-based carbon feedstock. Exemplary carbohydrates include but are not limited to glucose, sucrose, xylose, arabinose and glycerol.
[0097] In some embodiments, the reductive TCA cycle, coupled with carbon monoxide dehydrogenase and/or hydrogenase enzymes (FIGS. 8a and 9a insert), can be employed to allow syngas, CO.sub.2, CO, H.sub.2, and/or other gaseous carbon source utilization by microorganisms. Synthesis gas (syngas), in particular is a mixture of primarily H.sub.2 and CO, sometimes including some amounts of CO.sub.2, that can be obtained via gasification of any organic feedstock, such as coal, coal oil, natural gas, biomass, or waste organic matter. Numerous gasification processes have been developed, and most designs are based on partial oxidation, where limiting oxygen avoids full combustion, of organic materials at high temperatures (500-1500.degree. C.) to provide syngas as a 0.5:1-3:1 H.sub.2/CO mixture. In addition to coal, biomass of many types has been used for syngas production and represents an inexpensive and flexible feedstock for the biological production of renewable chemicals and fuels. Carbon dioxide can be provided from the atmosphere or in condensed from, for example, from a tank cylinder, or via sublimation of solid CO.sub.2. Similarly, CO and hydrogen gas can be provided in reagent form and/or mixed in any desired ratio. Other gaseous carbon forms can include, for example, methanol or similar volatile organic solvents.
[0098] The components of synthesis gas and/or other carbon sources can provide sufficient CO.sub.2, reducing equivalents, and ATP for the reductive TCA cycle to operate. One turn of the RICA cycle assimilates two moles of CO.sub.2 into one mole of acetyl-CoA and requires 2 ATP and 4 reducing equivalents. CO and/or H.sub.2 can provide reducing equivalents by means of carbon monoxide dehydrogenase and hydrogenase enzymes, respectively. Reducing equivalents can come in the form of NADH, NADPH, FADH, reduced quinones, reduced ferredoxins, thioredoxins, and reduced flavodoxins. The reducing equivalents, particularly NADH, NADPH, and reduced ferredoxin, can serve as cofactors for the RTCA cycle enzymes, for example, malate dehydrogenase, fumarate reductase, alpha-ketoglutarate:ferredoxin oxidoreductase (alternatively known as 2-oxoglutarate:ferredoxin oxidoreductase, alpha-ketoglutarate synthase, or 2-oxoglutarate synthase), pyruvate:ferredoxin oxidoreductase and isocitrate dehydrogenase. The electrons from these reducing equivalents can alternatively pass through an ion-gradient producing electron transport chain where they are passed to an acceptor such as oxygen, nitrate, oxidized metal ions, protons, or an electrode. The ion-gradient can then be used for ATP generation via an ATP synthase or similar enzyme.
[0099] In some embodiments, a non-naturally occurring microbial organism has a 1,3-butanediol pathway and includes at least one exogenous nucleic acid encoding a 1,3-butanediol pathway enzyme expressed in a sufficient amount to produce 1,3-butanediol; wherein the non-naturally occurring microbial organism further includes:
[0100] A non-naturally occurring microbial organism having a 1,3-butanediol pathway, wherein said microbial organism comprises at least one exogenous nucleic acid encoding a 1,3-butanediol pathway enzyme expressed in a sufficient amount to produce 1,3-butanediol; said non-naturally occurring microbial organism further comprising:
[0101] (i) a reductive TCA pathway, wherein said microbial organism comprises at least one exogenous nucleic acid encoding a reductive TCA pathway enzyme selected from the group consisting of an ATP-citrate lyase, citrate lyase, a fumarate reductase, and an alpha-ketoglutarate:ferredoxin oxidoreductase;
[0102] (ii) a reductive TCA pathway, wherein said microbial organism comprises at least one exogenous nucleic acid encoding a reductive TCA pathway enzyme selected from the group consisting of a pyruvate:ferredoxin oxidoreductase, a phosphoenolpyruvate carboxylase, a phosphoenolpyruvate carboxykinase, a CO dehydrogenase, and an H.sub.2 hydrogenase; or
[0103] (iii) at least one exogenous nucleic acid encodes an enzyme selected from the group consisting of a CO dehydrogenase, an H.sub.2 hydrogenase, and combinations thereof;
[0104] wherein said 1,3-butanediol pathway comprises a pathway selected from the group consisting of:
[0105] (a) (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP dehydrogenase; (3) a 2-amino-4-hydroxypentanoate aminotransferase or oxidoreductase (deaminating); (4) a 2-oxo-4-hydroxypentanoate decarboxylase; and (5) a 3-hydroxybutyraldehyde reductase;
[0106] (b) (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP aminotransferase or oxidoreductase (deaminating); (3) a 2,4-dioxopentanoate decarboxylase; (4) a 3-oxobutyraldehyde reductase (ketone reducing); and (5) a 3-hydroxybutyraldehyde reductase;
[0107] (c) (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP aminotransferase or oxidoreductase (deaminating); (3) a 2,4-dioxopentanoate decarboxylase; (4) a 3-oxobutyraldehyde reductase (aldehyde reducing); and (5) a 4-hydroxy-2-butanone reductase;
[0108] (d) (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP decarboxylase; (3) a 4-aminobutan-2-one aminotransferase or oxidoreductase (deaminating); (4) a 3-oxobutyraldehyde reductase (ketone reducing); and (5) a 3-hydroxybutyraldehyde reductase;
[0109] (e) (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP decarboxylase; (3) a 4-aminobutan-2-one aminotransferase or oxidoreductase (deaminating); (4) a 3-oxobutyraldehyde reductase (aldehyde reducing); and (5) a 4-hydroxy-2-butanone reductase;
[0110] (f) (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP decarboxylase; (3) a 4-aminobutan-2-one ammonia-lyase; (4) a butenone hydratase; and (5) a 4-hydroxy-2-butanone reductase;
[0111] (g) (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP ammonia-lyase; (3) an acetylacrylate decarboxylase; (4) a butenone hydratase; and (5) a 4-hydroxy-2-butanone reductase;
[0112] (h) (1) an acetoacetyl-CoA reductase (CoA-dependent, aldehyde forming); (2) a 3-oxobutyraldehyde reductase (ketone reducing); and (3) a 3-hydroxybutyraldehyde reductase;
[0113] (i) (1) an acetoacetyl-CoA reductase (CoA dependent, alcohol forming) and (2) a 4-hydroxy-2-butanone reductase;
[0114] (j) (1) an acetoacetyl-CoA reductase (CoA-dependent, aldehyde forming); (2) a 3-oxobutyraldehyde reductase (aldehyde reducing); and (3) a 4-hydroxy-2-butanone reductase;
[0115] (k) (1) an acetoacetyl-CoA reductase (ketone reducing) and (2) a 3-hydroxybutyryl-CoA reductase (alcohol forming);
[0116] (l) (1) an acetoacetyl-CoA reductase (ketone reducing); (2) a 3-hydroxybutyryl-CoA reductase (aldehyde forming); and (3) a 3-hydroxybutyraldehyde reductase;
[0117] (m) (1) a 4-hydroxybutyryl-CoA dehydratase; (2) a crotonase; and (3) a 3-hydroxybutyryl-CoA reductase (alcohol forming); and
[0118] (n) (1) a 4-hydroxybutyryl-CoA dehydratase; (2) a crotonase; (3) a 3-hydroxybutyryl-CoA reductase (aldehyde forming); and (4) a 3-hydroxybutyraldehyde reductase;
[0119] (o) (1) a succinyl-CoA transferase, a succinyl-CoA synthetase or a succinyl-CoA ligase, (2) a succinyl-CoA reductase (aldehyde forming), (3) a 4-hydroxybutyrate dehydrogenase, (4) a 4-hydroxybutyrate kinase, (5) a phosphotrans-4-hydroxybutyrylase, (6) a 4-hydroxybutyryl-CoA dehydratase, (7) a crotonase, (8) a 3-hydroxybutyryl-CoA reductase (aldehyde forming), and (9) a 3-hydroxybutanal reductase;
[0120] (p) (1) (i) an alpha-ketoglutarate decarboxylase, or (ii) (a) a glutamate dehydrogenase and/or a glutamate transaminase, (b) a glutamate decarboxylase, and (c) a 4-aminobutyrate dehydrogenase and/or a 4-aminobutyrate transaminase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyrate kinase, (4) a phosphotrans-4-hydroxybutyrylase, (5) a 4-hydroxybutyryl-CoA dehydratase; (6) a crotonase, (7) a 3-hydroxybutyryl-CoA reductase (aldehyde forming), and (8) a 3-hydroxybutanal reductase;
[0121] (q) (1) (i) an alpha-ketoglutarate decarboxylase, or (ii) (a) a glutamate dehydrogenase and/or a glutamate transaminase, (b) a glutamate decarboxylase, and (c) a 4-aminobutyrate dehydrogenase and/or a 4-aminobutyrate transaminase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (4) a 4-hydroxybutyryl-CoA dehydratase, (5) a crotonase, (6) a 3-hydroxybutyryl-CoA reductase (aldehyde forming), and (7) a 3-hydroxybutanal reductase;
[0122] (r) (1) (i) an alpha-ketoglutarate decarboxylase, or (ii) (a) a glutamate dehydrogenase and/or a glutamate transaminase, (b) a glutamate decarboxylase, and (c) a 4-aminobutyrate dehydrogenase and/or a 4-aminobutyrate transaminase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyrate kinase, (4) a phosphotrans-4-hydroxybutyrylase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, and (7) a 3-hydroxybutyryl-CoA reductase (alcohol forming);
[0123] (s) (1) (i) an alpha-ketoglutarate decarboxylase, or (ii) (a) a glutamate dehydrogenase and/or a glutamate transaminase, (b) a glutamate decarboxylase, and (c) a 4-aminobutyrate dehydrogenase and/or a 4-aminobutyrate transaminase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (4) a 4-hydroxybutyryl-CoA dehydratase, (5) a crotonase, and (6) a 3-hydroxybutyryl-CoA reductase (alcohol forming);
[0124] (t) (1) (i) an alpha-ketoglutarate decarboxylase, or (ii) (a) a glutamate dehydrogenase and/or a glutamate transaminase, (b) a glutamate decarboxylase, and (c) a 4-aminobutyrate dehydrogenase and/or a 4-aminobutyrate transaminase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyrate kinase, (4) a phosphotrans-4-hydroxybutyrylase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, (7) a 3-hydroxybutyryl-CoA hydrolase, transferase or synthetase, and (8) a 3-hydroxybutyrate reductase;
[0125] (u) (1) (i) an alpha-ketoglutarate decarboxylase, or (ii) (a) a glutamate dehydrogenase and/or a glutamate transaminase, (b) a glutamate decarboxylase, and (c) a 4-aminobutyrate dehydrogenase and/or a 4-aminobutyrate transaminase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (4) a 4-hydroxybutyryl-CoA dehydratase, (5) a crotonase, (6) a 3-hydroxybutyryl-CoA hydrolase, transferase or synthetase, and (7) a 3-hydroxybutyrate reductase.
[0126] (v) (1) a succinate reductase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyrate kinase, (4) a phosphotrans-4-hydroxybutyrylase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, (7) a 3-hydroxybutyryl-CoA reductase (aldehyde forming), and (8) a 3-hydroxybutanal reductase;
[0127] (w) (1) a succinate reductase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (4) a 4-hydroxybutyryl-CoA dehydratase, (5) a crotonase, (6) a 3-hydroxybutyryl-CoA reductase (aldehyde forming), (7) a 3-hydroxybutanal reductase;
[0128] (x) (1) a succinate reductase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyrate kinase, (4) a phosphotrans-4-hydroxybutyrylase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, and (7) a 3-hydroxybutyryl-CoA reductase (alcohol forming);
[0129] (y) (1) a succinate reductase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (4) a 4-hydroxybutyryl-CoA dehydratase, (5) a crotonase, and (6) a 3-hydroxybutyryl-CoA reductase (alcohol forming);
[0130] (z) (1) a succinate reductase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyrate kinase, (4) a phosphotrans-4-hydroxybutyrylase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, (7) a 3-hydroxybutyryl-CoA hydrolase, transferase or synthetase, and (8) a 3-hydroxybutyrate reductase;
[0131] (aa) (1) a succinate reductase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (4) a 4-hydroxybutyryl-CoA dehydratase, (5) a crotonase, (6) a 3-hydroxybutyryl-CoA hydrolase, transferase or synthetase, and (7) a 3-hydroxybutyrate reductase;
[0132] (bb) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (aldehyde forming), (3) a 4-hydroxybutyrate dehydrogenase, (5) a 4-hydroxybutyrate kinase, (6) a pPhosphotrans-4-hydroxybutyrylase, (7) a 4-hydroxybutyryl-CoA dehydratase, (8) a crotonase, and (9) a 3-hydroxybutyryl-CoA reductase (alcohol forming);
[0133] (cc) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (aldehyde forming), (3) a 4-hydroxybutyrate dehydrogenase, (4) a 4-hydroxybutyrate kinase, (5) a phosphotrans-4-hydroxybutyrylase, (6) a 4-hydroxybutyryl-CoA dehydratase, (7) a crotonase, (8) a 3-hydroxybutyryl-CoA hydrolase, transferase or synthetase, and (9) a 3-hydroxybutyrate reductase;
[0134] (dd) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (alcohol forming), (3) a 4-hydroxybutyrate kinase, (4) a phosphotrans-4-hydroxybutyrylase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, (7) a 3-hydroxybutyryl-CoA reductase (aldehyde forming), and (8) a 3-hydroxybutanal reductase;
[0135] (ee) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (alcohol forming), (3) a 4-hydroxybutyrate kinase, (4) a phosphotrans-4-hydroxybutyrylase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, and (7) a 3-hydroxybutyryl-CoA reductase (alcohol forming);
[0136] (ff) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (alcohol forming), (3) a 4-hydroxybutyrate kinase, (4) a phosphotrans-4-hydroxybutyrylase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, (7) a 3-hydroxybutyryl-CoA hydrolase, transferase or synthetase, and (8) a 3-hydroxybutyrate reductase;
[0137] (gg) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (aldehyde forming), (3) a 4-hydroxybutyrate dehydrogenase, (4) a 4-hydroxybutyryl-CoA transferase, or 4-hydroxybutyryl-CoA synthetase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, (7) a 3-hydroxybutyryl-CoA reductase (aldehyde forming), and (8) a 3-hydroxybutanal reductase;
[0138] (hh) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (aldehyde limning), (3) a 4-hydroxybutyrate dehydrogenase, (4) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, and (7) a 3-hydroxybutyryl-CoA reductase (alcohol forming);
[0139] (ii) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (aldehyde forming), (3) a 4-hydroxybutyrate dehydrogenase, (4) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, (7) a 3-hydroxybutyryl-CoA hydrolase, transferase or synthetase, (8) a 3-hydroxybutyrate reductase;
[0140] (jj) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (alcohol forming), (3) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (4) a 4-hydroxybutyryl-CoA dehydratase, (5) a crotonase, (6) a 3-hydroxybutyryl-CoA reductase (aldehyde forming), and (7) a 3-hydroxybutanal reductase;
[0141] (kk) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (alcohol forming), (3) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (4) a 4-hydroxybutyryl-CoA dehydratase, (5) a crotonase, and (6) a 3-hydroxybutyryl-CoA reductase (alcohol forming); and
[0142] (ll) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (alcohol forming), (3) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, (7) a 3-hydroxybutyryl-CoA hydrolase, transferase or synthetase, and (8) a 3-hydroxybutyrate reductase.
[0143] In some embodiments, the non-naturally occurring microbial organism (e.g., having pathway (i)) further includes an exogenous nucleic acid encoding an enzyme selected from a pyruvate:ferredoxin oxidoreductase, an aconitase, an isocitrate dehydrogenase, a succinyl-CoA synthetase, a succinyl-CoA transferase, a fumarase, a malate dehydrogenase, an acetate kinase, a phosphotransacetylase, an acetyl-CoA synthetase, an NAD(P)H:ferredoxin oxidoreductase, ferredoxin, and combinations thereof.
[0144] In some embodiments, the non-naturally occurring microbial organism (e.g., having pathway (ii)) further includes an exogenous nucleic acid encoding an enzyme selected from an aconitase, an isocitrate dehydrogenase, a succinyl-CoA synthetase, a succinyl-CoA transferase, a fumarase, a malate dehydrogenase, and combinations thereof.
[0145] In some embodiments, the non-naturally occurring microbial organism includes two, three, four, five, six, seven, eight or nine exogenous nucleic acids, each encoding a 1,3-BDO pathway enzyme.
[0146] In some embodiments, the non-naturally occurring microbial organism comprises exogenous nucleic acids encoding each of the enzymes of at least one of the 1,3-butanediol pathways selected from the group consisting of (a)-(ll).
[0147] In some embodiments, the non-naturally occurring microbial organism has at least one exogenous nucleic acid is a heterologous nucleic acid.
[0148] In some embodiments, the non-naturally occurring microbial organism is in a substantially anaerobic culture medium.
[0149] The reductive TCA cycle was first reported in the green sulfur photosynthetic bacterium Chlorobium limicola (Evans et al., Proc. Natl. Acad. Sci. U.S.A. 55:928-934 (1966)). Similar pathways have been characterized in some prokaryotes (proteobacteria, green sulfur bacteria and thermophillic Knallgas bacteria) and sulfur-dependent archaea (Hugler et al., J. Bacteriol. 187:3020-3027 (2005; Hugler et al., Environ. Microbiol. 9:81-92 (2007). In some cases, reductive and oxidative (Krebs) TCA cycles are present in the same organism (Hugler et al., supra (2007); Siebers et al., J. Bacteriol. 186:2179-2194 (2004)). Some methanogens and obligate anaerobes possess incomplete oxidative or reductive TCA cycles that may function to synthesize biosynthetic intermediates (Ekiel et al., J. Bacteriol. 162:905-908 (1985); Wood et al., FEMS Microbiol. Rev. 28:335-352 (2004)).
[0150] The key carbon-fixing enzymes of the reductive TCA cycle are alpha-ketoglutarate:ferredoxin oxidoreductase, pyruvate:ferredoxin oxidoreductase and isocitrate dehydrogenase. Additional carbon may be fixed during the conversion of phosphoenolpyruvate to oxaloacetate by phosphoenolpyruvate carboxylase or phosphoenolpyruvate carboxykinase or during the conversion of pyruvate to malate by malic enzyme and during the conversion of pyruvate to oxaloacetate by pyruvate carboxylase.
[0151] Many of the enzymes in the TCA cycle are reversible and can catalyze reactions in the reductive and oxidative directions. However, some TCA cycle reactions are irreversible in vivo and thus different enzymes are used to catalyze these reactions in the directions required for the reverse TCA cycle. These reactions are: (1) conversion of citrate to oxaloacetate and acetyl-CoA, (2) conversion of fumarate to succinate, and (3) conversion of succinyl-CoA to alpha-ketoglutarate. In the TCA cycle, citrate is formed from the condensation of oxaloacetate and acetyl-CoA. The reverse reaction, cleavage of citrate to oxaloacetate and acetyl-CoA, is ATP-dependent and catalyzed by ATP-citrate lyase, or citryl-CoA synthetase and citryl-CoA lyase. Alternatively, citrate lyase can be coupled to acetyl-CoA synthetase, an acetyl-CoA transferase, or phosphotransacetylase and acetate kinase to form acetyl-CoA and oxaloacetate from citrate. The conversion of succinate to fumarate is catalyzed by succinate dehydrogenase while the reverse reaction is catalyzed by fumarate reductase. In the TCA cycle succinyl-CoA is formed from the NAD(P).sup.+ dependent decarboxylation of alpha-ketoglutarate by the alpha-ketoglutarate dehydrogenase complex. The reverse reaction is catalyzed by alpha-ketoglutarate:ferredoxin oxidoreductase.
[0152] An organism capable of utilizing the reverse tricarboxylic acid cycle to enable production of acetyl-CoA-derived products on 1) CO, 2) CO.sub.2 and H.sub.2, 3) CO and CO.sub.2, 4) synthesis gas comprising CO and H.sub.2, and 5) synthesis gas or other gaseous carbon sources comprising CO, CO.sub.2, and H.sub.2 can include any of the following enzyme activities: ATP-citrate lyase, citrate lyase, aconitase, isocitrate dehydrogenase, alpha-ketoglutarate:ferredoxin oxidoreductase, succinyl-CoA synthetase, succinyl-CoA transferase, fumarate reductase, fumarase, malate dehydrogenase, acetate kinase, phosphotransacetylase, acetyl-CoA synthetase, acetyl-CoA transferase, pyruvate:ferredoxin oxidoreductase, NAD(P)H:ferredoxin oxidoreductase, carbon monoxide dehydrogenase, hydrogenase, and ferredoxin (see FIGS. 8A and 9A). Enzymes and the corresponding genes required for these activities are described herein below.
[0153] Carbon from syngas or other gaseous carbon sources can be fixed via the reverse TCA cycle and components thereof. Specifically, the combination of certain carbon gas-utilization pathway components with the pathways for formation of 1,3-butanediol from acetyl-CoA results in high yields of these products by providing an efficient mechanism for fixing the carbon present in carbon dioxide, fed exogenously or produced endogenously from CO, into acetyl-CoA.
[0154] In some embodiments, a 1,3-butanediol pathway in a non-naturally occurring microbial organism of the invention can utilize any combination of (1) CO, (2) CO.sub.2, (3) H.sub.2, or mixtures thereof to enhance the yields of biosynthetic steps involving reduction, including addition to driving the reductive TCA cycle.
[0155] In some embodiments a non-naturally occurring microbial organism having a 1,3-butanediol pathway includes at least one exogenous nucleic acid encoding a reductive TCA pathway enzyme. The at least one exogenous nucleic acid is selected from an ATP-citrate lyase, citrate lyase, a fumarate reductase, an isocitrate dehydrogenase, an aconitase, an isocitrate dehydrogenase, and an alpha-ketoglutarate:ferredoxin oxidoreductase; and at least one exogenous enzyme selected from a carbon monoxide dehydrogenase, a hydrogenase, a NAD(P)H:ferredoxin oxidoreductase, and a ferredoxin, expressed in a sufficient amount to allow the utilization of (1) CO, (2) CO.sub.2, (3) H.sub.2, (4) CO.sub.2 and H.sub.2, (5) CO and CO.sub.2, (6) CO and H.sub.2, or (7) CO, CO.sub.2, and H.sub.2.
[0156] In some embodiments a method includes culturing a non-naturally occurring microbial organism having a 1,3-butanediol pathway also comprising at least one exogenous nucleic acid encoding a reductive TCA pathway enzyme. The at least one exogenous nucleic acid is selected from an ATP-citrate lyase, citrate lyase, a fumarate reductase, an isocitrate dehydrogenase, an aconitase, an isocitrate dehydrogenase, and an alpha-ketoglutarate:ferredoxin oxidoreductase. Additionally, such an organism can also include at least one exogenous enzyme selected from a carbon monoxide dehydrogenase, a hydrogenase, a NAD(P)H:ferredoxin oxidoreductase, and a ferredoxin, expressed in a sufficient amount to allow the utilization of (1) CO, (2) CO.sub.2, (3) H.sub.2, (4) CO.sub.2 and H.sub.2, (5) CO and CO.sub.2, (6) CO and H.sub.2, or (7) CO, CO.sub.2, and H.sub.2 to produce a product.
[0157] In some embodiments a non-naturally occurring microbial organism having a 1,3-butanediol pathway further includes at least one exogenous nucleic acid encoding a reductive TCA pathway enzyme expressed in a sufficient amount to enhance carbon flux through acetyl-CoA. The at least one exogenous nucleic acid is selected from an ATP-citrate lyase, citrate lyase, a fumarate reductase, an isocitrate dehydrogenase, an aconitase, an isocitrate dehydrogenase, a pyruvate:ferredoxin oxidoreductase and an alpha-ketoglutarate:ferredoxin oxidoreductase.
[0158] In some embodiments a non-naturally occurring microbial organism having a 1,3-butanediol pathway includes at least one exogenous nucleic acid encoding an enzyme expressed in a sufficient amount to enhance the availability of reducing equivalents in the presence of carbon monoxide and/or hydrogen, thereby increasing the yield of redox-limited products via carbohydrate-based carbon feedstock. The at least one exogenous nucleic acid is selected from a carbon monoxide dehydrogenase, a hydrogenase, an NAD(P)H:ferredoxin oxidoreductase, and a ferredoxin. In some embodiments, the present invention provides a method for enhancing the availability of reducing equivalents in the presence of carbon monoxide or hydrogen thereby increasing the yield of redox-limited products via carbohydrate-based carbon feedstock, such as sugars or gaseous carbon sources, the method includes culturing this non-naturally occurring microbial organism under conditions and for a sufficient period of time to produce 1,3-butanediol.
[0159] In some embodiments, the non-naturally occurring microbial organism having a 1,3-butanediol pathway includes two exogenous nucleic acids each encoding a reductive TCA pathway enzyme. In some embodiments, the non-naturally occurring microbial organism having a 1,3-butanediol pathway includes three exogenous nucleic acids each encoding a reductive TCA pathway enzyme. In some embodiments, the non-naturally occurring microbial organism includes three exogenous nucleic acids encoding an ATP-citrate lyase, a fumarate reductase, and an alpha-ketoglutarate:ferredoxin oxidoreductase. In some embodiments, any one of the three exogenous nucleic acids can be an isocitrate dehydrogenase. In some embodiments, the non-naturally occurring microbial organism includes three exogenous nucleic acids encoding a citrate lyase, a fumarate reductase, and an alpha-ketoglutarate:ferredoxin oxidoreductase. In some embodiments, the non-naturally occurring microbial organism includes four exogenous nucleic acids encoding a pyruvate:ferredoxin oxidoreductase; a phosphoenolpyruvate carboxylase or a phosphoenolpyruvate carboxykinase, a CO dehydrogenase; and an H2 hydrogenase. In some embodiments, the non-naturally occurring microbial organism includes two exogenous nucleic acids encoding a CO dehydrogenase and an H2 hydrogenase.
[0160] In some embodiments, the non-naturally occurring microbial organisms having a 1,3-butanediol pathway further include an exogenous nucleic acid encoding an enzyme selected from a pyruvate:ferredoxin oxidoreductase, an aconitase, an isocitrate dehydrogenase, a succinyl-CoA synthetase, a succinyl-CoA transferase, a fumarase, a malate dehydrogenase, an acetate kinase, a phosphotransacetylase, an acetyl-CoA synthetase, an NAD(P)H:ferredoxin oxidoreductase, and combinations thereof.
[0161] In some embodiments, the non-naturally occurring microbial organism having a 1,3-butanediol pathway further includes an exogenous nucleic acid encoding an enzyme selected from carbon monoxide dehydrogenase, acetyl-CoA synthase, ferredoxin, NAD(P)H:ferredoxin oxidoreductase and combinations thereof.
[0162] In some embodiments, the non-naturally occurring microbial organism having a 1,3-butanediol pathway utilizes a carbon feedstock selected from (1) CO, (2) CO.sub.2, (3) CO.sub.2 and H.sub.2, (4) CO and H.sub.2, or (5) CO, CO.sub.2, and H.sub.2. In some embodiments, the non-naturally occurring microbial organism having a 1,3-butanediol pathway utilizes hydrogen for reducing equivalents. In some embodiments, the non-naturally occurring microbial organism having a 1,3-butanediol pathway utilizes CO for reducing equivalents. In some embodiments, the non-naturally occurring microbial organism having a 1,3-butanediol pathway utilizes combinations of CO and hydrogen for reducing equivalents.
[0163] In some embodiments, the non-naturally occurring microbial organism having a 1,3-butanediol pathway further includes one or more nucleic acids encoding an enzyme selected from a phosphoenolpyruvate carboxylase, a phosphoenolpyruvate carboxykinase, a pyruvate carboxylase, and a malic enzyme.
[0164] In some embodiments, the non-naturally occurring microbial organism having a 1,3-butanediol pathway further includes one or more nucleic acids encoding an enzyme selected from a malate dehydrogenase, a fumarase, a fumarate reductase, a succinyl-CoA synthetase, and a succinyl-CoA transferase.
[0165] In some embodiments, the non-naturally occurring microbial organism having a 1,3-butanediol pathway further includes at least one exogenous nucleic acid encoding a citrate lyase, an ATP-citrate lyase, a citryl-CoA synthetase, a citryl-CoA lyase, an aconitase, an isocitrate dehydrogenase, a succinyl-CoA synthetase, a succinyl-CoA transferase, a fumarase, a malate dehydrogenase, an acetate kinase, a phosphotransacetylase, an acetyl-CoA synthetase, an acetyl-CoA transferase, and a ferredoxin.
[0166] It is understood by those skilled in the art that the above-described pathways for increasing product yield can be combined with any of the pathways disclosed herein, including those pathways depicted in the figures. One skilled in the art will understand that, depending on the pathway to a desired product and the precursors and intermediates of that pathway, a particular pathway for improving product yield, as discussed herein above and in the examples, or combination of such pathways, can be used in combination with a pathway to a desired product to increase the yield of that product or a pathway intermediate.
[0167] In an additional embodiment, the invention provides a non-naturally occurring microbial organism having a 1,3-butanediol pathway, wherein the non-naturally occurring microbial organism comprises at least one exogenous nucleic acid encoding an enzyme or protein that converts a substrate to a product selected from the group consisting of pyruvate to alanine, alanine to 2-amino-4-oxopentanoate, 2-amino-4-oxopentanoate to 2-amino-4-hydroxypentanoate, 2-amino-4-oxopentanoate to 2,4-dioxopentanoate, 2-amino-4-oxopentanoate to 4-aminobutan-2-one, 2-amino-4-oxopentanoate to acetylacrylate, 2-amino-4-hydroxypentanoate to 2-oxo-4-hydroxypentanoate, 2-oxo-4-hydroxypentanoate to 3-hydroxybutyraldehyde, 2,4-dioxopentanoate to 3-oxobutyraldehyde, 3-oxobutyraldehyde to 3-hydroxybutryaldehyde, 3-oxobutyraldehdye to 4-hydroxy-2-butanone, 4-aminobutan-2-one to 3-oxobutyraldehyde, 4-aminobutan-2-one to butanone, butanone to 4-hydroxy-2-butanone and acetylacrylate to butenone. One skilled in the art will understand that these are merely exemplary and that any of the substrate-product pairs disclosed herein suitable to produce a desired product and for which an appropriate activity is available for the conversion of the substrate to the product can be readily determined by one skilled in the art based on the teachings herein. Thus, the invention provides a non-naturally occurring microbial organism containing at least one exogenous nucleic acid encoding an enzyme or protein, where the enzyme or protein converts the substrates and products of a 1,3-butanediol pathway, such as that shown in FIG. 8B.
[0168] In an additional embodiment, the invention provides a non-naturally occurring microbial organism having a 1,3-butanediol pathway, wherein the non-naturally occurring microbial organism comprises at least one exogenous nucleic acid encoding an enzyme or protein that converts a substrate to a product selected from the group consisting of succinate to succinyl-CoA, succinate to succinic semialdehyde, succinyl-CoA to succinic semialdehyde, alpha-ketoglutarate to succinic semialdehyde, succinyl-CoA to 4-hydroxybutyrate, succinic semialdehyde to 4-hydroxybutyrate, 4-hydroxybutyrate to 4-hydroxybutyryl phosphate, 4-hydroxybutyrate to 4-hydroxybutyryl-CoA, 4-hydroxybutyryl phosphate to 4-hydroxybutyryl CoA, 4-hydroxybutyryl-CoA to crotonyl-CoA, crotonyl-CoA to 3-hydroxybutyryl-CoA, 3-hydroxybutyryl-CoA to 3-hydroxybutyrate, 3-hydroxybutyrate to 1,3-butanediol, 3-hydroxybutyryl-CoA to 1,3-butanediol, 3-hydroxybutyryl-CoA to 3-hydroxybutanal, 3-hydroxybutanal to 1,3-butanediol. One skilled in the art will understand that these are merely exemplary and that any of the substrate-product pairs disclosed herein suitable to produce a desired product and for which an appropriate activity is available for the conversion of the substrate to the product can be readily determined by one skilled in the art based on the teachings herein. Thus, the invention provides a non-naturally occurring microbial organism containing at least one exogenous nucleic acid encoding an enzyme or protein, where the enzyme or protein converts the substrates and products of a 1,3-butanediol pathway, such as that shown in FIG. 9B.
[0169] While generally described herein as a microbial organism that contains a 1,3-butanediol pathway, it is understood that the invention additionally provides a non-naturally occurring microbial organism comprising at least one exogenous nucleic acid encoding a 1,3-butanediol pathway enzyme expressed in a sufficient amount to produce an intermediate of a 1,3-butanediol pathway. For example, as disclosed herein, a 1,3-butanediol pathway is exemplified in FIGS. 1-3, 8A/B, and 9A/B. Therefore, in addition to a microbial organism containing a 1,3-butanediol pathway that produces 1,3-butanediol, the invention additionally provides a non-naturally occurring microbial organism comprising at least one exogenous nucleic acid encoding a 1,3-butanediol pathway enzyme, where the microbial organism produces a 1,3-butanediol pathway intermediate, for example, alanine, 2-amino-4-pentanoate, 2-amino-4-hydroxypentanoate, 2-oxo-4-hydroxypentanoate, 3-hydroxybutyraldehyde, 2,4-dioxopentanoate, 3-oxobutyraldehyde, 4-aminobutan-2-one, acetylacrylate, butanone, 4-hydroxy-2-butanone, succinic semialdehyde, 4-hydroxybutyrate, 4-hydroxybutyryl phosphate, 4-hydroxybutyryl-CoA, crotonyl-CoA, 3-hydroxybutyryl-CoA, 3-hydroxybutyrate, and 3-hydroxybutanal.
[0170] It is understood that any of the pathways disclosed herein, as described in the Examples and exemplified in the Figures, including the pathways of FIGS. 1-3, 8A/B, and 9A/B can be utilized to generate a non-naturally occurring microbial organism that produces any pathway intermediate or product, as desired. As disclosed herein, such a microbial organism that produces an intermediate can be used in combination with another microbial organism expressing downstream pathway enzymes to produce a desired product. However, it is understood that a non-naturally occurring microbial organism that produces a 1,3-butanediol pathway intermediate can be utilized to produce the intermediate as a desired product.
[0171] Successfully engineering any of these pathways entails identifying an appropriate set of enzymes with sufficient activity and specificity, cloning their corresponding genes into a production host, optimizing fermentation conditions, and assaying for product formation following fermentation. To engineer a production host for the production of any of the aforementioned products, one or more exogenous DNA sequence(s) can be expressed in microorganisms. In addition, the microorganisms can have endogenous gene(s) functionally deleted. These modifications will enable the production of 1,3-BDO using renewable feedstocks.
[0172] Below, are described a number of biochemically characterized genes capable of encoding enzymes that catalyze each of the steps shown in FIGS. 1, 2 3, 8B, and 9B. Although we describe this method for E. coli, one skilled in the art can apply these teachings to essentially any other organism. Specifically, genes are listed that are native to E. coli in addition to genes in other organisms that can be applied to catalyze the appropriate transformations when properly cloned and expressed.
[0173] The invention is described herein with general reference to the metabolic reaction, reactant or product thereof, or with specific reference to one or more nucleic acids or genes encoding an enzyme associated with or catalyzing, or a protein associated with, the referenced metabolic reaction, reactant or product. Unless otherwise expressly stated herein, those skilled in the art will understand that reference to a reaction also constitutes reference to the reactants and products of the reaction. Similarly, unless otherwise expressly stated herein, reference to a reactant or product also references the reaction, and reference to any of these metabolic constituents also references the gene or genes encoding the enzymes that catalyze or proteins involved in the referenced reaction, reactant or product. Likewise, given the well known fields of metabolic biochemistry, enzymology and genomics, reference herein to a gene or encoding nucleic acid also constitutes a reference to the corresponding encoded enzyme and the reaction it catalyzes or a protein associated with the reaction as well as the reactants and products of the reaction.
[0174] As disclosed herein, intermediates en route to 1,3-butanediol can be carboxylic acids or CoA esters thereof, such as 4-hydroxy butyrate, 3-hydroxybutyrate, their CoA esters, as well as crotonyl-CoA. Any carboxylic acid intermediate can occur in various ionized forms, including fully protonated, partially protonated, and fully deprotonated forms. Accordingly, the suffix "-ate," or the acid form, can be used interchangeably to describe both the free acid form as well as any deprotonated form, in particular since the ionized form is known to depend on the pH in which the compound is found. It is understood that carboxylate intermediates includes ester forms of carboxylate products or pathway intermediates, such as O-carboxylate and S-carboxylate esters. O- and S-carboxylates can include lower alkyl, that is C1 to C6, branched or straight chain carboxylates. Some such O- or S-carboxylates include, without limitation, methyl, ethyl, n-propyl, n-butyl, i-propyl, sec-butyl, and tert-butyl, pentyl, hexyl O- or S-carboxylates, any of which can further possess an unsaturation, providing for example, propenyl, butenyl, pentyl, and hexenyl O- or S-carboxylates. O-carboxylates can be the product of a biosynthetic pathway. Exemplary O-carboxylates accessed via biosynthetic pathways can include, without limitation, methyl 4-hydroxybutyrate, methyl-3-hydroxybutyrate, ethyl 4-hydroxybutyrate, ethyl 3-hydroxybutyrate, n-propyl 4-hydroxybutyrate, and n-propyl 3-hydroxybutyrate. Other biosynthetically accessible O-carboxylates can include medium to long chain groups, that is C7-C22, O-carboxylate esters derived from fatty alcohols, such heptyl, octyl, nonyl, decyl, undecyl, lauryl, tridecyl, myristyl, pentadecyl, cetyl, palmitolyl, heptadecyl, stearyl, nonadecyl, arachidyl, heneicosyl, and behenyl alcohols, any one of which can be optionally branched and/or contain unsaturations. O-carboxylate esters can also be accessed via a biochemical or chemical process, such as esterification of a free carboxylic acid product or transesterification of an O- or S-carboxylate. S-carboxylates are exemplified by CoA S-esters, cysteinyl S-esters, alkylthioesters, and various aryl and heteroaryl thioesters.
[0175] All transformations depicted in FIGS. 1-3, 8B and 9B fall into the 8 general categories of transformations shown in Table 1. Below is described a number of biochemically characterized genes in each category. Specifically listed are genes that can be applied to catalyze the appropriate transformations in FIGS. 1-3, 8B, and 9B when properly cloned and expressed. Exemplary genes for each of the steps in FIGS. 1-3, 8B and 9B are provided further below in Tables 35-37.
[0176] Table 1 shows the enzyme types useful to convert common central metabolic intermediates into 1,3-butanediol. The first three digits of each label correspond to the first three Enzyme Commission number digits which denote the general type of transformation independent of substrate specificity.
TABLE-US-00001 TABLE 1 LABEL FUNCTION 1.1.1.a Oxidoreductase (ketone to hydroxyl or aldehyde to alcohol) 1.1.1.c Oxidoreductase (2 step, acyl-CoA to alcohol) 1.2.1.b Oxidoreductase (acyl-CoA to aldehyde) 1.4.1.a Oxidoreductase (deaminating) 2.3.1.b Acyltransferase 2.6.1.a Aminotransferase 4.1.1.a Carboxy-lyase 4.2.1.a Hydro-lyase 4.3.1.a Ammonia-lyase
[0177] Numerous transformation in FIGS. 1, 2 and 3 fall into the category of oxidoreductases that reduce an aldehyde to alcohol. For example, Steps D and P in FIG. 1 catalyzed by 3-oxobutyraldehyde reductase (aldehyde reducing) and 3-hydroxybutyraldehyde reductase respectively fall into this category. Similarly, Steps C and E in FIG. 2 catalyzed by 3-hydroxybutyraldehyde reductase and 3-oxobutyraldehdye reductase (aldehyde reducing) respectively are also oxidoreductases that convert the aldehyde functionality to alcohol. Pathways in FIG. 3 involve oxidoreductases such as 3-hydroxybutyraldehdye reductase in Step D.
[0178] Exemplary genes encoding enzymes that catalyze the conversion of an aldehyde to alcohol (i.e., alcohol dehydrogenase or equivalently aldehyde reductase) include alrA encoding a medium-chain alcohol dehydrogenase for C2-C14 (Tani et al., Appl. Environ. Microbiol., 66:5231-5235 (2000)), ADH2 from Saccharomyces cerevisiae (Atsumi et al., Nature, 451:86-89 (2008)), yqhD from E. coli which has preference for molecules longer than C3 (Sulzenbacher et al., J. of Molecular Biology, 342:489-502 (2004)), and bdh I and bdh II from C. acetobutylicum which converts butyraldehyde into butanol (Walter et al., J. of Bacteriology, 174:7149-7158 (1992)). The gene product of yqhD catalyzes the reduction of acetaldehyde, malondialdehyde, propionaldehyde, butyraldehyde, and acrolein using NADPH as the cofactor (Perez et al., J. Biol. Chem., 283:7346-7353 (2008)). The adhA gene product from Zymomonas mobilis has been demonstrated to have activity on a number of aldehydes including formaldehyde, acetaldehyde, propionaldehyde, butyraldehyde, and acrolein (Kinoshita et al., Appl. Microbiol. Biotechnol, 22:249-254 (1985)). Additional aldehyde reductase candidates are encoded by bdh in C. saccharoperbutylacetonicum and Cbei.sub.--1722, Cbei.sub.--2181 and Cbei.sub.--2421 in C. beijerinckii.
[0179] Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 2.
TABLE-US-00002 TABLE 2 Protein GENBANK ID GI NUMBER ORGANISM alrA BAB12273.1 9967138 Acinetobacter sp. strain M-1 ADH2 NP_014032.1 6323961 Saccharomyces cerevisiae yqhD NP_417484.1 16130909 Escherichia coli bdh I NP_349892.1 15896543 Clostridium acetobutylicum bdh II NP_349891.1 15896542 Clostridium acetobutylicum adhA YP_162971.1 56552132 Zymomonas mobilis bdh BAF45463.1 124221917 Clostridium saccharoperbutyl- acetonicum Cbei_1722 YP_001308850 150016596 Clostridium beijerinckii Cbei_2181 YP_001309304 150017050 Clostridium beijerinckii Cbei_2421 YP_001309535 150017281 Clostridium beijerinckii
[0180] Enzymes exhibiting 3-hydroxybutyraldehyde reductase activity (EC 1.1.1.61) also fall into this category. Such enzymes have been characterized in Ralstonia eutropha (Bravo et al., J. Forensic Sci., 49:379-387 (2004)), Clostridium kluyveri (Wolff et al., Protein Expr. Purif., 6:206-212 (1995)) and Arabidopsis thaliana (Breitkreuz et al., J. Biol. Chem., 278:41552-41556 (2003)). Yet another gene is the alcohol dehydrogenase adhI from Geobacillus thermoglucosidasius (Jeon et al., J. Biotechnol., 135:127-133 (2008)). Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 3.
TABLE-US-00003 TABLE 3 PROTEIN GENBANK ID GI NUMBER ORGANISM 4hbd YP_726053.1 113867564 Ralstonia eutropha H16 4hbd L21902.1 146348486 Clostridium kluyveri DSM 555 4hbd Q94B07 75249805 Arabidopsis thaliana adhI AAR91477.1 40795502 Geobacillus thermoglucosidasius M10EXG
[0181] Another exemplary enzyme is 3-hydroxyisobutyrate dehydrogenase which catalyzes the reversible oxidation of 3-hydroxyisobutyrate to methylmalonate semialdehyde. This enzyme participates in valine, leucine and isoleucine degradation and has been identified in bacteria, eukaryotes, and mammals. The enzyme encoded by P84067 from Thermus thermophilus HB8 has been structurally characterized (Lokanath et al., J. Mol. Biol., 352:905-917 (2005)). The reversibility of the human 3-hydroxyisobutyrate dehydrogenase was demonstrated using isotopically-labeled substrate (Manning et al., Biochem J., 231:481-484 (1985)). Additional genes encoding this enzyme include 3hidh in Homo sapiens (Hawes et al., Methods Enzymol, 324:218-228 (2000)) and Oryctolagus cuniculus (Hawes et al., supra; Chowdhury et al., Biosci. Biotechnol Biochem., 60:2043-2047 (1996)), mmsB in Pseudomonas aeruginosa and Pseudomonas putida (Liao et al., US patent 20050221466), and dhat in Pseudomonas putida (Aberhart et al., J. Chem. Soc., 6:1404-1406 (1979); Chowdhury et al., supra; Chowdhury et al., Biosci. Biotechnol Biochem., 67:438-441 (2003)). Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 4.
TABLE-US-00004 TABLE 4 PROTEIN GENBANK ID GI NUMBER ORGANISM P84067 P84067 75345323 Thermus thermophilus 3hidh P31937.2 12643395 Homo sapiens 3hidh P32185.1 416872 Oryctolagus cuniculus mmsB P28811.1 127211 Pseudomonas aeruginosa mmsB NP_746775.1 26991350 Pseudomonas putida dhat Q59477.1 2842618 Pseudomonas putida
[0182] Oxidoreductases that convert a ketone functionality to the corresponding hydroxyl group are also synthetic steps in the disclosed pathways. Notably, Reactions L, O and H in FIG. 1 catalyzed by AKP dehydrogenase, 3-oxobutyraldehyde reductase (ketone reducing), 4-hydroxy-2-butanone reductase respectively are transformations of this category. The two latter transformations are also encountered in Steps B and F respectively in FIG. 2. On similar lines, the acetoacetyl-CoA reductase in Step G of FIG. 2 reduces acetoacetyl-CoA to 3-hydroxybutyryl-CoA.
[0183] The reduction of 4-oxo group of 2-amino-4-oxopentanoate (AKP) by a dehydrogenase yields 2-amino-4-hydroxypentanoate (FIG. 1, step L). This reaction is very similar to the NAD(P)H-dependent reduction of aspartate semialdehyde to homoserine catalyzed by homoserine dehydrogenase (EC 1.1.13). In many organisms, including E. coli, homoserine dehydrogenase is a bifunctional enzyme that also catalyzes the ATP-dependent conversion of aspartate to aspartyl-4-phosphate (Starnes et al., Biochemistry, 11:677-687 (1973)). The functional domains are catalytically independent and connected by a linker region (Sibilli et al., J. Biol. Chem., 256:10228-10230 (1981)) and both domains are subject to allosteric inhibition by threonine. The homoserine dehydrogenase domain of the E. coli enzyme, encoded by thrA, was separated from the aspartate kinase domain, characterized, and found to exhibit high catalytic activity and reduced inhibition by threonine (James et al., Biochemistry, 41:3720-3725 (2002)). This can be applied to other bifunctional threonine kinases including, for example, hom1 of Lactobacillus plantarum (Cahyanto et al., Microbiology, 152:205-112 (2006)) and Arabidopsis thaliana. The monofunctional homoserine dehydrogenases encoded by hom6 in S. cerevisiae (Jacques et al., Biochem. Biophys. Acta, 1544:28-41 (2001)) and hom2 in Lactobacillus plantarum (Cahyanto et al., supra) have been functionally expressed and characterized in E. coli. Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 5.
TABLE-US-00005 TABLE 5 PROTEIN GENBANK ID GI NUMBER ORGANISM thrA AAC73113.1 1786183 Escherichia coli K12 akthr2 O81852 75100442 Arabidopsis thaliana hom6 CAA89671 1015880 Saccharomyces cerevisiae hom1 CAD64819 28271914 Lactobacillus plantarum hom2 CAD63186 28270285 Lactobacillus plantarum
[0184] Acetoacetyl-CoA reductase (Step G, FIG. 2) catalyzing the reduction of acetoacetyl-CoA to 3-hydroxybutyryl-CoA participates in the acetyl-CoA fermentation pathway to butyrate in several species of Clostridia and has been studied in detail (Jones et al., Microbiol. Rev., 50:484-524 (1986)). The enzyme from Clostridium acetobutylicum, encoded by hbd, has been cloned and functionally expressed in E. coli (Youngleson et al., J. Bacteriol., 171:6800-6807 (1989)). Additionally, subunits of two fatty acid oxidation complexes in E. coli, encoded by fadB and fadJ, function as 3-hydroxyacyl-CoA dehydrogenases (Binstock et al., Methods Enzymol., 71C:403-411 (1981)). Yet other genes demonstrated to reduce acetoacetyl-CoA to 3-hydroxybutyryl-CoA are phbB from Zoogloea ramigera (Ploux et al., Eur. J. Biochem., 174:177-182 (1988)) and phaB from Rhodobacter sphaeroides (Alber et al., Mol. Microbiol., 61:297-309 (2006)). The former gene is NADPH-dependent, its nucleotide sequence has been determined (Peoples et al., Mol. Microbiol. 3:349-357 (1989)) and the gene has been expressed in E. coli. Substrate specificity studies on the gene led to the conclusion that it could accept 3-oxopropionyl-CoA as a substrate besides acetoacetyl-CoA (Ploux et al., supra). Additional genes include Hbd1 (C-terminal domain) and Hbd2 (N-terminal domain) in Clostridium kluyveri (Hillmer and Gottschalk, Biochim. Biophys. Acta 3334:12-23 (1974)) and HSD17B10 in Bos taurus (Wakil et al., J. Biol. Chem., 207:631-638 (1954)). Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 6.
TABLE-US-00006 TABLE 6 Protein GENBANK ID GI NUMBER ORGANISM fadB P21177.2 119811 Escherichia coli fadJ P77399.1 3334437 Escherichia coli Hbd2 EDK34807.1 146348271 Clostridium kluyveri Hbd1 EDK32512.1 146345976 Clostridium kluyveri hbd P52041.2 Clostridium acetobutylicum HSD17B10 O02691.3 3183024 Bos Taurus phbB P23238.1 130017 Zoogloea ramigera phaB YP_353825.1 77464321 Rhodobacter sphaeroides
[0185] A number of similar enzymes have been found in other species of Clostridia and in Metallosphaera sedula (Berg et al., Archaea. Science, 318:1782-1786 (2007)) as shown in Table 7.
TABLE-US-00007 TABLE 7 Protein GenBank ID GI number Organism Hbd NP_349314.1 NP_349314.1 Clostridium acetobutylicum Hbd AAM14586.1 AAM14586.1 Clostridium beijerinckii Msed_1423 YP_001191505 YP_001191505 Metallosphaera sedula Msed_0399 YP_001190500 YP_001190500 Metallosphaera sedula Msed_0389 YP_001190490 YP_001190490 Metallosphaera sedula Msed_1993 YP_001192057 YP_001192057 Metallosphaera sedula
[0186] An exemplary alcohol dehydrogenase that converts a ketone to a hydroxyl group is the secondary alcohol dehydrogenase that was shown to convert acetone to isopropanol in C. beijerinckii (Ismaiel et al., J. Bacteriol., 175:5097-5105 (1993)) and T. brockii (Lamed et al., Biochem. J., 195:183-190 (1981); Peretz et al., Biochemistry, 28:6549-6555 (1989)). The gene product of adhA from Pyrococcus furiosus, which exhibits maximum activity on 2-pentanol and pyruvaldehyde, was shown to have very broad specificity which includes isopropanol and acetone (Van der et al., Eur. J. Biochem., 268:3062-3068 (2001)). Yet another secondary alcohol dehydrogenase with activity on isopropanol and acetone is encoded by the gene product of adh-A from Rhodococcus ruber (Edegger et al., Chem. Commun. (Camb), 2402-2404 (2006); Kosjek et al., Biotechnol. Bioeng., 86:55-62 (2004)). These genes along with others are listed below in Table 8.
TABLE-US-00008 TABLE 8 Protein GenBank ID GI number Organism adh AAA23199.2 60592974 Clostridium beijerinckii NRRL B593 adh P14941.1 113443 Thermoanaerobacter brockii HTD4 adhA AAC25556 3288810 Pyrococcus furiosus adh-A CAD36475 21615553 Rhodococcus ruber
[0187] Alternatively, there exist several exemplary alcohol dehydrogenases that convert a ketone to a hydroxyl functional group. Two such enzymes from E. coli are encoded by malate dehydrogenase (mdh) and lactate dehydrogenase (ldhA). In addition, lactate dehydrogenase from Ralstonia eutropha has been shown to demonstrate high activities on substrates of various chain lengths such as lactate, 2-oxobutyrate, 2-oxopentanoate and 2-oxoglutarate (Steinbuchel et al., Eur. J. Biochem., 130:329-334 (1983)). Conversion of the oxo functionality to the hydroxyl group can also be catalyzed by 2-keto-1,3-butanediol reductase, an enzyme reported to be found in rat and in human placenta (Suda et al., Arch. Biochem. Biophys., 176:610-620 (1976); Suda et al., Biochem. Biophys. Res. Commun., 77:586-591 (1977)). All of these enzymes can provide a 3-oxobutyraldehyde reductase, and a 4-hydroxy-2-butanone reductase. An additional enzyme for these steps is the mitochondrial 3-hydroxybutyrate dehydrogenase (bdh) from the human heart which has been cloned and characterized (Marks et al., J. Biol. Chem. 267:15459-15463 (1992)). This enzyme is a dehydrogenase that operates on a 3-hydroxyacid. Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 9.
TABLE-US-00009 TABLE 9 Protein GenBank ID GI number Organism mdh AAC76268.1 1789632 Escherichia coli ldhA NP_415898.1 16129341 Escherichia coli ldh YP_725182.1 113866693 Ralstonia eutropha bdh AAA58352.1 177198 Homo sapiens
[0188] A number of organisms can catalyze the reduction of 4-hydroxy-2-butanone to 1,3-butanediol, including those belonging to the genus Bacillus, Brevibacterium, Candida, and Klebsiella among others, as described by Matsuyama et al. (1995).
[0189] Several transformations in FIGS. 2 and 3 rely on the two-step reduction of acyl-CoA to the corresponding alcohol. For example, Steps D and I in FIG. 2, involving the acetoacetyl-CoA reductase (CoA-dependent, alcohol forming) and 3-hydroxybutyryl-CoA reductase (alcohol forming), and Step E in FIG. 3 involving 3-hydroxybutyryl-CoA reductase (alcohol forming), shows such a transformation.
[0190] Exemplary two-step oxidoreductases that convert an acyl-CoA to alcohol include those that transform substrates such as acetyl-CoA to ethanol (e.g., adhE from E. coli (Kessler et al., FEBS. Lett., 281:59-63 (1991)) and butyryl-CoA to butanol (e.g. adhE2 from C. acetobutylicum (Fontaine et al., J. Bacteriol., 184:821-830 (2002)). In addition to reducing acetyl-CoA to ethanol, the enzyme encoded by adhE in Leuconostoc mesenteroides has been shown to oxidize the branched chain compound isobutyraldehyde to isobutyryl-CoA (Kazahaya et al., J. Gen. Appl. Microbiol., 18:43-55 (1972); Koo et al., Biotechnol. Lett., 27:505-510 (2005)). Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 10.
TABLE-US-00010 TABLE 10 Protein GenBank ID GI Number Organism adhE NP_415757.1 16129202 Escherichia coli adhE2 AAK09379.1 12958626 Clostridium acetobutylicum adhE AAV66076.1 55818563 Leuconostoc mesenteroides
[0191] Another exemplary enzyme can convert malonyl-CoA to 3-HP. An NADPH-dependent enzyme with this activity has characterized in Chloroflexus aurantiacus where it participates in the 3-hydroxypropionate cycle (Hugler et al., J. Bacteriol., 184:2404-2410 (2002); Strauss et al., Eur. J. Biochem., 215:633-643 (1993)). This enzyme, with a mass of 300 kDa, is highly substrate-specific and shows little sequence similarity to other known oxidoreductases (Hugler et al., supra). No enzymes in other organisms have been shown to catalyze this specific reaction; however there is bioinformatic evidence that other organisms can have similar pathways (Klatt et al., Environ. Microbiol., 9:2067-2078 (2007)). Enzymes in other organisms including Roseiflexus castenholzii, Erythrobacter sp. NAP1 and marine gamma proteobacterium HTCC2080 can be inferred by sequence similarity. Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 11.
TABLE-US-00011 TABLE 11 Protein GenBank ID GI Number Organism mcr AAS20429.1 42561982 Chloroflexus aurantiacus Rcas_2929 YP_001433009.1 156742880 Roseiflexus castenholzii NAP1_02720 ZP_01039179.1 85708113 Erythrobacter sp. NAP1 MGP2080_00535 ZP_01626393.1 119504313 marine gamma proteobacterium HTCC2080
[0192] Longer chain acyl-CoA molecules can be reduced by enzymes such as the jojoba (Simmondsia chinensis) FAR which encodes an alcohol-forming fatty acyl-CoA reductase. Its overexpression in E. coli resulted in FAR activity and the accumulation of fatty alcohol (Metz et al., Plant Physiology, 122:635-644 (2000)) (FAR, AAD38039.1, 5020215, Simmondsia chinensis).
[0193] The pathways disclosed herein involve numerous oxidoreductase-type transformations that convert an acyl-CoA to an aldehyde. Specifically, Steps A and H in FIG. 2 catalyzed by acetoacetyl-CoA reductase (aldehyde forming) and 3-hydroxybutyryl-CoA reductase (aldehyde forming), and Step C from FIG. 3 showing the transformation catalyzed by 3-hydroxybutyryl-CoA reductase.
[0194] Several acyl-CoA dehydrogenases are capable of reducing an acyl-CoA to its corresponding aldehyde. Exemplary genes that encode such enzymes include the Acinetobacter calcoaceticus acr1 encoding a fatty acyl-CoA reductase (Reiser et al., J. of Bacteriology, 179:2969-2975 (1997)), the Acinetobacter sp. M-1 fatty acyl-CoA reductase (Ishige et al., Appl. Environ. Microbiol., 68:1192-1195 (2002)), and a CoA- and NADP-dependent succinate semialdehyde dehydrogenase encoded by the sucD gene in Clostridium kluyveri (Sohling et al., J. Bacteriol., 178:871-880 (1996)). SucD of P. gingivalis is another succinate semialdehyde dehydrogenase (Takahashi et al., J. Bacteriol., 182:4704-4710 (2000)). The enzyme acylating acetaldehyde dehydrogenase in Pseudomonas sp, encoded by bphG, is yet another enzyme demonstrated to oxidize and acylate acetaldehyde, propionaldehyde, butyraldehyde, isobutyraldehyde and formaldehyde (Powlowski et al., J. Bacteriol., 175:377-385 (1993)). In addition to reducing acetyl-CoA to ethanol, the enzyme encoded by adhE in Leuconostoc mesenteroides has been shown to oxidize the branched chain compound isobutyraldehyde to isobutyryl-CoA (Kazahaya et al., supra; Koo et al., supra). Butyraldehyde dehydrogenase catalyzes a similar reaction, conversion of butyryl-CoA to butyraldehyde, in solventogenic organisms such as Clostridium saccharoperbutylacetonicum (Kosaka et al., Biosci. Biotechnol. Biochem., 71:58-61 (2007)). Additional aldehyde dehydrogenase enzyme candidates are found in Desulfatibacillum alkenivorans, Citrobacter koseri, Salmonella enterica, Lactobacillus brevis and Bacillus selenitireducens. Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 12.
TABLE-US-00012 TABLE 12 Protein GenBank ID GI Number Organism acr1 YP_047869.1 50086359 Acinetobacter calcoaceticus acr1 AAC45217 1684886 Acinetobacter baylyi acr1 BAB85476.1 18857901 Acinetobacter sp. Strain M-1 sucD P38947.1 172046062 Clostridium kluyveri sucD NP_904963.1 34540484 Porphyromonas gingivalis bphG BAA03892.1 425213 Pseudomonas sp adhE AAV66076.1 55818563 Leuconostoc mesenteroides bld AAP42563.1 31075383 Clostridium saccharoperbutylacetonicum ald ACL06658.1 218764192 Desulfatibacillum alkenivorans AK-01 ald YP_001452373 157145054 Citrobacter koseri ATCC BAA- 895 pduP NP_460996.1 16765381 Salmonella enterica Typhimurium pduP ABJ64680.1 116099531 Lactobacillus brevis ATCC 367 BselDRAFT_1651 ZP_02169447 163762382 Bacillus selenitireducens MLS10
[0195] An additional enzyme type that converts an acyl-CoA to its corresponding aldehyde is malonyl-CoA reductase which transforms malonyl-CoA to malonic semialdehyde. Malonyl-CoA reductase is a key enzyme in autotrophic carbon fixation via the 3-hydroxypropionate cycle in thermoacidophilic archaeal bacteria (Berg et al., supra; Thauer, R. K., Science, 318:1732-1733 (2007)). The enzyme utilizes NADPH as a cofactor and has been characterized in Metallosphaera and Sulfolobus spp (Alber et al., J. Bacteriol., 188:8551-8559 (2006); Hugler et al., supra). The enzyme is encoded by Msed.sub.--0709 in Metallosphaera sedula (Alber et al., supra; Berg et al., supra). A gene encoding a malonyl-CoA reductase from Sulfolobus tokodaii was cloned and heterologously expressed in E. coli (Alber et al., supra). This enzyme has also been shown to catalyze the conversion of methylmalonyl-CoA to its corresponding aldehyde (2007). Although the aldehyde dehydrogenase functionality of these enzymes is similar to the bifunctional dehydrogenase from Chloroflexus aurantiacus, there is little sequence similarity. Both malonyl-CoA reductase enzymes have high sequence similarity to aspartate-semialdehyde dehydrogenase, an enzyme catalyzing the reduction and concurrent dephosphorylation of aspartyl-4-phosphate to aspartate semialdehyde. Additional genes can be found by sequence homology to proteins in other organisms including Sulfolobus solfataricus and Sulfolobus acidocaldarius and have been listed below. Yet another enzyme for CoA-acylating aldehyde dehydrogenase is the ald gene from Clostridium beijerinckii (Toth et al., Appl. Environ. Microbiol., 65:4973-4980 (1999)). This enzyme has been reported to reduce acetyl-CoA and butyryl-CoA to their corresponding aldehydes. This gene is very similar to eutE that encodes acetaldehyde dehydrogenase of Salmonella typhimurium and E. coli (Toth et al., supra). Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 13.
TABLE-US-00013 TABLE 13 Protein GenBank ID GI Number Organism MSED_0709 YP_001190808.1 146303492 Metallosphaera sedula mcr NP_378167.1 15922498 Sulfolobus tokodaii asd-2 NP_343563.1 15898958 Sulfolobus solfataricus Saci_2370 YP_256941.1 70608071 Sulfolobus acidocaldarius Ald AAT66436 9473535 Clostridium beijerinckii eutE AAA80209 687645 Salmonella typhimurium eutE P77445 2498347 Escherichia coli
[0196] The oxidative deamination of amino groups to their corresponding oxo groups is catalyzed by deaminating oxidoreductases in the EC class 1.4.1. Such enzymes utilize NAD.sup.+, NADP.sup.+ or FAD.sup.+ as acceptor. Enzymes in this class can convert 2-amino-4-oxopentanoate to 2,4-dioxopentanoate (FIG. 1, Step B), 2-amino-4-hydroxypentanoate to 2-oxo-4-hydroxypentanoate (FIG. 1, Step M) and 4-aminobutan-2-one to 3-oxobutyraldehyde (FIG. 1, Step K). Exemplary oxidoreductases operating on similar substrates include glutamate dehydrogenase (deaminating), encoded by gdhA, leucine dehydrogenase (deaminating), encoded by ldh, and aspartate dehydrogenase (deaminating), encoded by nadX. The gdhA gene product from Escherichia coli (McPherson et al., Nucleic. Acids Res. 11:5257-5266 (1983); Korber et al., J. Mol. Biol. 234:1270-1273 (1993)), gdh from Thermotoga maritima (Kort et al., Extremophiles 1:52-60 (1997); Lebbink et al., J. Mol. Biol. 280:287-296 (1998); Lebbink et al., J. Mol. Biol. 289:357-369 (1999)), and gdhA1 from Halobacterium salinarum (Ingoldsby et al., Gene. 349:237-244 (2005)) catalyze the reversible interconversion of glutamate to 2-oxoglutarate and ammonia, while favoring NADP(H), NAD(H), or both, respectively. Additional glutamate dehydrogenase gene candidates are found in Bacillus subtilis (Khan et al., Biosci. Biotechnol Biochem. 69:1861-1870 (2005)), Nicotiana tabacum (Purnell et al., Planta 222:167-180 (2005)), Oryza sativa (Abiko et al., Plant Cell Physiol 46:1724-1734 (2005)), Haloferax mediterranei (Diaz et al., Extremophiles. 10:105-115 (2006)), Halobactreium salinarum (Hayden et al., FEMS Microbiol Lett. 211:37-41 (2002)) and yeast (Roca et al., Appl Environ. Microbiol 69:4732-4736 (2003)). The Nicotiana tabacum enzyme is composed of alpha and beta subunits encoded by gdh1 and gdh2 (Purnell et al., Planta 222:167-180 (2005)). The ldh gene of Bacillus cereus encodes the LeuDH protein that accepts a wide of range of substrates including leucine, isoleucine, valine, and 2-aminobutanoate (Stoyan et al., J. Biotechnol 54:77-80 (1997); Ansorge et al., Biotechnol Bioeng. 68:557-562 (2000)). The nadX gene from Thermotoga maritime encoding for the aspartate dehydrogenase is involved in the biosynthesis of NAD (Yang et al., J. Biol. Chem. 278:8804-8808 (2003)). Data related to the sequences for each of these exemplary gene products can be found using the GenBank accession numbers shown below in Table 14.
TABLE-US-00014 TABLE 14 Protein GenBank ID GI Number Organism gdhA P00370 118547 Escherichia coli gdh P96110.4 6226595 Thermotoga maritima gdhA1 NP_279651.1 15789827 Halobacterium salinarum rocG NP_391659.1 16080831 Bacillus subtilis gdh1 AAR11534.1 38146335 Nicotiana tabacum gdh2 AAR11535.1 38146337 Nicotiana tabacum GDH Q852M0 75243660 Oryza sativa GDH Q977U6 74499858 Haloferax mediterranei GDH P29051 118549 Halobactreium salinarum GDH2 NP_010066.1 6319986 Saccharomyces cerevisiae ldh P0A393 61222614 Bacillus cereus nadX NP_229443.1 15644391 Thermotoga maritima
[0197] An enzyme with 4-aminobutan-2-one oxidoreductase (deaminating) activity is required to convert 4-aminobutan-2-one to its corresponding aldehyde (FIG. 1, Step K). Exemplary candidates include 3,5-diaminohexanoate dehydrogenase (EC 1.4.1.11) and lysine 6-dehydrogenase (EC 1.4.1.18). 3,5-Diaminohexanoate dehydrogenase interconverts 3-amino acids and 3-oxoacids and has been characterized in organisms that ferment lysine. The gene encoding 3,5-diaminohexanoate dehydrogenase, kdd, was recently identified in Fusobacterium nucleatum (Kreimeyer et al., J. Biol. Chem. 282:7191-7197 (2007)). The enzyme has been purified and characterized in other organisms (Baker et al., J Biol. Chem. 247:7724-7734 (1972); Baker et al., Biochemistry 13:292-299 (1974)) but the genes associated with these enzymes are not known. Candidates in other sequenced organisms can be inferred by sequence homology. Lysine 6-dehydrogenase, encoded by the lysDH genes, catalyzes the conversion of primary amines to their corresponding aldehydes. This enzyme naturally catalyzes the reversible oxidative deamination of the 6-amino group of L-lysine to form 2-aminoadipate-6-semialdehyde (Misono et al., J Bacteriol. 150:398-401 (1982)). Exemplary enzymes are found in Geobacillus stearothermophilus (Heydari et al., Appl Environ. Microbiol 70:937-942 (2004)), Agrobacterium tumefaciens (Hashimoto et al., J Biochem. 106:76-80 (1989); Misono and Nagasaki, J Bacteriol. 150:398-401 (1982)), and Achromobacter denitrificans (Ruldeekulthamrong et al., BMB. Rep. 41:790-795 (2008)). Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 15.
TABLE-US-00015 TABLE 15 Protein GenBank ID GI Number Organism kdd AAL93966.1 19713113 Fusobacterium nucleatum lysDH BAB39707 13429872 Geobacillus stearothermophilus lysDH NP_353966 15888285 Agrobacterium tumefaciens lysDH AAZ94428 74026644 Achromobacter denitrificans
[0198] 2-Amino-4-oxopentanoate (AKP) thiolase or AKP thiolase (AKPT) (Step 1, FIG. 1) is a pyridoxal phosphate-dependent enzyme participating in ornithine degradation in Clostridium sticklandii (Jeng et al., A. Biochemistry, 13:2898-2903 (1974); Kenklies et al., Microbiology, 145:819-826 (1999)). A gene cluster encoding the alpha and beta subunits of AKPT (or-2 (ortA) and or-3 (ortB)) was recently identified and the biochemical properties of the enzyme were characterized (Fonknechten et al., J. Bacteriol., In Press (2009)). The enzyme is capable of operating in both directions and reacts with the D-isomer of alanine. Enzyme engineering can be performed to optimize function with L-alanine as a substrate. AKPT from Clostridium sticklandii has been characterized but its protein sequence has not yet been published. Enzymes with high sequence homology are found in Clostridium difficile, Alkaliphilus metalliredigenes QYF, Thermoanaerobacter sp. X514, and Thermoanaerobacter tengcongensis MB4 (Fonknechten et al, supra). Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 16.
TABLE-US-00016 TABLE 16 Protein GenBank ID GI Number Organism ortA (A) YP_001086914.1 126698017 Clostridium difficile 630 ortB (.beta.) YP_001086915.1 126698018 Clostridium difficile 630 Amet_2368 YP_001320181.1 150390132 Alkaliphilus (.alpha.) metalliredigenes QYF Amet_2369 YP_001320182.1 150390133 Alkaliphilus (.beta.) metalliredigenes QYF Teth514_1478 YP_001663101.1 167040116 Thermoanaerobacter (.alpha.) sp. X514 Teth514_1479 YP_001663102.1 167040117 Thermoanaerobacter (.beta.) sp. X514 TTE1235 (.alpha.) NP_622858.1 20807687 Thermoanaerobacter tengcongensis MB4 thrC (.beta.) NP_622859.1 20807688 Thermoanaerobacter tengcongensis MB4
[0199] The conversion of 2-amino-4-oxopentanoate (AKP) to 2,4-dioxopentanoate (Step B, FIG. 1) is accomplished by 2-amino-4-oxopentanoate aminotransferase or oxidoreductase (deaminating). Selection of an appropriate enzyme for this transformation is dependent on the stereochemistry of the substrate. For example, if the substrate is in the D-configuration, a D-amino acid aminotransferase (EC 2.6.1.21) can be utilized, whereas the L-stereoisomer can utilize an L-aminotransferase such as aspartate aminotransferase (EC 2.6.1.1).
[0200] Aspartate aminotransferase transfers an amino group from aspartate to alpha-ketoglutarate, forming glutamate and oxaloacetate. Aspartate is similar in structure to 2-amino-4-oxopentanoate. This conversion is catalyzed by, for example, the gene products of aspC from Escherichia coli (Yagi et al., FEBS Lett., 100:81-84 (1979); Yagi et al., Methods Enzymol., 133:83-89 (1985)), AAT2 from Saccharomyces cerevisiae (Yagi et al., J. Biochem., 92:35-43 (1982)) and ASP5 from Arabidopsis thaliana (Kwok et al., J. Exp. Bot., 55:595-604 (2004); De la et al., Plant J., 46:414-425 (2006); Wilkie et al., Protein Expr. Purif., 12:381-389 (1998)). The enzyme from Rattus norvegicus has been shown to transaminate alternate substrates such as 2-aminohexanedioic acid and 2,4-diaminobutyric acid (Recasens et al., Biochemistry, 19:4583-4589 (1980)). Aminotransferases that work on other amino-acid-like substrates can also catalyze this transformation. Valine aminotransferase catalyzes the conversion of valine and pyruvate to 2-ketoisovalerate and alanine. The E. coli gene, avtA, encodes one such enzyme (Whalen et al., J. Bacteriol., 150:739-746 (1982)). This gene product also catalyzes the amination of .alpha.-ketobutyrate to generate .alpha.-aminobutyrate, although the amine donor in this reaction has not been identified (Whalen et al., J. Bacteriol., 158:571-574 (1984)). An additional candidate is alpha-aminoadipate transaminase (EC 2.6.1.39), an enzyme that participates in lysine biosynthesis and degradation in some organisms. The enzyme from Thermus thermophilus, encoded by lysN, is active with several alternate substrates including oxaloacetate, 2-oxoisocaproate, 2-oxoisovalerate, and 2-oxo-3-methylvalerate (Miyazaki et al., Microbiol. 150:2327-2334 (2004)). A similar enzyme from Homo sapiens has been characterized (Okuno et al., Enz. Prot. 47:136-148 (1993)). Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 17.
TABLE-US-00017 TABLE 17 Protein GenBank ID GI Number Organism aspC NP_415448.1 16128895 Escherichia coli AAT2 P23542.3 1703040 Saccharomyces cerevisiae ASP5 P46248.2 20532373 Arabidopsis thaliana got2 P00507 112987 Rattus norvegicus avtA YP_026231.1 49176374 Escherichia coli lysN BAC76939.1 31096548 Thermus thermophilus AadAT-II Q8N5Z0.2 46395904 Homo sapiens
[0201] When the substrate is present as the D-stereoisomer, transamination can be catalyzed by D-aminotransferase (EC 2.6.1.21), also known as D-amino acid aminotransferase and D-alanine aminotransferase (DAAT). This class of enzymes is noted for its broad substrate specificity, which is species-specific. The D-aminotransferase from Bacillus species YM-1, encoded by dat, has been cloned, sequenced (Tanizawa et al., J. Biol. Chem., 264:2450-2454 (1989)) and the crystal structure has been solved (Peisach et al., Biochemistry, 37:4958-4967 (1998)). This enzyme has also been the subject of protein engineering studies to alter the substrate specificity (Gutierrez et al., Eur. J. Biochem, 267:7218-7223 (2000); Gutierrez et al., Protein Eng., 11:53-58 (1998)). Additional genes are found in Bacillus licheniformis ATCC 10716 (Taylor et al., Biochim. Biophys. Acta., 1350:38-40 (1997)), Staphylococcus haemolyticus (Pucci et al., J. Bacteriol., 177:336-342 (1995)) and Bacillus subtilis (Martinez-Carrion et al., J. Biol. Chem., 240:3538-3546 (1965)). Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 18.
TABLE-US-00018 TABLE 18 Protein GenBank ID GI Number Organism dat P19938 118222 Bacillus sp. YM-1 dat P54692 1706292 Bacillus licheniformis ATCC 10716 dat P54694 1706294 Staphylococcus haemolyticus dat O07597.1 3121979 Bacillus subtilis
[0202] In reaction K of FIG. 1, 4-aminobutan-2-one is transaminated to form 3-oxobutanal. This transformation can likely be catalyzed by an aminotransferase that interconverts terminal amines and aldehydes. Exemplary candidate enzymes are beta-alanine/alpha-ketoglutarate aminotransferase, GABA aminotransferase, 3-amino-2-methylpropionate transaminase, lysine-6-aminotransferase, 2,4-diaminobutanoate transaminase, putrescine aminotransferase and diamine aminotransferase.
[0203] Cargill has developed and patented a beta-alanine/alpha-ketoglutarate aminotransferase for producing 3-HP from beta-alanine via malonyl-semialdehyde (Chandra et al., ARch. Microbiol., 176:443-451 (2001)). The gene product of SkPYD4 in Saccharomyces kluyveri was also shown to preferentially use beta-alanine as the amino group donor (Aberhart et al., J. Chem. Soc. 6:1404-1406 (1979)). SkUGA1 encodes a homologue of Saccharomyces cerevisiae GABA aminotransferase, UGA1 (Ichikawa et al., J. Mol. Catalysis A-Chem., 256:106-112 (2006)), whereas SkPYD4 encodes an enzyme involved in both .beta.-alanine and GABA transamination (Aberthart et al., Supra). 3-amino-2-methylpropionate transaminase catalyzes the transformation from methylmalonate semialdehyde to 3-amino-2-methylpropionate. The enzyme has been characterized in Rattus norvegicus and Sus scrofa and is encoded by Abat (Chopra et al., Protein Expr. Purif, 25:533-540 (2002), Kuznetsova et al., FEMS Microbiol. Rev., 29:263-279 (2005)). Enzyme candidates in other organisms with high sequence homology to 3-amino-2-methylpropionate transaminase include Gta-1 in C. elegans and gabT in Bacillus subtilus. Additionally, one of the native GABA aminotransferases in E. coli, encoded by gene gabT, has been shown to have broad substrate specificity (Fontaine et al., J. Bacteriol., 184:821-830 (2002), Kanamasa et al., Appl. Microbiol Biotechnol., 80:223-229 (2008)). The gene puuE encodes the other 4-aminobutyrate transaminase in E. coli (Drummond et al., J. Biol. Chem., 235:318-325 (1960)).
[0204] Lysine-6-aminotransferase converts lysine to alpha-aminoadipate semialdehyde. Candidate enzymes have been characterized in Candida utilis (Hammer et al., J Basic Microbiol 32:21-27 (1992)), Flavobacterium lutescens (Fujii et al., J Biochem. 128:391-397 (2000)) and Streptomyces clavuligenus (Romero et al., J Ind. Microbiol Biotechnol 18:241-246 (1997)). A recombinant lysine-6-aminotransferase from S. clavuligenus was functionally expressed in E. coli (Tobin et al., J Bacteriol. 173:6223-6229 (1991)). The F. lutescens enzyme is specific to alpha-ketoglutarate as the amino acceptor (Soda et al., Biochemistry 7:4110-4119 (1968)). An enzyme with diaminobutanoate transaminase activity is encoded by the dat gene product in Acinetobacter baumanii (Ikai et al., J Bacteriol. 179:5118-5125 (1997)). In addition to its natural substrate, 2,4-diaminobutyrate, DAT transaminates the terminal amines of lysine, 4-aminobutyrate and ornithine. Candidate putrescine aminotransferase enzymes are encoded by ygjG in E. coli and spuC of Pseudomonas aeruginosa (Lu et al., J Bacteriol. 184:3765-3773 (2002)). The ygiG gene product reacts with the alternate substrates cadaverine, spermidine and 1,7-diaminoheptanoate (Samsonova et al., BMC. Microbiol 3:2 (2003); Kim, J Biol. Chem. 239:783-786 (1964)).
[0205] Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 19.
TABLE-US-00019 TABLE 19 Protein GenBank ID GI Number Organism SkyPYD4 ABF58893.1 98626772 Saccharomyces kluyveri SkUGA1 ABF58894.1 98626792 Saccharomyces kluyveri UGA1 NP_011533.1 6321456 Saccharomyces cerevisiae Abat P50554.3 122065191 Rattus norvegicus Abat P80147.2 120968 Sus scrofa Gta-1 Q21217.1 6016091 Caenorhabditis elegans gabT P94427.1 6016090 Bacillus subtilis gabT P22256.1 16130576 Escherichia coli K12 puuE NP_415818.1 16129263 Escherichia coli K12 lat BAB13756.1 10336502 Flavobacterium lutescens lat AAA26777.1 153343 Streptomyces clavuligenus dat P56744.1 6685373 Acinetobacter baumanii ygjG NP_417544 145698310 Escherichia coli spuC AAG03688 9946143 Pseudomonas aeruginosa
[0206] In FIG. 1, Step C, 2,4-dioxopentanoate is decarboxylated to form 3-oxobutyraldehyde by 2,4-dioxopentanoate decarboxylase. 2,4-dioxopentanoate is similar to the native substrates of pyruvate decarboxylase (EC 4.1.1.1) and benzoylformate decarboxylase (EC 4.1.1.7). Pyruvate decarboxylase (PDC), also termed keto-acid decarboxylase, is a key enzyme in alcoholic fermentation, catalyzing the decarboxylation of pyruvate to acetaldehyde. The enzyme from Saccharomyces cerevisiae has a broad substrate range for aliphatic 2-keto acids including 2-ketobutyrate, 2-ketovalerate, 3-hydroxypyruvate and 2-phenylpyruvate (Li et al., Biochemistry, 38:10004-10012 (1999)). This enzyme has been extensively studied, engineered for altered activity, and functionally expressed in E. coli (Killenberg-Jabs et al., Eur. J. Biochem., 268:1698-1704 (2001); Li et al., supra; Schure et al., Appl. Environ. Microbiol., 64:1303-1307 (1998)). The PDC from Zymomonas mobilus, encoded by pdc, also has a broad substrate range and has been a subject of directed engineering studies to alter the affinity for different substrates (Siegert et al., Protein Eng. Des. Sel., 18:345-357 (2005)). The crystal structure of this enzyme is available (Killenberg-Jabs, supra). Other well-characterized PDC enzymes include the enzymes from Acetobacter pasteurians (Chandra et al., Arch. Microbiol. 176:443-451 (2001)) and Kluyveromyces lactis (Krieger et al., Eur. J. Biochem., 269:3256-3263 (2002)). Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 20.
TABLE-US-00020 TABLE 20 Protein GenBank ID GI Number Organism pdc P06672.1 118391 Zymomonas mobilis pdc1 P06169 30923172 Saccharomyces cerevisiae pdc Q8L388 20385191 Acetobacter pasteurians pdc1 Q12629 52788279 Kluyveromyces lactis
[0207] Like PDC, benzoylformate decarboxylase (EC 4.1.1.7) has a broad substrate range and has been the target of enzyme engineering studies. The enzyme from Pseudomonas putida has been extensively studied and crystal structures of this enzyme are available (Polovnikova et al., Biochemistry 42:1820-1830 (2003); Hasson et al., Biochemistry, 37:9918-9930 (1998)). Site-directed mutagenesis of two residues in the active site of the Pseudomonas putida enzyme altered the affinity (Km) of naturally and non-naturally occurring substrates (Siegert et al., supra). The properties of this enzyme have been further modified by directed engineering (Lingen et al., Chembiochem, 4:721-726 (2003); Lingen et al., Protein Eng., 15:585-593 (2002)). The enzyme from Pseudomonas aeruginosa, encoded by mdlC, has also been characterized experimentally (Barrowman et al., FEMS Microbiology Letters, 34:57-60 (1986)). Additional genes from Pseudomonas stutzeri, Pseudomonas fluorescens and other organisms can be inferred by sequence homology or identified using a growth selection system developed in Pseudomonas putida (Henning et al., Appl. Environ. Microbiol., 72:7510-7517 (2006)). Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 21.
TABLE-US-00021 TABLE 21 Protein GenBank ID GI Number Organism mdlC P20906.2 3915757 Pseudomonas putida mdlC Q9HUR2.1 81539678 Pseudomonas aeruginosa dpgB ABN80423.1 126202187 Pseudomonas stutzeri ilvB-1 YP_260581.1 70730840 Pseudomonas fluorescens
[0208] A third enzyme capable of decarboxylating 2-oxoacids is alpha-ketoglutarate decarboxylase (KGD). The substrate range of this class of enzymes has not been studied to date. The KDC from Mycobacterium tuberculosis (Tian et al., Proc. Natl. Acad. Sci. USA, 102:10670-10675 (2005)) has been cloned and has been functionally expressed in E. coli at Genomatica. KDC enzyme activity has been detected in several species of Rhizobia including Bradyrhizobium japonicum and Mesorhizobium loti (Green et al., J. Bacteriol., 182:2838-2844 (2000)). Although the KDC-encoding gene(s) have not been isolated in these organisms, the genome sequences are available and several genes in each genome are annotated as putative KDCs. A KDC from Euglena gracilis has also been characterized but the gene associated with this activity has not been identified to date (Shigeoka et al., Arch. Biochem. Biophys., 288:22-28 (1991)). The first twenty amino acids starting from the N-terminus were sequenced MTYKAPVKDVKFLLDKVFKV (Shigeoka et al., supra). The gene can be identified by testing genes containing this N-terminal sequence for KDC activity. Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 22.
TABLE-US-00022 TABLE 22 Protein GenBank ID GI Number Organism kgd O50463.4 160395583 Mycobacterium tuberculosis kgd NP_767092.1 27375563 Bradyrhizobium japonicum USDA110 kgd NP_105204.1 13473636 Mesorhizobium loti
[0209] A fourth enzyme for catalyzing this step is the branched chain alpha-ketoacid decarboxylase (BCKA). This class of enzymes has been shown to act on a variety of compounds varying in chain length from 3 to 6 carbons (Oku et al., J. Biol. Chem., 263:18386-18396 (1988); Smit et al., Appl. Environ. Microbiol., 71:303-311 (2005)). The enzyme in Lactococcus lactis has been characterized on a variety of branched and linear substrates including 2-oxobutanoate, 2-oxohexanoate, 2-oxopentanoate, 3-methyl-2-oxobutanoate, 4-methyl-2-oxobutanoate and isocaproate (Smit et al., supra). The enzyme has been structurally characterized (Berthold et al., D. Biol. Crystallogr., 63:1217-1224 (2007)). Sequence alignments between the Lactococcus lactis enzyme and the pyruvate decarboxylase of Zymomonas mobilus indicate that the catalytic and substrate recognition residues are nearly identical (Siegert et al., supra), so this enzyme is readily amenable to directed engineering. Additional BCKA genes can be identified by homology to the Lactococcus lactis protein sequence (kdcA, AAS49166.1, 44921617, Lactococcus lactis). Many of the high-scoring BLASTp hits to this enzyme are annotated as indolepyruvate decarboxylases (EC 4.1.1.74). Indolepyruvate decarboxylase (IPDA) is an enzyme that catalyzes the decarboxylation of indolepyruvate to indoleacetaldehyde in plants and plant bacteria.
[0210] 2-amino-4-ketopentanoate is decarboxylated to form 4-aminobutan-2-one by AKP decarboxylase in Step E of FIG. 1. This transformation can be catalyzed by an amino acid decarboxylase. Selection of an appropriate decarboxylase depends on the stereochemical configuration of 4-amino-4-oxopentanoate. When this compound is in a D-configuration, a D-amino acid decarboxylase can be utilized. One such D-amino acid decarboxylase is diaminopimelate decarboxylase (DDC, EC 4.1.1.20). This enzyme decarboxylates the D-stereocenter of meso-diaminopimelate, catalyzing the final step of lysine biosynthesis. DDC has been studied in many organisms including E. coli (Momany et al., D. Biol. Crystallogr., 58:549-552 (2002)), Mycobacterium tuberculosis (Kefala et al., Acta. Crystallogr. Sect. F. Struct. Biol. Cryst. Commun., 61:782-784 (2005); Gokulan et al., J. Biol. Chem., 278:18588-18596 (2003); Andersen et al., Gene, 124:105-109 (1993)), Methylophilus methylotrophus (Tsujimoto et al., J. Biotechnol, 124:327-337 (2006)), and Helicobacter pylori (Hu et al., J. Biol. Chem., 283:21284-21293 (2008)). Alternately, the ornithine decarboxylase (EC 4.1.1.17) from Homo sapiens has a weak activity on the D-isomer of ornithine (Qu et al., Biochem. J., 375:465-470 (2003); Fitzgerald et al., DNA, 8:623-634 (1989)) and can be used for the decarboxylation in step E. Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 23.
TABLE-US-00023 TABLE 23 Protein GenBank ID GI Number Organism lysA NP_417315.1 16130742 Escherichia coli lysA AAA25361.1 149964 Mycobacterium tuberculosis lysA BAC92756.1 37196770 Methylophilus methylotrophus lysA ABW70801.1 158523325 Helicobacter pylori odc1 AA59969.1 386989 Homo sapiens
[0211] When 2-amino-4-ketopentanoate exhibits L-stereochemistry, an amino acid decarboxylase such as aspartate decarboxylase (EC 4.1.1.11), ornithine decarboxylase (EC 4.1.1.17) or lysine decarboxylase (EC 4.1.1.18) can be utilized. An exemplary enzyme is aspartate decarboxylase (EC 4.1.1.11). 2-Amino-4-ketopentanoate bears structural similarity to aspartate, the native substrate of this enzyme. Aspartate decarboxylase participates in pantothenate biosynthesis and is encoded by panD in Escherichia coli (Dusch et al., Appl. Environ. Microbiol., 65:1530-1539 (1999); Ramjee et al., Biochem. J., 323:661-669 (1997); Merkel et al., FEMS Microbiol. Lett., 143:247-252 (1996); Schmitzberger et al., EMBO J., 22:6193-6204 (2003)). The enzymes from Mycobacterium tuberculosis (Chopra et al., Protein Expr. Purif., 25:533-540 (2002)) and Corynebacterium glutamicum (Dusch et al., supra) have been expressed and characterized in E. coli. Lysine decarboxylase enzymes are encoded in the E. coli genome by genes cadA and ldcC. A lysine decarboxylase analogous to CadA was recently identified in Vibrio parahaemolyticus (Tanaka et al., J. Appl. Microbiol. 104:1283-1293 (2008)). The lysine decarboxylase from Selenomonas ruminantium, encoded by ldc, bears sequence similarity to eukaryotic ornithine decarboxylases, and accepts both L-lysine and L-ornithine as substrates (Takatsuka et al., Biosci. Biotechnol Biochem. 63:1843-1846 (1999)). Ornithine decarboxylase enzyme candidates are found in Nicotiana glutinosa (Lee et al., Biochem. J. 360:657-665 (2001)), Lactobacillus sp. 30a (Guirard et al., J Biol. Chem. 255:5960-5964 (1980)) and Vibrio vulnificus (Lee et al., J Biol. Chem. 282:27115-27125 (2007)). The residues involved in substrate specificity Vibrio vulnificus have been elucidated (Lee et al., supra).
[0212] Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 24.
TABLE-US-00024 TABLE 24 Protein GenBank ID GI Number Organism panD P0A790 67470411 Escherichia coli panD Q9X4N0 18203593 Corynebacterium glutanicum panD P65660.1 54041701 Mycobacterium tuberculosis cadA AAA23536. 145458 Escherichia coli ldcC AAC73297.1 1786384 Escherichia coli ldc O50657.1 13124043 Selenomonas ruminantium cadA AB124819.1 44886078 Vibrio parahaemolyticus AF323910.1:1..1299 AAG45222.1 12007488 Nicotiana glutinosa odc1 P43099.2 1169251 Lactobacillus sp. 30a VV2_1235 NP_763142.1 27367615 Vibrio vulnificus
[0213] In reaction J (FIG. 1), acetylacrylate is decarboxylated to 2-oxobutene by acetoacrylate decarboxylase. An enzyme catalyzing this transformation has not been identified to date, but similar reactions are catalyzed by the enzymes aconitate decarboxylase, 4-oxalocrotonate decarboxylase and cinnamate decarboxylase.
[0214] Aconitate decarboxylase catalyzes the final step in itaconate biosynthesis in a strain of Candida and also in the filamentous fungus Aspergillus terreus (Bonnarme et al., J. Bacteriol., 177:3573-3578 (1995); Willke et al., Appl. Microbiol. Biotechnol., 56:289-295 (2001)). A cis-aconitate decarboxylase (CAD) (EC 4.1.16), encoded by ATEG.sub.--09971, has been identified and extensively studied in Aspergillus terreus and other related fungi. Recently, the gene has been cloned and functionally characterized (Kanamasa et al., Appl. Biotechnol., 80:223-229 (2008)) and (WO/2009/014437).
[0215] 4-oxalocronate decarboxylase has been isolated from numerous organisms and characterized. Genes encoding this enzyme include dmpH and dmpE in Pseudomonas sp. (strain 600) (Shingler et al., J. Bacteriol., 174:711-724 (1992)), xylII and xylIII from Pseudomonas putida (Kato et al., Arch. Microbiol., 168:457-463 (1997); Stanley et al., Biochemistry, 39:3514 (2000); Lian et al., J. Am. Chem. Soc., 116:10403-10411 (1994)) and Reut_B5691 and Reut_B5692 from Ralstonia eutropha JMP134 (Hughes et al., J. Bacteriol., 158:79-83 (1984)). The genes encoding the enzyme from Pseudomonas sp. (strain 600) have been cloned and expressed in E. coli (Shingler et al., supra). Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 25.
TABLE-US-00025 TABLE 25 Protein GenBank ID GI Number Organism dmpH CAA43228.1 45685 Pseudomonas sp. CF600 dmpE CAA43225.1 45682 Pseudomonas sp. CF600 xylII YP_709328.1 111116444 Pseudomonas putida xylIII YP_709353.1 111116469 Pseudomonas putida Reut_B5691 YP_299880.1 73539513 Ralstonia eutropha JMP134 Reut_B5692 YP_299881.1 73539514 Ralstonia eutropha JMP134 ATEG_09971 EAU29420.1 114187720 Aspergillus terreus
[0216] An additional class of decarboxylases has been characterized that catalyze the conversion of cinnamate (phenylacrylate) and substituted cinnamate derivatives to the corresponding styrene derivatives. These enzymes are common in a variety of organisms and specific genes encoding these enzymes that have been cloned and expressed in E. coli are: pad 1 from Saccharomyces cerevisae (Clausen et al., Gene, 142:107-112 (1994)), pdc from Lactobacillus plantarum (Barthelmebs et al., Appl. Environ. Microbiol., 67:1063-1069 (2001); Rodriguez et al., J. Agric. Food Chem., 56:3068-3072 (2008); Qi et al., Biochem. J., 375:465-470 (2007)), pofK (pad) from Klebsiella oxytoca (Uchiyama et al., Biosci. Biotechnol. Biochem., 72:116-123 (2008); Hashidoko et al., Biosci. Biotech. Biochem., 58:217-218 (1994)), Pedicoccus pentosaceus (Barthelmebs et al., supra) and padC from Bacillus subtilis and Bacillus pumilus (Cavin et al., Appl. Environ. Microbiol., 64:1466-1471 (1998)). A ferulic acid decarboxylase from Pseudomonas fluorescens also has been purified and characterized (Huang et al., J. Bacteriol., 176:5912-5918 (1994)). Importantly, this class of enzymes has been shown to be stable and does not require either exogenous or internally bound co-factors, thus making these enzymes ideally suitable for biotransformations (Sariaslani, F. S., Annu. Rev. Microbiol., 61:51-69 (2007)). Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 26.
TABLE-US-00026 TABLE 26 Protein GenBank ID GI Number Organism pad1 AAB64980.1 1165293 Saccharomyces cerevisae pdc AAC45282.1 1762616 Lactobacillus plantarum pad BAF65031.1 149941608 Klebsiella oxytoca padC NP_391320.1 16080493 Bacillus subtilis pad YP_804027.1 116492292 Pedicoccus pentosaceus pad CAC18719.1 11691810 Bacillus pumilus
[0217] An additional enzyme for decarboxylation is acetoacetate decarboxylase (EC 4.1.1.4), an enzyme that decarboxylates acetoacetate to acetone and has therefore been studied for its role in bacterial solventogenesis. Exemplary bacterial enzymes have been characterized from Clostridium acetobutylicum (Benner et al., J. Am. Chem. So. 103:993-994 (1981); Hlghbarger et al., Biochemistry 35:41-46 (1996); Petersen et al., Appl. Environ. Microbiol. 56:3491-3498 (1990); Rozzel et al. J. Am. Chem. Soc. 106:4937-4941 (1984)) Clostridium saccharoperbutylacetonicum (Kosaka, et al., Biosci. Biotechnol Biochem. 71:58-68 (2007)) and Clostridium beijerinckii (Ravagnani et al. Mol. Microbiol. 37:1172-1185 (2000)). Acetoacetate decarboxylase activity has also been demonstrated in Pseudomonas putida and Bacillus polymyxa but genes are not associated with this activity to date (Matiasek et al., Curr. Microbiol. 42: 276-281 (2001)). Bacterial genes in other organisms such as Clostridium botulinum and Bacillus amyloliquefaciens can be identified by sequence homology. In humans and other mammals, acetoacetate decarboxylase catalyzes the final step of the ketone-body pathway (Kalapos, Biochim. Biophys. Acta 1621:122-139 (2003)), but genes associated with this activity have not been identified to date. Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 27.
TABLE-US-00027 TABLE 27 Protein GenBank ID GI Number Organism adc NP_149328.1 15004868 Clostridium acetobutylicum adc AAP42566.1 31075386 Clostridium saccharoperbutyl- acetonicum cbei_3835 YP_001310906.1 150018652 Clostridium beijerinckii CLL_A2135 YP_001886324.1 187933144 Clostridium botulinum RBAM_030030 YP_001422565.1 154687404 Bacillus amyloliquefaciens
[0218] All the aforementioned gene candidates can also be used to catalyze the decarboxylation of 2-oxo-4-hydroxypentanoate to 3-hydroxybutyraldehyde in Step N of FIG. 1.
[0219] Butenone hydratase (Step G, FIG. 1), 4-hydroxybutyryl-CoA dehydratase (Step A, FIG. 3) and crotonase (Step A, FIG. 3) are hydrolyase-type transformations. Specifically, the hydration of butenone to 4-hydroxy-2-butanone (Step G, FIG. 1) can be accomplished by an enzyme in the hydratase family of enzymes. Enzymes that can carry out this transformation include fumarate hydratase (EC 4.2.1.2), 2-(hydroxymethyl)glutarate dehydratase (EC 4.2.1.-), dimethylmaleate hydratase (EC 4.2.1.85) and citramalate hydrolyase (EC 4.2.1.34).
[0220] Fumarate hydratase enzymes naturally catalyze the reversible hydration of fumarate to malate. Although the ability of fumarate hydratase to react with butanone as a substrate has not been described in the literature, a wealth of structural information is available for this enzyme and other researchers have successfully engineered the enzyme to alter activity, inhibition and localization (Weaver, T., B. Biol. Crystallogr., 61:1395-1401 (2005)). E. coli has three fumarases: FumA, FumB, and FumC that are regulated by growth conditions. FumB is oxygen sensitive and only active under anaerobic conditions. FumA is active under microanaerobic conditions, and FumC is the only active enzyme in aerobic growth (Tseng et al., J. Bacteriol., 183:461-467 (2001); Woods et al., Biochem. Biophys. Acta., 954:14-26 (1988); Guest et al., J. Gen. Microbiol., 131:2971-2984 (1985)). Additional enzymes are found in Campylobacter jejuni (Smith et al., Int. J. Biochem. Cell Biol., 31:961-975 (1999)), Thermus thermophilus (Mizobata et al., Arch. Biochem. Biophys., 355:49-55 (1998)) and Rattus norvegicus (Kobayashi et al., J. Biochem., 89:1923-1931 (1981)). Similar enzymes with high sequence homology include fum1 from Arabidopsis thaliana and fumC from Corynebacterium glutamicum. The MmcBC fumarase from Pelotomaculum thermopropionicum is another class of fumarase with two subunits (Shimoyama et al., FEMS Microbiol. Lett., 270:207-213 (2007)). Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 28.
TABLE-US-00028 TABLE 28 Protein GenBank ID GI Number Organism fumA NP_416129.1 16129570 Escherichia coli fumB NP_418546.1 16131948 Escherichia coli fumC NP_416128.1 16129569 Escherichia coli fumC O69294 9789756 Campylobacter jejuni fumC P84127 75427690 Thermus thermophilus fumH P14408 120605 Rattus norvegicus fum1 P93033 39931311 Arabidopsis thaliana fumC Q8NRN8 39931596 Corynebacterium glutamicum MmcB YP_001211906 147677691 Pelotomaculum thermopropionicum MmcC YP_001211907 147677692 Pelotomaculum thermopropionicum
[0221] Two additional hydratase enzymes are 2-(hydroxymethyl)glutarate dehydratase and dimethylmaleate hydratase, enzymes studied for their role in nicontinate catabolism in Eubacterium barkeri (formerly Clostridium barkeri) (Alhapel et al., Proc. Natl. Acad. Sci. USA, 103:12341-12346 (2006)). 2-(Hydroxymethyl)glutarate dehydratase is a [4Fe-4S]-containing enzyme that dehydrates 2-(hydroxymethyl)glutarate to 2-methylene-glutarate. This enzyme is encoded by hmd in Eubacterium barkeri (Alhapel et al., supra). Similar enzymes with high sequence homology are found in Bacteroides capillosus, Anaerotruncus colihominis, and Natranaerobius thermophilius. These enzymes are homologous to the alpha and beta subunits of [4Fe-4S]-containing bacterial serine dehydratases (e.g., E. coli enzymes encoded by tdcG, sdhB, and sdaA). Dimethylmaleate hydratase (EC 4.2.1.85) is a reversible Fe.sup.2+-dependent and oxygen-sensitive enzyme in the aconitase family that hydrates dimethylmaeate to form (2R,3S)-2,3-dimethylmalate. This enzyme is encoded by dmdAB in Eubacterium barkeri (Alhapel, et al., supra; Kollmann-Koch et al., Physiol. Chem., 365:847-857 (1984)). Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 29.
TABLE-US-00029 TABLE 29 Protein GenBank ID GI Number Organism hmd ABC88407.1 86278275 Eubacterium barkeri BACCAP_02294 ZP_02036683.1 154498305 Bacteroides capillosus ATCC 29799 ANACOL_02527 ZP_02443222.1 167771169 Anaerotruncus colihominis DSM 17241 NtherDRAFT_2368 ZP_02852366.1 169192667 Natranaerobius thermophilus JW/NM-WN-LF dmdA ABC88408 86278276 Eubacterium barkeri dmdB ABC88409.1 86278277 Eubacterium barkeri
[0222] An additional enzyme is 2-methylmalate dehydratase, also called citramalate hydrolyase, a reversible hydrolyase that catalyzes the alpha, beta elimination of water from citramalate to form mesaconate. This enzyme has been purified and characterized in Clostridium tetanomorphum (Wang et al., J. Biol. Chem., 244:2516-2526 (1969)). The activity of this enzyme has also been detected in several bacteria in the genera Citrobacter and Morganella in the context of the glutamate degradation VI pathway (Kato et al., supra). Genes encoding this enzyme have not been identified in any organism to date.
[0223] Hydration of crotonyl-CoA to form 3-hydroxybutyryl-CoA (Step B, FIG. 3) is catalyzed by a crotonase (EC 4.2.1.55). These enzymes are required for n-butanol formation in some organisms, particularly Clostridial species, and also comprise one step of the 3-hydroxypropionate/4-hydroxybutyrate cycle in thermoacidophilic Archaea of the genera Sulfolobus, Acidianus, and Metallosphaera. Exemplary genes encoding crotonase enzymes can be found in C. acetobutylicum (Boynton et al., J. Bacteriol., 178:3015-3024 (1996)), C. kluyveri (Hillmer et al., FEBS Lett., 21:351-354 (1972)), and Metallosphaera sedula (Berg et al., supra). Enoyl-CoA hydratases, which are involved in fatty acid beta-oxidation and/or the metabolism of various amino acids, can also catalyze the hydration of crotonyl-CoA to form 3-hydroxybutyryl-CoA (Roberts et al., Arch. Microbiol., 117:99-108 (1978); Agnihotri et al., Bioorg. Med. Chem., 11:9-20 (2003); Conrad et al., J. Bacteriol., 118:103-111 (1974)). An exemplary enoyl-CoA hydratase is the gene product of ech from Pseudomonas putida (Roberts et al., supra). The enoyl-CoA hydratases, phaA and phaB, of P. putida have been indicated to carry out the hydroxylation of double bonds during phenylacetate catabolism (Olivera et al., Proc. Natl. Acad. Sci USA, 95:6419-6424 (1998)). The paaA and paaB from P. fluorescens catalyze analogous transformations (Olivera et al., supra). Lastly, a number of Escherichia coli genes have been shown to demonstrate enoyl-CoA hydratase functionality including maoC (Park et al., J. Bacteriol., 185:5391-5397 (2003)), paaF (Ismail et al., Eur. J. Biochem., 270:3047-3054 (2003); Park et al., Appl. Biochem. Biotechnol., 113-116:335-346 (2004); Park et al., Biotechnol Bioeng., 86:681-686 (2004)) and paaG (Ismail et al., supra; Park et al., supra; Park et al., supra). Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 30.
TABLE-US-00030 TABLE 30 Protein GenBank ID GI Number Organism crt NP_349318.1 15895969 Clostridium acetobutylicum crt1 YP_001393856 153953091 Clostridium kluyveri DSM 555 ech NP_745498.1 26990073 Pseudomonas putida phaA ABF82233.1 26990002 Pseudomonas putida phaB ABF82234.1 26990001 Pseudomonas putida paaA NP_745427.1 106636093 Pseudomonas fluorescens paaB NP_745426.1 106636094 Pseudomonas fluorescens maoC NP_415905.1 16129348 Escherichia coli paaF NP_415911.1 16129354 Escherichia coli paaG NP_415912.1 16129355 Escherichia coli
[0224] Alternatively, the E. coli gene products of fadA and fadB encode a multienzyme complex involved in fatty acid oxidation that exhibits enoyl-CoA hydratase activity (Haller et al., Biochemistry 39:4622-4629 (2000); Martinez-Carrion et al., J. Biol. Chem. 240:3538-3546 (1965); Matthies et al., Appl. Environ. Micriobiol. 58:1435-1439 (1992)). Knocking out a negative regulator encoded by fadR can be utilized to activate the fadB gene product (Jeng et al., A. Biochemistry 13:2898-2903 (1974)). The fadI and fadJ genes encode similar functions and are naturally expressed under anaerobic conditions (Atsumi et al., Nature 451:86-89 (2008)). Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 31.
TABLE-US-00031 TABLE 31 Protein GenBank ID GI Number Organism fadA YP_026272.1 49176430 Escherichia coli fadB NP_418288.1 16131692 Escherichia coli fadI NP_416844.1 16130275 Escherichia coli fadJ NP_416843.1 16130274 Escherichia coli fadR NP_415705.1 16129150 Escherichia coli
[0225] The reversible condensation of 4-hydroxybutyryl-CoA to crotonyl-CoA (Step A, FIG. 3) is catalyzed by the bifunctional enzyme 4-hydroxybutyryl-CoA dehydratase/vinylacetyl-CoA .DELTA.-isomerase. This enzyme first dehydrates 4-hydroxybutyryl-CoA to vinylacetyl-CoA, which subsequently rearranges to form crotonoyl-CoA. The enzymes from Clostridium kluyveri and C. aminobutyrium have been purified, characterized, and sequenced at the N-terminal domain (Scherf et al., Eur. J. Biochem., 215:421-429 (1993); Scherf et al., Arch. Microbiol., 161:239-245 (1994)). The abfD genes from C. aminobutyrium and C. kluyveri match exactly with these N-terminal amino acid sequences, and have been indicated to encode the 4-hydroxybutyrul-CoA dehydratases/vinylacetyl-CoA A-isomerase activities. Similar genes are identified through homology from genome projects, including abfD from Porphyromonas gingivalis and Msed.sub.--1220 from Metallosphaera sedula. Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 32.
TABLE-US-00032 TABLE 32 Protein GenBank ID GI Number Organism abfD YP_001396399.1 153955634 Clostridium kluyveri abfD P55792 84028213 Clostridium aminobutyricum abfD YP_001928843 188994591 Porphyromonas gingivalis Msed_1220 YP_001191305.1 146303989 Metallosphaera sedula
[0226] Deamination of 2-amino-4-ketopentanoate (FIG. 1, Reaction I) and of 4-aminobutan-2-one (Step F, FIG. 1) can be accomplished by AKP ammonia-lyase and 4-aminobutan-2-one ammonia-lyase respectively. These deaminations are very similar to the deamination of aspartate to fumarate by aspartase. The enzyme has been extensively studied and several crystal structures are available. The E. coli enzyme has been shown to react with alternate substrates such as aspartatephenylmethylester, asparagine, benzyl-aspartate and malate (Ma et al., Ann. N.Y. Acad. Sci., 672:60-65 (1992). In a separate study, directed evolution has been implemented on this enzyme to alter substrate specificity (Asano et al., Biomol. Eng., 22:95-101 (2005)). Enzymes with aspartase functionality have also been characterized in Haemophilus influenzae (Sjostrom et al., Biochem. Biophys. Acta., 1324:182-190 (1997)), Pseudomonas fluorescens (Takagi et al., J. Biochem., 96:545-552 (1984)), Bacillus subtilus (Sjostrom et al., supra) and Serratia marcescens (Takagi et al., J. Bacteriol., 161:1-6 (1985)). Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 33.
TABLE-US-00033 TABLE 33 Protein GenBank ID GI Number Organism aspA NP_418562 90111690 Escherichia coli aspA P44324.1 1168534 Haemophilus influenzae aspA P07346.1 114273 Pseudomonas fluorescens ansB P26899.1 251757243 Bacillus subtilus aspA P33109.1 416661 Serratia marcescens
[0227] A similar ammonia lyase reaction is catalyzed by methylaspartase (EC 4.3.1.2), an enzyme participating in the glutamate fermentation route via mesaconate (Kato et al., supra). This enzyme, also known as beta-methylaspartase and 3-methylaspartate ammonia-lyase, naturally catalyzes the deamination of threo-3-methylasparatate to mesaconate. The 3-methylaspartase from Clostridium tetanomorphum has been cloned, functionally expressed in E. coli, and crystallized (Asuncion et al., 57:731-733 (2001); Asuncion et al., J Biol Chem. 277:8306-8311 (2002); Botting et al., 27:2953-2955 (1988); Goda et al., 31:10747-10756 (1992)). In Citrobacter amalonaticus, this enzyme is encoded by BAA28709 (Kato et al., Arch. Microbiol 168:457-463 (1997)). 3-Methylaspartase has also been crystallized from E. coli YG1002 (Asano et al., FEMS Microbiol Lett. 118:255-258 (1994)) although the protein sequence is not listed in public databases such as GenBank. Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 34.
TABLE-US-00034 TABLE 34 Protein GenBank ID GI Number Organism mal AAB24070.1 259429 Clostridium tetanomorphum BAA28709 BAA28709.1 3184397 Citrobacter amalonaticus
[0228] Referring now to FIG. 8B, gene candidates for alanine dehydrogenase alanine aminotransferase are shown below which convert pyruvate to alanine.
TABLE-US-00035 TABLE 35 glutamate-pyruvate Escherichia coli Accession: AAC76384.1 aminotransferase (alaB) K-12 substr. GI: 1789759 MG1655 glutamate-pyruvate Escherichia coli Accession: AAC75350.1 aminotransferase (alaA) K-12 substr. GI: 1788627 MG1655 glutamate-pyruvate Escherichia coli Accession: AAC75438.1 aminotransferase (alaC) K-12 substr. GI: 1788722 MG1655 alanine transaminase Arabidopsis Accession: AEE30370.1 (AOAT1) thaliana col GI: 332192249 alanine transaminase Arabidopsis Accession: AEE35084.1 (AOAT2) thaliana col GI: 332196963 tryptophan Arabidopsis Accession: aminotransferase (TAA1) thaliana col NP_177213.1 GI: 15223183 alanine aminotransferase Homo sapiens Accession: (GPT2) NP_597700.1 GI: 19263340 alanine aminotransferase Homo sapiens Accession: AAB20194.1 (GPT) GI: 238134 alanine aminotransferase Clostridium propionicum alanine dehydrogenase Phormidium Accession: BAA24455.1 (ald) lapideum GI: 2804515 alanine dehydrogenase Enterobacter Accession: BAA77513.1 (aladh) aerogenes GI: 4803749 L-alanine dehydrogenase Bacillus cereus L-alanine dehydrogenase Bacillus subtilis Accession: (ald) NP_391071.1 GI: 16080244 alanine dehydrogenase Bilophila Accession: AF269148.1 wadsworthia GI: 13661832 RZATAU
[0229] Five requisite pathways to achieve the biosynthesis of 4-HB are exemplified herein and shown for purposes of illustration in FIG. 9B. One requisite 4-HB biosynthetic pathway includes the biosynthesis of 4-HB from succinate (the succinate pathway). The enzymes participating in this 4-HB pathway include succinate reductase, 4-hydroxybutanoate dehydrogenase (Steps F and C, FIG. 9B). Another requisite 4-HB biosynthetic pathway includes the biosynthesis from succinate through succinyl-CoA (the succinyl-CoA pathway). The enzymes participating in this 4-HB pathway include succinyl-CoA synthetase, CoA-dependent succinic semialdehyde dehydrogenase, and 4-hydroxybutanoate dehydrogenase (Steps A, I and K, FIG. 9B). Three other requisite 4-HB biosynthetic pathways include the biosynthesis of 4-HB from .alpha.-ketoglutarate (the .alpha.-ketoglutarate pathways). Hence, a third requisite 4-HB biosynthetic pathway is the biosynthesis of succinic semialdehyde through succinyl-CoA (Steps V, B, and C, FIG. 9B). AKG can be converted into succinyl-CoA by alpha-ketoglutarate dehydrogenase. This is then transformed into succinate semialdehyde and 4-HB as described earlier. Yet another pathway (Steps Q, R, S, T, U, FIG. 9B) for synthesizing 4-HB entails conversion of AKG into glutamate via glutamate dehydrogenase or glutamate transaminase. Glutamate is decarboxylated to form 4-aminobutyrate by glutamate decarboxylase and then 4-aminobutyrate dehydrogenase or 4-aminobutyrate transaminase can convert it into succinate semialdehyde. The final 4-HB biosynthetic pathway described here also includes the biosynthesis of 4-HB from .alpha.-ketoglutarate (Steps I and C, FIG. 9B), but utilizes .alpha.-ketoglutarate decarboxylase to catalyze succinic semialdehyde synthesis. 4-hydroxybutanoate dehydrogenase catalyzes the conversion of succinic semialdehyde to 4-HB. 4-hydroxybutanoate dehydrogenase catalyzes the conversion of succinic semialdehyde to 4-HB. 4HB can further be converted into 4-HB-CoA by 4-HB-CoA ligase, 4-HB-CoA synthetase or 4-HB-CoA transferase. Alternatively, the conversion of 4-HB to 4-HB CoA can be carried out by 4-Hydroxybutyrate kinase and Phosphotrans-4-hydroxybutyrylase (Steps D and E, FIG. 9B). Each of these 4-HB and 4HB-CoA biosynthetic pathways, their substrates, reactants and products are described further below in the Examples.
[0230] The non-naturally occurring microbial organisms of the invention can be produced by introducing expressible nucleic acids encoding one or more of the enzymes participating in one or more 4-HB biosynthetic pathways. Depending on the host microbial organism chosen for biosynthesis, nucleic acids for some or all of a particular 4-HB biosynthetic pathway can be expressed. For example, if a chosen host is deficient in both enzymes in the succinate to 4-HB pathway and this pathway is selected for 4-HB biosynthesis, then expressible nucleic acids for both succinate reductase and 4-hydroxybutanoate dehydrogenase are introduced into the host for subsequent exogenous expression. Alternatively, if the chosen host exhibits endogenous succinate reductase, but is deficient in 4-hydroxybutanoate dehydrogenase then an encoding nucleic acid is needed for this enzyme to achieve 4-HB biosynthesis.
[0231] In like fashion, where 4-HB biosynthesis is selected to occur through the succinate to succinyl-CoA pathway (the succinyl-CoA pathway), encoding nucleic acids for host deficiencies in the enzymes succinyl-CoA synthetase, CoA-dependent succinic semialdehyde dehydrogenase and/or 4-hydroxybutanoate dehydrogenase are to be exogenously expressed in the recipient host. Selection of 4-HB biosynthesis through the .alpha.-ketoglutarate to succinic semialdehyde pathway (the .alpha.-ketoglutarate pathway) can utilize exogenous expression for host deficiencies in one or more of the enzymes for glutamate dehydrogenase, glutamate transaminase, glutamate decarboxylase, 4-aminobutyrate dehydrogenase, 4-aminobutyrate transaminase, and/or 4-hydroxybutanoate dehydrogenase.
[0232] Depending on the 4-HB biosynthetic pathway constituents of a selected host microbial organism, the non-naturally occurring microbial 4-HB biocatalysts of the invention will include at least one exogenously expressed 4-HB pathway-encoding nucleic acid and up to all encoding nucleic acids for one or more 4-HB biosynthetic pathways. For example, 4-HB-CoA biosynthesis can be established from all five pathways in a host deficient in 4-hydroxybutanoate dehydrogenase through exogenous expression of a 4-hydroxybutanoate dehydrogenase encoding nucleic acid. In contrast, 4-HB biosynthesis can be established from all five pathways in a host deficient in all eleven enzymes through exogenous expression of all eight of CoA-independent succinic semialdehyde dehydrogenase, succinyl-CoA synthetase, CoA-dependent succinic semialdehyde dehydrogenase, glutamate: succinic semialdehyde transaminase, glutamate decarboxylase, .alpha.-ketoglutarate decarboxylase and 4-hydroxybutanoate dehydrogenase.
[0233] Given the teachings and guidance provided herein, those skilled in the art will understand that the number of encoding nucleic acids to introduce in an expressible form will, at least, parallel the 4-HB pathway deficiencies of the selected host microbial organism. Therefore, a non-naturally occurring microbial organism of the invention can have one, two, three, four, five, six or seven or up to eleven nucleic acids encoding the above enzymes constituting one or more 4-HB biosynthetic pathways. In some embodiments, the non-naturally occurring microbial organisms also can include other genetic modifications that facilitate or optimize 4-HB-CoA biosynthesis or that confer other useful functions onto the host microbial organism. One such other functionality can include, for example, augmentation of the synthesis of one or more of the 4-HB-CoA pathway precursors such as succinate, succinyl-CoA and/or .alpha.-ketoglutarate.
[0234] In some embodiments, a non-naturally occurring microbial organism of the invention is generated from a host that contains the enzymatic capability to synthesize 4-HB. In this specific embodiment it can be useful to increase the synthesis or accumulation of a 4-HB pathway product to, for example, drive 4-HB pathway reactions toward 4-HB production. Increased synthesis or accumulation can be accomplished by, for example, overexpression of nucleic acids encoding one or more of the above-described 4-HB pathway enzymes. Over expression of the 4-HB pathway enzyme or enzymes can occur, for example, through exogenous expression of the endogenous gene or genes, or through exogenous expression of the heterologous gene or genes. Therefore, naturally occurring organisms can be readily generated to be non-naturally 4-HB producing microbial organisms of the invention through overexpression of one, two, three, four, five, six or seven nucleic or all eleven acids encoding 4-HB biosynthetic pathway enzymes. In addition, a non-naturally occurring organism can be generated by mutagenesis of an endogenous gene that results in an increase in activity of an enzyme in the 4-HB biosynthetic pathway.
[0235] Non-naturally occurring microbial organisms also can be generated which biosynthesize 1,3-BDO. As with the 4-HB producing microbial organisms of the invention, the 1,3-BDO producing microbial organisms also can produce intracellularly or secret the 1,3-BDO into the culture medium. Following the teachings and guidance provided previously for the construction of microbial organisms that synthesize 4-HB, additional 1,3-BDO pathways can be incorporated into the 4-HB producing microbial organisms to generate organisms that also synthesize 1,3-BDO and other 1,3-BDO family compounds. The non-naturally occurring microbial organisms of the invention capable of 1,3-BDO biosynthesis circumvent these chemical synthesis using 4-HB as an entry point as illustrated in FIG. 9B.
[0236] The additional 1,3-BDO pathways to introduce into 4-HB producers include, for example, the exogenous expression in a host deficient background or the overexpression of one or more of the enzymes exemplified in FIG. 9B. An initial step in the entry pathway to 1,3-BDO is the conversion of 4-hydroxybutyrate to 4-hydroxybutyryl-CoA using 4-hydroxybutyrate:CoA transferase or 4-hydroxybutyryl-CoA synthetase (or ligase) the combination of butyrate kinase and phosphotransbutyrylase. Accordingly, the additional initial 1,3-BDO pathways to introduce into 4-HB producers to produce 4-hydroxybutyryl-CoA include, for example, the exogenous expression in a host deficient background or the overexpression of one or more of a 4-hydroxybutyrate:CoA transferase, 4-hydroxybutyryl-CoA synthetase (or ligase), butyrate kinase or phosphotransbutyrylase. In the absence of endogenous acyl-CoA synthetase capable of modifying 4-HB, the non-naturally occurring 1,3-BDO producing microbial organisms can further include an exogenous acyl-CoA synthetase selective for 4-HB, or the combination of multiple enzymes that have as a net reaction conversion of 4-HB into 4-HB-CoA. As exemplified further below in the Examples, butyrate kinase and phosphotransbutyrylase exhibit 1,3-BDO pathway activity and catalyze the conversions illustrated in FIG. 9B with a 4-HB substrate. Therefore, these enzymes also can be referred to herein as 4-hydroxybutyrate kinase and phosphotranshydroxybutyrylase respectively. Once 4-hydroxybutyryl-CoA is generated it can then be utilized for the biosynthesis of 1,3-BDO following the subsequent steps shown FIG. 9B.
[0237] Step A of FIG. 9B involves CoA synthetase or ligase reactions for succinate as the substrate. Exemplary genes encoding enzymes likely to carry out these transformations include the sucCD genes of E. coli which naturally form a succinyl-CoA synthetase complex. This enzyme complex naturally catalyzes the formation of succinyl-CoA from succinate with the contaminant consumption of one ATP, a reaction which is reversible in vivo (Buck et al., Biochem. 24:6245-6252 (1985)).
TABLE-US-00036 TABLE 36 Gene Accession No. GI No. Organism sucC NP_415256.1 16128703 Escherichia coli sucD AAC73823.1 1786949 Escherichia coli
[0238] Additional exemplary CoA-ligases include the rat dicarboxylate-CoA ligase for which the sequence is yet uncharacterized (Vamecq et al., Biochemical Journal 230:683-693 (1985)), either of the two characterized phenylacetate-CoA ligases from P. chrysogenum (Lamas-Maceiras et al., Biochem. J. 395:147-155 (2005); Wang et al., Biochem Biophy Res Commun 360(2):453-458 (2007)), the phenylacetate-CoA ligase from Pseudomonas putida (Martinez-Blanco et al., J. Biol. Chem. 265:7084-7090 (1990)), and the 6-carboxyhexanoate-CoA ligase from Bacillus subtilis (Bower et al., J. Bacteriol. 178(14):4122-4130 (1996)). Additional candidate enzymes are acetoacetyl-CoA synthetases from Mus musculus (Hasegawa et al., Biochim Biophys Acta 1779:414-419 (2008)) and Homo sapiens (Ohgami et al., Biochem Pharmacol 65:989-994 (2003)) which naturally catalyze the ATP-dependant conversion of acetoacetate into acetoacetyl-CoA. 4-hydroxybutyryl-CoA synthetase activity has been demonstrated in Metallosphaera sedula (Berg et al., Science 318:1782-1786 (2007)). This function has been tentatively assigned to the Msed.sub.--1422 gene.
TABLE-US-00037 TABLE 37 Gene Accession No. GI No. Organism phl CAJ15517.1 77019264 Penicillium chrysogenum phlB ABS19624.1 152002983 Penicillium chrysogenum paaF AAC24333.2 22711873 Pseudomonas putida bioW NP_390902.2 50812281 Bacillus subtilis AACS NP_084486.1 21313520 Mus musculus AACS NP_076417.2 31982927 Homo sapiens Msed_1422 YP_001191504 146304188 Metallosphaera sedula
[0239] ADP-forming acetyl-CoA synthetase (ACD, EC 6.2.1.13) is another candidate enzyme that couples the conversion of acyl-CoA esters to their corresponding acids with the concurrent synthesis of ATP. Several enzymes with broad substrate specificities have been described in the literature. ACD I from Archaeoglobus fulgidus, encoded by AF1211, was shown to operate on a variety of linear and branched-chain substrates including acetyl-CoA, propionyl-CoA, butyryl-CoA, acetate, propionate, butyrate, isobutyrate, isovalerate, succinate, fumarate, phenylacetate, indoleacetate (Musfeldt et al., J Bacteriol 184:636-644 (2002)). The enzyme from Haloarcula marismortui (annotated as a succinyl-CoA synthetase) accepts propionate, butyrate, and branched-chain acids (isovalerate and isobutyrate) as substrates, and was shown to operate in the forward and reverse directions (Brasen et al., Arch Microbiol 182:277-287 (2004)). The ACD encoded by PAE3250 from hyperthermophilic crenarchaeon Pyrobaculum aerophilum showed the broadest substrate range of all characterized ACDs, reacting with acetyl-CoA, isobutyryl-CoA (preferred substrate) and phenylacetyl-CoA (Brasen et al., supra). The enzymes from A. fulgidus, H. marismortui and P. aerophilum have all been cloned, functionally expressed, and characterized in E. coli (Musfeldt et al., supra; Brasen et al., supra).
TABLE-US-00038 TABLE 38 Gene Accession No. GI No. Organism AF1211 NP_070039.1 11498810 Archaeoglobus fulgidus DSM 4304 scs YP_135572.1 55377722 Haloarcula marismortui ATCC 43049 PAE3250 NP_560604.1 18313937 Pyrobaculum aerophilum str. IM2
[0240] The conversion of succinate to succinate semialdehyde (Step F) can be catalyzed by a carboxylic acid reductase. One notable carboxylic acid reductase can be found in Nocardia iowensis which catalyzes the magnesium, ATP and NADPH-dependent reduction of carboxylic acids to their corresponding aldehydes (Venkitasubramanian et al., J Biol. Chem. 282:478-485 (2007)). This enzyme is encoded by the car gene and was cloned and functionally expressed in E. coli (Venkitasubramanian et al., J Biol. Chem. 282:478-485 (2007)). Expression of the npt gene product improved activity of the enzyme via post-transcriptional modification. The npt gene encodes a specific phosphopantetheine transferase (PPTase) that converts the inactive apo-enzyme to the active holo-enzyme. The natural substrate of this enzyme is vanillic acid and the enzyme exhibits broad acceptance of aromatic and aliphatic substrates (Venkitasubramanian et al. "Biocatalytic Reduction of Carboxylic Acids: Mechanism and Applications" Chapter 15 in Biocatalysis in the Pharmaceutical and Biotechnology Industires, ed. R. N. Patel, CRC Press LLC, Boca Raton, Fla. (2006)). Additional car and npt genes can be identified based on sequence homology.
TABLE-US-00039 TABLE 39A Gene Accession No. GI No. Organism car AAR91681.1 40796035 Nocardia iowensis (sp. NRRL 5646) npt ABI83656.1 114848891 Nocardia iowensis (sp. NRRL 5646)
TABLE-US-00040 TABLE 39B Gene Accession No. GI No. Organism fadD9 YP_978699.1 121638475 Mycobacterium bovis BCG BCG_2812c YP_978898.1 121638674 Mycobacterium bovis BCG nfa20150 YP_118225.1 54023983 Nocardia farcinica IFM 10152 nfa40540 YP_120266.1 54026024 Nocardia farcinica IFM 10152 SGR_6790 YP_001828302.1 182440583 Streptomyces griseus subsp. griseus NBRC 13350 SGR_665 YP_001822177.1 182434458 Streptomyces griseus subsp. griseus NBRC 13350
[0241] An additional enzyme candidate found in Streptomyces griseus is encoded by the griC and griD genes. This enzyme is believed to convert 3-amino-4-hydroxybenzoic acid to 3-amino-4-hydroxybenzaldehyde as deletion of either griC or griD led to accumulation of extracellular 3-acetylamino-4-hydroxybenzoic acid, a shunt product of 3-amino-4-hydroxybenzoic acid metabolism (Suzuki, et al., J. Antibiot. 60(6):380-387 (2007)). Co-expression of griC and griD with SGR.sub.--665, an enzyme similar in sequence to the Nocardia iowensis npt, may be beneficial.
TABLE-US-00041 TABLE 40 Gene Accession No. GI No. Organism griC 182438036 YP_001825755.1 Streptomyces griseus subsp. griseus NBRC 13350 griD 182438037 YP_001825756.1 Streptomyces griseus subsp. griseus NBRC 13350
[0242] An enzyme with similar characteristics, alpha-aminoadipate reductase (AAR, EC 1.2.1.31), participates in lysine biosynthesis pathways in some fungal species. This enzyme naturally reduces alpha-aminoadipate to alpha-aminoadipate semialdehyde. The carboxyl group is first activated through the ATP-dependent formation of an adenylate that is then reduced by NAD(P)H to yield the aldehyde and AMP. Like CAR, this enzyme utilizes magnesium and requires activation by a PPTase. Enzyme candidates for AAR and its corresponding PPTase are found in Saccharomyces cerevisiae (Morris et al., Gene 98:141-145 (1991)), Candida albicans (Guo et al., Mol. Genet. Genomics 269:271-279 (2003)), and Schizosaccharomyces pombe (Ford et al., Curr. Genet. 28:131-137 (1995)). The AAR from S. pombe exhibited significant activity when expressed in E. coli (Guo et al., Yeast 21:1279-1288 (2004)). The AAR from Penicillium chrysogenum accepts S-carboxymethyl-L-cysteine as an alternate substrate, but did not react with adipate, L-glutamate or diaminopimelate (Hijarrubia et al., J Biol. Chem 278:8250-8256 (2003)). The gene encoding the P. chrysogenum PPTase has not been identified to date and no high-confidence hits were identified by sequence comparison homology searching.
TABLE-US-00042 TABLE 41 Gene Accession No. GI No. Organism LYS2 AAA34747.1 171867 Saccharomyces cerevisiae LYS5 P50113.1 1708896 Saccharomyces cerevisiae LYS2 AAC02241.1 2853226 Candida albicans LYS5 AAO26020.1 28136195 Candida albicans Lys1p P40976.3 13124791 Schizosaccharomyces pombe Lys7p Q10474.1 1723561 Schizosaccharomyces pombe Lys2 CAA74300.1 3282044 Penicillium chrysogenum
[0243] The conversion of succinyl-CoA to succinate semialdehyde (Step B, FIG. 9B) is catalyzed by an aldehyde forming succinyl-CoA reductase. Several acyl-CoA dehydrogenases are capable of reducing an acyl-CoA to its corresponding aldehyde. Exemplary genes that encode such enzymes include the Acinetobacter calcoaceticus acr1 encoding a fatty acyl-CoA reductase (Reiser and Somerville, J. Bacteriology 179:2969-2975 (1997)), the Acinetobacter sp. M-1 fatty acyl-CoA reductase (Ishige et al. Appl. Environ. Microbiol. 68:1192-1195 (2002)), and a CoA- and NADP-dependent succinate semialdehyde dehydrogenase encoded by the sucD gene in Clostridium kluyveri (Sohling and Gottschalk J Bacteriol 178:871-80 (1996); Sohling and Gottschalk J Bacteriol. 178:871-880 (1996)). SucD of P. gingivalis is another aldehyde-forming succinyl-CoA reductase (Takahashi et al. J. Bacteriol. 182:4704-4710 (2000)). The enzyme acylating acetaldehyde dehydrogenase in Pseudomonas sp, encoded by bphG, is yet another as it has been demonstrated to oxidize and acylate acetaldehyde, propionaldehyde, butyraldehyde, isobutyraldehyde and formaldehyde (Powlowski et al. J Bacteriol. 175:377-385 (1993)). In addition to reducing acetyl-CoA to ethanol, the enzyme encoded by adhE in Leuconostoc mesenteroides has been shown to oxidize the branched chain compound isobutyraldehyde to isobutyryl-CoA (Koo et al., Biotechnol Lett. 27:505-510 (2005)). Butyraldehyde dehydrogenase catalyzes a similar reaction, conversion of butyryl-CoA to butyraldehyde, in solventogenic organisms such as Clostridium saccharoperbutylacetonicum (Kosaka et al., Biosci. Biotechnol Biochem. 71:58-68 (2007)).
TABLE-US-00043 TABLE 42 Gene Accession No. GI No. Organism acr1 YP_047869.1 50086359 Acinetobacter calcoaceticus acr1 AAC45217 1684886 Acinetobacter baylyi acr1 BAB85476.1 18857901 Acinetobacter sp. Strain M-1 sucD P38947.1 730847 Clostridium kluyveri sucD NP_904963.1 34540484 Porphyromonas gingivalis bphG BAA03892.1 425213 Pseudomonas sp adhE AAV66076.1 55818563 Leuconostoc mesenteroides bld AAP42563.1 31075383 Clostridium saccharoperbutylacetonicum
[0244] An additional enzyme type that converts an acyl-CoA to its corresponding aldehyde is malonyl-CoA reductase which transforms malonyl-CoA to malonic semialdehyde. Malonyl-CoA reductase is a key enzyme in autotrophic carbon fixation via the 3-hydroxypropionate cycle in thermoacidophilic archael bacteria (Berg et al. Science 318:1782-1786 (2007); Thauer, R. K. Science 318:1732-1733 (2007)). The enzyme utilizes NADPH as a cofactor and has been characterized in Metallosphaera and Sulfolobus spp (Alber et al. J. Bacteriol. 188:8551-8559 (2006); Hugler et al. J. Bacteriol. 184:2404-2410 (2002)). The enzyme is encoded by Msed.sub.--0709 in Metallosphaera sedula (Alber et al. J. Bacteriol. 188:8551-8559 (2006); Berg et al. Science 318:1782-1786 (2007)). A gene encoding a malonyl-CoA reductase from Sulfolobus tokodaii was cloned and heterologously expressed in E. coli (Alber et al. J. Bacteriol. 188:8551-8559 (2006)). Although the aldehyde dehydrogenase functionality of these enzymes is similar to the bifunctional dehydrogenase from Chloroflexus aurantiacus, there is little sequence similarity. Both malonyl-CoA reductase enzyme candidates have high sequence similarity to aspartate-semialdehyde dehydrogenase, an enzyme catalyzing the reduction and concurrent dephosphorylation of aspartyl-4-phosphate to aspartate semialdehyde. Additional gene candidates can be found by sequence homology to proteins in other organisms including Sulfolobus solfataricus and Sulfolobus acidocaldarius. Yet another candidate for CoA-acylating aldehyde dehydrogenase is the ald gene from Clostridium beijerinckii (Toth et al., Appl Environ. Microbiol 65:4973-4980 (1999)). This enzyme has been reported to reduce acetyl-CoA and butyryl-CoA to their corresponding aldehydes. This gene is very similar to eutE that encodes acetaldehyde dehydrogenase of Salmonella typhimurium and E. coli (Toth et al., Appl Environ. Microbiol 65:4973-4980 (1999)).
TABLE-US-00044 TABLE 43 Gene Accession No. GI No. Organism Msed_0709 YP_001190808.1 146303492 Metallosphaera sedula mcr NP_378167.1 15922498 Sulfolobus tokodaii asd-2 NP_343563.1 15898958 Sulfolobus solfataricus Saci_2370 YP_256941.1 70608071 Sulfolobus acidocaldarius Ald AAT66436 49473535 Clostridium beijerinckii eutE AAA80209 687645 Salmonella typhimurium eutE P77445 2498347 Escherichia coli
[0245] Glutamate dehydrogenase (Step Q, FIG. 9B) and 4-aminobutyrate dehydrogenase (Step T, FIG. 9B) can be catalyzed by aminating oxidoreductases. Enzymes in this EC class (1.4.1.a) catalyze the oxidative deamination of alpha-amino acids with NAD.sup.+ or NADP.sup.+ as acceptor, and the reactions are typically reversible. Exemplary oxidoreductases operating on amino acids include glutamate dehydrogenase (deaminating), encoded by gdhA, leucine dehydrogenase (deaminating), encoded by ldh, and aspartate dehydrogenase (deaminating), encoded by nadX. The gdhA gene product from Escherichia coli (Korber et al. J. Mol. Biol. 234:1270-1273 (1993); McPherson and Wootton Nucleic. Acids Res. 11:5257-5266 (1983)), gdh from Thermotoga maritima (Kort et al. Extremophiles 1:52-60 (1997); Lebbink, et al. J. Mol. Biol. 280:287-296 (1998)); Lebbink et al. J. Mol. Biol. 289:357-369 (1999)), and gdhA1 from Halobacterium salinarum (Ingoldsby et al. Gene 349:237-244 (2005)) catalyze the reversible interconversion of glutamate to 2-oxoglutarate and ammonia, while favoring NADP(H), NAD(H), or both, respectively. The ldh gene of Bacillus cereus encodes the LeuDH protein that has a wide of range of substrates including leucine, isoleucine, valine, and 2-aminobutanoate (Ansorge and Kula Biotechnol Bioeng. 68:557-562 (2000); Stoyan et al. J. Biotechnol 54:77-80 (1997)). The nadX gene from Thermotoga maritime encoding for the aspartate dehydrogenase is involved in the biosynthesis of NAD (Yang et al. J. Biol. Chem. 278:8804-8808 (2003)).
TABLE-US-00045 TABLE 44 Gene Accession No. GI No. Organism gdhA P00370 118547 Escherichia coli gdh P96110.4 6226595 Thermotoga maritima gdhA1 NP_279651.1 15789827 Halobacterium salinarum ldh P0A393 61222614 Bacillus cereus nadX NP_229443.1 15644391 Thermotoga maritima
[0246] Additional glutamate dehydrogenase gene candidates are found in Bacillus subtilis (Khan et al., Biosci. Biotechnol Biochem. 69:1861-1870 (2005)), Nicotiana tabacum (Purnell et al., Planta 222:167-180 (2005)), Oryza sativa (Abiko et al., Plant Cell Physiol 46:1724-1734 (2005)), Haloferax mediterranei (Diaz et al., Extremophiles. 10:105-115 (2006)) and Halobactreium salinarum (Hayden et al., FEMS Microbiol Lett. 211:37-41 (2002)). The Nicotiana tabacum enzyme is composed of alpha and beta subunits encoded by gdh1 and gdh2 (Purnell et al., Planta 222:167-180 (2005)). Overexpression of the NADH-dependent glutamate dehydrogenase was found to improve ethanol production in engineered strains of S. cerevisiae (Roca et al., Appl Environ. Microbiol 69:4732-4736 (2003)).
TABLE-US-00046 TABLE 45 Gene Accession No. GI No. Organism rocG NP_391659.1 16080831 Bacillus subtilis gdh1 AAR11534.1 38146335 Nicotiana tabacum gdh2 AAR11535.1 38146337 Nicotiana tabacum GDH Q852M0 75243660 Oryza sativa GDH Q977U6 74499858 Haloferax mediterranei GDH P29051 118549 Halobactreium salinarum GDH2 NP_010066.1 6319986 Saccharomyces cerevisiae
[0247] An exemplary enzyme for catalyzing the conversion of aldehydes to their corresponding primary amines is lysine 6-dehydrogenase (EC 1.4.1.18), encoded by the lysDH genes. The lysine 6-dehydrogenase (deaminating), encoded by lysDH gene, catalyze the oxidative deamination of the .epsilon.-amino group of L-lysine to form 2-aminoadipate-6-semialdehyde, which in turn nonenzymatically cyclizes to form .DELTA.1-piperideine-6-carboxylate (Misono and Nagasaki J. Bacteriol. 150:398-401 (1982)). The lysDH gene from Geobacillus stearothermophilus encodes a thermophilic NAD-dependent lysine 6-dehydrogenase (Heydari et al. Appl Environ. Microbiol 70:937-942 (2004)). The lysDH gene from Aeropyrum pernix K1 is identified through homology from genome projects. Additional enzymes can be found in Agrobacterium tumefaciens (Hashimoto et al., J Biochem. 106:76-80 (1989); Misono et al., J Bacteriol. 150:398-401 (1982)) and Achromobacter denitrificans (Ruldeekulthamrong et al., BMB. Rep. 41:790-795 (2008)).
TABLE-US-00047 TABLE 46 Gene Accession No. GI No. Organism lysDH BAB39707 13429872 Geobacillus stearothermophilus lysDH NP_147035.1 14602185 Aeropyrum pernix K1 lysDH NP_353966 15888285 Agrobacterium tumefaciens lysDH AAZ94428 74026644 Achromobacter denitrificans
[0248] An enzyme that converts 3-oxoacids to 3-amino acids is 3,5-diaminohexanoate dehydrogenase (EC 1.4.1.11), an enzyme found in organisms that ferment lysine. The gene encoding this enzyme, kdd, was recently identified in Fusobacterium nucleatum (Kreimeyer et al., 282:7191-7197 (2007)). The enzyme has been purified and characterized in other organisms (Baker et al., 247:7724-7734 (1972); Baker et al., 13:292-299 (1974)) but the genes associated with these enzymes are not known. Candidates in Myxococcus xanthus, Porphyromonas gingivalis W83 and other sequenced organisms can be inferred by sequence homology.
TABLE-US-00048 TABLE 47 Gene Accession No. GI No. Organism kdd AAL93966.1 19713113 Fusobacterium nucleatum mxan_439 ABF87267.1 108462082 Myxococcus xanthus pg_1069 AAQ66183.1 34397119 Porphyromonas gingivalis
[0249] Steps R and U in FIG. 9B can be catalyzed by aminotransferases that reversibly convert an aldehyde or ketone to an amino group. Common amino donor/acceptor combinations include glutamate/alpha-ketoglutarate, alanine/pyruvate, and aspartate/oxaloacetate. Several enzymes have been shown to convert aldehydes to primary amines, and vice versa. Lysine-6-aminotransferase (EC 2.6.1.36) is one exemplary enzyme capable of forming a primary amine. This enzyme function, converting lysine to alpha-aminoadipate semialdehyde, has been demonstrated in yeast and bacteria. Candidates from Candida utilis (Hammer et al., J Basic Microbiol 32:21-27 (1992)), Flavobacterium lutescens (Fujii et al., J Biochem. 128:391-397 (2000)) and Streptomyces clavuligenus (Romero et al., J Ind. Microbiol Biotechnol 18:241-246 (1997)) have been characterized. A recombinant lysine-6-aminotransferase from S. clavuligenus was functionally expressed in E. coli (Tobin et al., J Bacteriol. 173:6223-6229 (1991)). The F. lutescens enzyme is specific to alpha-ketoglutarate as the amino acceptor (Soda et al., 7:4110-4119 (1968)). Other enzymes which convert aldehydes to terminal amines include the dat gene product in Acinetobacter baumanii encoding 2,4-diaminobutanoate:2-ketoglutarate 4-transaminase (Ikai et al., J Bacteriol. 179:5118-5125 (1997)). In addition to its natural substrate, 2,4-diaminobutyrate, DAT transaminates the terminal amines of lysine, 4-aminobutyrate and ornithine.
TABLE-US-00049 TABLE 48 Gene Accession No. GI No. Organism lat BAB13756.1 10336502 Flavobacterium lutescens lat AAA26777.1 153343 Streptomyces clavuligenus dat P56744.1 6685373 Acinetobacter baumanii
[0250] The conversion of an aldehyde to a terminal amine can also be catalyzed by gamma-aminobutyrate transaminase (GABA transaminase or 4-aminobutyrate transaminase). This enzyme naturally interconverts succinic semialdehyde and glutamate to 4-aminobutyrate and alpha-ketoglutarate and is known to have a broad substrate range (Schulz et al., 56:1-6 (1990); Liu et al., 43:10896-10905 (2004)). The two GABA transaminases in E. coli are encoded by gabT (Bartsch et al., J Bacteriol. 172:7035-7042 (1990)) and puuE (Kurihara et al., J. Biol. Chem. 280:4602-4608 (2005)). GABA transaminases in Mus musculus, Pseudomonas fluorescens, and Sus scrofa have been shown to react with a range of alternate substrates including 6-aminocaproic acid (Cooper, 113:80-82 (1985); SCOTT et al., 234:932-936 (1959)).
TABLE-US-00050 TABLE 49 Gene Accession No. GI No. Organism gabT NP_417148.1 16130576 Escherichia coli puuE NP_415818.1 16129263 Escherichia coli Abat NP_766549.2 37202121 Mus musculus gabT YP_257332.1 70733692 Pseudomonas fluorescens abat NP_999428.1 47523600 Sus scrofa
[0251] Additional enzyme candidates for interconverting aldehydes and primary amines are putrescine transminases or other diamine aminotransferases. The E. coli putrescine aminotransferase is encoded by the ygjG gene and the purified enzyme also was able to transaminate cadaverine and spermidine (Samsonova et al., BMC. Microbiol 3:2 (2003)). In addition, activity of this enzyme on 1,7-diaminoheptane and with amino acceptors other than 2-oxoglutarate (e.g., pyruvate, 2-oxobutanoate) has been reported (Samsonova et al., BMC. Microbiol 3:2 (2003); KIM, 239:783-786 (1964)). A putrescine aminotransferase with higher activity with pyruvate as the amino acceptor than alpha-ketoglutarate is the spuC gene of Pseudomonas aeruginosa (Lu et al., J Bacteriol. 184:3765-3773 (2002)).
TABLE-US-00051 TABLE 50 Gene Accession No. GI No. Organism ygjG NP_417544 145698310 Escherichia coli spuC AAG03688 9946143 Pseudomonas aeruginosa
[0252] Enzymes that transaminate 3-oxoacids include GABA aminotransferase (described above), beta-alanine/alpha-ketoglutarate aminotransferase and 3-amino-2-methylpropionate aminotransferase. Beta-alanine/alpha-ketoglutarate aminotransferase (WO08027742) reacts with beta-alanine to form malonic semialdehyde, a 3-oxoacid. The gene product of SkPYD4 in Saccharomyces kluyveri was shown to preferentially use beta-alanine as the amino group donor (Andersen et al., Gene 124:105-109 (1993)). SkUGA1 encodes a homologue of Saccharomyces cerevisiae GABA aminotransferase, UGA1 (Ramos et al., Eur. J. Biochem. 149:401-404 (1985)), whereas SkPYD4 encodes an enzyme involved in both beta-alanine and GABA transamination (Andersen and Hansen, Gene 124:105-109 (1993)). 3-Amino-2-methylpropionate transaminase catalyzes the transformation from methylmalonate semialdehyde to 3-amino-2-methylpropionate. The enzyme has been characterized in Rattus norvegicus and Sus scrofa and is encoded by Abat (Tamaki et al., 324:376-389 (2000); Kakimoto et al., 156:374-380 (1968)).
TABLE-US-00052 TABLE 51 Gene Accession No. GI No. Organism SkyPYD4 ABF58893.1 98626772 Lachancea kluyveri SkUGA1 ABF58894.1 98626792 Lachancea kluyveri UGA1 NP_011533.1 6321456 Saccharomyces cerevisiae Abat P50554.3 122065191 Rattus norvegicus Abat P80147.2 120968 Sus scrofa
[0253] Several aminotransferases transaminate the amino groups of amino acids to form 2-oxoacids. Aspartate aminotransferase is an enzyme that naturally transfers an oxo group from oxaloacetate to glutamate, forming alpha-ketoglutarate and aspartate. Aspartate is similar in structure to OHED and 2-AHD. Aspartate aminotransferase activity is catalyzed by, for example, the gene products of aspC from Escherichia coli (Yagi et al., 100:81-84 (1979): Yagi et al., 113:83-89 (1985)), AAT2 from Saccharomyces cerevisiae (Yagi et al., 92:35-43 (1982)) and ASP5 from Arabidopsis thaliana (Kwok et al., 55:595-604 (2004); de la et al., 46:414-425 (2006); Wilkie et al., Protein Expr. Purif. 12:381-389 (1998)). The enzyme from Rattus norvegicus has been shown to transaminate alternate substrates such as 2-aminohexanedioic acid and 2,4-diaminobutyric acid (Recasens et al., 19:4583-4589 (1980)). Aminotransferases that work on other amino-acid substrates may also be able to catalyze this transformation. Valine aminotransferase catalyzes the conversion of valine and pyruvate to 2-ketoisovalerate and alanine. The E. coli gene, avtA, encodes one such enzyme (Whalen et al., J. Bacteriol. 150:739-746 (1982)). This gene product also catalyzes the transamination of alpha-ketobutyrate to generate .alpha.-aminobutyrate, although the amine donor in this reaction has not been identified (Whalen et al., J. Bacteriol. 158:571-574 (1984)). The gene product of the E. coli serC catalyzes two reactions, phosphoserine aminotransferase and phosphohydroxythreonine aminotransferase (Lam et al., J. Bacteriol. 172:6518-6528 (1990)), and activity on nonphosphorylated substrates could not be detected (Drewke et al., FEBS. Lett. 390:179-182 (1996)).
TABLE-US-00053 TABLE 52 Gene Accession No. GI No. Organism aspC NP_415448.1 16128895 Escherichia coli AAT2 P23542.3 1703040 Saccharomyces cerevisiae ASP5 P46248.2 20532373 Arabidopsis thaliana Got2 P00507 112987 Rattus norvegicus avtA YP_026231.1 49176374 Escherichia coli serC NP_415427.1 16128874 Escherichia coli
[0254] Another enzyme candidate is alpha-aminoadipate aminotransferase (EC 2.6.1.39), an enzyme that participates in lysine biosynthesis and degradation in some organisms. This enzyme interconverts 2-aminoadipate and 2-oxoadipate, using alpha-ketoglutarate as the amino acceptor. Gene candidates are found in Homo sapiens (Okuno et al., Enzyme Protein 47:136-148 (1993)) and Thermus thermophilus (Miyazaki et al., 150:2327-2334 (2004)). The Thermus thermophilus enzyme, encoded by lysN, is active with several alternate substrates including oxaloacetate, 2-oxoisocaproate, 2-oxoisovalerate, and 2-oxo-3-methylvalerate.
TABLE-US-00054 TABLE 53 Gene Accession No. GI No. Organism lysN BAC76939.1 31096548 Thermus thermophilus AadAT-II Q8N5Z0.2 46395904 Homo sapiens
[0255] Alpha-ketoglutarate decarboxylase (Step I, FIG. 9B) and glutamate decarboxylase (Step S, FIG. 9B) all involve the decarboxylation of an alpha-ketoacid. The decarboxylation of keto-acids is catalyzed by a variety of enzymes with varied substrate specificities, including pyruvate decarboxylase (EC 4.1.1.1), benzoylformate decarboxylase (EC 4.1.1.7), alpha-ketoglutarate decarboxylase and branched-chain alpha-ketoacid decarboxylase.
[0256] Pyruvate decarboxylase (PDC), also termed keto-acid decarboxylase, is a key enzyme in alcoholic fermentation, catalyzing the decarboxylation of pyruvate to acetaldehyde. The enzyme from Saccharomyces cerevisiae has a broad substrate range for aliphatic 2-keto acids including 2-ketobutyrate, 2-ketovalerate, 3-hydroxypyruvate and 2-phenylpyruvate. This enzyme has been extensively studied, engineered for altered activity, and functionally expressed in E. coli (Killenberg-Jabs et al., 268:1698-1704 (2001); Li et al., Biochemistry. 38:10004-10012 (1999); ter Schure et al., Appl. Environ. Microbiol. 64:1303-1307 (1998)). The PDC from Zymomonas mobilus, encoded by pdc, also has a broad substrate range and has been a subject of directed engineering studies to alter the affinity for different substrates (Siegert et al., 18:345-357 (2005)). The crystal structure of this enzyme is available (Killenberg-Jabs et al., 268:1698-1704 (2001)). Other well-characterized PDC candidates include the enzymes from Acetobacter pasteurians (Chandra et al., 176:443-451 (2001)) and Kluyveromyces lactis (Krieger et al., 269:3256-3263 (2002)).
TABLE-US-00055 TABLE 54 Gene Accession No. GI No. Organism pdc P06672.1 118391 Zymomonas mobilus pdc1 P06169 30923172 Saccharomyces cerevisiae pdc AM21208 20385191 Acetobacter pasteurians pdc1 Q12629 52788279 Kluyveromyces lactis
[0257] Like PDC, benzoylformate decarboxylase (EC 4.1.1.7) has a broad substrate range and has been the target of enzyme engineering studies. The enzyme from Pseudomonas putida has been extensively studied and crystal structures of this enzyme are available (Polovnikova et al., 42:1820-1830 (2003); Hasson et al., 37:9918-9930 (1998)). Site-directed mutagenesis of two residues in the active site of the Pseudomonas putida enzyme altered the affinity (Km) of naturally and non-naturally occurring substrates (Siegert et al., 18:345-357 (2005)). The properties of this enzyme have been further modified by directed engineering (Lingen et al., Chembiochem. 4:721-726 (2003); Lingen et al., Protein Eng 15:585-593 (2002)). The enzyme from Pseudomonas aeruginosa, encoded by mdlC, has also been characterized experimentally (Barrowman et al., 34:57-60 (1986)). Additional gene candidates from Pseudomonas stutzeri, Pseudomonas fluorescens and other organisms can be inferred by sequence homology or identified using a growth selection system developed in Pseudomonas putida (Henning et al., Appl. Environ. Microbiol. 72:7510-7517 (2006)).
TABLE-US-00056 TABLE 55 Gene Accession No. GI No. Organism mdlC P20906.2 3915757 Pseudomonas putida mdlC Q9HUR2.1 81539678 Pseudomonas aeruginosa dpgB ABN80423.1 126202187 Pseudomonas stutzeri ilvB-1 YP_260581.1 70730840 Pseudomonas fluorescens
[0258] A third enzyme capable of decarboxylating 2-oxoacids is alpha-ketoglutarate decarboxylase (KGD). The substrate range of this class of enzymes has not been studied to date. The KDC from Mycobacterium tuberculosis (Tian et al., 102:10670-10675 (2005)) has been cloned and functionally expressed in other internal projects at Genomatica. However, it is not an ideal candidate for strain engineering because it is large (.about.130 kDa) and GC-rich. KDC enzyme activity has been detected in several species of rhizobia including Bradyrhizobium japonicum and Mesorhizobium loti (Green et al., 182:2838-2844 (2000)). Although the KDC-encoding gene(s) have not been isolated in these organisms, the genome sequences are available and several genes in each genome are annotated as putative KDCs. A KDC from Euglena gracilis has also been characterized but the gene associated with this activity has not been identified to date (Shigeoka et al., 288:22-28 (1991)). The first twenty amino acids starting from the N-terminus were sequenced MTYKAPVKDVKFLLDKVFKV (Shigeoka and Nakano, 288:22-28 (1991)). The gene could be identified by testing candidate genes containing this N-terminal sequence for KDC activity.
TABLE-US-00057 TABLE 56 Gene Accession No. GI No. Organism kgd O50463.4 160395583 Mycobacterium tuberculosis kgd NP_767092.1 27375563 Bradyrhizobium japonicum kgd NP_105204.1 13473636 Mesorhizobium loti
[0259] A fourth candidate enzyme for catalyzing this reaction is branched chain alpha-ketoacid decarboxylase (BCKA). This class of enzyme has been shown to act on a variety of compounds varying in chain length from 3 to 6 carbons (Oku et al., 263:18386-18396 (1988); Smit et al., 71:303-311 (2005)). The enzyme in Lactococcus lactis has been characterized on a variety of branched and linear substrates including 2-oxobutanoate, 2-oxohexanoate, 2-oxopentanoate, 3-methyl-2-oxobutanoate, 4-methyl-2-oxobutanoate and isocaproate (Smit et al., 71:303-311 (2005)). The enzyme has been structurally characterized (Berg et al., 318:1782-1786 (2007)). Sequence alignments between the Lactococcus lactis enzyme and the pyruvate decarboxylase of Zymomonas mobilus indicate that the catalytic and substrate recognition residues are nearly identical (Siegert et al., 18:345-357 (2005)), so this enzyme would be a promising candidate for directed engineering. Decarboxylation of alpha-ketoglutarate by a BCKA was detected in Bacillus subtilis; however, this activity was low (5%) relative to activity on other branched-chain substrates (Oku and Kaneda, 263:18386-18396 (1988)) and the gene encoding this enzyme has not been identified to date. Additional BCKA gene candidates can be identified by homology to the Lactococcus lactis protein sequence. Many of the high-scoring BLASTp hits to this enzyme are annotated as indolepyruvate decarboxylases (EC 4.1.1.74). Indolepyruvate decarboxylase (IPDA) is an enzyme that catalyzes the decarboxylation of indolepyruvate to indoleacetaldehyde in plants and plant bacteria.
TABLE-US-00058 Gene Accession No. GI No. Organism kdcA AAS49166.1 44921617 Lactococcus lactis
[0260] Recombinant branched chain alpha-keto acid decarboxylase enzymes derived from the E1 subunits of the mitochondrial branched-chain keto acid dehydrogenase complex from Homo sapiens and Bos taurus have been cloned and functionally expressed in E. coli (Davie et al., 267:16601-16606 (1992); Wynn et al., 267:12400-12403 (1992); Wynn et al., 267:1881-1887 (1992)). In these studies, the authors found that co-expression of chaperonins GroEL and GroES enhanced the specific activity of the decarboxylase by 500-fold (Wynn et al., 267:12400-12403 (1992)). These enzymes are composed of two alpha and two beta subunits.
TABLE-US-00059 TABLE 57 Gene Accession No. GI No. Organism BCKDHB NP_898871.1 34101272 Homo sapiens BCKDHA NP_000700.1 11386135 Homo sapiens BCKDHB P21839 115502434 Bos taurus BCKDHA P11178 129030 Bos taurus
[0261] Conversion of succinate semialdehyde to 4-hydroxybutyrate (Step C, FIG. 9B) can be catalyzed by an oxidoreducatse that converts an aldehyde to alcohol. Exemplary genes encoding enzymes that catalyze the conversion of an aldehyde to alcohol, that is, alcohol dehydrogenase or equivalently aldehyde reductase, include alrA encoding a medium-chain alcohol dehydrogenase for C2-C14 (Tani et al. Appl. Environ. Microbiol. 66:5231-5235 (2000)), ADH2 from Saccharomyces cerevisiae (Atsumi et al. Nature 451:86-89 (2008)), yqhD from E. coli which has preference for molecules longer than C3 (Sulzenbacher et al. Journal of Molecular Biology 342:489-502 (2004)), and bdh I and bdh II from C. acetobutylicum which converts butyrylaldehyde into butanol (Walter et al. Journal of Bacteriology 174:7149-7158 (1992)). The protein sequences for each of these exemplary gene products, if available, can be found using the following GenBank accession numbers:
TABLE-US-00060 TABLE 58 Gene Accession No. GI No. Organism alrA BAB12273.1 9967138 Acinetobacter sp. Strain M-1 ADH2 NP_014032.1 6323961 Saccharomyces cerevisiae yqhD NP_417484.1 16130909 Escherichia coli bdh I NP_349892.1 15896543 Clostridium acetobutylicum bdh II NP_349891.1 15896542 Clostridium acetobutylicum
[0262] Enzymes exhibiting 4-hydroxybutyrate dehydrogenase activity (EC 1.1.1.61) have been characterized in Ralstonia eutropha (Bravo et al. J. Forensic Sci. 49:379-387 (2004), Clostridium kluyveri (Wolff et al. Protein Expr. Purif. 6:206-212 (1995)) and Arabidopsis thaliana (Breitkreuz et al. J. Biol. Chem. 278:41552-41556 (2003)).
TABLE-US-00061 TABLE 59 Gene Accession No. GI No. Organism 4-HBd YP_726053.1 113867564 Ralstonia eutropha H16 4-HBd EDK35022.1 146348486 Clostridium kluyveri DSM 555 4-HBd Q94B07 75249805 Arabidopsis thaliana
[0263] The adhl gene from Geobacillus thermoglucosidasius M10EXG (Jeon et al., J Biotechnol 135:127-133 (2008)) has been indicated to exhibit high activity on both 4-hydroxybutanal and butanal. Thus this enzyme exhibits 1,4-butanediol dehydrogenase activity.
TABLE-US-00062 TABLE 60 Gene Accession No. GI No. Organism adh1 AAR91477.1 40795502 Geobacillus thermoglucosidasius M10EXG
[0264] Another exemplary enzyme is 3-hydroxyisobutyrate dehydrogenase which catalyzes the reversible oxidation of 3-hydroxyisobutyrate to methylmalonate semialdehyde. This enzyme participates in valine, leucine and isoleucine degradation and has been identified in bacteria, eukaryotes, and mammals. The enzyme encoded by P84067 from Thermus thermophilus HB8 has been structurally characterized (Lokanath et al. J Mol Biol 352:905-17 (2005)). The reversibility of the human 3-hydroxyisobutyrate dehydrogenase was demonstrated using isotopically-labeled substrate (Manning et al. Biochem J 231:481-484 (1985)). Additional genes encoding this enzyme include 3hidh in Homo sapiens (Hawes et al. Methods Enzymol. 324:218-228 (2000)) and Oryctolagus cuniculus (Chowdhury et al. Biosci. Biotechnol Biochem. 60:2043-2047 (1996); Hawes et al. Methods Enzymol. 324:218-228 (2000)), mmsb in Pseudomonas aeruginosa, and dhat in Pseudomonas putida (Aberhart et al. J Chem. Soc. [Perkin 1] 6:1404-1406 (1979); Chowdhury et al. Biosci. Biotechnol Biochem. 67:438-441 (2003); Chowdhury et al. Biosci. Biotechnol Biochem. 60:2043-2047 (1996)).
TABLE-US-00063 TABLE 61 Gene Accession No. GI No. Organism P84067 P84067 75345323 Thermus thermophilus mmsb P28811.1 127211 Pseudomonas aeruginosa dhat Q59477.1 2842618 Pseudomonas putida 3hidh P31937.2 12643395 Homo sapiens 3hidh P32185.1 416872 Oryctolagus cuniculus
[0265] Several 3-hydroxyisobutyrate dehydrogenase enzymes have also been shown to convert malonic semialdehyde to 3-hydroxyproprionic acid (3-HP). Three gene candidates exhibiting this activity are mmsB from Pseudomonas aeruginosa PAO1(62), mmsB from Pseudomonas putida KT2440 (Liao et al., US Publication 2005/0221466) and mmsB from Pseudomonas putida E23 (Chowdhury et al., Biosci. Biotechnol. Biochem. 60:2043-2047 (1996)). An enzyme with 3-hydroxybutyrate dehydrogenase activity in Alcaligenes faecalis M3A has also been identified (Gokam et al., U.S. Pat. No. 7,393,676; Liao et al., US Publication No. 2005/0221466). Additional gene candidates from other organisms including Rhodobacter spaeroides can be inferred by sequence similarity.
TABLE-US-00064 TABLE 62 Gene Accession No. GI No. Organism mmsB AAA25892.1 151363 Pseudomonas aeruginosa mmsB NP_252259.1 15598765 Pseudomonas aeruginosa PAO1 mmsB NP_746775.1 26991350 Pseudomonas putida KT2440 mmsB JC7926 60729613 Pseudomonas putida E23 orfB1 AAL26884 16588720 Rhodobacter spaeroides
[0266] The conversion of malonic semialdehyde to 3-HP can also be accomplished by two other enzymes: NADH-dependent 3-hydroxypropionate dehydrogenase and NADPH-dependent malonate semialdehyde reductase. An NADH-dependent 3-hydroxypropionate dehydrogenase is thought to participate in beta-alanine biosynthesis pathways from propionate in bacteria and plants (Rathinasabapathi, B. Journal of Plant Pathology 159:671-674 (2002); Stadtman, E. R. J. Am. Chem. Soc. 77:5765-5766 (1955)). This enzyme has not been associated with a gene in any organism to date. NADPH-dependent malonate semialdehyde reductase catalyzes the reverse reaction in autotrophic CO.sub.2-fixing bacteria. Although the enzyme activity has been detected in Metallosphaera sedula, the identity of the gene is not known (Alber et al. J. Bacteriol. 188:8551-8559 (2006)).
[0267] Step V in FIG. 9B depicts the conversion of alpha-ketoglutarate to succinyl-CoA. This reaction is catalyzed by alpha-ketoglutarate dehydrogenase, These enzymes are multi-enzyme complexes that catalyze a series of partial reactions which result in acylating oxidative decarboxylation of 2-keto-acids. This 2-keto-acid dehydrogenase complex occupies key positions in intermediary metabolism, and enzyme activity is typically tightly regulated (Fries et al. Biochemistry 42:6996-7002 (2003)). The enzyme shares a complex but common structure composed of multiple copies of three catalytic components: alpha-ketoacid decarboxylase (E1), dihydrolipoamide acyltransferase (E2) and dihydrolipoamide dehydrogenase (E3). The E3 component is shared among all 2-keto-acid dehydrogenase complexes in an organism, while the E1 and E2 components are encoded by different genes. The enzyme components are present in numerous copies in the complex and utilize multiple cofactors to catalyze a directed sequence of reactions via substrate channeling. The overall size of these dehydrogenase complexes is very large, with molecular masses between 4 and 10 million Da (that is, larger than a ribosome).
[0268] Activity of enzymes in the 2-keto-acid dehydrogenase family is normally low or limited under anaerobic conditions in E. coli. Increased production of NADH (or NADPH) could lead to a redox-imbalance, and NADH itself serves as an inhibitor to enzyme function. Engineering efforts have increased the anaerobic activity of the E. coli pyruvate dehydrogenase complex (Kim et al. Appl. Environ. Microbiol. 73:1766-1771 (2007); Kim et al. J. Bacteriol. 190:3851-3858) 2008); Zhou et al. Biotechnol. Lett. 30:335-342 (2008)). For example, the inhibitory effect of NADH can be overcome by engineering an H322Y mutation in the E3 component (Kim et al. J. Bacteriol. 190:3851-3858 (2008)). Structural studies of individual components and how they work together in complex provide insight into the catalytic mechanisms and architecture of enzymes in this family (Aevarsson et al. Nat. Struct. Biol. 6:785-792 (1999); Zhou et al. Proc. Natl. Acad. Sci. U.S.A. 98:14802-14807 (2001)). The substrate specificity of the dehydrogenase complexes varies in different organisms, but generally branched-chain ketoacid dehydrogenases have the broadest substrate range.
[0269] Alpha-ketoglutarate dehydrogenase (AKGD) converts alpha-ketoglutarate to succinyl-CoA and is the primary site of control of metabolic flux through the TCA cycle (Hansford, R. G. Curr. Top. Bioenerg. 10:217-278 (1980)). Encoded by genes sucA, sucB and lpd in E. coli, AKGD gene expression is downregulated under anaerobic conditions and during growth on glucose (Park et al. Mol. Microbiol. 15:473-482 (1995)). Structural studies of the catalytic core of the E2 component pinpoint specific residues responsible for substrate specificity (Knapp et al. J. Mol. Biol. 280:655-668 (1998)). The Bacillus subtilis AKGD, encoded by odhAB (E1 and E2) and pdhD (E3, shared domain), is regulated at the transcriptional level and is dependent on the carbon source and growth phase of the organism (Resnekov et al. Mol. Gen. Genet. 234:285-296 (1992)). In yeast, the LPD1 gene encoding the E3 component is regulated at the transcriptional level by glucose (Roy and Dawes J. Gen. Microbiol. 133:925-933 (1987)). The E1 component, encoded by KGD1, is also regulated by glucose and activated by the products of HAP2 and HAP3 (Repetto and Tzagoloff Mol. Cell Biol. 9:2695-2705 (1989)). The AKGD enzyme complex, inhibited by products NADH and succinyl-CoA, is well-studied in mammalian systems, as impaired function of has been linked to several neurological diseases (Tretter and dam-Vizi Philos. Trans. R. Soc. Lond B Biol. Sci. 360:2335-2345 (2005)).
TABLE-US-00065 TABLE 63 Gene Accession No. GI No. Organism sucA NP_415254.1 16128701 Escherichia coli str. K12 sucB NP_415255.1 16128702 Escherichia coli str. K12 lpd NP_414658.1 16128109 Escherichia coli str. K12 odhA P23129.2 51704265 Bacillus subtilis odhB P16263.1 129041 Bacillus subtilis pdhD P21880.1 118672 Bacillus subtilis KGD1 NP_012141.1 6322066 Saccharomyces cerevisiae KGD2 NP_010432.1 6320352 Saccharomyces cerevisiae LPD1 NP_116635.1 14318501 Saccharomyces cerevisiae
[0270] Steps A and H in FIG. 9B involve CoA transferase activities. The gene products of cat1, cat2, and cat3 of Clostridium kluyveri have been shown to exhibit succinyl-CoA, 4-hydroxybutyryl-CoA, and butyryl-CoA acetyltransferase activities (Seedorf et al. Proc Natl Acad Sci U.S.A. 105(6):2128-2133 (2008); Sohling and Gottschalk J Bacteriol 178(3):871-880 (1996)). Similar CoA transferase activities are also present in Trichomonas vaginalis (van Grinsven et al., J. Biol. Chem. 283:1411-1418 (2008)) and Trypanosoma brucei (Riviere et al., J. Biol. Chem. 279:45337-45346 (2004)).
TABLE-US-00066 TABLE 64 Gene Accession No. GI No. Organism cat1 P38946.1 729048 Clostridium kluyveri cat2 P38942.2 1705614 Clostridium kluyveri cat3 EDK35586.1 146349050 Clostridium kluyveri TVAG_395550 XP_001330176 123975034 Trichomonas vaginalis G3 Tb11.02.0290 XP_828352 71754875 Trypanosoma brucei
[0271] An additionally useful enzyme for this type of transformation is acyl-CoA:acetate-CoA transferase, also known as acetate-CoA transferase (EC 2.8.3.8), which has been shown to transfer the CoA moiety to acetate from a variety of branched and linear acyl-CoA substrates, including isobutyrate (Matthies and Schink Appl Environ Microbiol 58:1435-1439 (1992)), valerate (Vanderwinkel et al. Biochem. Biophys. Res Commun. 33:902-908 (1968)) and butanoate (Vanderwinkel, supra (1968)). This enzyme is encoded by atoA (alpha subunit) and atoD (beta subunit) in E. coli sp. K12 (Korolev et al. Acta Crystallogr. D Biol Crystallogr. 58:2116-2121 (2002); Vanderwinkel, supra (1968)). Similar enzymes exist in Corynebacterium glutamicum ATCC 13032 (Duncan et al., 68:5186-5190 (2002)), Clostridium saccharoperbutylacetonicum (Kosaka et al., Biosci. Biotechnol Biochem. 71:58-68 (2007)), and Clostridium acetobutylicum (Cary et al., 56:1576-1583 (1990); Wiesenbom et al., 55:323-329 (1989)).
TABLE-US-00067 TABLE 65 Gene Accession No. GI No. Organism atoA P76459.1 2492994 Escherichia coli K12 atoD P76458.1 2492990 Escherichia coli K12 actA YP_226809.1 62391407 Corynebacterium glutamicum cg0592 YP_224801.1 62389399 Corynebacterium glutamicum ctfA NP_149326.1 15004866 Clostridium acetobutylicum ctfB NP_149327.1 15004867 Clostridium acetobutylicum ctfA AAP42564.1 31075384 Clostridium saccharoperbutylacetonicum ctfB AAP42565.1 31075385 Clostridium saccharoperbutylacetonicum
[0272] The glutaconate-CoA-transferase (EC 2.8.3.12) enzyme from anaerobic bacterium Acidaminococcus fermentans reacts with diacid glutaconyl-CoA and 3-butenoyl-CoA (Mack and Buckel FEBS Lett. 405:209-212 (1997)). The genes encoding this enzyme are gctA and gctB. This enzyme has reduced but detectable activity with other CoA derivatives including glutaryl-CoA, 2-hydroxyglutaryl-CoA, adipyl-CoA and acrylyl-CoA (Buckel et al. Eur. J. Biochem. 118:315-321 (1981)). The enzyme has been cloned and expressed in E. coli (Mac et al. Eur. J. Biochem. 226:41-51 (1994)).
TABLE-US-00068 TABLE 66 Gene Accession No. GI No. Organism gctA CAA57199.1 559392 Acidaminococcus fermentans gctB CAA57200.1 559393 Acidaminococcus fermentans
[0273] Step H of FIG. 9B involves CoA synthetase or ligase reactions for 4-HB as the substrate. Exemplary CoA-ligases include the rat dicarboxylate-CoA ligase for which the sequence is yet uncharacterized (Vamecq et al., Biochemical Journal 230:683-693 (1985)), either of the two characterized phenylacetate-CoA ligases from P. chrysogenum (Lamas-Maceiras et al., Biochem. J. 395:147-155 (2005); Wang et al., Biochem Biophy Res Commun 360(2):453-458 (2007)), the phenylacetate-CoA ligase from Pseudomonas putida (Martinez-Blanco et al., J. Biol. Chem. 265:7084-7090 (1990)), and the 6-carboxyhexanoate-CoA ligase from Bacillus subtilis (Bower et al., J. Bacteriol. 178(14):4122-4130 (1996)). Additional candidate enzymes are acetoacetyl-CoA synthetases from Mus musculus (Hasegawa et al., Biochim Biophys Acta 1779:414-419 (2008)) and Homo sapiens (Ohgami et al., Biochem Pharmacol 65:989-994 (2003)) which naturally catalyze the ATP-dependant conversion of acetoacetate into acetoacetyl-CoA. 4-hydroxybutyryl-CoA synthetase activity has been demonstrated in Metallosphaera sedula (Berg et al., Science 318:1782-1786 (2007)). This function has been tentatively assigned to the Msed.sub.--1422 gene.
TABLE-US-00069 TABLE 67 Gene Accession No. GI No. Organism phl CAJ15517.1 77019264 Penicillium chrysogenum phlB ABS19624.1 152002983 Penicillium chrysogenum paaF AAC24333.2 22711873 Pseudomonas putida bioW NP_390902.2 50812281 Bacillus subtilis AACS NP_084486.1 21313520 Mus musculus AACS NP_076417.2 31982927 Homo sapiens Msed_1422 YP_001191504 146304188 Metallosphaera sedula
[0274] ADP-forming acetyl-CoA synthetase (ACD, EC 6.2.1.13) is another candidate enzyme that couples the conversion of acyl-CoA esters to their corresponding acids with the concurrent synthesis of ATP. Several enzymes with broad substrate specificities have been described in the literature. ACD I from Archaeoglobus fulgidus, encoded by AF1211, was shown to operate on a variety of linear and branched-chain substrates including acetyl-CoA, propionyl-CoA, butyryl-CoA, acetate, propionate, butyrate, isobutyrate, isovalerate, succinate, fumarate, phenylacetate, indoleacetate (Musfeldt et al., J Bacteriol 184:636-644 (2002)). The enzyme from Haloarcula marismortui (annotated as a succinyl-CoA synthetase) accepts propionate, butyrate, and branched-chain acids (isovalerate and isobutyrate) as substrates, and was shown to operate in the forward and reverse directions (Brasen et al., Arch Microbiol 182:277-287 (2004)). The ACD encoded by PAE3250 from hyperthermophilic crenarchaeon Pyrobaculum aerophilum showed the broadest substrate range of all characterized ACDs, reacting with acetyl-CoA, isobutyryl-CoA (preferred substrate) and phenylacetyl-CoA (Brasen et al., supra). The enzymes from A. fulgidus, H. marismortui and P. aerophilum have all been cloned, functionally expressed, and characterized in E. coli (Musfeldt et al., supra; Brasen et al., supra).
TABLE-US-00070 TABLE 68 Gene Accession No. GI No. Organism AF1211 NP_070039.1 11498810 Archaeoglobus fulgidus DSM 4304 scs YP_135572.1 55377722 Haloarcula marismortui ATCC 43049 PAE3250 NP_560604.1 18313937 Pyrobaculum aerophilum str. IM2
[0275] Exemplary phosphate transferring acyltransferases that transform 4-hydroxybutyryl-CoA to 4-hydroxybutyryl-phosphate include phosphotransacetylase, encoded by pta, and phosphotransbutyrylase, encoded by ptb. The pta gene from E. coli encodes an enzyme that can convert acetyl-CoA into acetyl-phosphate, and vice versa (Suzuki, T. Biochim. Biophys. Acta 191:559-569 (1969)). This enzyme can also utilize propionyl-CoA instead of acetyl-CoA forming propionate in the process (Hesslinger et al. Mol. Microbiol 27:477-492 (1998)). Similarly, the ptb gene from C. acetobutylicum encodes an enzyme that can convert butyryl-CoA into butyryl-phosphate (Walter et al. Gene 134(1): p. 107-11 (1993)); Huang et al. J Mol Microbiol Biotechnol 2(1): p. 33-38 (2000). Additional ptb genes can be found in butyrate-producing bacterium L2-50 (Louis et al. J. Bacteriol. 186:2099-2106 (2004)) and Bacillus megaterium (Vazquez et al. Curr. Microbiol 42:345-349 (2001)).
TABLE-US-00071 TABLE 69 Gene Accession No. GI No. Organism pta NP_416800.1 16130232 Escherichia coli ptb NP_349676 15896327 Clostridium acetobutylicum ptb AAR19757.1 38425288 butyrate-producing bacterium L2-50 ptb CAC07932.1 10046659 Bacillus megaterium
[0276] The conversion of 4-hydroxybutyryl-phosphate to 4-hydroxybutanal can be catalyzed by an oxidoreductase in the EC class 1.2.1. Aspartate semialdehyde dehydrogenase (ASD, EC 1.2.1.11) catalyzes the NADPH-dependent reduction of 4-aspartyl phosphate to aspartate-4-semialdehyde. ASD participates in amino acid biosynthesis and recently has been studied as an antimicrobial target (Hadfield et al., 40:14475-14483 (2001)). The E. coli ASD structure has been solved (Hadfield et al., 289:991-1002 (1999)) and the enzyme has been shown to accept the alternate substrate beta-3-methylaspartyl phosphate (Shames et al., 259:15331-15339 (1984)). The Haemophilus influenzae enzyme has been the subject of enzyme engineering studies to alter substrate binding affinities at the active site (Blanco et al., Acta Crystallogr. D. Biol. Crystallogr. 60:1388-1395 (2004); Blanco et al., Acta Crystallogr. D. Biol. Crystallogr. 60:1808-1815 (2004)). Other ASD candidates are found in Mycobacterium tuberculosis (Shafiani et al., J Appl Microbiol 98:832-838 (2005)), Methanococcus jannaschii (Faehnle et al., 353:1055-1068 (2005)), and the infectious microorganisms Vibrio cholera and Heliobacter pylori (Moore et al., Protein Expr. Purif. 25:189-194 (2002)). A related enzyme candidate is acetylglutamylphosphate reductase (EC 1.2.1.38), an enzyme that naturally reduces acetylglutamylphosphate to acetylglutamate-5-semialdehyde, found in S. cerevisiae (Pauwels et al., Eur. J Biochem. 270:1014-1024 (2003)), B. subtilis (O'Reilly et al., 140 (Pt 5):1023-1025 (1994)) and other organisms.
TABLE-US-00072 TABLE 70 Gene Accession No. GI No. Organism asd NP_417891.1 16131307 Escherichia coli asd YP_248335.1 68249223 Haemophilus influenzae asd AAB49996 1899206 Mycobacterium tuberculosis VC2036 NP_231670 15642038 Vibrio cholera asd YP_002301787.1 210135348 Heliobacter pylori ARG5,6 NP_010992.1 6320913 Saccharomyces cerevisiae argC NP_389001.1 16078184 Bacillus subtilis
[0277] Other exemplary enzymes in this class include glyceraldehyde 3-phosphate dehydrogenase which converts glyceraldehyde-3-phosphate into D-glycerate 1,3-bisphosphate (e.g., E. coli gapA (Branlant et al., Eur. J. Biochem. 150:61-66 (1985))), N-acetyl-gamma-glutamyl-phosphate reductase which converts N-acetyl-L-glutamate-5-semialdehyde into N-acetyl-L-glutamyl-5-phosphate (e.g., E. coli argC (Parsot et al., Gene. 68:275-283 (1988))), and glutamate-5-semialdehyde dehydrogenase which converts L-glutamate-5-semialdehyde into L-glutamyl-5-phospate (e.g., E. coli proA (Smith et al., J. Bacteriol. 157:545-551 (1984a))). Genes encoding glutamate-5-semialdehyde dehydrogenase enzymes from Salmonella typhimurium (Mahan et al., J Bacteriol. 156:1249-1262 (1983)) and Campylobacter jejuni (Louie et al., 240:29-35 (1993)) were cloned and expressed in E. coli.
TABLE-US-00073 TABLE 71 Gene Accession No. GI No. Organism gapA P0A9B2.2 71159358 Escherichia coli argC NP_418393.1 16131796 Escherichia coli proA NP_414778.1 16128229 Escherichia coli proA NP_459319.1 16763704 Salmonella typhimurium proA P53000.2 9087222 Campylobacter jejuni
[0278] In some embodiments, the 2-amino-4-ketopentanoate (AKP) thiolase encoded by one or more genes selected from the group consisting of ortA (.alpha.), ortB (.beta.), Amet.sub.--2368 (.alpha.), Amet.sub.--2369 (.beta.), Teth514.sub.--1478 (.alpha.), Teth514.sub.--1479 (.beta.), TTE1235 (.alpha.), and thrC (.beta.).
[0279] In some embodiments, the AKP dehydrogenase is encoded by one or more genes selected from the group consisting of thrA, akthr2, hom6, hom1, hom2, fadB, fadJ, Hbd2, Hbd1, hbd, HSD17B10, phbB, phaB, Msed.sub.--1423, Msed.sub.--0399, Msed.sub.--0389, Msed.sub.--1993, adh, adhA, adh-A, mdh, ldhA, ldh, and bdh.
[0280] In some embodiments, the 2-amino-4-hydroxypentanoate aminotransferase is encoded by one or more genes selected from the group consisting of aspC, AAT2, ASP5, got2, avtA, lysN, AadAT-II, dat, lat, ygjG, spuC, SkyPYD4, SkUGA1, UGA1, Abat, Abat, Gta-1, gabT, and puuE.
[0281] In some embodiments, the 2-amino-4-hydroxypentanoate oxidoreductase (deaminating) is encoded by one or more genes selected from the group consisting of gdhA, gdh, gdhA1, rocG, gdh1, gdh2, GDH, GDH2, ldh and nadX.
[0282] In some embodiments, the 2-oxo-4-hydroxypentanoate decarboxylase is encoded by one or more genes selected from the group consisting of pdc, pdc1, mdlC, dpgB, ilvB-1, kgd, kdcA, lysA, panD, cadA, ldc, ldcC, AF323910.1:1 . . . 1299, odc1, VV2.sub.--1235, dmpH, dmpE, xylII, xylIII, Reut_B5691, Reut_B5692, CAD, pad1, pofK (pad), padC, pad, adc, cbei.sub.--3835, CLL_A2135, RBAM.sub.--030030,
[0283] In some embodiments, the 3-hydroxybutyraldehdye reductase is encoded by one or more genes selected from the group consisting of alrA, ADH2, yqhD, bdh I, bdh II, adhA, 4hbd, adhI, P84067, mmsb, dhat, and 3hidh.
[0284] In some embodiments, the AKP aminotransferase is encoded by one or more genes selected from the group consisting of aspC, AAT2, ASP5, got2, avtA, lysN, AadAT-II, dat, lat, ygjG, spuC, SkyPYD4, SkUGA1, UGA1, Abat, Gta-1, gabT, and puuE.
[0285] In some embodiments, the AKP oxidoreductase (deaminating) is encoded by one or more genes selected from the group consisting of gdhA, gdh, gdhA1, rocG, gdh1, gdh2, GDH, GDH2, ldh and nadX. In some embodiments, the 2,4-dioxopentanoate decarboxylase is encoded by one or more genes selected from the group consisting of pdc, pdc1, mdlC, dpgB, ilvB-1, kgd, kdcA, lysA, panD, cadA, ldc, ldcC, AF323910.1:1 . . . 1299, odc1, VV2.sub.--1235, dmpH, dmpE, xylII, xylIII, Reut_B5691, Reut_B5692, CAD, pad1, padC, and pad, adc, cbei.sub.--3835, CLL_A2135, RBAM.sub.--030030.
[0286] In some embodiments, the 3-oxobutyraldehyde reductase (ketone reducing) is encoded by one or more genes selected from the group consisting of thrA, akthr2, hom6, hom1, hom2, fadB, fadJ, Hbd2, Hbd1, hbd, HSD17B10, phbB, phaB, Msed.sub.--1423, Msed.sub.--0399, Msed.sub.--0389, Msed.sub.--1993, adh, adhA, adh-A, mdh, ldhA, ldh, and bdh.
[0287] In some embodiments, the 3-oxobutyraldehyde reductase (aldehyde reducing) is encoded by one or more genes selected from the group consisting of alrA, ADH2, yqhD, bdh I, bdh II, adhA, 4hbd, adhI, P84067, mmsb, dhat, and 3hidh.
[0288] In some embodiments, the 4-hydroxy-2-butanone reductase is encoded by one or more genes selected from the group consisting of thrA, akthr2, hom6, hom1, hom2, fadB, fadJ, Hbd2, Hbd1, hbd, HSD17B10, phbB, phaB, Msed.sub.--1423, Msed.sub.--0399, Msed.sub.--0389, Msed.sub.--1993, adh, adhA, adh-A, mdh, ldhA, ldh, and bdh.
[0289] In some embodiments, the AKP decarboxylase is encoded by one or more genes selected from the group consisting of pdc, pdc1, mdlC, dpgB, ilvB-1, kgd, kdcA, lysA, panD, cadA, ldc, ldcC, AF323910.1:1 . . . 1299, odc1, VV2.sub.--1235, dmpH, dmpE, xylII, xylIII, Reut_B5691, Reut_B5692, CAD, pad1, pofK(pad), padC, pad.
[0290] In some embodiments, the 4-aminobutan-2-one aminotransferase is encoded by one or more genes selected from the group consisting of aspC, AAT2, ASP5, got2, avtA, lysN, AadAT-II, dat, lat, ygjG, spuC, SkyPYD4, SkUGA1, UGA1, Abat, Gta-1, gabT, and puuE.
[0291] In some embodiments, the 4-aminobutan-2-one oxidoreductase (deaminating) is encoded by one or more genes selected from the group consisting of gdhA, gdh, gdhA1, rocG, gdh1, gdh2, GDH, GDH2, ldh, nadX, kdd and lysDH.
[0292] In some embodiments, the 4-aminobutan-2-one ammonia-lyase is encoded by one or more genes selected from the group consisting of aspA, ansB, mal and BAA28709.
[0293] In some embodiments, the butenone hydratase is encoded by one or more genes selected from the group consisting of fumA, fumB, fumC, fumH, fum1, MmcB, MmcC, hmd, BACCAP.sub.--02294, ANACOL.sub.--02527, NtherDRAFT.sub.--2368, dmdA, dmdB, crt, crt1, ech paaA, paaB, phaA, phaB, maoC, paaF, paaG, abfD, Msed.sub.--1220, fadA, fadB, fadI, fadJ, and fadR.
[0294] In some embodiments, the AKP ammonia-lyase is encoded by one or more genes selected from the group consisting of aspA, ansB, mal and BAA28709.
[0295] In some embodiments, the acetylacrylate decarboxylase is encoded by one or more genes selected from the group consisting of pdc, pdc1, mdlC, dpgB, ilvB-1, kgd, kdcA, lysA, panD, cadA, ldc, ldcC, AF323910.1:1 . . . 1299, odc1, VV2.sub.--1235, dmpH, dmpE, xylII, xylIII, Reut_B5691, Reut_B5692, CAD, pad1, pofK (pad), padC, pad, adc, cbei.sub.--3835, CLL_A2135, RBAM.sub.--030030)
[0296] In some embodiments, the acetoacetyl-CoA reductase (CoA-dependent, aldehyde forming) is encoded by one or more genes selected from the group consisting of acr1, sucD, bphG, bld, adhE, Msed.sub.--0709, mcr, asd-2, Saci.sub.--2370, Ald, and eutE.
[0297] In some embodiments, the acetoacetyl-CoA reductase (CoA-dependent, alcohol forming) is encoded by one or more genes selected from the group consisting of adhE, adhE2, mcr, Rcas.sub.--2929, NAP1.sub.--02720, MGP2080.sub.--00535, and FAR.
[0298] In some embodiments, the acetoacetyl-CoA reductase (ketone reducing) is encoded by one or more genes selected from the group consisting of thrA, akthr2, hom6, hom1, hom2, fadB, fadJ, Hbd2, Hbd1, hbd, HSD17B10, phbB, phaB, Msed.sub.--1423, Msed.sub.--0399, Msed.sub.--0389, Msed.sub.--1993, adh, adhA, adh-A, mdh, ldhA, ldh, and bdh.
[0299] In some embodiments, the 3-hydroxybutyryl-CoA reductase (aldehyde forming) is encoded by one or more genes selected from the group consisting of acr1, sucD, bphG, bld, adhE, Msed.sub.--0709, mcr, asd-2, SacI.sub.--2370, Ald, and eutE.
[0300] In some embodiments, the 3-hydroxybutyryl-CoA reductase (alcohol forming) is encoded by one or more genes selected from the group consisting of adhE, adhE2, mcr, Rcas.sub.--2929, NAP1.sub.--02720, MGP2080.sub.--00535, and FAR.
[0301] In some embodiments, the 4-hydroxybutyryl-CoA dehydratase is encoded by one or more genes selected from the group consisting of fumA, fumB, fumC, fumH, fum1, MmcB, MmcC, hmd, BACCAP.sub.--02294, ANACOL.sub.--02527, NtherDRAFT.sub.--2368, dmdA, dmdB, crt, crt1, ech, paaA, paaB, phaA, phaB, maoC, paaF, paaG, abfD, Msed.sub.--1220, fadA, fadB, fadI, fadJ, and fadR.
[0302] In some embodiments, the crotonase is encoded by one or more genes selected from the group consisting of fumA, fumB, fumC, fumH, fumI, MmcB, MmcC, hmd, BACCAP.sub.--02294, ANACOL.sub.--02527, NtherDRAFT.sub.--2368, dmdA, dmdB, crt, crt1, ech paaA, paaB, phaA, phaB, maoC, paaF, paaG, abfD, Msed.sub.--1220, fadA, fadB, fadI, fadJ, and fadR.
[0303] In some embodiments, the 3-hydroxybutyryl-CoA hydrolase, transferase, or synthetase is encoded by one or more genes selected from a group consisting of acot12, ACH1, acot8, tesB, acot8, teas, ybgC, paaI, pbdB, gctA, gctB, and hibCH.
[0304] In some embodiments, 3-hydroxybutyrate reductase is encoded by one or more genes selected from a group consisting of car, npt, fadD9, BCG.sub.--2812c, nfa20150, nfa40540, SGR.sub.--6790, SGR.sub.--665, griC, griD.
[0305] The non-naturally occurring microbial organisms of the invention can be produced by introducing expressible nucleic acids encoding one or more of the enzymes or proteins participating in one or more 1,3-butanediol biosynthetic pathways. Depending on the host microbial organism chosen for biosynthesis, nucleic acids for some or all of a particular 1,3-butanediol biosynthetic pathway can be expressed. For example, if a chosen host is deficient in one or more enzymes or proteins for a desired biosynthetic pathway, then expressible nucleic acids for the deficient enzyme(s) or protein(s) are introduced into the host for subsequent exogenous expression. Alternatively, if the chosen host exhibits endogenous expression of some pathway genes, but is deficient in others, then an encoding nucleic acid is needed for the deficient enzyme(s) or protein(s) to achieve 1,3-butanediol biosynthesis. Thus, a non-naturally occurring microbial organism of the invention can be produced by introducing exogenous enzyme or protein activities to obtain a desired biosynthetic pathway or a desired biosynthetic pathway can be obtained by introducing one or more exogenous enzyme or protein activities that, together with one or more endogenous enzymes or proteins, produces a desired product such as 1,3-butanediol.
[0306] Host microbial organisms can be selected from, and the non-naturally occurring microbial organisms generated in, for example, bacteria, yeast, fungus or any of a variety of other microorganisms applicable to fermentation processes. Exemplary bacteria include species selected from Escherichia coli, Klebsiella oxytoca, Anaerobiospirillum succiniciproducens, Actinobacillus succinogenes, Mannheimia succiniciproducens, Rhizobium etli, Bacillus subtilis, Corynebacterium glutamicum, Gluconobacter oxydans, Zymomonas mobilis, Lactococcus lactis, Lactobacillus plantarum, Streptomyces coelicolor, Clostridium acetobutylicum, Pseudomonas fluorescens, and Pseudomonas putida. Exemplary yeasts or fungi include species selected from Saccharomyces cerevisiae, Schizosaccharomyces pombe, Kluyveromyces lactis, Kluyveromyces marxianus, Aspergillus terreus, Aspergillus niger, Pichia pastoris, Rhizopus arrhizus, Rhizobus oryzae, Yarrowia lipolytica, and the like. E. coli is a particularly useful host organism since it is a well characterized microbial organism suitable for genetic engineering. Other particularly useful host organisms include yeast such as Saccharomyces cerevisiae. It is understood that any suitable microbial host organism can be used to introduce metabolic and/or genetic modifications to produce a desired product.
[0307] Depending on the 1,3-butanediol biosynthetic pathway constituents of a selected host microbial organism, the non-naturally occurring microbial organisms of the invention will include at least one exogenously expressed 1,3-butanediol pathway-encoding nucleic acid and up to all encoding nucleic acids for one or more 1,3-butanediol biosynthetic pathways. For example, 1,3-butanediol biosynthesis can be established in a host deficient in a pathway enzyme or protein through exogenous expression of the corresponding encoding nucleic acid. In a host deficient in all enzymes or proteins of a 1,3-butanediol pathway, exogenous expression of all enzyme or proteins in the pathway can be included, although it is understood that all enzymes or proteins of a pathway can be expressed even if the host contains at least one of the pathway enzymes or proteins. For example, exogenous expression of all enzymes or proteins in a pathway for production of 1,3-butanediol can be included.
[0308] Given the teachings and guidance provided herein, those skilled in the art will understand that the number of encoding nucleic acids to introduce in an expressible form will, at least, parallel the 1,3-butanediol pathway deficiencies of the selected host microbial organism. Therefore, a non-naturally occurring microbial organism of the invention can have one, two, three, four, five, up to all nucleic acids encoding the enzymes or proteins constituting a 1,3-butanediol biosynthetic pathway disclosed herein. In some embodiments, the non-naturally occurring microbial organisms also can include other genetic modifications that facilitate or optimize 1,3-butanediol biosynthesis or that confer other useful functions onto the host microbial organism. One such other functionality can include, for example, augmentation of the synthesis of one or more of the 1,3-butanediol pathway precursors such as acetyl-CoA.
[0309] Generally, a host microbial organism is selected such that it produces the precursor of a 1,3-butanediol pathway, either as a naturally produced molecule or as an engineered product that either provides de novo production of a desired precursor or increased production of a precursor naturally produced by the host microbial organism. For example, acetyl-CoA is produced naturally in a host organism such as E. coli. A host organism can be engineered to increase production of a precursor, as disclosed herein. In addition, a microbial organism that has been engineered to produce a desired precursor can be used as a host organism and further engineered to express enzymes or proteins of a 1,3-butanediol pathway.
[0310] In some embodiments, a non-naturally occurring microbial organism of the invention is generated from a host that contains the enzymatic capability to synthesize 1,3-butanediol. In this specific embodiment it can be useful to increase the synthesis or accumulation of a 1,3-butanediol pathway product to, for example, drive 1,3-butanediol pathway reactions toward 1,3-butanediol production. Increased synthesis or accumulation can be accomplished by, for example, overexpression of nucleic acids encoding one or more of the above-described 1,3-butanediol pathway enzymes or proteins. Overexpression of the enzyme or enzymes and/or protein or proteins of the 1,3-butanediol pathway can occur, for example, through exogenous expression of the endogenous gene or genes, or through exogenous expression of the heterologous gene or genes. Therefore, naturally occurring organisms can be readily generated to be non-naturally occurring microbial organisms of the invention, for example, producing 1,3-butanediol, through overexpression of one, two, three, four, five, that is, up to all nucleic acids encoding 1,3-butanediol biosynthetic pathway enzymes or proteins. In addition, a non-naturally occurring organism can be generated by mutagenesis of an endogenous gene that results in an increase in activity of an enzyme in the 1,3-butanediol biosynthetic pathway.
[0311] In particularly useful embodiments, exogenous expression of the encoding nucleic acids is employed. Exogenous expression confers the ability to custom tailor the expression and/or regulatory elements to the host and application to achieve a desired expression level that is controlled by the user. However, endogenous expression also can be utilized in other embodiments such as by removing a negative regulatory effector or induction of the gene's promoter when linked to an inducible promoter or other regulatory element. Thus, an endogenous gene having a naturally occurring inducible promoter can be up-regulated by providing the appropriate inducing agent, or the regulatory region of an endogenous gene can be engineered to incorporate an inducible regulatory element, thereby allowing the regulation of increased expression of an endogenous gene at a desired time. Similarly, an inducible promoter can be included as a regulatory element for an exogenous gene introduced into a non-naturally occurring microbial organism.
[0312] It is understood that, in methods of the invention, any of the one or more exogenous nucleic acids can be introduced into a microbial organism to produce a non-naturally occurring microbial organism of the invention. The nucleic acids can be introduced so as to confer, for example, 1,3-butanediol biosynthetic pathway onto the microbial organism. Alternatively, encoding nucleic acids can be introduced to produce an intermediate microbial organism having the biosynthetic capability to catalyze some of the required reactions to confer 1,3-butanediol biosynthetic capability. For example, a non-naturally occurring microbial organism having 1,3-butanediol biosynthetic pathway can comprise at least two exogenous nucleic acids encoding desired enzymes or proteins. For example, the non-naturally occurring microbial organism can comprise at least two exogenous nucleic acids encoding an acetoacetyl-CoA reductase (CoA-dependent, alcohol forming) and a 4-hydroxy,2-butanone reductase (FIG. 2, steps D and F). Thus, it is understood that any combination of two or more enzymes or proteins of a biosynthetic pathway can be included in a non-naturally occurring microbial organism of the invention. Similarly, it is understood that any combination of three or more enzymes or proteins of a biosynthetic pathway can be included in a non-naturally occurring microbial organism of the invention and so forth, as desired, so long as the combination of enzymes and/or proteins of the desired biosynthetic pathway results in production of the corresponding desired product. For example, the non-naturally occurring microbial organism can comprise at least three exogenous nucleic acids encoding an acetoacetyl-CoA reductase (CoA-dependent, aldehyde forming), a 3-oxobutyraldehyde reductase (ketone reducing), and a 3-hydroxybutyraldehyde reductase (FIG. 2, steps A, B and C). Similarly, any combination of four, or more enzymes or proteins of a biosynthetic pathway as disclosed herein can be included in a non-naturally occurring microbial organism of the invention, as desired, so long as the combination of enzymes and/or proteins of the desired biosynthetic pathway results in production of the corresponding desired product. For example, the non-naturally occurring microbial organism can comprise at least five exogenous nucleic acids encoding an AKP thiolase, an AKP decarboxylase, a 4-aminobutan-2-one ammonia-lyase, a butenone hydratase, and a 4-hydroxy,2-butanone reductase, (FIG. 8B, steps A, E, F, G and H). Other individual pathways depicted in the figures are also contemplated embodiments of the compositions and methods provided herein.
[0313] In addition to the biosynthesis of 1,3-butanediol as described herein, the non-naturally occurring microbial organisms and methods of the invention also can be utilized in various combinations with each other and with other microbial organisms and methods well known in the art to achieve product biosynthesis by other routes. For example, one alternative to produce 1,3-butanediol other than use of the 1,3-butanediol producers is through addition of another microbial organism capable of converting 1,3-butanediol pathway intermediate to 1,3-butanediol. One such procedure includes, for example, the fermentation of a microbial organism that produces 1,3-butanediol pathway intermediate. The 1,3-butanediol pathway intermediate can then be used as a substrate for a second microbial organism that converts the 1,3-butanediol pathway intermediate to 1,3-butanediol. The 1,3-butanediol pathway intermediate can be added directly to another culture of the second organism or the original culture of the 1,3-butanediol pathway intermediate producers can be depleted of these microbial organisms by, for example, cell separation, and then subsequent addition of the second organism to the fermentation broth can be utilized to produce the final product without intermediate purification steps.
[0314] In other embodiments, the non-naturally occurring microbial organisms and methods of the invention can be assembled in a wide variety of subpathways to achieve biosynthesis of, for example, 1,3-butanediol. In these embodiments, biosynthetic pathways for a desired product of the invention can be segregated into different microbial organisms, and the different microbial organisms can be co-cultured to produce the final product. In such a biosynthetic scheme, the product of one microbial organism is the substrate for a second microbial organism until the final product is synthesized. For example, the biosynthesis of 1,3-butanediol can be accomplished by constructing a microbial organism that contains biosynthetic pathways for conversion of one pathway intermediate to another pathway intermediate or the product. Alternatively, 1,3-butanediol also can be biosynthetically produced from microbial organisms through co-culture or co-fermentation using two organisms in the same vessel, where the first microbial organism produces 1,3-butanediol intermediate and the second microbial organism converts the intermediate to 1,3-butanediol.
[0315] Given the teachings and guidance provided herein, those skilled in the art will understand that a wide variety of combinations and permutations exist for the non-naturally occurring microbial organisms and methods of the invention together with other microbial organisms, with the co-culture of other non-naturally occurring microbial organisms having subpathways and with combinations of other chemical and/or biochemical procedures well known in the art to produce 1,3-butanediol.
[0316] Sources of encoding nucleic acids for 1,3-butanediol pathway enzyme or protein can include, for example, any species where the encoded gene product is capable of catalyzing the referenced reaction. Such species include both prokaryotic and eukaryotic organisms including, but not limited to, bacteria, including archaea and eubacteria, and eukaryotes, including yeast, plant, insect, animal, and mammal, including human. Exemplary species for such sources include, for example, Escherichia coli, as well as other exemplary species disclosed herein or available as source organisms for corresponding genes. However, with the complete genome sequence available for now more than 550 species (with more than half of these available on public databases such as the NCBI), including 395 microorganism genomes and a variety of yeast, fungi, plant, and mammalian genomes, the identification of genes encoding the requisite 1,3-butanediol biosynthetic activity for one or more genes in related or distant species, including for example, homologues, orthologs, paralogs and nonorthologous gene displacements of known genes, and the interchange of genetic alterations between organisms is routine and well known in the art. Accordingly, the metabolic alterations allowing biosynthesis of 1,3-butanediol described herein with reference to a particular organism such as E. coli can be readily applied to other microorganisms, including prokaryotic and eukaryotic organisms alike. Given the teachings and guidance provided herein, those skilled in the art will know that a metabolic alteration exemplified in one organism can be applied equally to other organisms.
[0317] In some instances, such as when an alternative 1,3-butanediol biosynthetic pathway exists in an unrelated species, 1,3-butanediol biosynthesis can be conferred onto the host species by, for example, exogenous expression of a paralog or paralogs from the unrelated species that catalyzes a similar, yet non-identical metabolic reaction to replace the referenced reaction. Because certain differences among metabolic networks exist between different organisms, those skilled in the art will understand that the actual gene usage between different organisms can differ. However, given the teachings and guidance provided herein, those skilled in the art also will understand that the teachings and methods of the invention can be applied to all microbial organisms using the cognate metabolic alterations to those exemplified herein to construct a microbial organism in a species of interest that will synthesize 1,3-butanediol.
[0318] Host microbial organisms can be selected from, and the non-naturally occurring microbial organisms generated in, for example, bacteria, yeast, fungus or any of a variety of other microorganisms applicable to fermentation processes. Exemplary bacteria include species selected from Escherichia coli, Klebsiella oxytoca, Anaerobiospirillum succiniciproducens, Actinobacillus succinogenes, Mannheimia succiniciproducens, Rhizobium etli, Bacillus subtilis, Corynebacterium glutamicum, Gluconobacter oxydans, Zymomonas mobilis, Lactococcus lactis, Lactobacillus plantarum, Streptomyces coelicolor, Clostridium acetobutylicum, Pseudomonas fluorescens, and Pseudomonas putida. Exemplary yeasts or fungi include species selected from Saccharomyces cerevisiae, Schizosaccharomyces pombe, Kluyveromyces lactis, Kluyveromyces marxianus, Aspergillus terreus, Aspergillus niger and Pichia pastoris. E. coli is a particularly useful host organism since it is a well characterized microbial organism suitable for genetic engineering. Other particularly useful host organisms include yeast such as Saccharomyces cerevisiae.
[0319] Methods for constructing and testing the expression levels of a non-naturally occurring 1,3-butanediol-producing host can be performed, for example, by recombinant and detection methods well known in the art. Such methods can be found described in, for example, Sambrook et al., Molecular Cloning: A Laboratory Manual, Third Ed., Cold Spring Harbor Laboratory, New York (2001); and Ausubel et al., Current Protocols in Molecular Biology, John Wiley and Sons, Baltimore, Md. (1999).
[0320] Exogenous nucleic acid sequences involved in a pathway for production of 1,3-butanediol can be introduced stably or transiently into a host cell using techniques well known in the art including, but not limited to, conjugation, electroporation, chemical transformation, transduction, transfection, and ultrasound transformation. For exogenous expression in E. coli or other prokaryotic cells, some nucleic acid sequences in the genes or cDNAs of eukaryotic nucleic acids can encode targeting signals such as an N-terminal mitochondrial or other targeting signal, which can be removed before transformation into prokaryotic host cells, if desired. For example, removal of a mitochondrial leader sequence led to increased expression in E. coli (Hoffmeister et al., J. Biol. Chem. 280:4329-4338 (2005)). For exogenous expression in yeast or other eukaryotic cells, genes can be expressed in the cytosol without the addition of leader sequence, or can be targeted to mitochondrion or other organelles, or targeted for secretion, by the addition of a suitable targeting sequence such as a mitochondrial targeting or secretion signal suitable for the host cells. Thus, it is understood that appropriate modifications to a nucleic acid sequence to remove or include a targeting sequence can be incorporated into an exogenous nucleic acid sequence to impart desirable properties. Furthermore, genes can be subjected to codon optimization with techniques well known in the art to achieve optimized expression of the proteins.
[0321] An expression vector or vectors can be constructed to include one or more 1,3-butanediol biosynthetic pathway encoding nucleic acids as exemplified herein operably linked to expression control sequences functional in the host organism. Expression vectors applicable for use in the microbial host organisms of the invention include, for example, plasmids, phage vectors, viral vectors, episomes and artificial chromosomes, including vectors and selection sequences or markers operable for stable integration into a host chromosome. Additionally, the expression vectors can include one or more selectable marker genes and appropriate expression control sequences. Selectable marker genes also can be included that, for example, provide resistance to antibiotics or toxins, complement auxotrophic deficiencies, or supply critical nutrients not in the culture media. Expression control sequences can include constitutive and inducible promoters, transcription enhancers, transcription terminators, and the like which are well known in the art. When two or more exogenous encoding nucleic acids are to be co-expressed, both nucleic acids can be inserted, for example, into a single expression vector or in separate expression vectors. For single vector expression, the encoding nucleic acids can be operationally linked to one common expression control sequence or linked to different expression control sequences, such as one inducible promoter and one constitutive promoter. The transformation of exogenous nucleic acid sequences involved in a metabolic or synthetic pathway can be confirmed using methods well known in the art. Such methods include, for example, nucleic acid analysis such as Northern blots or polymerase chain reaction (PCR) amplification of mRNA, or immunoblotting for expression of gene products, or other suitable analytical methods to test the expression of an introduced nucleic acid sequence or its corresponding gene product. It is understood by those skilled in the art that the exogenous nucleic acid is expressed in a sufficient amount to produce the desired product, and it is further understood that expression levels can be optimized to obtain sufficient expression using methods well known in the art and as disclosed herein.
[0322] The invention provides a method for producing 1,3-BDO that includes culturing the non-naturally occurring microbial organism disclosed herein, under conditions and for a sufficient period of time to produce 1,3-BDO, including organisms that incorporate one, two, three, four, five, up to all exogenous nucleic acids encoding enzymes that complete a 1,3-BDO pathway. The 1,3-BDO pathways include a set of 1,3-BDO pathway enzymes, where the set of 1,3-BDO pathway enzymes are identified as above, namely: (a) (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP dehydrogenase; (3) a 2-amino-4-hydroxypentanoate aminotransferase or oxidoreductase (deaminating); (4) a 2-oxo-4-hydroxypentanoate decarboxylase; and (5) a 3-hydroxybutyraldehyde reductase; (b) (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP aminotransferase or oxidoreductase (deaminating); (3) a 2,4-dioxopentanoate decarboxylase; (4) a 3-oxobutyraldehyde reductase (ketone reducing); and (5) a 3-hydroxybutyraldehyde reductase; (c) (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP aminotransferase or oxidoreductase (deaminating); (3) a 2,4-dioxopentanoate decarboxylase; (4) a 3-oxobutyraldehyde reductase (aldehyde reducing); and (5) a 4-hydroxy-2-butanone reductase; (d) (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP decarboxylase; (3) a 4-aminobutan-2-one aminotransferase or oxidoreductase (deaminating); (4) a 3-oxobutyraldehyde reductase (ketone reducing); and (5) a 3-hydroxybutyraldehyde reductase; (e) (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP decarboxylase; (3) a 4-aminobutan-2-one aminotransferase or oxidoreductase (deaminating); (4) a 3-oxobutyraldehyde reductase (aldehyde reducing); and (5) a 4-hydroxy-2-butanone reductase; (f) (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP decarboxylase; (3) a 4-aminobutan-2-one ammonia-lyase; (4) a butanone hydratase; and (5) a 4-hydroxy-2-butanone reductase; (g) (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP ammonia-lyase; (3) an acetylacrylate decarboxylase; (4) a butanone hydratase; and (5) a 4-hydroxy-2-butanone reductase; (h) (1) an acetoacetyl-CoA reductase (CoA-dependent, aldehyde forming); (2) a 3-oxobutyraldehyde reductase (ketone reducing); and (3) a 3-hydroxybutyraldehyde reductase; (i) (1) an acetoacetyl-CoA reductase (CoA dependent, alcohol faulting) and (2) a 4-hydroxy-2-butanone reductase; (j) (1) an acetoacetyl-CoA reductase (CoA-dependent, aldehyde forming); (2) a 3-oxobutyraldehyde reductase (aldehyde reducing); and (3) a 4-hydroxy-2-butanone reductase; (k) (1) an acetoacetyl-CoA reductase (ketone reducing) and (2) a 3-hydroxybutyryl-CoA reductase (alcohol forming); (l) (1) an acetoacetyl-CoA reductase (ketone reducing); (2) a 3-hydroxybutyryl-CoA reductase (aldehyde forming); and (3) a 3-hydroxybutyraldehyde reductase; (m) (1) a 4-hydroxybutyryl-CoA dehydratase; (2) a crotonase; and (3) a 3-hydroxybutyryl-CoA reductase (alcohol forming); and (n) (1) a 4-hydroxybutyryl-CoA dehydratase; (2) a crotonase; (3) a 3-hydroxybutyryl-CoA reductase (aldehyde forming); and (4) a 3-hydroxybutyraldehyde reductase.
[0323] In some embodiments, a method for producing 1,3-BDO includes culturing a non-naturally occurring microbial organism provided herein under conditions and for a sufficient period of time to produce 1,3-BDO. In some embodiments, the non-naturally occurring microbial organism has a 1,3-butanediol pathway, wherein said microbial organism comprises at least one exogenous nucleic acid encoding a 1,3-butanediol pathway enzyme expressed in a sufficient amount to produce 1,3-butanediol. In some embodiments, the non-naturally occurring microbial organism includes at least one of
[0324] (i) a reductive TCA pathway, wherein said microbial organism comprises at least one exogenous nucleic acid encoding a reductive TCA pathway enzyme selected from the group consisting of an ATP-citrate lyase, citrate lyase, a fumarate reductase, and an alpha-ketoglutarate:ferredoxin oxidoreductase;
[0325] (ii) a reductive TCA pathway, wherein said microbial organism comprises at least one exogenous nucleic acid encoding a reductive TCA pathway enzyme selected from the group consisting of a pyruvate:ferredoxin oxidoreductase, a phosphoenolpyruvate carboxylase, a phosphoenolpyruvate carboxykinase, a CO dehydrogenase, and an H.sub.2 hydrogenase; or
[0326] (iii) at least one exogenous nucleic acid encodes an enzyme selected from the group consisting of a CO dehydrogenase, an H.sub.2 hydrogenase, and combinations thereof;
[0327] wherein said 1,3-butanediol pathway comprises a pathway selected from the group consisting of:
[0328] (a) (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP dehydrogenase; (3) a 2-amino-4-hydroxypentanoate aminotransferase or oxidoreductase (deaminating); (4) a 2-oxo-4-hydroxypentanoate decarboxylase; and (5) a 3-hydroxybutyraldehyde reductase;
[0329] (b) (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP aminotransferase or oxidoreductase (deaminating); (3) a 2,4-dioxopentanoate decarboxylase; (4) a 3-oxobutyraldehyde reductase (ketone reducing); and (5) a 3-hydroxybutyraldehyde reductase;
[0330] (c) (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP aminotransferase or oxidoreductase (deaminating); (3) a 2,4-dioxopentanoate decarboxylase; (4) a 3-oxobutyraldehyde reductase (aldehyde reducing); and (5) a 4-hydroxy-2-butanone reductase;
[0331] (d) (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP decarboxylase; (3) a 4-aminobutan-2-one aminotransferase or oxidoreductase (deaminating); (4) a 3-oxobutyraldehyde reductase (ketone reducing); and (5) a 3-hydroxybutyraldehyde reductase;
[0332] (e) (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP decarboxylase; (3) a 4-aminobutan-2-one aminotransferase or oxidoreductase (deaminating); (4) a 3-oxobutyraldehyde reductase (aldehyde reducing); and (5) a 4-hydroxy-2-butanone reductase;
[0333] (f) (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP decarboxylase; (3) a 4-aminobutan-2-one ammonia-lyase; (4) a butenone hydratase; and (5) a 4-hydroxy-2-butanone reductase;
[0334] (g) (1) a 2-amino-4-ketopentanoate (AKP) thiolase; (2) an AKP ammonia-lyase; (3) an acetylacrylate decarboxylase; (4) a butenone hydratase; and (5) a 4-hydroxy-2-butanone reductase;
[0335] (h) (1) an acetoacetyl-CoA reductase (CoA-dependent, aldehyde forming); (2) a 3-oxobutyraldehyde reductase (ketone reducing); and (3) a 3-hydroxybutyraldehyde reductase;
[0336] (i) (1) an acetoacetyl-CoA reductase (CoA dependent, alcohol forming) and (2) a 4-hydroxy-2-butanone reductase;
[0337] (j) (1) an acetoacetyl-CoA reductase (CoA-dependent, aldehyde forming); (2) a 3-oxobutyraldehyde reductase (aldehyde reducing); and (3) a 4-hydroxy-2-butanone reductase;
[0338] (k) (1) an acetoacetyl-CoA reductase (ketone reducing) and (2) a 3-hydroxybutyryl-CoA reductase (alcohol forming);
[0339] (l) (1) an acetoacetyl-CoA reductase (ketone reducing); (2) a 3-hydroxybutyryl-CoA reductase (aldehyde forming); and (3) a 3-hydroxybutyraldehyde reductase;
[0340] (m) (1) a 4-hydroxybutyryl-CoA dehydratase; (2) a crotonase; and (3) a 3-hydroxybutyryl-CoA reductase (alcohol forming); and
[0341] (n) (1) a 4-hydroxybutyryl-CoA dehydratase; (2) a crotonase; (3) a 3-hydroxybutyryl-CoA reductase (aldehyde forming); and (4) a 3-hydroxybutyraldehyde reductase;
[0342] (o) (1) a succinyl-CoA transferase, a succinyl-CoA synthetase or a succinyl-CoA ligase, (2) a succinyl-CoA reductase (aldehyde forming), (3) a 4-hydroxybutyrate dehydrogenase, (4) a 4-hydroxybutyrate kinase, (5) a phosphotrans-4-hydroxybutyrylase, (6) a 4-hydroxybutyryl-CoA dehydratase, (7) a crotonase, (8) a 3-hydroxybutyryl-CoA reductase (aldehyde forming), and (9) a 3-hydroxybutanal reductase;
[0343] (p) (1) (i) an alpha-ketoglutarate decarboxylase, or (ii) (a) a glutamate dehydrogenase and/or a glutamate transaminase, (b) a glutamate decarboxylase, and (c) a 4-aminobutyrate dehydrogenase and/or a 4-aminobutyrate transaminase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyrate kinase, (4) a phosphotrans-4-hydroxybutyrylase, (5) a 4-hydroxybutyryl-CoA dehydratase; (6) a crotonase, (7) a 3-hydroxybutyryl-CoA reductase (aldehyde forming), and (8) a 3-hydroxybutanal reductase;
[0344] (q) (1) (i) an alpha-ketoglutarate decarboxylase, or (ii) (a) a glutamate dehydrogenase and/or a glutamate transaminase, (b) a glutamate decarboxylase, and (c) a 4-aminobutyrate dehydrogenase and/or a 4-aminobutyrate transaminase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (4) a 4-hydroxybutyryl-CoA dehydratase, (5) a crotonase, (6) a 3-hydroxybutyryl-CoA reductase (aldehyde forming), and (7) a 3-hydroxybutanal reductase;
[0345] (r) (1) (i) an alpha-ketoglutarate decarboxylase, or (ii) (a) a glutamate dehydrogenase and/or a glutamate transaminase, (b) a glutamate decarboxylase, and (c) a 4-aminobutyrate dehydrogenase and/or a 4-aminobutyrate transaminase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyrate kinase, (4) a phosphotrans-4-hydroxybutyrylase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, and (7) a 3-hydroxybutyryl-CoA reductase (alcohol forming);
[0346] (s) (1) (i) an alpha-ketoglutarate decarboxylase, or (ii) (a) a glutamate dehydrogenase and/or a glutamate transaminase, (b) a glutamate decarboxylase, and (c) a 4-aminobutyrate dehydrogenase and/or a 4-aminobutyrate transaminase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (4) a 4-hydroxybutyryl-CoA dehydratase, (5) a crotonase, and (6) a 3-hydroxybutyryl-CoA reductase (alcohol forming);
[0347] (t) (1) (i) an alpha-ketoglutarate decarboxylase, or (ii) (a) a glutamate dehydrogenase and/or a glutamate transaminase, (b) a glutamate decarboxylase, and (c) a 4-aminobutyrate dehydrogenase and/or a 4-aminobutyrate transaminase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyrate kinase, (4) a phosphotrans-4-hydroxybutyrylase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, (7) a 3-hydroxybutyryl-CoA hydrolase, transferase or synthetase, and (8) a 3-hydroxybutyrate reductase;
[0348] (u) (1) (i) an alpha-ketoglutarate decarboxylase, or (ii) (a) a glutamate dehydrogenase and/or a glutamate transaminase, (b) a glutamate decarboxylase, and (c) a 4-aminobutyrate dehydrogenase and/or a 4-aminobutyrate transaminase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (4) a 4-hydroxybutyryl-CoA dehydratase, (5) a crotonase, (6) a 3-hydroxybutyryl-CoA hydrolase, transferase or synthetase, and (7) a 3-hydroxybutyrate reductase.
[0349] (v) (1) a succinate reductase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyrate kinase, (4) a phosphotrans-4-hydroxybutyrylase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, (7) a 3-hydroxybutyryl-CoA reductase (aldehyde forming), and (8) a 3-hydroxybutanal reductase;
[0350] (w) (1) a succinate reductase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (4) a 4-hydroxybutyryl-CoA dehydratase, (5) a crotonase, (6) a 3-hydroxybutyryl-CoA reductase (aldehyde forming), (7) a 3-hydroxybutanal reductase;
[0351] (x) (1) a succinate reductase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyrate kinase, (4) a phosphotrans-4-hydroxybutyrylase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, and (7) a 3-hydroxybutyryl-CoA reductase (alcohol forming);
[0352] (y) (1) a succinate reductase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (4) a 4-hydroxybutyryl-CoA dehydratase, (5) a crotonase, and (6) a 3-hydroxybutyryl-CoA reductase (alcohol forming);
[0353] (z) (1) a succinate reductase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyrate kinase, (4) a phosphotrans-4-hydroxybutyrylase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, (7) a 3-hydroxybutyryl-CoA hydrolase, transferase or synthetase, and (8) a 3-hydroxybutyrate reductase;
[0354] (aa) (1) a succinate reductase, (2) a 4-hydroxybutyrate dehydrogenase, (3) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (4) a 4-hydroxybutyryl-CoA dehydratase, (5) a crotonase, (6) a 3-hydroxybutyryl-CoA hydrolase, transferase or synthetase, and (7) a 3-hydroxybutyrate reductase;
[0355] (bb) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (aldehyde forming), (3) a 4-hydroxybutyrate dehydrogenase, (5) a 4-hydroxybutyrate kinase, (6) a pPhosphotrans-4-hydroxybutyrylase, (7) a 4-hydroxybutyryl-CoA dehydratase, (8) a crotonase, and (9) a 3-hydroxybutyryl-CoA reductase (alcohol forming);
[0356] (cc) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (aldehyde forming), (3) a 4-hydroxybutyrate dehydrogenase, (4) a 4-hydroxybutyrate kinase, (5) a phosphotrans-4-hydroxybutyrylase, (6) a 4-hydroxybutyryl-CoA dehydratase, (7) a crotonase, (8) a 3-hydroxybutyryl-CoA hydrolase, transferase or synthetase, and (9) a 3-hydroxybutyrate reductase;
[0357] (dd) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (alcohol forming), (3) a 4-hydroxybutyrate kinase, (4) a phosphotrans-4-hydroxybutyrylase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, (7) a 3-hydroxybutyryl-CoA reductase (aldehyde forming), and (8) a 3-hydroxybutanal reductase;
[0358] (ee) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (alcohol forming), (3) a 4-hydroxybutyrate kinase, (4) a phosphotrans-4-hydroxybutyrylase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, and (7) a 3-hydroxybutyryl-CoA reductase (alcohol forming);
[0359] (ff) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (alcohol forming), (3) a 4-hydroxybutyrate kinase, (4) a phosphotrans-4-hydroxybutyrylase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, (7) a 3-hydroxybutyryl-CoA hydrolase, transferase or synthetase, and (8) a 3-hydroxybutyrate reductase;
[0360] (gg) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (aldehyde forming), (3) a 4-hydroxybutyrate dehydrogenase, (4) a 4-hydroxybutyryl-CoA transferase, or 4-hydroxybutyryl-CoA synthetase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, (7) a 3-hydroxybutyryl-CoA reductase (aldehyde forming), and (8) a 3-hydroxybutanal reductase;
[0361] (hh) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (aldehyde forming), (3) a 4-hydroxybutyrate dehydrogenase, (4) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, and (7) a 3-hydroxybutyryl-CoA reductase (alcohol forming);
[0362] (ii) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (aldehyde forming), (3) a 4-hydroxybutyrate dehydrogenase, (4) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, (7) a 3-hydroxybutyryl-CoA hydrolase, transferase or synthetase, (8) a 3-hydroxybutyrate reductase;
[0363] (jj) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (alcohol forming), (3) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (4) a 4-hydroxybutyryl-CoA dehydratase, (5) a crotonase, (6) a 3-hydroxybutyryl-CoA reductase (aldehyde forming), and (7) a 3-hydroxybutanal reductase;
[0364] (kk) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (alcohol forming), (3) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (4) a 4-hydroxybutyryl-CoA dehydratase, (5) a crotonase, and (6) a 3-hydroxybutyryl-CoA reductase (alcohol forming); and
[0365] (ll) (1) a succinyl-CoA transferase, succinyl-CoA synthetase or succinyl-CoA ligase, (2) a succinyl-CoA reductase (alcohol forming), (3) a 4-hydroxybutyryl-CoA transferase or 4-hydroxybutyryl-CoA synthetase, (5) a 4-hydroxybutyryl-CoA dehydratase, (6) a crotonase, (7) a 3-hydroxybutyryl-CoA hydrolase, transferase or synthetase, and (8) a 3-hydroxybutyrate reductase.
[0366] In some embodiments, the method includes a non-naturally occurring microbial organism (e.g., having pathway (i)) that further includes an exogenous nucleic acid encoding an enzyme selected from a pyruvate:ferredoxin oxidoreductase, an aconitase, an isocitrate dehydrogenase, a succinyl-CoA synthetase, a succinyl-CoA transferase, a fumarase, a malate dehydrogenase, an acetate kinase, a phosphotransacetylase, an acetyl-CoA synthetase, an NAD(P)H:ferredoxin oxidoreductase, ferredoxin, and combinations thereof.
[0367] In some embodiments, the method includes a non-naturally occurring microbial organism (e.g., having pathway (ii)) that further includes an exogenous nucleic acid encoding an enzyme selected from an aconitase, an isocitrate dehydrogenase, a succinyl-CoA synthetase, a succinyl-CoA transferase, a fumarase, a malate dehydrogenase, and combinations thereof.
[0368] In some embodiments, the method includes a microbial organism that includes two, three, four, five, six, seven, eight or nine exogenous nucleic acids, each encoding a 1,3-BDO pathway enzyme.
[0369] In some embodiments, the method includes a microbial organism that comprises exogenous nucleic acids encoding each of the enzymes of at least one of the 1,3-butanediol pathways selected from the group consisting of (a)-(ll).
[0370] In some embodiments, the method includes at least one exogenous nucleic acid that is a heterologous nucleic acid.
[0371] In some embodiments, the method includes the non-naturally occurring microbial organism in a substantially anaerobic culture medium.
[0372] Suitable purification and/or assays to test for the production of 1,3-butanediol can be performed using well known methods. Suitable replicates such as triplicate cultures can be grown for each engineered strain to be tested. For example, product and byproduct formation in the engineered production host can be monitored. The final product and intermediates, and other organic compounds, can be analyzed by methods such as HPLC (High Performance Liquid Chromatography), GC-MS (Gas Chromatography-Mass Spectroscopy) and LC-MS (Liquid Chromatography-Mass Spectroscopy) or other suitable analytical methods using routine procedures well known in the art. The release of product in the fermentation broth can also be tested with the culture supernatant. Byproducts and residual glucose can be quantified by HPLC using, for example, a refractive index detector for glucose and alcohols, and a UV detector for organic acids (Lin et al., Biotechnol. Bioeng. 90:775-779 (2005)), or other suitable assay and detection methods well known in the art. The individual enzyme or protein activities from the exogenous DNA sequences can also be assayed using methods well known in the art (see, for example, WO/2008/115840 and Hanai et al., Appl. Environ. Microbiol. 73:7814-7818 (2007)).
[0373] The 1,3-butanediol can be separated from other components in the culture using a variety of methods well known in the art. Such separation methods include, for example, extraction procedures as well as methods that include continuous liquid-liquid extraction, pervaporation, membrane filtration, membrane separation, reverse osmosis, electrodialysis, distillation, crystallization, centrifugation, extractive filtration, ion exchange chromatography, size exclusion chromatography, adsorption chromatography, and ultrafiltration. All of the above methods are well known in the art.
[0374] Any of the non-naturally occurring microbial organisms described herein can be cultured to produce and/or secrete the biosynthetic products of the invention. For example, the 1,3-butanediol producers can be cultured for the biosynthetic production of 1,3-butanediol.
[0375] For the production of 1,3-butanediol, the recombinant strains are cultured in a medium with carbon source and other essential nutrients. It is sometimes desirable and can be highly desirable to maintain anaerobic conditions in the fermenter to reduce the cost of the overall process. Such conditions can be obtained, for example, by first sparging the medium with nitrogen and then sealing the flasks with a septum and crimp-cap. For strains where growth is not observed anaerobically, microaerobic conditions can be applied by perforating the septum with a small hole for limited aeration. Exemplary anaerobic conditions have been described previously and are well-known in the art. Exemplary aerobic and anaerobic conditions are described, for example, in United States Publication No. US-2009-0047719, filed Aug. 10, 2007. Fermentations can be performed in a batch, fed-batch or continuous manner, as disclosed herein.
[0376] If desired, the pH of the medium can be maintained at a desired pH, in particular neutral pH, such as a pH of around 7 by addition of a base, such as NaOH or other bases, or acid, as needed to maintain the culture medium at a desirable pH. The growth rate can be determined by measuring optical density using a spectrophotometer (600 nm), and the glucose uptake rate by monitoring carbon source depletion over time.
[0377] In addition to renewable feedstocks such as those exemplified above, the 1,3-butanediol microbial organisms of the invention also can be modified for growth on syngas as its source of carbon. In this specific embodiment, one or more proteins or enzymes are expressed in the 1,3-butanediol producing organisms to provide a metabolic pathway for utilization of syngas or other gaseous carbon source.
[0378] Organisms of the present invention can utilize, and the growth medium can include, for example, any carbohydrate source which can supply a source of carbon to the non-naturally occurring microorganism. Such sources include, for example, sugars such as glucose, xylose, arabinose, galactose, mannose, fructose and starch. Other sources of carbohydrate include, for example, renewable feedstocks and biomass. Exemplary types of biomasses that can be used as feedstocks in the methods of the invention include cellulosic biomass, hemicellulosic biomass and lignin feedstocks or portions of feedstocks. Such biomass feedstocks contain, for example, carbohydrate substrates useful as carbon sources such as glucose, xylose, arabinose, galactose, mannose, fructose and starch. Given the teachings and guidance provided herein, those skilled in the art will understand that renewable feedstocks and biomass other than those exemplified above also can be used for culturing the microbial organisms of the invention for the production of 1,3-butanediol.
[0379] In addition to renewable feedstocks such as those exemplified above, the 1,3-butanediol microbial organisms of the invention also can be modified for growth on syngas as its source of carbon. In this specific embodiment, one or more proteins or enzymes are expressed in the 1,3-butanediol producing organisms to provide a metabolic pathway for utilization of syngas or other gaseous carbon source.
[0380] Synthesis gas, also known as syngas or producer gas, is the major product of gasification of coal and of carbonaceous materials such as biomass materials, including agricultural crops and residues. Syngas is a mixture primarily of H.sub.2 and CO and can be obtained from the gasification of any organic feedstock, including but not limited to coal, coal oil, natural gas, biomass, and waste organic matter. Gasification is generally carried out under a high fuel to oxygen ratio. Although largely H.sub.2 and CO, syngas can also include CO.sub.2 and other gases in smaller quantities. Thus, synthesis gas provides a cost effective source of gaseous carbon such as CO and, additionally, CO.sub.2.
[0381] The Wood-Ljungdahl pathway catalyzes the conversion of CO and H.sub.2 to acetyl-CoA and other products such as acetate. Organisms capable of utilizing CO and syngas also generally have the capability of utilizing CO.sub.2 and CO.sub.2/H.sub.2 mixtures through the same basic set of enzymes and transformations encompassed by the Wood-Ljungdahl pathway. H.sub.2-dependent conversion of CO.sub.2 to acetate by microorganisms was recognized long before it was revealed that CO also could be used by the same organisms and that the same pathways were involved. Many acetogens have been shown to grow in the presence of CO.sub.2 and produce compounds such as acetate as long as hydrogen is present to supply the necessary reducing equivalents (see for example, Drake, Acetogenesis, pp. 3-60 Chapman and Hall, New York, (1994)). This can be summarized by the following equation:
2CO.sub.2+4H.sub.2+nADP+nPi.fwdarw.CH.sub.3COOH+2H.sub.2O+nATP
[0382] Hence, non-naturally occurring microorganisms possessing the Wood-Ljungdahl pathway can utilize CO.sub.2 and H.sub.2 mixtures as well for the production of acetyl-CoA and other desired products.
[0383] The Wood-Ljungdahl pathway is well known in the art and consists of 12 reactions which can be separated into two branches: (1) methyl branch and (2) carbonyl branch. The methyl branch converts syngas to methyl-tetrahydrofolate (methyl-THF) whereas the carbonyl branch converts methyl-THF to acetyl-CoA. The reactions in the methyl branch are catalyzed in order by the following enzymes or proteins: ferredoxin oxidoreductase, formate dehydrogenase, formyltetrahydrofolate synthetase, methenyltetrahydrofolate cyclodehydratase, methylenetetrahydrofolate dehydrogenase and methylenetetrahydrofolate reductase. The reactions in the carbonyl branch are catalyzed in order by the following enzymes or proteins: methyltetrahydrofolate:corrinoid protein methyltransferase (for example, AcsE), corrinoid iron-sulfur protein, nickel-protein assembly protein (for example, AcsF), ferredoxin, acetyl-CoA synthase, carbon monoxide dehydrogenase and nickel-protein assembly protein (for example, CooC). Following the teachings and guidance provided herein for introducing a sufficient number of encoding nucleic acids to generate a 1,3-butanediol pathway, those skilled in the art will understand that the same engineering design also can be performed with respect to introducing at least the nucleic acids encoding the Wood-Ljungdahl enzymes or proteins absent in the host organism. Therefore, introduction of one or more encoding nucleic acids into the microbial organisms of the invention such that the modified organism contains the complete Wood-Ljungdahl pathway will confer syngas utilization ability.
[0384] Additionally, the reductive (reverse) tricarboxylic acid cycle coupled with carbon monoxide dehydrogenase and/or hydrogenase activities can also be used for the conversion of CO, CO.sub.2 and/or H.sub.2 to acetyl-CoA and other products such as acetate. Organisms capable of fixing carbon via the reductive TCA pathway can utilize one or more of the following enzymes: ATP citrate-lyase, citrate lyase, aconitase, isocitrate dehydrogenase, alpha-ketoglutarate:ferredoxin oxidoreductase, succinyl-CoA synthetase, succinyl-CoA transferase, fumarate reductase, fumarase, malate dehydrogenase, NAD(P)H:ferredoxin oxidoreductase, carbon monoxide dehydrogenase, and hydrogenase. Specifically, the reducing equivalents extracted from CO and/or H.sub.2 by carbon monoxide dehydrogenase and hydrogenase are utilized to fix CO.sub.2 via the reductive TCA cycle into acetyl-CoA or acetate. Acetate can be converted to acetyl-CoA by enzymes such as acetyl-CoA transferase, acetate kinase/phosphotransacetylase, and acetyl-CoA synthetase. Acetyl-CoA can be converted to the 1,3-butanediol precursors, glyceraldehyde-3-phosphate, phosphoenolpyruvate, and pyruvate, by pyruvate:ferredoxin oxidoreductase and the enzymes of gluconeogenesis. Following the teachings and guidance provided herein for introducing a sufficient number of encoding nucleic acids to generate a 1,3-butanediol pathway, those skilled in the art will understand that the same engineering design also can be performed with respect to introducing at least the nucleic acids encoding the reductive TCA pathway enzymes or proteins absent in the host organism. Therefore, introduction of one or more encoding nucleic acids into the microbial organisms of the invention such that the modified organism contains a reductive TCA pathway can confer syngas utilization ability.
[0385] Accordingly, given the teachings and guidance provided herein, those skilled in the art will understand that a non-naturally occurring microbial organism can be produced that secretes the biosynthesized compounds of the invention when grown on a carbon source such as a carbohydrate, syngas, CO and/or CO2. Such compounds include, for example, 1,3-butanediol and any of the intermediate metabolites in the 1,3-butanediol pathway. All that is required is to engineer in one or more of the required enzyme or protein activities to achieve biosynthesis of the desired compound or intermediate including, for example, inclusion of some or all of the 1,3-butanediol biosynthetic pathways. Accordingly, the invention provides a non-naturally occurring microbial organism that produces and/or secretes 1,3-butanediol when grown on a carbohydrate or other carbon source and produces and/or secretes any of the intermediate metabolites shown in the 1,3-butanediol pathway when grown on a carbohydrate or other carbon source. The 1,3-butanediol producing microbial organisms of the invention can initiate synthesis from an intermediate, for example, acetyl-CoA.
[0386] The non-naturally occurring microbial organisms of the invention are constructed using methods well known in the art as exemplified herein to exogenously express at least one nucleic acid encoding a 1,3-butanediol pathway enzyme or protein in sufficient amounts to produce 1,3-butanediol. It is understood that the microbial organisms of the invention are cultured under conditions sufficient to produce 1,3-butanediol. Following the teachings and guidance provided herein, the non-naturally occurring microbial organisms of the invention can achieve biosynthesis of 1,3-butanediol resulting in intracellular concentrations between about 0.1-200 mM or more. Generally, the intracellular concentration of 1,3-butanediol is between about 3-150 mM, particularly between about 5-125 mM and more particularly between about 8-100 mM, including about 10 mM, 20 mM, 50 mM, 80 mM, or more. Intracellular concentrations between and above each of these exemplary ranges also can be achieved from the non-naturally occurring microbial organisms of the invention.
[0387] In some embodiments, culture conditions include anaerobic or substantially anaerobic growth or maintenance conditions. Exemplary anaerobic conditions have been described previously and are well known in the art. Exemplary anaerobic conditions for fermentation processes are described herein and are described, for example, in U.S. publication 2009/0047719, filed Aug. 10, 2007. Any of these conditions can be employed with the non-naturally occurring microbial organisms as well as other anaerobic conditions well known in the art. Under such anaerobic or substantially anaerobic conditions, the 1,3-butanediol producers can synthesize 1,3-butanediol at intracellular concentrations of 5-10 mM or more as well as all other concentrations exemplified herein. It is understood that, even though the above description refers to intracellular concentrations, 1,3-butanediol producing microbial organisms can produce 1,3-butanediol intracellularly and/or secrete the product into the culture medium.
[0388] In addition to the culturing and fermentation conditions disclosed herein, growth condition for achieving biosynthesis of 1,3-butanediol can include the addition of an osmoprotectant to the culturing conditions. In certain embodiments, the non-naturally occurring microbial organisms of the invention can be sustained, cultured or fermented as described herein in the presence of an osmoprotectant. Briefly, an osmoprotectant refers to a compound that acts as an osmolyte and helps a microbial organism as described herein survive osmotic stress. Osmoprotectants include, but are not limited to, betaines, amino acids, and the sugar trehalose. Non-limiting examples of such are glycine betaine, praline betaine, dimethylthetin, dimethylsulfonioproprionate, 3-dimethylsulfonio-2-methylproprionate, pipecolic acid, dimethylsulfonioacetate, choline, L-carnitine and ectoine. In one aspect, the osmoprotectant is glycine betaine. It is understood to one of ordinary skill in the art that the amount and type of osmoprotectant suitable for protecting a microbial organism described herein from osmotic stress will depend on the microbial organism used. The amount of osmoprotectant in the culturing conditions can be, for example, no more than about 0.1 mM, no more than about 0.5 mM, no more than about 1.0 mM, no more than about 1.5 mM, no more than about 2.0 mM, no more than about 2.5 mM, no more than about 3.0 mM, no more than about 5.0 mM, no more than about 7.0 mM, no more than about 10 mM, no more than about 50 mM, no more than about 100 mM or no more than about 500 mM.
[0389] In some embodiments, the carbon feedstock and other cellular uptake sources such as phosphate, ammonia, sulfate, chloride and other halogens can be chosen to alter the isotopic distribution of the atoms present in 1,3-butanediol or any 1,3-butanediol pathway intermediate. The various carbon feedstock and other uptake sources enumerated above will be referred to herein, collectively, as "uptake sources." Uptake sources can provide isotopic enrichment for any atom present in the product 1,3-butanediol or 1,3-butanediol pathway intermediate including any 1,3-butanediol impurities, or for side products generated in reactions diverging away from a 1,3-butanediol pathway. Isotopic enrichment can be achieved for any target atom including, for example, carbon, hydrogen, oxygen, nitrogen, sulfur, phosphorus, chloride or other halogens.
[0390] In some embodiments, the uptake sources can be selected to alter the carbon-12, carbon-13, and carbon-14 ratios. In some embodiments, the uptake sources can be selected to alter the oxygen-16, oxygen-17, and oxygen-18 ratios. In some embodiments, the uptake sources can be selected to alter the hydrogen, deuterium, and tritium ratios. In some embodiments, the uptake sources can be selected to alter the nitrogen-14 and nitrogen-15 ratios. In some embodiments, the uptake sources can be selected to alter the sulfur-32, sulfur-33, sulfur-34, and sulfur-35 ratios. In some embodiments, the uptake sources can be selected to alter the phosphorus-31, phosphorus-32, and phosphorus-33 ratios. In some embodiments, the uptake sources can be selected to alter the chlorine-35, chlorine-36, and chlorine-37 ratios.
[0391] In some embodiments, a target isotopic ratio of an uptake source can be obtained via synthetic chemical enrichment of the uptake source. Such isotopically enriched uptake sources can be purchased commercially or prepared in the laboratory. In some embodiments, a target isotopic ratio of an uptake source can be obtained by choice of origin of the uptake source in nature. In some embodiments, the isotopic ratio of a target atom can be varied to a desired ratio by selecting one or more uptake sources. An uptake source can be derived from a natural source, as found in nature, or from a man-made source, and one skilled in the art can select a natural source, a man-made source, or a combination thereof, to achieve a desired isotopic ratio of a target atom. An example of a man-made uptake source includes, for example, an uptake source that is at least partially derived from a chemical synthetic reaction. Such isotopically enriched uptake sources can be purchased commercially or prepared in the laboratory and/or optionally mixed with a natural source of the uptake source to achieve a desired isotopic ratio. In some embodiments, a target atom isotopic ratio of an uptake source can be achieved by selecting a desired origin of the uptake source as found in nature. For example, as discussed herein, a natural source can be a biobased derived from or synthesized by a biological organism or a source such as petroleum-based products or the atmosphere. In some such embodiments, a source of carbon, for example, can be selected from a fossil fuel-derived carbon source, which can be relatively depleted of carbon-14, or an environmental or atmospheric carbon source, such as CO2, which can possess a larger amount of carbon-14 than its petroleum-derived counterpart.
[0392] The unstable carbon isotope carbon-14 or radiocarbon makes up for roughly 1 in 10.sup.12 carbon atoms in the earth's atmosphere and has a half-life of about 5700 years. The stock of carbon is replenished in the upper atmosphere by a nuclear reaction involving cosmic rays and ordinary nitrogen (.sup.14N). Fossil fuels contain no carbon-14, as it decayed long ago. Burning of fossil fuels lowers the atmospheric carbon-14 fraction, the so-called "Suess effect".
[0393] Methods of determining the isotopic ratios of atoms in a compound are well known to those skilled in the art. Isotopic enrichment is readily assessed by mass spectrometry using techniques known in the art such as accelerated mass spectrometry (AMS), Stable Isotope Ratio Mass Spectrometry (SIRMS) and Site-Specific Natural Isotopic Fractionation by Nuclear Magnetic Resonance (SNIF-NMR). Such mass spectral techniques can be integrated with separation techniques such as liquid chromatography (LC), high performance liquid chromatography (HPLC) and/or gas chromatography, and the like.
[0394] In the case of carbon, ASTM D6866 was developed in the United States as a standardized analytical method for determining the biobased content of solid, liquid, and gaseous samples using radiocarbon dating by the American Society for Testing and Materials (ASTM) International. The standard is based on the use of radiocarbon dating for the determination of a product's biobased content. ASTM D6866 was first published in 2004, and the current active version of the standard is ASTM D6866-11 (effective Apr. 1, 2011). Radiocarbon dating techniques are well known to those skilled in the art, including those described herein.
[0395] The biobased content of a compound is estimated by the ratio of carbon-14 (.sup.14C) to carbon-12 (.sup.12C). Specifically, the Fraction Modern (Fm) is computed from the expression: Fm=(S-B)/(M-B), where B, S and M represent the .sup.14C/.sup.12C ratios of the blank, the sample and the modern reference, respectively. Fraction Modern is a measurement of the deviation of the .sup.14C/.sup.12C ratio of a sample from "Modern." Modern is defined as 95% of the radiocarbon concentration (in AD 1950) of National Bureau of Standards (NBS) Oxalic Acid I (i.e., standard reference materials (SRM) 4990b) normalized to .delta..sup.13C.sub.VPDB=-19 per mil. (Olsson, The use of Oxalic acid as a Standard. in, Radiocarbon Variations and Absolute Chronology, Nobel Symposium, 12th Proc., John Wiley & Sons, New York (1970)). Mass spectrometry results, for example, measured by ASM, are calculated using the internationally agreed upon definition of 0.95 times the specific activity of NBS Oxalic Acid I (SRM 4990b) normalized to .delta..sup.13C.sub.VPDB=-19 per mil. This is equivalent to an absolute (AD 1950) .sup.14C/.sup.12C ratio of 1.176.+-.0.010.times.10.sup.-12 (Karlen et al., Arkiv Geofysik, 4:465-471 (1968)). The standard calculations take into account the differential uptake of one isotope with respect to another, for example, the preferential uptake in biological systems of C.sup.12 over C.sup.13 over C.sup.14, and these corrections are reflected as a Fm corrected for .delta..sup.13.
[0396] An oxalic acid standard (SRM 4990b or HOx 1) was made from a crop of 1955 sugar beet. Although there were 1000 lbs made, this oxalic acid standard is no longer commercially available. The Oxalic Acid II standard (HOx 2; N.I.S.T designation SRM 4990 C) was made from a crop of 1977 French beet molasses. In the early 1980's, a group of 12 laboratories measured the ratios of the two standards. The ratio of the activity of Oxalic acid II to 1 is 1.2933.+-.0.001 (the weighted mean). The isotopic ratio of HOx II is -17.8 per mille. ASTM D6866-11 suggests use of the available Oxalic Acid II standard SRM 4990 C (Hox2) for the modern standard (see discussion of original vs. currently available oxalic acid standards in Mann, Radiocarbon, 25(2):519-527 (1983)). A Fm=0% represents the entire lack of carbon-14 atoms in a material, thus indicating a fossil (for example, petroleum based) carbon source. A Fm=100%, after correction for the post-1950 injection of carbon-14 into the atmosphere from nuclear bomb testing, indicates an entirely modern carbon source. As described herein, such a "modern" source includes biobased sources.
[0397] As described in ASTM D6866, the percent modern carbon (pMC) can be greater than 100% because of the continuing but diminishing effects of the 1950s nuclear testing programs, which resulted in a considerable enrichment of carbon-14 in the atmosphere as described in ASTM D6866-11. Because all sample carbon-14 activities are referenced to a "pre-bomb" standard, and because nearly all new biobased products are produced in a post-bomb environment, all pMC values (after correction for isotopic fraction) must be multiplied by 0.95 (as of 2010) to better reflect the true biobased content of the sample. A biobased content that is greater than 103% suggests that either an analytical error has occurred, or that the source of biobased carbon is more than several years old.
[0398] ASTM D6866 quantifies the biobased content relative to the material's total organic content and does not consider the inorganic carbon and other non-carbon containing substances present. For example, a product that is 50% starch-based material and 50% water would be considered to have a Biobased Content=100% (50% organic content that is 100% biobased) based on ASTM D6866. In another example, a product that is 50% starch-based material, 25% petroleum-based, and 25% water would have a Biobased Content=66.7% (75% organic content but only 50% of the product is biobased). In another example, a product that is 50% organic carbon and is a petroleum-based product would be considered to have a Biobased Content=0% (50% organic carbon but from fossil sources). Thus, based on the well known methods and known standards for determining the biobased content of a compound or material, one skilled in the art can readily determine the biobased content and/or prepared downstream products that utilize of the invention having a desired biobased content.
[0399] Applications of carbon-14 dating techniques to quantify bio-based content of materials are known in the art (Currie et al., Nuclear Instruments and Methods in Physics Research B, 172:281-287 (2000)). For example, carbon-14 dating has been used to quantify bio-based content in terephthalate-containing materials (Colonna et al., Green Chemistry, 13:2543-2548 (2011)). Notably, polypropylene terephthalate (PPT) polymers derived from renewable 1,3-propanediol and petroleum-derived terephthalic acid resulted in Fm values near 30% (i.e., since 3/11 of the polymeric carbon derives from renewable 1,3-propanediol and 8/11 from the fossil end member terephthalic acid) (Currie et al., supra, 2000). In contrast, polybutylene terephthalate polymer derived from both renewable 1,4-butanediol and renewable terephthalic acid resulted in bio-based content exceeding 90% (Colonna et al., supra, 2011).
[0400] Accordingly, in some embodiments, the present invention provides 1,3-butanediol or a 1,3-butanediol intermediate that has a carbon-12, carbon-13, and carbon-14 ratio that reflects an atmospheric carbon, also referred to as environmental carbon, uptake source. For example, in some aspects the 1,3-butanediol or a 1,3-butanediol intermediate can have an Fm value of at least 10%, at least 15%, at least 20%, at least 25%, at least 30%, at least 35%, at least 40%, at least 45%, at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 98% or as much as 100%. In some such embodiments, the uptake source is CO.sub.2. In some embodiments, the present invention provides 1,3-butanediol or a 1,3-butanediol intermediate that has a carbon-12, carbon-13, and carbon-14 ratio that reflects petroleum-based carbon uptake source. In this aspect, the 1,3-butanediol or a 1,3-butanediol intermediate can have an Fm value of less than 95%, less than 90%, less than 85%, less than 80%, less than 75%, less than 70%, less than 65%, less than 60%, less than 55%, less than 50%, less than 45%, less than 40%, less than 35%, less than 30%, less than 25%, less than 20%, less than 15%, less than 10%, less than 5%, less than 2% or less than 1%. In some embodiments, the present invention provides 1,3-butanediol or a 1,3-butanediol intermediate that has a carbon-12, carbon-13, and carbon-14 ratio that is obtained by a combination of an atmospheric carbon uptake source with a petroleum-based uptake source. Using such a combination of uptake sources is one way by which the carbon-12, carbon-13, and carbon-14 ratio can be varied, and the respective ratios would reflect the proportions of the uptake sources.
[0401] Further, the present invention relates to the biologically produced 1,3-butanediol or 1,3-butanediol intermediate as disclosed herein, and to the products derived therefrom, wherein the 1,3-butanediol or a 1,3-butanediol intermediate has a carbon-12, carbon-13, and carbon-14 isotope ratio of about the same value as the CO.sub.2 that occurs in the environment. For example, in some aspects the invention provides: bioderived 1,3-butanediol or a bioderived 1,3-butanediol intermediate having a carbon-12 versus carbon-13 versus carbon-14 isotope ratio of about the same value as the CO.sub.2 that occurs in the environment, or any of the other ratios disclosed herein. It is understood, as disclosed herein, that a product can have a carbon-12 versus carbon-13 versus carbon-14 isotope ratio of about the same value as the CO.sub.2 that occurs in the environment, or any of the ratios disclosed herein, wherein the product is generated from bioderived 1,3-butanediol or a bioderived 1,3-butanediol intermediate as disclosed herein, wherein the bioderived product is chemically modified to generate a final product. Methods of chemically modifying a bioderived product of 1,3-butanediol, or an intermediate thereof, to generate a desired product are well known to those skilled in the art, as described herein. The invention further provides organic solvents, polyurethane resins, polyester resins, hypoglycaemic agents, butadiene and/or butadiene-based products having a carbon-12 versus carbon-13 versus carbon-14 isotope ratio of about the same value as the CO.sub.2 that occurs in the environment, wherein the organic solvents, polyurethane resins, polyester resins, hypoglycaemic agents, butadiene and/or butadiene-based products are generated directly from or in combination with bioderived 1,3-butanediol or a bioderived 1,3-butanediol intermediate as disclosed herein.
[0402] 1,3-butanediol is a chemical used in commercial and industrial applications and is also used as a raw material in the production of a wide range of products. Non-limiting examples of such applications and products include organic solvents, polyurethane resins, polyester resins, hypoglycaemic agents, butadiene and/or butadiene-based products organic solvents, polyurethane resins, polyester resins, hypoglycaemic agents, butadiene and/or butadiene-based products. Accordingly, in some embodiments, the invention provides biobased used as a raw material in the production of a wide range of products comprising one or more bioderived 1,3-butanediol or bioderived 1,3-butanediol intermediate produced by a non-naturally occurring microorganism of the invention or produced using a method disclosed herein.
[0403] As used herein, the term "bioderived" means derived from or synthesized by a biological organism and can be considered a renewable resource since it can be generated by a biological organism. Such a biological organism, in particular the microbial organisms of the invention disclosed herein, can utilize feedstock or biomass, such as, sugars or carbohydrates obtained from an agricultural, plant, bacterial, or animal source. Alternatively, the biological organism can utilize atmospheric carbon. As used herein, the term "biobased" means a product as described above that is composed, in whole or in part, of a bioderived compound of the invention. A biobased or bioderived product is in contrast to a petroleum derived product, wherein such a product is derived from or synthesized from petroleum or a petrochemical feedstock.
[0404] In some embodiments, the invention provides organic solvents, polyurethane resins, polyester resins, hypoglycaemic agents, butadiene and/or butadiene-based products comprising bioderived 1,3-butanediol or bioderived 1,3-butanediol intermediate, wherein the bioderived 1,3-butanediol or bioderived 1,3-butanediol intermediate includes all or part of the 1,3-butanediol or 1,3-butanediol intermediate used in the production of organic solvents, polyurethane resins, polyester resins, hypoglycaemic agents, butadiene and/or butadiene-based products. Thus, in some aspects, the invention provides biobased organic solvents, polyurethane resins, polyester resins, hypoglycaemic agents, butadiene and/or butadiene-based products comprising at least 2%, at least 3%, at least 5%, at least 10%, at least 15%, at least 20%, at least 25%, at least 30%, at least 35%, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, at least 95%, at least 98% or 100% bioderived 1,3-butanediol or bioderived 1,3-butanediol intermediate as disclosed herein. Additionally, in some aspects, the invention provides biobased organic solvents, polyurethane resins, polyester resins, hypoglycaemic agents, butadiene and/or butadiene-based products wherein the 1,3-butanediol or 1,3-butanediol intermediate used in its production is a combination of bioderived and petroleum derived 1,3-butanediol or 1,3-butanediol intermediate. For example, biobased organic solvents, polyurethane resins, polyester resins, hypoglycaemic agents, butadiene and/or butadiene-based products can be produced using 50% bioderived 1,3-butanediol and 50% petroleum derived 1,3-butanediol or other desired ratios such as 60%/40%, 70%/30%, 80%/20%, 90%/10%, 95%/5%, 100%/0%, 40%/60%, 30%/70%, 20%/80%, 10%/90% of bioderived/petroleum derived precursors, so long as at least a portion of the product comprises a bioderived product produced by the microbial organisms disclosed herein. It is understood that methods for producing organic solvents, polyurethane resins, polyester resins, hypoglycaemic agents, butadiene and/or butadiene-based products using the bioderived 1,3-butanediol or bioderived 1,3-butanediol intermediate of the invention are well known in the art.
[0405] The non-naturally occurring microbial organisms of the invention are constructed using methods well known in the art as exemplified herein to exogenously express at least one nucleic acid encoding a 1,3-butanediol pathway enzyme or protein in sufficient amounts to produce 1,3-butanediol. It is understood that the microbial organisms of the invention are cultured under conditions sufficient to produce 1,3-butanediol. Following the teachings and guidance provided herein, the non-naturally occurring microbial organisms of the invention can achieve biosynthesis of 1,3-butanediol resulting in intracellular concentrations between about 0.1-2000 mM or more. Generally, the intracellular concentration of 1,3-butanediol is between about 3-1800 mM, particularly between about 5-1700 mM and more particularly between about 8-1600 mM, including about 100 mM, 200 mM, 500 mM, 800 mM, or more. Intracellular concentrations between and above each of these exemplary ranges also can be achieved from the non-naturally occurring microbial organisms of the invention.
[0406] In some embodiments, culture conditions include anaerobic or substantially anaerobic growth or maintenance conditions. Exemplary anaerobic conditions have been described previously and are well known in the art. Exemplary anaerobic conditions for fermentation processes are described herein and are described, for example, in U.S. patent application No. US 2009/0047719, filed Aug. 10, 2007. Any of these conditions can be employed with the non-naturally occurring microbial organisms as well as other anaerobic conditions well known in the art. Under such anaerobic conditions, the 1,3-butanediol producers can synthesize 1,3-butanediol at intracellular concentrations of 5-10 mM or more as well as all other concentrations exemplified herein. It is understood that, even though the above description refers to intracellular concentrations, 1,3-butanediol producing microbial organisms can produce 1,3-butanediol intracellularly and/or secrete the product into the culture medium.
[0407] In addition to the culturing and fermentation conditions disclosed herein, growth condition for achieving biosynthesis of 1,3-butanediol can include the addition of an osmoprotectant to the culturing conditions. In certain embodiments, the non-naturally occurring microbial organisms of the invention can be sustained, cultured or fermented as described herein in the presence of an osmoprotectant. Briefly, an osmoprotectant refers to a compound that acts as an osmolyte and helps a microbial organism as described herein survive osmotic stress. Osmoprotectants include, but are not limited to, betaines, amino acids, and the sugar trehalose. Non-limiting examples of such are glycine betaine, praline betaine, dimethylthetin, dimethylsulfonioproprionate, 3-dimethylsulfonio-2-methylproprionate, pipecolic acid, dimethylsulfonioacetate, choline, L-carnitine and ectoine. In one aspect, the osmoprotectant is glycine betaine. It is understood to one of ordinary skill in the art that the amount and type of osmoprotectant suitable for protecting a microbial organism described herein from osmotic stress will depend on the microbial organism used. The amount of osmoprotectant in the culturing conditions can be, for example, no more than about 0.1 mM, no more than about 0.5 mM, no more than about 1.0 mM, no more than about 1.5 mM, no more than about 2.0 mM, no more than about 2.5 mM, no more than about 3.0 mM, no more than about 5.0 mM, no more than about 7.0 mM, no more than about 10 mM, no more than about 50 mM, no more than about 100 mM or no more than about 500 mM.
[0408] The culture conditions can include, for example, liquid culture procedures as well as fermentation and other large scale culture procedures. As described herein, particularly useful yields of the biosynthetic products of the invention can be obtained under anaerobic or substantially anaerobic culture conditions.
[0409] As described herein, one exemplary growth condition for achieving biosynthesis of 1,3-butanediol includes anaerobic culture or fermentation conditions. In certain embodiments, the non-naturally occurring microbial organisms of the invention can be sustained, cultured or fermented under anaerobic or substantially anaerobic conditions. Briefly, anaerobic conditions refers to an environment devoid of oxygen. Substantially anaerobic conditions include, for example, a culture, batch fermentation or continuous fermentation such that the dissolved oxygen concentration in the medium remains between 0 and 10% of saturation. Substantially anaerobic conditions also includes growing or resting cells in liquid medium or on solid agar inside a sealed chamber maintained with an atmosphere of less than 1% oxygen. The percent of oxygen can be maintained by, for example, sparging the culture with an N2/CO2 mixture or other suitable non-oxygen gas or gases.
[0410] The culture conditions described herein can be scaled up and grown continuously for manufacturing of 1,3-butanediol. Exemplary growth procedures include, for example, fed-batch fermentation and batch separation; fed-batch fermentation and continuous separation, or continuous fermentation and continuous separation. All of these processes are well known in the art. Fermentation procedures are particularly useful for the biosynthetic production of commercial quantities of 1,3-butanediol. Generally, and as with non-continuous culture procedures, the continuous and/or near-continuous production of 1,3-butanediol will include culturing a non-naturally occurring 1,3-butanediol producing organism of the invention in sufficient nutrients and medium to sustain and/or nearly sustain growth in an exponential phase. Continuous culture under such conditions can include, for example, growth for 1 day, 2, 3, 4, 5, 6 or 7 days or more. Additionally, continuous culture can include longer time period of 1 week, 2, 3, 4 or 5 or more weeks and up to several months. Alternatively, organisms of the invention can be cultured for hours, if suitable for a particular application. It is to be understood that the continuous and/or near-continuous culture conditions also can include all time intervals in between these exemplary periods. It is further understood that the time of culturing the microbial organism of the invention is for a sufficient period of time to produce a sufficient amount of product for a desired purpose.
[0411] Fermentation procedures are well known in the art. Briefly, fermentation for the biosynthetic production of 1,3-butanediol can be utilized in, for example, fed-batch fermentation and batch separation; fed-batch fermentation and continuous separation, or continuous fermentation and continuous separation. Examples of batch and continuous fermentation procedures are well known in the art.
[0412] In addition to the above fermentation procedures using the 1,3-butanediol producers of the invention for continuous production of substantial quantities of 1,3-butanediol, the 1,3-butanediol producers also can be, for example, simultaneously subjected to chemical synthesis procedures to convert the product to other compounds or the product can be separated from the fermentation culture and sequentially subjected to chemical or enzymatic conversion to convert the product to other compounds, if desired. For example, 1,3-butanediol can be dehydrated to provide 1,3-butadiene.
[0413] To generate better producers, metabolic modeling can be utilized to optimize growth conditions. Modeling can also be used to design gene knockouts that additionally optimize utilization of the pathway (see, for example, U.S. patent publications US 2002/0012939, US 2003/0224363, US 2004/0029149, US 2004/0072723, US 2003/0059792, US 2002/0168654 and US 2004/0009466, and U.S. Pat. No. 7,127,379). Modeling analysis allows reliable predictions of the effects on cell growth of shifting the metabolism towards more efficient production of 1,3-butadiene.
[0414] In some embodiments, syngas can be used as a carbon feedstock. Important process considerations for a syngas fermentation are high biomass concentration and good gas-liquid mass transfer (Bredwell et al., Biotechnol Prog., 15:834-844 (1999). The solubility of CO in water is somewhat less than that of oxygen. Continuously gas-sparged fermentations can be performed in controlled fermenters with constant off-gas analysis by mass spectrometry and periodic liquid sampling and analysis by GC and HPLC. The liquid phase can function in batch mode. Fermentation products such as alcohols, organic acids, and residual glucose along with residual methanol are quantified by HPLC (Shimadzu, Columbia Md.), for example, using an Aminex.RTM. series of HPLC columns (for example, HPX-87 series) (BioRad, Hercules Calif.), using a refractive index detector for glucose and alcohols, and a UV detector for organic acids. The growth rate is determined by measuring optical density using a spectrophotometer (600 nm). All piping in these systems is glass or metal to maintain anaerobic conditions. The gas sparging is performed with glass fits to decrease bubble size and improve mass transfer. Various sparging rates are tested, ranging from about 0.1 to 1 vvm (vapor volumes per minute). To obtain accurate measurements of gas uptake rates, periodic challenges are performed in which the gas flow is temporarily stopped, and the gas phase composition is monitored as a function of time.
[0415] In order to achieve the overall target productivity, methods of cell retention or recycle are employed. One method to increase the microbial concentration is to recycle cells via a tangential flow membrane from a sidestream. Repeated batch culture can also be used, as previously described for production of acetate by Moorella (Sakai et al., J Biosci. Bioeng, 99:252-258 (2005)). Various other methods can also be used (Bredwell et al., Biotechnol Prog., 15:834-844 (1999); Datar et al., Biotechnol Bioeng, 86:587-594 (2004)). Additional optimization can be tested such as overpressure at 1.5 atm to improve mass transfer (Najafpour et al., Enzyme and Microbial Technology, 38[1-2], 223-228 (2006)).
[0416] Once satisfactory performance is achieved using pure H2/CO as the feed, synthetic gas mixtures are generated containing inhibitors likely to be present in commercial syngas. For example, a typical impurity profile is 4.5% CH4, 0.1% C2H2, 0.35% C2H6, 1.4% C2H4, and 150 ppm nitric oxide (Datar et al., Biotechnol Bioeng, 86:587-594 (2004)). Tars, represented by compounds such as benzene, toluene, ethylbenzene, p-xylene, o-xylene, and naphthalene, are added at ppm levels to test for any effect on production. For example, it has been shown that 40 ppm NO is inhibitory to C. carboxidivorans (Ahmed et al., Biotechnol Bioeng, 97:1080-1086 (2007)). Cultures are tested in shake-flask cultures before moving to a fermentor. Also, different levels of these potential inhibitory compounds are tested to quantify the effect they have on cell growth. This knowledge is used to develop specifications for syngas purity, which is utilized for scale up studies and production. If any particular component is found to be difficult to decrease or remove from syngas used for scale up, an adaptive evolution procedure is utilized to adapt cells to tolerate one or more impurities.
[0417] Advances in the field of protein engineering make it feasible to alter any of the enzymes disclosed herein to act efficiently on substrates not known to be natural to them. Below are several examples of broad-specificity enzymes from diverse classes of interest and methods that have been used for evolving such enzymes to act on non-natural substrates.
[0418] One class of enzymes in the pathways disclosed herein is the oxidoreductases that interconvert ketones or aldehydes to alcohols (1.1.1). Enzymes in this class that can operate on a wide range of substrates. An alcohol dehydrogenase (1.1.1.1) purified from the soil bacterium Brevibacterium sp KU 1309 (Hirano et al., J. Biosci. Bioeng. 100:318-322 (2005)) was shown to operate on a plethora of aliphatic as well as aromatic alcohols with high activities. Table 72 shows the activity of the enzyme and its Km on different alcohols. The enzyme is reversible and has very high activity on several aldehydes also as shown in Table 73.
TABLE-US-00074 TABLE 72 RELATIVE ACTIVITY K.sub.M SUBSTRATE (%) (MM) 2-Phenylethanol 100 0.025 (S)-2-Phenylpropanol 156 0.157 (R)-2-Phenylpropanol 63 0.020 Benzyl alcohol 199 0.012 3-Phenylpropanol 135 0.033 Ethanol 76 1-Butanol 111 1-Octanol 101 1-Dodecanol 68 1-Phenylethanol 46 2-Propanol 54
[0419] In this Table, the activity of 2-phenylethanol, corresponding to 19.2 U/mg, was taken as 100%.
TABLE-US-00075 TABLE 73 RELATIVE ACTIVITY .kappa..sub.M SUBSTRATE (%) (MM) Phenylacetaldehyde 100 0.261 2-Phenylpropionaldehyde 188 0.864 1-Octylaldehyde 87 Acetophenone 0
[0420] Lactate dehydrogenase (1.1.1.27) from Ralstonia eutropha is another enzyme that has been demonstrated to have high activities on several 2-oxoacids such as 2-oxobutyrate, 2-oxopentanoate and 2-oxoglutarate (a C5 compound analogous to 2-oxoadipate) (Steinbuchel et al., supra). Column 2 in Table 74 demonstrates the activities of ldhA from R. eutropha (formerly A. eutrophus) on different substrates (Steinbuchel et al., supra).
TABLE-US-00076 TABLE 74 Activity of L(+)- L(+)- D(-)- lactate lactate dehydro- lactate dehydro-genase genase from dehydro-genase from rabbit from Substrate A. eustrophus % muscle L. leischmanii Glyoxylate 8.7 23.9 5.0 Pyruvate 100.0 100.0 100.0 2-Oxobutyrate 107.0 18.6 1.1 2-Oxovalerate 125.0 0.7 0.0 3-Methyl-2- 28.5 0.0 0.0 oxobutyrate 3-Methyl-2- 5.3 0.0 0.0 oxovalerate 4-Methyl-2- 39.0 1.4 1.1 oxopentanoate Oxaloacetate 0.0 33.1 23.1 2-Oxoglutarate 79.6 0.0 0.0 3-Fluoropyruvate 33.6 74.3 40.0
[0421] Oxidoreductases that can convert 2-oxoacids to their acyl-CoA counterparts (1.2.1) have been shown to accept multiple substrates as well. For example, branched-chain 2-keto-acid dehydrogenase complex (BCKAD), also known as 2-oxoisovalerate dehydrogenase (1.2.1.25), participates in branched-chain amino acid degradation pathways, converting 2-keto acids derivatives of valine, leucine and isoleucine to their acyl-CoA derivatives and CO.sub.2. In some organisms including Rattus norvegicus (Paxton et al., Biochem. J. 234:295-303 (1986)) and Saccharomyces cerevisiae (Sinclair et al., Biochem. Mol. Biol. Int. 31:911-922 (1993)), this complex has been shown to have a broad substrate range that includes linear oxo-acids such as 2-oxobutanoate and alpha-ketoglutarate, in addition to the branched-chain amino acid precursors.
[0422] Members of yet another class of enzymes, namely aminotransferases (2.6.1), have been reported to act on multiple substrates. Aspartate aminotransferase (aspAT) from Pyrococcus fursious has been identified, expressed in E. coli and the recombinant protein characterized to demonstrate that the enzyme has the highest activities towards aspartate and alpha-ketoglutarate but lower, yet significant activities towards alanine, glutamate and the aromatic amino acids (Ward et al., Archaea. 1:133-141 (2002)). In another instance, an aminotransferase identified from Leishmania mexicana and expressed in E. coli Vernal et al., FEMS Microbiol. Lett. 229:217-222 (2003)) was reported to have a broad substrate specificity towards tyrosine (activity considered 100% on tyrosine), phenylalanine (90%), tryptophan (85%), aspartate (30%), leucine (25%) and methionine (25%) respectively (Vernal et al., Mol. Biochem. Parasitoi. 96:83-92 (1998)). Similar broad specificity has been reported for a tyrosine aminotransferase from Trypanosoma cruzi, even though both of these enzymes have a sequence homology of only 6%. Note that the latter enzyme can accept leucine, methionine as well as tyrosine, phenylalanine, tryptophan and alanine as efficient amino donors (Nowicki et al., Biochim. Biophys. Acta 1546: 268-281 (2001)).
[0423] In contrast to these examples where the enzymes naturally have broad substrate specificities, numerous enzymes have been modified using directed evolution to broaden their specificity towards their non-natural substrates. Alternatively, the substrate preference of an enzyme has also been changed using directed evolution. For example, it has been reported that the enantioselectivity of a lipase from Pseudomonas aeruginosa was improved significantly. This enzyme hydrolyzed p-nitrophenyl 2-methyldecanoate with only 2% enantiomeric excess (ee) in favor of the (S)-acid. However, after four successive rounds of error-prone mutagenesis and screening, a variant was produced that catalyzed the requisite reaction with 81% ee Reetz et al., Angew. Chem. Int. Ed Engl. 36:2830-2832 (1997)).
[0424] Directed evolution methods have made possible the modification of an enzyme to function on an array of unnatural substrates. The substrate specificity of the lipase in P. aeruginosa was broadened by randomization of amino acid residues near the active site. This allowed for the acceptance of alpha-substituted carboxylic acid esters by this enzyme Reetz et al., Angew. Chem. Int. Ed Engl. 44:4192-4196 (2005)). In another successful attempt, DNA shuffling was employed to create an Escherichia coli aminotransferase that accepted .beta.-branched substrates, which were poorly accepted by the wild-type enzyme (Yano et al., Proc. Natl. Acad. Sci. U.S.A 95:5511-5515 (1998)). Specifically, at the end of four rounds of shuffling, the activity of aspartate aminotransferase for valine and 2-oxovaline increased by up to five orders of magnitude, while decreasing the activity towards the natural substrate, aspartate, by up to 30-fold. Recently, an algorithm was used to design a retro-aldolase that could be used to catalyze the carbon-carbon bond cleavage in a non-natural and non-biological substrate, 4-hydroxy-4-(6-methoxy-2-naphthyl)-2-butanone. These algorithms used different combinations of four different catalytic motifs to design new enzymes and 20 of the selected designs for experimental characterization had four-fold improved rates over the uncatalyzed reaction (Jiang et al., Science 319:1387-1391 (2008)). Thus, not only are these engineering approaches capable of expanding the array of substrates on which an enzyme can act, but allow the design and construction of very efficient enzymes. For example, a method of DNA shuffling (random chimeragenesis on transient templates or RACHITT) was reported to lead to an engineered monooxygenase that had an improved rate of desulfurization on complex substrates as well as 20-fold faster conversion of a non-natural substrate (Coco et al. Nat. Biotechnol. 19:354-359 (2001)). Similarly, the specific activity of a sluggish mutant triosephosphate isomerase enzyme was improved up to 19-fold from 1.3 fold (Hermes et al., Proc. Natl. Acad. Sci. U.S.A 87:696-700 (1990)). This enhancement in specific activity was accomplished by using random mutagenesis over the whole length of the protein and the improvement could be traced back to mutations in six amino acid residues.
[0425] The effectiveness of protein engineering approaches to alter the substrate specificity of an enzyme for a desired substrate has also been demonstrated. Isopropylmalate dehydrogenase from Thermus thermophilus was modified by changing residues close to the active site so that it could now act on malate and D-lactate as substrates (Fujita et al., Biosci. Biotechnol Biochem. 65:2695-2700 (2001)). In this study as well as in others, it was pointed out that one or a few residues could be modified to alter the substrate specificity. A case in point is the dihydroflavonol 4-reductase for which a single amino acid was changed in the presumed substrate-binding region that could preferentially reduce dihydrokaempferol Johnson et al., Plant J. 25:325-333 (2001)). The substrate specificity of a very specific isocitrate dehydrogenase from Escherichia coli was changed from isocitrate to isopropylmalate by changing one residue in the active site (Doyle et al., Biochemistry 40:4234-4241 (2001)). In a similar vein, the cofactor specificity of a NAD+-dependent 1,5-hydroxyprostaglandin dehydrogenase was altered to NADP+ by changing a few residues near the N-terminal end Cho et al., Arch. Biochem. Biophys. 419:139-146 (2003)). Sequence analysis and molecular modeling analysis were used to identify the key residues for modification, which were further studied by site-directed mutagenesis.
[0426] A fucosidase was evolved from a galactosidase in E. coli by DNA shuffling and screening (Zhang et al., Proc Natl Acad Sci US.A 94:4504-4509 (1997)). Similarly, aspartate aminotransferase from E. coli was converted into a tyrosine aminotransferase using homology modeling and site-directed mutagenesis (Onuffer et al., Protein Sci. 4:1750-1757 (1995)). Site-directed mutagenesis of two residues in the active site of benzoylformate decarboxylase from P. putida reportedly altered the affinity (Km) towards natural and non-natural substrates Siegert et al., Protein Eng Des Sel 18:345-357 (2005)). Cytochrome c peroxidase (CCP) from Saccharomyces cerevisiae was subjected to directed molecular evolution to generate mutants with increased activity against the classical peroxidase substrate guaiacol, thus changing the substrate specificity of CCP from the protein cytochrome c to a small organic molecule. After three rounds of DNA shuffling and screening, mutants were isolated which possessed a 300-fold increased activity against guaiacol and up to 1000-fold increased specificity for this substrate relative to that for the natural substrate (Iffland et al., Biochemistry 39:10790-10798 (2000)).
[0427] In some cases, enzymes with different substrate preferences than both the parent enzymes have been obtained. For example, biphenyl-dioxygenase-mediated degradation of polychlorinated biphenyls was improved by shuffling genes from two bacteria, Pseudomonas pseudoalcaligens and Burkholderia cepacia (Kumamaru et al., Nat. Biotechnol 16, 663-666 (1998)). The resulting chimeric biphenyl oxygenases showed different substrate preferences than both the parental enzymes and enhanced the degradation activity towards related biphenyl compounds and single aromatic ring hydrocarbons such as toluene and benzene which were originally poor substrates for the enzyme.
[0428] It is not only possible to change the enzyme specificity but also to enhance the activities on those substrates on which the enzymes naturally have low activities. One study demonstrated that amino acid racemase from P. putida that had broad substrate specificity (on lysine, arginine, alanine, serine, methionine, cysteine, leucine and histidine among others) but low activity towards tryptophan could be improved significantly by random mutagenesis Kino et al., Appl. Microbiol. Biotechnol. 73:1299-1305 (2007)). Similarly, the active site of the bovine BCKAD was engineered to favor alternate substrate acetyl-CoA (Meng et al. Biochemistry 33:12879-12885 (1994)). An interesting aspect of these approaches is that even when random methods have been applied to generate these mutated enzymes with efficacious activities, the exact mutations or structural changes that confer the improvement in activity can be identified. For example, in the aforementioned study, the mutations that facilitated improved activity on tryptophan could be traced back to two different positions.
[0429] Directed evolution has also been used to express proteins that are difficult to express. For example, by subjecting the horseradish peroxidase to random mutagenesis and gene recombination, mutants could be extracted that had more than 14-fold activity than the wild type (Lin et al., Biotechnol. Frog. 15:467-471 (1999)).
[0430] A final example of directed evolution shows the extensive modifications to which an enzyme can be subjected to achieve a range of desired functions. The enzyme, lactate dehydrogenase from Bacillus stearothermophilus was subjected to site-directed mutagenesis, and three amino acid substitutions were made at sites that were indicated to determine the specificity towards different hydroxyacids (Clarke et al., Biochem. Biophys. Res. Commun. 148:15-23 (1987)). After these mutations, the specificity for oxaloacetate over pyruvate was increased to 500 in contrast to the wild type enzyme that had a catalytic specificity for pyruvate over oxaloacetate of 1000. This enzyme was further engineered using site-directed mutagenesis to have activity towards branched-chain substituted pyruvates (Wilks et al., Biochemistry 29:8587-8591 (1990)). Specifically, the enzyme had a 55-fold improvement in Kcat for alpha-ketoisocaproate. Three structural modifications were made in the same enzyme to change its substrate specificity from lactate to malate. The enzyme was highly active and specific towards malate (Wilks et al., Science 242:1541-1544 (1988)). The same enzyme from B. stearothermophilus was subsequently engineered to have high catalytic activity towards alpha-keto acids with positively charged side chains, such as those containing ammonium groups (Hogan et al., Biochemistry 34:4225-4230 (1995)). Mutants with acidic amino acids introduced at position 102 of the enzyme favored binding of such side chain ammonium groups. The results obtained proved that the mutants showed up to 25-fold improvements in kcat/Km values for omega-amino-alpha-keto acid substrates. This enzyme was also structurally modified to function as a phenyllactate dehydrogenase instead of a lactate dehydrogenase (Wilks et al., Biochemistry 31:7802-7806 (1992)). Restriction sites were introduced into the gene for the enzyme which allowed a region of the gene to be excised. This region coded for a mobile surface loop of polypeptide (residues 98-110) which normally seals the active site vacuole from bulk solvent and is a major determinant of substrate specificity. The variable length and sequence loops were inserted into the cut gene and used to synthesize hydroxyacid dehydrogenases with altered substrate specificities. With one longer loop construction, activity with pyruvate was reduced one-million-fold but activity with phenylpyruvate was largely unaltered. A switch in specificity (kcat/Km) of 390,000-fold was achieved. The 1700:1 selectivity of this enzyme for phenylpyruvate over pyruvate is that required in a phenyllactate dehydrogenase.
[0431] As indicated above, directed evolution is a powerful approach that involves the introduction of mutations targeted to a specific gene in order to improve and/or alter the properties of an enzyme. Improved and/or altered enzymes can be identified through the development and implementation of sensitive high-throughput screening assays that allow the automated screening of many enzyme variants (e.g., >104). Iterative rounds of mutagenesis and screening typically are performed to afford an enzyme with optimized properties. Computational algorithms that can help to identify areas of the gene for mutagenesis also have been developed and can significantly reduce the number of enzyme variants that need to be generated and screened.
[0432] Numerous directed evolution technologies have been developed (for reviews, see Hibbert, E. G., F. Baganz, H. C. Hailes, J. M. Ward, G. J. Lye, J. M. Woodley, and P. A. Dalby, 2005, Directed evolution of biocatalytic processes. Biomol. Eng 22:11-19; Huisman, G. W. and J. J. Lalonde, 2007, Enzyme evolution for chemical process applications, p. 717-742. In R. N. Patel (ed.), Biocatalysis in the pharmaceutical and biotechnology industries. CRC Press; Otten, L. G. and W. J. Quax. 2005. Directed evolution: selecting today's biocatalysts. Biomol. Eng 22:1-9; and Sen, S., D. Venkata, V, and B. Mandal, 2007, Developments in directed evolution for improving enzyme functions. Appl Biochem. Biotechnol 143:212-223) to be effective at creating diverse variant libraries and these methods have been successfully applied to the improvement of a wide range of properties across many enzyme classes.
[0433] Enzyme characteristics that have been improved and/or altered by directed evolution technologies include, for example, selectivity/specificity--for conversion of non-natural substrates; temperature stability--for robust high temperature processing; pH stability--for bioprocessing under lower or higher pH conditions; substrate or product tolerance--so that high product titers can be achieved; binding (Km)--broadens substrate binding to include non-natural substrates; inhibition (Ki)--to remove inhibition by products, substrates, or key intermediates; activity (kcat)--increases enzymatic reaction rates to achieve desired flux; expression levels--increases protein yields and overall pathway flux; oxygen stability--for operation of air sensitive enzymes under aerobic conditions; and anaerobic activity--for operation of an aerobic enzyme in the absence of oxygen.
[0434] The following exemplary methods have been developed for the mutagenesis and diversification of genes to target desired properties of specific enzymes. Any of these can be used to alter/optimize activity of a decarboxylase enzyme.
[0435] EpPCR (Pritchard, L., D. Come, D. Kell, J. Rowland, and M. Winson, 2005, A general model of error-prone PCR. J Theor. Biol 234:497-509) introduces random point mutations by reducing the fidelity of DNA polymerase in PCR reactions by the addition of Mn2+ ions, by biasing dNTP concentrations, or by other conditional variations. The five step cloning process to confine the mutagenesis to the target gene of interest involves: 1) error-prone PCR amplification of the gene of interest; 2) restriction enzyme digestion; 3) gel purification of the desired DNA fragment; 4) ligation into a vector; 5) transformation of the gene variants into a suitable host and screening of the library for improved performance. This method can generate multiple mutations in a single gene simultaneously, which can be useful. A high number of mutants can be generated by EpPCR, so a high-throughput screening assay or a selection method (especially using robotics) is useful to identify those with desirable characteristics.
[0436] Error-prone Rolling Circle Amplification (epRCA) (Fujii, R., M. Kitaoka, and K. Hayashi, 2004, One-step random mutagenesis by error-prone rolling circle amplification. Nucleic Acids Res 32:e145; and Fujii, R., M. Kitaoka, and K. Hayashi, 2006, Error-prone rolling circle amplification: the simplest random mutagenesis protocol. Nat. Protoc. 1:2493-2497) has many of the same elements as epPCR except a whole circular plasmid is used as the template and random 6-mers with exonuclease resistant thiophosphate linkages on the last 2 nucleotides are used to amplify the plasmid followed by transformation into cells in which the plasmid is re-circularized at tandem repeats. Adjusting the Mn2+ concentration can vary the mutation rate somewhat. This technique uses a simple error-prone, single-step method to create a full copy of the plasmid with 3-4 mutations/kbp. No restriction enzyme digestion or specific primers are required. Additionally, this method is typically available as a kit.
[0437] DNA or Family Shuffling (Stemmer, W. P. 1994, DNA shuffling by random fragmentation and reassembly: in vitro recombination for molecular evolution. Proc Natl Acad Sci US.A 91:10747-10751; and Stemmer, W. P. 1994. Rapid evolution of a protein in vitro by DNA shuffling. Nature 370:389-391) typically involves digestion of 2 or more variant genes with nucleases such as Dnase I or EndoV to generate a pool of random fragments that are reassembled by cycles of annealing and extension in the presence of DNA polymerase to create a library of chimeric genes. Fragments prime each other and recombination occurs when one copy primes another copy (template switch). This method can be used with >1 kbp DNA sequences. In addition to mutational recombinants created by fragment reassembly, this method introduces point mutations in the extension steps at a rate similar to error-prone PCR. The method can be used to remove deleterious random neutral mutations that might confer antigenicity.
[0438] Staggered Extension (StEP) (Zhao, H., L. Giver, Z. Shao, J. A. Affholter, and F. H. Arnold, 1998, Molecular evolution by staggered extension process (StEP) in vitro recombination. Nat. Biotechnol 16:258-261) entails template priming followed by repeated cycles of 2 step PCR with denaturation and very short duration of annealing/extension (as short as 5 sec). Growing fragments anneal to different templates and extend further, which is repeated until full-length sequences are made. Template switching means most resulting fragments have multiple parents. Combinations of low-fidelity polymerases (Taq and Mutazyme) reduce error-prone biases because of opposite mutational spectra.
[0439] In Random Priming Recombination (RPR) random sequence primers are used to generate many short DNA fragments complementary to different segments of the template. (Shao, Z., H. Zhao, L. Giver, and F. H. Arnold, 1998, Random-priming in vitro recombination: an effective tool for directed evolution. Nucleic Acids Res 26:681-683.) Base misincorporation and mispriming via epPCR give point mutations. Short DNA fragments prime one another based on homology and are recombined and reassembled into full-length by repeated thermocycling. Removal of templates prior to this step assures low parental recombinants. This method, like most others, can be performed over multiple iterations to evolve distinct properties. This technology avoids sequence bias, is independent of gene length, and requires very little parent DNA for the application.
[0440] In Heteroduplex Recombination linearized plasmid DNA is used to form heteroduplexes that are repaired by mismatch repair. (Volkov, A. A., Z. Shao, and F. H. Arnold. 1999. Recombination and chimeragenesis by in vitro heteroduplex formation and in vivo repair. Nucleic Acids Res 27:e18; and Volkov, A. A., Z. Shao, and F. H. Arnold. 2000. Random chimeragenesis by heteroduplex recombination. Methods Enzymol. 328:456-463.) The mismatch repair step is at least somewhat mutagenic. Heteroduplexes transform more efficiently than linear homoduplexes. This method is suitable for large genes and whole operons.
[0441] Random Chimeragenesis on Transient Templates (RACHITT) (Coco, W. M., W. E. Levinson, M. J. Crist, H. J. Hektor, A. Darzins, P. T. Pienkos, C. H. Squires, and D. J. Monticello, 2001, DNA shuffling method for generating highly recombined genes and evolved enzymes. Nat. Biotechnol 19:354-359) employs Dnase I fragmentation and size fractionation of ssDNA. Homologous fragments are hybridized in the absence of polymerase to a complementary ssDNA scaffold. Any overlapping unhybridized fragment ends are trimmed down by an exonuclease. Gaps between fragments are filled in, and then ligated to give a pool of full-length diverse strands hybridized to the scaffold (that contains U to preclude amplification). The scaffold then is destroyed and is replaced by a new strand complementary to the diverse strand by PCR amplification. The method involves one strand (scaffold) that is from only one parent while the priming fragments derive from other genes; the parent scaffold is selected against. Thus, no reannealing with parental fragments occurs. Overlapping fragments are trimmed with an exonuclease. Otherwise, this is conceptually similar to DNA shuffling and StEP. Therefore, there should be no siblings, few inactives, and no unshuffled parentals. This technique has advantages in that few or no parental genes are created and many more crossovers can result relative to standard DNA shuffling.
[0442] Recombined Extension on Truncated templates (RETT) entails template switching of unidirectionally growing strands from primers in the presence of unidirectional ssDNA fragments used as a pool of templates. (Lee, S. H., E. J. Ryu, M. J. Kang, E.-S. Wang, Z. C. Y. Piao, K. J. J. Jung, and Y. Shin, 2003, A new approach to directed gene evolution by recombined extension on truncated templates (RETT). J. Molec. Catalysis 26:119-129.) No DNA endonucleases are used. Unidirectional ssDNA is made by DNA polymerase with random primers or serial deletion with exonuclease. Unidirectional ssDNA are only templates and not primers. Random priming and exonucleases don't introduce sequence bias as true of enzymatic cleavage of DNA shuffling/RACHITT. RETT can be easier to optimize than StEP because it uses normal PCR conditions instead of very short extensions. Recombination occurs as a component of the PCR steps--no direct shuffling. This method can also be more random than StEP due to the absence of pauses.
[0443] In Degenerate Oligonucleotide Gene Shuffling (DOGS) degenerate primers are used to control recombination between molecules; (Bergquist, P. L. and M. D. Gibbs, 2007, Degenerate oligonucleotide gene shuffling. Methods Mol. Biol. 352:191-204; Bergquist, P. L., R. A. Reeves, and M. D. Gibbs, 2005, Degenerate oligonucleotide gene shuffling (DOGS) and random drift mutagenesis (RNDM): two complementary techniques for enzyme evolution. Biomol. Eng 22:63-72; Gibbs, M. D., K. M. Nevalainen, and P. L. Bergquist, 2001, Degenerate oligonucleotide gene shuffling (DOGS): a method for enhancing the frequency of recombination with family shuffling. Gene 271:13-20) this can be used to control the tendency of other methods such as DNA shuffling to regenerate parental genes. This method can be combined with random mutagenesis (epPCR) of selected gene segments. This can be a good method to block the reformation of parental sequences. No endonucleases are needed. By adjusting input concentrations of segments made, one can bias towards a desired backbone. This method allows DNA shuffling from unrelated parents without restriction enzyme digests and allows a choice of random mutagenesis methods.
[0444] Incremental Truncation for the Creation of Hybrid Enzymes (ITCHY) creates a combinatorial library with 1 base pair deletions of a gene or gene fragment of interest. (Ostermeier et al., Proc Natl Acad Sci US.A. 96:3562-3567 (1999); Ostermeier et al., 1999 Nat. Biotechnol. 17:1205-1209 (1999)) Truncations are introduced in opposite direction on pieces of 2 different genes. These are ligated together and the fusions are cloned. This technique does not require homology between the 2 parental genes. When ITCHY is combined with DNA shuffling, the system is called SCRATCHY (see below). A major advantage of both is no need for homology between parental genes; for example, functional fusions between an E. coli and a human gene were created via ITCHY. When ITCHY libraries are made, all possible crossovers are captured.
[0445] Thio-Incremental Truncation for the Creation of Hybrid Enzymes (THIO-ITCHY) is almost the same as ITCHY except that phosphothioate dNTPs are used to generate truncations. (Lutz, S., M. Ostermeier, and S. J. Benkovic, 2001, Rapid generation of incremental truncation libraries for protein engineering using alpha-phosphothioate nucleotides. Nucleic Acids Res 29:E16) Relative to ITCHY, THIO-ITCHY can be easier to optimize, provide more reproducibility, and adjustability.
[0446] SCRATCHY--ITCHY combined with DNA shuffling is a combination of DNA shuffling and ITCHY; therefore, allowing multiple crossovers. (Lutz et al., Proc Natl Acad Sci US.A. 98:11248-11253 (2001).) SCRATCHY combines the best features of ITCHY and DNA shuffling. Computational predictions can be used in optimization. SCRATCHY is more effective than DNA shuffling when sequence identity is below 80%.
[0447] In Random Drift Mutagenesis (RNDM) mutations made via epPCR followed by screening/selection for those retaining usable activity. (Bergquist et al., Biomol. Eng. 22:63-72 (2005).) Then, these are used in DOGS to generate recombinants with fusions between multiple active mutants or between active mutants and some other desirable parent. Designed to promote isolation of neutral mutations; its purpose is to screen for retained catalytic activity whether or not this activity is higher or lower than in the original gene. RNDM is usable in high throughput assays when screening is capable of detecting activity above background. RNDM has been used as a front end to DOGS in generating diversity. The technique imposes a requirement for activity prior to shuffling or other subsequent steps; neutral drift libraries are indicated to result in higher/quicker improvements in activity from smaller libraries. Though published using epPCR, this could be applied to other large-scale mutagenesis methods.
[0448] Sequence Saturation Mutagenesis (SeSaM) is a random mutagenesis method that: 1) generates pool of random length fragments using random incorporation of a phosphothioate nucleotide and cleavage; this pool is used as a template to 2) extend in the presence of "universal" bases such as inosine; 3) replication of a inosine-containing complement gives random base incorporation and, consequently, mutagenesis. (Wong et al., Biotechnol J. 3:74-82 (2008); Wong Nucleic Acids Res 32:e26; Wong et al. Anal. Biochem. 341:187-189 (2005).) Using this technique it can be possible to generate a large library of mutants within 2-3 days using simple methods. This is very non-directed compared to mutational bias of DNA polymerases. Differences in this approach makes this technique complementary (or alternative) to epPCR.
[0449] In Synthetic Shuffling, overlapping oligonucleotides are designed to encode "all genetic diversity in targets" and allow a very high diversity for the shuffled progeny. (Ness, et al., Nat. Biotechnol 20:1251-1255 (2002).) In this technique, one can design the fragments to be shuffled. This aids in increasing the resulting diversity of the progeny. One can design sequence/codon biases to make more distantly related sequences recombine at rates approaching more closely related sequences and it doesn't require possessing the template genes physically.
[0450] Nucleotide Exchange and Excision Technology NexT exploits a combination of dUTP incorporation followed by treatment with uracil DNA glycosylase and then piperidine to perform endpoint DNA fragmentation. (Muller et al., Nucleic Acids Res 33:e117 (2005)) The gene is reassembled using internal PCR primer extension with proofreading polymerase. The sizes for shuffling are directly controllable using varying dUPT::dTTP ratios. This is an end point reaction using simple methods for uracil incorporation and cleavage. One can use other nucleotide analogs such as 8-oxo-guanine with this method. Additionally, the technique works well with very short fragments (86 bp) and has a low error rate. Chemical cleavage of DNA means very few unshuffled clones.
[0451] In Sequence Homology-Independent Protein Recombination (SHIPREC) a linker is used to facilitate fusion between 2 distantly/unrelated genes; nuclease treatment is used to generate a range of chimeras between the two. Result is a single crossover library of these fusions. (Sieber, V., C. A. Martinez, and F. H. Arnold. 2001. Libraries of hybrid proteins from distantly related sequences. Nat. Biotechnol 19:456-460.) This produces a limited type of shuffling; mutagenesis is a separate process. This technique can create a library of chimeras with varying fractions of each of 2 unrelated parent genes. No homology is needed. SHIPREC was tested with a heme-binding domain of a bacterial CP450 fused to N-terminal regions of a mammalian CP450; this produced mammalian activity in a more soluble enzyme.
[0452] In Gene Site Saturation Mutagenesis (GSSM) the starting materials are a supercoiled dsDNA plasmid with insert and 2 primers degenerate at the desired site for mutations. (Kretz, K. A., T. H. Richardson, K. A. Gray, D. E. Robertson, X. Tan, and J. M. Short, 2004, Gene site saturation mutagenesis: a comprehensive mutagenesis approach. Methods Enzymol. 388:3-11.) Primers carry the mutation of interest and anneal to the same sequence on opposite strands of DNA; mutation in the middle of the primer and .about.20 nucleotides of correct sequence flanking on each side. The sequence in the primer is NNN or NNK (coding) and MNN (noncoding) (N=all 4, K=G, T, M=A, C). After extension, DpnI is used to digest dam-methylated DNA to eliminate the wild-type template. This technique explores all possible amino acid substitutions at a given locus (i.e., one codon). The technique facilitates the generation of all possible replacements at one site with no nonsense codons and equal or near-equal representation of most possible alleles. It does not require prior knowledge of structure, mechanism, or domains of the target enzyme. If followed by shuffling or Gene Reassembly, this technology creates a diverse library of recombinants containing all possible combinations of single-site up-mutations. The utility of this technology combination has been demonstrated for the successful evolution of over 50 different enzymes, and also for more than one property in a given enzyme.
[0453] Combinatorial Cassette Mutagenesis (CCM) involves the use of short oligonucleotide cassettes to replace limited regions with a large number of possible amino acid sequence alterations. (Reidhaar-Olson, J. F., J. U. Bowie, R. M. Breyer, J. C. Hu, K. L. Knight, W. A. Lim, M. C. Mossing, D. A. Parsell, K. R. Shoemaker, and R. T. Sauer, 1991, Random mutagenesis of protein sequences using oligonucleotide cassettes. Methods Enzymol. 208:564-586; and Reidhaar-Olson, J. F. and R. T. Sauer, 1988, Combinatorial cassette mutagenesis as a probe of the informational content of protein sequences. Science 241:53-57.) Simultaneous substitutions at 2 or 3 sites are possible using this technique. Additionally, the method tests a large multiplicity of possible sequence changes at a limited range of sites. It has been used to explore the information content of lambda repressor DNA-binding domain.
[0454] Combinatorial Multiple Cassette Mutagenesis (CMCM) is essentially similar to CCM except it is employed as part of a larger program: 1) Use of epPCR at high mutation rate to 2) ID hot spots and hot regions and then 3) extension by CMCM to cover a defined region of protein sequence space. (Reetz, M. T., S. Wilensek, D. Zha, and K. E. Jaeger, 2001, Directed Evolution of an Enantioselective Enzyme through Combinatorial Multiple-Cassette Mutagenesis. Angew. Chem. Int. Ed Engl. 40:3589-3591.) As with CCM, this method can test virtually all possible alterations over a target region. If used along with methods to create random mutations and shuffled genes, it provides an excellent means of generating diverse, shuffled proteins. This approach was successful in increasing, by 51-fold, the enantioselectivity of an enzyme.
[0455] In the Mutator Strains technique conditional is mutator plasmids allow increases of 20- to 4000-X in random and natural mutation frequency during selection and to block accumulation of deleterious mutations when selection is not required. (Selifonova, 0., F. Valle, and V. Schellenberger, 2001, Rapid evolution of novel traits in microorganisms. Appl Environ Microbiol 67:3645-3649.) This technology is based on a plasmid-derived mutD5 gene, which encodes a mutant subunit of DNA polymerase III. This subunit binds to endogenous DNA polymerase III and compromises the proofreading ability of polymerase III in any of the strain that harbors the plasmid. A broad-spectrum of base substitutions and frameshift mutations occur. In order for effective use, the mutator plasmid should be removed once the desired phenotype is achieved; this is accomplished through a temperature sensitive origin of replication, which allows plasmid curing at 41.degree. C. It should be noted that mutator strains have been explored for quite some time (e.g., see Winter and coworkers, 1996, J. Mol. Biol. 260, 359-3680. In this technique very high spontaneous mutation rates are observed. The conditional property minimizes non-desired background mutations. This technology could be combined with adaptive evolution to enhance mutagenesis rates and more rapidly achieve desired phenotypes.
[0456] "Look-Through Mutagenesis (LTM) is a multidimensional mutagenesis method that assesses and optimizes combinatorial mutations of selected amino acids." (Rajpal, A., N. Beyaz, L. Haber, G. Cappuccilli, H. Yee, R. R. Bhatt, T. Takeuchi, R. A. Lerner, and R. Crea, 2005, A general method for greatly improving the affinity of antibodies by using combinatorial libraries. Proc Natl Acad Sci US.A 102:8466-8471.) Rather than saturating each site with all possible amino acid changes, a set of 9 is chosen to cover the range of amino acid R-group chemistry. Fewer changes per site allows multiple sites to be subjected to this type of mutagenesis. A >800-fold increase in binding affinity for an antibody from low nanomolar to picomolar has been achieved through this method. This is a rational approach to minimize the number of random combinations and should increase the ability to find improved traits by greatly decreasing the numbers of clones to be screened. This has been applied to antibody engineering, specifically to increase the binding affinity and/or reduce dissociation. The technique can be combined with either screens or selections.
[0457] Gene Reassembly is a DNA shuffling method that can be applied to multiple genes at one time or to creating a large library of chimeras (multiple mutations) of a single gene. (on the world-wide web at www.verenium.com/Pages/Technology/EnzymeTech/TechEnzyTGR.html) Typically this technology is used in combination with ultra-high-throughput screening to query the represented sequence space for desired improvements. This technique allows multiple gene recombination independent of homology. The exact number and position of cross-over events can be pre-determined using fragments designed via bioinformatic analysis. This technology leads to a very high level of diversity with virtually no parental gene reformation and a low level of inactive genes. Combined with GSSM, a large range of mutations can be tested for improved activity. The method allows "blending" and "fine tuning" of DNA shuffling, e.g. codon usage can be optimized.
[0458] In Silico Protein Design Automation PDA is an optimization algorithm that anchors the structurally defined protein backbone possessing a particular fold, and searches sequence space for amino acid substitutions that can stabilize the fold and overall protein energetics. (Hayes, R. J., J. Bentzien, M. L. Ary, M. Y. Hwang, J. M. Jacinto, J. Vielmetter, A. Kundu, and B. I. Dahiyat, 2002, Combining computational and experimental screening for rapid optimization of protein properties. Proc Natl Acad Sci US.A 99:15926-15931.) This technology allows in silico structure-based entropy predictions in order to search for structural tolerance toward protein amino acid variations. Statistical mechanics is applied to calculate coupling interactions at each position--structural tolerance toward amino acid substitution is a measure of coupling. Ultimately, this technology is designed to yield desired modifications of protein properties while maintaining the integrity of structural characteristics. The method computationally assesses and allows filtering of a very large number of possible sequence variants (1050). Choice of sequence variants to test is related to predictions based on most favorable thermodynamics and ostensibly only stability or properties that are linked to stability can be effectively addressed with this technology. The method has been successfully used in some therapeutic proteins, especially in engineering immunoglobulins. In silico predictions avoid testing extraordinarily large numbers of potential variants. Predictions based on existing three-dimensional structures are more likely to succeed than predictions based on hypothetical structures. This technology can readily predict and allow targeted screening of multiple simultaneous mutations, something not possible with purely experimental technologies due to exponential increases in numbers.
[0459] Iterative Saturation Mutagenesis (ISM) involves 1) Use knowledge of structure/function to choose a likely site for enzyme improvement. 2) Saturation mutagenesis at chosen site using Stratagene QuikChange (or other suitable means). 3) Screen/select for desired properties. 4) With improved clone(s), start over at another site and continue repeating. (Reetz, M. T. and J. D. Carballeira, 2007, Iterative saturation mutagenesis (ISM) for rapid directed evolution of functional enzymes. Nat. Protoc. 2:891-903; and Reetz, M. T., J. D. Carballeira, and A. Vogel, 2006, Iterative saturation mutagenesis on the basis of B factors as a strategy for increasing protein thermostability. Angew. Chem. Int. Ed Engl. 45:7745-7751.) This is a proven methodology assures all possible replacements at a given position are made for screening/selection.
[0460] Any of the aforementioned methods for mutagenesis can be used alone or in any combination. Additionally, any one or combination of the directed evolution methods can be used in conjunction with adaptive evolution techniques.
[0461] To generate better producers, metabolic modeling can be utilized to optimize growth conditions. Modeling can also be used to design gene knockouts that additionally optimize utilization of the pathway (see, for example, U.S. patent publications US 2002/0012939, US 2003/0224363, US 2004/0029149, US 2004/0072723, US 2003/0059792, US 2002/0168654 and US 2004/0009466, and U.S. Pat. No. 7,127,379). Modeling analysis allows reliable predictions of the effects on cell growth of shifting the metabolism towards more efficient production of 1,3-butanediol.
[0462] One computational method for identifying and designing metabolic alterations favoring biosynthesis of a desired product is the OptKnock computational framework (Burgard et al., Biotechnol. Bioeng. 84:647-657 (2003)). OptKnock is a metabolic modeling and simulation program that suggests gene deletion strategies that result in genetically stable microorganisms which overproduce the target product. Specifically, the framework examines the complete metabolic and/or biochemical network of a microorganism in order to suggest genetic manipulations that force the desired biochemical to become an obligatory byproduct of cell growth. By coupling biochemical production with cell growth through strategically placed gene deletions or other functional gene disruption, the growth selection pressures imposed on the engineered strains after long periods of time in a bioreactor lead to improvements in performance as a result of the compulsory growth-coupled biochemical production. Lastly, when gene deletions are constructed there is a negligible possibility of the designed strains reverting to their wild-type states because the genes selected by OptKnock are to be completely removed from the genome. Therefore, this computational methodology can be used to either identify alternative pathways that lead to biosynthesis of a desired product or used in connection with the non-naturally occurring microbial organisms for further optimization of biosynthesis of a desired product.
[0463] Briefly, OptKnock is a term used herein to refer to a computational method and system for modeling cellular metabolism. The OptKnock program relates to a framework of models and methods that incorporate particular constraints into flux balance analysis (FBA) models. These constraints include, for example, qualitative kinetic information, qualitative regulatory information, and/or DNA microarray experimental data. OptKnock also computes solutions to various metabolic problems by, for example, tightening the flux boundaries derived through flux balance models and subsequently probing the performance limits of metabolic networks in the presence of gene additions or deletions. OptKnock computational framework allows the construction of model formulations that enable an effective query of the performance limits of metabolic networks and provides methods for solving the resulting mixed-integer linear programming problems. The metabolic modeling and simulation methods referred to herein as OptKnock are described in, for example, U.S. 2002/0168654, WO 2002/055995, and U.S. 2009/0047719.
[0464] Another computational method for identifying and designing metabolic alterations favoring biosynthetic production of a product is a metabolic modeling and simulation system termed SimPheny.RTM.. This computational method and system is described in, for example, U.S. 2003/0233218, filed Jun. 14, 2002, and in WO/2003/106998. SimPheny.RTM. is a computational system that can be used to produce a network model in silico and to simulate the flux of mass, energy or charge through the chemical reactions of a biological system to define a solution space that contains any and all possible functionalities of the chemical reactions in the system, thereby determining a range of allowed activities for the biological system. This approach is referred to as constraints-based modeling because the solution space is defined by constraints such as the known stoichiometry of the included reactions as well as reaction thermodynamic and capacity constraints associated with maximum fluxes through reactions. The space defined by these constraints can be interrogated to determine the phenotypic capabilities and behavior of the biological system or of its biochemical components.
[0465] These computational approaches are consistent with biological realities because biological systems are flexible and can reach the same result in many different ways. Biological systems are designed through evolutionary mechanisms that have been restricted by fundamental constraints that all living systems must face. Therefore, constraints-based modeling strategy embraces these general realities. Further, the ability to continuously impose further restrictions on a network model via the tightening of constraints results in a reduction in the size of the solution space, thereby enhancing the precision with which physiological performance or phenotype can be predicted.
[0466] Given the teachings and guidance provided herein, those skilled in the art will be able to apply various computational frameworks for metabolic modeling and simulation to design and implement biosynthesis of a desired compound in host microbial organisms. Such metabolic modeling and simulation methods include, for example, the computational systems exemplified above as SimPheny.RTM. and OptKnock. For illustration of the invention, some methods are described herein with reference to the OptKnock computation framework for modeling and simulation. Those skilled in the art will know how to apply the identification, design and implementation of the metabolic alterations using OptKnock to any of such other metabolic modeling and simulation computational frameworks and methods well known in the art.
[0467] The methods described above will provide one set of metabolic reactions to disrupt. Elimination of each reaction within the set or metabolic modification can result in a desired product as an obligatory product during the growth phase of the organism. Because the reactions are known, a solution to the bilevel OptKnock problem also will provide the associated gene or genes encoding one or more enzymes that catalyze each reaction within the set of reactions. Identification of a set of reactions and their corresponding genes encoding the enzymes participating in each reaction is generally an automated process, accomplished through correlation of the reactions with a reaction database having a relationship between enzymes and encoding genes.
[0468] Once identified, the set of reactions that are to be disrupted in order to achieve production of a desired product are implemented in the target cell or organism by functional disruption of at least one gene encoding each metabolic reaction within the set. One particularly useful means to achieve functional disruption of the reaction set is by deletion of each encoding gene. However, in some instances, it can be beneficial to disrupt the reaction by other genetic aberrations including, for example, mutation, deletion of regulatory regions such as promoters or cis binding sites for regulatory factors, or by truncation of the coding sequence at any of a number of locations. These latter aberrations, resulting in less than total deletion of the gene set can be useful, for example, when rapid assessments of the coupling of a product are desired or when genetic reversion is less likely to occur.
[0469] To identify additional productive solutions to the above described bilevel OptKnock problem which lead to further sets of reactions to disrupt or metabolic modifications that can result in the biosynthesis, including growth-coupled biosynthesis of a desired product, an optimization method, termed integer cuts, can be implemented. This method proceeds by iteratively solving the OptKnock problem exemplified above with the incorporation of an additional constraint referred to as an integer cut at each iteration. Integer cut constraints effectively prevent the solution procedure from choosing the exact same set of reactions identified in any previous iteration that obligatorily couples product biosynthesis to growth. For example, if a previously identified growth-coupled metabolic modification specifies reactions 1, 2, and 3 for disruption, then the following constraint prevents the same reactions from being simultaneously considered in subsequent solutions. The integer cut method is well known in the art and can be found described in, for example, Burgard et al., Biotechnol. Prog. 17:791-797 (2001). As with all methods described herein with reference to their use in combination with the OptKnock computational framework for metabolic modeling and simulation, the integer cut method of reducing redundancy in iterative computational analysis also can be applied with other computational frameworks well known in the art including, for example, SimPheny.RTM..
[0470] The methods exemplified herein allow the construction of cells and organisms that biosynthetically produce a desired product, including the obligatory coupling of production of a target biochemical product to growth of the cell or organism engineered to harbor the identified genetic alterations. Therefore, the computational methods described herein allow the identification and implementation of metabolic modifications that are identified by an in silico method selected from OptKnock or SimPheny.RTM.. The set of metabolic modifications can include, for example, addition of one or more biosynthetic pathway enzymes and/or functional disruption of one or more metabolic reactions including, for example, disruption by gene deletion.
[0471] As discussed above, the OptKnock methodology was developed on the premise that mutant microbial networks can be evolved towards their computationally predicted maximum-growth phenotypes when subjected to long periods of growth selection. In other words, the approach leverages an organism's ability to self-optimize under selective pressures. The OptKnock framework allows for the exhaustive enumeration of gene deletion combinations that force a coupling between biochemical production and cell growth based on network stoichiometry. The identification of optimal gene/reaction knockouts requires the solution of a bilevel optimization problem that chooses the set of active reactions such that an optimal growth solution for the resulting network overproduces the biochemical of interest (Burgard et al., Biotechnol. Bioeng. 84:647-657 (2003)).
[0472] An in silico stoichiometric model of E. coli metabolism can be employed to identify essential genes for metabolic pathways as exemplified previously and described in, for example, U.S. patent publications US 2002/0012939, US 2003/0224363, US 2004/0029149, US 2004/0072723, US 2003/0059792, US 2002/0168654 and US 2004/0009466, and in U.S. Pat. No. 7,127,379. As disclosed herein, the OptKnock mathematical framework can be applied to pinpoint gene deletions leading to the growth-coupled production of a desired product. Further, the solution of the bilevel OptKnock problem provides only one set of deletions. To enumerate all meaningful solutions, that is, all sets of knockouts leading to growth-coupled production formation, an optimization technique, termed integer cuts, can be implemented. This entails iteratively solving the OptKnock problem with the incorporation of an additional constraint referred to as an integer cut at each iteration, as discussed above.
[0473] As disclosed herein, a nucleic acid encoding a desired activity of a 1,3-butanediol pathway can be introduced into a host organism. In some cases, it can be desirable to modify an activity of a 1,3-butanediol pathway enzyme or protein to increase production of 1,3-butanediol. For example, known mutations that increase the activity of a protein or enzyme can be introduced into an encoding nucleic acid molecule. Additionally, optimization methods can be applied to increase the activity of an enzyme or protein and/or decrease an inhibitory activity, for example, decrease the activity of a negative regulator.
[0474] One such optimization method is directed evolution. Directed evolution is a powerful approach that involves the introduction of mutations targeted to a specific gene in order to improve and/or alter the properties of an enzyme. Improved and/or altered enzymes can be identified through the development and implementation of sensitive high-throughput screening assays that allow the automated screening of many enzyme variants (for example, >10.sup.4). Iterative rounds of mutagenesis and screening typically are performed to afford an enzyme with optimized properties. Computational algorithms that can help to identify areas of the gene for mutagenesis also have been developed and can significantly reduce the number of enzyme variants that need to be generated and screened. Numerous directed evolution technologies have been developed (for reviews, see Hibbert et al., Biomol. Eng 22:11-19 (2005); Huisman and Lalonde, In Biocatalysis in the pharmaceutical and biotechnology industries pgs. 717-742 (2007), Patel (ed.), CRC Press; Otten and Quax. Biomol. Eng 22:1-9 (2005); and Sen et al., Appl Biochem. Biotechnol 143:212-223 (2007)) to be effective at creating diverse variant libraries, and these methods have been successfully applied to the improvement of a wide range of properties across many enzyme classes. Enzyme characteristics that have been improved and/or altered by directed evolution technologies include, for example: selectivity/specificity, for conversion of non-natural substrates; temperature stability, for robust high temperature processing; pH stability, for bioprocessing under lower or higher pH conditions; substrate or product tolerance, so that high product titers can be achieved; binding (K.sub.m), including broadening substrate binding to include non-natural substrates; inhibition (K.sub.i), to remove inhibition by products, substrates, or key intermediates; activity (kcat), to increases enzymatic reaction rates to achieve desired flux; expression levels, to increase protein yields and overall pathway flux; oxygen stability, for operation of air sensitive enzymes under aerobic conditions; and anaerobic activity, for operation of an aerobic enzyme in the absence of oxygen.
[0475] A number of exemplary methods have been developed for the mutagenesis and diversification of genes to target desired properties of specific enzymes. Such methods are well known to those skilled in the art. Any of these can be used to alter and/or optimize the activity of a 1,3-butanediol pathway enzyme or protein. Such methods include, but are not limited to EpPCR, which introduces random point mutations by reducing the fidelity of DNA polymerase in PCR reactions (Pritchard et al., J Theor. Biol. 234:497-509 (2005)); Error-prone Rolling Circle Amplification (epRCA), which is similar to epPCR except a whole circular plasmid is used as the template and random 6-mers with exonuclease resistant thiophosphate linkages on the last 2 nucleotides are used to amplify the plasmid followed by transformation into cells in which the plasmid is re-circularized at tandem repeats (Fujii et al., Nucleic Acids Res. 32:e145 (2004); and Fujii et al., Nat. Protoc. 1:2493-2497 (2006)); DNA or Family Shuffling, which typically involves digestion of two or more variant genes with nucleases such as Dnase I or EndoV to generate a pool of random fragments that are reassembled by cycles of annealing and extension in the presence of DNA polymerase to create a library of chimeric genes (Stemmer, Proc Natl Acad Sci USA 91:10747-10751 (1994); and Stemmer, Nature 370:389-391 (1994)); Staggered Extension (StEP), which entails template priming followed by repeated cycles of 2 step PCR with denaturation and very short duration of annealing/extension (as short as 5 sec) (Zhao et al., Nat. Biotechnol. 16:258-261 (1998)); Random Priming Recombination (RPR), in which random sequence primers are used to generate many short DNA fragments complementary to different segments of the template (Shao et al., Nucleic Acids Res 26:681-683 (1998)).
[0476] Additional methods include Heteroduplex Recombination, in which linearized plasmid DNA is used to form heteroduplexes that are repaired by mismatch repair (Volkov et al, Nucleic Acids Res. 27:e18 (1999); and Volkov et al., Methods Enzymol. 328:456-463 (2000)); Random Chimeragenesis on Transient Templates (RACHITT), which employs Dnase I fragmentation and size fractionation of single stranded DNA (ssDNA) (Coco et al., Nat. Biotechnol. 19:354-359 (2001)); Recombined Extension on Truncated templates (RETT), which entails template switching of unidirectionally growing strands from primers in the presence of unidirectional ssDNA fragments used as a pool of templates (Lee et al., J. Molec. Catalysis 26:119-129 (2003)); Degenerate Oligonucleotide Gene Shuffling (DOGS), in which degenerate primers are used to control recombination between molecules; (Bergquist and Gibbs, Methods Mol. Biol 352:191-204 (2007); Bergquist et al., Biomol. Eng 22:63-72 (2005); Gibbs et al., Gene 271:13-20 (2001)); Incremental Truncation for the Creation of Hybrid Enzymes (ITCHY), which creates a combinatorial library with 1 base pair deletions of a gene or gene fragment of interest (Ostermeier et al., Proc. Natl. Acad. Sci. USA 96:3562-3567 (1999); and Ostermeier et al., Nat. Biotechnol. 17:1205-1209 (1999)); Thio-Incremental Truncation for the Creation of Hybrid Enzymes (THIO-ITCHY), which is similar to ITCHY except that phosphothioate dNTPs are used to generate truncations (Lutz et al., Nucleic Acids Res 29:E16 (2001)); SCRATCHY, which combines two methods for recombining genes, ITCHY and DNA shuffling (Lutz et al., Proc. Natl. Acad. Sci. USA 98:11248-11253 (2001)); Random Drift Mutagenesis (RNDM), in which mutations made via epPCR are followed by screening/selection for those retaining usable activity (Bergquist et al., Biomol. Eng. 22:63-72 (2005)); Sequence Saturation Mutagenesis (SeSaM), a random mutagenesis method that generates a pool of random length fragments using random incorporation of a phosphothioate nucleotide and cleavage, which is used as a template to extend in the presence of "universal" bases such as inosine, and replication of an inosine-containing complement gives random base incorporation and, consequently, mutagenesis (Wong et al., Biotechnol. J. 3:74-82 (2008); Wong et al., Nucleic Acids Res. 32:e26 (2004); and Wong et al., Anal. Biochem. 341:187-189 (2005)); Synthetic Shuffling, which uses overlapping oligonucleotides designed to encode "all genetic diversity in targets" and allows a very high diversity for the shuffled progeny (Ness et al., Nat. Biotechnol. 20:1251-1255 (2002)); Nucleotide Exchange and Excision Technology NexT, which exploits a combination of dUTP incorporation followed by treatment with uracil DNA glycosylase and then piperidine to perform endpoint DNA fragmentation (Muller et al., Nucleic Acids Res. 33:e117 (2005)).
[0477] Further methods include Sequence Homology-Independent Protein Recombination (SHIPREC), in which a linker is used to facilitate fusion between two distantly related or unrelated genes, and a range of chimeras is generated between the two genes, resulting in libraries of single-crossover hybrids (Sieber et al., Nat. Biotechnol. 19:456-460 (2001)); Gene Site Saturation Mutagenesis.TM. (GSSM.TM.), in which the starting materials include a supercoiled double stranded DNA (dsDNA) plasmid containing an insert and two primers which are degenerate at the desired site of mutations (Kretz et al., Methods Enzymol. 388:3-11 (2004)); Combinatorial Cassette Mutagenesis (CCM), which involves the use of short oligonucleotide cassettes to replace limited regions with a large number of possible amino acid sequence alterations (Reidhaar-Olson et al. Methods Enzymol. 208:564-586 (1991); and Reidhaar-Olson et al. Science 241:53-57 (1988)); Combinatorial Multiple Cassette Mutagenesis (CMCM), which is essentially similar to CCM and uses epPCR at high mutation rate to identify hot spots and hot regions and then extension by CMCM to cover a defined region of protein sequence space (Reetz et al., Angew. Chem. Int. Ed Engl. 40:3589-3591 (2001)); the Mutator Strains technique, in which conditional is mutator plasmids, utilizing the mutD5 gene, which encodes a mutant subunit of DNA polymerase III, to allow increases of 20 to 4000-X in random and natural mutation frequency during selection and block accumulation of deleterious mutations when selection is not required (Selifonova et al., Appl. Environ. Microbiol. 67:3645-3649 (2001)); Low et al., J. Mol. Biol. 260:359-3680 (1996)).
[0478] Additional exemplary methods include Look-Through Mutagenesis (LTM), which is a multidimensional mutagenesis method that assesses and optimizes combinatorial mutations of selected amino acids (Rajpal et al., Proc. Natl. Acad. Sci. USA 102:8466-8471 (2005)); Gene Reassembly, which is a DNA shuffling method that can be applied to multiple genes at one time or to create a large library of chimeras (multiple mutations) of a single gene (Tunable GeneReassembly.TM. (TGR.TM.) Technology supplied by Verenium Corporation), in Silico Protein Design Automation (PDA), which is an optimization algorithm that anchors the structurally defined protein backbone possessing a particular fold, and searches sequence space for amino acid substitutions that can stabilize the fold and overall protein energetics, and generally works most effectively on proteins with known three-dimensional structures (Hayes et al., Proc. Natl. Acad. Sci. USA 99:15926-15931 (2002)); and Iterative Saturation Mutagenesis (ISM), which involves using knowledge of structure/function to choose a likely site for enzyme improvement, performing saturation mutagenesis at chosen site using a mutagenesis method such as Stratagene QuikChange (Stratagene; San Diego Calif.), screening/selecting for desired properties, and, using improved clone(s), starting over at another site and continue repeating until a desired activity is achieved (Reetz et al., Nat. Protoc. 2:891-903 (2007); and Reetz et al., Angew. Chem. Int. Ed Engl. 45:7745-7751 (2006)).
[0479] Any of the aforementioned methods for mutagenesis can be used alone or in any combination. Additionally, any one or combination of the directed evolution methods can be used in conjunction with adaptive evolution techniques, as described herein.
[0480] It is understood that modifications which do not substantially affect the activity of the various embodiments of this invention are also included within the definition of the invention provided herein. Accordingly, the following examples are intended to illustrate but not limit the present invention.
Example I
1,3-Butanediol Synthesis Via Alanine
[0481] This example describes the generation of a microbial organism capable of producing 1,3-butanediol using the alanine pathway in FIG. 1 via Steps A, B, C, D and H.
[0482] Escherichia coli is used as a target organism to engineer a 1,3-butanediol-producing pathway as shown in FIG. 1. E. coli provides a good host for generating a non-naturally occurring microorganism capable of producing 1,3-butanediol. E. coli is amenable to genetic manipulation and is known to be capable of producing various products, like ethanol, acetic acid, formic acid, lactic acid, and succinic acid, effectively under anaerobic or microaerobic conditions.
[0483] To generate an E. coli strain engineered to produce 1,3-butanediol, nucleic acids encoding the enzymes utilized in the alanine pathway as described previously, are expressed in E. coli using well known molecular biology techniques (see, for example, Sambrook, supra, 2001; Ausubel supra, 1999; Roberts et al., supra, 1989).
[0484] In particular, the ortA (YP.sub.--001086914.1), ortB (YP.sub.--001086915.1), dat (P19938), and pdc (P06672) genes encoding the AKP thiolase, AKP aminotransferase and 2,4-dioxopentanoate decarboxylase activities, respectively, are cloned into the pZE13 vector (Expressys, Ruelzheim, Germany) under the PA1/lacO promoter. In addition, the yqhD (NP.sub.--417484.1) and adh (AAA23199.2) genes encoding 3-oxobutyraldehdye reductase (aldehyde reducing) and 4-hydroxy,2-butanone reductase, respectively are cloned into the pZA33 vector (Expressys, Ruelzheim, Germany) under the PA1/lacO promoter. The two sets of plasmids are transformed into E. coli strain MG1655 to express the proteins and enzymes required for 1,3-butanediol synthesis via the alanine pathway. Note that E. coli possesses the ability to form D-alanine.
[0485] The resulting genetically engineered organism is cultured in glucose containing medium following procedures well known in the art (see, for example, Sambrook et al., supra, 2001). The expression of alanine pathway genes is corroborated using methods well known in the art for determining polypeptide expression or enzymatic activity, including for example, Northern blots, PCR amplification of mRNA, immunoblotting. Enzymatic activities of the expressed enzymes are confirmed using assays specific for the individually activities. The ability of the engineered E. coli strain to produce 1,3-butanediol is confirmed using HPLC, gas chromatography-mass spectrometry (GCMS) or liquid chromatography-mass spectrometry (LCMS).
[0486] Microbial strains engineered to have a functional 1,3-butanediol synthesis pathway are further augmented by optimization for efficient utilization of the pathway. Briefly, the engineered strain is assessed to determine whether any of the exogenous genes are expressed at a rate limiting level. Expression is increased for any enzymes expressed at low levels that can limit the flux through the pathway by, for example, introduction of additional gene copy numbers.
[0487] To generate better producers, metabolic modeling is utilized to optimize growth conditions. Modeling is also used to design gene knockouts that additionally optimize utilization of the pathway (see, for example, U.S. patent publications US 2002/0012939, US 2003/0224363, US 2004/0029149, US 2004/0072723, US 2003/0059792, US 2002/0168654 and US 2004/0009466, and in U.S. Pat. No. 7,127,379). Modeling analysis allows reliable predictions of the effects on cell growth of shifting the metabolism towards more efficient production of 1,3-butanediol. One modeling method is the bilevel optimization approach, OptKnock (Burgard et al., Biotechnol. Bioengineer. 84:647-657 (2003)), which is applied to select gene knockouts that collectively result in better production of 1,3-butanediol. Adaptive evolution also can be used to generate better producers of, for example, alanine or 2-amino-4-oxopentanoate intermediates or the 1,3-butanediol product. Adaptive evolution is performed to improve both growth and production characteristics (Fong and Palsson, Nat. Genet. 36:1056-1058 (2004); Alper et al., Science 314:1565-1568 (2006)). Based on the results, subsequent rounds of modeling, genetic engineering and adaptive evolution can be applied to the 1,3-butanediol producer to further increase production.
[0488] For large-scale production of 1,3-butanediol, the above alanine pathway-containing organism is cultured in a fermenter using a medium known in the art to support growth of the organism under anaerobic conditions. Fermentations are performed in either a batch, fed-batch or continuous manner. Anaerobic conditions are maintained by first sparging the medium with nitrogen and then sealing culture vessel (e.g., flasks can be sealed with a septum and crimp-cap). Microaerobic conditions also can be utilized by providing a small hole for limited aeration. The pH of the medium is maintained at a pH of 7 by addition of an acid, such as H2SO4. The growth rate is determined by measuring optical density using a spectrophotometer (600 nm), and the glucose uptake rate by monitoring carbon source depletion over time. Byproducts such as undesirable alcohols, organic acids, and residual glucose can be quantified by HPLC (Shimadzu) with an HPX-087 column (BioRad), using a refractive index detector for glucose and alcohols, and a UV detector for organic acids, Lin et al., Biotechnol. Bioeng., 775-779 (2005).
Example II
1,3-BDO Synthesis Using Acetoacetyl-CoA as the Intermediate
[0489] This Example describes the generation of a microbial organism capable of producing 1,3-butanediol using acetoacetyl-CoA as the precursor (Steps G, H and I in FIG. 2).
[0490] Escherichia coli is used as a target organism to engineer the pathway through Steps G (conversion of acetoacetyl-CoA into 3-hydroxybutyryl-CoA), H (conversion of 3-hydroxybutyryl-CoA into 3-hydroxybutyraldehyde) and I (conversion of 3-hydroxybutyraldehyde into 1,3-butanediol) in FIG. 2. E. coli provides a good host for generating a non-naturally occurring microorganism capable of producing 1,3-butanediol. E. coli is amenable to genetic manipulation and is known to be capable of producing various products, like ethanol, acetic acid, formic acid, lactic acid, and succinic acid, effectively under anaerobic or microaerobic conditions.
[0491] To generate an E. coli strain engineered to produce 1,3-butanediol, nucleic acids encoding the enzymes utilized in the disclosed pathway (Steps G, H and I) as described previously, are expressed in E. coli using well known molecular biology techniques (see, for example, Sambrook, supra, 2001; Ausubel supra, 1999; Roberts et al., supra, 1989). Note that E. coli has a native thiolase encoded by atop (Accession number: NP.sub.--416728.1) that condenses two molecules of acetyl-CoA to form acetoacetyl-CoA.
[0492] Further, hbd (NP.sub.--349314.1) encoding acetoacetyl-CoA reductase (ketone reducing), is cloned into the pZE 13 vector (Expressys, Ruelzheim, Germany) under the PA1/lacO promoter. The plasmid is transformed into E. coli strain MG1655 to express the enzyme required for the formation of 3-hydroxybutyryl-CoA via acetoacetyl-CoA. An aldehyde dehydrogenase (selected from Table 75 below) that converts 3-hydroxybutyryl-CoA into 3-hydroxybutyraldehyde, and an alcohol dehydrogenase (selected from Table 76 below) that further reduces 3-hydroxybutyraldehyde into 1,3-BDO are also cloned into the pZE13 vector under the PA1/lacO promoter.
[0493] The resulting genetically engineered organism is cultured in glucose containing medium following procedures well known in the art (see, for example, Sambrook et al., supra, 2001). The expression of the pathway genes is corroborated using methods well known in the art for determining polypeptide expression or enzymatic activity, including, for example, Northern blots, PCR amplification of mRNA, immunoblotting. Enzymatic activities of the expressed enzymes are confirmed using assays specific for the individually activities. The ability of the engineered E. coli strain to produce 1,3-butanediol is confirmed using HPLC, gas chromatography-mass spectrometry (GCMS) or liquid chromatography-mass spectrometry (LCMS).
[0494] Microbial strains engineered to have a functional 1,3-butanediol synthesis pathway are further augmented by optimization for efficient utilization of the pathway. Briefly, the engineered strain is assessed to determine whether any of the exogenous genes are expressed at a rate limiting level. Expression is increased for any enzymes expressed at low levels that can limit the flux through the pathway by, for example, introduction of additional gene copy numbers.
[0495] To generate better producers, metabolic modeling is utilized to optimize growth conditions. Modeling is also used to design gene knockouts that additionally optimize utilization of the pathway (see, for example, U.S. patent publications US 2002/0012939, US 2003/0224363, US 2004/0029149, US 2004/0072723, US 2003/0059792, US 2002/0168654 and US 2004/0009466, and in U.S. Pat. No. 7,127,379). Modeling analysis allows reliable predictions of the effects on cell growth of shifting the metabolism towards more efficient production of 1,3-butanediol. One modeling method is the bilevel optimization approach, OptKnock (Burgard et al., Biotechnol. Bioengineer. 84:647-657 (2003)), which is applied to select gene knockouts that collectively result in better production of 1,3-butanediol. Adaptive evolution also can be used to generate better producers of, for example, the acetyl-CoA intermediate or the 1,3-butanediol product. Adaptive evolution is performed to improve both growth and production characteristics (Fong and Palsson, Nat. Genet. 36:1056-1058 (2004); Alper et al., Science 314:1565-1568 (2006)). Based on the results, subsequent rounds of modeling, genetic engineering and adaptive evolution can be applied to the 1,3-butanediol producer to further increase production.
[0496] For large-scale production of 1,3-butanediol, the recombinant organism is cultured in a fermenter using a medium known in the art to support growth of the organism under anaerobic conditions. Fermentations are performed in either a batch, fed-batch or continuous manner. Anaerobic conditions are maintained by first sparging the medium with nitrogen and then sealing culture vessel (e.g., flasks can be sealed with a septum and crimp-cap). Microaerobic conditions also can be utilized by providing a small hole for limited aeration. The pH of the medium is maintained at a pH of 7 by addition of an acid, such as H2SO4. The growth rate is determined by measuring optical density using a spectrophotometer (600 nm), and the glucose uptake rate by monitoring carbon source depletion over time. Byproducts such as undesirable alcohols, organic acids, and residual glucose can be quantified by HPLC (Shimadzu) with an HPX-087 column (BioRad), using a refractive index detector for glucose and alcohols, and a UV detector for organic acids (Lin et al., Biotechnol. Bioeng., 90:775-779 (2005)).
[0497] Several aldehyde dehydrogenases were tested for activity on 3-hydroxybutyryl-CoA. Crude lysates of bacteria, each strain carrying one out of six genes listed in Table 75 below encoding for an aldehyde dehydrogenase was tested for activity on 3-hydroxybutyryl-CoA by measuring the release of CoA moiety. The genes that were tested and were found to have significant activity on 3-HBCoA encode the proteins with the following accession and GI numbers:
TABLE-US-00077 TABLE 75 Protein GenBank ID GI Number Organism bld AAP42563.1 31075383 Clostridium saccharoperbutyl- acetonicum ald ACL06658.1 218764192 Desulfatibacillum alkenivorans AK-01 ald YP_001452373 157145054 Citrobacter koseri ATCC BAA-895 pduP NP_460996.1 16765381 Salmonella enterica Typhimurium pduP ABJ64680.1 116099531 Lactobacillus brevis ATCC 367 BselDRAFT_1651 ZP_02169447 163762382 Bacillus selenitireducens MLS10
[0498] To correct for background activity in the lysate, measured activities were compared to a negative control without ALD gene (vector only, "Vo"). FIG. 4 shows the specific activity of each of the tested genes on 3-hydroxybutyryl-CoA. The gene ids are shown on the x-axis.
[0499] Further, bld (GenBank ID: AAP42563.1, GI number: 31075383) was also tested for activity on 3-HBCoA. The following FIG. 5 shows the activity of the gene on 3-hydroxybutyryl-CoA before and after dialysis.
[0500] Alcohol dehydrogenases that were tested for activity on 3-hydroxybutyraldehyde and demonstrated to have significant activity are listed below.
TABLE-US-00078 TABLE 76 Protein GenBank ID GI Number Organism Bdh (Cbei_2181) YP_001309304 150017050 Clostridium beijerinckii Bdh (Cbei_1722) YP_001309535.1 150016596 Clostridium beijerinckii Bdh (Cbei_2421) YP_001309535.1 150017281 Clostridium beijerinckii
[0501] The following protocol was used to demonstrate alcohol dehydrogenase activity (i.e., conversion of 3-hydroxybutyraldehyde to 1,3-BDO) and combined aldehyde and alcohol dehydrogenase activities (i.e., conversion of 3-hydroxybutyryl-CoA into 1,3-BDO).
[0502] Chemically competent cells were transformed with plasmids containing either an aldehyde dehydrogenase or an alcohol dehydrogenase (listed in Tables 75 and 76 above). Colonies from the plates were picked and grown in LB plus 100 ug/ml carbenecillin overnight, then 0.6 mL was used to inoculate 60 mL culture of each alcohol dehydrogenase, or 1.5 mL was used to inoculate a 500 mL culture of each aldehyde dehydrogenase. Cells were grown at 37.degree. C. to an O.D. of .about.0.7 and induced with IPTG. The cultures were incubated at 30.degree. C. during protein expression for 4 hours. The cell cultures were divided into 30 ml aliquots, centrifuged and the cell pellets were stored at -80.degree. C. A sample of the cell culture was used to estimate final cell density.
[0503] Combinations of alcohol dehydrogenases and aldehyde dehydrogenases were screened in a 96-well plate format with 3-hydroxybutyryl-CoA as a substrate plus a control (no substrate). Alternatively, for testing the alcohol dehydrogenases activity, only the alcohol dehydrogenases were added with and without the substrate, 3-hydroxybutyraldehyde. Preparation of cell lysates was performed on ice in the coldroom (4.degree. C.). Final cell density was used to calculate the quantity of Bug Buster cell lysis reagent for each cell pellet. Lysozyme (10 uL) and benzonase (10 uL) were added to 35 ml bugbuster and gently inverted to mix. First, 50 tun of dithiothreitol (100 mM stock) was added to the pellet, then 0.5 ml per O.D. of 1.0 (at 600 nm) of the Bug Buster plus enzyme mixture was added to the cell pellet and gently mixed to resuspend.
[0504] To each well, 50 ul of 1 M MOPS (pH=7.5), and 25 ul of cofactor mixture (4 mM NADH and 4 mM NADPH), both 100 uL aldehyde dehydrogenase cell lysate, 150 uL alcohol dehydrogenase cell lysate or only 150 uL alcohol dehydrogenase cell lysate was added and gently mixed. Then, the relevant substrate was added to the wells. 25 mg of 3-hydroxybutyryl CoA was resuspended in 250 uL water and 5 ul was added to each well testing for both alcohol and aldehyde dehydrogenase activities for a final concentration of 1.8 mM. For testing only the alcohol dehydrogenase activity, 50 uL of 3-hydroxybutyraldehyde (prepared by mixing 0.6 ml acetaldehyde in 5 ml water plus catalytic base (one pellet of NaOH) Guthrie, J. P. (reference attached) was added to each well. The final concentration of 3-hydroxybutyraldehyde in each well was approximately 50 mM. The 96-deepwell plate was sealed with a plastic PCR seal and incubated at 30.degree. C. shaking overnight (18 hours total). Because protein and cell debris form precipitates during the incubation period, the plates were centrifuged for 10 min at 4500.times.g, and the supernate was filtered through a Whatman 96-well filter plate (0.45 .mu.m) prior to LC-MS analysis. Samples were analyzed for 1,3-butanediol formation.
[0505] FIG. 6 shows 1,3-BDO concentrations when 3-hydroxybutyraldehyde was added as a substrate and in the control samples with no substrate. The GI numbers for the alcohol dehydrogenases are shown.
[0506] FIG. 7 shows 1,3-BDO concentrations when 3-hydroxybutyryl-CoA was added as a substrate and in the control samples with no substrate. The GI numbers for the alcohol dehydrogenases are shown. The GI number for the aldehyde dehydrogenase tested in conjunction is 163762382.
Example III
1,3-BDO Synthesis Using 4-Hydroxybutyryl-CoA as the Intermediate
[0507] This Example describes the generation of a microbial organism capable of producing 1,3-butanediol using 4-hydroxybutyryl-CoA as the precursor (Steps A, B and E in FIG. 3).
[0508] Escherichia coli is used as a target organism to engineer the pathway through Steps A, B and E in FIG. 3. E. coli provides a good host for generating a non-naturally occurring microorganism capable of producing 1,3-butanediol. E. coli is amenable to genetic manipulation and is known to be capable of producing various products, like ethanol, acetic acid, formic acid, lactic acid, and succinic acid, effectively under anaerobic or microaerobic conditions.
[0509] To generate an E. coli strain engineered to produce 1,3-butanediol, nucleic acids encoding the enzymes utilized in the disclosed pathway (Steps A, B and E) as described previously, are expressed in E. coli using well known molecular biology techniques (see, for example, Sambrook, supra, 2001; Ausubel supra, 1999; Roberts et al., supra, 1989). A recombinant strain that has ben engineered to produce significant quantities of 4-hydroxybutyryl-CoA has been described by the applicants previously (Burk et al. (US 20090075351) and will be used for inserting the proposed pathway to 1,3-butanediol.
[0510] Further, abfD (YP.sub.--3001396399.1), crt (NP.sub.--349318.1) and adhE2 (AAK09379.1) genes encoding 4-hydroxybutyryl-CoA dehydratase, crotonase and 3-hydroxybutyryl-CoA reductase (alcohol forming) activities respectively, are cloned into the pZE 13 vector (Expressys, Ruelzheim, Germany) under the PA1/lacO promoter. The plasmid is transformed into the recombinant E. coli strain producing 4-hydroxybutyryl-CoA to express the proteins and enzymes required for 1,3-butanediol synthesis from this metabolite.
[0511] The resulting genetically engineered organism is cultured in glucose containing medium following procedures well known in the art (see, for example, Sambrook et al., supra, 2001). The expression of the pathway genes is corroborated using methods well known in the art for determining polypeptide expression or enzymatic activity, including, for example, Northern blots, PCR amplification of mRNA, immunoblotting. Enzymatic activities of the expressed enzymes are confirmed using assays specific for the individually activities. The ability of the engineered E. coli strain to produce 1,3-butanediol is confirmed using HPLC, gas chromatography-mass spectrometry (GCMS) or liquid chromatography-mass spectrometry (LCMS).
[0512] Microbial strains engineered to have a functional 1,3-butanediol synthesis pathway are further augmented by optimization for efficient utilization of the pathway. Briefly, the engineered strain is assessed to determine whether any of the exogenous genes are expressed at a rate limiting level. Expression is increased for any enzymes expressed at low levels that can limit the flux through the pathway by, for example, introduction of additional gene copy numbers.
[0513] To generate better producers, metabolic modeling is utilized to optimize growth conditions. Modeling is also used to design gene knockouts that additionally optimize utilization of the pathway (see, for example, U.S. patent publications US 2002/0012939, US 2003/0224363, US 2004/0029149, US 2004/0072723, US 2003/0059792, US 2002/0168654 and US 2004/0009466, and in U.S. Pat. No. 7,127,379). Modeling analysis allows reliable predictions of the effects on cell growth of shifting the metabolism towards more efficient production of 1,3-butanediol. One modeling method is the bilevel optimization approach, OptKnock (Burgard et al., Biotechnol. Bioengineer. 84:647-657 (2003)), which is applied to select gene knockouts that collectively result in better production of 1,3-butanediol. Adaptive evolution also can be used to generate better producers of, for example, the acetyl-CoA intermediate or the 1,3-butanediol product. Adaptive evolution is performed to improve both growth and production characteristics (Fong and Palsson, Nat. Genet. 36:1056-1058 (2004); Alper et al., Science 314:1565-1568 (2006)). Based on the results, subsequent rounds of modeling, genetic engineering and adaptive evolution can be applied to the 1,3-butanediol producer to further increase production.
[0514] For large-scale production of 1,3-butanediol, the recombinant organism is cultured in a fermenter using a medium known in the art to support growth of the organism under anaerobic conditions. Fermentations are performed in either a batch, fed-batch or continuous manner. Anaerobic conditions are maintained by first sparging the medium with nitrogen and then sealing culture vessel (e.g., flasks can be sealed with a septum and crimp-cap). Microaerobic conditions also can be utilized by providing a small hole for limited aeration. The pH of the medium is maintained at a pH of 7 by addition of an acid, such as H2SO4. The growth rate is determined by measuring optical density using a spectrophotometer (600 nm), and the glucose uptake rate by monitoring carbon source depletion over time. Byproducts such as undesirable alcohols, organic acids, and residual glucose can be quantified by HPLC (Shimadzu) with an HPX-087 column (BioRad), using a refractive index detector for glucose and alcohols, and a UV detector for organic acids (Lin et al., Biotechnol Bioeng. 90:775-779 (2005)).
Example IV
Exemplary Hydrogenase and CO Dehydrogenase Enzymes for Extracting Reducing Equivalents from Syngas and Exemplary Reductive TCA Cycle Enzymes
[0515] Enzymes of the reductive TCA cycle useful in the non-naturally occurring microbial organisms of the present invention include one or more of ATP-citrate lyase and three CO.sub.2-fixing enzymes: isocitrate dehydrogenase, alpha-ketoglutarate:ferredoxin oxidoreductase, pyruvate:ferredoxin oxidoreductase. The presence of ATP-citrate lyase or citrate lyase and alpha-ketoglutarate:ferredoxin oxidoreductase indicates the presence of an active reductive TCA cycle in an organism. Enzymes for each step of the reductive TCA cycle are shown below.
[0516] ATP-citrate lyase (ACL, EC 2.3.3.8), also called ATP citrate synthase, catalyzes the ATP-dependent cleavage of citrate to oxaloacetate and acetyl-CoA. ACL is an enzyme of the RTCA cycle that has been studied in green sulfur bacteria Chlorobium limicola and Chlorobium tepidum. The alpha(4)beta(4) heteromeric enzyme from Chlorobium limicola was cloned and characterized in E. coli (Kanao et al., Eur. J. Biochem. 269:3409-3416 (2002). The C. limicola enzyme, encoded by aclAB, is irreversible and activity of the enzyme is regulated by the ratio of ADP/ATP. A recombinant ACL from Chlorobium tepidum was also expressed in E. coli and the holoenzyme was reconstituted in vitro, in a study elucidating the role of the alpha and beta subunits in the catalytic mechanism (Kim and Tabita, J. Bacteriol. 188:6544-6552 (2006). ACL enzymes have also been identified in Balnearium lithotrophicum, Sulfurihydrogenibium subterraneum and other members of the bacterial phylum Aquificae (Hugler et al., Environ. Microbiol. 9:81-92 (2007)). This activity has been reported in some fungi as well. Exemplary organisms include Sordaria macrospora (Nowrousian et al., Curr. Genet. 37:189-93 (2000), Aspergillus nidulans, Yarrowia lipolytica (Hynes and Murray, Eukaryotic Cell, July: 1039-1048, (2010) and Aspergillus niger (Meijer et al. J. Ind. Microbiol. Biotechnol. 36:1275-1280 (2009). Other candidates can be found based on sequence homology. Information related to these enzymes is tabulated below:
TABLE-US-00079 TABLE 77 Protein GenBank ID GI Number Organism aclA BAB21376.1 12407237 Chlorobium limicola aclB BAB21375.1 12407235 Chlorobium limicola aclA AAM72321.1 21647054 Chlorobium tepidum aclB AAM72322.1 21647055 Chlorobium tepidum aclA ABI50076.1 114054981 Balnearium lithotrophicum aclB ABI50075.1 114054980 Balnearium lithotrophicum aclA ABI50085.1 114055040 Sulfurihydrogenibium subterraneum aclB ABI50084.1 114055039 Sulfurihydrogenibium subterraneum aclA AAX76834.1 62199504 Sulfurimonas denitrificans aclB AAX76835.1 62199506 Sulfurimonas denitrificans acl1 XP_504787.1 50554757 Yarrowia lipolytica acl2 XP_503231.1 50551515 Yarrowia lipolytica SPBC1703.07 NP_596202.1 19112994 Schizosaccharomyces pombe SPAC22A12.16 NP_593246.1 19114158 Schizosaccharomyces pombe acl1 CAB76165.1 7160185 Sordaria macrospora acl2 CAB76164.1 7160184 Sordaria macrospora aclA CBF86850.1 259487849 Aspergillus nidulans aclB CBF86848 259487848 Aspergillus nidulans
[0517] In some organisms the conversion of citrate to oxaloacetate and acetyl-CoA proceeds through a citryl-CoA intermediate and is catalyzed by two separate enzymes, citryl-CoA synthetase (EC 6.2.1.18) and citryl-CoA lyase (EC 4.1.3.34) (Aoshima, M., Appl. Microbiol. Biotechnol. 75:249-255 (2007). Citryl-CoA synthetase catalyzes the activation of citrate to citryl-CoA. The Hydrogenobacter thermophilus enzyme is composed of large and small subunits encoded by ccsA and ccsB, respectively (Aoshima et al., Mol. Micrbiol. 52:751-761 (2004)). The citryl-CoA synthetase of Aquifex aeolicus is composed of alpha and beta subunits encoded by sucC1 and sucD1 (Hugler et al., Environ. Microbiol. 9:81-92 (2007)). Citryl-CoA lyase splits citryl-CoA into oxaloacetate and acetyl-CoA. This enzyme is a homotrimer encoded by ccl in Hydrogenobacter thermophilus (Aoshima et al., Mol. Microbiol. 52:763-770 (2004)) and aq.sub.--150 in Aquifex aeolicus (Hugler et al., supra (2007)). The genes for this mechanism of converting citrate to oxaloacetate and citryl-CoA have also been reported recently in Chlorobium tepidum (Eisen et al., PNAS 99(14): 9509-14 (2002).
TABLE-US-00080 TABLE 78 Protein GenBank ID GI Number Organism ccsA BAD17844.1 46849514 Hydrogenobacter thermophilus ccsB BAD17846.1 46849517 Hydrogenobacter thermophilus sucC1 AAC07285 2983723 Aquifex aeolicus sucD1 AAC07686 2984152 Aquifex aeolicus ccl BAD17841.1 46849510 Hydrogenobacter thermophilus aq_150 AAC06486 2982866 Aquifex aeolicus CT0380 NP_661284 21673219 Chlorobium tepidum CT0269 NP_661173.1 21673108 Chlorobium tepidum CT1834 AAM73055.1 21647851 Chlorobium tepidum
[0518] Oxaloacetate is converted into malate by malate dehydrogenase (EC 1.1.1.37), an enzyme which functions in both the forward and reverse direction. S. cerevisiae possesses three copies of malate dehydrogenase, MDH1 (McAlister-Henn and Thompson, J. Bacteriol. 169:5157-5166 (1987), MDH2 (Minard and McAlister-Henn, Mol. Cell. Biol. 11:370-380 (1991); Gibson and McAlister-Henn, J. Biol. Chem. 278:25628-25636 (2003)), and MDH3 (Steffan and McAlister-Henn, J. Biol. Chem. 267:24708-24715 (1992)), which localize to the mitochondrion, cytosol, and peroxisome, respectively. E. coli is known to have an active malate dehydrogenase encoded by mdh.
TABLE-US-00081 TABLE 79 Protein GenBank ID GI Number Organism MDH1 NP_012838 6322765 Saccharomyces cerevisiae MDH2 NP_014515 116006499 Saccharomyces cerevisiae MDH3 NP_010205 6320125 Saccharomyces cerevisiae Mdh NP_417703.1 16131126 Escherichia coli
[0519] Fumarate hydratase (EC 4.2.1.2) catalyzes the reversible hydration of fumarate to malate. The three fumarases of E. coli, encoded by fumA, fumB and fumC, are regulated under different conditions of oxygen availability. FumB is oxygen sensitive and is active under anaerobic conditions. FumA is active under microanaerobic conditions, and FumC is active under aerobic growth conditions (Tseng et al., J. Bacteriol. 183:461-467 (2001); Woods et al., Biochim. Biophys. Acta 954:14-26 (1988); Guest et al., J. Gen. Microbiol. 131:2971-2984 (1985)). S. cerevisiae contains one copy of a fumarase-encoding gene, FUM1, whose product localizes to both the cytosol and mitochondrion (Sass et al., J. Biol. Chem. 278:45109-45116 (2003)). Additional fumarase enzymes are found in Campylobacter jejuni (Smith et al., Int. J. Biochem. Cell. Biol. 31:961-975 (1999)), Thermus thermophilus (Mizobata et al., Arch. Biochem. Biophys. 355:49-55 (1998)) and Rattus norvegicus (Kobayashi et al., J. Biochem. 89:1923-1931 (1981)). Similar enzymes with high sequence homology include fum1 from Arabidopsis thaliana and fumC from Corynebacterium glutamicum. The MmcBC fumarase from Pelotomaculum thermopropionicum is another class of fumarase with two subunits (Shimoyama et al., FEMS Microbiol. Lett. 270:207-213 (2007)).
TABLE-US-00082 TABLE 80 Protein GenBank ID GI Number Organism fumA NP_416129.1 16129570 Escherichia coli fumB NP_418546.1 16131948 Escherichia coli fumC NP_416128.1 16129569 Escherichia coli FUM1 NP_015061 6324993 Saccharomyces cerevisiae fumC Q8NRN8.1 39931596 Corynebacterium glutamicum fumC O69294.1 9789756 Campylobacter jejuni fumC P84127 75427690 Thermus thermophilus fumH P14408.1 120605 Rattus norvegicus MmcB YP_001211906 147677691 Pelotomaculum thermopropionicum MmcC YP_001211907 147677692 Pelotomaculum thermopropionicum
[0520] Fumarate reductase catalyzes the reduction of fumarate to succinate. The fumarate reductase of E. coli, composed of four subunits encoded by frdABCD, is membrane-bound and active under anaerobic conditions. The electron donor for this reaction is menaquinone and the two protons produced in this reaction do not contribute to the proton gradient (Iverson et al., Science 284:1961-1966 (1999)). The yeast genome encodes two soluble fumarate reductase isozymes encoded by FRDS1 (Enomoto et al., DNA Res. 3:263-267 (1996)) and FRDS2 (Muratsubaki et al., Arch. Biochem. Biophys. 352:175-181 (1998)), which localize to the cytosol and promitochondrion, respectively, and are used during anaerobic growth on glucose (Arikawa et al., FEMS Microbiol. Lett. 165:111-116 (1998)).
TABLE-US-00083 TABLE 81 Protein GenBank ID GI Number Organism FRDS1 P32614 418423 Saccharomyces cerevisiae FRDS2 NP_012585 6322511 Saccharomyces cerevisiae frdA NP_418578.1 16131979 Escherichia coli frdB NP_418577.1 16131978 Escherichia coli frdC NP_418576.1 16131977 Escherichia coli frdD NP_418475.1 16131877 Escherichia coli
[0521] The ATP-dependent acylation of succinate to succinyl-CoA is catalyzed by succinyl-CoA synthetase (EC 6.2.1.5). The product of the LSC1 and LSC2 genes of S. cerevisiae and the sucC and sucD genes of E. coli naturally form a succinyl-CoA synthetase complex that catalyzes the formation of succinyl-CoA from succinate with the concomitant consumption of one ATP, a reaction which is reversible in vivo (Buck et al., Biochemistry 24:6245-6252 (1985)). These proteins are identified below:
TABLE-US-00084 TABLE 82 Protein GenBank ID GI Number Organism LSC1 NP_014785 6324716 Saccharomyces cerevisiae LSC2 NP_011760 6321683 Saccharomyces cerevisiae sucC NP_415256.1 16128703 Escherichia coli sucD AAC73823.1 1786949 Escherichia coli
[0522] Alpha-ketoglutarate:ferredoxin oxidoreductase (EC 1.2.7.3), also known as 2-oxoglutarate synthase or 2-oxoglutarate:ferredoxin oxidoreductase (OFOR), forms alpha-ketoglutarate from CO2 and succinyl-CoA with concurrent consumption of two reduced ferredoxin equivalents. OFOR and pyruvate:ferredoxin oxidoreductase (PFOR) are members of a diverse family of 2-oxoacid:ferredoxin (flavodoxin) oxidoreductases which utilize thiamine pyrophosphate, CoA and iron-sulfur clusters as cofactors and ferredoxin, flavodoxin and FAD as electron carriers (Adams et al., Archaea. Adv. Protein Chem. 48:101-180 (1996)). Enzymes in this class are reversible and function in the carboxylation direction in organisms that fix carbon by the RTCA cycle such as Hydrogenobacter thermophilus, Desulfobacter hydrogenophilus and Chlorobium species (Shiba et al. 1985; Evans et al., Proc. Natl. Acad. ScI. U.S.A. 55:92934 (1966); Buchanan, 1971). The two-subunit enzyme from H. thermophilus, encoded by korAB, has been cloned and expressed in E. coli (Yun et al., Biochem. Biophys. Res. Commun. 282:589-594 (2001)). A five subunit OFOR from the same organism with strict substrate specificity for succinyl-CoA, encoded by forDABGE, was recently identified and expressed in E. coli (Yun et al. 2002). The kinetics of CO.sub.2 fixation of both H. thermophilus OFOR enzymes have been characterized (Yamamoto et al., Extremophiles 14:79-85 (2010)). A CO2-fixing OFOR from Chlorobium thiosulfatophilum has been purified and characterized but the genes encoding this enzyme have not been identified to date. Enzyme candidates in Chlorobium species can be inferred by sequence similarity to the H. thermophilus genes. For example, the Chlorobium limicola genome encodes two similar proteins. Acetogenic bacteria such as Moorella thermoacetica are predicted to encode two OFOR enzymes. The enzyme encoded by Moth.sub.--0034 is predicted to function in the CO2-assimilating direction. The genes associated with this enzyme, Moth.sub.--0034 have not been experimentally validated to date but can be inferred by sequence similarity to known OFOR enzymes.
[0523] OFOR enzymes that function in the decarboxylation direction under physiological conditions can also catalyze the reverse reaction. The OFOR from the thermoacidophilic archaeon Sulfolobus sp. strain 7, encoded by ST2300, has been extensively studied (Zhang et al. 1996. A plasmid-based expression system has been developed for efficiently expressing this protein in E. coli (Fukuda et al., Eur. J. Biochem. 268:5639-5646 (2001)) and residues involved in substrate specificity were determined (Fukuda and Wakagi, Biochim. Biophys. Acta 1597:74-80 (2002)). The OFOR encoded by Ape1472/Ape1473 from Aeropyrum pernix str. K1 was recently cloned into E. coli, characterized, and found to react with 2-oxoglutarate and a broad range of 2-oxoacids (Nishizawa et al., FEBS Lett. 579:2319-2322 (2005)). Another exemplary OFOR is encoded by oorDABC in Helicobacter pylori (Hughes et al. 1998). An enzyme specific to alpha-ketoglutarate has been reported in Thauera aromatica (Dorner and Boll, J, Bacteriol. 184 (14), 3975-83 (2002). A similar enzyme can be found in Rhodospirillum rubrum by sequence homology. A two subunit enzyme has also been identified in Chlorobium tepidum (Eisen et al., PNAS 99(14): 9509-14 (2002)).
TABLE-US-00085 TABLE 83 Protein GenBank ID GI Number Organism korA BAB21494 12583691 Hydrogenobacter thermophilus korB BAB21495 12583692 Hydrogenobacter thermophilus forD BAB62132.1 14970994 Hydrogenobacter thermophilus forA BAB62133.1 14970995 Hydrogenobacter thermophilus forB BAB62134.1 14970996 Hydrogenobacter thermophilus forG BAB62135.1 14970997 Hydrogenobacter thermophilus forE BAB62136.1 14970998 Hydrogenobacter thermophilus Clim_0204 ACD89303.1 189339900 Chlorobium limicola Clim_0205 ACD89302.1 189339899 Chlorobium limicola Clim_1123 ACD90192.1 189340789 Chlorobium limicola Clim_1124 ACD90193.1 189340790 Chlorobium limicola Moth_1984 YP_430825.1 83590816 Moorella thermoacetica Moth_1985 YP_430826.1 83590817 Moorella thermoacetica Moth_0034 YP_428917.1 83588908 Moorella thermoacetica ST2300 NP_378302.1 15922633 Sulfolobus sp. strain 7 Ape1472 BAA80470.1 5105156 Aeropyrum pernix Ape1473 BAA80471.2 116062794 Aeropyrum pernix oorD NP_207383.1 15645213 Helicobacter pylori oorA NP_207384.1 15645214 Helicobacter pylori oorB NP_207385.1 15645215 Helicobacter pylori oorC NP_207386.1 15645216 Helicobacter pylori CT0163 NP_661069.1 21673004 Chlorobium tepidum CT0162 NP_661068.1 21673003 Chlorobium tepidum korA CAA12243.2 19571179 Thauera aromatica korB CAD27440.1 19571178 Thauera aromatica Rru_A2721 YP_427805.1 83594053 Rhodospirillum rubrum Rru_A2722 YP_427806.1 83594054 Rhodospirillum rubrum
[0524] Isocitrate dehydrogenase catalyzes the reversible decarboxylation of isocitrate to 2-oxoglutarate coupled to the reduction of NAD(P).sup.+. IDH enzymes in Saccharomyces cerevisiae and Escherichia coli are encoded by IDP1 and icd, respectively (Haselbeck and McAlister-Henn, J. Biol. Chem. 266:2339-2345 (1991); Nimmo, H. G., Biochem. J. 234:317-2332 (1986)). The reverse reaction in the reductive TCA cycle, the reductive carboxylation of 2-oxoglutarate to isocitrate, is favored by the NADPH-dependent CO.sub.2-fixing IDH from Chlorobium limicola and was functionally expressed in E. coli (Kanao et al., Eur. J. Biochem. 269:1926-1931 (2002)). A similar enzyme with 95% sequence identity is found in the C. tepidum genome in addition to some other candidates listed below.
TABLE-US-00086 TABLE 84 Protein GenBank ID GI Number Organism Icd ACI84720.1 209772816 Escherichia coli IDP1 AAA34703.1 171749 Saccharomyces cerevisiae Idh BAC00856.1 21396513 Chlorobium limicola Icd AAM71597.1 21646271 Chlorobium tepidum icd NP_952516.1 39996565 Geobacter sulfurreducens icd YP_393560. 78777245 Sulfurimonas denitrificans
[0525] In H. thermophilus the reductive carboxylation of 2-oxoglutarate to isocitrate is catalyzed by two enzymes: 2-oxoglutarate carboxylase and oxalosuccinate reductase. 2-Oxoglutarate carboxylase (EC 6.4.1.7) catalyzes the ATP-dependent carboxylation of alpha-ketoglutarate to oxalosuccinate (Aoshima and Igarashi, Mol. Microbiol. 62:748-759 (2006)). This enzyme is a large complex composed of two subunits. Biotinylation of the large (A) subunit is required for enzyme function (Aoshima et al., Mol. Microbiol. 51:791-798 (2004)). Oxalosuccinate reductase (EC 1.1.1.-) catalyzes the NAD-dependent conversion of oxalosuccinate to D-threo-isocitrate. The enzyme is a homodimer encoded by icd in H. thermophilus. The kinetic parameters of this enzyme indicate that the enzyme only operates in the reductive carboxylation direction in vivo, in contrast to isocitrate dehydrogenase enzymes in other organisms (Aoshima and Igarashi, J. Bacteriol. 190:2050-2055 (2008)). Based on sequence homology, gene candidates have also been found in Thiobacillus denitrilicans and Thermocrinis albus.
TABLE-US-00087 TABLE 85 Protein GenBank ID GI Number Organism cfiA BAF34932.1 116234991 Hydrogenobacter thermophilus cifB BAF34931.1 116234990 Hydrogenobacter thermophilus Icd BAD02487.1 38602676 Hydrogenobacter thermophilus Tbd_1556 YP_315314 74317574 Thiobacillus denitrificans Tbd_1555 YP_315313 74317573 Thiobacillus denitrificans Tbd_0854 YP_314612 74316872 Thiobacillus denitrificans Thal_0268 YP_003473030 289548042 Thermocrinis albus Thal_0267 YP_003473029 289548041 Thermocrinis albus Thal_0646 YP_003473406 289548418 Thermocrinis albus
[0526] Aconitase (EC 4.2.1.3) is an iron-sulfur-containing protein catalyzing the reversible isomerization of citrate and iso-citrate via the intermediate cis-aconitate. Two aconitase enzymes are encoded in the E. coli genome by acnA and acnB. AcnB is the main catabolic enzyme, while AcnA is more stable and appears to be active under conditions of oxidative or acid stress (Cunningham et al., Microbiology 143 (Pt 12):3795-3805 (1997)). Two isozymes of aconitase in Salmonella typhimurium are encoded by acnA and acnB (Horswill and Escalante-Semerena, Biochemistry 40:4703-4713 (2001)). The S. cerevisiae aconitase, encoded by ACO1, is localized to the mitochondria where it participates in the TCA cycle (Gangloff et al., Mol. Cell. Biol. 10:3551-3561 (1990)) and the cytosol where it participates in the glyoxylate shunt (Regev-Rudzki et al., Mol. Biol. Cell. 16:4163-4171 (2005)).
TABLE-US-00088 TABLE 86 Protein GenBank ID GI Number Organism acnA AAC7438.1 1787531 Escherichia coli acnB AAC73229.1 2367097 Escherichia coli HP0779 NP_207572.1 15645398 Helicobacter pylori 26695 H16_B0568 CAJ95365.1 113529018 Ralstonia eutropha DesfrDRAFT_3783 ZP_07335307.1 303249064 Desulfovibrio fructosovorans JJ Suden_1040 ABB44318.1 78497778 Sulfurimonas denitrificans (acnB) Hydth_0755 ADO45152.1 308751669 Hydrogenobacter thermophilus CT0543 (acn) AAM71785.1 21646475 Chlorobium tepidum Clim_2436 YP_001944436.1 189347907 Chlorobium limicola Clim_0515 ACD89607.1 189340204 Chlorobium limicola acnA NP_460671.1 16765056 Salmonella typhimurium acnB NP_459163.1 16763548 Salmonella typhimurium ACO1 AAA34389.1 170982 Saccharomyces cerevisiae
[0527] Pyruvate:ferredoxin oxidoreductase (PFOR) catalyzes the reversible oxidation of pyruvate to form acetyl-CoA. The PFOR from Desulfovibrio africanus has been cloned and expressed in E. coli resulting in an active recombinant enzyme that was stable for several days in the presence of oxygen (Pieulle et al., J. Bacteriol. 179:5684-5692 (1997)). Oxygen stability is relatively uncommon in PFORs and is believed to be conferred by a 60 residue extension in the polypeptide chain of the D. africanus enzyme. Two cysteine residues in this enzyme form a disulfide bond that protects it against inactivation in the form of oxygen. This disulfide bond and the stability in the presence of oxygen has been found in other Desulfovibrio species also (Vita et al., Biochemistry, 47: 957-64 (2008)). The M. thermoacetica PFOR is also well characterized (Menon and Ragsdale, Biochemistry 36:8484-8494 (1997)) and was shown to have high activity in the direction of pyruvate synthesis during autotrophic growth (Furdui and Ragsdale, J. Biol. Chem. 275:28494-28499 (2000)). Further, E. coli possesses an uncharacterized open reading frame, ydbK, encoding a protein that is 51% identical to the M. thermoacetica PFOR. Evidence for pyruvate oxidoreductase activity in E. coli has been described (Blaschkowski et al., Eur. J. Biochem. 123:563-569 (1982)). PFORs have also been described in other organisms, including Rhodobacter capsulatas (Yakunin and Hallenbeck, Biochimica et Biophysica Acta 1409 (1998) 39-49 (1998)) and Choloboum tepidum (Eisen et al., PNAS 99(14): 9509-14 (2002)). The five subunit PFOR from H. thermophilus, encoded by porEDABG, was cloned into E. coli and shown to function in both the decarboxylating and CO.sub.2-assimilating directions (Ikeda et al. 2006; Yamamoto et al., Extremophiles 14:79-85 (2010)). Homologs also exist in C. carboxidivorans P7. Several additional PFOR enzymes are described in the following review (Ragsdale, S. W., Chem. Rev. 103:2333-2346 (2003)). Finally, flavodoxin reductases (e.g., fgrB from Helicobacter pylori or Campylobacter jejuni) (St Maurice et al., J. Bacteriol. 189:4764-4773 (2007)) or Rnf-type proteins (Seedorf et al., Proc. Natl. Acad. Sci. U.S.A. 105:2128-2133 (2008); and Herrmann, J. Bacteriol 190:784-791 (2008)) provide a means to generate NADH or NADPH from the reduced ferredoxin generated by PFOR. These proteins are identified below.
TABLE-US-00089 TABLE 87 Protein GenBank ID GI Number Organism DesfrDRAFT_0121 ZP_07331646.1 303245362 Desulfovibrio fructosovorans JJ Por CAA70873.1 1770208 Desulfovibrio africanus por YP_012236.1 46581428 Desulfovibrio vulgaris str. Hildenborough Dde_3237 ABB40031.1 78220682 DesulfoVibrio desulfuricans G20 Ddes_0298 YP_002478891.1 220903579 Desulfovibrio desulfuricans subsp. desulfuricans str. ATCC 27774 Por YP_428946.1 83588937 Moorella thermoacetica YdbK NP_415896.1 16129339 Escherichia coli nifJ (CT1628) NP_662511.1 21674446 Chlorobium tepidum CJE1649 YP_179630.1 57238499 Campylobacter jejuni nifJ ADE85473.1 294476085 Rhodobacter capsulatus porE BAA95603.1 7768912 Hydrogenobacter thermophilus porD BAA95604.1 7768913 Hydrogenobacter thermophilus porA BAA95605.1 7768914 Hydrogenobacter thermophilus porB BAA95606.1 776891 Hydrogenobacter thermophilus porG BAA95607.1 7768916 Hydrogenobacter thermophilus FqrB YP_001482096.1 157414840 Campylobacter jejuni HP1164 NP_207955.1 15645778 Helicobacter pylori RnfC EDK33306.1 146346770 Clostridium kluyveri RnfD EDK33307.1 146346771 Clostridium kluyveri RnfG EDK33308.1 146346772 Clostridium kluyveri RnfE EDK33309.1 146346773 Clostridium kluyveri RnfA EDK33310.1 146346774 Clostridium kluyveri RnfB EDK33311.1 146346775 Clostridium kluyveri
[0528] The conversion of pyruvate into acetyl-CoA can be catalyzed by several other enzymes or their combinations thereof. For example, pyruvate dehydrogenase can transform pyruvate into acetyl-CoA with the concomitant reduction of a molecule of NAD into NADH. It is a multi-enzyme complex that catalyzes a series of partial reactions which results in acylating oxidative decarboxylation of pyruvate. The enzyme comprises of three subunits: the pyruvate decarboxylase (E1), dihydrolipoamide acyltransferase (E2) and dihydrolipoamide dehydrogenase (E3). This enzyme is naturally present in several organisms, including E. coli and S. cerevisiae. In the E. coli enzyme, specific residues in the E1 component are responsible for substrate specificity (Bisswanger, H., J. Biol. Chem. 256:815-82 (1981); Bremer, J., Eur. J. Biochem. 8:535-540 (1969); Gong et al., J. Biol. Chem. 275:13645-13653 (2000)). Enzyme engineering efforts have improved the E. coli PDH enzyme activity under anaerobic conditions (Kim et al., J. Bacteriol. 190:3851-3858 (2008); Kim et al., Appl. Environ. Microbiol. 73:1766-1771 (2007); Zhou et al., Biotechnol. Lett. 30:335-342 (2008)). In contrast to the E. coli PDH, the B. subtilis complex is active and required for growth under anaerobic conditions (Nakano et al., J. Bacteriol. 179:6749-6755 (1997)). The Klebsiella pneumoniae PDH, characterized during growth on glycerol, is also active under anaerobic conditions (5). Crystal structures of the enzyme complex from bovine kidney (18) and the E2 catalytic domain from Azotobacter vinelandii are available (4). Yet another enzyme that can catalyze this conversion is pyruvate formate lyase. This enzyme catalyzes the conversion of pyruvate and CoA into acetyl-CoA and formate. Pyruvate formate lyase is a common enzyme in prokaryotic organisms that is used to help modulate anaerobic redox balance. Exemplary enzymes can be found in Escherichia coli encoded by pf1/3 (Knappe and Sawers, FEMS. Microbiol Rev. 6:383-398 (1990)), Lactococcus lactis (Melchiorsen et al., Appl Microbiol Biotechnol 58:338-344 (2002)), and Streptococcus mutans (Takahashi-Abbe et al., Oral. Microbiol Immunol. 18:293-297 (2003)). E. coli possesses an additional pyruvate formate lyase, encoded by tdcE, that catalyzes the conversion of pyruvate or 2-oxobutanoate to acetyl-CoA or propionyl-CoA, respectively (Hesslinger et al., Mol. Microbiol 27:477-492 (1998)). Both pflB and tdcE from E. coli require the presence of pyruvate formate lyase activating enzyme, encoded by pflA. Further, a short protein encoded by yfiD in E. coli can associate with and restore activity to oxygen-cleaved pyruvate formate lyase (Vey et al., Proc. Natl. Acad. Sci. U.S.A. 105:16137-16141 (2008). Note that pflA and pflB from E. coli were expressed in S. cerevisiae as a means to increase cytosolic acetyl-CoA for butanol production as described in WO/2008/080124]. Additional pyruvate formate lyase and activating enzyme candidates, encoded by pfl and act, respectively, are found in Clostridium pasteurianum (Weidner et al., J. Bacteriol. 178:2440-2444 (1996)).
[0529] Further, different enzymes can be used in combination to convert pyruvate into acetyl-CoA. For example, in S. cerevisiae, acetyl-CoA is obtained in the cytosol by first decarboxylating pyruvate to form acetaldehyde; the latter is oxidized to acetate by acetaldehyde dehydrogenase and subsequently activated to form acetyl-CoA by acetyl-CoA synthetase. Acetyl-CoA synthetase is a native enzyme in several other organisms including E. coli (Kumari et al., J. Bacteriol. 177:2878-2886 (1995)), Salmonella enterica (Starai et al., Microbiology 151:3793-3801 (2005); Starai et al., J. Biol. Chem. 280:26200-26205 (2005)), and Moorella thermoacetica (described already). Alternatively, acetate can be activated to form acetyl-CoA by acetate kinase and phosphotransacetylase. Acetate kinase first converts acetate into acetyl-phosphate with the accompanying use of an ATP molecule. Acetyl-phosphate and CoA are next converted into acetyl-CoA with the release of one phosphate by phosphotransacetylase. Both acetate kinase and phosphotransacetylyase are well-studied enzymes in several Clostridia and Methanosarcina thermophila.
[0530] Yet another way of converting pyruvate to acetyl-CoA is via pyruvate oxidase. Pyruvate oxidase converts pyruvate into acetate, using ubiquione as the electron acceptor. In E. coli, this activity is encoded by poxB. PoxB has similarity to pyruvate decarboxylase of S. cerevisiae and Zymomonas mobilis. The enzyme has a thiamin pyrophosphate cofactor (Koland and Gennis, Biochemistry 21:4438-4442 (1982)); O'Brien et al., Biochemistry 16:3105-3109 (1977); O'Brien and Gennis, J. Biol. Chem. 255:3302-3307 (1980)) and a flavin adenine dinucleotide (FAD) cofactor. Acetate can then be converted into acetyl-CoA by either acetyl-CoA synthetase or by acetate kinase and phosphotransacetylase, as described earlier. Some of these enzymes can also catalyze the reverse reaction from acetyl-CoA to pyruvate.
[0531] For enzymes that use reducing equivalents in the form of NADH or NADPH, these reduced carriers can be generated by transferring electrons from reduced ferredoxin. Two enzymes catalyze the reversible transfer of electrons from reduced ferredoxins to NAD(P).sup.+, ferredoxin:NAD+ oxidoreductase (EC 1.18.1.3) and ferredoxin:NADP+ oxidoreductase (FNR, EC 1.18.1.2). Ferredoxin:NADP+ oxidoreductase (FNR, EC 1.18.1.2) has a noncovalently bound FAD cofactor that facilitates the reversible transfer of electrons from NADPH to low-potential acceptors such as ferredoxins or flavodoxins (Blaschkowski et al., Eur. J. Biochem. 123:563-569 (1982); Fujii et al., 1977). The Helicobacter pylori FNR, encoded by HP1164 (fqrB), is coupled to the activity of pyruvate:ferredoxin oxidoreductase (PFOR) resulting in the pyruvate-dependent production of NADPH (St et al. 2007). An analogous enzyme is found in Campylobacter jejuni (St et al. 2007). A ferredoxin:NADP+ oxidoreductase enzyme is encoded in the E. coli genome by fpr (Bianchi et al. 1993). Ferredoxin:NAD+ oxidoreductase utilizes reduced ferredoxin to generate NADH from NAD+. In several organisms, including E. coli, this enzyme is a component of multifunctional dioxygenase enzyme complexes. The ferredoxin:NAD+ oxidoreductase of E. coli, encoded by hcaD, is a component of the 3-phenylproppionate dioxygenase system involved in involved in aromatic acid utilization (Diaz et al. 1998). NADH:ferredoxin reductase activity was detected in cell extracts of Hydrogenobacter thermophilus strain TK-6, although a gene with this activity has not yet been indicated (Yoon et al. 2006). NADP oxidoreductase of C. kluyveri, encoded by nfnAB, catalyzes the concomitant reduction of ferredoxin and NAD+ with two equivalents of NADPH (Wang et al, J Bacteriol 192: 5115-5123 (2010)). Finally, the energy-conserving membrane-associated Rnf-type proteins (Seedorf et al., Proc. Natl. Acad. Sci. U.S.A. 105:2128-2133 (2008); Herrmann et al., J. Bacteriol. 190:784-791 (2008)) provide a means to generate NADH or NADPH from reduced ferredoxin. Additional ferredoxin:NAD(P)+ oxidoreductases have been annotated in Clostridium carboxydivorans P7 and Clostridium ljungdahli.
TABLE-US-00090 TABLE 88 Protein GenBank ID GI Number Organism HP1164 NP_207955.1 15645778 Helicobacter pylori RPA3954 CAE29395.1 39650872 Rhodopseudomonas palustris fpr BAH29712.1 225320633 Hydrogenobacter thermophilus yumC NP_391091.2 255767736 Bacillus subtilis CJE0663 AAW35824.1 57167045 Campylobacter jejuni fpr P28861.4 399486 Escherichia coli hcaD AAC75595.1 1788892 Escherichia coli LOC100282643 NP_001149023.1 226497434 Zea mays NfnA YP_001393861.1 153953096 Clostridium kluyveri NfnB YP_001393862.1 153953097 Clostridium kluyveri RnfC EDK33306.1 146346770 Clostridium kluyveri RnfD EDK33307.1 146346771 Clostridium kluyveri RnfG EDK33308.1 146346772 Clostridium kluyveri RnfE EDK33309.1 146346773 Clostridium kluyveri RnfA EDK33310.1 146346774 Clostridium kluyveri RnfB EDK33311.1 146346775 Clostridium kluyveri CcarbDRAFT_2639 ZP_05392639.1 255525707 Clostridium carboxidivorans P7 CcarbDRAFT_2638 ZP_05392638.1 255525706 Clostridium carboxidivorans P7 CcarbDRAFT_2636 ZP_05392636.1 255525704 Clostridium carboxidivorans P7 CcarbDRAFT_5060 ZP_05395060.1 255528241 Clostridium carboxidivorans P7 CcarbDRAFT_2450 ZP_05392450.1 255525514 Clostridium carboxidivorans P7 CcarbDRAFT_1084 ZP_05391084.1 255524124 Clostridium carboxidivorans P7 CLJU_c11410 ADK14209.1 300434442 Clostridium ljungdahli (RnfB) CLJU_c11400 ADK14208.1 300434441 Clostridium ljungdahli (RnfA) CLJU_c11390 ADK14207.1 300434440 Clostridium ljungdahli (RnfE) CLJU_c11380 ADK14206.1 300434439 Clostridium ljungdahli (RnfG) CLJU_c11370 ADK14205.1 300434438 Clostridium ljungdahli (RnfD) CLJU_c11360 ADK14204.1 300434437 Clostridium ljungdahli (RnfC)
[0532] Ferredoxins are small acidic proteins containing one or more iron-sulfur clusters that function as intracellular electron carriers with a low reduction potential. Reduced ferredoxins donate electrons to Fe-dependent enzymes such as ferredoxin-NADP+ oxidoreductase, pyruvate:ferredoxin oxidoreductase (PFOR) and 2-oxoglutarate:ferredoxin oxidoreductase (OFOR). The H. thermophilus gene fdx1 encodes a [4Fe-4S]-type ferredoxin that is required for the reversible carboxylation of 2-oxoglutarate and pyruvate by OFOR and PFOR, respectively (Yamamoto et al., Extremophiles 14:79-85 (2010)). The ferredoxin associated with the Sulfolobus solfataricus 2-oxoacid:ferredoxin reductase is a monomeric dicluster [3Fe-4S][4Fe-4S] type ferredoxin (Park et al. 2006). While the gene associated with this protein has not been fully sequenced, the N-terminal domain shares 93% homology with the zfx ferredoxin from S. acidocaldarius. The E. coli genome encodes a soluble ferredoxin of unknown physiological function, fdx. Some evidence indicates that this protein can function in iron-sulfur cluster assembly (Takahashi and Nakamura, 1999). Additional ferredoxin proteins have been characterized in Helicobacter pylori (Mukhopadhyay et al. 2003) and Campylobacter jejuni (van Vliet et al. 2001). A 2Fe-2S ferredoxin from Clostridium pasteurianum has been cloned and expressed in E. coli (Fujinaga and Meyer, Biochemical and Biophysical Research Communications, 192(3): (1993)). Acetogenic bacteria such as Moorella thermoacetica, Clostridium carboxidivorans P7, Clostridium ljungdahli and Rhodospirillum rubrum are predicted to encode several ferredoxins, listed in the table below.
TABLE-US-00091 TABLE 89 Protein GenBank ID GI Number Organism fdx1 BAE02673.1 68163284 Hydrogenobacter thermophilus M11214.1 AAA83524.1 144806 Clostridium pasteurianum Zfx AAY79867.1 68566938 Sulfolobus acidocalarius Fdx AAC75578.1 1788874 Escherichia coli hp_0277 AAD07340.1 2313367 Helicobacter pylori fdxA CAL34484.1 112359698 Campylobacter jejuni Moth_0061 ABC18400.1 83571848 Moorella thermoacetica Moth_1200 ABC19514.1 83572962 Moorella thermoacetica Moth_1888 ABC20188.1 83573636 Moorella thermoacetica Moth_2112 ABC20404.1 83573852 Moorella thermoacetica Moth_1037 ABC19351.1 83572799 Moorella thermoacetica CcarbDRAFT_4383 ZP_05394383.1 255527515 Clostridium carboxidivorans P7 CcarbDRAFT_2958 ZP_05392958.1 255526034 Clostridium carboxidivorans P7 CcarbDRAFT_2281 ZP_05392281.1 255525342 Clostridium carboxidivorans P7 CcarbDRAFT_5296 ZP_05395295.1 255528511 Clostridium carboxidivorans P7 CcarbDRAFT_1615 ZP_05391615.1 255524662 Clostridium carboxidivorans P7 CcarbDRAFT_1304 ZP_05391304.1 255524347 Clostridium carboxidivorans P7 cooF AAG29808.1 11095245 Carboxydothermus hydrogenoformans fdxN CAA35699.1 46143 Rhodobacter capsulatus Rru_A2264 ABC23064.1 83576513 Rhodospirillum rubrum Rru_A1916 ABC22716.1 83576165 Rhodospirillum rubrum Rru_A2026 ABC22826.1 83576275 Rhodospirillum rubrum cooF AAC45122.1 1498747 Rhodospirillum rubrum fdxN AAA26460.1 152605 Rhodospirillum rubrum Alvin_2884 ADC63789.1 288897953 Allochromatium vinosum DSM 180 fdx YP_002801146.1 226946073 Azotobacter vinelandii DJ CKL_3790 YP_001397146.1 153956381 Clostridium kluyveri DSM 555 fer1 NP_949965.1 39937689 Rhodopseudomonas palustris CGA009 fdx CAA12251.1 3724172 Thauera aromatica CHY_2405 YP_361202.1 78044690 Carboxydothermus hydrogenoformans fer YP_359966.1 78045103 Carboxydothermus hydrogenoformans fer AAC83945.1 1146198 Bacillus subtilis fdx1 NP_249053.1 15595559 Pseudomonas aeruginosa PA01 yfhL AP_003148.1 89109368 Escherichia coli K-12 CLJU_c00930 ADK13195.1 300433428 Clostridium ljungdahli CLJU_c00010 ADK13115.1 300433348 Clostridium ljungdahli CLJU_c01820 ADK13272.1 300433505 Clostridium ljungdahli CLJU_c17980 ADK14861.1 300435094 Clostridium ljungdahli CLJU_c17970 ADK14860.1 300435093 Clostridium ljungdahli CLJU_c22510 ADK15311.1 300435544 Clostridium ljungdahli CLJU_c26680 ADK15726.1 300435959 Clostridium ljungdahli CLJU_c29400 ADK15988.1 300436221 Clostridium ljungdahli
[0533] Succinyl-CoA transferase catalyzes the conversion of succinyl-CoA to succinate while transferring the CoA moiety to a CoA acceptor molecule. Many transferases have broad specificity and can utilize CoA acceptors as diverse as acetate, succinate, propionate, butyrate, 2-methylacetoacetate, 3-ketohexanoate, 3-ketopentanoate, valerate, crotonate, 3-mercaptopropionate, propionate, vinylacetate, and butyrate, among others.
[0534] The conversion of succinate to succinyl-CoA can be carried by a transferase which does not require the direct consumption of an ATP or GTP. This type of reaction is common in a number of organisms. The conversion of succinate to succinyl-CoA can also be catalyzed by succinyl-CoA:Acetyl-CoA transferase. The gene product of cat1 of Clostridium kluyveri has been shown to exhibit succinyl-CoA: acetyl-CoA transferase activity (Sohling and Gottschalk, J. Bacteriol. 178:871-880 (1996)). In addition, the activity is present in Trichomonas vaginalis (van Grinsven et al. 2008) and Trypanosoma brucei (Riviere et al. 2004). The succinyl-CoA:acetate CoA-transferase from Acetobacter aceti, encoded by aarC, replaces succinyl-CoA synthetase in a variant TCA cycle (Mullins et al. 2008). Similar succinyl-CoA transferase activities are also present in Trichomonas vaginalis (van Grinsven et al. 2008), Trypanosoma brucei (Riviere et al. 2004) and Clostridium kluyveri (Sohling and Gottschalk, 1996c). The beta-ketoadipate:succinyl-CoA transferase encoded by pcaI and pcaJ in Pseudomonas putida is yet another candidate (Kaschabek et al. 2002). The aforementioned proteins are identified below.
TABLE-US-00092 TABLE 90 Protein GenBank ID GI Number Organism cat1 P38946.1 729048 Clostridium kluyveri TVAG_395550 XP_001330176 123975034 Trichomonas vaginalis G3 Tb11.02.0290 XP_828352 71754875 Trypanosoma brucei pcaI AAN69545.1 24985644 Pseudomonas putida pcaJ NP_746082.1 26990657 Pseudomonas putida aarC ACD85596.1 189233555 Acetobacter aceti
[0535] An additional exemplary transferase that converts succinate to succinyl-CoA while converting a 3-ketoacyl-CoA to a 3-ketoacid is succinyl-CoA:3:ketoacid-CoA transferase (EC 2.8.3.5). Exemplary succinyl-CoA:3:ketoacid-CoA transferases are present in Helicobacter pylori (Corthesy-Theulaz et al. 1997), Bacillus subtilis, and Homo sapiens (Fukao et al. 2000; Tanaka et al. 2002). The aforementioned proteins are identified below.
TABLE-US-00093 TABLE 91 Protein GenBank ID GI Number Organism HPAG1_0676 YP_627417 108563101 Helicobacter pylori HPAG1_0677 YP_627418 108563102 Helicobacter pylori ScoA NP_391778 16080950 Bacillus subtilis ScoB NP_391777 16080949 Bacillus subtilis OXCT1 NP_000427 4557817 Homo sapiens OXCT2 NP_071403 11545841 Homo sapiens
[0536] Converting succinate to succinyl-CoA by succinyl-CoA:3:ketoacid-CoA transferase requires the simultaneous conversion of a 3-ketoacyl-CoA such as acetoacetyl-CoA to a 3-ketoacid such as acetoacetate. Conversion of a 3-ketoacid back to a 3-ketoacyl-CoA can be catalyzed by an acetoacetyl-CoA:acetate:CoA transferase. Acetoacetyl-CoA:acetate:CoA transferase converts acetoacetyl-CoA and acetate to acetoacetate and acetyl-CoA, or vice versa. Exemplary enzymes include the gene products of atoAD from E. coli (Hanai et al., Appl Environ Microbiol 73:7814-7818 (2007), ctfAB from C. acetobutylicum (Jojima et al., Appl Microbiol Biotechnol 77:1219-1224 (2008), and ctfAB from Clostridium saccharoperhutylacetonicum (Kosaka et al., Biosci. Biotechnol Biochem. 71:58-68 (2007)) are shown below.
TABLE-US-00094 TABLE 92 Protein GenBank ID GI Number Organism AtoA NP_416726.1 2492994 Escherichia coli AtoD NP_416725.1 2492990 Escherichia coli CtfA NP_149326.1 15004866 Clostridium acetobutylicum CtfB NP_149327.1 15004867 Clostridium acetobutylicum CtfA AAP42564.1 31075384 Clostridium saccharoperbutylacetonicum CtfB AAP42565.1 31075385 Clostridium saccharoperbutylacetonicum
[0537] Yet another possible CoA acceptor is benzylsuccinate. Succinyl-CoA:(R)-Benzylsuccinate CoA-Transferase functions as part of an anaerobic degradation pathway for toluene in organisms such as Thauera aromatica (Leutwein and Heider, J. Bact. 183(14) 4288-4295 (2001)). Homologs can be found in Azoarcus sp. T, Aromatoleum aromaticum EbN1, and Geobacter metallireducens GS-15. The aforementioned proteins are identified below.
TABLE-US-00095 TABLE 93 Protein GenBank ID GI Number Organism bbsE AAF89840 9622535 Thauera aromatica Bbsf AAF89841 9622536 Thauera aromatica bbsE AAU45405.1 52421824 Azoarcus sp. T bbsF AAU45406.1 52421825 Azoarcus sp. T bbsE YP_158075.1 56476486 Aromatoleum aromaticum EbN1 bbsF YP_158074.1 56476485 Aromatoleum aromaticum EbN1 Gmet_1521 YP_384480.1 78222733 Geobacter metallireducens GS-15 Gmet_1522 YP_384481.1 78222734 Geobacter metallireducens GS-15
[0538] Additionally, ygfH encodes a propionyl CoA:succinate CoA transferase in E. coli (Haller et al., Biochemistry, 39(16) 4622-4629). Close homologs can be found in, for example, Citrobacter youngae ATCC 29220, Salmonella enterica subsp. arizonae serovar, and Yersinia intermedia ATCC 29909. The aforementioned proteins are identified below.
TABLE-US-00096 TABLE 94 Protein GenBank ID GI Number Organism ygfH NP_417395.1 16130821 Escherichia coli str. K-12 substr. MG1655 CIT292_04485 ZP_03838384.1 227334728 Citrobacter youngae ATCC 29220 SARI_04582 YP_001573497.1 161506385 Salmonella enterica subsp. arizonae serovar yinte0001_14430 ZP_04635364.1 238791727 Yersinia intermedia ATCC 29909
[0539] Citrate lyase (EC 4.1.3.6) catalyzes a series of reactions resulting in the cleavage of citrate to acetate and oxaloacetate. The enzyme is active under anaerobic conditions and is composed of three subunits: an acyl-carrier protein (ACP, gamma), an ACP transferase (alpha), and a acyl lyase (beta). Enzyme activation uses covalent binding and acetylation of an unusual prosthetic group, 2'-(5''-phosphoribosyl)-3-'-dephospho-CoA, which is similar in structure to acetyl-CoA. Acylation is catalyzed by CitC, a citrate lyase synthetase. Two additional proteins, CitG and CitX, are used to convert the apo enzyme into the active holo enzyme (Schneider et al., Biochemistry 39:9438-9450 (2000)). Wild type E. coli does not have citrate lyase activity; however, mutants deficient in molybdenum cofactor synthesis have an active citrate lyase (Clark, FEMS Microbiol. Lett. 55:245-249 (1990)). The E. coli enzyme is encoded by citEFD and the citrate lyase synthetase is encoded by citC (Nilekani and SivaRaman, Biochemistry 22:4657-4663 (1983)). The Leuconostoc mesenteroides citrate lyase has been cloned, characterized and expressed in E. coli (Bekal et al., J. Bacteriol. 180:647-654 (1998)). Citrate lyase enzymes have also been identified in enterobacteria that utilize citrate as a carbon and energy source, including Salmonella typhimurium and Klebsiella pneumoniae (Bott, Arch. Microbiol. 167: 78-88 (1997); Bott and Dimroth, Mol. Microbiol. 14:347-356 (1994)). The aforementioned proteins are tabulated below.
TABLE-US-00097 TABLE 95 Protein GenBank ID GI Number Organism citF AAC73716.1 1786832 Escherichia coli Cite AAC73717.2 87081764 Escherichia coli citD AAC73718.1 1786834 Escherichia coli citC AAC73719.2 87081765 Escherichia coli citG AAC73714.1 1786830 Escherichia coli citX AAC73715.1 1786831 Escherichia coli citF CAA71633.1 2842397 Leuconostoc mesenteroides Cite CAA71632.1 2842396 Leuconostoc mesenteroides citD CAA71635.1 2842395 Leuconostoc mesenteroides citC CAA71636.1 3413797 Leuconostoc mesenteroides citG CAA71634.1 2842398 Leuconostoc mesenteroides citX CAA71634.1 2842398 Leuconostoc mesenteroides citF NP_459613.1 16763998 Salmonella typhimurium cite AAL19573.1 16419133 Salmonella typhimurium citD NP_459064.1 16763449 Salmonella typhimurium citC NP_459616.1 16764001 Salmonella typhimurium citG NP_459611.1 16763996 Salmonella typhimurium citX NP_459612.1 16763997 Salmonella typhimurium citF CAA56217.1 565619 Klebsiella pneumoniae cite CAA56216.1 565618 Klebsiella pneumoniae citD CAA56215.1 565617 Klebsiella pneumoniae citC BAH66541.1 238774045 Klebsiella pneumoniae citG CAA56218.1 565620 Klebsiella pneumoniae citX AAL60463.1 18140907 Klebsiella pneumoniae
[0540] Acetate kinase (EC 2.7.2.1) catalyzes the reversible ATP-dependent phosphorylation of acetate to acetylphosphate. Exemplary acetate kinase enzymes have been characterized in many organisms including E. coli, Clostridium acetobutylicum and Methanosarcina thermophila (Ingram-Smith et al., J. Bacteriol. 187:2386-2394 (2005); Fox and Roseman, J. Biol. Chem. 261:13487-13497 (1986); Winzer et al., Microbioloy 143 (Pt 10):3279-3286 (1997)). Acetate kinase activity has also been demonstrated in the gene product of E. coli purT (Marolewski et al., Biochemistry 33:2531-2537 (1994). Some butyrate kinase enzymes (EC 2.7.2.7), for example buk1 and buk2 from Clostridium acetobutylicum, also accept acetate as a substrate (Hartmanis, M. G., J. Biol. Chem. 262:617-621 (1987)).
TABLE-US-00098 TABLE 96 Protein GenBank ID GI Number Organism ackA NP_416799.1 16130231 Escherichia coli Ack AAB18301.1 1491790 Clostridium acetobutylicum Ack AAA72042.1 349834 Methanosarcina thermophila purT AAC74919.1 1788155 Escherichia coli buk1 NP_349675 15896326 Clostridium acetobutylicum buk2 Q97II1 20137415 Clostridium acetobutylicum
[0541] The formation of acetyl-CoA from acetylphosphate is catalyzed by phosphotransacetylase (EC 2.3.1.8). The pta gene from E. coli encodes an enzyme that reversibly converts acetyl-CoA into acetyl-phosphate (Suzuki, T., Biochim. Biophys. Acta 191:559-569 (969)). Additional acetyltransferase enzymes have been characterized in Bacillus subtilis (Rado and Hoch, Biochim. Biophys. Acta 321:114-125 (1973), Clostridium kluyveri (Stadtman, E., Methods Enzymol. 1:5896-599 (1955), and Thermotoga maritima (Bock et al., J. Bacteriol. 181:1861-1867 (1999)). This reaction is also catalyzed by some phosphotranbutyrylase enzymes (EC 2.3.1.19) including the ptb gene products from Clostridium acetobutylicum (Wiesenborn et al., App. Environ. Microbiol. 55:317-322 (1989); Walter et al., Gene 134:107-111 (1993)). Additional ptb genes are found in butyrate-producing bacterium L2-50 (Louis et al., J. Bacteriol. 186:2099-2106 (2004) and Bacillus megaterium (Vazquez et al., Curr. Microbiol. 42:345-349 (2001).
TABLE-US-00099 TABLE 97 Protein GenBank ID GI Number Organism Pta NP_416800.1 71152910 Escherichia coli Pta P39646 730415 Bacillus subtilis Pta A5N801 146346896 Clostridium kluyveri Pta Q9X0L4 6685776 Thermotoga maritima Ptb NP_349676 34540484 Clostridium acetobutylicum Ptb AAR19757.1 38425288 butyrate-producing bacterium L2-50 Ptb CAC07932.1 10046659 Bacillus megaterium
[0542] The acylation of acetate to acetyl-CoA is catalyzed by enzymes with acetyl-CoA synthetase activity. Two enzymes that catalyze this reaction are AMP-forming acetyl-CoA synthetase (EC 6.2.1.1) and ADP-forming acetyl-CoA synthetase (EC 6.2.1.13). AMP-forming acetyl-CoA synthetase (ACS) is the predominant enzyme for activation of acetate to acetyl-CoA. Exemplary ACS enzymes are found in E. coli (Brown et al., J. Gen. Microbiol. 102:327-336 (1977)), Ralstonia eutropha (Priefert and Steinbuchel, J. Bacteriol. 174:6590-6599 (1992)), Methanothermobacter thermautotrophicus (Ingram-Smith and Smith, Archaea 2:95-107 (2007)), Salmonella enterica (Gulick et al., Biochemistry 42:2866-2873 (2003)) and Saccharomyces cerevisiae (Jogl and Tong, Biochemistry 43:1425-1431 (2004)). ADP-forming acetyl-CoA synthetases are reversible enzymes with a generally broad substrate range (Musfeldt and Schonheit, J. Bacteriol. 184:636-644 (2002)). Two isozymes of ADP-forming acetyl-CoA synthetases are encoded in the Archaeoglobus fulgidus genome by are encoded by AF1211 and AF1983 (Musfeldt and Schonheit, supra (2002)). The enzyme from Haloarcula marismortui (annotated as a succinyl-CoA synthetase) also accepts acetate as a substrate and reversibility of the enzyme was demonstrated (Brasen and Schonheit, Arch. Microbiol. 182:277-287 (2004)). The ACD encoded by PAE3250 from hyperthermophilic crenarchaeon Pyrobaculum aerophilum showed the broadest substrate range of all characterized ACDs, reacting with acetate, isobutyryl-CoA (preferred substrate) and phenylacetyl-CoA (Brasen and Schonheit, supra (2004)). Directed evolution or engineering can be used to modify this enzyme to operate at the physiological temperature of the host organism. The enzymes from A. fulgidus, H. marismortui and P. aerophilum have all been cloned, functionally expressed, and characterized in E. coli (Brasen and Schonheit, supra (2004); Musfeldt and Schonheit, supra (2002)). Additional candidates include the succinyl-CoA synthetase encoded by sucCD in E. coli (Buck et al., Biochemistry 24:6245-6252 (1985)) and the acyl-CoA ligase from Pseudomonas putida (Fernandez-Valverde et al., Appl. Environ. Microbiol. 59:1149-1154 (1993)). The aforementioned proteins are tabulated below.
TABLE-US-00100 TABLE 98 Protein GenBank ID GI Number Organism acs AAC77039.1 1790505 Escherichia coli acoE AAA21945.1 141890 Ralstonia eutropha acs1 ABC87079.1 86169671 Methanothermobacter thermautotrophicus acs1 AAL23099.1 16422835 Salmonella enterica ACS1 Q01574.2 257050994 Saccharomyces cerevisiae AF1211 NP_070039.1 11498810 Archaeoglobus fulgidus AF1983 NP_070807.1 11499565 Archaeoglobus fulgidus scs YP_135572.1 55377722 Haloarcula marismortui PAE3250 NP_560604.1 18313937 Pyrobaculum aerophilum str. IM2 sucC NP_415256.1 16128703 Escherichia coli sucD AAC73823.1 1786949 Escherichia coli paaF AAC24333.2 22711873 Pseudomonas putida
[0543] The product yields per C-mol of substrate of microbial cells synthesizing reduced fermentation products such as 1,3-butanediol, are limited by insufficient reducing equivalents in the carbohydrate feedstock. Reducing equivalents, or electrons, can be extracted from synthesis gas components such as CO and H2 using carbon monoxide dehydrogenase (CODH) and hydrogenase enzymes, respectively. The reducing equivalents are then passed to acceptors such as oxidized ferredoxins, oxidized quinones, oxidized cytochromes, NAD(P)+, water, or hydrogen peroxide to form reduced ferredoxin, reduced quinones, reduced cytochromes, NAD(P)H, H2, or water, respectively. Reduced ferredoxin and NAD(P)H are particularly useful as they can serve as redox carriers for various Wood-Ljungdahl pathway and reductive TCA cycle enzymes.
[0544] Here, we show specific examples of how additional redox availability from CO and/or H2 can improve the yields of reduced products such as 1,3-butanediol. The maximum theoretical yield to produce 1,3-butanediol from glucose is 1.09 mole 1,3-butanediol per mole of glucose under aerobic conditions via the pathways shown in FIG. 8B or 1.09 mole 1,3-butanediol per mole of glucose under aerobic conditions via the pathways shown in FIG. 9B. Gasification of glucose to form syngas will result in the maximum theoretical yield of 1.09 moles of 1,3-BDO per mole of glucose consumed, assuming that 6 moles of CO and 6 moles of H2 are obtained from glucose.
6CO+6H.sub.2.fwdarw.1.091C.sub.4H.sub.10O.sub.2+1.636CO.sub.2+0.545H.sub- .2
[0545] When additional reducing equivalents are provided, the yield can be improved to 2 mol/mol glucose.
1C.sub.6H.sub.12O.sub.6+2CO+8H.sub.2.fwdarw.2C.sub.4H.sub.10O.sub.2+4H.s- ub.2O
[0546] When both feedstocks of sugar and syngas are available, the syngas components CO and H.sub.2 can be utilized together or separately (from any source) to generate reducing equivalents by employing the hydrogenase and/or CO dehydrogenase. The reducing equivalents generated from CO and/or hydrogen will be utilized to power the glucose to 1,3-butanediol production pathways. Theoretically, all carbons in glucose will be conserved, thus resulting in a maximal theoretical yield to produce 1,3-butanediol from glucose.
[0547] As shown in above example, a combined feedstock strategy where syngas is combined with a sugar-based feedstock or other carbon substrate can greatly improve the theoretical yields. In this co-feeding approach, syngas components H2 and/or CO can be utilized by the hydrogenase and CO dehydrogenase to generate reducing equivalents, that can be used to power chemical production pathways in which the carbons from sugar or other carbon substrates will be maximally conserved and the theoretical yields improved. Such improvements provide environmental and economic benefits and greatly enhance sustainable chemical production.
[0548] As shown in above example, a combined feedstock strategy where syngas is combined with a sugar-based feedstock or other carbon substrate can greatly improve the theoretical yields. In this co-feeding approach, syngas components H2 and CO can be utilized by the hydrogenase and CO dehydrogenase to generate reducing equivalents, that can be used to power chemical production pathways in which the carbons from sugar or other carbon substrates will be maximally conserved and the theoretical yields improved. In case of 1,3-butanediol production from glucose or sugar, the theoretical yields improve from 1.09 mol 1,3-butanediol per mol of glucose to 2 mol 1,3-butanediol per mol of glucose. Such improvements provide environmental and economic benefits and greatly enhance sustainable chemical production.
[0549] Herein below the enzymes and the corresponding genes used for extracting redox from syngas components are described. CODH is a reversible enzyme that interconverts CO and CO2 at the expense or gain of electrons. The natural physiological role of the CODH in ACS/CODH complexes is to convert CO2 to CO for incorporation into acetyl-CoA by acetyl-CoA synthase. Nevertheless, such CODH enzymes are suitable for the extraction of reducing equivalents from CO due to the reversible nature of such enzymes. Expressing such CODH enzymes in the absence of ACS allows them to operate in the direction opposite to their natural physiological role (i.e., CO oxidation).
[0550] In M. thermoacetica, C. hydrogenoformans, C. carboxidivorans P7, and several other organisms, additional CODH encoding genes are located outside of the ACS/CODH operons. These enzymes provide a means for extracting electrons (or reducing equivalents) from the conversion of carbon monoxide to carbon dioxide. The M. thermoacetica gene (GenBank Accession Number: YP.sub.--430813) is expressed by itself in an operon and is believed to transfer electrons from CO to an external mediator like ferredoxin in a "Ping-pong" reaction. The reduced mediator then couples to other reduced nicolinamide adenine dinucleotide phosphate (NAD(P)H) carriers or ferredoxin-dependent cellular processes (Ragsdale, Annals of the New York Academy of Sciences 1125: 129-136 (2008)). The genes encoding the C. hydrogenoformans CODH-II and CooF, a neighboring protein, were cloned and sequenced (Gonzalez and Robb, FEMS Microbiol Lett. 191:243-247 (2000)). The resulting complex was membrane-bound, although cytoplasmic fractions of CODH-II were shown to catalyze the formation of NADPH suggesting an anabolic role (Svetlitchnyi et al., J Bacteriol. 183:5134-5144 (2001)). The crystal structure of the CODH-II is also available (Dobbek et al., Science 293:1281-1285 (2001)). Similar ACS-free CODH enzymes can be found in a diverse array of organisms including Geobacter metallireducens GS-15, Chlorobium phaeobacteroides DSM 266, Clostridium cellulolyticum H10, Desulfovibrio desulfuricans subsp. desulfuricans str. ATCC 27774, Pelobacter carbinolicus DSM 2380, and Campylobacter curvus 525.92.
TABLE-US-00101 TABLE 99 Protein GenBank ID GI Number Organism CODH (putative) YP_430813 83590804 Moorella thermoacetica CODH-II (CooS-II) YP_358957 78044574 Carboxydothermus hydrogenoformans CooF YP_358958 78045112 Carboxydothermus hydrogenoformans CODH (putative) ZP_05390164.1 255523193 Clostridium carboxidivorans P7 CcarbDRAFT_0341 ZP_05390341.1 255523371 Clostridium carboxidivorans P7 CcarbDRAFT_1756 ZP_05391756.1 255524806 Clostridium carboxidivorans P7 CcarbDRAFT_2944 ZP_05392944.1 255526020 Clostridium carboxidivorans P7 CODH YP_384856.1 78223109 Geobacter metallireducens GS-15 Cpha266_0148 YP_910642.1 119355998 Chlorobium (cytochrome c) phaeobacteroides DSM 266 Cpha266_0149 YP_910643.1 119355999 Chlorobium (CODH) phaeobacteroides DSM 266 Ccel_0438 YP_002504800.1 220927891 Clostridium cellulolyticum H10 Ddes_0382 YP_002478973.1 220903661 Desulfovibrio desulfuricans subsp. (CODH) desulfuricans str. ATCC 27774 Ddes_0381 YP_002478972.1 220903660 Desulfovibrio desulfuricans subsp. (CooC) desulfuricans str. ATCC 27774 Pcar_0057 YP_355490.1 7791767 Pelobacter carbinolicus DSM (CODH) 2380 Pcar_0058 YP_355491.1 7791766 Pelobacter carbinolicus DSM (CooC) 2380 Pcar_0058 YP_355492.1 7791765 Pelobacter carbinolicus DSM (HypA) 2380 CooS (CODH) YP_001407343.1 154175407 Campylobacter curvus 525.92 CLJU_c09110 ADK13979.1 300434212 Clostridium ljungdahli CLJU_c09100 ADK13978.1 300434211 Clostridium ljungdahli CLJU_c09090 ADK13977.1 300434210 Clostridium ljungdahli
[0551] In some cases, hydrogenase encoding genes are located adjacent to a CODH. In Rhodospirillum rubrum, the encoded CODH/hydrogenase proteins form a membrane-bound enzyme complex that has been indicated to be a site where energy, in the form of a proton gradient, is generated from the conversion of CO and H.sub.2O to CO.sub.2 and H.sub.2 (Fox et al., J Bacteriol. 178:6200-6208 (1996)). The CODH-I of C. hydrogenoformans and its adjacent genes have been proposed to catalyze a similar functional role based on their similarity to the R. rubrum CODH/hydrogenase gene cluster (Wu et al., PLoS Genet. 1:e65 (2005)). The C. hydrogenoformans CODH-I was also shown to exhibit intense CO oxidation and CO.sub.2 reduction activities when linked to an electrode (Parkin et al., J Am. Chem. Soc. 129:10328-10329 (2007)). The protein sequences of exemplary CODH and hydrogenase genes can be identified by the following GenBank accession numbers.
TABLE-US-00102 TABLE 100 GI Protein GenBank ID Number Organism CODH-I (CooS-I) YP_360644 78043418 Carboxydothermus hydrogenoformans CooF YP_360645 78044791 Carboxydothermus hydrogenoformans HypA YP_360646 78044340 Carboxydothermus hydrogenoformans CooH YP_360647 78043871 Carboxydothermus hydrogenoformans CooU YP_360648 78044023 Carboxydothermus hydrogenoformans CooX YP_360649 78043124 Carboxydothermus hydrogenoformans CooL YP_360650 78043938 Carboxydothermus hydrogenoformans CooK YP_360651 78044700 Carboxydothermus hydrogenoformans CooM YP_360652 78043942 Carboxydothermus hydrogenoformans CooC YP_360654.1 78043296 Carboxydothermus hydrogenoformans CooA-1 YP_360655.1 78044021 Carboxydothermus hydrogenoformans CooL AAC45118 1515468 Rhodospirillum rubrum CooX AAC45119 1515469 Rhodospirillum rubrum CooU AAC45120 1515470 Rhodospirillum rubrum CooH AAC45121 1498746 Rhodospirillum rubrum CooF AAC45122 1498747 Rhodospirillum rubrum CODH (CooS) AAC45123 1498748 Rhodospirillum rubrum CooC AAC45124 1498749 Rhodospirillum rubrum CooT AAC45125 1498750 Rhodospirillum rubrum CooJ AAC45126 1498751 Rhodospirillum rubrum
[0552] Native to E. coli and other enteric bacteria are multiple genes encoding up to four hydrogenases (Sawers, G., Antonie Van Leeuwenhoek 66:57-88 (1994); Sawers et al., J Bacteriol. 164:1324-1331 (1985); Sawers and Boxer, Eur. J Biochem. 156:265-275 (1986); Sawers et al., J Bacteriol. 168:398-404 (1986)). Given the multiplicity of enzyme activities, E. coli or another host organism can provide sufficient hydrogenase activity to split incoming molecular hydrogen and reduce the corresponding acceptor. E. coli possesses two uptake hydrogenases, Hyd-1 and Hyd-2, encoded by the hyaABCDEF and hybOABCDEFG gene clusters, respectively (Lukey et al., How E. coli is equipped to oxidize hydrogen under different redox conditions, J Biol Chem published online Nov. 16, 2009). Hyd-1 is oxygen-tolerant, irreversible, and is coupled to quinone reduction via the hyaC cytochrome. Hyd-2 is sensitive to O.sub.2, reversible, and transfers electrons to the periplasmic ferredoxin hybA which, in turn, reduces a quinone via the hybB integral membrane protein. Reduced quinones can serve as the source of electrons for fumarate reductase in the reductive branch of the TCA cycle. Reduced ferredoxins can be used by enzymes such as NAD(P)H:ferredoxin oxidoreductases to generate NADPH or NADH. They can alternatively be used as the electron donor for reactions such as pyruvate ferredoxin oxidoreductase, AKG ferredoxin oxidoreductase, and 5,10-methylene-H4folate reductase.
TABLE-US-00103 TABLE 101 Protein GenBank ID GI Number Organism HyaA AAC74057.1 1787206 Escherichia coli HyaB AAC74058.1 1787207 Escherichia coli HyaC AAC74059.1 1787208 Escherichia coli HyaD AAC74060.1 1787209 Escherichia coli HyaE AAC74061.1 1787210 Escherichia coli HyaF AAC74062.1 1787211 Escherichia coli
TABLE-US-00104 TABLE 102 Protein GenBank ID GI Number Organism HybO AAC76033.1 1789371 Escherichia coli HybA AAC76032.1 1789370 Escherichia coli HybB AAC76031.1 2367183 Escherichia coli HybC AAC76030.1 1789368 Escherichia coli HybD AAC76029.1 1789367 Escherichia coli HybE AAC76028.1 1789366 Escherichia coli HybF AAC76027.1 1789365 Escherichia coli HybG AAC76026.1 1789364 Escherichia coli
[0553] The hydrogen-lyase systems of E. coli include hydrogenase 3, a membrane-bound enzyme complex using ferredoxin as an acceptor, and hydrogenase 4 that also uses a ferredoxin acceptor. Hydrogenase 3 and 4 are encoded by the hyc and hyf gene clusters, respectively. Hydrogenase 3 has been shown to be a reversible enzyme (Maeda et al., Appl Microbiol Biotechnol 76(5):1035-42 (2007)). Hydrogenase activity in E. coli is also dependent upon the expression of the hyp genes whose corresponding proteins are involved in the assembly of the hydrogenase complexes (Jacobi et al., Arch. Microbiol 158:444-451 (1992); Rangarajan et al., J. Bacteriol. 190:1447-1458 (2008)).
TABLE-US-00105 TABLE 103 Protein GenBank ID GI Number Organism HycA NP_417205 16130632 Escherichia coli HycB NP_417204 16130631 Escherichia coli HycC NP_417203 16130630 Escherichia coli HycD NP_417202 16130629 Escherichia coli HycE NP_417201 16130628 Escherichia coli HycF NP_417200 16130627 Escherichia coli HycG NP_417199 16130626 Escherichia coli HycH NP_417198 16130625 Escherichia coli HycI NP_417197 16130624 Escherichia coli
TABLE-US-00106 TABLE 104 Protein GenBank ID GI Number Organism HyfA NP_416976 90111444 Escherichia coli HyfB NP_416977 16130407 Escherichia coli HyfC NP_416978 90111445 Escherichia coli HyfD NP_416979 16130409 Escherichia coli HyfE NP_416980 16130410 Escherichia coli HyfF NP_416981 16130411 Escherichia coli HyfG NP_416982 16130412 Escherichia coli HyfH NP_416983 16130413 Escherichia coli HyfI NP_416984 16130414 Escherichia coli HyfJ NP_416985 90111446 Escherichia coli HyfR NP_416986 90111447 Escherichia coli
TABLE-US-00107 TABLE 105 Protein GenBank ID GI Number Organism HypA NP_417206 16130633 Escherichia coli HypB NP_417207 16130634 Escherichia coli HypC NP_417208 16130635 Escherichia coli HypD NP_417209 16130636 Escherichia coli HypE NP_417210 226524740 Escherichia coli HypF NP_417192 16130619 Escherichia coli
[0554] The M. thermoacetica hydrogenases are suitable for a host that lacks sufficient endogenous hydrogenase activity. M. thermoacetica can grow with CO2 as the exclusive carbon source indicating that reducing equivalents are extracted from H2 to enable acetyl-CoA synthesis via the Wood-Ljungdahl pathway (Drake, H. L., J. Bacteriol. 150:702-709 (1982); Drake and Daniel, Res. Microbiol. 155:869-883 (2004); Kellum and Drake, J. Bacteriol. 160:466-469 (1984)). M. thermoacetica has homologs to several hyp, hyc, and hyf genes from E. coli. The protein sequences encoded for by these genes are identified by the following GenBank accession numbers.
[0555] Proteins in M. thermoacetica whose genes are homologous to the E. coli hyp genes are shown below.
TABLE-US-00108 TABLE 106 Protein GenBank ID GI Number Organism Moth_2175 YP_431007 83590998 Moorella thermoacetica Moth_2176 YP_431008 83590999 Moorella thermoacetica Moth_2177 YP_431009 83591000 Moorella thermoacetica Moth_2178 YP_431010 83591001 Moorella thermoacetica Moth_2179 YP_431011 83591002 Moorella thermoacetica Moth_2180 YP_431012 83591003 Moorella thermoacetica Moth_2181 YP_431013 83591004 Moorella thermoacetica
[0556] Proteins in M. thermoacetica that are homologous to the E. coli Hydrogenase 3 and/or 4 proteins are listed in the following table.
TABLE-US-00109 TABLE 107 Protein GenBank ID GI Number Organism Moth_2182 YP_431014 83591005 Moorella thermoacetica Moth_2183 YP_431015 83591006 Moorella thermoacetica Moth_2184 YP_431016 83591007 Moorella thermoacetica Moth_2185 YP_431017 83591008 Moorella thermoacetica Moth_2186 YP_431018 83591009 Moorella thermoacetica Moth_2187 YP_431019 83591010 Moorella thermoacetica Moth_2188 YP_431020 83591011 Moorella thermoacetica Moth_2189 YP_431021 83591012 Moorella thermoacetica Moth_2190 YP_431022 83591013 Moorella thermoacetica Moth_2191 YP_431023 83591014 Moorella thermoacetica Moth_2192 YP_431024 83591015 Moorella thermoacetica
[0557] In addition, several gene clusters encoding hydrogenase functionality are present in M. thermoacetica and their corresponding protein sequences are provided below.
TABLE-US-00110 TABLE 108A Protein GenBank ID GI Number Organism Moth_0439 YP_429313 83589304 Moorella thermoacetica Moth_0440 YP_429314 83589305 Moorella thermoacetica Moth_0441 YP_429315 83589306 Moorella thermoacetica Moth_0442 YP_429316 83589307 Moorella thermoacetica Moth_0809 YP_429670 83589661 Moorella thermoacetica Moth_0810 YP_429671 83589662 Moorella thermoacetica Moth_0811 YP_429672 83589663 Moorella thermoacetica Moth_0812 YP_429673 83589664 Moorella thermoacetica Moth_0814 YP_429674 83589665 Moorella thermoacetica Moth_0815 YP_429675 83589666 Moorella thermoacetica Moth_0816 YP_429676 83589667 Moorella thermoacetica Moth_1193 YP_430050 83590041 Moorella thermoacetica Moth_1194 YP_430051 83590042 Moorella thermoacetica Moth_1195 YP_430052 83590043 Moorella thermoacetica Moth_1196 YP_430053 83590044 Moorella thermoacetica Moth_1717 YP_430562 83590553 Moorella thermoacetica Moth_1718 YP_430563 83590554 Moorella thermoacetica Moth_1719 YP_430564 83590555 Moorella thermoacetica Moth_1883 YP_430726 83590717 Moorella thermoacetica Moth_1884 YP_430727 83590718 Moorella thermoacetica Moth_1885 YP_430728 83590719 Moorella thermoacetica Moth_1886 YP_430729 83590720 Moorella thermoacetica Moth_1887 YP_430730 83590721 Moorella thermoacetica Moth_1888 YP_430731 83590722 Moorella thermoacetica Moth_1452 YP_430305 83590296 Moorella thermoacetica Moth_1453 YP_430306 83590297 Moorella thermoacetica Moth_1454 YP_430307 83590298 Moorella thermoacetica
[0558] Genes encoding hydrogenase enzymes from C. ljungdahli are shown below.
TABLE-US-00111 TABLE 108B Protein GenBank ID GI Number Organism CLJU_c20290 ADK15091.1 300435324 Clostridium ljungdahli CLJU_c07030 ADK13773.1 300434006 Clostridium ljungdahli CLJU_c07040 ADK13774.1 300434007 Clostridium ljungdahli CLJU_c07050 ADK13775.1 300434008 Clostridium ljungdahli CLJU_c07060 ADK13776.1 300434009 Clostridium ljungdahli CLJU_c07070 ADK13777.1 300434010 Clostridium ljungdahli CLJU_c07080 ADK13778.1 300434011 Clostridium ljungdahli CLJU_c14730 ADK14541.1 300434774 Clostridium ljungdahli CLJU_c14720 ADK14540.1 300434773 Clostridium ljungdahli CLJU_c14710 ADK14539.1 300434772 Clostridium ljungdahli CLJU_c14700 ADK14538.1 300434771 Clostridium ljungdahli CLJU_c28670 ADK15915.1 300436148 Clostridium ljungdahli CLJU_c28660 ADK15914.1 300436147 Clostridium ljungdahli CLJU_c28650 ADK15913.1 300436146 Clostridium ljungdahli CLJU_c28640 ADK15912.1 300436145 Clostridium ljungdahli
[0559] Ralstonia eutropha H16 uses hydrogen as an energy source with oxygen as a terminal electron acceptor. Its membrane-bound uptake [NiFe]-hydrogenase is an "O2-tolerant" hydrogenase (Cracknell, et al. Proc Nat Acad Sci, 106(49) 20681-20686 (2009)) that is periplasmically-oriented and connected to the respiratory chain via a b-type cytochrome (Schink and Schlegel, Biochim. Biophys. Acta, 567, 315-324 (1979); Bernhard et al., Eur. J. Biochem. 248, 179-186 (1997)). R. eutropha also contains an O.sub.2-tolerant soluble hydrogenase encoded by the Hox operon which is cytoplasmic and directly reduces NAD+ at the expense of hydrogen (Schneider and Schlegel, Biochim. Biophys. Acta 452, 66-80 (1976); Burgdorf, J. Bact. 187(9) 3122-3132 (2005)). Soluble hydrogenase enzymes are additionally present in several other organisms including Geobacter sulfurreducens (Coppi, Microbiology 151, 1239-1254 (2005)), Synechocystis str. PCC 6803 (Germer, J. Biol. Chem., 284(52), 36462-36472 (2009)), and Thiocapsa roseopersicina (Rakhely, Appl. Environ. Microbiol. 70(2) 722-728 (2004)). The Synechocystis enzyme is capable of generating NADPH from hydrogen. Overexpression of both the Hox operon from Synechocystis str. PCC 6803 and the accessory genes encoded by the Hyp operon from Nostoc sp. PCC 7120 led to increased hydrogenase activity compared to expression of the Hox genes alone (Germer, J. Biol. Chem. 284(52), 36462-36472 (2009)).
TABLE-US-00112 TABLE 109 Protein GenBank ID GI Number Organism HoxF NP_942727.1 38637753 Ralstonia eutropha H16 HoxU NP_942728.1 38637754 Ralstonia eutropha H16 HoxY NP_942729.1 38637755 Ralstonia eutropha H16 HoxH NP_942730.1 38637756 Ralstonia eutropha H16 HoxW NP_942731.1 38637757 Ralstonia eutropha H16 HoxI NP_942732.1 38637758 Ralstonia eutropha H16 HoxE NP_953767.1 39997816 Geobacter sulfurreducens HoxF NP_953766.1 39997815 Geobacter sulfurreducens HoxU NP_953765.1 39997814 Geobacter sulfurreducens HoxY NP_953764.1 39997813 Geobacter sulfurreducens HoxH NP_953763.1 39997812 Geobacter sulfurreducens GSU2717 NP_953762.1 39997811 Geobacter sulfurreducens HoxE NP_441418.1 16330690 Synechocystis str. PCC 6803 HoxF NP_441417.1 16330689 Synechocystis str. PCC 6803 Unknown NP_441416.1 16330688 Synechocystis str. PCC function 6803 HoxU NP_441415.1 16330687 Synechocystis str. PCC 6803 HoxY NP_441414.1 16330686 Synechocystis str. PCC 6803 Unknown NP_441413.1 16330685 Synechocystis str. PCC function 6803 Unknown NP_441412.1 16330684 Synechocystis str. PCC function 6803 HoxH NP_441411.1 16330683 Synechocystis str. PCC 6803 HypF NP_484737.1 17228189 Nostoc sp. PCC 7120 HypC NP_484738.1 17228190 Nostoc sp. PCC 7120 HypD NP_484739.1 17228191 Nostoc sp. PCC 7120 Unknown NP_484740.1 17228192 Nostoc sp. PCC 7120 function HypE NP_484741.1 17228193 Nostoc sp. PCC 7120 HypA NP_484742.1 17228194 Nostoc sp. PCC 7120 HypB NP_484743.1 17228195 Nostoc sp. PCC 7120 Hox1E AAP50519.1 37787351 Thiocapsa roseopersicina Hox1F AAP50520.1 37787352 Thiocapsa roseopersicina Hox1U AAP50521.1 37787353 Thiocapsa roseopersicina Hox1Y AAP50522.1 37787354 Thiocapsa roseopersicina Hox1H AAP50523.1 37787355 Thiocapsa roseopersicina
[0560] Several enzymes and the corresponding genes used for fixing carbon dioxide to either pyruvate or phosphoenolpyruvate to form the TCA cycle intermediates, oxaloacetate or malate are described below.
[0561] Carboxylation of phosphoenolpyruvate to oxaloacetate is catalyzed by phosphoenolpyruvate carboxylase. Exemplary PEP carboxylase enzymes are encoded by ppc in E. coli (Kai et al., Arch. Biochem. Biophys. 414:170-179 (2003), ppcA in Methylobacterium extorquens AM1 (Arps et al., J. Bacteriol. 175:3776-3783 (1993), and ppc in Corynebacterium glutamicum (Eikmanns et al., Mol. Gen. Genet. 218:330-339 (1989).
TABLE-US-00113 TABLE 110 Protein GenBank ID GI Number Organism Ppc NP_418391 16131794 Escherichia coli ppcA AAB58883 28572162 Methylobacterium extorquens Ppc ABB53270 80973080 Corynebacterium glutamicum
[0562] An alternative enzyme for converting phosphoenolpyruvate to oxaloacetate is PEP carboxykinase, which simultaneously forms an ATP while carboxylating PEP. In most organisms PEP carboxykinase serves a gluconeogenic function and converts oxaloacetate to PEP at the expense of one ATP. S. cerevisiae is one such organism whose native PEP carboxykinase, PCK1, serves a gluconeogenic role (Valdes-Hevia et al., FEBS Lett. 258:313-316 (1989). E. coli is another such organism, as the role of PEP carboxykinase in producing oxaloacetate is believed to be minor when compared to PEP carboxylase, which does not form ATP, possibly due to the higher K.sub.m for bicarbonate of PEP carboxykinase (Kim et al., Appl. Environ. Microbiol. 70:1238-1241 (2004)). Nevertheless, activity of the native E. coli PEP carboxykinase from PEP towards oxaloacetate has been recently demonstrated in ppc mutants of E. coli K-12 (Kwon et al., J. Microbiol. Biotechnol. 16:1448-1452 (2006)). These strains exhibited no growth defects and had increased succinate production at high NaHCO.sub.3 concentrations. Mutant strains of E. coli can adopt Pck as the dominant CO2-fixing enzyme following adaptive evolution (Zhang et al. 2009). In some organisms, particularly rumen bacteria, PEP carboxykinase is quite efficient in producing oxaloacetate from PEP and generating ATP. Examples of PEP carboxykinase genes that have been cloned into E. coli include those from Mannheimia succiniciproducens (Lee et al., Biotechnol. Bioprocess Eng. 7:95-99 (2002)), Anaerobiospirillum succiniciproducens (Laivenieks et al., Appl. Environ. Microbiol. 63:2273-2280 (1997), and Actinobacillus succinogenes (Kim et al. supra). The PEP carboxykinase enzyme encoded by Haemophilus influenza is effective at forming oxaloacetate from PEP.
TABLE-US-00114 TABLE 111 Protein GenBank ID GI Number Organism PCK1 NP_013023 6322950 Saccharomyces cerevisiae pck NP_417862.1 16131280 Escherichia coli pckA YP_089485.1 52426348 Mannheimia succiniciproducens pckA O09460.1 3122621 Anaerobiospirillum succiniciproducens pckA Q6W6X5 75440571 Actinobacillus succinogenes pckA P43923.1 1172573 Haemophilus influenza
[0563] Pyruvate carboxylase (EC 6.4.1.1) directly converts pyruvate to oxaloacetate at the cost of one ATP. Pyruvate carboxylase enzymes are encoded by PYC1 (Walker et al., Biochem. Biophys. Res. Commun. 176:1210-1217 (1991) and PYC2 (Walker et al., supra) in Saccharomyces cerevisiae, and pyc in Mycobacterium smegmatis (Mukhopadhyay and Purwantini, Biochim. Biophys. Acta 1475:191-206 (2000)).
TABLE-US-00115 TABLE 112 Protein GenBank ID GI Number Organism PYC1 NP_011453 6321376 Saccharomyces cerevisiae PYC2 NP_009777 6319695 Saccharomyces cerevisiae Pyc YP_890857.1 118470447 Mycobacterium smegmatis
[0564] Malic enzyme can be applied to convert CO.sub.2 and pyruvate to malate at the expense of one reducing equivalent. Malic enzymes for this purpose can include, without limitation, malic enzyme (NAD-dependent) and malic enzyme (NADP-dependent). For example, one of the E. coli malic enzymes (Takeo, J. Biochem. 66:379-387 (1969)) or a similar enzyme with higher activity can be expressed to enable the conversion of pyruvate and CO.sub.2 to malate. By fixing carbon to pyruvate as opposed to PEP, malic enzyme allows the high-energy phosphate bond from PEP to be conserved by pyruvate kinase whereby ATP is generated in the formation of pyruvate or by the phosphotransferase system for glucose transport. Although malic enzyme is typically assumed to operate in the direction of pyruvate formation from malate, overexpression of the NAD-dependent enzyme, encoded by maeA, has been demonstrated to increase succinate production in E. coli while restoring the lethal .DELTA.pfl-.DELTA.ldhA phenotype under anaerobic conditions by operating in the carbon-fixing direction (Stols and Donnelly, Appl. Environ. Microbiol. 63(7) 2695-2701 (1997)). A similar observation was made upon overexpressing the malic enzyme from Ascaris suum in E. coli (Stols et al., Appl. Biochem. Biotechnol. 63-65(1), 153-158 (1997)). The second E. coli malic enzyme, encoded by maeB, is NADP-dependent and also decarboxylates oxaloacetate and other alpha-keto acids (Iwakura et al., J. Biochem. 85(5):1355-65 (1979)).
TABLE-US-00116 TABLE 113 Protein GenBank ID GI Number Organism maeA NP_415996 90111281 Escherichia coli maeB NP_416958 16130388 Escherichia coli NAD-ME P27443 126732 Ascaris suum
[0565] The enzymes used for converting oxaloacetate (formed from, for example, PEP carboxylase, PEP carboxykinase, or pyruvate carboxylase) or malate (formed from, for example, malic enzyme or malate dehydrogenase) to succinyl-CoA via the reductive branch of the TCA cycle are malate dehydrogenase, fumarate dehydratase (fumarase), fumarate reductase, and succinyl-CoA transferase. The genes for each of the enzymes are described herein above.
[0566] Enzymes, genes and methods for engineering pathways from succinyl-CoA to various products into a microorganism are now known in the art. The additional reducing equivalents obtained from CO and/or H2, as disclosed herein, improve the yields of 1,3-butanediol when utilizing carbohydrate-based feedstock. For example, 1,3-butanediol can be produced from succinyl-CoA via succinate semialdehyde, 4-hydroxybutyrate, 4-hydroxybutyryl-phosphate, 4-hydroxybutyrl-CoA, crotonyl-CoA, 3-hydroxybutyryl-CoA, 3-hydroxybutanal, and/or 3-hydroxybutryate. Exemplary enzymes for the conversion succinyl-CoA to 1,3-butanediol include: A. Succinyl-CoA transferase, or Succinyl-CoA synthetase (or succinyl-CoA ligase), B. Succinyl-CoA reductase (aldehyde forming), C. 4-Hydroxybutyrate dehydrogenase, D. 4-Hydroxybutyrate kinase, E. Phosphotrans-4-hydroxybutyrylase, F. Succinate reductase, G. Succinyl-CoA reductase (alcohol forming), H. 4-Hydroxybutyryl-CoA transferase, or 4-Hydroxybutyryl-CoA synthetase, I. Alpha-ketoglutarate decarboxylase or (Q. Glutamate dehydrogenase and/or R. Glutamate transaminase; S. Glutamate decarboxylase; T. 4-aminobutyrate dehydrogenase and/or U. 4-aminobutyrate transaminase), J. 4-hydroxybutyryl-CoA dehydratase, K. crotonase, L. 3-hydroxybutyryl-CoA reductase (aldehyde forming), M. 3-hydroxybutanal reductase, N. 3-hydroxybutyryl-CoA reductase (alcohol forming), O. 3-hydroxybutyryl-CoA hydrolase, transferase, or synthetase, P. 3-hydroxybutyrate reductase.
[0567] Enzymes, genes and methods for engineering pathways from glycolysis intermediates to various products into a microorganism are known in the art. The additional reducing equivalents obtained from CO and H2, as described herein, improve the yields of all these products on carbohydrates. For example, 1,3-butanediol can be produced from the glycolysis intermediate, pyruvate. Exemplary enzymes for the conversion of pyruvate to 1,3-butanediol include alanine dehydrogenase alanine aminotransferase; the remaining enzymatic transformations shown are carried out by the following enzymes: A) AKP thiolase, B) AKP aminotransferase or AKP oxidoreductase (deaminating), C) 2,4-dioxopentanoate decarboxylase, D) 3-oxobutyraldehyde reductase (aldehyde reducing), E) AKP decarboxylase, F) 4-aminobutan-2-one ammonia-lyase, G) Butenone hydratase, H) 4-hydroxy,2-butanone reductase, I) AKP ammonia-lyase, J) acetylacrylate decarboxylase, K) 4-aminobutan-2-one aminotransferase or 4-aminobutan-2-one oxidoreductase (deaminating), L) AKP dehydrogenase, M) 2-amino-4-hydroxypentanoate aminotransferase or 2-amino-4-hydroxypentanoate oxidoreductase (deaminating), N) 2-oxo-4-hydroxypentanoate decarboxylase, O) 3-oxobutyraldehyde reductase (ketone reducing), and P) 3-hydroxybutyraldehdye reductase.
Example V
Methods for Handling CO and Anaerobic Cultures
[0568] This example describes methods used in handling CO and anaerobic cultures.
[0569] A. Handling of CO in small quantities for assays and small cultures. CO is an odorless, colorless and tasteless gas that is a poison. Therefore, cultures and assays that utilized CO required special handling. Several assays, including CO oxidation, acetyl-CoA synthesis, CO concentration using myoglobin, and CO tolerance/utilization in small batch cultures, called for small quantities of the CO gas that were dispensed and handled within a fume hood. Biochemical assays called for saturating very small quantities (<2 mL) of the biochemical assay medium or buffer with CO and then performing the assay. All of the CO handling steps were performed in a fume hood with the sash set at the proper height and blower turned on; CO was dispensed from a compressed gas cylinder and the regulator connected to a Schlenk line. The latter ensures that equal concentrations of CO were dispensed to each of several possible cuvettes or vials. The Schlenk line was set up containing an oxygen scrubber on the input side and an oil pressure release bubbler and vent on the other side. Assay cuvettes were both anaerobic and CO-containing. Therefore, the assay cuvettes were tightly sealed with a rubber stopper and reagents were added or removed using gas-tight needles and syringes. Secondly, small (.about.50 mL) cultures were grown with saturating CO in tightly stoppered serum bottles. As with the biochemical assays, the CO-saturated microbial cultures were equilibrated in the fume hood using the Schlenk line setup. Both the biochemical assays and microbial cultures were in portable, sealed containers and in small volumes making for safe handling outside of the fume hood. The compressed CO tank was adjacent to the fume hood.
[0570] Typically, a Schlenk line was used to dispense CO to cuvettes, each vented. Rubber stoppers on the cuvettes were pierced with 19 or 20 gage disposable syringe needles and were vented with the same. An oil bubbler was used with a CO tank and oxygen scrubber. The glass or quartz spectrophotometer cuvettes have a circular hole on top into which a Kontes stopper sleeve, Sz7 774250-0007 was fitted. The CO detector unit was positioned proximal to the fume hood.
[0571] B. Handling of CO in larger quantities fed to large-scale cultures. Fermentation cultures are fed either CO or a mixture of CO and H2 to simulate syngas as a feedstock in fermentative production. Therefore, quantities of cells ranging from 1 liter to several liters can include the addition of CO gas to increase the dissolved concentration of CO in the medium. In these circumstances, fairly large and continuously administered quantities of CO gas are added to the cultures. At different points, the cultures are harvested or samples removed. Alternatively, cells are harvested with an integrated continuous flow centrifuge that is part of the fermenter.
[0572] The fermentative processes are carried out under anaerobic conditions. In some cases, it is uneconomical to pump oxygen or air into fermenters to ensure adequate oxygen saturation to provide a respiratory environment. In addition, the reducing power generated during anaerobic fermentation may be needed in product formation rather than respiration. Furthermore, many of the enzymes for various pathways are oxygen-sensitive to varying degrees. Classic acetogens such as M. thermoacetica are obligate anaerobes and the enzymes in the Wood-Ljungdahl pathway are highly sensitive to irreversible inactivation by molecular oxygen. While there are oxygen-tolerant acetogens, the repertoire of enzymes in the Wood-Ljungdahl pathway might be incompatible in the presence of oxygen because most are metallo-enzymes, key components are ferredoxins, and regulation can divert metabolism away from the Wood-Ljungdahl pathway to maximize energy acquisition. At the same time, cells in culture act as oxygen scavengers that moderate the need for extreme measures in the presence of large cell growth.
[0573] C. Anaerobic chamber and conditions. Exemplary anaerobic chambers are available commercially (see, for example, Vacuum Atmospheres Company, Hawthorne Calif.; MBraun, Newburyport Mass.). Conditions included an O2 concentration of 1 ppm or less and 1 atm pure N2. In one example, 3 oxygen scrubbers/catalyst regenerators were used, and the chamber included an O2 electrode (such as Teledyne; City of Industry Calif.). Nearly all items and reagents were cycled four times in the airlock of the chamber prior to opening the inner chamber door. Reagents with a volume >5 mL were sparged with pure N2 prior to introduction into the chamber. Gloves are changed twice/yr and the catalyst containers were regenerated periodically when the chamber displays increasingly sluggish response to changes in oxygen levels. The chamber's pressure was controlled through one-way valves activated by solenoids. This feature allowed setting the chamber pressure at a level higher than the surroundings to allow transfer of very small tubes through the purge valve.
[0574] The anaerobic chambers achieved levels of O2 that were consistently very low and were needed for highly oxygen sensitive anaerobic conditions. However, growth and handling of cells does not usually require such precautions. In an alternative anaerobic chamber configuration, platinum or palladium can be used as a catalyst that requires some hydrogen gas in the mix. Instead of using solenoid valves, pressure release can be controlled by a bubbler. Instead of using instrument-based O2 monitoring, test strips can be used instead.
[0575] D. Anaerobic microbiology. Small cultures were handled as described above for CO handling. In particular, serum or media bottles are fitted with thick rubber stoppers and aluminum crimps are employed to seal the bottle. Medium, such as Terrific Broth, is made in a conventional manner and dispensed to an appropriately sized serum bottle. The bottles are sparged with nitrogen for .about.30 min of moderate bubbling. This removes most of the oxygen from the medium and, after this step, each bottle is capped with a rubber stopper (such as Bellco 20 mm septum stoppers; Bellco, Vineland, N.J.) and crimp-sealed (Bellco 20 mm). Then the bottles of medium are autoclaved using a slow (liquid) exhaust cycle. At least sometimes a needle can be poked through the stopper to provide exhaust during autoclaving; the needle needs to be removed immediately upon removal from the autoclave. The sterile medium has the remaining medium components, for example buffer or antibiotics, added via syringe and needle. Prior to addition of reducing agents, the bottles are equilibrated for 30-60 minutes with nitrogen (or CO depending upon use). A reducing agent such as a 100.times.150 mM sodium sulfide, 200 mM cysteine-HCl is added. This is made by weighing the sodium sulfide into a dry beaker and the cysteine into a serum bottle, bringing both into the anaerobic chamber, dissolving the sodium sulfide into anaerobic water, then adding this to the cysteine in the serum bottle. The bottle is stoppered immediately as the sodium sulfide solution generates hydrogen sulfide gas upon contact with the cysteine. When injecting into the culture, a syringe filter is used to sterilize the solution. Other components are added through syringe needles, such as B 12 (10 .quadrature.M cyanocobalamin), nickel chloride (NiCl2, 20 microM final concentration from a 40 mM stock made in anaerobic water in the chamber and sterilized by autoclaving or by using a syringe filter upon injection into the culture), and ferrous ammonium sulfate (final concentration needed is 100 .quadrature.M--made as 100-1000.times. stock solution in anaerobic water in the chamber and sterilized by autoclaving or by using a syringe filter upon injection into the culture). To facilitate faster growth under anaerobic conditions, the 1 liter bottles were inoculated with 50 mL of a preculture grown anaerobically. Induction of the pA1-lacO1 promoter in the vectors was performed by addition of isopropyl .beta.-D-1-thiogalactopyranoside (IPTG) to a final concentration of 0.2 mM and was carried out for about 3 hrs.
[0576] Large cultures can be grown in larger bottles using continuous gas addition while bubbling. A rubber stopper with a metal bubbler is placed in the bottle after medium addition and sparged with nitrogen for 30 minutes or more prior to setting up the rest of the bottle. Each bottle is put together such that a sterile filter will sterilize the gas bubbled in and the hoses on the bottles are compressible with small C clamps. Medium and cells are stirred with magnetic stir bars. Once all medium components and cells are added, the bottles are incubated in an incubator in room air but with continuous nitrogen sparging into the bottles.
Example VI
CO Oxidation (CODH) Assay
[0577] This example describes assay methods for measuring CO oxidation (CO dehydrogenase; CODH).
[0578] The 7 gene CODH/ACS operon of Moorella thermoacetica was cloned into E. coli expression vectors. The intact .about.10 kbp DNA fragment was cloned, and it is likely that some of the genes in this region are expressed from their own endogenous promoters and all contain endogenous ribosomal binding sites. These clones were assayed for CO oxidation, using an assay that quantitatively measures CODH activity. Antisera to the M. thermoacetica gene products was used for Western blots to estimate specific activity. M. thermoacetica is Gram positive, and ribosome binding site elements are expected to work well in E. coli. This activity, described below in more detail, was estimated to be .about. 1/50th of the M. thermoacetica specific activity. It is possible that CODH activity of recombinant E. coli cells could be limited by the fact that M. thermoacetica enzymes have temperature optima around 55.degree. C. Therefore, a mesophilic CODH/ACS pathway could be advantageous such as the close relative of Moorella that is mesophilic and does have an apparently intact CODH/ACS operon and a Wood-Ljungdahl pathway, Desulfitobacterium hafniense. Acetogens as potential host organisms include, but are not limited to, Rhodospirillum rubrum, Moorella thermoacetica and Desulfitobacterium hafniense.
[0579] CO oxidation is both the most sensitive and most robust of the CODH/ACS assays. It is likely that an E. coli-based syngas using system will ultimately need to be about as anaerobic as Clostridial (i.e., Moorella) systems, especially for maximal activity. Improvement in CODH should be possible but will ultimately be limited by the solubility of CO gas in water.
[0580] Initially, each of the genes was cloned individually into expression vectors. Combined expression units for multiple subunits/1 complex were generated. Expression in E. coli at the protein level was determined. Both combined M. thermoacetica CODH/ACS operons and individual expression clones were made.
[0581] CO oxidation assay. This assay is one of the simpler, reliable, and more versatile assays of enzymatic activities within the Wood-Ljungdahl pathway and tests CODH (Seravalli et al., Biochemistry 43:3944-3955 (2004)). A typical activity of M. thermoacetica CODH specific activity is 500 U at 55.degree. C. or .about.60 U at 25.degree. C. This assay employs reduction of methyl viologen in the presence of CO. This is measured at 578 nm in stoppered, anaerobic, glass cuvettes.
[0582] In more detail, glass rubber stoppered cuvettes were prepared after first washing the cuvette four times in deionized water and one time with acetone. A small amount of vacuum grease was smeared on the top of the rubber gasket. The cuvette was gassed with CO, dried 10 mM with a 22 Ga. needle plus an exhaust needle. A volume of 0.98 mL of reaction buffer (50 mM Hepes, pH 8.5, 2 mM dithiothreitol (DTT) was added using a 22 Ga. needle, with exhaust needled, and 100% CO. Methyl viologen (CH3 viologen) stock was 1 M in water. Each assay used 20 microliters for 20 mM final concentration. When methyl viologen was added, an 18 Ga needle (partial) was used as a jacket to facilitate use of a Hamilton syringe to withdraw the CH3 viologen. 4-5 aliquots were drawn up and discarded to wash and gas equilibrate the syringe. A small amount of sodium dithionite (0.1 M stock) was added when making up the CH3 viologen stock to slightly reduce the CH3 viologen. The temperature was equilibrated to 55.degree. C. in a heated Olis spectrophotometer (Bogart GA). A blank reaction (CH3 viologen+buffer) was run first to measure the base rate of CH3 viologen reduction. Crude E. coli cell extracts of ACS90 and ACS91 (CODH-ACS operon of M. thermoacetica with and without, respectively, the first cooC). 10 microliters of extract were added at a time, mixed and assayed. Reduced CH3 viologen turns purple. The results of an assay are shown in Table 114.
TABLE-US-00117 TABLE 114 Crude extract CO Oxidation Activities. ACS90 7.7 mg/ml ACS91 11.8 mg/ml Mta98 9.8 mg/ml Mta99 11.2 mg/ml Extract Vol OD/ U/ml U/mg ACS90 10 microliters 0.073 0.376 0.049 ACS91 10 microliters 0.096 0.494 0.042 Mta99 10 microliters 0.0031 0.016 0.0014 ACS90 10 microliters 0.099 0.51 0.066 Mta99 25 microliters 0.012 0.025 0.0022 ACS91 25 microliters 0.215 0.443 0.037 Mta98 25 microliters 0.019 0.039 0.004 ACS91 10 microliters 0.129 0.66 0.056 Averages ACS90 0.057 U/mg ACS91 0.045 U/mg Mta99 0.0018 U/mg
[0583] Mta98/Mta99 are E. coli MG1655 strains that express methanol methyltransferase genes from M. thermoacetia and, therefore, are negative controls for the ACS90 ACS91 E. coli strains that contain M. thermoacetica CODH operons.
[0584] If .about.1% of the cellular protein is CODH, then these figures would be approximately 100.times. less than the 500 U/mg activity of pure M. thermoacetica CODH. Actual estimates based on Western blots are 0.5% of the cellular protein, so the activity is about 50.times. less than for M. thermoacetica CODH. Nevertheless, this experiment demonstrates CO oxidation activity in recombinant E. coli with a much smaller amount in the negative controls. The small amount of CO oxidation (CH3 viologen reduction) seen in the negative controls indicates that E. coli may have a limited ability to reduce CH3 viologen.
[0585] To estimate the final concentrations of CODH and Mtr proteins, SDS-PAGE followed by Western blot analyses were performed on the same cell extracts used in the CO oxidation, ACS, methyltransferase, and corrinoid Fe--S assays. The antisera used were polyclonal to purified M. thermoacetica CODH-ACS and Mtr proteins and were visualized using an alkaline phosphatase-linked goat-anti-rabbit secondary antibody. The Westerns were performed and results are shown in FIG. 9. The amounts of CODH in ACS90 and ACS91 were estimated at 50 ng by comparison to the control lanes. Expression of CODH-ACS operon genes including 2 CODH subunits and the methyltransferase were confirmed via Western blot analysis. Therefore, the recombinant E. coli cells express multiple components of a 7 gene operon. In addition, both the methyltransferase and corrinoid iron sulfur protein were active in the same recombinant E. coli cells. These proteins are part of the same operon cloned into the same cells.
[0586] The CO oxidation assays were repeated using extracts of Moorella thermoacetica cells for the positive controls. Though CODH activity in E. coli ACS90 and ACS91 was measurable, it was at about 130-150.times. lower than the M. thermoacetica control. The results of the assay are shown in FIG. 11. Briefly, cells (M. thermoacetica or E. coli with the CODH/ACS operon; ACS90 or ACS91 or empty vector: pZA33S) were grown and extracts prepared as described above. Assays were performed as described above at 55.degree. C. at various times on the day the extracts were prepared. Reduction of methylviologen was followed at 578 nm over a 120 sec time course.
[0587] These results describe the CO oxidation (CODH) assay and results. Recombinant E. coli cells expressed CO oxidation activity as measured by the methyl viologen reduction assay.
Example VII
E. coli CO Tolerance Experiment and CO Concentration Assay (Myoglobin Assay)
[0588] This example describes the tolerance of E. coli for high concentrations of CO.
[0589] To test whether or not E. coli can grow anaerobically in the presence of saturating amounts of CO, cultures were set up in 120 ml serum bottles with 50 ml of Terrific Broth medium (plus reducing solution, NiCl2, Fe(II)NH4SO4, cyanocobalamin, IPTG, and chloramphenicol) as described above for anaerobic microbiology in small volumes. One half of these bottles were equilibrated with nitrogen gas for 30 min. and one half was equilibrated with CO gas for 30 min. An empty vector (pZA33) was used as a control, and cultures containing the pZA33 empty vector as well as both ACS90 and ACS91 were tested with both N2 and CO. All were inoculated and grown for 36 hrs with shaking (250 rpm) at 37.degree. C. At the end of the 36 hour period, examination of the flasks showed high amounts of growth in all. The bulk of the observed growth occurred overnight with a long lag.
[0590] Given that all cultures appeared to grow well in the presence of CO, the final CO concentrations were confirmed. This was performed using an assay of the spectral shift of myoglobin upon exposure to CO. Myoglobin reduced with sodium dithionite has an absorbance peak at 435 nm; this peak is shifted to 423 nm with CO. Due to the low wavelength and need to record a whole spectrum from 300 nm on upwards, quartz cuvettes must be used. CO concentration is measured against a standard curve and depends upon the Henry's Law constant for CO of maximum water solubility=970 micromolar at 20.degree. C. and 1 atm.
[0591] For the myoglobin test of CO concentration, cuvettes were washed 10.times. with water, 1.times. with acetone, and then stoppered as with the CODH assay. N2 was blown into the cuvettes for .about.10 min. A volume of 1 ml of anaerobic buffer (HEPES, pH 8.0, 2 mM DTT) was added to the blank (not equilibrated with CO) with a Hamilton syringe. A volume of 10 microliter myoglobin (.about.1 mM--can be varied, just need a fairly large amount) and 1 microliter dithionite (20 mM stock) were added. A CO standard curve was made using CO saturated buffer added at 1 microliter increments. Peak height and shift was recorded for each increment. The cultures tested were pZA33/CO, ACS90/CO, and ACS91/CO. Each of these was added in 1 microliter increments to the same cuvette. Midway through the experiment a second cuvette was set up and used. The results are shown in Table 115.
TABLE-US-00118 TABLE 115 Carbon Monoxide Concentrations, 36 hrs. Strain and Growth Conditions Final CO concentration (micromolar) pZA33-CO 930 ACS90-CO 638 494 734 883 ave 687 SD 164 ACS91-CO 728 812 760 611 ave. 728 SD 85
[0592] The results shown in Table 115 indicate that the cultures grew whether or not a strain was cultured in the presence of CO or not. These results indicate that E. coli can tolerate exposure to CO under anaerobic conditions and that E. coli cells expressing the CODH-ACS operon can metabolize some of the CO.
[0593] These results demonstrate that E. coli cells, whether expressing CODH/ACS or not, were able to grow in the presence of saturating amounts of CO. Furthermore, these grew equally well as the controls in nitrogen in place of CO. This experiment demonstrated that laboratory strains of E. coli are insensitive to CO at the levels achievable in a syngas project performed at normal atmospheric pressure. In addition, preliminary experiments indicated that the recombinant E. coli cells expressing CODH/ACS actually consumed some CO, probably by oxidation to carbon dioxide.
Example VIII
Exemplary Carboxylic Acid Reductases
[0594] This example describes the use of carboxylic acid reductases (CAR) to carry out the conversion of a carboxylic acid to an aldehyde.
[0595] Any intermediate carboxylic acid in a 1,3-butanediol pathway (or accessible carboxylic acid via its CoA derivative) can be converted to an aldehyde, if so desired. The conversion of unactivated acids to aldehydes can be carried out by an acid reductase. Examples of such conversions include, but are not limited, the conversion of 4-hydroxybutyrate, succinate, alpha-ketoglutarate, and 4-aminobutyrate to 4-hydroxybutanal, succinate semialdehyde, 2,5-dioxopentanoate, and 4-aminobutanal, respectively. One notable carboxylic acid reductase can be found in Nocardia iowensis which catalyzes the magnesium, ATP and NADPH-dependent reduction of carboxylic acids to their corresponding aldehydes (Venkitasubramanian et al., J. Biol. Chem. 282:478-485 (2007)). This enzyme is encoded by the car gene and was cloned and functionally expressed in E. coli (Venkitasubramanian et al., J. Biol. Chem. 282:478-485 (2007)). Expression of the npt gene product improved activity of the enzyme via post-transcriptional modification. The npt gene encodes a specific phosphopantetheine transferase (PPTase) that converts the inactive apo-enzyme to the active holo-enzyme. The natural substrate of this enzyme is vanillic acid, and the enzyme exhibits broad acceptance of aromatic and aliphatic substrates (Venkitasubramanian et al., in Biocatalysis in the Pharmaceutical and Biotechnology Industires, ed. R. N. Patel, Chapter 15, pp. 425-440, CRC Press LLC, Boca Raton, Fla. (2006)).
TABLE-US-00119 TABLE 116 Gene Accession No. GI No. Organism car AAR91681.1 40796035 Nocardia iowensis (sp. NRRL 5646) npt ABI83656.1 114848891 Nocardia iowensis (sp. NRRL 5646)
[0596] Additional car and npt genes can be identified based on sequence homology.
TABLE-US-00120 TABLE 117 Gene Accession No. GI No. Organism fadD9 YP_978699.1 121638475 Mycobacterium bovis BCG BCG_2812c YP_978898.1 121638674 Mycobacterium bovis BCG nfa20150 YP_118225.1 54023983 Nocardia farcinica IFM 10152 nfa40540 YP_120266.1 54026024 Nocardia farcinica IFM 10152 SGR_6790 YP_001828302.1 182440583 Streptomyces griseus subsp. griseus NBRC 13350 SGR_665 YP_001822177.1 182434458 Streptomyces griseus subsp. griseus NBRC 13350
[0597] An additional enzyme candidate found in Streptomyces griseus is encoded by the griC and griD genes. This enzyme is believed to convert 3-amino-4-hydroxybenzoic acid to 3-amino-4-hydroxybenzaldehyde as deletion of either griC or griD led to accumulation of extracellular 3-acetylamino-4-hydroxybenzoic acid, a shunt product of 3-amino-4-hydroxybenzoic acid metabolism (Suzuki, et al., J. Antibiot. 60(6):380-387 (2007)). Co-expression of griC and griD with SGR.sub.--665, an enzyme similar in sequence to the Nocardia iowensis npt, can be beneficial.
TABLE-US-00121 TABLE 118 Gene Accession No. GI No. Organism griC 182438036 YP_001825755.1 Streptomyces griseus subsp. griseus NBRC 13350 griD 182438037 YP_001825756.1 Streptomyces griseus subsp. griseus NBRC 13350 MSMEG_2956 YP_887275.1 YP_887275.1 Mycobacterium smegmatis MC2 155 MSMEG_5739 YP_889972.1 118469671 Mycobacterium smegmatis MC2 155 MSMEG_2648 YP_886985.1 118471293 Mycobacterium smegmatis MC2 155 MAP1040c NP_959974.1 41407138 Mycobacterium avium subsp. paratuberculosis K- 10 MAP2899c NP_961833.1 41408997 Mycobacterium avium subsp. paratuberculosis K- 10 MMAR_2117 YP_001850422.1 183982131 Mycobacterium marinum M MMAR_2936 YP_001851230.1 183982939 Mycobacterium marinum M MMAR_1916 YP_001850220.1 183981929 Mycobacterium marinum M TpauDRAFT_33060 ZP_04027864.1 227980601 Tsukamurella paurometabola DSM 20162 TpauDRAFT_20920 ZP_04026660.1 227979396 Tsukamurella paurometabola DSM 20162 CPCC7001_1320 ZP_05045132.1 254431429 Cyanobium PCC7001 DDBDRAFT_0187729 XP_636931.1 66806417 Dictyostelium discoideum AX4
[0598] An enzyme with similar characteristics, alpha-aminoadipate reductase (AAR, EC 1.2.1.31), participates in lysine biosynthesis pathways in some fungal species. This enzyme naturally reduces alpha-aminoadipate to alpha-aminoadipate semialdehyde. The carboxyl group is first activated through the ATP-dependent formation of an adenylate that is then reduced by NAD(P)H to yield the aldehyde and AMP. Like CAR, this enzyme utilizes magnesium and requires activation by a PPTase. Enzyme candidates for AAR and its corresponding PPTase are found in Saccharomyces cerevisiae (Morris et al., Gene 98:141-145 (1991)), Candida albicans (Guo et al., Mol. Genet. Genomics 269:271-279 (2003)), and Schizosaccharomyces pombe (Ford et al., Curr. Genet. 28:131-137 (1995)). The AAR from S. pombe exhibited significant activity when expressed in E. coli (Guo et al., Yeast 21:1279-1288 (2004)). The AAR from Penicillium chrysogenum accepts S-carboxymethyl-L-cysteine as an alternate substrate, but did not react with adipate, L-glutamate or diaminopimelate (Hijarrubia et al., J. Biol. Chem. 278:8250-8256 (2003)). The gene encoding the P. chrysogenum PPTase has not been identified to date.
TABLE-US-00122 TABLE 119 Gene Accession No. GI No. Organism LYS2 AAA34747.1 171867 Saccharomyces cerevisiae LYS5 P50113.1 1708896 Saccharomyces cerevisiae LYS2 AAC02241.1 2853226 Candida albicans LYS5 AAO26020.1 28136195 Candida albicans Lys1p P40976.3 13124791 Schizosaccharomyces pombe Lys7p Q10474.1 1723561 Schizosaccharomyces pombe Lys2 CAA74300.1 3282044 Penicillium chrysogenum
[0599] Cloning and Expression of Carboxylic Acid Reductase.
[0600] Escherichia coli is used as a target organism to engineer the pathway for 1,3-butanediol. E. coli provides a good host for generating a non-naturally occurring microorganism capable of producing 1,3-butanedial. E. coli is amenable to genetic manipulation and is known to be capable of producing various intermediates and products effectively under various oxygenation conditions.
[0601] To generate a microbial organism strain such as an E. coli strain engineered to produce 1,3-butanedial, nucleic acids encoding a carboxylic acid reductase and phosphopantetheine transferase are expressed in E. coli using well known molecular biology techniques (see, for example, Sambrook, supra, 2001; Ausubel supra, 1999). In particular, car genes from Nocardia iowensis (designated 720), Mycobacterium smegmatis mc(2)155 (designated 890), Mycobacterium avium subspecies paratuberculosis K-10 (designated 891) and Mycobacterium marinum M (designated 892) were cloned into pZS*13 vectors (Expressys, Ruelzheim, Germany) under control of PA1/lacO promoters. The npt (ABI83656.1) gene (i.e., 721) was cloned into the pKJL33S vector, a derivative of the original mini-F plasmid vector PML31 under control of promoters and ribosomal binding sites similar to those used in pZS*13.
[0602] The car gene (GNM.sub.--720) was cloned by PCR from Nocardia genomic DNA. Its nucleic acid and protein sequences are shown in FIGS. 12A and 12B, respectively. A codon-optimized version of the npt gene (GNM.sub.--721) was synthesized by GeneArt (Regensburg, Germany). Its nucleic acid and protein sequences are shown in FIGS. 13A and 13B, respectively. The nucleic acid and protein sequences for the Mycobacterium smegmatis mc(2)155 (designated 890), Mycobacterium avium subspecies paratuberculosis K-10 (designated 891) and Mycobacterium marinum M (designated 892) genes and enzymes can be found in FIGS. 14, 15, and 16, respectively. The plasmids are transformed into a host cell to express the proteins and enzymes required for 1,3-butanedial production.
[0603] Additional CAR variants were generated. A codon optimized version of CAR 891 was generated and designated 891GA. The nucleic acid and amino acid sequences of CAR 891 GA are shown in FIGS. 17A and 17B, respectively. Over 2000 CAR variants were generated. In particular, all 20 amino acid combinations were made at positions V295, M296, G297, G391, G421, D413, G414, Y415, G416, and S417, and additional variants were tested as well. Exemplary CAR variants include: E16K; Q95L; L100M; A1011T; K823E; T941S; H15Q; D198E; G446C; S392N; F699L; V8831; F467S; T987S; R12H; V295G; V295A; V295S; V295T; V295C; V295V; V295L; V2951; V295M; V295P; V295F; V295Y; V295W; V295D; V295E; V295N; V295Q; V295H; V295K; V295R; M296G; M296A; M296S; M296T; M296C; M296V; M296L; M296I; M296M; M296P; M296F; M296Y; M296W; M296D; M296E; M296N; M296Q; M296H; M296K; M296R; G297G; G297A; G297S; G297T; G297C; G297V; G297L; G297I; G297M; G297P; G297F; G297Y; G297W; G297D; G297E; G297N; G297Q; G297H; G297K; G297R; G391G; G391A; G391S; G391T; G391C; G391V; G391L; G391I; G391M; G391P; G391F; G391Y; G391W; G391D; G391E; G391N; G391Q; G391H; G391K; G391R; G421G; G421A; G421S; G421T; G421C; G421V; G421L; G421I G421M; G421P; G421F; G421Y; G421W; G421D; G421E; G421N; G421Q; G421H; G421K; G421R; D413G; D413A; D413S; D413T; D413C; D413V; D413L; D413I; D413M; D413P; D413F; D413Y; D413W; D413D; D413E; D413N; D413Q; D413H; D413K; D413R; G414G; G414A; G414S; G414T; G414C; G414V; G414L; G414I; G414M; G414P; G414F; G414Y; G414W; G414D; G414E; G414N; G414Q; G414H; G414K; G414R; Y415G; Y415A; Y415S; Y415T; Y415C; Y415V; Y415L; Y415I; Y415M; Y415P; Y415F; Y415Y; Y415W; Y415D; Y415E; Y415N; Y415Q; Y415H; Y415K; Y415R; G416G; G416A; G416S; G416T; G416C; G416V; G416L; G416I; G416M; G416P; G416F; G416Y; G416W; G416D; G416E; G416N; G416Q; G416H; G416K; G416R; S417G; S417A; S417S; S417T; S417C; S417V S417L; S417I; S417M; S417P; S417F; S417Y; S417W; S417D; S417E; S417N; S417Q; S417H; S417K; and S417R.
[0604] The CAR variants were screened for activity, and numerous CAR variants were found to exhibit CAR activity. This example describes the use of CAR for converting carboxylic acids to aldehydes.
TABLE-US-00123 TABLE 120 (Ref: FIG. 1) Step EC class Desired substrate Desired product Enzyme name Gene name A 2.3.1.b D-alanine 2-amino-4- AKP Thiolase ortA oxopentanoate ortB Amet_2368 Amet_2369 Teth514_1478 Teth514_1479 B 2.6.1.a 2-amino-4- 2,4- 2-amino-4- aspC oxopentanoate dioxopentanoate oxopentanoate aminotransferase or oxidoreductase (deaminating) avtA AAT2 dat dat ldh nadX C 4.1.1.a 2,4-dioxopentanoate 3-oxobutyr- 2,4-dioxopentanoate pdc aldehyde decarboxylase (3-oxobutanal) pdc1 mdlC kgd D 1.1.1.a 3-oxobutyraldehyde 4-hydroxy,2- 3-oxobutyraldehyde alrA butanone reductase (aldehyde reducing) ADH2 yqhD bdh I bdh II 4hbd ADHI mmsb P84067 E 4.1.1.a 2-amino-4- 4-aminobutan-2- 2-amino-4- lysA oxopentanoate one oxopentanoate decarboxylase lysA lysA odc1 panD panD panD F 4.3.1.a 4-aminobutan-2-one butenone 4-aminobutan-2-one aspA ammonia lyase aspA aspA ansB aspA G 4.2.1.a butenone 4-hydroxy,2- butenone hydratase fumA butanone fumC fumC fumC fumH hmd dmdA dmdB H 1.1.1.a 4-hydroxy,2- 1,3-butanediol 4-hydroxy,2- bdh butanone butanone reductase adh adhA ldh adh I 4.3.1.a 2-amino-4- acetylacrylate 2-amino-4- aspA oxopentanoate oxopentanoate ammonia lyase aspA aspA ansB aspA J 4.1.1.a acetylacrylate butenone acetylacrylate xylII decarboxylase xylIII dmpH dmpE pdc pad K 2.6.1.a 4-aminobutan-2-one 3-oxobutyr- 4-aminobutan-2-one SkyPYD4 aldehyde aminotransferase or (3-oxobutanal) oxidoreductase (deaminating) gabT Abat UGA1 kdd lysDH L 1.1.1.a 2-amino-4- 2-amino-4- 2-amino-4- thrA oxopentanoate hydroxypentanoate oxopentanoate dehydrogenase hom6 hom2 akthr2 hom1 M 2.6.1.a 2-amino-4- 2-oxo-4- 2-amino-4- aspC hydroxypentanoate hydroxypentanoate hydroxypentanoate aminotransferase or oxidoreductase (deaminating) avtA AAT2 dat dat ldh nadX N 4.1.1.a 2-oxo-4- 3-hydroxybutyr- 2-oxo-4- pdc hydroxypentanoate aldehyde hydroxypentanoate (3-hydroxybutanal) pdc1 mdlC kgd O 1.1.1.a 3-oxobutyraldehyde 3-hydroxybutyr- 3-oxobutyraldehyde bdh aldehyde reductase (ketone reducing) adh adhA ldh adh P 1.1.1.a 3-hydroxybutyr- 1,3-butanediol 3-hydroxybutyr- alrA aldehyde aldehyde reductase ADH2 yqhD bdh I bdh II 4hbd ADHI mmsb P84067 GenBank ID Step (if available) Organism Known Substrates A YP_001086914.1 Clostridium D-alanine difficile 630 YP_001086915.1 Clostridium D-alanine difficile 630 YP_001320181.1 Alkaliphilus D-alanine metalliredigenes QYF YP_001320182.1 Alkaliphilus D-alanine metalliredigenes QYF YP_001663101.1 Thermoanaero- D-alanine bacter sp. X514 YP_001663102.1 Thermoanaero- D-alanine bacter sp. X514 B NP_415448.1 Escherichia coli L-aspartate YP_026231.1 Escherichia coli L-alanine, L-valine P23542.3 Saccharomyces L-aspartate cerevisae P19938 Bacillus sp. YM-1 D-alanine, D-2- minobutanoate, D- aspartate O07597 Bacillus subtilis D-alanine, D-2- aminobutanoate, D-aspartate P0A393 Bacillus cereus L-leucine, L-valine, 2-aminobutanoate, L-isoleucine NP_229443.1 Thermotoga L-aspartate maritima C P06672.1 Zymomonas 2-ketobutyrate mobilus P06169 Saccharomyces 2-ketobutyrate, 3- cerevisae hydroxypyruvate P20906.2 Pseudomonas 2-ketobutyrate putdia O50463.4 Mycobacterium alpha-ketoglutarate tuberculosis D BAB12273.1 Acinetobacter sp. C2-C14 aldehydes Strain M-1 NP_014032.1 Saccharymyces propionaldehyde, cerevisiae isobutyraldehyde, butyraldehyde, 2- methylbutyraldehyde, 3- methylbutyraldehyde, 2-phenylacetaldehyde NP_417484.1 Escherichia coli acetaldehyde, malondialdehyde, propanaldehyde, butanaldehyde, and acrolein NP_349892.1 Clostridium butyraldehyde acetobutylicum NP_349891.1 Clostridium butyraldehyde acetobutylicum YP_726053.1 Ralstonia eutropha succinate H16 semialdehyde AAR91477.1 Geobacillus ethanol, 1-butanol, 1- thermo- pentanol, 1-heptanol, glucosidasius 1-hexanol, 1-octanol, M10EXG 2-propanol P28811.1 Pseudomonas 3- aeruginosa hydroxybutyraldehyde, malonic semialdehyde, methylmalonate semialdehyde P84067 Thermus methylmalonate thermophilus semialdehyde E NP_417315.1 Escherichia coli meso- diaminopimelate AAA25361.1 Mycobacterium meso- tuberculosis diaminopimelate BAC92756.1 Methylophilus meso- methylotrophus diaminopimelate AA59967.1 Homo sapiens D-ornithine P0A790 Escherichia coli L-aspartate Q9X4N0 Corynebacterium L-aspartate glutanicum P65660 Mycobacterium L-aspartate tuberculosis F NP_418562 Escherichia coli L-aspartate K12 subsp. MG1655 P44324.1 Haemophilus L-aspartate influenzae P07346.1 Pseudomonas L-aspartate fluorescens P26899.1 Bacillus subtilus L-aspartate P33109.1 Serratia L-aspartate marcescens G P0AC33 Escherichia coli fumarate K12 P05042 Escherichia coli fumarate K12 O69294 Campylobacter fumarate jejuni P84127 Thermus fumarate thermophilus P14408 Rattus norvegicus fumarate ABC88407.1 Eubacterium 2-methylene- barkeri glutarate ABC88408 Eubacterium dimethylmaleate barkeri ABC88409.1 Eubacterium dimethylmaleate barkeri H AAA58352.1 Homo sapiens 3-oxobutyrate AAA23199.2 Clostridium acetone beijerinckii NRRL B593 AAC25556 Pyrococuus 2-pentanaol, furiosus pyruvaldehyde YP_725182.1 Ralstonia eutropha lactate, 2-
oxobutyrate, 2-oxopentaonotae, 2-oxoglutarate P14941.1 Thermoanaero- acetone bacter brockii HTD4 I NP_418562 Escherichia coli L-aspartate K12 subsp. MG1655 P44324.1 Haemophilus L-aspartate influenzae P07346.1 Pseudomonas L-aspartate fluorescens P26899.1 Bacillus subtilus L-aspartate P33109.1 Serratia L-aspartate marcescens J YP_709328.1 Pseudomonas 4-oxalocrotonate putida YP_709353.1 Pseudomonas 4-oxalocrotonate putida CAA43228.1 Pseudomonas sp. 4-oxalocrotonate CF600 CAA43225.1 Pseudomonas sp. 4-oxalocrotonate CF600 U63827 Lactobacillus cinnamate and plantarum derivatives AB330293 Klebsiella oxytoca cinnamate and derivatives K ABF58893 Saccharomyces beta-alanine kluyveri P22256 Escherichia coli 4-aminobutyrate P50554 Rattus norvegicus 3-amino-2- methylpropionate NP_011533 Saccharomyces 4-aminobutyrate cerevisae AAL93966.1 Fusobacterium 3,5- nucleatum diaminohexanoate BAB39707 Geobacillus L-lysine stearothermophilus L AAC73113 Escherichia coli aspartate semialdehyde CAA89671 Saccharomyces aspartate cerevisae semialdehyde CAD63186 Lactobacillus aspartate plantarum semialdehyde O81852 Arabidopsis aspartate thaliana semialdehyde CAD64819 Lactobacillus aspartate plantarum semialdehyde M NP_415448.1 Escherichia coli L-aspartate YP_026231.1 Escherichia coli L-alanine, L-valine P23542.3 Saccharomyces L-aspartate cerevisae Bacillus sp. YM-1 P19938 D-alanine, D-2- aminobutanoate, D-aspartate Bacillus subtilis O07597 D-alanine, D-2-aminobutanoate, D-aspartate P0A393 Bacillus cereus L-leucine, L-valine, 2-aminobutanoate, L- isoleucine NP_229443.1 Thermotoga L-aspartate maritima N P06672.1 Zymomonas 2-ketobutyrate mobilus P06169 Saccharomyces 2-ketobutyrate, cerevisae 3-hydroxypyruvate P20906.2 Pseudomonas 2-ketobutyrate putdia O50463.4 Mycobacterium alpha-ketoglutarate tuberculosis O AAA58352.1 Homo sapiens 3-oxobutyrate AAA23199.2 Clostridium acetone beijerinckii NRRL B593 AAC25556 Pyrococuus 2-pentanaol, furiosus pyruvaldehyde YP_725182.1 Ralstonia eutropha lactate, 2-oxobutyrate, 2-oxopentaonotae, 2-oxoglutarate P14941.1 Thermoanaero- acetone bacter brockii HTD4 P BAB12273.1 Acinetobacter sp. C2-C14 aldehydes Strain M-1 NP_014032.1 Saccharymyces propionaldehyde, cerevisiae isobutyraldehyde, butyraldehyde, 2-methylbutyr- aldehyde, 3-methylbutyr- aldehyde, 2- phenylacetaldehyde NP_417484.1 Escherichia coli acetaldehyde, malondialdehyde, propanaldehyde, butanaldehyde, and acrolein NP_349892.1 Clostridium butyraldehyde acetobutylicum NP_349891.1 Clostridium butyraldehyde acetobutylicum YP_726053.1 Ralstonia eutropha succinate H16 semialdehyde AAR91477.1 Geobacillus ethanol, 1-butanol, thermoglucosidasius 1-pentanol, M10EXG 1-heptanol, 1-hexanol, 1-octanol, 2-propanol P28811.1 Pseudomonas 3-hydroxy- aeruginosa butyraldehyde, malonic semialdehyde, methylmalonate semialdehyde P84067 Thermus methylmalonate thermophilus semialdehyde
TABLE-US-00124 TABLE 121 (Ref: FIG. 2) EC GenBank ID (if Step class Desired substrate Desired product Enzyme name Gene name available) Organism Known Substrates A 1.2.1.b acetoacetyl-CoA 3-oxobutyraldehyde acetoacetyl- Ald AAT66436 Clostridium butyryl-CoA CoA reductase beijerinckii (aldehdye forming) sucD NP_904963.1 Porphyromonas succinyl-CoA gingivalis bphG BAA03892.1 Pseudomonas sp acetaldehyde, propionaldehyde, butyraldehyde, isobutyraldehyde and formaldehyde Msed_0709 YP_001190808.1 Metallosphaera malonyl-CoA sedula mcr NP_378167 Sulfolobus malonyl-CoA, tokodaii methylmalonyl- CoA B 1.1.1.a 3-oxobutyr- 3- 3-oxobutyr- bdh AAA58352.1 Homo sapiens 3-oxobutyrate aldehyde hydroxybutyraldehyde aldehyde reductase (ketone- reducing) adh AAA23199.2 Clostridium acetone beijerinckii NRRL B593 adhA AAC25556 Pyrococuus 2-pentanaol, furiosus pyruvaldehyde ldh YP_725182.1 Ralstonia lactate, 2- eutropha oxobutyrate, 2- oxopentaonotae, 2-oxoglutarate adh P14941.1 Thermoanaero- acetone bacter brockii HTD4 C 1.1.1.a 3-hydroxybutyr- 1,3-butanediol 3-hydroxy- alrA BAB12273.1 Acinetobacter sp. C2-C14 aldehyde butyraldehyde Strain M-1 aldehydes reductase ADH2 NP_014032.1 Saccharymyces propionaldehyde, cerevisiae isobutyraldehyde, butyraldehyde, 2-methylbutyr- aldehyde, 3-methylbutyr- aldehyde, 2-phenylacet- aldehyde yqhD NP_417484.1 Escherichia coli acetaldehyde, malondialdehyde, propanaldehyde, butanaldehyde, and acrolein bdh I NP_349892.1 Clostridium butyraldehyde acetobutylicum bdh II NP_349891.1 Clostridium butyraldehyde acetobutylicum 4hbd YP_726053.1 Ralstonia succinate eutropha H16 semialdehyde ADHI AAR91477.1 Geobacillus ethanol, thermoglucosidasius 1-butanol, M10EXG 1-pentanol, 1-heptanol, 1-hexanol, 1-octanol, 2-propanol mmsb P28811.1 Pseudomonas 3-hydroxybutyr- aeruginosa aldehyde, malonic semialdehyde, methylmalonate semialdehyde P84067 P84067 Thermus methylmalonate thermophilus semialdehyde D 1.1.1.c acetoacetyl-CoA 4-hydroxy,2-butanone acetoacetyl- adhE2 AAK09379.1 Clostridium butanoyl-CoA CoA reductase acetobutylicum (alcohol- forming) mcr AAS20429.1 Chloroflexus malonyl-CoA aurantiacus FAR AAD38039.1 Simmondsia long chain acyl- chinensis CoA E 1.1.1.a 3-oxobutyr- 4-hydroxy,2-butanone 3- alrA BAB12273.1 Acinetobacter sp. C2-C14 aldehyde oxobutyraldeh Strain M-1 aldehydes dye reductase (aldehyde reducing) ADH2 NP_014032.1 Saccharymyces propionaldehyde, cerevisiae isobutyraldehyde, butyraldehyde, 2- methyl- butyraldehyde, 3-methylbutyr- aldehyde, 2- phenyl- acetaldehyde yqhD NP_417484.1 Escherichia coli acetaldehyde, malondialdehyde, propanaldehyde, butanaldehyde, and acrolein bdh I NP_349892.1 Clostridium butyraldehyde acetobutylicum bdh II NP_349891.1 Clostridium butyraldehyde acetobutylicum 4hbd YP_726053.1 Ralstonia succinate eutropha H16 semialdehyde ADHI AAR91477.1 Geobacillus ethanol, thermoglucosidasius 1-butanol, M10EXG 1-pentanol, 1-heptanol, 1-hexanol, 1-octanol, 2-propanol mmsb P28811.1 Pseudomonas 3- aeruginosa hydroxy- butyraldehyde, malonic semialdehyde, methylmalonate semialdehyde P84067 P84067 Thermus methylmalonate thermophilus semialdehyde F 1.1.1.a 4-hydroxy, 1,3-butanediol 4-hydroxy,2- bdh AAA58352.1 Homo sapiens 3-oxobutyrate 2-butanone butanone reductase adh AAA23199.2 Clostridium acetone beijerinckii NRRL B593 adhA AAC25556 Pyrococuus 2-pentanaol, furiosus pyruvaldehyde ldh YP_725182.1 Ralstonia lactate, 2- eutropha oxobutyrate, 2- oxopentaonotae, 2-oxoglutarate adh P14941.1 Thermoanaero- acetone bacter brockii HTD4 G 1.1.1.a acetoacetyl-CoA 3-hydroxybutyryl-CoA acetaocetyl hbd NP_349314.1 Clostridium acetoacetyl-CoA CoA reductase acetobutylicum (ketone reducing) hbd AAM14586.1 Clostridium acetoacetyl-CoA beijerinckii Hbd2 EDK34807.1 Clostridium acetoacetyl-CoA kluyveri Hbd1 EDK32512.1 Clostridium acetoacetyl-CoA kluyveri Msed_1423 YP_001191505 Metallosphaera 3-hydroxybutyryl- sedula CoA (suspected) Msed_0399 YP_001190500 Metallosphaera 3-hydroxybutyryl- sedula CoA (suspected) Msed_0389 YP_001190490 Metallosphaera 3-hydroxybutyryl- sedula CoA (suspected) Msed_1993 YP_001192057 Metallosphaera 3-hydroxybutyryl- sedula CoA (suspected) fadB P21177.2 Escherichia coli 3-oxoacyl-CoA fadJ P77399.1 Escherichia coli 3-oxoacyl-CoA H 1.2.1.b 3-hydroxybutyryl- 3-hydroxybutyr- 3-hydroxy- Ald AAT66436 Clostridium butyryl-CoA CoA aldehyde butyryl-CoA beijerinckii redcutase (aldehyde forming) sucD NP_904963.1 Porphyromonas succinyl-CoA gingivalis bphG BAA03892.1 Pseudomonas sp acetaldehyde, propionaldehyde, butyraldehyde, isobutyraldehyde and formaldehyde Msed_0709 YP_001190808.1 Metallosphaera malonyl-CoA sedula mcr NP_378167 Sulfolobus malonyl-CoA, tokodaii methylmalonyl- CoA I 1.1.1.c 3-hydroxybutyryl- 1,3-butanediol 3-hydroxy- adhE2 AAK09379.1 Clostridium butanoyl-CoA CoA butyryl-CoA acetobutylicum reductase (alcohol forming) mcr AAS20429.1 Chloroflexus malonyl-CoA aurantiacus FAR AAD38039.1 Simmondsia long chain acyl- chinensis CoA
TABLE-US-00125 TABLE 122 (Ref: FIG. 3) EC Gene GenBank ID (if Step class Desired substrate Desired product Enzyme name name available) Organism Known Substrates A 4.2.1.a 4-hydroxybutyryl- crotonyl-CoA 4-hydroxy- abfD YP_001396399.1 Clostridium 4-hydroxybutyryl- CoA butyryl-CoA kluyveri DSM 555 CoA dehydratase abfD P55792 Clostridium 4-hydroxybutyryl- aminobutyricum CoA abfD YP_001928843 Porphyromonas 4-hydroxybutyryl- gingivalis ATCC CoA 33277 B 4.2.1.a crotonyl-CoA 3-hydroxybutyryl- crotonase crt NP_349318.1 Clostridium 3-hydroxybutyryl- CoA acetobutylicum CoA crt1 YP_001393856 Clostridium 3-hydroxybutyryl- kluyveri DSM 555 CoA crt YP_001929291.1 Porphyromonas example based on gingivalis ATCC sequence similarity 33277 paaA NP_745427.1 Pseudomonas enoyl-CoA, cis- putida dihydrodiol derivative of phenylacetyl-CoA paaB NP_745426.1 Pseudomonas enoyl-CoA, cis- putida dihydrodiol derivative of phenylacetyl-CoA phaA ABF82233.1 Pseudomonas enoyl-CoA, cis- fluorescens dihydrodiol derivative of phenylacetyl-CoA phaB ABF82234.1 Pseudomonas enoyl-CoA, cis- fluorescens dihydrodiol derivative of phenylacetyl-CoA maoC NP_415905.1 Escherichia coli enoyl-CoA, cis- dihydrodiol derivative of phenylacetyl-CoA paaF NP_415911.1 Escherichia coli enoyl-CoA, cis- dihydrodiol derivative of phenylacetyl-CoA paaG NP_415912.1 Escherichia coli enoyl-CoA, cis-dihydrodiol derivative of phenylacetyl-CoA C 1.2.1.b 3-hydroxybutyryl- 3-hydroxy- 3-hydroxy- Ald AAT66436 Clostridium butyryl-CoA CoA butyraldehyde butyryl-CoA beijerinckii reductase (aldehyde forming) sucD NP_904963.1 Porphyromonas succinyl-CoA gingivalis bphG BAA03892.1 Pseudomonas sp acetaldehyde, propionaldehyde, butyraldehyde, isobutyraldehyde and formaldehyde Msed_0709 YP_001190808.1 Metallosphaera malonyl-CoA sedula mcr NP_378167 Sulfolobus tokodaii malonyl-CoA, methylmalonyl- CoA D 1.1.1.a 3-hydroxy- 1,3-butanediol 3-hydroxy- alrA BAB12273.1 Acinetobacter sp. C2-C14 aldehydes butyraldehyde butyraldehyde Strain M-1 reductase ADH2 NP_014032.1 Saccharymyces propionaldehyde, cerevisiae isobutyraldehyde, butyraldehyde, 2-methylbutyr- aldehyde, 3-methylbutyr- aldehyde, 2-phenylacet- aldehyde yqhD NP_417484.1 Escherichia coli acetaldehyde, malondialdehyde, propanaldehyde, butanaldehyde, and acrolein bdh I NP_349892.1 Clostridium butyraldehyde acetobutylicum bdh II NP_349891.1 Clostridium butyraldehyde acetobutylicum 4hbd YP_726053.1 Ralstonia eutropha succinate H16 semialdehyde ADHI AAR91477.1 Geobacillus ethanol, 1-butanol, thermoglucosidasius 1-pentanol, M10EXG 1-heptanol, 1-hexanol, 1-octanol, 2-propanol mmsb P28811.1 Pseudomonas 3-hydroxy- aeruginosa butyraldehyde, malonic semialdehyde, methylmalonate semialdehyde P84067 P84067 Thermus methylmalonate thermophilus semialdehyde E 1.1.1.c 3-hydroxybutyryl- 1,3-butanediol 3-hydroxy- adhE2 AAK09379.1 Clostridium butanoyl-CoA CoA butyryl-CoA acetobutylicum reductase (alcohol forming) mcr AAS20429.1 Chloroflexus malonyl-CoA aurantiacus FAR AAD38039.1 Simmondsia long chain acyl- chinensis CoA
Sequence Listing
[0605] The present specification is being filed with a computer readable form (CRF) copy of the Sequence Listing. The CRF entitled 12956-143_SEQLIST.txt, which was created on Jun. 15, 2012 and is 77,805 bytes in size, is identical to the paper copy of the Sequence Listing and is incorporated herein by reference in its entirety.
[0606] Throughout this application various publications have been referenced. The disclosures of these publications in their entireties, including GenBank and GI number publications, are hereby incorporated by reference in this application in order to more fully describe the state of the art to which this invention pertains. Although the invention has been described with reference to the examples and embodiments provided above, it should be understood that various modifications can be made without departing from the spirit of the invention.
Sequence CWU 1
1
1213525DNANocardia iowensis 1atggcagtgg attcaccgga tgagcggcta
cagcgccgca ttgcacagtt gtttgcagaa 60gatgagcagg tcaaggccgc acgtccgctc
gaagcggtga gcgcggcggt gagcgcgccc 120ggtatgcggc tggcgcagat
cgccgccact gttatggcgg gttacgccga ccgcccggcc 180gccgggcagc
gtgcgttcga actgaacacc gacgacgcga cgggccgcac ctcgctgcgg
240ttacttcccc gattcgagac catcacctat cgcgaactgt ggcagcgagt
cggcgaggtt 300gccgcggcct ggcatcatga tcccgagaac cccttgcgcg
caggtgattt cgtcgccctg 360ctcggcttca ccagcatcga ctacgccacc
ctcgacctgg ccgatatcca cctcggcgcg 420gttaccgtgc cgttgcaggc
cagcgcggcg gtgtcccagc tgatcgctat cctcaccgag 480acttcgccgc
ggctgctcgc ctcgaccccg gagcacctcg atgcggcggt cgagtgccta
540ctcgcgggca ccacaccgga acgactggtg gtcttcgact accaccccga
ggacgacgac 600cagcgtgcgg ccttcgaatc cgcccgccgc cgccttgccg
acgcgggcag cttggtgatc 660gtcgaaacgc tcgatgccgt gcgtgcccgg
ggccgcgact taccggccgc gccactgttc 720gttcccgaca ccgacgacga
cccgctggcc ctgctgatct acacctccgg cagcaccgga 780acgccgaagg
gcgcgatgta caccaatcgg ttggccgcca cgatgtggca ggggaactcg
840atgctgcagg ggaactcgca acgggtcggg atcaatctca actacatgcc
gatgagccac 900atcgccggtc gcatatcgct gttcggcgtg ctcgctcgcg
gtggcaccgc atacttcgcg 960gccaagagcg acatgtcgac actgttcgaa
gacatcggct tggtacgtcc caccgagatc 1020ttcttcgtcc cgcgcgtgtg
cgacatggtc ttccagcgct atcagagcga gctggaccgg 1080cgctcggtgg
cgggcgccga cctggacacg ctcgatcggg aagtgaaagc cgacctccgg
1140cagaactacc tcggtgggcg cttcctggtg gcggtcgtcg gcagcgcgcc
gctggccgcg 1200gagatgaaga cgttcatgga gtccgtcctc gatctgccac
tgcacgacgg gtacgggtcg 1260accgaggcgg gcgcaagcgt gctgctcgac
aaccagatcc agcggccgcc ggtgctcgat 1320tacaagctcg tcgacgtgcc
cgaactgggt tacttccgca ccgaccggcc gcatccgcgc 1380ggtgagctgt
tgttgaaggc ggagaccacg attccgggct actacaagcg gcccgaggtc
1440accgcggaga tcttcgacga ggacggcttc tacaagaccg gcgatatcgt
ggccgagctc 1500gagcacgatc ggctggtcta tgtcgaccgt cgcaacaatg
tgctcaaact gtcgcagggc 1560gagttcgtga ccgtcgccca tctcgaggcc
gtgttcgcca gcagcccgct gatccggcag 1620atcttcatct acggcagcag
cgaacgttcc tatctgctcg cggtgatcgt ccccaccgac 1680gacgcgctgc
gcggccgcga caccgccacc ttgaaatcgg cactggccga atcgattcag
1740cgcatcgcca aggacgcgaa cctgcagccc tacgagattc cgcgcgattt
cctgatcgag 1800accgagccgt tcaccatcgc caacggactg ctctccggca
tcgcgaagct gctgcgcccc 1860aatctgaagg aacgctacgg cgctcagctg
gagcagatgt acaccgatct cgcgacaggc 1920caggccgatg agctgctcgc
cctgcgccgc gaagccgccg acctgccggt gctcgaaacc 1980gtcagccggg
cagcgaaagc gatgctcggc gtcgcctccg ccgatatgcg tcccgacgcg
2040cacttcaccg acctgggcgg cgattccctt tccgcgctgt cgttctcgaa
cctgctgcac 2100gagatcttcg gggtcgaggt gccggtgggt gtcgtcgtca
gcccggcgaa cgagctgcgc 2160gatctggcga attacattga ggcggaacgc
aactcgggcg cgaagcgtcc caccttcacc 2220tcggtgcacg gcggcggttc
cgagatccgc gccgccgatc tgaccctcga caagttcatc 2280gatgcccgca
ccctggccgc cgccgacagc attccgcacg cgccggtgcc agcgcagacg
2340gtgctgctga ccggcgcgaa cggctacctc ggccggttcc tgtgcctgga
atggctggag 2400cggctggaca agacgggtgg cacgctgatc tgcgtcgtgc
gcggtagtga cgcggccgcg 2460gcccgtaaac ggctggactc ggcgttcgac
agcggcgatc ccggcctgct cgagcactac 2520cagcaactgg ccgcacggac
cctggaagtc ctcgccggtg atatcggcga cccgaatctc 2580ggtctggacg
acgcgacttg gcagcggttg gccgaaaccg tcgacctgat cgtccatccc
2640gccgcgttgg tcaaccacgt ccttccctac acccagctgt tcggccccaa
tgtcgtcggc 2700accgccgaaa tcgtccggtt ggcgatcacg gcgcggcgca
agccggtcac ctacctgtcg 2760accgtcggag tggccgacca ggtcgacccg
gcggagtatc aggaggacag cgacgtccgc 2820gagatgagcg cggtgcgcgt
cgtgcgcgag agttacgcca acggctacgg caacagcaag 2880tgggcggggg
aggtcctgct gcgcgaagca cacgatctgt gtggcttgcc ggtcgcggtg
2940ttccgttcgg acatgatcct ggcgcacagc cggtacgcgg gtcagctcaa
cgtccaggac 3000gtgttcaccc ggctgatcct cagcctggtc gccaccggca
tcgcgccgta ctcgttctac 3060cgaaccgacg cggacggcaa ccggcagcgg
gcccactatg acggcttgcc ggcggacttc 3120acggcggcgg cgatcaccgc
gctcggcatc caagccaccg aaggcttccg gacctacgac 3180gtgctcaatc
cgtacgacga tggcatctcc ctcgatgaat tcgtcgactg gctcgtcgaa
3240tccggccacc cgatccagcg catcaccgac tacagcgact ggttccaccg
tttcgagacg 3300gcgatccgcg cgctgccgga aaagcaacgc caggcctcgg
tgctgccgtt gctggacgcc 3360taccgcaacc cctgcccggc ggtccgcggc
gcgatactcc cggccaagga gttccaagcg 3420gcggtgcaaa cagccaaaat
cggtccggaa caggacatcc cgcatttgtc cgcgccactg 3480atcgataagt
acgtcagcga tctggaactg cttcagctgc tctaa 352521174PRTNocardia
iowensis 2Met Ala Val Asp Ser Pro Asp Glu Arg Leu Gln Arg Arg Ile
Ala Gln1 5 10 15Leu Phe Ala Glu Asp Glu Gln Val Lys Ala Ala Arg Pro
Leu Glu Ala 20 25 30Val Ser Ala Ala Val Ser Ala Pro Gly Met Arg Leu
Ala Gln Ile Ala 35 40 45Ala Thr Val Met Ala Gly Tyr Ala Asp Arg Pro
Ala Ala Gly Gln Arg 50 55 60Ala Phe Glu Leu Asn Thr Asp Asp Ala Thr
Gly Arg Thr Ser Leu Arg65 70 75 80Leu Leu Pro Arg Phe Glu Thr Ile
Thr Tyr Arg Glu Leu Trp Gln Arg 85 90 95Val Gly Glu Val Ala Ala Ala
Trp His His Asp Pro Glu Asn Pro Leu 100 105 110Arg Ala Gly Asp Phe
Val Ala Leu Leu Gly Phe Thr Ser Ile Asp Tyr 115 120 125Ala Thr Leu
Asp Leu Ala Asp Ile His Leu Gly Ala Val Thr Val Pro 130 135 140Leu
Gln Ala Ser Ala Ala Val Ser Gln Leu Ile Ala Ile Leu Thr Glu145 150
155 160Thr Ser Pro Arg Leu Leu Ala Ser Thr Pro Glu His Leu Asp Ala
Ala 165 170 175Val Glu Cys Leu Leu Ala Gly Thr Thr Pro Glu Arg Leu
Val Val Phe 180 185 190Asp Tyr His Pro Glu Asp Asp Asp Gln Arg Ala
Ala Phe Glu Ser Ala 195 200 205Arg Arg Arg Leu Ala Asp Ala Gly Ser
Leu Val Ile Val Glu Thr Leu 210 215 220Asp Ala Val Arg Ala Arg Gly
Arg Asp Leu Pro Ala Ala Pro Leu Phe225 230 235 240Val Pro Asp Thr
Asp Asp Asp Pro Leu Ala Leu Leu Ile Tyr Thr Ser 245 250 255Gly Ser
Thr Gly Thr Pro Lys Gly Ala Met Tyr Thr Asn Arg Leu Ala 260 265
270Ala Thr Met Trp Gln Gly Asn Ser Met Leu Gln Gly Asn Ser Gln Arg
275 280 285Val Gly Ile Asn Leu Asn Tyr Met Pro Met Ser His Ile Ala
Gly Arg 290 295 300Ile Ser Leu Phe Gly Val Leu Ala Arg Gly Gly Thr
Ala Tyr Phe Ala305 310 315 320Ala Lys Ser Asp Met Ser Thr Leu Phe
Glu Asp Ile Gly Leu Val Arg 325 330 335Pro Thr Glu Ile Phe Phe Val
Pro Arg Val Cys Asp Met Val Phe Gln 340 345 350Arg Tyr Gln Ser Glu
Leu Asp Arg Arg Ser Val Ala Gly Ala Asp Leu 355 360 365Asp Thr Leu
Asp Arg Glu Val Lys Ala Asp Leu Arg Gln Asn Tyr Leu 370 375 380Gly
Gly Arg Phe Leu Val Ala Val Val Gly Ser Ala Pro Leu Ala Ala385 390
395 400Glu Met Lys Thr Phe Met Glu Ser Val Leu Asp Leu Pro Leu His
Asp 405 410 415Gly Tyr Gly Ser Thr Glu Ala Gly Ala Ser Val Leu Leu
Asp Asn Gln 420 425 430Ile Gln Arg Pro Pro Val Leu Asp Tyr Lys Leu
Val Asp Val Pro Glu 435 440 445Leu Gly Tyr Phe Arg Thr Asp Arg Pro
His Pro Arg Gly Glu Leu Leu 450 455 460Leu Lys Ala Glu Thr Thr Ile
Pro Gly Tyr Tyr Lys Arg Pro Glu Val465 470 475 480Thr Ala Glu Ile
Phe Asp Glu Asp Gly Phe Tyr Lys Thr Gly Asp Ile 485 490 495Val Ala
Glu Leu Glu His Asp Arg Leu Val Tyr Val Asp Arg Arg Asn 500 505
510Asn Val Leu Lys Leu Ser Gln Gly Glu Phe Val Thr Val Ala His Leu
515 520 525Glu Ala Val Phe Ala Ser Ser Pro Leu Ile Arg Gln Ile Phe
Ile Tyr 530 535 540Gly Ser Ser Glu Arg Ser Tyr Leu Leu Ala Val Ile
Val Pro Thr Asp545 550 555 560Asp Ala Leu Arg Gly Arg Asp Thr Ala
Thr Leu Lys Ser Ala Leu Ala 565 570 575Glu Ser Ile Gln Arg Ile Ala
Lys Asp Ala Asn Leu Gln Pro Tyr Glu 580 585 590Ile Pro Arg Asp Phe
Leu Ile Glu Thr Glu Pro Phe Thr Ile Ala Asn 595 600 605Gly Leu Leu
Ser Gly Ile Ala Lys Leu Leu Arg Pro Asn Leu Lys Glu 610 615 620Arg
Tyr Gly Ala Gln Leu Glu Gln Met Tyr Thr Asp Leu Ala Thr Gly625 630
635 640Gln Ala Asp Glu Leu Leu Ala Leu Arg Arg Glu Ala Ala Asp Leu
Pro 645 650 655Val Leu Glu Thr Val Ser Arg Ala Ala Lys Ala Met Leu
Gly Val Ala 660 665 670Ser Ala Asp Met Arg Pro Asp Ala His Phe Thr
Asp Leu Gly Gly Asp 675 680 685Ser Leu Ser Ala Leu Ser Phe Ser Asn
Leu Leu His Glu Ile Phe Gly 690 695 700Val Glu Val Pro Val Gly Val
Val Val Ser Pro Ala Asn Glu Leu Arg705 710 715 720Asp Leu Ala Asn
Tyr Ile Glu Ala Glu Arg Asn Ser Gly Ala Lys Arg 725 730 735Pro Thr
Phe Thr Ser Val His Gly Gly Gly Ser Glu Ile Arg Ala Ala 740 745
750Asp Leu Thr Leu Asp Lys Phe Ile Asp Ala Arg Thr Leu Ala Ala Ala
755 760 765Asp Ser Ile Pro His Ala Pro Val Pro Ala Gln Thr Val Leu
Leu Thr 770 775 780Gly Ala Asn Gly Tyr Leu Gly Arg Phe Leu Cys Leu
Glu Trp Leu Glu785 790 795 800Arg Leu Asp Lys Thr Gly Gly Thr Leu
Ile Cys Val Val Arg Gly Ser 805 810 815Asp Ala Ala Ala Ala Arg Lys
Arg Leu Asp Ser Ala Phe Asp Ser Gly 820 825 830Asp Pro Gly Leu Leu
Glu His Tyr Gln Gln Leu Ala Ala Arg Thr Leu 835 840 845Glu Val Leu
Ala Gly Asp Ile Gly Asp Pro Asn Leu Gly Leu Asp Asp 850 855 860Ala
Thr Trp Gln Arg Leu Ala Glu Thr Val Asp Leu Ile Val His Pro865 870
875 880Ala Ala Leu Val Asn His Val Leu Pro Tyr Thr Gln Leu Phe Gly
Pro 885 890 895Asn Val Val Gly Thr Ala Glu Ile Val Arg Leu Ala Ile
Thr Ala Arg 900 905 910Arg Lys Pro Val Thr Tyr Leu Ser Thr Val Gly
Val Ala Asp Gln Val 915 920 925Asp Pro Ala Glu Tyr Gln Glu Asp Ser
Asp Val Arg Glu Met Ser Ala 930 935 940Val Arg Val Val Arg Glu Ser
Tyr Ala Asn Gly Tyr Gly Asn Ser Lys945 950 955 960Trp Ala Gly Glu
Val Leu Leu Arg Glu Ala His Asp Leu Cys Gly Leu 965 970 975Pro Val
Ala Val Phe Arg Ser Asp Met Ile Leu Ala His Ser Arg Tyr 980 985
990Ala Gly Gln Leu Asn Val Gln Asp Val Phe Thr Arg Leu Ile Leu Ser
995 1000 1005Leu Val Ala Thr Gly Ile Ala Pro Tyr Ser Phe Tyr Arg
Thr Asp 1010 1015 1020Ala Asp Gly Asn Arg Gln Arg Ala His Tyr Asp
Gly Leu Pro Ala 1025 1030 1035Asp Phe Thr Ala Ala Ala Ile Thr Ala
Leu Gly Ile Gln Ala Thr 1040 1045 1050Glu Gly Phe Arg Thr Tyr Asp
Val Leu Asn Pro Tyr Asp Asp Gly 1055 1060 1065Ile Ser Leu Asp Glu
Phe Val Asp Trp Leu Val Glu Ser Gly His 1070 1075 1080Pro Ile Gln
Arg Ile Thr Asp Tyr Ser Asp Trp Phe His Arg Phe 1085 1090 1095Glu
Thr Ala Ile Arg Ala Leu Pro Glu Lys Gln Arg Gln Ala Ser 1100 1105
1110Val Leu Pro Leu Leu Asp Ala Tyr Arg Asn Pro Cys Pro Ala Val
1115 1120 1125Arg Gly Ala Ile Leu Pro Ala Lys Glu Phe Gln Ala Ala
Val Gln 1130 1135 1140Thr Ala Lys Ile Gly Pro Glu Gln Asp Ile Pro
His Leu Ser Ala 1145 1150 1155Pro Leu Ile Asp Lys Tyr Val Ser Asp
Leu Glu Leu Leu Gln Leu 1160 1165 1170Leu3669DNAArtificial
SequenceDescription of Artificial Sequence Synthetic codon
optimized phosphpantetheine transferase polynucleotide 3atgattgaaa
ccattctgcc tgcaggcgtt gaaagcgcag aactgctgga atatccggaa 60gatctgaaag
cacatccggc agaagaacat ctgattgcca aaagcgttga aaaacgtcgt
120cgtgatttta ttggtgcacg tcattgtgca cgtctggcac tggcagaact
gggtgaacct 180ccggttgcaa ttggtaaagg tgaacgtggt gcaccgattt
ggcctcgtgg tgttgttggt 240agcctgaccc attgtgatgg ttatcgtgca
gcagcagttg cacataaaat gcgctttcgc 300agcattggta ttgatgcaga
accgcatgca accctgccgg aaggtgttct ggatagcgtt 360agcctgccgc
cggaacgtga atggctgaaa accaccgata gcgcactgca tctggatcgt
420ctgctgtttt gtgcaaaaga agccacctat aaagcctggt ggccgctgac
agcacgttgg 480ctgggttttg aagaagccca tattaccttt gaaattgaag
atggtagcgc agatagcggt 540aatggcacct ttcatagcga actgctggtt
ccgggtcaga ccaatgatgg tggtacaccg 600ctgctgagct ttgatggtcg
ttggctgatt gcagatggtt ttattctgac cgcaattgcc 660tatgcctaa
6694222PRTArtificial SequenceDescription of Artificial Sequence
Synthetic codon optimized phosphpantetheine transferase polypeptide
4Met Ile Glu Thr Ile Leu Pro Ala Gly Val Glu Ser Ala Glu Leu Leu1 5
10 15Glu Tyr Pro Glu Asp Leu Lys Ala His Pro Ala Glu Glu His Leu
Ile 20 25 30Ala Lys Ser Val Glu Lys Arg Arg Arg Asp Phe Ile Gly Ala
Arg His 35 40 45Cys Ala Arg Leu Ala Leu Ala Glu Leu Gly Glu Pro Pro
Val Ala Ile 50 55 60Gly Lys Gly Glu Arg Gly Ala Pro Ile Trp Pro Arg
Gly Val Val Gly65 70 75 80Ser Leu Thr His Cys Asp Gly Tyr Arg Ala
Ala Ala Val Ala His Lys 85 90 95Met Arg Phe Arg Ser Ile Gly Ile Asp
Ala Glu Pro His Ala Thr Leu 100 105 110Pro Glu Gly Val Leu Asp Ser
Val Ser Leu Pro Pro Glu Arg Glu Trp 115 120 125Leu Lys Thr Thr Asp
Ser Ala Leu His Leu Asp Arg Leu Leu Phe Cys 130 135 140Ala Lys Glu
Ala Thr Tyr Lys Ala Trp Trp Pro Leu Thr Ala Arg Trp145 150 155
160Leu Gly Phe Glu Glu Ala His Ile Thr Phe Glu Ile Glu Asp Gly Ser
165 170 175Ala Asp Ser Gly Asn Gly Thr Phe His Ser Glu Leu Leu Val
Pro Gly 180 185 190Gln Thr Asn Asp Gly Gly Thr Pro Leu Leu Ser Phe
Asp Gly Arg Trp 195 200 205Leu Ile Ala Asp Gly Phe Ile Leu Thr Ala
Ile Ala Tyr Ala 210 215 22053522DNAMycobacterium smegmatis
5atgaccagcg atgttcacga cgccacagac ggcgtcaccg aaaccgcact cgacgacgag
60cagtcgaccc gccgcatcgc cgagctgtac gccaccgatc ccgagttcgc cgccgccgca
120ccgttgcccg ccgtggtcga cgcggcgcac aaacccgggc tgcggctggc
agagatcctg 180cagaccctgt tcaccggcta cggtgaccgc ccggcgctgg
gataccgcgc ccgtgaactg 240gccaccgacg agggcgggcg caccgtgacg
cgtctgctgc cgcggttcga caccctcacc 300tacgcccagg tgtggtcgcg
cgtgcaagcg gtcgccgcgg ccctgcgcca caacttcgcg 360cagccgatct
accccggcga cgccgtcgcg acgatcggtt tcgcgagtcc cgattacctg
420acgctggatc tcgtatgcgc ctacctgggc ctcgtgagtg ttccgctgca
gcacaacgca 480ccggtcagcc ggctcgcccc gatcctggcc gaggtcgaac
cgcggatcct caccgtgagc 540gccgaatacc tcgacctcgc agtcgaatcc
gtgcgggacg tcaactcggt gtcgcagctc 600gtggtgttcg accatcaccc
cgaggtcgac gaccaccgcg acgcactggc ccgcgcgcgt 660gaacaactcg
ccggcaaggg catcgccgtc accaccctgg acgcgatcgc cgacgagggc
720gccgggctgc cggccgaacc gatctacacc gccgaccatg atcagcgcct
cgcgatgatc 780ctgtacacct cgggttccac cggcgcaccc aagggtgcga
tgtacaccga ggcgatggtg 840gcgcggctgt ggaccatgtc gttcatcacg
ggtgacccca cgccggtcat caacgtcaac 900ttcatgccgc tcaaccacct
gggcgggcgc atccccattt ccaccgccgt gcagaacggt 960ggaaccagtt
acttcgtacc ggaatccgac atgtccacgc tgttcgagga tctcgcgctg
1020gtgcgcccga ccgaactcgg cctggttccg cgcgtcgccg acatgctcta
ccagcaccac 1080ctcgccaccg tcgaccgcct ggtcacgcag ggcgccgacg
aactgaccgc cgagaagcag 1140gccggtgccg aactgcgtga gcaggtgctc
ggcggacgcg tgatcaccgg attcgtcagc 1200accgcaccgc tggccgcgga
gatgagggcg ttcctcgaca tcaccctggg cgcacacatc 1260gtcgacggct
acgggctcac cgagaccggc gccgtgacac gcgacggtgt gatcgtgcgg
1320ccaccggtga tcgactacaa gctgatcgac gttcccgaac tcggctactt
cagcaccgac 1380aagccctacc cgcgtggcga actgctggtc aggtcgcaaa
cgctgactcc cgggtactac 1440aagcgccccg aggtcaccgc gagcgtcttc
gaccgggacg gctactacca caccggcgac 1500gtcatggccg agaccgcacc
cgaccacctg gtgtacgtgg accgtcgcaa caacgtcctc 1560aaactcgcgc
agggcgagtt cgtggcggtc gccaacctgg aggcggtgtt ctccggcgcg
1620gcgctggtgc gccagatctt cgtgtacggc aacagcgagc gcagtttcct
tctggccgtg 1680gtggtcccga cgccggaggc gctcgagcag tacgatccgg
ccgcgctcaa ggccgcgctg 1740gccgactcgc tgcagcgcac cgcacgcgac
gccgaactgc aatcctacga ggtgccggcc 1800gatttcatcg tcgagaccga
gccgttcagc gccgccaacg ggctgctgtc gggtgtcgga 1860aaactgctgc
ggcccaacct caaagaccgc tacgggcagc gcctggagca gatgtacgcc
1920gatatcgcgg ccacgcaggc caaccagttg cgcgaactgc ggcgcgcggc
cgccacacaa 1980ccggtgatcg acaccctcac ccaggccgct gccacgatcc
tcggcaccgg gagcgaggtg 2040gcatccgacg cccacttcac cgacctgggc
ggggattccc tgtcggcgct gacactttcg 2100aacctgctga gcgatttctt
cggtttcgaa gttcccgtcg gcaccatcgt gaacccggcc 2160accaacctcg
cccaactcgc ccagcacatc gaggcgcagc gcaccgcggg tgaccgcagg
2220ccgagtttca ccaccgtgca cggcgcggac gccaccgaga tccgggcgag
tgagctgacc 2280ctggacaagt tcatcgacgc cgaaacgctc cgggccgcac
cgggtctgcc caaggtcacc 2340accgagccac ggacggtgtt gctctcgggc
gccaacggct ggctgggccg gttcctcacg 2400ttgcagtggc tggaacgcct
ggcacctgtc ggcggcaccc tcatcacgat cgtgcggggc 2460cgcgacgacg
ccgcggcccg cgcacggctg acccaggcct acgacaccga tcccgagttg
2520tcccgccgct tcgccgagct ggccgaccgc cacctgcggg tggtcgccgg
tgacatcggc 2580gacccgaatc tgggcctcac acccgagatc tggcaccggc
tcgccgccga ggtcgacctg 2640gtggtgcatc cggcagcgct ggtcaaccac
gtgctcccct accggcagct gttcggcccc 2700aacgtcgtgg gcacggccga
ggtgatcaag ctggccctca ccgaacggat caagcccgtc 2760acgtacctgt
ccaccgtgtc ggtggccatg gggatccccg acttcgagga ggacggcgac
2820atccggaccg tgagcccggt gcgcccgctc gacggcggat acgccaacgg
ctacggcaac 2880agcaagtggg ccggcgaggt gctgctgcgg gaggcccacg
atctgtgcgg gctgcccgtg 2940gcgacgttcc gctcggacat gatcctggcg
catccgcgct accgcggtca ggtcaacgtg 3000ccagacatgt tcacgcgact
cctgttgagc ctcttgatca ccggcgtcgc gccgcggtcg 3060ttctacatcg
gagacggtga gcgcccgcgg gcgcactacc ccggcctgac ggtcgatttc
3120gtggccgagg cggtcacgac gctcggcgcg cagcagcgcg agggatacgt
gtcctacgac 3180gtgatgaacc cgcacgacga cgggatctcc ctggatgtgt
tcgtggactg gctgatccgg 3240gcgggccatc cgatcgaccg ggtcgacgac
tacgacgact gggtgcgtcg gttcgagacc 3300gcgttgaccg cgcttcccga
gaagcgccgc gcacagaccg tactgccgct gctgcacgcg 3360ttccgcgctc
cgcaggcacc gttgcgcggc gcacccgaac ccacggaggt gttccacgcc
3420gcggtgcgca ccgcgaaggt gggcccggga gacatcccgc acctcgacga
ggcgctgatc 3480gacaagtaca tacgcgatct gcgtgagttc ggtctgatct aa
352261173PRTMycobacterium smegmatis 6Met Thr Ser Asp Val His Asp
Ala Thr Asp Gly Val Thr Glu Thr Ala1 5 10 15Leu Asp Asp Glu Gln Ser
Thr Arg Arg Ile Ala Glu Leu Tyr Ala Thr 20 25 30Asp Pro Glu Phe Ala
Ala Ala Ala Pro Leu Pro Ala Val Val Asp Ala 35 40 45Ala His Lys Pro
Gly Leu Arg Leu Ala Glu Ile Leu Gln Thr Leu Phe 50 55 60Thr Gly Tyr
Gly Asp Arg Pro Ala Leu Gly Tyr Arg Ala Arg Glu Leu65 70 75 80Ala
Thr Asp Glu Gly Gly Arg Thr Val Thr Arg Leu Leu Pro Arg Phe 85 90
95Asp Thr Leu Thr Tyr Ala Gln Val Trp Ser Arg Val Gln Ala Val Ala
100 105 110Ala Ala Leu Arg His Asn Phe Ala Gln Pro Ile Tyr Pro Gly
Asp Ala 115 120 125Val Ala Thr Ile Gly Phe Ala Ser Pro Asp Tyr Leu
Thr Leu Asp Leu 130 135 140Val Cys Ala Tyr Leu Gly Leu Val Ser Val
Pro Leu Gln His Asn Ala145 150 155 160Pro Val Ser Arg Leu Ala Pro
Ile Leu Ala Glu Val Glu Pro Arg Ile 165 170 175Leu Thr Val Ser Ala
Glu Tyr Leu Asp Leu Ala Val Glu Ser Val Arg 180 185 190Asp Val Asn
Ser Val Ser Gln Leu Val Val Phe Asp His His Pro Glu 195 200 205Val
Asp Asp His Arg Asp Ala Leu Ala Arg Ala Arg Glu Gln Leu Ala 210 215
220Gly Lys Gly Ile Ala Val Thr Thr Leu Asp Ala Ile Ala Asp Glu
Gly225 230 235 240Ala Gly Leu Pro Ala Glu Pro Ile Tyr Thr Ala Asp
His Asp Gln Arg 245 250 255Leu Ala Met Ile Leu Tyr Thr Ser Gly Ser
Thr Gly Ala Pro Lys Gly 260 265 270Ala Met Tyr Thr Glu Ala Met Val
Ala Arg Leu Trp Thr Met Ser Phe 275 280 285Ile Thr Gly Asp Pro Thr
Pro Val Ile Asn Val Asn Phe Met Pro Leu 290 295 300Asn His Leu Gly
Gly Arg Ile Pro Ile Ser Thr Ala Val Gln Asn Gly305 310 315 320Gly
Thr Ser Tyr Phe Val Pro Glu Ser Asp Met Ser Thr Leu Phe Glu 325 330
335Asp Leu Ala Leu Val Arg Pro Thr Glu Leu Gly Leu Val Pro Arg Val
340 345 350Ala Asp Met Leu Tyr Gln His His Leu Ala Thr Val Asp Arg
Leu Val 355 360 365Thr Gln Gly Ala Asp Glu Leu Thr Ala Glu Lys Gln
Ala Gly Ala Glu 370 375 380Leu Arg Glu Gln Val Leu Gly Gly Arg Val
Ile Thr Gly Phe Val Ser385 390 395 400Thr Ala Pro Leu Ala Ala Glu
Met Arg Ala Phe Leu Asp Ile Thr Leu 405 410 415Gly Ala His Ile Val
Asp Gly Tyr Gly Leu Thr Glu Thr Gly Ala Val 420 425 430Thr Arg Asp
Gly Val Ile Val Arg Pro Pro Val Ile Asp Tyr Lys Leu 435 440 445Ile
Asp Val Pro Glu Leu Gly Tyr Phe Ser Thr Asp Lys Pro Tyr Pro 450 455
460Arg Gly Glu Leu Leu Val Arg Ser Gln Thr Leu Thr Pro Gly Tyr
Tyr465 470 475 480Lys Arg Pro Glu Val Thr Ala Ser Val Phe Asp Arg
Asp Gly Tyr Tyr 485 490 495His Thr Gly Asp Val Met Ala Glu Thr Ala
Pro Asp His Leu Val Tyr 500 505 510Val Asp Arg Arg Asn Asn Val Leu
Lys Leu Ala Gln Gly Glu Phe Val 515 520 525Ala Val Ala Asn Leu Glu
Ala Val Phe Ser Gly Ala Ala Leu Val Arg 530 535 540Gln Ile Phe Val
Tyr Gly Asn Ser Glu Arg Ser Phe Leu Leu Ala Val545 550 555 560Val
Val Pro Thr Pro Glu Ala Leu Glu Gln Tyr Asp Pro Ala Ala Leu 565 570
575Lys Ala Ala Leu Ala Asp Ser Leu Gln Arg Thr Ala Arg Asp Ala Glu
580 585 590Leu Gln Ser Tyr Glu Val Pro Ala Asp Phe Ile Val Glu Thr
Glu Pro 595 600 605Phe Ser Ala Ala Asn Gly Leu Leu Ser Gly Val Gly
Lys Leu Leu Arg 610 615 620Pro Asn Leu Lys Asp Arg Tyr Gly Gln Arg
Leu Glu Gln Met Tyr Ala625 630 635 640Asp Ile Ala Ala Thr Gln Ala
Asn Gln Leu Arg Glu Leu Arg Arg Ala 645 650 655Ala Ala Thr Gln Pro
Val Ile Asp Thr Leu Thr Gln Ala Ala Ala Thr 660 665 670Ile Leu Gly
Thr Gly Ser Glu Val Ala Ser Asp Ala His Phe Thr Asp 675 680 685Leu
Gly Gly Asp Ser Leu Ser Ala Leu Thr Leu Ser Asn Leu Leu Ser 690 695
700Asp Phe Phe Gly Phe Glu Val Pro Val Gly Thr Ile Val Asn Pro
Ala705 710 715 720Thr Asn Leu Ala Gln Leu Ala Gln His Ile Glu Ala
Gln Arg Thr Ala 725 730 735Gly Asp Arg Arg Pro Ser Phe Thr Thr Val
His Gly Ala Asp Ala Thr 740 745 750Glu Ile Arg Ala Ser Glu Leu Thr
Leu Asp Lys Phe Ile Asp Ala Glu 755 760 765Thr Leu Arg Ala Ala Pro
Gly Leu Pro Lys Val Thr Thr Glu Pro Arg 770 775 780Thr Val Leu Leu
Ser Gly Ala Asn Gly Trp Leu Gly Arg Phe Leu Thr785 790 795 800Leu
Gln Trp Leu Glu Arg Leu Ala Pro Val Gly Gly Thr Leu Ile Thr 805 810
815Ile Val Arg Gly Arg Asp Asp Ala Ala Ala Arg Ala Arg Leu Thr Gln
820 825 830Ala Tyr Asp Thr Asp Pro Glu Leu Ser Arg Arg Phe Ala Glu
Leu Ala 835 840 845Asp Arg His Leu Arg Val Val Ala Gly Asp Ile Gly
Asp Pro Asn Leu 850 855 860Gly Leu Thr Pro Glu Ile Trp His Arg Leu
Ala Ala Glu Val Asp Leu865 870 875 880Val Val His Pro Ala Ala Leu
Val Asn His Val Leu Pro Tyr Arg Gln 885 890 895Leu Phe Gly Pro Asn
Val Val Gly Thr Ala Glu Val Ile Lys Leu Ala 900 905 910Leu Thr Glu
Arg Ile Lys Pro Val Thr Tyr Leu Ser Thr Val Ser Val 915 920 925Ala
Met Gly Ile Pro Asp Phe Glu Glu Asp Gly Asp Ile Arg Thr Val 930 935
940Ser Pro Val Arg Pro Leu Asp Gly Gly Tyr Ala Asn Gly Tyr Gly
Asn945 950 955 960Ser Lys Trp Ala Gly Glu Val Leu Leu Arg Glu Ala
His Asp Leu Cys 965 970 975Gly Leu Pro Val Ala Thr Phe Arg Ser Asp
Met Ile Leu Ala His Pro 980 985 990Arg Tyr Arg Gly Gln Val Asn Val
Pro Asp Met Phe Thr Arg Leu Leu 995 1000 1005Leu Ser Leu Leu Ile
Thr Gly Val Ala Pro Arg Ser Phe Tyr Ile 1010 1015 1020Gly Asp Gly
Glu Arg Pro Arg Ala His Tyr Pro Gly Leu Thr Val 1025 1030 1035Asp
Phe Val Ala Glu Ala Val Thr Thr Leu Gly Ala Gln Gln Arg 1040 1045
1050Glu Gly Tyr Val Ser Tyr Asp Val Met Asn Pro His Asp Asp Gly
1055 1060 1065Ile Ser Leu Asp Val Phe Val Asp Trp Leu Ile Arg Ala
Gly His 1070 1075 1080Pro Ile Asp Arg Val Asp Asp Tyr Asp Asp Trp
Val Arg Arg Phe 1085 1090 1095Glu Thr Ala Leu Thr Ala Leu Pro Glu
Lys Arg Arg Ala Gln Thr 1100 1105 1110Val Leu Pro Leu Leu His Ala
Phe Arg Ala Pro Gln Ala Pro Leu 1115 1120 1125Arg Gly Ala Pro Glu
Pro Thr Glu Val Phe His Ala Ala Val Arg 1130 1135 1140Thr Ala Lys
Val Gly Pro Gly Asp Ile Pro His Leu Asp Glu Ala 1145 1150 1155Leu
Ile Asp Lys Tyr Ile Arg Asp Leu Arg Glu Phe Gly Leu Ile 1160 1165
117073522DNAMycobacterium avium 7atgtcgactg ccacccatga cgaacgactc
gaccgtcgcg tccacgaact catcgccacc 60gacccgcaat tcgccgccgc ccaacccgac
ccggcgatca ccgccgccct cgaacagccc 120gggctgcggc tgccgcagat
catccgcacc gtgctcgacg gctacgccga ccggccggcg 180ctgggacagc
gcgtggtgga gttcgtcacg gacgccaaga ccgggcgcac gtcggcgcag
240ctgctccccc gcttcgagac catcacgtac agcgaagtag cgcagcgtgt
ttcggcgctg 300ggccgcgccc tgtccgacga cgcggtgcac cccggcgacc
gggtgtgcgt gctgggcttc 360aacagcgtcg actacgccac catcgacatg
gcgctgggcg ccatcggcgc cgtctcggtg 420ccgctgcaga ccagcgcggc
aatcagctcg ctgcagccga tcgtggccga gaccgagccc 480accctgatcg
cgtccagcgt gaaccagctg tccgacgcgg tgcagctgat caccggcgcc
540gagcaggcgc ccacccggct ggtggtgttc gactaccacc cgcaggtcga
cgaccagcgc 600gaggccgtcc aggacgccgc ggcgcggctg tccagcaccg
gcgtggccgt ccagacgctg 660gccgagctgc tggagcgcgg caaggacctg
cccgccgtcg cggagccgcc cgccgacgag 720gactcgctgg ccctgctgat
ctacacctcc gggtccaccg gcgcccccaa gggcgcgatg 780tacccacaga
gcaacgtcgg caagatgtgg cgccgcggca gcaagaactg gttcggcgag
840agcgccgcgt cgatcaccct gaacttcatg ccgatgagcc acgtgatggg
ccgaagcatc 900ctctacggca cgctgggcaa cggcggcacc gcctacttcg
ccgcccgcag cgacctgtcc 960accctgcttg aggacctcga gctggtgcgg
cccaccgagc tcaacttcgt cccgcggatc 1020tgggagacgc tgtacggcga
attccagcgt caggtcgagc ggcggctctc cgaggccggg 1080gacgccggcg
aacgtcgcgc cgtcgaggcc gaggtgctgg ccgagcagcg ccagtacctg
1140ctgggcgggc ggttcacctt cgcgatgacg ggctcggcgc ccatctcgcc
ggagctgcgc 1200aactgggtcg agtcgctgct cgaaatgcac ctgatggacg
gctacggctc caccgaggcc 1260ggaatggtgt tgttcgacgg ggagattcag
cgcccgccgg tgatcgacta caagctggtc 1320gacgtgccgg acctgggcta
cttcagcacc gaccggccgc atccgcgcgg cgagctgctg 1380ctgcgcaccg
agaacatgtt cccgggctac tacaagcggg ccgaaaccac cgcgggcgtc
1440ttcgacgagg acggctacta ccgcaccggc gacgtgttcg ccgagatcgc
cccggaccgg 1500ctggtctacg tcgaccgccg caacaacgtg ctcaagctgg
cgcagggcga attcgtcacg 1560ctggccaagc tggaggcggt gttcggcaac
agcccgctga tccgccagat ctacgtctac 1620ggcaacagcg cccagcccta
cctgctggcg gtcgtggtgc ccaccgagga ggcgctggcc 1680tcgggtgacc
ccgagacgct caagcccaag atcgccgact cgctgcagca ggtcgccaag
1740gaggccggcc tgcagtccta cgaggtgccg cgcgacttca tcatcgagac
caccccgttc 1800agcctggaaa acggtctgct gaccgggatc cggaagctgg
cgtggccgaa actgaagcag 1860cactacgggg aacggctgga gcagatgtac
gccgacctgg ccgccggaca ggccaacgag 1920ctggccgagc tgcgccgcaa
cggtgcccag gcgccggtgt tgcagaccgt gagccgcgcc 1980gcgggcgcca
tgctgggttc ggccgcctcc gacctgtccc ccgacgccca cttcaccgat
2040ctgggcggag actcgttgtc ggcgttgaca ttcggcaacc tgctgcgcga
gatcttcgac 2100gtcgacgtgc cggtaggcgt gatcgtcagc ccggccaacg
acctggcggc catcgcgagc 2160tacatcgagg ccgagcggca gggcagcaag
cgcccgacgt tcgcctcggt gcacggccgg 2220gacgcgaccg tggtgcgcgc
cgccgacctg acgctggaca agttcctcga cgccgagacg 2280ctggccgccg
cgccgaacct gcccaagccg gccaccgagg tgcgcaccgt gctgctgacc
2340ggcgccaccg gcttcctggg ccgctacctg gccctggaat ggctggagcg
gatggacatg 2400gtggacggca aggtcatcgc cctggtccgg gcccgctccg
acgaggaggc acgcgcccgg 2460ctggacaaga ccttcgacag cggcgacccg
aaactgctcg cgcactacca gcagctggcc 2520gccgatcacc tggaggtcat
cgccggcgac aagggcgagg ccaatctggg cctgggccaa 2580gacgtttggc
aacgactggc cgacacggtc gacgtgatcg tcgaccccgc cgcgctggtc
2640aaccacgtgt tgccgtacag cgagctgttc gggcccaacg ccctgggcac
cgcggagctg 2700atccggctgg cgctgacgtc caagcagaag ccgtacacct
acgtgtccac catcggcgtg 2760ggcgaccaga tcgagccggg caagttcgtc
gagaacgccg acatccggca gatgagcgcc 2820acccgggcga tcaacgacag
ctacgccaac ggctatggca acagcaagtg ggccggcgag 2880gtgctgctgc
gcgaggcgca cgacctgtgc gggctgcccg tcgcggtgtt ccgctgcgac
2940atgatcctgg ccgacaccac gtatgccggg cagctcaacc tgccggacat
gttcacccgg 3000ctgatgctga gcctggtggc caccgggatc gcgcccggct
cgttctacga gctcgacgcc 3060gacggcaacc ggcagcgggc gcactacgac
ggcctgccgg tcgagttcat cgccgcggcg 3120atctcgacgc tgggttcgca
gatcaccgac agcgacaccg gcttccagac ctaccacgtg 3180atgaacccct
acgatgacgg cgtcggtctg gacgagtacg tcgattggct ggtggacgcc
3240ggctattcga tcgagcggat cgccgactac tccgaatggc tgcggcggtt
cgagacctcg 3300ctgcgggccc tgccggaccg gcagcgccag tactcgctgc
tgccgctgct gcacaactac 3360cgcacgccgg agaagccgat caacgggtcg
atagctccca ccgacgtgtt ccgggcagcg 3420gtgcaggagg cgaaaatcgg
ccccgacaaa gacattccgc acgtgtcgcc gccggtcatc 3480gtcaagtaca
tcaccgacct gcagctgctc gggctgctct aa 352281173PRTMycobacterium avium
8Met Ser Thr Ala Thr His Asp Glu Arg Leu Asp Arg Arg Val His Glu1 5
10 15Leu Ile Ala Thr Asp Pro Gln Phe Ala Ala Ala Gln Pro Asp Pro
Ala 20 25 30Ile Thr Ala Ala Leu Glu Gln Pro Gly Leu Arg Leu Pro Gln
Ile Ile 35 40 45Arg Thr Val Leu Asp Gly Tyr Ala Asp Arg Pro Ala Leu
Gly Gln Arg 50 55 60Val Val Glu Phe Val Thr Asp Ala Lys Thr Gly Arg
Thr Ser Ala Gln65 70 75 80Leu Leu Pro Arg Phe Glu Thr Ile Thr Tyr
Ser Glu Val Ala Gln Arg 85 90 95Val Ser Ala Leu Gly Arg Ala Leu Ser
Asp Asp Ala Val His Pro Gly 100 105 110Asp Arg Val Cys Val Leu Gly
Phe Asn Ser Val Asp Tyr Ala Thr Ile 115 120 125Asp Met Ala Leu Gly
Ala Ile Gly Ala Val Ser Val Pro Leu Gln Thr 130 135 140Ser Ala Ala
Ile Ser Ser Leu Gln Pro Ile Val Ala Glu Thr Glu Pro145 150 155
160Thr Leu Ile Ala Ser Ser Val Asn Gln Leu Ser Asp Ala Val Gln Leu
165 170 175Ile Thr Gly Ala Glu Gln Ala Pro Thr Arg Leu Val Val Phe
Asp Tyr 180 185 190His Pro Gln Val Asp Asp Gln Arg Glu Ala Val Gln
Asp Ala Ala Ala 195 200 205Arg Leu Ser Ser Thr Gly Val Ala Val Gln
Thr Leu Ala Glu Leu Leu 210 215 220Glu Arg Gly Lys Asp Leu Pro Ala
Val Ala Glu Pro Pro Ala Asp Glu225 230 235 240Asp Ser Leu Ala Leu
Leu Ile Tyr Thr Ser Gly Ser Thr Gly Ala Pro 245 250 255Lys Gly Ala
Met Tyr Pro Gln Ser Asn Val Gly Lys Met Trp Arg Arg 260 265 270Gly
Ser Lys Asn Trp Phe Gly Glu Ser Ala Ala Ser Ile Thr Leu Asn 275 280
285Phe Met Pro Met Ser His Val Met Gly Arg Ser Ile Leu Tyr Gly Thr
290 295 300Leu Gly Asn Gly Gly Thr Ala Tyr Phe Ala Ala Arg Ser Asp
Leu Ser305 310 315 320Thr Leu Leu Glu Asp Leu Glu Leu Val Arg Pro
Thr Glu Leu Asn Phe 325 330 335Val Pro Arg Ile Trp Glu Thr Leu Tyr
Gly Glu Phe Gln Arg Gln Val 340 345 350Glu Arg Arg Leu Ser Glu Ala
Gly Asp Ala Gly Glu Arg Arg Ala Val 355 360 365Glu Ala Glu Val Leu
Ala Glu Gln Arg Gln Tyr Leu Leu Gly Gly Arg 370 375 380Phe Thr Phe
Ala Met Thr Gly Ser Ala Pro Ile Ser Pro Glu Leu Arg385 390 395
400Asn Trp Val Glu Ser Leu Leu Glu Met His Leu Met Asp Gly Tyr Gly
405 410 415Ser Thr Glu Ala Gly Met Val Leu Phe Asp Gly Glu Ile Gln
Arg Pro 420 425 430Pro Val Ile Asp Tyr Lys Leu Val Asp Val Pro Asp
Leu Gly Tyr Phe 435
440 445Ser Thr Asp Arg Pro His Pro Arg Gly Glu Leu Leu Leu Arg Thr
Glu 450 455 460Asn Met Phe Pro Gly Tyr Tyr Lys Arg Ala Glu Thr Thr
Ala Gly Val465 470 475 480Phe Asp Glu Asp Gly Tyr Tyr Arg Thr Gly
Asp Val Phe Ala Glu Ile 485 490 495Ala Pro Asp Arg Leu Val Tyr Val
Asp Arg Arg Asn Asn Val Leu Lys 500 505 510Leu Ala Gln Gly Glu Phe
Val Thr Leu Ala Lys Leu Glu Ala Val Phe 515 520 525Gly Asn Ser Pro
Leu Ile Arg Gln Ile Tyr Val Tyr Gly Asn Ser Ala 530 535 540Gln Pro
Tyr Leu Leu Ala Val Val Val Pro Thr Glu Glu Ala Leu Ala545 550 555
560Ser Gly Asp Pro Glu Thr Leu Lys Pro Lys Ile Ala Asp Ser Leu Gln
565 570 575Gln Val Ala Lys Glu Ala Gly Leu Gln Ser Tyr Glu Val Pro
Arg Asp 580 585 590Phe Ile Ile Glu Thr Thr Pro Phe Ser Leu Glu Asn
Gly Leu Leu Thr 595 600 605Gly Ile Arg Lys Leu Ala Trp Pro Lys Leu
Lys Gln His Tyr Gly Glu 610 615 620Arg Leu Glu Gln Met Tyr Ala Asp
Leu Ala Ala Gly Gln Ala Asn Glu625 630 635 640Leu Ala Glu Leu Arg
Arg Asn Gly Ala Gln Ala Pro Val Leu Gln Thr 645 650 655Val Ser Arg
Ala Ala Gly Ala Met Leu Gly Ser Ala Ala Ser Asp Leu 660 665 670Ser
Pro Asp Ala His Phe Thr Asp Leu Gly Gly Asp Ser Leu Ser Ala 675 680
685Leu Thr Phe Gly Asn Leu Leu Arg Glu Ile Phe Asp Val Asp Val Pro
690 695 700Val Gly Val Ile Val Ser Pro Ala Asn Asp Leu Ala Ala Ile
Ala Ser705 710 715 720Tyr Ile Glu Ala Glu Arg Gln Gly Ser Lys Arg
Pro Thr Phe Ala Ser 725 730 735Val His Gly Arg Asp Ala Thr Val Val
Arg Ala Ala Asp Leu Thr Leu 740 745 750Asp Lys Phe Leu Asp Ala Glu
Thr Leu Ala Ala Ala Pro Asn Leu Pro 755 760 765Lys Pro Ala Thr Glu
Val Arg Thr Val Leu Leu Thr Gly Ala Thr Gly 770 775 780Phe Leu Gly
Arg Tyr Leu Ala Leu Glu Trp Leu Glu Arg Met Asp Met785 790 795
800Val Asp Gly Lys Val Ile Ala Leu Val Arg Ala Arg Ser Asp Glu Glu
805 810 815Ala Arg Ala Arg Leu Asp Lys Thr Phe Asp Ser Gly Asp Pro
Lys Leu 820 825 830Leu Ala His Tyr Gln Gln Leu Ala Ala Asp His Leu
Glu Val Ile Ala 835 840 845Gly Asp Lys Gly Glu Ala Asn Leu Gly Leu
Gly Gln Asp Val Trp Gln 850 855 860Arg Leu Ala Asp Thr Val Asp Val
Ile Val Asp Pro Ala Ala Leu Val865 870 875 880Asn His Val Leu Pro
Tyr Ser Glu Leu Phe Gly Pro Asn Ala Leu Gly 885 890 895Thr Ala Glu
Leu Ile Arg Leu Ala Leu Thr Ser Lys Gln Lys Pro Tyr 900 905 910Thr
Tyr Val Ser Thr Ile Gly Val Gly Asp Gln Ile Glu Pro Gly Lys 915 920
925Phe Val Glu Asn Ala Asp Ile Arg Gln Met Ser Ala Thr Arg Ala Ile
930 935 940Asn Asp Ser Tyr Ala Asn Gly Tyr Gly Asn Ser Lys Trp Ala
Gly Glu945 950 955 960Val Leu Leu Arg Glu Ala His Asp Leu Cys Gly
Leu Pro Val Ala Val 965 970 975Phe Arg Cys Asp Met Ile Leu Ala Asp
Thr Thr Tyr Ala Gly Gln Leu 980 985 990Asn Leu Pro Asp Met Phe Thr
Arg Leu Met Leu Ser Leu Val Ala Thr 995 1000 1005Gly Ile Ala Pro
Gly Ser Phe Tyr Glu Leu Asp Ala Asp Gly Asn 1010 1015 1020Arg Gln
Arg Ala His Tyr Asp Gly Leu Pro Val Glu Phe Ile Ala 1025 1030
1035Ala Ala Ile Ser Thr Leu Gly Ser Gln Ile Thr Asp Ser Asp Thr
1040 1045 1050Gly Phe Gln Thr Tyr His Val Met Asn Pro Tyr Asp Asp
Gly Val 1055 1060 1065Gly Leu Asp Glu Tyr Val Asp Trp Leu Val Asp
Ala Gly Tyr Ser 1070 1075 1080Ile Glu Arg Ile Ala Asp Tyr Ser Glu
Trp Leu Arg Arg Phe Glu 1085 1090 1095Thr Ser Leu Arg Ala Leu Pro
Asp Arg Gln Arg Gln Tyr Ser Leu 1100 1105 1110Leu Pro Leu Leu His
Asn Tyr Arg Thr Pro Glu Lys Pro Ile Asn 1115 1120 1125Gly Ser Ile
Ala Pro Thr Asp Val Phe Arg Ala Ala Val Gln Glu 1130 1135 1140Ala
Lys Ile Gly Pro Asp Lys Asp Ile Pro His Val Ser Pro Pro 1145 1150
1155Val Ile Val Lys Tyr Ile Thr Asp Leu Gln Leu Leu Gly Leu Leu
1160 1165 117093525DNAMycobacterium marinum 9atgtcgccaa tcacgcgtga
agagcggctc gagcgccgca tccaggacct ctacgccaac 60gacccgcagt tcgccgccgc
caaacccgcc acggcgatca ccgcagcaat cgagcggccg 120ggtctaccgc
taccccagat catcgagacc gtcatgaccg gatacgccga tcggccggct
180ctcgctcagc gctcggtcga attcgtgacc gacgccggca ccggccacac
cacgctgcga 240ctgctccccc acttcgaaac catcagctac ggcgagcttt
gggaccgcat cagcgcactg 300gccgacgtgc tcagcaccga acagacggtg
aaaccgggcg accgggtctg cttgttgggc 360ttcaacagcg tcgactacgc
cacgatcgac atgactttgg cgcggctggg cgcggtggcc 420gtaccactgc
agaccagcgc ggcgataacc cagctgcagc cgatcgtcgc cgagacccag
480cccaccatga tcgcggccag cgtcgacgca ctcgctgacg ccaccgaatt
ggctctgtcc 540ggtcagaccg ctacccgagt cctggtgttc gaccaccacc
ggcaggttga cgcacaccgc 600gcagcggtcg aatccgcccg ggagcgcctg
gccggctcgg cggtcgtcga aaccctggcc 660gaggccatcg cgcgcggcga
cgtgccccgc ggtgcgtccg ccggctcggc gcccggcacc 720gatgtgtccg
acgactcgct cgcgctactg atctacacct cgggcagcac gggtgcgccc
780aagggcgcga tgtacccccg acgcaacgtt gcgaccttct ggcgcaagcg
cacctggttc 840gaaggcggct acgagccgtc gatcacgctg aacttcatgc
caatgagcca cgtcatgggc 900cgccaaatcc tgtacggcac gctgtgcaat
ggcggcaccg cctacttcgt ggcgaaaagc 960gatctctcca ccttgttcga
agacctggcg ctggtgcggc ccaccgagct gaccttcgtg 1020ccgcgcgtgt
gggacatggt gttcgacgag tttcagagtg aggtcgaccg ccgcctggtc
1080gacggcgccg accgggtcgc gctcgaagcc caggtcaagg ccgagatacg
caacgacgtg 1140ctcggtggac ggtataccag cgcactgacc ggctccgccc
ctatctccga cgagatgaag 1200gcgtgggtcg aggagctgct cgacatgcat
ctggtcgagg gctacggctc caccgaggcc 1260gggatgatcc tgatcgacgg
agccattcgg cgcccggcgg tactcgacta caagctggtc 1320gatgttcccg
acctgggtta cttcctgacc gaccggccac atccgcgggg cgagttgctg
1380gtcaagaccg atagtttgtt cccgggctac taccagcgag ccgaagtcac
cgccgacgtg 1440ttcgatgctg acggcttcta ccggaccggc gacatcatgg
ccgaggtcgg ccccgaacag 1500ttcgtgtacc tcgaccgccg caacaacgtg
ttgaagctgt cgcagggcga gttcgtcacc 1560gtctccaaac tcgaagcggt
gtttggcgac agcccactgg tacggcagat ctacatctac 1620ggcaacagcg
cccgtgccta cctgttggcg gtgatcgtcc ccacccagga ggcgctggac
1680gccgtgcctg tcgaggagct caaggcgcgg ctgggcgact cgctgcaaga
ggtcgcaaag 1740gccgccggcc tgcagtccta cgagatcccg cgcgacttca
tcatcgaaac aacaccatgg 1800acgctggaga acggcctgct caccggcatc
cgcaagttgg ccaggccgca gctgaaaaag 1860cattacggcg agcttctcga
gcagatctac acggacctgg cacacggcca ggccgacgaa 1920ctgcgctcgc
tgcgccaaag cggtgccgat gcgccggtgc tggtgacggt gtgccgtgcg
1980gcggccgcgc tgttgggcgg cagcgcctct gacgtccagc ccgatgcgca
cttcaccgat 2040ttgggcggcg actcgctgtc ggcgctgtcg ttcaccaacc
tgctgcacga gatcttcgac 2100atcgaagtgc cggtgggcgt catcgtcagc
cccgccaacg acttgcaggc cctggccgac 2160tacgtcgagg cggctcgcaa
acccggctcg tcacggccga ccttcgcctc ggtccacggc 2220gcctcgaatg
ggcaggtcac cgaggtgcat gccggtgacc tgtccctgga caaattcatc
2280gatgccgcaa ccctggccga agctccccgg ctgcccgccg caaacaccca
agtgcgcacc 2340gtgctgctga ccggcgccac cggcttcctc gggcgctacc
tggccctgga atggctggag 2400cggatggacc tggtcgacgg caaactgatc
tgcctggtcc gggccaagtc cgacaccgaa 2460gcacgggcgc ggctggacaa
gacgttcgac agcggcgacc ccgaactgct ggcccactac 2520cgcgcactgg
ccggcgacca cctcgaggtg ctcgccggtg acaagggcga agccgacctc
2580ggactggacc ggcagacctg gcaacgcctg gccgacacgg tcgacctgat
cgtcgacccc 2640gcggccctgg tcaaccacgt actgccatac agccagctgt
tcgggcccaa cgcgctgggc 2700accgccgagc tgctgcggct ggcgctcacc
tccaagatca agccctacag ctacacctcg 2760acaatcggtg tcgccgacca
gatcccgccg tcggcgttca ccgaggacgc cgacatccgg 2820gtcatcagcg
ccacccgcgc ggtcgacgac agctacgcca atggctactc gaacagcaag
2880tgggccggcg aggtgctgtt gcgcgaggcg catgacctgt gtggcctgcc
ggttgcggtg 2940ttccgctgcg acatgatcct ggccgacacc acatgggcgg
gacagctcaa tgtgccggac 3000atgttcaccc ggatgatcct gagcctggcg
gccaccggta tcgcgccggg ttcgttctat 3060gagcttgcgg ccgacggcgc
ccggcaacgc gcccactatg acggtctgcc cgtcgagttc 3120atcgccgagg
cgatttcgac tttgggtgcg cagagccagg atggtttcca cacgtatcac
3180gtgatgaacc cctacgacga cggcatcgga ctcgacgagt tcgtcgactg
gctcaacgag 3240tccggttgcc ccatccagcg catcgctgac tatggcgact
ggctgcagcg cttcgaaacc 3300gcactgcgcg cactgcccga tcggcagcgg
cacagctcac tgctgccgct gttgcacaac 3360tatcggcagc cggagcggcc
cgtccgcggg tcgatcgccc ctaccgatcg cttccgggca 3420gcggtgcaag
aggccaagat cggccccgac aaagacattc cgcacgtcgg cgcgccgatc
3480atcgtgaagt acgtcagcga cctgcgccta ctcggcctgc tctaa
3525101174PRTMycobacterium marinum 10Met Ser Pro Ile Thr Arg Glu
Glu Arg Leu Glu Arg Arg Ile Gln Asp1 5 10 15Leu Tyr Ala Asn Asp Pro
Gln Phe Ala Ala Ala Lys Pro Ala Thr Ala 20 25 30Ile Thr Ala Ala Ile
Glu Arg Pro Gly Leu Pro Leu Pro Gln Ile Ile 35 40 45Glu Thr Val Met
Thr Gly Tyr Ala Asp Arg Pro Ala Leu Ala Gln Arg 50 55 60Ser Val Glu
Phe Val Thr Asp Ala Gly Thr Gly His Thr Thr Leu Arg65 70 75 80Leu
Leu Pro His Phe Glu Thr Ile Ser Tyr Gly Glu Leu Trp Asp Arg 85 90
95Ile Ser Ala Leu Ala Asp Val Leu Ser Thr Glu Gln Thr Val Lys Pro
100 105 110Gly Asp Arg Val Cys Leu Leu Gly Phe Asn Ser Val Asp Tyr
Ala Thr 115 120 125Ile Asp Met Thr Leu Ala Arg Leu Gly Ala Val Ala
Val Pro Leu Gln 130 135 140Thr Ser Ala Ala Ile Thr Gln Leu Gln Pro
Ile Val Ala Glu Thr Gln145 150 155 160Pro Thr Met Ile Ala Ala Ser
Val Asp Ala Leu Ala Asp Ala Thr Glu 165 170 175Leu Ala Leu Ser Gly
Gln Thr Ala Thr Arg Val Leu Val Phe Asp His 180 185 190His Arg Gln
Val Asp Ala His Arg Ala Ala Val Glu Ser Ala Arg Glu 195 200 205Arg
Leu Ala Gly Ser Ala Val Val Glu Thr Leu Ala Glu Ala Ile Ala 210 215
220Arg Gly Asp Val Pro Arg Gly Ala Ser Ala Gly Ser Ala Pro Gly
Thr225 230 235 240Asp Val Ser Asp Asp Ser Leu Ala Leu Leu Ile Tyr
Thr Ser Gly Ser 245 250 255Thr Gly Ala Pro Lys Gly Ala Met Tyr Pro
Arg Arg Asn Val Ala Thr 260 265 270Phe Trp Arg Lys Arg Thr Trp Phe
Glu Gly Gly Tyr Glu Pro Ser Ile 275 280 285Thr Leu Asn Phe Met Pro
Met Ser His Val Met Gly Arg Gln Ile Leu 290 295 300Tyr Gly Thr Leu
Cys Asn Gly Gly Thr Ala Tyr Phe Val Ala Lys Ser305 310 315 320Asp
Leu Ser Thr Leu Phe Glu Asp Leu Ala Leu Val Arg Pro Thr Glu 325 330
335Leu Thr Phe Val Pro Arg Val Trp Asp Met Val Phe Asp Glu Phe Gln
340 345 350Ser Glu Val Asp Arg Arg Leu Val Asp Gly Ala Asp Arg Val
Ala Leu 355 360 365Glu Ala Gln Val Lys Ala Glu Ile Arg Asn Asp Val
Leu Gly Gly Arg 370 375 380Tyr Thr Ser Ala Leu Thr Gly Ser Ala Pro
Ile Ser Asp Glu Met Lys385 390 395 400Ala Trp Val Glu Glu Leu Leu
Asp Met His Leu Val Glu Gly Tyr Gly 405 410 415Ser Thr Glu Ala Gly
Met Ile Leu Ile Asp Gly Ala Ile Arg Arg Pro 420 425 430Ala Val Leu
Asp Tyr Lys Leu Val Asp Val Pro Asp Leu Gly Tyr Phe 435 440 445Leu
Thr Asp Arg Pro His Pro Arg Gly Glu Leu Leu Val Lys Thr Asp 450 455
460Ser Leu Phe Pro Gly Tyr Tyr Gln Arg Ala Glu Val Thr Ala Asp
Val465 470 475 480Phe Asp Ala Asp Gly Phe Tyr Arg Thr Gly Asp Ile
Met Ala Glu Val 485 490 495Gly Pro Glu Gln Phe Val Tyr Leu Asp Arg
Arg Asn Asn Val Leu Lys 500 505 510Leu Ser Gln Gly Glu Phe Val Thr
Val Ser Lys Leu Glu Ala Val Phe 515 520 525Gly Asp Ser Pro Leu Val
Arg Gln Ile Tyr Ile Tyr Gly Asn Ser Ala 530 535 540Arg Ala Tyr Leu
Leu Ala Val Ile Val Pro Thr Gln Glu Ala Leu Asp545 550 555 560Ala
Val Pro Val Glu Glu Leu Lys Ala Arg Leu Gly Asp Ser Leu Gln 565 570
575Glu Val Ala Lys Ala Ala Gly Leu Gln Ser Tyr Glu Ile Pro Arg Asp
580 585 590Phe Ile Ile Glu Thr Thr Pro Trp Thr Leu Glu Asn Gly Leu
Leu Thr 595 600 605Gly Ile Arg Lys Leu Ala Arg Pro Gln Leu Lys Lys
His Tyr Gly Glu 610 615 620Leu Leu Glu Gln Ile Tyr Thr Asp Leu Ala
His Gly Gln Ala Asp Glu625 630 635 640Leu Arg Ser Leu Arg Gln Ser
Gly Ala Asp Ala Pro Val Leu Val Thr 645 650 655Val Cys Arg Ala Ala
Ala Ala Leu Leu Gly Gly Ser Ala Ser Asp Val 660 665 670Gln Pro Asp
Ala His Phe Thr Asp Leu Gly Gly Asp Ser Leu Ser Ala 675 680 685Leu
Ser Phe Thr Asn Leu Leu His Glu Ile Phe Asp Ile Glu Val Pro 690 695
700Val Gly Val Ile Val Ser Pro Ala Asn Asp Leu Gln Ala Leu Ala
Asp705 710 715 720Tyr Val Glu Ala Ala Arg Lys Pro Gly Ser Ser Arg
Pro Thr Phe Ala 725 730 735Ser Val His Gly Ala Ser Asn Gly Gln Val
Thr Glu Val His Ala Gly 740 745 750Asp Leu Ser Leu Asp Lys Phe Ile
Asp Ala Ala Thr Leu Ala Glu Ala 755 760 765Pro Arg Leu Pro Ala Ala
Asn Thr Gln Val Arg Thr Val Leu Leu Thr 770 775 780Gly Ala Thr Gly
Phe Leu Gly Arg Tyr Leu Ala Leu Glu Trp Leu Glu785 790 795 800Arg
Met Asp Leu Val Asp Gly Lys Leu Ile Cys Leu Val Arg Ala Lys 805 810
815Ser Asp Thr Glu Ala Arg Ala Arg Leu Asp Lys Thr Phe Asp Ser Gly
820 825 830Asp Pro Glu Leu Leu Ala His Tyr Arg Ala Leu Ala Gly Asp
His Leu 835 840 845Glu Val Leu Ala Gly Asp Lys Gly Glu Ala Asp Leu
Gly Leu Asp Arg 850 855 860Gln Thr Trp Gln Arg Leu Ala Asp Thr Val
Asp Leu Ile Val Asp Pro865 870 875 880Ala Ala Leu Val Asn His Val
Leu Pro Tyr Ser Gln Leu Phe Gly Pro 885 890 895Asn Ala Leu Gly Thr
Ala Glu Leu Leu Arg Leu Ala Leu Thr Ser Lys 900 905 910Ile Lys Pro
Tyr Ser Tyr Thr Ser Thr Ile Gly Val Ala Asp Gln Ile 915 920 925Pro
Pro Ser Ala Phe Thr Glu Asp Ala Asp Ile Arg Val Ile Ser Ala 930 935
940Thr Arg Ala Val Asp Asp Ser Tyr Ala Asn Gly Tyr Ser Asn Ser
Lys945 950 955 960Trp Ala Gly Glu Val Leu Leu Arg Glu Ala His Asp
Leu Cys Gly Leu 965 970 975Pro Val Ala Val Phe Arg Cys Asp Met Ile
Leu Ala Asp Thr Thr Trp 980 985 990Ala Gly Gln Leu Asn Val Pro Asp
Met Phe Thr Arg Met Ile Leu Ser 995 1000 1005Leu Ala Ala Thr Gly
Ile Ala Pro Gly Ser Phe Tyr Glu Leu Ala 1010 1015 1020Ala Asp Gly
Ala Arg Gln Arg Ala His Tyr Asp Gly Leu Pro Val 1025 1030 1035Glu
Phe Ile Ala Glu Ala Ile Ser Thr Leu Gly Ala Gln Ser Gln 1040 1045
1050Asp Gly Phe His Thr Tyr His Val Met Asn Pro Tyr Asp Asp Gly
1055 1060 1065Ile Gly Leu Asp Glu Phe Val Asp Trp Leu Asn Glu Ser
Gly Cys 1070 1075 1080Pro Ile Gln Arg Ile Ala Asp Tyr Gly Asp Trp
Leu Gln Arg Phe 1085 1090 1095Glu Thr Ala Leu Arg Ala Leu Pro Asp
Arg Gln Arg His Ser Ser 1100 1105 1110Leu Leu Pro Leu Leu His Asn
Tyr Arg Gln Pro Glu Arg Pro Val 1115 1120 1125Arg Gly Ser Ile Ala
Pro Thr Asp Arg Phe Arg Ala Ala Val Gln 1130 1135 1140Glu Ala Lys
Ile Gly Pro Asp Lys Asp Ile
Pro His Val Gly Ala 1145 1150 1155Pro Ile Ile Val Lys Tyr Val Ser
Asp Leu Arg Leu Leu Gly Leu 1160 1165 1170Leu113522DNAArtificial
SequenceDescription of Artificial Sequence Synthetic carboxylic
acid reductase polynucleotide designated 891GA 11atgagcaccg
caacccatga tgaacgtctg gatcgtcgtg ttcatgaact gattgcaacc 60gatccgcagt
ttgcagcagc acagccggat cctgcaatta ccgcagcact ggaacagcct
120ggtctgcgtc tgccgcagat tattcgtacc gttctggatg gttatgcaga
tcgtccggca 180ctgggtcagc gtgttgttga atttgttacc gatgcaaaaa
ccggtcgtac cagcgcacag 240ctgctgcctc gttttgaaac cattacctat
agcgaagttg cacagcgtgt tagcgcactg 300ggtcgtgcac tgagtgatga
tgcagttcat ccgggtgatc gtgtttgtgt tctgggtttt 360aatagcgttg
attatgccac cattgatatg gcactgggtg caattggtgc agttagcgtt
420ccgctgcaga ccagcgcagc aattagcagc ctgcagccga ttgttgcaga
aaccgaaccg 480accctgattg caagcagcgt taatcagctg tcagatgcag
ttcagctgat taccggtgca 540gaacaggcac cgacccgtct ggttgttttt
gattatcatc cgcaggttga tgatcagcgt 600gaagcagttc aggatgcagc
agcacgtctg agcagcaccg gtgttgcagt tcagaccctg 660gcagaactgc
tggaacgtgg taaagatctg cctgcagttg cagaaccgcc tgcagatgaa
720gatagcctgg cactgctgat ttataccagc ggtagcacag gtgcaccgaa
aggtgcaatg 780tatccgcaga gcaatgttgg taaaatgtgg cgtcgtggta
gcaaaaattg gtttggtgaa 840agcgcagcaa gcattaccct gaatttcatg
ccgatgagcc atgttatggg tcgtagcatt 900ctgtatggca ccctgggtaa
tggtggcacc gcatattttg cagcacgtag cgatctgagc 960accctgctgg
aagatctgga actggttcgt ccgaccgaac tgaattttgt tccgcgtatt
1020tgggaaaccc tgtatggtga atttcagcgt caggttgaac gtcgtctgag
cgaagctggc 1080gatgccggtg aacgtcgtgc agttgaagca gaagttctgg
cagaacagcg tcagtatctg 1140ctgggtggtc gttttacctt tgcaatgacc
ggtagcgcac cgattagtcc ggaactgcgt 1200aattgggttg aaagcctgct
ggaaatgcat ctgatggatg gctatggtag caccgaagca 1260ggtatggttc
tgtttgatgg cgaaattcag cgtccgcctg tgattgatta taaactggtt
1320gatgttccgg atctgggtta ttttagcacc gatcgtccgc atccgcgtgg
tgaactgctg 1380ctgcgtaccg aaaatatgtt tccgggttat tataaacgtg
cagaaaccac cgcaggcgtt 1440tttgatgaag atggttatta tcgtaccggt
gatgtgtttg cagaaattgc accggatcgt 1500ctggtttatg ttgatcgtcg
taataatgtt ctgaaactgg cacagggtga atttgtgacc 1560ctggccaaac
tggaagcagt ttttggtaat agtccgctga ttcgtcagat ttatgtgtat
1620ggtaatagcg cacagccgta tctgctggca gttgttgttc cgaccgaaga
ggcactggca 1680agcggtgatc cggaaaccct gaaaccgaaa attgcagata
gcctgcagca ggttgcaaaa 1740gaagcaggtc tgcagagcta tgaagttccg
cgtgatttta ttattgaaac caccccgttt 1800agcctggaaa atggtctgct
gaccggtatt cgtaaactgg catggccgaa actgaaacag 1860cattatggtg
aacgcctgga acaaatgtat gcagatctgg cagcaggtca ggcaaatgaa
1920ctggccgaac tgcgtcgtaa tggtgcacag gcaccggttc tgcagaccgt
tagccgtgca 1980gccggtgcaa tgctgggtag cgcagccagc gatctgagtc
cggatgcaca ttttaccgat 2040ctgggtggtg atagcctgag cgcactgacc
tttggtaatc tgctgcgtga aatttttgat 2100gttgatgtgc cggttggtgt
tattgttagt ccggctaatg atctggcagc cattgcaagc 2160tatattgaag
cagaacgtca gggtagcaaa cgtccgacct ttgcaagcgt tcatggtcgt
2220gatgcaaccg ttgttcgtgc agcagatctg accctggata aatttctgga
tgcagaaacc 2280ctggcagcag caccgaatct gccgaaaccg gcaaccgaag
ttcgtaccgt gctgctgaca 2340ggtgcaaccg gttttctggg tcgttatctg
gcactggaat ggctggaacg tatggatatg 2400gttgatggta aagttattgc
actggttcgt gcccgtagtg atgaagaagc acgcgcacgt 2460ctggataaaa
cctttgatag tggtgatccg aaactgctgg cacattatca gcagctggct
2520gcagatcatc tggaagttat tgccggtgat aaaggtgaag caaatctggg
tctgggtcag 2580gatgtttggc agcgtctggc agataccgtt gatgttattg
tggatccggc agcactggtt 2640aatcatgttc tgccgtatag cgaactgttt
ggtccgaatg cactgggcac cgcagaactg 2700attcgtctgg cactgaccag
caaacagaaa ccgtatacct atgttagcac cattggtgtt 2760ggcgatcaga
ttgaaccggg taaatttgtt gaaaatgccg atattcgtca gatgagcgca
2820acccgtgcaa ttaatgatag ctatgcaaat ggctacggca atagcaaatg
ggcaggcgaa 2880gttctgctgc gcgaagcaca tgatctgtgt ggtctgccgg
ttgcagtttt tcgttgtgat 2940atgattctgg ccgataccac ctatgcaggt
cagctgaatc tgccggatat gtttacccgt 3000ctgatgctga gcctggttgc
aaccggtatt gcaccgggta gcttttatga actggatgca 3060gatggtaatc
gtcagcgtgc acattatgat ggcctgccgg ttgaatttat tgcagcagcc
3120attagcaccc tgggttcaca gattaccgat agcgataccg gttttcagac
ctatcatgtt 3180atgaacccgt atgatgatgg tgttggtctg gatgaatatg
ttgattggct ggttgatgcc 3240ggttatagca ttgaacgtat tgcagattat
agcgaatggc tgcgtcgctt tgaaacctca 3300ctgcgtgcac tgccggatcg
tcagcgccag tatagcctgc tgccgctgct gcacaattat 3360cgtacaccgg
aaaaaccgat taatggtagc attgcaccga ccgatgtttt tcgtgcagcc
3420gttcaagaag ccaaaattgg tccggataaa gatattccgc atgttagccc
tccggtgatt 3480gttaaatata ttaccgatct gcagctgctg ggtctgctgt aa
3522121173PRTArtificial SequenceDescription of Artificial Sequence
Synthetic carboxylic acid reductase polypeptide designated 891GA
12Met Ser Thr Ala Thr His Asp Glu Arg Leu Asp Arg Arg Val His Glu1
5 10 15Leu Ile Ala Thr Asp Pro Gln Phe Ala Ala Ala Gln Pro Asp Pro
Ala 20 25 30Ile Thr Ala Ala Leu Glu Gln Pro Gly Leu Arg Leu Pro Gln
Ile Ile 35 40 45Arg Thr Val Leu Asp Gly Tyr Ala Asp Arg Pro Ala Leu
Gly Gln Arg 50 55 60Val Val Glu Phe Val Thr Asp Ala Lys Thr Gly Arg
Thr Ser Ala Gln65 70 75 80Leu Leu Pro Arg Phe Glu Thr Ile Thr Tyr
Ser Glu Val Ala Gln Arg 85 90 95Val Ser Ala Leu Gly Arg Ala Leu Ser
Asp Asp Ala Val His Pro Gly 100 105 110Asp Arg Val Cys Val Leu Gly
Phe Asn Ser Val Asp Tyr Ala Thr Ile 115 120 125Asp Met Ala Leu Gly
Ala Ile Gly Ala Val Ser Val Pro Leu Gln Thr 130 135 140Ser Ala Ala
Ile Ser Ser Leu Gln Pro Ile Val Ala Glu Thr Glu Pro145 150 155
160Thr Leu Ile Ala Ser Ser Val Asn Gln Leu Ser Asp Ala Val Gln Leu
165 170 175Ile Thr Gly Ala Glu Gln Ala Pro Thr Arg Leu Val Val Phe
Asp Tyr 180 185 190His Pro Gln Val Asp Asp Gln Arg Glu Ala Val Gln
Asp Ala Ala Ala 195 200 205Arg Leu Ser Ser Thr Gly Val Ala Val Gln
Thr Leu Ala Glu Leu Leu 210 215 220Glu Arg Gly Lys Asp Leu Pro Ala
Val Ala Glu Pro Pro Ala Asp Glu225 230 235 240Asp Ser Leu Ala Leu
Leu Ile Tyr Thr Ser Gly Ser Thr Gly Ala Pro 245 250 255Lys Gly Ala
Met Tyr Pro Gln Ser Asn Val Gly Lys Met Trp Arg Arg 260 265 270Gly
Ser Lys Asn Trp Phe Gly Glu Ser Ala Ala Ser Ile Thr Leu Asn 275 280
285Phe Met Pro Met Ser His Val Met Gly Arg Ser Ile Leu Tyr Gly Thr
290 295 300Leu Gly Asn Gly Gly Thr Ala Tyr Phe Ala Ala Arg Ser Asp
Leu Ser305 310 315 320Thr Leu Leu Glu Asp Leu Glu Leu Val Arg Pro
Thr Glu Leu Asn Phe 325 330 335Val Pro Arg Ile Trp Glu Thr Leu Tyr
Gly Glu Phe Gln Arg Gln Val 340 345 350Glu Arg Arg Leu Ser Glu Ala
Gly Asp Ala Gly Glu Arg Arg Ala Val 355 360 365Glu Ala Glu Val Leu
Ala Glu Gln Arg Gln Tyr Leu Leu Gly Gly Arg 370 375 380Phe Thr Phe
Ala Met Thr Gly Ser Ala Pro Ile Ser Pro Glu Leu Arg385 390 395
400Asn Trp Val Glu Ser Leu Leu Glu Met His Leu Met Asp Gly Tyr Gly
405 410 415Ser Thr Glu Ala Gly Met Val Leu Phe Asp Gly Glu Ile Gln
Arg Pro 420 425 430Pro Val Ile Asp Tyr Lys Leu Val Asp Val Pro Asp
Leu Gly Tyr Phe 435 440 445Ser Thr Asp Arg Pro His Pro Arg Gly Glu
Leu Leu Leu Arg Thr Glu 450 455 460Asn Met Phe Pro Gly Tyr Tyr Lys
Arg Ala Glu Thr Thr Ala Gly Val465 470 475 480Phe Asp Glu Asp Gly
Tyr Tyr Arg Thr Gly Asp Val Phe Ala Glu Ile 485 490 495Ala Pro Asp
Arg Leu Val Tyr Val Asp Arg Arg Asn Asn Val Leu Lys 500 505 510Leu
Ala Gln Gly Glu Phe Val Thr Leu Ala Lys Leu Glu Ala Val Phe 515 520
525Gly Asn Ser Pro Leu Ile Arg Gln Ile Tyr Val Tyr Gly Asn Ser Ala
530 535 540Gln Pro Tyr Leu Leu Ala Val Val Val Pro Thr Glu Glu Ala
Leu Ala545 550 555 560Ser Gly Asp Pro Glu Thr Leu Lys Pro Lys Ile
Ala Asp Ser Leu Gln 565 570 575Gln Val Ala Lys Glu Ala Gly Leu Gln
Ser Tyr Glu Val Pro Arg Asp 580 585 590Phe Ile Ile Glu Thr Thr Pro
Phe Ser Leu Glu Asn Gly Leu Leu Thr 595 600 605Gly Ile Arg Lys Leu
Ala Trp Pro Lys Leu Lys Gln His Tyr Gly Glu 610 615 620Arg Leu Glu
Gln Met Tyr Ala Asp Leu Ala Ala Gly Gln Ala Asn Glu625 630 635
640Leu Ala Glu Leu Arg Arg Asn Gly Ala Gln Ala Pro Val Leu Gln Thr
645 650 655Val Ser Arg Ala Ala Gly Ala Met Leu Gly Ser Ala Ala Ser
Asp Leu 660 665 670Ser Pro Asp Ala His Phe Thr Asp Leu Gly Gly Asp
Ser Leu Ser Ala 675 680 685Leu Thr Phe Gly Asn Leu Leu Arg Glu Ile
Phe Asp Val Asp Val Pro 690 695 700Val Gly Val Ile Val Ser Pro Ala
Asn Asp Leu Ala Ala Ile Ala Ser705 710 715 720Tyr Ile Glu Ala Glu
Arg Gln Gly Ser Lys Arg Pro Thr Phe Ala Ser 725 730 735Val His Gly
Arg Asp Ala Thr Val Val Arg Ala Ala Asp Leu Thr Leu 740 745 750Asp
Lys Phe Leu Asp Ala Glu Thr Leu Ala Ala Ala Pro Asn Leu Pro 755 760
765Lys Pro Ala Thr Glu Val Arg Thr Val Leu Leu Thr Gly Ala Thr Gly
770 775 780Phe Leu Gly Arg Tyr Leu Ala Leu Glu Trp Leu Glu Arg Met
Asp Met785 790 795 800Val Asp Gly Lys Val Ile Ala Leu Val Arg Ala
Arg Ser Asp Glu Glu 805 810 815Ala Arg Ala Arg Leu Asp Lys Thr Phe
Asp Ser Gly Asp Pro Lys Leu 820 825 830Leu Ala His Tyr Gln Gln Leu
Ala Ala Asp His Leu Glu Val Ile Ala 835 840 845Gly Asp Lys Gly Glu
Ala Asn Leu Gly Leu Gly Gln Asp Val Trp Gln 850 855 860Arg Leu Ala
Asp Thr Val Asp Val Ile Val Asp Pro Ala Ala Leu Val865 870 875
880Asn His Val Leu Pro Tyr Ser Glu Leu Phe Gly Pro Asn Ala Leu Gly
885 890 895Thr Ala Glu Leu Ile Arg Leu Ala Leu Thr Ser Lys Gln Lys
Pro Tyr 900 905 910Thr Tyr Val Ser Thr Ile Gly Val Gly Asp Gln Ile
Glu Pro Gly Lys 915 920 925Phe Val Glu Asn Ala Asp Ile Arg Gln Met
Ser Ala Thr Arg Ala Ile 930 935 940Asn Asp Ser Tyr Ala Asn Gly Tyr
Gly Asn Ser Lys Trp Ala Gly Glu945 950 955 960Val Leu Leu Arg Glu
Ala His Asp Leu Cys Gly Leu Pro Val Ala Val 965 970 975Phe Arg Cys
Asp Met Ile Leu Ala Asp Thr Thr Tyr Ala Gly Gln Leu 980 985 990Asn
Leu Pro Asp Met Phe Thr Arg Leu Met Leu Ser Leu Val Ala Thr 995
1000 1005Gly Ile Ala Pro Gly Ser Phe Tyr Glu Leu Asp Ala Asp Gly
Asn 1010 1015 1020Arg Gln Arg Ala His Tyr Asp Gly Leu Pro Val Glu
Phe Ile Ala 1025 1030 1035Ala Ala Ile Ser Thr Leu Gly Ser Gln Ile
Thr Asp Ser Asp Thr 1040 1045 1050Gly Phe Gln Thr Tyr His Val Met
Asn Pro Tyr Asp Asp Gly Val 1055 1060 1065Gly Leu Asp Glu Tyr Val
Asp Trp Leu Val Asp Ala Gly Tyr Ser 1070 1075 1080Ile Glu Arg Ile
Ala Asp Tyr Ser Glu Trp Leu Arg Arg Phe Glu 1085 1090 1095Thr Ser
Leu Arg Ala Leu Pro Asp Arg Gln Arg Gln Tyr Ser Leu 1100 1105
1110Leu Pro Leu Leu His Asn Tyr Arg Thr Pro Glu Lys Pro Ile Asn
1115 1120 1125Gly Ser Ile Ala Pro Thr Asp Val Phe Arg Ala Ala Val
Gln Glu 1130 1135 1140Ala Lys Ile Gly Pro Asp Lys Asp Ile Pro His
Val Ser Pro Pro 1145 1150 1155Val Ile Val Lys Tyr Ile Thr Asp Leu
Gln Leu Leu Gly Leu Leu 1160 1165 1170
References
D00001

D00002

D00003

D00004

D00005

D00006

D00007

D00008

D00009

D00010

D00011

D00012

D00013

D00014

D00015

D00016

D00017

D00018

D00019

D00020

D00021

D00022

D00023

D00024

S00001
XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.