Use In A Paint Of A Dry-ground Calcium Carbonate With A Copolymer Of (meth)acrylic Acid With An Alkoxy Or Hydroxy Polyalkyleneglycol Group
SUAU; Jean-Marc ; et al.
U.S. patent application number 13/226879 was filed with the patent office on 2011-12-29 for use in a paint of a dry-ground calcium carbonate with a copolymer of (meth)acrylic acid with an alkoxy or hydroxy polyalkyleneglycol group. This patent application is currently assigned to COATEX S.A.S.. Invention is credited to Olivier Guerret, Jacques Mongoin, Denis Ruhlmann, Jean-Marc SUAU.
| Application Number | 20110319544 13/226879 |
| Document ID | / |
| Family ID | 38896771 |
| Filed Date | 2011-12-29 |

| United States Patent Application | 20110319544 |
| Kind Code | A1 |
| SUAU; Jean-Marc ; et al. | December 29, 2011 |
USE IN A PAINT OF A DRY-GROUND CALCIUM CARBONATE WITH A COPOLYMER OF (METH)ACRYLIC ACID WITH AN ALKOXY OR HYDROXY POLYALKYLENEGLYCOL GROUP
Abstract
A paint comprising a calcium carbonate that is obtained by dry-grinding in the presence of a grinding aid agent comprising a copolymer of (meth)acrylic acid with a monomer containing an alkoxy or hydroxy polyalkyleneglycol group.
| Inventors: | SUAU; Jean-Marc; (Lucenay, FR) ; Mongoin; Jacques; (Quincieux, FR) ; Ruhlmann; Denis; (Genay, FR) ; Guerret; Olivier; (La Tour De Salvagny, FR) |
| Assignee: | COATEX S.A.S. Genay FR |
| Family ID: | 38896771 |
| Appl. No.: | 13/226879 |
| Filed: | September 7, 2011 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 12523207 | Jul 15, 2009 | |||
| PCT/IB08/00532 | Feb 26, 2008 | |||
| 13226879 | ||||
| Current U.S. Class: | 524/425 |
| Current CPC Class: | C09D 7/61 20180101; C01P 2006/22 20130101; C09C 3/10 20130101; C09C 1/021 20130101; C01P 2006/64 20130101; C01P 2006/60 20130101; C01P 2006/62 20130101; C08K 3/26 20130101 |
| Class at Publication: | 524/425 |
| International Class: | C09D 133/02 20060101 C09D133/02; C09D 129/10 20060101 C09D129/10; C09D 135/02 20060101 C09D135/02; C09D 7/14 20060101 C09D007/14; C09D 143/02 20060101 C09D143/02 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Mar 5, 2007 | FR | 07 01595 |
Claims
1-7. (canceled)
8. A paint, comprising a calcium carbonate which is dry-ground in the presence of a grinding aid agent comprising a copolymer comprising: a) at least one anionic monomer selected from the group consisting of acrylic acid and methacrylic acid; b) at least one non-ionic monomer of formula (I) ##STR00003## wherein m, n, p, and q are integers such that m, n, and p.ltoreq.150, q.gtoreq.1, and 5.ltoreq.(m+n+p)q.ltoreq.150, R.sub.1 and R.sub.2 independently represent hydrogen or a methyl or ethyl radical, R represents a radical containing an unsaturated polymerizable function selected from the group consisting of a vinylic, an acrylic ester, a methacrylic ester, a maleic ester, an acrylurethane, a methacrylurethane, an .alpha.-.alpha.' dimethyl-isopropenyl-benzylurethane, an allylurethane, an optionally substituted allylic ether, an optionally substituted vinylic ether, an ethylenically unsaturated amide, an ethylenically unsaturated imide, an acrylamide, and a methacrylamide; R' represents hydrogen or a hydrocarbon radical with 1 to 40 carbon atoms; and c) optionally, at least one other monomer selected from (meth)acrylic anhydride, a (meth)acrylamide, an acrylate with 1 to 20 carbon atoms in its ester radical, a methacrylate with 1 to 20 carbon atoms in its ester radical, a hydroxylated methacrylate, an aromatic vinylic monomer, an acrylate phosphate of ethylene glycol, an acrylate phosphate of oxyethylene glycol, an acrylate phosphate of oxypropylene glycol, an acrylate phosphate of oxyethylene and oxypropylene glycol, a methacrylate phosphate of ethylene glycol, a methacrylate phosphate of oxyethylene glycol, a methacrylate phosphate of oxypropylene glycol, and a methacrylate phosphate of oxyethylene and oxypropylene glycol.
9. The paint of claim 8, wherein said copolymer comprises, expressed as a percentage by weight of the monomers in which the sum of the percentages by weight of all the monomers equals 100%: a) 0.5% to 50% of the at least one anionic monomer; b) 50% to 99.5% of the at least one non-ionic monomer of formula (I); and c) 0% to 50% of the at least one other monomer.
10. The paint of claim 8, wherein said copolymer is obtained in acidic form and optionally distilled, and is, optionally, partially or totally neutralized by at least one neutralization agent having a monovalent or polyvalent cation, wherein the neutralization agent is at least one selected from the group consisting of ammonia, calcium hydroxide, calcium oxide, magnesium hydroxide, magnesium oxide, sodium hydroxide, potassium hydroxide, lithium hydroxide, an aliphatic primary amine, an aliphatic secondary amine, an aliphatic tertiary amine, a cyclic secondary amine, and a cyclic tertiary amine.
11. The paint of claim 8, wherein said copolymer is obtained by radical polymerization in solution, in a direct or reverse emulsion, in suspension or precipitation in a solvent, in the presence of at least one selected from the group consisting of a catalytic system and a chain transfer agent, controlled radical polymerization by nitroxide mediated polymerization (NMP), or by a cobaloxime, atom transfer radical polymerization (ATRP), or controlled radical polymerization by at least one sulphurated derivative selected from the group consisting of a carbamate, a dithioester, a trithiocarbonate, RAFT, and a xanthate.
12. The paint of claim 10, wherein said copolymer is totally or partially neutralized and, before or after the total or partial neutralization reaction, is treated and separated into several phases, according to static or dynamic processes, by at least one polar solvent selected from the group consisting of water, methanol, ethanol, propanol, isopropanol, a butanol, acetone, and tetrahydrofuran.
13. The paint of claim 8, wherein 0.05% to 5% by dry weight of the copolymer relative to a dry weight of calcium carbonate is used during a dry grinding of said calcium carbonate.
14. The paint of claim 8, wherein, in the copolymer, m, n, p, and q are integers such that: 15.ltoreq.(m+n+p)q.ltoreq.120.
15. The paint of claim 8, wherein, in the copolymer, R' is a hydrocarbon radical with 1 to 4 carbon atoms
16. The paint of claim 8, wherein, in the copolymer, R' is a methyl radical
17. The paint of claim 8, wherein, in the copolymer, the at least one other monomer is present and is methyl acrylate, ethyl acrylate, isopropyl acrylate, n-propyl acrylate, isobutyl acrylate, n-butyl acrylate, tert-butyl acrylate, 2-ethylhexyl acrylate, methyl methacrylate, or ethyl methacrylate.
18. The paint of claim 8, wherein, in the copolymer, the at least one other monomer is present and is hydroxyethyl methacrylate, hydroxypropyl methacrylate, styrene, .alpha.-methylstyrene, styrene sulfonate, or acrylamido-2-methyl-2-propane-sulfonic acid.
19. The paint of claim 8, wherein, in the copolymer, the at least one other monomer is present and is an acrylate phosphate of ethylene glycol, a methacrylate phosphate of ethylene glycol, an acrylate phosphate of oxyethylene glycol, an acrylate phosphate of oxypropylene glycol, an acrylate phosphate of oxyethylene and oxypropylene glycol, a methacrylate phosphate of oxyethylene glycol, a methacrylate phosphate of oxypropylene glycol, or a methacrylate phosphate of oxyethylene and oxypropylene glycol.
20. The paint of claim 8, wherein, in the copolymer, the at least one anionic monomer is present in 1% to 25% by weight, based on a sum of percentages by weight of all monomers being 100%.
21. The paint of claim 8, wherein, in the copolymer, the at least one anionic monomer is present in 5% to 20% by weight, based on a sum of percentages by weight of all monomers being 100%.
22. The paint of claim 8, wherein, in the copolymer, the at least one non-ionic monomer of formula (I) is present in 75% to 99% by weight, based on a sum of percentages by weight of all monomers being 100%.
23. The paint of claim 8, wherein, in the copolymer, the at least one non-ionic monomer of formula (I) is present in 80% to 95% by weight, based on a sum of percentages by weight of all monomers being 100%.
24. The paint of claim 8, wherein 0.1% to 3% by dry weight of the copolymer relative to a dry weight of the calcium carbonate is used in dry grinding.
25. The paint of claim 8, wherein 0.1% to 1% by dry weight of the copolymer relative to a dry weight of the calcium carbonate is used in dry grinding.
26. The paint of claim 8, wherein 1% to 5% by dry weight of the copolymer relative to a dry weight of the calcium carbonate is used in dry grinding.
27. The paint of claim 8, wherein the copolymer is partially or totally neutralized and the neutralization agent is triethanolamine or sodium hydroxide.
Description
[0001] The present invention concerns the field of paints, where the said paints contain at least one mineral matter which is a calcium carbonate obtained by dry grinding.
[0002] Grinding using a dry process (as opposed to in an aqueous medium) of calcium carbonate is described in the documents "Beitrag zur Aufklarung der Wirkungsweise von Mahlhilfsmitteln" ("Freiberger Forschungshefte" VEB Deutscher Verlag fur Grundstoffindustrie, Leipzig, Germany (1975)). and "Calcium Carbonate" (Birkhauser Verlag, 2001).
[0003] This grinding, which leads to a reduction in the sizes of the individual particles of calcium carbonate by mechanical action, is assisted by grinding agents which can be classified into the following 3 categories.
[0004] The first comprises the weak acids (formic, acetic, lactic, adipic as mentioned in document FR 2 863 914, or stearic as indicated in document EP 0 510 890), and their salts (sodium lignin sulfonate, sodium acetate, etc.). The second includes the weak bases, and notably the amines (see document GB 2 179 268). The third, and the most commonly used, since it is the most effective, includes the glycol-based alcohols, including notably diethylene glycol illustrated in documents WO 2002/081 573 and U.S. 2003/019 399.
[0005] Although they are still the most effective, the latter lead to calcium carbonates with a high quantity of volatile organic compounds (VOCs). For this reason, finding an alternative solution to these glycol-based grinding aid agents which is at least as effective as them in terms of the final application would constitute a major contribution to the state of the technique.
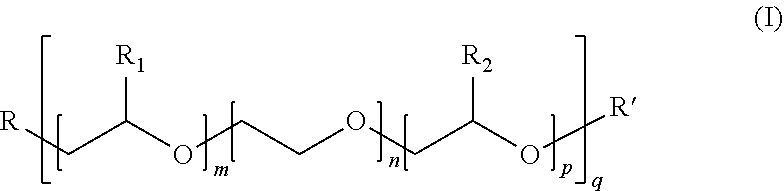
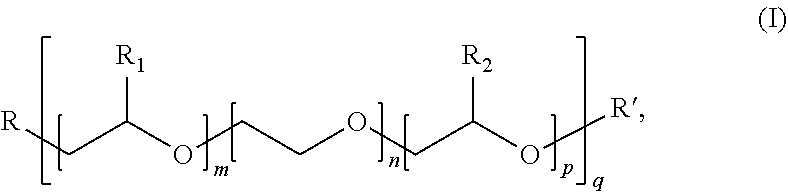
[0006] With this regard, a first object of the invention consists in the use, in a paint, of a calcium carbonate characterised in that the said carbonate is dry-ground in the presence of a grinding aid agent, firstly, and in that the grinding aid agent is, secondly, a copolymer, consisting: [0007] a) of at least one anionic monomer chosen from among acrylic acid, methacrylic acid and their blends, [0008] b) and of at least one non-ionic monomer, of formula (I):
##STR00001##
[0009] where: [0010] m, n, p and q are integers such that: m, n, p.ltoreq.150, q.gtoreq.1, and 5.ltoreq.(m+n+p)q.ltoreq.150, and preferentially 15.ltoreq.(m+n+p)q.ltoreq.120, [0011] R.sub.1 represents hydrogen or the methyl or ethyl radical, [0012] R.sub.2 represents hydrogen or the methyl or ethyl radical, [0013] R represents a radical containing an unsaturated polymerisable function, preferentially belonging to the group of vinylics, or to the group of acrylic, methacrylic, maleic esters, or to the group of unsaturated urethanes such as acrylurethane, methacrylurethane, .alpha.-.alpha.' dimethyl-isopropenyl-benzylurethane, allylurethane, or to the group of allylic or vinylic ethers, whether or not substituted, or again to the group of ethylenically unsaturated amides or imides, or again to the group constituted by acrylamide and methacrylamide, [0014] R' represents hydrogen or a hydrocarbonated radical with 1 to 40 carbon atoms, preferentially 1 to 4 carbon atoms, where R' is very preferentially the methyl radical,
[0015] or a blend of several monomers of formula (I), [0016] c) and possibly of at least one other monomer chosen from among (meth)acrylic anhydride, (meth)acrylamide, or from among the (meth)acrylic esters, such as preferentially the acrylates and methacrylates with 1 to 20 carbon atoms in their ester radical, such as very preferentially the methyl, ethyl, isopropyl, n-propyl, isobutyl, n-butyl, ter-butyl or 2-ethylhexyl acrylates, the methyl or ethyl methacrylates, the hydroxylated methacrylates such as the hydroxyethyl and hydroxypropyl methacrylates, or from among the aromatic vinylic monomers such as preferentially styrene, .alpha.-methylstyrene, styrene sulfonate, or acrylamido-2-methyl-2-propane-sulfonic acid, or from among the organophosphate monomers, such as preferentially the acrylate and methacrylate phosphates of ethylene glycol, or the acrylate and methacrylate phosphates of oxyethylene and/or oxypropylene glycol, and their blends.
[0017] This grinding aid agent may be designated using the expression of a copolymer of methacrylic acid with a monomer containing an alcoxy or hydroxy polyalkyleneglycol group.
[0018] The examples of the present Application show that, in comparison with a calcium carbonate which is dry ground with a diethylene glycol, a calcium carbonate according to the invention enables a cement to be obtained with a much higher degree of spreading.
[0019] This use is also characterised in that the said copolymer consists, expressed as a percentage by weight of the monomers (the sum of the percentages by weight of all the monomers equals 100%): [0020] a) 0.5% to 50%, preferentially 1% to 25%, and very preferentially 5% to 20%, of at least one anionic monomer chosen from among acrylic acid, methacrylic acid and their blends, [0021] b) of 50% to 99.5%, preferentially 75% to 99%, and very preferentially 80% to 95%, of at least one non-ionic monomer, of formula (I):
##STR00002##
[0022] where: [0023] m, n, p and q are integers such that: m, n, p.ltoreq.150, q.gtoreq.1, and 5.ltoreq.(m+n+p)q.ltoreq.150, and preferentially 15.ltoreq.(m+n+p)q.ltoreq.120, [0024] R.sub.1 represents hydrogen or the methyl or ethyl radical, [0025] R.sub.2 represents hydrogen or the methyl or ethyl radical, [0026] R represents a radical containing an unsaturated polymerisable function, preferentially belonging to the group of vinylics, or to the group of acrylic, methacrylic, maleic esters, or to the group of unsaturated urethanes such as acrylurethane, methacrylurethane, .alpha.-.alpha.' dimethyl-isopropenyl-benzylurethane, allylurethane, or to the group of allylic or vinylic ethers, whether or not substituted, or again to the group of ethylenically unsaturated amides or imides, or again to the group constituted by acrylamide and methacrylamide, [0027] R' represents hydrogen or a hydrocarbonated radical with 1 to 40 carbon atoms, preferentially 1 to 4 carbon atoms, where R' is very preferentially the methyl radical,
[0028] or a blend of several monomers of formula (I), [0029] c) 0% to 50% of at least one other monomer chosen from among (meth)acrylic anhydride, (meth)acrylamide, or from among the (meth)acrylic esters, such as preferentially the acrylates and methacrylates with 1 to 20 carbon atoms in their ester radical, such as very preferentially the methyl, ethyl, isopropyl, n-propyl, isobutyl, n-butyl, ter-butyl or 2-ethylhexyl acrylates, the methyl or ethyl methacrylates, the hydroxylated methacrylates such as the hydroxyethyl and hydroxypropyl methacrylates, or from among the aromatic vinylic monomers such as preferentially styrene, .alpha.-methylstyrene, styrene sulfonate, or acrylamido-2-methyl-2-propane-sulfonic acid, or from among the organophosphate monomers, such as preferentially the acrylate and methacrylate phosphates of ethylene glycol, or the acrylate and methacrylate phosphates of oxyethylene and/or oxypropylene glycol, and their blends.
[0030] This use is also characterised in that the said copolymer is obtained in the acidic form and possibly distilled, and is possibly partially or totally neutralised by one or more neutralisation agents having a monovalent or polyvalent cation, where the said agents are chosen preferentially from among ammonia or from among calcium, magnesium hydroxides and/or oxides, or from among sodium, potassium or lithium hydroxides, or from among the aliphatic and/or cyclic primary, secondary or tertiary amines, such as preferentially stearylamine, the ethanolamines (mono-, di- and triethanolamine), mono- and diethylamine, cyclohexylamine, methylcyclohexylamine, amino methyl propanol, morpholine, and preferentially in that the neutralisation agent is chosen from among triethanolamine and sodium hydroxide.
[0031] This use is also characterised in that the said copolymer is obtained by processes of radical polymerisation in solution, in a direct or reverse emulsion, in suspension or precipitation in solvents, in the presence of catalytic systems and chain transfer agents, or again by processes of controlled radical polymerisation, and preferentially by nitroxide mediated polymerisation (NMP) or by cobaloximes, by atom transfer radical polymerisation (ATRP), by controlled radical polymerisation by sulphurated derivatives, chosen from among carbamates, dithioesters or trithiocarbonates (RAFT) or xanthates.
[0032] This use is also characterised in that the said copolymer may possibly, before or after the total or partial neutralisation reaction, be treated and separated into several phases, according to static or dynamic processes known to the skilled man in the art, by one or more polar solvents belonging preferentially to the group constituted by water, methanol, ethanol, propanol, isopropanol, the butanols, acetone and tetrahydrofuran or their blends.
[0033] One of the phases then corresponds to the polymer used according to the invention.
[0034] This use is also characterised in that 0.05% to 5%, preferentially 0.1% to 3%, and very preferentially 0.1% to 1%, by dry weight of the said copolymer relative to the dry weight of calcium carbonate is used during the dry grinding of the said carbonate.
[0035] A second purpose of the invention consists of the paints obtained by the use of dry-ground calcium carbonate according to the invention.
EXAMPLES
[0036] In these examples, all the grinding operations are undertaken according to a method well known to the skilled man in the art (document FR 2 901 491 describes such a method). We shall confine ourselves here to indicating the final granulometry of the particles of calcium carbonate obtained after grinding (the skilled man in the art in reality knows how to modify the parameters of the process with a view to obtaining such a granulometry; however, one may also refer to the above-mentioned document).
[0037] In each of the tests n.degree. 1 to 4, a matt paint is produced in aqueous phase by blending whilst stirring of the different constituents, in the proportions in grams as indicated in table 1.
TABLE-US-00001 Test n.sup.o 1 2 3 4 Prior Art (PA)/Invention (IN) PA IN PA IN Water 292.0 292.0 292.0 292.0 Ammonium hydroxide (31% 2.0 2.0 2.0 2.0 solution) Coatex .TM. P90 (40% solution) 4.0 4.0 4.0 4.0 Mergal .TM. K6N 2.0 2.0 2.0 2.0 Byk .TM. 034 2.0 2.0 2.0 2.0 TiO2 RL68 41.0 41.0 41.0 41.0 Omyacoat .TM. 850 OG 215.0 215.0 215.0 215.0 CaCO3 dry ground according to 328.0 0 0 0 test n.sup.o1 CaCO3 dry ground according to 0 328.0 0 0 test n.sup.o2 CaCO3 dry ground according to 0 0 328.0 0 test n.sup.o3 CaCO3 dry ground according to 0 0 0 328.0 test n.sup.o4 Acronal .TM. 290D 82.0 82.0 82.0 82.0 Monoethylene glycol 10.0 10.0 10.0 10.0 Texanol .TM. 10.0 10.0 10.0 10.0 Rheo .TM. 3000 12.0 12.0 12.0 12.0 Total (g) 1000.0 1000.0 1000.0 1000.0 Coatex .TM. P90 designates a thickener sold by the company COATEX .TM., Rheo .TM. 3000 designates a thickener sold by the company COATEX .TM., Mergal .TM. K6N designates a bactericide sold by the company TROY .TM., Byk .TM. 034 designates an anti-foaming agent sold by the company BYK .TM., TiO2 RL68 designates a titanium dioxide powder sold by the company MILLENIUM .TM., Omyacoat .TM. 850 OG designates a calcium carbonate sold by the company OMYA .TM., Acronal .TM. 290 D designates a binder sold by the company BASF .TM., Texanol .TM. designates a coalescence agent sold by the company EASTMANN .TM.
Test No. 1
[0038] This test illustrates the prior art and uses a calcium carbonate obtained by dry grinding in the presence of 3000 ppm (relative to the dry weight of calcium carbonate) of a grinding agent of the prior art, which is diethylene glycol.
[0039] After grinding a calcium carbonate is obtained, 73.8% and 48.5% by weight of the particles of which have an average diameter lower respectively than 2 .mu.m and 1 .mu.m (as measured using a Sedigraph.TM. 5100).
Test No. 2
[0040] This test illustrates the prior art and uses a calcium carbonate obtained by dry grinding in the presence of 3000 ppm (relative to the dry weight of calcium carbonate) of a grinding agent of the invention which is a polymer consisting, by weight percentage, 81.5% of methoxy polyethylene glycol methacrylate of molecular weight 2000, 4.9% of methacrylic acid, and 13.6% of acrylic acid.
[0041] After grinding a calcium carbonate is obtained, 74.5% and 44.1% by weight of the particles of which have an average diameter lower respectively than 2 .mu.m and 1 .mu.m (as measured using a Sedigraph.TM. 5100).
Test No. 3
[0042] This test illustrates the prior art and uses a calcium carbonate obtained by dry grinding in the presence of 1400 ppm (relative to the dry weight of calcium carbonate) of a grinding agent of the prior art, which is diethylene glycol.
[0043] After grinding a calcium carbonate is obtained, 47.1% and 30.0% by weight of the particles of which have an average diameter lower respectively than 2 .mu.m and 1 .mu.m (as measured using a Sedigraph.TM. 5100).
Test No. 4
[0044] This test illustrates the prior art and uses a calcium carbonate obtained by dry grinding in the presence of 1400 ppm (relative to the dry weight of calcium carbonate) of a grinding agent of the invention which is a polymer consisting, by weight percentage, 81.5% of methoxy polyethylene glycol methacrylate of molecular weight 2000, 4.9% of methacrylic acid, and 13.6% of acrylic acid.
[0045] After grinding a calcium carbonate is obtained, 45.4% and 28.0% by weight of the particles of which have an average diameter lower respectively than 2 .mu.m and 1 .mu.m (as measured using a Sedigraph.TM. 5100).
[0046] The polymer used in tests n.degree. 2 and 4 is obtained by polymerisation techniques well known to the skilled man in the art.
[0047] In a first beaker the following are introduced at ambient temperature and whilst stirring: 259.02 of methoxy polyethylene glycol methacrylate of molecular weight equal to 2000 g/mol, a solution containing 15.36 g of methacrylic acid and 274.39 g of water, a solution containing 69.86 g of untreated water and 50.25 g of acrylic acid
[0048] In a second beaker the following are introduced at ambient temperature and whilst stirring: 2.38 g of thiolactic acid (of 98.5% mass purity) and 52.14 g of water.
[0049] In a third beaker the following are introduced at ambient temperature and whilst stirring: 2.84 g of ammonium persulphate (NH.sub.4).sub.2S.sub.2O.sub.8 and 51.90 g of water.
[0050] In a fourth beaker the following are introduced at ambient temperature and whilst stirring: 0.56 g of ammonium persulphate (NH.sub.4).sub.2S.sub.2O.sub.8 and 10.87 g of water.
[0051] In a single-casing cylindrical reactor, surmounted by a refrigerating column, by an anchor stirring system and 3 inlets, 351.39 g of water and 295.61 g of isopropanol are introduced. The reactive medium is heated whilst stirring (230 rpm) to 84.+-.2.degree. C. The contents of the first 3 beakers are introduced into the reactive medium in 2 hours. The introduction nozzles are then rinsed with 20.84 g of water (this water goes directly into the reactive medium). The solution of the 4.sup.th beaker is then added to the reactive medium, and the blend is fired for 1 hour at 84.+-.2.degree. C. (at 230 rpm). At the end of the reaction the device is rinsed with 52.13 g of water. The temperature is then raised and 604.78 g is distilled. The temperature is brought down to between 50 and 60.degree. C., and 69.34 g of sodium hydroxide at 50% mass in water is added to the reactive medium. Finally the medium is diluted with 20.85 g of water. It is cooled again before obtaining the polymer of the invention.
[0052] For each of the tests n.degree. 1 to 4, the Stormer.TM., ICI.TM. and Brookfield.TM. viscosities were determined according to the methods well known to the skilled man in the art.
[0053] Certain optical characteristics of the paints obtained were also measured, such as notably L whiteness, the "3 Hunterlab filters" Sub-tone, covering power or contrast ratio, and the level of brightness at angles equal to 60.degree. and 85.degree..
[0054] The methods used are notably described in document FR 2 872 815.
[0055] The results are shown in tables 2 and 3.
TABLE-US-00002 TABLE 2 Test Test Test Test Viscosities n.sup.o 1 n.sup.o 2 n.sup.o 3 n.sup.o 4 Time t = 0 .mu..sup.I (P) 1.9 1.9 1.6 1.6 .mu..sup.S (K.U) 102 106 99 100 .mu..sup.B.sub.10 (mPa s) 13700 15100 11600 11100 .mu..sup.B100 (mPa s) 3300 3600 2900 3000 Time t = 24 hours .mu..sup.B.sub.10 (mPa s) 18900 18400 12700 12300 .mu..sup.B100 (mPa s) 4900 4750 3500 3100 Instant t = 7 days .mu..sup.B.sub.10 (mPa s) 43900 32100 15400 12800 50.degree. C. .mu..sup.B.sub.10 (mPa s) 14000 8340 4500 3200 Instant t = .mu..sup.B.sub.10 (mPa s) Taken 42800 15800 13800 1 month 50.degree. C. .mu..sup.B.sub.10 (mPa s) in 12000 4700 3400 mass .mu..sup.I (P): ICI .TM. viscosity .mu..sup.S (K.U): Stormer .TM. viscosity .mu..sup.B.sub.10 (mPa s): Brookfield .TM. viscosity determined at 10 revolutions per minute .mu..sup.B.sub.100 (mPa s): Brookfield .TM. viscosity determined at 100 revolutions per minute
TABLE-US-00003 TABLE 3 Test n.sup.o 1 2 3 4 Whiteness. 99.2 99.1 98.7 98.6 Measurement of the "L" "3 Hunterlab filters" Sub-tone 2.2 2.3 2.3 2.2 Value of b Covering power of dry film 96.9% 96.8% 96.0% 95.9% Contrast ratio RC = Yn/Yb Level of brightness t = 72 h 60.degree. 4.7 4.6 4.8 4.7 85.degree. 32.9 33.5 14.9 15.6
[0056] By comparing the tests 2 by 2 (1 to 2, and 3 to 4), it is observed that the optical properties of the paint films are in the context of the invention at the same level as for the prior art.
[0057] Conversely, a clear drift of the Brookfield.TM. viscosity is observed in the case of paints formulated according to the prior art. At the same time, this viscosity remains much more stable in the context of the invention.
[0058] We have therefore succeeded in developing a dry-ground calcium carbonate which, in the paint field, has a more stable Brookfield viscosity, without modifying the optical properties of the dry film.
* * * * *
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.