Gastroresistant Pharmaceutical Formulations Containing Rifaximin
Viscomi; Giuseppe Claudio ; et al.
U.S. patent application number 12/695945 was filed with the patent office on 2010-12-30 for gastroresistant pharmaceutical formulations containing rifaximin. This patent application is currently assigned to ALFA WASSERMANN S.P.A.. Invention is credited to Ernesto Palazzini, Maria Rosaria Pantaleo, Giuseppe Claudio Viscomi, Villiam Zamboni.
| Application Number | 20100330129 12/695945 |
| Document ID | / |
| Family ID | 36694343 |
| Filed Date | 2010-12-30 |



| United States Patent Application | 20100330129 |
| Kind Code | A1 |
| Viscomi; Giuseppe Claudio ; et al. | December 30, 2010 |
Gastroresistant Pharmaceutical Formulations Containing Rifaximin
Abstract
The object of the invention consists of pharmaceutical formulations containing rifaximin in the shape of microgranules made gastroresistant by an insoluble polymer at pH values between 1.5 and 4.0 and soluble at pH values between 5.0 and 7.5, by their preparation and by their use in the manufacture of medicinal preparations useful in the treatment of inflammatory bowel diseases (IBD) and mainly Crohn's disease.
| Inventors: | Viscomi; Giuseppe Claudio; (Bologna, IT) ; Palazzini; Ernesto; (Bologna, IT) ; Zamboni; Villiam; (Bologna, IT) ; Pantaleo; Maria Rosaria; (Bologna, IT) |
| Correspondence Address: |
TechLaw LLP
10755 Scripps Poway Parkway, Suite 465
San Diego
CA
92131
US
|
| Assignee: | ALFA WASSERMANN S.P.A. Allano IT |
| Family ID: | 36694343 |
| Appl. No.: | 12/695945 |
| Filed: | January 28, 2010 |
Related U.S. Patent Documents
| Application Number | Filing Date | Patent Number | ||
|---|---|---|---|---|
| 11814628 | Jul 24, 2007 | |||
| PCT/EP2006/002022 | Mar 6, 2006 | |||
| 12695945 | ||||
| Current U.S. Class: | 424/400 ; 514/279 |
| Current CPC Class: | A61K 9/5026 20130101; A61K 31/437 20130101; A61K 31/44 20130101; A61K 9/2027 20130101; A61P 1/14 20180101; A61P 1/04 20180101; Y02A 50/30 20180101; A61K 9/28 20130101; Y02A 50/473 20180101; A61P 1/12 20180101; A61P 31/04 20180101; A61K 9/1635 20130101; A61K 9/5073 20130101; A61K 9/1652 20130101; A61K 9/2054 20130101; A61P 29/00 20180101; A61K 9/0095 20130101; A61K 31/395 20130101; A61P 1/00 20180101 |
| Class at Publication: | 424/400 ; 514/279 |
| International Class: | A61K 9/00 20060101 A61K009/00; A61K 31/437 20060101 A61K031/437; A61P 1/12 20060101 A61P001/12; A61P 29/00 20060101 A61P029/00; A61P 1/14 20060101 A61P001/14; A61P 31/04 20060101 A61P031/04 |
Foreign Application Data
| Date | Code | Application Number |
|---|---|---|
| Mar 7, 2005 | IT | BO 2005 A 000123 |
Claims
1. A pharmaceutical composition comprising a therapeutically effective amount of rifaximin or polymorph thereof, one or more release controlling agents and at least one pharmaceutically acceptable excipient.
2. The pharmaceutical composition according to claim 1, wherein the composition is formulated to delay release of rifaximin in the gastrointestinal tract.
3. The pharmaceutical composition according to claim 2, wherein the composition is formulated to delay release of rifaximin in the intestinal tract.
4. The pharmaceutical composition according to claim 2, wherein the composition is formulated to give binding properties in the gastrointestinal tract.
5. The pharmaceutical composition according to claim 4, wherein the binding properties are achieved with one or more binder compounds selected from the group consisting of cellulose, cellulose derivatives, starches, gums, synthetic gum, polyvinylpyrrolidone, sodium carboxymethyl cellulose, polyethylene glycol, gelatin, polypropylene glycol, alginic acid, alginate salts, sugars, and combinations thereof.
6. The pharmaceutical composition according to claim 5, wherein said sugars are selected from the group consisting of sucrose, glucose, dextrose, and lactose.
7. The pharmaceutical composition according to claim 2, wherein delay in release of rifaximin is achieved with excipients selected from the group consisting of proteins carbohydrates, polysaccharides, cellulose derivatives, acetylated glycerides, diethylphthalate, propylene glycol, polyethylene glycol, polysorbate, polyoxyethylenate esthers, poly vinylacetate phthalate, polyvinyl alcohol, acrylic polymer, acrylic methacrylic acid copolymers with an acrylic or methacrylic ester, polyvinyl alcohols, polyvinylpyrrolidone, polymers of acrylic and methacrylic esters, blends and copolymers or mixtures thereof, polyols, dextrans, sodium alginate, gums, xanthan gum, guar gum, and synthetic gum.
8. The pharmaceutical composition according to claim 7, wherein said proteins are selected from the group consisting of pectin, zein, and gelatin.
9. The pharmaceutical composition according to claim 7, wherein said carbohydrates are selected from the group consisting of starch and chitosan.
10. The pharmaceutical composition according to claim 7, wherein said polysaccharide is cellulose.
11. The pharmaceutical composition according to claim 7, wherein said cellulose derivatives are selected from the group consisting of methyl cellulose, ethyl cellulose, hydroxypropyl cellulose, hydroxypropylmethyl cellulose, hydroxypropyl cellulose acetate phthalate, cellulose acetate phthalate, cellulose acetate, cellulose acetate propionate, cellulose acetate butyrate, and sodium carboxymethyl cellulose.
12. The pharmaceutical composition according to claim 1, wherein the release controlling agent is a hydrophilic polymer or hydrophobic polymer or combination thereof.
13. The pharmaceutical composition according to claim 1, wherein the release controlling agent is a hydrophilic polymer.
14. The pharmaceutical composition according to claim 13, wherein the hydrophilic polymer is selected from the group consisting of carbohydrate, cellulose derivatives, gum, alginates, polyvinylacetate, polyvinylalcohol, povidone, polyvinylpyrrolidone, acrylic and methacrylic acid copolymers and mixtures thereof.
15. The pharmaceutical composition according to claim 14, wherein the carbohydrate is cellulose.
16. The pharmaceutical composition according to claim 14, wherein the cellulose derivative is ethylcellulose, hydroxypropylcellulose, or hydroxypropylmethylcellulose.
17. The pharmaceutical composition according to claim 14, wherein the gum is xanthan gum.
18. The pharmaceutical composition according to claim 12, wherein the hydrophobic polymer is selected from the group consisting of waxes, fatty acids and their esters, glyceryl ester, hydrogenated oil, castor oil, mineral oil, paraffin, stearate salts, stearic acid, talc, and mixture thereof.
19. The pharmaceutical composition according to claim 18, wherein the glyceryl ester is glyceryl monostearate or glyceryl distearate.
20. The pharmaceutical composition according to claim 18, wherein the stearate salt is magnesium stearate or calcium stearate.
21. The pharmaceutical composition according to claim 1, further comprising one or more solubilizing agents.
22. The pharmaceutical composition according to claim 21, wherein the solubilizing agent is selected from the group consisting of surfactants, polysorbates, polyoxyethylene esters, dextrins, waxes, monohydric alcohol esters, trialkyl citrates, lower alcohol fatty acid esters, glycerol acetates, acetin, diacetin, triacetin, glycerol fatty acid esters, monoglycerides, diglycerides, triglycerides, acetylated mono and diglycerides, propylene glycol esters, ethylene glycol esters, and combinations thereof.
23. The pharmaceutical composition according to claim 1, wherein the at least one pharmaceutically acceptable excipient is selected from the group consisting of binders, diluents, lubricants, surfactants, plasticizers, and glidants.
24. The pharmaceutical composition according to claim 23, wherein the diluent is selected from the group consisting of cellulose or cellulose derivatives, polyhydric alcohol, sugar alcohols, sulphate or phosphate salts of inorganic metals, and mixtures thereof.
25. The pharmaceutical composition according to claim 23, wherein the lubricant is selected from the group consisting of magnesium or calcium stearate, polyethylene glycol, stearic acid, hydrogenated vegetable oil, glyceryl stearate, cornstarch, talc, magnesium silicate, colloidal silicon dioxide, silicon hydrogel, and mixtures thereof.
26. The pharmaceutical composition according to claim 23, wherein the plasticizer is selected from the group consisting of acetylated monoglycerides, butyl phthalyl butyl glycolate, dibutyl tartrate, diethyl phthalate, dimethyl phthalate, ethyl phthalyl ethyl glycolate, glycerin, ethylene glycol, propylene glycol, triacetin citrate, triacetin, tripropinoin, diacetin, dibutyl phthalate, acetyl monoglyceride, polyethylene glycols, castor oil, triethyl citrate, polyhydric alcohols, acetate esters, gylcerol triacetate, acetyl triethyl citrate, dibenzyl phthalate, dihexyl phthalate, butyl octyl phthalate, diisononyl phthalate, butyl octyl phthalate, dioctyl azelate, epoxydised tallate, triisoctyl trimellitate, diethylhexyl phthalate, di-n-octyl phthalate, di-1-octyl phthalate, di-1-decyl phthalate, di-n-undecyl phthalate, di-n-tridecyl phthalate, tri-2-ethylhexyl trimellitate, di-2-ethylhexyl adipate, di-2-ethylhexyl sebacate, di-2-ethylhexyl azelate, dibutyl sebacate, glyceryl monocaprylate, and glyceryl monocaprate.
27. The pharmaceutical composition according to claim 23, wherein the glidant is selected from the group consisting of silicon dioxide, colloidal silica, powdered cellulose, talc, dicalcium phosphate, and mixtures thereof.
28. A pharmaceutical composition according to claim 1, wherein the composition is a one or multiple daily dose form comprising from 1 to 3000 mg of rifaximin in one or more polymorph forms or raw form.
29. A pharmaceutical composition according to claim 28, comprising 50 to 800 mg of rifaximin in one or more polymorph forms or raw form.
30. A pharmaceutical composition according to claim 28, wherein the unit dosage is 200 mg, or 400 mg or 550 mg or 600 mg or 800 mg of rifaximin in one or more polymorph forms or raw form.
31. A method of treating or preventing a gastrointestinal disorder in a subject in need thereof comprising administering to said subject the pharmaceutical composition of claim 1, wherein said gastrointestinal indication is selected from the group consisting of traveler's diarrhea, infectious diarrhea, an antibacterial prophylactic prior to colon surgery, Crohn's disease, small intestinal bacterial overgrowth, traveler's diarrhea prophylaxis, inflammation of intestinal mucous membrane, surgical prophylaxis and gastric dyspepsia.
32. The method according to claim 31, wherein the composition is used for the treatment of gastrointestinal inflammatory diseases.
33. The method according to claim 31, wherein the composition is used for the treatment of disease caused by E. coli.
34. The method according to claim 31, wherein said composition is further coated with a coating which includes a film coating, a sugar coating, and/or an enteric coating.
35. The method according to claim 34, wherein the coating comprises coating agents, plasticizers, antisticking agents, surfactants, coloring agents, or mixtures thereof.
36. A pharmaceutical composition according to claim 1, wherein the composition is formulated as a tablet, granule, capsule, microcapsule, tablet in capsule, or microgranules for suspension.
37. A pharmaceutical composition comprising a therapeutically effective amount of rifaximin or polymorph thereof, one or more release controlling agents and one or more pharmaceutically acceptable excipients, wherein said release controlling agents or excipients are selected from the group consisting of methacrylic acid ethylacrylate copolymer (1:1), methacrylic acid methylmethacrylate copolymer (1:2), polyvinyl acetate phthalate, polyvinylpyrrolidone, cellulose, microcrystalline cellulose, hydroxyethyl cellulose, hydroxypropyl cellulose, hydroxypropyl methylcellulose, carboxymethyl cellulose, methylcellulose, hydroxypropyl methyl cellulose, cellulose acetate, cellulose acetate butyrate, cellulose acetate propionate, ethyl cellulose, hydroxypropyl methylcellulose phthalate, hydroxypropyl cellulose acetate phthalate/cellulose acetate phthalate, cellulose acetate phthalate, cellulose acetate latex, hydroxypropyl methylcellulose, sodium carboxymethyl cellulose, acetylated glycerides, diethylphthalate, ethylene glycol, propylene glycol, polyethylene glycol, polysorbate, polyoxyethylenate esthers, amylose, chitosan, chondroitin sulphate, dextran, guar gum, inulin, adipates, azelates, benzoates, citrates, isoebucates, phthalates, sebacates, stearates, glycols, acetylated monoglycerides, butyl phthalyl butyl glycolate, dibutyl tartrate, diethyl phthalate, dimethyl phthalate, ethyl phthalyl ethyl glycolate, glycerin, triacetin citrate, triacetin, tripropinoin, diacetin, dibutyl phthalate, acetyl monoglyceride, castor oil, triethyl citrate, polyhydric alcohols, acetate esters, gylcerol triacetate, acetyl triethyl citrate, dibenzyl phthalate, dihexyl phthalate, butyl octyl phthalate, diisononyl phthalate, butyl octyl phthalate, dioctyl azelate, epoxydized tallate, triisoctyl trimellitate, diethylhexyl phthalate, di-n-octyl phthalate, di-1-octyl phthalate, di-1-decyl phthalate, di-n-undecyl phthalate, di-n-tridecyl phthalate, tri-2-ethylhexyl trimellitate, di-2-ethylhexyl adipate, di-2-ethylhexyl sebacate, di-2-ethylhexyl azelate, dibutyl sebacate, glyceryl monocaprylate, glyceryl monocaprate, fatty acids, fatty acid esters, waxes, zein, simethicone, polyvinyl alcohol, titanium dioxide, micronized silica, silica gel, fumed silica, colloidal silica, colloidal silicon dioxide, glycerol monostearate, magnesium trisilicate, magnesium stearate, sucrose, sorbitol, mannitol, glucose, dextrose, lactose, saccharine, acesulfame, neohesperedine, pectin, xanthan gum, agar, dicalcium phosphate, calcium sulphate, kaolin, sodium chloride, dry starch, starch, corn starch, potato starch, gelatin, synthetic gum, sodium alginate, talc, calcium stearate, stearic acid, hydrogenated vegetable, oils, croscarmelose, crospovidone, sodium starch glycolate, coloring agents, acetylated monoglycerides, citrate esters, albumin lake, iron oxide, and mixtures thereof.
38. A pharmaceutical composition comprising a therapeutically effective amount of rifaximin or polymorph thereof, one or more release controlling agents and one or more pharmaceutically acceptable excipients, wherein said composition is multi-layered.
39. A pharmaceutical composition according to claim 38, wherein at least one enteric layer is present.
40. A pharmaceutical composition according to claim 39, further comprising a hydrophilic film.
Description
[0001] This application is a continuation under 35 U.S.C. .sctn.120 of U.S. patent application Ser. No. 11/814,628, filed Jul. 24, 2007, which is a 35 U.S.C. .sctn.371 national phase filing of PCT Application No. PCT/EP2006/002022, filed Mar. 6, 2006, which claims priority to Italian Application No. BO 2005 A 000123, filed Mar. 7, 2005, the contents of each of which are hereby incorporated by reference.
[0002] The object of the invention consists of pharmaceutical formulations containing rifaximin in the shape of microgranules made gastroresistant by an insoluble polymer at pH values between 1.5 and 4.0 and soluble at pH values between 5.0 and 7.5, by their preparation and by their use in the manufacture of medicinal preparations useful in the treatment of inflammatory bowel diseases (IBD) and mainly Crohn's disease.
BACKGROUND OF THE INVENTION
[0003] The intestinal apparatus is affected by many inflammatory diseases generally capped as inflammatory bowel diseases. In particular, Crohn's disease is a severe chronic inflammatory disease affecting various levels of the digestive tract, from the mouth to the anus, particularly it can be observed in the last portion of the small intestine, either the ileum, the colon or both and sometimes in the mucous membrane of the colon and in the anal region as well. In the interested intestinal part, inflammation, swelling and ulceration occur in the whole intestinal wall causing stenosis, bleeding ulcers and pain, while the non-affected tissue portions appear normal. Crohn's disease exhibits alternate periods of inflammatory symptoms of variable gravity with symptoms such as: diarrhea, abdominal pain, weight loss often accompanied by rhagades or peri-rectal fistulas. From two-thirds to three-quarters of patients with Crohn's disease require surgery at some point in their lives. Surgery is used either to relieve symptoms that do not respond to medical therapy or to correct complications such as blockage, perforation, abscess, or bleeding in the intestine.
[0004] The role of the intestinal bacterial flora in the etiopathogenesis of the intestinal inflammatory diseases and in particular in Crohn's disease is evidenced by, for example, the frequency of localization to areas with high bacteria concentrations, see Jannowitz, H. D., in Inflamm. Bowel Dis., 1998, 44, 29-39; the deviation of the faecal flow determines remission of the endoscopic damages which reappear again at restoration of the canalization, see Rutgeerts, P., in Lancet, 1991, 338, 771-774; experimental models, e.g., knock-out mouse for the IL-10 gene or others, show that spontaneous colitis does not develop if a "germ-free" condition is maintained, see Blumberg R. S., in Curr. Opin. Immunol., 1999, 11(6), 648-56; inflammation of intestinal mucous membrane develops after the contact with faecal material, see Harper P. H., in Gut 1985, 26(3), 279-84; after surgical "curative" therapy consisting of ileocolic anastomosis, antibiotic treatment delays the development of both endoscopic and clinic relapses, see Cameron J. L. in Ann. Surg., 1992, 215, 546-52; and the presence of fistulae or abscess-sacs points out further the bacterial contribution to the disease development.
[0005] Crohn's disease has previously been treated with drugs that are able to decrease or control the inflammation, e.g., cortisones, salazopirine, mesalazine, immunosupressants, specific chemotherapeutics, antibiotics and protein inhibitors of the actions of the Tumor Necrosis Factor (TNF). During the treatment of the acute phase of the inflammatory bowel disease, stronger treatments are often necessary to ensure parenteral alimentation, to reconstitute the loss of proteins, liquids and salts, to permit the intestine to rest to facilitate the cicatrisation of ulcers. The purpose of the therapy is to decrease the frequency of the reappearance of symptoms and to reduce the seriousness acute episodes when they appear. However, with current therapies, acute episodes respond in about 50-70% of the cases, but relapses occur in 80% of the patients.
[0006] Antibiotics are usually used to decrease the growth of the luminal bacteria; to decrease the inflammatory state sustained as a result of the bacterial growth; to reduce symptoms of the acute phase of the disease, e.g., diarrhea, intestinal pain and meteorism; and to prevent and to cure septic complications, e.g., abscesses, fistulas and toxic state.
[0007] The most frequently used antibiotics are systemically absorbed, for example, metronidazole (active against some parasites along with many anaerobic bacteria) and ciprofloxacin (active against such bacteria as E. Coli and aerobic enterobacteria). Metronidrazol has been used at a dose of 10-20 mg/kg/day for 4 months (Sunterland, L. Gut, 199132, 1071-5), while ciprofloxacin has been used at a dose of 1000 mg/day for 6 weeks (Colombel J. F. in Am. J. Gastoenterol., 1999, 94, 674-8), while Prantera in Am. J. Gastoenterol., 1996, 91, 328-32, adopted the combination of the two antibiotics using metronidazole at the dose of 1000 mg/day and ciprofloxacin at the dose of 1000 mg/day for 12 weeks. The high systemic bioavailability of these antibiotics is at the root of their high incidence of side effects registered in long-term therapies, which negatively impacts their use. The incidence of side effects in the use of metronidazole ranges from 10% to 20%, depending on the dose and the treatment duration. The most frequent side-effects include metallic taste, gastric intolerance, nausea, glossitis, cephalea, vertigo, ataxia, convulsion and neurotoxicity. Peripheral neuropathy has been recorded in 50-85% of the long-term treated patients, which may regresses only after several months of therapeutic interruption. The percentage of side effects described in ciprofloxacin studies is variable and depends in part on the dosage and the duration of the treatment. The most frequent of the side effects are of gastrointestinal origin, but an increase of the transaminase and skin reactions have also been frequently described. Thus, there is a need in the art for a long-term treatment option for inflammatory diseases of the digestive tract, e.g., gastro enteric pathologies.
[0008] It is advantageous for pharmaceutical preparation used for treating inflammatory bowel diseases (e.g., gastro enteric pathologies) that are based on antibiotics to have one of more of the following characteristics: intestinal level activity, low absorption, bacteria level control in the intestinal lumen, wide spectrum of actions against the microbes (e.g., intestinal Gram-positive, Gram-negative, aerobic and anaerobic components), possibility of long term therapy without side effects, ease of administration to facilitate compliance even with the potential of high dosage necessity, e.g., long-term dosing and/or multiple dosing per day.
[0009] An antibiotic possessing several of these characteristics is rifaximin (INN; see The Merck Index, XIII Ed., 8304), which is characterized by a wide spectrum of action against many Gram-positive and Gram-negative bacteria, including aerobic and anaerobic bacteria. Bioavailability studies in healthy volunteers have shown that, when given orally, less than 1% of rifaximin is absorbed and it concentrates in the intestinal lumen and in the feces described herein (Descombe J. J. et al. Pharmacokinetic study of rifaximin after oral administration in healthy volunteers. Int J Clin Pharmacol Res, 14 (2), 51-56, (1994)). The absence of rifaximin absorption has been confirmed in patients affected by chronic bowel disease, (see Rizzello, Eur. J. Clin. Pharmacol. (1998) 54, 91-93). Moreover, the low absorption profile of rifaximin reduces the incidence of side effects and the unwanted risk of pharmacological interactions. Thus, rifaximin may be considered useful in the therapy of inflammatory chronic bowel disease and particularly in Crohn's disease. The potential efficacy of rifaximin in chronic inflammatory bowel diseases has been confirmed, see Gionchetti, P., Dig. Dis. Sci., 1999, 44, 1220-1, who hypothesized the use of rifaximin in patients with moderate or severe ulcerative colitis refractory to steroid-treatment.
[0010] Rifaximin has been described in Italian Patent IT 1154655 (1980) and EP 0161534 (1985), both of which are incorporated herein by reference in their entirety for all purposes. EP 016153 discloses a process for rifaximin production using rifamycin 0 as the starting material (The Merck Index, XIII Ed., 8301).
[0011] Guidance for rifaximin crystallization and drying are described in Italian Patent Application No. MI2003A002144 (2003), in European Patent Application No. EP 1557421 (2003); in U.S. patent application Ser. No. 10/728,090 (2003) in PCT Patent Application No WO2005/044823; all of which are incorporated herein by reference in their entirety for all purposes. The experimental conditions described in these patents allow yielding polymorphic forms of rifaximin named Form .alpha., Form .beta., Form .gamma., Form .delta. and Form .epsilon., respectively.
[0012] Rifaximin is approved in certain countries for the treatment of pathologies whose etiology is in part or totally due to intestinal acute and chronic infections sustained by Gram-positive and Gram-negative bacteria, with diarrhea syndromes, altered intestinal microbial flora, summer diarrhea-like episodes, traveler's diarrhea and enterocolitis; pre- and post-surgery prophylaxis of the infective complications in gastro intestinal surgery; and hyperammonaemia therapy as coadjutant. Rifaximin is currently marketed as tablets or capsules at the dosage of 100 mg and 200 mg, in a ready to use preparation for children, or as ointment for the treatment of topical infections.
[0013] Studies on commercially available samples, particularly 200 mg tablets, have shown a potential usefulness of rifaximin in the prevention of the relapse of Crohn's disease after endoscopic resection. However, the absence of a placebo group in the clinical trial does not allow to draw confident conclusions, see Rizzello, Gut., 2000, 47, Supp. 3, A12. However, the suggested posology the use of the rifaximin 200 mg tablets has to be considered sub optimal due to the need up to six tablets a day for three months, resulting in a poor patient compliance. The 200 mg tablets of rifaximin have also been used in the treatment of Crohn's disease with dosages of 600 mg/day for 16 weeks as described by Shafran, I., Am. J. Gastroenterol., 2003, 98 (Suppl.) S-250.
[0014] Thus, there is a need in-the-art for a rifaximin pharmaceutical formulation for the treatment of infections specifically located in the intestinal tract. Previous formulations, after administration, are released and spread between the stomach and the intestine. Thus, when the rifaximin finally reaches the intestinal tract, the concentration is too low resulting in the need for increasing dosages. To maximize the therapeutic efficacy of rifaximin in the treatment of bowel diseases, new pharmaceutical formulations are provided herein and include, for example, rifaximin microgranules coated with a gastroresistant film which dissolves releasing the antibiotic only in the intestinal tract. This novel formulation maximizes contact between the active ingredient and the intestinal mucous due, in part, to the high superficial area of the microgranules. The novel formulations also allow for ease of high and low dose administration, for example, in paediatric use.
[0015] The novel gastroresistant rifaximin formulations takes advantage form the pH difference between the gastric environment (e.g., values from about 1.5 to about 4.0, depending on the state of fast or in presence of meal) and the intestinal lumen (e.g., values from 5.0 to about 7.5, depending of the tracts considered).
[0016] The novel forms also utilize the polymorphic forms of rifaximin.
[0017] The coating of pharmaceutical microgranules with gastroresistant film is a technique known by many years in the pharmaceutical field. It is generally performed in two steps: granulation and coating. Nevertheless, many active substances, including rifaximin, are characterized by a very fine particle size, for example, in case of rifaximin approximately 50% of the particles has a particle diameter between 10 .mu.m and 40 .mu.m. In such condition it is very difficult using conventional systems like fluid bed coating or pan technology. Very often agglomeration occurs or random blend of coated and uncoated particles is commonly obtained.
[0018] We have found, and this is an object of the invention, that it is possible to obtain enteric-coated microgranules of rifaximin by applying the fluid bed technology, which surprisingly allows in one step and at the same time to perform the wet-granulation of the powder and the coating of the formed microgranules with a polymer resistant to the gastric environment, commonly called enteric coating. With this approach the chief disadvantages of the wet-granulation and microgranule coating, which are in separate steps involved as well as the time and labor necessary to carry out the entire procedure, especially on the large scale, are minimized. This result comes from a combination between the rifaximin properties and a proper balancing the quantity of rifaximin, of enteric polymer, of plasticizer, and process parameters.
[0019] The efficiency of this technology in providing a complete coating layer around rifaximin is demonstrated by SEM microscopy as reported in FIGS. 1a (Scanning electron microscopy of rifaximin gastroresistant microgranules) and 1b (Scanning electron microscopy of single granule of rifaximin gastroresistant microgranules), where it is clearly show that rifaximin is fully coated by the enteric polymer. The particles sizes are quite homogeneous without large clots or very fine powder. If present, one or both of these aspects would have negative impact in any further medicinal preparation.
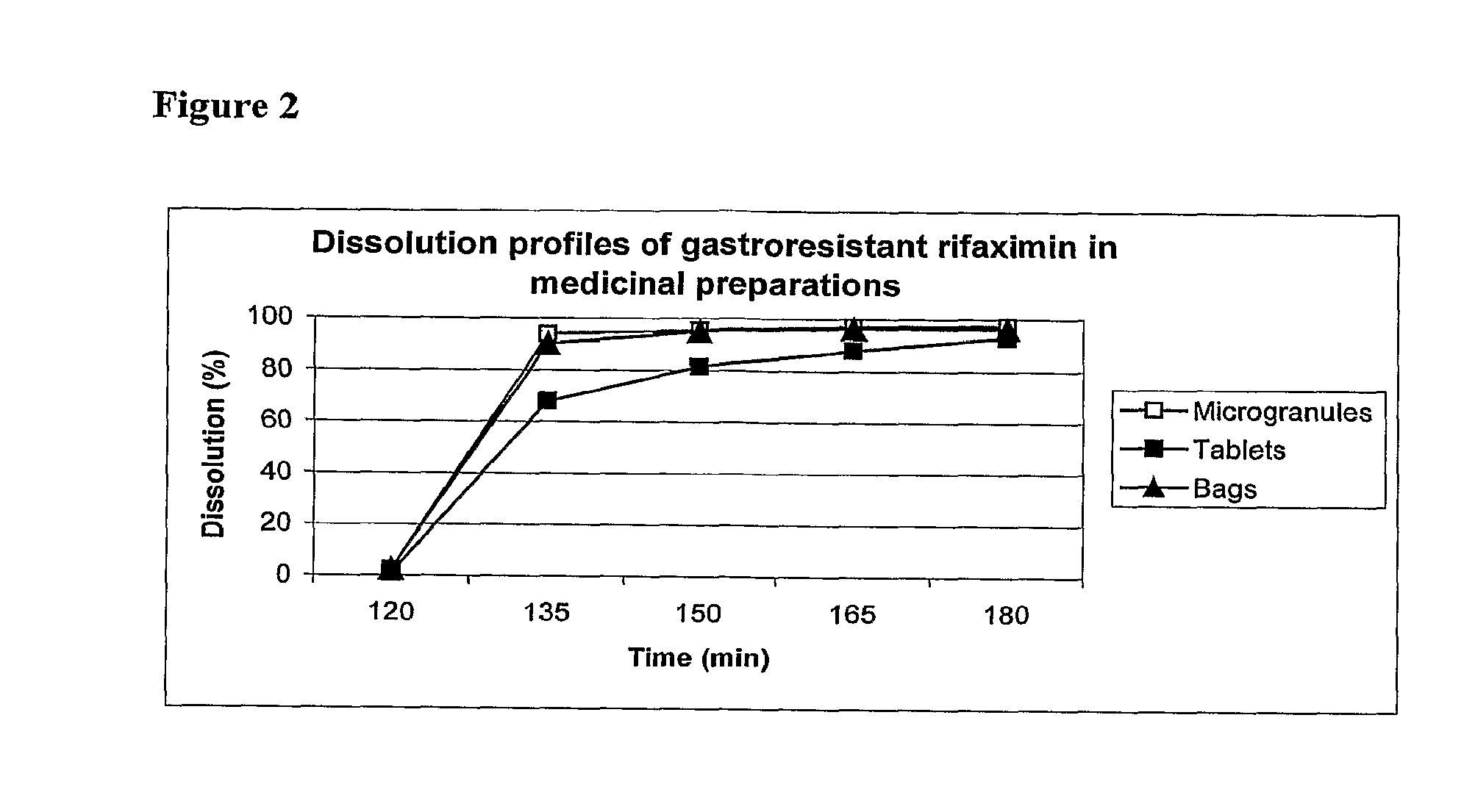
[0020] As confirmation of the completeness of the coating, the dissolution profile of the gastroresistant microgranules of rifaximin shows that rifaximin is completely retained at low pH and released at pH higher than 5.0, as reported in FIG. 2 (Dissolution profiles).
[0021] In order to maximize the release of the active ingredient near the intestinal mucous membrane it has been utilized high pH difference between the gastric environment, values from 1.5 to 4.0, depending on the state of fast or in presence of meal and the intestinal lumen, values from 5.0 to 7.5 depending of the tracts considered. For this purpose, enteric polymeric materials having the property to solubilize at pH values between 5.0 and 7.5 have been used, to include: methacrylic acid copolymers with an acrylic or methacrylic ester like methacrylic acid ethylacrylate copolymer (1:1) and methacrylic acid methylmethacrylate copolymer (1:2), polyvinyl acetate phthalate, hydroxypropyl cellulose acetate phthalate and cellulose acetate phthalate, products available on the market for example with the trademarks KOLLICOAT.RTM., EUDRAGIT.RTM., AQUATERIC.RTM., AQOAT.RTM..
[0022] The application of these gastroresistant films to rifaximin powder or granules is performed with conventional apparatus for fluid-bed coating technology. The film coating, dissolved in organic solvents or suspended in water, is applied by spraying on powders or granules maintained in suspension with air in fluid bed systems. The most used organic solvents are: methylene chloride, methyl alcohol, isopropyl alcohol, acetone, tri-ethyl acetate and ethyl alcohol. Alternatively, the polymeric gastroresistant material can be applied suspended in water. This technique is preferable because it doesn't need the use of solvents and so it avoids the toxicological and safety related problems.
[0023] Other excipients with anti-agglomerative properties, like talc; plasticizing properties, like acetylated glycerides, diethylphthalate, propylene glycol and polyethylene glycol; surfactants like polysorbate and polyoxyethylenate esters, anti-foam as well as anti-sticking agents can be added together with the polymeric material.
[0024] The successful application of the above mentioned technology to the coating of rifaximin powder is remarkable because it is not in the state-of-art of fluid-bed technology to spray the enteric polymer directly on the active ingredient without any preliminary treatment like granulation or layering the active ingredient on inert particles. Indeed, several drawbacks could occur without any powder pre-treatment such as large clamp formation, large range of particle diameter, inhomogeneous composition of microgranules, no uniform coating layer. The occurrence of some of these drawbacks is common with rifaximin, the powder of which is composed by a fine particles, and is extremely hydrophobic, electrostatic, hygroscopic and difficult to be mixed with common excipients in powder. Moreover it has a predisposition to segregate not allowing homogenous mixture. In presence of such unfavourable characteristics to get coated rifaximin would required the use of more than one step and a large quantity of excipients, which would limit the pharmaceutical strengths of human dosage.
[0025] As further advantage of the present invention, the gastroresistant microgranules of rifaximin prepared on the basis of the described technology of the present invention can directly be used to fill capsules or can be mixed with excipients and sweetener enhancers giving the possibility of an aqueous suspension administration.
[0026] In addiction and more remarkably the gastroresistant microgranules of rifaximin can also be directly used for tablet preparation through direct compression technology by adding conventional vehicles or carriers. As additional advantage, the tablets can be scored in order to modulate the dose strength or to be crushed to facilitate the ingestion without losing the gastroresistant property of the microgranules.
[0027] All these opportunities confer significant value to the technology described in the present invention to prepare gastroresistant microgranules of rifaximin, making it suitable for a wide modulation of dosages and pharmaceutical forms.
[0028] In conclusion, the present invention shows, with respect to other marketed rifaximin preparations, remarkable improvements that can be summarized on the possibility to manufacture in only one steps gastroresistant microgranules of rifaximin, which remain insoluble in the stomach (e.g., at a range of pH between about 1.5 and about 4.0) and soluble in the intestine (e.g., at higher pH, for example between about 5.5 and about 7.5.), to administer high dose, targeting the maximum release of the active ingredient in the intestine and at the same time maximizing its contact with the intestinal mucous membrane because of the high superficial area of the microgranules.
BRIEF DESCRIPTION OF DRAWINGS
[0029] FIG. 1A is an image of microgranules of rifaximin.
[0030] FIG. 1B is a closer image of a single microgranule.
[0031] FIG. 2 is a line graph which shows the dissolution profiles of gastroresistant rifaximin.
[0032] FIG. 3 is a scanning electron microscope image of rifaximin gastroresistant microgranules that are compressed in tablets.
DESCRIPTION OF INVENTION
[0033] The object of the present invention consists of pharmaceutical formulations containing microgranules of rifaximin coated with a gastroresistant polymer which is insoluble at pH values ranging between 1.5 and 4.0 and soluble at pH values ranging between 5.0 and 7.5, of their preparation and their use in intestinal inflammatory bowel diseases and in particular in Crohn's disease.
[0034] The microgranules may be from between about 1 micron to about 900 microns in diameter, or more preferably from between about 10 microns to about 500 microns in diameter.
[0035] The gastroresistance can be obtained using any material insoluble at pH values ranging between about 1 to about 4.9, from about 1.4 to about 4.2, or from about 1.5 and about 4.0. Suitable polymers may also be soluble at pH values ranging from between about 5.0 to about 7.0, 5.0 to about 7.5, or 5.0 and about 7.7 and above.
[0036] Polymeric materials utilized in the gastroresistant rifaximin formulations solubilize, as discussed above, at pH values consist with the intestinal lumen, for example, from between about 4.9 and about 7.7, and can be used as gastroresistant, entero-soluble coatings for drug release in the intestine when desired. Examples of suitable polymeric materials include, for example, acrylic polymers, methacrylic acid copolymers with an acrylic or methacrylic ester (e.g., methacrylic acid ethylacrylate copolymer (1:1) and methacrylic acid methylmethacrylate copolymer (1:2), polyvinyl acetate phthalate, hydroxypropyl cellulose acetate phthalate and cellulose acetate phthalate), as well as cellulose acetate phthalate, hydroxypropyl methylcellulose phthalate, polyvinyl acetate phthalate. Commercially available products include, for example, KOLLIKOAT.RTM., EDRAGIT.RTM. (e.g., EUDRAGIT 40), AQUATERIC, AQOAT.RTM..
[0037] The enteric materials, which are soluble at higher pH values, are frequently used for colon-specific delivery systems and are employable in the gastroresistant rifaximin formulations described herein. The enteric polymers used can also be modified by mixing with other coating products that are not pH sensitive. Examples of such coating products include, for example, the neutral methacrylic acid esters with a small portion of trimethylammonioethyl methacrylate chloride, sold currently under the trade names EUDRAGIT.RTM. and EUDRAGIT.RTM. RL; a neutral ester dispersion without any functional groups, sold under the trade names EUDRAGIT.RTM. NE3OD and EUDRAGIT.RTM. NE30, EUDRAGIT.RTM. 40; polysaccharides, like amylose, chitosan, chondroitin sulphate, dextran, guar gum, inulin and pectin; and other pH independent coating products.
[0038] The polymer is from between about 5% and about 75% of the weight of the microgranule. In other embodiments, the polymer is from between about 10% and about 60%, 20% and about 55%, about 30% to about 80%, or 25% and about 50% of the weight of the microgranule. The weight percent of the polymer to the weight of the microgranule can depend, in part, on the polymer used, the temperature of the polymer, the formulation (e.g., bag, pill, capsule, etc.), and the pH at which the polymer is soluble.
[0039] The gastroresistant rifaximin microgranules may further comprise one or more of a diluents, plasticizer, anti-agglomerative, anti-sticking, glidants, anti-foam surfactants, or coloring substances. These, along with other polymers and coating (e.g., protective coatings, over-coatings, and films) are described below.
[0040] Suitable ingredients can be incorporated into the coating formula such as plasticizers, which include, for example, adipates, azelates, benzoates, citrates, isoebucates, phthalates, sebacates, stearates and glycols. Representative plasticizers include acetylated monoglycerides, butyl phthalyl butyl glycolate, dibutyl tartrate, diethyl phthalate, dimethyl phthalate, ethyl phthalyl ethyl glycolate, glycerin, ethylene glycol, propylene glycol, triacetin citrate, triacetin, tripropinoin, diacetin, dibutyl phthalate, acetyl monoglyceride, polyethylene glycols, castor oil, triethyl citrate, polyhydric alcohols, acetate esters, gylcerol triacetate, acetyl triethyl citrate, dibenzyl phthalate, dihexyl phthalate, butyl octyl phthalate, diisononyl phthalate, butyl octyl phthalate, dioctyl azelate, epoxydised tallate, triisoctyl trimellitate, diethylhexyl phthalate, di-n-octyl phthalate, di-1-octyl phthalate, di-1-decyl phthalate, di-n-undecyl phthalate, di-n-tridecyl phthalate, tri-2-ethylhexyl trimellitate, di-2-ethylhexyl adipate, di-2-ethylhexyl sebacate, di-2-ethylhexyl azelate, dibutyl sebacate, glyceryl monocaprylate, and glyceryl monocaprate. Other various layers, as recognized by one of skill in the art are also envisioned. The amount of plasticizer used in the polymeric material typically ranges from about 10% to about 50%, for example, about 10, 20, 30, 40, or 50%, based on the weight of the dry polymer.
[0041] Optional modifying components of a protective layer which can be used over the enteric or other coatings include a water penetration barrier layer (semi-permeable polymer) which can be successively coated after the enteric or other coating to reduce the water penetration rate through the enteric coating layer and thus increase the lag time of the drug release. Coatings commonly known to one skilled in the art can be used for this purpose by coating techniques such as fluid bed coating using solutions of polymers in water or suitable organic solvents or by using aqueous polymer dispersions. For example, useful materials include cellulose acetate, cellulose acetate butyrate, cellulose acetate propionate, ethyl cellulose, fatty acids and their esters, waxes, zein, and aqueous polymer dispersions such as EUDRAGIT RS and RL 30D, EUDRAGIT.RTM. NE 30D, EUDRAGIT.RTM. 40, AQUACOAT.RTM., SURELEASE.RTM., cellulose acetate latex. Combinations of the polymers and hydrophilic polymers such as hydroxyethyl cellulose, hydroxypropyl cellulose (KLUCEL.RTM., Hercules Corp.), hydroxypropyl methylcellulose (METHOCEL.RTM., Dow Chemical Corp.), polyvinylpyrrolidone may also be used.
[0042] Anti-foaming agents can also be included in the gastroresistant rifaximin formulations. In one embodiment, the anti-foaming agent is simethicone. The amount of anti-foaming agent used typically comprises from 0% to 0.5% of the final formulation. Other agents can be added to improve the processability of a sealant or barrier layer. Such agents include, for example, talc, colloidal silica, polyvinyl alcohol, titanium dioxide, micronized silica, fumed silica, glycerol monostearate, magnesium trisilicate, and magnesium stearate, or a mixture thereof.
[0043] The amount of polymer to be used in the gastroresistant formulations is typically adjusted to achieve the desired drug delivery properties, including the amount of drug to be delivered, the rate and location of drug delivery, the time delay of drug release, and the size of the multiparticulates in the formulation. The combination of all solid components of the polymeric material, including co-polymers, fillers, plasticizers, and optional excipients and processing aids, typically provides about 1% to about 50% weight of the core.
[0044] The gastroresistant rifaximin microgranules comprise rifaximin in a polymorphous form and/or a raw form. The forms in any microgranule may be a mixture or may be a pure form. The form of rifaximin may depend, in part, on the form of the rifaximin that is coated, on the composition of excipients, and on the process used to form the microgranules. The rifaximin polymorphous forms are selected from Form .alpha., Form .beta., Form .gamma., Form .delta., or Form .epsilon. of rifaximin, mentioned above.
[0045] The mixture containing the gastroresistant material is prepared by suspending the components in demineralised water and homogenizing the suspension with an high speed mixing system, preferably an Ultra Turax homogeniser, in order to obtain a homogeneous suspension containing between 15% and 30% of solid particles. The homogeneous suspension containing the gastroresistant material can be applied by means of a coating system or a fluid bed apparatus.
[0046] In the current invention, fluid bed technology has been used. The mixture containing the active ingredient is maintained in suspension by a flux of warm air, at the same time the gastroresistant suspension is sprayed by means of a jet applied in a top part (top spray) or in a low part (bottom spray--Wurster system) of the apparatus. For example, a fluid bed apparatus type Glatt GPG 30 has been used with a Wurster system of 18 inch with a 1.8 mm spray jet.
[0047] Process parameters including the air entering temperature, the product temperature and the speed of film application are specifically controlled. The speed of film application and the air temperature are balanced to avoid overheating of the product resulting in a non-homogeneous gastroresistant microgranule formation (too fast drying of the product) or, an agglomeration of the mixture to be coated to slow drying of the product.
[0048] In formulating, for example, a 25 kg batch of gastroresistant rifaximin, a jet spray between 150 and 300 g/min may be used. A jet spray 150 and 250 g/min, and pressures between 1.0 and 1.5 bar may also be used. The speed and pressure may be independently manipulated. The product temperature, during the spraying is maintained at a constant temperature between about 20.degree. C. and about 40.degree. C. The air temperature in entrance may also be regulated at between about 40.degree. C. and about 75.degree. C., preferably between about 60.degree. C. and about 70.degree. C.
[0049] The obtained gastroresistant microgranules are formulated for medical preparations in order to obtain, after adding water, a suspension with pleasant taste for the patients. Sweeteners agents like: sucrose, sorbitol, mannitol, saccharine, acesulfame, neohesperedine; suspending agents like polyvinyl pyrrolidone (PVP), sodium carboxymethyl cellulose, pectin, xanthan gum, agar and glidants like silica gel can be added to the gastroresistant microgranules to this end.
[0050] The gastroresistant microgranules are mixed with the above mentioned excipients in a suitable apparatus like a biconical mixer or V mixer for the time necessary to obtain the homogeneity of the gastroresistant microgranules inside the mixture. The ratio between gastroresistant microgranules and excipients is between 1:0.1 and 1:10, preferably between 1:0.5 and 1:5. The obtained mixture can be divided in bags containing a quantity of rifaximin between 1 mg and 3000 mg, preferably between 50 mg and 800 mg.
[0051] The obtained gastroresistant microgranules of rifaximin can be directly compressed in tablet after having mixed with appropriate excipients such as diluents such as dicalcium phosphate, calcium sulphate, cellulose, microcrystalline cellulose (AVICEL.RTM.), hydroxypropyl methyl cellulose, corn starch, lactose, kaolin, mannitol, sodium chloride, dry starch; binders such as starch, gelatin, sugars as sucrose, glucose, dextrose, lactose, synthetic gum, sodium alginate, carboxymethyl cellulose, methylcellulose, polyvinylpyrrolidone, polyethylene glycol, ethylcellulose, water, waxes, alcohol; lubricants such as talc, magnesium stearate, calcium stearate, stearic acid, hydrogenated vegetable, oils, polyethylenglycol; glidants such as colloidal silicon dioxide, talc; disintegrants such as corn and potato starch, croscarmelose, crospovidone, sodium starch glycolate, coloring agents, sweeteners such as sucrose, sorbitol, mannitol, saccharine, acesulfame, neohesperedine.
[0052] Conventional technology and apparatus known to expert-of-art of tablet preparation can be applied. The gastroresistant microgranules are mixed with the above mentioned excipients in a suitable apparatus like a biconical mixer or V mixer for the time necessary to obtain the homogeneity of the gastroresistant microgranules inside the mixture.
[0053] The gastroresistant granules of rifaximin have good properties in respect of ability to flow freely, cohesiveness and lubrication, therefore the ratio between gastroresistant microgranules and excipients is between 1:0.2 and 1:0.05, preferably between 1:0.15 and 1:0.1. The obtained mixture can be pressed in order to obtain, using a suitable punch, tablets containing a quantity of rifaximin between 50 mg and 600 mg, preferably between 100 mg and 500 mg. As described above the favorable properties of Rifaximin gastroresistant microgranules allow achieving a suitable blend for direct compression with the addiction of minimal quantity of excipients. The possibility to obtain tablets using a blend containing up to 93% of gastroresistant microgranules presents a further advantage: it allows to maintain the dose of 400 mg in a suitable size to maintain a good compliance for the patient.
[0054] Tablets can be successively coated with a conventional hydrophilic film to achieve taste-masking properties and improve appearance. Suitable materials could be: hydroxyethyl cellulose, hydroxypropyl cellulose (KLUCEL.RTM., Hercules Corp.), hydroxypropyl methylcellulose (METHOCEL.RTM., Dow Chemical Corp.), polyvinylpyrrolidone.
[0055] The tablet containing rifaximin gastroresistant microgranules can be film-coated following conventional procedure known to someone skilled in the art selecting as polymer one or more of cellulose and its substitutes such as hydropropylcellulose hydromethylcellulose, hydropropyl-methylcellulose. Alternatives to the cellulose ethers are certain acrylics, such as metacrylate and methylmetacrylate copolymers. Polymers can be employed as solutions either aqueous or organic solvent-based system. Incorporating a plasticiser the flexibility of the coating film is improved; by addition of plasticisers it is reduced the risk of film cracking and it is improved the adhesion of the film to the substrate. Examples of typical plasticisers include glycerin, propylene glicol, polyethylene glycols, triacetin, acetylated monoglycerides, citrate esthers and phtalate esthers. Colorants usually are used to improve the appearance of the product. Water-soluble and/or organic solvent-soluble dyes can be used like albumin lake, titanium dioxide, iron oxide. Finally, stabilisers such as EDTA can be added to the coating.
[0056] The picture shown in FIG. 3 (Scanning electron microscopy of rifaximin gastroresistant microgranules compressed in the tablets) and the data of FIG. 2 show that the compression does not alter the integrity of the gastroresistant layer of the microgranules compressed in the tablets.
[0057] Furthermore, the obtained gastroresistant microgranules of rifaximin have such favorable properties regarding the particle size and the capacity of flow freely to be directly used, with the addiction of inert diluents and glidants to fill in hard gelatin capsules. Examples of typical diluents include dicalcium phosphate, calcium sulphate, cellulose, microcrystalline cellulose, hydroxypropylmethylcellulose, corn starch, lactose, kaolin, mannitol, sodium chloride, dry starch, from between about 1 and to about 225 mg. In this case the density of gastroresistant microgranules, between 0.25 and 0.45 mg/ml allows to fill about 140-250 mg of Rifaximin in conventional 000 hard gelatin capsules according to the content of Rifaximin in the gastroresistant microgranules.
[0058] All the medicine preparation, namely thermo welded bags, tablets and capsules can be usefully used in the therapy of inflammatory bowel disease to include Crohn's disease.
[0059] The following examples have to be considered as a further illustration of the object of the invention and not as a limitation.
Example 1
Rifaximin Preparation in Gastroresistant Microgranules
[0060] In a fluid bed apparatus, Glatt GPC 30, with a Wurster system of 18 inches with a 1.8 mm spray jet, 25000 g of rifaximin powder and 125 g of Aerosil as fluidiser are loaded. Contemporaneously in a mixer under agitation a suspension is prepared using 48107 g of demineralised water, 9281 g of methacrylic acid ethylacrylate copolymer marketed under the trademark KOLLICOAT.RTM. MAE 100 P, 1392 g propylglycol, 2475 g of talc, 557 g of titanium dioxide FU and 62 g of iron oxide E 172. The solid components of the suspension are homogeneously mixed in demineralised water with an high speed homogeniser (Ultra Turrax). The prepared suspension feeds the spray system of the fluid bed apparatus and nebulized, at a pressure between 1.0 and 1.5 bar, trough the 1.8 mm nozzle on the mixture of rifaximin powder and Aerosil 200 maintained in suspension in the fluid bed by a warm air flow.
[0061] The applied conditions are described in table 1:
TABLE-US-00001 TABLE 1 Pre-warm Application of Process parameters phase coating solution Drying Air flow in entrance 400 .+-. 100 550 .+-. 100 350 .+-. 50 (m.sup.3/hour) Air temperature in entrance 60 .+-. 2 60.degree. C. .+-. 10 50 .+-. 2 (.degree. C.) Product temperature (.degree. C.) 32 25-27 30 .+-. 2 Jet pressure (bar) 1-1.5 .+-. 0.1 (initial phase) Jet speed (gamin) 150-200
[0062] The obtained microgranules are submitted to granulometry analysis by Light Scattering technology using Malvern Mastersizer 2000 apparatus 5 obtaining the following results: 100%<200 micron [0063] 99.17%<150 micron [0064] 90.03%<100 micron [0065] 48.37%<50 micron [0066] 6.20%<10 micron
[0067] The rifaximin in the gastroresistant microgranule preparation corresponds to 61.4% of the total particle weight.
Example 2
SEM Microscopy of Gastroresistant Microgranules of Rifaximin
[0068] A SEM Philips 515 instrument is used for the observations.
[0069] Rifaximin gastroresistant microgranules are sputtered with gold by current stream of 30 mA, getting an Au-layer of about 100 nm. An accelerating voltage of 15 kV is applied.
[0070] The images are digitally recorded with a CCD camera.
[0071] An image of microgranules of rifaximin is shown in FIG. 1A, while 20 in FIG. 1B a detail of a single microgranule is shown.
Example 3
[0072] Gastroresistant Microgranules of Rifaximin Prepared in Thermo Welded Bags
[0073] 9.12 Kg of gastroresistant rifaximin microgranules prepared according to the example 1, 19.58 Kg of sorbitol, 0.49 Kg of aspartame, 0.21 Kg of anhydrous citric acid, 2.10 Kg of pectin, 2.10 Kg of mannitol, 0.21 Kg of neohesperidine DC, 1.12 Kg of cherry flavour and 0.07 Kg of silica gel are sieved on a sieve with mesh of 0.5 mm and then mixed for 20 minutes in a V mixer. The resulting mixture is divided in thermo welded bags containing 5 grams of product corresponding to 800 mg of rifaximin. In the following Table 2 the composition of the medicinal speciality, thermo welded bag, is reported:
TABLE-US-00002 TABLE 2 Amount Components (mg) % Gastroresistant rifaximin microgranules 1303 26.06 (corresponding to 800 mg of rifaximin) Aspartame 70 1.40 Anhydrous citric acid 30 0.60 Pectin 300 6.00 Mannitol 300 6.00 Neohesperidin DC 30 0.60 Sorbitol 2797 55.94 Cherry-flavour 160 3.20 Silica gel 10 0.20
Example 4
Gastroresistant Microgranules of Rifaximin Prepared in Compressed Tablets
[0074] 9.3 Kg of gastroresistant rifaximin microgranules prepared according to the example 1,593 g of Sodium Starch Glycolate, 100 g of magnesium stearate are sieved on a sieve with mesh of 0.5 mm and then mixed for 20 minutes in a V mixer. The resulting mixture is compressed using a rotary tabletting machine (Fette 1200) equipped with oblong, scored 19.times.9 mm punches at the final weight of 718 mg (corresponding to a content of 400 mg of rifaximin).
[0075] The tablet composition is reported in Table 3.
TABLE-US-00003 TABLE 3 Amount Tablet composition mg % Rifaximin gastroresistant 650.00 90.53 microgranules (corresponding to 400 mg of rifaximin) sodium 34.95 4.87 carboxymethylcellulose Avicel PH 101 24.31 3.34 Mg-stearate 8.74 1.21 718.00 100.00
[0076] The tablets are then coated, using conventional pan equipment, with a hydroxypropylmethylcellulose film in order to improve appearance and achieve taste mask properties. The unitary film composition is reported in Table 4:
TABLE-US-00004 TABLE 4 Amount Coating composition (mg) HPMC 14.07 Titanium dioxide 4.10 Na-EDTA 0.05 Propylene glycol 1.37 Red Iron Oxide E 172 0.41
Example 5
Gastroresistant Microgranules of Rifaximin Prepared in Hard Capsule
[0077] 9.0 Kg of gastroresistant rifaximin microgranules prepared according to the example 1, are blended and sieved on 0.5 mm with 110 g of talc and 1.1 kg of lactose. The resulting mixture is introduced in hard gelatin capsules type 000 using a conventional equipment like Zanasi LZ64 at a final weight of 461.00, corresponding to a content of about 270 mg of rifaximin. The capsule composition is reported in Table 5.
TABLE-US-00005 TABLE 5 Amount Capsule composition mg % Rifaximin gastroresistant 406.00 88.01 granules (corresponding to 270 mg of rifaximin) Talc 5.00 1.01 Lactose 50.00 10.8
Example 6
Dissolution Performance of Gastroresistant Microgranule of Rifaximin Medicinal Preparations
[0078] The gastroresistance of the pharmaceutical preparation is evaluated according to what described at page 247 of the US Pharmacopeia (USP), 28.sup.a Ed.
[0079] The dissolution test of medicinal preparations containing gastroresistant microgranules of rifaximin, described in examples 1, 3 and 4 and consisting of rifaximin gastroresistant microgranules, thermo welded bags containing rifaximin gastroresistant microgranules, and tablets containing rifaximin gastroresistant microgranules, respectively, are evaluated by using the following conditions:
[0080] Equipment: SOTAX AT7 Smart
[0081] Medium: HCl 0.1 N, pH 1; after 2 hours a phosphate buffer with 2% of Sodium Lauryl Sulphate is added to bring up the pH to 6.8
[0082] Stirring speed: 100 rpm
[0083] Temperature: 37.degree. C.
[0084] Sampling time: 120, 135, 150, and 180 min.
[0085] The content of dissolved rifaximin is measured by a HPLC method.
[0086] The results, reported in Table 6, are the average of six measures and are expressed as percent of dissolution over the total amount of rifaximin.
TABLE-US-00006 TABLE 6 Time Dissolution ( % ) Medium & pH (min) Microgranules Tablets Bags HC1 0.1 N, pH 1 120 2.41 1.07 2.57 Phosphate buffer, pH 6.8 135 93.8 67.9 90.3 Phosphate buffer, pH 6.8 150 95.4 81.6 95.1 Phosphate buffer, pH 6.8 165 97.2 88.1 96.4 Phosphate buffer, pH 6.8 180 97.4 93.1 96.2
[0087] After 12 months of storage at 25.degree. C. microgranules, prepared as in Example 1, show a similar dissolution profile, precisely a dissolution of 2.2% after 120 min at pH 1 in 0.1 N hydrochloric acid and of 91.1% after 60 min in phosphate buffer at pH 6.8.
Example 7
Treatment of Crohn's Disease
[0088] The medicinal rifaximin preparation containing gastroresistant microgranules described in example 3 has been used in a clinical multi-center randomised trial versus placebo in patients affected by Crohn's disease. 55 Crohn's disease patients in acute, mild to moderate grade, phase, having CDAI (Crohn's Disease Active Index) value between 200 and 300, have been recruited. The primary end point was represented by the percentage of the patients in clinic remission defined as CDAI lower than 150 points at the end of the study. The patients, randomised in two groups: group A, of 27 patients and group B of 28 patients, have been treated for 12 weeks according to the following therapeutic schemes:
[0089] Group A: rifaximin 800 mg, administrated 2 times a day for a total 15 dosage equal to 1600 mg/die;
[0090] Group B: placebo, administrated 2 times in a day in a such quantity to correspond to the content of the dose of the active principle.
[0091] The primary end point, the clinical remission after 12 weeks of therapy, is achieved by 51.9% of the patients with the gastroresistant formulation and by 32.1% of the patient treated with placebo. Moreover only one patients of the group treated with rifaximin has been forced early to leave the clinical trial because of the therapeutic failure, while nine patients treated with placebo discontinued the treatment.
[0092] The results are summarized in Table 7
TABLE-US-00007 TABLE 7 Number of clinical Number of therapeutic Group remission failure A (rifaximin) 27 patients 14 (51.9%) 1 (3.4%) B (placebo) 28 patients 9 (32.1%) 9 (32.1%)
Example 8
Treatment of Crohn's Patients Characterized by a Protein C 5 Reactive Value Higher than Normal
[0093] At the beginning of the treatment, 31 patients had a protein C reactive value, an index of inflammation in course, higher than normal. The patients have been divided into two groups: one of 16 treated with rifaximin and the other treated with placebo, as described in example 3.
[0094] The primary end point, the clinical remission, has been obtained in 62.5% of the patients treated with the new formulation of rifaximin and in 20.5% only of the patients treated with placebo. Moreover, none of the patients of the subgroup treated with rifaximin dropped from the study for therapeutic failure, unlike 6 of the patients of the subgroup treated with placebo.
[0095] The Table 8 shows the obtained results.
TABLE-US-00008 TABLE 8 Subgroup with protein C value Number of clinical Number of higher than normal values remission therapeutic failure 16 patients treated with rifaximin 10 (62.5%) 0 (0%) 15 patients treated with placebo 3 (20%) 6 (40%)
[0096] The incidence of side effects has been similar in the two groups confirming the excellent tolerability of the rifaximin formulation in continuous and prolonged use.
* * * * *
D00000

D00001

D00002

D00003

D00004

XML
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.