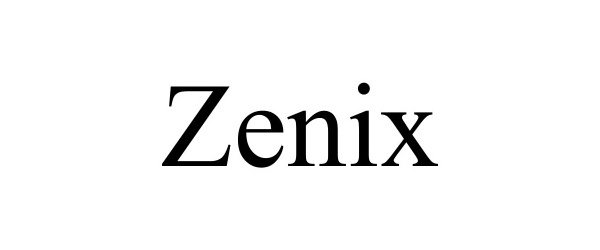ZENIX
ZOLL Medical Corporation

Mark For: ZENIX® trademark registration is intended to cover the categories of
| Research |
|
| Serial Number | 90799500 |
| Registration Number | 1666501 |
| Mark Literal Elements | ZENIX |
| Mark Drawing Type | 4 - STANDARD CHARACTER MARK |
| Mark Type | TRADEMARK |
| Register | PRINCIPAL |
| Current Location | INTENT TO USE SECTION 2022-03-22 |
| Basis | 1(b) |
| Class Status | ACTIVE |
| Primary US Classes |
|
| Primary International Class |
|
| Filed Use | No |
| Current Use | No |
| Intent To Use | Yes |
| Filed ITU | Yes |
| 44D Filed | No |
| 44E Current | No |
| 66A Current | No |
| Current Basis | No |
| No Basis | No |
| Attorney Name | Debra S. Serota |
| Attorney Docket Number | 046440627001 |
Timeline
| 2021-06-28 | Application Filed |
| 2021-07-01 | Location: NEW APPLICATION PROCESSING |
| 2021-07-01 | Status: Live/Pending |
| 2021-07-01 | Transaction Date |
| 2022-01-25 | Published |
| 2022-03-22 | Location: INTENT TO USE SECTION |
| 2022-03-22 | Status: Notice of Allowance (NOA) sent (issued) to the applicant. Applicant must file a Statement of Use or Extension Request within six |
Trademark Parties (Applicants & Owners)
| Party: | |
| Address | 269 Mill Road Chelmsford, MASSACHUSETTS UNITED STATES 018244105 |
| Legal Entity Type | Corporation |
| Legal Entity State | MASSACHUSETTS |
Documents
| 2021-06-28 | ||
| 2021-06-28 | ||
| 2021-09-13 | ||
| 2021-09-23 | ||
| 2021-12-16 | ||
| 2021-12-16 | ||
| 2021-12-17 | ||
| 2021-12-21 | ||
| 2022-01-05 | ||
| 2022-01-05 | ||
| 2022-01-25 | ||
| 2022-03-22 |
Attorney of Record
Good, Services, and Codes
IC 010. US 026 039 044. G & S: defibrillators; defibrillator parts and accessories; defibrillators with communication hardware and communication software; patient medical monitors, namely, defibrillators; defibrillator component parts featuring instantaneous feedback and information regarding chest compressions; defibrillator component parts featuring recorded software for instantaneous feedback and information regarding ventilation; defibrillators featuring recorded software that filters cardiopulmonary resuscitation (CPR) artifacts out of the electrocardiographic (ECG) waveform; defibrillators featuring recorded software for analyzing electrocardiographic (ECG) rhythms; medical apparatus, namely, respiratory equipment and monitors therefor; patient medical monitors for diagnosing cardiac arrest; patient medical monitors for monitoring critical care; medical devices for monitoring vital signs, blood properties and respiratory events; patient medical monitors, as a component of a medical device, for monitoring vital signs, blood properties and respiratory events; medical devices for monitoring and providing feedback for use during cardiopulmonary resuscitation (CPR) chest compressions and ventilation; medical devices for monitoring and providing feedback for use during cardiopulmonary resuscitation (CPR) chest compressions; medical devices for monitoring and providing feedback for use during cardiopulmonary resuscitation(CPR) ventilation; medical ventilators; medical ventilator sold together with defibrillators; medical apparatus and instruments for monitoring vital signs, blood properties and respiratory events, and downloadable software for recording, processing and transmitting medical data, for use therewith, sold as a unit; medical apparatus and instruments for monitoring vital signs, blood properties and respiratory events sold together with a tablet computer monitor; defibrillator featuring recorded software, for storing and sharing patient and medical event information
| International Codes: | 10 |
| U.S. Codes: | 026,039,044 |
| Type Code | Type |
|---|---|
| GS0101 | defibrillators; defibrillator parts and accessories; defibrillators with communication hardware and communication software; defibrillators for use as patient monitors; defibrillator component parts featuring instantaneous feedback and information regarding chest compressions; defibrillator component parts featuring software for instantaneous feedback and information regarding ventilation; software as a feature of a defibrillator, to filter CPR artifact out of the ECG waveform; software as a feature of a defibrillator, for analyzing ECG rhythms; medical apparatus, namely, respiratory equipment and monitors therefor; patient monitors for diagnosing cardiac arrest; patient monitors for critical care; medical devices for monitoring vital signs, blood properties and respiratory events; patient monitors, as a component of a medical device, for monitoring vital signs, blood properties and respiratory events; medical devices for monitoring and providing feedback on CPR namely chest compressions and ventilation; medical devices for monitoring and providing feedback on CPR namely chest compressions; medical devices for monitoring and providing feedback on CPR namely ventilation; medical ventilators; medical ventilator sold together with defibrillators; medical apparatus and instruments for monitoring vital signs, blood properties and respiratory events, and downloadable software for recording, processing and transmitting medical data, for use therewith, sold as a unit; medical apparatus and instruments for monitoring vital signs, blood properties and respiratory events sold together with a tablet computer; software as a feature of a defibrillator, for storing and sharing patient and medical event information |
Trademark Filing History
| Description | Date | Proceeding Number |
|---|---|---|
| NOA E-MAILED - SOU REQUIRED FROM APPLICANT | 2022-03-22 | |
| PUBLISHED FOR OPPOSITION | 2022-01-25 | |
| OFFICIAL GAZETTE PUBLICATION CONFIRMATION E-MAILED | 2022-01-25 | |
| NOTIFICATION OF NOTICE OF PUBLICATION E-MAILED | 2022-01-05 | |
| APPROVED FOR PUB - PRINCIPAL REGISTER | 2021-12-20 | |
| NOTIFICATION OF EXAMINERS AMENDMENT E-MAILED | 2021-12-16 | 6328 |
| EXAMINERS AMENDMENT E-MAILED | 2021-12-16 | 6328 |
| EXAMINERS AMENDMENT -WRITTEN | 2021-12-16 | 96140 |
| EXAMINER'S AMENDMENT ENTERED | 2021-12-16 | 88888 |
| NOTIFICATION OF NON-FINAL ACTION E-MAILED | 2021-09-23 | 6325 |
| NON-FINAL ACTION WRITTEN | 2021-09-23 | 96140 |
| NON-FINAL ACTION E-MAILED | 2021-09-23 | 6325 |
| ASSIGNED TO EXAMINER | 2021-09-13 | 96140 |
| NEW APPLICATION OFFICE SUPPLIED DATA ENTERED IN TRAM | 2021-09-10 | |
| NEW APPLICATION ENTERED IN TRAM | 2021-07-01 |
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.