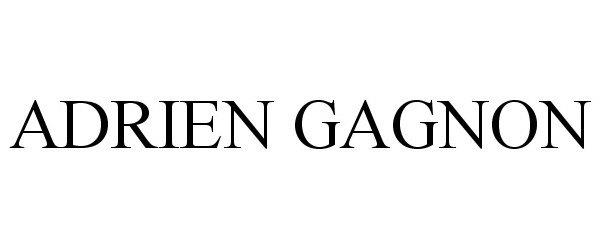ADRIEN GAGNON
SANTÉ NATURELLE A.G. Ltée
Mark For: ADRIEN GAGNON® trademark registration is intended to cover the categories of
| Research |
|
| Serial Number | 88976227 |
| Registration Number | 5940312 |
| Mark Literal Elements | ADRIEN GAGNON |
| Mark Drawing Type | 4 - STANDARD CHARACTER MARK |
| Mark Type | TRADEMARK |
| Register | PRINCIPAL |
| Current Location | PUBLICATION AND ISSUE SECTION 2019-11-13 |
| Basis | 1(a) |
| Class Status | ACTIVE |
| Primary US Classes |
|
| Primary International Class |
|
| Filed Use | No |
| Current Use | Yes |
| Intent To Use | No |
| Filed ITU | Yes |
| 44D Filed | No |
| 44E Current | No |
| 66A Current | No |
| Current Basis | No |
| No Basis | No |
| Attorney Name | Joseph V. Myers III |
| Attorney Docket Number | 105933000002 |
| Law Office Assigned | L40 |
| Employee Name | PARK, JENNY K |
Timeline
| 2016-11-00 | Date of First Use |
| 2016-11-01 | Date of First Use |
| 2016-11-01 | Date of Use In Commerce |
| 2018-07-03 | Application Filed |
| 2019-01-29 | Published |
| 2019-01-29 | Published for Opposition |
| 2019-11-13 | Location: PUBLICATION AND ISSUE SECTION |
| 2019-12-17 | Status: Live/Registered |
| 2019-12-17 | Status: Registered. The registration date is used to determine when post-registration maintenance documents are due. |
| 2019-12-17 | Trademark Registered |
| 2019-12-17 | Transaction Date |
Trademark Parties (Applicants & Owners)
| Party: | |
| Address | 3555 Matte Blvd., Door C Brossard, Quebec CANADA J4Y2P4 |
| Legal Entity Type | Limited Liability Company |
| Legal Entity State | CANADA |
| Party: | |
| Address | 3555 Matte Blvd., Door C Brossard, Quebec CA J4Y2P4 |
| Legal Entity Type | Limited Liability Company |
| Legal Entity State | CA |
*multiple parties listed, check assignment documents for ownership information
Documents
| JPEG | 2018-07-03 | |
| MULTI | 2018-07-03 | |
| XML | 2018-07-23 | |
| MULTI | 2018-07-24 | |
| XML | 2018-10-30 | |
| XML | 2018-10-30 | |
| XML | 2018-12-10 | |
| MULTI | 2018-12-20 | |
| MULTI | 2018-12-20 | |
| XML | 2019-01-09 | |
| XML | 2019-01-09 | |
| XML | 2019-01-29 | |
| XML | 2019-03-26 | |
| XML | 2019-09-25 | |
| XML | 2019-09-27 | |
| MULTI | 2019-09-27 | |
| MULTI | 2019-10-29 | |
| JPEG | 2019-10-29 | |
| MULTI | 2019-10-29 | |
| APPLICATION/XML | 2019-11-12 | |
| MULTI | 2019-11-13 | |
| APPLICATION/XML | 2019-11-14 | |
| MULTI | 2019-11-14 | |
| 2019-12-17 |
Attorney of Record
Good, Services, and Codes
IC 005. US 006 018 044 046 051 052. G & S: Natural products for human use, namely, a formula composed of saw palmetto, pygeum, stinging nettle, white deadnettle, common heather and bearberry, all in the form of capsules for relieving benign prostate hyperplasia symptoms; collagen, in the form of tablets for relieving joint pain and symptoms related to arthritis and osteoarthritis and for contributing to the formation of healthy cartilage; Natural products for human use, namely, capsules containing plant extracts to assist in weight loss; Natural products for human use, namely, plant and vitamin extracts in the form of ampoules and capsules for soothing respiratory symptoms due to a cold, flu, bronchitis and for increasing the bio-availability of Vitamin C by decreasing its elimination and oxidation; Natural products for human use, namely, plant extracts in the form of infusions, with purifying, eliminative, diuretic and laxative actions; Natural products for human use, namely, a supplement formula made of vitamins, plants and adaptogenic roots in the form of energizing ampoules, to increase resistance to stress and decrease the feeling of fatigue; Natural products for human use, namely, a supplement formula made of adaptogenic plants and roots in the form of energizing ampoules, to increase resistance to stress and decrease the feeling of fatigue, to decrease hyperglycemia and stimulate weakened immune systems and cognitive functions; Natural products for human use, namely, plant and root extracts in the form of capsules and ampoules to assist in slowing the advancement of benign prostate hypertrophy, controlling sleep disorders; a fatty acid and vitamin formula in the form of capsules for physical endurance, cholesterol level management and for the maintenance of good health; Natural products for human use, namely, plant extracts and antioxidant roots and fish oils in the form of capsules, gelcaps, liquids, to improve the cognitive functions of patients suffering from Alzheimer's or minor memory disorders, to contribute to the development and proper functioning of the brain; Natural products for human use, namely, type II collagen containing chondroitin and hyaluronic acid in the form of capsules for maintaining connective tissues, namely, joints and skin; Natural products for human use, namely, gel in the form of ampoules to combat fatigue. FIRST USE: 20161100. FIRST USE IN COMMERCE: 20161100
| International Codes: | 5 |
| U.S. Codes: | 006,018,044,046,051,052 |
| Type Code | Type |
|---|---|
| GS0051 | Natural products for human use, namely, a formula composed of saw palmetto, pygeum, stinging nettle, white deadnettle, common heather and bearberry, all in the form of capsules for relieving benign prostate hyperplasia symptoms; collagen, in the form of tablets for relieving joint pain and symptoms related to arthritis and osteoarthritis and for contributing to the formation of healthy cartilage; Natural products for human use, namely, capsules containing plant extracts to assist in weight loss; Natural products for human use, namely, plant and vitamin extracts in the form of ampoules and capsules for soothing respiratory symptoms due to a cold, flu, bronchitis and for increasing the bio-availability of Vitamin C by decreasing its elimination and oxidation; Natural products for human use, namely, plant extracts in the form of infusions, with purifying, eliminative, diuretic and laxative actions; Natural products for human use, namely, a supplement formula made of vitamins, plants and adaptogenic roots in the form of energizing ampoules, to increase resistance to stress and decrease the feeling of fatigue; Natural products for human use, namely, a supplement formula made of adaptogenic plants and roots in the form of energizing ampoules, to increase resistance to stress and decrease the feeling of fatigue, to decrease hyperglycemia and stimulate weakened immune systems and cognitive functions; Natural products for human use, namely, plant and root extracts in the form of capsules and ampoules to assist in slowing the advancement of benign prostate hypertrophy, controlling sleep disorders; a fatty acid and vitamin formula in the form of capsules for physical endurance, cholesterol level management and for the maintenance of good health; Natural products for human use, namely, plant extracts and antioxidant roots and fish oils in the form of capsules, gelcaps, liquids, to improve the cognitive functions of patients suffering from Alzheimer's or minor memory disorders, to contribute to the development and proper functioning of the brain; Natural products for human use, namely, type II collagen containing chondroitin and hyaluronic acid in the form of capsules for maintaining connective tissues, namely, joints and skin; Natural products for human use, namely, gel in the form of ampoules to combat fatigue |
| GS0051 | Natural products for human use, namely, a formula composed of saw palmetto, pygeum, stinging nettle, white deadnettle, common heather and bearberry, all in the form of capsules for relieving benign prostate hyperplasia symptoms; collagen, in the form of tablets for relieving joint pain and symptoms related to arthritis and osteoarthritis and for contributing to the formation of healthy cartilage; Natural products for human use, namely, capsules containing plant extracts to assist in weight loss; Natural products for human use, namely, plant and vitamin extracts in the form of ampoules and capsules for soothing respiratory symptoms due to a cold, flu, bronchitis and for increasing the bio-availability of Vitamin C by decreasing its elimination and oxidation; Natural products for human use, namely, plant extracts in the form of infusions, with purifying, eliminative, diuretic and laxative actions; Natural products for human use, namely, a supplement formula made of vitamins, plants and adaptogenic roots in the form of energizing ampoules, to increase resistance to stress and decrease the feeling of fatigue; Natural products for human use, namely, a supplement formula made of adaptogenic plants and roots in the form of energizing ampoules, to increase resistance to stress and decrease the feeling of fatigue, to decrease hyperglycemia and stimulate weakened immune systems and cognitive functions; Natural products for human use, namely, plant and root extracts in the form of capsules and ampoules to assist in slowing the advancement of benign prostate hypertrophy, controlling sleep disorders; a fatty acid and vitamin formula in the form of capsules for physical endurance, cholesterol level management and for the maintenance of good health; Natural products for human use, namely, plant extracts and antioxidant roots and fish oils in the form of capsules, gelcaps, liquids, to improve the cognitive functions of patients suffering from Alzheimer's or minor memory disorders, to contribute to the development and proper functioning of the brain; Natural products for human use, namely, type II collagen containing chondroitin and hyaluronic acid in the form of capsules for maintaining connective tissues, namely, joints and skin; Natural products for human use, namely, gel in the form of ampoules to combat fatigue |
Trademark Filing History
| Description | Date | Proceeding Number |
|---|---|---|
| REGISTERED-PRINCIPAL REGISTER | 2019-12-17 | |
| NOTICE OF ACCEPTANCE OF STATEMENT OF USE E-MAILED | 2019-11-14 | |
| ALLOWED PRINCIPAL REGISTER - SOU ACCEPTED | 2019-11-13 | |
| STATEMENT OF USE PROCESSING COMPLETE | 2019-11-12 | 70565 |
| DIVISIONAL PROCESSING COMPLETE | 2019-11-12 | |
| CASE ASSIGNED TO INTENT TO USE PARALEGAL | 2019-11-12 | 70565 |
| USE AMENDMENT FILED | 2019-10-29 | 70565 |
| TEAS STATEMENT OF USE RECEIVED | 2019-10-29 | |
| TEAS REQUEST TO DIVIDE RECEIVED | 2019-10-29 | |
| DIVISIONAL REQUEST RECEIVED | 2019-10-29 | |
| NOTICE OF APPROVAL OF EXTENSION REQUEST E-MAILED | 2019-09-27 | |
| TEAS EXTENSION RECEIVED | 2019-09-25 | |
| EXTENSION 1 GRANTED | 2019-09-25 | 98765 |
| EXTENSION 1 FILED | 2019-09-25 | 98765 |
| NOA E-MAILED - SOU REQUIRED FROM APPLICANT | 2019-03-26 | |
| PUBLISHED FOR OPPOSITION | 2019-01-29 | |
| OFFICIAL GAZETTE PUBLICATION CONFIRMATION E-MAILED | 2019-01-29 | |
| NOTIFICATION OF NOTICE OF PUBLICATION E-MAILED | 2019-01-09 | |
| TEAS/EMAIL CORRESPONDENCE ENTERED | 2018-12-19 | 68171 |
| CORRESPONDENCE RECEIVED IN LAW OFFICE | 2018-12-19 | 68171 |
| ASSIGNED TO LIE | 2018-12-19 | 68171 |
| APPROVED FOR PUB - PRINCIPAL REGISTER | 2018-12-19 | |
| ASSIGNED TO LIE | 2018-12-14 | 68552 |
| TEAS RESPONSE TO OFFICE ACTION RECEIVED | 2018-12-10 | |
| NOTIFICATION OF NON-FINAL ACTION E-MAILED | 2018-10-30 | 6325 |
| NON-FINAL ACTION WRITTEN | 2018-10-30 | 81848 |
| NON-FINAL ACTION E-MAILED | 2018-10-30 | 6325 |
| ASSIGNED TO EXAMINER | 2018-10-23 | 81848 |
| TEAS VOLUNTARY AMENDMENT RECEIVED | 2018-07-23 | |
| TEAS AMENDMENT ENTERED BEFORE ATTORNEY ASSIGNED | 2018-07-23 | 88889 |
| NEW APPLICATION OFFICE SUPPLIED DATA ENTERED IN TRAM | 2018-07-09 |
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.