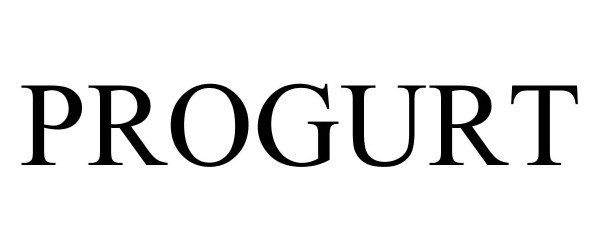PROGURT
Marilyn Beson
Mark For: PROGURT® trademark registration is intended to cover the categories of
| Research |
|
| Serial Number | 88429799 |
| Registration Number | 6182704 |
| Mark Literal Elements | PROGURT |
| Mark Drawing Type | 4 - STANDARD CHARACTER MARK |
| Mark Type | TRADEMARK |
| Register | PRINCIPAL |
| Current Location | TMEG LAW OFFICE 101 - EXAMINING ATTORNEY ASSIGNED 2020-02-07 |
| Basis | 1(a) 44(d) |
| Class Status | ACTIVE |
| Primary US Classes |
|
| Primary International Class |
|
| Filed Use | No |
| Current Use | Yes |
| Intent To Use | No |
| Filed ITU | No |
| 44D Filed | Yes |
| 44E Current | No |
| 66A Current | No |
| Current Basis | No |
| No Basis | No |
| Attorney Name | Susan L. Stuart |
| Attorney Docket Number | HENR-2019001 |
| Law Office Assigned | L10 |
| Employee Name | DOMBROW, COLLEEN M |
Timeline
| 2005-05-00 | Date of First Use |
| 2005-05-01 | Date of First Use |
| 2005-05-01 | Date of Use In Commerce |
| 2019-05-14 | Application Filed |
| 2019-08-07 | Location: TMEG LAW OFFICE 101 - EXAMINING ATTORNEY ASSIGNED |
| 2019-08-07 | Status: Live/Pending |
| 2019-08-07 | Transaction Date |
| 2020-02-07 | Location: TMEG LAW OFFICE 101 - EXAMINING ATTORNEY ASSIGNED |
| 2020-02-07 | Status: An Office action suspending further action on the application has been sent (issued) to the applicant. To view all documents in |
| 2020-10-27 | Trademark Registered |
Trademark Parties (Applicants & Owners)
| Party: | |
| Address | 16 Sunnyridge Road Arcadia, NSW AUSTRALIA 2159 |
| Legal Entity Type | Individual |
Documents
| JPEG | 2019-05-14 | |
| MULTI | 2019-05-14 | |
| MULTI | 2019-05-29 | |
| JPEG | 2019-05-29 | |
| MULTI | 2019-06-05 | |
| XML | 2019-08-07 | |
| XML | 2019-08-07 | |
| XML | 2019-08-07 | |
| XML | 2020-02-05 | |
| MULTI | 2020-02-07 | |
| XML | 2020-02-07 | |
| MULTI | 2020-06-11 | |
| MULTI | 2020-06-16 | |
| XML | 2020-07-02 | |
| XML | 2020-07-02 | |
| MULTI | 2020-07-03 | |
| MULTI | 2020-07-03 | |
| XML | 2020-07-22 | |
| XML | 2020-07-22 | |
| XML | 2020-08-11 | |
| 2020-10-27 | ||
| MULTI | 2021-01-12 | |
| 2021-05-04 |
Attorney of Record
Good, Services, and Codes
IC 005. US 006 018 044 046 051 052. G & S: Pharmaceutical preparations for digestive health, cardiovascular health, immune function and blood sugar management; probiotic preparations for medical use; probiotic substances, namely, probiotic supplements; probiotic bacterial preparations, namely, probiotic supplements; probiotic cultures, namely, bacteriological culture mediums; cultures for medical use, namely, bacteriological cultures for medical purposes; bacterial cultures, namely, bacteriological culture mediums; bacteriological cultural media, namely, media for bacteriological cultures; bacterial preparations for medical use; biological preparations for medical use, namely, biological preparations for the treatment of digestive disorders, cardiovascular health, immune function and blood sugar management; bacterial preparations for nutritional purposes for medical use; biological preparations for nutritional purposes, namely, biological preparations for the treatment of digestive health, cardiovascular health, immune function and blood sugar management; cultures of micro-organisms for medical or veterinary use; dietetic foods adapted for medical use; dietetic substances adapted for medical use, namely, dietetic beverages adapted for medical use; meal replacement beverages adapted for medical use; food for babies; food for infants; milk powder for babies; milk powder for infants; dietary supplements; vitamins; vitamin formulations, namely, vitamins for mixing with yogurt; mineral supplements; mineral supplements for foodstuffs; nutritional food additives for medical purposes in the nature of natural food extracts derived from human breast milk; nutritional supplements; bacteriological preparations for medical use; albuminous preparations for medical purposes, namely, albumin dietary supplements; homogenized food adapted for medical purposes; baby milk powder; lacteal flour for babies; milk sugar for medical purposes; lactose for medical purposes; malted milk beverages for medical purposes; albuminous foodstuffs in the nature of nutritional supplements for medical purposes; mineral food supplements. FIRST USE: 20050500. FIRST USE IN COMMERCE: 20050500
| International Codes: | 5 |
| U.S. Codes: | 006,018,044,046,051,052 |
| Type Code | Type |
|---|---|
| GS0051 | Pharmaceuticals; pharmaceutical preparations; probiotic preparations; probiotic substances; probiotic bacterial preparations; probiotic cultures; bacterial cultures; cultures for medical use; bacteriological cultural media; lactic cultures for food industry; bacterial preparations for medical use; biological preparations for medical use; bacterial preparations for nutritional purposes; biological preparations for nutritional purposes; cultures of micro-organisms; dietetic foods adapted for medical use; dietetic substances adapted for medical use; dietetic beverages adapted for medical use; meal replacements adapted for medical use; food for babies; food for infants; milk powder for babies; milk powder for infants; dietary supplements; vitamins; vitamin formulations; mineral supplements; mineral supplements for foodstuffs; nutritional additives; nutritional supplements; nutritional cultures; bacteriological preparations for medical use; albuminous preparations for medical purposes; food preparations adapted for medical purposes; lacteal flour; milk sugar; lactose; baby milk powder; lacteal flour for babies; malted milk beverages for medical purposes; albuminous milk; albuminous foodstuffs for medical purposes; nutritional additives for medical purposes; mineral food supplements |
| GS0051 | Pharmaceutical preparations for digestive health, cardiovascular health, immune function and blood sugar management; probiotic preparations for medical use; probiotic substances, namely, probiotic supplements; probiotic bacterial preparations, namely, probiotic supplements; probiotic cultures, namely, bacteriological culture mediums; cultures for medical use, namely, bacteriological cultures for medical purposes; bacterial cultures, namely, bacteriological culture mediums; bacteriological cultural media, namely, media for bacteriological cultures; bacterial preparations for medical use; biological preparations for medical use, namely, biological preparations for the treatment of digestive disorders, cardiovascular health, immune function and blood sugar management; bacterial preparations for nutritional purposes for medical use; biological preparations for nutritional purposes, namely, biological preparations for the treatment of digestive health, cardiovascular health, immune function and blood sugar management; cultures of micro-organisms for medical or veterinary use; dietetic foods adapted for medical use; dietetic substances adapted for medical use, namely, dietetic beverages adapted for medical use; meal replacement beverages adapted for medical use; food for babies; food for infants; milk powder for babies; milk powder for infants; dietary supplements; vitamins; vitamin formulations, namely, vitamins for mixing with yogurt; mineral supplements; mineral supplements for foodstuffs; nutritional food additives for medical purposes in the nature of natural food extracts derived from human breast milk; nutritional supplements; bacteriological preparations for medical use; albuminous preparations for medical purposes, namely, albumin dietary supplements; homogenized food adapted for medical purposes; baby milk powder; lacteal flour for babies; milk sugar for medical purposes; lactose for medical purposes; malted milk beverages for medical purposes; albuminous foodstuffs in the nature of nutritional supplements for medical purposes; mineral food supplements |
Trademark Filing History
| Description | Date | Proceeding Number |
|---|---|---|
| CORRECTION UNDER SECTION 7 PROCESSED | 2021-03-29 | 76874 |
| CASE ASSIGNED TO POST REGISTRATION PARALEGAL | 2021-02-02 | 76874 |
| TEAS SECTION 7 REQUEST RECEIVED | 2021-01-12 | |
| REGISTERED-PRINCIPAL REGISTER | 2020-10-27 | |
| PUBLISHED FOR OPPOSITION | 2020-08-11 | |
| OFFICIAL GAZETTE PUBLICATION CONFIRMATION E-MAILED | 2020-08-11 | |
| NOTIFICATION OF NOTICE OF PUBLICATION E-MAILED | 2020-07-22 | |
| NOTIFICATION OF EXAMINERS AMENDMENT E-MAILED | 2020-07-02 | 6328 |
| EXAMINERS AMENDMENT E-MAILED | 2020-07-02 | 6328 |
| EXAMINERS AMENDMENT -WRITTEN | 2020-07-02 | 81089 |
| EXAMINER'S AMENDMENT ENTERED | 2020-07-02 | 88888 |
| APPROVED FOR PUB - PRINCIPAL REGISTER | 2020-07-02 | |
| TEAS/EMAIL CORRESPONDENCE ENTERED | 2020-06-13 | 70884 |
| CORRESPONDENCE RECEIVED IN LAW OFFICE | 2020-06-13 | 70884 |
| TEAS RESPONSE TO SUSPENSION INQUIRY RECEIVED | 2020-06-11 | |
| SUSPENSION LETTER WRITTEN | 2020-02-07 | 81089 |
| NOTIFICATION OF LETTER OF SUSPENSION E-MAILED | 2020-02-07 | 6332 |
| LETTER OF SUSPENSION E-MAILED | 2020-02-07 | 6332 |
| TEAS/EMAIL CORRESPONDENCE ENTERED | 2020-02-06 | 88889 |
| TEAS RESPONSE TO OFFICE ACTION RECEIVED | 2020-02-05 | |
| CORRESPONDENCE RECEIVED IN LAW OFFICE | 2020-02-05 | 88889 |
| NOTIFICATION OF NON-FINAL ACTION E-MAILED | 2019-08-07 | 6325 |
| NON-FINAL ACTION WRITTEN | 2019-08-07 | 81089 |
| NON-FINAL ACTION E-MAILED | 2019-08-07 | 6325 |
| ASSIGNED TO EXAMINER | 2019-07-30 | 81089 |
| TEAS/EMAIL CORRESPONDENCE ENTERED | 2019-06-04 | 70884 |
| CORRESPONDENCE RECEIVED IN LAW OFFICE | 2019-06-04 | 70884 |
| ASSIGNED TO LIE | 2019-06-03 | 70884 |
| NEW APPLICATION OFFICE SUPPLIED DATA ENTERED IN TRAM | 2019-05-30 | |
| TEAS VOLUNTARY AMENDMENT RECEIVED | 2019-05-29 | |
| NEW APPLICATION ENTERED IN TRAM | 2019-05-17 |
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.