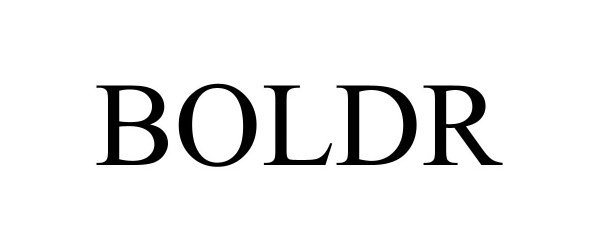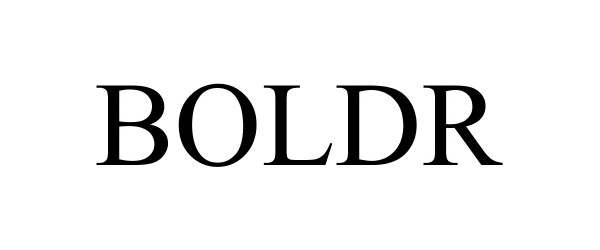BOLDR
Boldr Beverages Llc
Mark For: BOLDR™ trademark registration is intended to cover the categories of
| Research |
|
| Serial Number | 88169850 |
| Mark Literal Elements | BOLDR |
| Mark Drawing Type | 4 - STANDARD CHARACTER MARK |
| Mark Type | TRADEMARK |
| Register | PRINCIPAL |
| Current Location | TMO LAW OFFICE 126- EXAMINING ATTORNEY ASSIGNED 2020-03-24 |
| Basis | 1(b) |
| Class Status | ACTIVE |
| Primary US Classes |
|
| Primary International Class |
|
| Filed Use | No |
| Current Use | No |
| Intent To Use | Yes |
| Filed ITU | Yes |
| 44D Filed | No |
| 44E Current | No |
| 66A Current | No |
| Current Basis | No |
| No Basis | No |
| Attorney Name | Susan L. Heller |
| Attorney Docket Number | 182889.01000 |
| Law Office Assigned | N80 |
| Employee Name | RIBAUDO, MICHELLE RACH |
Timeline
| 2018-10-25 | Application Filed |
| 2019-02-09 | Location: TMO LAW OFFICE 126- EXAMINING ATTORNEY ASSIGNED |
| 2019-02-09 | Status: Live/Pending |
| 2019-04-25 | Transaction Date |
| 2020-03-24 | Location: TMO LAW OFFICE 126- EXAMINING ATTORNEY ASSIGNED |
| 2020-03-24 | Status: Abandoned because the applicant failed to respond or filed a late response to an Office action. To view all documents in this fi |
Trademark Parties (Applicants & Owners)
| Party: | |
| Address | 5434 RUSSELL AVE. LOS ANGELES, CALIFORNIA UNITED STATES 90027 |
| Legal Entity Type | Limited Liability Company |
| Legal Entity State | CALIFORNIA |
| Party: | |
| Address | 5434 Russell Ave Los Angeles CA US 90027 |
| Legal Entity Type | |
| Legal Entity State | US |
*multiple parties listed, check assignment documents for ownership information
Documents
| JPEG | 2018-10-25 | |
| MULTI | 2018-10-25 | |
| APPLICATION/XML | 2019-02-01 | |
| MULTI | 2019-02-09 | |
| XML | 2019-04-24 | |
| MULTI | 2019-08-02 | |
| APPLICATION/XML,IMAGE/JPEG | 2019-08-06 | |
| MULTI | 2019-09-11 | |
| APPLICATION/XML | 2020-03-24 |
Attorney of Record
Good, Services, and Codes
(ABANDONED)
IC 005. US 006 018 044 046 051 052. G & S: Non-Medical Nutriceutical products solely in nature of powders for use as a dietary supplement, expressly excluding any liquid beverage, namely, nutritionally fortified beverages; Dietary supplements, expressly excluding nutritionally fortified beverages; Dietary supplements, namely, protein-based, nutrient-dense snack bars; powdered nutritional supplement drink mix containing one or more of the following ingredients, soy, oat oil, creatine, soy oil, dextrose, protein, non-fat milk, milk protein, or whey protein concentrate; dietary supplements; nutritional supplements for weight loss purposes in the nature of protein-based drinks, protein-fortified meal replacement bars, protein-fortified energy bars, and protein supplements formed and packaged as bars; protein supplements; ready to drink nutritional supplement beverages for use as a meal replacement beverages adapted for medical purposes; nutritionally fortified beverages for medical purposes; nutritional shakes for use as a meal substitute for medical purposes; dietary and nutritional supplements containing protein; food supplements; nutritional supplements; dietary supplements in the nature of powdered drink mixes and weight loss powders; dietary supplement drinks, namely, nutritional supplements for weight loss, health and bodybuilding; dietary and nutritional food supplements and powdered drink mixes containing soluble and insoluble fiber; dietary and nutritional supplement for weight loss, diet, health, sports nutrition, body building, muscle gain and training; nutritional supplements in the form of non-fruit meal replacement bars and powders for medical purposes; nutritional supplements in the form of non-chocolate meal replacement bars and powders for medical purposes; chocolate-based meal replacement bars and powders for weight loss purposes; nutritional supplement meal replacement bars and powders for boosting energy; Dietary and nutritional supplements, in gel, bar, powder and liquid form; vitamin supplements; mineral supplements; vitamins; vitamin, mineral, dietary and nutritional supplements
| International Codes: | 5 |
| U.S. Codes: | 006,018,044,046,051,052 |
| Type Code | Type |
|---|---|
| GS0051 | Non-Medical Nutriceutical products solely in nature of powders, expressly excluding any liquid beverage, namely, nutritionally fortified beverages; Dietary supplements, expressly excluding nutritionally fortified beverages; Dietary supplements, namely, protein-based, nutrient-dense snack bars; powdered nutritional supplement drink mix containing one or more of the following ingredients, soy, oat oil, creatine, soy oil, dextrose, protein, non-fat milk, milk protein, or whey protein concentrate; dietary supplements; nutritional supplements in the nature of protein-based drinks, protein-fortified meal replacement bars, protein-fortified snack bars, and protein-based food bars; protein supplements; ready to drink nutritional beverages for use as a meal substitute for medical purposes; nutritionally fortified beverages for medical purposes; nutritional shakes for use as a meal substitute for medical purposes; dietary and nutritional supplements containing protein; food supplements; nutritional supplements; dietary supplements in the nature of powdered drink mixes and weight loss powders; dietary supplement drinks, namely, nutritional supplements for weight loss, health and bodybuilding; dietary and nutritional food supplements and powdered drink mixes containing soluble and insoluble fiber; dietary and nutritional supplement for weight loss, diet, health, sports nutrition, body building, muscle gain and training; nutritional supplements in the form of non-fruit meal replacement bars and powders and nutritional supplements in the form of non-chocolate meal replacement bars and powders; chocolate-based meal replacement bars and powders for weight loss purposes; nutritional supplement meal replacement bars and powders for boosting energy; Dietary and nutritional supplements, in gel, bar, powder and liquid form; vitamin supplements; mineral supplements; vitamins; vitamin, mineral, dietary and nutritional supplements |
Trademark Filing History
| Description | Date | Proceeding Number |
|---|---|---|
| ABANDONMENT NOTICE E-MAILED - FAILURE TO RESPOND | 2020-03-24 | |
| ABANDONMENT - FAILURE TO RESPOND OR LATE RESPONSE | 2020-03-24 | |
| NOTIFICATION OF FINAL REFUSAL EMAILED | 2019-09-11 | |
| FINAL REFUSAL WRITTEN | 2019-09-11 | 93984 |
| FINAL REFUSAL E-MAILED | 2019-09-11 | |
| TEAS/EMAIL CORRESPONDENCE ENTERED | 2019-08-03 | 88889 |
| TEAS RESPONSE TO OFFICE ACTION RECEIVED | 2019-08-02 | |
| CORRESPONDENCE RECEIVED IN LAW OFFICE | 2019-08-02 | 88889 |
| AUTOMATIC UPDATE OF ASSIGNMENT OF OWNERSHIP | 2019-07-03 | |
| TEAS REVOKE/APP/CHANGE ADDR OF ATTY/DOM REP RECEIVED | 2019-04-24 | |
| ATTORNEY/DOM.REP.REVOKED AND/OR APPOINTED | 2019-04-24 | |
| NOTIFICATION OF NON-FINAL ACTION E-MAILED | 2019-02-09 | 6325 |
| NON-FINAL ACTION WRITTEN | 2019-02-09 | 93984 |
| NON-FINAL ACTION E-MAILED | 2019-02-09 | 6325 |
| ASSIGNED TO EXAMINER | 2019-02-01 | 93984 |
| NEW APPLICATION OFFICE SUPPLIED DATA ENTERED IN TRAM | 2018-11-08 | |
| NEW APPLICATION ENTERED IN TRAM | 2018-10-29 |
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.