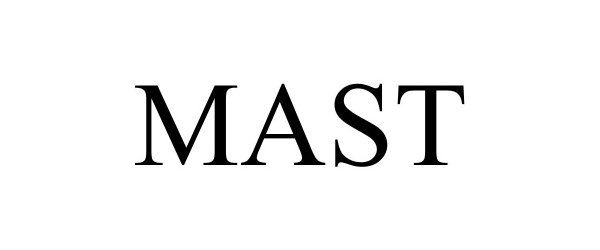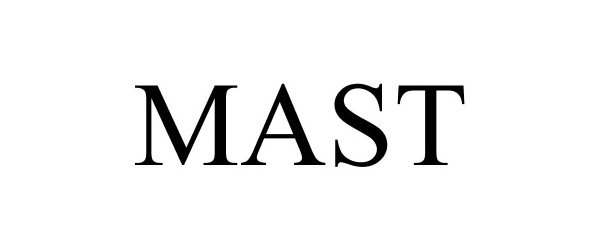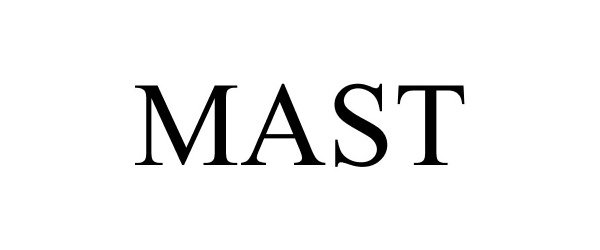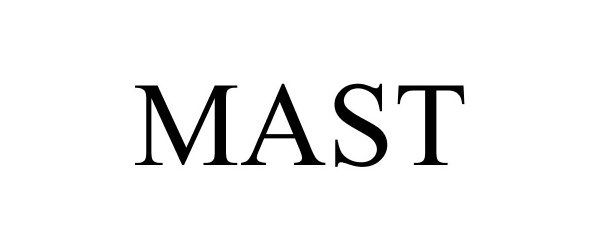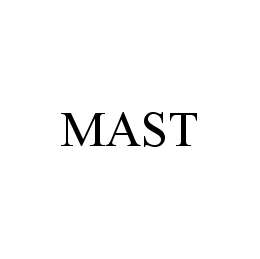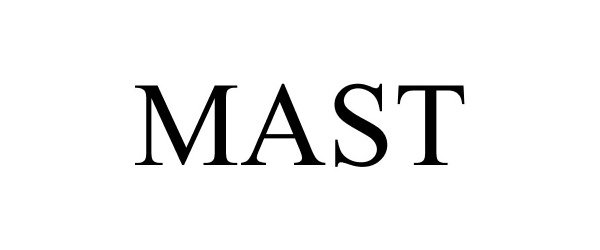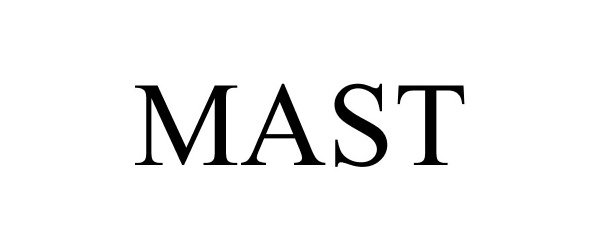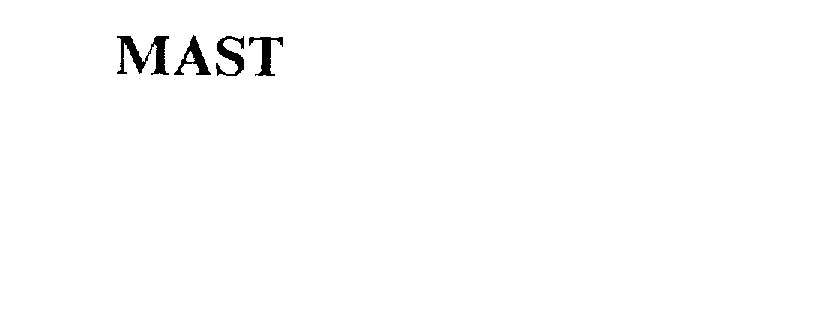MAST
Mast Group Limited
| Research |
|
| Serial Number | 79209271 |
| Registration Number | 6082227 |
| Mark Drawing Code | 4000: Illustration: Drawing with word(s)/letter(s)/number(s) in Block form |
| Domestic Representative | Jeffrey A. Smith |
| Attorney Name | Jeffrey A. Smith |
| Attorney Docket Number | THMPS-0046-T |
| Law Office Assigned | L40 |
| Employee Name | SHANOSKI, JOANNA MARIE |
Timeline
| 2016-11-25 | Application Filed |
| 2019-08-30 | Location: TMEG LAW OFFICE 104 - EXAMINING ATTORNEY ASSIGNED |
| 2019-08-30 | Status: Live/Pending |
| 2019-11-06 | Transaction Date |
| 2020-06-23 | Trademark Registered |
Trademark Applicants & Owners
| Owner: | |
| Address | Mast House, Derby Road Bootle Merseyside L20 1EA GB |
| Legal Entity Type | Private Limited Company |
| Legal Entity State | GB |
Documents
Click the blue "Refresh" button to load certificates, specimines, application, and other documents.
Attorney of Record
Good, Services, and Codes
| International Codes: | 1 |
| U.S. Codes: | 001,005,006,010,026,046 |
| International Codes: | 5 |
| U.S. Codes: | 006,018,044,046,051,052 |
| International Codes: | 9 |
| U.S. Codes: | 021,023,026,036,038 |
| Type Code | Type |
|---|---|
| GS0011 | Chemicals used in industry and science; reagents for scientific and research use and clinical and medical laboratory use; chemical reagents for scientific and research use and clinical and medical laboratory use; molecular reagents for scientific and research use and clinical and medical laboratory use; biological reagents for scientific and research use and clinical and medical laboratory use; microbiological reagents for scientific and research use and clinical and medical laboratory use; antibiotic susceptibility reagents for scientific and research use and clinical and medical laboratory use; cultivation and growth media for the cultivation and growth of bacteria, fungi and yeast from clinical specimens or reference strains use in laboratories, including near patient testing facilities, for diagnostic research purposes; chemical, molecular, biological and microbiological reagents for scientific and research use and clinical and medical laboratory use in kit form; chemical reagents in kit form for conducting enzyme-linked immunosorbent assays (ELISA) for scientific and research use and clinical and medical laboratory use; chemical reagents in kit form for conducting thermal amplification assays for scientific and research use and clinical and medical laboratory use; chemical reagents in kit form for thermal amplification or detection of nucleic targets for scientific and research use and clinical and medical laboratory use; assay preparations for scientific and research use and clinical and ; tests for detecting antibodies, other than for medical or veterinary use comprising immunofluorescence tests, enzyme immunoassays and western blot products for use for scientific and research use and clinical or medical laboratory use Chemical media supplements and test kits containing these goods for use in laboratories, including near patient testing facilities, for diagnostic purposes including identification and susceptibility testing for scientific and research use and clinical or medical laboratory use; chemicals for use in the cryopreservation of organisms for scientific use, research use and clinical or medical laboratory use; chemical reagents for molecular biology scientific research and for use in clinical and medical laboratories; bacterial culture media for use in laboratories, including near patient testing facilities for diagnostic purposes for clinical or medical laboratory use; reagents for use in scientific apparatus for antibiotic susceptibility analysis; nutrients for use in scientific apparatus for microbiological analysis; Biochemical reagents for the identification and detection of bacteria; biochemical reagents for clinical or medical laboratory use; bacterial agglutinating antisera for clinical or medical laboratory use; Laboratory chemicals, namely, stained febrile antigens; Laboratory chemicals, namely, latex for agglutination tests |
| GS0011 | Chemicals used in industry and science; reagents for scientific and research use and clinical and medical laboratory use; chemical reagents for scientific and research use and clinical and medical laboratory use; molecular reagents for scientific and research use and clinical and medical laboratory use; biological reagents for scientific and research use and clinical and medical laboratory use; microbiological reagents for scientific and research use and clinical and medical laboratory use; antibiotic susceptibility reagents for scientific and research use and clinical and medical laboratory use; cell growth media for the cultivation and growth of bacteria, fungi and yeast cells from clinical specimens or reference strains for use in laboratories, near patient testing laboratory facilities, and for diagnostic research purposes; Kits comprising chemical, molecular, biological and microbiological reagents for scientific, research, and clinical and medical laboratory use; Kits comprising chemical reagents for conducting enzyme-linked immunosorbent assays (ELISA) for scientific and research use and clinical and medical laboratory use; Kits comprising chemical reagents for conducting thermal amplification assays for scientific and research use, and clinical and medical laboratory use; Kits comprising chemical reagents for thermal amplification or detection of nucleic targets for scientific and research use, and clinical and medical laboratory use; assay preparations for scientific and research use and clinical laboratory use; Test kits for detecting antibodies, other than for medical or veterinary use, comprising immunofluorescence dyes, enzyme immunoassay reagents, and Western blot electrophoreses and protein gel reagents, all for use for scientific and research use, and clinical or medical laboratory use; Chemical cell culture media supplements and test kits comprised of chemical cell culture media supplements for use in laboratories, near patient testing facilities for research purposes related to identification and susceptibility testing for scientific and research use, and clinical or medical laboratory use; chemicals for use in the cryopreservation of organisms for scientific use, research use and clinical or medical laboratory use; chemical reagents for molecular biology scientific research and for use in clinical and medical laboratories; cell culture media for use in laboratories, near patient testing facilities for diagnostic research purposes, and clinical or medical laboratory use; reagents for use in scientific apparatus for antibiotic susceptibility analysis; nutrients for use in scientific apparatus for microbiological analysis; Biochemical reagents for the identification and detection of bacteria for non-medical purposes; biochemical reagents for clinical or medical laboratory use; bacterial agglutinating antisera for clinical or medical laboratory use; Laboratory chemicals, namely, stained febrile antigens; Laboratory chemicals, namely, latex for agglutination tests |
Trademark Filing History
| Description | Date | Event Coding |
|---|---|---|
| SN ASSIGNED FOR SECT 66A APPL FROM IB | 2017-06-08 | 1 REPR M: |
| NEW APPLICATION OFFICE SUPPLIED DATA ENTERED IN TRAM | 2017-06-09 | 2 NWOS I:Incoming Correspondence |
| ASSIGNED TO EXAMINER | 2017-06-09 | 3 DOCK D:Assigned to Examiner |
| APPLICATION FILING RECEIPT MAILED | 2017-06-13 | 4 MAFR O:Outgoing Correspondence |
| NON-FINAL ACTION WRITTEN | 2017-06-27 | 5 CNRT R:Renewal |
| NON-FINAL ACTION (IB REFUSAL) PREPARED FOR REVIEW | 2017-06-28 | 6 RFCR E:E-Mail |
| REFUSAL PROCESSED BY MPU | 2017-07-08 | 7 RFRR P: |
| NON-FINAL ACTION MAILED - REFUSAL SENT TO IB | 2017-07-08 | 8 RFCS P: |
| REFUSAL PROCESSED BY IB | 2017-07-28 | 9 RFNT P: |
| TEAS REVOKE/APP/CHANGE ADDR OF ATTY/DOM REP RECEIVED | 2018-01-04 | 10 REAP I:Incoming Correspondence |
| ATTORNEY/DOM.REP.REVOKED AND/OR APPOINTED | 2018-01-04 | 11 ARAA I:Incoming Correspondence |
| TEAS RESPONSE TO OFFICE ACTION RECEIVED | 2018-01-08 | 12 TROA I:Incoming Correspondence |
| ASSIGNED TO LIE | 2018-01-23 | 13 ALIE A:Allowance for Publication |
| CORRESPONDENCE RECEIVED IN LAW OFFICE | 2018-01-24 | 14 CRFA I:Incoming Correspondence |
| TEAS/EMAIL CORRESPONDENCE ENTERED | 2018-01-24 | 15 TEME I:Incoming Correspondence |
| SUSPENSION LETTER WRITTEN | 2018-02-12 | 16 CNSL R:Renewal |
| LETTER OF SUSPENSION E-MAILED | 2018-02-12 | 17 GNSL S: |
| NOTIFICATION OF LETTER OF SUSPENSION E-MAILED | 2018-02-12 | 18 GNS3 O:Outgoing Correspondence |
| REPORT COMPLETED SUSPENSION CHECK CASE STILL SUSPENDED | 2018-08-12 | 19 RCSC S: |
| NOTIFICATION OF POSSIBLE OPPOSITION CREATED, TO BE SENT TO IB | 2018-11-14 | 20 OPNR P: |
| NOTIFICATION OF POSSIBLE OPPOSITION SENT TO IB | 2018-11-14 | 21 OPNS P: |
| NOTIFICATION OF POSSIBLE OPPOSITION - PROCESSED BY IB | 2018-11-30 | 22 OPNX P: |
| REPORT COMPLETED SUSPENSION CHECK CASE STILL SUSPENDED | 2019-02-12 | 23 RCSC S: |
| LIE CHECKED SUSP - TO ATTY FOR ACTION | 2019-08-15 | 24 RCCK S: |
| NON-FINAL ACTION WRITTEN | 2019-08-30 | 25 CNRT R:Renewal |
| NON-FINAL ACTION E-MAILED | 2019-08-30 | 26 GNRT O:Outgoing Correspondence |
| NOTIFICATION OF NON-FINAL ACTION E-MAILED | 2019-08-30 | 27 GNRN O:Outgoing Correspondence |
| IRREGULARITY ON REFUSAL FROM IB | 2019-10-23 | 28 IRRF M: |
| IRREGULARITY FROM IB EXAMINED BY MPU | 2019-11-06 | 29 IRRH M: |
| TEAS RESPONSE TO OFFICE ACTION RECEIVED | 2020-02-27 | 30 TROA I:Incoming Correspondence |
| CORRESPONDENCE RECEIVED IN LAW OFFICE | 2020-03-02 | 31 CRFA I:Incoming Correspondence |
| TEAS/EMAIL CORRESPONDENCE ENTERED | 2020-03-02 | 32 TEME I:Incoming Correspondence |
| APPROVED FOR PUB - PRINCIPAL REGISTER | 2020-03-03 | 33 CNSA O:Outgoing Correspondence |
| NOTIFICATION OF NOTICE OF PUBLICATION E-MAILED | 2020-03-18 | 34 NONP E:E-Mail |
| NOTICE OF START OF OPPOSITION PERIOD CREATED, TO BE SENT TO IB | 2020-03-18 | 35 OP2R P: |
| NOTIFICATION OF POSSIBLE OPPOSITION SENT TO IB | 2020-03-18 | 36 OPNS P: |
| PUBLISHED FOR OPPOSITION | 2020-04-07 | 37 PUBO A:Allowance for Publication |
| OFFICIAL GAZETTE PUBLICATION CONFIRMATION E-MAILED | 2020-04-07 | 38 NPUB E:E-Mail |
| REGISTERED-PRINCIPAL REGISTER | 2020-06-23 | 39 R.PR A:Allowance for Publication |
| NOTIFICATION PROCESSED BY IB | 2020-08-14 | 40 GPNX P: |
| FINAL DISPOSITION NOTICE CREATED, TO BE SENT TO IB | 2020-09-23 | 41 FICR P: |
| FINAL DISPOSITION PROCESSED | 2020-10-08 | 42 FIMP P: |
| FINAL DISPOSITION NOTICE SENT TO IB | 2020-10-09 | 43 FICS P: |
| FINAL DECISION TRANSACTION PROCESSED BY IB | 2020-11-01 | 44 FINO P: |
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.