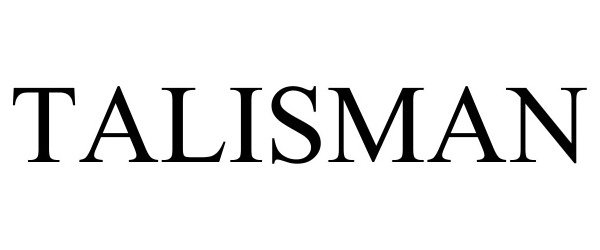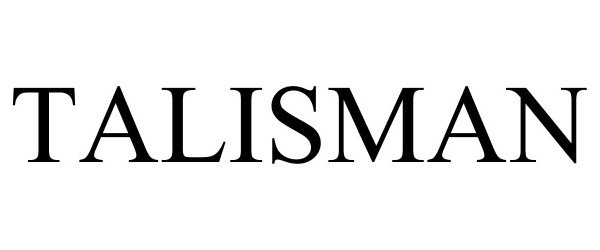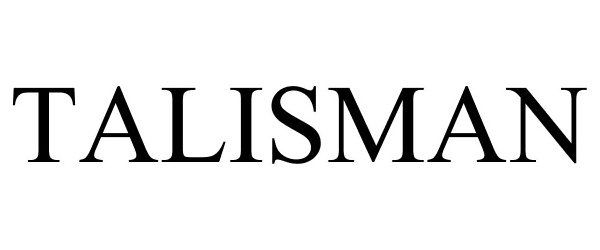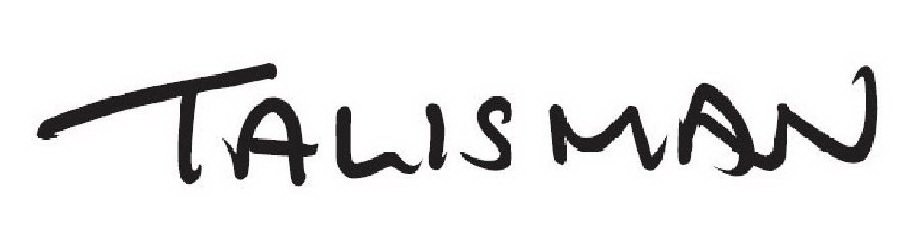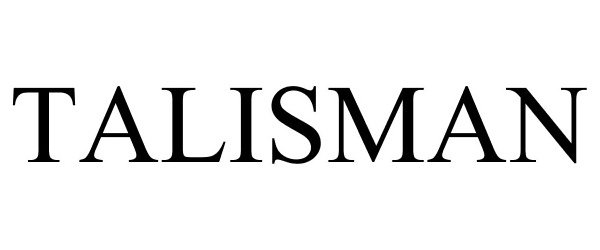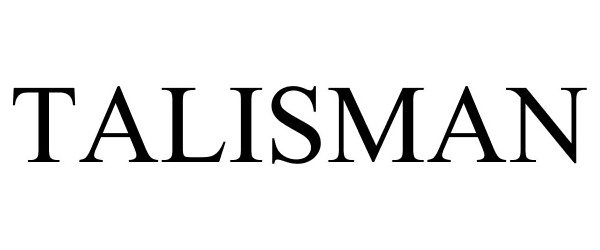TALISMAN
Aventis Pharma Sa

Mark For: TALISMAN® trademark registration is intended to cover the categories of
| Research |
|
| Serial Number | 76483857 |
| Registration Number | 3147095 |
| Mark Literal Elements | TALISMAN |
| Mark Drawing Type | 1 - TYPESET WORD(S) /LETTER(S) /NUMBER(S) |
| Mark Type | SERVICE MARK |
| Standard Character Claim | No |
| Register | PRINCIPAL |
| Current Location | PUBLICATION AND ISSUE SECTION 2006-09-26 |
| Basis | 44(e) |
| Class Status | SECTION 8 - CANCEL |
| Primary US Classes |
|
| Primary International Class |
|
| Filed Use | No |
| Current Use | No |
| Intent To Use | No |
| Filed ITU | Yes |
| 44D Filed | No |
| 44E Current | Yes |
| 66A Current | No |
| Current Basis | No |
| No Basis | No |
| Cancellation Code | 2 |
| Domestic Representative | Finnegan, Henderson, Farabow, Garrett & Dunner, L.L. |
| Attorney Name | B. Brett Heavner |
| Attorney Docket Number | 0888.0553 |
| Law Office Assigned | L70 |
| Employee Name | HOFFMAN, DAVID AARON |
Timeline
| 2003-01-21 | Application Filed |
| 2006-07-04 | Published |
| 2006-07-04 | Published for Opposition |
| 2006-09-26 | Location: PUBLICATION AND ISSUE SECTION |
| 2006-09-26 | Trademark Registered |
| 2013-05-03 | Cancelled |
| 2013-05-03 | Status: Dead/Cancelled |
| 2013-05-03 | Status: Registration cancelled because registrant did not file an acceptable declaration under Section 8. To view all documents in this |
| 2018-07-08 | Transaction Date |
Trademark Parties (Applicants & Owners)
| Party: | |
| Address | 20 AVENUE RAYMOND ARON 92160 ANTONY FRANCE |
| Legal Entity Type | Corporation |
| Legal Entity State | FRANCE |
Documents
| IMAGE/TIFF | 2003-01-21 | |
| IMAGE/TIFF | 2003-01-21 | |
| APPLICATION/XML,IMAGE/JPEG | 2003-07-03 | |
| IMAGE/TIFF | 2003-07-03 | |
| IMAGE/TIFF | 2003-12-30 | |
| APPLICATION/XML | 2004-01-23 | |
| APPLICATION/XML,IMAGE/JPEG | 2005-01-28 | |
| IMAGE/TIFF,IMAGE/JPEG | 2005-07-13 | |
| APPLICATION/XML,IMAGE/JPEG | 2005-07-29 | |
| IMAGE/TIFF | 2005-11-08 | |
| APPLICATION/XML | 2005-12-19 | |
| APPLICATION/XML | 2006-05-02 | |
| APPLICATION/XML,IMAGE/TIFF | 2006-05-06 | |
| APPLICATION/XML | 2006-06-14 | |
| IMAGE/TIFF | 2006-09-26 |
Attorney of Record
Good, Services, and Codes
(CANCELLED)
IC 042. US 100 101. G & S: Scientific research and development; medical research; research and development relating to pharmaceutical preparations; clinical trial services namely, design and implementation of clinical trials; medical and scientific research, namely, conducting clinical trials; collection of data derived from clinical trials; testing of pharmaceutical preparations for the treatment of cardiovascular, arterial and arteriovenous circulatory disorders, peripheral arterial disease, and other related conditions; advisory and consultancy services relating to all the aforesaid services(CANCELLED)
IC 044. US 100 101. G & S: Medical services, namely, the treatment of cardiovascular arterial and arteriovenous circulatory disorders, peripheral arterial disease, cancer, central nervous system diseases and disorders, neurological disorders, musculoskeletal disorders, allergies, hypertension, stroke, inflammation, and inflammatory diseases; medical diagnostic services, namely, conducting genetic testing to identify medical conditions and diseases; compilation and preparation of medical reports; advisory and consultancy services relating to cardiovascular arterial and arteriovenous circulatory disorders, peripheral arterial disease, and other related conditions
| International Codes: | 42 |
| U.S. Codes: | 100,101 |
| International Codes: | 44 |
| U.S. Codes: | 100,101 |
| Type Code | Type |
|---|---|
| GS0421 | Scientific research and development; medical research; research and development relating to pharmaceutical preparations; clinical trial services namely, design and implementation of clinical trials; medical and scientific research, namely, conducting clinical trials; collection of data derived from clinical trials; testing of pharmaceutical preparations for the treatment of cardiovascular, arterial and arteriovenous circulatory disorders, peripheral arterial disease, and other related conditions; advisory and consultancy services relating to all the aforesaid services |
Trademark Filing History
| Description | Date | Proceeding Number |
|---|---|---|
| CANCELLED SEC. 8 (6-YR) | 2013-05-03 | |
| AUTOMATIC UPDATE OF ASSIGNMENT OF OWNERSHIP | 2010-12-09 | |
| REGISTERED-PRINCIPAL REGISTER | 2006-09-26 | |
| EXPARTE APPEAL TERMINATED | 2006-09-26 | 483857 |
| PUBLISHED FOR OPPOSITION | 2006-07-04 | |
| NOTICE OF PUBLICATION | 2006-06-14 | |
| LAW OFFICE PUBLICATION REVIEW COMPLETED | 2006-05-12 | 74221 |
| ASSIGNED TO LIE | 2006-05-12 | 74221 |
| APPROVED FOR PUB - PRINCIPAL REGISTER | 2006-05-05 | |
| EXAMINERS AMENDMENT MAILED | 2006-05-03 | |
| EXAMINERS AMENDMENT -WRITTEN | 2006-05-02 | 81855 |
| EXAMINER'S AMENDMENT ENTERED | 2006-05-02 | 88888 |
| JURISDICTION RESTORED TO EXAMINING ATTORNEY | 2006-04-24 | 483857 |
| EX PARTE APPEAL-INSTITUTED | 2006-01-31 | 483857 |
| ACTION DENYING REQ FOR RECON MAILED | 2005-12-19 | |
| ACTION CONTINUING FINAL - COMPLETED | 2005-12-19 | 81855 |
| ASSIGNED TO EXAMINER | 2005-11-29 | 81855 |
| AMENDMENT FROM APPLICANT ENTERED | 2005-11-21 | 68552 |
| PAPER RECEIVED | 2005-11-08 | |
| CORRESPONDENCE RECEIVED IN LAW OFFICE | 2005-11-08 | 68552 |
| FINAL REFUSAL WRITTEN | 2005-07-29 | 72505 |
| FINAL REFUSAL MAILED | 2005-07-29 | |
| AMENDMENT FROM APPLICANT ENTERED | 2005-07-20 | 73793 |
| PAPER RECEIVED | 2005-07-13 | |
| CORRESPONDENCE RECEIVED IN LAW OFFICE | 2005-07-13 | 73793 |
| NON-FINAL ACTION WRITTEN | 2005-01-28 | 72505 |
| NON-FINAL ACTION MAILED | 2005-01-28 | |
| REPORT COMPLETED SUSPENSION CHECK CASE STILL SUSPENDED | 2004-07-28 | |
| LETTER OF SUSPENSION MAILED | 2004-01-26 | |
| PAPER RECEIVED | 2003-12-30 | |
| CORRESPONDENCE RECEIVED IN LAW OFFICE | 2003-12-30 | |
| NON-FINAL ACTION MAILED | 2003-07-08 | |
| ASSIGNED TO EXAMINER | 2003-07-02 | 72505 |
uspto.report is an independent third-party trademark research tool that is not affiliated, endorsed, or sponsored by the United States Patent and Trademark Office (USPTO) or any other governmental organization. The information provided by uspto.report is based on publicly available data at the time of writing and is intended for informational purposes only.
While we strive to provide accurate and up-to-date information, we do not guarantee the accuracy, completeness, reliability, or suitability of the information displayed on this site. The use of this site is at your own risk. Any reliance you place on such information is therefore strictly at your own risk.
All official trademark data, including owner information, should be verified by visiting the official USPTO website at www.uspto.gov. This site is not intended to replace professional legal advice and should not be used as a substitute for consulting with a legal professional who is knowledgeable about trademark law.